Application of aldo-keto reductase in catalytic generation of (R)-4-chlorine-3-hydroxy ethyl butyrate
A technology of ethyl hydroxybutyrate and ethyl chloroacetoacetate is applied in the directions of oxidoreductase, introduction of foreign genetic material using a carrier, recombinant DNA technology, etc., to achieve the effects of high yield, high yield and lower production costs
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
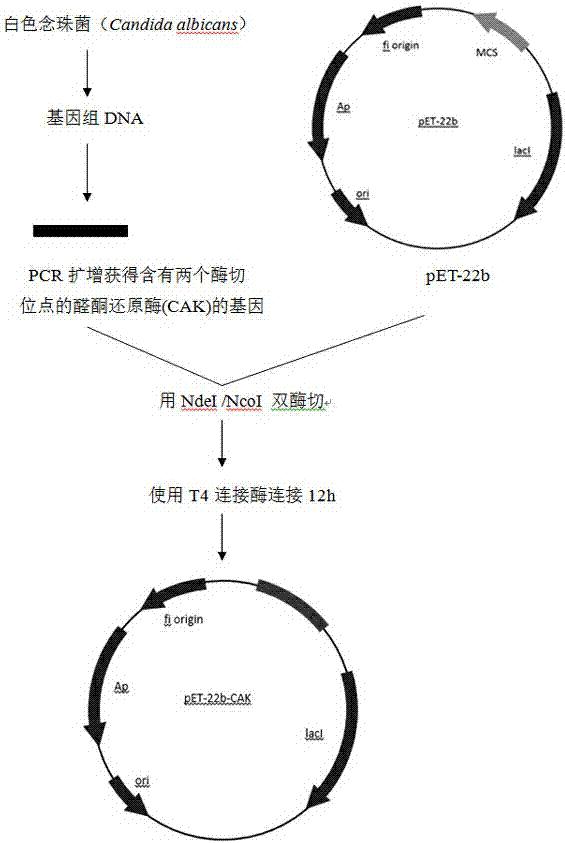
[0026] Embodiment 1: recombinant escherichia coli E. coli Construction of Rosetta(pET-22b-CAK)
[0027] 1. Acquisition of aldehyde and ketone reductase gene
[0028] Candida albicans Candida albicans (purchased from Centraalbureauvoor Schimmelcultures (CBS) Fungal Biodiversiry Centre), medium YPD (g·L -1 ): yeast extract 10, peptone 20, glucose 20, add distilled water to 1L.
[0029] Candida albicans Candida albicans Inoculated in 5mL YPD liquid medium and cultured at 30°C until the logarithmic growth phase, and the genome was extracted using a genomic DNA extraction kit (Beijing Tianwei Bioengineering Co., Ltd. Yeast Genome Extraction Kit, GD2415 Yeast gDNA Kit).
[0030] The primers used to amplify the target fragment from the genome are provided with enzyme cutting sites, and the primer sequences are as follows:
[0031] Upstream primers (including Nd) is: 5'- GGAATTCCATATGCCAGCTCAATTGCA-3'
[0032] Downstream primers (including NCOI ) is: 5'- CATGCCATGGTTAATC...
Embodiment 2
[0038] Example 2: Expression of aldehyde and ketone reductase and preparation of crude enzyme solution
[0039] pick recombinant bacteria E. coli Rosseta (pET-22b-CAK) and Escherichia coli Rosseta (DE3) were cultured in LB liquid medium containing 100 μg / mL ampicillin and 34 μg / mL chloramphenicol overnight at 37°C with shaking. Then they were inoculated into fresh LB medium containing 100 μg / mL ampicillin and 34 μg / mL chloramphenicol according to the inoculum amount of 2%, and cultivated to OD at 37 °C. 600 At about 0.6, add IPTG to a final concentration of 0.8 mmol·L -1 , 25°C, 200rpm, after induction of expression for 10 h, centrifuge at low temperature (4°C, 5000rpm, 15min), resuspend the bacteria sludge with 100mM sodium phosphate buffer (pH7.0), and sonicate the cells (power 300W, ultrasonic 2s, intermittent 1s , a total of 5min), low-temperature centrifugation (4°C, 12000rpm, 15min), the supernatant is the crude enzyme solution of aldehyde and ketone reductase.
Embodiment 3
[0040] Embodiment 3: Determination of aldehyde and ketone reductase enzyme activity
[0041] The enzyme reaction system includes 100mM sodium phosphate buffer (pH7.0), 5mM NADPH, 20mM COBE, and the decrease of absorbance at 340nm is measured at 30°C. Enzyme activity is defined as the amount of enzyme required to oxidize 1 μmol NADPH per minute as one enzyme activity unit U. Protein concentration was determined by the Brandford method. The results showed that the recombinant bacteria E. coli The specific enzyme activity of Rosseta (pET22b-CAK) is 8.1U / mg protein.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More