Lipase mutant with improved heat stability, and construction method thereof
A technology for thermostability and lipase, applied in the field of lipase mutants and their construction, can solve the problems of limited application scope, poor thermostability, restricting the development and application of lipase, etc., achieving broad market prospects and improving thermostability , the effect of high practical application value
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0039] Embodiment 1 Cloning of wild-type Candida antarctica lipase B gene
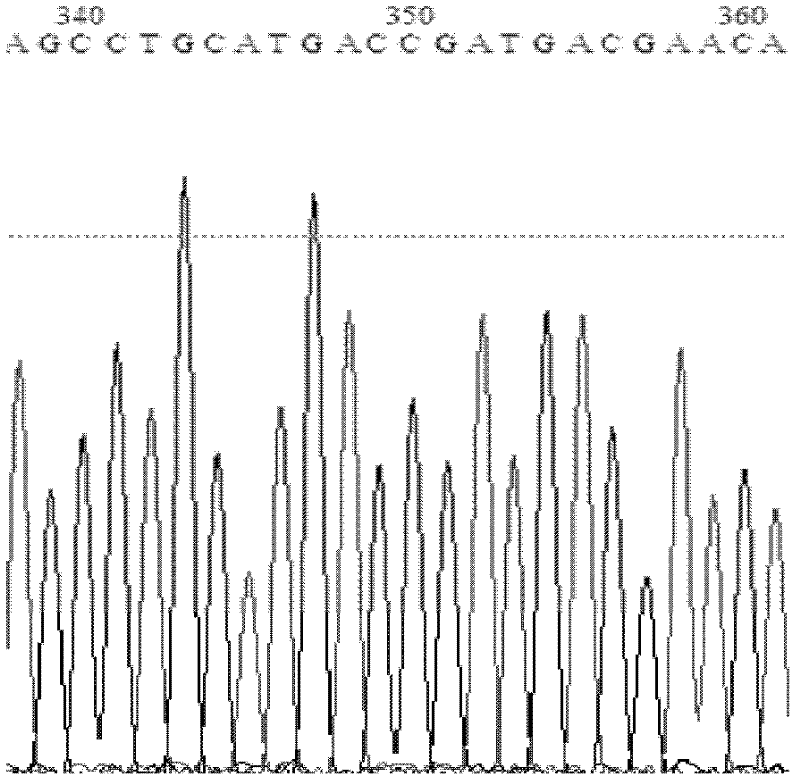
[0040] The wild-type Candida antarctica lipase B gene was synthesized by Nanjing GenScript, through the upstream primer 5'-AATA CCATGG CTCTACCTTCCGGTTCG-3' (underlined base is the recognition site of restriction endonuclease NcoI) and downstream primer 5'-TAA CTCGAG GGGGGTGACGATGCCGGA-3' (the underlined base is the restriction endonuclease XhoI recognition site) to amplify the target gene. The PCR reaction uses Takara's PrimeSTAR polymerase. The PCR reaction conditions are: 98°C for 2min, then 98°C for 10sec, 55°C 15sec at ℃, 1min at 72℃, a total of 25 cycles; the last 10min at 72℃. After the reaction, the PCR amplification product was detected by 1% agarose gel electrophoresis, and a band with a size of 1 kb was obtained, which was consistent with the expected result. DpnI digests the template, recovers, and purifies the target fragment. It is double-digested with restriction enzymes NcoI and XhoI ...
Embodiment 2
[0041] Example 2 Structural Analysis and Hot Spot Determination of Candida Antarctica Lipase B
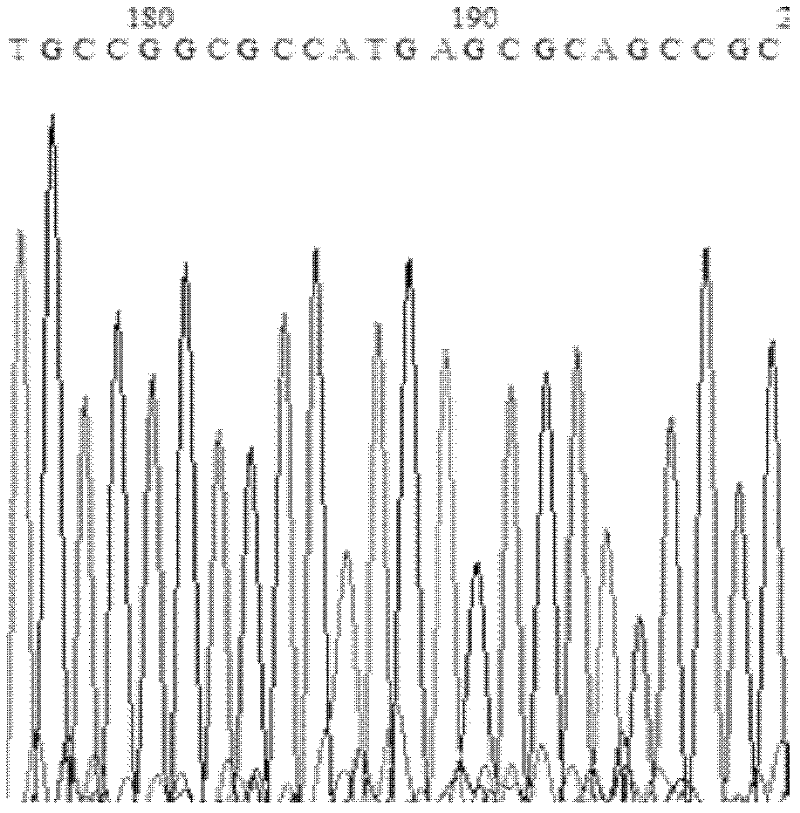
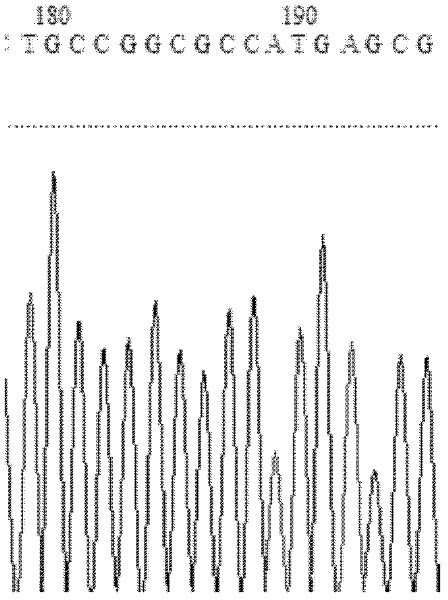
[0042] The crystal structure of Candida antarctica lipase B (ID: 1TCA) was analyzed. The catalytic triad of lipase is: serine at the 104th position from the N-terminus, aspartic acid at the 187th position, and histidine. Select the amino acid residues around the catalytic serine to analyze the B factor, and determine a series of higher residues of the B factor (from the N-terminus, the 278th leucine, the 285th valine, the 277th leucine, Glycine at position 281, aspartic acid at position 223) as a hotspot for saturation mutation.
Embodiment 3
[0043] Example 3 Establishment of Saturation Mutant Library and Screening of Mutants
[0044] Use the following method to screen the variant of Candida antarctica lipase B with better thermal stability, and the specific process includes the following steps:
[0045] 1. Construct a saturated mutant library according to hotspots and import it into host cells
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More