Staphylococcal immunotherapeutics via donor selection and donor stimulation
a staphylococcal and immunotherapy technology, applied in the field of biological products for the treatment, prevention and diagnosis of bacterial infections, can solve the problems of more serious invasive infections, difficult to eradicate rapid progressive and highly destructive joint diseases, and serious medical problems of bacterial arthritis acquired by the patient, so as to improve the rate of opsonization and phagocytosis, and enhance the intracellular killing of staphylococcus bacteria
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
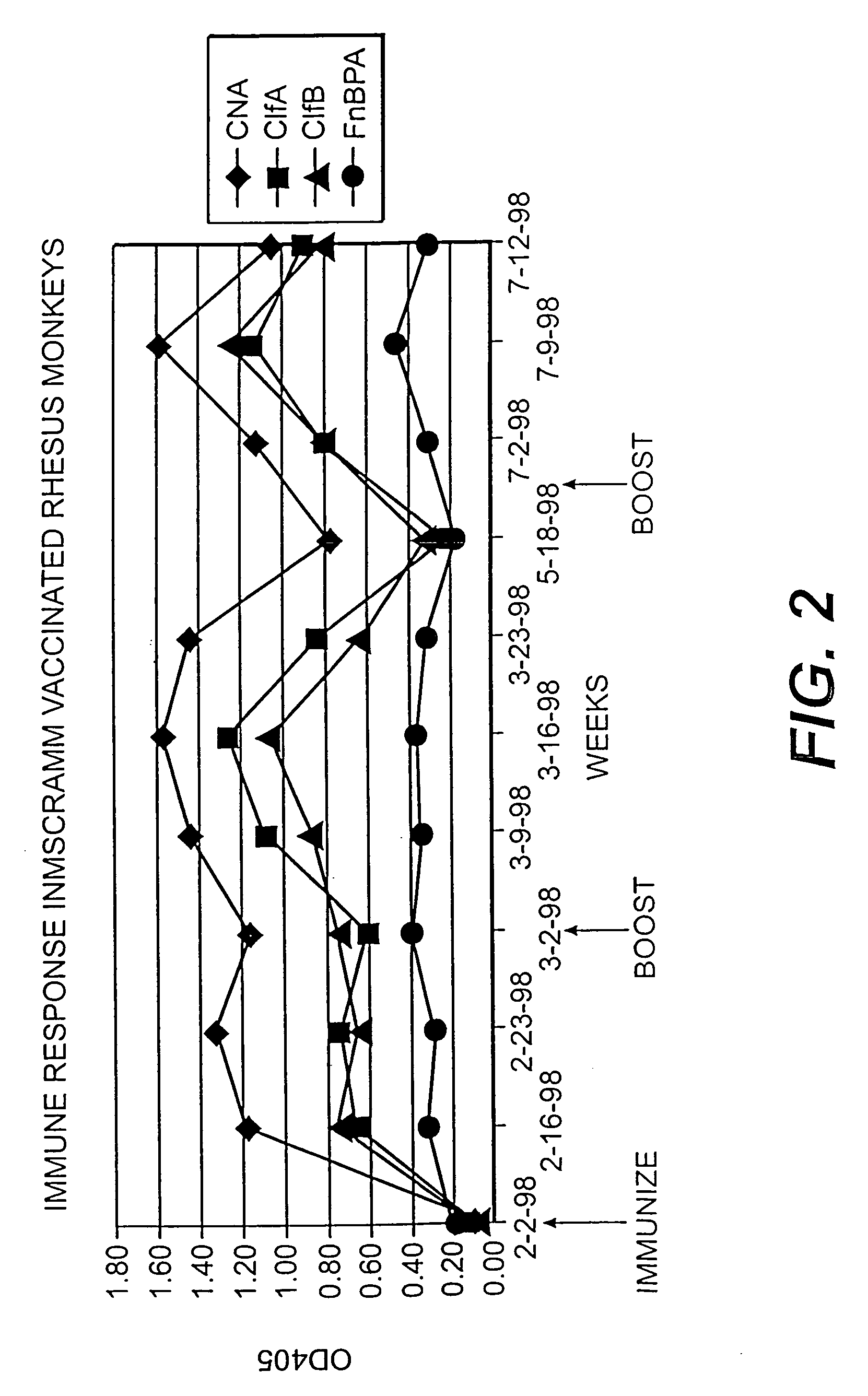
Preparation of Prototype Four Component MSCRAMM Vaccine
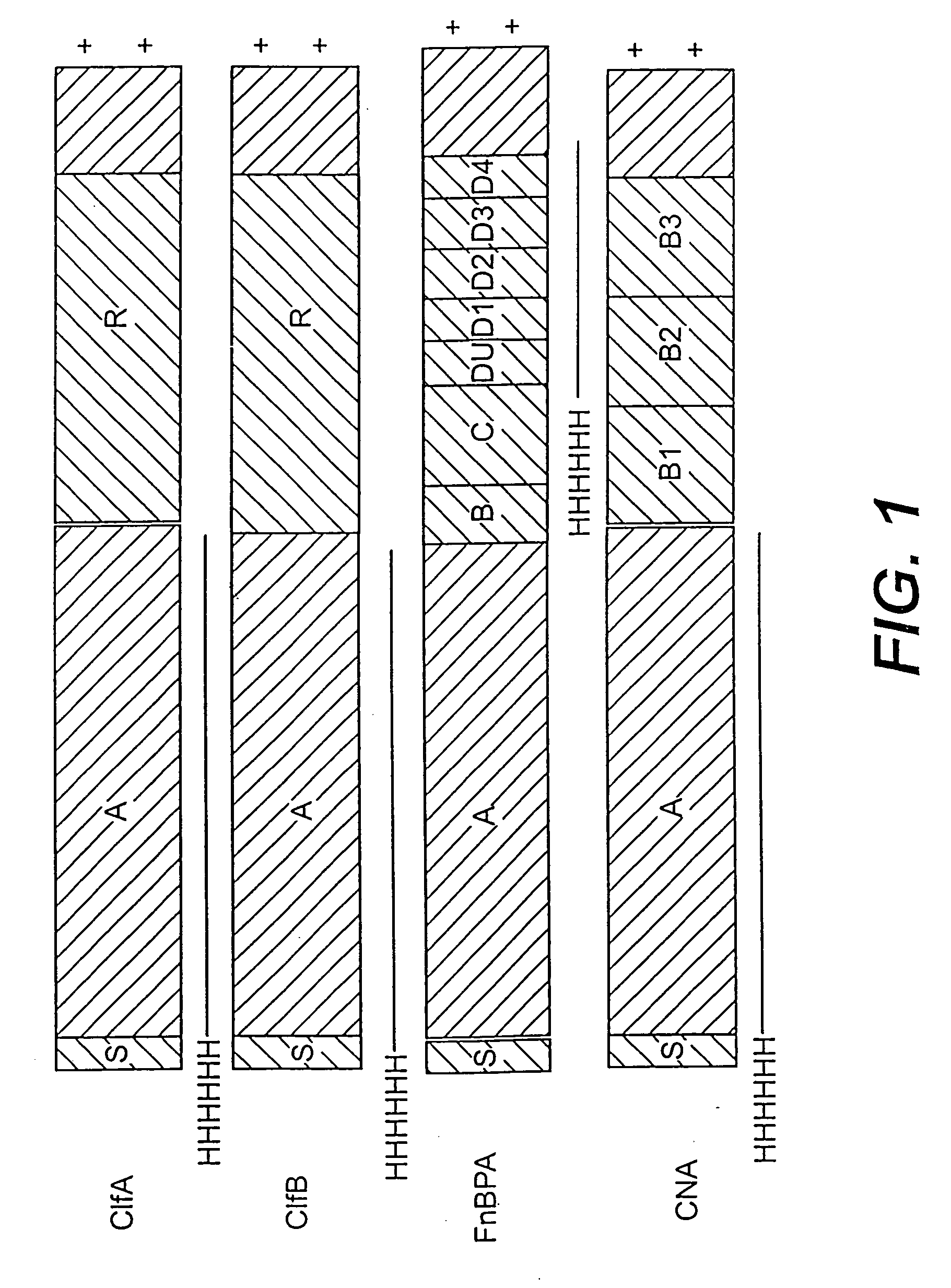
[0226] A series of recombinant proteins, representing domains from the collagen, Fn, and Fbg-binding MSCRAMMs (FIG. 1), were overexpressed in E. coli and affinity purified by metal chelating chromatography as previously described (see, e.g., Job et al., Biochemistry. 33 (20):6086-6092, 1994; Patti et al., J. Biol. Chem. 270, 12005-12011, 1995; McDevitt et al., Mol. Micro. 11 (2):237-248, 1994; Ni Eidhin et al., Infect. Immun. Submitted, 1998). Used were the following: amino acids contained in the recombinant collagen-binding MSCRAMM expressed from CNA (M55, such as disclosed in co-pending U.S. patent application Ser. No. 08 / 856,253, incorporated herein by reference); amino acids contained in the recombinant fibrinogen-binding MSCRAMM-expressed from clfA (Region A, such as disclosed in U.S. patent application Ser. No. 08 / 293,728, incorporated herein by reference); amino acids contained in the recombinant fibrinogen-binding MSCRA...
example 2
Example of Growing E. coli Strains for Production of Recombinant Proteins
[0227] Overnight cultures of E. coli JM101 or TOP 3 cells (Stratagene) harboring the recombinant plasmids were diluted 1:50 in 1 L of Luria Broth (Gibco BRL) containing 50 mg / mL ampicillin. E. coli cells were grown until the culture reached an OD600 of 0.5-0.8. Expression of the recombinant proteins was induced by adding IPTG to a final concentration of 0.2 mM. After a three hour induction period, cells were collected by centrifugation, resuspended in 15 mL of Buffer A (5 mM imidazole, 0.5 M NaCl, 20 mM Tris-HCl, pH 7.9) and lysed by passage through a French press twice at 20,000 lb. / in2. Cell debris was removed by centrifugation at 50,000×g for 10 min and the supernatant was passed through a 0.45 μM filter.
example 3
Purification of HIS6 Containing Recombinant Proteins Expressed from pQE-30 (Qiagen®; Qiagen Inc., Chatsworth, Calif.) or PV-4 Based Recombinant Plasmids
[0228] The recombinant proteins were purified by immobilized metal chelate chromatography, using a column of iminodiacetic acid / Sepharose® 6B Fast Flow (Sigma, St. Louis, Mo.) charged with Ni2+; (Porath et al. 1975; Hochuli et al. 1988). The HIS6 tagged proteins were purified by immobilized metal chelate affinity chromatography. More specifically, a column containing iminodiacetic acid Sepharose® 6B FF, connected to a FPLC® system (Pharmacia), was charged with 150 mM Ni++ and equilibrated with buffer A (5 mM imidazole, 0.5 M NaCl, 20 mM Tris, pH 7.9). After equilibration, the bacterial supernatant was applied to the column and the column was washed with 10 bed volumes of buffer A. Subsequently, the column was eluted with buffer B (200 mM imidazole, 0.5 M NaCl, 20 mM Tris, pH 7.9). The eluate was monitored for protein by the absorban...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time | aaaaa | aaaaa |
| molecular weights | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More