Induced phase transition method for the production of microparticles containing hydrophobic active agents
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Lipophilic Active Principles
[0084]750 mg of the polymer Resomer® RG-756 is dissolved in 15 mL ethyl formate and transferred to a double-walled steel vessel (inside height 11.0 cm, inside diameter 4 cm). 5 mL of an aqueous 50 mmol Tris(hydroxymethyl)aminomethane solution (pH 7.4) containing 20 mg Budesonide is then dispersed in the polymer solution for 4 minutes at 9,000 rpm at room temperature by means of a mechanical agitator (Dispermat FT, VMA-Getzmann GmbH, 2 cm dissolver disk).
[0085]50 mL of a 50 mmol citrate buffer solution (pH 6.0) containing 4% Pluronic F-68 is then added as continuous phase during agitation at 9,000 rpm. After a dispersal time of 30 seconds, the microparticle suspension is transferred to a 500 mL two-necked flask and agitated with a magnetic stirrer. The solvent ethyl formate is then eliminated at 20° C. by application of vacuum, by introduction of nitrogen or air or by extraction with water. After 5 hours, the suspension is washed with 5 L water or an aqueo...
example 2
[0087]750 mg of the polymer Resomer® RG-756 is dissolved, together with 20 mg Budesonide in 15 mL ethyl formate and transferred to a double-walled steel vessel (inside height 11.0 cm, inside diameter 4 cm). 5 mL of an aqueous 50 mmol
Tris(hydroxymethyl)aminomethane solution (pH 7.4) is added to the solution and dispersed in the polymer solution for 4 minutes at 9,000 rpm by means of a mechanical agitator (Dispermat FT, VMA-Getzmann GmbH, 2 cm dissolver disk).
[0088]50 mL of an aqueous citrate buffer solution (50 mmol and pH 6.0) container Pluronic F-68 is added to the formed dispersion during agitation (9,000 rpm). After a disposal time of 30 seconds, the microparticle suspension is transferred to a 500 mL two-necked flask and processed further as in Example 1.
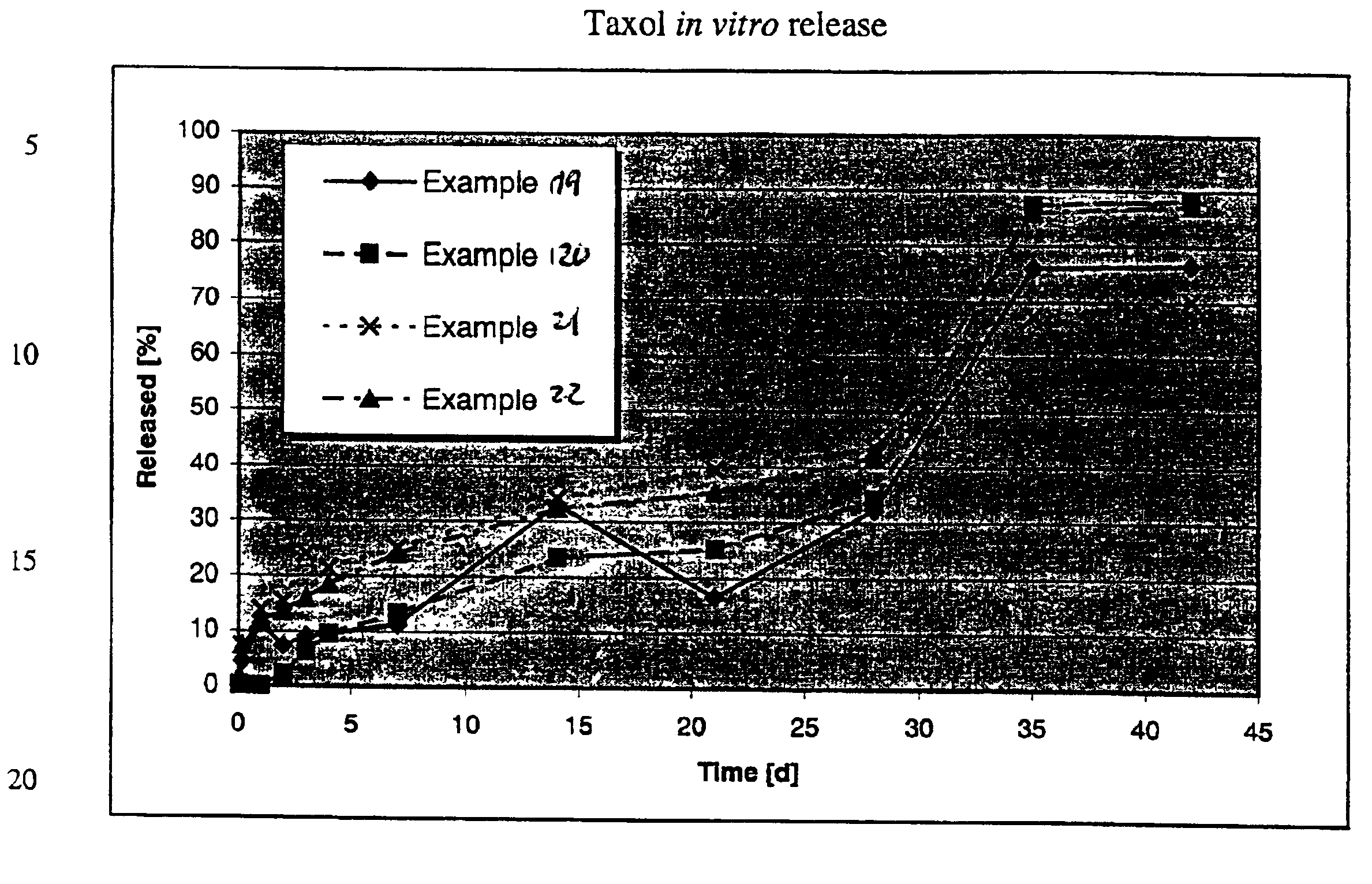
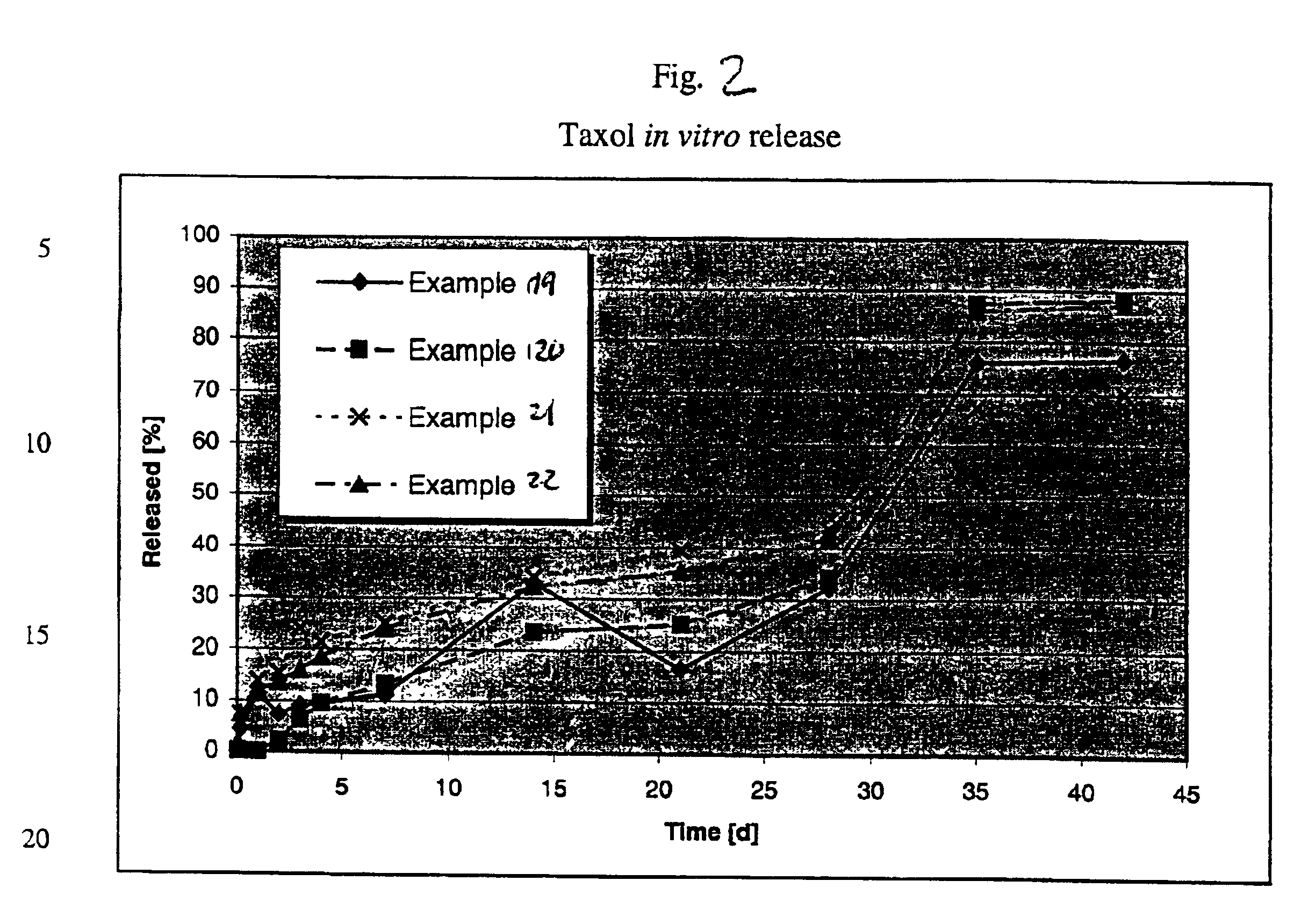
[0089]The lyophilizate, resuspended with water or an aqueous solution, contains microcapsules with an active principle content of 2.2%. The microcapsules have a diameter from 0.2 to 20 μm with the active agent budesonide embedde...
example 3
[0090]750 mg of the polymer Resomer® RG-756 is dissolved in 15 mL ethyl formate and transferred to a double-walled steel vessel (inside height 11.0 cm, inside diameter 4 cm). 5 mL of an aqueous suspension of 20 mg Taxol and a 50 mmol
Tris(hydroxymethyl)aminomethane solution (pH 7.4) is then dispersed in the polymer solution for 4 minutes at 9,000 rpm at room temperature by means of a mechanical agitator (Dispermat FT, VMA-Getzmann GmbH, 2 cm dissolver disk).
[0091]50 mL of an aqueous citrate buffer solution (50 mmol and pH 6.0) containing 4% Pluronic F-68 is then added to the formed dispersion during agitation (9,000 rpm). After a dispersal time of 30 seconds, the microparticle suspension is transferred to a 500 mL two-necked flask and agitated with a magnetic stirrer. The solvent ethyl formate is then eliminated at 20° C. by application of vacuum, by introduction of nitrogen or air or by extraction with water. After 5 hours, the suspension is washed with 5 L water or an aqueous solut...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Viscosity | aaaaa | aaaaa |
| Solubility (mass) | aaaaa | aaaaa |
| Water solubility | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More