Anti-inflammatory polypeptide BMP14, and preparation method and application thereof
A technology of BMP14 and solid-phase synthesis, which is applied to the preparation method of peptides, anti-inflammatory agents, chemical instruments and methods, etc., can solve the problems of difficult clinical application, poor stability in vivo, hemolytic cytotoxicity, etc., and achieve low cost, The effect of good stability and high quality controllability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0035] (1) According to the designed amino acid sequence:
[0036] Ile-Asn-Leu-Arg-Val-Ile-Ala-Cys-Leu-Val-Arg-Lys-Ile-Leu, the crude polypeptide was synthesized by solid phase synthesis;
[0037] (2) desalting and purifying the crude polypeptide by HPLC reverse-phase column chromatography, and identifying its purity until the purity of the polypeptide is not less than 95%;
[0038] HPLC purification and identification method:
[0039] Dissolve 0.1 mg of the sample to be tested in 1 mL of ultrapure water containing 0.1% trifluoroacetic acid. If there are undissolved impurities, filter them with a 0.45 μm filter membrane. Mobile phase A is 0.1% trifluoroacetic acid-water, mobile phase B It is 0.1% trifluoroacetic acid-acetonitrile. After the baseline is stable, start to load the sample. The sample volume is 50 μL; the chromatographic column is a silica gel alkyl bonded phase C18 column (4.6mm×300mm, colloidal particle size 5μm, pore size 100A) , using a binary mobile phase gr...
experiment example 1
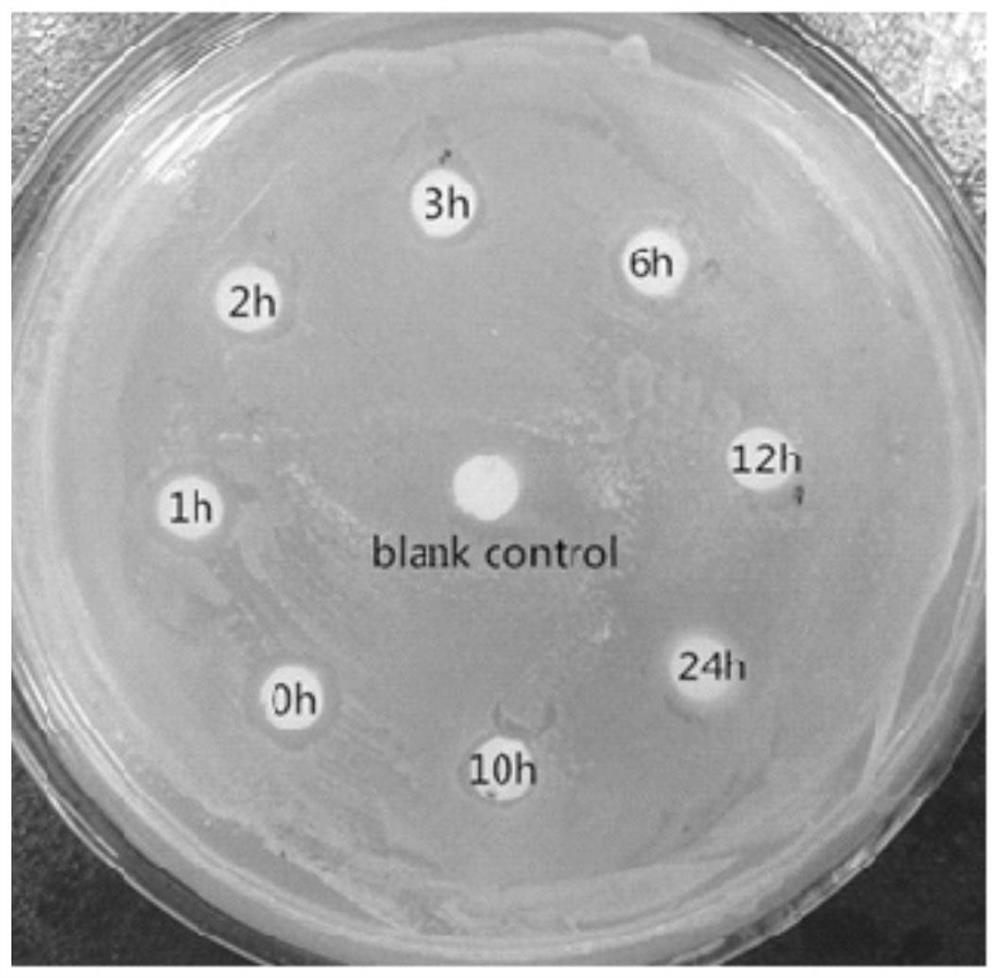
[0044] Antibacterial activity detection
[0045] The minimum inhibitory concentration is the lowest drug concentration that can inhibit the growth and reproduction of bacteria. The experiment adopts the method of two-fold gradient dilution, and the specific experimental operation is as follows: prepare fresh bacterial liquid, use an ultraviolet spectrophotometer to detect the OD600 of the bacterial liquid, according to 1OD600=1×109CFU / ml, use fresh LB liquid medium to dilute the above bacterial liquid The concentration was diluted and adjusted to 2×105 CFU / ml. After that, add 100 μl of normal saline to the sterile 96-well plate in advance, add the sample to be tested in the first well, perform two-fold gradient dilution of the sample to be tested in turn, and then add 100 μl of 2×105 CFU / ml to each well. Use a pipette gun to blow and mix the bacterial solution, and then place it in a constant temperature incubator at 37°C for overnight culture. Finally, use a microplate reade...
experiment example 2
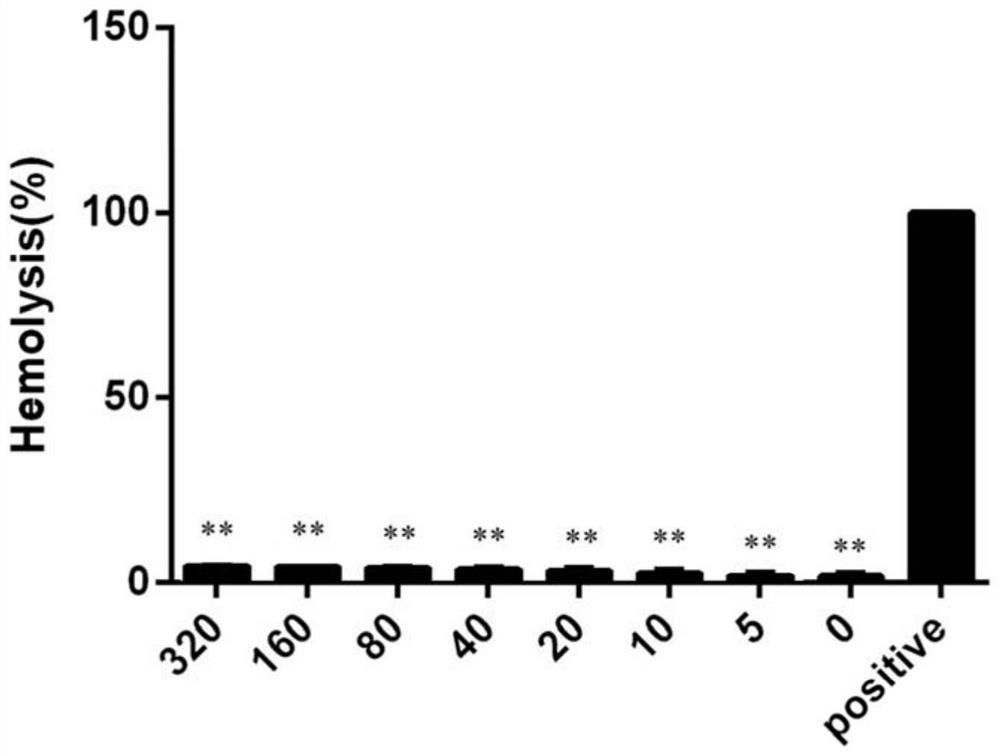
[0051] Hemolytic Activity and Cytotoxicity Assays
[0052] Use physiological saline to adjust the density dilution of the washed red blood cells to 107-108 cells / ml. At the same time, prepare the samples to be tested into different gradient concentrations, place the two at a constant temperature of 37 ° C for 30 min, and then centrifuge at 1000 rpm for 5 min. A microplate reader was used to detect the absorbance of the supernatant at 540 nm. In this experiment, physiological saline was used as a negative control, and the same volume of TritonX-100 (10%) was used as a positive control, and the hemolytic activity was directly proportional to the light absorption value at 540 nm.
[0053] Hemolytic activity as attached figure 1 As shown, the hemolytic activity of BMP14 was low.
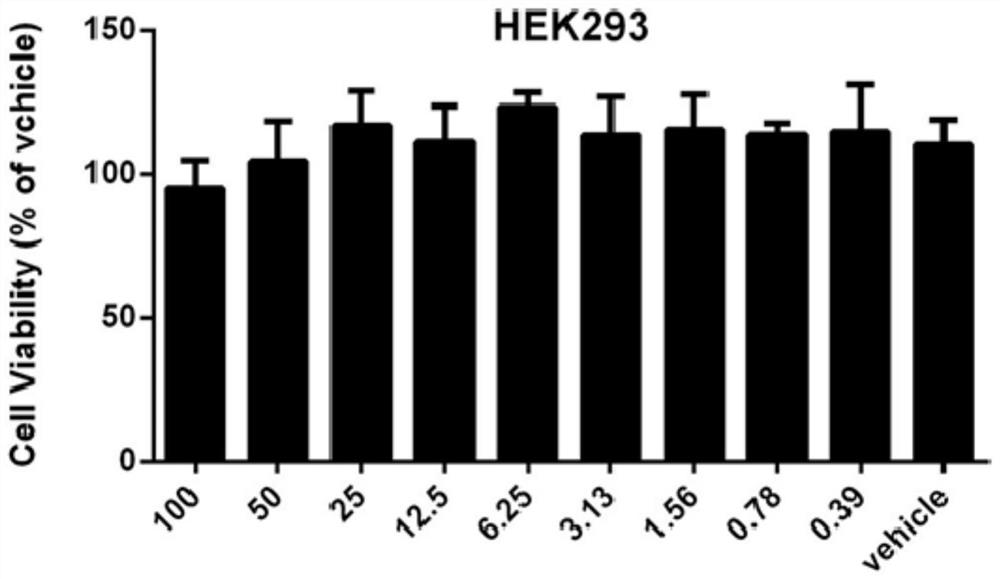
[0054] Cytotoxicity assays were performed using the human embryonic kidney cell line HEK293T. When the cells grow well and the density reaches 80% of the bottom of the bottle, discard the medium, wash...
PUM
| Property | Measurement | Unit |
|---|---|---|
| control rate | aaaaa | aaaaa |
| control rate | aaaaa | aaaaa |
| control rate | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap