Patents
Literature
18299 results about "Thin walled" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
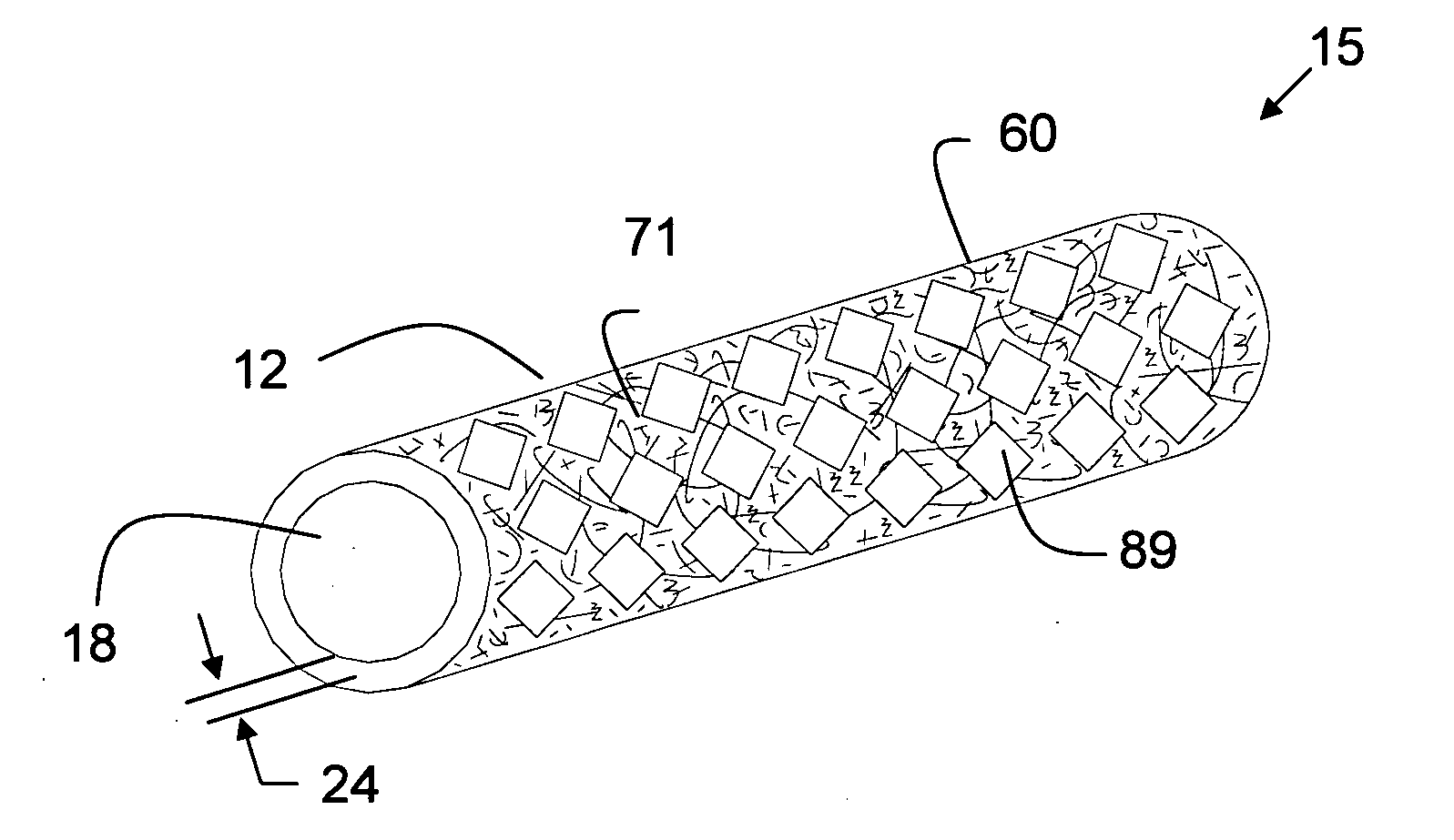
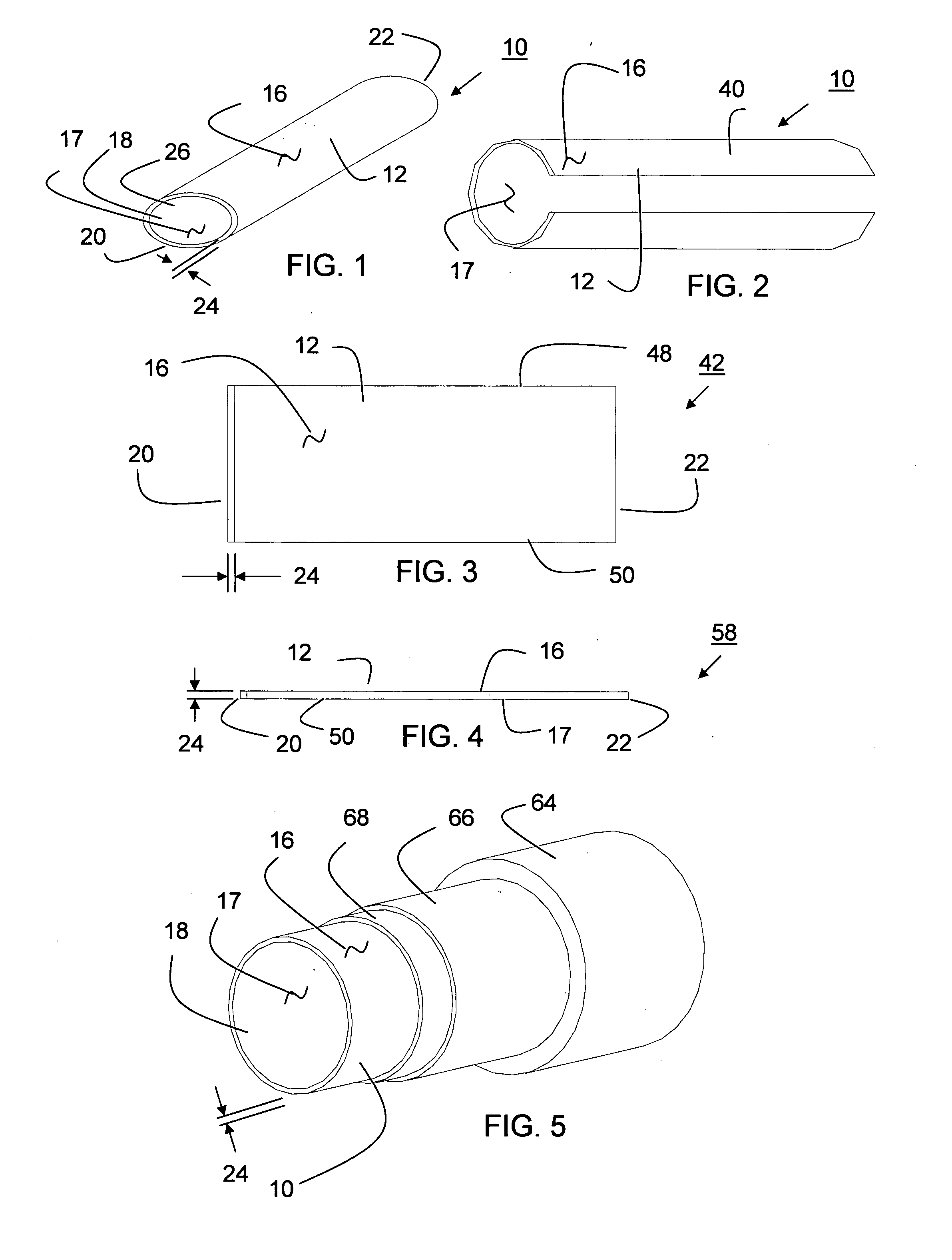
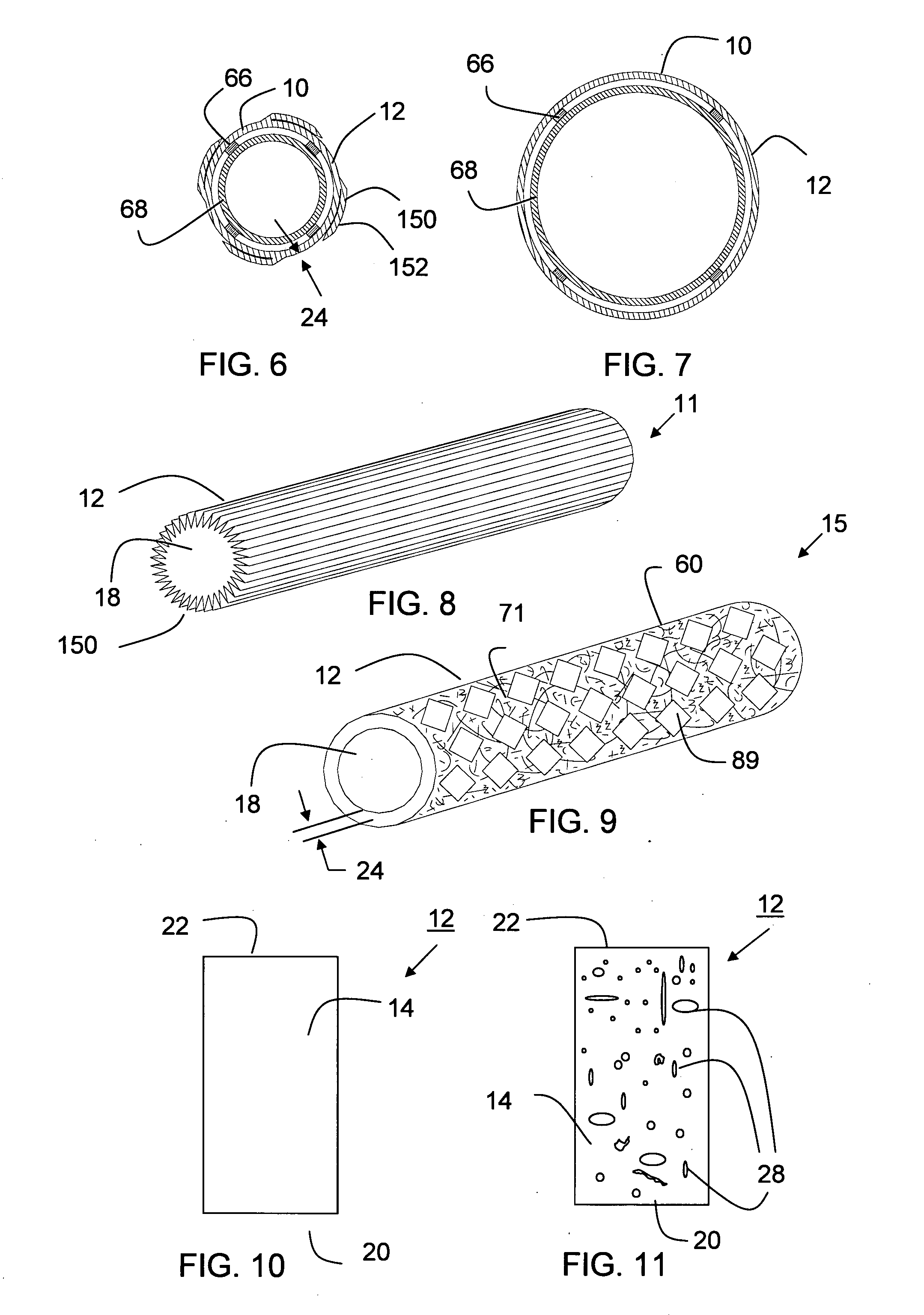
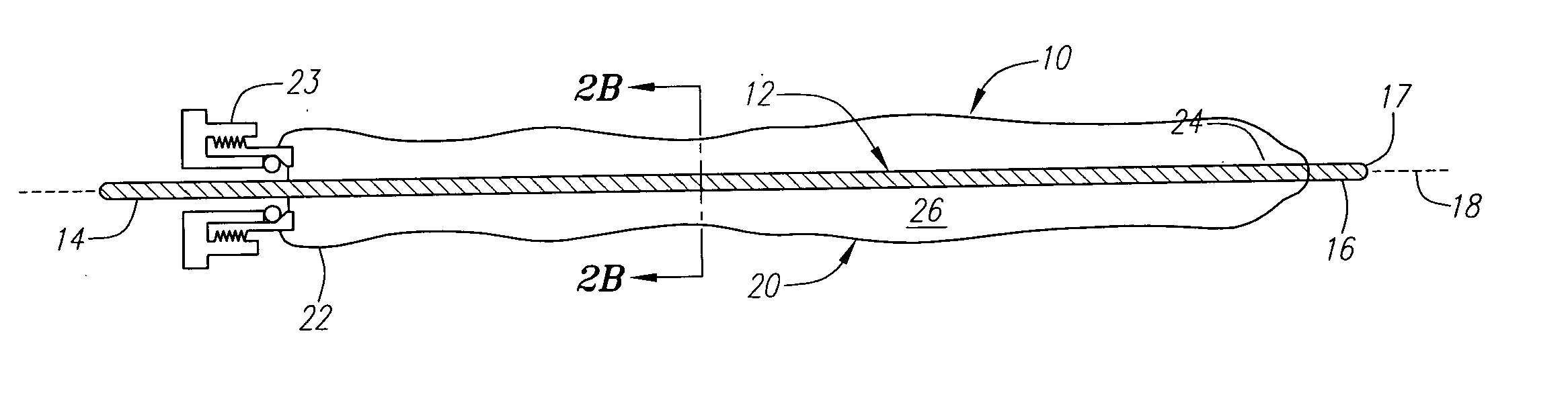
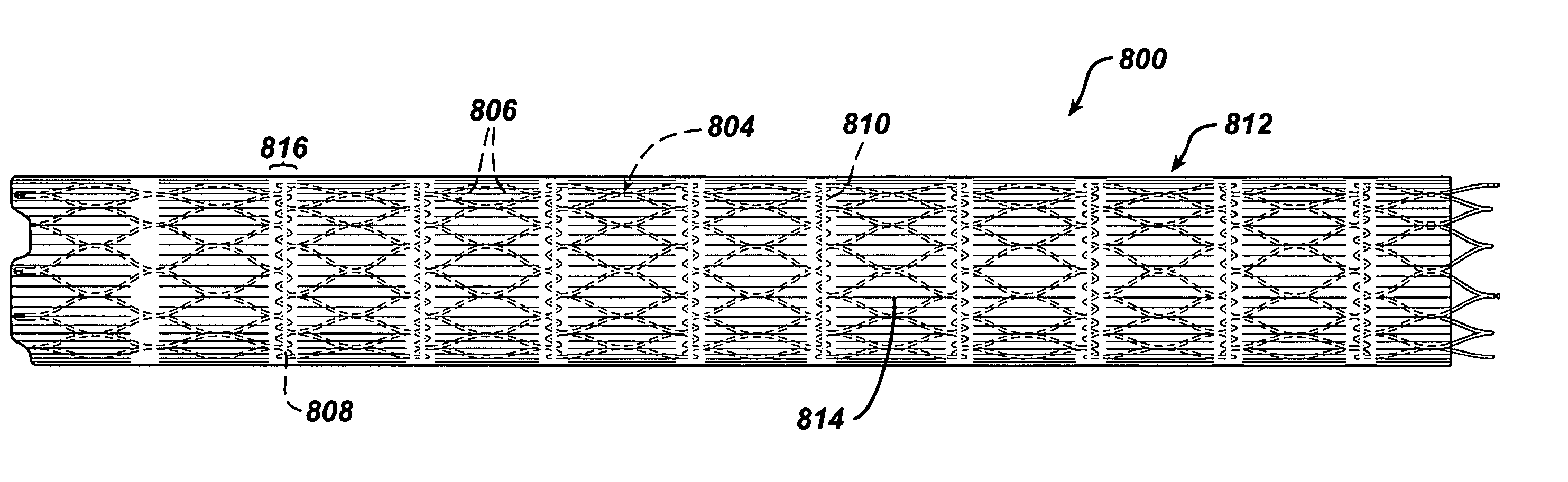
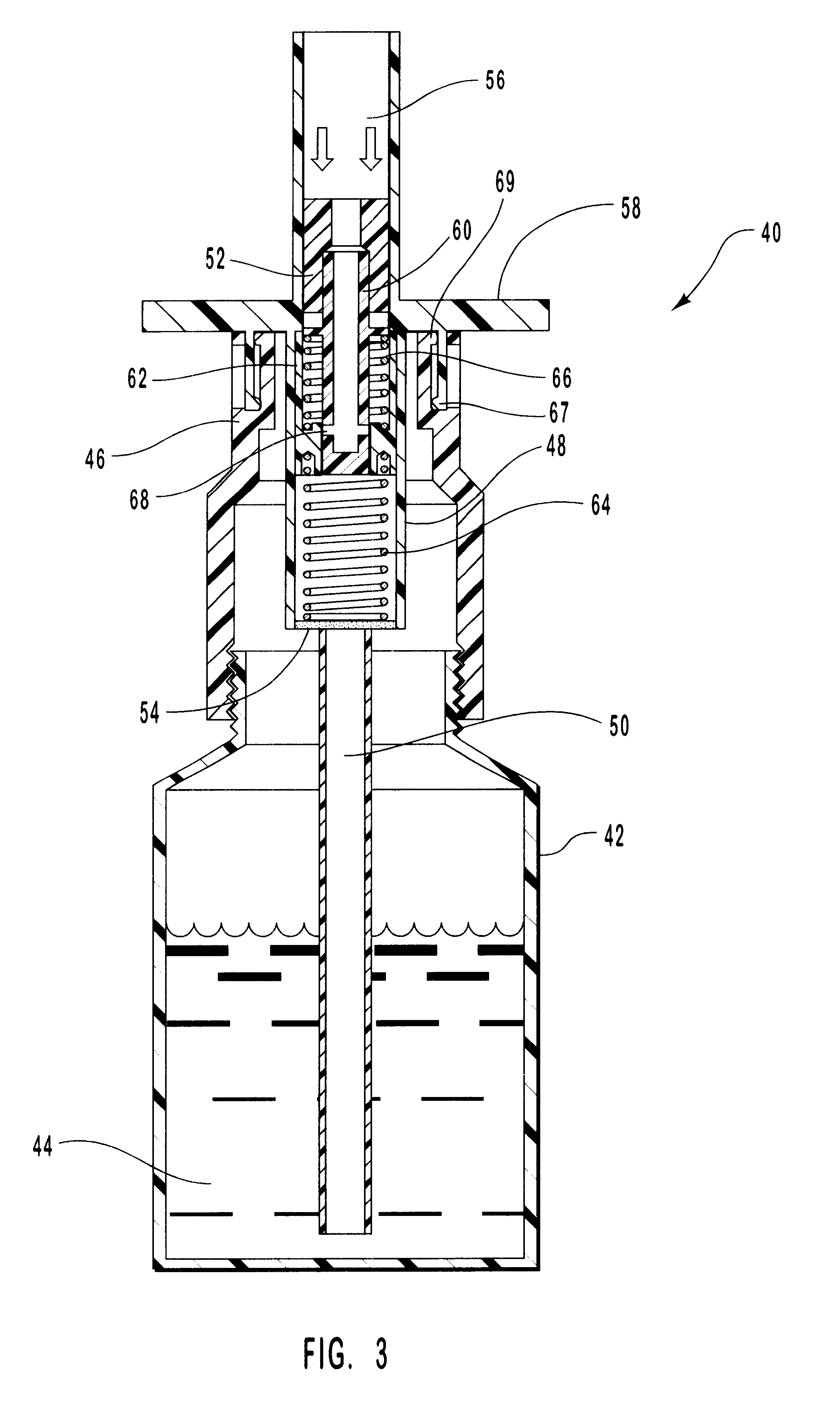
Tear and abrasion resistant expanded material and reinforcement
InactiveUS20070207186A1Increase flexibilityLess complex manufacturing processStentsSurgeryDiseaseEngineering
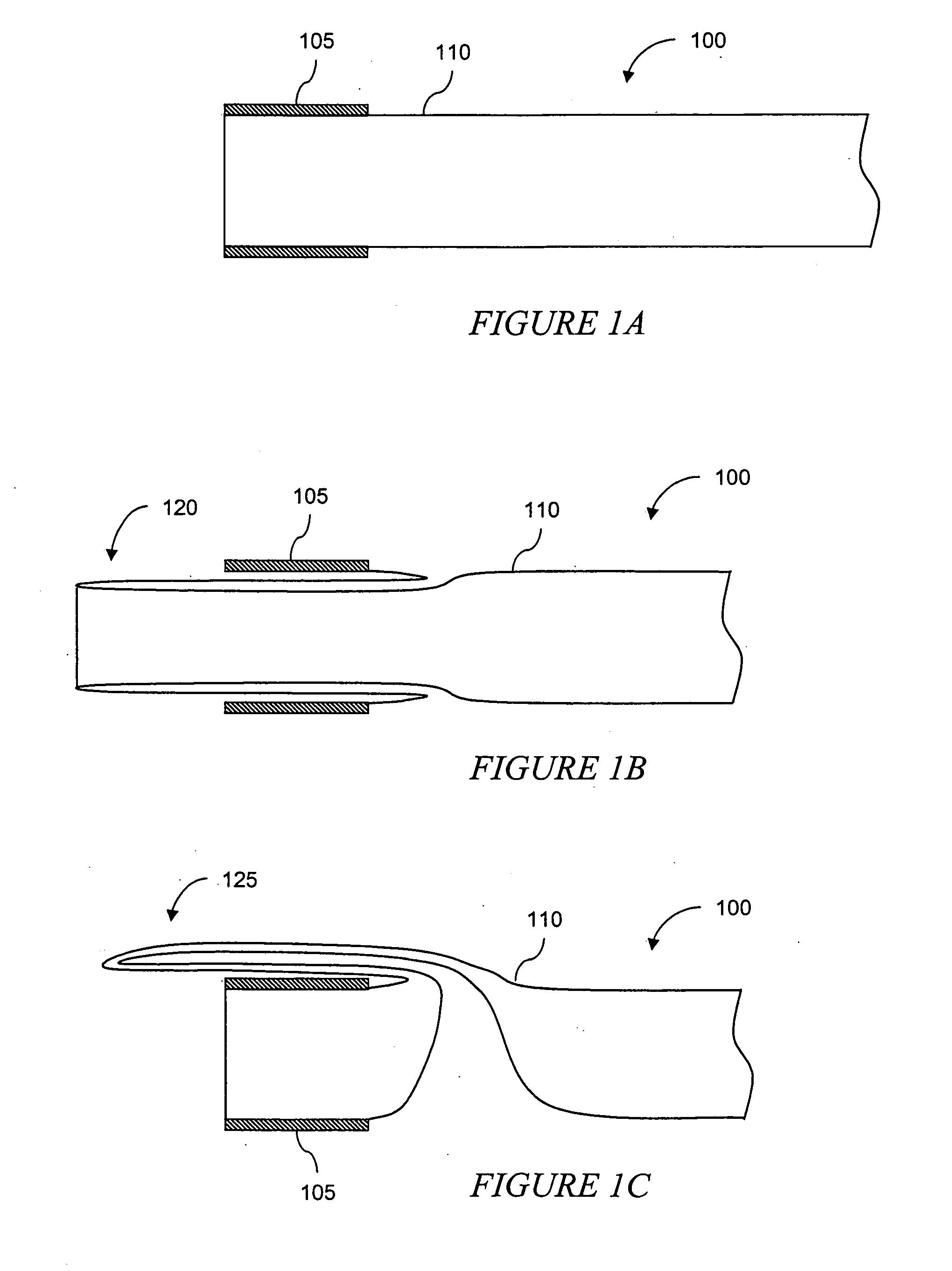
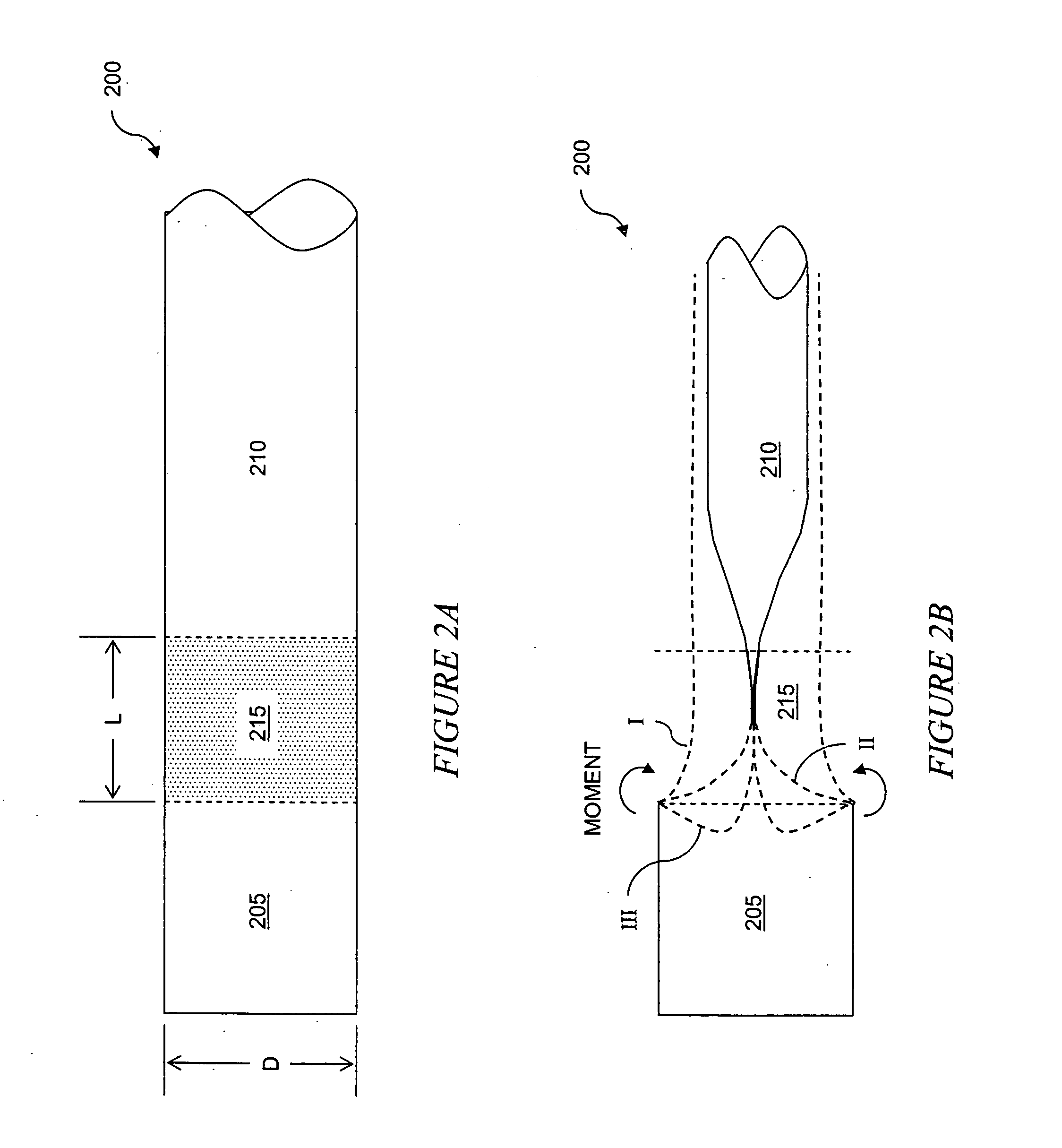
The present invention is a more durable expanded material that enables thinner wall thicknesses and a more flexible reinforcement suitable for stenting. The present invention is especially useful in the construction of grafts, stents, and stent-grafts which are used, for example, in repairing or replacing blood vessels that are narrowed or occluded by disease, aneurismal blood vessels, or other medical treatments. The inventive material and configurations allow expansion or contraction in size or adjustment in size in an incremental manner so that the optimum size, shape, and fit with other objects can be obtained. The present invention is also optionally capable of more accurately delivering one or more active ingredients such as drugs over longer periods of time. The present invention optionally includes surface modifications and additives that increase the surface adhesion of active ingredients, coatings, or combinations thereof. Finally, the present invention optionally includes growing cells on the inventive material so that the expanded material, reinforcement, or combinations thereof are useful, for example, in producing lab-grown blood vessels or organs.
Owner:SCANLON JOHN JAMES +1
Expandable guide sheath and apparatus with distal protection and methods for use
Apparatus and methods provide distal protection while accessing blood vessels within a patient's vasculature. A flexible sheath and distal protection element, e.g., a balloon or filter, are carried by a stiffening member. The sheath is lubricious and has a relatively thin wall, thereby providing a collapsible / expandable guide for delivering fluids and / or instruments. The sheath is advanced into a blood vessel in a contracted condition, expanded to an enlarged condition to define a lumen, and the distal protection element is deployed within the vessel beyond the sheath. Fluids and / or instruments are introduced into the vessel via the sheath lumen, the distal protection element retaining the fluids and / or capturing emboli released by the instruments. Upon completing the procedure, the sheath, distal protection element, and stiffening member are removed from the vessel.
Owner:MEDTRONIC INC
Systems and leads with a radially segmented electrode array and methods of manufacture
A method of making a lead for a stimulation device includes forming at least one pre-electrode in the shape of a ring, the at least one pre-electrode comprises at least two thin-walled portions separated by at least two thick-walled portions; disposing the at least one pre-electrode near a distal end of a lead body; joining at least one conductor to each thick-walled portion of the at least one pre-electrode; and grinding the lead body and the at least one pre-electrode to remove the thin-walled portions of the at least one pre-electrode to form segmented electrodes from the thick-walled portions of the at least one pre-electrode.
Owner:BOSTON SCI NEUROMODULATION CORP
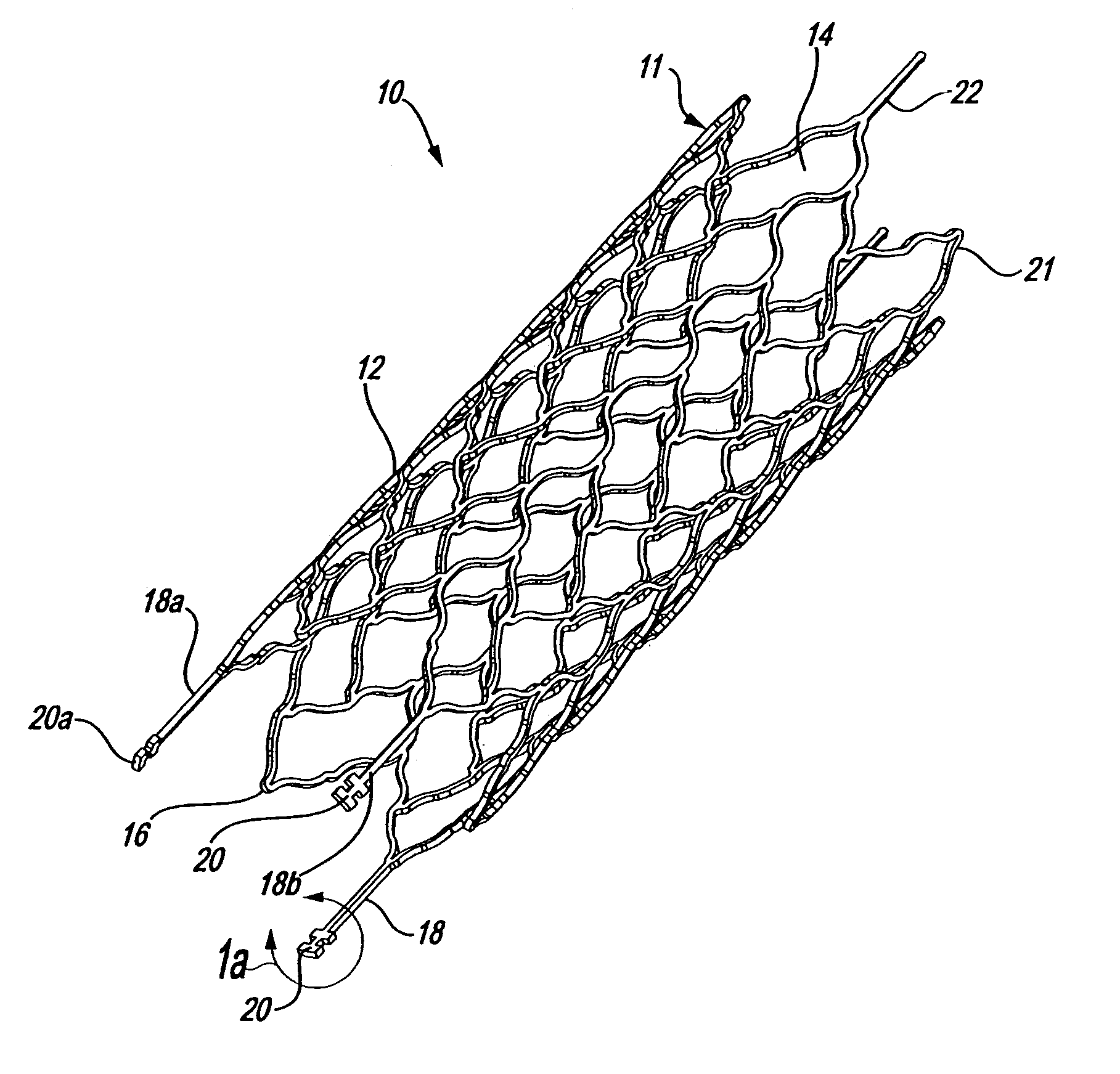
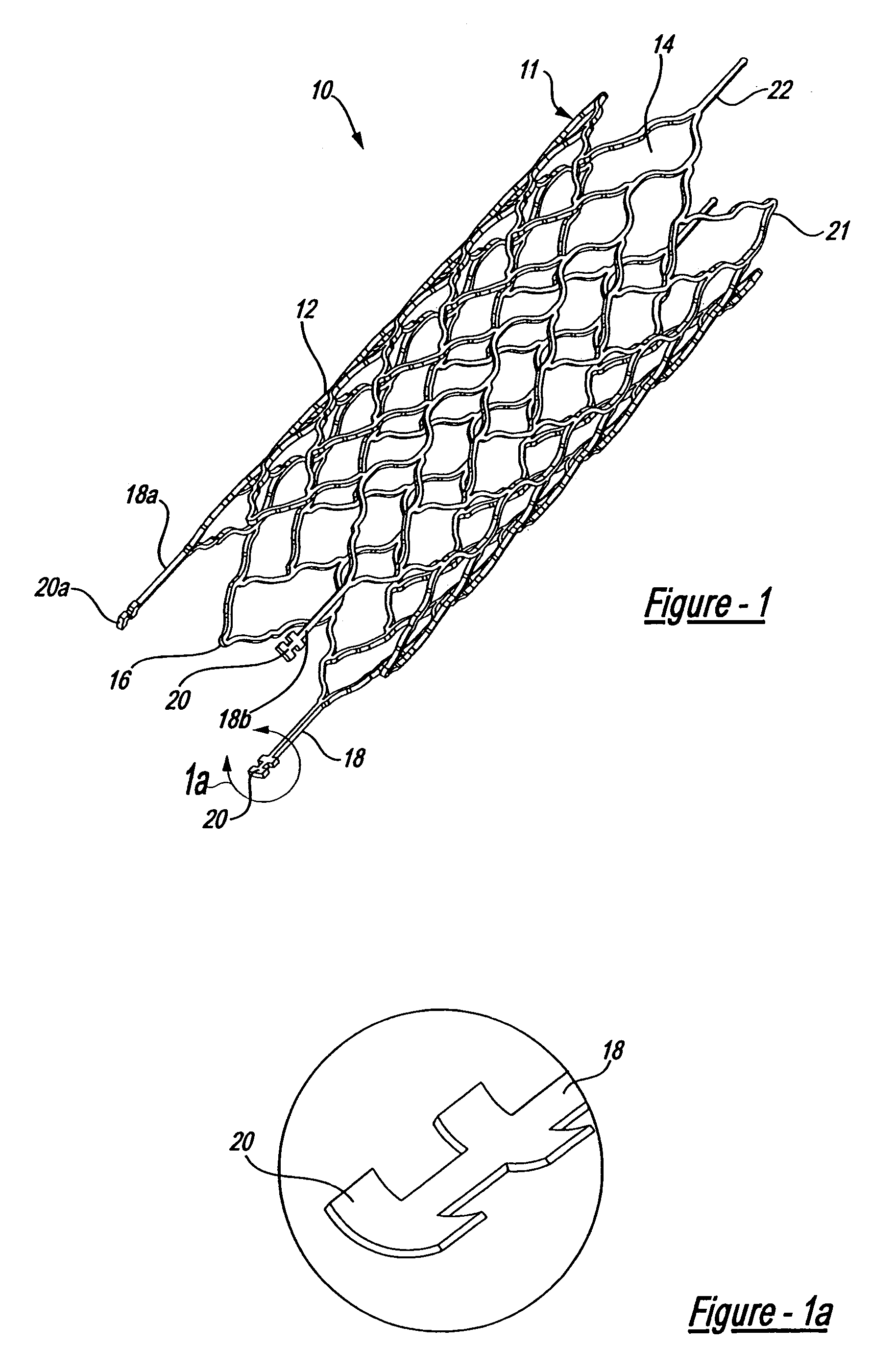
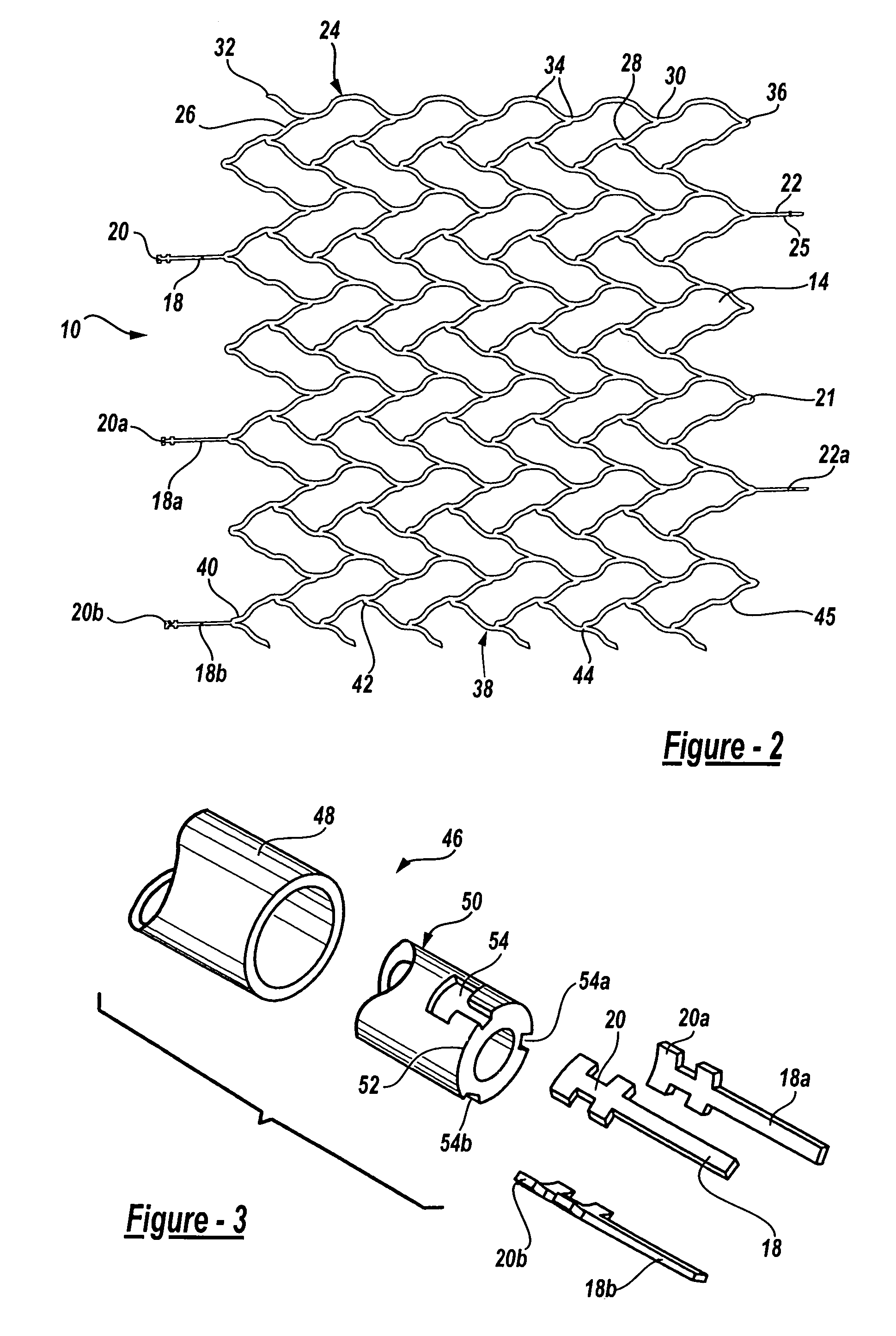
Intravascular stent device
A very small diameter intravascular stent device which may be used to occlude or partially occlude an aneurysm in the human brain which is comprised of a thin-walled skeletal cylindrical tube formed of undulating or sinusoidal elements which, when compressed, nest tightly with each other.
Owner:CODMAN & SHURTLEFF INC
Hollow floor slab with small dense ribs and its production process
InactiveCN1637217ALarge spanSafe and reliable structureFloorsForms/shuttering/falseworksFloor slabLine tubing
The hollow floor slab includes column, solid belt, solid reinforced beam with dense ribs, reinforced facing board, and composite box with lower thin bottom board. The production process includes: a) laying and tying reinforcing steel bar in the floor slab formwork; b) laying the steel wire net and other reinforcing material; c) setting and fixing made composite thin box; d) tying reinforcing steel bar for the facing board; e) fixing reinforcing bars with iron wire; and f) embedding prefabricated pipeline inside the facing board and the dense ribs. The present invention may produce flat sealed floor slab cavity with dense ribs in once deposition.
Owner:HUNAN BDF ENERGY SAVING TECH
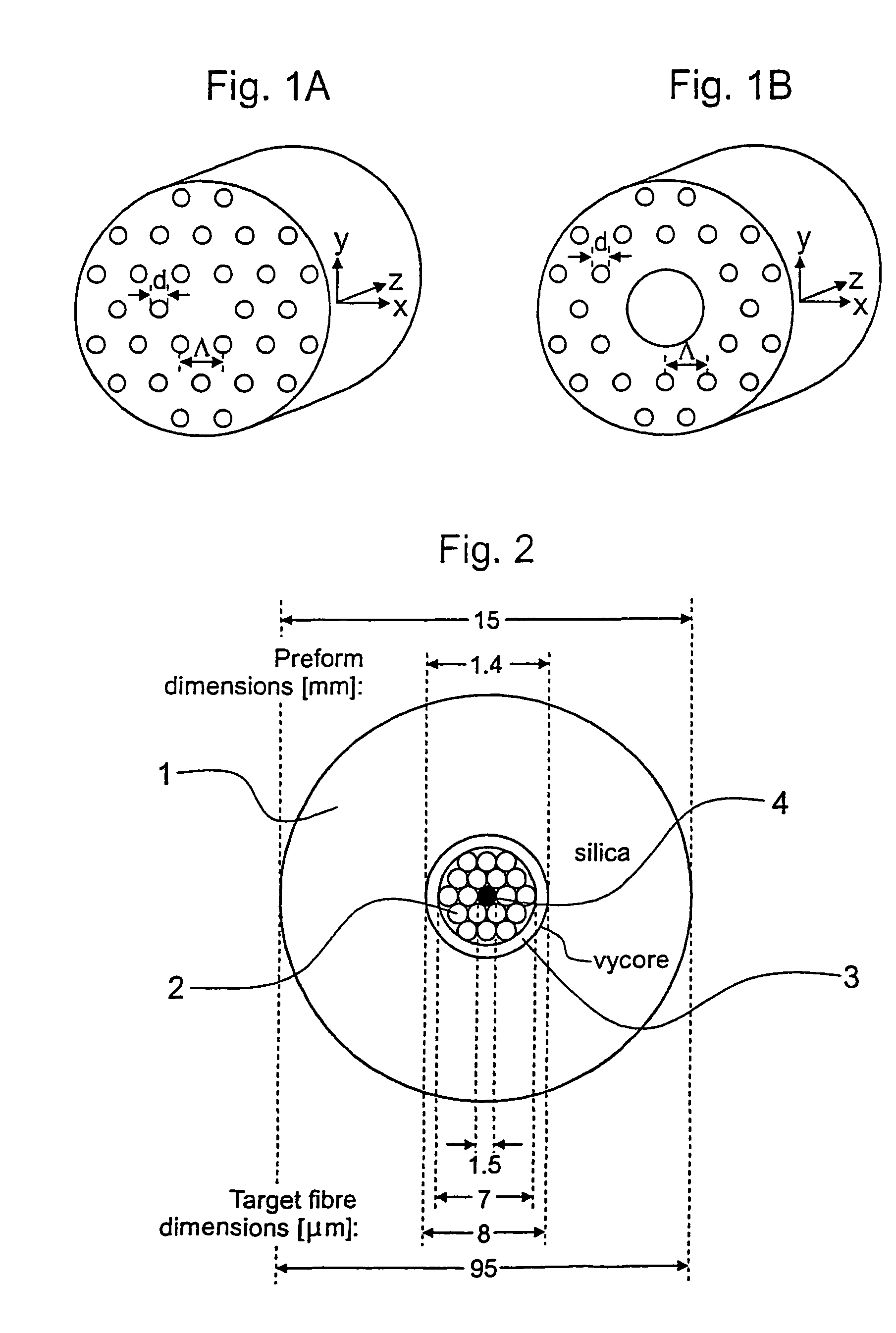
Holey optical fibres
An optical fiber structure having a holey fiber arranged in a holey outer support structure made up of holey tubes encased in a thin walled outer jacket. The holey fiber may have a solid core surrounded by a holey cladding having a plurality of rings of holes. With the invention it is possible to produce robust, coated and jacketed fibers with microstructured core features of micrometer size relatively easily using existing fiber fabrication technology. This improvement is a result of the outer holey structure which reduces the thermal mass of the supporting structure and makes it possible to reliably and controllably retain small hole features during the fiber fabrication process.
Owner:UNIV OF SOUTHAMPTON
System and method for removal of material from a blood vessel using a small diameter catheter
InactiveUS20080228209A1Increase flexibilityMaximize supportCannulasExcision instrumentsThrombusThree vessels
This invention provides a small diameter snare device and device for thrombus removal consisting of a hollow, elongate, thin-walled outer sheath. A single central core wire extends through the entire length of the sheath. The outer diameter of the core wire is sized close to the inner diameter of the sheath while allowing for axial sliding, in order to maximize the support to the body portion of the snare device. A tool tip or “capture segment” at the distal end of the sheath and core wire can be controllably expanded to engage a thrombus and remove the thrombus from the blood vessel.
Owner:VASCULAR SOLUTIONS
Plasma processing apparatus
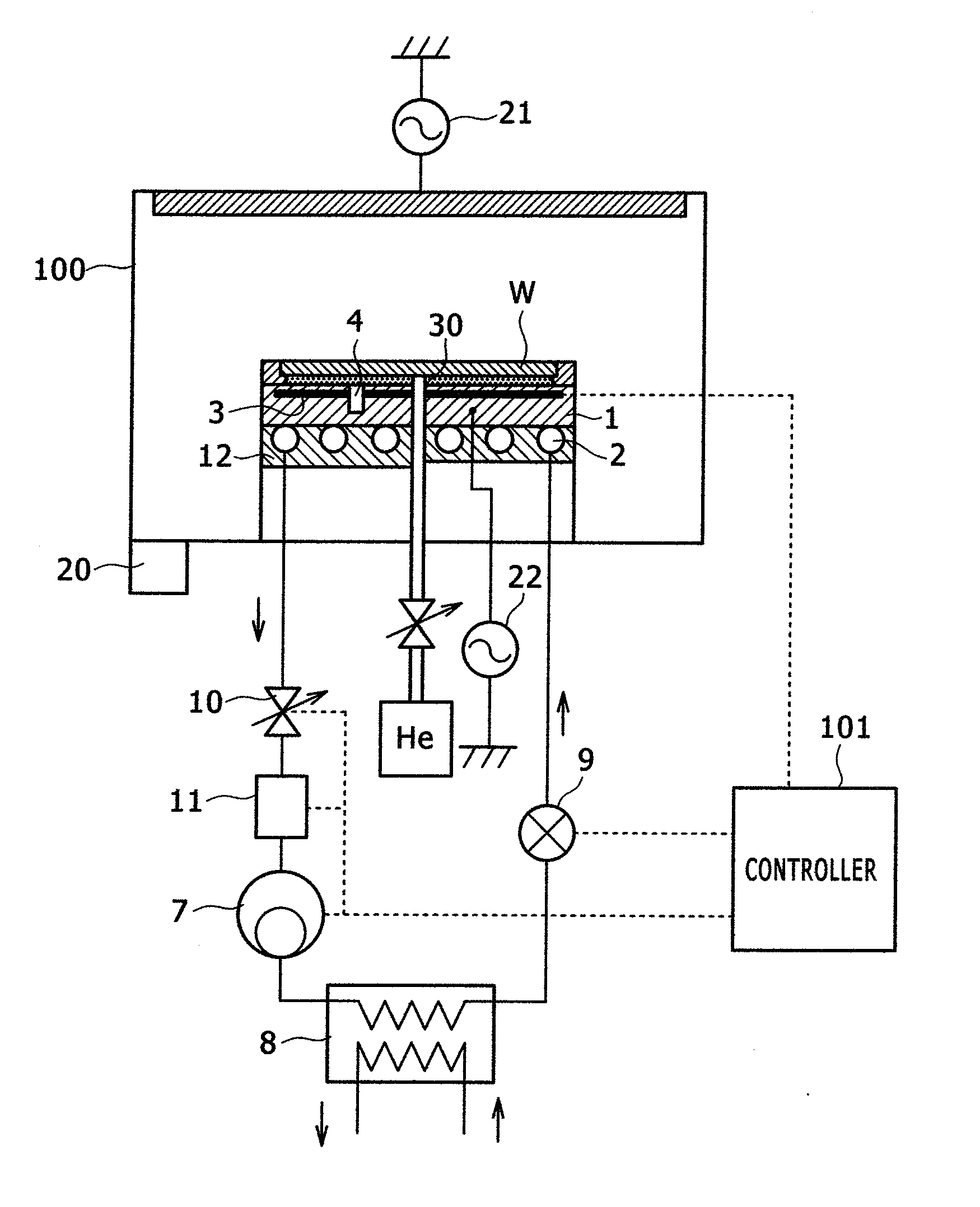
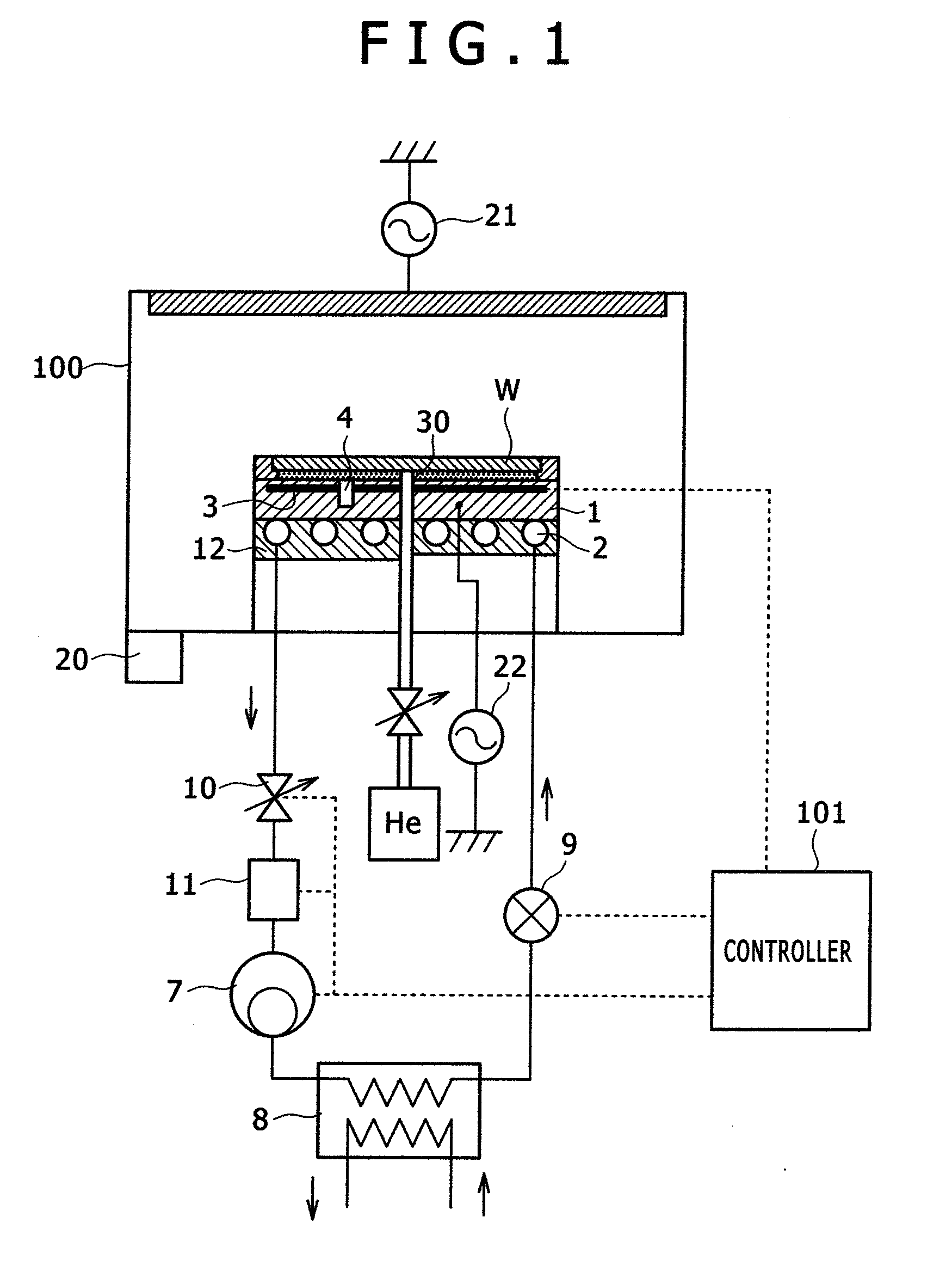
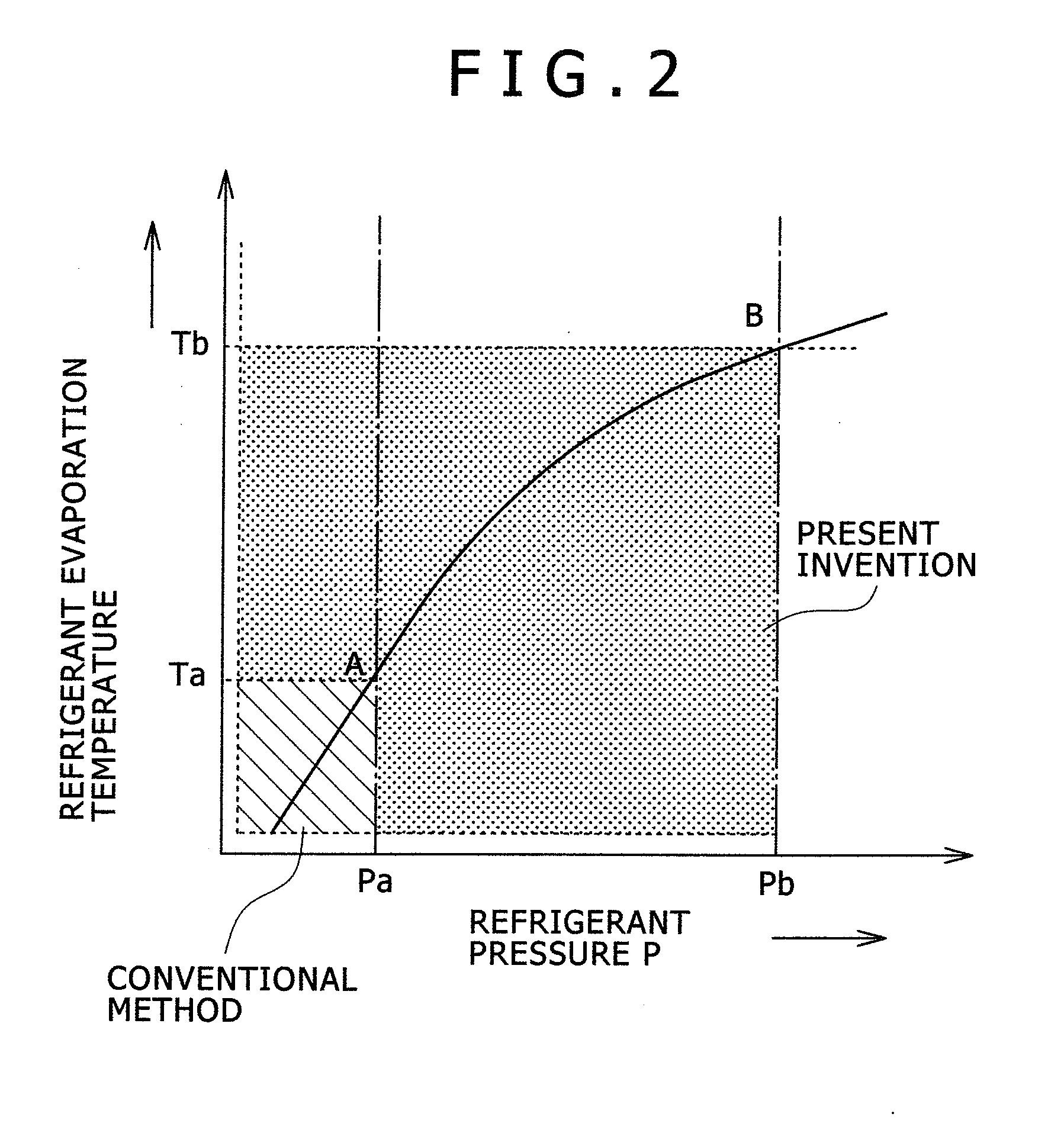
ActiveUS20080289767A1High bias powerWide temperature rangeMechanical apparatusDomestic cooling apparatusIn planeEtching
The present invention provides a temperature control unit for an electrostatic adsorption electrode that is capable of controlling the wafer temperature rapidly over a wide temperature range without affecting in-plane uniformity while high heat input etching is conducted with high wafer bias power applied. A refrigerant flow path provided in the electrostatic adsorption electrode serves as an evaporator. The refrigerant flow path is connected to a compressor, a condenser, and a first expansion valve to form a direct expansion type refrigeration cycle. A second expansion valve is installed between the electrostatic adsorption electrode and the compressor to adjust the flow rate of a refrigerant. This makes it possible to compress the refrigerant in the refrigerant flow path of the electrostatic adsorption electrode and adjust the wafer temperature to a high level by raising the refrigerant evaporation temperature. Further, a thin-walled cylindrical refrigerant flow path is employed so that the thin-walled cylinder is deformed only slightly by the refrigerant pressure.
Owner:HITACHI HIGH-TECH CORP
Eversion resistant sleeves
ActiveUS20060161187A1Reduce coefficient of frictionIncrease stiffnessOesophagiIntravenous devicesThin walledDigestive tract
The invention relates to improved means for preventing eversion and subsequent obstruction of thin-walled, floppy gastrointestinal liners implanted in the digestive tract of an animal. The implantable devices include an anchor adapted for attachment within a natural body lumen and a thin-walled, floppy sleeve open at both ends and defining a lumen therebetween. A substantial length of the sleeve has material characteristics that result in the sleeve being prone to eversion in the presence of retrograde pressures. Exemplary eversion-resistant features provide an increased stiffness and / or an increased friction coefficient between the anchor and the proximal end of the sleeve to resist eversion. In some embodiments, the eversion-resistant feature includes an anti-buckling element, such as a wire coupled along the substantial length of the sleeve.
Owner:GI DYNAMICS
Device for measuring pressure points to be applied by a compressive orthotic device
The device comprises a rigid former reproducing the volume of a portion of the body and suitable for receiving the compressive orthosis. The former (10) incorporates a plurality of sensors (22) distributed over various points of the former and configured in such a manner as to avoid significantly modifying the surface profile of the former, the sensors essentially measuring the pressure applied locally on the former by the orthosis at the location of the sensor and perpendicularly to the surface of the former. Advantageously, at the location of the measurement point, each sensor comprises a thin wall capable of being subjected to microdeformation under the effect of the pressure applied by the orthosis, and means such as a strain gauge bridge, for example. The thin wall can constitute a portion of a support pellet which is fitted to the former in such a manner that its outside surface, which includes the thin wall, is flush with the outside surface of the former.
Owner:INNOTHERA TOPIC INT
Device and method for vascular access
ActiveUS20060064159A1Reduce kinksReduce kinking in said catheter portionOther blood circulation devicesSurgeryVeinGraft portion
Vascular access systems for performing hemodialysis are disclosed. The vascular access system contemplates a catheter section adapted for insertion into a vein and a graft section adapted for attachment to an artery. The catheter section may have metal or polymer wall reinforcements that allow the use of thin-walled, small outer diameter conduits for the vascular access system. One or more of the adhered, embedded or bonded conduit reinforcement structures may be removable without significant damage to the conduit sections to facilitate attachment of the sections, or to a connector between the sections. Various self-sealing materials are provided for use in the vascular access system, as well as temporary access sites and flow control / sensor systems.
Owner:MERIT MEDICAL SYST INC
System and method for removal of material from a blood vessel using a small diameter catheter
InactiveUS20070118165A1Increase flexibilityMaximize supportCannulasExcision instrumentsDistal portionThrombus
This invention provides a small diameter snare device and device for thrombus removal consisting of a hollow, elongate, thin-walled outer sheath constructed from polymer, at least at a distal part thereof. A single central core wire extends through the entire length of the sheath. The outer diameter of the core wire is sized close to the inner diameter of the sheath while allowing for axial sliding, in order to maximize the support to the body portion of the snare device. The distal end of the core wire has a tapered section of reduced diameter or cross section to provide a “guidewire-like” flexibility to the distal portion of the device. A second wire (or wires) of about fifty percent or less of the inner diameter of the sheath is (are) shaped to form a snare loop or radially extended tool tip that is attached to the distal most portion of the central core wire. The snare loop is typically circular or oval shaped and can also be multiplanar (for example, a twisted, figure eight shape) so as to increase the ability to ensnare and capture objects. The tool tip can be a planar or multi-planar, proximally directed structure that, when deployed, is adapted to engage the thrombus from behind after piercing the thrombus with the sheath while the tool tip is retracted.
Owner:VASCULAR SOLUTIONS
Anti-buckling sleeve
The invention relates to improved means for preventing buckling and therefore eversion of thin-walled, flexible, floppy gastrointestinal liners implanted in the digestive tract of an animal. The implantable devices include an anchor adapted for attachment within a natural body lumen and a thin-walled, floppy sleeve open at both ends and defining a lumen therebetween. A substantial length of the sleeve has material characteristics that result in the sleeve being prone to buckling and therefore eversion in the presence of retrograde pressures. Exemplary anti-buckling mechanisms provide an increased stiffness and / or an increased friction coefficient between the anchor and the proximal end of the sleeve to resist buckling and therefore eversion. In some embodiments, the anti-buckling mechanism is as a wire coupled along the substantial length of the sleeve.
Owner:GI DYNAMICS
Ventilation interface for sleep apnea therapy
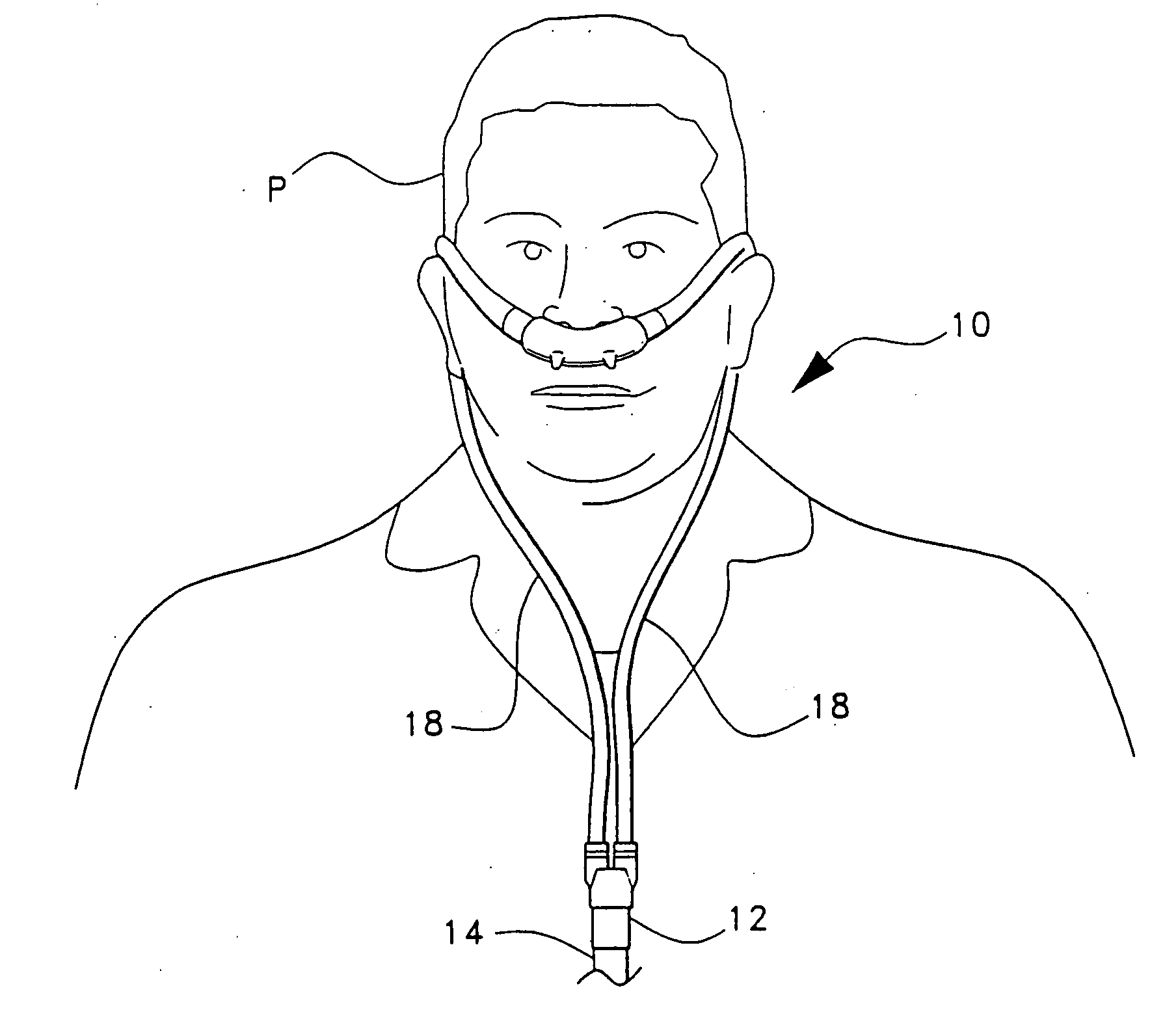
InactiveUS20070137653A1Great volume of airIncrease volumeRespiratory masksMedical devicesThin walledAirflow
The ventilation interface for sleep apnea therapy interfaces a ventilation device to the patient's airways. The ventilation interface includes a pair of nasal inserts made from flexible, resilient silicone which are oval shaped in cross-section and slightly tapered from a base proximal the ventilation supply to the distal tip end. A bead flange is disposed about the exterior of each insert at the distal end of the insert. A bleed port for release of exhaled air is defined through a conical vent projecting normally to the path of the incoming air flow, and continues through a nipple extending to the exterior of the air conduit. In one embodiment, a pair of nasal inserts are integral with a nasal cannula body, with bleed ports axially aligned with each insert. In another embodiment, each insert is independently connected to a separate, thin-walled, flexible supply line.
Owner:WOOD THOMAS J
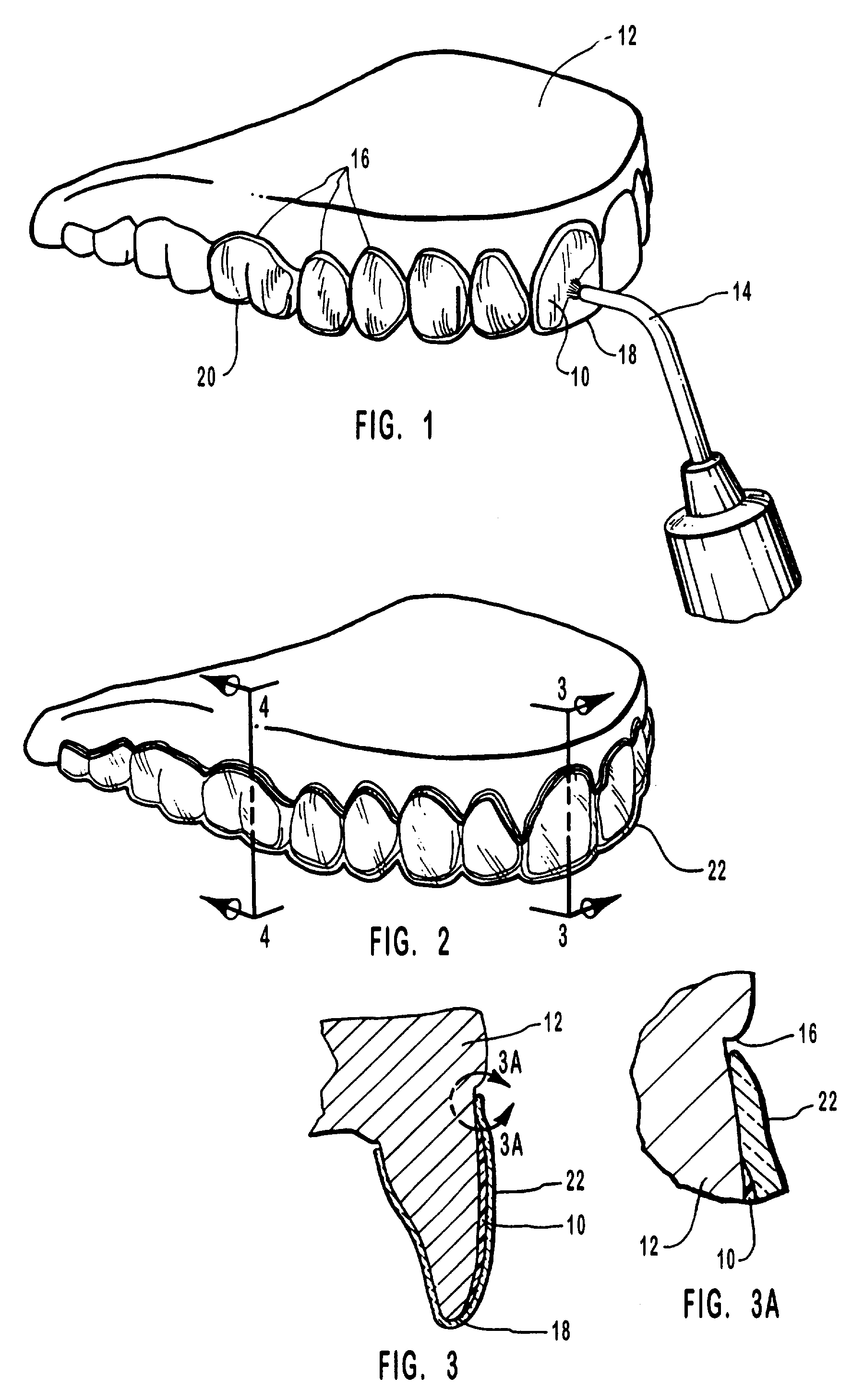
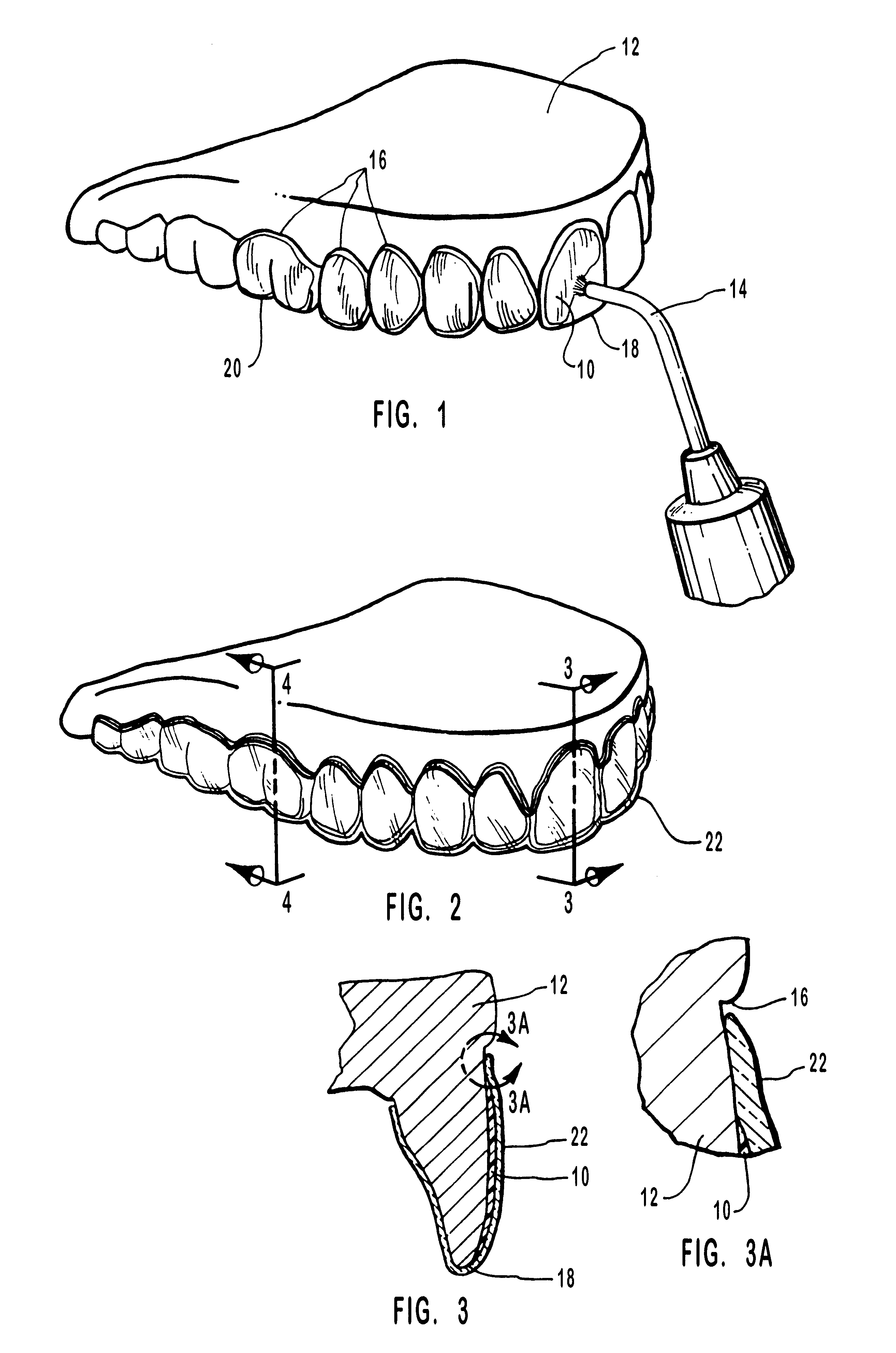
Methods for bleaching, opacifying and desensitizing teeth
InactiveUS6368576B1Trend downReduce sensitivityCosmetic preparationsTeeth fillingPotassium nitrateCarbamide peroxide
Owner:ULTRADENT PROD INC
Endovascular graft with differentiable porosity along its length
Medical devices, and in particular implantable medical devices, may be coated to minimize or substantially eliminate a biological organism's reaction to the introduction of the medical device to the organism. The medical devices may be coated with any number of biocompatible materials. Therapeutic drugs, agents or compounds may be mixed with the biocompatible materials and affixed to at least a portion of the medical device such as a stent-graft. These therapeutic drugs, agents or compounds may also further reduce a biological organism's reaction to the introduction of the medical device to the organism. In addition, these therapeutic drugs, agents and / or compounds may be utilized to promote healing, including the formation of blood clots. A stent-graft fabricated from a thin-walled, high strength material provides for a more durable and lower profile endoprosthesis. The stent-graft comprises one or more stent segments covered with a fabric formed by the weaving, knitting or braiding of a biocompatible, high tensile strength, abrasion resistant, highly durable yarn such as ultra high molecular weight polyethylene. The one or more stent segments may be balloon expandable or self-expanding. The fabric may be attached to the stent segments utilizing any number of known materials and techniques. In addition, the pore size of the graft material may be varied.
Owner:CORDIS CORP
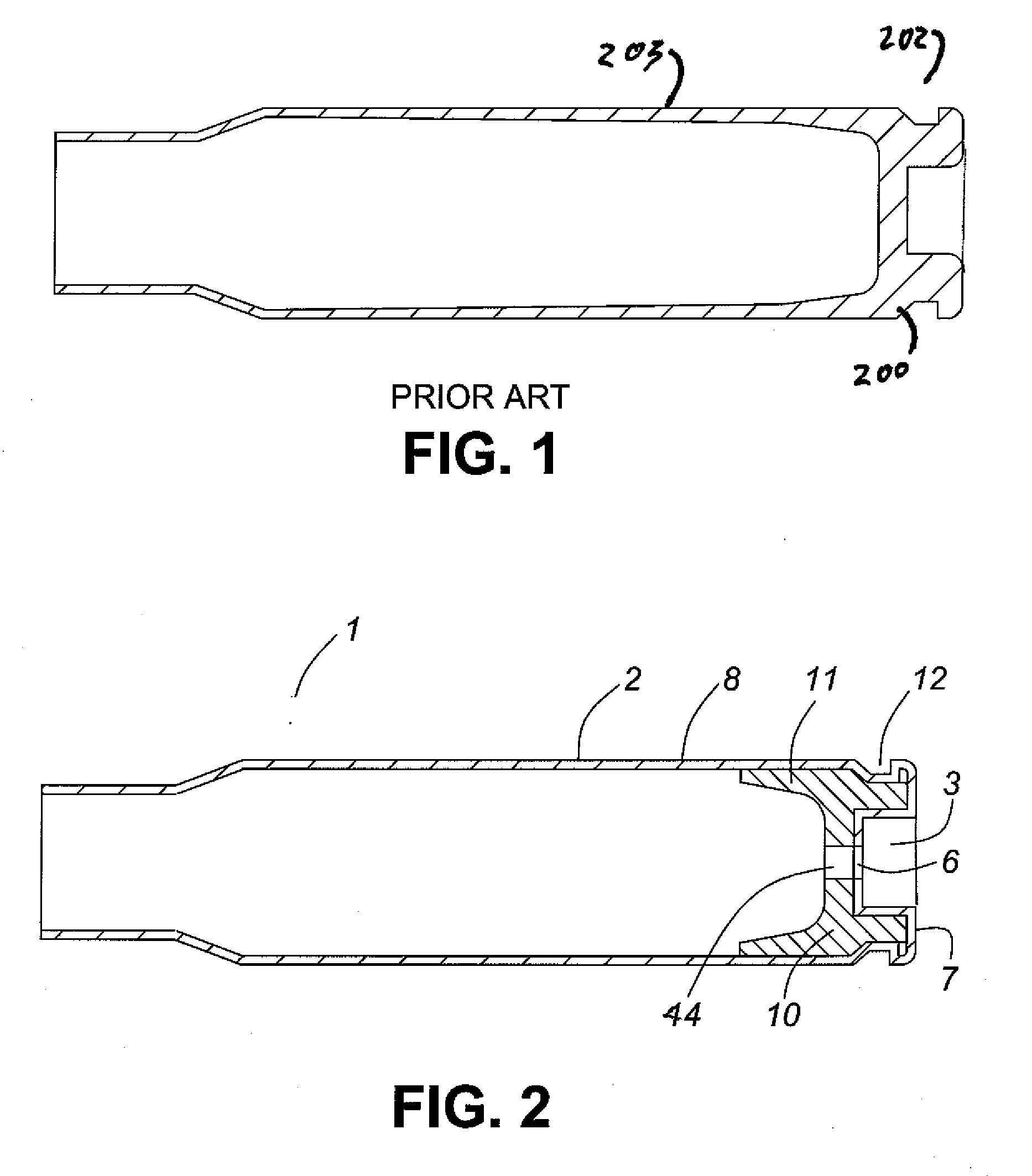
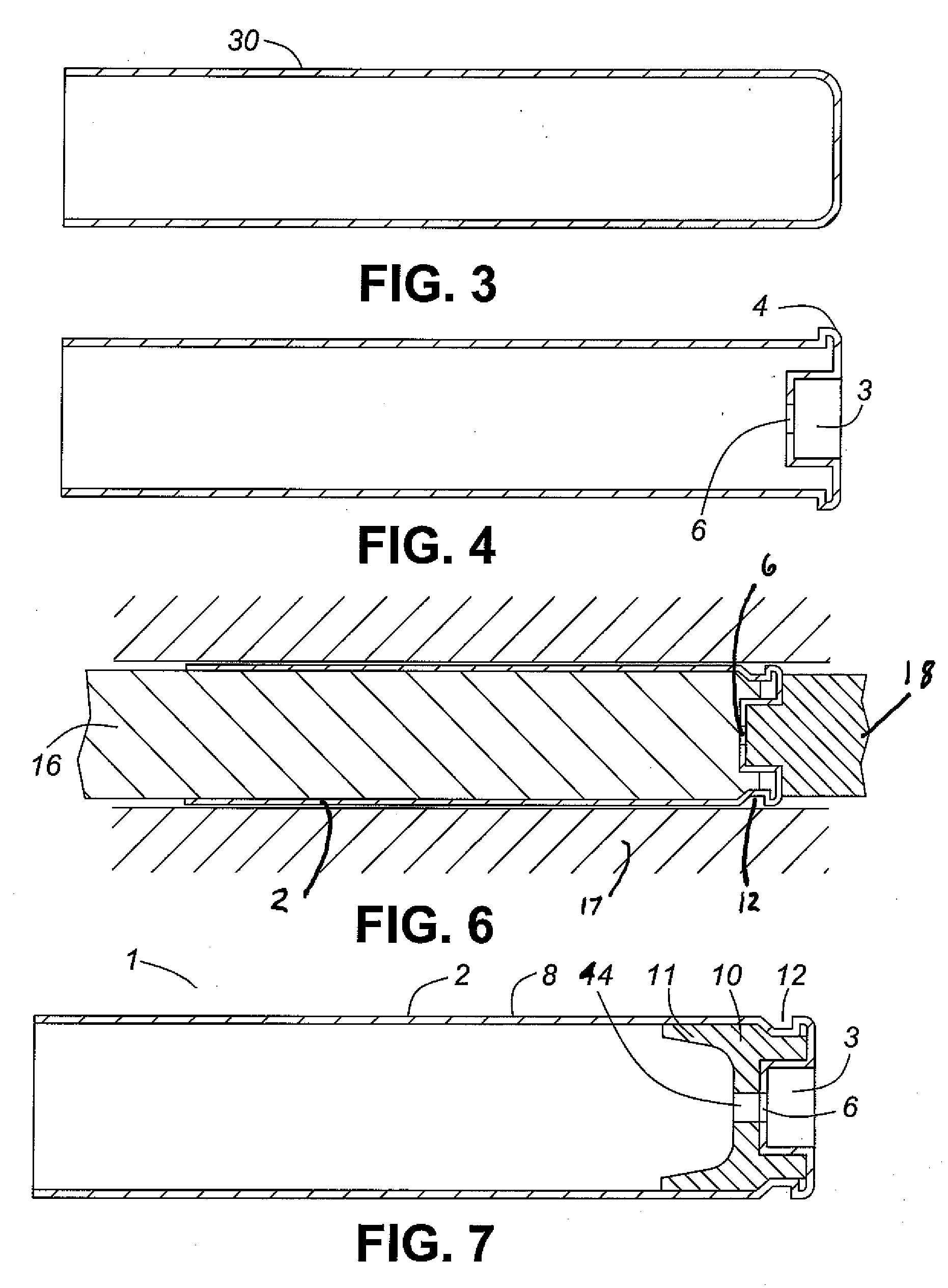
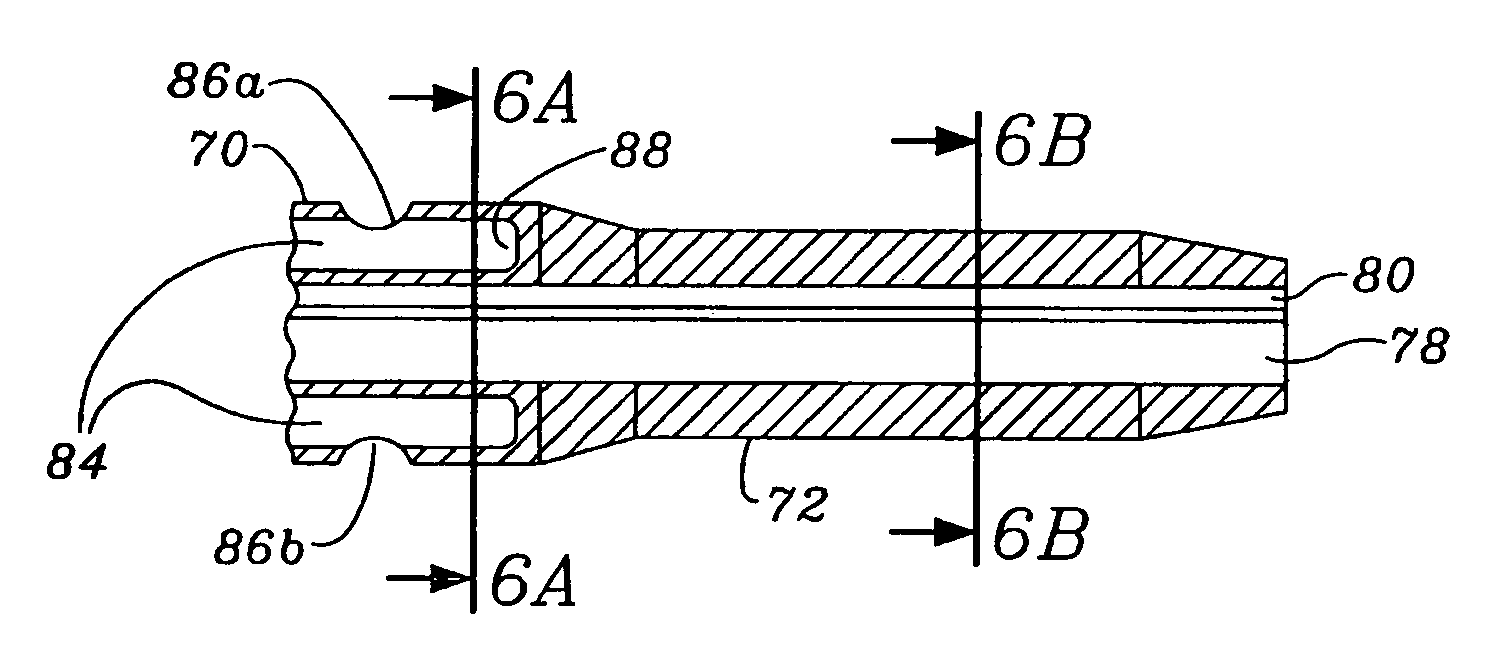
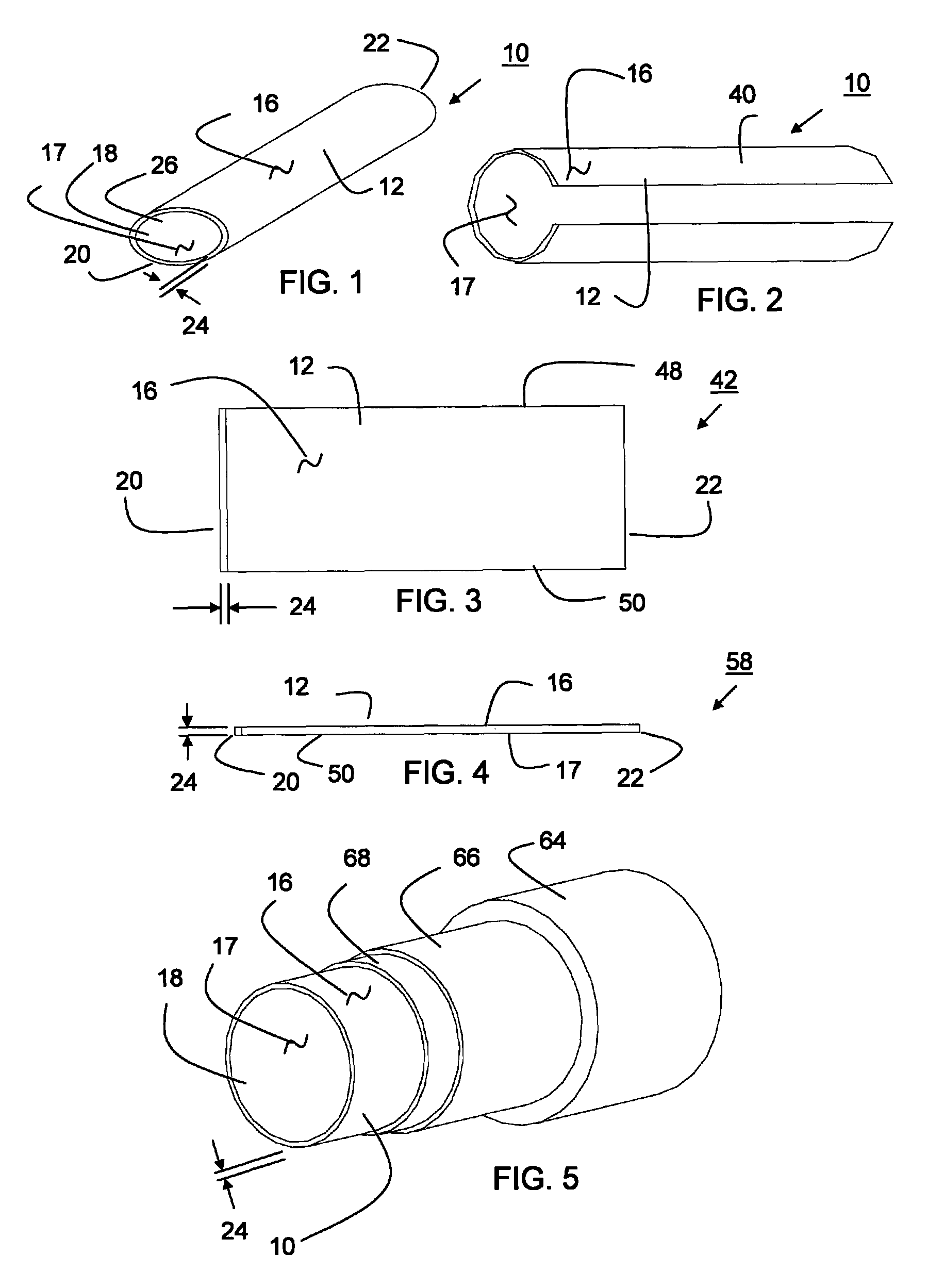
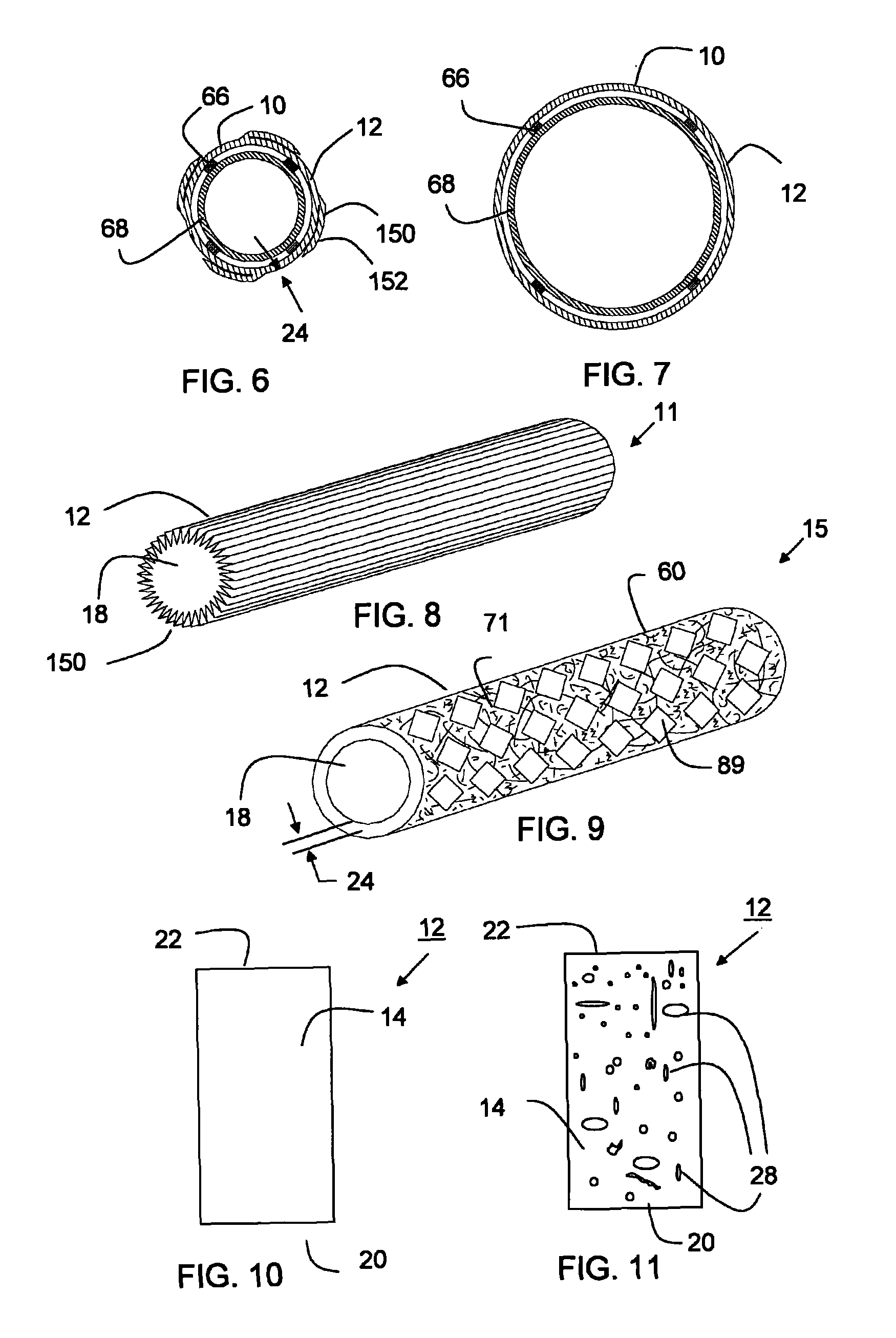
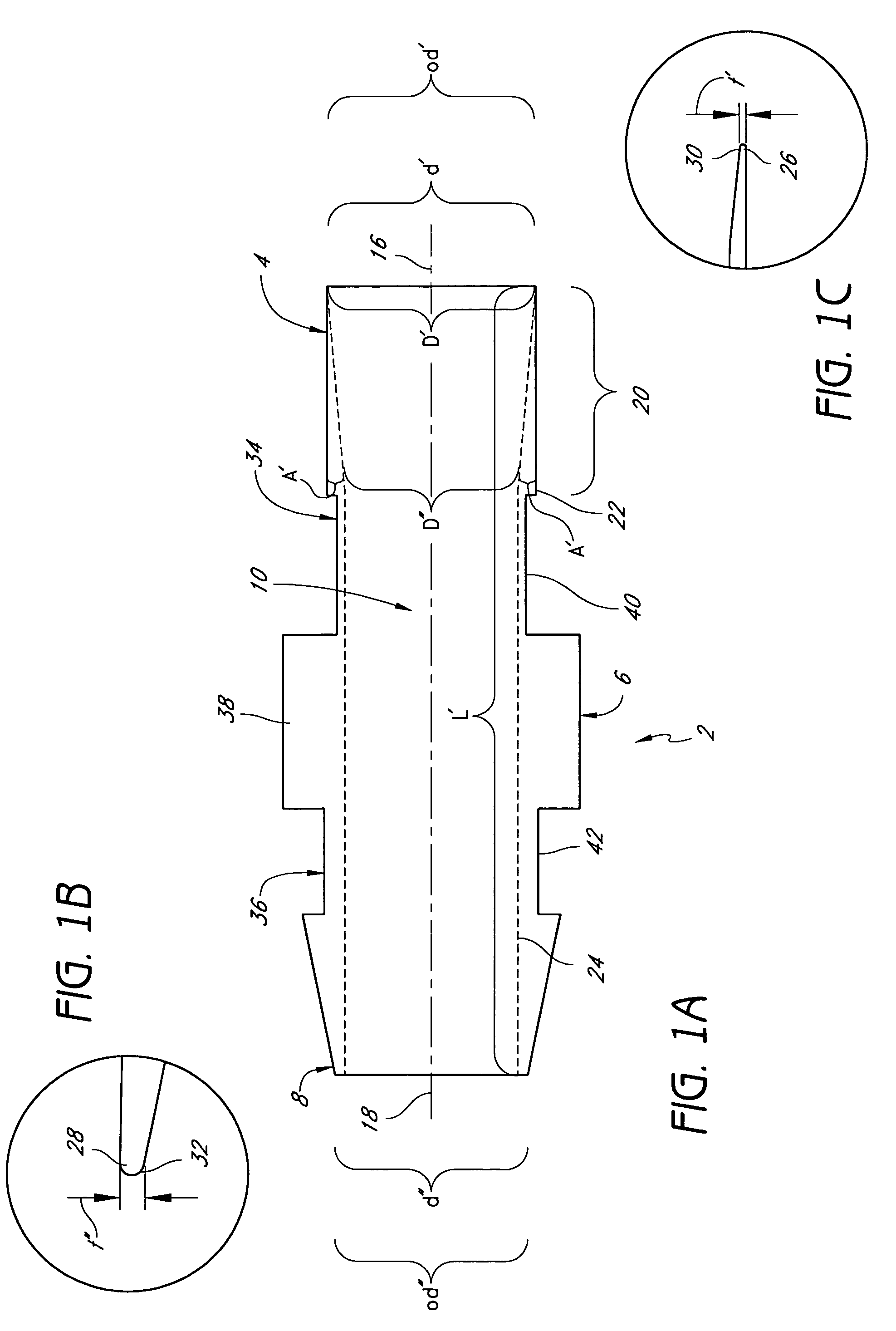
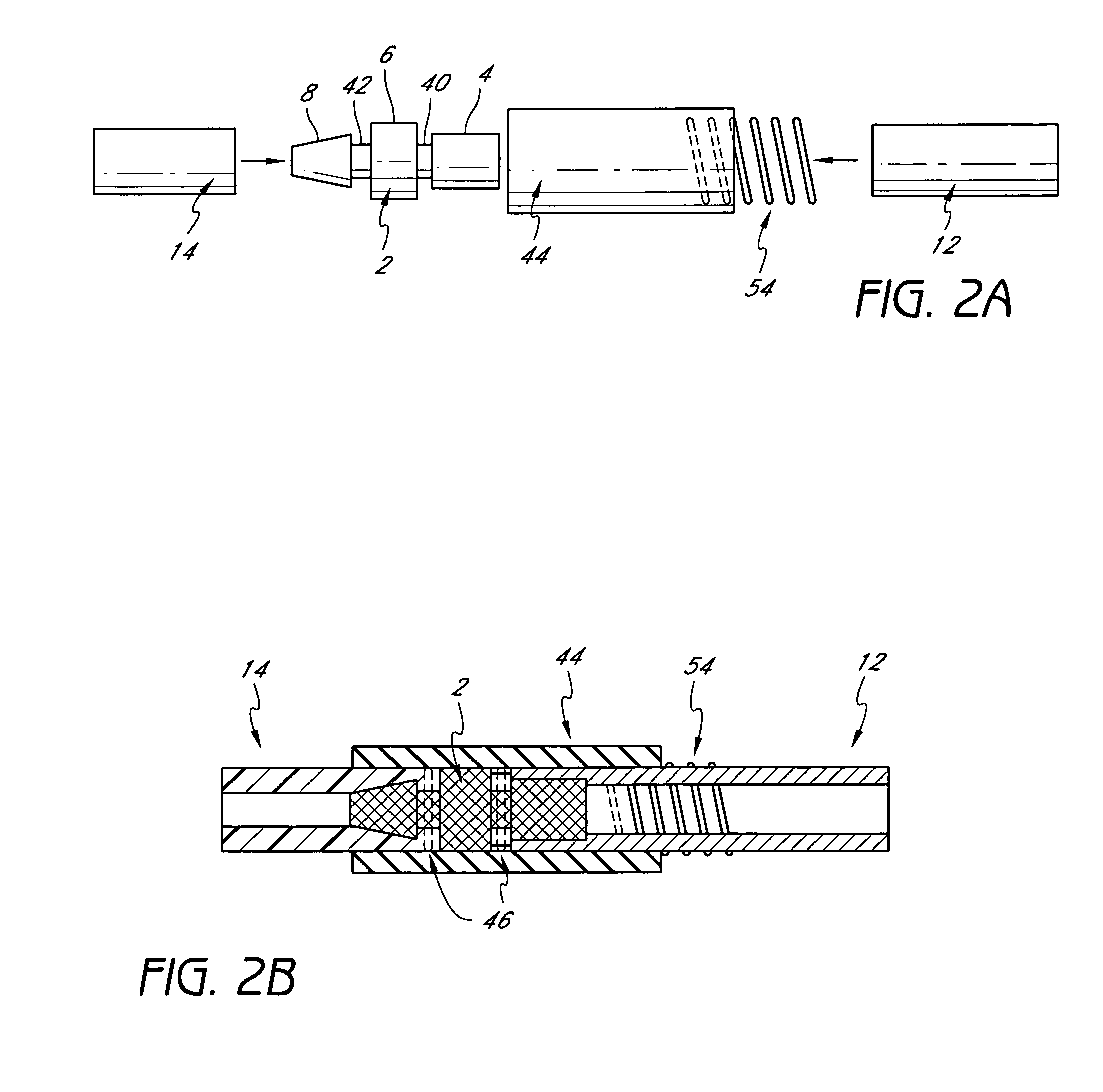
Thin walled, two component cartridge casing
A cartridge or ammunition casing is made of two components, one forming the outer casing sleeve and head end and the other comprising a plug seated within the head end to protect the outer casing material from propellant gases. The outer cylindrical sidewall and end panel of the casing may be made of a material selected from the group consisting of stainless steel, steel, pre-coated carbon steel, brass or brass-type alloys, aluminum, hardened aluminum alloys, and suitable polymeric plastic material such as nylon derivatives and VECTRA™. The inner plug may be made of a material selected from the group consisting of aluminum, brass, steel, stainless steel, and suitable polymeric materials.
Owner:TECH CORPORATION CO LTD
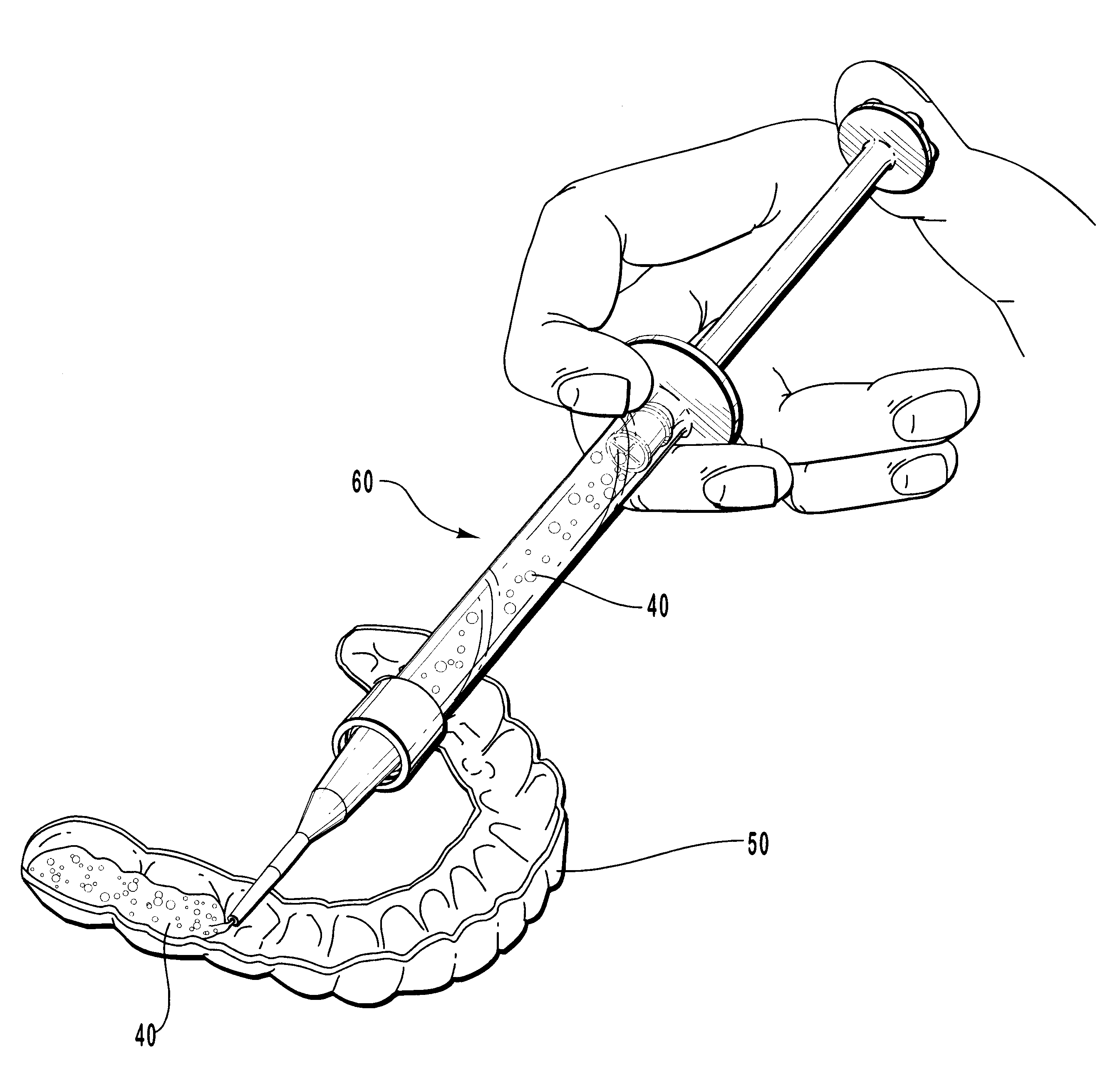
One-part dental compositions and methods for bleaching and desensitizing teeth
InactiveUS6309625B1Reduce sensitivityTrend downCosmetic preparationsGum massagePotassium nitrateMedicine
Composition and methods that include potassium nitrate for whitening and / or reducing tooth sensitivity. The dental compositions may optionally include a dental bleaching agent, such as hydrogen peroxide or carbamide peroxide. The dental compositions may be applied directly to the person's teeth, or they may be loaded into a comfortable fitting, flexible, thin-walled dental tray and placed over the person's teeth. In that case, the dental compositions will include a tackifying agent, such as carboxypolymethylene, dispersed within a solvent, which assists the composition in retaining the dental tray over the person's teeth as a result of the adhesive properties of the dental composition rather than due to mechanical interlocking of the tray over the person's teeth. The dental compositions may further include anticariogenic and antimicrobial agents.
Owner:ULTRADENT PROD INC
Expandable guide sheath and apparatus and methods using such sheaths
Apparatus and methods are provided for accessing body lumens and / or for delivering instruments into body lumens, e.g., vessels within a patient's vasculature. A flexible sheath is provided that is expandable from a contracted condition to an enlarged condition wherein the sheath at least partially defines a lumen therein. The sheath is lubricious and has a relatively thin wall, thereby providing a collapsible / expandable guide for delivering fluids and / or instruments through tortuous anatomy and / or into relatively narrow passages. The sheath is advanced from an entry site to a body lumen in the contracted condition. Once the sheath reaches a target body lumen, the sheath is expanded to the enlarged condition, thereby defining a lumen within the sheath, and fluids and / or instruments are introduced into the body lumen via the sheath lumen. Upon completing the procedure, the sheath is removed from the body lumen.
Owner:THE BOARD OF TRUSTEES OF THE LELAND STANFORD JUNIOR UNIV
Multiple lumen catheter having a soft tip
A multiple lumen catheter having a soft, tapered multiple lumen distal tip for insertion into a body vessel. One of the lumens is sized to pass over a guidewire such that the catheter can be inserted into the body vessel using the Seldinger technique. At least one medical implement lumen is used for placement or positioning of a biomedical sensor or other medical implement. For example, at least one optical fiber passing through the medical implement lumen may transmit and receive light at the distal tip for measuring oxygen saturation of the blood. The catheter may have a cylindrical catheter body to which the soft distal tip attaches. The soft tip reduces the possibility of vessel or tissue puncture and abrasion. The tip is constructed of a soft plastic or pliable material that yields easily when force is applied. For example, the tip may be made of a softer material than the catheter body, or if made of the same material, the tip can be configured with thinner walls or a higher air-to-material ratio cross-section. Various geometrical configurations and combinations of materials can be used to decrease the flexible resistance of the tip to an applied load. One particular useful application for the catheter of the present invention is as a central venous catheter equipped with fibers for measuring oximetry. The fibers extend to the distal end of the tip and are preferably secured therein with minimal adhesive so as to limit the stiffness added to the tip. One particular useful construction is to secure the fibers within the medical implement lumen using adhesive only along a length of between about 0.5–3.5 mm.
Owner:EDWARDS LIFESCIENCES CORP
Fibrillated biodegradable prosthesis
The present invention is a more durable expanded material that enables thinner wall thicknesses and a more flexible reinforcement suitable for stenting. The present invention is especially useful in the construction of grafts, stents, and stent-grafts which are used, for example, in repairing or replacing blood vessels that are narrowed or occluded by disease, aneurismal blood vessels, or other medical treatments. The inventive material and configurations allow expansion or contraction in size or adjustment in size in an incremental manner so that the optimum size, shape, and fit with other objects can be obtained. The present invention is also optionally capable of more accurately delivering one or more active ingredients such as drugs over longer periods of time. The present invention optionally includes surface modifications and additives that increase the surface adhesion of active ingredients, coatings, or combinations thereof. Finally, the present invention optionally includes growing cells on the inventive material so that the expanded material, reinforcement, or combinations thereof are useful, for example, in producing lab-grown blood vessels or organs.
Owner:SCANLON JOHN JAMES +1
Compositions and methods for whitening and desensitizing teeth
InactiveUS6306370B1Reduce sensitivityTrend downCosmetic preparationsImpression capsPotassium nitrateMedicine
Composition and methods that include potassium nitrate for whitening and / or reducing tooth sensitivity. The dental compositions may optionally include a dental bleaching agent, such as hydrogen peroxide or carbamide peroxide. The dental compositions may be applied directly to the person's teeth, or they may be loaded into a comfortable fitting, flexible, thin-walled dental tray and placed over the person's teeth. In that case, the dental compositions will include a tackifying agent, such as carboxypolymethylene, which assists the composition in retaining the dental tray over the person's teeth as a result of the adhesive properties of the dental composition rather than due to mechanical interlocking of the tray over the person's teeth. The dental compositions may further include anticariogenic and antimicrobial agents.
Owner:ULTRADENT PROD INC
Injection molding of polymeric material
Injection molding systems and methods useful for making microcellular foamed materials are provided as well as microcellular articles. Pressure drop rate and shear rate are important features in some embodiments, and the invention provides systems for controlling these parameters in an injection molding system. Another aspect involves an injection molding system including a nucleator that is upstream of a pressurized mold. Another aspect involves an extrusion system with the reciprocating screw for forming a single phase solution of non-nucleated blowing agent and polymeric material. Another aspect involves very thin walled microcellular material and very thin walled polymeric material. Another aspect provides a method for producing high weight reductions in very thin-walled parts with surfaces that have no noticeable differences from non-foamed parts.
Owner:TREXEL
Kinetic stent
The disclosed stent is formed of an elongate, flexible duct having a very thin wall and a preformed diameter, length, and shape. The stent is constructed of a woven tubular structure of multiple strands or elements. The woven tubular structure is thermally set to a predetermined diameter and length, so that the "at rest" or natural condition of the tubular structure is predictable. A retention or holding member can be formed at one or both of the ends of the stent. This retention member can be reduced in diameter for insertion into the body passage. The woven tubular structure provides a path for fluids to flow in and around the stent, while a patent lumen is being developed. The woven tubular structure allows the stent to be extended or stretched over a guidewire or other noncompressive member, to thereby reduce the diameter of the stent for insertion of the stent into a body passage.
Owner:APPL MEDICAL RESOURCES CORP
Impact energy management method and system
ActiveUS7774866B2Little strengthExtension of timeChemical protectionHeat protectionElectrical resistance and conductanceEngineering
An impact-absorbing protective structure comprises one or more compressible cells. Each cell is in the form of a thin-walled plastic enclosure defining an inner, fluid-filled chamber with at least one small orifice through which fluid resistively flows. Each cell includes an initially resistive mechanism that resists collapse during an initial phase of an impact and that then yields to allow the remainder of the impact to be managed by the venting of fluid through the orifice. The initially resistive mechanism may be implemented by providing the cell with semi-vertical side walls of an appropriate thickness or by combining a resiliently collapsible ring with the cell. After the initially resistive mechanism yields to the impact, the remainder of the impact is managed by the fluid venting through the orifice. The cell properties can be readily engineered to optimize the impact-absorbing response of the cell to a wide range of impact energies.
Owner:XENITH
Methods and kits for maxillary dental anesthesia by means of a nasal deliverable anesthetic
Methods and systems for anesthetizing a portion or all of a patient's maxillary dental arch using a nasal delivered anesthetizing composition. The process generates anesthesia sufficient for facilitation of operative dentistry, endodontics, periodontics or oral surgery for teeth of the maxillary arch. The dental nasal spray process consists of inserting one or more dispensing devices through the patient's nostril and delivering metered dosages of anesthetic solution or gel into the nasal cavity. The process may utilize a single solution which is a mixture of anesthetic agents, vasoconstricting agents and other physiological inert agents or two separate solutions, wherein one solution contains the vasoconstricting agents and the other solution contains the anesthetic agents. Anesthetic diffusion through the thin walls of the nasal cavity allows for the blocking of nerve impulses originating from the maxillary dentition and surrounding tissues. Anesthesia of specific oral regions such as right versus left sides of the dental arch, anterior versus posterior teeth, and soft tissue anesthesia may be controlled through modification of the dosage volume and the selection of right or left nostril insertion and agent delivery.
Owner:ST RENATUS +1
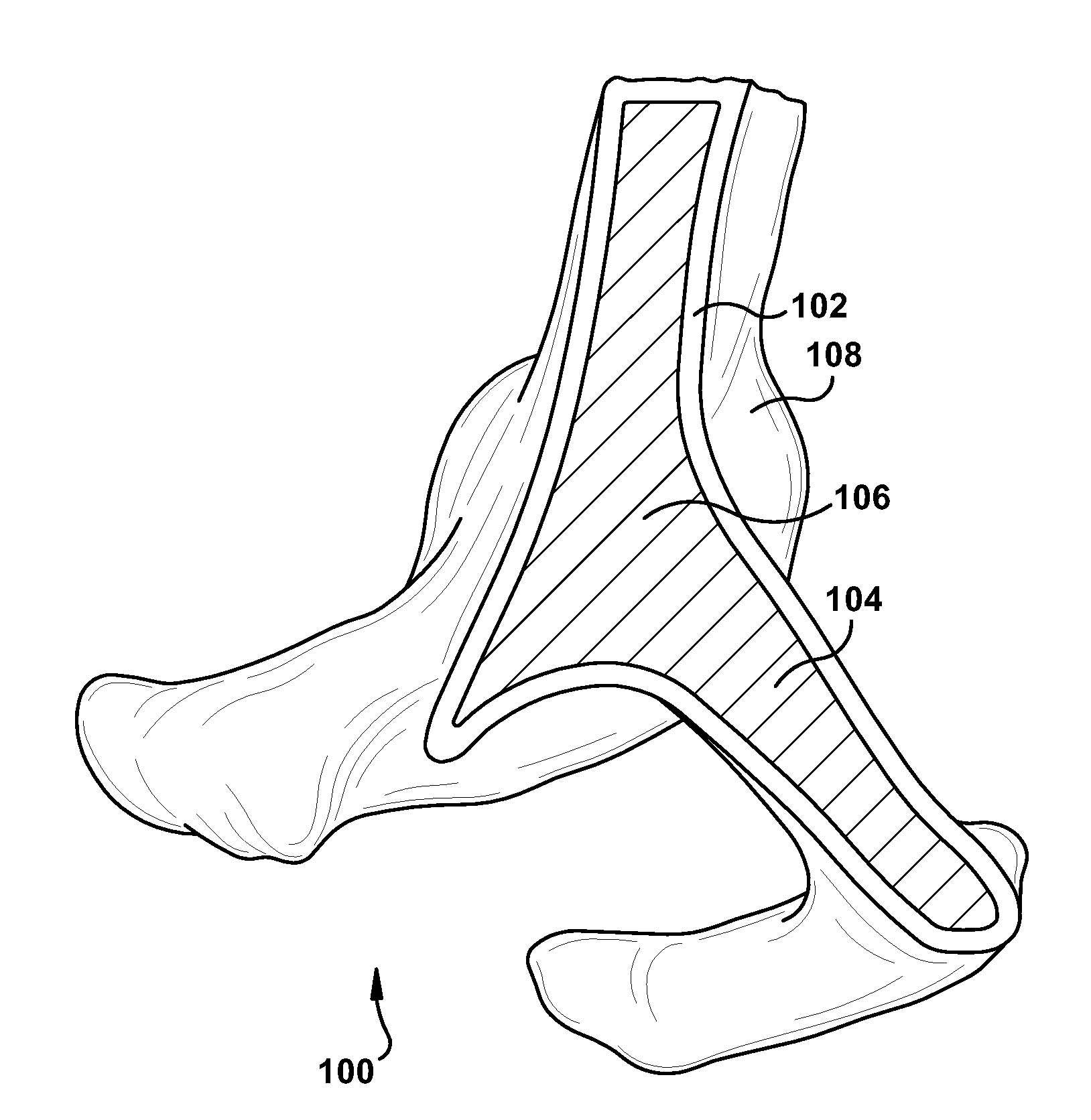
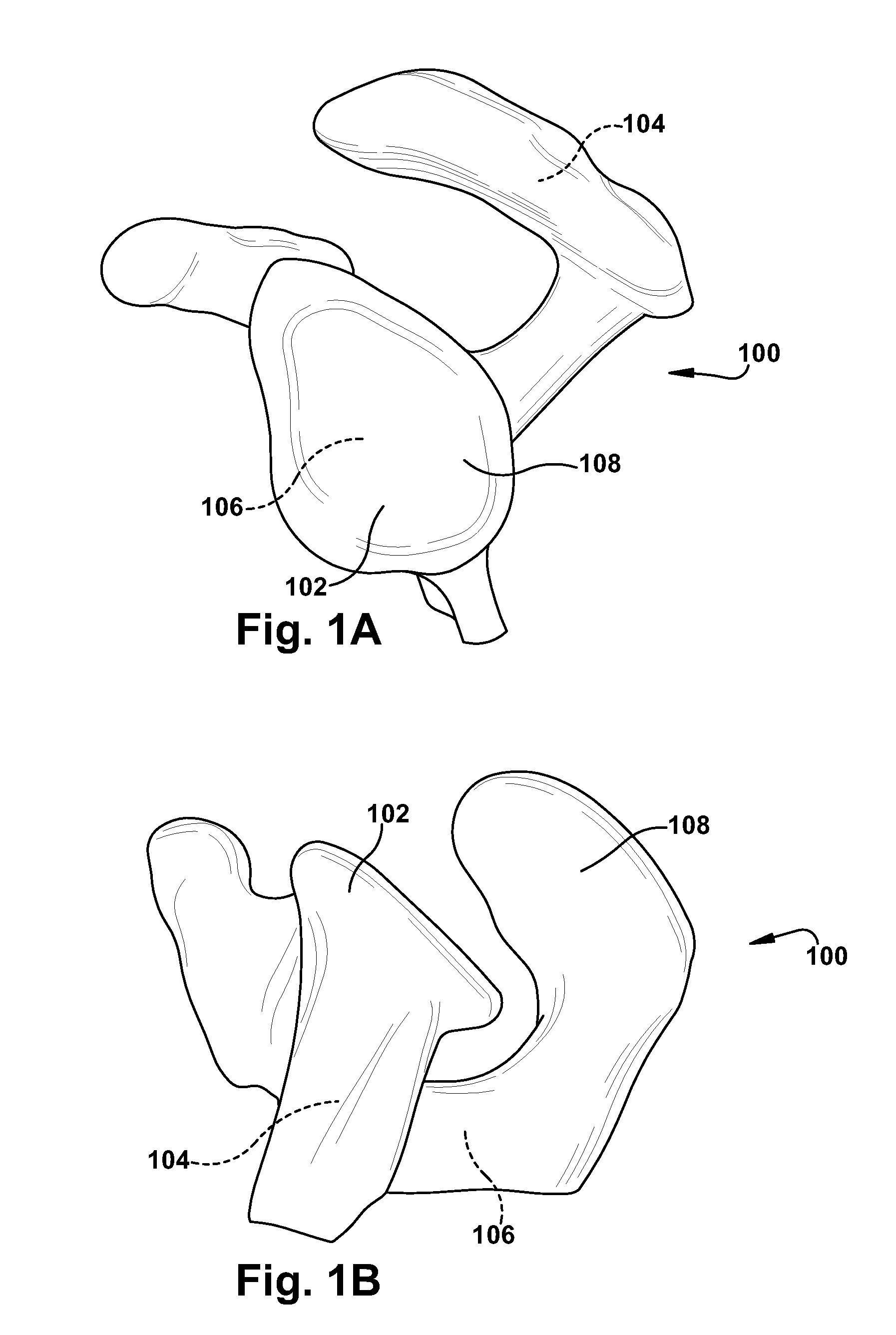
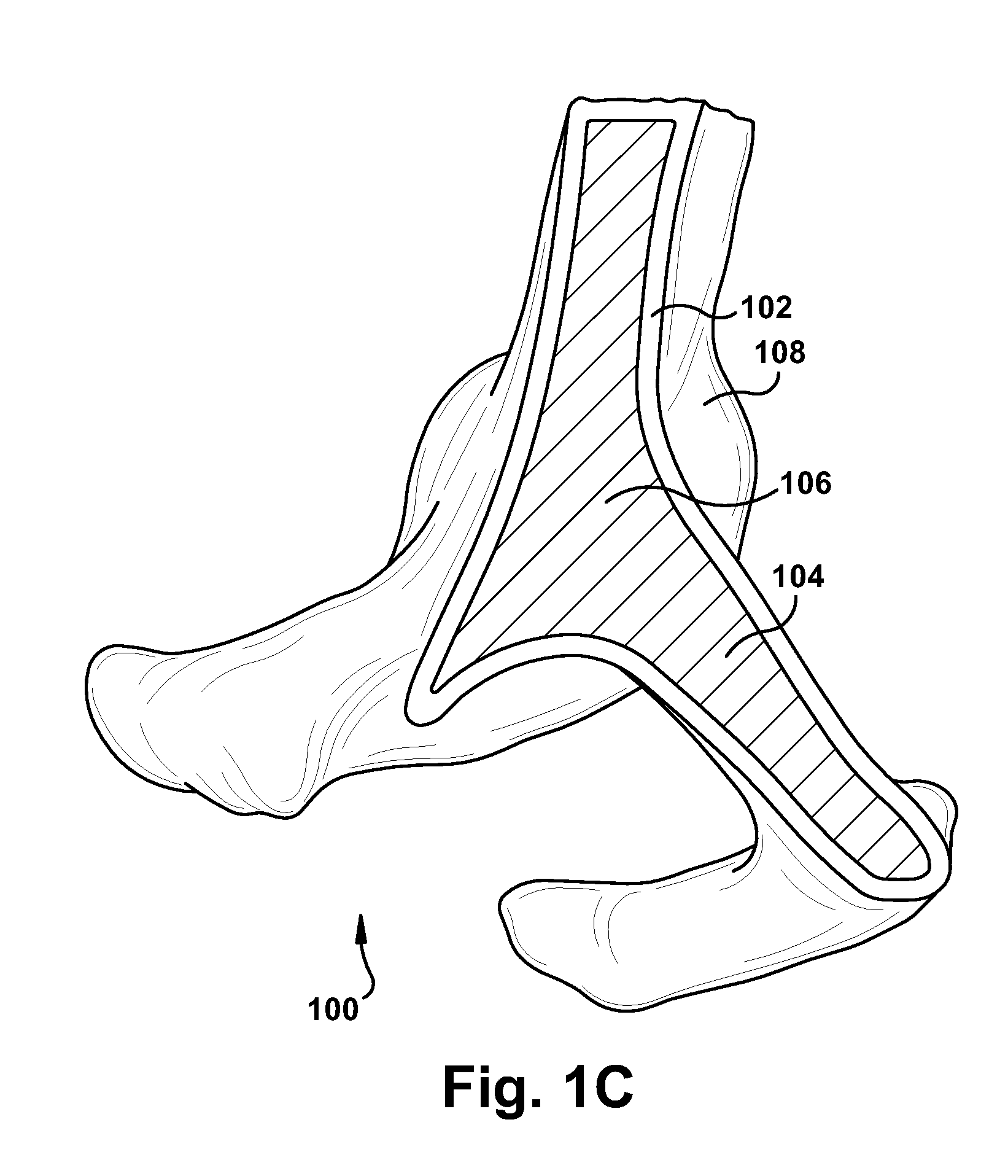
Intravascular stent device
Disclosed is a very small diameter intravascular stent, or aneurysm cover, which may be used to occlude, or partially occlude, an aneurysm in the human brain. The stent is comprised of a thin-walled skeletal tubular member formed of interconnected sinusoidal members to thereby form a pattern of cells along the skeletal tubular member. The stent also includes anchor members, which are comprised of a longitudinal leg member having a radiopaque coil disposed about the leg member, which serve to retain the stent during delivery of the stent.
Owner:CODMAN & SHURTLEFF INC
Synthetic bone model and method for providing same
InactiveUS20130085590A1Additive manufacturing apparatusEducational modelsFilling materialsSynthetic bone
A method for providing a synthetic bone model of a subject bone includes providing a file with data representing a three-dimensional subject bone. Manufacturing instructions are generated based upon at least a portion of the data. The manufacturing instructions are transferred to a manufacturing device. A thin-walled outer shell of the synthetic bone model is created directly from the manufacturing instructions using the manufacturing device. The outer shell defines an inner cavity. A filler material is placed within at least a portion of the inner cavity. A synthetic bone model is also disclosed.
Owner:THE CLEVELAND CLINIC FOUND
Device and method for vascular access
ActiveUS7762977B2Reduce kinksReduce kinking in said catheter portionOther blood circulation devicesSurgeryVeinHaemodialysis machine
Vascular access systems for performing hemodialysis are disclosed. The vascular access system contemplates a catheter section adapted for insertion into a vein and a graft section adapted for attachment to an artery. The catheter section may have metal or polymer wall reinforcements that allow the use of thin-walled, small outer diameter conduits for the vascular access system. One or more of the adhered, embedded or bonded conduit reinforcement structures may be removable without significant damage to the conduit sections to facilitate attachment of the sections, or to a connector between the sections. Various self-sealing materials are provided for use in the vascular access system, as well as temporary access sites and flow control / sensor systems.
Owner:MERIT MEDICAL SYST INC
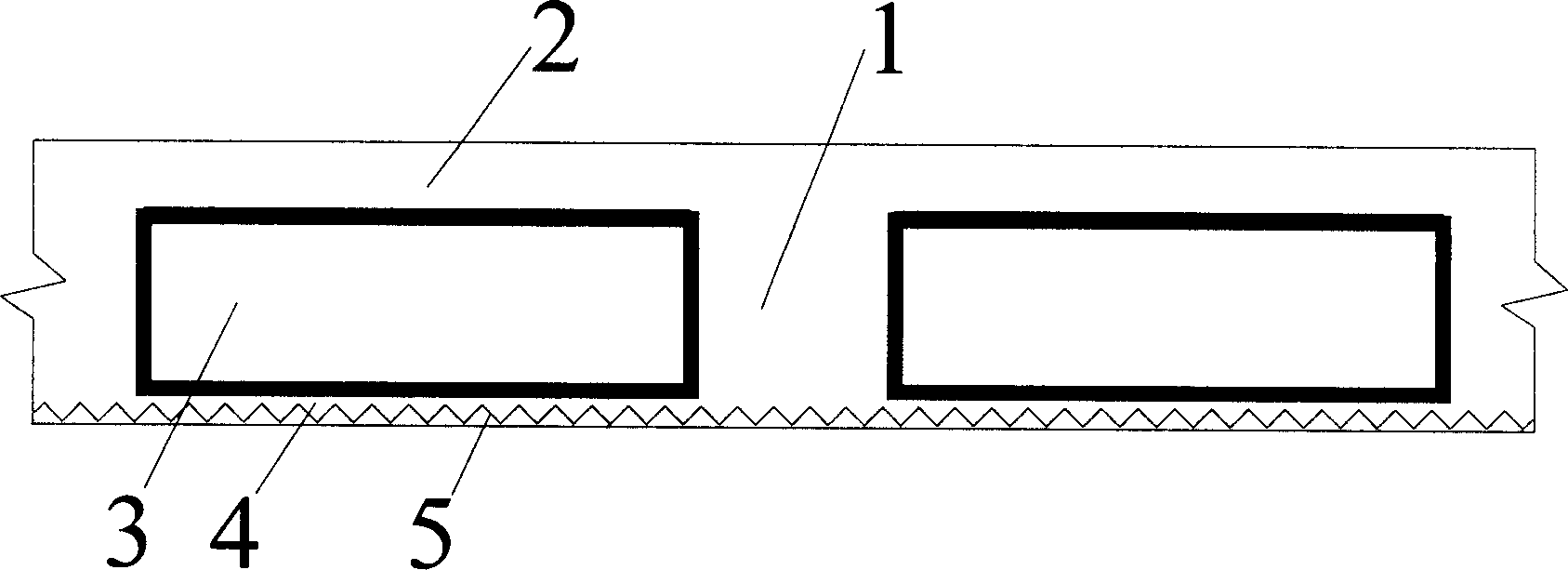
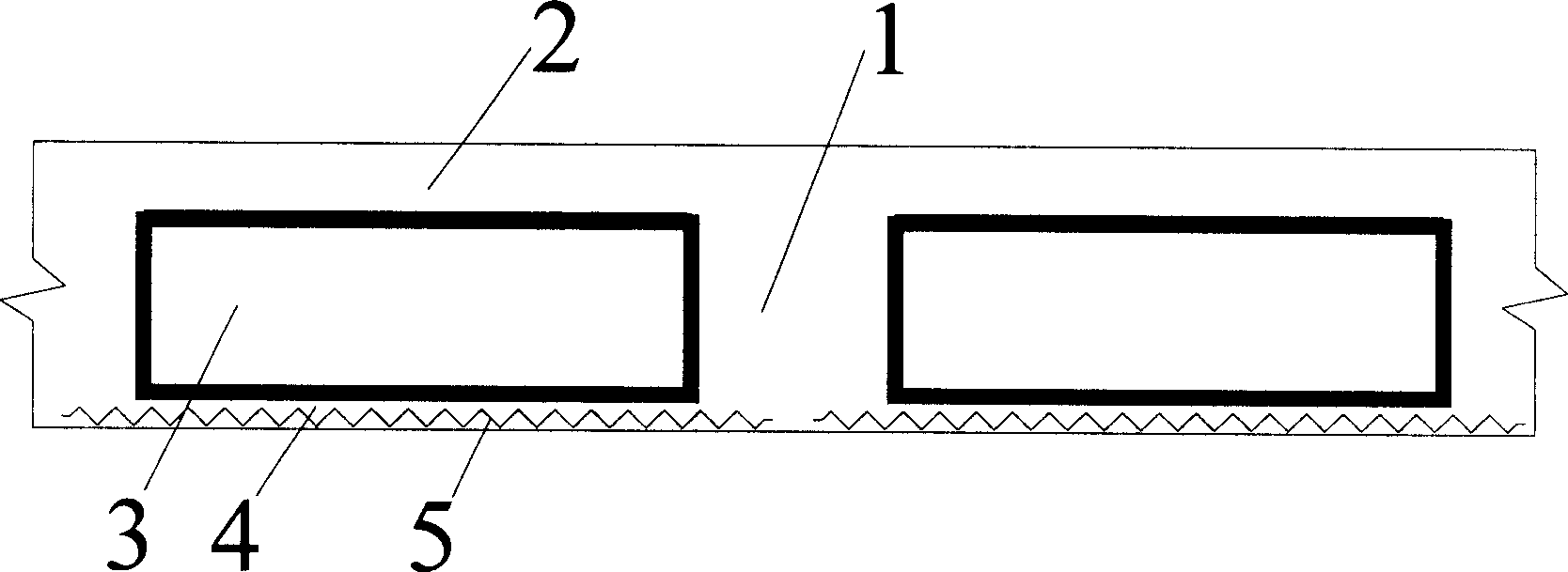
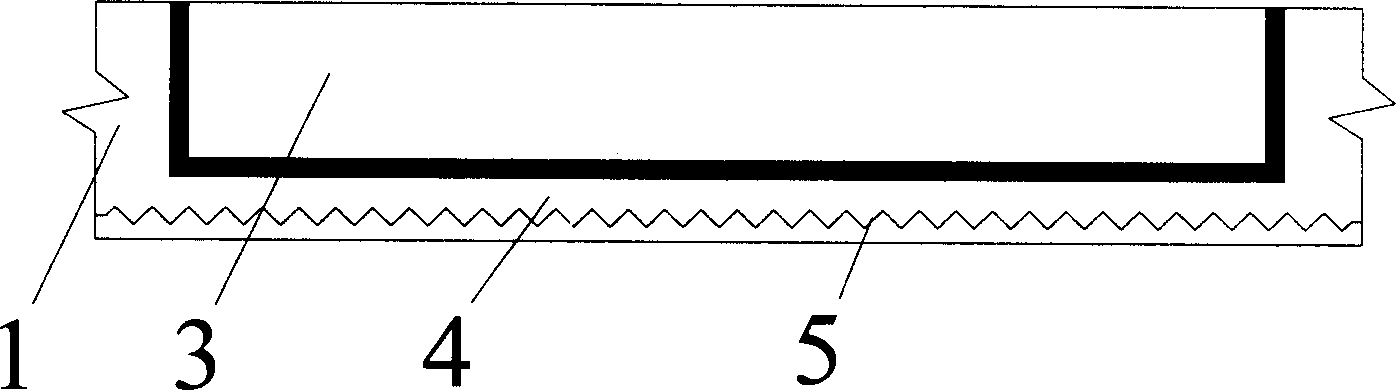
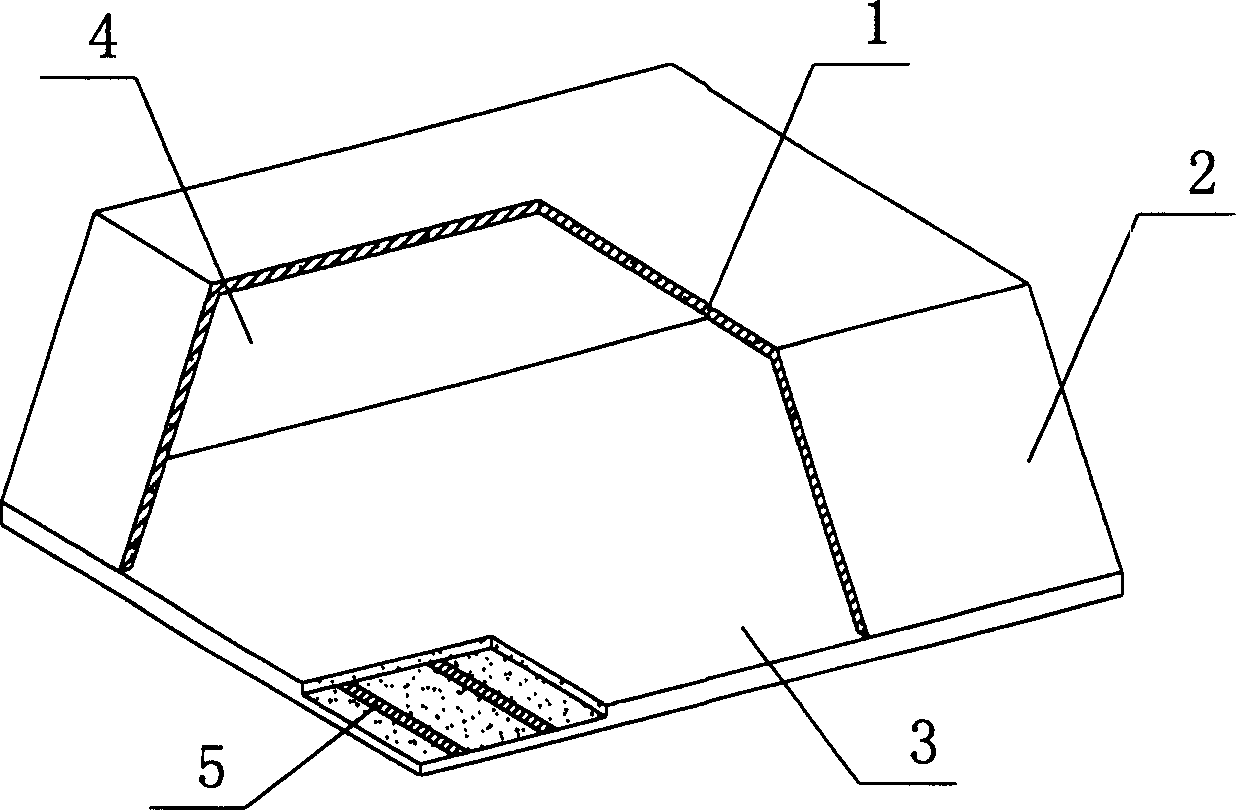
Thin-wall form member for hollowing out concrete
A thin-wall hollow form for concrete to in-situ pour hollow non-beam floor is composed of top plate, peripheral wall and bottom plate, and is characterized by that its bottom plate contains at least one thin strip for improving its resistance to impact and collision. Its advantages are simple structure, high strength, toughness and resistance to deformation and vibration, light weight and low cost.
Owner:湖南邱则有专利战略策划有限公司