PCR fluorescence detection kit of clostridium difficile toxin B and application of kit
A Clostridium difficile toxin and fluorescence detection technology, which is applied in the determination/testing of microorganisms, DNA/RNA fragments, recombinant DNA technology, etc., can solve the problem of unbalanced sensitivity and specificity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0081] Screening of Primers for PCR Fluorescent Detection Kit of Clostridium difficile Toxin B
[0082] 1. Sample Collection
[0083] Take diarrhea feces and send them for inspection in time. 16 samples were collected, all clinical samples were from Shenzhen Children's Hospital.
[0084] 2. Sample extraction
[0085] (1) Take internal reference fragments (2×10 4 Copes / mL) 10 μl was added to 500 μl sample treatment solution (10 mM Tris-HCl, pH=8.0; 6M guanidine hydrochloride; 25 mM NaOH; 1% TritonX-100; 1% NP-40; 1 mM EDTA);
[0086] (2) Take 100-200 mg of feces sample. If the sample is liquid, draw 0.15-0.20 ml of sample, add 500 μl of sample treatment solution, shake and mix, and treat at 100°C for 10 minutes;
[0087] (3) Centrifuge at 12,000g for 5 minutes;
[0088] (4) Take 400 μl supernatant, add 200 μl absolute ethanol, and mix well;
[0089] (5) Transfer the suspension to a spin column, centrifuge at 12,000g for 1 minute, and install the spin column into a new col...
Embodiment 2
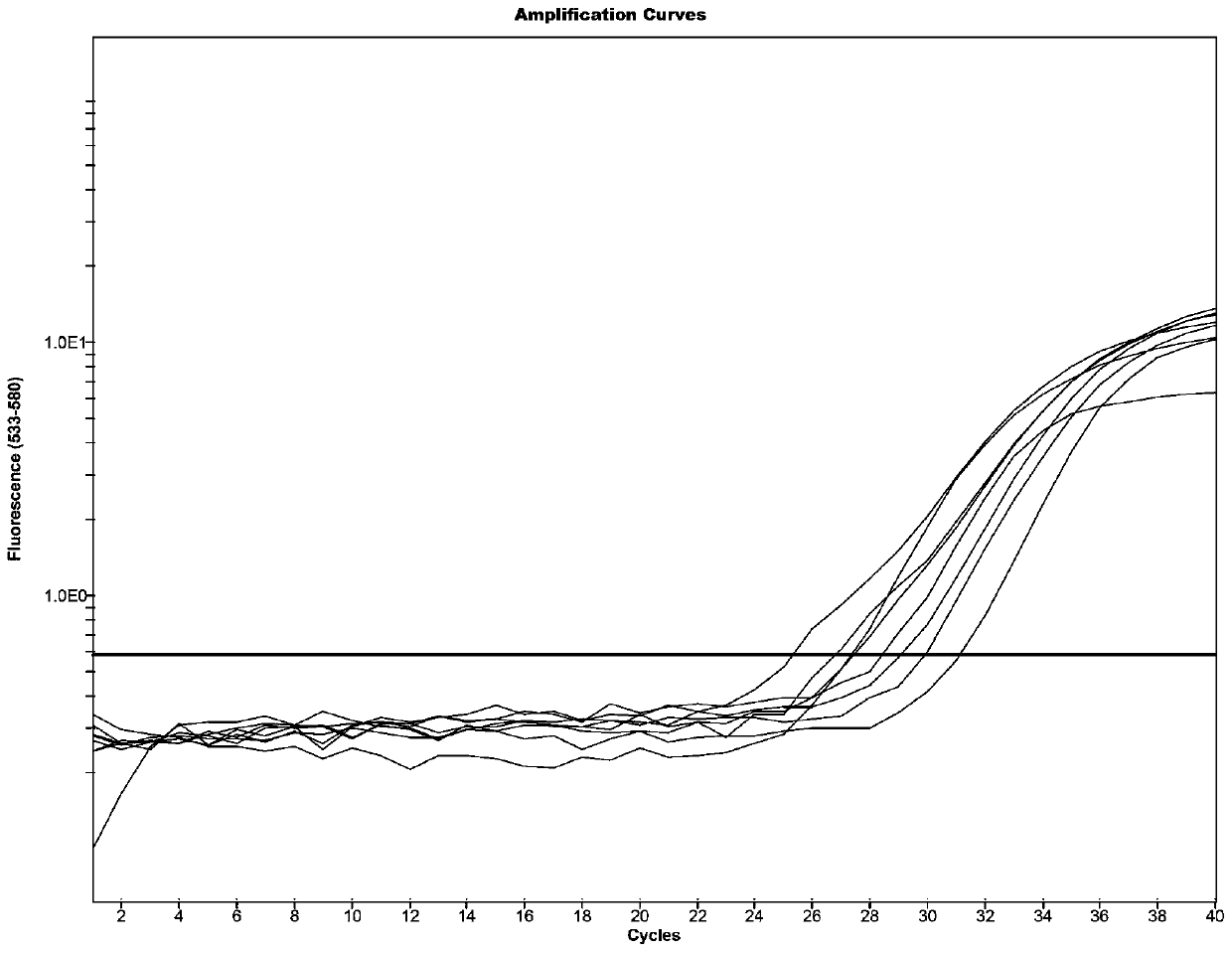
[0130] Specificity and Sensitivity Evaluation of PCR Fluorescence Detection Kit for Clostridium difficile Toxin B
[0131] 1. Sample preparation:
[0132] (1) Positive sample: Cultivate and clone the engineering bacteria of Clostridium difficile toxin B gene and extract the plasmid, measure the concentration with Qubit, then calculate its concentration, and dilute the plasmid with AE buffer to 1.0×10 5 copies / mL~1.0×10 2 copies / mL four concentration gradients.
[0133] (2) Negative samples: physiological saline and nucleic acids of Escherichia coli, Salmonella, Enterococcus faecium, and Enterococcus faecalis (confirmed by mass spectrometry and Sanger sequencing for bacteria obtained from feces culture).
[0134] 2. Sample adding system:
[0135] The PCR reaction solution includes PCR reaction solution A and PCR reaction solution B. Wherein the reaction solution A includes 12.5mM Tris-HCl, pH9.0, 50mM KCl, 0.125% X-100, 3.125mM MgCl 2 , 0.44625mM dATP, 0.44625mMdGTP, 0.4...
Embodiment 3
[0153] The PCR fluorescence detection kit of Clostridium difficile toxin B of the present invention and Comparison experiment of Clostridium difficile glutamate dehydrogenase antigen and toxin detection kit (enzyme-linked immunochromatography)
[0154] 1. Sample Collection
[0155] Take diarrhea feces and send them for inspection in time. A total of 97 samples were collected, all clinical samples were from Shenzhen Children's Hospital.
[0156] 2. Clostridium difficile glutamate dehydrogenase antigen and toxin detection kit (ELISA) (National Food and Drug Administration (Jin) Zi 2014 No. 3403929) operation process:
[0157] (1) Before use, place all reagents and corresponding number of reaction plates at room temperature.
[0158] (2) Prepare a small test tube for each sample and mark it. If necessary, it can be used as an external quality control.
[0159] (3) Take the diluent (bottle with a black dropper cap), and add 750 μL (the second scale from the tip) of the dilu...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More