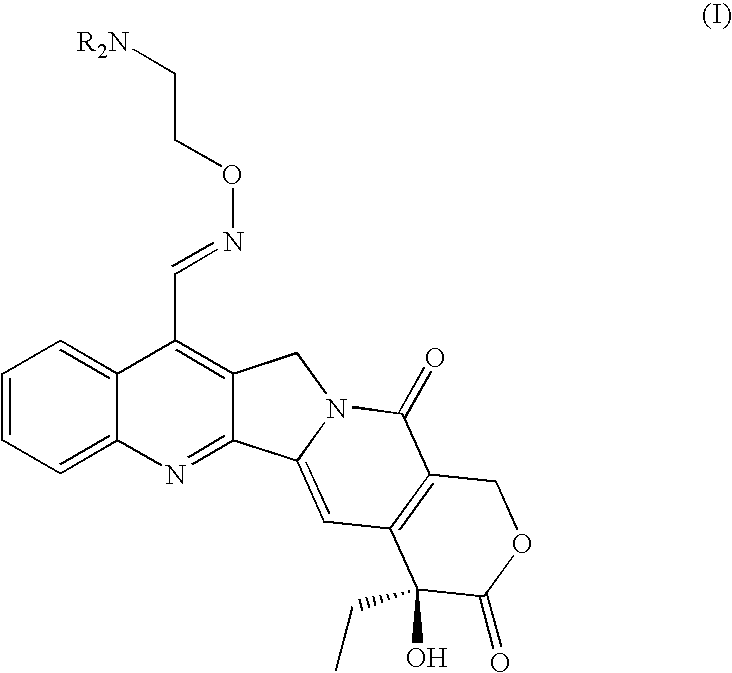
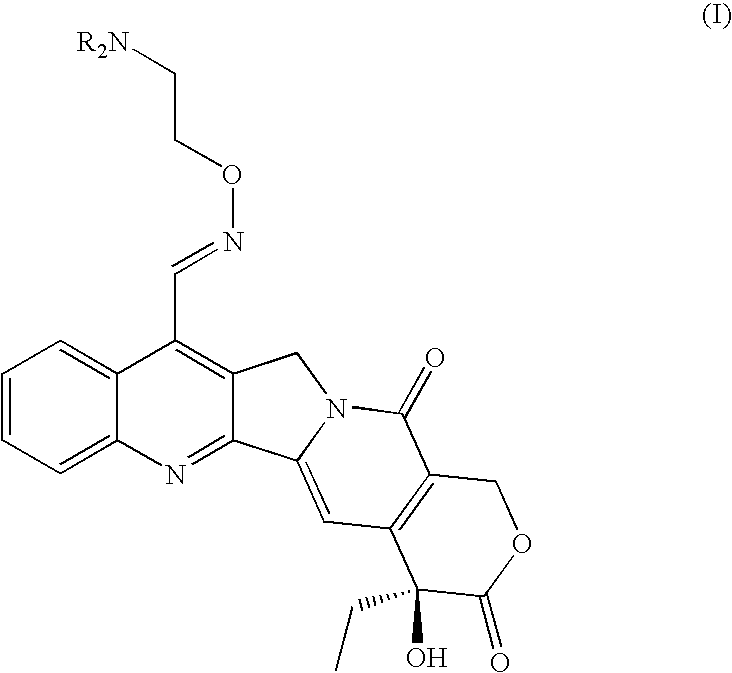
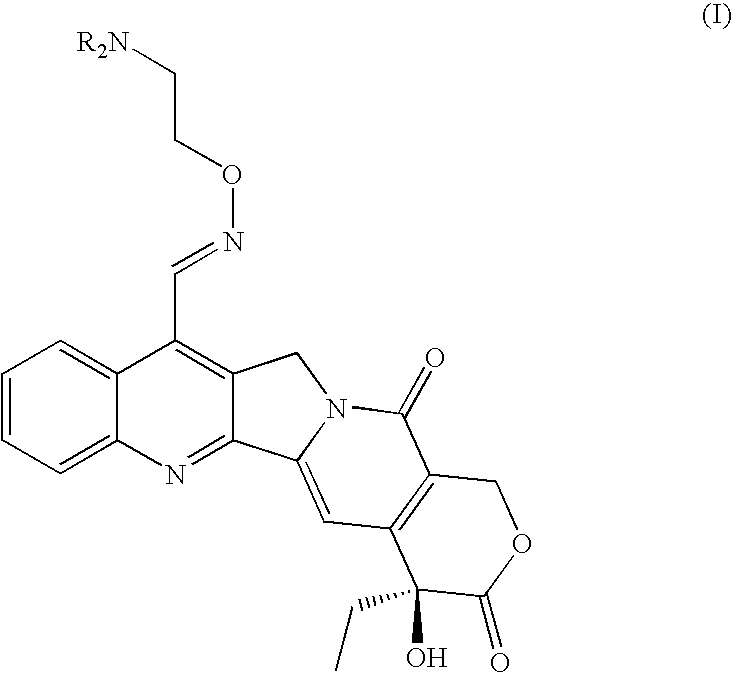
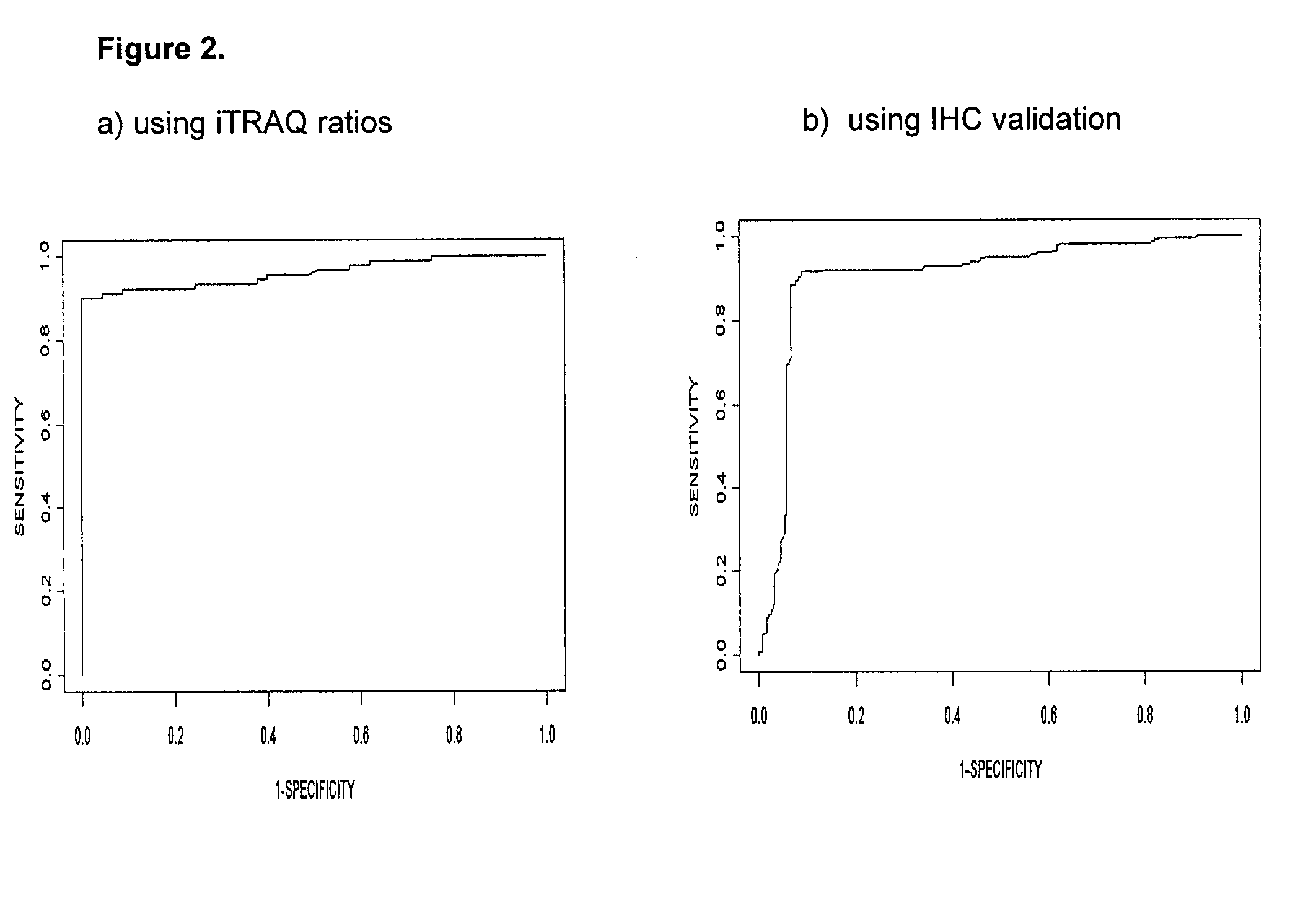Patents
Literature
43 results about "Head and neck carcinoma" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Head and neck cancer originates in the tissues and organs of the head and neck area. Most head and neck cancers begin in the squamous cells that line the mouth, nose and throat. These types of cancer are generally referred to as squamous cell carcinomas and may include cancer that develops in the larynx, throat, lips, mouth, nose or salivary glands.
Biomarkers for the Identification Monitoring and Treatment of Head and Neck Cancer
InactiveUS20100099093A1Microbiological testing/measurementDisease diagnosisCancer cellHead and neck cancer
Owner:THE DNA REPAIR
Biomarkers for Head-And-Neck Cancers and Precancers
The invention provides markers and methods for detecting head-and-neck precancers, (including OPLs), cancers and related disease conditions in a subject. The invention also provides localization and imaging methods for head-and-neck precancers (including OPLs) and cancers, along with kits for carrying out methods of the invention. The invention further provides therapeutic applications for head-and-neck precancers (including OPLs) and cancers which employ head-and-neck precancer and cancer markers, polynucleotides encoding the markers, and binding agents for the markers.
Owner:WALFISH PAUL +1
Monoclonal antibodies against claudin-18 for treatment of cancer
The present invention provides antibodies useful as therapeutics for treating and / or preventing diseases associated with cells expressing CLD18, including tumor-related diseases such as gastric cancer, esophageal cancer, pancreatic cancer, lung cancer, ovarian cancer, colon cancer, hepatic cancer, head-neck cancer, and cancer of the gallbladder.
Owner:ASTELLAS PHARMA INC +1
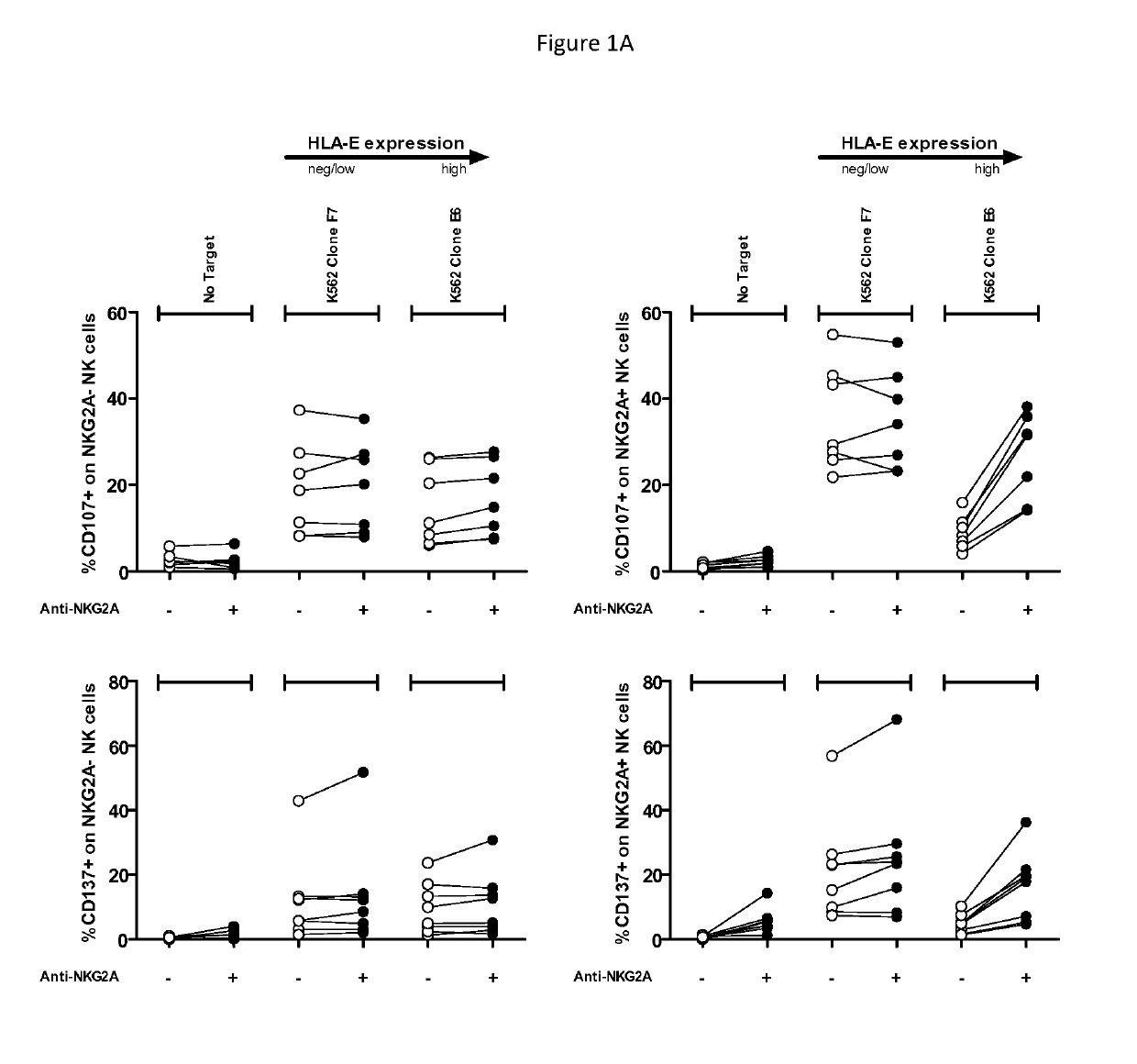
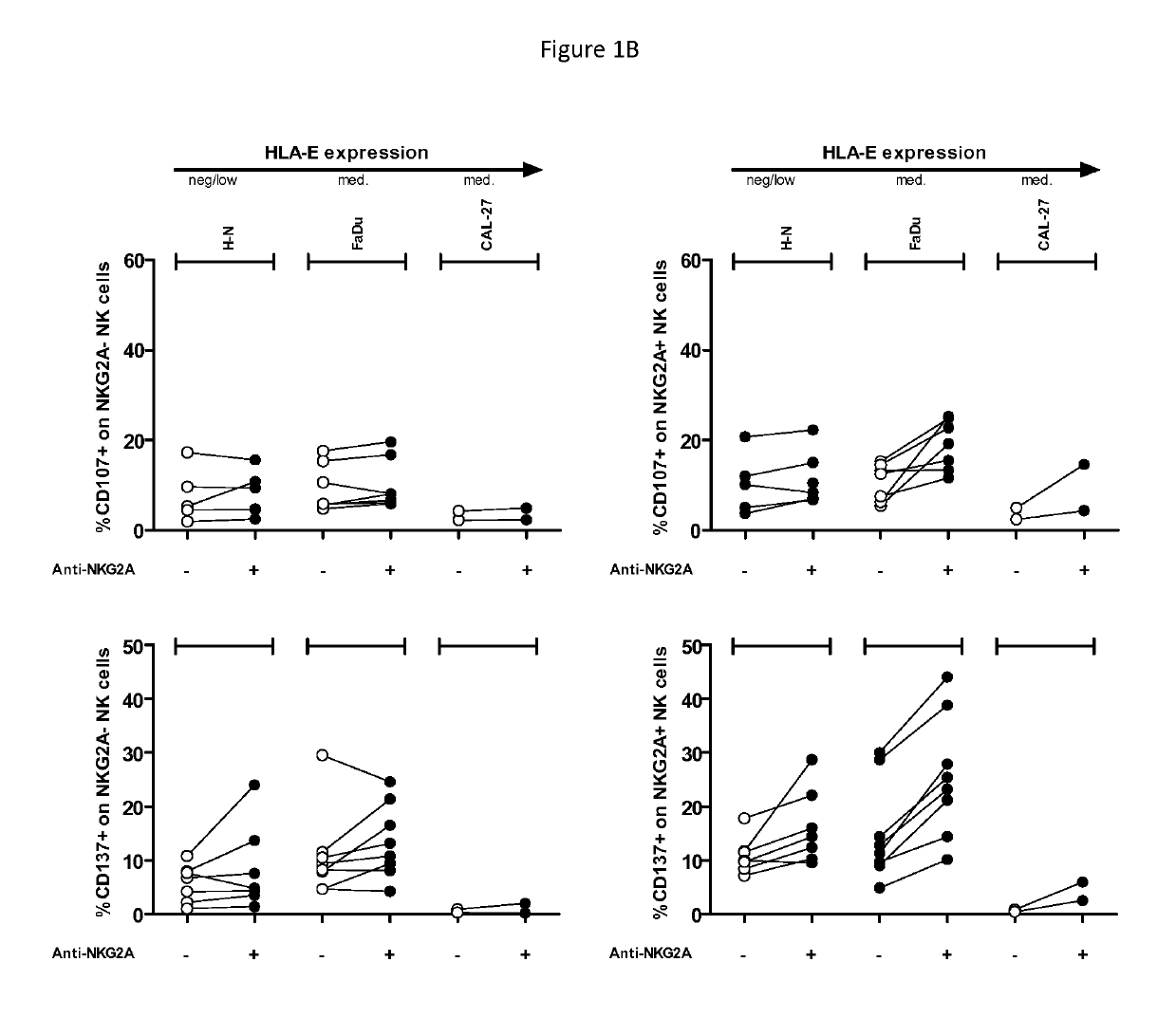
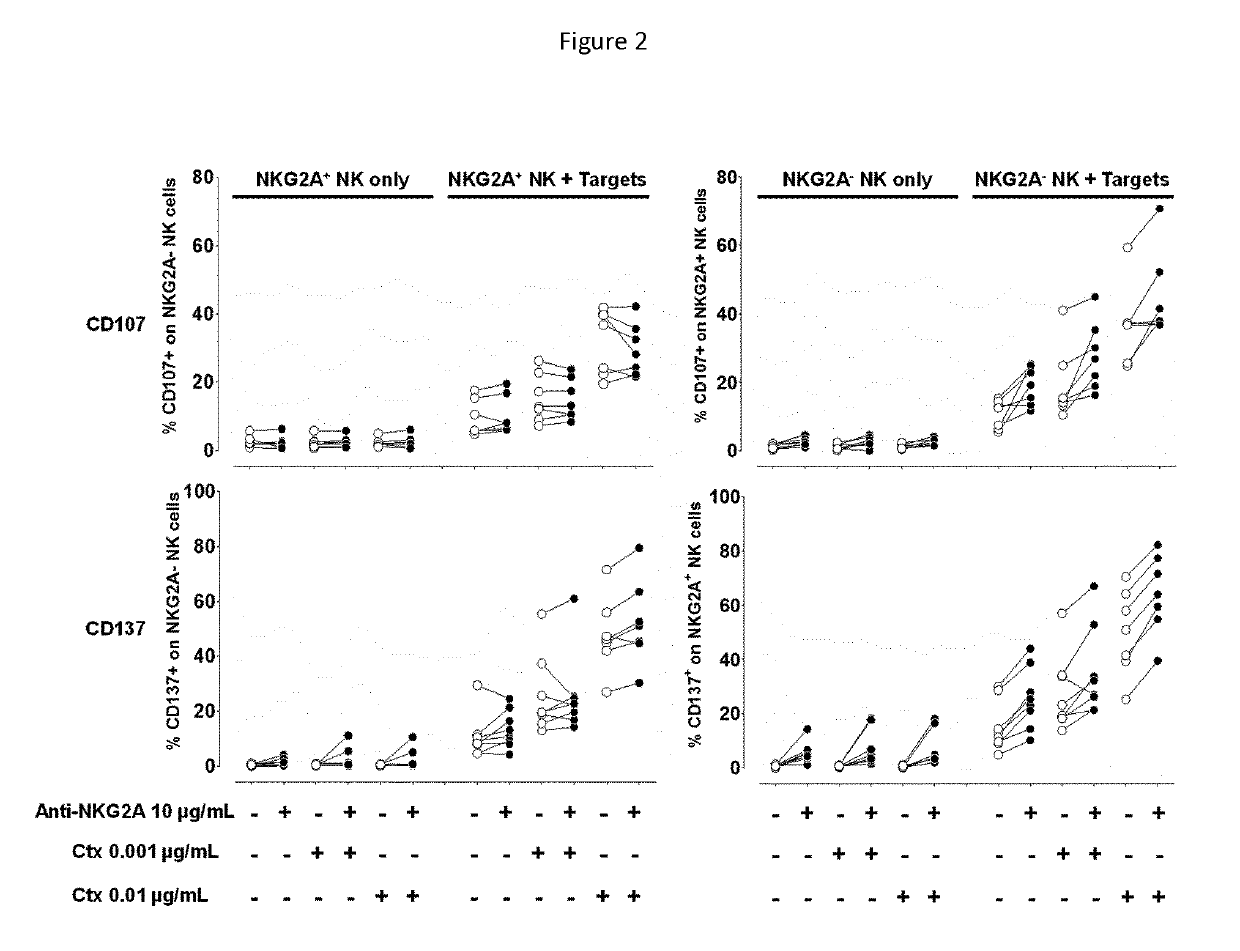
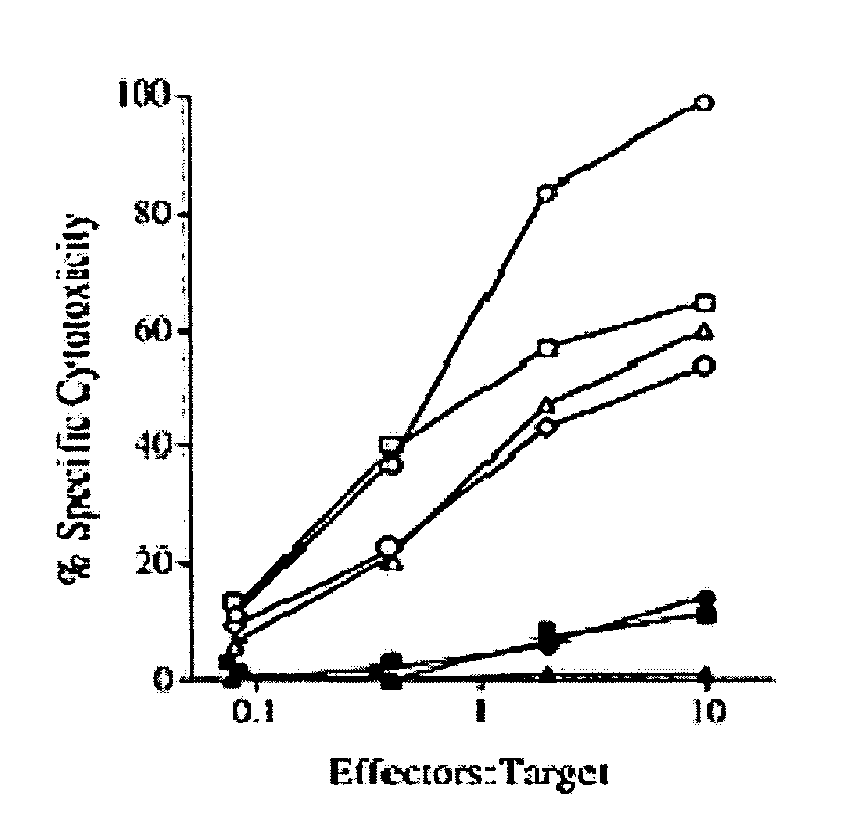
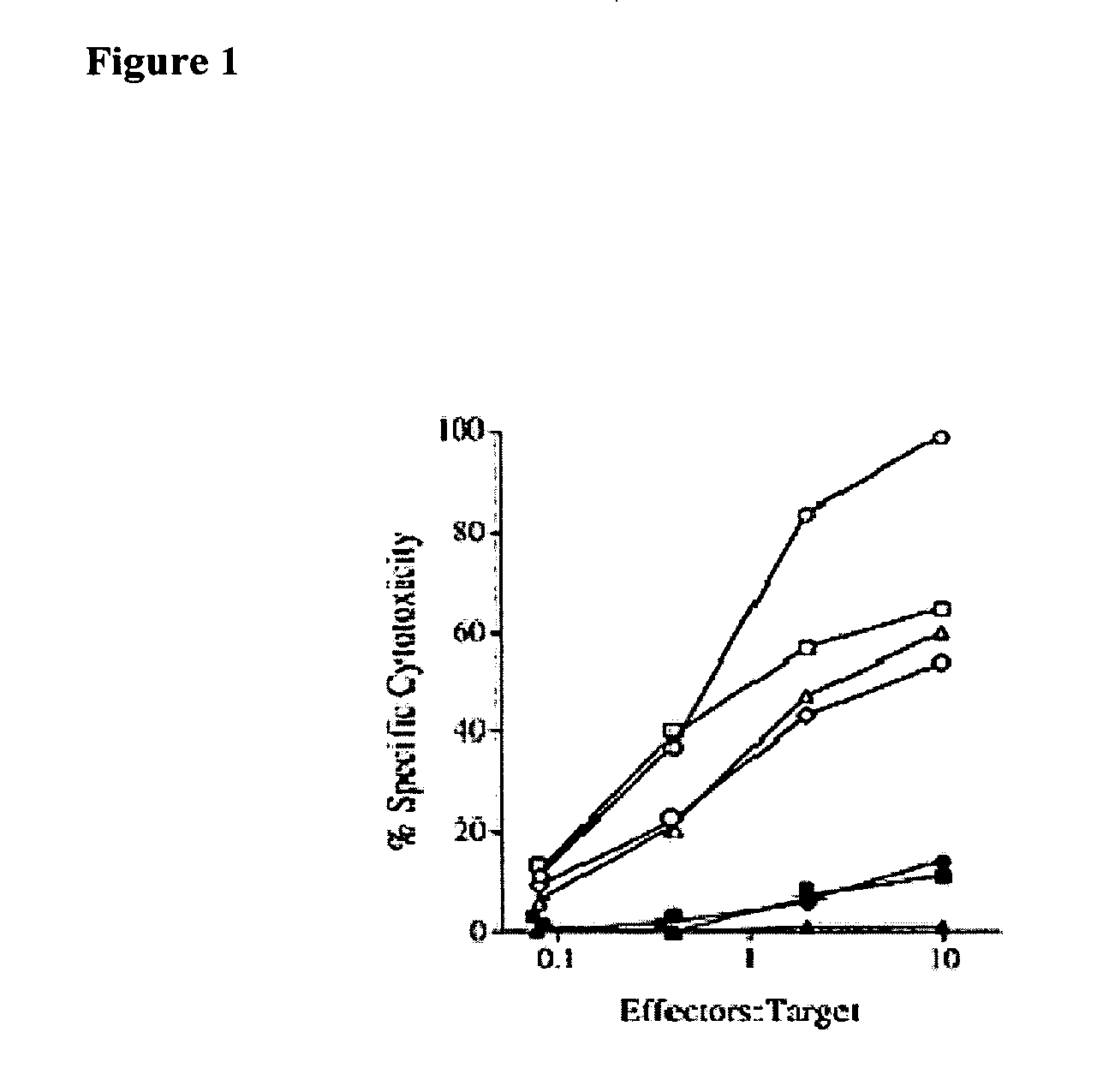
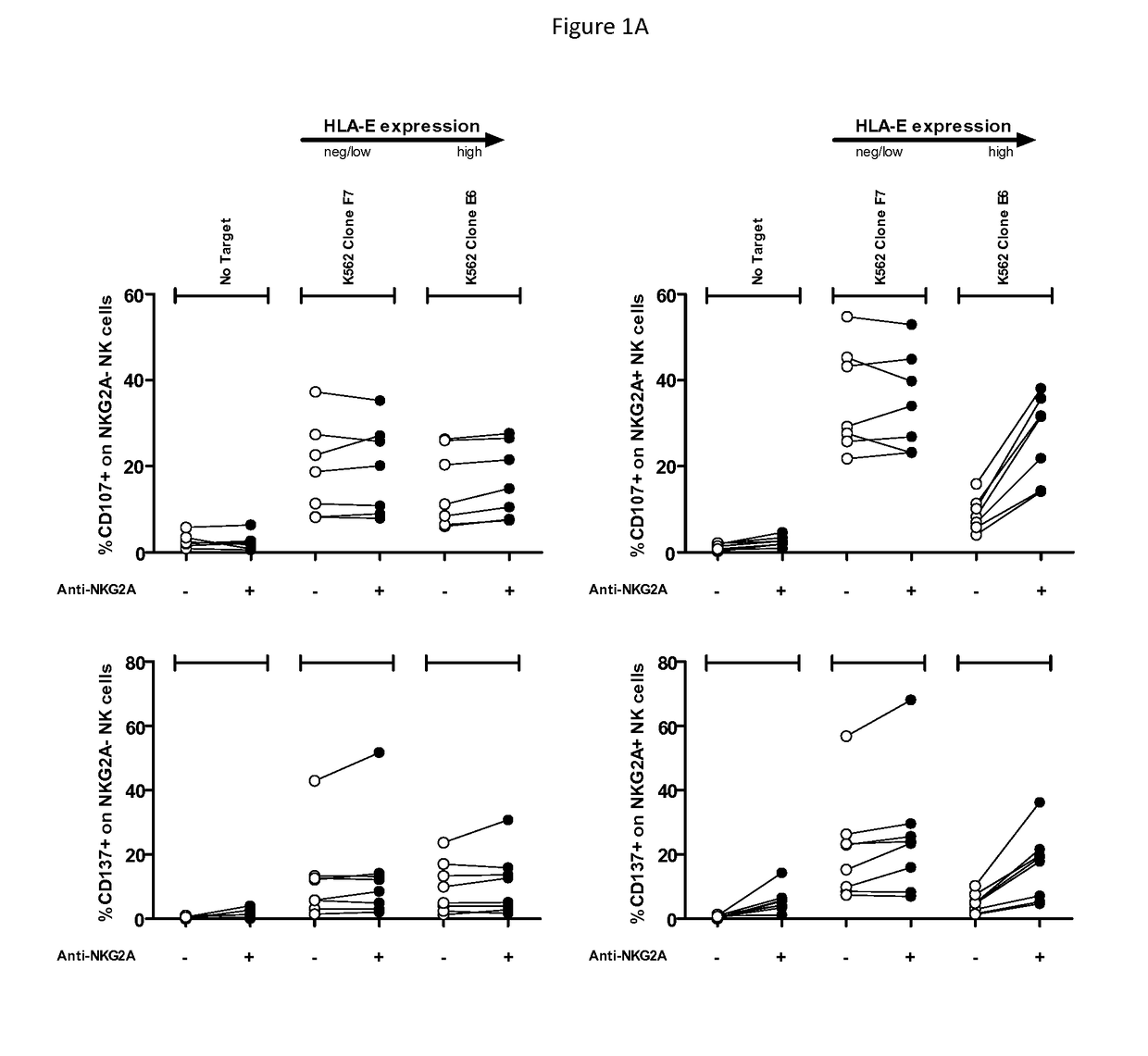
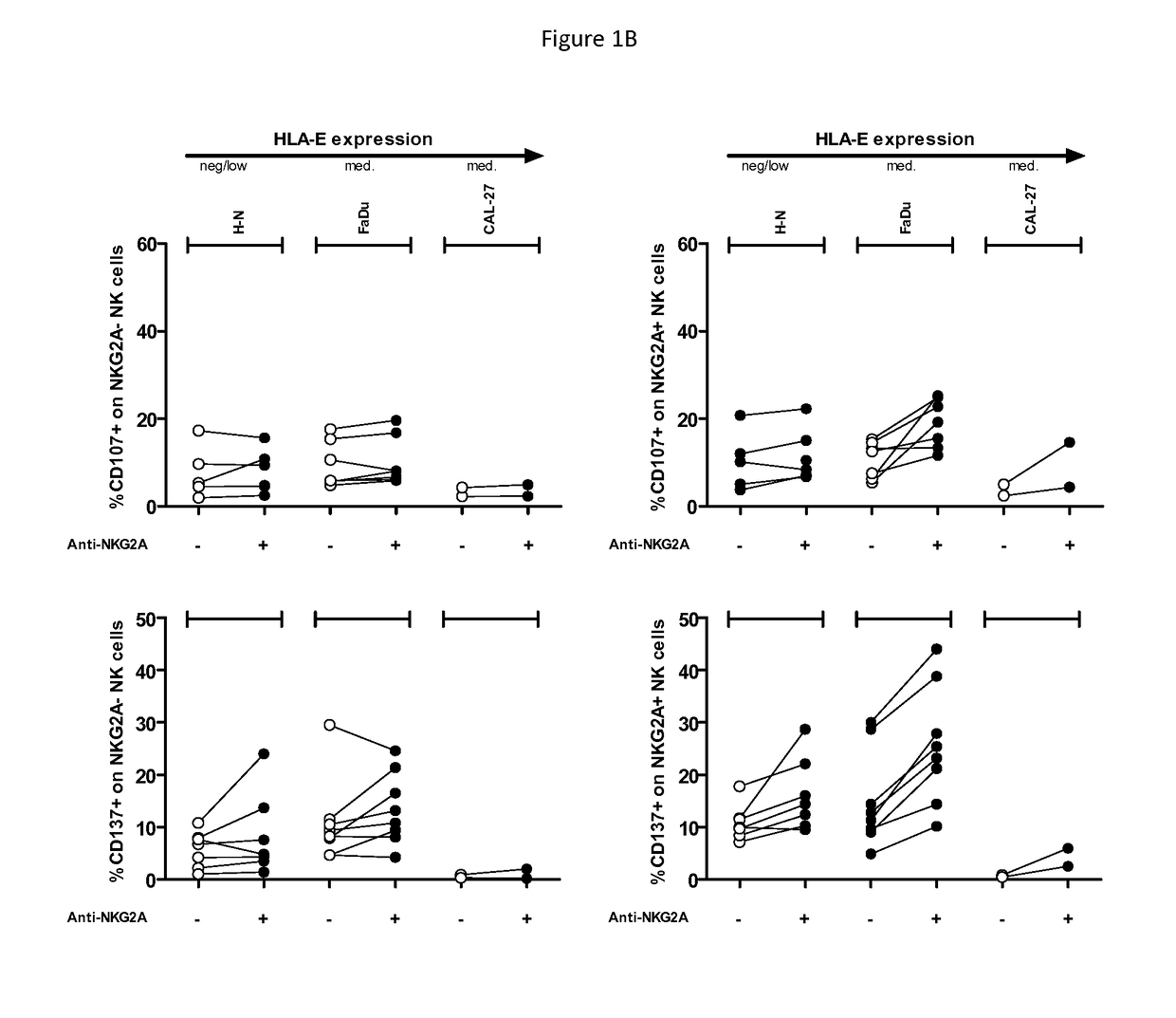
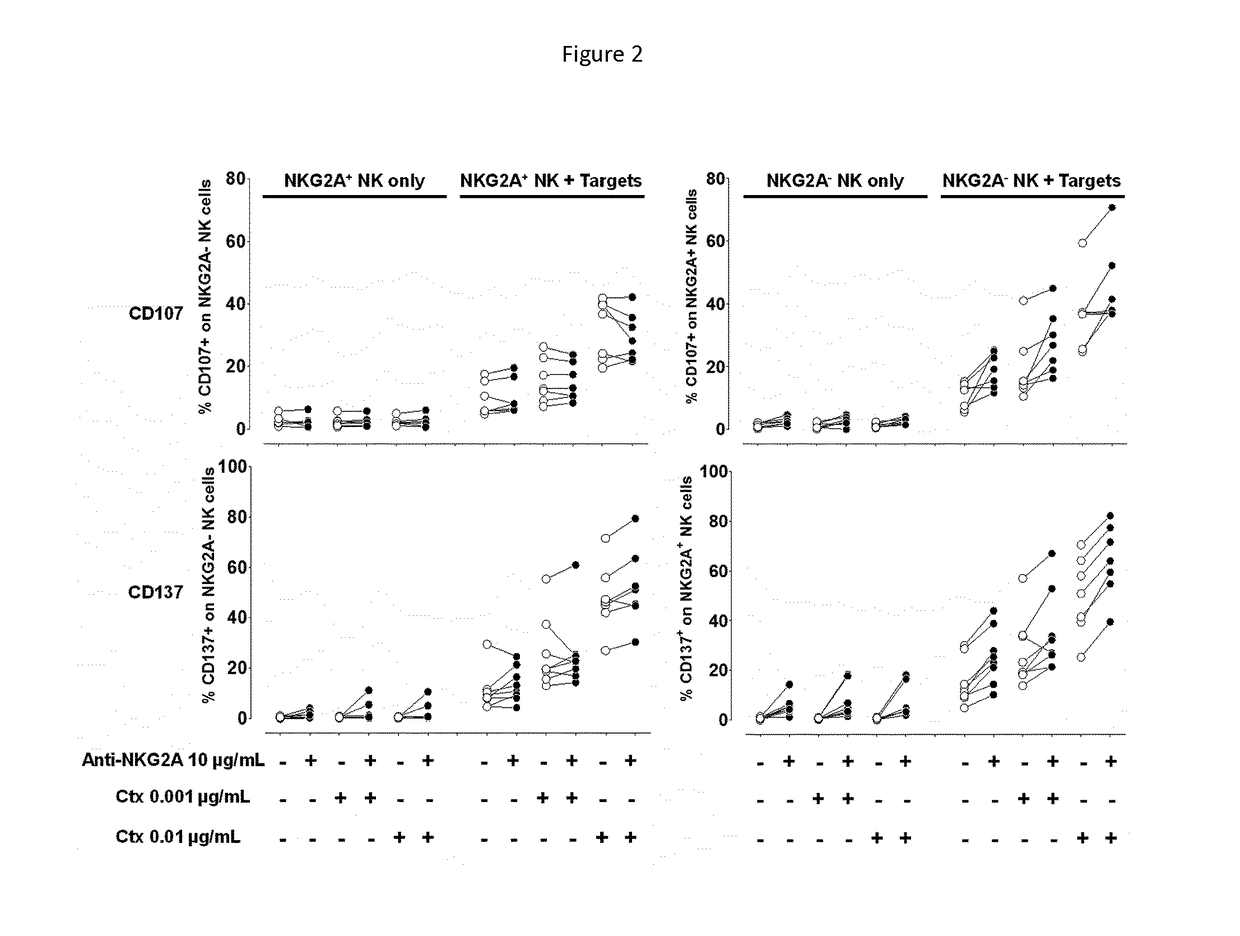
Treatment of cancers using anti-NKG2A agents
ActiveUS10329348B2Effectively eliminate head and neck cancer cellsHigh expressionImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsSquamous CarcinomasTumor Escape
The present invention relates to HLA-E as a tumor escape mechanism in head and neck cancer. The invention relates to methods for the treatment of head and neck cancer, notably HLA-E expressing head and neck squamous cell carcinoma, using antibodies that specifically bind and inhibit human NKG2A.
Owner:INNATE PHARMA SA
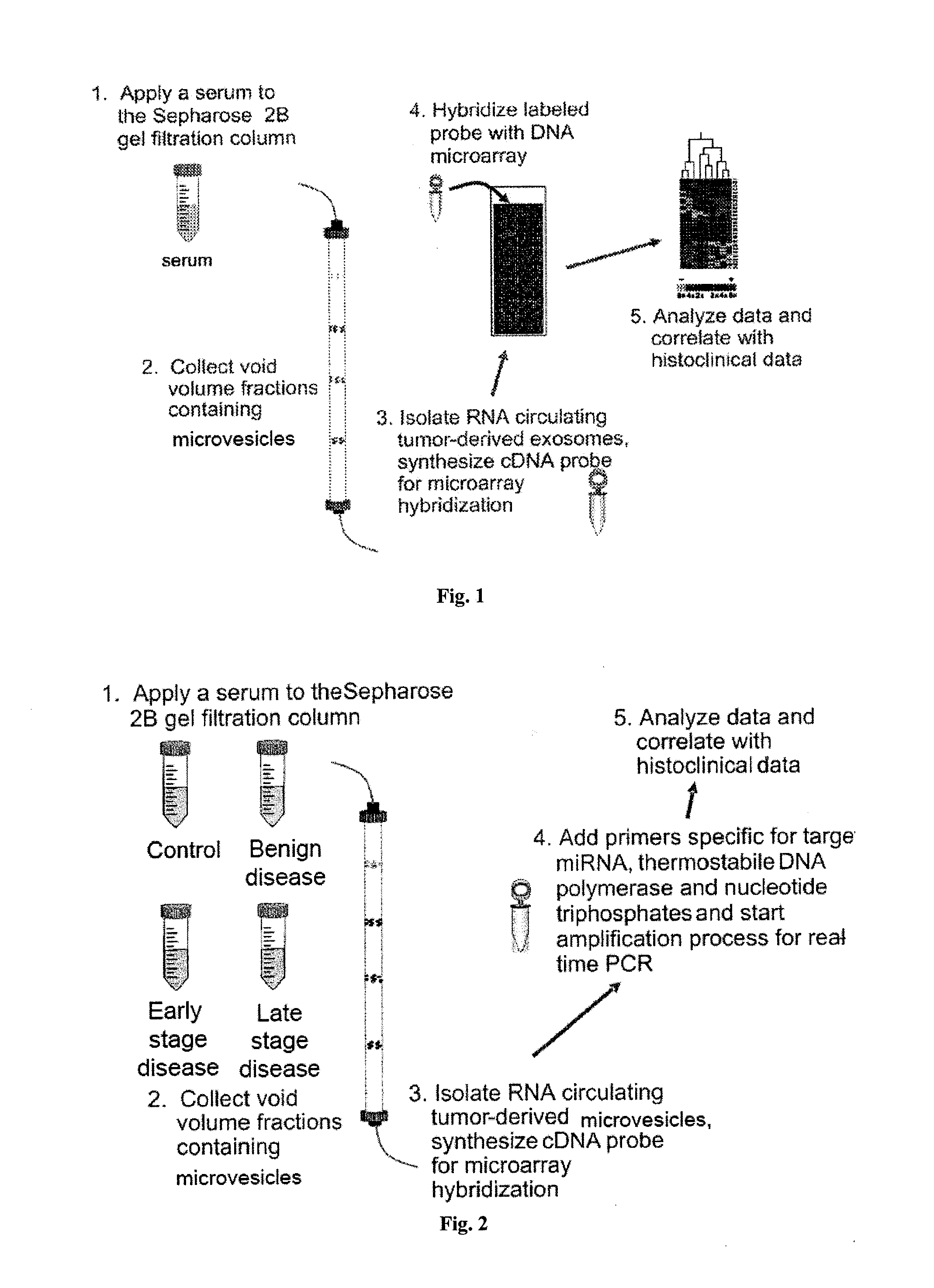
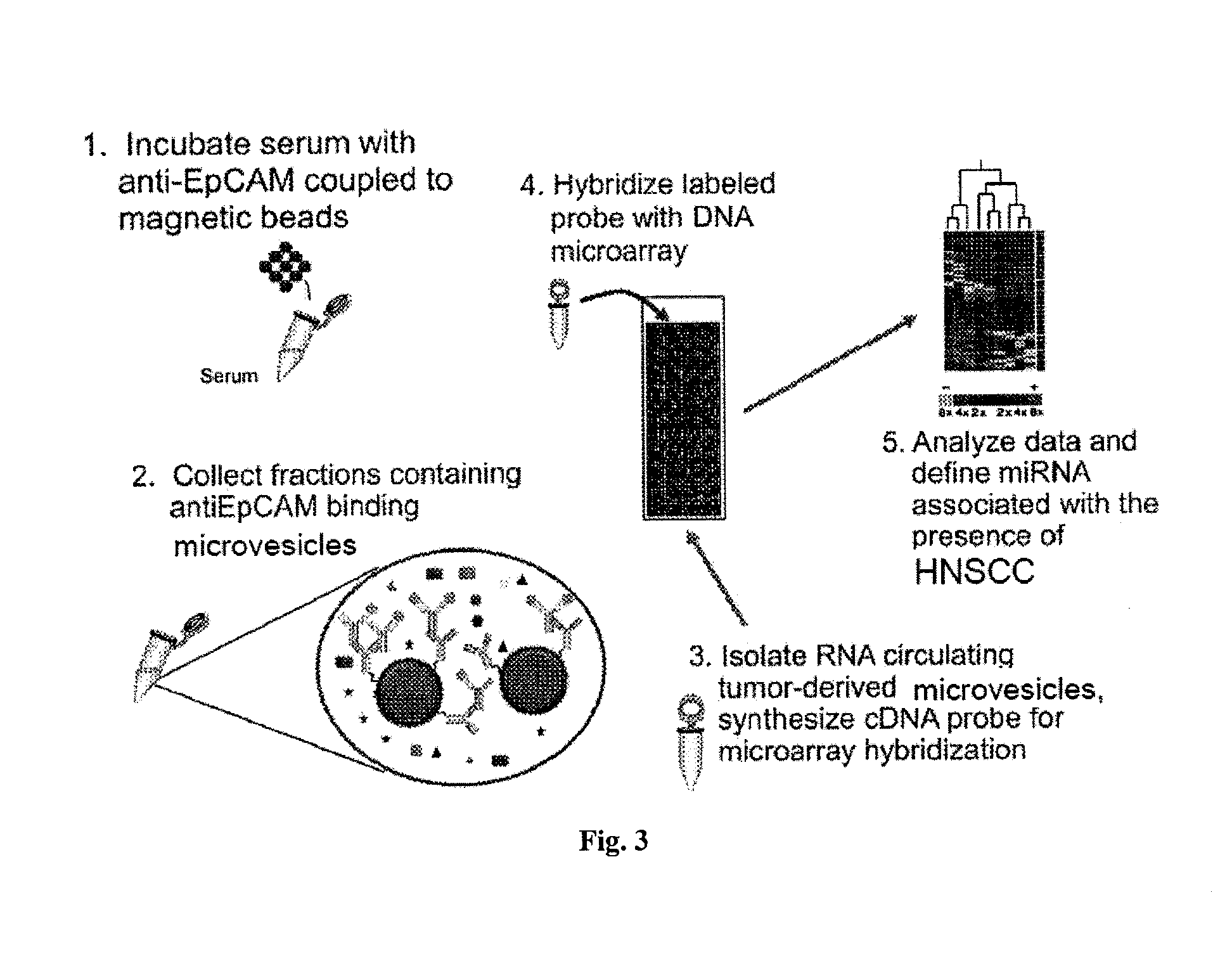
Microrna biomarkers
InactiveUS20150018227A1Component separationMicrobiological testing/measurementSubject matterMicroRNA
The presently disclosed subject matter provides methods for characterization of and evaluation of treatment and / or progression of a lung cancer or a head and neck cancer by measuring amounts of one or more RNAs present in microvesicles isolated from a biological sample from the subject.
Owner:UNIV OF LOUISVILLE RES FOUND INC
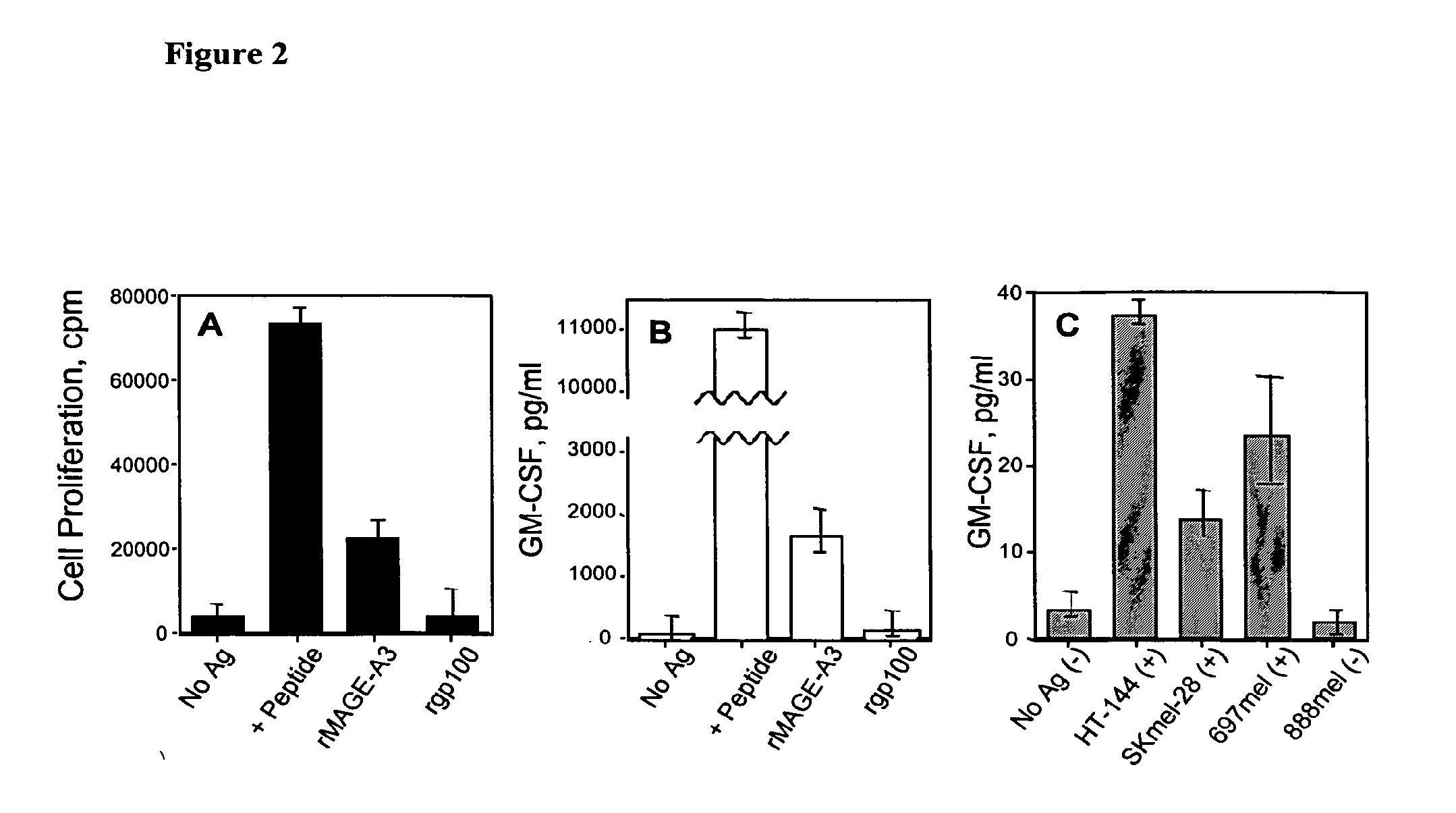
Mage-a3/hpv 16 peptide vaccines for head and neck cancer
ActiveUS20080044407A1Tumor rejection antigen precursorsPeptide/protein ingredientsSquamous CarcinomasPeptide vaccine
Owner:MAYO FOUND FOR MEDICAL EDUCATION & RES +1
Treatment of cancers using Anti-nkg2a agents
ActiveUS20170313773A1Eliminate effectiveEffectively eliminate head and neck cancer cellsImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsParanasal Sinus CarcinomaAntiendomysial antibodies
The present disclosure relates to HLA-E as a tumor escape mechanism in head and neck cancer. The disclosure relates to methods for the treatment of head and neck cancer, notably HLA-E expressing head and neck squamous cell carcinoma, using antibodies that specifically bind and inhibit human NKG2A.
Owner:INNATE PHARMA SA
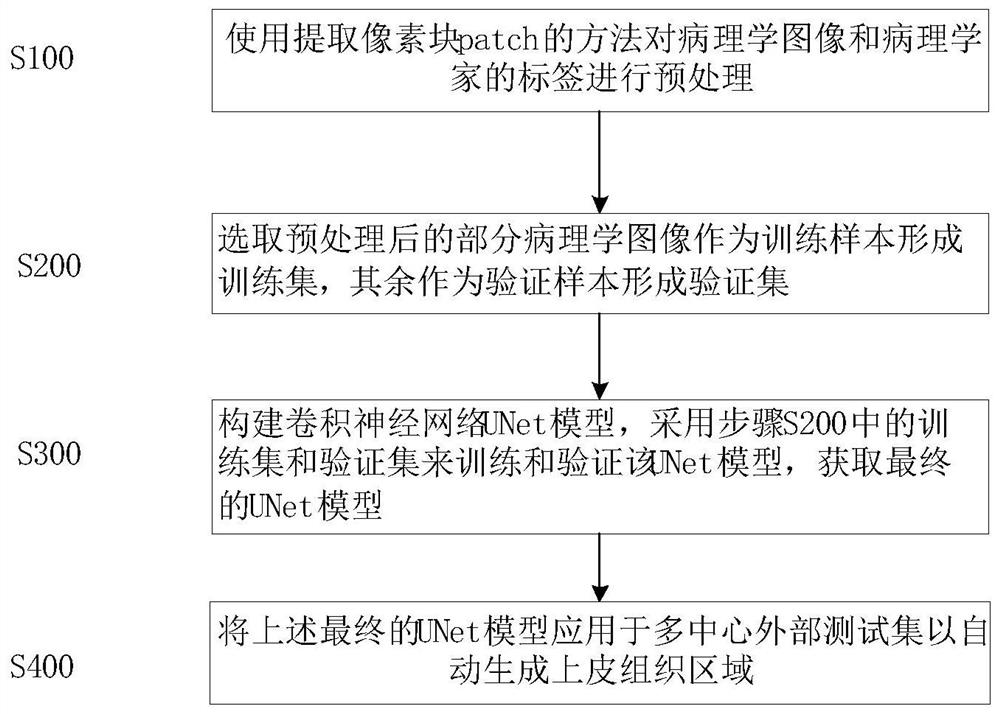
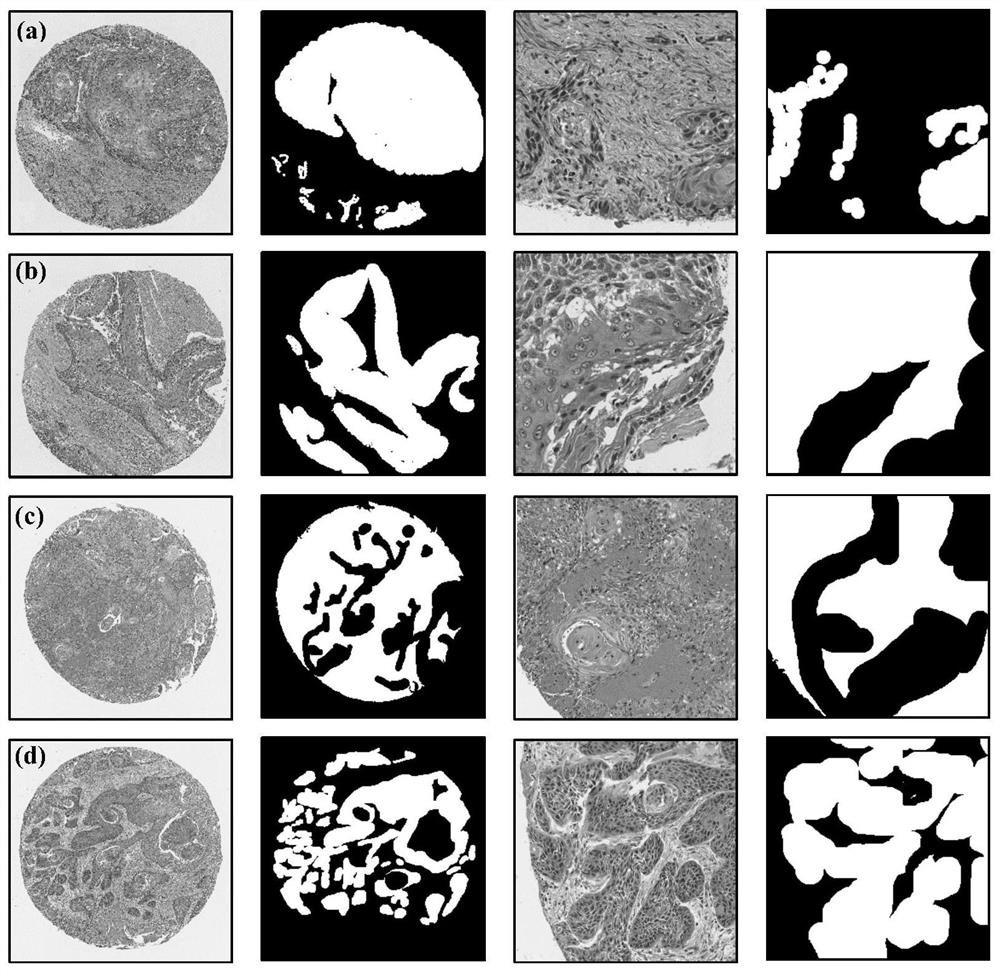
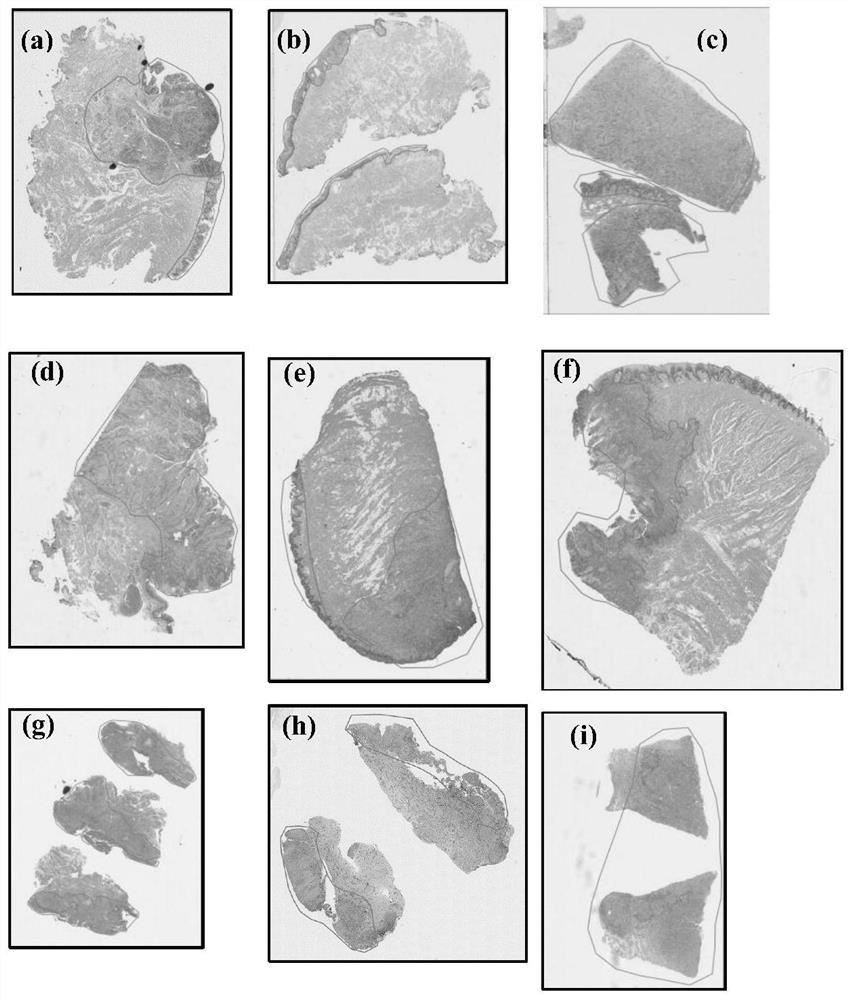
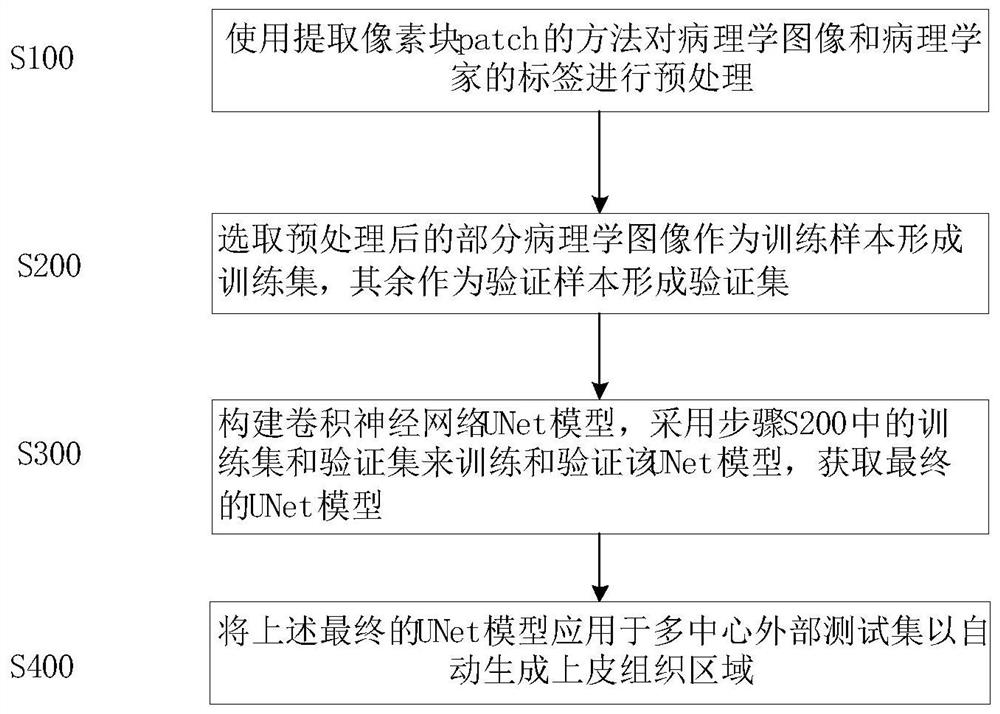
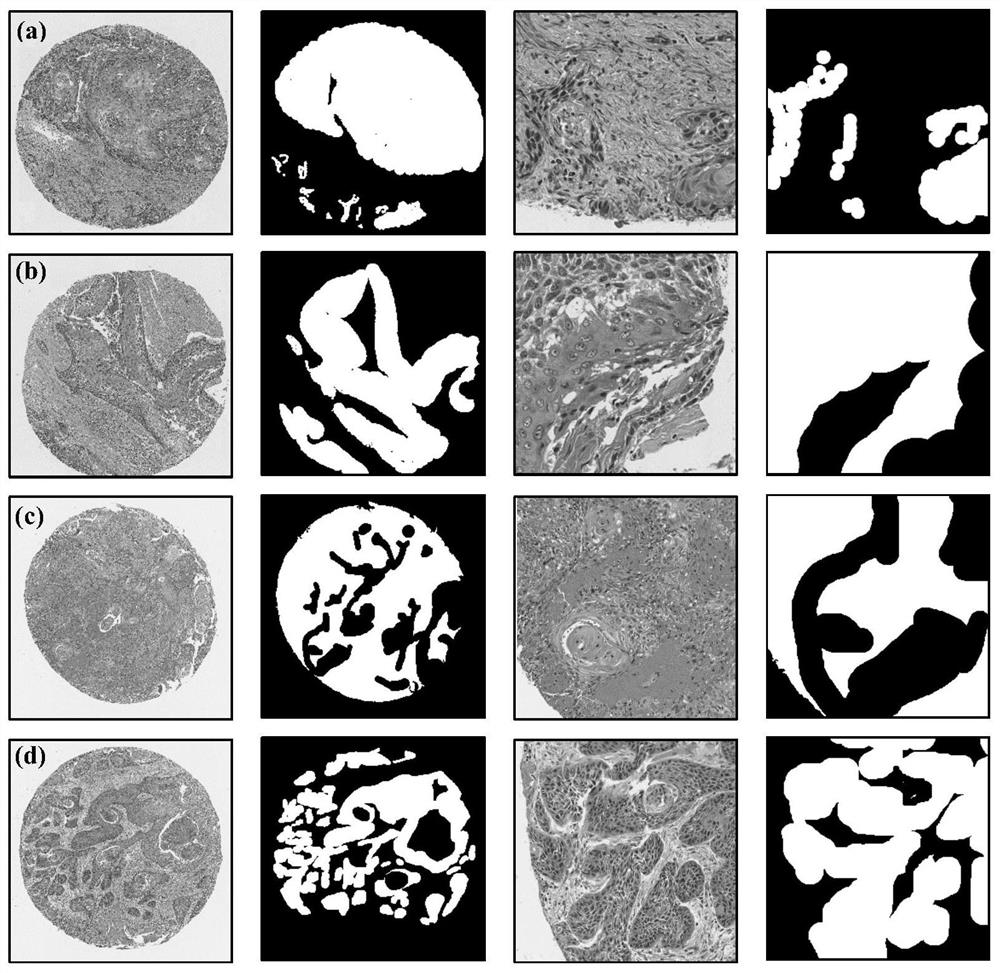
Automatic segmentation method for oral cancer epithelial tissue region of pathology image
The invention discloses an automatic segmentation method for an oral cancer epithelial tissue region of a pathological image. The method comprises the following steps: S100, preprocessing the pathological image and a label of a pathologist by using a method for extracting a pixel block patch; s200, selecting part of the preprocessed pathological images as training samples to form a training set, and taking the rest pathological images as verification samples to form a verification set; s300, constructing a convolutional neural network UNet model, and training and verifying the UNet model by adopting the training set and the verification set in the step S200 to obtain a final UNet model; and S400, applying the final UNet model to a multi-center external test set to automatically generate an epithelial tissue region. According to the method, training, verification and testing are not carried out on each data set in head and neck cancer detection, only TMA data is used for training, testing is carried out on a multi-center external WSI data set, and the method is more persuasive to unknown images.
Owner:SHAANXI NORMAL UNIV +1
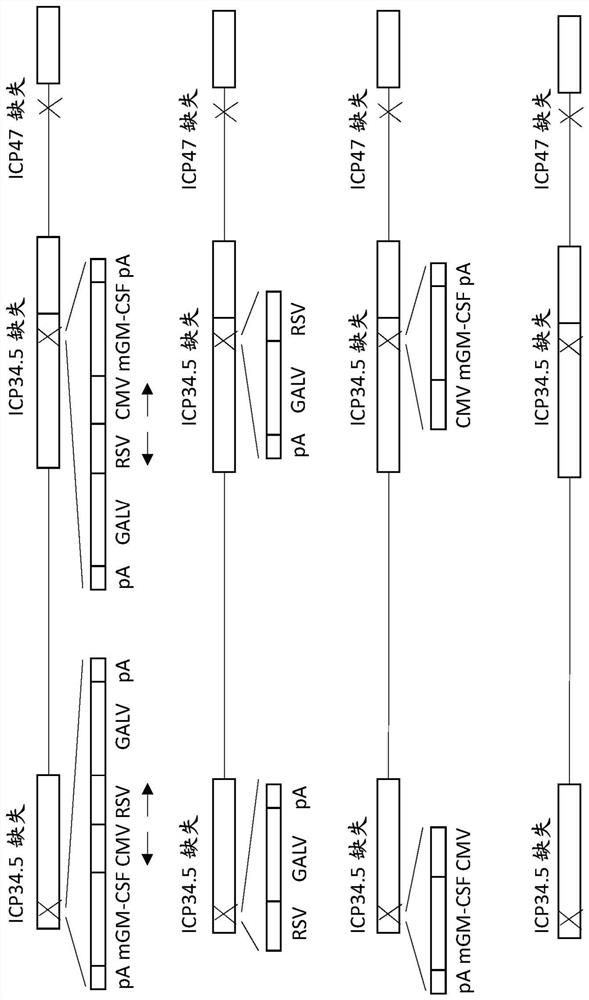
Treatment using oncolytic virus
An oncolytic virus is for use in a method of treating or preventing cutaneous squamous cell carcinoma (CSCC), renal cell carcinoma (RCC), non-small cell lung cancer (NSCLC), triple negative breast cancer (TNBC), small cell lung cancer (SCLC), advanced recurrent head and neck cancer, squamous cell carcinoma of the head and neck (SCCHN), nasopharyngeal carcinoma (NPC), hepatocellular carcinoma (HCC), anal cancer, colorectal cancer (CRC), basal cell carcinoma (BCC), Merkel cell carcinoma, appendiceal carcinoma, sarcoma of the skin, recurrent melanoma after surgery, advanced or metastatic urothelial carcinoma, liver metastases, microsatellite instability high cancer (MSI-H), mixed advanced solid tumors, virally caused cancer, locoregionally advanced cancer, pediatric cancer, cancer in patientswith no or minimal pre-existing anti-cancer immunity, cancer as first line therapy, cancer in previously treated patients, cancer in patients who have not received checkpoint blockade therapy, and / orcancer in patients who have received checkpoint blockade therapy, wherein the oncolytic virus is, or is derived from, a clinical isolate which has been selected by comparing the abilities of a panelof three or more clinical isolates of the same viral species to kill tumor cells of two or more tumor cell lines in vitro and selecting a clinical isolate which is capable of killing cells of two or more tumor cell lines more rapidly and / or at a lower dose in vitro than one or more of the other clinical isolates in the panel; comprises (i) a fusogenic protein-encoding gene; and (ii) an immune stimulatory molecule or an immune stimulatory molecule-encoding gene; comprises (i) a GM-CSF-encoding gene; and (ii) an immune co-stimulatory pathway activating molecule or an immune co-stimulatory pathway activating molecule-encoding gene; and / or comprises a gene encoding a CTLA-4 inhibitor.
Owner:REPLIMUNE
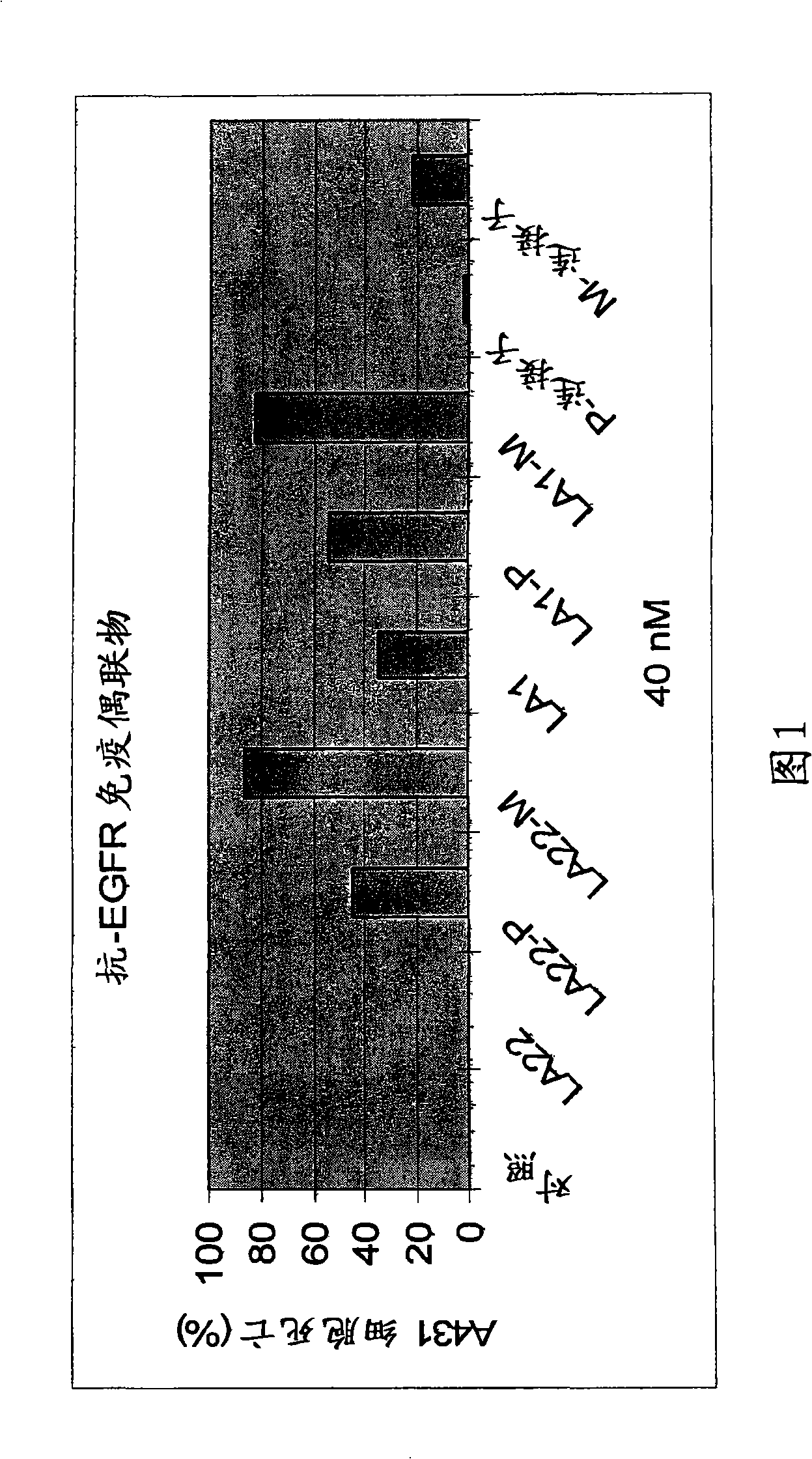
Antibodies for the treatment of cancers
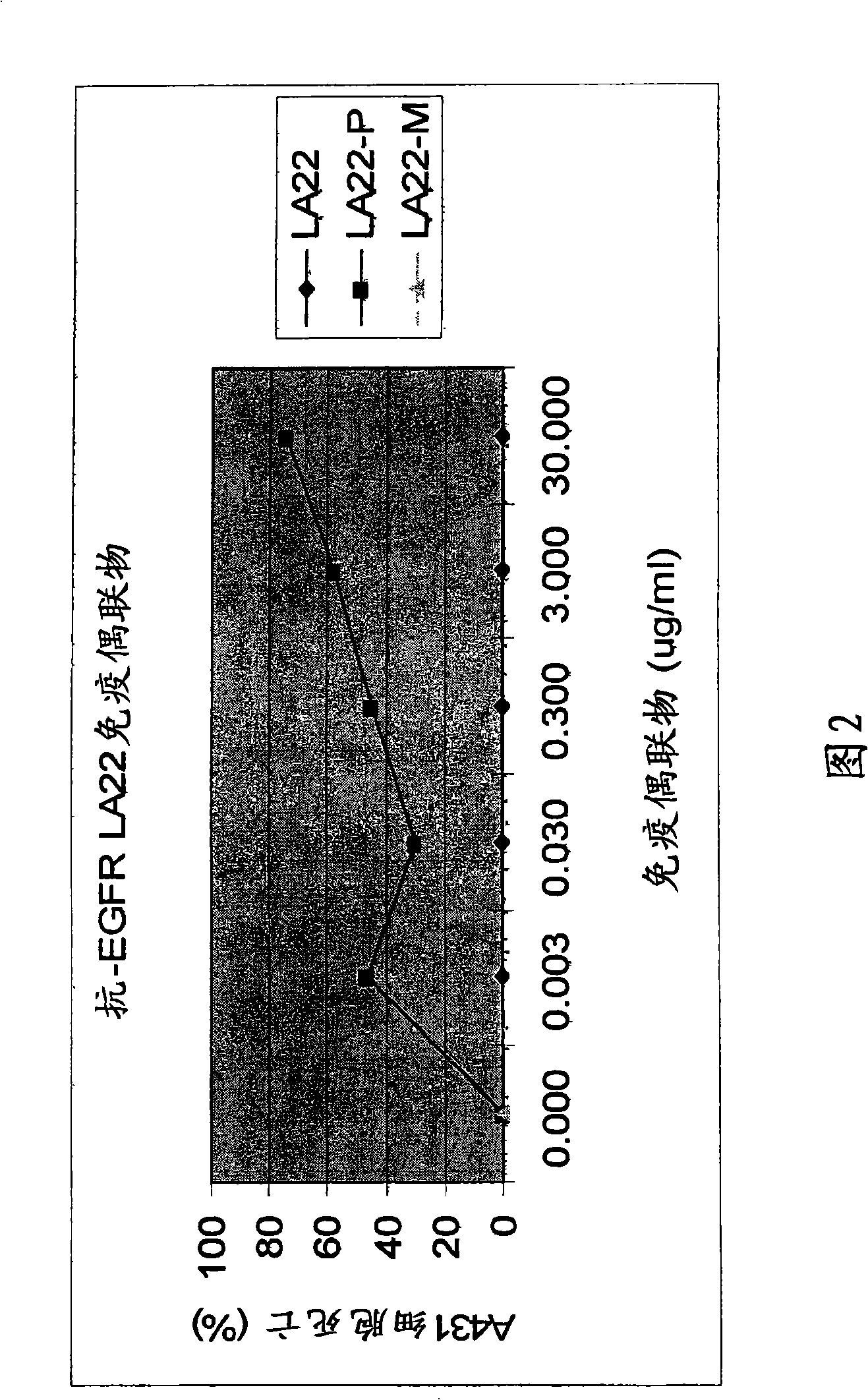
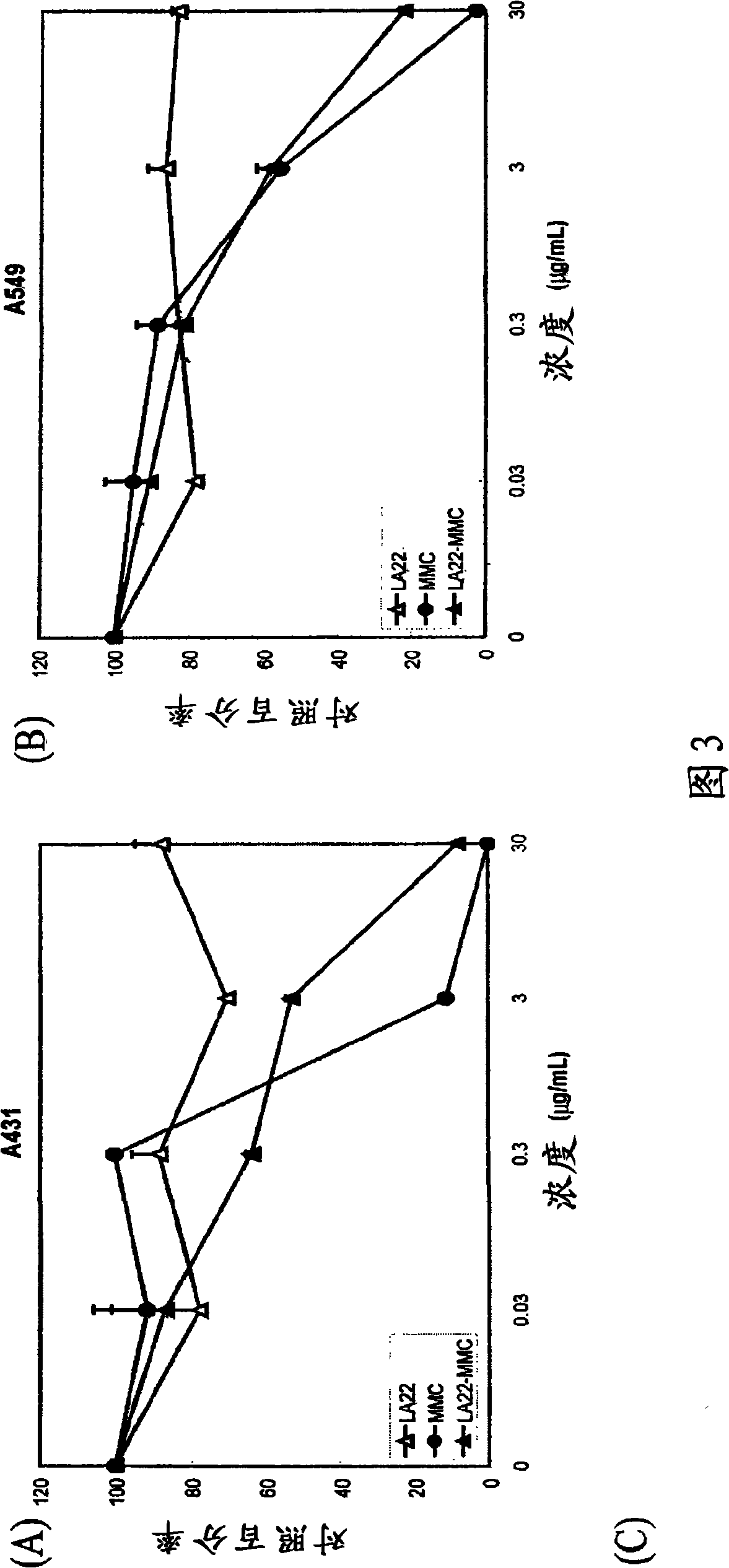
The present invention features monoclonal antibodies LA1 or LA22 conjugated with mitomycin C, pingyangmycin or other anti-cellular agents. The present invention also features other anti-EGFR antibodies conjugated with mitomycin C or pingyangmycin. The antibodies of the present invention can be used to treat cancers, including but not limited to, those of epithelial origin, such as glioblastoma or cancer of the lung, breast, head and neck, and bladder.
Owner:WELSON PHARMA
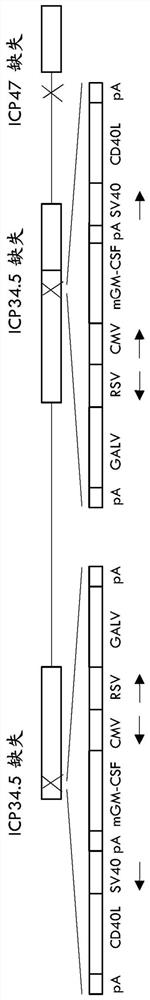
Treatment using oncolytic virus
PendingUS20210252135A1Direct effectImprove immunityPeptide/protein ingredientsViral antigen ingredientsAppendiceal carcinomaSquamous Carcinomas
An oncolytic virus for use in a method of treating or preventing cutaneous squamous cell carcinoma (CSCC), renal cell carcinoma (RCC), non-small cell lung cancer (NSCLC), triple negative breast cancer (TNBC), small cell lung cancer (SCLC), advanced recurrent head and neck cancer, squamous cell carcinoma of the head and neck (SCCHN), nasopharyngeal carcinoma (NPC), hepatocellular carcinoma (HCC), anal cancer, colorectal cancer (CRC), basal cell carcinoma (BCC), Merkel cell carcinoma, appendiceal carcinoma, sarcoma of the skin, recurrent melanoma after surgery, advanced or metastatic urothelialcarcinoma, liver metastases, microsatellite instability high cancer (MSI-H), mixed advanced solid tumors, virally caused cancer, locoregionally advanced cancer, pediatric cancer, cancer in patients with no or minimal pre-existing anti-cancer immunity, cancer as first line therapy, cancer in previously treated patients, cancer in patients who have not received checkpoint blockade therapy, and / or cancer in patients who have received checkpoint blockade therapy, wherein the oncolytic virus: is, or is derived from, a clinical isolate which has been selected by comparing the abilities of a panel of three or more clinical isolates of the same viral species to kill tumor cells of two or more tumor cell lines in vitro and selecting a clinical isolate which is capable of killing cells of two or more tumor cell lines more rapidly and / or at a lower dose in vitro than one or more of the other clinical isolates in the panel; comprises (i) a fusogenic protein-encoding gene; and (ii) an immune stimulatory molecule or an immune stimulatory molecule-encoding gene; comprises (i) a GM-CSF-encoding gene; and (ii) an immune co-stimulatory pathway activating molecule or an immune co-stimulatory pathway activating molecule-encoding gene; and / or comprises a gene encoding a CTLA-4 inhibitor.
Owner:REPLIMUNE
Method of inhibiting osteoclast activity
InactiveUS20140178376A1Peptide/protein ingredientsAntibody mimetics/scaffoldsMelanomaSquamous Carcinomas
Methods for inhibiting osteoclastogenesis by administering a soluble RANK polypeptide are disclosed. Such methods can be used to treat a variety of different cancers, including bone cancer, multiple myeloma, melanoma, breast cancer, squamous cell carcinoma, lung cancer, prostate cancer, hematologic cancers, head and neck cancer and renal cancer.
Owner:IMMUNEX CORP
Heteroaryl fused ring compound as well as preparation method and application thereof
PendingCN112979654AStrong inhibitory activityLow inhibitory doseOrganic active ingredientsOrganic chemistryDiseaseAryl
The invention provides a novel substituted heteroaryl fused ring compound and a preparation method thereof, and the novel substituted heteroaryl fused ring compound can be used as a TRK inhibitor and has a wide and good application prospect in TRK kinase mediated diseases such as melanoma, lung cancer, head and neck cancer, pancreatic cancer, breast cancer, thyroid cancer and glioma.
Owner:SCINNOHUB PHARM CO LTD
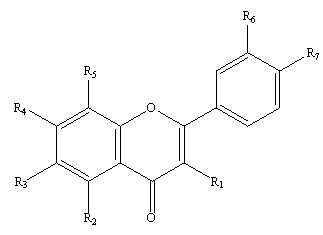
Isolicoflavonol-containing antitumor drug and application thereof
ActiveCN102908340AMeet the requirements of therapeutically effective doseOrganic active ingredientsOrganic chemistryOfficinalProstate cancer
The invention provides an isolicoflavonol-containing antitumor drug, wherein suspension type injections, tablets and microcapsules are processed and manufactured by taking isolicoflavonol as the active ingredient and combining with pharmaceutically acceptable auxiliary materials and carriers. The antitumor drug is used for preventing and treating breast cancer, ovarian cancer, cervical cancer and colon cancer and usual gastrointestinal cancer, lung cancer, kidney cancer, bladder cancer, prostate cancer, skin cancer, head and neck cancer, brain tumor or leukemia. Confirmed by pharmacological experiments and antitumor effect comparing evaluation, the isolicoflavonol coming from traditional Chinese medicine liquorice has the function of inhibiting the generation of new blood vessels and is capable of achieving the purpose of resisting tumors through inhibiting the generation of the new blood vessels of the tumors. The invention provides an industrial preparation method of the isolicoflavonol serving as a medicinal material, comprising extraction, separation, grain size specification and other concrete process requirements.
Owner:BIOLOGY INST OF SHANDONG ACAD OF SCI +1
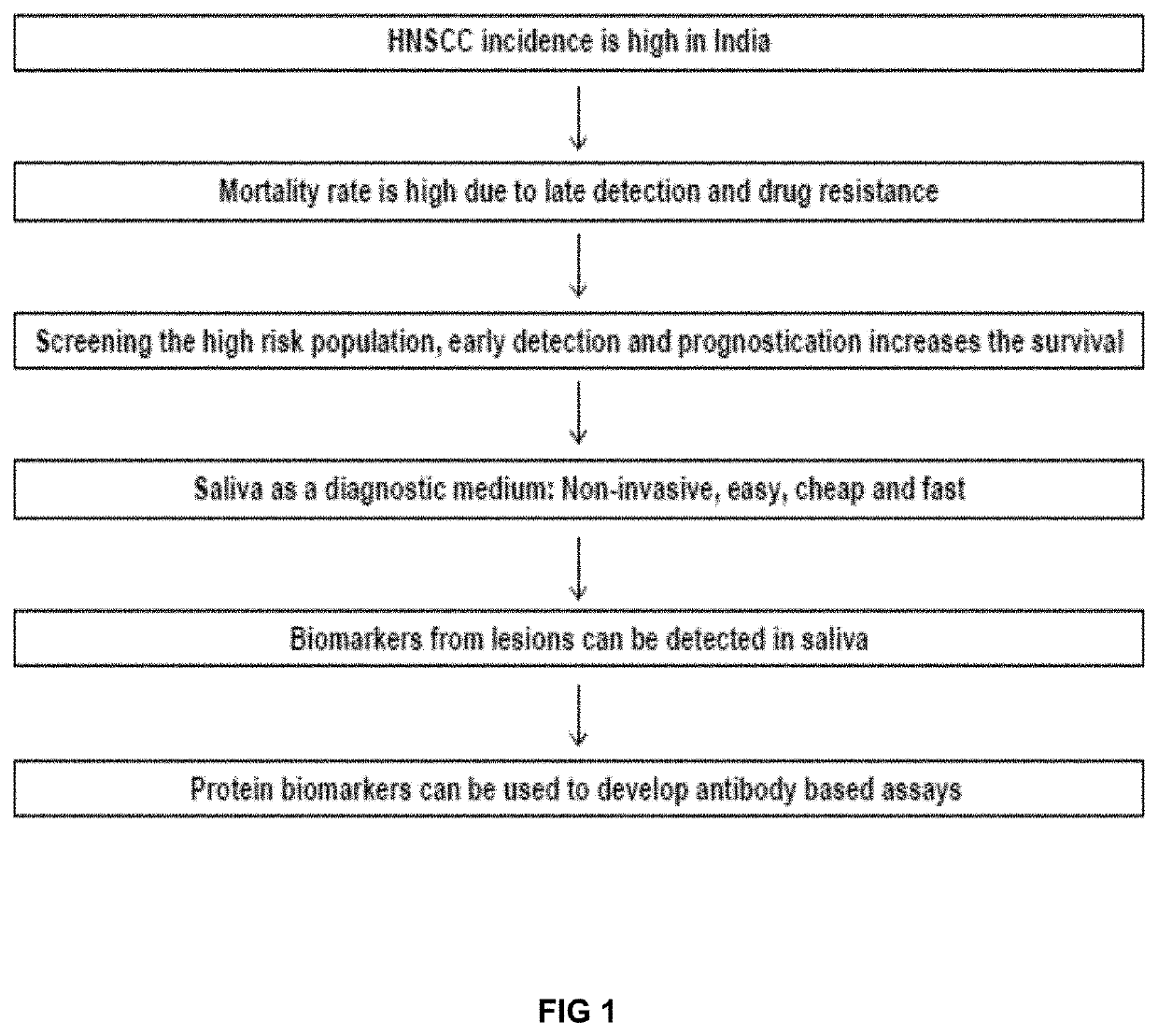
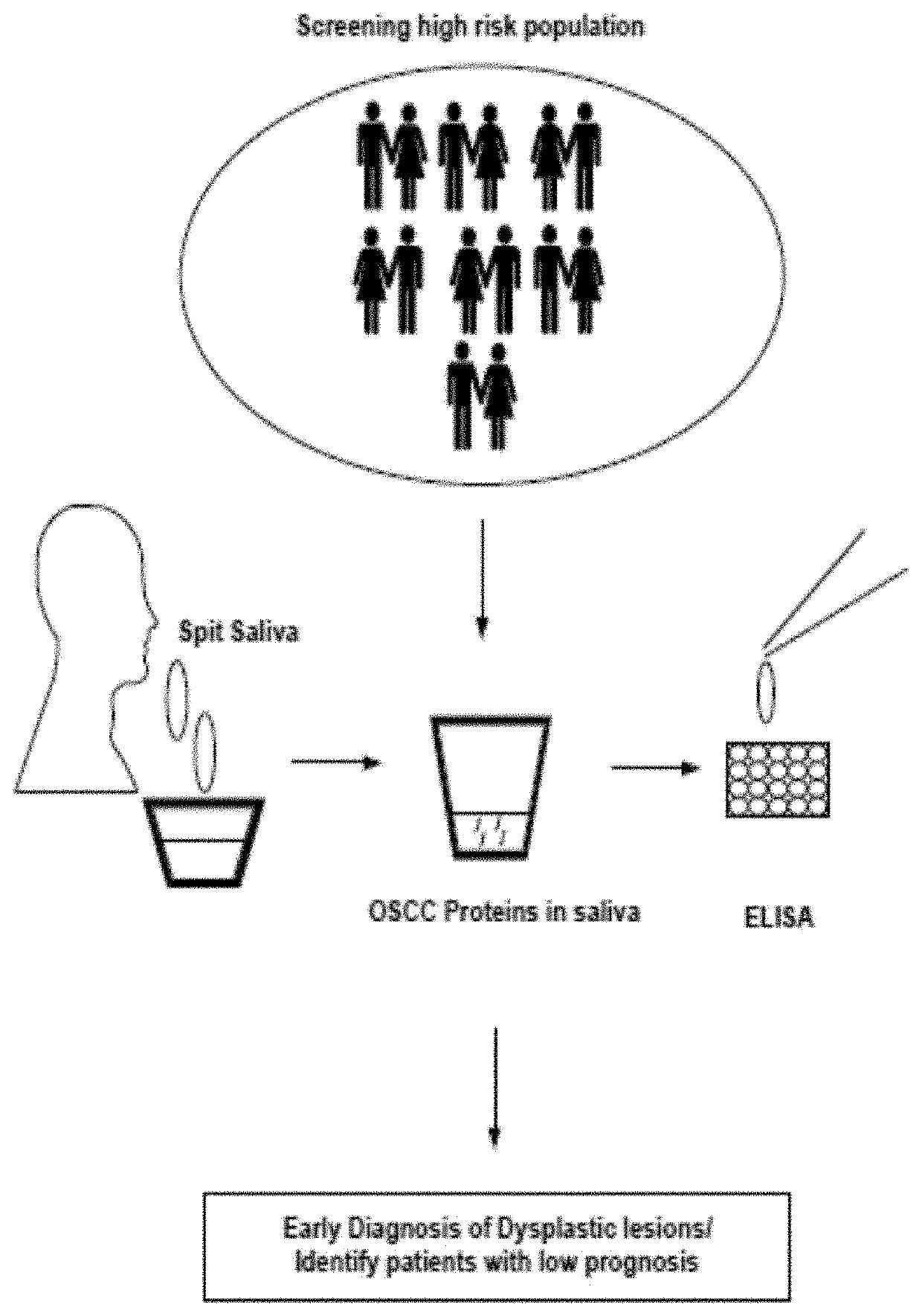
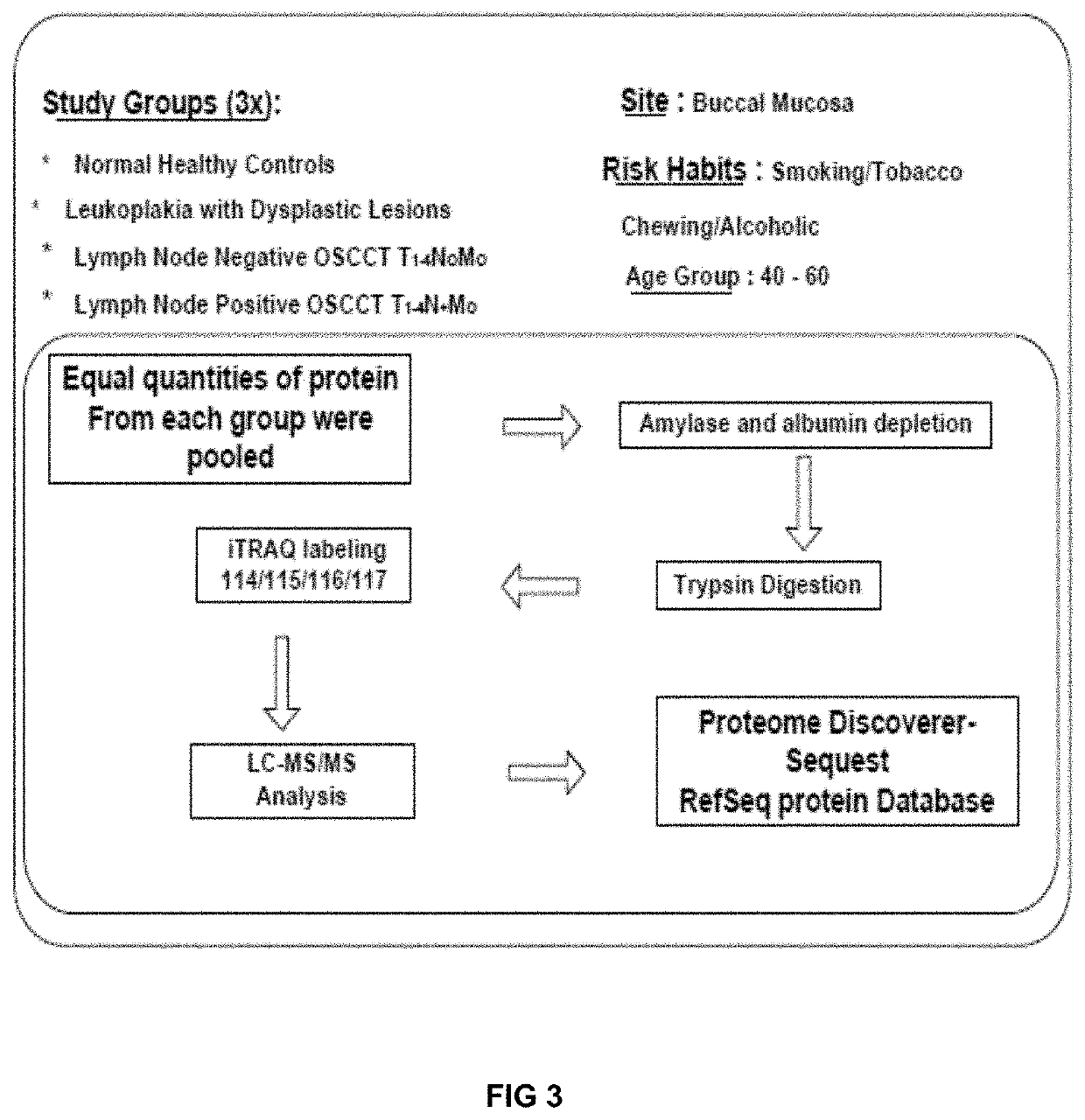
Sall.ivary protein biomarkers for the diagnosis and prognosis of head and neck cancers, and precancers
PendingUS20210018507A1Effective clinical diagnosisEffective diagnosisDisease diagnosisProtein profilingNon invasive
The present disclosure relates to a panel of biomarkers for the detection / diagnosis and prognosis of head and neck squamous cell carcinoma (HNSCC). Biomarkers for the detection / diagnosis of carcinoma cells identified by proteomic profiling and, detection / identification of selected proteins that are differentially regulated, in a biological sample from an individual as candidate markers. The biomarkers are useful for non-invasive early detection / prognosis focusing on the protein profiling of saliva at different stages of oral pre-cancer and cancer progression. Furthermore, the present invention deals with novel methods of diagnosing and for providing a prognosis for oral cancer and periodontal disease. In addition, the invention also provides kits that are useful for the practice of the methods of the invention.
Owner:MAZUMDAR SHAW MEDICAL FOUND +3
Automatic segmentation method for oral cancer epithelial tissue region of pathology image
The invention discloses an automatic segmentation method for an oral cancer epithelial tissue region of a pathological image. The method comprises the following steps: S100, preprocessing the pathological image and a label of a pathologist by using a method for extracting a pixel block patch; S200, selecting part of the preprocessed pathological images as training samples to form a training set, and taking the rest pathological images as verification samples to form a verification set; S300, constructing a convolutional neural network UNet model, and training and verifying the UNet model by adopting the training set and the verification set in the step S200 to obtain a final UNet model; and S400, applying the final UNet model to a multi-center external test set to automatically generate anepithelial tissue region. According to the method, training, verification and testing are not carried out on each data set in head and neck cancer detection, only TMA data is used for training, testing is carried out on a multi-center external WSI data set, and the method is more persuasive to unknown images.
Owner:SHAANXI NORMAL UNIV
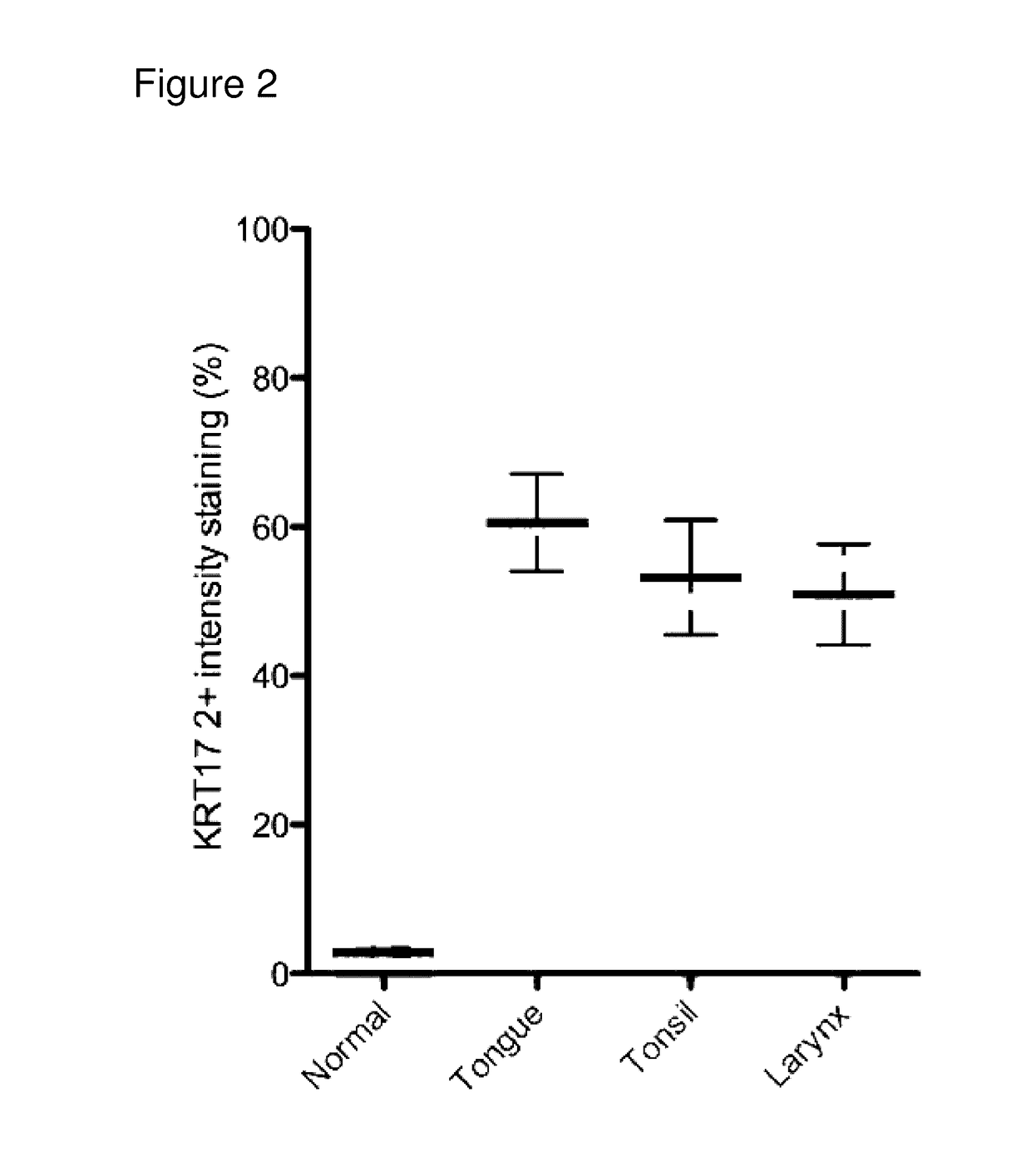
Keratin 17 as a biomarker for head and neck cancers
InactiveUS20170082632A1Reduced incidence of survivalShort survival timeMicrobiological testing/measurementDisease diagnosisKeratin 17Oncology
The current disclosure provides methods for detecting and analyzing KRT17 expression in a sample obtained from a subject. The current disclosure also pertains to methods and kits for identifying a mammalian subject with head and neck squamous cell carcinoma. The current disclosure further provides methods and kits for determining the likelihood of survival of a subject having head and neck squamous cell carcinoma.
Owner:THE RES FOUND OF STATE UNIV OF NEW YORK
Development of prognostic markers from the saliva of head and neck cancer patients
The present invention relates to biomarkers that are useful for the detection of cancer. The invention further relates to biomarkers and methods of using biomarkers for the early detection of head and neck cancer.
Owner:UNIV OF SOUTHERN CALIFORNIA
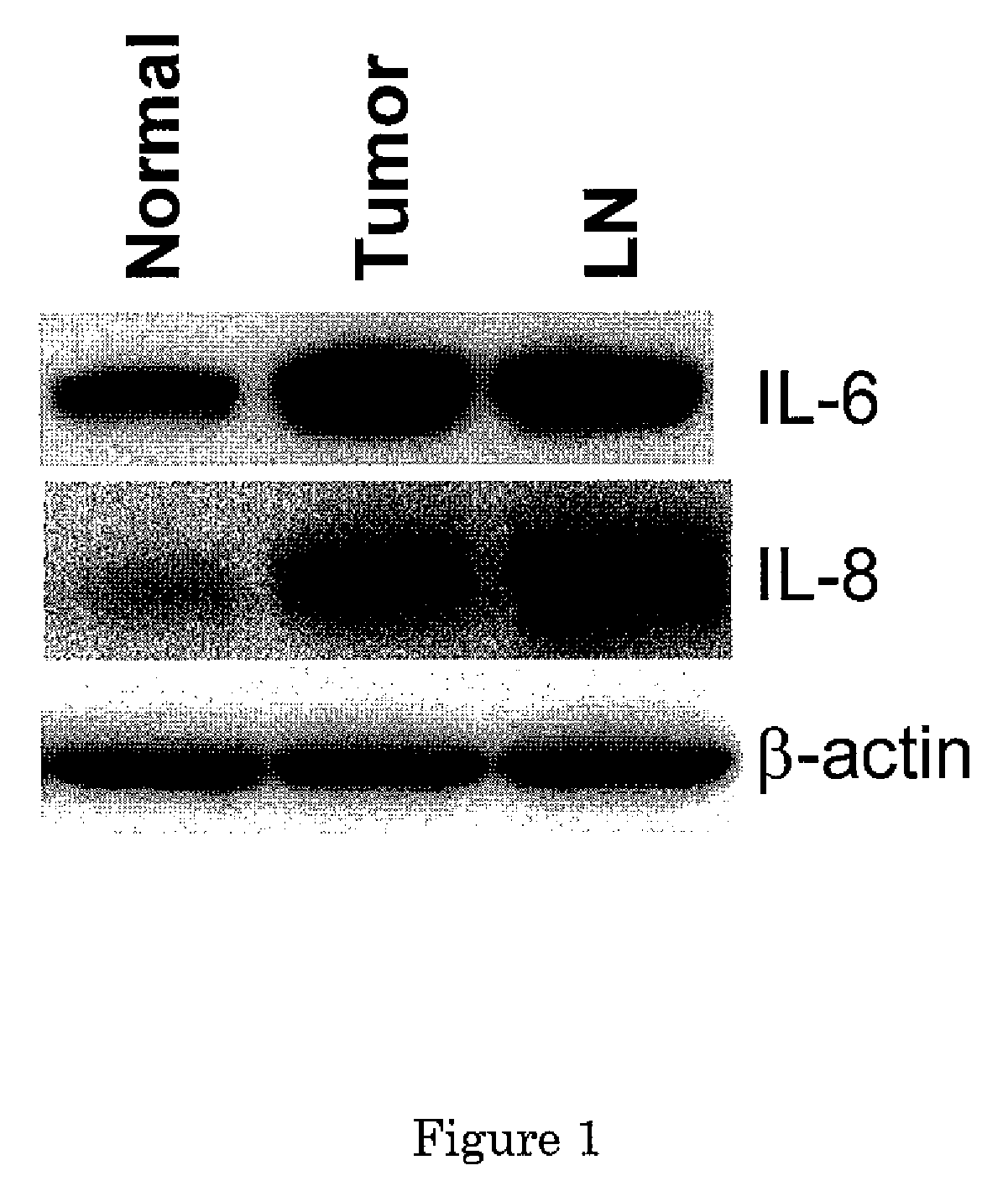
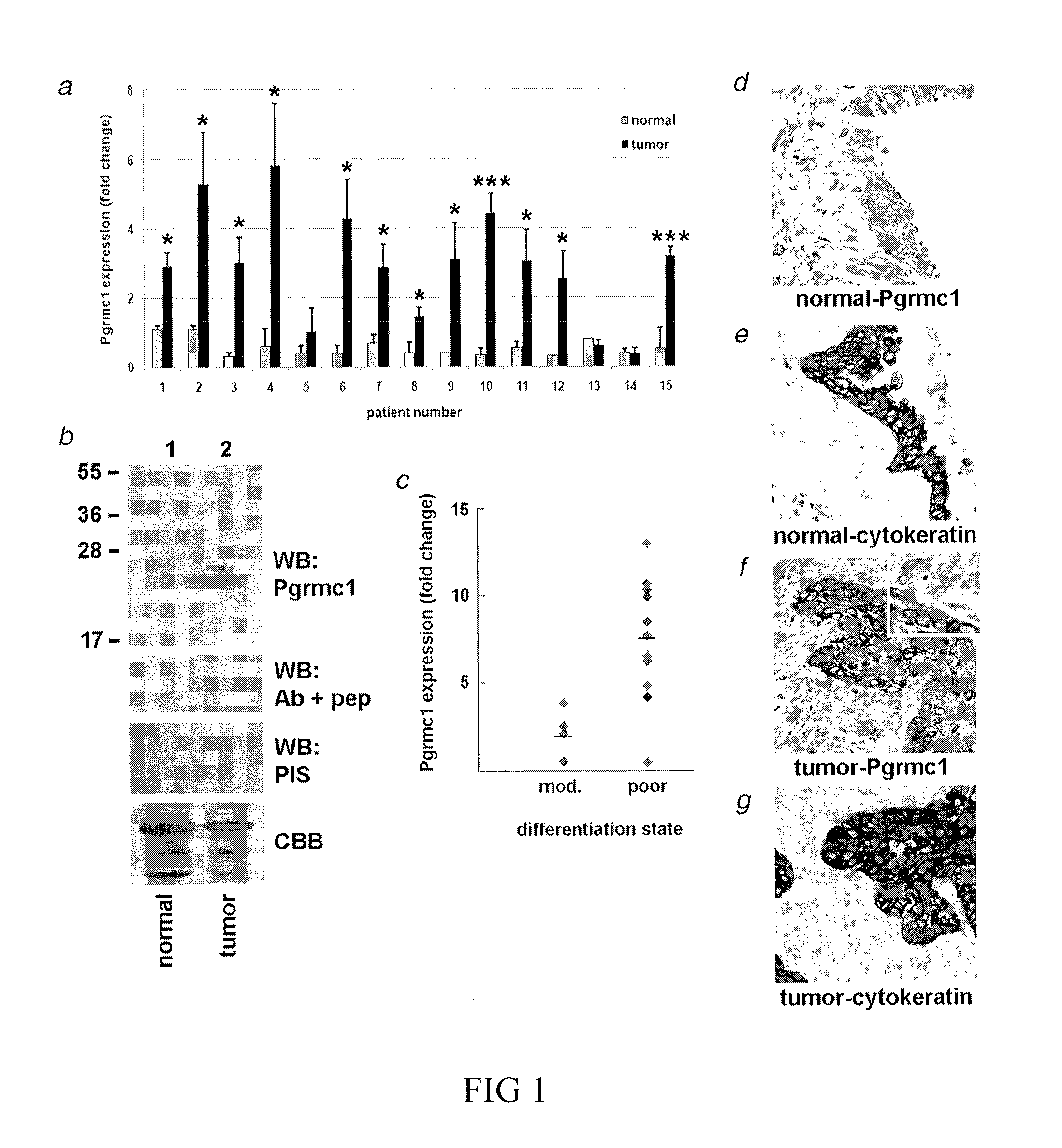
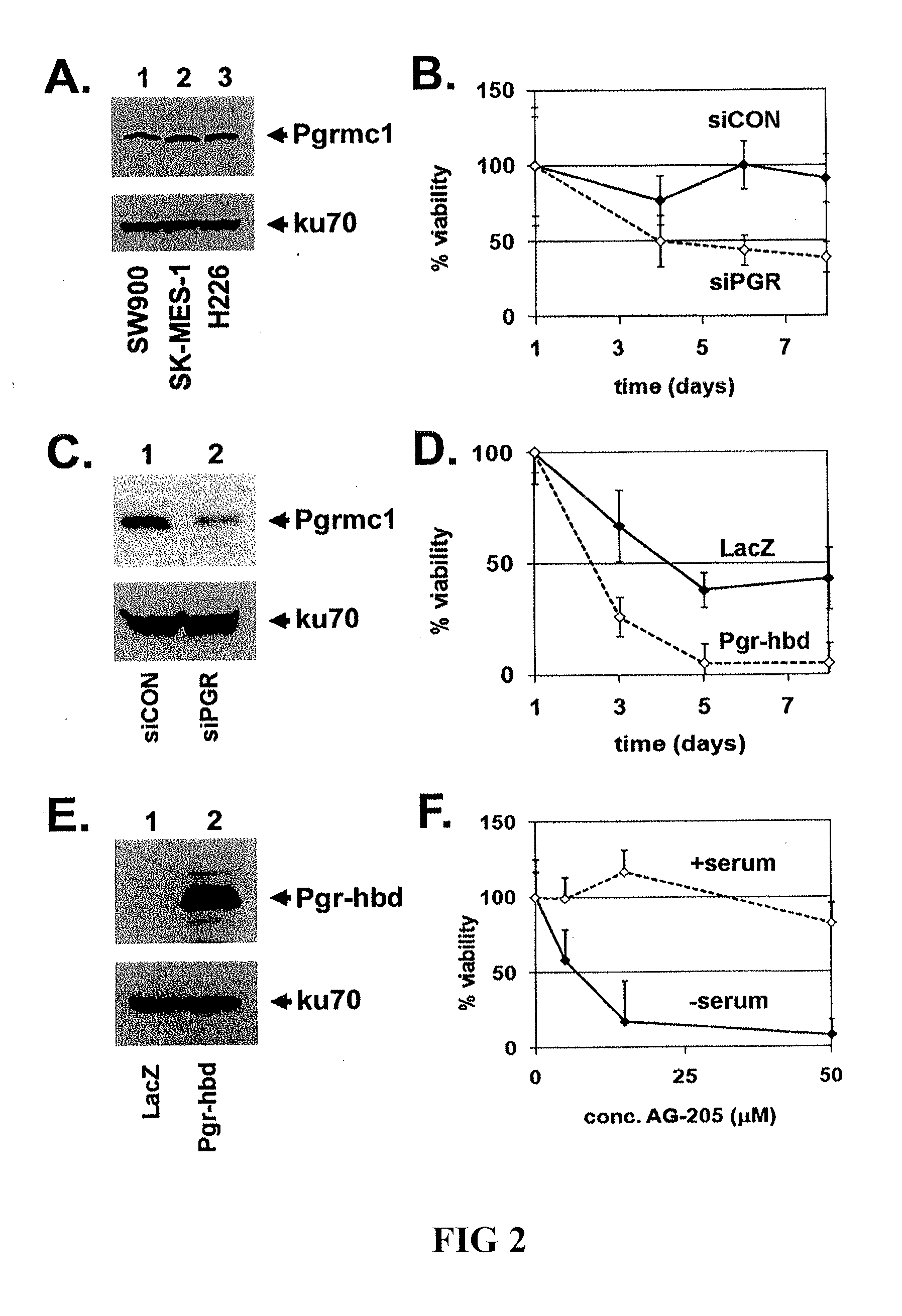
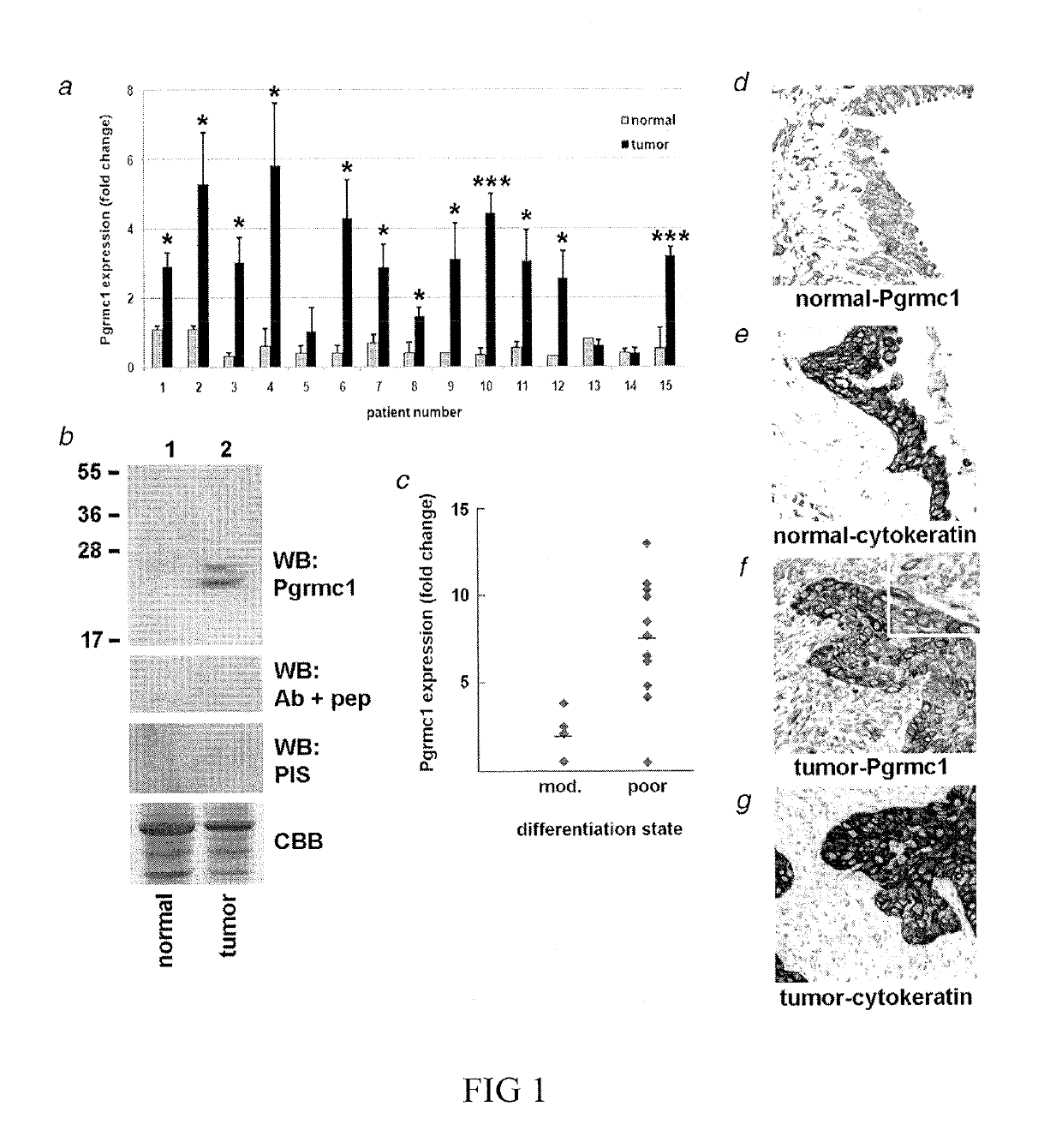
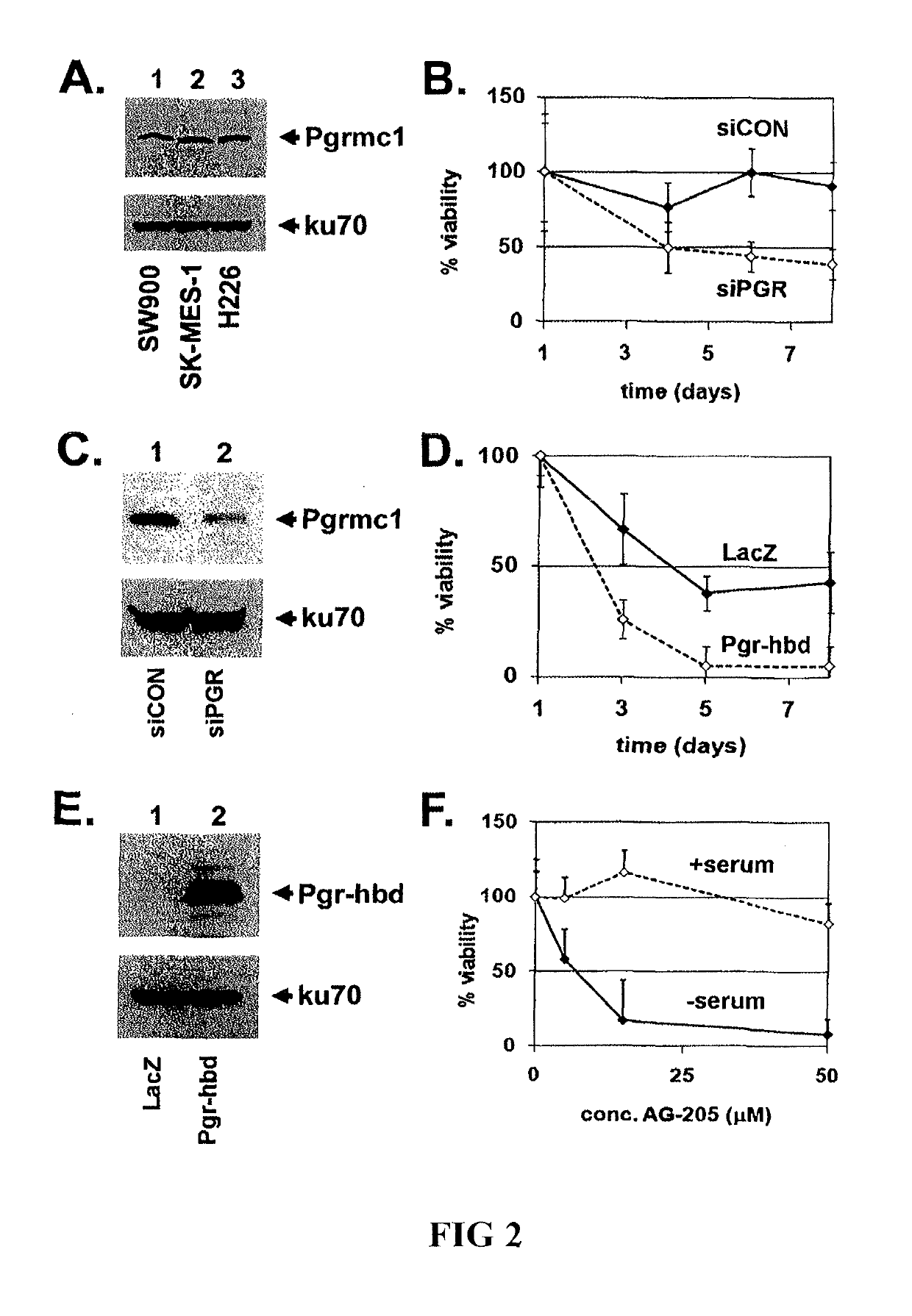
Secreted Tumor-Associated Cytochrome as a Blood-Based Biomarker for Cancer
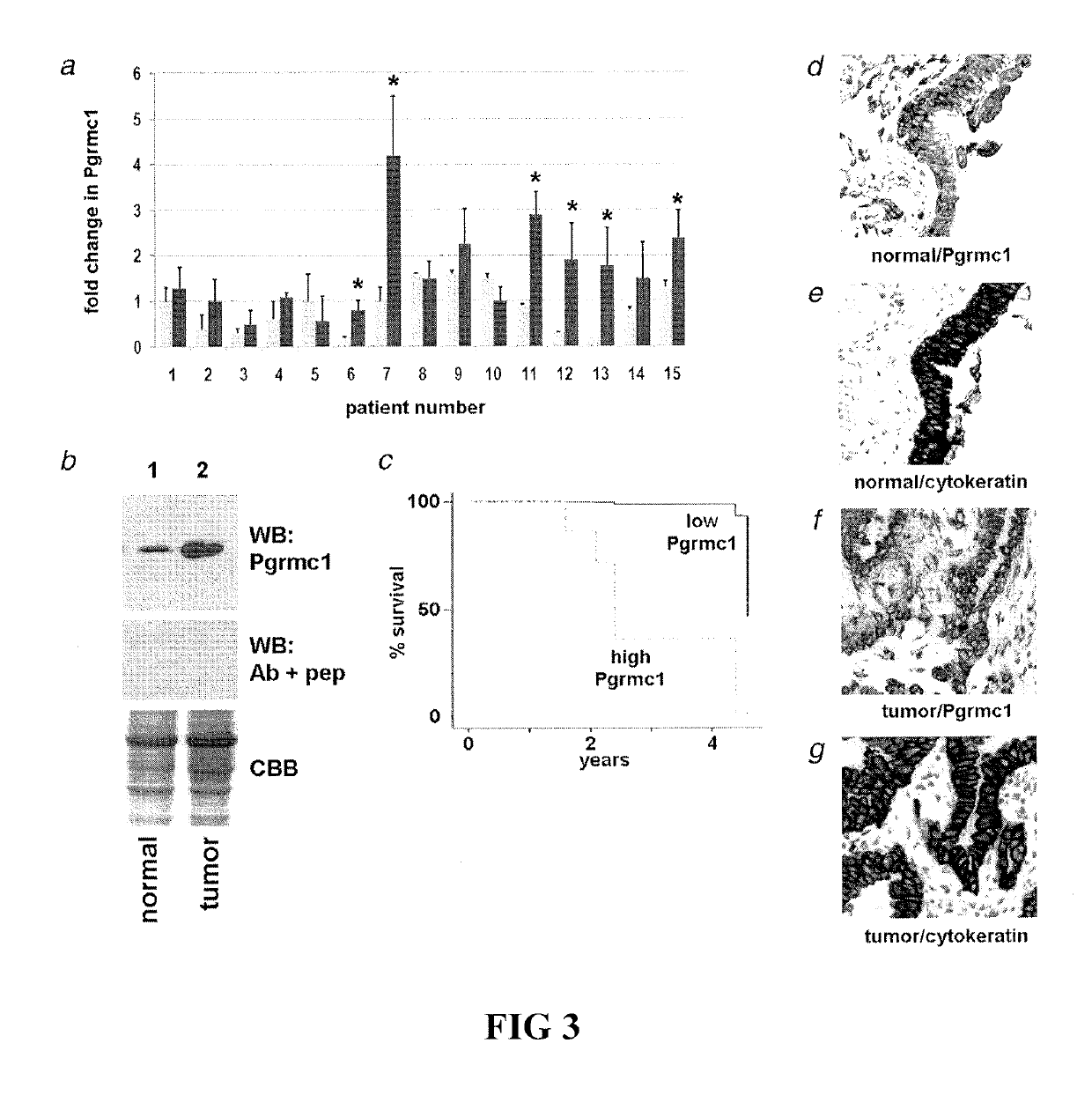
Disclosed herein are methods for detecting Pgrmc1 in bodily fluids, e.g., blood, plasma and serum, wherein detection of Pgrmc1 is a biomarker for the presence of cancer, e.g., lung cancer or head or neck cancer. Pgrmc1 levels in bodily fluids may be used to predict patient prognosis, e.g., survival and response to therapy.
Owner:UNIV OF KENTUCKY RES FOUND
Combination therapy for head and neck cancer
ActiveUS20170072010A1Lower Level RequirementsReduce in quantityPeptide/protein ingredientsPharmaceutical delivery mechanismAgonistOncology
This invention relates to pharmaceutical composition and methods of using RXR agonist and / or RAR agonist for the treatment or prevention of head and neck cancer.
Owner:CORNELL UNIVERSITY
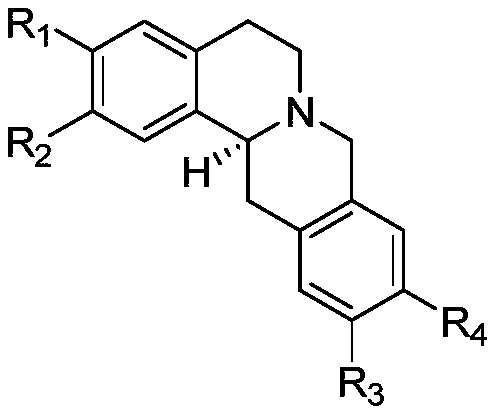
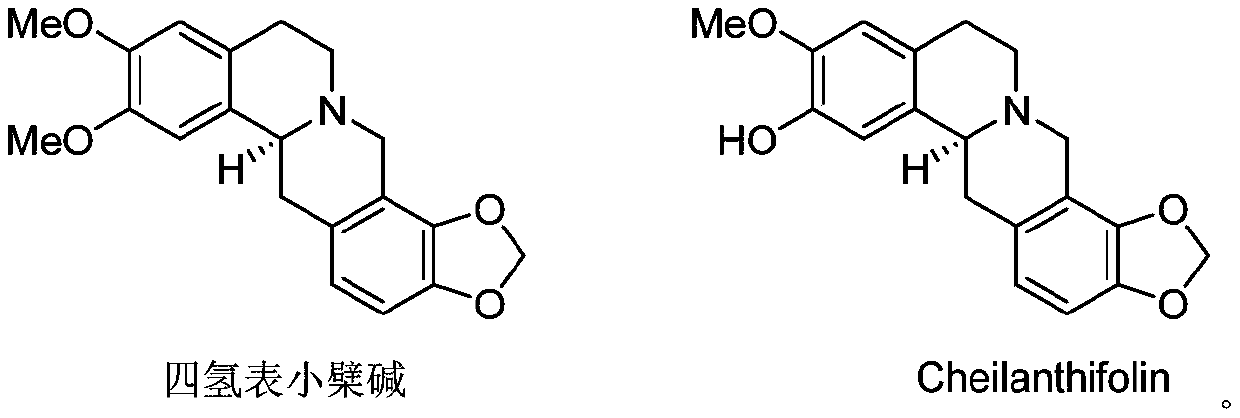
Application of protoberberine-type compound with TDO selective inhibition activity in medicament preparation
The invention discloses the application of a protoberberine-type compound in preparation of TDO inhibitors or in preparation of medicaments for treating or preventing TDO-mediated diseases. Results ofenzyme activity inhibition experiments show that compounds, including tetrahydroepiberberine and Cheilanthifolin, have the inhibitory effect on the tryptophan 2,3-dioxygenase, and can be used for preparing the TDO inhibitors and preparing the medicaments or derivatives for treating or preventing the TDO-mediated diseases, wherein the TDO-mediated diseases mainly include tumors such as liver cancer, spongioblastoma, mesothelioma, head and neck cancer, non-small cell lung cancer, bladder cancer and breast cancer, or metabolic disorders such as depression, organ transplant rejection, neuropathicdisorders and obesity, or autoimmune diseases, and in the diseases, the TDO expression is up-regulated or the activity is obviously enhanced.
Owner:KUNMING INST OF BOTANY - CHINESE ACAD OF SCI
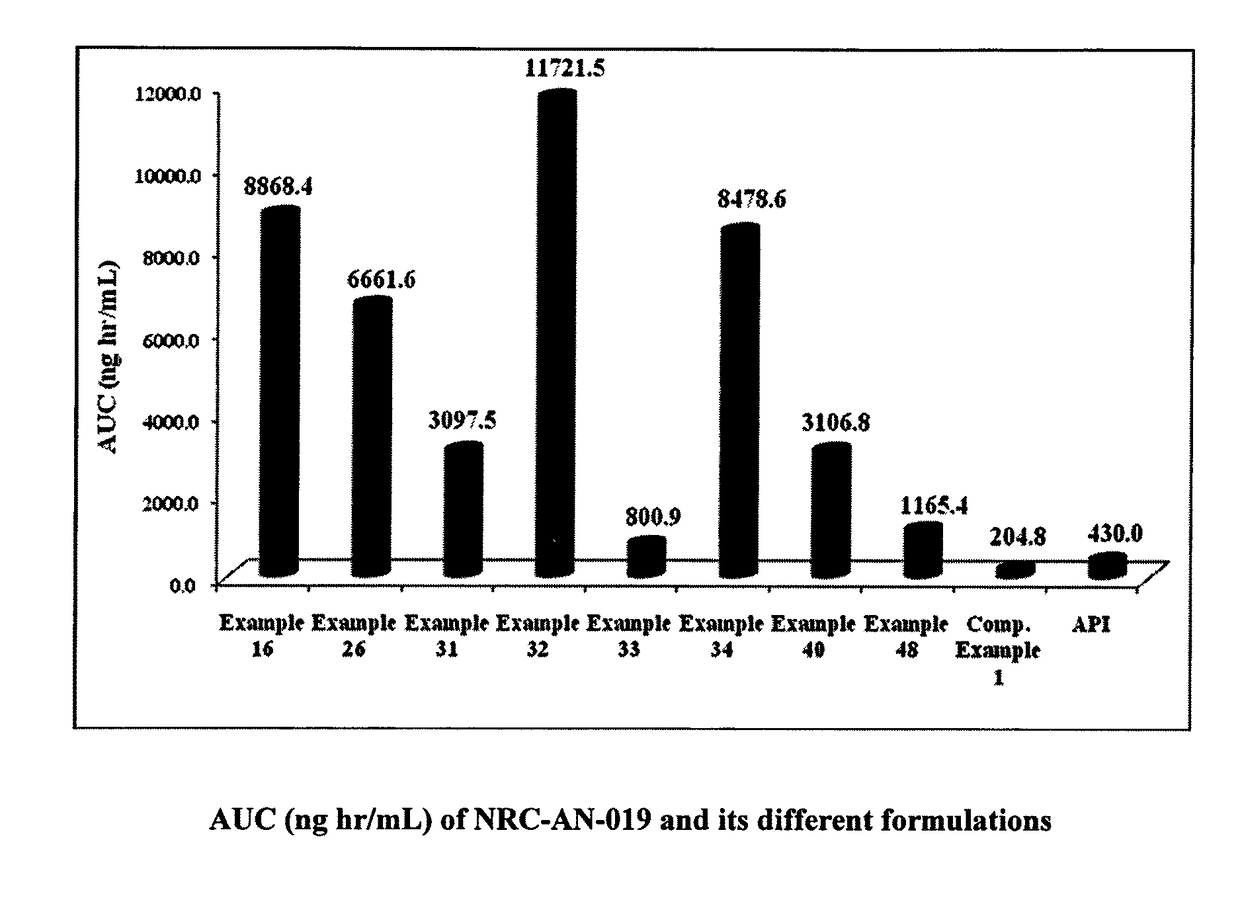
Formulation comprising phenylaminopyrimidine derivative as active agent
InactiveUS9895367B2Easy to useImprove the level ofBiocideOrganic active ingredientsBcr-Abl tyrosine-kinase inhibitorMyeloid leukemia
An oral pharmaceutical formulation containing an effective amount of NRC-AN-019 including its pharmaceutically acceptable salts and polymorphs such as Form I, Form II and Form III thereof to improve the bioavailability intended for self-emulsification upon its contact with the gastro-intestinal fluid. The invention also relates to a process for the preparation of oral solution containing NRC-AN-019 in an effective concentration for the better therapy against Chronic Myeloid Leukemia as BCR-ABL tyrosine kinase inhibitor and against other tumors such as head and neck cancer, prostate cancer and the like.
Owner:NATCO PHARMA LTD
Broad-spectrum Anti-cancer treatment based on iminocamptothecin derivatives
A subclass of camptothecin derivatives is disclosed to be useful for the preparation of a medicament for the treatment of a cancer or tumor pathology selected from the group consisting of head and neck carcinoma, pancreas carcinoma, melanoma, bladder carcinoma, mesothelioma and epidermoid skin carcinoma.
Owner:SIGMA TAU IND FARMACEUTICHE RIUNITE SPA +1
Treatment with Anti-alpha2 integrin antibodies
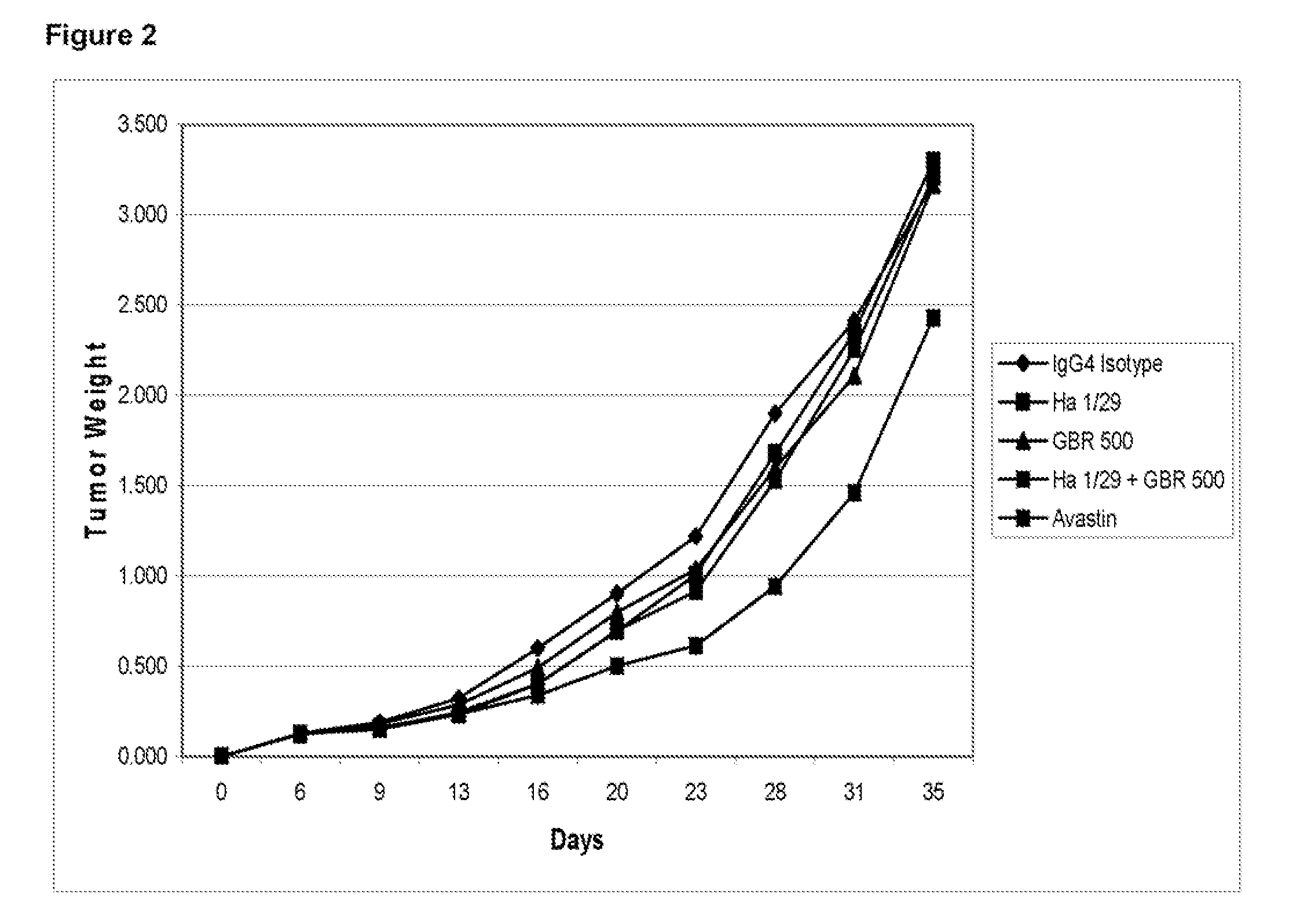
ActiveUS20100158904A1Inhibit bindingAvoid stickingNervous disorderMuscular disorderUterine carcinomaLymphoid Tumor
The invention relates to treatment of cancer. More specifically the invention relates to methods of treating cancer selected from the group consisting of squamous cell cancer, lung cancer including small-cell lung cancer, non-small cell lung cancer, adenocarcinoma of the lung, and squamous carcinoma of the lung, cancer of the peritoneum, hepatocellular cancer, gastric or stomach cancer including gastrointestinal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, hepatoma, breast cancer, colon cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney or renal cancer, liver cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma and various types of head and neck cancer, as well as B-cell lymphoma including low grade / follicular non-Hodgkin's lymphoma (NHL); small lymphocytic (SL) NHL; intermediate grade / follicular NHL; intermediate grade diffuse NHL; high grade immunoblastic NHL; high grade lymphoblastic NHL; high grade small non-cleaved cell NHL; bulky disease NHL; mantle cell lymphoma; AIDS-related lymphoma; and Waldenstrom's Macroglobulinemia; chronic lymphocytic leukemia (CLL); acute lymphoblastic leukemia (ALL); Hairy cell leukemia; chronic myeloblastic leukemia; and post-transplant lymphoproliferative disorder (PTLD), as well as abnormal vascular proliferation associated with phakomatoses, edema such as that associated with brain tumors, Meigs' syndrome, melanoma, mesothelioma, multiple myeloma, fibrosarcoma, osteosarcoma and epidermoid carcinoma, by administering antibodies directed to α2β1 integrin.
Owner:ICHNOS SCI SA
Secreted tumor-associated cytochrome as a blood-based biomarker for cancer
Disclosed herein are methods for detecting Pgrmc1 in bodily fluids, e.g., blood, plasma and serum, wherein detection of Pgrmc1 is a biomarker for the presence of cancer, e.g., lung cancer or head or neck cancer. Pgrmc1 levels in bodily fluids may be used to predict patient prognosis, e.g., survival and response to therapy.
Owner:UNIV OF KENTUCKY RES FOUND
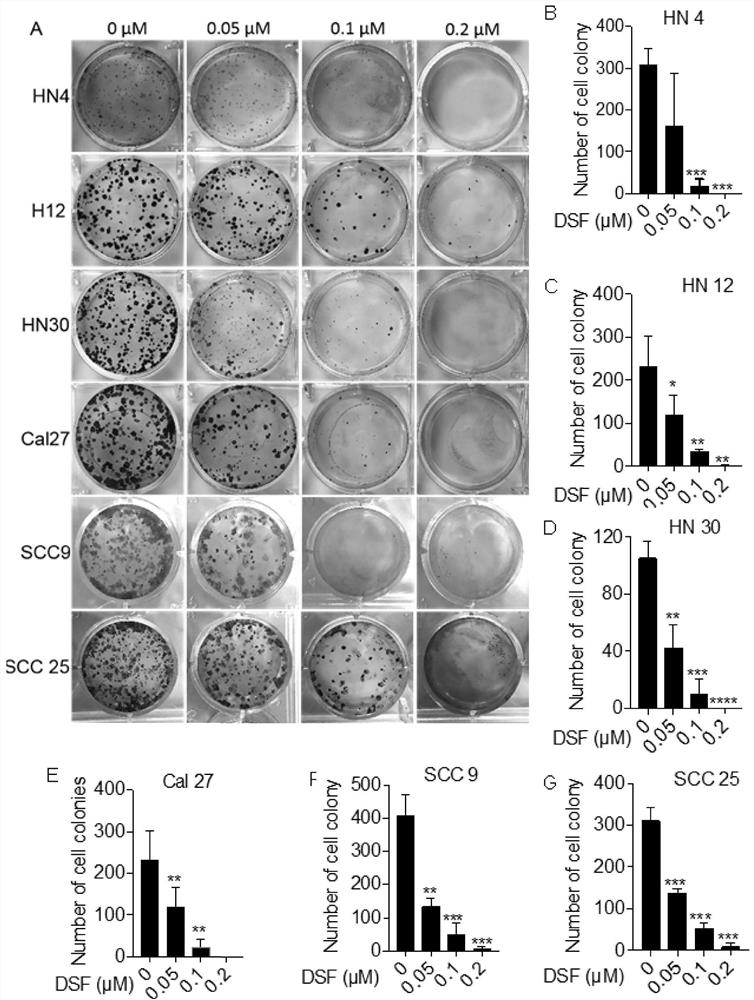
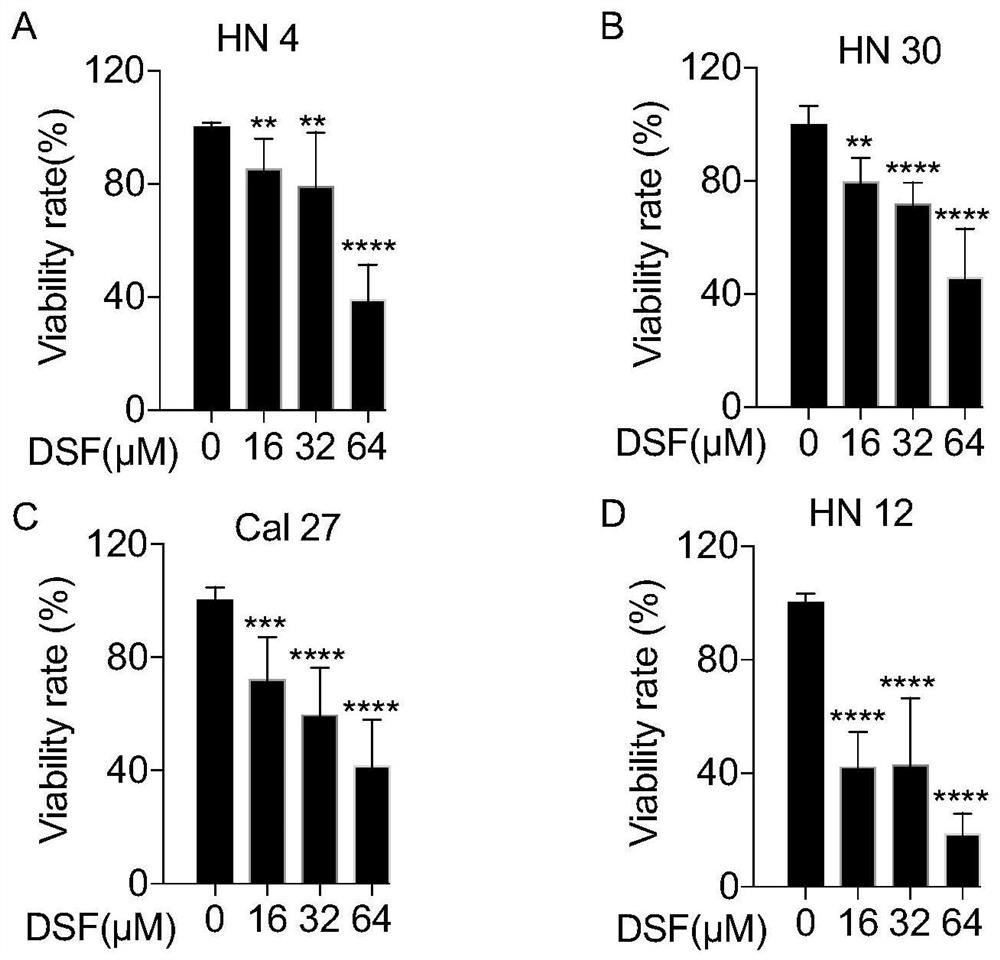
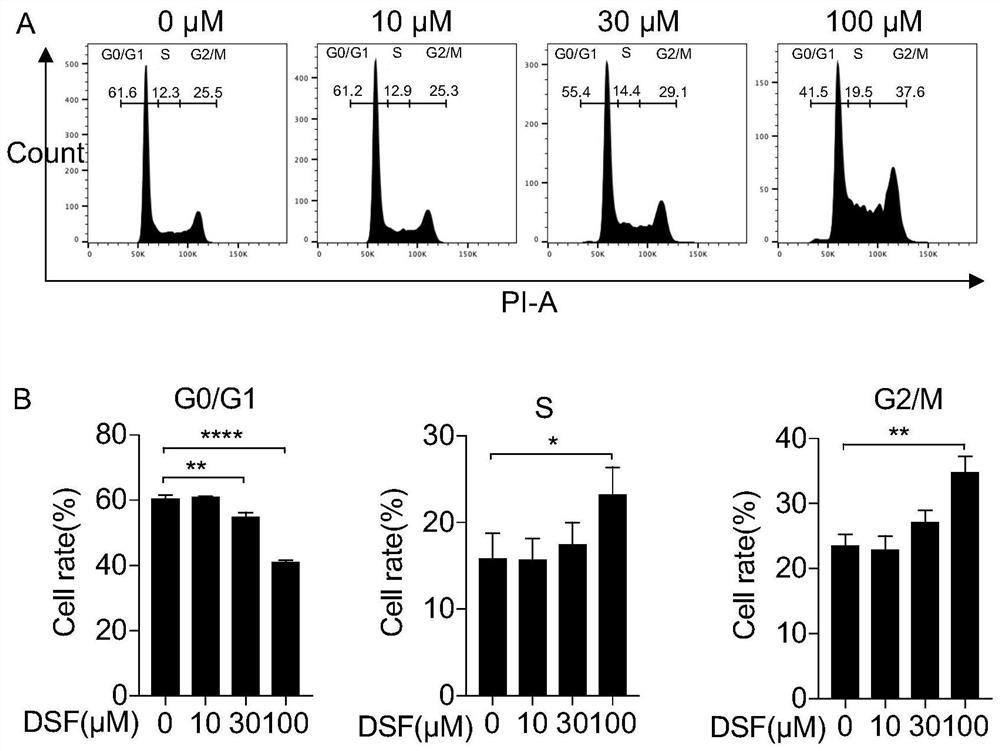
Application of disulfiram in preparation of drugs capable of resisting head and neck cancer and fibrosis
InactiveCN111803477AImprove side effectsImprove securityOrganic active ingredientsRespiratory disorderDiseaseOncology
The invention discloses an application of disulfiram in preparation of drugs capable of resisting head and neck cancer and drugs capable of resisting liver fibrosis. According to the invention, the disulfiram is discovered to have a proliferation inhibition effect and an apoptosis promotion effect on head and neck cancer cells for the first time, and the disulfiram is proved to be a potential clinical treatment drug for head and neck cancer diseases. The head and neck cancer refers to a head and neck cancer including oral and maxillofacial tumors (oral cancer, gingival cancer and buccal cancer) and neck tumors. The disulfiram is also discovered to have a relieving effect on lung fibrosis for the first time; and the disulfiram is indicated to have an important significance on clinical treatment of the lung fibrosis and provide a new direction for treatment of the lung fibrosis. The lung fibrosis refers to idiopathic lung fibrosis and clinical secondary lung fibrosis.
Owner:SHANGHAI JIAO TONG UNIV
Treatment of head and neck cancer
PendingCN112004828AImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsOncologyHead and neck
This invention relates to the use of NKG2A-targeting agents for the treatment of cancers, notably head and neck cancers, in a patient having received prior treatment with cetuximab. This invention also provides advantageous combination regimens for use with NKG2A-targeting agents for the treatment of cancers.
Owner:INNATE PHARMA SA
Methods for detecting head and neck cancer
The present invention relates to the field of pharmacogenomics and in particular to detecting the presence or absence of methylated genomic DNA derived from head and neck cancer cells in biological samples such as body fluids that contain circulating DNA from the cancer cells. This detection is useful for an early and reliable diagnosis of head and neck cancer and the invention provides methods and oligonucleotides suitable for this purpose.
Owner:EPIGENOMICS AG
Polypeptides for improved response to Anti-cancer therapy
The present disclosure provides E2F5 mimetic polypeptides. Further provided are methods for the treatment of cancer, such as head and neck cancer, comprising administering the E2F5 mimetic polypeptides alone or in combination with an additional anti-cancer therapy, such as a chemotherapeutic.
Owner:MUSC FOUND FOR RES DEV
Compositions and methods for diagnosis and treatment of cancer
The present invention relates to the diagnosis and treatment of diseases expressing Fibronectin Extra Domain B (EDB), such as diseases characterized by tissue remodeling and / or angiogenesis, in particular cancerous diseases, such as head and neck, brain, colorectal, lung, prostate and breast cancer. More particularly, the invention concerns peptides targeting fibronectin extra domain B.
Owner:生物技术欧洲股份公司
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com