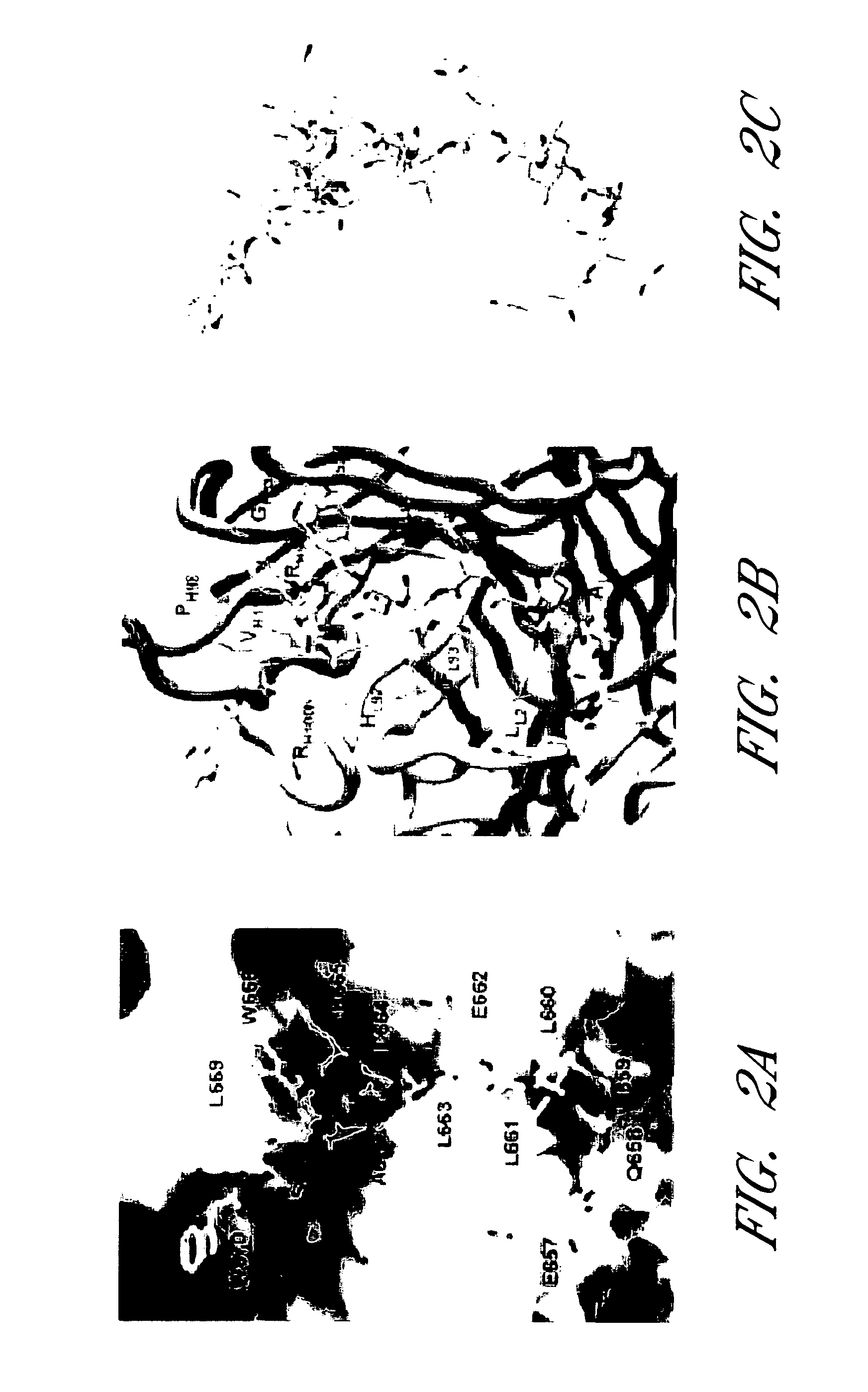
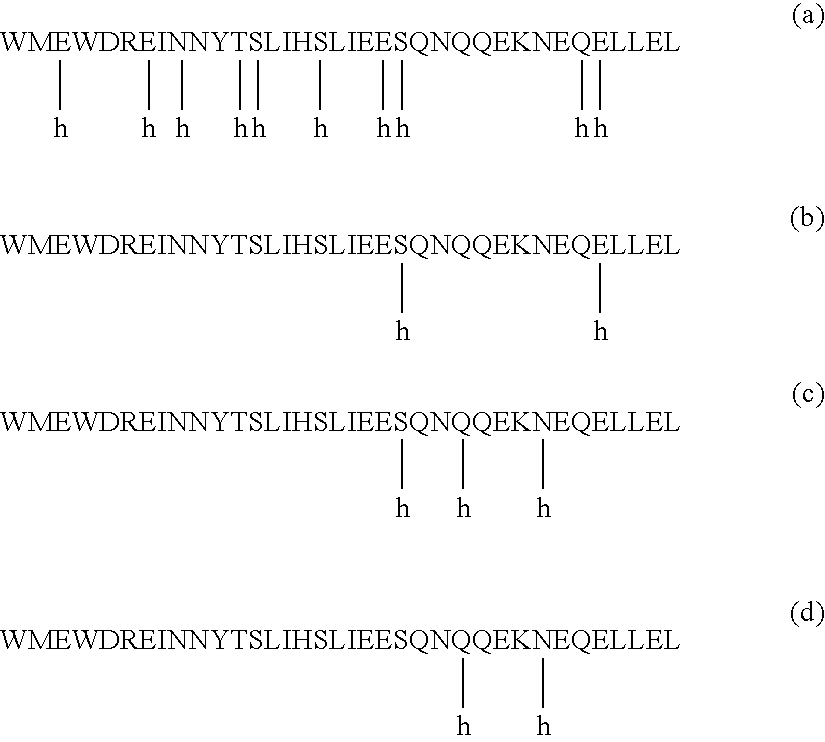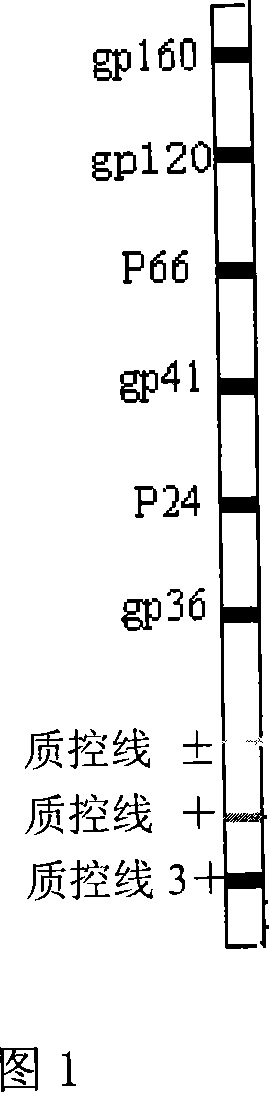Patents
Literature
165 results about "Gp41" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
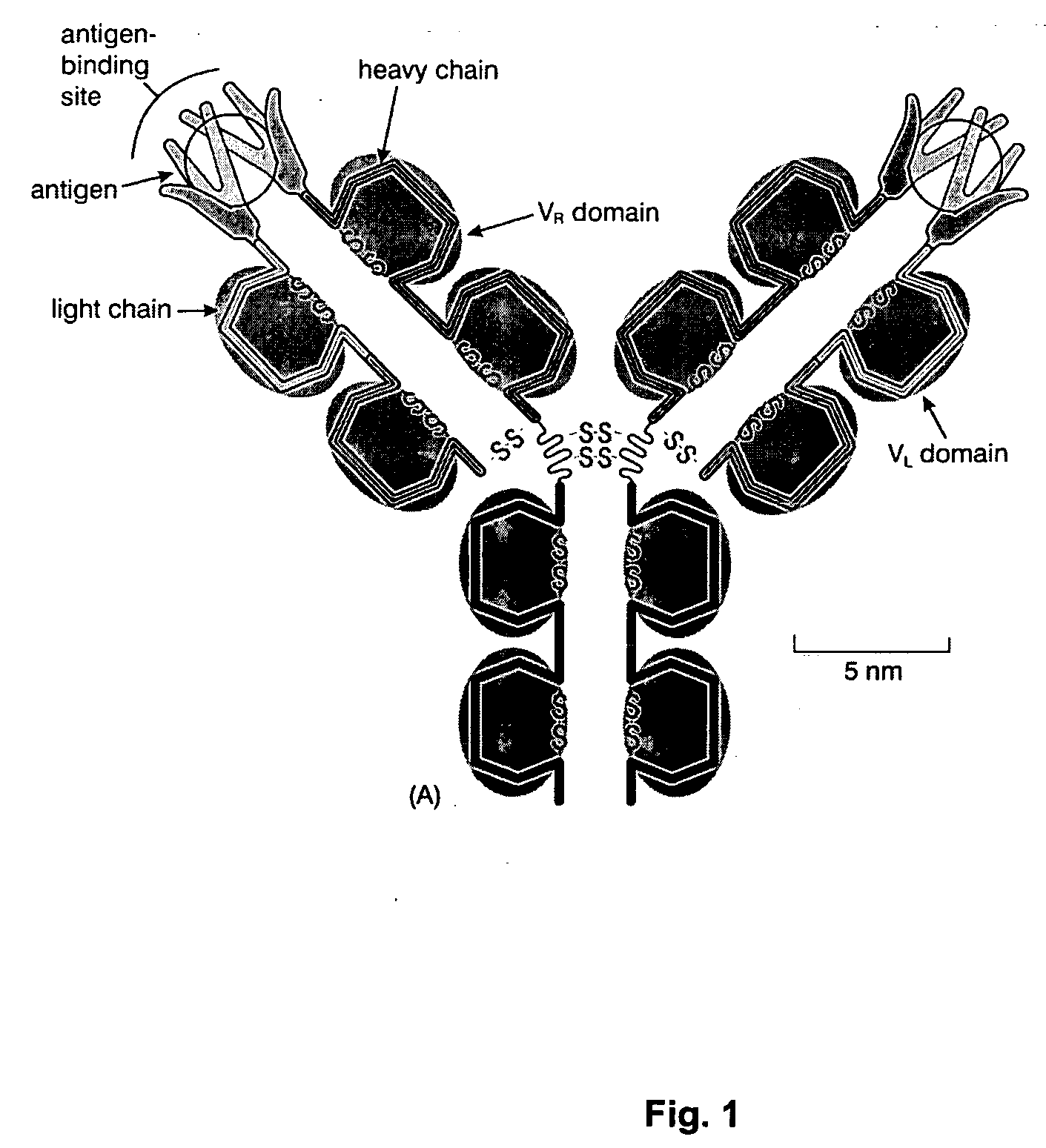
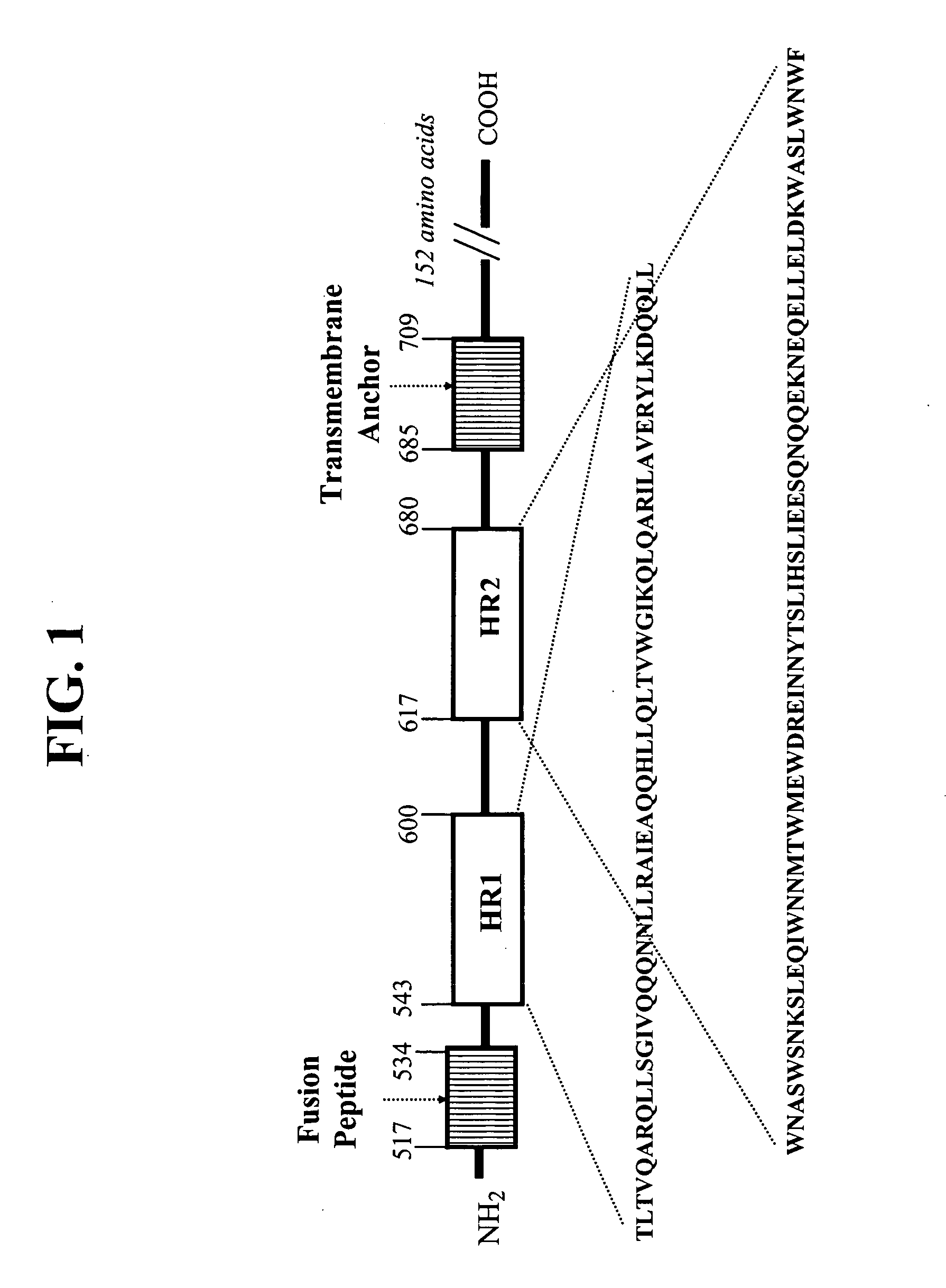
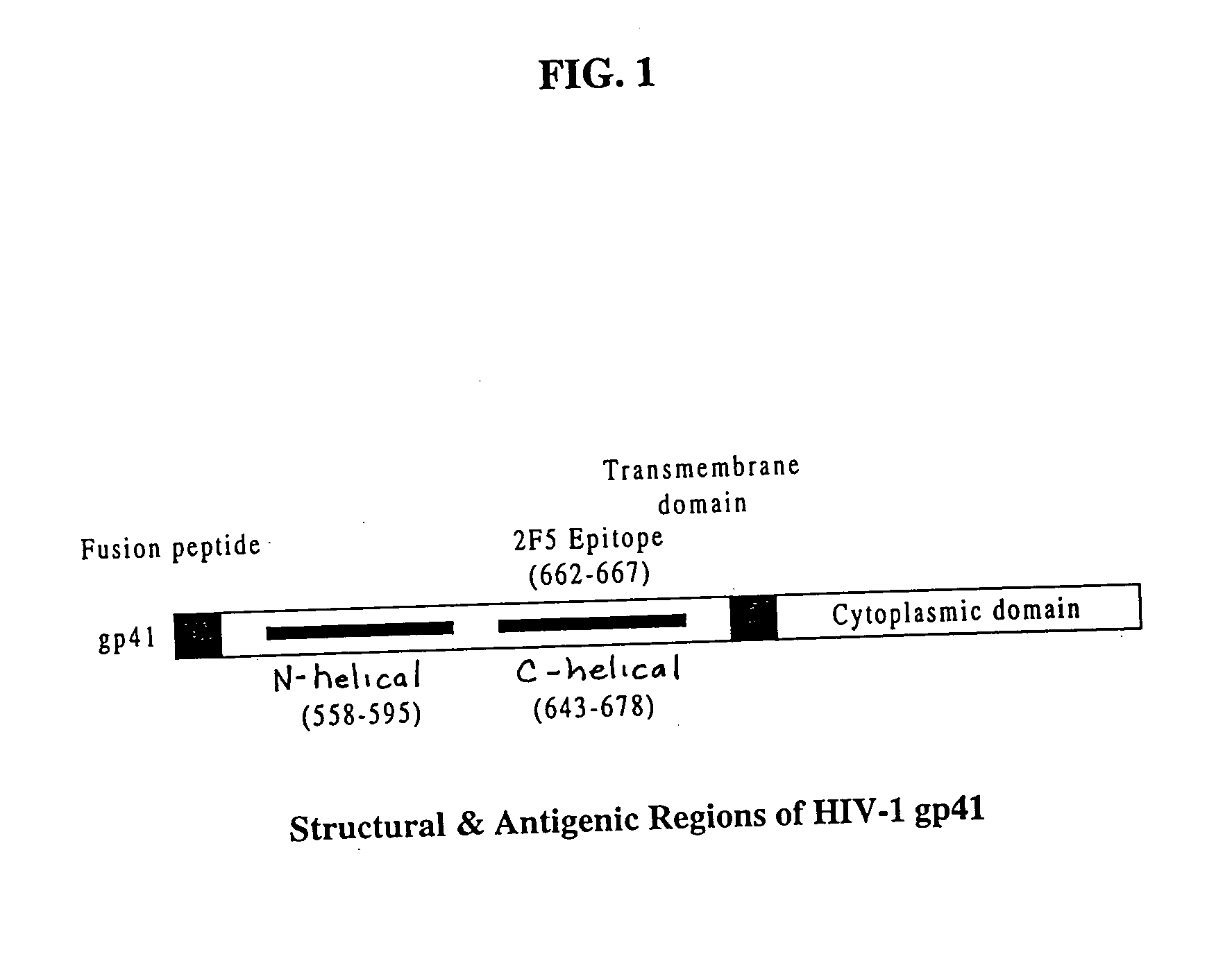
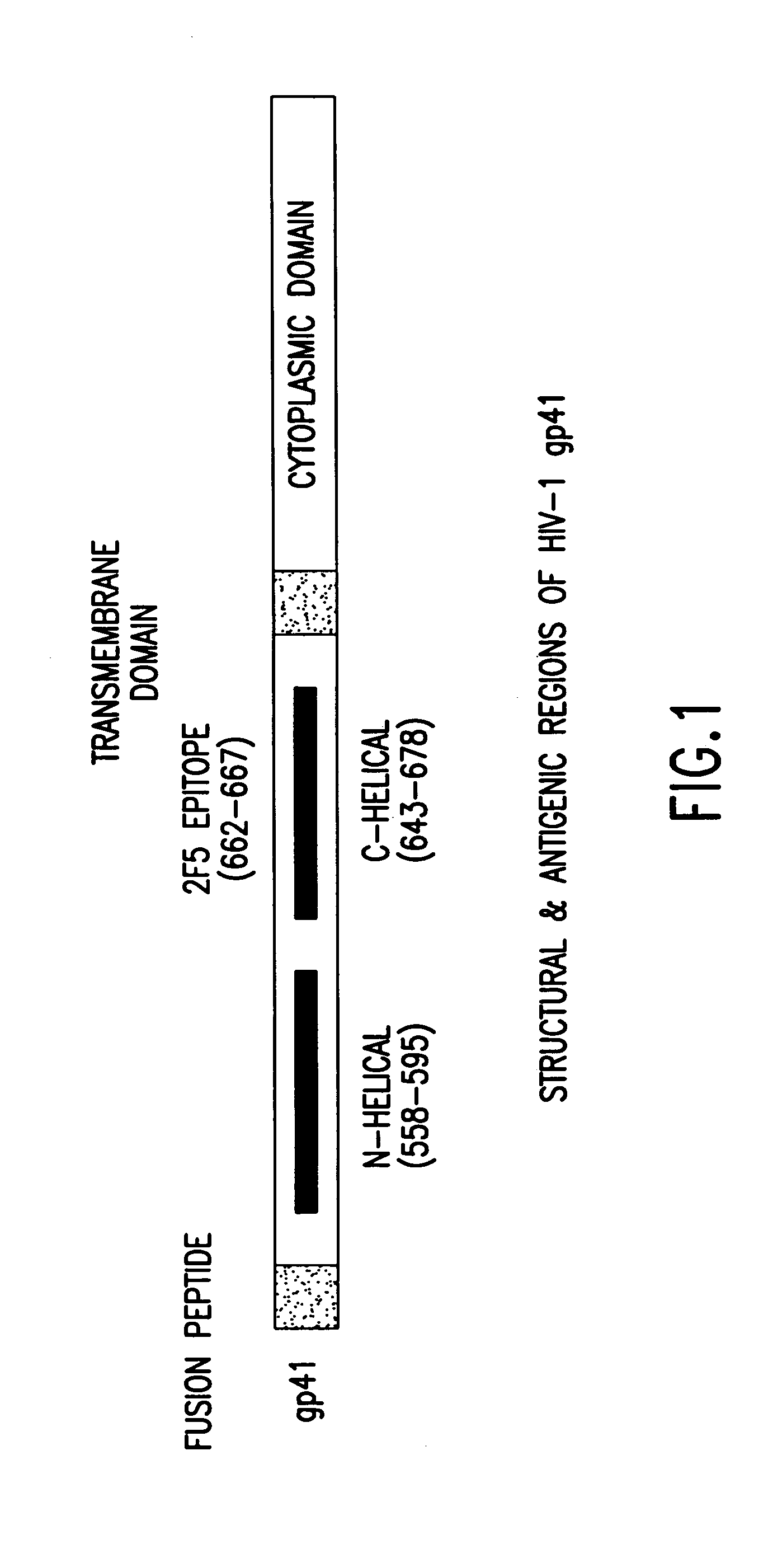
Gp41 also known as glycoprotein 41 is a subunit of the envelope protein complex of retroviruses, including human immunodeficiency virus (HIV). Gp41 is a transmembrane protein that contains several sites within its ectodomain that are required for infection of host cells. As a result of its importance in host cell infection, it has also received much attention as a potential target for HIV vaccines.
Measles virus peptides with antifusogenic and antiviral activities
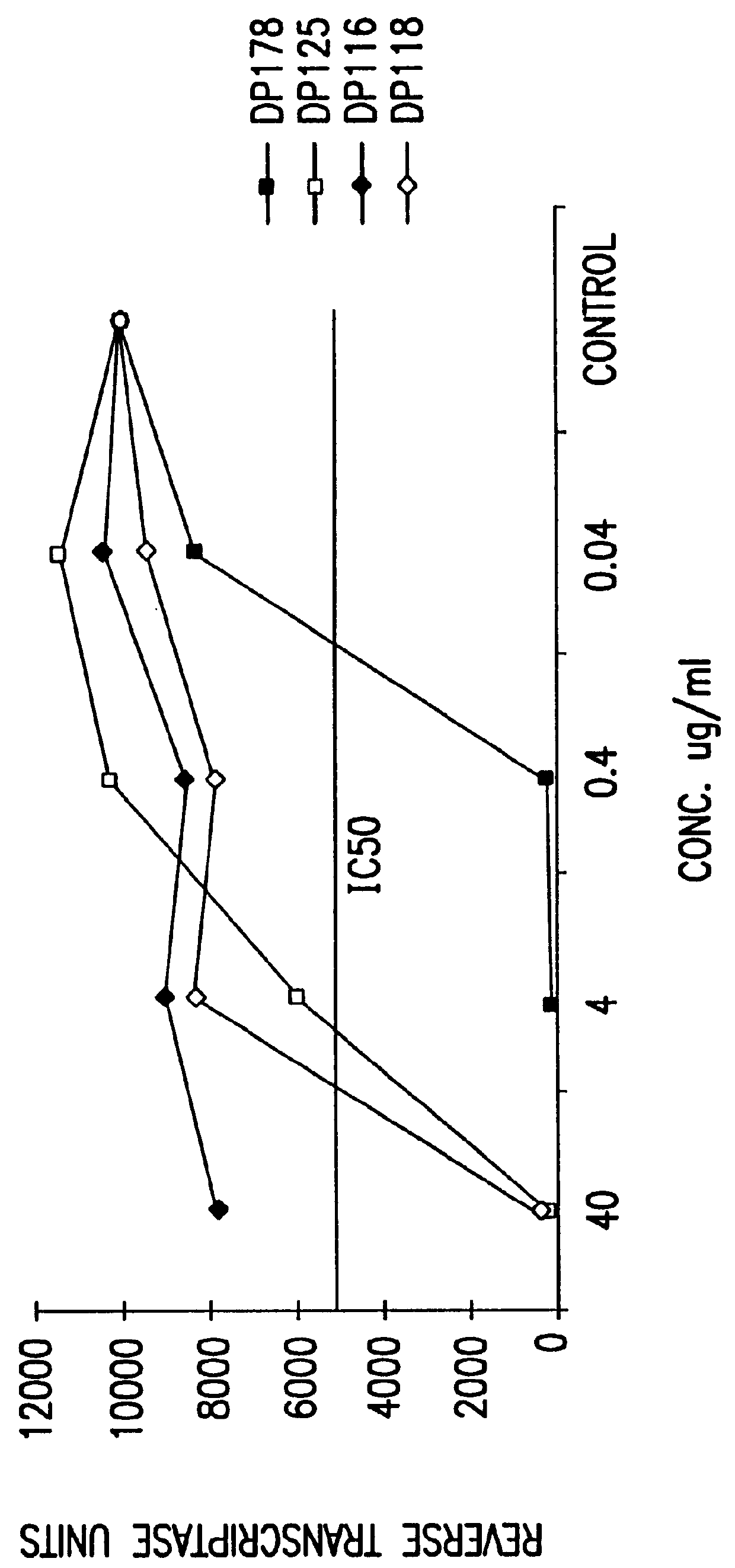
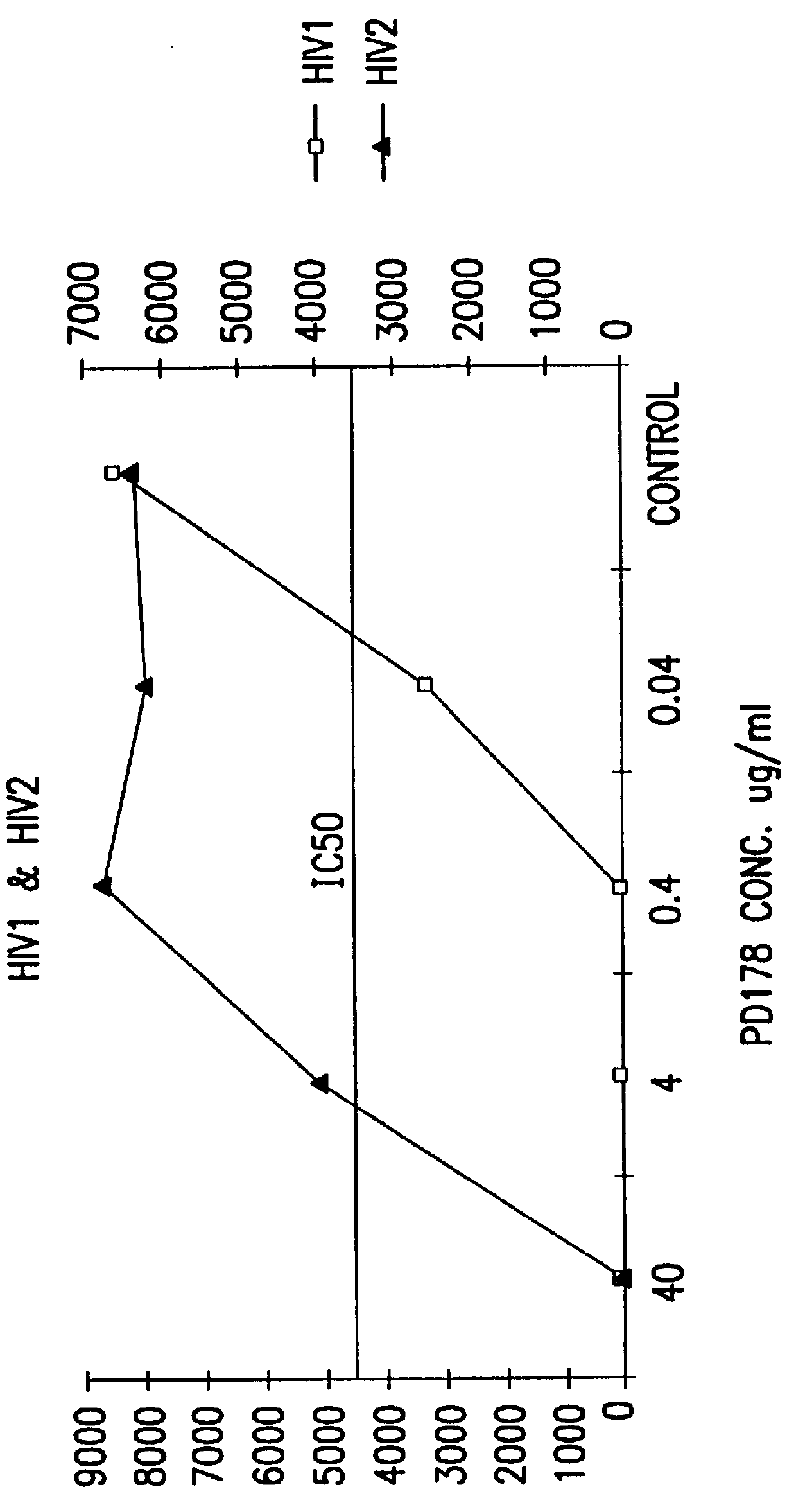
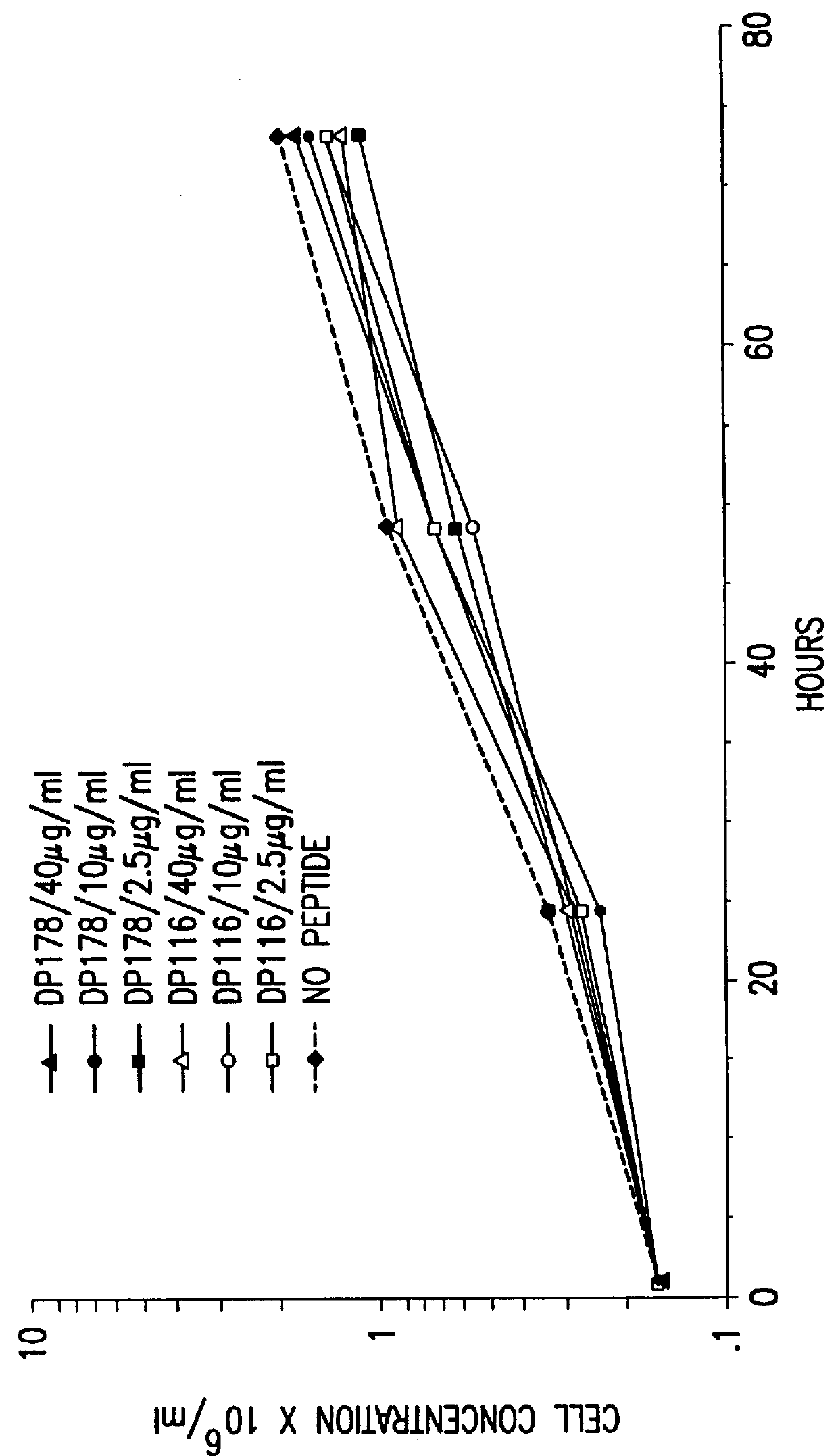
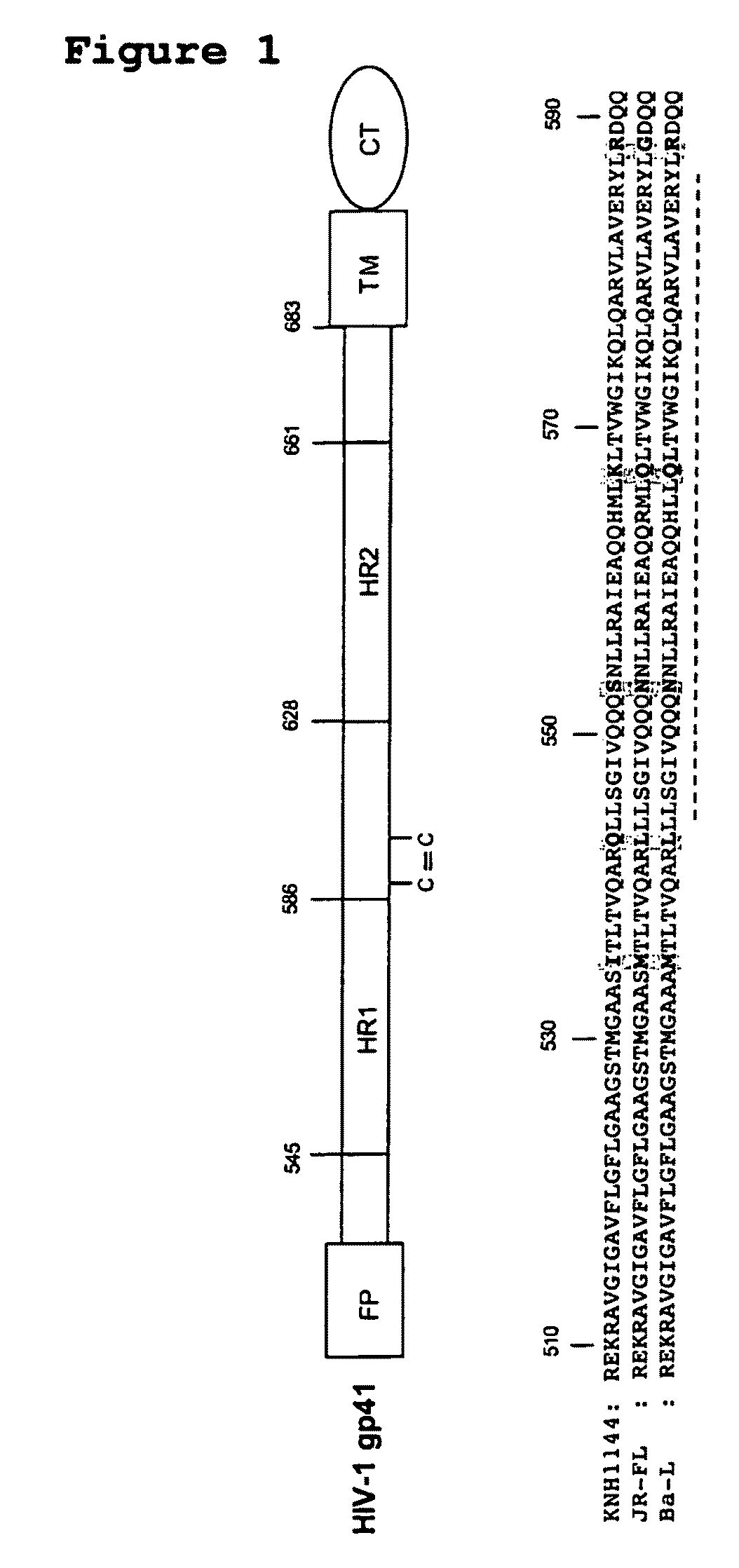
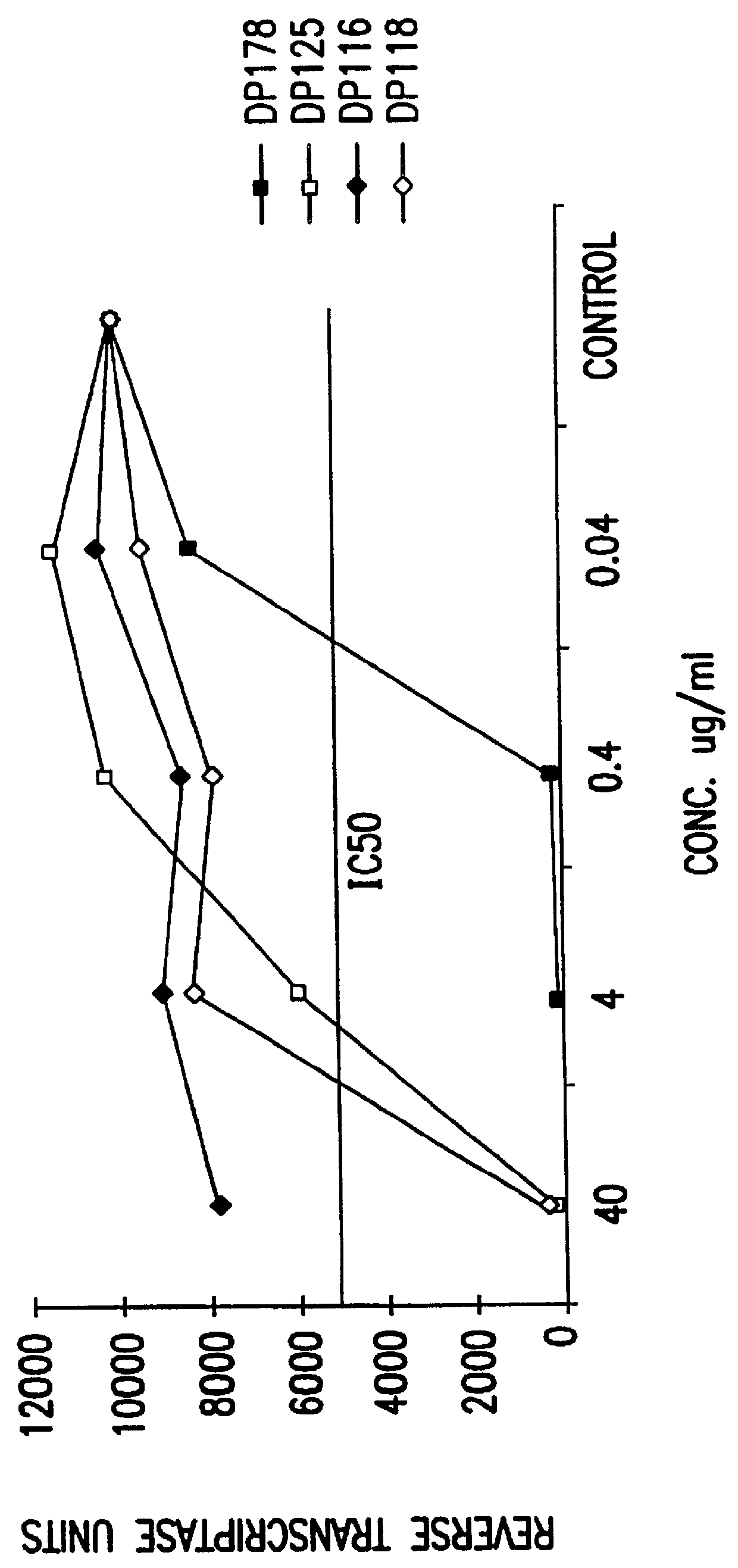
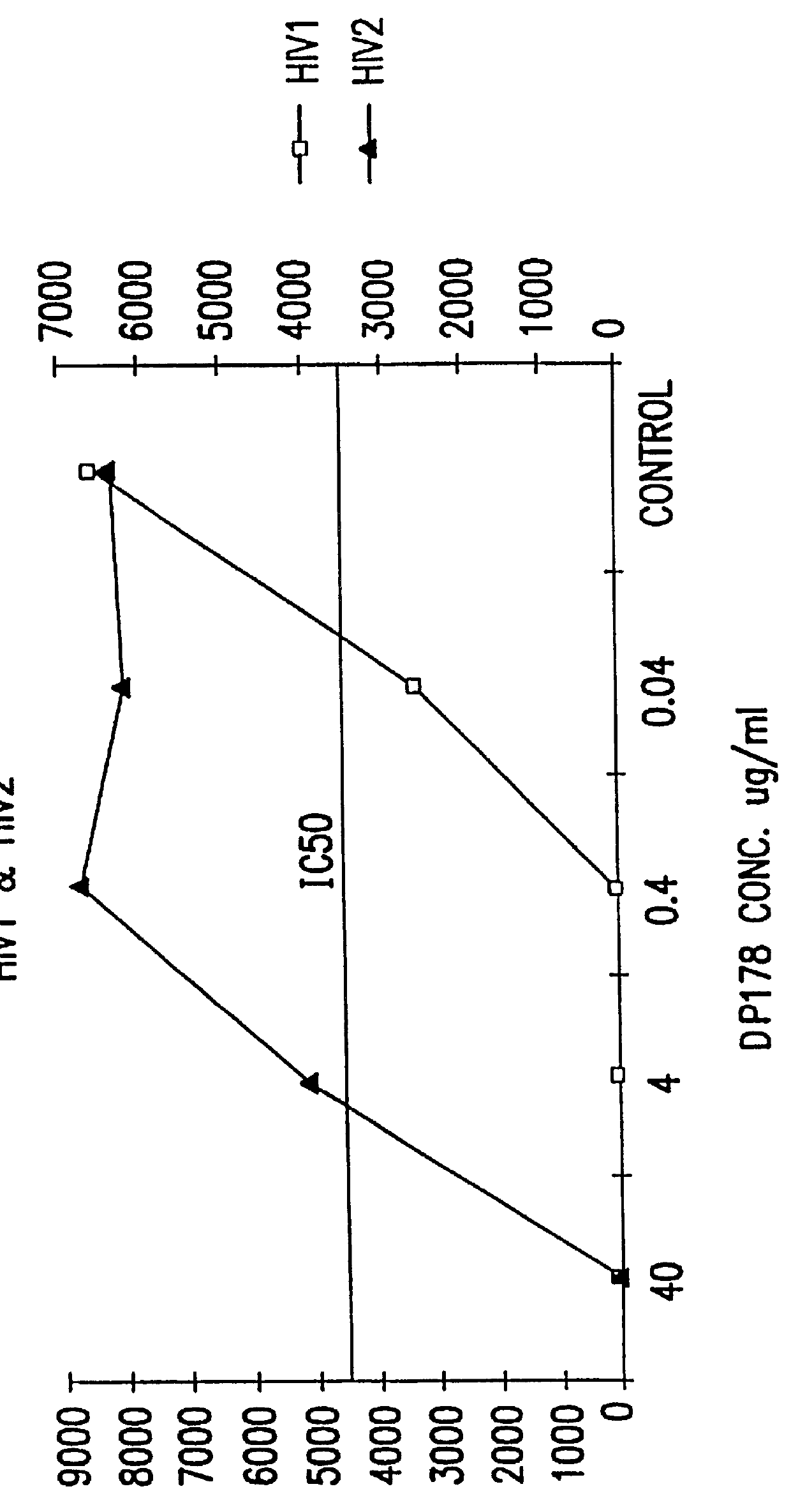
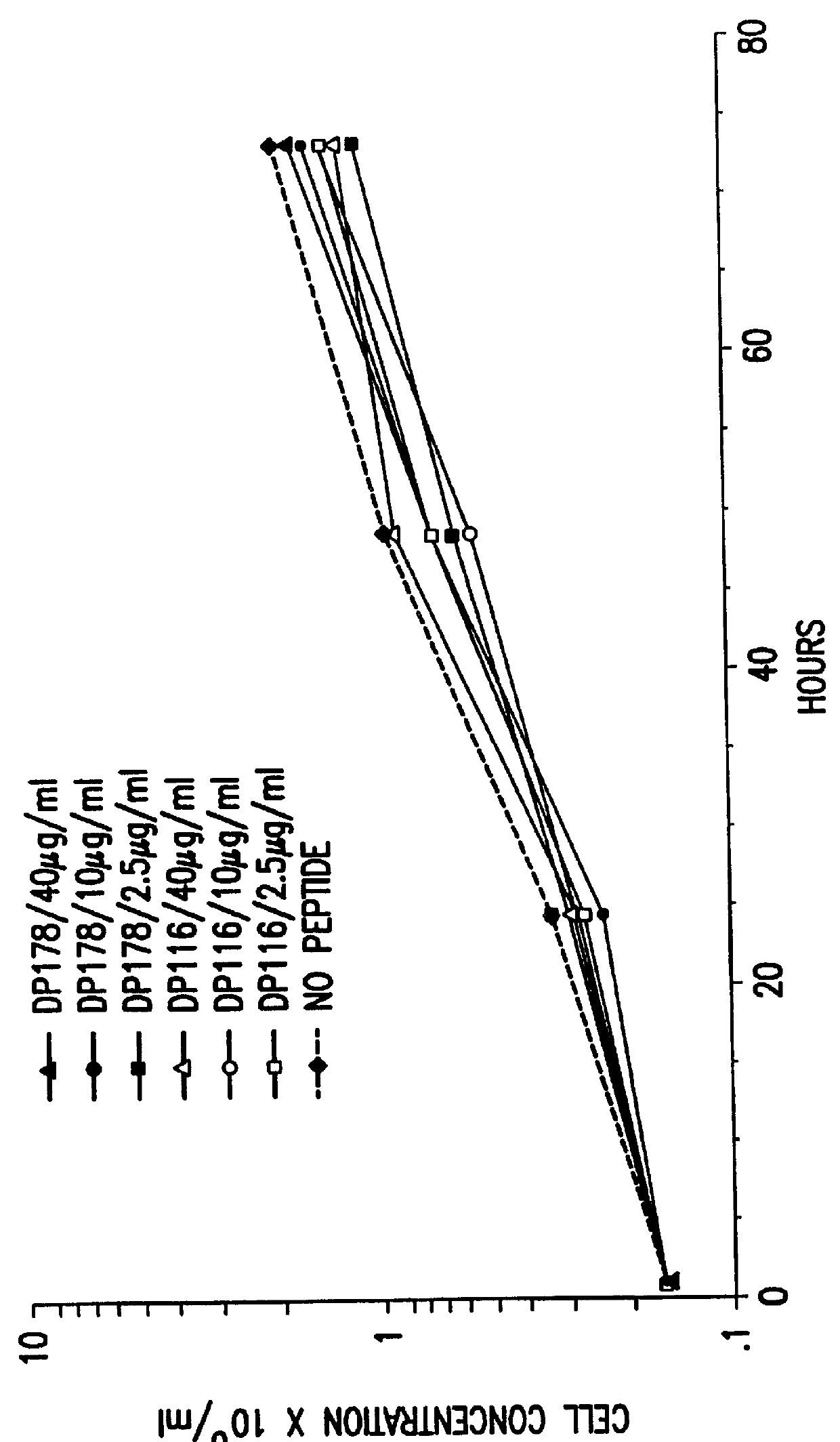
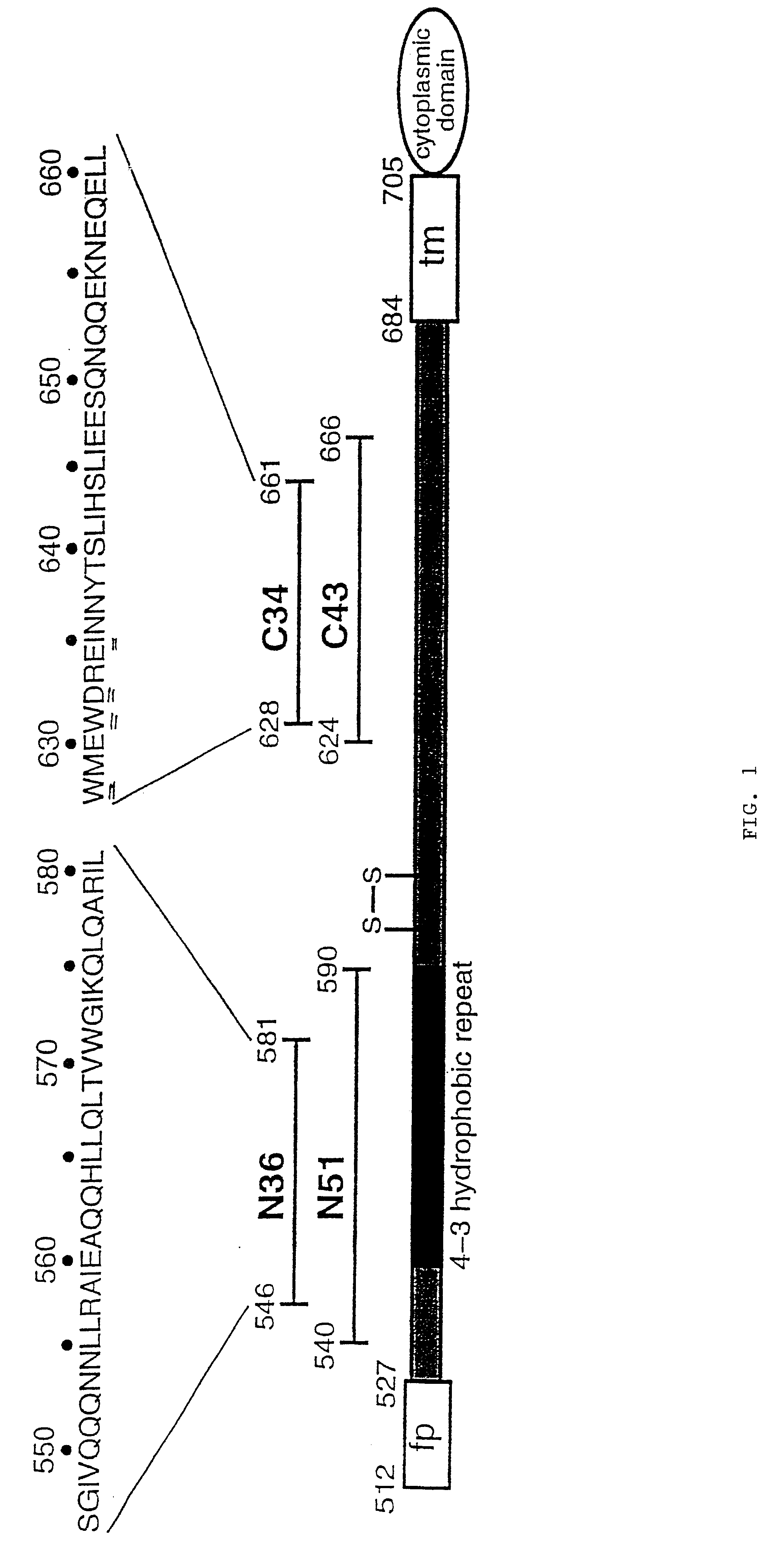
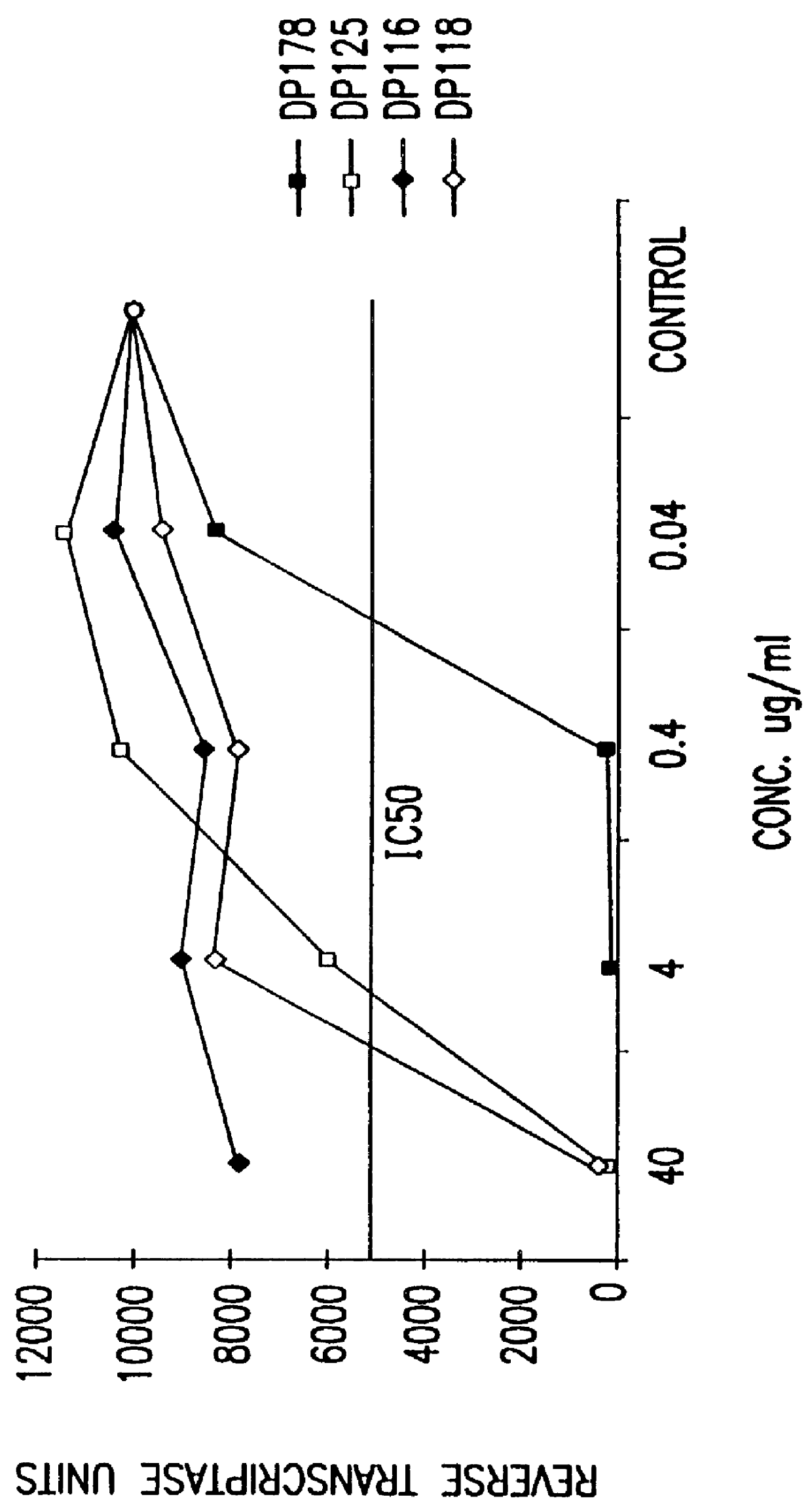
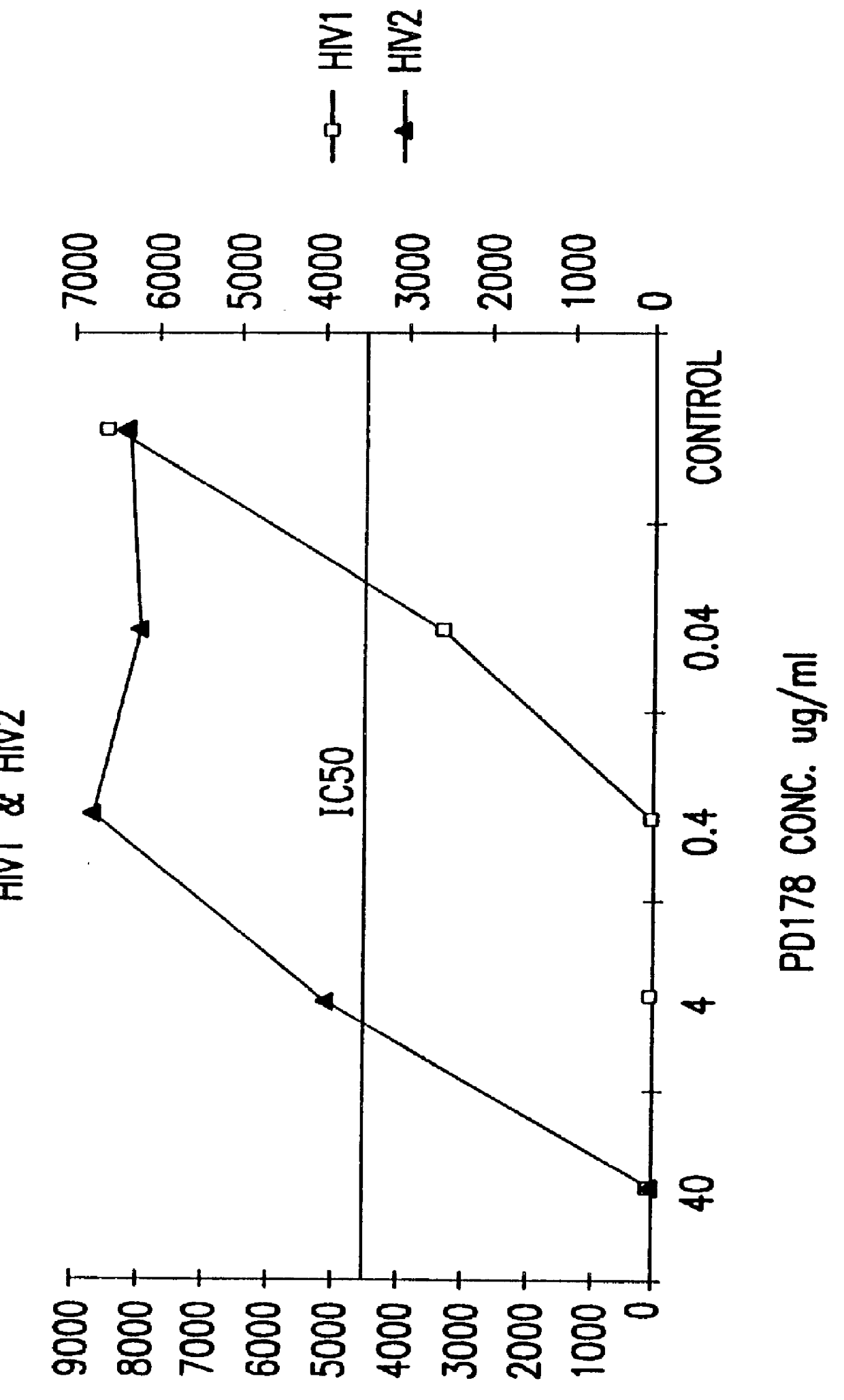
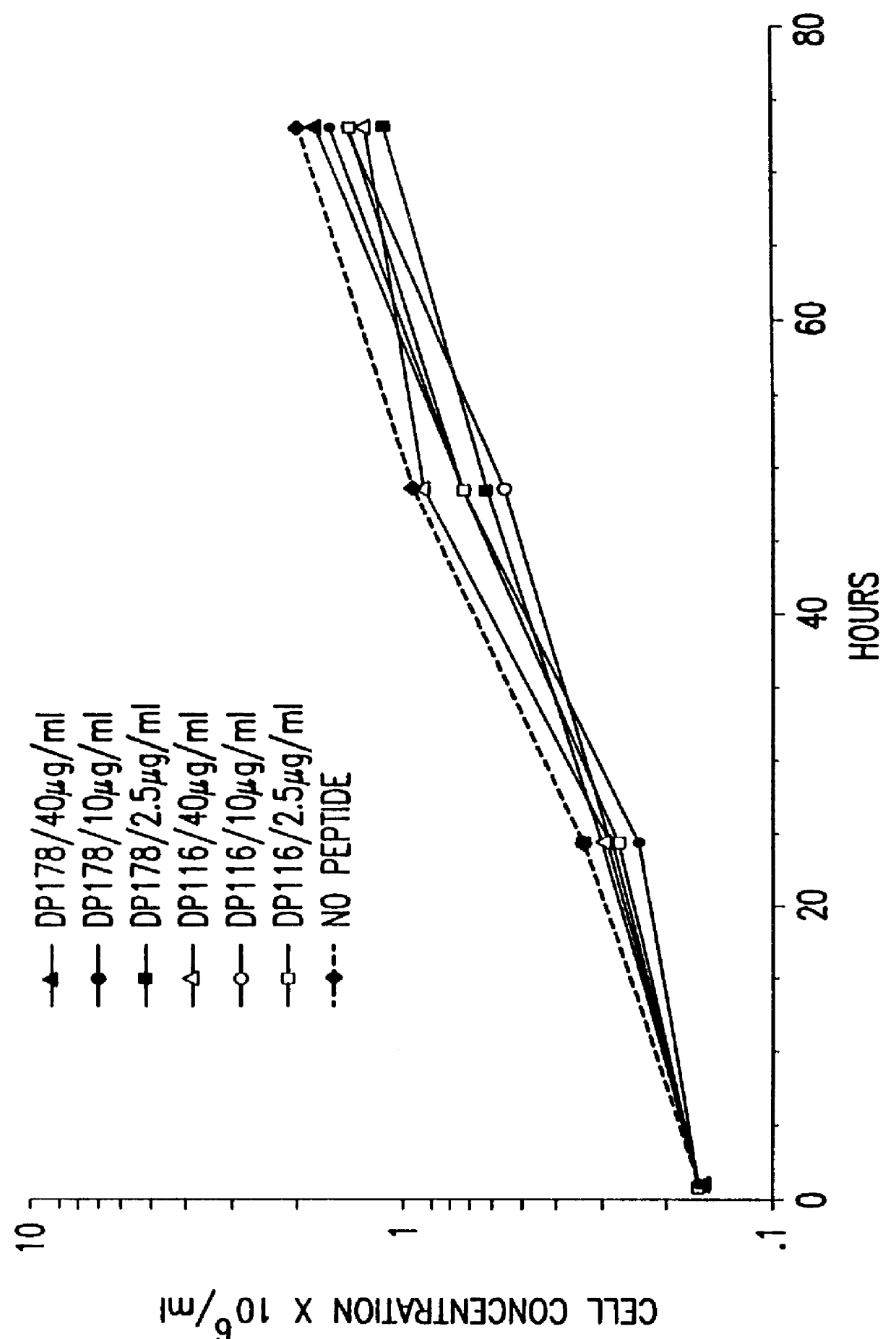
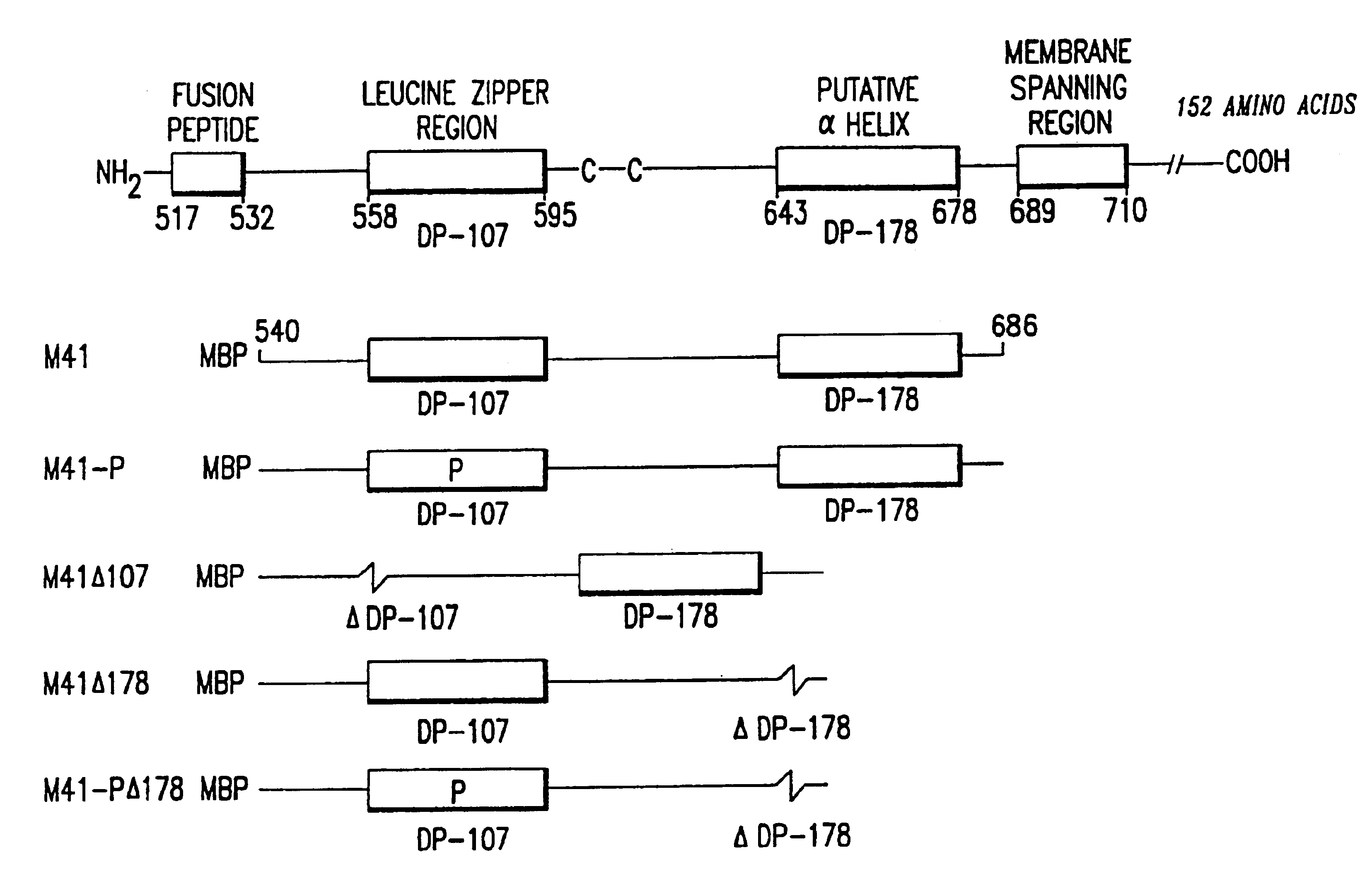
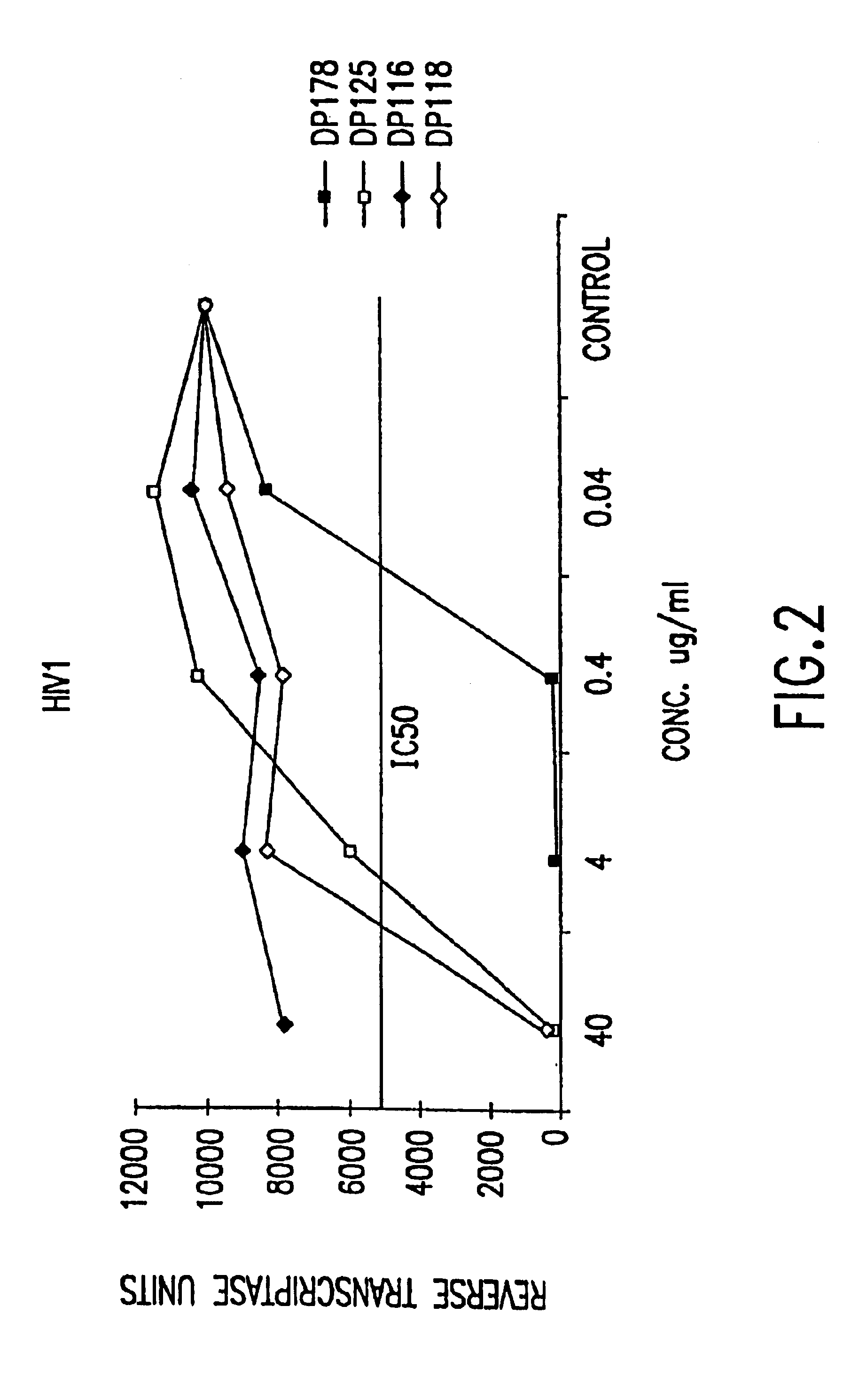
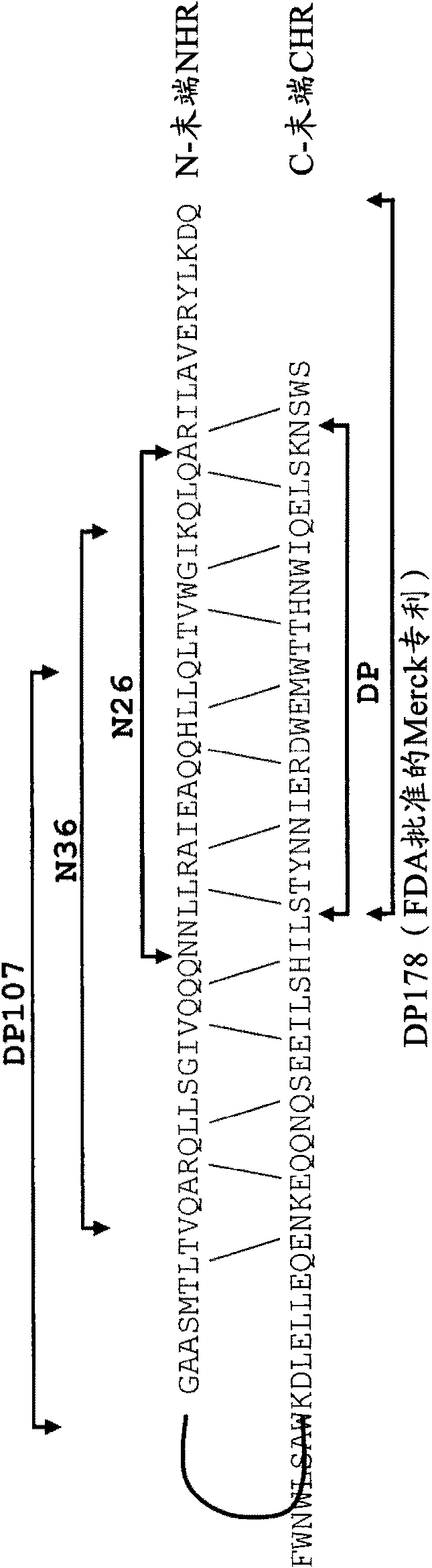
The present invention relates to peptides which exhibit potent anti-retroviral activity. The peptides of the invention comprise DP178 (SEQ ID:1) peptide corresponding to amino acids 638 to 673 of the HIV-1LAI gp41 protein, and fragments, analogs and homologs of DP178. The invention further relates to the uses of such peptides as inhibitory of human and non-human retroviral, especially HIV, transmission to uninfected cells.
Owner:TRIMERIS
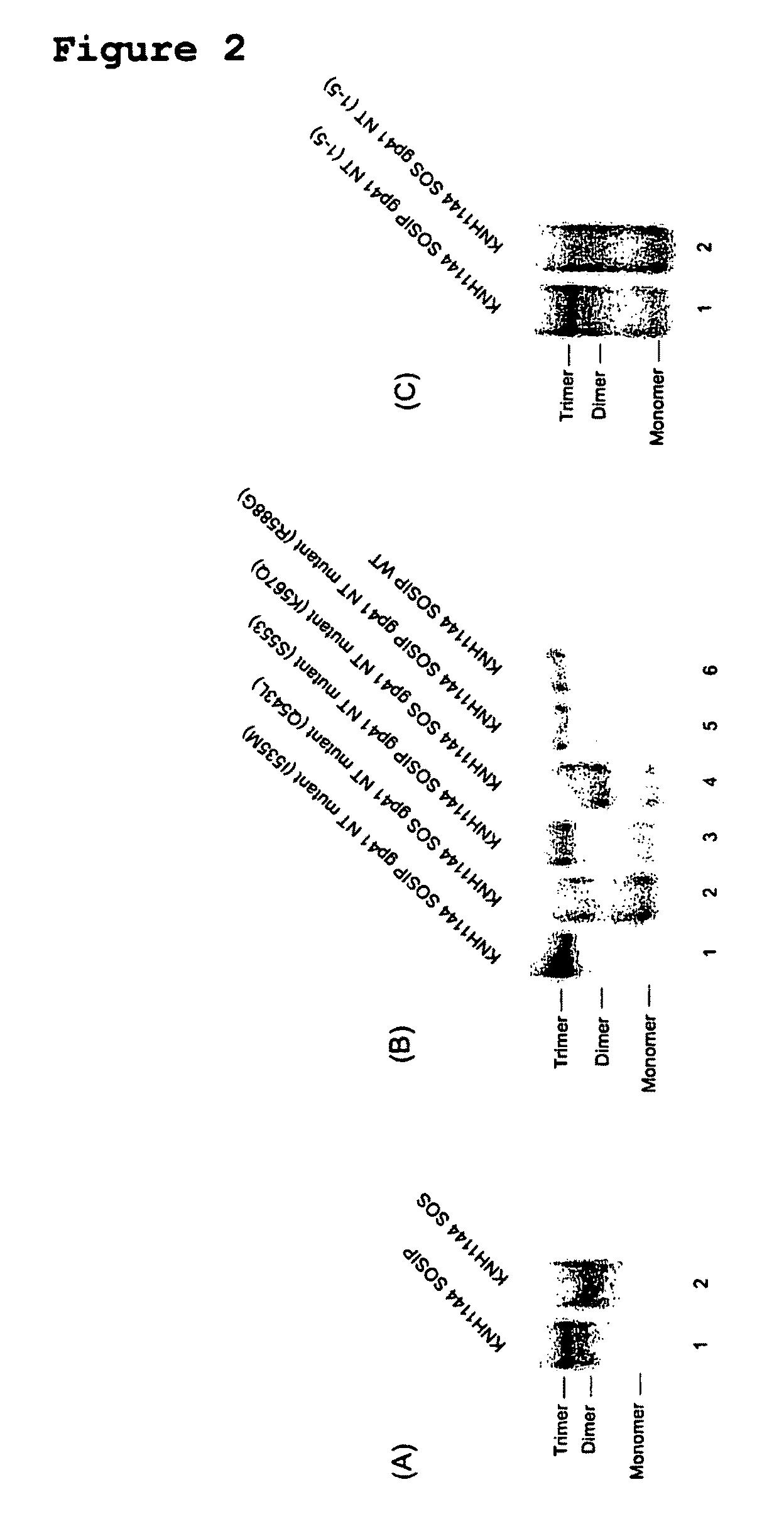
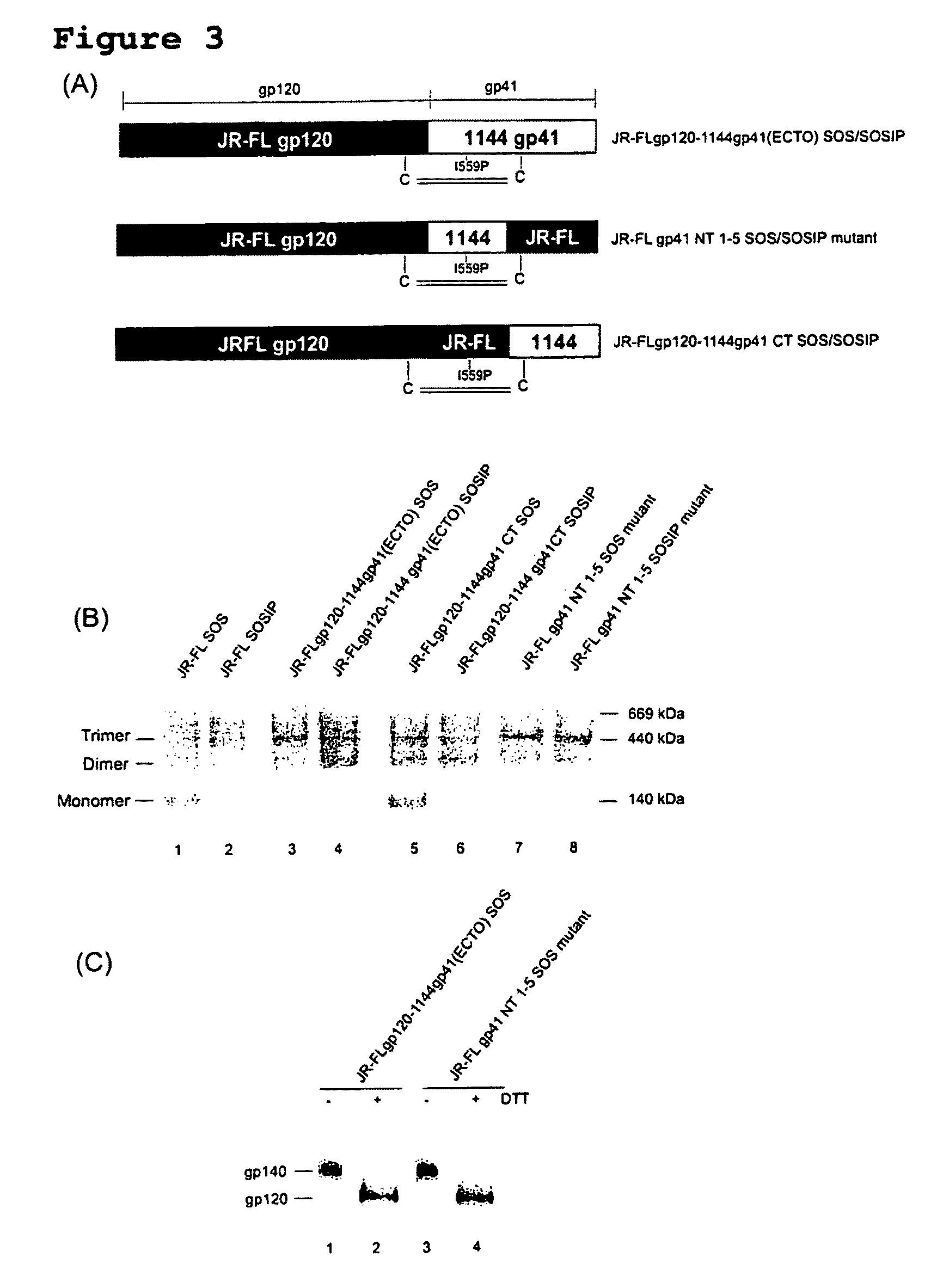
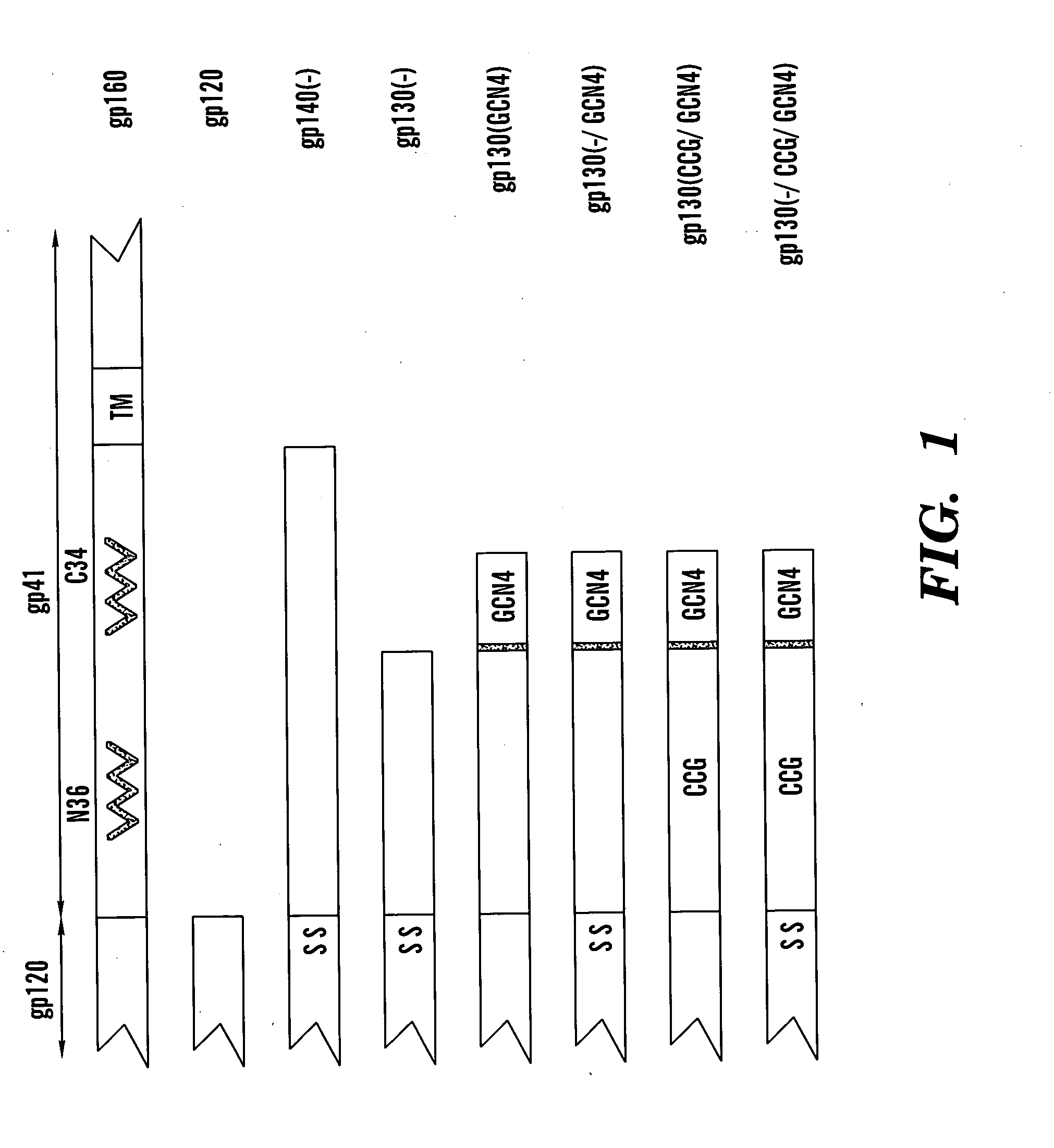
Soluble, stabilized, proteolytically cleaved, trimeric HIV-1 gp140 proteins comprising modifications in the N-terminus of the gp41 ectodomain
InactiveUS7939083B2Improve stabilityOverall antigenic structure of the trimer not adversely affectedViral antigen ingredientsVirus peptidesArginineGlutamine
Owner:CORNELL RES FOUNDATION INC +1
Stabilized soluble glycoprotein trimers
InactiveUS6911205B2Stabilize trimerSimplifies isolationPeptide/protein ingredientsAntibody mimetics/scaffoldsHiv envelopeGp41
Owner:THE TRUSTEES OF COLUMBIA UNIV IN THE CITY OF NEW YORK
Synthetic peptide inhibitors of HIV transmission
The present invention relates to peptides which exhibit potent anti-retroviral activity. The peptides of the invention comprise DP-178 (SEQ ID:1) ptide corresponding to amino acids 638 to 673 of the HIV-1LAI gp41 protein, and fragments, analogs and homologs of DP-178. The invention further relates to the uses of such peptides as inhibitory of human and non-human retroviral, especially HIV, transmission to uninfected cells.
Owner:DUKE UNIV
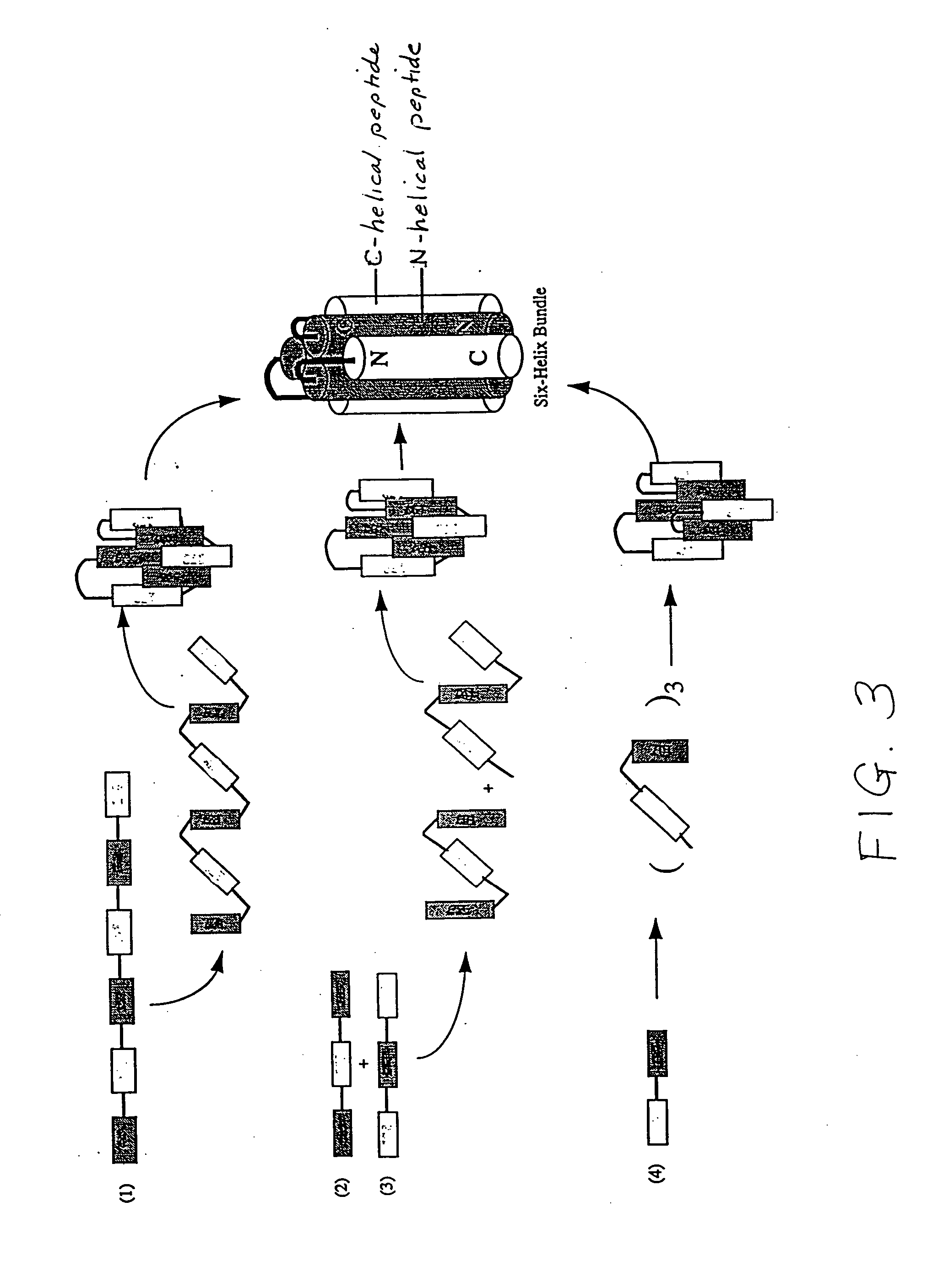
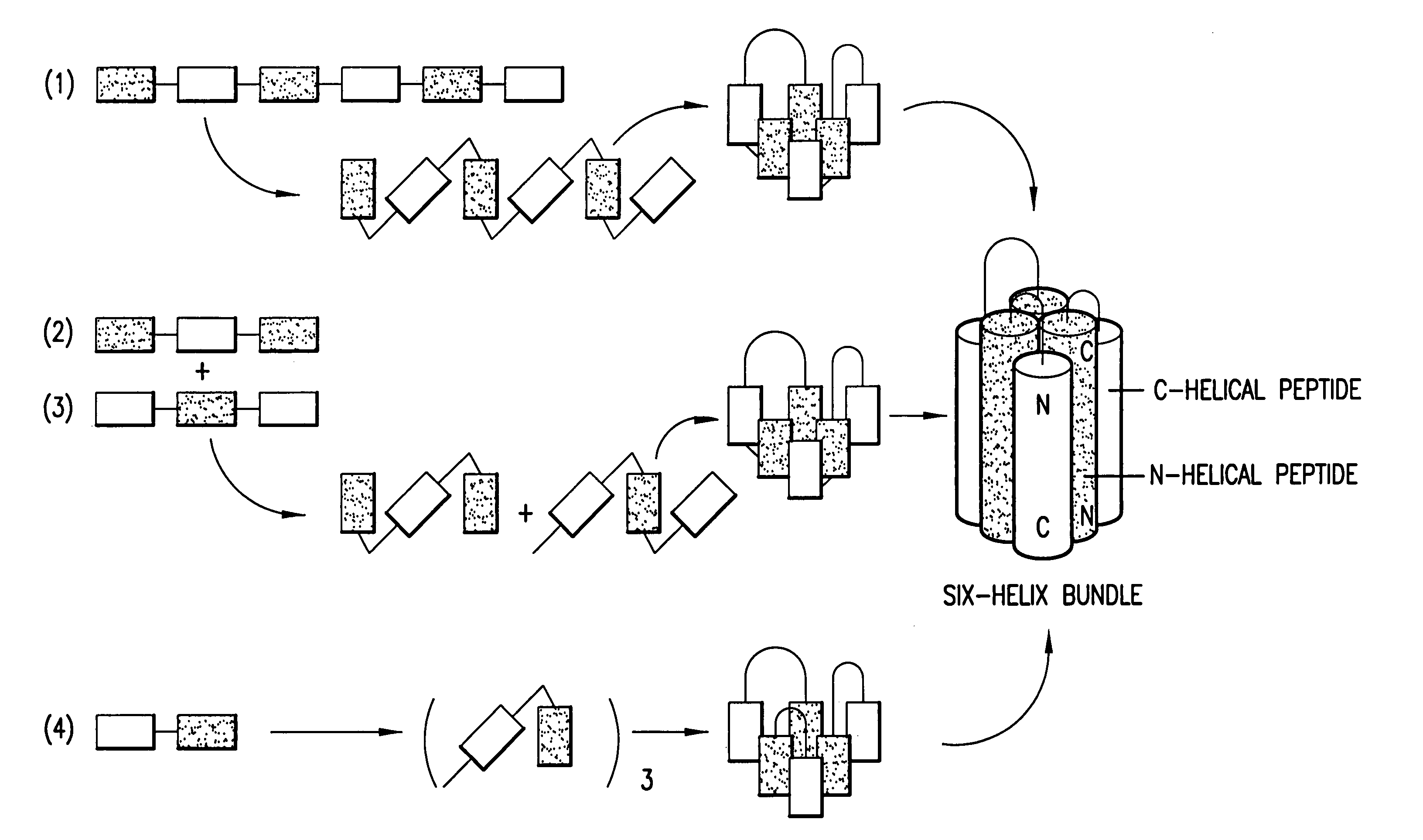
Core structure of gp41 from the HIV envelope glycoprotein
InactiveUS6506554B1Increase infectivityHigh activityPeptide/protein ingredientsMicrobiological testing/measurementHiv envelopeDesigning drug
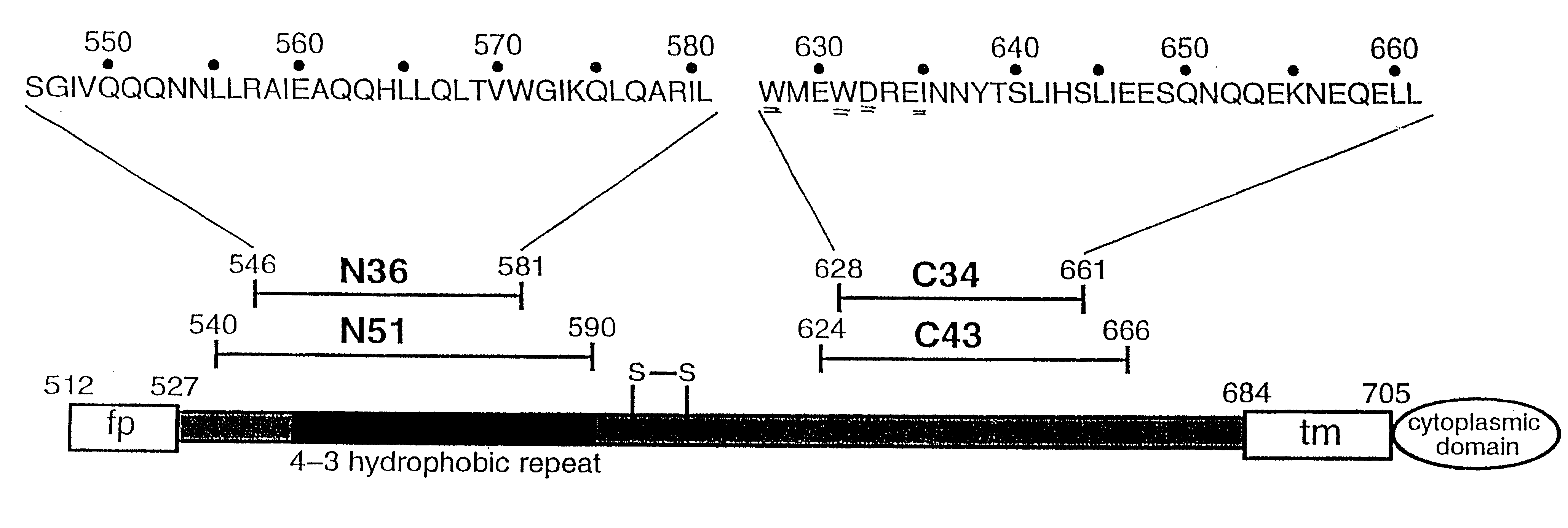
Described are the crystal structure of the alpha-helical domain of the gp41 component of HIV-1 envelope glycoprotein which represents the core of fusion-active gp41, methods of identifying and designing drugs which inhibit gp41 function and drugs which do so.
Owner:WHITEHEAD INST FOR BIOMEDICAL RES
Screening assays for compounds that inhibit membrane fusion-associated events
The present invention relates to peptides which exhibit potent anti-retroviral activity. The peptides of the invention comprise DP178 (SEQ ID:1) peptide corresponding to amino acids 638 to 673 of the HIV-1LAI gp41 protein, and fragments, analogs and homologs of DP178. The invention further relates to the uses of such peptides as inhibitory of human and non-human retroviral, especially HIV, transmission to uninfected cells.
Owner:TRIMERIS
Fusion protein construct and method for inducing HIV-specific serum IgG and secretory IgA antibodies in-vivo
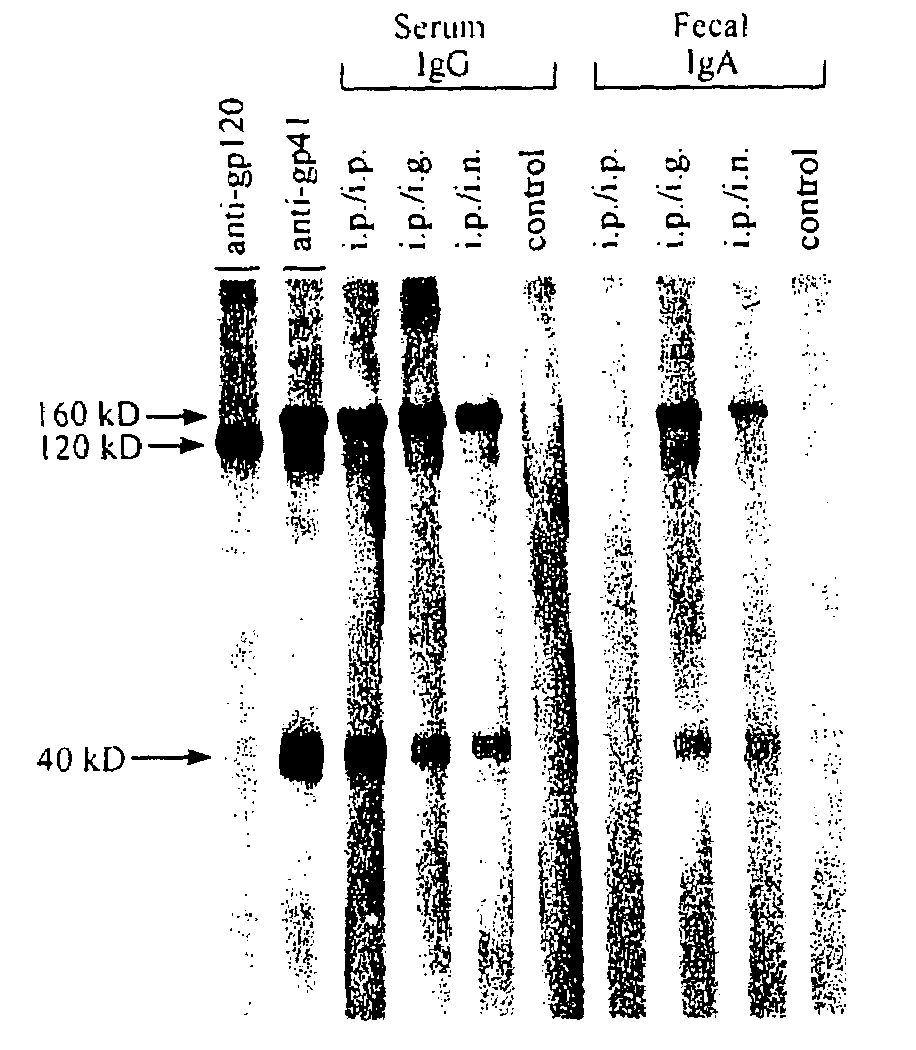
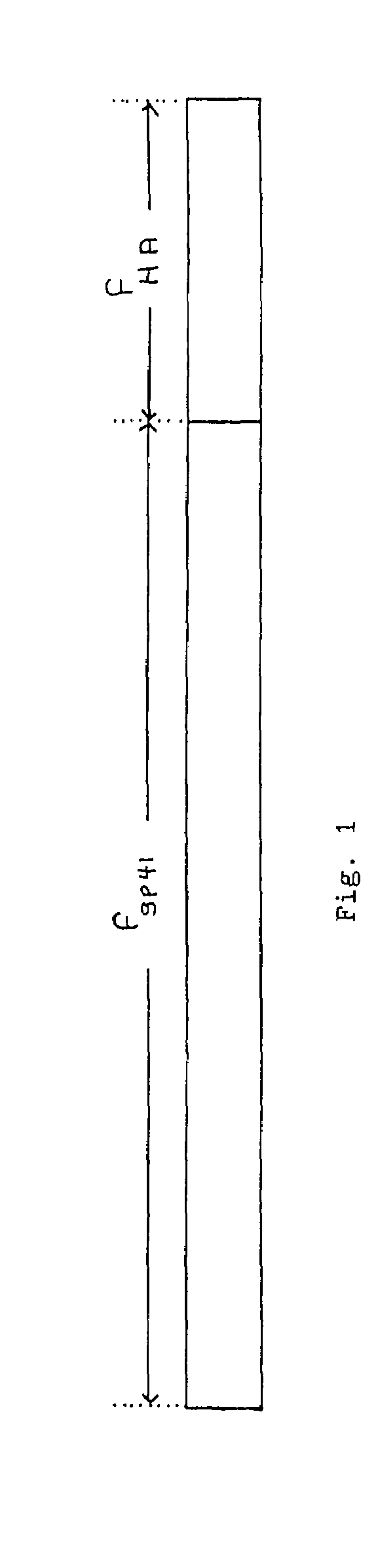
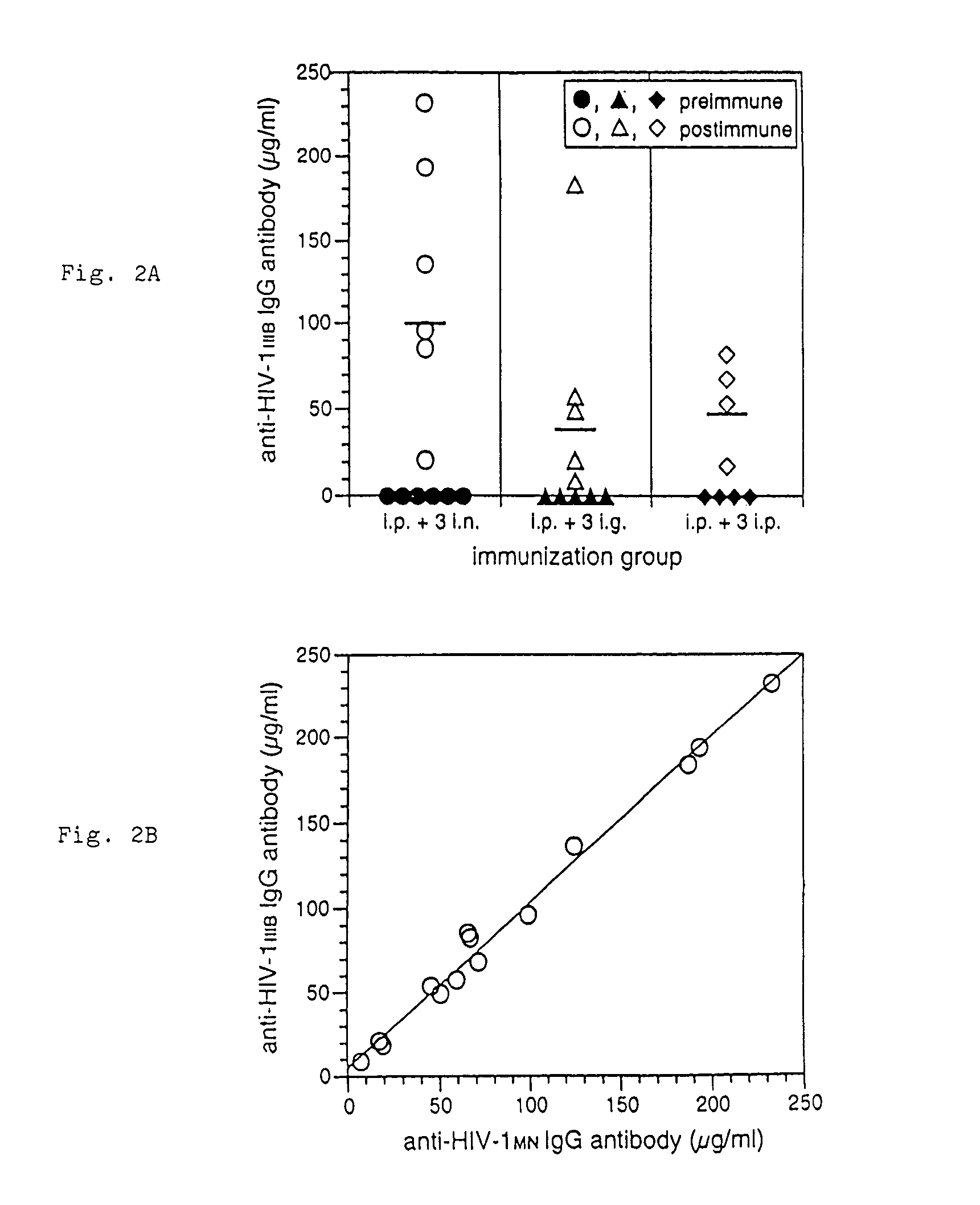
The present invention provides a fusion protein construct (gp41HA) consisting of the ectodomain of the HIV-1IIIB envelope glycoprotein gp41 fused to a fragment of the influenza virus HA2 hemagglutinin protein. Immunization in-vivo via an intraperitoneal prime followed by intranasal or intragastric boosts with gp41HA induces high concentrations of serum IgG antibodies and fecal IgA antibodies that reacted with gp41 in HIV-1IIIB viral lysate and are cross-reactive with gp41 in HIV-1MN lysate. Followup analyses by indirect immunofluorescence showed that both serum IgG and fecal IgA recognized human peripheral blood mononuclear cells infected with either syncytium-inducing (SI) or non-syncytium-inducing (NSI) North American HIV-1 field isolates, but not uninfected cells.
Owner:CHILDRENS MEDICAL CENT CORP
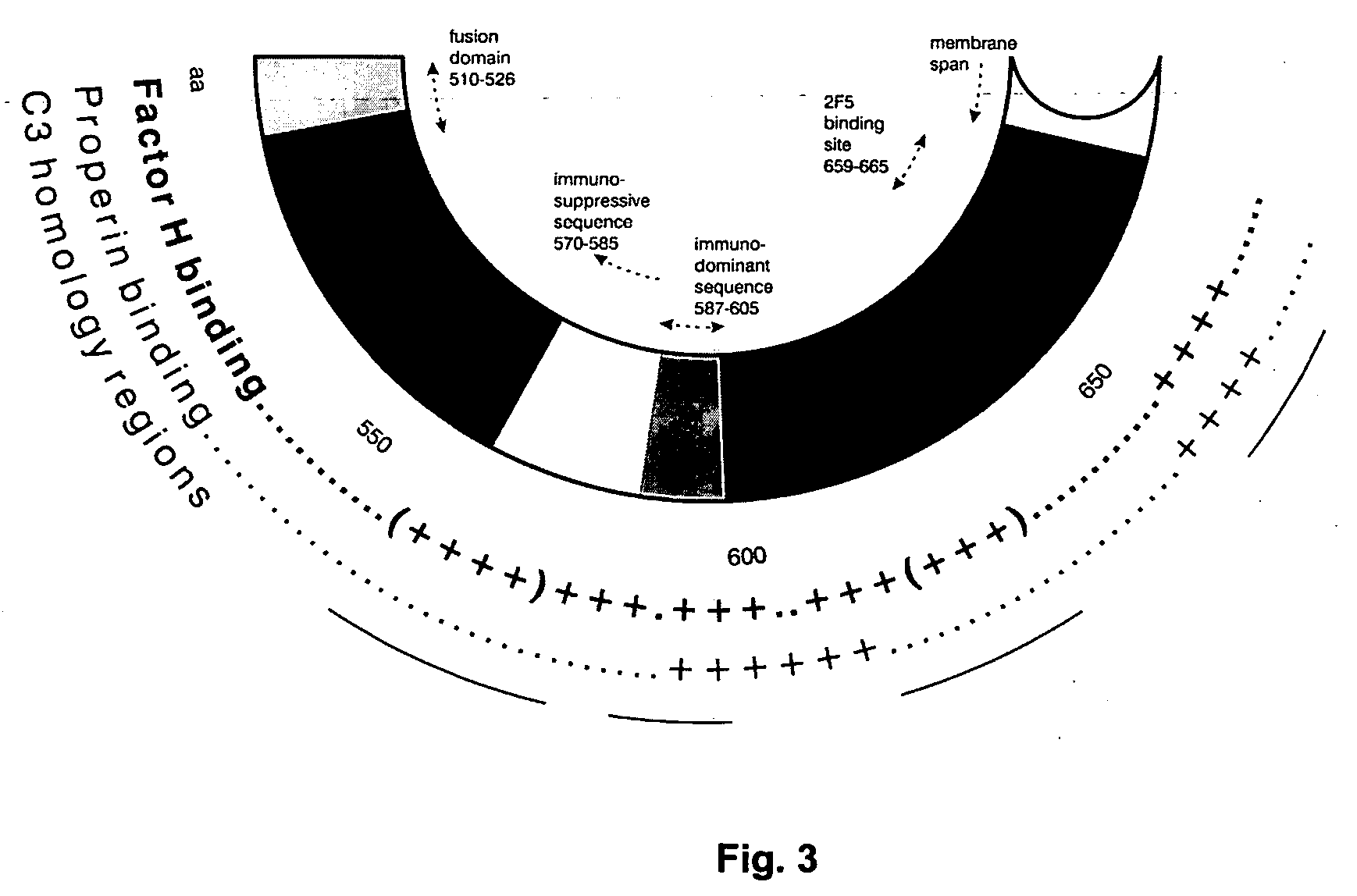
Immunogenic composition and method of developing a vaccine based on factor H binding sites
InactiveUS20050112139A1Rapid decrease in viremiaEasy to copyVirus peptidesPharmaceutical delivery mechanismBinding siteAutoimmune responses
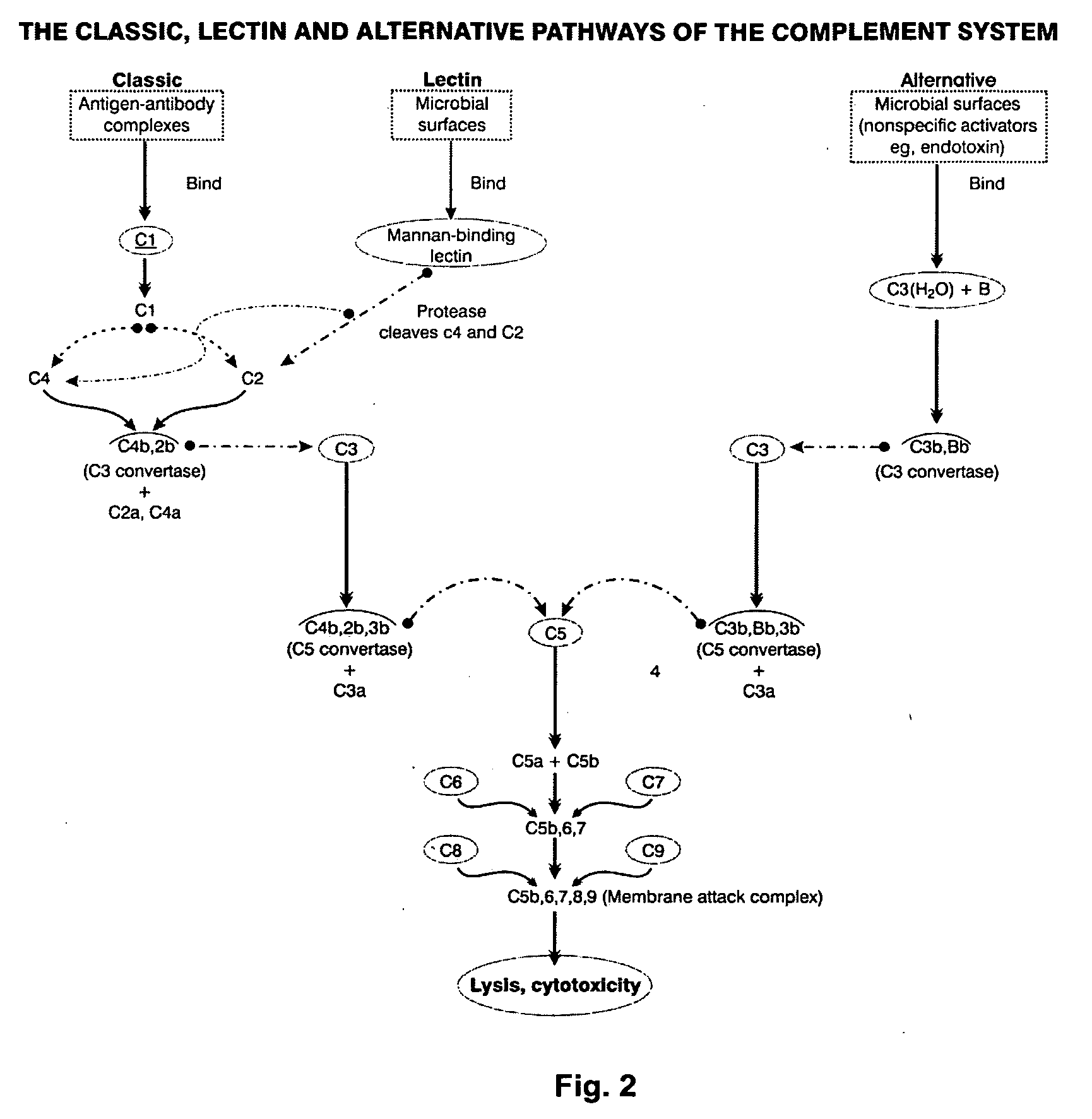
An immunogenic composition able to provide an immune response to Human Complement Factor H binding on one or more HIV glycoproteins is disclosed, which is characterized by at least one binding site epitope of the glycoproteins. Such immunogenic composition wherein the glycoprotein comprises gp120, gp41, or both glycoproteins gp120 and gp41 is hereby disclosed. Sialic acid is removed to enhance immune recognition of the composition and to impair Factor H binding. A medication having an inhibitive action on autoimmune response by specific inhibition of the cleavage of C3b by Factor H into inactive cell fragments.
Owner:KARP NELSON M
Anti-viral compositions comprising heterocyclic substituted phenyl furans and related compounds
InactiveUS20060287319A1Potent anti-HIV activityInhibit HIV replicationBiocideAntiviralsFuranFluorescence
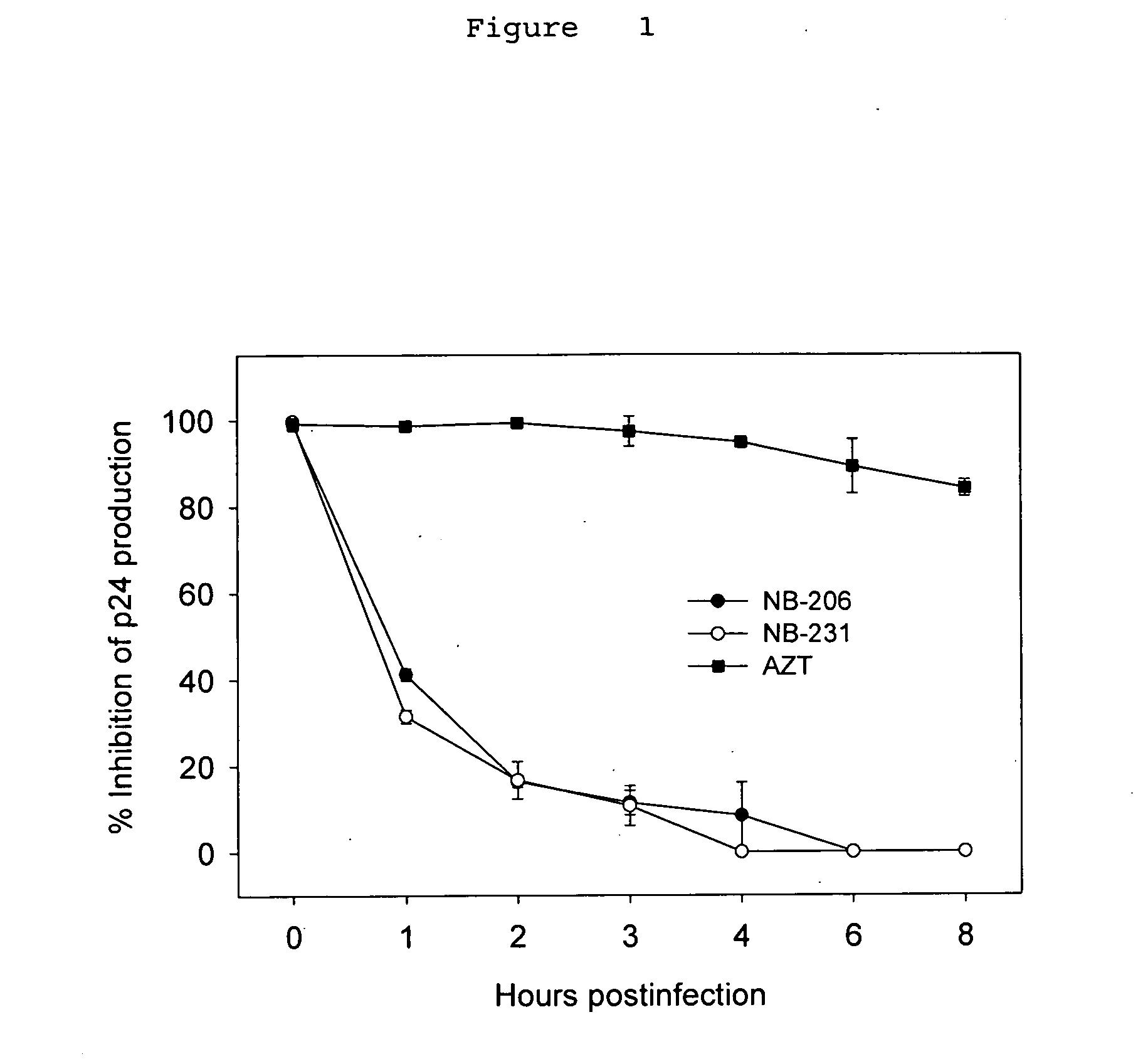
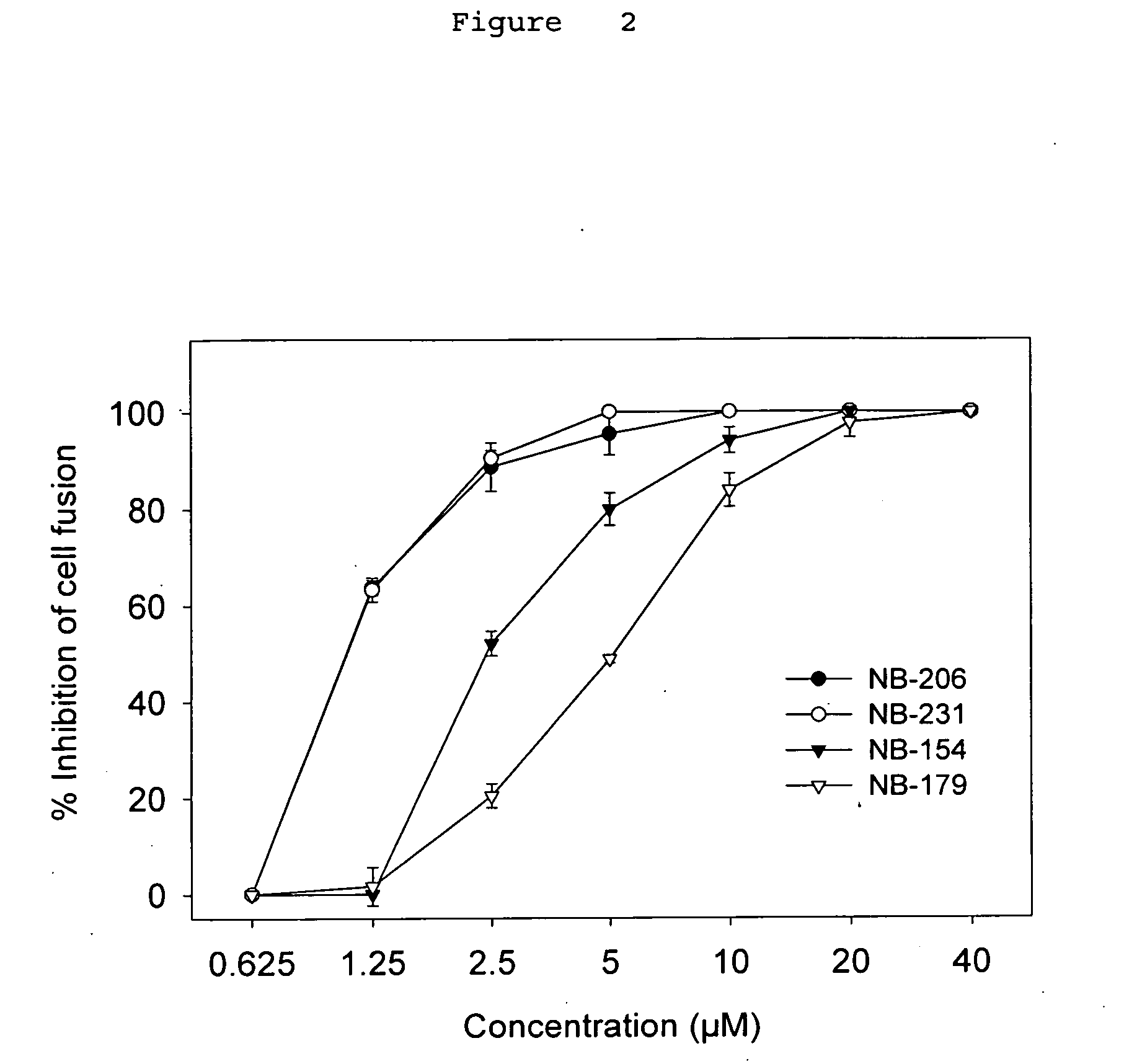
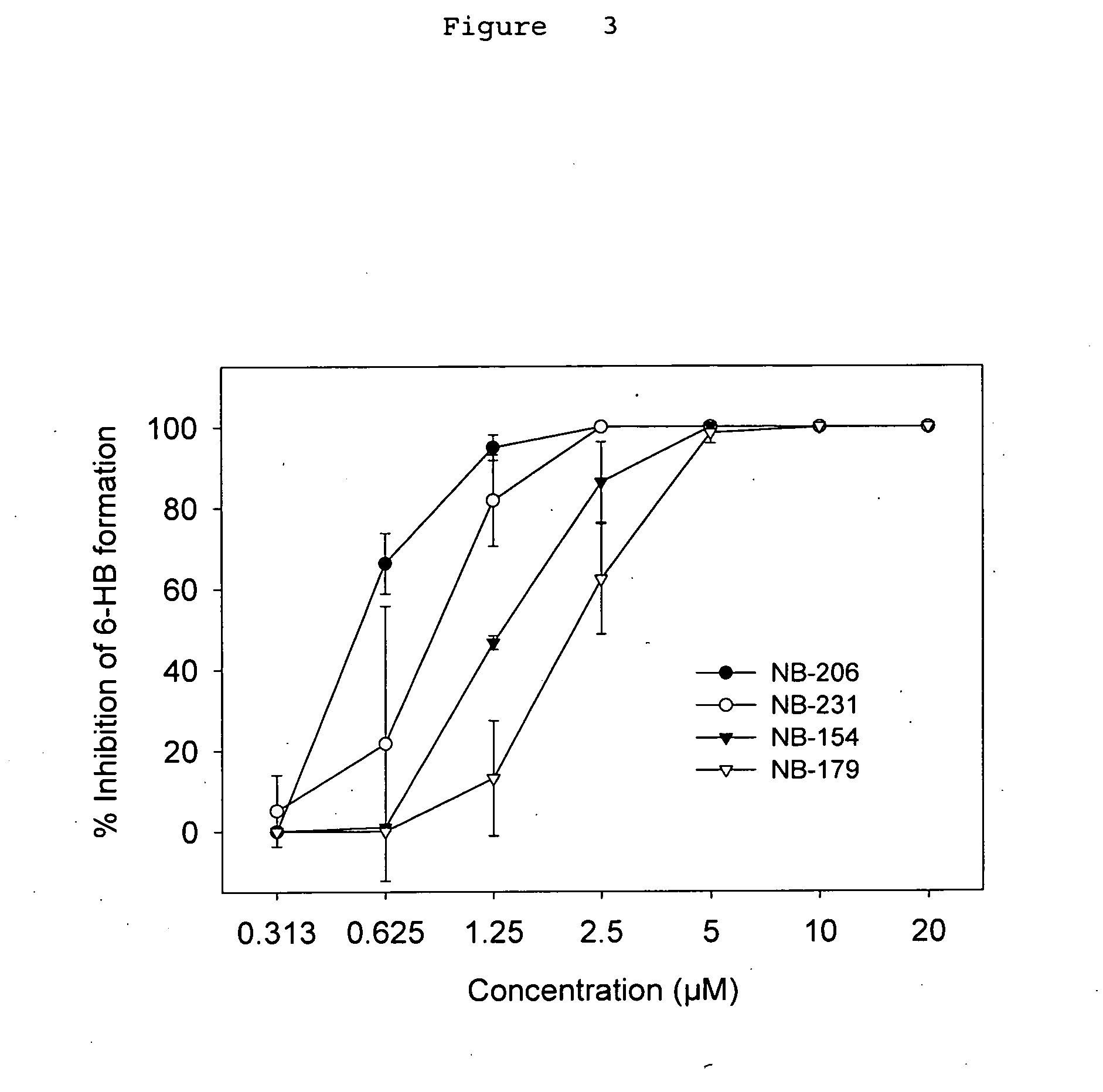
A group of compounds that inhibit HIV replication by blocking HIV entry was identified. One representative compound, designated NB-206, and its analogs inhibited HIV replication (p24 production) with IC50 values at nanomolar levels. It was proved that NB-206 and its analogs are HIV entry inhibitors by targeting the HIV gp41 since: 1) they inhibited HIV-mediated cell fusion; 2) they inhibited HIV replication only when they were added to the cells less than one hour after virus addition; 3) they blocked the formation of the gp41 core that is detected by sandwich enzyme linked immunosorbent assay (ELISA) using a conformation-specific MAb NC-1; and 4) they inhibited the formation of the gp41 six-helix bundle revealed by fluorescence native-polyacrylamide gel electrophoresis (FN-PAGE). These results suggested that NB-206 and its analogs may interact with the hydrophobic cavity and block the formation of the fusion-active gp41 coiled coil domain, resulting in inhibition of HIV-1 mediated membrane fusion and virus entry.
Owner:NEW YORK BLOOD CENT
Stabilization of envelope glycoprotein trimers by disulfide bonds introduced into a gp41 glycoprotein ectodomain
The present application is directed to stabilized envelope glycoprotein trimers. The trimers are stabilized by introducing disulfide bonds at certain sites in the gp41 ectodomain. DNA molecules encoding such trimers can be used to generate an immunogenic reaction.
Owner:DANA FARBER CANCER INST INC
Trimer of HIV env gene expression product
InactiveUS6737067B1Peptide/protein ingredientsViral antigen ingredientsAdjuvantRecombinant glycoprotein
The invention concerns a purified recombinant glycoprotein having the following properties: a) an adherence capacity to CD4; b) an affinity with a anti-gp120 antibody capable of neutralizing in vitro HIV cell infection; c) an affinity with an anti-gp41 antibody; d) a timeric form free from inter-chain disulphide bonds. The invention also concerns a vaccine comprising said purified glycoprotein and an adjuvant. The invention further concerns a method for obtaining said glycoprotein, which consists in expressing, by means of genetic recombining techniques, a glycoprotein corresponding to the properties a), b) and c) mentioned above; purifying it, and subjecting it to steps involving at least a reducing agent, an ionic detergent and / or a neutral detergent in conditions leading to a glycoprotein having said properties.
Owner:AVENTIS PASTUER LTD
Antigen constructs useful in the detection and differentiation of antibodies to HIV
Isolated HIV-1 Group O env polypeptides obtained from the HIV-1 isolate HAM112 are claimed, as well as (a) antigen constructs comprising fusions of one or more of each of HIV-1 Group O env polypeptides and HIV-1 Group M env polypeptide and (b) further antigen constructs containing additional Group O sequences and especially the gp41 IDR of isolate HAM112. Also claimed are polynucleotide sequences encoding the above, expression vectors comprising the same, host cells transformed thereby, and immunoassay methods and kits utilizing the antigen constructs of the invention.
Owner:ABBOTT LAB INC
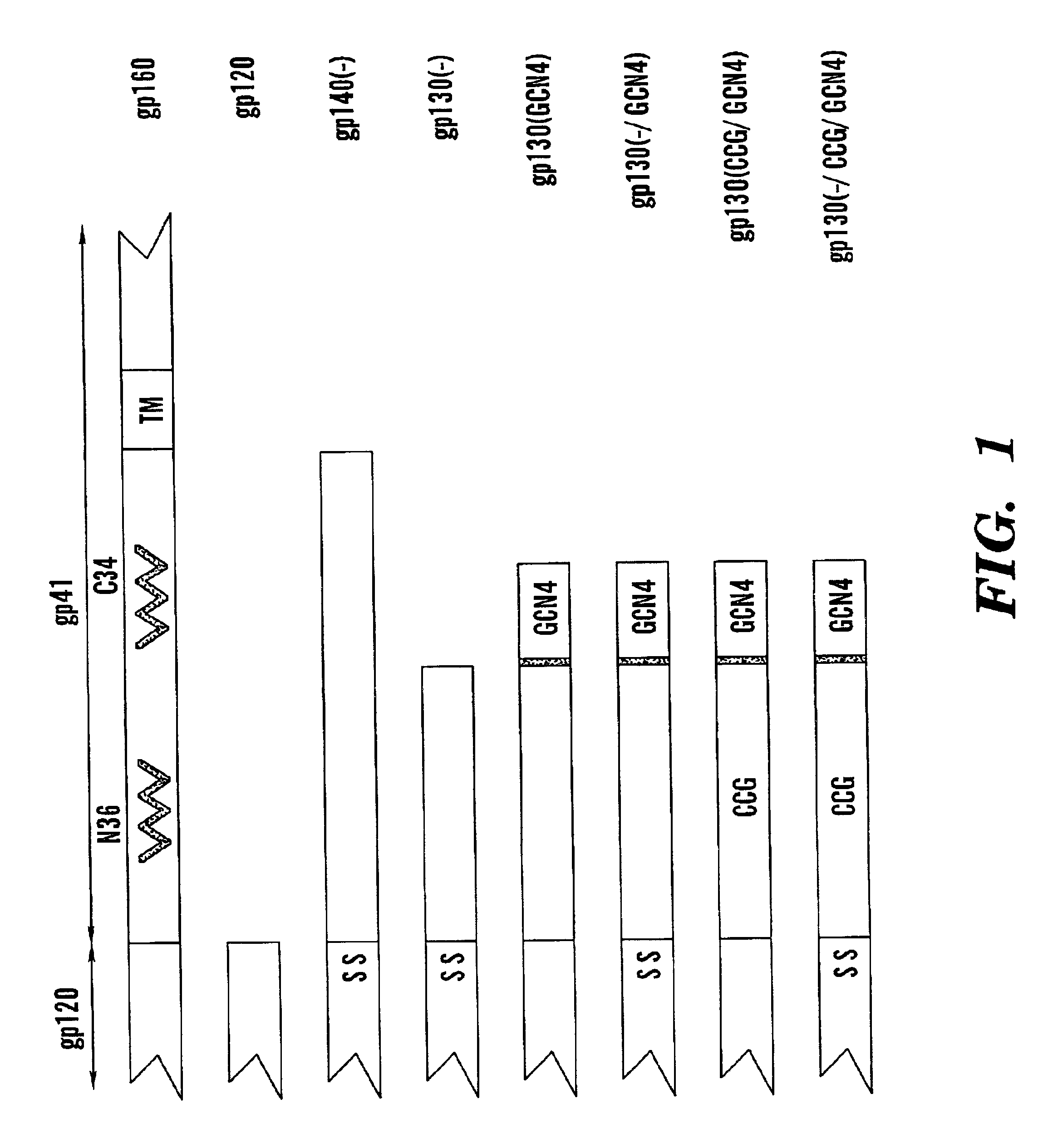
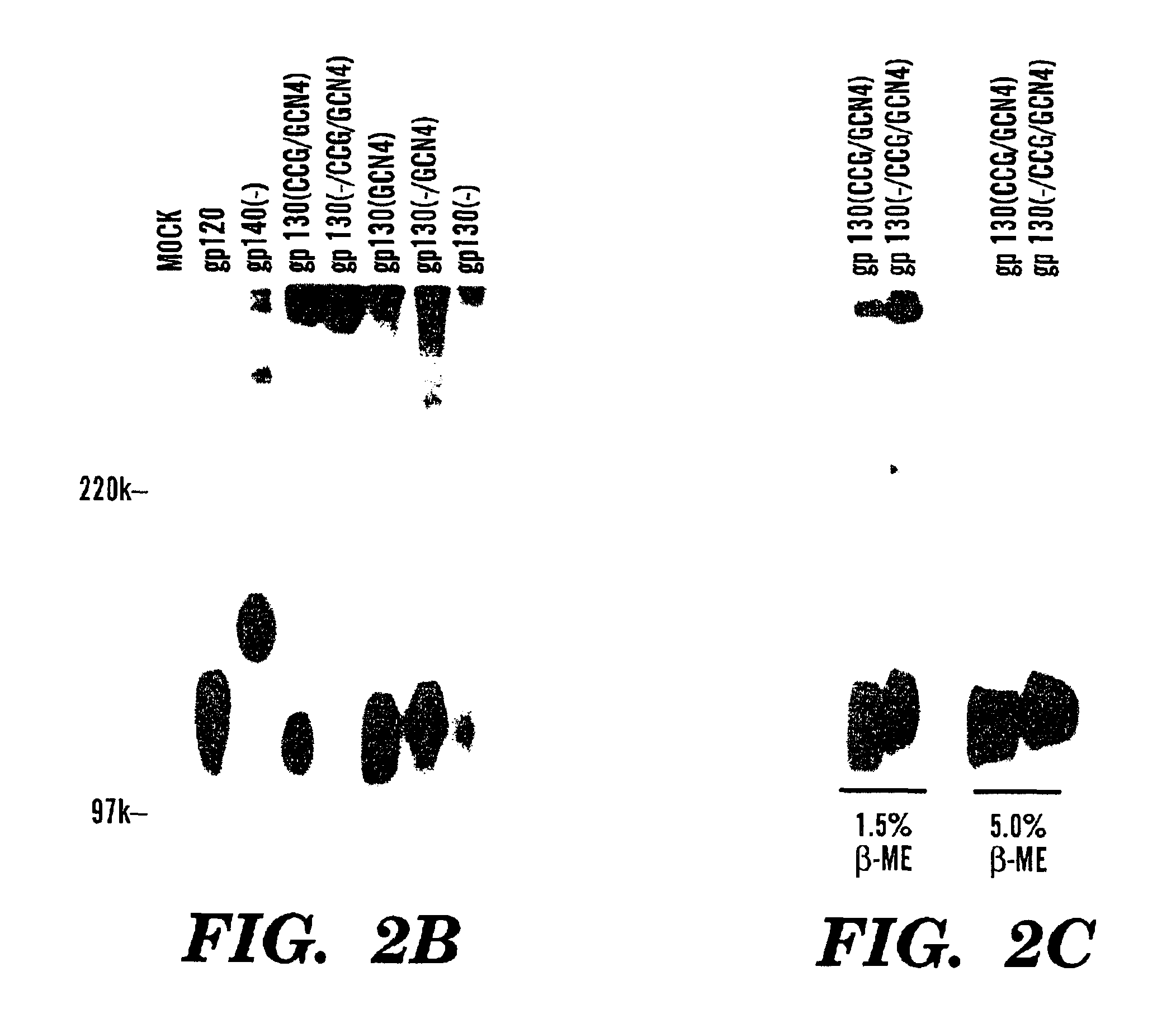
Stabilized soluble glycoprotein trimers
InactiveUS20050106177A1Stabilize trimerSimplifies isolationPeptide/protein ingredientsAntibody mimetics/scaffoldsHiv envelopeGp41
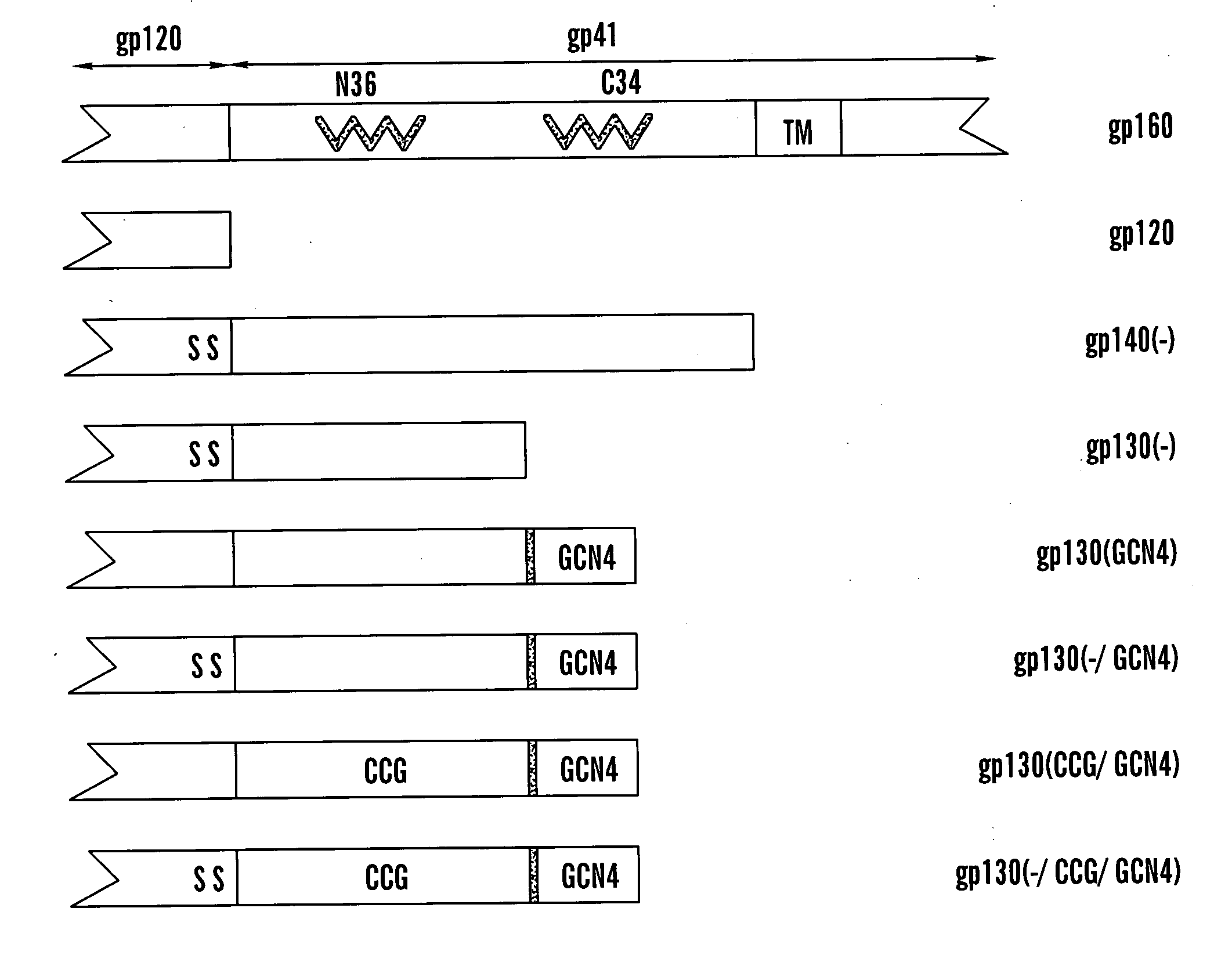
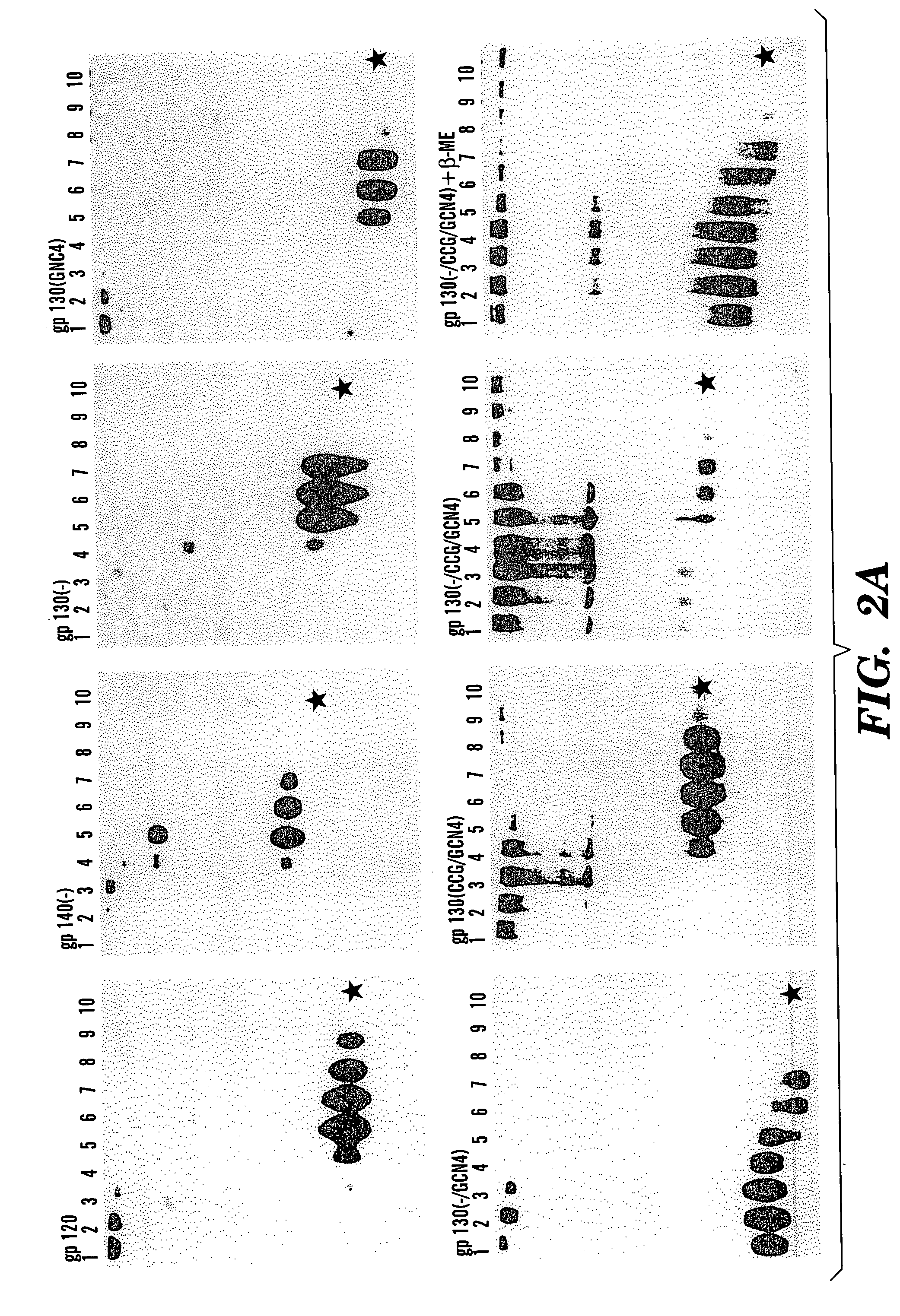
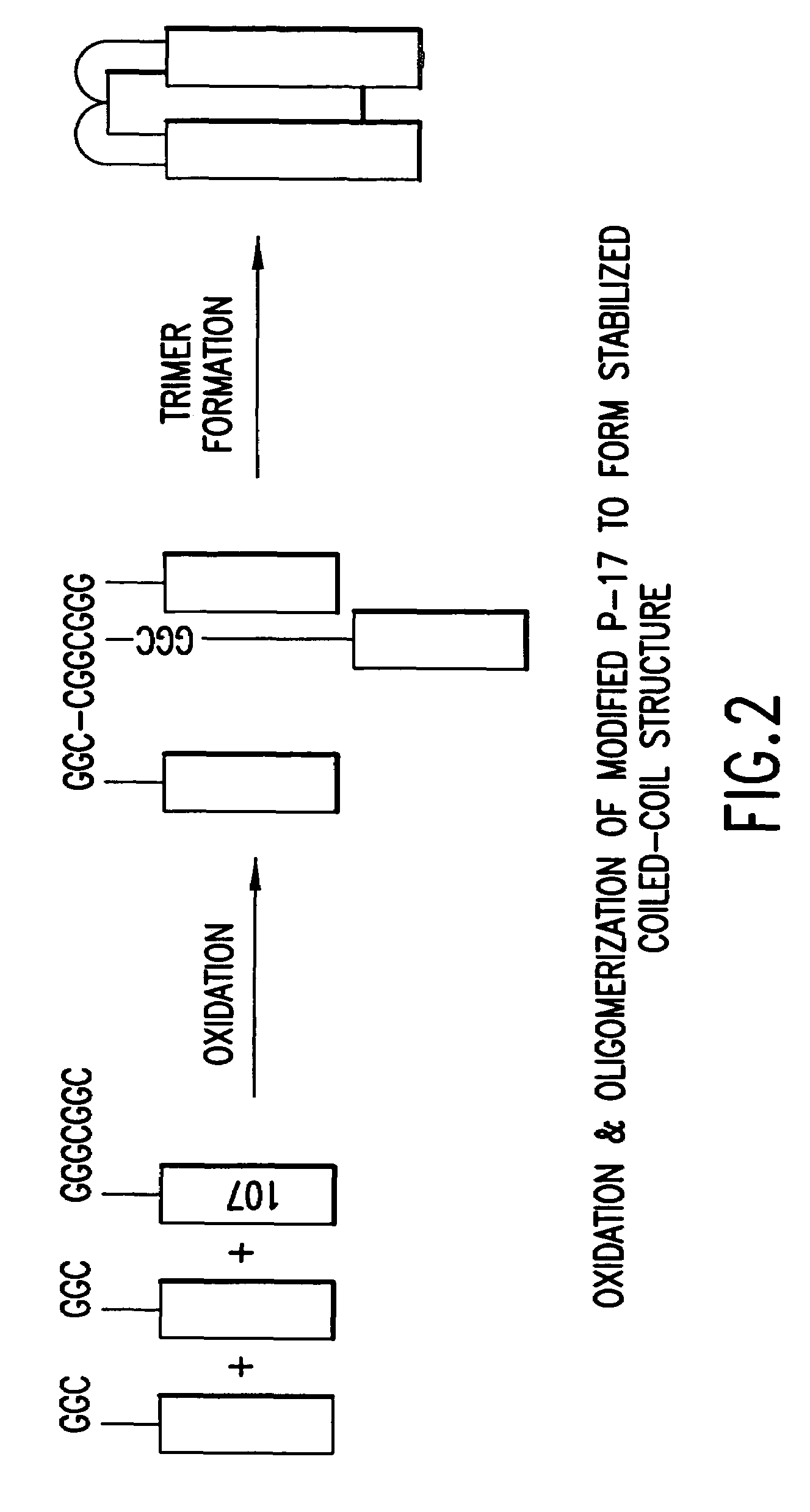
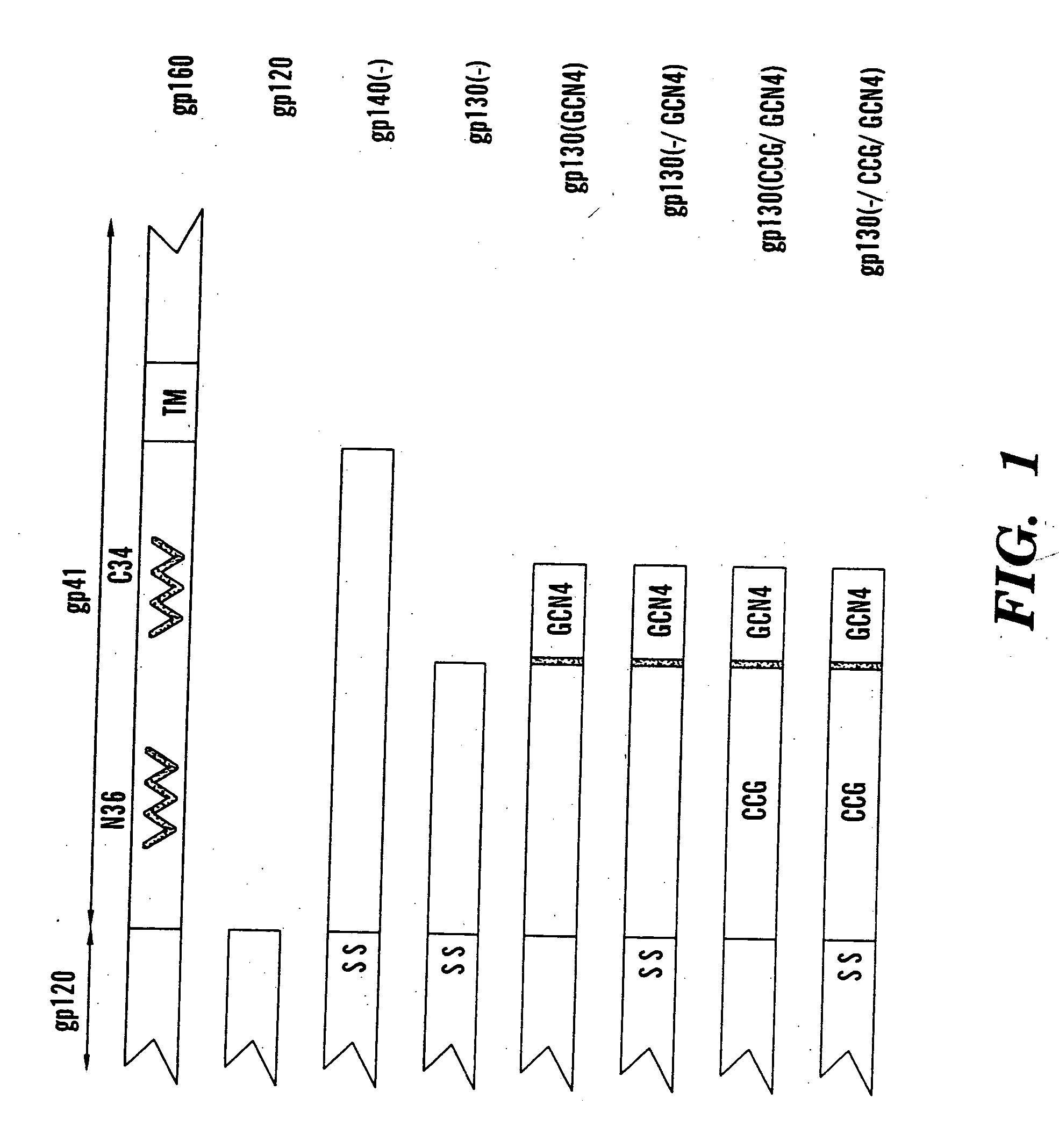
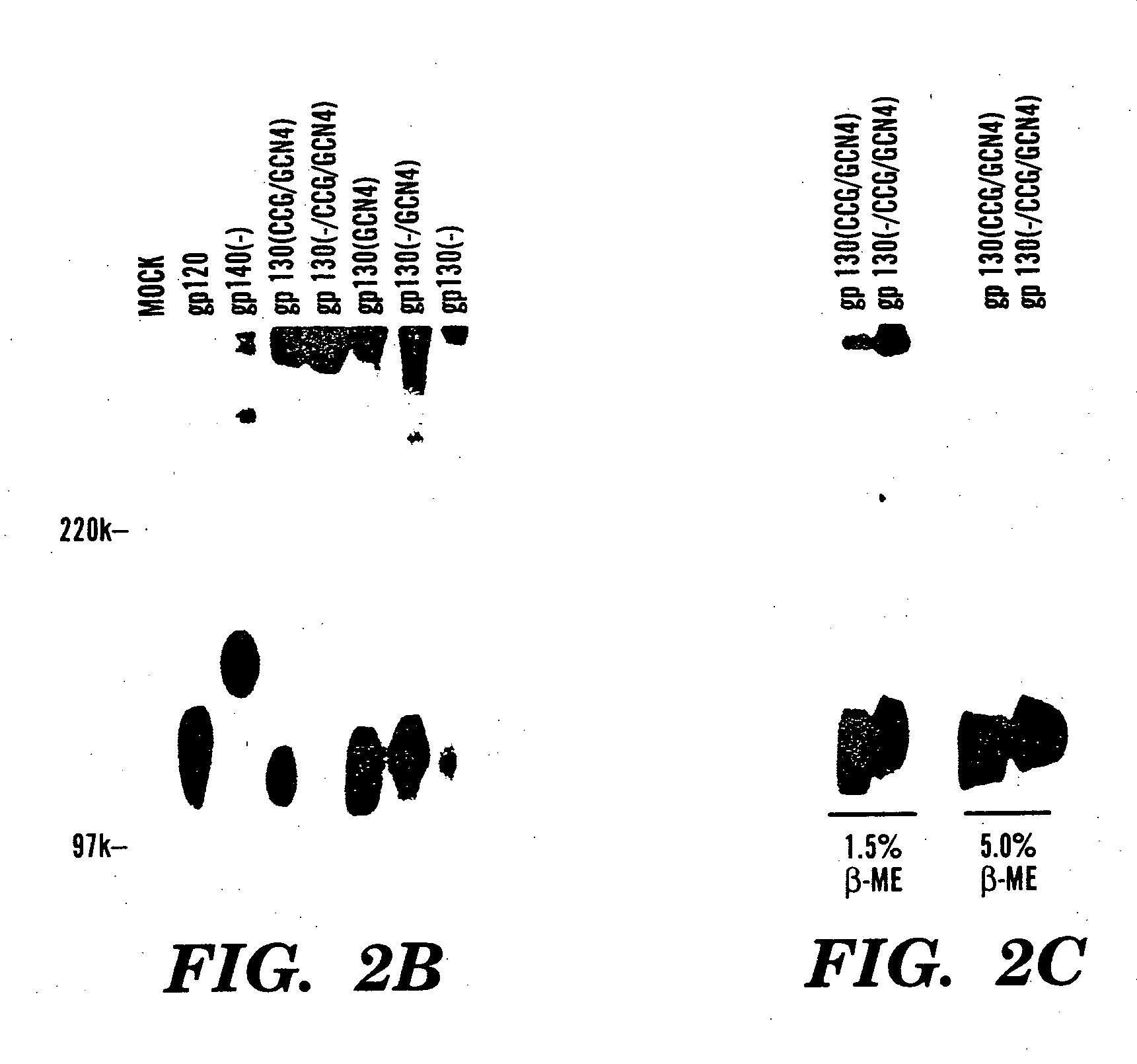
The present application is directed to stabilized HIV envelope glycoprotein trimers. The trimers are stabilized by introducing trimeric motifs, preferably the GCN4 coiled coil or the fibritin trimeric domain, at certain sites, for example in the gp41 ectodomain. These stabilized trimers or DNA molecules encoding such trimers can be used to generate an immunogenic reaction. The trimers can also be used in assays to screen for molecules that interact with them—and to identify molecules that interact with specific sites.
Owner:THE TRUSTEES OF COLUMBIA UNIV IN THE CITY OF NEW YORK
HIV gp41 HR2-derived synthetic peptides, and their use in therapy to inhibit transmission of human immunodeficiency virus
InactiveUS20060247416A1Improve biological activityStrong antiviral activityPolypeptide with localisation/targeting motifSugar derivativesImmunodeficiency virusAmino acid substitution
Provided are synthetic peptides based on a native sequence of HIV gp41 HR2 except that the synthetic peptides have a plurality of amino acid replacements comprising (a) a helix-promoting amino acid, or (b) a combination of helix-promoting amino acids, and charged amino acids introduced to form ion pairs in the synthetic peptide; wherein the synthetic peptides demonstrate an unexpected, improved biological activity, as compared to a peptide having an amino acid sequence without the plurality of amino acid substitutions. Also provided are polynucleotides encoding synthetic peptide, and methods of using these synthetic peptides in inhibition of, or as compositions to inhibit, transmission of HIV to a target cell.
Owner:TRIMERIS
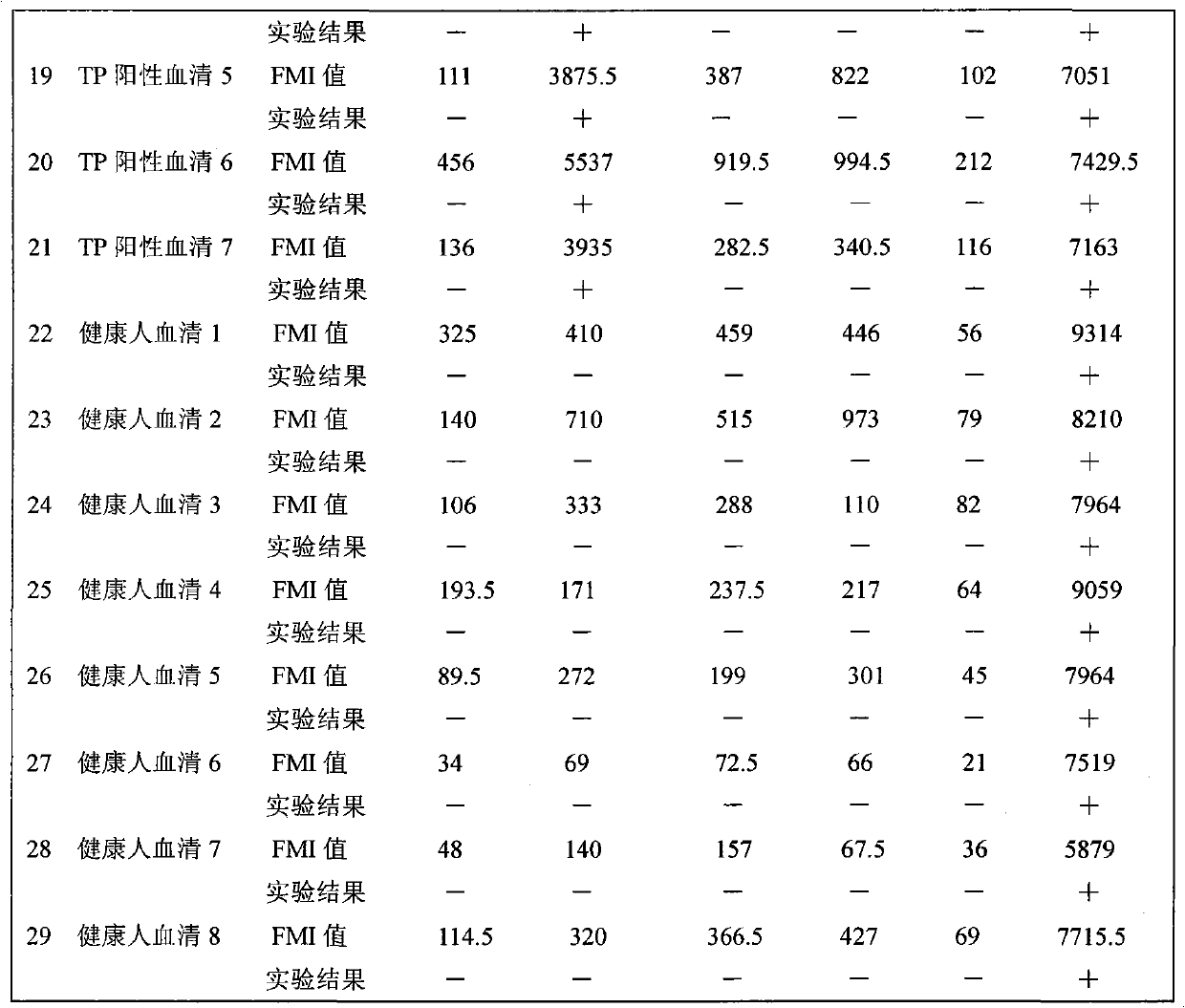
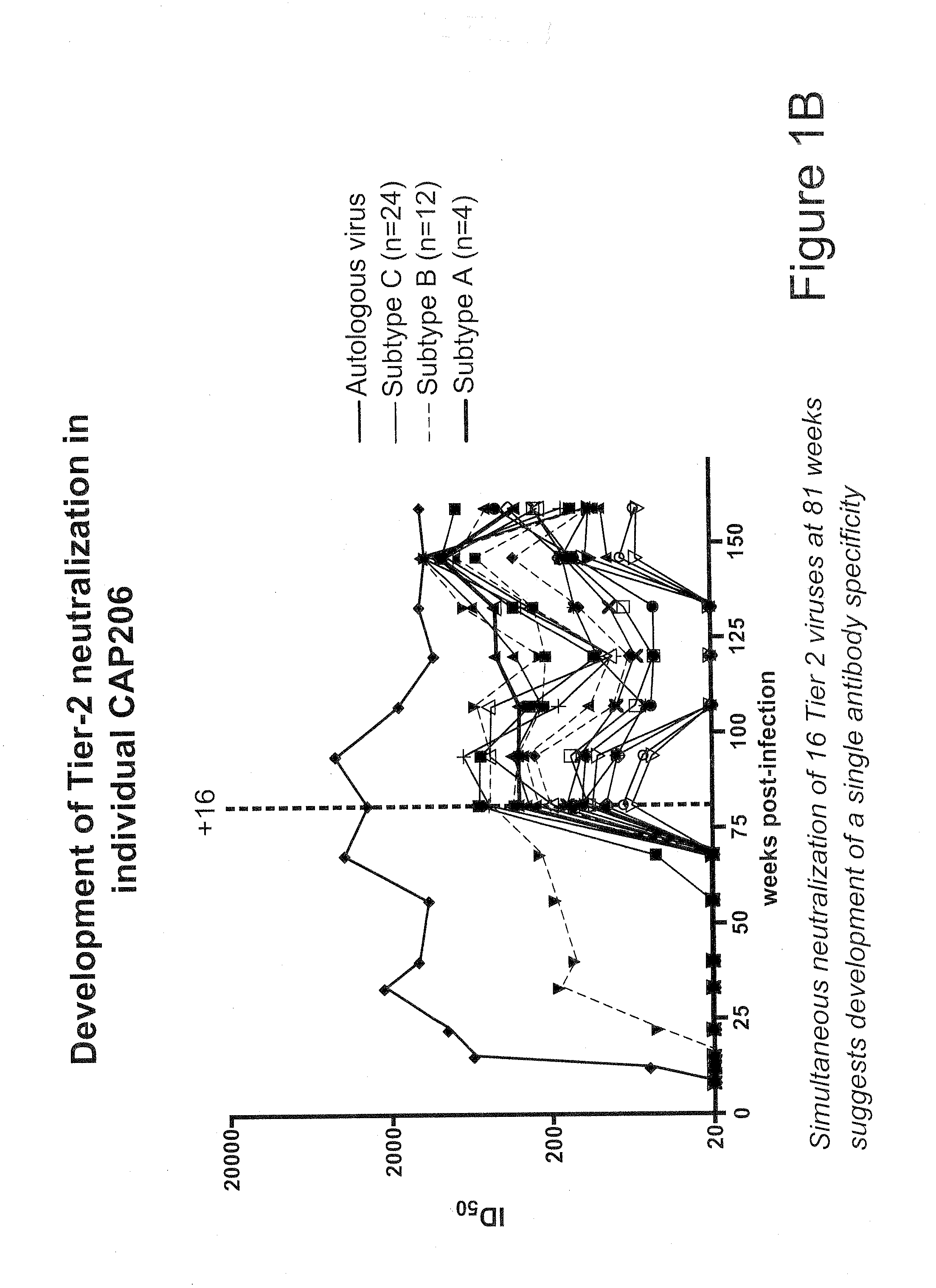
Methods of eliciting broadly neutralizing antibodies targeting HIV-1 gp41
The present invention is directed to the induction and characterization of a humoral immune response targeting "entry-relevant" gp41 structures. In its broadest aspect, the present invention is directed to methods of raising a neutralizing antibody response to a broad spectrum of HIV strains and isolates. The present invention targets particular molecular conformations or structures that occur at the cell surface of HIV during viral entry into host cells. Such a humoral response can be generated in vivo as a prophylactic measure in individuals to reduce or inhibit the ability of HIV to infect uninfected cells in the individual's body. Such a response can also be employed to raise antibodies against "entry relevant" gp41 structures. These antibodies can be employed for therapeutic uses, and as tools for further illuminating the mechanism of HIV cell entry.
Owner:UNITED STATES OF AMERICA +1
Modified HIV-1 clade C envelope glycoprotein immunogens comprising deletions in the gp120/gp41 cleavage site and gp41 fusion domain
The present invention relates, in general, to an immunogen and, in particular, to an immunogen for inducing antibodies that neutralize a wide spectrum of HIV primary isolates and / or to an immunogen that induces a T cell immune response. The invention also relates to a method of inducing anti-HIV antibodies, and / or to a method of inducing a T cell immune response, using such an immunogen. The invention further relates to nucleic acid sequences encoding the present immunogens.
Owner:DUKE UNIV
Methods of eliciting broadly neutralizing antibodies targeting HIV-1 gp41
The present invention is directed to the induction and characterization of a humoral immune response targeting “entry-relevant” gp41 structures. In its broadest aspect, the present invention is directed to methods of raising a neutralizing antibody response to a broad spectrum of HIV strains and isolates. The present invention targets particular molecular conformations or structures that occur at the cell surface of HIV during viral entry into host cells. Such a humoral response can be generated in vivo as a prophylactic measure in individuals to reduce or inhibit the ability of HIV to infect uninfected cells in the individual's body. Such a response can also be employed to raise antibodies against “entry relevant” gp41 structures. These antibodies can be employed for therapeutic uses, and as tools for further illuminating the mechanism of HIV cell entry.
Owner:UNITED STATES OF AMERICA +1
Methods and compositions for inhibition of membrane fusion-associated events, including HIV transmission
The present invention relates to peptides which exhibit potent anti-retroviral activity. The peptides of the invention comprise DP178 (SEQ ID:1) peptide corresponding to amino acids 638 to 673 of the HIV-1LAI gp41 protein, and fragments, analogs and homologs of DP178. The invention further relates to the uses of such peptides as inhibitory of human and non-human retroviral, especially HIV, transmission to uninfected cells.
Owner:TRIMERIS
Chimeric HIV fusion proteins as vaccines
InactiveUS20120213811A1Polypeptide with localisation/targeting motifAntibody mimetics/scaffoldsPseudomonas aeruginosa exotoxin AReticulum cell
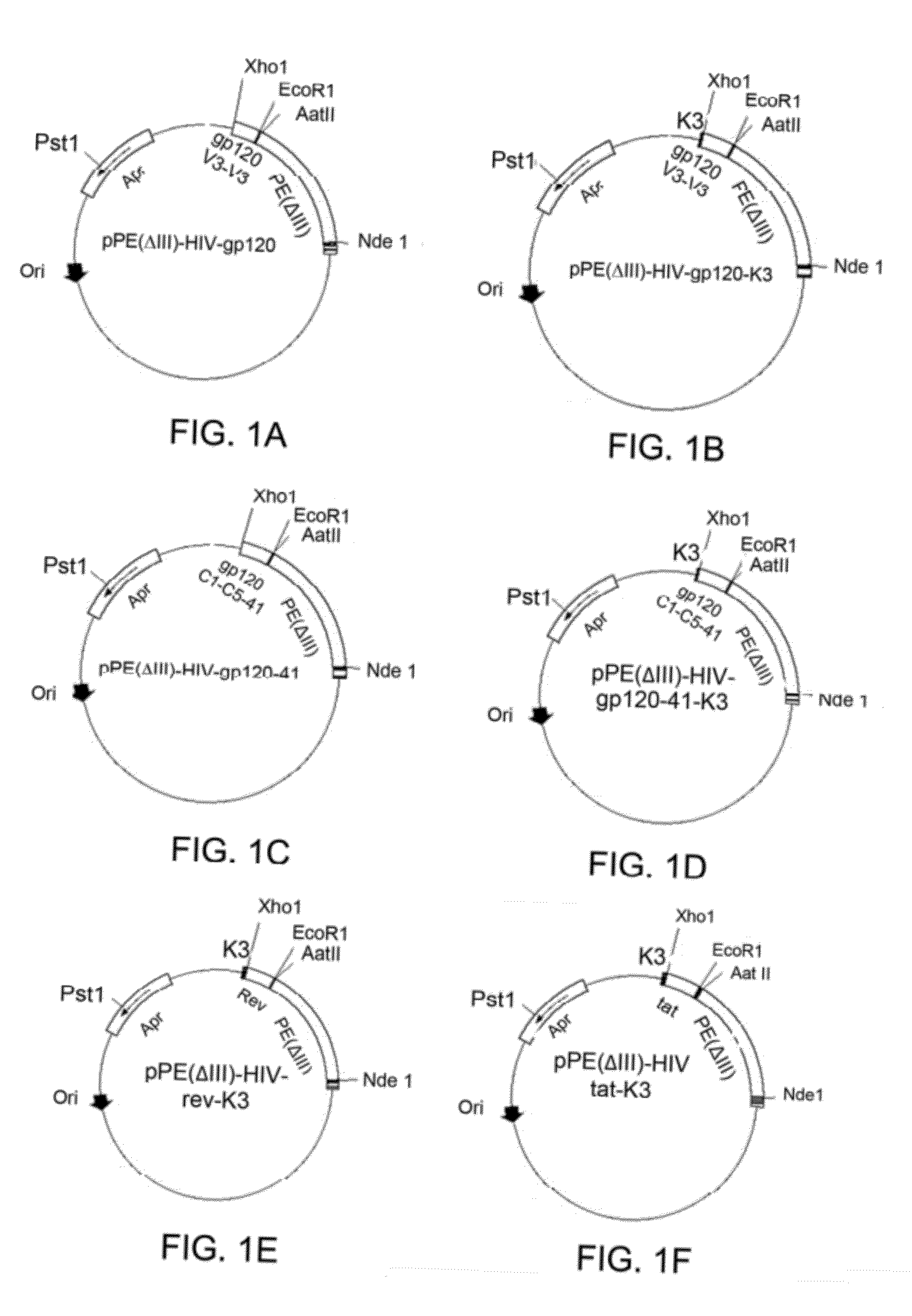
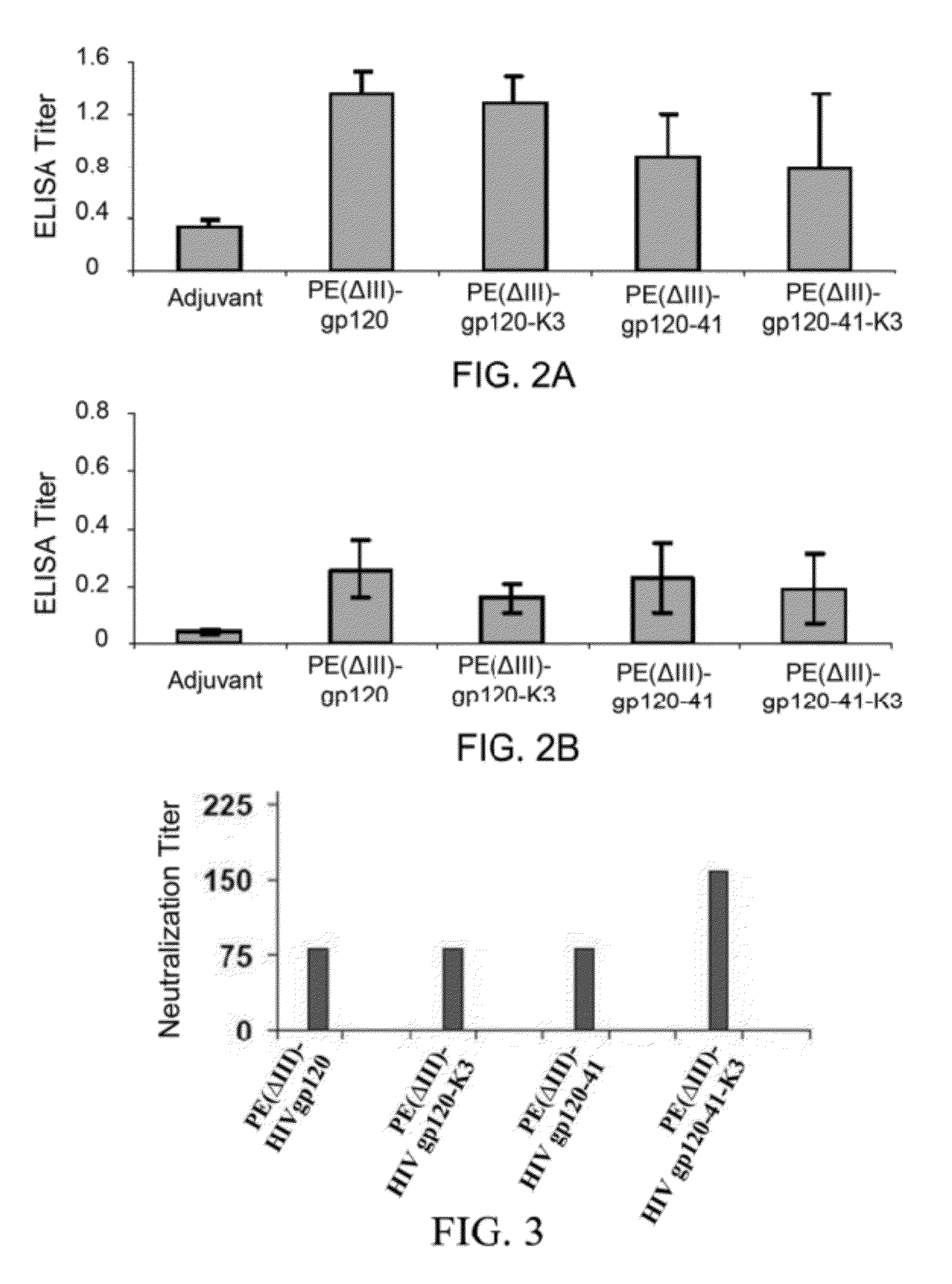
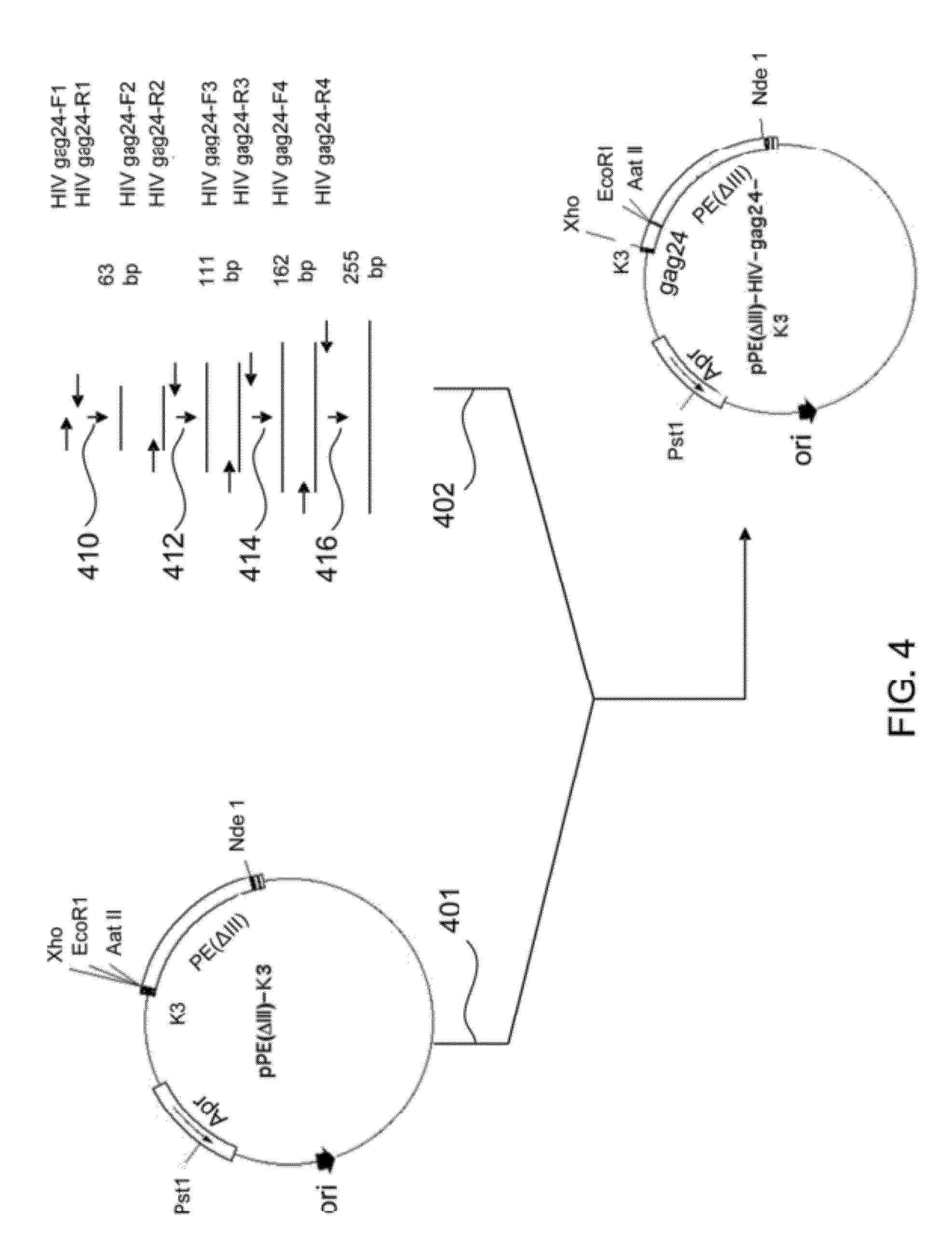
A method for treating HIV infection is disclosed. The method comprises administering to a patient in need thereof a therapeutically effective amount of a fusion protein comprising: a) a Pseudomonas Exotoxin A (PE) peptide comprising a binding domain and a PE translocation domain, the PE peptide being devoid of cytotoxic domain III; b) optionally gag24, being fused to the PE peptide; c) a fragment of gp120 C1 domain, being fused to the PE peptide or fused to the gag24 if the gag24 is present; d) a fragment of gp 120 C5 domain, being fused to the fragment of gp120 C1 domain; e) a fragment of gp41 amino acid sequence, being fused to the fragment of gp 120 C5 domain, and f) optionally an endoplasmic reticulum retention sequence, being fused to the C-terminus of the fragment of gp41.
Owner:THEVAX GENETICS VACCINE
Methods and compositions for the inhibition of HIV infection of t cells
InactiveUS20090053220A1Inhibit bindingAntiviralsAntibody ingredientsAntiendomysial antibodiesSoluble CD4
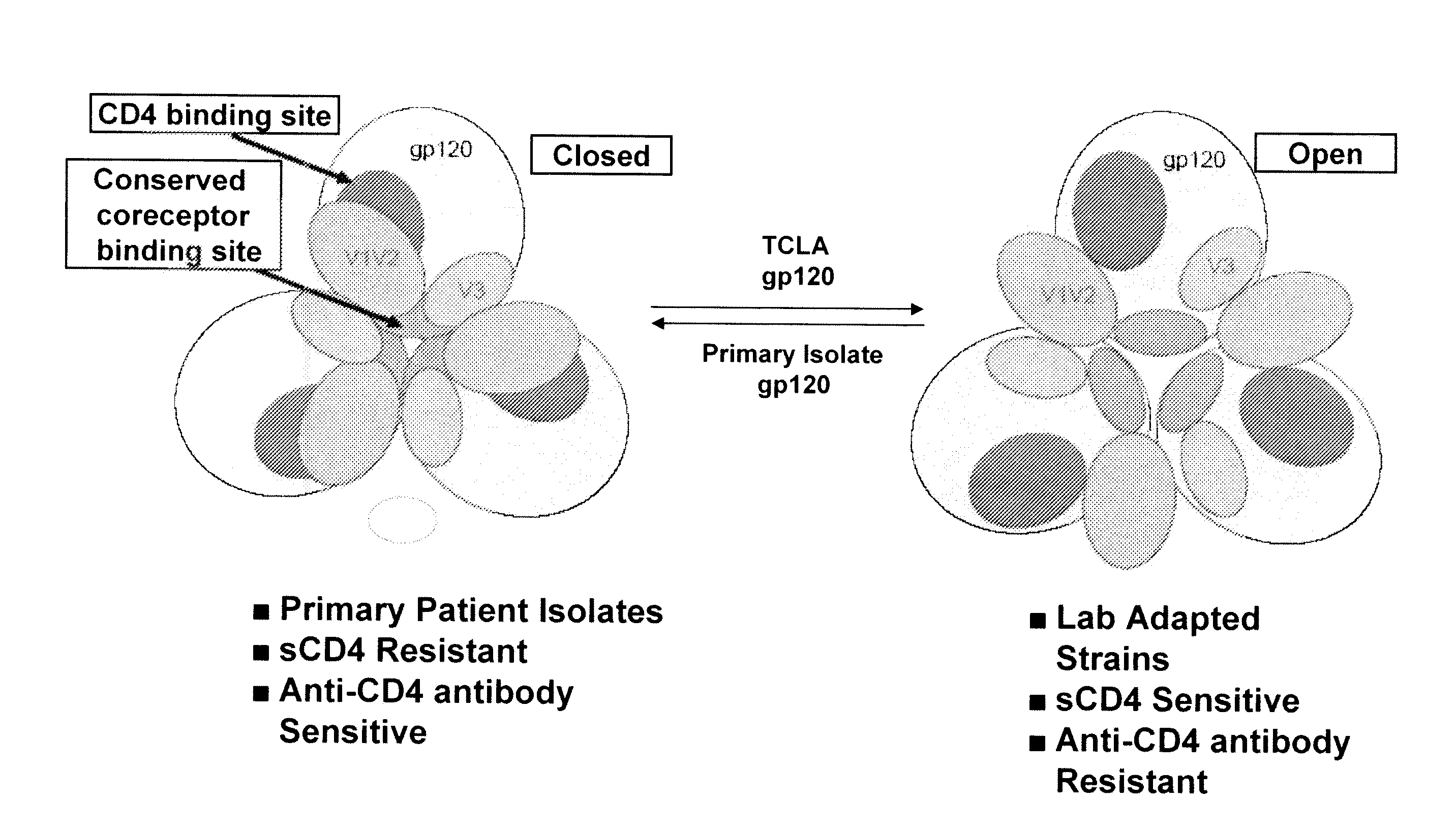
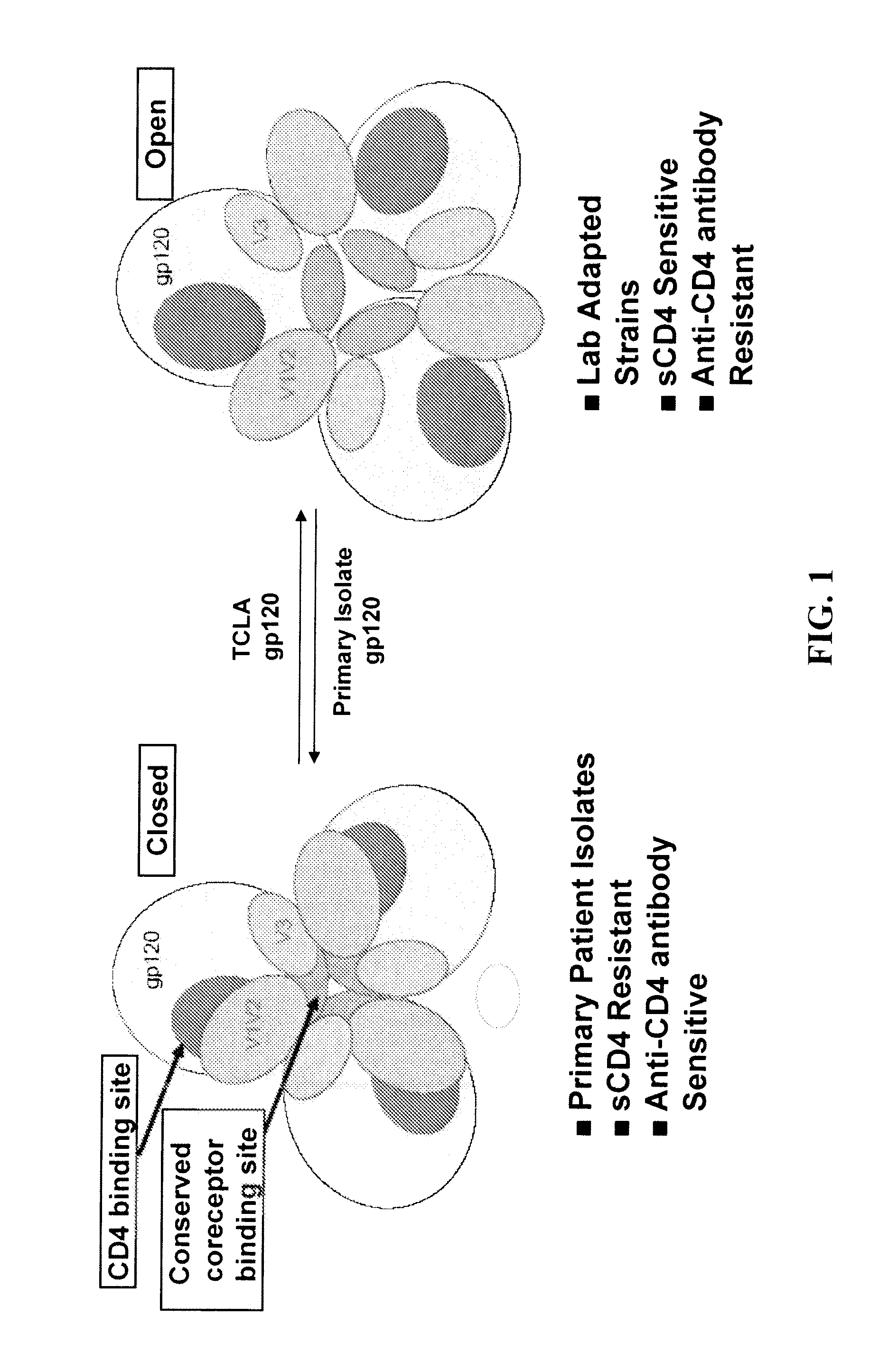
The present invention is based upon the surprising discovery that exposure of a non-resistant HIV to a first entry inhibitor, such as an anti-CD4 antibody or a co-receptor inhibitor, which like all current HIV drugs selects for mutations that result in a resistant HIV, surprisingly results in HIV viruses much more susceptible to neutralization by a second entry inhibitor, such as soluble CD4 (sCD4) or an HIV gp41 inhibitor. Therefore, the present invention provides methods and compositions for inhibiting HIV-1 infection in a subject that overcomes the problem of drug resistance.
Owner:GENENTECH INC
Human immunodeficiency virus antibody confirmations reagent
InactiveCN101097216AFor long-term storageMaterial analysis by observing effect on chemical indicatorBiological testingHuman immunodeficiency virus antibodyImmunodeficiency virus
The invention relates to a kind of affirmation regent for antibody of human immunodeficiency virus, and the diagnosing regent consists of HIV-I type specificity protein gp160, gp120, gp41, p66, p24, mixed fiber film which is encysted by HIV-II specificity protein gp36, enzyme conjugates and other component, and the principle of indirect process is provided for the affirmation of antibody of HIV-I and HIV-II type.
Owner:曹健荣 +1
Polypeptides comprising epitopes of HIV gp41 and methods of use
An isolated polypeptide that has an amino acid sequence that is substantially homologous to consecutive amino acids of a c-terminal portion of the membrane proximal external region (MPER) of gp41 includes an immunogenic epitope reactive with at least one anti-HIV antibody.
Owner:CASE WESTERN RESERVE UNIV
Polypeptide of controlling IIIv virus fusion and its use
InactiveCN1955190AEasy to synthesizeHigh activityPeptide/protein ingredientsVirus peptidesSolubilityMechanism of action
This invention provides a category of polypeptide that restrain HIV virus to mix together. According to the mechanism of action of HIV virus and adaptor molecule which is on the surface of cellular membrane in the process of membrane fusion, this invention designs an active polypeptide which can bind with virus surface glycoprotein gp41. Effect situs of mentioned polypeptide is at trimer NHR lateral 'long point' and bilateralis that is in gp41 molecule, it can effectively restrain reproduction of HIV virus. The polypeptide of this invention compared with present medicine, has small molecular weight, high activity, good water solubility. Using polypeptide that is mentioned by this invention can prepare medicine which can restrain many kinds of viral infection.
Owner:INST OF BIOENG ACAD OF MILITARY MEDICAL SCI OF THE CHINESE +1
Lipopeptide inhibitors of HIV-1
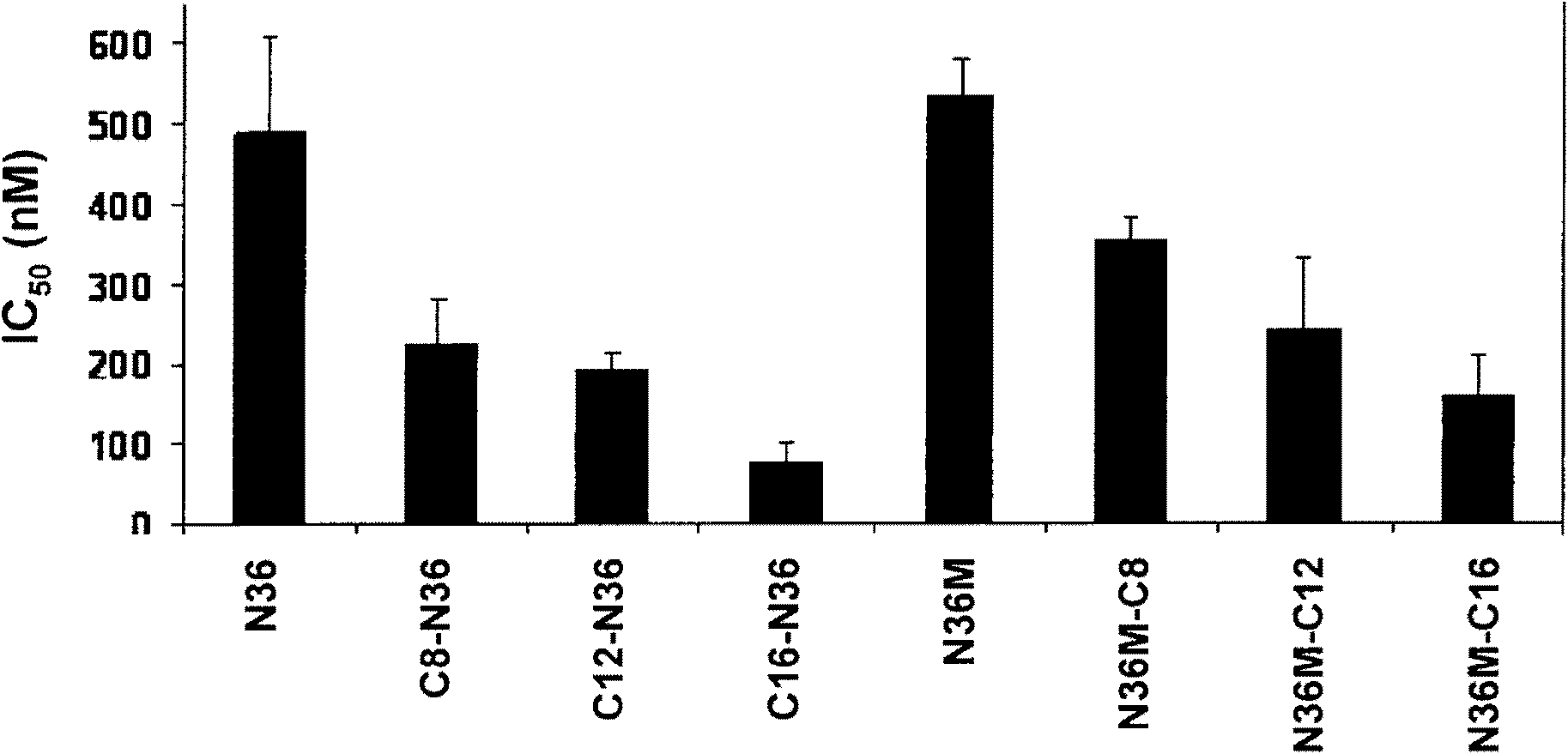
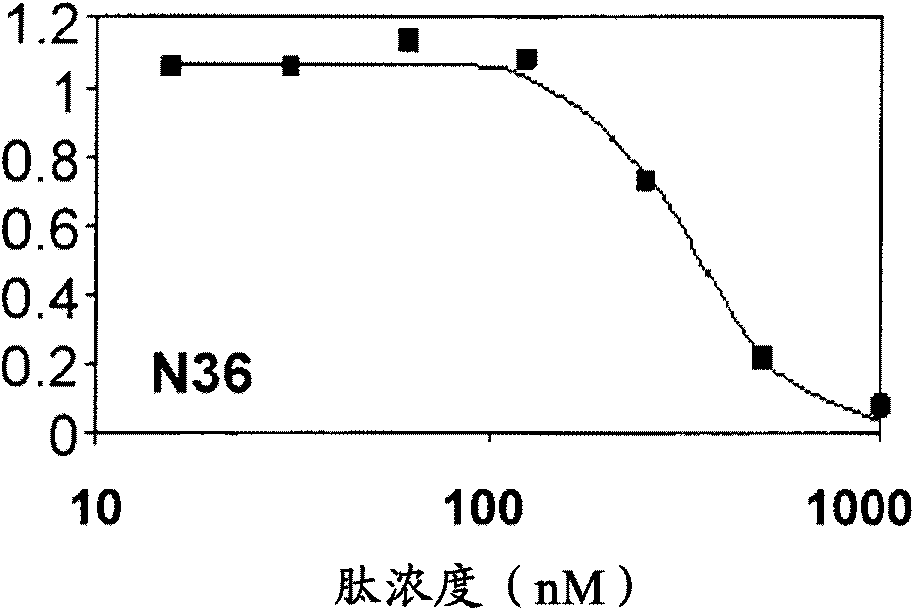
The invention provides lipophilic conjugates comprising a short isolated peptide coupled to a hydrophobic moiety, the peptide comprising a sequence derived from the HIV-1 gp41 N-terminal heptad repeat domain, said peptide after conjugation to the hydrophobic moiety possesses anti-fusogenic activity higher than prior to conjugation. The lipophilic conjugates are suitable for treatment of infections caused by human and non-human retroviruses, especially HIV.
Owner:YEDA RES & DEV CO LTD
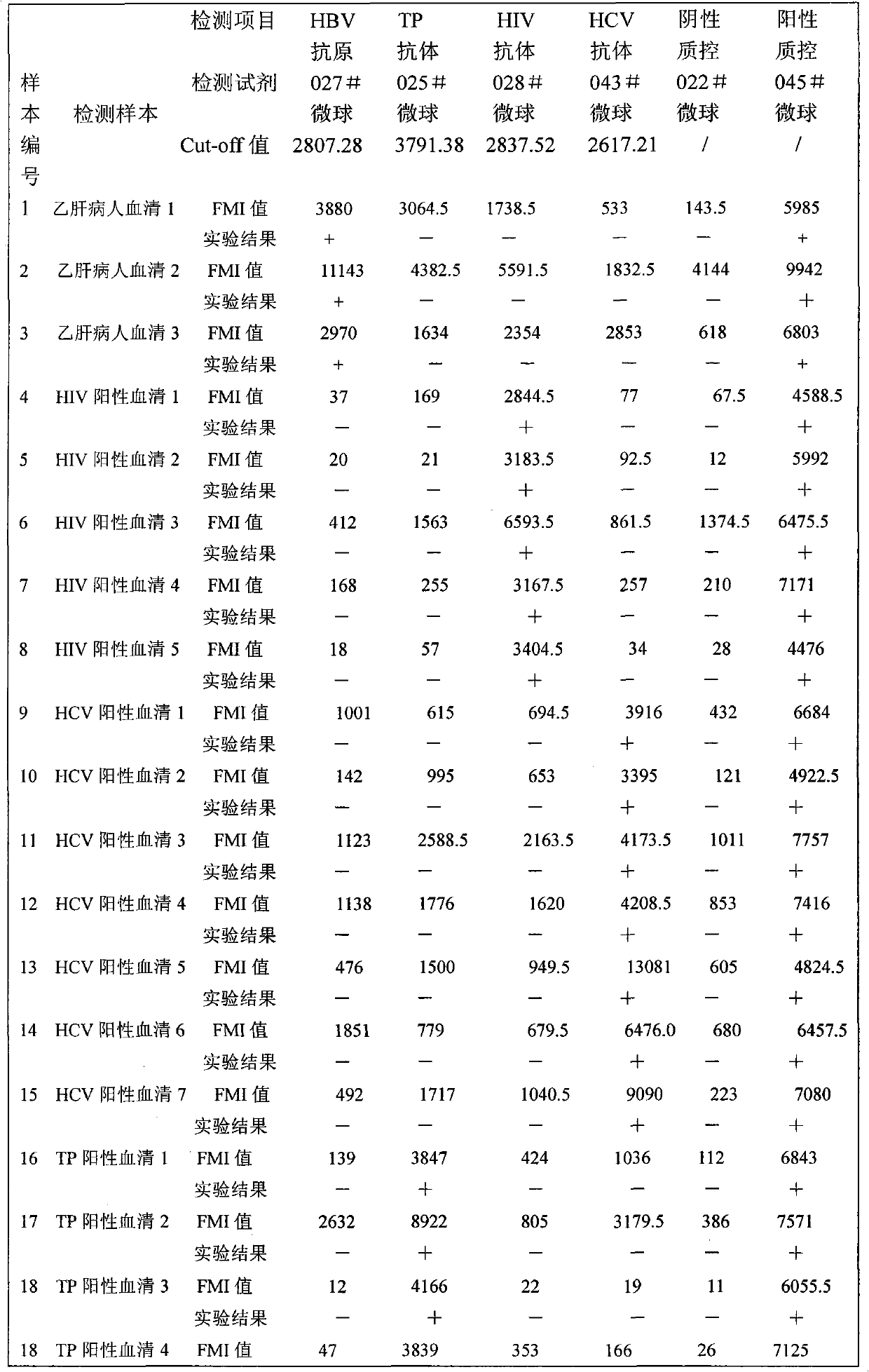
Suspension chip detection method for simultaneously detecting hepatitis B virus, hepatitis C virus, treponema pallidum and human immunodeficiency virus pathogens
The invention relates to a suspension chip detection method for simultaneously detecting hepatitis B virus, hepatitis C virus, treponema pallidum and human immunodeficiency virus pathogens. Plastic color coded microspheres with diameter of between 5.5 and 6.5 mu m are taken as vectors for a suspension chip; and the carriers are coated with monoclonal hepatitis B virus surface antibodies, HIV1, gP41 antigens, HCV NS3 antigens or treponema pallidum TP15 antigens. Through the suspension chip, the hepatitis B virus, hepatitis C virus, treponema pallidum and human immunodeficiency virus pathogens in a sample to be detected can be very conveniently and quickly detected at the same time; and the method has the advantages of accurate and reliable detection result, high specificity, small using amount of samples and high-throughput detection.
Owner:中华人民共和国陕西出入境检验检疫局
Hiv-1 antibodies
Owner:HAYNES BARTON F +4
Neutralizing gp41 antibodies and their use
Monoclonal neutralizing antibodies are disclosed that specifically bind to the HIV-1 gp41 membrane-proximal external region (MPER). Also disclosed are compositions including the disclosed antibodies that specifically bind gp41, nucleic acids encoding these antibodies, expression vectors including the nucleic acids, and isolated host cells that express the nucleic acids. The antibodies and compositions disclosed herein can be used for detecting the presence of HIV-1 in a biological sample, or detecting an HIV-1 infection or diagnosing AIDS in a subject. In additional, the broad neutralization breadth of the disclosed antibodies makes them ideal for treating a subject with an HIV infection. Thus, disclosed are methods of treating and / or preventing HIV infection.
Owner:US DEPT OF HEALTH & HUMAN SERVICES
HIV (human immunodeficiency virus) antibody recognition reagent
The invention relates to an HIV (human immunodeficiency virus) antibody recognition reagent. The HIV antibody recognition reagent comprises a piece of HIV antibody test paper, a washing solution, a developing system, a positive control substance, a negative control substance and an immunochromatographic result interpretoscope; the HIV antibody test paper is coated with 10 protein stripes including a HIV1 gp160 stripe, a gp120 stripe, a P66 stripe, a p51 stripe, a gp41 stripe, a p24 stripe, a p17 stripe, a HIV2 gp36 stripe and two human IgG (immunoglobulin G) stripes with different concentrations; and the developing system comprises a collaurum or biotin-labeled anti-human IgG or SPA and a biotin-labeled avidin. During testing, a sample is put on the HIV antibody test paper, unrelated proteins are removed by washing, the stripes are developed by using the developing system, and the result is judged with the immunochromatographic result interpretoscope or naked eyes according to the developing condition of the stripes. The HIV antibody recognition reagent provided by the invention is easy and convenient to operate, is suitable for field testing and is economic, practical and the like.
Owner:齐明山 +1
Stabilized soluble glycoprotein trimers
InactiveUS20050220817A1Stabilize trimerSimplifies isolationAntibody mimetics/scaffoldsVirus peptidesHiv envelopeGp41
The present application is directed to stabilized HIV envelope glycoprotein trimers. The trimers are stabilized by introducing trimeric motifs, preferably the GCN4 coiled coil or the fibritin trimeric domain, at certain sites, for example in the gp41 ectodomain. These stabilized trimers or DNA molecules encoding such trimers can be used to generate an immunogenic reaction. The trimers can also be used in assays to screen for molecules that interact with them—and to identify molecules that interact with specific sites.
Owner:DANA FARBER CANCER INST INC
HIV vaccine immunogens and immunization strategies to elicit broadly-neutralizing anti-HIV-1 antibodies against the membrane proximal domain of HIV GP41
This invention relates to novel peptide immunogens that generate an immune response in mammals against HIV gp41, to pharmaceutical compositions that comprise such immunogens, and to methods of treating Immunodeficiency disease, especially HIV infection and AIDS, that employ such pharmaceutical compositions.
Owner:US DEPT OF HEALTH & HUMAN SERVICES