Supplemented and unsupplemented tissue sealants, methods of their production and use
a technology of tissue sealant and supplement, which is applied in the direction of peptide/protein ingredients, unknown materials, and macromolecule non-active ingredients, etc. it can solve the problems of inability to establish if fgf growth factor is chemotactic for fibroblasts, inconsistent use of fgf growth factor to promote wound healing, and inability to possess true wound healing properties. , to achieve the effect of increasing the longevity and stability of fg, reducing thrombo
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of HBGF-1 for Supplementation of FG
[0244]An 800 ml culture of recombinant E. coli containing a plasmid that included DNA encoding HBGF-1β was prepared. After induction and culturing for 24 hours at 37° C., the cells were centrifuged and the supernatant was discarded. The cell pellet was resuspended in 25 mls of 20 mM phosphate buffer, containing 0.15 M NaCl, pH 7.3. The suspended cells were disrupted with a cell disrupter and the cell debris was separated from the resulting solution by centrifugation at 5000 g for 20 min.
[0245]The pellet was discarded and the supernatant containing the solubilized HBGF-1β and the other bacterial proteins was loaded onto a 2.6 cm diameter by 10 cm high column of Heparin-Sepharose™ (Pharmacia Fine Chemicals, Upsala, Sweden). The column was washed with 5 column volumes of 0.15 M NaCl in 20 mM phosphate buffer, pH 7.3, and then was eluted with a 0.15 M NaCl in 20 mM phosphate buffer to 2.0 M NaCl gradient.
[0246]The eluate was monitored by UV...
example 2
Stability of HBGF-1
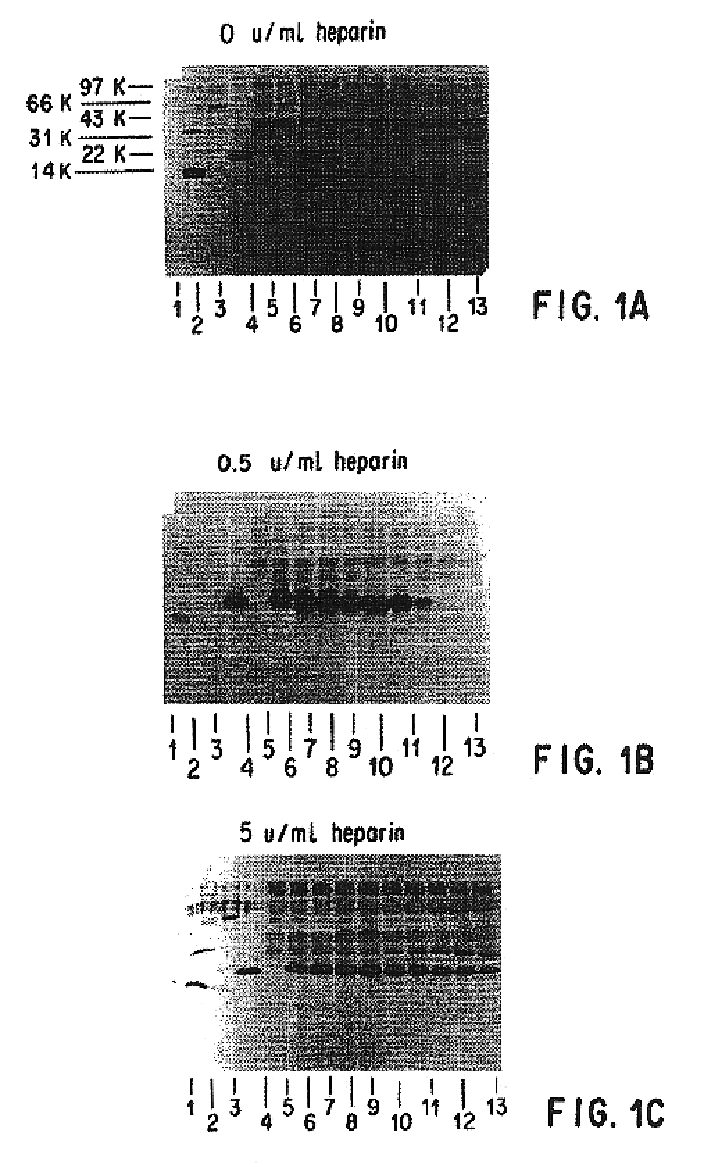
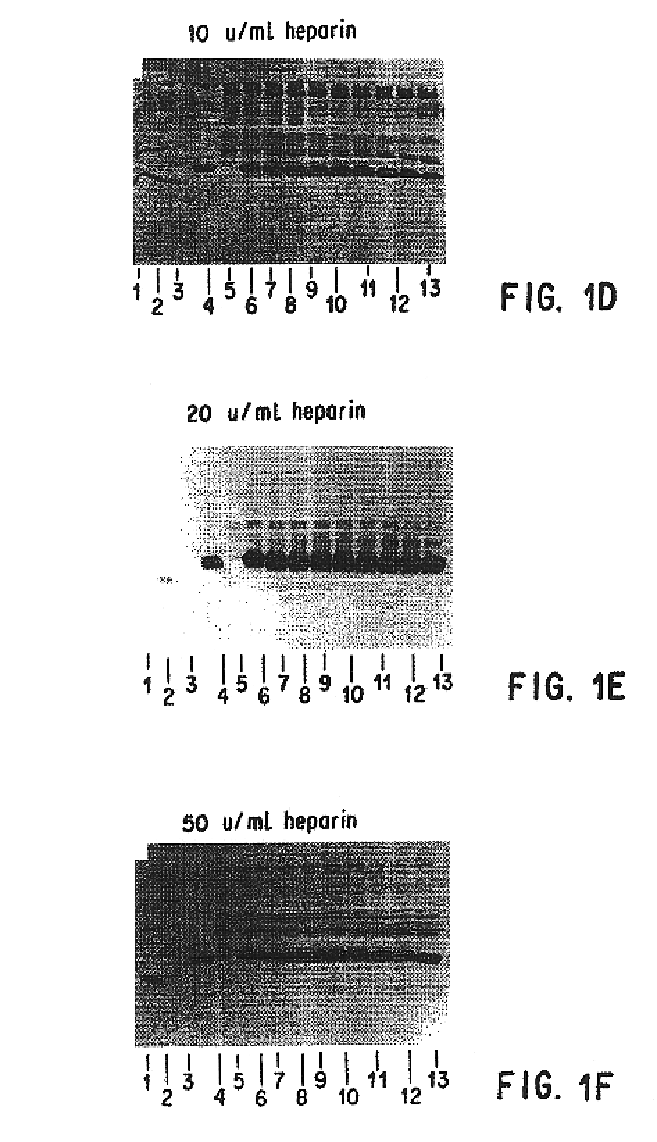
[0249]It was necessary to add an ingredient to the FG that would inhibit or prevent the digestion of HBGF-1β by thrombin (Lobb, Biochem. 27:2572-2578 (1988)), which is a component of FG. Heparin, which adsorbs to HBGF-1, was selected and tested to determine whether it could protect HBGF-1 from digestion by thrombin and any other proteolytic components of the FG. The stability of HBGF-1 in the presence of increasing concentrations of heparin was assessed.
[0250]Solutions containing HBGF-1β (10 μg / ml), thrombin (250 U / ml), and increasing concentrations of heparin (0, 0.5, 5, 10, 20, and 50 U / ml) were incubated at 37° C. Aliquots were periodically removed from the incubating solutions and were frozen and stored at −70° C. for further testing.
[0251]After the incubation was complete, the samples were thawed and separated on 15 % SDS polyacrylamide gels under reducing conditions according to the method of Laemmli (Nature 227:680 (1970)). The gel was then electroblotted o...
example 3
The Biological Activity of HBGF-1β after Incubation in the Presence of Heparin and Thrombin
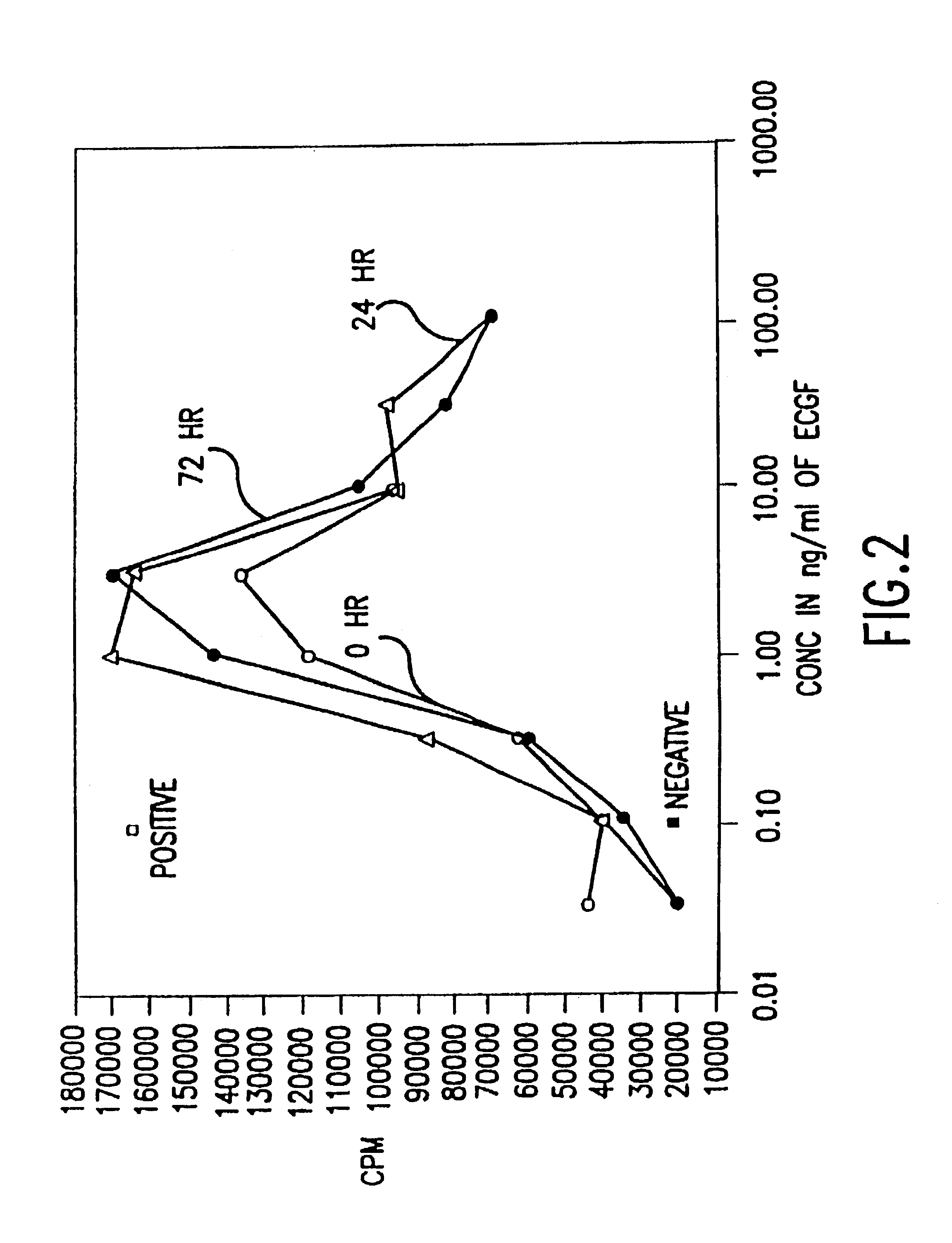
[0252]The biological activity of HBGF-1 in the incubation mixture that contained 5 U / ml of heparin, and was described in Example 2, was measured using an 3H-thymidine incorporation assay with NIH 3T3 cells.
[0253]NIH 3T3 cells were introduced into 96 well plates and were incubated at 37° C. under starvation conditions in Dulbecco's Modified Medium (DMEM; GIBCO, Grand island, N.Y.) with 0.5% fetal bovine serum (BCS; GIBCO, Grand Island, N.Y.) until the cells reached 30 to 50% confluence. Two days later, varying dilutions of HBGF-1 from the samples prepared in Example 2 were added to each well without changing the medium. Diluent (incubation buffer) was added in place of growth factor for the negative controls and DMEM with 10% BCS, which contains growth factors needed for growth, was added in place of the HBGF-1 sample for the positive controls.
[0254]After incubation at 37° C. for 18 hours, 0.25...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| concentrations | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More