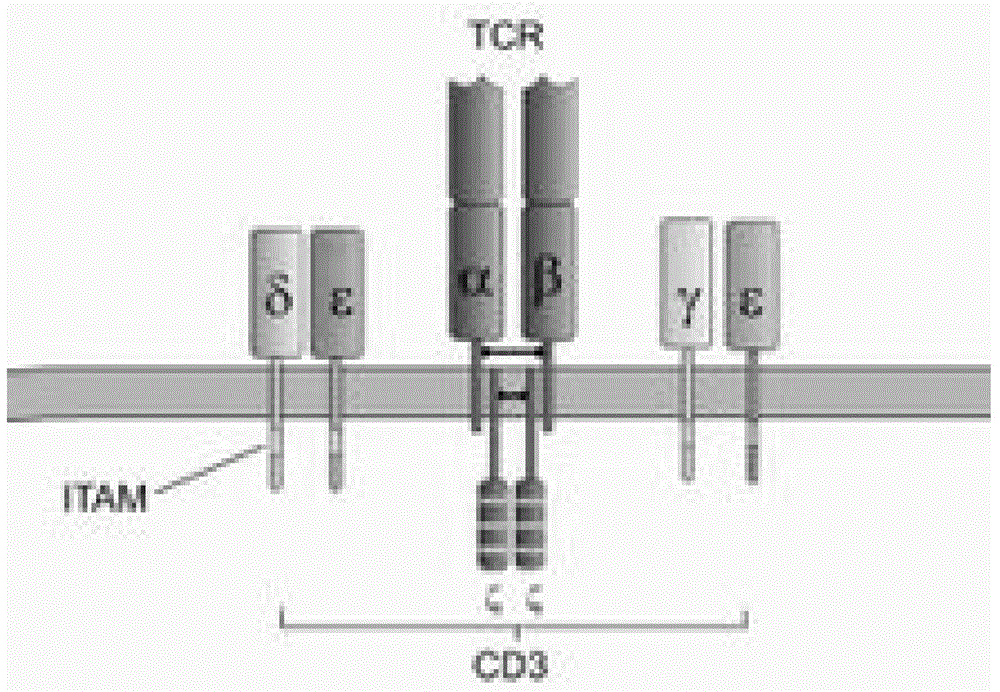
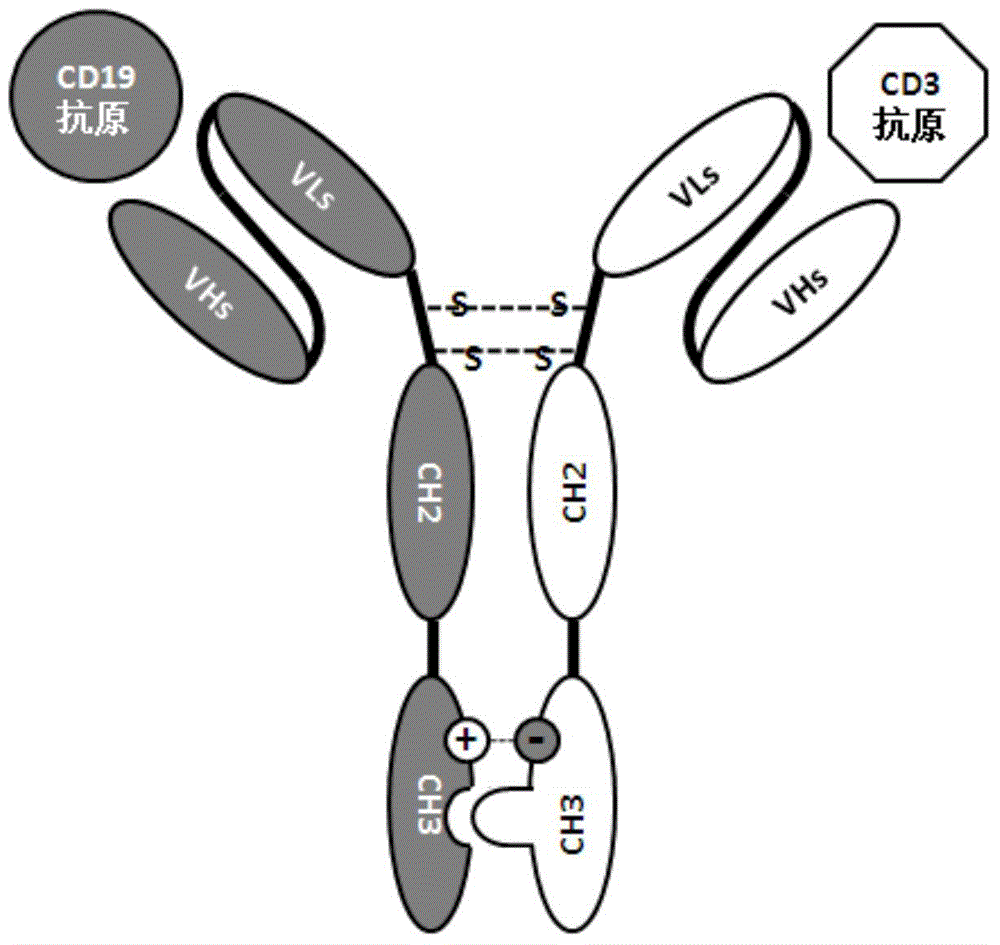
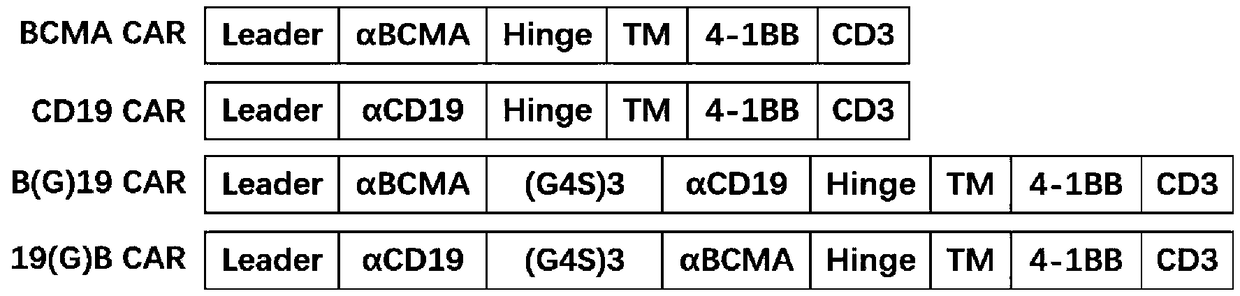
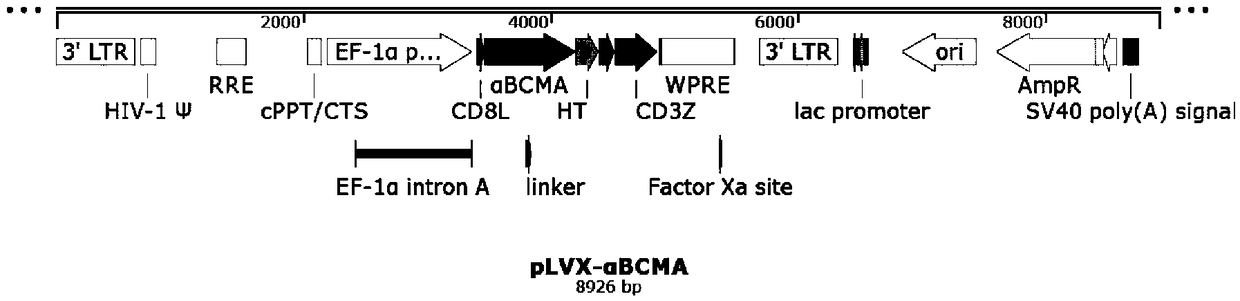
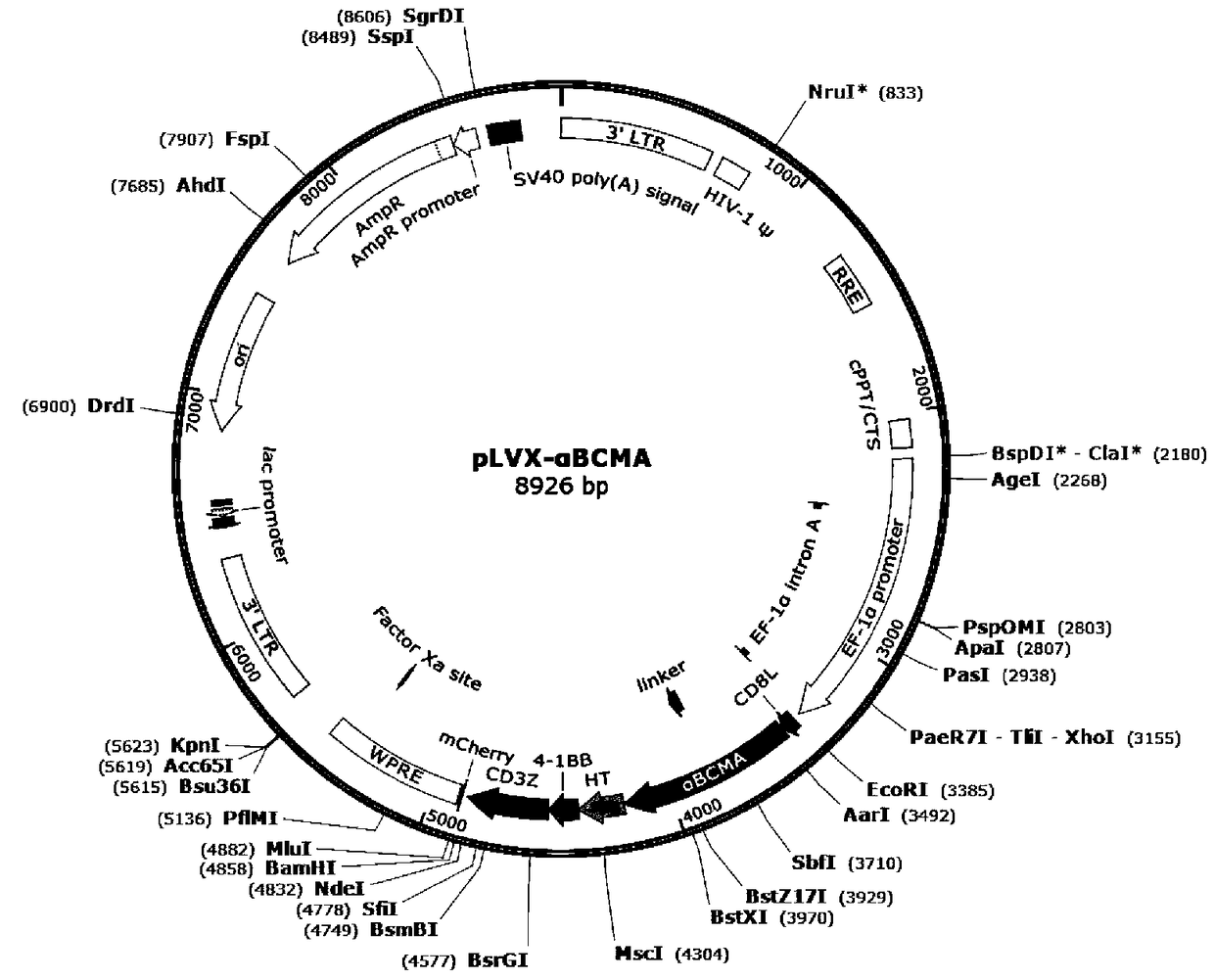
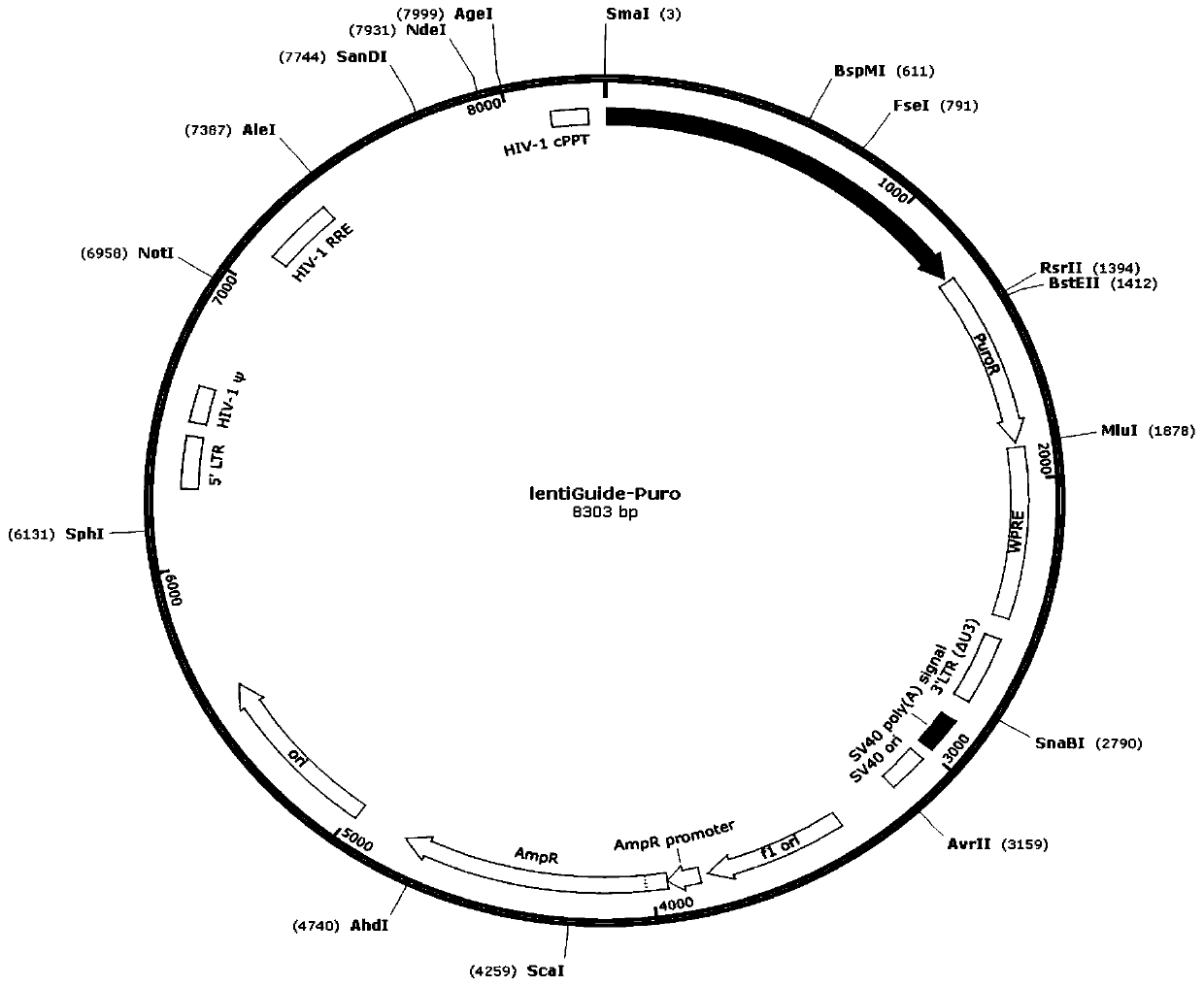
Patents
Literature
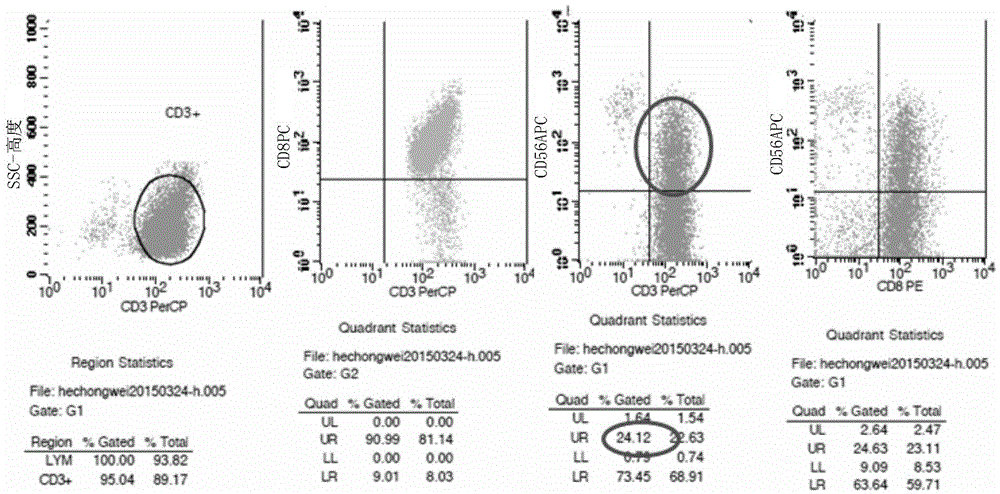
375 results about "CD19" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
B-lymphocyte antigen CD19, also known as CD19 molecule (Cluster of Differentiation 19), B-Lymphocyte Surface Antigen B4, T-Cell Surface Antigen Leu-12 and CVID3 is a transmembrane protein that in humans is encoded by the gene CD19. In humans, CD19 is expressed in all B lineage cells, except for plasma cells, and in follicular dendritic cells. CD19 plays two major roles in human B cells. It acts as an adaptor protein to recruit cytoplasmic signaling proteins to the membrane and it works within the CD19/CD21 complex to decrease the threshold for B cell receptor signaling pathways. Due to its presence on all B cells, it is a biomarker for B lymphocyte development, lymphoma diagnosis and can be utilized as a target for leukemia immunotherapies.
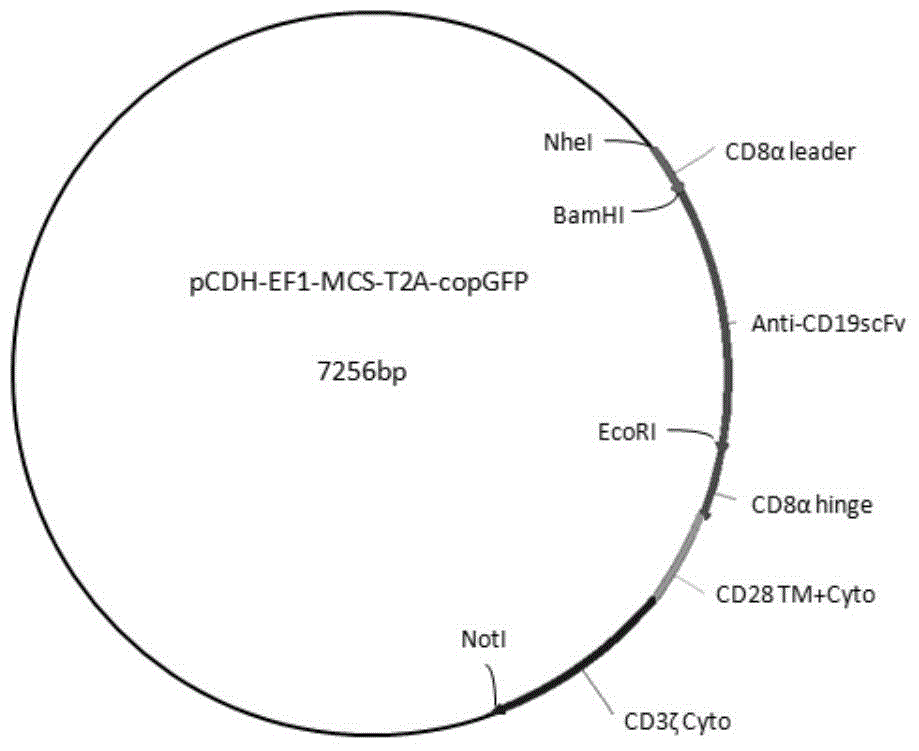
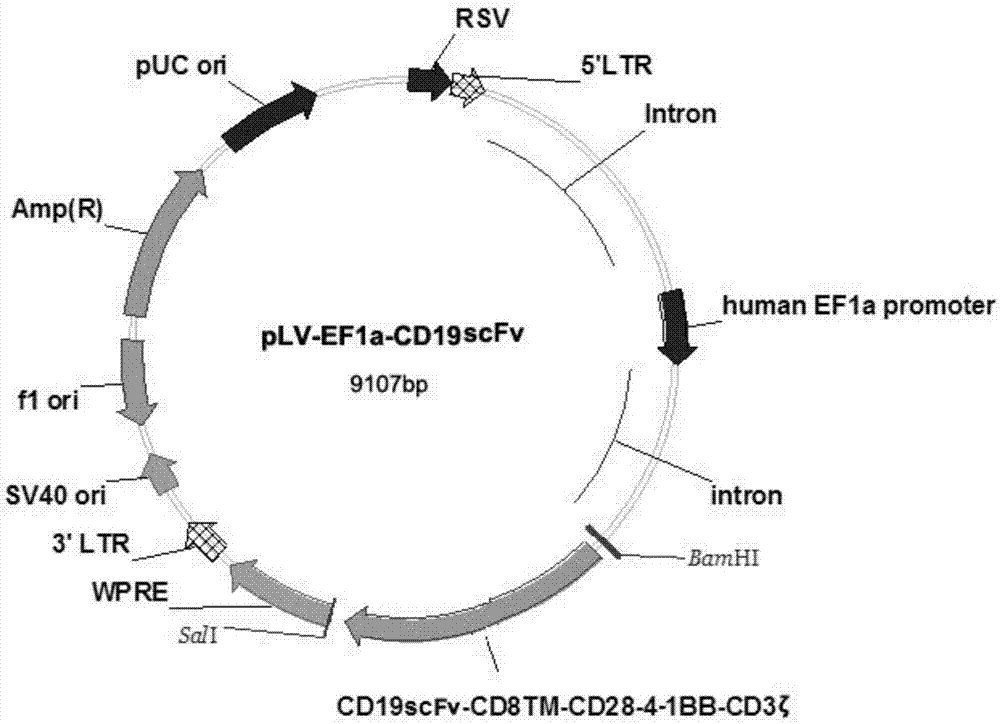
CD19-specific chimeric T cell receptor
InactiveUS7446179B2Peptide/protein ingredientsAntibody mimetics/scaffoldsIntracellular signallingTransmembrane domain
The present invention relates to a genetically engineered, CD19-specific chimeric T cell receptor and to immune cells expressing the chimeric receptor The present invention also relates to the use of such cells for cellular immunotherapy of CD9+ malignancies and for abrogating any untoward B cell function. The chimeric receptor is a single chain scFvFc:ζ receptor where scFvFc designates the extracellular domain, scFv designates the VH and VL chains of a single chain monoclonal antibody to CD19, Fc represents at least part of a constant region of an IgG1, and ζ represents the intracellular signaling domain of the zeta chain of human CD3. The extracellular domain scFvFc and the intracellular domain ζ are linked by a transmembrane domain such as the transmembrane domain of CD4. In one aspect, the chimeric receptor comprises amino acids 23-634 of SEQ I DNO:2. The present invention further relates to a method of making a redirected T cell expressing a chimeric T cell receptor by electroporation using naked DNA encoding the receptor.
Owner:CITY OF HOPE
Chimeric antigen receptor hCD19scFv-CD8a-CD-28-CD3zata and application thereof
ActiveCN104788573AConfirmed specific killing effectMammal material medical ingredientsHybrid peptidesAntigen receptorHeavy chain
The invention discloses a chimeric antigen receptor hCD19scFv-CD8a-CD-28-CD3zata and application thereof. The chimeric antigen receptor is serially connected by an anti-human CD19 monoclonal antibody H119a light chain and heavy chain variable region (hCD19scFv), human CD8a hinge region, a human CD28 transmembrane region, an intracellular region and a human CD3zata intracellular region. The chimeric antigen receptor is used for modifying T lymphocyte, and the modified T lymphocyte (CAR-T cell) can be used for treating the tumor with positive surface CD19.
Owner:JUVENTAS CELL THERAPY LTD
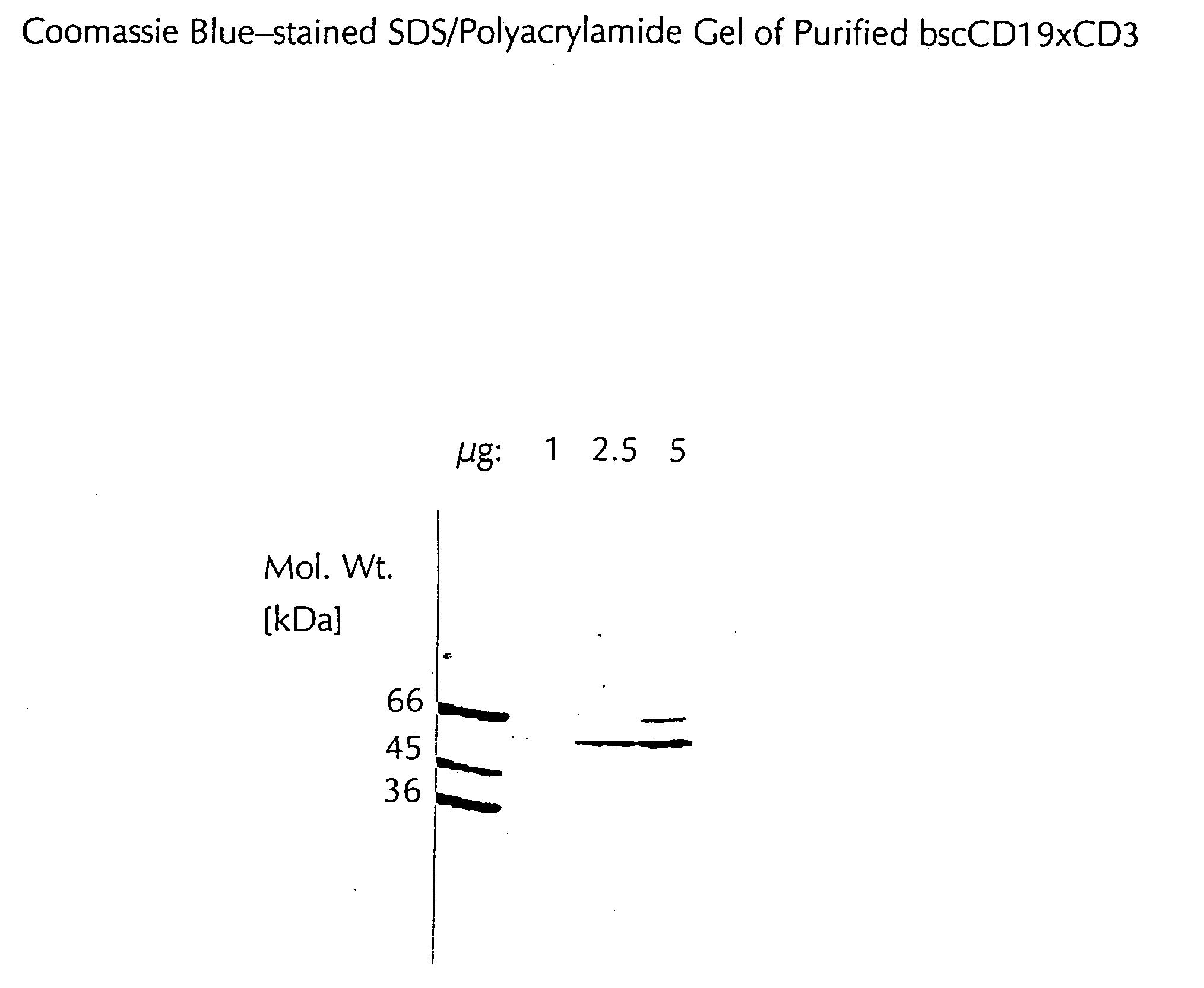
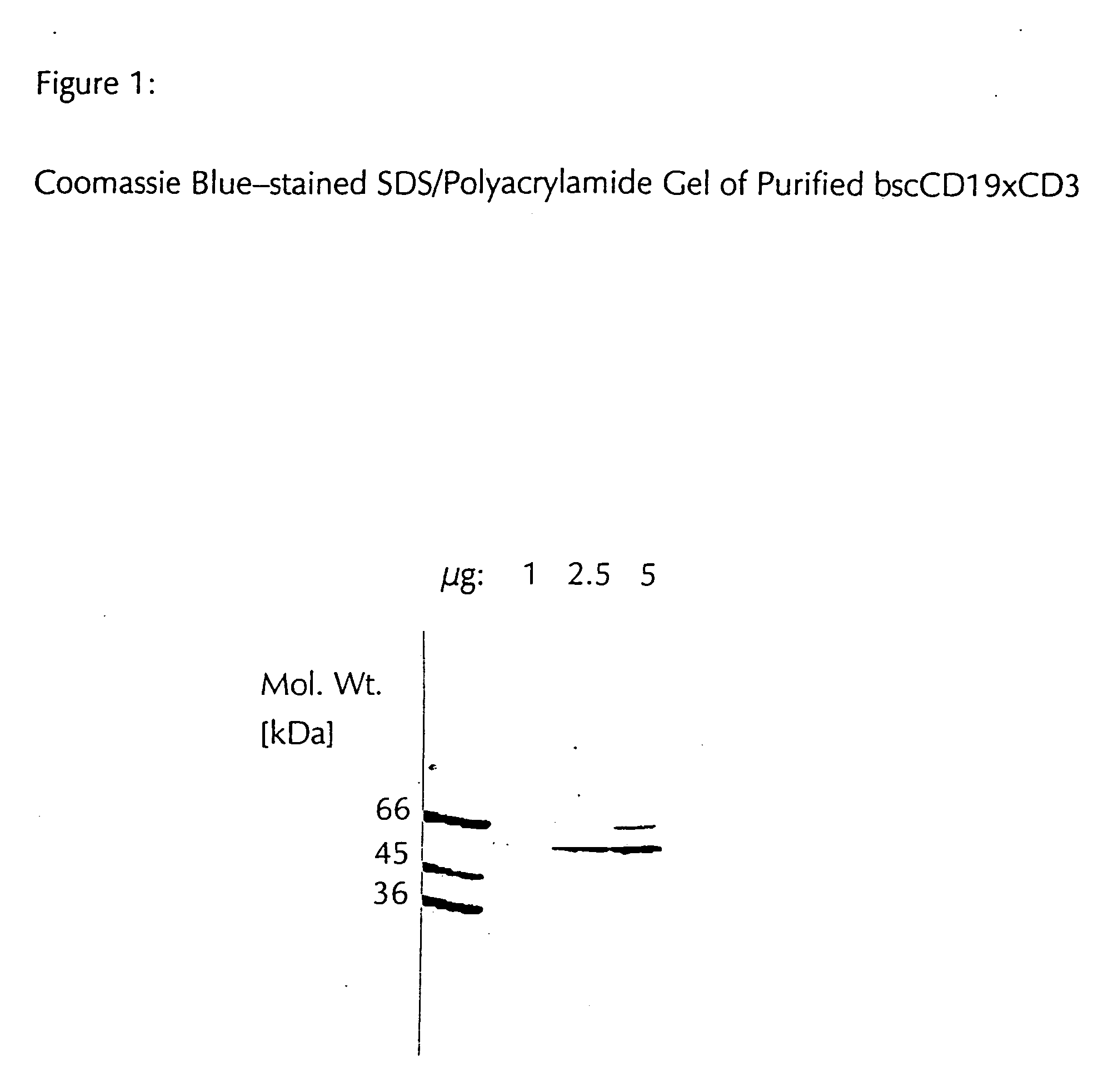
CD19xCD3 specific polypeptides and uses thereof
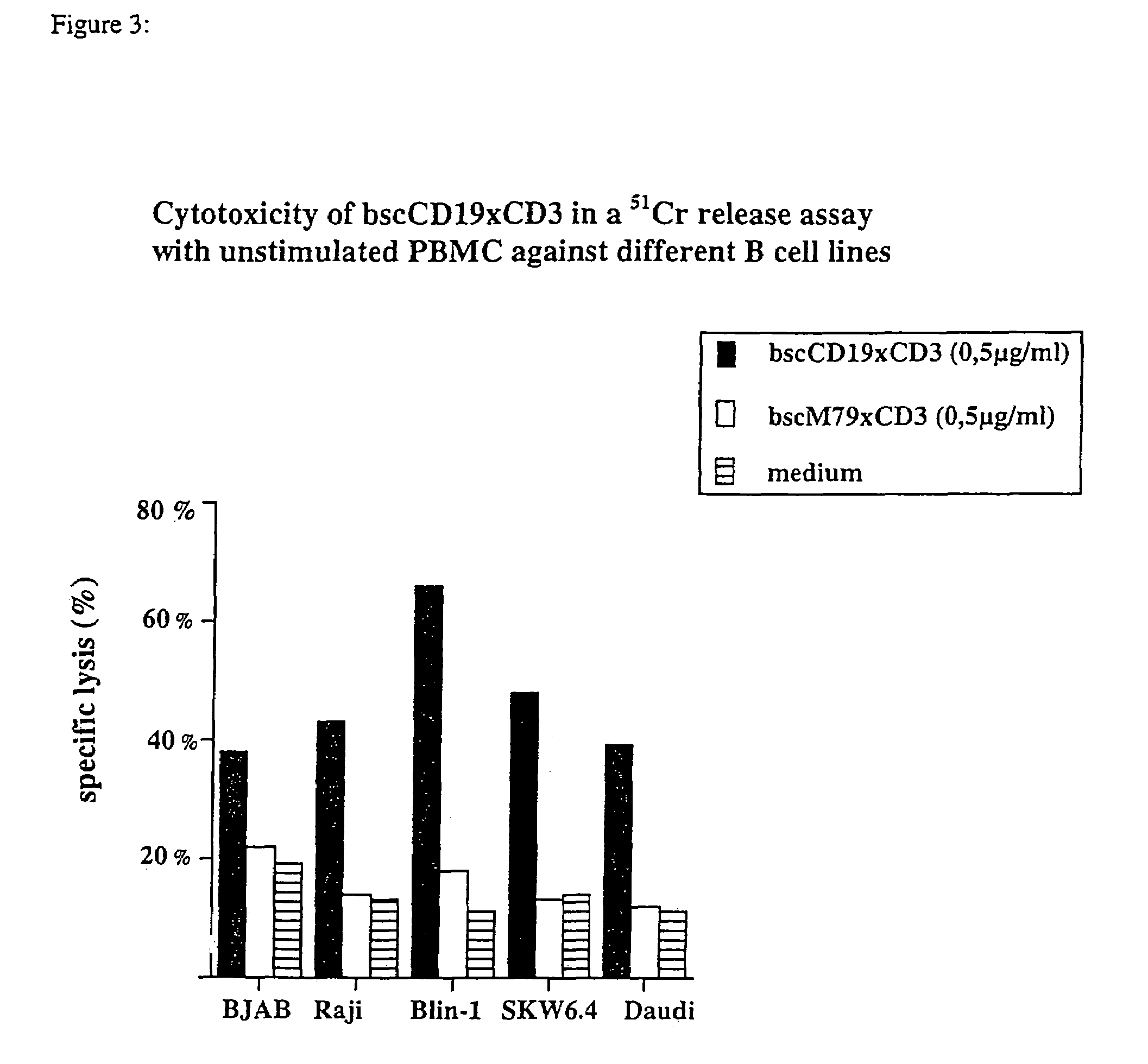
Described are novel single-chain multifunctional polypeptides comprising at least two binding sites specific for the CD19 and CD3 antigen, respectively. Further provided are polypeptides, wherein the above-described polypeptide comprises at least one further domain, preferably of pre-determined function. Furthermore, polynucleotides encoding said polypeptides as well as to vectors comprising said polynucleotides and host cells transformed therewith and their use in the production of said polypeptides are described. In addition, compositions, preferably pharmaceutical and diagnostic compositions are provided comprising any of the afore-described polypeptides, polynucleotides or vectors. Described is also the use of the afore-mentioned polypeptides, polynucleotides and vectors for the preparation of pharmaceutical compositions for immunotherapy, preferably against B-cell malignancies such as non-Hodgkin lymphoma.
Owner:AMGEN RES (MUNICH) GMBH
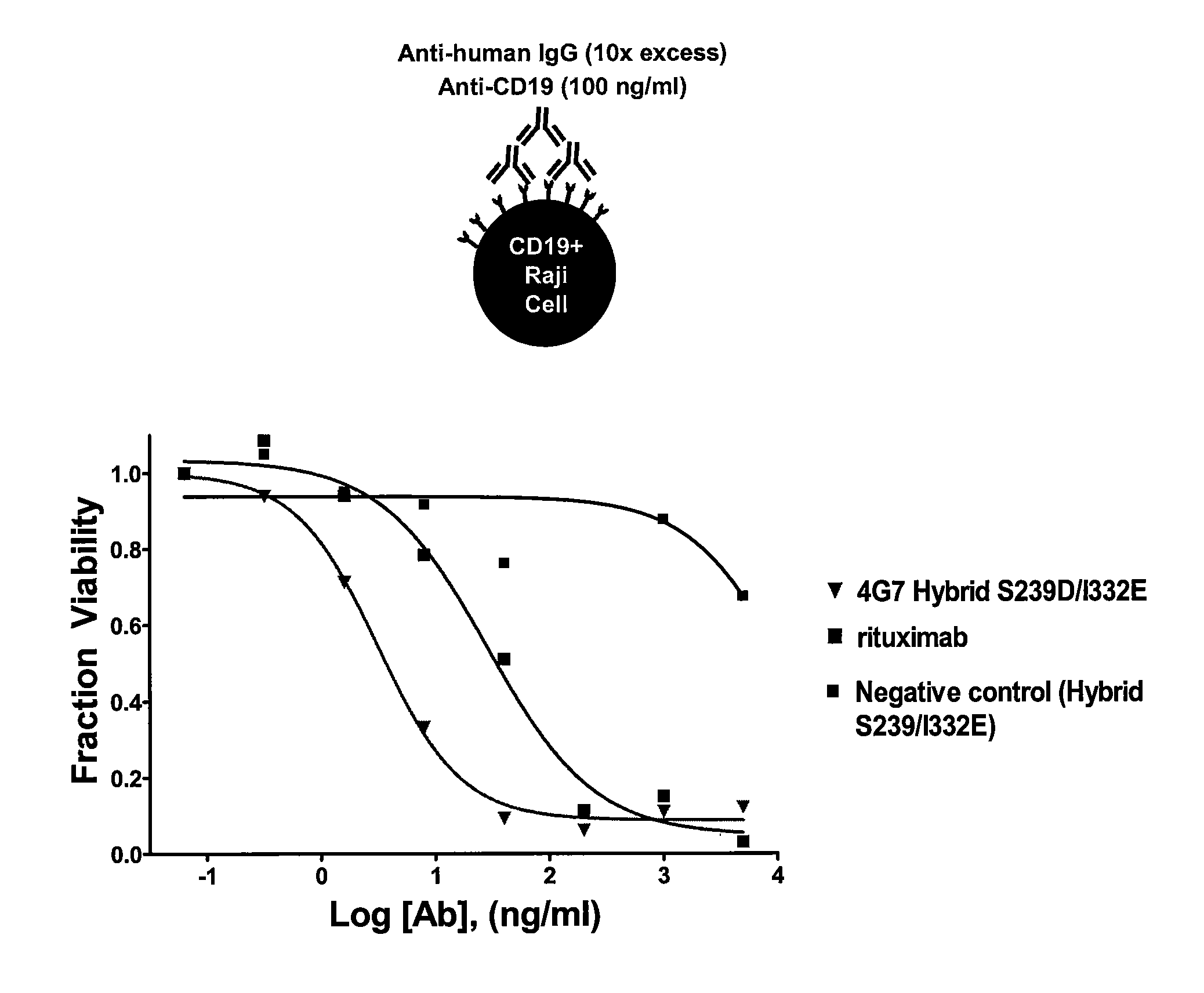
Optimized antibodies that target cd19
The present invention describes antibodies that target CD19, wherein the antibodies comprise at least one modification relative to a parent antibody, wherein the modification alters affinity to an FcγR or alters effector function as compared to the parent antibody. Also disclosed are methods of using the antibodies of the invention.
Owner:XENCOR INC
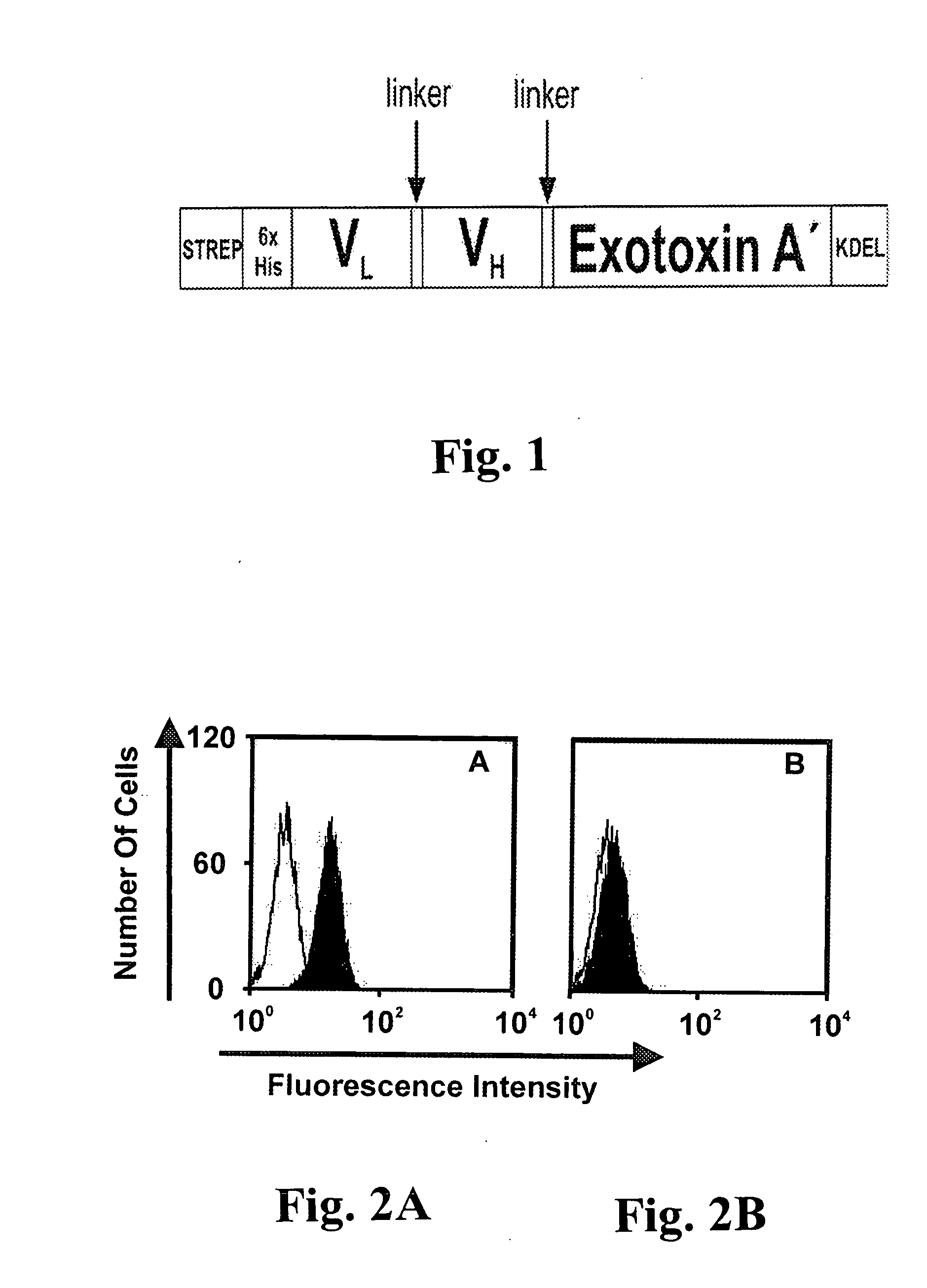
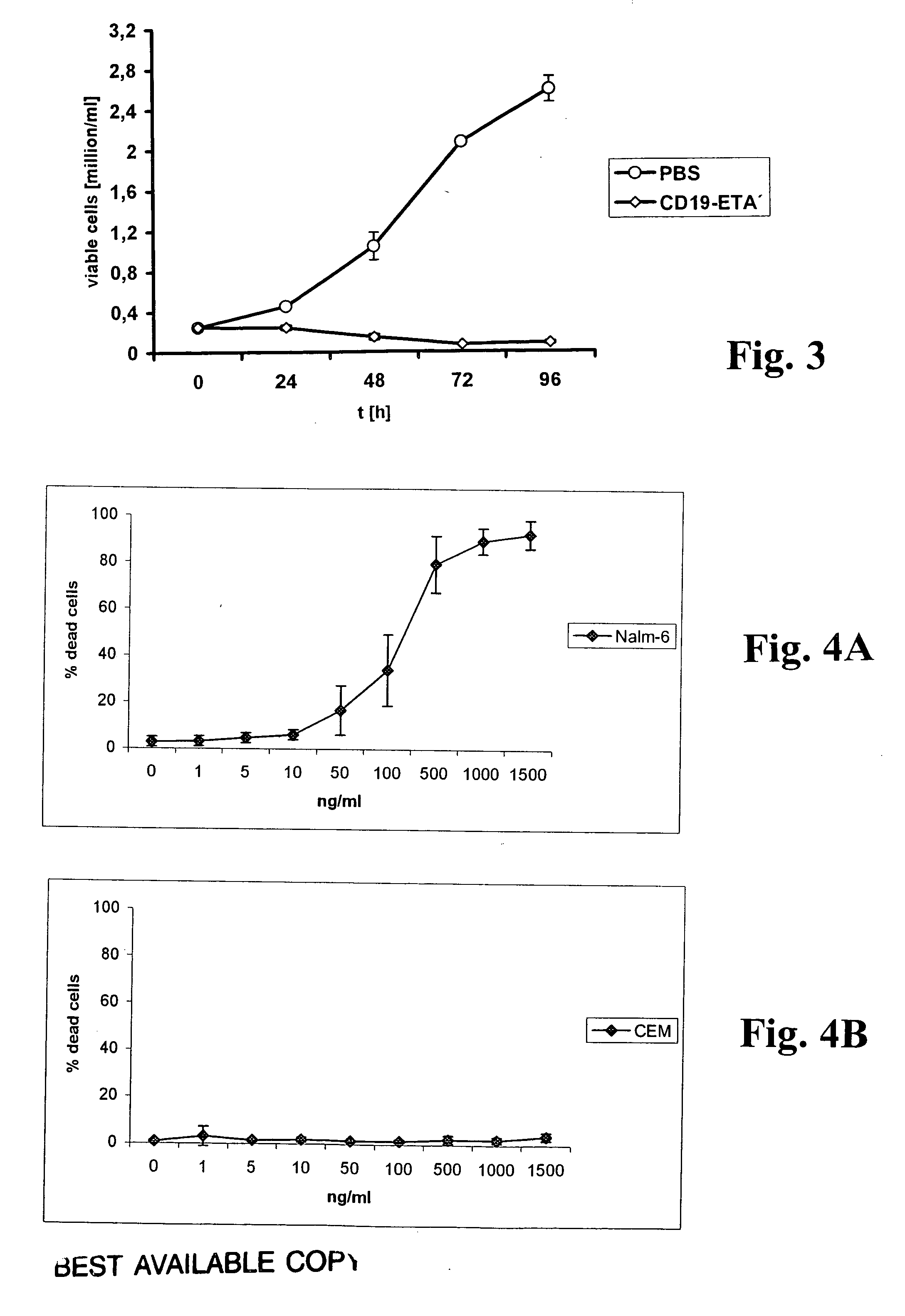
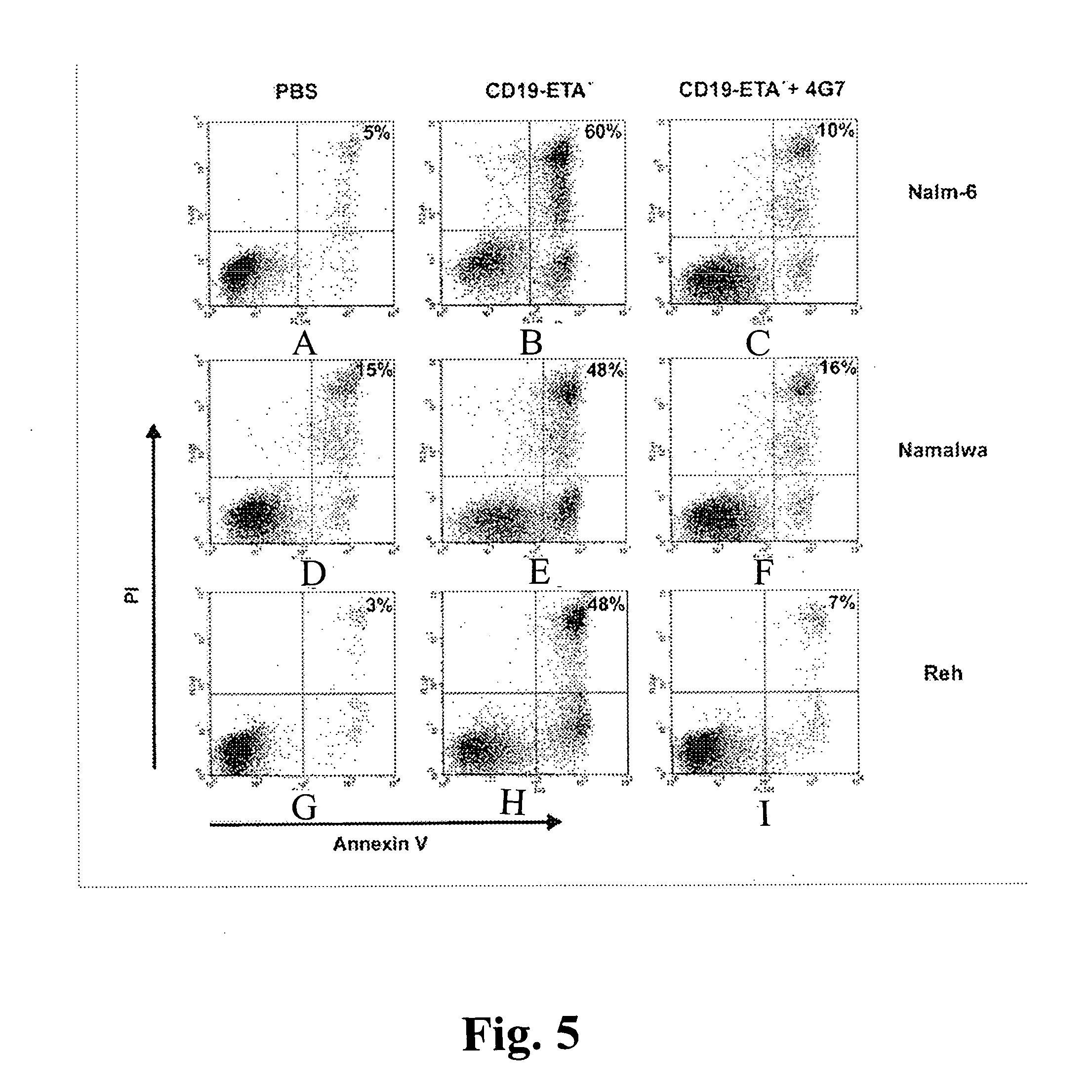
CD19-specific immunotoxin and treatment method
InactiveUS20070178103A1Easy to transportResistant to extracellular cleavagePolypeptide with localisation/targeting motifHybrid immunoglobulinsAutoimmune conditionBacterial exotoxin
An immunotoxin for use in, and a method for treating a subject having a cancer associated with malignant B-lineage cells or an autoimmune condition, are disclosed. The immunotoxin includes (a) an anti-CD19 antibody lacking an Fc fragment, (b) a modified exotoxin A protein having both Domains II and III, but lacking Domain I, and (c) a peptide linker joining the C-terminal end of the antibody to the N-terminal end of the modified exotoxin A protein. The linker is substantially resistant to extracellular cleavage. The modified exotoxin A protein may be further modified to include a C-terminal KDEL sequence (SEQ ID NO: 6) that promotes transport of the protein to the endoplasmic reticulum of cells that have taken up the immunotoxin.
Owner:FRIEDRICH ALEXANDER UNIV ERLANGEN NURNBERG
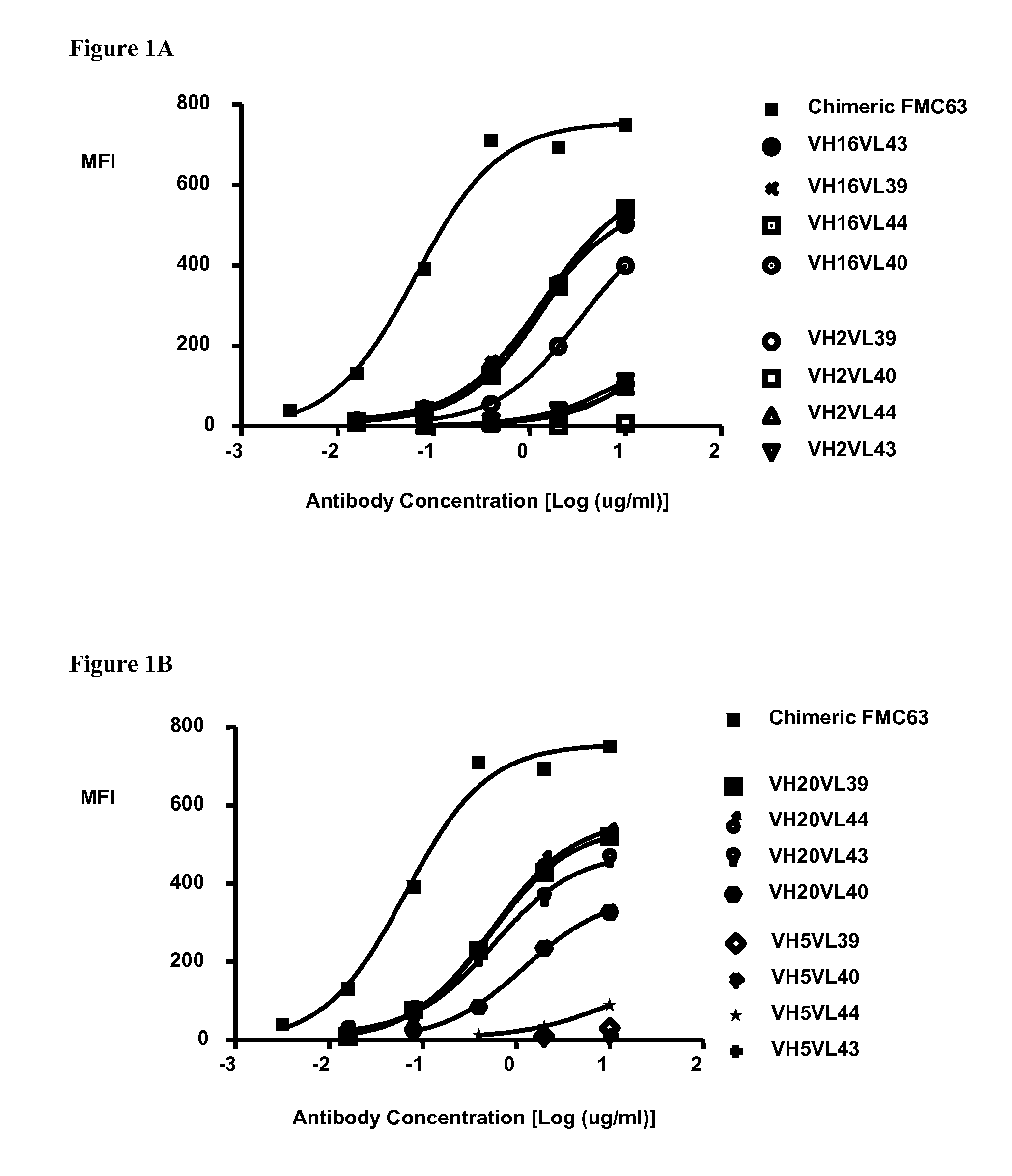
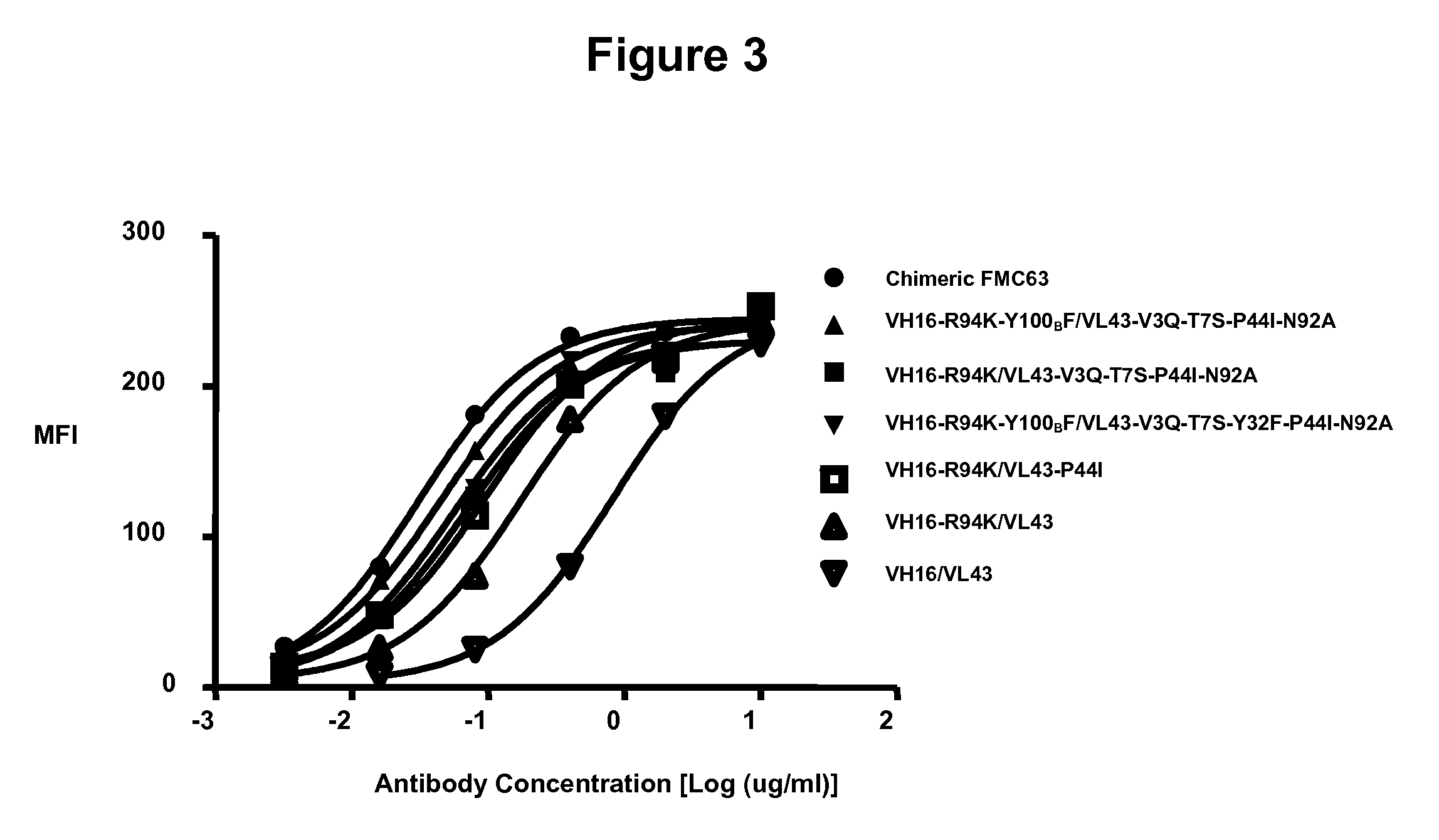
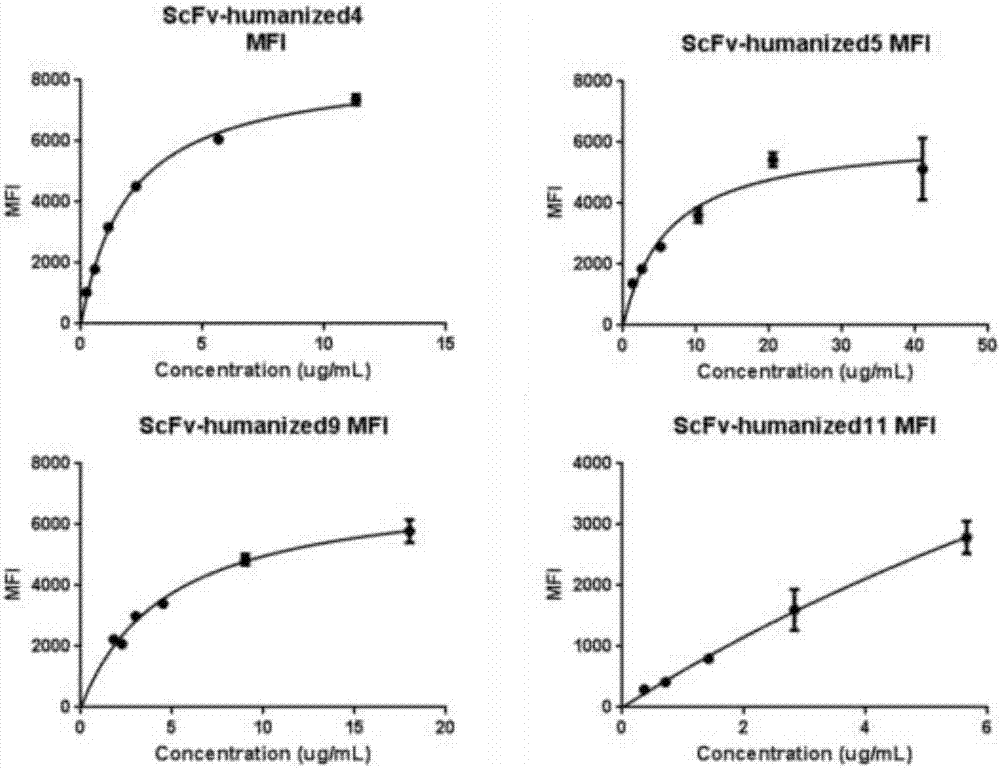
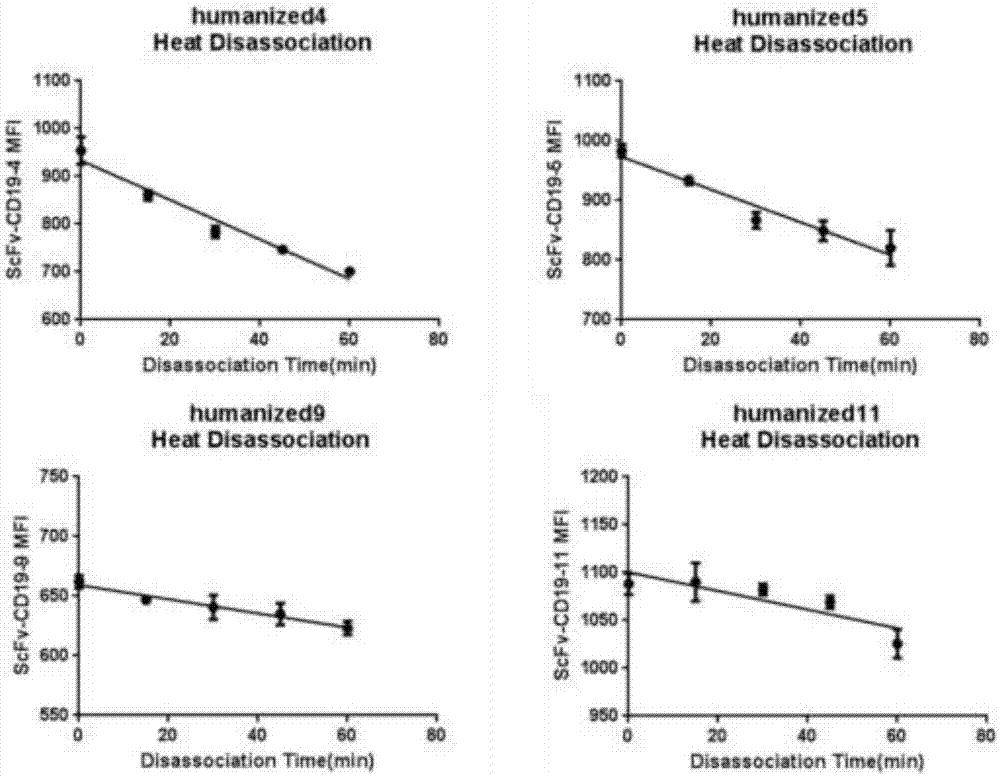
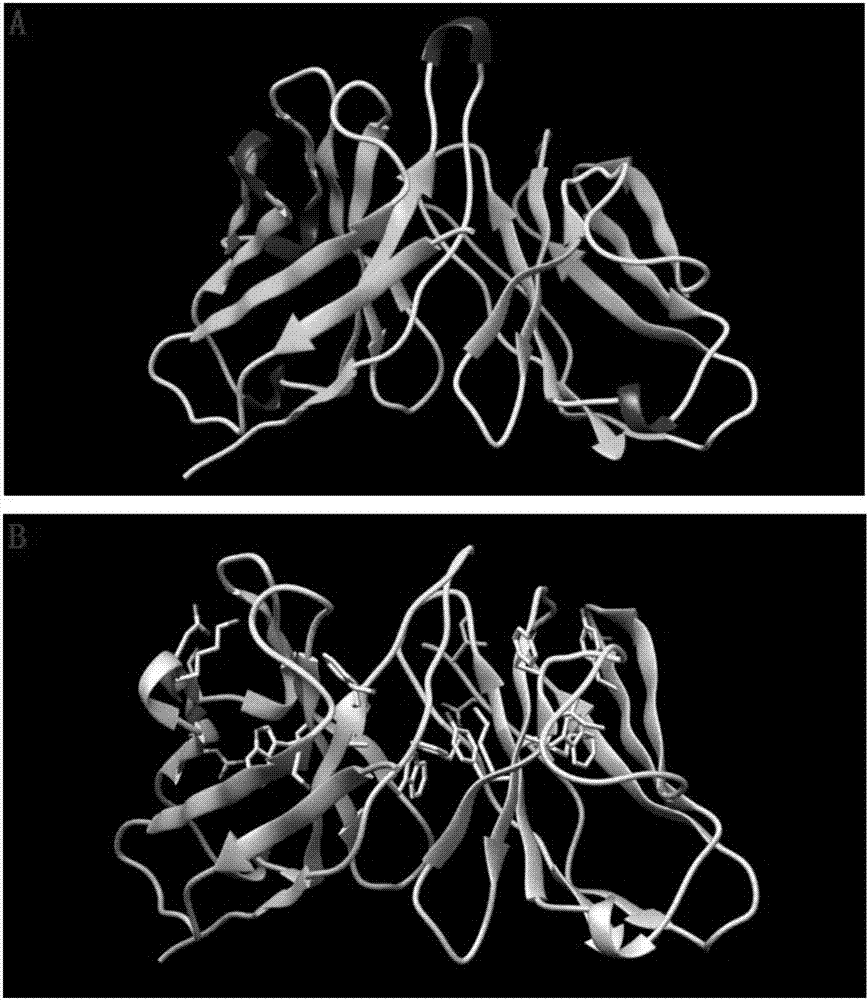
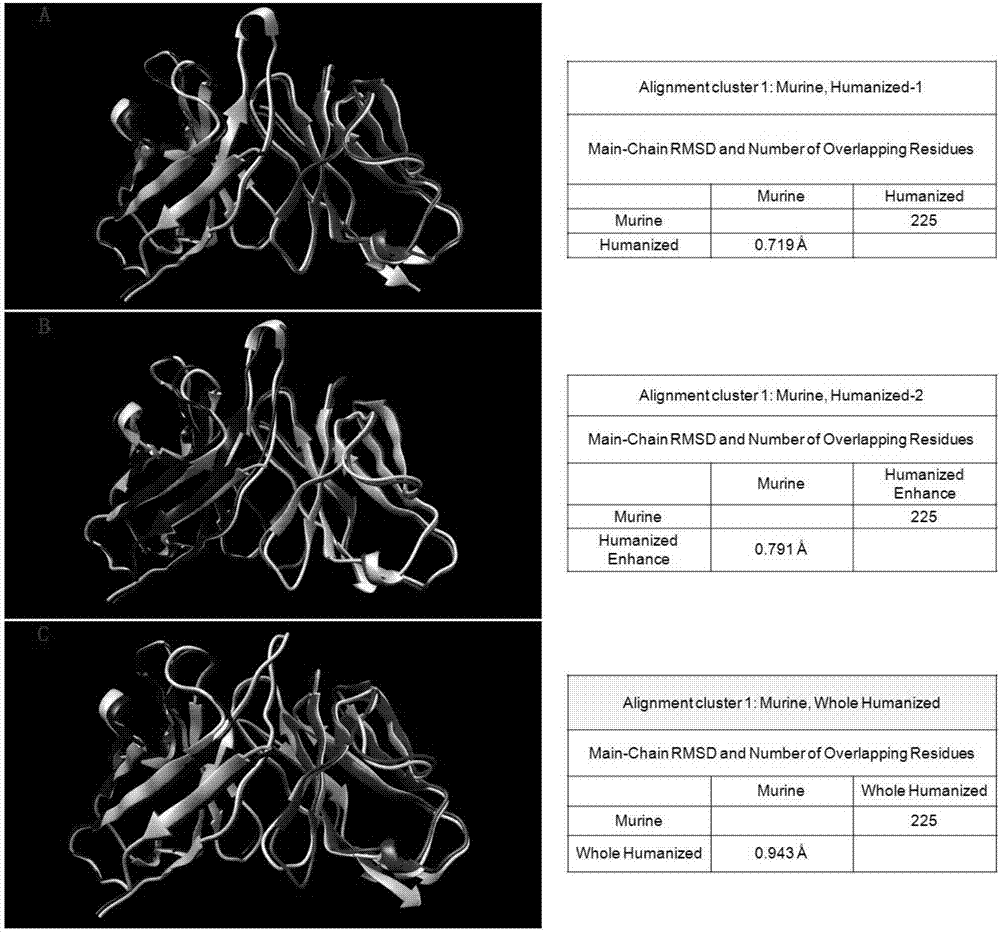
Humanized antibodies that bind to CD19 and their uses
InactiveUS20100215651A1Improved complement dependent cytotoxicityAntipyreticAnalgesicsHeavy chainHumanized antibody
The present invention relates to humanized antibodies or fragments thereof that bind to human CD19. More specifically, the present invention relates to a humanized antibody or fragment thereof that binds to human CD19 comprising a heavy chain CDR1 comprising the amino acid sequence of SEQ ID NO: 27, and / or a heavy chain CDR2 comprising the amino acid sequence of SEQ ID NO: 28, and / or a heavy chain CDR3 comprising the amino acid sequence of SEQ ID NO: 29; and / or comprising a light chain CDR1 comprising the amino acid sequence of SEQ ID NO: 30, and / or a light chain CDR2 comprising the amino acid sequence of SEQ ID NO: 31 and / or a light chain CDR3 comprising the amino acid sequence of SEQ ID NO: 32.
Owner:ICHNOS SCI SA
CD19 targeting chimeric antigen receptor and NKT cell, and preparation method thereof and applications thereof
ActiveCN105418765AEnhance specific killing activityProlong survival timePeptide/protein ingredientsGenetic material ingredientsAntigenHinge region
The present invention discloses a chimeric antigen receptor, a gene and a recombinant expression vector thereof, an engineered CD19 targeting NKT cell and applications thereof. The chimeric antigen receptor is CD19ScFv-CD8-CD137-CD3 zeta, and consists of a hinge region and a transmembrane region of the CD19ScFv and CD8, an intracellular signal structural domain of CD137, and an intracellular signal structural domain of CD3 zeta, and the the hinge region, the transmembrane region, the intracellular signal structural domain of CD137 and the intracellular signal structural domain of CD3 zeta are connected in series. When used to treat advanced stage CD19-positive B-cell acute lymphocytic leukemia, the NKT cell modified by the chimeric antigen receptor CD19ScFv-CD8-CD137-CD3 zeta provided by the present invention has good specific killing activity on leukemic cells, and has certain therapeutic effect on advanced stage CD19-positive B-cell acute lymphocytic leukemia patients who are repeatedly subjected to therapy such as radiotherapy, chemotherapy and symptomatic therapy by other drugs but cannot recover obviously.
Owner:CELLULAR BIOMEDICINE GRP SHANGHAI +1
Chimeric antigen receptor of anti-human CD19 antigen and application thereof
ActiveCN107226867ANo side effectsIncreased proliferationPolypeptide with localisation/targeting motifImmunoglobulin superfamilyCancer cellSide effect
The invention belongs to the field of gene engineering, and relates to chimeric antigen receptor of anti-human CD19 antigen and application thereof. The chimeric antigen receptor of anti-human CD19 antigen comprises polypeptide (scFv) for recognizing human CD19 antigen, a hinge region, a transmembrane region and an intracellular signal domain which are connected in sequence, wherein an amino acid sequence of the polypeptide (scFv) for recognizing human CD19 antigen is as shown in SEQ ID NO.1, SEQ ID NO.2, SEQ ID NO.3 or SEQ ID NO.4. The chimeric antigen receptor can be more stably expressed in T lymphocyte, has the capacity for properly removing cancer cell, can not only maintain the positive rate of CD19 targeted chimeric antigen receptor during the cell culture of a patient, but also enhance the capacity for CAR-T multiplication and tumor killing, does not have toxic and / or side effects on antigen-negative cell, and can be applied to the targeted therapy of tumor.
Owner:CHONGQING PRECISION BIOTECH CO LTD
Novel CD19xCD3 specific polypeptides and uses thereof
Described are novel single-chain multifunctional polypeptides comprising at least two binding sites specific for the CD19 and CD3 antigen, respectively. Further provided are polypeptides, wherein the above-described polypeptide comprises at least one further domain, preferably of pre-determined function. Furthermore, polynucleotides encoding said polypeptides as well as to vectors comprising said polynucleotides and host cells transformed therewith and their use in the production of said polypeptides are described. In addition, compositions, preferably pharmaceutical and diagnostic compositions are provided comprising any of the afore-described polypeptides, polynucleotides or vectors. Described is also the use of the afore-mentioned polypeptides, polynucleotides and vectors for the preparation of pharmaceutical compositions for immunotherapy, preferably against B-cell malignancies such as non-Hodgkin lymphoma.
Owner:AMGEN RES (MUNICH) GMBH
Pharmaceutical compositions comprising bispecific anti-cd3, anti-cd19 antibody constructs for the treatment of b-cell related disorders
The present invention relates to a pharmaceutical composition comprising a bispecific single chain antibody construct comprising human CD3 and human CD19 specific binding domains, wherein the corresponding variable heavy chain regions (VH) and the corresponding variable light chain region (VL) from N-terminus to C-terminus in the following order: VH (CD19)-VL (CD19)-VH (CD3)-VL (CD3), VH (CD3)-VL (CD3)-VH (CD19)-VL (CD19) or VH (CD3)-VL (CD3)-VL (CD19)-VH (CD19). In addition, the present invention discloses the preparation method of the pharmaceutical composition and the medical / pharmaceutical use of the specific bispecific single chain antibody molecule specific to human CD3 antigen and human CD19 antigen.
Owner:AMGEN RES (MUNICH) GMBH
Treatment of cancer using humanized Anti-cd19 chimeric antigen receptor
ActiveUS20190135940A1Good curative effectPolypeptide with localisation/targeting motifImmunoglobulin superfamilyBinding domainCancer research
The invention provides compositions and methods for treating diseases associated with expression of CD19. The invention also relates to chimeric antigen receptor (CAR) specific to CD19, vectors encoding the same, and recombinant T cells comprising the CD19 CAR. The invention also includes methods of administering a genetically modified T cell expressing a CAR that comprises a CD19 binding domain.
Owner:NOVARTIS AG +1
Humanized monoclonal antibody targeting to human CD19 antigen
ActiveCN107312091AEfficient killingNo side effectsImmunoglobulins against cell receptors/antigens/surface-determinantsMammal material medical ingredientsSingle-Chain AntibodiesTreatment effect
The invention discloses a humanized monoclonal antibody targeting to human CD19 antigen. The humanized monoclonal antibody is applied to construction and preparation of chimeric antigen receptor T cells; and subsequent in-vitro experiments verify the tumor treatment effect of the chimeric antigen receptor T cells. A chimeric antigen receptor targeting to the human CD19 antigen is composed of a leader peptide, a single-chain antibody targeting to the human CD19 antigen, a hinge area for connection with ScFv, a transmembrane domain and an immune receptor tyrosine activation motif; a lentiviral expression vector is constructed from the chimeric antigen receptor and then used for infection of immune cells, so the chimeric antigen receptor targeting to the human CD19 antigen can be expressed in the immune cells; and tumors expressing the CD19 antigen are killed by recognizing the CD19 antigen on the surfaces of tumor cells, so the purpose of tumor treatment is achieved. The invention provides a novel tool for targeting therapy of tumors.
Owner:CHONGQING PRECISION BIOTECH CO LTD
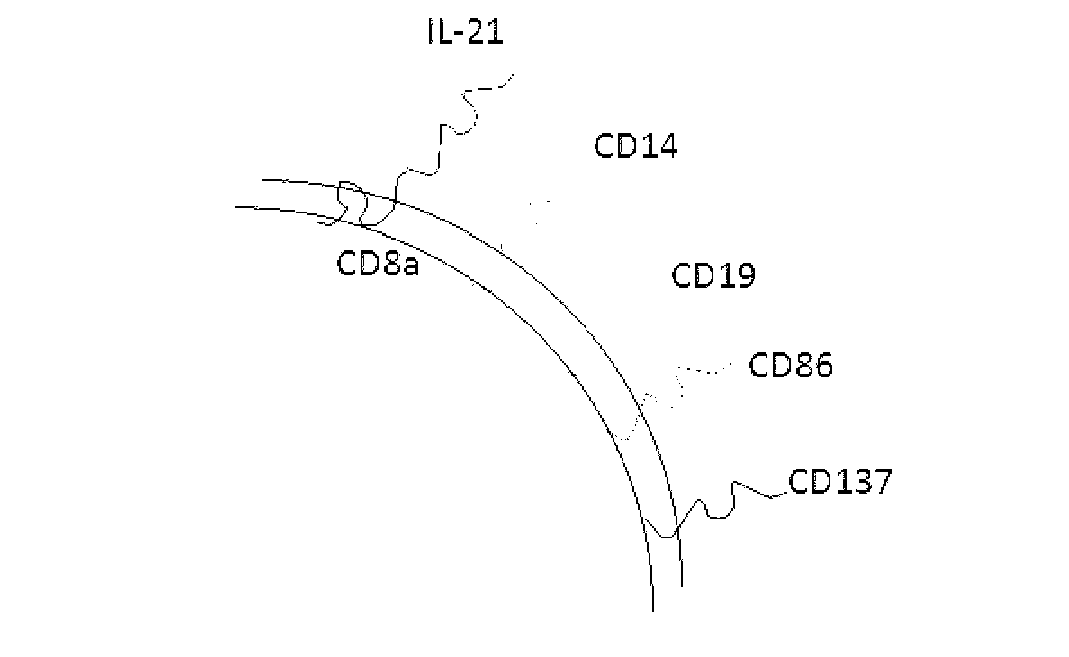
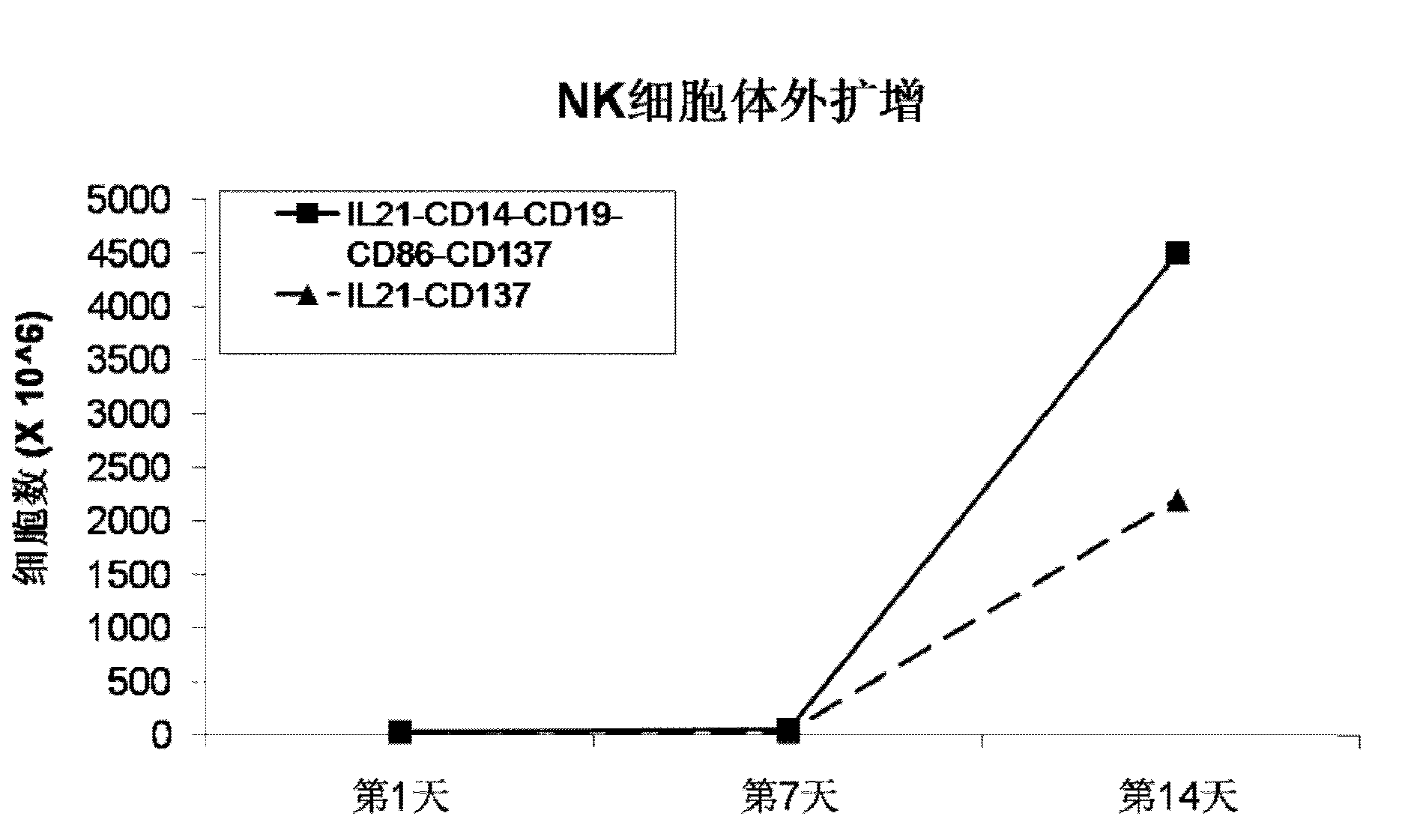
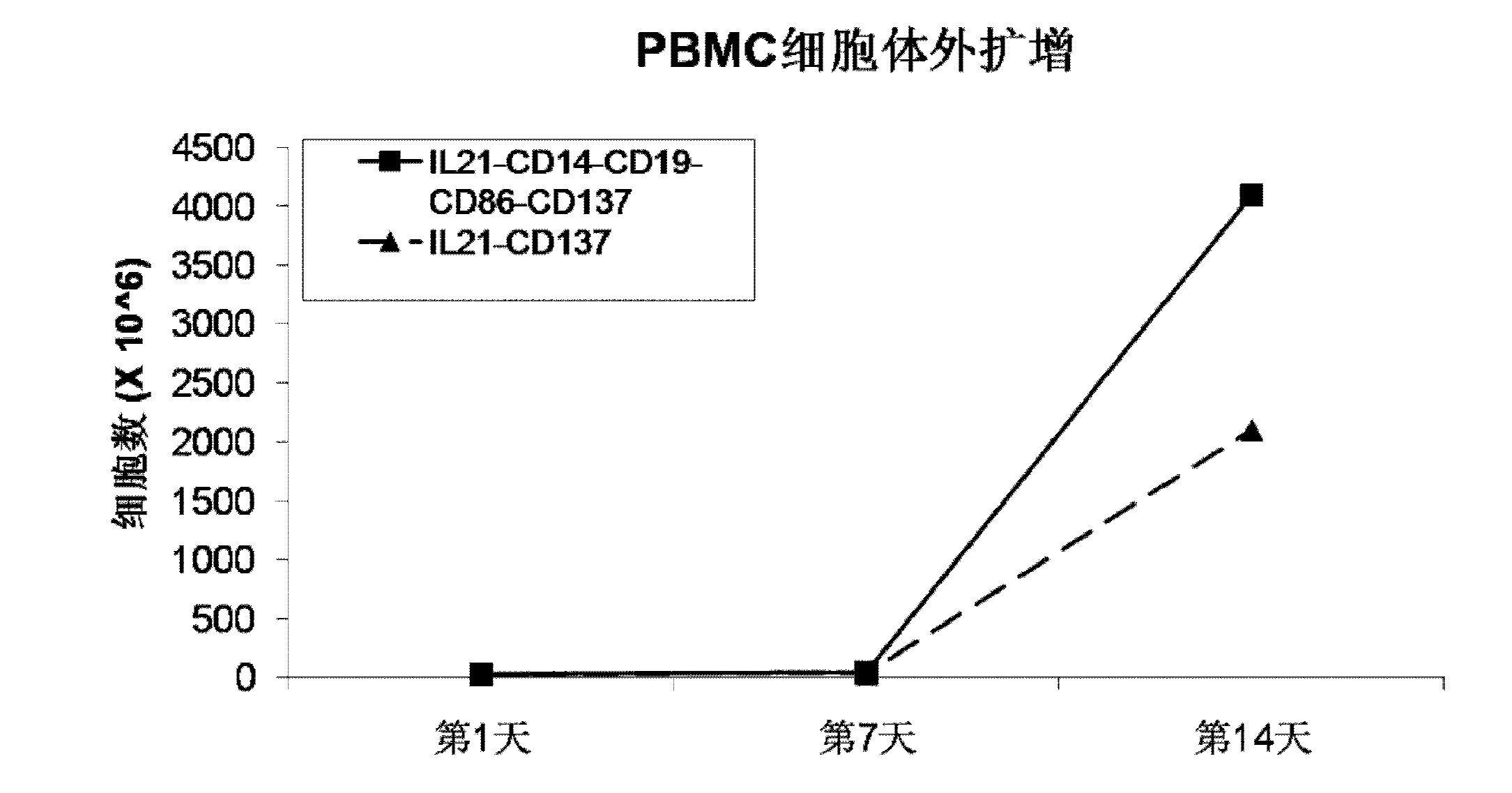
Method for amplification and activation of NK cells by K562 cells
ActiveCN103232973AHelp identify and killHelp activate recognition and killMammal material medical ingredientsBlood/immune system cellsNatural Killer Cell Inhibitory ReceptorsCD86
The invention discloses a method for amplification and activation of NK cells by K562 cells. The method comprises that through synergism of K562 cells transfected by transmembrane interleukin 21, CD14, CD19, CD86 and CD137, and low-concentration interleukin 2, NK cells are subjected to directed amplification and activation. Compared with the existing similar compounds, the compound provided by the invention has a stronger lymphocyte amplification and activation capability and higher efficiency. The method has wide prospects in immunological therapy.
Owner:杭州中赢生物医疗科技有限公司
Treatment of cancer using Anti-cd19 chimeric antigen receptor
PendingUS20190388471A1Enhances activity of CAR-expressingGood curative effectPolypeptide with localisation/targeting motifImmunoglobulin superfamilyDiseaseEnzyme Inhibitor Agent
The invention provides compositions and methods for treating diseases associated with expression of CD19, e.g., by administering a recombinant T cell comprising the CD19 CAR as described herein, in combination with a kinase inhibitor, e.g., a kinase inhibitor described herein. The invention also provides kits and compositions described herein.
Owner:THE TRUSTEES OF THE UNIV OF PENNSYLVANIA +1
CD19 targeted CAR (chimeric antigen receptor)-T cell, preparation method and application
InactiveCN107287164AIncrease lethalityHigh transduction efficiencyGenetically modified cellsMammal material medical ingredientsCAR T-cell therapySingle-Chain Antibodies
The invention provides a CD19 targeted CAR (chimeric antigen receptor)-T cell, a preparation method and an application and relates to the field of immune cells. The activation capacity of T cells in CAR-T cell therapy can be improved, the problem of insufficient transfection efficiency can be solved, and the CAR-T cell has a high killing capacity for CD19. The CD19 targeted CAR-T cell expresses a CD19 targeted CAR gene on surface, and the CD19 targeted CAR gene is formed by connecting a single-chain antibody CD19ScFv of CD19, a hinge region and a transmembrane region of CD8, an intracellular signal structure of CD28, an intracellular signal structure of 4-1BB and an intracellular signal structural domain of CD3zeta in series, and the base sequence of the CD19 targeted CAR gene is shown as SEQ ID NO:2.
Owner:青岛见康华美医学检验有限公司
Anti-CD19 full humanized antibody or antibody segment as well as method and application thereof
ActiveCN107880128ATo achieve the purpose of healingPolypeptide with localisation/targeting motifImmunoglobulin superfamilyComplementarity determining regionHeavy chain
The invention discloses an anti-CD19 full humanized antibody or antibody segment as well as a method and an application thereof. The antibody or the antibody segment contains a heavy chain and a lightchain, wherein the heavy chain and the light chain comprise variable regions; the variable regions comprise complementary determining regions; the complementary determining regions CDR1, CDR2 and CDR3 of the heavy chain are separately represented by HCDR1, HCDR2 and HCDR3; the complementary determining regions CDR1, CDR2 and CDR3 of the light chain are separately represented by LCDR1, LCDR2 and LCDR3; the amino acid sequence of the HCDR1 comprises SED ID NO: 3; the amino acid sequence of the HCDR2 comprises SEQ ID NO: 4; the amino acid sequence of the HCDR3 comprises SEQ ID NO: 5; the amino acid sequence of the LCDR1 comprises SEQ ID NO: 6; and the amino acid sequence of the LCDR2 comprises SEQ ID NO: 7. The antibody or the antibody segment can be combined with a humanized CD19 protein with high specificity, and by means of engineered expression integration by means of a chimeric antigen receptor cell technology in a cell T, the obtained chimeric antigen receptor cell T can be used for treating hematological cancers related to expression of CD19.
Owner:CHANGZHOU VELOX PHARMA SCI & TECH CO LTD
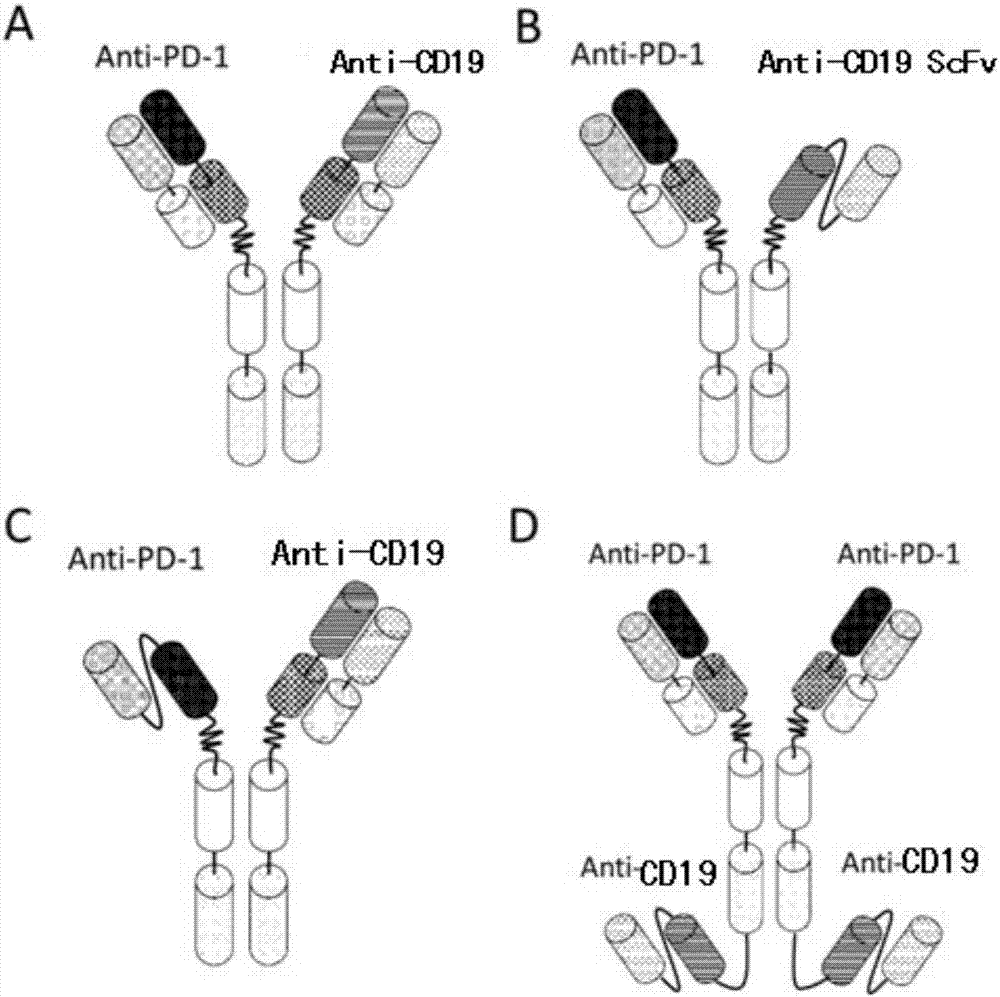
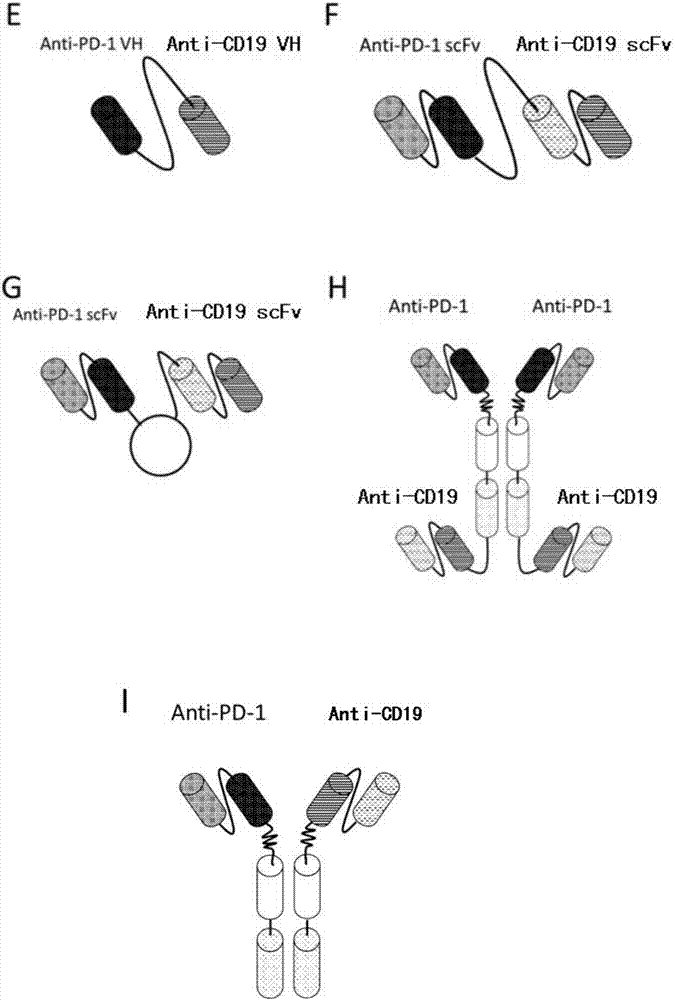
Anti-PD1 and anti-CD19 bispecific antibody and applications thereof
ActiveCN106939050AImprove biological activityGood biological stabilityHybrid immunoglobulinsAntibody ingredientsCell Surface AntigensTherapeutic effect
The present invention relates to anti-PD1 and anti-CD19 bispecific antibody and applications thereof, wherein the anti-PD1 and anti-CD19 bispecific antibody, or a variant, or a functional fragment thereof comprises: a domain capable of specifically recognizing and binding an immune cell surface antigen PD-1, wherein the domain comprises the heavy chain variable region (anti-PD-1 VH) of an anti-PD-1 specific antibody; and a domain capable of specifically recognizing and binding CD19, wherein the domain comprises the heavy chain variable region (anti-CD19 VH) of an anti-CD19 specific antibody. According to the present invention, the bispecific antibody has the whole human sequence, such that the occurrence of HAMA in clinical treatment application is effectively avoided, the occurrence of drug resistance can be effectively reduced, and the utilization efficiency and the treatment effect of the medicine can be improved; and the anti-PD-1 and anti-CD19 bispecific antibody can well mediate T cells to efficiently and specifically kill B-lymphocyte derived tumor cells compared to the existing anti-PD-1 specific antibody and the existing anti-CD19 specific antibody.
Owner:SHUNHAO CELL BIOTECHNOLOGY (TIANJIN) CO LTD
Humanized antibodies that bind to CD19 and their uses
The present invention relates to humanized antibodies or fragments thereof that bind to human CD19. More specifically, the present invention relates to a humanized antibody or fragment thereof that binds to human CD19 comprising a heavy chain CDR1 comprising the amino acid sequence of SEQ ID NO: 27, and / or a heavy chain CDR2 comprising the amino acid sequence of SEQ ID NO: 28, and / or a heavy chain CDR3 comprising the amino acid sequence of SEQ ID NO: 29; and / or comprising a light chain CDR1 comprising the amino acid sequence of SEQ ID NO: 30, and / or a light chain CDR2 comprising the amino acid sequence of SEQ ID NO: 31 and / or a light chain CDR3 comprising the amino acid sequence of SEQ ID NO: 32.
Owner:ICHNOS SCI SA
Anti-CD19 monoclonal antibody and preparation method thereof
ActiveCN105837689AEfficient identificationHigh affinityImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsProtein targetWestern blot
The invention provides an anti-CD19 monoclonal antibody, and a preparation method and an application thereof. The anti-CD19 monoclonal antibody can effectively recognize cells with CD19 highly expressed, and can be successfully used for Western Blot detection. An affinity test shows that the monoclonal antibody has the extremely high affinity to target protein.
Owner:PERSONGEN ANKE CELLULAR THERAPEUTICS CO LTD
Treatment of cancer using humanized anti-CD19 chimeric antigen receptor
ActiveUS10221245B2Good curative effectPolypeptide with localisation/targeting motifImmunoglobulin superfamilyCD19-specific chimeric antigen receptorAntigen receptors
The invention provides compositions and methods for treating diseases associated with expression of CD19. The invention also relates to chimeric antigen receptor (CAR) specific to CD19, vectors encoding the same, and recombinant T cells comprising the CD19 CAR. The invention also includes methods of administering a genetically modified T cell expressing a CAR that comprises a CD19 binding domain.
Owner:THE TRUSTEES OF THE UNIV OF PENNSYLVANIA
Anti-CD47 Antibodies and Methods of Use Thereof
ActiveUS20140303354A1High selectivityHigh affinityHybrid immunoglobulinsImmunoglobulins against cell receptors/antigens/surface-determinantsBinding siteBispecific monoclonal antibody
The invention relates to monoclonal and / or monovalent antibodies that bind CD47. The invention relates to monoclonal and / or monovalent antibodies that bind CD19. The invention also relates to novel bispecific monoclonal antibodies carrying a different specificity for each binding site of the immunoglobulin molecule, where one of the binding sites is specific for CD47. The invention also relates to novel bispecific monoclonal antibodies carrying a different specificity for each binding site of the immunoglobulin molecule, where one of the binding sites is specific for CD19.
Owner:NOVIMMUNE
CD19-based chimeric antigen receptor and application thereof
PendingCN108383914ALess prone to stormsGood killing effectPeptide/protein ingredientsAntibody mimetics/scaffoldsAbnormal tissue growthSide effect
The invention relates to a CD19-based chimeric antigen receptor and application thereof, in particular to a lentivirus vector material built by a chimeric antigen receptor T (CAR-T) cell technology using a tumor specific target point CD19 as the basis, a method, and application thereof to anti-tumor treatment. The chimeric antigen receptor is formed by serially connecting an antigen combination structure domain, a membrane spaning structure domain, a costimulatory signal conduction region, a CD3 zeta signal conduction structure domain and an inducible suicide fusion structure domain, wherein the antigen combination structure domain is combined with the tumor surface antigen; the tumor surface antigen is CD19. The chimeric antigen receptor is subjected to specific gene transformation on theT cell stimulation signals. Compared with other chimeric antigen receptors, the chimeric antigen receptor provided by the invention has a better reaction effect and higher safety, so that the CAR-T cells have higher immune effects and low side effects; the treatment effect and safety of the CAR-T cells are enhanced.
Owner:BEIJING MEIKANG JIMIAN BIOTECH CO LTD
Humanized anti-CD19 antigen binding fragment
ActiveCN107383196ALow immunogenicity in vivoLong life in vivoPolypeptide with localisation/targeting motifImmunoglobulin superfamilyDiseaseSingle-Chain Antibodies
The invention provides a composition and a method for treating CD19 expression related diseases. The invention also relates to an application of humanized anti-CD19 single chain antibody with low immunogenicity as a CD19 antigen binding fragment, which is used for constructing a chimeric antigen receptor (CAR) specific to CD19, a carrier for coding the chimeric antigen receptor, and a recombined T-cell containing anti-CD19CAR. The invention also comprises a method for applying a genetically modified T-cell which expresses CAR containing the humanized anti-CD19 antigen binding fragment.
Owner:GUANGZHOU BIO GENE TECH CO LTD
Construction and application of bispecific antibody CD19*CD3
ActiveCN104829726AIncrease lethalityIncreased immunotherapyHybrid immunoglobulinsImmunoglobulins against cell receptors/antigens/surface-determinantsBispecific antibodyTumor cells
The invention provides a bispecific antibody which is composed of a single-chain unit A and a single-chain unit B, wherein the single-chain unit A, aiming to surface antigen CD19 of immune cells, has a specifically combining capability while the single-chain unit B, aiming to surface antigen CD3 of tumor cells, has a specifically combining capability. The single-chain unit A and the single-chain unit B both include a single-chain variable fragment (ScFv) fused with an Fc fragment. The application also provides a preparation of the bispecific antibody and medicinal applications of these antibodies.
Owner:WUHAN YZY BIOPHARMA CO LTD
BCMA and CD19-targeted duplex-specific chimeric antigen receptor and application thereof
ActiveCN109485734AEnhance the kill signalAvoid target escapeMammal material medical ingredientsImmunoglobulinsTreatment effectSide effect
The invention relates to a BCMA and CD19-targeted duplex-specific chimeric antigen receptor and application thereof. The chimeric antigen receptor comprises an extracellular structural domain, a membrane spanning structural domain and at least one intracellular structural domain which can be bound with an antigen, wherein the extracellular structural domain comprises an anti-BCMA antigen structural domain and an anti-CD-19 antigen structural domain. The chimeric antigen receptor has relatively small clinic side effects and relatively high safety, a solid tumor can be effectively shrunk, and the treatment effect of tumors can be effectively improved.
Owner:GUANGZHOU BIO GENE TECH CO LTD
Specific chimeric antigen receptor T cells targeting CD19 and preparation and clinical application thereof
PendingCN110272493AEffective targeted attackHigh kill rateVirusesAntipyreticSide effectCD19-specific chimeric antigen receptor
The invention relates to specific chimeric antigen receptor T cells targeting CD19, and a preparation method and clinical application thereof. The present invention constructs a specific chimeric antigen receptor targeting CD19 and immune response cells modified by the chimeric antigen receptor based on a targeted human CD19 single-chain antibody sequence. The novel modified immune response cells can effectively target and attack a plurality of tumor cells, especially tumor cells with positive CD19 expressiion, and can be used to prepare a preparation for the treatment of tumors. The method for preparing the modified immune response cells targeting CD19 is simple, and the obtained modified immune response cells targeting CD19 have a high killing rate on tumor cells. Clinical verification shows that: after a million-grade low-dose back transfusion, patients with recurrent and refractory advanced CD19-positive lymphoma get a significant clinical symptom relief after two weeks of treatment, almost complete relief curative effect is obtained on the 77th day, and no fever caused by cytokine release syndrome and any neurotoxic side effect occur.
Owner:NANJING KAEDI BIOTECH INC
Chimeric Antigen Receptor (CAR) Comprising a CD19-Binding Domain
ActiveUS20180044417A1Reduces T-cell exhaustionImproved clinical responsePolypeptide with localisation/targeting motifImmunoglobulin superfamilyChimeric antigen receptorHeavy chain
There is provided a chimeric antigen receptor (CAR) comprising a CD19-binding domain which comprises a) a heavy chain variable region (VH) having complementarity determining regions (CDRs) with the following sequences: CDR1-GY-AFSSS (SEQ ID No. 1); CDR2-YPGDED (SEQ ID No. 2) CDR3-SLLYGDYLDY (SEQ ID No. 3); and b) a light chain variable region (VL) having CDRs with the following sequences: CDR1-SASSSVSYMH (SEQ ID No. 4); CDR2-DTSKLAS (SEQ ID No. 5) CDR3-QQWNINPLT (SEQ ID No. 6). There is also provided a cell comprising such a CAR, and the use of such a cell in the treatment of cancer, in particular a B cell malignancy.
Owner:AUTOLUS LIMIED
Anti-CD19 antibody and preparation method and use thereof
ActiveCN107793478AHigh affinityIncrease lethalityAntibody mimetics/scaffoldsGenetically modified cellsHeavy chainSingle-Chain Antibodies
The invention relates to the technical field of biotechnology and particularly relates to an anti-CD19 antibody and a preparation method and use thereof. CDR in a heavy chain variable region of the anti-CD19 antibody comprises CDR-H1 with an amino acid sequence shown in the formula of SEQ ID No. 1, CDR-H2 with an amino acid sequence shown in the formula of SEQ ID No. 2 and CDR-H3 with an amino acid sequence shown in the formula of SEQ ID No. 3. The CDR of a light chain variable region of the anti-CD19 antibody comprises CDR-L1 with an amino acid sequence shown in the formula of SEQ ID No. 4, CDR-L2 with an amino acid sequence shown in the formula of SEQ ID No. 5 and CDR-L3 with an amino acid sequence shown in the formula of SEQ ID No. 6. The FMC63 scFv is screened in affinity maturation through a phage display technology so that a high affinity single chain antibody against CD19 is obtained.
Owner:HUADAO SHANGHAI BIOPHARMA CO LTD
Bispecific CD3 and CD19 antigen binding constructs
Owner:ZYMEWORKS INC