Patents
Literature
1672results about "Transvascular endocardial electrodes" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
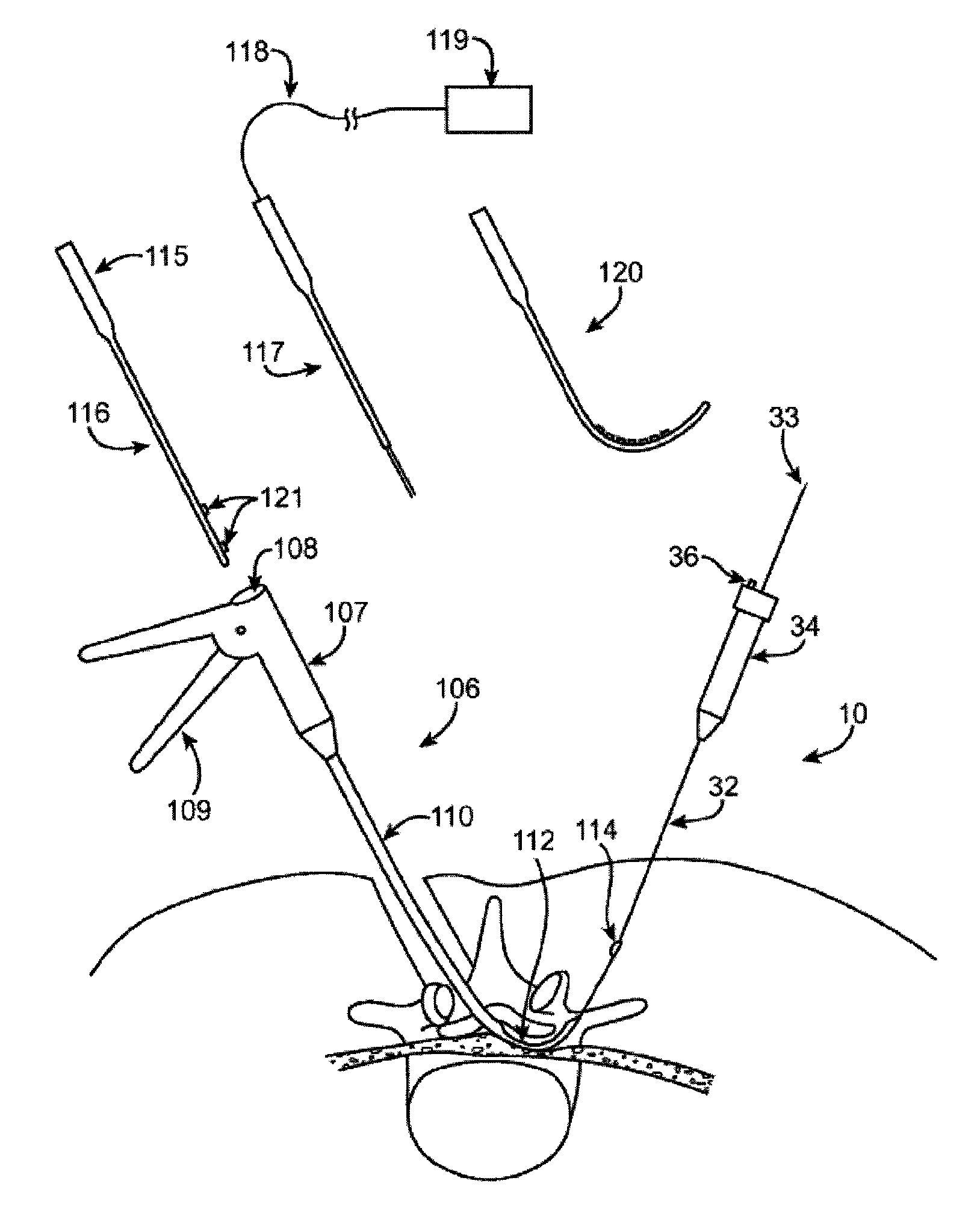
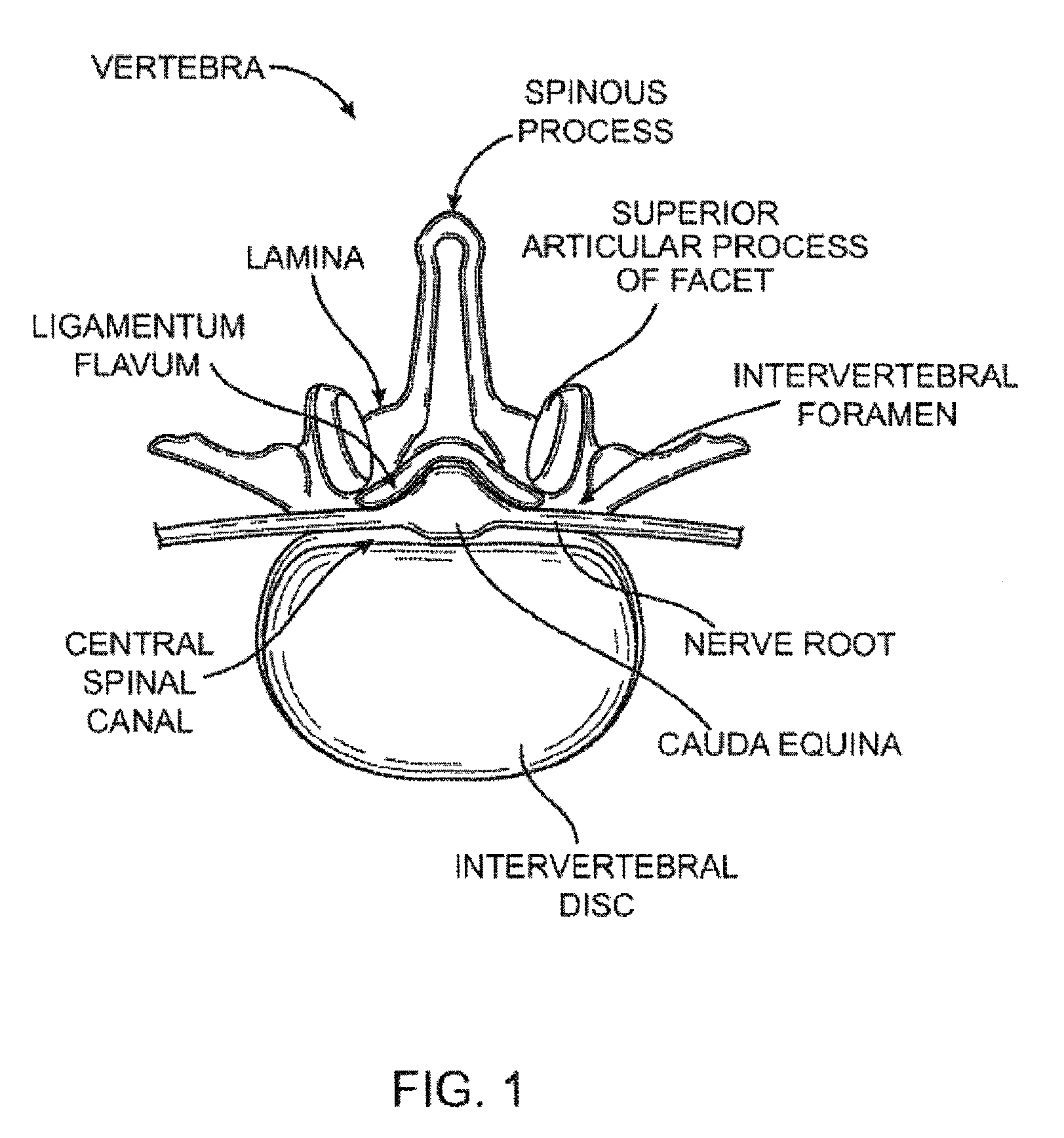
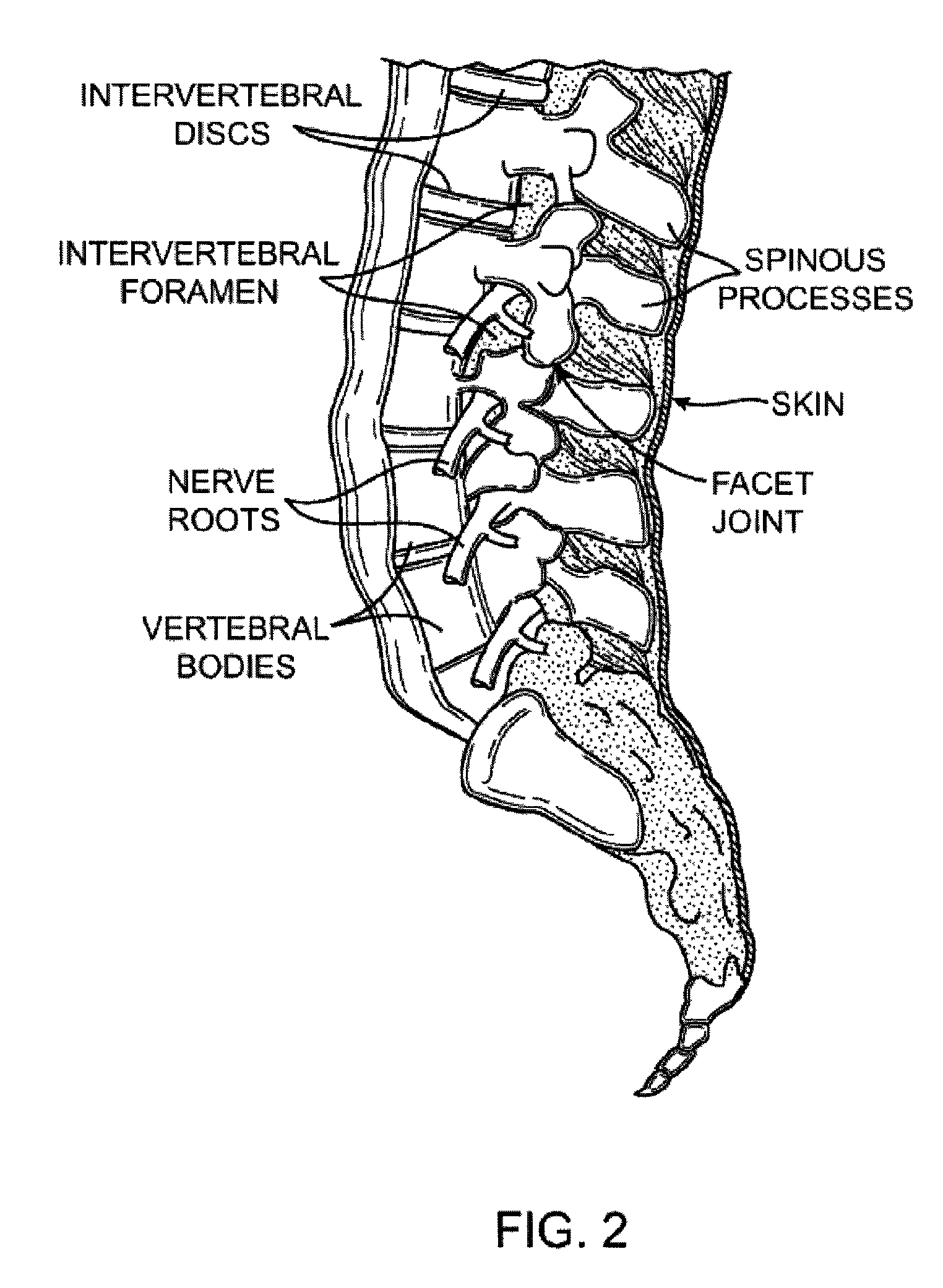
Guidewire exchange systems to treat spinal stenosis
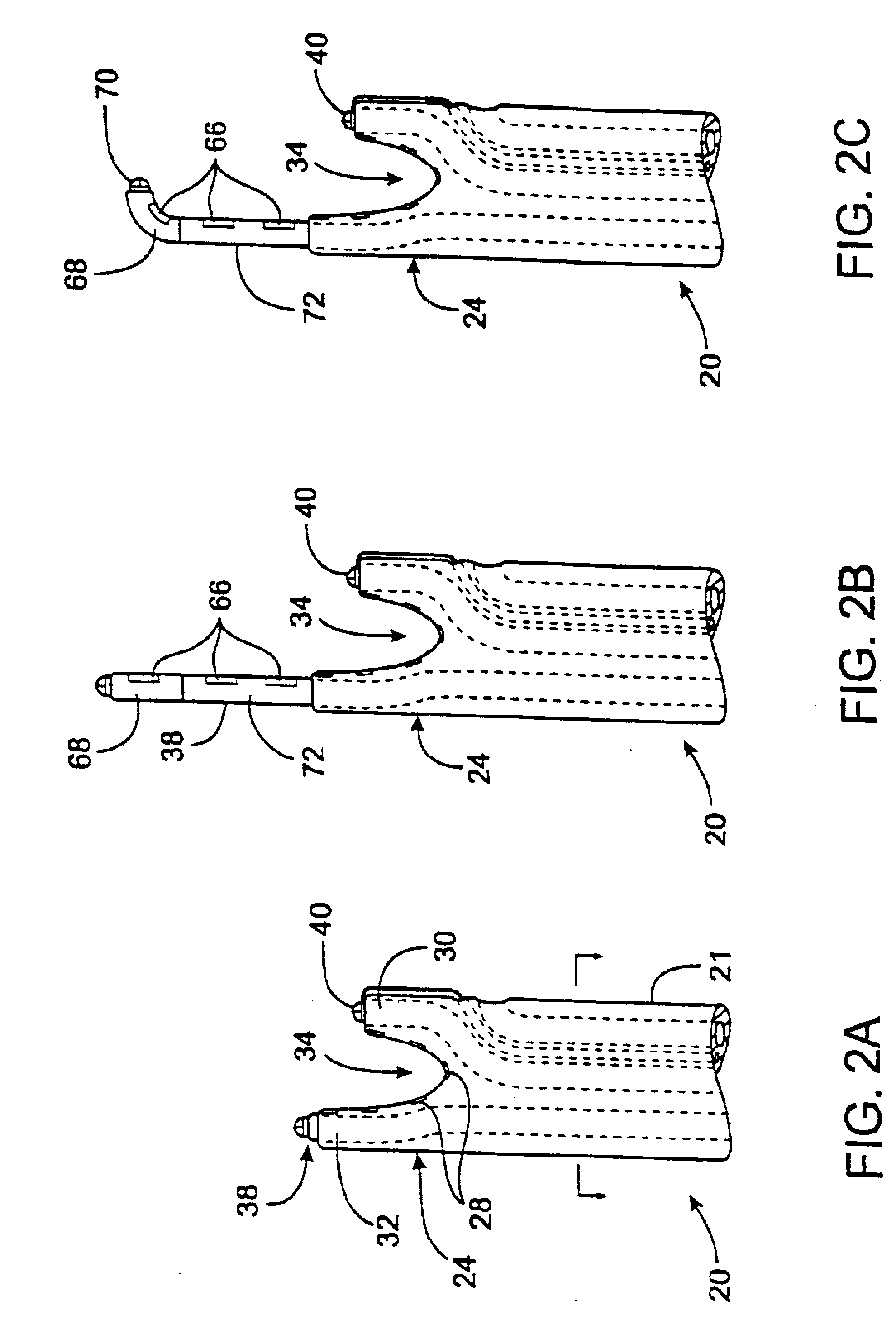
Guidewire exchange systems, devices and methods, for positioning and actuating surgical devices in a desired position between two tissues in a patient's body are described. A guidewire may be coupled to a surgical device for positioning and actuating (e.g., urging against a target tissue). The guidewire may be exchanged between different surgical devices during the same procedure, and the guidewire and surgical devices may be releaseably or permanently coupled. The surgical device generally includes one or more guidewire coupling members. A system may include a guidewire and a surgical device having a guidewire coupling member. Methods, devices and systems may be used in open, less-invasive or percutaneous surgical procedures.
Owner:SPINAL ELEMENTS INC +1
Body implantable lead including one or more conductive polymer electrodes and methods for fabricating same
InactiveUS6999821B2Transvascular endocardial electrodesDiagnostic recording/measuringElectrical conductorCoronary sinus
A body implantable lead comprises a lead body including a conductive polymer electrode disposed along a distal end portion of the lead body for performing one or more of the functions consisting of pacing, sensing, cardioversion and defibrillation. An electrical conductor, preferably in the form of a multistrand cable conductor, couples the conductive polymer electrode with a proximal end of the lead body. The conductive polymer electrode encapsulates the conductor and is in electrical contact therewith along the length, and preferably along substantially the entire length, of the conductive polymer electrode. The lead body may comprise a multilumen polymer housing, the conductor being contained within one of the lumens of the housing. The conductive polymer electrode may be disposed within a window formed in the lead body. Alternatively, the conductive polymer electrode may comprise multiple electrode sections within a corresponding number of windows formed in the lead body and spaced apart along the length thereof. Further, the window and the conductive polymer electrode disposed therein may extend helically about the lead body. Because of its flexibility and because it can have a small diameter, the lead of the invention is particularly advantageous for implantation in the small, tortuous vessels of the coronary sinus region of the heart for left side stimulation and / or sensing.Methods of fabricating lead bodies incorporating conductive polymer electrodes are also disclosed.
Owner:PACESETTER INC
Rotatable lead introducer
Minimally invasive introducers and methods that can be used for rotationally securing devices within the human body. Introducers can include a distal element for releasably engaging a lead head controllable from a proximal control located outside of the body. An inner stem can extend between a proximal portion and a distal portion, and be pivotally and rotatably coupled to the distal lead engagement mechanism. An outer tube can be rotatably disposed over the inner stem and be flexibly coupled over the pivot to rotationally drive the distal element. A helical epicardial-myocardial lead electrode can be secured and oriented straight ahead and introduced through a port or small incision with the introducer in a straight configuration. The introducer can then be bent and rotated to screw the helical electrode into the heart.
Owner:WILSON GREATBATCH LTD
Devices and methods for transluminal or transthoracic interstitial electrode placement
ActiveUS7191015B2Epicardial electrodesTransvascular endocardial electrodesElectrode placementDevice implant
Methods and devices for implanting pacing electrodes or other apparatus, or for delivering substances, to the heart of other tissues within the body. A guided tissue penetrating catheter is inserted into a body lumen (e.g., blood vessel) or into a body cavity or space (e.g., the pericardial space) and a penetrator is advanced from the catheter to a target location. In some embodiments, a substance or an apparatus (such as an electrode) may be delivered through a lumen in the penetrator. In other embodiments, a guidewire may be advanced through the penetrator, the penetrating catheter may then be removed and an apparatus (e.g., electrode) may then be advanced over that guidewire. Also disclosed are various implantable electrodes and electrode anchoring apparatus.
Owner:MEDTRONIC VASCULAR INC
Apparatus and method for fixing electrodes in a blood vessel
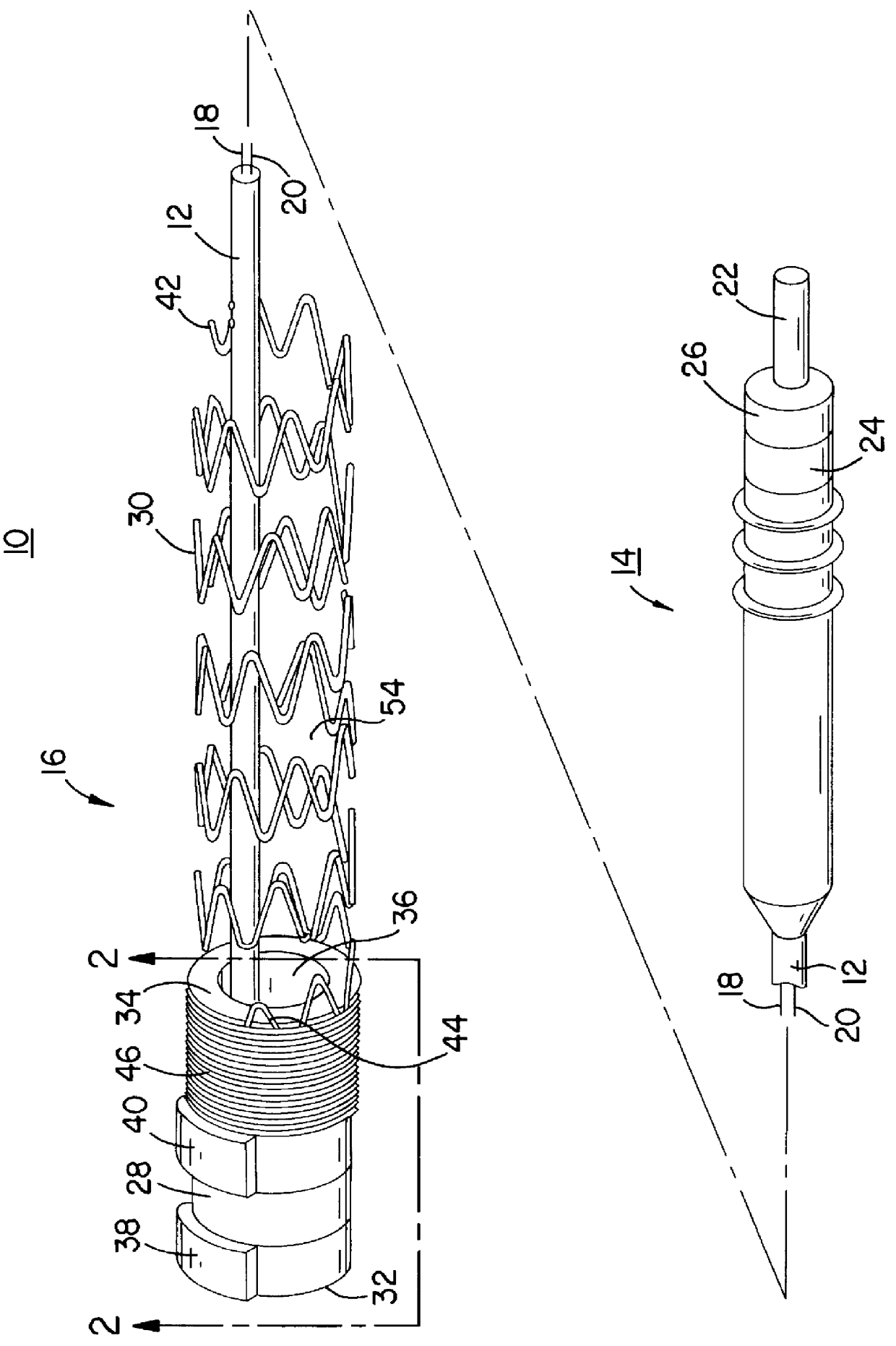
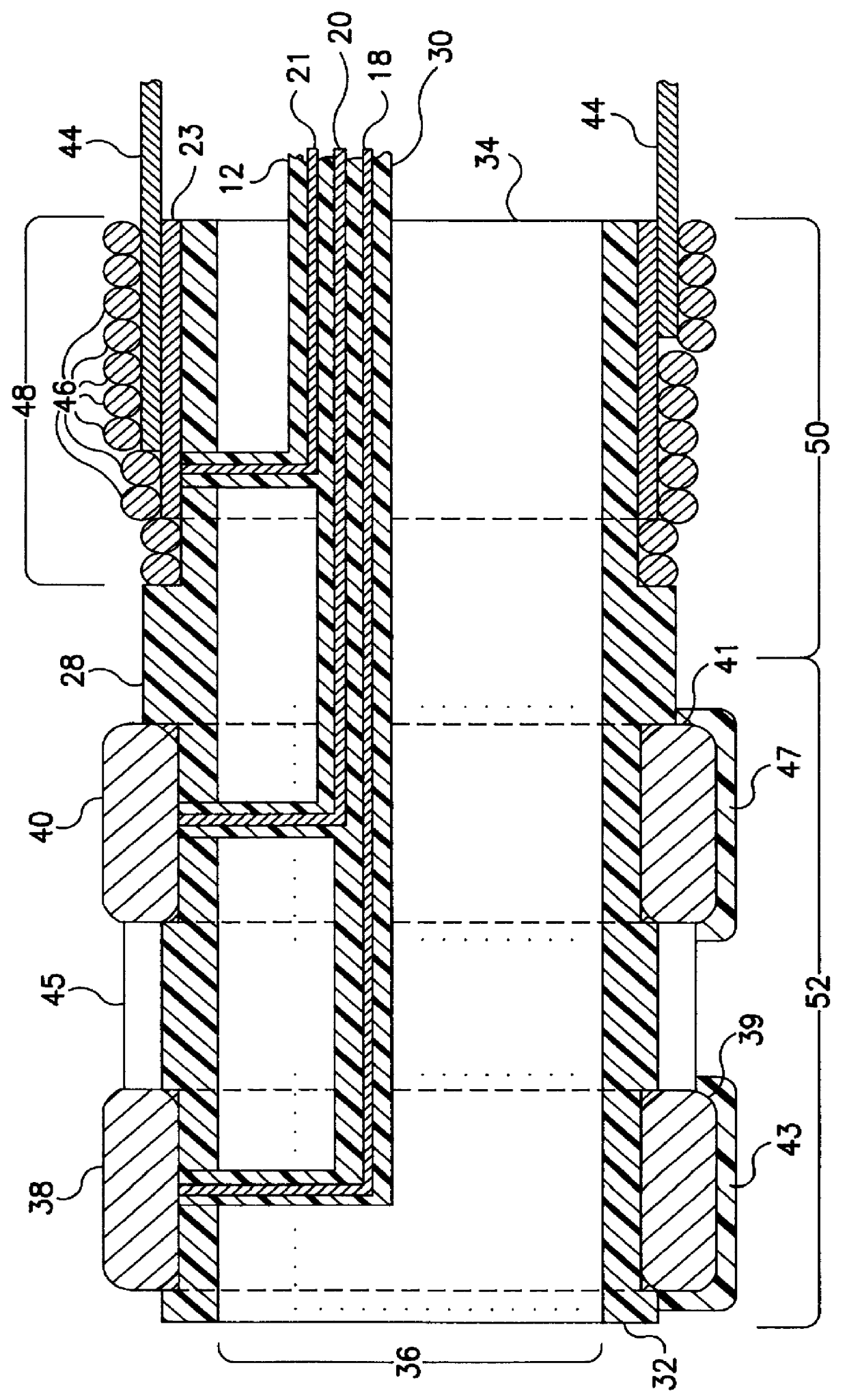
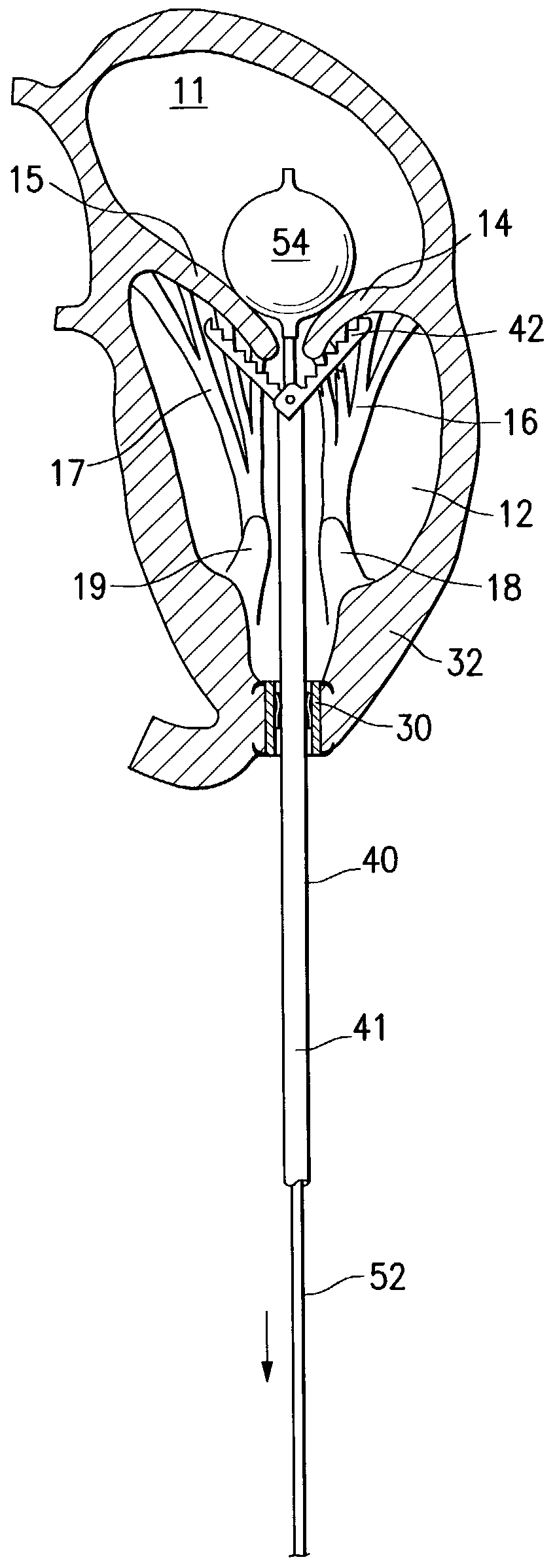
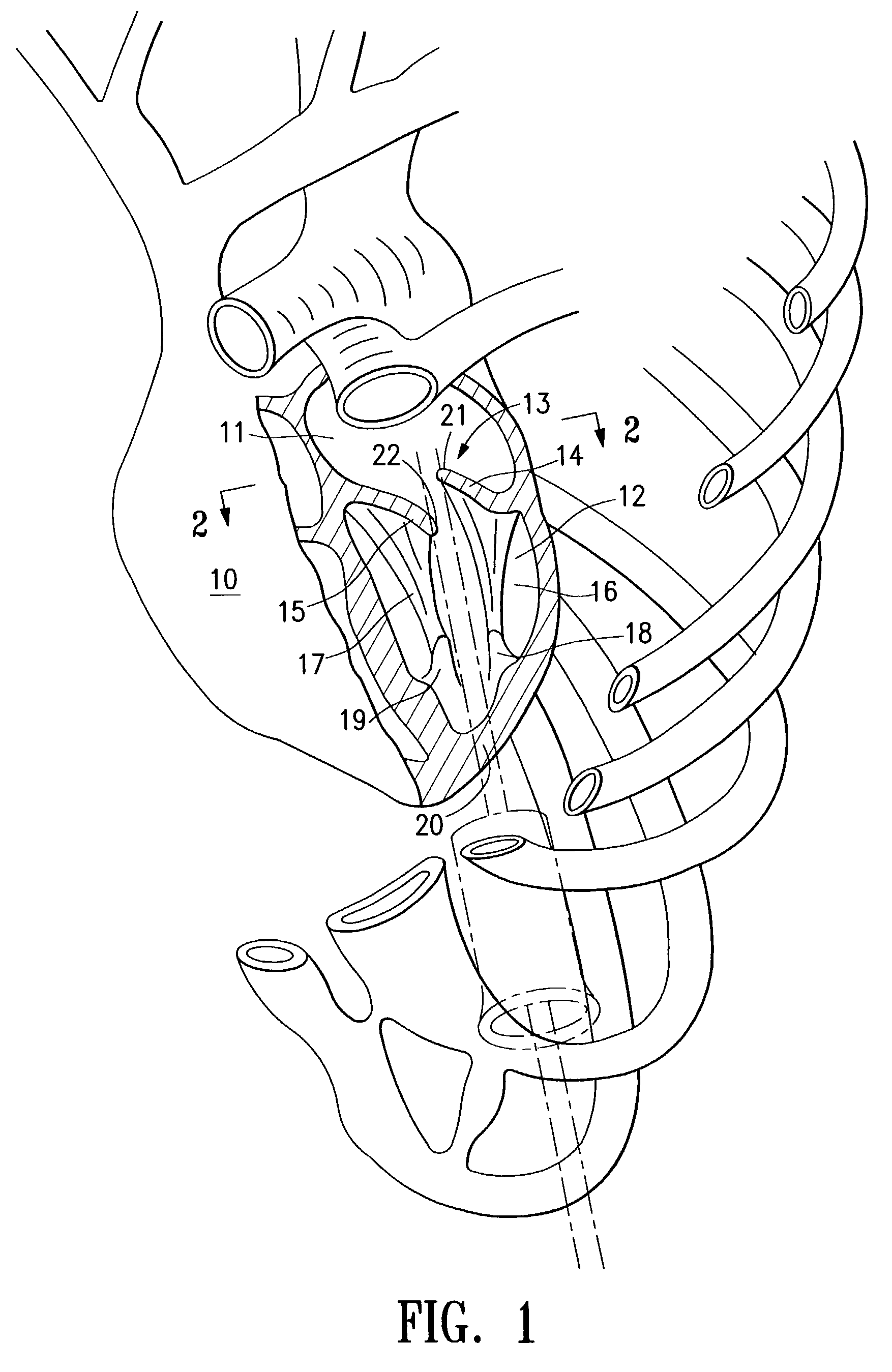
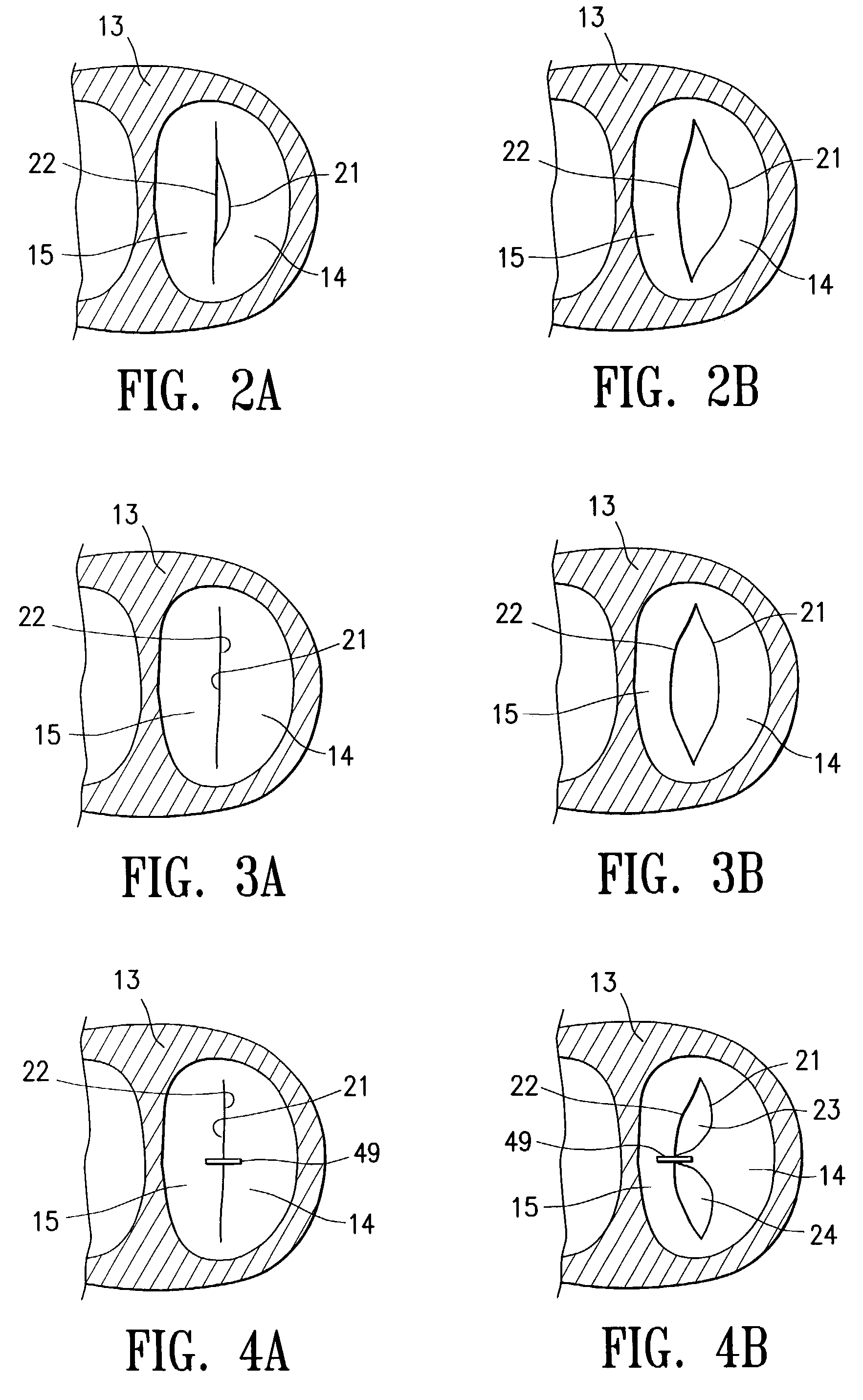
InactiveUS6161029ASimple equipmentElectrocardiographyTransvascular endocardial electrodesBlood vessel wallsLeft Ventricles
Endocardial implantable cardiac leads are disclosed for applying electrical stimulation to and / or sensing electrical activity of the heart at one or more distal electrode positioned at a cardiac implantation site within a cardiac vessel adjacent to and at a desired orientation to the left ventricle or atrium of the heart. The distal electrode(s) is supported by a tubular electrode support having a diameter large enough to bear against the blood vessel wall and a support lumen that allows blood to flow through it. A retention stent extends proximally from a distal stent end fixed to the tubular electrode support to a proximal stent end. After advancement to the cardiac implantation site employing a lead delivery mechanism, the retention stent is expandable from a collapsed stent state in which the outer diameter of the retention stent is less than the inner diameter of the vessel to an expanded stent state. The expanded stent is lodged against the blood vessel wall and inhibits movement of the stent and distal electrode support. The expanded stent lumen is aligned with the electrode support lumen for allowing blood to flow through the aligned electrode support lumen and expanded stent lumen. The retention stent may take any of the known forms that can be introduced in the collapsed stent state within an introducer lumen or mounted to an introducer and can either self-expand upon release in the blood vessel or be mechanically expanded within the blood vessel.
Owner:MEDTRONIC INC
High-sensitivity pressure-sensing probe
ActiveUS20090138007A1Accurate inductionTransvascular endocardial electrodesEndoscopesMagnetic transducersSacroiliac joint
A medical probe includes an insertion tube, having a longitudinal axis and having a distal end. A distal tip is disposed at the distal end of the insertion tube and is configured to be brought into contact with a body tissue. A joint couples the distal tip to the distal end of the insertion tube. A joint sensor, contained within the probe, senses a position of the distal tip relative to the distal end of the insertion tube. The joint sensor includes first and second subassemblies, which are disposed within the probe on opposite, respective sides of the joint and each include one or more magnetic transducers.
Owner:BIOSENSE WEBSTER (ISRAEL) LTD
Access and tissue modification systems and methods
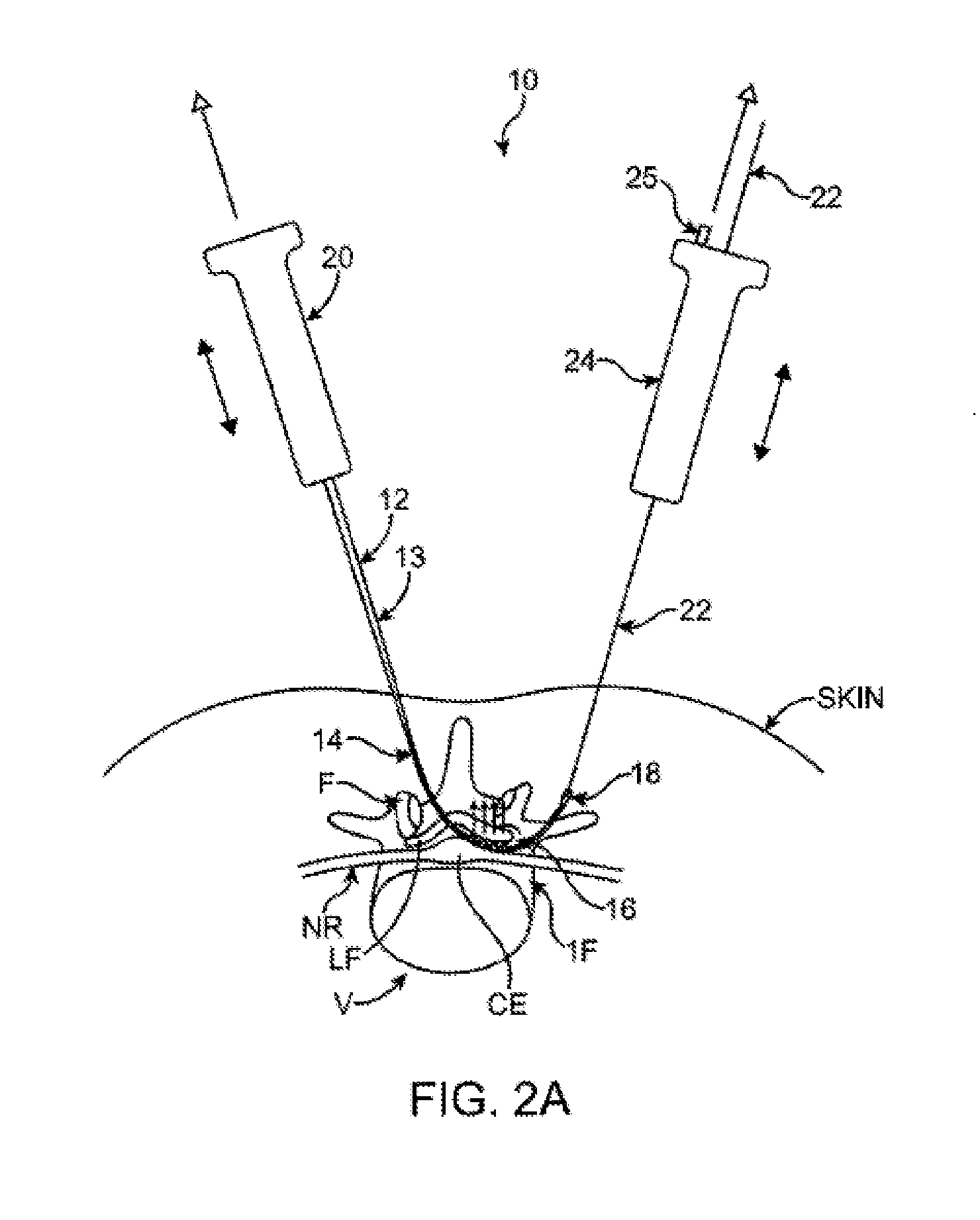
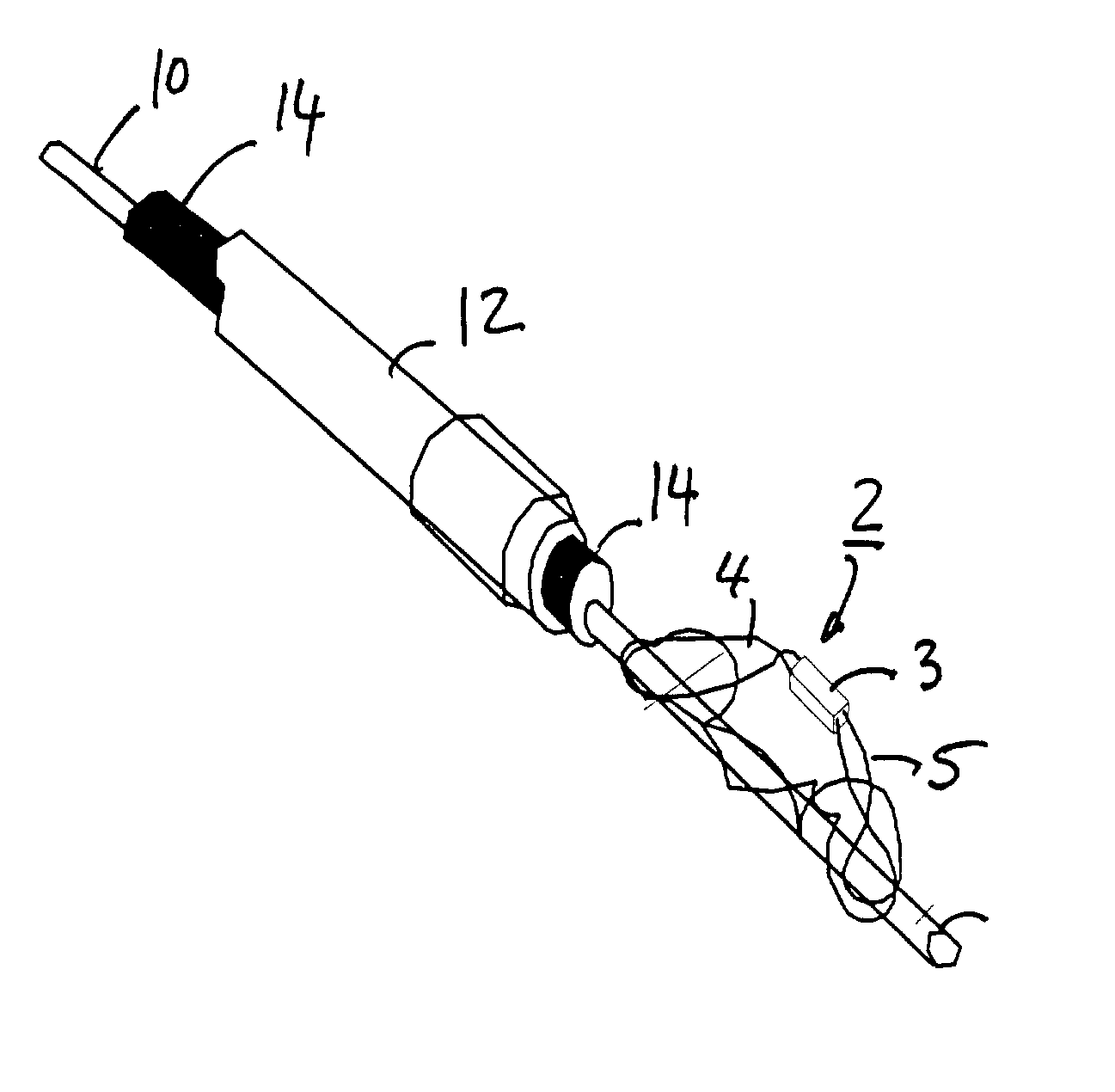
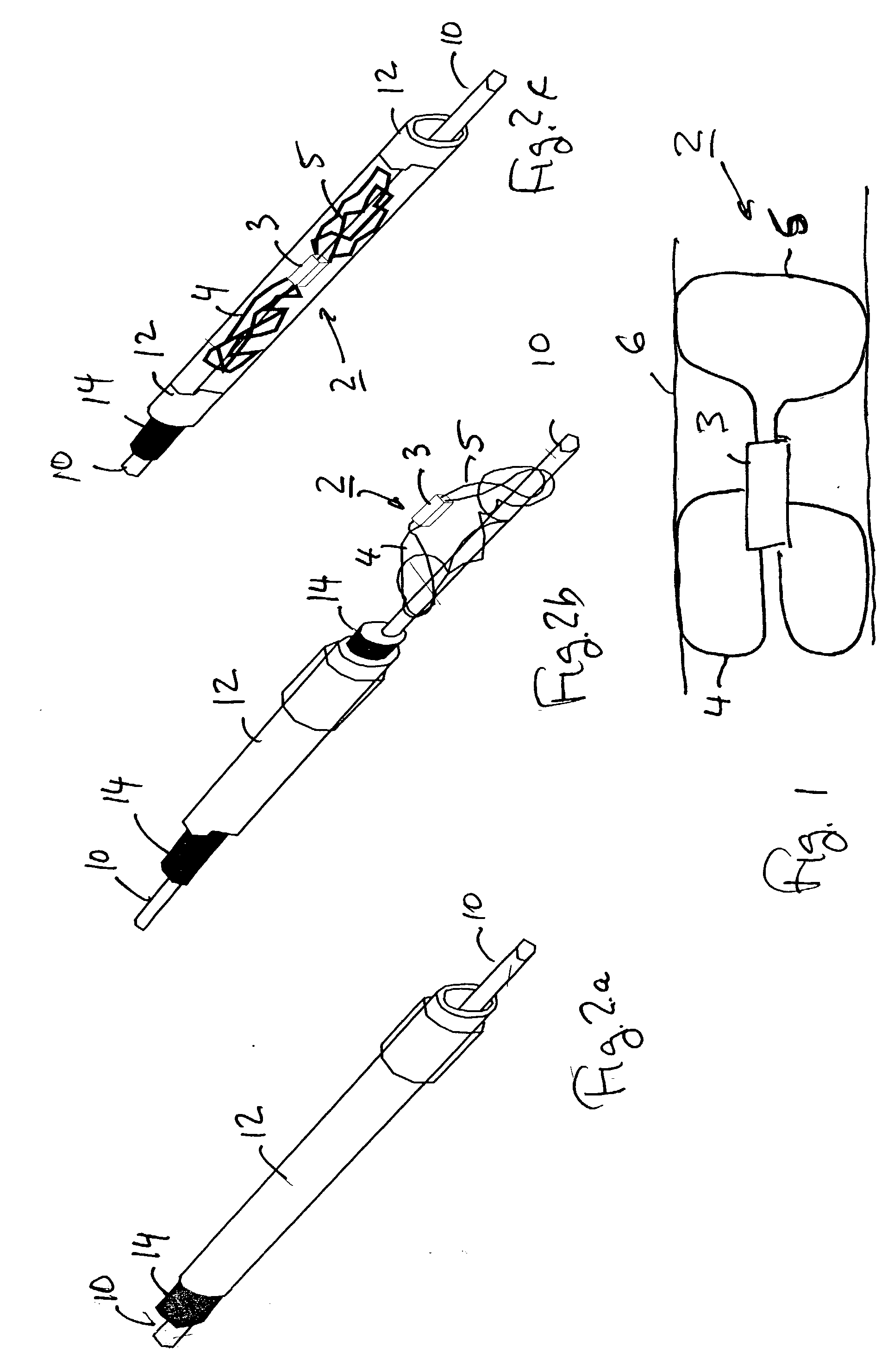
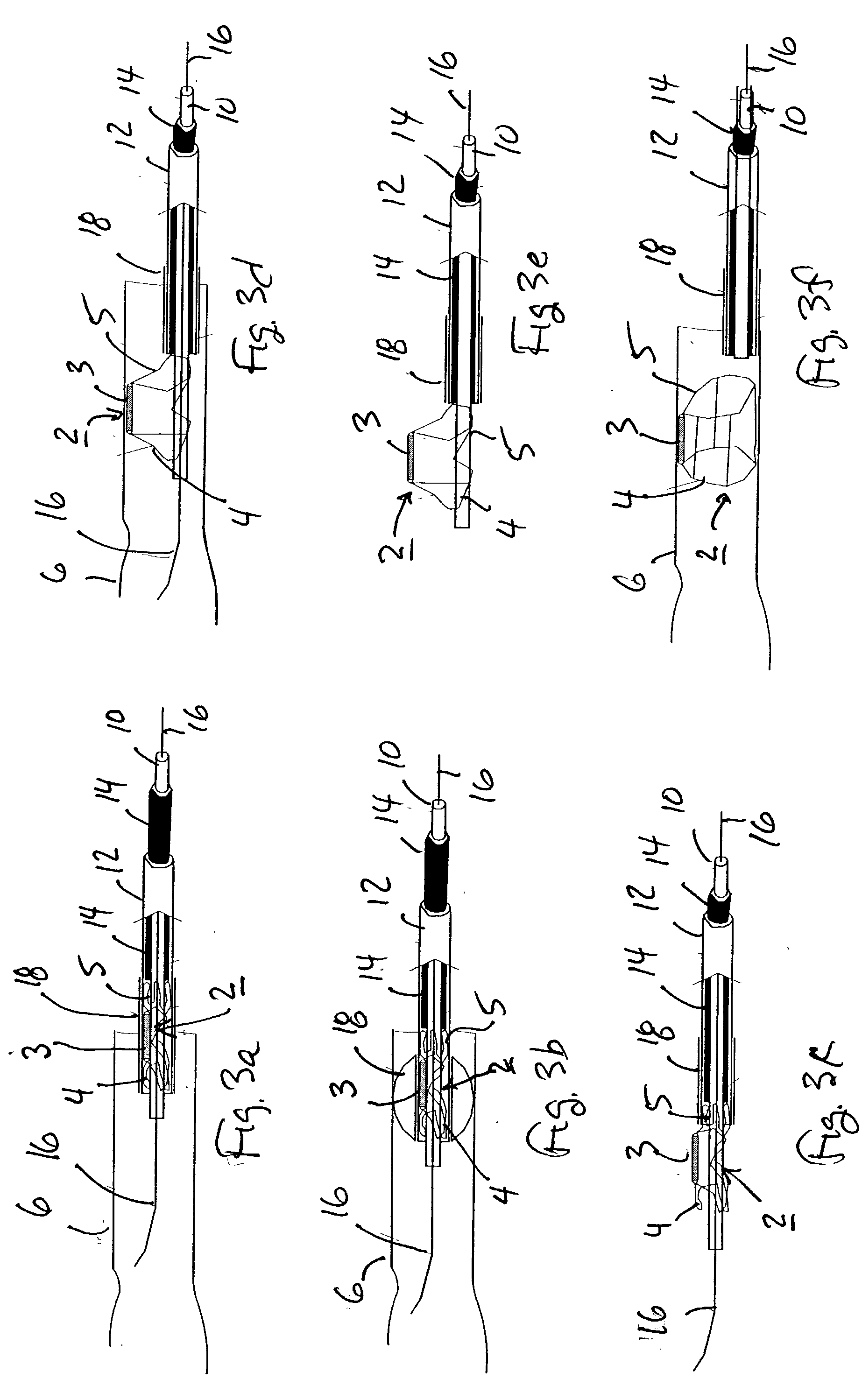
Described herein are methods and systems for precisely placing and / or manipulating devices within the body by first positioning a guidewire or pullwire through the body from a first location, around a curved pathway, and out of the body through a second location, so that the distal and proximal ends of the guidewire extend from the body, then pulling a device into position using the guidewire. The device to be positioned within the body is coupled to the proximal end of the guidewire, and the device is pulled into the body by pulling on the distal end of the guidewire that extends from the body. The device may be bimanually manipulated by pulling the guidewire distally, and an attachment to the device that extends proximally, allowing control of both the proximal and the distal ends. In this manner devices (and particularly implants such as innerspinous distracters, stimulating leads, and disc slings) may be positioned and / or manipulated within the body. Devices to modify tissue may also be positioned or manipulated so that a target tissue within the body is modified.
Owner:BAXANO
Deployment device, system and method for medical implantation
A deployment device for deploying a self-expansible medical implant at a target location in a body cavity, is provided. While generally, the expansion of self-expansible structures tends to be abrupt, and the impact of expansion may cause injury, a two-stage expansion process of the present invention minimizes the impact of exapnsion. Additionally, an ability to manuever the medical imlant into position, after the first stage of expansion, provides for accurate positioning. Thus the present invention is of a deployment device for precise and well-controlled manner of deployment, so as to minimize damage to the cavity wall and to position the implant accurately at the target location. The deployment device includes: an inner tube; an outer tube; and an implant received on the inner tube and enclosed by the outer tube. The implant has a self-expansible anchoring element which is in a contracted condition when enclosed by the outer tube, expands to a partially-expanded condition when the outer tube is retracted, and expands to a fully-expanded condition when the inner tube is removed.. The implant is deployed by introducing the deployment device to the target location in the body cavity; retracting the outer tube with respect to the implant such that the anchoring element self-expands from its contracted condition to its partially-expanded condition; and withdrawing the inner tube from the implant such that the anchoring element self-expands from its partially-expanded condition to its fully-expanded condition to firmly fix the implant at the target location within the body cavity.
Owner:REMON MEDICAL TECH
Method and apparatus for shunting induced currents in an electrical lead
InactiveUS20030083726A1Transvascular endocardial electrodesHeart defibrillatorsElectricityPower flow
An electrical lead includes an elongate body having a proximal end portion and a distal end portion, a first electrode disposed adjacent and joined to the distal end portion of the elongate body. Current flow within the first electrode is limited when a predetermined condition occurs, such as the generation of an electromagnetic field having a predetermined frequency range. The medical electrical lead may further comprise one or more second electrodes disposed adjacent the first electrode and joined to the elongate body to shunt current to body tissue when the predetermined condition occurs.
Owner:MEDTRONIC INC
Method and device for electronically controlling the beating of a heart using venous electrical stimulation of nerve fibers
An electro-stimulation device includes a pair of electrodes for connection to at least one location in the body that affects or regulates the heartbeat. The electro-stimulation device both electrically arrests the heartbeat and stimulates the heartbeat. A pair of electrodes are provided for connection to at least one location in the body that affects or regulates the heartbeat. The pair of electrodes may be connected to an intravenous catheter for transvenous stimulation of the appropriate nerve. A first switch is connected between a power supply and the electrodes for selectively supplying current from the power supply to the electrodes to augment any natural stimuli to the heart and thereby stop the heart from beating. A second switch is connected between the power supply and the electrodes for selectively supplying current from the power supply to the electrodes to provide an artificial stimulus to initiate heartbeating. In another aspect, the invention is directed to a method for arresting the beat of a heart in a living body comprising the steps of connecting the pair of electrodes to at least one location in the body that affects or regulates the heartbeat and supplying an electrical current to the electrodes of sufficient amplitude and duration to arrest the heartbeat. The device may also serve to still the lungs by input to a respirator or by stimulation of the phrenic nerve during surgical procedures.
Owner:MEDTRONIC INC
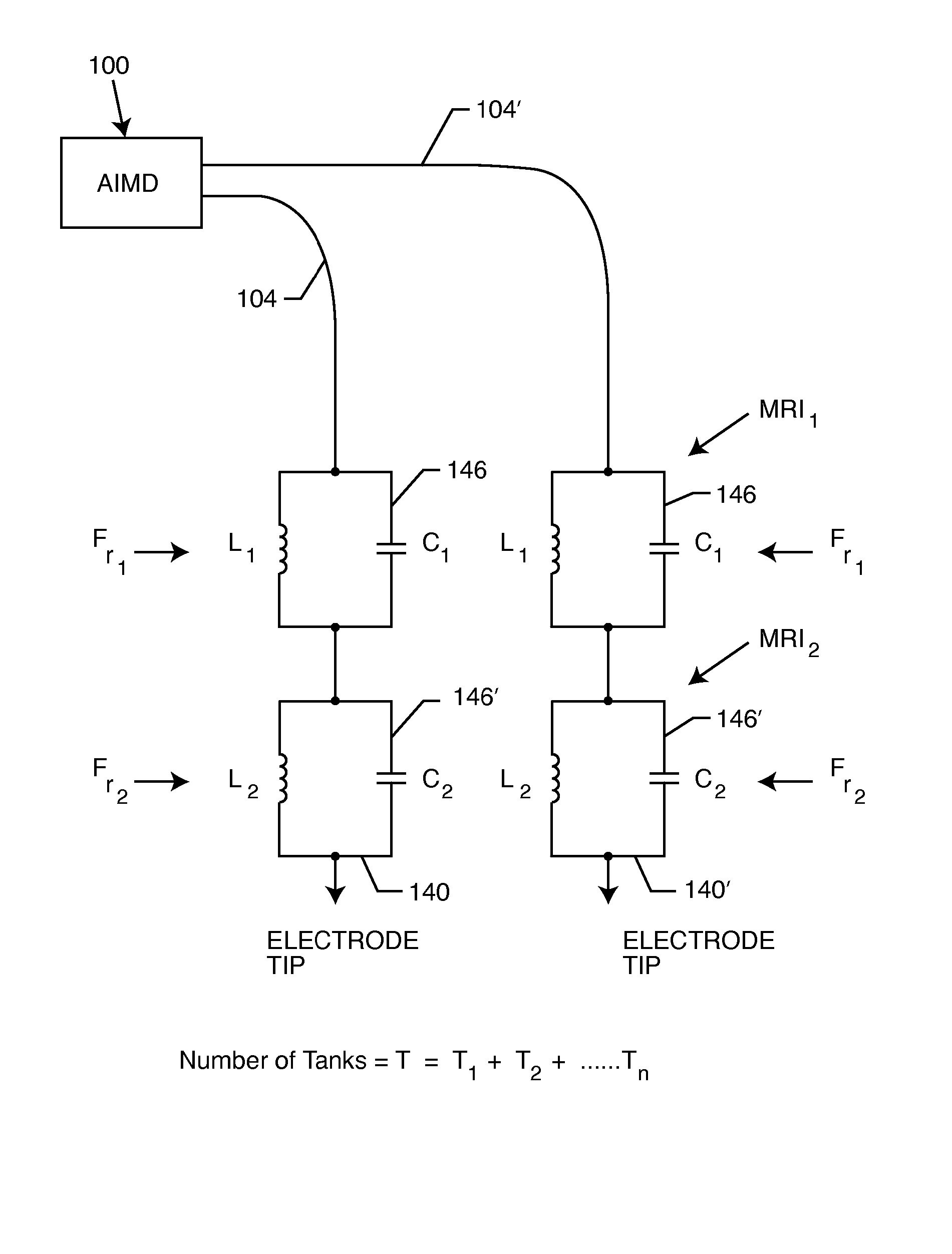
Non-ferromagnetic tank filters in lead wires of active implantable medical devices to enhance MRI compatibility
InactiveUS20080049376A1High impedanceMuch smaller and volumetrically efficientMultiple-port networksAnti-noise capacitorsCapacitanceEngineering
A TANK filter is provided for a lead wire of an active medical device (AMD). The TANK filter includes a capacitor in parallel with an inductor. The parallel capacitor and inductor are placed in series with the lead wire of the AMD, wherein values of capacitance and inductance are selected such that the TANK filter is resonant at a selected frequency. In a preferred form, the TANK filter reduces or even eliminates the use of ferro-magnetic materials, and instead uses non-ferromagnetic materials so as to reduce or eliminate MRI image artifacts or the force or torque otherwise associated during an MRI image scan.
Owner:WILSON GREATBATCH LTD
Cardiovascular anchoring device and method of deploying same
InactiveUS7149587B2Efficient deploymentElectrocardiographyTransvascular endocardial electrodesMeasurement deviceOrgan wall
An anchoring device and a delivery method thereof can effectively provide a means for securing an implant to a wall of an internal organ within a patient in a variety of clinical applications. In one embodiment, an anchoring device used to retain a cardiac pressure measurement device is provided. The device is implanted in the body by deforming it to a small cross section profile, sliding it through a low profile delivery device and ejecting from the delivery device at a targeted site. The anchoring mechanism, when ejected from the delivery device, reverts back to pre-formed configuration and engages opposite sides of an organ wall, thereby anchoring the implant in the organ wall.
Owner:PACESETTER INC
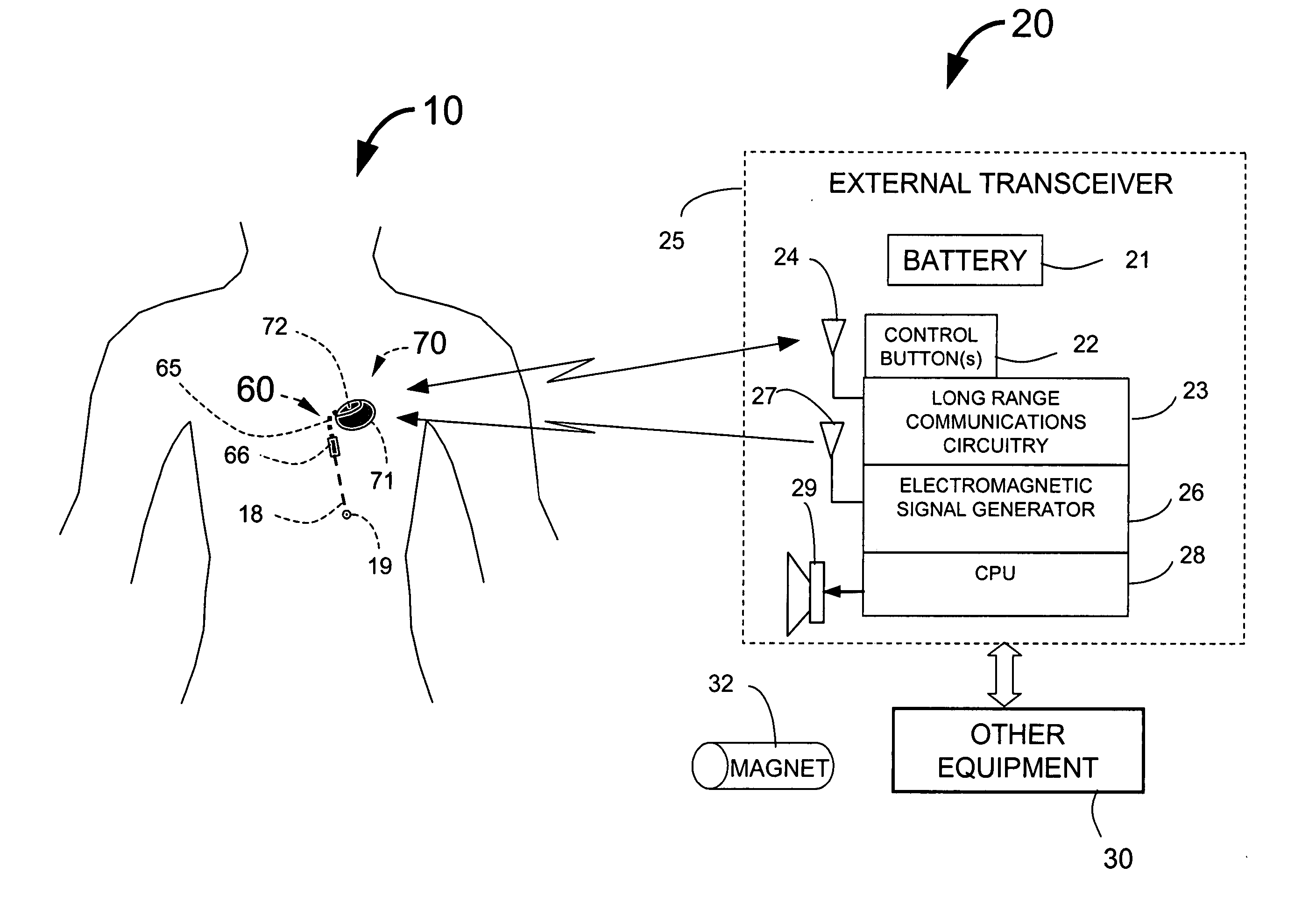
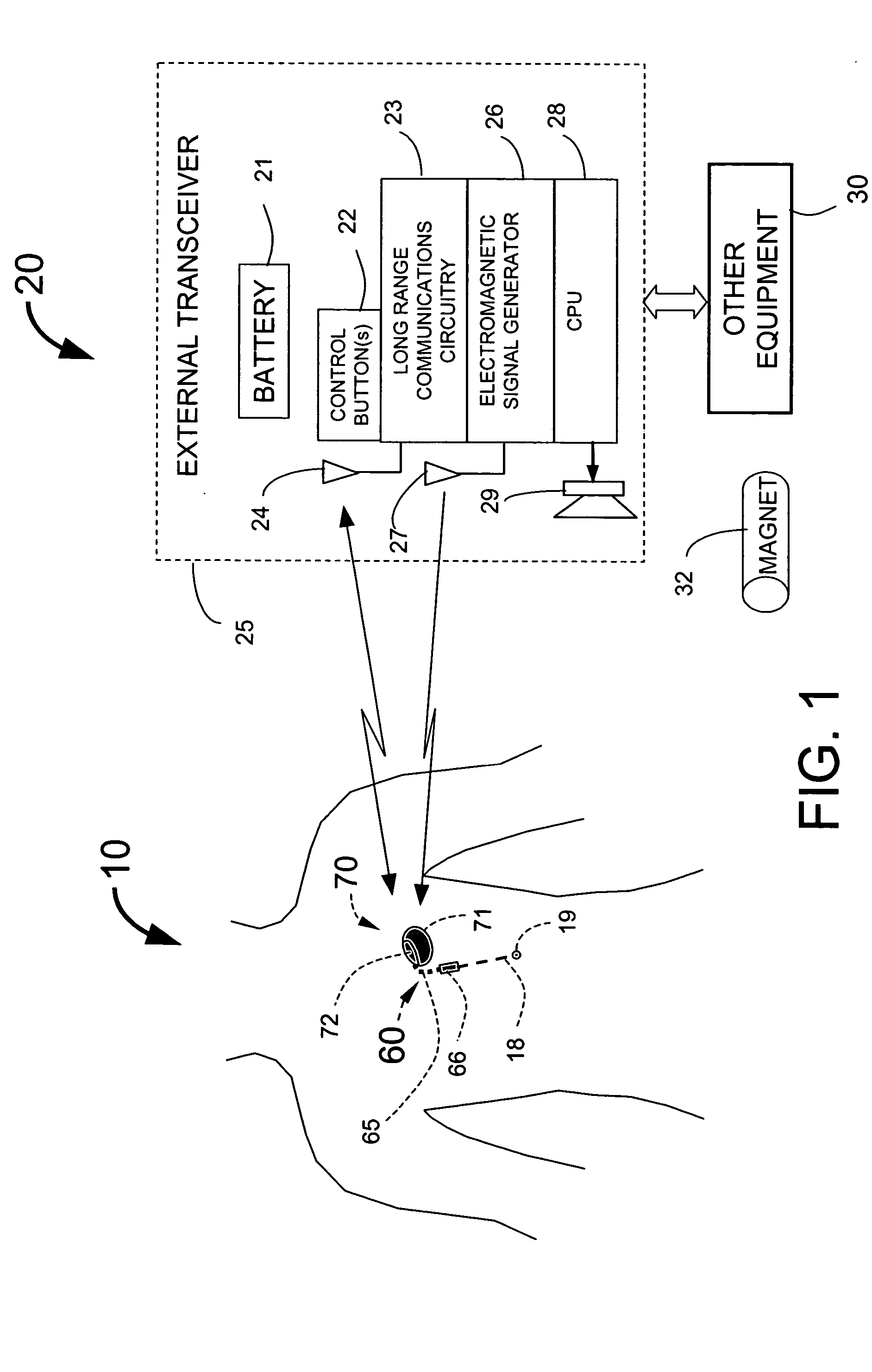
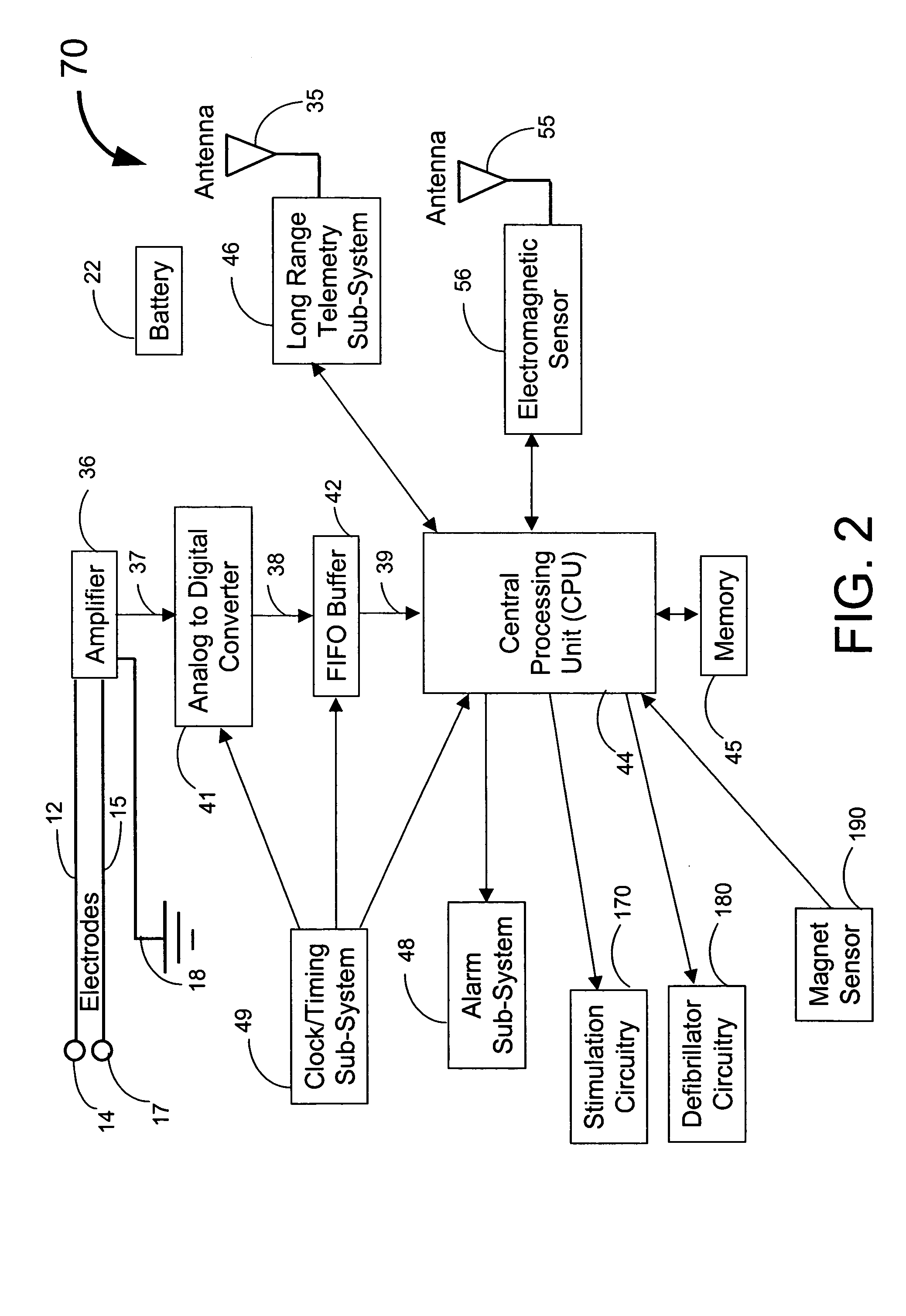
Implantable medical system with long range telemetry
InactiveUS20050113886A1Minimize and eliminate chanceStrong enoughTransvascular endocardial electrodesExternal electrodesImplanted deviceTelemetry Equipment
An implantable medical system for implantation within the body of a patient is provided. The system includes an implanted device having an implant casing and a long range telemetry sub-system housed therein. The system also includes an implantable lead operationally coupled to the implanted device and an antenna coupled to the implant casing to extend therefrom. The antenna is operationally coupled to the long-range telemetry sub-system to enable wireless bi-directional communication between the long range telemetry sub-system and predetermined external equipment disposed outside the body of the patient.
Owner:ANGEL MEDICAL SYST +1
Implant delivery catheter system and methods for its use
Catheter systems and methods for implanting helical or dart-like implants into the myocardium or other body tissue. The catheter system includes a helix for fixing the distal end of the catheter to the myocardium, an implant held by the helix, mechanisms for driving the fixation helix into the myocardium, and mechanisms for driving the implant into the myocardium, removing the fixation helix and leaving the implant behind. The implant may be coated, filled, or made of a drug or drug eluting compound, or drug delivery matrix of any composition.
Owner:BIOCARDIA
Steerable catheter with preformed distal shape and method for use
InactiveUS6096036AReduce curvatureTransvascular endocardial electrodesSurgical instrument detailsHeart chamberCurve shape
A catheter has a stylet formed of a shape-retentive and resilient material having a preformed curved shape at its distal end resulting in the catheter sheath having the preformed curved shape. The catheter sheath has a plurality of electrodes at its distal end for contacting selected biological tissue for imparting ablation energy thereto. The catheter sheath also has an axially mounted tendon for causing deflection of the distal end. The stylet material permits straightening the catheter sheath during insertion into the patient and advancing the electrodes to the target tissue. Upon removal of the straightening forces, such as by entry into a chamber of the heart, the stylet material resumes its preformed curved distal shape thereby forcing the catheter distal end with the electrodes into the same preformed curved shape. The operator may place the curved distal end into contact with the target tissue and axially move the tendon as desired to gain greater control over the bend in the distal end of the catheter sheath to adjust the radius of curvature of the distal end to obtain greater contact of the electrodes with the heart tissue. Preferably, the stylet is formed of nitinol.
Owner:CARDIAC PACEMAKERS INC
Pacemaker Retrieval Systems and Methods
A catheter system for retrieving a leadless cardiac pacemaker from a patient is provided. The cardiac pacemaker can include a docking or retrieval feature configured to be grasped by the catheter system. In some embodiments, the retrieval catheter can include a snare configured to engage the retrieval feature of the pacemaker. The retrieval catheter can include a torque shaft selectively connectable to a docking cap and be configured to apply rotational torque to a pacemaker to be retrieved. Methods of delivering the leadless cardiac pacemaker with the delivery system are also provided.
Owner:PACESETTER INC
RFID detection and identification system for implantable medical lead systems
A system for identifying active implantable medical devices (AIMD) and lead systems implanted in a patient using a radio frequency identification (RFID) tag having retrievable information relating to the AIMD, lead system and / or patient. The RFID tag may store information about the AIMD manufacturer, model number, serial number; lead wire system placement information and manufacturer information; MRI compatibility due to the incorporation of bandstop filters; patient information, and physician and / or hospital information and other relevant information. The RFID tag may be affixed or disposed within the AIMD or lead wires of the lead system, or surgically implanted within a patient adjacent to the AIMD or lead wire system.
Owner:WILSON GREATBATCH LTD
Method and anchor for medical implant placement, and method of anchor manufacture
ActiveUS20050065589A1Transvascular endocardial electrodesCatheterDistal portionMonitoring physiological parameters
An anchor and procedure for placing a medical implant, such as for monitoring physiological parameters. The anchor includes a central body in which a medical implant can be received. arms and members extend radially from first and second ends, respectively, of the central body. Each member defines a leg extending toward distal portions of the arms to provide a clamping action. The anchor and its implant are placed by coupling first and second guidewires to first and second portions of the anchor, placing an end of a delivery catheter in a wall where implantation is desired, inserting the anchor in the catheter with the guidewires to locate the anchor within the wall, deploying the arms of the anchor at one side of the wall followed by deployment of the members at the opposite side of the wall, and thereafter decoupling the guidewires from the anchor.
Owner:UIM PRESSURE IMPLANT INC
Methods and devices for ablation
InactiveUS6840936B2Promote progressGap can be presentUltrasonic/sonic/infrasonic diagnosticsCannulasBiomedical engineeringMembrane configuration
Owner:ST JUDE MEDICAL ATRIAL FIBRILLATION DIV
Treatment for patient with congestive heart failure
Owner:TRANSCARDIAC THERAPEUTICS
Method and system for nerve stimulation prior to and during a medical procedure
InactiveUS7184829B2Prevent beatingStimulate beatingTransvascular endocardial electrodesHeart defibrillatorsSacral nerve stimulationNerve stimulation
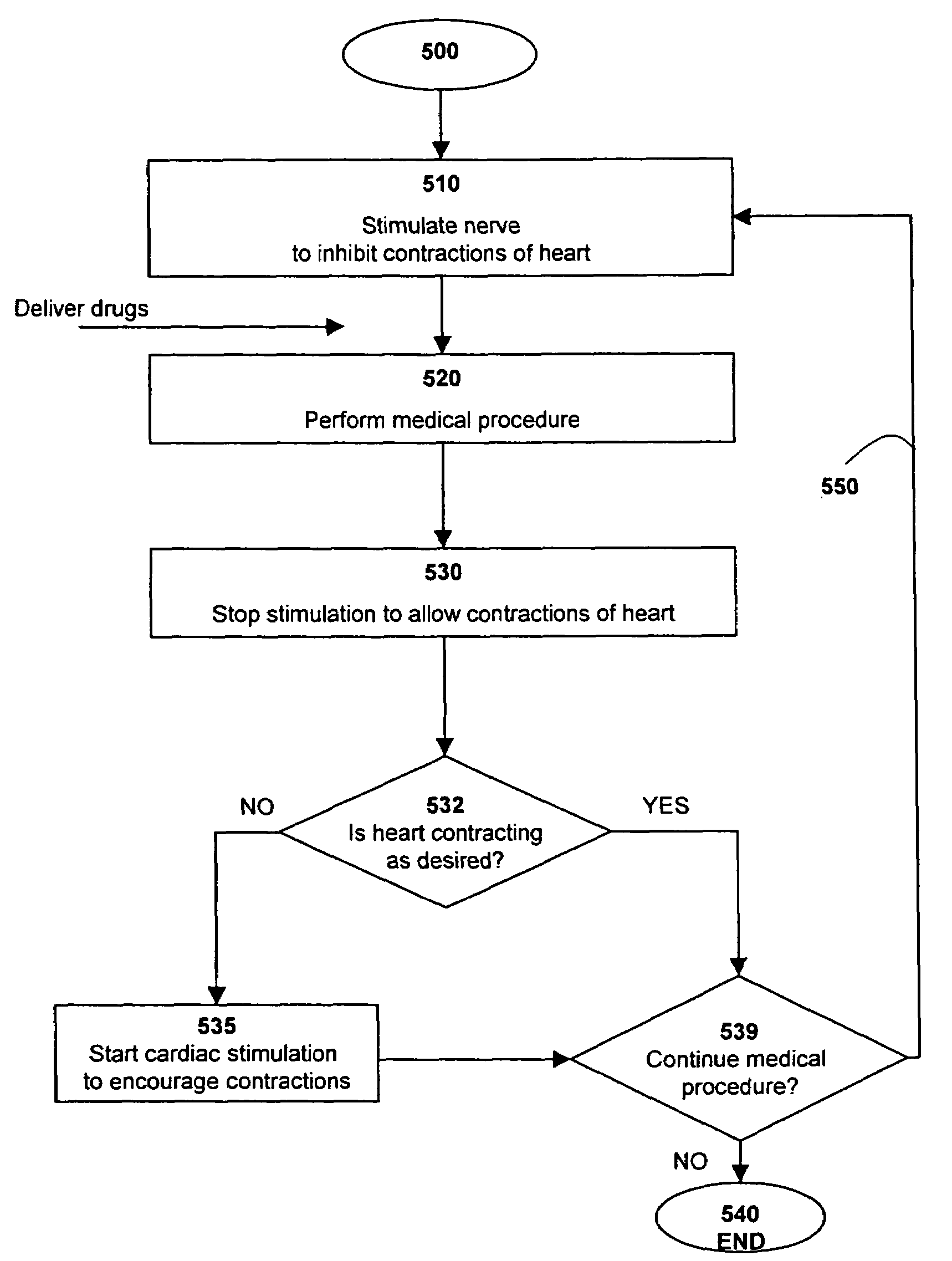
A method of performing a medical procedure, such as surgery, is provided. A nerve is stimulated in order to adjust the beating of the heart to a first condition, such as a stopped or slowed condition. The medical procedure is performed on the heart or another organ. The stimulation of the nerve is stopped in order to adjust the beating of the heart to a second condition, such as a beating condition. The heart itself may also be stimulated to a beating condition, such as by pacing. The stimulation of the nerve may be continued in order to allow the medical procedure to be continued. Systems and devices for performing the medical procedure are also provided.
Owner:MEDTRONIC INC
Fixed length anchor and pull mitral valve device and method
InactiveUS6976995B2Good effectSuture equipmentsTransvascular endocardial electrodesCoronary sinusMitral valve leaflet
A device effects the mitral valve annulus geometry of a heart. The device includes first and second anchors configured to be positioned within the coronary sinus of the heart adjacent the mitral valve annulus of the heart and a fixed length connecting member permanently attached to the first and second anchors. With the first anchor anchored in the coronary sinus, the second anchor may be displaced proximally to effect the geometry of the mitral valve annulus and released to maintain the effect on the mitral valve geometry.
Owner:CARDIAC DIMENSIONS
Conditioning of coupled electromagnetic signals on a lead
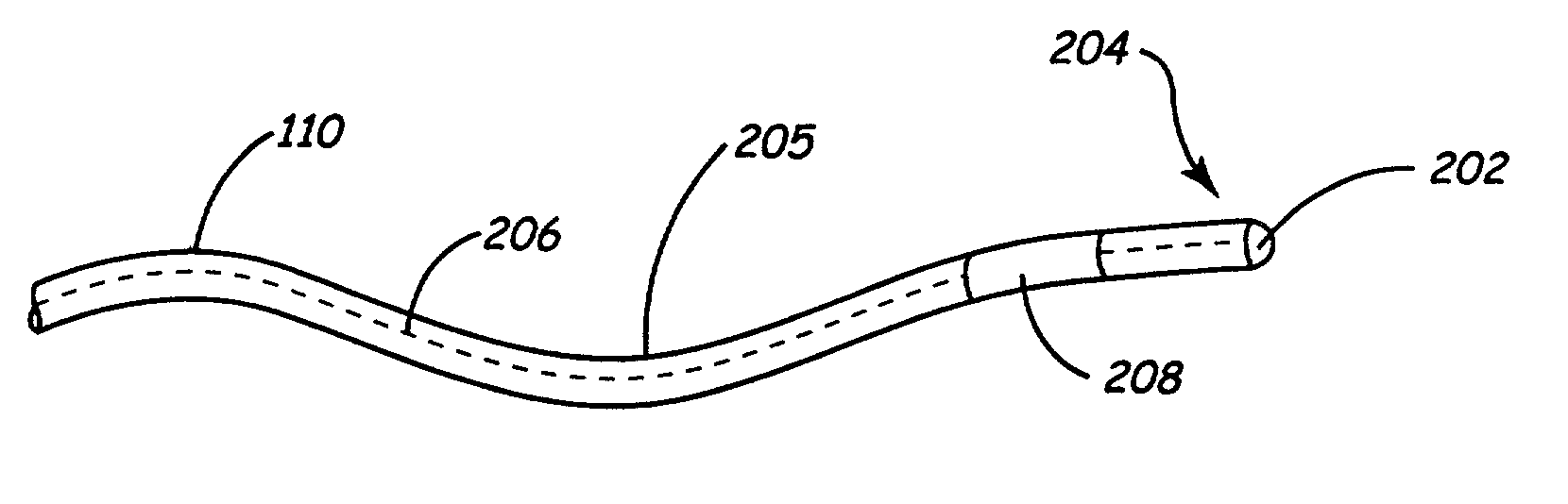
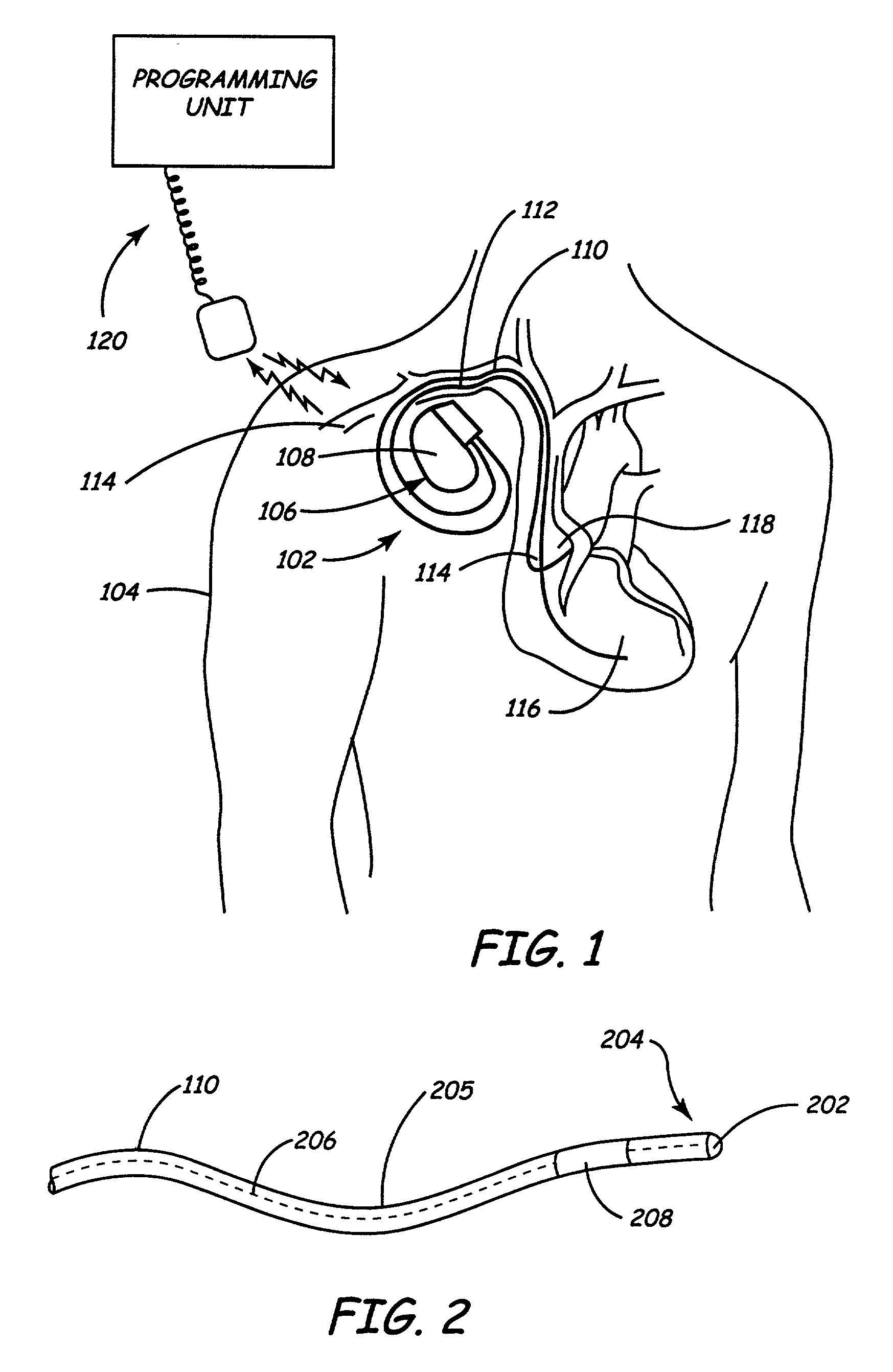
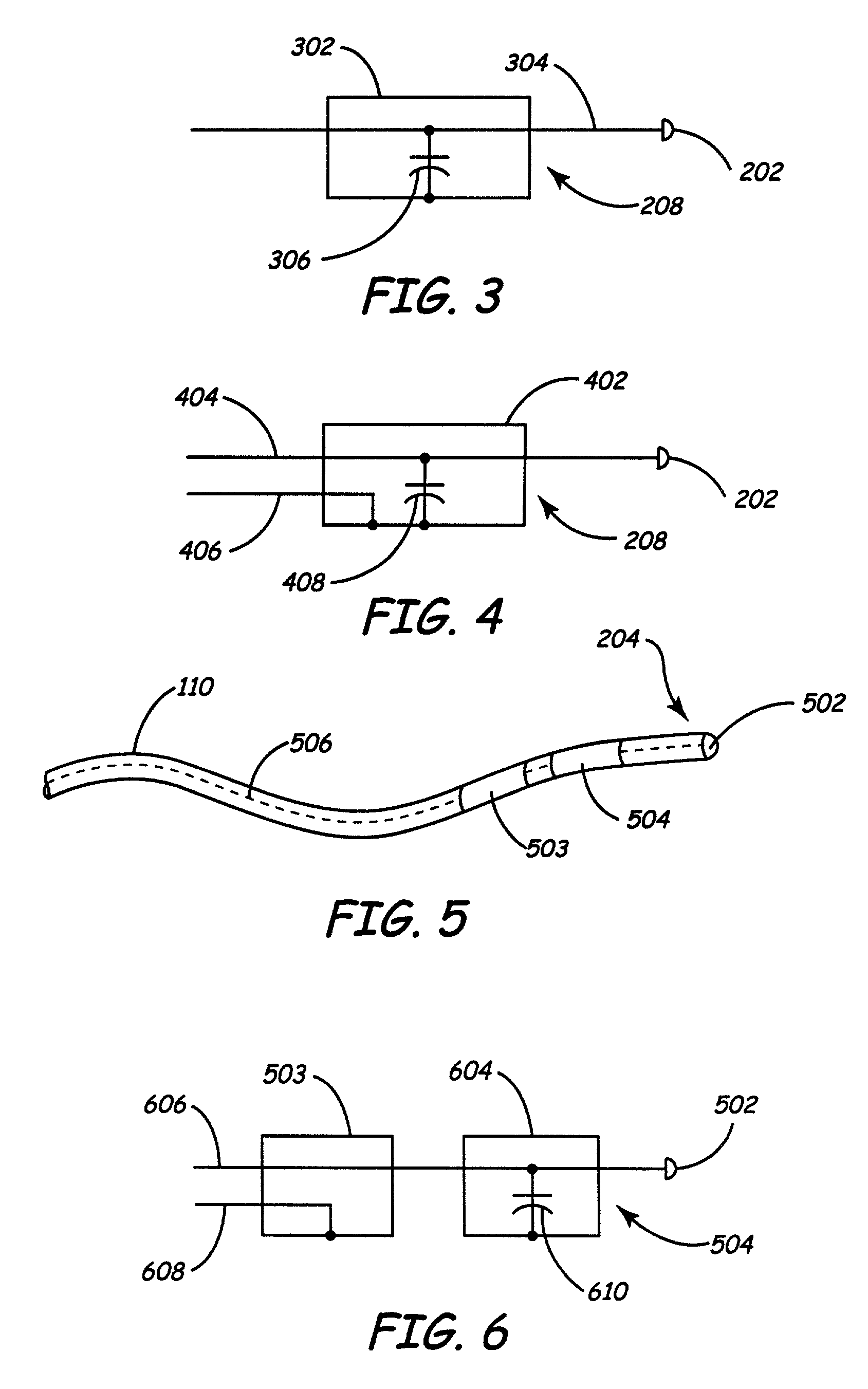
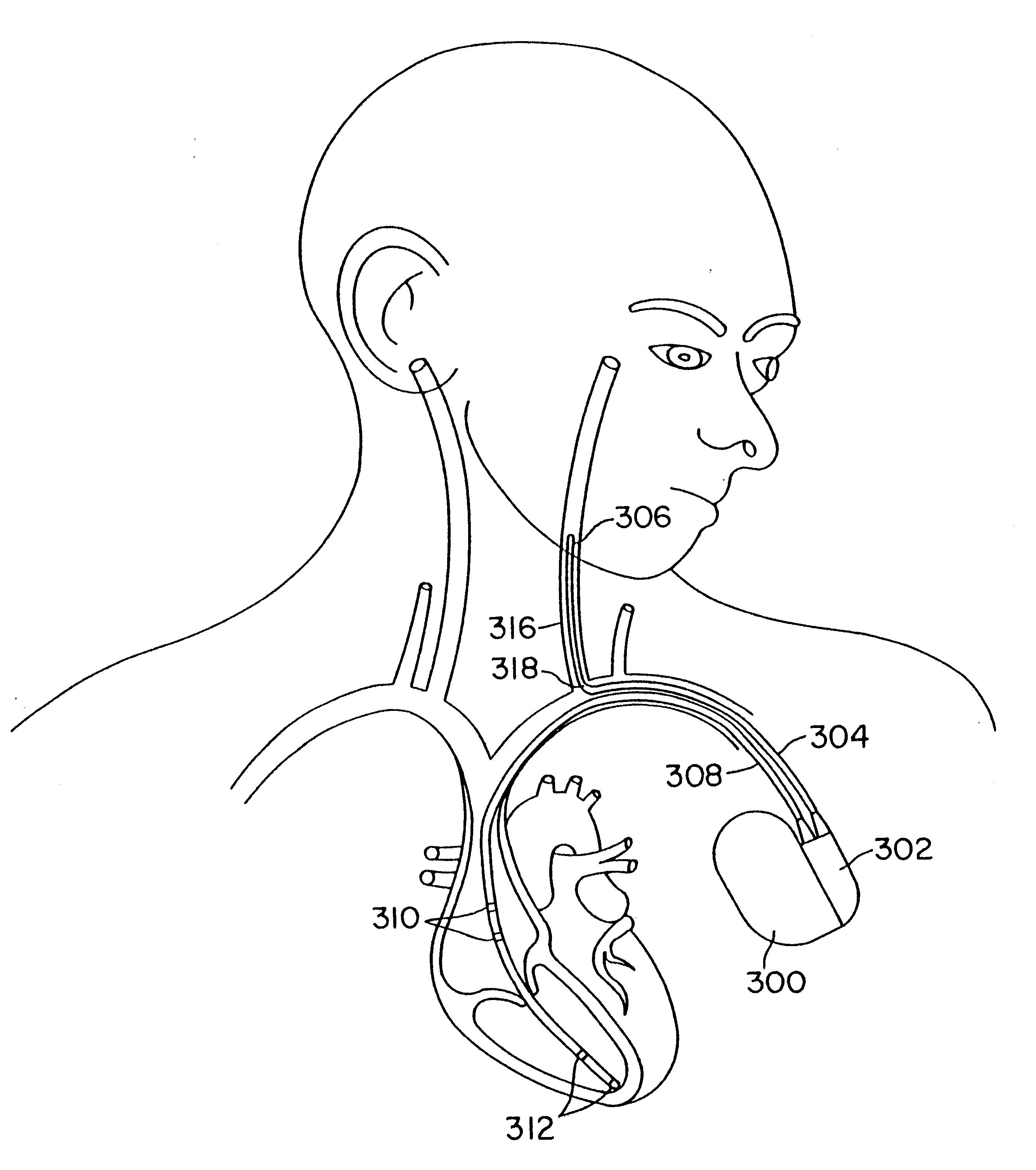
InactiveUS7013180B2Reducing coupled currentTotal current dropTransvascular endocardial electrodesSurgeryElectromagnetic shieldingEngineering
Owner:MEDTRONIC INC
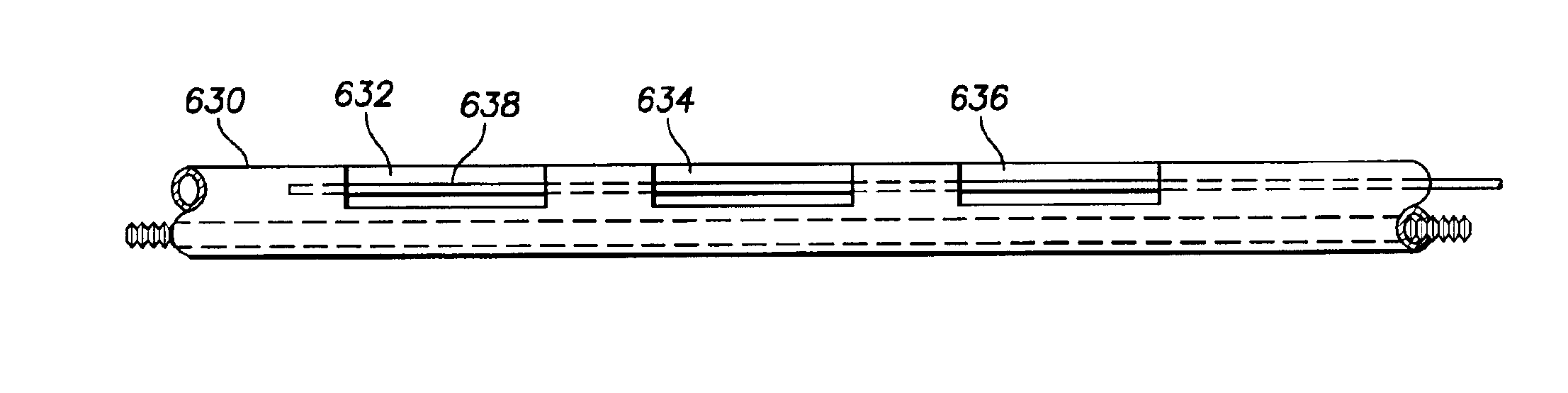
Electrode support
InactiveUS20080114230A1Electrode increaseTransvascular endocardial electrodesDiagnostic recording/measuringLead structureBiomedical engineering
Electrode supports configured to provide a foundation for a segmented electrode on a flexible lead structure are provided. Also provided are electrode structures, leads that include the same, implantable pulse generators that include the leads, as well as systems and kits having components thereof, and methods of making and using the subject devices.
Owner:PROTEUS DIGITAL HEALTH INC
Delivery Catheter Systems and Methods
A leadless cardiac pacemaker comprises a housing, a plurality of electrodes coupled to an outer surface of the housing, and a pulse delivery system hermetically contained within the housing and electrically coupled to the electrode plurality, the pulse delivery system configured for sourcing energy internal to the housing, generating and delivering electrical pulses to the electrode plurality. Systems and methods for delivering the leadless cardiac pacemaker with delivery catheters are also provided. In some embodiments, the delivery catheters include first and second coaxial shafts configured to apply rotational torque to the pacemaker. In other embodiments, the pacemaker is held in place on the catheter with a tether.
Owner:NANOSTIM
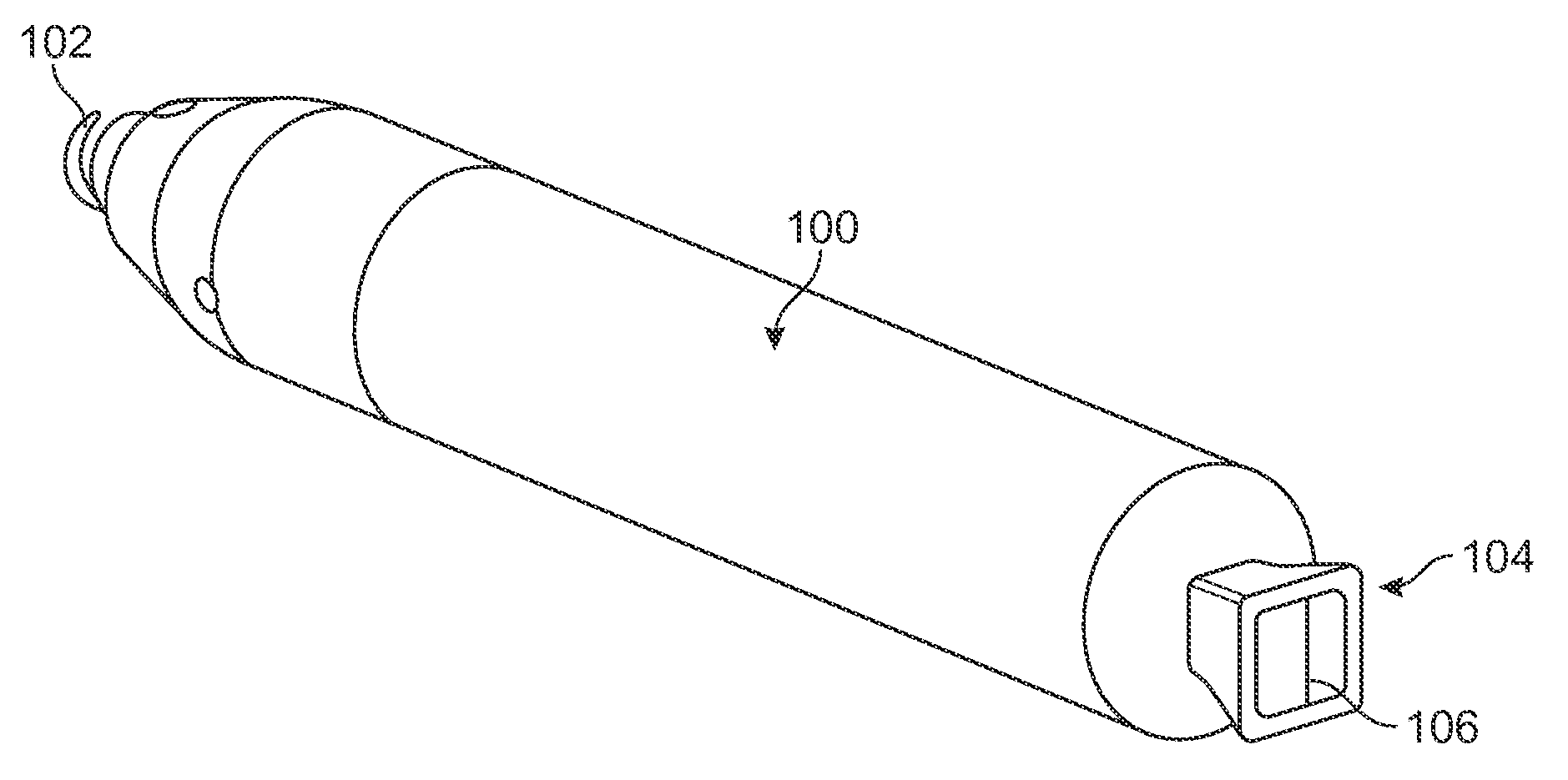
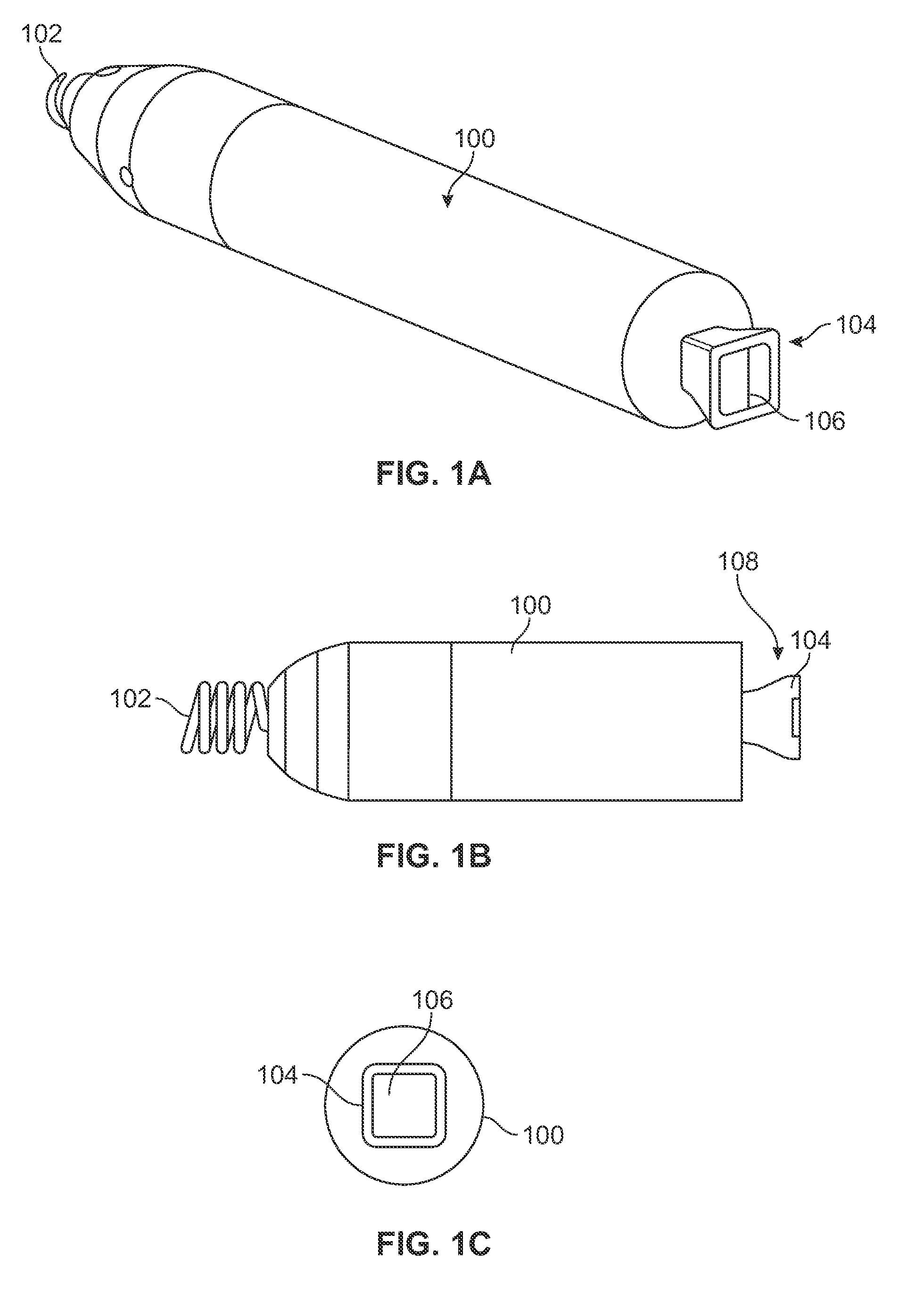
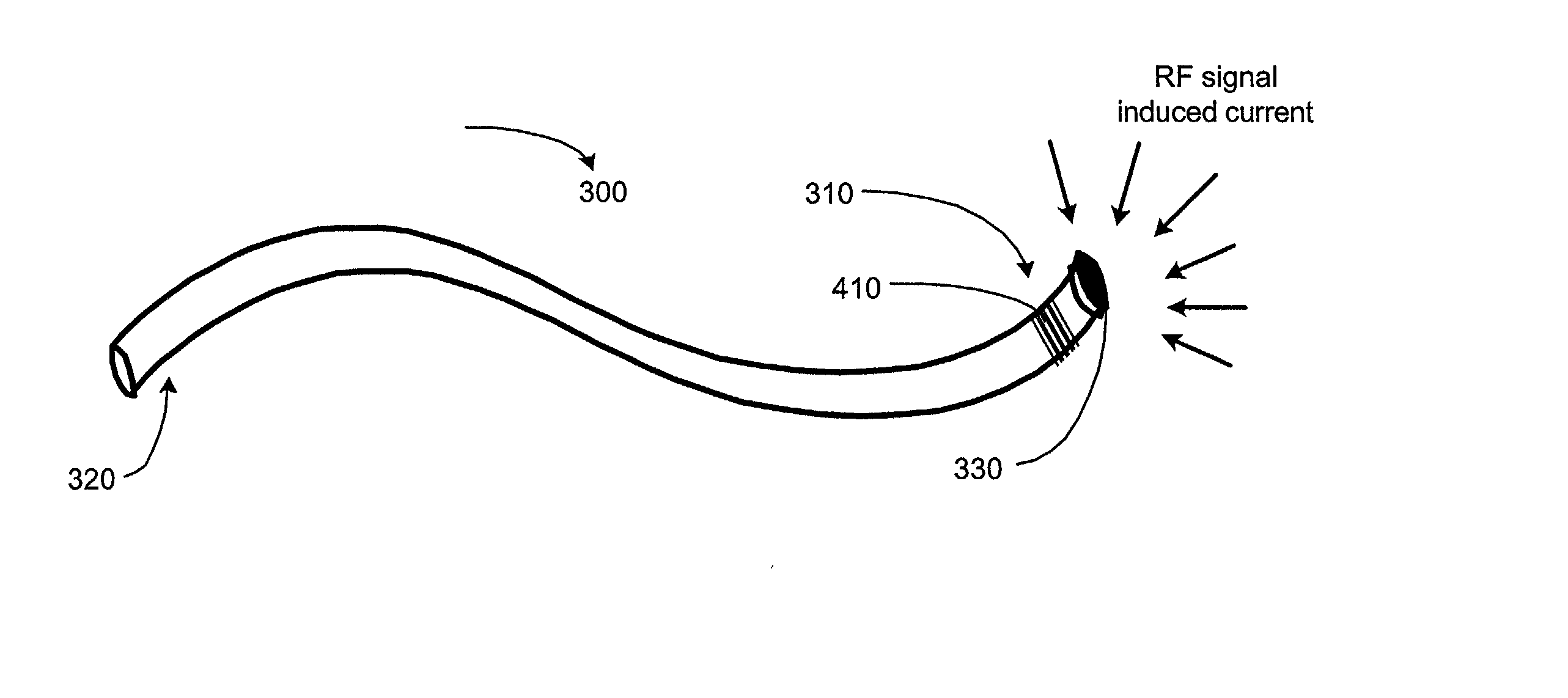
Electromagnetic trap for a lead
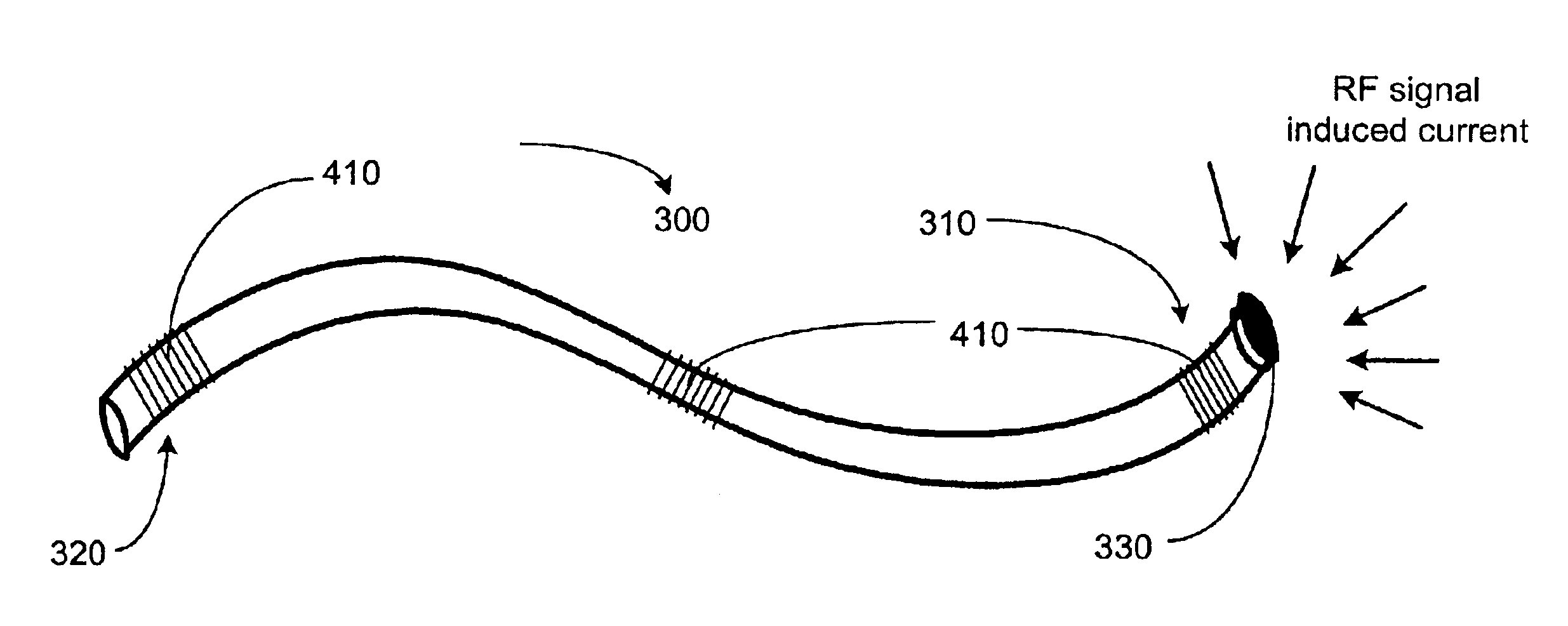
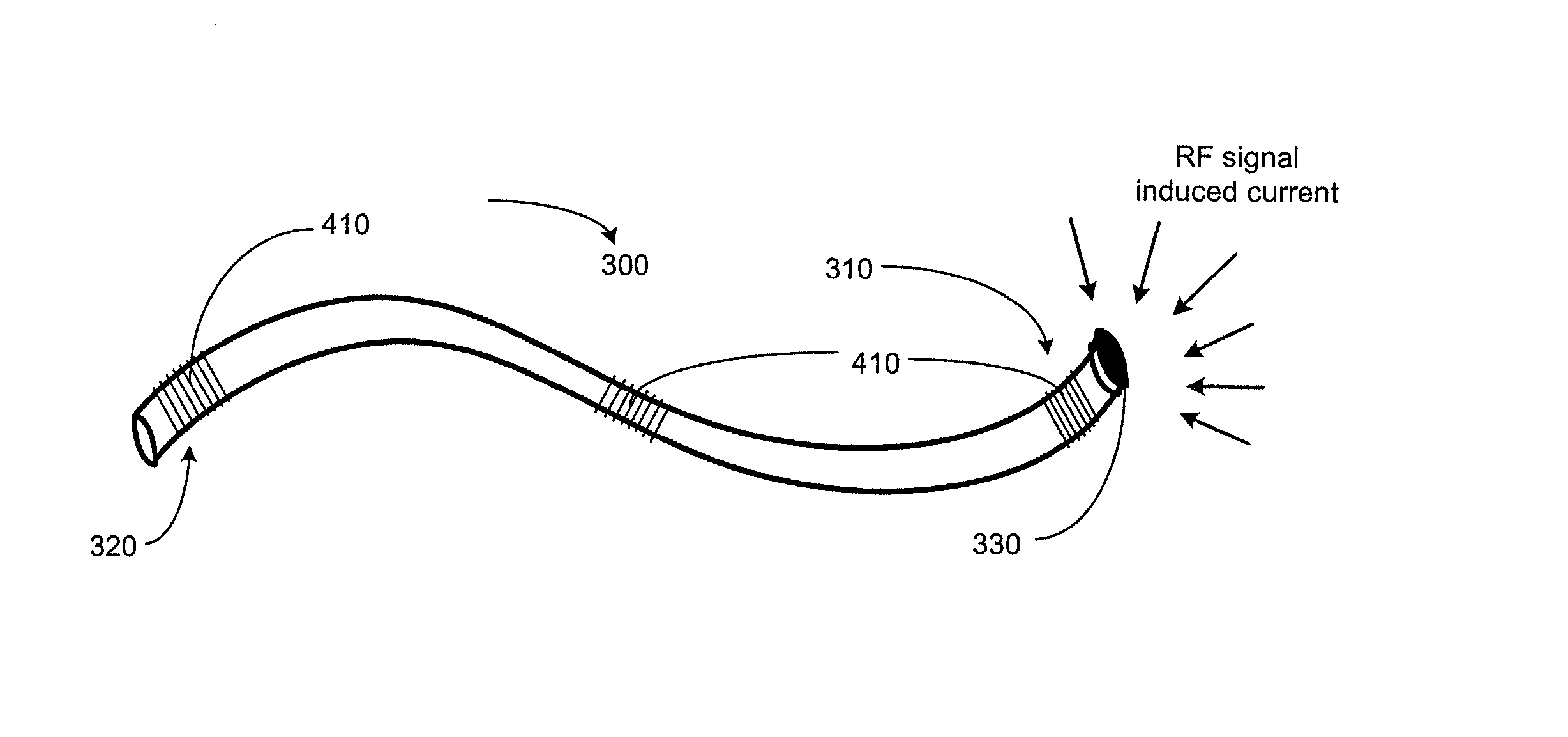
A method and an apparatus for trapping induced current resulting from an electromagnetic field. Embodiments of the present invention provide for an elongate body having a proximal and a distal end portion and a coil wound about the distal end, the coil to provide an electromagnetic trap for filtering radio frequency (RF) signal-induced currents.
Owner:MEDTRONIC INC
Implantable pressure transducer system optimized for anchoring and positioning
InactiveUS20050288722A1Long-term stabilityFacilitate optimal healingTransvascular endocardial electrodesSurgical needlesTransducerBiomedical engineering
This invention relates generally to systems and methods for optimizing the performance and minimizing complications related to implanted sensors, such as pressure sensors, for the purposes of detecting, diagnosing and treating cardiovascular disease in a medical patient. Systems and methods for anchoring implanted sensors to various body structures is also provided.
Owner:PACESETTER INC
Methods and devices for ablation
InactiveUS6858026B2Promote progressGap can be presentUltrasonic/sonic/infrasonic diagnosticsCannulasBiomedical engineeringMembrane configuration
Owner:ST JUDE MEDICAL ATRIAL FIBRILLATION DIV
Positive fixation percutaneous epidural neurostimulation lead
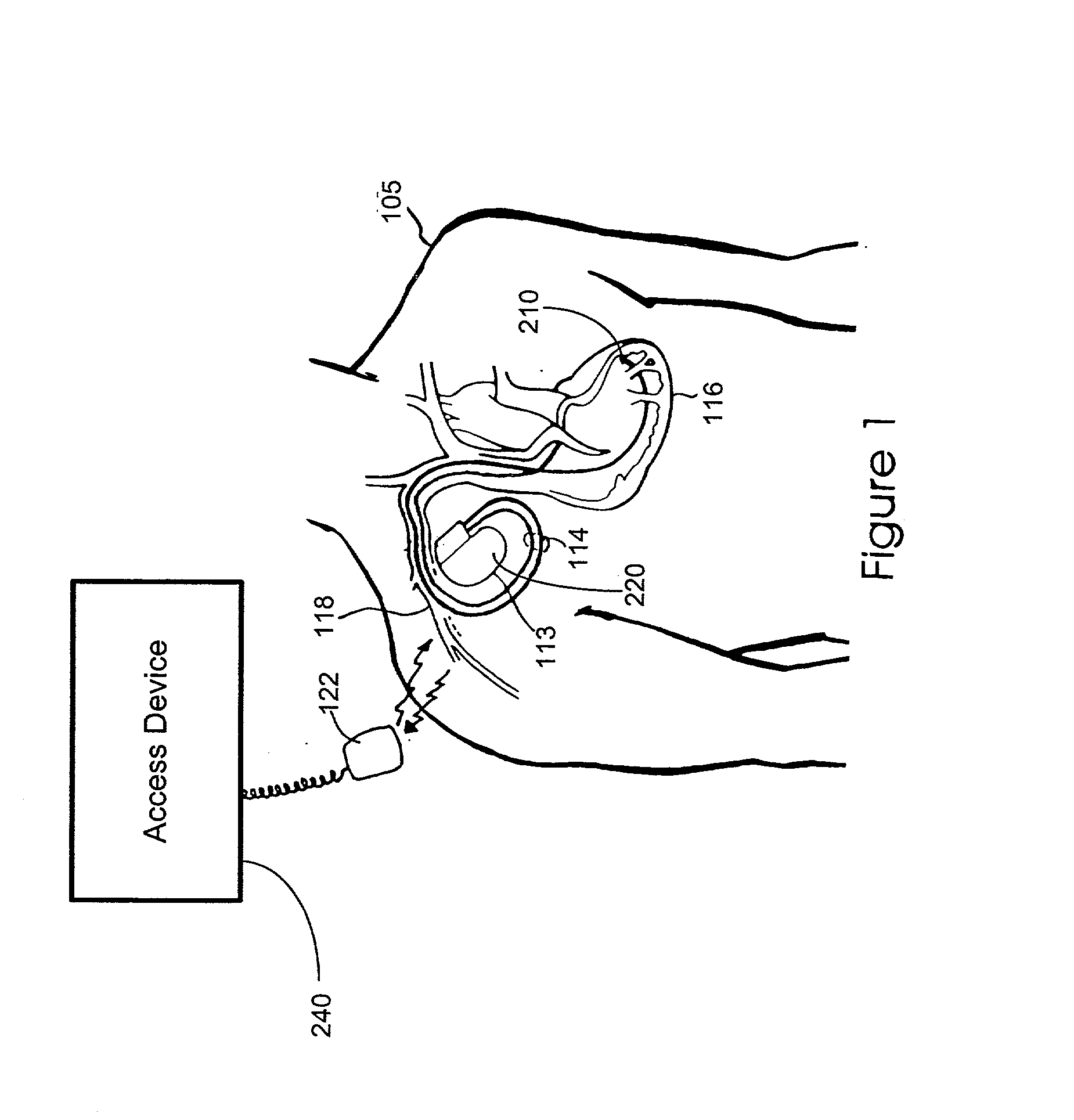
InactiveUS20060041295A1Direction easyInhibit migrationSpinal electrodesTransvascular endocardial electrodesLinear configurationSignal generator
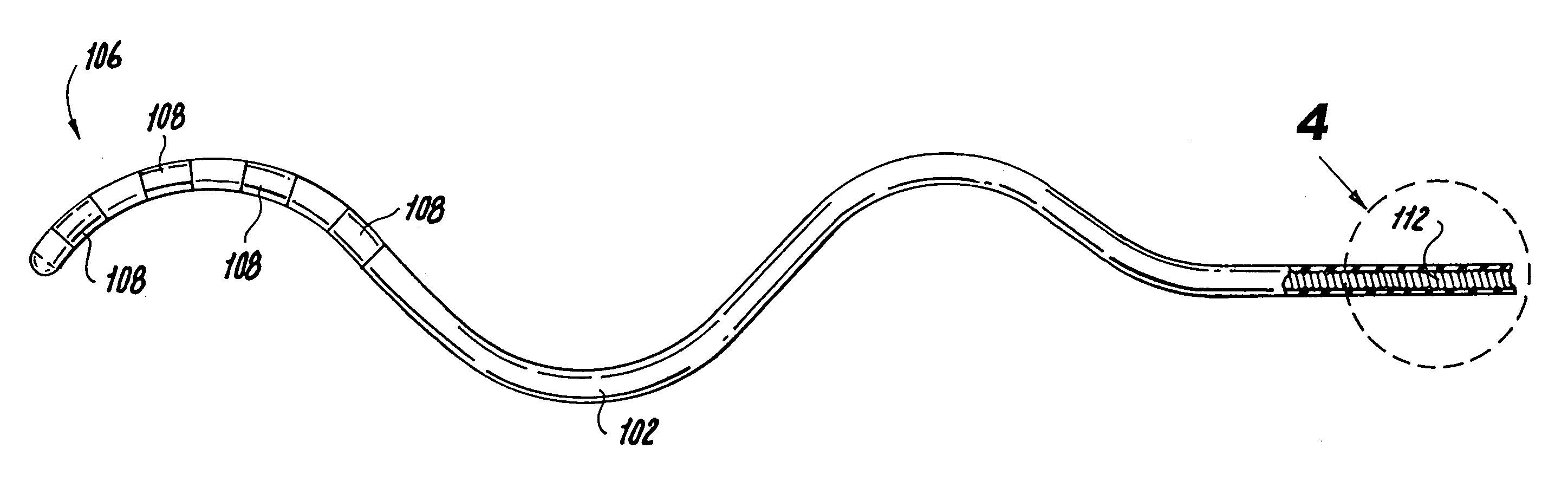
Disclosed is a lead for percutaneous insertion into an epidural space of a spinal canal, which includes an elongated lead body having opposed proximal and distal end portions. At least one electrode for stimulating a patient is operatively associated with the distal end portion of the lead body. Structure for conducting signals extends through the lead body to connect the electrode to a connecting structure operatively associated with the proximal end portion of the lead body. The connecting structure is capable of engaging a signal generator such that signals can be conducted from a signal generator to the electrode. The distal end portion of the lead body is adapted for movement between a first state, in which the distal end portion has a generally linear configuration, and a second state, in which the distal end portion has an undulating configuration.
Owner:NEUROPOINT MEDICAL
Conditioning of coupled electromagnetic signals on a lead
A method and an apparatus for reducing coupled electrical energy resulting from an electromagnetic field. Embodiments of the present invention provide for an elongate body having a proximal end portion, a middle portion, and a distal end portion and at least one coil wound about at least one of the proximal end portion, the middle portion, and the distal end portion, the coil to provide for filtering of radio frequency (RF) signal-coupled electrical energy.
Owner:MEDTRONIC INC