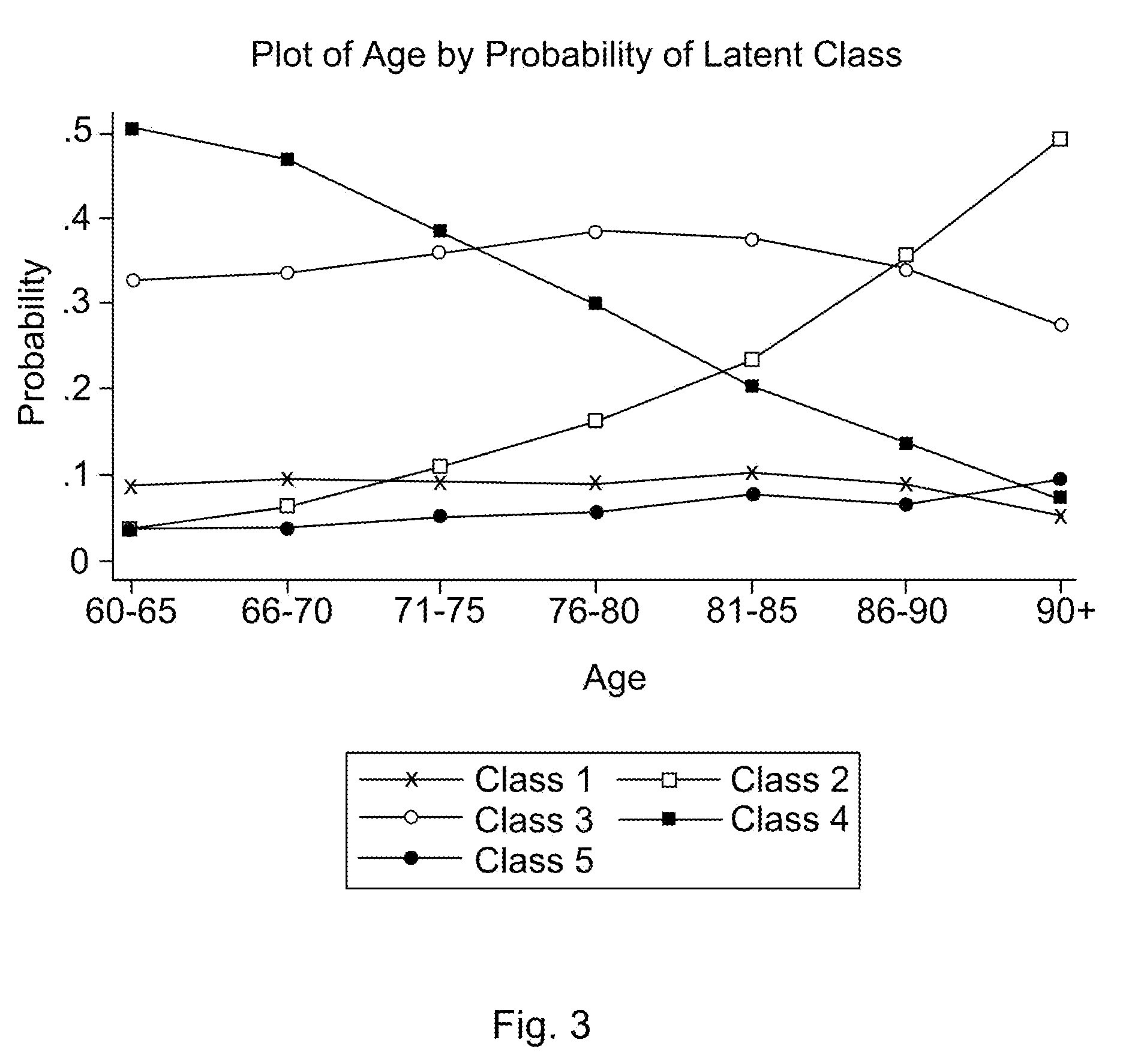Patents
Literature
173 results about "Eye disorder" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Impairment of health or a condition of abnormal functioning of the organ of sight.
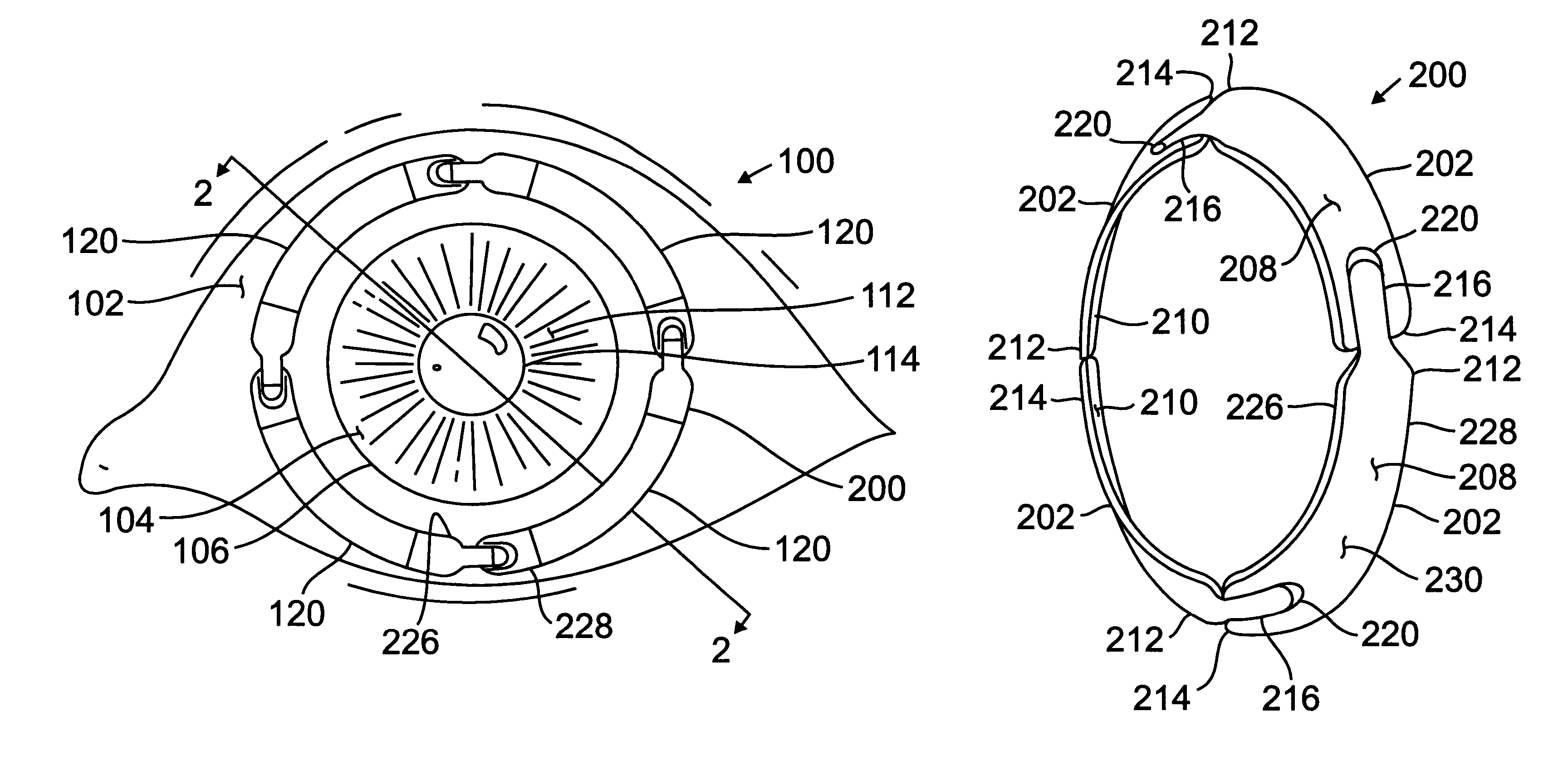
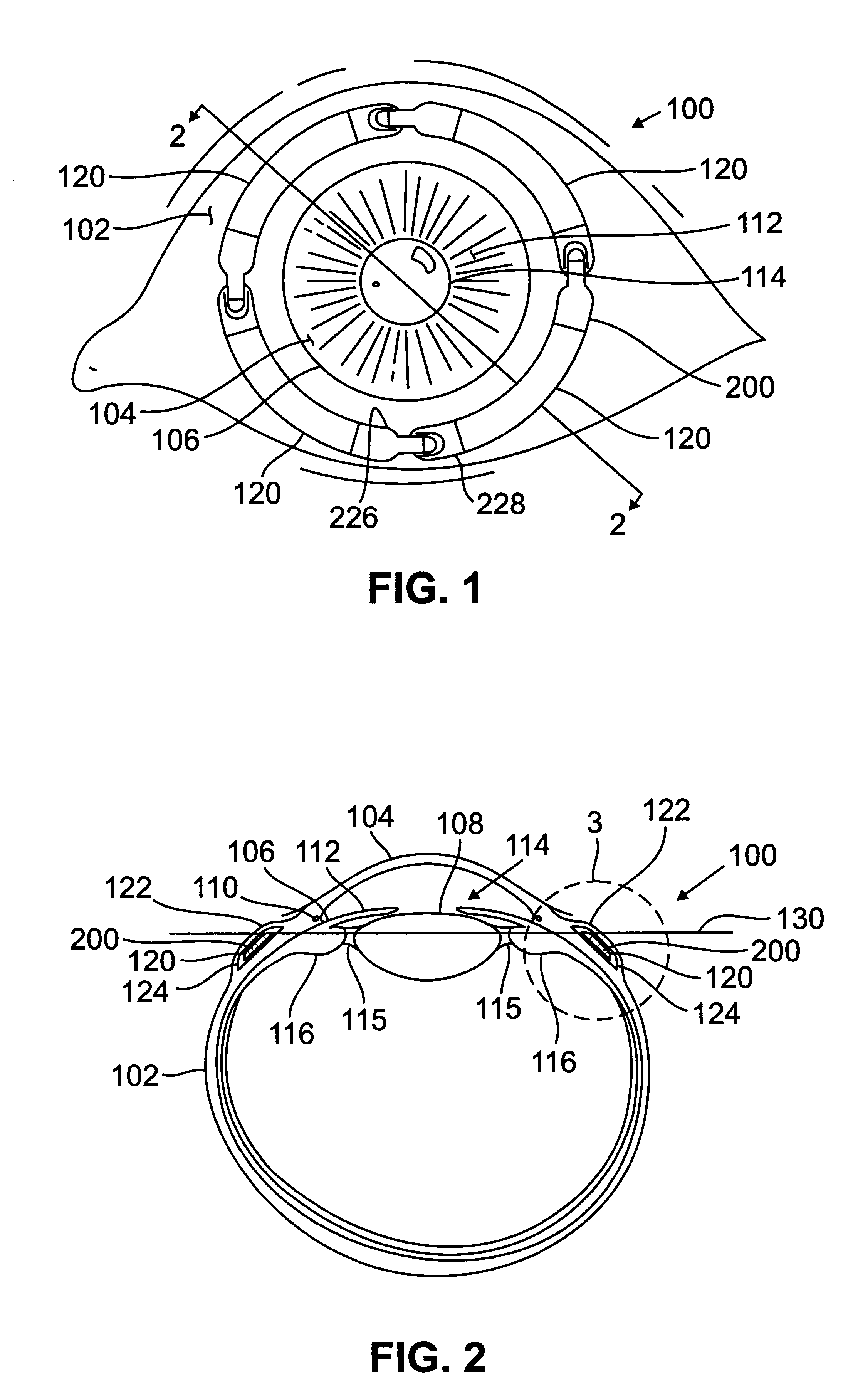
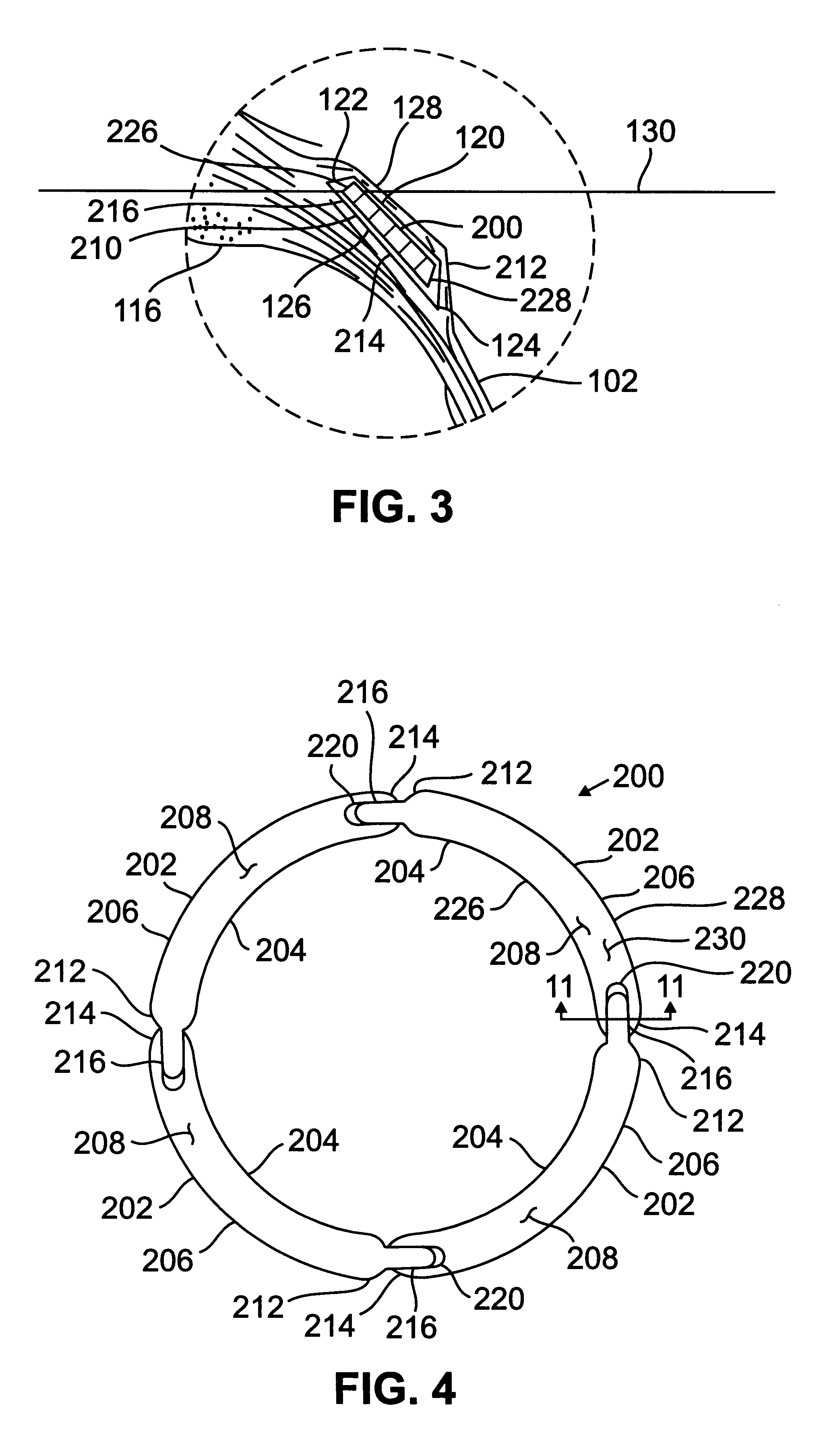
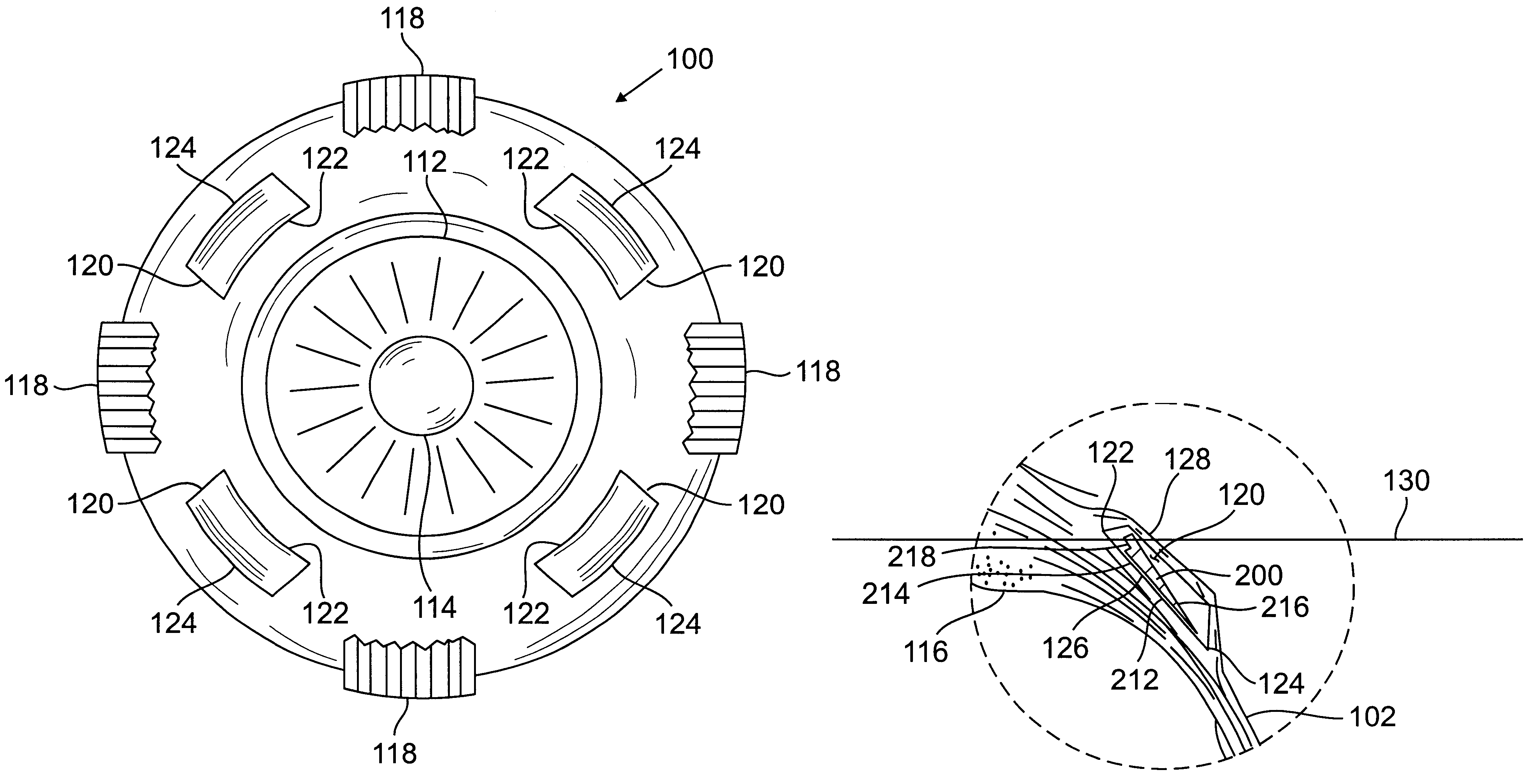
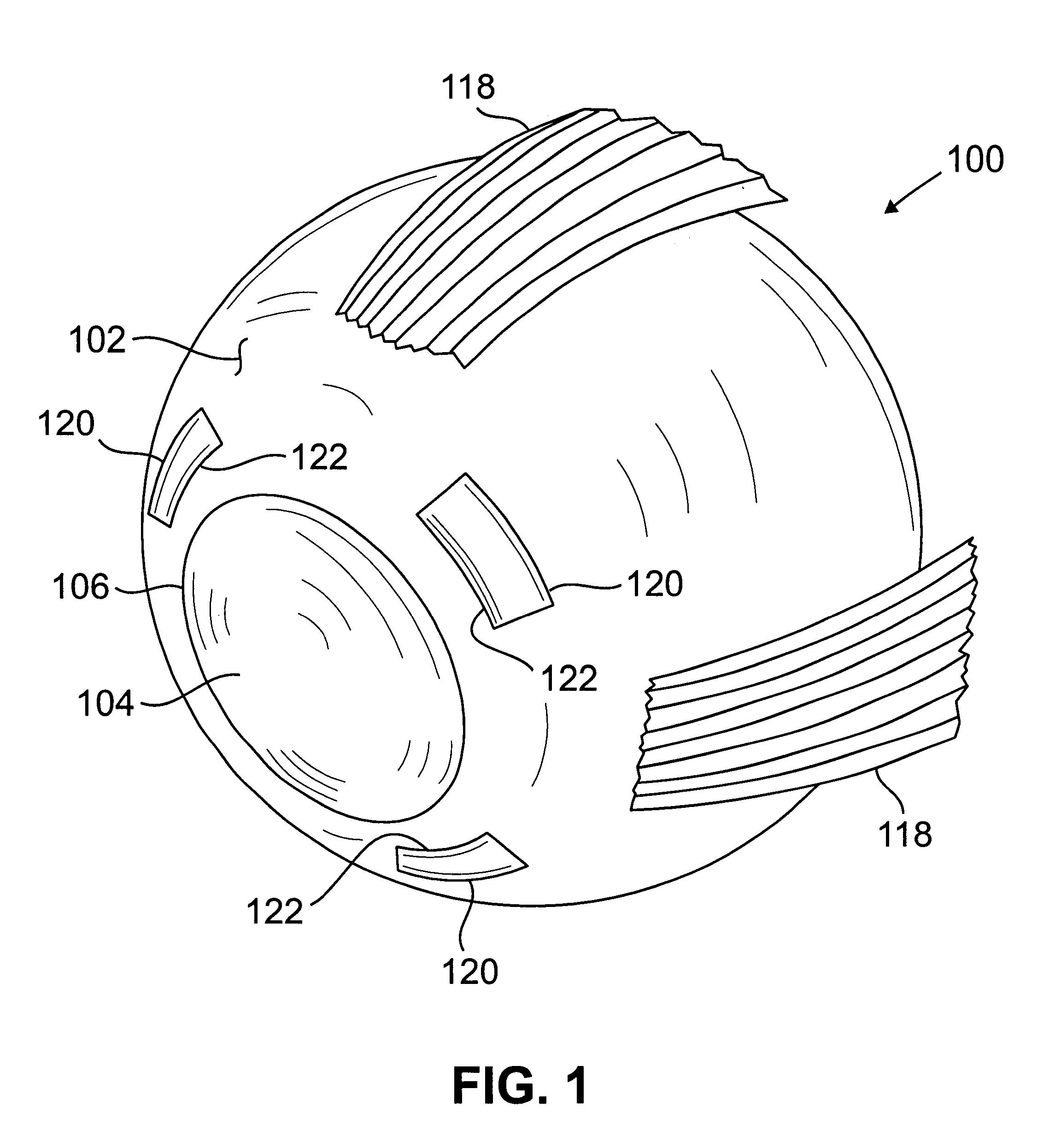
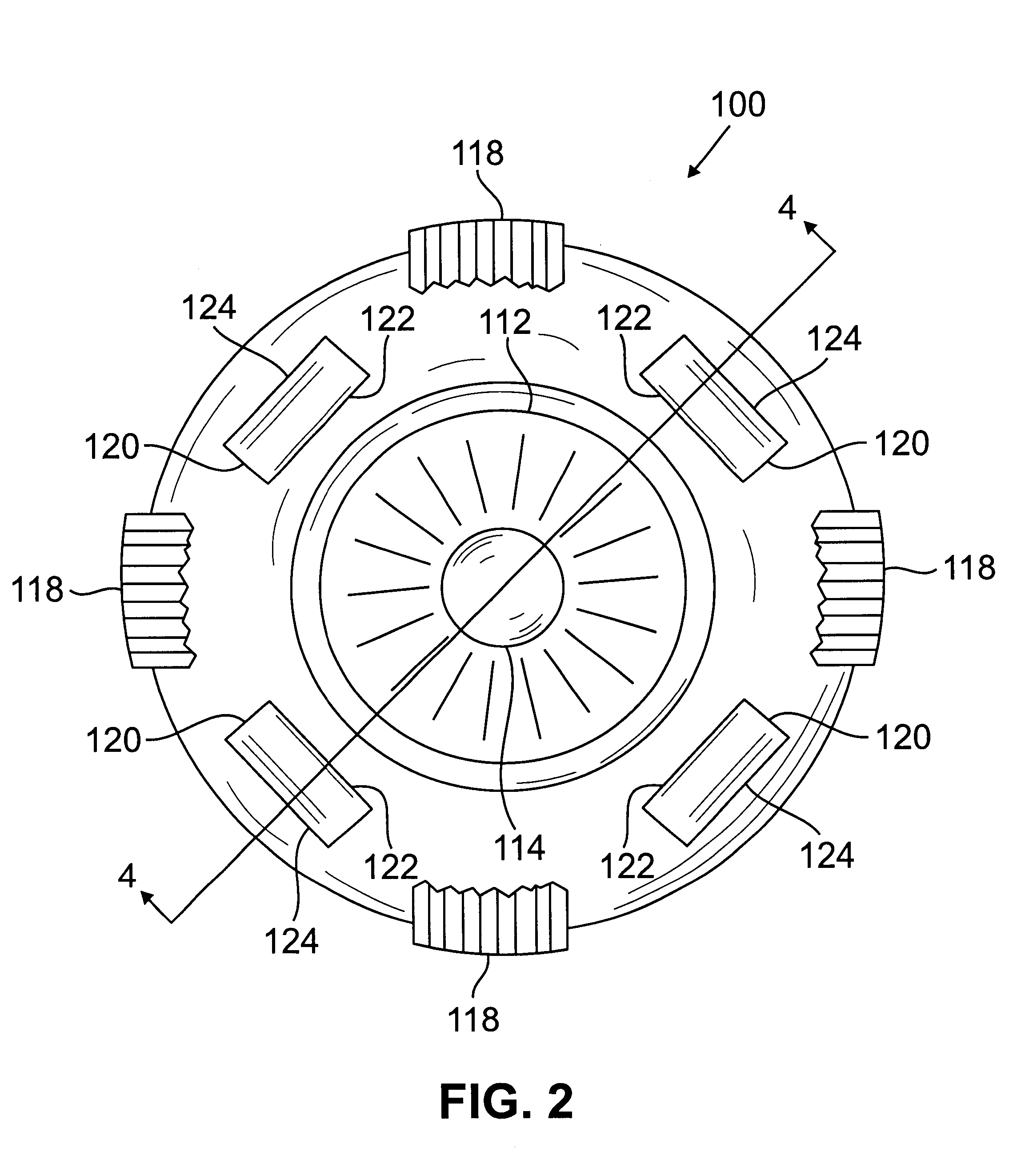
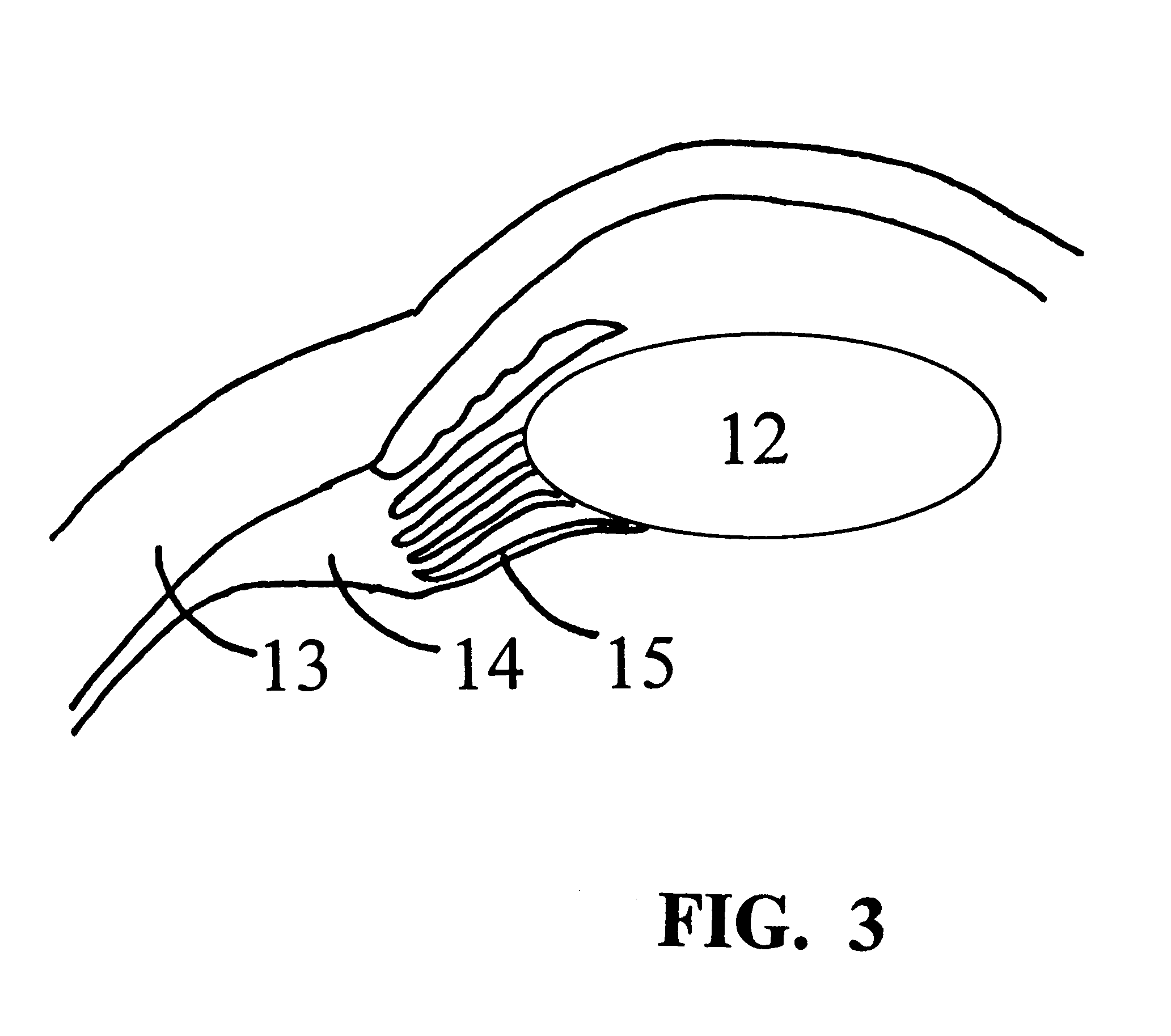
Segmented scleral band for treatment of presbyopia and other eye disorders
InactiveUS6197056B1Increase the effective working distanceLaser surgeryEye implantsDiseaseCiliary body
A segmented scleral expansion band adapted for implantation within or fastening to a segment of the sclera of an eye lying outside of and adjacent to the ciliary body of the eye, is formed from a number of arcuate segments, curved to match the curvature of the globe of the eye, and joined together at each end to form a complete scleral expansion band. The band is implanted in the sclera of the eye by forming circumferential tunnels, inserting the band segments through the tunnels, and joining the ends of the segments to form a complete scleral expansion band. The scleral expansion band is useful in treating presbyopia and other ocular disorders.
Owner:REFOCUS OCULAR INC
Scleral prosthesis for treatment of presbyopia and other eye disorders
InactiveUS6280468B1Increase the effective working distanceIncrease the working distanceLaser surgeryEye implantsDiseaseOpen angle glaucoma
Presbyopia is treated by implanting within a plurality of elongated pockets formed in the tissue of the sclera of the eye transverse to a meridian of the eye, a prosthesis having an elongated body having a first surface and a second surface opposite the first surface to contact the base and flap of the scleral pocket. The first and second surfaces are spaced apart a distance so that the implanted prosthesis exerts an outward force on the flap of the scleral pocket which results in an outward traction on at least the anterior margin of the scleral pocket. The combined effect of the implanted prostheses is to exert a radially outward traction on the sclera in the region overlying the ciliary body which expands the sclera in the affected region together with the underlying ciliary body. The expansion of the ciliary body restores the effective working distance of the ciliary muscle in the presbyopic eye and thereby increases the amplitude of accommodation. Hyperopia, primary open angle glaucoma and / or ocular hypertension can be treated by increasing the effective working distance of the ciliary muscle according to the invention. A preferred embodiment of the scleral prosthesis has a major surface adapted to contact the base or flap of the pocket and an opposite surface or ridge spaced from the major surface.
Owner:REFOCUS GROUP
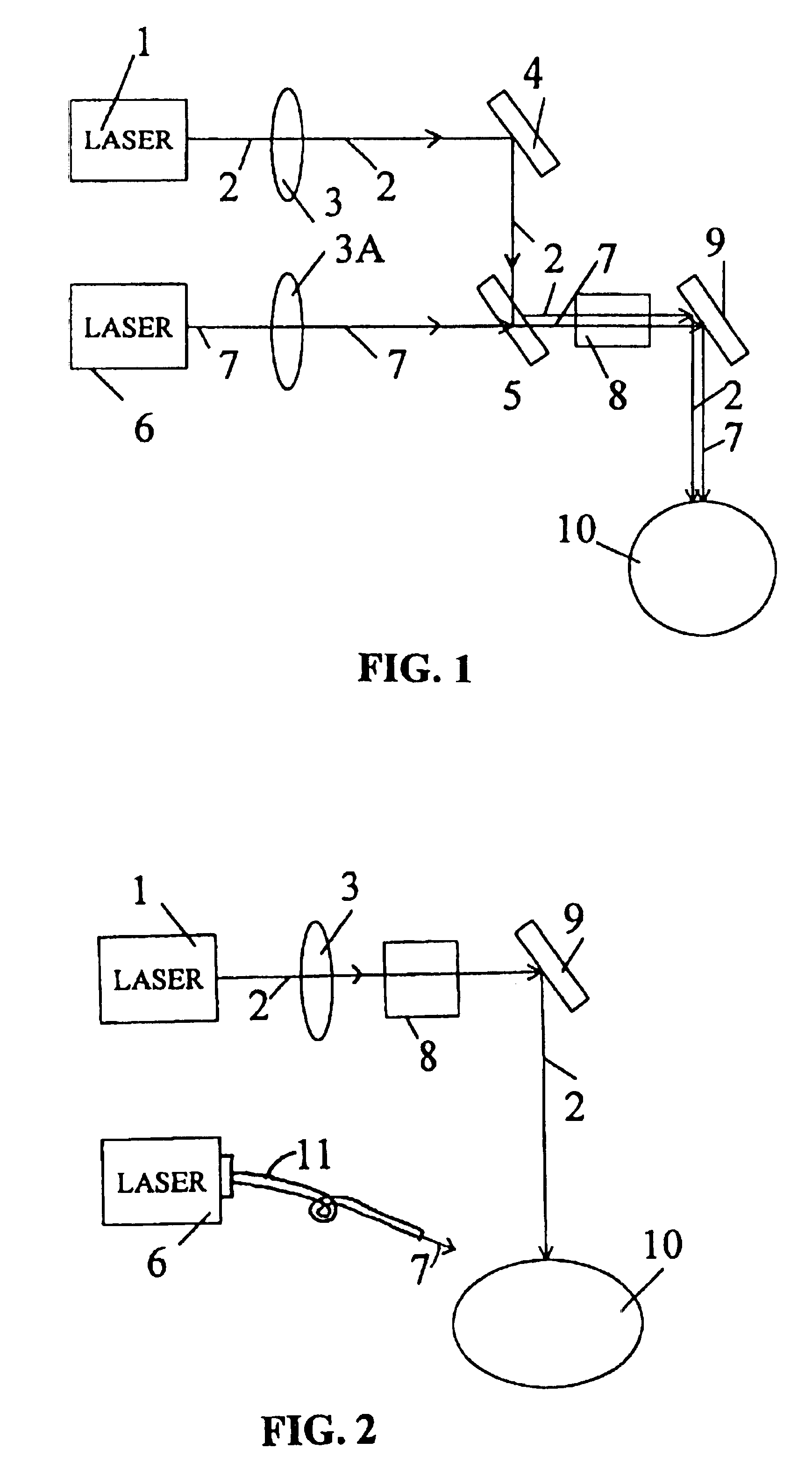
Treatment of presbyopia and other eye disorders using a scanning laser system
InactiveUS6263879B1Efficient and accurate expansionPreventing of open angle glaucomaLaser surgeryDiagnosticsDiseaseGlaucoma
Presbyopia is treated by a method which uses ablative lasers to ablate the sclera tissue and increase the accommodation of the ciliary body. Tissue bleeding is prevented by an ablative laser having a wavelength of between 0.15 and 3.2 micron. A scanning system is proposed to perform various patterns on the sclera area of the cornea to treat presbyopia and to prevent other eye disorder such as glaucoma. Laser parameters are determined for accurate sclera expansion.
Owner:NEOS OCULAR
Heterocyclic compounds and their uses
ActiveUS20090137581A1Low inhibitory potencyInhibitory activityBiocideSenses disorderDiseaseMyeloid leukemia
Substituted bicyclic heteroaryls and compositions containing them, for the treatment of general inflammation, arthritis, rheumatic diseases, osteoarthritis, inflammatory bowel disorders, inflammatory eye disorders, inflammatory or unstable bladder disorders, psoriasis, skin complaints with inflammatory components, chronic inflammatory conditions, including but not restricted to autoimmune diseases such as systemic lupus erythematosis (SLE), myestenia gravis, rheumatoid arthritis, acute disseminated encephalomyelitis, idiopathic thrombocytopenic purpura, multiples sclerosis, Sjoegren's syndrome and autoimmune hemolytic anemia, allergic conditions including all forms of hypersensitivity, The present invention also enables methods for treating cancers that are mediated, dependent on or associated with p110δ activity, including but not restricted to leukemias, such as Acute Myeloid leukaemia (AML) Myelo-dysplastic syndrome (MDS) myelo-proliferative diseases (MPD) Chronic Myeloid Leukemia (CML) T-cell Acute Lymphoblastic leukaemia (T-ALL) B-cell Acute Lymphoblastic leukaemia (B-ALL) Non Hodgkins Lymphoma (NHL) B-cell lymphoma and solid tumors, such as breast cancer.
Owner:AMGEN INC
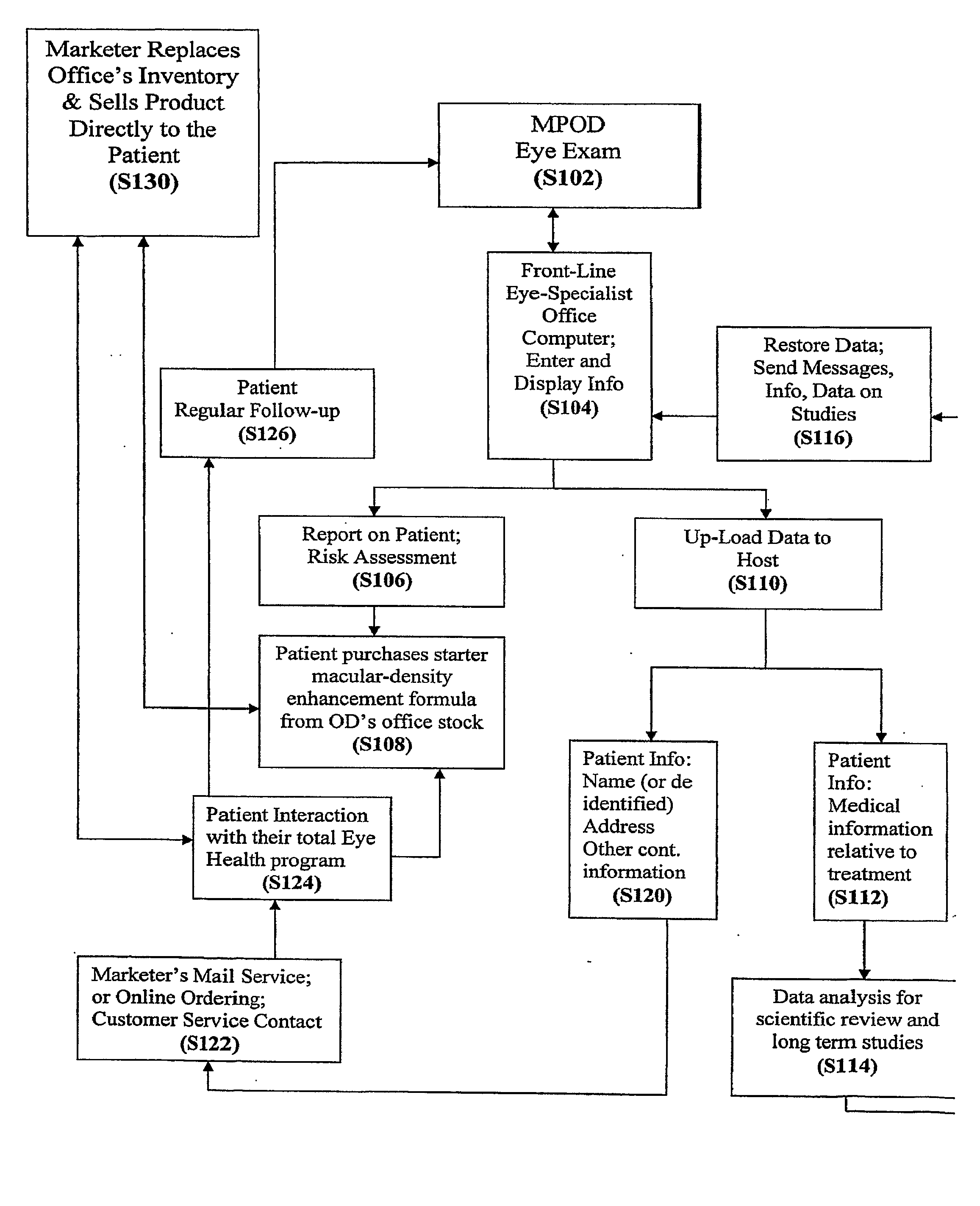
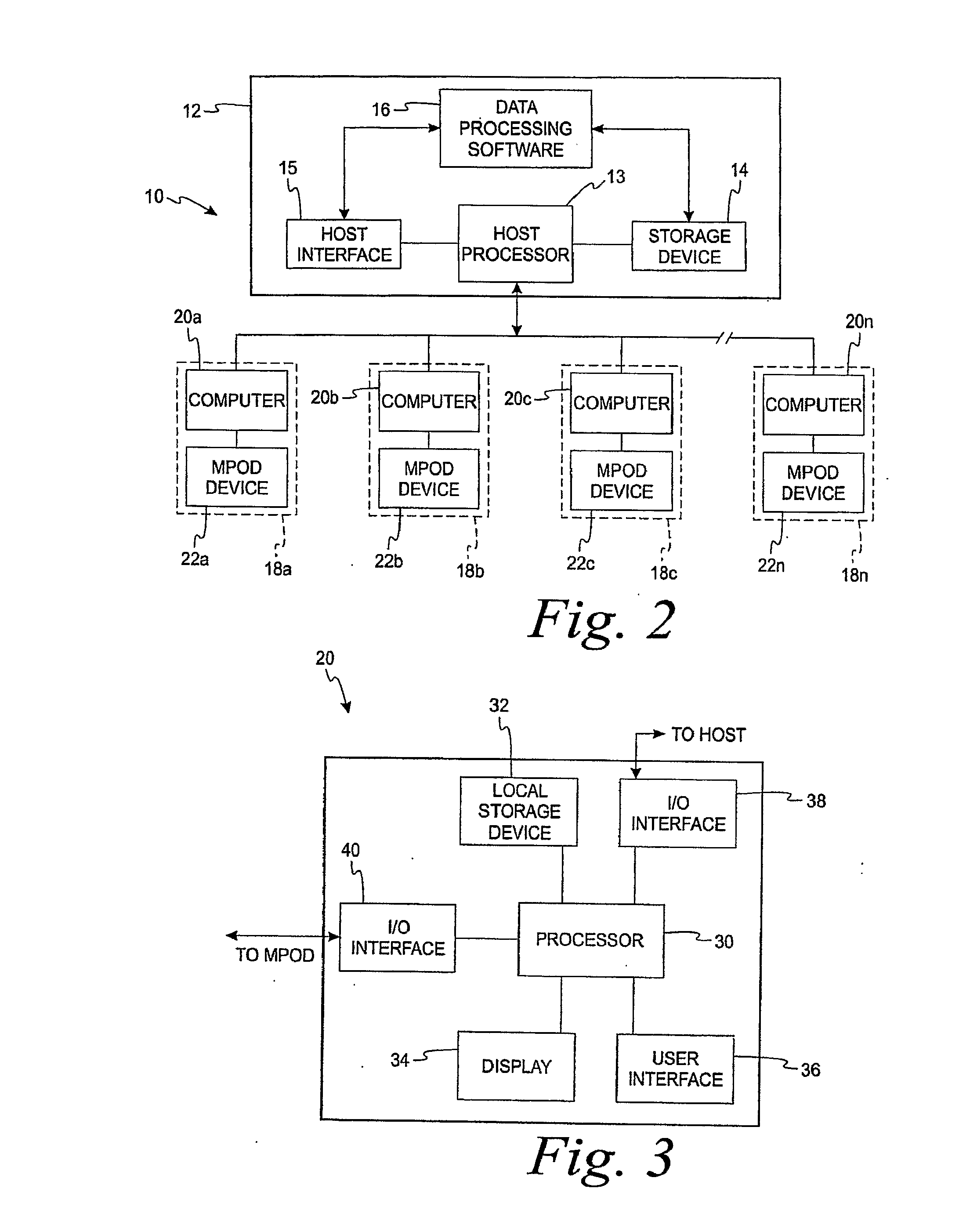
Diagnostic, Prescriptive, and Data-Gathering System and Method For Macular Pigment Deficits and Other Eye Disorders
ActiveUS20100241450A1Compact and inexpensive and well-suited for placement and operationInexpensive and convenientDrug and medicationsNutrition controlPatient dataMedical prescription
A macular health measurement and storage system comprises a plurality of macular-pigment measurement machine for measuring macular pigment density in humans, a plurality of computers each of which is associated with a corresponding one the macular-pigment measuring machines, and a central host. The plurality of macular-pigment measurement machines include a device for receiving macular pigment data from a patient, at least one data transfer port, and at least one processor that enables the transfer of the macular pigment data from the transfer port. The plurality of computers include a first port coupled to the data transfer port of the corresponding macular-pigment measurement machine for receiving the macular pigment data. Each of the computers includes a second port for transferring patient data. The central host is coupled to the second ports on each of the plurality of computers. The central host includes a storage device for storing the patient data.
Owner:ZEAVISION LLC
Compositions and methods for treatment of eye disorders
The present invention provides compounds and methods for the treatment of LFA-1 mediated diseases. In particular, LFA-1 antagonists are described herein and these antagonists are used in the treatment of LFA-1 mediated diseases. One aspect of the invention provides for diagnosis of an LFA-1 mediated disease and administration of a LFA-1 antagonist, after the patient is diagnosed with a LFA-1 mediated disease. In some embodiments, the LFA-1 mediated diseases treated are dry eye disorders. Also provided herein are methods for identifying compounds which are LFA-1 antagonists.
Owner:NOVARTIS PHARM CORP
Scleral prosthesis for treatment of presbyopia and other eye disorders
InactiveUS6299640B1Increase the effective working distanceIncrease the working distanceLaser surgeryEye implantsDiseaseOpen angle glaucoma
Presbyopia is treated by implanting within a plurality of elongated pockets formed in the tissue of the sclera of the eye transverse to a meridian of the eye, a prosthesis having an elongated base member having an inward surface adapted to be placed against the inward wall of the pocket and having a ridge on the inward surface of the base extending along at least a major portion of the major dimension of the base. The combined effect of the implanted prostheses is to exert a radially outward traction on the sclera in the region overlying the ciliary body which expands the sclera in the affected region together with the underlying ciliary body. The expansion of the ciliary body restores the effective working distance of the ciliary muscle in the presbyopic eye and thereby increases the amplitude of accommodation. Hyperopia, primary open angle glaucoma and / or ocular hypertension can be treated by increasing the effective working distance of the ciliary muscle according to the invention.
Owner:REFOCUS GROUP
Treatment of eye disorders with sirtuin modulators
InactiveUS20070014833A1Prevention of effect of generalInhibition effectBiocideSenses disorderDiseaseVisual acuity
Sirtuin modulators, particularly sirtuin activators, are useful in treating vision impairment. In general, the sirtuin modulators inhibit the progression of vision impairment resulting from various eye disorders. The invention also includes pharmaceutically acceptable formulations of sirtuin modulators, particular ophthalmically acceptable formulations.
Owner:SIRTRIS PHARMA INC
Treatment of eye disorders with sirtuin modulators
InactiveUS20060292099A1Prevention of effect of generalInhibition effectOrganic active ingredientsCosmetic preparationsDiseaseVisual acuity
Sirtuin modulators, particularly sirtuin activators, are useful in treating vision impairment. In general, the sirtuin modulators inhibit the progression of vision impairment resulting from various eye disorders. The invention also includes pharmaceutically acceptable formulations of sirtuin modulators, particular ophthalmically acceptable formulations.
Owner:SIRTRIS PHARMA INC
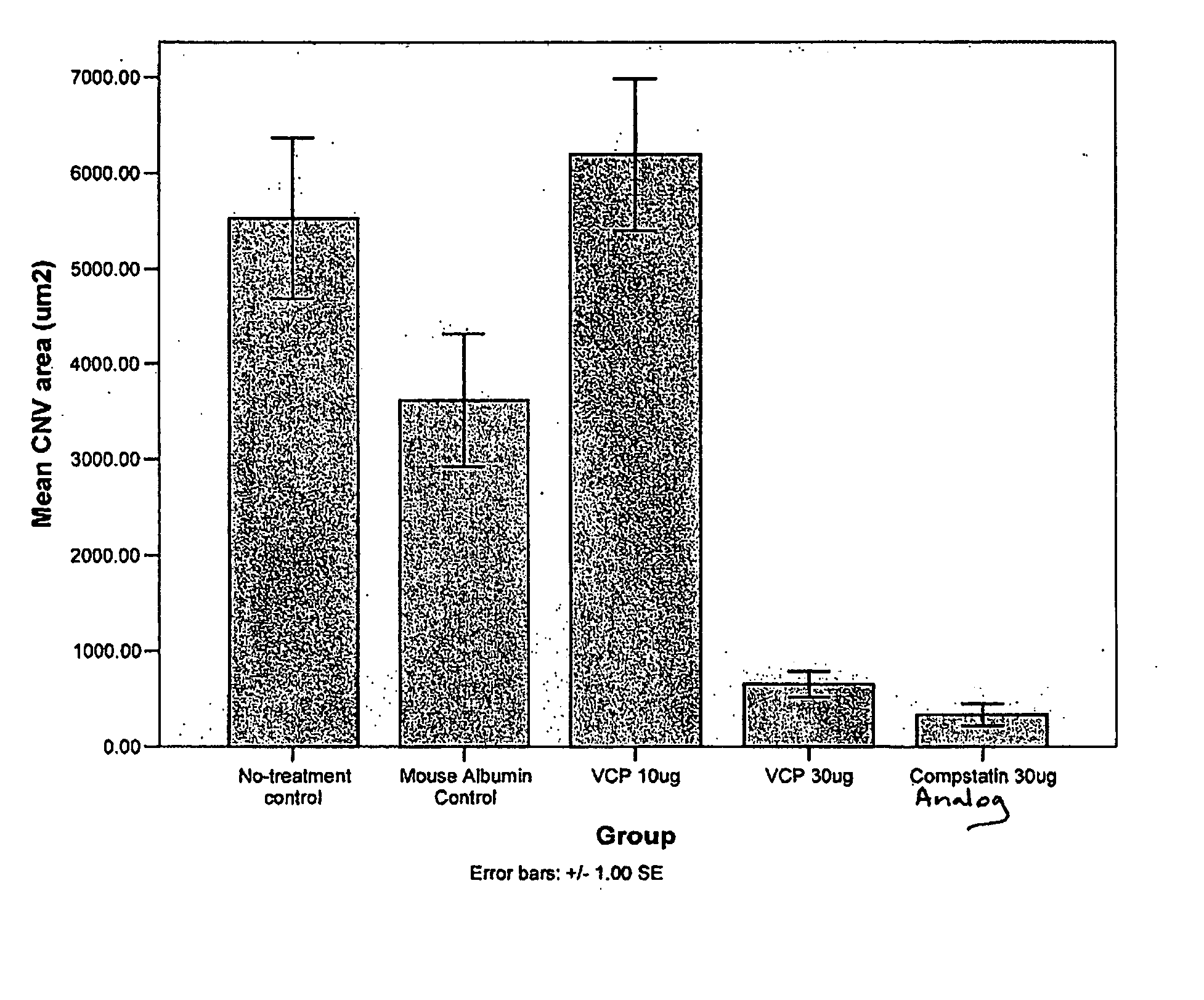
Compstatin and analogs thereof for eye disorders
ActiveUS20070238654A1Inhibit expressionImprove in vivo stabilityCompounds screening/testingOrganic active ingredientsDiseaseRetinal neovascularization
The present invention features the use of compstatin and complement inhibiting analogs thereof for treating and / or preventing age related macular degeneration and other conditions involving macular degeneration, choroidal neovascularization, and / or retinal neovascularization. The invention also provides compositions comprising compstatin or a complement inhibiting analog thereof and a second therapeutic agent. The invention also provides compositions comprising compstatin or a complement inhibiting analog thereof and a gel-forming material, e.g., soluble collagen, and methods of administering the compositions.
Owner:APELLIS PHARMA
Heterocyclic compounds and their uses
InactiveUS20100331306A1Inhibit biological activityLow inhibitory potencyBiocideSenses disorderDiseaseB-cell acute lymphoblastic leukaemia
Substituted bicyclic heteroaryls and compositions containing them, for the treatment of general inflammation, arthritis, rheumatic diseases, osteoarthritis, inflammatory bowel disorders, inflammatory eye disorders, inflammatory or unstable bladder disorders, psoriasis, skin complaints with inflammatory components, chronic inflammatory conditions, including but not restricted to autoimmune diseases such as systemic lupus erythematosis (SLE), myestenia gravis, rheumatoid arthritis, acute disseminated encephalomyelitis, idiopathic thrombocytopenic purpura, multiples sclerosis, Sjoegren's syndrome and autoimmune hemolytic anemia, allergic conditions including all forms of hypersensitivity. The present invention also enables methods for treating cancers that are mediated, dependent on or associated with p110δ activity, including but not restricted to leukemias, such as Acute Myeloid leukaemia (AML) Myelo-dysplastic syndrome (MDS) myelo-proliferative diseases (MPD) Chronic Myeloid Leukemia (CML) T-cell Acute Lymphoblastic leukaemia (T-ALL) B-cell Acute Lymphoblastic leukaemia (B-ALL) Non Hodgkins Lymphoma (NHL) B-cell lymphoma and solid tumors, such as breast cancer.
Owner:AMGEN INC
Methods and compositions for treating eye disorders
InactiveUS20040234532A1Efficiently provideAntibacterial agentsBacterial antigen ingredientsUveitisViral Conjunctivitis
The present invention provides methods of treating an eye disorder. The methods comprise a step of locally administering a Clostridial toxin to the eye of a patient to treat the disorder. The eye disorder may be associated with an inflammation of the eye, including for example, bacterial conjunctivitis, fungal conjunctivitis, viral conjunctivitis, uveitis, keratic precipitates, macular edema, and inflammation response after intra-ocular lens implantation. The Clostridial toxin may be produced by a Clostridial beratti, a Clostridia butyricum, a Clostridial tetani bacterium and / or a Clostridial botulinum.
Owner:ALLERGAN INC
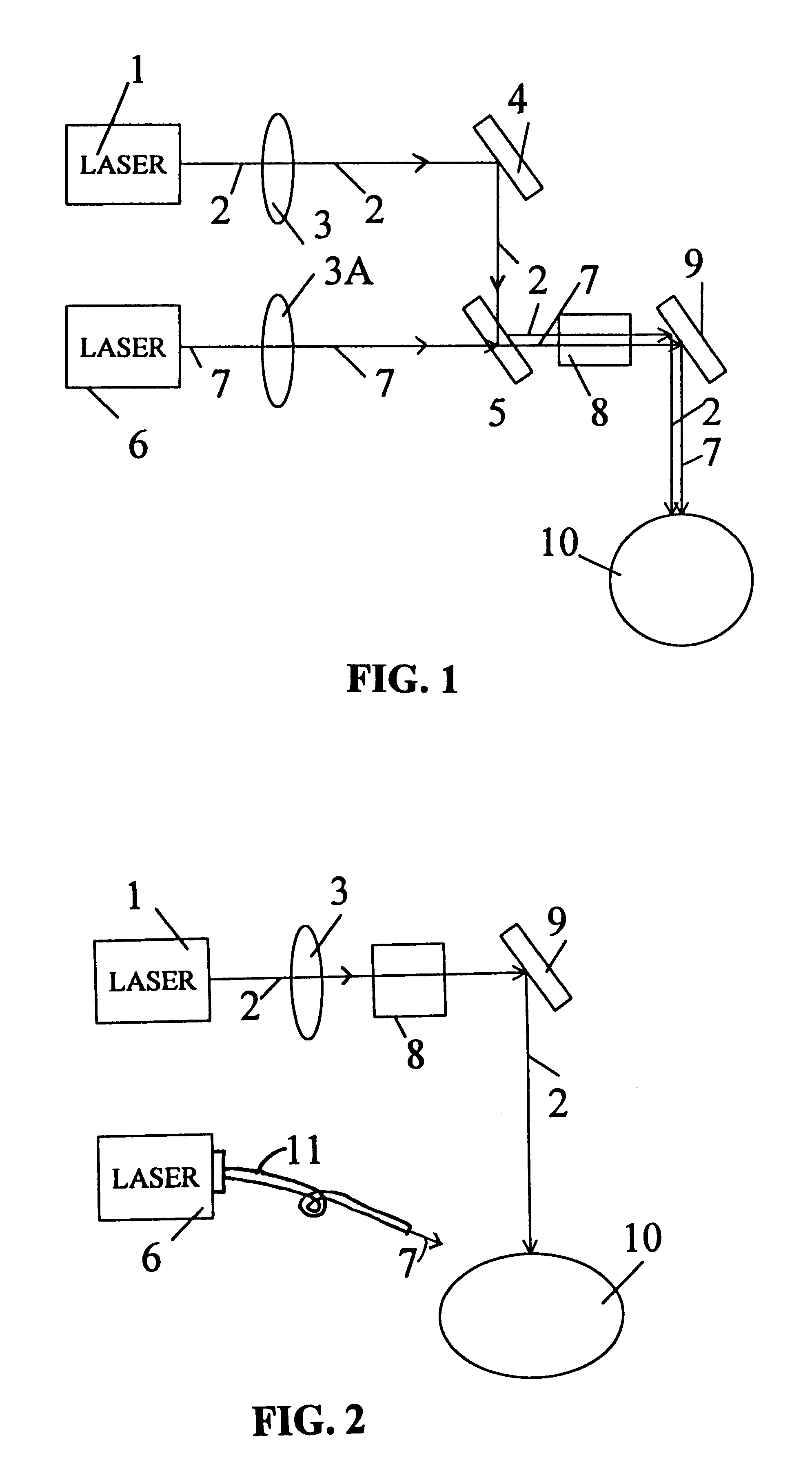
Treatment of eye disorders using articulated-arm coupled ultraviolet lasers
InactiveUS20060129141A1Increase flexibilityIncrease spacingLaser surgerySurgical instrument detailsIntra ocular pressureDisease
Surgical method and apparatus for presbyopia correction and glaucoma by laser removal a portion of the sclera and / or ciliary tissue are disclosed. The disclosed preferred embodiments of the system consists of a beam spot controller, an articulated arm and an attached end-piece. The basic laser beam includes UV laser having wavelength ranges of (0.19-0.36) microns, generated from UV excimer lasers of ArF, XeCl or solid state lasers of Nd:YLF, Nd:YAG, Ti:sapphire with harmonic generation using nonlinear crystals. Presbyopia is treated by ablation of the treated surface tissue in predetermined patterns outside the limbus to increase the accommodation of the eye. Glaucoma is treated by decreasing of intra ocular pressure of the laser surgery.
Owner:NEW VISION
Agent for treating eye diseases
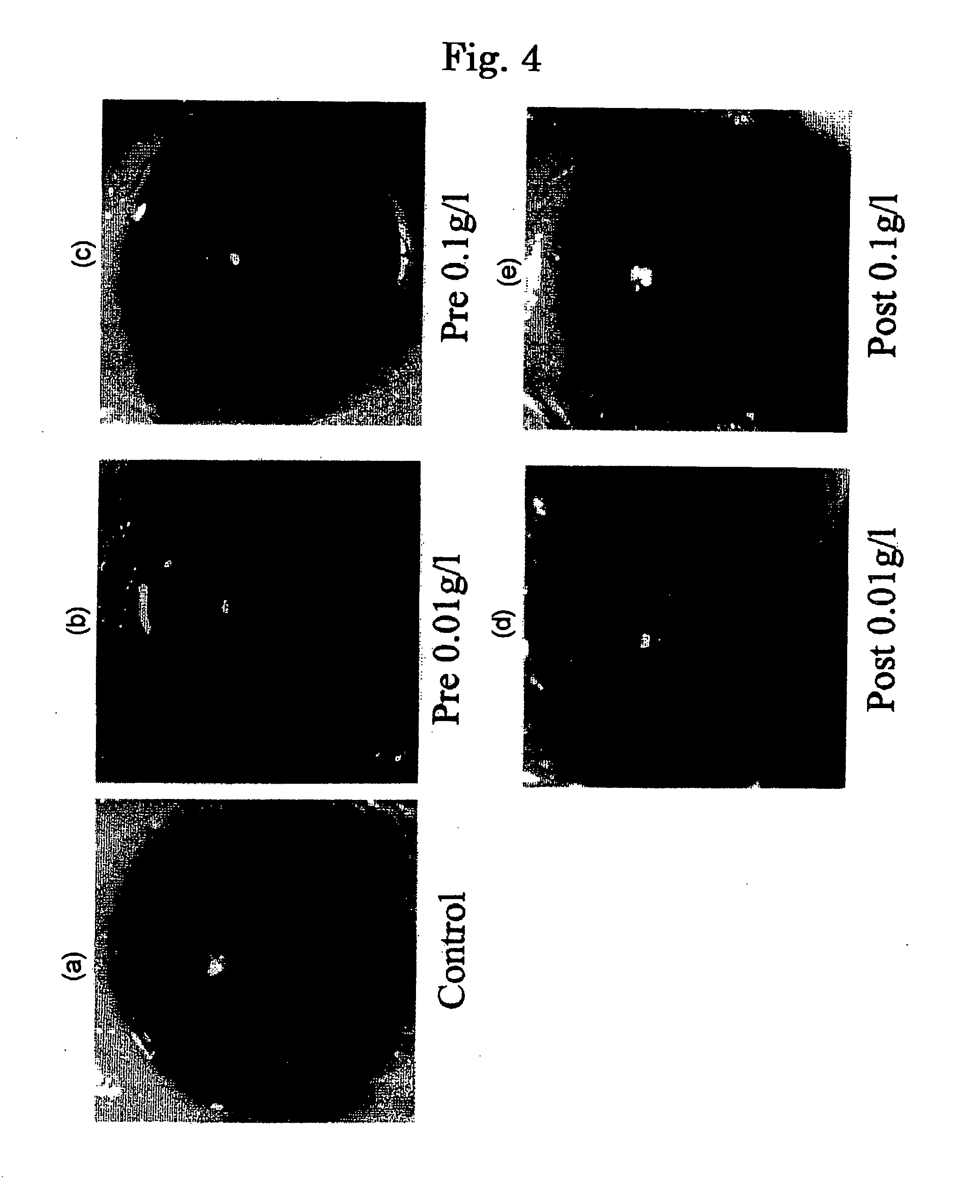
An agent for treating various kinds of diseases which contains at least one selected from the group consisting of sexual steroid hormone such as estrogen or its metabolites, its derivative, structural analogues thereof, estrogen acting substance or SERM non-feminizing estrogen (non-hormonal estrogen), and an activator of sirtuin and has a form of eye drops or eye washes, oral preparation, etc. An agent for treating eye diseases which has excellent treatment effects, reduced in side action can be provided.
Owner:ADVANCED MEDICINE RES INST
Heterocyclic compounds and their uses
Substituted bicyclic heteroaryls and compositions containing them, for the treatment of general inflammation, arthritis, rheumatic diseases, osteoarthritis, inflammatory bowel disorders, inflammatory eye disorders, inflammatory or unstable bladder disorders, psoriasis, skin complaints with inflammatory components, chronic inflammatory conditions, including but not restricted to autoimmune diseases such as systemic lupus erythematosis (SLE), myestenia gravis, rheumatoid arthritis, acute disseminated encephalomyelitis, idiopathic thrombocytopenic purpura, multiples sclerosis, Sjoegren's syndrome and autoimmune hemolytic anemia, allergic conditions including all forms of hypersensitivity, The present invention also enables methods for treating cancers that are mediated, dependent on or associated with p110δ activity, including but not restricted to leukemias, such as Acute Myeloid leukaemia (AML) Myelo-dysplastic syndrome (MDS) myelo-proliferative diseases (MPD) Chronic Myeloid Leukemia (CML) T-cell Acute Lymphoblastic leukaemia (T-ALL) B-cell Acute Lymphoblastic leukaemia (B-ALL) Non Hodgkins Lymphoma (NHL) B-cell lymphoma and solid tumors, such as breast cancer.
Owner:AMGEN INC
Compositions and methods for treating eye disorders and conditions
Owner:THE SCHEPENS EYE RES INST +1
Inflammatory eye disorders
ActiveUS20130336557A1Improve reflectivityIncrease in sizeOrganic active ingredientsSenses disorderClinical studyOcular disease
Provided herein are methods of evaluating efficacy of a treatment in a subject having eye inflammation (e.g., a subject having dry eye syndrome) and selecting a subject for participation in a clinical study. Also provided are methods of treating a subject having eye inflammation (e.g., a subject having dry eye syndrome).
Owner:MASSACHUSETTS EYE & EAR INFARY
Methods for treating eye disorders
Owner:TATTON WILLIAM G
Heterocyclic compounds and their uses
Substituted bicyclic heteroaryls and compositions containing them, for the treatment of general inflammation, arthritis, rheumatic diseases, osteoarthritis, inflammatory bowel disorders, inflammatory eye disorders, inflammatory or unstable bladder disorders, psoriasis, skin complaints with inflammatory components, chronic inflammatory conditions, including but not restricted to autoimmune diseases such as systemic lupus erythematosis (SLE), myestenia gravis, rheumatoid arthritis, acute disseminated encephalomyelitis, idiopathic thrombocytopenic purpura, multiples sclerosis, Sjoegren's syndrome and autoimmune hemolytic anemia, allergic conditions including all forms of hypersensitivity, The present invention also enables methods for treating cancers that are mediated, dependent on or associated with p110δ activity, including but not restricted to leukemias, such as Acute Myeloid leukaemia (AML) Myelodysplastic syndrome (MDS) myelo-proliferative diseases (MPD) Chronic Myeloid Leukemia (CML) T-cell Acute Lymphoblastic leukaemia (T-ALL) B-cell Acute Lymphoblastic leukaemia (B-ALL) Non Hodgkins Lymphoma (NHL) B-cell lymphoma and solid tumors, such as breast cancer.
Owner:AMGEN INC
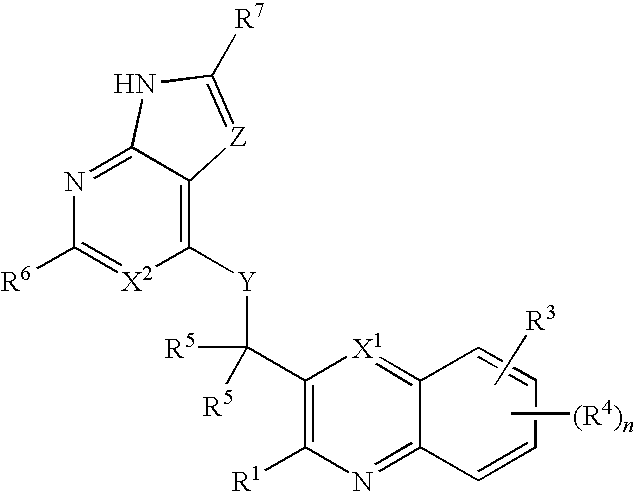
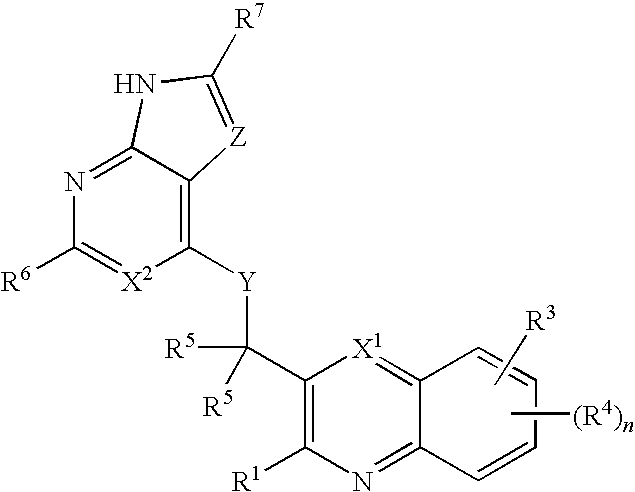
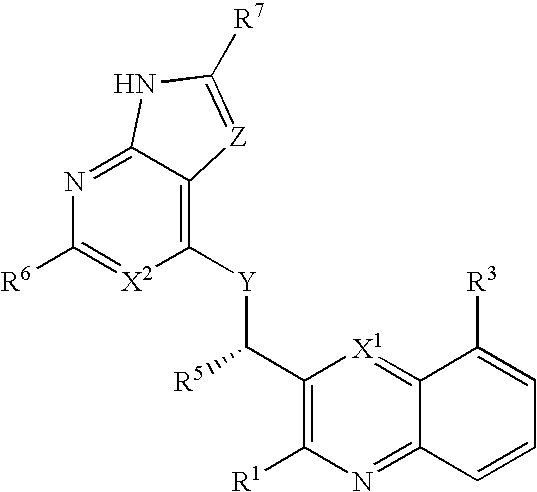
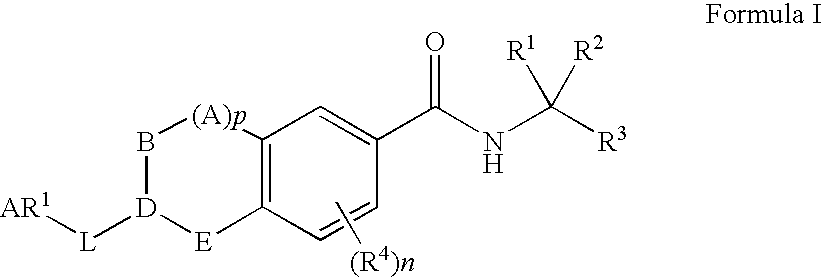
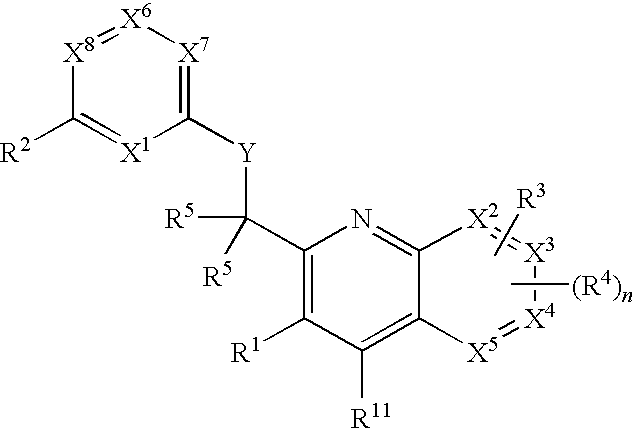
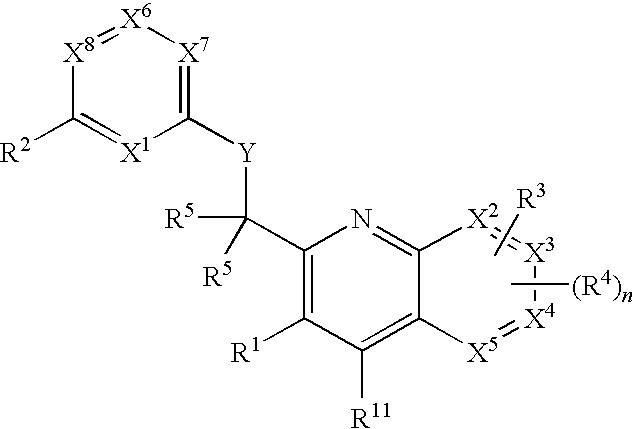
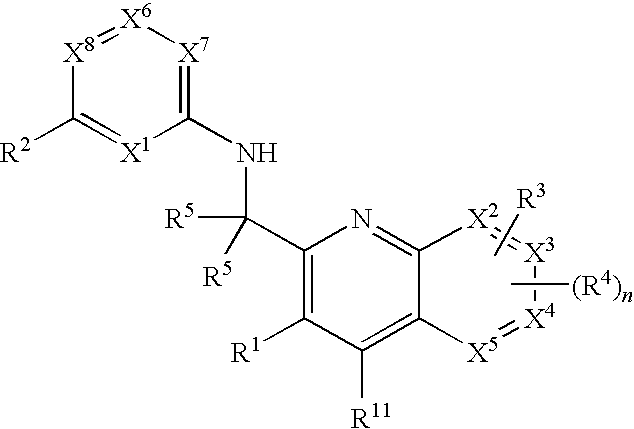
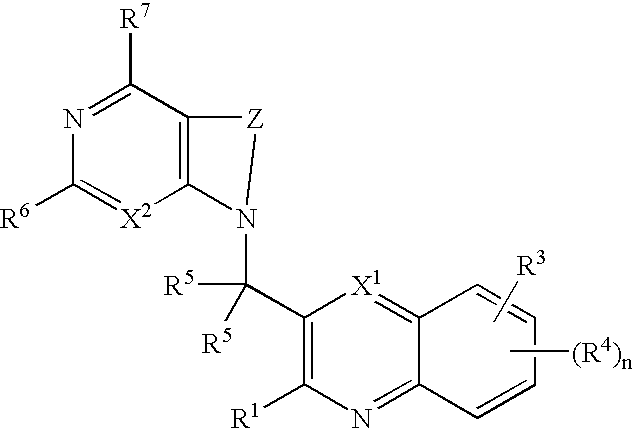
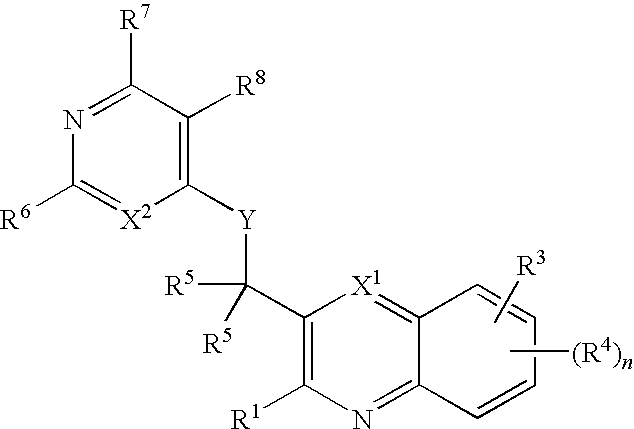
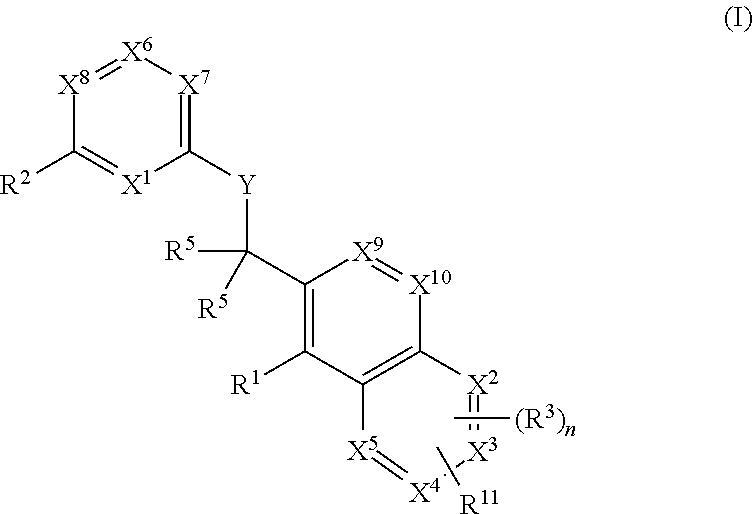
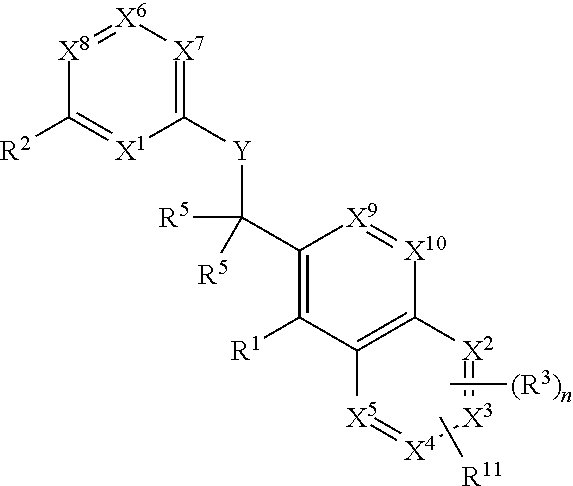
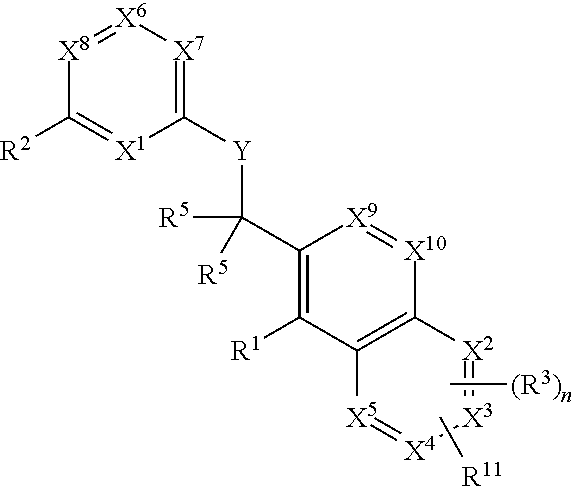
Substituted quinolines and their uses in treatment of inflammatory and related conditions
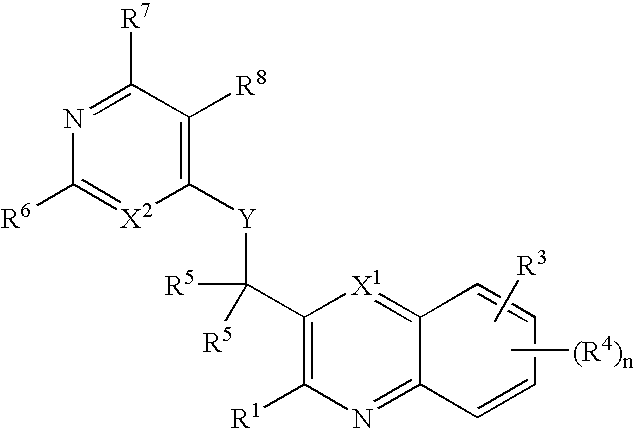
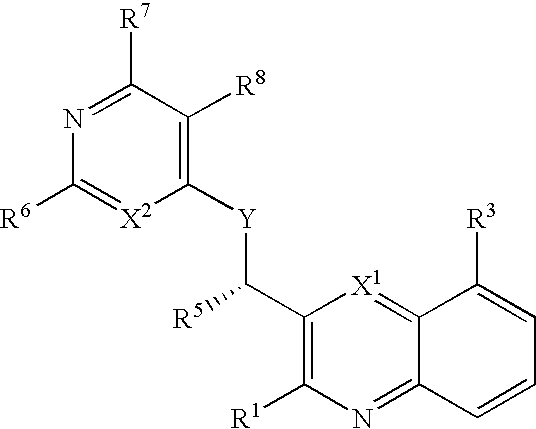
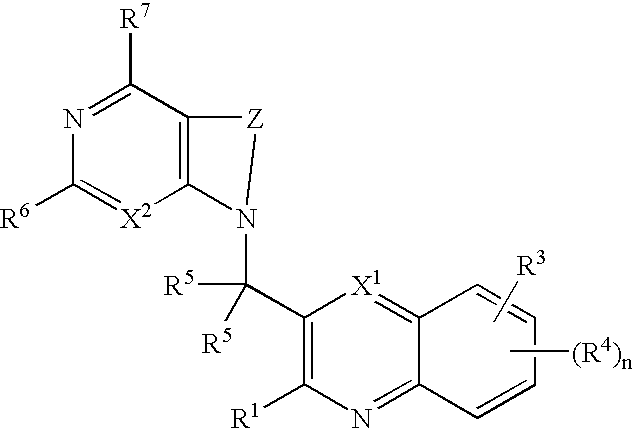
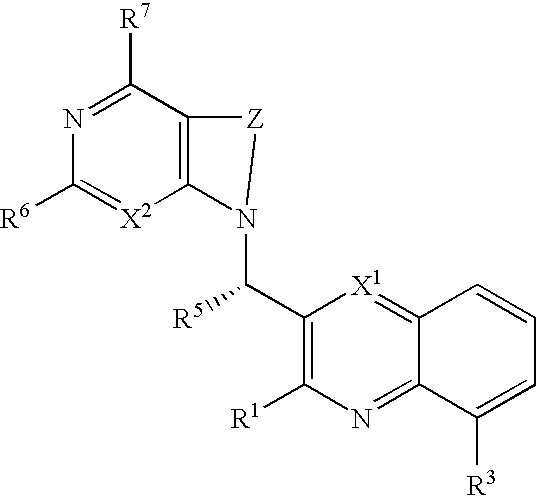
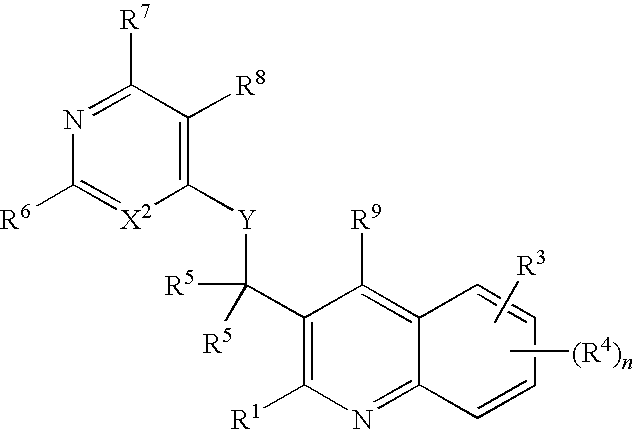
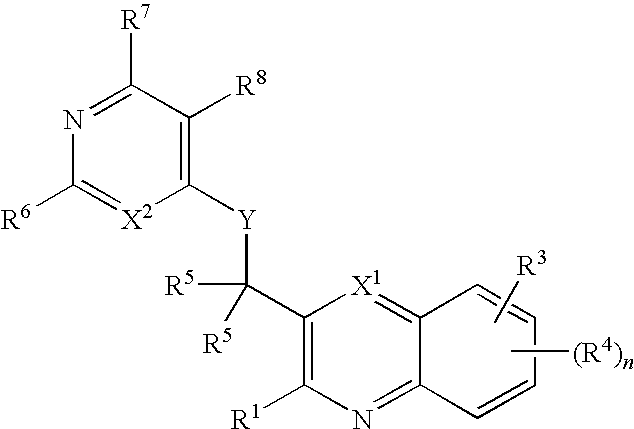
Substituted bicyclic heteroaryls having the general formulaand compositions containing them, for the treatment of general inflammation, arthritis, rheumatic diseases, osteoarthritis, inflammatory bowel disorders, inflammatory eye disorders, inflammatory or unstable bladder disorders, psoriasis, skin complaints with inflammatory components, chronic inflammatory conditions, including but not restricted to autoimmune diseases such as systemic lupus erythematosis (SLE), myestenia gravis, rheumatoid arthritis, acute disseminated encephalomyelitis, idiopathic thrombocytopenic purpura, multiples sclerosis, Sjoegren's syndrome and autoimmune hemolytic anemia, allergic conditions including all forms of hypersensitivity, The present invention also enables methods for treating cancers that are mediated, dependent on or associated with p110δ activity, including but not restricted to leukemias, such as Acute Myeloid leukaemia (AML) Myelo-dysplastic syndrome (MDS) myelo-proliferative diseases (MPD) Chronic Myeloid Leukemia (CML) T-cell Acute Lymphoblastic leukaemia (T-ALL) B-cell Acute Lymphoblastic leukaemia (B-ALL) Non Hodgkins Lymphoma (NHL) B-cell lymphoma and solid tumors, such as breast cancer.
Owner:AMGEN INC
Compositions and methods for treating eye disorders and conditions
Owner:THE SCHEPENS EYE RES INST +1
Methods of treating epithelial lesions
InactiveUS20060188471A1Altered pharmacokinetic and pharmacodynamic profileIncreased serum half-lifeBiocideSenses disorderDiseaseTrefoil domain
The invention features methods of preventing or treating epithelial cell lesions in a mammal by administering a composition containing a therapeutically effective amount of a trefoil domain-containing polypeptide, or a trefoil peptide fragment, and a mucoadhesive excipient. The invention further features methods of preventing or treating an eye disorder, e.g., dry eye, by topically administering to the eye a composition containing a therapeutically effective amount of a trefoil domain-containing polypeptide, or a trefoil peptide fragment, and a mucoadhesive excipient. Compositions containing a trefoil domain-containing polypeptide, or a trefoil peptide fragment, and a mucoadhesive excipient may be formulated in combination with one or more additional therapeutic agents and used in the methods of the invention.
Owner:THE GENERAL HOSPITAL CORP +1
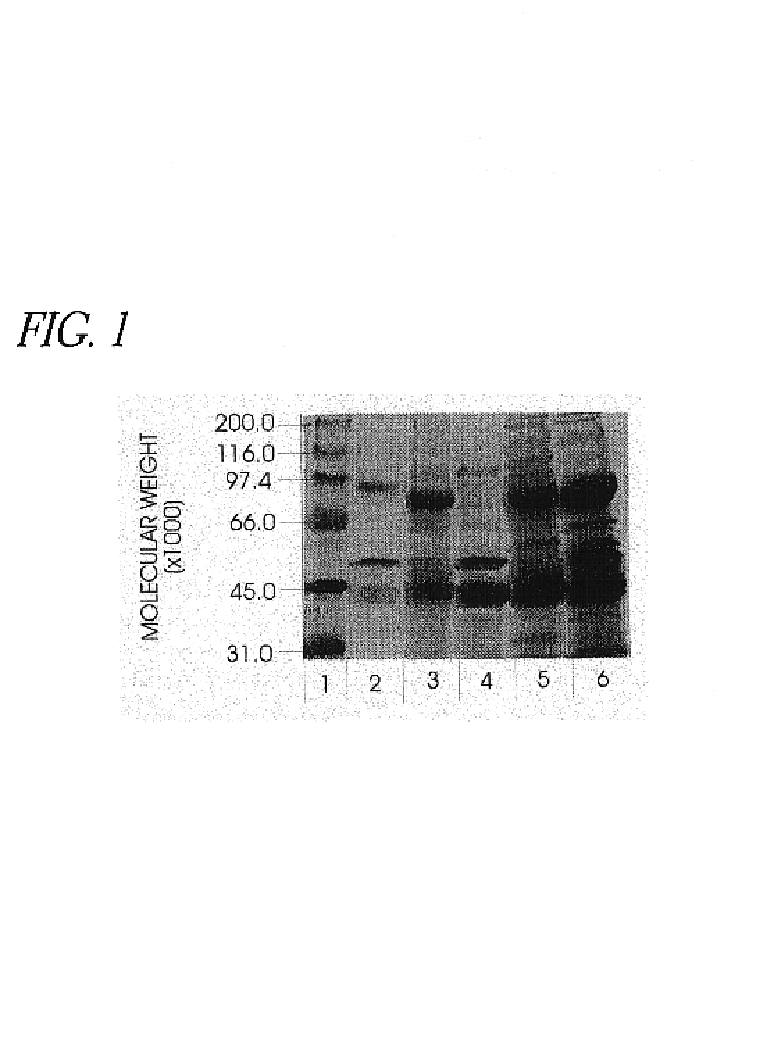
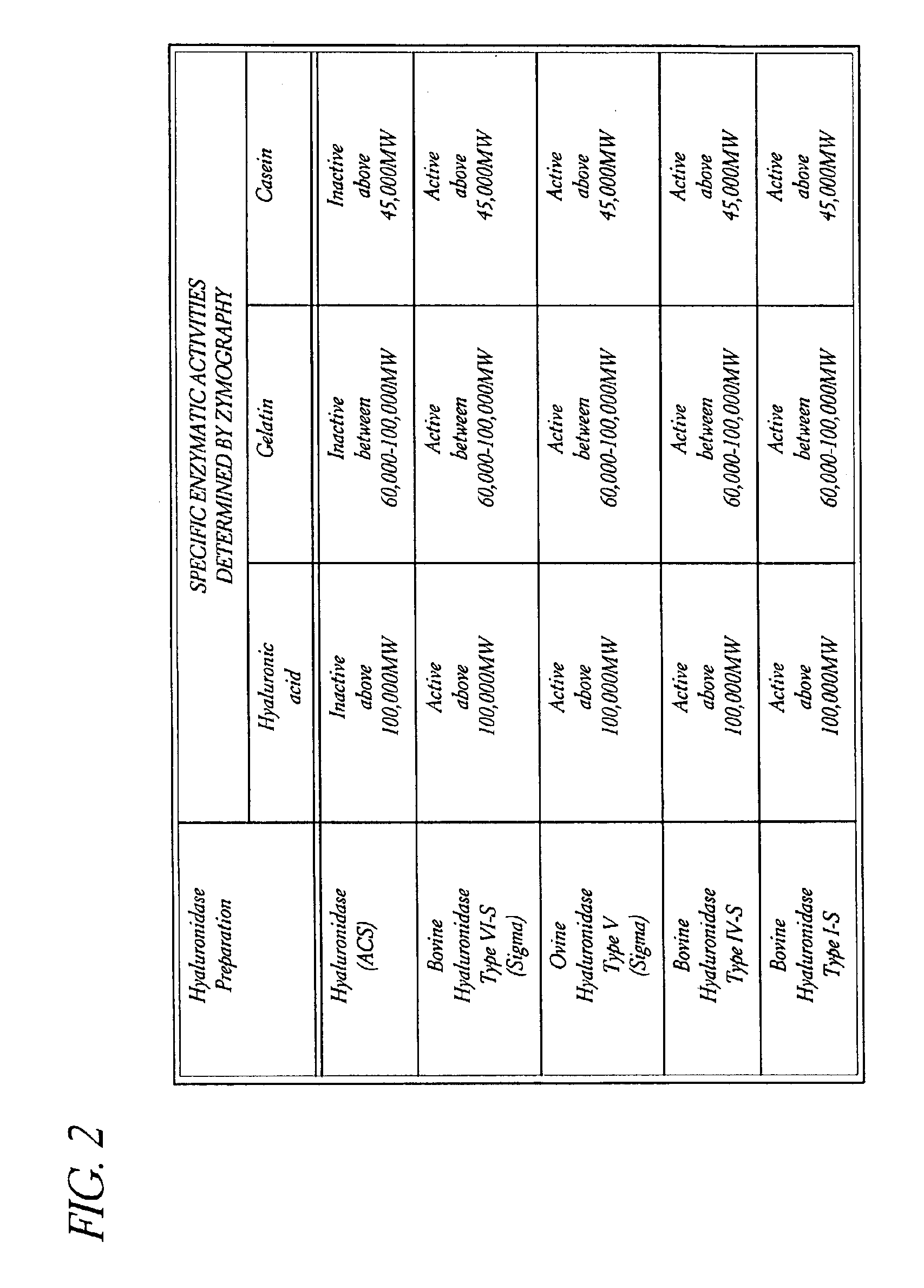
Use of hyaluronidase in the manufacture of an ophthalmic preparation for liquefying vitreous humor in the treatment of eye disorders
InactiveUS6863886B2Improve clearance rateIncreases rate of liquid exchangeBiocidePeptide/protein ingredientsDiseaseHyaluronidase
An enzymatic method is provided for treating ophthalmic disorders of the mammalian eye. Prevention of neovascularization and the increased rate of clearance from the vitreous of materials toxic to retina is accomplished by administering an amount of hyaluronidase effective to liquefy the vitreous humor of the treated eye without causing toxic damage to the eye. Liquefaction of the vitreous humor increases the rate of liquid exchange from the vitreal chamber. This increase in exchange removes those materials and conditions whose presence causes ophthalmological and retinal damage.
Owner:BAUSCH & LOMB PHARMA HLDG
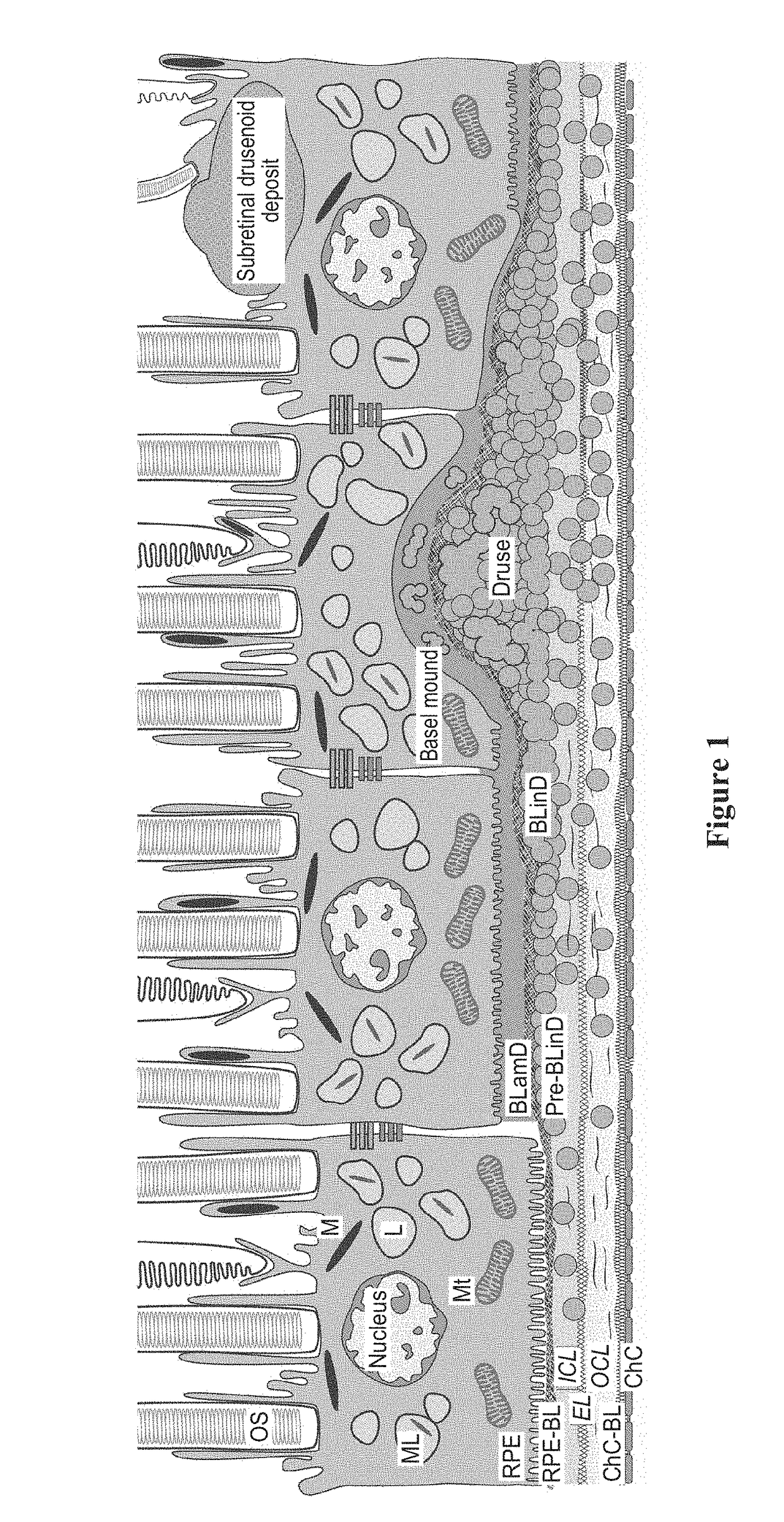
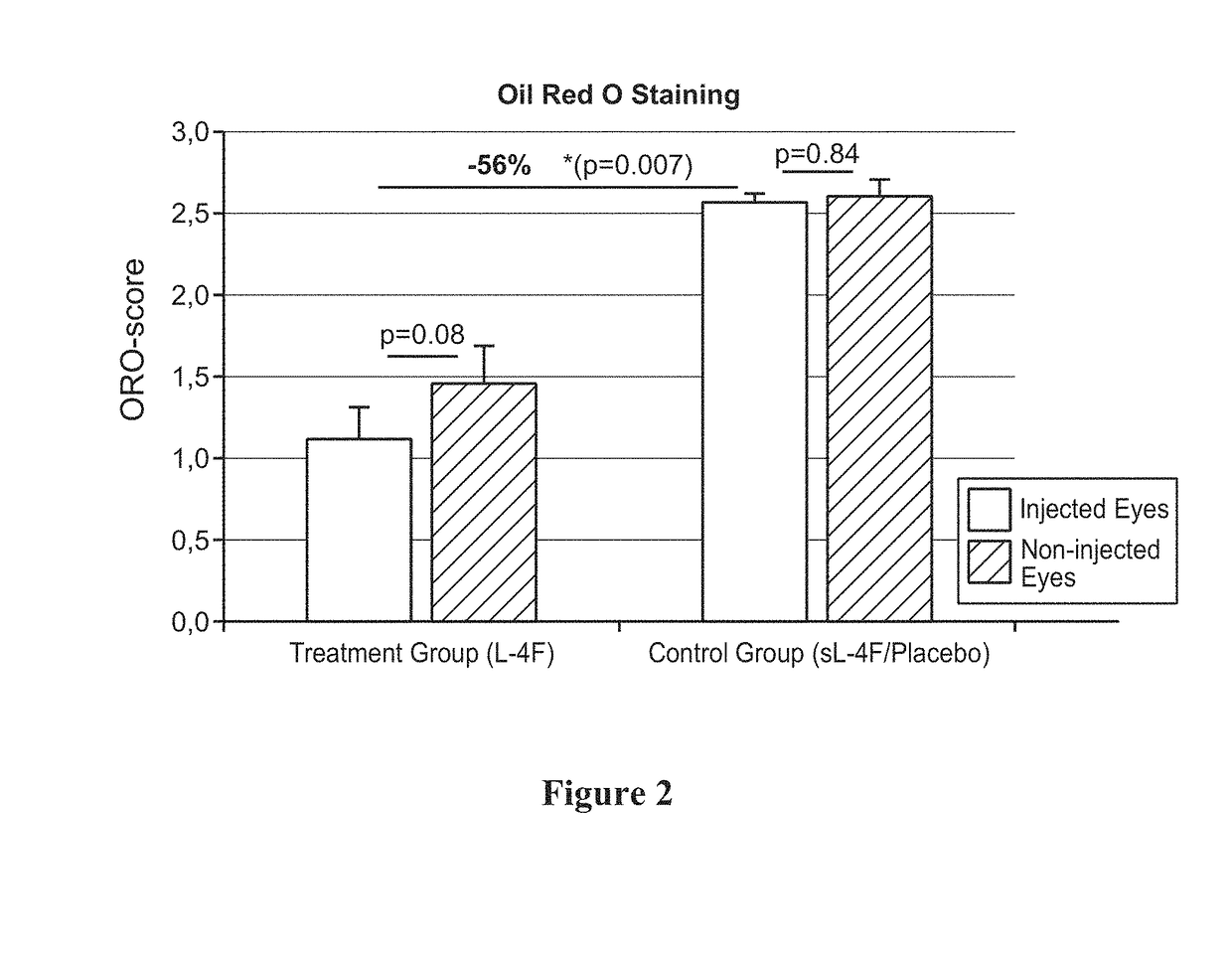
Topical delivery of therapeutic agents using cell-penetrating peptides for the treatment of age-related macular degeneration and other eye diseases
ActiveUS20190015521A1Hydroxy compound active ingredientsPeptide/protein ingredientsAntioxidantApolipoproteins E
The present disclosure provides therapeutic agents for the treatment of age-related macular degeneration (AMD) and other eye disorders. One or more therapeutic agents can be used to treat any stages (including the early, intermediate and advance stages) of AMD, and any phenotypes of AMD, including geographic atrophy (including non-central GA and central GA) and neovascularization (including types 1, 2 and 3 NV). In some embodiments, the one or more therapeutic agents are or include an anti-dyslipidemic agent, an antioxidant, an anti-inflammatory agent, a complement inhibitor, a neuroprotector or an anti-angiogenic agent, or any combination thereof. In certain embodiments, the one or more therapeutic agents are or include an anti-dyslipidemic agent (e.g., an apolipoprotein mimetic or / and a statin). In some embodiments, the one or more therapeutic agents are mixed with, non-covalently associated with or covalently bonded to a cell-penetrating peptide (CPP), encapsulated in CPP-conjugated nanoparticles, micelles or liposomes, or modified (e.g., stapled, prenylated, lipidated or coupled to a small-molecule α-helix mimic) to acquire membrane-translocating ability. In certain embodiments, the one or more therapeutic agents are administered by eye drop.
Owner:MACREGEN INC
Method for treating visual impairment through the prophylactic administration of a Morinda citrifolia-based naturaceutical
InactiveUS20030134002A1Promote resultsInhibit and prevent and reverse macular degenerationBiocideUnknown materialsDiabetic retinopathyRetinitis pigmentosa
Implementation of the present invention takes place in association with the utilization of one or more processed products produced from the Indian Mulberry plant, scientifically known as Morinda citrifolia L., to treat one or more eye disorders that affect vision, such as glaucoma, diabetic retinopathy, retinitis pigmentosa, cataracts, age-related macular degeneration, night blindness, color blindness, and other related conditions. The processed Morinda citrifolia products from the Indian Mulberry plant may be in the form of a dietary supplement, eye drops, or in another suitable form.
Owner:TAHITIAN NONI INT INC
Viral complement control proteins for eye disorders
InactiveUS20060142191A1Inhibit expressionImprove in vivo stabilityBiocideSenses disorderDiseaseCOMPLEMENT REGULATORS
The present invention provides compositions and methods for treating and / or preventing age related macular degeneration and other conditions involving macular degeneration or choroidal neovascularization. Certain of the compositions comprise a poxvirus complement control protein or a complement binding fragment or variant thereof. Other compositions comprise a poxvirus complement control protein linked to a moiety that binds to a component present on or at the surface of cell or noncellular molecular entity, e.g., a component present in the eye of a subject at risk of or suffering from age related macular degeneration or a related condition or choroidal neovascularization. Certain of the methods comprise administering a poxvirus complement control protein or complement binding fragment or variant thereof to a subject.
Owner:POTENTIA PHARMA INC
Sustained release implants and methods for subretinal delivery of bioactive agents to treat or prevent retinal disease
InactiveUS8003124B2Quantity maximizationDesired flexibilitySenses disorderEye implantsDiseaseActive agent
The invention relates to sustained release implants and to methods for treating eyes, particularly the eyes of mammals having eye disorders or diseases. By using the implants and methods described herein, the delivery of the one or more bioactive agents can be localized at a desired treatment site, particularly the choroid and the retina.
Owner:SURMODICS INC
Treatment of presbyopia and other eye disorders using a scanning laser system
Presbyopia is treated by a method which uses ablative lasers to ablate the sclera tissue and increase the accommodation of the ciliary body. Tissue bleeding is prevented by an ablative laser having a wavelength of between 0.15 and 3.2 micron. A scanning system is proposed to perform various patterns on the sclera area of the cornea to treat presbyopia and to prevent other eye disorder such as glaucoma. Laser parameters are determined for accurate sclera expansion.REEXAMINATION RESULTSThe questions raised in reexamination request no. 90 / 006,090, filed Aug. 22, 2001, have been considered and the results thereof are reflected in this reissue patent which constitutes the reexamination certificate required by 35 U.S.C. 307 as provided in 37 CFR 1.570(e), for ex parte reexaminations, or the reexamination certificate required by 35 U.S.C. 316 as provided in 37 CFR 1.997(e) for inter partes reexaminations.
Owner:SURGILIGHT
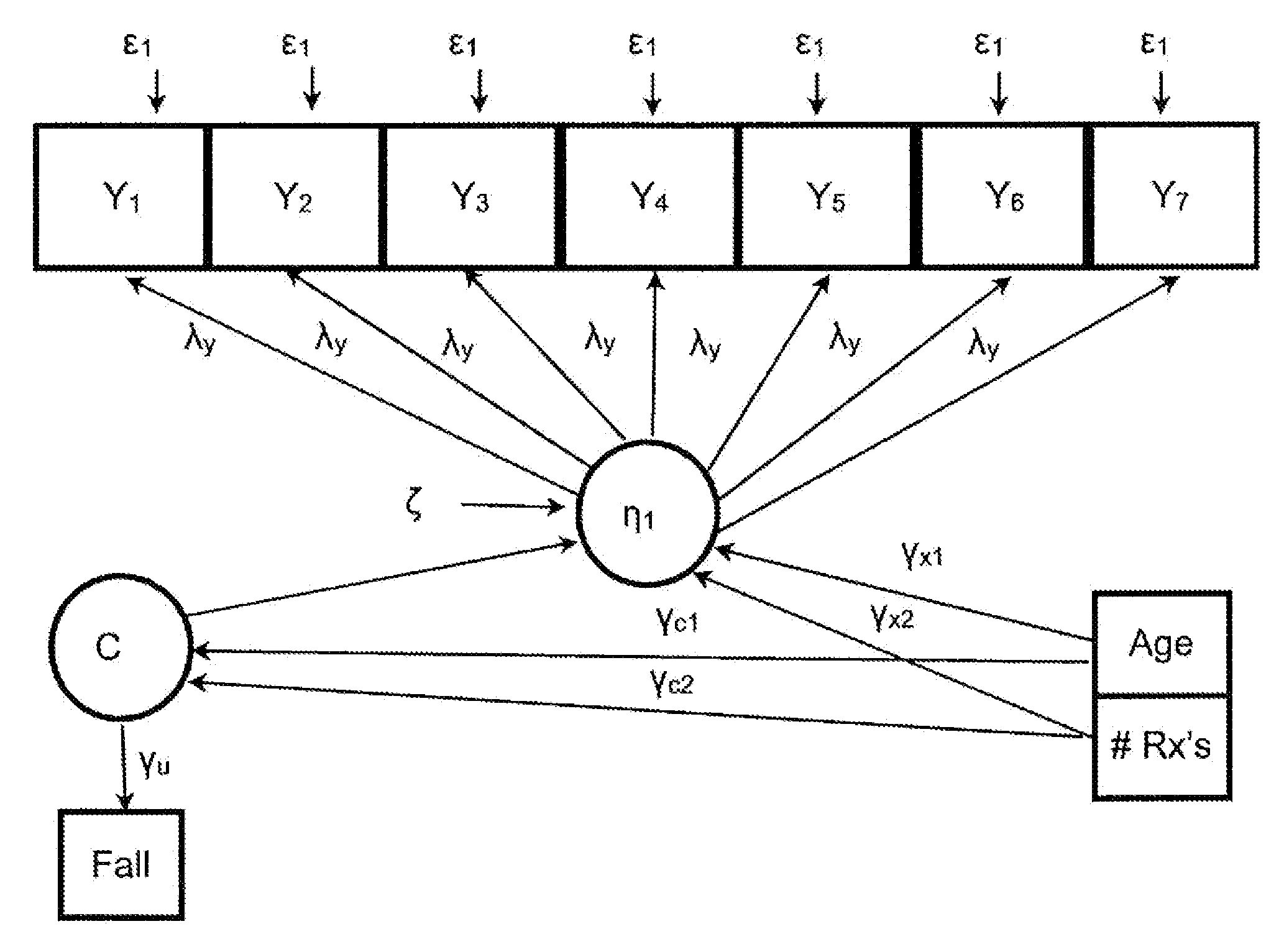
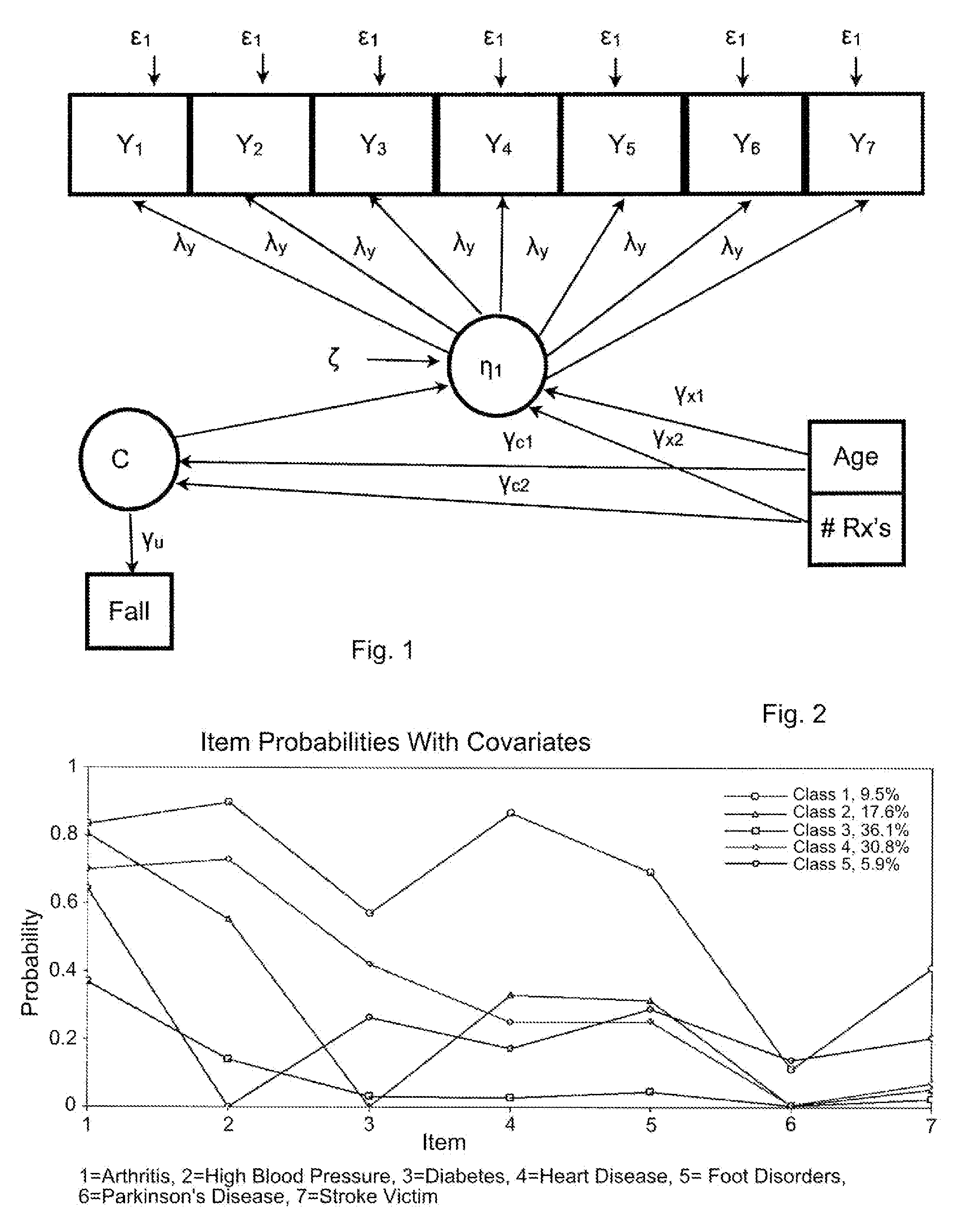
Statistical model for predicting falling in humans
Dependent variables believed to contribute to a likelihood of falling are analyzed using a latent class analysis. The dependent variables are biomedical factors, which may include, for example, arthritis, high blood pressure, diabetes, foot disorders, Parkinson's Disease, stroke, eye disorder, limb disorder, or proprioceptive disorder. Data pertaining to the biomedical factors is gathered from a population of individuals at risk of falling. Covariate data, including for example age and the number of prescriptions taken, is further analyzed against latent class data. For a particular group of at risk individuals, a set of five classes produced useful results broadly corresponding to groups representing individuals who have: good health; a range of diseases; Parkinson's Disease; arthritis; and high blood pressure. A probability of falling is determined, relative to the group of individuals with good health.
Owner:NOVA SOUTHEASTERN UNIVERSITY
Heterocyclic compounds and their uses
Substituted bicyclic heteroaryls and compositions containing them, for the treatment of general inflammation, arthritis, rheumatic diseases, osteoarthritis, inflammatory bowel disorders, inflammatory eye disorders, inflammatory or unstable bladder disorders, psoriasis, skin complaints with inflammatory components, chronic inflammatory conditions, including but not restricted to autoimmune diseases such as systemic lupus erythematosis (SLE), myestenia gravis, rheumatoid arthritis, acute disseminated encephalomyelitis, idiopathic thrombocytopenic purpura, multiples sclerosis, Sjoegren's syndrome and autoimmune hemolytic anemia, allergic conditions including all forms of hypersensitivity. The present invention also enables methods for treating cancers that are mediated, dependent on or associated with pi 105 activity, including but not restricted to leukemias, such as Acute Myeloid leukaemia (AML) Myelo-dysplastic syndrome (MDS) myelo-proliferative diseases (MPD) Chronic Myeloid Leukemia (CML) T-cell Acute Lymphoblastic leukaemia (T-ALL) B-cell Acute Lymphoblastic leukaemia (B-ALL) Non Hodgkins Lymphoma (NHL) B-cell lymphoma and solid tumors, such as breast cancer.
Owner:AMGEN INC