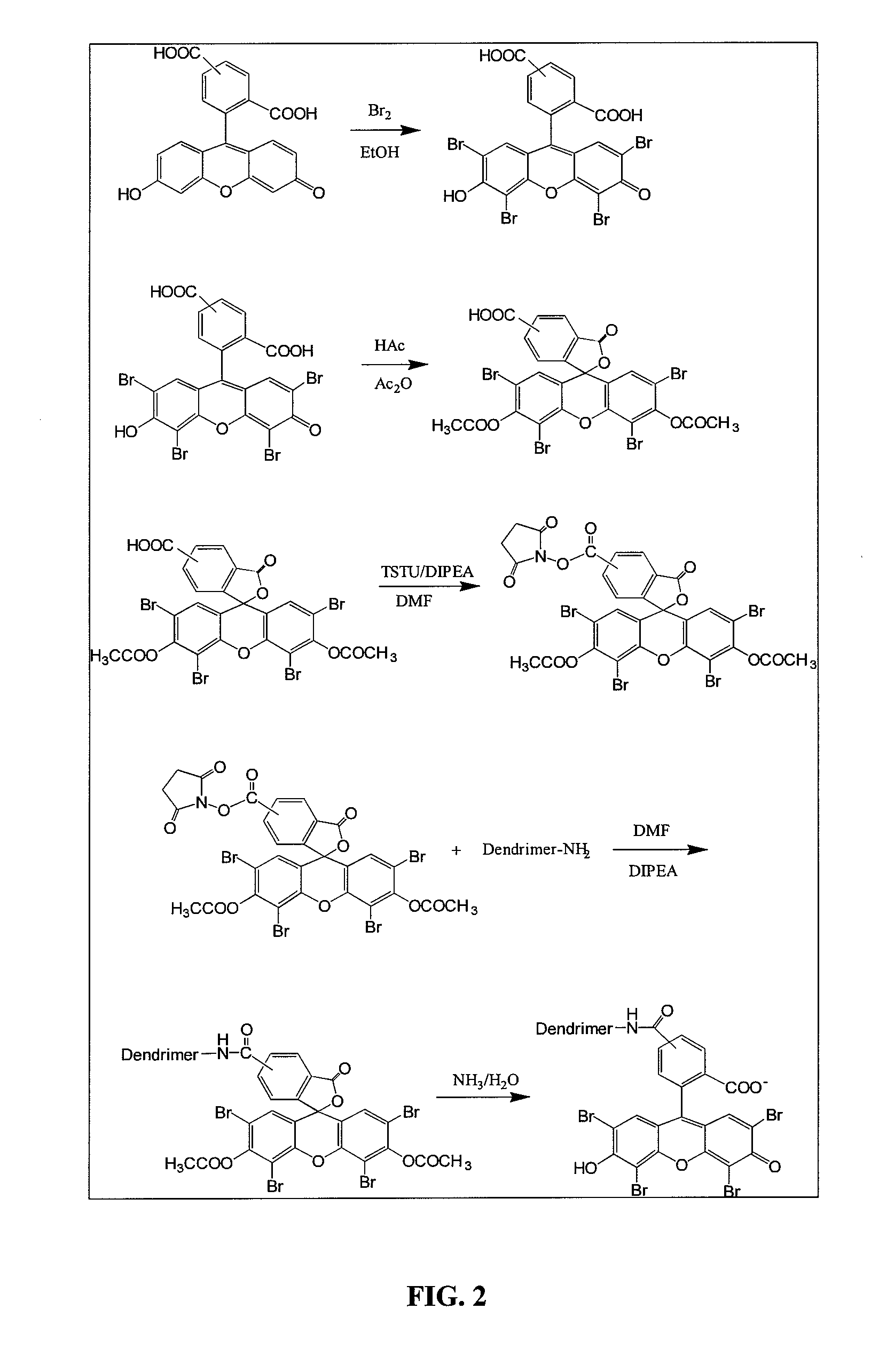
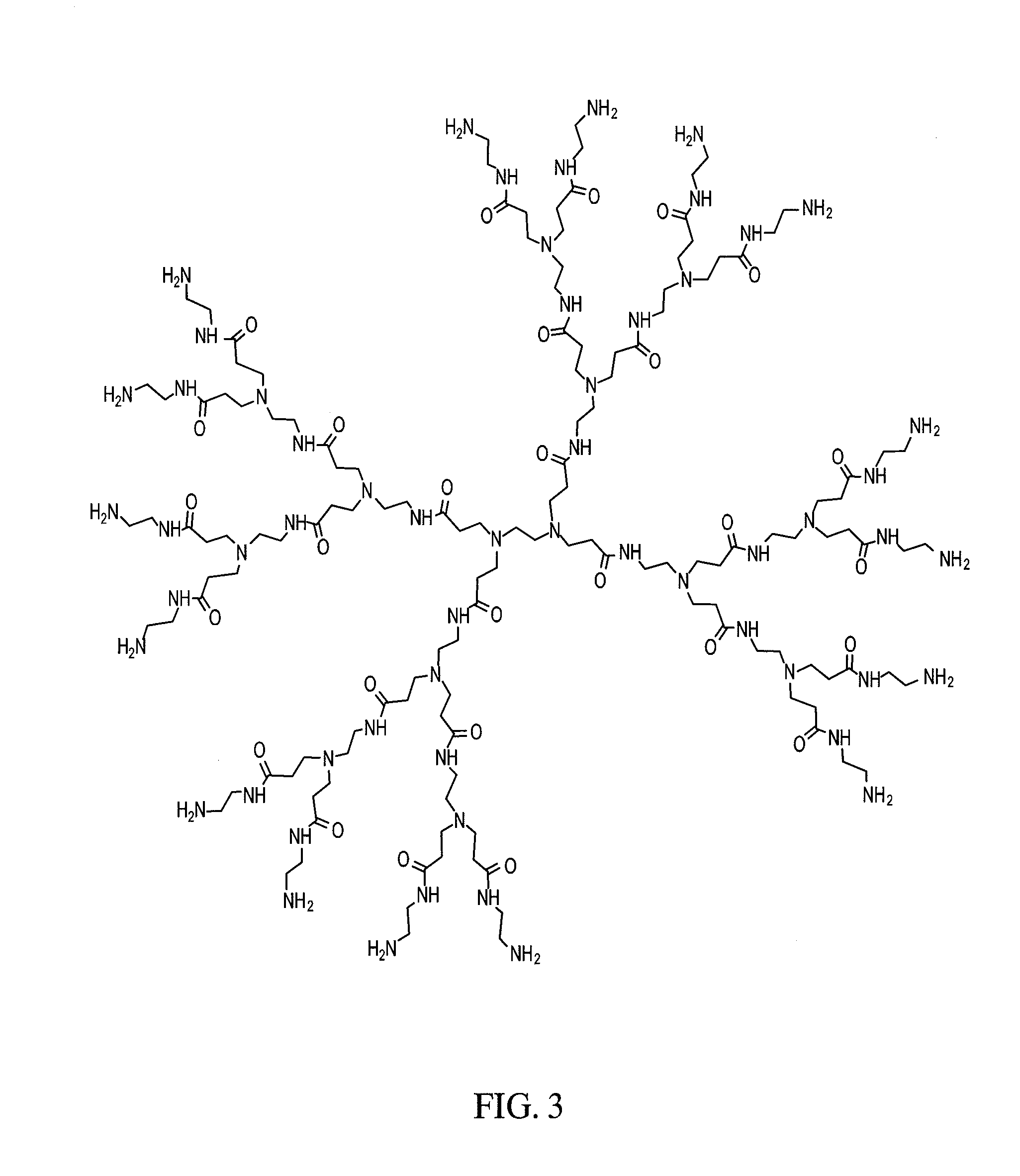
In diabetes, glucose saturates the
blood stream, but it cannot be transported into the cells where it is needed and utilized.
As a result, the cells of the body are starved of needed energy, which leads to the wasted appearance of many patients with poorly controlled insulin-dependent diabetes.
Prior to the discovery of insulin and its use as a treatment for diabetes, the only available treatment was starvation followed predictably by death.
Death still occurs today with insulin treatment from over dosage of insulin, which results in extreme
hypoglycemia and
coma followed by death unless reversed by someone who can quickly get glucose into the patient.
Also, death still occurs from major under dosage of insulin, which leads to hyperglycemia and ketoacidosis that can result in
coma and death if not properly and urgently treated.
While diabetes is not commonly a fatal
disease thanks to the treatments available to diabetics today, none of the standard treatments can replace the body's minute-to-minute production of insulin and precise control of glucose
metabolism.
Therefore, the average blood glucose levels in diabetics generally remain too high.
Since the incidence of diabetes is rising, the costs of diabetes care will occupy an ever-increasing fraction of total healthcare expenditures unless steps are taken promptly to meet the challenge.
The medical, emotional and financial
toll of diabetes is enormous, and increase as the numbers of those suffering from diabetes grows.
When the number of beta cells drops to a
critical level (10% of normal), blood glucose levels no longer can be controlled and progression to total insulin production failure is almost inevitable.
The multiple daily injections of insulin do not adequately mimic the body's minute-to-minute production of insulin and precise control of glucose
metabolism.
Eventually, however, the beta cells are gradually exhausted because they have to produce large amounts of excess insulin due to the
elevated blood glucose levels.
Ultimately, the overworked beta cells die and
insulin secretion fails, bringing with it a concomitant rise in blood glucose to sufficient levels that it can only be controlled by exogenous insulin injections.
However, high levels of glucose are toxic to beta cells, causing a progressive decline of function and
cell death.
This form of diabetes is due to one of several genetic errors in insulin-producing cells that
restrict their ability to process the glucose that enters via special glucose receptors.
Beta cells in patients with MODY cannot produce insulin correctly in response to glucose, which results in hyperglycemia.
The currently available medical treatments for insulin-dependent diabetes are limited to insulin administration and
pancreas transplantation with either whole pancreata or pancreatic segments.
Despite rigorous attention to maintaining a
healthy diet, exercise
regimen, and always injecting the proper amount of insulin, many other factors can adversely affect a person's blood-
sugar including stress, hormonal changes, periods of growth, illness, infection and fatigue.
Insulin-dependent diabetes is a life threatening disease, which requires never-ending vigilance.
Although
pancreas transplants are very successful in helping people with insulin-dependent diabetes improve their
blood sugar control without the need for insulin injections and reduce their long-term complications, there are a number of drawbacks to whole pancreas transplants.
Some risks in taking these immuno-suppressive drugs are the increased incidence of infections and tumors that can be life threatening in their own right.
The risks inherent in the operative procedure, the requirement for life-long
immunosuppression of the patient to prevent rejection of the transplant, and the morbidity and
mortality rate associated with this
invasive procedure, illustrate the serious disadvantages associated with whole pancreas transplantation for the treatment of diabetes.
Due to its relatively
large size, there are few sites in the body able to accommodate it for the treatment of a disease like diabetes.
Since it grows into the body and the contained cells are not expected to survive for more than a few years, multiple
cell removals and reloading of new cells is required for the long-term application of this device.
It has proven quite difficult to flush and reload this type of device while at the same time maintaining the critical
cell compartment distance for
oxygen diffusion.
For the diabetes product, it has been quite difficult to place this device into the intraperitoneal cavity of large animals, while maintaining its integrity.
This has been due to the difficulty in securing it in the
abdomen so that the intestines cannot cause it to move or
wrinkle, which may damage or break the device.
This type has shown
efficacy in
large animal diabetic trials, but has been plagued by problems in the access to the vascular site.
Both
thrombosis and hemorrhage have complicated the development of this approach with it currently being abandoned as a clinically relevant product.
Due to low packing densities, the required
cell mass for encapsulation causes the length of this type of hollow to approach many meters.
Therefore, this approach was abandoned for treating diabetes since it was not clinically relevant.
In addition, sealing the open ends of the
fiber is not trivial and strength has been a problem depending upon the extravascular site.
However, nearly 25 years have passed since these first reports without the ability to demonstrate
clinical efficacy.
One of the problems associated with microcapsules is their relatively
large size in combination with low packing densities of cells, especially for the treatment of diabetes.
The permselectivity of pure alginate capsules has been difficult to control with the vast majority being wide open in terms of molecular weight cutoff.
However,
polylysine and most other similar molecules invite an inflammatory reaction requiring an additional third
coating of alginate to reduce the host's response to the
capsule.
In addition, it has been difficult to produce very pure alginates that are not reactive within the host after implantation.
Trying to reduce the size of the alginate microcapsules causes two major problems.
First, the production of very large quantities of empty capsules without any cells.
Second, the formation of smaller capsules results in poorly coated cells.
There is no force to keep the contained cells within the center of the microcapsule, which causes the risk of incomplete coatings to go up exponentially with the decrease in the size of the capsules.
The stringent requirements of encapsulating polymers for
biocompatibility,
chemical stability, immunoprotection and resistance to cellular overgrowth
restrict the applicability of prior art methods of encapsulating cells and other biological materials.
These biomaterials are considered biocompatible if they produce a minimal or no adverse response in the body.
Thus, they can interact with implanted material through any of these various regions, resulting in cellular proliferation at the
implant surface.
Due to the inability of those of skill in the art to provide one or more important properties of successful
cell encapsulation, none of the encapsulation technologies developed in the past have resulted in a clinical product.
This process produces many cytokines that will certainly diffuse through the
capsule and most likely destroy the encapsulated cells.
Most devices tested to date have failed in part by their lack of
biocompatibility in the host.
Small pores capable of keeping out immune cytokines also cause the death of the encapsulated cells from a lack of
diffusion of nutritional elements and waste products.
Relevant Size—Many devices are of such a
large size that the number of practical implantation sites in the host is limited.
This would further lower the
oxygen concentration to a point where the cells cannot adequately function or survive.
Many device designs have not considered the fact that encapsulated cells have a limited lifetime in the host and require regular replacement.
Another risk has been elevated portal
venous pressures from large volumes of injected
islet tissues that are not sufficiently purified.
This also leads to portal venous
thrombosis as a complication of this procedure.
Attempts at
subcutaneous implantation of encapsulated islets have been unable to produce sustainable results in the treatment of diabetes, probably due to some or all of the scientific challenges described above.
One of skill in the art would be unable to determine the desired volume of encapsulated cells needed to administer to a subject.
 Login to View More
Login to View More