Patents
Literature
582 results about "Paranasal Sinus Carcinoma" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
A malignant epithelial neoplasm arising in the paranasal sinus.
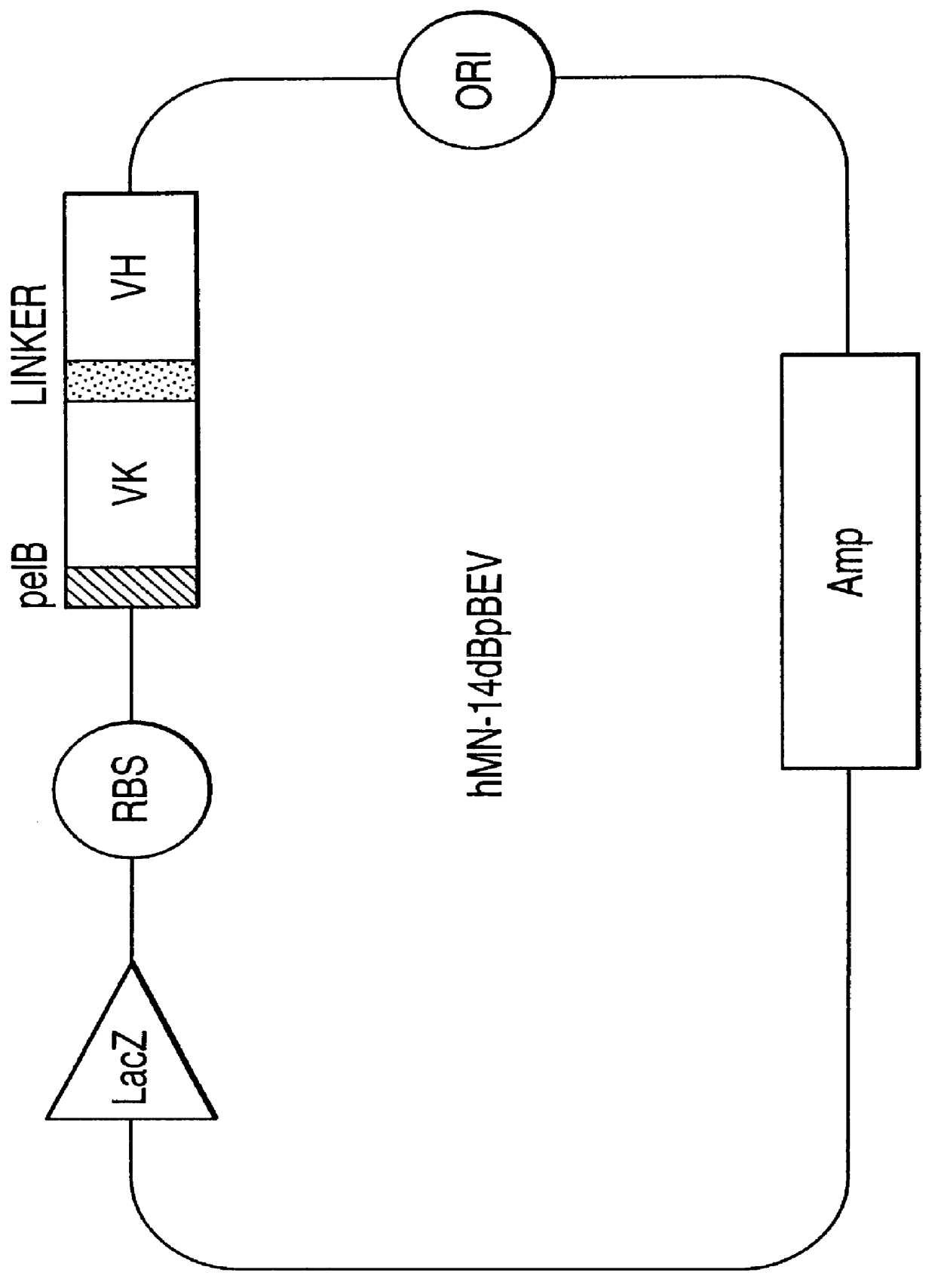
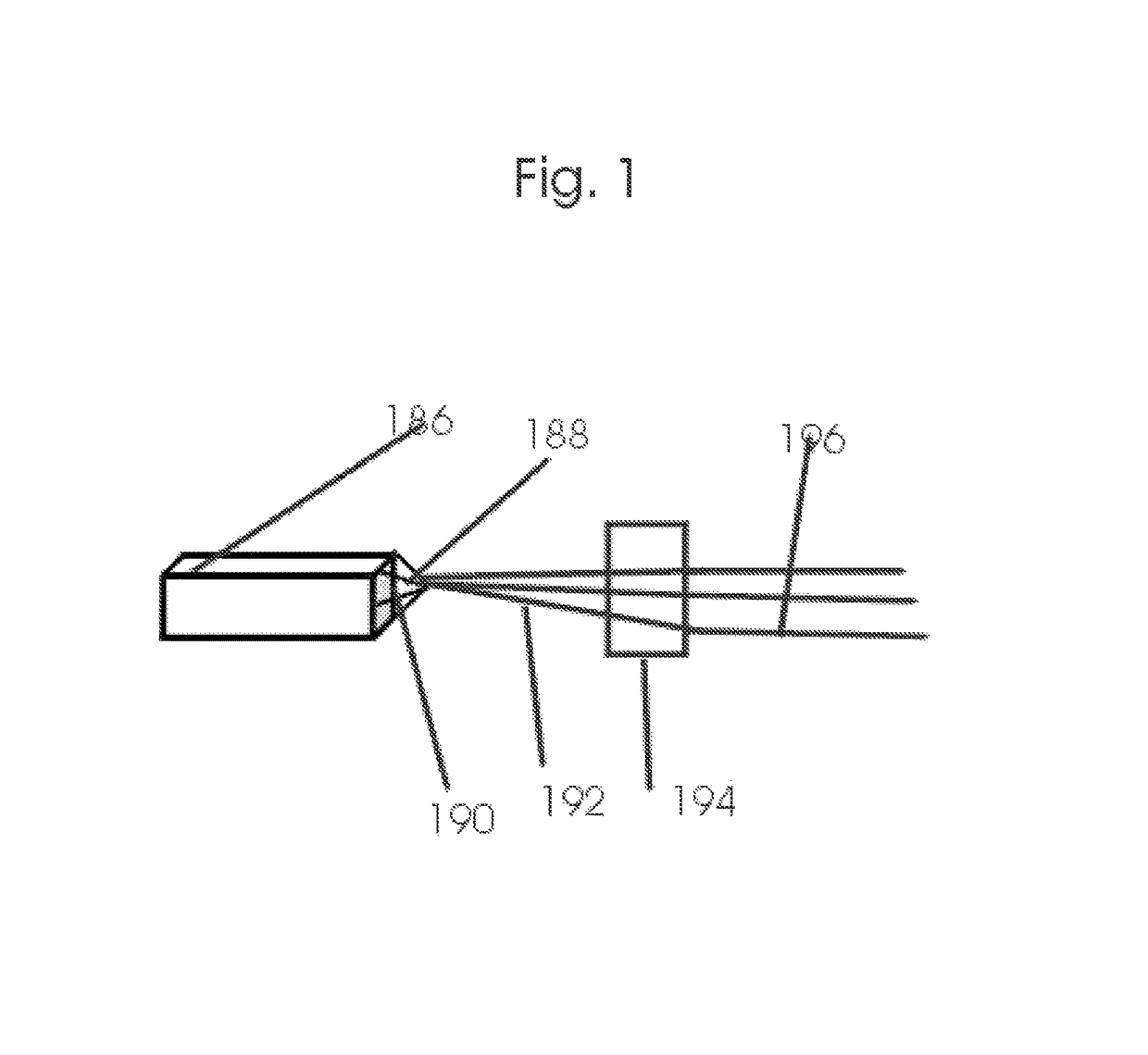
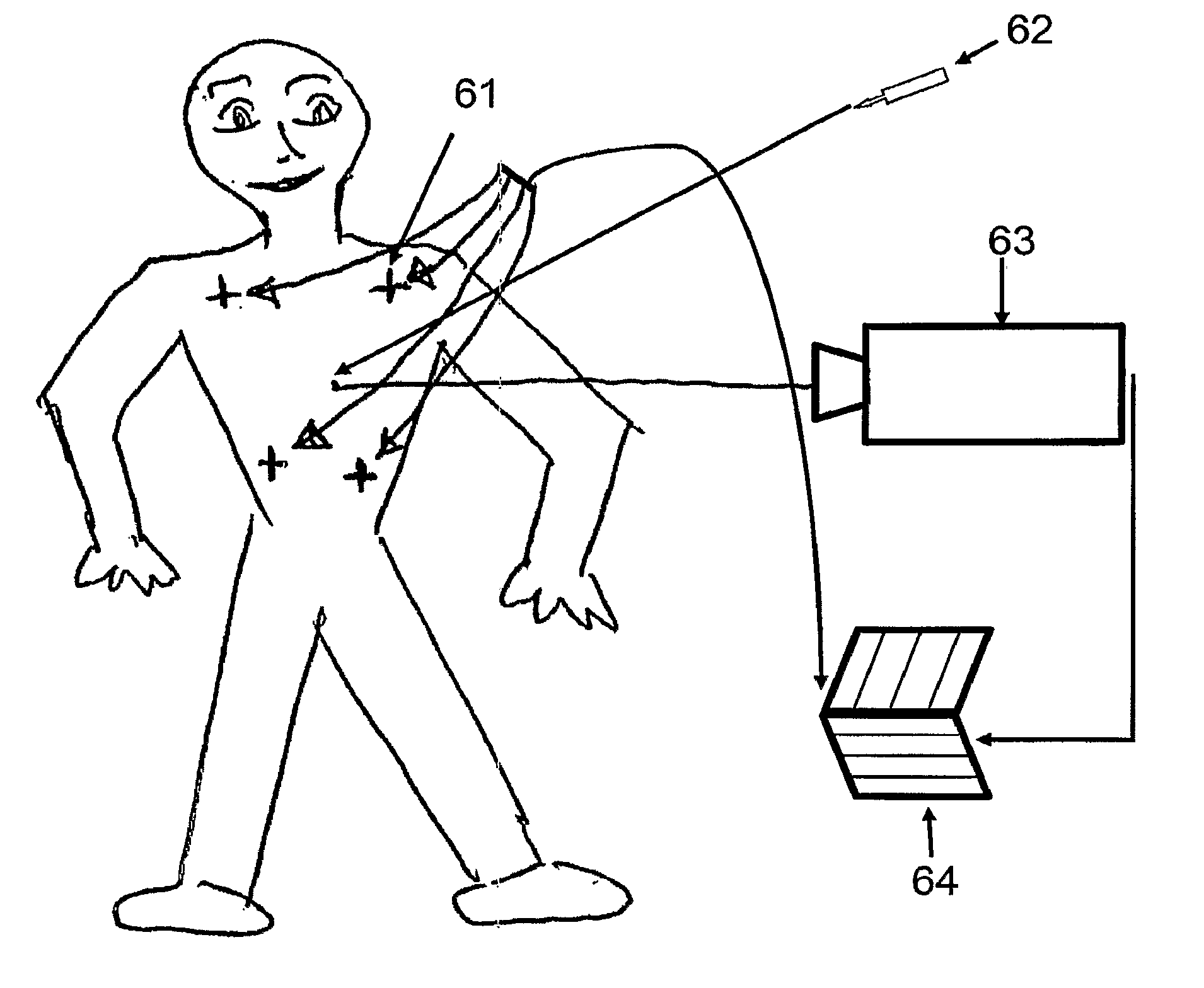
Intraoperative, intravascular, and endoscopic tumor and lesion detection, biopsy and therapy
InactiveUS6096289ADiscriminationImprove discriminationUltrasonic/sonic/infrasonic diagnosticsSurgeryNon malignantAntibody fragments
Methods are provided for close-range intraoperative, endoscopic and intravascular deflection and treatment of lesions, including tumors and non-malignant lesions. The methods use antibody fragments or subfragments labeled with isotopic and non-isotopic agents. Also provided are methods for detection and treatment of lesions with photodynamic agents and methods of treating lesions with a protein conjugated to an agent capable of being activated to emit Auger electron or other ionizing radiation. Compositions and kits useful in the above methods are also provided.
Owner:IMMUNOMEDICS INC
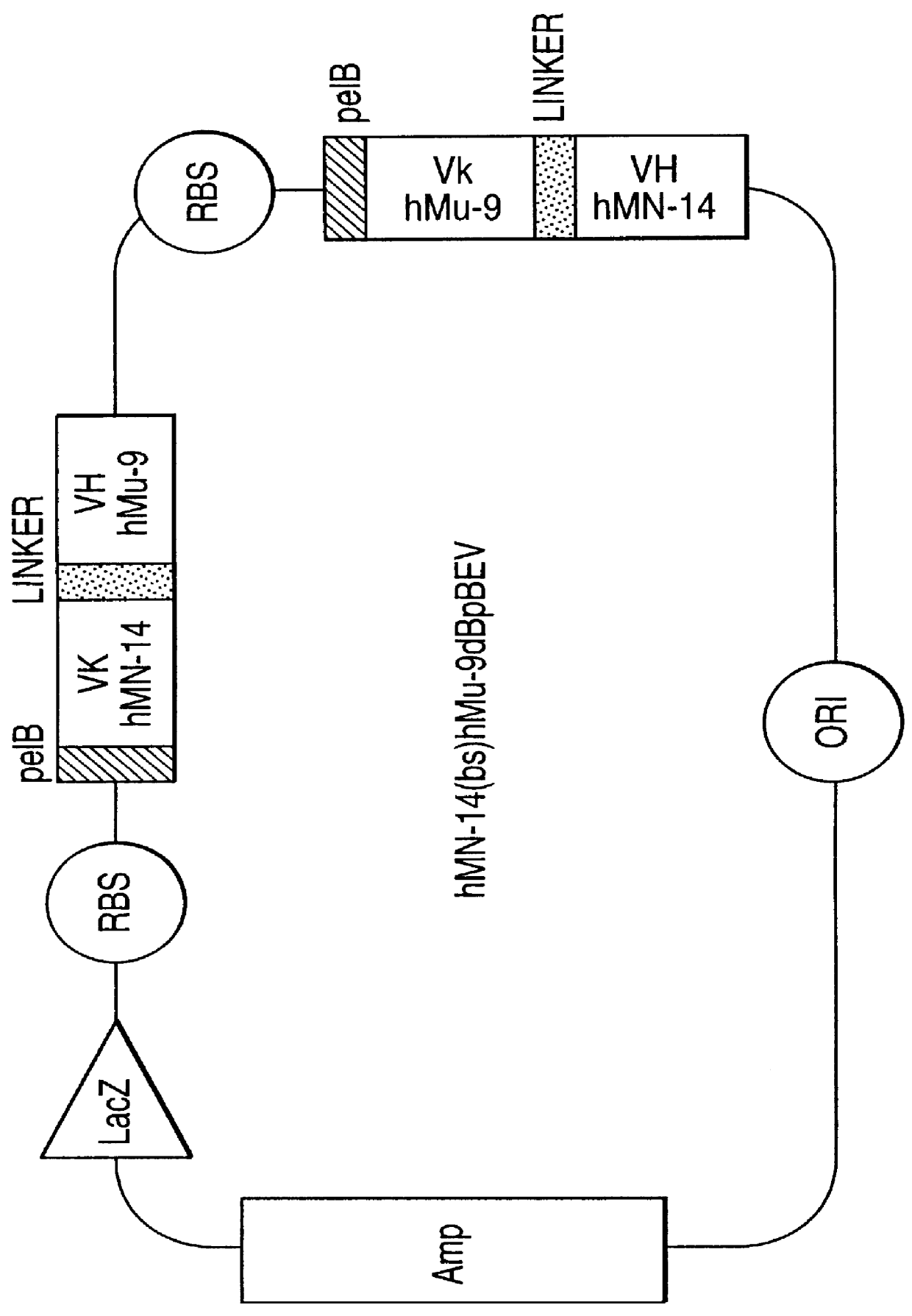
Combining Radioimmunotherapy and Antibody-Drug Conjugates for Improved Cancer Therapy
InactiveUS20110070156A1Organic active ingredientsHybrid immunoglobulinsParanasal Sinus CarcinomaAntiendomysial antibodies
Described herein are compositions and methods of use of radionuclide-antibody conjugates (for RAIT) and drug-antibody conjugates (ADC). The combination of RAIT and ADC was more efficacious than either RAIT alone, ADC alone, or the sum of effects of RAIT and ADC. The unexpected synergy resulted in decreased tumor growth rate and increased survival, with a high incidence of tumor-free survival in Capan-1 human pancreatic cancer xenografts in nude mice.
Owner:IMMUNOMEDICS INC
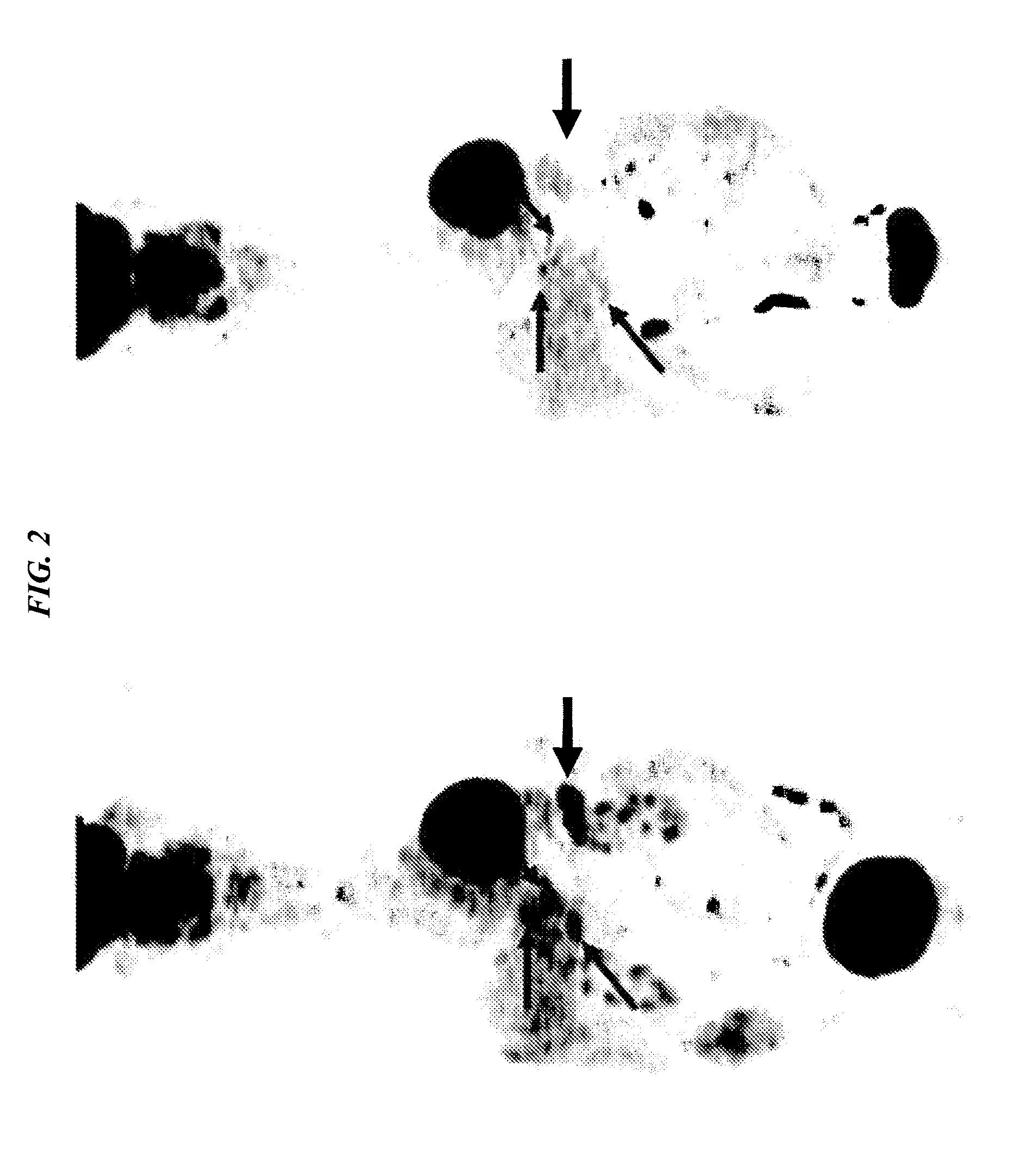
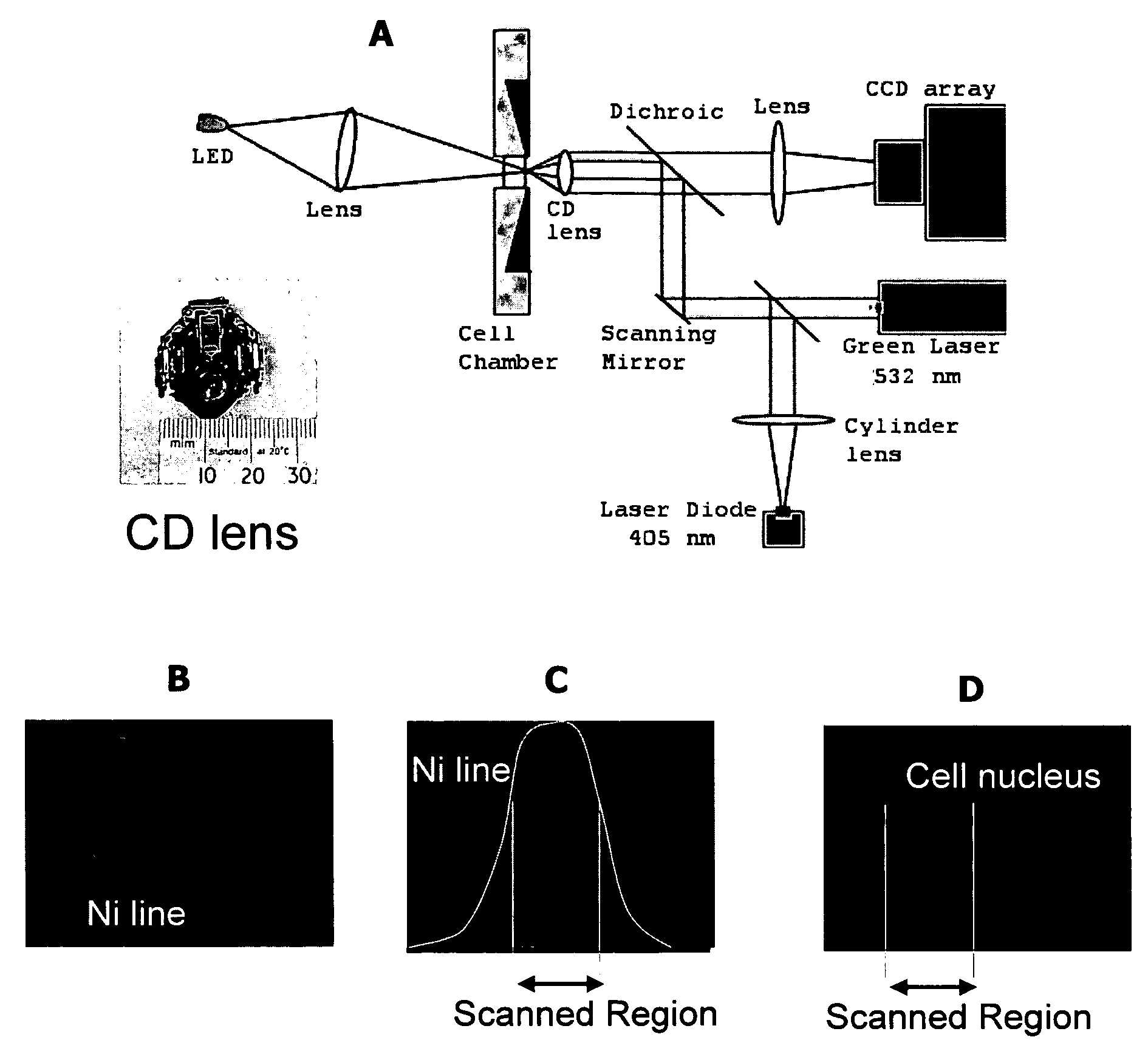
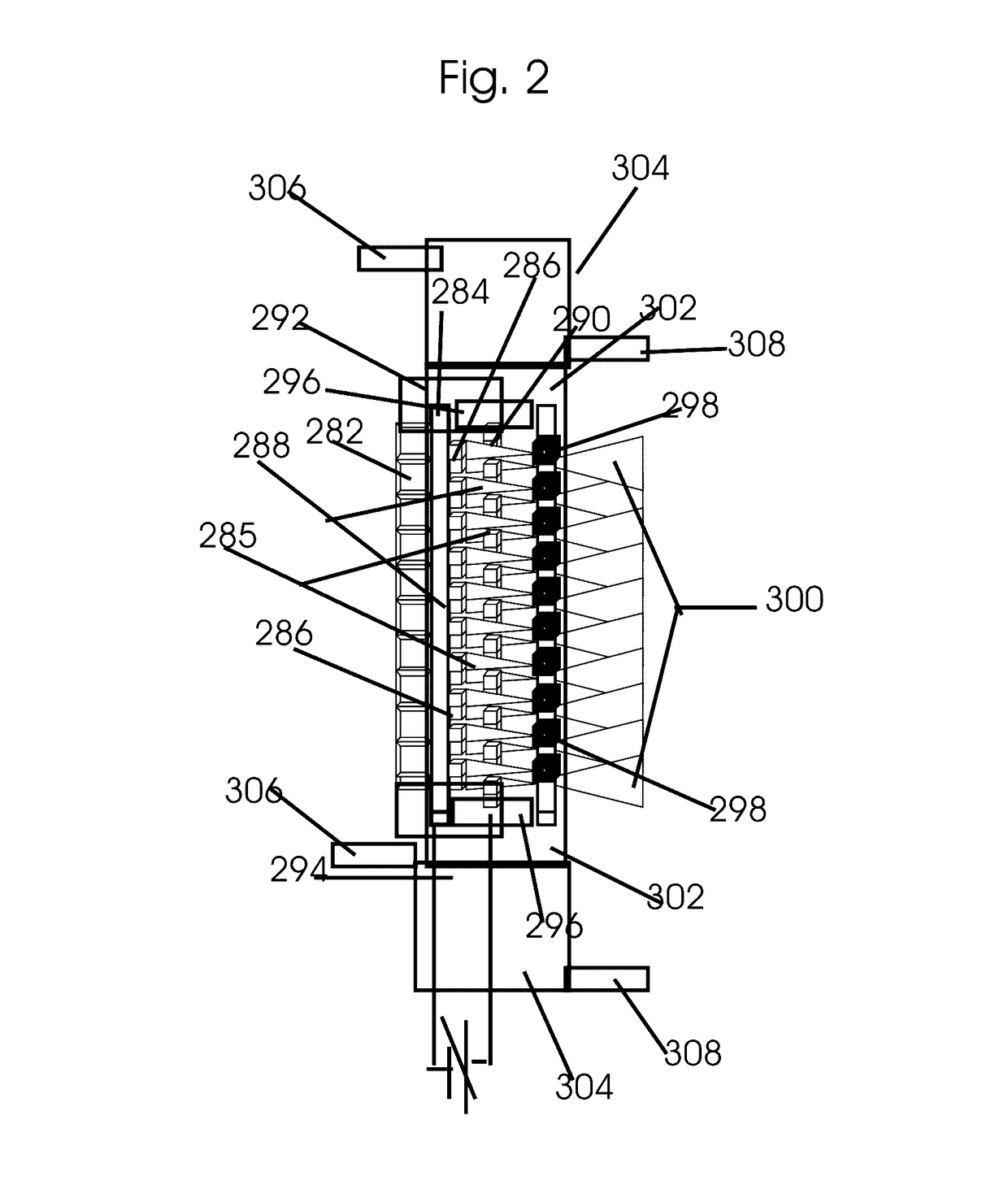
Device and method for analytical cell imaging
InactiveUS20060094109A1Bioreactor/fermenter combinationsBiological substance pretreatmentsBiopsyCirculating tumor cell
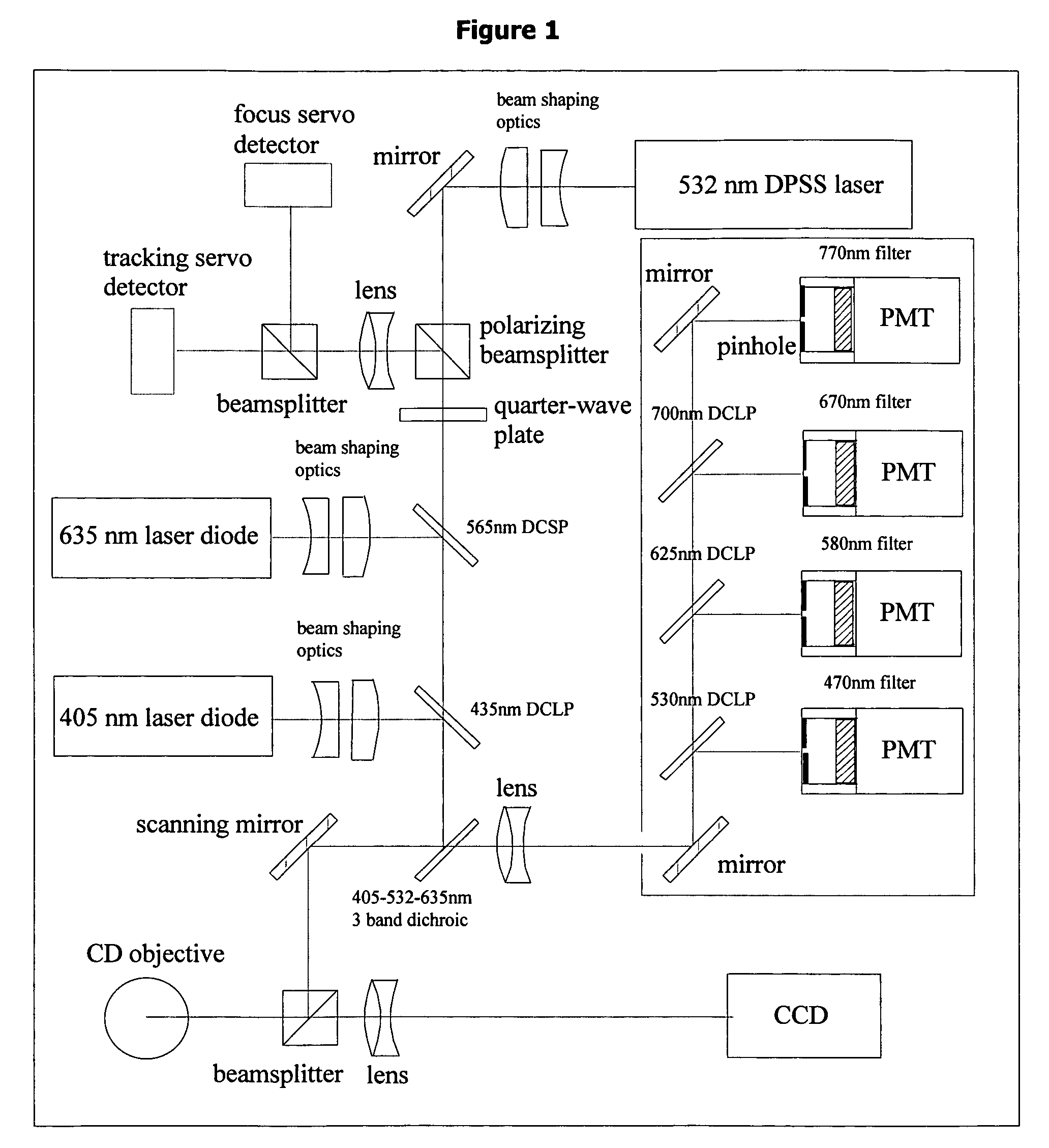
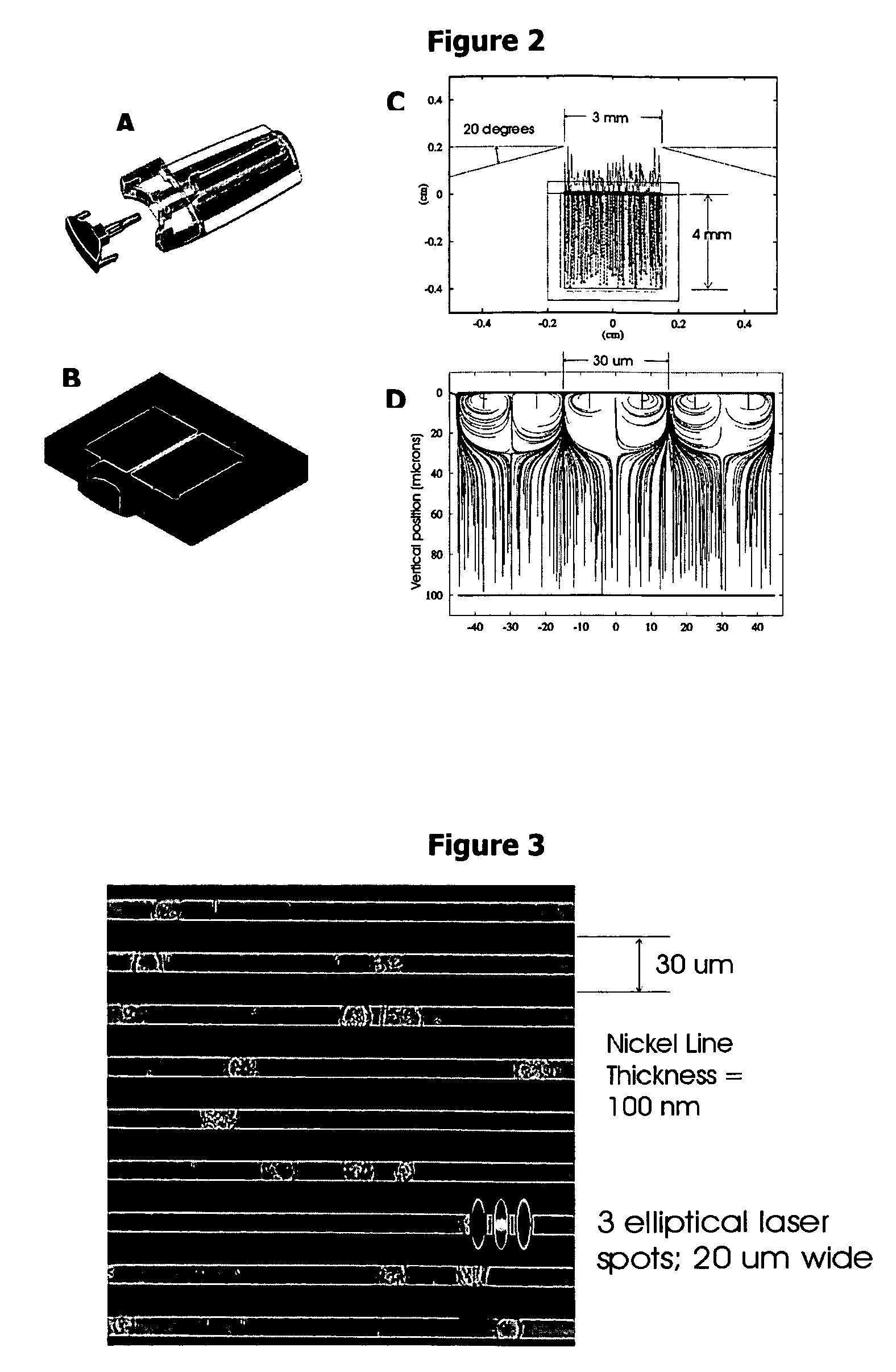
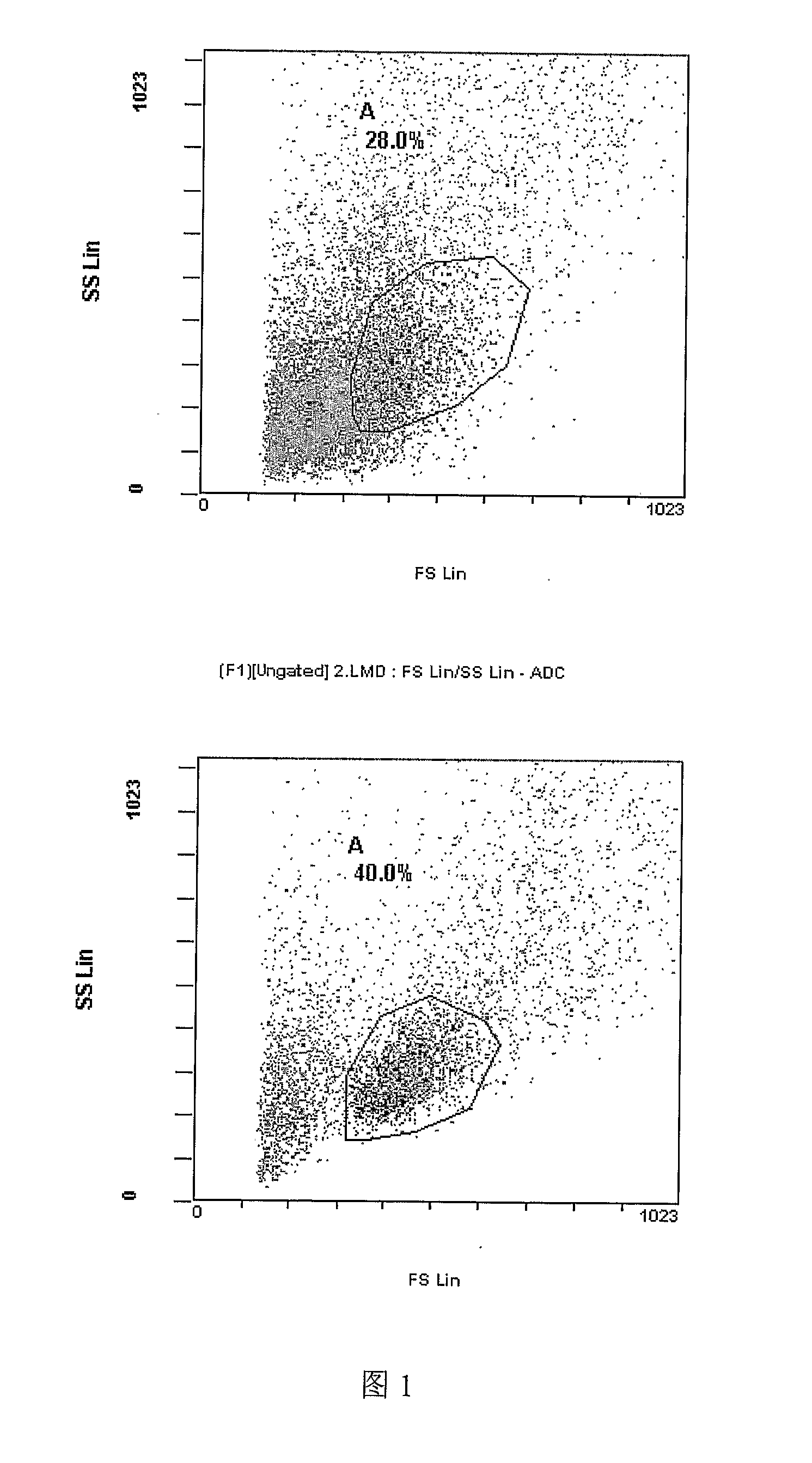
In patients with carcinomas tumor cells are shed into the blood, enumeration and characterization of these cells offers the opportunity to obtain a “real time” biopsy of the tumor and may improve the management of the disease. The frequency of circulating tumor cells is rare (<1 cell / ml) and technology is needed that has sufficient sensitivity and specificity to enumerate and characterize these cells. The present system was developed to provide an immunophenotype, fluorescence wave forms as well as images of immunomagnetically enriched cells. Blood volumes ranging from 7.5-30 ml are immunomagnetically enriched for epithelial cells. The sample volume is reduced to 320 μl and inserted into an analysis chamber. Upon introduction of the chamber in a magnetic field, the immunomagnetically tagged cells rise out of the sample and align between nickel lines (period 30 μm, space 15 μm) that are present on the viewing surface of the chamber. A multi laser system is used to detect the fluorescence emitted by DAPI, Phycoerythrin and Allophycocyan labeled and magnetically aligned cells. Compact disk optics are used to maintain alignment and focus of the laser beams onto nickel lines while moving the chamber. The chamber is scanned with a speed of 10 mm / sec and the entire chamber is analyzed in approximately 5 minutes. The fluorescent signals obtained from the events provide an immunophenotype similar to that of a flow cytometer. The fluorescence waveforms improve the characterization of the events and add to the classification as background, cellular debris and cells. Since the cell locations are preserved, objects that immunophenotypically classify as epithelial cells can be revisited for further analysis. Bright field and fluorescent images of the selected objects are captured to confirm that the identified objects are tumor cells.
Owner:IMMUNIVEST
(1H-Ind0l-7-Yl)-(Pyrimidin-2-Ylamino)Methanone Derivatives and Related Compounds as Igf-R1 Inhibitors for the Treatment of Cancer
InactiveUS20080194605A1Inhibiting, regulating and/or modulating signal transductionEfficiently employedOrganic active ingredientsBiocideDiseaseKinase
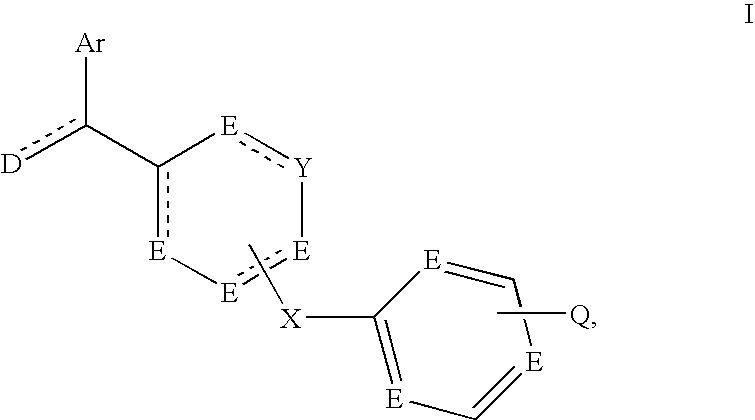
The invention relates to compounds of the formula (I), in which Ar denotes a mono- or bicyclic aromatic homo- or heterocycle having 1 to 4 N, O and / or S atoms and 5 to 10 skeleton atoms, which may be unsubstituted or mono-, di- or trisubstituted by carbonyl oxygen, Hal, A, OH, OA, NH2, NHA, NA2, NO2, CN, OCN, SCN, COOH, COOA, CONH2, CONHA, CONA2, NHCOA, NHCONH2, NHSO2A, CHO, COA, SO2NH2 and / or S(O)9A, A denotes unbranched, branched or cyclic alkyl having 1-14 C atoms, in which one or two CH2 groups may be replaced by O or S atoms and / or by —CR═CH— groups and / or in addition 1-7 H atoms may be replaced by F and / or Cl, Hal: F, Cl, Br or I, D: NH, NH2, NA2, NHA, CH2, CH3, OH, OA, O or S, E: CH2, CH, NH or N, Y: E or a saturated or unsaturated bond, X: CH2, O or NH, Q: Hal, A, OH, OA, NH2, NHA, NA2, NO2, CN, OCN, SCN, COOH, COOA, CONH2, CONHA, CONA2, NHCOA, NHCONH2, NHSO2A, CHO, COA, SO2NH2 or X-M, to the preparation and use thereof for the preparation of a medicament for the treatment of diseases, in particular tumours and / or diseases in the development or course of which kinases are involved.
Owner:MERCK PATENT GMBH
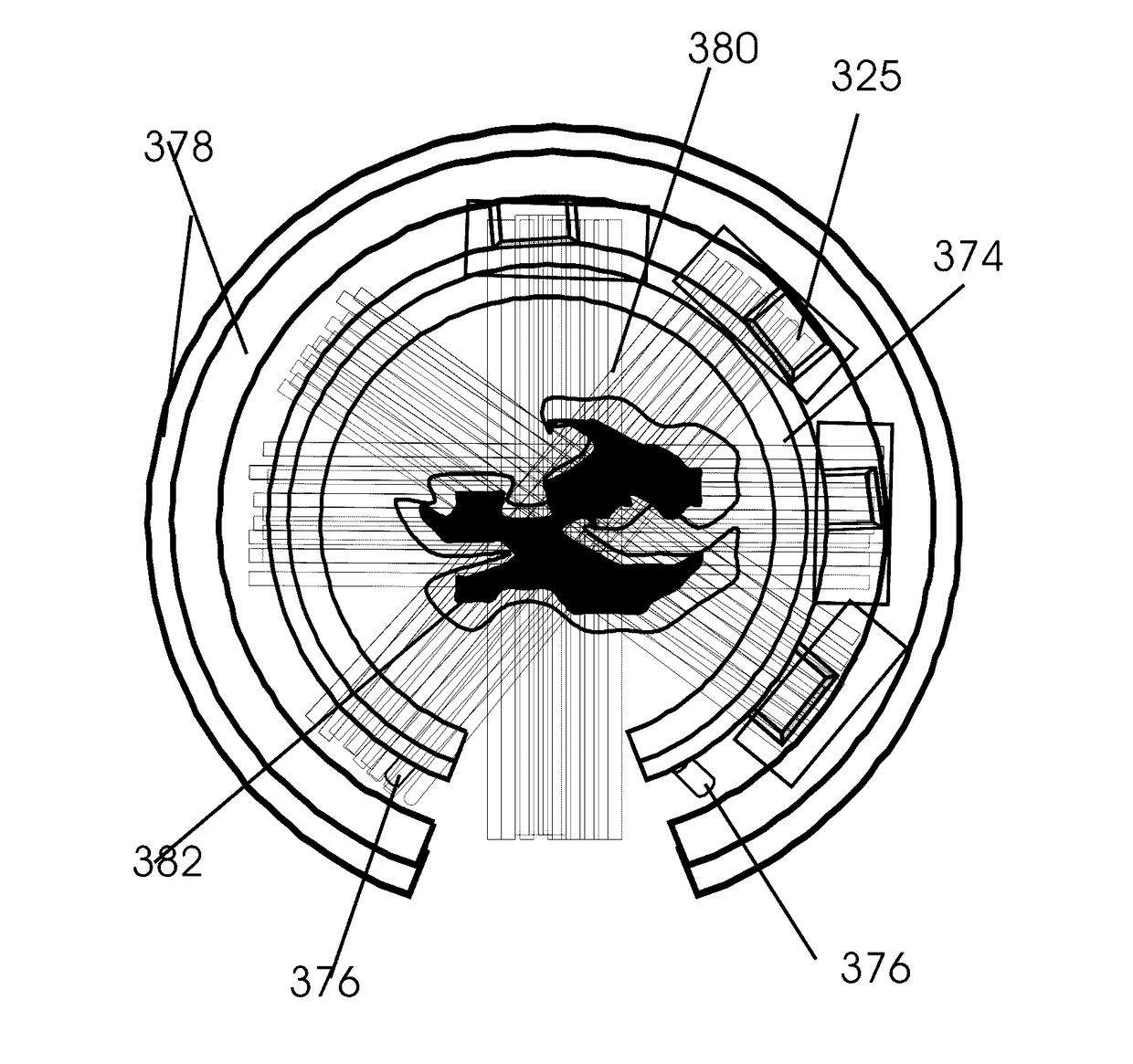
Method of image guided intraoperative simultaneous several ports microbeam radiation therapy with microfocus X-ray tubes
This invention pertains to a method of low-cost intraoperative all field simultaneous parallel microbeam single fraction few seconds duration 100 to 1,000 Gy and higher dose radiosurgery with micro-electro-mechanical systems (MEMS)-carbon nanotube based microaccelerators. It ablates cancer cells including the mesenchymal epithelial transformation associated cancer stem cells. Microbeam brachy-therapeutic radiosurgery is performed. Microaccelerators are configured for simultaneous parallel microbeam emission from varying angels to an isocentric tumor. Their additive dose rate at the isocentric tumor is in the range of 10,000 to 20,000 Gy / s. It eliminates most tumor recurrence and metastasis which enhances cancer cure rates. It also exposes cancer antigens which induces cancer immunity. Stereotactic breast core biopsy is combined with, positron emission tomography and computerized tomography and phase-contrast imaging. Parallel microbeam brachytherapy preserves normal breast appearance. Migration of normal stem cells from unirradiated valley regions heals the radiation damage to the normal tissue.
Owner:SAHADEVAN VELAYUDHAN
Methods and materials for detecting colorectal cancer and adenoma
InactiveUS20130012410A1High detectionHigh level of sensitivityMicrobiological testing/measurementLibrary screeningFOXE1Mammal
The present invention provides methods and materials related to the detection of colorectal neoplasm-specific markers (e.g., markers associated with colorectal cancer, markers associated with adenoma) in or associated with a subject's stool sample. In particular, the present invention provides methods and materials for identifying mammals (e.g., humans) having a colorectal neoplasm by detecting the presence and level of indicators of colorectal neoplasia such as, for example, long DNA (e.g., quantified by Alu PCR) and the presence and level of tumor-associated gene alterations (e.g., mutations in KRAS, APC, melanoma antigen gene, p53, BRAF, BAT26, PIK3CA) or epigenetic alterations (e.g., DNA methylation) (e.g., CpG methylation) (e.g., CpG methylation in coding or regulatory regions of bmp-3, bmp-4, SFRP2, vimentin, septin9, ALX4, EYA4, TFPI2, NDRG4, FOXE1) in DNA from a stool sample obtained from the mammal.
Owner:MAYO FOUND FOR MEDICAL EDUCATION & RES
Compositions and Methods for Tumor-Targeted Delivery of Effector Molecules
InactiveUS20070298012A1Reduce Toxicity RiskReduce riskHeavy metal active ingredientsBiocideParanasal Sinus CarcinomaMelanoma
The present application discloses the preparation and use of attenuated tumor-targeted bacteria vectors for the delivery of one or more primary effector molecule(s) to the site of a solid tumor. The primary effector molecule(s) of the invention is used in the methods of the invention to treat a solid tumor cancer such as a carcinoma, melanoma, lymphoma, or sarcoma. The invention relates to the surprising discovery that effector molecules, which may be toxic when administered systemically to a host, can be delivered locally to tumors by attenuated tumor-targeted bacteria with reduced toxicity to the host. The application also discloses to the delivery of one or more optional effector molecule(s) (termed secondary effector molecules) which may be delivered by the attenuated tumor-targeted bacteria in conjunction with the primary effector molecule(s).
Owner:NANOTHERAPEUTICS INC
Muir-Torre-like syndrome in Fhit deficient mice
InactiveUS20060075511A1Drug screeningNucleic acid vectorParanasal Sinus CarcinomaGastrointestinal cancer
The invention provides nonhuman transgenic animals with a disrupted FHIT gene. The invention further provides transgenic mice in which one or both Fhit alleles have been inactivated. Preferably, the Fhit-deficient mice develop multiple tumors of both visceral and sebaceous origin, similar to those of Muir-Torre familial cancer syndrome. The present invention further relates to the generation of these transgenic mice and their use as model systems to study the effects of carcinogenic agents in promoting clonal expansion of neoplastic cells in cancers, preferably gastrointestinal cancers of which Muir-Torre syndrome is a subset. The invention further relates to testing therapeutic agents for their efficacy in the prevention and treatment of cancer, preferably gastrointestinal cancer.
Owner:CROCE CARLO +1
Non-invasive screening of skin diseases by visible/near-infrared spectroscopy
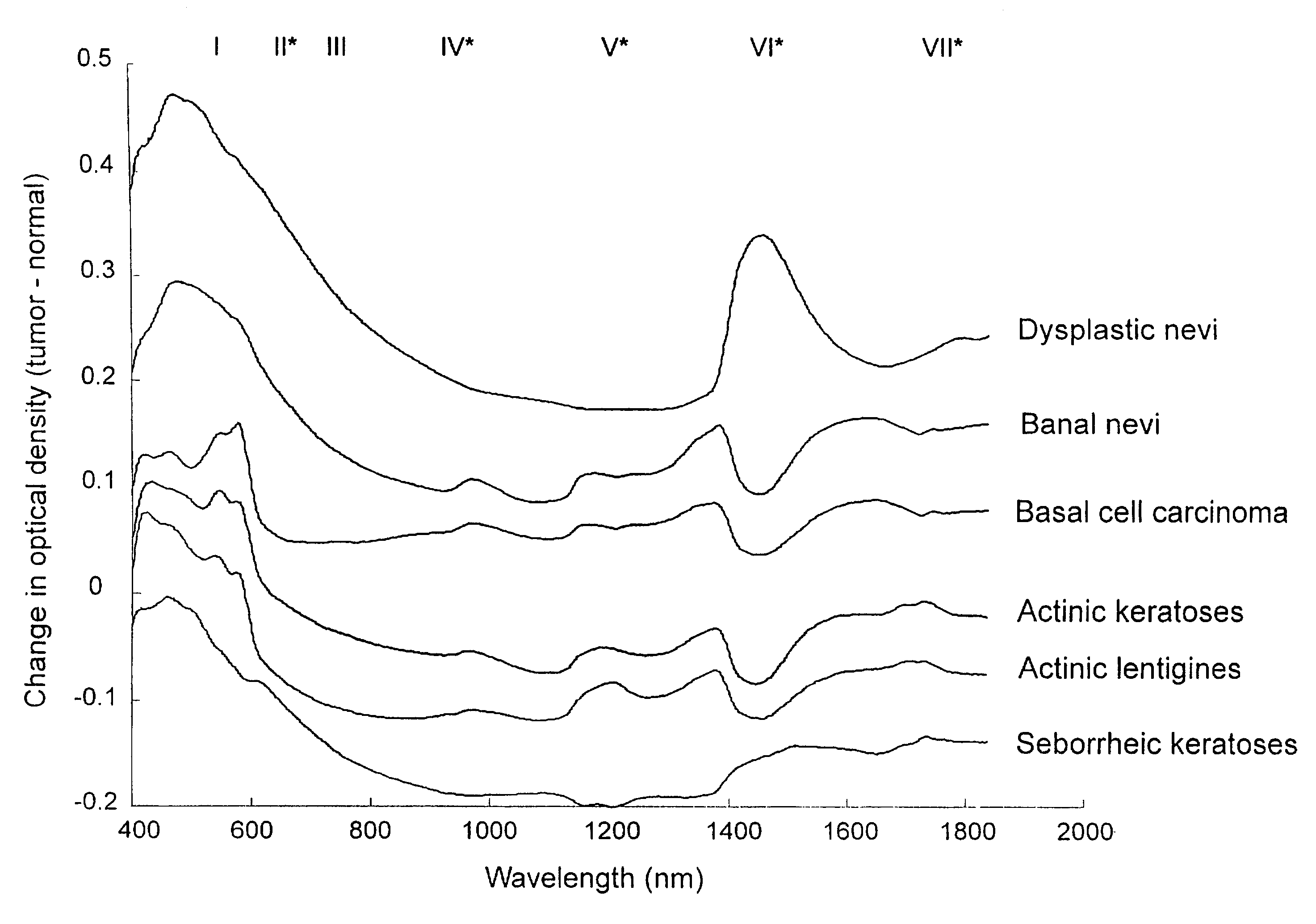
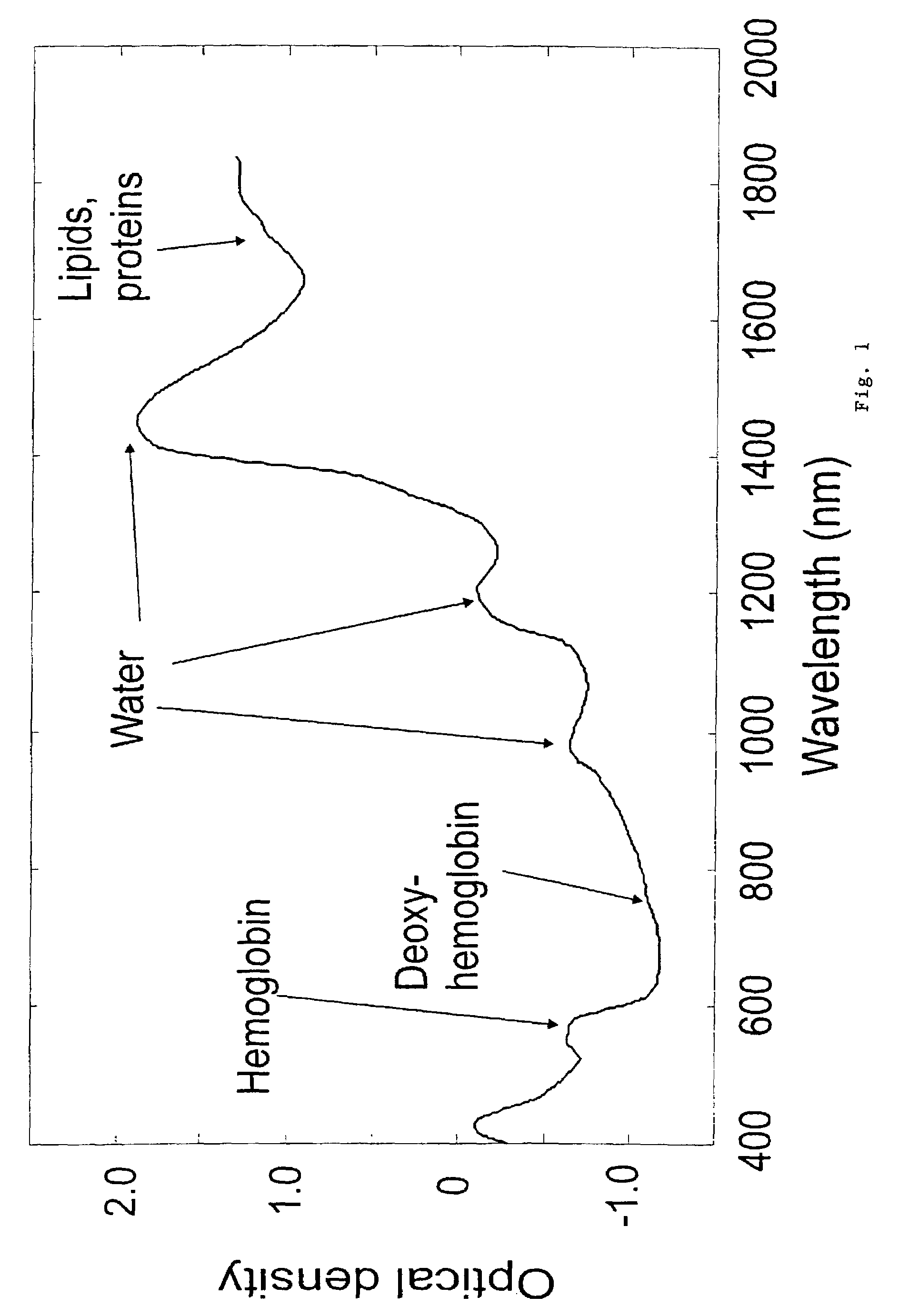
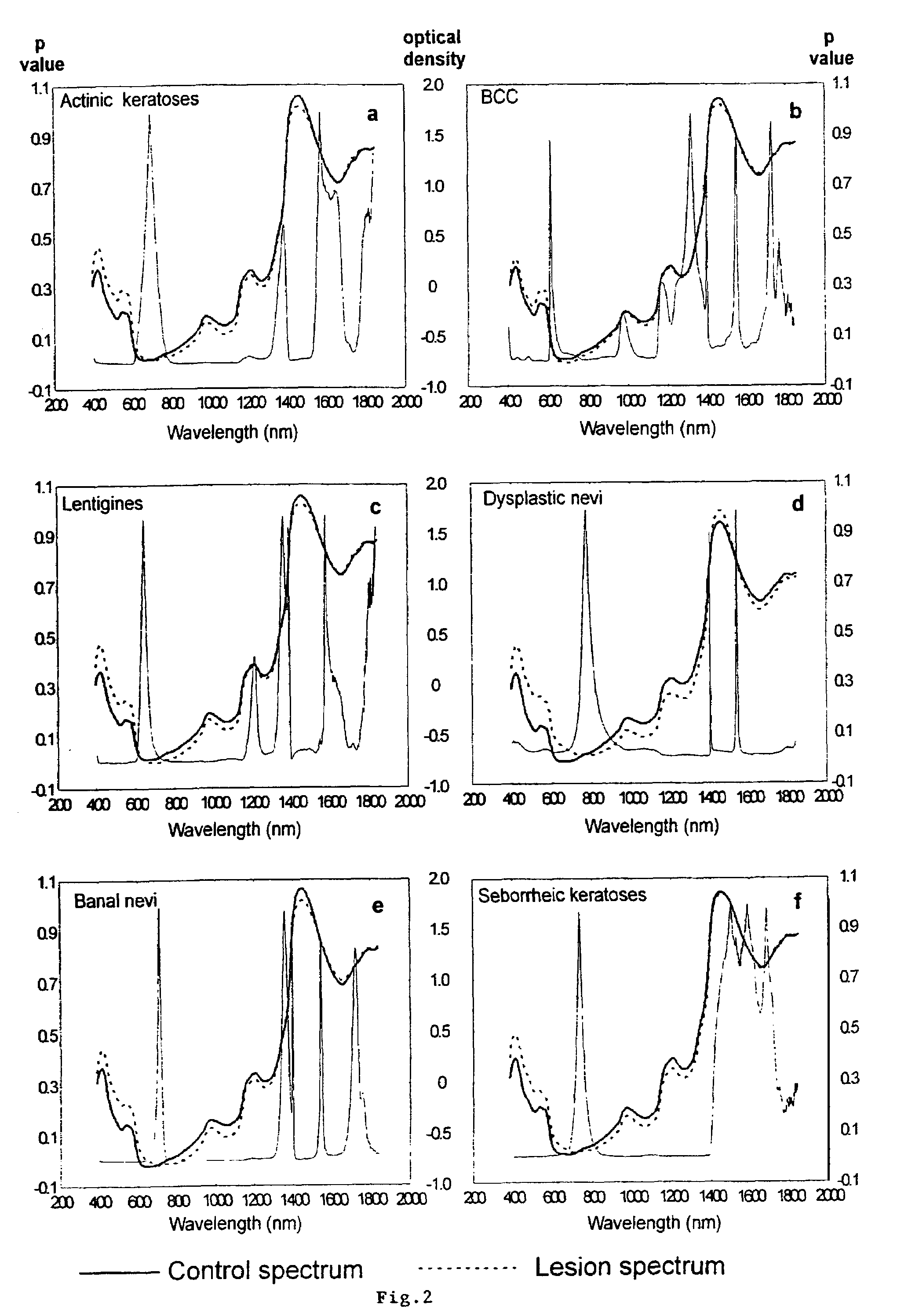
A non-invasive tool for skin disease diagnosis would be a useful clinical adjunct. The purpose of this study was to determine whether visible / near-infrared spectroscopy can be used to non-invasively characterize skin diseases. In-vivo visible- and near-infrared spectra (400-2500 nm) of skin neoplasms (actinic keratoses, basal cell carcinomata, banal common acquired melanocytic nevi, dysplastic melanocytic nevi, actinic lentigines and seborrheic keratoses) were collected by placing a fiber optic probe on the skin. Paired t-tests, repeated measures analysis of variance and linear discriminant analysis were used to determine whether significant spectral differences existed and whether spectra could be classified according to lesion type. Paired t-tests showed significant differences (p<0.05) between normal skin and skin lesions in several areas of the visible / near-infrared spectrum. In addition, significant differences were found between the lesion groups by analysis of variance. Linear discriminant analysis classified spectra from benign lesions compared to pre-malignant or malignant lesions with high accuracy. Visible / near-infrared spectroscopy is a promising non-invasive technique for the screening of skin diseases.
Owner:NAT RES COUNCIL OF CANADA
Diagnostic tests for predicting prognosis, recurrence, resistance or sensitivity to therapy and metastatic status in cancer
InactiveUS20140342946A1Low costReduce morbidityMicrobiological testing/measurementProtein nucleotide librariesParanasal Sinus CarcinomaDiagnostic test
The present invention describes a method utilizing a set of genes or gene products whose altered expression in cancer tissue, particularly head and neck cancer and other carcinomas, or its adjacent normal tissues predicts (a) probability of recurrence in time after treatment (b) sensitivity or resistance to therapies or (c) probability of metastasis at the time of initial discovery of the tumor. Furthermore, the invention describes methods of determining the molecular signature in tumor tissues, tissues adjacent to the tumor, or in saliva by using DNA microarray techniques, quantitative real-time PCR, immunohistochemistry or other methods that are used for determining gene or gene product expression levels.
Owner:KURIAKOSE MONI ABRAHAM +1
Treating or preventing the early stages of degeneration of articular cartilage or subchondral bone in mammals using carprofen and derivatives
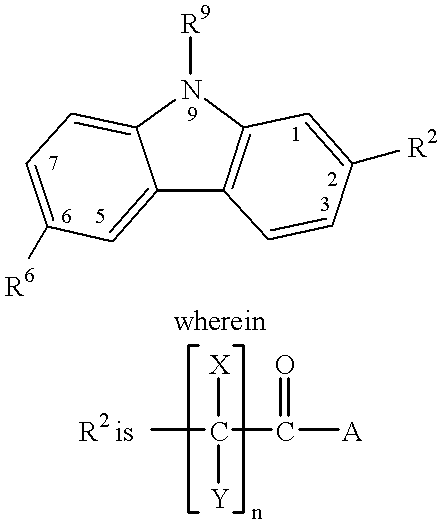
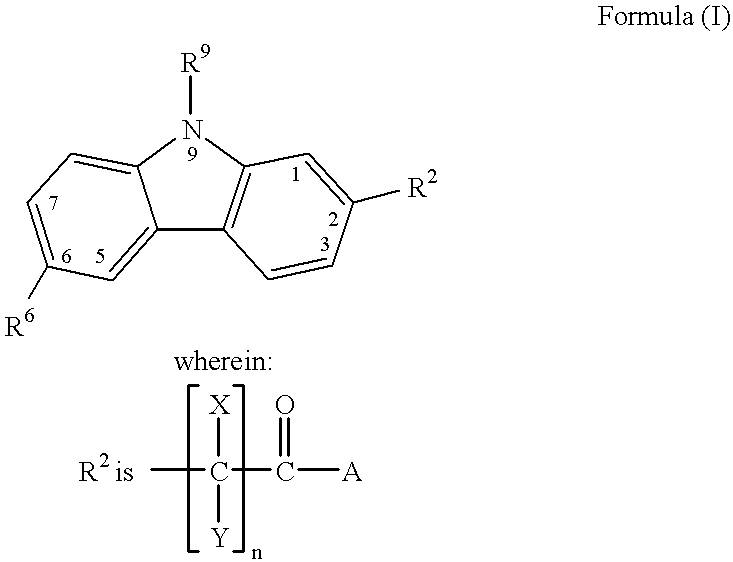
Treating or preventing the early stages of degeneration of articular cartilage or subchondral bone in the affected joint of a mammal is accomplished by administering a chondroprotective compound of Formula (I):where A is hydroxy, (C1-C4)alkoxy, amino, hydroxy-amino, mono-(C1-C2)alkylamino, di-(C1-C2)alkylamino; X and Y are independently H or (C1-C2)alkyl; and n is 1 or 2; R6 is halogen, (C1-C3)alkyl, trifluoromethyl, or nitro; R9 is H; (C1-C2)alkyl; phenyl or phenyl-(C1-C2)alkyl, where phenyl is optionally mono-substituted by fluoro or chloro; -C(=O)-R, where R is (C1-C2)alkyl or phenyl, optionally mono-substituted by fluoro or chloro; or -C(=O)-O-R', where R1 is (C1-C2)alkyl.This treatment ameliorates, diminishes, actively treats, reverses or prevents any injury, damage or loss of articular cartilage or subchondral bone subsequent to said early stage of said degeneration. Whether or not a mammal needs such treatment is determined by whether or not it exhibits a statistically significant deviation from normal standard values in synovial fluid or membrane from the affected joint, with respect to at least five of the following substances: increased interleukin-1 beta (IL-1beta); increased tumor necrosis factor alpha (TNFalpha); increased ratio of IL-1beta to IL-1 receptor antagonist protein (IRAP); increased expression of p55 TNF receptors (p55 TNF-R); increased interleukin-6 (IL-6); increased leukemia inhibitory factor (LIF); decreased insulin-like growth factor-1 (IGF-1); decreased transforming growth factor beta (TGFbeta); decreased platelet-derived growth factor (PDGF); decreased basic fibroblast growth factor (b-FGF); increased keratan sulfate; increased stromelysin; increased ratio of stromelysin to tissue inhibitor of metalloproteases (TIMP); increased osteocalcin; increased alkaline phosphatase; increased cAMP responsive to hormone challenge; increased urokinase plasminogen activator (uPA); increased cartilage oligomeric matrix protein; and increased collagenase.
Owner:PFIZER INC +1
Compositions and methods for the treatment of cancer
InactiveUS20040087531A1Growth inhibitionOrganic active ingredientsGenetic material ingredientsParanasal Sinus CarcinomaOncology
Owner:TELERMAN ADAM +3
American cockroaches effective fraction for anti-tumor prepared by macroporous adsorption resin and use
ActiveCN101214262AAnthropod material medical ingredientsPharmaceutical delivery mechanismSolventChemistry
The present invention relates to a periplaneta americana extract effective part with cytotoxic activity, a preparation method and a medical purpose thereof. The periplaneta americana extract effective part of the present invention is refined from fresh or dry blattidae periplaneta americana body which is extracted by alcohol water, processed for macroporous resin column chromatography and is eluted by alcohol solvent. The periplaneta americana extract effective part obtained by the present invention has obvious cytotoxicity and internal anticancer efficacy towards oral epidermoid carcinoma (KB) cell, poorly differentiated gastric abenocarcinoma tumour (BGC-823) cell, human chronic myelogonium leukaemia cancer (K562) cell and human myelogonium leukaemia cancer cell (HL-60) and can be used for preparing for a drug and a health care product for remedying the tumours.
Owner:KUNMING SINOWAY NATURAL PHARMA
Methods for determining the prognosis for cancer patients using TUCAN
InactiveUS20070178502A1Improve survivalPeptide/protein ingredientsAntibody mimetics/scaffoldsOncologyNeoplastic cell
The invention provides methods for determining a prognosis for survival for a cancer patient. One method involves (a) measuring a level of a TUCAN in a neoplastic cell-containing sample from the cancer patient, and (b) comparing the level of TUCAN in the sample to a reference level of TUCAN, wherein a low level of TUCAN in the sample correlates with increased survival of the patient. Another method involves (a) measuring a level of TUCAN in a neoplastic cell-containing sample from the cancer patient, and (b) classifying the patient as belonging to either a first or second group of patients, wherein the first group of patients having low levels of TUCAN is classified as having an increased likelihood of survival compared to the second group of patients having high levels of TUCAN.
Owner:THE BURNHAM INST
Cancer therapy
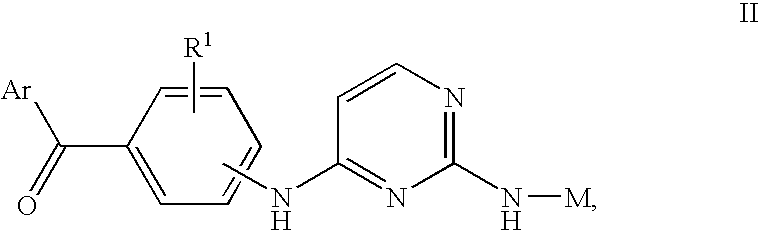
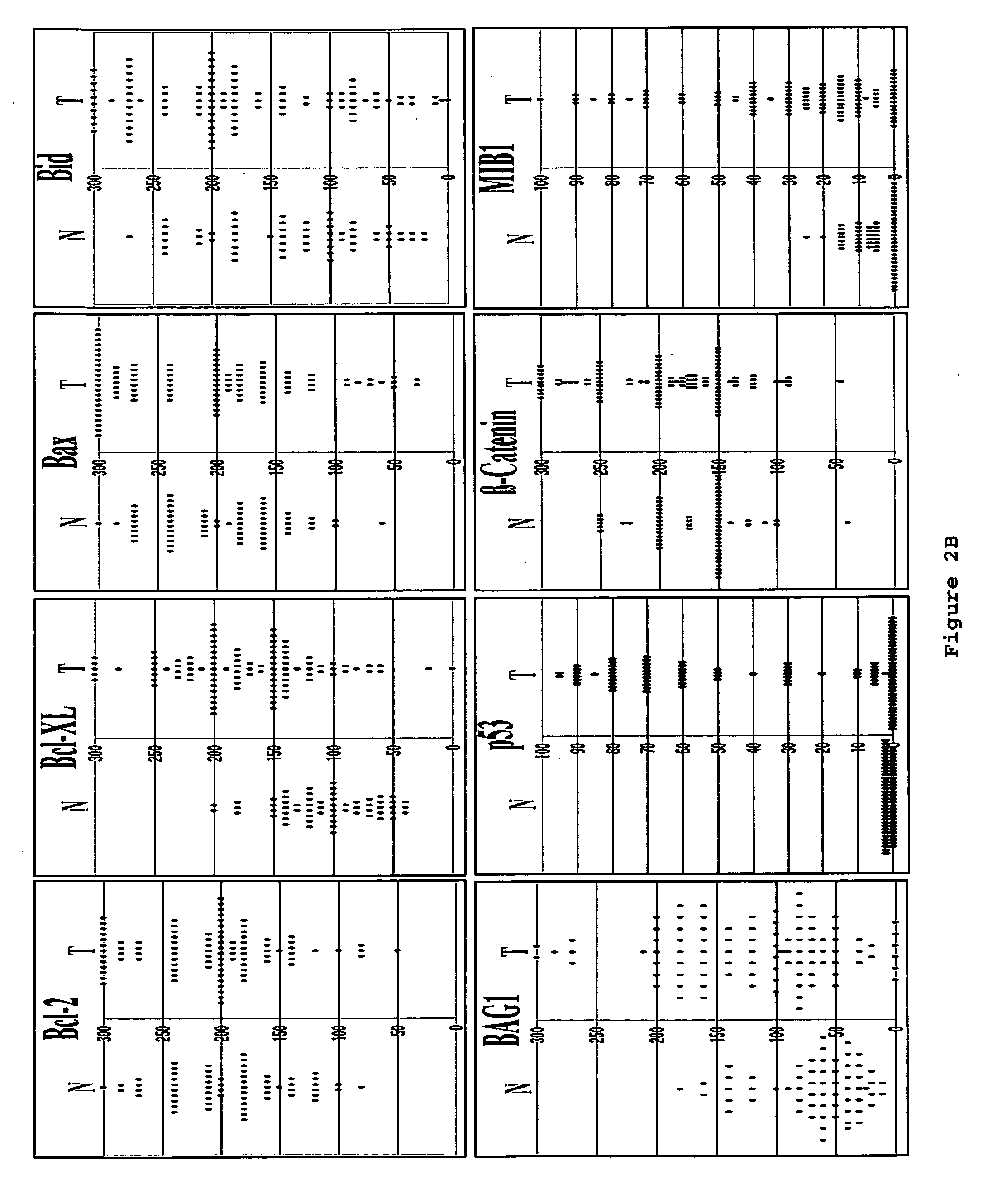
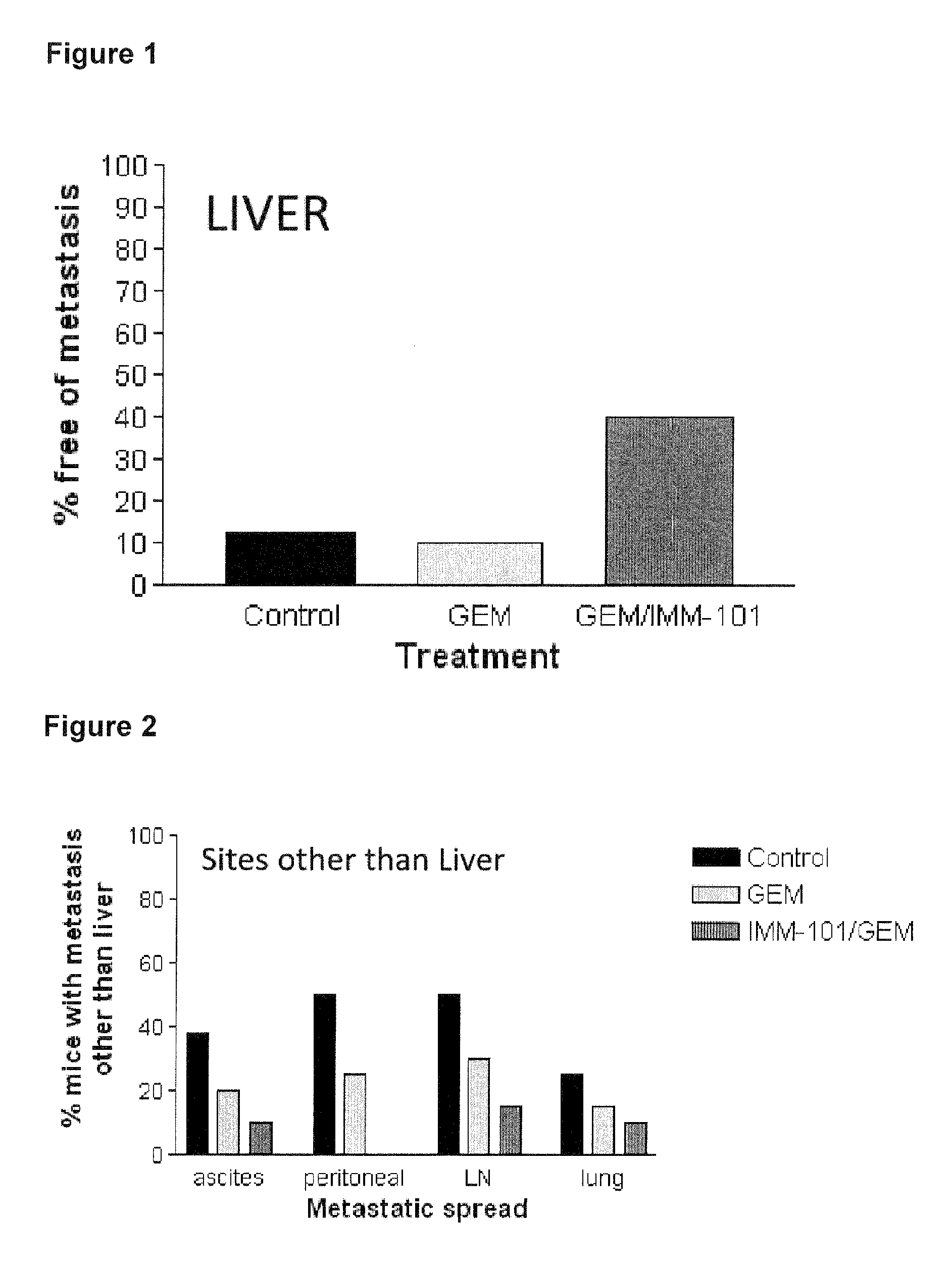
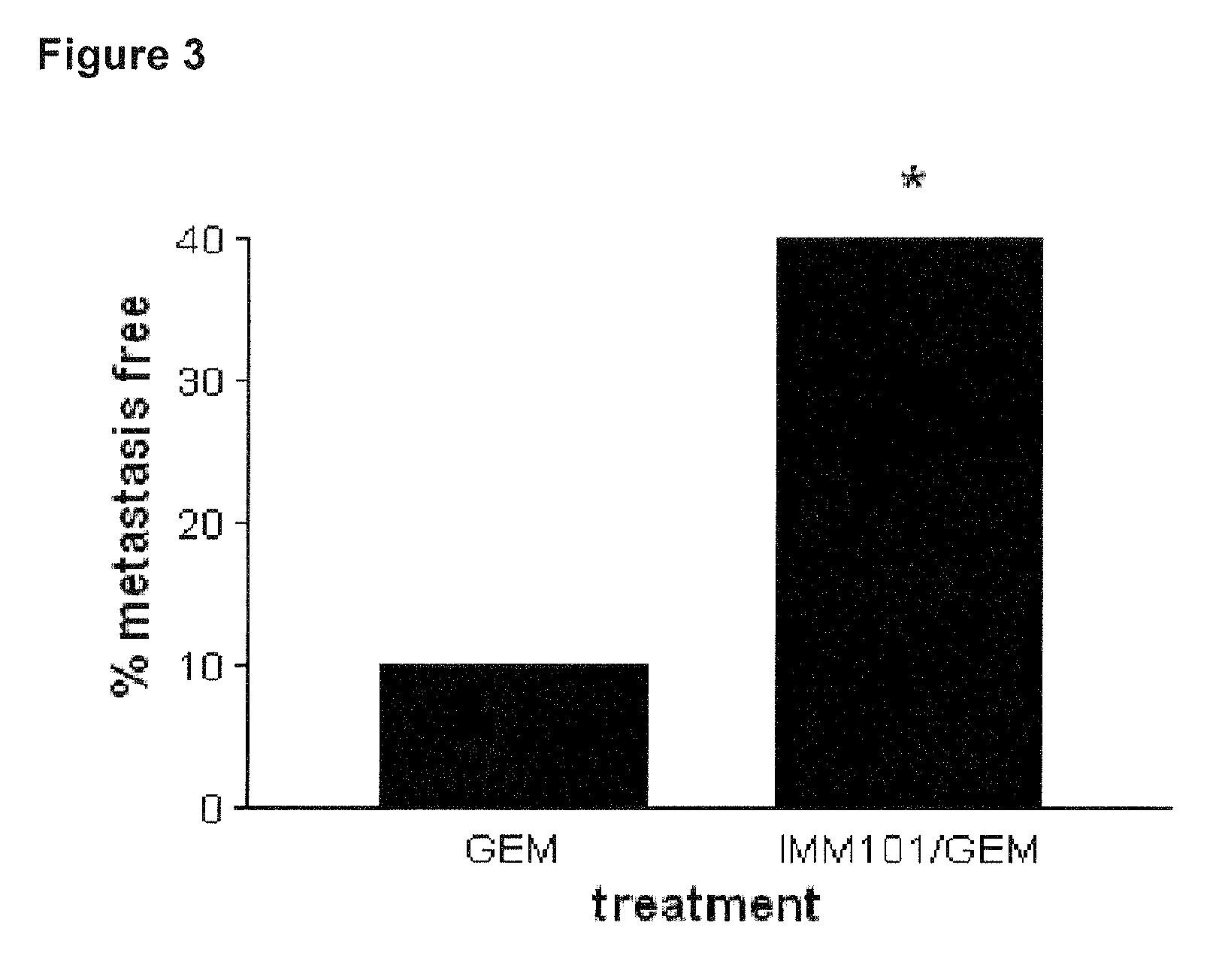
ActiveUS20130209517A1Great therapeutic responseBalanced immunoregulatoryBiocideBacterial antigen ingredientsParanasal Sinus CarcinomaPyrimidine analogue
The present invention relates to a method of preventing, treating or inhibiting the development of tumors or metastases in a subject and to an immunomodulator for use in such therapy, in combination with a chemotherapeutic agent. An aspect the present invention is a method of preventing, treating, reducing, inhibiting and / or controlling the formation or establishment of metastasis of a primary neoplasia, tumor or cancer at one or more sites distinct from a primary neoplasia, tumor or cancer, in a subject intended to undergo chemotherapy, wherein the method comprises administering to the subject, a therapeutically effective amount of an antimetabolite pyrimidine analogue and an immunomodulator.
Owner:IMMODULON THERAPEUTICS
Methods for culturing and identifying nasopharyngeal carcinoma organs
ActiveCN109679915AImprove consistencyEfficient formationMicrobiological testing/measurementCell culture active agentsCancer cellNasopharyngeal carcinoma
The application relates to a culture medium for nasopharyngeal carcinoma organs, and methods for culturing and identifying the nasopharyngeal carcinoma organs. According to the culture and growth characteristics of nasopharyngeal carcinoma-derived cells, a variety of cytokine components are selected and blended according to a certain ratio; the contents of cytokines and signal pathway regulatory factors in the culture medium are suitable; the nasopharyngeal carcinoma cells can effectively form organoids in a three-dimensional (3D) environment. The methods for culturing and identifying the nasopharyngeal carcinoma organs as well as the unique culture medium for the nasopharyngeal carcinoma organs can be used for successfully and stably culturing the nasopharyngeal carcinoma organs which have pathophysiological features and genotypes being highly consistent with those of the parental tumor and have high similar form of organization.
Owner:NANFANG HOSPITAL OF SOUTHERN MEDICAL UNIV
Diagnostic system for the detection of skin cancer
ActiveUS20090220415A1Improves transdermal penetrationQuick analysisUltrasonic/sonic/infrasonic diagnosticsCompounds screening/testingFluoProbesParanasal Sinus Carcinoma
Methods and compositions are provided for the analysis of skin surfaces to determine the presence of neoplastic tissue. In the methods of the invention, a composition comprising a florescent probe that binds to a specific neoplasia associated marker is applied topically to the area of interest. After topical administration, the probe preferentially binds to markers associated in neoplastic lesions in situ, which binding is detected with a compact illumination unit that provides illumination at a wavelength appropriate for image acquisition. The illumination unit comprises a light source and fiber optic bundle to direct the light towards the area of examination. A detection unit is used to capture and record an image of the area of investigation. The detection unit may be a digital camera, film camera, etc. A mapping module may also be provided to catalogue the site of examination.
Owner:ORLUCENT INC
Method for detection and diagnosis of lung and pancreatic cancers from imaging scans
ActiveUS20200160997A1Improve performanceLarge gainImage enhancementImage analysisPulmonary noduleParanasal Sinus Carcinoma
A method of detecting and diagnosing cancers characterized by the presence of at least one nodule / neoplasm from an imaging scan is presented. To detect nodules in an imaging scan, a 3D CNN using a single feed forward pass of a single network is used. After detection, risk stratification is performed using a supervised or an unsupervised deep learning method to assist in characterizing the detected nodule / neoplasm as benign or malignant. The supervised learning method relies on a 3D CNN used with transfer learning and a graph regularized sparse MTL to determine malignancy. The unsupervised learning method uses clustering to generate labels after which label proportions are used with a novel algorithm to classify malignancy. The method assists radiologists in improving detection rates of lung nodules to facilitate early detection and minimizing errors in diagnosis.
Owner:UNIV OF CENT FLORIDA RES FOUND INC
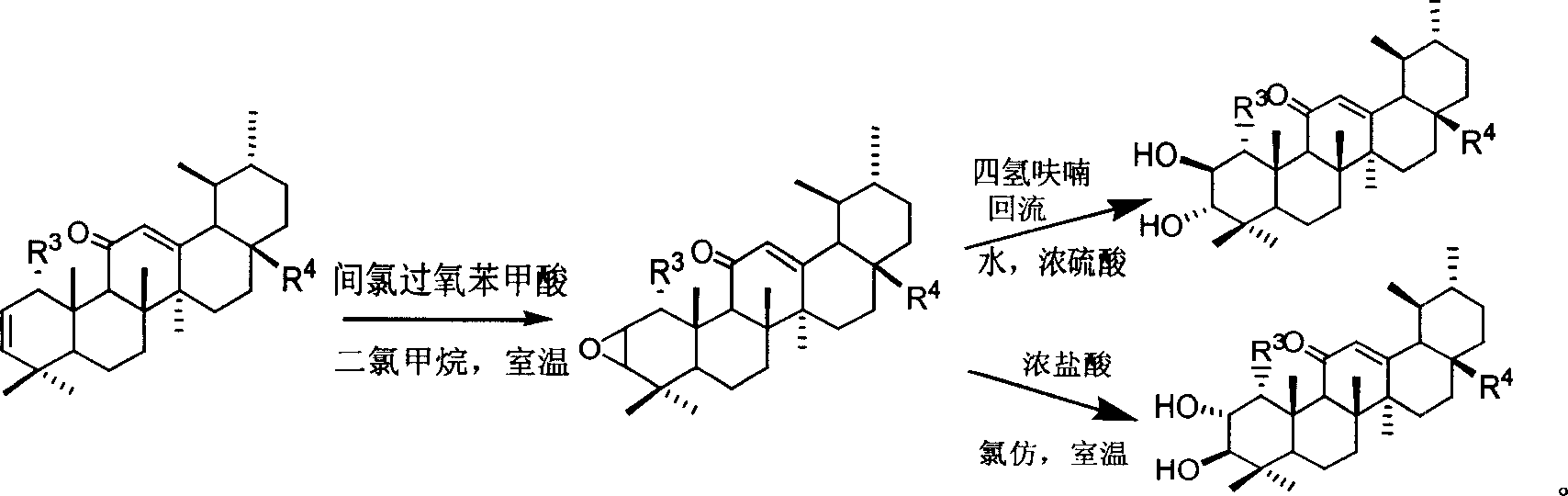
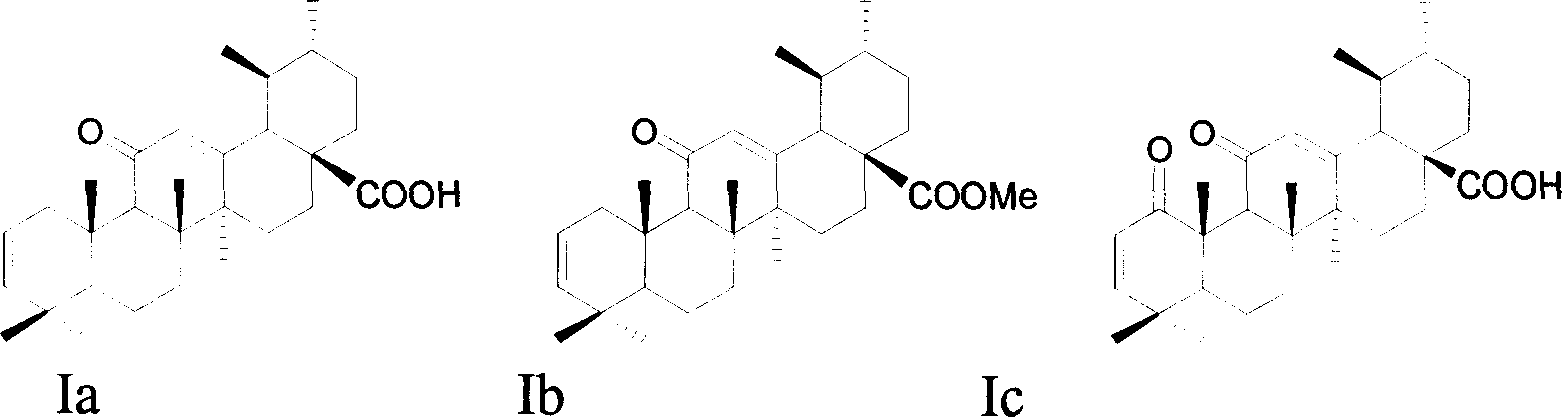
A macrocyclic oxidation substituted pentacyclic triterpanoids derivative and preparation method and use thereof
InactiveCN101117349AStrong inhibitory activityOrganic active ingredientsMetabolism disorderAlgluceraseAnti-Tumor Drugs
The present invention relates to a pentacyclic triterpanoid derivative of multiple-oxide substitution of the A ring and the medicine salt or solvate of the derivative, and the present invention also relates to the preparation method, the drug combination, and medical use of the derivative. The compound of the present invention has the functions of inhibiting the activity of six human tumor cell strains in vitro, such as human prostate cancer cell (PC-3), nasopharyngeal carcinoma cells (CNE), oral squamous carcinoma cell(KB), human lung cancer cell (A549), human hepatoma cell (BEL-7404), and human cervix cancer cell (Hela), and the function of the invention is at the same magnitude of the positive control of cisplatin, thereby the compound can be used as expected antitumor drug. The compound of the present invention also inhibits the alpha glucosidase strongly, and the inhibiting effect is greater than the positive control of acarbose, thereby the compound can be used as expected medicine for preventing and treating diabetes and the treatment of the virus diseases.
Owner:ZHEJIANG HISUN PHARMA CO LTD
Intraoperative, intravascular and endoscopic tumor and lesion detection, biopsy and therapy
InactiveUS20010006618A1DiscriminationImprove discriminationUltrasonic/sonic/infrasonic diagnosticsSurgeryNon malignantAntibody fragments
Methods are provided for close-range intraoperative, endoscopic and intravascular detection and treatment of lesions, including tumors and non-malignant lesions. The methods use antibody fragments or subfragments labeled with isotopic and non-isotopic agents. Also provided are methods for detection and treatment of lesions with photodynamic agents and methods of treating lesions with a protein conjugated to an agent capable of being activated to emit Auger electron or other ionizing radiation. Compositions and kits useful in the above methods are also provided.
Owner:IMMUNOMEDICS INC
Tumor correlated albumen, coding gene and application thereof
InactiveCN101054413AInhibit malignant transformationPeptide/protein ingredientsGenetic material ingredientsAntineoplastic drugGene expression
The present invention discloses a tumour-related protein and its coding gene. The tumour-related protein is as follows: (a) a protein composed of sequence 4 amino acid residue in sequence list; (b) a protein derived from (a) by substituting and / or deleting and / or adding one or several amino acid residue of sequence 4 amino acid residue in sequence list The recombinant mammal gene expression vector containing tumour-related protein coding gene can be used as antineoplastic drug for preventing and / or treating lung cancer, esophageal carcinoma or cancer of larynx.
Owner:CANCER INST & HOSPITAL CHINESE ACADEMY OF MEDICAL SCI +1
LNA oligonucleotides and the treatment of cancer
InactiveUS20060154888A1Organic active ingredientsPeptide/protein ingredientsCancer cell linesApoptosis induction
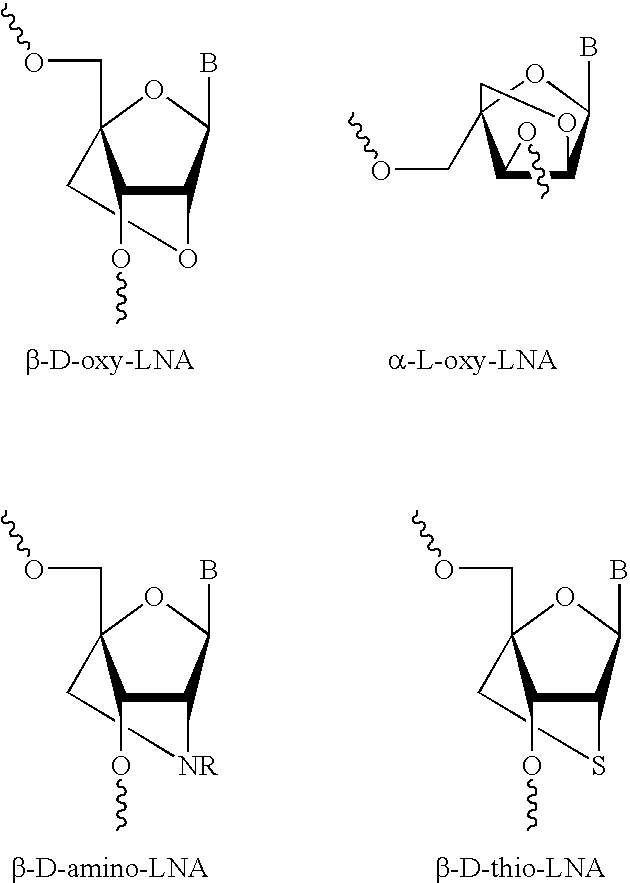
The present disclosure concerns LNA oligonucleotides having a (sub)sequence of the general formula 5′-(MeCx)(Tx)MeCxAsAstscscsastsgsgsMeCxAx(Gx)(c)-3′, and preferably of the general formula 5′-MeCxTxMeCxAsastscscsastsgsgsMeCxAxGxc-3′, wherein capital letters designate an LNA nucleotide analogue selected from β-D-oxy-LNA, β-D-thio-LNA, β-D-amino-LNA and α-L-oxy-LNA, small letters designate a deoxynucleotide, and underline designates either an LNA nucleotide analogue as defined above or a deoxynucleotide. Such LNA oligonucleotides exhibit surprisingly good properties with respect to inhibition of the expression of Survivin by means of an anti-sense mechanism, and thereby lead to reduction or inhibition of tumour development in vivo. The LNA oligonucleotides are superior to other LNA oligonucletides targeting Surviving mRNA measured by functional read outs such as apoptosis induction and proliferation inhibition, and is potent in down-regulating Survivin mRNA and protein in transfected cancer cell lines, and induce apoptosis in combination with Taxol superior compared to other LNA oligonucleotides.
Owner:ENZON PHARM INC
Composition and method for treating tumor
ActiveUS20100104660A1Improving immunogenicityGood anticancer effectHeavy metal active ingredientsBiocideMedicineDrug carrier
Methods for treating neoplasm, tumors and cancers, using one or more tumor treating drug carriers, haptens and anticancer drugs, alone or in combination with other antineoplastic agents or treatments, are provided. Also provided are compositions, and kits containing the composition for affecting the therapy.
Owner:YU BAOFA
Tissue chip used for tumour early stage diagnosis and preparation device
Three kinds of tissues including cancer tissue, precancerosis and corresponding normal tissue are sliced up, dyed, marked, and positioned. Receptor holes are prepared by leading designed lattice array mould paper to paste on surface of wax block of receptor. Wax block with tissue core bar is prepared by using perforating needle and puncture needle for tissue. Common cancer such as lung cancer, nasopharyngeal carcinoma, oesophagus cancer etc. and having integrated clinical data and pathology features are selected. Through in situ hybridization, testing mRNA of relevant gene and expression of protein on tissue chip, consistent result between the invented product and traditional test is validated. In the product, cellular morphology is clear and even, and there is no fallen off tissue point. The invention is applicable to filter cancers, early diagnosis and forecasting prognosis.
Owner:中南大学湘雅医学院肿瘤研究所
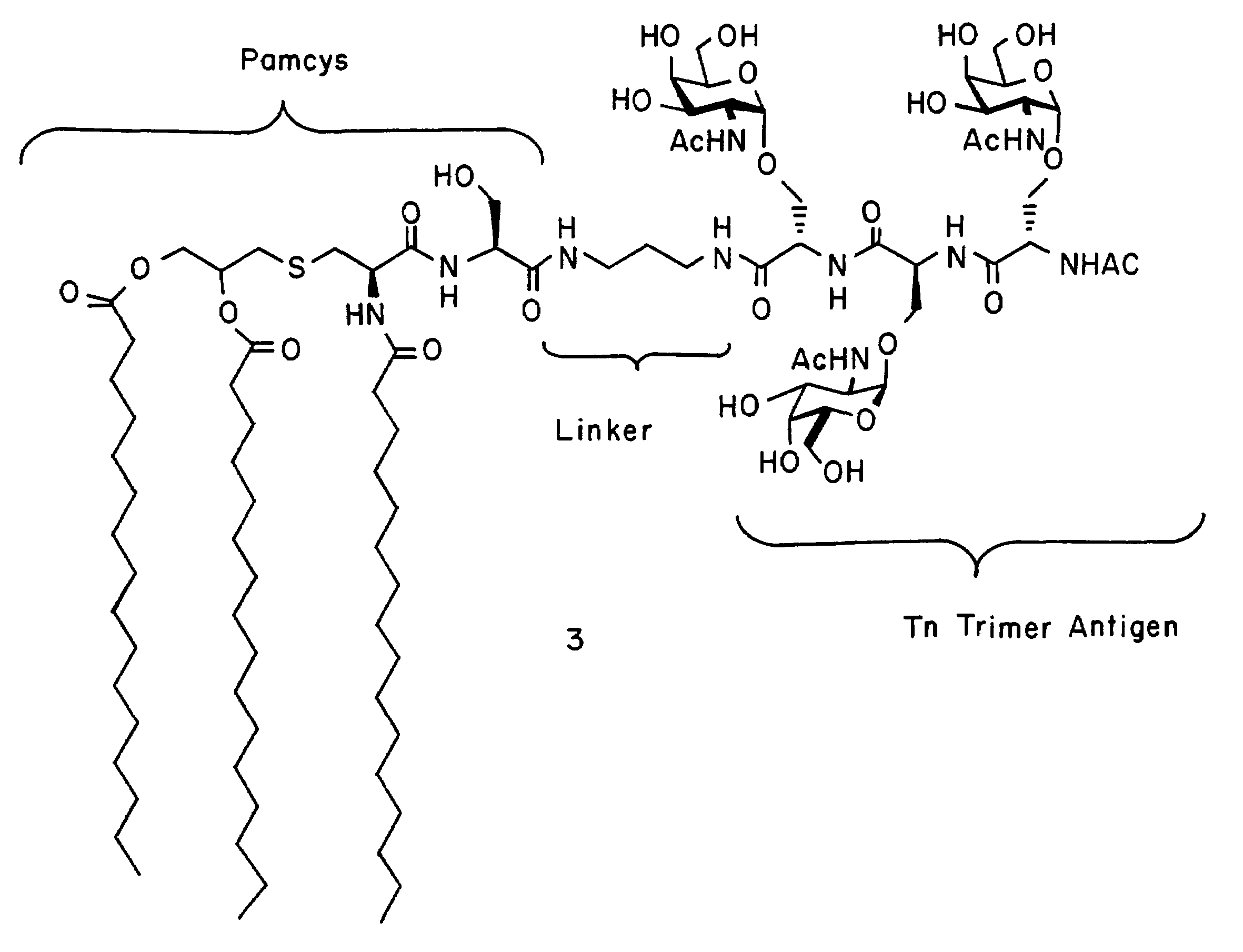
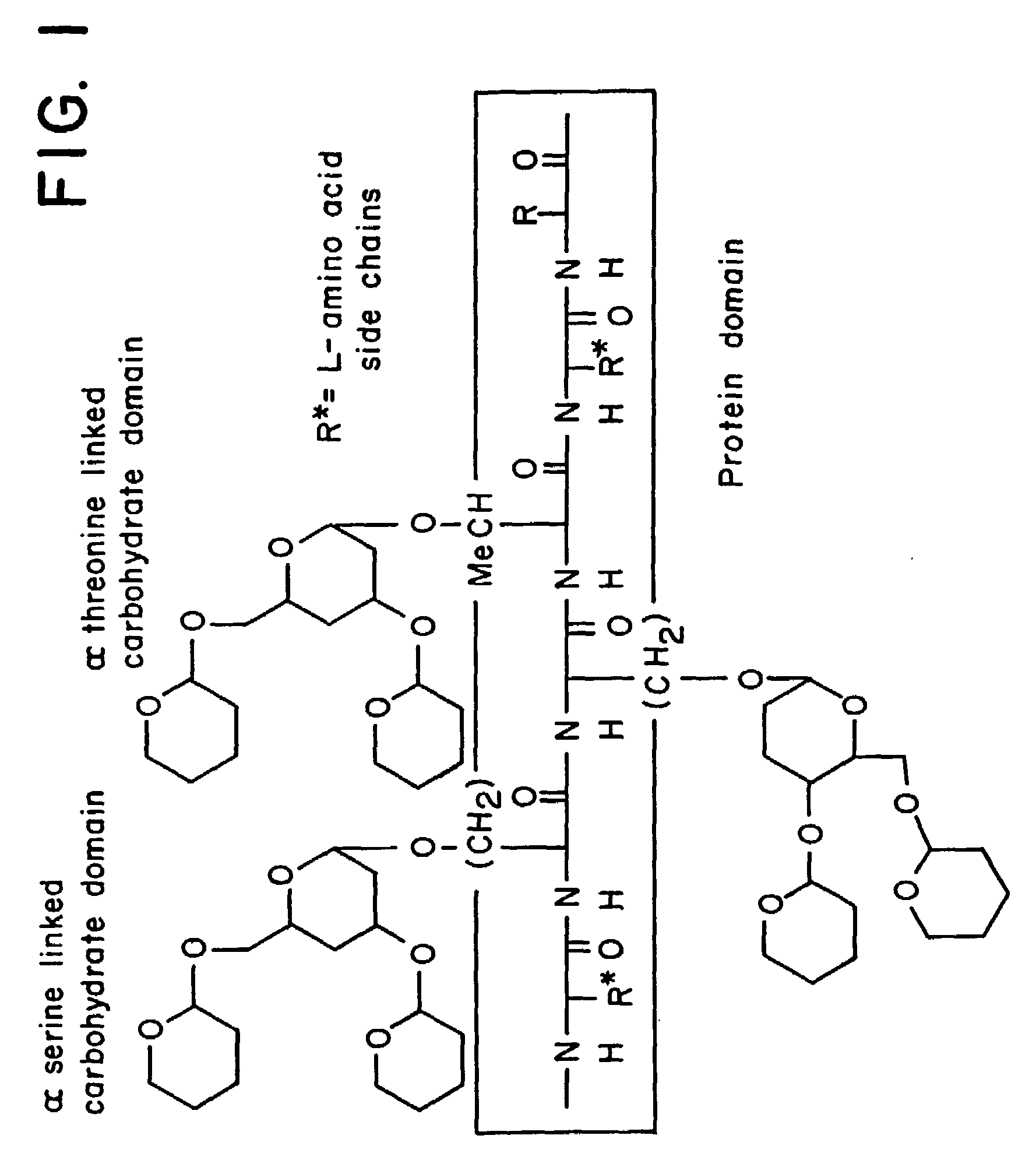
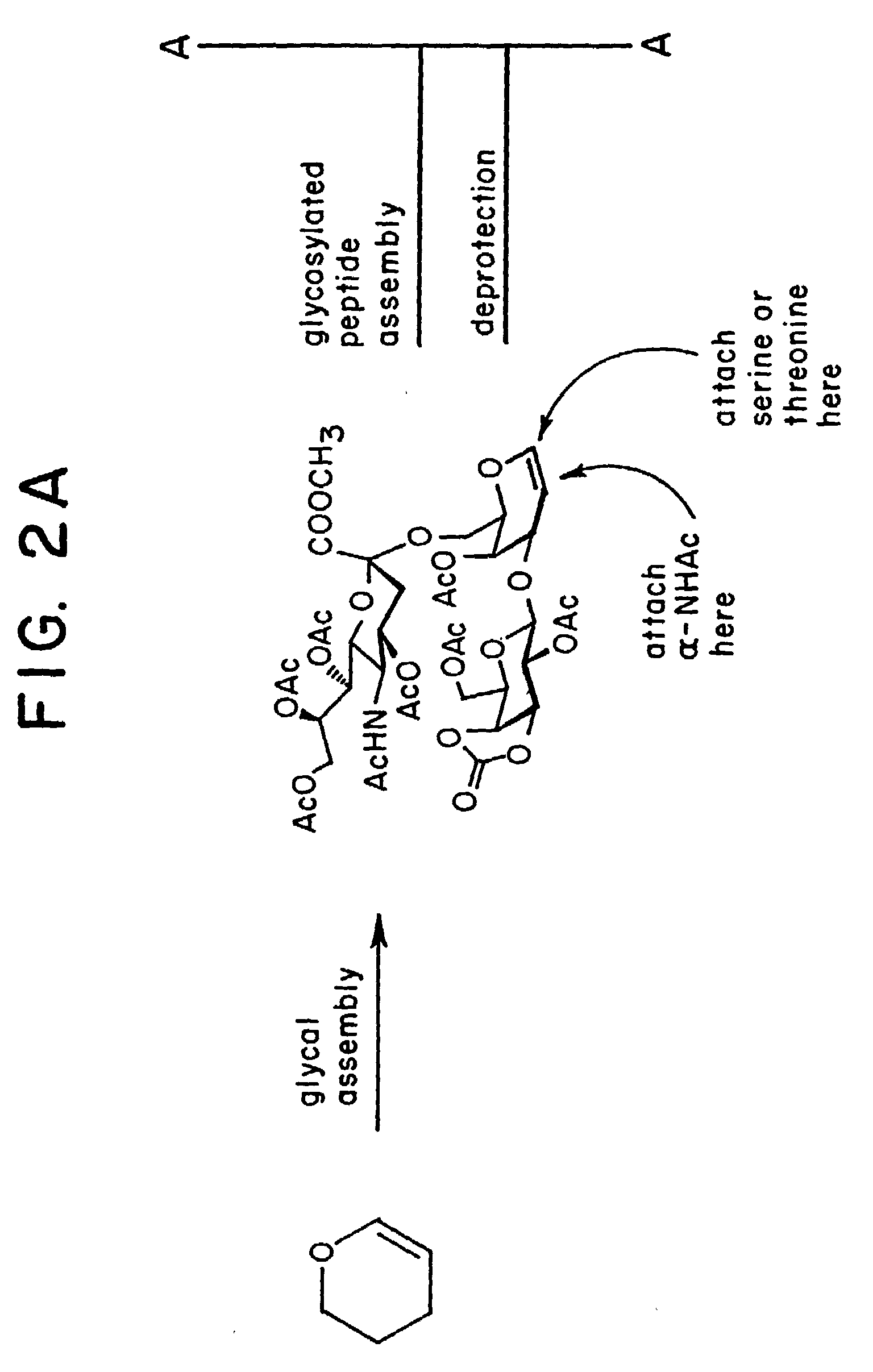
alpha-O-linked glycoconjugates, methods of preparation and uses thereof
The present invention provides novel α-O-linked glycoconjugates such as α-O-linked glycopeptides, as well convergent methods for synthesis thereof. The general preparative approach is exemplified by the synthesis of the mucin motif commonly found on epithelial tumor cell surfaces. The present invention further provides compositions and methods of treating cancer using the α-O-linked glycoconjugates.
Owner:SLOAN KETTERING INST FOR CANCER RES
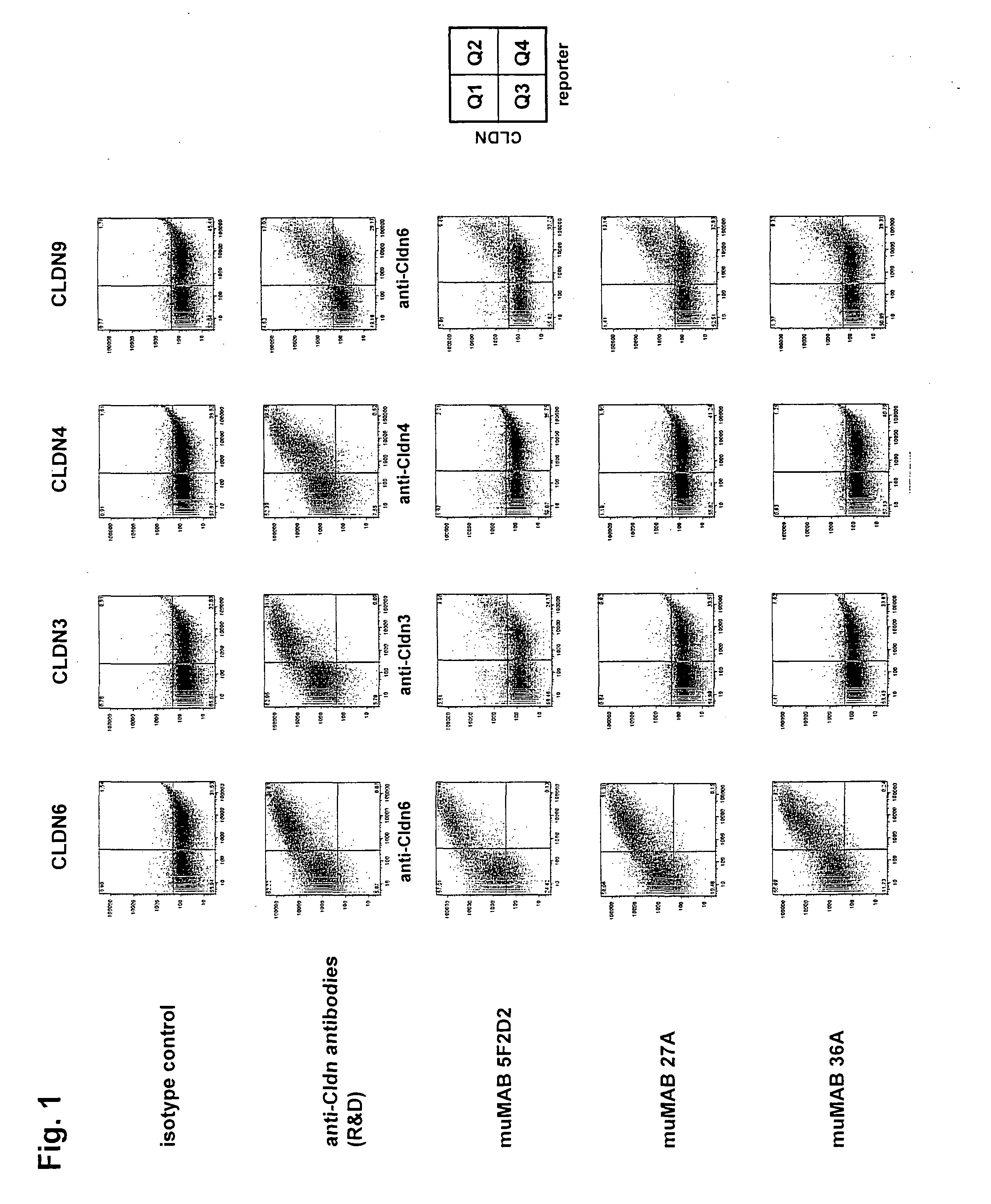
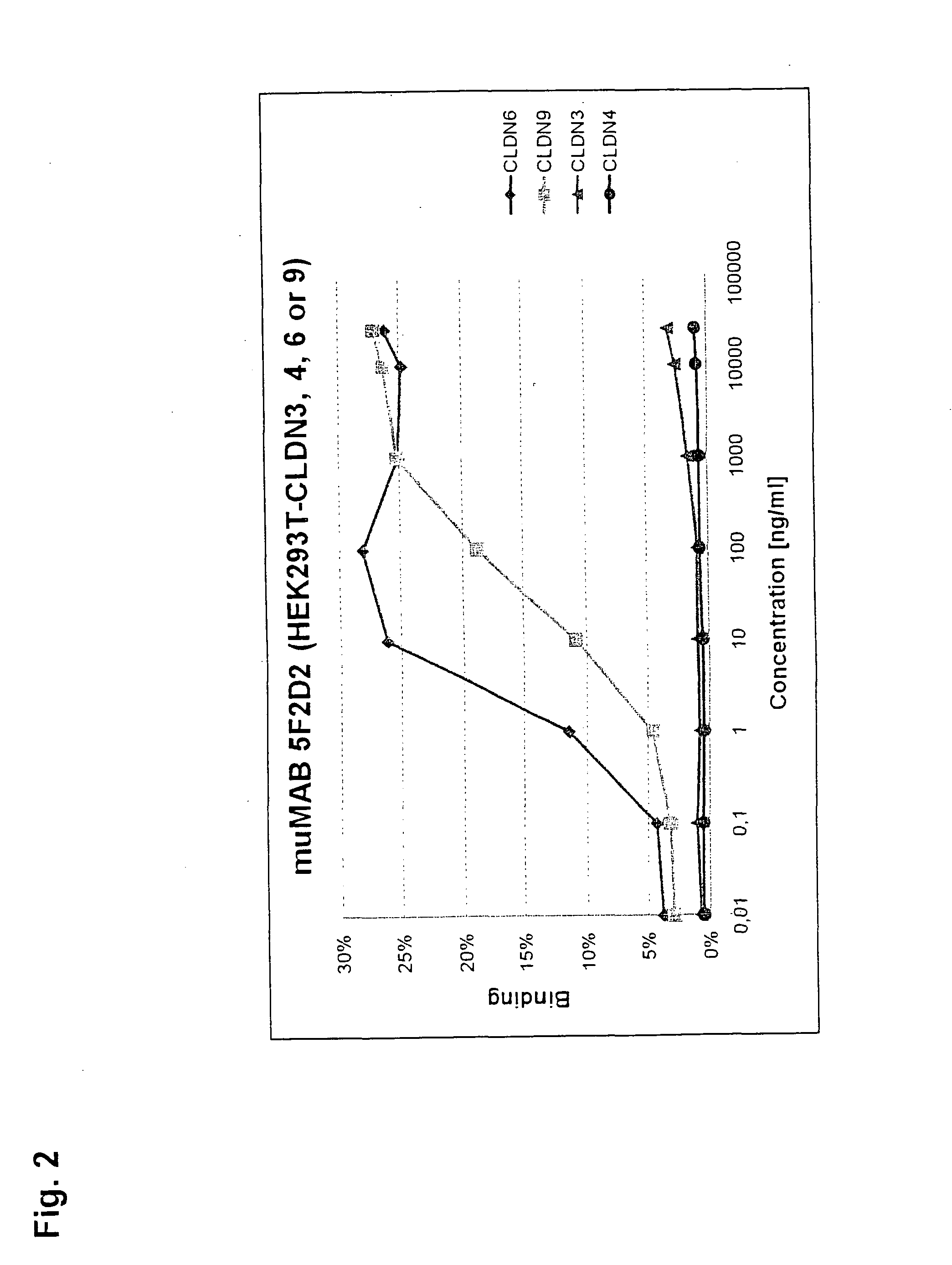
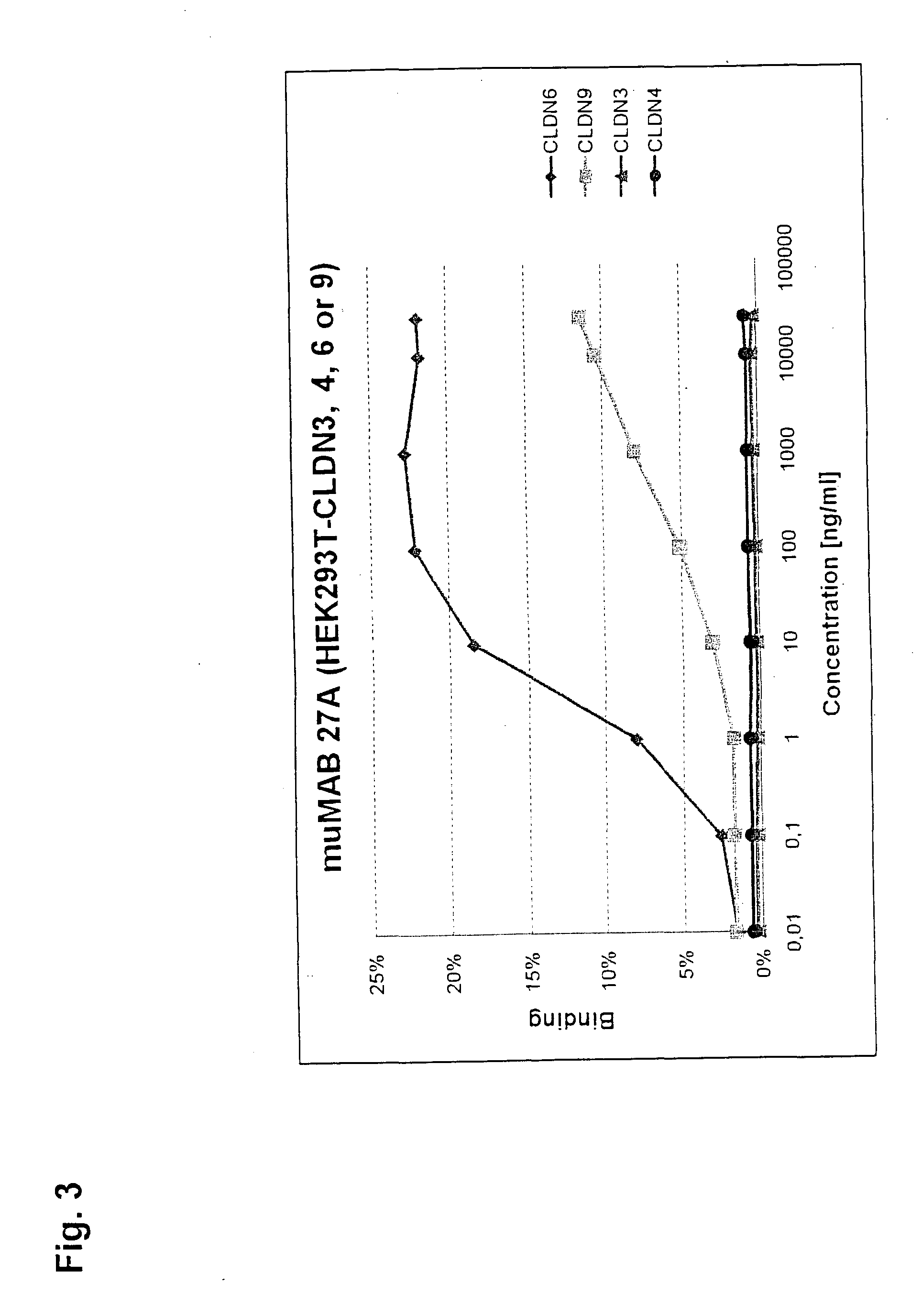
Cancer therapy using cldn6 target-directed antibodies in vivo
ActiveUS20130183305A1Improve survivalReduces tumor cell growthAnimal cellsCell receptors/surface-antigens/surface-determinantsAntibodyWilms tumour
The invention relates to the treatment and / or prevention of tumor diseases associated with cells expressing CLD-N6, in particular cancer and cancer metastasis using antibodies which bind to CLDN6. The present application demonstrates that the binding of antibodies to CLDN6 on the surface of tumor cells is sufficient to inhibit growth of the tumor and to prolong survival and extend the lifespan of tumor patients. Furthermore, binding of antibodies to CLDN6 is efficient in inhibiting growth of CLDN6 positive germ cell tumors such as teratocarcinomas or embryonal carcinomas, in particular germ cell tumors of the testis.
Owner:JOHANNES GUTENBERG UNIV +1
SNP marker related to primary hepatocellular carcinoma auxiliary diagnosis and application of SNP marker
InactiveCN104745710AHelps reflect disease stateQuickly and accurately grasp the conditionMicrobiological testing/measurementDNA/RNA fragmentationHepatic carcinomaWilms' tumor
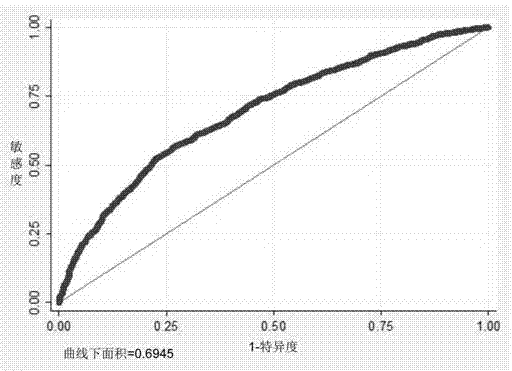
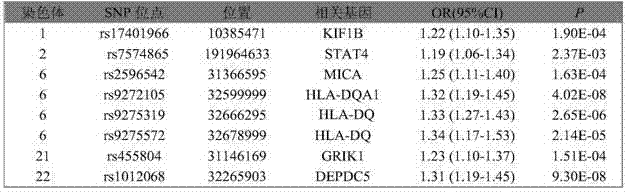
The invention belongs to the genetic engineering and tumour medicine fields and discloses an SNP marker related to primary hepatocellular carcinoma auxiliary diagnosis and an application of the SNP marker. The marker is a combination of rs7574865, rs1012068, rs17401966, rs2596542, rs455804, rs9272105, rs9275319 and rs9275572. The marker can be used for preparing a hepatic carcinoma auxiliary diagnosis kit.
Owner:SHANGHAI ROSETTA BIOTECH CO LTD
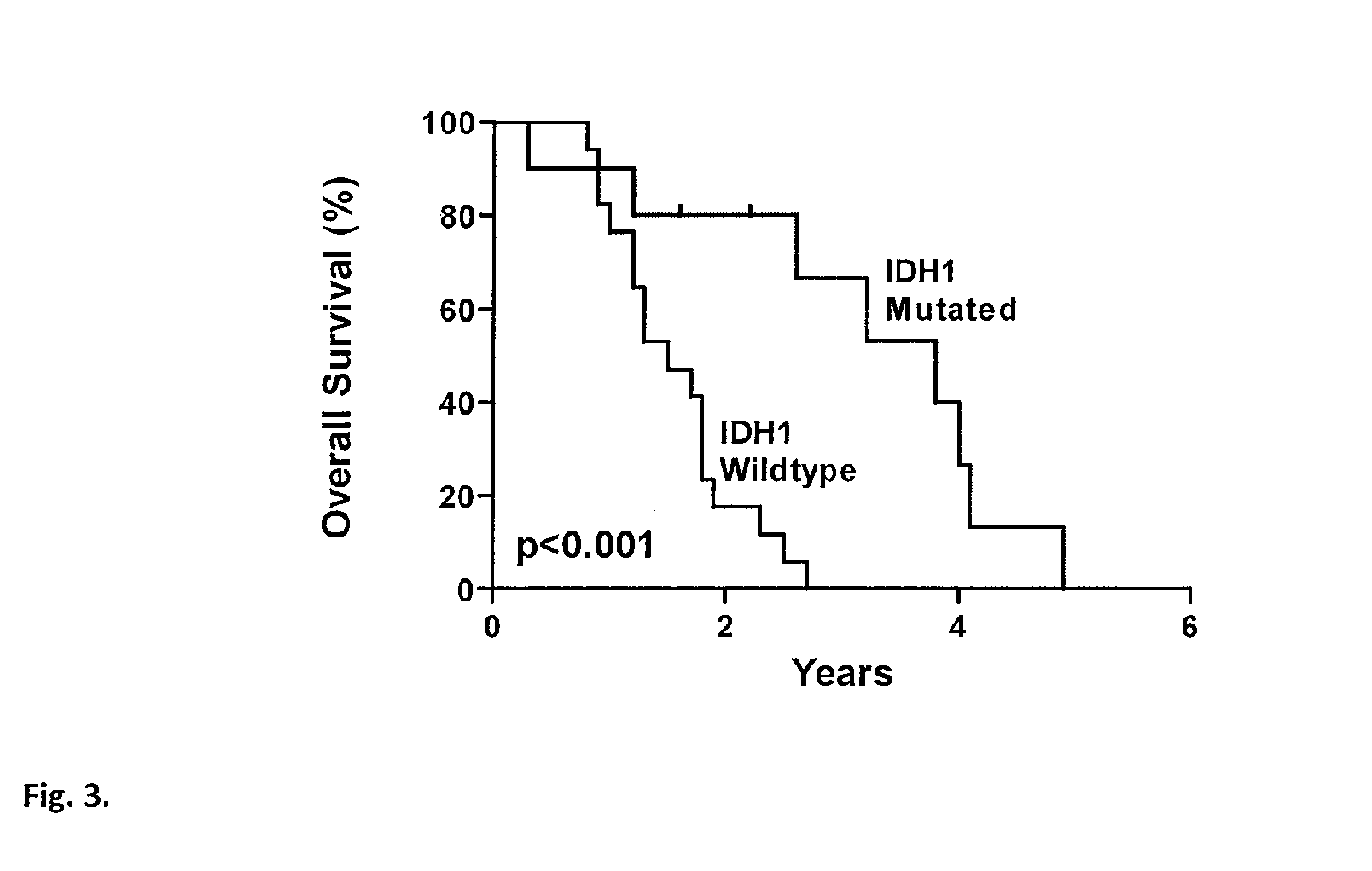
Genetic alterations in isocitrate dehydrogenase and other genes in malignant glioma
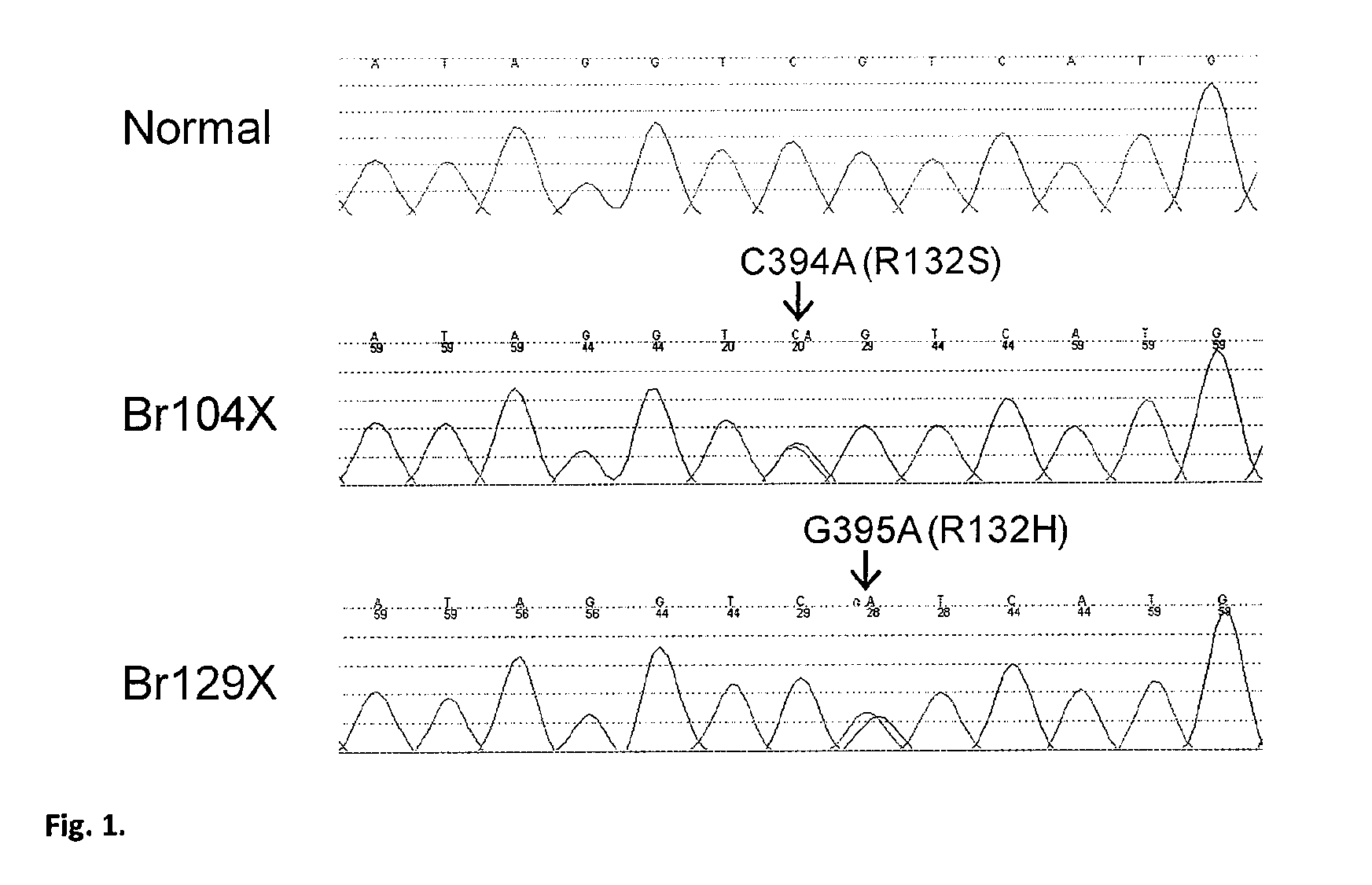
We found mutations of the R132 residue of isocitrate dehydrogenase 1 (IDH1) in the majority of grade II and III astrocytomas and oligodendrogliomas as well as in glioblastomas that develop from these lower grade lesions. Those tumors without mutations in IDH1 often had mutations at the analogous R172 residue of the closely related IDH2 gene. These findings have important implications for the pathogenesis and diagnosis of malignant gliomas.
Owner:THE JOHN HOPKINS UNIV SCHOOL OF MEDICINE +1
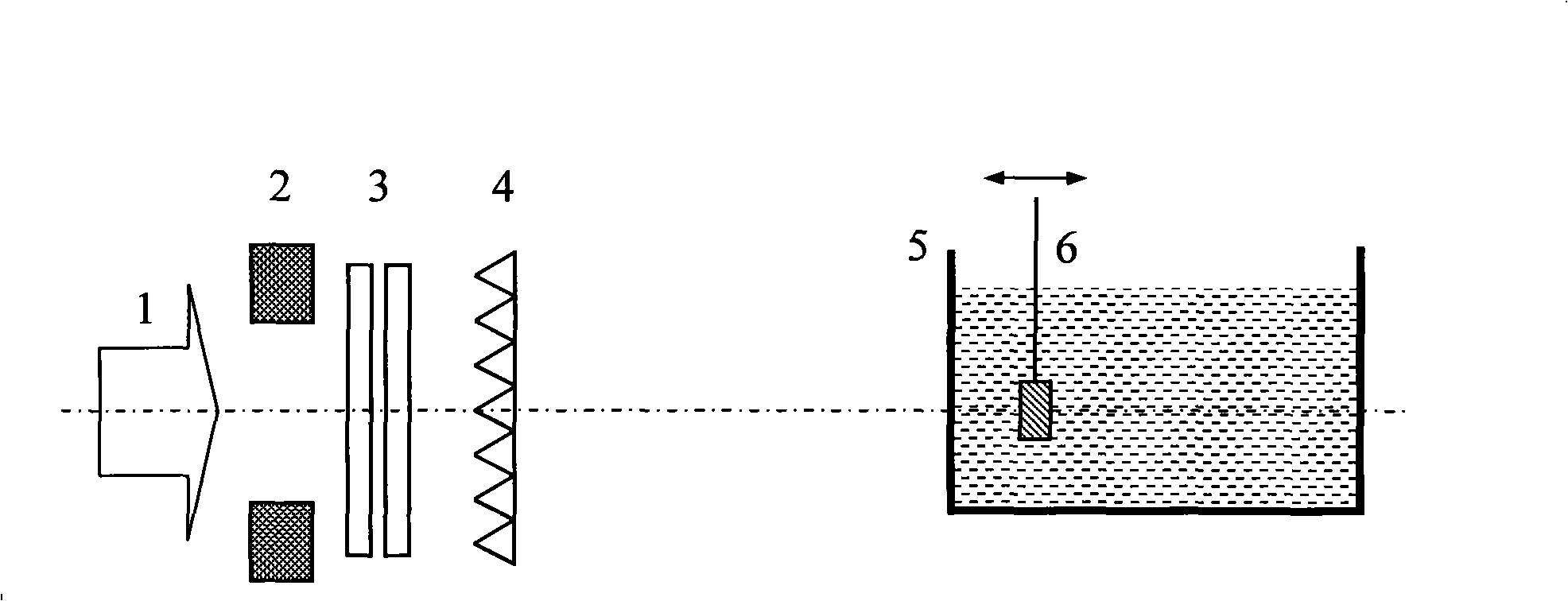
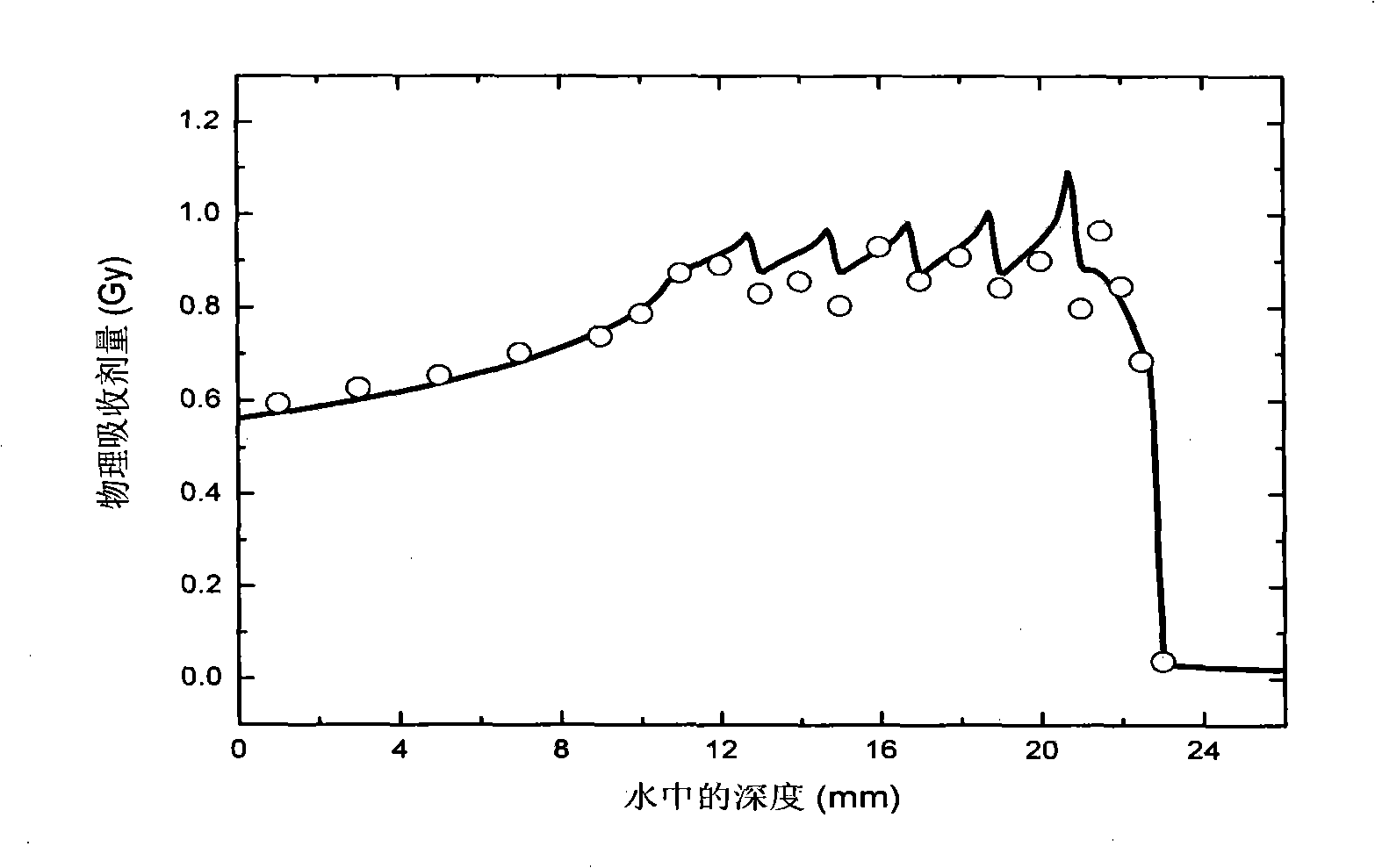
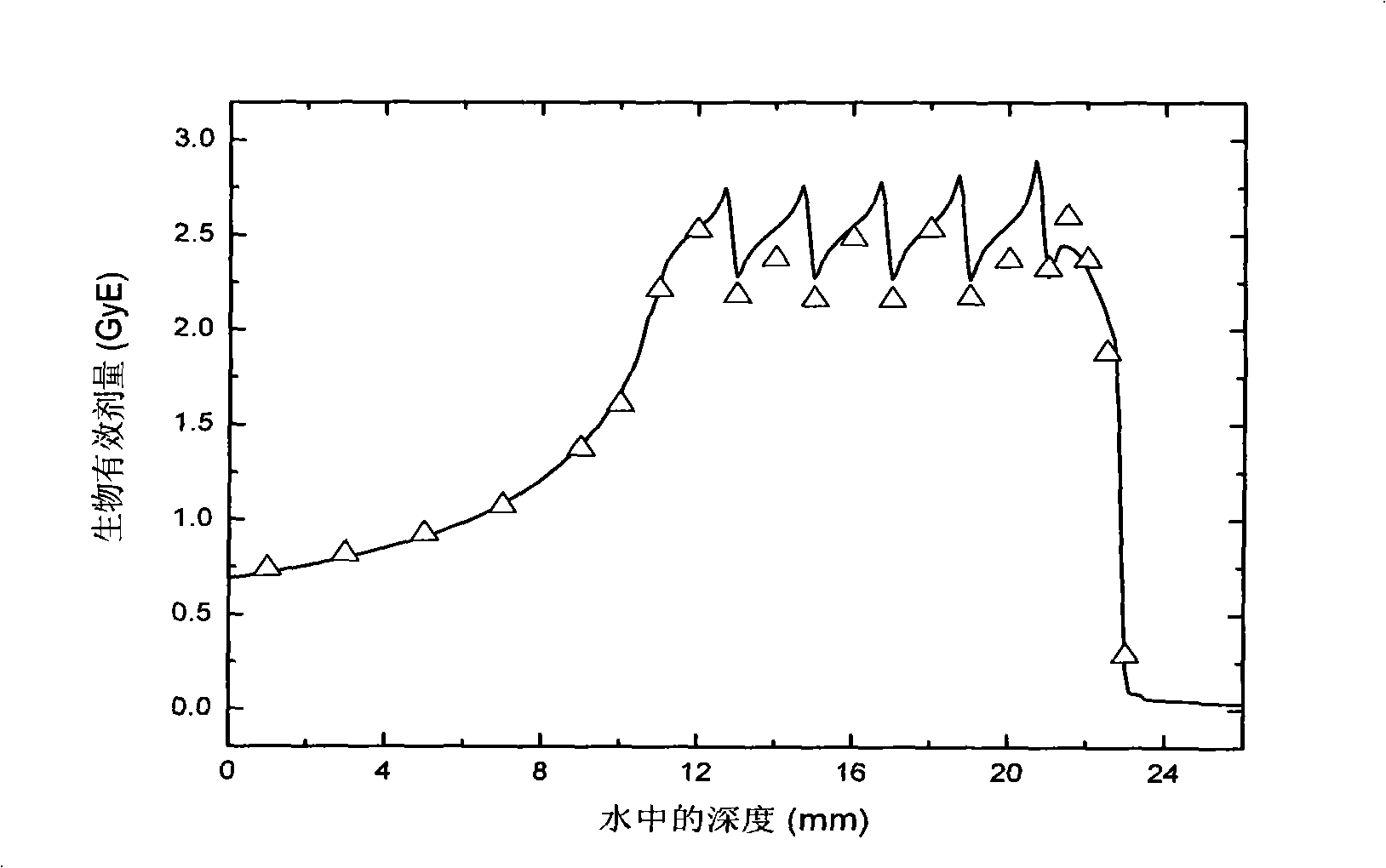
Dose monitoring detector calibration device and method in heavy ion beam treating carcinoma
ActiveCN101285887A3D Conformal Radiation Therapy ControlImprove treatment efficiencyDosimetersBragg peakTumor target
The invention relates to a device and a method for demarcating and calibrating a dose monitoring detector in heavy ion beam cancer treatment. The structure of the device is characterized in that a collimator, the dose monitoring detector, a mini ridge-shaped filter, a water tank and a standard ionization chamber are arranged on a beam flux axis in sequence. The standard ionization chamber is arranged inside the water tank. The depth position of an irradiation beam mini spread-out Bragg peak in water is obtained by measuring absorbed dose of the standard ionization chamber at different depth in aqueous medium. At the depth position, the dose monitoring detector is demarcated and calibrated by the standard ionization chamber so as to obtain demarcating and calibrating factors of the measurement of the dose monitoring detector for the mini spread-out Bragg peak cancer treatment beam with a Gauss arrangement. With the demarcating and calibrating factors, the entire process of three-dimensional conformal irradiation therapy with uniform physical absorption dose or uniform biological effective dose in a tumor target volume can be conveniently controlled, the requirements of the treatment of different clinical cases in practical clinical treatment are satisfied, and the treatment efficiency of a treatment device is improved.
Owner:INST OF MODERN PHYSICS CHINESE ACADEMY OF SCI
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com