Due to the high
salinity of concentrated seawater (nearly twice that of ordinary seawater) and the presence of pretreatment chemicals, a large amount of direct
discharge into the sea will cause marine ecological
pollution; it also wastes
freshwater resources and various salt resources in concentrated seawater
[0005] (2) The combination of traditional salt field tanning and salt
chemical industry is the mainstream way of comprehensive utilization of marine and brine resources, but there are shortcomings such as being seriously affected by seasons, relying on a large number of salt fields, and
low resource utilization.
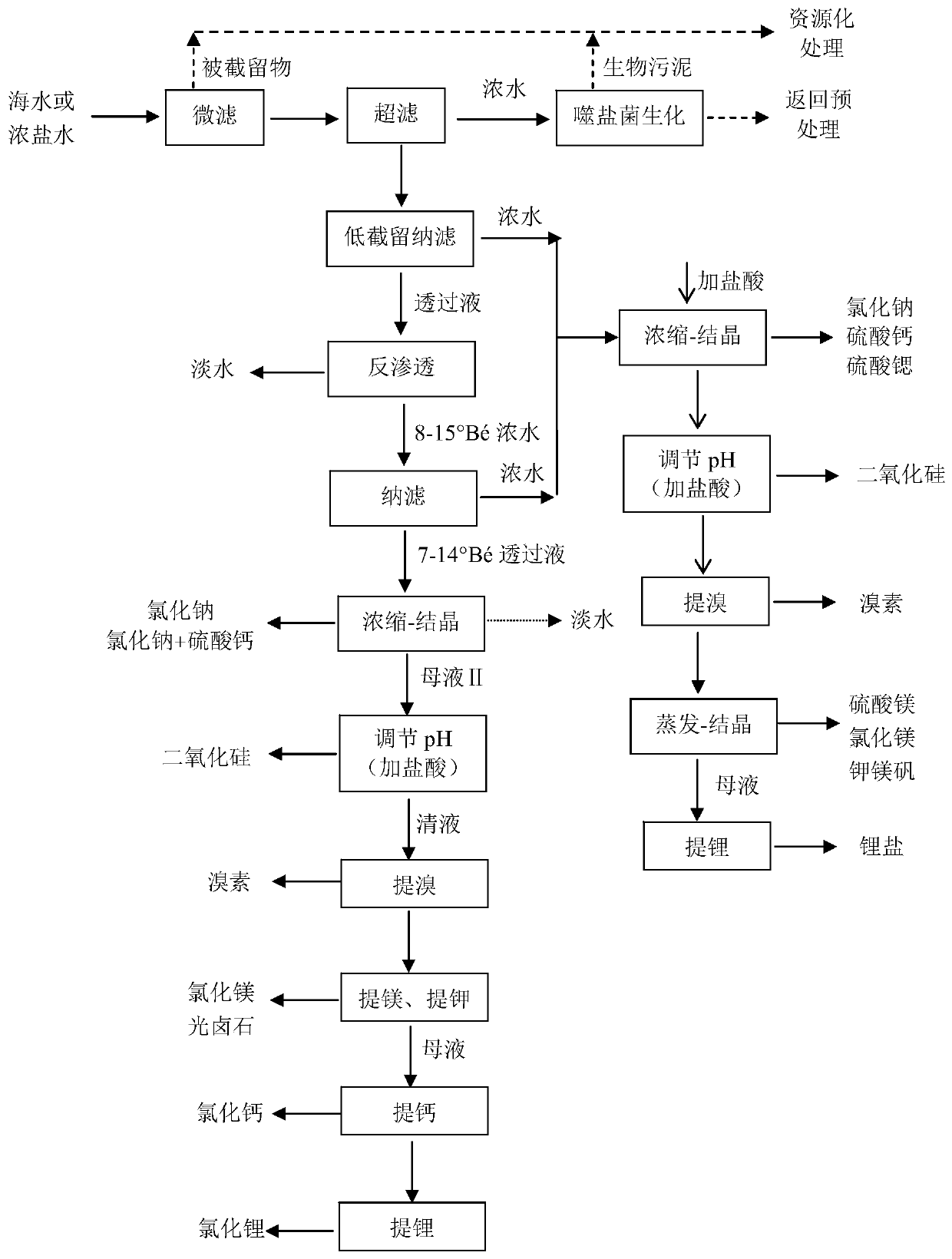
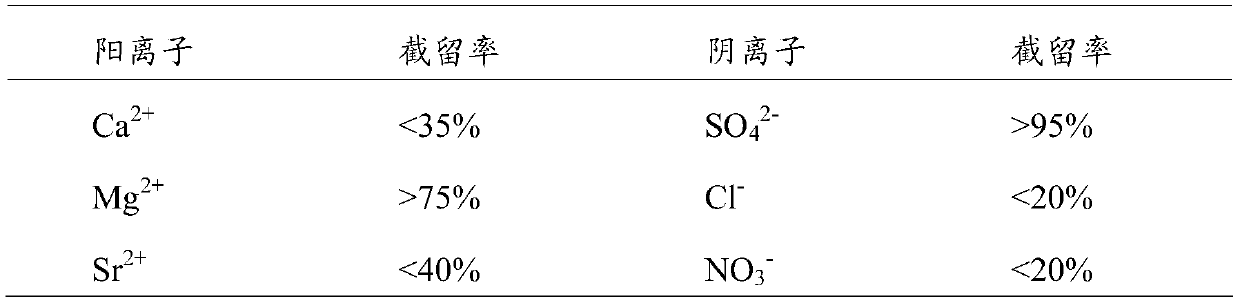
Since ordinary nanofiltration membranes have a relatively high
rejection rate for all
divalent ions, sulfate ions and
calcium ions that are prone to scaling are mainly retained on the concentrated water side of nanofiltration. The use of ordinary nanofiltration membranes in the patent can effectively reduce the The risk of
fouling of the follow-up concentration equipment of the filtered permeate, and the improvement of the purity of the
solid salt, etc., but when using this method to treat the feed liquid with
high calcium ion and
sulfate ion content (such as concentrated seawater by-products of
seawater desalination plants, underground brine, etc.) , the yield of nanofiltration permeate will be very limited, because ordinary nanofiltration membranes do not have the ability to fundamentally and selectively resolve sulfate ions and calcium ions in them. If the concentration multiple is increased, ordinary nanofiltration may cause
Calcium sulfate scaling on the concentrated water side of the membrane, so the economical maximization of the seawater comprehensive treatment process cannot be realized
At the same time, the ordinary nanofiltration concentrated water obtained by using the patented process
route is enriched with a large number of
divalent ions, especially sulfate ions and calcium ions that are easy to scale. Even if the nanofiltration concentrated water is subjected to multi-effect
evaporation or steam compression
Distillation-seed method concentrates
crystallization, calcium ions in the concentrated water (that is, more than 68% of the calcium ions in the
raw material liquid) and most of the
sulfate radicals will still be almost worthless calcium sulfate (rather than the higher value CaCl 2 , MgSO 4 etc.), and the equipment has the risk of
fouling, so it is only piled up as waste (partially used as building materials or used for
land reclamation), which is a serious waste of resources
Therefore, the introduction of ordinary nanofiltration process into the process of membrane or thermal
desalination can only solve the problem of calcium sulfate scaling when the nanofiltration permeate is concentrated in depth, and can obtain pure concentrated brine with a
high concentration. The
recovery rate does not increase, and the comprehensive utilization still loses sulfate ions and most of the calcium ions in seawater, and still mainly produces magnesium chloride, which has low
market acceptance rather than mainly producing magnesium sulfate (
chlorine-free
fertilizer). High degree of product
In addition, ordinary nanofiltration cannot be used directly for concentrated brines with
high concentration such as concentrated seawater or underground brine (
salt water concentration ≥ 8%), and it is only suitable for situations with
low salt concentrations
[0010] In summary, the current
conventional treatment processes for seawater and concentrated brine cannot avoid the scaling problem of calcium sulfate, the problem of a large number of by-products of low-value calcium sulfate salt, and the problem of not being able to obtain high value-added products such as
potassium sulfate and magnesium sulfate; high value-added The extraction technology of salt resources in seawater and brine remains to be further developed
 Login to View More
Login to View More  Login to View More
Login to View More