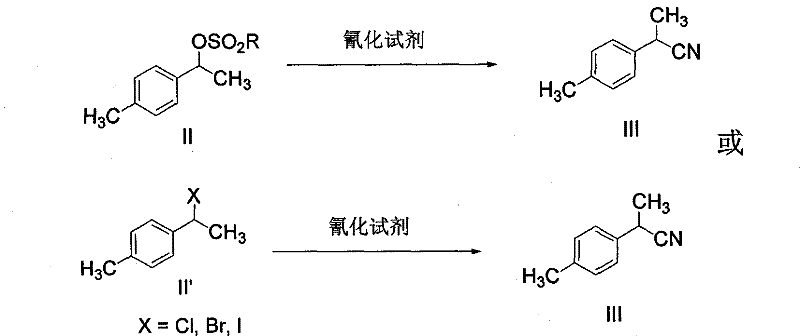
Method for synthesizing loxoprofen sodium
A technique for loxoprofen sodium and a synthetic method, which is applied in the field of pharmaceutical and chemical engineering, and can solve problems such as harsh synthetic conditions, difficult catalyst recycling, and complex raw materials
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
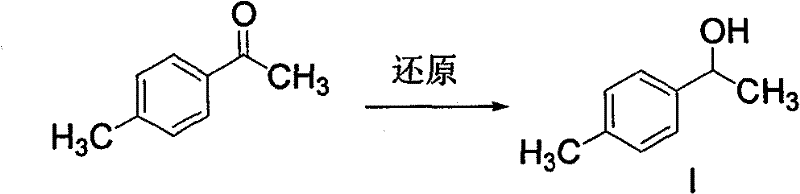
[0104] Example 1 Preparation of 1-(4-methylphenyl)-1-ethanol (compound I)
[0105]6.7g (0.05mol) of 4-methylacetophenone, 30ml of methanol, stir evenly, cool down in an ice-water bath, add 2.28g (0.06mol) of sodium borohydride, react at 30°C for 3 hours, add 20ml of water, stir evenly, water The layer was extracted with dichloromethane, and the layers were separated. The organic phase was washed with water, dried over anhydrous sodium sulfate, filtered, and the filtrate was evaporated to remove the solvent under reduced pressure to obtain 6.56 g of 1-(4-methylphenyl)-1-ethanol (Compound I), with a yield of 96.4%, detected by HPLC The content is more than 99%.
Embodiment 2
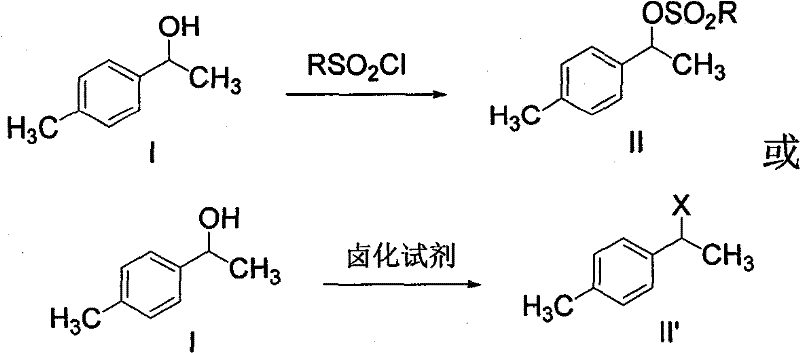
[0106] The preparation of embodiment 2 (1-(4-methylphenyl)-1-ethanol) sulfonate (compound II)
[0107] 6.8g (0.05mol) of compound I, 9.1g (0.09mol) of triethylamine, 30ml of dichloromethane, stirred, and cooled in an ice-water bath. 10.3 g (0.09 mol) of methanesulfonyl chloride was slowly added dropwise at below 15°C. After the dropwise addition was continued and stirred for 3 hours, the heating was stopped, and the layers were separated. The organic phase was washed with saturated sodium bicarbonate until pH = 7, dried over anhydrous sodium sulfate, filtered, and the solvent was evaporated under reduced pressure to obtain 9.81 g of compound II. The yield is 91.7%, and the content detected by HPLC is 95%.
Embodiment 3
[0108] Example 3 Preparation of 1-(4-methylphenyl)-1-chloroethane (compound II')
[0109] 61g (0.448mol) of compound I, 200ml of carbon tetrachloride, and 80g (0.67mol) of thionyl chloride were added dropwise at room temperature. After dropping, the mixture was heated to reflux for 3 hours. Cool down to room temperature, add an appropriate amount of water to wash the organic phase, and then wash the organic phase with saturated sodium bicarbonate until the pH of the aqueous layer = 7, and add anhydrous sodium sulfate to dry. Filtration, carbon tetrachloride was distilled off under reduced pressure to obtain 65 g of compound II', the yield was 93.5%, and the content detected by HPLC was 97%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More