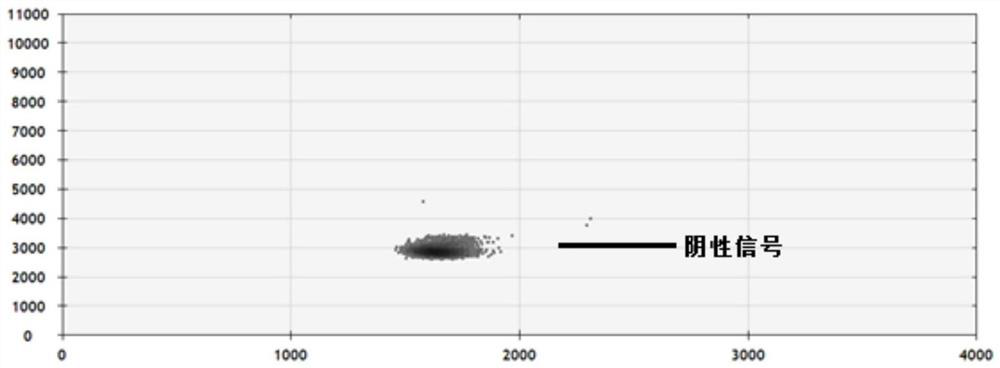
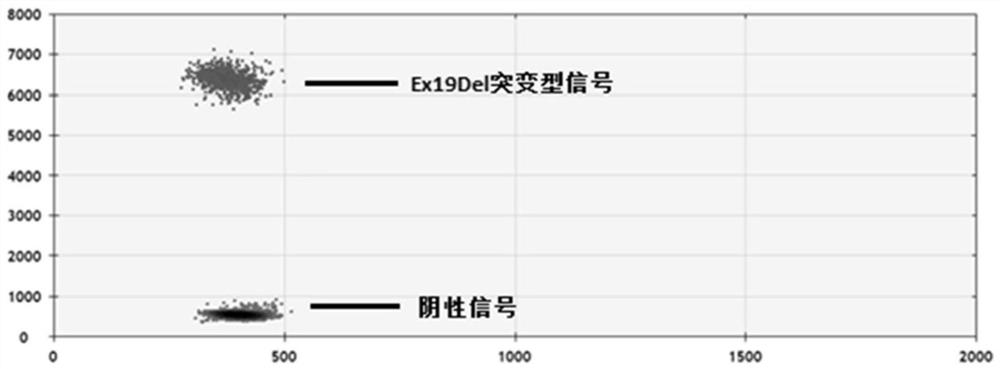
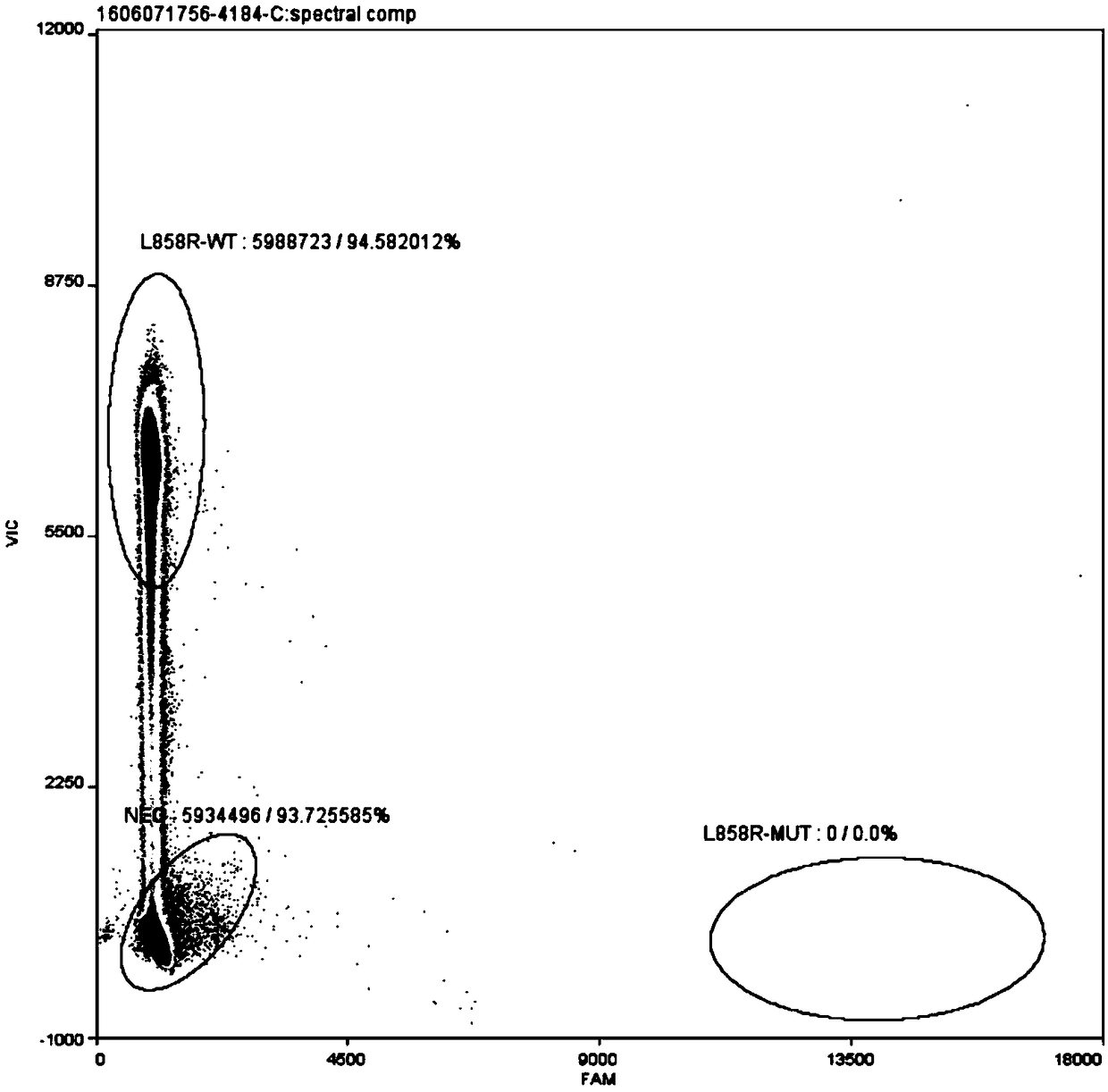
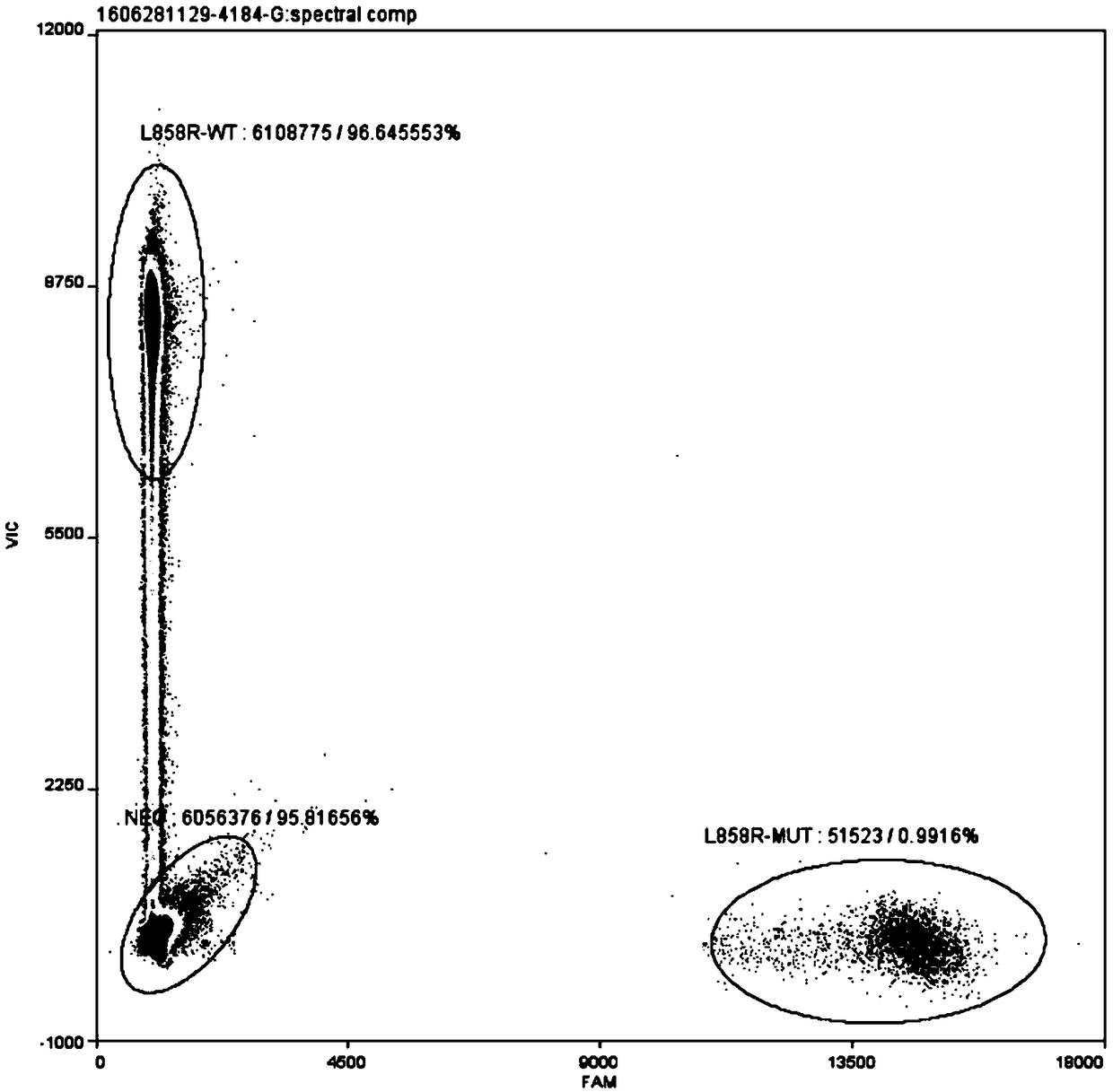
Patents
Literature
30 results about "L858r mutation" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
About 40-45% of the sensitizing mutations are point mutations in exon 21, the most common being L858R (At the “point” in the 858th position, the normal amino acid leucine (L) is switched out for an arginine (R), which changes the protein function).
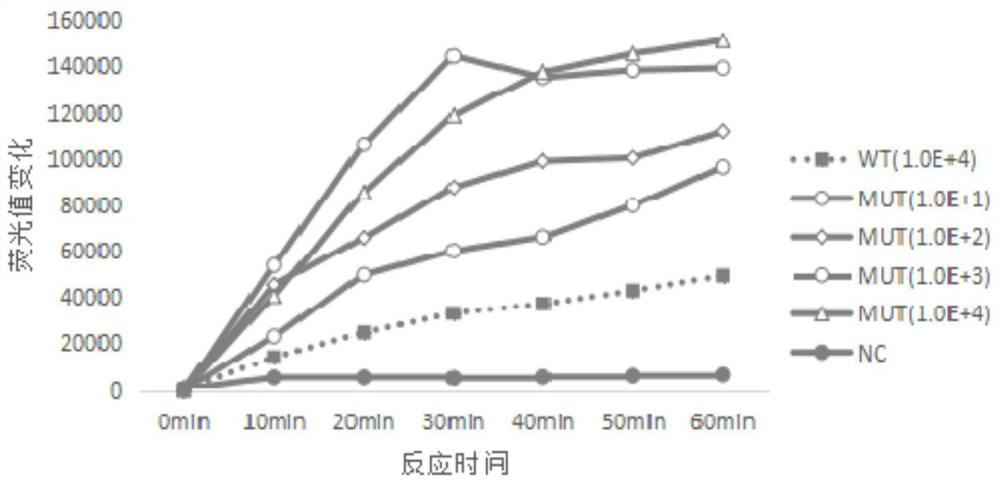
Kit, reaction system and method for detecting human EGFR gene mutation
InactiveCN106434941AGood choiceHigh sensitivityMicrobiological testing/measurementFluorescenceEGFR Gene Mutation
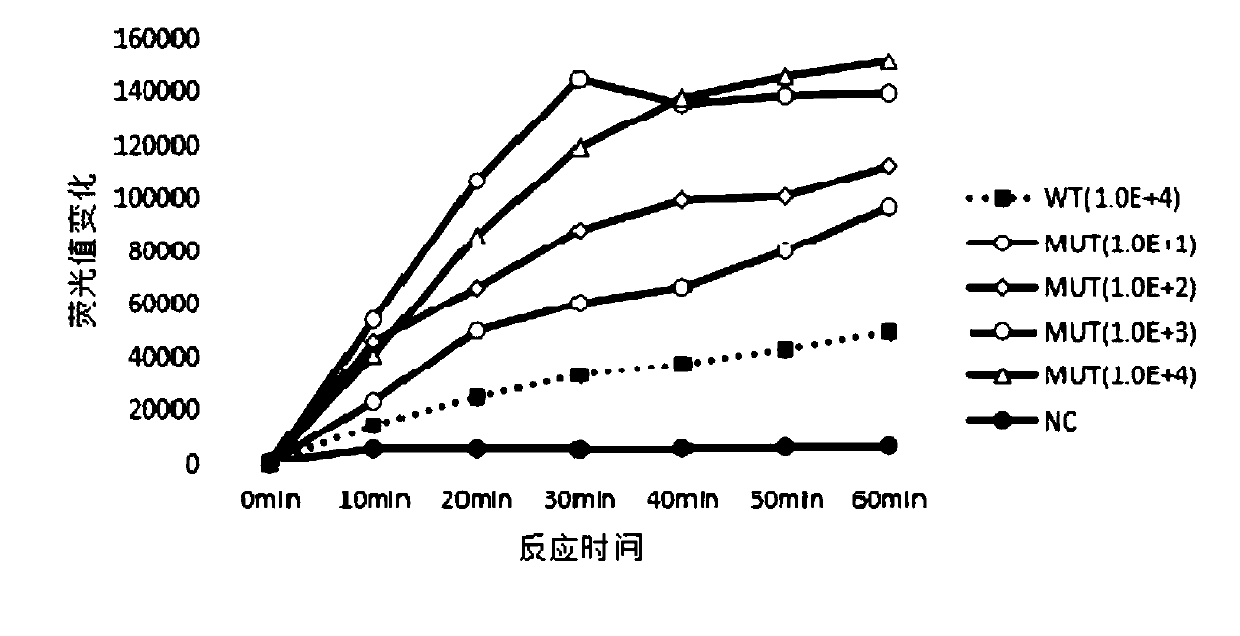
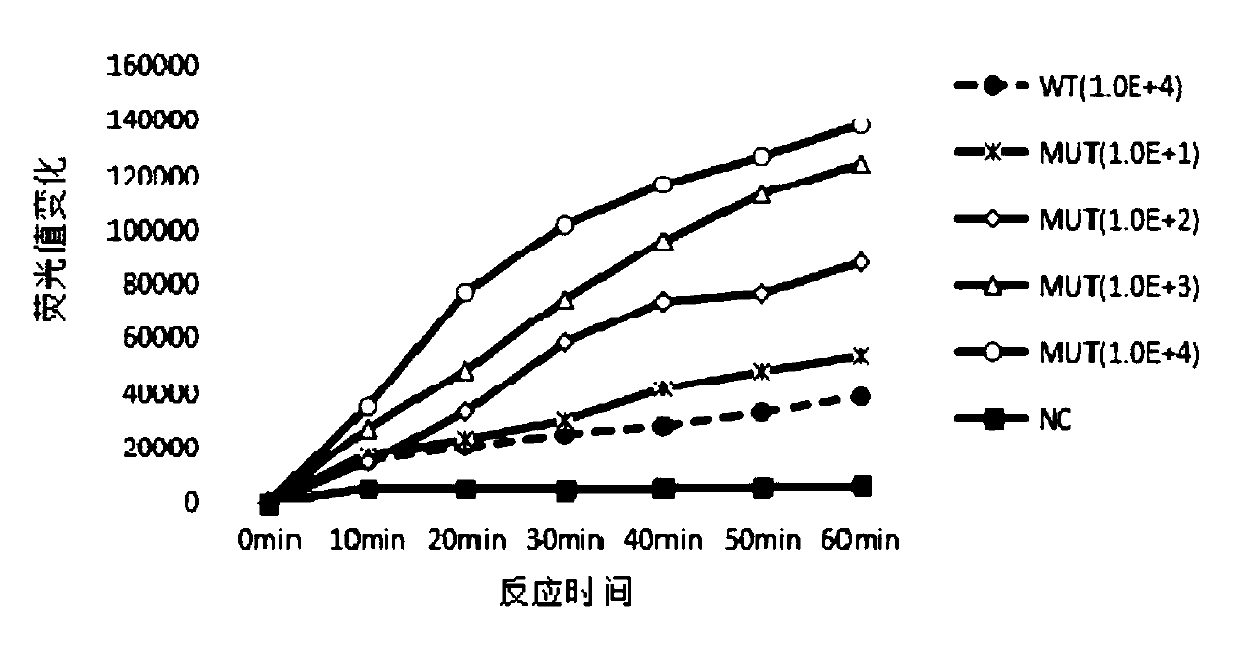
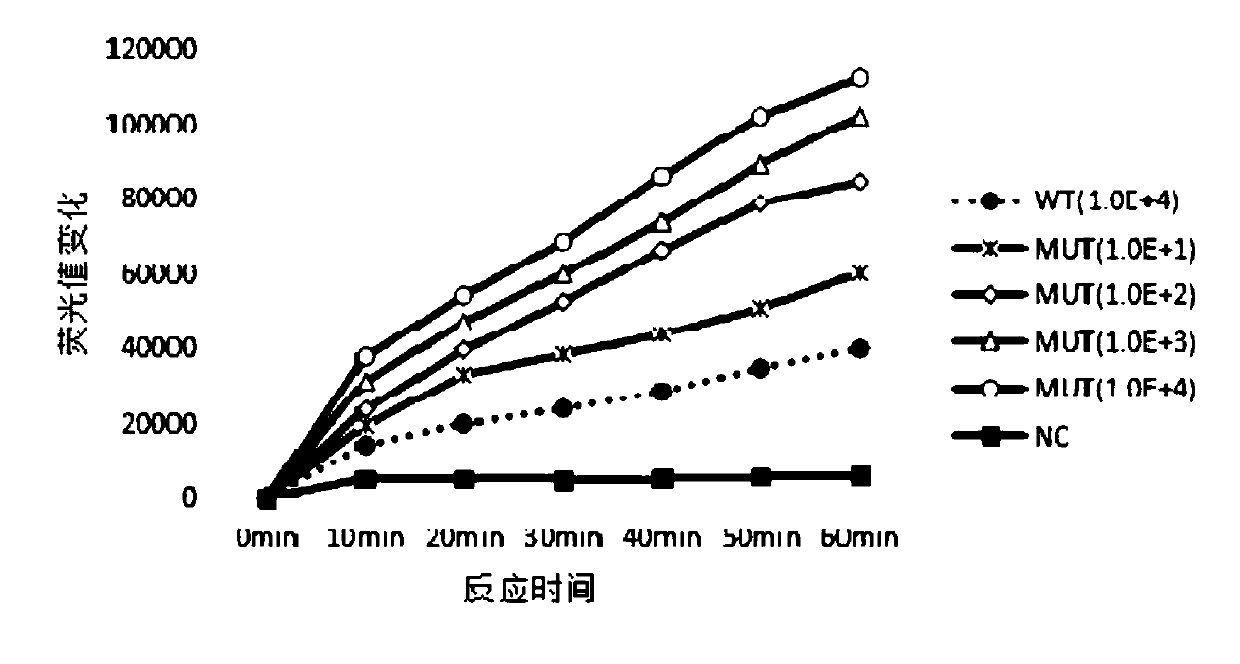
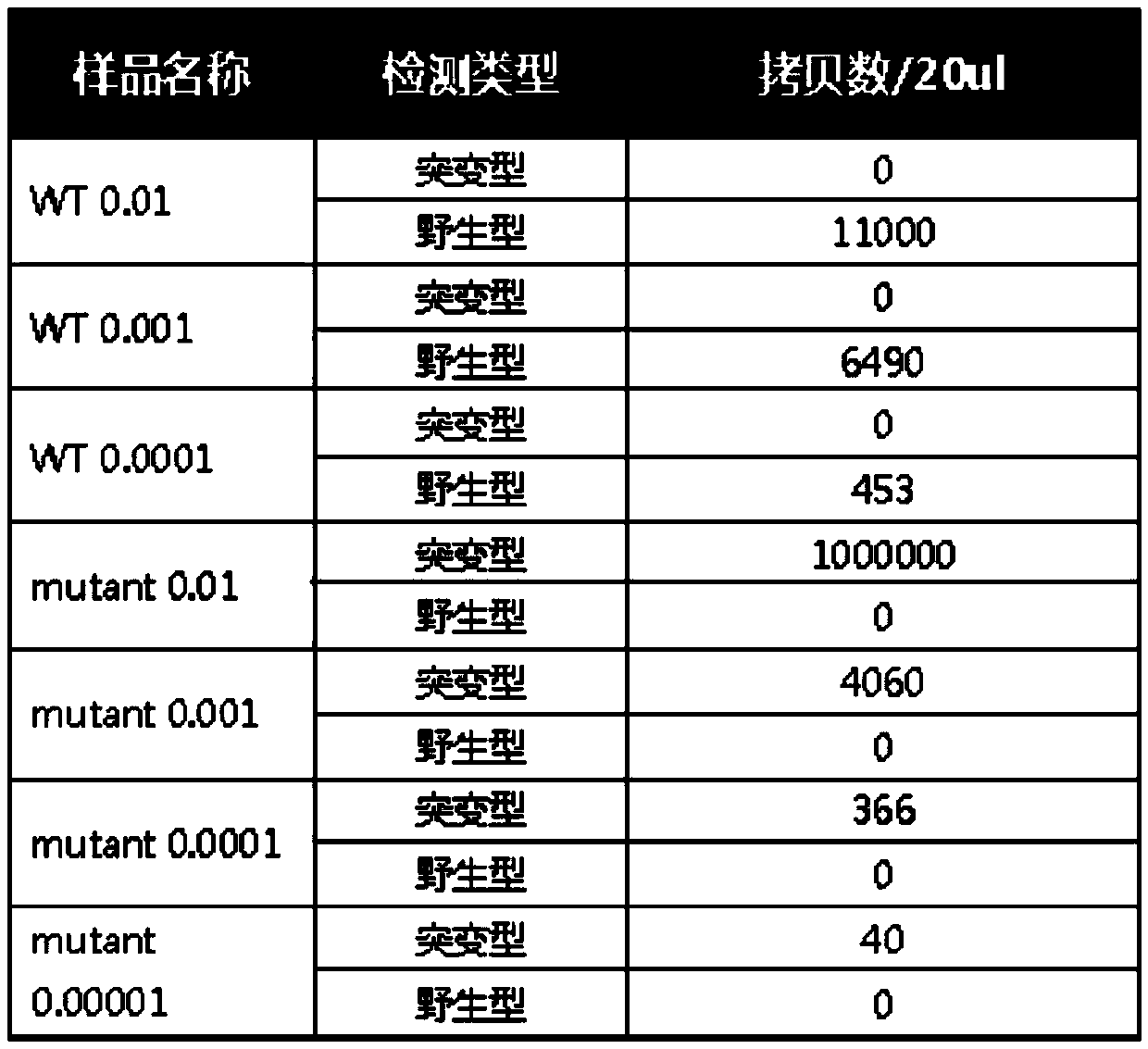
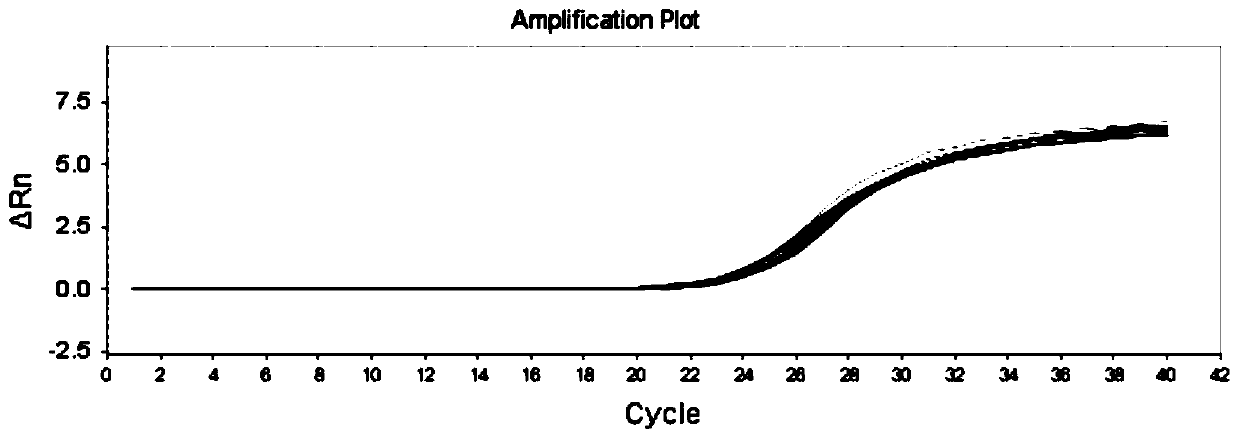
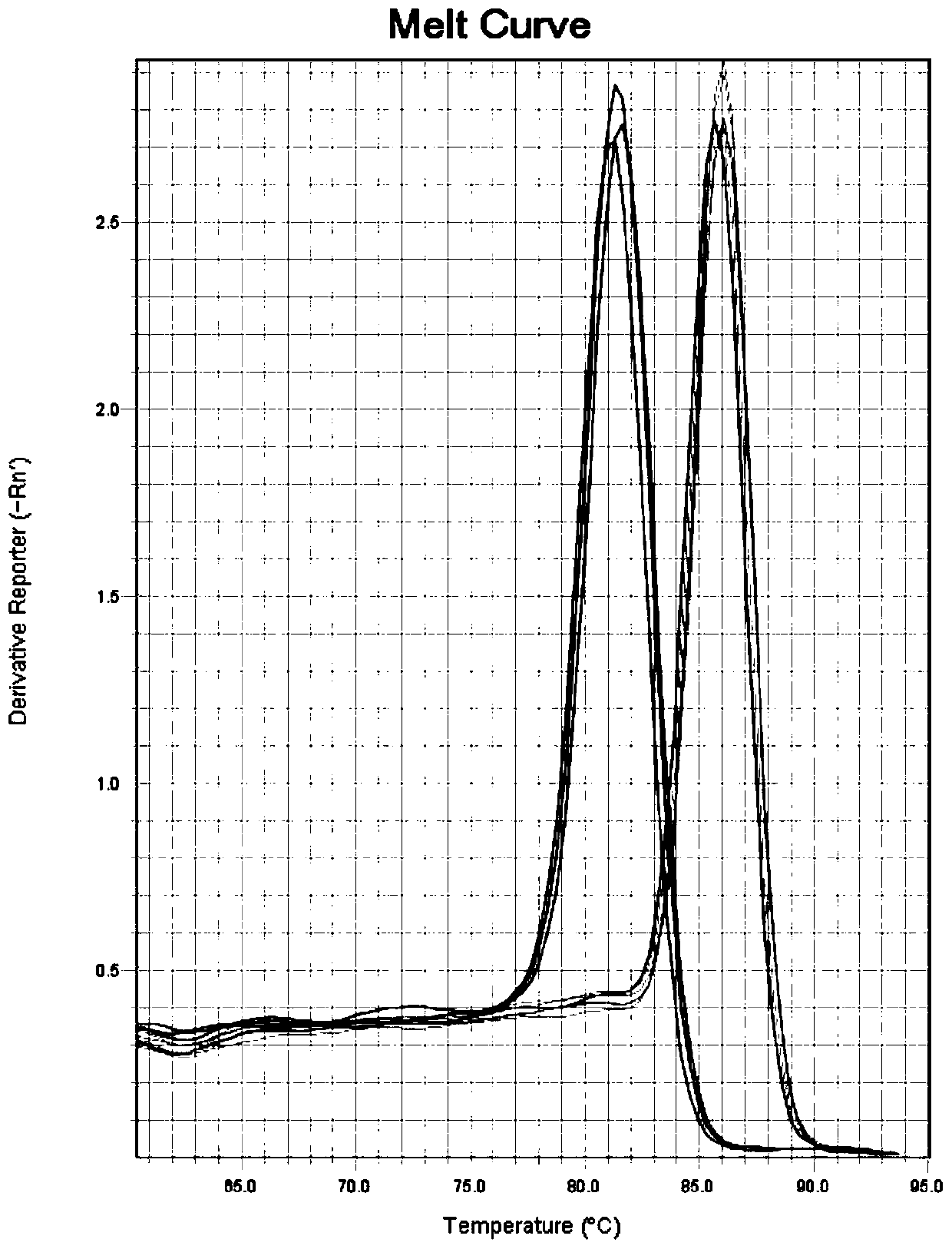
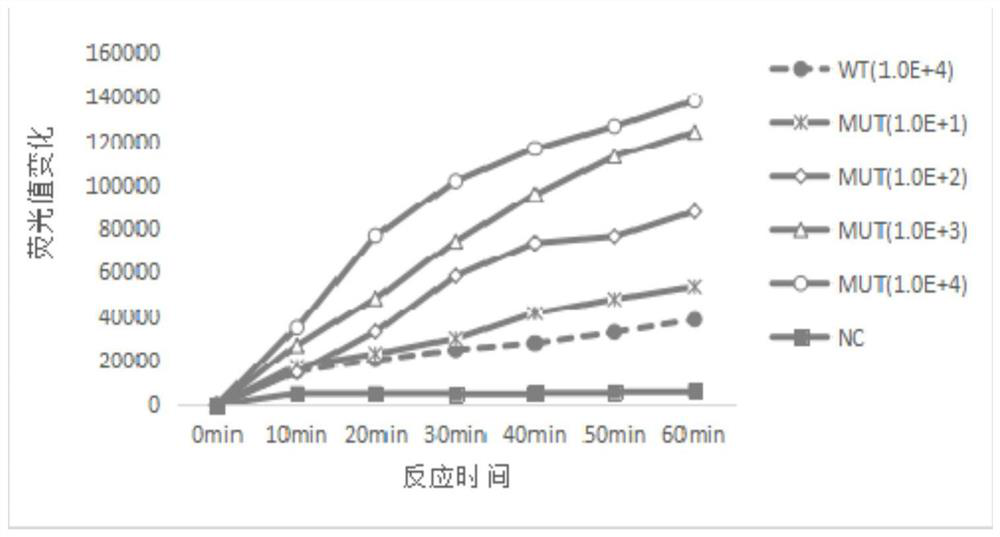
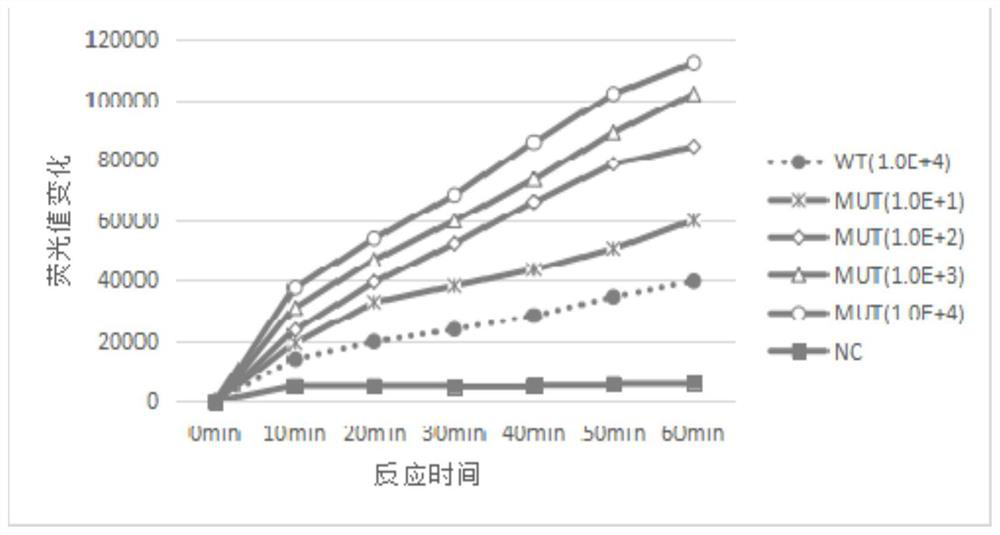
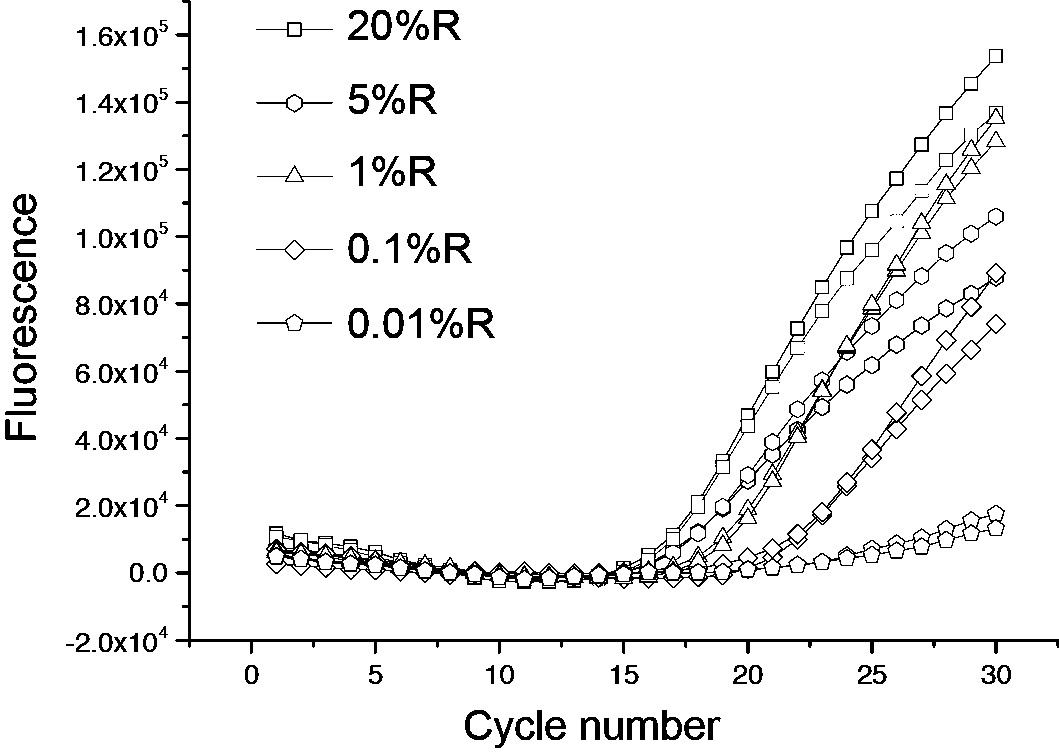
The invention discloses a kit, reaction system and method for detecting human EGFR gene mutation. The kit comprises an upstream primer, a downstream primer, a fluorescent probe for detecting an L858R mutation and a fluorescent probe for detecting wild types. The primers with specific sequences and the probes with the specific sequences are designed in the kit according to the L858R mutation of an EGFR gene. In an optimized reaction system, high-sensitivity detection of the L858R mutation of the EGFR gene is achieved through a digital PCR platform. Besides, negativity and positivity of the L858R mutation are detected, meanwhile the proportion of the mutation in a sample can be obtained, sources of samples capable of being detected in the kit are further diversified, the samples can be tumor tissue and ctDNAs, and the application range of the kit, the reaction system and the method is widened.
Owner:GENETRON HEALTH (BEIJING) CO LTD
Methods and compositions to detect mutations in plasma using exosomal RNA and cell free DNA from non-small cell lung cancer patients
ActiveUS20190376128A1Stimulate immune responseHigh sensitivityMicrobiological testing/measurementDiseaseCell free
The present invention relates generally to methods and kits for detecting one or more biomarkers, such as an Epidermal Growth Factor Receptor (EGFR) mutation, e.g., T790M mutation, L858R mutation, one or more exon19 insertions and / or one or more exon19 deletions in the EGFR gene, in a biological sample to aid in diagnosis, prognosis, monitoring, or therapy selection for a disease such as, for example, cancer. The methods and kits are useful in aiding in diagnosis, prognosis, monitoring, or therapy selection for lung cancer, e.g., non-small cell lung cancer (NSCLC).
Owner:EXOSOME DIAGNOSTICS
Standard product for detecting genes of clinical medications for lung cancer and application thereof
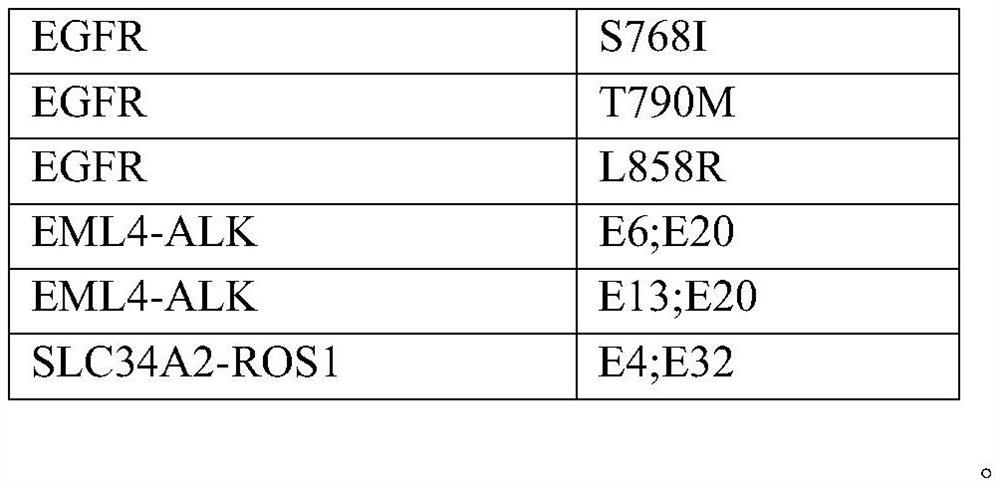
ActiveCN107345253AVerify accuracyAccurate guidanceMicrobiological testing/measurementDNA/RNA fragmentationReference productROS1
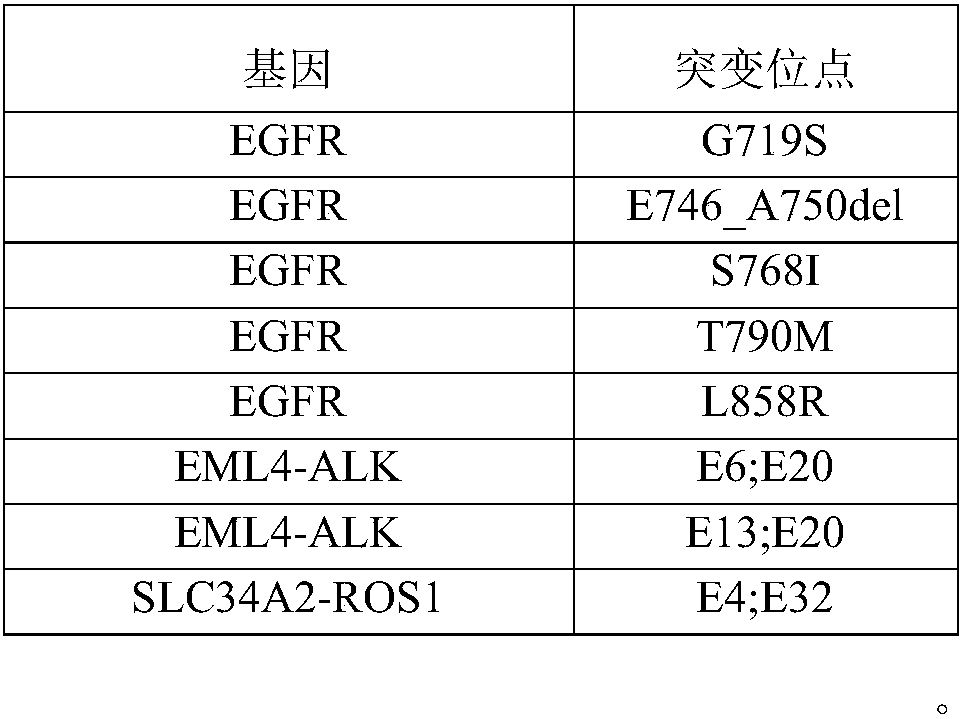
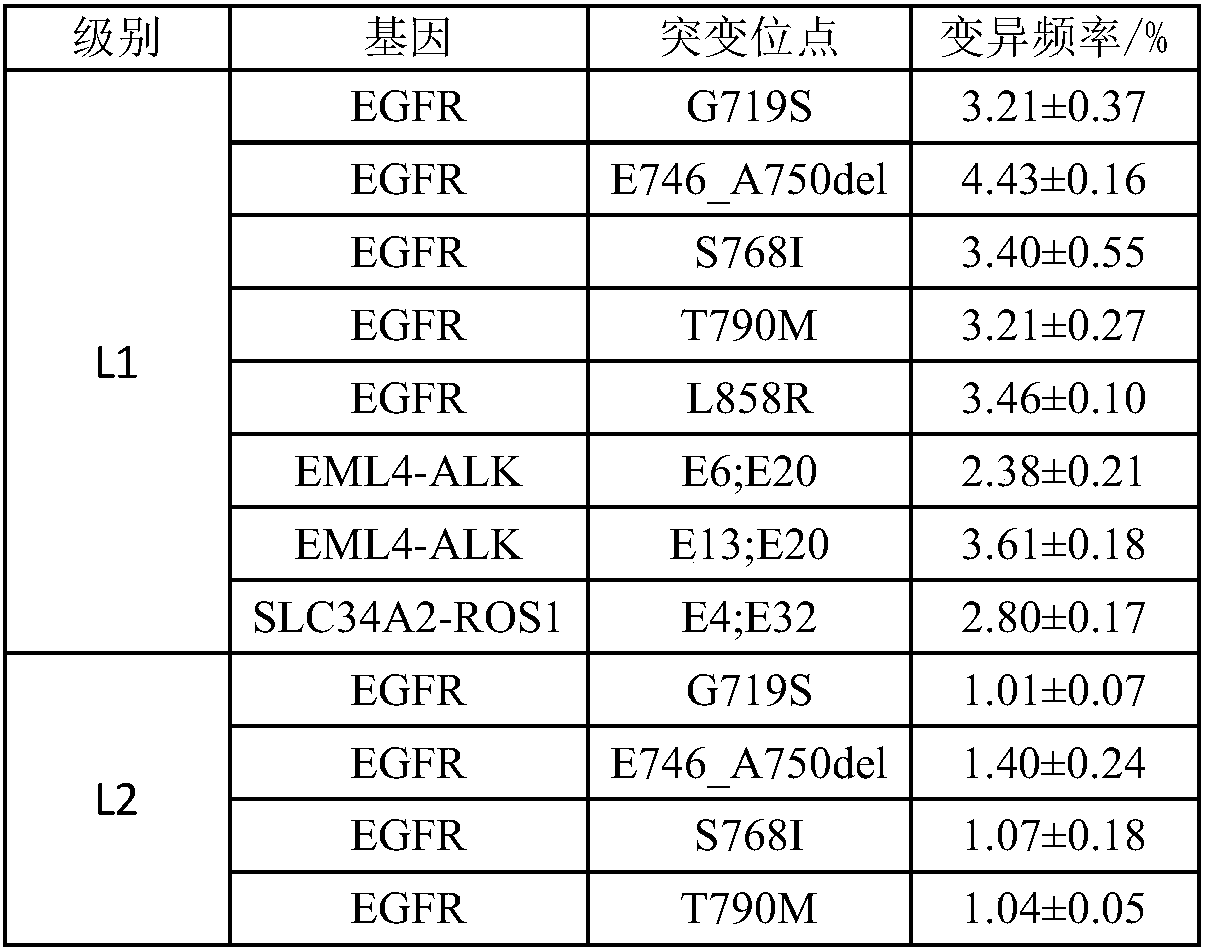
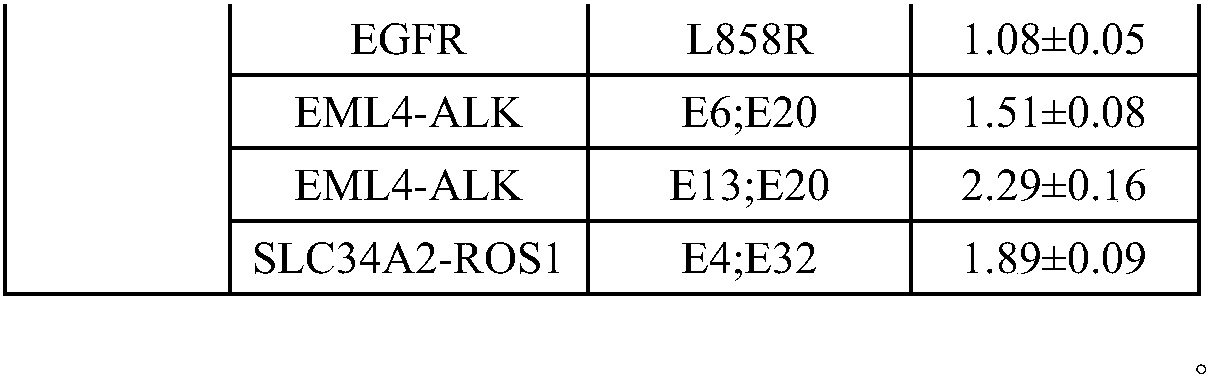
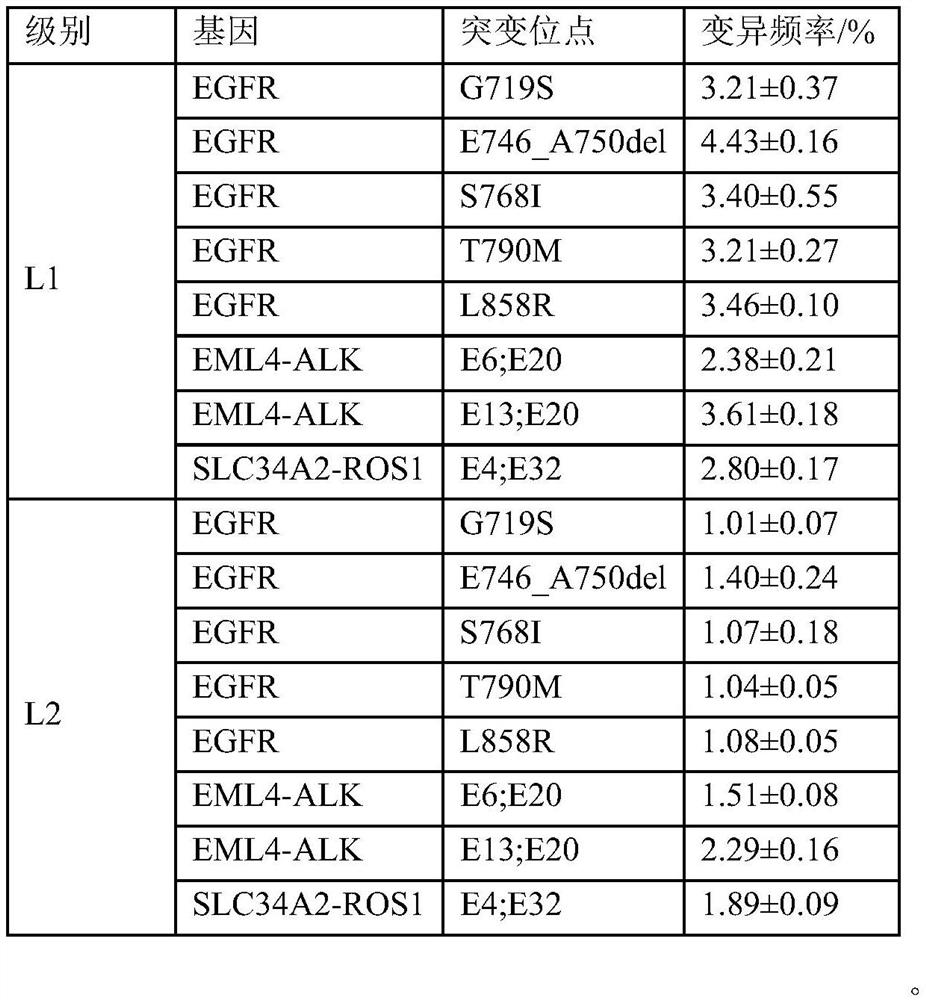
The invention discloses a standard product for detecting genes of clinical medications for lung cancer and an application thereof, wherein the standard product comprises a lowest-detection-limit reference product at least comprising two different levels of variation-frequency DNA mixtures; and each level of variation-frequency DNA mixture comprises the following mutation sites: EGFR gene G719S, E746_A750del, S768I, T790M and L858R mutation sites, EML4-ALK gene E6, E20 and E13, E20 mutation site, SLC34A2-ROS1 gene E4, and E32 mutation site. By application of the technical scheme disclosed by the invention, whether a kit can detect the gene mutation and the gene frequency can be judged according to results, the accuracy of detection results of the existing kit can be verified and more accurate guidance also can be provided for clinical medications.
Owner:GENE CRAB BIOTECH CO +1
EGFR inhibitor for targetedly treating cancer and preparation method and application thereof
ActiveCN105777716ALittle side effectsOrganic chemistryAntineoplastic agentsSide effectDouble mutation
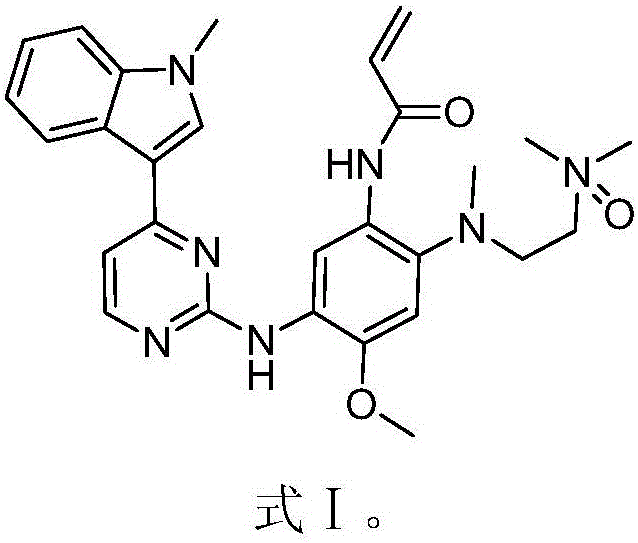
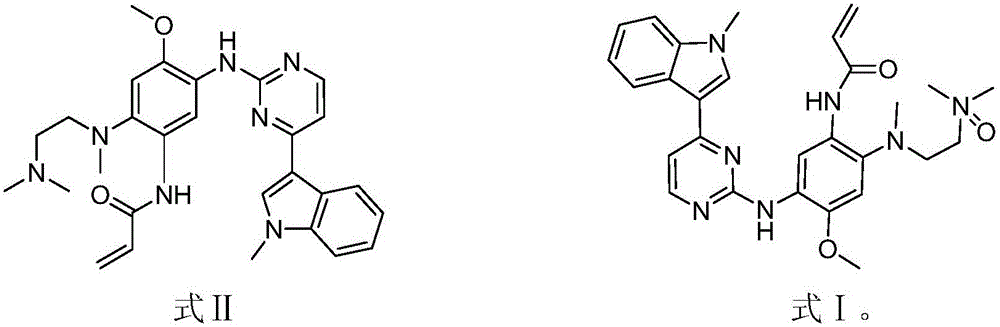
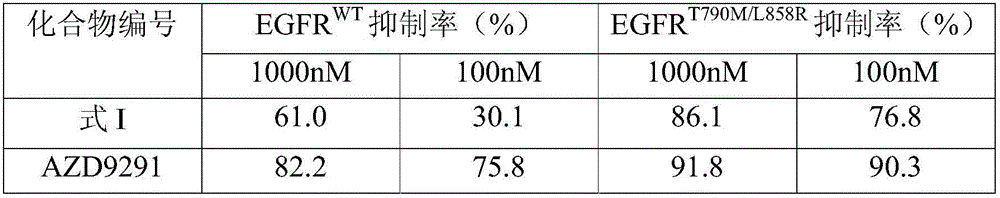
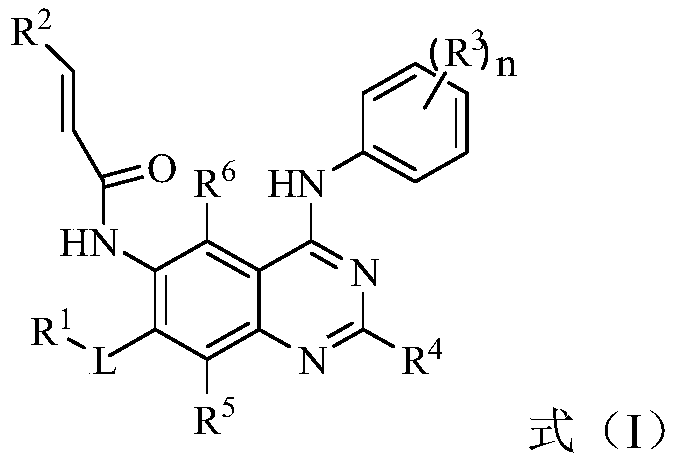
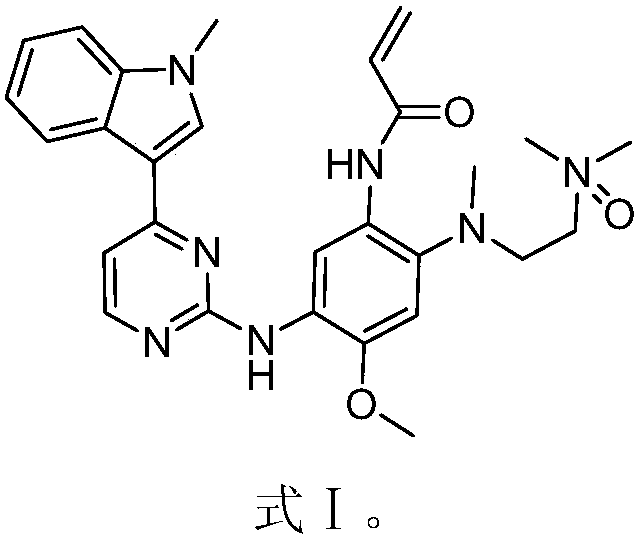
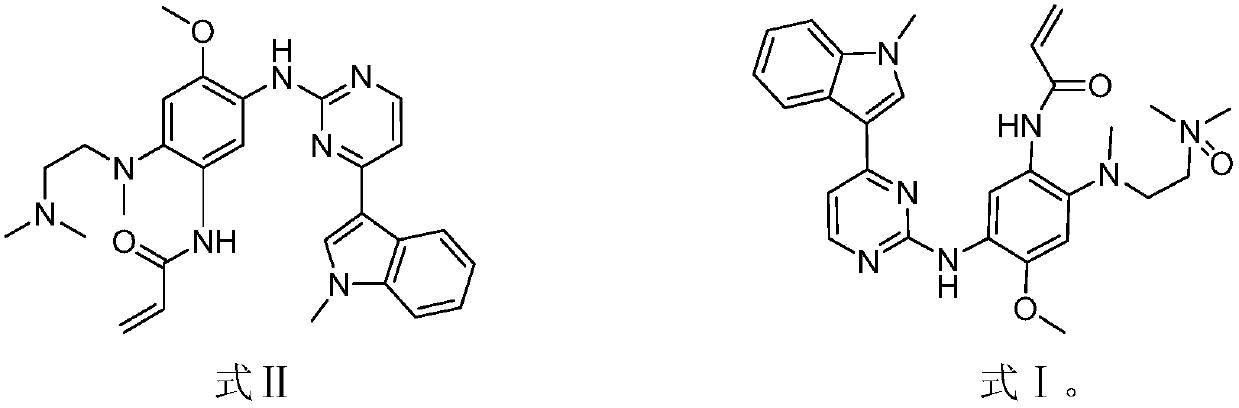
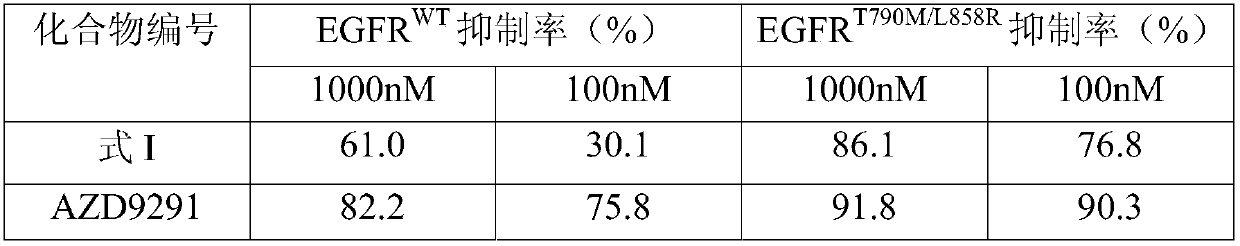
The invention discloses an EGFR inhibitor for targetedly treating a cancer and a preparation method and application thereof.The structural formula of the EGFR inhibitor is shown in the formula I.The EGFR inhibitor can be used for preventing and / or treating the cancer such as a human skin caner or a lung cancer.The EGFR inhibitor can selectively inhibit cell lines of EGFR double mutation (EGFRT790M and L858R), and the inhibitory activity to EGFR wild type cells is weak; therefore, the EGFR inhibitor can be used for treating lung cancer patients with EGFRT790M and L858R mutation, and the side effects are fewer, wherein the side effects are caused by inhibiting wild type EGFR, for example, Afatinib.
Owner:TSINGHUA UNIV
EGFR (epidermal growth factor receptor) inhibitor for targeted therapy of cancers, and preparation method and application thereof
ActiveCN105585557ANovel chemical structureLittle side effectsOrganic active ingredientsOrganic chemistryChemical structureSide effect
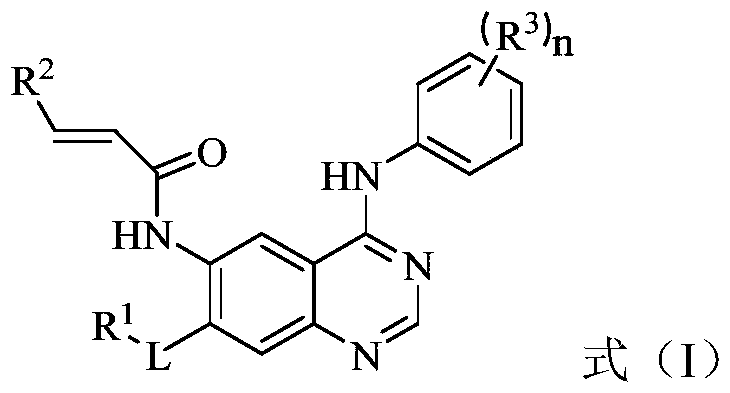
The invention discloses an EGFR (epidermal growth factor receptor) inhibitor for targeted therapy of cancers, and a preparation method and application thereof. The structural formula of the EGFR inhibitor is disclosed as Formula I, wherein R is H, OH, NR', C1-C3 alkyl, C1-C3 alkenyl, aryl or heterocycle, and R' is C1-C3 alkyl. The EGFR inhibitor can be used for preventing and / or treating cancers, such as human skin squamous carcinoma or lung cancer. Compared with the existing inhibitors (such as AZD9291, afatinib and the like), the EGFR inhibitor disclosed by the invention has novel chemical structure. The EGFR inhibitor can selectively inhibit cell lines of EGFR double mutants (EGFRT790M / L858R), and has lower inhibition activities for EGFR wild type cells. Therefore, the EGFR inhibitor disclosed by the invention can be used for treating the patient with lung cancer with EGFRT790M / L858R mutants, and has lower side effect (caused by the inhibition of the wild type EGFR, such as afatinib).
Owner:TSINGHUA UNIV
Method for detecting EGFR L858R locus in lung cancer by ddPCR and application thereof
InactiveCN109762878ALow nucleic acid contentLow Mutation Sequence ContentMicrobiological testing/measurementWild typeBlood plasma
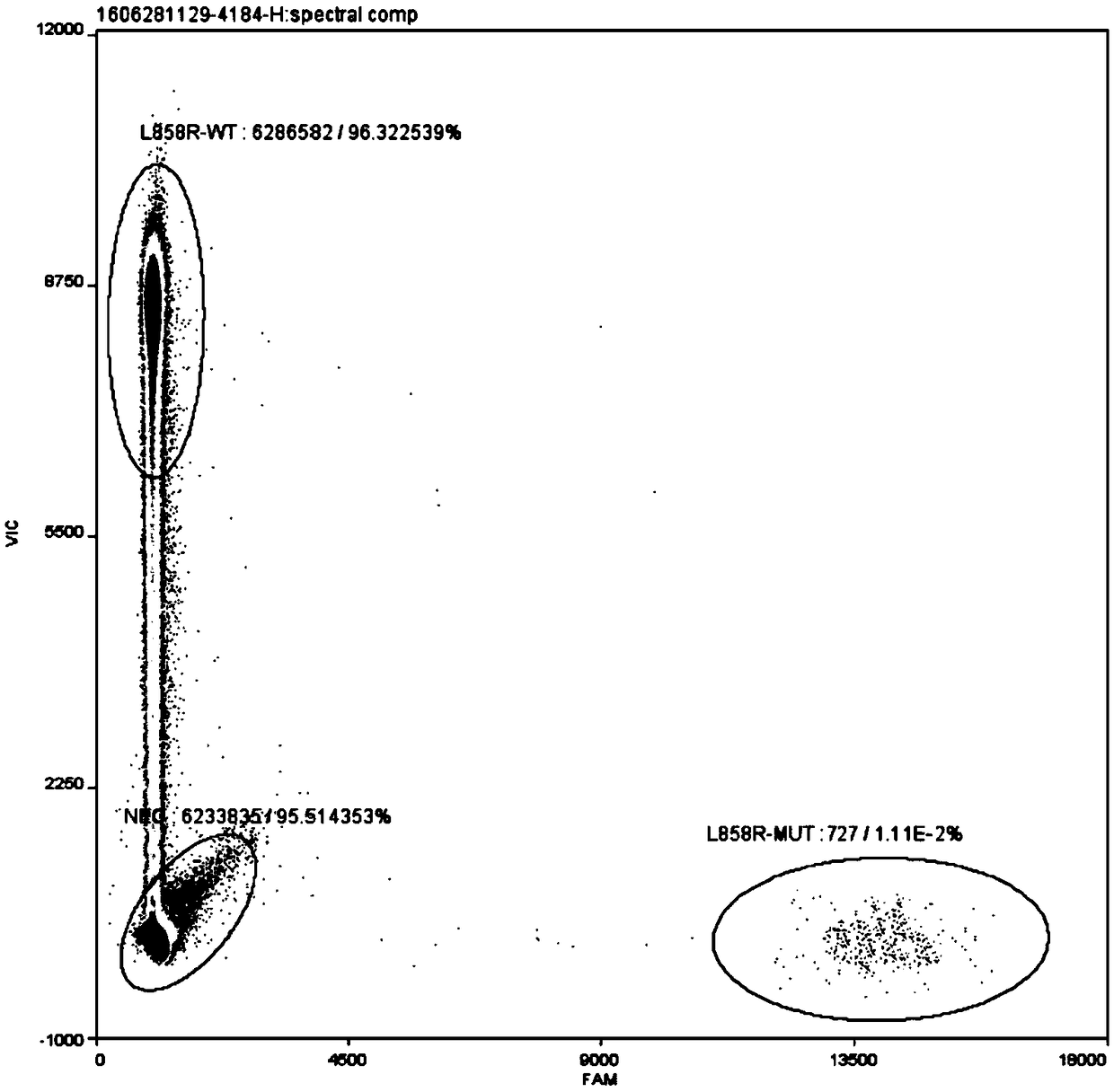
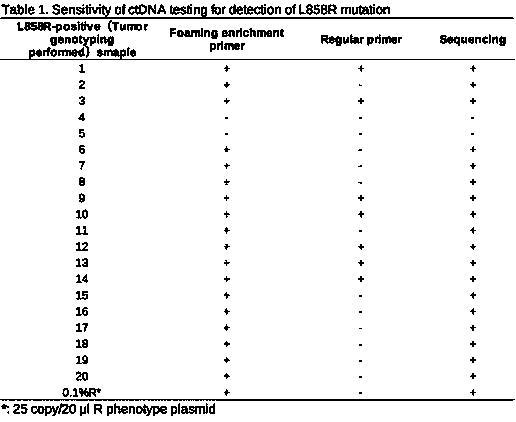
The invention discloses a method for detecting an EGFR L858R locus in lung cancer by ddPCR and an application thereof. According to the sequence of an EGFR gene, a pair of amplified primers includingan upstream primer and a downstream primer are designed; according to the mutation sequence of L858R (2573T>G, COSM6224), a probe for detecting a wild type sequence and a probe for detecting a mutation sequence are designed. The primers and the probes are used for ddPCR and used for detecting and analyzing important loci related to targeted medication for lung cancer in ctDNA. Firstly, DNA is extracted from peripheral plasma of lung cancer patients, and the mutation condition of the mutation locus L858R of EGFR is detected by the ddPCR technology, so as to guide the patients to use drugs. Themethod can be used for diagnosis of early-stage lung cancer such as stage I, II and III A of tumor, can detect 0.0625% EGFR gene L858R mutation, and has the sensitivity reaching 10 times or more thatof conventional methods.
Owner:铭时医疗科技(宁波)有限公司
EGFR (Epidermal Growth Factor Receptor)/L858R mutant liquid biopsy kit and application thereof
InactiveCN106755551AAmplification arrestThe detection process is fastMicrobiological testing/measurementWild typeTherapeutic effect
The invention provides an EGFR gene L858R mutant liquid biopsy kit and application thereof. The EGFR gene L858R mutant liquid biopsy quantitative detection kit is characterized by comprising an EGFR gene upstream primer, an EGFR mutant gene downstream enrichment primer (a wild blocking primer), an EGFR wild gene downstream enrichment primer (a mutant blocking primer), a qPCR reaction system reagent and mutant and wild type EGFR positive plasmids. The kit disclosed by the invention can be used for relative quantification of mutant target genes of minimally invasive liquid biopsy of cancer patients and achieves important clinical effects on early screening, auxiliary diagnosis, recurrence monitoring and therapeutic effect evaluation of tumors. In addition, because use of a probe is not involved in the invention, the kit is low in cost and can be popularized on a large scale, and the liquid biopsy cost is reduced.
Owner:陈超
Detection kit and detection method for gene mutation of lung cancer-related molecular markers
ActiveCN110904239ALow costSimple methodMicrobiological testing/measurementDNA/RNA fragmentationGenes mutationBRAF Gene Mutation
The invention provides crRNAs for gene mutation of lung cancer-related molecular markers, a detection kit and a detection method thereof. The crRNAs comprise: a crRNA at the EGFR-T790M mutation site:as shown in any one of SEQ ID NO.16-17; a crRNA at the EGFR-L858R mutation site: as shown in any one of SEQ ID NO.18-19; a crRNA at the BRAF gene mutation site: as shown in any one of SEQ ID NO.20-21;a crRNA at the ALK gene mutation site: as shown in any one of SEQ ID NO.22-23; and a crRNA at the ROS1 gene mutation site: as shown in any one of SEQ ID NO.24-25. According to the invention, the 5 crRRNAs related to gene mutation targets of lung cancer-related molecular markers are designed, and the crRNAs are adopted in combination with a CRISPR-cpf1 system to carry out mutation detection. The method of the invention has the characteristics of simplicity, fast speed, high sensitivity and strong specificity.
Owner:江苏博嘉生物医学科技有限公司
Specific primer, probe and kit for detecting EGFR gene L858R locus
InactiveCN105505924AAccurate detectionHigh sensitivityMicrobiological testing/measurementDNA/RNA fragmentationMutation frequencyNucleotide sequencing
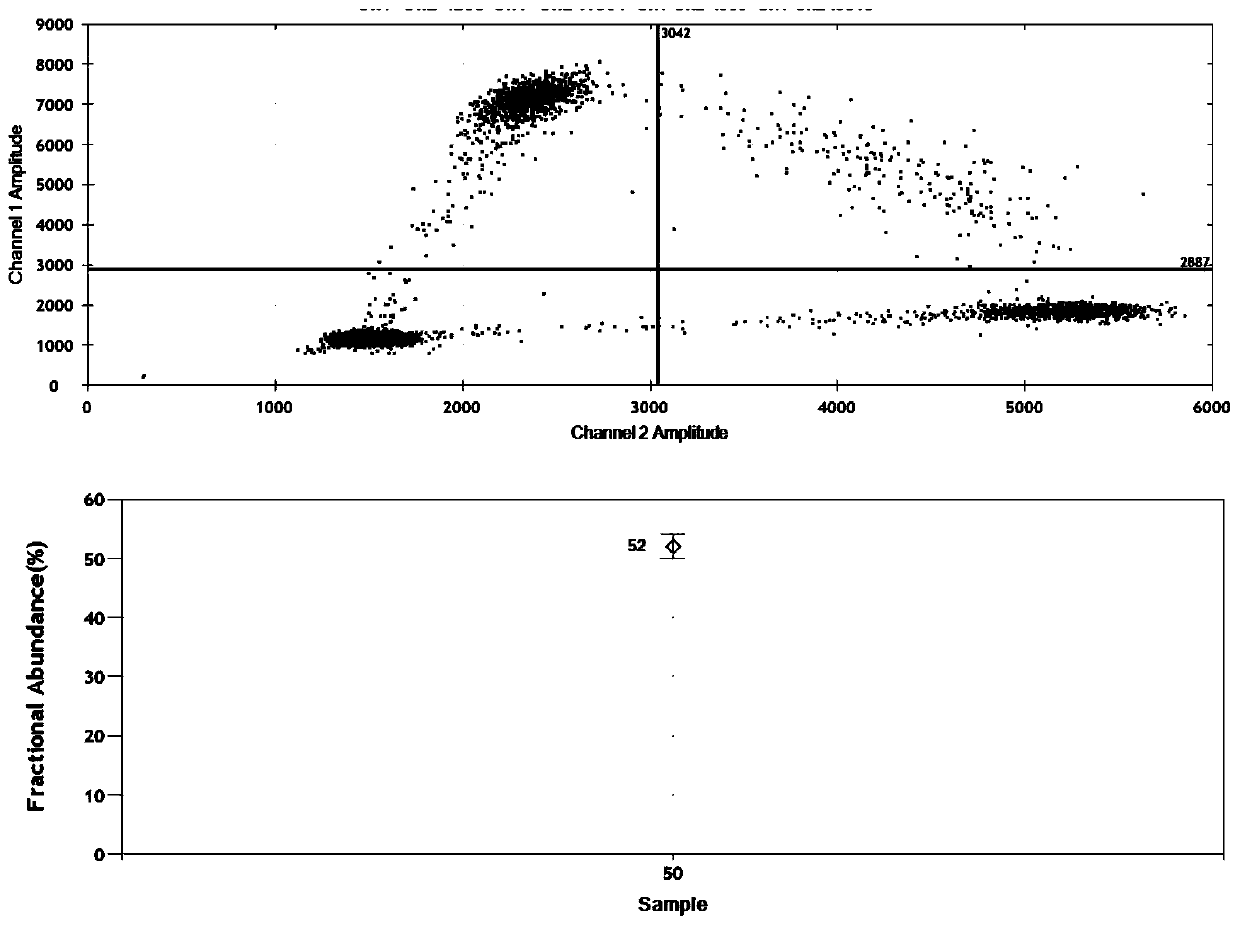
The invention relates to molecular detection, and particularly discloses a specific primer, probe and kit for detecting an EGFR gene L858R locus. A nucleotide sequence of the specific primer is shown as SEQ ID NO. 1 to 2, and a nucleotide sequence of the probe is shown as SEQ ID NO. 3 to 4. The specific primer and the MGB-Taqman probe provided by the invention adopt a probe method to detect the EGFR gene L858R, and are not only suitable for detecting nucleic acid from a tumor tissue and a paraffin-embedded tissue, but also suitable for detecting a mutant locus of the EGFR gene L858R in free circulating nucleic acid of blood; and moreover, by combining a detection system and a micro-drop digital PCR system, the mutation frequency of a nucleic acid sample can be accurately detected.
Owner:BEIJING CENT FOR PHYSICAL & CHEM ANALYSIS
Primer and probe used for detecting EGFR TK1 sensitive mutant
InactiveCN108504725AReduce detection pointsReduce testing costsMicrobiological testing/measurementDNA/RNA fragmentationNucleotideMicrobiology
The invention discloses a primer and a probe used for detecting EGFR TK1 sensitive mutant. The nucleotide sequence of the primer disclosed by the invention is as shown in SEQ ID NO:1-13 and SEQ ID NO:16-17; and the nucleotide sequence of the probe disclosed by the invention is as shown in SEQ ID NO:14-15 and SEQ ID NO:18. The primer and the probe disclosed by the invention have the benefits that in detection, the mutation type with very low mutation rate is deleted, thus reducing total time required for experiment and reducing experiment cost under the condition of ensuring other ARMSPCR technical advantages; and the design locus still covers more than 90% of the EGFR TK1 sensitive mutant. In addition, relatively good detection efficiency can be obtained by brand-new designed detection primer and probe for 19 exon deletion mutation and L858R mutation.
Owner:SHANGHAI PERMED BIOMEDICAL CO LTD
Method and reagent kit for detecting human EGFR gene T790M and L858R mutations
PendingCN109852676AImprove scalabilityEasy to detectMicrobiological testing/measurementDNA/RNA fragmentationDouble mutationWilms' tumor
The invention provides a primer and probe combination for a digital PCR detection system for double mutations of human EGFR gene T790M and L858R, the digital PCR detection system for the double mutations of the human EGFR gene T790M and L858R, a digital PCR detection reagent kit for the double mutations of the human EGFR gene T790M and L858R, and a detection method of the double mutations of the human EGFR gene T790M and L858R. High sensitivity detection of the T790M and L858R is realized by the method of the digital PCR platform in the dual system, and so the cost is reduced and the consumption of samples is reduced, especially for ctDNA with scarce samples. Moreover, the detection of other sample types such as tumor tissues is facilitated, and certain medication guidance is provided forpatients with non-small cell lung cancer.
Owner:DALIAN GENTALKER BIO-TECH CO LTD
Specific primer, probe and kit for detecting EGFR gene L858R mutation based on digital PCR technology
PendingCN110066873AImprove featuresConvenient and efficientMicrobiological testing/measurementDNA/RNA fragmentationNucleotideNucleotide sequencing
The invention provides a specific primer, probe and kit for detecting an EGFR gene L858R mutation based on a digital PCR technology, wherein the specific primer used for detecting the EGFR gene L858Rmutation based on the digital PCR technology comprises an upstream primer and a downstream primer, the nucleotide sequence of the upstream primer is shown as SEQ ID No.1-4, and the nucleotide sequenceof the downstream primer is shown as SEQ ID No.5-7. The specific primer of the EGFR gene L858R mutation has good amplification specificity and amplification efficiency; meanwhile, the kit for detecting the EGFR gene L858R mutation based on the digital PCR technology can carry out absolute quantification on the L858R mutation frequency, and has high specificity and higher detection sensitivity. The method is not only suitable for detecting DNA derived from pathological tissues or sections, but also suitable for detecting the EGFR gene L858R mutation in blood free DNA.
Owner:DELUTONG SHIJIAZHUANG BIOTECH CO LTD
Primer probe and kit for detecting EGFR (epidermal growth factor receptor) gene L858R mutation and application of primer probe and kit
ActiveCN113355423ASimple and fast operationShort detection timeMicrobiological testing/measurementDNA/RNA fragmentationMedicineMutation detection
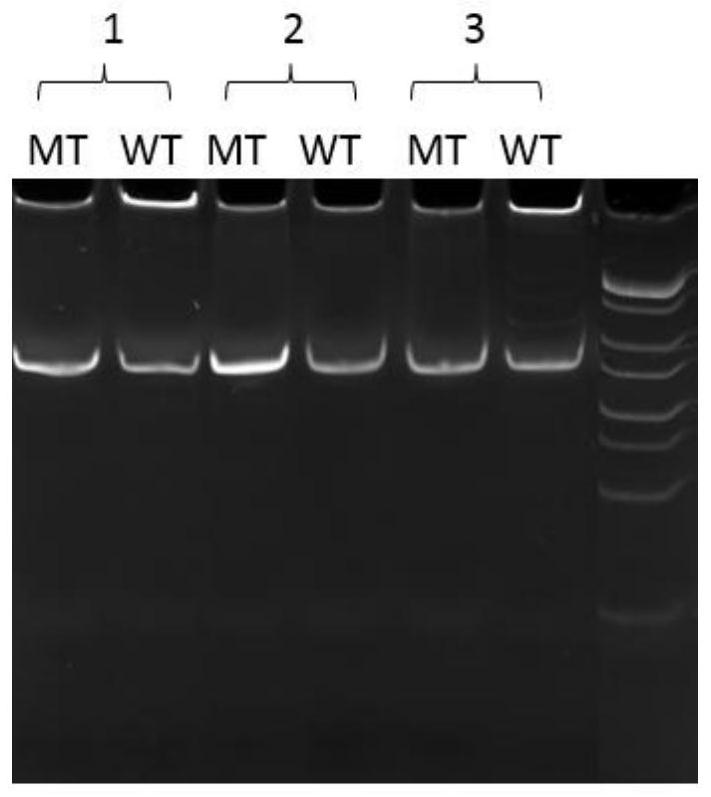
The invention provides a primer probe for detecting L858R mutation of an EGFR (epidermal growth factor receptor) gene, and belongs to the technical field of gene detection. An upstream primer sequence of the L858R mutation is as shown in SEQ ID NO: 1; a downstream primer sequence of the L858R mutation is as shown in SEQ ID NO: 2; and the L858R mutation probe is a gold-labeled probe, and the sequence of the L858R mutation probe is as shown in SEQ ID NO: 3. The primer probe used in the invention has high specificity, accuracy and sensitivity. According to the primer probe, an RPA amplification technology is combined with a lateral flow chromatography test strip to detect the EGFR gene L858R mutation for the first time, the detection time is short, the operation is simple and convenient, and the cost is low.
Owner:ENFIN BIOTECH (JIANGSU) CO LTD
Novel EGFR and alk dual inhibitor
The present application provides a compound of formula I, which is a EGFR and ALK dual inhibitor and can be used alone or in combination with other therapeutic agents to treat diseases such as non-small cell lung cancer. The compounds of the present application are useful in the treatment of diseases carrying the EGFR wild-type gene, or carrying the EGFR T790M mutant gene and / or the EGFR L858R mutant gene and / or the EGFR delE746_A750 mutant gene, or in the treatment of diseases carrying the ALK wild-type gene, ALK F1174L mutant gene and / or ALK F1196M gene and / or EML4-ALK mutant gene and / or NPM-ALK mutant gene, and can be used in the first-line treatment of anaplastic lymphoma kinase (ALK) positive late-stage non-small cell Lung cancer.
Owner:PRECEDO PHARMA CO LTD
Multiple enrichment detection method of low-frequency mutation relevant to non-small cell lung cancer target medicine
ActiveCN110438210AEfficient suppressionReduce testing costsMicrobiological testing/measurementDNA/RNA fragmentationFluorescenceWild type
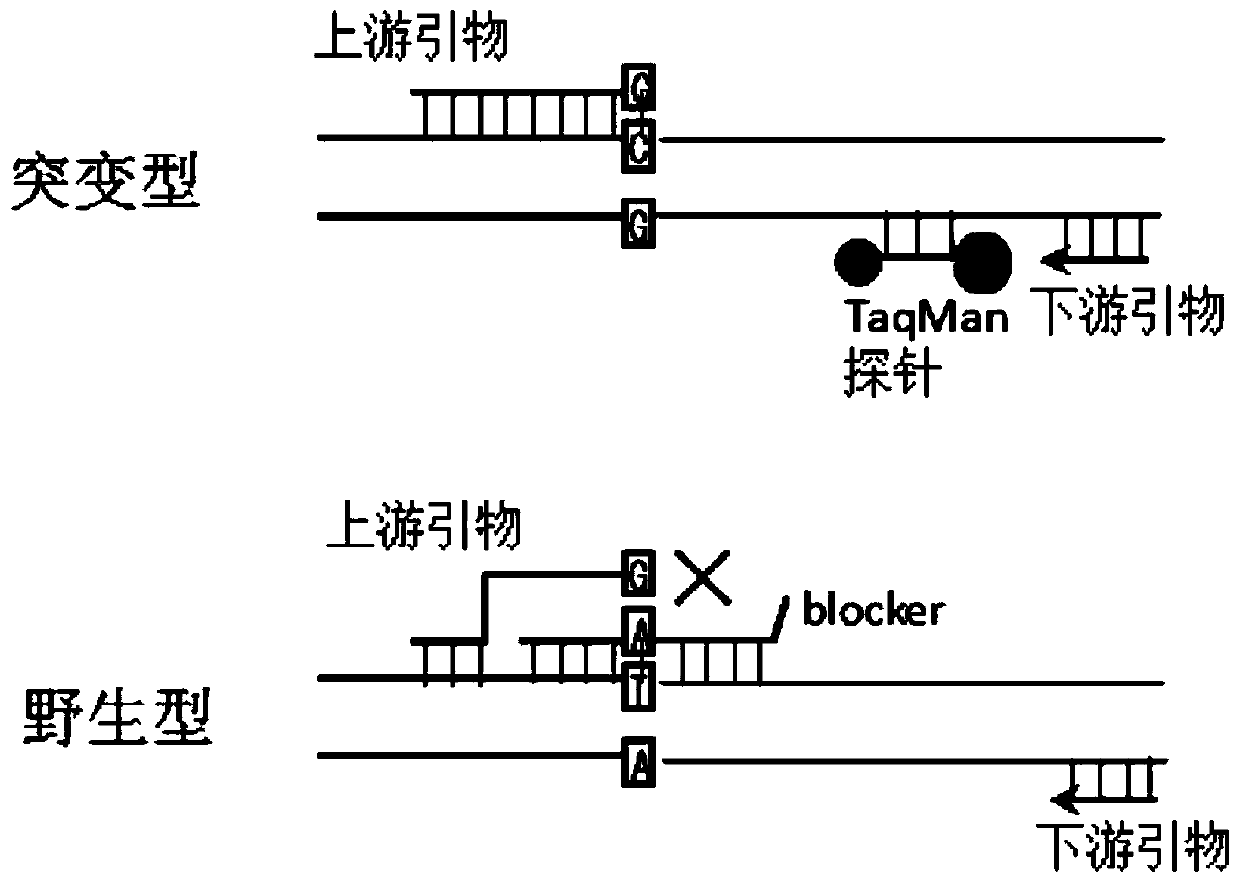
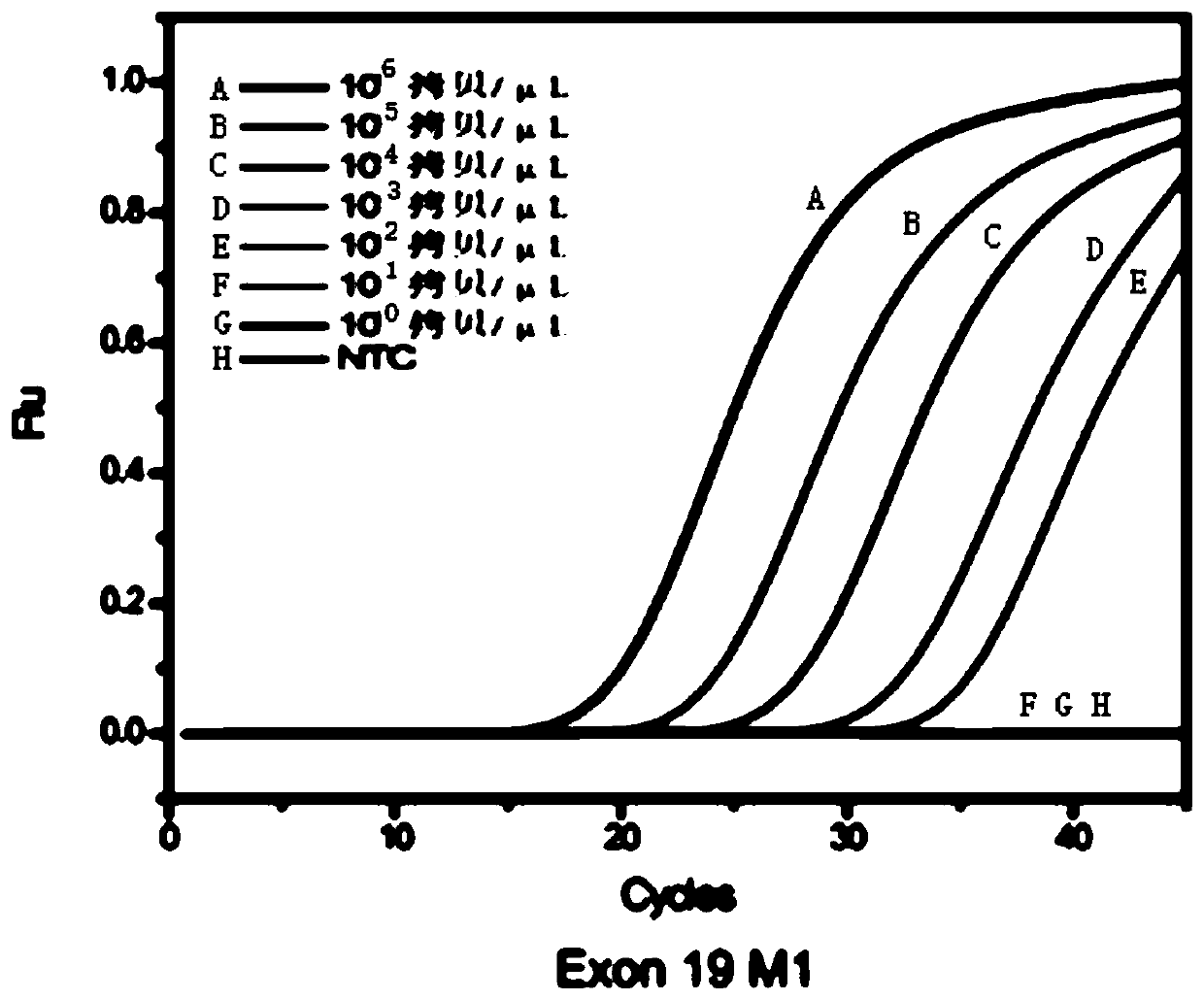
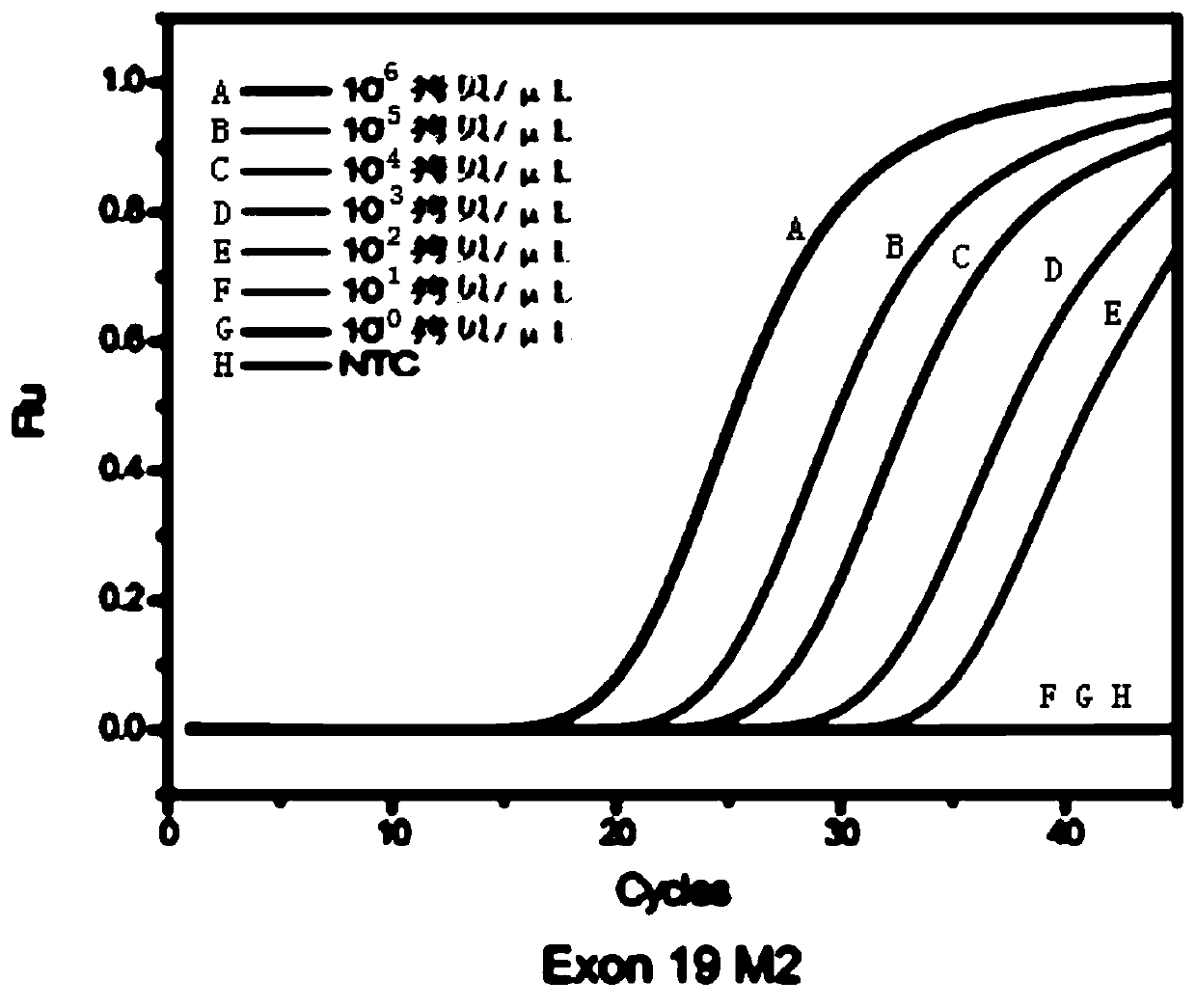
The invention relates to a multiple enrichment detection method of low-frequency mutation relevant to a non-small cell lung cancer target medicine. Four mutations of 2235_2249del-15 and 2236_2250del-15 which are highest in frequency in deletion mutations on an exon 19 of a human EGFR gene, a T790M mutation on an exon 20, and an L858R mutation on an exon 21 are used as targets, primers, blockers and TaqMan probes marking different fluorescence are respectively designed in accordance with each mutation site, an amplification curve is drawn through collecting fluorescence signals in the amplification course, the Cq value of the amplification curve and a threshold determined through a standard curve are compared, and whether samples have corresponding mutations or not is judged. The system disclosed by the invention is optimized in accordance with cfDNA, so that the system can realize amplification and detection on a target sequence under the situation that the formwork gragmentation degree is high and the wild type background is powerful. The method concurrently has the characteristics of being sensitive and multiple, the detection operations are convenient, quick and accurate, and the method has large application value.
Owner:FUDAN UNIV
Digital PCR kit for detecting human EGFR gene mutation
PendingCN113584132ALow costImprove throughputMicrobiological testing/measurementDNA/RNA fragmentationFluoProbesMedicine
Owner:TARGETINGONE TECH (BEIJING) CORP
Optimization method of EGFR gene L858R mutation digital PCR detection system and detection product
PendingCN109295225APlay a decisive roleThe result is accurateMicrobiological testing/measurementEnzyme digestionMutation frequency
The invention relates to an optimization method of a EGFR gene L858R mutation digital PCR detection system, wherein the detection system includes upstream primers, downstream primers, wild-type probes, mutant-type probes, a wild-type template and a mutant-type template; the wild-type template is normal human gDNA after enzyme digestion, and the mutant-type template is a mutant plasmid inserted with a EGFR gene L858R mutant fragment after enzyme digestion; a standard substance is prepared from the mutant-type template and the wild-type template according to a certain proportion of copy number;reaction is performed with a medium mutation frequency standard substance as a template by digital PCR, a data statistical graph is prepared according to the reaction data, and a wild-type fluorescentregion and a mutant-type fluorescent region are selected. Reaction is performed with the wild-type template as the template by digital PCR, and the background threshold value is determined. A detection product optimized by the optimization method of the EGFR gene L858R mutation digital PCR detection system has higher accuracy degree.
Owner:上海赛安生物医药科技股份有限公司
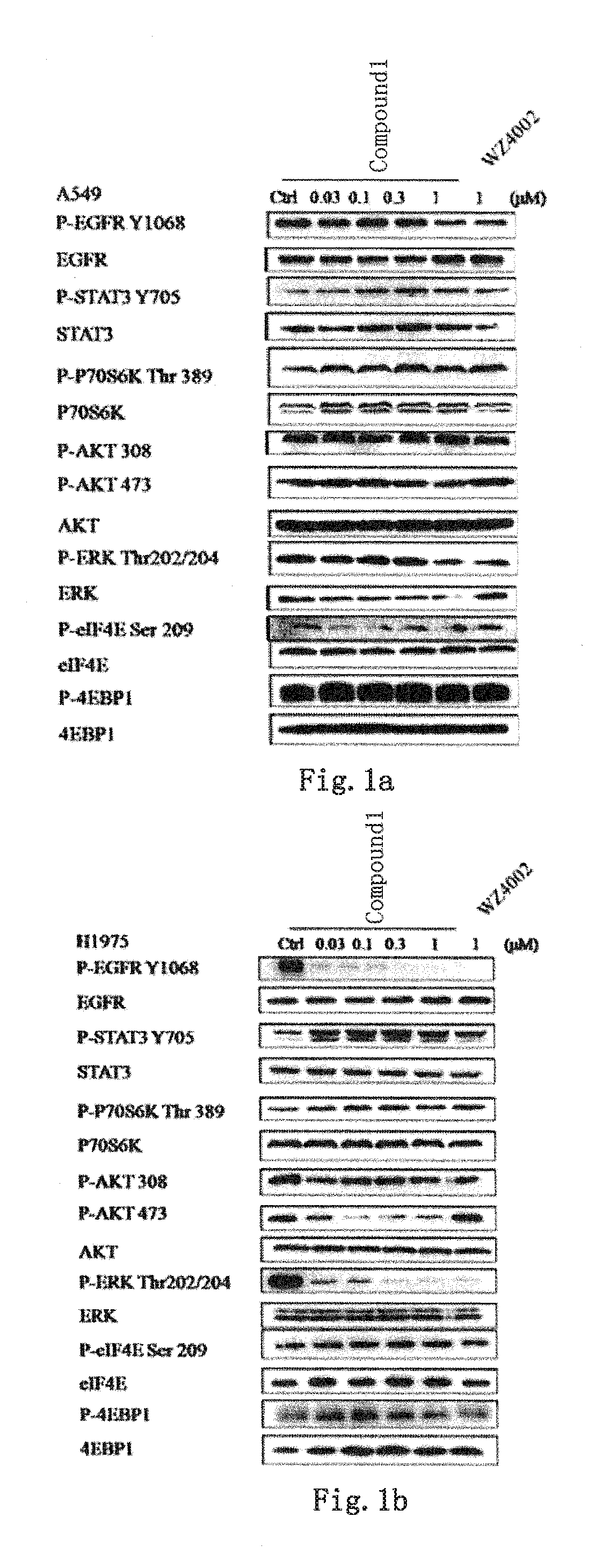
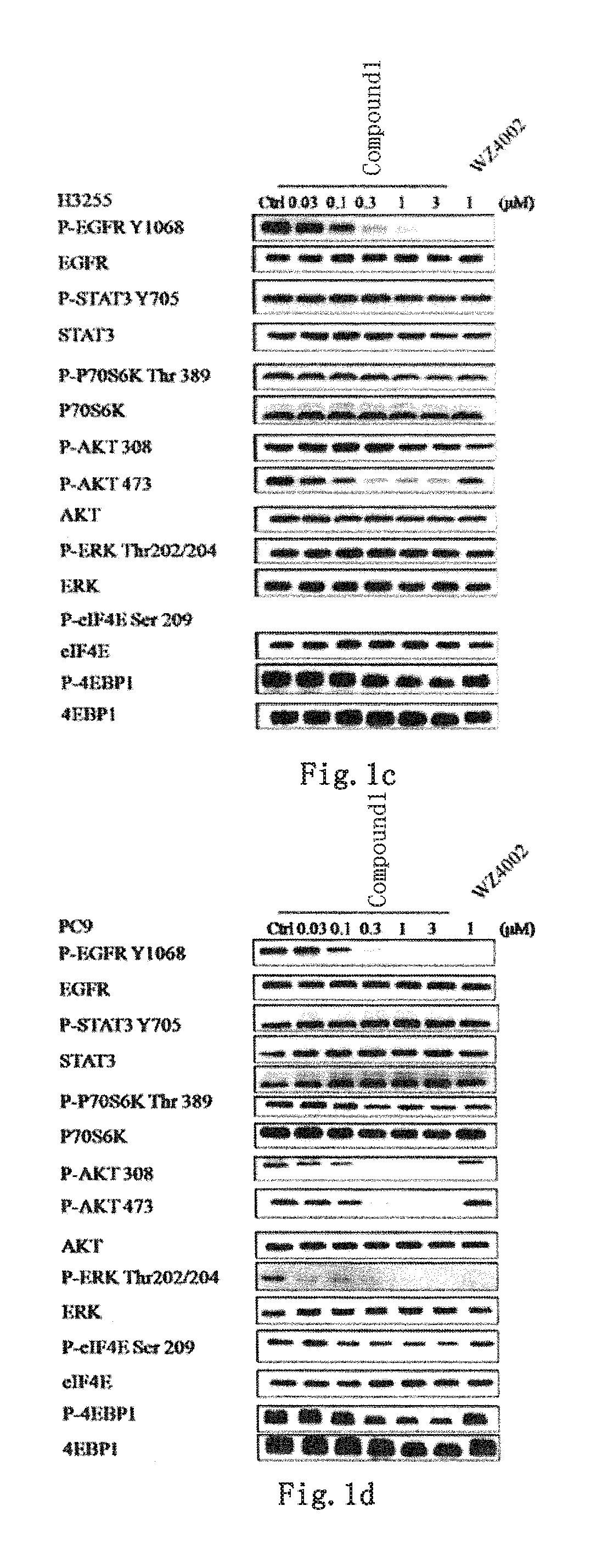
Novel application of quinazoline derivative type tyrosine kinase inhibitor
PendingCN110498804AOrganic active ingredientsOrganic chemistry methodsTyrosine-kinase inhibitorQuinazoline
The invention discloses novel application of a quinazoline derivative type tyrosine kinase inhibitor, belongs to the technical field of medicines, and in particular relates to novel application of a compound of a formula (I) shown in the description, and a stereisomer, a pharmaceutically acceptable salt and a crystal forma thereof, and in the formula, R1, R2, R3, R4, R5, R6, L and n are defined inthe description. The invention specifically relates to the compounds and application of medicine preparations and medicine compositions with the compounds in preparing medicines for treating tumor patients with EGFR_T790M mutation, EGFR_L858R mutation, EGFR_C797S mutation, EGFR_d746-750 mutation, EGFR_T790M / L858R mutation, EGFR_T790M / C797S mutation, EGFR_T790M / d746-750 mutation, EGFR_C797S / L858Rmutation, EGFR_L858R / d746-750 mutation, EGFR_C797S / d746-750 mutation, EGFR_L858R / T790M / C797S mutation, EGFT_d746-750 / T790M / C797S mutation or EGFR_d746-750 / L858R / C797S.
Owner:SHANDONG XUANZHU PHARMA TECH CO LTD
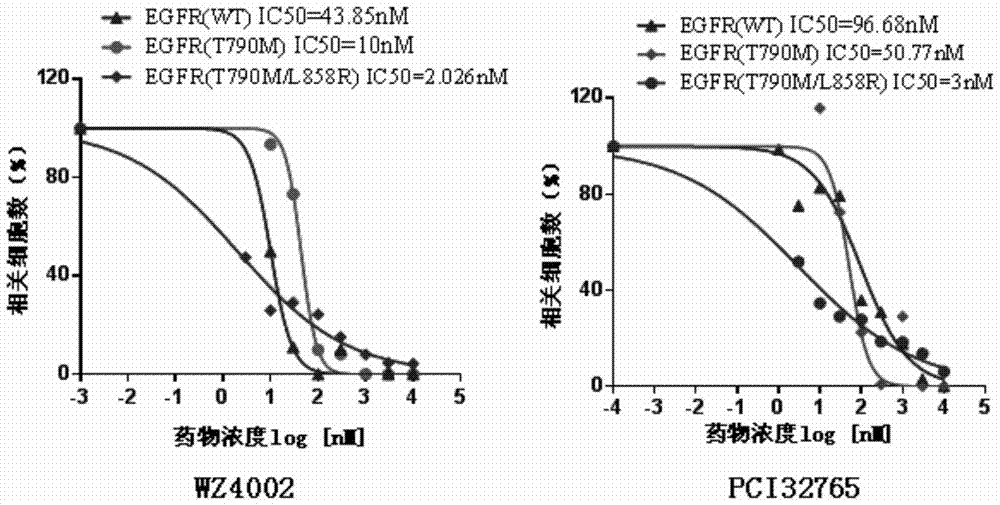
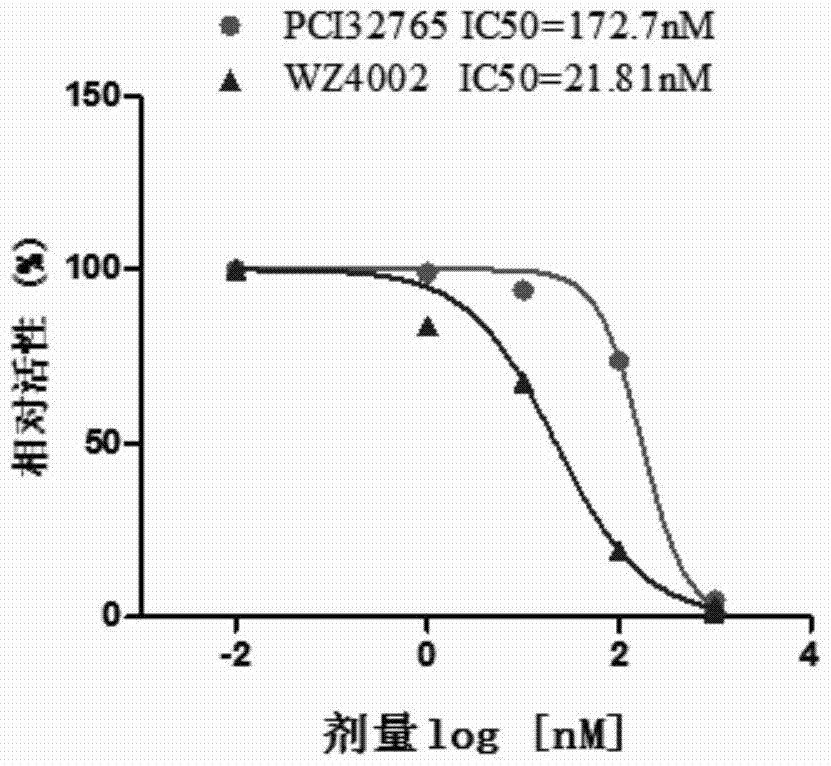
Uses of ibrutinib
InactiveCN104825455BInhibition amountPromote apoptosisOrganic active ingredientsAntineoplastic agentsDouble mutationEGFR T790M
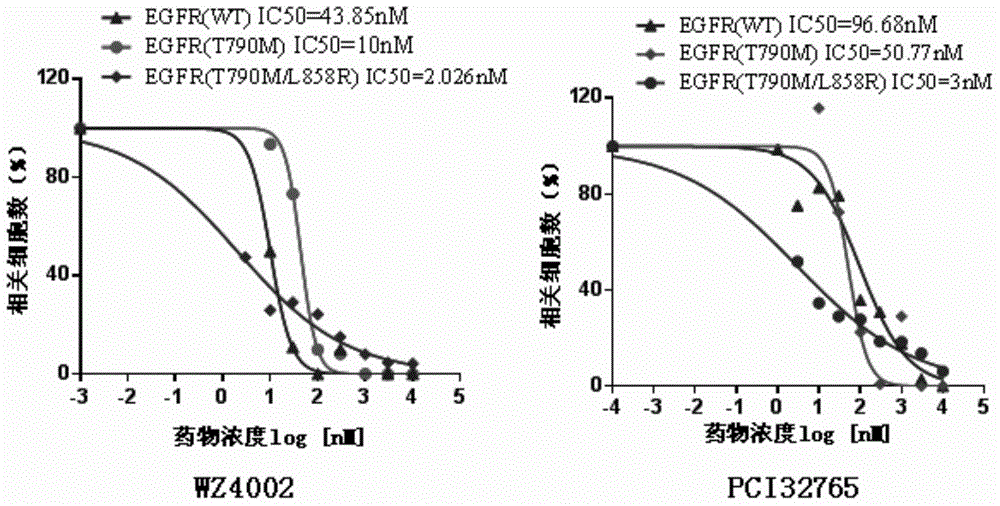
Disclosed is a new use of ibrutinib (PCI-32765). In particular, the present invention has found that ibrutinib can treat non-small cell lung cancer carrying EGFR T790M mutations and / or EGFR L858R mutations and / or EGFR delE746_A750 mutations, especially non-small cell lung cancer carrying EGFR L858R and EGFR T790M double mutations.
Owner:HEFEI INSTITUTES OF PHYSICAL SCIENCE - CHINESE ACAD OF SCI
Detection kit and detection method for gene mutation of lung cancer-related molecular markers
ActiveCN110904239BLow costSimple methodMicrobiological testing/measurementDNA/RNA fragmentationGenes mutationBRAF Gene Mutation
The invention provides crRNAs for gene mutation of lung cancer-related molecular markers, a detection kit and a detection method thereof. The crRNAs comprise: a crRNA at the EGFR-T790M mutation site:as shown in any one of SEQ ID NO.16-17; a crRNA at the EGFR-L858R mutation site: as shown in any one of SEQ ID NO.18-19; a crRNA at the BRAF gene mutation site: as shown in any one of SEQ ID NO.20-21;a crRNA at the ALK gene mutation site: as shown in any one of SEQ ID NO.22-23; and a crRNA at the ROS1 gene mutation site: as shown in any one of SEQ ID NO.24-25. According to the invention, the 5 crRRNAs related to gene mutation targets of lung cancer-related molecular markers are designed, and the crRNAs are adopted in combination with a CRISPR-cpf1 system to carry out mutation detection. The method of the invention has the characteristics of simplicity, fast speed, high sensitivity and strong specificity.
Owner:江苏博嘉生物医学科技有限公司
EGFR and ALK dual inhibitor
The present application provides a compound of formula I, which is a EGFR and ALK dual inhibitor and can be used alone or in combination with other therapeutic agents to treat diseases such as non-small cell lung cancer. The compounds of the present application are useful in the treatment of diseases carrying the EGFR wild-type gene, or carrying the EGFR T790M mutant gene and / or the EGFR L858R mutant gene and / or the EGFR delE746_A750 mutant gene, or in the treatment of diseases carrying the ALK wild-type gene, ALK F1174L mutant gene and / or ALK F1196M gene and / or EML4-ALK mutant gene and / or NPM-ALK mutant gene, and can be used in the first-line treatment of anaplastic lymphoma kinase (ALK) positive late-stage non-small cell Lung cancer.
Owner:PRECEDO PHARMA CO LTD
Primer probe, kit and application for detection of egfr gene l858r mutation
ActiveCN113355423BSimple and fast operationShort detection timeMicrobiological testing/measurementDNA/RNA fragmentationGenes mutationMutation detection
The invention provides a primer probe for detecting the EGFR gene L858R mutation, which belongs to the technical field of gene detection. The upstream primer sequence of the L858R mutation is shown in SEQ ID NO: 1; the downstream primer sequence of the L858R mutation is shown in SEQ ID Shown in NO:2; the probe for the L858R mutation is a gold-labeled probe, and its sequence is shown in SEQ ID NO:3. The primer probe used in the present invention has high specificity, accuracy and sensitivity. The invention adopts the RPA amplification technology combined with the lateral flow chromatography test strip to detect the EGFR gene L858R mutation for the first time, and the detection time is short, the operation is simple and the cost is low.
Owner:ENFIN BIOTECH (JIANGSU) CO LTD
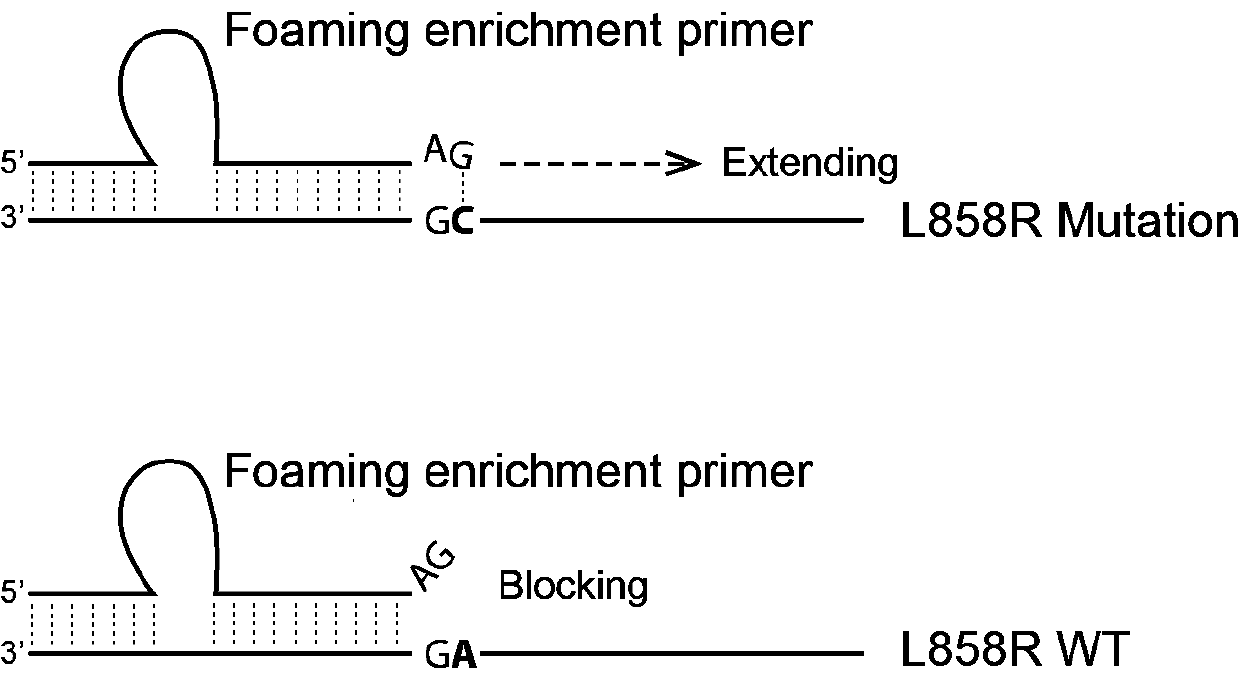
EGFR/L858R mutation hypersensitivity detection kit
PendingCN110343745AEasy to operateThe detection process is fastMicrobiological testing/measurementSerum freeWild type
The invention provides an EGFR / L858R mutation hypersensitivity detection kit and application thereof. The EGFR / L858R mutation hypersensitivity detection kit is characterized by comprising an L858R mutation upstream foaming enrichment primer, an EGFR gene downstream primer, a qPCR reaction system reagent and positive plasmids of mutant and wild type EGFR genes. The kit can be used for qualitative detection of lung cancer EGFR / L858R mutant serum free DNA, and has a lower limit of experimental detection of 0.1%. A detection method using the kit only relates to dye method qPCR, does not relate tosynthesis and modification of probes, is low in detection cost, and is beneficial to saving gene detection cost of vast patients.
Owner:陈超
New application of ibrutinib
InactiveCN104825455AInhibition amountPromote apoptosisOrganic active ingredientsAntineoplastic agentsDouble mutationEGFR T790M
The invention discloses a new application of ibrutinib (PCI-32765). In particular, it is found that the ibrutinib can treat non-small cell lung cancer carrying EGFR T790M mutation and / or EGFR L858R mutation and / or EDFR delE746-A750 mutation, and especially, can treat the non-small cell lung cancer carrying double-mutation of EGFR L858R and EGFR T790M.
Owner:HEFEI INSTITUTES OF PHYSICAL SCIENCE - CHINESE ACAD OF SCI
Novel kinase inhibitor against wild-type and mutant EGFR
The present application provides a compound of Formula (I) as an inhibitor against wild-type EGFR and / or mutant EGFR, which may be used for treating human non-small cell lung cancer individually or in combination with other therapeutic agent(s). The compound of Formula (I) of the present application may be used for treating patients of drug-resistant human non-small cell lung cancer harboring wild-type EGFR and / or EGFR T790M mutation and / or EGFR L858R mutation and / or EGFR delE746_A750 mutation.
Owner:PRECEDO PHARMA CO LTD
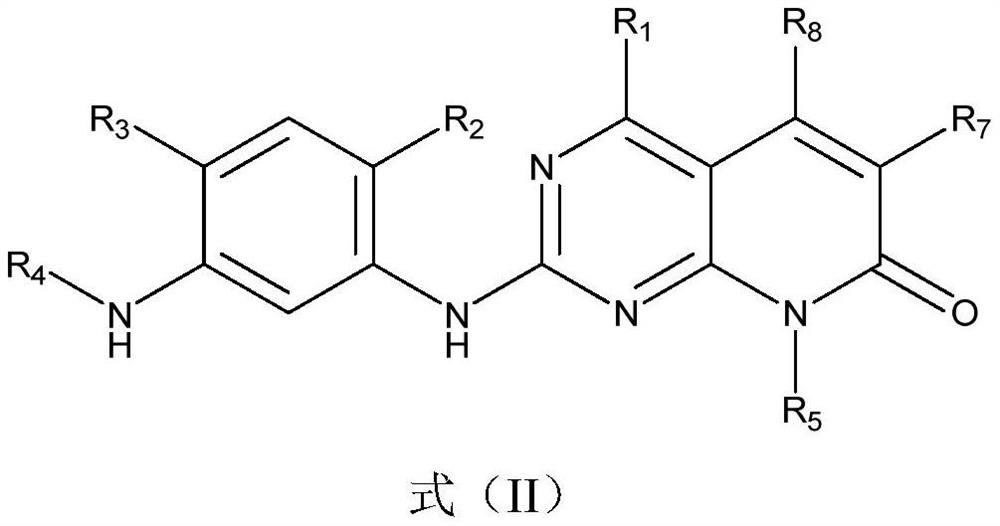
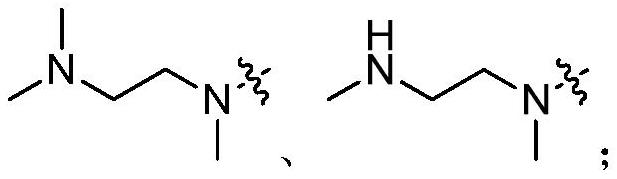
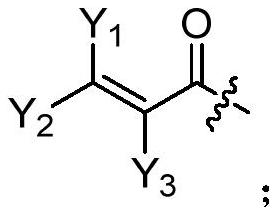
A kind of compound containing pyrimidine ring, egfr inhibitor and application thereof
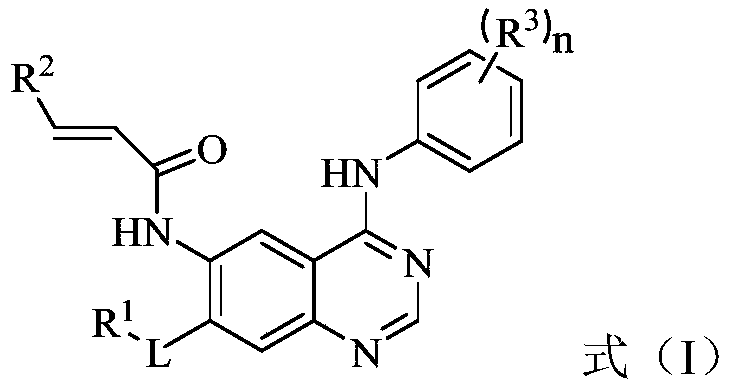
The present invention relates to the technical field of pharmaceutical compositions and relates to a compound containing a pyrimidine ring, an epidermal growth factor receptor (EGFR) inhibitor and application thereof. The compound containing the pyrimidine ring comprises a compound represented by formula (I) or a pharmaceutically acceptable salt, stereoisomer, solvate or prodrug thereof. The EGFR inhibitor comprises the compound containing the pyrimidine ring. The compound can inhibit one or more activating or resistant mutations in EGFR and can be used to prepare drugs for controlling EGFR tyrosine kinase activity or treating EGFR related diseases, such as cancers, diabetes, immune system diseases, neurodegenerative diseases or cardiovascular diseases, and is particularly suitable for preparing drugs for treating non-small cell lung cancer caused by EGFR mutations including sensitive mutations (such as an L858R mutation or an exon 19 deletion mutation) and drug-resistance mutations (such as an EGFR T790M mutation).
Owner:TETRANOV PHARMA CO LTD
Lung cancer clinical drug gene detection standard and its application
ActiveCN107345253BVerify accuracyAccurate guidanceMicrobiological testing/measurementDNA/RNA fragmentationGenes mutationReference product
The invention discloses a standard product for detecting genes of clinical medications for lung cancer and an application thereof, wherein the standard product comprises a lowest-detection-limit reference product at least comprising two different levels of variation-frequency DNA mixtures; and each level of variation-frequency DNA mixture comprises the following mutation sites: EGFR gene G719S, E746_A750del, S768I, T790M and L858R mutation sites, EML4-ALK gene E6, E20 and E13, E20 mutation site, SLC34A2-ROS1 gene E4, and E32 mutation site. By application of the technical scheme disclosed by the invention, whether a kit can detect the gene mutation and the gene frequency can be judged according to results, the accuracy of detection results of the existing kit can be verified and more accurate guidance also can be provided for clinical medications.
Owner:GENE CRAB BIOTECH CO +1
Digital PCR detection kit for EGFR gene L858R mutation site
InactiveCN109295180AAccurate detectionEasy to detectMicrobiological testing/measurementFresh TissueMutation detection
The invention discloses a digital PCR detection kit for an EGFR gene L858R mutation site. According to the invention, EGFR gene L858R mutation is adopted as the detection object, through optimized combination of specific primers and fluorescent probe, accurate, simple and rapid simultaneous detection of EGFR gene L858R mutation can be achieved, and the ability of mutation detection is as high as 0.1%. The kit can effectively detect L858R mutation in patients' free DNA, and still has the same amplification and detection ability on a short fragment DNA obtained from formaldehyde-fixed and paraffin-embedded sample as that of a fresh tissue sample DNA.
Owner:XIAMEN SPACEGEN BIOTECH CO LTD
A digital pcr kit for detecting mutations in human egfr gene
ActiveCN110564856BLow costImprove throughputMicrobiological testing/measurementDNA/RNA fragmentationGenes mutationFluoProbes
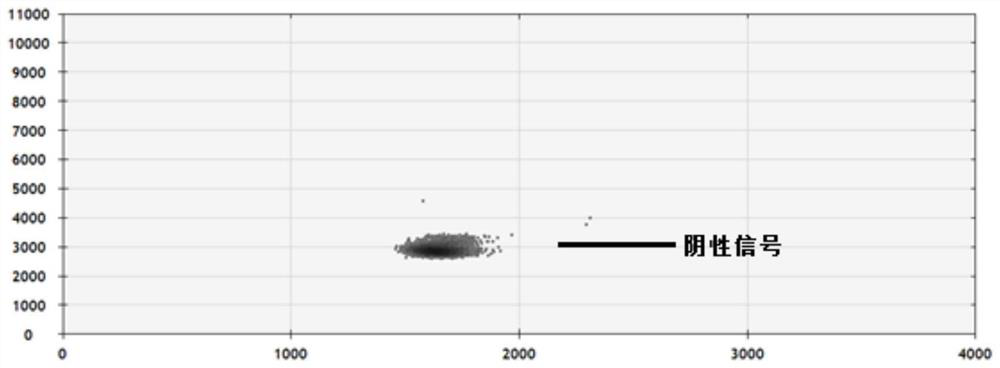
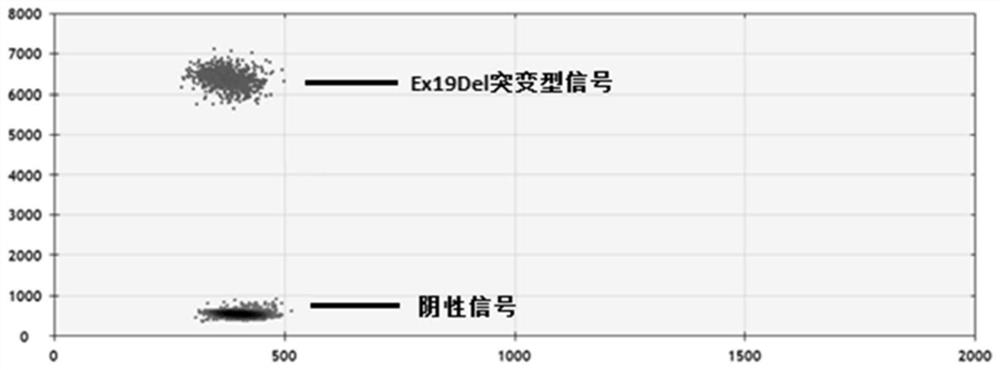
The invention provides a digital PCR kit for detecting human EGFR gene mutations, the digital PCR kit includes simultaneous detection of multiple Ex19Del mutations and L858R mutations in the same reaction tube digital PCR reaction system, wherein the Ex19Del site uses multiple ARMS primers were used to identify the mutant type, and TaqMan fluorescent probe was used to identify the wild type and mutant type at the L858R site; after PCR amplification, the Ex19Del mutant type, L858R mutant type, and L858R wild type were detected by the position and intensity of the fluorescent signal, and compared Absolute quantification is performed directly. The kit of the present invention has high throughput and low cost, and realizes the detection of the Ex19Del25 site and the L858R site in one tube, which improves the detection throughput and reduces the cost of reagent consumables.
Owner:TARGETINGONE TECH (BEIJING) CORP
A kind of EGFR inhibitor for targeted therapy of cancer and its preparation method and application
The invention discloses an EGFR inhibitor for targetedly treating a cancer and a preparation method and application thereof.The structural formula of the EGFR inhibitor is shown in the formula I.The EGFR inhibitor can be used for preventing and / or treating the cancer such as a human skin caner or a lung cancer.The EGFR inhibitor can selectively inhibit cell lines of EGFR double mutation (EGFRT790M and L858R), and the inhibitory activity to EGFR wild type cells is weak; therefore, the EGFR inhibitor can be used for treating lung cancer patients with EGFRT790M and L858R mutation, and the side effects are fewer, wherein the side effects are caused by inhibiting wild type EGFR, for example, Afatinib.
Owner:TSINGHUA UNIV