High molecular weight major outer membrane protein of moraxella
a molecular weight, moraxella technology, applied in the field of high, can solve the problems of serious disease, speech and cognitive impairment in children, hearing, and speech impairment in children
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0128] This Example illustrates the generation of a non-clumping strain (RH408) of M. catarrhalis.
[0129] M. catarrhalis strain 4223, a clumping strain (a common property of Moraxella strains), was inoculated into several flasks containing 20 mL of brain heat infusion (BHI) broth, and the cultures were incubated with shaking (170 rpm) overnight at 37.degree. C. Five mL of each overnight culture were transferred to five individual 1 mL tubes, and were left sitting undisturbed at room temperature for 3 to 8 hours, to allow bacteria to sediment. One hundred .mu.L of the cleared upper phase of each culture were used to inoculate 25 mL of BHI broth and cultures were incubated overnight at 37.degree. C., as described above. This passaging was repeated six times, using 25 .mu.L of cleared culture to inoculate 25 mL of BHI for each overnight culture. Non-clumping bacterial cultures were identified by measuring the absorbency A.sub.578 at intervals over a 3 hour time period, in order to compa...
example 2
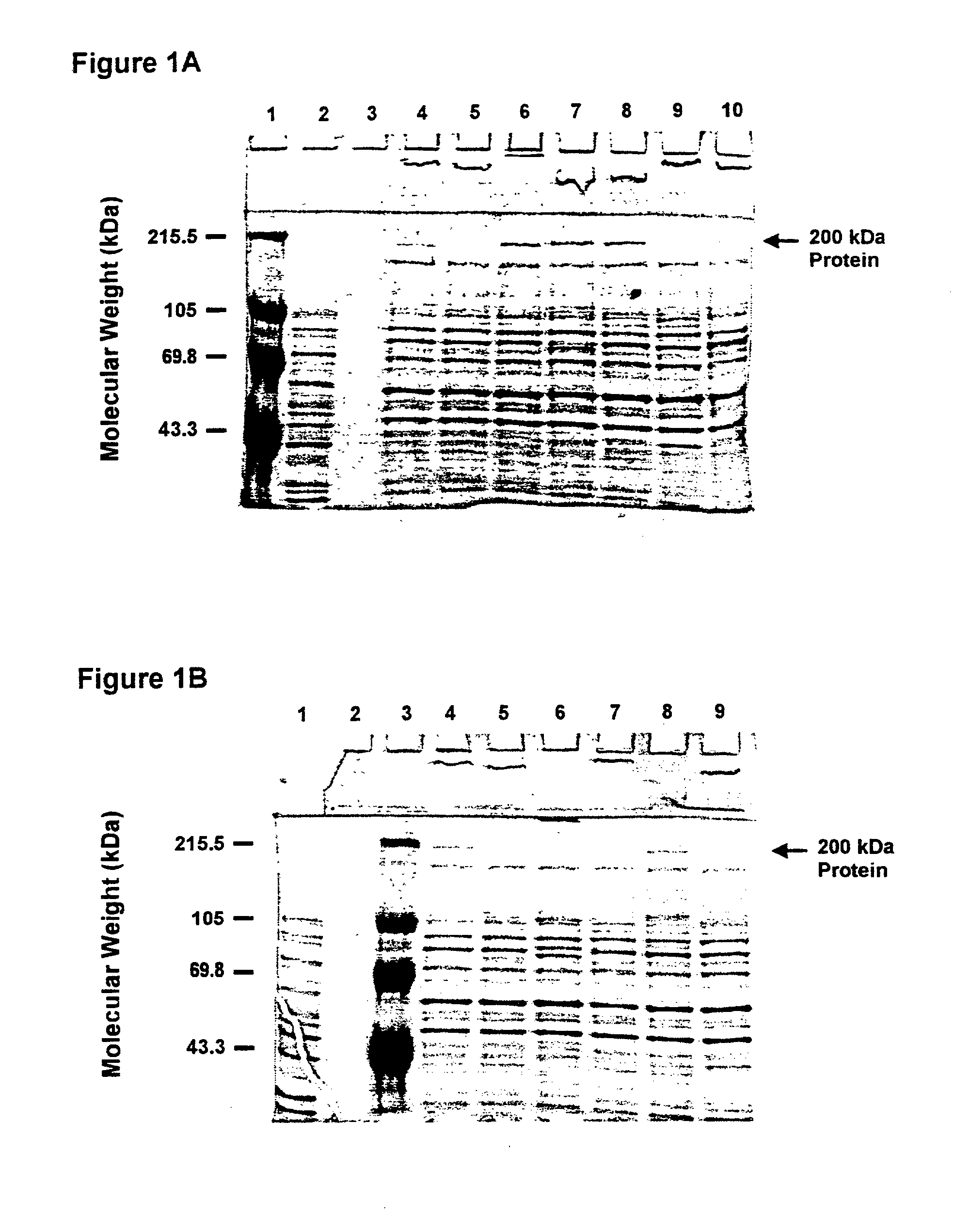
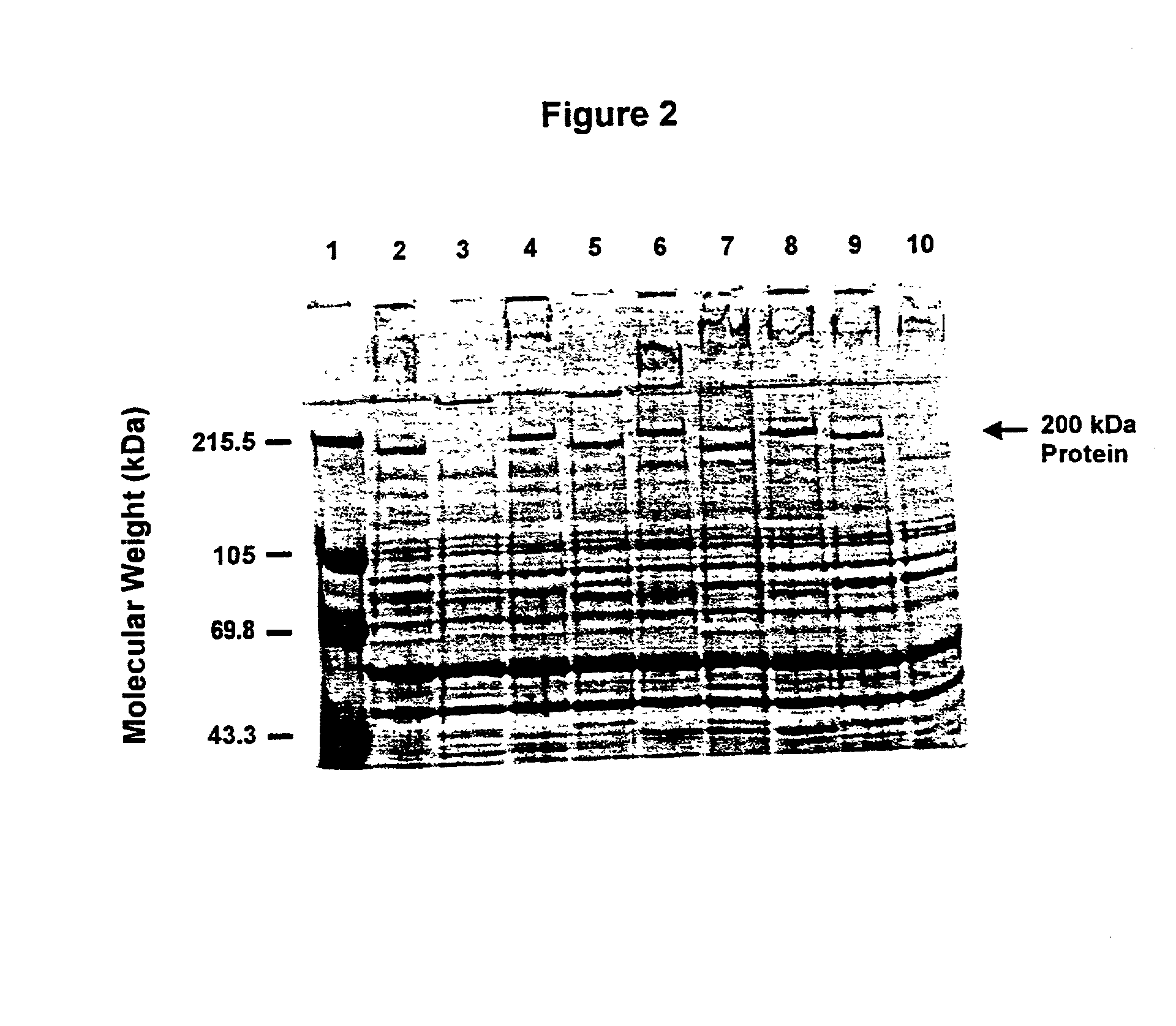
[0130] This Example illustrates the identification of the about 200 kDa outer membrane protein of Moraxella catarrhalis.
[0131] M. catarrhalis strains 4223, RH408, 5191, 8185, M2, M5, ATCC 25240, 3, 56, 135, 585 were grown in brain heart infusion (BHI) broth. The culture was incubated overnight with aeration at 37.degree. C.
[0132] M. catarrhalis cells were sonicated and total. protein was determined using the BCA assay system (Pierce, Rockford, Ill.). Ten .mu.g of total protein were mixed with the SDS-PAGE sample buffer containing 0.3M Tris-HCl (pH 8.0), 50% glycerol, 10% SDS, 20% .beta.-mercaptoethanol and 0.01% bromophenol blue, boiled for 5 minutes and loaded on each lane of SDS-PAGE gel (0.75 mm thick, 7.5% acrylamide). The gels were run at 200 V for 1 hour. Proteins were visualized by staining gels with a solution containing 0.13% Coomassie brilliant blue, 10% acetic acid and 45% methanol. Excess stain was removed with a destaining solution of 5% ethanol and 7.5% acetic acid.
[01...
example 3
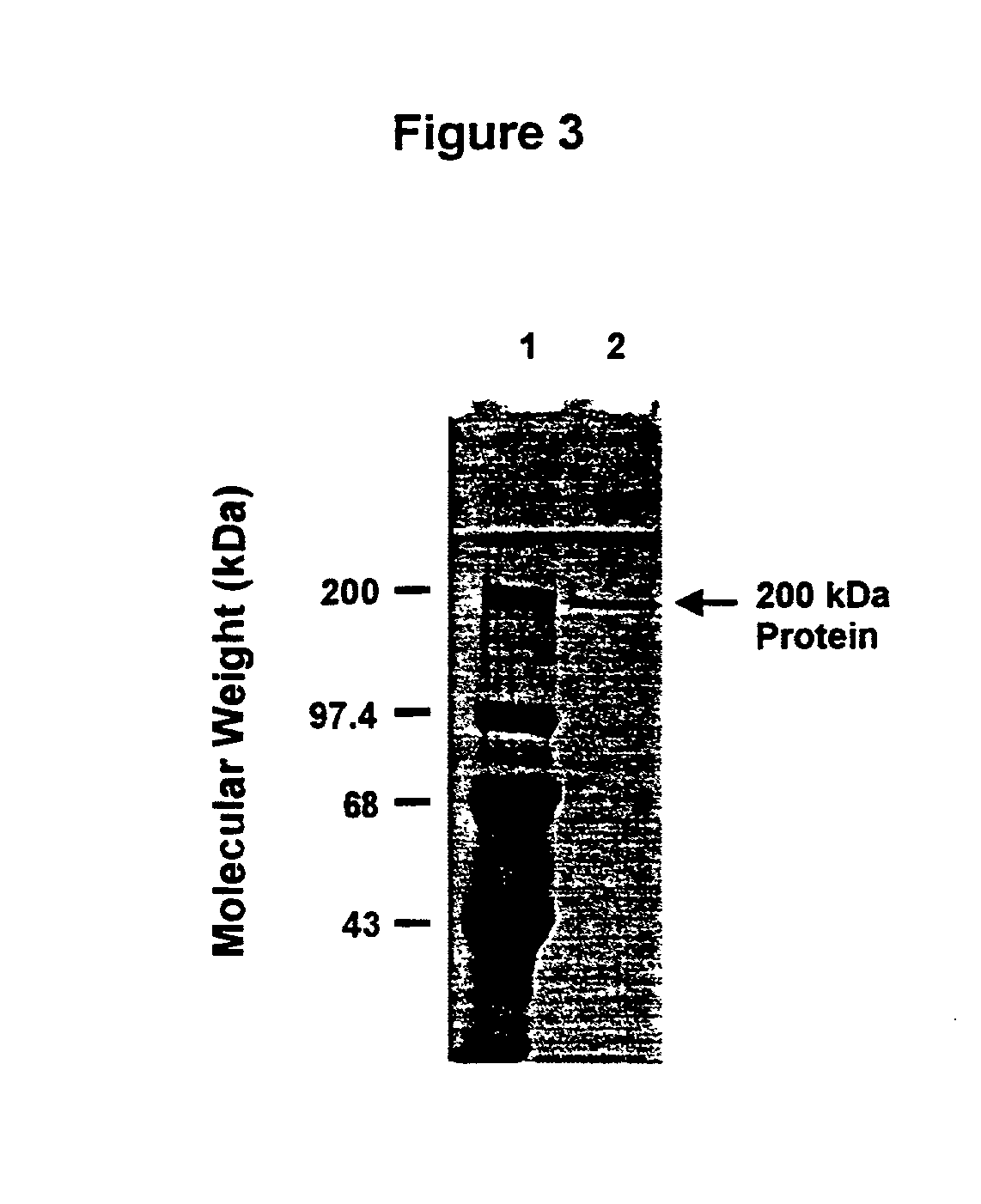
[0138] This Example illustrates the detection of antibodies specific for the about 200 kDa outer membrane protein in a serum obtained from a convalescent patient having recovered from otitis media due to M. catarrhalis.
[0139] After separation by SDS-PAGE, bacterial proteins were transferred from polyacrylamide gels to prepared PVDF (polyvinylidene fluoride; Millipore) membranes at a constant voltage of 70 V for 1.5 h in a buffer system consisting of 3 g Tris, 14,4 g glycine and 200 ml methanol per liter at 4.degree. C. Membranes with transferred proteins were blocked with Blocking Reagent (from Boehringer Mannheim) diluted in TBS (0.1M Tris, 0.15M Nacl) at room temperature for 30 min. Blots were exposed to convalescent antiserum diluted 1:500 in Blocking Reagent / TBS with 0.1% Tween 20 for 2 hours at room temperature. This patient had otitis media and the M. catarrhalis strain isolated from the patient's ear fluid was M. catarrhalis CJ7. Blots were then washed 2 times in Blocking Rea...
PUM
| Property | Measurement | Unit |
|---|---|---|
| apparent molecular mass | aaaaa | aaaaa |
| wt % | aaaaa | aaaaa |
| wt % | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More