[0021]The methods and systems of the invention have the
advantage of converting bio-based feedstocks, optionally without any additional costly purification steps to form higher
energy density product of lower
oxygen / carbon ratio including
higher alkanes, olefins, and aromatics. The invention also reduces the amount of unwanted byproducts, thereby improving the overall yield of converting carbohydrates to higher hydrocarbons. Another
advantage of the present invention includes the use of similar catalysts for multiple reaction steps, offering the potential to combine reactions when desired. A further
advantage of the present invention provides the combination or
elimination of steps required to convert bio-based feedstocks to fuel, thereby reducing capital costs. While not intending to be limited by theory, it is believed that some carbohydrates may thermally degrade at the conditions needed to produce higher hydrocarbons. In addition, the inclusion of some higher polyols in a reaction to form high hydrocarbons may result in excessive condensation so that the resulting higher hydrocarbons are unusable for transportation fuels (e.g., tars). As an advantage of the present process, the
hydrogenolysis reaction may allow for the formation of stable hydroxyl intermediates that can withstand the conditions needed to form higher hydrocarbons and allow for the formation of desired higher hydrocarbons that may be used for fuels or as other industrial chemicals. Advantages of specific embodiments will be described in more detail below.
[0022]An embodiment of the invention comprises providing a carbohydrate feed; contacting at least a portion of the carbohydrate feed directly with
hydrogen in the presence of a
hydrogenolysis catalyst to produce a first reaction product comprising stable hydroxyl intermediates; contacting at least a portion of the first reaction product comprising the stable hydroxyl intermediates with a
dehydrogenation catalyst to form a second reaction product; and contacting at least a portion of the second reaction product with a condensation catalyst comprising a base functionality to form a fuel blend.
[0023]Carbohydrates are the most abundant, naturally occurring biomolecules.
Plant materials store carbohydrates either as sugars, starches, celluloses, lignocelluloses, hemicelluloses, and any combination thereof. In one embodiment, the carbohydrates include monosaccharides, polysaccharides or mixtures of monosaccharides and polysaccharides. As used herein, the term “monosaccharides” refers to hydroxy aldehydes or hydroxy ketones that cannot be hydrolyzed to smaller units. Examples of monosaccharides include, but are not limited to, dextrose, glucose,
fructose and
galactose. As used herein, the term “polysaccharides” refers to saccharides comprising two or more
monosaccharide units. Examples of polysaccharides include, but are not limited to,
sucrose,
maltose,
cellobiose,
cellulose and
lactose. Carbohydrates are produced during
photosynthesis, a process in which
carbon dioxide is converted into organic compounds as a way to store energy. The carbohydrates are highly reactive compounds that can be easily oxidized to generate energy, carbon dioxide, and water. The presence of
oxygen in the molecular structure of carbohydrates contributes to the reactivity of the compound.
Water soluble carbohydrates react with
hydrogen over catalyst(s) to generate stable hydroxyl intermediates comprising polyols and
sugar alcohols, either by hydrogenation, hydrogenolysis or both.
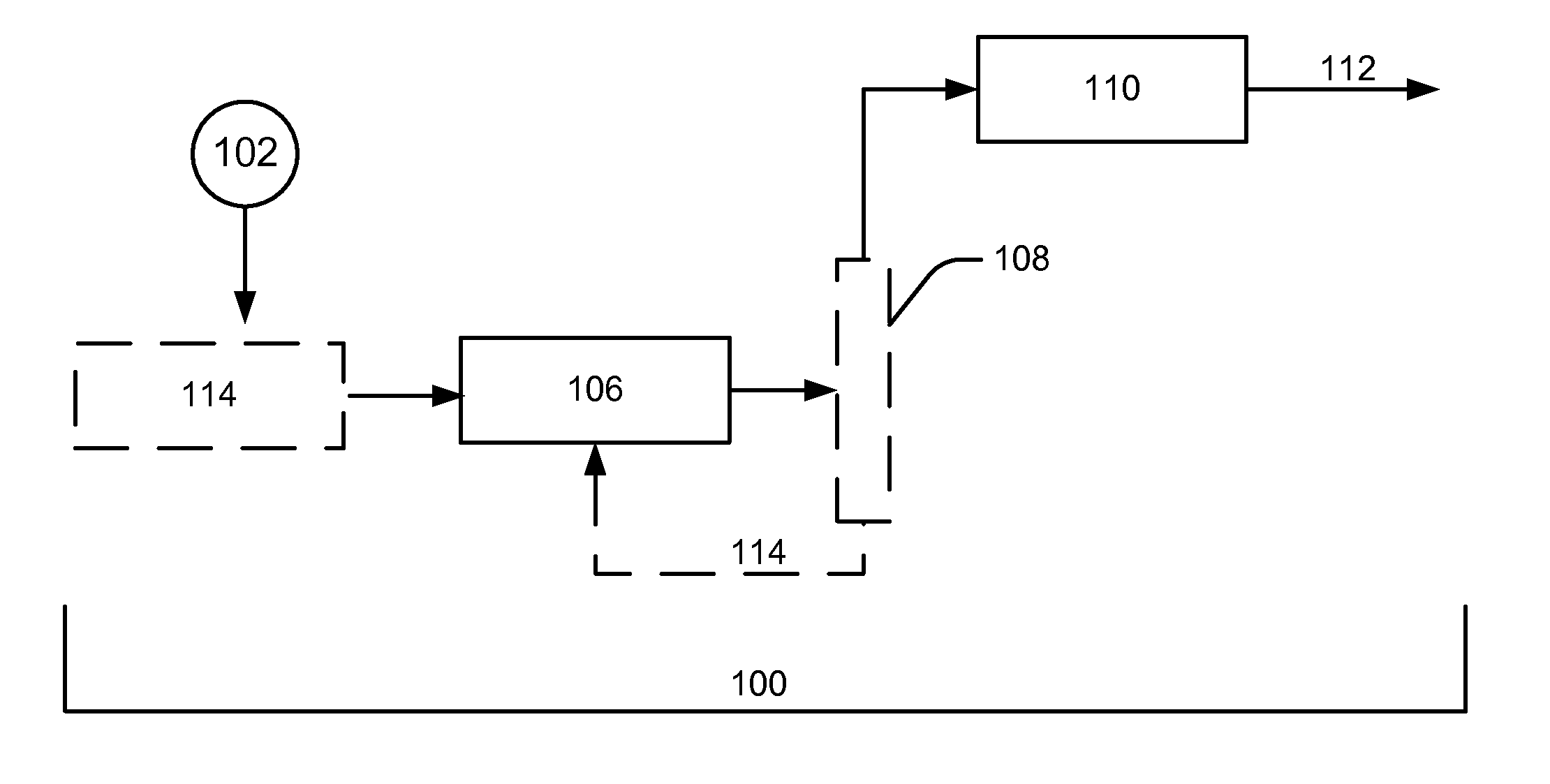
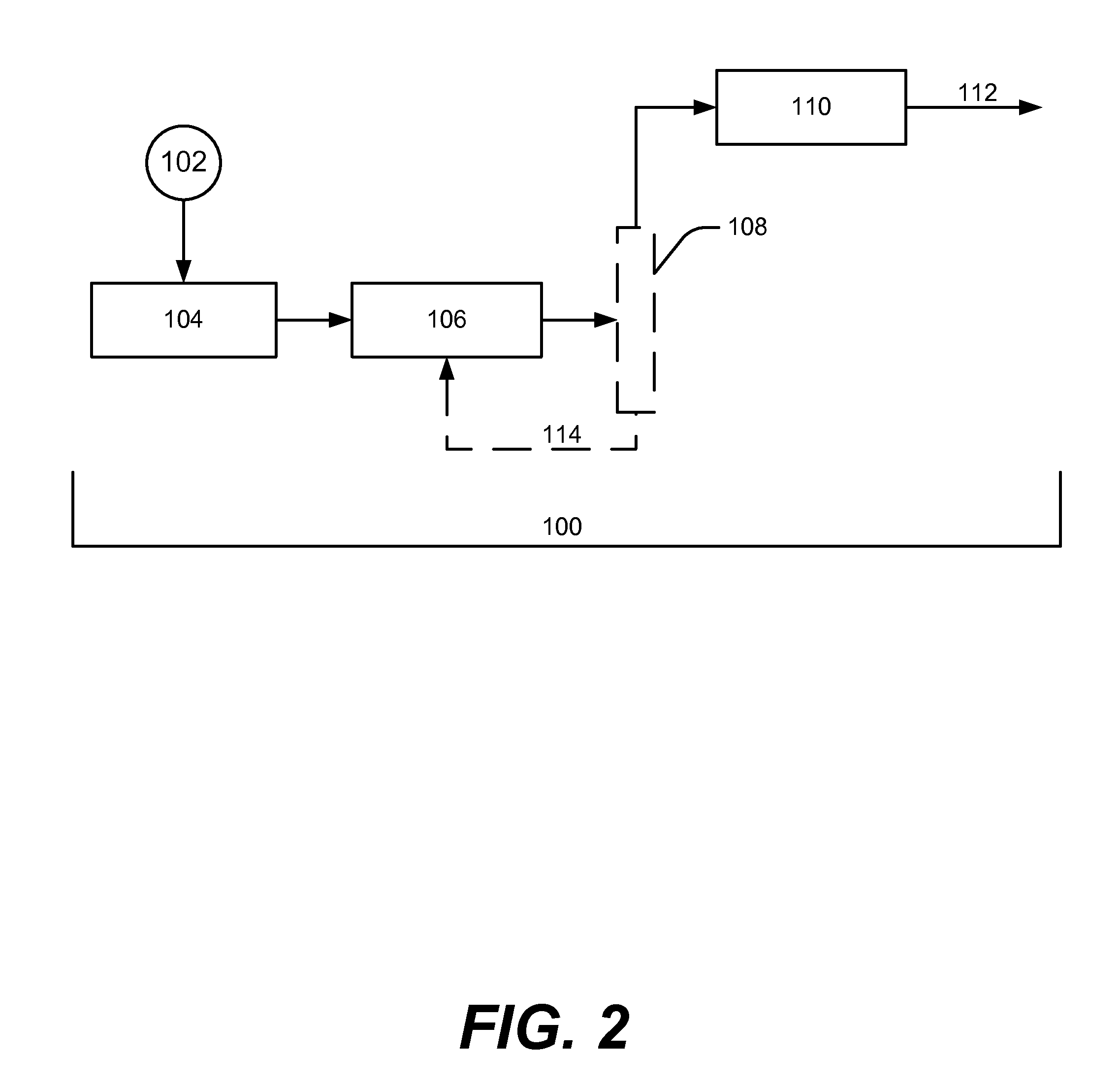
[0024]In the embodiment shown in FIG. 1, the carbohydrates are optionally reacted in a
hydrogenation reaction and then a hydrogenolysis reaction to form suitable stable hydroxyl intermediates that comprise alcohols and polyols for the
condensation reaction 110. In an embodiment of the invention, the
hydrogenation reaction is optional and the hydrogenolysis reaction alone is suitable to form the desired stable hydroxyl intermediates. In another embodiment of the invention, the carbohydrates are passed through the hydrogenolysis reaction prior to being passed through the
hydrogenation reaction (thus hydrogenolysis reaction 106 and hydrogenation reaction 104 are reversed from the order shown in FIG. 1). In an embodiment of the invention, the hydrogenation and hydrogenolysis reactions occur in the same vessel to generate stable hydroxyl intermediates to be fed into a
processing reaction. In an embodiment, a separation step (e.g., water removal) could be conducted prior to the hydrogenolysis reaction.
[0025]The carbohydrates may originate from any suitable source. In an embodiment, the carbohydrates fed to the process may be derived from an organic source (e.g., sugars and starches from corn or sugarcane). In another embodiment, the carbohydrates are derived from bio-based feedstocks. Bio-based feedstocks can include biomass. As used herein, the term “biomass” means organic materials produced by plants (e.g., leaves, roots, seeds and stalks), and microbial and animal metabolic wastes. Common sources of biomass include: agricultural wastes (e.g., corn stalks,
straw, seed hulls, sugarcane leavings,
bagasse, nutshells, and
manure from cattle, poultry, and hogs); wood materials (e.g., wood or
bark,
sawdust, timber slash, and mill
scrap); municipal waste, (e.g.,
waste paper and
yard clippings); and energy crops, (e.g., poplars, willows, switch grass, alfalfa, prairie bluestream, corn, and soybean). The term “biomass” also refers to the primary building blocks of all the above, including, but not limited to, saccharides, lignins, celluloses, hemicelluloses, and starches. Useful carbohydrates in the invention include, but are not limited to, carbohydrates that can be converted to hydrocarbons under suitable
reaction conditions. Suitable carbohydrates in the invention include any carbohydrate soluble in water or an
organic solvent having one or more carbon atoms and at least one
oxygen atom. The carbohydrates may also have an oxygen to carbon ratio from about 0.5:1 to about 1:1.2.
[0026]In one embodiment of the invention, the bio-based feedstock is optionally first hydrolyzed in a
liquid medium such as an
aqueous solution or
aqueous solution with
organic solvent (e.g., a recycled portion of the stable hydroxyl intermediates), to obtain a soluble carbohydrate
stream for use in the process. Various biomass
hydrolysis methods may be suitable, including, but not limited to,
acid hydrolysis,
alkaline hydrolysis,
enzymatic hydrolysis, and
hydrolysis using hot-compressed water. In certain embodiments, the
hydrolysis reaction can occur at a temperature between 100° C. and 250° C. and pressure between 0.1 MPa and 10,000 kPa. In embodiments including strong acid and
enzymatic hydrolysis, the hydrolysis reaction can occur at temperatures as low as ambient
temperature and pressure between 100 kPa and 10,000 kPa. In some embodiments, the hydrolysis reaction may comprise a hydrolysis catalyst (e.g., a
metal or acid catalyst) to aid in the hydrolysis reaction. The hydrolysis catalyst can be any catalyst capable of effecting a hydrolysis reaction. For example, suitable hydrolysis catalysts include, but are not limited to, acid catalysts, base catalysts,
metal catalysts, and any combination thereof. Acid catalysts can include organic acids such as
acetic acid,
formic acid, and
levulinic acid. In an embodiment, the acid catalyst can be generated as a byproduct during the hydrogenation and / or hydrogenolysis reactions. In certain embodiments, the hydrolysis of the bio-based feestocks can occur in conjunction with the hydrogenation and / or hydrogenolysis reactions. In such embodiments, a co-catalyst or catalytic support may be added to the hydrogenation and / or hydrogenolysis reactions to facilitate the hydrolysis reaction.
 Login to View More
Login to View More  Login to View More
Login to View More