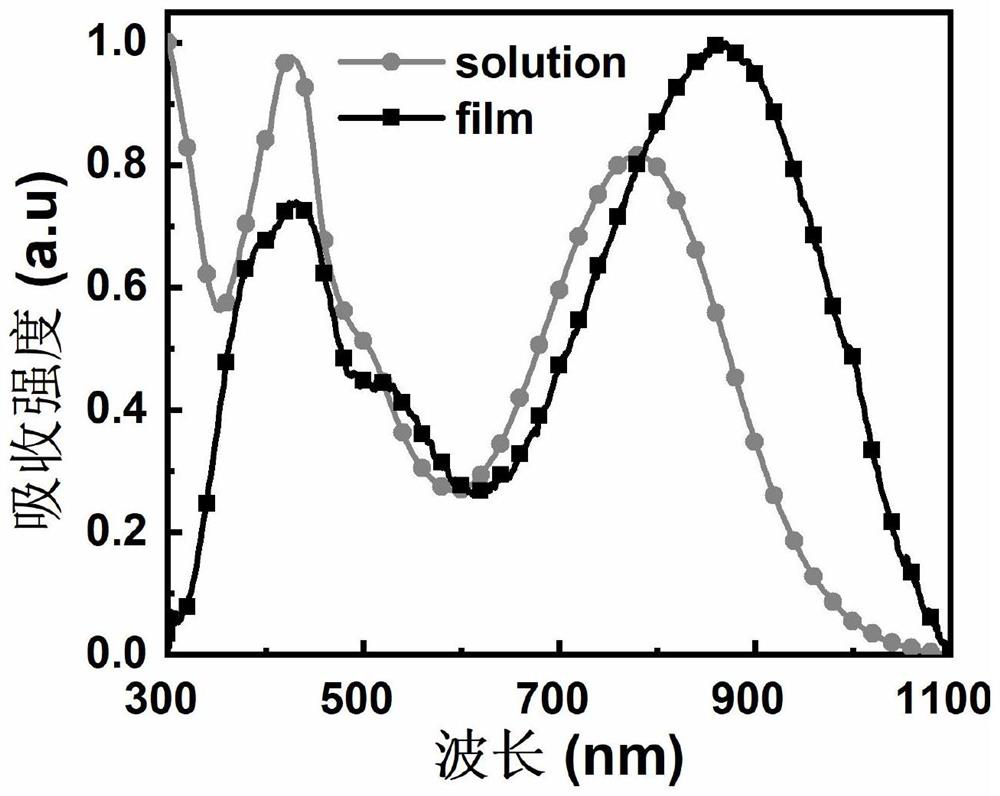
Photoelectric and photothermal conversion material containing condensed ring electron withdrawing end group and preparation method and application thereof
A light-to-heat conversion material and electron-absorbing technology, applied in circuits, photovoltaic power generation, electrical components, etc., to achieve the effects of easy purification, high yield, and low polarity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0046]
[0047] (1) Add 3-bromofluoren-9-one (100mg, 0.39mmol), 5,6-dichloro-3-(dicyanomethylene)inden-1-one (113mg, 0.43mmol) and solvent tetrahydrofuran (15mL), after stirring evenly, add 0.2ml of pyridine and 0.2ml of titanium tetrachloride solution, and heat to reflux at 60°C for 1h. After the reaction was completed, it was cooled to room temperature, quenched by adding ice water, extracted three times with dichloromethane, collected the organic layer, and removed the solvent by rotary evaporation. 1 vol / vol) was used as the eluent and dried in vacuo to obtain 153 mg of fused-ring electron-withdrawing terminal compound A1 (red solid, yield 77.6%). The proton spectrum result of this product is: 1H-NMR (CDCl 3 ,500MHz)δ8.60(d,J=8.0Hz,1H),8.46(d,J=8.4Hz,1H),8.01(d,J=1.6Hz,1H),7.65(m,2H),7.48( m,2H), 7.43–7.39(m,2H).
[0048] (2) The fused-ring electron-withdrawing terminal compound A1 (82mg, 0.162mmol), 6,6,12,12-tetrakis(4-hexylphenyl)-6,12-dihydrodithiophene[2,3-d :...
Embodiment 2
[0058]
[0059] (1) Add 3-bromofluoren-9-one (280mg, 1.08mmol), 5,6-difluoro-3-(dicyanomethylene)inden-1-one (300mg, 1.29mmol) and solvent tetrahydrofuran (40mL), after stirring evenly, add 0.6ml of pyridine and 0.6ml of titanium tetrachloride solution, and heat to reflux at 70°C for 2h. After the reaction was completed, it was cooled to room temperature, quenched by adding ice water, extracted three times with dichloromethane, collected the organic layer, and removed the solvent by rotary evaporation. 1 vol / vol) was used as the eluent, and vacuum-dried to obtain 480 mg of fused-ring electron-withdrawing terminal compound A2 (red solid, yield 84.2%). The proton spectrum result of this product is: 1 H-NMR (CDCl 3 ,500MHz)δ8.47(d,J=8.4Hz,1H),8.40(m,1H),7.75(m,1H),7.68(m,1H),7.62(d,J=1.9Hz,1H), 7.49(m,1H),7.44–7.38(m,2H),7.25–7.23(m,1H).
[0060] (2) The fused-ring electron-withdrawing terminal compound A2 (38mg, 0.081mmol), 6,6,12,12-tetrakis(4-hexylphenyl)-6,12-dihydrodi...
Embodiment 3
[0063]
[0064] The fused ring electron-withdrawing terminal compound A2 (97mg, 0.206mmol), 4,4,9,9-tetraoctyl-4,9-dihydro-s-indolo[1,2-b:5,6 -b']dithiophene-2,7-bistrimethyltin (IDT-Sn) (80mSg, 0.086mmol), catalyst trisdibenzylideneacetone dipalladium (7.8mg, 0.0086mmol), ligand tri(o Methylphenyl)phosphine (8.1mg, 0.0258mmol) and solvent toluene (16mL) were added into the reaction vessel, stirred evenly at room temperature under nitrogen protection, and then heated to 110°C for 12h. After the reaction was completed, it was cooled to room temperature, extracted three times with dichloromethane, the organic layer was collected, and the solvent was removed by rotary evaporation. The solvent was removed and vacuum-dried to obtain 62 mg of photoelectric and photothermal conversion material LY3 (dark solid, yield 52.1%).
[0065] Example 4:
PUM
| Property | Measurement | Unit |
|---|---|---|
| thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More