Patents
Literature
839 results about "Anti virals" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
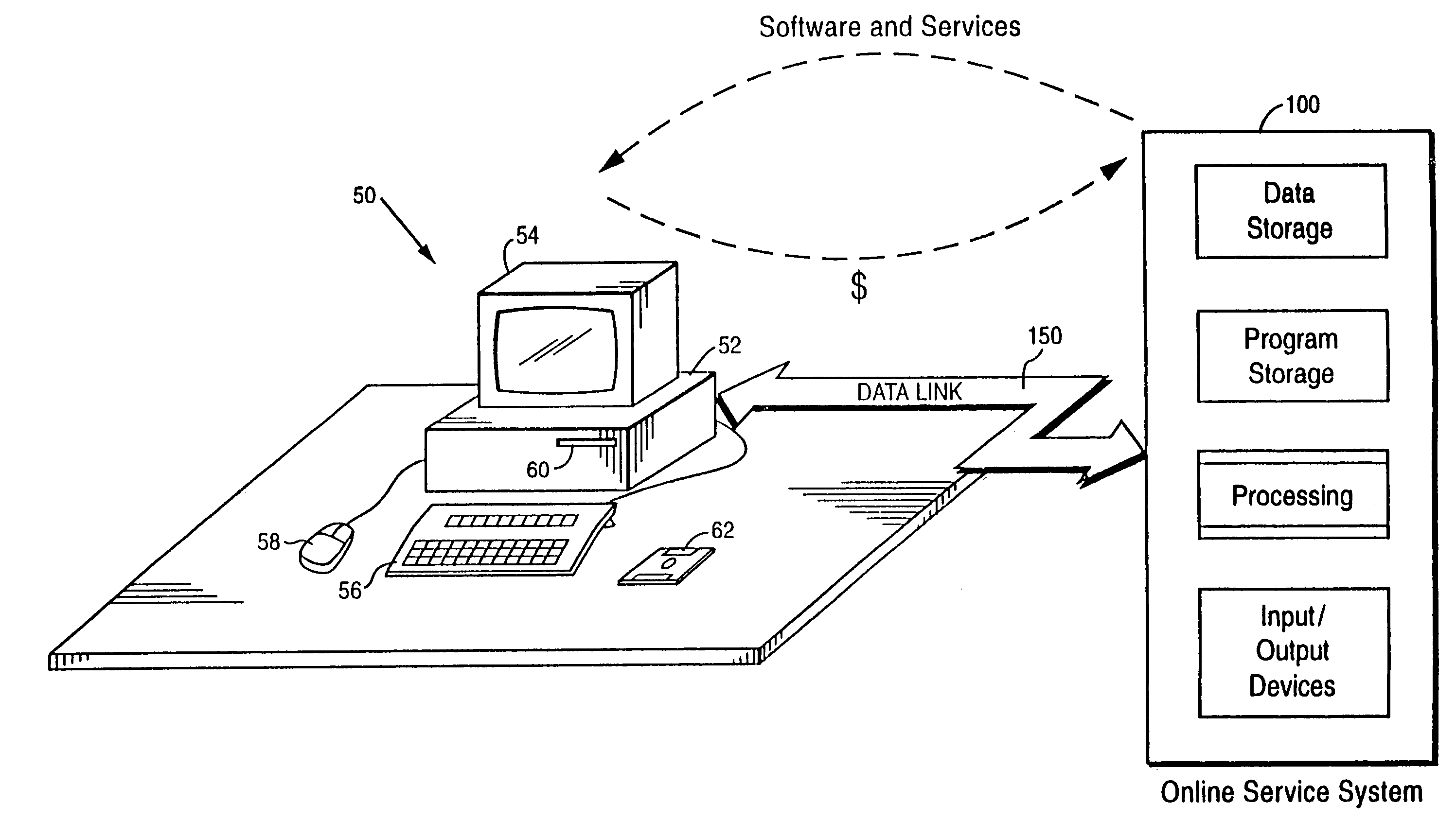
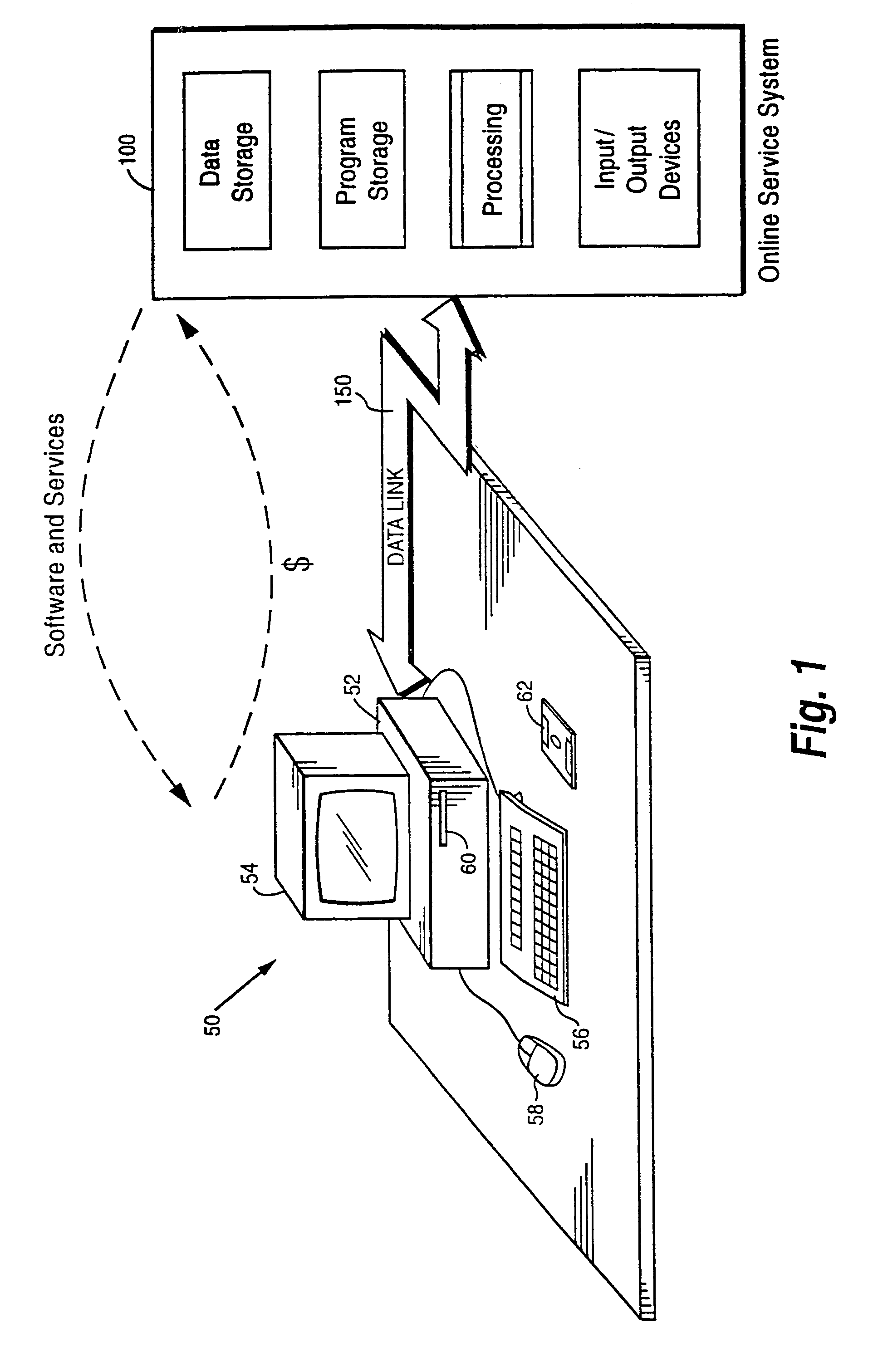
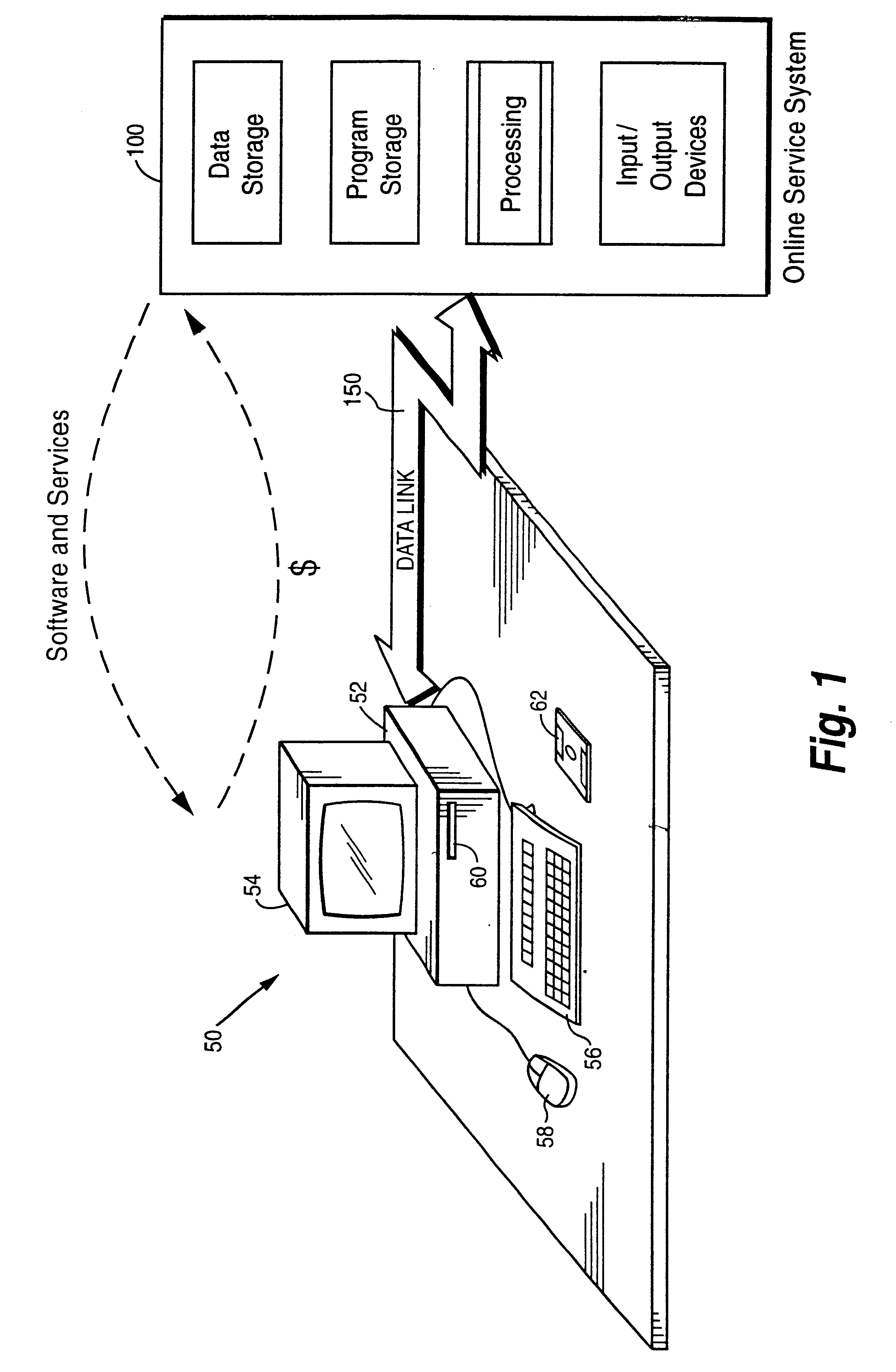
Internet download systems and methods providing software to internet computer users for local execution
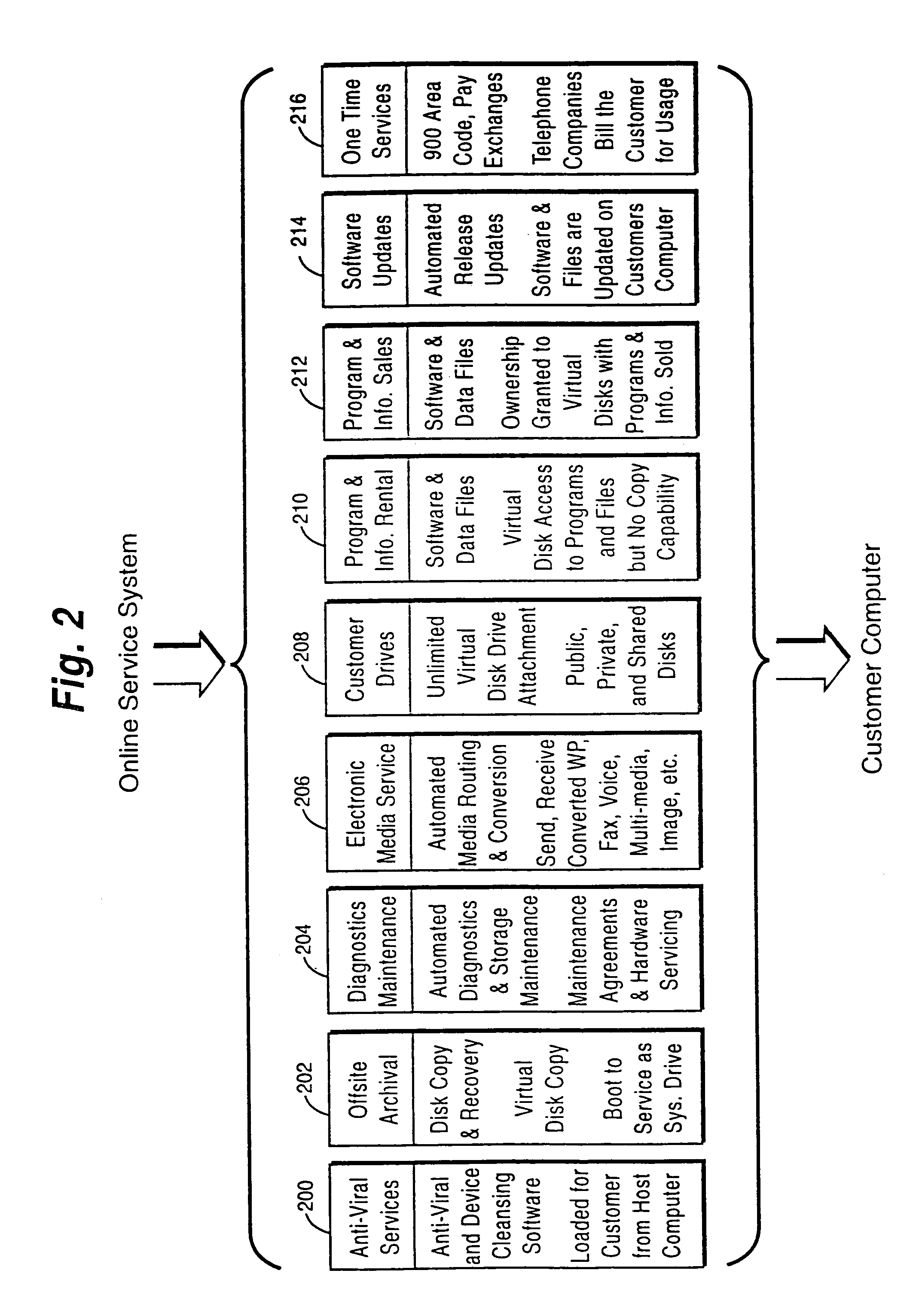
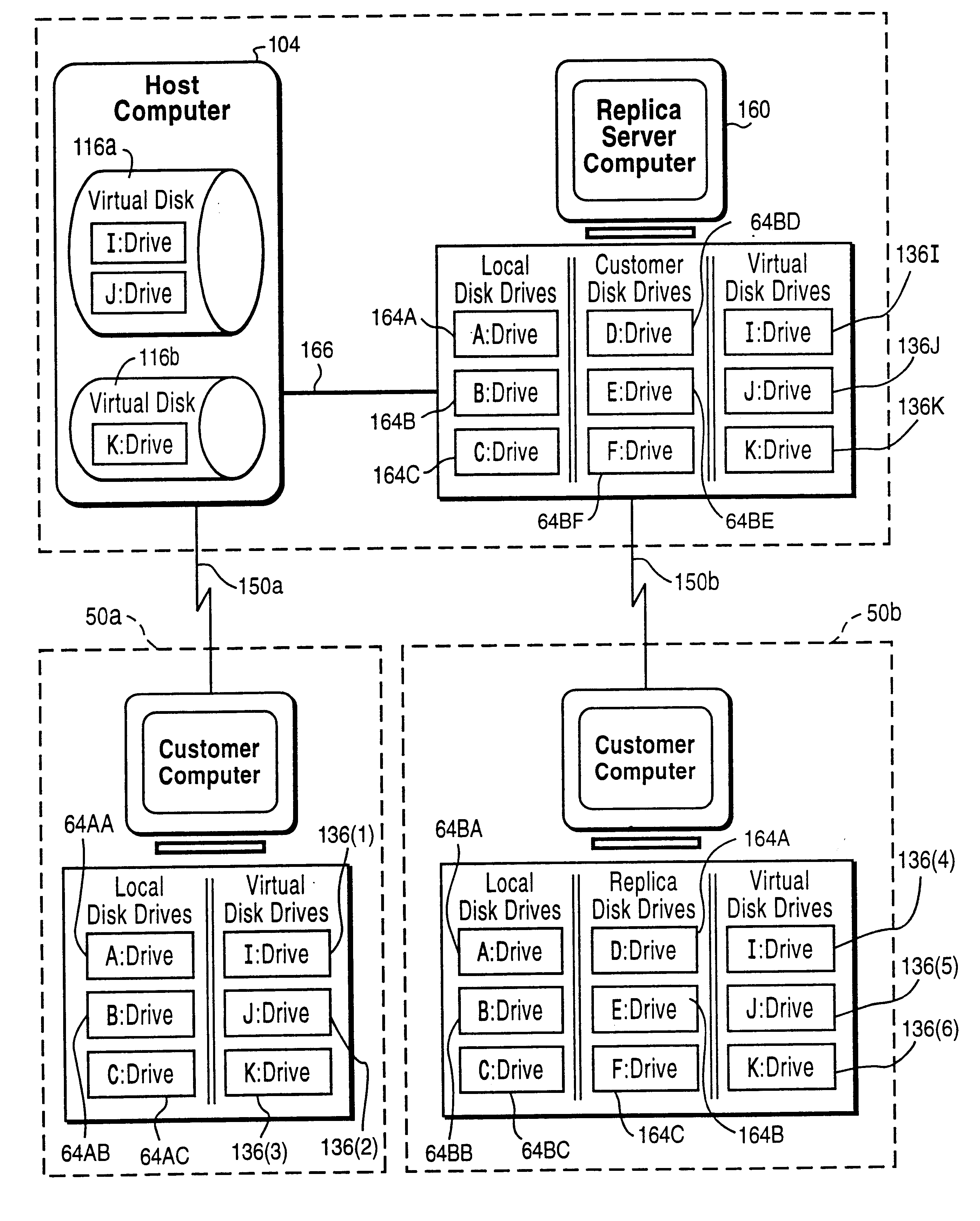
A customer computer connects to an online service provider by phone, Internet, or other telecommunications link. The link gives the customer access to additional processing and storage resources such as virtual storage, processing power and / or additional software or data through interaction between the customer computer and an online service provider computer over the link. The additional resources made available to the customer computer enhance the customers' local needs through access to virtual storage, a more powerful processor of similar type for program execution, and / or online support services such as software rental, software sales, release update services, anti-viral services, data backup and recovery services, diagnostic services and / or repair services.
Owner:OASIS RES +6
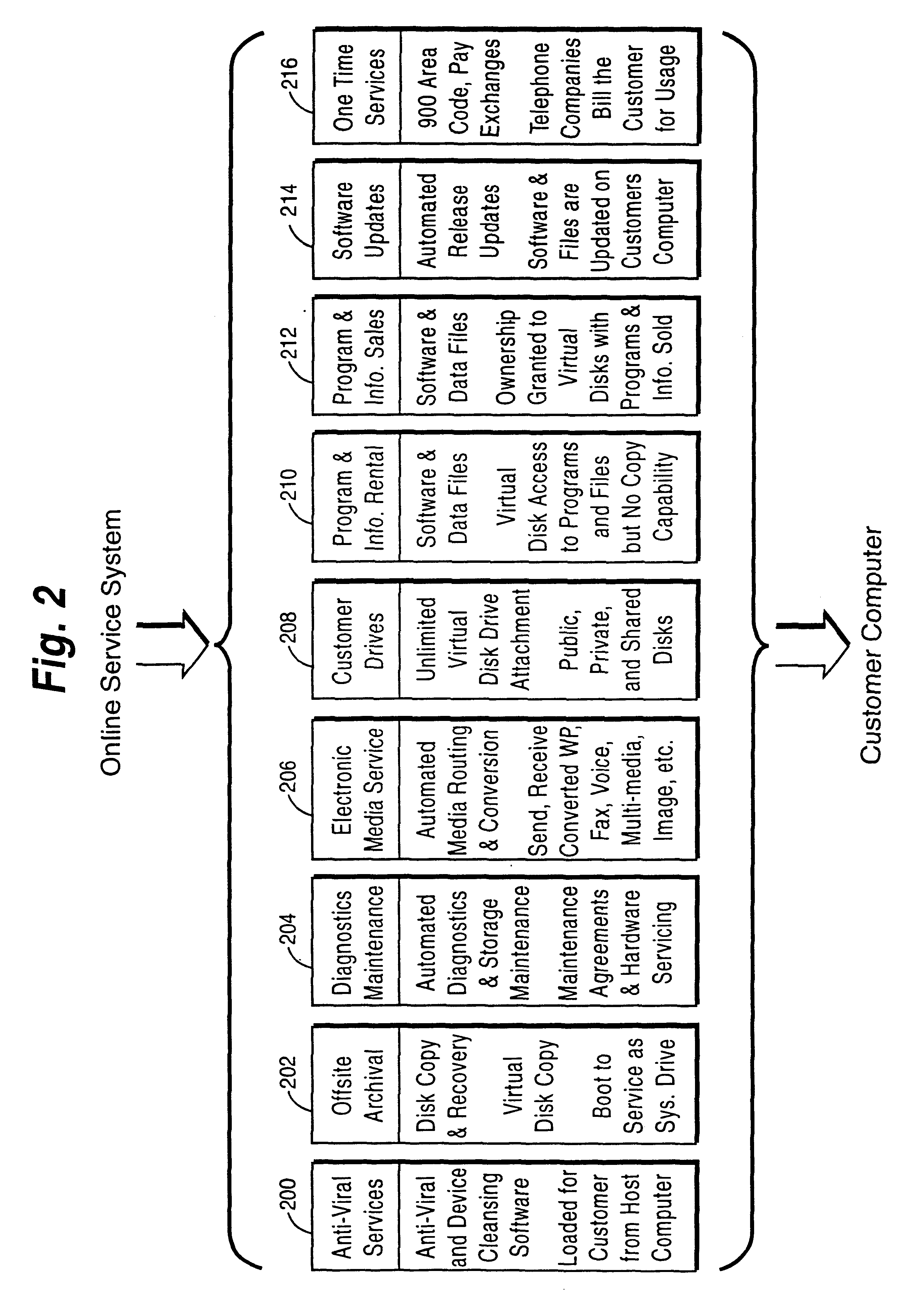
Online computer services including help desk, anti-virus and/or application service features
InactiveUS6327579B1Guaranteed uptimeRelieving userFinanceResource allocationAnti virusTelecommunications link
A customer computer connects to an online service provider by phone, Internet, or other telecommunications link. The link gives the customer access to additional processing and storage resources such as virtual storage, processing power and / or additional software or data through interaction between the customer computer and an online service provider computer over the link. The additional resources made available to the customer computer enhance the customers' local needs through access to virtual storage, a more powerful processor of similar type for program execution, and / or online support services such as software rental, software sales, release update services, anti-viral services, data backup and recovery services, diagnostic services and / or repair services.
Owner:EMC CORP +6
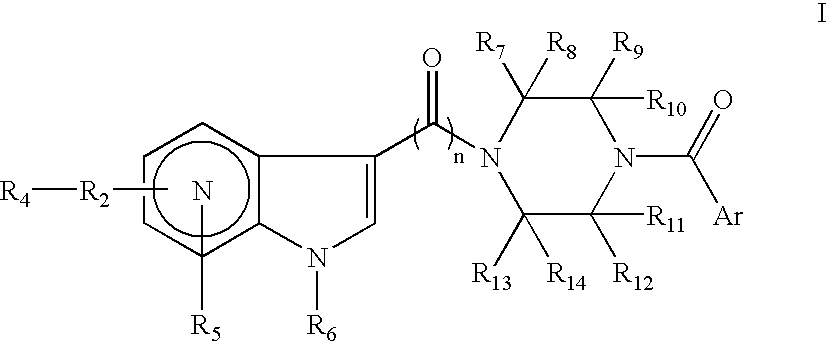
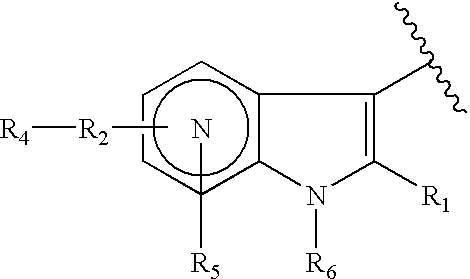
Antiviral azaindole derivatives
The invention comprises compounds of the class of azaindole piperazine diamide derivatives, compositions thereof and their use as anti-viral agents, and particularly for treating HIV infection.
Owner:VIIV HEALTHCARE UK (NO 5) LTD
Use of Oligonucleotides with Modified Bases as Antiviral Agents
The present invention relates to the use of oligonucleotides having modified nucleobases to inhibit gene expression and / or replication of viruses in a subject. The modified nucleobases may be mercapto-modified bases or hydroxy-modified nucleobases. It is contemplated that the oligonucleotides further comprise a nuclease complex which enhances anti-viral activity of the oligonucleotides.
Owner:BALTIC TECH DEV
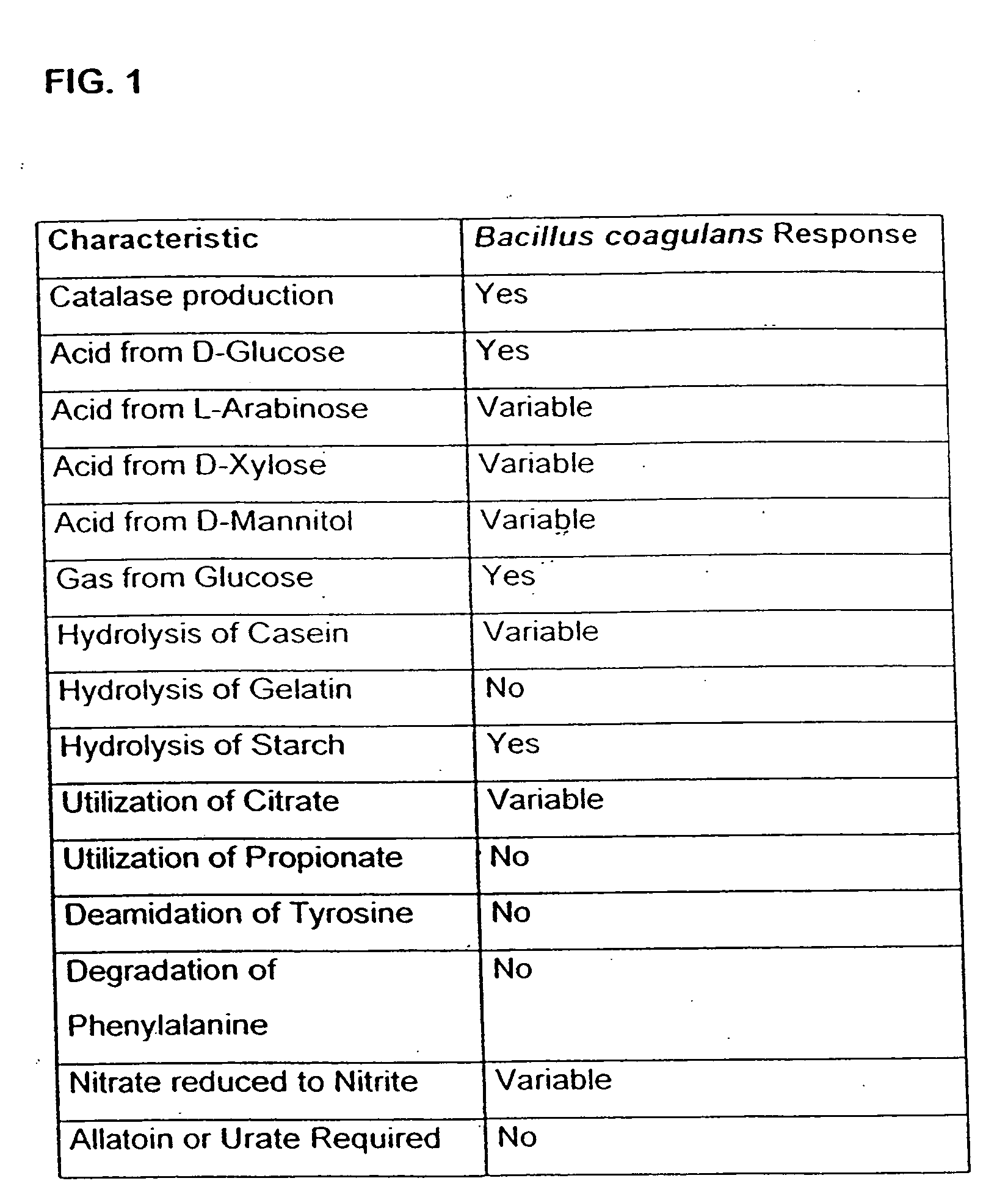
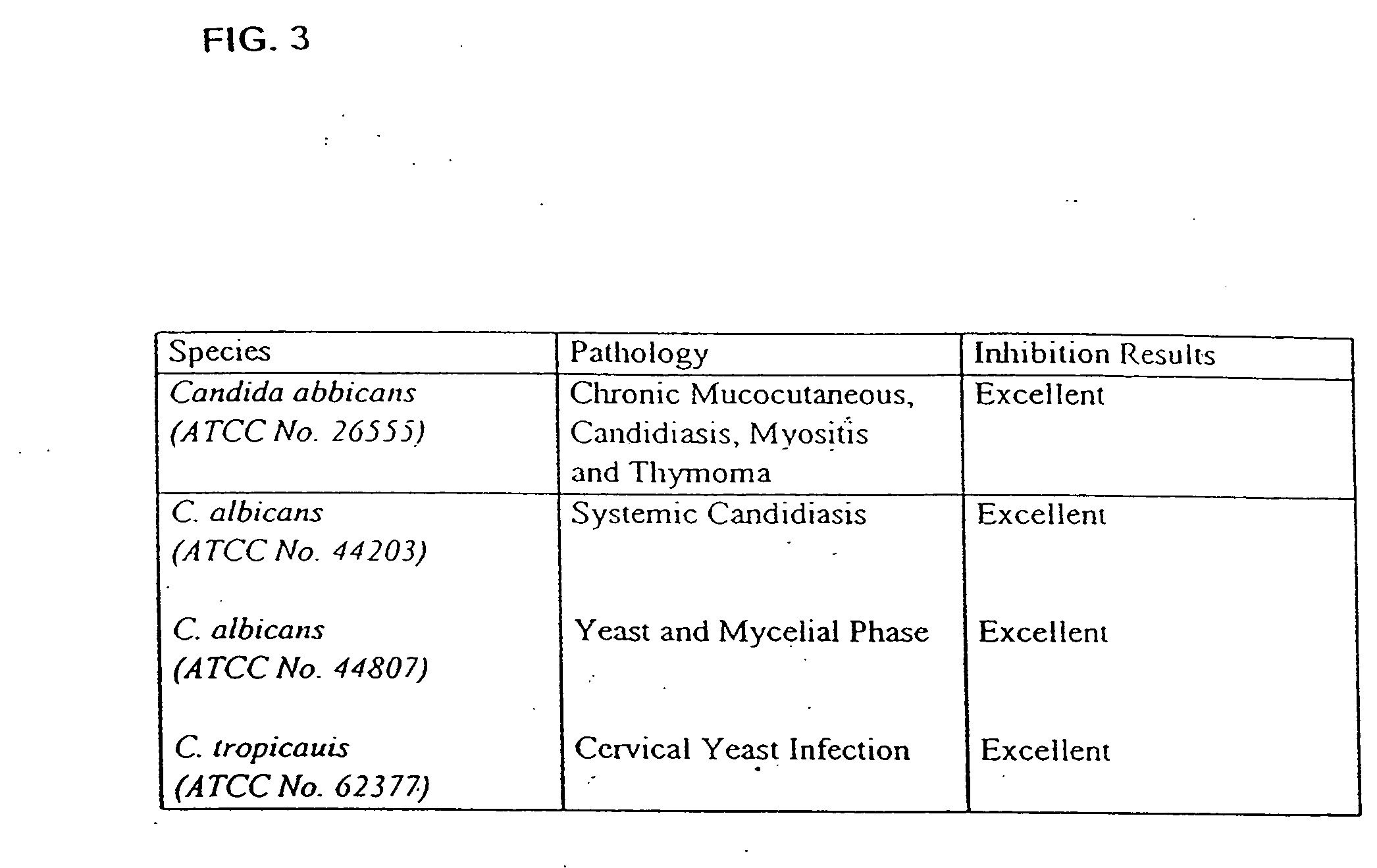
Probiotic, lactic acid-producing bacteria and uses thereof
InactiveUS20060099197A1Good curative effectMitigating deleterious side-effectsAntibacterial agentsBiocideMicrobial agentAnti fungal
The present invention discloses compositions and methodologies for the utilization of probiotic organisms in therapeutic compositions. More specifically, the present invention relates to the utilization of one or more species or strains of lactic acid-producing bacteria, preferably strains of Bacillus coagulans, for the control of gastrointestinal tract pathogens, including antibiotic-resistant gastrointestinal tract pathogens, and their associated diseases by both a reduction in the rate of colonization and the severity of the deleterious physiological effects of the colonization of the antibiotic-resistant pathogen. In addition, the present invention relates to the utilization of therapeutic compounds comprised of lactic acid—producing bacteria and anti-microbial agents such as antibiotics, anti-fungal compounds, anti-yeast compounds, or anti-viral compounds. The present invention also discloses methodologies for: (i) the selective breeding and isolation of probiotic, lactic acid-producing bacterial strains which possess resistance or markedly decreased sensitivity to anti-microbial agents (e.g., antibiotics, anti-fungal agents, anti-yeast agents, and anti-viral agents); and (ii) treating or preventing bacteria-mediated infections of the gastrointestinal tract by use of the aforementioned probiotic bacterial strains with or without the concomitant administration of antibiotics. While the primary focus is on the treatment of gastrointestinal tract infections, the therapeutic compositions of the present invention may also be administered to buccal, vaginal, optic, and like physiological locations.
Owner:GANEDEN BIOTECH
Anti-Viral Compounds
ActiveUS20100168138A1Lower Level RequirementsOrganic active ingredientsBiocideHepacivirusAnti virals
Owner:ABBVIE INC
Anti-viral nucleoside analogs and methods for treating viral infections, especially HIV infections
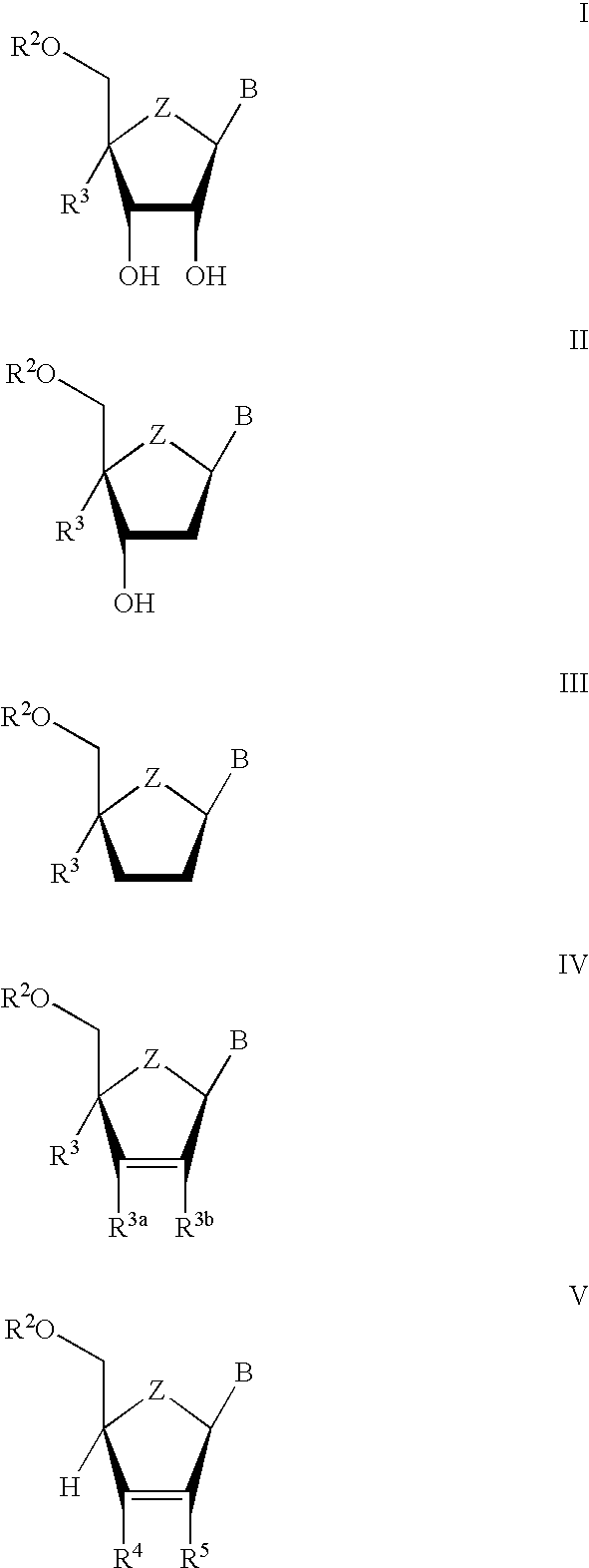
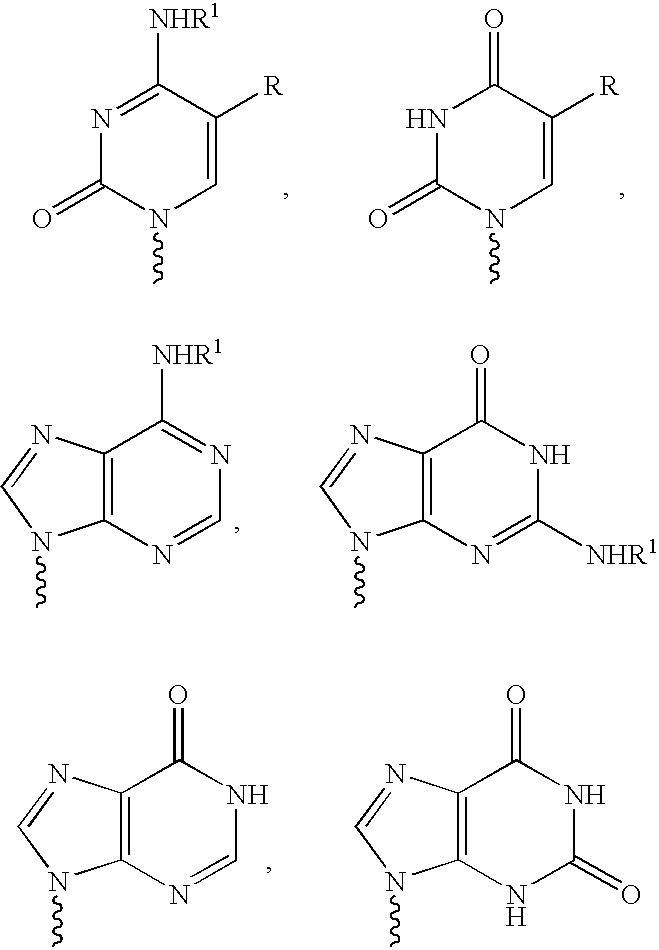
The present invention relates to novel compounds according to the to the general formulas I, II, III, IV or V: wherein B is nucleoside base according to the structure: R is H, F, Cl, Br, I, C1-C4 alkyl (preferably CH3), -C=N, -C=C-Ra, X is H, C1-C4 alkyl (preferably, CH3), F, Cl, Br or I; Z is O or CH2, with the proviso that Z is CH2 and not O when the compound is according to general formula II, R<3 >is -C=C-H and R<2 >is H or a phosphate, diphosphate, triphosphate or phosphotriester group; R<1 >is H, an acyl group, a C1-C20 alkyl or an ether group; R<2 >is H, an acyl group, a C1-C20 alkyl or ether group, a phosphate, diphosphate, triphosphate, phosphodiester group or a group; Nu is a radical of a biologically active antiviral compound such that an amino group or hydroxyl group from said biologically active antiviral compound forms a phosphate, phosphoramidate, carbonate or urethane group with the adjacent moiety; R<8 >is H, or a C1-C20 alkyl or ether group, preferably a C1-C12 alkyl group; k is 0-12, preferably, 0-2; R<3 >is selected from a C1-C4 alkyl (preferably, CH3), -(CH2)n-C=C-Ra, R<3a >and R<3b >are independently selected from H, F, Cl, Br or I; R<4 >and R<5 >are independently selected from H, F, Cl, Br, I, OH, C1-C4 alkyl (preferably, CH3), -(CH2)n-C=C-Ra, with the proviso that R<4 >and R<5 >are not both H; Ra is H, F, Cl, Br, I, or -C1-C4 alkyl, preferably H or CH3; Y is H, F, Cl, Br, I or -C1-C4 alkyl, preferably H or CH3; and n is 0, 1, 2, 3, 4 or 5, preferably 0, 1 or 2; and their anomers, pharmaceutically acceptable salts, solvates, or polymorphs thereof.
Owner:YALE UNIV
Double-stranded and single-stranded RNA molecules with 5 ' triphosphates and their use for inducing interferon
InactiveUS20060178334A1Strong responseInhibits and prevents viral infectionSugar derivativesGenetic material ingredientsSingle-Stranded RNADouble stranded
Double-stranded and single-stranded RNA molecules, and their use in methods for inducing interferon are provided. The interferon induction provides anti-viral and other medically useful effects, such as anti-cancer effects. Also provided are methods for reducing or inhibiting interferon induction exhibited by such molecules, particularly siRNA and shRNA molecules produced in vitro.
Owner:CITY OF HOPE
2' and 3'-nucleoside prodrugs for treating Flaviviridae infections
InactiveUS20070015905A1Inhibit HCV polymerase activityInhibit polymerase activityBiocideSugar derivativesDiseaseReverse transcriptase
2′ and 3′-Prodrugs of 1′, 2′, 3′ or 4′-branched β-D or β-L nucleosides, or their pharmaceutically acceptable salts and derivatives are described, which are useful in the prevention and treatment of Flaviviridae infections and other related conditions. These modified nucleosides provide superior results against flaviviruses and pestiviruses, including hepatitis C virus and viruses generally that replicate through an RNA dependent RNA reverse transcriptase. Compounds, compositions, methods and uses are provided for the treatment of Flaviviridae infection, including HCV infection, that include the administration of an effective amount of the prodrugs of the present invention, or their pharmaceutically acceptable salts or derivatives. These drugs may optionally be administered in combination or alteration with further anti-viral agents to prevent or treat Flaviviridae infections and other related conditions.
Owner:INDENIX PHARM LLC +3
2' and 3'-nucleoside prodrugs for treating Flaviviridae infections
2′ and 3′-Prodrugs of 1′, 2′, 3′ or 4′-branched β-D or β-L nucleosides, or their pharmaceutically acceptable salts and derivatives are described, which are useful in the prevention and treatment of Flaviviridae infections and other related conditions. These modified nucleosides provide superior results against flaviviruses and pestiviruses, including hepatitis C virus and viruses generally that replicate through an RNA dependent RNA reverse transcriptase. Compounds, compositions, methods and uses are provided for the treatment of Flaviviridae infection, including HCV infection, that include the administration of an effective amount of the prodrugs of the present invention, or their pharmaceutically acceptable salts or derivatives. These drugs may optionally be administered in combination or alteration with further anti-viral agents to prevent or treat Flaviviridae infections and other related conditions.
Owner:THE CENT NAT DEL LA RECH SCIQUE +3
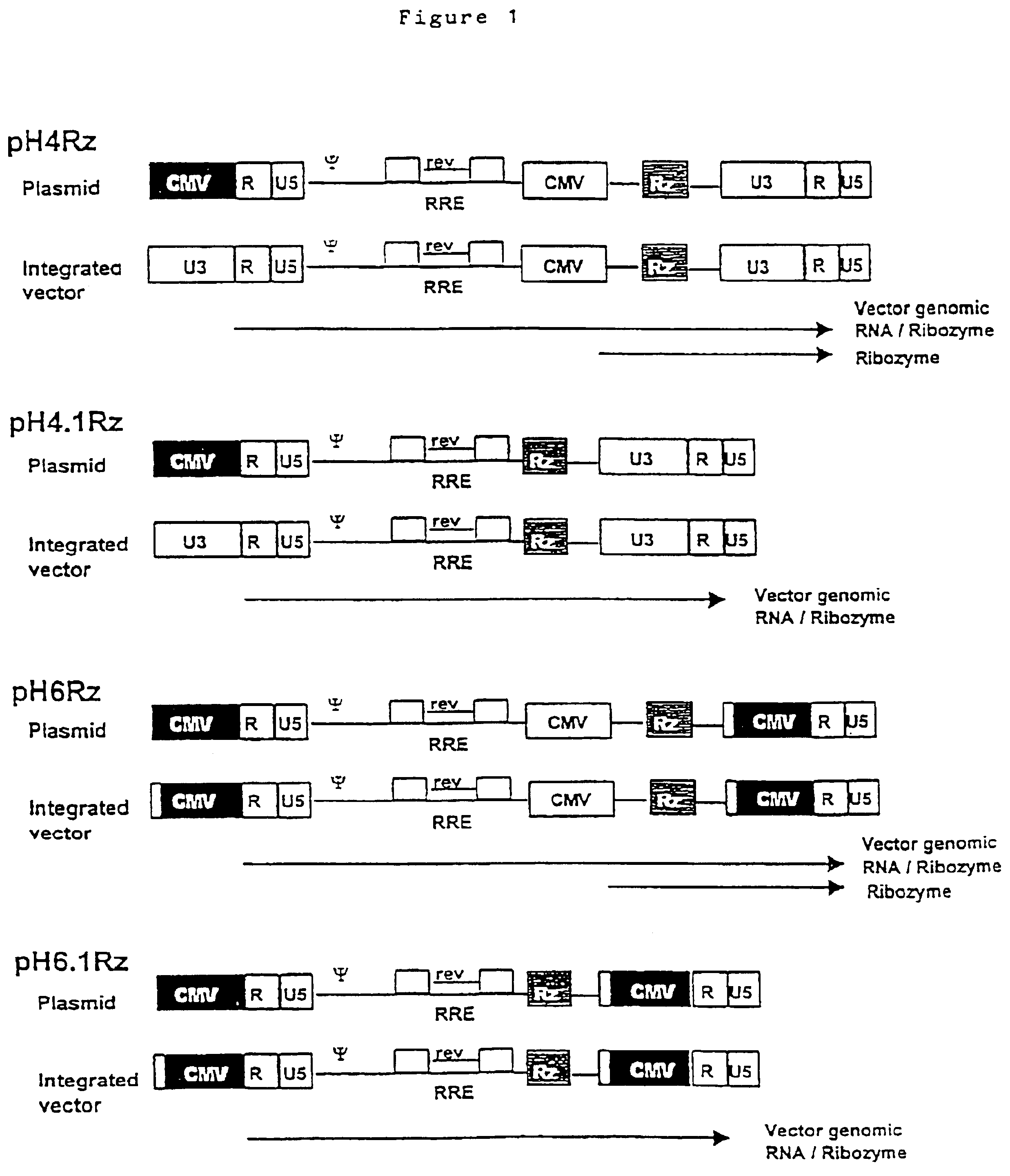
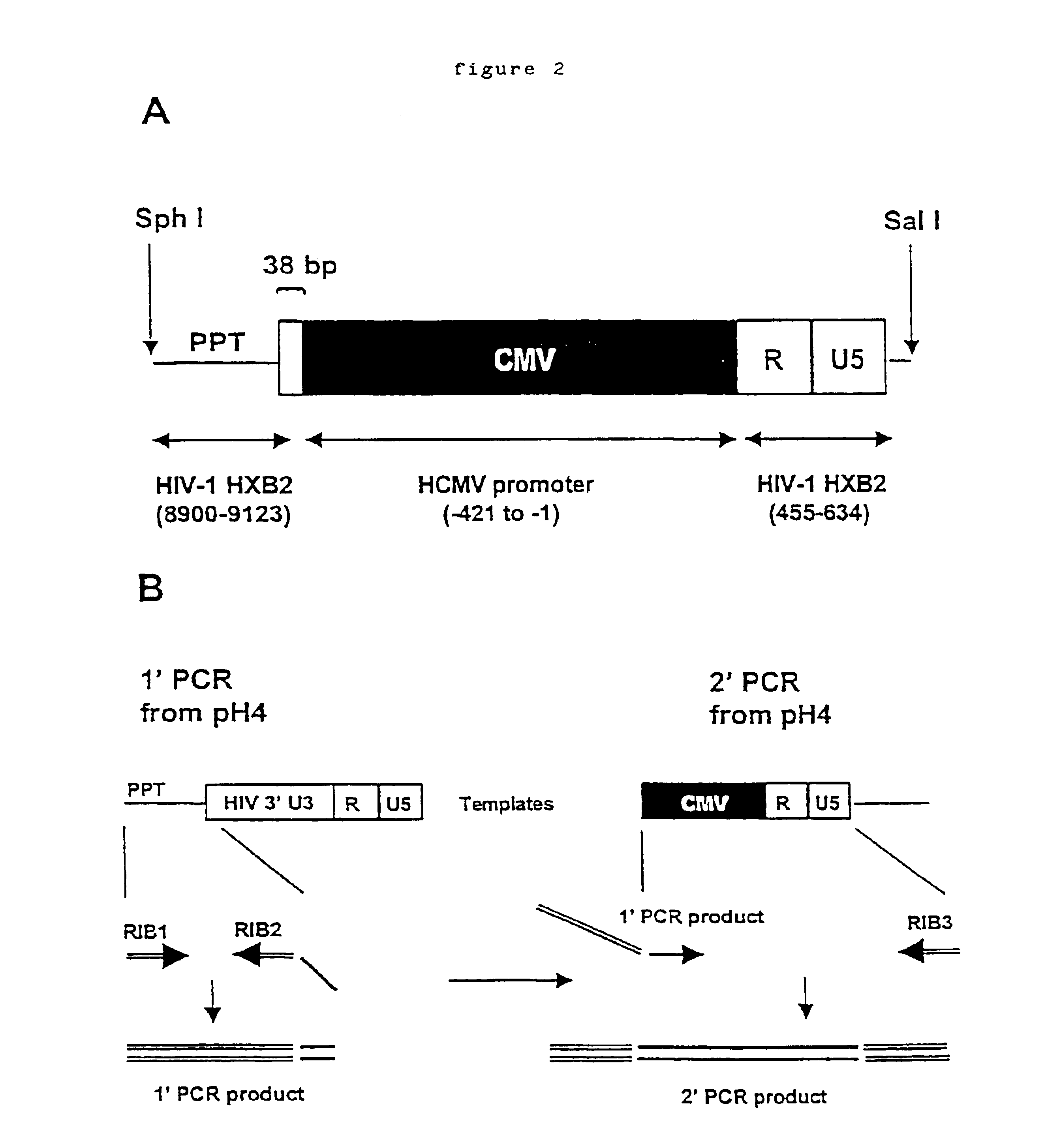
Anti-viral vectors
InactiveUS6783981B1Efficient exportPeptide/protein ingredientsGenetic material ingredientsNucleotideRNase P
A viral vector production system is provided which system comprises: (i) a viral genome comprising at least one first nucleotide sequence encoding a gene product capable of binding to and effecting the cleavage, directly or indirectly, of a second nucleotide sequence, or transcription product thereof, encoding a viral polypeptide required for the assembly of viral particles, (ii) a third nucleotide sequence encoding said viral polypeptide required for the assembly of the viral genome into viral particles, which third nucleotide sequence has a different nucleotide sequence to the second nucleotide sequence such that said third nucleotide sequence, or transcription product thereof, is resistant to cleavage directed by said gene product; wherein at least one of the gene products is an external guide sequence capable of binding to and effecting the cleavage by RNase P of the second nucleotide sequence. The viral vector production system may be used to produce viral particles for use in treating or preventing viral infection.
Owner:OXFORD BIOMEDICA (UK) LTD
Antiviral compounds
ActiveUS20060122123A1Increase concentrationEfficient targetingBiocideDipeptide ingredientsCombinatorial chemistryPhosphorus
The invention is related to phosphorus substituted anti-viral inhibitory compounds, compositions containing such compounds, and therapeutic methods that include the administration of such compounds, as well as to processes and intermediates useful for preparing such compounds.
Owner:GILEAD SCI INC
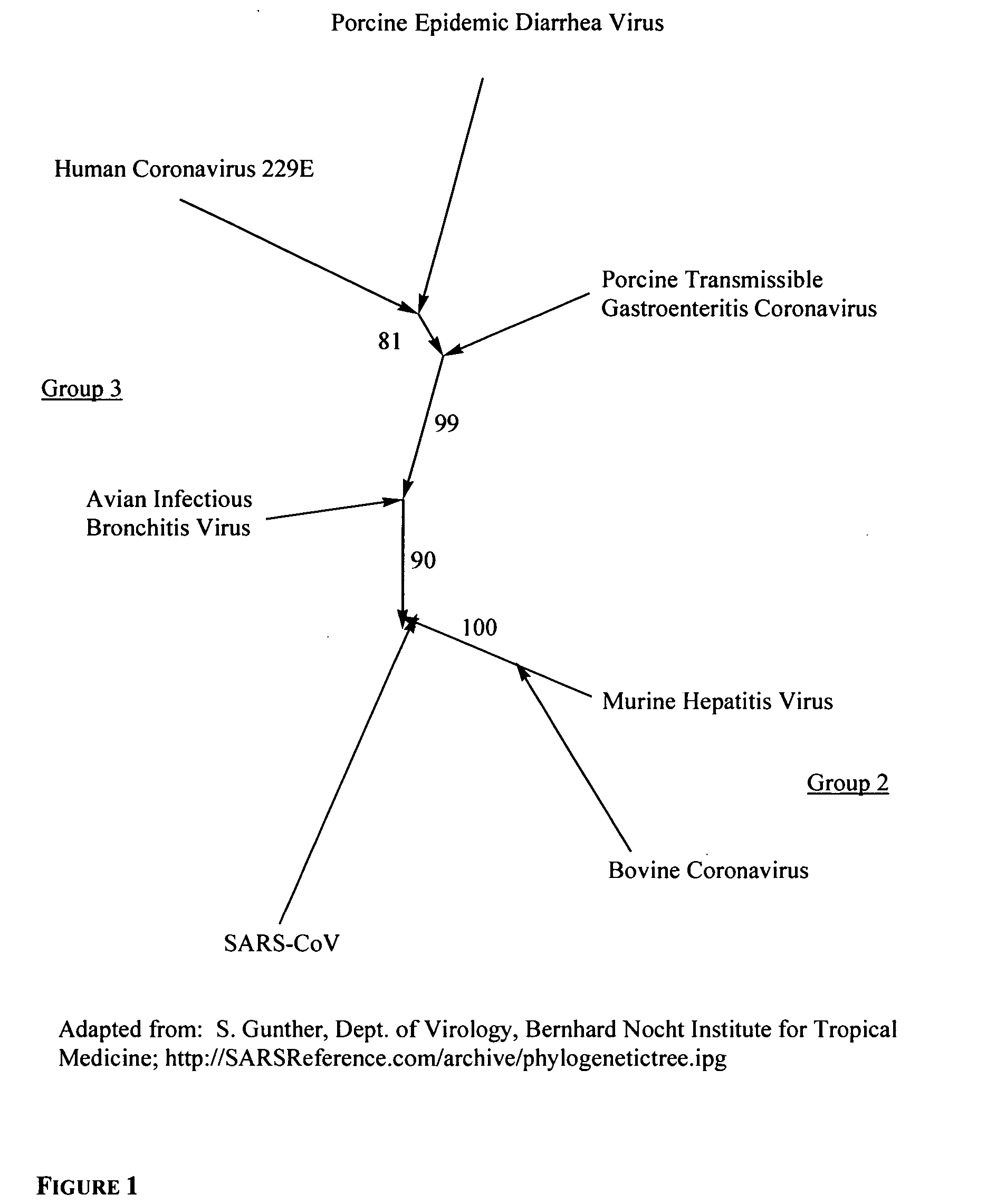
Phospholipids for the treatment of infection by togaviruses, herpes viruses and coronaviruses
InactiveUS20050187192A1BiocidePhosphorous compound active ingredientsHerpes simplex virus DNACompound (substance)
Provided are compounds, methods and pharmaceutical compositions for treating a host, especially a human, infected with a togavirus, herpes virus and / or coronavirus, and in particular SARS-CoV, cytomegalovirus or varicella-zoster virus. The method in one embodiment comprises administering to that host an effective amount of an anti-togavirus, anti-herpes virus and / or anti-coronavirus phospholipid or a pharmaceutically acceptable salt or prodrug thereof. The phospholipid compound is, e.g., a 3-alkylamido-2-alkoxypropylphosphocholine compound or salt thereof. The compound may be administered alone or in combination and / or alternation with one or more other anti-viral agents.
Owner:KUCERA PHARMA
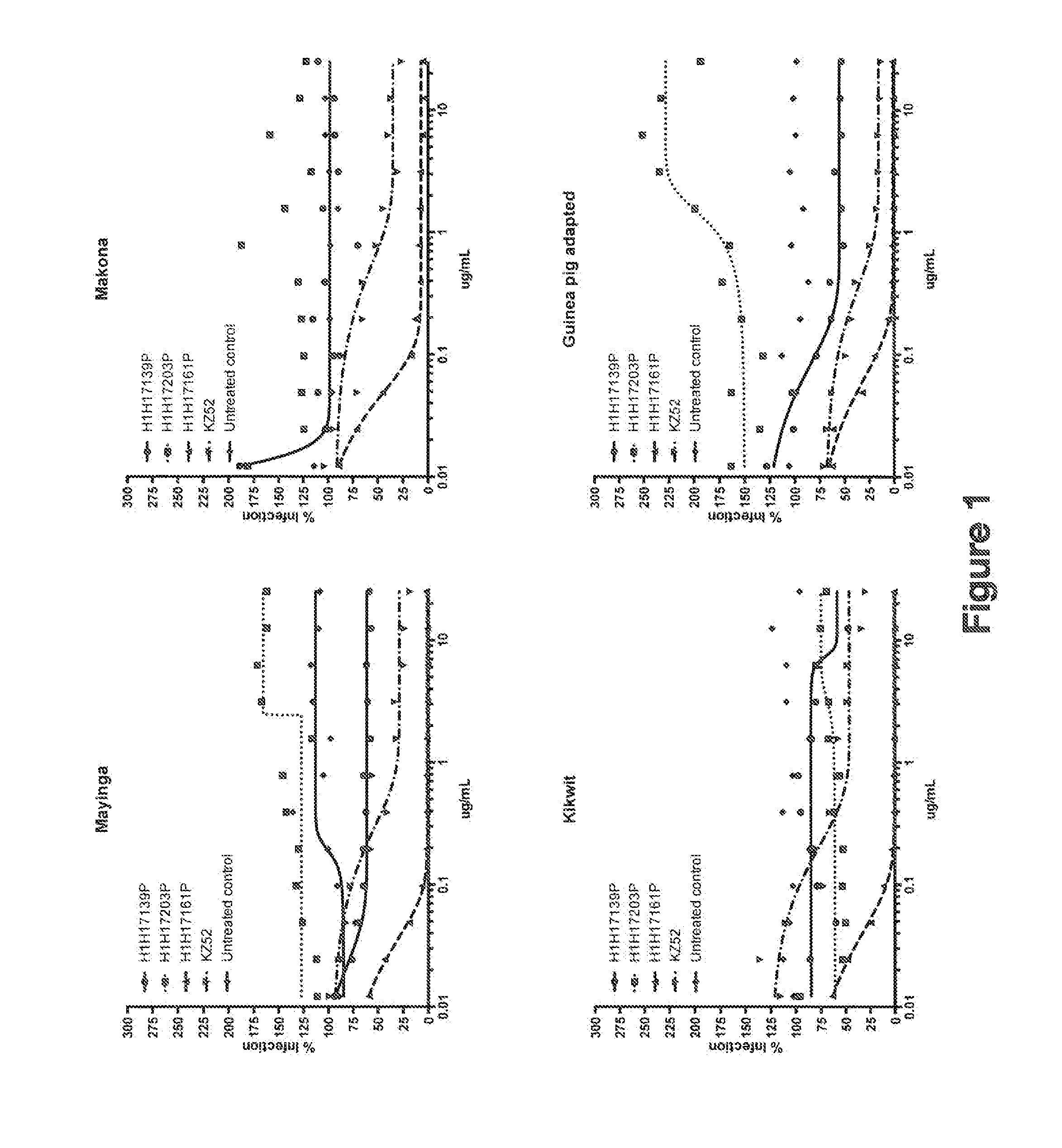
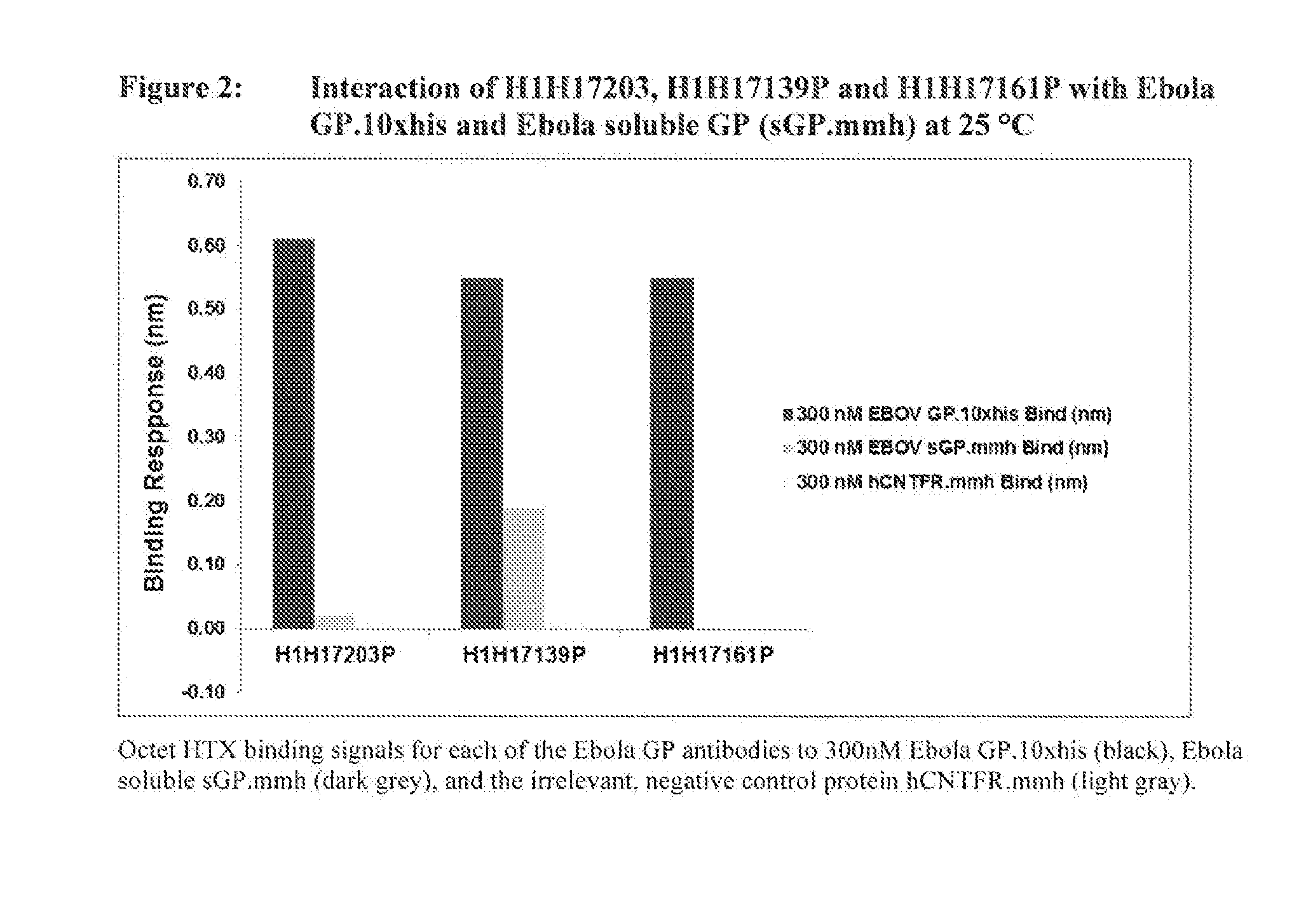
Human Antibodies to Ebola Virus Glycoprotein
ActiveUS20160215040A1Inhibiting and neutralizing activityAvoid enteringImmunoglobulins against virusesAntiviralsViral glycoproteinAntigen Binding Fragment
The present invention provides monoclonal antibodies, or antigen-binding fragments thereof, that bind to Ebola virus glycoproteins, pharmaceutical compositions comprising the antibodies and methods of use. The antibodies of the invention are useful for inhibiting or neutralizing Ebola virus activity, thus providing a means of treating or preventing Ebola virus infection in humans. In some embodiments, the invention provides for use of one or more antibodies that bind to the Ebola virus for preventing viral attachment and / or entry into host cells. The antibodies of the invention may be used prophylactically or therapeutically and may be used alone or in combination with one or more other anti-viral agents or vaccines.
Owner:REGENERON PHARM INC
Dosing regimen for Flaviviridae therapy
An anti-hepatitis C agent which is an anti-metabolite to the host and cannot be administered on a daily or chronic basis as is usual in anti-viral therapy (referred to below as an “anti-HCV anti-metabolite”), can be administered using a traditional anti-cancer dosing regimen (for example via intravenous or parenteral injection), over a period of one, two, three, four, five, six, or seven days followed by cessation of therapy until rebound of the viral load is noted. This dosing regimen runs counter to conventional antiviral experience, wherein effective agents are usually administered over at least fourteen days of sustained therapy, and typically on an indefinite daily basis.
Owner:PHARMASSET
Antiviral compounds
InactiveUS8178491B2Inhibitory and pharmacokinetic propertyImprove oral bioavailabilityBiocideDigestive systemCombinatorial chemistryAnti virals
Owner:GILEAD SCI INC
Purine nucleoside analogues for treating Flaviviridae including hepatitis C
This invention is directed to a method for treating a host, especially a human, infected with hepatitis C, flavivirus and / or pestivirus, comprising administering to that host an effective amount of an anti-HCV biologically active pentofuranonucleoside where the pentofuranonucleoside base is an optionally substituted 2-azapurine. The optionally substituted pentofuranonucleoside, or a salt or prodrug thereof, may be administered alone or in combination with one or more optionally substituted pentofuranonucleosides or other anti-viral agents.
Owner:THE CENT NAT DEL LA RECH SCIQUE +2
Long Acting Biologically Active Conjugates
InactiveUS20070207952A1High level of drugPoor adhesionBiocidePeptide/protein ingredientsImmunodeficiency virusIn vivo
The invention provides biologically active compounds that may be reacted with macromolecules, such as albumin, to form covalent linked complexes wherein the resulting complexes exhibit a desired biological activity in vivo. More specifically, the complexes are isolated complexes comprising a biologically active moiety covalently bound to a linking group and a protein. The complexes are prepared by conjugating a biologically active moiety, for example, a renin inhibitor or a viral fusion inhibitor peptide, with purified and isolated protein. The complexes have extended lifetimes in the bloodstream as compared to the unconjugated molecule, and exhibit biological activity for extended periods of time as compared to the unconjugated molecule. The invention also provides anti-viral compounds that are inhibitors of viral infection and / or exhibit anti-fusiogenic properties. In particular, this invention provides compounds having inhibiting activity against viruses such as human immunodeficiency virus (HIV), respiratory syncytial virus (RSV), human parainfluenza virus (HPV), measles virus (MeV), and simian immunodeficiency virus (SIV) and that have extended duration of action for the treatment of viral infections.
Owner:SEQUOIA PHARMACEUTICALS INC
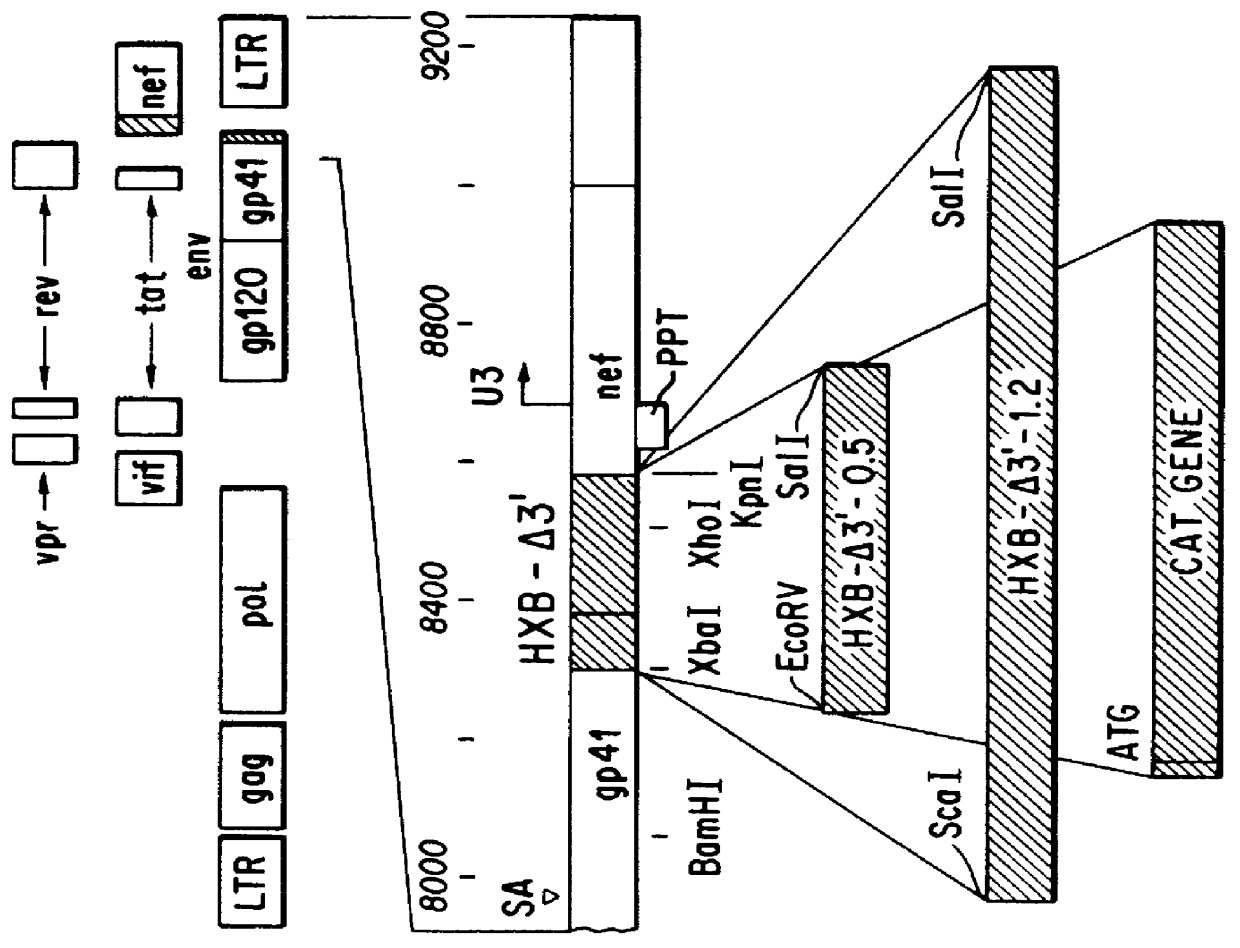
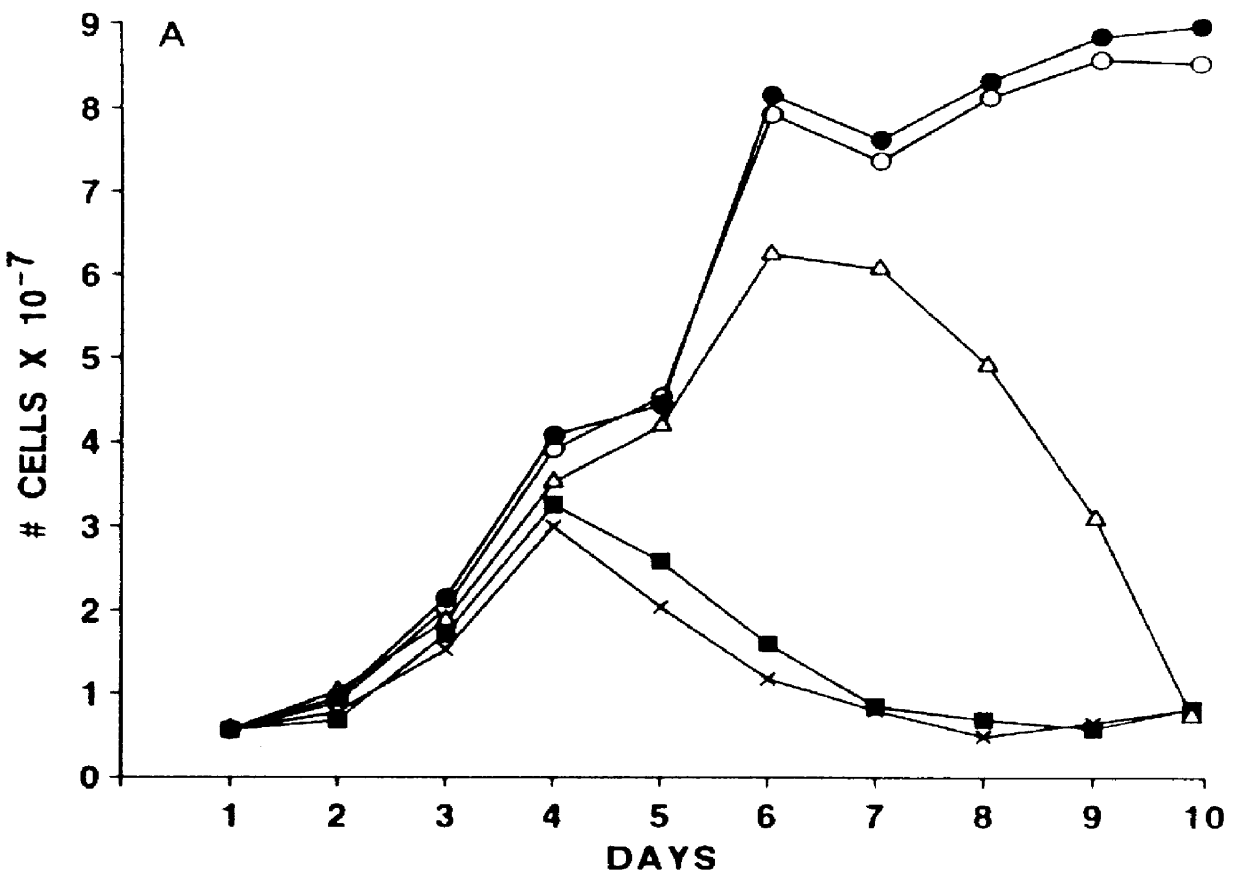
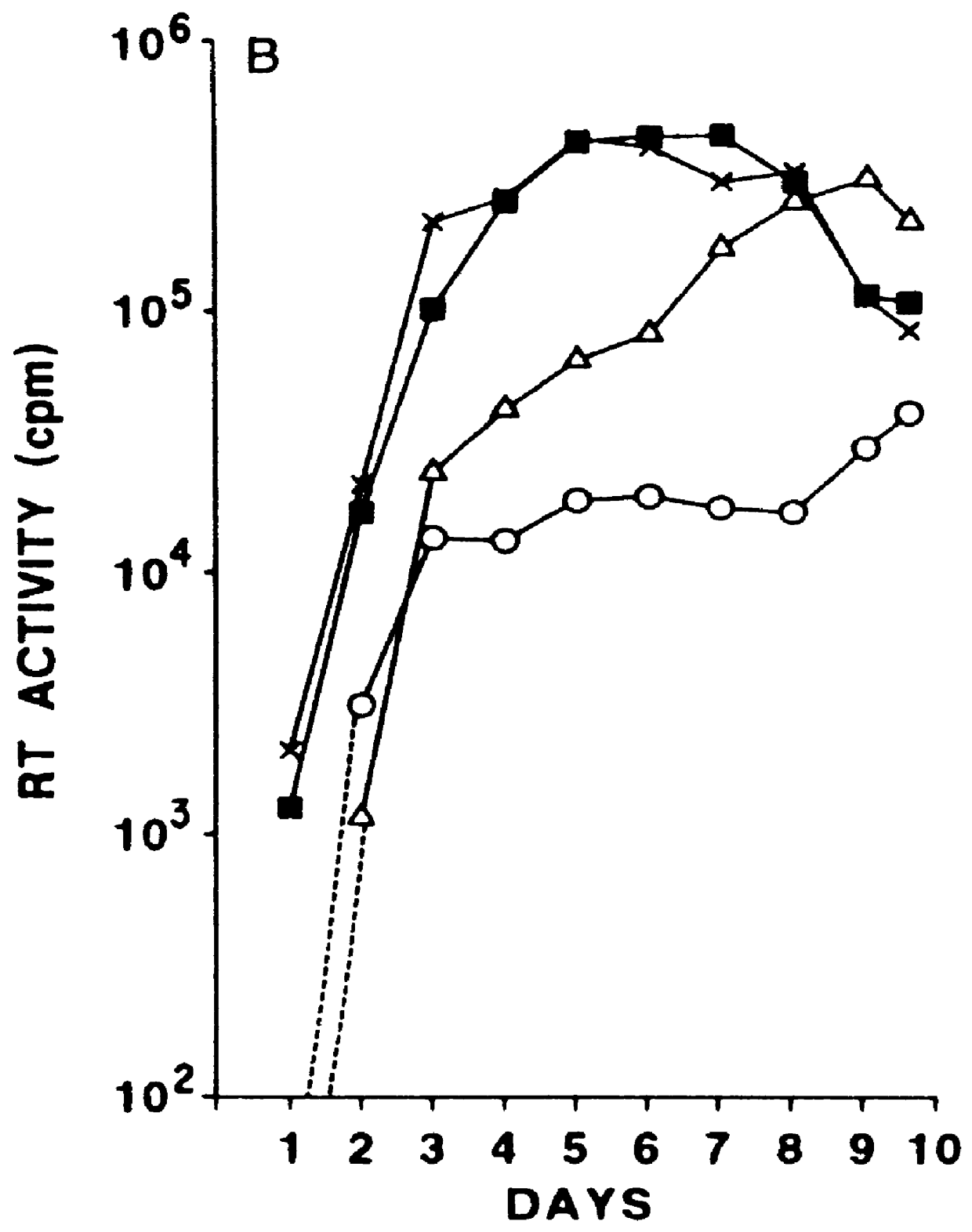
Vector comprising a replication competent HIV-1 provirus and a heterologous gene
InactiveUS6033902AQuick analysisSmall sizeMicrobiological testing/measurementVirus peptidesHeterologousRetroviral provirus
A vector comprising an HIV segment and a heterologous gene segment, which produces a replication competent and an infective HIV virus is disclosed. When the heterologous gene is a marker gene, the spread of the virus can be observed in both in vitro and in vivo systems. The use of this vector in establishing methods for screening anti-viral compounds is also disclosed.
Owner:DANA FARBER CANCER INST INC
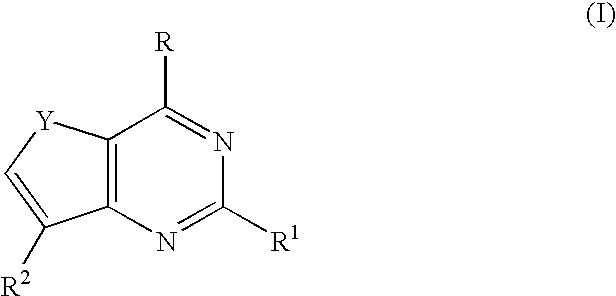
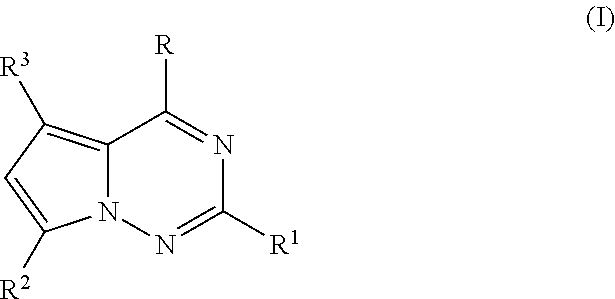
Therapeutic furopyrimidines and thienopyrimidines
The invention provides compounds of formula I, II, and III as described herein, as well as pharmaceutical compositions comprising the compounds, and synthetic methods and intermediates that are useful for preparing the compounds. The compounds of formula I, II, and III are useful as anti-viral agents and / or as anti-cancer agents.
Owner:BIOCRYST PHARM INC
Anti-viral treatment and assay to screenfor Anti-viral agent
InactiveUS20130085133A1Improve palatabilityImprove stabilityBiocideSugar derivativesCytopathic effectAntiviral drug
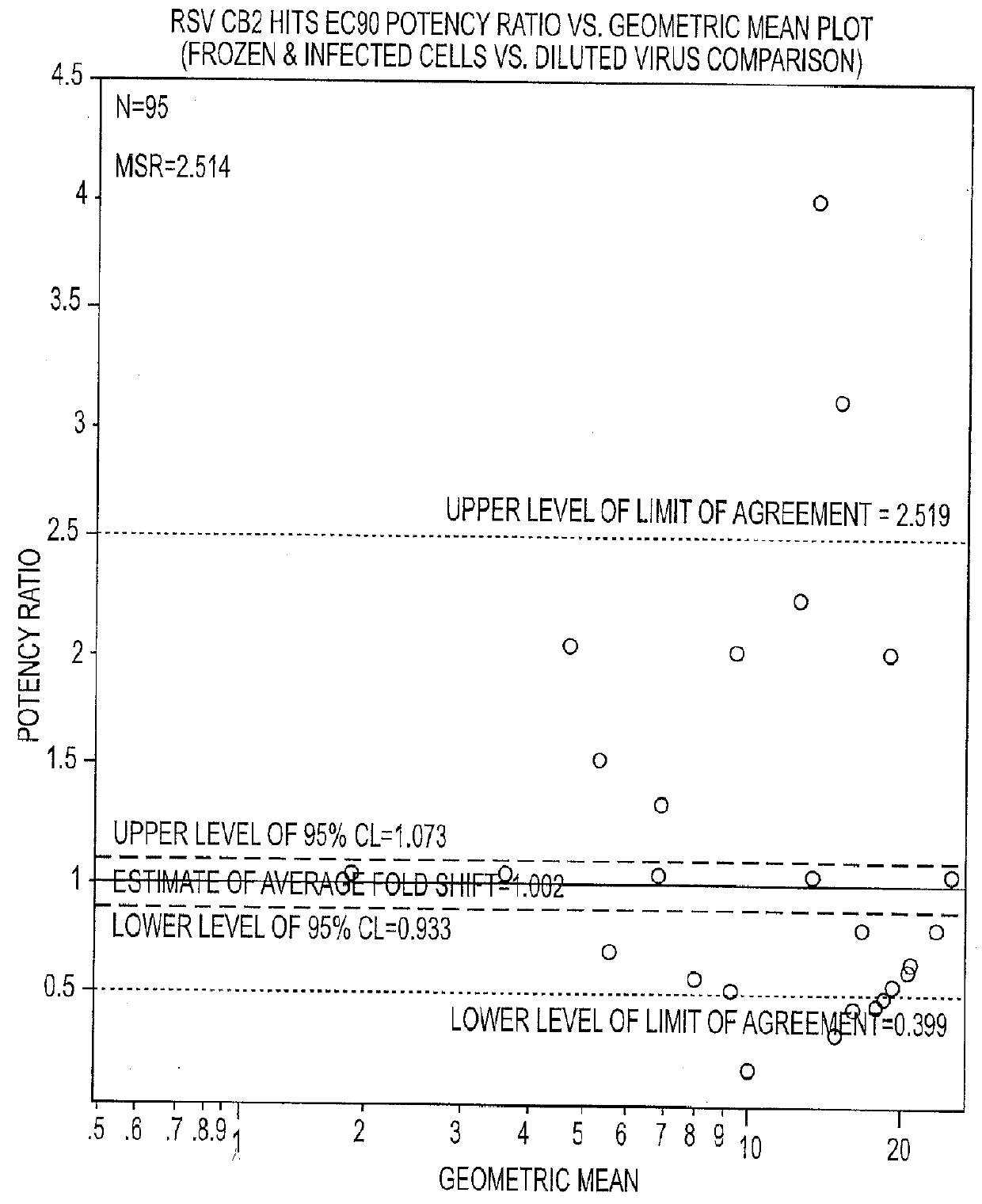
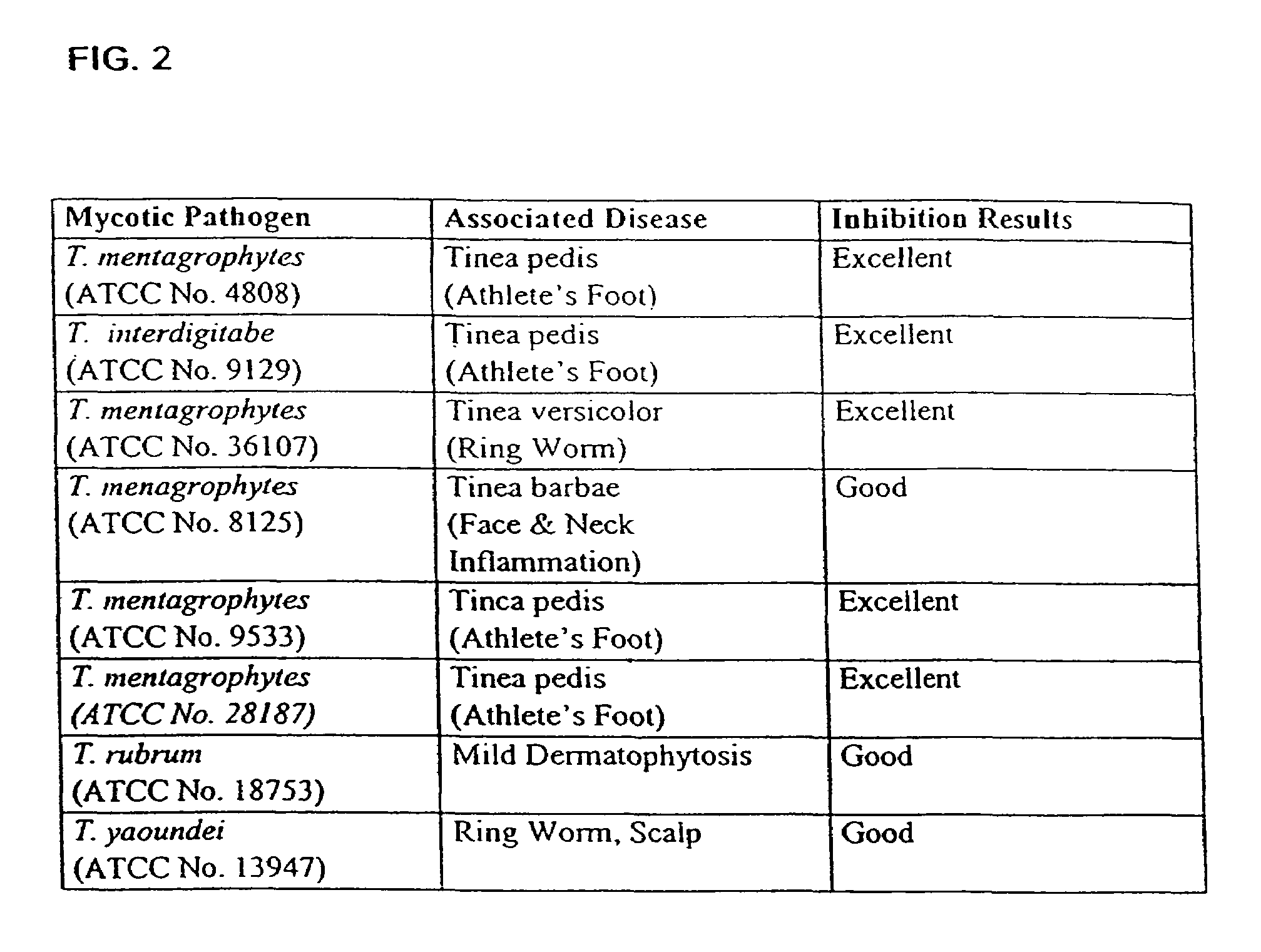
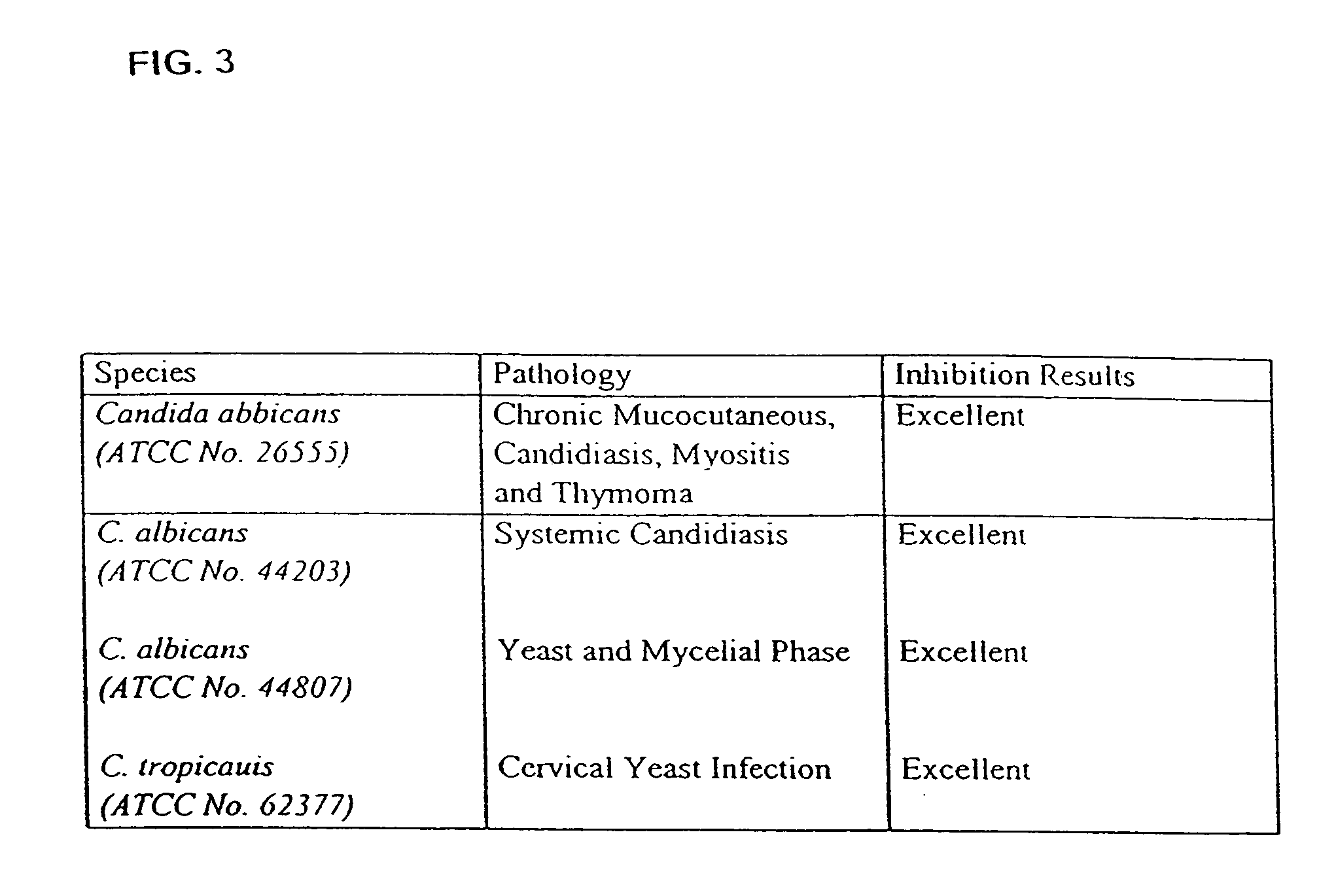
The present disclosure relates to novel compounds of formulas (1) through (19) and to a method for treating humans infected with a virus including various respiratory viruses such as members of the Paramyxoviridae family (respiratory syncytial virus (RSV), human metapneumovirus (HMPV), human parainfluenza virus (HPIV), measles virus, and mumps virus) with a compound of formulas (1) through (19). The present disclosure also relates to a cytopathic effect (CPE)-based assay that will assess virus-induced CPE for screening of compounds for treating viral diseases or inhibiting a virus.
Owner:SOUTHERN RES INST & IP
Probiotic, lactic acid-producing bacteria and uses thereof
InactiveUS7708988B2Good curative effectMitigating deleterious side-effectsAntibacterial agentsBiocideDiseaseMicrobial agent
The present invention discloses compositions and methodologies for the utilization of probiotic organisms in therapeutic compositions. More specifically, the present invention relates to the utilization of one or more species or strains of lactic acid-producing bacteria, preferably strains of Bacillus coagulans, for the control of gastrointestinal tract pathogens, including antibiotic-resistant gastrointestinal tract pathogens, and their associated diseases by both a reduction in the rate of colonization and the severity of the deleterious physiological effects of the colonization of the antibiotic-resistant pathogen. In addition, the present invention relates to the utilization of therapeutic compounds comprised of lactic acid-producing bacteria and anti-microbial agents such as antibiotics, anti-fungal compounds, anti-yeast compounds, or anti-viral compounds. The present invention also discloses methodologies for: (i) the selective breeding and isolation of probiotic, lactic acid-producing bacterial strains which possess resistance or markedly decreased sensitivity to anti-microbial agents (e.g., antibiotics, anti-fungal agents, anti-yeast agents, and anti-viral agents); and (ii) treating or preventing bacteria-mediated infections of the gastrointestinal tract by use of the aforementioned probiotic bacterial strains with or without the concomitant administration of antibiotics. While the primary focus is on the treatment of gastrointestinal tract infections, the therapeutic compositions of the present invention may also be administered to buccal, vaginal, optic, and like physiological locations.
Owner:GANEDEN BIOTECH
Antiviral nucleoside analogs
ActiveUS20100015094A1Useful in preparationBiocideSaccharide with heterocyclic radicalsAnticarcinogenCombinatorial chemistry
The invention provides compounds of Formula (I), as described herein, as well as pharmaceutical compositions comprising the compounds, and synthetic methods and intermediates that are useful for preparing the compounds. The compounds of Formula (I) are useful as anti-viral agents and / or as anti-cancer agents.
Owner:BIOCRYST PHARM INC
Anti-Viral Compounds
InactiveUS20130045229A1Less side effectsLess susceptibleBiocideOrganic active ingredientsRNA Virus InfectionsViral infection
Disclosed herein are compounds and related compositions for the treatment of viral infection, including RNA viral infection, and compounds that can modulate the RIG-I pathway in vertebrate cells, including compounds that can activate the RIG-I pathway.
Owner:KINETA
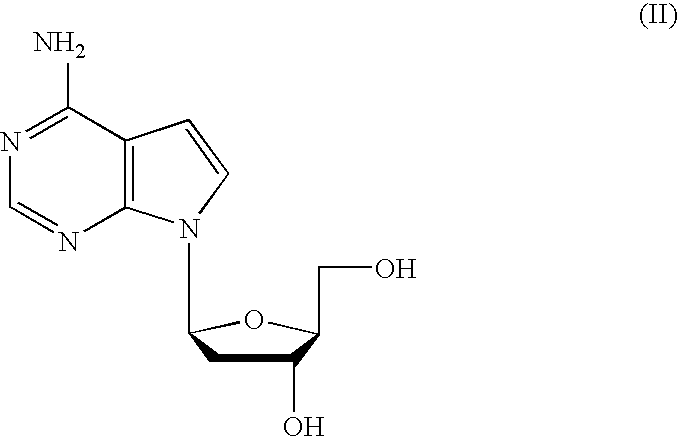
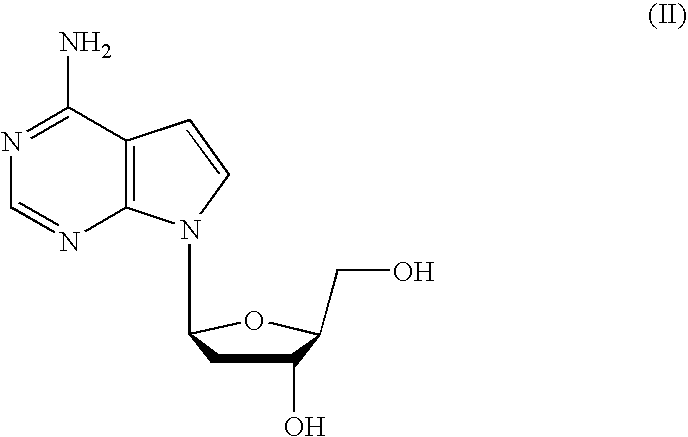
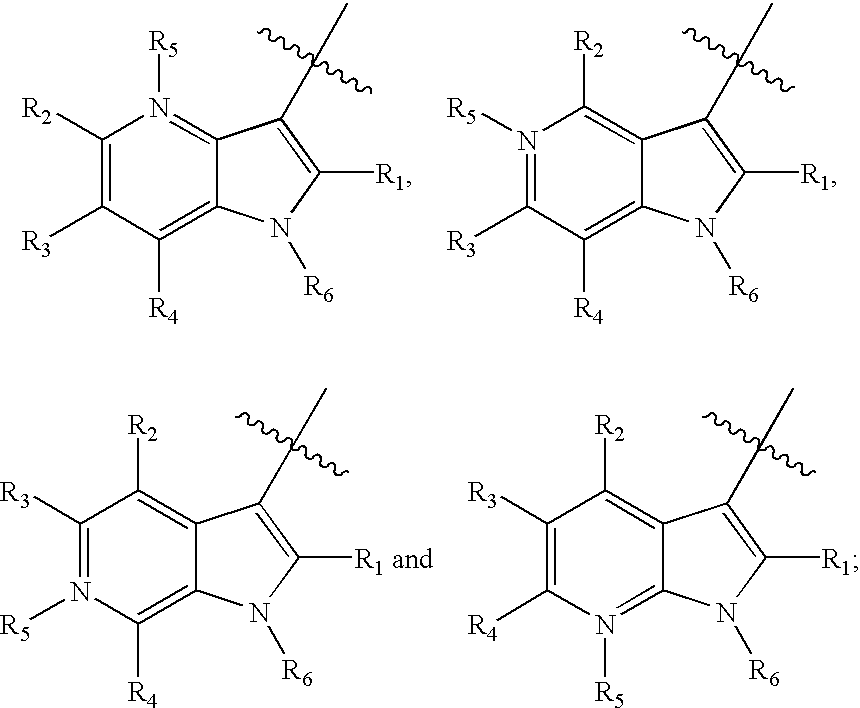
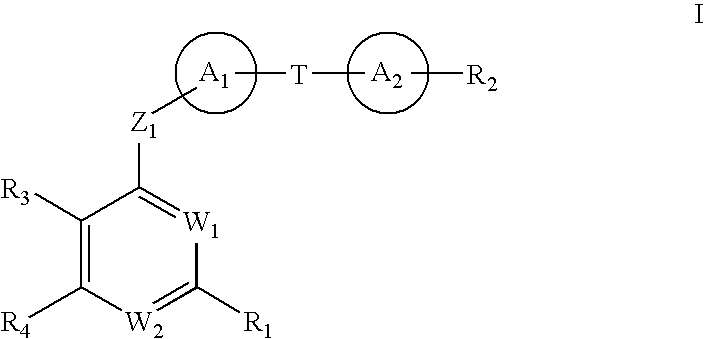
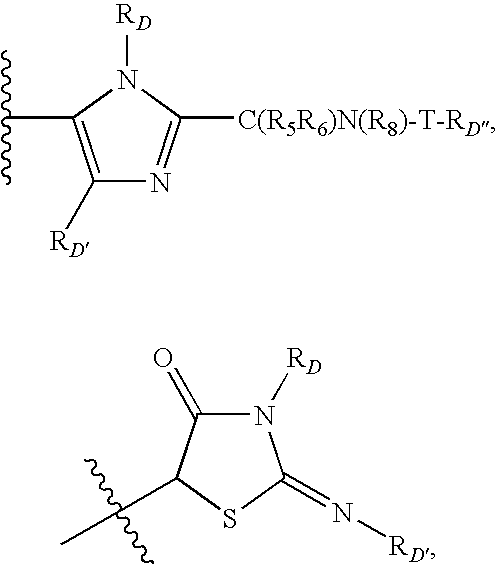
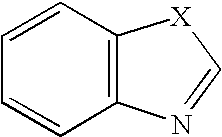
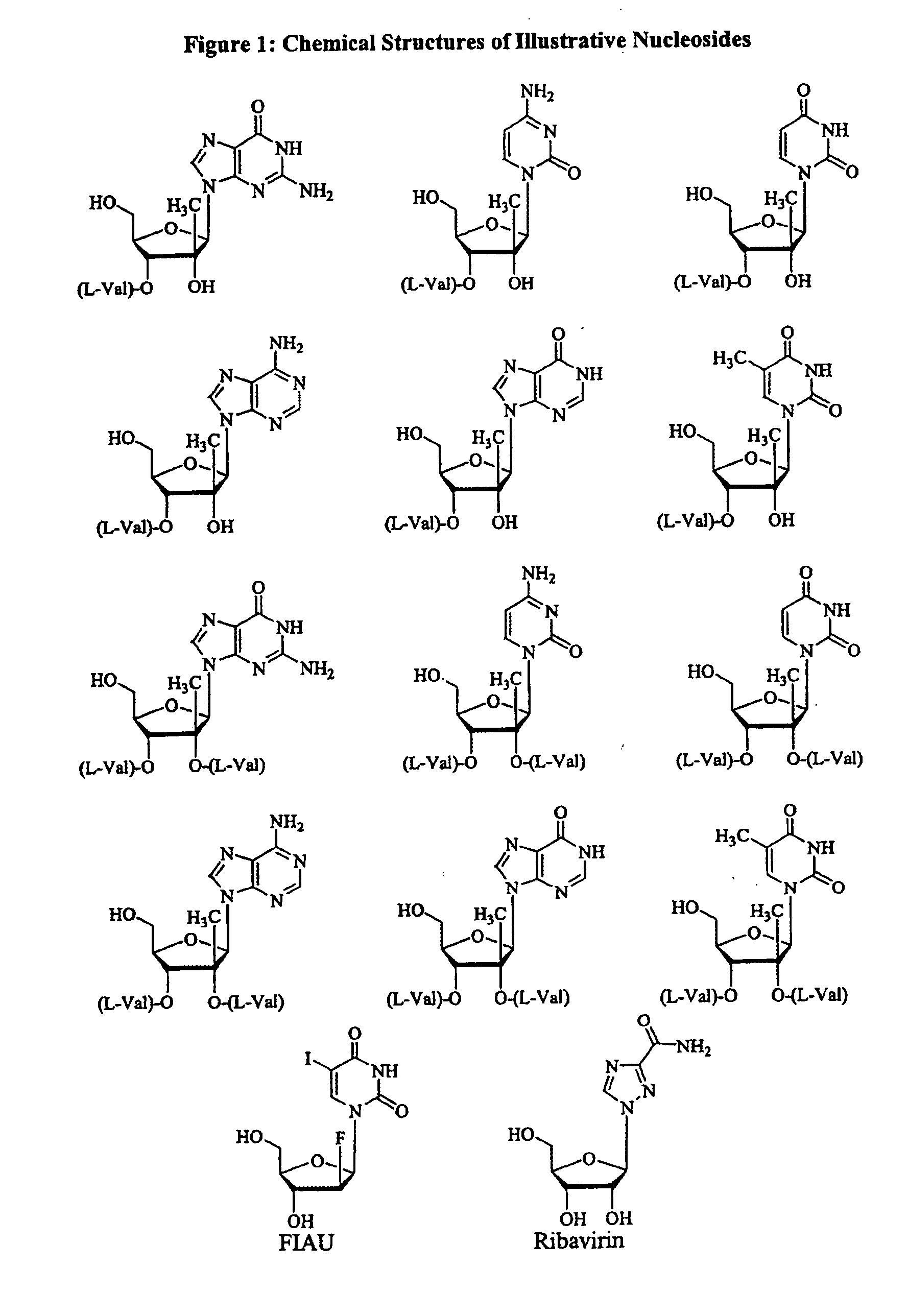
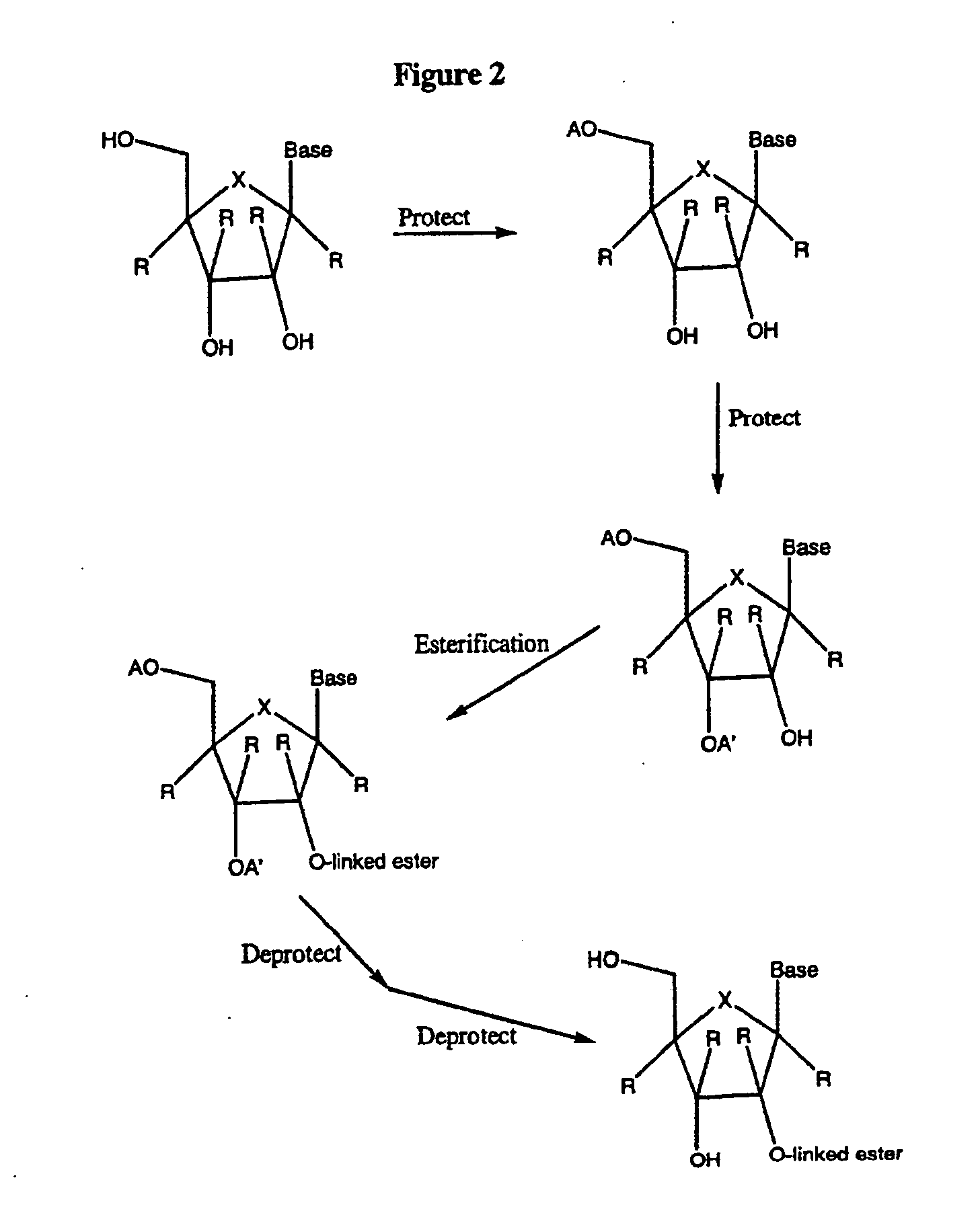
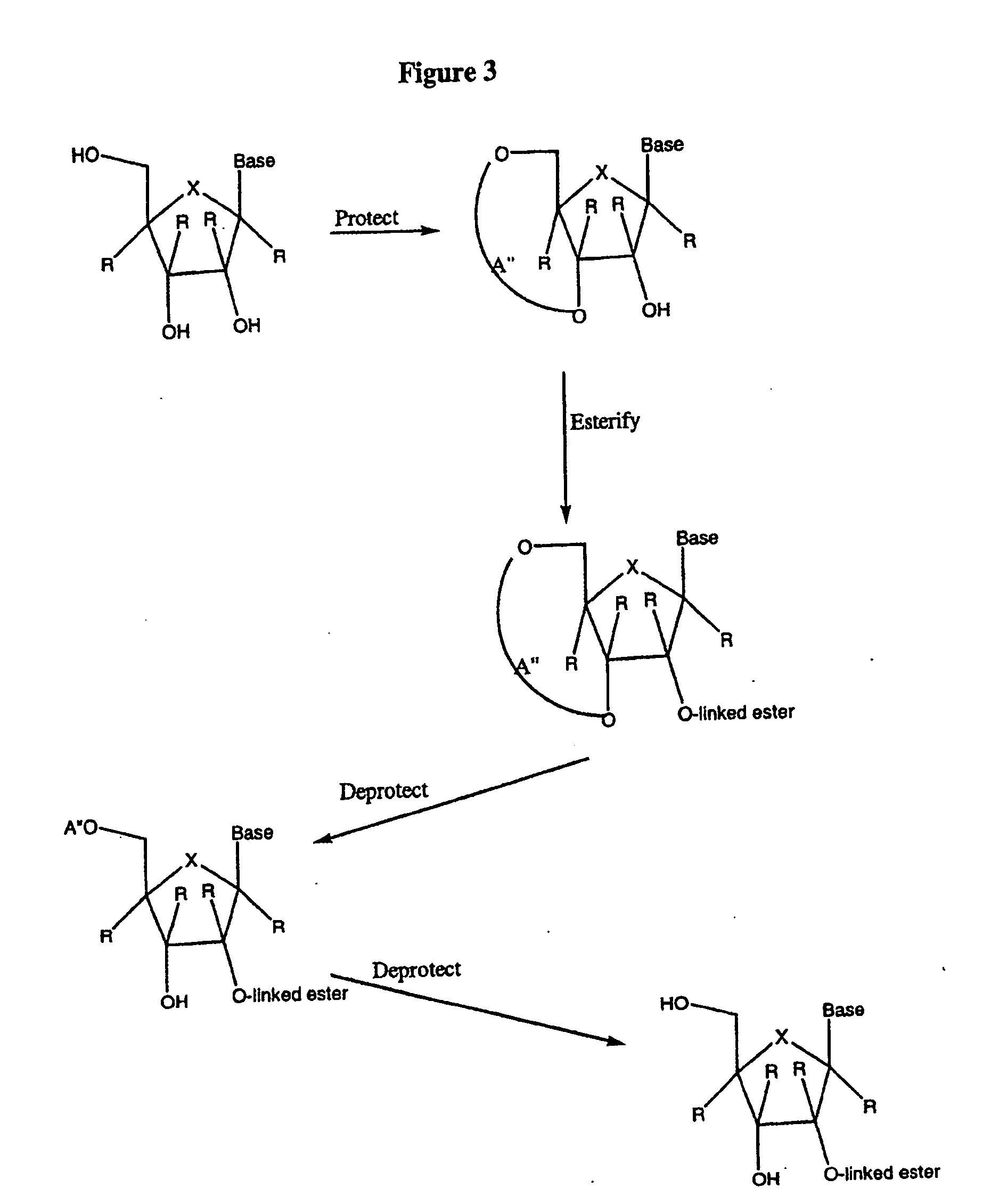
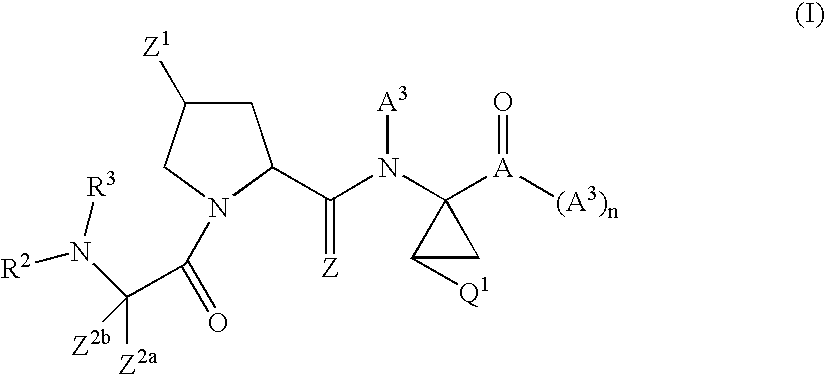
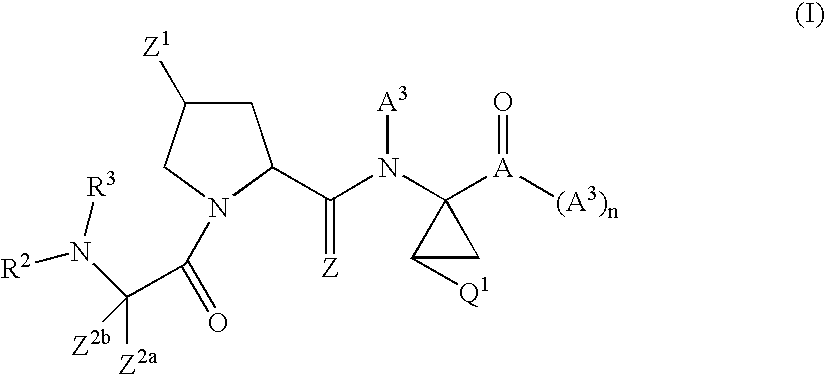
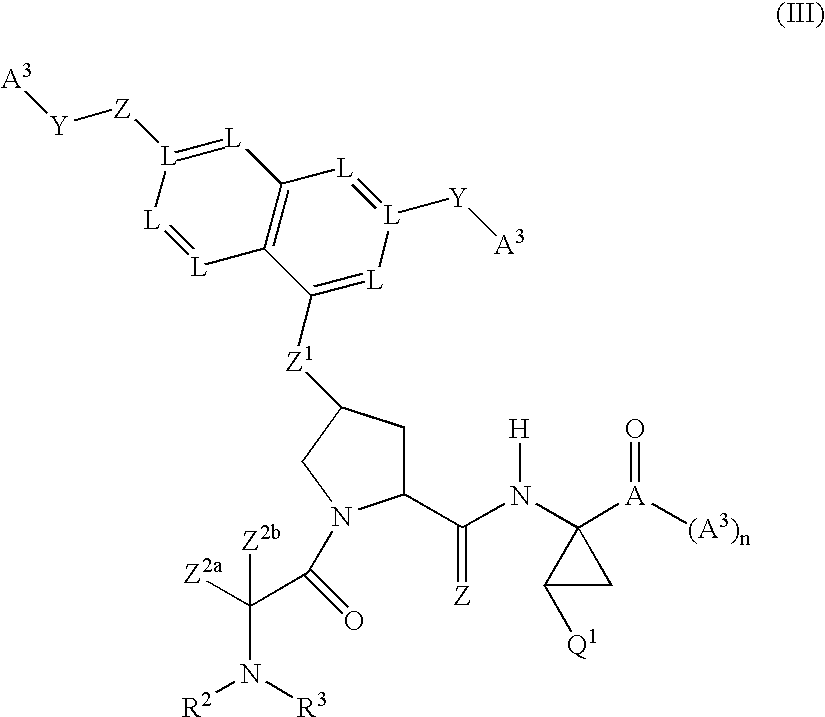
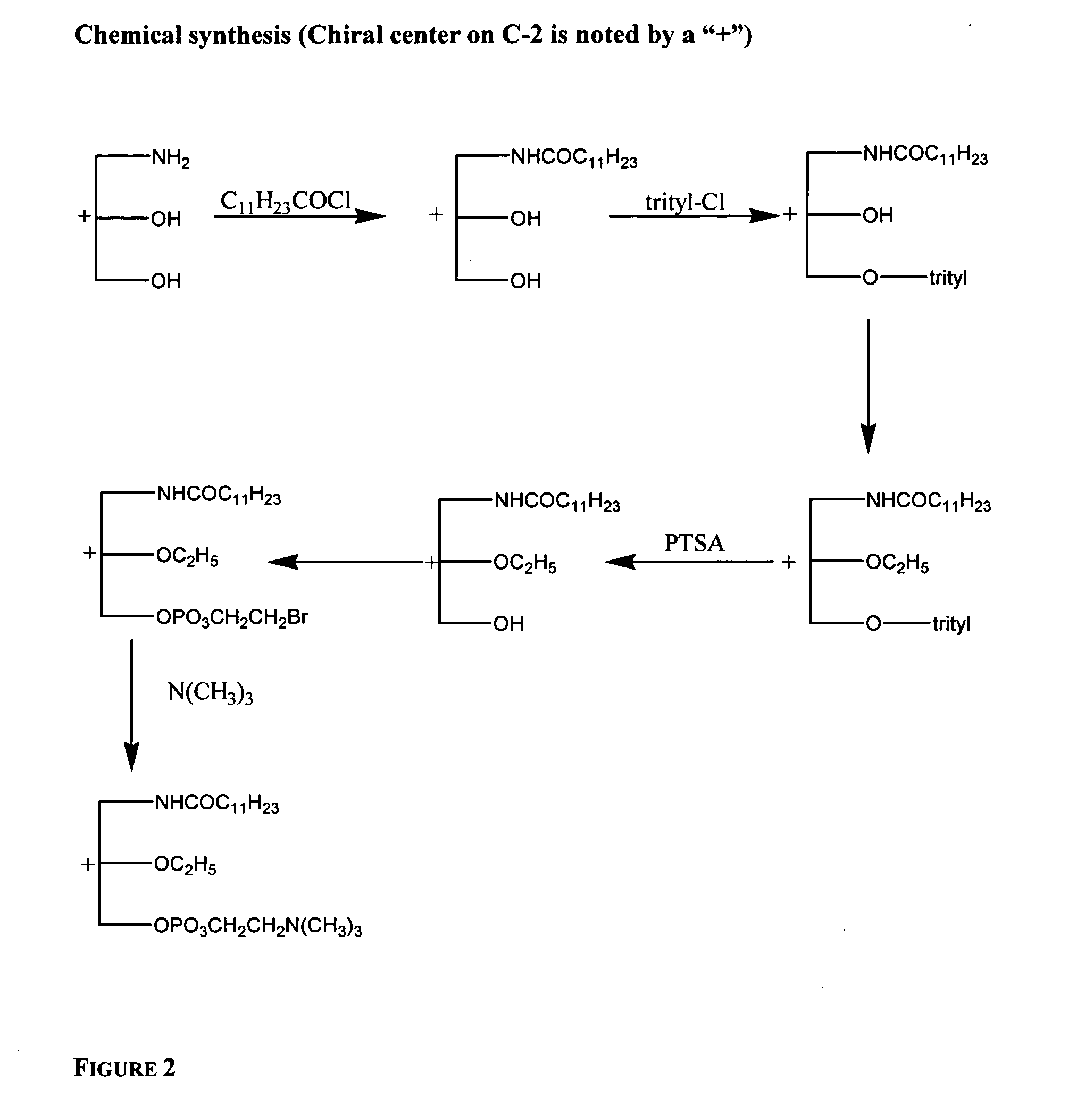
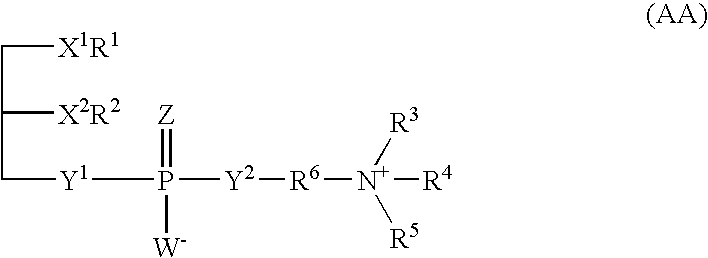
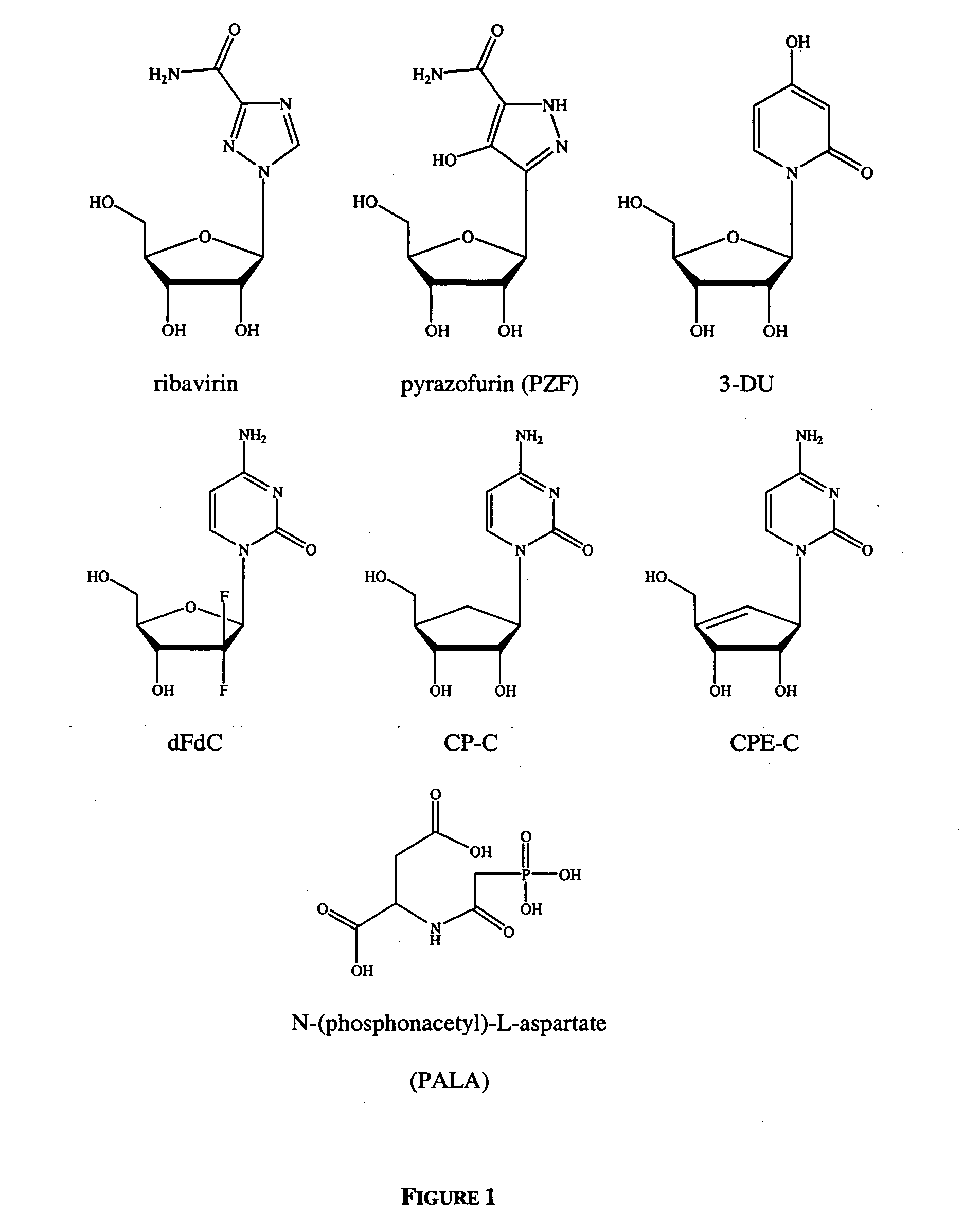
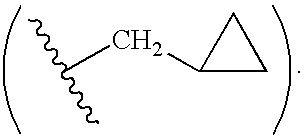
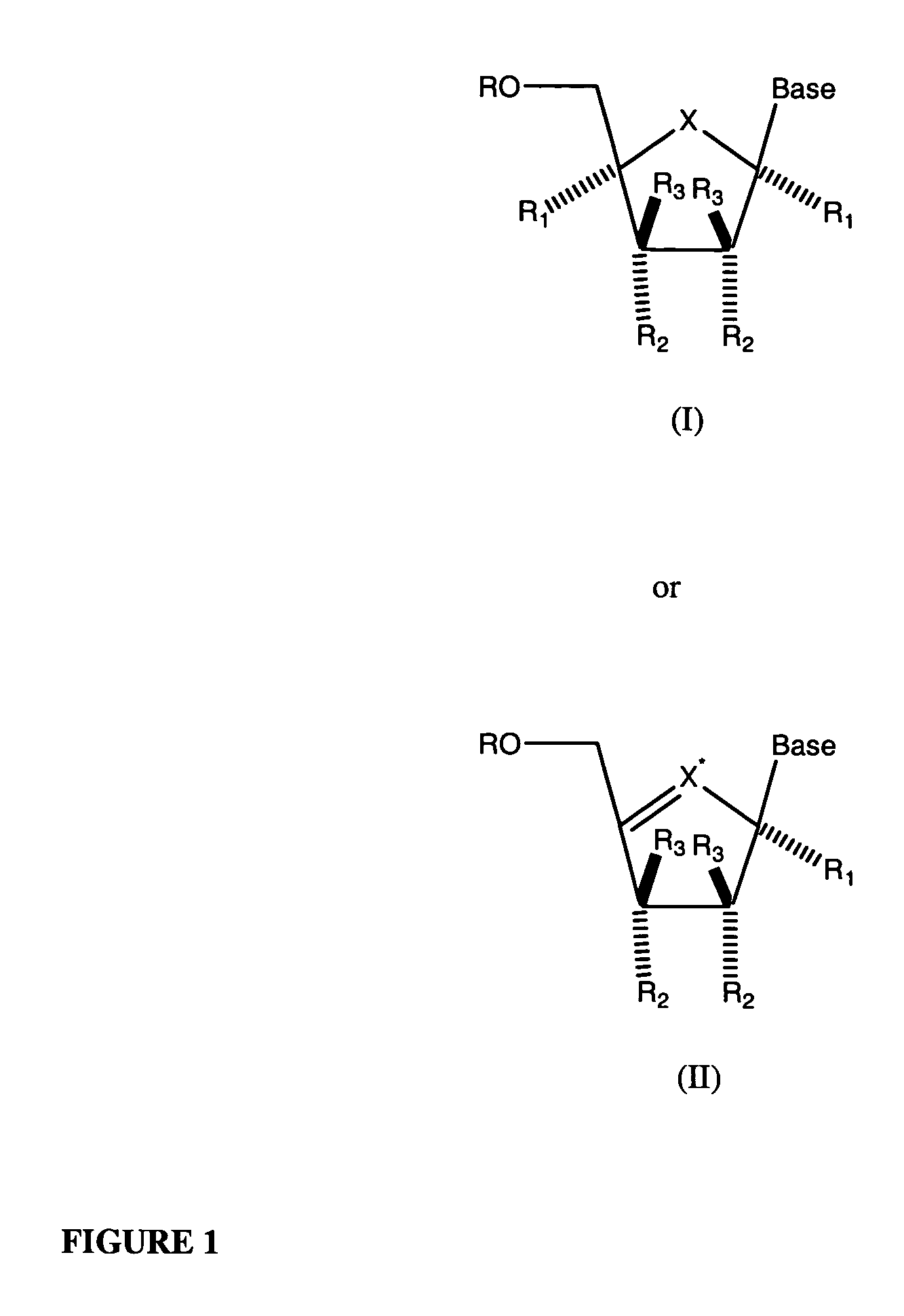
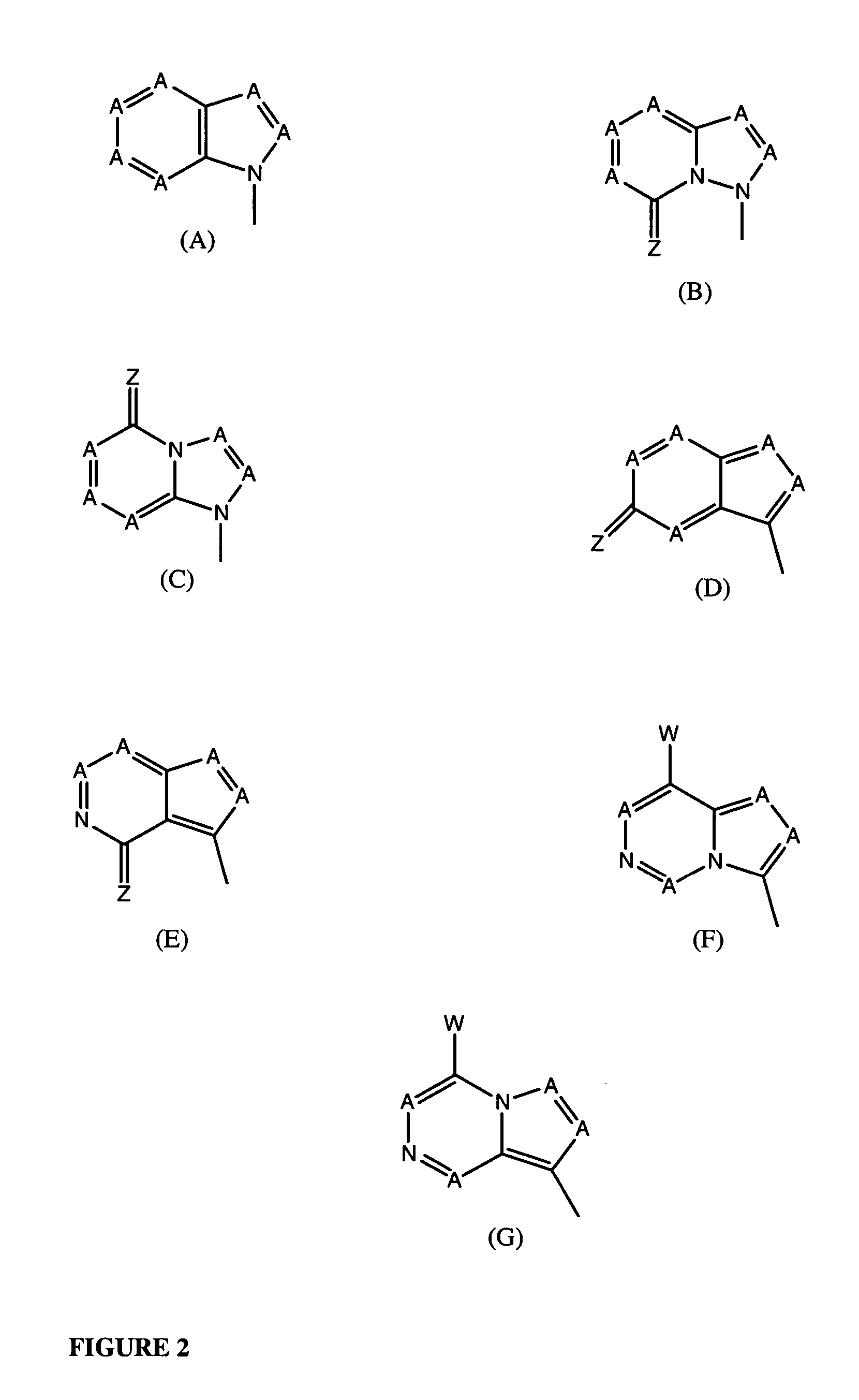
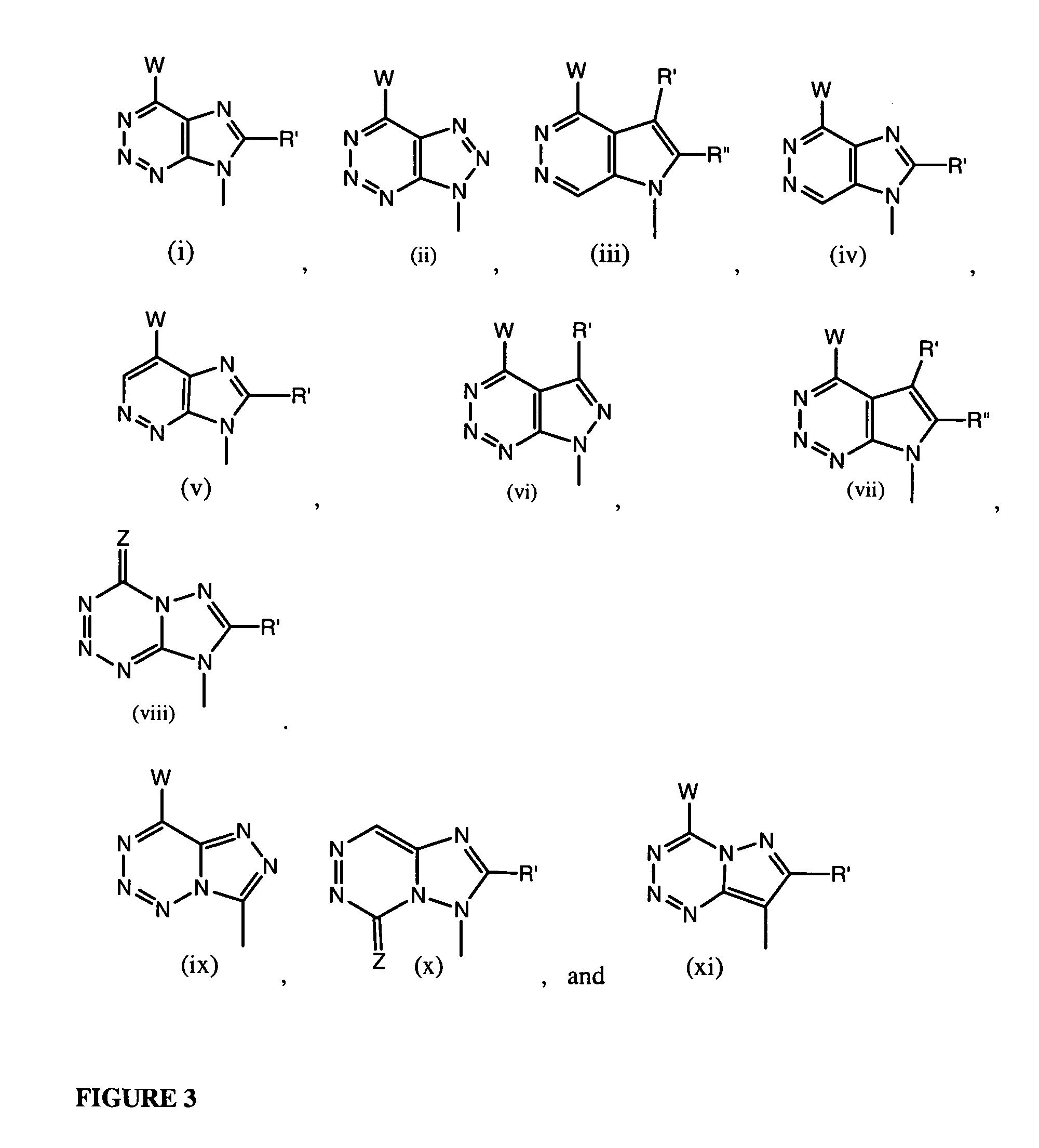
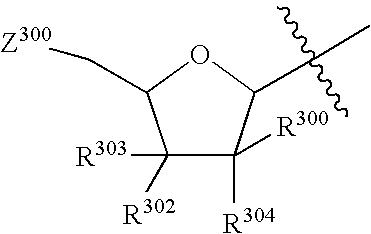
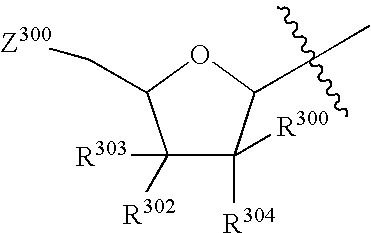
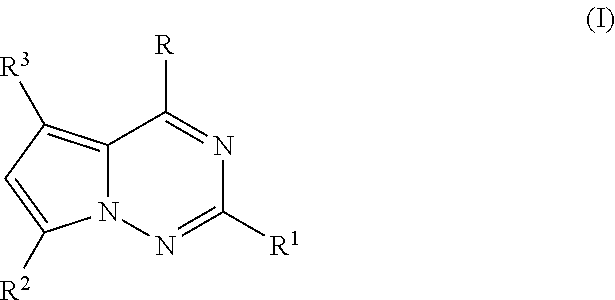
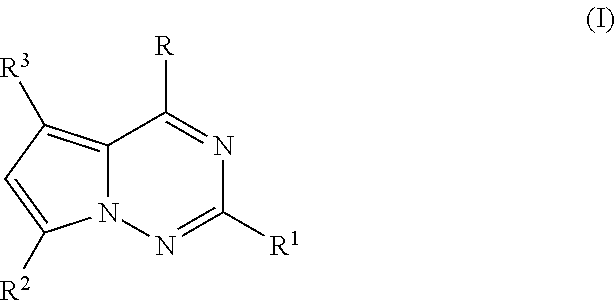
Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer
Owner:BIOCRYST PHARM INC
Anti-viral 7-deaza D-nucleosides and uses thereof
The present invention relates generally to anti-viral compounds, particularly anti-viral 7-deaza D-nucleosides and analogues, or derivatives thereof. The invention also relates to the use of such compounds to treat or prevent hepatitis B virus (HBV) infections, and to the use of such compounds to examine the biological mechanisms of HBV infection.
Owner:MIGENIX INC (CA)
Anti-viral 7-deaza L-nucleosides
The present invention comprises 7-deaza L-nucleosides having unexpectedly high inhibitory activity against the hepatitis B virus. The invention further comprises pharmaceutical compositions comprising such compounds as well as methods of treating mammals, particularly humans, infected with HBV and other viral infections.
Owner:MICROLOGIX BIOTECH INC
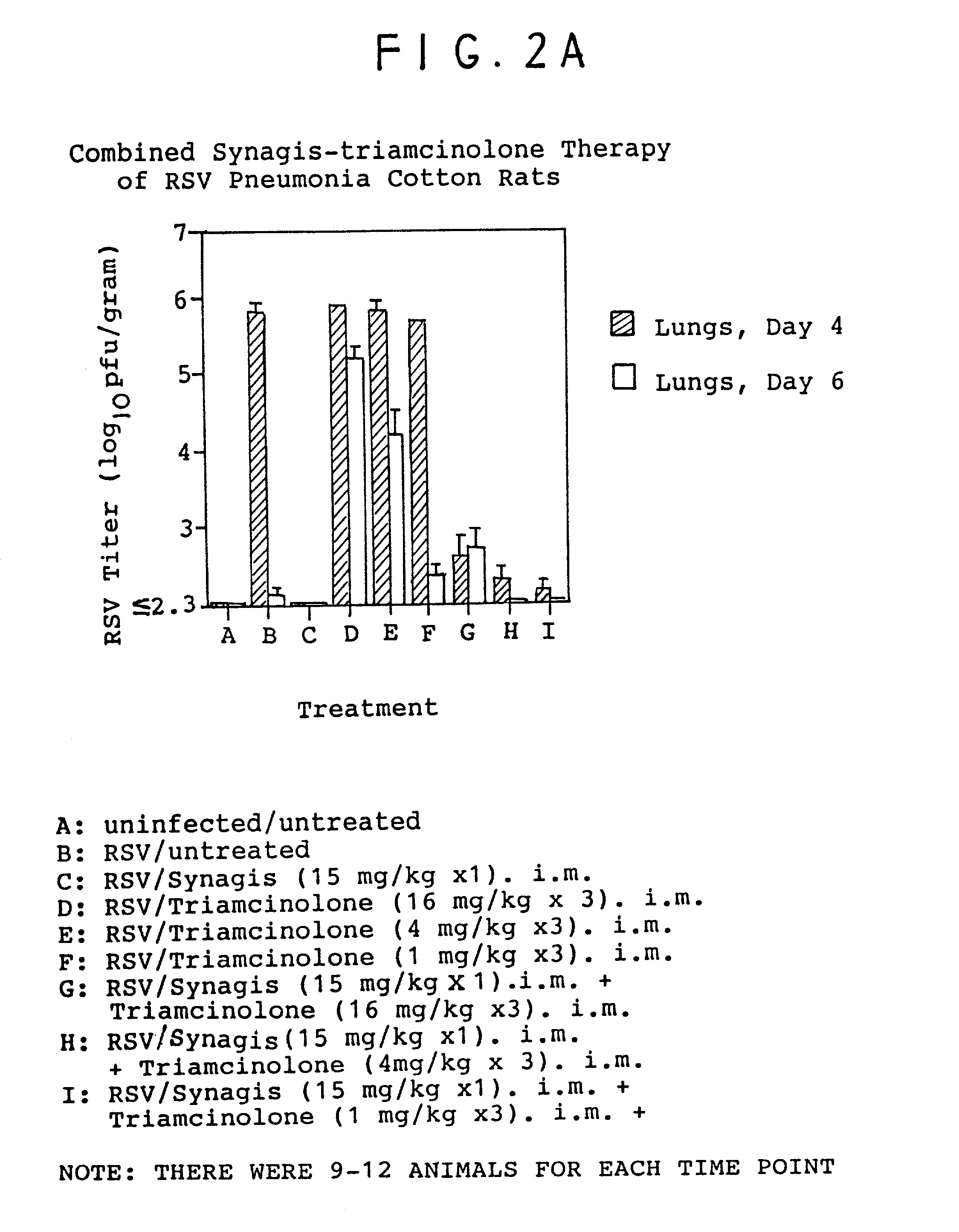
Combination therapy of respiratory diseases using antibodies and anti-inflammatory agents
InactiveUS7208162B2Useful in treatmentAntibacterial agentsImmunoglobulins against virusesInterleukin 6Disease
Therapeutically effective anti-viral compositions, useful especially against respiratory diseases caused or mediated by respiratory syncytial virus (RSV) are disclosed, wherein said compositions comprise at least one anti-RSV antibody, including high affinity antibodies, and an additional anti-inflammatory agent, especially corticosteroids, as well as anti-inflammatory antibodies, especially anti-interleukin-6. Also disclosed are methods of using such compositions to treat and / or prevent respiratory diseases. Such compositions may optionally contain other non-antibody anti-viral agents.
Owner:AREXIS +1
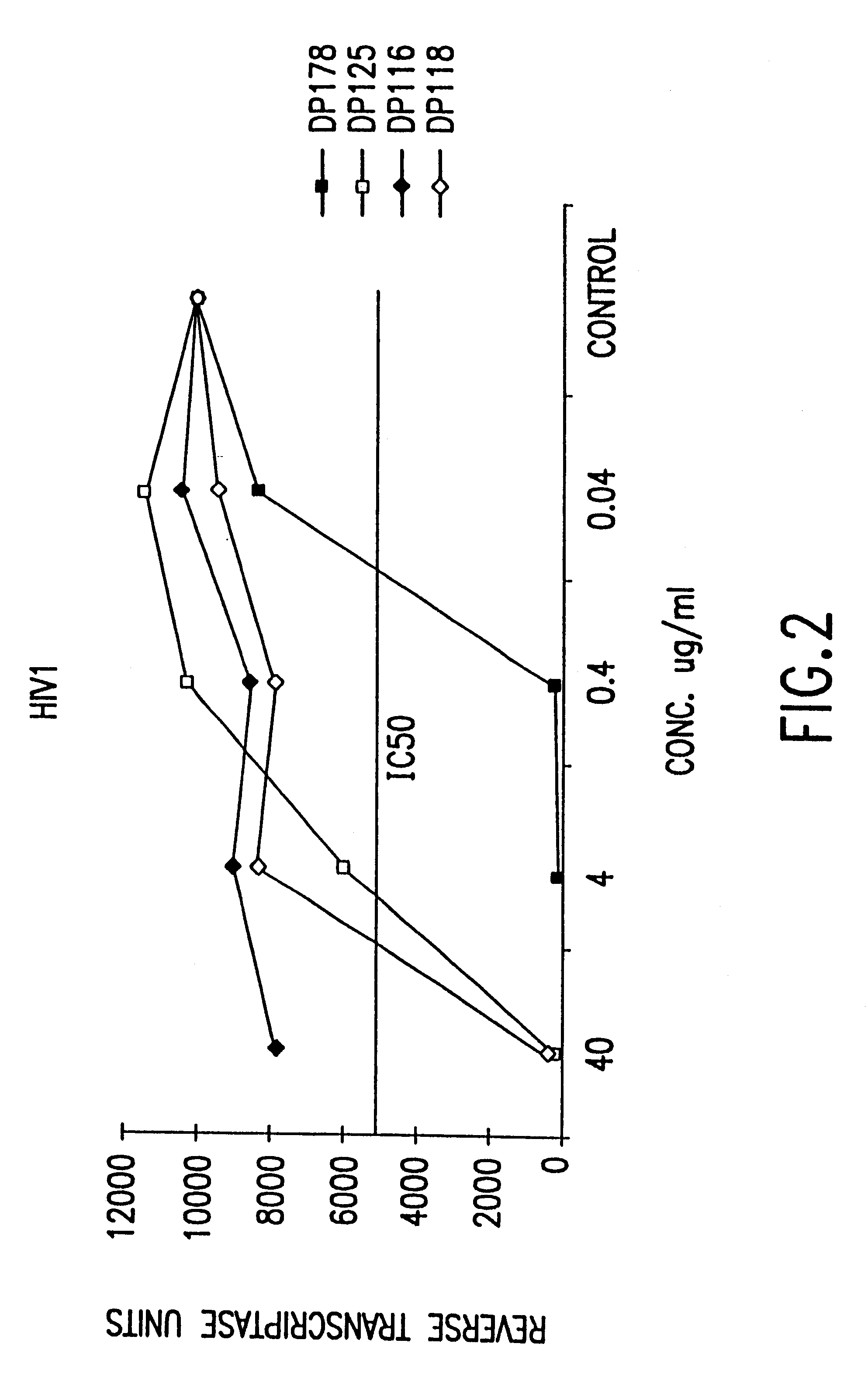
Methods for the inhibition of epstein-barr virus transmission employing anti-viral peptides capable of abrogating viral fusion and transmission
InactiveUS6518013B1SsRNA viruses negative-sensePeptide/protein ingredientsCell membraneViral life cycle
Fusion of the viral envelope, or infected cell membranes with uninfected cell membranes, is an essential step in the viral life cycle. Recent studies involving the human immunodeficiency virus type 1(HIV-1) demonstrated that synthetic peptides (designated DP-107 and DP-178) derived from potential helical regions of the transmembrane (TM) protein, gp41, were potent inhibitors of viral fusion and infection. A computerized antiviral searching technology (C.A.S.T.) that detects related structural motifs (e.g., ALLMOTI 5, 107x178x4, and PLZIP) in other viral proteins was employed to identify similar regions in the Epstein-Barr virus (EBV). Several conserved heptad repeat domains that are predicted to form coiled-coil structures with antiviral activity were identified in the EBV genome. Synthetic peptides of 16 to 39 amino acids derived from these regions were prepared and their antiviral activities assessed in a suitable in vitro screening assay. These peptides proved to be potent inhibitors of EBV fusion. Based upon their structural and functional equivalence to the known HIV-1 inhibitors DP-107 and DP-178, these peptides should provide a novel approach to the development of targeted therapies for the treatment of EBV infections.
Owner:TRIMERIS
Immunotherapy for chronic hepatitis c virus infection
InactiveUS20110256098A1Increase the number ofReduce in quantitySsRNA viruses positive-sensePeptide/protein ingredientsChronic viral hepatitis CInterferon therapy
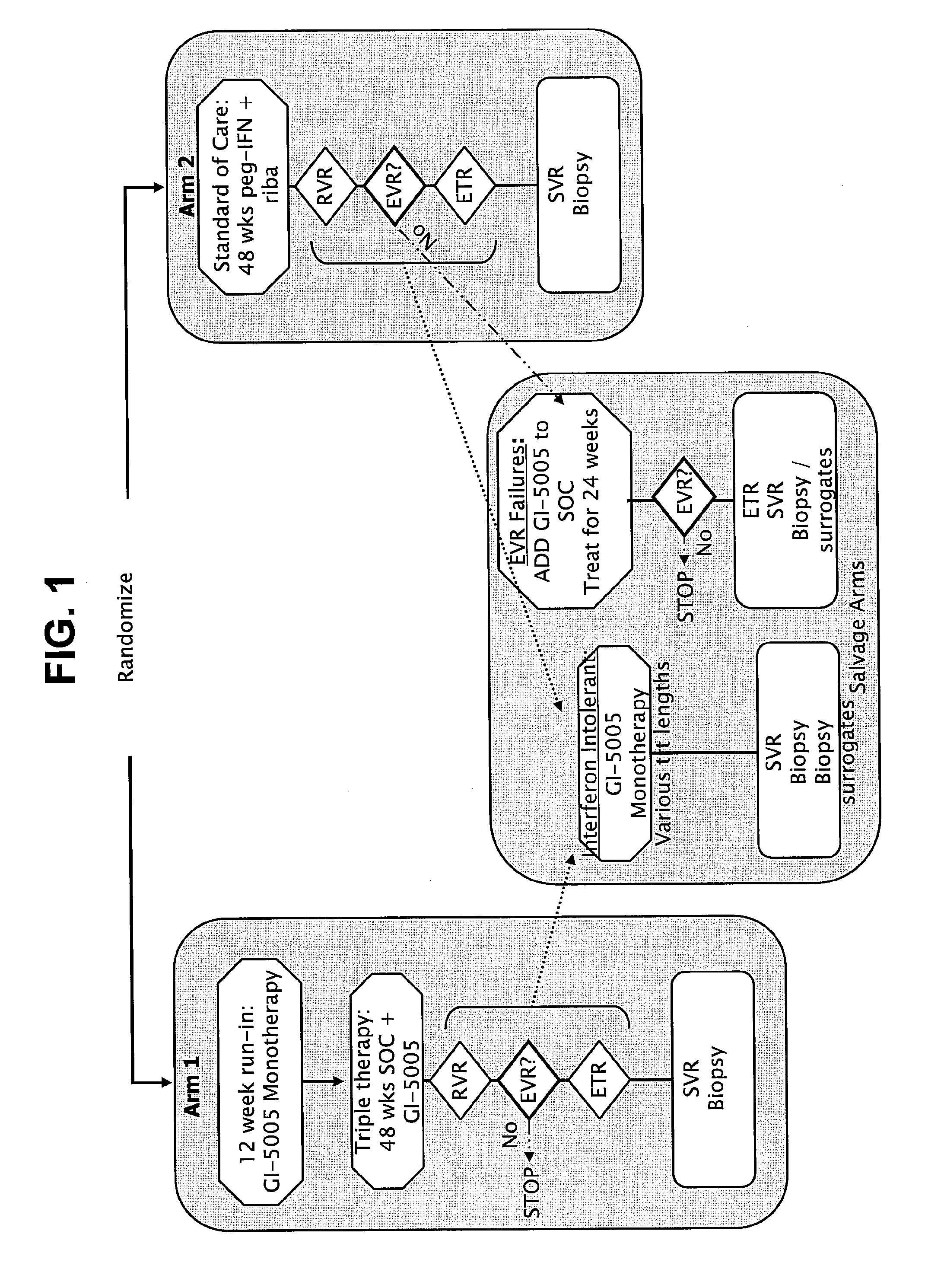
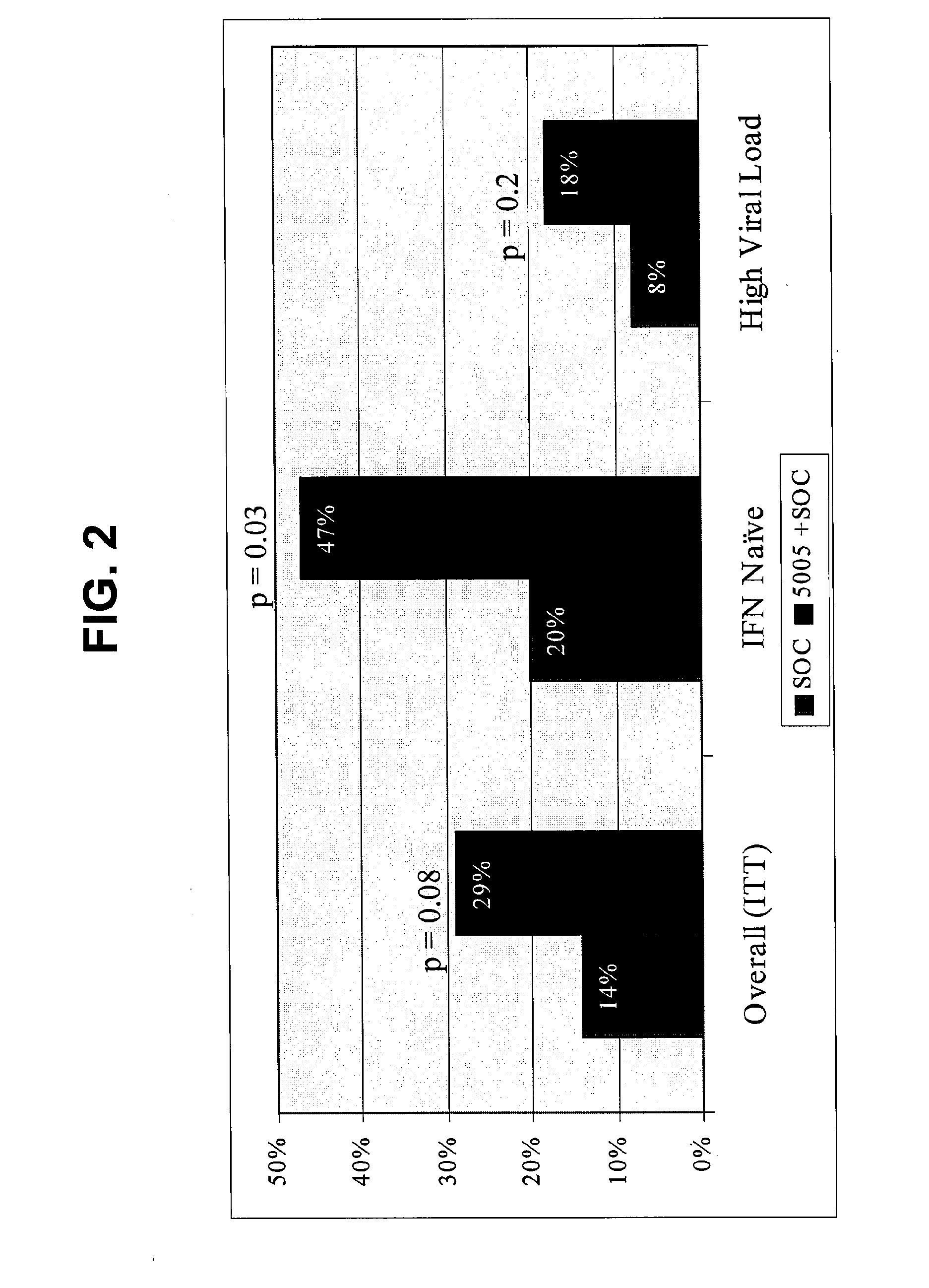
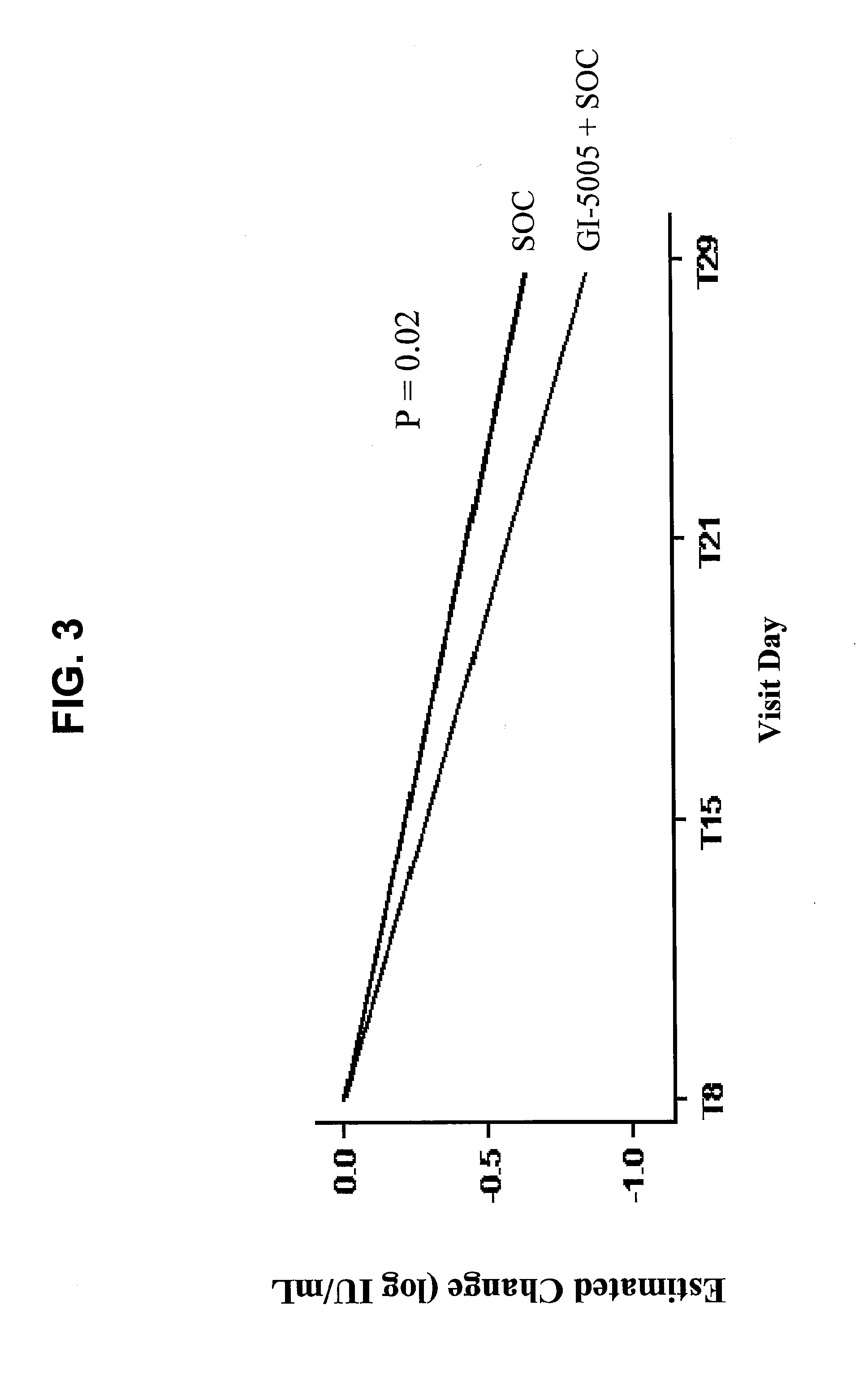
Disclosed are uses of immunotherapeutic compositions in combination with Standard of Care (SOC), or interferon therapy combined with anti-viral therapy, for the improved treatment of chronic hepatitis C virus (HCV) infection and related conditions, including liver function. The compositions, kits and uses of the invention, as compared to the use of SOC therapy alone: improves the rate of early response to therapy as measured by early virologic markers (e.g., RVR and EVR), enlarges the pool of patients who will have sustained responses to therapy over the long term, offers shortened courses of therapy for certain patients, enables “rescue” of patients who are non-responders or intolerant to SOC therapy, improves liver function and / or reduces liver damage in patients, and enables the personalization of HCV therapy for a patient, which can result in dose sparing, improved patient compliance, reduced side effects, and improved long term therapeutic outcomes.
Owner:GLOBE IMMUNE INC



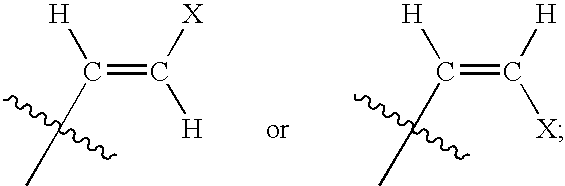


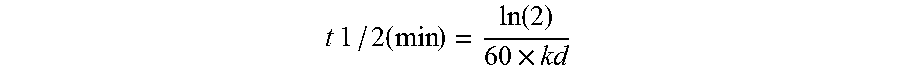




![Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer](https://images-eureka.patsnap.com/patent_img/3d019e20-9f9e-41d5-99b4-d31b4ff3c58e/US08242085-20120814-C00001.png)
![Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer](https://images-eureka.patsnap.com/patent_img/3d019e20-9f9e-41d5-99b4-d31b4ff3c58e/US08242085-20120814-C00002.png)
![Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer Tetrahydrofuro [3,4-D] dioxolane compounds for use in the treatment of viral infections and cancer](https://images-eureka.patsnap.com/patent_img/3d019e20-9f9e-41d5-99b4-d31b4ff3c58e/US08242085-20120814-C00003.png)