Patents
Literature
203 results about "Cell killing" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Method of treatment using ligand-immunogen conjugates
InactiveUS7033594B2Improve recognitionEnhance endogenous immune response-mediated eliminationAntibacterial agentsBiocideBinding siteCytotoxicity
A method and pharmaceutical composition are provided for enhancing the endogenous immune response-mediated elimination of a population of pathogenic cells in a host animal wherein the pathogenic cells preferentially express, uniquely express, or overexpress a binding site for a particular ligand. The invention comprises administering the ligand conjugated to an immunogen to a host animal harboring the population of pathogenic cells. Antibodies, preexisting or administered to the host animal to establish a passive immunity, directed against the immunogen bind to the ligand-immunogen conjugate resulting in elimination of the pathogenic cells by the host's immune response. At least one additional therapeutic factor is administered selected from the group consisting of a cell killing agent, a tumor penetration enhancer, a chemotherapeutic agent, antimicrobial agent, a cytotoxic immune cell, and a compound capable of stimulating an endogenous immune response wherein the compound does not bind to the ligand-immunogen conjugate.
Owner:PURDUE RES FOUND INC
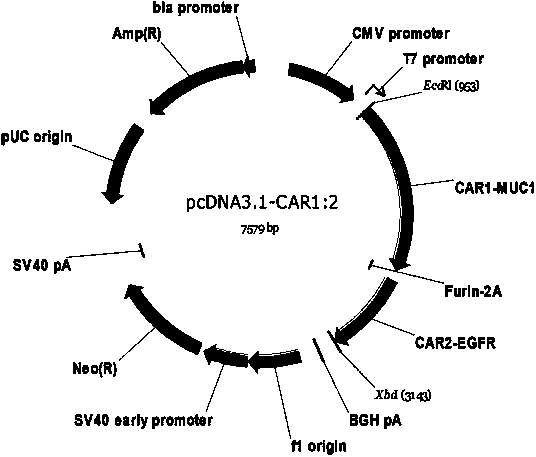
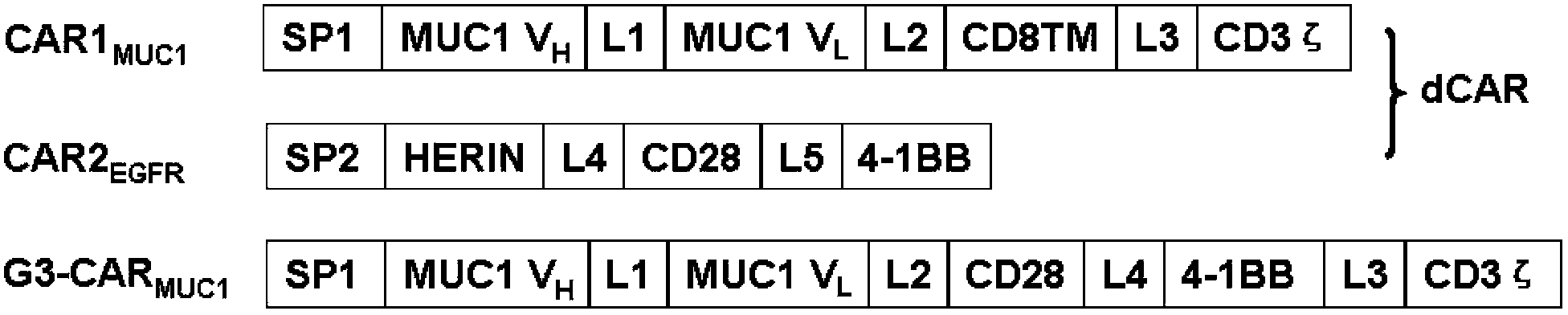
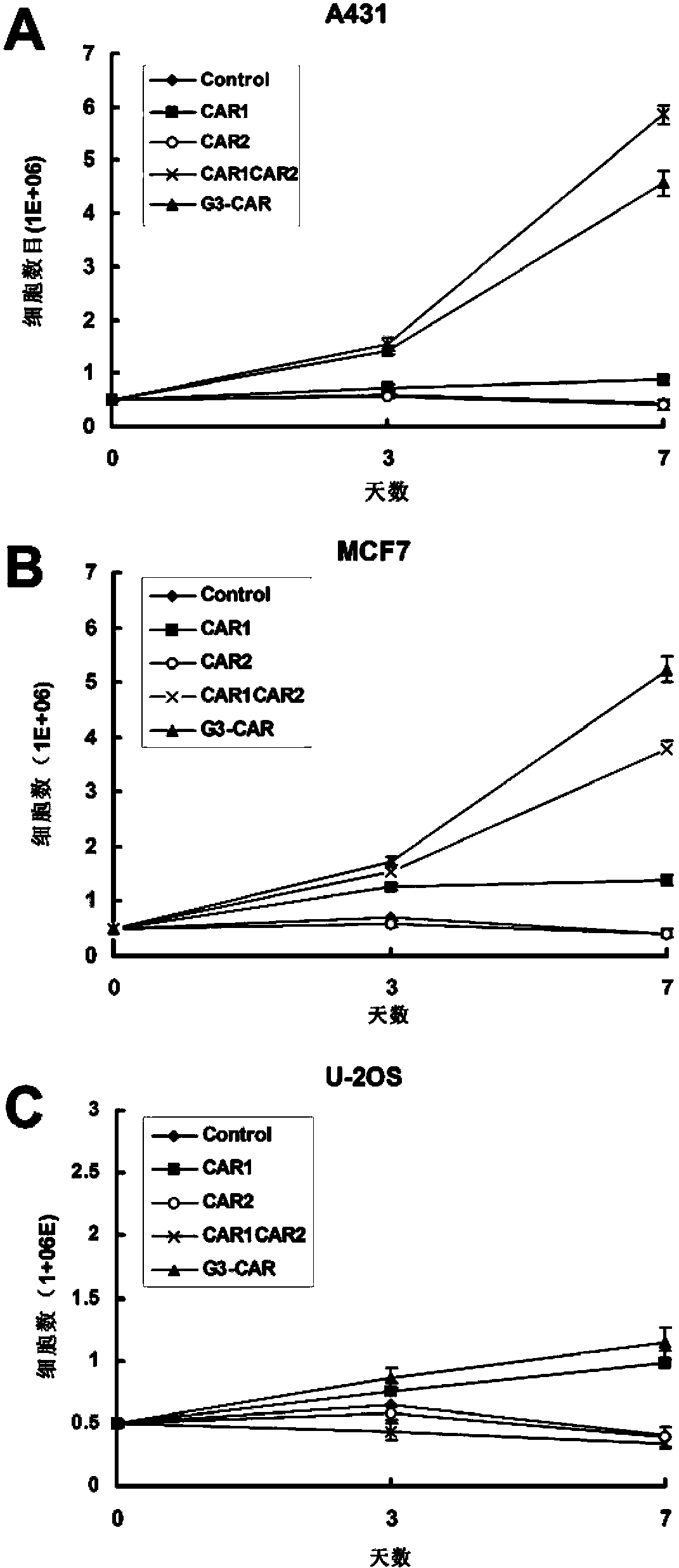
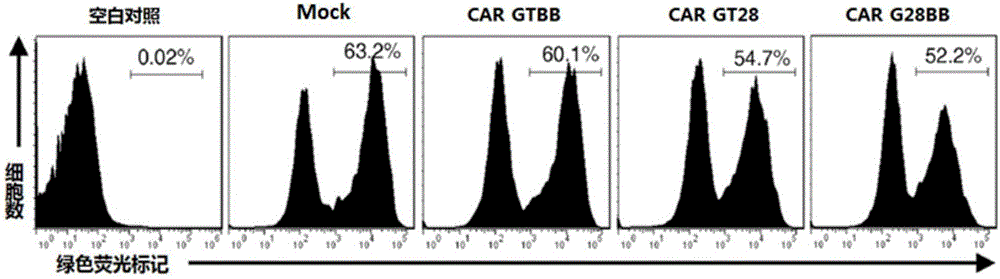
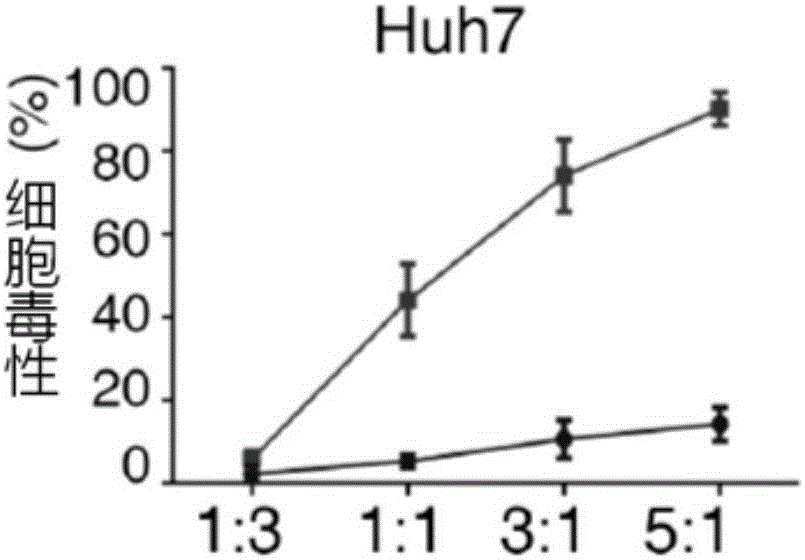
Chimeric antigen receptor combining EGFR (epidermal growth factor receptor) family proteins and composition and uses thereof
ActiveCN103483453AFix security issuesImprove proliferative abilityPeptide/protein ingredientsGenetic material ingredientsEpidermal Growth Factor Receptor KinaseAntigen
The present invention belongs to the fields of molecular biology and immunology and relates to a chimeric antigen receptor combining EGFR (epidermal growth factor receptor) family proteins and a composition and uses thereof The present invention particularly relates to the chimeric antigen receptor comprising polypeptides efficiently combining the EGFR family proteins and transmembrane domains, wherein the polypeptides efficiently combining EGFR family proteins comprise HERIN. The chimeric antigen receptor can promote the proliferation ability and killing effect of T cell at the premise of maintaining the killing specificity of the T cell.
Owner:SHANGHAI CELL THERAPY GRP CO LTD
Dual-signal independent chimeric antigen receptors (dsCAR) and uses thereof
ActiveCN103483452APromote proliferationHigh activityPeptide/protein ingredientsAntibody mimetics/scaffoldsAntigen receptorsViral infectious disease
The invention relates to chimeric antigen receptors (CAR), particularly relates to dual-signal independent chimeric antigen receptors (dsCAR), and also relates to immune response cells of the dual-signal independent chimeric antigen receptors (dsCAR) and uses of the immune response cells in preparation of drugs for treatment of malignant tumor and virus infected diseases. In detail, the dual-signal independent chimeric antigen receptors (dsCAR) can respectively identify two different family antigens of tumor cells and can respectively transmit two T-cell-activation related signals. One of the CAR can transmit a first T-cell-activation related signal by combing a ligand of a tumor specific antigen or a tumor-associated antigen to decide T-cell killing specificity, and the other CAR can transmit a second T-cell-activation related signal by combing a ligand of a membrane receptor (such as EGFR (epidermal growth factor receptor) family protein) widely expressed by the tumor cells to promote T cell activation, proliferation and survival. The dual-signal independent chimeric antigen receptors (dsCAR) can avoid the potential safety problems on the basis of maintaining curative effects of second generation and third generation CAR.
Owner:SHANGHAI CELL THERAPY GRP CO LTD
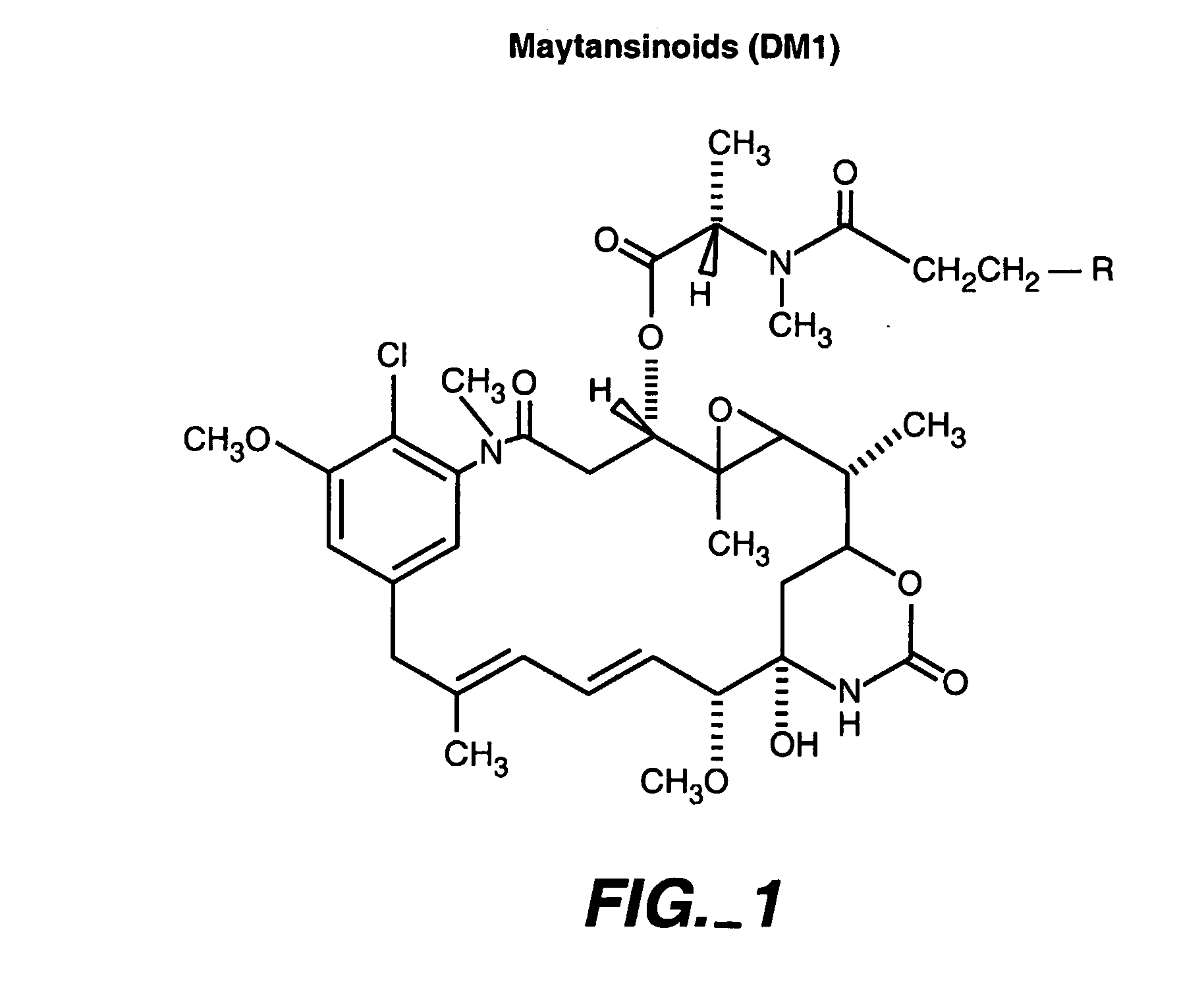
Methods for the identification of polypeptide antigens associated with disorders involving aberrant cell proliferation and compositions useful for the treatment of such disorders
Methods and compositions for the development of effective cancer therapies using mitotic inhibitors which have limited general toxicity to normal, non-cancerous cells and tissues are provided. The methods and compositions utilize cytotoxic compounds comprised of a cell-binding agent (e.g., antibodies) conjugated to an anti-mitotic compound (e.g., maytansinoids). The invention further provides antibodies which are substantially incapable of inducing antibody-dependent cell-mediated cytotoxicity (ADCC) and / or complement dependent cytotoxicity (CDC), thereby ensuring that the therapeutic effect is mediated primarily by the anti-mitotic component of the cytotoxic compound, rather than by indirect cell killing via ADCC and / or CDC. The antibodies of the invention further are capable of differentiating between polypeptide antigens which are more highly expressed on proliferating cancer cells as compared to proliferating non-cancer cells.
Owner:GENENTECH INC
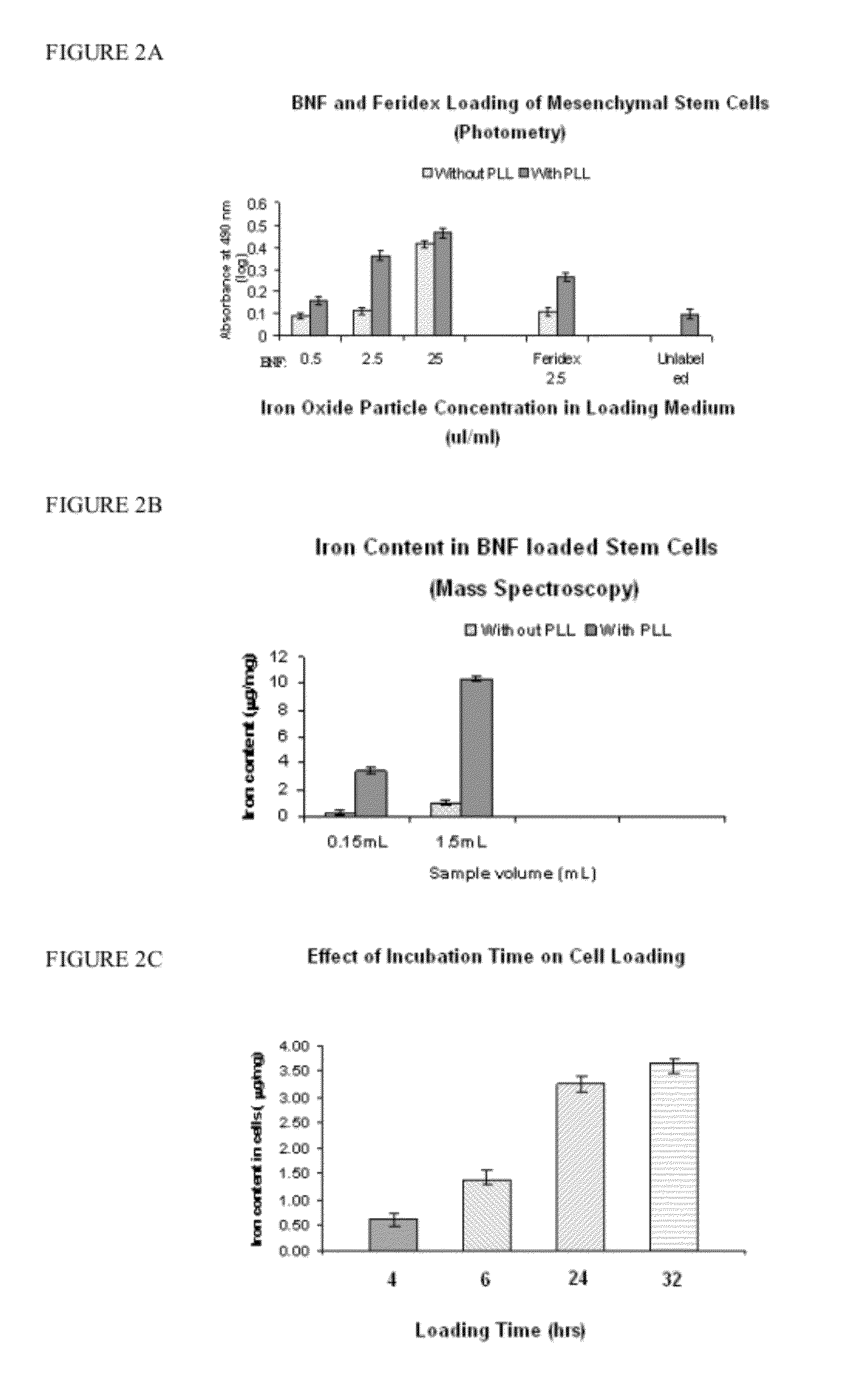
Nanoparticle loaded stem cells and their use in MRI guided hyperthermia
InactiveUS20120283503A1Enhanced MR propertyEnough timeElectrotherapyMedical devicesMri guidedMagnetite Nanoparticles
The present invention provides stem cells loaded with bi-functional magnetic nanoparticles (nanoparticle-loaded stem cells (NLSC)) that both: a) heat in an alternating magnetic field (AMF); and b) provide MRI contrast enhancement for MR-guided hyperthermia. The nanoparticles in the NLSC are non-toxic, and do not alter stem cell proliferation and differentiation, the nanoparticles do however, become heated in an alternating magnetic field, enabling therapeutic applications for cancer treatment. NLSC can deliver hyperthermia to hypoxic areas in tumors for sensitization of those areas to subsequent treatment, thus delivering therapy to the most treatment-resistant tumor regions. The heating of diseased tissue either results in direct cell killing or makes the tumor more susceptible to radio- and / or chemotherapy. The NLSC of the present invention can be used for MR image-guided hyperthermia in oncology, in stem cell research for cell tracking and heating, and for elimination of mis-injected stem cells.
Owner:OSTROVSKA LYUBOV PHD
Reduction of adverse inflammation
InactiveUS20050025804A1Reduce the possibilityReduction of the flux of nutrientsBiocideNervous disorderPeroxynitriteIsomerization
Reduction of the likelihood of adverse inflammatory reaction to an implant or a transplant is achieved through several mechanisms including the catalysis of isomerization of peroxynitrite by a hydrogel-bound peroxynitrite isomerization catalysts. A second mechanism controls acceptable and unacceptable dimensions of surface features of implants, such as vascular stents. A third mechanism fabricates implants from materials which are substantially free from alloys transition metals which produce ions of which catalyze cell killing radical formation.
Owner:HELLER ADAM
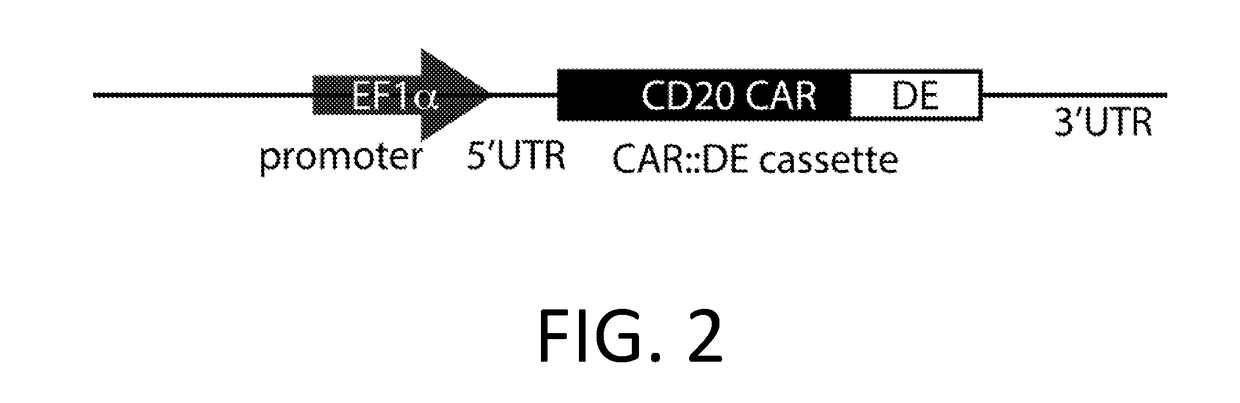
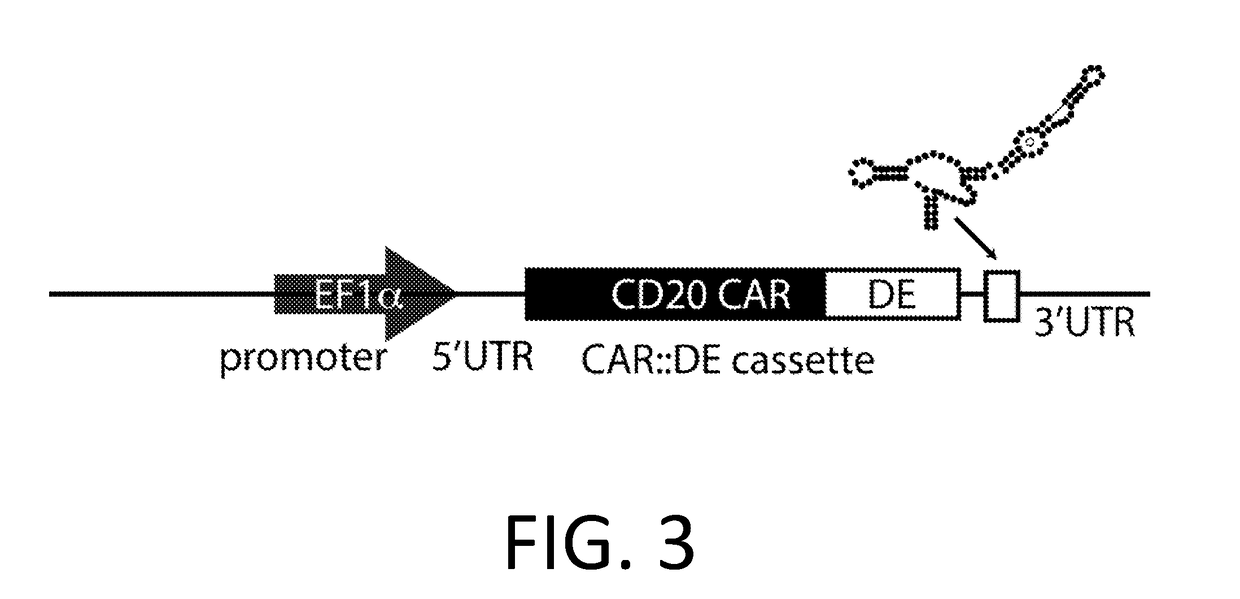
Smart CAR Devices and DE CAR Polypeptides for Treating Disease and Methods for Enhancing Immune Responses
ActiveUS20170157176A1Increase killIncrease the number of grainsPolypeptide with localisation/targeting motifAntibody mimetics/scaffoldsDiseaseApoptosis
In an aspect, the present invention relates generally to the field of treating disease with CAR devices, Smart CAR devices, DE CAR devices, and / or Smart-DE CAR devices. The present invention also relates generally to the genetic modification of cytotoxic T-lymphocytes to reduce target cell killing by apoptosis and / or increase production of lytic proteins at desired times. In an aspect, the invention relates to the use of these genetically modified T-lymphocytes and / or natural killer cells with CAR devices, Smart CAR devices, DE CAR devices, and / or Smart-DE CAR devices to enhance the immune response against a disease.
Owner:CHIMERA BIOENG INC
Engineering and delivery of therapeutic compositions of freshly isolated cells
ActiveUS8450112B2Simple procedureGood treatment effectGenetic material ingredientsFermentationNatural Killer Cell Inhibitory ReceptorsCD8
The present invention relates to the transient modification of cells. In particular embodiments, the cells are immune systems, such as PBMC, PBL, T (CD3+ and / or CD8+) and Natural Killer (NK) cells. The modified cells provide a population of cells that express a genetically engineered chimeric receptor which can be administered to a patient therapeutically. The present invention further relates to methods that deliver mRNA coding for the chimeric receptor to unstimulated resting PBMC, PBL, T (CD3+ and / or CD8+) and NK cells and which delivers the mRNA efficiently to the transfected cells and promotes significant target cell killing.
Owner:MAXCYTE
Method for simultaneous and efficient amplification of CD<3+>CD<56+>CIK cells and CD<3->CD<56+>NK cells
ActiveCN104357390AHigh purityHigh activityBlood/immune system cellsAdoptive cellular immunotherapySerum free media
The invention discloses a method for simultaneous and efficient amplification of CD<3+>CD<56+>CIK cells and CD<3->CD<56+>NK cells. The method comprises the steps as follows: the concentration of separated PBMC (peripheral blood mononuclear cells) is adjusted by a serum-free medium containing autologous plasma, an Anti-CD16 antibody, IL-2 and IL-15 are added, and then the mixture is transferred into a T175 culture flask for culture; an Anti-CD3 antibody and an Anti-CD137 antibody are added; a serum-free medium containing the autologous plasma, IL-2 and IL-15 is supplemented every two days according to the cell growth condition; the cell concentration is controlled to be about 1.5*10<6> / ml; and after culture is performed for 14-21 days, large quantities of high-purity CD<3+>CD<56+>CIK cells and CD<3->CD<56+>NK cells can be obtained simultaneously, and the total cell quantity can reach an effective value of the cell quantity required for adoptive cellular immunotherapy clinically for tumor. The method for simultaneous and efficient amplification of the CD<3+>CD<56+>CIK cells and the CD<3->CD<56+>NK cells is simple, convenient, effective and high in cell killing activity.
Owner:HRYZ (SHENZHEN) BIOTECH CO +1
Method of treatment using ligand-immunogen conjugates
InactiveUS20060067946A1Improve recognitionEnhance endogenous immune response-mediated eliminationAntibacterial agentsBiocideBinding siteCytotoxicity
A method and pharmaceutical composition are provided for enhancing the endogenous immune response-mediated elimination of a population of pathogenic cells in a host animal wherein the pathogenic cells preferentially express, uniquely express, or overexpress a binding site for a particular ligand. The invention comprises administering the ligand conjugated to an immunogen to a host animal harboring the population of pathogenic cells. Antibodies, preexisting or administered to the host animal to establish a passive immunity, directed against the immunogen bind to the ligand-immunogen conjugate resulting in elimination of the pathogenic cells by the host's immune response. At least one additional therapeutic factor is administered selected from the group consisting of a cell killing agent, a tumor penetration enhancer, a chemotherapeutic agent, antimicrobial agent, a cytotoxic immune cell, and a compound capable of stimulating an endogenous immune response wherein the compound does not bind to the ligand-immunogen conjugate.
Owner:PURDUE RES FOUND INC
Visualization and quantitation of cellular cytotoxicity using cell-permeable fluorogenic protease substrates and caspase activity indicator markers
ActiveUS20070184493A1Microbiological testing/measurementTetrapeptide ingredientsFluorescenceApoptosis pathways
This invention provides a non-radioactive assay to monitor and quantify the target-cell killing activities mediated by cytotoxic T lymphocytes (CTLs). This assay is predicated on the discovery that apoptosis pathway activation and, in particular, granzyme B activity, provides a measure of cytotoxic effector cell activity. In one embodiment, measurement of CTL-induced granzyme B activation in target cells is achieved through detection of the specific cleavage of fluorogenic granzyme B substrates. This assay reliably detects antigen-specific CTL killing of target cells, and provides a more sensitive, more informative and safer alternative to the standard 51Cr-release assay most often used to quantify CTL responses. The assay can be used to study CTL-mediated killing of primary host target cells of different cell lineages, and enables the study of antigen-specific cellular immune responses in real time at the single-cell level. As such, the assay can provide a valuable tool for studies of infectious disease pathogenesis and development of new vaccines and immunotherapies.
Owner:ONCOIMMUNIN
Chemotherapeutic methods and compositions
ActiveUS20100209415A1Enhance cell killing activityEnhance killing activityOrganic active ingredientsHeavy metal active ingredientsCell-Extracellular MatrixECM Protein
Disclosed herein are methods and compositions for enhancing the cell-killing activity of anti-neoplastic agents by inhibiting the activity of a lysyl oxidase-type enzyme. Also disclosed are methods for screening for chemotherapeutic agents, and for molecules that enhance the activity of chemotherapeutic agents, using cells grown on an extracellular matrix.
Owner:GILEAD BIOLOGICS
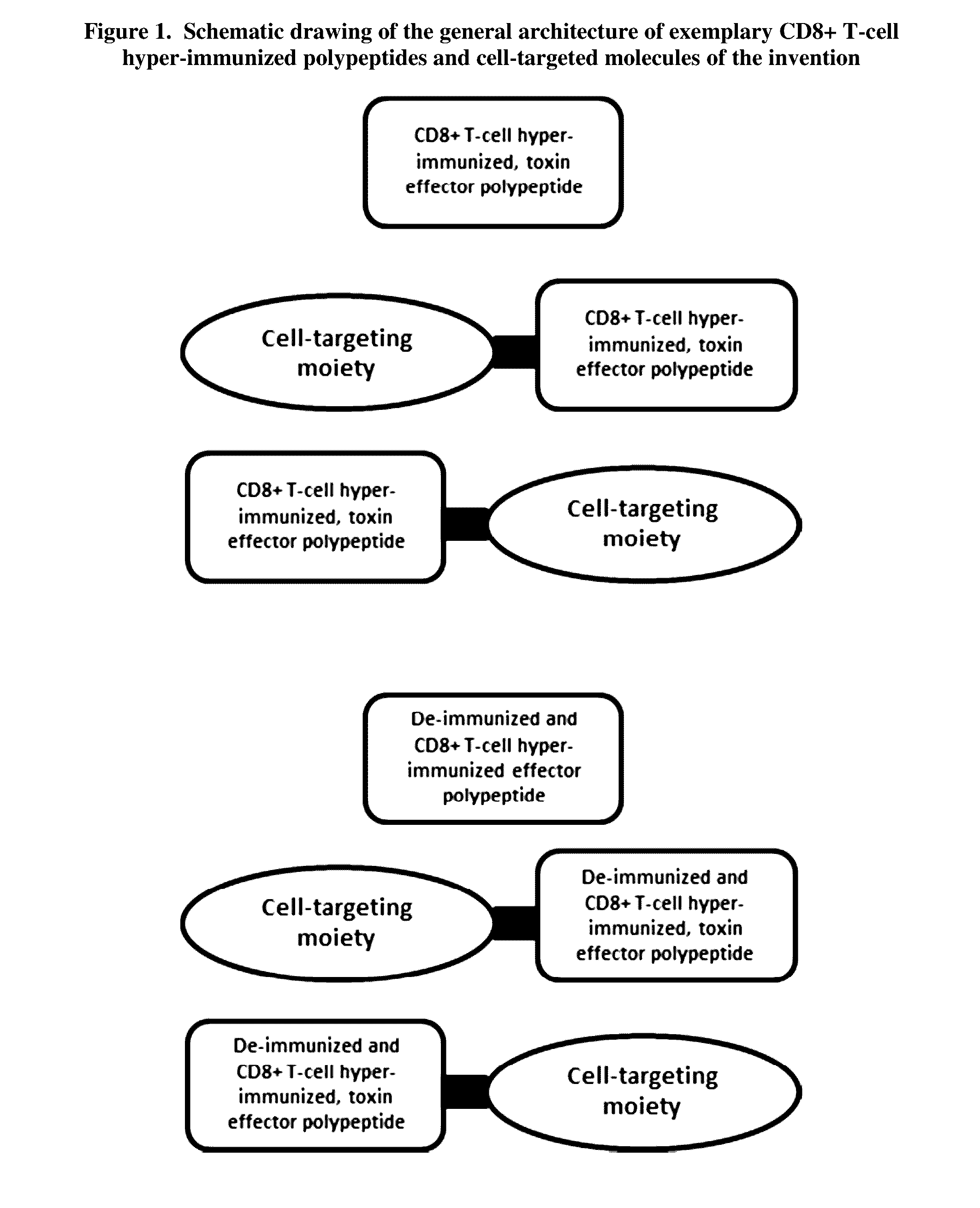
Mhc class i epitope delivering polypeptides and cell-targeted molecules for direct cell killing and immune stimulation via mhc class i presentation and methods regarding the same
InactiveUS20160347798A1Increase probabilityReduce probabilityAntibacterial agentsHydrolasesMHC class IHeterologous
The present invention is directed to T-cell epitope delivering polypeptides which deliver one or more CD8+ T-cell epitopes to the MHC class I presentation pathway of a cell, including toxin-derived polypeptides which comprise embedded T-cell epitopes and are de-immunized. The present invention provides cell-targeted, CD8+ T-cell epitope delivering molecules for the targeted delivery of cytotoxicity to certain cells, e.g., infected or malignant cells, for the targeted killing of specific cell types, and the treatment of a variety of diseases, disorders, and conditions, including cancers, immune disorders, and microbial infections. The present invention also provides methods of generating polypeptides capable of delivering one or more heterologous T-cell epitopes to the MHC class I presentation pathway, including polypeptides which are 1) B-cell and / or CD4+ T-cell de-immunized, 2) comprise embedded T-cell epitopes, and / or 3) comprises toxin effectors which retain toxin functions.
Owner:MOLECULAR TEMPLATES
Antibody induced cell membrane wounding
ActiveUS8377435B2Enhanced cell membrane wounding and killing of cellReduced viabilityOrganic active ingredientsNervous disorderAntigenCell Surface Antigens
Compositions and methods for inducing cell membrane wounding, cell permeabilization and cell killing are provided. The composition comprises a polyvalent agent that binds to a highly expressed cell surface antigen present on the surface of a cell. Preferably, the cell surface antigen is associated with the cytoskeleton of the cell. A preferred polyvalent agent is an IgM, and enhanced cell wounding and killing can be provided by the addition of a crosslinking agent. At sublethal concentrations in vivo, the cell wounding antibodies permeabilize cells and dramatically enhance response to chemotherapeutic agents, even in patients refractory to the chemotherapeutic agents.
Owner:MCURE BIOSCIENCES INC +1
Photosensitive antibody-phuorophore conjugates
InactiveCN103781495AAllow treatmentGarment special featuresEnergy modified materialsFluorescenceCell Surface Proteins
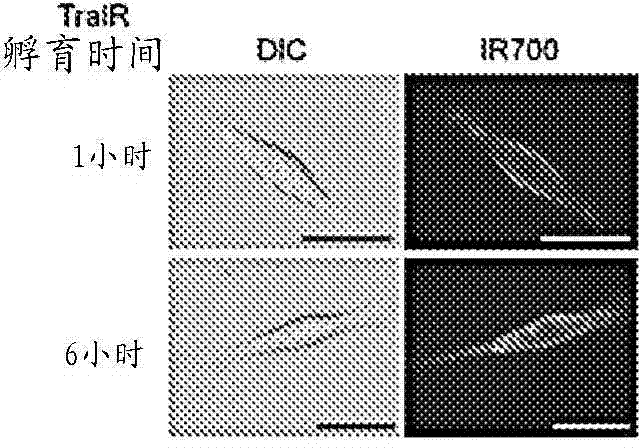
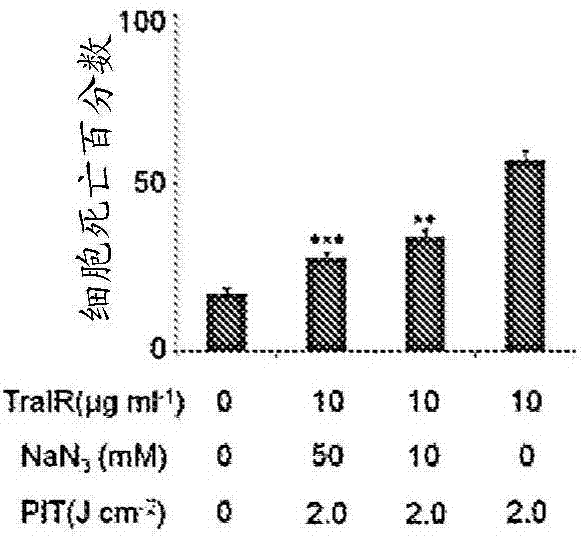
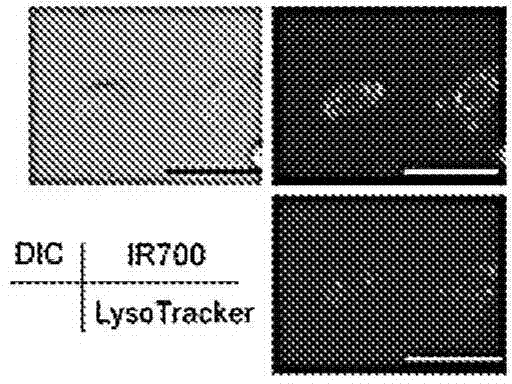
The present disclosure relates to compositions and methods of killing cells. In particular examples, the method includes contacting a cell having a cell surface protein with a therapeutically effective amount of an antibody-IR700 molecule, wherein the antibody specifically binds to the cell surface protein, such as a tumor-specific antigen on the surface of a tumor cell. The cell is subsequently irradiated, such as at a wavelength of 660 to 740 nm at a dose of at least 1 J cm<-2>. The cell is also contacted with one or more therapeutic agents (such as an anti-cancer agent), for example about 0 to 8 hours after irradiating the cell, thereby killing the cell. Also provided are methods of imaging cell killing in real time, using fluorescence lifetime imaging. Also provided are wearable devices that include an article of clothing, jewelry, or covering; and an NIR LED incorporated into the article, which can be used with the disclosed methods.
Owner:UNITED STATES OF AMERICA
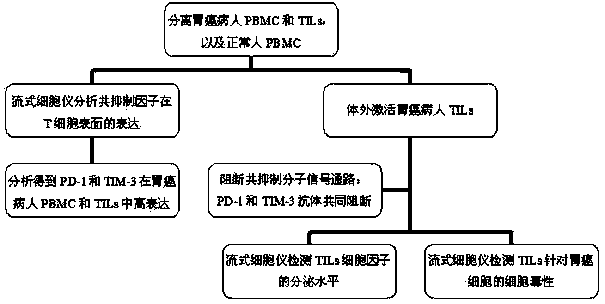
Application of co-blocking of PD-1 and TIM-3 signal paths to anti-stomach-cancer treatment
InactiveCN103721255AEnhance the effect of killing gastric cancer cellsAntibody ingredientsAntineoplastic agentsTreatment effectSide effect
The invention relates to application of co-blocking of PD-1 and TIM-3 signal paths to anti-stomach-cancer treatment, and in particular relates to a blocking agent of the PD-1 signal path, a blocking agent of the TIM-3 signal path and application of the blocking agents together with the conventional anti-stomach-cancer treatment medicaments to preparation of a medicinal composition for resisting stomach cancer. The blocking agents or a combination of the blocking agents and the conventional anti-stomach-cancer medicaments treats stomach cancer by enhancing the stomach cancer cell killing effect of T cells. The composition has a remarkable treatment effect, and has the advantages of small toxic and side effects and cooperation with the conventional stomach cancer treatment medicaments.
Owner:SUZHOU UNIV
Cryopreservation solution for preserving congenital intrathoracic kidney (CIK) cells and application thereof
InactiveCN103210903ANo significant difference in proliferative activityNo significant difference in killing activityDead animal preservationSerum free mediaCD8
The invention discloses a cryopreservation solution for preserving congenital intrathoracic kidney (CIK) cells and application thereof. The cryopreservation solution for preserving the CIK cells is prepared according to the following methods: adding dimethyl sulfoxide and dextran into a lymphocyte serum-free medium, so that the volume percent content of the dimethyl sulfoxide is 5-15 percent, and the volume percent content of the dextran is 5-15 percent. Compared with the cells before cryopreservation, the cells cryopreserved by employing the cryopreservation solution have the following advantages: (1) the cellular morphology does not have obvious difference; (2) immunophenotype (CD3+CD56+CD8+) does not have obvious difference; and (3) the cell proliferation activity and cell killing activity do not have obvious difference.
Owner:XINXIANG MEDICAL UNIV
Engineering and Delivery of Therapeutic Compositions of Freshly Isolated Cells
ActiveUS20090257991A1Improve biological activitySimple procedureGenetic material ingredientsFermentationNatural Killer Cell Inhibitory ReceptorsCD8
The present invention relates to the transient modification of cells. In particular embodiments, the cells are immune systems, such as PBMC, PBL, T (CD3+ and / or CD8+) and Natural Killer (NK) cells. The modified cells provide a population of cells that express a genetically engineered chimeric receptor which can be administered to a patient therapeutically. The present invention further relates to methods that deliver mRNA coding for the chimeric receptor to unstimulated resting PBMC, PBL, T (CD3+ and / or CD8+) and NK cells and which delivers the mRNA efficiently to the transfected cells and promotes significant target cell killing.
Owner:MAXCYTE
Killing cell for high-efficiency and stable expression of antibodies and use thereof
PendingCN107523545AAntibacterial agentsAntibody mimetics/scaffoldsAntigen Binding FragmentAutoimmune disease
The invention relates to a killing cell for high-efficiency and stable expression of antibodies and a use thereof. Specifically, the invention provides the transgenic killing cell; a genome of the killing cell is stably integrated with an expression cassette containing an encoding sequence of human Fc segment-containing antibodies, or containing an encoding sequence of chimeric antigen receptors and inhibitory antibodies or activated type antibodies, and both ends of the expression cassette contain inverted terminal repeated sequences of a transposon. The killing cell can stably express the human Fc segment-containing antibodies or the chimeric antigen receptors and antigen-binding fragments derived from interested antibodies with high level while maintaining cell killing toxic effect. In addition, for preventing systemic toxicity and autoimmune diseases caused by over expression of antibodies due to in-vivo continuous proliferation of immune cells for stable expression of the antibodies, a molecular braking system is also introduced. With use of monoclonal antibody agents appearing on the market, the killing cell integrated with the antibody expression cassette can be quickly eliminated, and the safety of treatment is effectively improved.
Owner:SHANGHAI CELL THERAPY RES INST +1
Recombinant lentivirus and application thereof
ActiveCN106749675ASignificant in vivo and in vitro amplificationSignificant tumor killing effectMammal material medical ingredientsImmunoglobulinsAbnormal tissue growthMicro environment
The invention relates to the field of tumor cellular immunotherapy, and in particular relates to a recombinant lentivirus and application thereof. The recombinant lentivirus comprises a chimeric antigen receptor, wherein the chimeric antigen receptor mainly comprises signal peptide, an antigen recognition domain, a transmembrane domain, an intracellular co-stimulation signal transduction domain and a CD3 zeta signal transduction domain which are serially connected; the intracellular co-stimulation signal transduction domain mainly comprises a human TLR2 (Toll Like Receptor 2) intracellular domain. A GPC3 CAT T (Glypican 3 CAT T) cell prepared from the recombinant lentivirus has an intense cell killing effect on liver cancer cells, a Th1 cell factor can be highly expressed, a tumor killing effect caused by non-CAR T (Chimeric Antigen Receptor T) cell can be stimulated to the maximum extent, escape and potential reoccurrence risk of GPC 3-tumor cells can be effectively prevented, the tumor cells can be killed by T cells expressing the chimeric antigen receptor, normal tissue can be slightly damaged, a tumor immunosuppression micro environment can be broken through, and thus a relatively good treatment effect on solid tumor can be achieved.
Owner:SHENZHEN IN VIVO BIOMEDICINE TECH LTD
Targeted double stranded rna mediated cell killing
ActiveUS20070010467A1Improve efficacyAchieve rapidityNervous disorderHydroxy compound active ingredientsDouble strandDouble stranded rna
A method of killing a specific target cell / tissue is disclosed. The method comprises exposing the specific target cell / tissue to a composition-of-matter comprising a double stranded RNA molecule associated with a targeting moiety selected capable of targeting to the specific target cell / tissue, thereby killing the specific target cell / tissue.
Owner:TARGIMMUNE THERAPEUTICS AG
Chemotherapeutic methods and compositions
ActiveUS20130017207A1High activityEnhance killing activityOrganic active ingredientsHeavy metal active ingredientsCell-Extracellular MatrixECM Protein
Disclosed herein are methods and compositions for enhancing the cell-killing activity of anti-neoplastic agents by inhibiting the activity of a lysyl oxidase-type enzyme. Also disclosed are methods for screening for chemotherapeutic agents, and for molecules that enhance the activity of chemotherapeutic agents, using cells grown on an extracellular matrix.
Owner:GILEAD BIOLOGICS
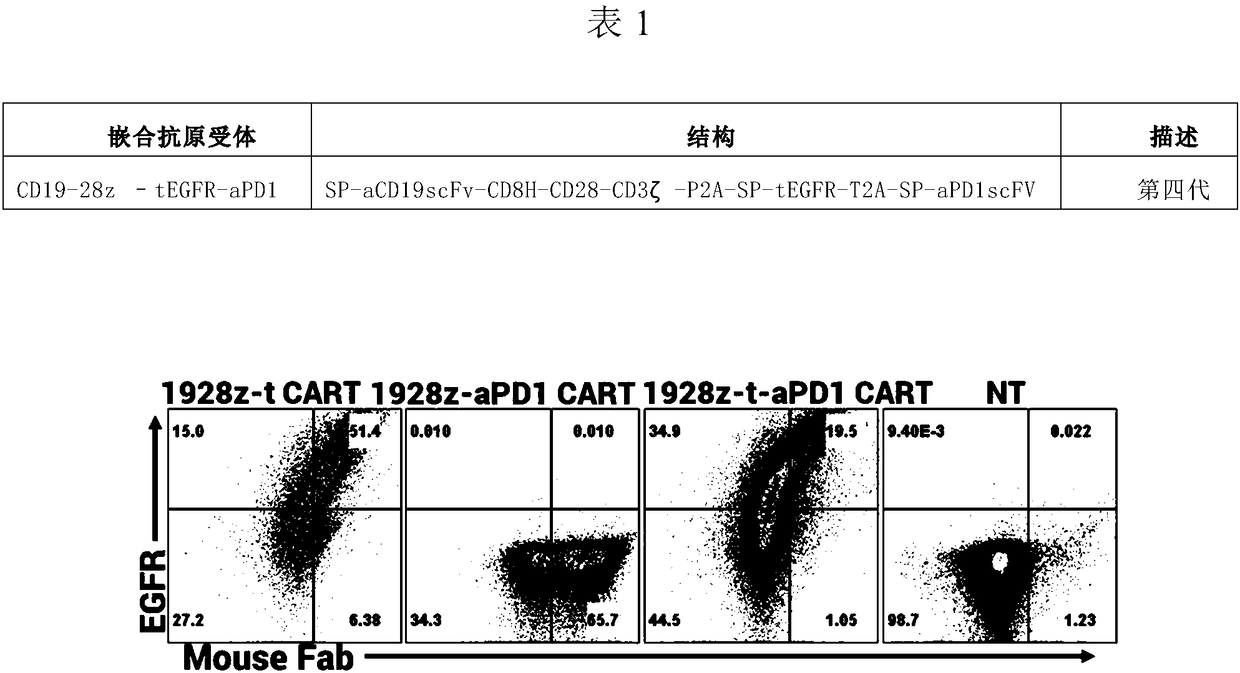
CD19 targeted chimeric antigen receptor, method of dual-modifying same, and application of the CD19 targeted chimeric antigen receptor
ActiveCN108330133APolypeptide with localisation/targeting motifImmunoglobulin superfamilySingle-Chain AntibodiesPolynucleotide
The invention relates to a CD19 targeted chimeric antigen receptor and an application thereof, and particularly provides a polynucleotide sequence, which is selected from: (1) a polynucleotide sequence which contains, in a successively connected manner, an encoding sequence of anti-CD19 single-chain antibody, an encoding sequence of human CD8[alpha] hinge zone, an encoding sequence of human CD28 transmembrane zone, an encoding sequence of human CD28 intracellular zone, an encoding sequence of human CD3 [zeta] intracellular zone, and optionally, a fraction, which contains an extracellular domain III and an extracellular domain IV of EGFR, and an encoding sequence of anti-human PD1 sequence fraction; and (2) a complementary sequence of the polynucleotide sequence (1). The invention also provides related fusion proteins, a carrier containing the encoding sequences, and applications of the fusion proteins, encoding sequences and carrier. The CAR-T cell has strong killing effect on specifictumor cells, and can reach more than 90% in killing efficiency on the specific tumor cells under the multiplicity of infection of 1:2. The CAR-T cell can secrete the PD1 antibody and has regulation effect on immunosuppression micro-environments.
Owner:HRAIN BIOTECHNOLOGY CO LTD
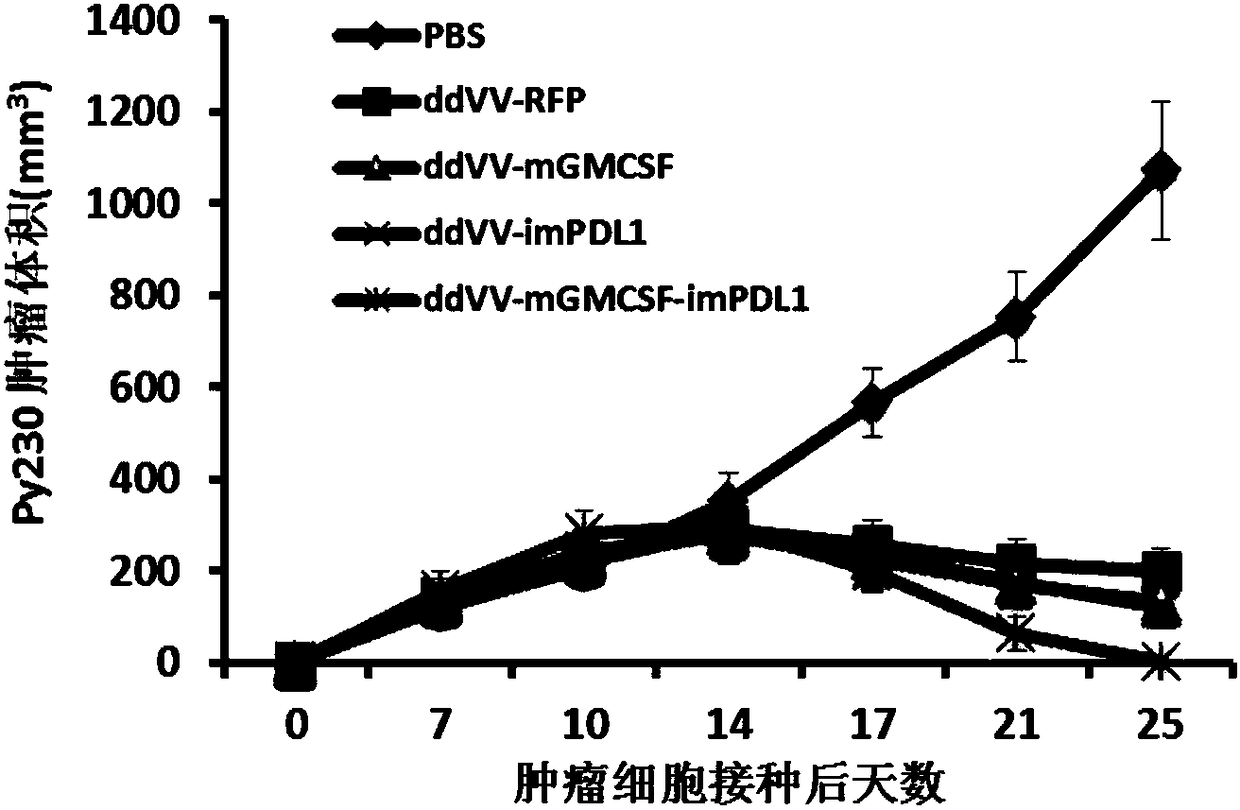
Oncolytic virus construction body, oncolytic virus and application thereof
InactiveCN108728488APrevent proliferationSignificant effectAntibody mimetics/scaffoldsUnknown materialsOncolytic adenovirusTumor specific
The invention provides an oncolytic virus construction body, an oncolytic virus and application thereof. The oncolytic virus construction body comprises a first nucleic acid molecule and a second nucleic acid molecule, wherein the first nucleic acid molecule encodes a secreted fusion protein molecule; the secreted fusion protein molecule is used for inhibiting immune checkpoints; and the second nucleic acid molecule encodes an immunostimulant molecule. According to the embodiment of the invention, the construction body encodes fusion protein and immunostimulant molecules for inhibiting the immune checkpoints and can inhibit immunosuppression mechanism mediated by the immune checkpoints and immunoreactions which causes individual tumor specificity so as to realize generalized and effectivetumor cell killing.
Owner:LIFESEQ LTDRP
Preparation method and application of CAR-T cell targeting B7H3
ActiveCN109880804AHas a lethal effectEnhance killing activityMammal material medical ingredientsAntineoplastic agentsBiologyLung cancer
The invention relates to a preparation method of a CAR-T cell targeting B7H3. The preparation method includes first preparing a PBMC cell; then co-transfecting a 293T cell with a shuttle plasmid LV-B7H3 containing the CAR structure, a helper plasmid psPAX2 and an envelope plasmid VSV-G to obtain a packaged B7H3-CAR virus; then taking a PBMC cell, using anti-human CD3 and anti-human CD28 as activators, culturing and activating for 48 hours and adding the B7H3-CAR virus for infection. By means of the preparation scheme, the expression of IFN-gamma in the CAR-T cell is increased, and the cell killing activity is high. The CAR-T cell targeting B7H3 has a killing effect on various solid tumor cells, has high killing activity, is safe and effective, and can be used for immunotherapy of kidney cancer, lung cancer, liver cancer, glioma, ovarian cancer, breast cancer and the like.
Owner:XUZHOU MEDICAL UNIV
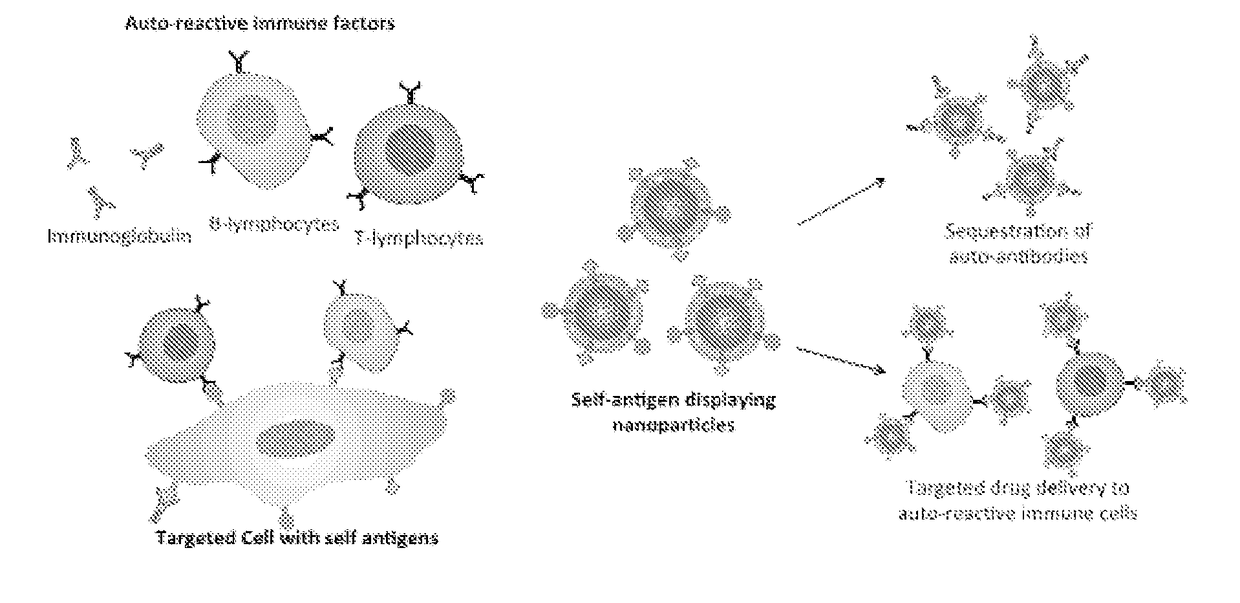
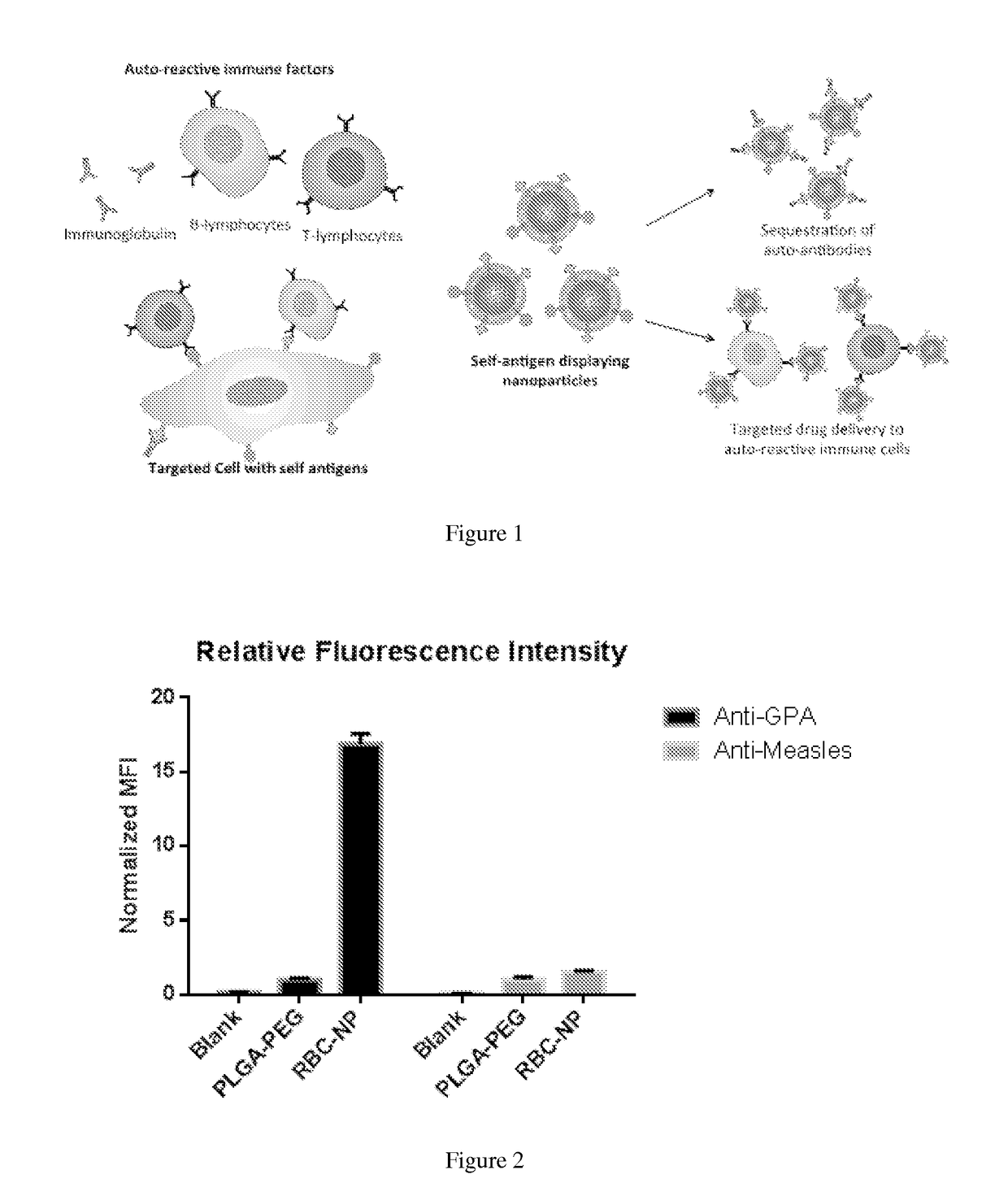
Self-Antigen Displaying Nanoparticles Targeting Auto-Reactive Immune Factors and Uses Thereof
PendingUS20170274059A1Normalize the immune regulationRelieve symptomsMammal material medical ingredientsCarrier-bound antigen/hapten ingredientsNanoparticleCytotoxic drug
The invention provides a composition, and method of use thereof, comprising self-antigen displaying nanoparticles to target auto-reactive immune components for treating and / or preventing the autoimmune diseases associated therewith. The nanoparticles can also be loaded with cytotoxic drugs for targeted cell killing or with immune-tolerizing compounds to normalize the immune regulation.
Owner:RGT UNIV OF CALIFORNIA
Recombinant fusion protein and polynucleotide construct for immunotoxin production
InactiveUS20080292646A1Effective internalizingBacteriaAntibody mimetics/scaffoldsNucleotidePseudomonas tolaasii
The present invention relates to a polynucleotide construct encoding a fusion protein consisting of a domain which binds the immunoglobulin Fc region, genetically fused to a truncated form of Pseudomonas exotoxin A (PE). In particular, the invention discloses the fusion protein, ZZ-PE38, and further provides immunotoxins, formed from complexes of the fusion protein with antibodies for targeted cell killing.
Owner:RAMOT AT TEL AVIV UNIV LTD
Antibody induced cell membrane wounding
ActiveUS20060153854A1Enhanced cell membrane wounding and killing of cellReduced viabilityOrganic active ingredientsNervous disorderAntigenCell Surface Antigens
Compositions and methods for inducing cell membrane wounding, cell permeabilization and cell killing are provided. The composition comprises a polyvalent agent that binds to a highly expressed cell surface antigen present on the surface of a cell. Preferably, the cell surface antigen is associated with the cytoskeleton of the cell. A preferred polyvalent agent is an IgM, and enhanced cell wounding and killing can be provided by the addition of a crosslinking agent. At sublethal concentrations in vivo, the cell wounding antibodies permeabilize cells and dramatically enhance response to chemotherapeutic agents, even in patients refractory to the chemotherapeutic agents.
Owner:MCURE BIOSCIENCES INC +1
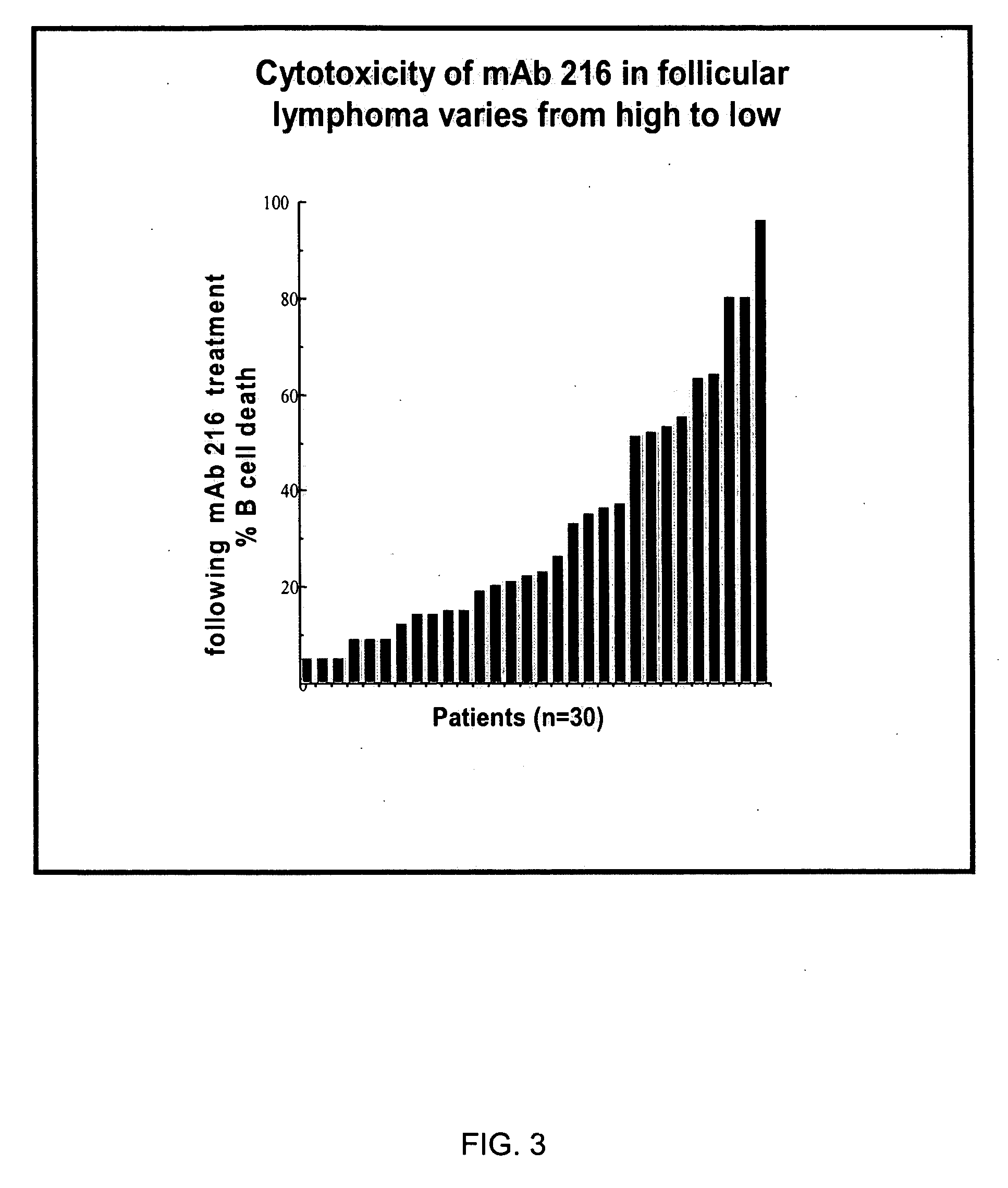
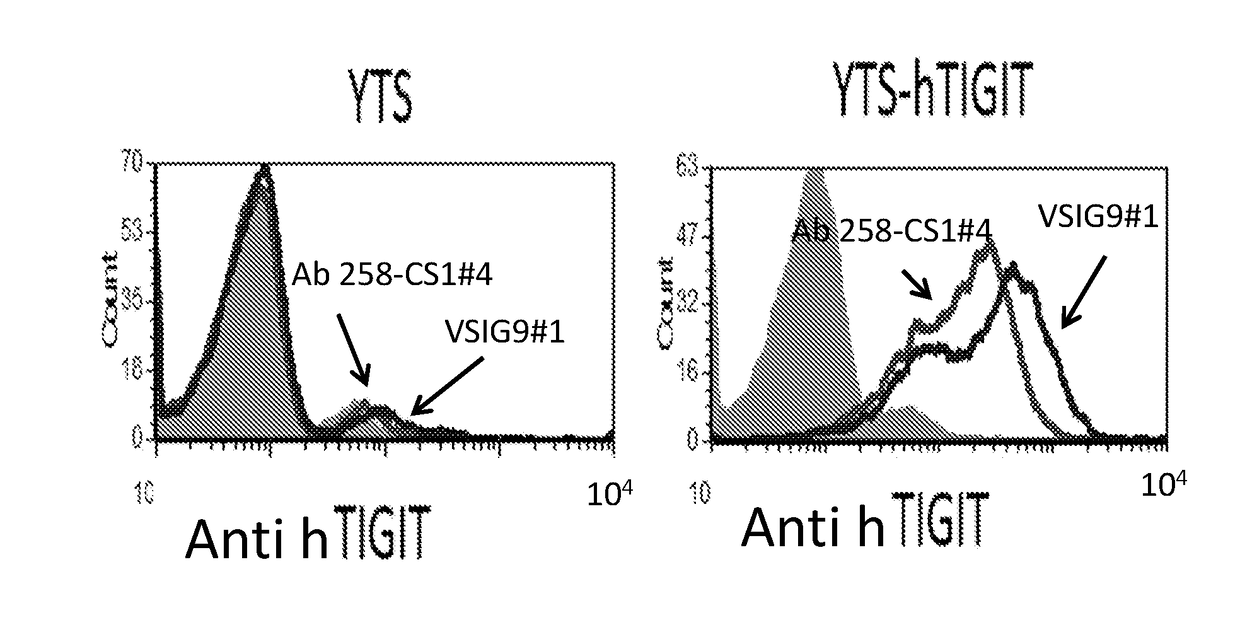
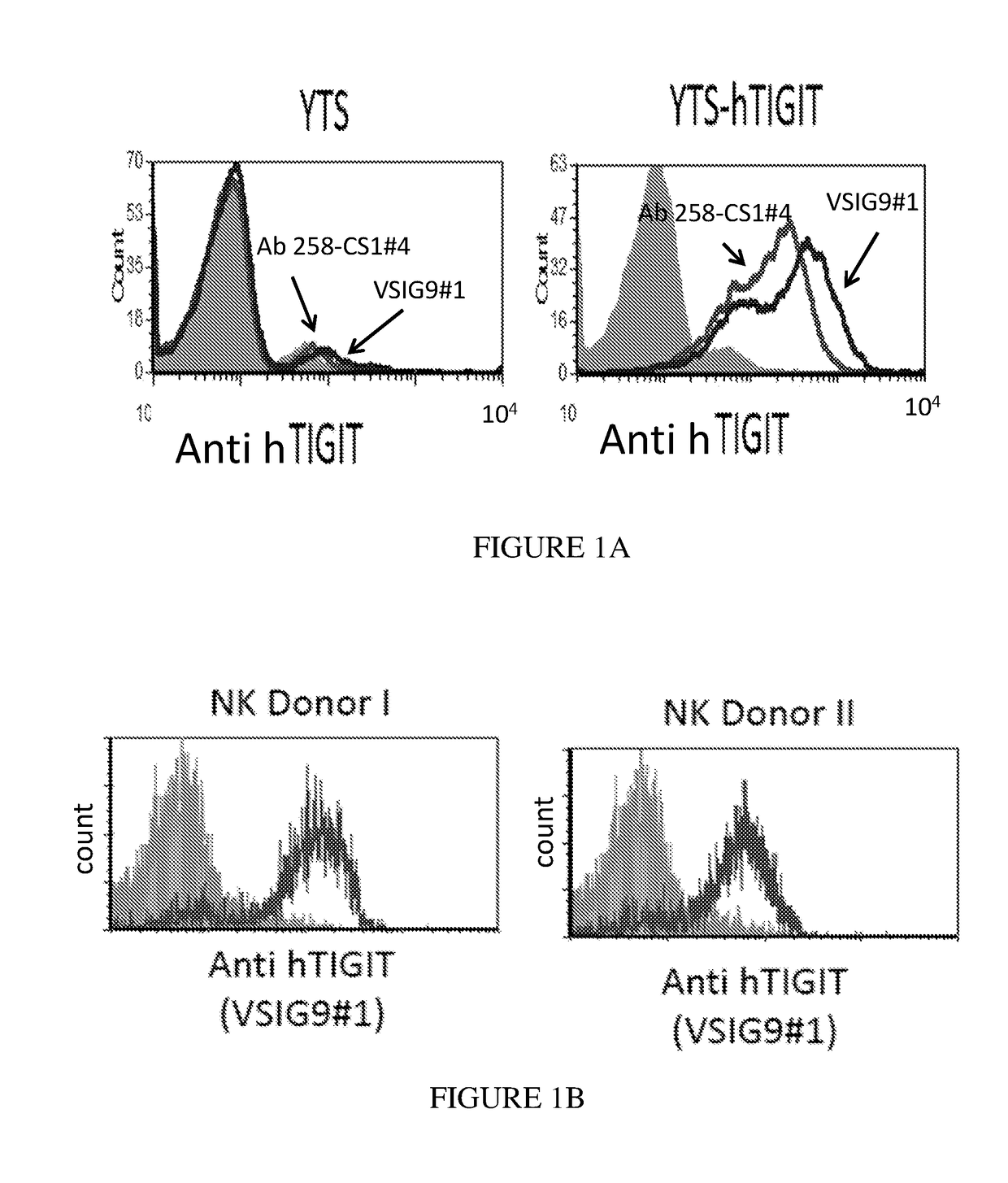
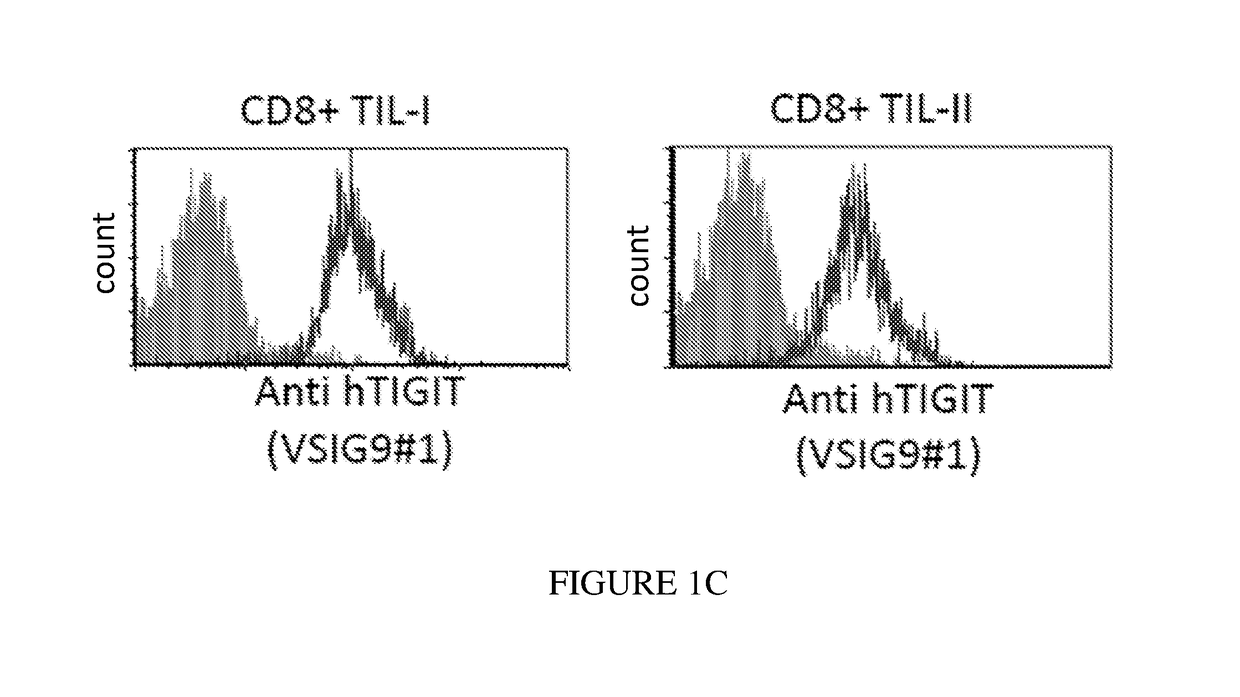
Antibodies specific to human t-cell immunoglobulin and itim domain (TIGIT)
ActiveUS20180185480A1Increasing and stimulating immune responseLess side effectsImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsNatural Killer Cell Inhibitory ReceptorsInhibitory receptors
Provided is a natural killer (NK) cell killing of various tumors is inhibited in the presence of ligands of TIGIT, an inhibitory receptor present on all human NK cells and on various T cells. Monoclonal antibodies that recognize TIGIT and inhibit its suppressive activity on T-cells are provided as well as pharmaceutical compositions including them and methods for their use in cancer immunotherapy and in diagnosis and treatment of immune disorders.
Owner:YISSUM RES DEV CO OF THE HEBREWUNIVERSITY OF JERUSALEM LTD +1
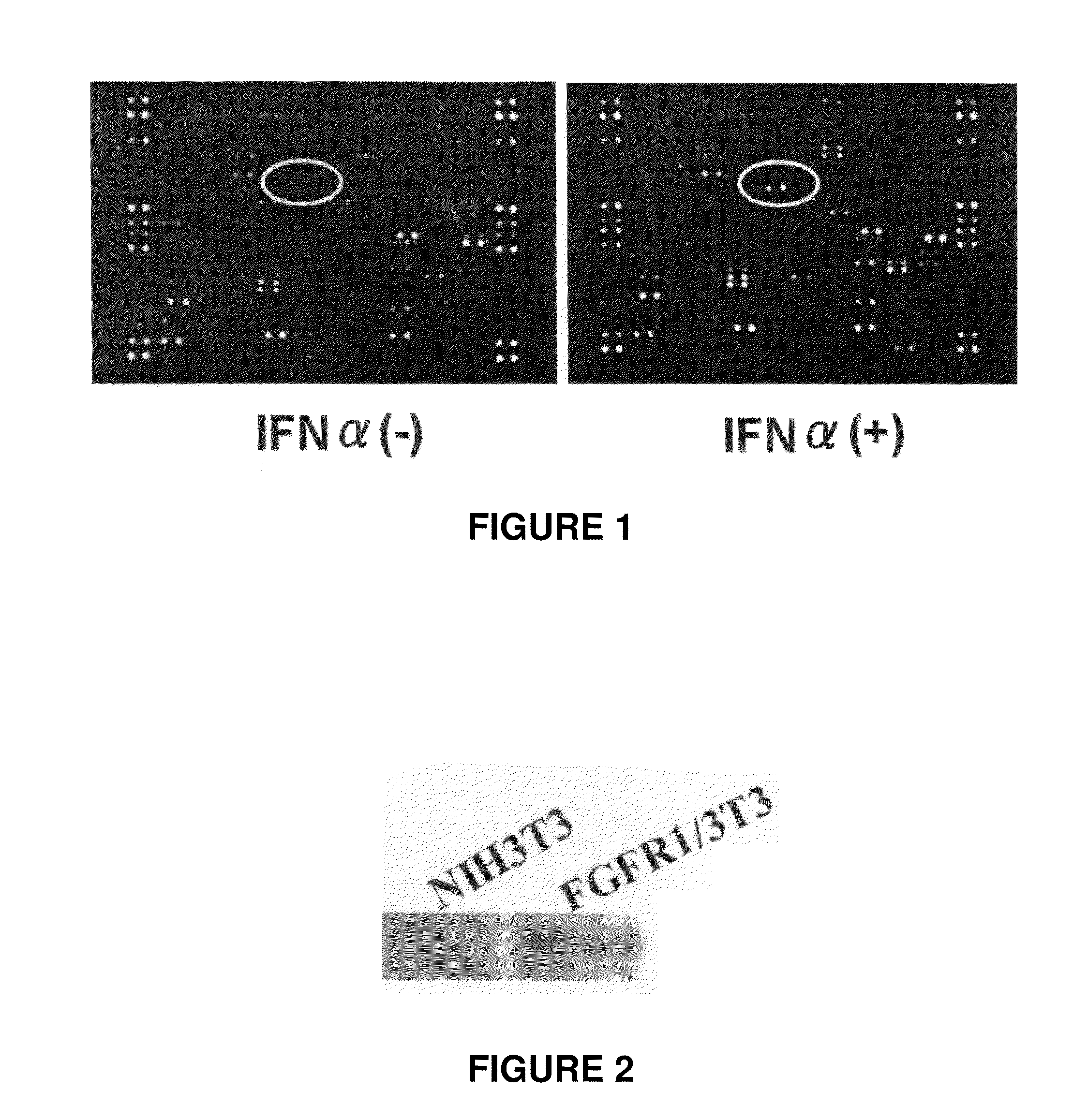
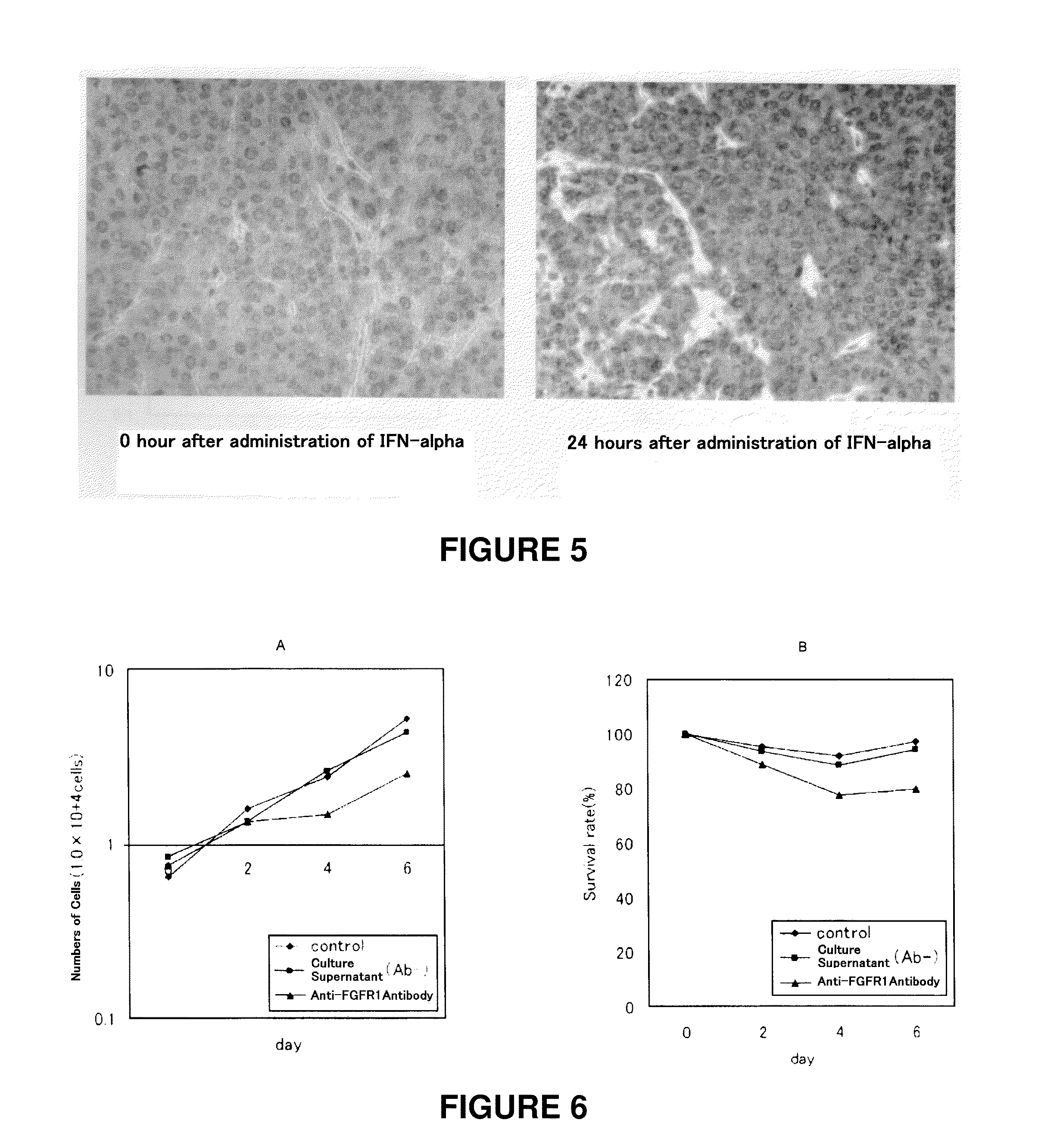
Therapeutic agent for cancer, and method for treatment of cancer
InactiveUS20110129524A1High expressionImprove aimingBiocidePeptide/protein ingredientsTherapeutic effectCancer therapy
[Problems] To provide a technique which enables an effective antibody therapy for cancer which targets for FGFR1 without the need of using any effective antibody having high specificity and a potent cell-killing activity.[Means for Solving Problems] Disclosed are: a therapeutic agent for cancer, which comprises an enhancer of the expression of a fibroblast growth factor receptor-1 and an anti-fibroblast growth factor receptor-1 antibody; and a method for the treatment of cancer using the therapeutic agent.
Owner:THE UNIV OF TOKYO +1