Patents
Literature
1658results about "Powdered material dispensing" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
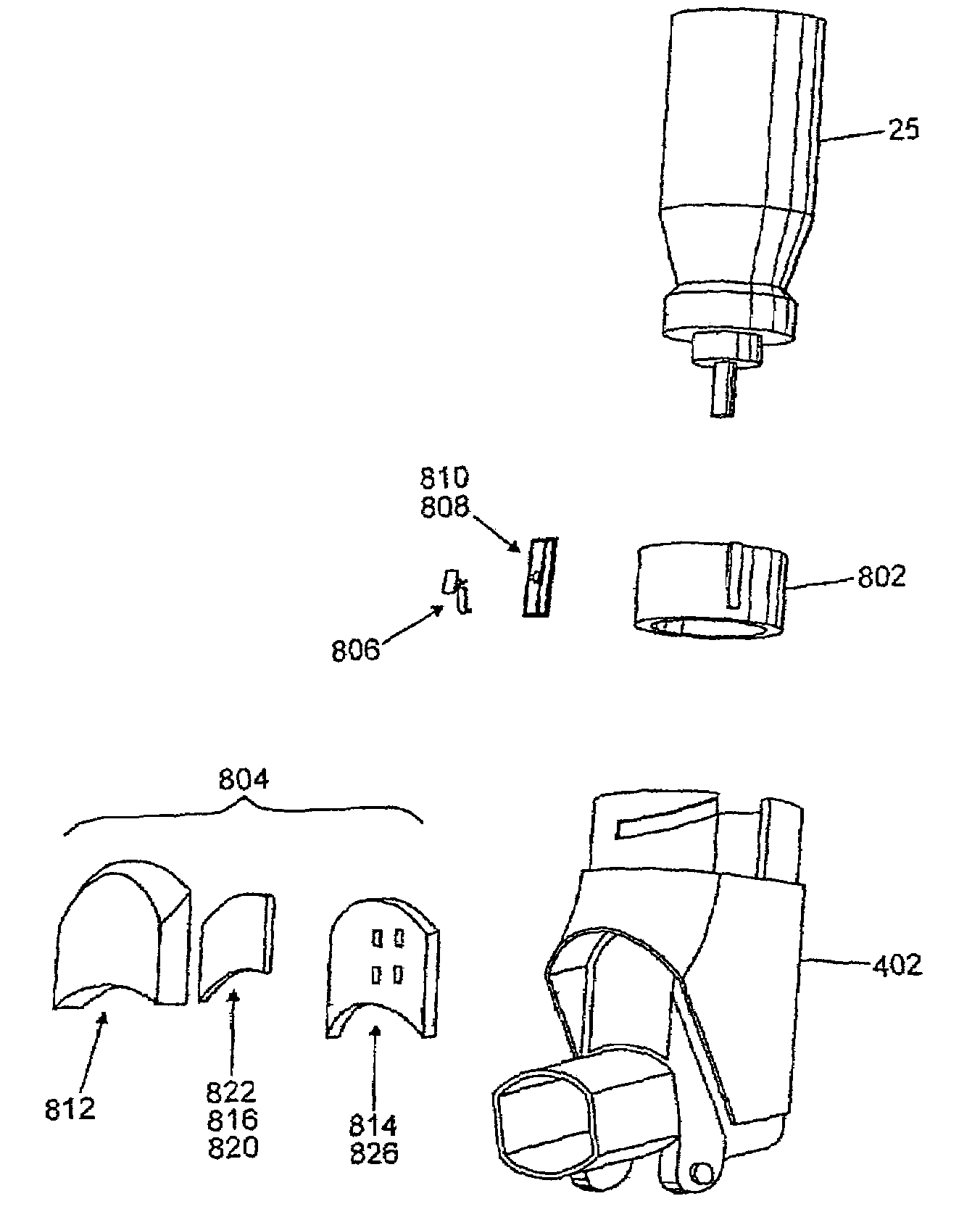
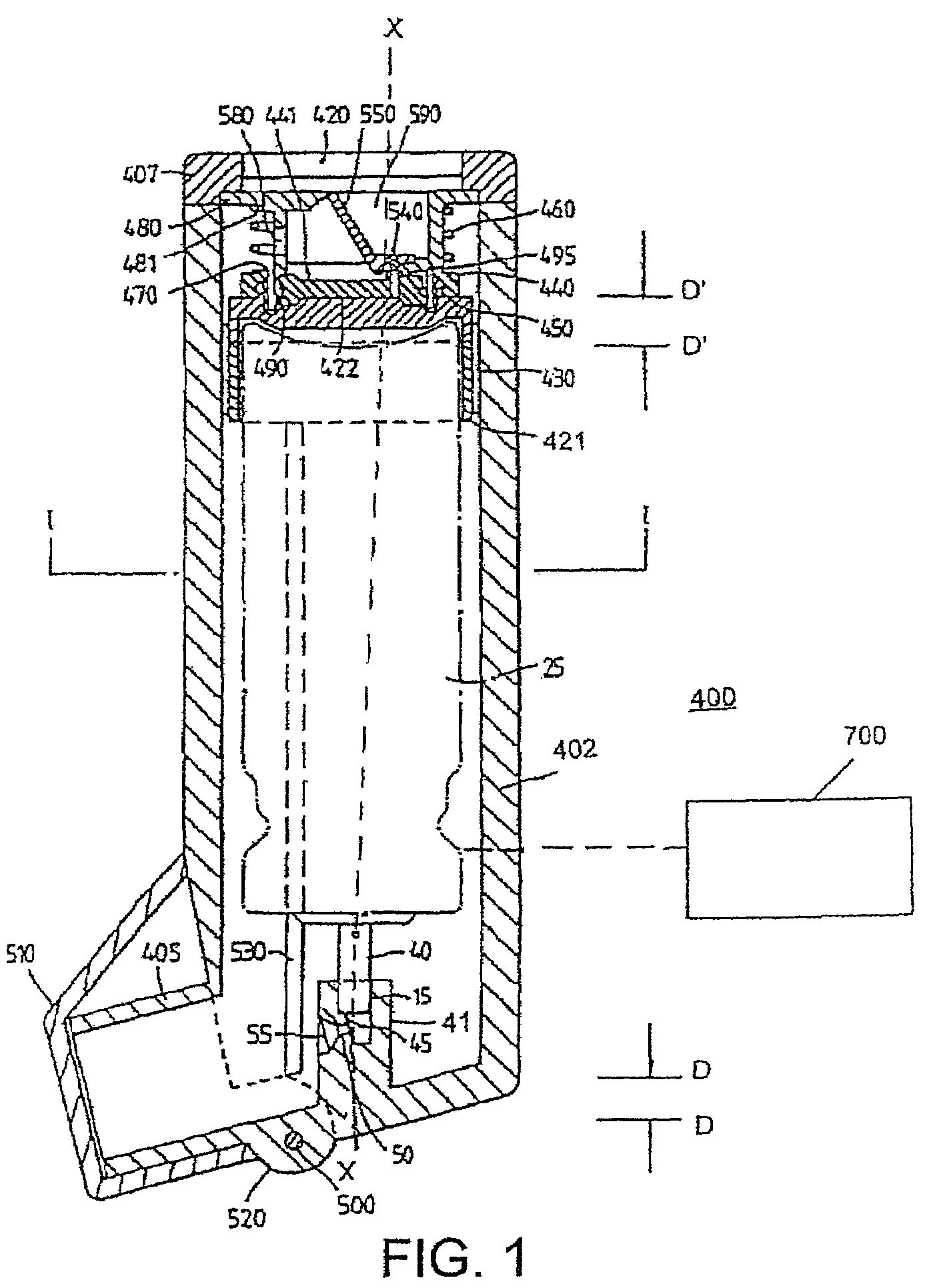
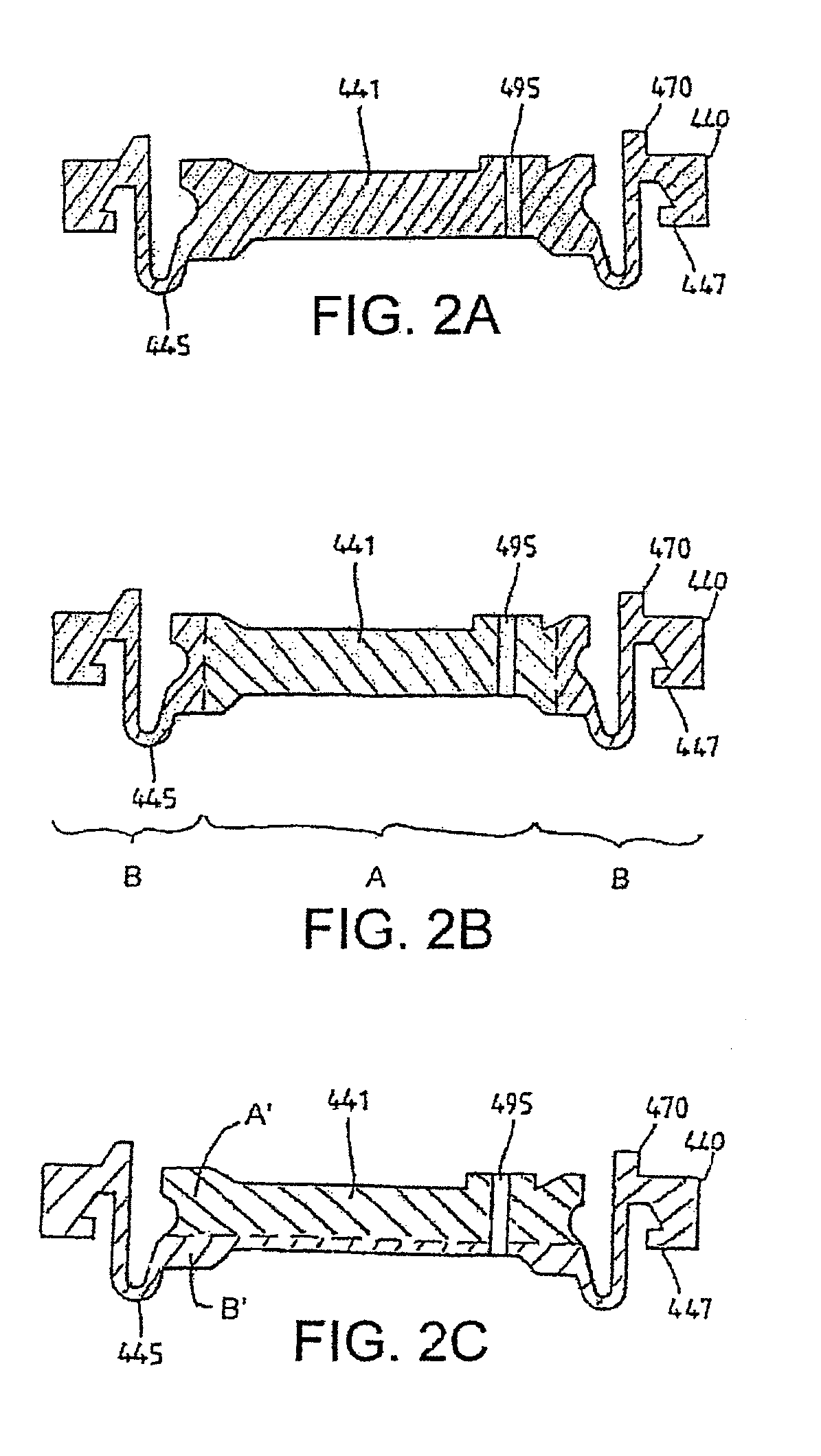
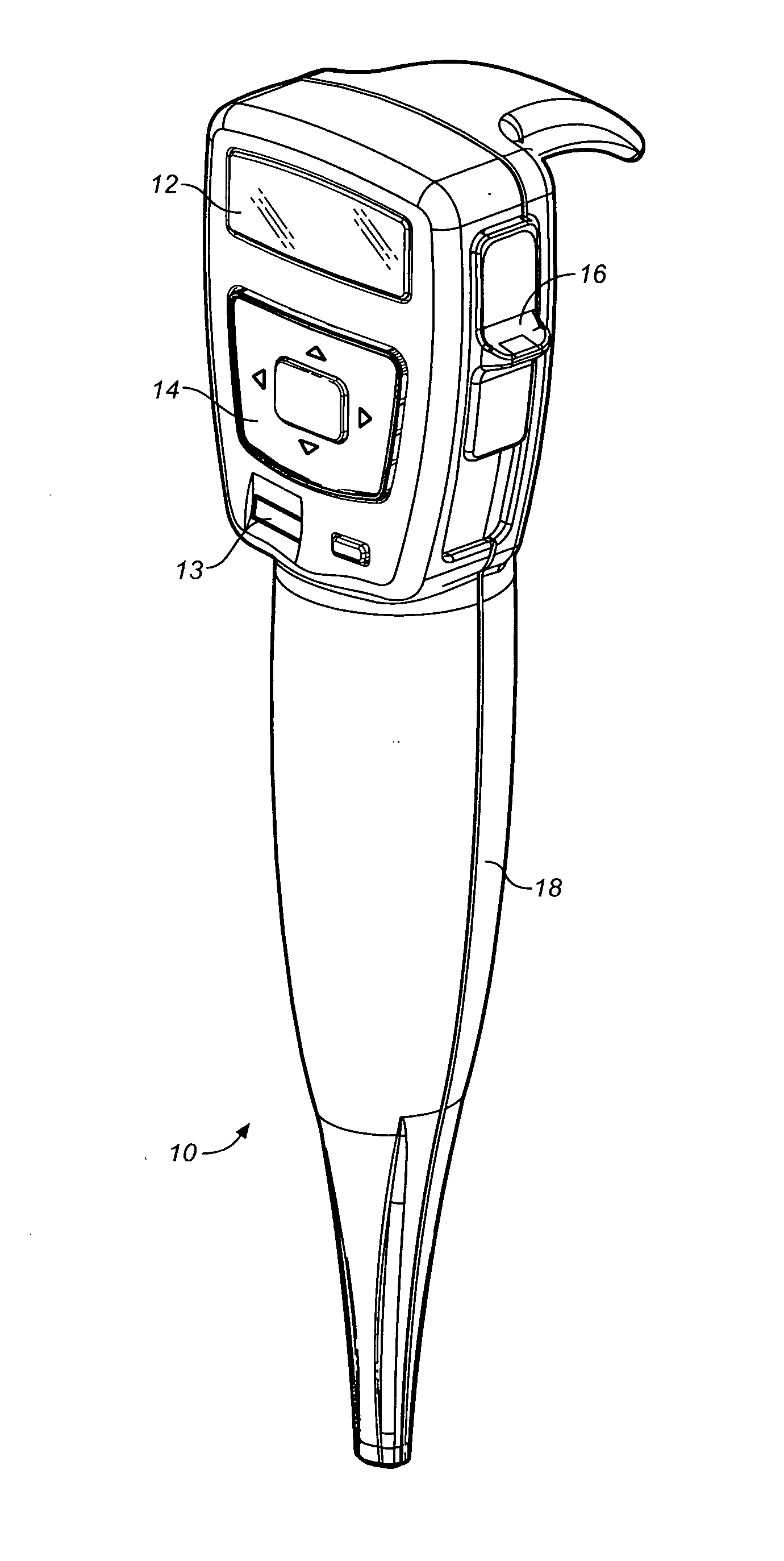
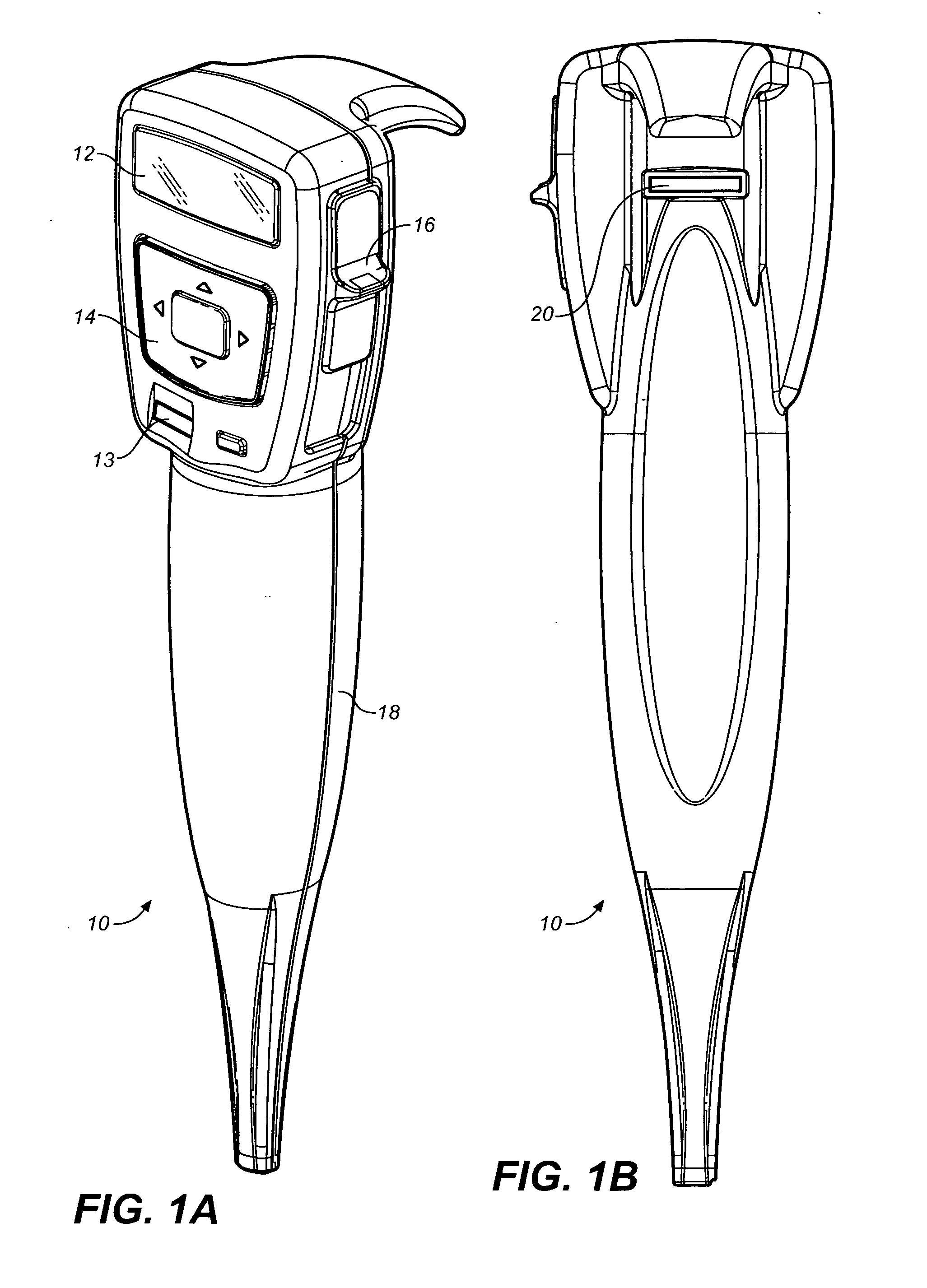
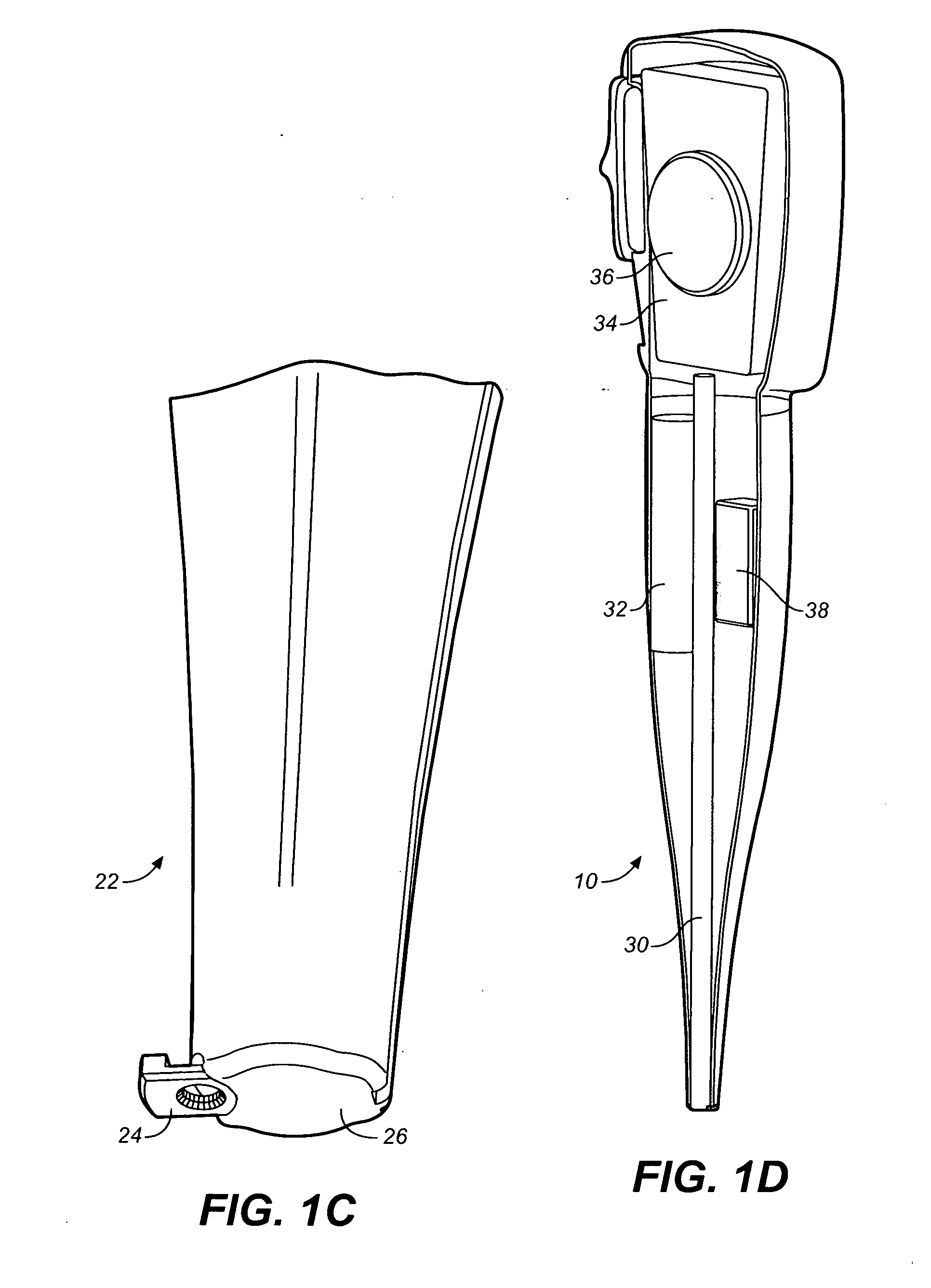
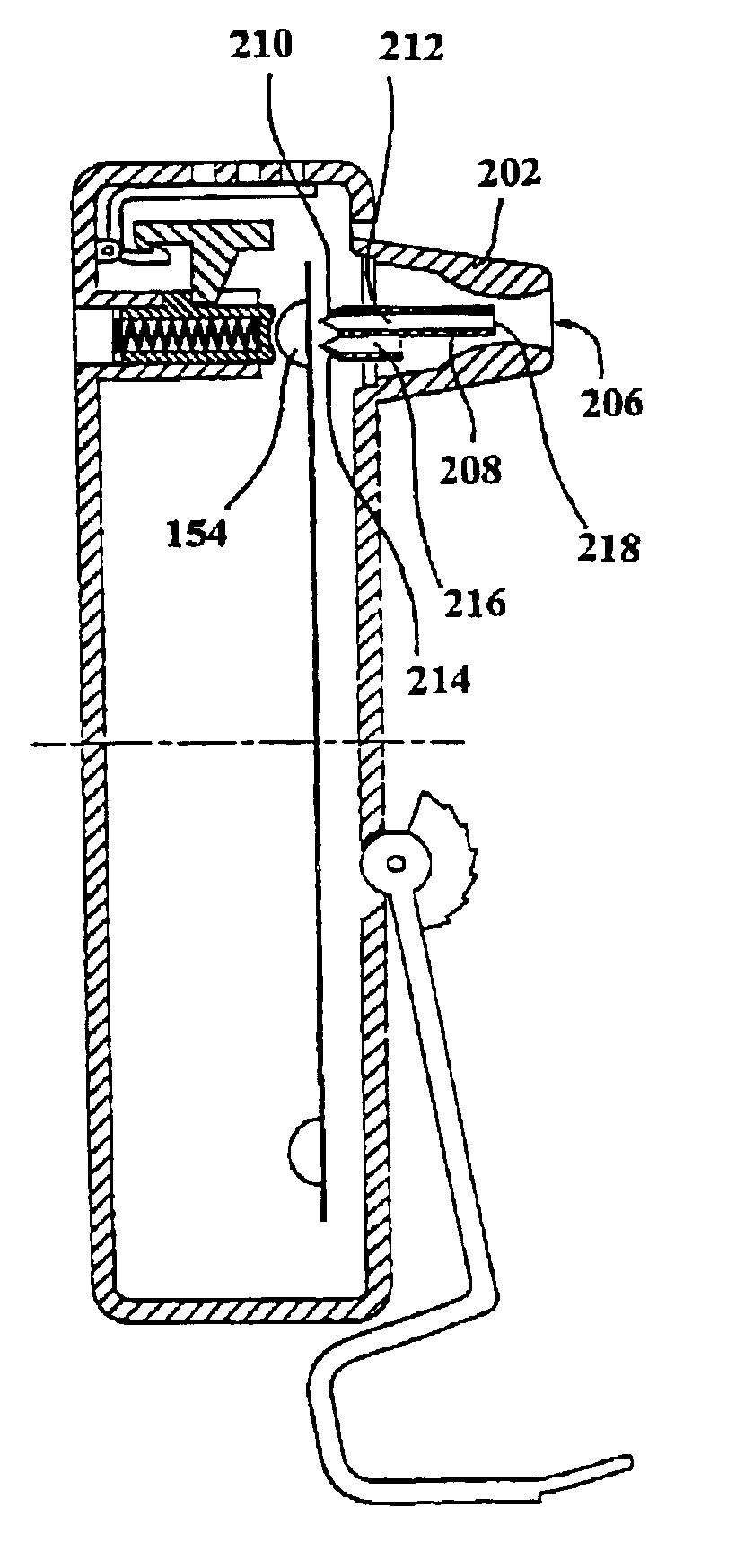
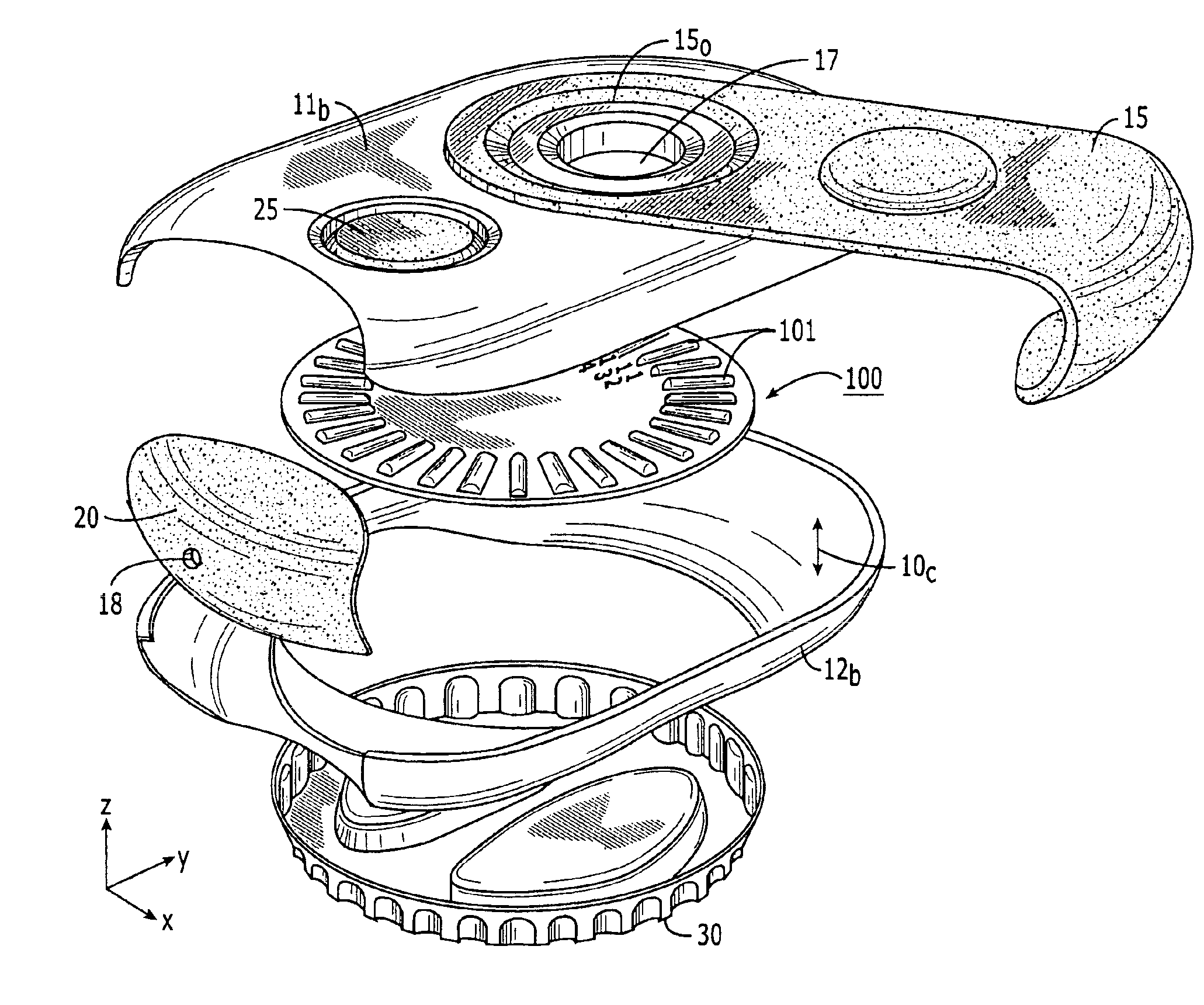
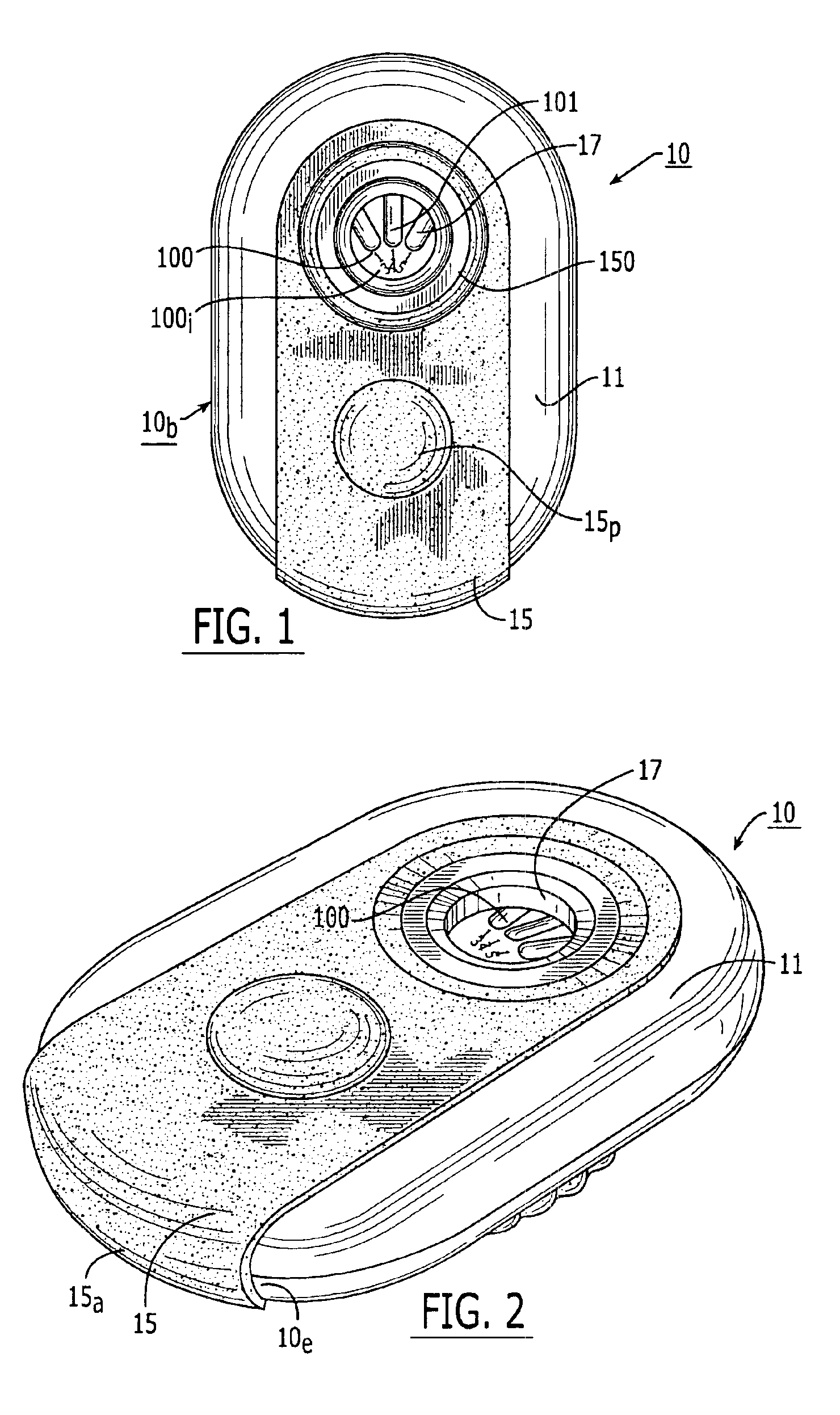
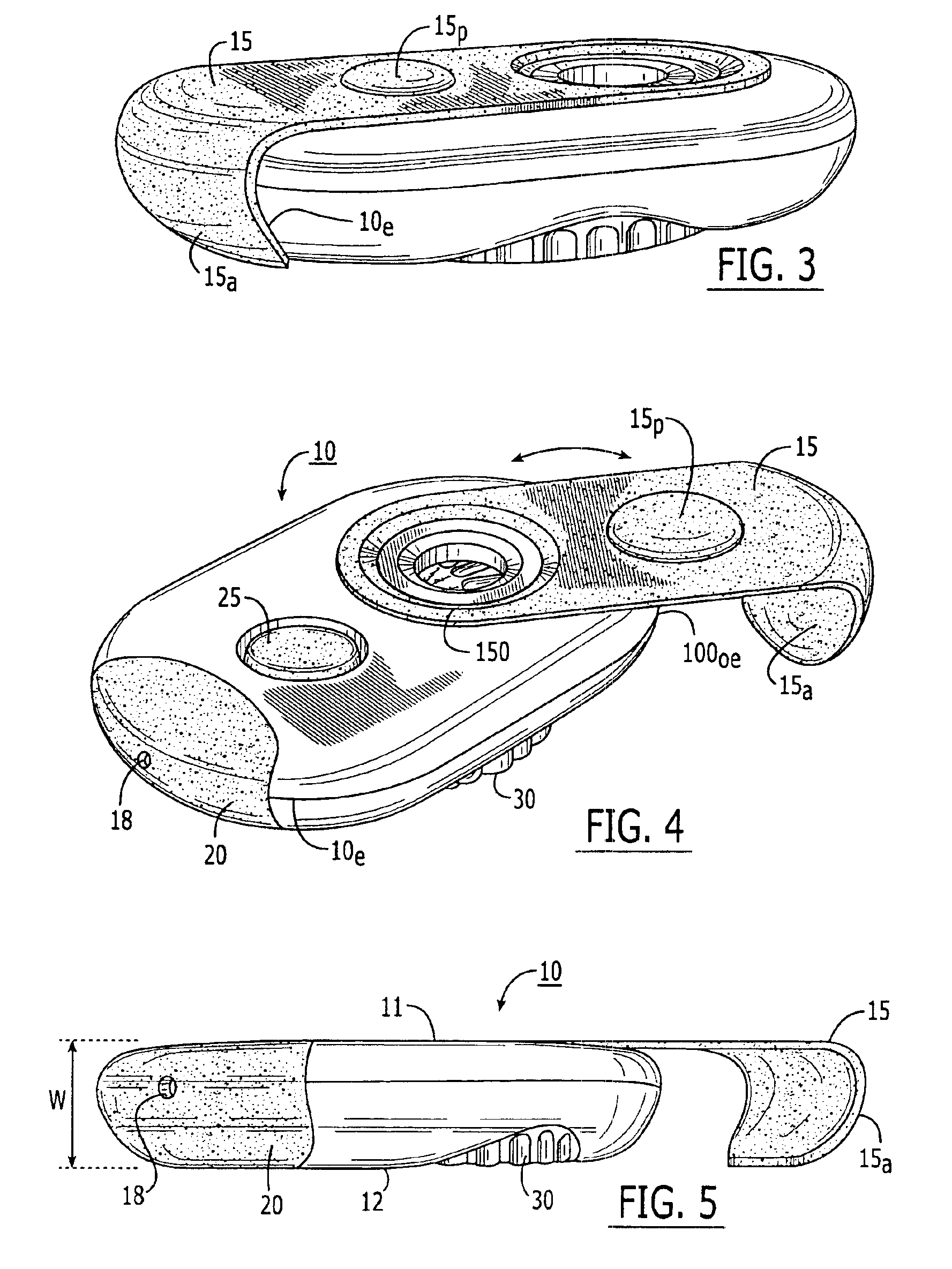
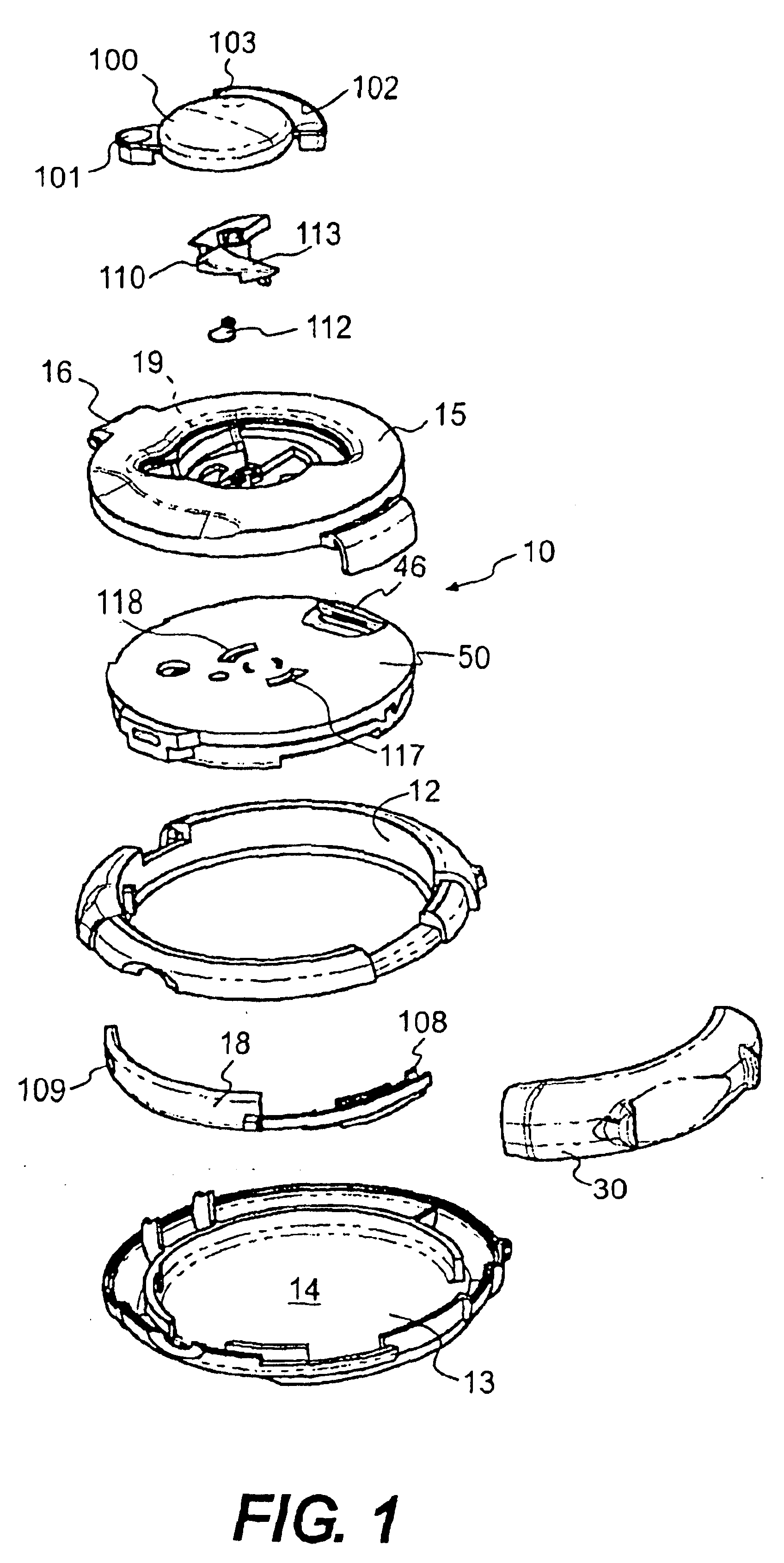
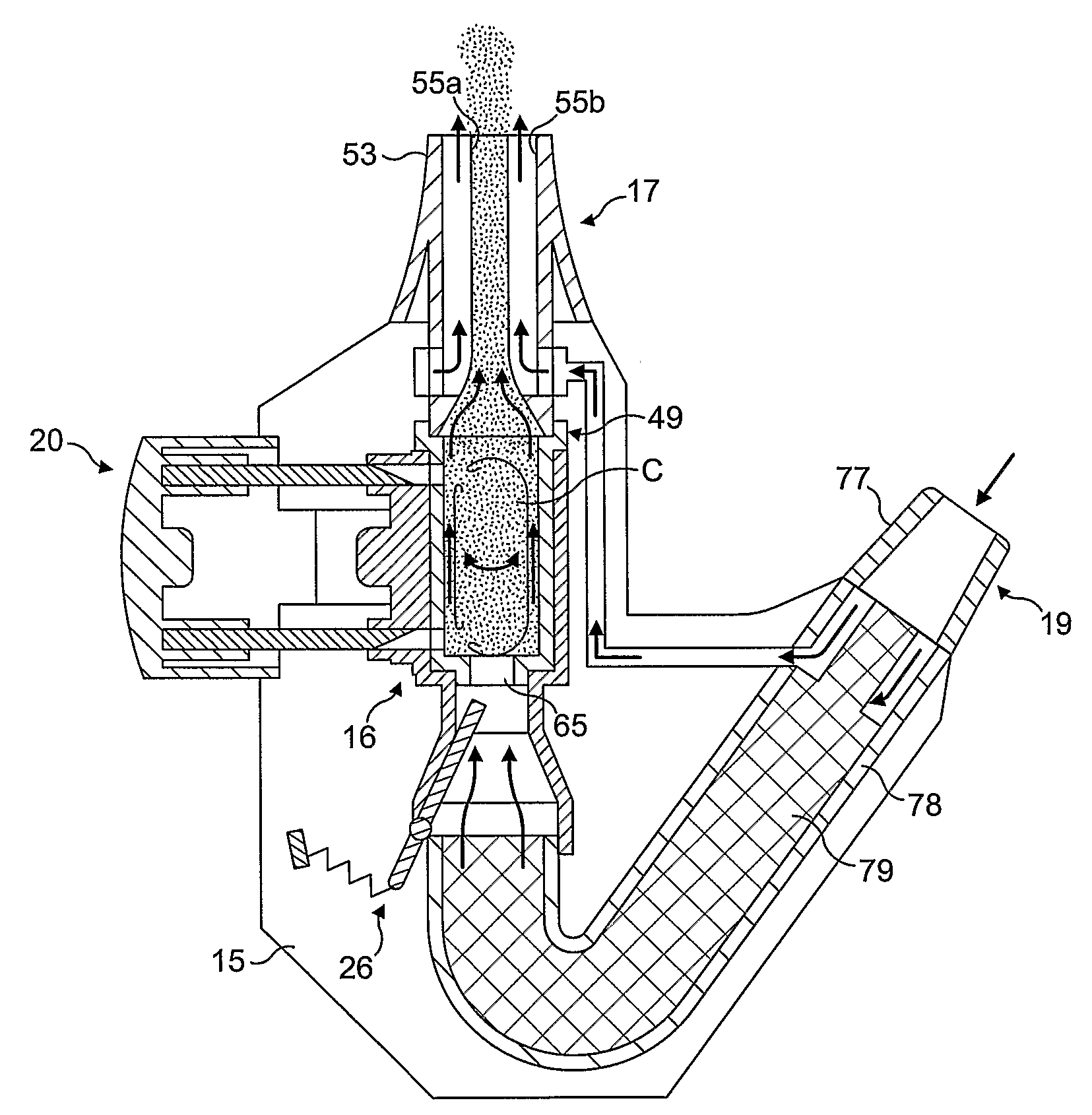
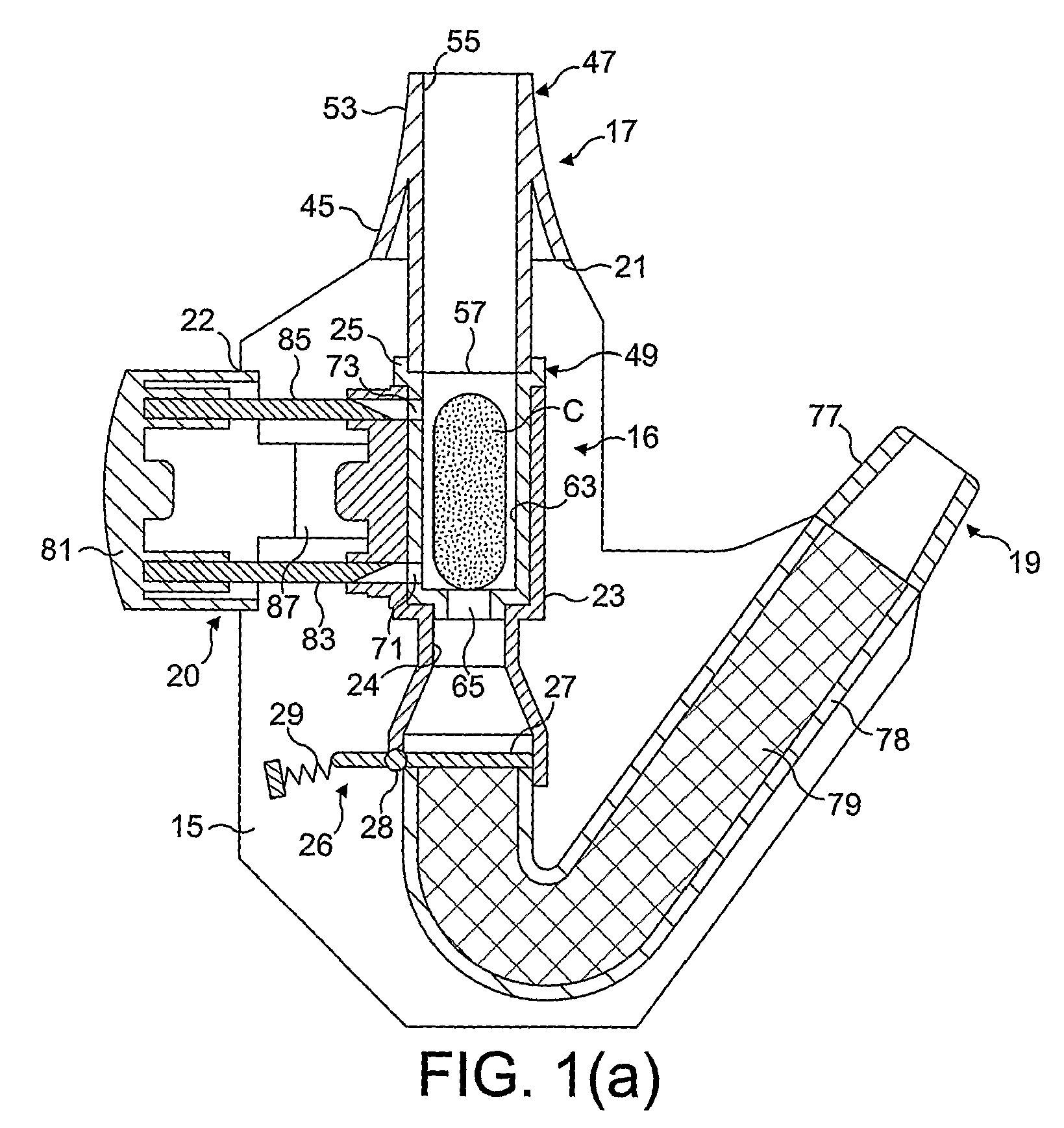
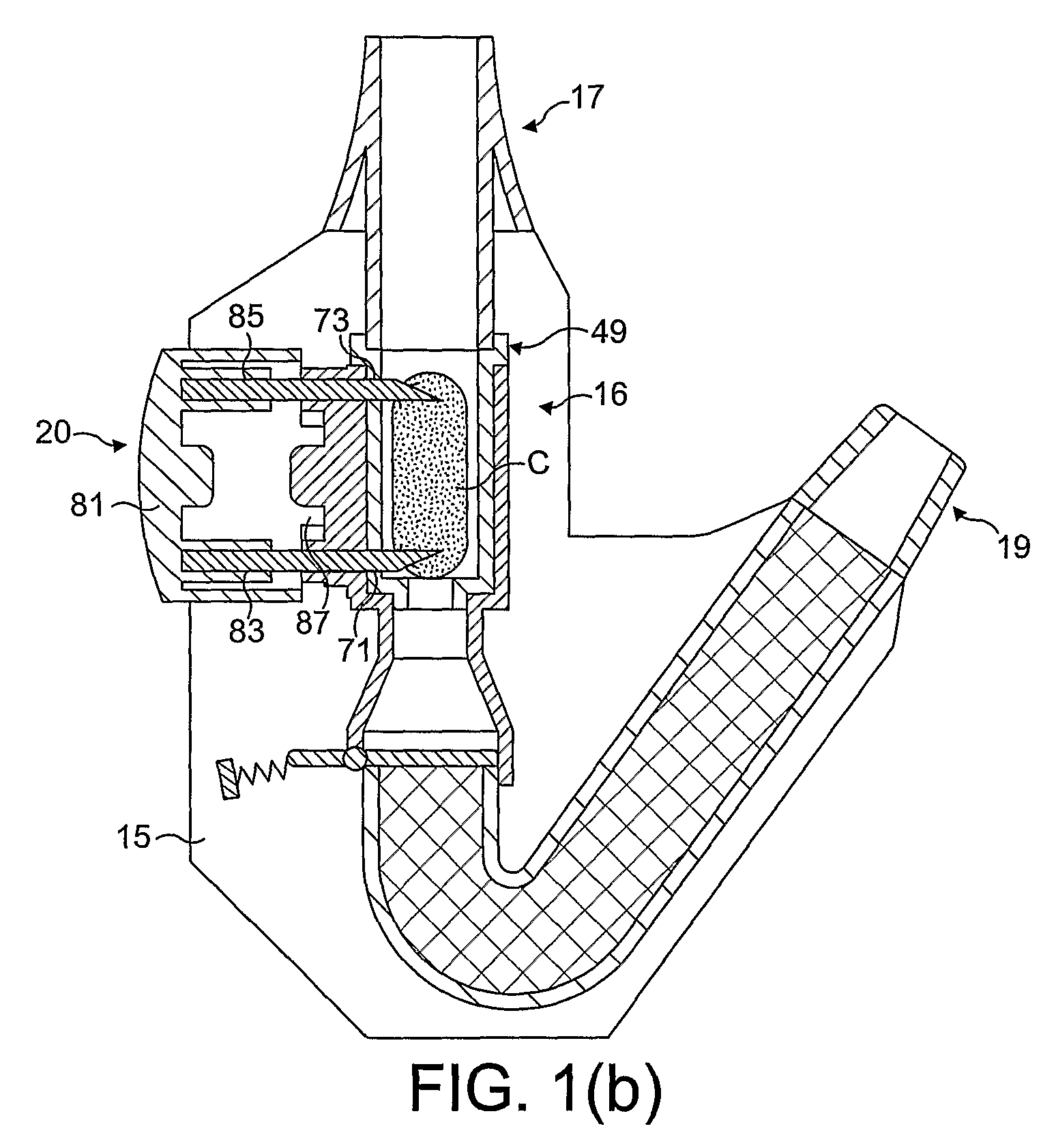
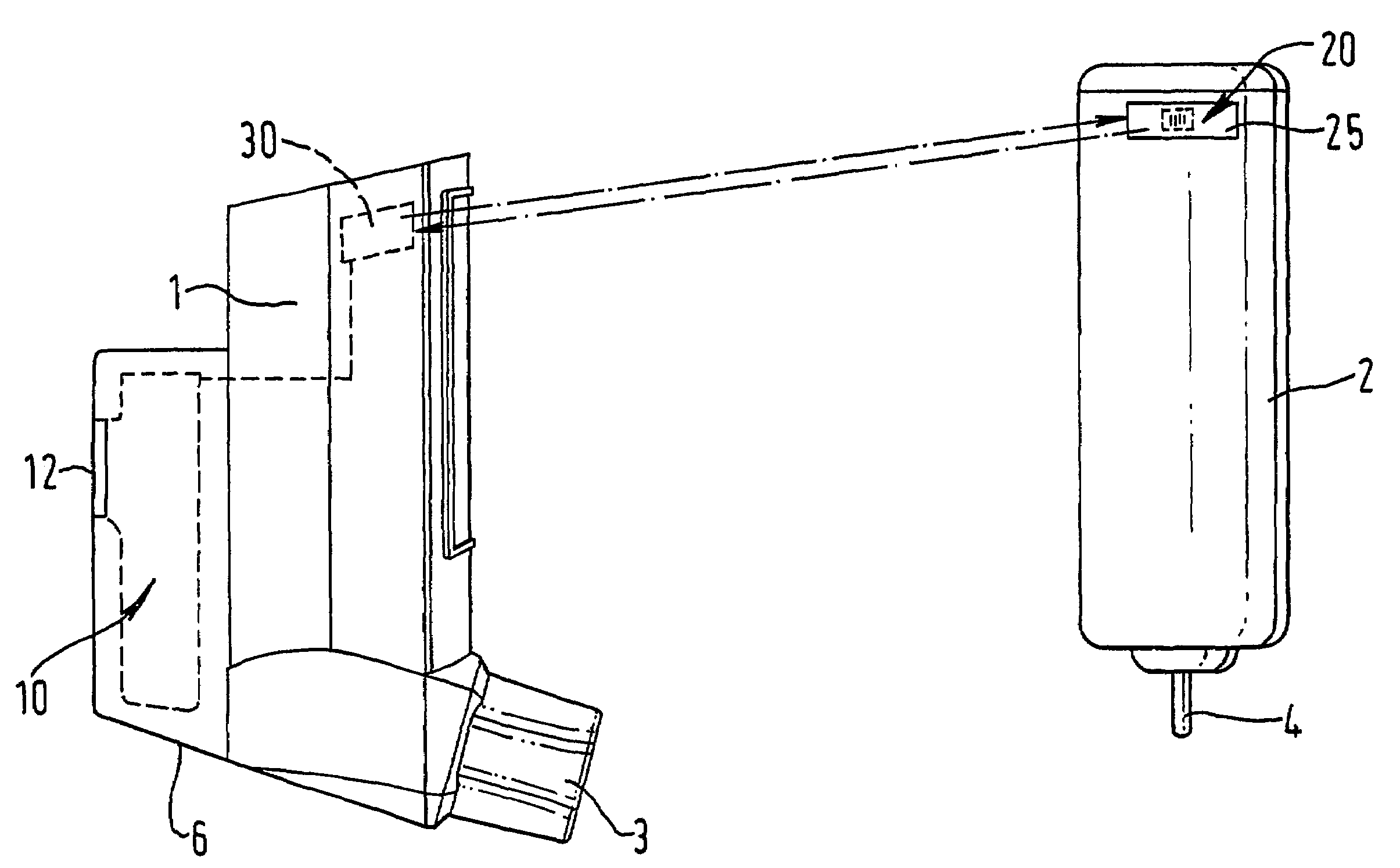
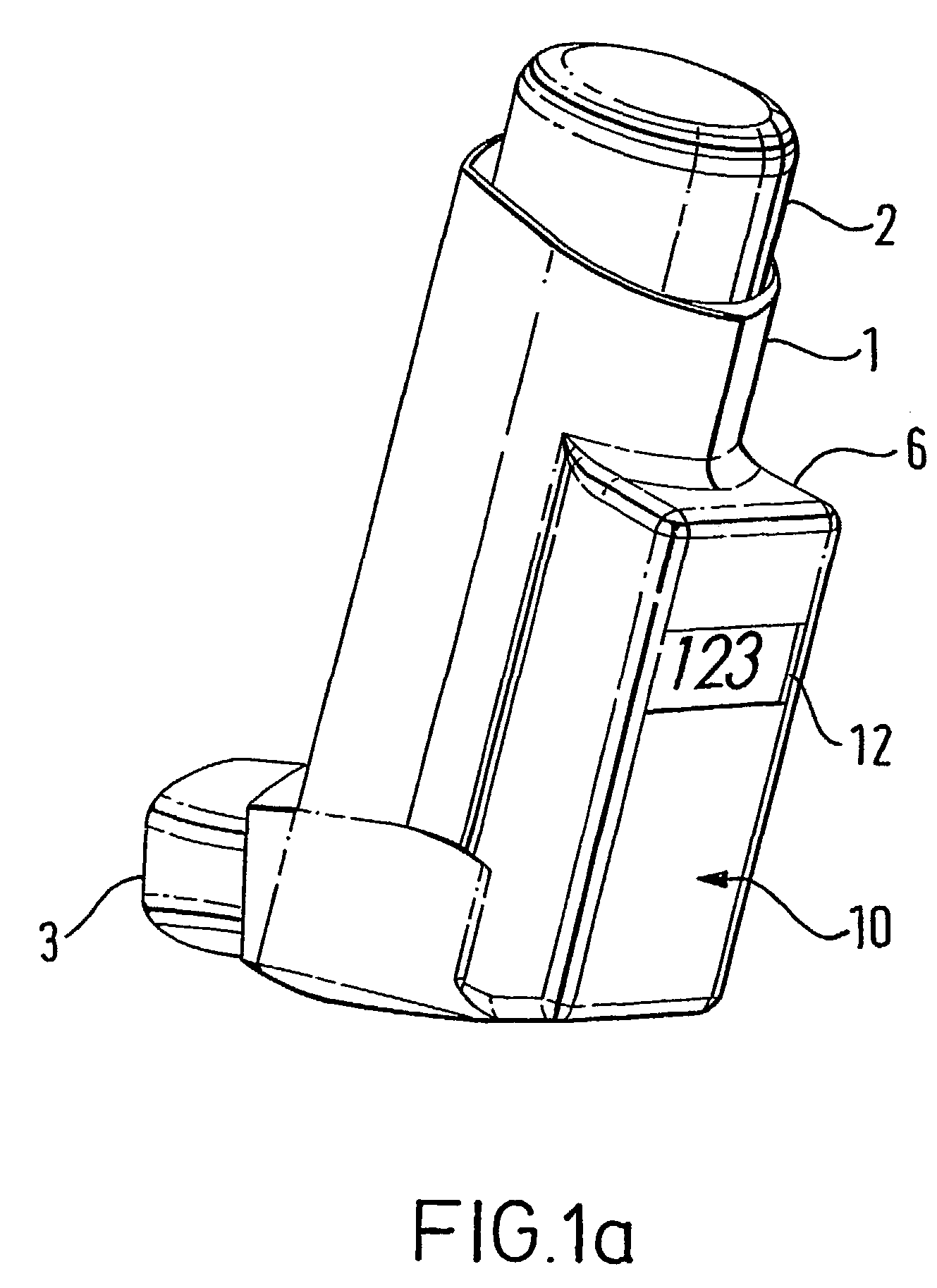
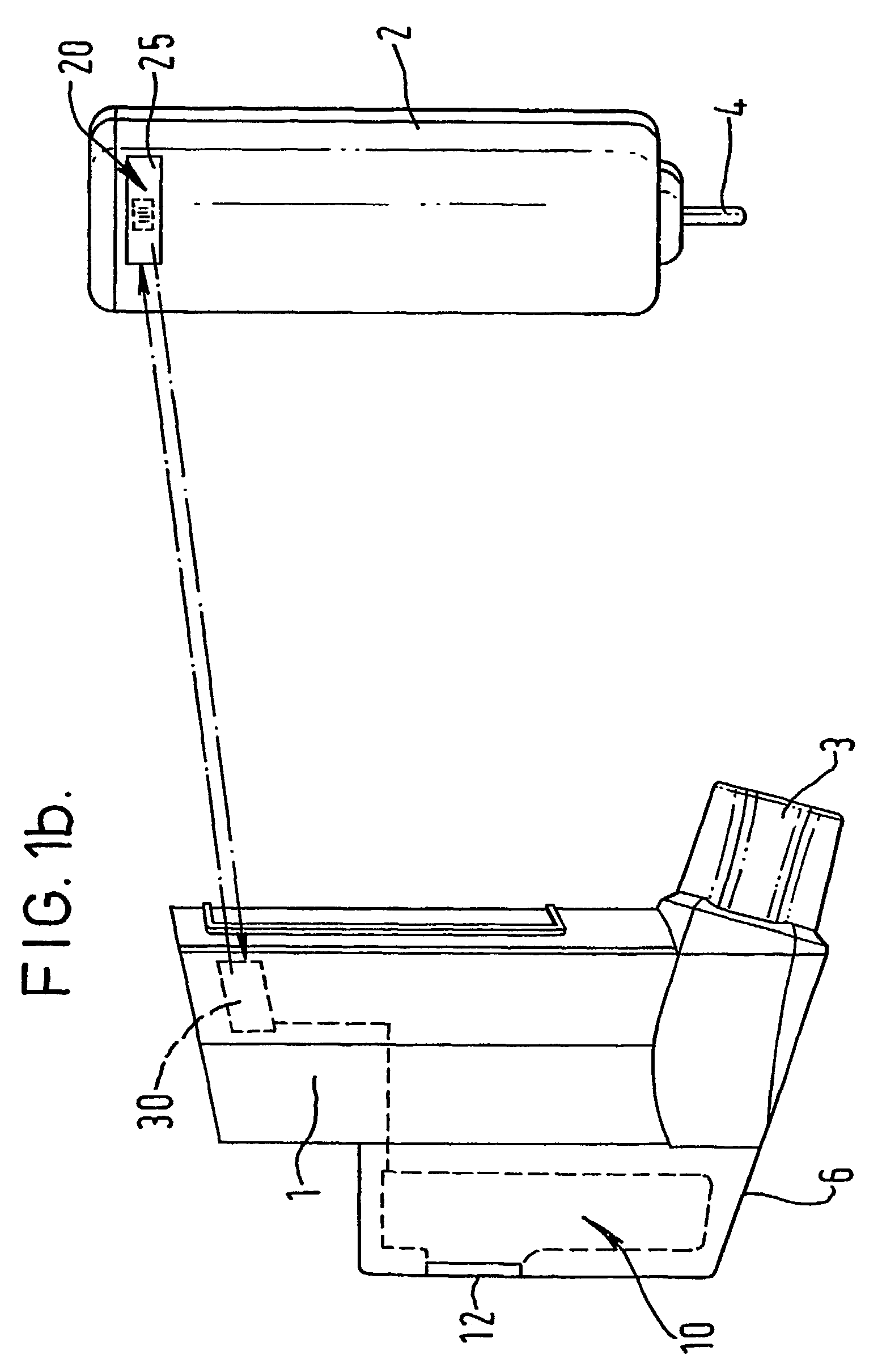
Medicament dispensing device with a display indicative of the state of an internal medicament reservoir
ActiveUS7331340B2Liquid surface applicatorsPowdered material dispensingDisplay deviceBiomedical engineering
A metered dose inhaler for use with a removable pressurized aerosol canister, or reservoir, having a display for indicating to a user the state of the canister. A memory device on the canister or a housing which houses the canister stores information indicative of doses dispensed from, or remaining in, the canister. That information is processed to provide and display information representative of the state of the canister.
Owner:IVAX CORP
Drug storage and dispensing devices and systems comprising the same
ActiveUS20070186923A1Minimizing saliva influxPowdered material dispensingDrug and medicationsDrug StorageBiomedical engineering
Drug storage and dispensing devices for dispensing a drug dosage form to a patient are disclosed. The dispensing device has a programmable lock-out feature for locking the dispensing device and is capable of detecting the identity of a user. The invention further provides a method for the treatment of subject, by administering to the subject a drug dosage form using a dispensing device of the invention.
Owner:ACEIRX PHARM INC
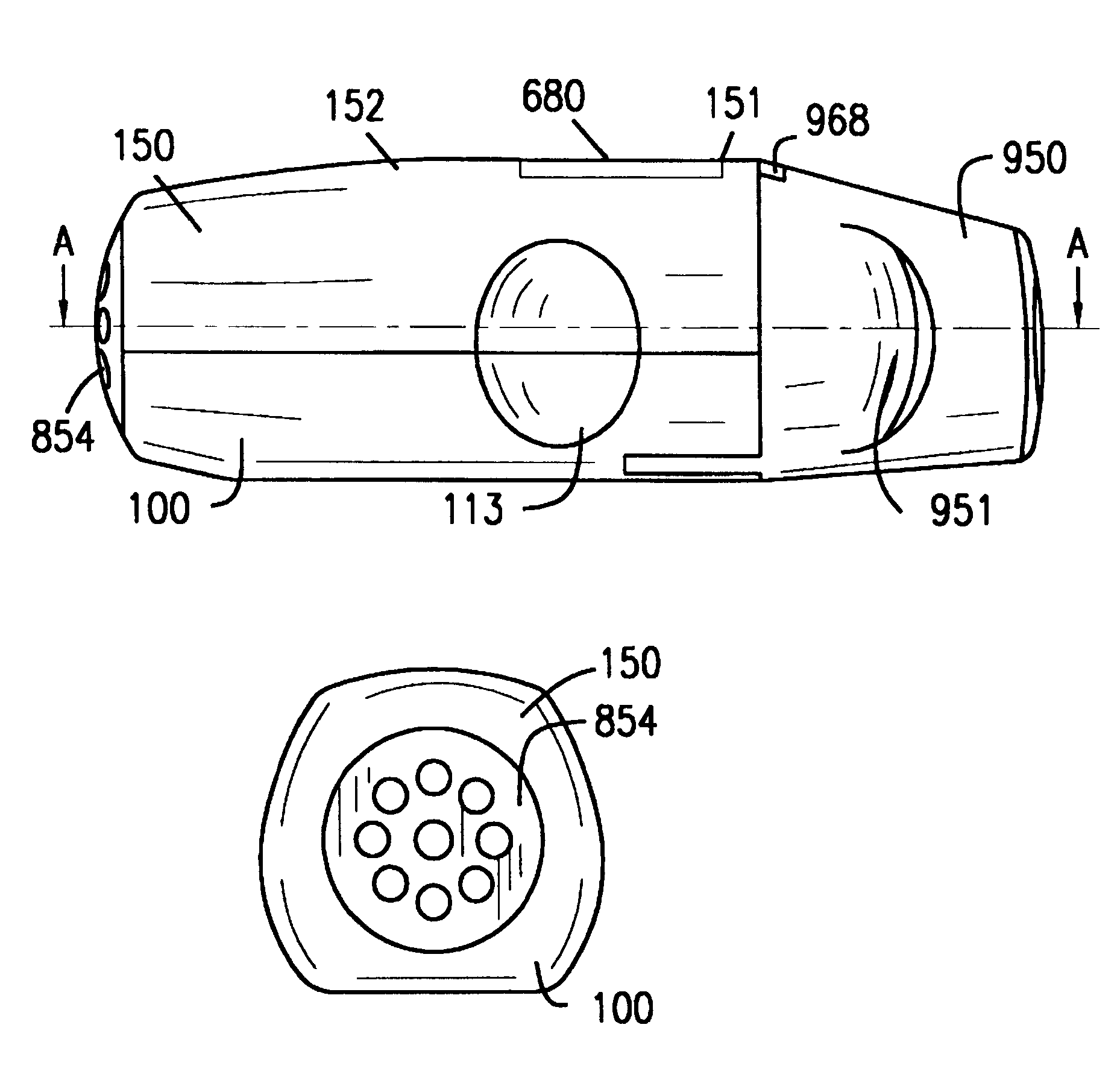
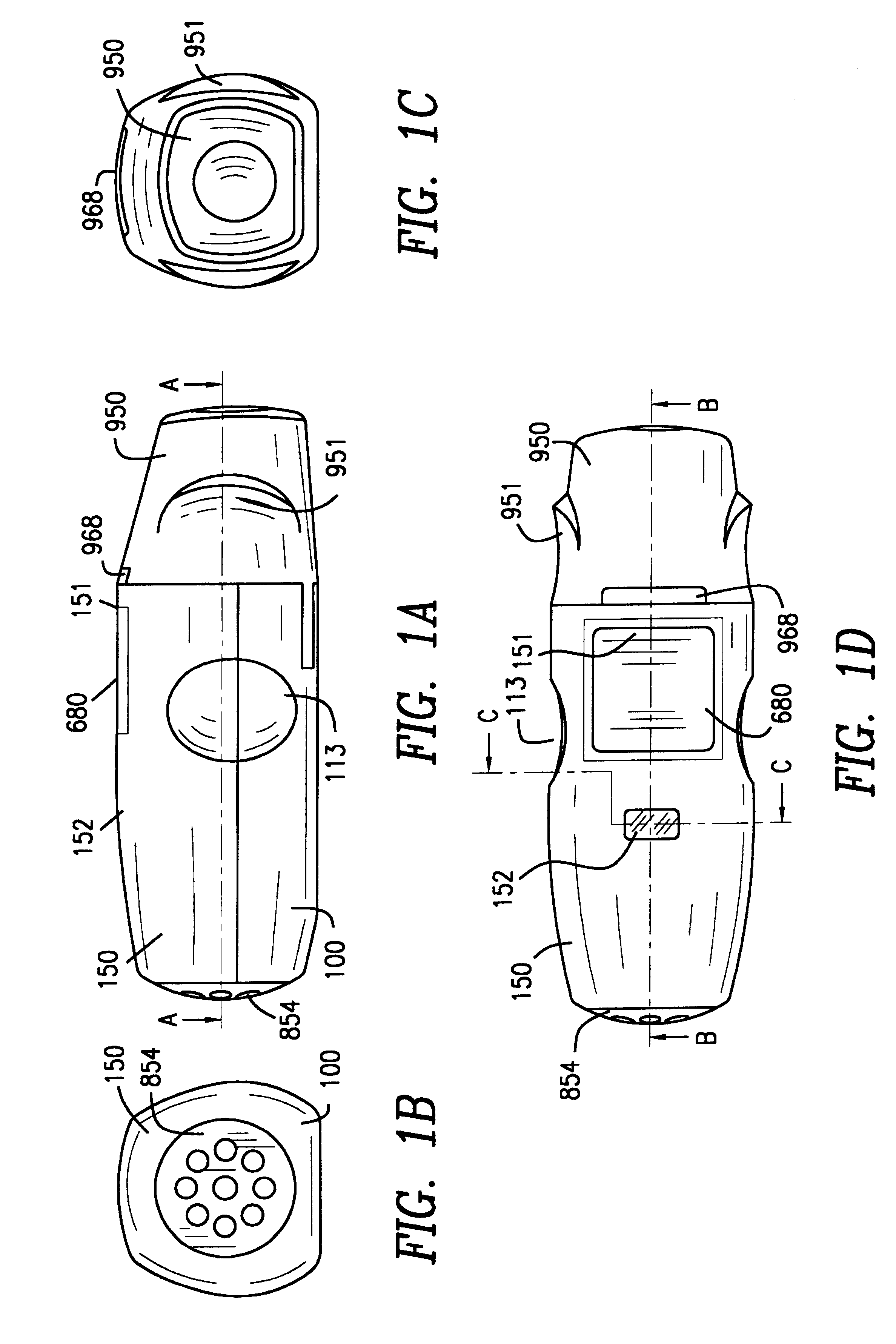
Inhaler for multiple dosed administration of a pharmacological dry powder
InactiveUS6182655B1Avoid possibilityReducing powder flow rateRespiratorsLiquid surface applicatorsMouth pieceEngineering
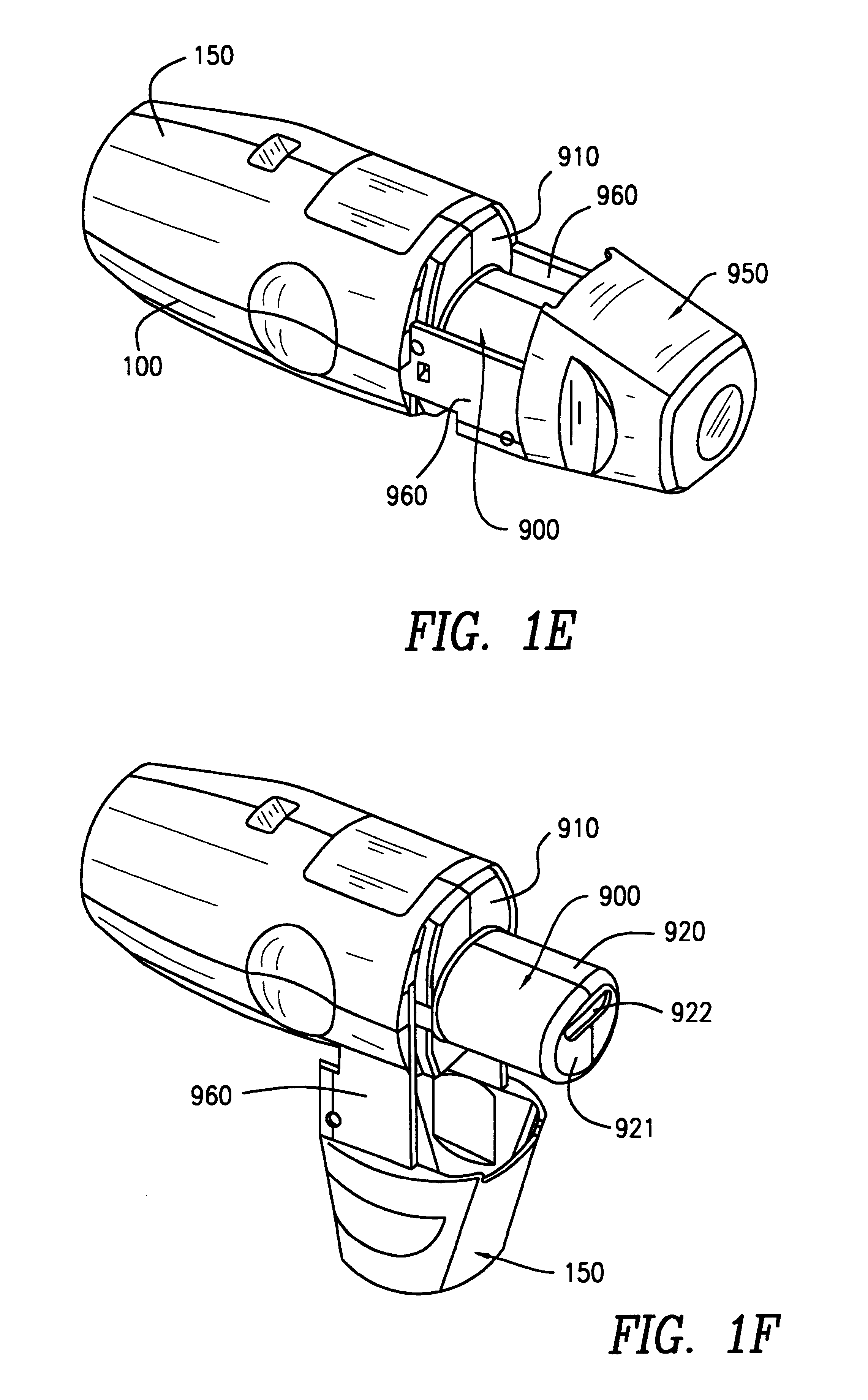
An inhaler for multiple dosed administration of a pharmacological dry powder consists externally of a housing (100,150) and of a protective cap (950) which can be removed from a special mouthpiece (900) fitted on the housing. Arranged on the inside there are a slide rail (200), a dosing slide (300), a shutter (400), a carriage (500), a funnel arrangement (600), a counter device (700), a valve shield (800) and a valve guide (850). Removal of the protective cap (950) initiates the dosing, with a dose received in the dosing cavity (302) being transported to the mouth-piece (900) by means of the dosing slide (300). Only upon application of a defined minimum intensity of inhalation is the shutter (400) moved by the suctioned valve shield (800), as a result of which the dose is released for inhalation. Completed with an electronic module and a controllable nozzle, all inhalation-relevant data can be recorded and the flow conditions regulated.
Owner:JAGOTEC AG
Delivery of oral drugs
InactiveUS20010020147A1Comfortable and convenient motionComfortable and convenient feelPowder deliveryLiquid surface applicatorsMean diameterHuman patient
Disclosed is a system for delivery of a drug comprising a multiple unit dosing device comprising a housing and an actuator, said device containing multiple doses of multiparticulates comprising drug particles, said device upon actuation delivering a unit dose of said multiparticulates, said drug particles having a mean diameter of greater than 10 mum to about 1 mm such that an effective dose of said drug cannot be delivered into the lower lung of a human patient. Also disclosed are novel methods, devices and dosage forms for delivering a drug.
Owner:PHARMAKODEX LTD
Methods and compositions for pulmonary administration of a TNFa inhibitor
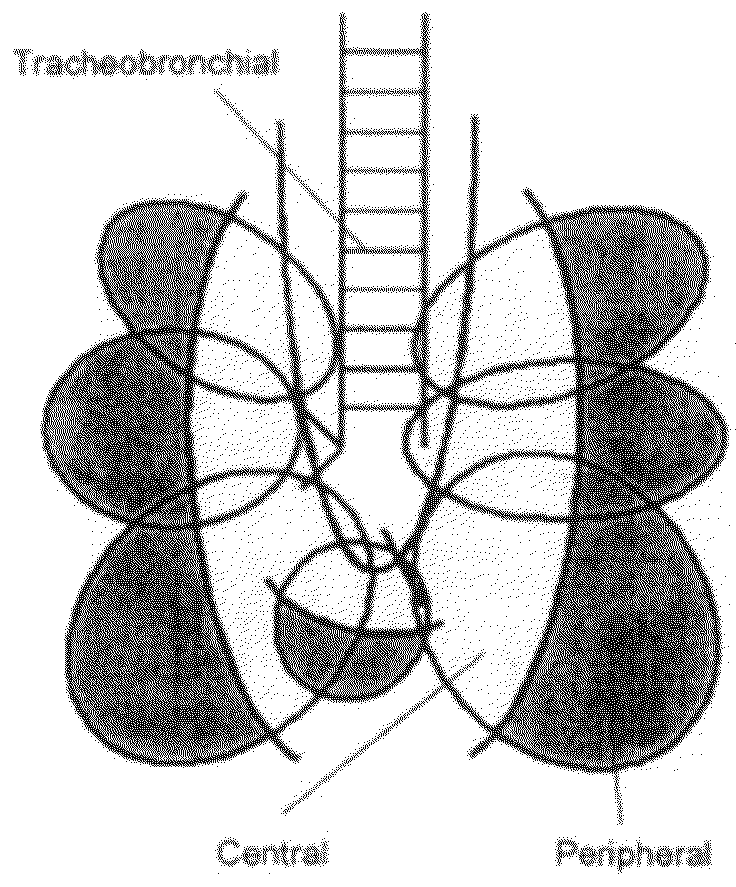
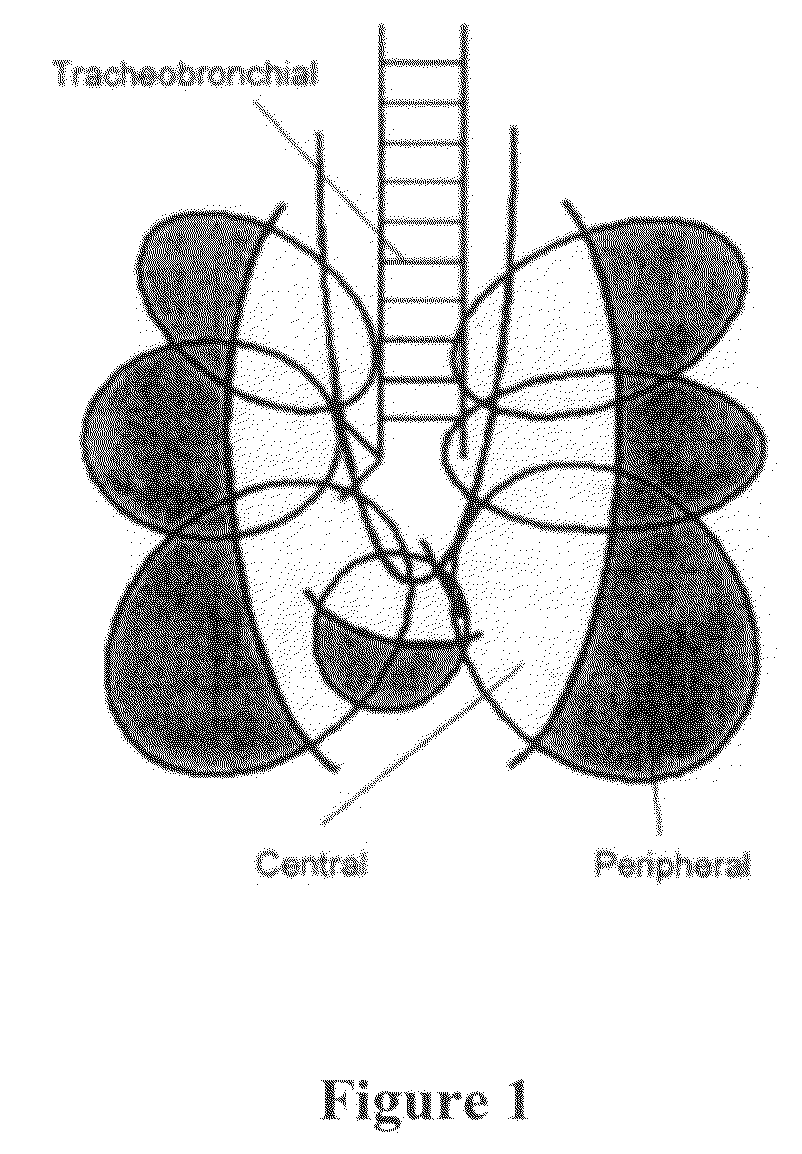
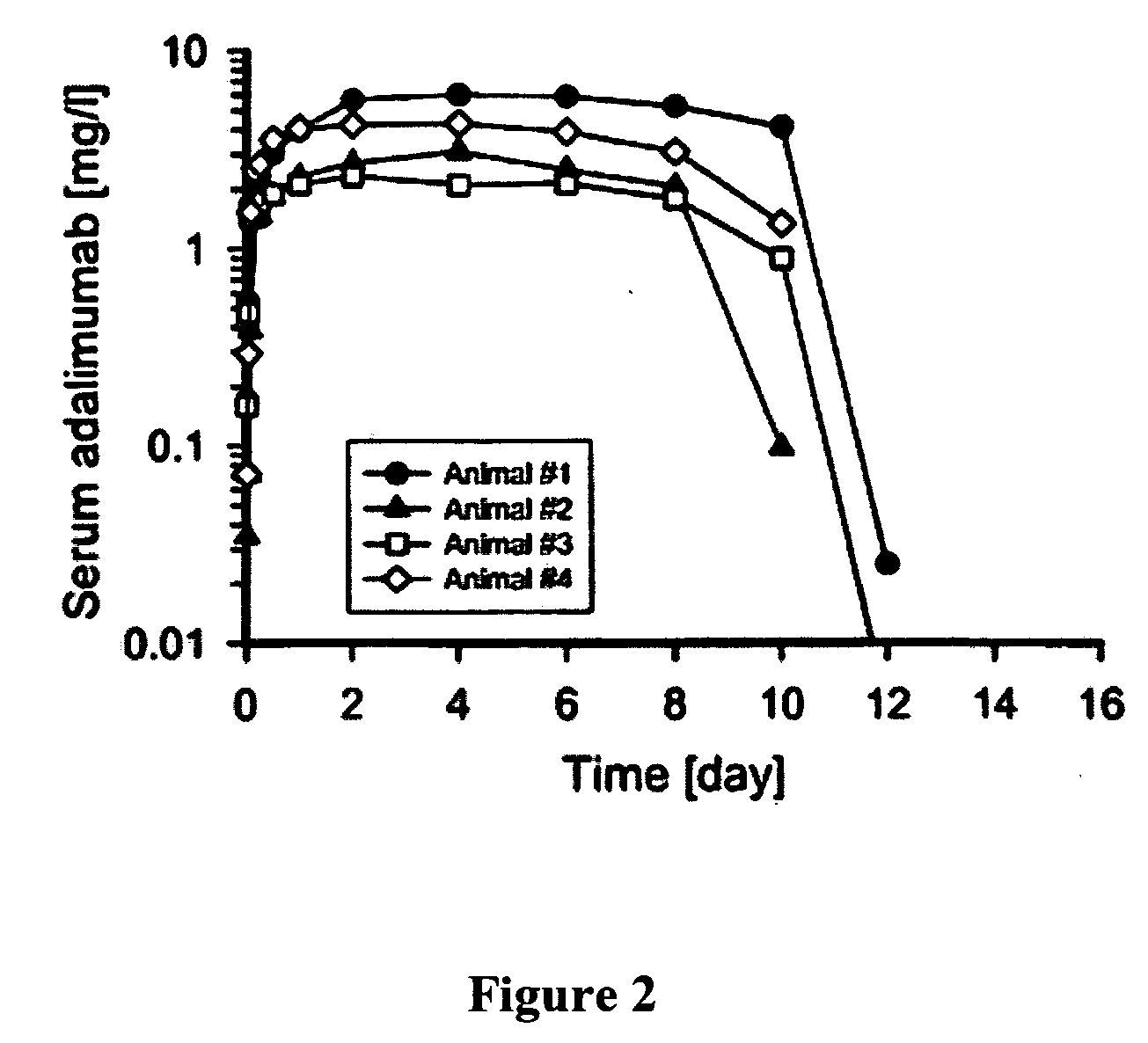
The invention describes methods of pulmonary delivery of a TNFα inhibitor to a subject having a disorder in which TNFα is detrimental, such that the disorder is treated. Also included is a method of achieving systemic circulation of a TNFα inhibitor in a subject comprising administering the TNFα inhibitor to the central lung region or the peripheral lung region of the subject via inhalation, such that systemic circulation of the TNFα inhibitor is achieved.
Owner:ABBVIE BIOTECHNOLOGY LTD
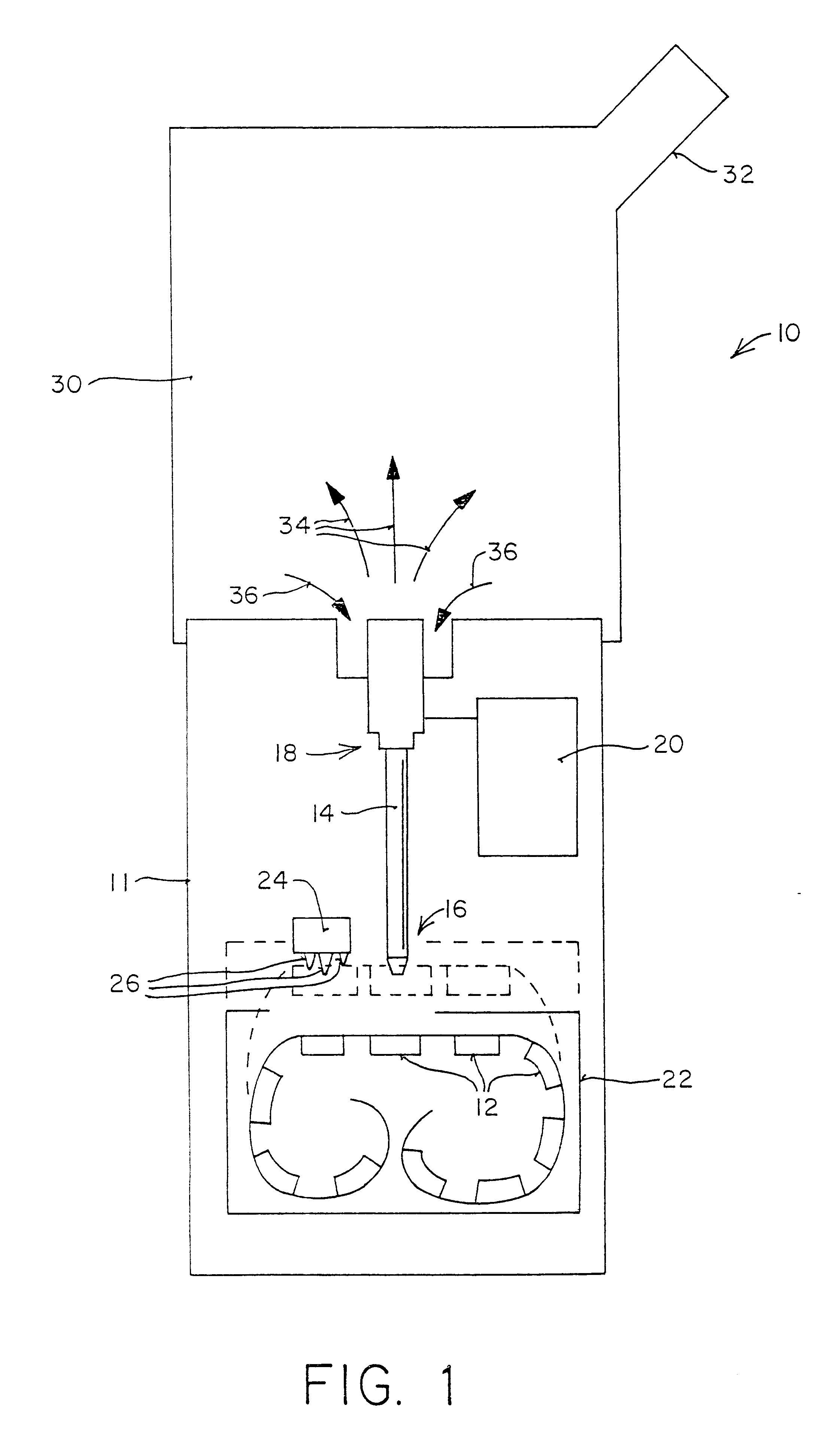
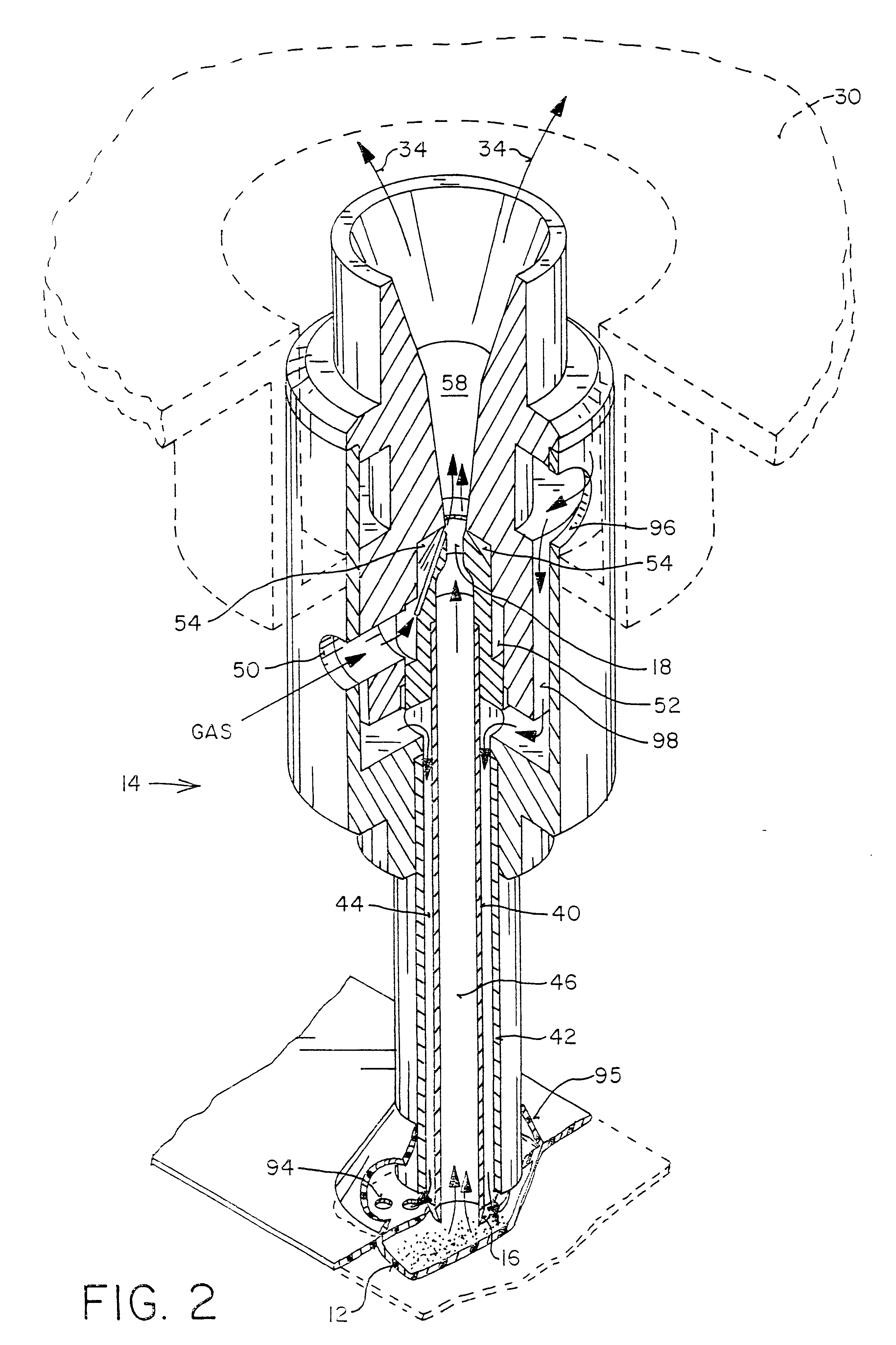
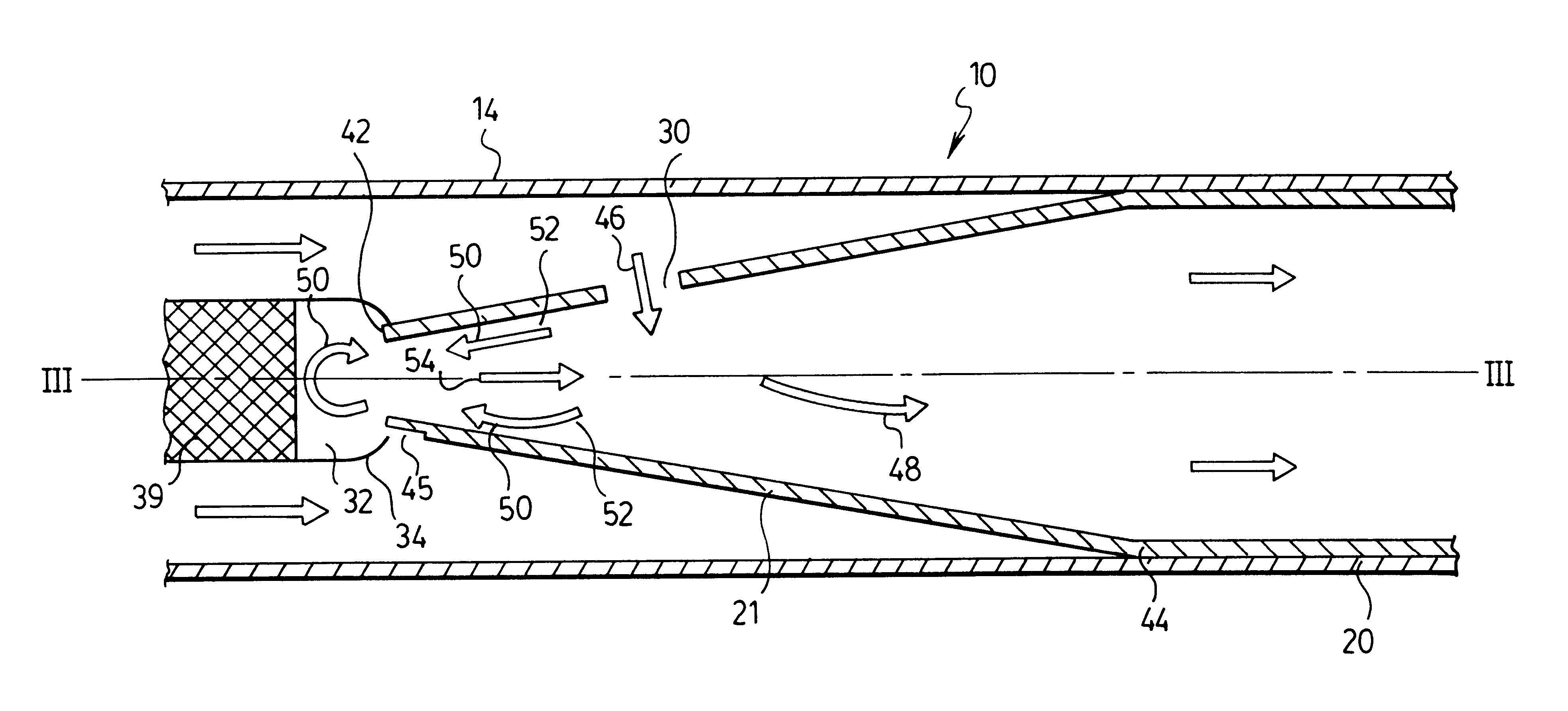
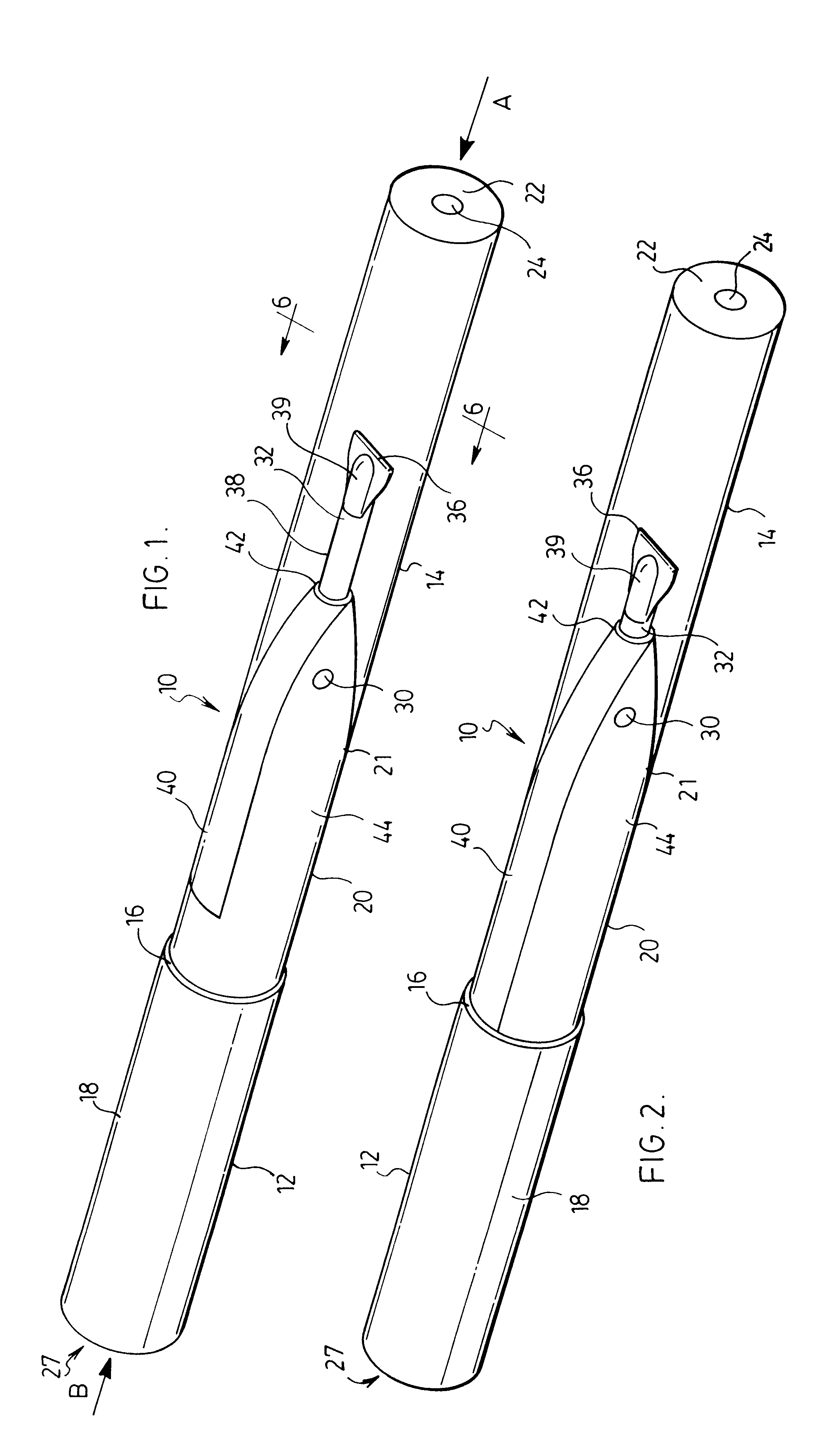
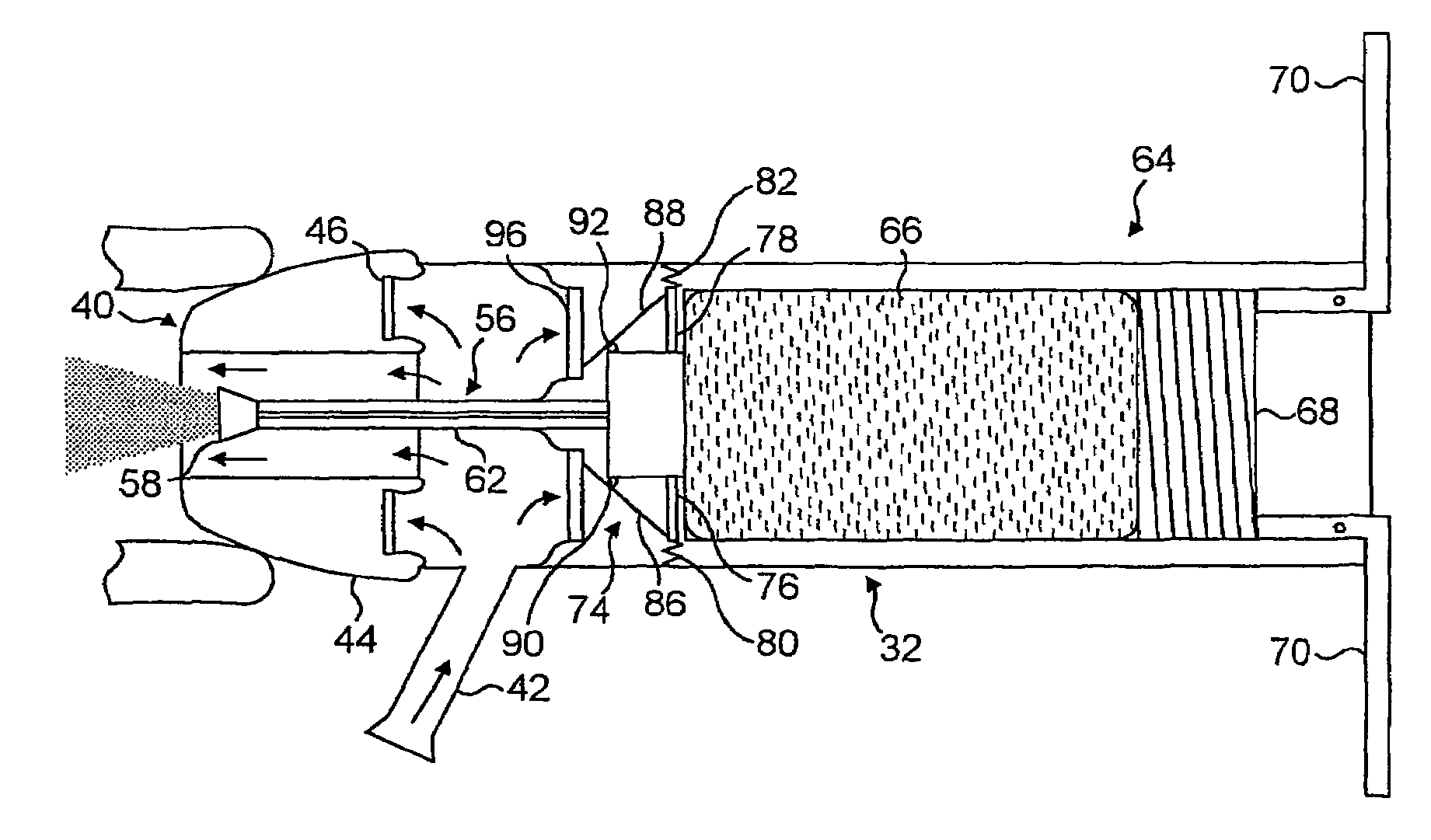
Apparatus and methods for dispersing dry powder medicaments
InactiveUS6543448B1Prevent retractionRespiratorsLiquid surface applicatorsProximateReciprocating motion
A method for aerosolizing a powdered medicament comprises coupling a powder inlet end of a feed tube with a penetration in a receptacle containing the powder. Powder is drawn upward through the tube and dispersed in a high pressure gas stream flowing past a portion of the feed tube. Apparatus comprise the feed tube mounted within a base enclosure proximate a holder for one or more receptacles, which may be in the form of a cartridge containing a plurality of receptacles formed in a continuous web. The cartridge may be reciprocated relative to the feed tube and a separate piercing mechanism in order to sequentially piercing the receptacle and thereafter couple the feed tube through the resulting penetration for extracting the powder. Alternatively, penetration(s) through the receptacle may be formed as the feed tube is coupled, or some penetrations formed prior to coupling with other penetrations formed at the time of coupling.
Owner:NOVARTIS FARMA
Aerosol delivery apparatus with positive expiratory pressure capacity
InactiveUS6848443B2Continuous regulationRespiratorsLiquid surface applicatorsPositive pressureAerosol delivery
An apparatus and method for performing positive pressure (PP) therapy alone or in combination with an aerosol delivery apparatus. The positive pressure apparatus includes a positive pressure valve having a continuously variable respiratory window. The PP valve may be associated with a patient respiratory system interface alone, such as, but not limited to, a mask or mouthpiece, or in combination with an aerosol delivery apparatus.
Owner:TRUDELL MEDICAL INT INC
Programmable multi-dose intranasal drug delivery device
InactiveUS6948492B2Avoid diversionAvoid abuseRespiratorsLiquid surface applicatorsNasal sprayBiological activation
An apparatus and method for the self-administration of a plurality of doses of an intranasal liquid pharmaceutical composition, including opioid analgesics, that includes a drug delivery device containing a plurality of sealed vials, each vial containing a predetermined volume of the pharmaceutical composition, a pump assembly for conveying the liquid pharmaceutical composition from the interior of the vial and discharging it as a nasal spray in response to manual activation by the patient, and programmable means for sequentially advancing a vial to the ready position after passage of a prescribed time interval following the last activation of the delivery device.
Owner:UNIV OF KENTUCKY RES FOUND
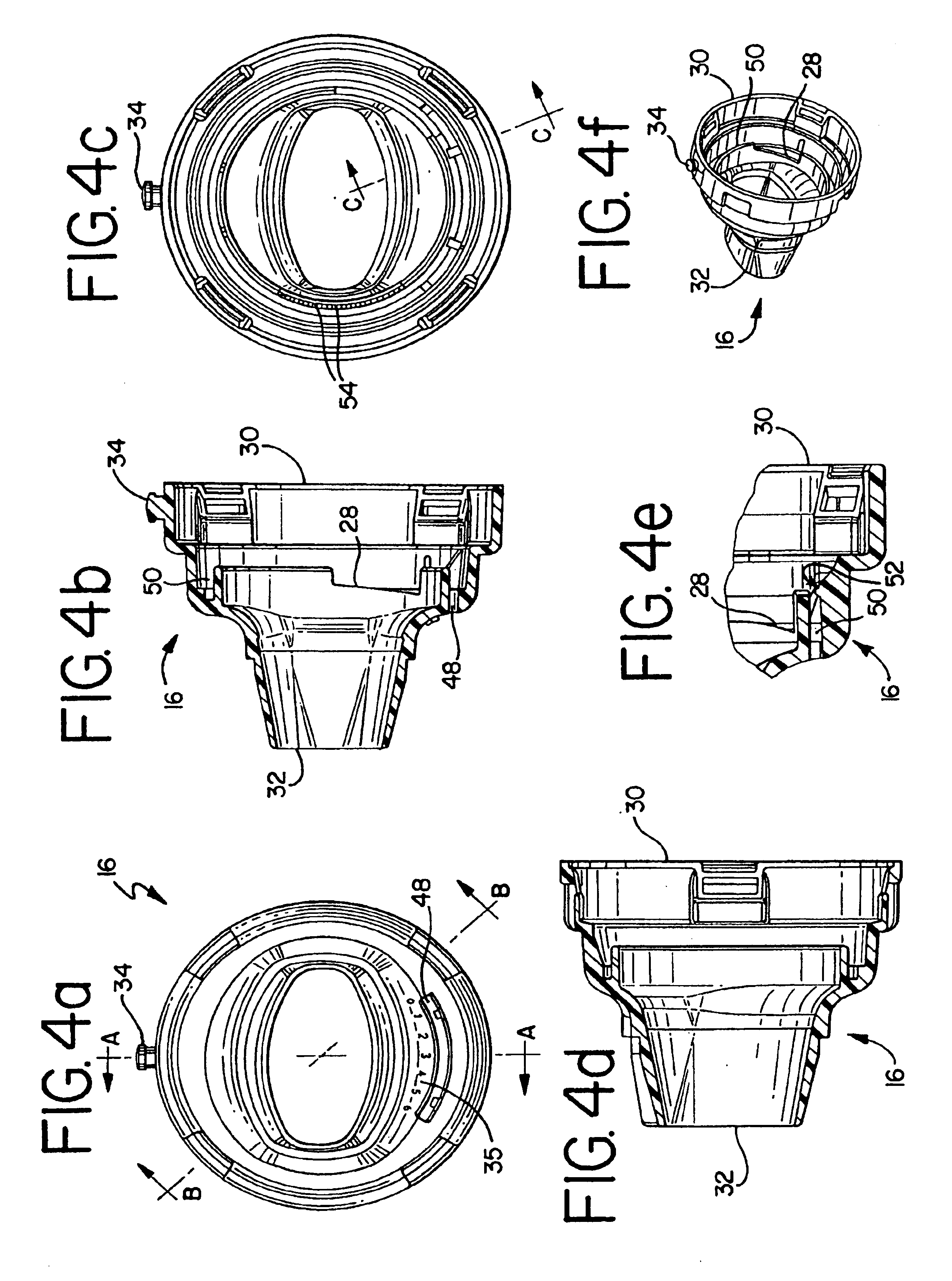
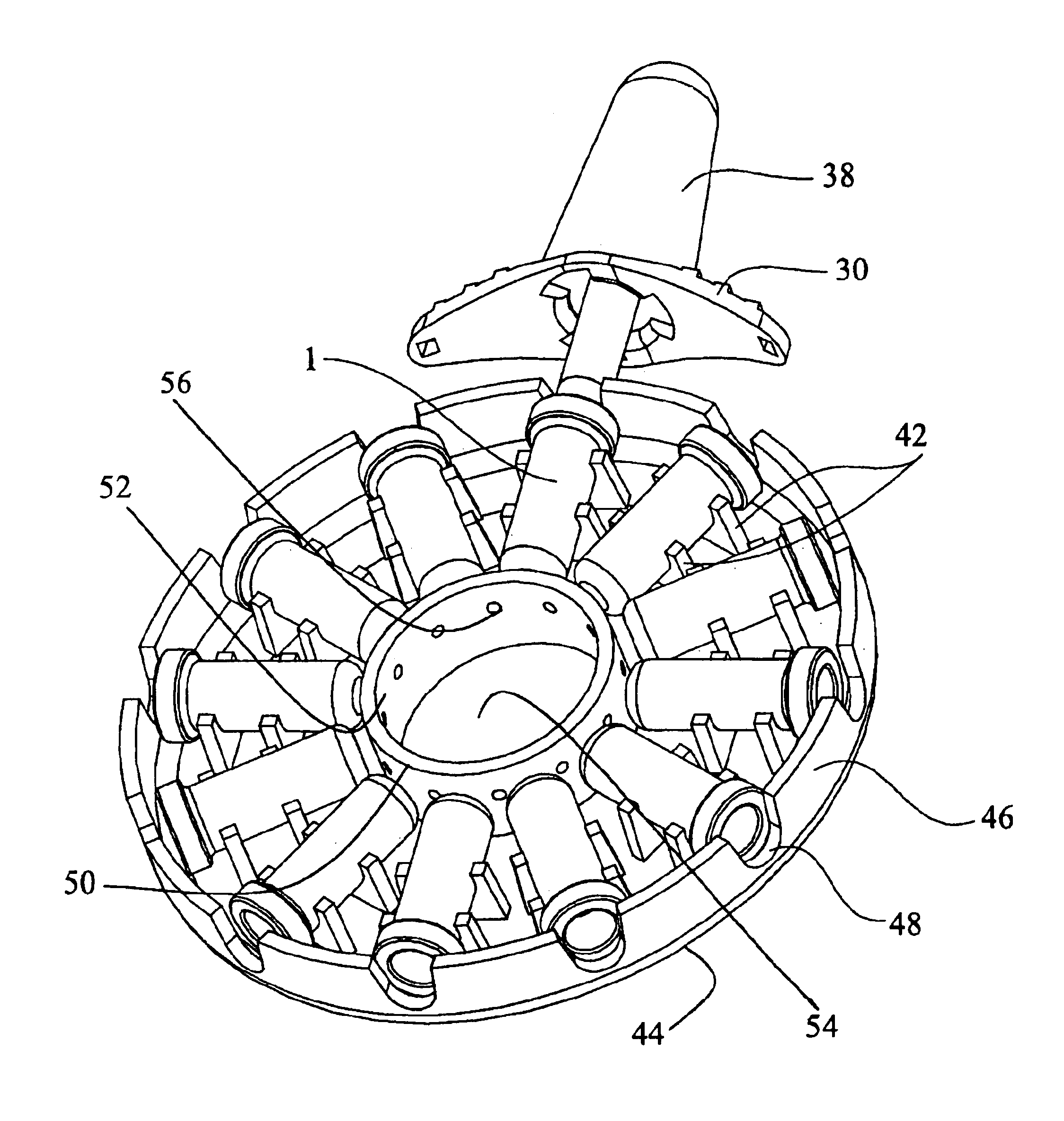
Inhaler
InactiveUS6880555B1Lifting efficiencyGood effectRespiratorsLiquid surface applicatorsInhalationMedicine
An inhaler for medicament in powder form with an opening intended for inhalation. The powder medicament is arranged in the inhaler in a number of enclosures, each enclosure including a specific dose of medicament. A member is provided for enabling access to the dose of medicament. The member is arranged and designed such that it is able to be inserted inside the enclosure and establish at least one outlet passage, between the interior of the enclosure and the inhalation opening, through which outlet passage the medicament is delivered to the patient upon inhalation.
Owner:SHL MEDICAL AG
Drugs And Gene Carrier Particles That Rapidly Move Through Mucous Barriers
ActiveUS20080166414A1Easy adhesionPromote complexationPowder deliveryOrganic active ingredientsCrystallographyMucus
Owner:THE JOHNS HOPKINS UNIVERSITY SCHOOL OF MEDICINE
Inhaler
InactiveUS6234169B1Reduce negative impactHigh simulationRespiratorsLiquid surface applicatorsParticulatesInhalation
An inhaler for use by an individual to inhale a particulate medicament from a reservoir comprises a chamber having a first end connectable to the reservoir to be in air flow communication therewith, a second end for delivering the medicament to the individual upon inhalation and a conduit defining an air flow path extending between the first end and the second end; and, an orifice in the chamber between the first end and the second end, the orifice utilizing the Coanda Effect when the reservoir is in air flow communication with the chamber and upon inhalation by the individual to draw medicament from the reservoir into the air flow path.
Owner:SANSA BARBADOS
Dry powder inhaler
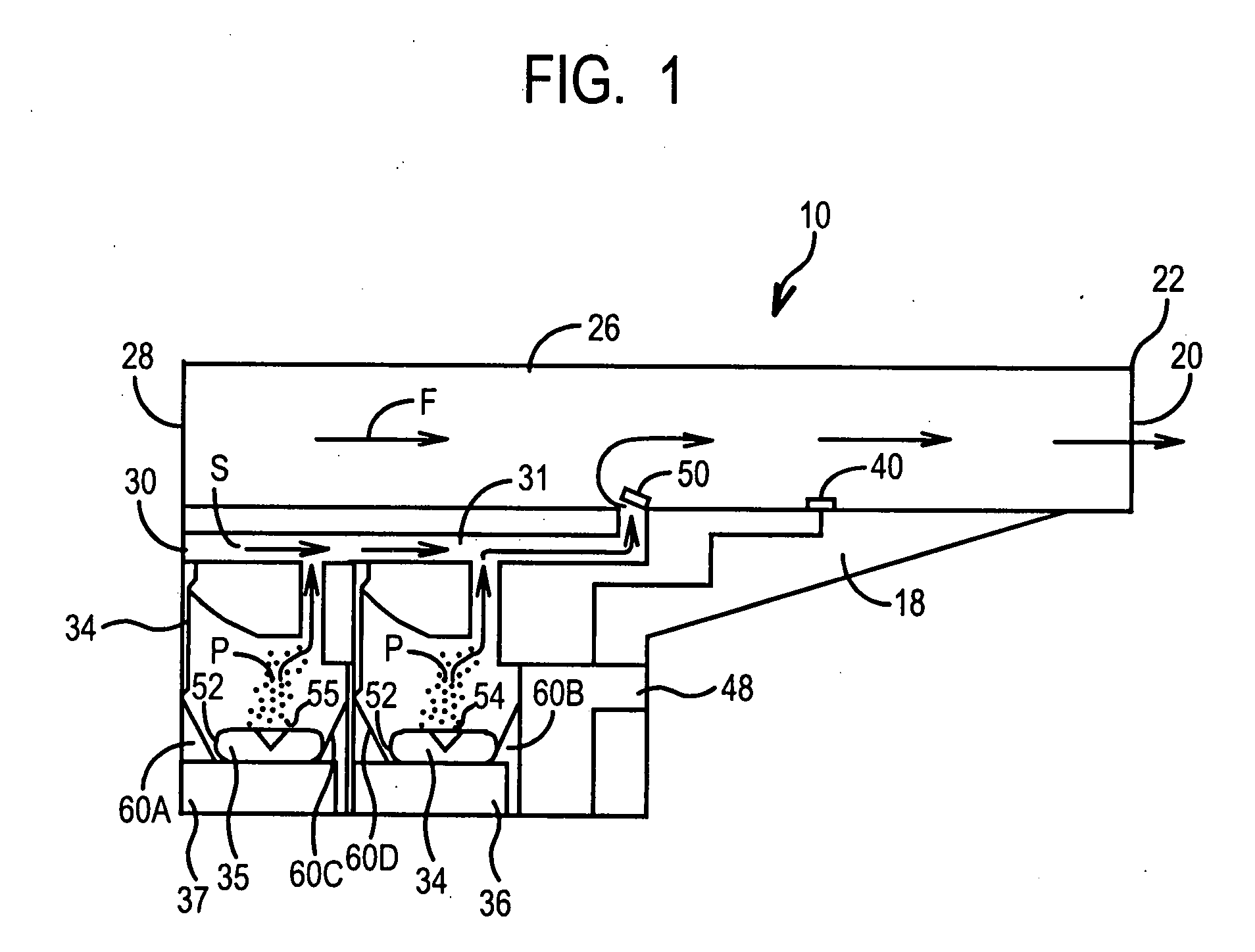
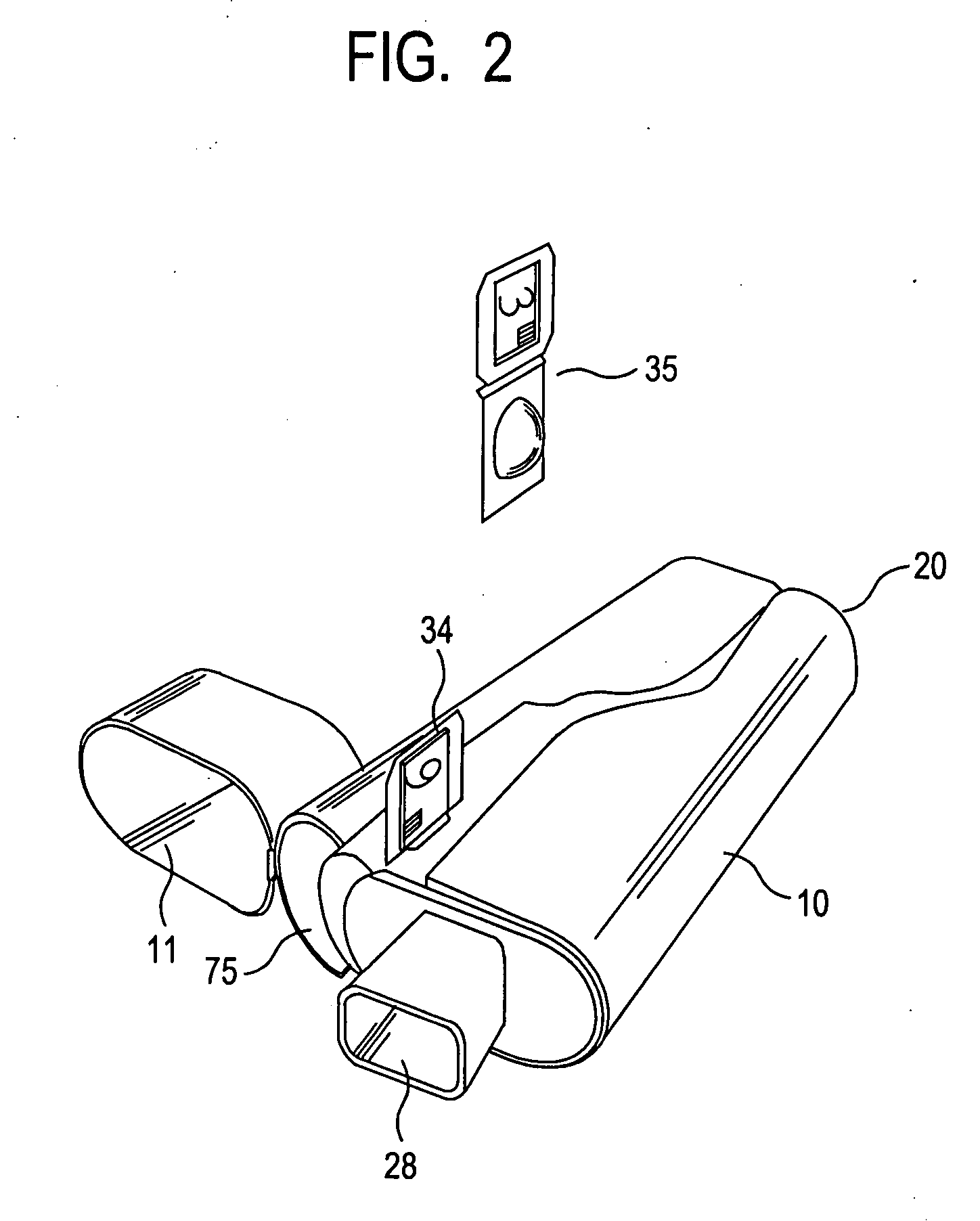
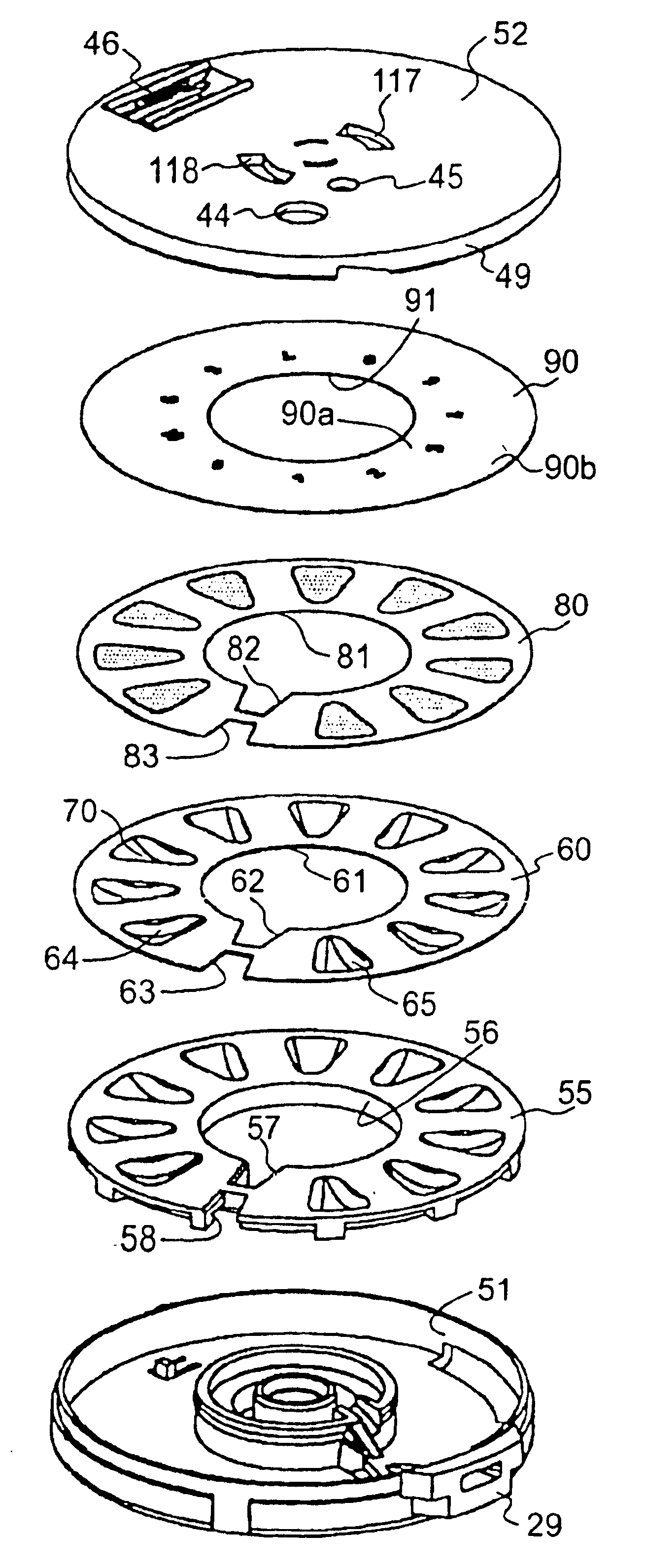
ActiveUS20070235029A1Small accurate volumePrecise deliveryRespiratorsLiquid surface applicatorsMicroDoseAerosolize
A new dry powder inhaler is developed as a pulmonary medicine delivery device for dispersing precise tiny dosages (10 μg-50 mg) of pure carrier-free ultra-fine powdered medicament (<5 μm aerodynamics particle size) into a patient's lung. The powder is drawn from the blister cell and dispersed through an outlet tube assisted by two air streams. The first air stream goes through a the blister cell from its upstream side, to significantly fluidize the medicament in the dose to flow upward. The second one extracts the fluidized powder from downstream of the blister cell for further deagglomeration and dispersion of the medicament powder by shear force. The rotating multi-dose blister can hold up to 60 doses, which are pre-metered with pure ultra-fine powdered medicament. So that it has higher drug loading capability in small volumes, compared to most current dry powder inhalers, which usually use some excipient. The inhaler efficiently disperse the aerosolized medicament in the air stream to the deep interior of patient's lung. The fine particle fraction (<4.7 μm) is reported to reach as high as 80% using this inhaler.
Owner:NINGBO INHAL PHARMA CO LTD
Powder formulation disintegrating system and method for dry powder inhalers
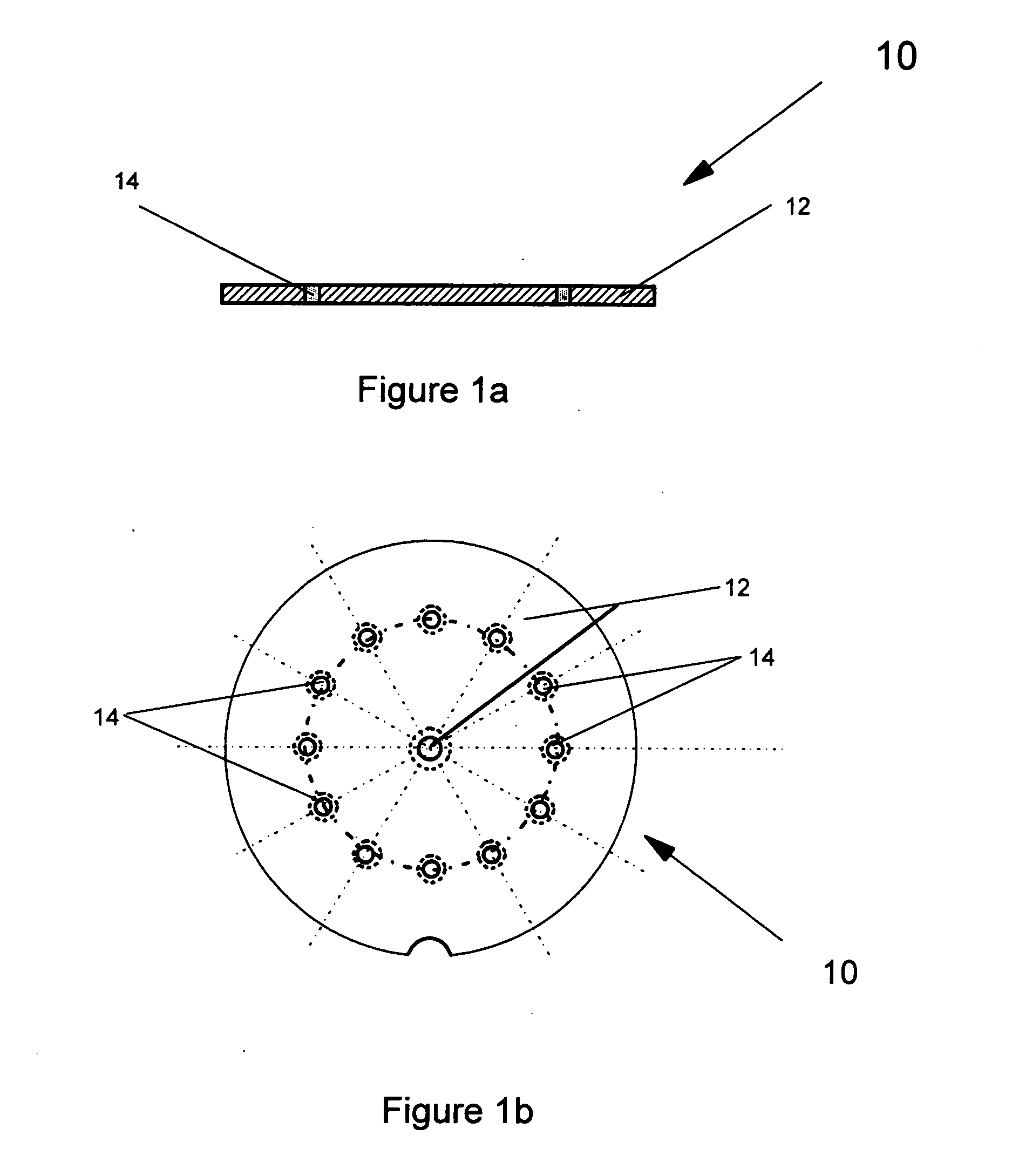
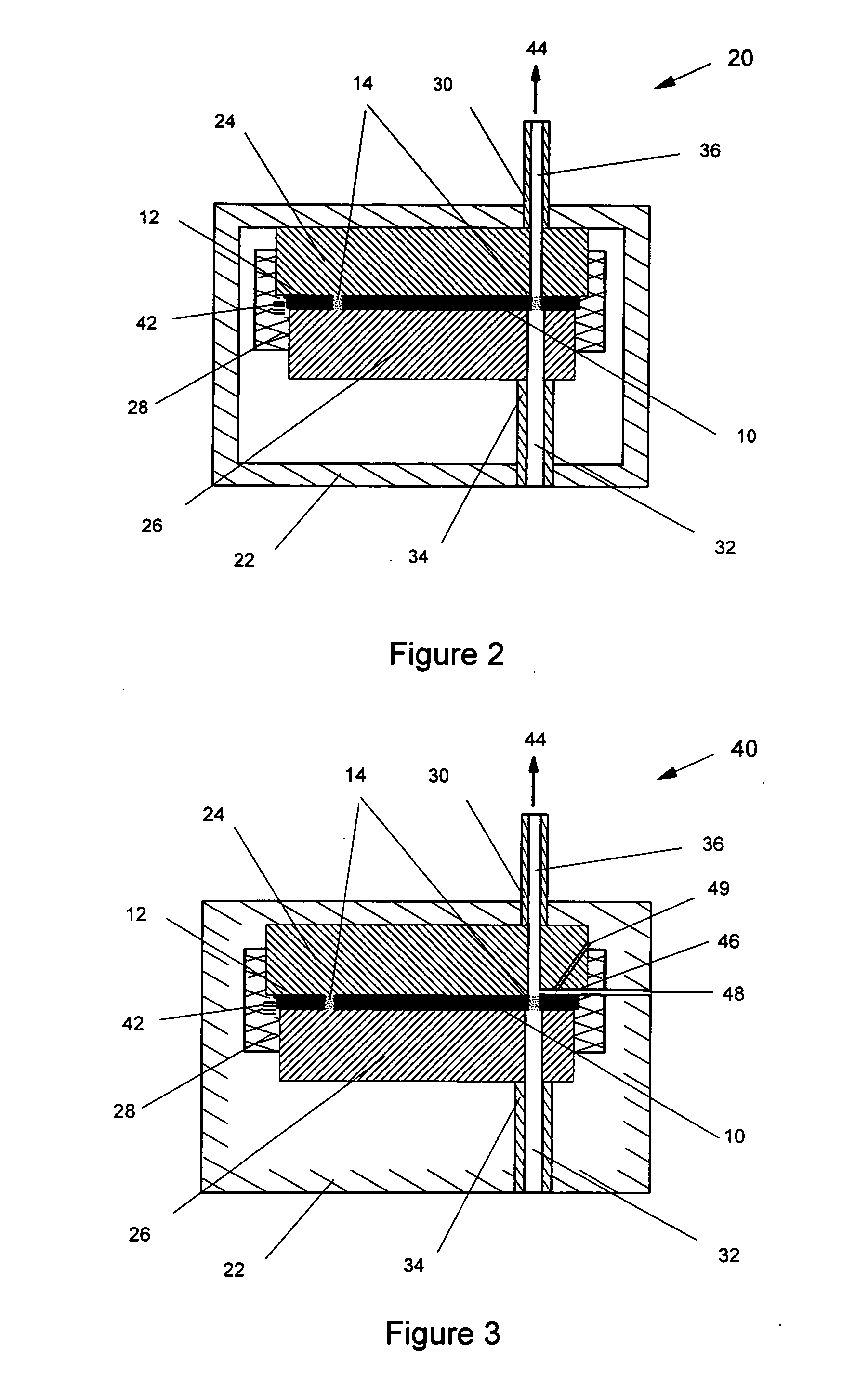
A disperser for dry powders which can be used with different dose systems, dose weights ranging from 2 to 25 mg and different types of powder formulation. In one embodiment, the disperser acts both as a de-agglomeration (disintegration; aerosolization) means and as an air classifier for especially adhesive mixtures. Only fine drug particles are emitted whereas the larger agglomerates and carrier crystals are retained by the disperser. Another embodiment enables time controlled release of carrier crystals in these mixtures. Yet another embodiment has optimized performance with spherical pellets, containing no carrier crystals. Other possible embodiments of the invention make it possible to control the total inhaler resistance and the powder deposition in the upper respiratory tract by means of the addition of a so-called sheath flow of clean air. Modifications also enable carrier retainment in the mouthpiece and elimination of the tangential flow component of the discharge cloud.
Owner:ASTRAZENECA AB
Unit dose cartridge and dry powder inhaler
InactiveUS7464706B2Broad inhalation tidal volume range of human breathMaintain good propertiesRespiratorsLiquid surface applicatorsMedicineCheck valve
A dry powder inhaler having improved aerodynamic properties for diluting, dispersing, and metering drug particles for increasing the efficiency of pulmonary drug delivery to a patient is described. The inhaler comprises, in general, a housing having an air intake, an air flow-control / check-valve, a mixing section and a mouthpiece. A cartridge loaded with a single dose of medicament can be installed in the mixing section.
Owner:MANNKIND CORP
Dry powder inhalers, related blister devices, and associated methods of dispensing dry powder substances and fabricating blister packages
InactiveUS6889690B2Easy to optimizeLimit amount of resistanceSmall article dispensingLiquid surface applicatorsPowder InhalerInhalation
The present invention includes dry powder inhalers and associated multi-dose dry powder packages for holding inhalant formulated dry powder substances and associated fabrication and dispensing methods. The multi-dose package can include a platform body comprising at least one thin piezoelectric polymer material layer defining at least a portion of a plurality of spatially separated discrete elongate dry powder channels having an associated length, width and height; and a metallic material attached to selected portions of the piezoelectric polymer material including each of the regions corresponding to the elongate dry powder channels to, in operation, define active energy releasing vibratory channels. In operation, the elongate channels can be selectively individually activated to vibrate upon exposure to an electrical input.The dry powder inhaler includes an elongate body having opposing first and second outer primary surfaces with a cavity therebetween and having opposing top and bottom end portions and a multi-dose sealed blister package holding a plurality of discrete meted doses of a dry powder inhalable product located in the cavity of the elongate body. The inhaler also includes an inhalation port formed in the bottom end portion of the elongate body, the inhalation port configured to be in fluid communication with at least one of the discrete meted doses during use and a cover member that is pivotably attached to the elongate body so that it remains attached to the body during normal operational periods of use and moves to a first closed position to overlie the inhalation port at the bottom end portion of the body during periods of non-use and moves to a second open position away from the inhalation port during periods of use to allow a user to access the inhalation port.
Owner:ORIEL THERAPEUTICS INC
Nebuliser for the production of aerosolized medication
A nebuliser to deliver a medicament that includes a housing having a reservoir for the medicament, an aerosol generator that can be supplied the medicament from the reservoir, where the generator aerosolizes at least a portion of the medicament into an aerosol, a gas venting inlet to permit a gas to enter the nebuliser and form a mixture with the aerosol, and a passage through which the mixture of the aerosol and the gas is delivered to an outlet port of the nebuliser.
Owner:NOVARTIS AG
Inhalation device
ActiveUS20070151562A1Inhibit migrationRespiratorsLiquid surface applicatorsInhalation DevicesDelivery system
The present invention provides for the integration of drug dispersion methods into a drug or medicine delivery system. The drug dispersion methods used include shear (e.g., air across a drug, with or without a gas assist), capillary flow or a venturi effect, mechanical means such as spinning, vibration, or impaction, and turbulence (e.g., using mesh screens, or restrictions in the air path). These methods of drug dispersion allow for all of the drug in the system to be released, allowing control of the dosage size. These methods also provide for drug metering, fluidization, entrainment, deaggragation and deagglomeration. The present invention also provides for the integration of a drug sealing system into the device. The drug sealing system provides a way of blocking the migration of drug from one area of the package to another. The drug seal system can also provide a method of tightly containing the drug until the package is opened, of directing airflow through the package and of managing and containing the drug during the package / device manufacturing process.
Owner:MANTA DEVICES
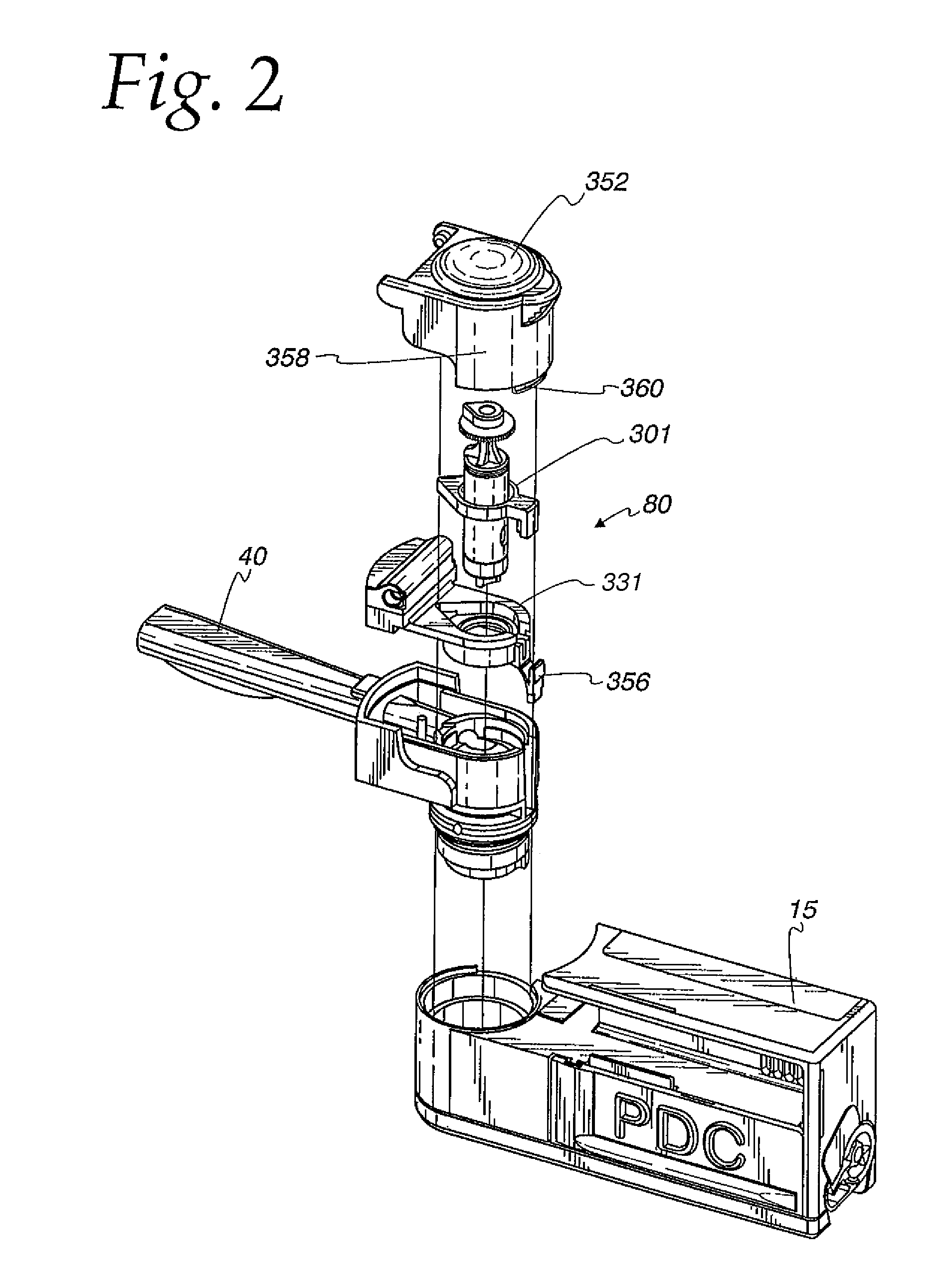
Powdered medication inhaler
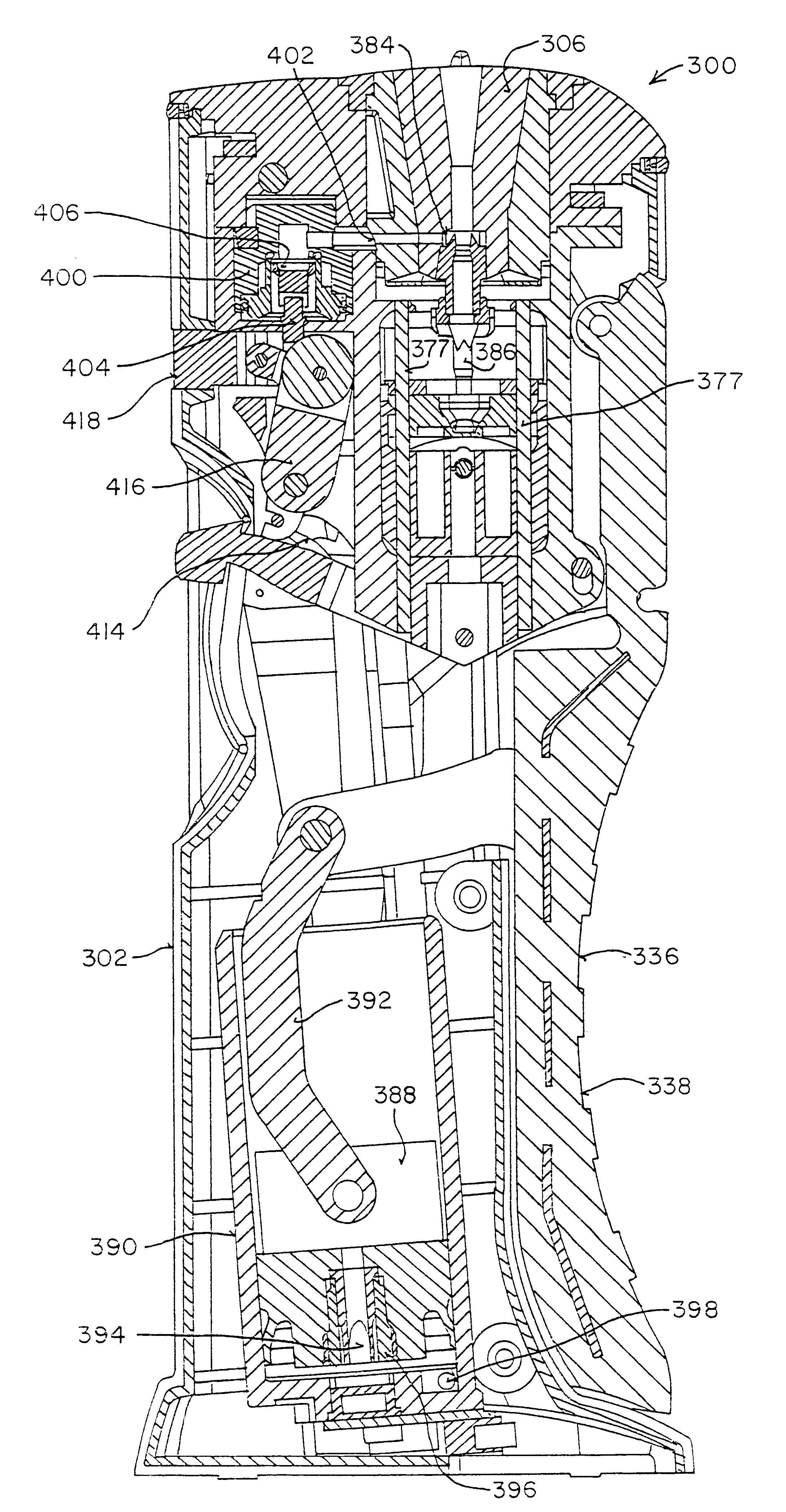
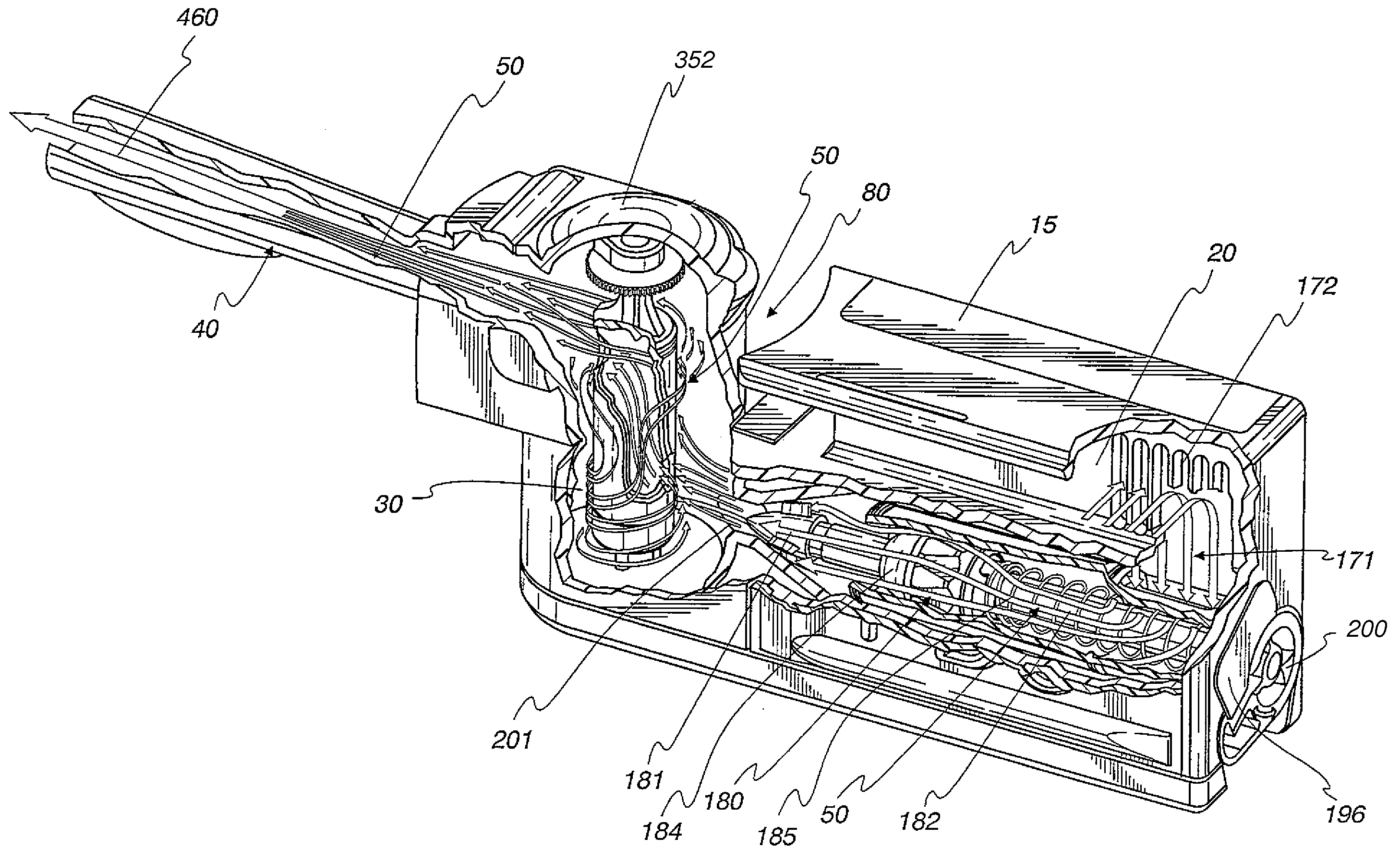
A powder dispenser includes a reservoir body including a supply of powder and an inhalation conduit; a driving body for rotating the reservoir body and including upper recesses and two spring fingers in lower driving recesses thereof; a rotatable metering plate for carrying a metered amount of powder from the supply to the inhalation conduit, and having an underside with ribs; a gas permeable retainer welded to the ribs; a spring biasing the metering plate toward the reservoir body; a nozzle having spiked ribs welded in the upper recesses of the driving body and including a chimney with vertical flutes; an adapter non-rotatably mounted with respect to the metering plate and including two locking recesses for receiving the spring fingers for locking engagement and two helical cam tracks with a square cross-section; a closure cap covering the driving body and including priming ribs biasing the spring fingers out of the locking recesses and engaging with the locking recesses to rotate the driving body, and two cams riding within the cam tracks; a base non-rotatably connected with the metering plate; and a counter rotatably mounted on the base and including rotatable counter rings providing a visual count of the number of doses of powder to be dispensed, and a pawl assembly engaging with gear teeth of the counter rings for rotating the same, the pawl assembly including an outer wall, a pawl and a pawl spring integrally molded as a single piece.
Owner:MERCK SHARP & DOHME CORP
Nasal delivery devices
InactiveUS7347201B2Small particle sizeAvoid inhalationRespiratorsLiquid surface applicatorsNostrilNasal cavity
An exhalation breath-actuated nasal delivery device for and a method of delivering a substance to a nasal cavity of a subject, the delivery device comprising: a nosepiece (40) for fitting to a nostril of a subject; a mouthpiece (42) through which the subject in use exhales; and delivery unit (64), as one of a mechanical delivery pump (66) or a nebulizer (115), for delivering a substance to the nosepiece (40); and an actuation mechanism (74) for actuating the delivery unit in response to oral exhalation through the mouthpiece, and preferably when at least one or both of the pressure at or the flow rate through the nosepiece exceeds a predetermined threshold.
Owner:OPTINOSE INC
Variable dose inhalation device
InactiveUS20070240712A1Easy to manageImprove complianceRespiratorsLiquid surface applicatorsInhalerDrug
Owner:MICRODOSE THERAPEUTX INC
Treatment of Asthma and Chronic Obstructive Pulmonary Disease With Anti-proliferate and Anti-inflammatory Drugs
InactiveUS20080175887A1Promote absorptionOrganic active ingredientsPowdered material dispensingDiseaseObstructive Pulmonary Diseases
Embodiments of the present invention provide a method for treatment of respiratory disorders such as asthma, chronic obstructive pulmonary disease, and chronic sinusitis, including cystic fibrosis, interstitial fibrosis, chronic bronchitis, emphysema, bronchopulmonary dysplasia and neoplasia. The method involves administration, preferably oral, nasal or pulmonary administration, of anti-inflammatory and anti-proliferative drugs (rapamycin or paclitaxel and their analogues).
Owner:LUTONIX INC
Inhaler
An inhaler for delivering metered doses of powdered medicament, the inhaler having a plurality of compartments spaced in an array and each arranged to contain a metered dose of the medicament, a lever to displace the compartments one by one into line with an inhalation aperture that constitutes a mouthpiece, each compartment including inner and outer edges, the plurality of compartments being closed by a sealing layer, the inhaler further having a mechanism adapted to lift the sealing layer off the inner and outer edges of the compartment to open an air passageway defined by the compartment and the sealing layer so that, in use, on inhalation through the mouthpiece, air flow in the air flow passageway picks up and entrains the powder in the compartment to be drawn with the air out of the inhaler through the mouthpiece.
Owner:ADVENT PHARMA PTY LTD
Aerosol processing and inhalation method and system for high dose rate aerosol drug delivery
ActiveUS20070144514A1Increase dose rateRisk minimizationRespiratorsLiquid surface applicatorsSolvent vaporCounter flow
A method and system is disclosed which is capable of delivering at a high dose rate, respirable solid aerosols derived from aqueous- or nonaqueous-based solutions containing the desired therapeutic agent(s). The method and system comprises the integration of an aerosol generator, an aerosol evaporator, an aerosol concentrator, and an aerosol flow regulator. The aerosol generator generates 10-30 μm droplets, with a narrow size distribution. The aerosol jet is arrested by a coaxial counter-flow heated air jet, and evaporated rapidly by annular swirling heated air. Most of the air, together with the unwanted solvent vapor, is removed from the aerosol stream during the process of aerosol concentration. The output aerosol carries the dry particles to be inhaled by the patient. The respiratory-governed control of aerosol fluid generation system delivers fluid containing the test agent of interest (drug or toxin) to the aerosol generator throughout inhalation.
Owner:KAER BIOTHERAPEUTICS CORP
Portable ventilator system
ActiveUS20060144396A1Reduce power consumptionSmall sizeLiquid surface applicatorsPowdered material dispensingPortable ventilatorsEngineering
A portable ventilator uses a Roots-type blower as a compressor to reduce both the size and power consumption of the ventilator. Various functional aspects of the ventilator are delegated to multiple subassemblies having dedicated controllers and software that interact with a ventilator processor to provide user interface functions, exhalation control and flow control servos, and monitoring of patient status. The ventilator overcomes noise problems through the use of a noise attenuating system comprising noise reducing pressure compensating orifices on the Roots blower housing and multiple noise reducing chambers. The ventilator is configured with a highly portable form factor, and may be used as a stand-alone device or as a docked device having a docking cradle with enhanced interface and monitoring capabilities.
Owner:VYAIRE MEDICAL 203 INC
Nasal Delivery Devices
InactiveUS20080289629A1Block deliveryReduce the impactRespiratorsLiquid surface applicatorsNasal cavityMoisture
A nasal delivery device for and method of delivering substance to a nasal cavity of a subject, the delivery device comprising: a container-receiving unit comprising a container chamber for receiving a substance-containing container which contains substance to be delivered to the nasal cavity of the subject, the container chamber including an inlet and an outlet; a nosepiece unit including a nosepiece for fitting to a nasal cavity of the subject and being in fluid communication with the outlet of the container chamber; a mouthpiece unit including a mouthpiece in fluid communication with the inlet of the container chamber and through which the subject in use exhales, such as to entrain substance from the container and deliver the same through the nosepiece; and moisture-mitigation means for mitigating an effect of moisture in an exhaled breath on the entrainment of substance from the container, which means are provided, for example, by providing the container in a replaceable container-containing member, by a pressure-sensitive valve which normally closes the fluid connection between the container chamber and the mouthpiece, and a temperature regulator upstream of the container chamber.
Owner:OPTINOSE AS
Apparatus and methods for handling materials in a 3-D printer
ActiveUS20080006334A1Improve efficiencyProcess uneconomicalLiquid surface applicatorsPowdered material dispensingComputer printing3d printer
The present invention is directed towards methods and apparatus for handling powder in a 3D printer. The invention includes a means of transporting powder from multiple sources to a powder dispensing apparatus with minimal user intervention, thus reducing contamination of the 3D printer and surrounding area with loose powder, while also providing a means of improving the recycling of powder for re-use.
Owner:3D SYST INC
Powder inhaler
InactiveUS6230707B1Good dispersion propertiesEasily reaches lungRespiratorsLiquid surface applicatorsPowder InhalerEngineering
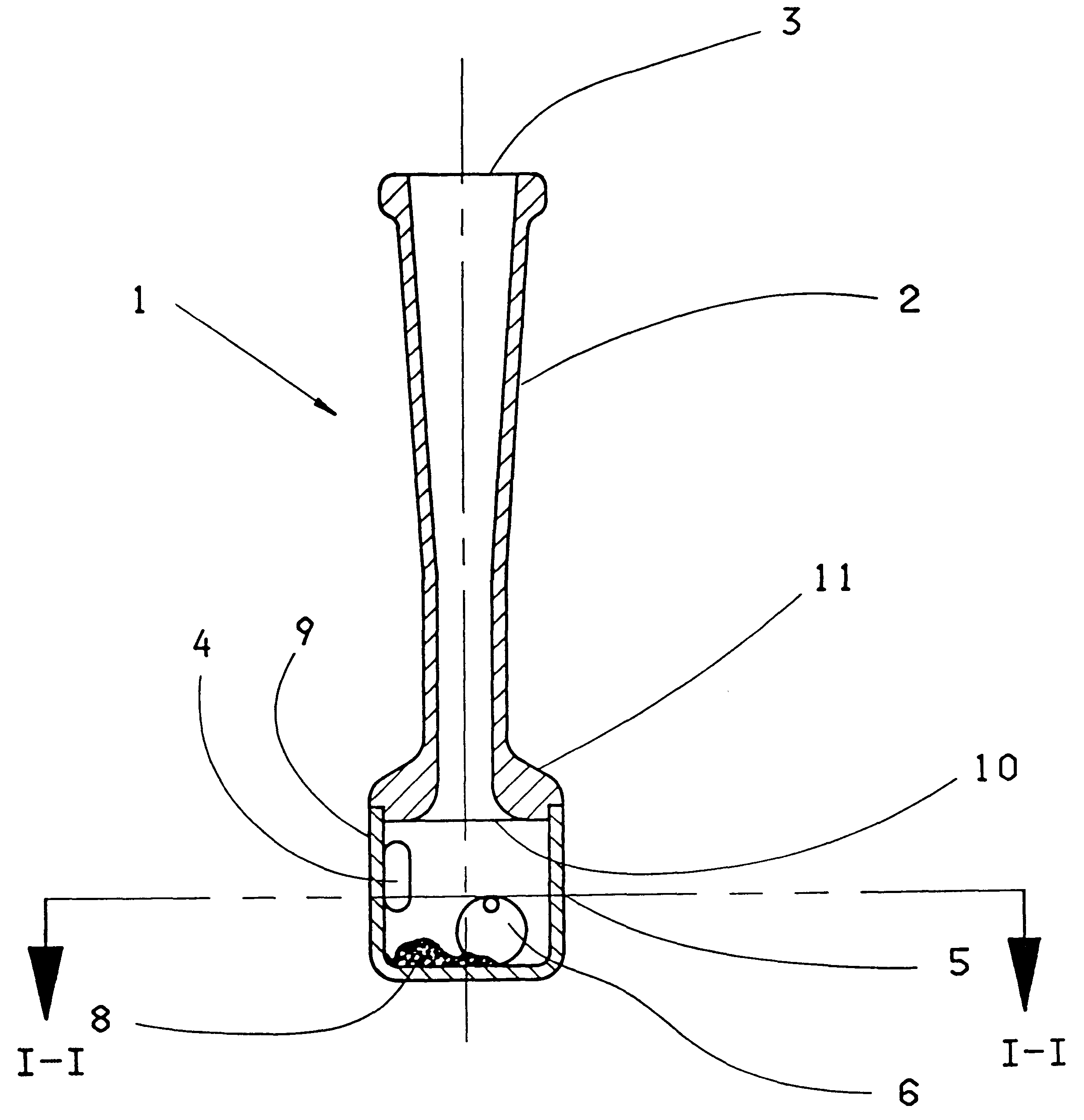
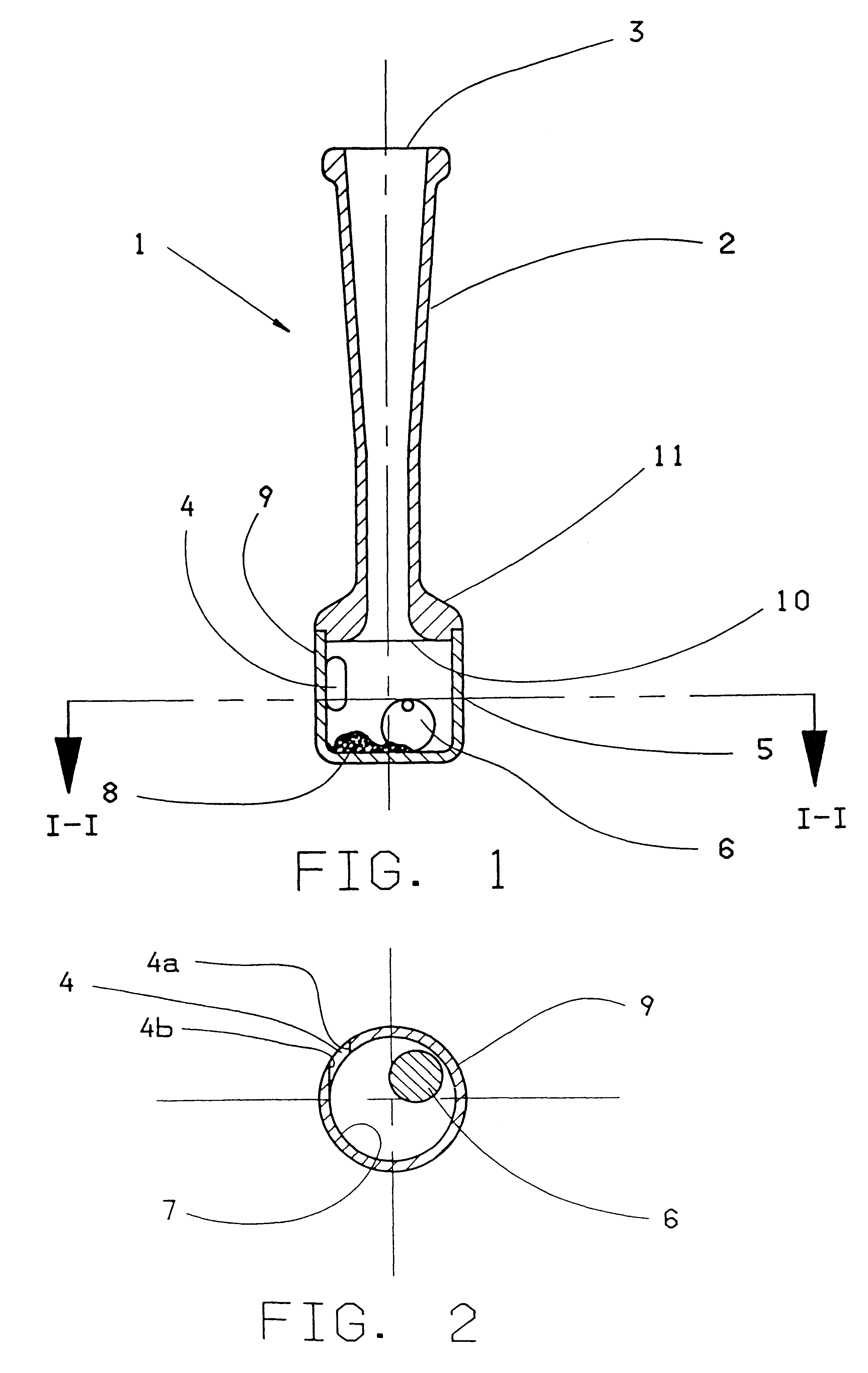
The invention concerns an inhaler device (1) having a hollow tubular member (2, 102, 402) connected to a chamber (5, 105, 405). The tubular member has a first opening (3, 103, 203) at one end through which air can be sucked and the chamber (5, 105, 405) has a hole (4, 104, 204) therein for entry of air. When air is sucked through the first opening, air enters the chamber through the hole swirls and moves towards the first opening (3, 103, 203). To maintain or increase the swirling effect of the air a single restriction (10, 110) is arranged between the opening and the hole (4, 104, 204). A powdered substance (8) within the chamber is picked up by the swirling air within the chamber and is uniformly and finely divided by the swirling effect of the air. The effect can be enhanced by adding a movable element (6) such as a ball inside the chamber (5) and / or by providing a central core element (112, 412) inside the chamber.
Owner:AVENTIS PHARMA LTD
Medicament dispenser
InactiveUS7191777B2Readily and accurately transferableAssist detectionLiquid surface applicatorsPowdered material dispensingMedication DispenserBody shape
A medicament dispenser for use in the storage, presentation and / or dispensing of medicament comprising a body shaped for receipt of a medicament container. In association with the body, there is a first transceiver for transmitting and receiving data. A medicament container is receivable by the body. In association with the medicament container, there is a second transceiver for transmitting and receiving data. Data is transferable in two-way fashion from the first transceiver to the second transceiver. The medicament dispenser may be supplied in kit of parts form.
Owner:GLAXO GROUP LTD
Inverted aerosol dispenser
An inverted aerosol dispensing device is disclosed having an undercap rotatably secured to the aerosol container with a bottom portion of the undercap supporting the aerosol container on a supporting surface to store the aerosol dispensing device in an inverted position. The undercap is rotatable into a first rotational position for enabling an actuator to discharge an aerosol product in a generally downwardly direction. The undercap is rotatable into a second rotational position for inhibiting the actuator from moving the valve stem.
Owner:SEAQUIST PERFECT DISPENSING FOREIGN INC
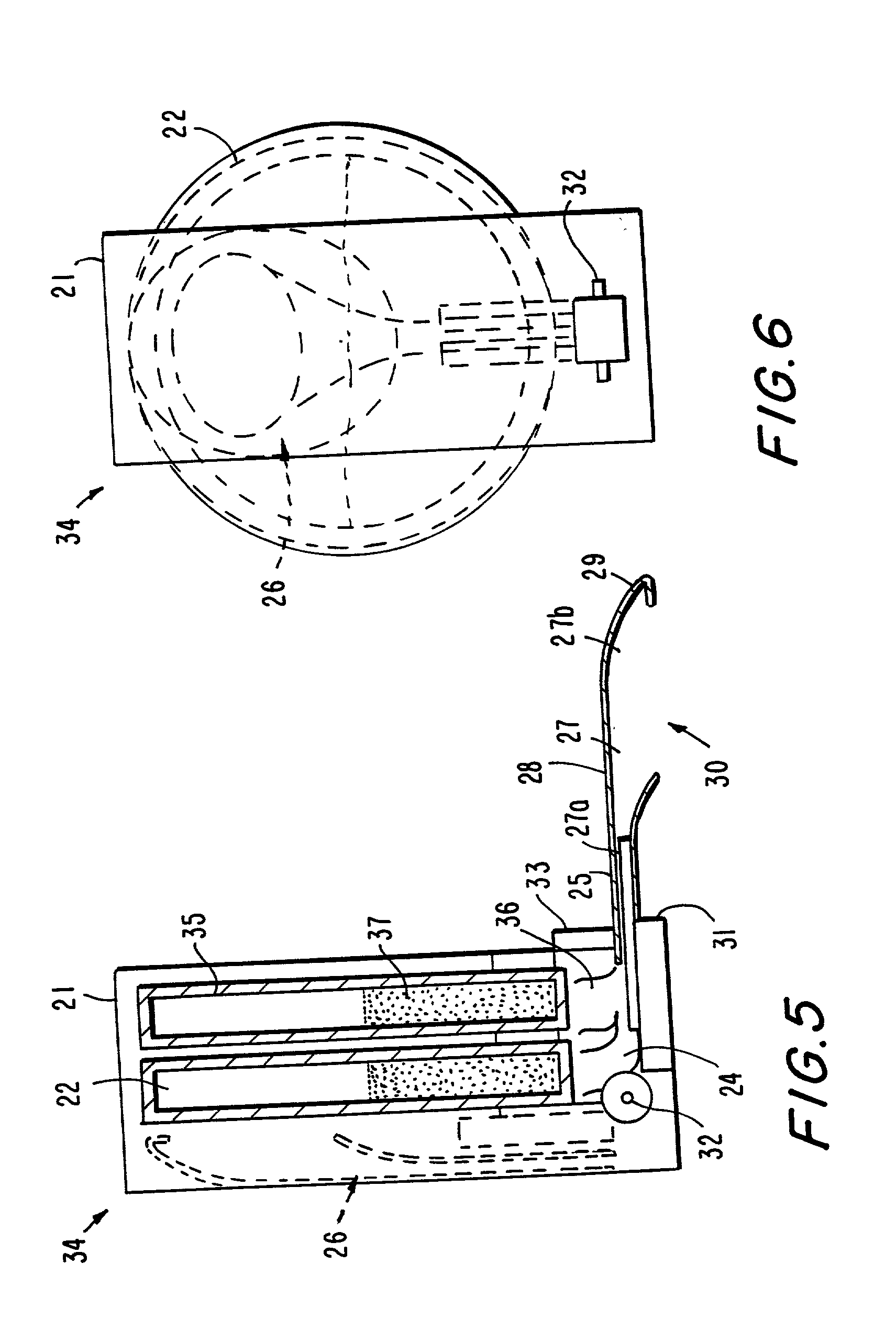
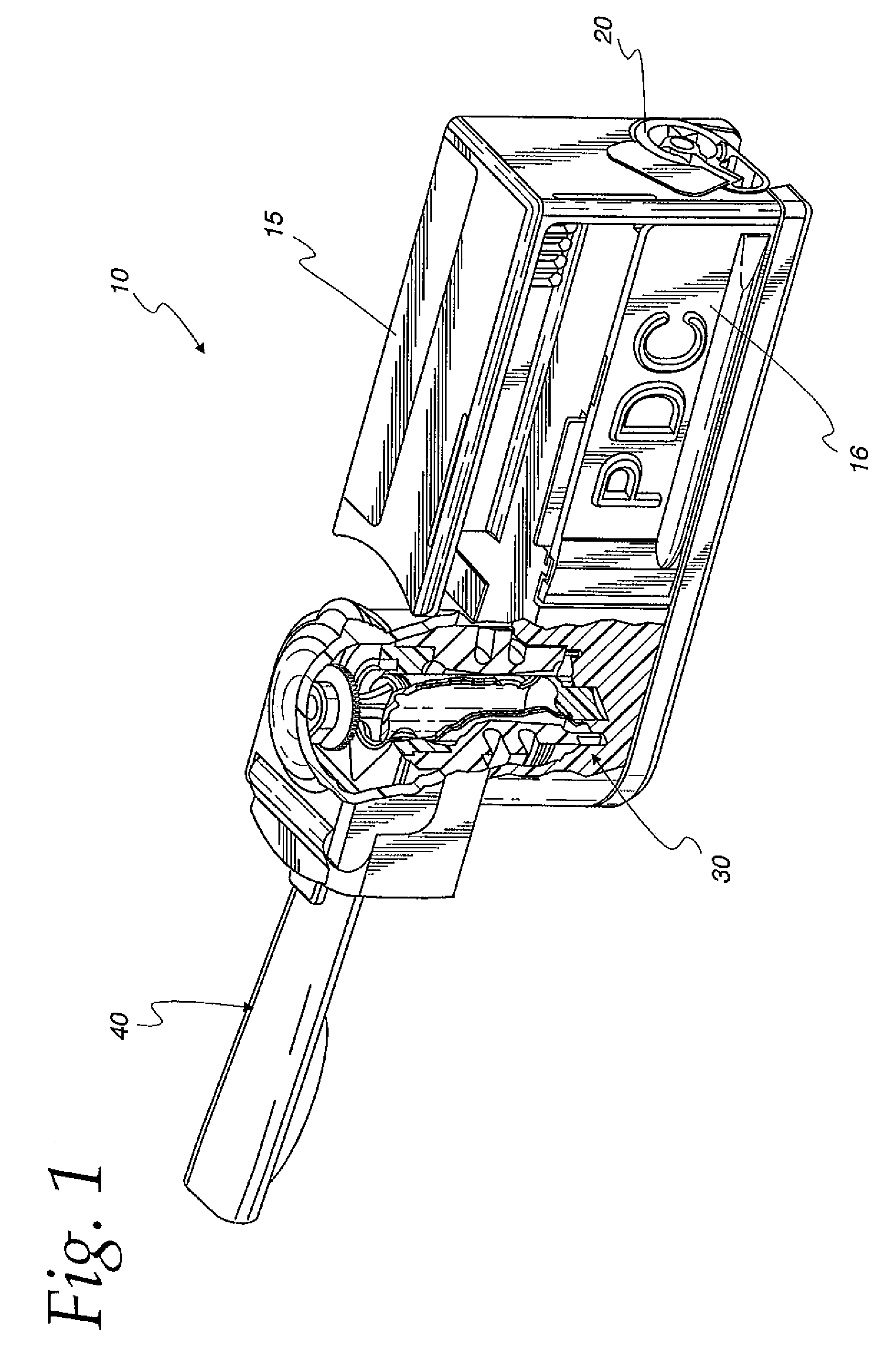
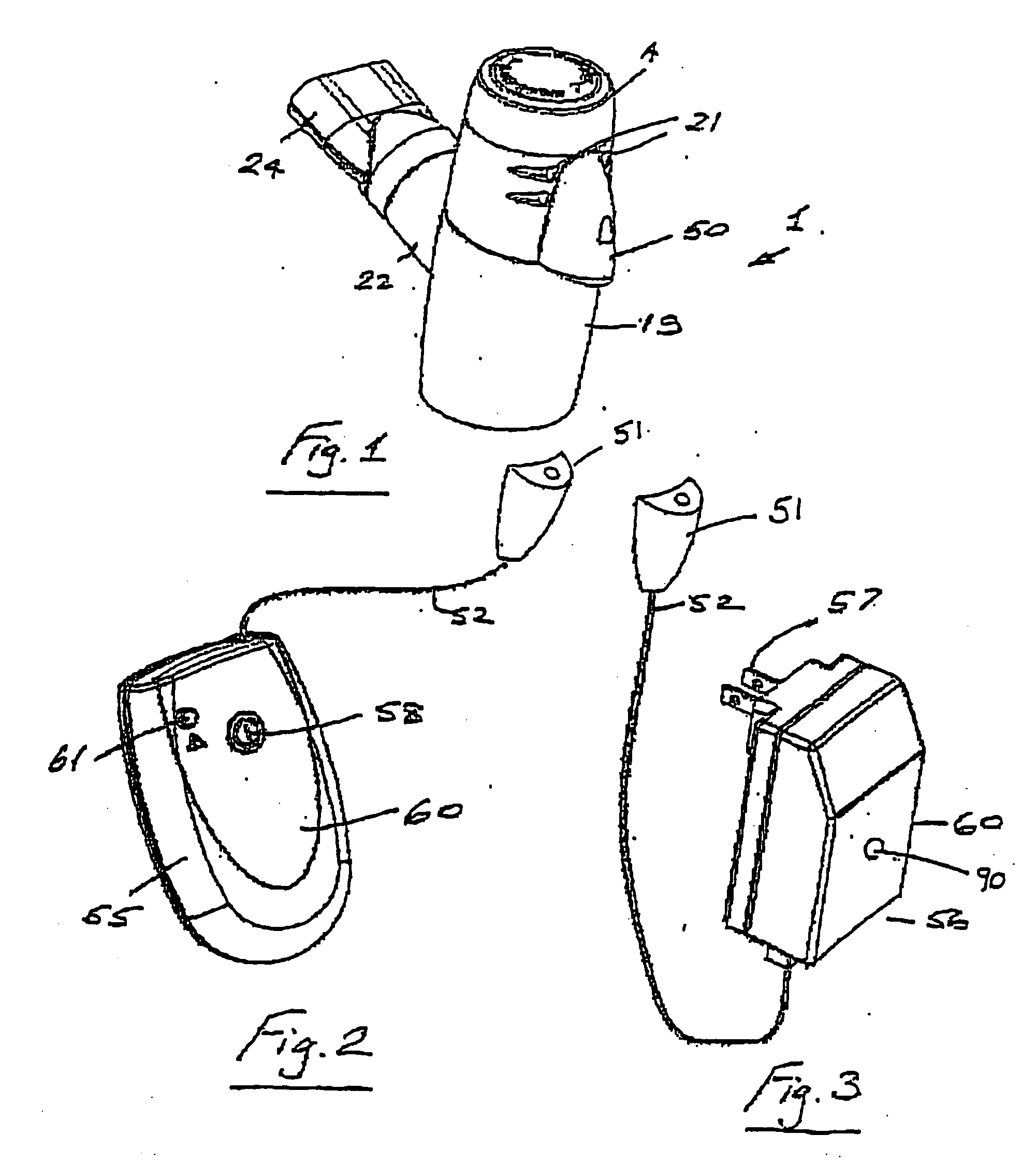
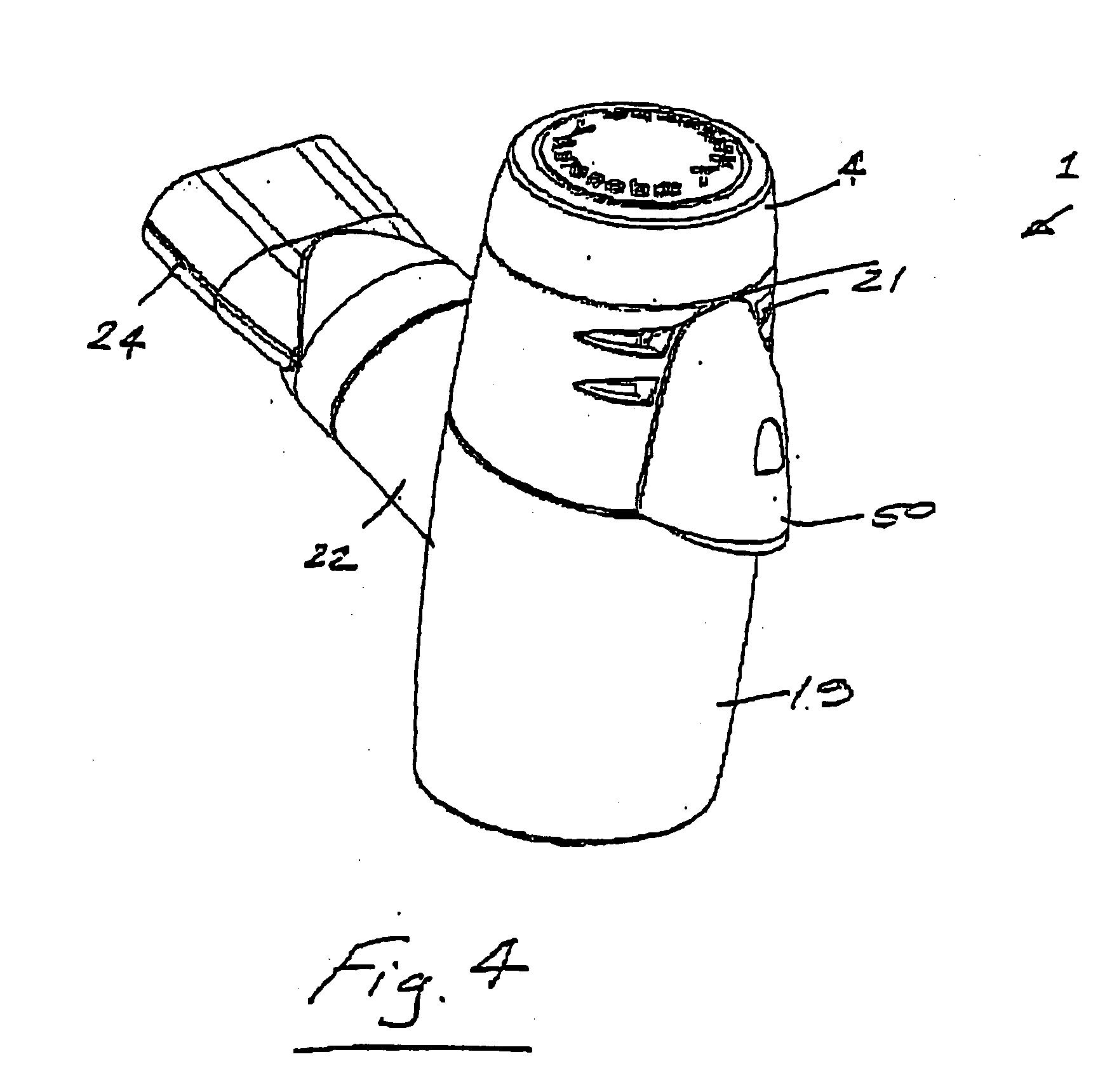
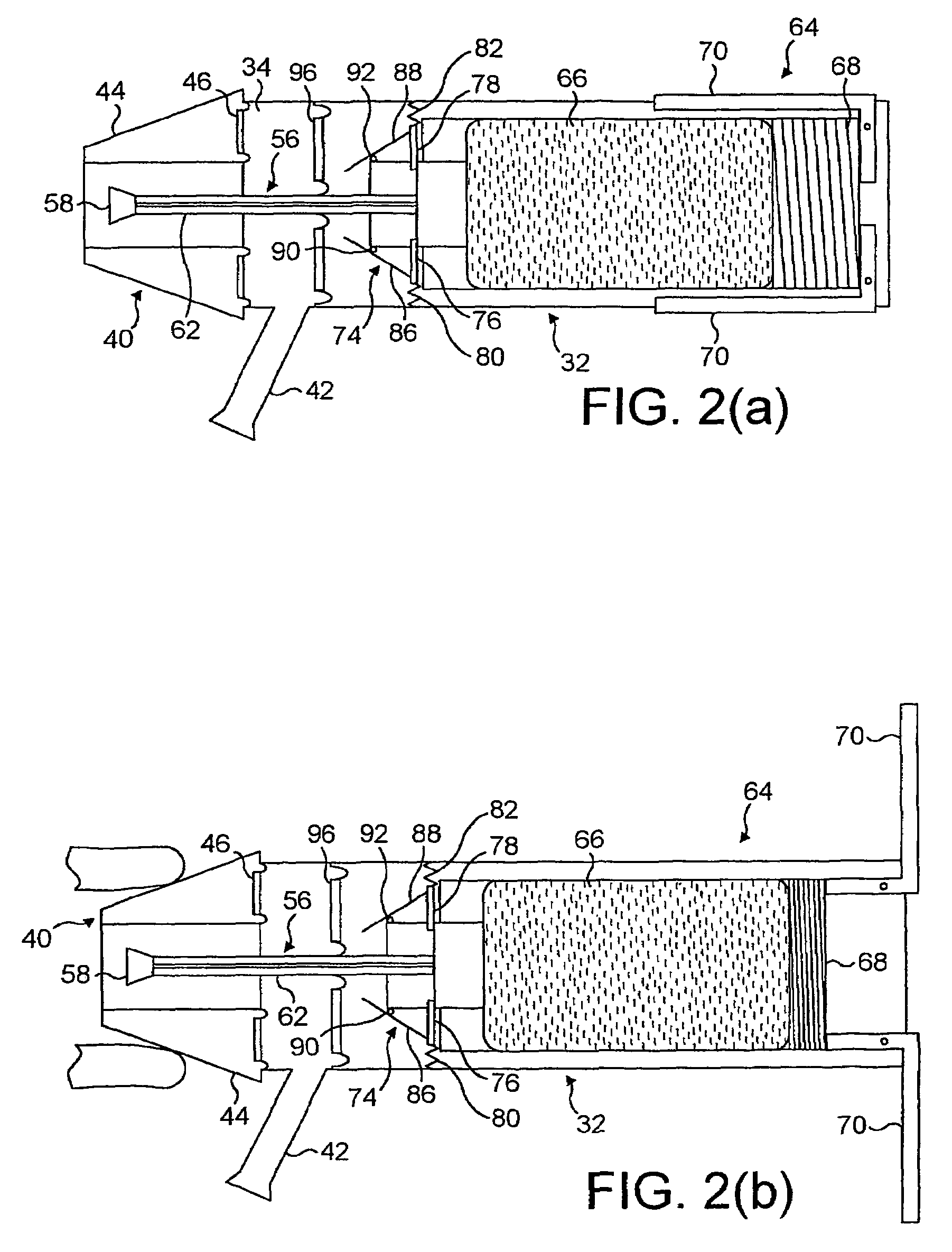
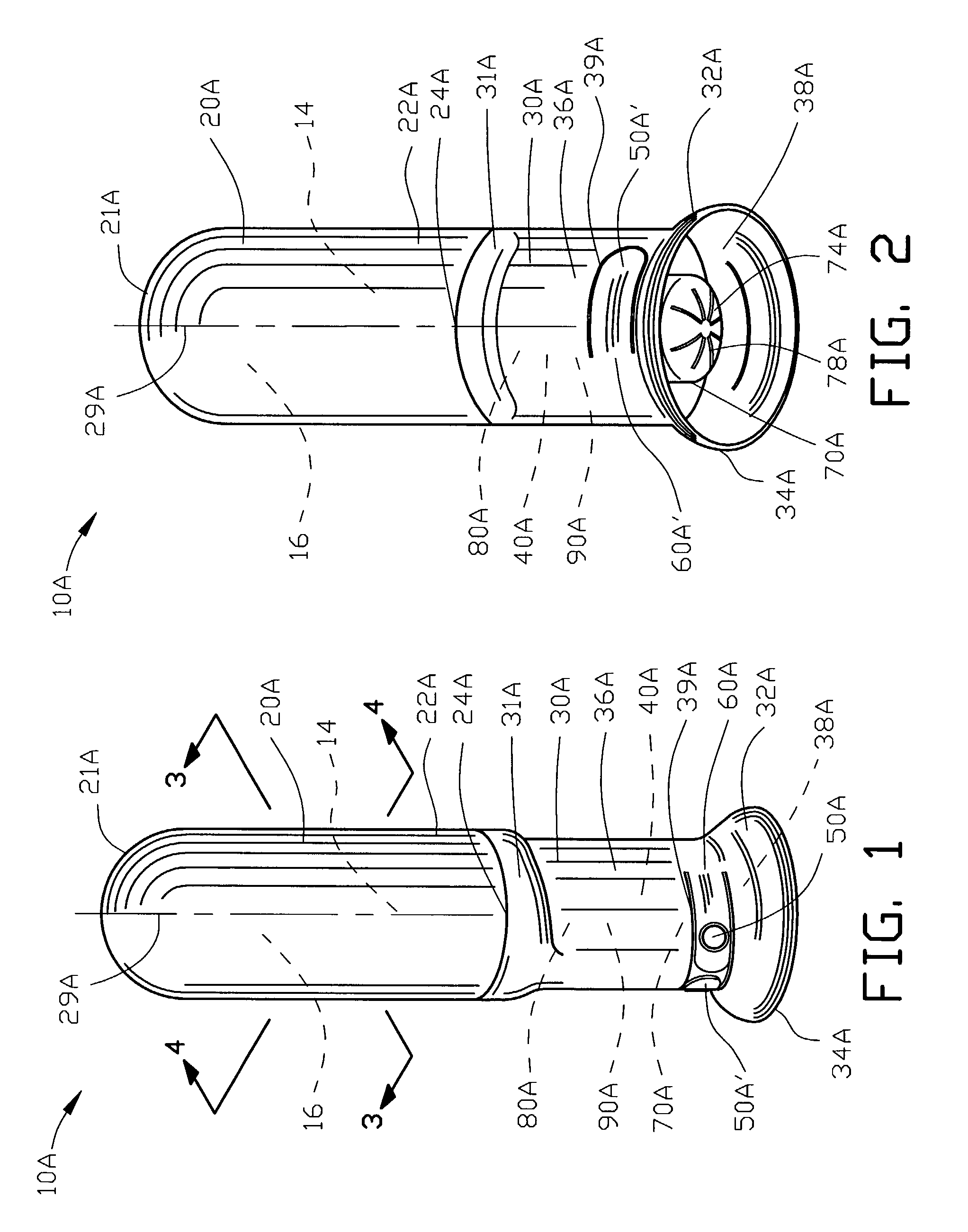
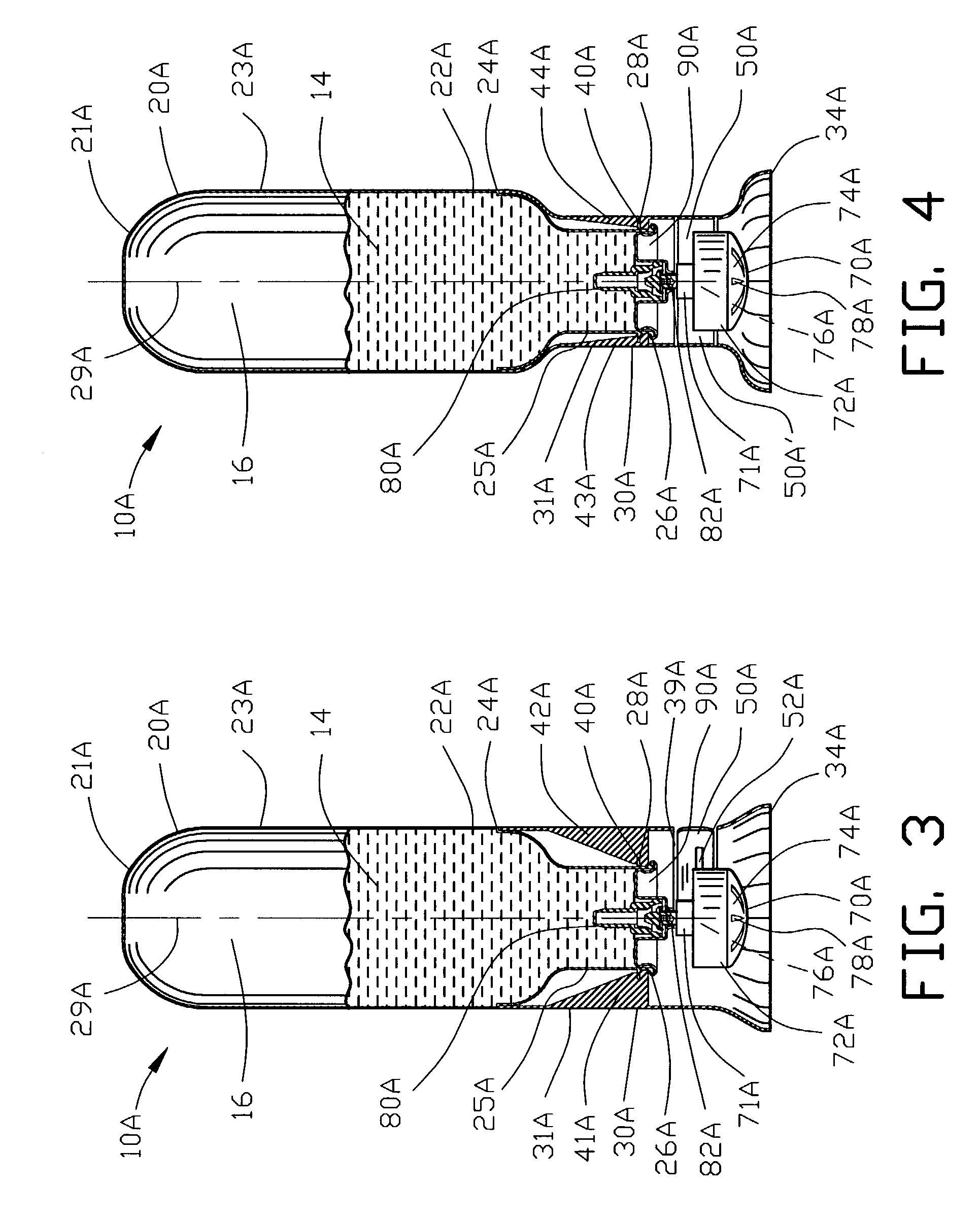
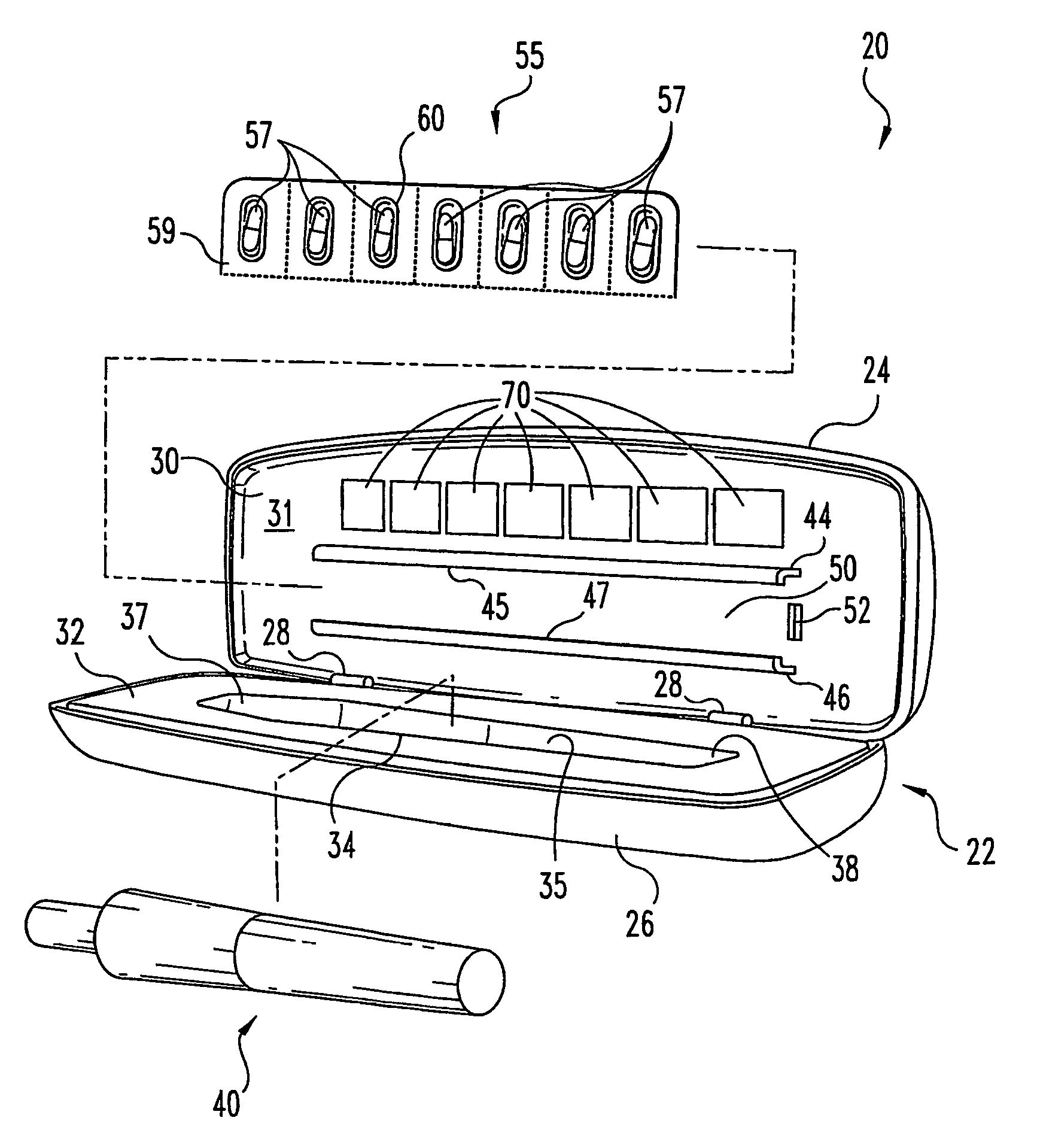
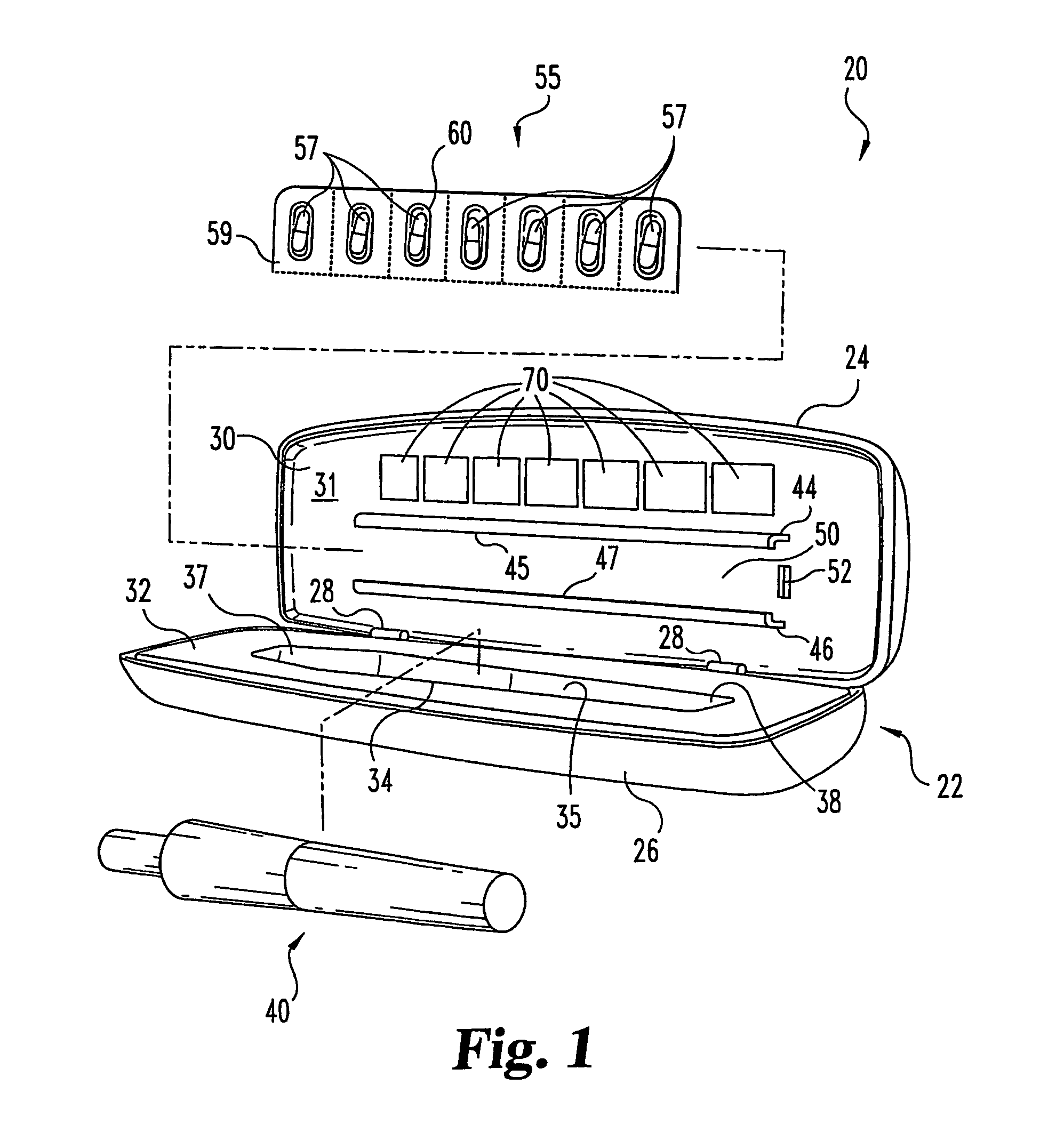
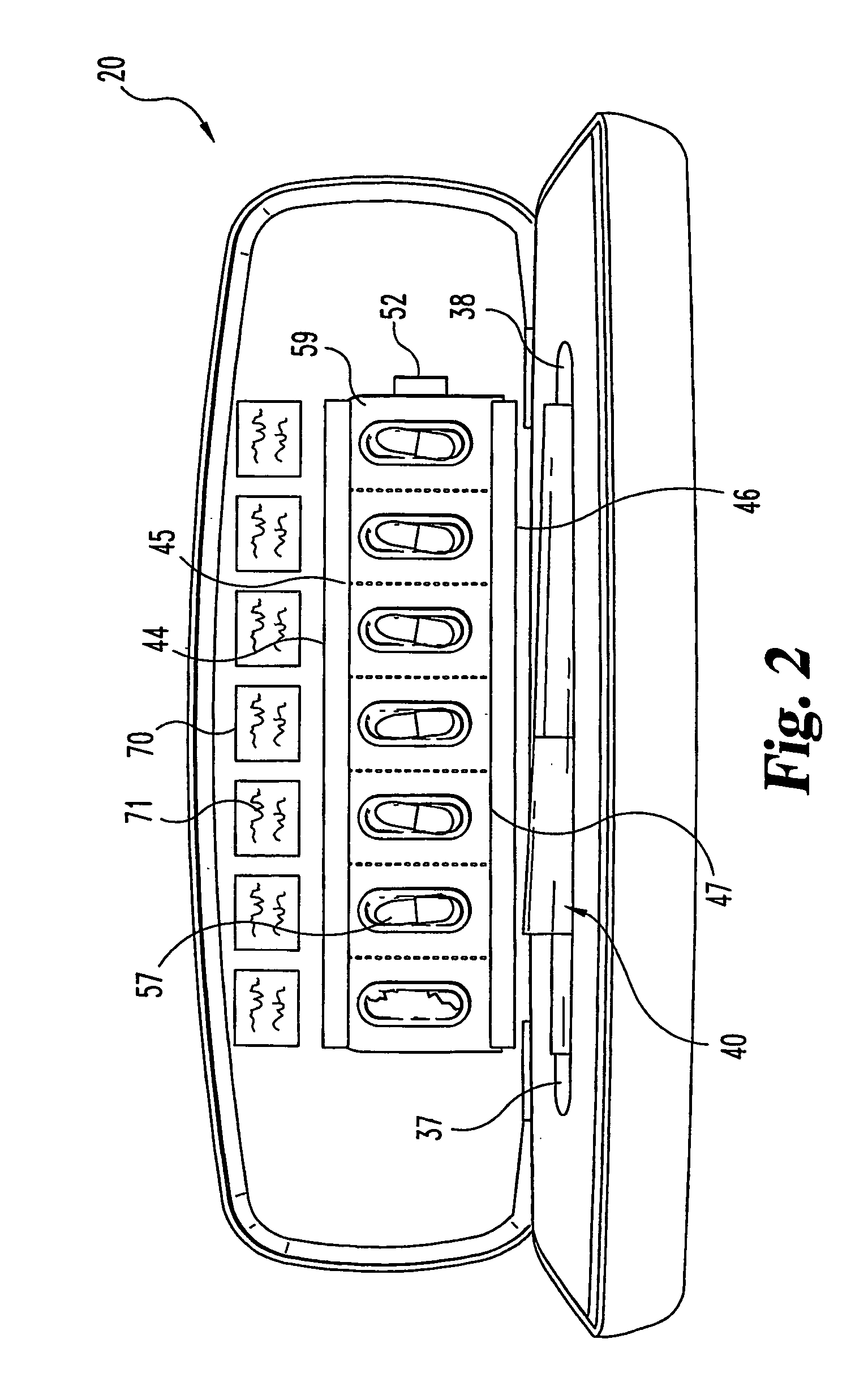
Portable medication inhalation kit
InactiveUS7093595B2Easy and intuitive to useCost-effectiveLiquid surface applicatorsPowdered material dispensingGuidance systemMedication dose
A portable kit (20) in which is conveniently retained the materials required for a user to pulmonarily delivery inhaleable medication. The kit (20) includes a carrying case (22) in which a plurality of individual doses (5) of an inhaleable medication are removably mounted. An inhaler (40) is also mountable in the case (22), and when removed from the case (22) is operable to pulmonarily administer one of the medication doses (57) loaded by a user into the inhaler (40). A dosing guidance system (70) is also provided within the case (22) in registry with the plurality of individual doses (57) of medication, which dosing guidance system (70) provides a quick, visual reminder to assist the user in taking doses as prescribed by her physician.
Owner:ELI LILLY & CO