Patents
Literature
64 results about "Anterior capsule" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
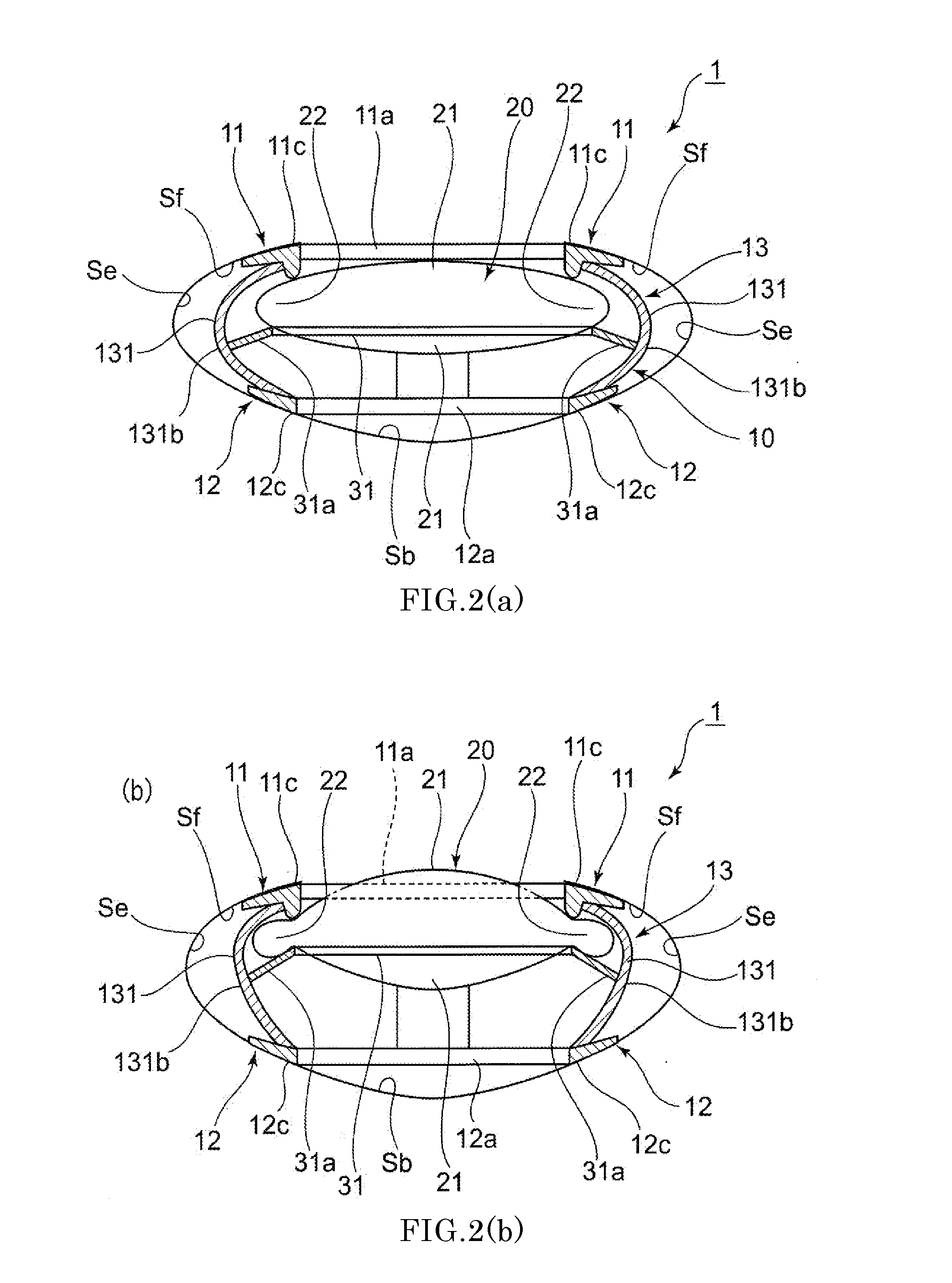
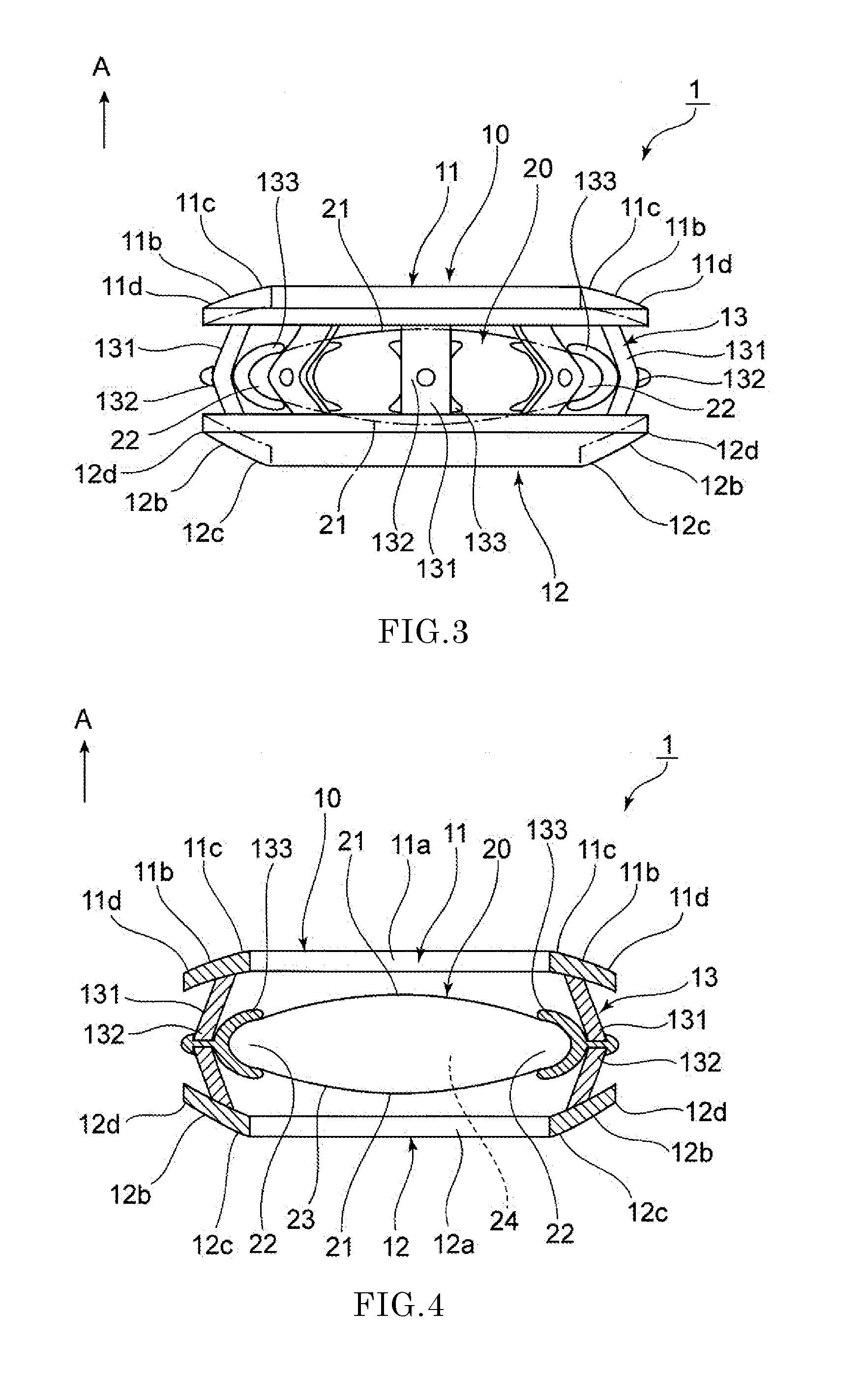
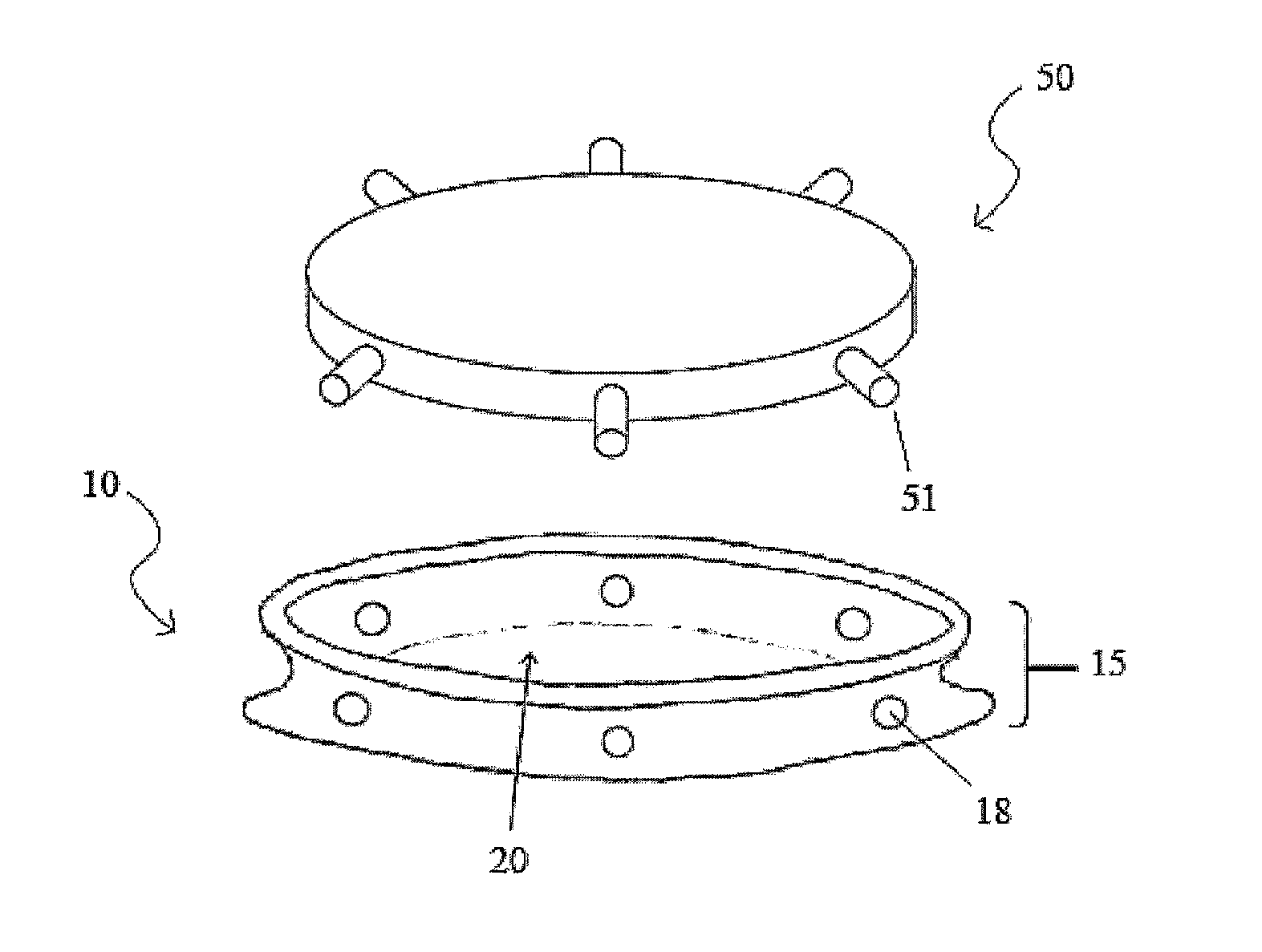
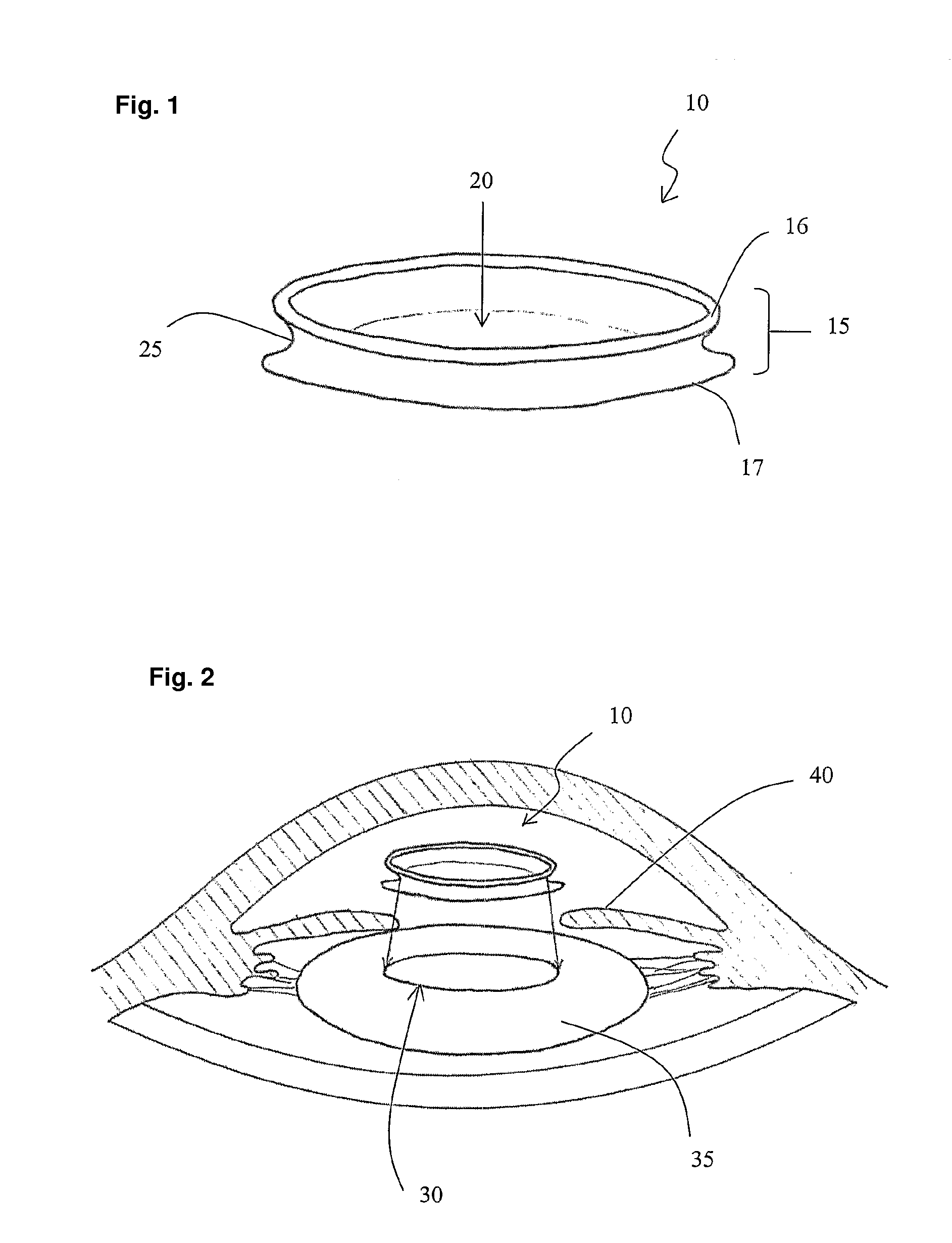
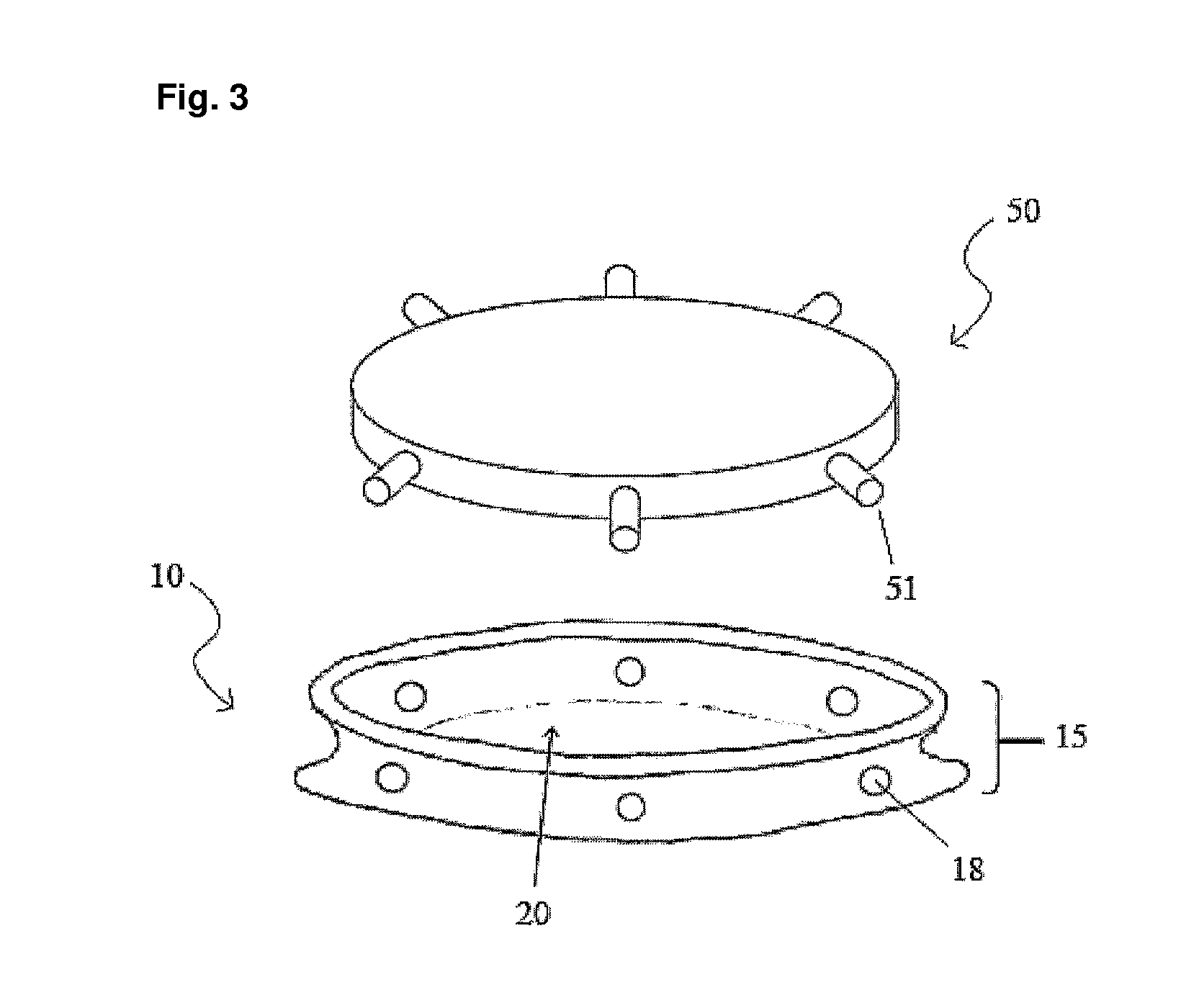
Intraocular lens and method for preventing secondary opacification
InactiveUS6027531APrevent proliferationImprove stabilityDiagnosticsSurgeryCataract extractionPosterior capsulorhexis
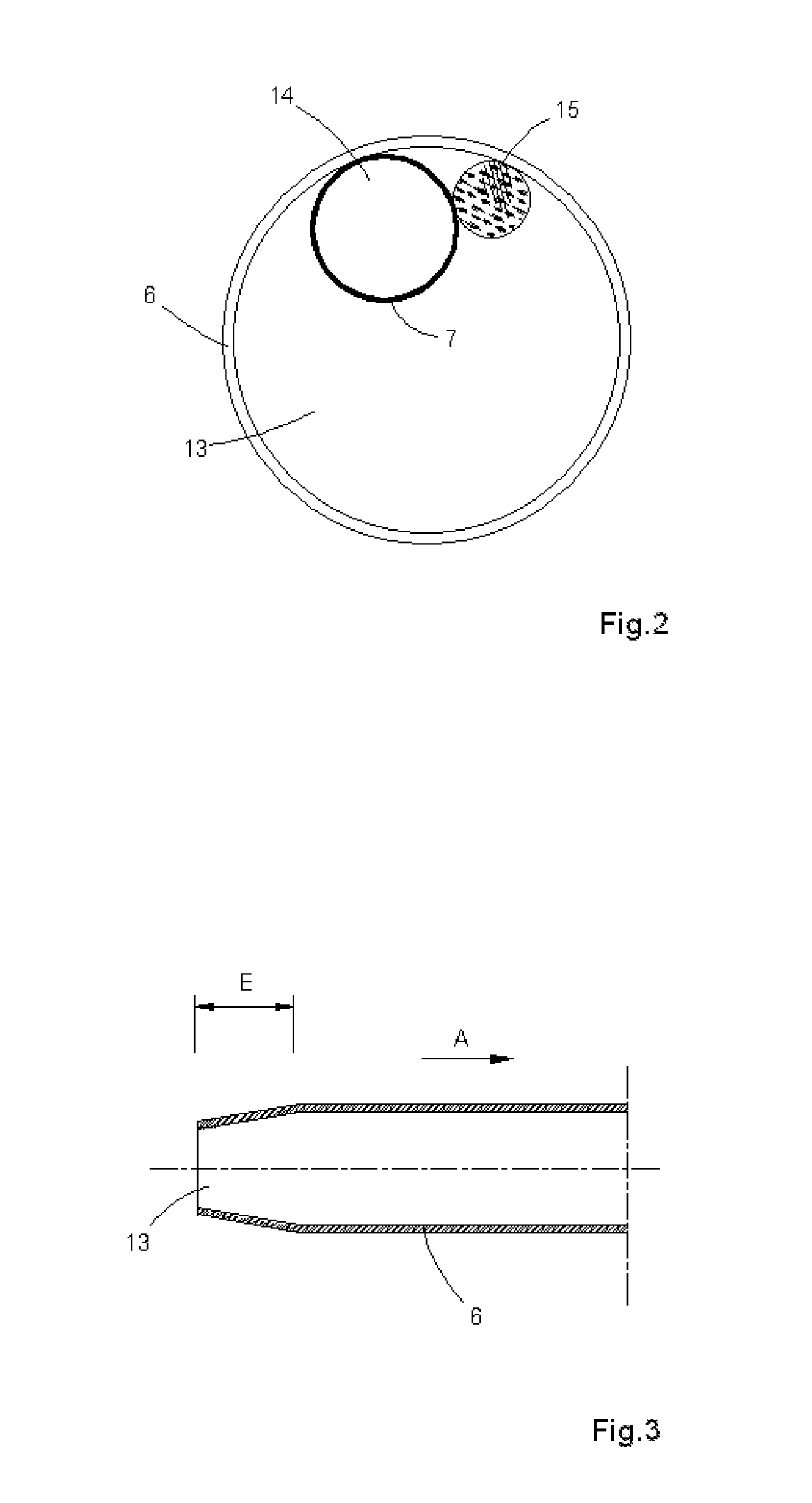
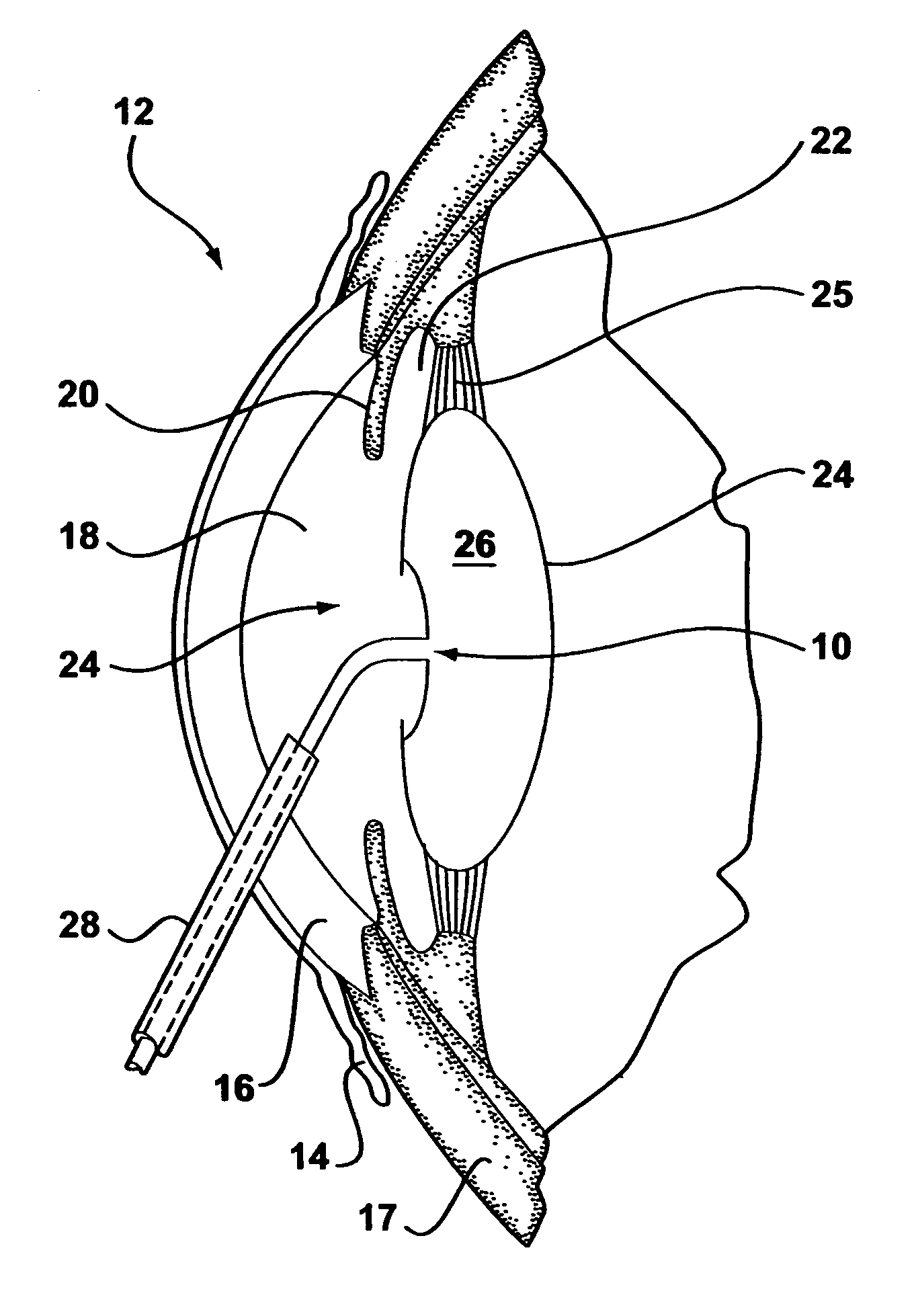
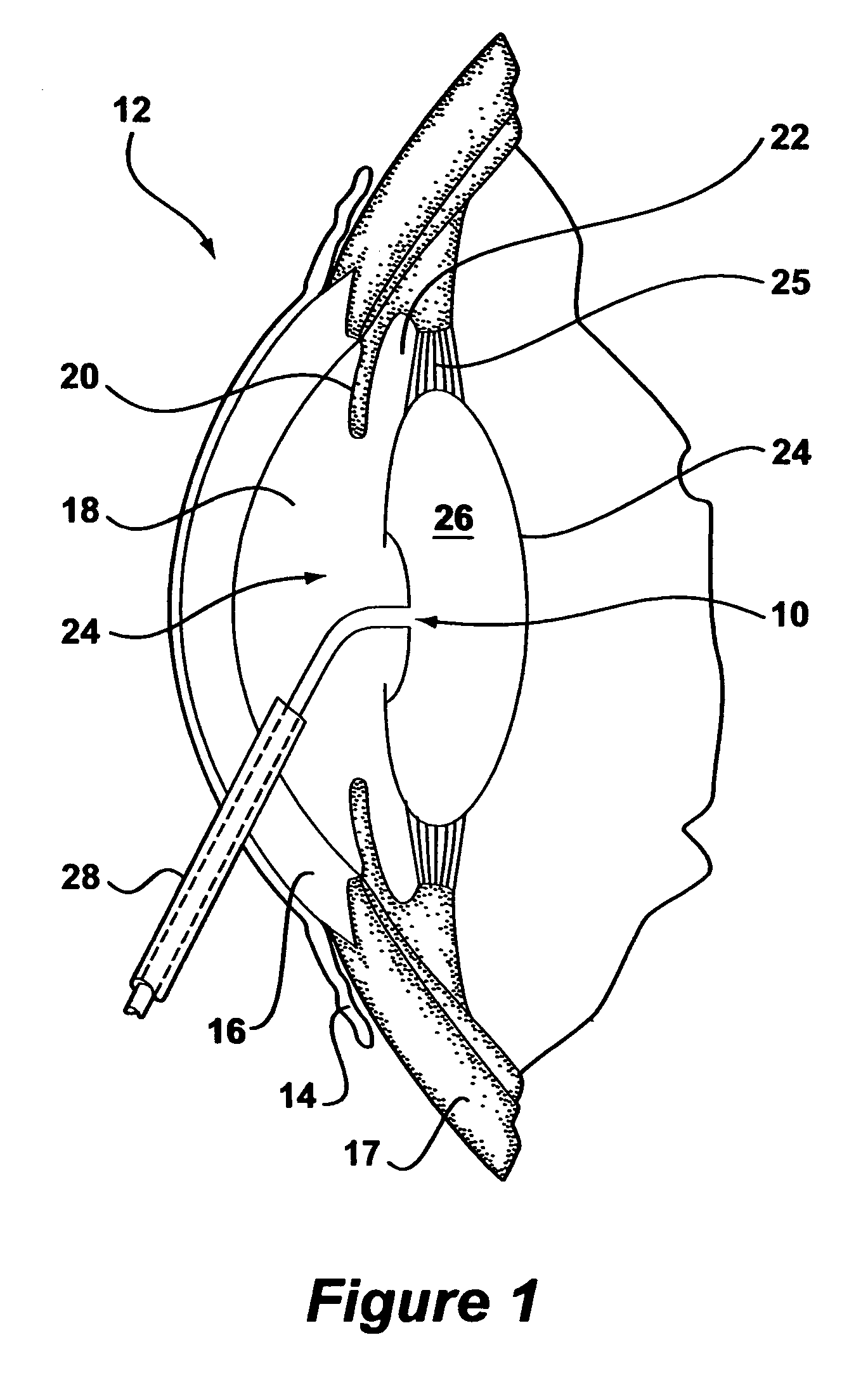
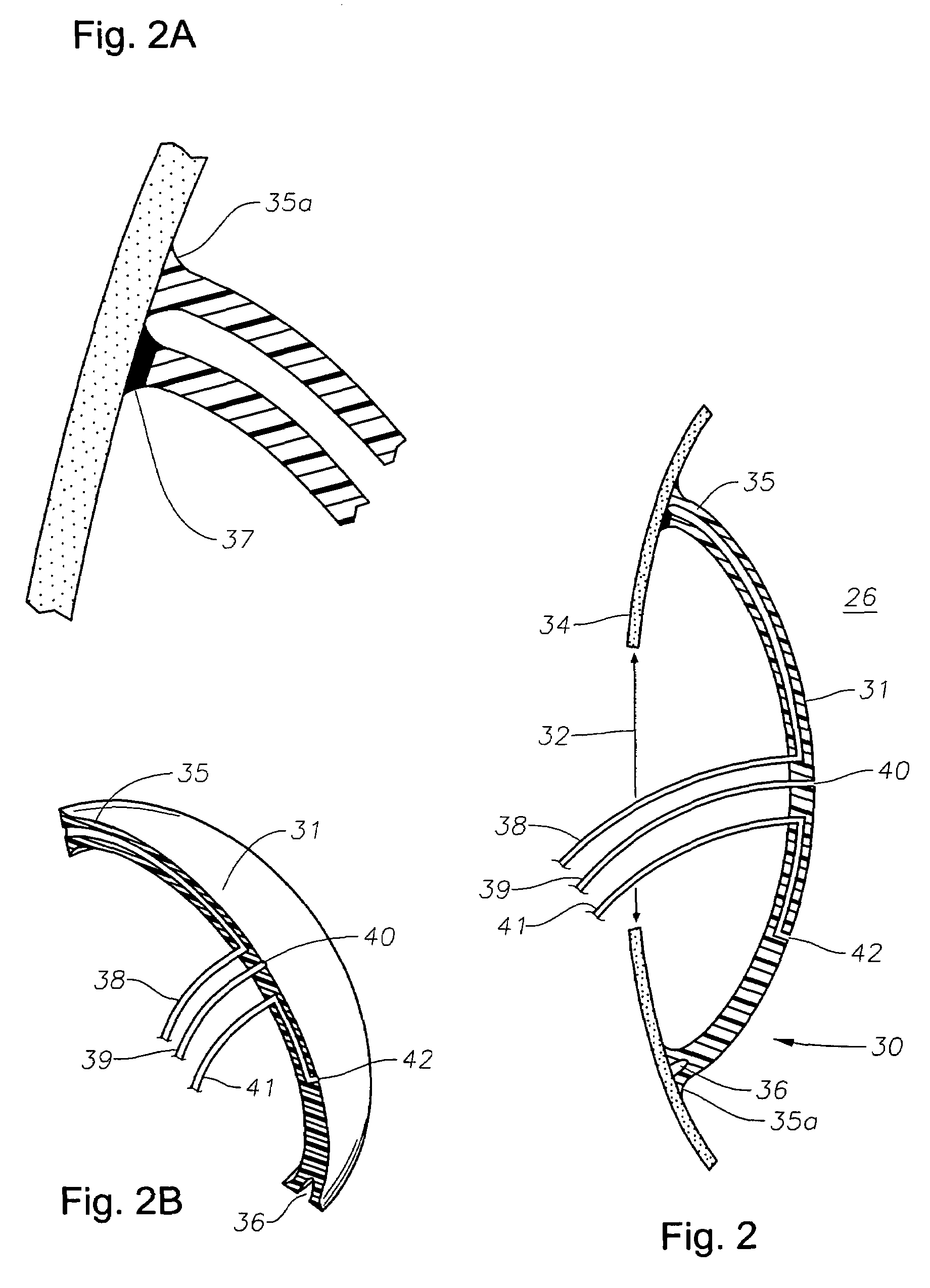
An intraocular lens for use in extracapsular cataract extraction has a haptic pat that surrounds the optical pat of the lens and further contains a groove of such shape to accommodate the anterior and posterior capsules of the lens bag after anterior capsulorhexis, extracapsular cataract extraction and posterior capsulorhexis. The lens is preferably inserted in a calibrated, circular and continuous combined anterior and posterior capsulorhexis, slightly smaller than the inner circumference of the groove as to induce a stretching of the rims of the capsular openings. This new approach is believed to prevent the appearance of secondary opacification of the capsules, allows a very stable fixation of the intraocular lens and ensures a tight separation between the anterior and posterior segment of the eye. This new principle of insertion is called the bag-in-the-lens technique, in contrast with the classical lens in-the-bag technique.
Owner:TASSIGNON MARIE JOSE B R
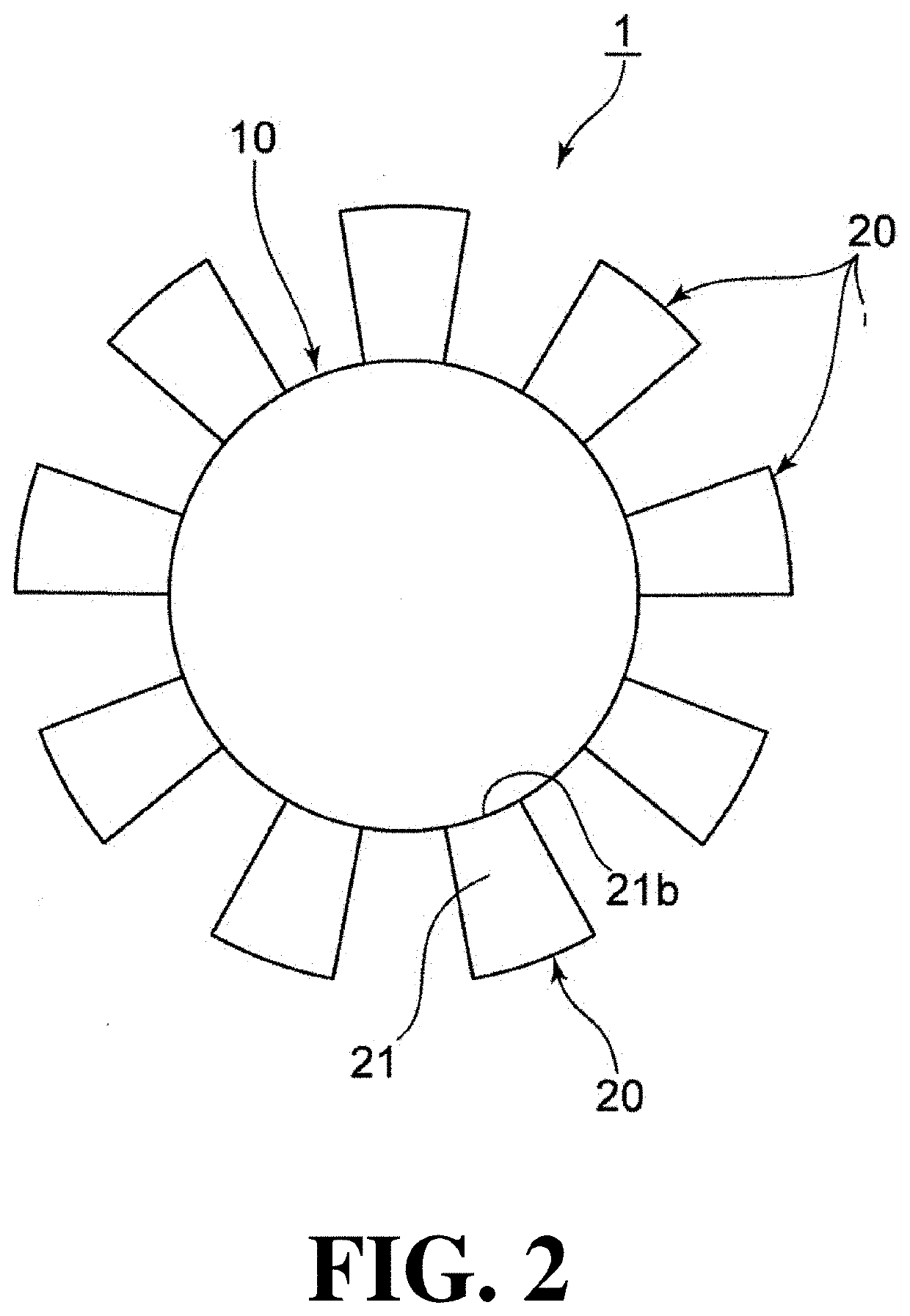
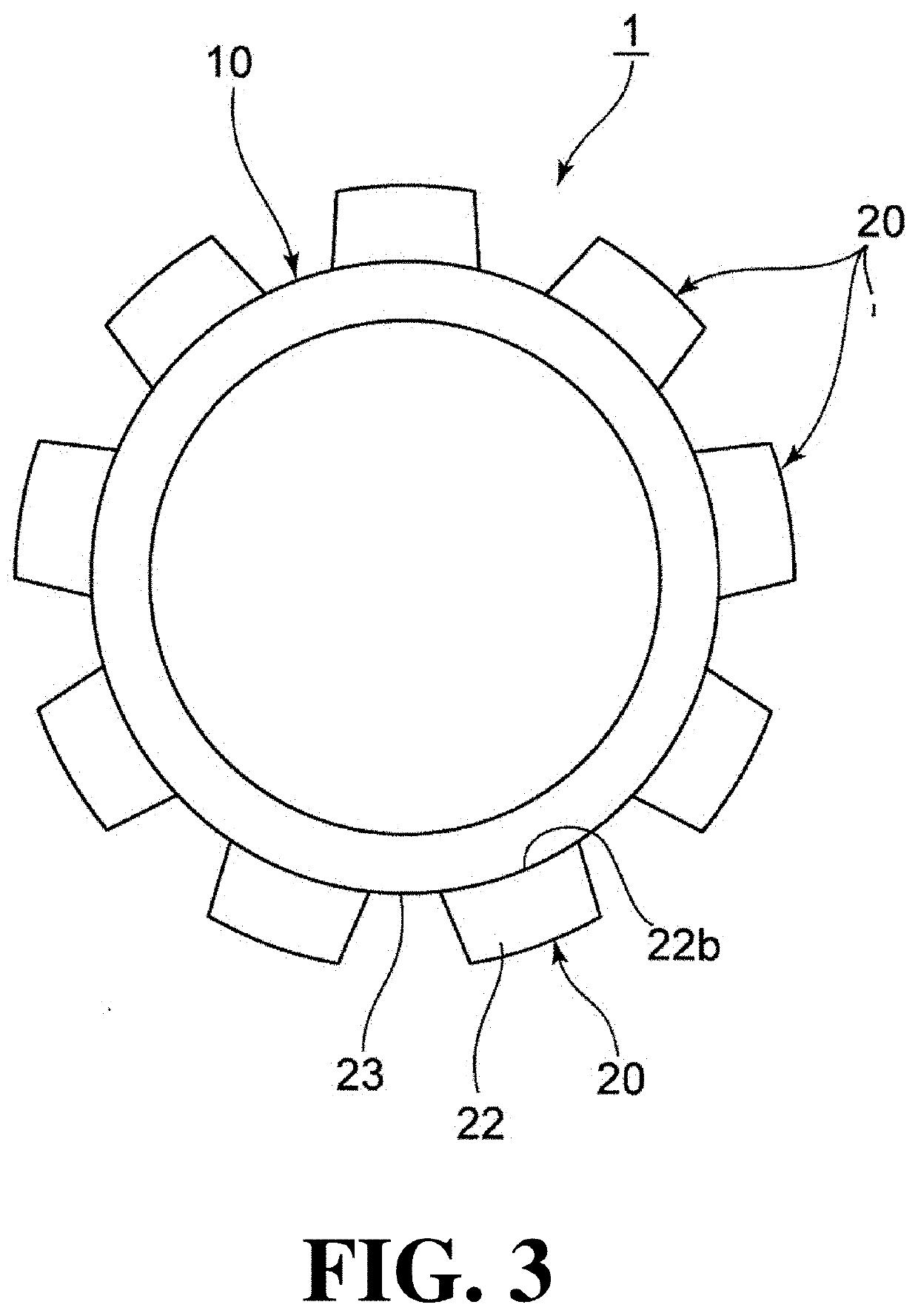
Capsularhexis device
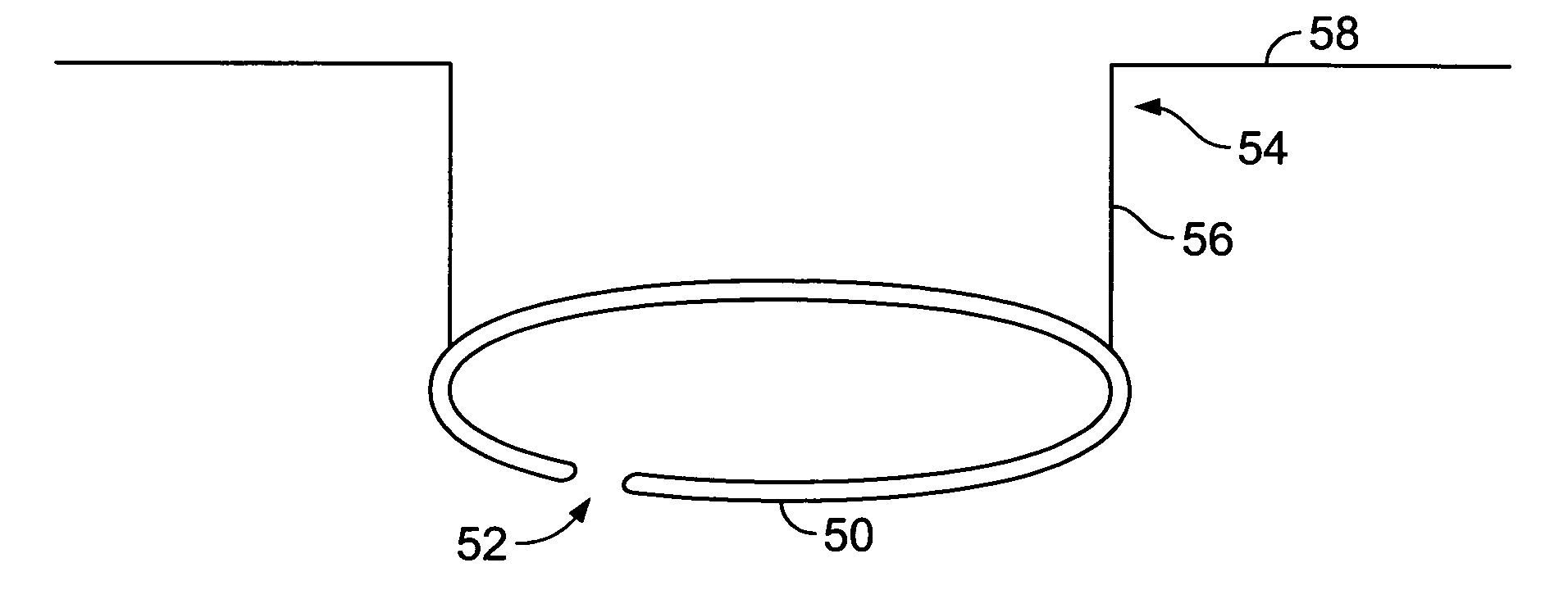
A flexible ring having a diameter suitable for an anterior chamber rhexis. Imbedded in the ring is a heating element or filament. Alternatively, the ring may contain a pair of adjacent, circular electrodes. The electrodes or heating filament are placed against the anterior capsule and electrical current is applied, causes localized heating around the electrodes or filament. The flexible nature of the ring and electrodes allow the ring to be folded so as to pass through a relatively small (<3 mm) incision, yet retain its full ring-shape once within the anterior chamber of the eye.
Owner:ALCON INC
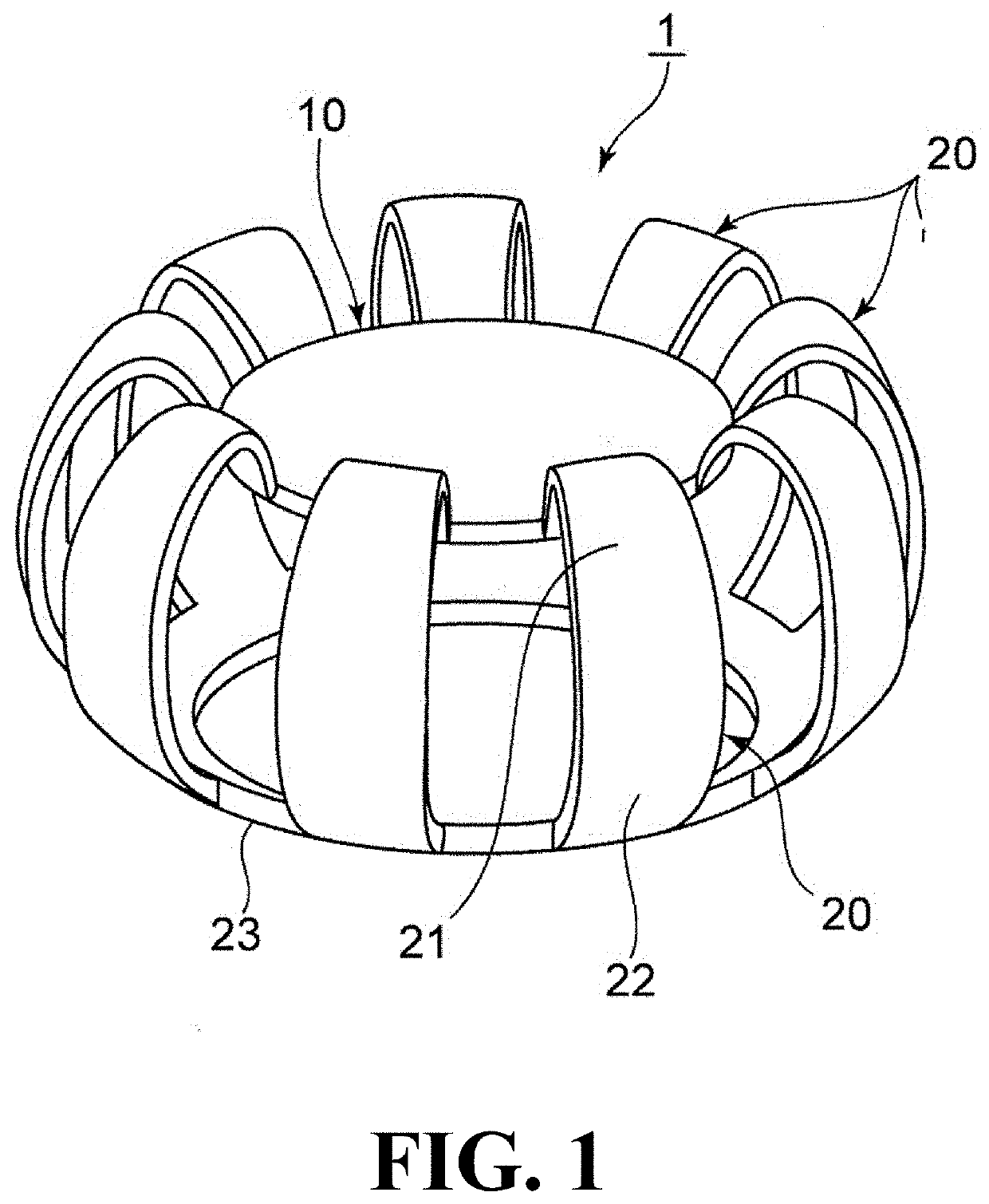
Tensioning rings for anterior capsules and accommodative intraocular lenses for use therewith
InactiveUS20160000558A1Reducing expander ring pressureReduce tensionIntraocular lensContinuous curvilinear capsulorrhexisCentripetal force
A tensioning device for attaching to the anterior capsule of an eye, and accommodative intraocular lens systems employing the device. The tensioning device includes a biocompatible, elastically reconfigurable ring for restoring at least a portion of the anterior capsule centripetal forces lost by capsulorhexis. The tensioning device also includes a plurality of penetrators configured for attaching the ring to the anterior capsule. The plurality of penetrators is biocompatible with the eye and partially embedded in a part of the ring configured for facing the anterior capsule.
Owner:HONIGSBAUM RICHARD F
System and method of performing femtosecond laser accomodative capsulotomy
Disclosed is a system and method for making a first incision in an anterior capsule of a capsular bag, the first incision being less than or equal to approximately 3.5 mm in diameter and making a second incision in the anterior capsule, the second incision being less than or equal to approximately 3.0 mm in diameter. The first incision and the second incision are positioned off-center from a center portion of the anterior capsule. The method includes performing lens fragmentation of a lens in the capsular bag to yield lens material, inserting a first instrument into the first incision, inserting a second first instrument into the second incision and removing the lens material via one of the first instrument and the second instrument and through one of the first incision and the second incision. The tensile structure of the anterior portion of the capsular bag is maintained such that accommodation exists within the eye after insertion of the intraocular lens.
Owner:IANCHULEV PRAVOSLAVA
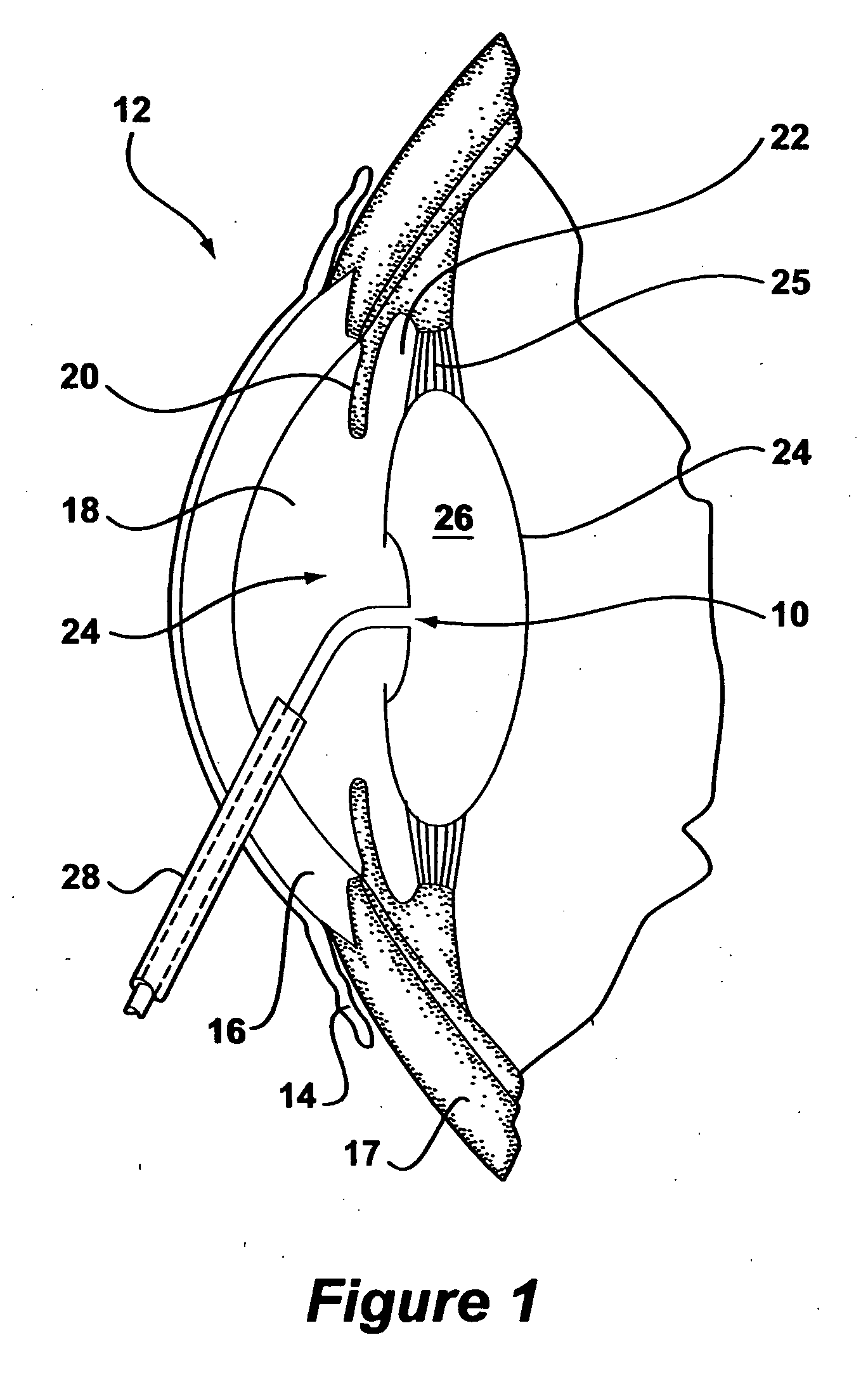
Maintaining preoperative position of the posterior lens capsule after cataract surgery
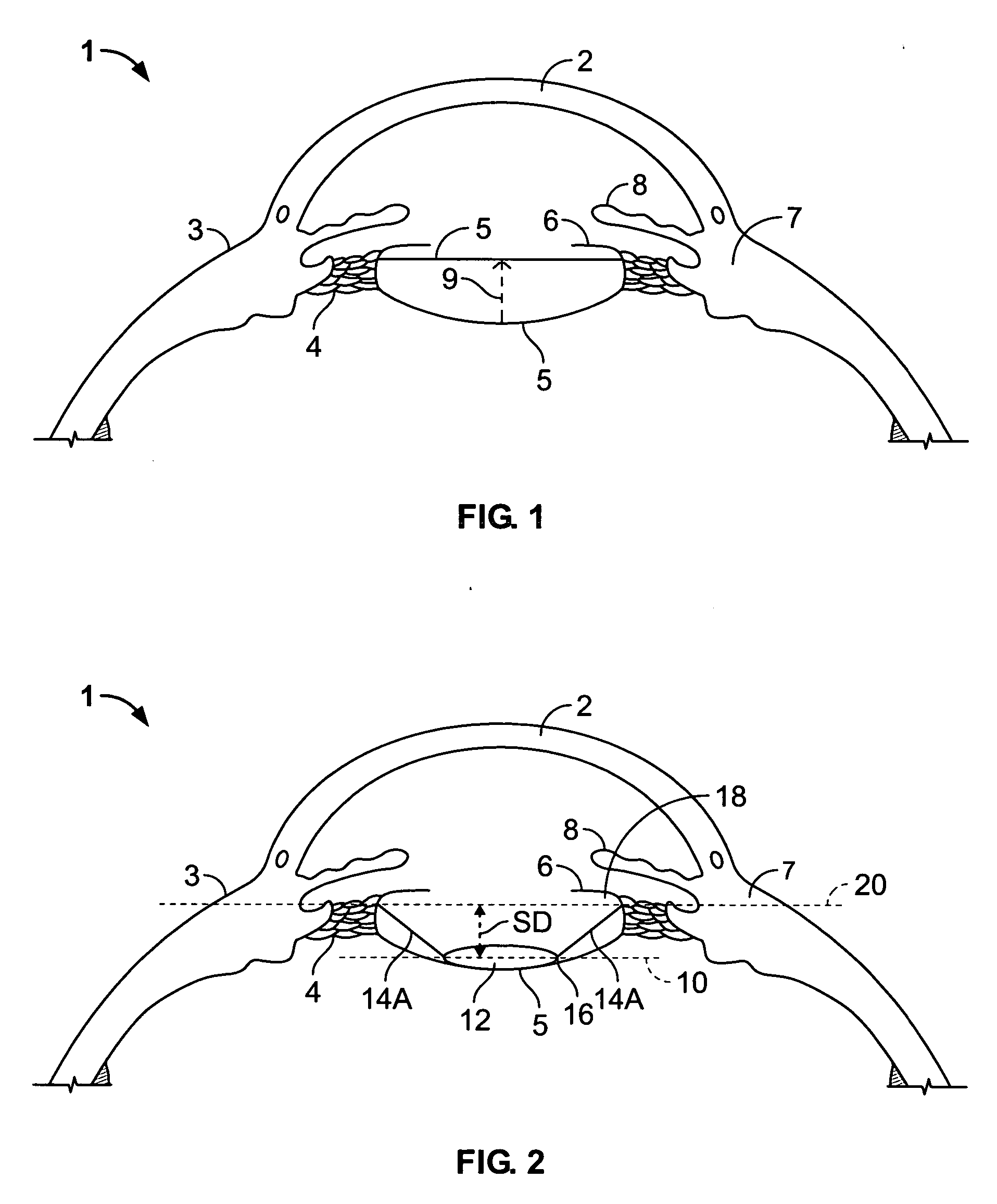
Intraocular lens implant that includes a lens optic and lens haptics configured to maintain a preoperative position of the posterior lens capsule after cataract removal and insertion of a lens implant. The lens haptics have proximal and distal portions, with the distal portions lying in a common plane and the lens optic extending in a lens optic plane. The distance between the planes may be at least substantially the same dimension as or larger than a shift distance that the posterior lens capsule would otherwise traverse between its normal anatomical location and its shifted anatomical location where it not constrained. The shifted anatomical location arises naturally after both removal of cataract lens material and removal of a portion of an anterior capsule.
Owner:NOVARTIS AG
Lenticular capsule-expanding device
InactiveUS20150289970A1High precisionAdjustment function can be effectivelyIntraocular lensCataract surgeryEngineering
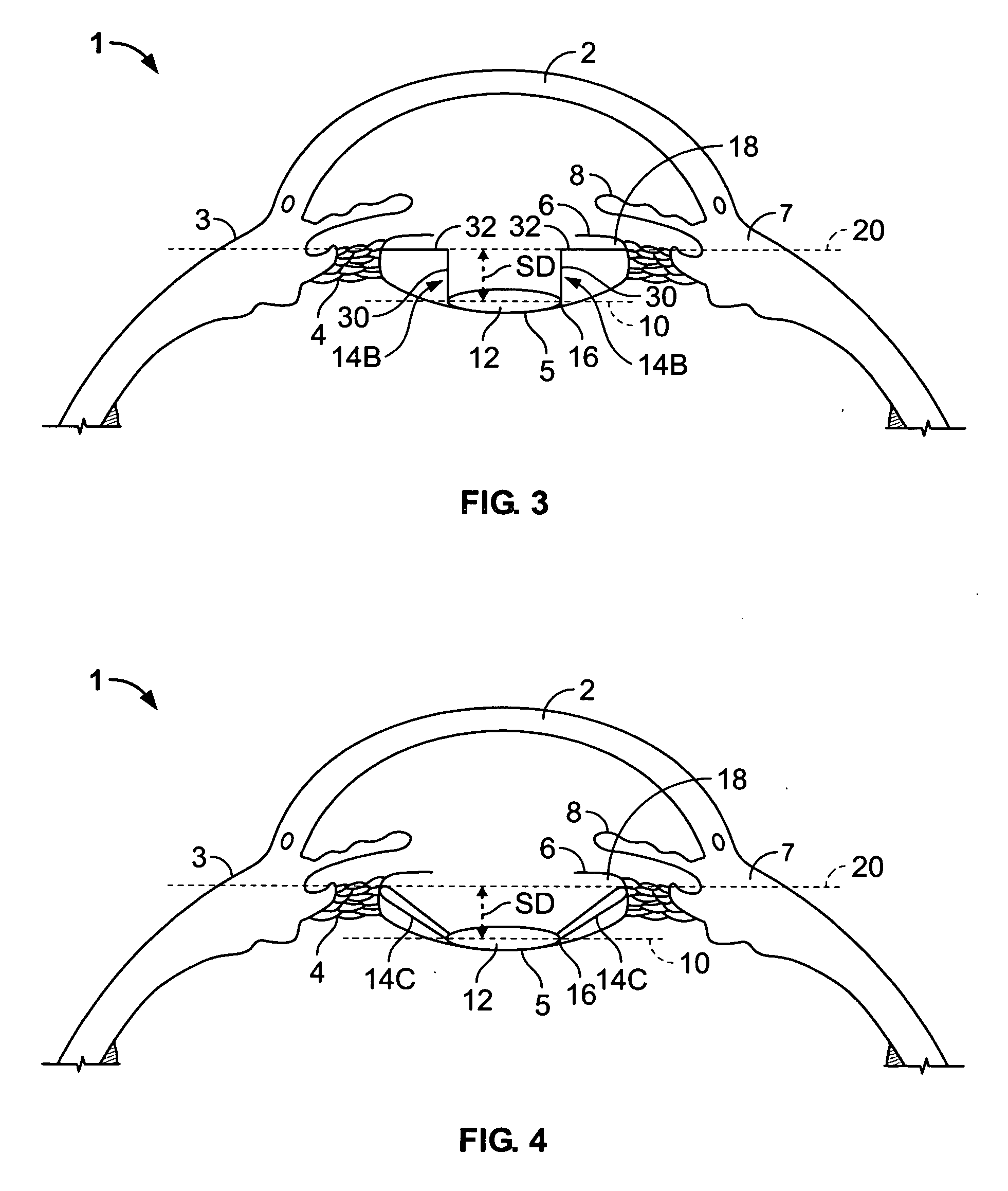
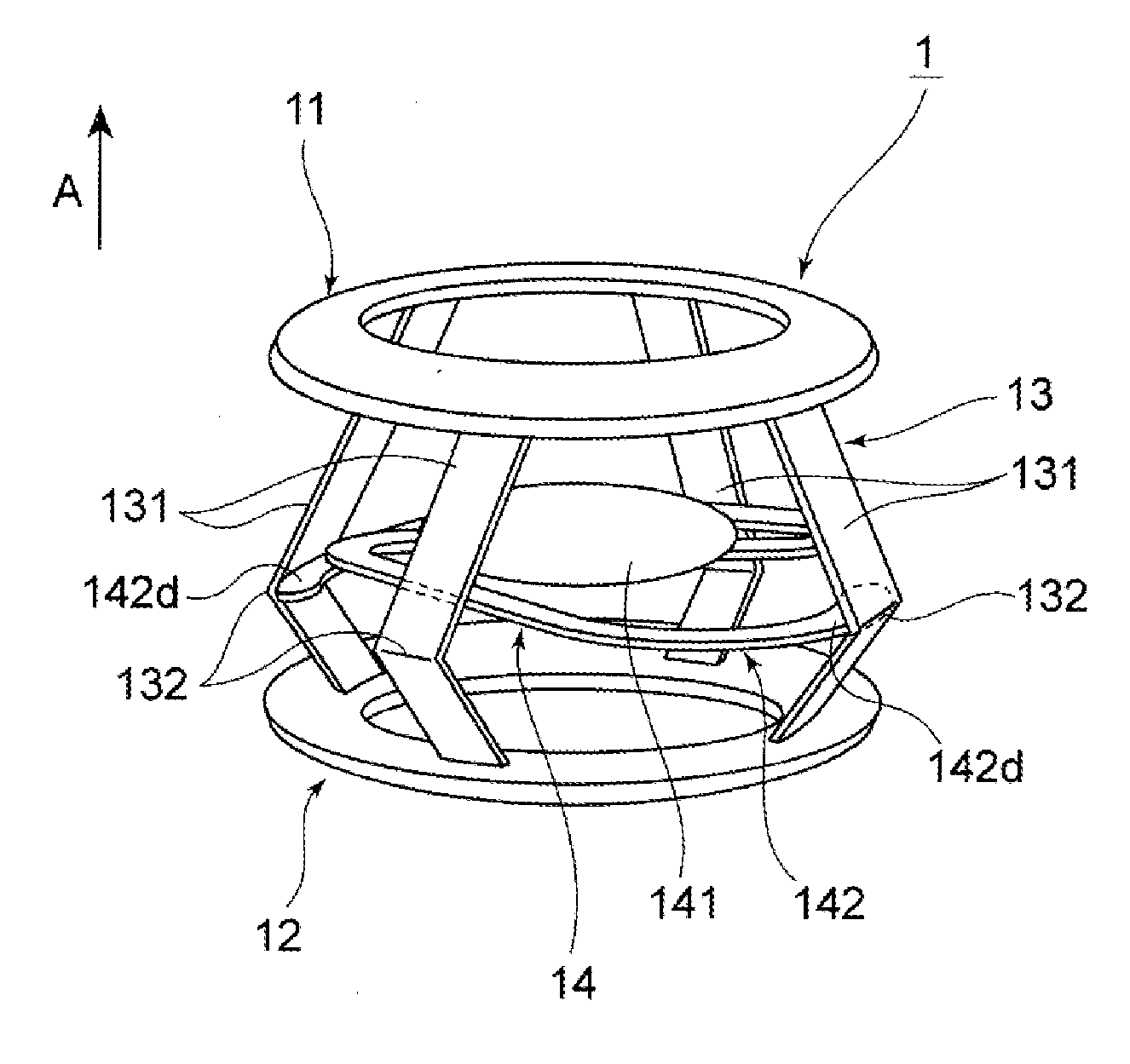
A lenticular capsule-expanding device is arranged in a lens capsule in a cataract surgery, etc. The device includes a front supporting section to be arranged in contact with an inner surface of an anterior capsule, the front supporting section, a rear supporting section to be arranged in contact with an inner surface of a posterior capsule, the rear supporting section being arranged behind the front supporting section, and a connecting section that connects the front and rear supporting sections in a manner as to have an urging force for separating the front and rear supporting sections in a separating direction. By the urging force of the connecting section, the front supporting section presses against the inner surface of the anterior capsule and the rear supporting section presses against the inner surface of the posterior capsule.
Owner:XLENS TECH INC
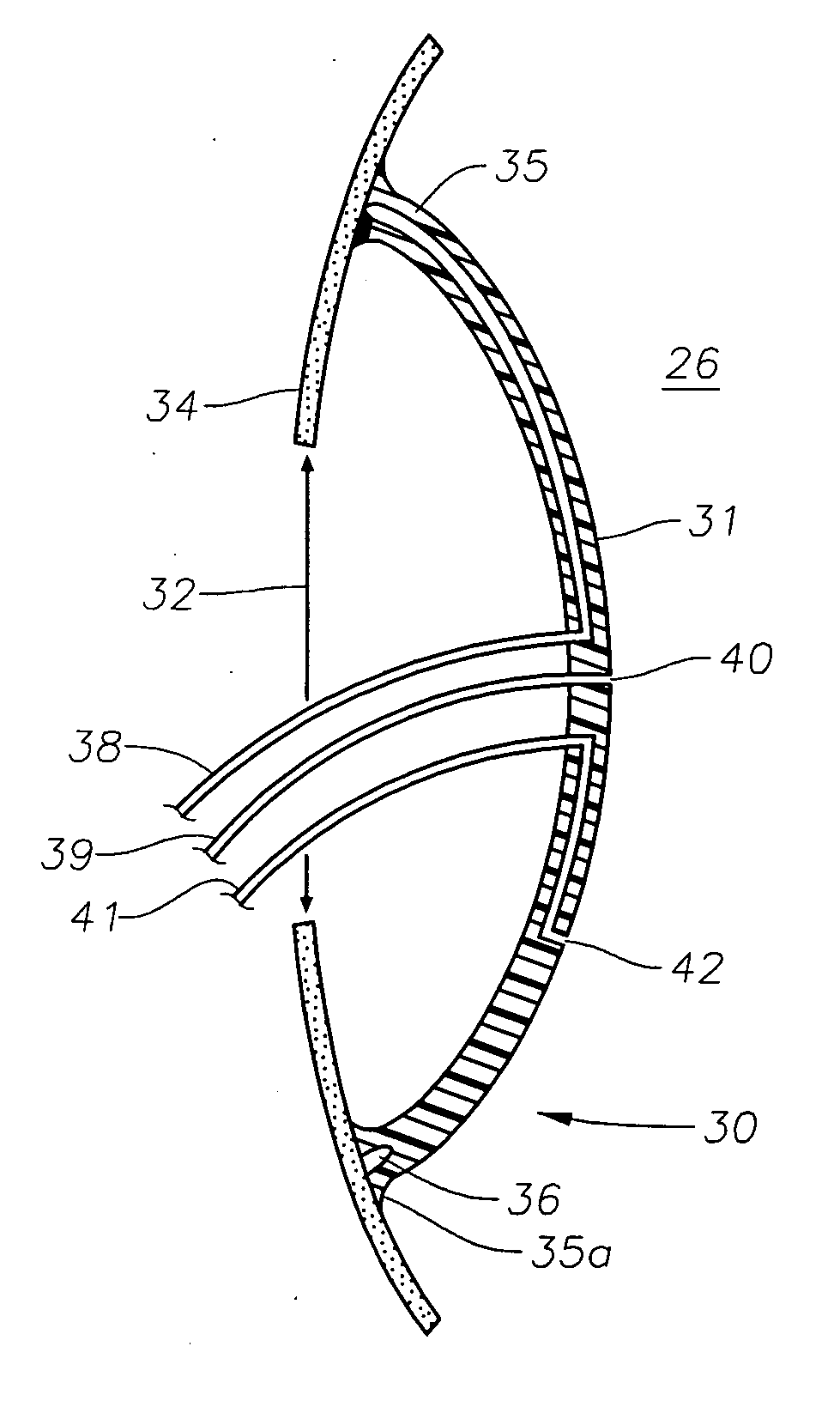
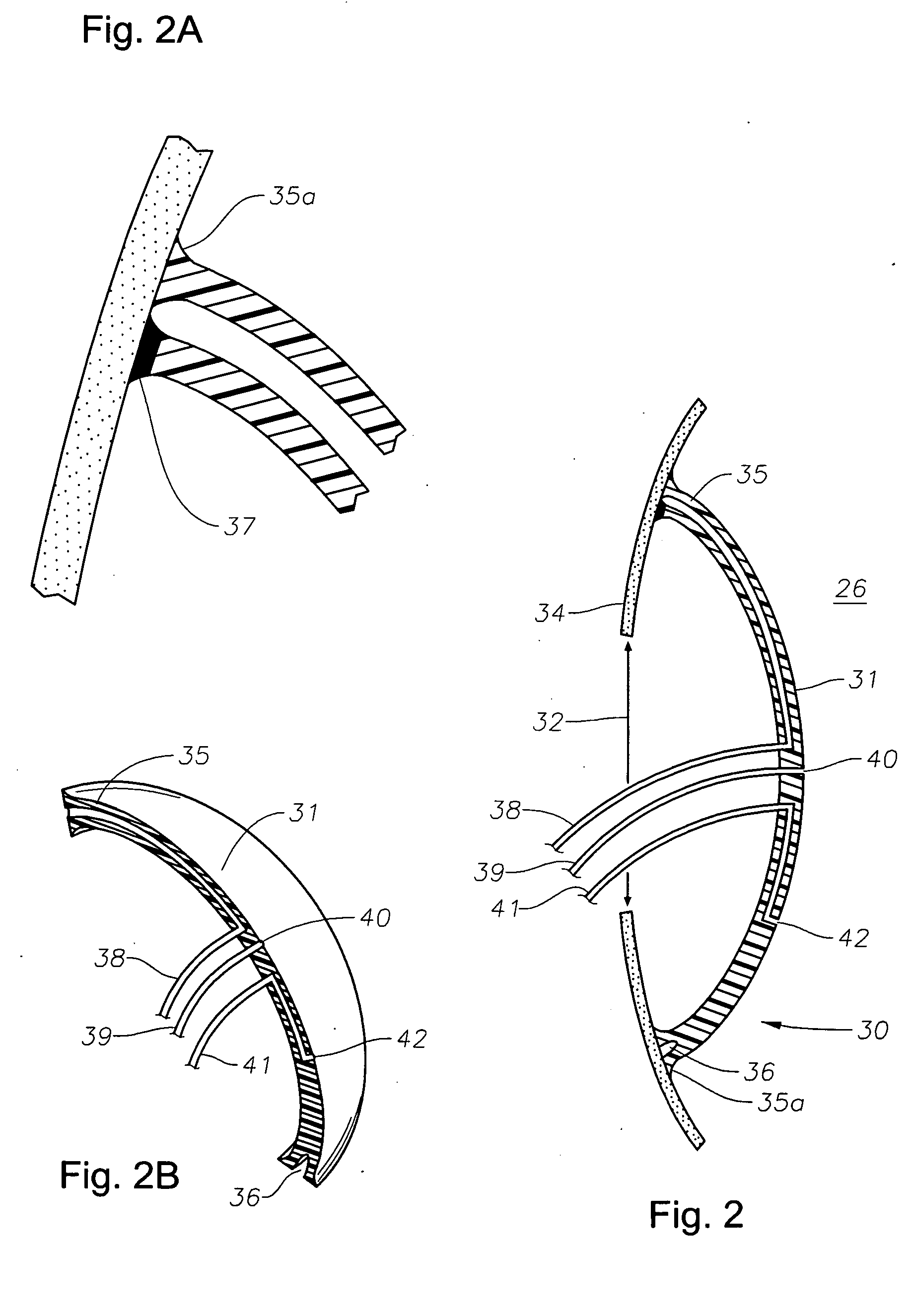
Accommodating intraocular lens
ActiveUS20160310263A1Reduce diameter of lensModerate strengthIntraocular lensEngineeringPhakic intraocular lens
An intraocular lens includes a lens capsule expanding device and an optical portion. A front supporting portion makes contact with an inner surface of an anterior capsule. A rear supporting portion makes contact with an inner surface of a posterior capsule while facing the front supporting portion. A connecting portion connects the front supporting portion and the rear supporting portion so as to have a biasing force to separate the front supporting portion and the rear supporting portion. Due to the biasing force, the front supporting portion presses the inner surface of the anterior capsule and the rear supporting portion presses the inner surface of the posterior capsule. The optical portion changes the curvature of a central portion according to movement of the connecting portion that moves the front supporting portion and the rear supporting portion closer to or away from each other with movement of the lens capsule.
Owner:MIRAI EYE INC
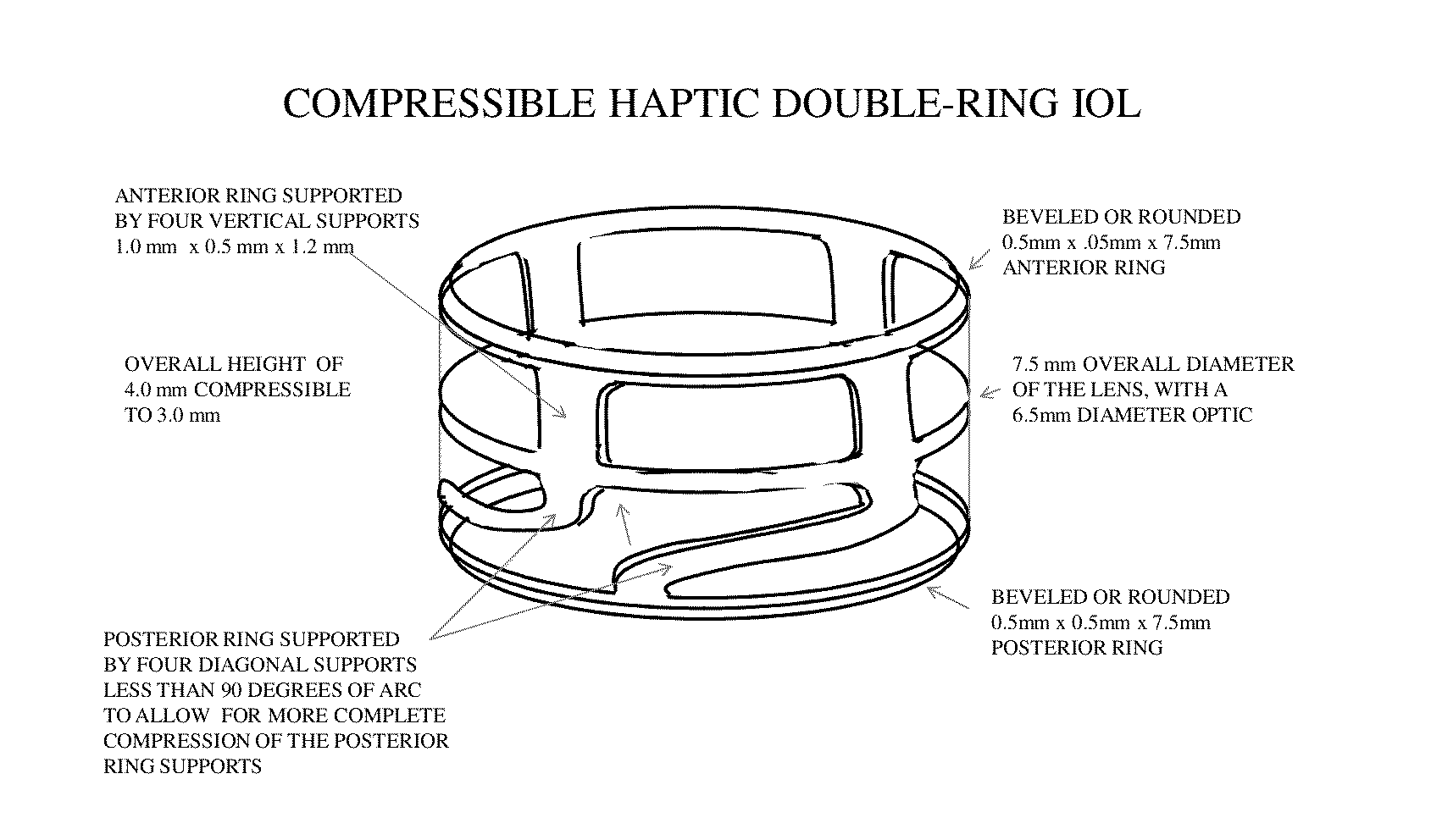
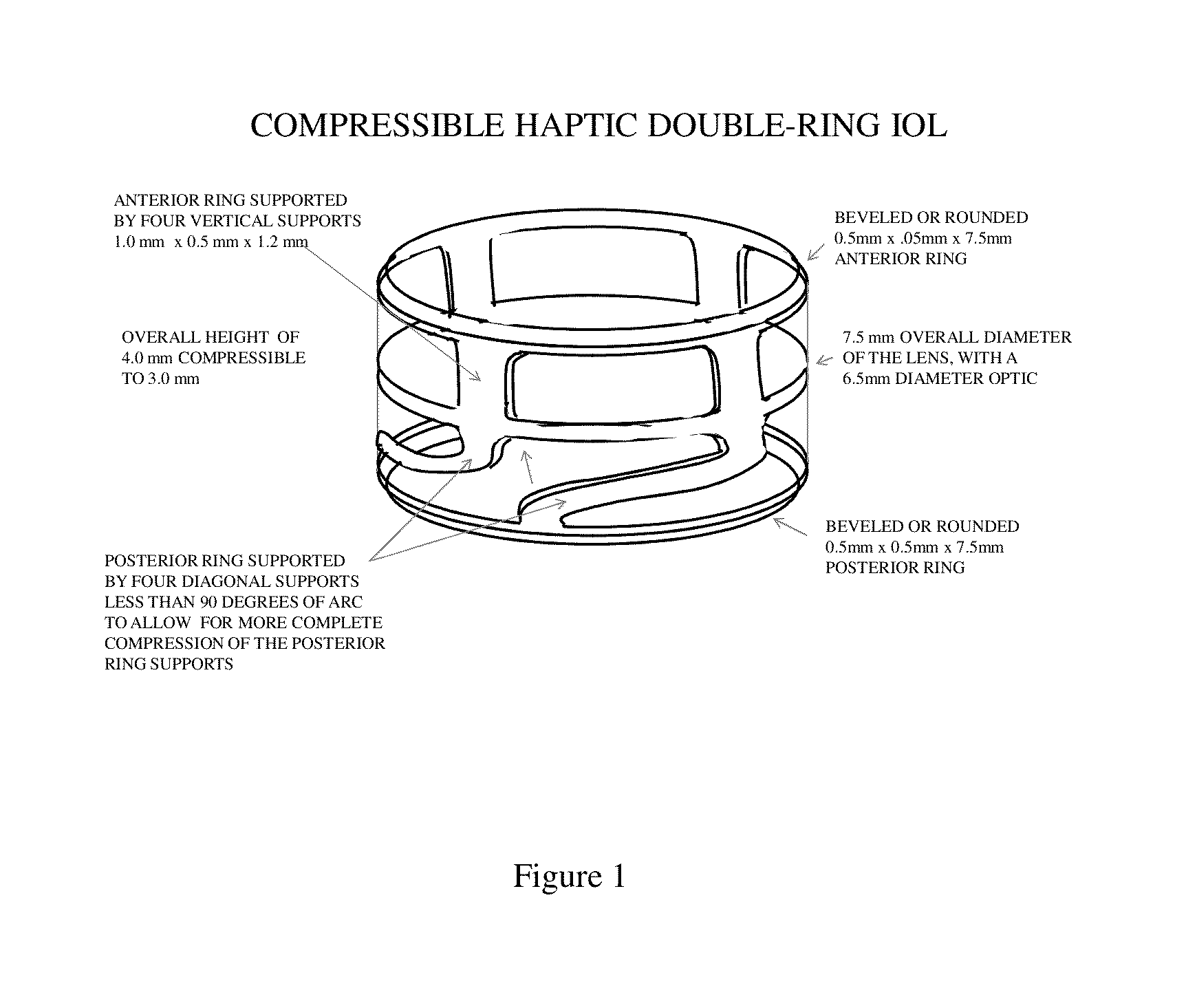
Intraocular lens rings
InactiveUS20130184816A1Suitable and consistent visual acuityLong lasting fitIntraocular lensAqueous humorEquator
The invention is directed to a lens that comprises an optic and at least two haptic rings, one positioned to rest against the posterior capsule distally outward from the optic zone, the other to rest on the anterior capsule some distance from the equator. The haptic rings of the lens are connected by segments of haptic material that may be arched or straight, and sections of open space to provide for ample circulation of the aqueous humor. The optic is suspended between the two haptic rings such that the distance between the optic and the anterior ring is constant while the distance between the optic and the posterior ring may vary according to the overall capsular dimensions of the eye of the patient.
Owner:ANEW IOL TECH
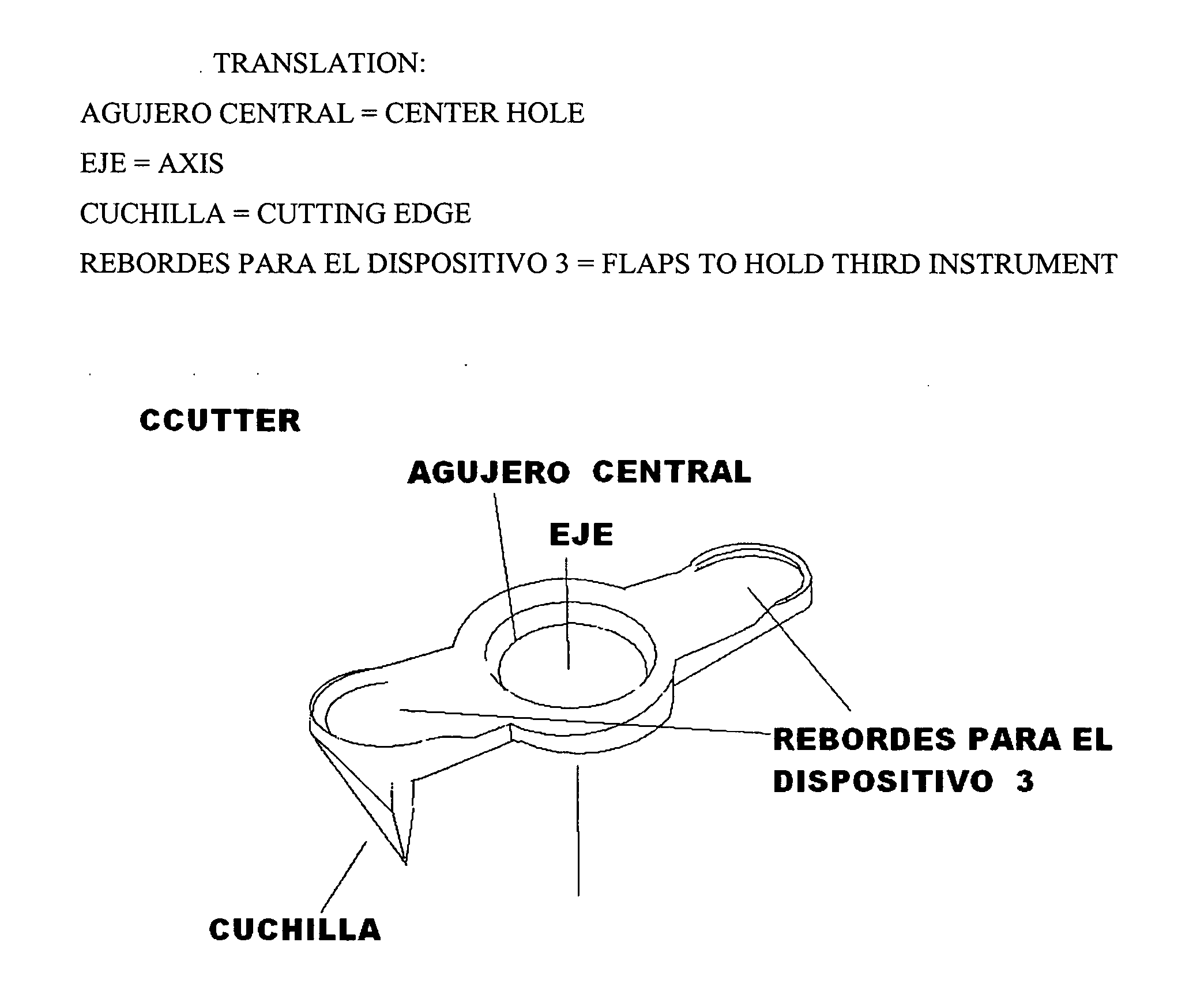
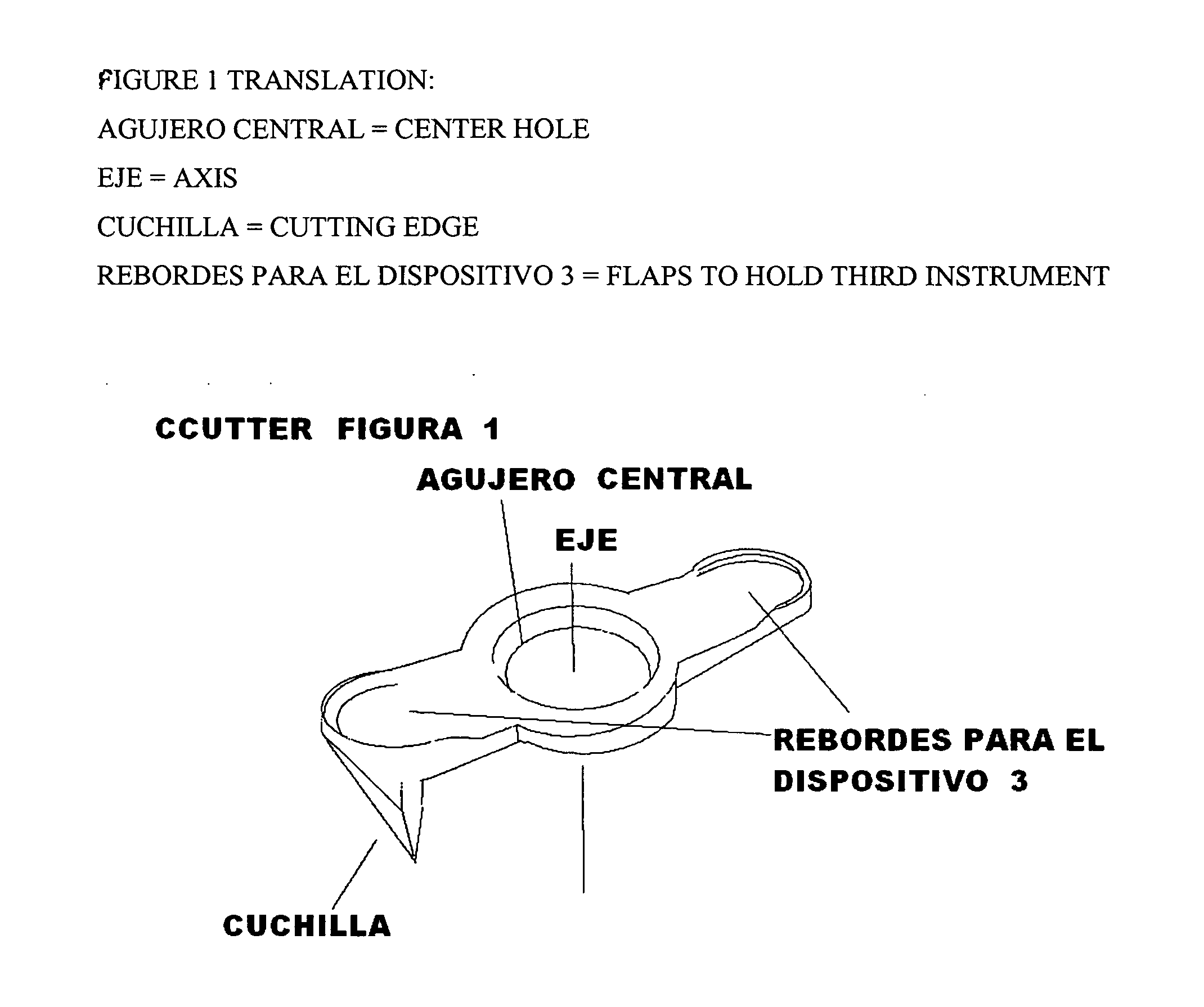
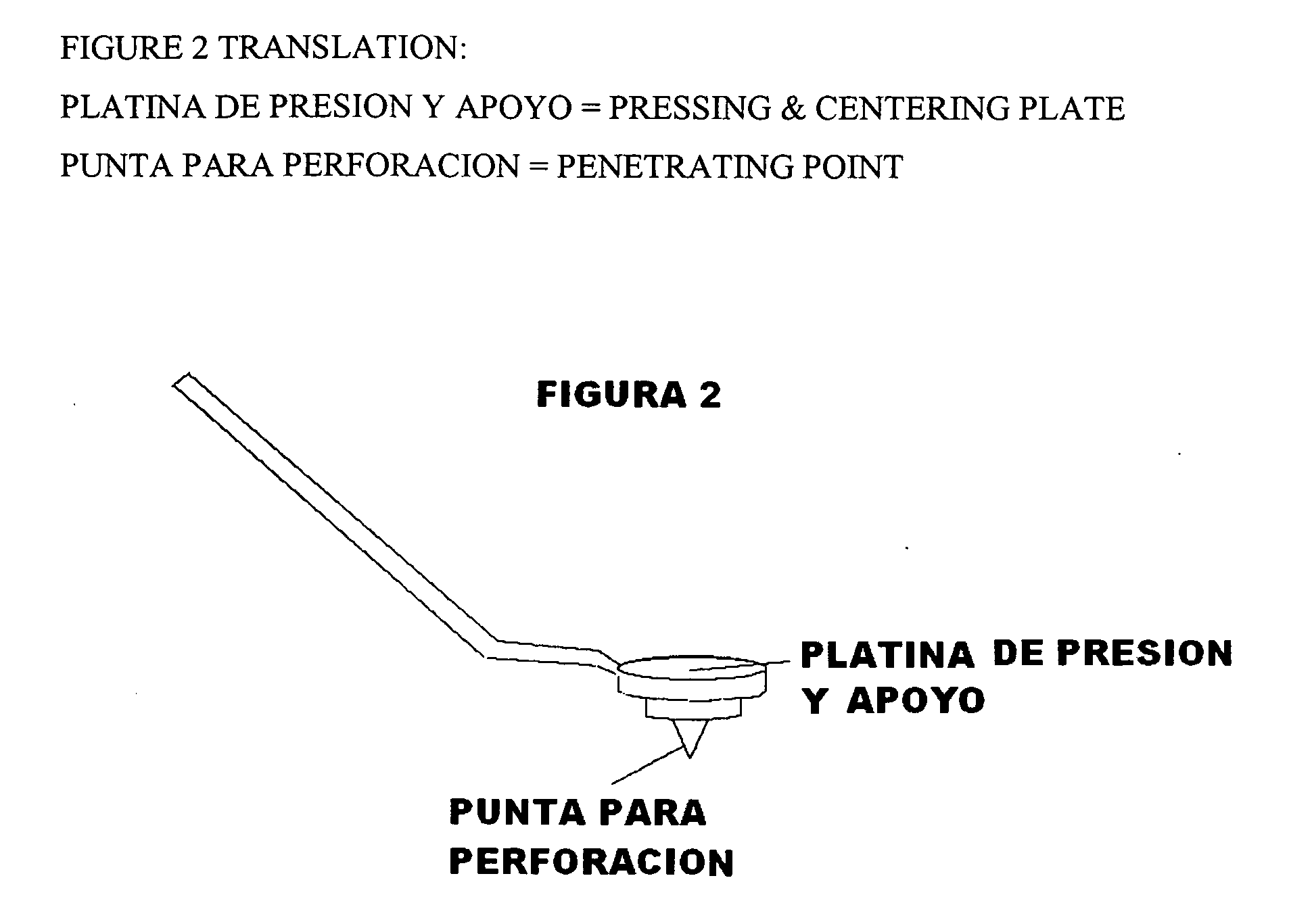
Ccutter
The CCUTTER is an innovative ophthalmologic surgical instrument that greatly facilitates the performance of cataract surgery. Cataract surgery is the most frequently performed surgery in the United States, with over one million cataract surgeries done each year. A cataract is a clouding of the eye's internal lens, which interferes with the individual's ability to see clearly. During surgery, a continuous circular tear (called capsulorhexis) is made in the anterior capsule of the crystalline lens of the eye, and the front surface of the cataract is opened to allow access to the clouded tissue inside. The cloudy portion is then removed, leaving the thin clear back surface of the lens (called the posterior capsule) in place. A lens implant is then placed in the shell of the natural lens, and the incision is closed. The CCUTTER is designed to improve the predictability and consistency of performing the capsulorhexis during cataract surgery.
Owner:BENITEZ PEDRO +1
Instrument system and procedure for phacoemulsification
An instrument system for phacoemulsification includes a femto- or picosecond laser device configured to dissect a lens of a patient's eye into lens fragments, open an anterior capsule of the lens of the eye, and make an incision in the eyeball so as to provide access to the lens of the eye. The system also includes device for fragmenting and aspirating the lens fragments through the access incision. A monitoring device is provided for monitoring results achieved by the phacoemulsification. A control device is configured to control the femto- or picosecond laser device and the device for fragmenting and aspirating the lens fragments, subject to parameters of the eye to be treated or of given surgical steps.
Owner:CARL ZEISS MEDITEC AG
Capsular opacification blocking lens
The invention is directed to a lens that comprises an optic and two haptic rings, one positioned to rest against the posterior capsule distally outward from the optic zone, the other to rest on the anterior capsule some distance from the equator. The haptic rings of the lens are connected by segments of haptic material that may be arched or straight, and sections of open space to provide for ample circulation of the aqueous humor. The optic is positioned against the posterior capsule as close as possible to the nodal zone of the eye.
Owner:ANEW IOL TECH
Automated ophthalmic device for performance of capsulorhexis
InactiveUS20050228419A1Reduction and elimination of complicationSmall sizeEye surgeryMechanical componentsHand held
A mechanical surgical device and device are provided for allowing the user to form a circular incision in an intraocular tissue, such as the anterior capsule of the eye, as part of an anterior capsulorhexis. Unlike the prior art, the device of the present invention is comprised of mechanical components, where the mechanical components drive a cutting member in a motion around a fixed pivot point to perform a continuous curvilinear capsulorhexis. Where the pivot point is the location of the slit incision in the cornea or the sclera through which the cutting tip is inserted to access the anterior capsule bag. The present device is hand held, compact and relatively light in weight. The present capsulorhexis device requires only a 1 mm incision in the corneal or scleral tissue. The present invention does not require the manual skill of the user to perform a circular incision.
Owner:AVANTGARDE DEVICES
Eye surgery training simulator
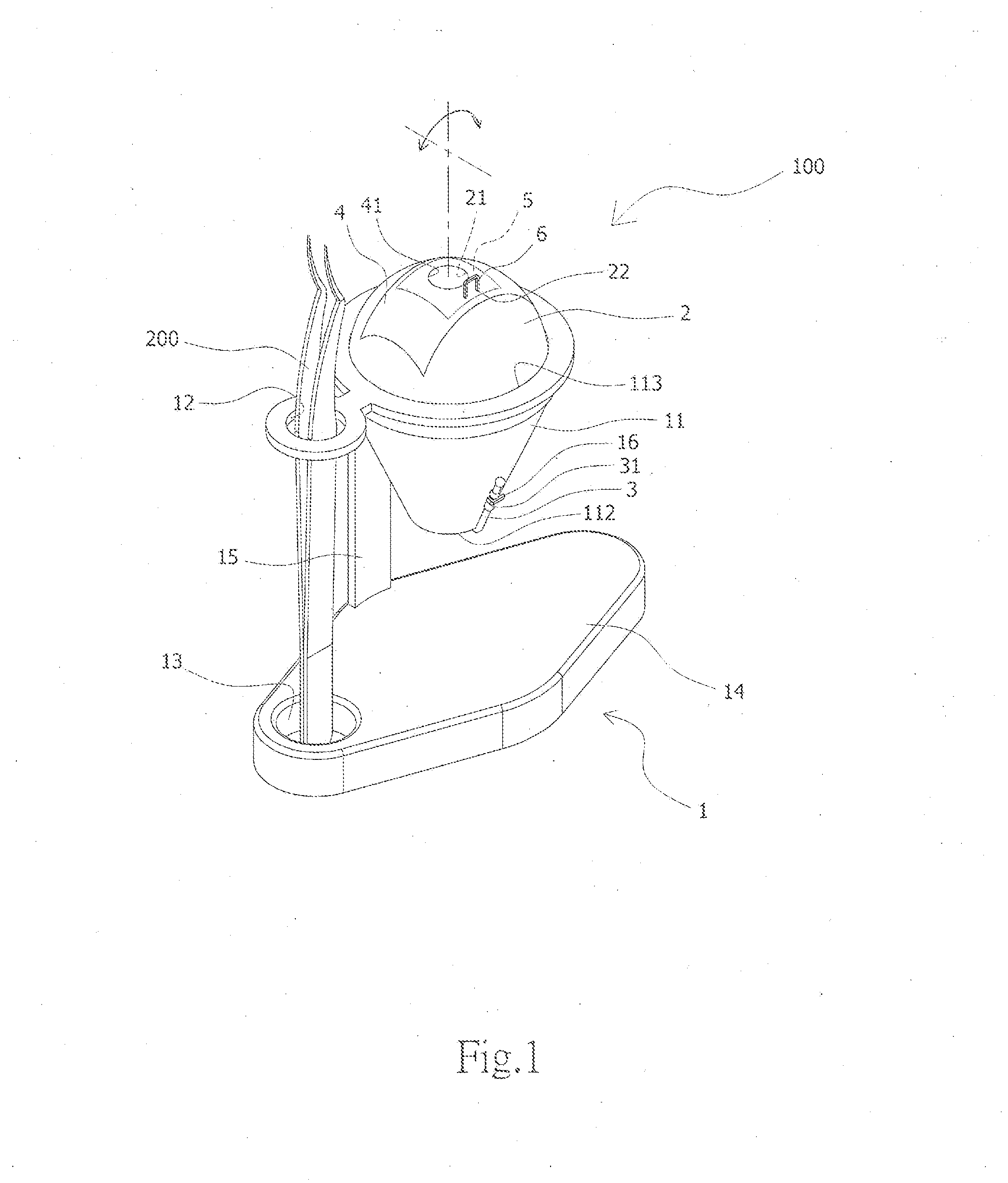
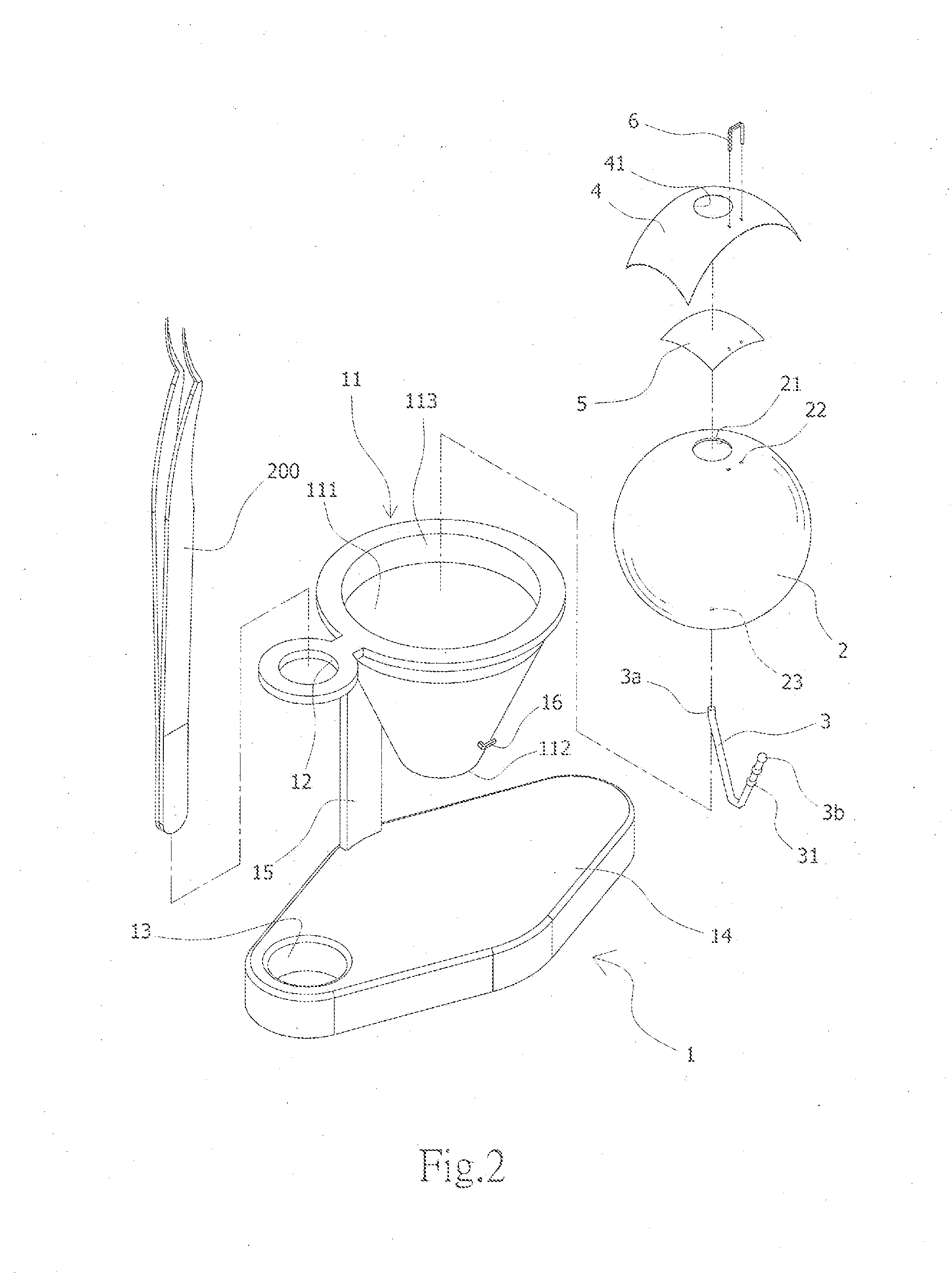
InactiveUS20160098944A1Reduce learning costsIncrease success rateEducational modelsElastic componentSurgery training
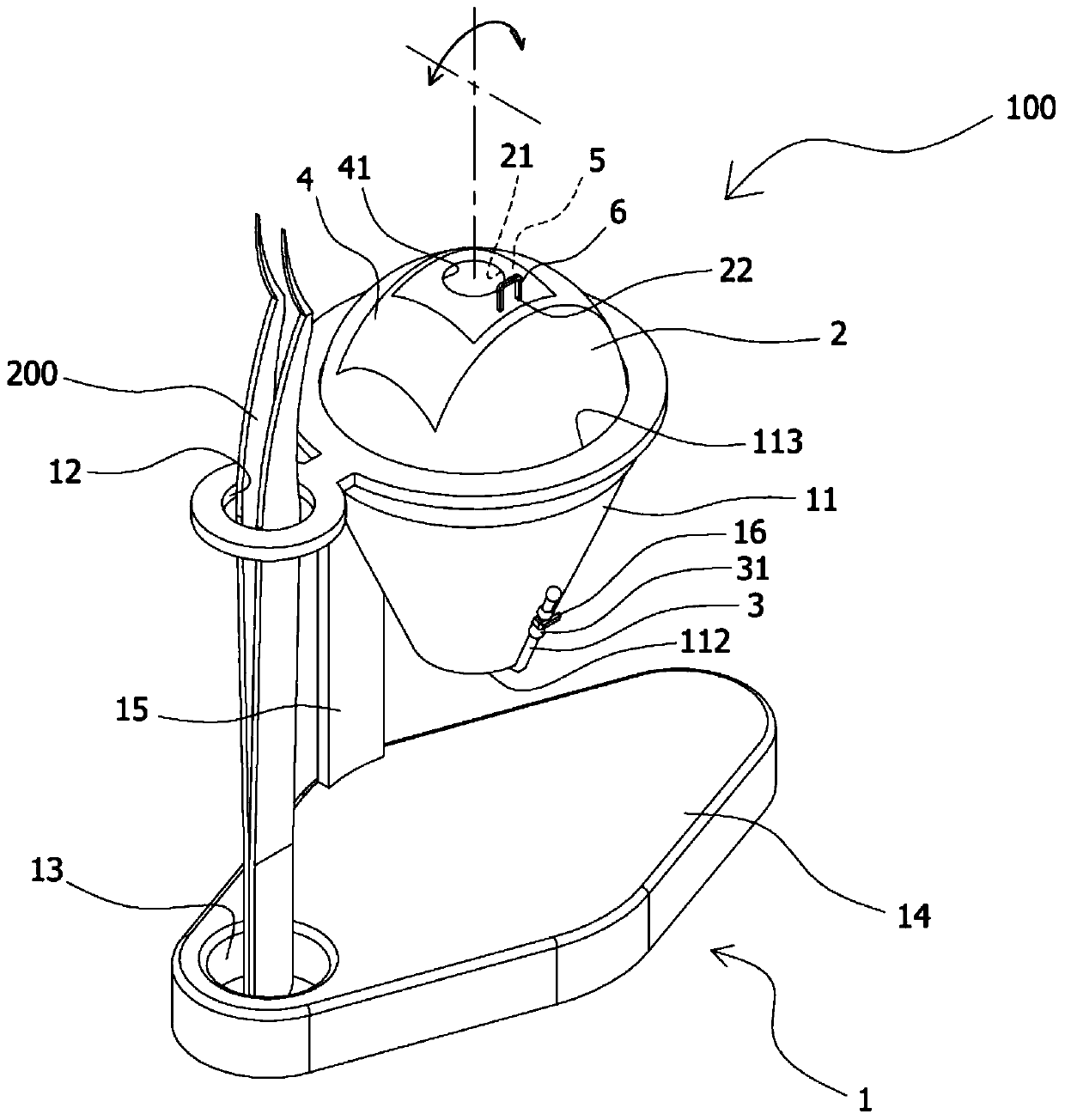
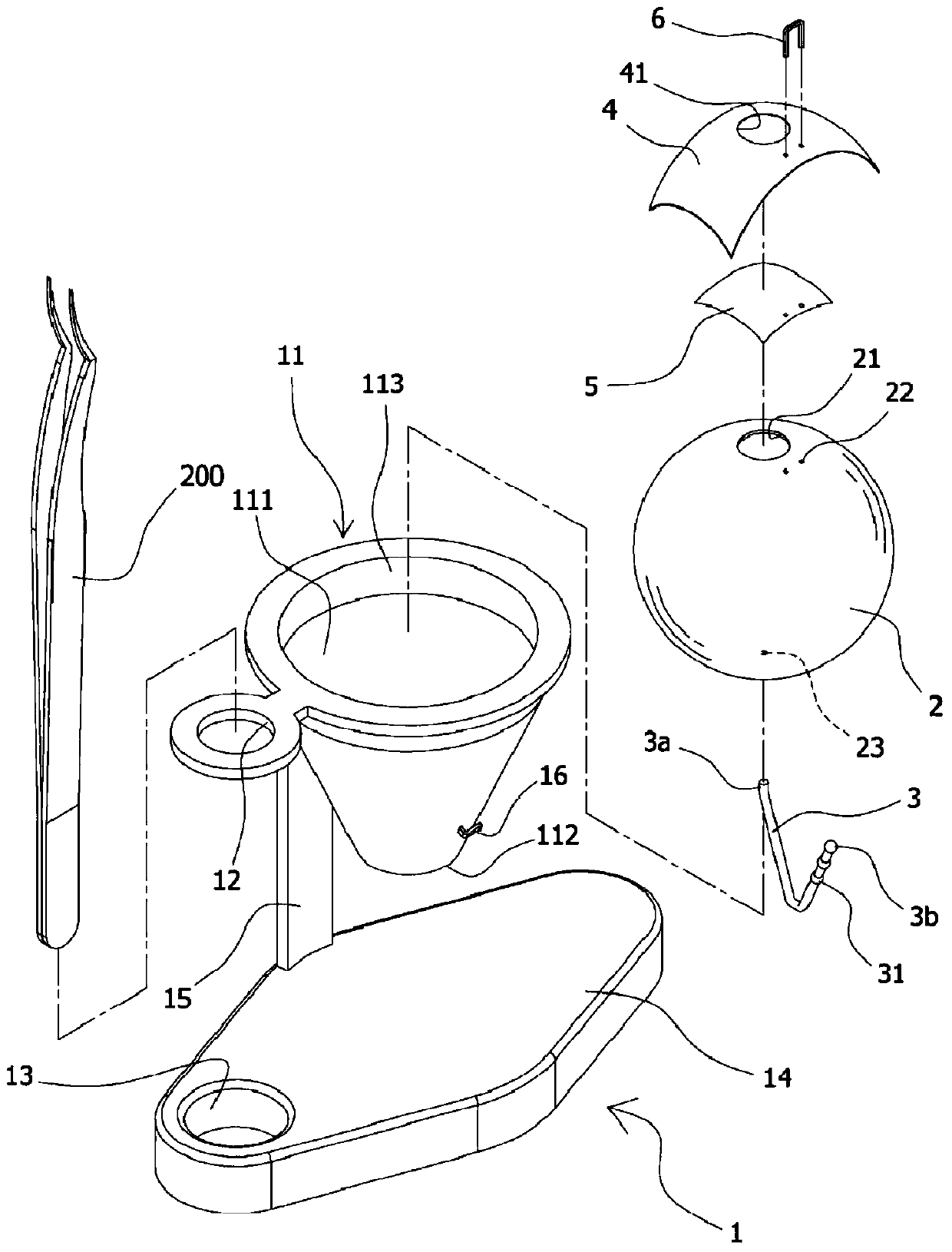
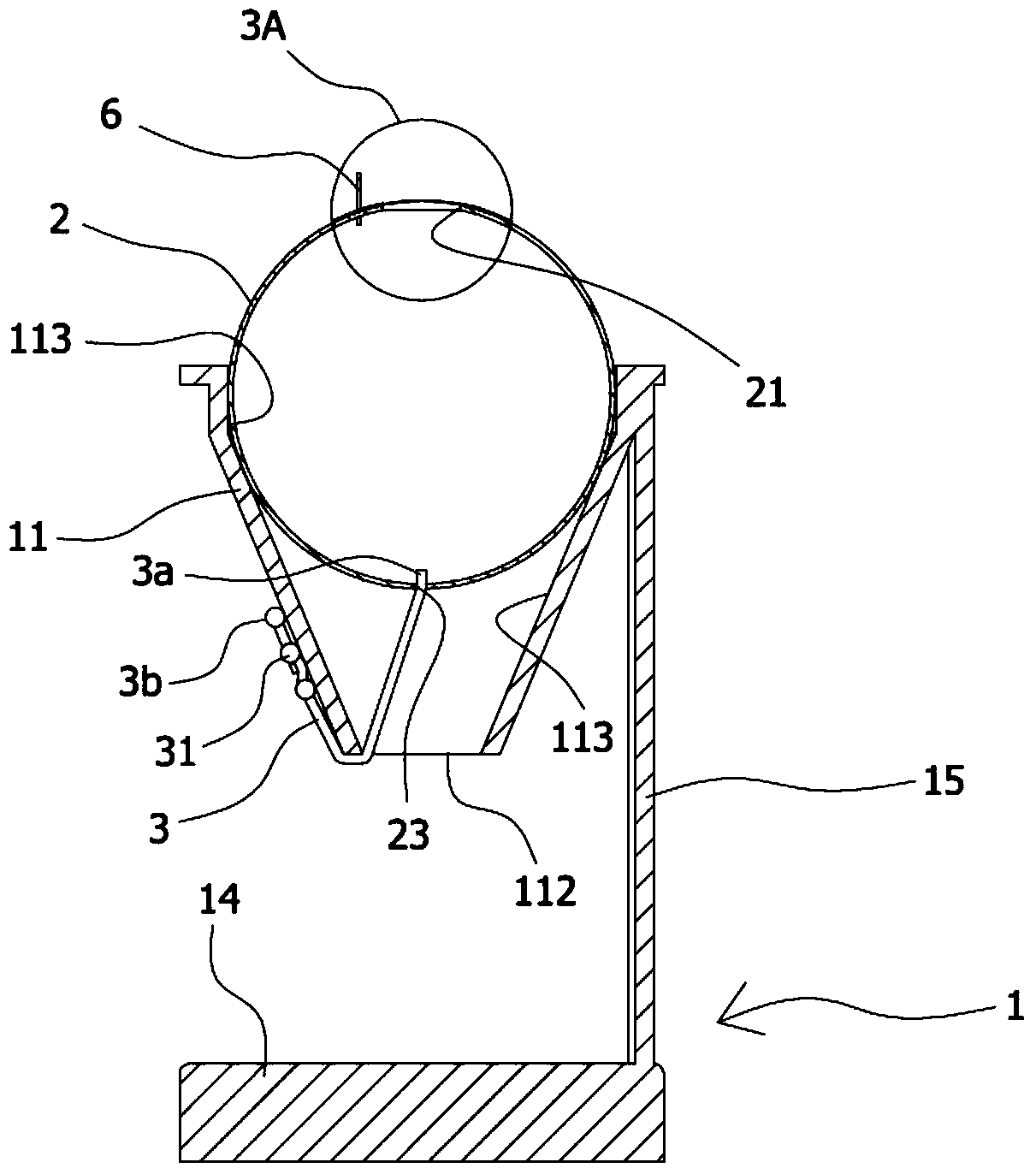
The present invention provides an eye surgery training simulator that can reduce learning cost and simulate the eyes in a more real way to allow surgery performers in practice to increase the success rate of surgery. The surgery training simulator compromise: a base with the accommodating tank, a hollow sphere inside said accommodating tank with a punched hole at the center of its top to simulate the eyeball rotation; a thin film covering the punched hole of said hollow sphere to simulate anterior capsule; a positioning sheet attached to the top surface of the hollow sphere with a hole corresponding to the punched hole; a positioning plug-in at the side of said punched hole that can limit the displacement of the capsulorhexis forceps; and at least an elastic component that can simulate eyeball muscle by pulling the hollow sphere and restore it after the rotation of the hollow sphere.
Owner:UNIVERSAL VISION BIOTECHNOLOGY CO LTD
Accommodating intraocular lens
ActiveUS20160310264A1Avoid it happening againAccommodation powerIntraocular lensMedicinePhakic intraocular lens
An intraocular lens includes a lens capsule expanding device and an optical portion. The lens capsule expanding device includes a front supporting portion to make contact with an inner surface of an anterior capsule. A rear supporting portion provided on a rear side of the front supporting portion makes contact with an inner surface of a posterior capsule while facing the front supporting portion. A connecting portion connects the front supporting portion and the rear supporting portion so as to have biasing force in a direction of separating the front supporting portion and the rear supporting portion from each other. Due to the biasing force, the front supporting portion presses the inner surface of the anterior capsule and the rear supporting portion presses the inner surface of the posterior capsule. The optical portion changes the curvature of a central portion according to movement of the connecting portion.
Owner:MIRAI EYE INC
Apparatus and methods for isolating lens capsule fluids
InactiveUS20060271188A1Increase the sealing areaReduce pressureEye surgeryWound drainsMembrane configurationFlange
A device to be inserted within a lens capsule during ocular surgery to contain fluids, including therapeutic and optical materials, used for irrigation within the capsule is provided. The device includes a deformable membrane within a ring. The ring may include flanges that increase sealing area between the ring and the posterior surface of an anterior capsule. One or more ports are provided for irrigation within the capsule. Reduced pressure in an area around the periphery of the membrane may be used for forming a sealing area between the device and the posterior surface of the capsule. Method for inserting the device into the capsule and performing irrigation or aspiration for polishing and for deforming the lens capsule is provided. The device may be placed in an eye for permanent isolation of fluids-within the lens capsule from tissue and fluids outside the capsule.
Owner:BROWN DAVID C
Eye surgery training simulator
The invention provides an eye surgery training simulator. The eye surgery training simulator is advantageous in that the learning costs can be reduced, the simulation is closer to the real state of the eyes, the practice of the performer can be facilitated, and the surgery success rate can be increased. The surgery training simulator comprises a seat body provided with an accommodation groove body; a hollow ball body, which is disposed in the accommodation groove body, is provided with a penetrating hole in the central part of the top end, and is capable of simulating rotation of an eyeball; a thin film sheet body, which is disposed on the penetrating hole in the hollow ball body in a covered manner, and is capable of simulating a front anterior capsule; a positioning sheet body, which is attached to the top surface of the hollow ball body, is disposed on the thin film sheet body in a covered manner at the same time, is provided with a hole corresponding to the penetrating hole, and can be attached repeatedly; a positioning inserting member, which is disposed next to the penetrating hole, and is capable of limiting displacement of capsule tearing tweezers; and at least one elastic assembly, which is used to simulate the eyeball muscle, is used for the traction of the hollow ball body, and is used to reset the hollow ball body after the rotation.
Owner:UNIVERSAL VISION BIOTECHNOLOGY CO LTD
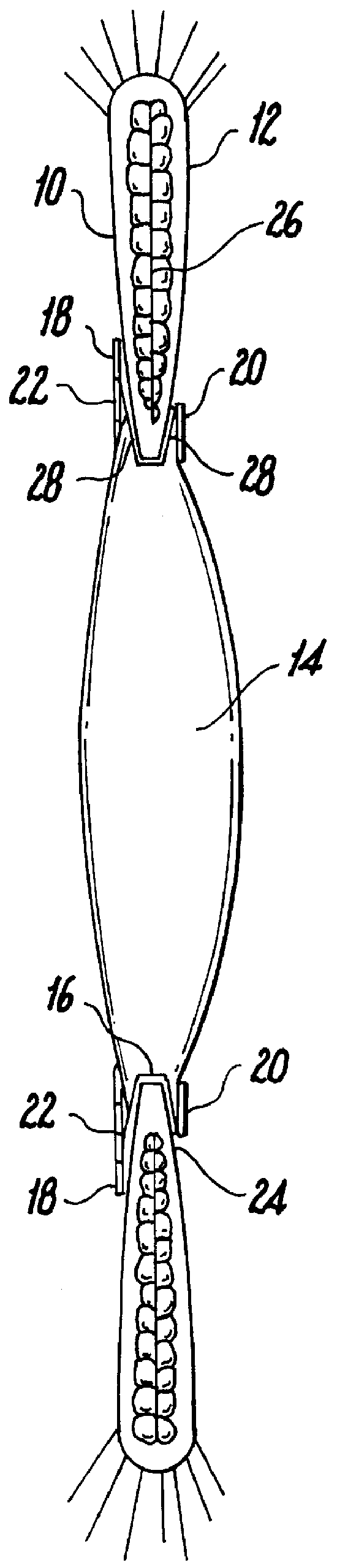
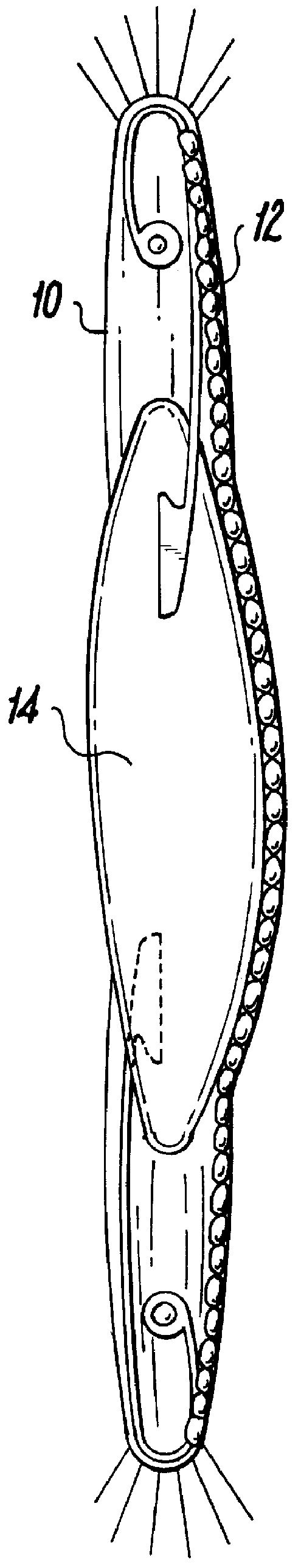
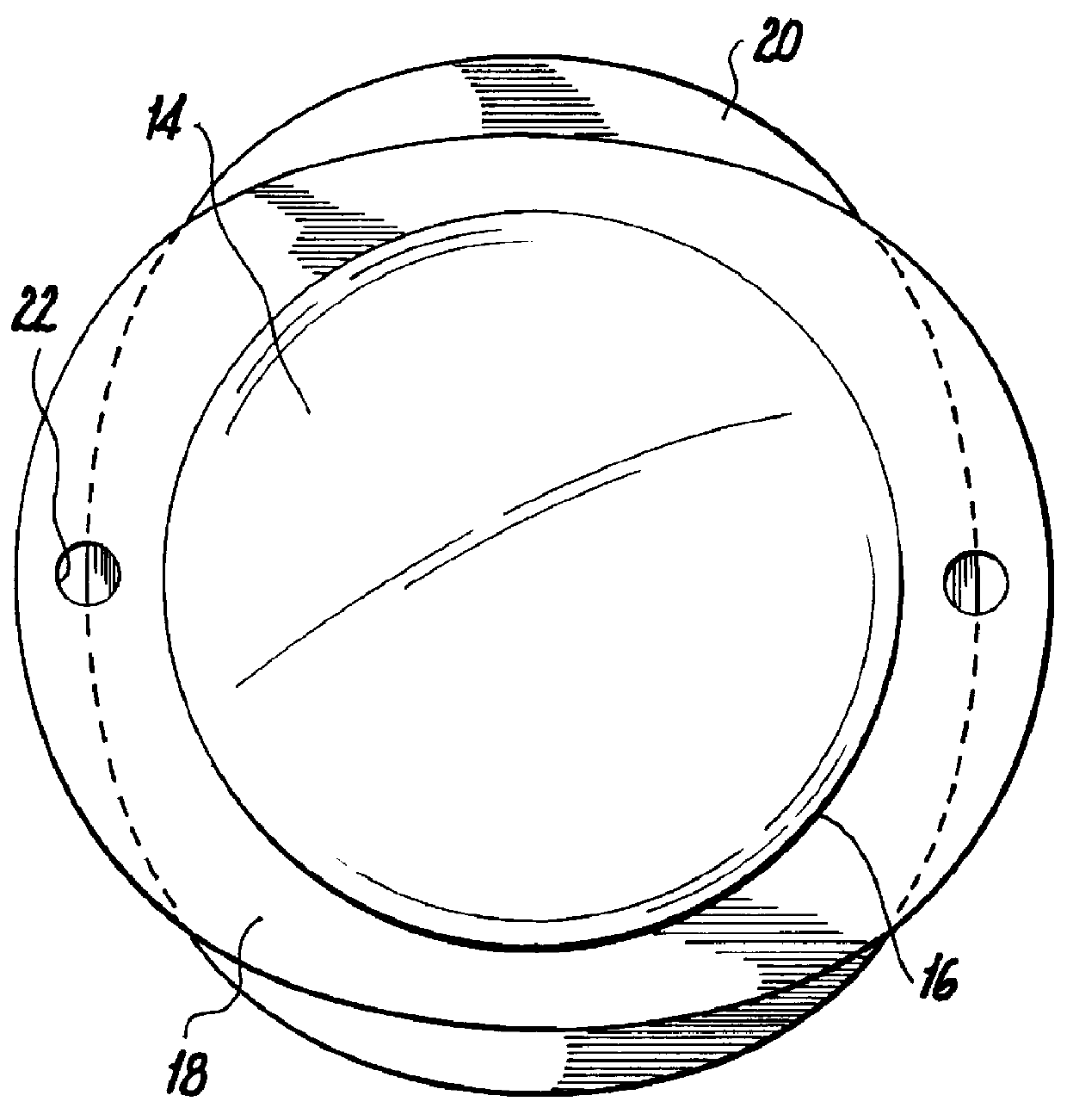
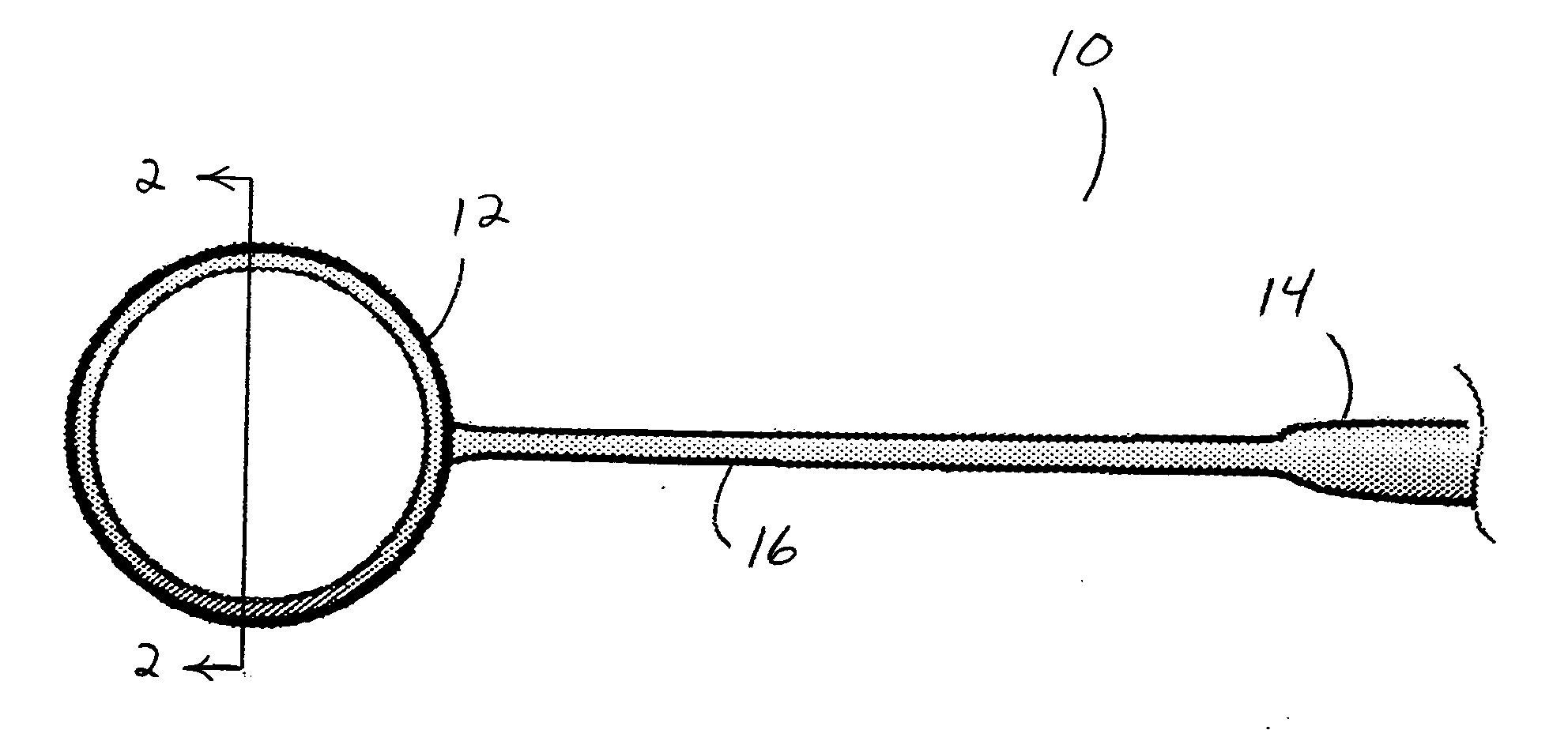
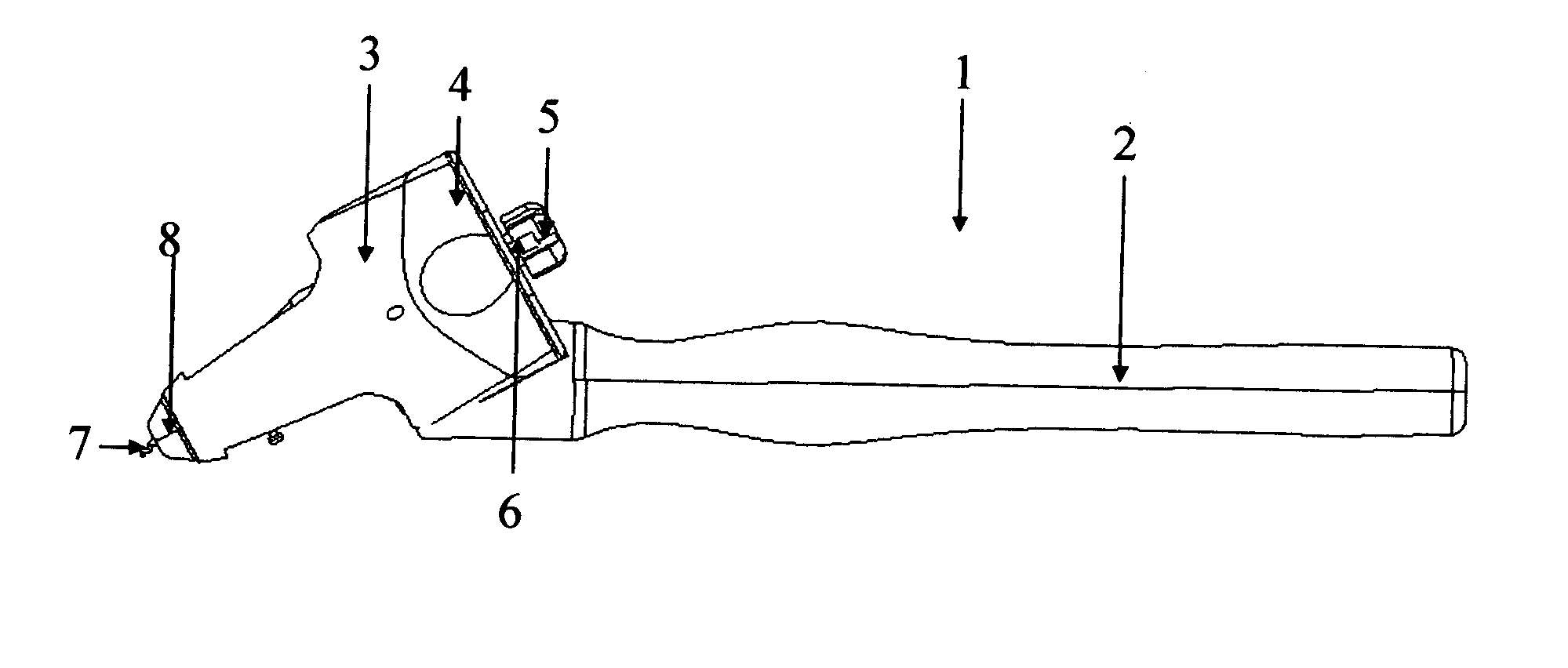
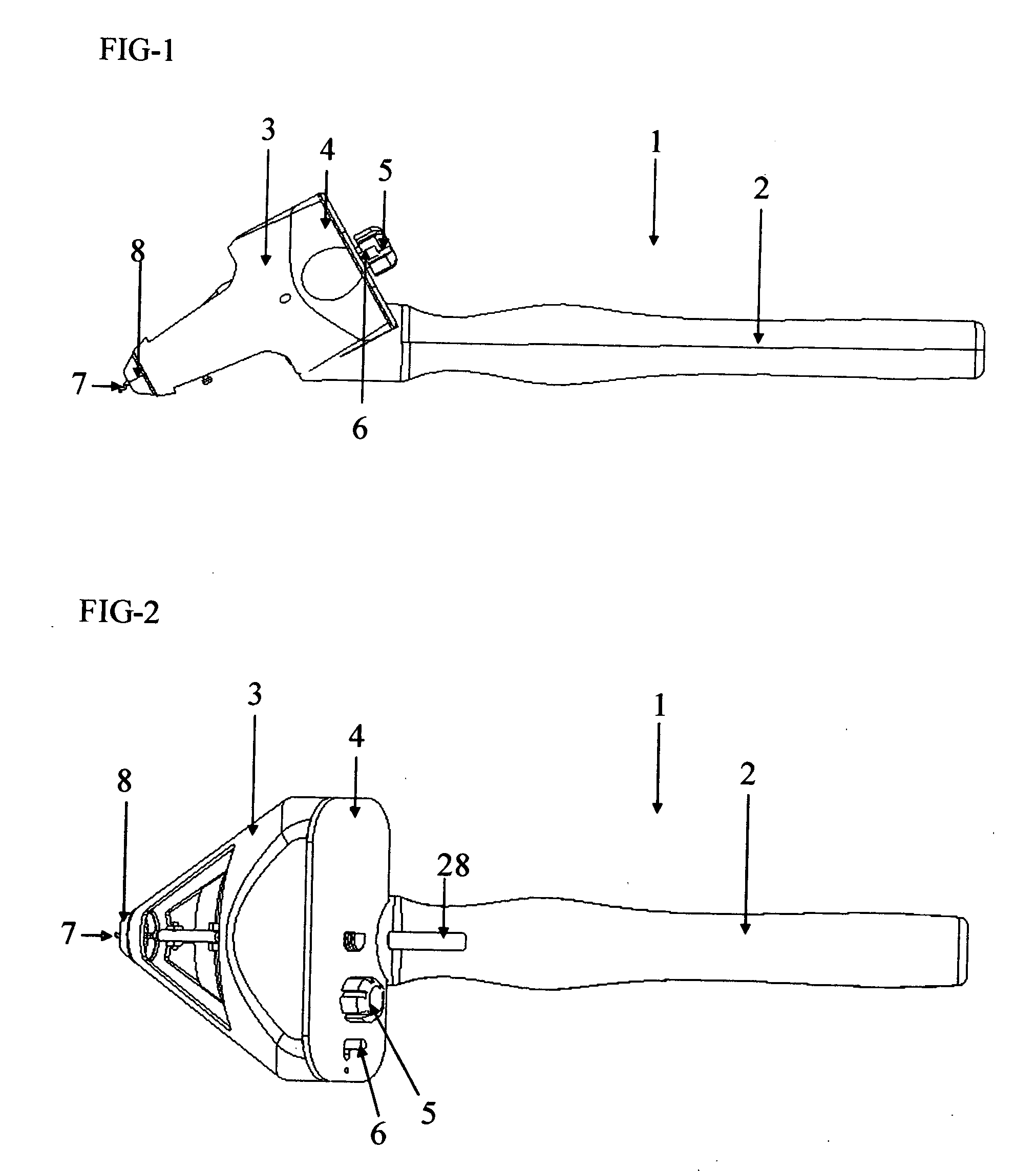
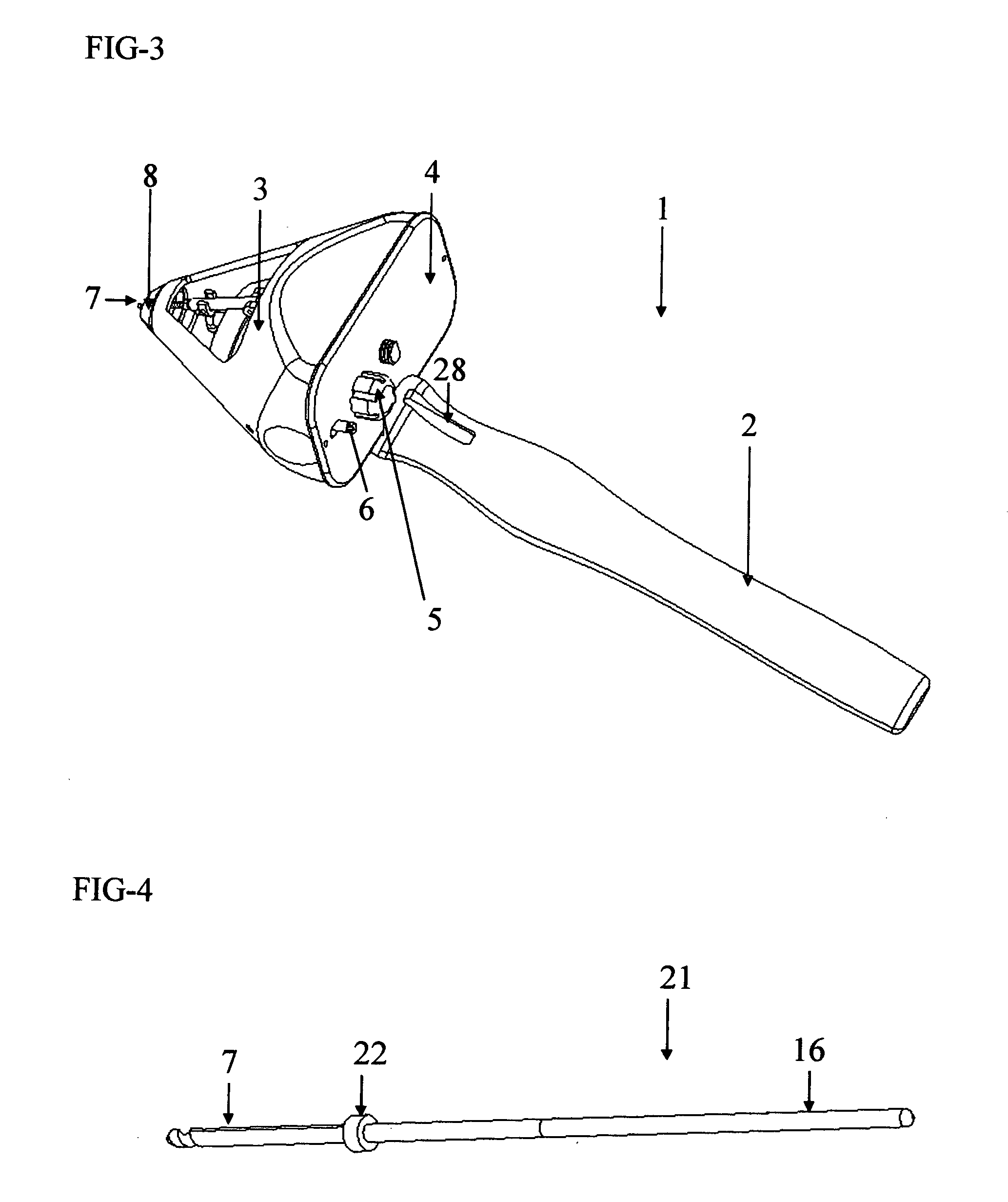
Capsulotomy devices and methods
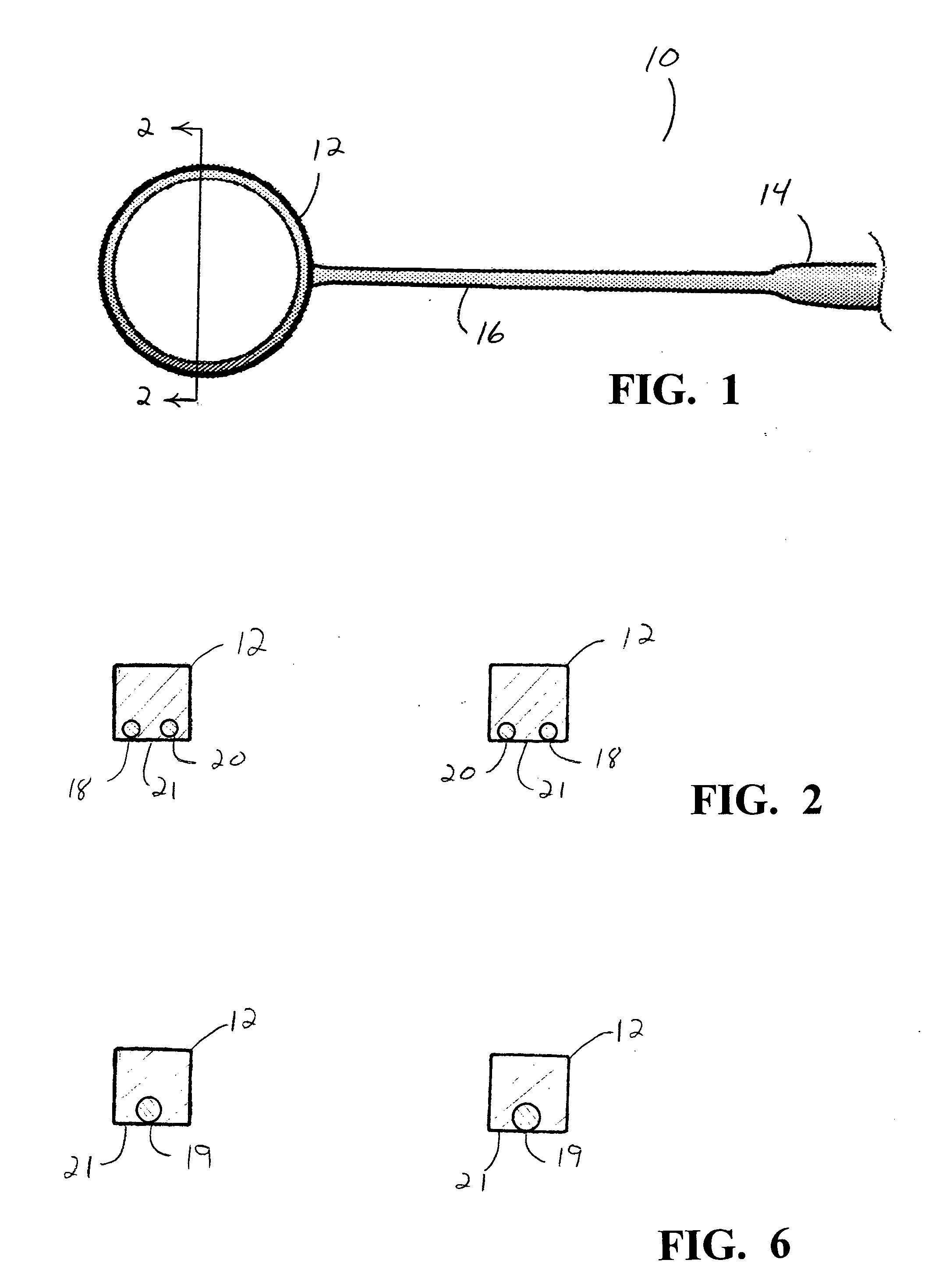
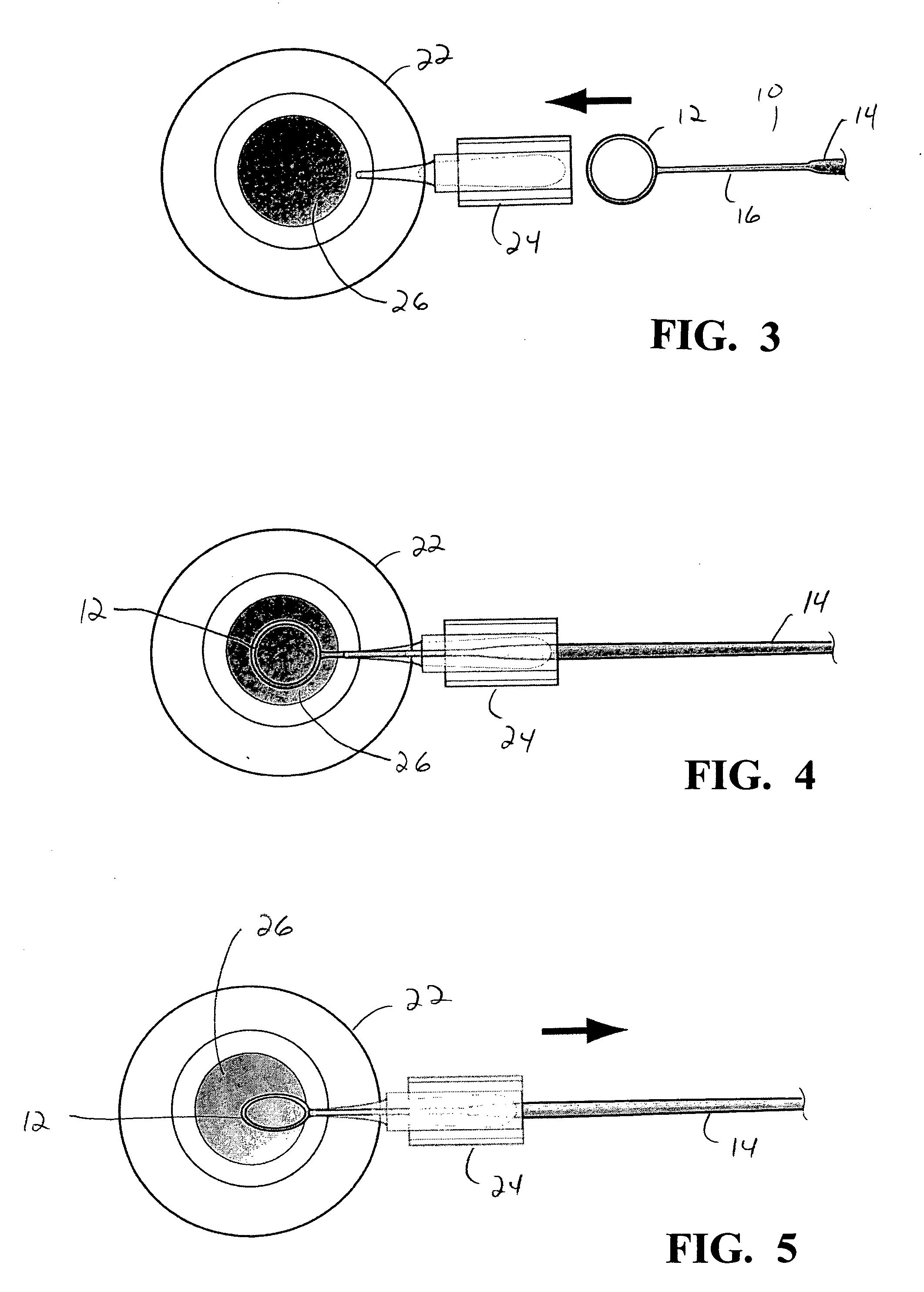
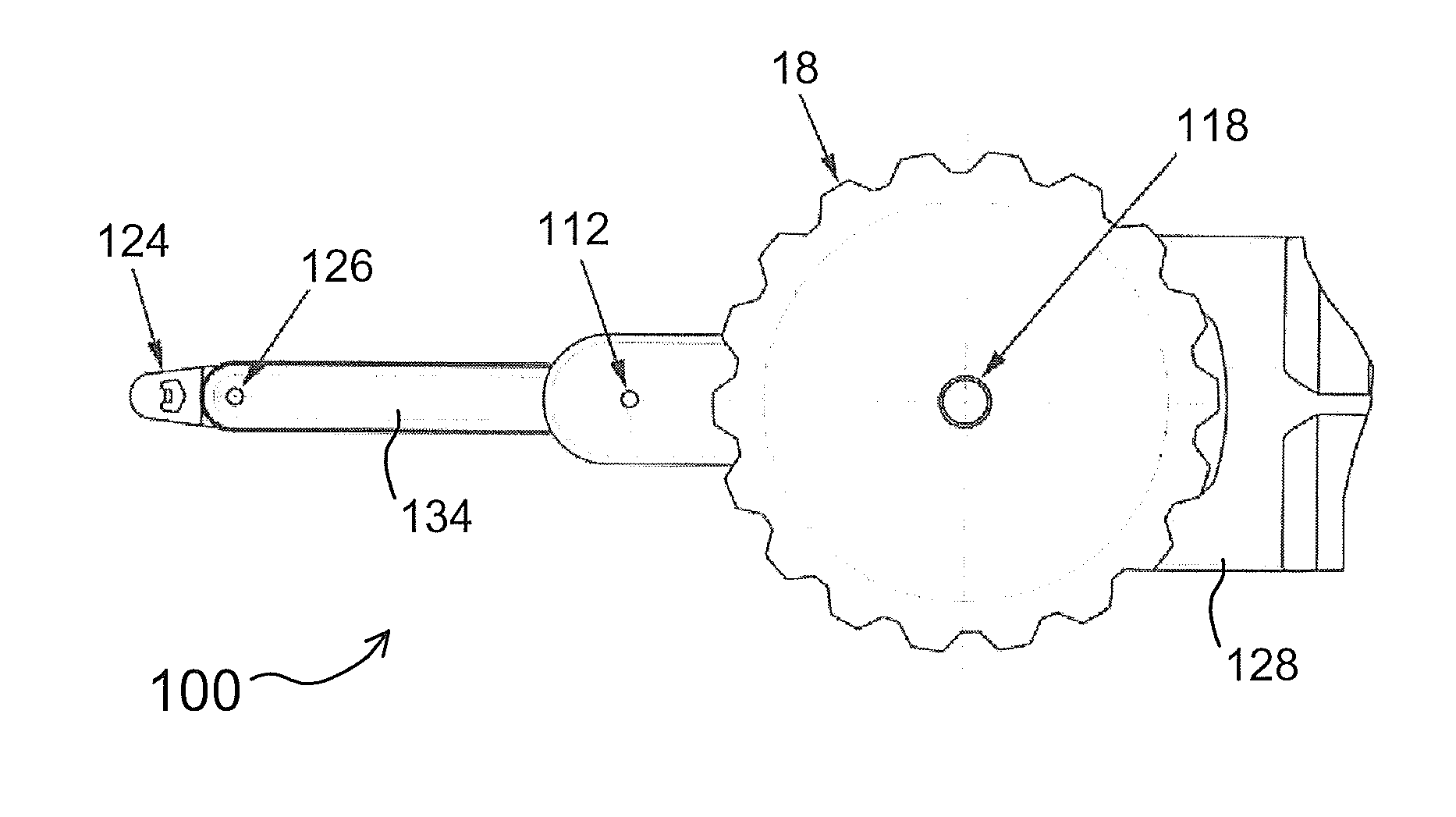
An Anterior Capsule Remover (“ACR”) device allows an ophthalmologist to safely and efficiently remove an anterior portion of the lens capsule at the beginning of cataract surgery. The device allows the surgeon to safely, efficiently and consistently create a circular central opening of a predetermined diameter in the top of the lens capsule. An exemplary anterior capsule remover apparatus includes a handle and a distal shaft that holds a blade or cutter. One or more o-rings attached to a thumbwheel and to the blade allow the surgeon to easily create a circular, central opening in the anterior capsule of predetermined diameter.
Owner:OJO
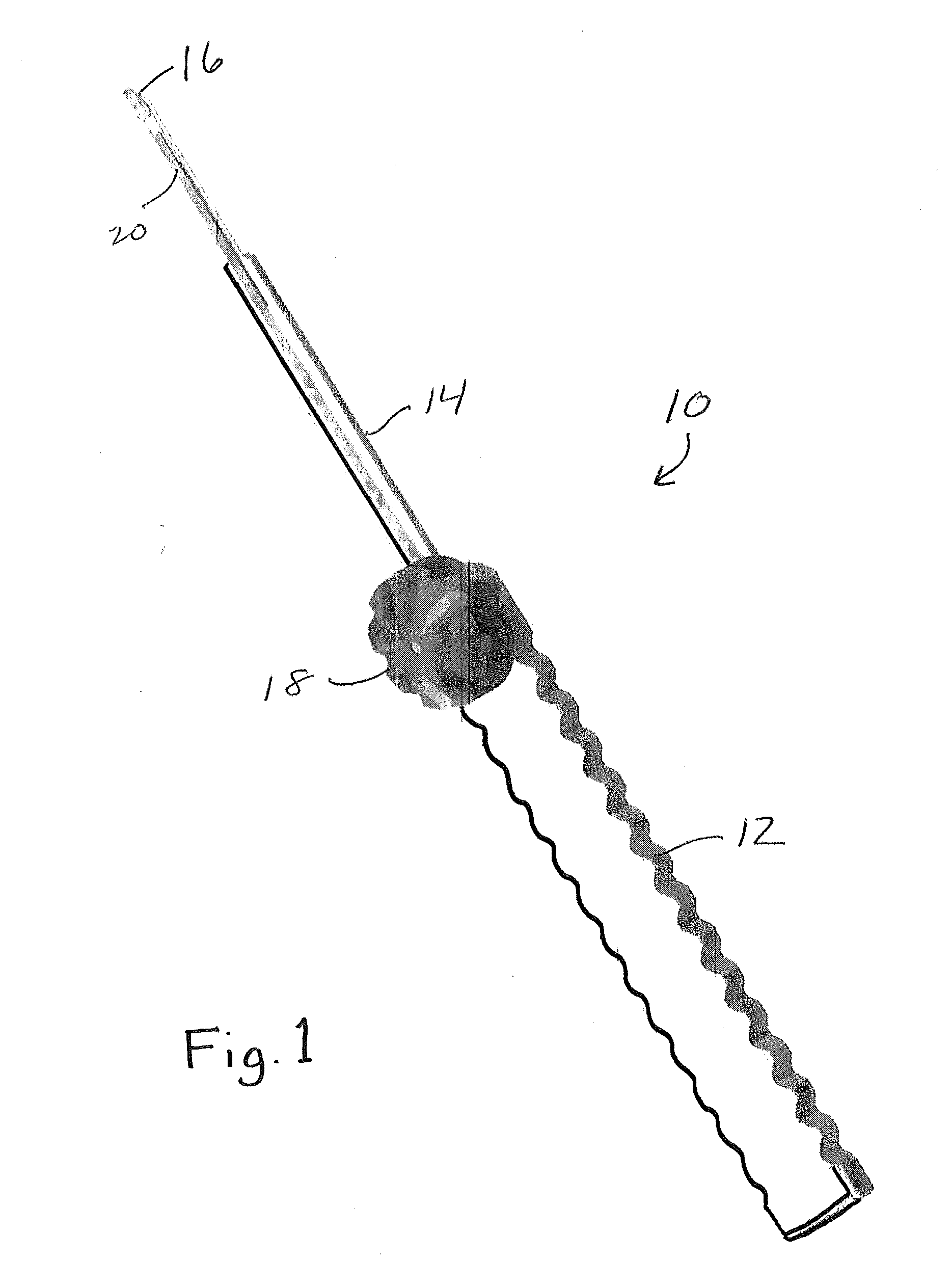
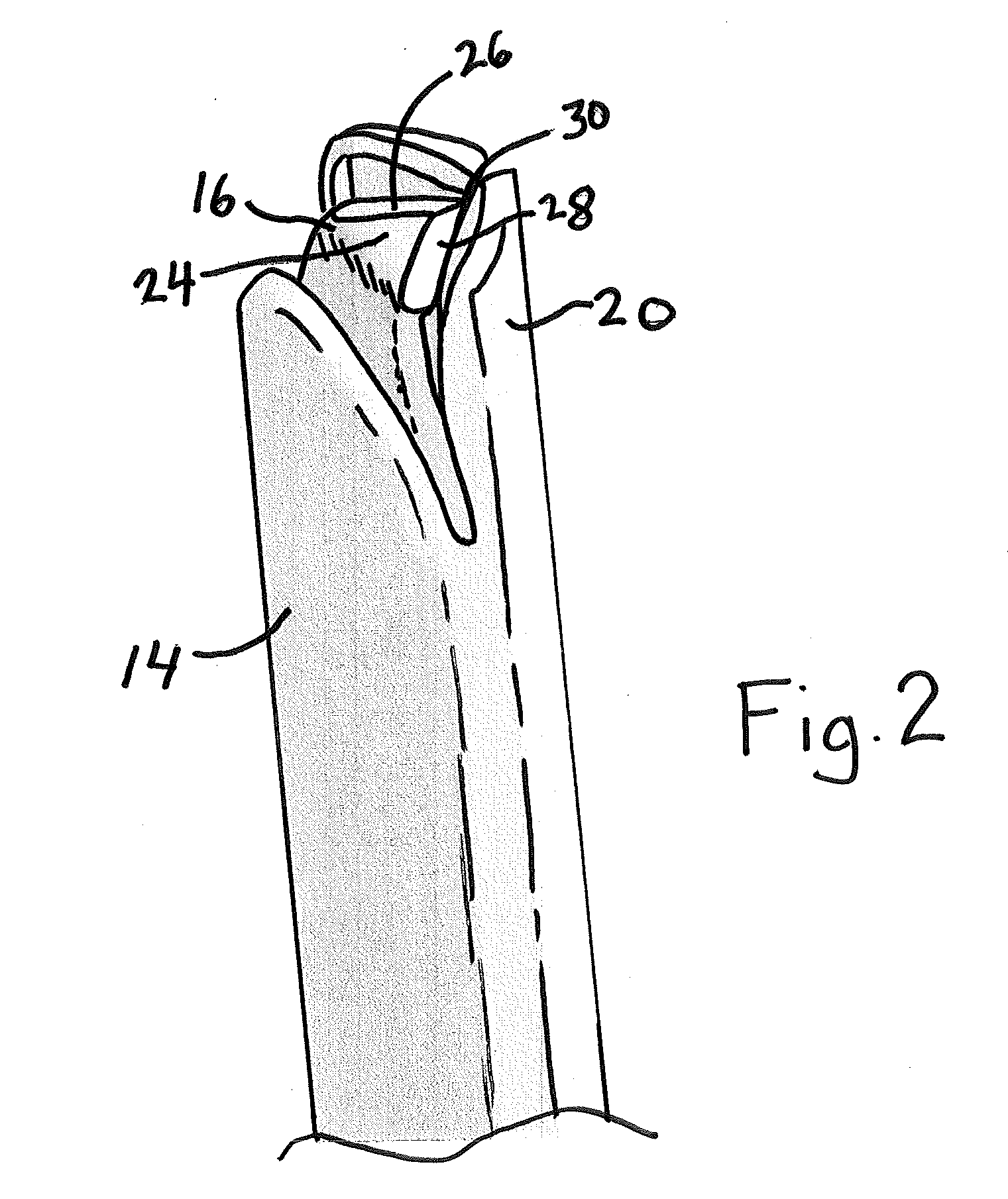
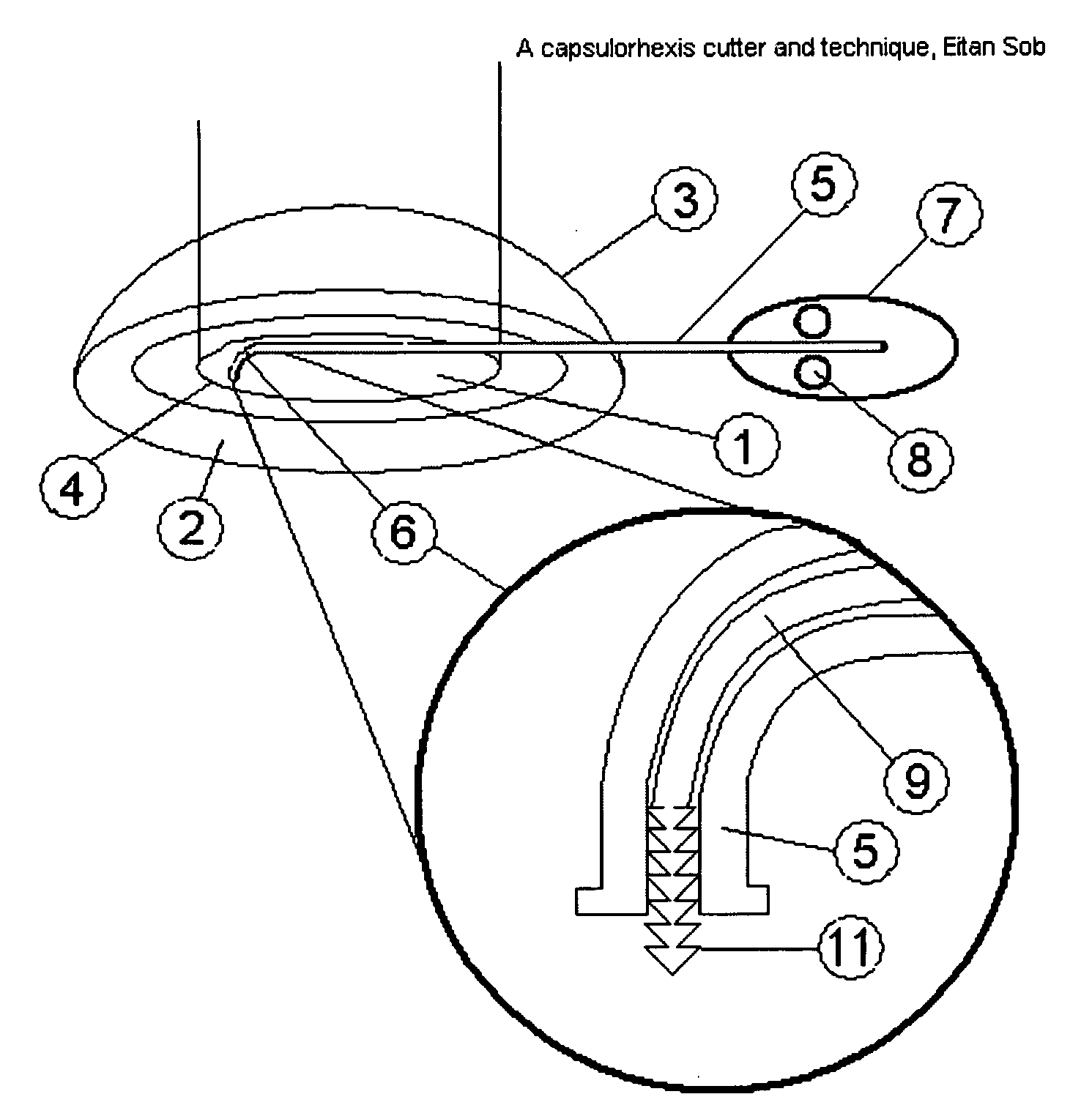
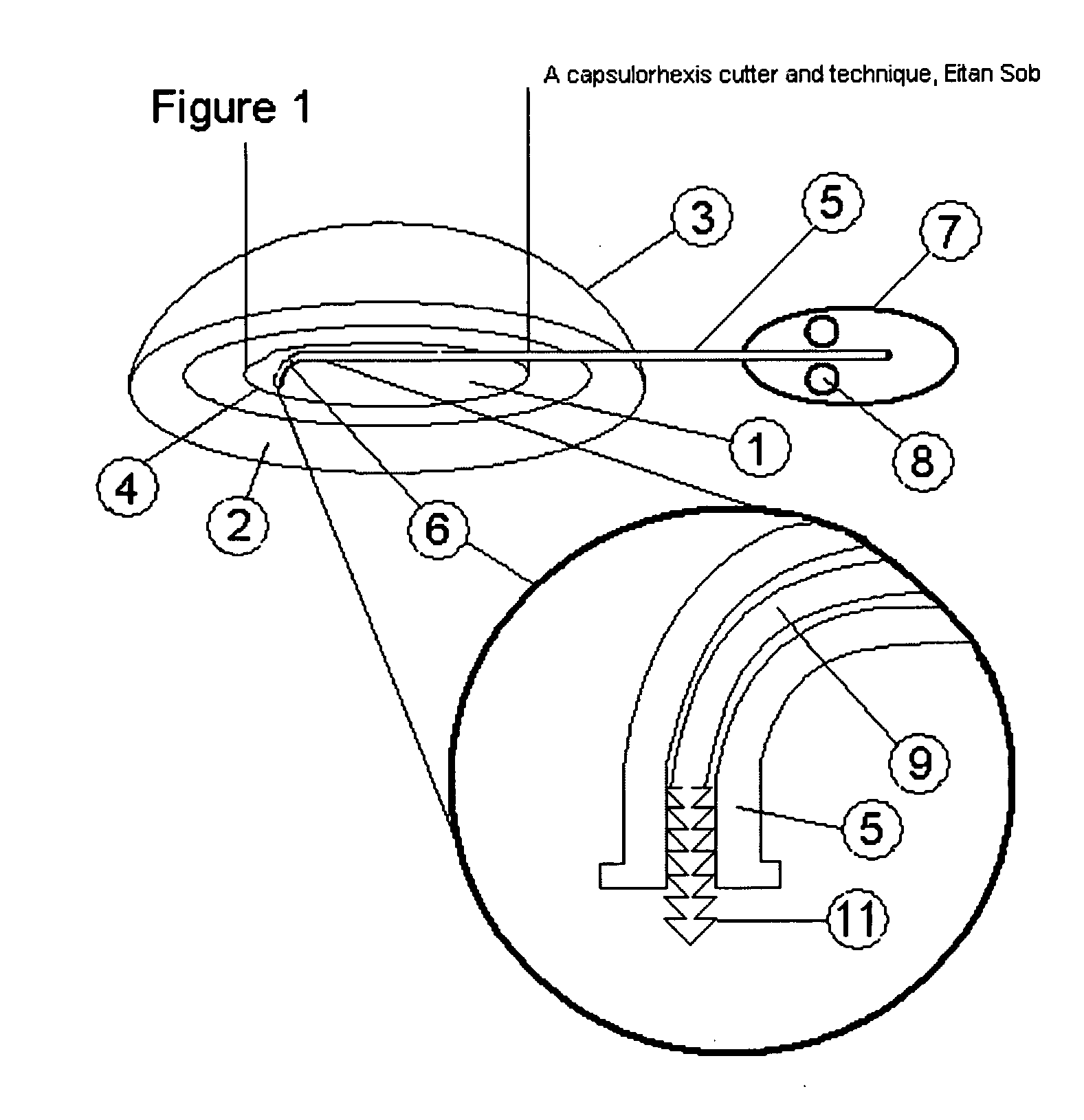
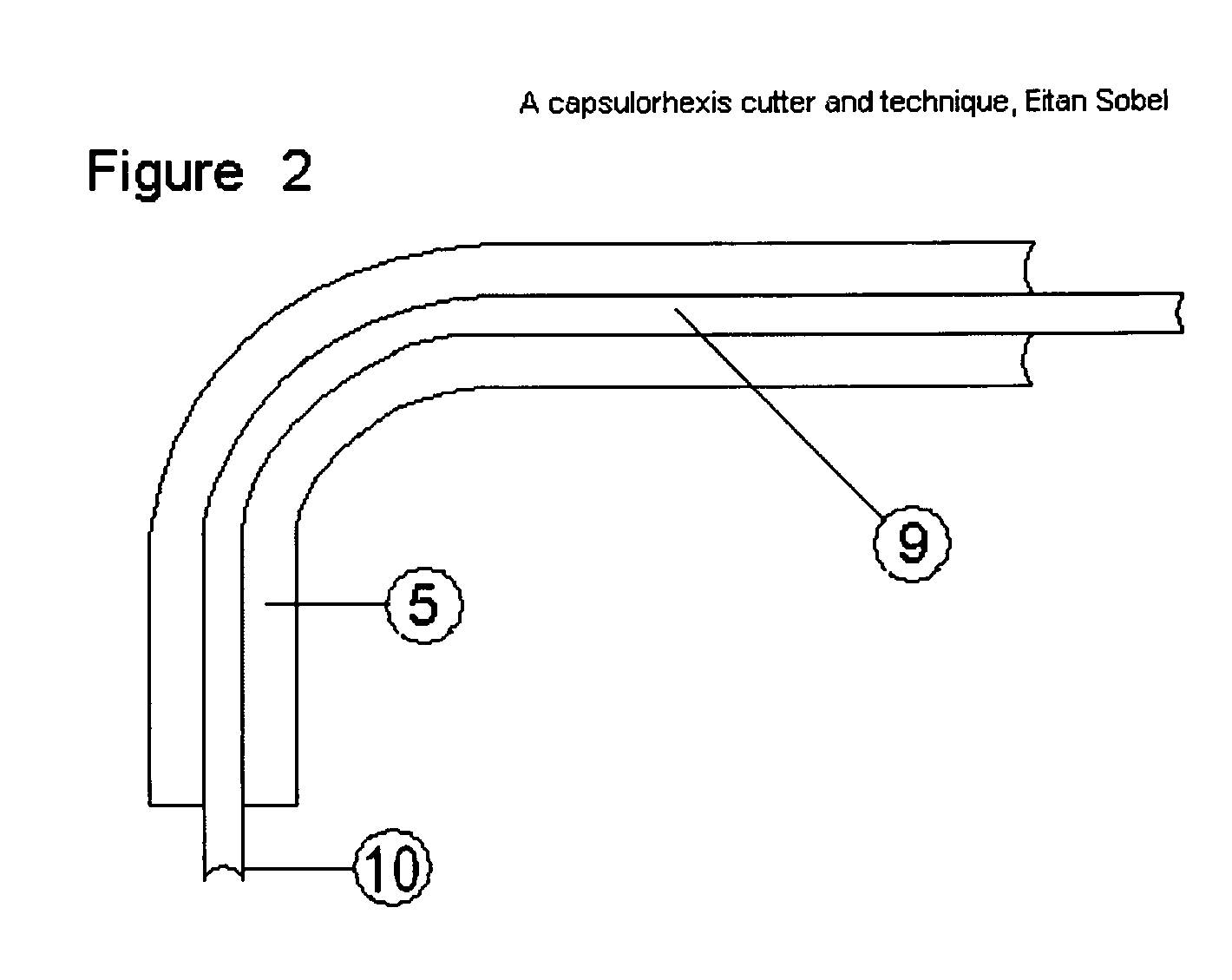
Capsulorhexis cutter and technique
A device for performing a capsulorhexis incision inserted via a small wound cut in the corneal or sclera tissue. The device includes a motorized cutting element and a handle The motorized cutting element has mechanical means to create a cut in the anterior capsule tissue along a planned capsulorhexis line, said mechanical means tear small fragments of the anterior capsule in a fashion that may be similar to either a saw, a rotating chipping bit, a continuous hole puncher or a combination of those mechanical means thereby, creating a relatively smooth cut without the risk of undesired capsular tear.
Owner:SOBEL EITAN
Surgical training eye apparatus
An apparatus for teaching and practicing an ophthalmologic surgical technique of creating the continuous curvilinear capsulorhexis comprises a housing with a first base end and a second suction cup end for holding a malleable body; a flexible film or cellophane-type membrane covers the operating area of the malleable body; this flexible film can be held into place on the first base end with a first cap with an aperture or opening; in between the first base end and the second suction cup end, there can be a flexible stock with a threaded or friction connection. There can also be a second cap addition, which simulates a cornea and anterior chamber, which can be filled with viscoelastic material, which can increase the pressure in the eye and flattens the anterior capsule.
Owner:STOLL STUART
Method of maintaining the structure of an opening in the anterior or posterior capsule
ActiveUS20130013061A1Rigid enoughReduces and minimizes glareIntraocular lensCorrective surgeryIntraocular lens insertion
A method of maintaining the structure of an opening in the anterior or posterior capsule formed by a capsulorhexis whereby a device is inserted into opening in the anterior or posterior capsule, the device having a main body including a peripheral portion and an opening therethrough, wherein the peripheral portion engages with the inside peripheral edge of the opening in the anterior or posterior capsule, wherein the device is inserted into the opening in the anterior or posterior capsule after an intraocular lens has been inserted into the capsular bag of an eye during cataract corrective surgery.
Owner:CORONEO MINAS THEODORE
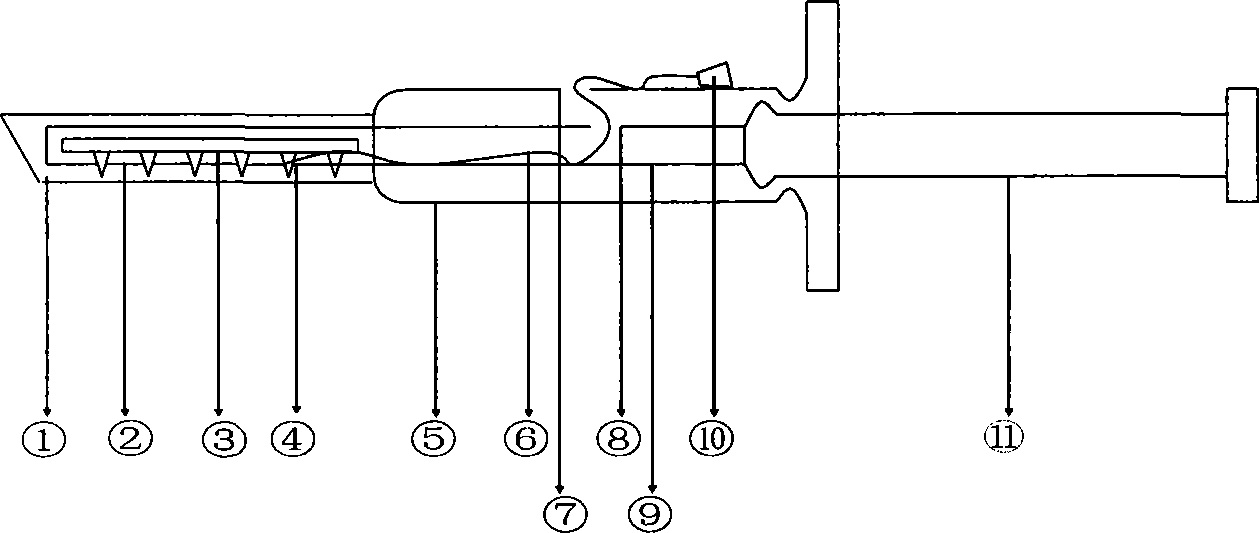
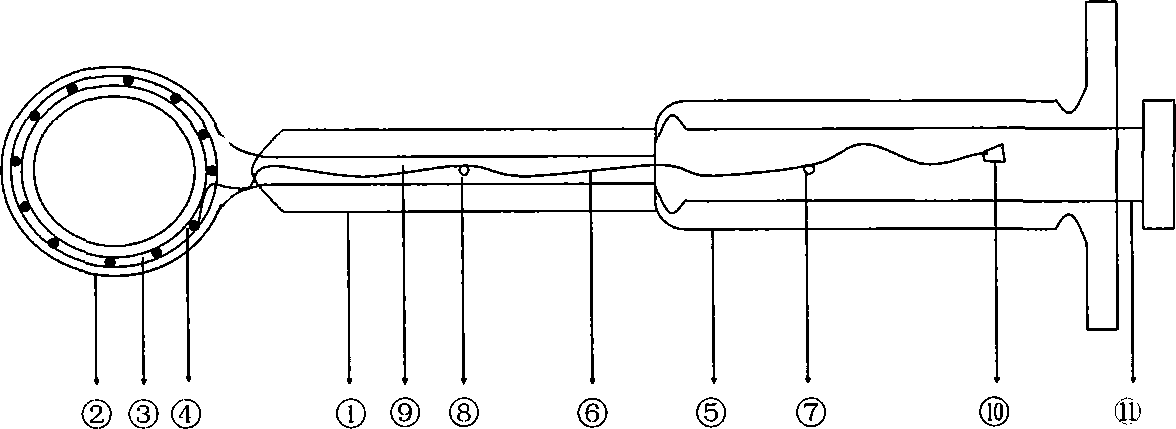
Automatic vesicle gashing device
InactiveCN101669852AEasy exitReduce surgical skill level requirementsEye surgeryBiomedical engineeringMembrane configuration
The invention relates to an apparatus for automatically gashing crystalline lens anterior capsule membrane into a circle with a diameter as required during the implementation of cataract operation. The apparatus comprises a power control box capable of being connected with a soft shaft, a handle, and a vesicle gashing cutter made of memory metal. The inner part of the handle comprises the soft shaft, an internal core, and a transmission rod with a clamping device. The vesicle gashing cutter is straight when pulled back to the tube, and restored to be circular based on memory shape when pushedout of the tube; and the anterior capsule membrane is cut or sheared by the cutter head along the arc line so as to form smoothly circular shape with corresponding diameter. The vesicle gashing cutteris simply structured and convenient to be operated, can get into the atria easily and automatically, thereby improving the success ratio of operation and reducing the operation difficulty.
Owner:王伟 +1
Apparatus and methods for isolating lens capsule fluids
A device to be inserted within a lens capsule during ocular surgery to contain fluids, including therapeutic and optical materials, used for irrigation within the capsule is provided. The device includes a deformable membrane within a ring. The ring may include flanges that increase sealing area between the ring and the posterior surface of an anterior capsule. One or more ports are provided for irrigation within the capsule. Reduced pressure in an area around the periphery of the membrane may be used for forming a sealing area between the device and the posterior surface of the capsule. Method for inserting the device into the capsule and performing irrigation or aspiration for polishing and for deforming the lens capsule is provided. The device may be placed in an eye for permanent isolation of fluids-within the lens capsule from tissue and fluids outside the capsule.
Owner:BROWN DAVID C
Multicavity three-sac prostate capsule dilating and tissue cracking shifting catheter
The invention discloses a multicavity three-sac prostate capsule dilating and tissue cracking shifting catheter which comprises a catheter main body, wherein a catheterization cavity, a front sac cavity, a back sac cavity, an inner sac cavity and a front flushing cavity are arranged in the catheter main body; a front flushing opening and a catheterization port communicating to the front flushing cavity and the catheterization cavity are separately formed in the front end of the catheter main body; a front sac communicating to the front sac cavity is arranged on the back part of the catheterization port; a back sac communicating to the back sac cavity is arranged on the back part of the front sac; and an inner sac communicating to the inner sac cavity is arranged at the front end in the front sac. The catheter can be also effectively fixed to the affected part while meeting an operating demand of dilating the membranous urethra and the prostate urethra in steps or in turns, so that traction positioning and hemostasis by compression of the catheter are achieved. According to the catheter flushed by a lot of water flows, caused wounds are minimal, patients have small pain, and the cure rate can be increased effectively and the postoperative recovery is accelerated.
Owner:黄正
Preparation method of ophthalmological viscoelastic agent with selective anterior capsule staining function
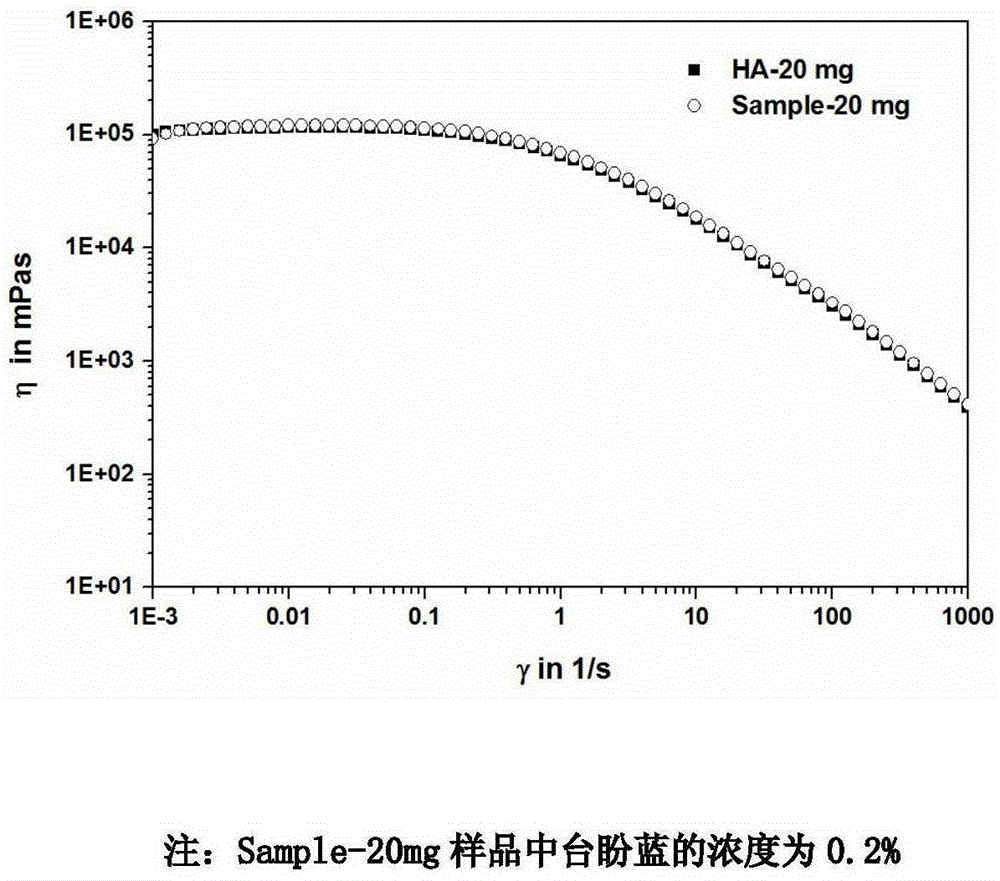
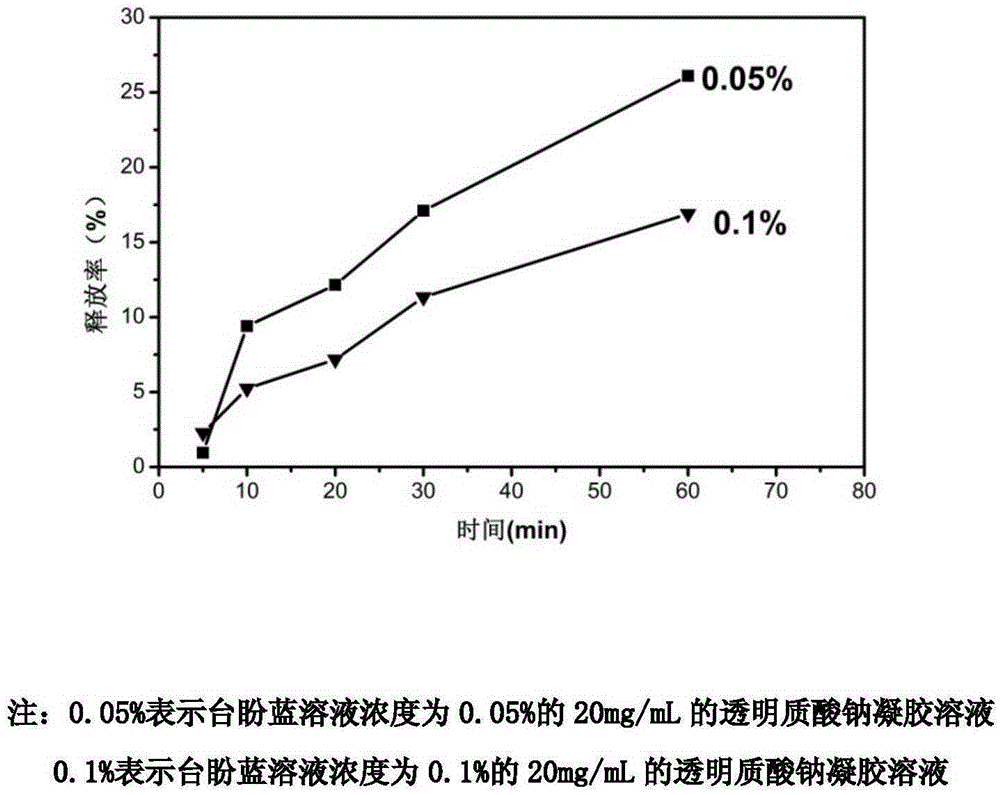
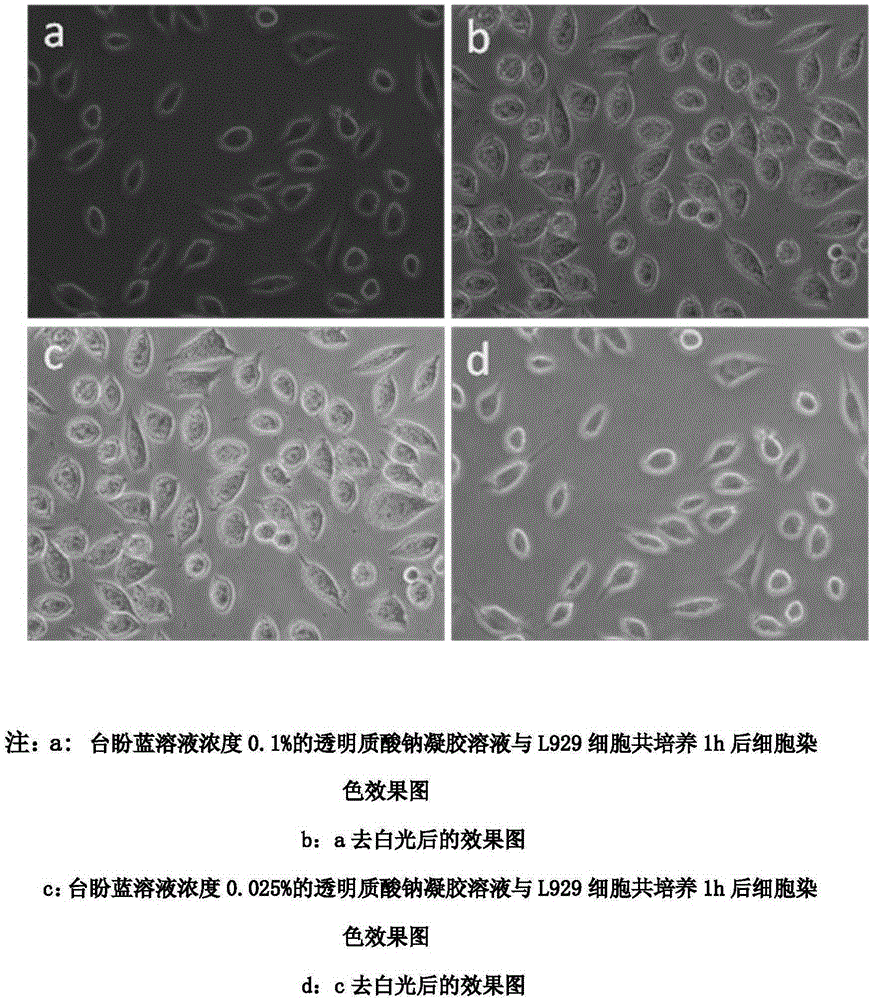
InactiveCN105903088AImprove cleanlinessIncrease success rateSurgeryHigh pressureBiological materials
The invention relates to the technical field of medical biomaterials. Cataract treatment becomes the ophthalmic clinical attention focus and a main treatment means comprises combination of lens extraction and artificial lens implantation. At present, the main clinical problem comprises intraocular pressure increasing caused by a low anterior lens capsule tear success rate and residual of a viscoelastic agent in the eye. The invention provides the preparation method of the ophthalmological viscoelastic agent with the selective anterior capsule staining function. The preparation method comprises adding a Trypan blue staining agent with a concentration of 0.002-0.6% into a material and carrying out high temperature and high pressure sterilization. The use of the staining agent does not change viscoelastic agent gel properties and an in-vitro release rate in 60min is about 15% so that the staining agent can be continuously and slowly released to an operation part so that selective staining is realized and damage to tissue cells is prevented. Based on the characteristic, the viscoelastic agent can be effectively removed through staining agent instruction after an operation.
Owner:SHANGHAI QISHENG BIOLOGICAL PREPARATION CO LTD
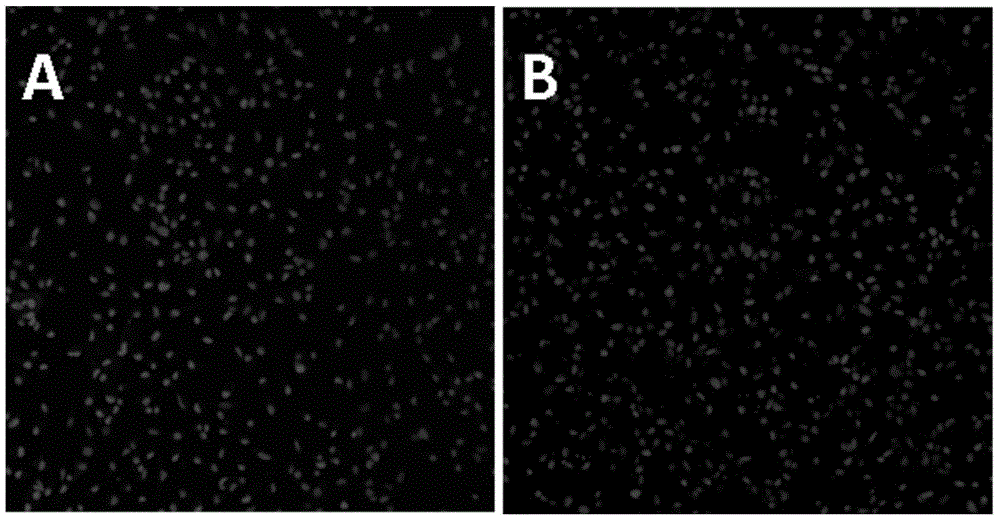
A separation culture method for lens epithelium stem cells
ActiveCN104789521AHigh activitySimple methodArtificially induced pluripotent cellsNon-embryonic pluripotent stem cellsCuticleCulture fluid
A separation culture method for lens epithelium stem cells is disclosed. The method includes following steps: (1) cutting a lens anterior capsule into small pieces, digesting, and paving the obtained cells to a culture flask coated with a medium, a culture plate coated with a medium or a culture dish coated with a medium; and (2) adding an MEM culture solution containing 15-25% of FBS, 50-150 mug / L of a human epidermal growth factor (FGF), 5-10 mg / L of insulin, 5-10 mg / L of hydrocortisone, 5-10 mg / L of a vibrio cholera toxin, 0.01-0.05 mg / L of 3,3',5-iodo-L-para-hydroxyphenylalanine, 1-5 mg / L of adenine, 5-10 mg / L of glycine, 6-12 mg / L of alanine, 10-16 mg / L of asparagines, 10-16 mg / L of aspartic acid, 12-18 mg / L of glutamic acid, 8-15 mg / L of proline and 7-14 mg / L of serine, putting into an incubator having a temperature of 37 DEG C, culturing for 2-3 days to form clone, and forming lens stem cells with monolayer activity after 10-14 days. A separation culture solution for the lens epithelium stem cells is also disclosed. The method can separate and obtain the primary lens epithelium stem cells. The stem cells are high in activity. The method is simple, convenient, easy in operation and good in application prospect.
Owner:GUANGZHOU KANGRUI BIOLOGICAL PHARMA TECH CO LTD
Capsule opening shaping instrument
The invention provides an instrument which is applied to cataract surgery and used for performing ostomy on an anterior capsule conveniently and safely. A cutter is a closed circular ring which is made of plastic and good in toughness; tooth-shaped projections are distributed at the bottom end of the circular ring in an equally spaced mode; a supporting ring is a hollow circular ring which is made of plastic and good in toughness; the supporting ring can accommodate the cutter and match with the cutter; the cutter can rotate freely in the supporting ring; the bottom end of the supporting ring is provided with an open area and the width of the open area is about 1.5 mm; the tooth-shaped projections which are arranged at the bottom end of the cutter in an annular shape are located in the open area; the tail end of the supporting ring is connected with a pushing rod through a connecting rod; a user can push the connecting rod into a sleeve from a handle container by pushing the pushing rod and meanwhile the supporting ring can be pushed out of the sleeve through the connecting rod; two holes are formed above the sleeve and the handle container respectively; a pulling rope penetrates the holes; one end of the pulling rope is fixed on one of the tooth-shaped projections of the cutter, the other end of the pulling rope is arranged outside the handle container, and the tail end of the pulling rope is connected with a rope drop; the diameter of the rope drop is larger than that of the hole which is formed above the handle container. The supporting ring is pushed out, the pulling rope is pulled, the cutter cuts through the anterior capsule, and accordingly the ostomy shaping is achieved.
Owner:尹澜
Accommodative intraocular lens
ActiveUS20200276011A1Reduce diameter of lensReduce the overall diameterIntraocular lensEngineeringMechanical engineering
An accommodative intraocular lens capable of effectively exerting a focus adjustment function includes an optical portion and a plurality of support portions arranged around the optical portion. The support portion includes an anterior support portion and a posterior support portion, and the anterior support portion presses an anterior capsule and the posterior support portion presses a posterior capsule by the elastic force of the support portion. When the lens capsule is in a distance vision state or in a near vision state, as the pressing force of the anterior capsule against the anterior support portion increases or decreases, the anterior support portion deflects backward or returns forward while maintaining the radial position of the base end portion, so that the tip end portion of the anterior support portion moves backward or forward greatly while maintaining the radial position, and the optical portion moves backward or forward accordingly.
Owner:MIRAI EYE INC
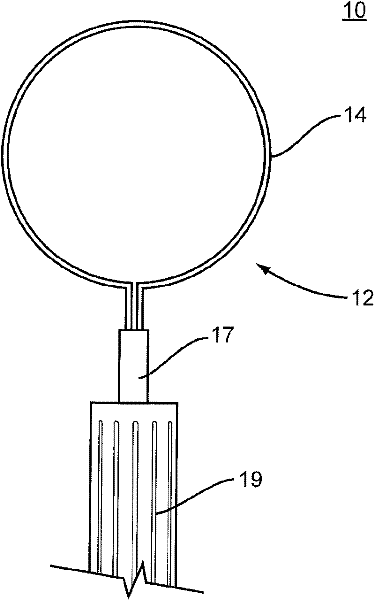
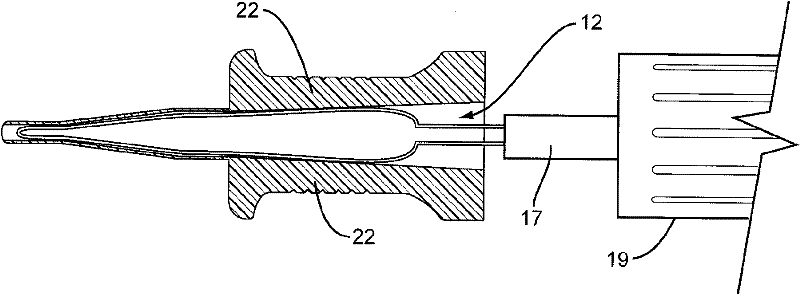
Capsularhexis device with flexible heating element
ActiveCN102176887AEye surgerySurgical instruments for heatingWire rodElectrical resistance and conductance
A capsularhexis device (10) comprises a resistive-heating element (12) formed from an electrically resistive, superelastic wire (14) formed into a loop. A bare surface of this loop is applied to the anterior capsule and electrically heated to define a rhexis boundary, such that a smooth circular or oval capsule portion may be removed. In some embodiments, the superelastic wire is formed to include a loop, so that first and second ends of the wire are adjacent to one another and extend away from the loop to form a lead section. An electrically insulating material (17) may completely or partly surround the first and second ends of the wire so that the first and second ends of the superelastic wire are electrically separated. A handle (19) engages at least a portion of the lead section so that the loop-shaped heating element may be moved in and out of the anterior chamber.
Owner:ALCON INC
Accommodative intraocular lens that ejects post capsular opacification and self-centers
ActiveUS20180104047A1Reduces potential warpageGood removal effectIntraocular lensPosterior capsule opacificationAnterior surface
Described is an accommodating intraocular lens with a bi-convex, bi-aspheric, smooth surfaced optic held inside an anterior annulus via tabs. A second larger diameter annulus is positioned posteriorly and connects via a sloped surface to where the annuluses are at a maximum separation when viewing NEAR objects and minimum separation in the FAR position. The sloped surface is cut into ribbons, tabs and / or other annuluses without pushing the surfaces into the capsule when implanted; therefore, only the anterior and posterior annuluses have a force component against the capsule. The proximal edge of the anterior annulus is anterior to the apex of the anterior surface of the optic. The anterior capsule resting on the annulus leaves space for hydration of the capsule and reduces potential warpage of the optic. The annulus edge is designed to scrape posterior capsular opacification from the capsule.
Owner:CALLAHAN WAYNE B
Capsulotomy devices and methods
An Anterior Capsule Remover (“ACR”) device allows an ophthalmologist to safely and efficiently remove an anterior portion of the lens capsule at the beginning of cataract surgery. The device allows the surgeon to safely, efficiently and consistently create a circular central opening of a predetermined diameter in the top of the lens capsule. An exemplary anterior capsule remover apparatus includes a handle and a distal shaft that holds a blade or cutter. One or more o-rings attached to a thumbwheel and to the blade allow the surgeon to easily create a circular, central opening in the anterior capsule of predetermined diameter.
Owner:OJO