Patents
Literature
659 results about "Regulatory T cell" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
The regulatory T cells (Tregs /ˈtiːrɛɡ/), formerly known as suppressor T cells, are a subpopulation of T cells that modulate the immune system, maintain tolerance to self-antigens, and prevent autoimmune disease. Tregs are immunosuppressive and generally suppress or downregulate induction and proliferation of effector T cells. Tregs express the biomarkers CD4, FOXP3, and CD25 and are thought to be derived from the same lineage as naïve CD4 cells. Because effector T cells also express CD4 and CD25, Tregs are very difficult to effectively discern from effector CD4+, making them difficult to study. Recent research has found that the cytokine TGFβ is essential for Tregs to differentiate from naïve CD4+ cells and is important in maintaining Treg homeostasis.
Constitutive expression of costimulatory ligands on adoptively transferred T lymphocytes
ActiveUS8252592B2Increase self-toleranceIncrease and reduces immune responseFused cellsFermentationRegulatory T cellLymphocyte
Owner:MEMORIAL SLOAN KETTERING CANCER CENT
Composition for inducing proliferation or accumulation of regulatory t cells
InactiveUS20130149339A1Induced proliferationInduce accumulationAntibacterial agentsCompound screeningRegulatory T cellT cell
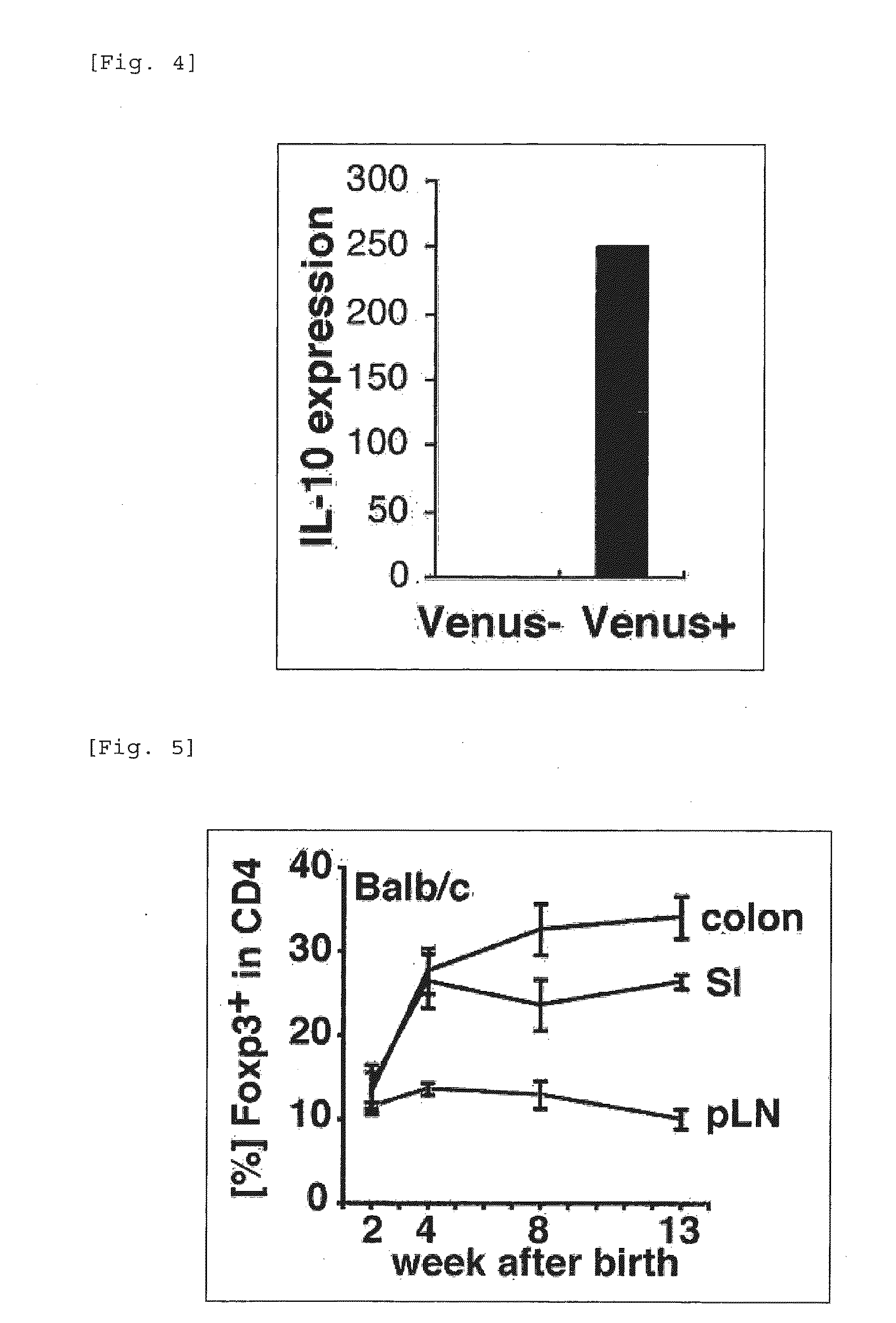
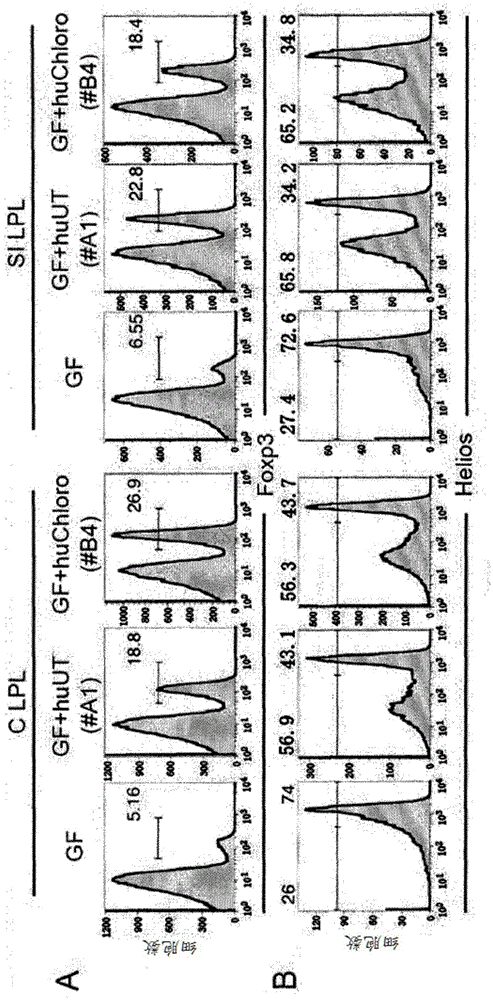
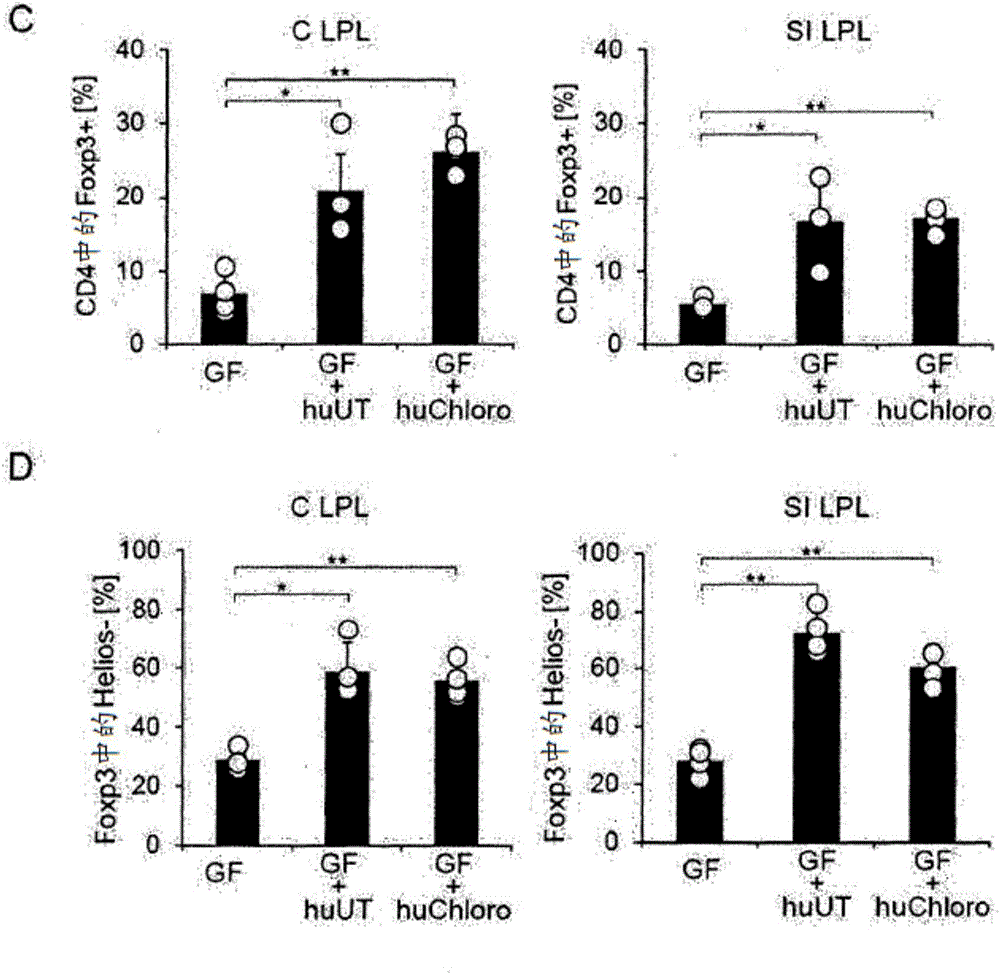
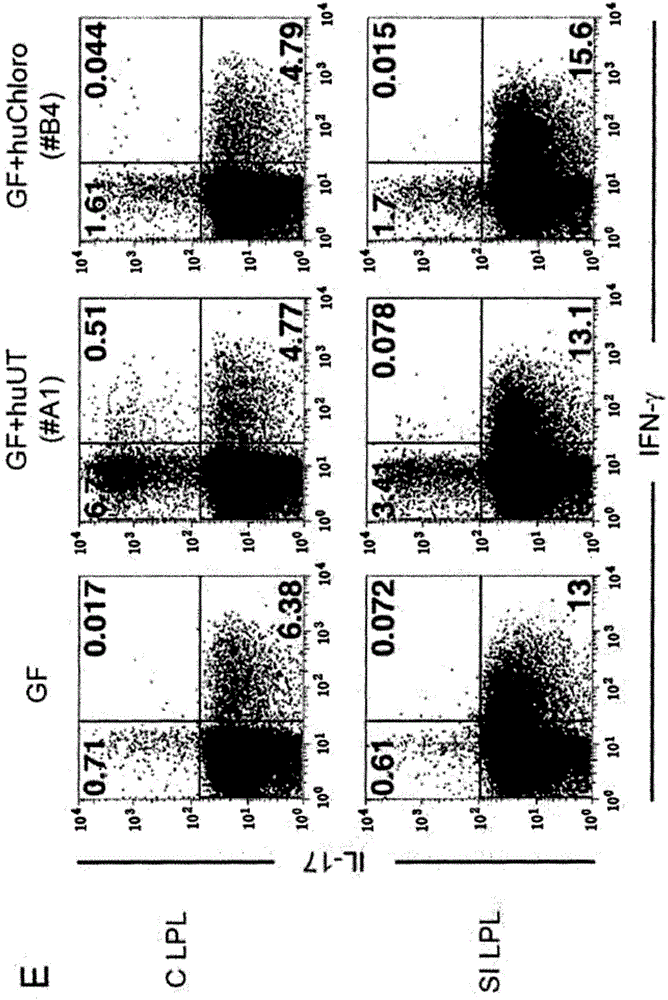
It was found that bacteria belonging to the genus Clostridium induce accumulation of regulatory T cells (Treg cells) in the colon. Moreover, the present inventors found that regulatory T cells (Treg cells) induced by from these bacteria suppressed proliferation of effector T-cells. From these findings, the present inventors found that the use of bacteria belonging to the genus Clostridium or a physiologically active substance derived therefrom made it possible to induce proliferation or accumulation of regulatory T cells (Treg cells), and further to suppress immune functions.
Owner:THE UNIV OF TOKYO
Compositions and Methods for Targeted Immunomodulatory Antibodies and Fusion Proteins
ActiveUS20130039911A1Function increaseOrganic active ingredientsPeptide/protein ingredientsTumor targetAntitumor immunity
The present invention is based on the seminal discovery that targeted immunomodulatory antobodies and fusion proteins can counter act or reverse immune tolerance of cancer cells. Cancer cells are able to escape elimination by chemotherapeutic agents or tumor-targeted antobodies via specific immunosuppressive mechanisms in the tumor microenvironment and such ability of cancer cells is recognized as immune tolerance. Such immuno-suppressive mechanisms include immunosuppressive cytokines (for example, Transforming growth factor beta (TGF-β)) and regulatory T cells and / or immunosuppressive myeloid dendritic cells (DCs). By conteracting tumor-induced immune tolerance, the present invention provides effective compositions and methods for cancer treatment, optional in combination with another existing cancer treatment. The present invention provides strategies to counteract tumor-induced immune tolerance and enhance the antitumor efficacy of chemotherapy by activating and leveraging T cell-mediated adaptive antitumor immunity against resistant or disseminated cancer cells.
Owner:THE JOHN HOPKINS UNIV SCHOOL OF MEDICINE
Composition for inducing proliferation or accumulation of regulatory t cells
ActiveUS20150143557A1Induce proliferation and accumulationImmunity in a living organism can be suppressedAntibacterial agentsCompound screeningRegulatory T cellT cell
It was found that bacteria belonging to the genus Clostridium induce accumulation of regulatory T cells (Treg cells) in the colon. Moreover, the present inventors found that regulatory T cells (Treg cells) induced by from these bacteria suppressed proliferation of effector T-cells. From these findings, the present inventors found that the use of bacteria belonging to the genus Clostridium or a physiologically active substance derived therefrom made it possible to induce proliferation or accumulation of regulatory T cells (Treg cells), and further to suppress immune functions.
Owner:THE UNIV OF TOKYO
Compositions and methods for targeted immunomodulatory antibodies and fusion proteins
ActiveUS8993524B2Function increaseOrganic active ingredientsPeptide/protein ingredientsTumor targetAntitumor immunity
The present invention is based on the seminal discovery that targeted immunomodulatory antibodies and fusion proteins can counter act or reverse immune tolerance of cancer cells. Cancer cells are able to escape elimination by chemotherapeutic agents or tumor-targeted antibodies via specific immunosuppressive mechanisms in the tumor microenvironment and such ability of cancer cells is recognized as immune tolerance. Such immune suppressive mechanisms include immunosuppressive cytokines (for example, Transforming growth factor beta (TGF-β) and regulatory T cells and / or immunosuppressive myeloid dendritic cells (DCs). By counteracting tumor-induced immune tolerance, the present invention provides effective compositions and methods for cancer treatment, optional in combination with another existing cancer treatment. The present invention provides strategies to counteract tumor-induced immune tolerance and enhance the antitumor efficacy of chemotherapy by activating and leveraging T cell-mediated adaptive antitumor immunity against resistant or disseminated cancer cells.
Owner:THE JOHN HOPKINS UNIV SCHOOL OF MEDICINE
Micro-RNAs modulating immunity and inflammation
InactiveUS8415096B2Effective redirectingTherapy is simpleOrganic active ingredientsSugar derivativesDiseaseRegulatory T cell
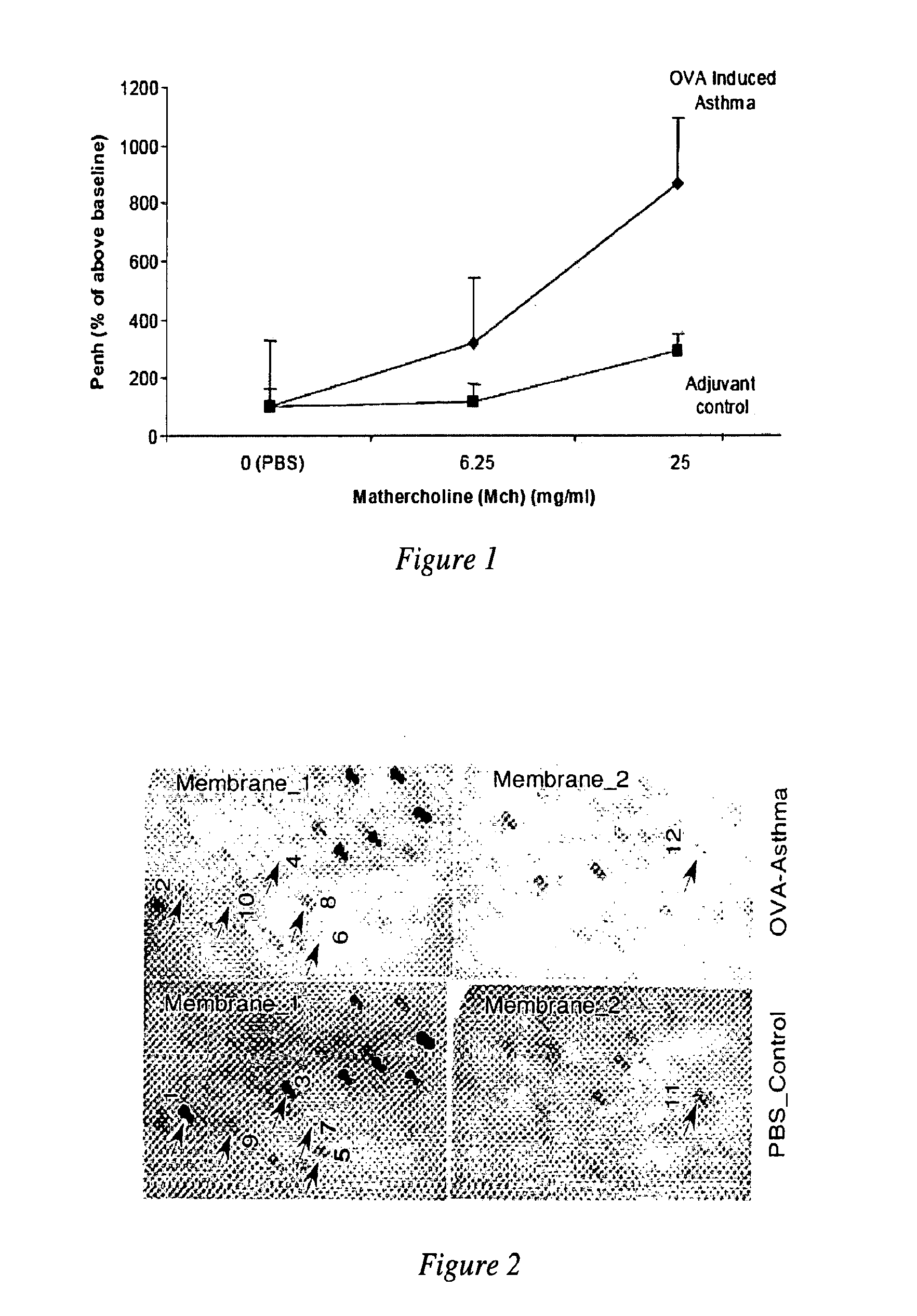
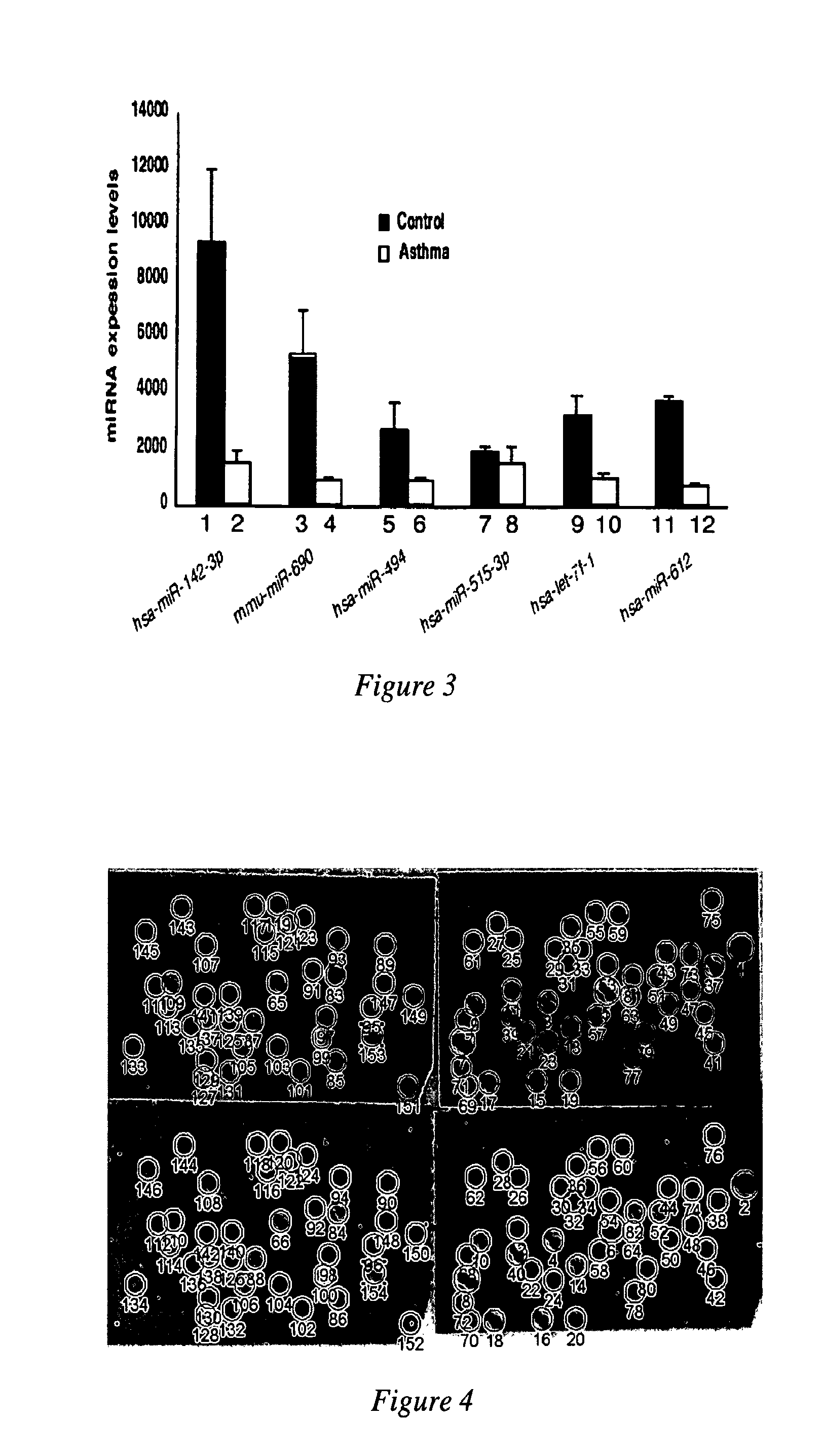
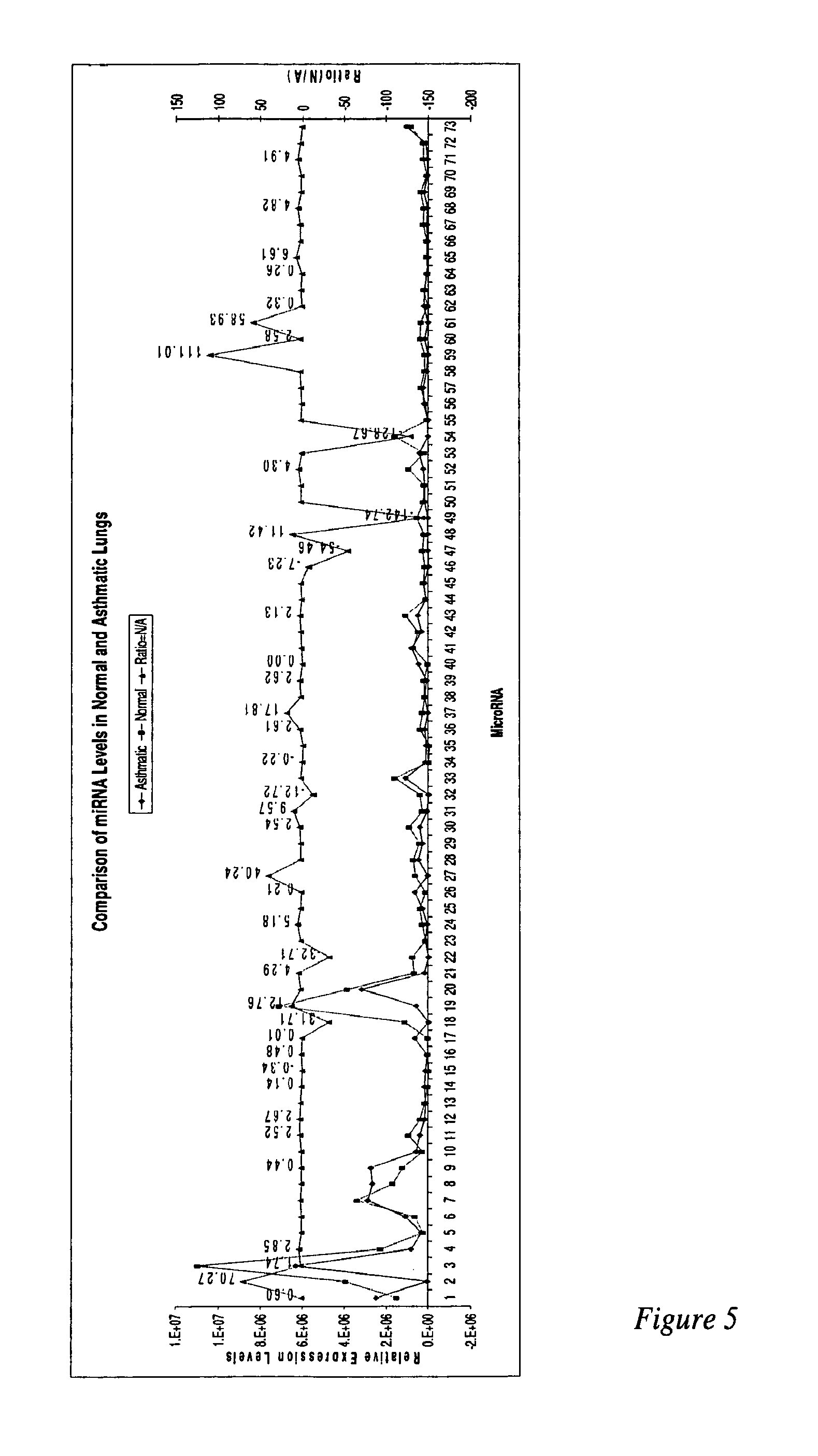
MicroRNAs are shown to be up- and / or down-regulated in inflammation and immune cells using a mouse model of asthma and regulatory T cells as source of RNA, respectively. Modulating the expression of these microRNAs can be effective in redirecting inflammation and immunity and hence, can be beneficial as biomarkers or as therapeutic agents against diverse human immunologic and inflammatory diseases.
Owner:UNIV OF SOUTH FLORIDA
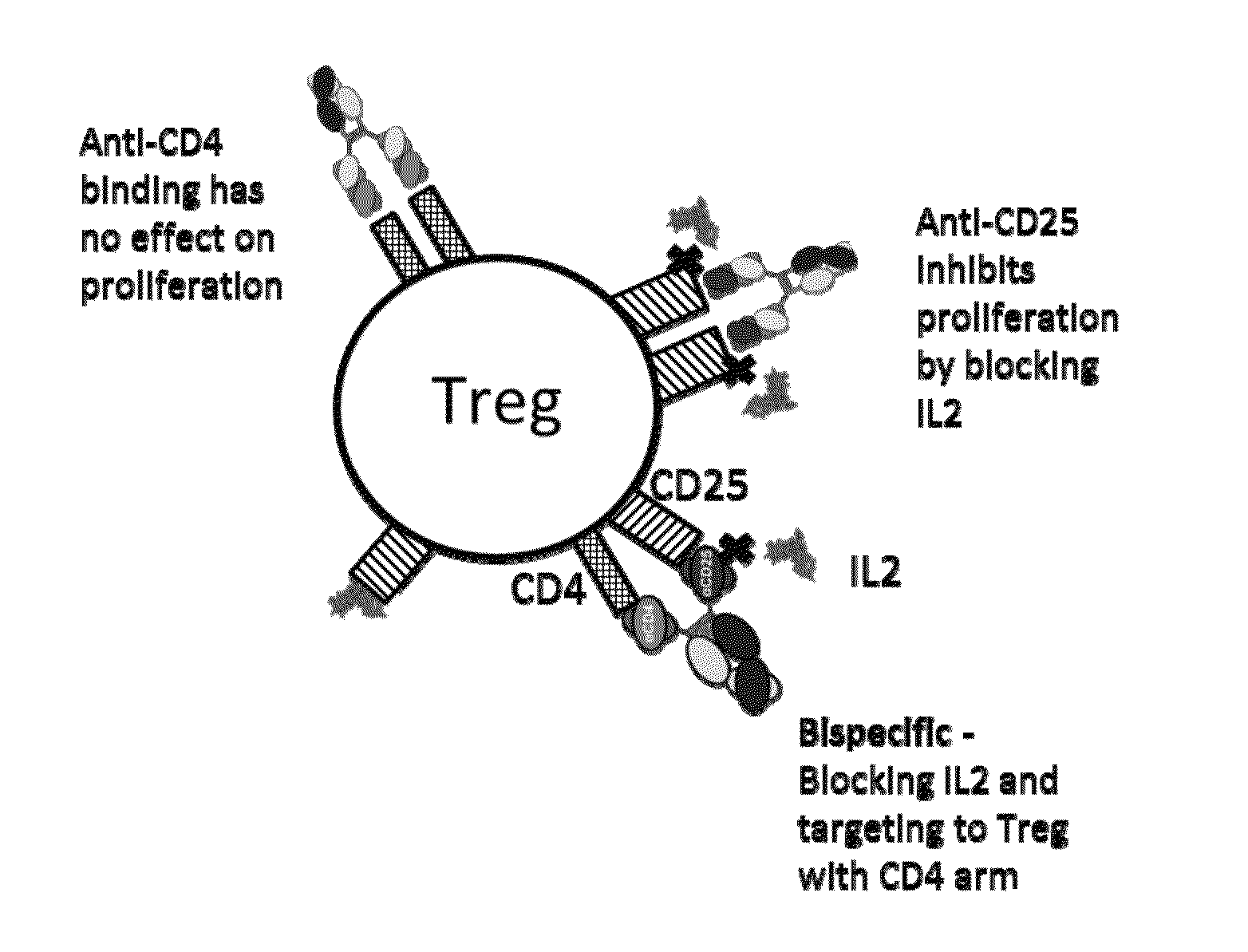
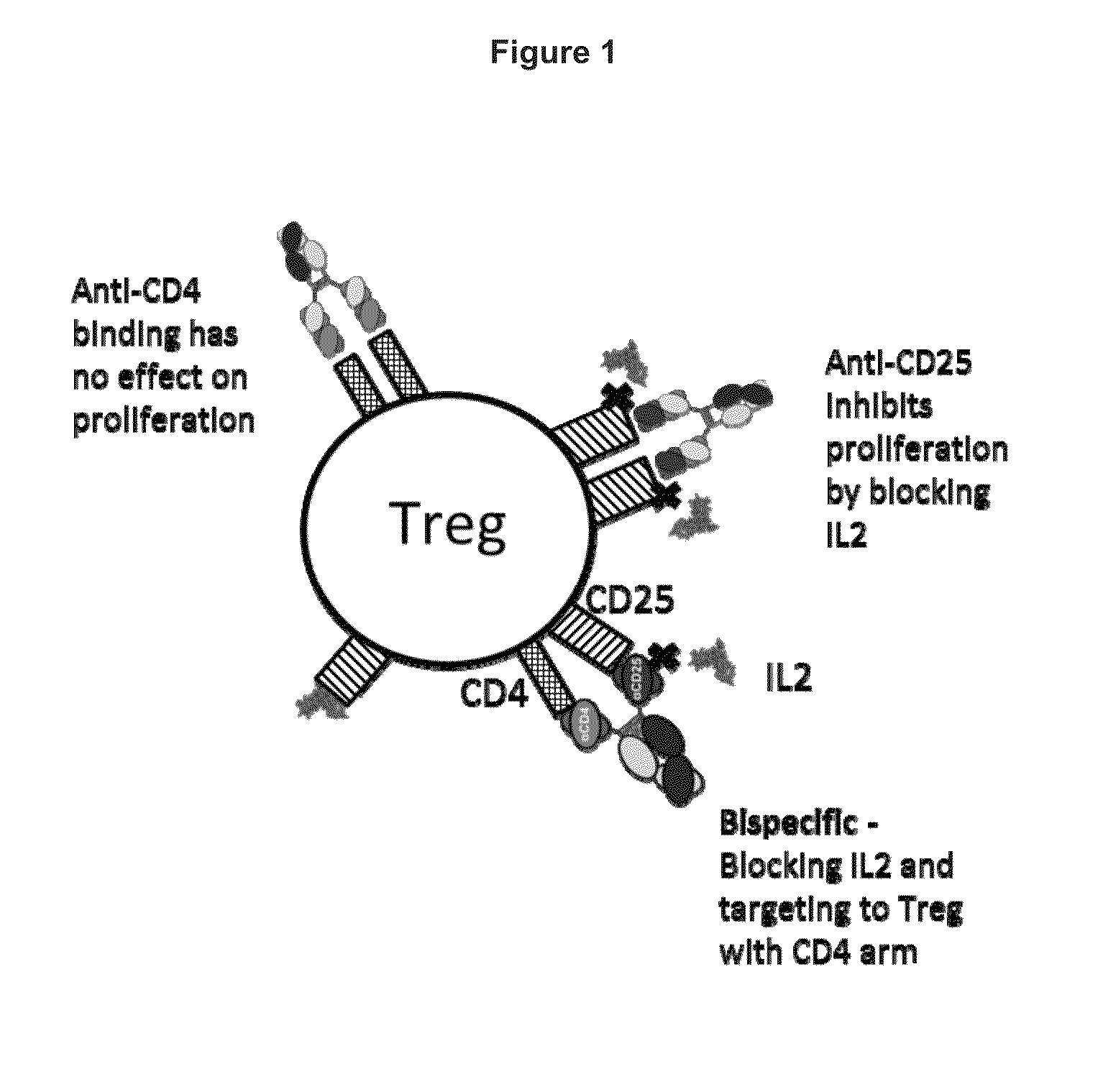
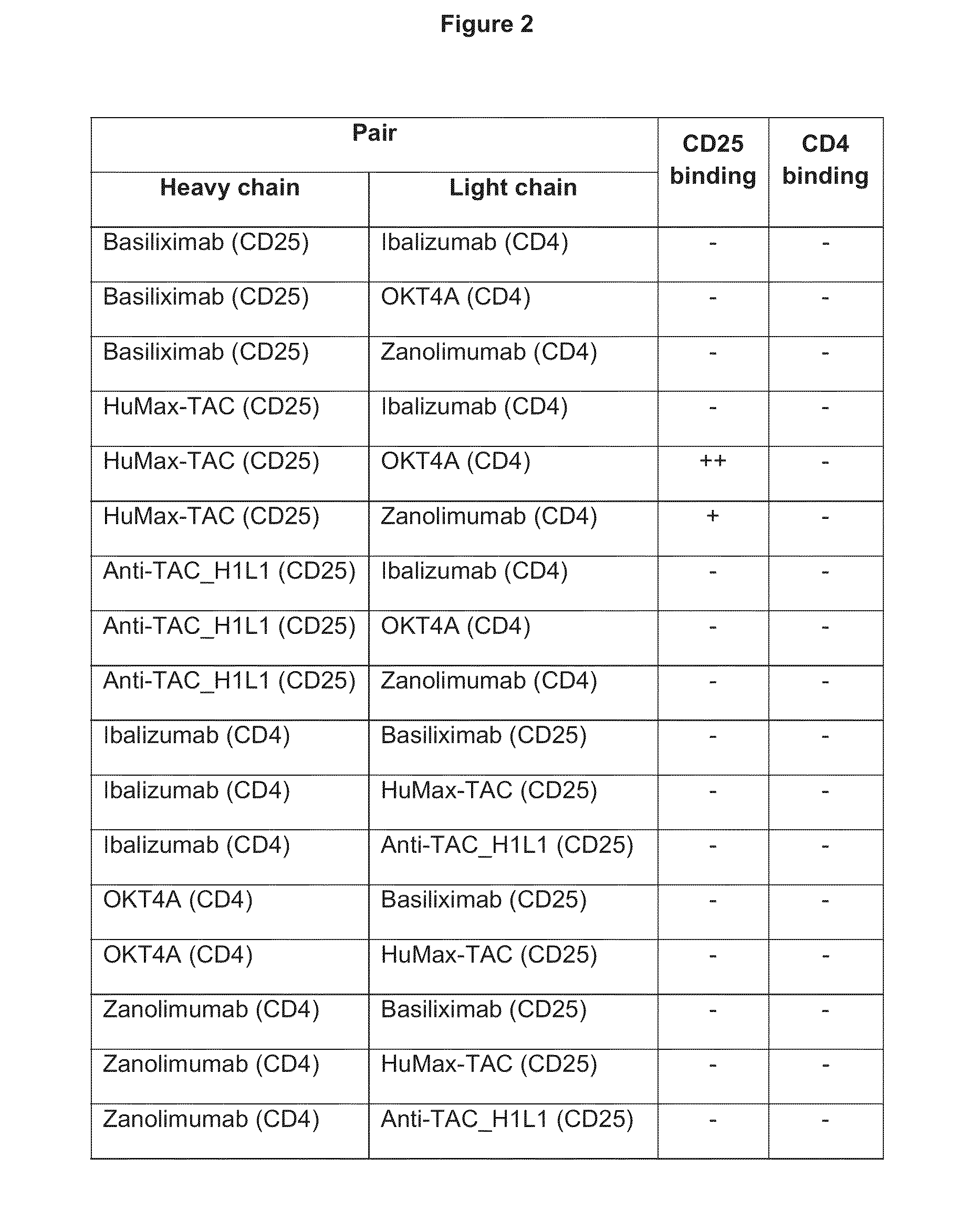
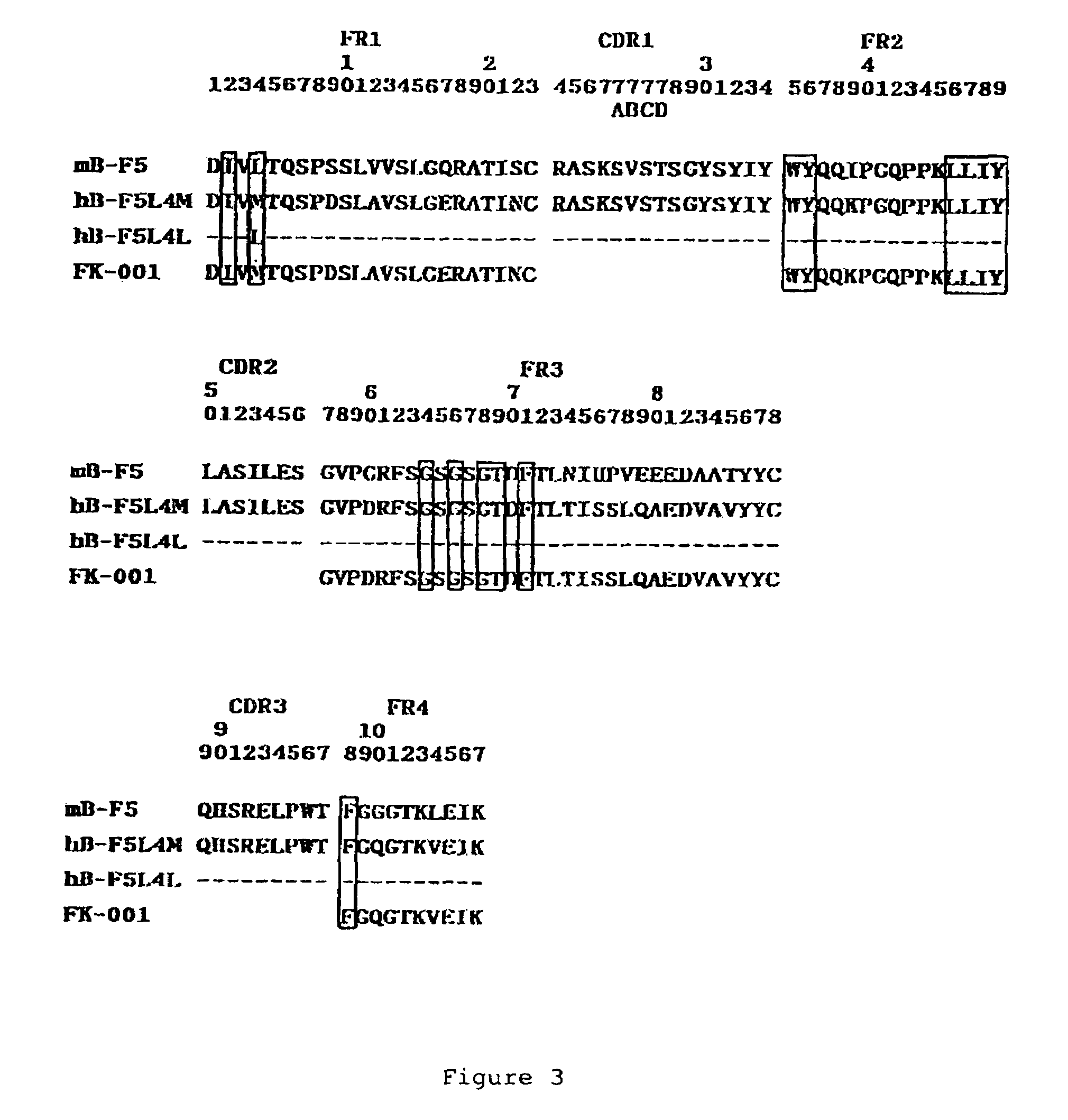
Humanized anti-CD4 antibody with immunosuppressive properties
A humanized antibody derived from mouse monoclonal anti-CD4 antibody B-F5 is able to activate CD25+CD4+ regulatory T cells and is useful for preparing immunosuppressive compositions.
Owner:OGEN GMBH
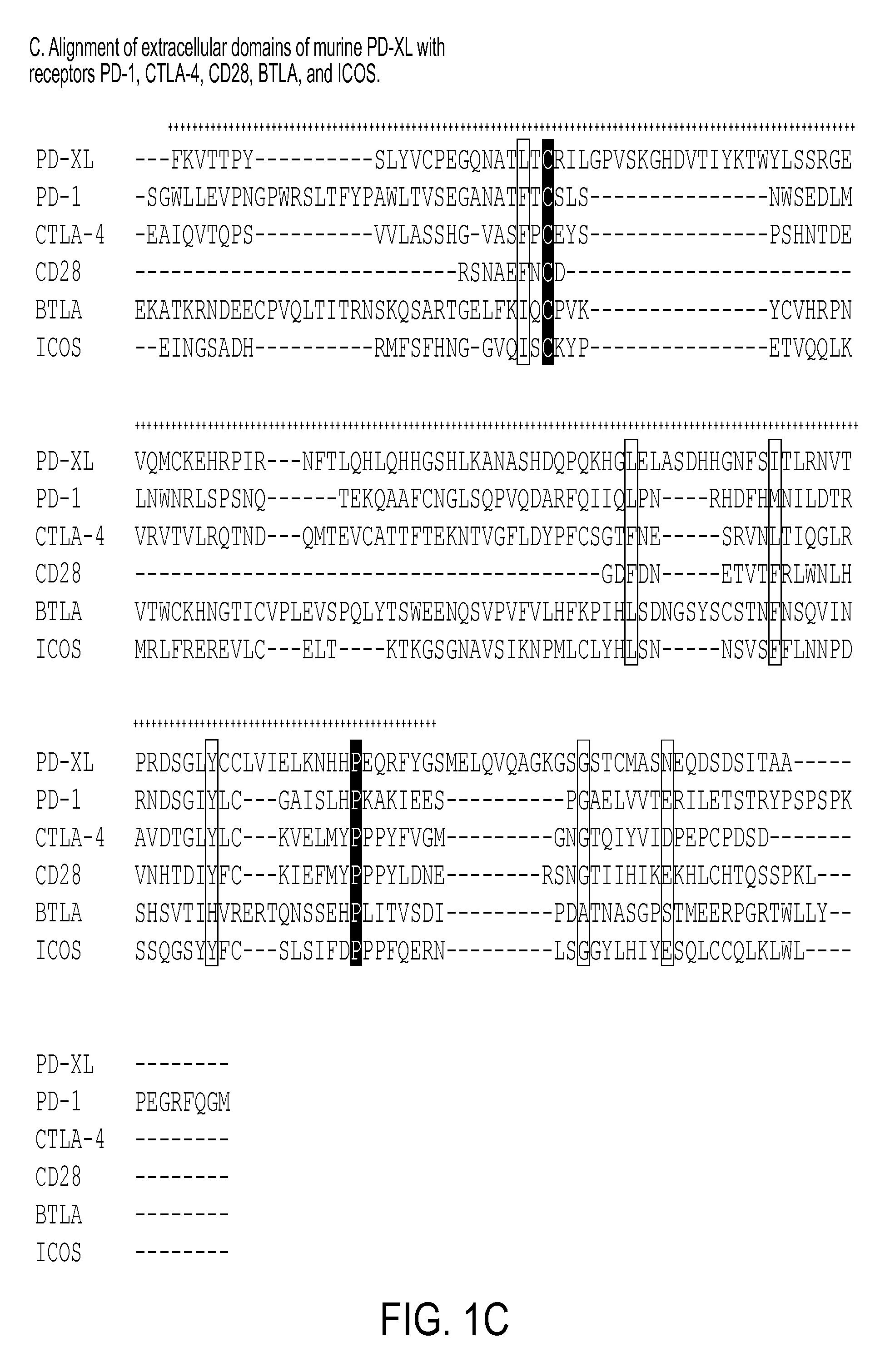
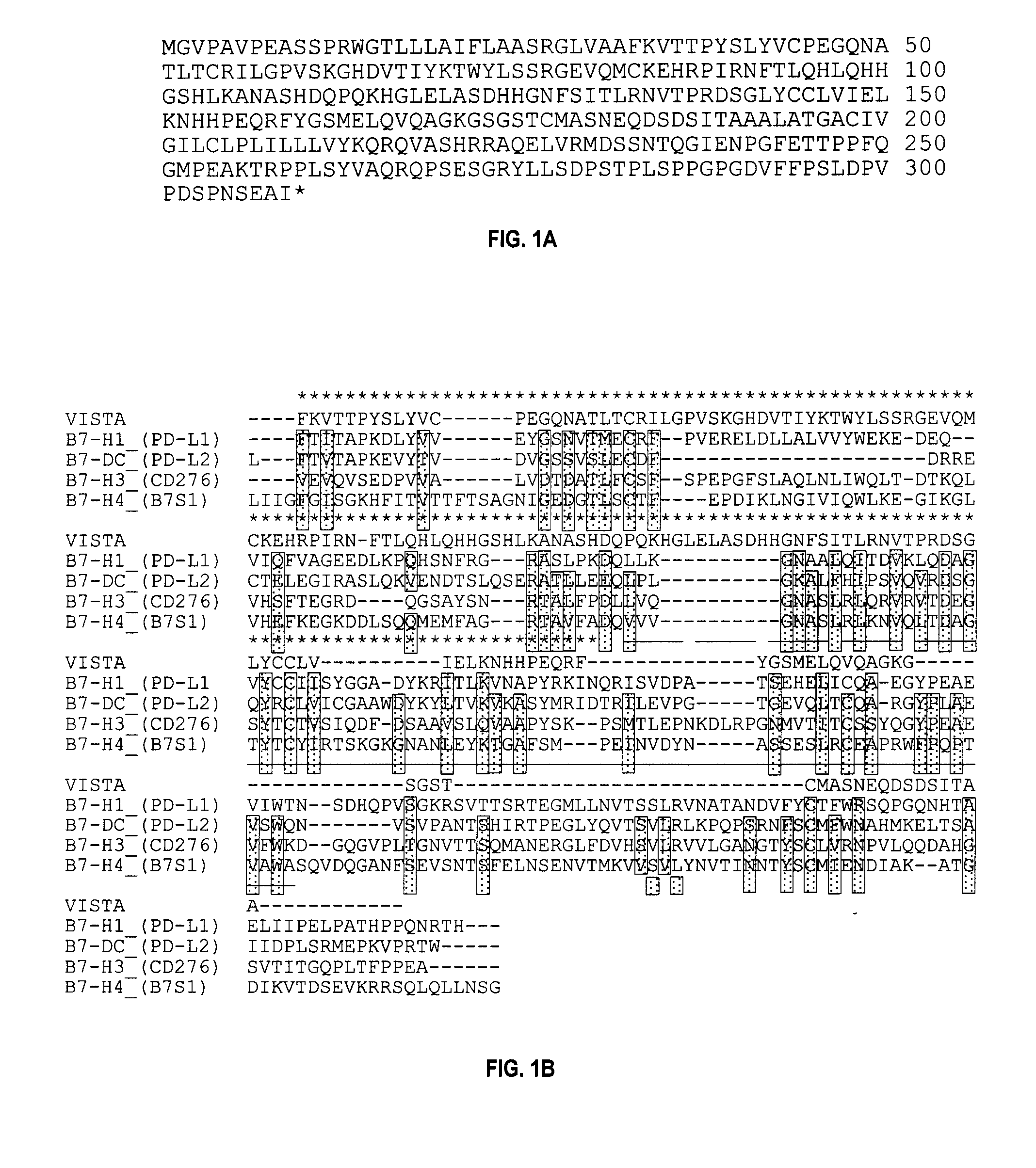
Use of PD-L3 proteins and PD-L3 specific antibodies or antibody fragments to regulate CD4+ and CD8+ T cell immunity
The present invention relates to novel regulatory T cell proteins. One protein, designated PD-L3, resembles members of the PD-L1 family, and co-stimulates αCD3 proliferation of T cells in vitro. A second, TNF-like, protein has also been identified as being upregulated upon αCD3 / αGITR stimulation. This protein has been designated Treg-sTNF. Proteins, antibodies, activated T cells and methods for using the same are disclosed.
Owner:TRUSTEES OF DARTMOUTH COLLEGE THE
Treatment of autoimmune disorders
Disclosed herein are methods for the treatment of autoimmune or immune related diseases or disorders. Also disclosed are methods for treating such autoimmune or immune related diseases or disorders with the administration of expanded populations of regulatory T cells. Also disclosed herein are methods of treating autoimmune or immune related diseases or disorders by administering an amount of expanded regulatory T cells to the body of a patient effective to reduce or prevent the symptoms of the autoimmune or immune related disease or disorder.
Owner:TORREY PINES INST FOR MOLECULAR STUDIES
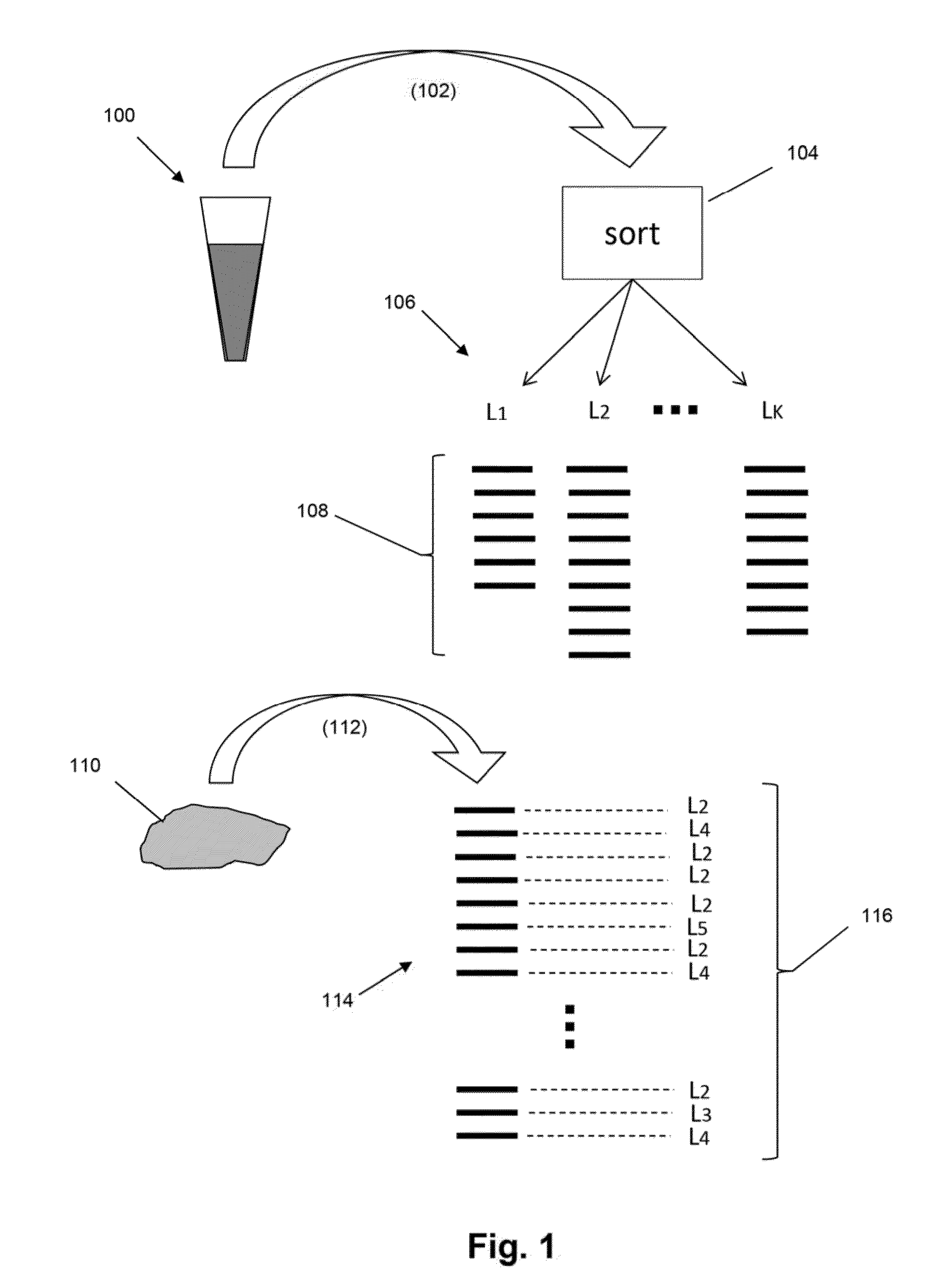
Detection and measurement of tissue-infiltrating lymphocytes
ActiveUS20130150252A1Microbiological testing/measurementLibrary member identificationDiseaseRegulatory T cell
The present invention is drawn to methods for measuring numbers, levels, and / or ratios of cells, such as lymphocytes, infiltrated into a solid tissue, such as a tumor or a tissue affected by an autoimmune disease, and to methods for making patient prognoses based on such measurements. In one aspect, methods of the invention comprise sorting lymphocytes from an accessible tissue, such as peripheral blood, into functional subsets, such as cytotoxic T cells and regulatory T cells, and generating clonotype profiles of each subset. An inaccessible disease-affected tissue is sampled and one or more clonotype profiles are generated. From the latter clonotype profiles, levels lymphocytes in each of the functional subsets are determined in the disease-affected tissue by their clonotypes, which are identified from lymphocytes sorted into subsets from the accessible tissue.
Owner:ADAPTIVE BIOTECH
Regulatory t cells and methods of making and using same
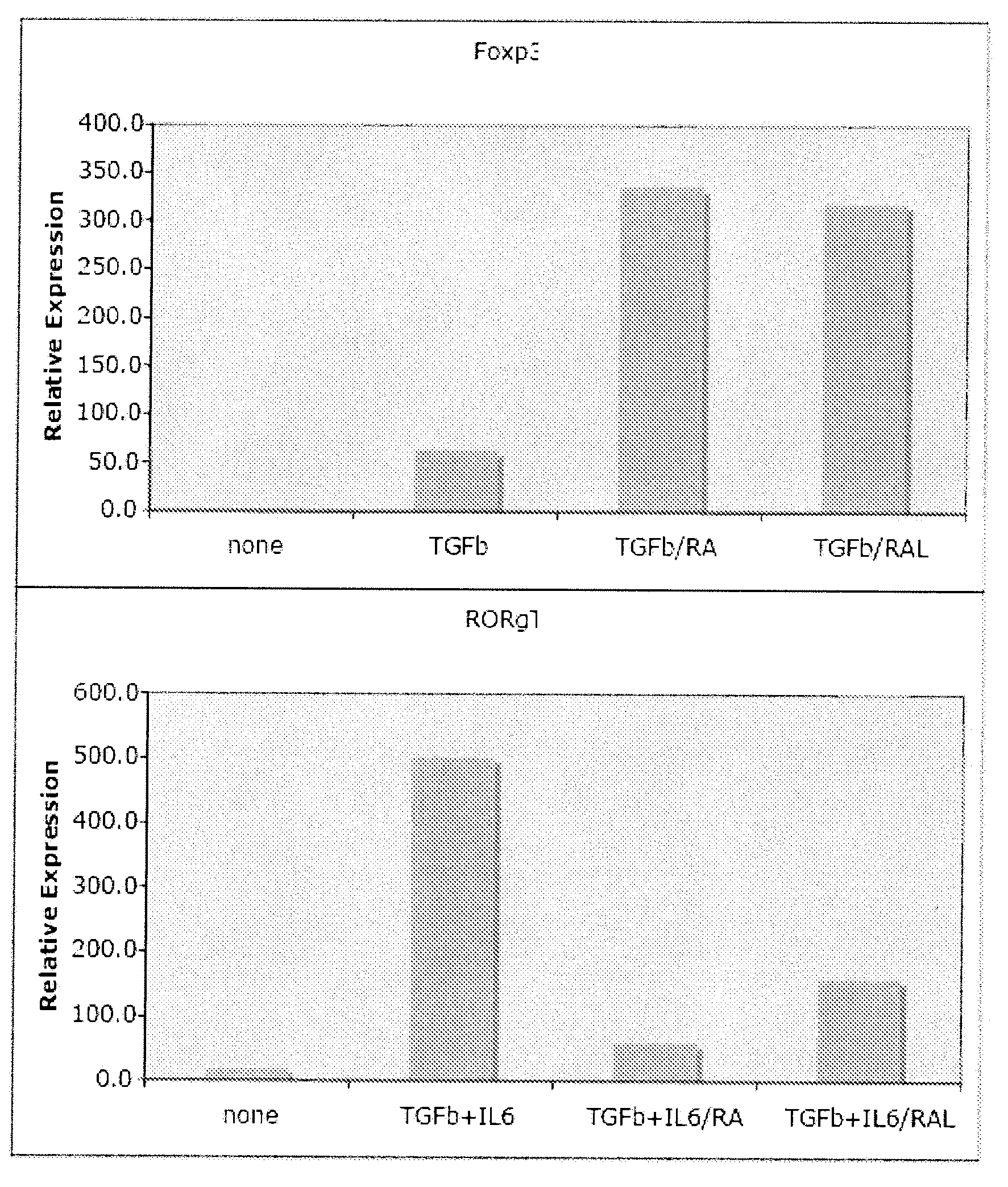
InactiveUS20090136470A1Inhibiting IL-6-driven induction of Th-1Adjust balanceBiocideSenses disorderRegulatory T cellPeroxisome
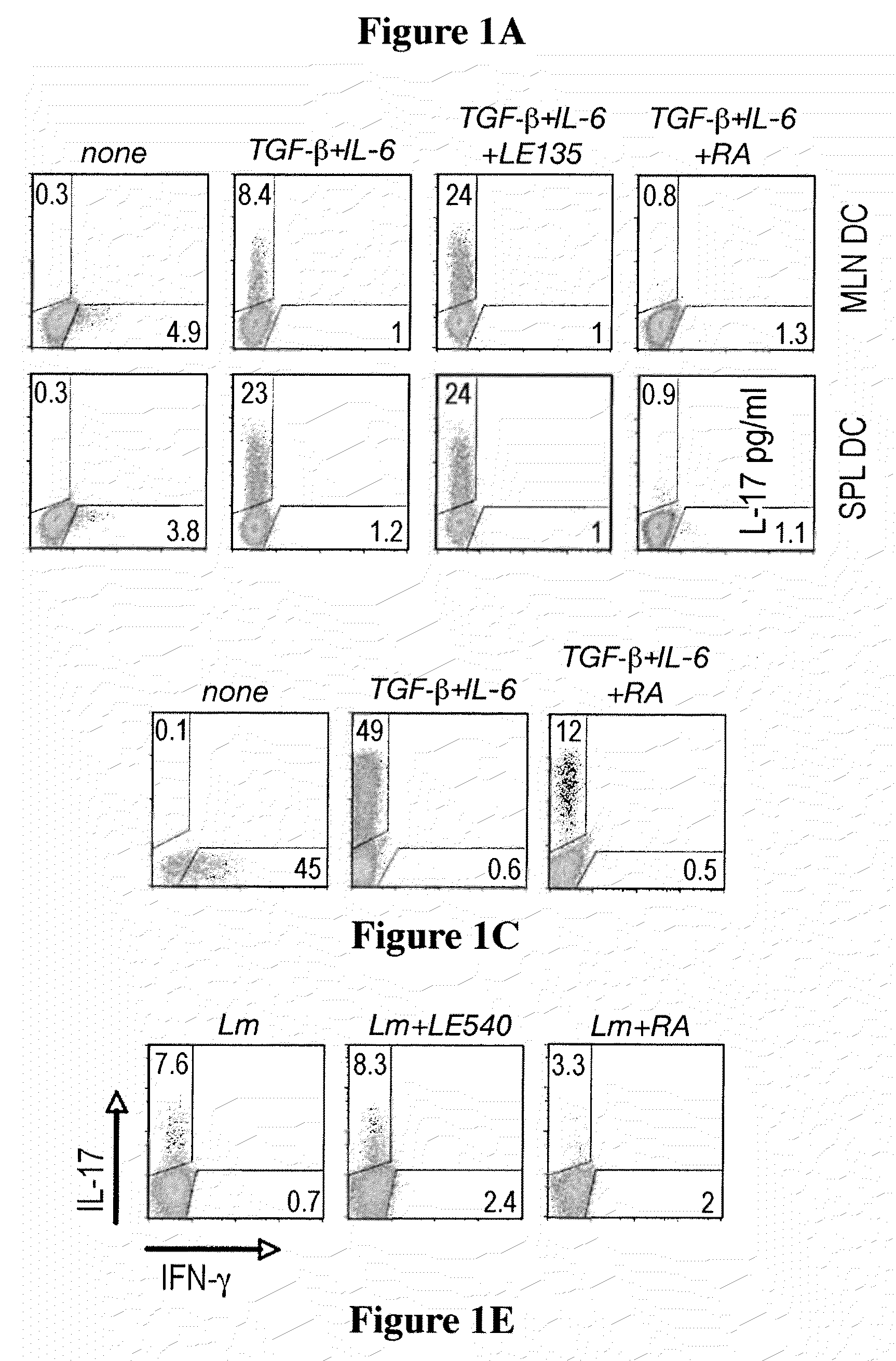
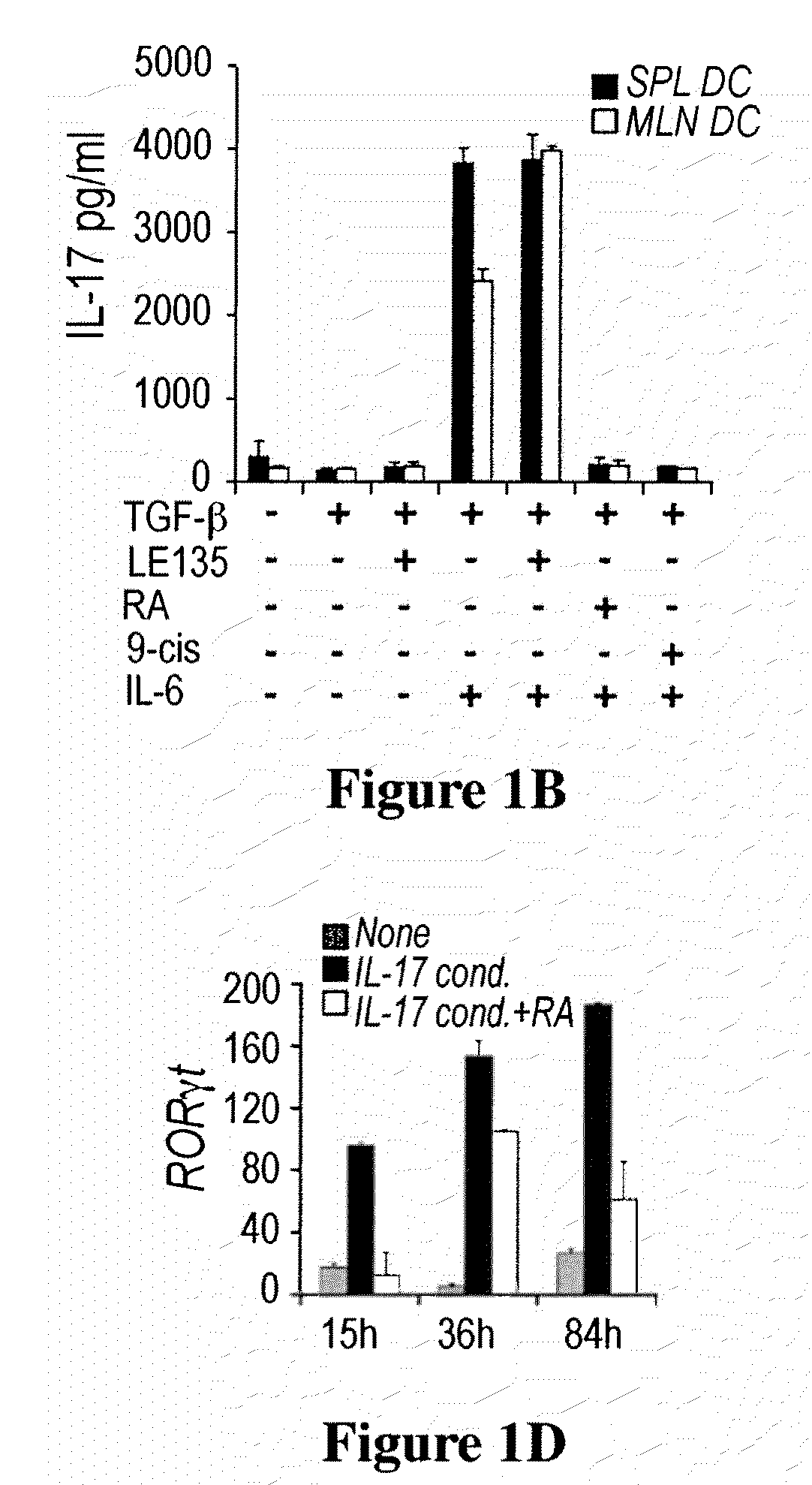
Methods of stimulating or increasing differentiation to regulatory T cells, cultures of regulatory T cells and methods of reducing or decreasing an immune response, inflammation or an inflammatory response, among other things, are provided. Methods include, among other things, contacting blood cells or T cells with an amount of TGF-beta or a TGF-beta analogue and a retinoic acid receptor agonist, or an amount of a retinoid X receptor (RXR) or peroxisome proliferator activated receptor-gamma (PPARgamma) agonist, sufficient to stimulate or increase differentiation to regulatory T cells. Cultures of regulatory T cells include T cells that express a marker associated with regulatory T cells, such as cultures in which regulatory T cells represent, for example, 30% or more of the total number of cells in the culture.
Owner:LA JOLLA INST FOR ALLERGY & IMMUNOLOGY
Human-derived bacteria that induce proliferation or accumulation of regulatory t cells
InactiveCN104160014APromote proliferationPromote accumulationBacteriaAntipyreticBacteroidesRegulatory T cell
Species of human-derived bacteria belonging to the Clostridia class have been shown to induce accumulation of regulatory T cells (Treg cells) in the colon and suppress immune functions. Pharmaceutical compositions containing these bacteria can be used to prevent and treat immune-mediated diseases such as autoimmune diseases.
Owner:THE UNIV OF TOKYO +1
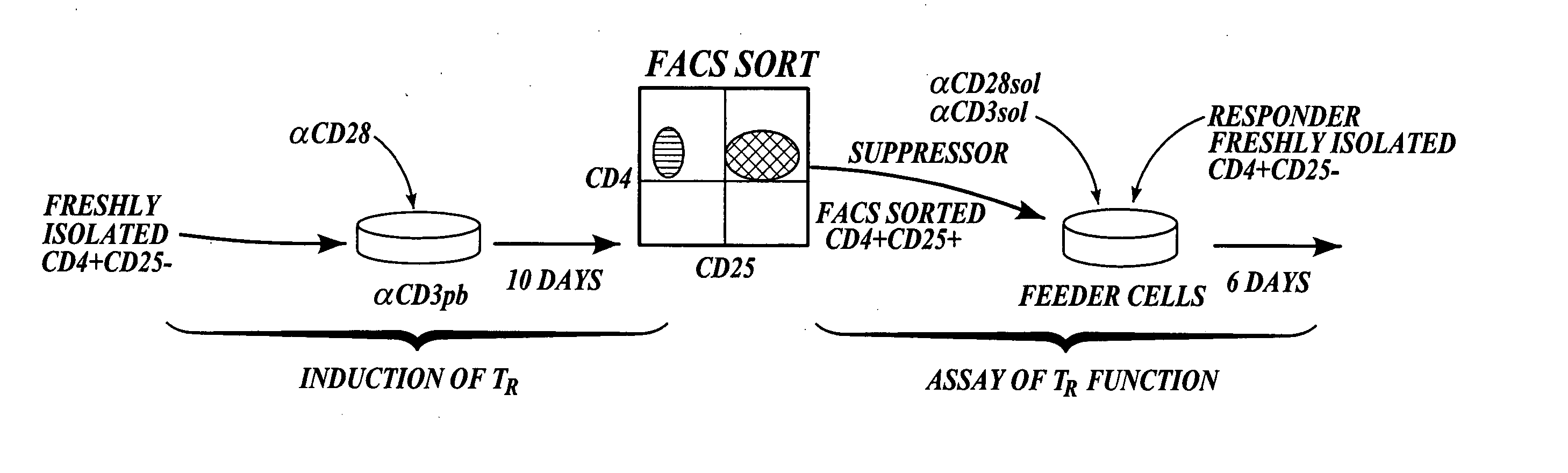
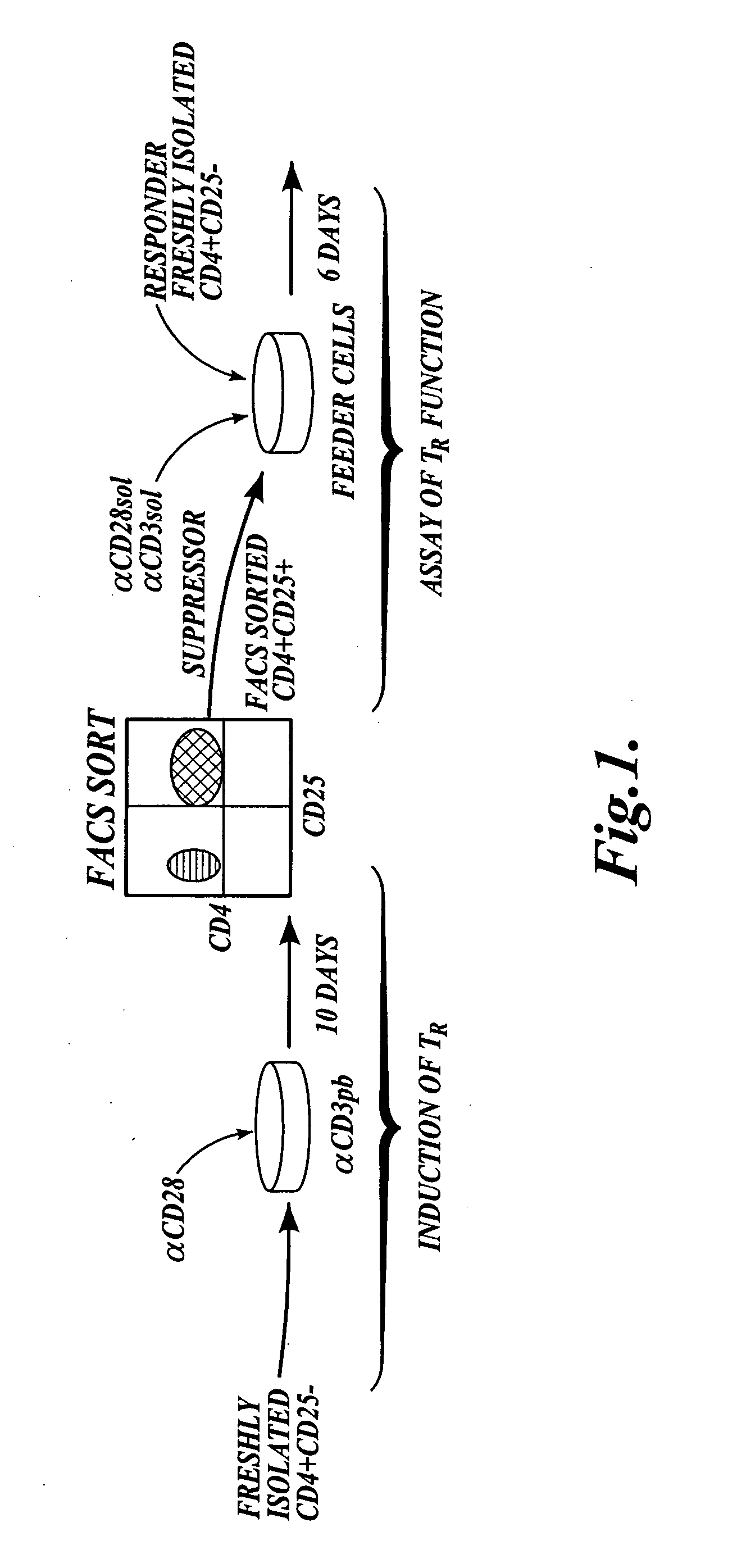
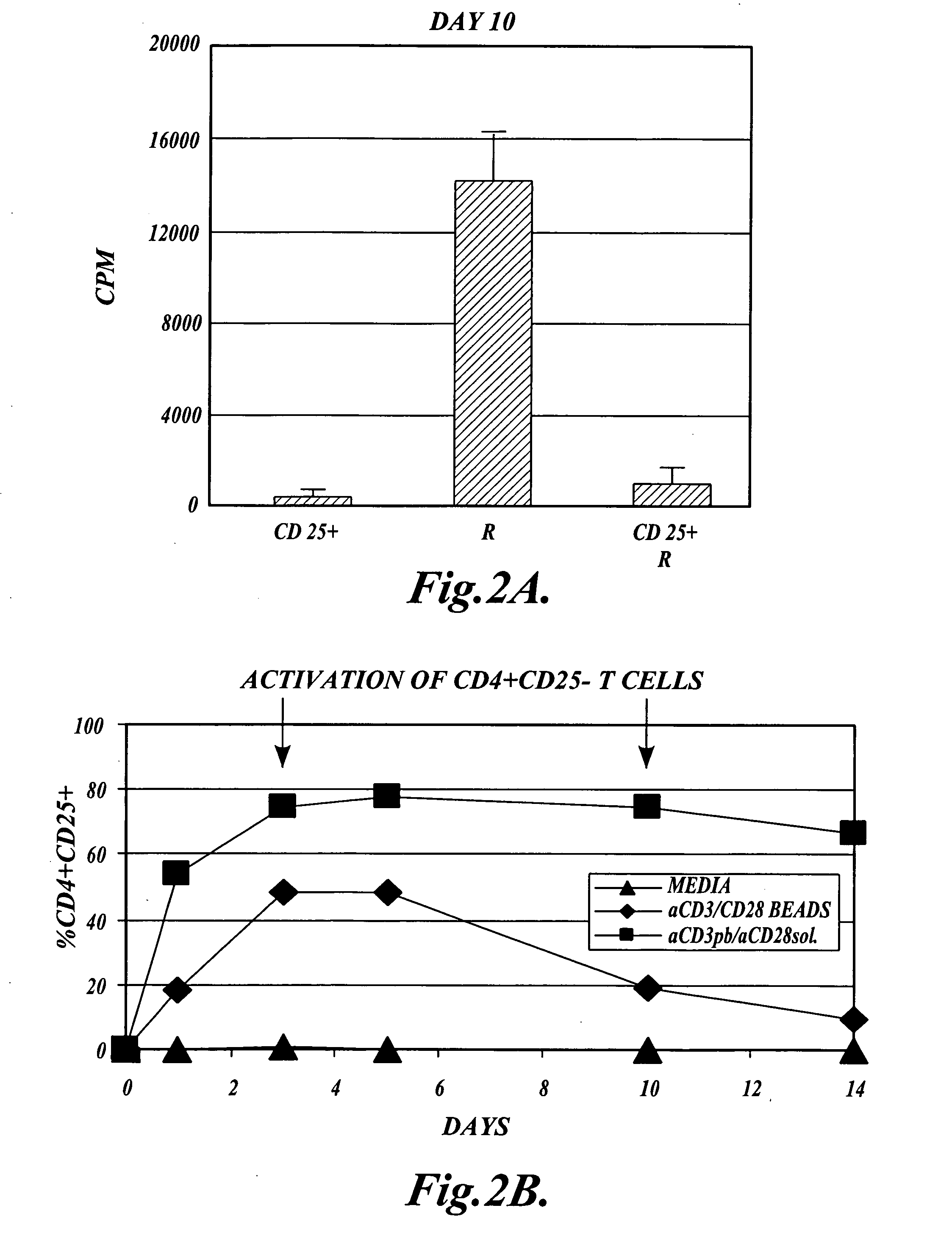
Methods of generating antigen-specific CD4+CD25+regulatory T cells, compositions and methods of use
ActiveUS8053235B2Reduce riskReduce severityArtificial cell constructsDead animal preservationRegulatory T cellSelect agent
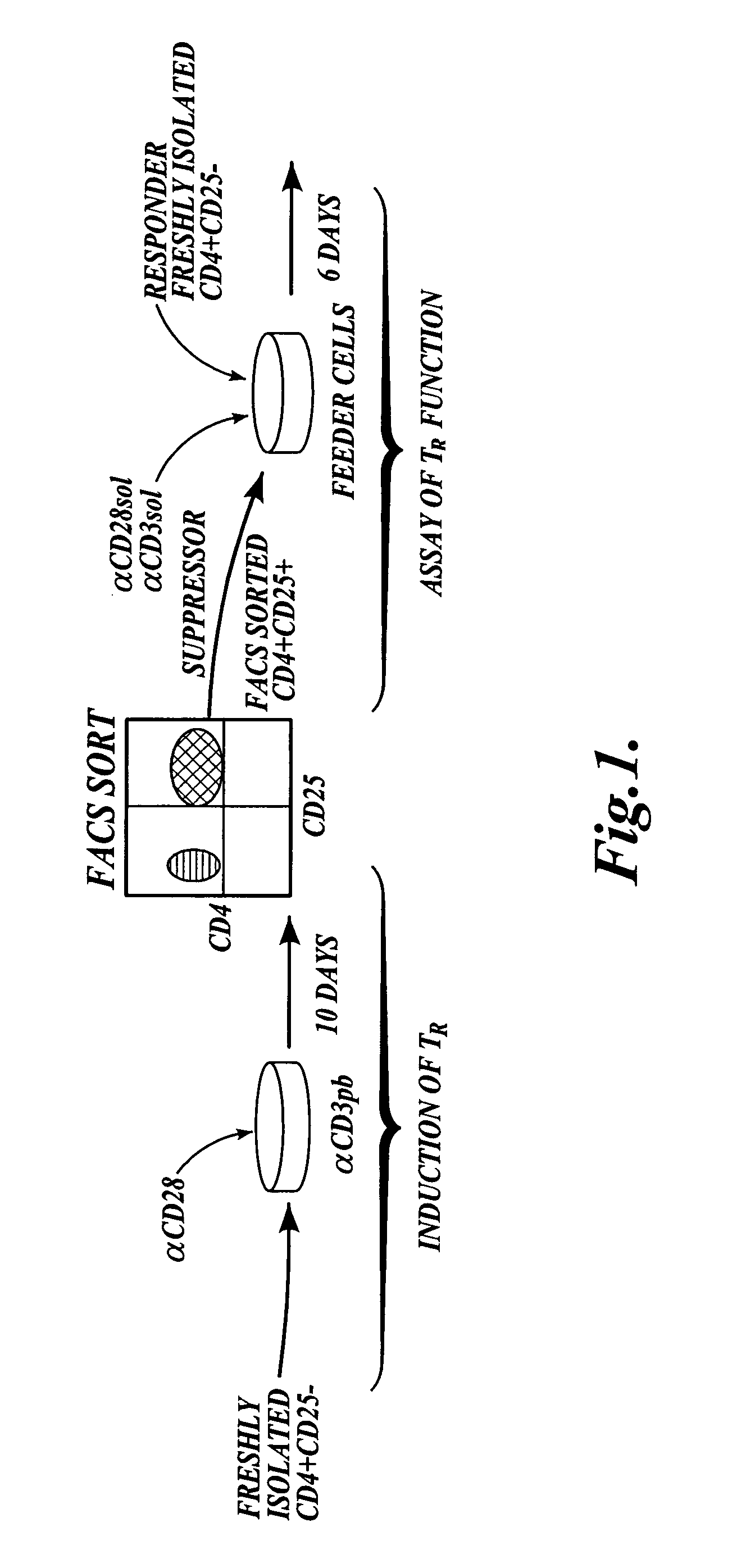
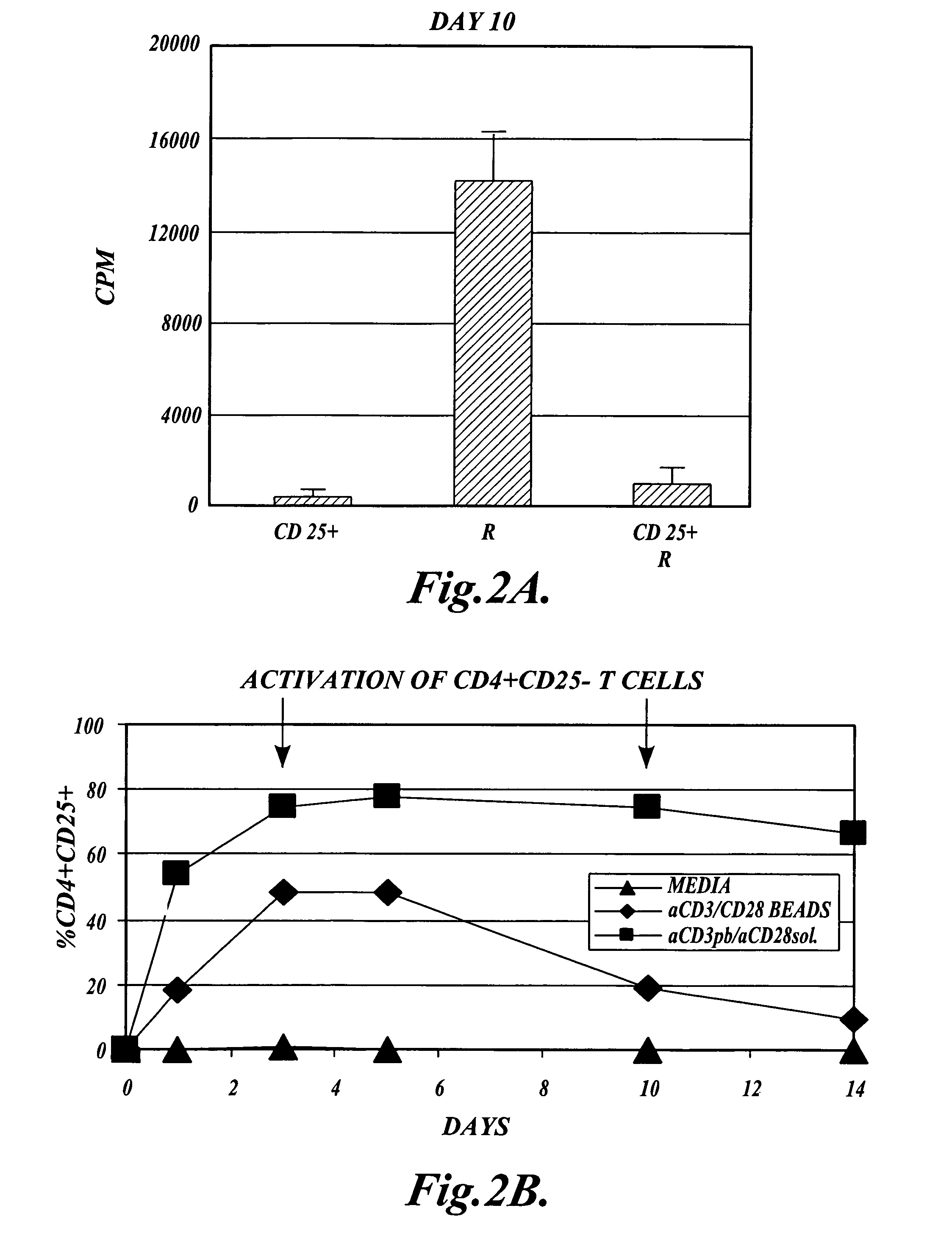
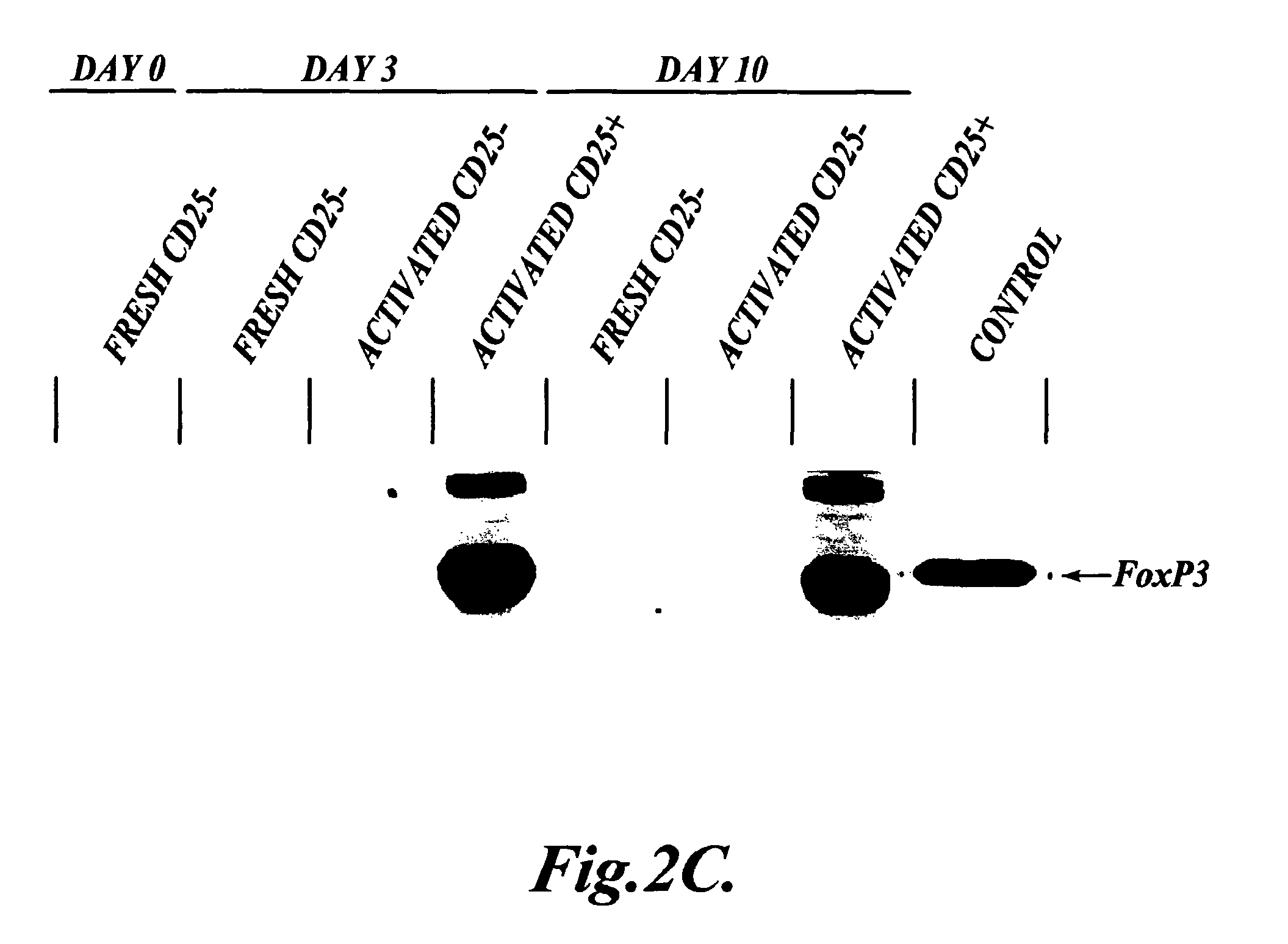
The present invention provides methods for generating mammalian T cell populations comprising antigen-specific CD4+CD25+ regulatory T cells from freshly isolated CD4+CD25− T cells. The method comprises selecting CD4+CD25− T cells from a sample obtained from a mammalian subject; determining the MHC Class II type of the subject; inducing the generation of antigen-specific regulatory T cells by contacting the isolated CD4+CD25− T cells in a culture vessel with an induction agent for a time period sufficient to generate antigen-specific CD4+CD25+ regulatory T cells; and selecting the CD4+CD25+ antigen-specific regulatory T cells by sorting the cells in the induction culture with a selection agent comprising at least one artificial multimeric MHC Class II / peptide complex that corresponds to the MHC Class II type of the subject.
Owner:BENAROYA RES INST AT VIRGINIA MASON
Method to inhibit T cell interactions with soluble B7
InactiveUS6887471B1Suppression problemPeptide/protein ingredientsAntibody mimetics/scaffoldsRegulatory T cellT cell mediated immunity
Owner:BRISTOL MYERS SQUIBB CO
Regulatory T cells suppress autoimmunity
InactiveUS20050186207A1Prevent proliferationBiocideMetabolism disorderRegulatory T cellAutoimmune responses
The invention provides methods for producing an autoantigen-specific regulatory T Cell enriched composition, and resultant compositions and methods of use.
Owner:RGT UNIV OF CALIFORNIA
Compositions and methods for immunotherapy
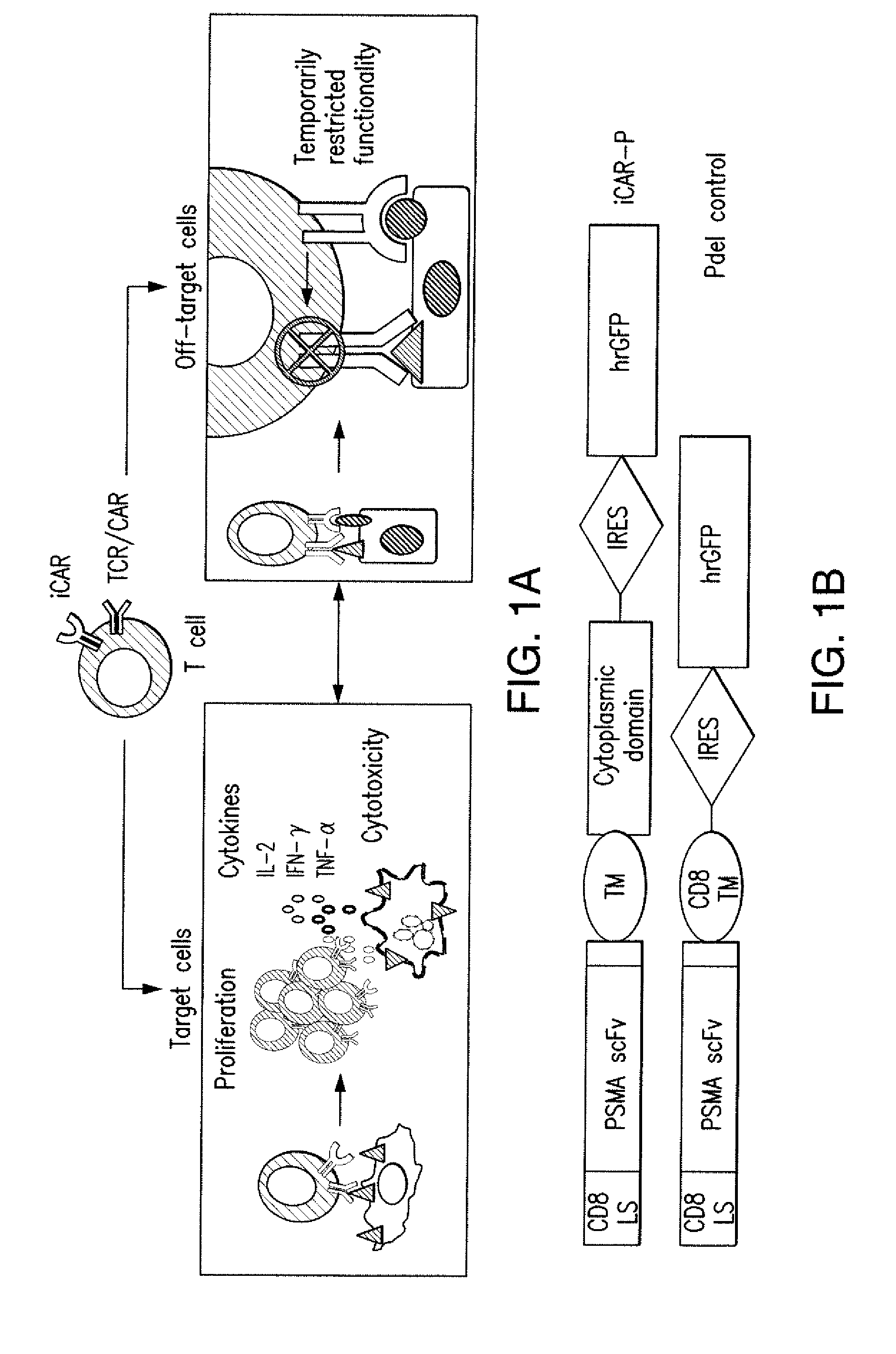
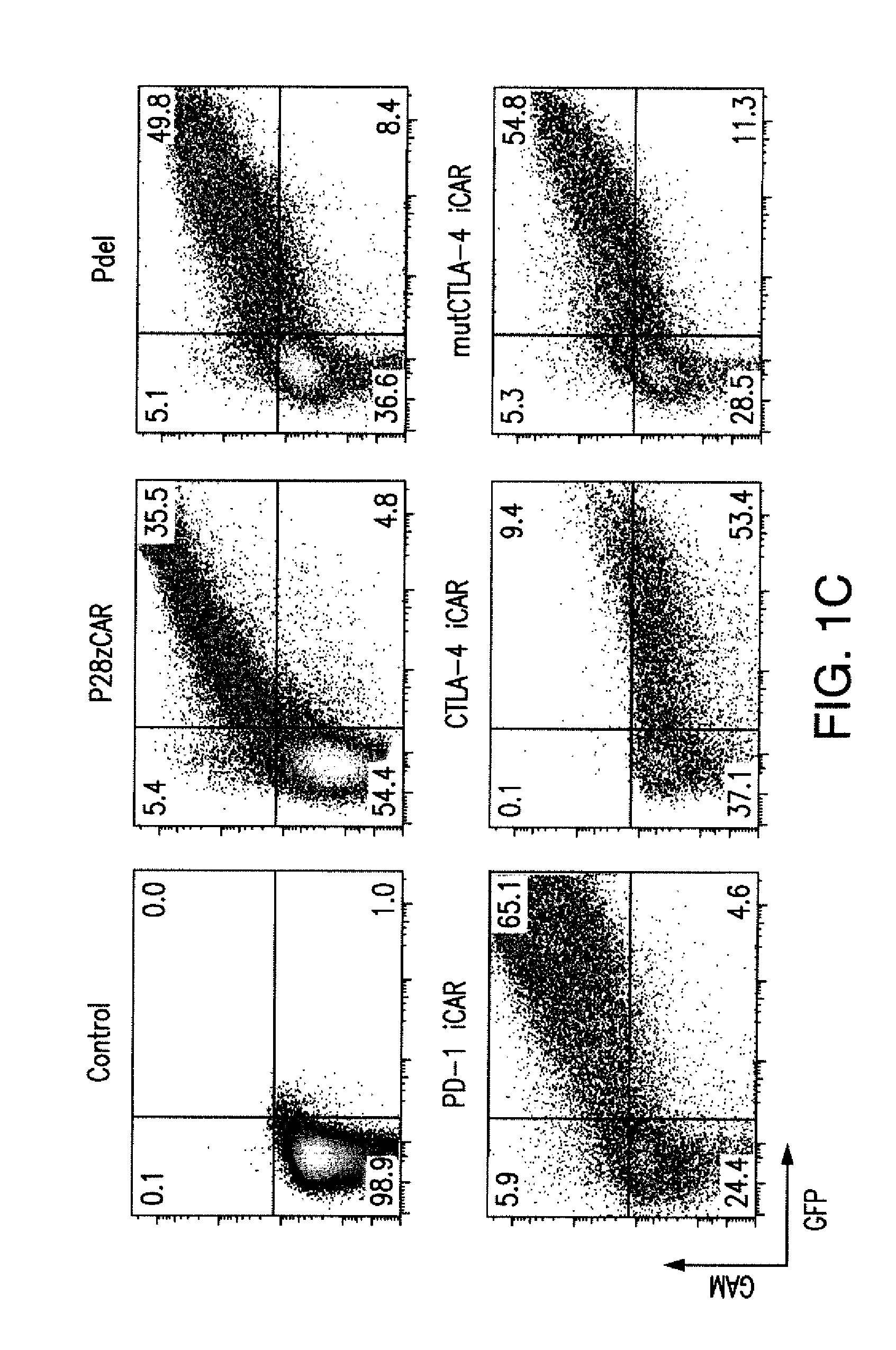
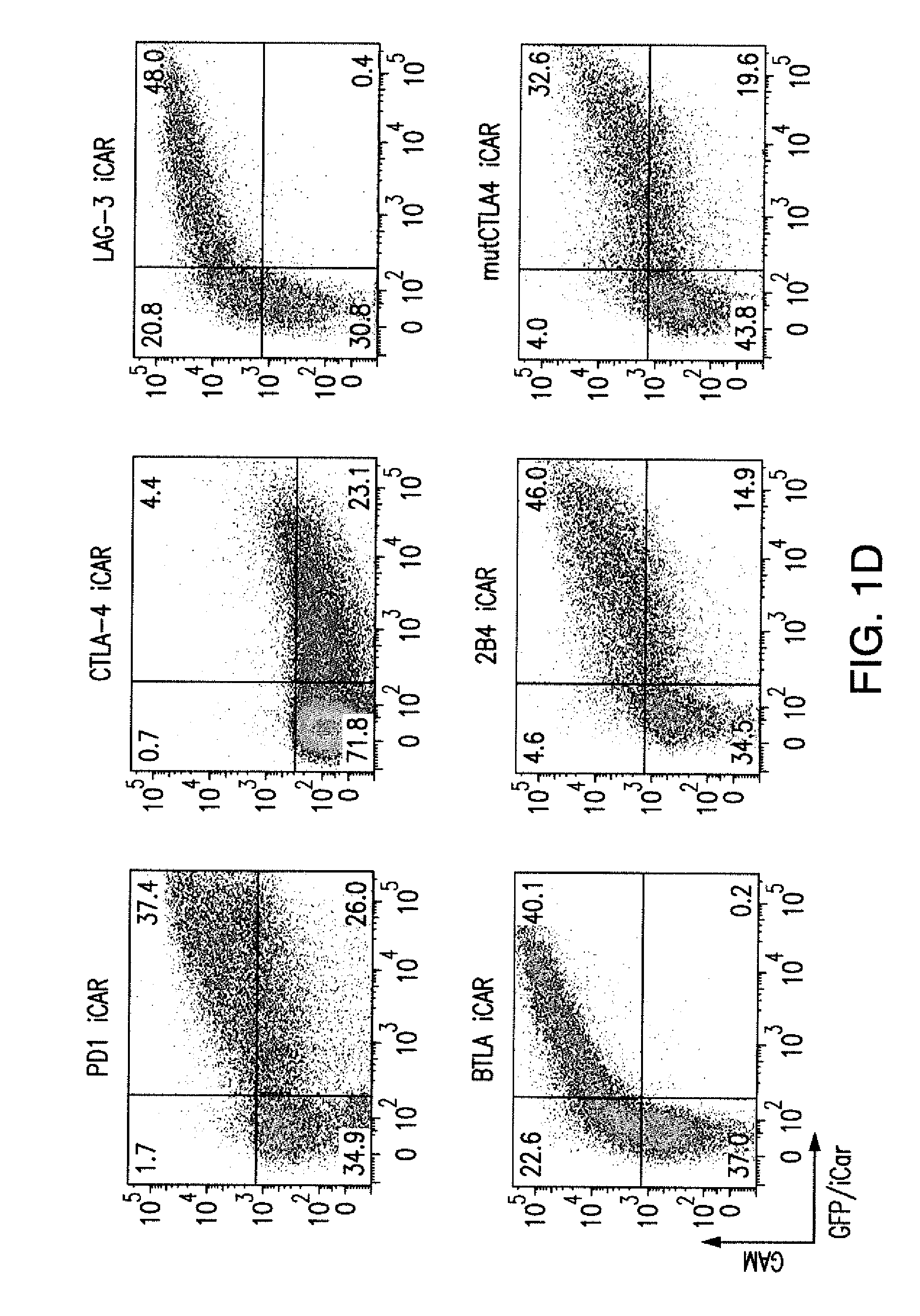
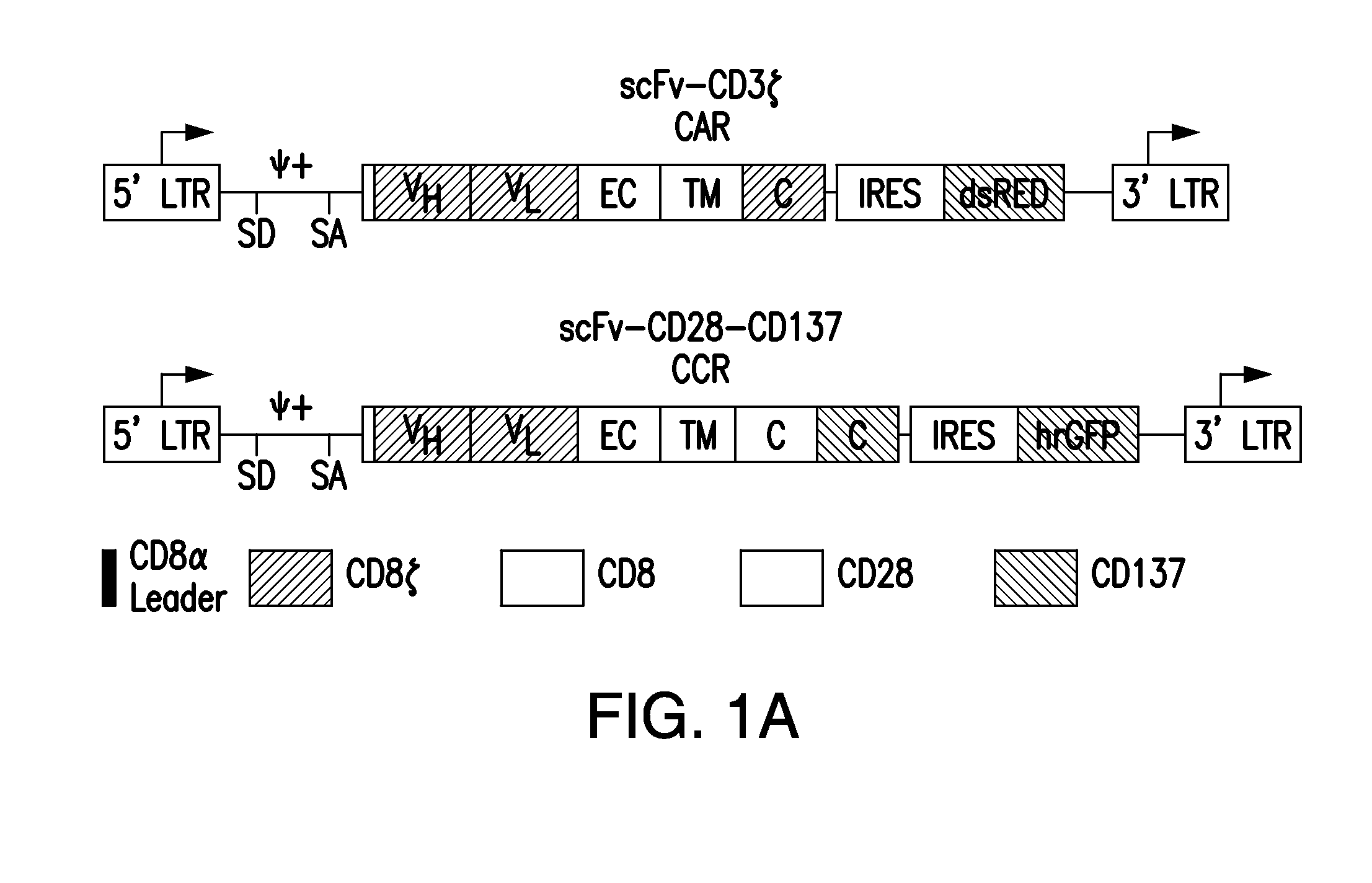
ActiveUS20150376296A1Decrease in immune activityReduces and eliminates immune activityAntibody mimetics/scaffoldsMuscular disorderRegulatory T cellChimeric antigen receptor
The present invention provides immunoresponsive cells, including T cells, cytotoxic T cells, regulatory T cells, and Natural Killer (NK) cells, expressing an antigen recognizing receptor and an inhibitory chimeric antigen receptor (iCAR). Methods of using the immunoresponsive cell include those for the treatment of neoplasia and other pathologies where an increase in an antigen-specific immune response is desired.
Owner:MEMORIAL SLOAN KETTERING CANCER CENT
Methods for converting or inducing protective immunity
InactiveUS20090214533A1Reduced activityReduce functionAntibody ingredientsImmunoglobulinsRegulatory T cellImmune complex deposition
The invention is based in part on the finding that suppressing regulatory T cell function is needed in order to convert passive immunity into active antigen-specific immunity. Generally, the methods of the invention comprise at least the combination of: (1) increasing the amount of immune complexes in the subject, wherein the immune complex comprises a target antigen and a immunoglobulin molecule comprising (i) a variable region specific to the target antigen and (ii) a Fc receptor binding region; and (2) inhibiting regulatory T cell function or decreasing / depleting the regulatory T cell population in the subject.
Owner:THE TRUSTEES OF COLUMBIA UNIV IN THE CITY OF NEW YORK
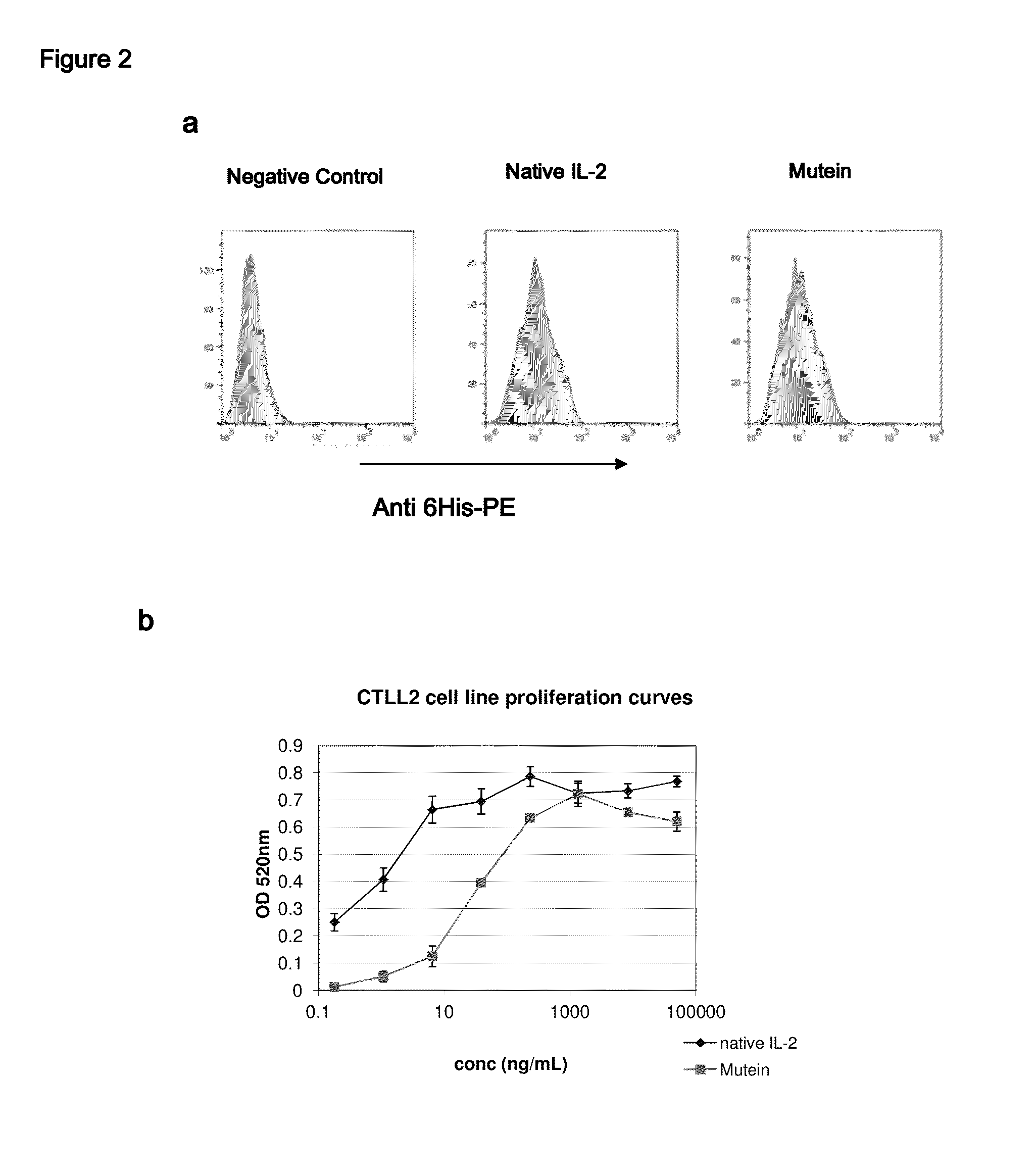
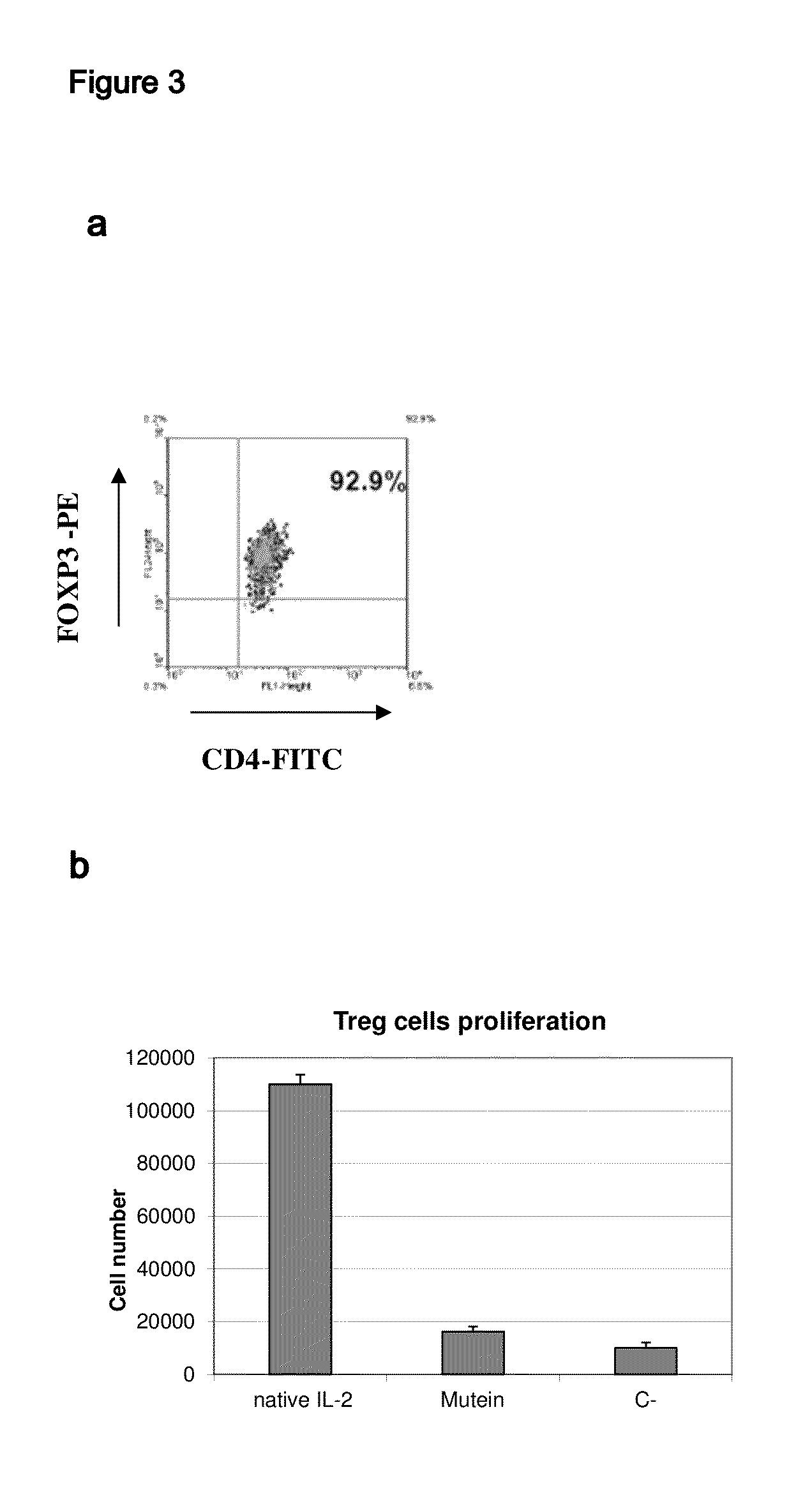
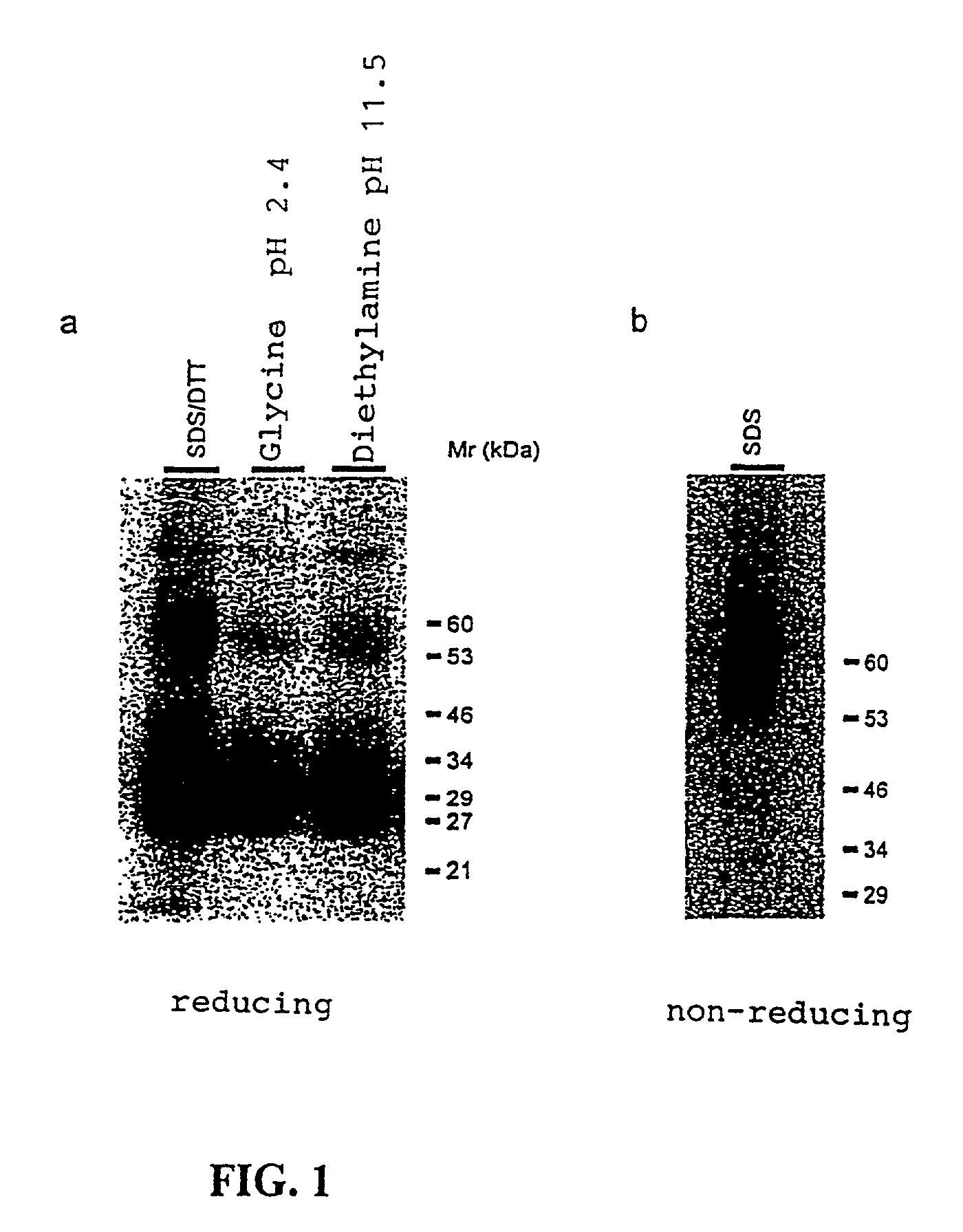
IL-2 derivative polypeptides
ActiveUS9206243B2Good treatment effectLow ability to stimulateAntibacterial agentsPeptide/protein ingredientsRegulatory T cellInfectious Disorder
The present invention relates to polypeptides which share primary sequence with human IL-2, except for several amino acids that have been mutated. The mutations introduced substantially reduce the ability of these polypeptides to stimulate in vitro and in vivo regulatory T cells (T CD4+CD25+FoxP3+) and make them more effective in the therapy of murine transplantable tumors. Also includes therapeutic uses of these mutated variants, used alone or in combination with vaccines for the therapy of diseases such as cancer or infections where the activity of regulatory T cells (Tregs) is relevant. In another aspect the present invention relates to pharmaceutical compositions comprising as active principle the polypeptides disclosed. Finally, the present invention relates to the therapeutic use of the polypeptides and pharmaceutical compositions disclosed due to their modulating effect of the immune system on diseases like cancer and chronic infectious diseases.
Owner:CENT DE INMUNOLOGIA MOLECULAR CENT DE INMUNOLO
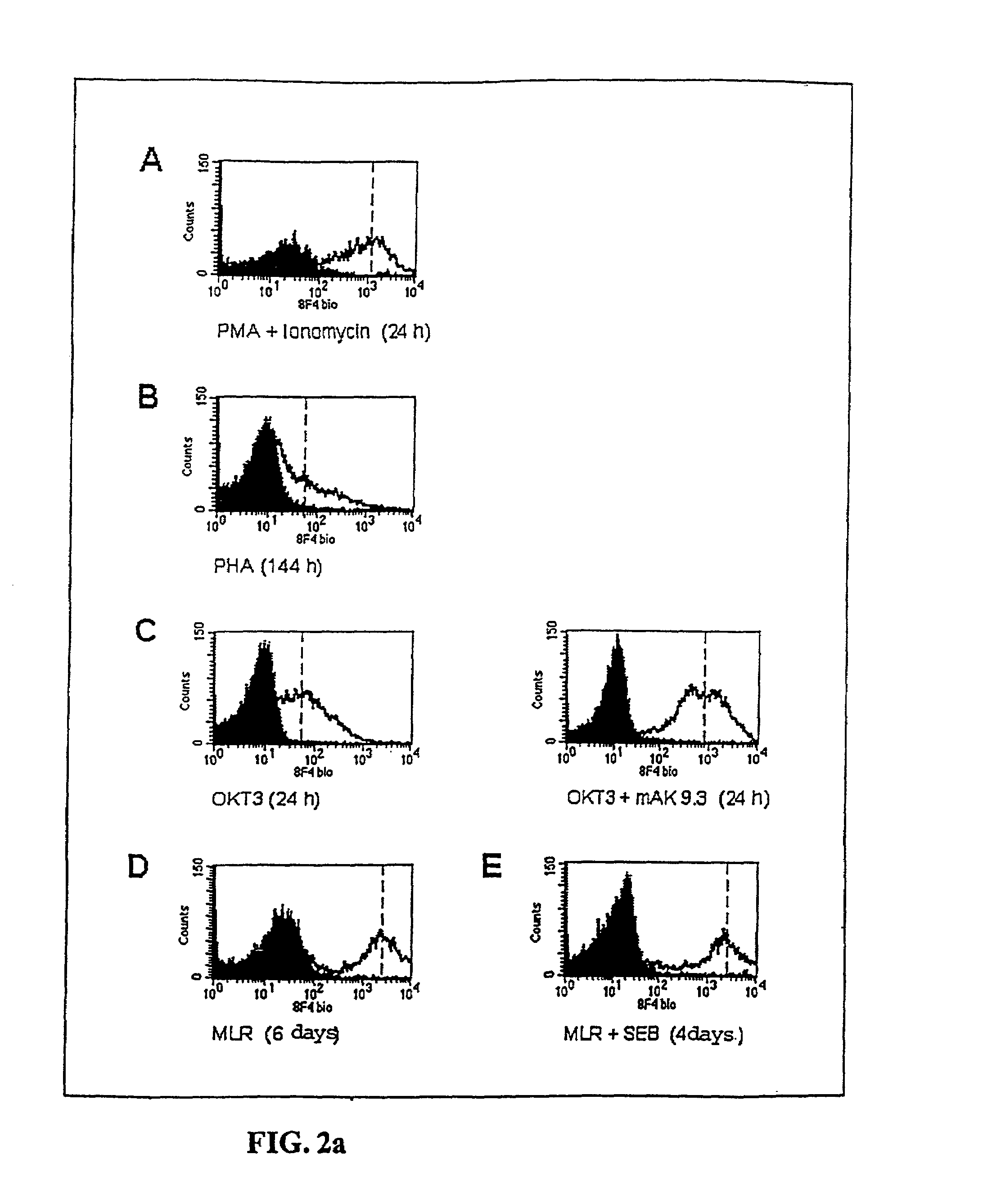
Methods for treatment of asthmatic disorders using a monoclonal antibody to 8F4 polypeptide
The present invention relates to methods and compositions for the prevention and treatment and prevention of immune system disorders, including cancer, AIDS, asthmatic disorders, autoimmune diseases, organ transplant rejection and chronic viral diseases such as HCV or HBV infections. The therapeutic methods of the invention comprise administering molecules that modulate the activity of 8F4, thereby modulating costimulation of T cells. The present invention further provides monoclonal antibodies against the 8F4 molecule and hybridoma cells which produce said monoclonal antibodies. Pharmaceutical compositions comprising molecules that modulate the activity of 8F4 are also provided.
Owner:BUNDESREPUBLIK DEUTSCHLAND
T cell regulation
InactiveUS20060240024A1Increases magnitudeSimple methodAntibacterial agentsNervous disorderRegulatory T cellAutoimmune responses
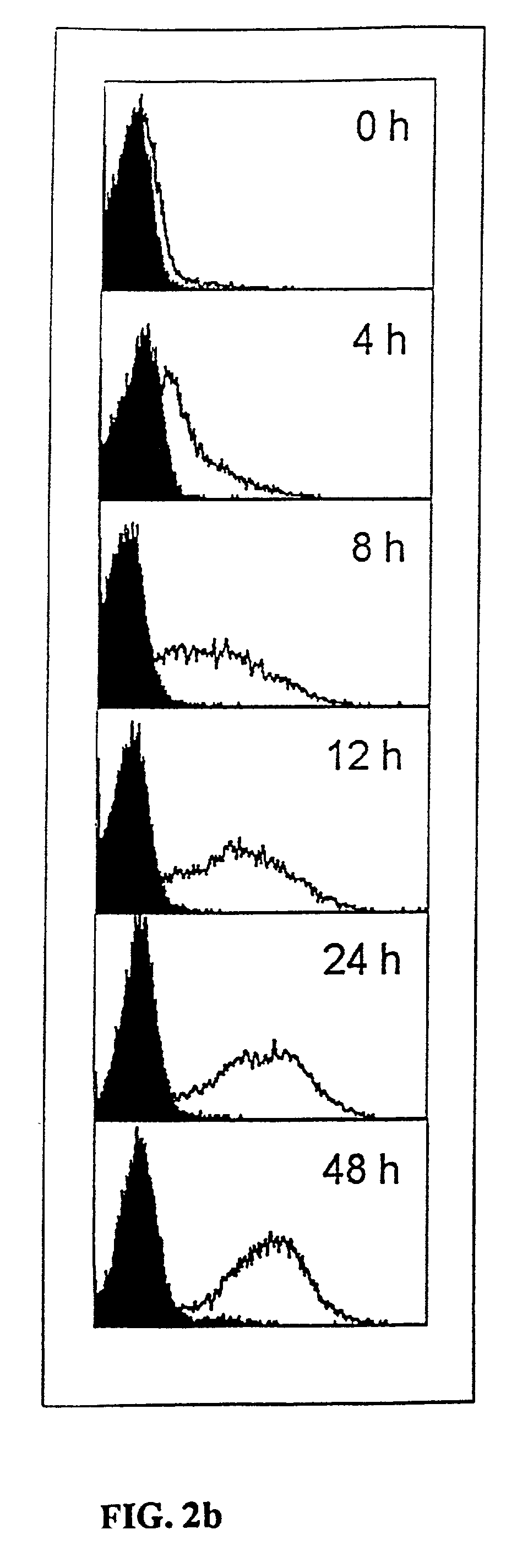
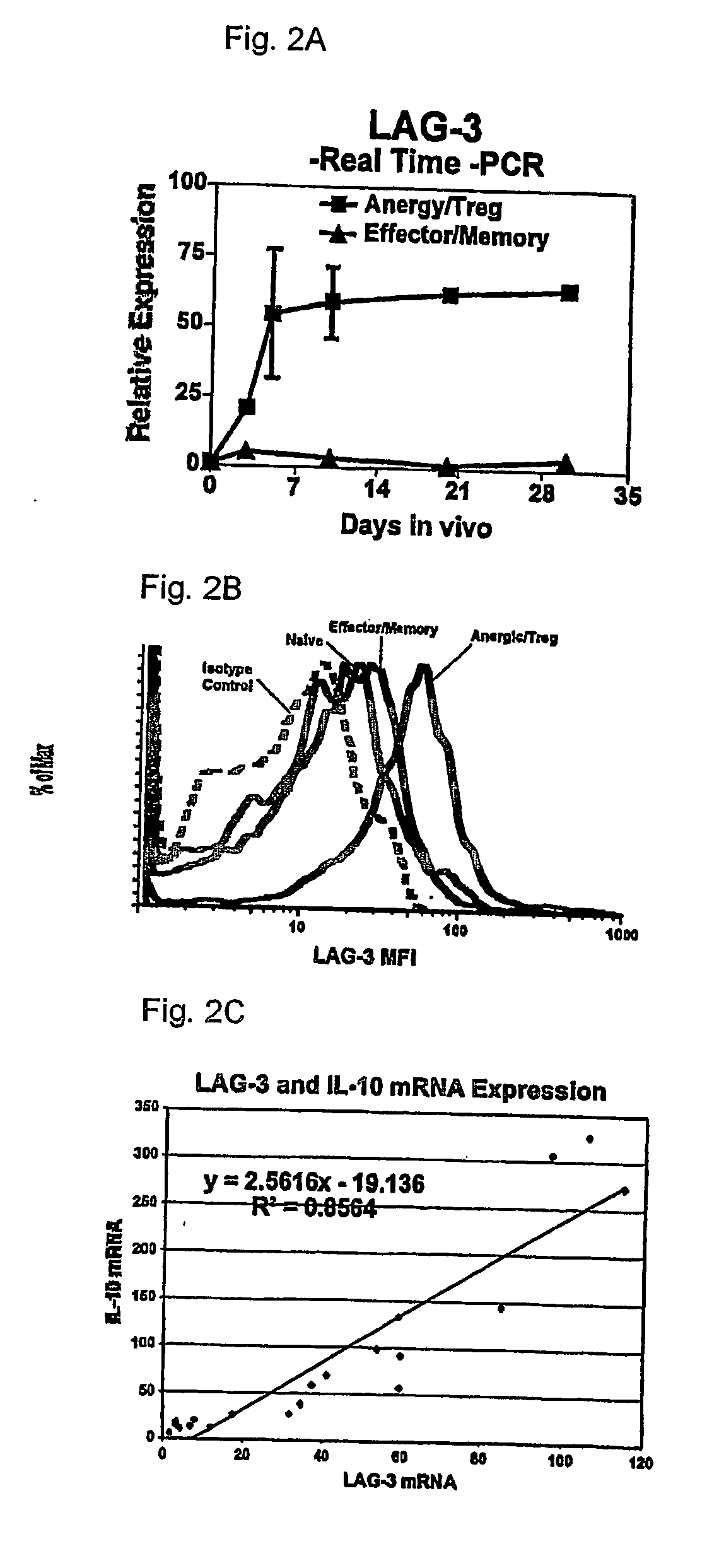
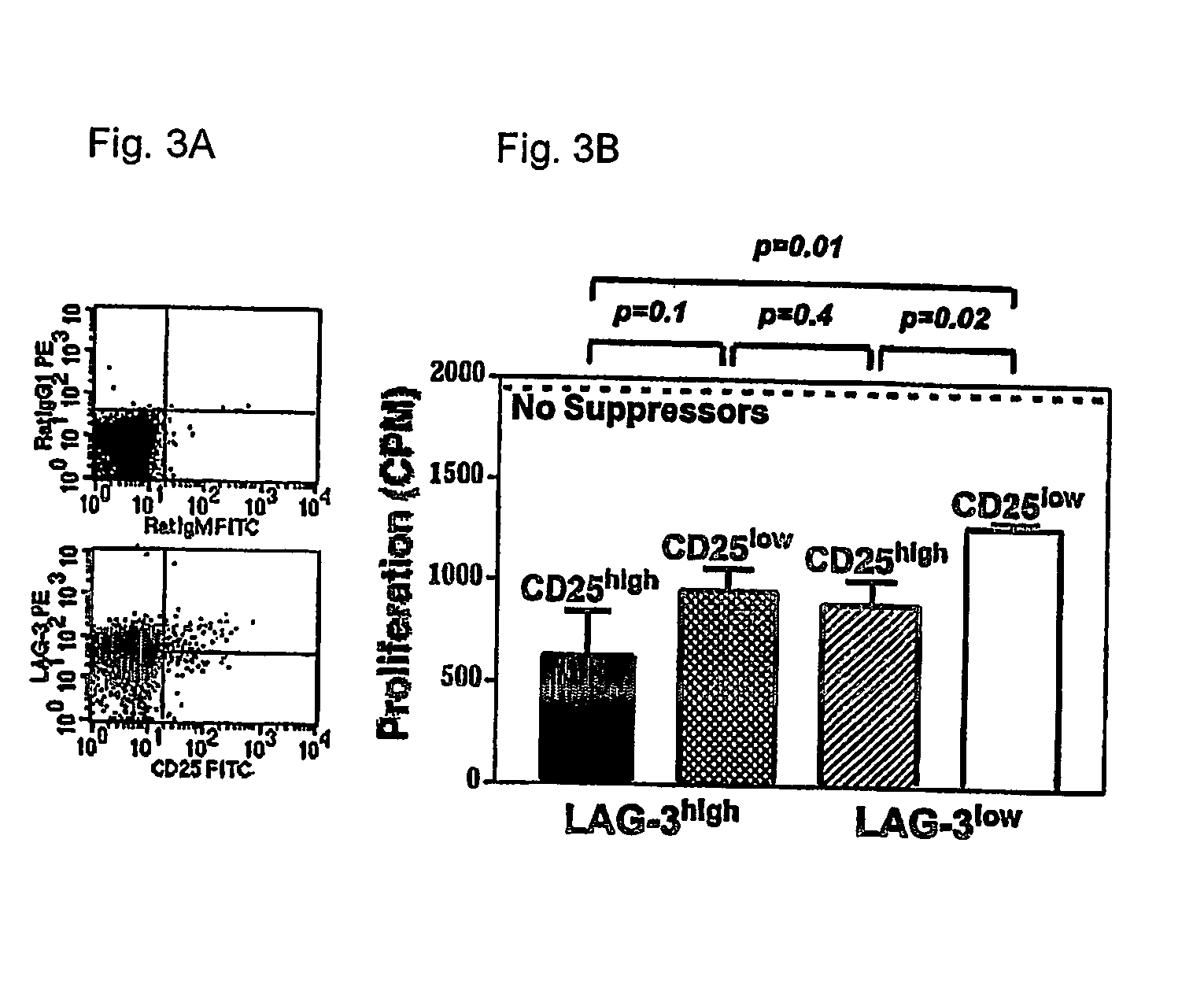
Regulatory T cells (Treg) limit autoimmunity but can also attenuate the magnitude of anti-pathogen and anti-tumor immunity. Understanding the mechanism of Treg function and therapeutic manipulation of Treg in vivo requires identification of Treg selective receptors. A comparative analysis of gene expression arrays from antigen specific CD4+ T cells differentiating to either an effector / memory or a regulatory phenotype revealed Treg selective expression of LAG-3 (CD223), a CD4-related molecule that binds MHC class II. LAG-3 expression on CD4+ T cells correlates with the cells' in vitro suppressor activity, and ectopic expression of LAG-3 on CD4 T cells confers suppressor activity on the T cells. Antibodies to LAG-3 inhibit suppression both in vitro and in vivo. LAG-3 marks regulatory T cell populations and contributes to their suppressor activity.
Owner:ST JUDE CHILDRENS RES HOSPITAL INC +1
Regulatory t cell mediator proteins and uses thereof
ActiveUS20110027278A1Eliminate the effects ofPeptide/protein ingredientsAntibody mimetics/scaffoldsRegulatory T cellAllergy
The present invention relates to novel regulatory T cell proteins. One protein, designated PD-L3, resembles members of the PD-L1 family, and co-stimulates αCD3 proliferation of T cells in vitro. A second, TNF-like, protein has also been identified as being upregulated upon αCD3 / αGITR stimulation. This protein has been designated Treg-sTNF. Proteins, antibodies, activated T cells and methods for using the same are disclosed.In particular methods of using these proteins and compounds, preferably antibodies, which bind or modulate (agonize or antagonize) the activity of these proteins, as immune modulators and for the treatment of cancer, autoimmune disease, allergy, infection and inflammatory conditions, e.g. multiple sclerosis is disclosed
Owner:TRUSTEES OF DARTMOUTH COLLEGE THE
Methods of generating antigen-specific CD4+CD25+ regulatory T cells, compositions and methods of use
ActiveUS20060115899A1Reduce riskReduce severityArtificial cell constructsDead animal preservationRegulatory T cellSelect agent
The present invention provides methods for generating mammalian T cell populations comprising antigen-specific CD4+CD25+ regulatory T cells from freshly isolated CD4+CD25− T cells. The method comprises selecting CD4+CD25− T cells from a sample obtained from a mammalian subject; determining the MHC Class II type of the subject; inducing the generation of antigen-specific regulatory T cells by contacting the isolated CD4+CD25− T cells in a culture vessel with an induction agent for a time period sufficient to generate antigen-specific CD4+CD25+ regulatory T cells; and selecting the CD4+CD25+ antigen-specific regulatory T cells by sorting the cells in the induction culture with a selection agent comprising at least one artificial multimeric MHC Class II / peptide complex that corresponds to the MHC Class II type of the subject.
Owner:BENAROYA RES INST AT VIRGINIA MASON
Molecules preferentially associated with effector T cells or regulatory T cells and methods of their use
The present invention is based, at least in part, on the finding that certain molecules are preferentially associated with effector T cells or regulatory T cells. Accordingly, immune responses by one or the other subset of cells can be preferentially modulated. The invention pertains, e.g., to methods of modulating (e.g., up- or down-modulating), the balance between the activation of regulatory T cells and effector T cells leading to modulation of immune responses and to compositions useful in modulating those responses. The invention also pertains to methods useful in diagnosing, treating, or preventing conditions that would benefit from modulating effector T cell function relative to regulatory T cell function or from modulating regulatory T cell function relative to effector T cell function in a subject. The subject methods and compositions are especially useful in the diagnosis, treatment or prevention of conditions characterized by a too-vigorous effector T cell response to antigens associated with the condition, in the diagnosis, treatment or prevention of conditions characterized by a weak effector T cell response, in the diagnosis, treatment or prevention of conditions characterized by a too-vigorous regulatory T cell response, or in the diagnosis, treatment, or prevention of conditions characterized by a weak regulatory T cell response.
Owner:TOLERX INC
Regulatory T Cell Mediator Proteins and Uses Thereof
The present invention relates to novel regulatory T cell proteins. One protein, designated PD-L3, resembles members of the PD-L1 family, and co-stimulates αCD3 proliferation of T cells in vitro. A second, TNF-like, protein has also been identified as being upregulated upon αCD3 / αGITR stimulation. This protein has been designated Treg-sTNE Proteins, antibodies, activated T cells and methods for using the same are disclosed.
Owner:TRUSTEES OF DARTMOUTH COLLEGE THE
Compositions and methods for immunotherapy
ActiveUS20150342993A1Decrease number of tumorShrink tumorAntibody mimetics/scaffoldsImmunoglobulins against cell receptors/antigens/surface-determinantsRegulatory T cellT cell
The present invention provides immunoresponsive cells, including T cells, cytotoxic T cells, regulatory T cells, and Natural Killer (NK) cells, expressing at least one of an antigen recognizing receptor and one of a chimeric costimulatory receptor. Methods of using the immunoresponsive cell include those for the treatment of neoplasia and other pathologies where an increase in an antigen-specific immune response is desired.
Owner:MEMORIAL SLOAN KETTERING CANCER CENT
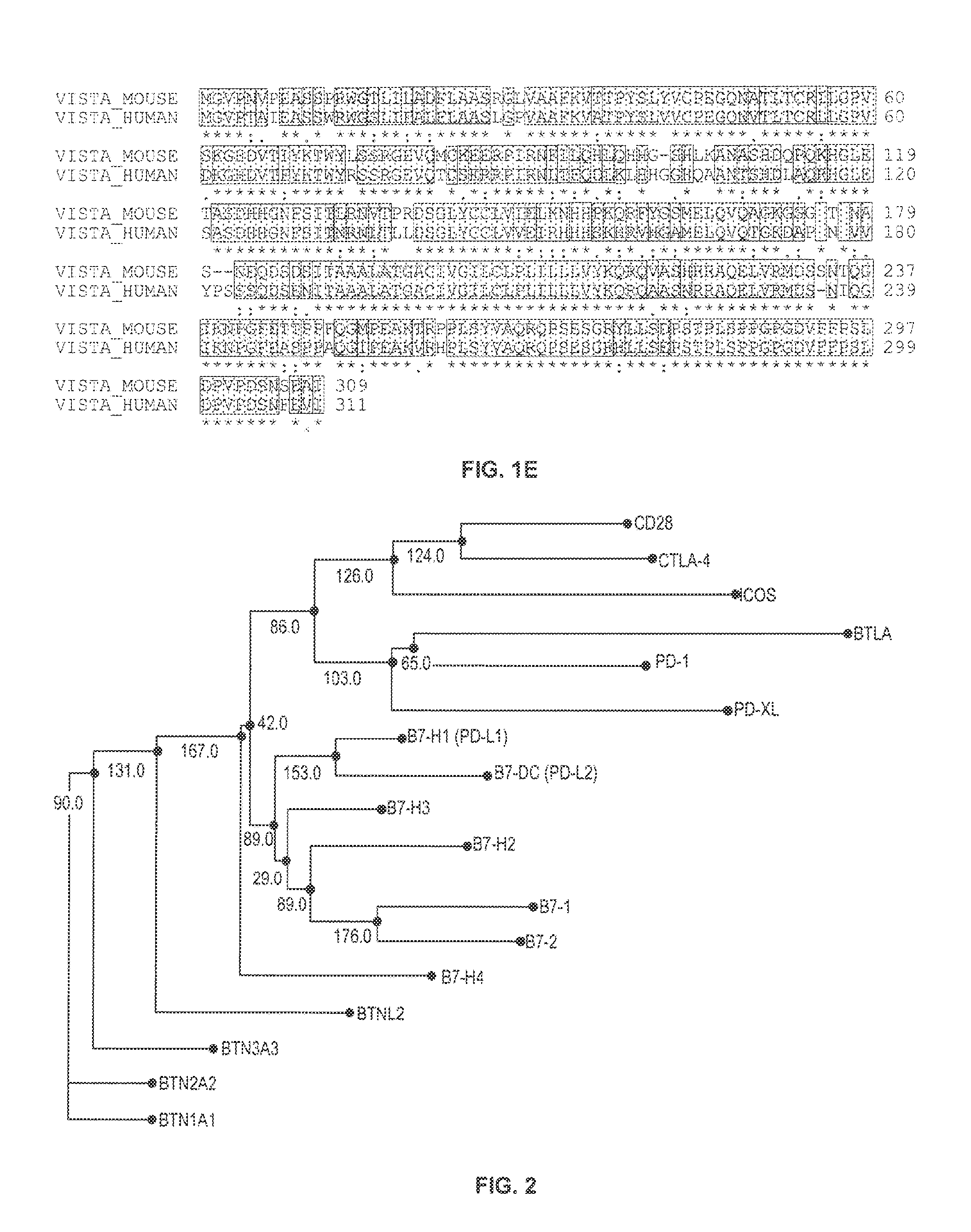
VISTA-Ig for Treatment of Autoimmune, Allergic and Inflammatory Disorders
ActiveUS20140056892A1Relieve T cell exhaustionImprove immunityImmunoglobulin superfamilyPeptide/protein ingredientsImmunologic disordersRegulatory T cell
The present invention relates to a fusion proteins comprising regulatory T cell protein, VISTA (V-domain Immunoglobulin Suppressor of T cell Activation (PD-L3) and an immunoglobulin protein (Ig). The invention also provides the use of VISTA polypeptides, multimeric VISTA polypeptides, VISTA-conjugates (e.g., VISTA-Ig), and VISTA antagonists for the treatment of autoimmune disease, allergy, and inflammatory conditions.
Owner:TRUSTEES OF DARTMOUTH COLLEGE THE +1
Agent for treating disease
ActiveUS20110059082A1Easy to manageIncrease doseNervous disorderAntipyreticRegulatory T cellAutoimmune condition
The present invention provides a pharmaceutical composition for treating an autoimmune disease comprising a pharmaceutically acceptable carrier and an agent capable of activating CD4+CD25+ regulatory T cells, wherein the composition is to be administered to a subject in a dose of the agent from 10 mg to 200 mg.
Owner:OGEN GMBH
Agent for treating disease
ActiveUS20110059083A1Easy to manageEffective treatmentNervous disorderAntipyreticDiseaseRegulatory T cell
The present invention provides pharmaceutical composition for treating an autoimmune disease comprising a pharmaceutically acceptable carrier and an agent capable of activating CD4+CD25+ regulatory T cells, wherein the composition is to be administered to a subject at most every 3 days.
Owner:OGEN GMBH
Use of cell adhesion inhibitor for the mobilization of antigen presenting cells and immune cells in a cell mixture (AIM) from the peripheral blood and methods of use
Disclosed is a method to recover an antigen presenting cells (APCs) and immune cells rich mixture (AIM) from peripheral blood mononuclear cells (PBMC) mobilized with one or more cell adhesion inhibitors for the preparation of an AIM vaccine or an AIM adoptive immunotherapy preparation. In addition, AIM mobilization can be enhanced by priming, simultaneously or in sequence, one or more of a combination of different chemical compounds, cytokines, hormones, growth factors, etc. The interaction of chemokines and chemokine receptors enable tumor cells attachment or in close proximity to antigen presenting cells and immune cells which possess similar receptors in a micro niche environment. Severing the chemokine / chemokine receptor linkage by a cell adhesion inhibitor will release these specifically primed cell mixtures into the peripheral blood. The collection of these cells from the peripheral blood has never been described and is the basis of this invention. AIM cells can either be used alone or better still, be induced into more target specific preparations with additions, modifications and incubation, pre or post cell adhesion inhibitors mobilization, with vaccines, different target specific antigens, peptides, chemotherapeutic agents, oncolytic viral therapeutic agents, cytokines, co-stimulatory molecules, anti-regulatory T cell therapeutic agents, anti-CTLA4, anti-PD1 molecules and other methodologies of immunological enhancement known to the art. The AIM vaccine or AIM adoptive immunotherapy preparation can then be used, but not limited to, the treatment of cancer and other diseases.
Owner:YEUNG ALEX WAH HIN +1