Magnetic-particle chemiluminescence kit used for detecting monensin and applications of the kit
A chemiluminescence reagent, the technology of monensin, which is applied to a magnetic particle chemiluminescence kit for detecting monensin and its application field, can solve the problem of low false positive rate, influence of reaction time and temperature, poor reagent stability, etc. problem, to achieve the effect of low detection time, fast detection and high sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0035] Embodiment 1 The preparation of specific components of the kit
[0036] 1) Preparation of monensin artificial antigen
[0037] The immunogen was obtained by coupling monensin with bovine serum albumin (BSA) using the activated ester method (NHS-DCC).
[0038] 3) Preparation of monoclonal antibody
[0039] Animal immunization: Immunize Balb / c mice with the immunogen at a dose of 100 μg / mouse to produce antiserum.
[0040] Cell fusion and cloning: Splenocytes from immunized Balb / c mice were fused with SP2 / 0 myeloma cells at a ratio of 9:1 to obtain hybridoma cell lines of monoclonal antibodies.
[0041] Cell cryopreservation and recovery: the hybridoma cells were made into 1×10 cryopreservation medium 9 cells / ml for long-term storage in liquid nitrogen. When recovering, take out the cryopreservation tube, put it into a 37°C water bath to thaw quickly, remove the cryopreservation solution by centrifugation, and transfer it to a culture bottle for culture.
[0042] Pre...
Embodiment 2
[0052] Example 2 The formation of the kit
[0053] Construct the magnetic particle direct competition chemiluminescent detection kit for detecting monensin so that it contains the following components:
[0054] Fluorescent marker for FITC-labeled monensin monoclonal antibody
[0055] Luminescent marker of ABEI-labeled monensin hapten
[0056] Separation reagent of paramagnetic nanobeads coated with goat anti-FITC monoclonal antibody
[0057] Monensin standard solution (0ng / ml, 0.02ng / ml, 0.06ng / ml, 0.18ng / ml, 0.54ng / ml, 1.6ng / ml), the standard diluent is pH7.2, 0.03%NaN 3 , 0.1mol / L PBS buffer solution. The percentage content is a mass percentage content.
[0058] The concentrations of the Monens quality control solution are 0.05ng / ml and 1.0ng / ml respectively, and the dilution solution of the quality control solution is pH7.2, 0.03%NaN 3 , 0.1mol / L PBS buffer solution. The percentage content is a mass percentage content.
[0059] Concentrated lotion pH7.2, 0.2-0.4% Twe...
Embodiment 3
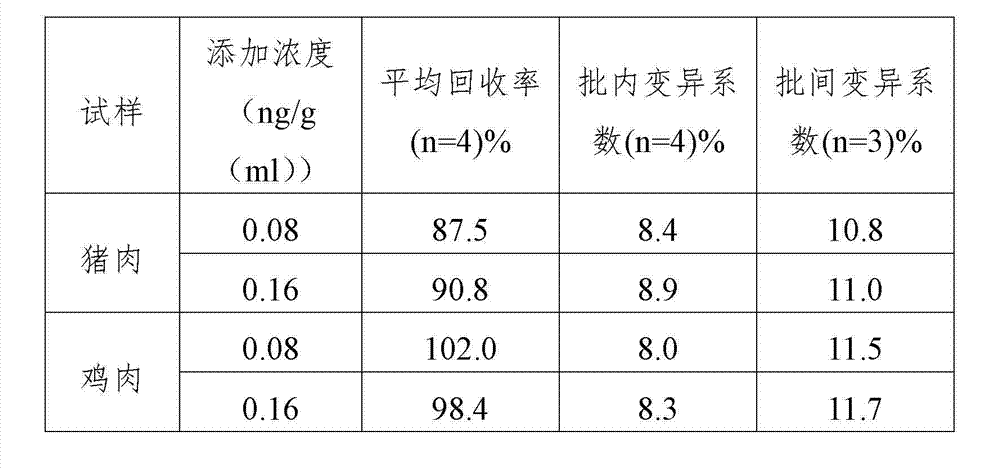
[0060] Example 3 Detection of monensin in actual samples
[0061] 1. Sample pretreatment
[0062] (1) Pork / liver pretreatment method
[0063] Homogenize the sample with a homogenizer; weigh 3.0±0.05g of the homogenate into a 50ml polystyrene centrifuge tube, add 5ml of methanol, then add 1ml of deionized water, shake for 5min, above 3000g, at room temperature (20-25°C) Centrifuge for 5min; take 2ml of supernatant to a 50ml plastic centrifuge tube, add 2ml of 1M sodium hydroxide solution and mix well, then add 6ml of n-hexane, vortex for 3min, centrifuge at room temperature (20-25℃) for 5min; take 3ml Put the supernatant into a 10ml glass test tube, dry it under nitrogen flow at 50-60°C, add 1ml reconstitution working solution and vortex for 2-3min; take 50μl for analysis, sample dilution factor: 2.
[0064] (2) Chicken / liver pretreatment method
[0065] Homogenize the sample with a homogenizer; weigh 3.0±0.05g of the homogenate into a 50ml polystyrene centrifuge tube, add 5...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Sensitivity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com