Paracetamol, caffein, artificial cow-bezoar and chlorphenamine maleate capsule and preparation method thereof
The technology of Kahuangmin capsule and caffeine is applied in the field of aminocahuangmin capsule and its preparation, which can solve the problems of poor bioavailability of aminocahuangmin capsule, high frequency of taking medicine, short duration of drug effect, etc. The effect of action time, drug effect time is long, and uniformity is good.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
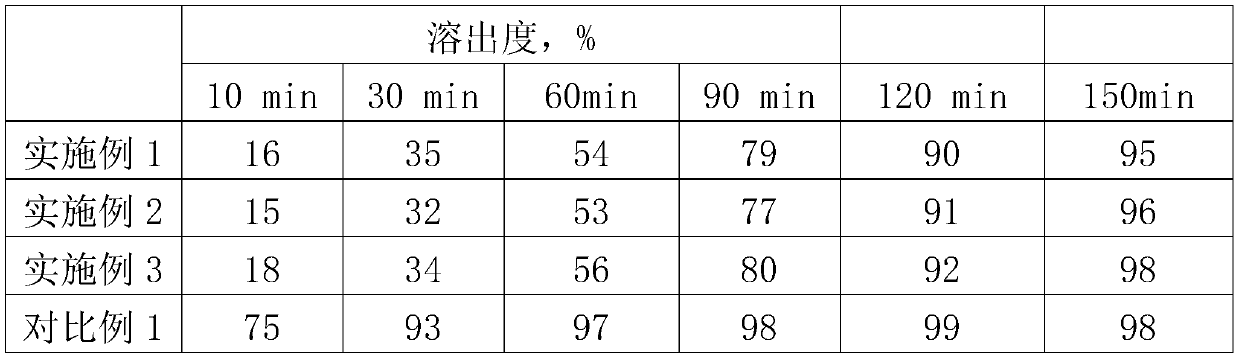
Examples
Embodiment 1
[0023] Anka Huangmin capsules, by weight, its content includes the following components:
[0024] 70 parts of paracetamol, 2 parts of caffeine, 0.5 parts of chlorpheniramine maleate, 2 parts of artificial bezoar, 4 parts of filler, 2 parts of adhesive, 5 parts of sustained release agent, and 2 parts of lubricant.
[0025] Wherein the filler is a mixture of microcrystalline cellulose, lactose and starch;
[0026] Wherein binder is sodium carboxymethyl cellulose;
[0027] Wherein the sustained-release agent is ethyl cellulose;
[0028] Wherein the lubricant is a mixture of talcum powder and magnesium stearate.
[0029] Preparation method, concrete steps include:
[0030] S1. Weighing para-acetylaminophenol, caffeine, chlorpheniramine maleate and artificial bezoar in proportion and then micronizing them;
[0031] S2. Add filler and binder to the micropowder prepared in S1, add water with 5-10% of the mass of the micropowder, and wet granulate;
[0032] S3. The wet granules a...
Embodiment 2
[0037] Anka Huangmin capsules, by weight, its content includes the following components:
[0038] 75 parts of acetaminophen, 3 parts of caffeine, 1 part of chlorpheniramine maleate, 3 parts of artificial bezoar, 5 parts of filler, 2 parts of adhesive, 5 parts of sustained release agent, and 1 part of lubricant.
[0039] Wherein the filler is one or more of pregelatinized starch, mannitol, calcium hydrogen phosphate, calcium dihydrogen phosphate;
[0040] Wherein the binder is one or more of sodium carboxymethyl cellulose, hydroxypropyl cellulose, methyl cellulose, polyvinylpyrrolidone;
[0041] Wherein the sustained-release agent is one or both of ethyl cellulose, ethylene-vinyl acetate copolymer, and polymethacrylate;
[0042] Wherein the lubricant is one or both of micropowder silica gel and sodium stearate fumarate.
[0043] Preparation method, concrete steps include:
[0044] S1. Weighing para-acetylaminophenol, caffeine, chlorpheniramine maleate and artificial bezoar i...
Embodiment 3
[0051] Anka Huangmin capsules, by weight, its content includes the following components:
[0052] 80 parts of paracetamol, 5 parts of caffeine, 1 part of chlorpheniramine maleate, 3 parts of artificial bezoar, 6 parts of filler, 3 parts of adhesive, 6 parts of sustained release agent, and 3 parts of lubricant.
[0053] Wherein the filler is one or more of microcrystalline cellulose, lactose, starch, pregelatinized starch, mannitol, calcium hydrogen phosphate, and calcium dihydrogen phosphate;
[0054] Wherein the binder is one or more of sodium carboxymethyl cellulose, hydroxypropyl cellulose, methyl cellulose, polyvinylpyrrolidone;
[0055] Wherein the sustained-release agent is one or more of ethyl cellulose, ethylene-vinyl acetate copolymer, polymethacrylate;
[0056] Wherein the lubricant is one or more of talcum powder, magnesium stearate, micropowder silica gel and sodium stearate fumarate.
[0057] Preparation method, concrete steps include:
[0058] S1. Weighing par...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com