Acinetobacter baumanniihy pothetical protein A1S-1462 protein and preparation method and application thereof
An Acinetobacter baumannii and protein technology, applied in the biological field, can solve the problems of complex components, large toxic and side effects, and achieve the effects of high expression and controllable quality and safety.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
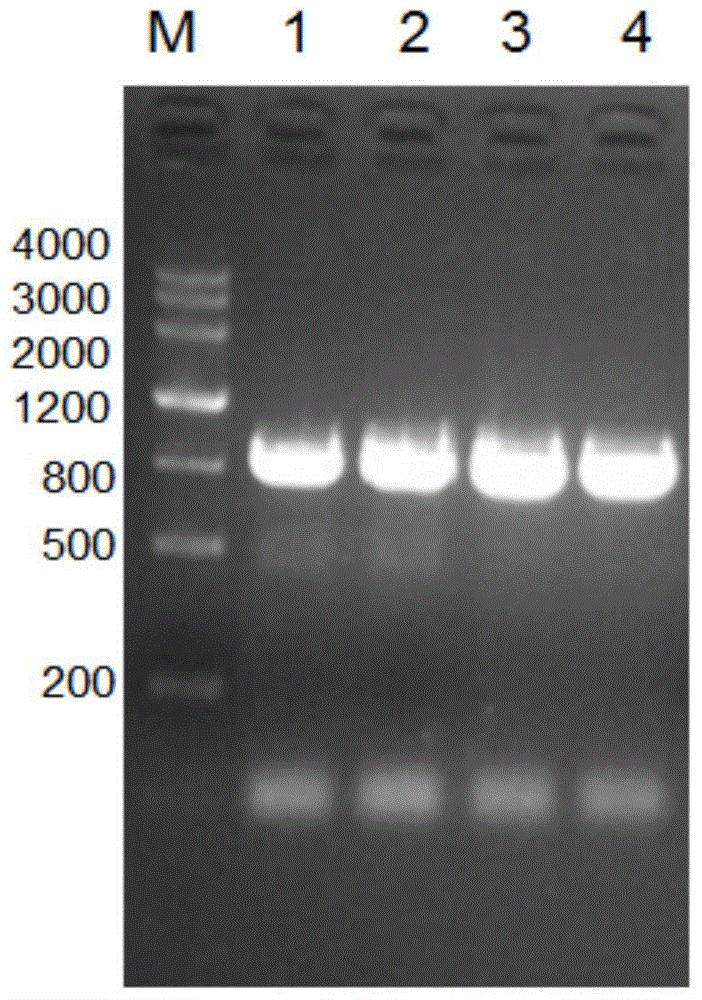
[0079] Example 1: Cloning of hypothetical proteins from Acinetobacter baumannii
[0080] 1. Firstly, according to the gene sequence of Acinetobacter baumannii 17978 standard strain A1S_1462, use bioinformatics software to conduct structural analysis. For the analysis results, see the attached Image 6 , so as to determine the gene fragment of the hypothetical protein that needs to be amplified.
[0081] 2. According to the analysis results, the PCR method was used to amplify the gene fragment of the hypothetical protein using the whole genome of Acinetobacter baumannii 17978 as a template. The amplification steps were as follows:
[0082] 1) Design the PCR primers as follows, which are SEQ ID NO: 7-8 (the base sequence of the restriction site is underlined)
[0083] Forward primer PA1S1462B1: SEQ ID NO.7
[0084] 5'-CGC GGATCC ATGGCACCTGTAAAAGAACAAAAAAT-3'
[0085] BamH Ⅰ
[0086] Reverse primer PA1S1462N2: SEQ ID NO.8
[0087] 5'-TTAT GCGGCCGC CTTACTTTTTTTGTAGCTGCG-3'...
Embodiment 2
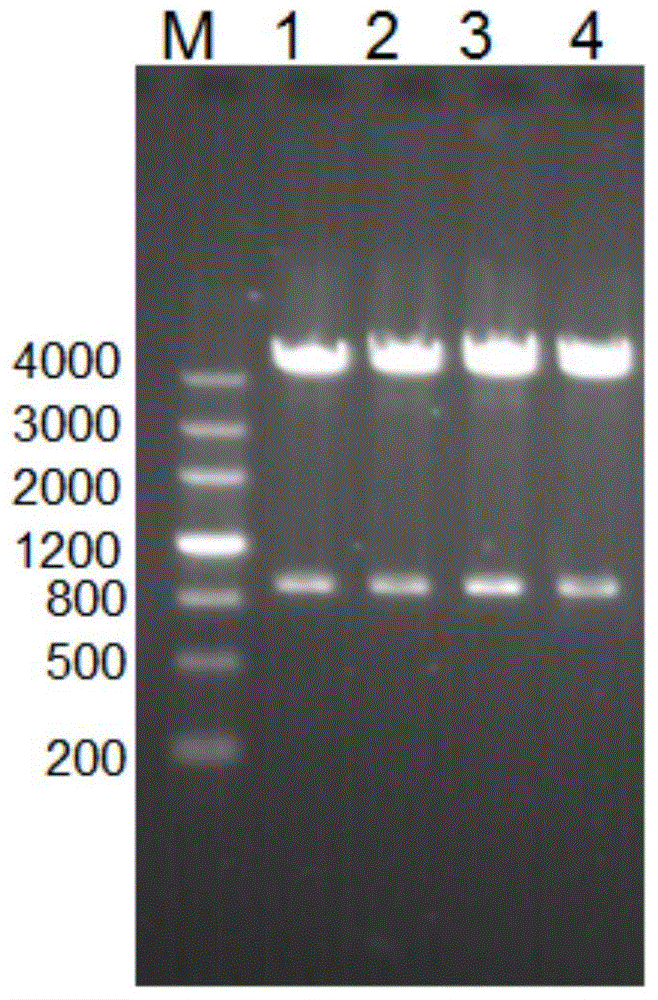
[0117] Example 2: Induced expression, purification and identification of expression form of hypothetical protein of Acinetobacter baumannii-17978 in prokaryotic expression system-Escherichia coli
[0118] 1. Induced expression of target protein
[0119] 1) Take 100 μL of the pGEX-6P-2-A1S_1462 / XL-1blue bacterial solution that was correctly identified by double enzyme digestion and add it to 10 mL of Amp-resistant TB medium, cultivate overnight at 100 rpm at 37°C, take 2 ml of the overnight cultured bacterial solution and add it to 18 mL In the Amp-resistant TB medium (the rest of the bacterial solution is stored in a refrigerator at 4°C for later use), culture at 37°C for 2-3 hours at a rotation speed of 250rpm, and when the secondary activation reaches OD600 of 0.8-1.2, add 4.4 μL of IPTG to make it The final concentration was 200 μM, and then placed on a shaker to induce expression at 30°C for 3 hours, and then induced expression at 16°C overnight.
[0120] 2) Take out the ...
Embodiment 3
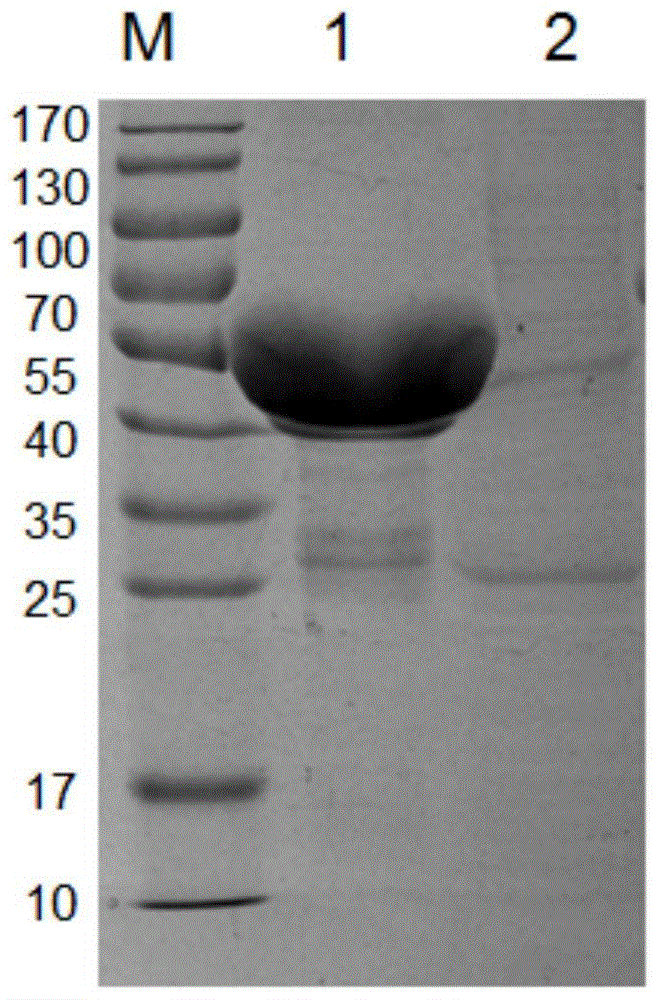
[0125] Example 3: Preparation of hypothetical protein antigens
[0126] 1. Amplify culture to obtain protein
[0127] Take 400 μL of the spare pGEX-6P-2-A1S_1462 / XL-1blue bacterial solution stored in a refrigerator at 4°C and add it to 100 mL of LB medium containing Amp resistance for one activation, culture at 200 rpm at 37°C overnight, and add to 2L containing Amp Perform secondary activation in resistant LB medium, culture at 37°C for 3-4h until OD600 is 1.2, add 420ml of IPTG (final concentration: 200uM) and induce in a shaker at 30°C for 3h, then centrifuge at 6000rpm for 5min to collect the bacteria , after adding 80ml of PBS to resuspend the bacterial cells, the bacterial solution was ultrasonically lysed for 30 minutes, and the supernatant was collected by centrifugation as above and combined with 4ml of glutathione-Sepharose 4B; then SDS-PAGE gel electrophoresis was performed, and the results are shown in Figure 4 , it can be seen that a large number of hypothetical...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More