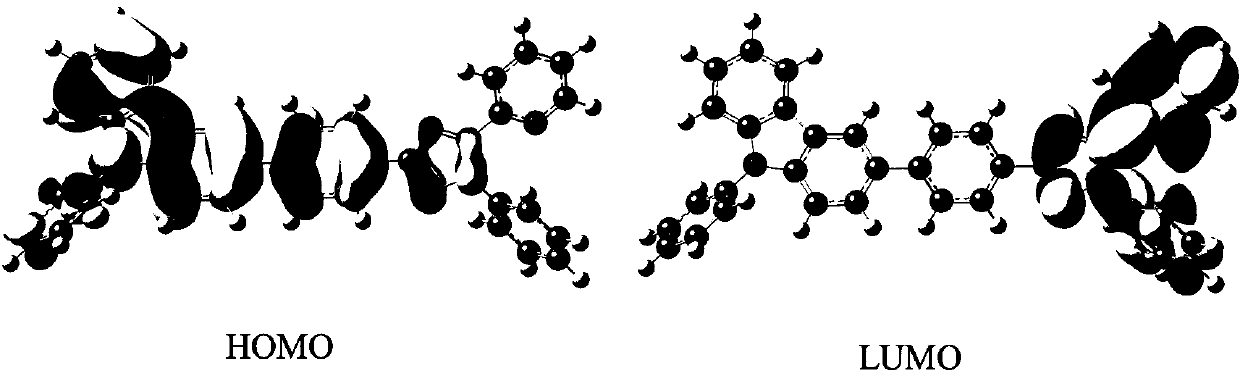
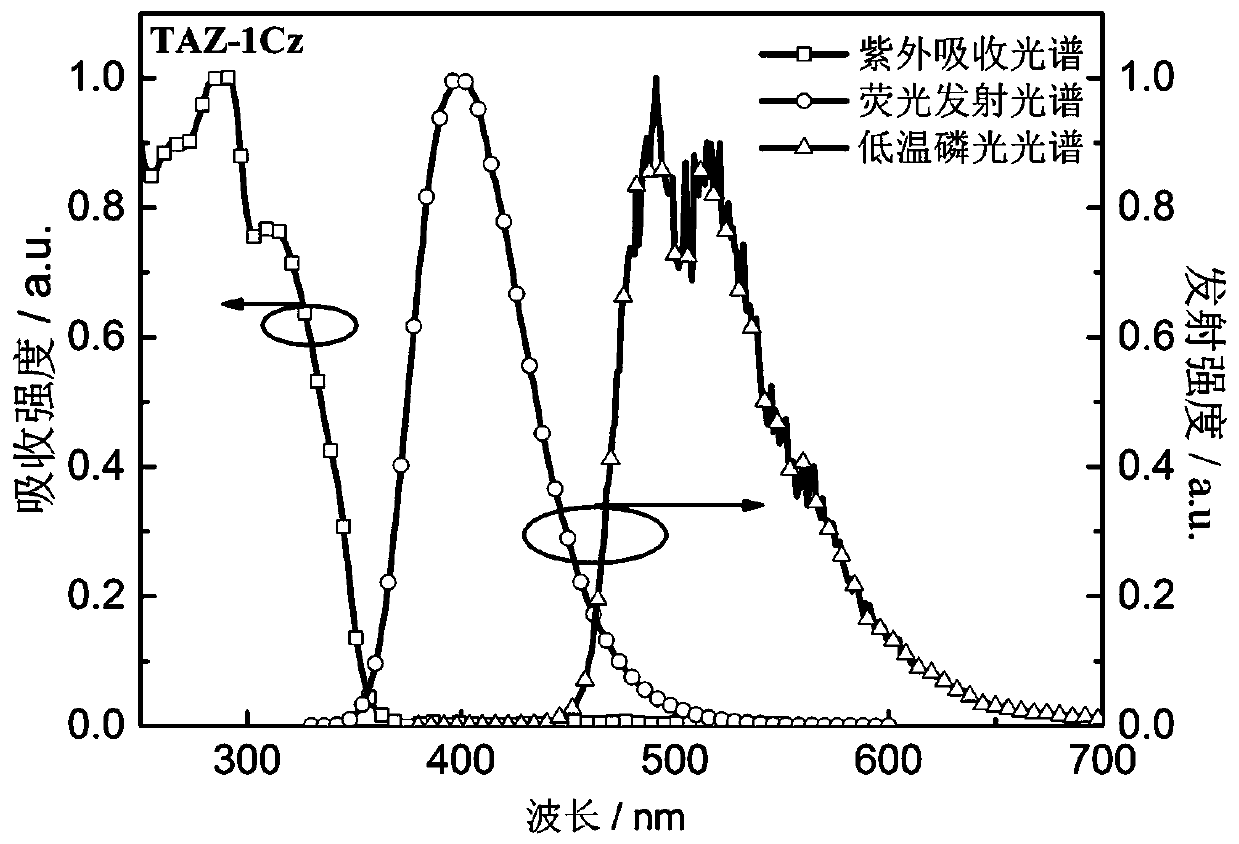
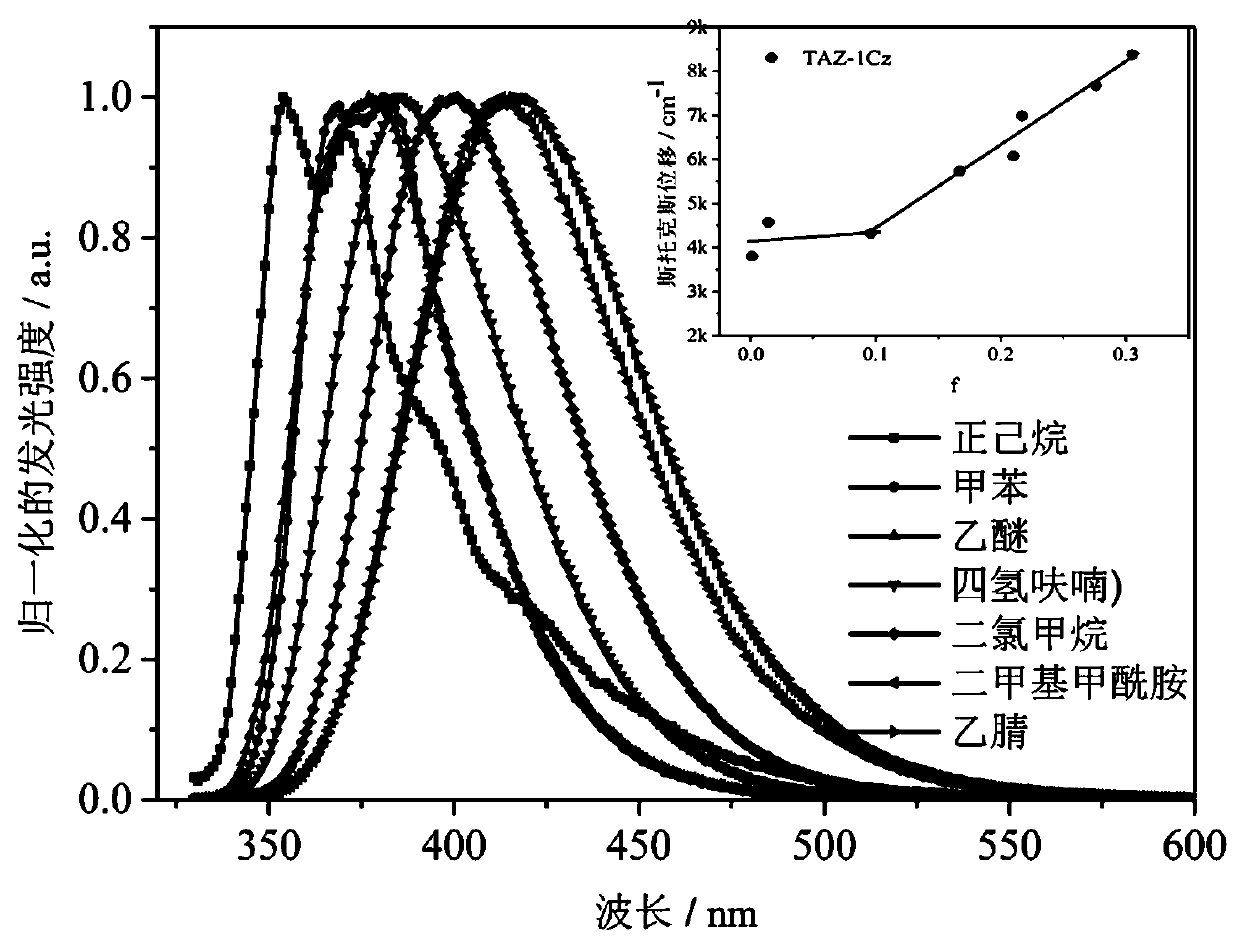
Dark blue fluorescent material based on 1,2,4-triazole derivatives and preparation method thereof
A fluorescent material, dark blue technology, applied in the field of non-doped fluorescent materials, can solve the problems of not being able to prepare non-doped fluorescent devices, unable to realize charge transfer excited state, unable to red shift, etc., to improve radiation excitation. effect of electron utilization rate, improving electron transport capability, and high radiation exciton utilization rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0051] 2-cyanopyridine (5.21g, 50mmol), hydrazine hydrate (2.50g, 50mmol), and 25mL of ethanol were sequentially added to a 250mL two-necked round-bottomed flask, and reacted at a low temperature of 0°C for 8 hours to form a viscous light yellow paste. The excess ethanol was removed under vacuum at room temperature, the solid was washed with a small amount of ether, filtered, and dried under vacuum at room temperature for 3 hours to obtain white crystals of (2-pyridine)aminohydrazone.
[0052] Add (2-pyridine)aminohydrazone (4.08g, 30mmol), sodium carbonate (3.15g, 30mmol), 4-bromobenzoyl chloride (7.00g, 30mmol), and 30mL tetrahydrofuran into a 250mL two-necked round-bottomed flask, and react at room temperature for 6h ,filter. The filtrate was dehydrated and ring-closed in 30ml of ethylene glycol at high temperature for 30min, filtered, dried in vacuo for 8h, and recrystallized from ethanol to obtain 2-(3-(4-bromophenyl)-1H-1,2,4-tri Azol-5-yl) pyridine white needle-like cr...
Embodiment 2
[0069] Take 2-(1-phenyl-3-(4-bromophenyl)-1H-1,2,4-triazol-5-yl)pyridine (1.47g, 4mmol) prepared in Example 1, and ( 3-(9H-carbazol-9-yl)phenylboronic acid (1.72g, 6mmol), tetrakis(triphenylphosphine)palladium (90mg, 0.08mmol), sodium carbonate (1.59g, 15mmol), and 100mL tetrahydrofuran were added together In a 250mL two-necked round-bottomed flask, heated to reflux temperature for 24h reaction.Reaction solution was cooled to room temperature, added 50mL water, extracted 3 times with dichloromethane, each dichloromethane consumption 50mL.Merge the organic layers, washed several times, without Dry over magnesium sulfate, filter, distill under reduced pressure, and then purify by petroleum ether / ethyl acetate (2:1) column chromatography to obtain 9-(4'-(1-phenyl-5-(pyridin-2-yl )-1H-1,2,4-triazol-3-yl)-[1,1'-biphenyl]-3-yl)-9H-carbazole (TAZ-2Cz) white solid, yield 60% .
[0070] The structural formula of the prepared TAZ-2Cz is as follows.
[0071]
[0072] 1 H NMR (600 ...
Embodiment 3
[0079] Take 2-(1-phenyl-3-(4-bromophenyl)-1H-1,2,4-triazol-5-yl)pyridine (1.88g, 5mmol) prepared in Example 1, and ( 9-Phenyl-9H-carbazol-3-yl)boronic acid (1.72g, 6mmol), tetrakis(triphenylphosphine)palladium (90mg, 0.08mmol), sodium carbonate (1.59g, 15mmol), and 100mL THF together Added to a 250mL two-neck round bottom flask, heated to reflux temperature for 24h. The reaction solution was cooled to room temperature, 50 mL of water was added, and extracted three times with dichloromethane, each time using 50 mL of dichloromethane. The organic layers were combined, washed several times with water, dried over anhydrous magnesium sulfate, filtered, distilled under reduced pressure, and then purified by petroleum ether / ethyl acetate (3:1) column chromatography to obtain 9-phenyl-3-(4-( 1-Phenyl-5-(pyridin-2-yl)-1H-1,2,4-triazol-3-yl)phenyl)-9H-carbazole (TAZ-3Cz) white solid, yield 67 %.
[0080] The structural formula of the prepared TAZ-3Cz is as follows.
[0081]
[00...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Maximum current efficiency | aaaaa | aaaaa |
| Maximum power efficiency | aaaaa | aaaaa |
| Maximum current efficiency | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap