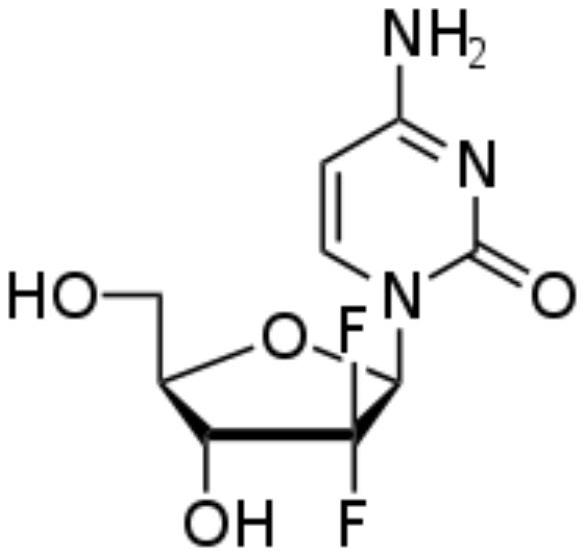
Preparation method of antitumor drug gemcitabine hydrochloride
A technology of gemcitabine hydrochloride and anti-tumor drugs, which is applied in the field of preparation of anti-tumor drug gemcitabine hydrochloride, can solve problems such as easy peroxidation, low purity, and large price fluctuations, and achieve mild reaction process conditions, simple reaction conditions, and environmental pollution small effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0046] The present invention will be further described below in conjunction with specific embodiments and drawings.
[0047] The specific technical scheme is as follows:
[0048] A preparation method of antineoplastic drug gemcitabine hydrochloride, specifically comprising the steps of:
[0049] (1), preparation of antineoplastic drug gemcitabine hydrochloride intermediate:
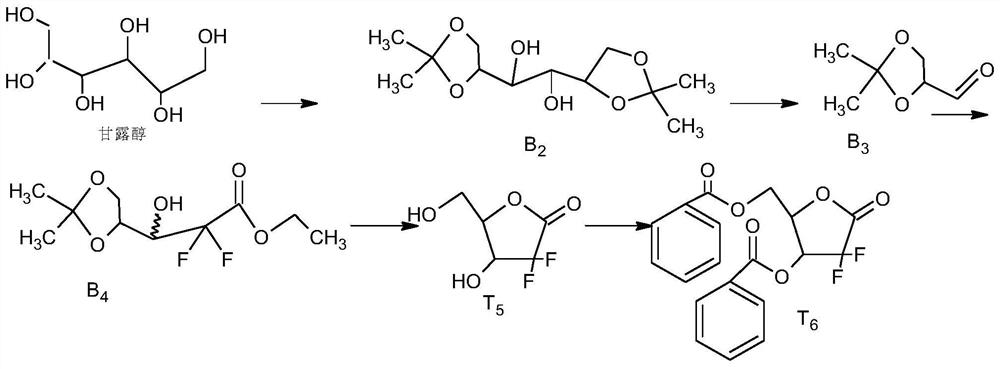
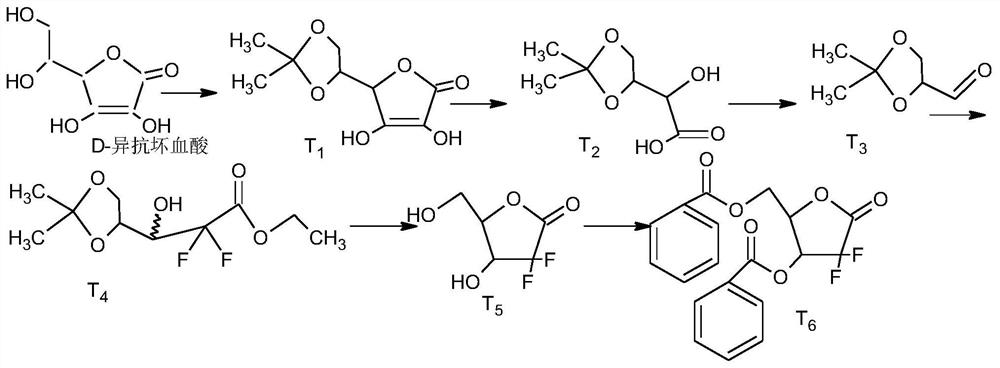
[0050] Step1, synthesis of T1:
[0051] Add 250kg of D-isoascorbic acid, 600kg of acetone, 300kg of 2,2-dimethoxypropane, and 5kg of p-toluenesulfonic acid into the reaction kettle. 6kg of triethylamine, and control the temperature below 50°C, evaporate the reaction solvent to dryness under reduced pressure, and proceed to the next step;
[0052] Step2, synthesis of T2:
[0053] Start stirring, add 1000kg of pre-configured 25% sodium carbonate aqueous solution and 3kg of defoamer, control it between 20-30°C, add 550kg of 18%-25% hydrogen peroxide dropwise, after the dropwise addition, slowly heat up t...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More