Homogeneous-phase catalytic rectification process for preparing propionic ester through ester-ester exchange
A homogeneous catalysis and propionate technology, which is applied in the preparation of carboxylate, the preparation of mutual reaction between ester groups, and the chemical industry, etc. Strict requirements and other problems, to achieve the effect of stable catalyst performance without deactivation, low cost and easy separation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] Using methyl propionate and propyl acetate as raw materials, the ratio of propyl propionate to methyl acetate is 1:1, and the excess methyl propionate is recycled.
[0042] The dosage of the catalyst KOH is 0.3-5wt% of the total mass sum of the raw materials, and the ratio of the cross-sectional area of the initial distillation section to the side line section is 1:1.
[0043] The operating conditions are as follows:
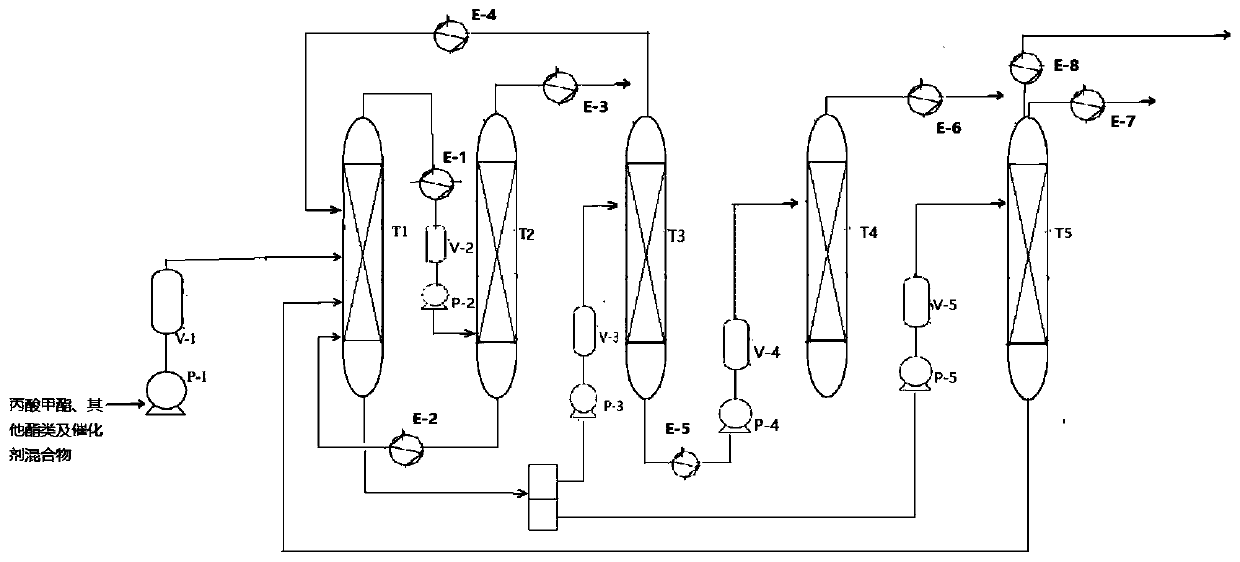
[0044] Distillation tower T1: tower diameter 1000mm; tower height 19000mm; the number of plates in the public rectification section, initial distillation section, side line section, and public stripping section are 10, 20, 30, and 15 respectively; the pressure inside the tower is 1 MPa; The temperature is 60°C; the bottom temperature of the common stripping section is 71°C; the bottom temperature of the side line section is 74°C.
[0045] Distillation tower T2: tower diameter 3000mm; tower height 40000mm; the number of trays in the public rectification...
Embodiment 2
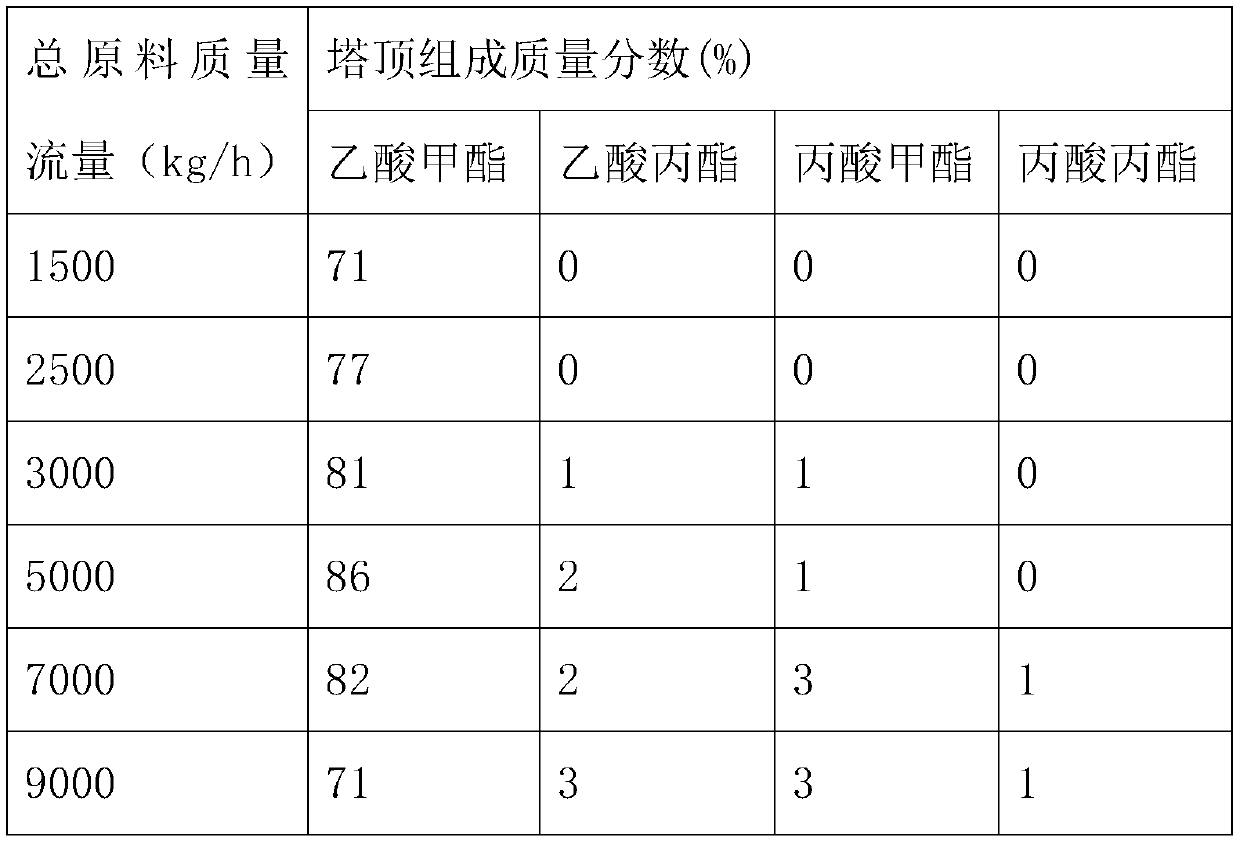
[0052] Under the operating conditions of Example 1, when the total mass flow rate of raw materials is 5000kg / h, changing the molar ratio of the raw materials in the first reactive distillation column of T1, the changes in the mass fractions of the components at the top of the tower are shown in Table 2.
[0053] Table 2 Effect of different molar ratios of raw materials on the mass fraction of each component at the top of the first reactive distillation column of T1
[0054]
[0055] It can be seen from Table 2 that with the increase of the molar ratio of raw materials, the mass fraction of methyl acetate at the top of the tower gradually decreases, and the mass fraction of ethyl propyl ester gradually increases. When the molar ratio of propyl acetate and methyl propionate is 1:2 is the best raw material ratio.
Embodiment 3
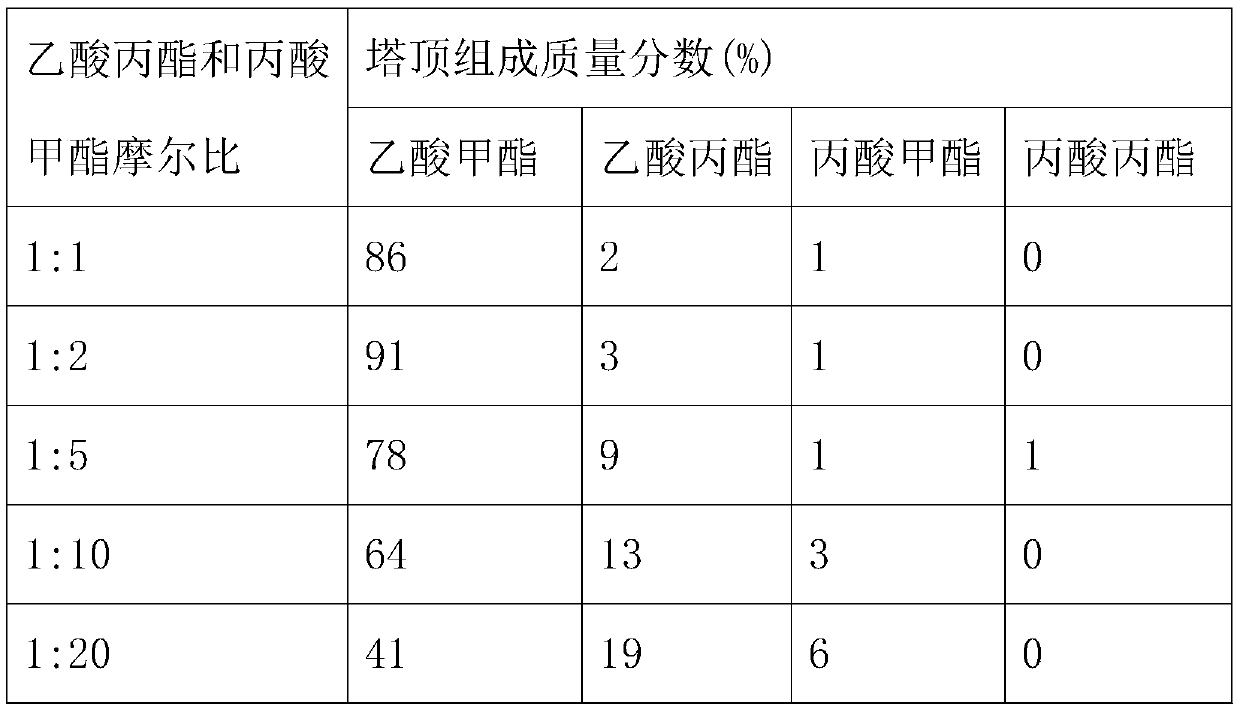
[0057] Under the operating conditions of Example 2, the total raw material mass flow rate is 5000kg / h, and when the molar ratio of propyl acetate and methyl propionate is 1:2, the reflux ratio of the first reactive distillation column of T1 is changed, and the components at the top of the column The quality fraction changes are shown in Table 3.
[0058] Table 3 Effects of different reflux ratios on the mass fraction of each component at the top of the first reactive distillation column of T1
[0059]
[0060]
[0061] As can be seen from Table 3, along with the increase of reflux ratio, the mass fraction of methyl acetate at the top of the tower increases gradually, and the content of methyl propionate, propyl propionate and propyl acetate decreases, and the increase of visible reflux ratio contributes to more The methyl acetate, the light component at the top of the tower, returns to the tower to participate in the reaction again to increase the conversion rate of the ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More