Patents
Literature
60results about How to "Situations to avoid safety accidents" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Charging gun with dragging protection function
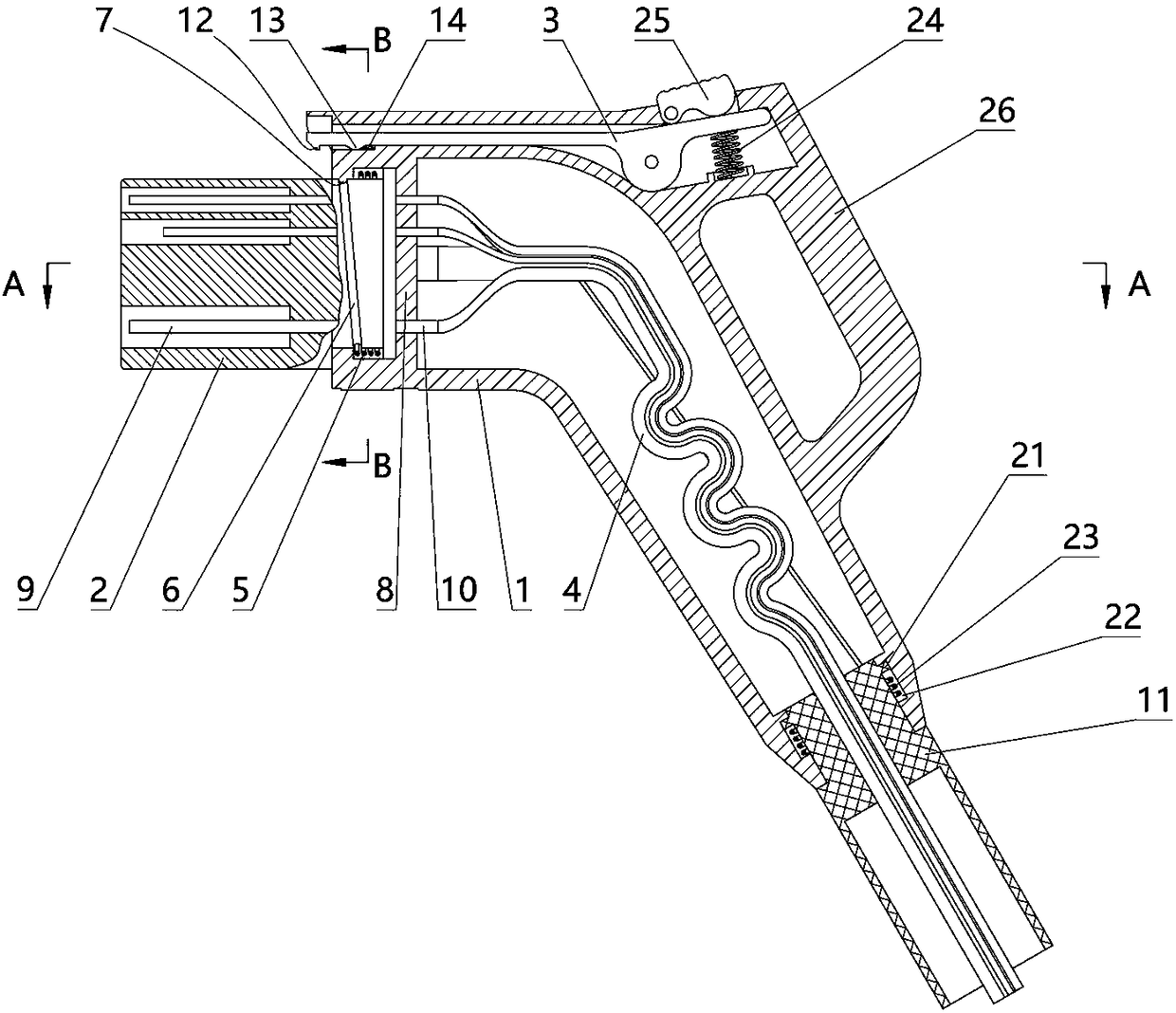
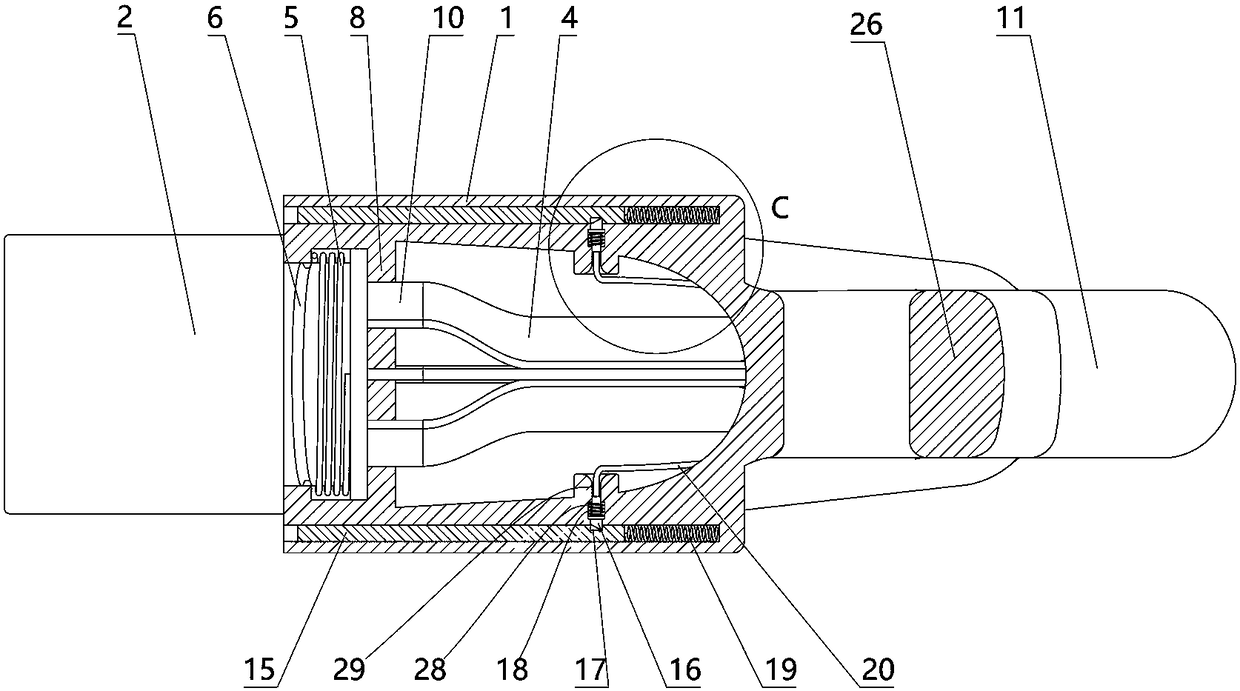
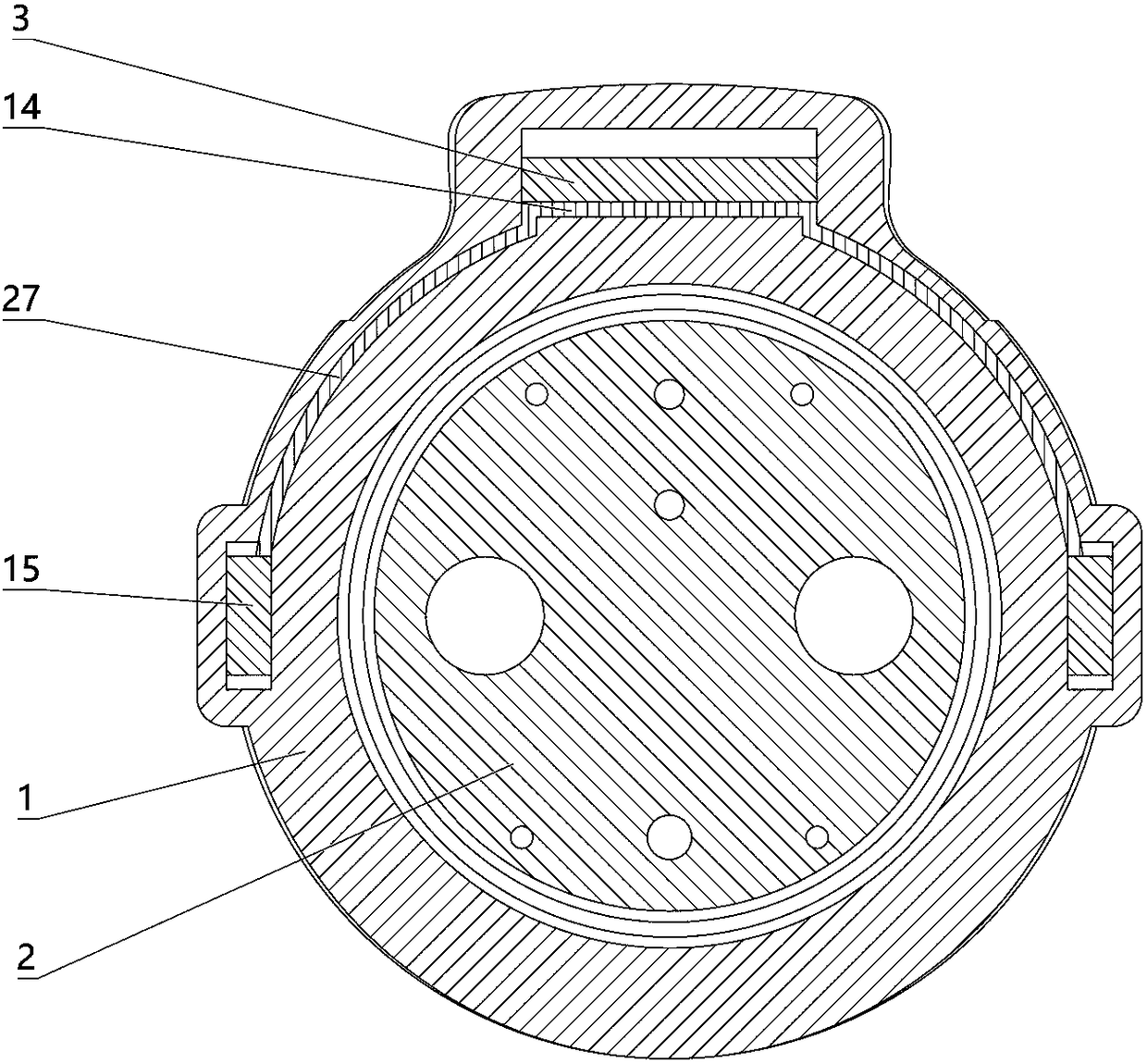
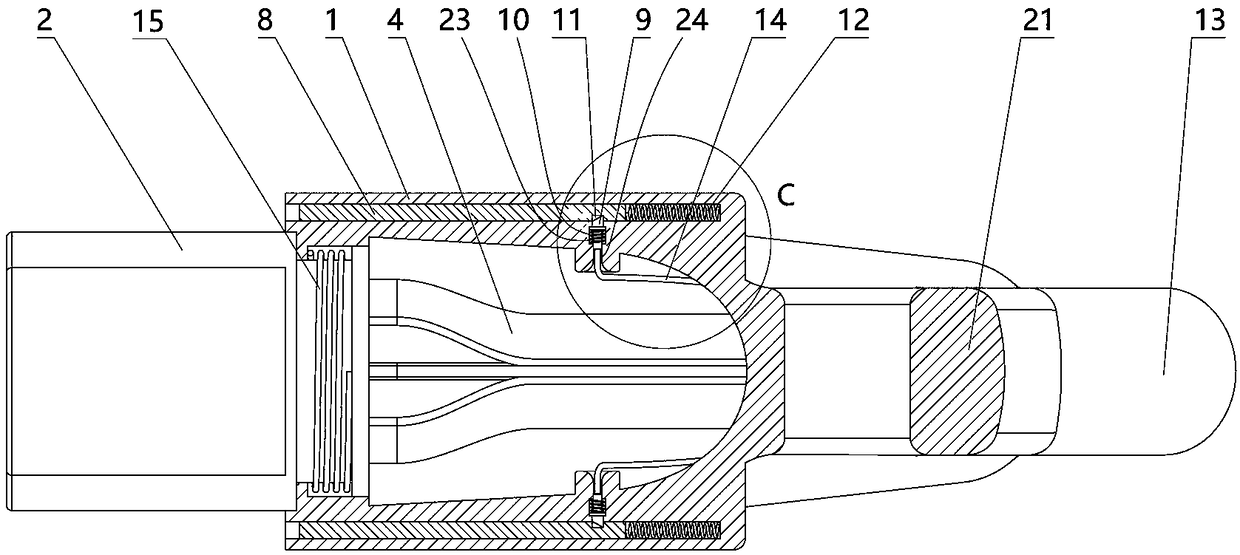
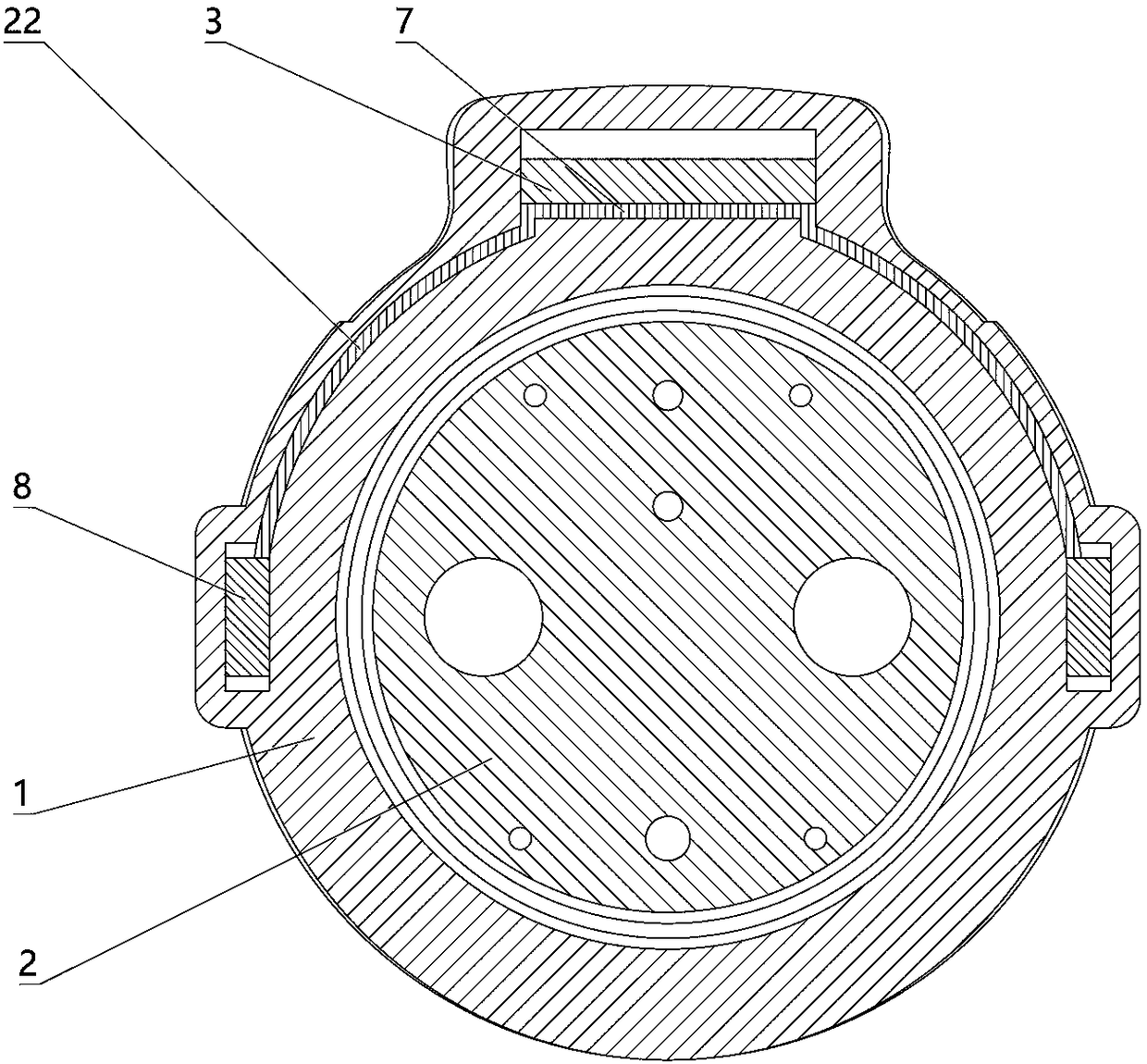
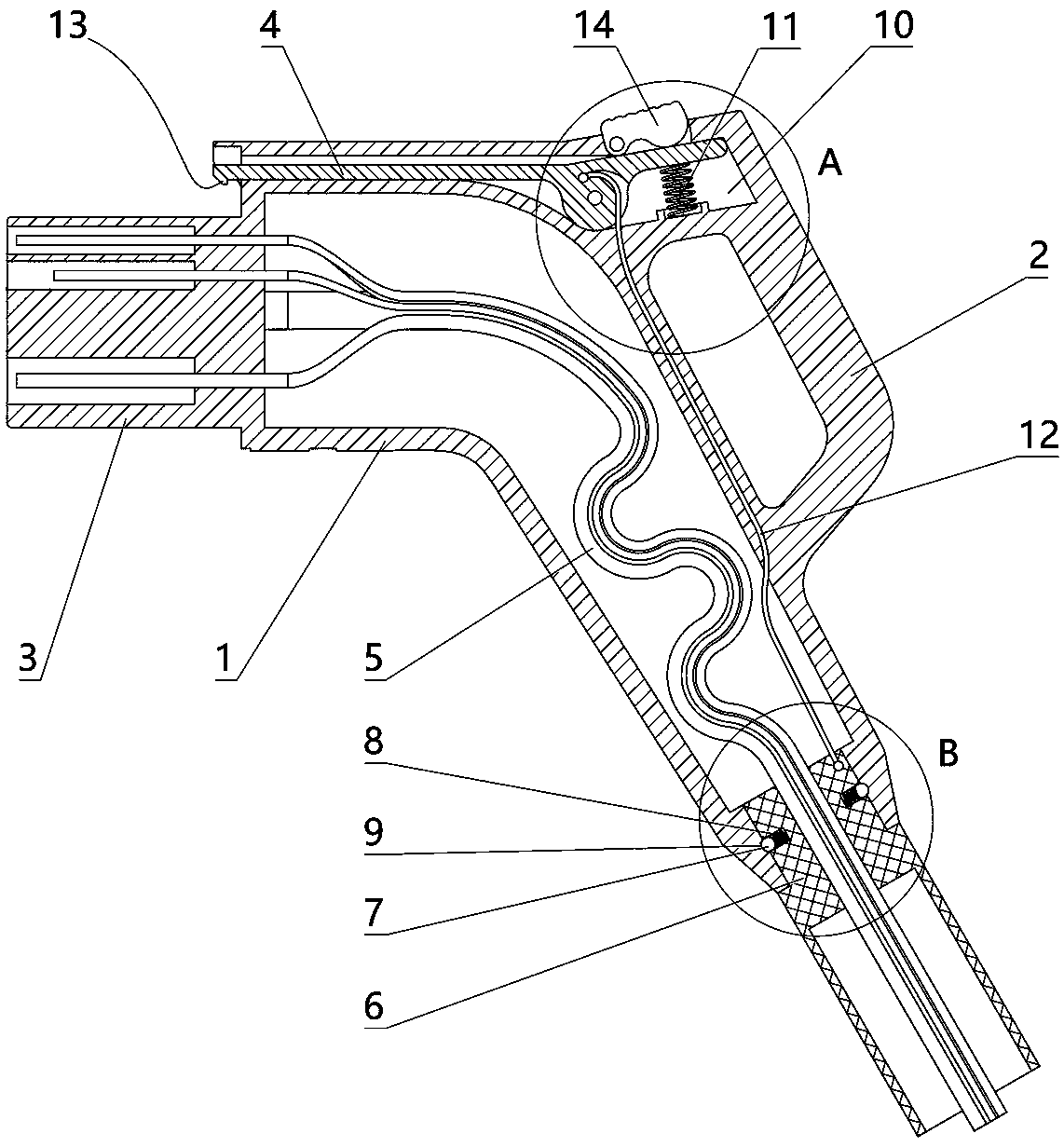
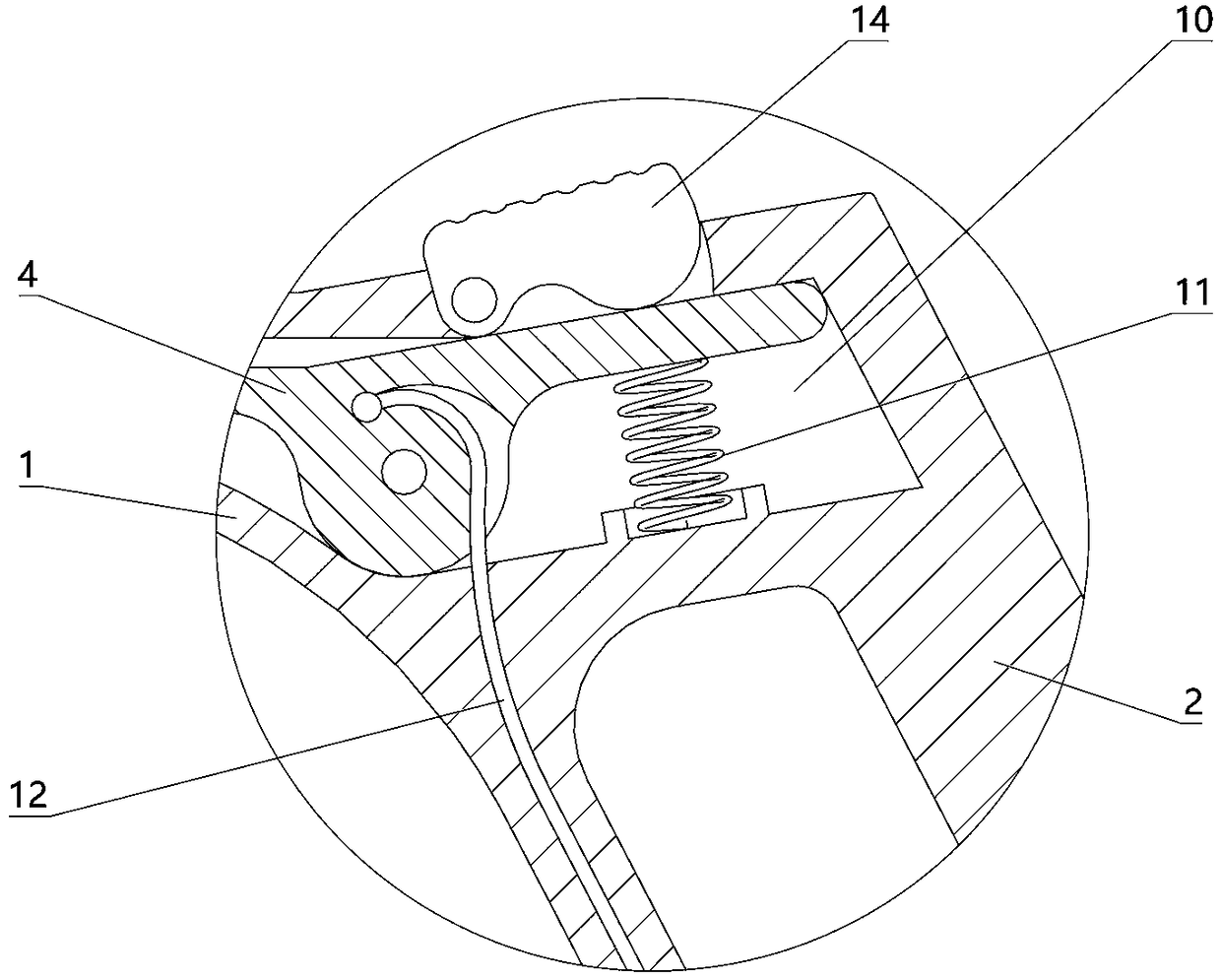
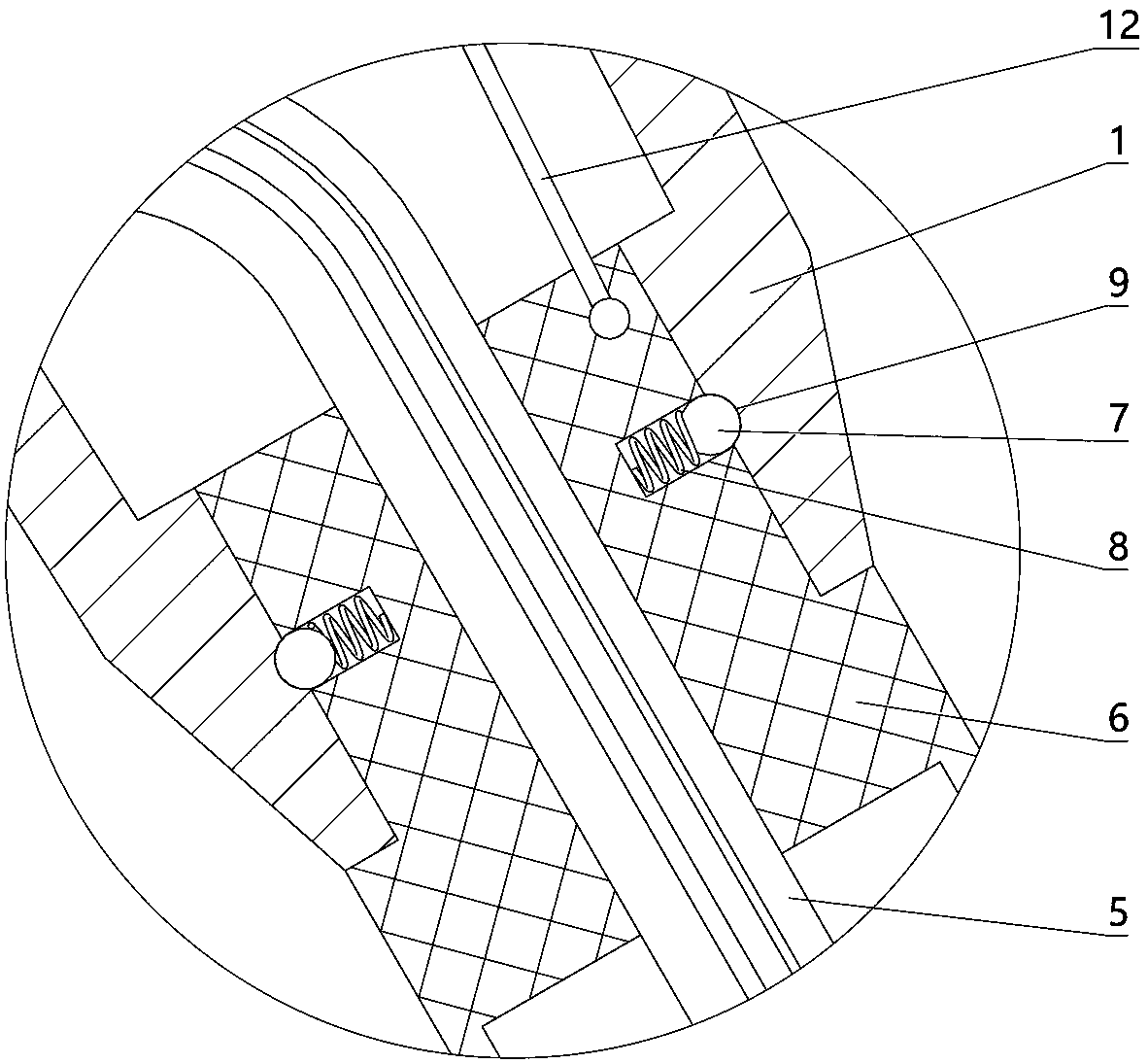
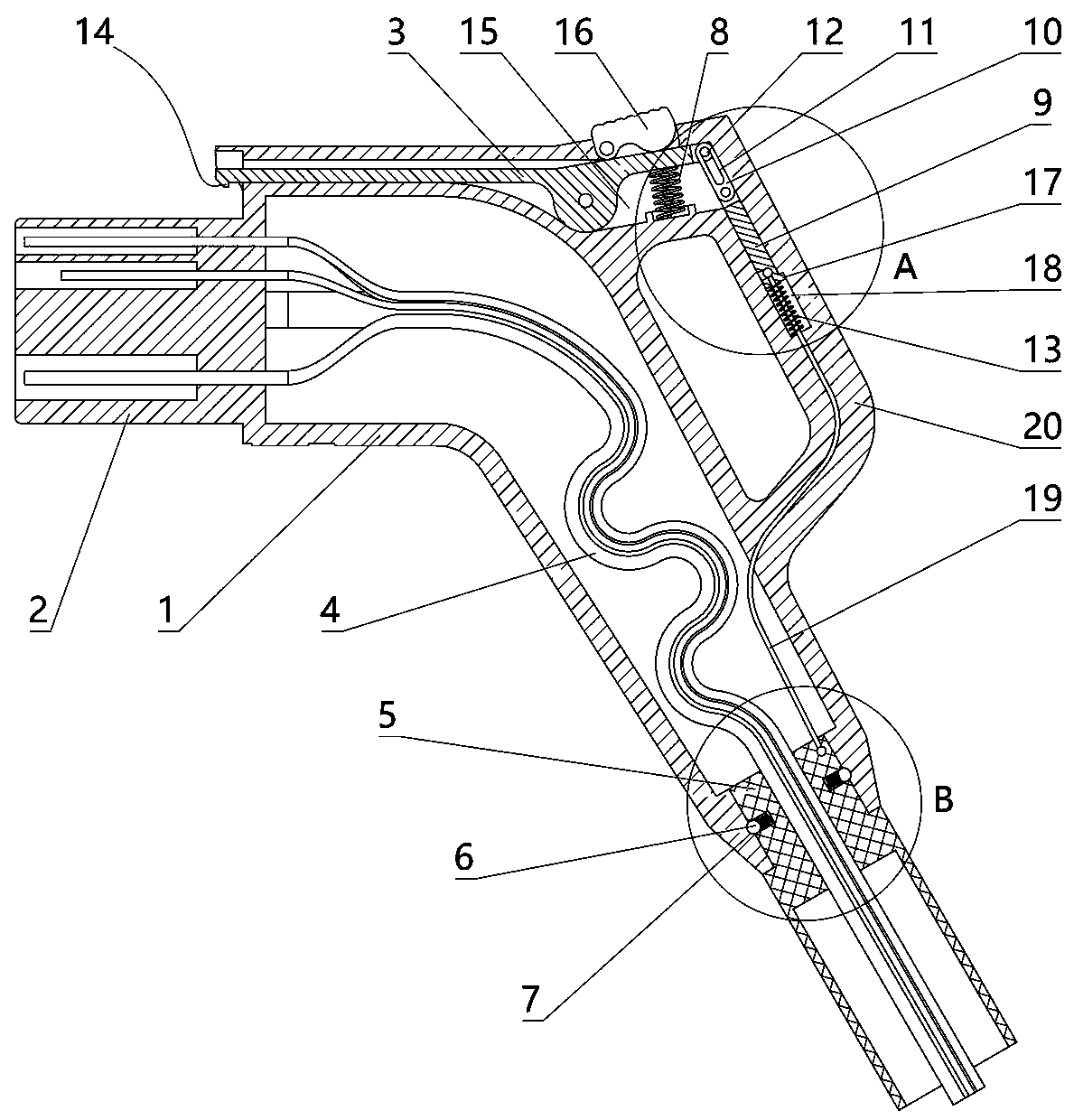
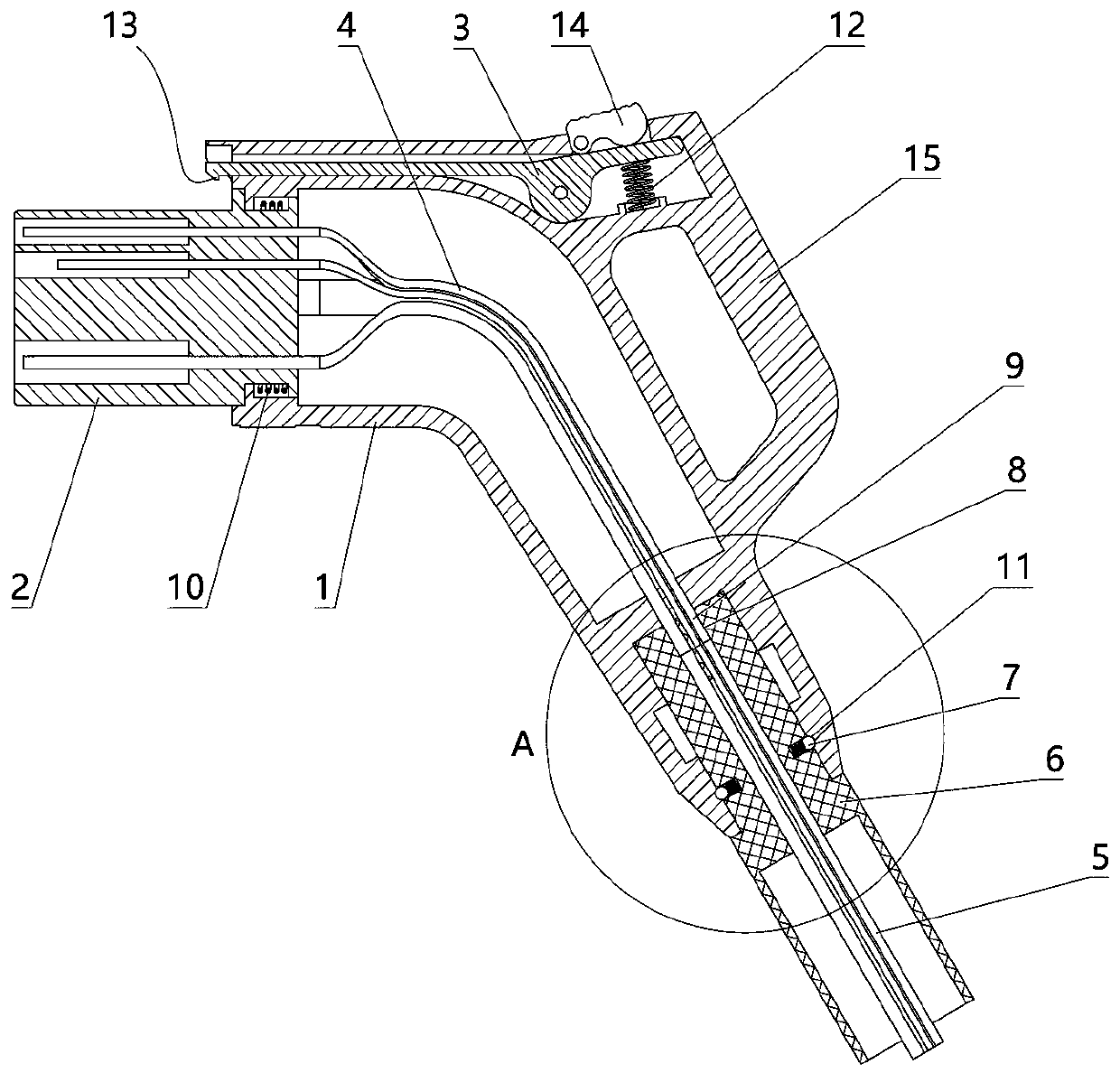
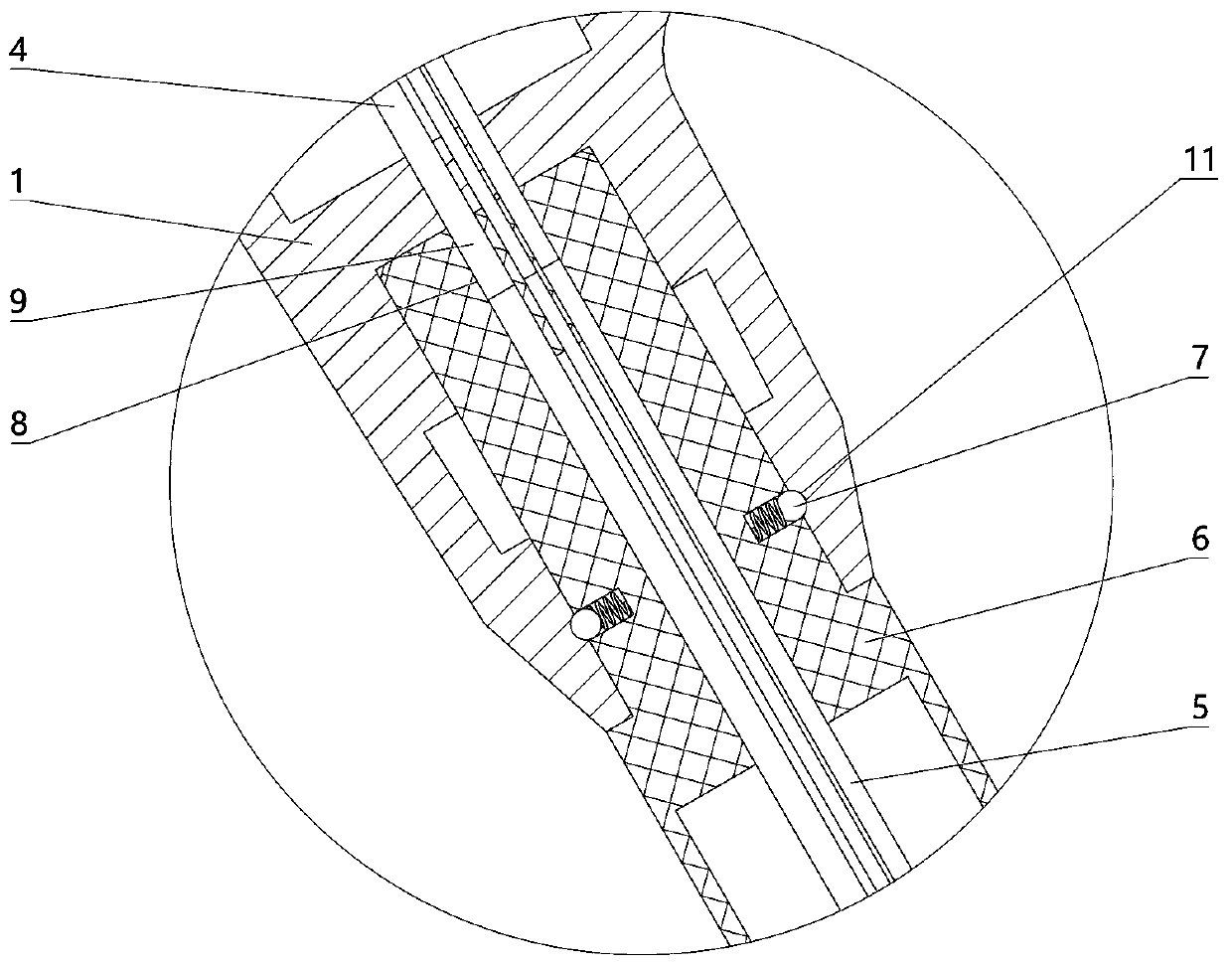
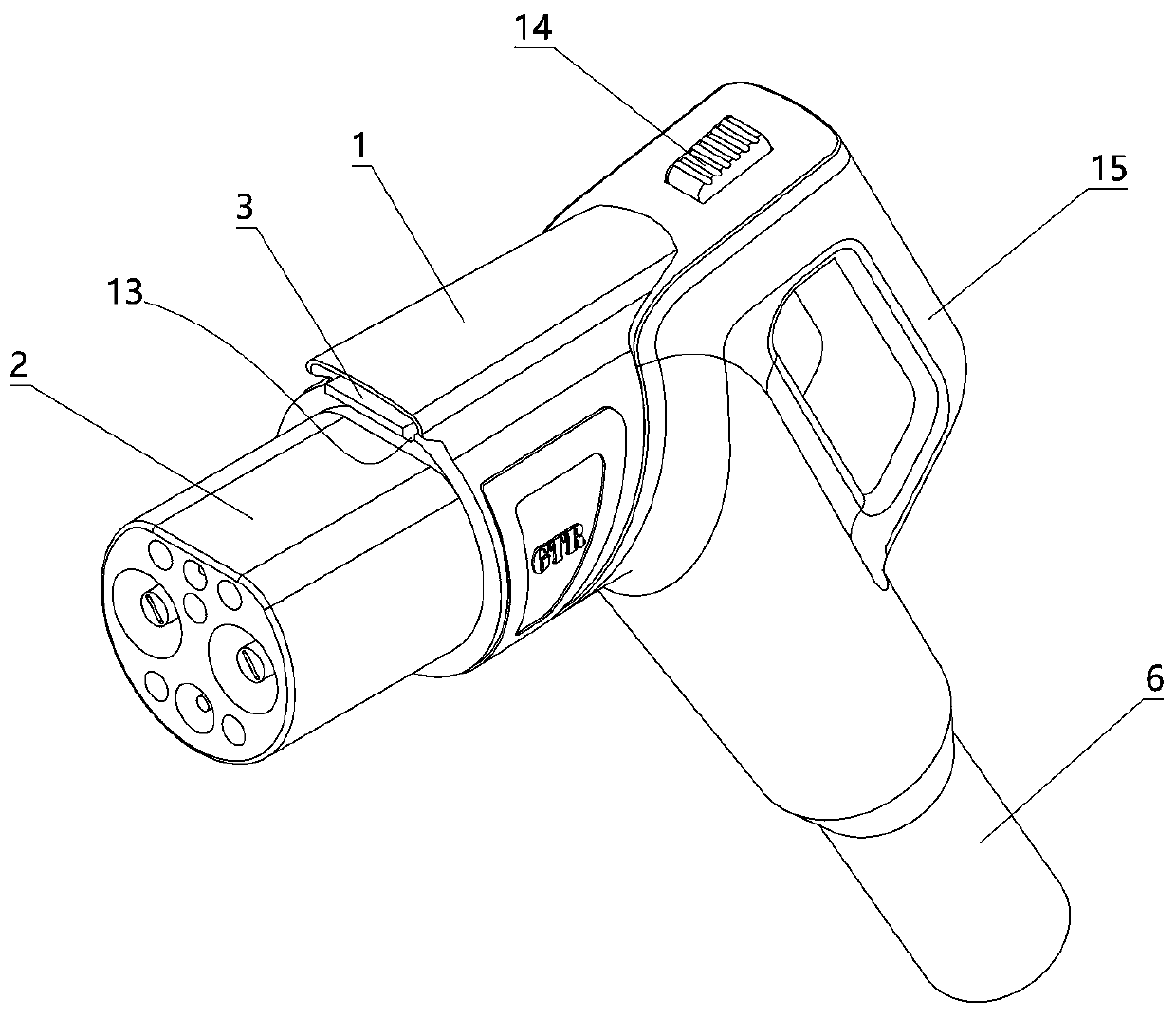
The invention relates to a charging gun with a dragging protection function. The charging gun comprises a gun body with an inner cavity, a plug, a locking part and a wiring harness; the plug is rotationally connected with the gun body; the plug is sleeved with a torsional spring; the two ends of the torsional spring are connected with the gun body and the plug respectively; a circumferential cam groove, which is closed and inclined along the rotary axis of the plug, is formed in the plug; a cam push rod matched with the circumferential cam groove is arranged on the gun body; the cam push rod is arranged in the circumferential cam groove; a terminal placed in the inner cavity is arranged on the gun body; a plurality of horizontally arranged ejector pins are arranged on the plug, multiple contact conductors are mounted on the terminal, and the contact conductors are in contact with the ejector pins in a one-to-one correspondence manner; an inserting-connecting sleeve is glidingly inserted and mounted on the gun body; the wiring harness is fixedly connected with the inserting-connecting sleeve; and one end of the wiring harness is placed in the inner cavity and connected with the terminal. By virtue of the charging gun, the normal charging use of a vehicle can be satisfied while it is ensured that the charging gun can be in a power-down state quickly in suffering from pulling force so as to be separated from the vehicle elastically and safely in an emergency condition.
Owner:乐清市荣盛引进电器有限公司
Gun head bounce-off type charging gun
ActiveCN108718019AExquisite structureEasy to operateVehicle connectorsCharging stationsMechanical engineeringCable harness
The present invention relates to a gun head bounce-off type charging gun which comprises a gun body provided with an inner cavity, a plug, a locking element and a wire harness. The locking element isinstalled on the gun body in a manner of rotating left and right. A left end of the locking element has a lock catch which protrudes downwards and a circular-arc lug boss. The gun body is provided with a sliding block which can slide out of the gun body to the left. The sliding block is placed at the right side of the circular-arc lug boss and comes into contact with the circular-arc lug boss. Thegun body is provided with an ejector rod horizontally disposed and sliding out of the gun body to the left. The sliding block is connected with the ejector rod. The gun body is provided with a wedgeblock which slides perpendicularly to the lengthwise direction of the ejector rod. The ejector rod is provided with a clamping groove. The wedge block is sleeved with a first spring. Under the effectof the elasticity of the first spring, a wedge end of the wedge block slides into the clamping groove. The ejector rod is connected with the gun body through a force accumulation spring. A plug jacketis inserted into the gun body in a sliding mode. A pull rope is disposed in the inner cavity. One end of the pull rope is connected with the wedge block, and the other end of the pull rope is connected with the plug jacket. The wire harness is fixedly connected with the plug jacket. One end of the wire harness is placed in the inner cavity and is connected with the plug.
Owner:山东路盛源新能源科技有限公司
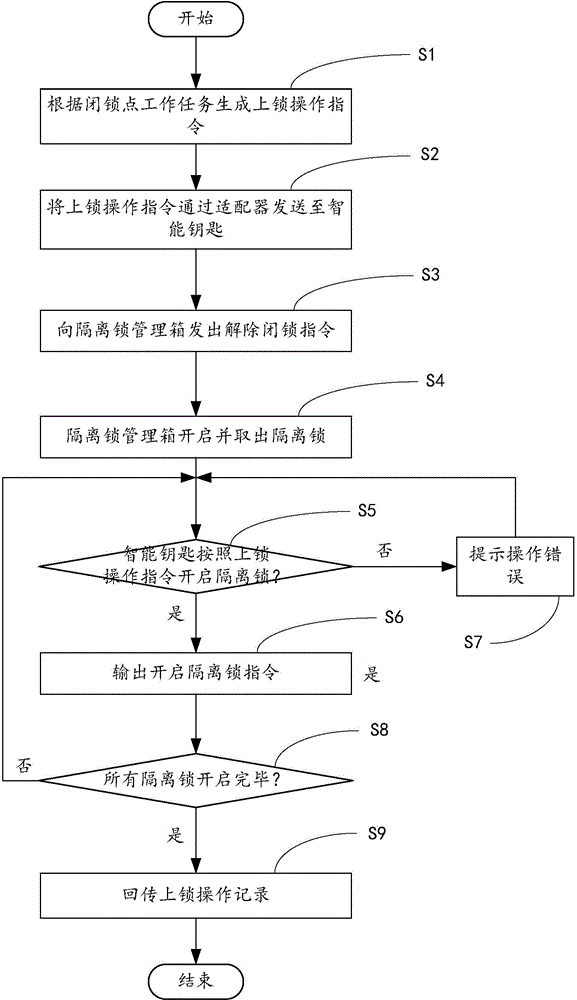
Maintenance isolation locking system and method
InactiveCN104680634AAvoid human managementReduce management costsData processing applicationsIndividual entry/exit registersInformatizationWork task
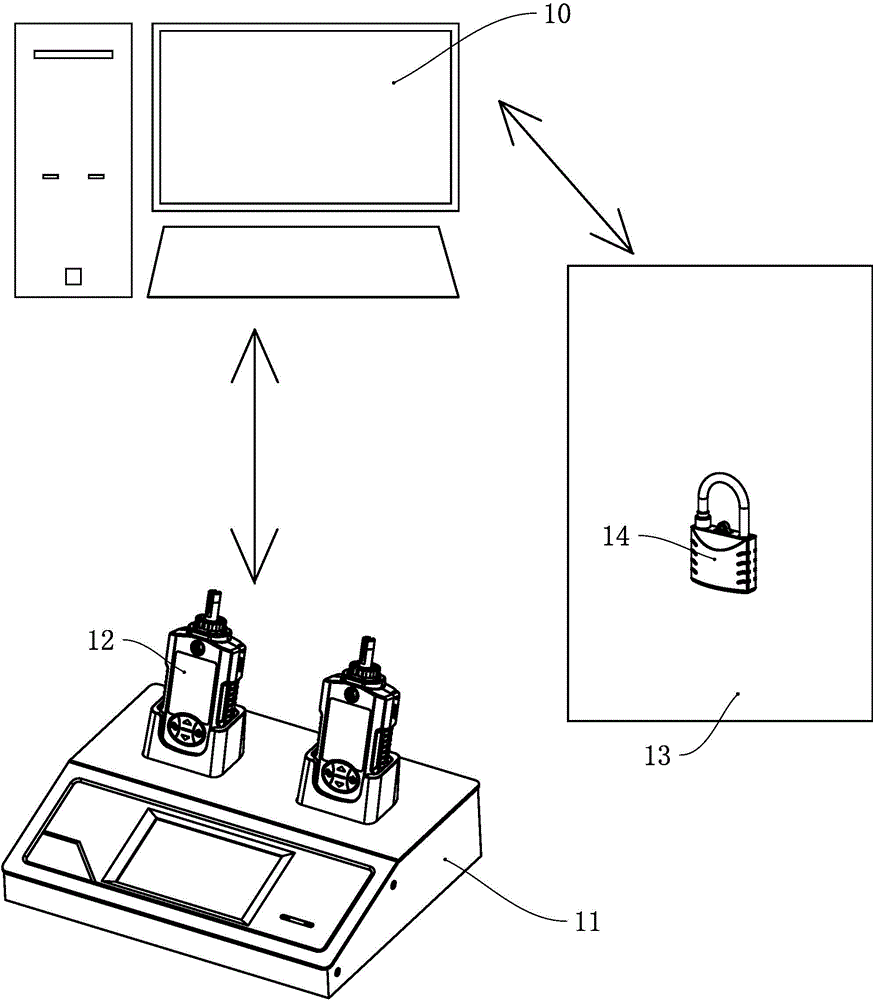
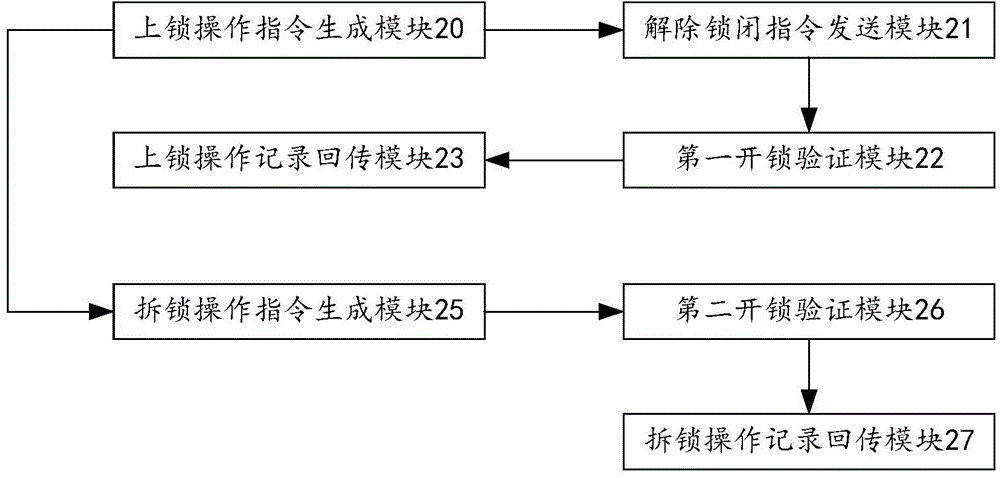
The invention provides a maintenance isolation locking system and method. The system comprises a locking operation instruction generation module, a lock-up release instruction sending module and a first unlocking verification module, wherein the locking operation instruction generation module is used for generating a locking operation instruction according to a work task of a lock-up point requiring locking and sending the locking operation instruction to an intelligent key; the lock-up release instruction sending module is used for sending a lock-up release instruction of an isolation lock required to be unlocked by the intelligent key to an isolation lock management box according to the locking operation instruction; the first unlocking verification module is used for verifying whether the isolation lock to be opened by the intelligent key is correct or not according to the locking operation instruction and outputting an instruction for opening the isolation lock after the condition that the isolation lock to be opened by the intelligent key is correct is determined. The method is an informatization management method for realizing implementation, monitoring and dismantling of a maintenance isolation measure through application of the maintenance isolation locking system. Therefore, the management efficiency of the information process of the safe isolation measure as well as the lock in electric power system maintenance can be improved, the isolation lock is effectively prevented from being lost, and safety accidents caused by faulty operation of the isolation lock are prevented.
Owner:ZHUHAI VALWELL ELECTRIC TECH
New energy vehicle charging gun
ActiveCN108749610AExquisite structureEasy to operateCoupling device detailsElectric vehicle charging technologyEngineeringElectrical and Electronics engineering
The invention relates to a new energy vehicle charging gun. The new energy vehicle charging gun comprises a gun body in which a cavity is formed, a handle, a plug, a locking piece and a wiring harness; a clamping sleeve capable of sliding is inserted in the gun body; multiple clamping beads distributed on a sliding contact part of the clamping sleeve and the gun body are arranged on the clamping sleeve; first springs corresponding to the clamping beads one to one are arranged on the clamping sleeve; clamping grooves matched with the clamping beads are formed in an inserting pars, correspondingto the clamping sleeve, of the gun body; the clamping beads slide to the external side direction of the clamping sleeve under elastic effects of the first springs so that one parts of the clamping beads are placed in the clamping grooves; the wiring harness is fixedly connected with the clamping sleeve; one end of the wiring harness is connected with the plug; the locking piece is mounted on thegun body in a left-to-right rotating manner; a second spring is arranged on the gun body; the locking piece rotate anticlockwise under an elastic effect of the second spring; the new energy vehicle charging gun further comprises a pull rope; one end of the pull rope is connected with the locking piece; and the other end of the pull rope is connected with the clamping sleeve.
Owner:宁波里尔汽车技术有限公司
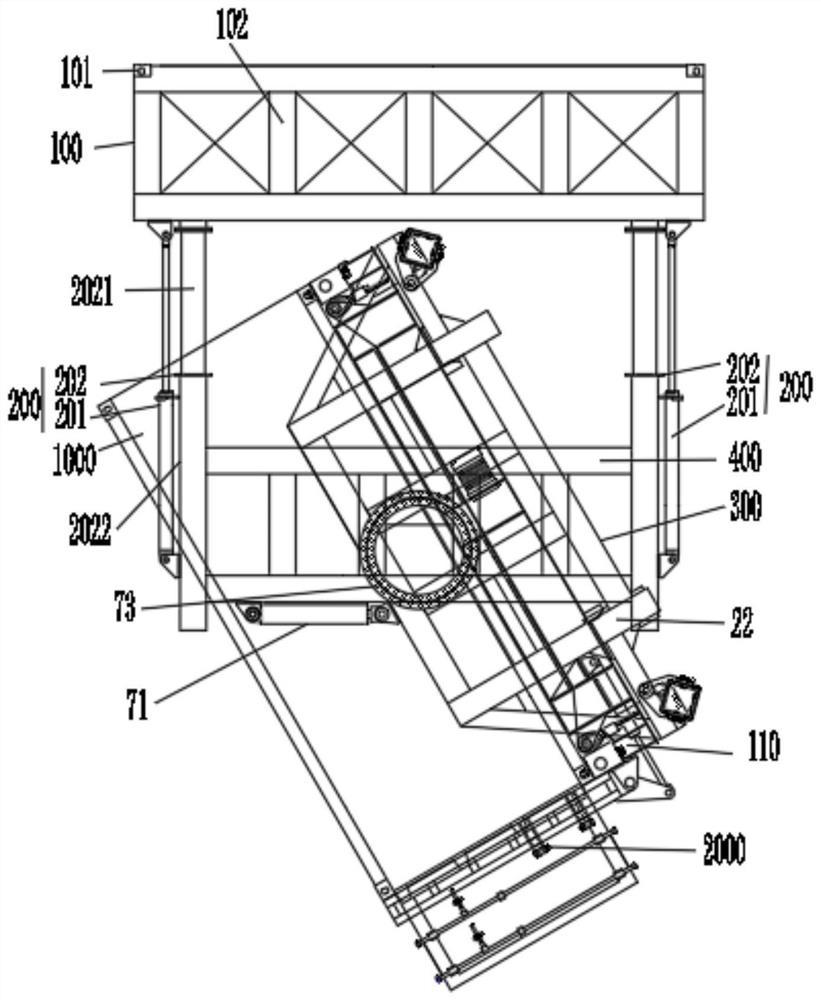
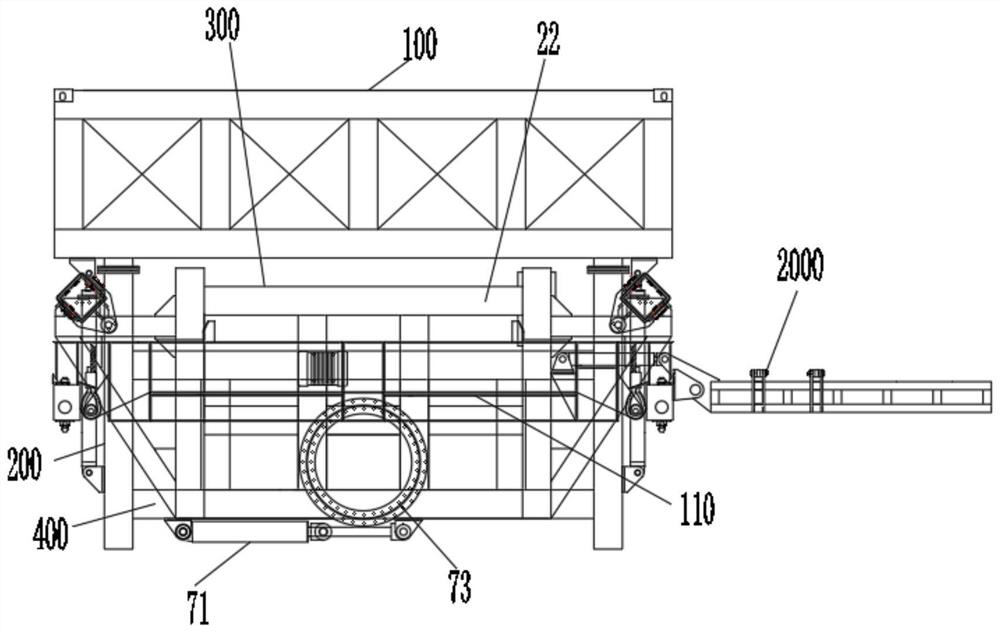
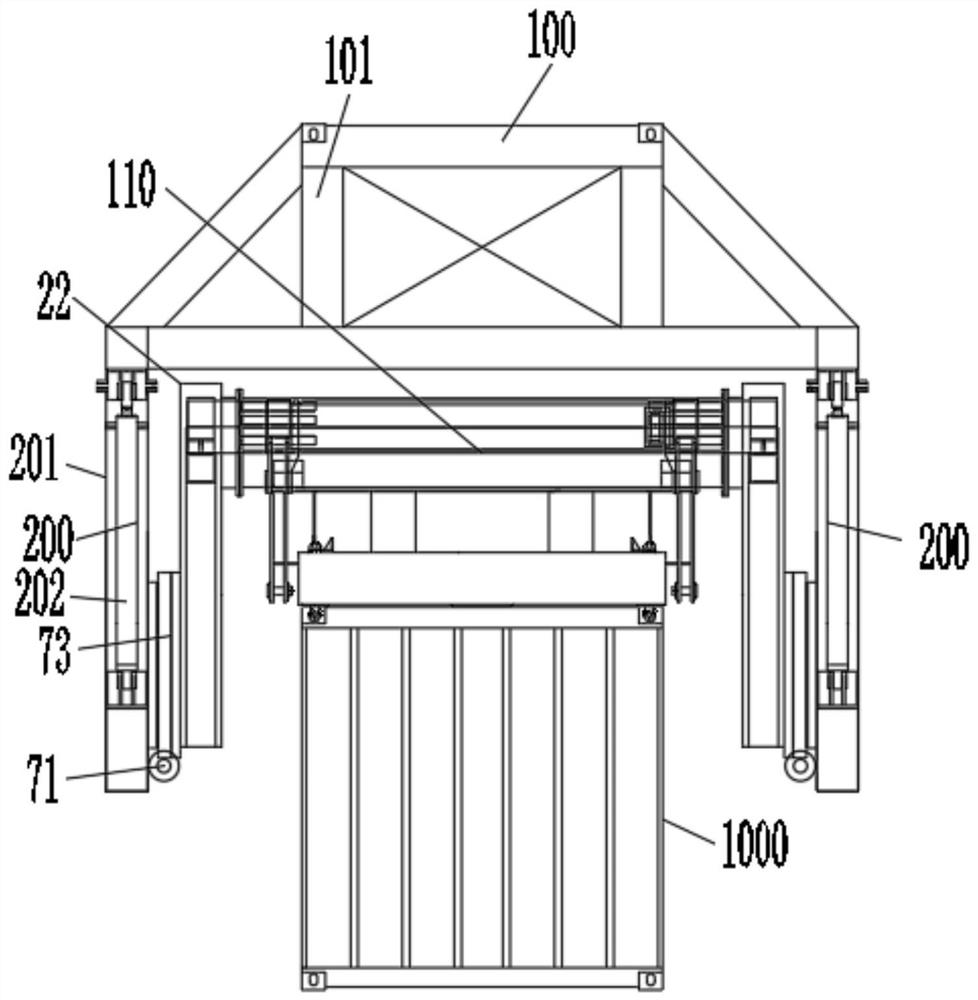
Lifting appliance special for overhead turnover unloading of container
PendingCN111824947AImprove loading efficiencyReduce the intensity of manual operationLoading/unloadingLoad-engaging elementsStructural engineeringMechanical engineering
The invention discloses a lifting appliance special for overhead turnover unloading of a container. The lifting appliance is characterized by comprising a lifting appliance connecting piece, a liftingbody and a stretchable device. The two ends of the stretchable device are connected with the lifting connecting piece and the lifting body respectively. The stretchable device drives the distance between the lifting connecting piece and the lifting body to become larger or smaller. A turnover mechanism for lifting the container is arranged on the lifting body. The turnover mechanism is used for turning over the lifted container to be horizontal or perpendicular or inclined, and a lifting connecting part is arranged on the lifting connecting piece. The lifting appliance has the technical effects that the unloading efficiency is high, the manual operation intensity is lowered, the cost is low, the environmental-friendly performance is good, the damage of cargo is little, unloading is clean,and safe operability is good.
Owner:苗林展
Container door opening or closing device of container
PendingCN111204594ARealize non-artificialAchieve securityVehicle locksLarge containersStructural engineeringMechanical engineering
The invention discloses a container door opening or closing device of a container. The container door opening or closing device comprises a hanger used for hanging the container and is characterized in that an automatic turnover mechanism is installed at one end of the hanger, a frame is fixed to the automatic turnover mechanism, a container door opening or closing mechanism is installed on the frame, and a container door lock rod locking mechanism is installed on the container door opening or closing mechanism; and after the automatic turnover mechanism turns over the frame to fit one port face of the container, the container door lock rod locking mechanism detachably locks container door lock rods of the container, and then the container door opening or closing mechanism moves to open orclose container doors. The container door opening or closing device structurally achieves the technical advantages of non-labor, high efficiency, safety, controllable operation and the like.
Owner:苗林展
Novel container hoisting equipment
PendingCN111362113AImprove work efficiency and safety of operationImprove efficiencyNon-mechanical controlsPower-operated mechanismStructural engineeringManual handling
The invention discloses novel container hoisting equipment. The novel container hoisting equipment comprises a hoisting tool, a container door opening and closing device is installed at one end of thehoisting tool, and a container body protection mechanism for pressing and protecting the outer side wall of a container is further installed on the hoisting tool. When the structure is applied to container overturning operation, the functions of automatically opening and closing a container door and preventing a container body from deforming are achieved, furthermore, opening and closing of the container door, a container body protective plate and a pressing plate are operated in a non-manual manner, and therefore the hoisting equipment has the advantages of being high in efficiency, safe, controllable in operation, good in protection performance and the like.
Owner:苗林展
Novel material unloading equipment and novel material unloading device
PendingCN111994651ALow costPromote environmental protectionConveyorsBase supporting structuresButt jointIndustrial engineering
The invention discloses novel material unloading equipment and a novel material unloading device. The novel material unloading equipment comprises an unloading frame, an unloading conveying mechanism,a lifting driving mechanism and an unloading funnel, wherein the unloading funnel is arranged at the bottom of an unloading channel of the unloading frame; the bottom end of the unloading conveying mechanism is rotationally connected with the unloading frame; the lifting driving mechanism is fixed on the unloading frame, and a lifting driving part thereof is connected with the unloading conveyingmechanism; the bottom end of the unloading conveying mechanism is in butt joint with a discharge port of the unloading funnel; and an overturning mechanism is arranged on the unloading frame. The novel material unloading equipment has such technical effects as high efficiency, low cost, excellent environmental protection, low goods loss, thorough unloading, high stacking height, safety and operability.
Owner:苗林展
Mold limiting device and sing method thereof
InactiveCN109454168ASituations to avoid safety accidentsAchieve regulationShaping toolsEngineeringMagnet
The invention discloses a mold limiting device. The mold limiting device comprises a limiting block and a supporting column; the supporting column is arranged on a lower mold; one end of the limitingblock is matched with the supporting column, and the other end of the limiting block makes contact with an upper mold; and the limiting block and the supporting column are made of magnets which are the same in magnetism. Through the principle that the like poles of the magnets repel, when a mold is opened, the limiting block automatically bounces away from the supporting column so that the purposethat the limiting device is automatically taken down can be achieved, and the situation that in the prior art, safety accidents occur due to the fact that an operator forgets to take down the limiting device is avoided. The invention further discloses a using method of the mold limiting device.
Owner:WUHU FEICHI AUTOMOTIVE COMPONENTS TECH
Non-avoidance stereoscopic parking device
The invention relates to a non-avoidance stereoscopic parking device and belongs to the technical field of stereoscopic parking devices. The non-avoidance stereoscopic parking device comprises bases,large arms, lifting hydraulic cylinders, connecting pieces, platform arms and a parking platform, wherein the large arms, the lifting hydraulic cylinders and the connecting pieces are all arranged onthe bases; the connecting pieces are arranged on the upper sides of the large arms; the lifting hydraulic cylinders are arranged on the lower sides of the large arms; one ends of the large arms are hinged to the bases and the other ends of the large arms are hinged to the lower ends of the platform arms; one ends of the connecting pieces are hinged to one ends of the bases, and the other ends of the connecting pieces are hinged to the upper ends of the platform arms; the large arms, the bases, the connecting pieces and the platform arms form parallelogram mechanisms by being arranged around; one ends of the lifting hydraulic cylinders are hinged to the bases, and the other ends of the lifting hydraulic cylinders are hinged to the large arms; the lifting hydraulic cylinders are equipped with lifting self-locking mechanisms; and the parking platform is fixedly connected with the platform arms. The stereoscopic parking device of the invention has the advantages of simple structure, smallsize, low cost and wide application range.
Owner:GUANGXI UNIVERSITY OF TECHNOLOGY
Floating tip cone device for vertical spool take-up machine
The invention discloses a floating tip cone device for a vertical spool take-up machine, and belongs to the technical field of steel wire drawing production equipment. The floating tip cone device comprises a support, a sleeve seat frame and a rotating shaft assembly, wherein the sleeve seat frame is fixedly mounted at the tail end of the support, the rotating shaft assembly is rotationally mounted in a sleeve seat, and the rotating shaft assembly comprises a rotating shaft. The support is fixedly mounted on the take-up machine, so that the support is perpendicular to the central axis of a centering mandrel, the tail end of the rotating shaft is in contact fit with a through hole in the centering mandrel, so that the central axis of the rotating shaft and the central axis of the centeringmandrel are collinear; and after a rotating belt wheel rotates to drive a power rotating disc, the centering mandrel is pressed by the rotating shaft, the spool take-up speed can be kept to be largerthan 8m / s, the problem that when the take-up speed is larger than 8m / s, the spool and the centering mandrel start to generate relative motion, and consequently machine body vibration is increased is solved, and the problems that equipment components fall off and fly out, and safety accidents are caused to operators due to the fact that the components fly out are avoided.
Owner:GUIZHOU AEROSPACE NANHAI SCI & TECH
Novel single-hand pressing pluggable socket
InactiveCN111834816AEasy to operateAchieve installationCoupling device detailsEngineeringStructural engineering
Owner:宁波变雪电子科技有限公司
Automatic welding manipulator with high efficiency and safe operation
InactiveCN113134698AOperational securityImprove securityWelding/cutting auxillary devicesAuxillary welding devicesGratingRobot hand
The invention relates to the technical field of automation equipment, and discloses an automatic welding manipulator with high efficiency and safe operation. The automatic welding manipulator comprises a protective cover, wherein a workbench is arranged in the protective cover, and a stepping motor is arranged in the workbench; a rotary table in transmission connection with the stepping motor is arranged at the top of the workbench, and a positioning assembly is arranged at the top of the rotary table; and a safety optical grating is arranged on the outer wall of the protective cover. According to the automatic welding manipulator with high efficiency and safe operation, welding equipment is surrounded by the safety optical grating, so that the situation that the equipment collides with the outside in the running process is avoided; the safety optical grating is used for detecting the body of a worker entering and exiting the protective cover, so that safety accidents caused by the fact that the worker does not completely exit from the protective cover and the equipment runs are avoided, and the simple and convenient multi-stage protection effect is achieved; and therefore, the safety performance of the automatic welding manipulator is improved.
Owner:晋江荣达机械有限公司
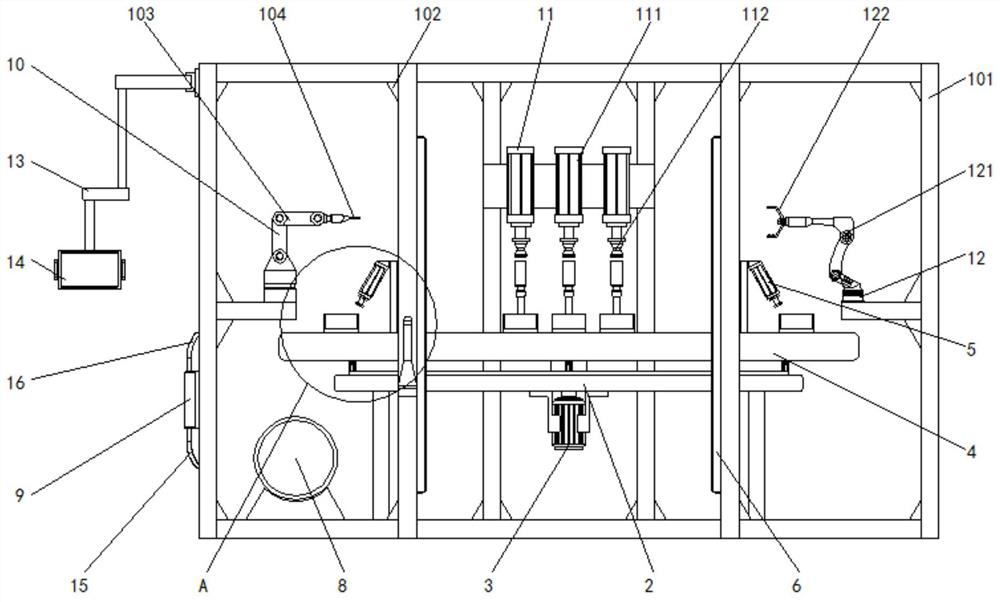
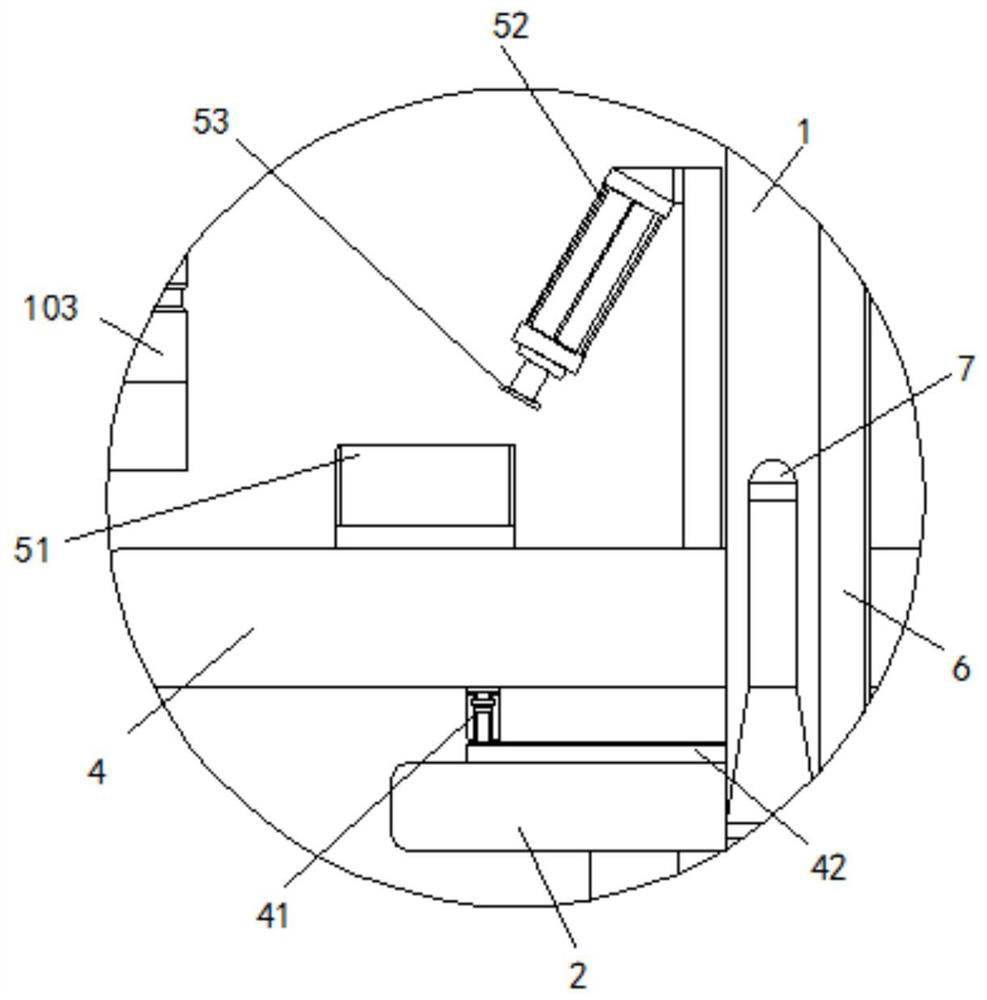
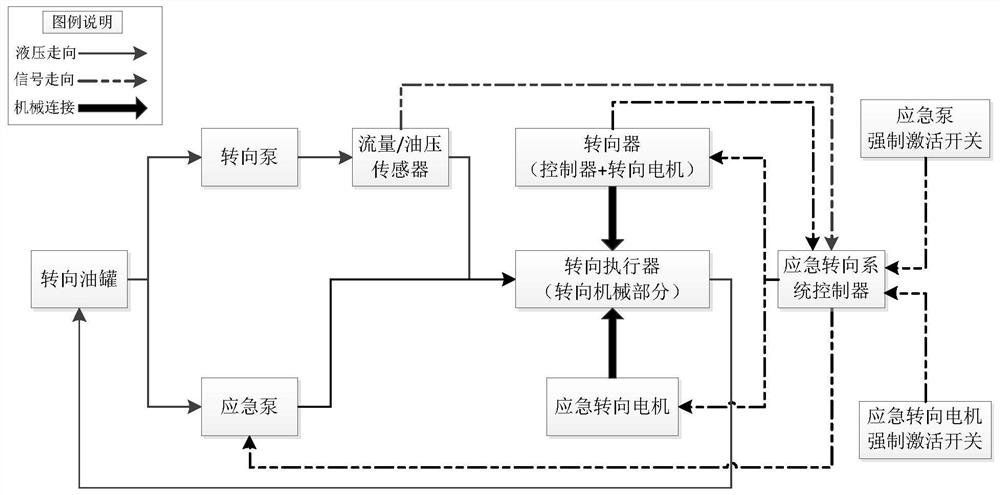
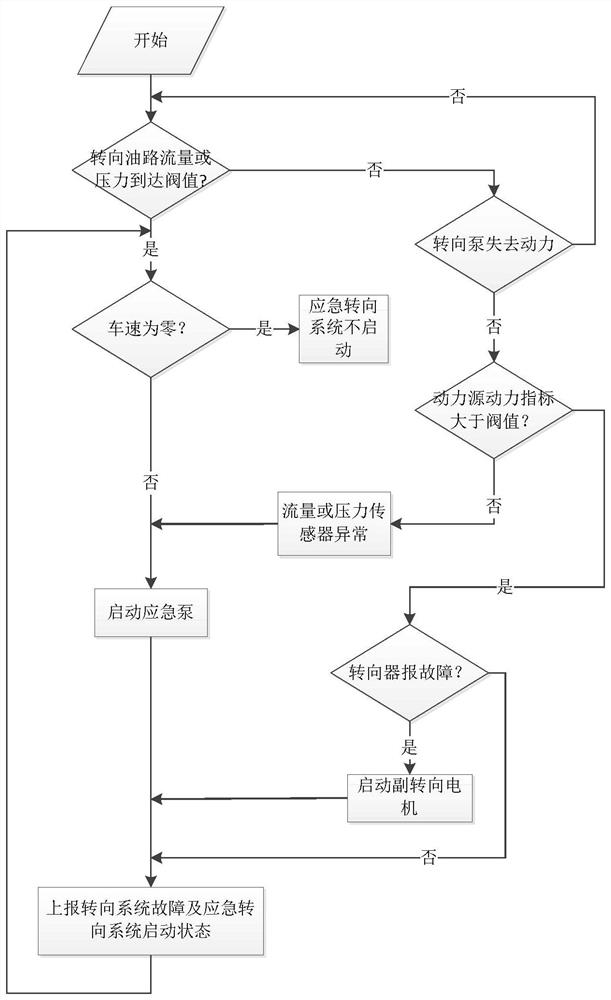
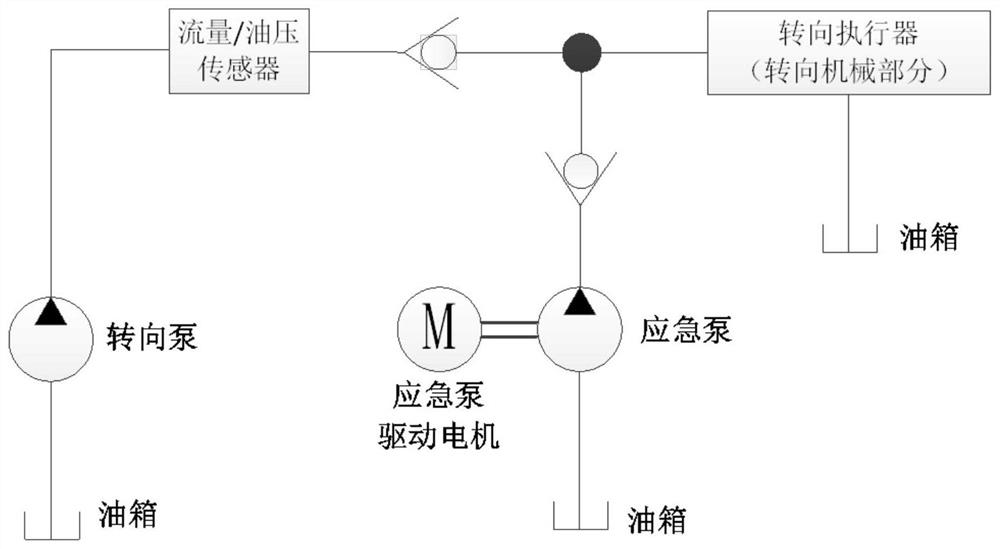
Emergency steering system and method for automatic drive-by-wire vehicle
PendingCN112960034ASteering function is normalAvoid safety accidentsFluid steeringElectrical steeringOil pressureSteering system
The invention discloses an emergency steering system for an automatic drive-by-wire vehicle. The system comprises an oil pressure or flow detection sensor used for detecting the oil pressure or flow of hydraulic oil between oil paths connected with a steering pump and a steering actuator, an oil path switch arranged between the oil paths, an emergency steering system controller outputting a signal I when the oil pressure value or the flow value is lower than a preset corresponding threshold value, and an emergency pump, wherein after the emergency pump receives the signal I, the emergency pump is started to input hydraulic oil into the steering actuator. The system has the advantages of automatically starting the emergency pump and guaranteeing the normal steering function of the vehicle. The invention further discloses an emergency steering method for the automatic drive-by-wire vehicle. The emergency steering method comprises the steps of detecting the oil pressure or flow of hydraulic oil between the oil paths connected with the steering pump and the steering actuator; when the oil pressure or flow is lower than the preset threshold value, closing the oil paths, and outputting the signal I; and after the emergency pump receives the signal I, starting the emergency pump to input hydraulic oil into the steering actuator. The method has the advantages of automatically starting the emergency pump and guaranteeing the normal steering function of the vehicle.
Owner:西安主函数智能科技有限公司
Rosuvastatin calcium tablet and preparation process thereof
PendingCN111548312AImprove productivityReduce manufacturing costOrganic active ingredientsOrganic chemistryNaCl - Sodium chloridePyrimidine
The invention relates to the technical field of pharmaceutical preparations and discloses a rosuvastatin calcium tablet. [4-[4-fluorophenyl-6-(1-methylethyl)-2-[N_ methyl-(N-methylsulfonyl) amino]-5-pyrimidyl] methyltriphenylphosphonium bromide, (4R-cis)-6-formyl-2, 2-dimethyl-1, 3-dioxane-4-cyclopentyl acetate, potassium carbonate, dimethyl sulfoxide, methylbenzene, acetonitrile, ethyl acetate, methanol, isopropanol, sodium hydroxide, sodium chloride and calcium chloride. According to the rosuvastatin calcium tablet and the preparation process thereof, a plurality of experiments prove that: when reaction temperature is about 75 DEG C in intermediate synthesis, alkali is the optimal experimental condition of the potassium carbonate, and reaction raw materials can react completely, the generation of multiple impurities is prevented, productivity can be improved, and production cost is reduced; a hydrochloric acid solution is added in a rosuvastatin cyclopentyl ester synthesis process toneutralize redundant acid and alkali, so that the generation of the impurities is further reduced; and therefore, high-purity production is achieved.
Owner:ABA CHEM CORP
Chain guiding-type freight elevator
The invention discloses a chain-guided freight elevator. A chain-guided freight elevator comprises a bearing platform for carrying goods, a square bracket for installing the bearing platform, and two brackets for lifting and lowering the bearing platform. Motion-guided guide chains, four force-bearing seats for supporting the weight of the entire elevator, a driving device for driving the carrying platform to move up and down, and a control device for controlling the operation of the entire elevator; the carrying platform can move along the The guide chain moves up and down; the two guide chains are parallel and symmetrically arranged on the opposite sides of the square bracket, and the bearing platform is arranged in the square bracket; the four water chestnuts on the top of the square bracket are respectively connected with the four The force-bearing bases are connected; the four force-bearing bases are installed on the flat ground of the highest floor of the multi-storey building; the guide chain and the control device are electrically connected with the driving device. The invention has the characteristics of stable operation, effective prevention of misoperation and high safety performance.
Owner:重庆旭彩装订有限公司
Driving shaft, rotor and centrifugal machine thereof
PendingCN114405693AReduce operational burdenEasy to operateCentrifugesDrive shaftClassical mechanics
Owner:RWD LIFE SCI
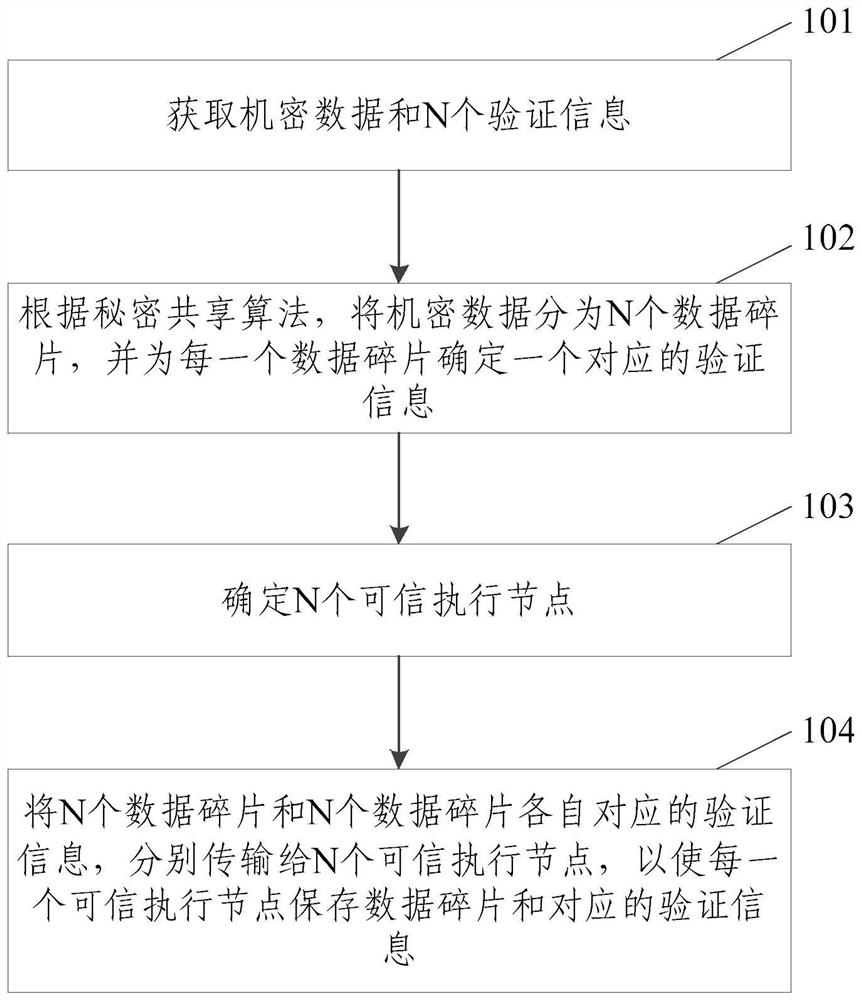
Confidential data management method, electronic device, storage medium and program product
PendingCN114117471AReduce the risk of collusionImprove safety and reliabilityDigital data protectionShardEngineering
The invention relates to the technical field of block chains, in particular to a confidential data management method, electronic equipment, a storage medium and a program product. The method is applied to a client side and comprises the steps that confidential data and N pieces of verification information are obtained, and N is an integer larger than 0; according to a secret sharing algorithm, dividing the confidential data into N data fragments, and determining one piece of corresponding verification information for each data fragment; n trusted execution nodes are determined, and the trusted execution nodes are nodes containing trusted execution environments; and respectively transmitting the N data fragments and the verification information corresponding to the N data fragments to the N trusted execution nodes, so that each trusted execution node stores the data fragments and the corresponding verification information. The method is used for overcoming the defect that a confidential data management mechanism is poor in safety and reliability in the prior art, and high reliability of confidential data management is achieved.
Owner:冯伊娜
Insulating pull rod with operation point capable of being monitored
InactiveCN105762689AEasy to monitorConvenient lightingLighting elementsContact operating partsPower gridOperation point
The invention discloses an insulating pull rod with an operation point capable of being monitored. The insulating pull rod comprises an operation segment, a joint segment, and a rod body, wherein the units are connected successively. A camera is arranged on the rod body and is installed on the rod body by a mounting support; and a viewfinder window of the camera faces the operation segment of the insulating pull rod. A transparent illumination chamber is arranged between the rod body and the joint segment; and an illumination lamp is installed in the illumination chamber. When the insulating pull rod is in use, a worker can adjust the angle between the camera mounting support and the rod body in advance according to any demand before usage of the insulating pull rod; and during the maintenance process, a corresponding operation point position image is obtained. Good illumination for the operation point can be realized only by turning on the illumination lamp at night or in an environment with dark light, so that the camera can obtain a clear image; and thus the worker can monitor the operation point conveniently during the maintenance process, so that an operation error can be avoided and thus a safety accident of a power grid can be avoided.
Owner:JURONG HUAYUAN ELECTRICAL APPLIANCE EQUIP
Stable combined electric vehicle frame made of extruded profiles
ActiveCN114132419AIncreased stabilityQuick supportPassenger cyclesCycle safety equipmentsVehicle frameElectric cars
The invention provides a stable combined electric vehicle frame made of extruded profiles, and relates to the technical field of electric vehicle frames, the stable combined electric vehicle frame comprises a blocking frame and an inclination sensor, a goods shelf is welded to one end of the blocking frame, the cross section of the goods shelf is in a shape like a Chinese character'ri ', and a stable structure is arranged on the surface of the blocking frame and comprises two supporting rods; the two supporting rods are slidably inserted into the surface of the blocking frame, the arc face of each supporting rod is sleeved with a spring A, the two ends of each spring A are fixedly connected with the corresponding supporting rod and the blocking frame respectively, a containing groove is formed in the lower end of each supporting rod, a spring B is fixedly connected to the inner wall of each containing groove, and a bottom plate is fixedly connected to the lower end of each spring B. By means of the frame, the problems that after a traditional frame is assembled into an electric vehicle in the later period, the stability of the frame is ordinary, and especially for a three-wheel electric vehicle to go uphill, the frame of the traditional electric vehicle is prone to rollover when running on an uphill road section are solved.
Owner:无锡众行车业科技有限公司
High-altitude automatic downward transporting device for engineering garbage
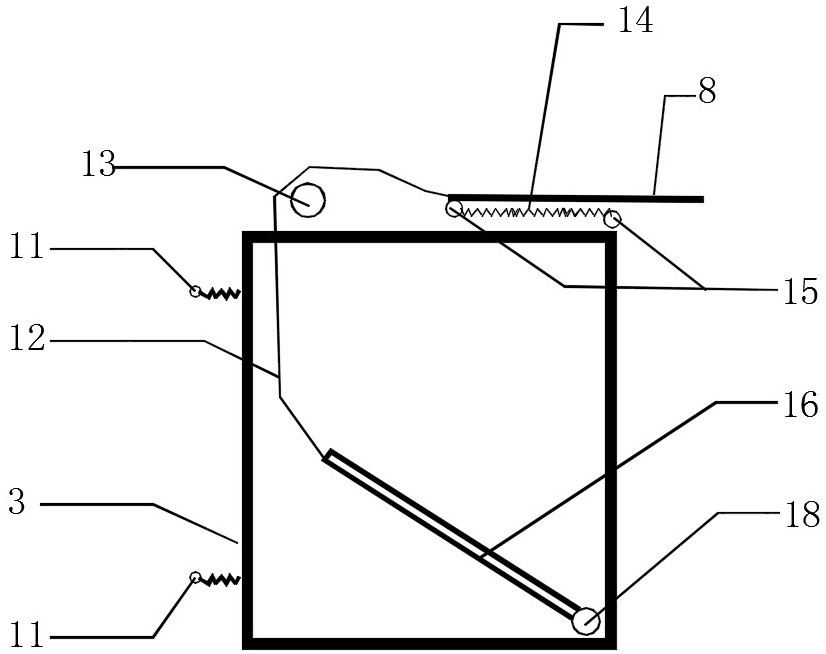
InactiveCN113309320AEnsure personal safetyReduce transportation pressureVertical ductsClassical mechanicsEngineering
The invention discloses a high-altitude automatic downward transporting device for engineering garbage. The high-altitude automatic downward transporting device comprises conveying belts (1), rotating wheels (2), hoppers (3), telescopic positioning rods (8) and a movable supporting plate (16), wherein grooves (9) matched with the corresponding conveying belts are respectively formed in the inner side and the outer side of each rotating wheel; and the two conveying belts go around the corresponding grooves of the upper rotating wheel and the lower rotating wheel respectively to form a closed continuous whole. The hoppers are fixedly connected with the two conveying belts through four elastic supports (11), when a constructor pulls the conveying belts to pull the empty hopper to a floor plate where garbage needs to be conveyed downwards, the corresponding telescopic positioning rod on the top of the hopper pops up and is placed on the floor plate to position the hopper, when the constructor loads the garbage on the floor into the hopper to reach a certain weight, the corresponding telescopic positioning rod is separated from the floor plate to descend the hopper, and the second hopper automatically descends to repeat the work of the first hopper, so that floor garbage downward transportation work is circularly and continuously performed.
Owner:JIANGSU TIANYU CONSTR GRP CO LTD
Remote driving assistance method and system
InactiveCN111572475AImprove securityImprove reliabilityCharacter and pattern recognitionElectric/fluid circuitSound recognitionAutopilot
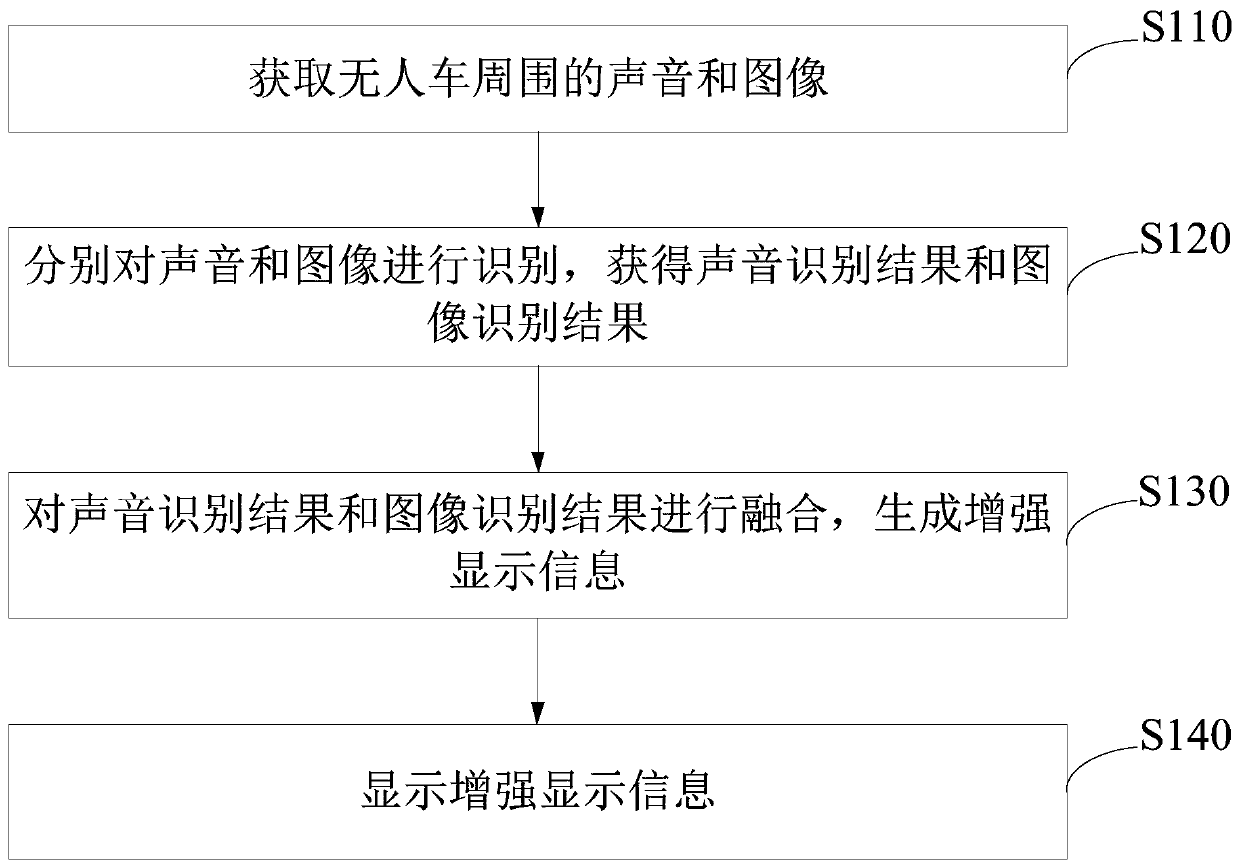
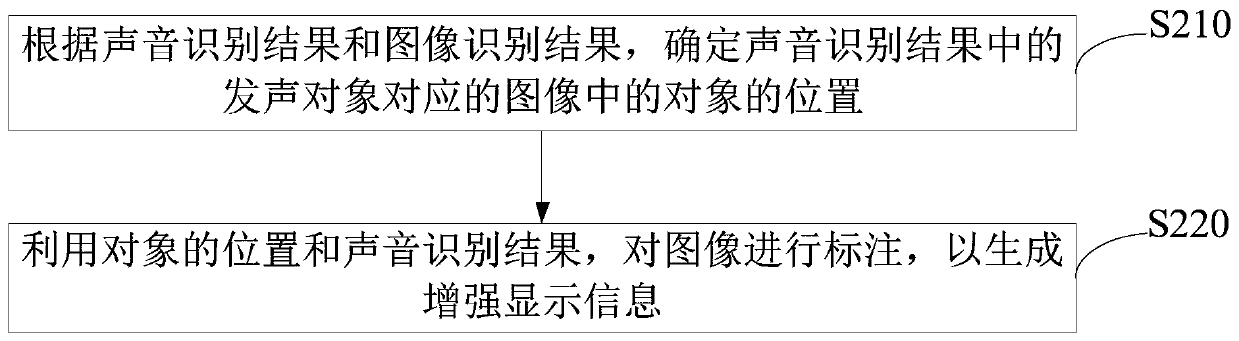
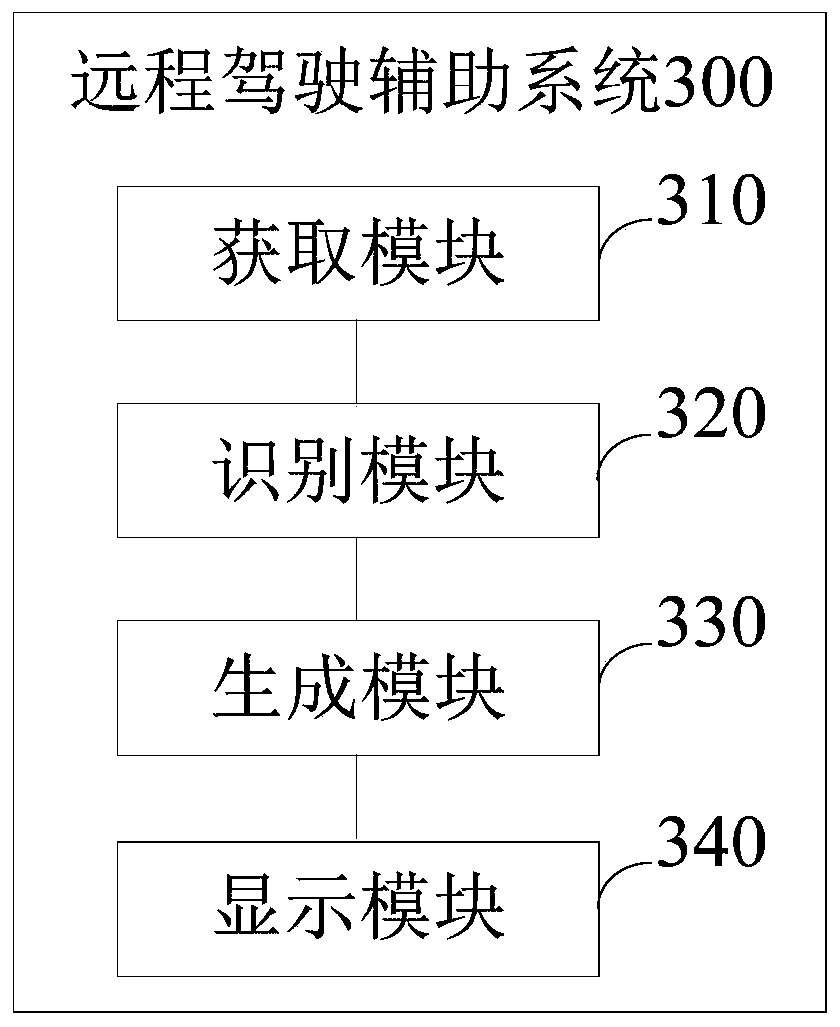
The embodiment of the invention provides a remote driving assistance method and system. The remote driving assistance method comprises the steps that sounds and images around an unmanned vehicle are acquired; the sound and the image are respectively identified to obtain a sound identification result and an image identification result; the voice identification result and the image identification result are fused to generate enhanced display information; and the enhanced display information is displayed. By means of the technical scheme, the workload of a remote operation user is reduced, the situation that safety accidents happen to the unmanned vehicle due to the fact that the remote operation user inaccurately judges the voice can be avoided, and then the safety of the unmanned vehicle with the advantages of automatic driving, unmanned driving and the like is guaranteed.
Owner:NEOLIX TECH CO LTD
Hub polishing machine for engineering vehicle
PendingCN109382734AEnsure connection stabilityAvoid warpingBelt grinding machinesCentrifugal forceRotating disc
The invention relates to the technical field of engineering machinery, in particular to a hub polishing machine for an engineering vehicle. A rotary base is used for installing a hub, a rotating driving assembly is installed below a supporting plate to drive the hub to rotate, and a polishing assembly is arranged above the rotating base and used for grinding the surface of the hub; a stop assemblyis arranged on the side, on the rotary base, of the supporting plate; the stop assembly comprises a rotating disc, a rotating disc driving assembly, brake blocks and a return assembly; the rotating disc is rotationally connected with the lower portion of the rotary base, the brake blocks are uniformly distributed on the end face of one side of the rotary disc in the circumferential direction, andthe return assembly is arranged between the brake blocks and the rotating disc; the return assembly is used for driving the brake blocks to rotate towards the central axis of the rotating disc, and the rotating disc drive assembly is used for driving the rotating disc to rotate, that is, the rotating disc provides centrifugal force to the brake blocks so as to make the brake blocks extend outwards to abut against the inner ring of the hub. The problems that when a large hub is polished, the single-side force is too large, accordingly the hub upwarps or even turns over, and the polishing quality is affected are solved.
Owner:臧其亮
An electric vehicle charging gun
ActiveCN108551039BEasy to useSafe escapeCoupling device detailsStructural engineeringElectric vehicle
The invention relates to an electric vehicle charging gun, comprising a gun body provided with an inner cavity, a plug, a locking member and a wire harness, wherein a clamping sleeve is inserted on the gun body in a sliding manner; the clamping sleeve and the gun body are fixedly connected in a sliding manner through at least one ball plunger; the wire harness is fixedly connected with the clamping sleeve, and one end of the wire harness is placed in the inner cavity and connected with the plug; the locking member is laterally rotatably mounted on the gun body; the gun body is provided with afirst spring, and the locking member rotates counterclockwise under the elastic force of the first spring; the gun body is provided with a sliding rod that slides up and down, the upper end of the sliding rod is provided with a connecting rod, the lower end of the connecting rod is hinged with the upper end of the sliding rod, the upper end of the connecting rod is provided with a straight groove,the right end of the locking member is provided with a hinge shaft, and the hinge shaft is arranged in the straight groove; a second spring is arranged on the gun body, and the sliding rod slides upward through the elastic force of the second spring; and the sliding rod is connected with the clamping sleeve. The electric vehicle charging gun in the invention can satisfy normal use when charging the vehicle and also can ensure that the charging gun is pulled off safely from the vehicle in case of emergency.
Owner:安徽广德佳欣电磁制动器有限公司
Sintering machine blowing device capable of adjusting blowing starting point in self-adaptation manner and blowing method thereof
PendingCN108120294AImprove securityEnhanced auxiliary sintering effectCharge treatment typeFurnace typesSafety coefficientFuel gas
The invention discloses a sintering machine blowing device capable of adjusting blowing starting point in a self-adaptation manner. The device comprises a sintering machine trolley, a blowing cover and a gas blowing device, the gas blowing device comprises a gas blowing main pipe, a gas blowing branch pipe and a gas blowing pipe bundle, the sintering machine trolley is located in the blowing cover, the gas blowing main pipe is arranged on the outer side of the blowing cover, the gas blowing pipe bundle is arranged above the sintering machine trolley, the gas blowing pipe bundle is located in the blowing cover, one end of the gas blowing branch pipe is connected with the gas blowing main pipe, the other end of the gas blowing branch pipe is connected with the gas blowing pipe bundle, the gas blowing pipe bundle comprises multiple gas blowing pipes which are provided with gas blowing holes. The device is characterized in that multiple air boxes are arranged below the sintering machine trolley, and an air box temperature measuring device is arranged at the position, on each air box, of the lower portion of the sintering machine trolley. According to the device, the blowing starting point can be adjusted in the self-adaptation manner, defects in the prior art can be effectively overcome, safety coefficients can be improved, and the assisting effect is ensured.
Owner:ZHONGYE-CHANGTIAN INT ENG CO LTD
Low-temperature denitration catalyst wet zone production equipment and process
PendingCN113357898AImprove drying efficiencyHigh degree of mechanizationDrying chambers/containersDrying machines with progressive movementsProcess engineeringEngineering
The invention discloses low-temperature denitration catalyst wet zone production equipment and process, and relates to the technical field of denitration catalyst production equipment. The low-temperature denitration catalyst wet zone production equipment comprises a drying chamber, a second conveying line is arranged in the middle of the drying chamber in a penetrating mode, a first conveying line parallel to the second conveying line is arranged on the left side of the drying chamber, a fifth conveying line is arranged between the front end of the first conveying line and the front end of the second conveying line, the fifth conveying line is perpendicular to the first conveying line and the second conveying line, and a third ferry vehicle is arranged on the fifth conveying line. The equipment has the beneficial effects that a conveying area is arranged to convey a skip car, a circulating area is arranged to thoroughly dry the skip car and then send out the skip car, and the drying efficiency is improved; a first ferry vehicle, a second ferry vehicle and the third ferry vehicle are arranged for transferring the skip car, the mechanization degree is improved, and human resource investment is reduced; and a protective net is arranged to avoid safety accidents caused by mistakenly entering an operation area by people without duties.
Owner:郑州登电昊南热陶瓷材料有限公司
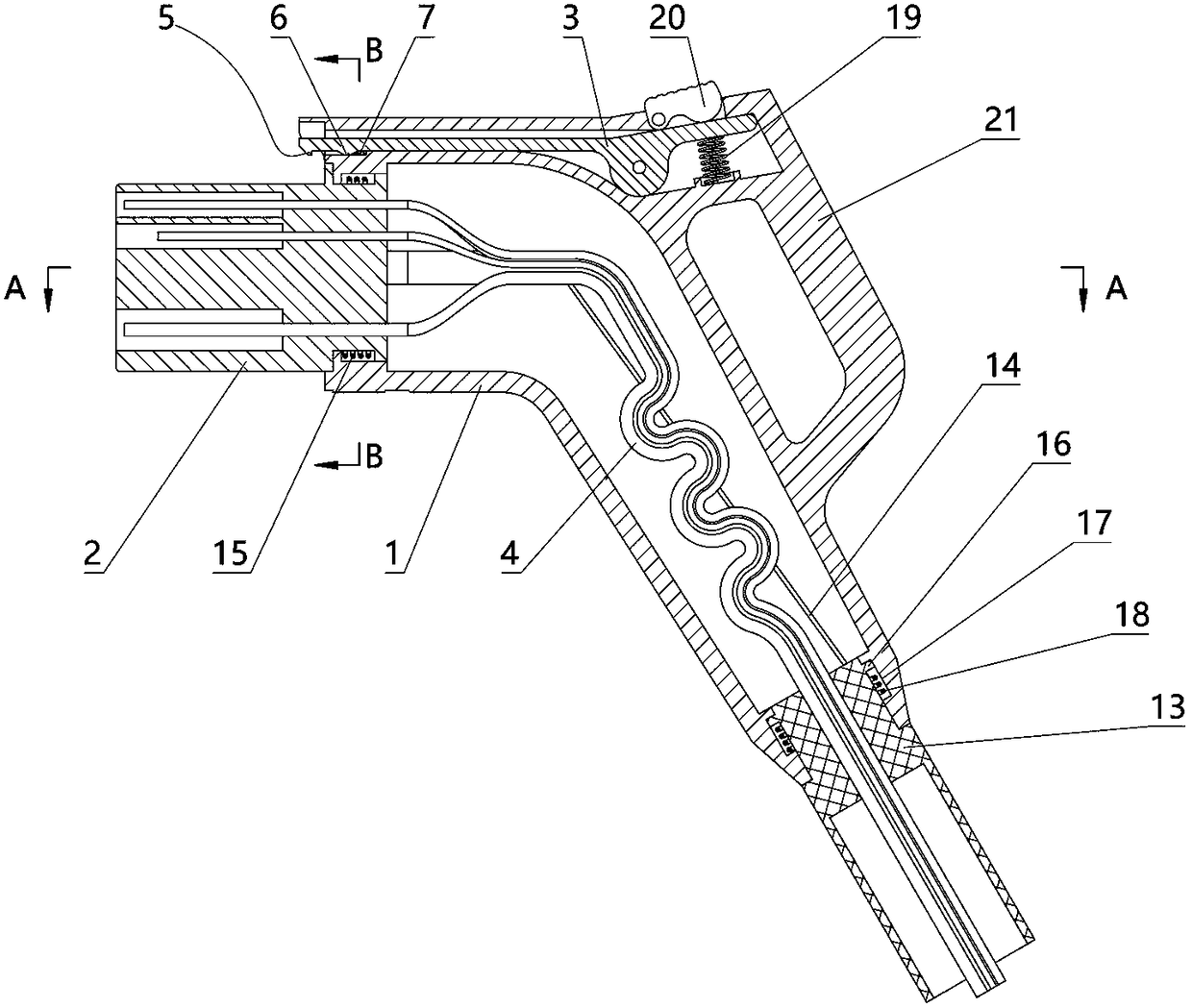
A charging gun capable of forced power-off
ActiveCN108598774BEasy to useAvoid breakingCharging stationsCoupling device detailsControl theoryTorsion spring
The invention relates to a charging gun allowing forced power off. The charging gun comprises a gun body with an inner cavity, a plug, a lock piece, a wire harness, and a cable, wherein a plug sleeveis inserted in the gun body in a sliding manner, the plug sleeve and the gun body are in slide positioning connection through at least one ball plunger, an insert end of the plug sleeve has a plug hole, the gun body has the plug matched with the plug hole, the plug is inserted in the plug hole, the wire harness is in the inner cavity, one end of the wire harness is connected with the plug, the other end of the wire harness is connected with the plug, one end of the cable is connected with the plug hole, the plug and the gun body are connected in a rotating manner, a torsion spring is sheathedat the right of the plug, and two ends of the torsion spring are respectively connected with the gun body and the plug. The charging gun can be used normally when a vehicle is charged, and in emergency, it is ensured that the charging gun cuts off the power when being pulled and releases the vehicle safely.
Owner:湖州浪佩智能科技有限公司
Dust collector
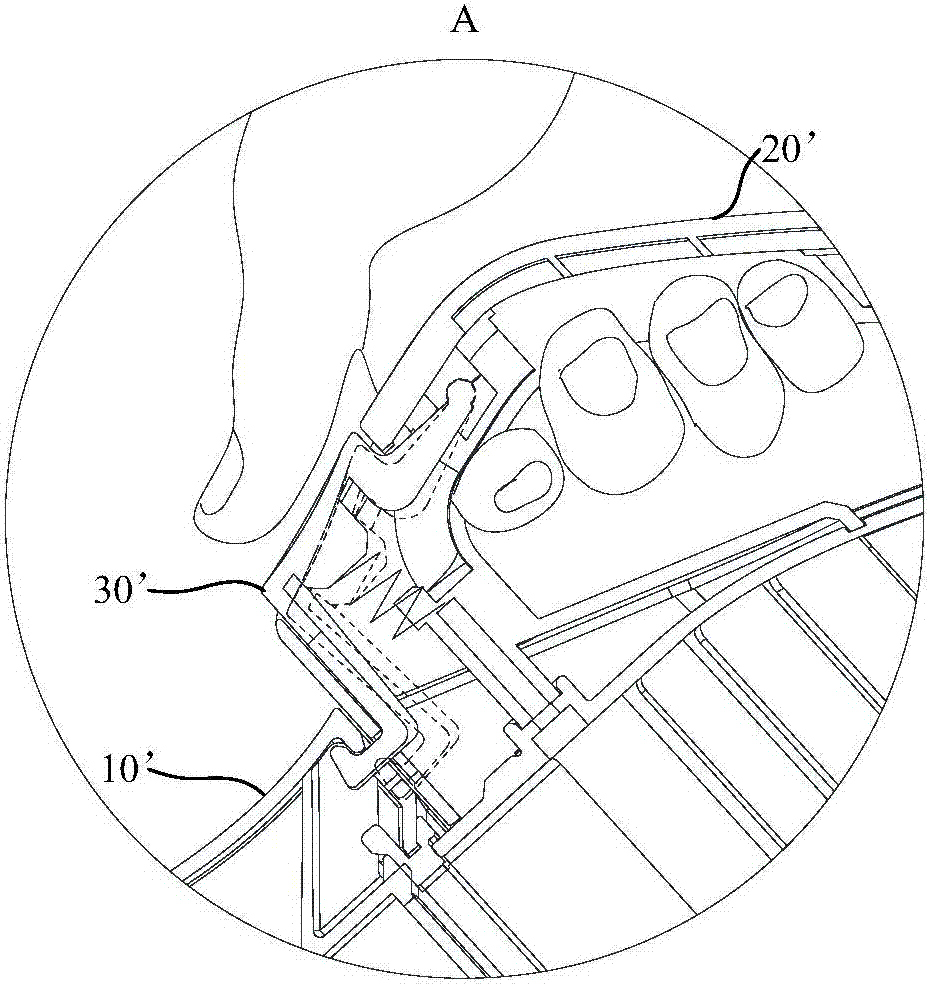
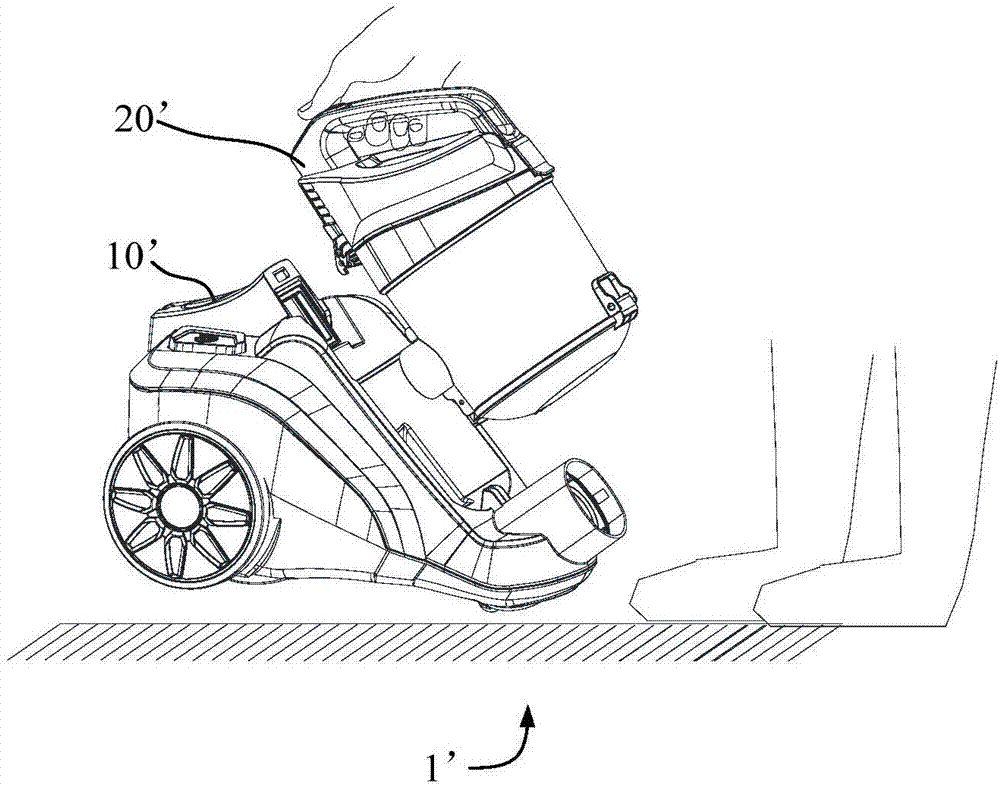
The invention provides a dust collector. The dust collector comprises a machine body and a dust cup, wherein the machine body is provided with an installation part and a first limiting part which is close to the installation part; the dust cup is arranged on the installation cup and provided with a second limiting part, and the first limiting part is located above the second limiting part; when the dust collector is lifted, the dust cup tends to be separated from the machine body due to the action of gravity, so that the first limiting part and the second limiting part move relative to each other, and the second limiting part abuts against the first limiting part; when the dust collector is placed on the flat ground, the second limiting part is separated from the first limiting part. When a user lifts the dust collector, the machine body moves relative to the dust cup, the second limiting part abuts against the first limiting part and prevents the dust cup from moving relative to the machine body, and therefore the machine body cannot be separated from the dust cup; furthermore, when the dust collector is placed on the flat ground, the first limiting part and the second limiting part are separated and do not move relative to each other, the user can conveniently take down the dust cup, and the dust cup is separated from the machine body.
Owner:JIANGSU MIDEA CLEANING APPLIANCES
Integrated reverse-hitting internal-insulation non-dismantling formwork outer wall and preparation method
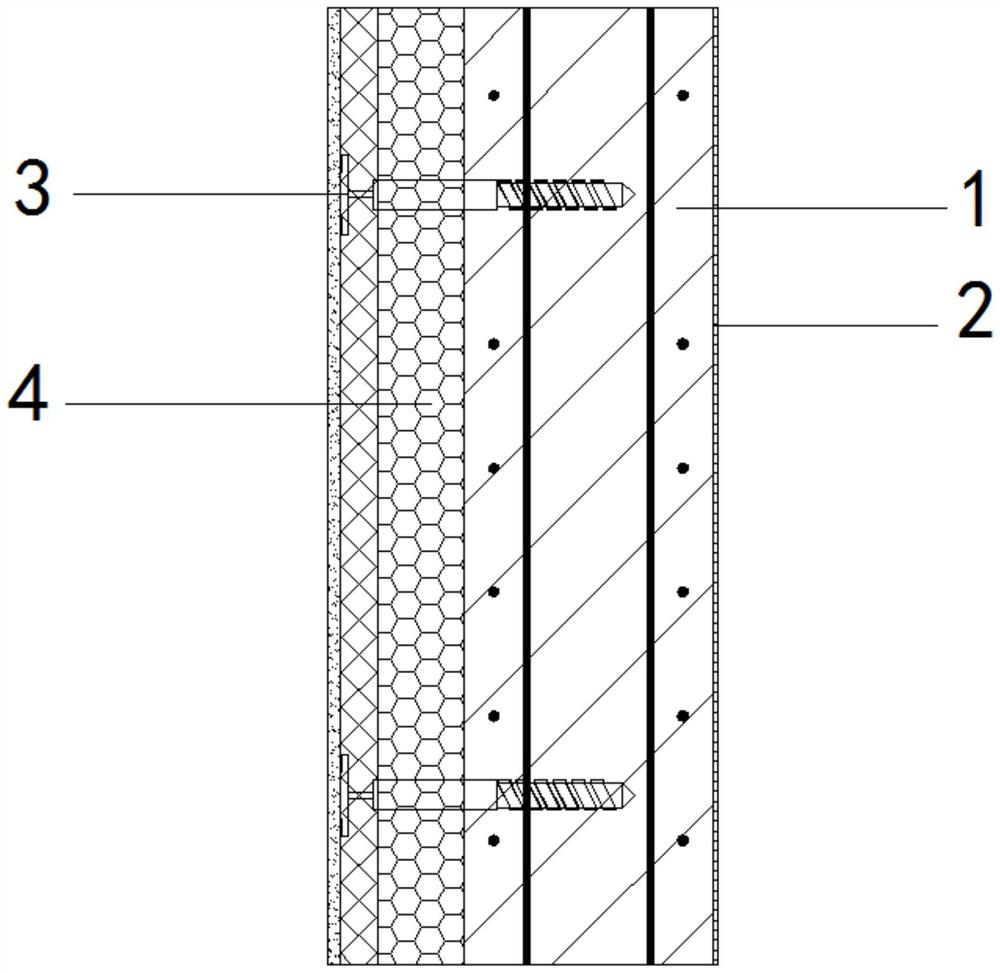
PendingCN114525860ASituations to avoid safety accidentsCovering/liningsWallsClassical mechanicsHeat conservation
The invention relates to the technical field of building external wall heat preservation, in particular to an integrated reverse-hitting internal heat preservation non-dismantling formwork external wall and a preparation method. The integrated reverse-hitting internal heat preservation non-dismantling formwork external wall comprises an external facing, a YZ non-dismantling formwork, a YZ connecting piece and a YZ limiting piece; the outer facing is installed on one side of the shear wall, the YZ non-dismantling formwork is arranged on the side, away from the outer facing, of the shear wall, the YZ connecting piece is connected with the YZ non-dismantling formwork and the shear wall, the YZ limiting piece is matched with the YZ connecting piece, and therefore the outer wall is composed of the facing layer, the shear wall, the YZ connecting piece and the YZ non-dismantling built-in heat preservation formwork, and the heat preservation and protection effects can be effectively achieved; and safety accidents caused by falling of the outer wall body of the building in the whole life cycle are avoided.
Owner:沈阳玖弘建筑科技有限公司
Material boxing equipment and feeding device thereof
PendingCN111846994AImprove loading efficiencyReduce the intensity of manual operationConveyorsLarge containersWoven fabricCushion
The invention relates to the technical field of textiles, and discloses a tension adjusting device in a non-woven fabric bundling process. The tension adjusting device comprises a supporting frame, wherein the back surface of the supporting frame is movably connected with a bundling roller; a stabilizing rod is fixedly mounted at the top end of the supporting frame; the bottom end of the stabilizing rod is movably connected with a roller; the outer wall of the roller is wound with fabric; the fabric is wound with a fixed roller; and the front surface of the fixed roller is movably connected with a roller frame. According to the tension adjusting device in the non-woven fabric bundling process, a top block is matched with cushion gas blocks for use, so that when tension of the fabric is toogreat, the top block in a stressed roller is stressed to move upwards; in a process that the top block moves upwards, four bent rods are in contact, and a coil spring rotates, so that a moving spaceof a movable rod is increased, and the bent rods rotate and stretch; and when the bent rods rotate and stretch, the four cushion gas blocks extrude each other, and gas in the cushion gas blocks is extruded out through gas leaking holes, so that the effects of extruding the gas into a gas pressure rod and triggering constant gas pressure unbalance in the gas pressure rod are achieved.
Owner:苗林展
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com