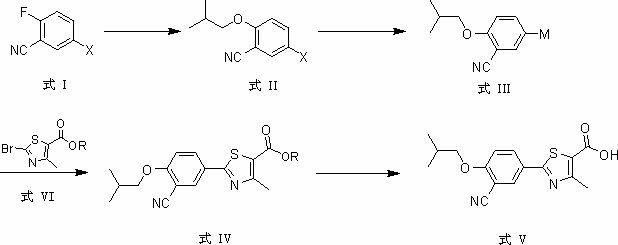
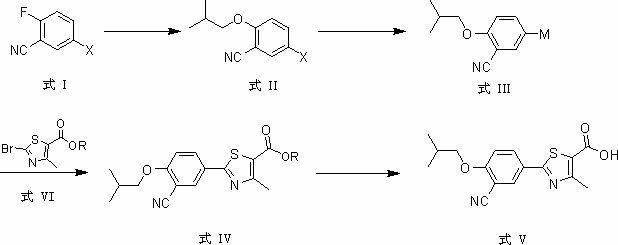
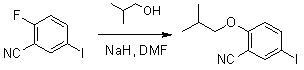
At present, most of the reported synthetic methods of febuxostat adopt the traditional method of constructing and synthesizing thiazole, usually the synthetic
route is relatively long (generally more than six steps), and the conversion of functional groups in the molecules involved is very cumbersome; secondly, The starting materials of various synthetic routes are expensive, and even in some steps, highly toxic, corrosive and polluting chemical reagents such as
sodium cyanide,
trifluoroacetic acid, urotropine,
hydrogen sulfide, and
phosphorus pentasulfide are required , has a greater
adverse effect on the operator and the environment
In addition, some reactions require the use of diazotization reactions, which are not suitable for control in the operation of large-scale reactions, which makes industrial production more difficult
Moreover, the total yield of the synthetic method reported in the existing literature is low, generally lower than 30%, which also makes it unsuitable for industrial production
[0003]In the traditional synthetic method, when constructing and introducing cyano group in the molecule: JP06345724, EP0513379A1, WO9209279, CN101386604 disclose in the
raw material through existing on the
benzene ring Two substituents, the cyano group positions it; JP10139770 discloses a
substitution reaction through the existing
leaving group on the
benzene ring in the intermediate; WO9209279 in the
raw material or intermediate Amino groups undergo diazotization reaction; these methods must use highly toxic
potassium cyanide,
sodium cyanide and other cyanides, the safety of the process is very low, and the environment is very destructive
[0004] When constructing a ring-closing reaction to generate a thiazole ring, the cyano group in the
raw material or intermediate is converted into a
thioamide using a
thio reagent: US20050075503 and US2005027128 disclose the use of Passing
hydrogen sulfide gas into the solution to prepare under pressure, the reaction is very dangerous, highly corrosive, and environmentally destructive, which is not conducive to large-scale production; CN101391988 discloses a one-pot method for the synthesis of thiazole rings, avoiding the use of
hydrogen sulfide, but the cost of raw materials is relatively high High; CN101412699 discloses the use of HCl / DMF
system to synthesize thiazole ring, avoiding the use of gas operation, and the yield is high, but the reaction cycle is very long and the post-treatment is cumbersome
When converting the
amide on the raw material or intermediate benzene ring into
thioamide, JP06293746 discloses the use of highly toxic
phosphorus pentasulfide, which is not conducive to large-scale production
[0005] When introducing an
aldehyde group on the benzene ring of the intermediate after constructing the thiazole ring: JP11060552 and JP1045733 disclose the use of polyphosphoric acid as a
solvent, and
polymerization occurs during the reaction. Viscous, unfavorable for stirring and low yield, more impurities, unfavorable for production; CN101412699 discloses the use of
trifluoroacetic acid as a
solvent, and the yield is greatly improved, but the price and cost of
trifluoroacetic acid are high, difficult to recycle and highly corrosive , low safety; CN102002017 discloses the use of mixed acids as solvents, but the acid
pollution is still not improved
[0006]In the traditional method of constructing thiazole ring, functional
group transformation needs to be carried out many times, the transformation is cumbersome, and the reaction steps are increased. Generally, more than six steps are required. For example, China Medical Journal In [2009,40(1):1-5], the final compound was synthesized after nine steps of reaction
 Login to View More
Login to View More  Login to View More
Login to View More