Patents
Literature
49 results about "Ascomycin" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
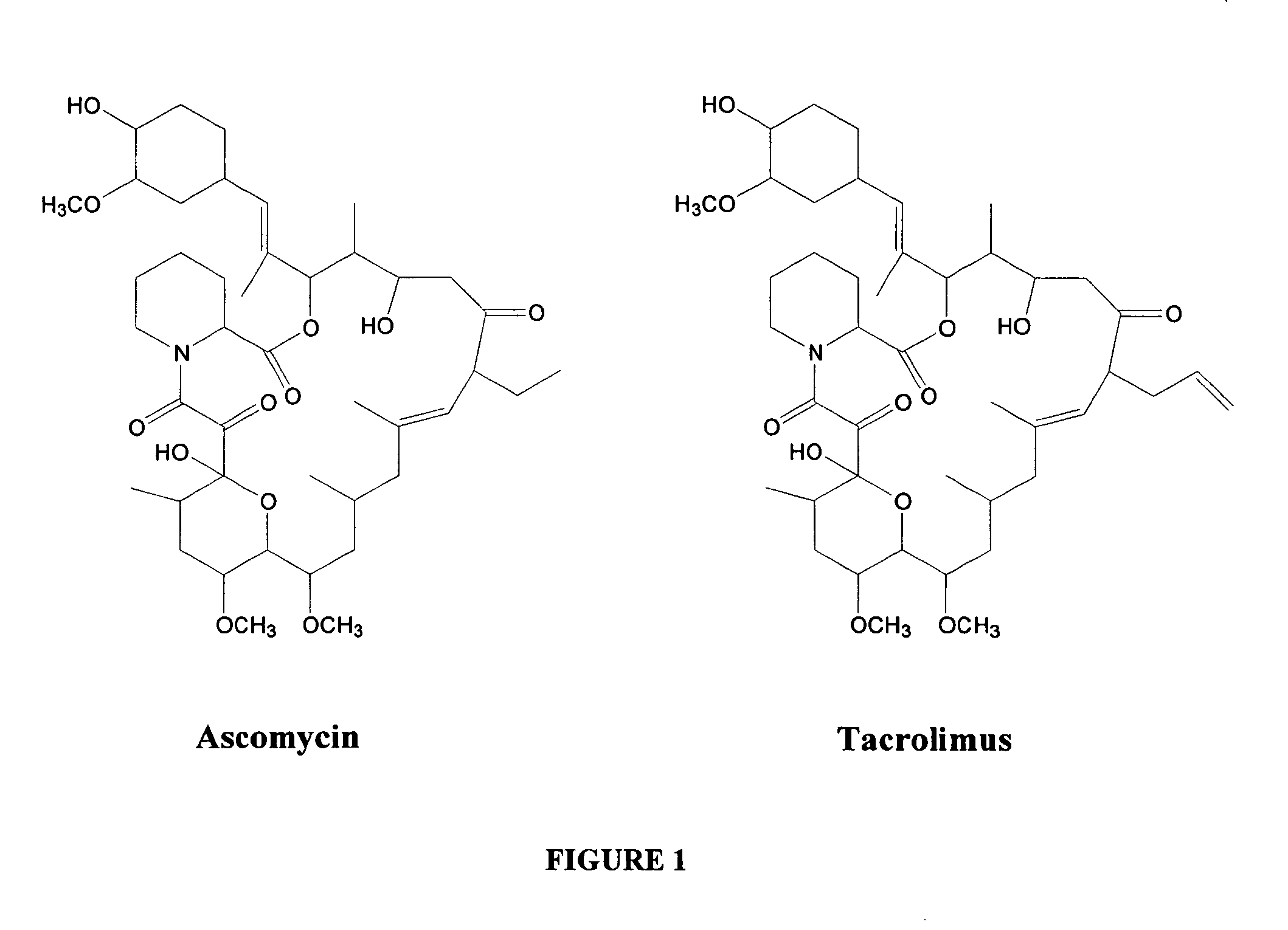
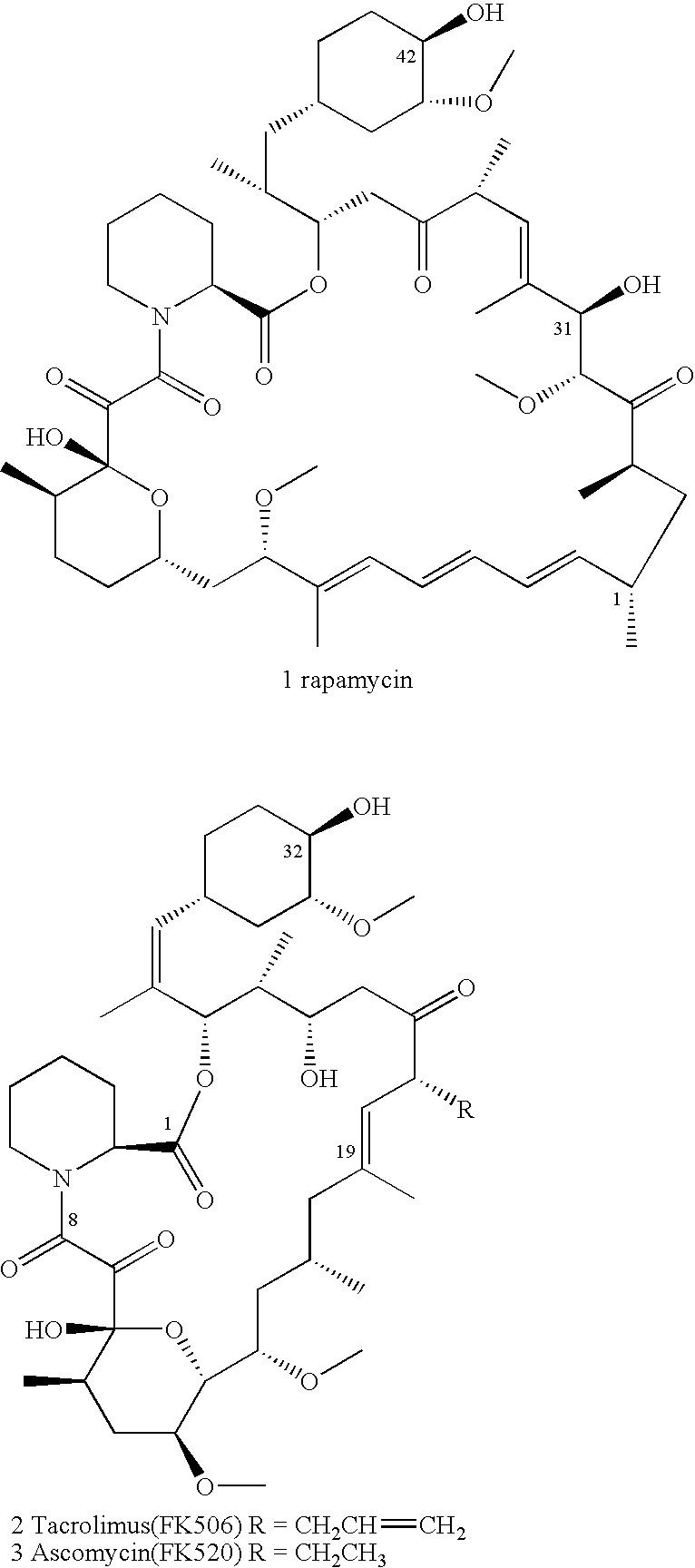
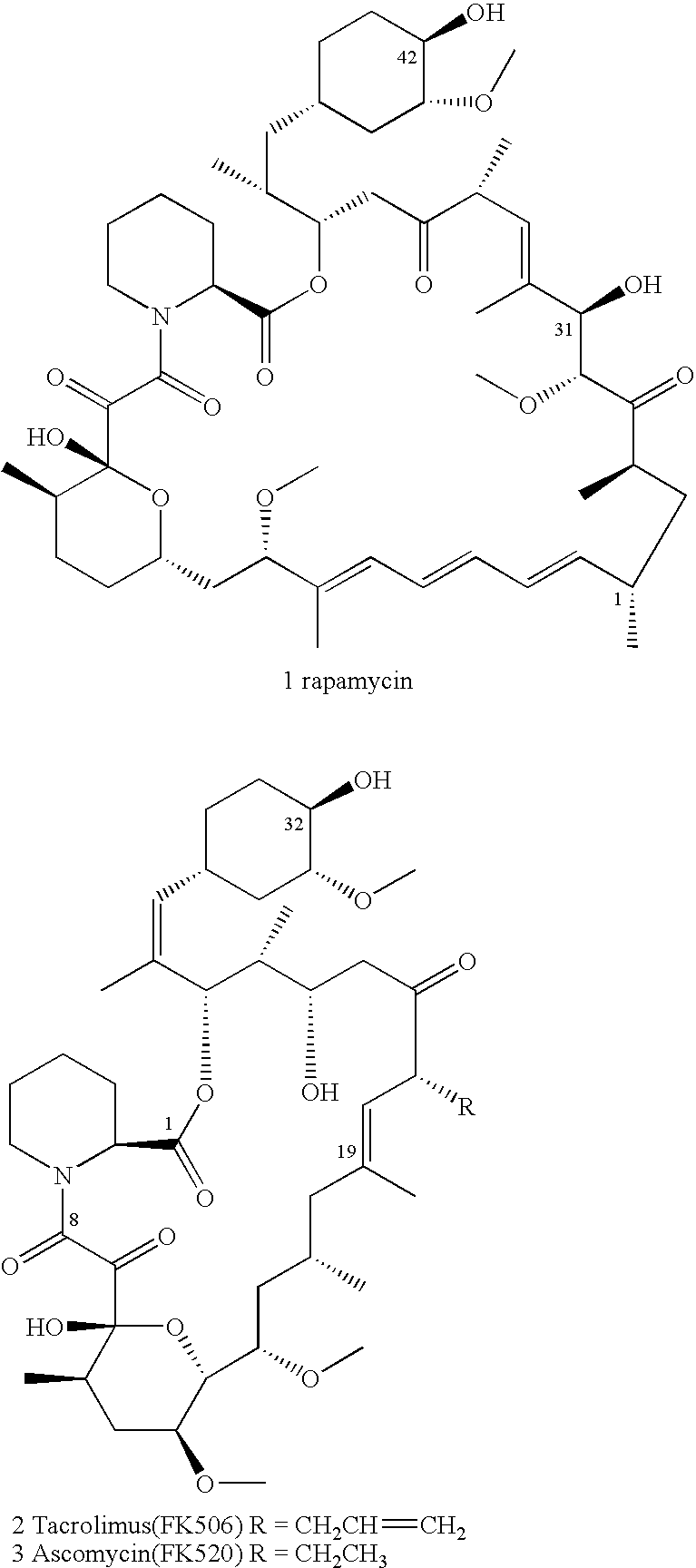
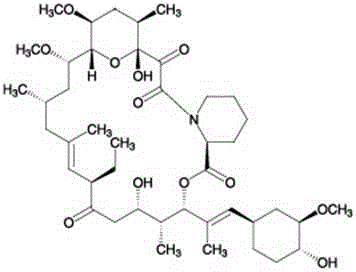
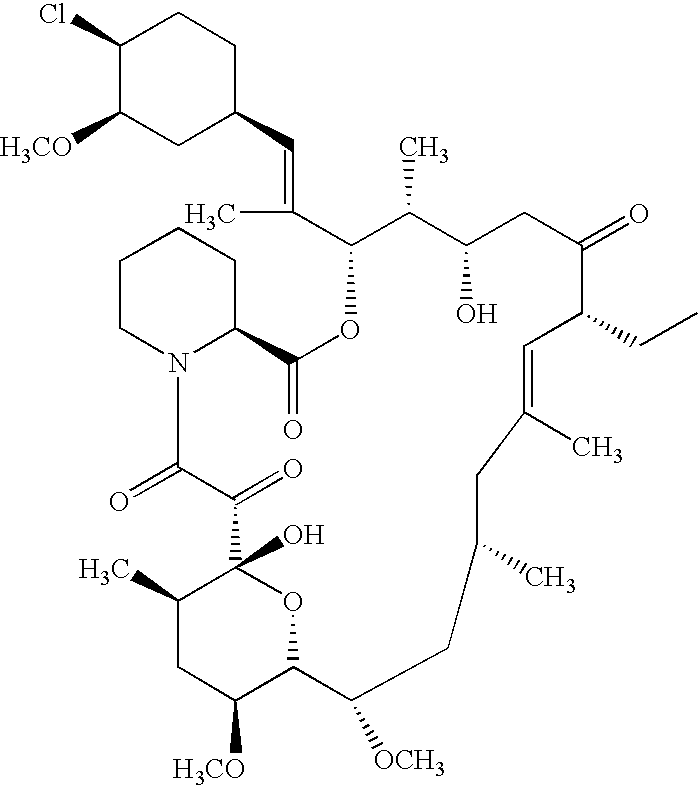
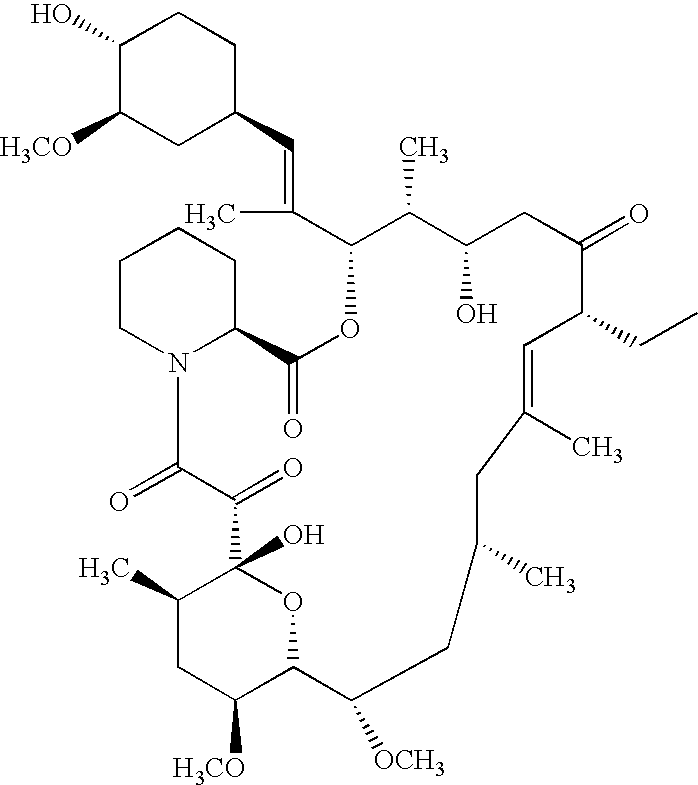
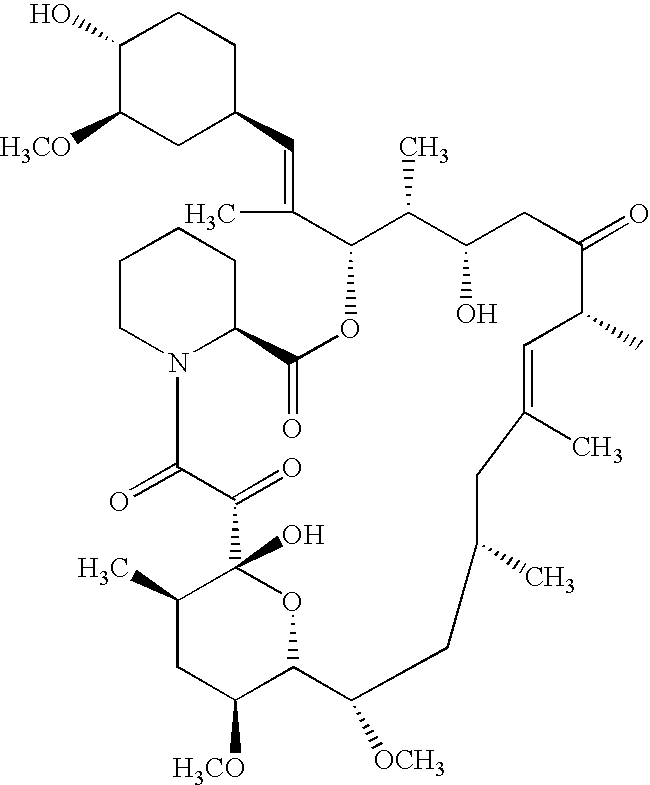
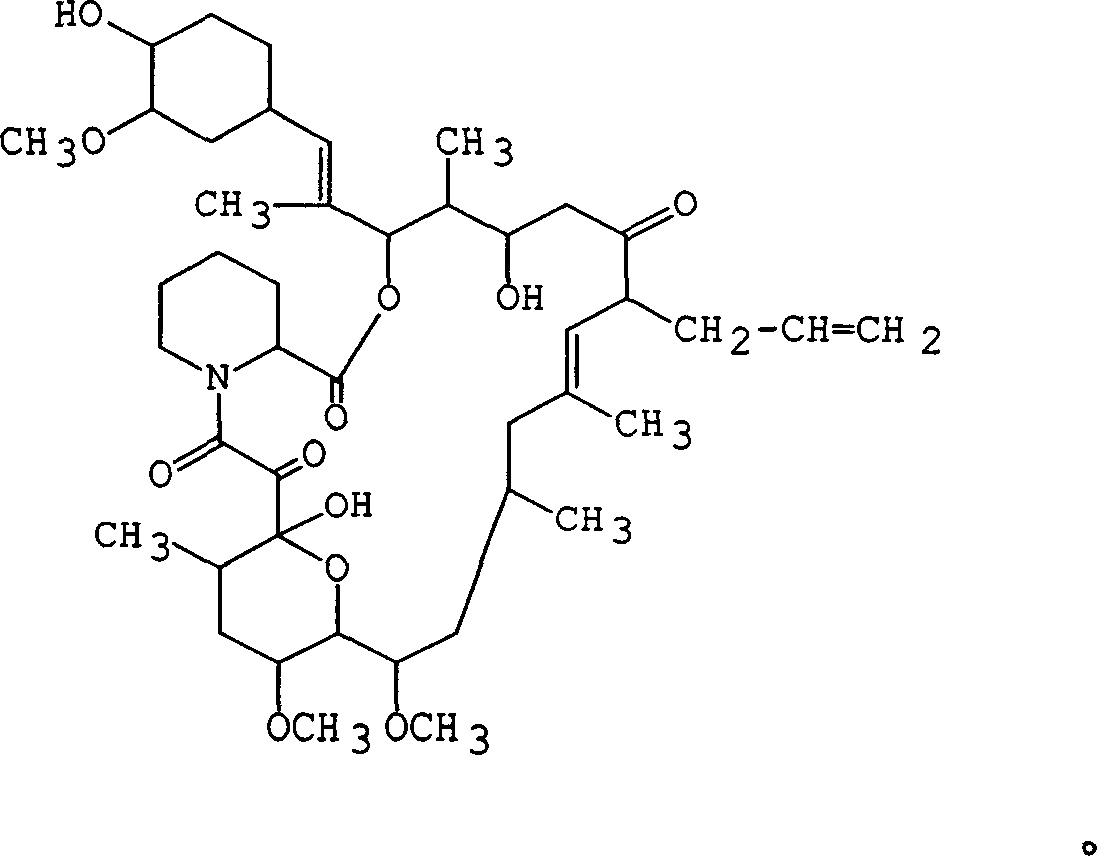
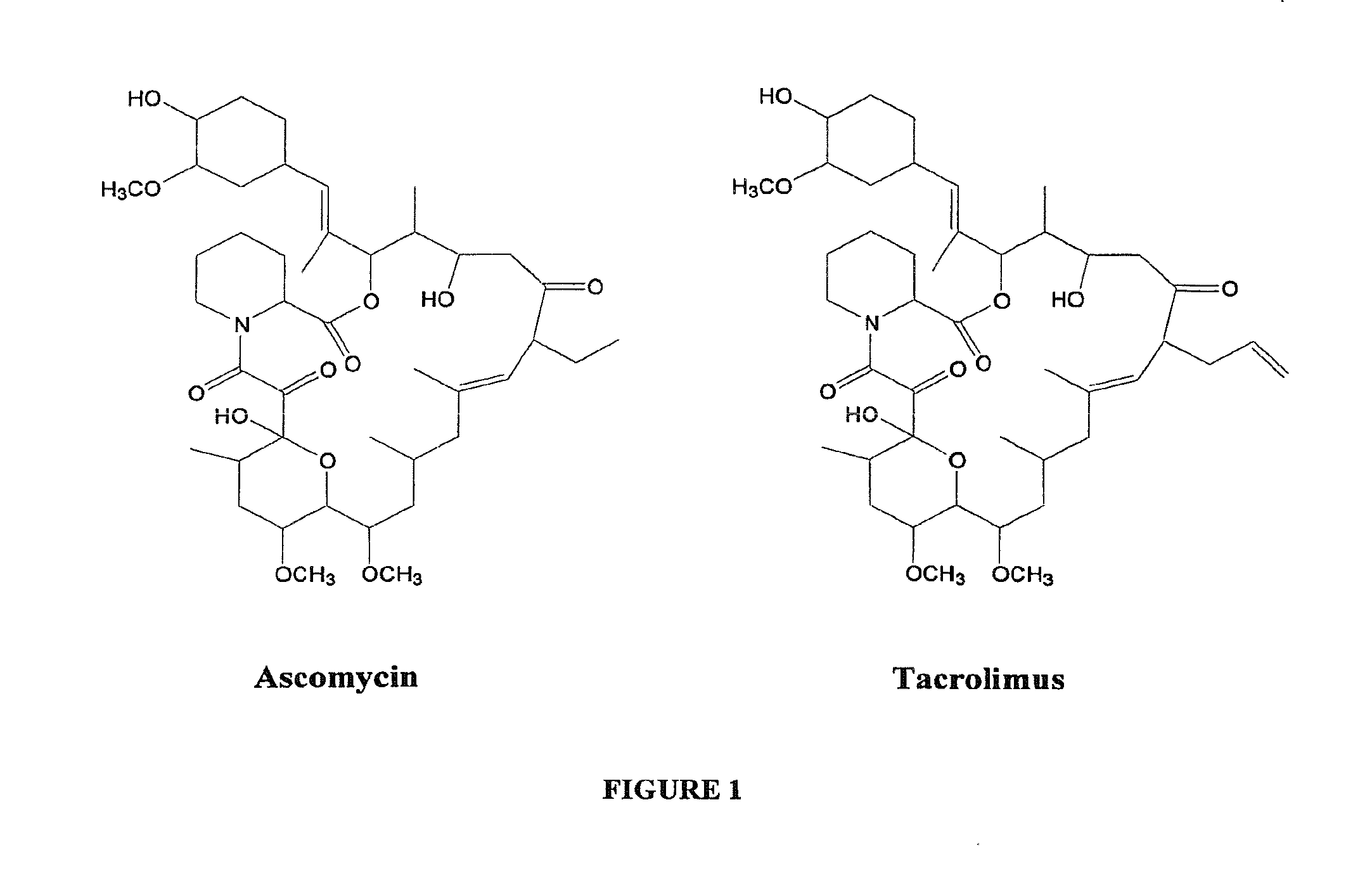
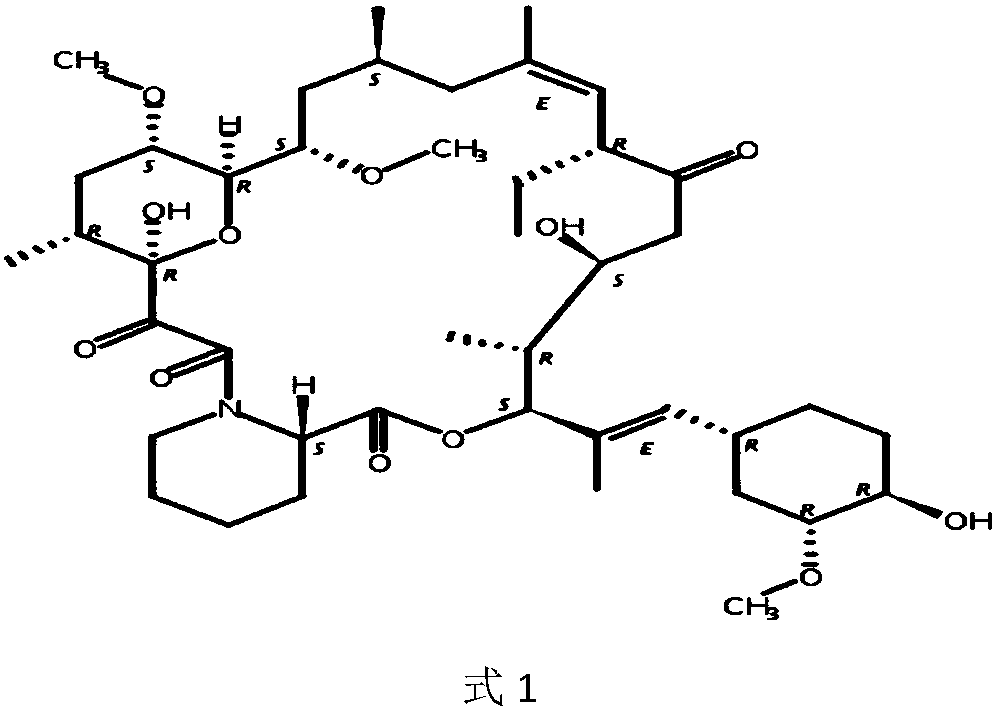
Ascomycin, also called Immunomycin, FR-900520, FK520, is an ethyl analog of tacrolimus (FK506) with strong immunosuppressant properties. It has been researched for the treatment of autoimmune diseases and skin diseases, and to prevent rejection after an organ transplant.
Treatment of ocular disease
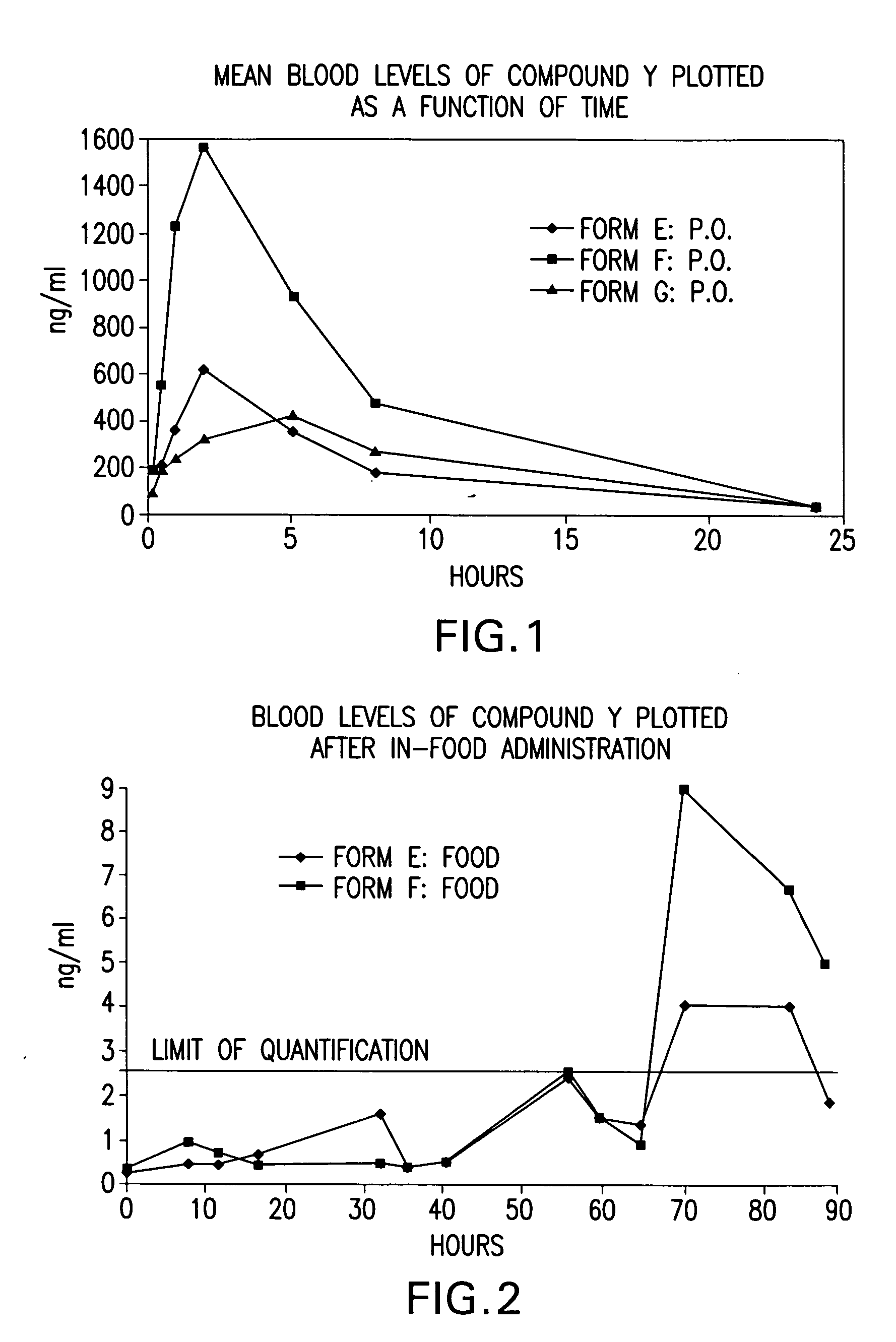
A formulation to treat ocular conditions such as dry eye disease, as well as other conditions, is disclosed. Rapamycin and / or ascomycin is administered intraocularly, such as by topical application, injection into the eye, or implantation in or on the eye. For example, a topical administration may contain between about 50 pg / ml drug to about 50 μg / ml drug in a formulation which may be applied at bedtime or throughout the day. For injection, a dose of about 50 pg / ml to about 200 μg / ml may be used. Rapamycin and / or ascomycin may also be administered in milligram quantities as a surgical implant, for example, in a diffusible walled reservoir sutured to the wall of the sclera, or may be contained within an inert carrier such as microspheres or liposomes to provide a slow-release drug delivery system.
Owner:PEYMAN GHOLAM A DR
Treatment of ocular disease
InactiveUS20050025810A1Prevent and decrease time of onsetReduce severityBiocideSenses disorderMicrosphereSurgical implant
A formulation to treat ocular conditions such as dry eye disease, as well as other conditions, is disclosed. Rapamycin and / or ascomycin is administered intraocularly, such as by topical application, injection into the eye, or implantation in or on the eye. For example, a topical administration may contain between about 50 pg / ml drug to about 50 μg / ml drug in a formulation which may be applied at bedtime or throughout the day. For injection, a dose of about 50 pg / mi to about 200 μg / ml may be used. Rapamycin and / or ascomycin may also be administered in milligram quantities as a surgical implant, for example, in a diffusible walled reservoir sutured to the wall of the sclera, or may be contained within an inert carrier such as microspheres or liposomes to provide a slow-release drug delivery system.
Owner:PEYMAN GHOLAM A DR
Ocular solutions
InactiveUS7083803B2Reduce inflammationReduce bacterial growthBiocideSenses disorderDiseaseEverolimus
Ocular solutions containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions. The solution may contain a supratherapeutic concentration of agent(s) so that a therapeutic concentration of a topically administered solution accumulates in a diseased ocular structure sufficient to treat the disease.
Owner:PEYMAN GHOLAM A DR
Ocular solutions
InactiveUS20050063996A1Reduce turbidityReduce inflammationAntibacterial agentsBiocideEverolimusCell migration
Ocular solutions containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial, and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions.
Owner:PEYMAN GHOLAM A DR
Treatment of ocular disease
InactiveUS20060263409A1Reduce onset timeReduce severityBiocideSenses disorderMicrosphereSurgical implant
A formulation to treat ocular conditions such as dry eye disease, as well as other conditions, is disclosed. Rapamycin and / or ascomycin is administered intraocularly, such as by topical application, injection into the eye, or implantation in or on the eye. For example, a topical administration may contain between about 50 pg / ml drug to about 50 μg / ml drug in a formulation which may be applied at bedtime or throughout the day. For injection, a dose of about 50 pg / ml to about 200 μg / ml may be used. Rapamycin and / or ascomycin may also be administered in milligram quantities as a surgical implant, for example, in a diffusible walled reservoir sutured to the wall of the sclera, or may be contained within an inert carrier such as microspheres or liposomes to provide a slow-release drug delivery system.
Owner:MINU
Ocular solutions
InactiveUS20060228394A1Reduce inflammationReduce bacterial growthAntibacterial agentsBiocideEverolimusOcular structure
Ocular solutions containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial, and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions. The solution may contain a supratherapeutic concentration of agent(s) so that a therapeutic concentration of a topically administered solution accumulates in a diseased ocular structure sufficient to treat the disease. The agent(s) may be formulated with polymers or other components for extended or slow release to provide a substantially constant concentration over the course of treatment.
Owner:MINU
Ocular solutions
InactiveUS7087237B2Reduce inflammationReduce bacterial growthAntibacterial agentsBiocideEverolimusMacrolide resistance
Containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial, and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions.
Owner:PEYMAN GHOLAM A DR
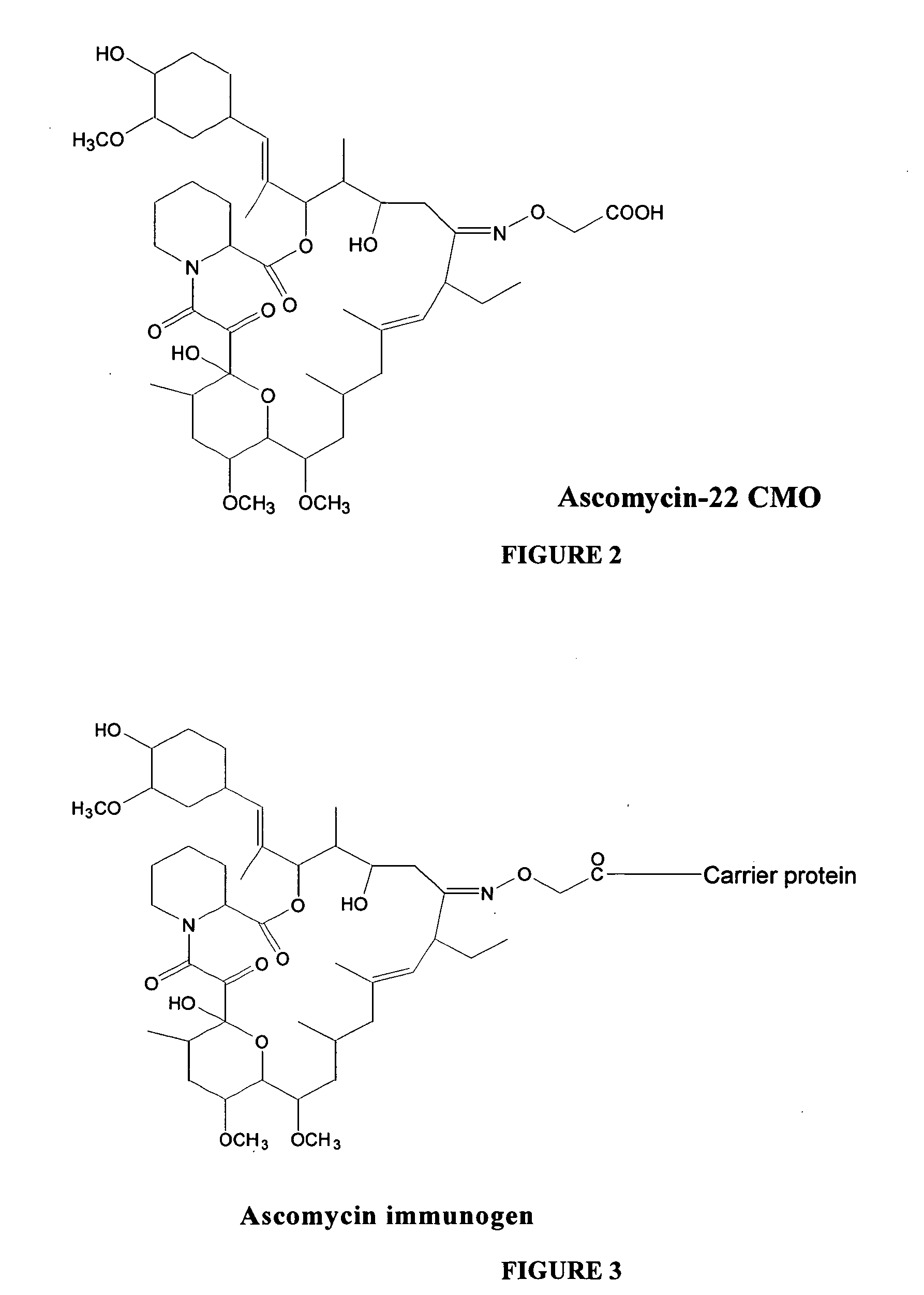
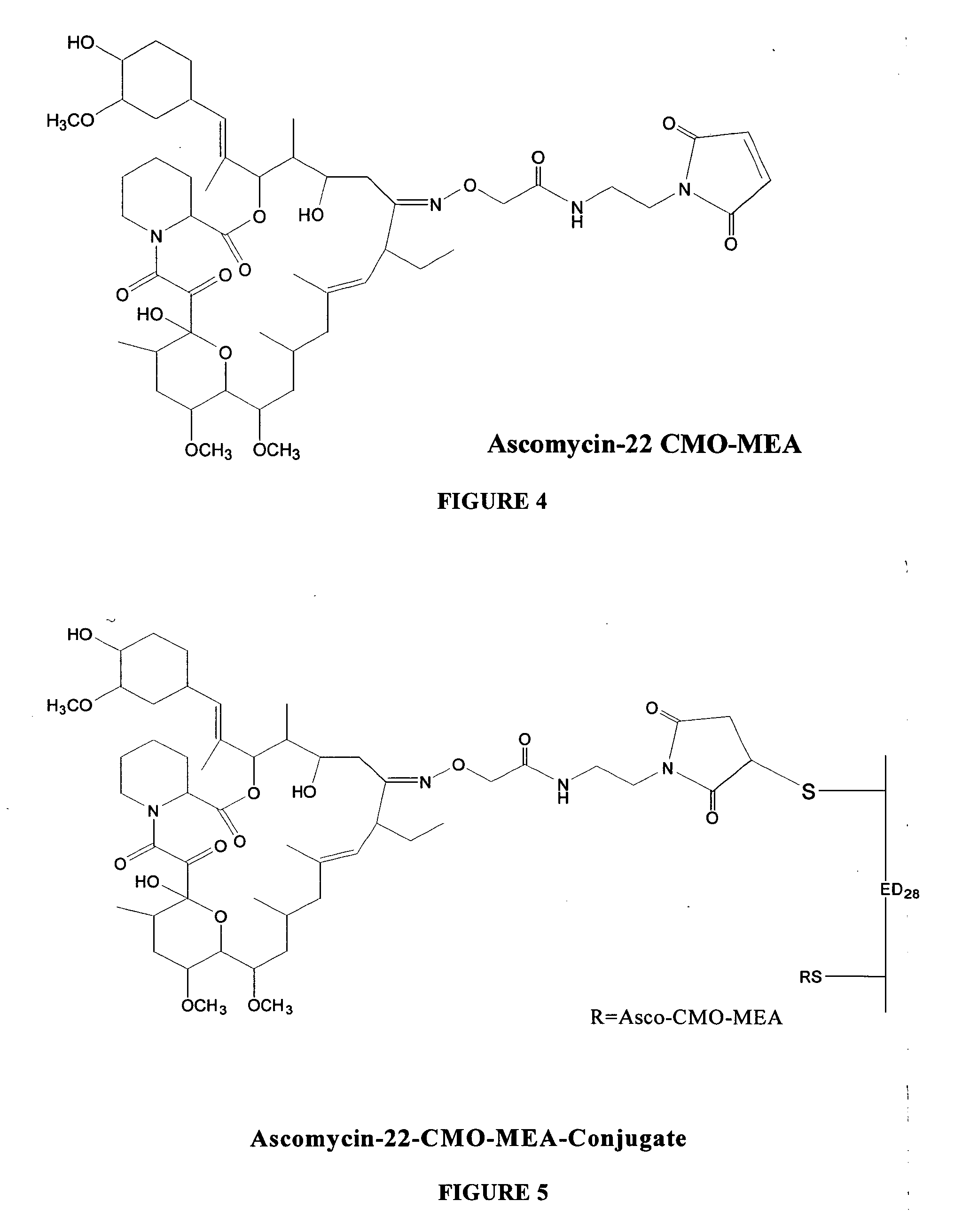
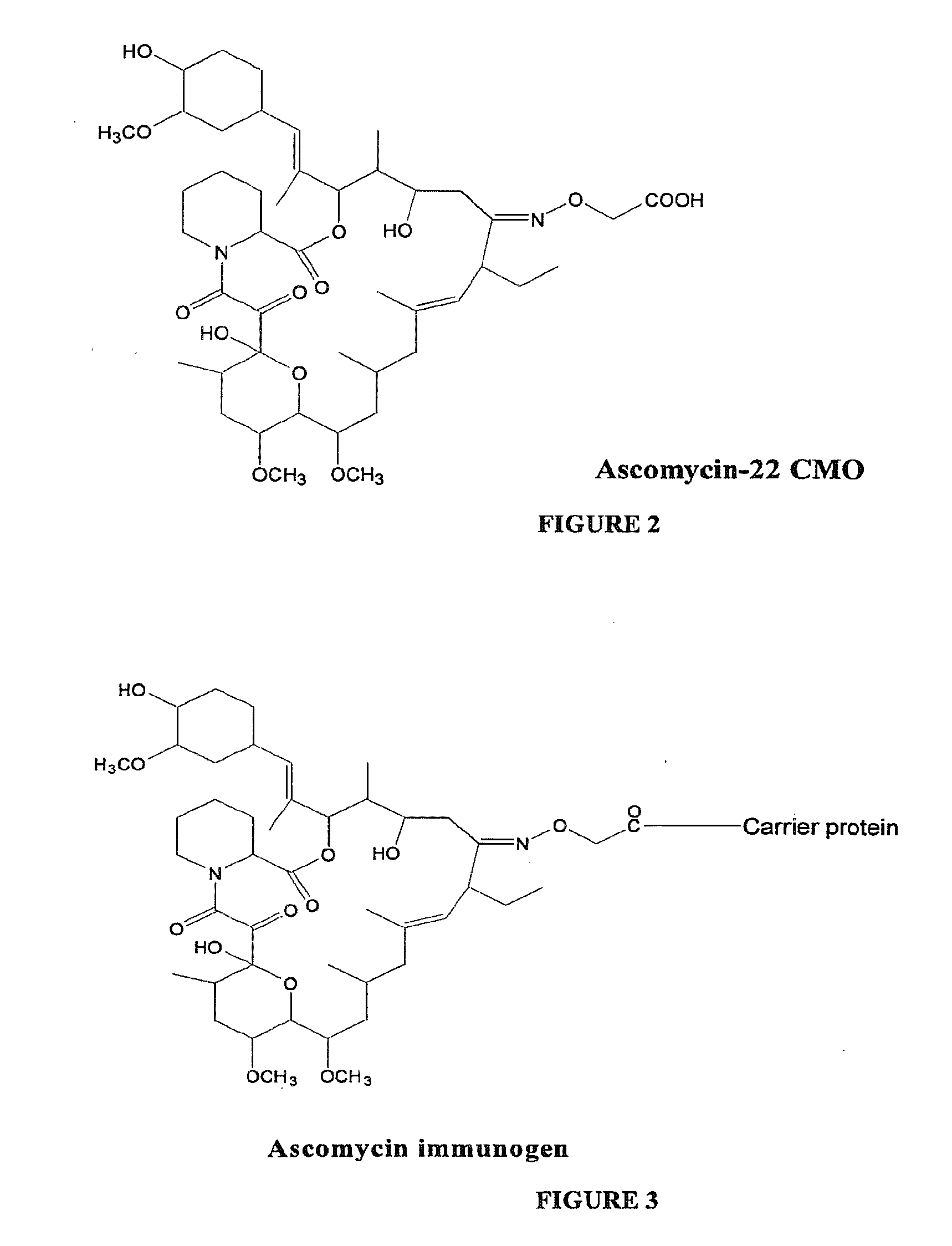
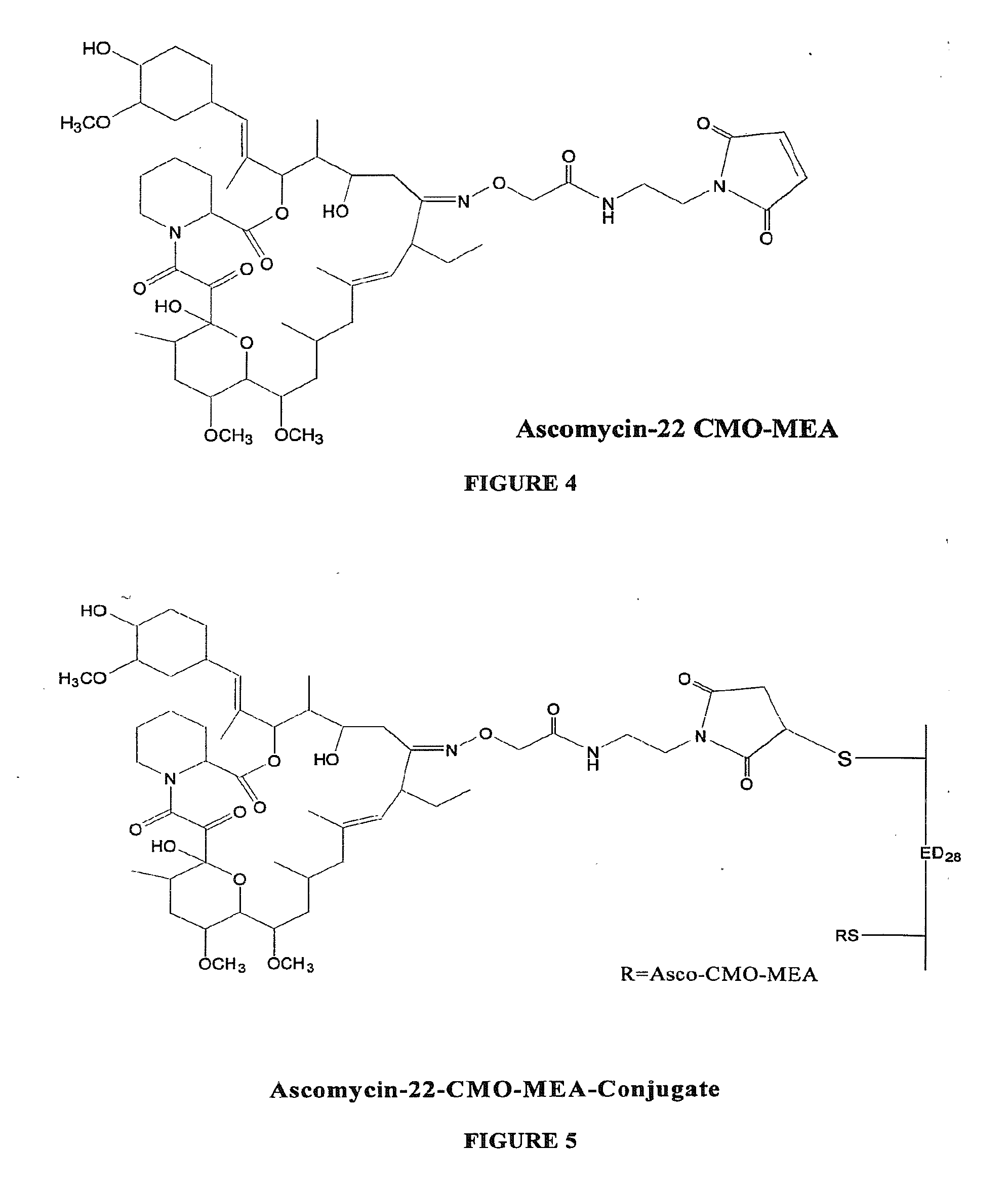
Hapten, immunogens and derivatives of ascomycin useful for preparation of antibodies and immunoassays
The invention teaches derivatives of ascomycin and methods of preparing immunogens and other conjugates useful in immunoassays for quantitatively measuring concentrations of tacrolimus in patient specimens. Antibodies produced from the disclosed immunogens capable of binding to tacrolimus with cross-reactivity of no more than 5% with each of 15-O-demethyl tacrolimus, 31-O-demethyl tacrolimus, and 13,31-O-didemethyl tacrolimus, less than 40% with 13-O-demethyl tacrolimus, and less than 1% with cyclosporin, rapamycin, mycophenolic acid, prednisone, hydrocortisol, and prednisolone are described. Further, immunoassays for measuring the concentration of tacrolimus using such antibodies are taught.
Owner:MICROGENICS CORP
Ocular solutions
InactiveUS20050063997A1Reduce turbidityReduce inflammationBiocideSenses disorderEverolimusOcular structure
Ocular solutions containing at least one macrolide antibiotic and / or mycophenolic acid provide anti-inflammatory, anti-cell proliferation, anti-cell migration, anti-angiogenesis, antimicrobial, and antifungal effects. In one embodiment, the solution is administered intraocularly after cataract surgery before insertion of a replacement intraocular lens, resulting in reduced posterior capsular opacification which may eliminate the need for a subsequent surgery. The solution may be one that is invasively administered, for example, an irrigation or volume replacement solution containing at least one macrolide antibiotic such as tacrolimus, sirolimus, everolimus, cyclosporine, and ascomycin, or mycophenolic acid. The solution may be one that is non-invasively or topically administered in the form of drops, ointments, gels, creams, etc. and may include eye lubricants and contact lens solutions. The solution may contain a supratherapeutic concentration of agent(s) so that a therapeutic concentration of a topically administered solution accumulates in a diseased ocular structure sufficient to treat the disease. The agent(s) may be formulated with polymers or other components for extended or slow release to provide a substantially constant concentration over the course of treatment.
Owner:PEYMAN GHOLAM A DR
Refining method of crude tacrolimus
ActiveCN101712685AEasy to separateAchieve separationIon-exchange process apparatusOrganic chemistryOrganic solventElution
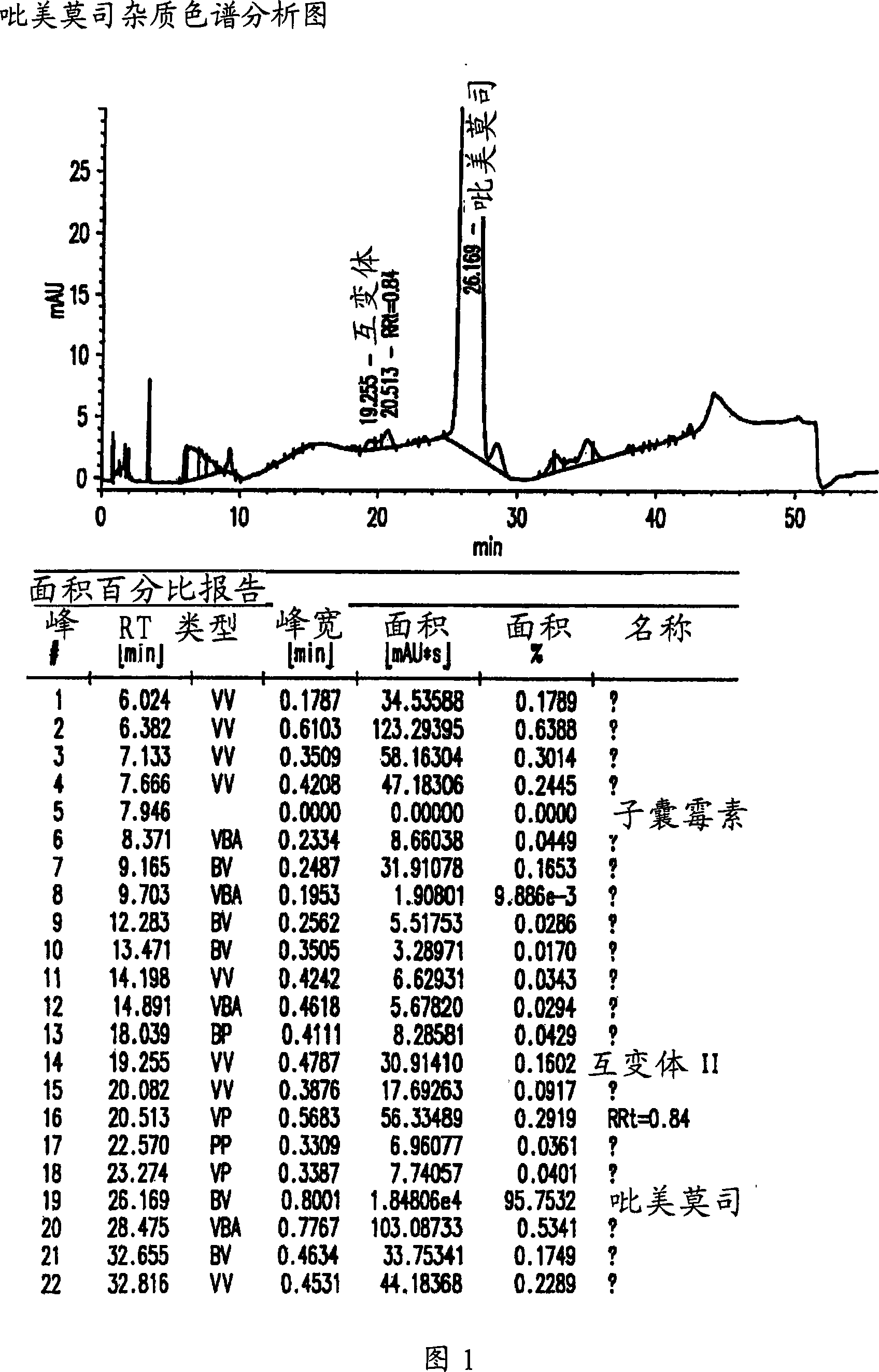
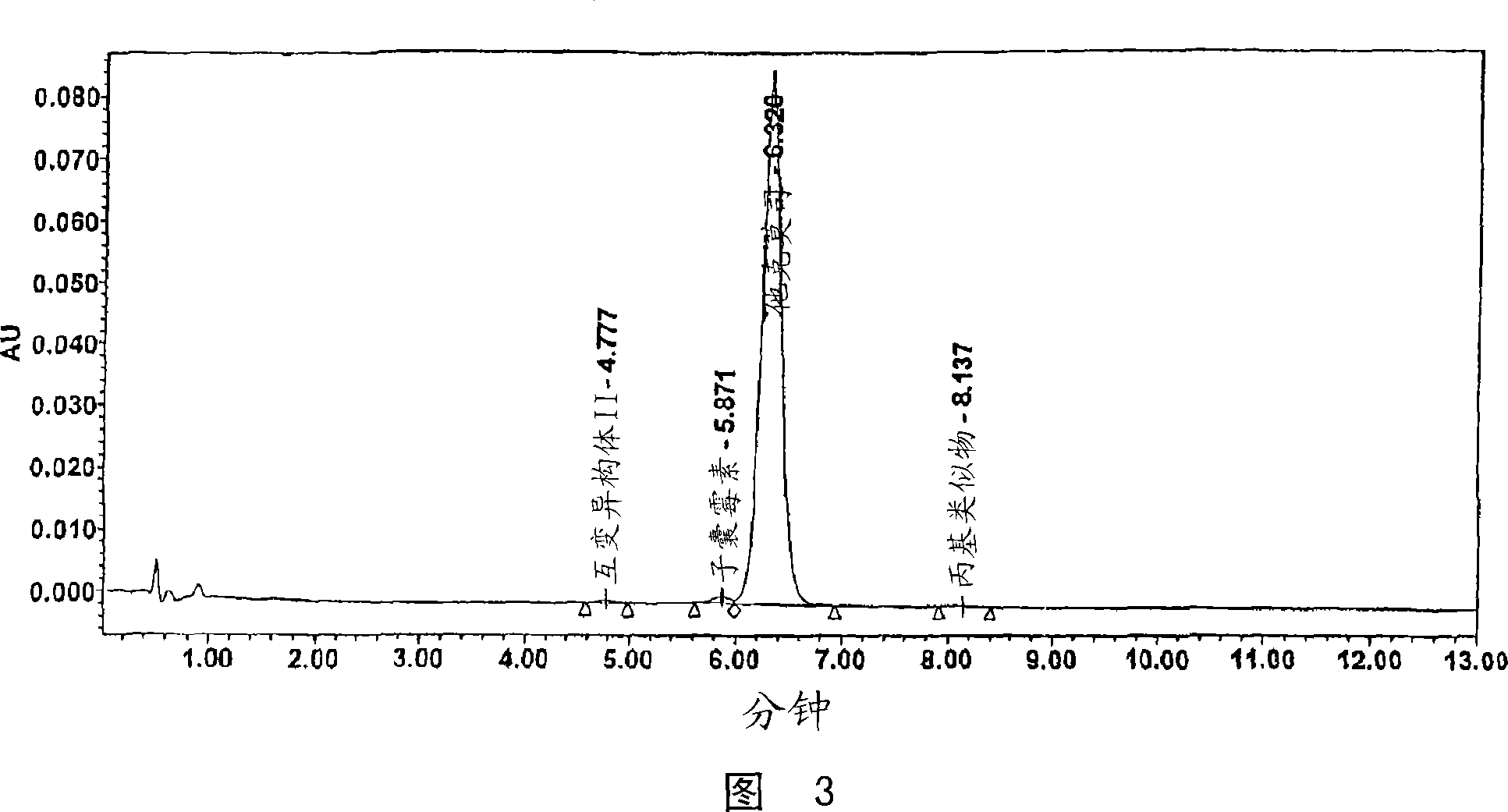
The invention belongs to the technical field of medicinal biotechnology field, specifically relates to a refining method of macrolide powerful immunosuppressant tacrolimus and in particular to a method for refining tacrolimus through reversed-phase high performance liquid preparative chromatography. The method uses crude tacrolimus as raw material to dissolve in organic solvent and adopts C18 reversed-phase preparative liquid chromatography to realize the separation and purification of tacrolimus, ascomycin and dihydro-tacrolimus. The refining method of the invention adopts the proper elution system and has good stability, the impurities can be completely separated under the condition of preparative chromatography, the primary loading quantity can be 200mg, the purity of the obtained tacrolimus can be over 98.5% through HPLC detection; and the whole refining process has simple operation and good universality, is easy to popularize and is suitable for industrial production.
Owner:LUNAN PHARMA GROUP CORPORATION
Processes for preparing water-soluble polyethylene glycol conjugates of macrolide immunosuppressants
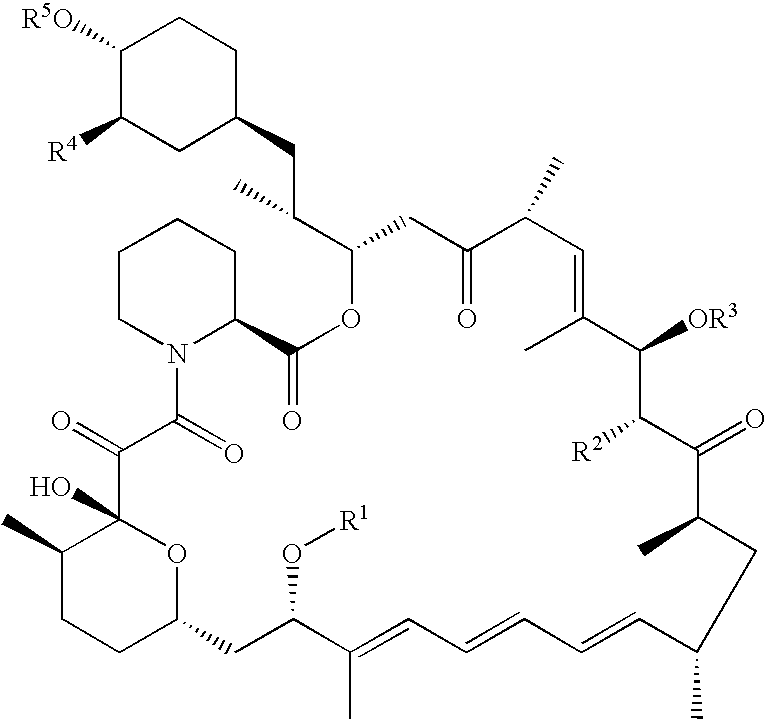
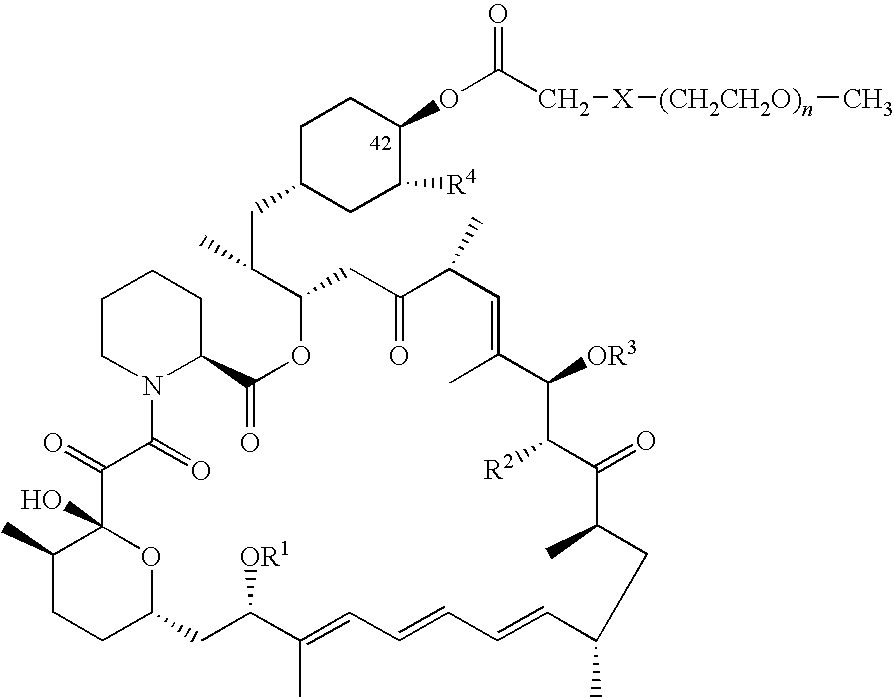
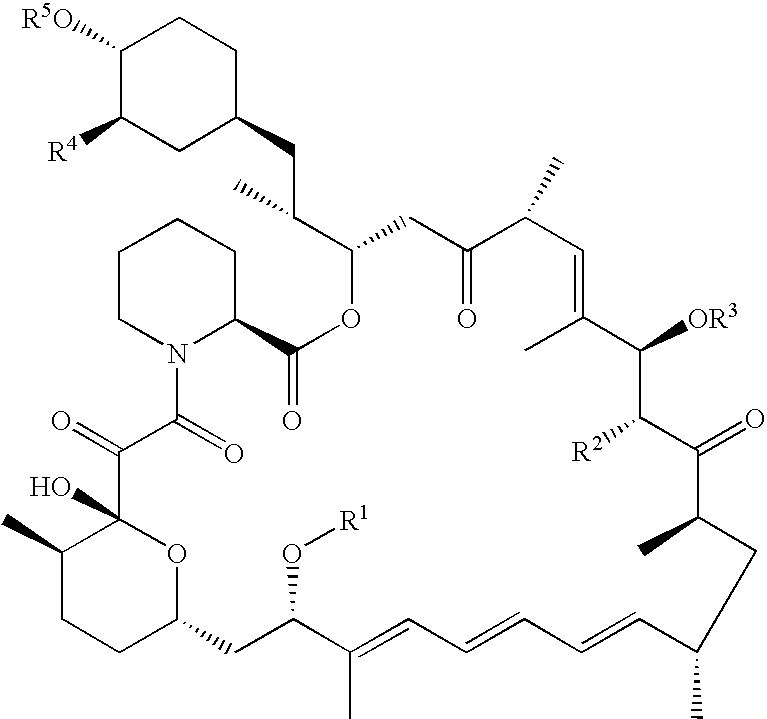
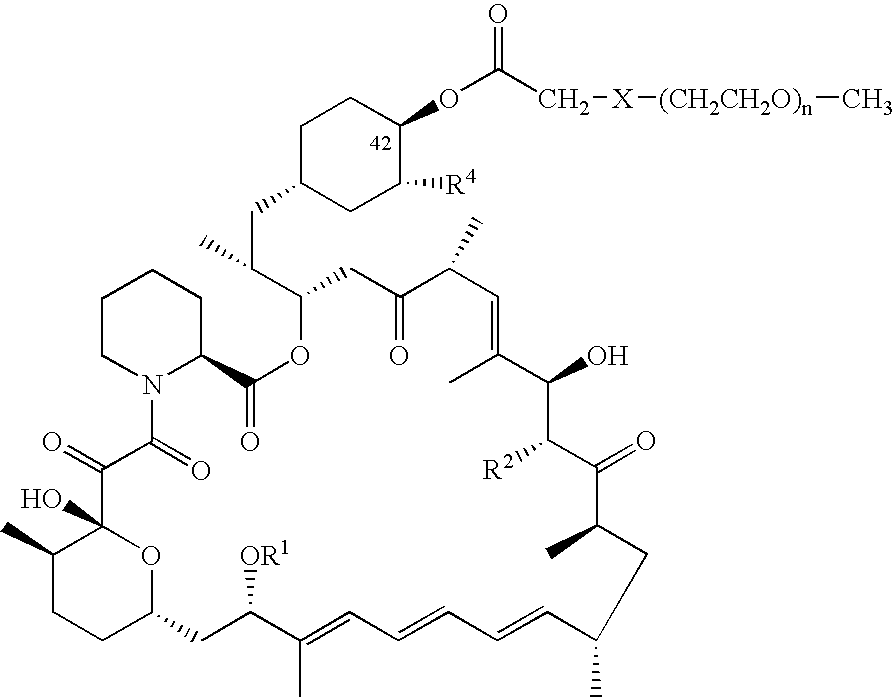
Processes are described for preparing 42-pegylated rapamycins including reacting a rapamycin with an acylating agent in the presence of a lipase to form an acylated rapamycin and reacting the acylated rapamycin with a methoxy poly(ethylene glycol) derivative in the presence of a base. Also described are processes for preparing 32-pegylated tacrolimus and / or ascomycin using these steps.
Owner:WYETH LLC
Methods of preparing pimecrolimus
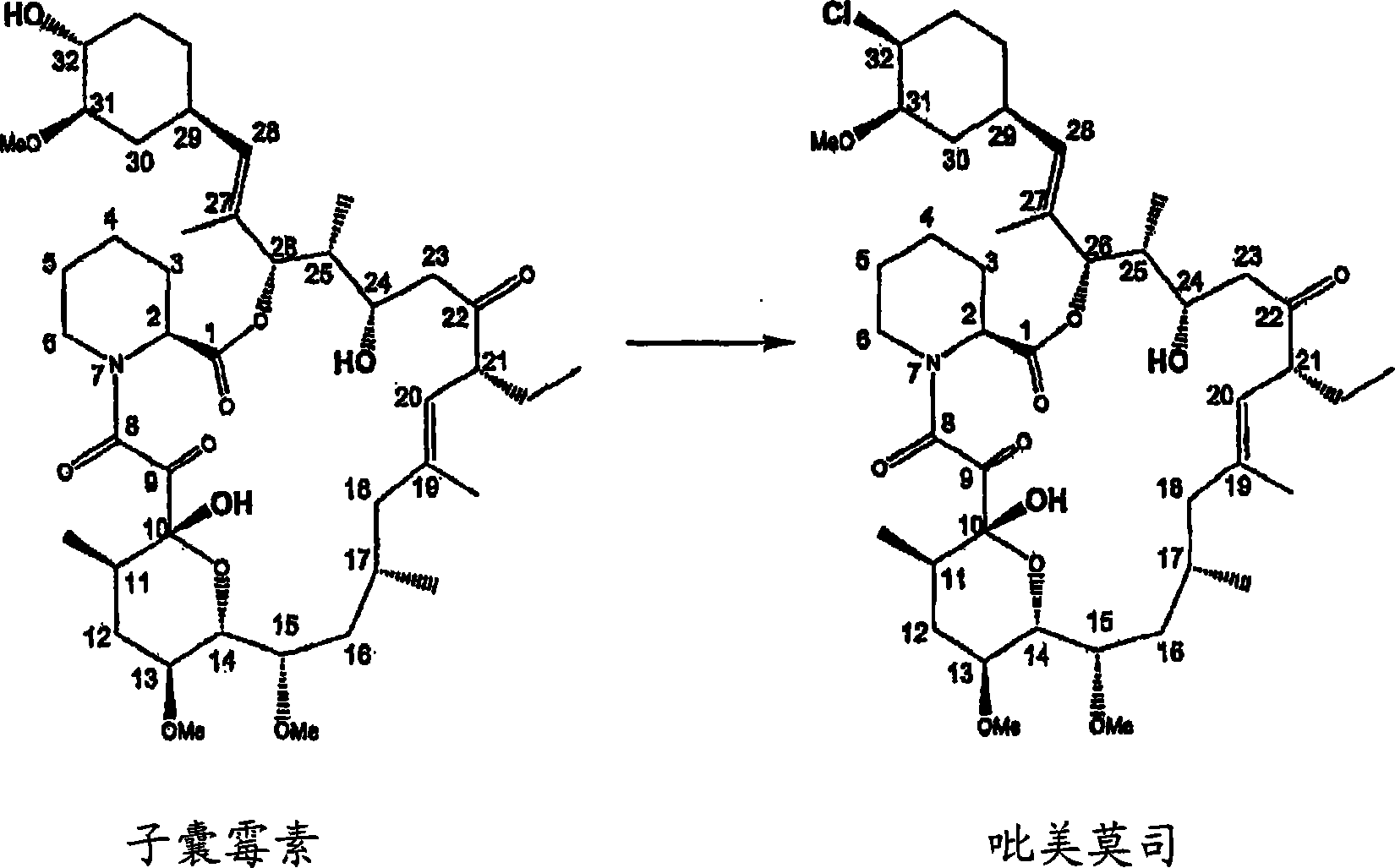
The present invention provides a process for the preparation of pimecrolimus from ascomycin in which ascomycin is reacted with a conversion reagent that converts ascomycin to its activated derivative at C-32. The activated ascomycin is then reacted with chloride ion. The process of the invention requires fewer process steps than prior art processes, and does not require the protection of the ascomycin C-24 hydroxyl group or the purification of the activated ascomycin derivative.
Owner:TEVA GYOGYSZERGYAR RESVENYTARSASAG
Processes for preparing water-soluble polyethylene glycol conjugates of macrolide immunosuppressants
Processes are described for preparing 42-pegylated rapamycins including reacting a rapamycin with an acylating agent in the presence of a lipase to form an acylated rapamycin and reacting the acylated rapamycin with a methoxy poly(ethylene glycol) derivative in the presence of a base. Also described are processes for preparing 32-pegylated tacrolimus and / or ascomycin using these steps.
Owner:WYETH LLC
Method of preparing ascomycin through fermentation
InactiveCN105671102ASlow growth rateRaise the fermentation unitMicroorganism based processesFermentationBiotechnologyStreptomyces hygroscopicus
The invention discloses a method of preparing ascomycin through fermentation with streptomyces hygroscopicus. On the basis of the difference on metabolism between on a fermentation pot and a fermentation bottle of the ascomycin in the prior art, a material supplying process for the fermentation bottle is disclosed, namely, various acids or carbon sources are supplied during the middle stage of fermentation to control the pH of a fermentation liquid, to form similar metabolism status between the fermentation pot and the fermentation bottle, thereby increasing fermentation unit and reducing production cost. The method is simple in operation, is mild in conditions. The raw materials are wide in sources and low in cost. The method is low in demand on equipment and is suitable for industrial production.
Owner:CHONGQING QIANTAI BIOLOGICAL MEDICINE
Method for breeding high-yield ascomycin strain by performing femtosecond laser mutagenesis on streptomyces hygroscopicus ascomycota subspecies and culture medium preparation
InactiveCN102250872ANot suitable for damageShort pulse timeMutant preparationElectrical/wave energy microorganism treatmentStreptomyces hygroscopicusAscomycota
The invention discloses a method for breeding an high-yield ascomycin strain by performing femtosecond laser mutagenesis on streptomyces hygroscopicus ascomycota subspecies and a culture medium preparation method; the method comprises the following steps: at room temperature, mixing mature slant spores of the streptomyces hygroscopicus ascomycota subspecies (ATCC14891) and sterile water to obtain monospore suspension containing 106-107 spores per milliliter; irradiating the spore suspension for 1-10 min by using the titanium sapphire femtosecond laser with centre wavelength of 800 nm, pulse width of 150 fs and frequency of 76 MHz under a irradiation power of 10-30m W; properly diluting the spore suspension, coating on a solid panel to culture, then screening by using a shake flask to obtain a high-yield ascomycin mutation strain. The method provided by the invention has the advantages that the used femtosecond laser mutagenesis method is feasible and safe to operate; the mutation effect is better than that of the traditional physical and chemical mutagenesis method, and the femtosecond laser mutagenesis method has great popularization value in breeding of microorganism pharmaceutical strains; by using the femtosecond laser irradiation to perform the mutagenesis, the high-yield ascomycin mutation strain can be bred. The positive mutation rate of the mutation strain is 5-30%, and fermenting unit is improved by 10-60% in comparison with that of an original strain.
Owner:TIANJIN UNIV
Process for isolation of crystalline tacrolimus
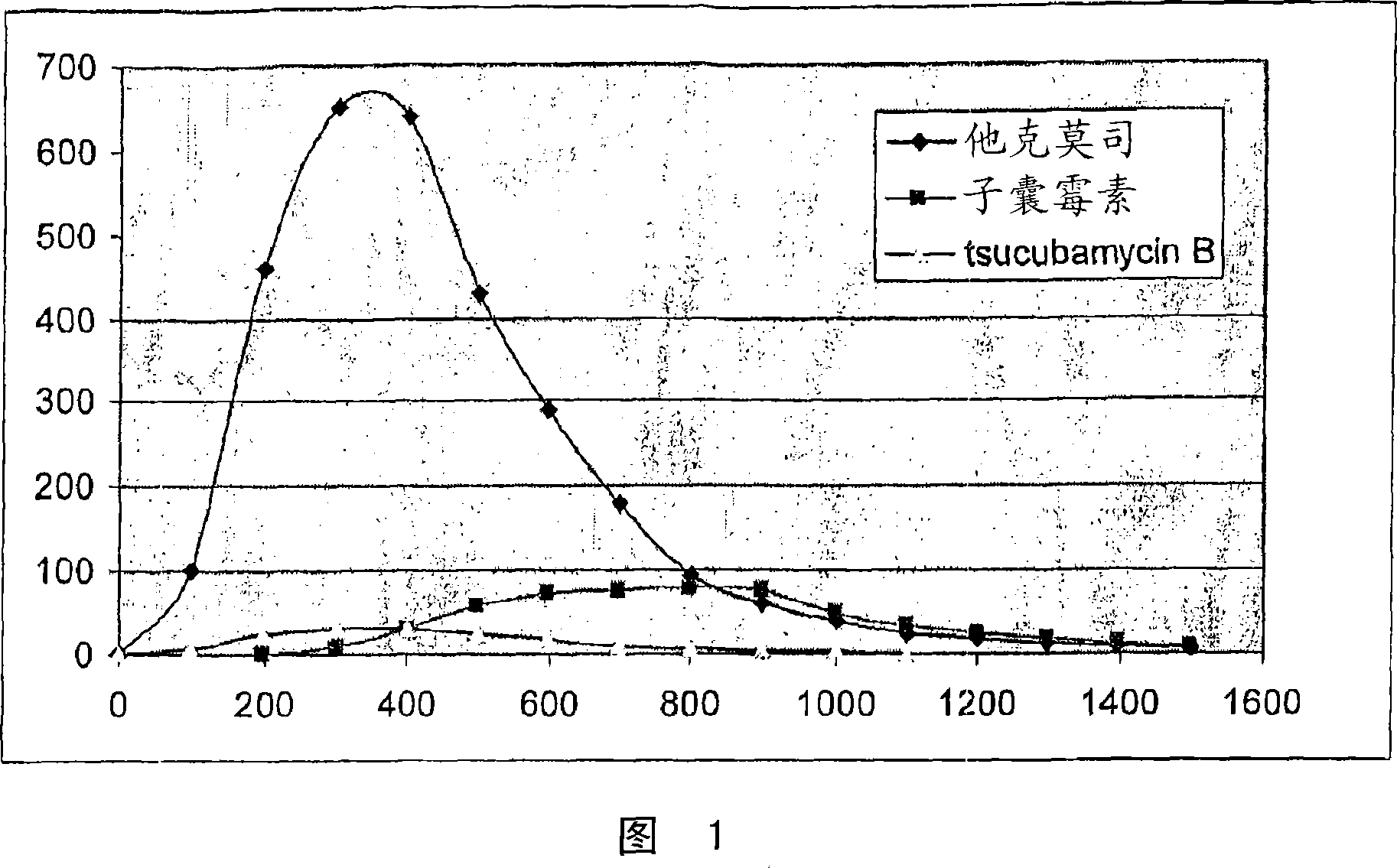
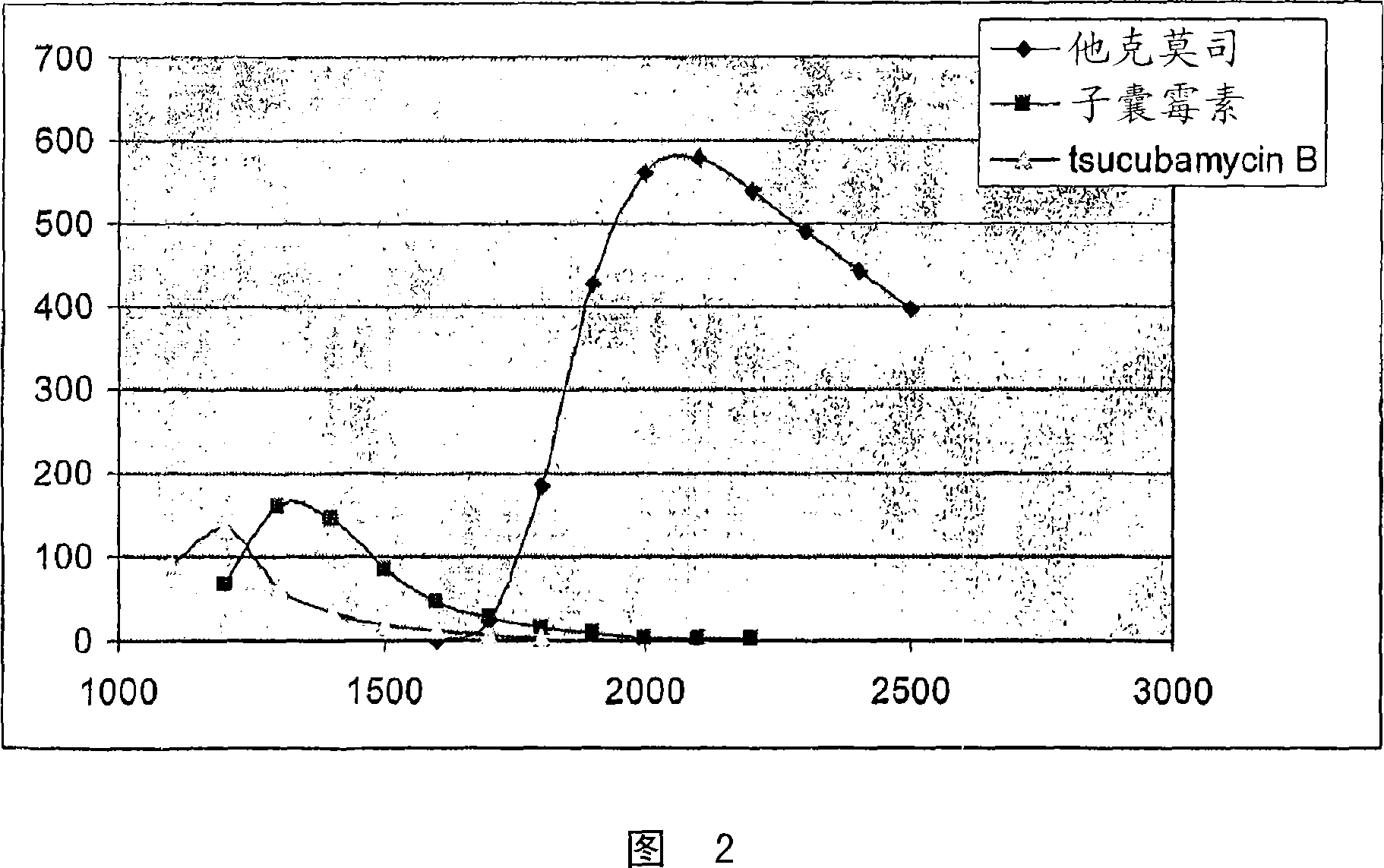
The invention is a simple process for separation of tacrolimus and its analogues, ascomycin and tsucubamycin B and preparation of enough pure crystalline tacrolimus. The process takes advantage of surprising properties of tacrolimus and involves extraction, purification and crystallization to produce purified crystalline tacrolimus.
Owner:IVAX PHARMA
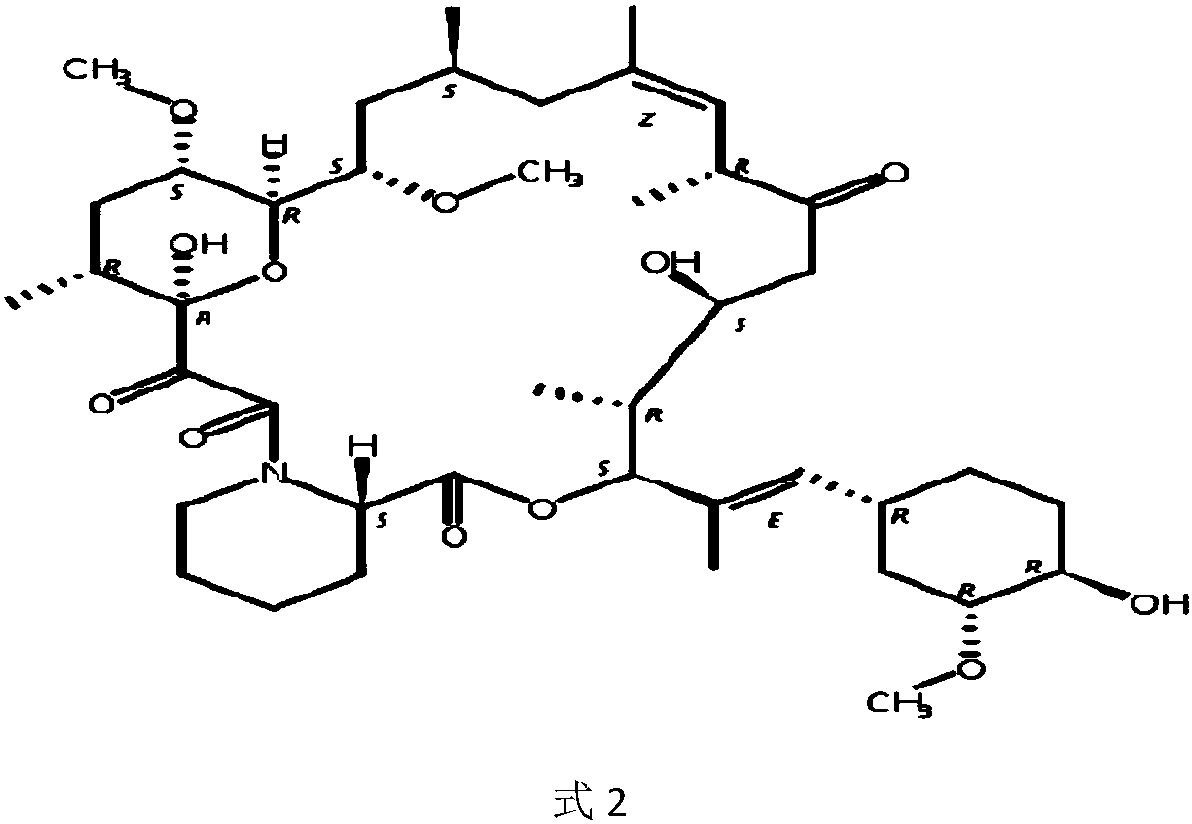
Ascomycin and pimecrolimus having reduced levels of desmethylascomycin and 32-deoxy-32-epichloro-desmethylascomycin respectively, and methods for preparation thereof
Owner:TEVA PHARM USA INC
Method for purification of ascomycin from streptomyces fermentation liquid
Belonging to the field of bioengineering, the invention discloses a method for purification of ascomycin from a streptomyces fermentation liquid. The method adopts the techniques of fermentation liquid filtration, mycelium ultrasonication, macroporous resin adsorption, silicagel column chromatography and crystallization so as to obtain pure ascomycin. By employing ultrasonication, the method of the invention has advantages of substantially improved extraction efficiency, convenient operation, simple process, low cost, and high efficiency, thus being suitable for industrial production.
Owner:NANJING UNIV OF TECH
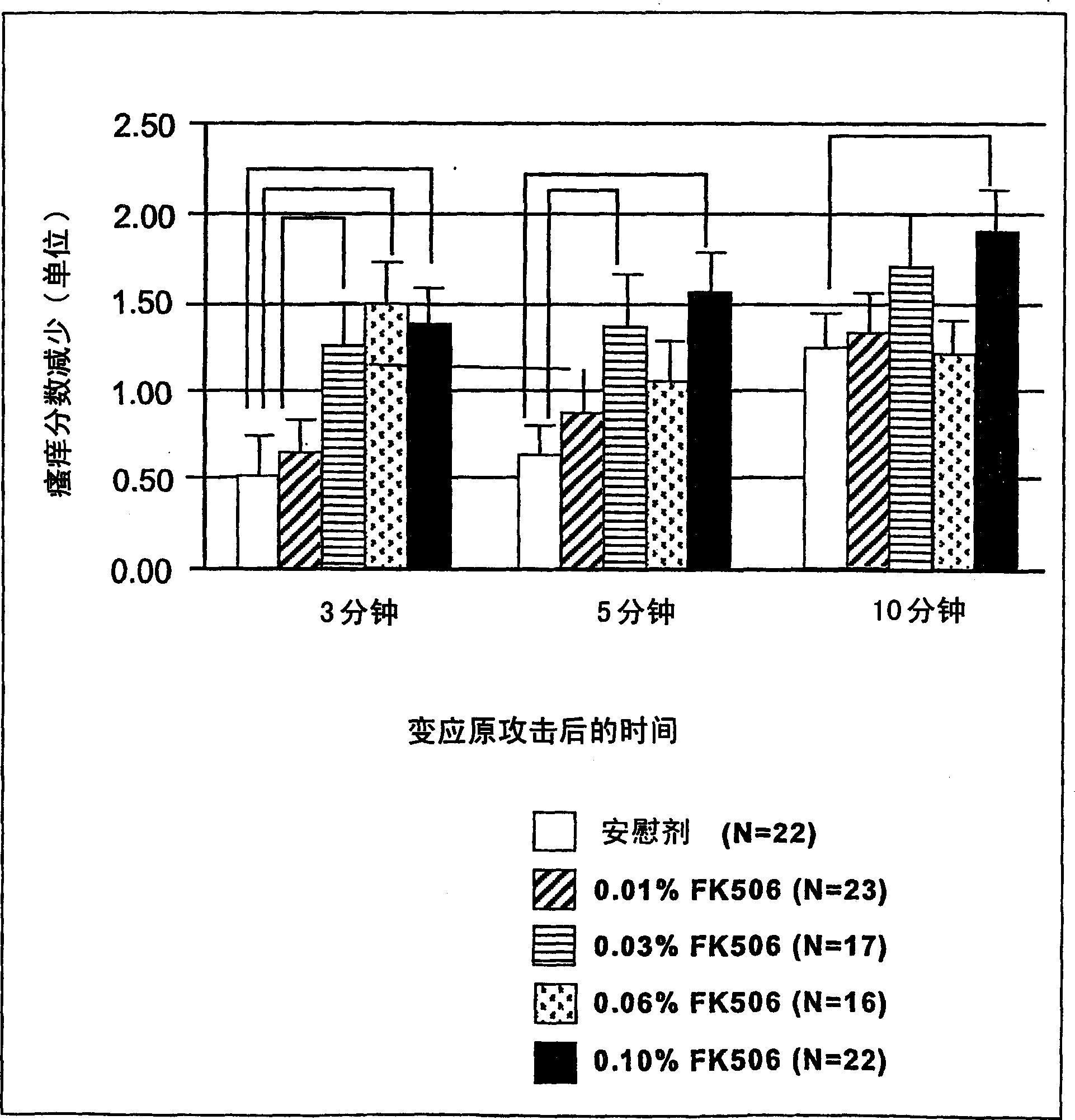
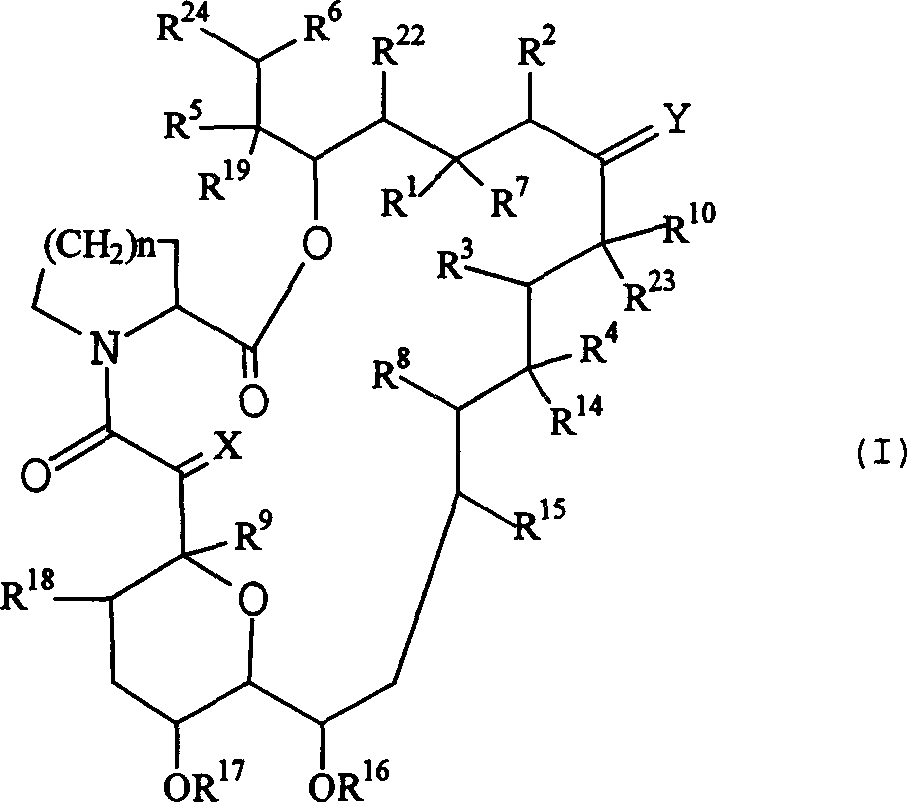
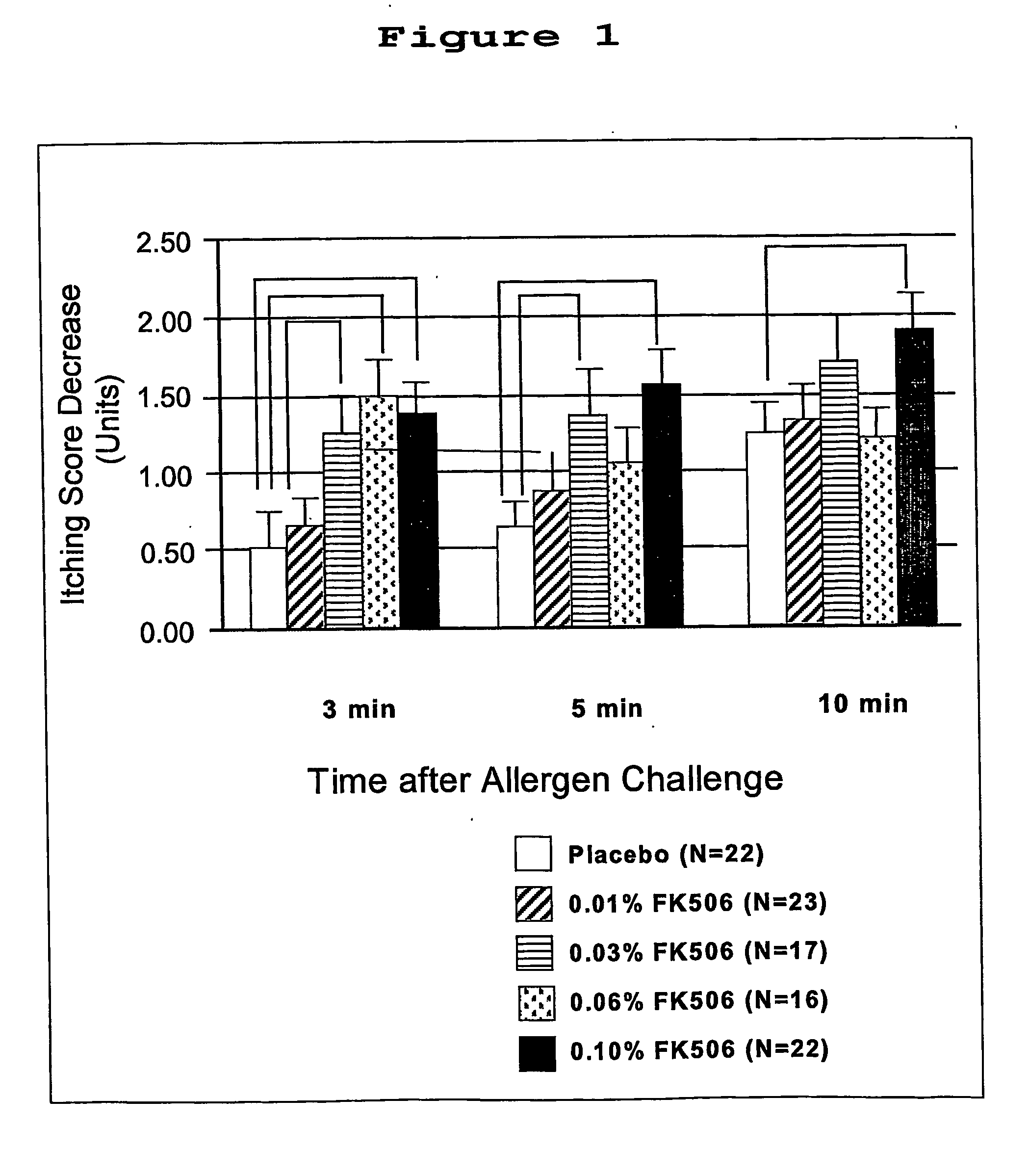
Pharmaceutical compositions comprising FK506 derivatives and the ir use for the treatment of allergic diseases
The invention provides ophthalmic compositions and methods treating the symptoms of ocular allergies. The principle active ingredient in these compositions and methods is macrolide compound, such as tacrolimus, ascomycin and rapamycin and their derivatives. Optimal concentrations and dosing regimens are provided.
Owner:SUCAMPO PHARMA
Pharmaceutical compositions comprising fk506 derivatives and their use for the treatment of allergic diseases
The invention provides ophthalmic compositions and methods treating the symptoms of ocular allergies. The principle active ingredient in these compositions and methods is macrolide compound, such as tacrolimus, ascomycin and rapamycin and their derivatives. Optimal concentrations and dosing regimens are provided.
Owner:SUCAMPO PHARMA
Hapten, Immunogens and Derivatives of Ascomycin Useful for Preparation of Antibodies and Immunoassays
The invention teaches derivatives of ascomycin and methods of preparing immunogens and other conjugates useful in immunoassays for quantitatively measuring concentrations of tacrolimus in patient specimens. Antibodies produced from the disclosed immunogens capable of binding to tacrolimus with cross-reactivity of no more than 5% with each of 15-O-demethyl tacrolimus, 31-O-demethyl tacrolimus, and 13,31-O-didemethyl tacrolimus, less than 40% with 13-O-demethyl tacrolimus, and less than 1% with cyclosporin, rapamycin, mycophenolic acid, prednisone, hydrocortisol, and prednisolone are described. Further, immunoassays for measuring the concentration of tacrolimus using such antibodies are taught.
Owner:MICROGENICS CORP
Streptomyces hygroscopicus mutant strain for highly yielding ascomycin, and application thereof
ActiveCN107245460AIncrease productionStable potencyBacteriaMicroorganism based processesStreptomyces hygroscopicusIndustrial fermentation
The invention relates to a strain obtained through adopting Streptomyces hygroscopicus FIM-311 as an original strain and screening using a normal pressure and room temperature plasma mutation technology. The strain is Streptomyces hygroscopicus FIM-311-80, can highly yield the ascomycin through fermentation, greatly increases the output of the ascomycin, and can be applied to industrial fermentation production; and the strain FIM-311-80 has a good stability, the ascomycin production tilter of continuously passed four generations is basically stable and keeps the same high level, and the strain FIM-311-80 can be used as a production strain used for further researches and development.
Owner:FUJIAN INST OF MICROBIOLOGY
Method for isolating and purifying ascomycin from streptomyces fermentation broth
The invention provides a method for separating and purifying ascomycin from streptomyces fermentation broth. The method comprises the following steps: filter-pressing the streptomyces fermentation broth with a plate frame to obtain thalli, extracting the thalli with an organic solvent at room temperature for multiple times, combining extracting solutions, concentrating the extracting solution and then transferring to macroporous resin and silica gel columns for chromatography, concentrating and crystallizing the obtained ascomycin with a mixed solvent, transferring the crystallized crude product to a dynamic axial high-pressure chromatographic column for isolation, and then concentrating and crystallizing the obtained ascomycin with the mixed solvent. In the invention, the prepared ascomycin has the purity of 98% or more and the yield of 80 to 90%. The method for separating and purifying ascomycin from the streptomyces fermentation broth, provided by the invention, has the advantages of greatly shortening the extraction time and improving the efficiency, having convenience in operation, simple process, low cost, easiness in industrial production and the like, and providing a feasible isolation process for the production of ascomycin.
Owner:WUXI FORTUNE PHARMA
Method for one-step refining of tacrolimus, dihydrotacrolimus and ascomycin in crude tacrolimus
The invention relates to a method for one-step refining of tacrolimus, dihydrotacrolimus and ascomycin in crude tacrolimus. The method comprises the following steps: developing the crude tacrolimus inthe head of a stainless steel chromatographic column filled with a special packing material, performing chromatographic elution with an eluent containing a solvent and water according to a ratio of 60:40, having a pH value, adjusted with an acid, of 4.5 and also containing 5% of a developer, and performing distribution collection, pure product concentration, crystallization and recrystallizationto obtain pure tacrolimus, pure dihydrotacrolimus and pure ascomycin. The HPLC purity of the tacrolimus, dihydrotacrolimus and ascomycin which are obtained through the method is more than 98%. The method has the advantages of convenience in operation, simple process and low cost, can be used in pilot production, and is very suitable for industrial production.
Owner:TIANJIN LINGSHI BIOTECH DEV CO LTD
Purification method for ascomycin
InactiveCN108409751AEfficient separationQuality assuranceOrganic chemistry methodsAlcoholPurification methods
The invention discloses a purification method for ascomycin. The method comprises the following steps: gathering ascomycin loading solution by using a macroreticular resin, collecting eluant and concentrating, adding an extraction agent to concentrated solution and extracting, and concentrating an organic phase to be dry powder; and dissolving the dry powder to be clear methyl alcohol or acetonitrile solution, and preparing a pure ascomycin product in high purity by using a liquid chromatograph. The preparation technology has the good removal effect to a key impurity FK523 in the ascomycin, and the prepared product has the high purity.
Owner:HISUN PHARMACEUTICAL (HANGZHOU) CO LTD +1
A kind of bactericidal composition containing picoxystrobin
ActiveCN108294004BReduce pollutionReduce the amount of applicationBiocideFungicidesBiotechnologyFungal disease
The invention relates to a bactericidal composition. Active components of the composition contain picoxystrobin and ascomycin, wherein the weight ratio of the picoxystrobin to the ascomycin ranges from (1 to 1) to (1 to 100). The invention further relates to a method for preventing and controlling agricultural fungal diseases by utilizing the composition and application of the composition.
Owner:上海永阔生物医药科技有限公司
Method for promoting microbes to synthesize ascomycin by using exogenous additive and preparation of seed liquor and culture medium
InactiveCN102250982BIncrease synthesisImprove conversion rateMicroorganism based processesFermentationBiotechnologyStreptomyces hygroscopicus
The invention discloses a method for promoting microbes to synthesize ascomycin by using an exogenous additive and a preparation of a culture medium, and belongs to the technical field of biologic synthesis of ascomycins. The method is characterized in that, on the basis of the characteristic that the ascomycin biologic synthesis process refers to the assembly of various precursors, at the beginning of the aerobic fermentation or in the process of aerobic fermentation, 0.5-3.0 g of shikimic acid, 0.5-3.0 g of isoleucine and 1-5 g of soybean oil are added in every one liter of a culture mediumfor enhancing the accumulation of ascomycin synthesis precursors DHCHC, malonyl and methyl malonyl, promoting the metabolism of the ascomycin synthesis pathway, thereby improving the yield of the ascomycin. The method provided by the invention is simple to operate and is low in cost because no extra device and labor is increased and only low additional investment is input; in the step of fermenting and culturing streptomyces hygroscopicus ascomycota subspecies, the fermenting unit of the ascomycin is improved by 55.9-114.7% as compared with a control group.
Owner:TIANJIN UNIV
Topical compositions comprising ascomycins
InactiveCN1213767CFew ingredientsEasy to manufactureAntipyreticAnalgesicsChemical compositionDermatology
Owner:迈德制药卢森堡有限公司
Method for increasing yield of ascomycin by using polyhydroxybutyrate as intracellular carbon library
ActiveCN112301072AIncrease productionSolve the problem of insufficient supply of carbon sourcesMicroorganism based processesEnzymesBiotechnologyStreptomyces hygroscopicus
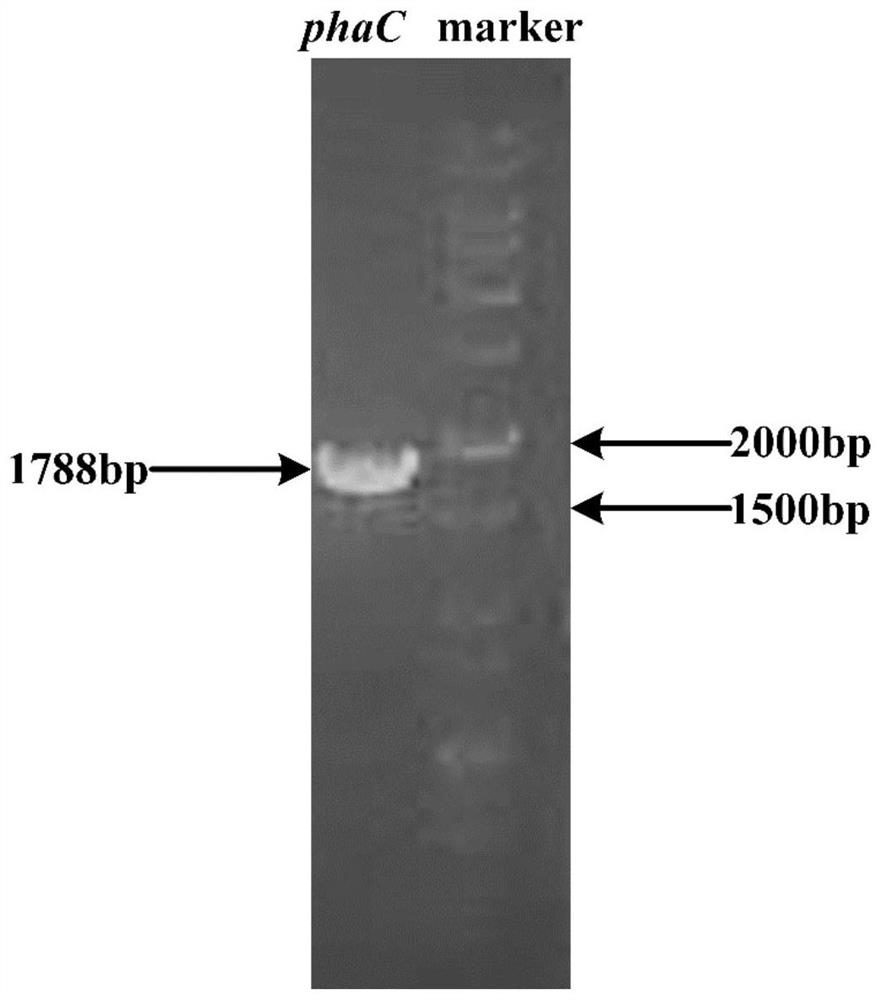
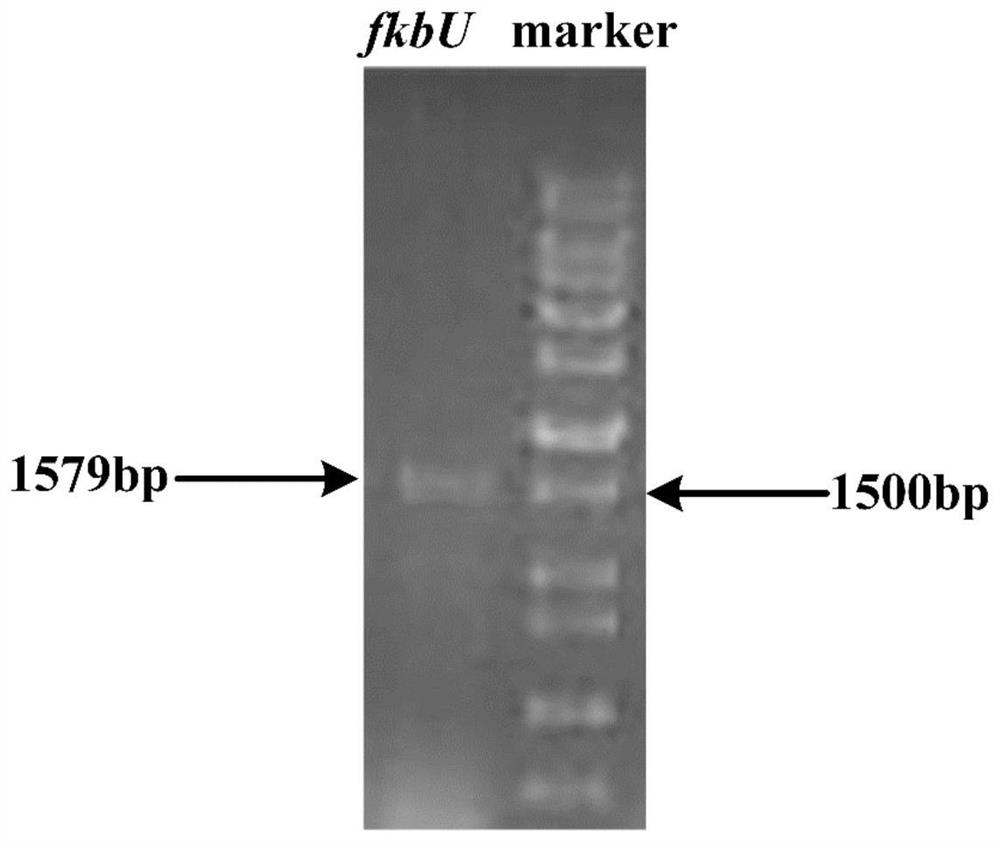
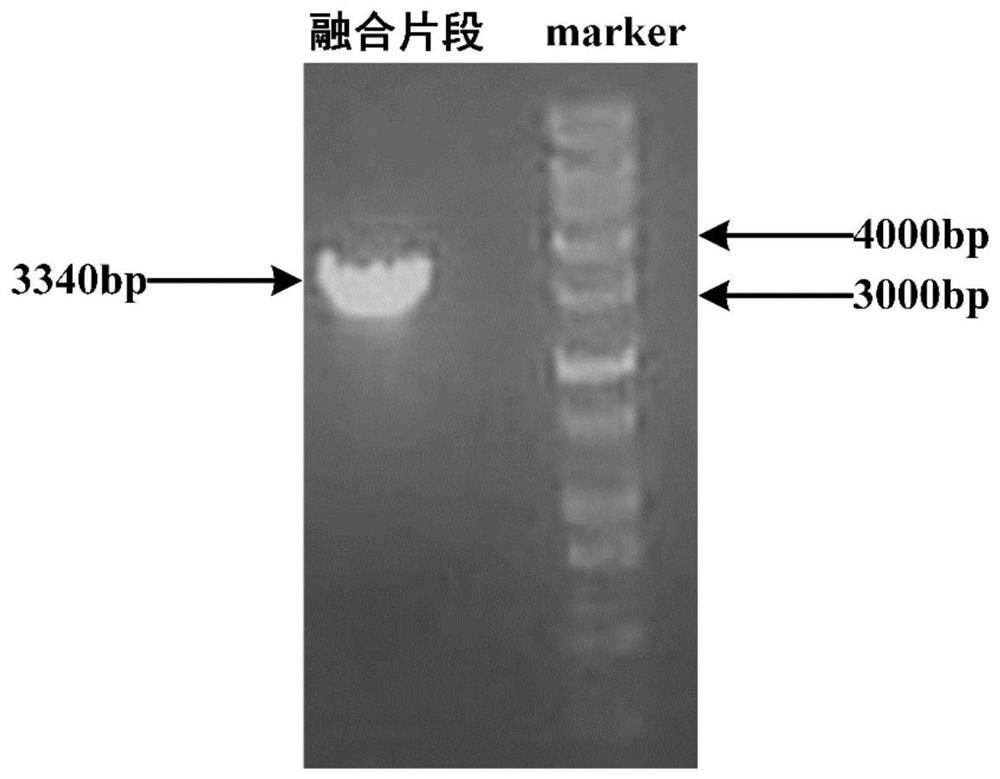
The invention provides a method for increasing the yield of ascomycin by using polyhydroxybutyrate as an intracellular carbon library. A polyhydroxybutyrate synthetic gene phaC from ralstonia eutrophaand a polyhydroxybutyrate decomposition gene fkbU from streptomyces hygroscopicus ascus subspecies are integrated in series to a genome of an original strain Streptomyces hygroscopicus var.ascomyceticus ATCC14891 by using a genetic engineering technology, and continuous production of ascomycin is promoted by exerting the effects of polyhydroxybutyrate on storage, degradation and reuse of a carbonsource, so that the fermentation yield of the ascomycin in the genetic engineering strain reaches 626.30 mg / L, which is increased by 211% compared with the original strain ATCC14891. The method provided by the invention provides a new strategy for high yield of the ascomycin, and the method has potential application value in industrial production of FK520.
Owner:TIANJIN UNIV