Therapeutic encapsulated embryonic stem cells and mesenchymal stromal cells
a technology of embryonic stem cells and mesenchymal stromal cells, which is applied in the direction of biocide, drug composition, skeletal/connective tissue cells, etc., can solve the problems of msc tissue persistence, potential msc differentiation and/or msc migration away from the injury site, and is difficult to resolve, control and quantify. , to achieve the effect of reducing the inflammatory aspects of trauma-mediated tissue damage, promoting tissue repair, and reducing the inflammatory aspects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
ES Cell Culture
[0132]All cell cultures were incubated in a humidified 37° C., 5% CO2 environment. The ES cell line D3 (ATCC, Manassas, Va.) was maintained in an undifferentiated state in T-75 gelatin-coated flasks (Biocoat, BD-Biosciences, Bedford, Mass.) in Knockout Dulbecco's modified Eagles medium (Gibco, Grand Island, N.Y.) containing 15% knockout serum (Gibco), 4 mM L-glutamine (Gibco), 100 U / ml penicillin (Gibco), 100 U / ml streptomycin (Gibco), 10 mg / ml gentamicin (Gibco), 1,000 U / ml ESGRO™ (Chemicon, Temecula, Calif.) and 0.1 mM 2-mercaptoethanol (Sigma-Aldrich, St. Louis, Mo.). Media was changed every two days until plates were confluent. ES cultures were split and passaged every 6 days. Following media aspiration, cells were washed with 10 mL of phosphate buffered solution (PBS) (Gibco), detached using 3 mL of trypsin EDTA (Gibco) for 3 minutes, and subsequently 12 mL of Knockout DMEM was added. Cells were then replated in gelatin-coated T-75 flasks at a density of 1 millio...
example 2
Alginate Encapsulation
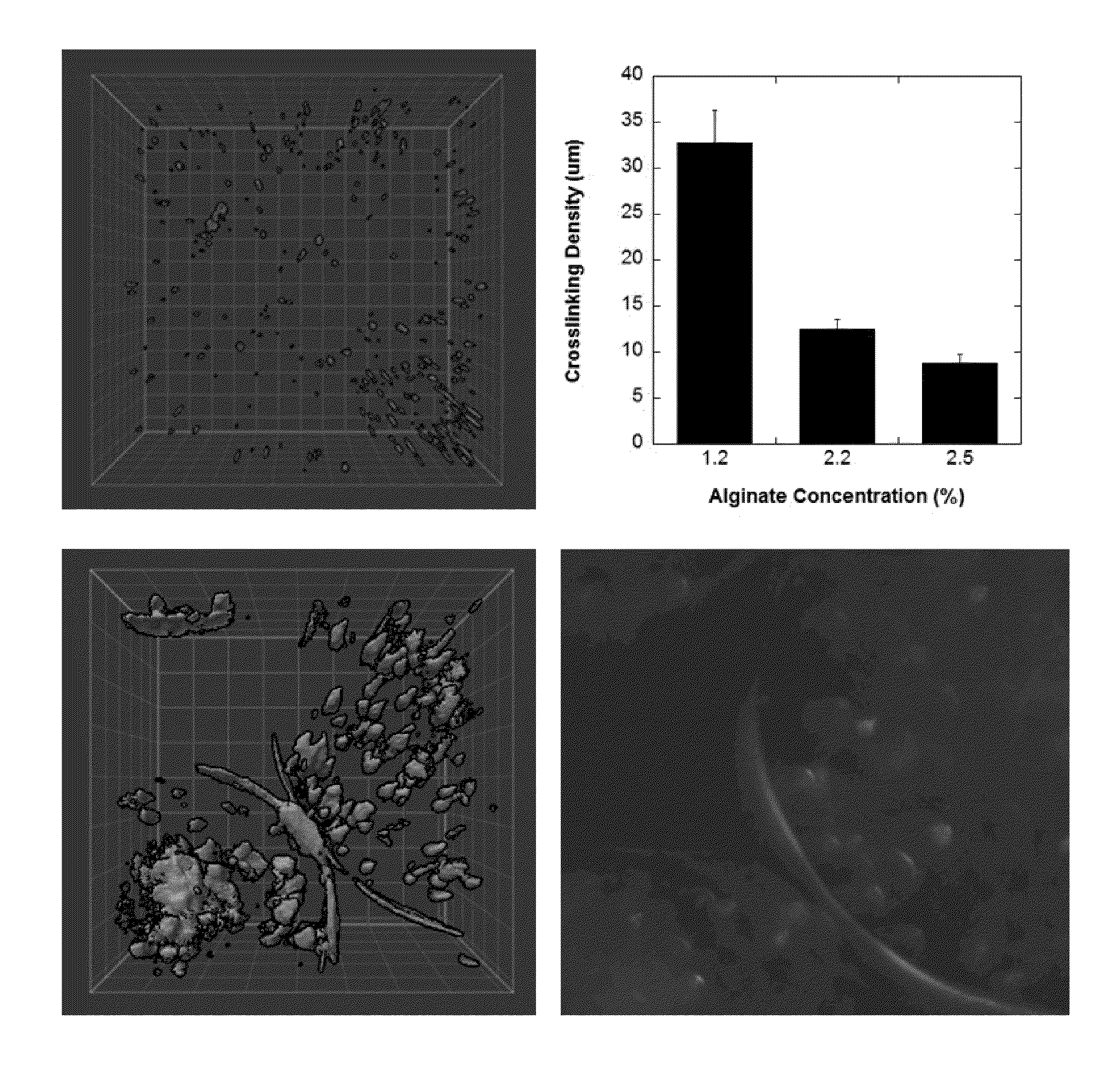
[0133]Alginate solution was prepared by dissolving 2.2 g of alginic acid sodium salt (MW: 100,000-200,000 g / mol, G Content: 65%-70%, Sigma-Aldrich) in 100 mL of Ca2+ free DMEM (Gibco), using a heated magnetic stir plate at a temperature of 65° C. The solution was then filtered using a 45 μm syringe filter (Fisher Scientific, Pittsburgh, Pa.). To create the cell-alginate mixture, 1 mL aliquot of cell suspension with a seeding density of 5×107 cells / mL was added to 9 mL of either 1.2%, 1.7%, 2.2% or 2.5% (w / v) alginate solution to yield a final cell seeding density of 5×106 cells / mL. This solution was transferred to a 10 mL syringe, and was connected to a syringe pump (KD Scientific, MA). Alginate beads were generated using an electrostatic bead generator (Nisco, Zurich, Switzerland) at a flow rate of 40 mL / h, and an applied voltage of 6.4 kV. The beads were extruded into a 200 mL bath of CaCl2 (100 mM), containing 145 mM NaCl, and 10 mM MOPS (all from Sigma-Aldr...
example 3
Assessment of Cell Proliferation and Neural Specific Protein Expressions
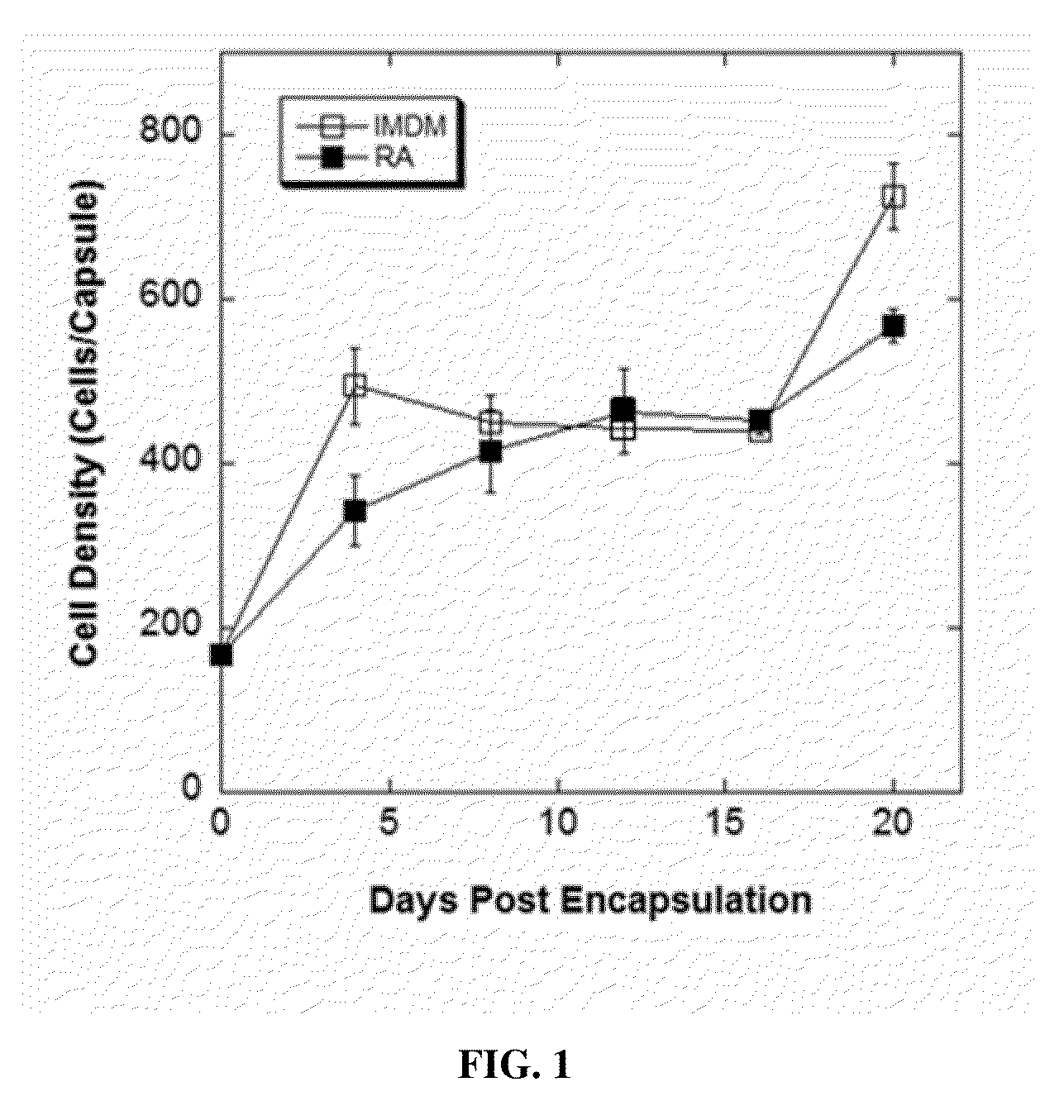
[0134]Under an optimized encapsulation condition (i.e. 2.2% w / v alginate and 5×106 cells / mL), experiments were designed to assess cell proliferation and the expression of an array of neural special markers during a 20-day differentiation period. Encapsulated cells were cultured in the presence or absence of RA, recovered on days 4, 8, 12, 16 and 20 post encapsulation by depolymerizating the alginate microcapsules, and cell number and viability were determined. As indicated in FIG. 1, encapsulated cell numbers in the presence or absence of retinoic acid were similar. The cell proliferation in both conditions exhibited biphasic kinetic properties, and the cultures ultimately reached a final density 2.5 times greater than the initial seeding density. Cell viability was greater than 95% in both conditions and throughout the culture period.
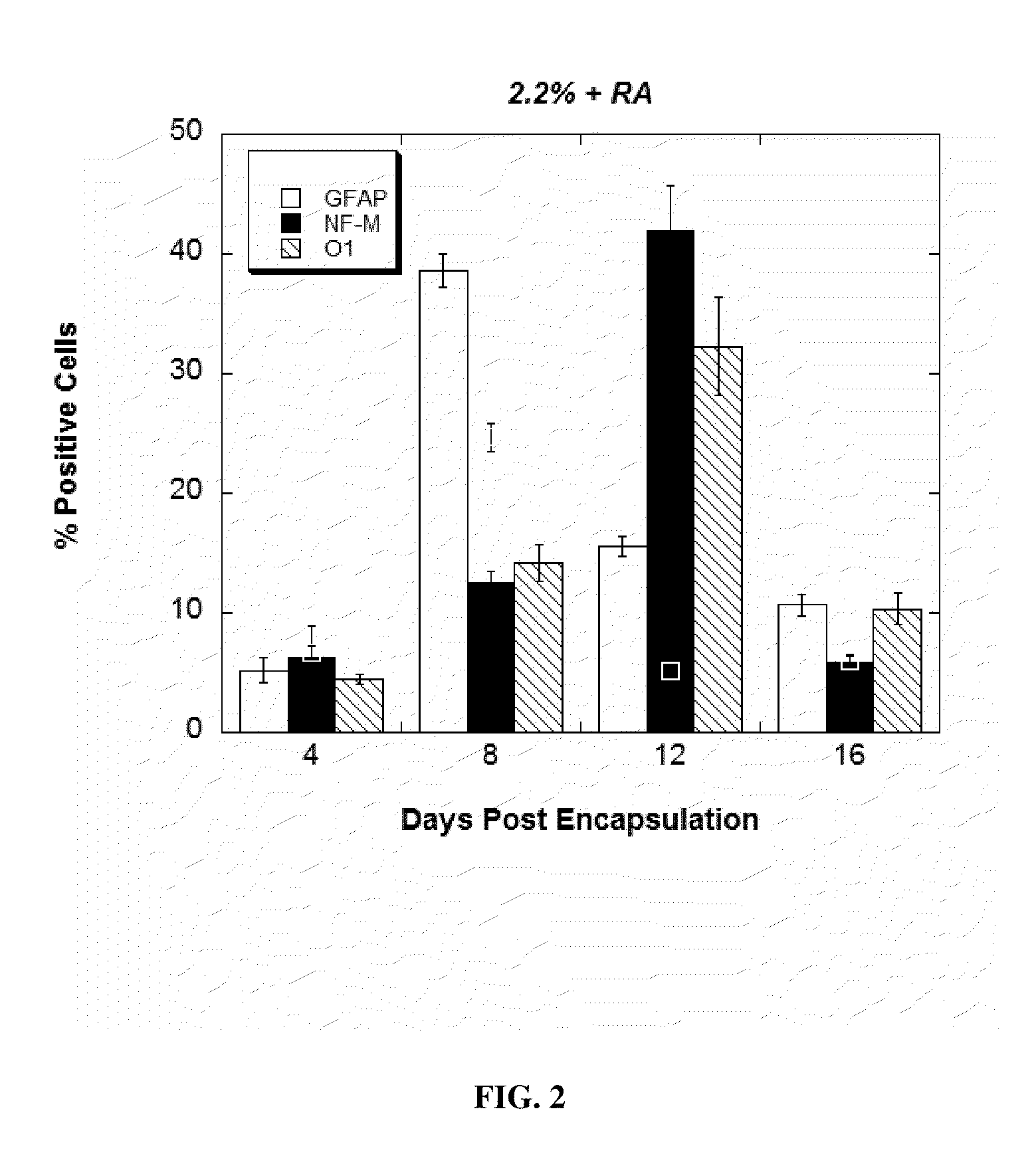
[0135]To assess the effect of RA on lineage commitment within the alginate mic...
PUM
| Property | Measurement | Unit |
|---|---|---|
| step size | aaaaa | aaaaa |
| cell seeding density | aaaaa | aaaaa |
| cell seeding density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More