2,4,7-Trisubstituted fluorene compounds and their electronic devices
A technology of fluorene compounds and electronic devices, which is applied in the field of organic optoelectronic materials to achieve good film-forming properties, thermal stability, high luminous efficiency, and improved electron transmission efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
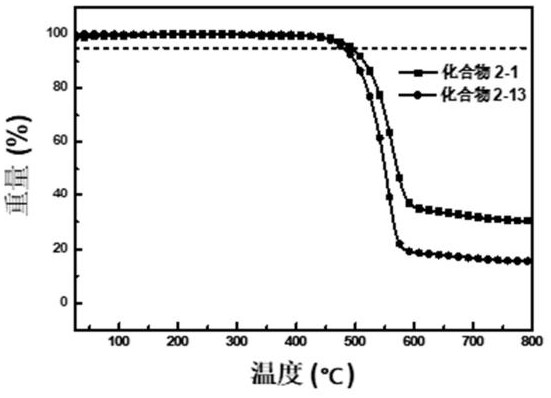
[0296] Embodiment 1: the synthesis of compound 2-1
[0297] (Synthesis of Compound M1)
[0298] The synthetic route of compound M1 is as follows:
[0299]
[0300] In a 100 mL two-necked flask, 1.6 g (6.2 mmol) of 4-bromo-fluorenone was dissolved in 50 mL of dichloromethane, and stirred in an ice bath. Add 0.67 mL (12.8 mmol) of liquid bromine dropwise with a constant pressure dropping funnel. After the addition was complete, the system was gradually warmed up to room temperature and reacted for 6 hours. After the reaction was complete, the reaction solution was poured into a saturated sodium bisulfite solution, extracted three times with dichloromethane, the organic phase was dried over anhydrous sodium sulfate, and the solvent was removed by spinning to obtain a crude product. The crude product was separated and purified on a silica gel column with an eluent of petroleum ether:dichloromethane=5:1 (volume ratio) to obtain 2.3 g of M1 with a yield of 90%. MS (EI): m / z 4...
Embodiment 2
[0311] Embodiment 2: the synthesis of compound 2-13
[0312] (Synthesis of compound M3)
[0313] The synthetic route of compound M3 is as follows:
[0314]
[0315] Under nitrogen, add 4.1 g (10 mmol) M1, 4.2 g (40 mmol) anhydrous sodium carbonate, 9.7 g (35 mmol) 2-boronic acid-4,6-diphenyl -1,3,5-triazine, 115.4 mg (0.1 mmol) tetrakis (triphenylphosphine) palladium and 100 mL mixed solvent (toluene: water: ethanol = 5:1:1 (volume ratio)), the system was heated to Reflux and react overnight under reflux. After the reaction was completed, the heating was stopped, and the reaction system was cooled to room temperature by itself. The reaction solution was poured into about 200 mL of water and extracted with dichloromethane. The organic phase was dried over anhydrous sodium sulfate, concentrated under reduced pressure, and further purified by column chromatography (350 mesh silica gel, eluent: petroleum ether: dichloromethane = 4:1 (volume ratio)) to obtain a yellow solid ...
Embodiment 3
[0322] Embodiment 3: the synthesis of compound 1-23
[0323] (Synthesis of compound M4)
[0324] The synthetic route of compound M4 is as follows:
[0325]
[0326] Under nitrogen, 2.6 g (10 mmol) methyl o-iodobenzoate, 4.2 g (40 mmol) anhydrous sodium carbonate, 1.9 g (10 mmol) 2,4-di Chlorophenylboronic acid, 115.4 mg (0.1 mmol) tetrakis(triphenylphosphine) palladium and 100 mL mixed solvent (toluene: water: ethanol = 5:1:1 (volume ratio)), the system was heated to reflux and under reflux React overnight. After the reaction was completed, the heating was stopped, and the reaction system was cooled to room temperature by itself. The reaction solution was poured into about 200 mL of water and extracted with dichloromethane. The organic phase was dried with anhydrous sodium sulfate, concentrated under reduced pressure, and further purified by column chromatography (350 mesh silica gel, eluent: petroleum ether: dichloromethane = 2:3 (volume ratio)) to obtain a white liqui...
PUM
| Property | Measurement | Unit |
|---|---|---|
| thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com