However, this form of preparing and
dispensing medications has many disadvantages including that a large proportion of adjuvants that must be added to obtain a size able to be handled, that a larger medication form requires additional storage space, and that dispensing includes counting the tablets which has a tendency for inaccuracy.
While tablets may be broken into smaller pieces or even crushed as a means of overcoming
swallowing difficulties, this is not a suitable solution for many tablet or
pill forms.
For example, crushing or destroying the tablet or
pill form to facilitate
ingestion, alone or in admixture with food, may also destroy the
controlled release properties.
However, historically films and the process of making
drug delivery systems therefrom have suffered from a number of unfavorable characteristics that have not allowed them to be used in practice.
Examination of films made in accordance with the process disclosed in Fuchs, however, reveals that such films suffer from the aggregation or conglomeration of particles, i.e., self-aggregation, making them inherently non-uniform.
When large dosages are involved, a small change in the dimensions of the film would lead to a large difference in the amount of active per film.
Since sheets of film are usually
cut into unit doses, certain doses may therefore be devoid of or contain an insufficient amount of active for the recommended treatment.
Failure to achieve a high degree of accuracy with respect to the amount of
active ingredient in the
cut film can be harmful to the patient.
For this reason, dosage forms formed by processes such as Fuchs, would not likely meet the stringent standards of governmental or regulatory agencies, such as the U.S. Federal
Drug Administration ("FDA"), relating to the variation of active in dosage forms.
Schmidt specifically pointed out that the methods disclosed by Fuchs did not provide a uniform film and recognized that that the creation of a non-uniform film necessarily prevents accurate dosing, which as discussed above is especially important in the pharmaceutical area.
Moreover, his process is a multi-step process that adds expense and complexity and is not practical for commercial use.
These methods have the
disadvantage of requiring additional components, which translates to additional cost and manufacturing steps.
Furthermore, both methods employ the use the conventional time-consuming
drying methods such as a high-temperature air-bath using a
drying oven, drying tunnel, vacuum drier, or other such drying equipment.
Such processes also run the risk of exposing the active, i.e., a
drug, or
vitamin C, or other components to
prolonged exposure to
moisture and elevated temperatures, which may render it ineffective or even harmful.
In addition to the concerns associated with degradation of an active during extended
exposure to
moisture, the conventional drying methods themselves are unable to provide uniform films.
Uniformity is particularly difficult to achieve via conventional drying methods where a relatively thicker film, which is well-suited for the incorporation of a
drug active, is desired.
Thicker uniform films are more difficult to achieve because the surfaces of the film and the inner portions of the film do not experience the same external conditions simultaneously during drying.
Thus, observation of relatively thick films made from such conventional
processing shows a non-uniform structure caused by
convection and intermolecular forces and requires greater than 10%
moisture to remain flexible.
The amount of free moisture can often interfere over time with the drug leading to
potency issues and therefore inconsistency in the final product.
The difficulty in achieving a uniform film is directly related to the Theological properties and the process of water
evaporation in the film-forming composition.
The result of the repeated destruction and reformation of the film surface is observed as a "
ripple effect" which produces an uneven, and therefore non-uniform film.
Frequently, depending on the
polymer, a surface will seal so tightly that the remaining water is difficult to remove, leading to very long drying times, higher temperatures, and higher energy costs.
Air can be trapped in the composition during the mixing process or later during the
film making process, which can leave voids in the film product as the moisture evaporates during the drying stage.
The film frequently collapse around the voids resulting in an uneven film surface and therefore, non-uniformity of the final film product.
This situation also provides a non-uniform film in that the spaces, which are not uniformly distributed, are occupying area that would otherwise be occupied by the film composition.
None of the above-mentioned patents either addresses or proposes a solution to the problems caused by air that has been introduced to the film.
The absence of a uniform thickness detrimentally affects uniformity of component distribution throughout the area of a given film.
Uncontrolled air currents, either above or below the film, can create non-uniformity in the final film products.
Further, increasing the number of particles leads to a hindered
settling effect based on the solids
volume fraction.
More particles suspended in the
liquid phase results in decreased velocity.
The
rheology requirements for the inventive compositions and films are quite severe.
Such dried upper portions serve as a barrier to further vapor release as the portions beneath are dried, which results in non-uniform films.
Any top fluid flow, such as air, also must not overcome the
inherent viscosity of the film-forming composition.
Moreover, air velocities are desirably below the yield values of the film, i.e., below any
force level that can move the liquids in the film-forming compositions.
Furthermore, bottom drying also tends to result in a lower internal film temperature as compared to top drying.
Such lower internal film temperatures often result in decreased
drug degradation and decreased loss of certain
volatiles, such as flavors.
Films containing sensitive
active components, however, may face degradation problems at high temperatures.
For instance, if the
active component is a drug or bioactive material, this may adversely affect the safety or
efficacy of the final pharmaceutical product.
Additionally, highly volatile materials will tend to be quickly released from this film upon
exposure to conventional drying methods.
Moreover, temperature has a significant effect on the rate of such reactions.
Therefore, it is commonly understood that exposing an active component to high temperatures will initiate and / or accelerate undesirable degradation reactions.
Proteins from bovine extract are also known to degrade at such low temperatures.
Due to the short heat
exposure and evaporative cooling, the film components such as drag or volatile actives remain unaffected by high temperatures.
In contrast, skinning on the top surface traps liquid carrier molecules of increased energy within the film, thereby causing the temperature within the film to rise and exposing active components to high, potentially deleterious temperatures.
Second, thermal mixing occurs within the film due to bottom heating and absence of surface skinning.
Pockets of heat in the film result in the formation of particle aggregates or danger areas within the film and subsequent non-uniformity.
The formation of such aggregates or agglomerations is undesirable because it leads to non-uniform films in which the active may be randomly distributed.
Such uneven distribution may lead to large differences in the amount of active per film, which is problematic from a safety and
efficacy perspective.
Although the film surfaces may be exposed to a temperature above that at which the active component degrades, the film interior may not reach this temperature.
Moreover, such particles are desirably not fully encased or fully embedded into the film, but remain exposed to the surface of the film, such as in the case where the particles are partially embedded or partially encased.
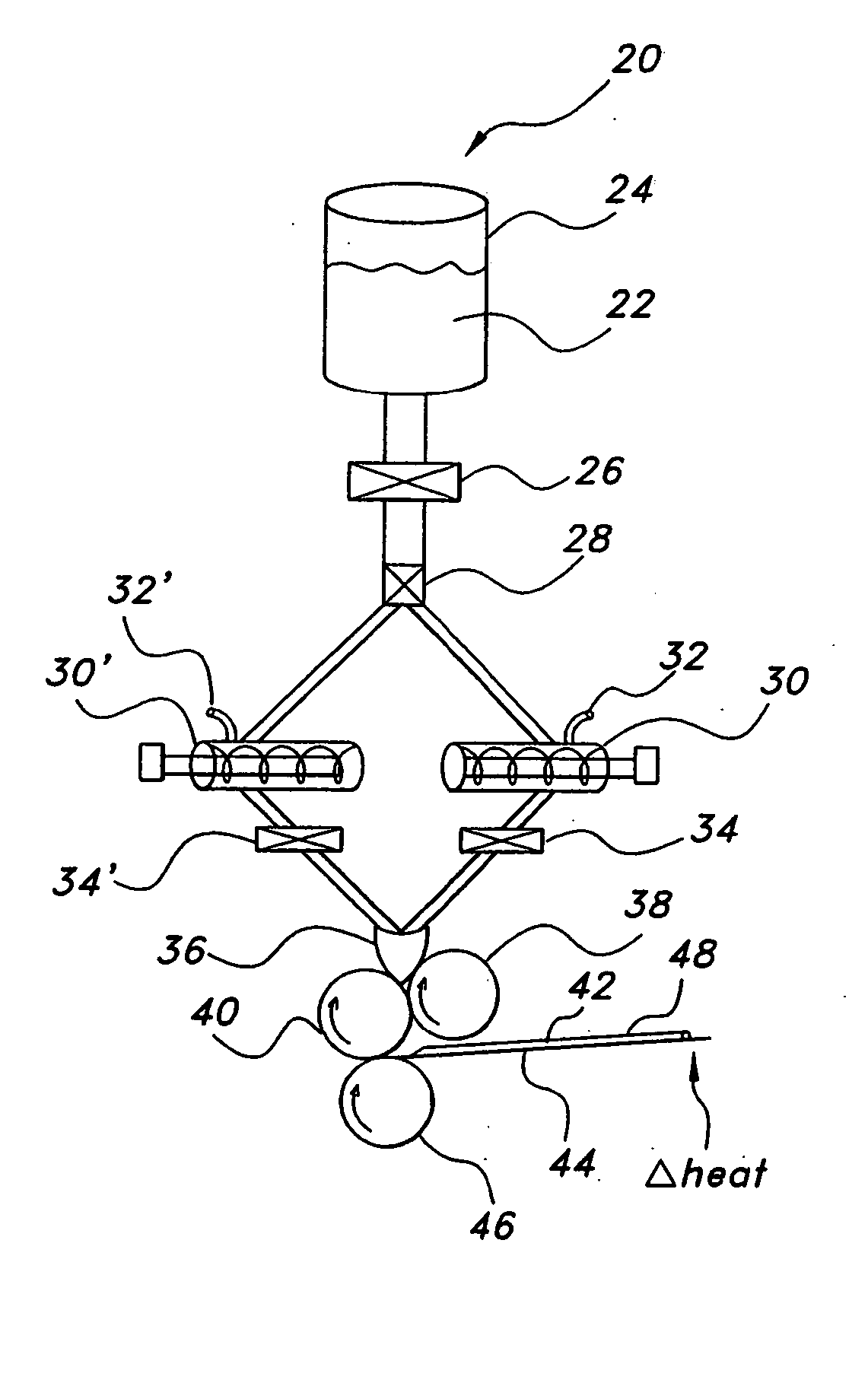
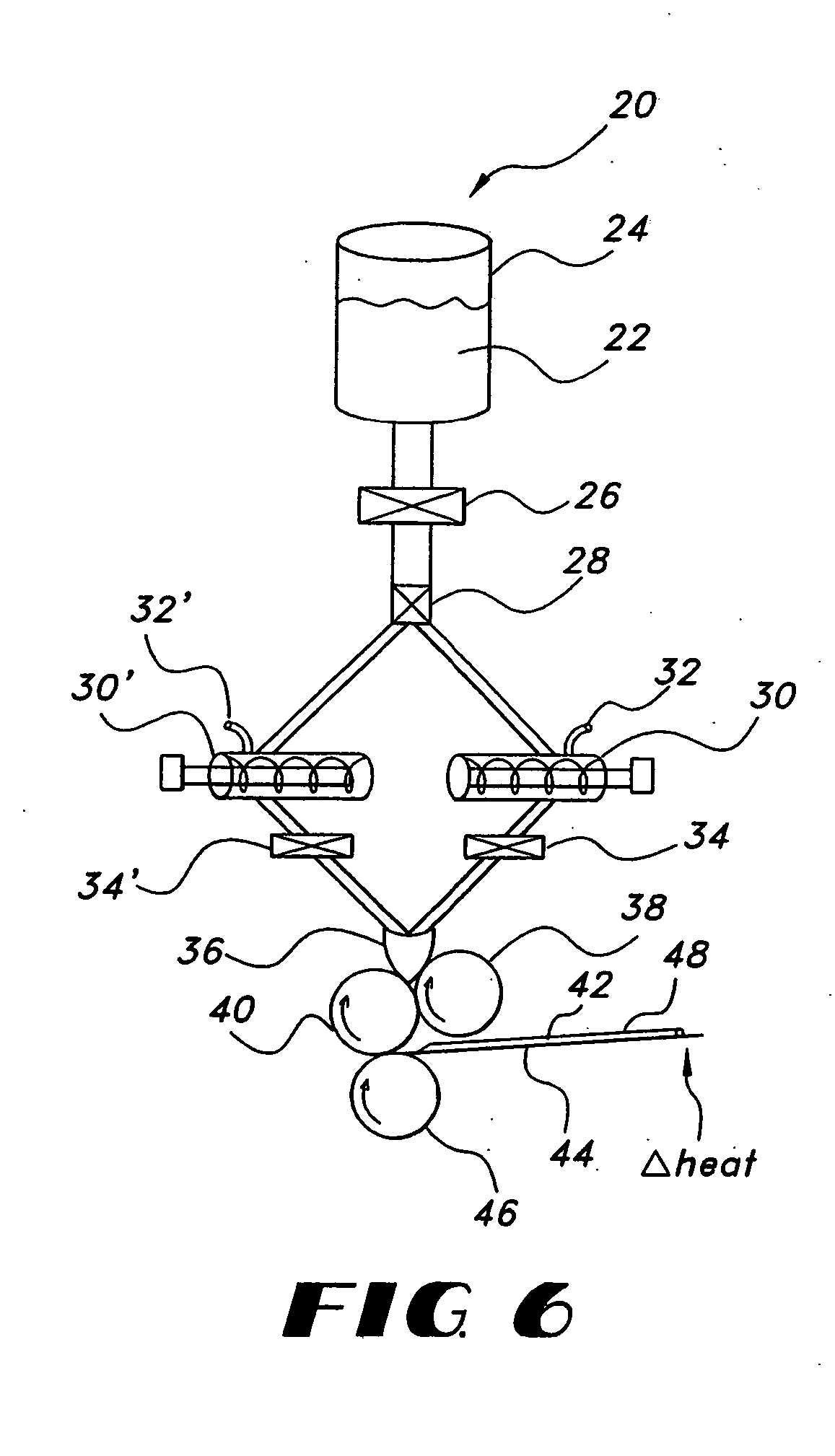
Lifting of the film 42 may not only result in non-uniformity in the film or otherwise, but may also result in non-controlled
processing of the film 42 as the film 42 and / or substrate 44 lift away from the
processing equipment.
For example, thin films are difficult to inadvertently aspirate and provide an increased
patient compliance because they need not be swallowed like a tablet.
Furthermore, thin films may not be crushed in the manner of
controlled release tablets which is a problem leading to abuse of drugs such as Oxycontin.
As described above, such entrapped air may lead to non-uniform films.
In this way,
simethicone reduces the
surface tension of bubbles air located in the solution, such as foam bubbles, causing their collapse.
 Login to View More
Login to View More