Gadolinium-functionalized copper sulfide nanoparticle and preparation method and application thereof
A nanoparticle and copper sulfide technology, applied in the field of nanomedicine, can solve the problems of restricting large-scale production, harsh reaction conditions, and fast clearance rate in the body, and achieve good clinical medical transformation application prospects, improve image contrast, and good photothermal performance effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0033] A preparation method of gadolinium functionalized copper sulfide nanoparticles, comprising the following steps:
[0034] (1) dissolving disodium edetate in an acidic solution under magnetic stirring at 55° C. to obtain an aqueous solution of disodium edetate, the acidic solution being a hydrochloric acid solution with a pH value of 5, and the The molar concentration of disodium edetate aqueous solution is 11mmol / L;
[0035] (2) Add gadolinium chloride hexahydrate to the disodium edetate aqueous solution of step (1) under magnetic stirring, carry out chelation reaction, obtain Gd 3+ In the chelate solution, the molar ratio of gadolinium chloride hexahydrate to disodium edetate is 0.18-0.45:1, and the time for the chelation reaction is preferably 30 minutes.
[0036] (3) Bovine serum albumin, polyethylene glycol and copper chloride dihydrate are dissolved in the Gd of step (2) under magnetic stirring 3+ Chelate solution, obtain mixed solution, bovine serum albumin, polyet...
Embodiment 1
[0044] (1) Weigh 32.8 mg of disodium edetate and add it to 8 ml of solvent, and fully stir and dissolve it under the condition of 55°C with a magnetic stirrer;
[0045] (2) Weigh 5.9 mg of gadolinium chloride hexahydrate and stir for 30 min under magnetic stirring at room temperature to obtain a chelated solution.
[0046] (3) Weigh 10 mg of bovine serum albumin, 10 mg of polyethylene glycol, and 23.9 mg of copper chloride dihydrate, add the above-mentioned chelated solution under magnetic stirring at room temperature, and stir for 5 minutes to obtain a light blue mixed solution.
[0047] (4) Add 2ml (168.1mg) sodium sulfide solution to the above mixed solution under normal temperature conditions in a magnetic stirrer, the volume ratio of sodium sulfide nonahydrate solution and mixed solution is 1:4, stir for 5 minutes, the color of the reaction solution gradually turns black Green, the nanoparticle nuclear reaction solution is obtained. Continue to stir the nanoparticle nucl...
Embodiment 2
[0049] (1) Weigh 32.8 mg of disodium edetate and add it into 8 ml of solvent, and fully stir and dissolve in a magnetic stirrer at 55°C.
[0050] (2) Weigh 10.4 mg of gadolinium chloride hexahydrate and stir for 30 minutes under magnetic stirring at room temperature to obtain a chelated solution.
[0051] (3) Weigh 10 mg of bovine serum albumin, 10 mg of polyethylene glycol, and 23.9 mg of copper chloride dihydrate, add the above-mentioned chelated solution under magnetic stirring at room temperature, and stir for 5 minutes to obtain a light blue mixed solution.
[0052] (4) Add 2ml (168.1mg) sodium sulfide solution to the above mixed solution under normal temperature conditions in a magnetic stirrer, the volume ratio of sodium sulfide nonahydrate solution and mixed solution is 1:4, stir for 5 minutes, the color of the reaction solution gradually turns black Green, the nanoparticle nuclear reaction solution is obtained. The nanoparticle nuclear reaction solution was stirred f...
PUM
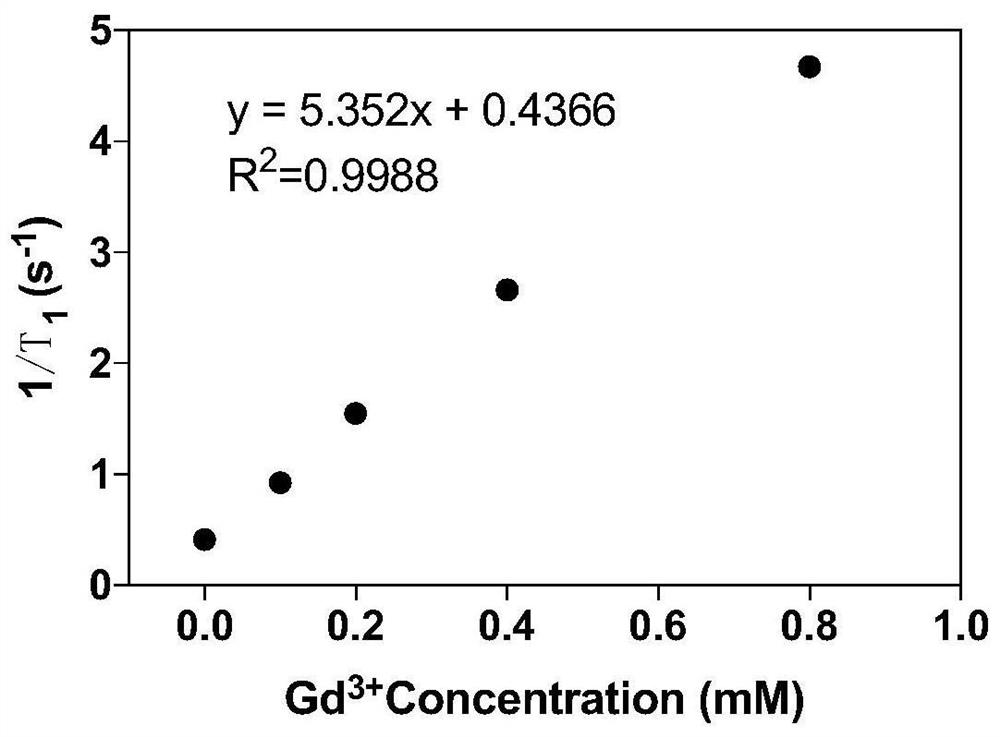
| Property | Measurement | Unit |
|---|---|---|
| Relaxation rate | aaaaa | aaaaa |
| Particle size | aaaaa | aaaaa |
| Relaxation rate | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com