[0004]
Ammonia nitrogen refers to the free ammonia (NH 3 ) and
ammonium ions (NH 4 +) The nitrogen in the form of nitrogen mainly comes from the
decomposition of organic nitrogen and the reduction of
nitrate nitrogen and nitrite nitrogen.
Ammonia nitrogen has a great
impact on
human health and the
ecological environment: ammonia nitrogen in water is converted into nitrite under certain conditions. Drinking, nitrite will combine with
protein to form strong carcinogenic nitrosamines, which is extremely harmful to
human health; the harm of ammonia nitrogen to
aquatic organisms mainly includes reduced
food intake, slowed growth,
tissue damage, reduced
oxygen transport between tissues, It can lead to hyperactivity of
aquatic organisms, loss of balance in the water, convulsions, and even death in severe cases. The fish among them are sensitive to ammonia nitrogen. When the ammonia nitrogen content is high, the fish will die; ammonia nitrogen is very polluting to the marine environment. Can not be ignored; Therefore, the ammonia nitrogen concentration value is one of the important
water quality monitoring indicators of close attention in
aquaculture nursery and culture, and the mensuration of the ammonia nitrogen concentration in the
mariculture water body is the important foundation of
mariculture production management and scientific research work; In the prior art, The methods for determining the concentration of ammonia nitrogen mainly include Nessler's
reagent colorimetric method,
ion chromatography,
gas phase molecular absorption method,
indophenol blue
spectrophotometry and
hypobromite oxidation method, wherein Nessler's
reagent colorimetric method utilizes mercuric
iodide and The alkaline solution of
potassium iodide reacts with ammonia to form a light reddish brown colloidal compound. The
chromaticity is proportional to the concentration of ammonia nitrogen. The concentration of ammonia nitrogen is calculated after measuring the
absorbance in the
wavelength range of 410-425nm. However, due to the large amount of
calcium and
Magnesium ions, while
calcium and
magnesium ions react with Nessler's
reagent, resulting in
turbidity of the
water sample, which interferes with the determination accuracy of the ammonia nitrogen concentration value, and is not suitable for the determination of the ammonia nitrogen concentration value in
seawater; the
ion chromatography and
gas phase molecular absorption method used The
measuring instrument has the problems of high purchase price and high maintenance cost, which is not convenient for popularization and use; the
indophenol blue spectrophotometric method has a long reaction time and is not suitable for the rapid determination of ammonia nitrogen value in
seawater, and its use is limited; the
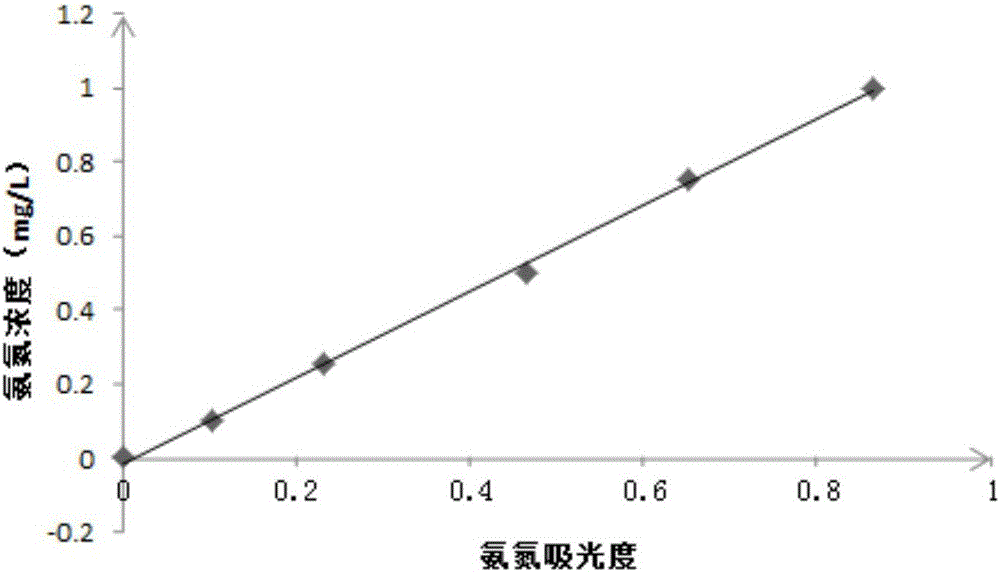
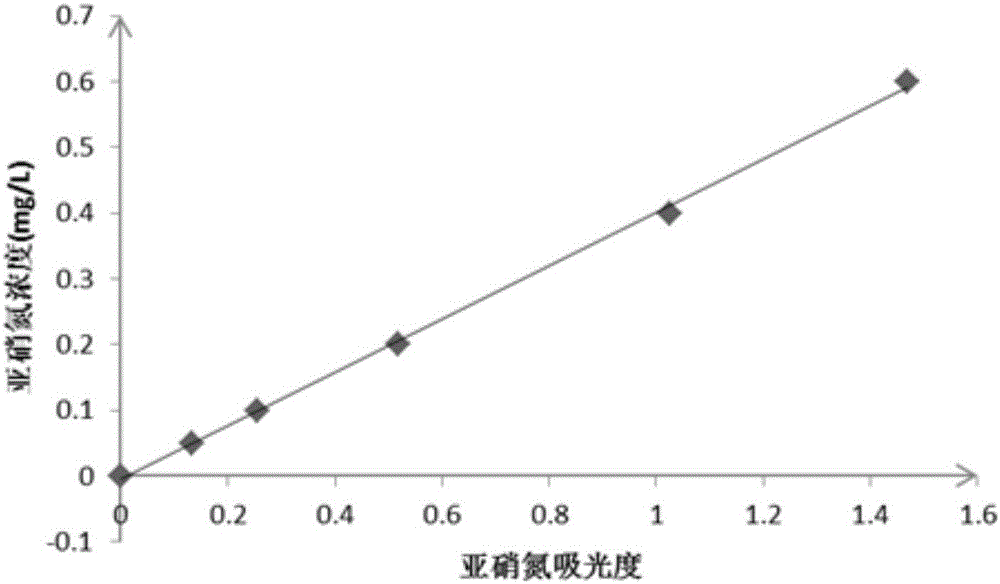
hypobromite oxidation method There is no highly toxic substance in the determination process, and the determination is fast and convenient. In the
national standard, the total concentration of ammonia nitrogen and nitrite nitrogen is measured by the
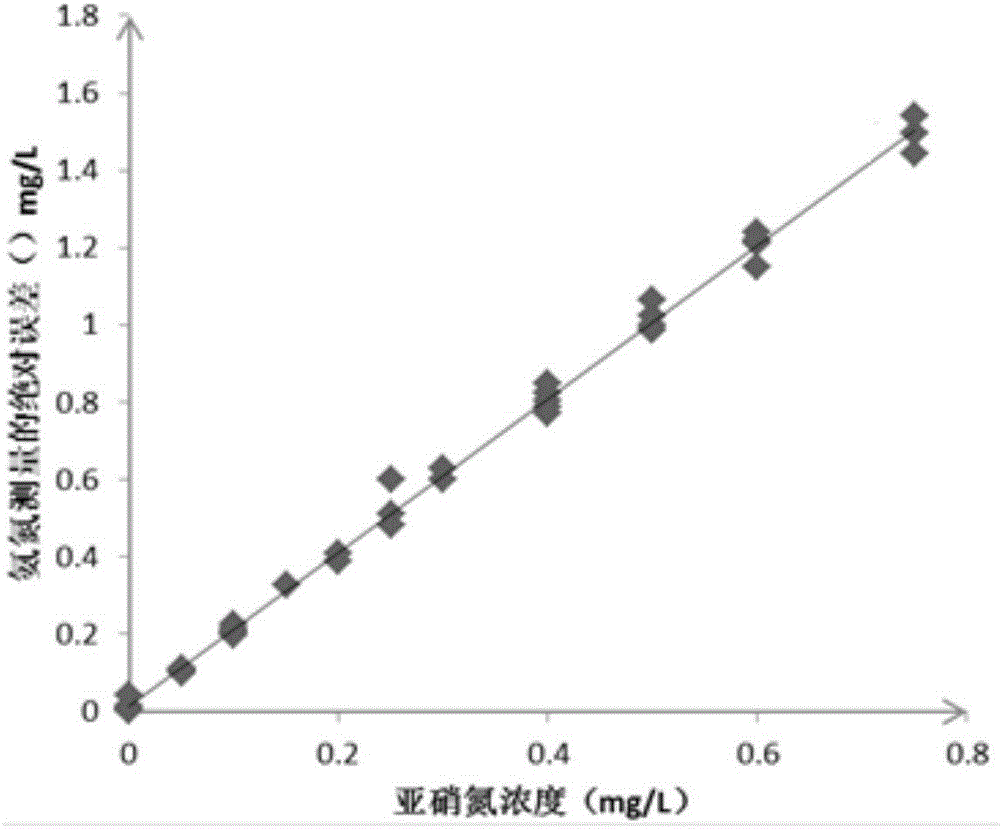
hypobromite oxidation method, and then the concentration of nitrite nitrogen is deducted to obtain the determination result of ammonia nitrogen. Experiments have confirmed that in the presence of nitrite nitrogen, when using the hypobromite oxidation method to measure the ammonia nitrogen value of water, the measured value of ammonia nitrogen is quite different from the actual value, and there is a non-negligible error, even in the presence of lower concentrations of nitrite nitrogen In the case of nitrite nitrogen, there are still non-negligible errors. The higher the concentration of nitrite nitrogen, the greater the difference between the measured value and the actual value of ammonia nitrogen concentration, and the concentration of nitrite nitrogen is linearly positively correlated with the absolute error of ammonia nitrogen concentration measurement; Therefore, in water bodies where nitrite nitrogen exists, it is difficult to accurately measure the concentration of ammonia nitrogen in
seawater with the traditional
national standard method. At present, there is no accurate, economical and efficient method for determining the concentration of ammonia nitrogen in seawater.
 Login to View More
Login to View More  Login to View More
Login to View More