Patents
Literature
979 results about "Cardiac cycle" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
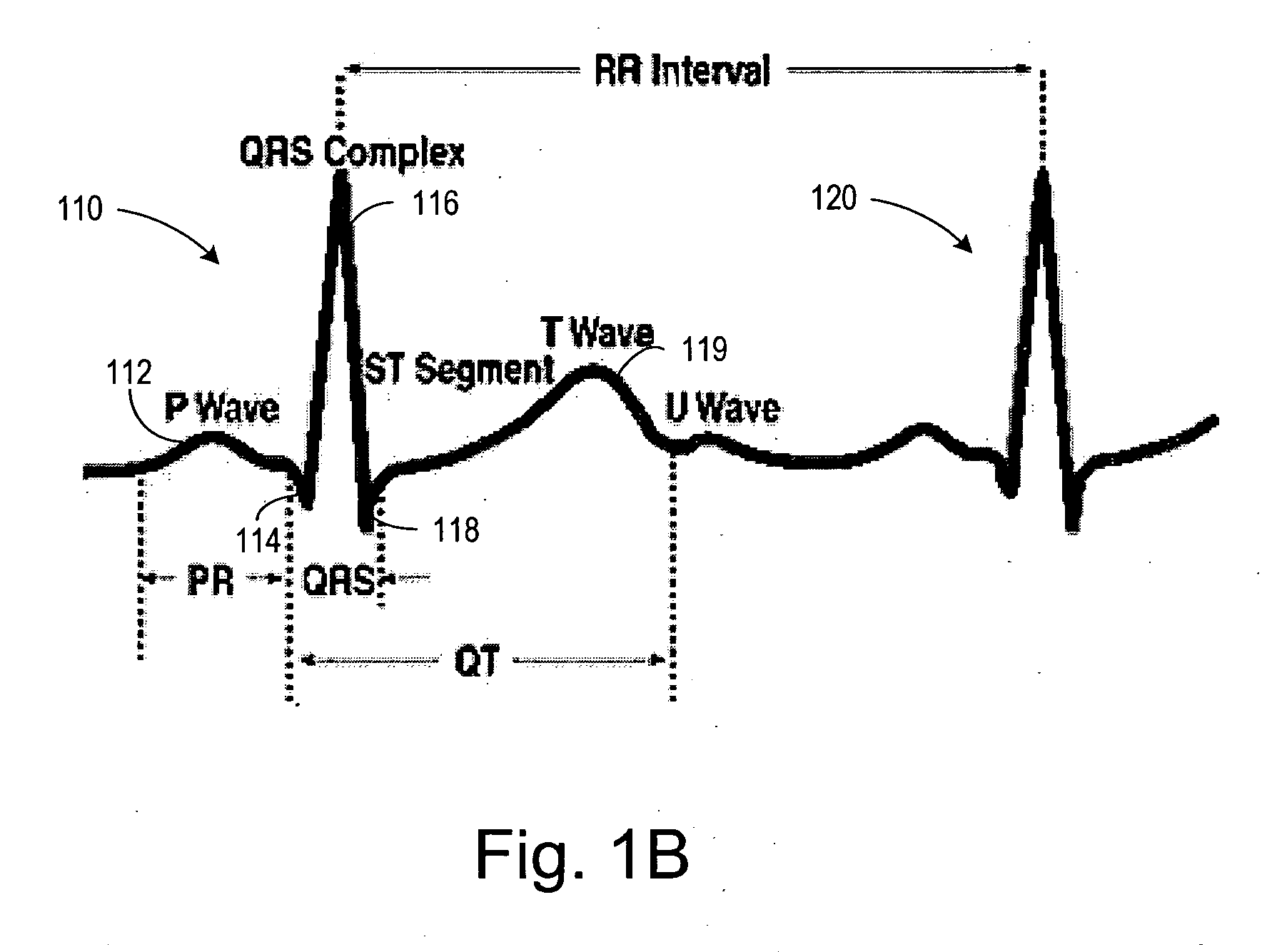
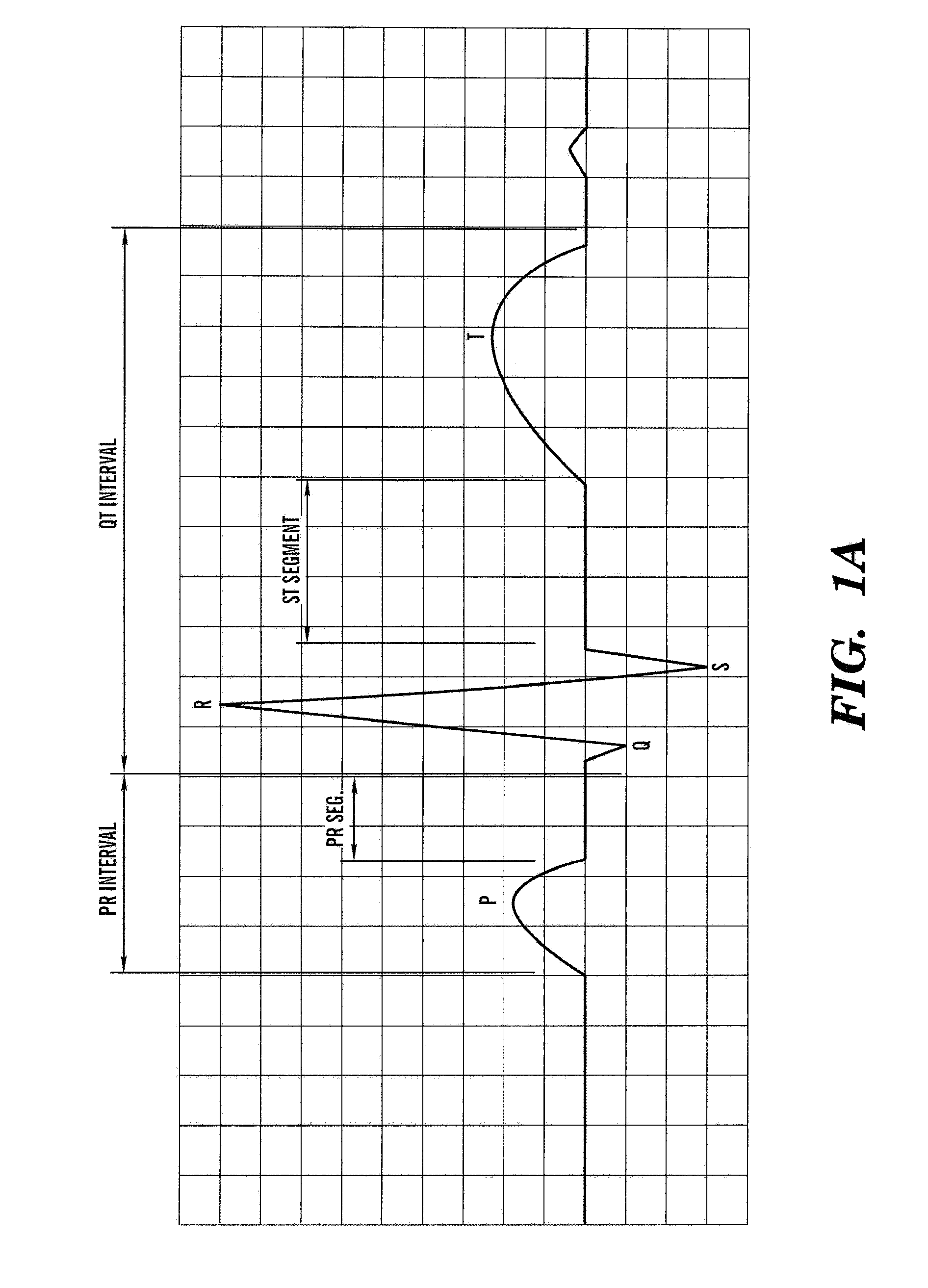
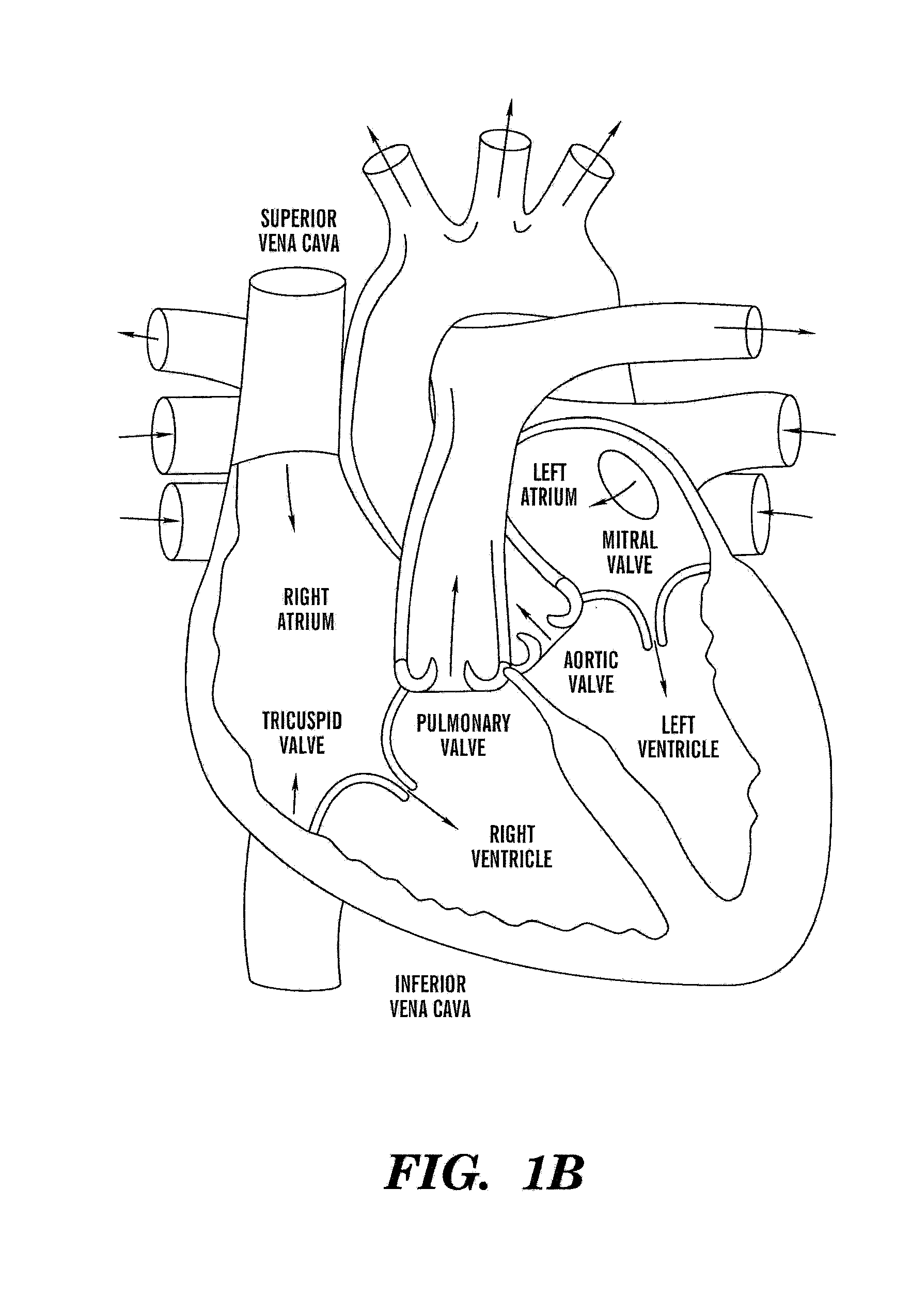
The cardiac cycle is the performance of the human heart from the ending of one heartbeat to the beginning of the next. It consists of two periods: one during which the heart muscle relaxes and refills with blood, called diastole (/daɪˈæstəli/), followed by a period of robust contraction and pumping of blood, dubbed systole (/sɪsˈtəli/). After emptying, the heart immediately relaxes and expands to receive another influx of blood returning from the lungs and other systems of the body, before again contracting to pump blood to the lungs and those systems. A normally performing heart must be fully expanded before it can efficiently pump again. Assuming a healthy heart and a typical rate of 70 to 75 beats per minute, each cardiac cycle, or heartbeat, takes about 0.8 seconds to complete the cycle.
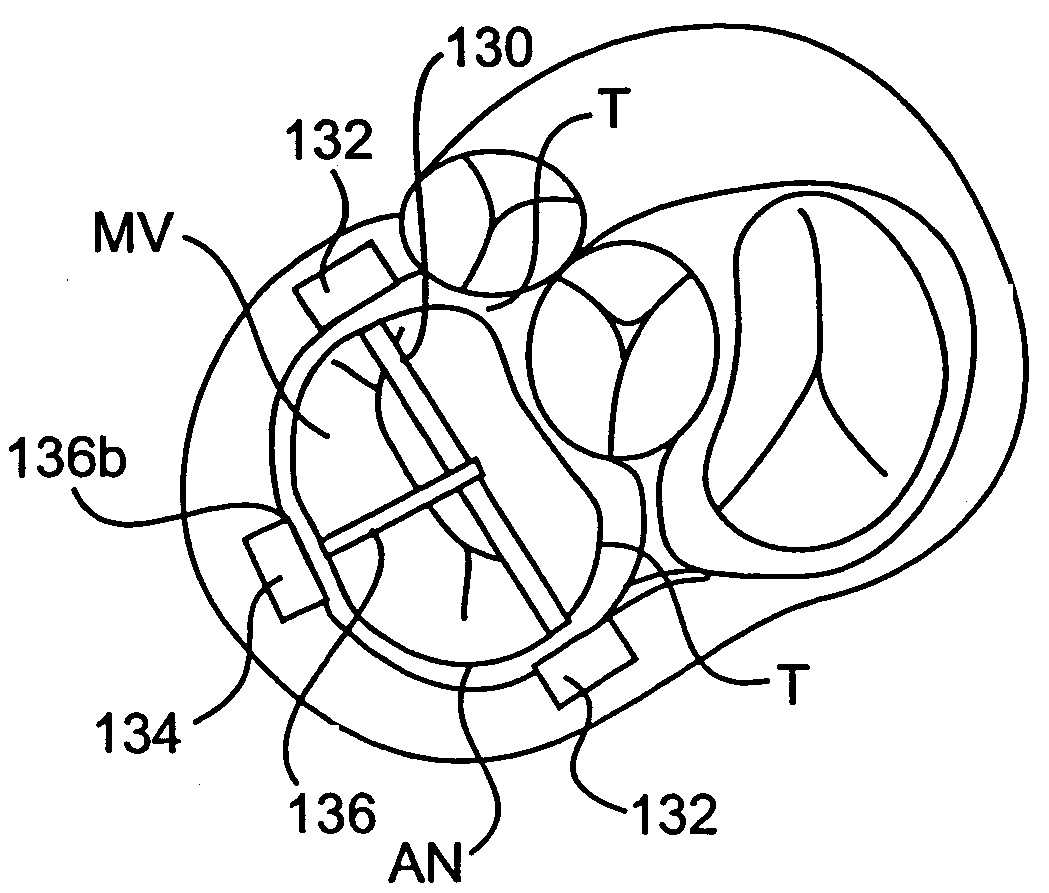
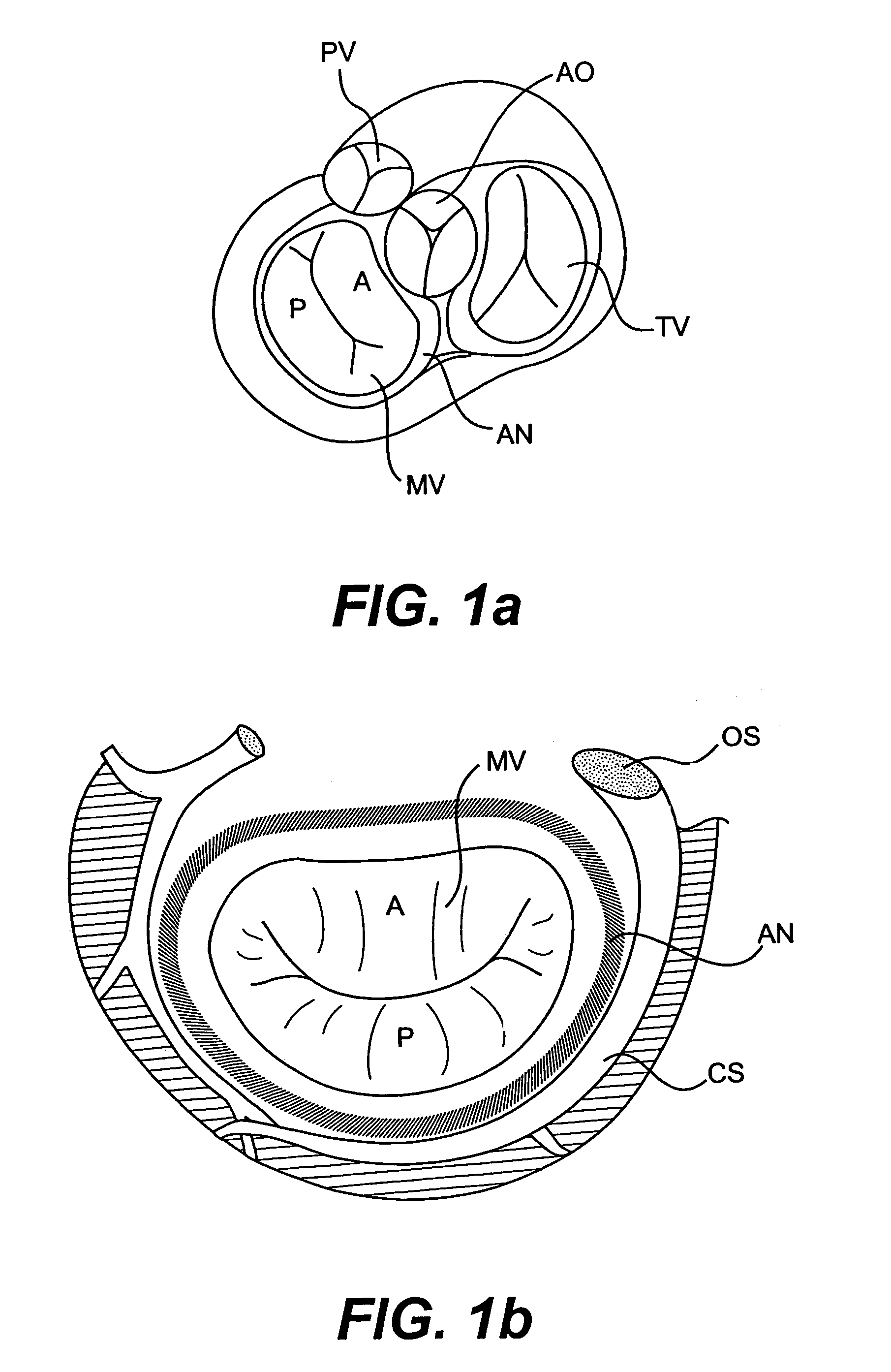
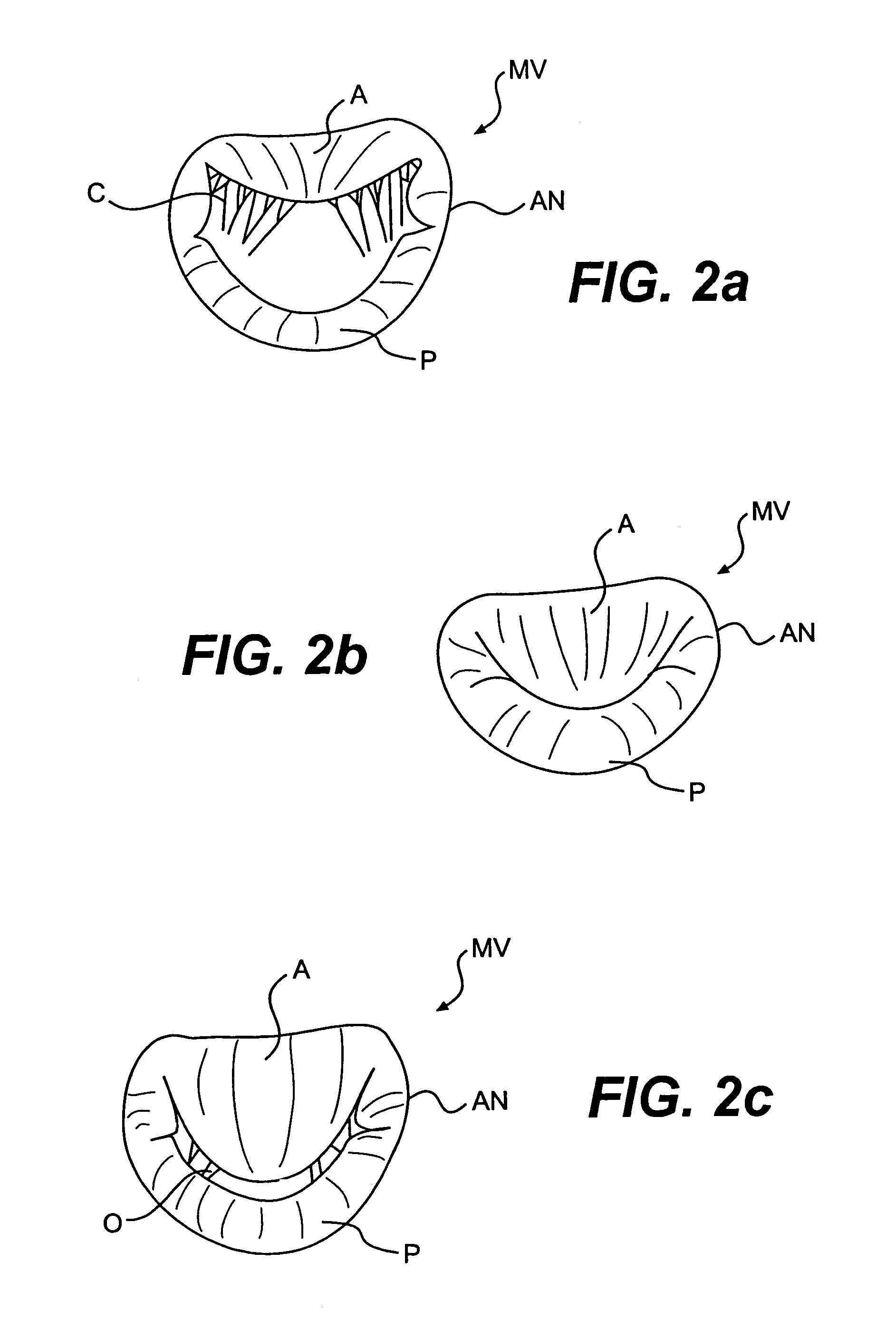
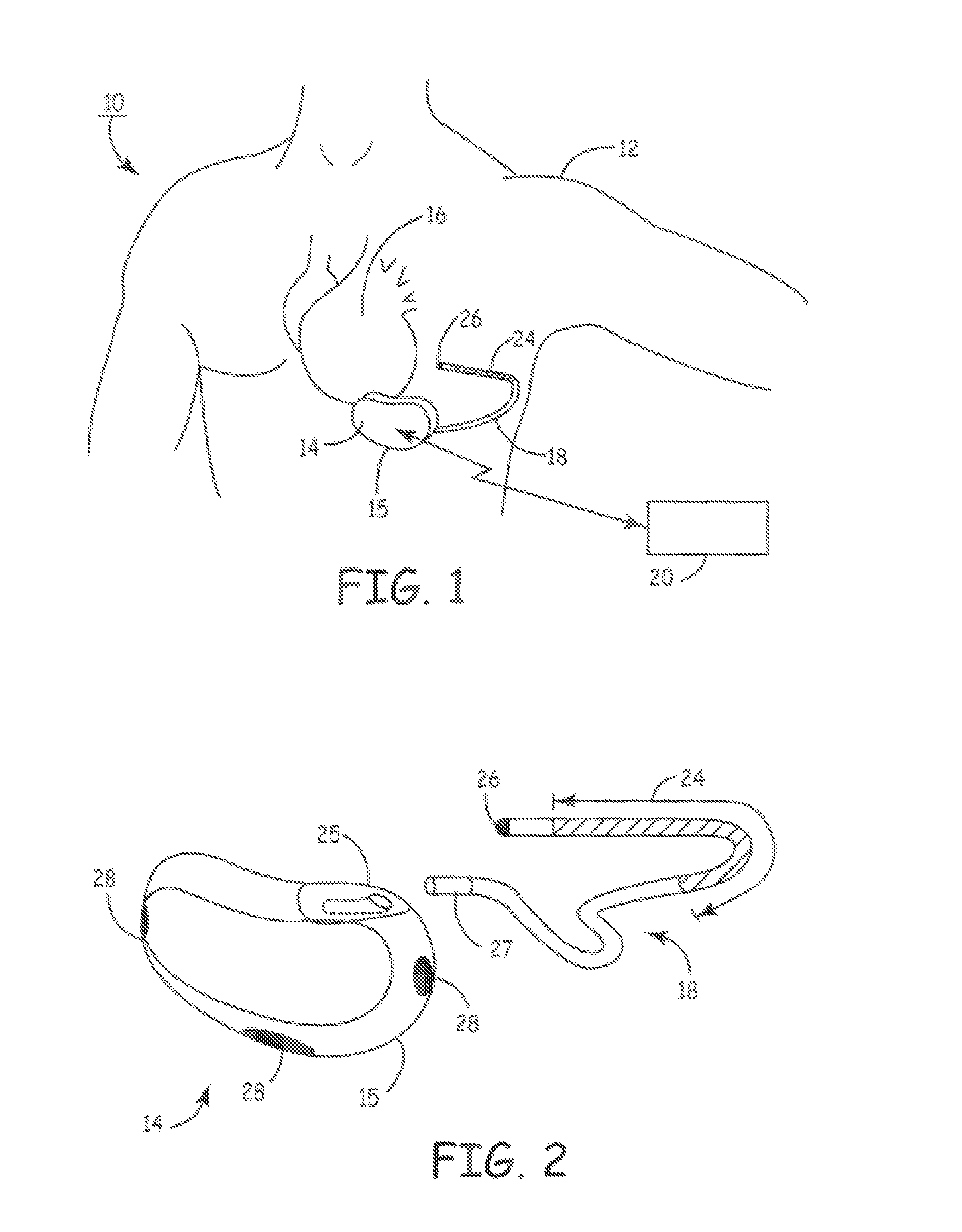
Devices and methods for heart valve treatment
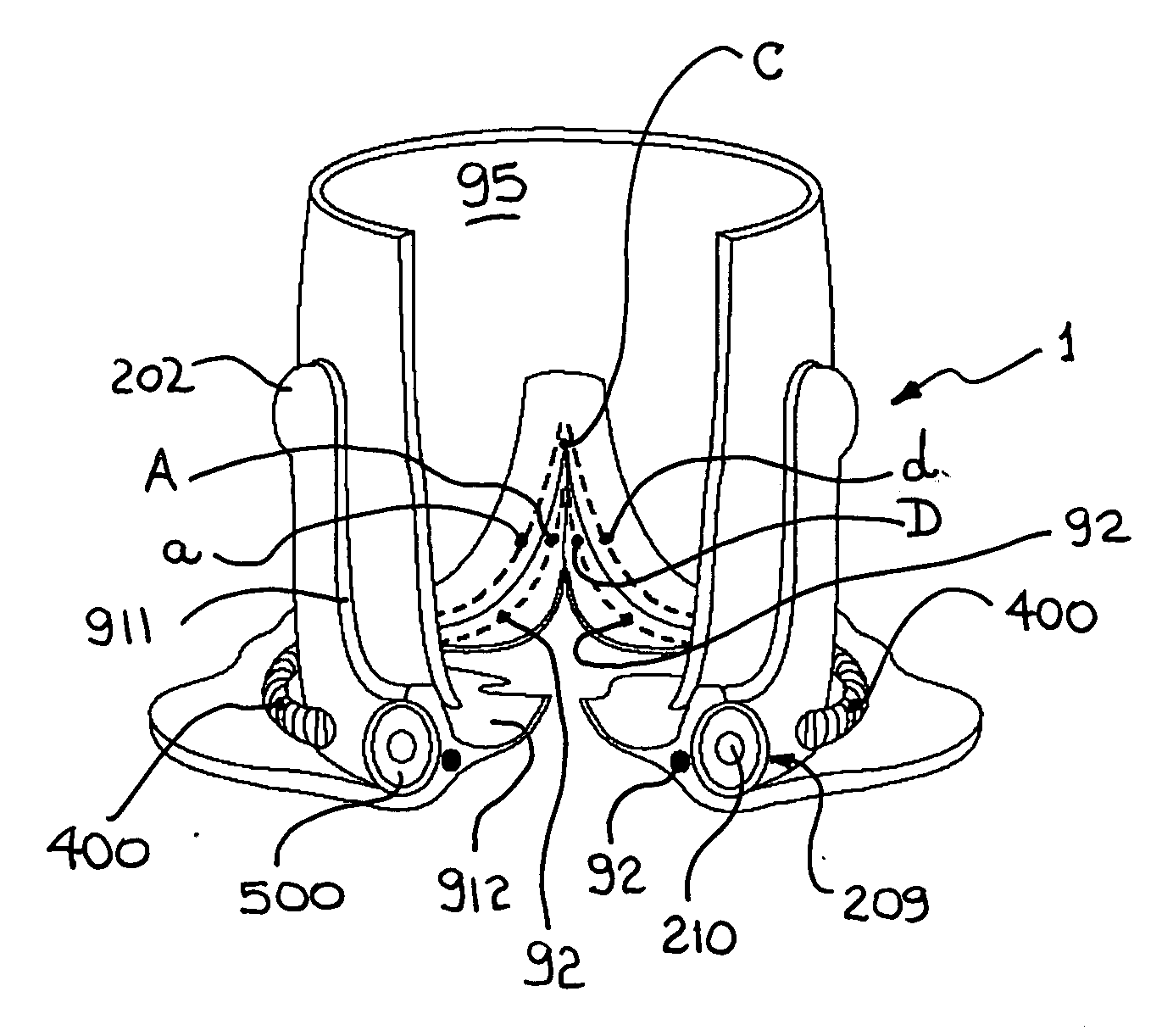
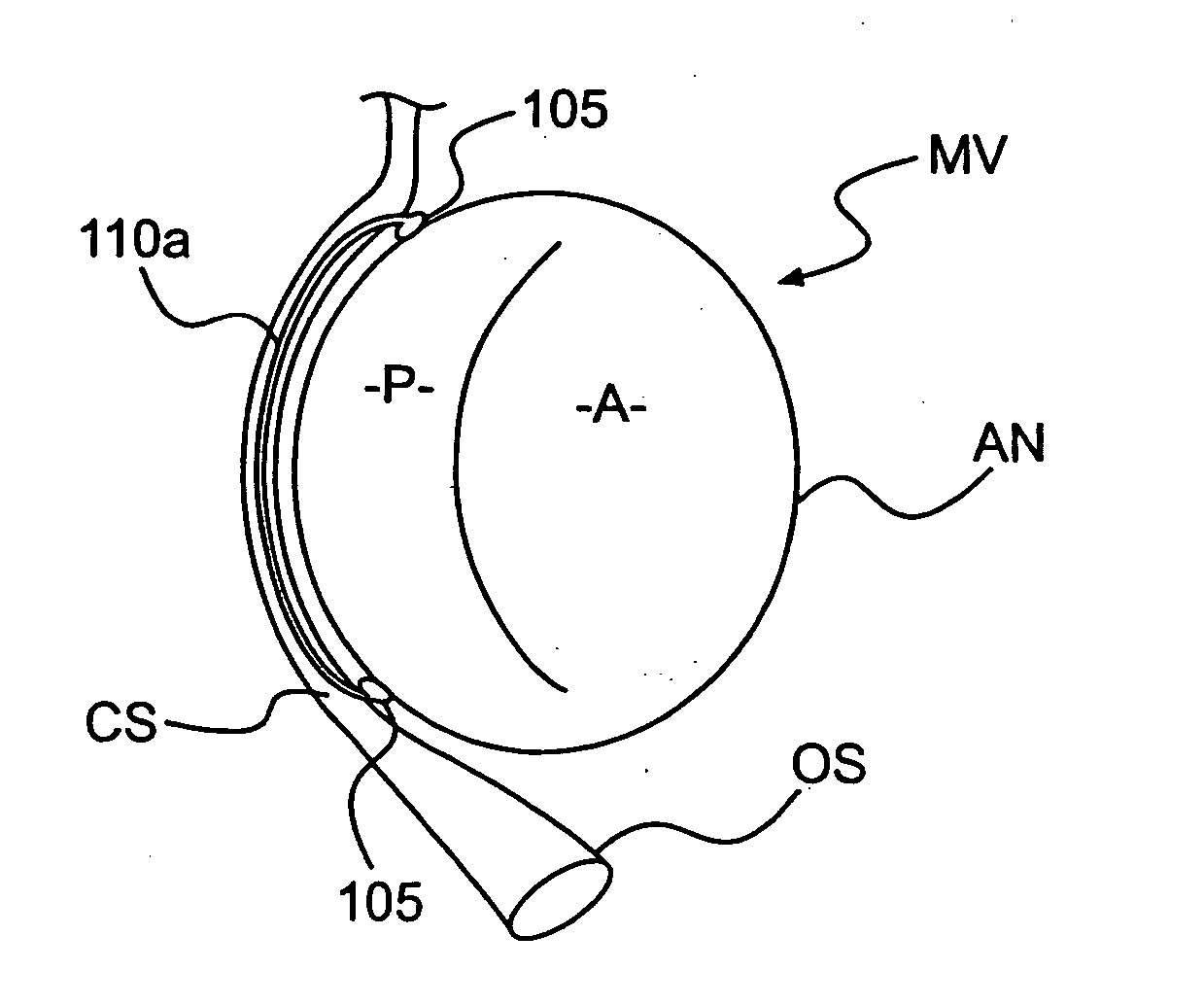
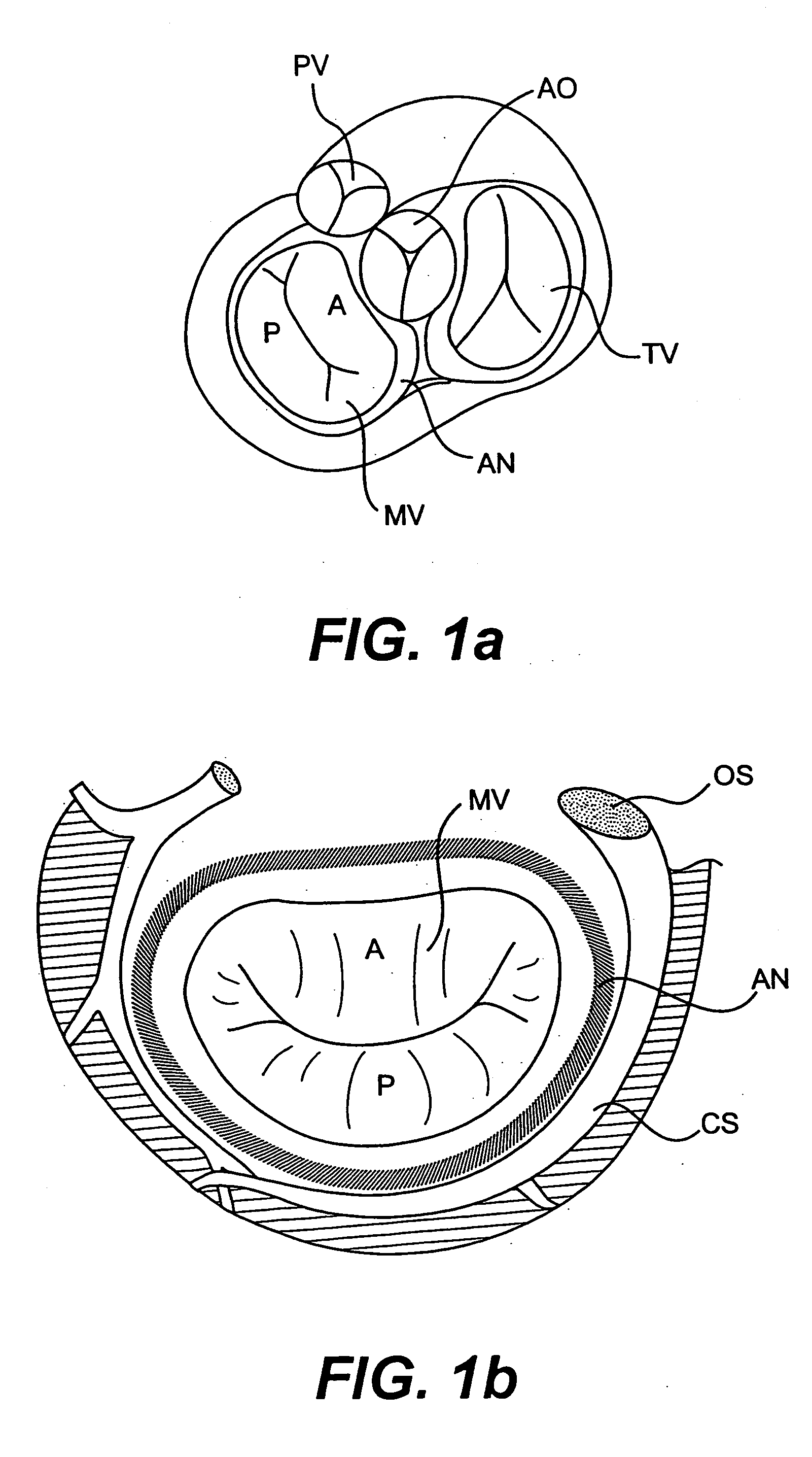
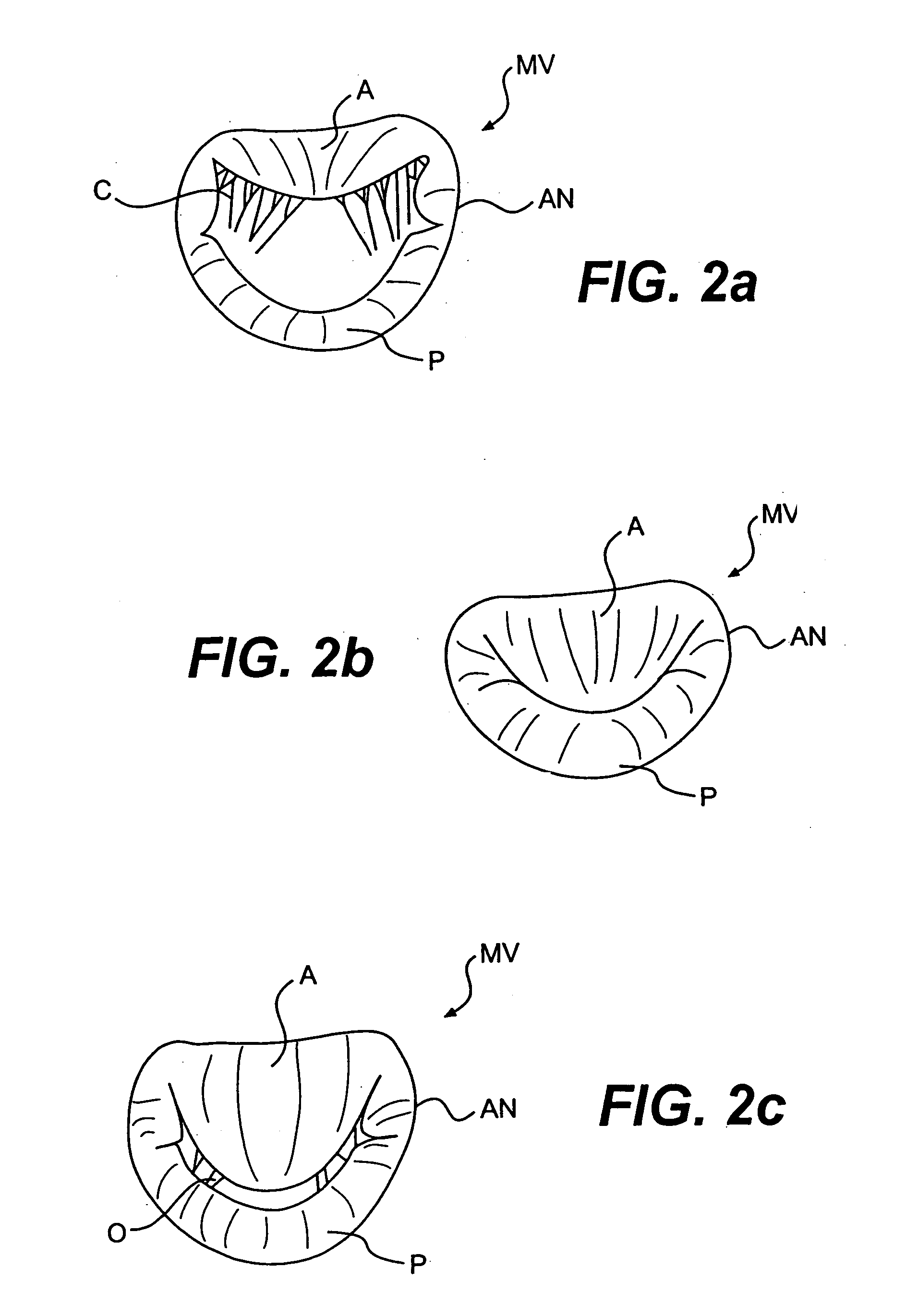
Devices and methods for treating heart valves include members that assist the valve in closing during at least a portion of the cardiac cycle. Such devices include members configured to alter the shape of a valve annulus, reposition at least one papillary muscle, and / or plug an orifice of the valve so as to provide a coaptation surface for the valve leaflets.
Owner:EDWARDS LIFESCIENCES LLC
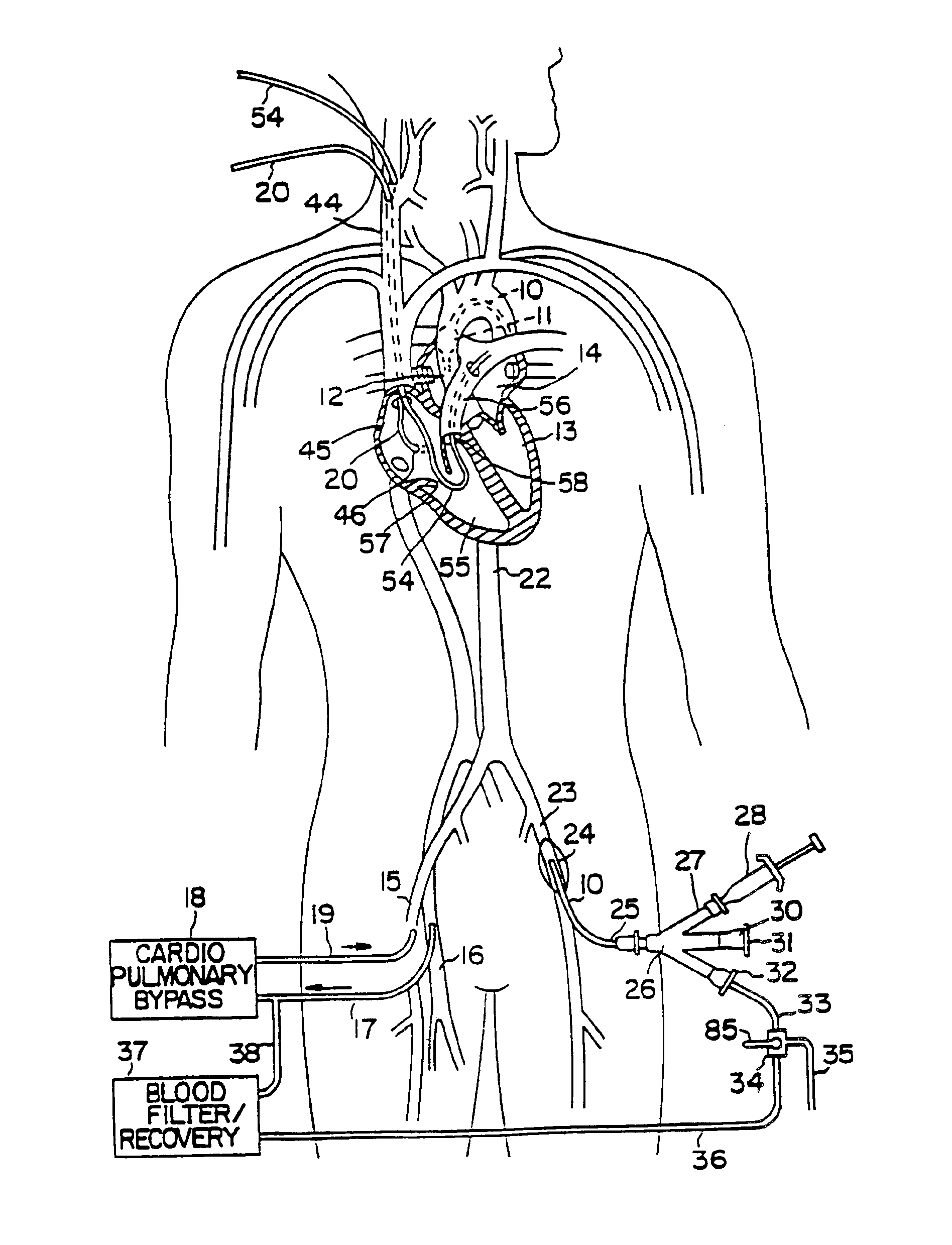
System for cardiac procedures
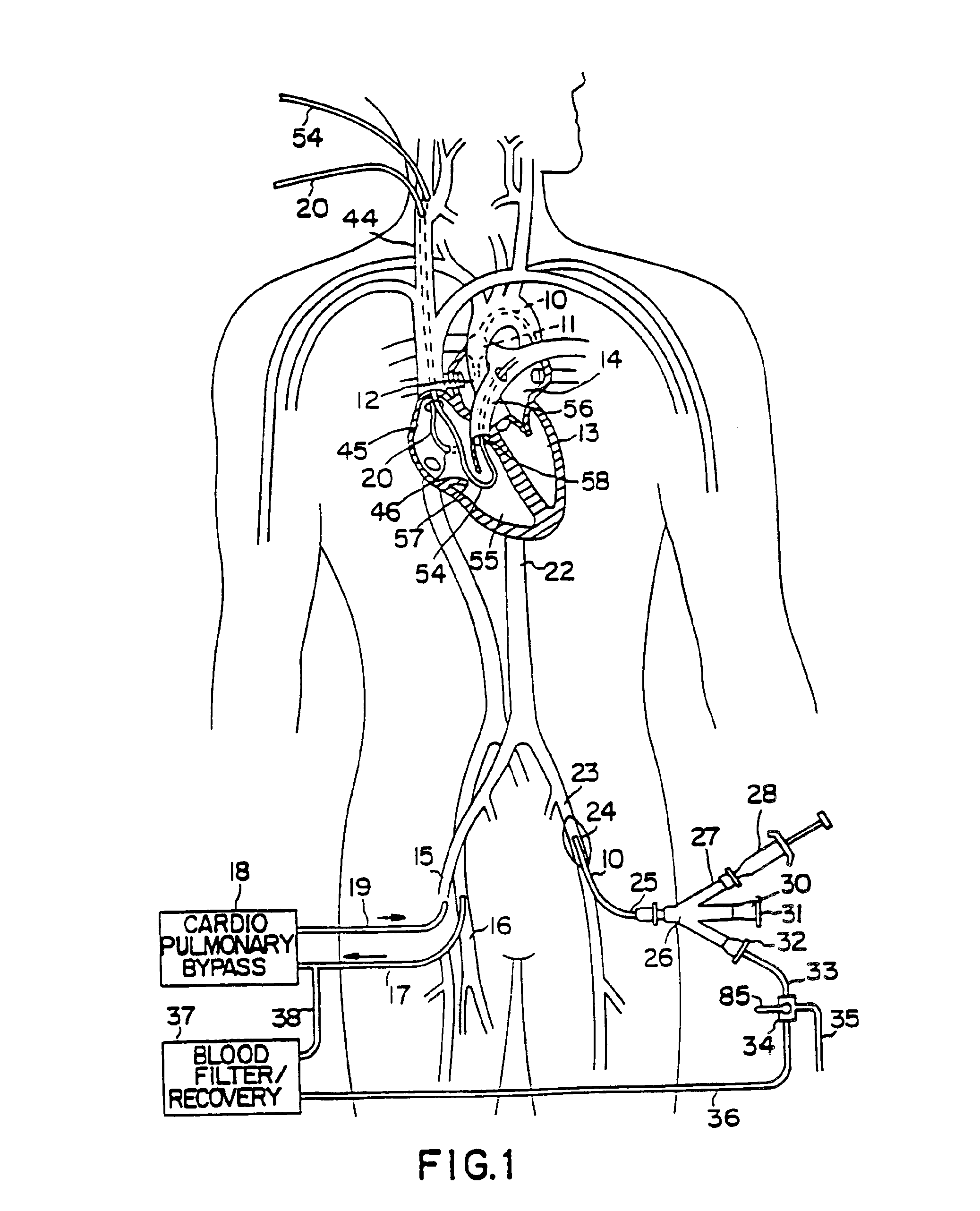
A system for accessing a patient's cardiac anatomy which includes an endovascular aortic partitioning device that separates the coronary arteries and the heart from the rest of the patient's arterial system. The endovascular device for partitioning a patient's ascending aorta comprises a flexible shaft having a distal end, a proximal end, and a first inner lumen therebetween with an opening at the distal end. The shaft may have a preshaped distal portion with a curvature generally corresponding to the curvature of the patient's aortic arch. An expandable means, e.g. a balloon, is disposed near the distal end of the shaft proximal to the opening in the first inner lumen for occluding the ascending aorta so as to block substantially all blood flow therethrough for a plurality of cardiac cycles, while the patient is supported by cardiopulmonary bypass. The endovascular aortic partitioning device may be coupled to an arterial bypass cannula for delivering oxygenated blood to the patient's arterial system. The heart muscle or myocardium is paralyzed by the retrograde delivery of a cardioplegic fluid to the myocardium through patient's coronary sinus and coronary veins, or by antegrade delivery of cardioplegic fluid through a lumen in the endovascular aortic partitioning device to infuse cardioplegic fluid into the coronary arteries. The pulmonary trunk may be vented by withdrawing liquid from the trunk through an inner lumen of an elongated catheter. The cardiac accessing system is particularly suitable for removing the aortic valve and replacing the removed valve with a prosthetic valve.
Owner:EDWARDS LIFESCIENCES LLC
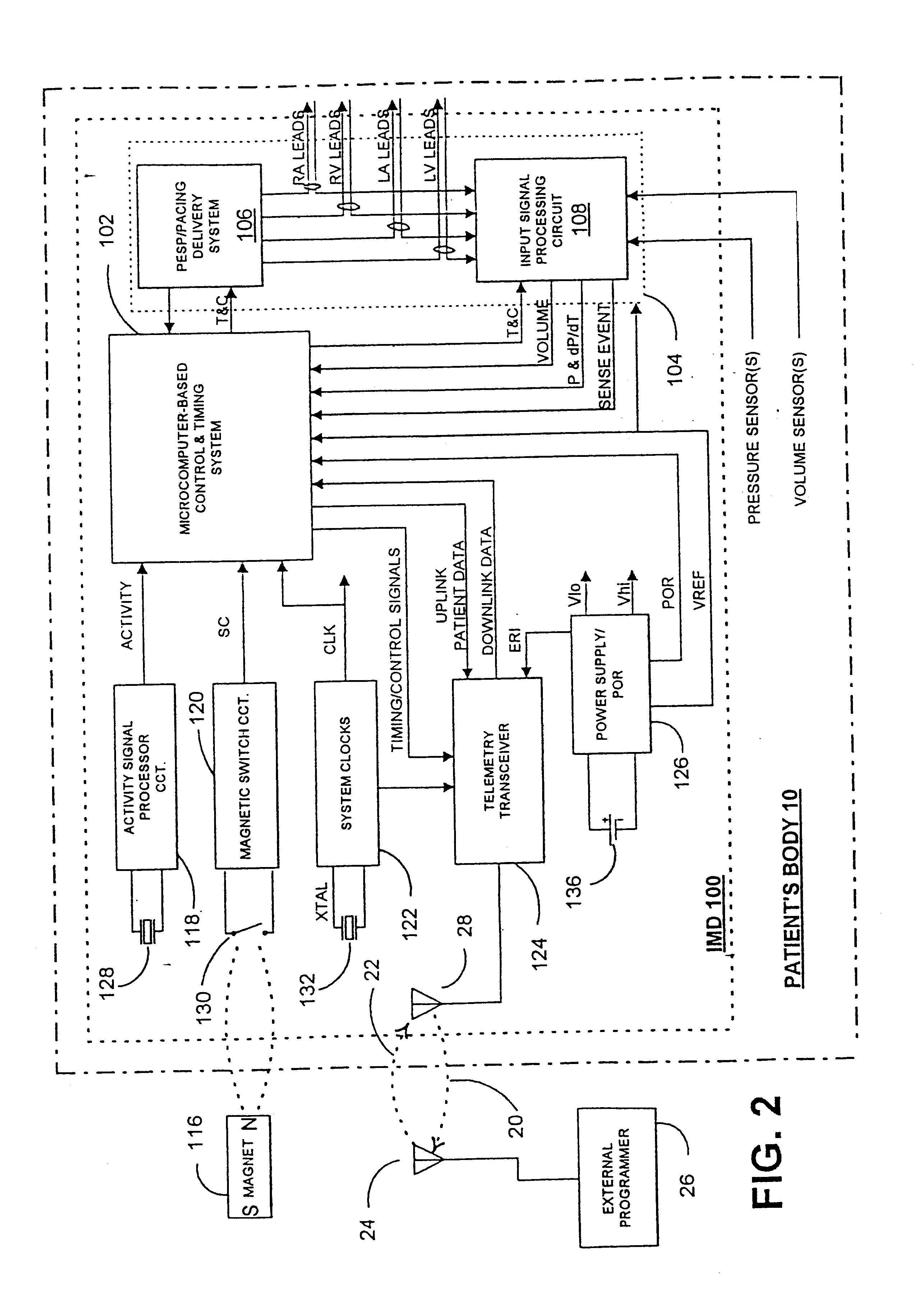
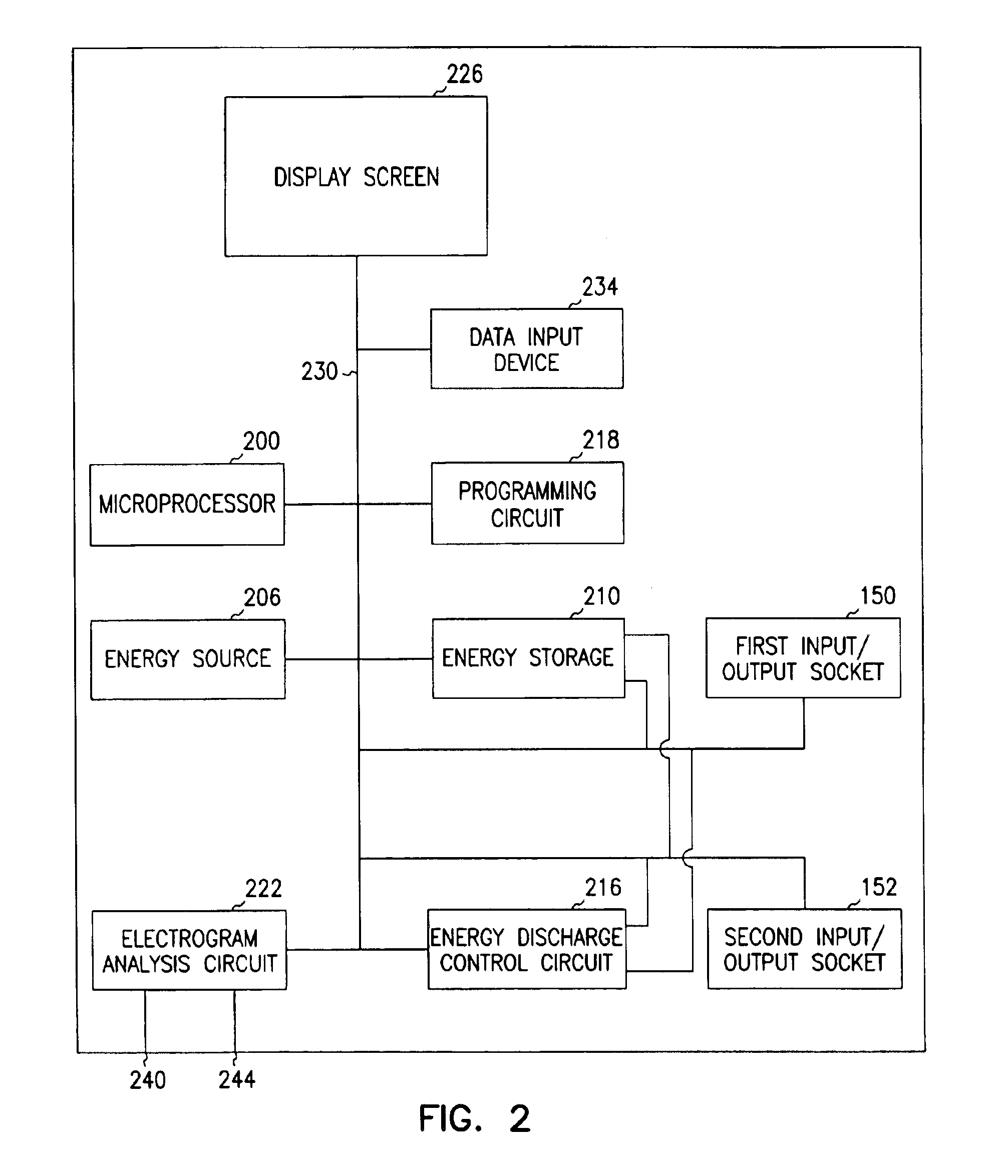
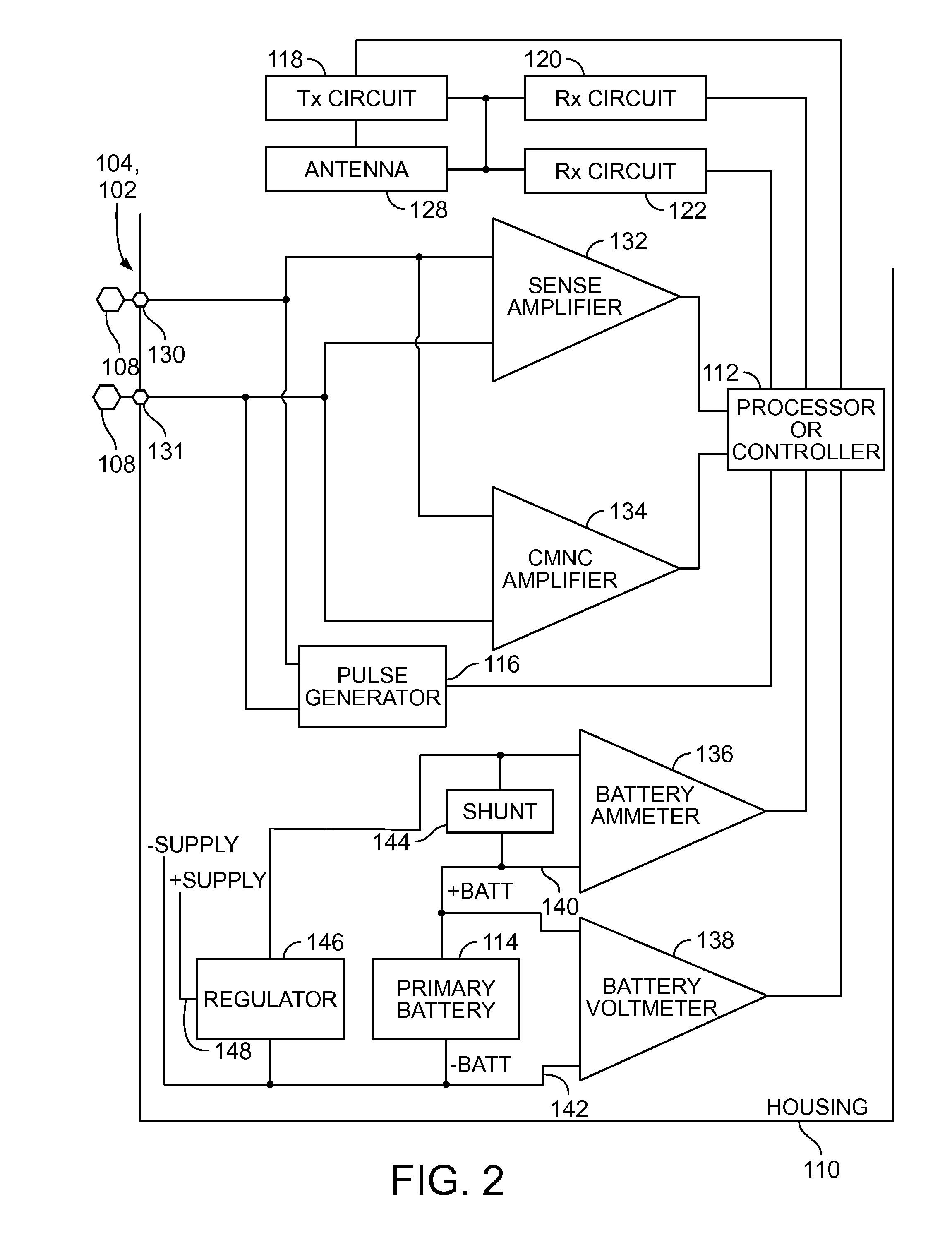
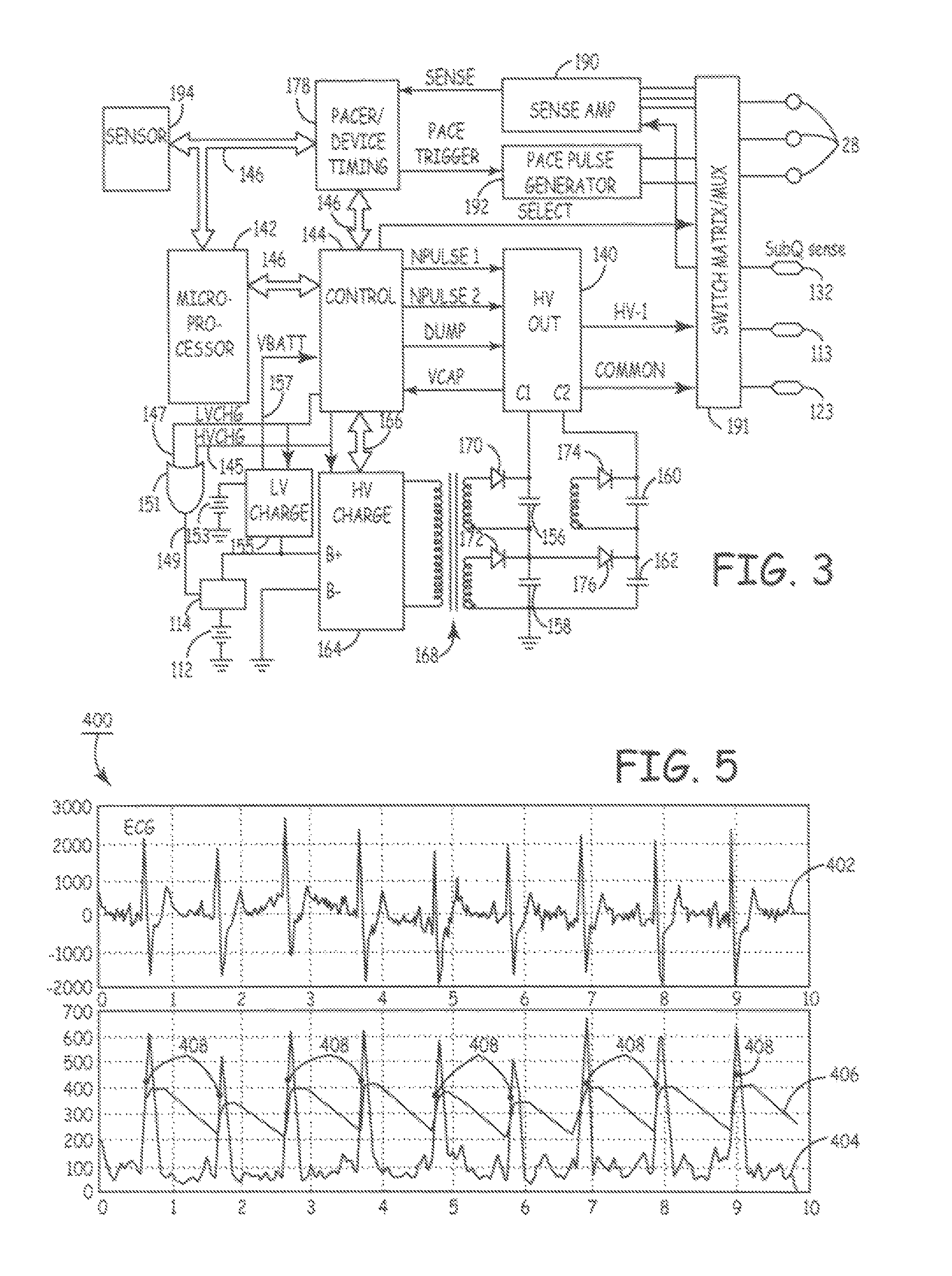
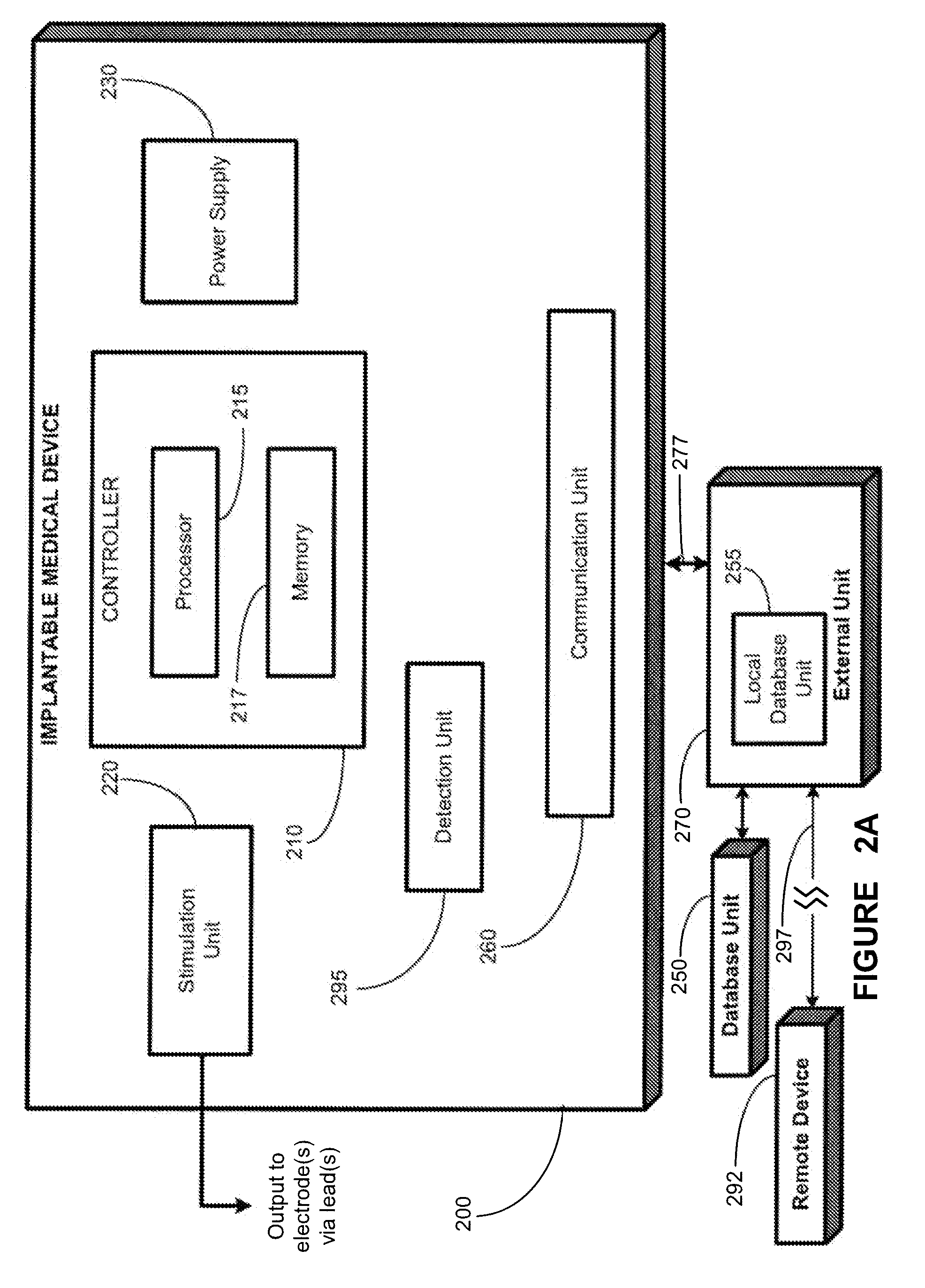
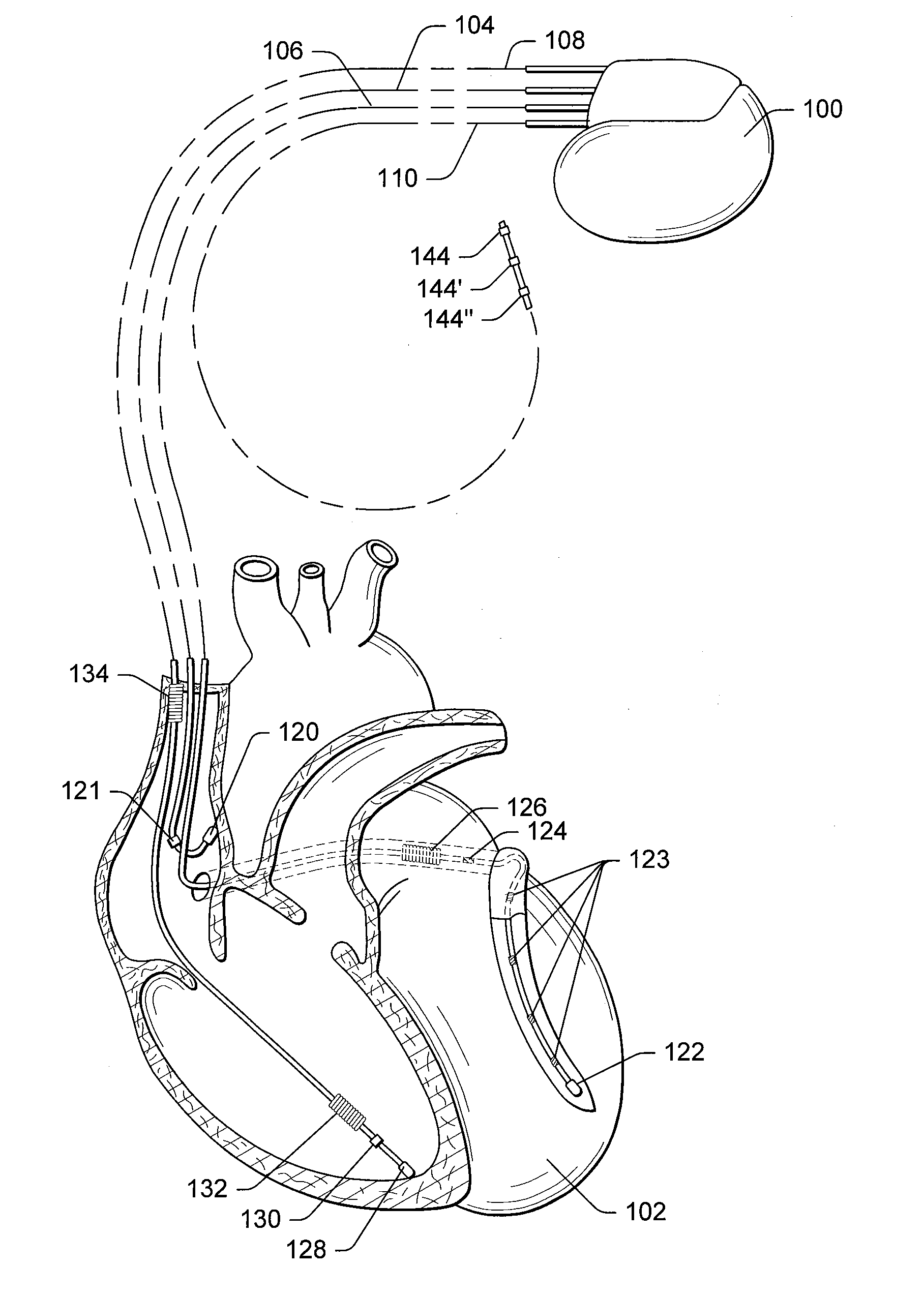
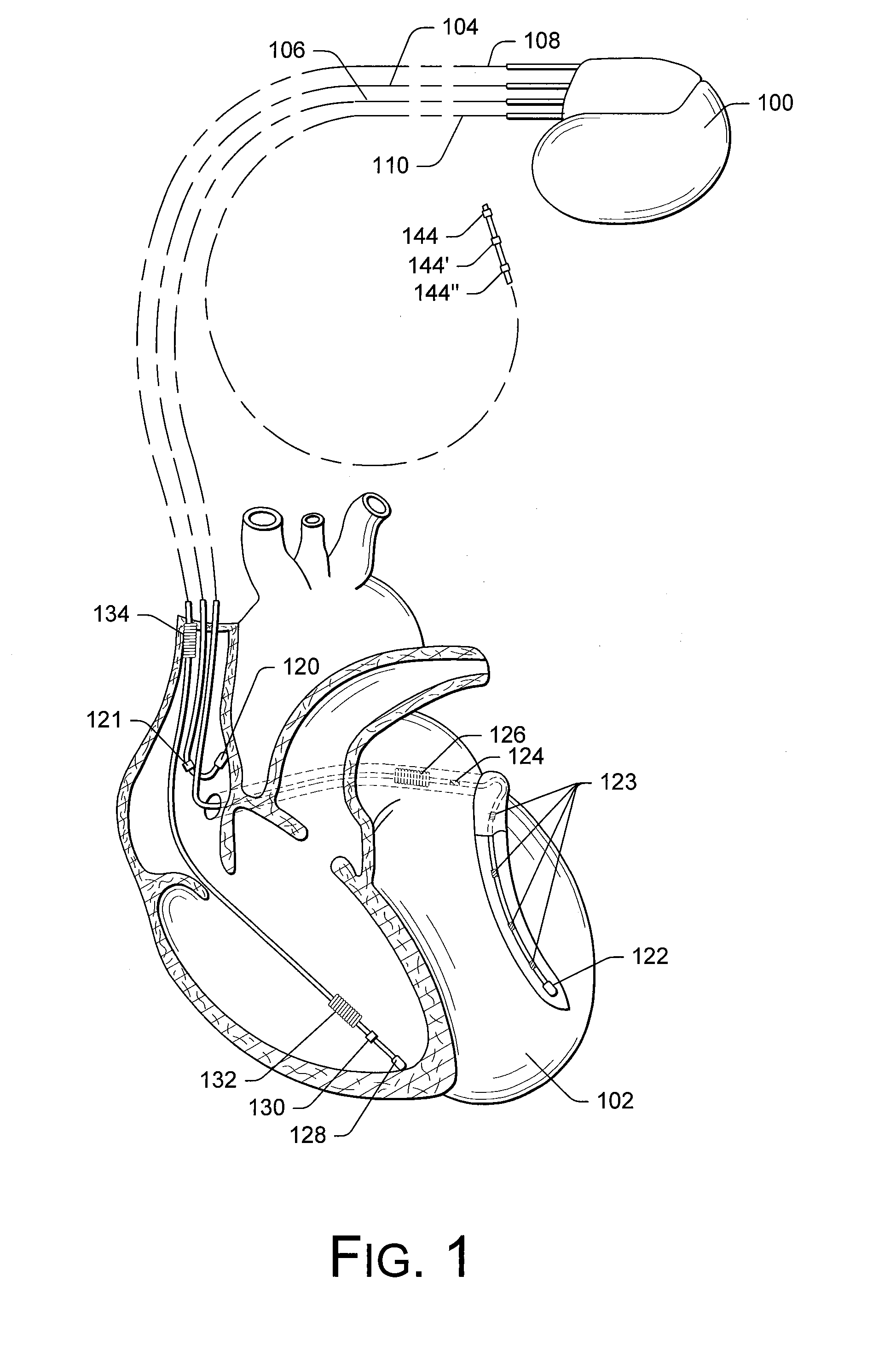
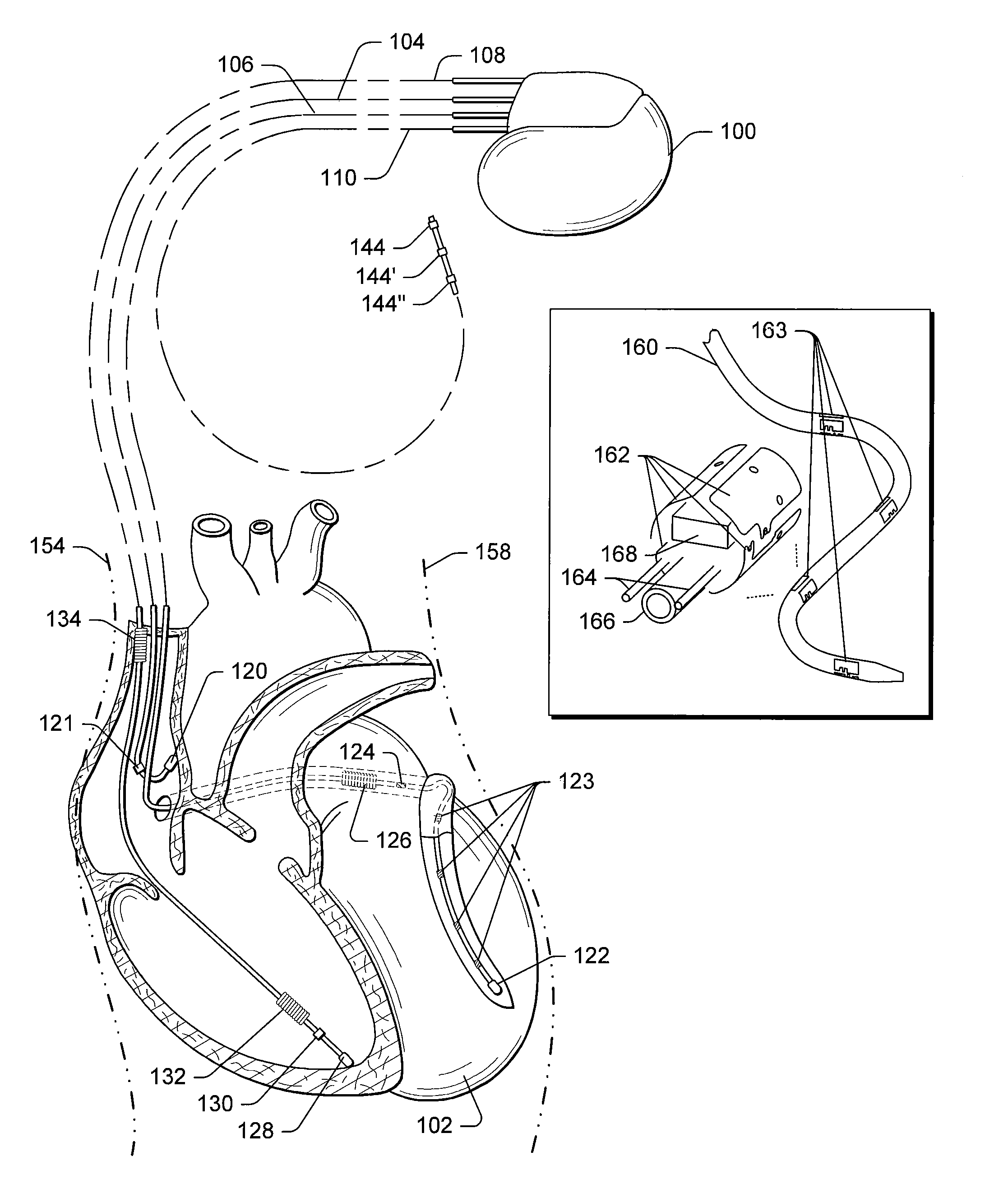
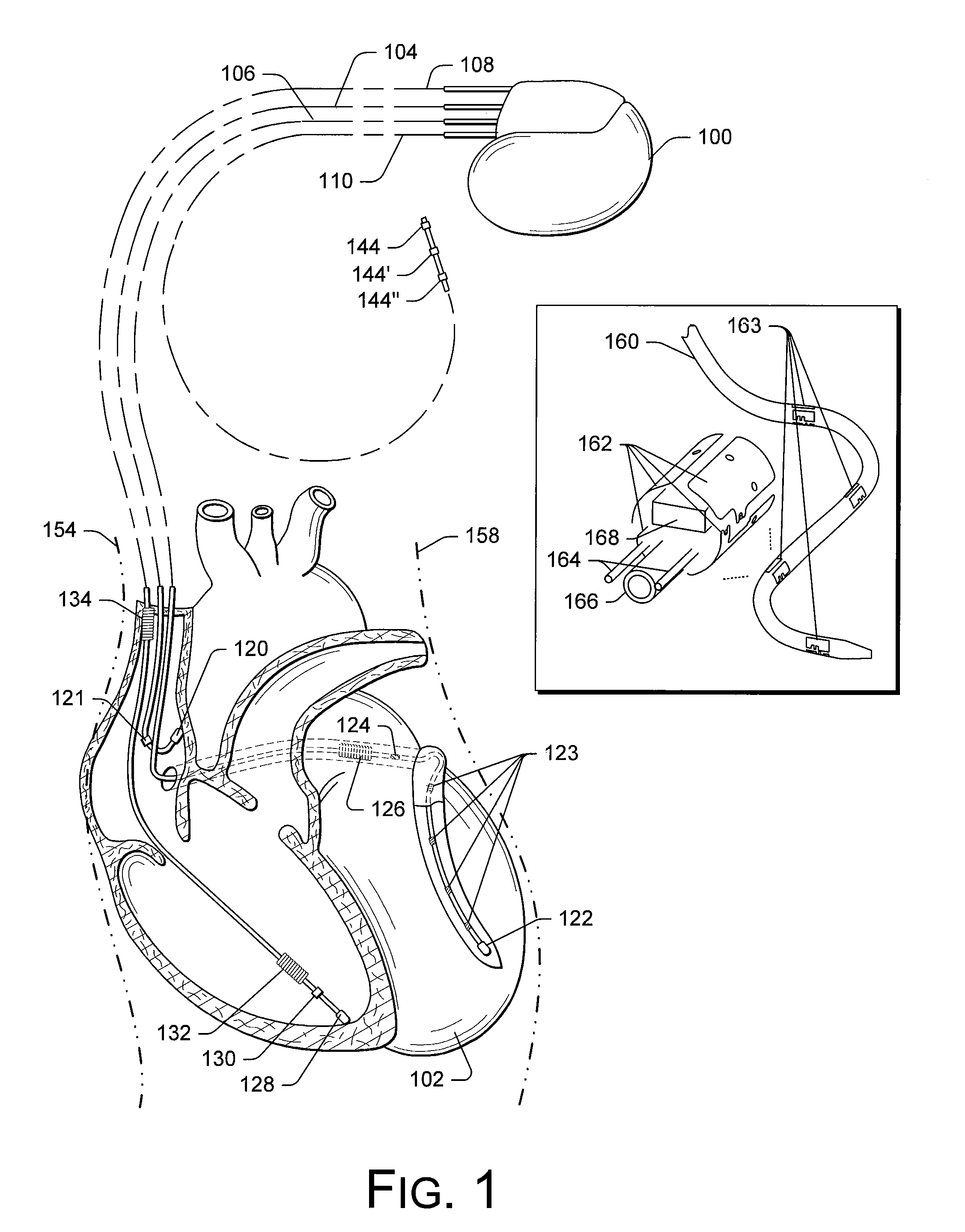
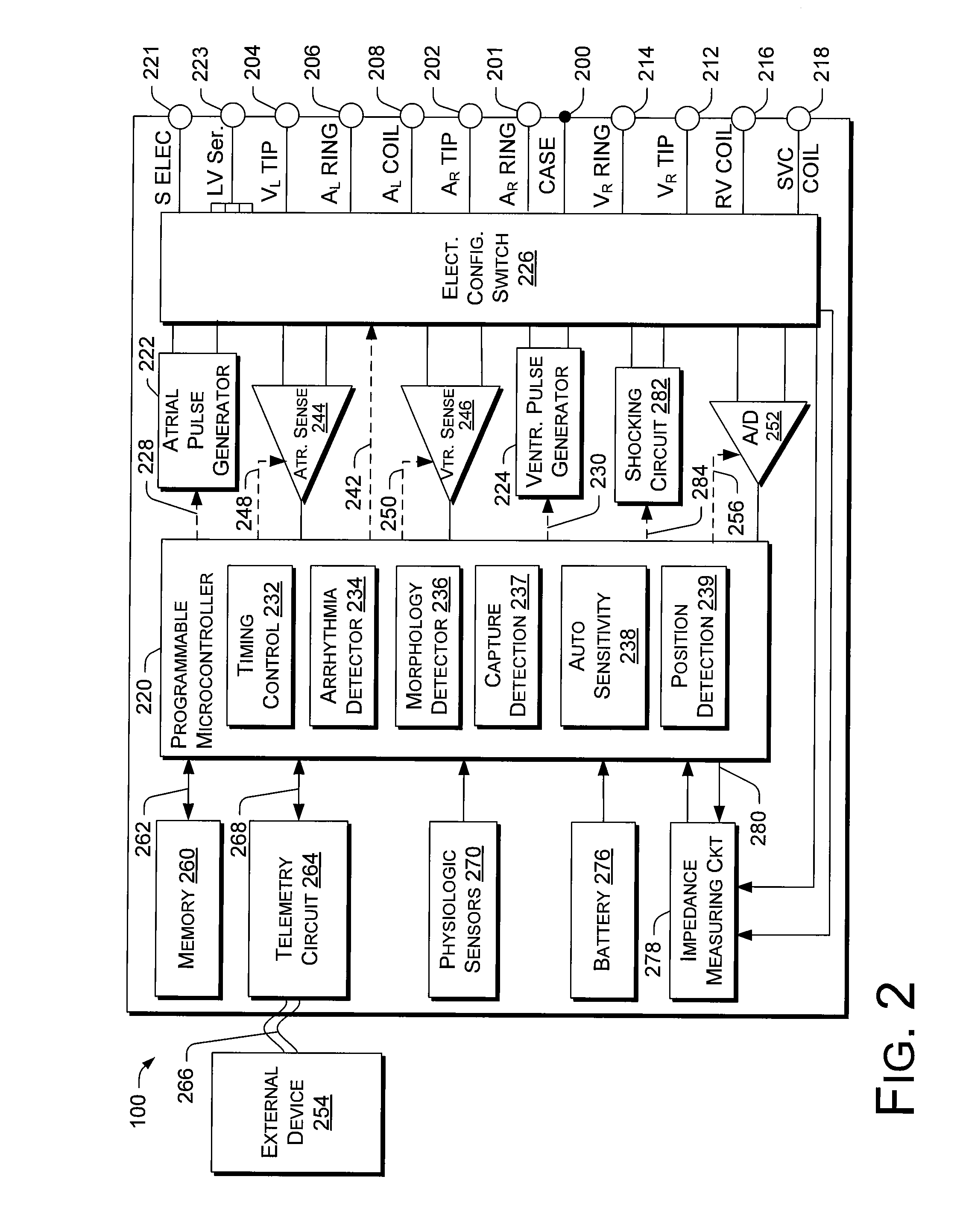
Implantable medical device for treating cardiac mechanical dysfunction by electrical stimulation
InactiveUS6738667B2Increase contractilityEasy to relaxCatheterHeart stimulatorsCardiac cycleHeart chamber
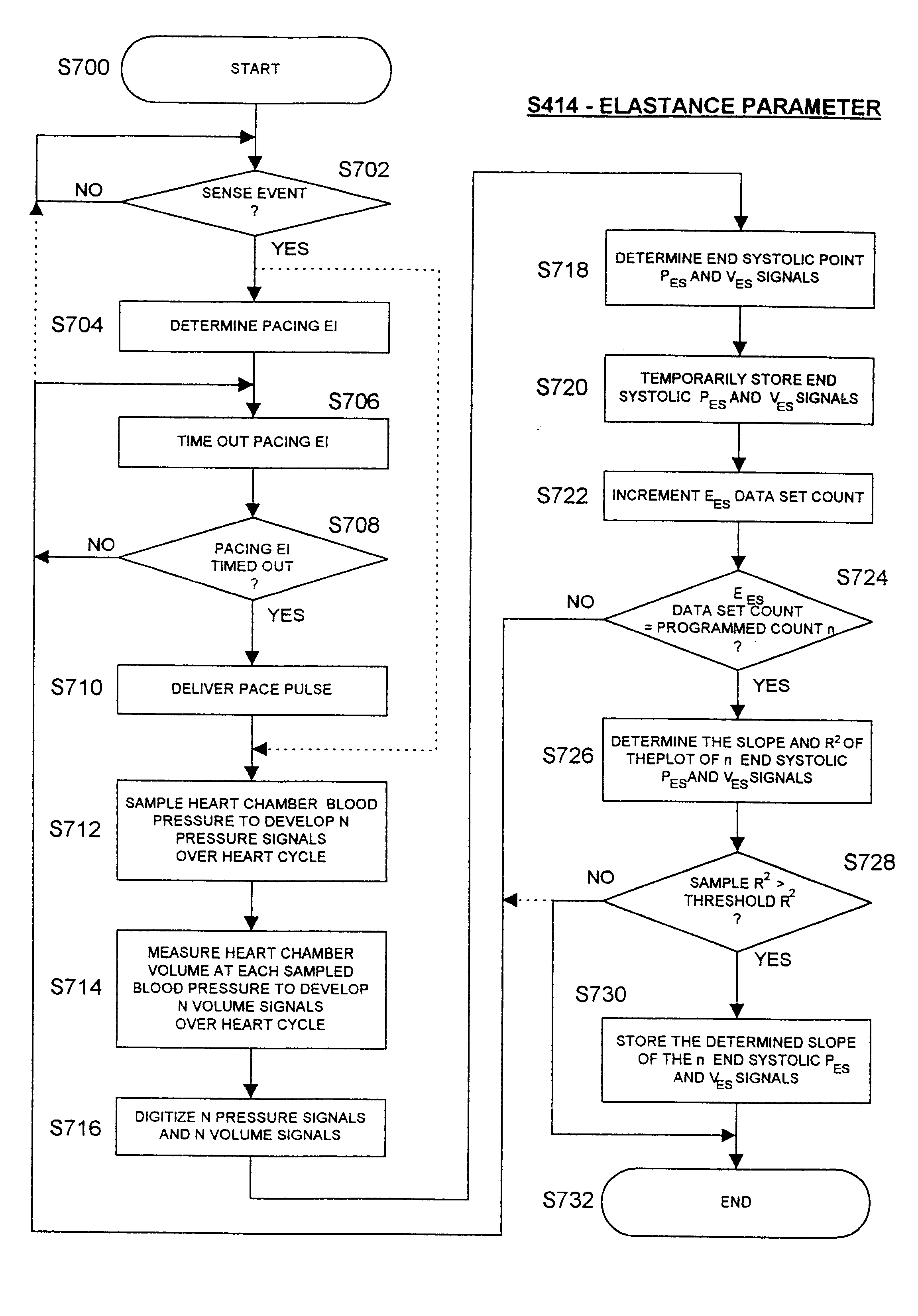
An implantable stimulator and monitor measures a group of heart failure parameters indicative of the state of heart failure employing EGM signals, measures of blood pressure including absolute pressure P, developed pressure (DP=systolic P-diastolic P), and / or dP / dt, and measures of heart chamber volume (V) over one or more cardiac cycles. These parameters include: (1) relaxation or contraction time constant tau (.tau.); (2) mechanical restitution (MR), i.e., the mechanical response of a heart chamber to premature stimuli applied to the heart chamber; (3) recirculation fraction (RF), i.e., the rate of decay of PESP effects over a series of heart cycles; and (4) end systolic elastance (E.sub.ES), i.e., the ratios of end systolic blood pressure P to volume V. These heart failure parameters are determined periodically regardless of patient posture and activity level. The physician can determine whether a particular therapy is appropriate, prescribe the therapy for a period of time while again accumulating the stored patient data for a later review and assessment to determine whether the applied therapy is beneficial or not, thereby enabling periodic changes in therapy, if appropriate. Drug therapies and electrical stimulation therapies, including PESP stimulation, and pacing therapies including single chamber, dual chamber and multi-chamber (bi-atrial and / or bi-ventricular) pacing can be delivered. In patient's prone to malignant tachyarrhythmias, the assessment of heart failure state can be taken into account in setting parameters of detection or classification of tachyarrhythmias and the therapies that are delivered.
Owner:MEDTRONIC INC
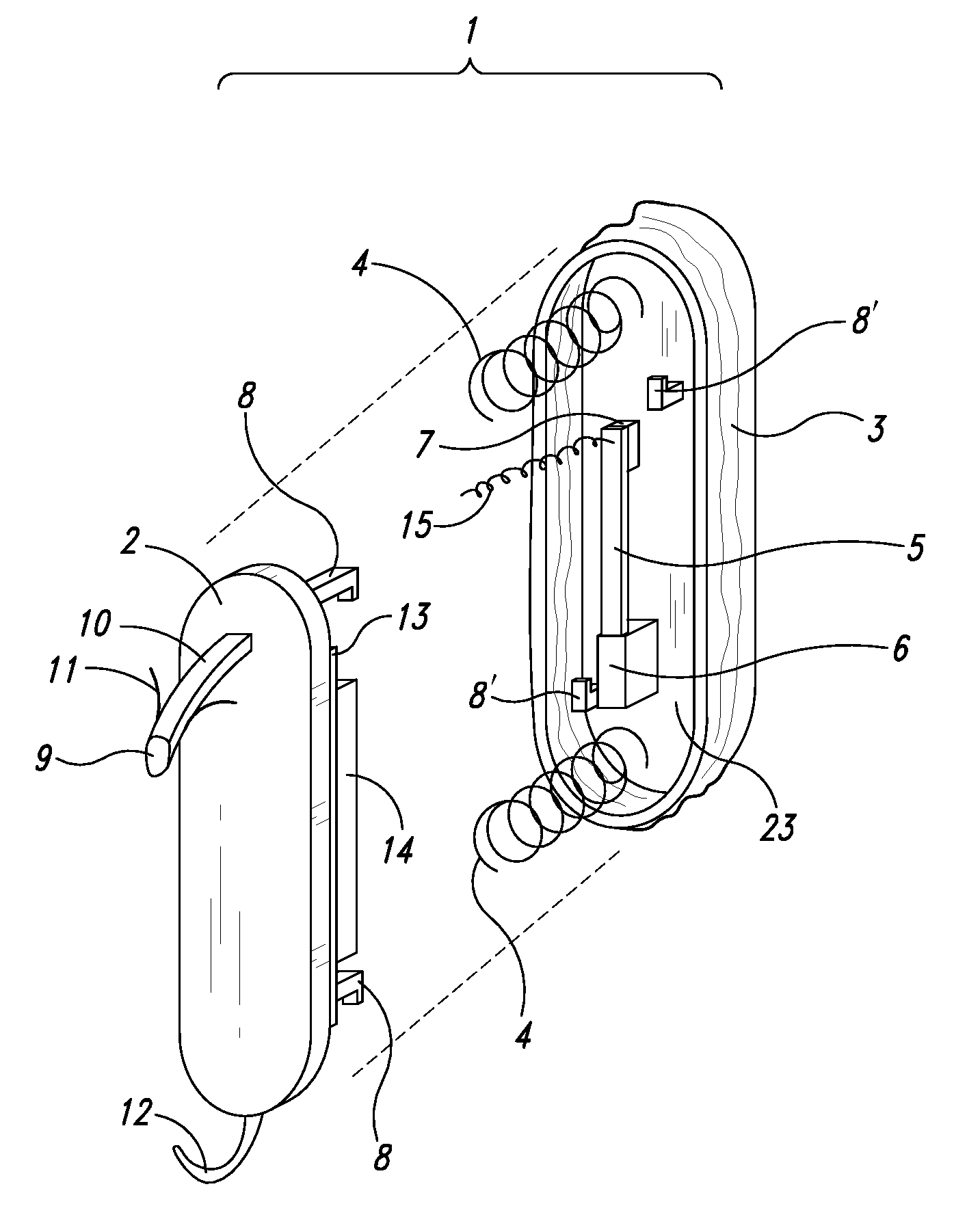
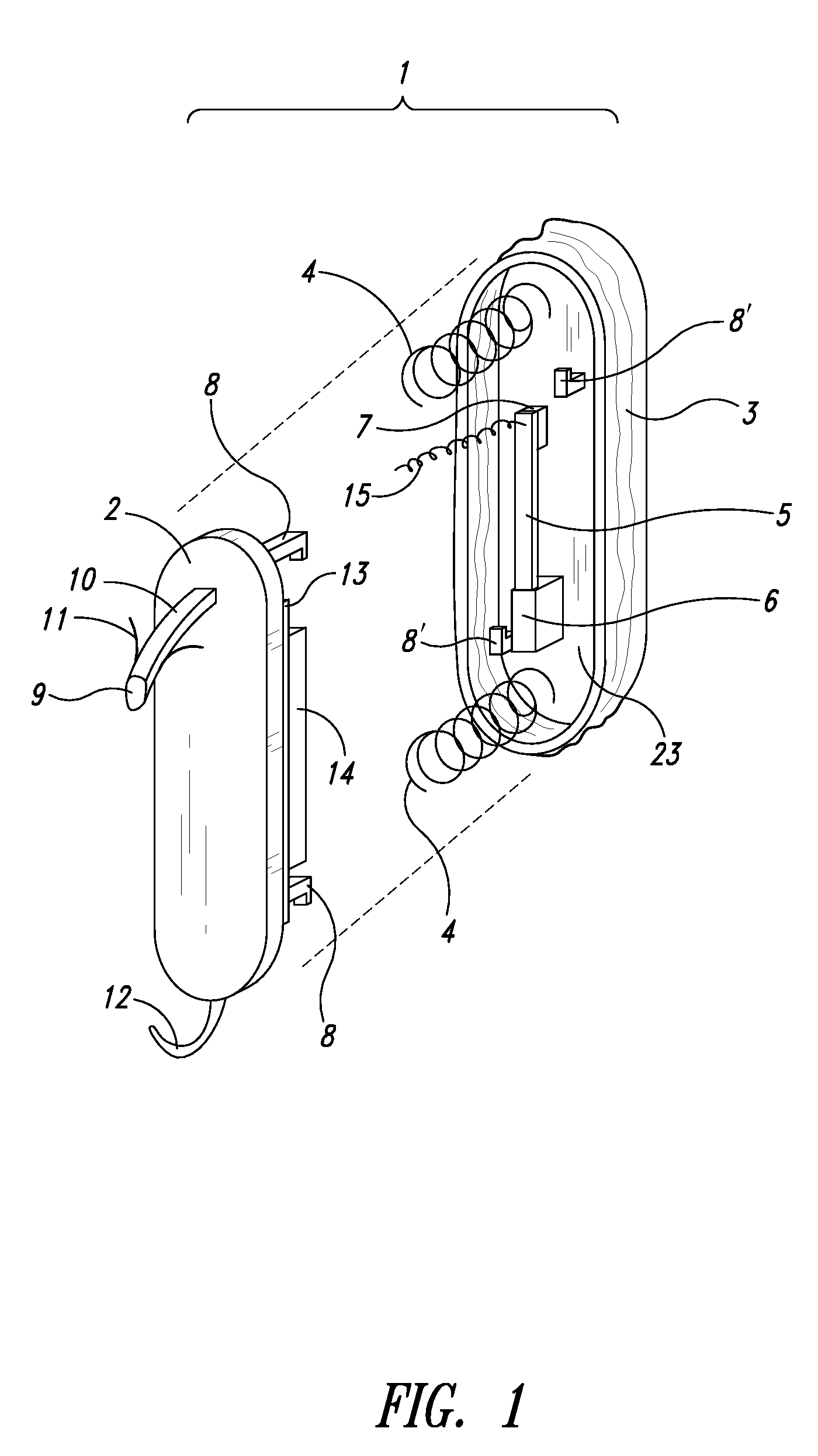
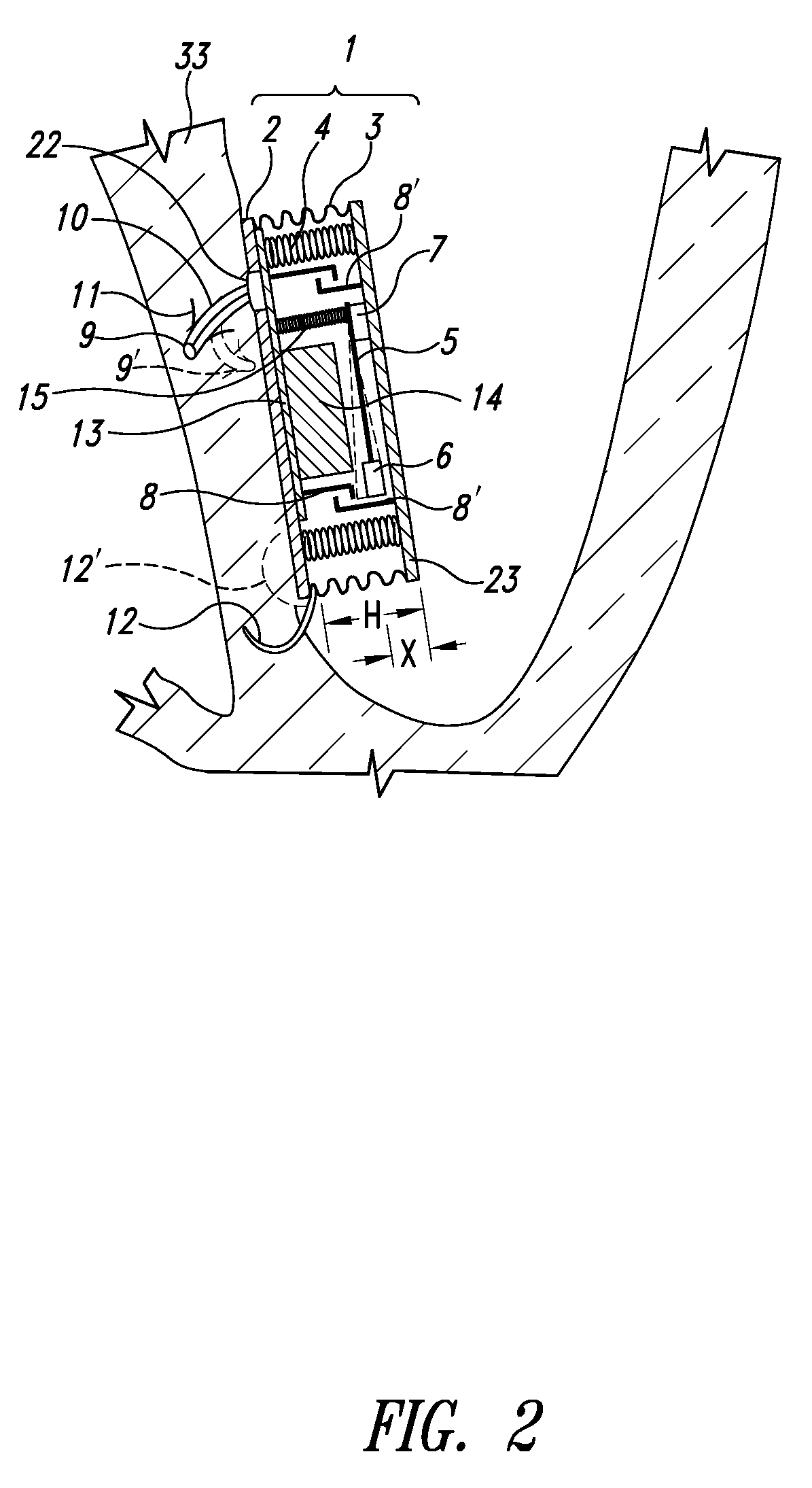
Self-powered resonant leadless pacemaker
InactiveUS20070293904A1Increases natural velocity and acceleration of heart muscleExtended durationElectrotherapyCardiac cycleCardiac pacemaker electrode
A self-powered medical device, for example a pacemaker uses the variations of blood pressure inside the heart or a major artery to create a mechanical resonance in an electromagnetic or piezoelectric generator. The resonance extends the time power is generated during the cardiac cycle. The pressure variations compress a bellows carrying the resonant generator. The inside of the bellows may be evacuated to a partial or full vacuum, and a spring restores the bellows to the desired equilibrium point, acting against the blood pressure. The current pulses are stored in a capacitor. Eliminating the battery allows dramatic miniaturization of the medical device to the point it can be implanted at the point of desired stimulation via a catheter.
Owner:LG RES PARTNERSHIP
Aortic annuloplasty ring
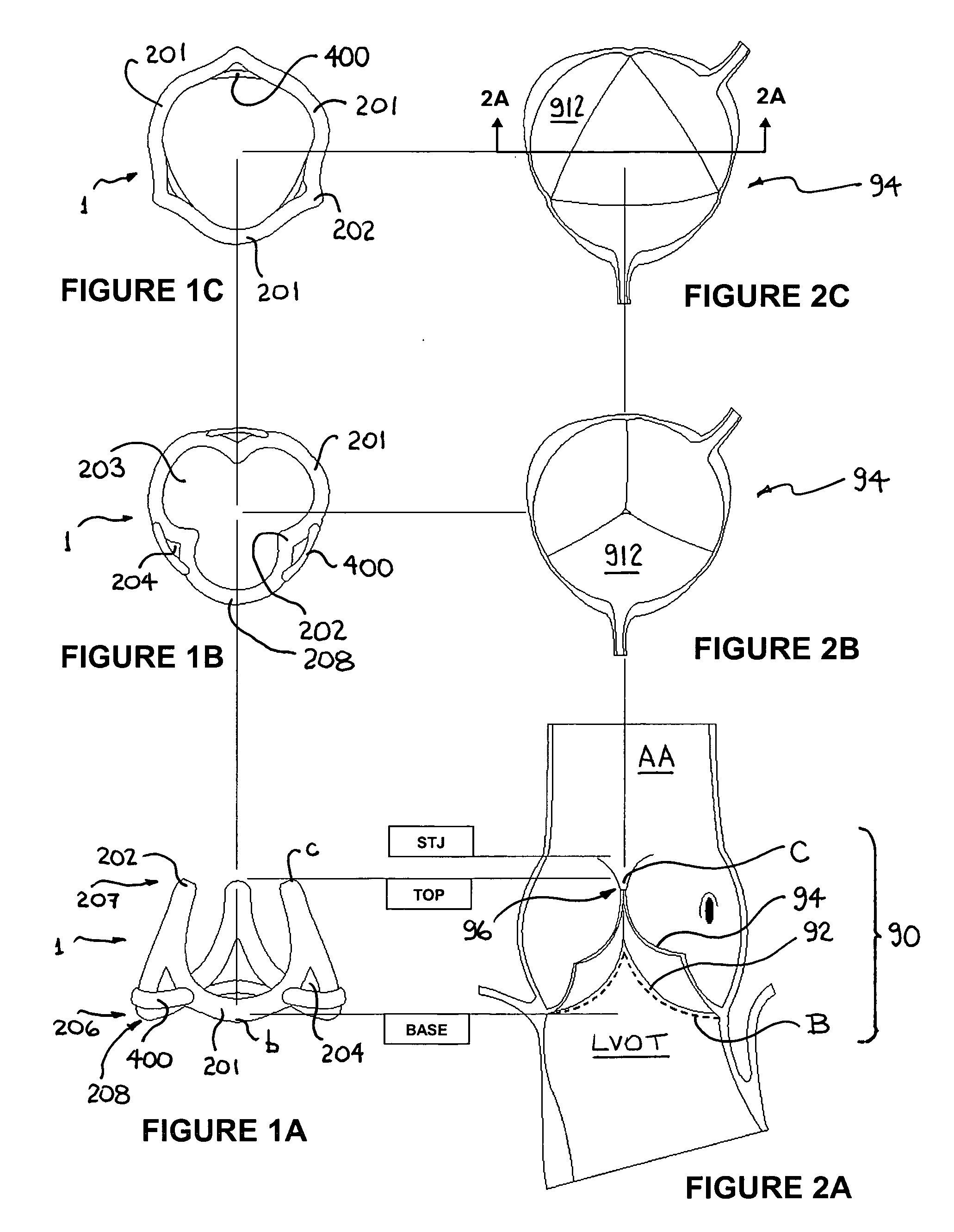
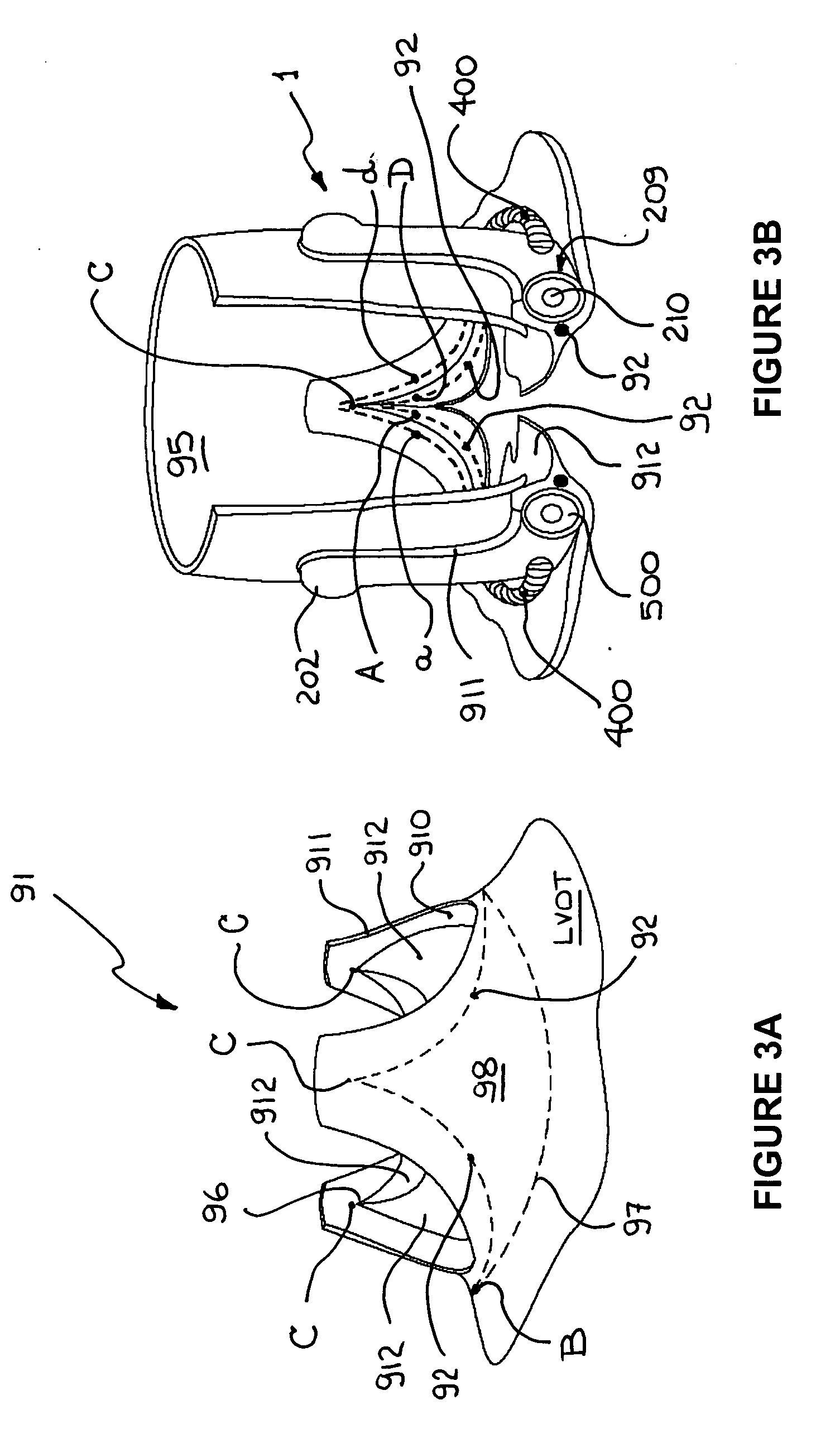
ActiveUS20060015179A1Preserve and restore normal aortic rootPreserve and restore and valve leafletAnnuloplasty ringsBlood vesselsCardiac cycleAnnuloplasty rings
An annuloplasty ring to resize a dilated aortic root during valve sparing surgery includes a scalloped space frame having three trough sections connected to define three crest sections. The annuloplasty ring is mounted outside the aortic root, and extends in height between a base plane and a spaced apart commissure plane of the aortic root. At least two adjacent trough sections are coupled by an annulus-restraining member or tether that limits the maximum deflection of the base of the annuloplasty ring. In use, the tether is preferably located in proximity to the base plane of the aortic root. The annuloplasty ring is movable between a first, substantially conical configuration occurring during a diastolic phase of the cardiac cycle, and a second, substantially cylindrical configuration occurring during a systolic phase of the cardiac cycle. The attachment of the annuloplasty ring in proximity to the cardiac valve annulus allows the ring to regulate the dimensions of a dynamic aortic root during the different phases of the cardiac cycle.
Owner:CORONEO
Devices and methods for heart valve treatment
Devices and methods for treating heart valves include members that assist the valve in closing during at least a portion of the cardiac cycle. Such devices include members configured to alter the shape of a valve annulus, reposition at least one papillary muscle, and / or plug an orifice of the valve so as to provide a coaptation surface for the valve leaflets.
Owner:EDWARDS LIFESCIENCES LLC
Method for reducing restenosis in the presence of an intravascular stent
InactiveUS6939345B2Reduce generationLot of damageDilatorsDiagnostic recording/measuringCoronary arteriesProximate
A first electrode is positioned within an artery proximate an implanted intravascular stent. A second electrode is positioned at a separate location relative the position of the first electrode. Electrical energy is then delivered between the first and the second electrodes to produce an electrical field adjacent the implanted intravascular stent. When a intravascular stent is implanted in a coronary artery, the delivery of the electrical energy is coordinated to cardiac cycles detected in sensed cardiac signals, where the delivery of the electrical energy between the first electrode and the second electrode occurs during a predetermined portion of the cardiac cycle.
Owner:CARDIAC PACEMAKERS INC
Cardiac activation sequence monitoring for ischemia detection
InactiveUS20060116593A1Convenient wireless communicationElectrocardiographyHeart stimulatorsImplantable ElectrodesCardiac cycle
Cardiac monitoring and / or stimulation methods and systems that provide one or more of monitoring, diagnosing, defibrillation, and pacing. Cardiac signal separation is employed to detect, monitor, track and / or trend ischemia using cardiac activation sequence information. Ischemia detection may involve sensing composite cardiac signals using implantable electrodes, and performing a signal separation that produces one or more cardiac activation signal vectors associated with one or more cardiac activation sequences. A change in the signal vector may be detected using subsequent separations. The change may be an elevation or depression of the ST segment of a cardiac cycle or other change indicative of myocardial ischemia, myocardial infarction, or other pathological change. The change may be used to predict, quantify, and / or qualify an event such as an arrhythmia, a myocardial infarction, or other pathologic change. Information associated with the vectors may be stored and used to track the vectors.
Owner:CARDIAC PACEMAKERS INC
Method and system for detecting and analyzing heart mecahnics
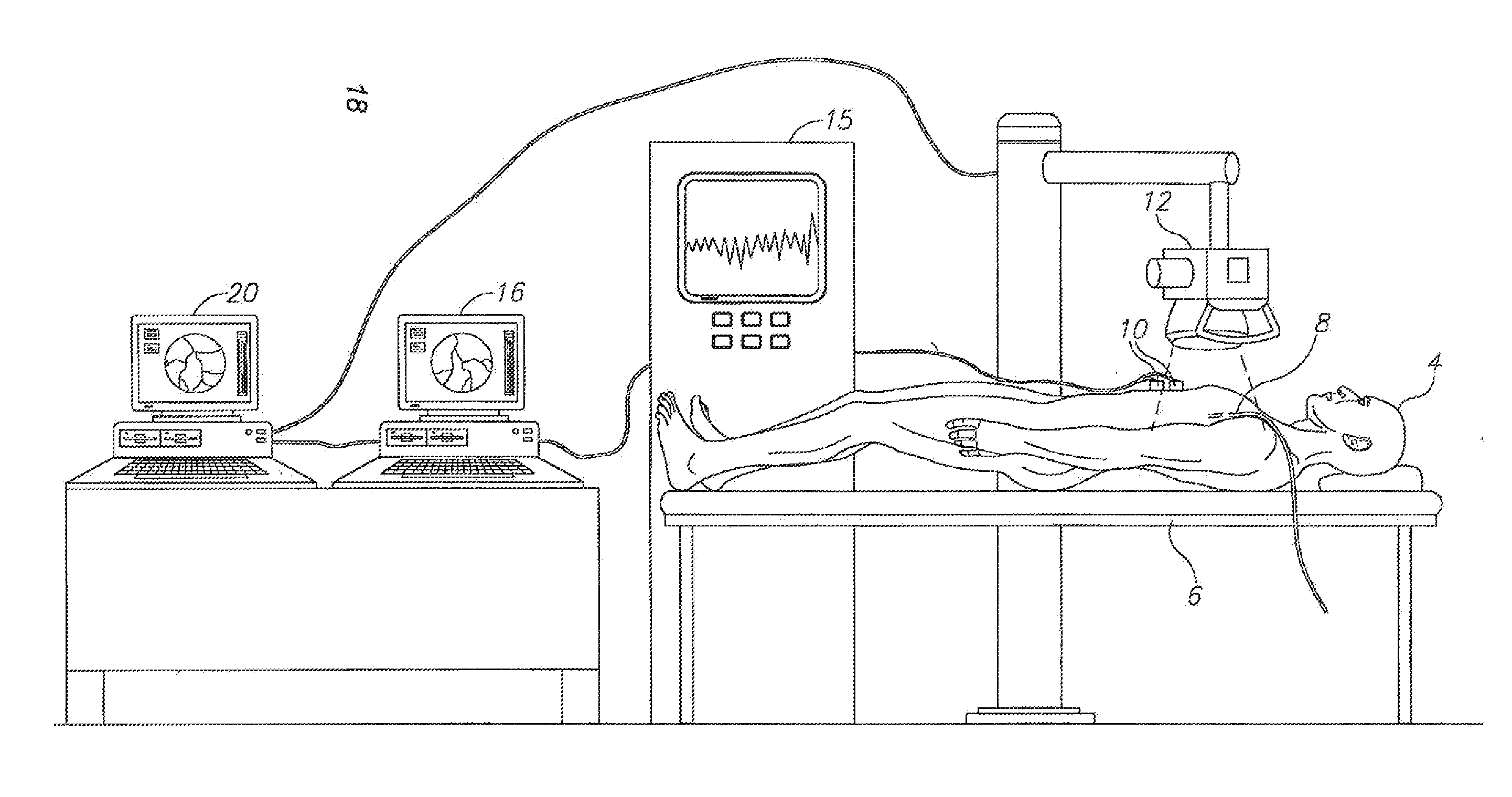
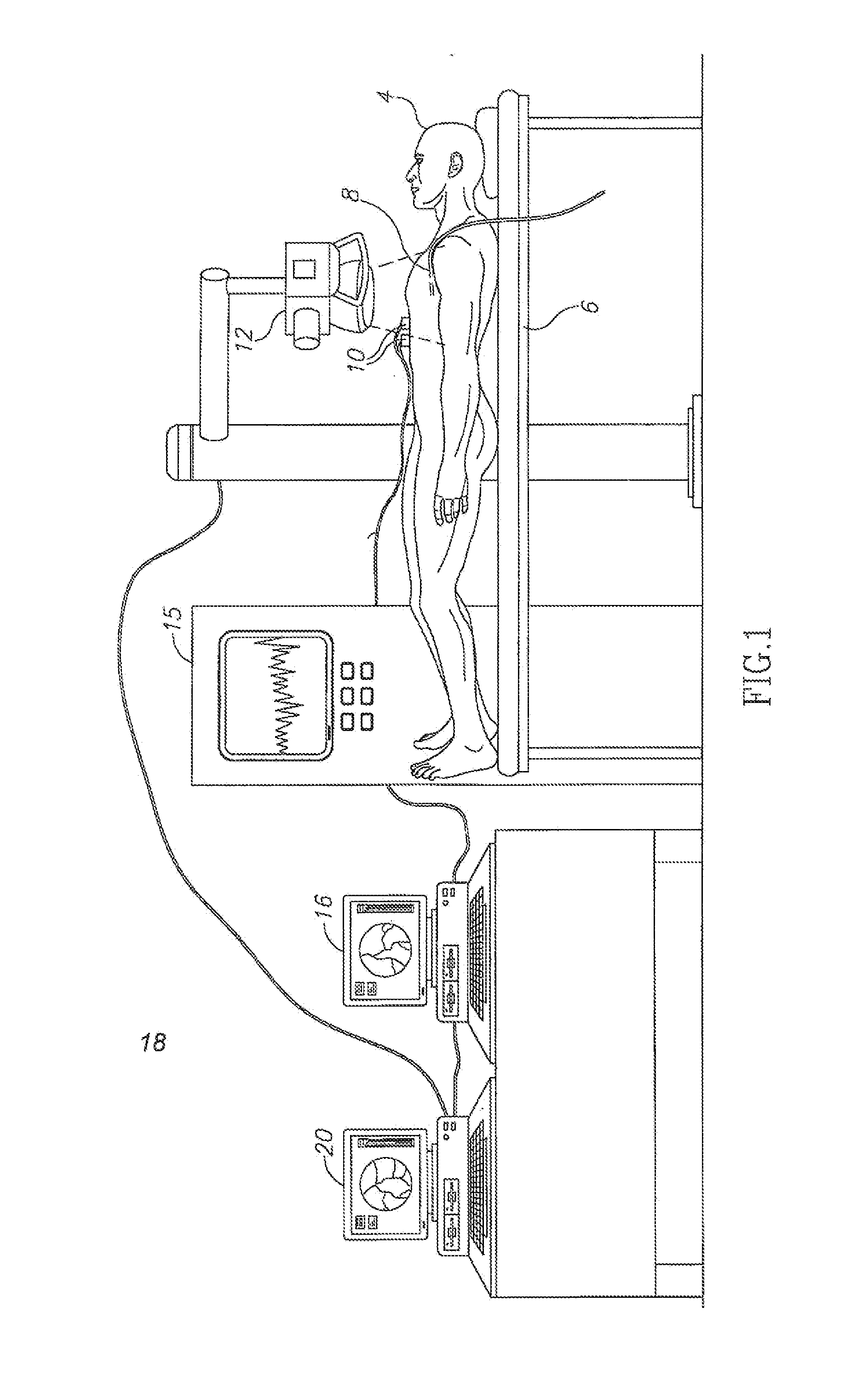
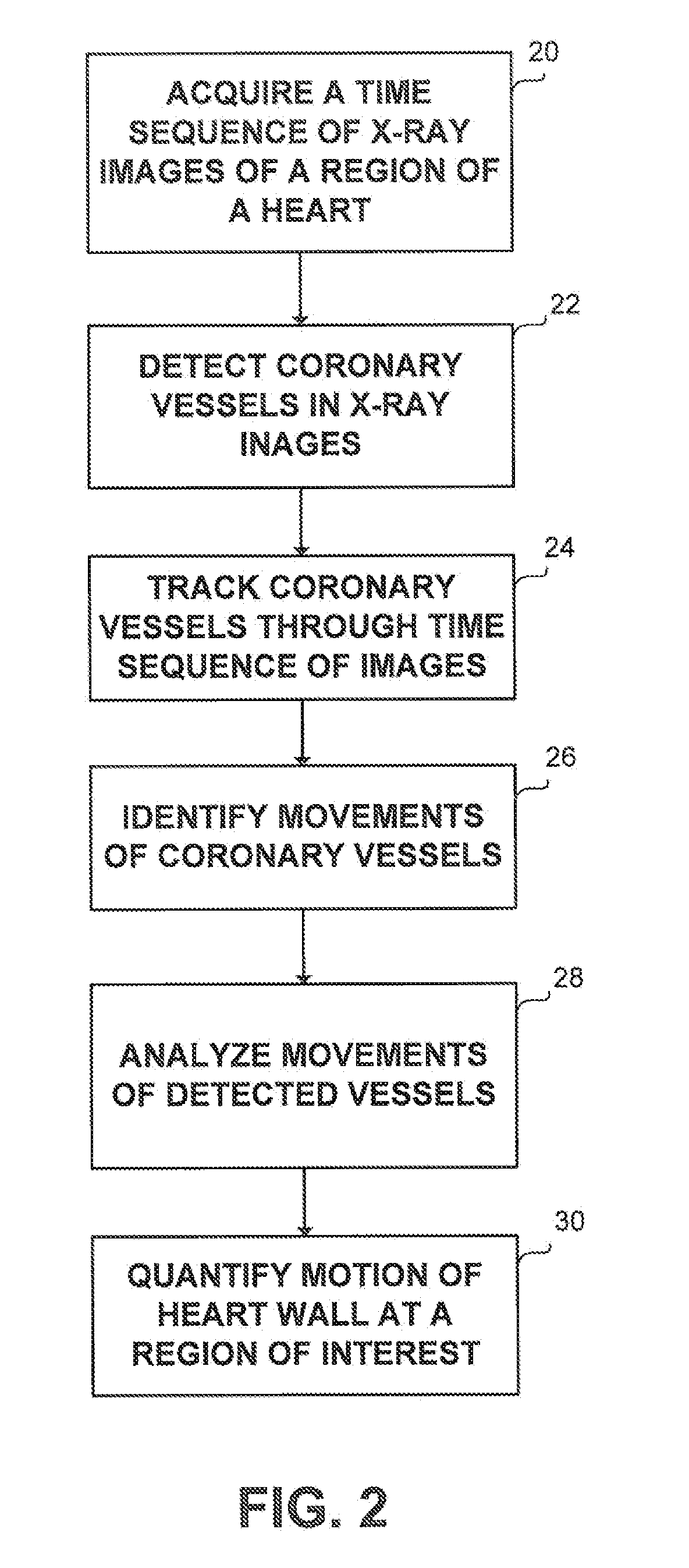
Method and apparatus for detecting and analyzing heart mechanical activity at a region of interest of a patient's heart are provided. The method comprises acquiring a time sequence of 2-dimensional X-ray images of a region of interest over at least part of a cardiac cycle; detecting coronary vessels in the X-ray images; tracking the coronary vessels through the sequence of images to identify movements of the coronary vessels; and analyzing the movements of the coronary vessels to quantify at least one parameter characterizing heart wall motion in the region of interest.
Owner:MEDTRONIC INC
Contraction status assessment
An implantable medical device receives at least one sensor signal representing inter-movement between a basal region of a heart ventricle and a ventricle apex during at least a portion of a systolic phase of a cardiac cycle. A parameter processor calculates a contraction status parameter value based on the at least one sensor signal. This contraction status parameter value represents an elongation of the ventricle following onset of ventricular activation during a cardiac cycle. The contraction status parameter value is stored in a memory as a diagnostic parameter representing a current contraction status of a subject's heart.
Owner:ST JUDE MEDICAL
Stress reduction apparatus and method
InactiveUS6908424B2Reduce maximum wall stress experienceRelieve pressureSuture equipmentsHeart valvesCardiac cycleStress reduction
The device and method for reducing heart wall stress. The device can be one which reduces wall stress throughout the cardiac cycle or only a portion of the cardiac cycle. The device can be configured to begin to engage, to reduce wall stress during diastolic filling, or begin to engage to reduce wall stress during systolic contraction. Furthermore, the device can be configured to include at least two elements, one of which engages full cycle and the other which engages only during a portion of the cardiac cycle.
Owner:EDWARDS LIFESCIENCES LLC
Intracardiac device for restoring the functional elasticity of the cardiac structures, holding tool for the intracardiac device, and method for implantation of the intracardiac device in the heart
ActiveUS20100030014A1Minimize tissutal reactionReduce thrombogenic capacityAnnuloplasty ringsBandagesCardiac cyclePlasma viscosity
An intracardiac device for restoring the functional elasticity of the cardiac structures, in particular for the treatment of cardiomyopathies and or valvulopathies, by storing energy from the cardiac structures and ceding energy to the cardiac structures during the cardiac cycle, has an elongated shape, is at least partially wound in coils and is attachable to a cardiac structure; the coils are selected in material, number and dimension so as to allow an elastic elongation of the intracardiac device higher than 10% of the rest length of the intracardiac device and are exposed, in use, to the blood flow.
Owner:CUBE
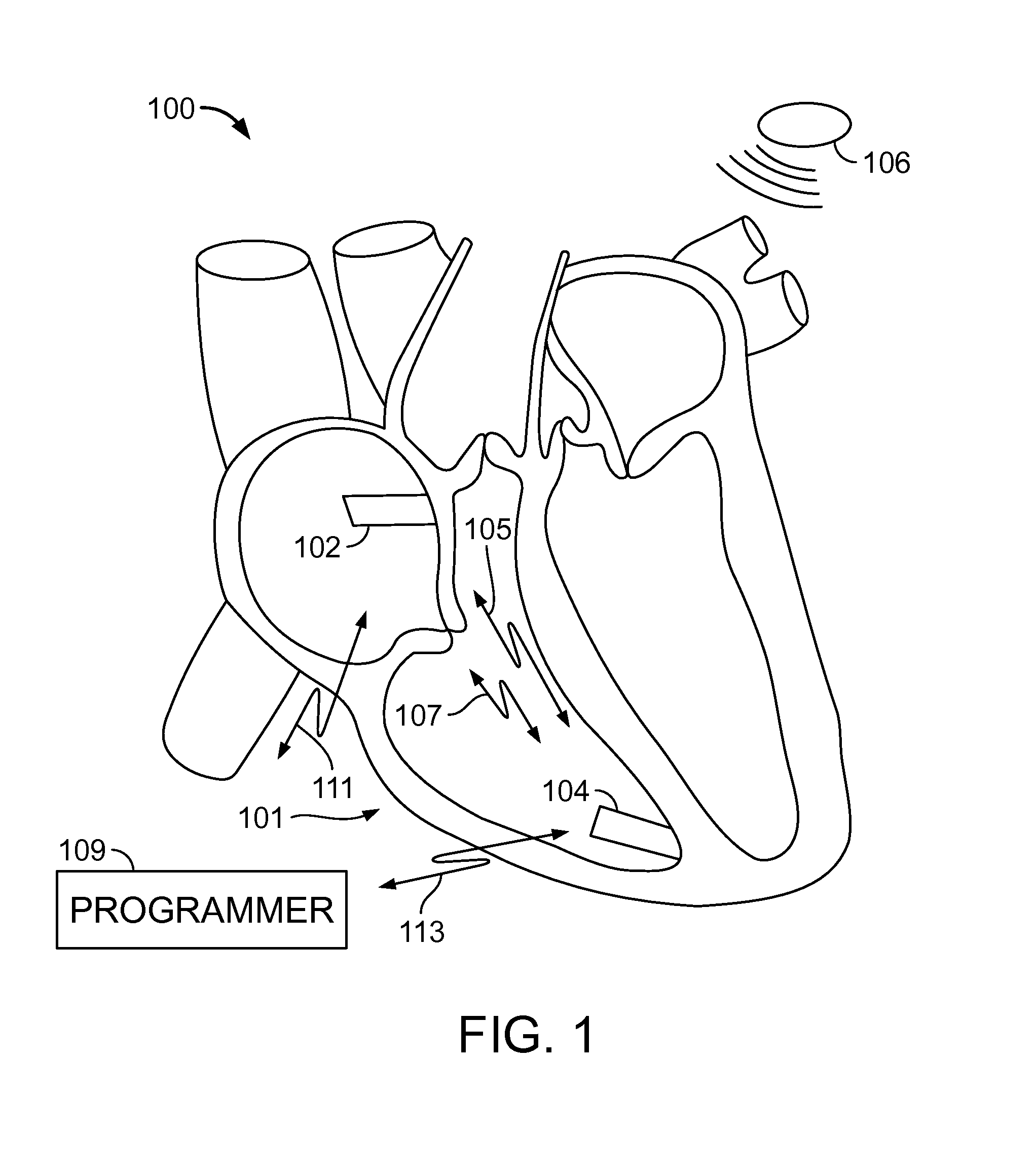
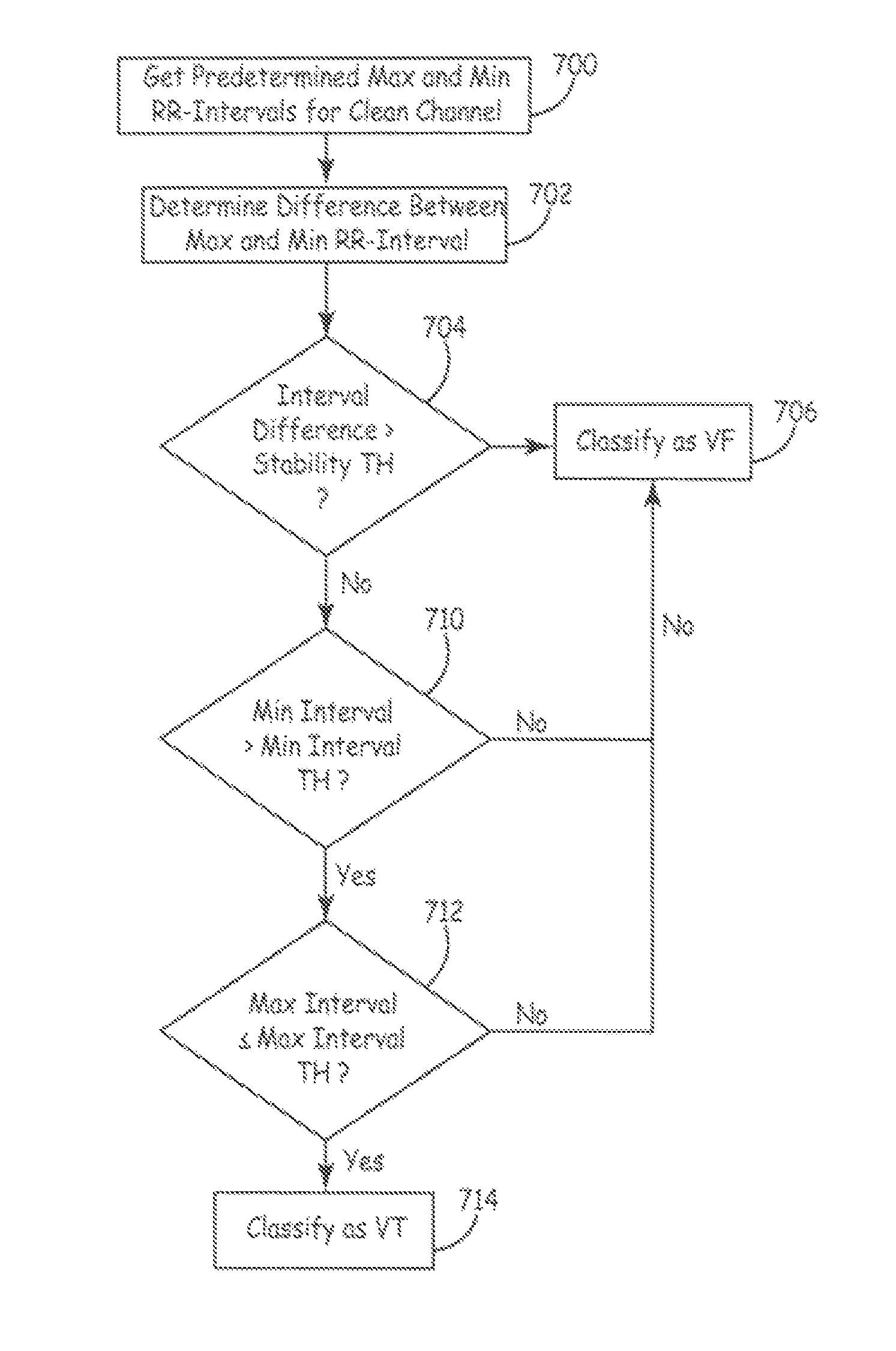
Classification of supraventricular and ventricular cardiac rhythms using cross channel timing algorithm
InactiveUS6889081B2ElectrocardiographyMedical automated diagnosisCardiac cycleVentricular tachycardia
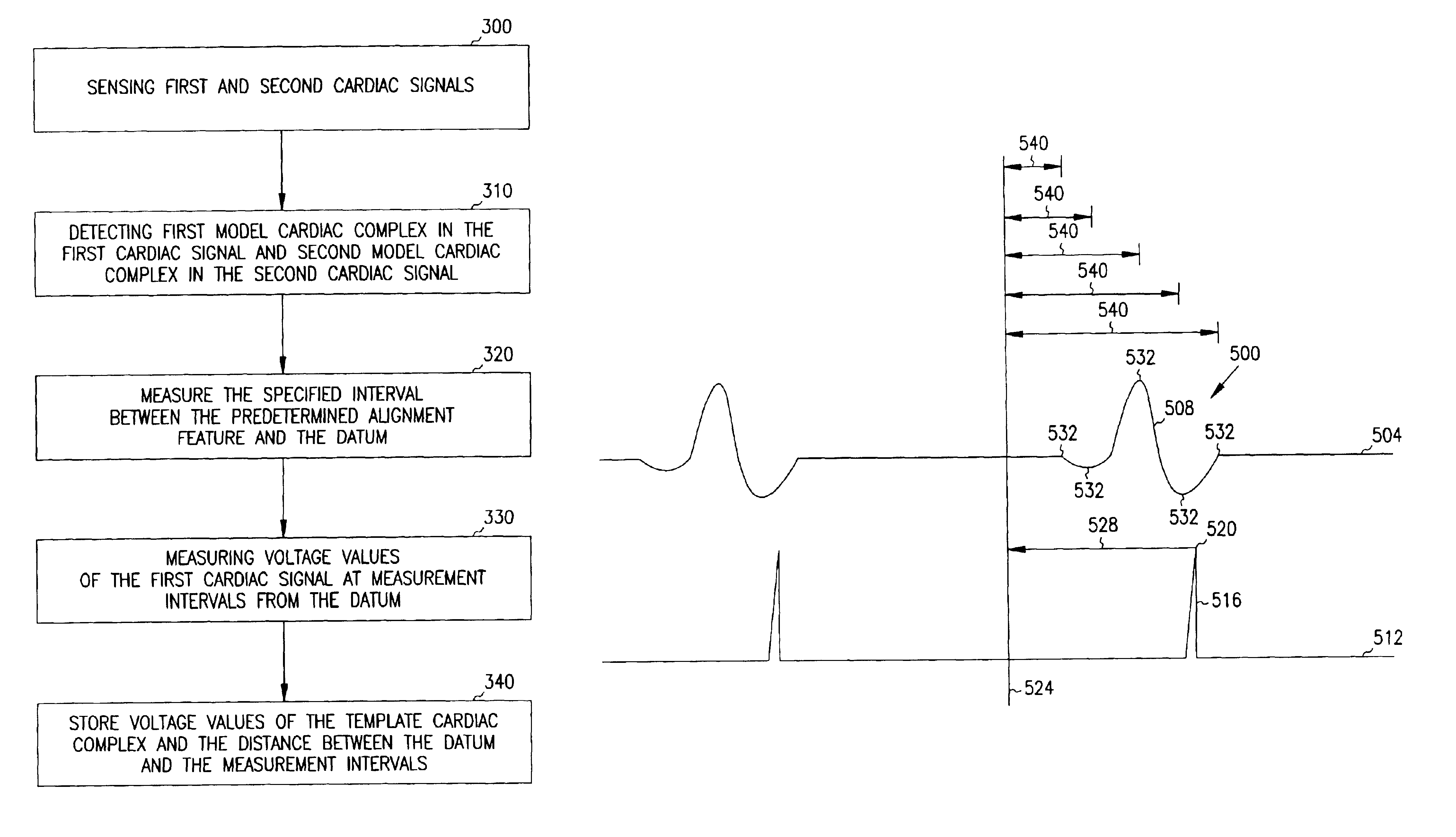
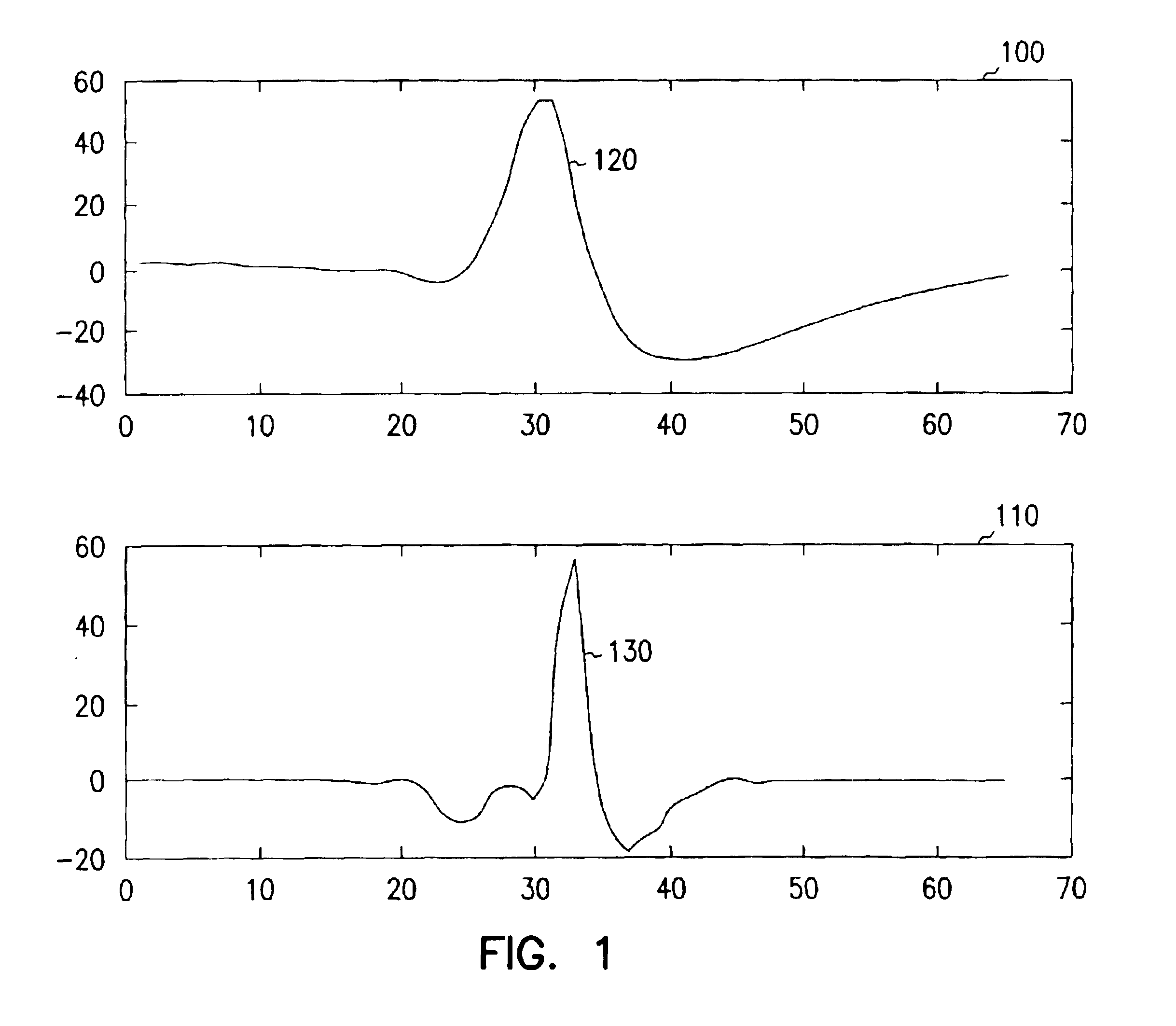
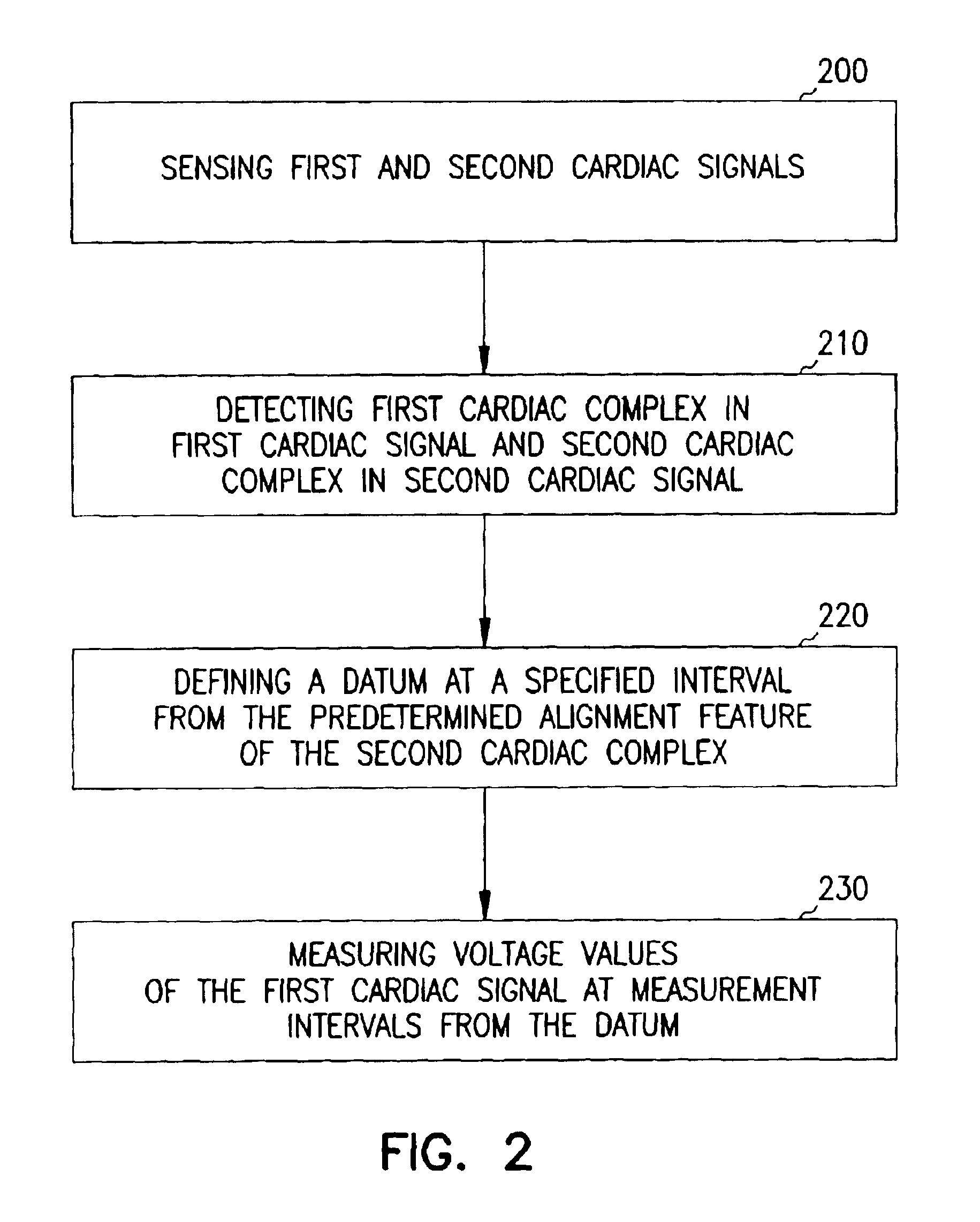
A system and method for classifying cardiac complexes sensed during a tachycardia episode. A first cardiac signal and a second cardiac signal are sensed, where the first cardiac signal has a voltage. A first cardiac complex and a second cardiac complex of a cardiac cycle are detected in the first and second cardiac signal, respectively. A predetermined alignment feature is identified in the second cardiac complex. A datum is defined, or positioned, at a specified interval from the predetermined alignment feature of the second cardiac complex. Voltage values are then measured from the first cardiac complex at each of two or more measurement intervals from the datum. The voltage values are then compared voltage values measured from NSR cardiac complexes to classify the first cardiac complex is either a ventricular tachycardia complex or a supraventricular tachycardiac complex.
Owner:CARDIAC PACEMAKERS INC
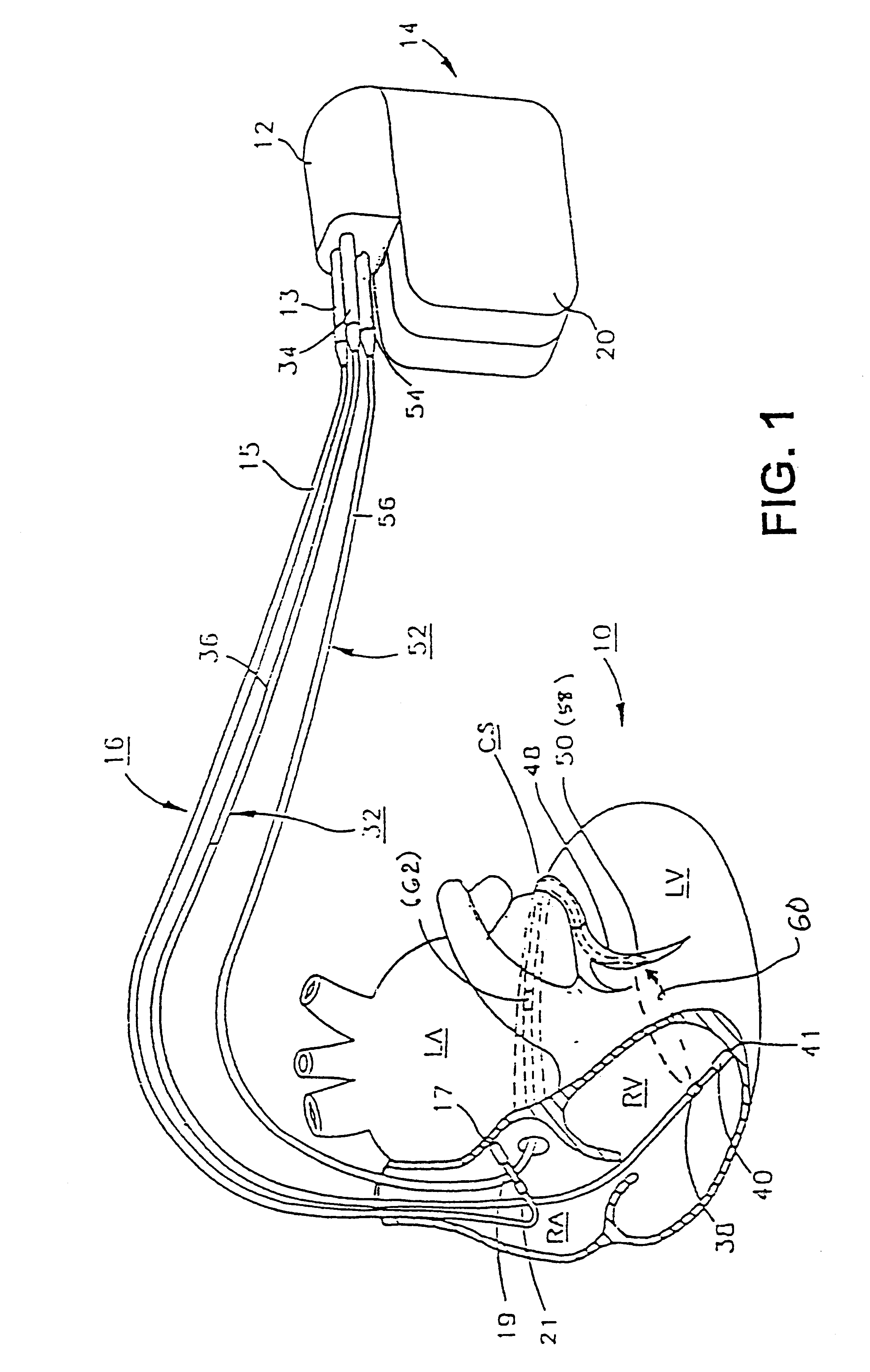
Leadless dual-chamber pacing system and method
In accordance with an embodiment, a system and method is provided for providing communications between first and second implantable medical devices (IMDs). The system comprises a first implantable medical device (IMD) configured to transmit a first event message during or preceding a first cardiac cycle, and a second IMD configured to receive the first event message, wherein receipt of the first event message configures the second IMD to generate pacing pulses for only a predetermined number (“n”) of consecutive cardiac cycles, wherein n is an integer equal to or greater than 2. The method comprises transmitting a first event message during or preceding a first cardiac cycle from the first IMD, receiving the first event message at the second IMD, in response to receiving the first event message, arming the second IMD to generate a pacing pulse for a second cardiac cycle, and arming the second IMD to generate a pacing pulse for only a predetermined number of consecutive cardiac cycles immediately subsequent to the second cardiac cycle.
Owner:PACESETTER INC
Method and apparatus for detecting arrhythmias in a subcutaneous medical device
A method and apparatus for detecting a cardiac event in a medical device that includes sensing cardiac signals from a plurality of electrodes forming a first sensing vector and a second sensing vector, advancing from a first state to a second state in response to processing of an interval associated with a cardiac cycle of the sensed cardiac signals, and advancing from the second state in response to processing of a cardiac signal sensed during a predetermined sensing window.
Owner:MEDTRONIC INC
In-vivo method and device for improving diastolic function of the left ventricle
InactiveUS7186210B2Function increaseReduce hydrostatic pressureSuture equipmentsHeart valvesLeft ventricular sizeCardiac cycle
A method and device featuring at least one component providing a potential to kinetic converted elastic, magnetic repulsion, or, an elastic and magnetic repulsion, pushing, pulling, or, pulling and pushing, type of radially outward expansive force or pressure to an inner, outer, intermediate, and, combination thereof, wall region of the left ventricle, for reducing intraluminal hydrostatic pressure of the left ventricle (LV filling pressure) during the ventricular diastolic stage of the cardiac cycle, thereby, improving diastolic function of the left ventricle of the heart in subjects having a condition of diastolic heart failure (DHF), while minimally disturbing systolic function of the heart. The expansive force or pressure is in a range of about 5–40 mm Hg, whereby, left ventricular end diastolic pressure (LVEDP) is reduced down to the normal range of about 6–12 mm Hg, during ventricular diastole of the heart.
Owner:RELAXIS
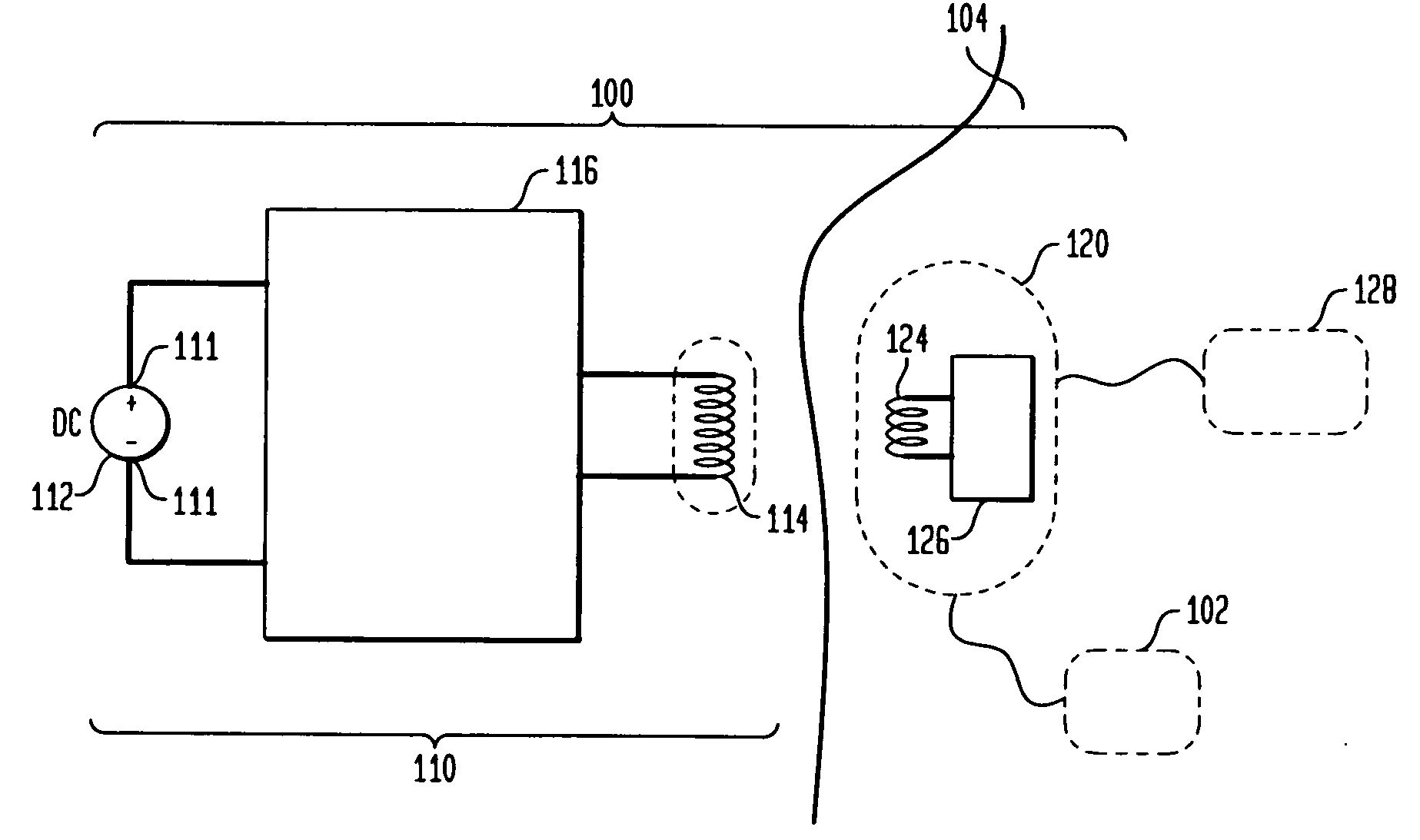
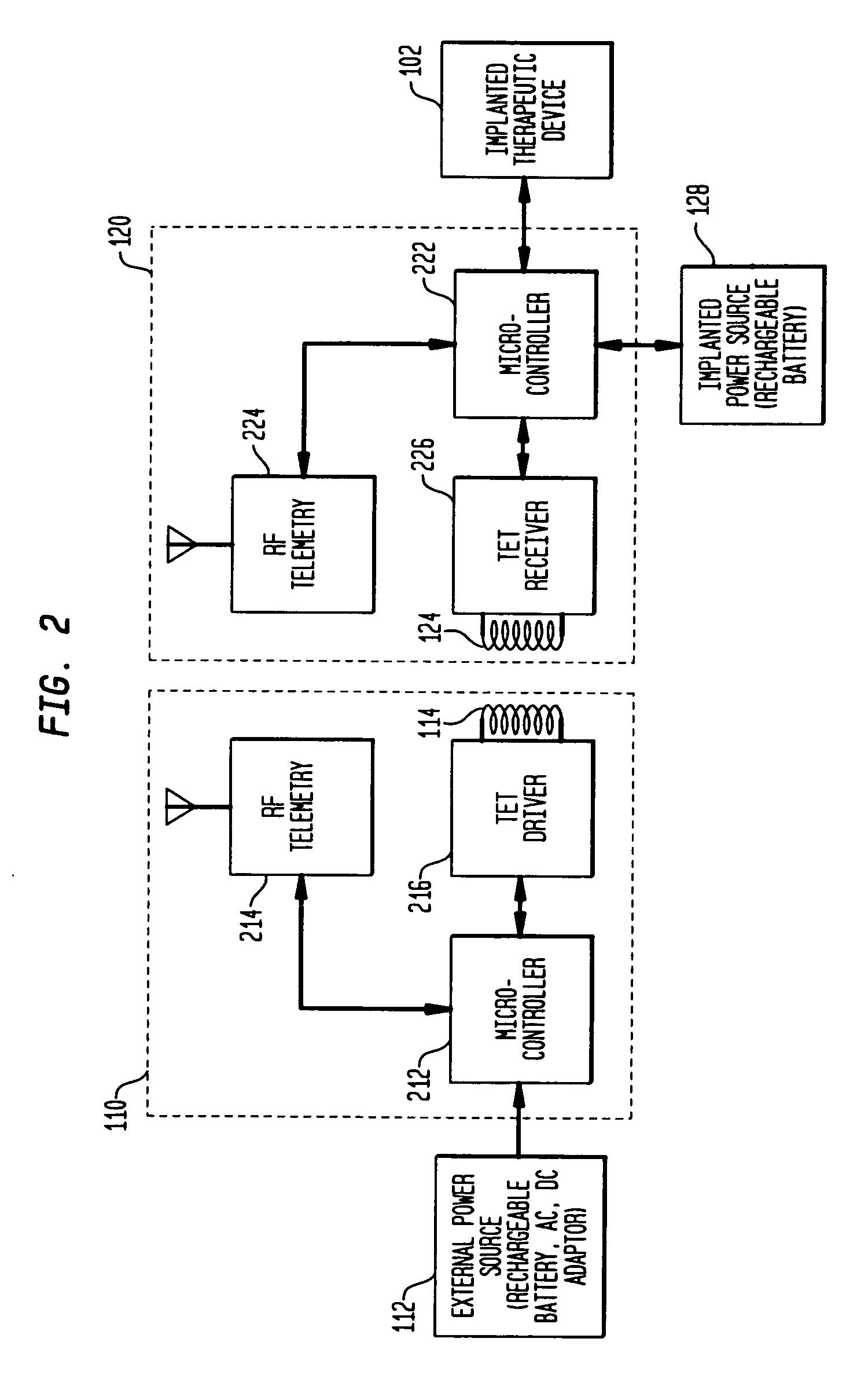
Tet system for implanted medical device
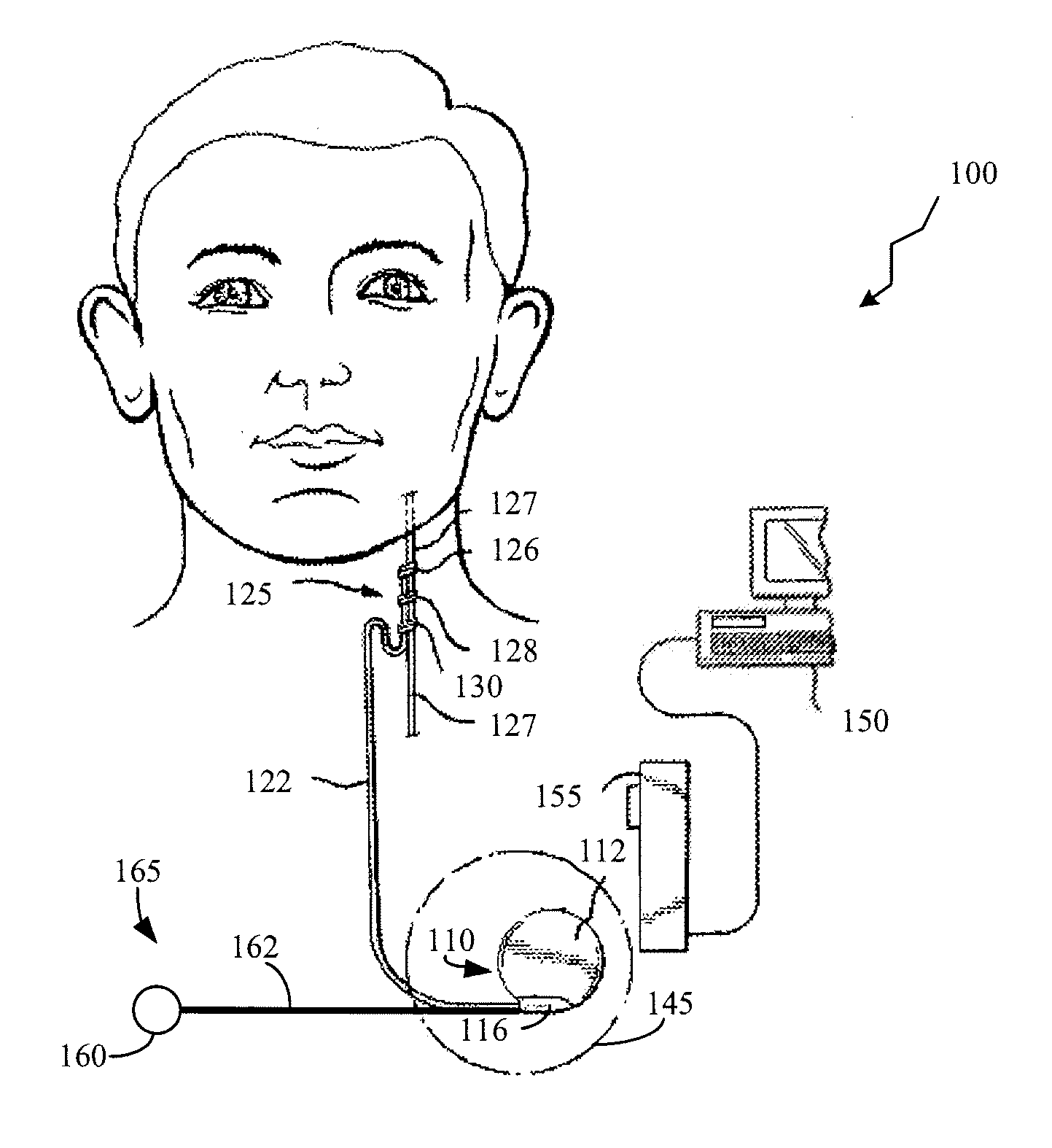
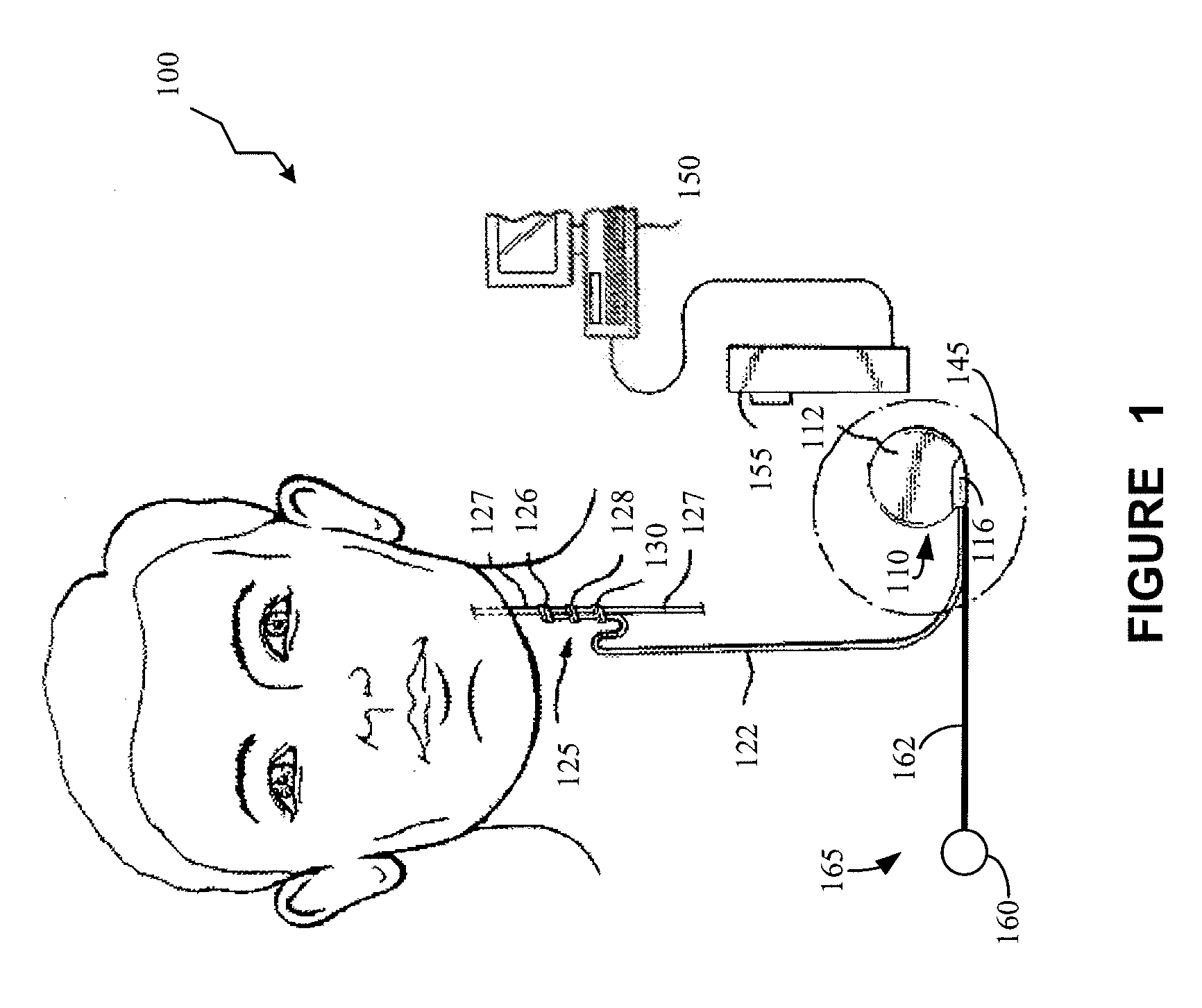
ActiveUS20100063347A1Current will often varyControl devicesMedical devicesCardiac cycleImplanted device
A TET system is provided which is operable to vary an amount of power transmitted from an external power supply to an implantable power unit in accordance with a monitored condition of the implantable power unit. In such way, the amount of power supplied to the implantable power unit for operating a pump, for example, can be varied in accordance with a cardiac cycle, so as to maintain the monitored condition in the power circuit within a desired range throughout the cardiac cycle.
Owner:HEARTWARE INC
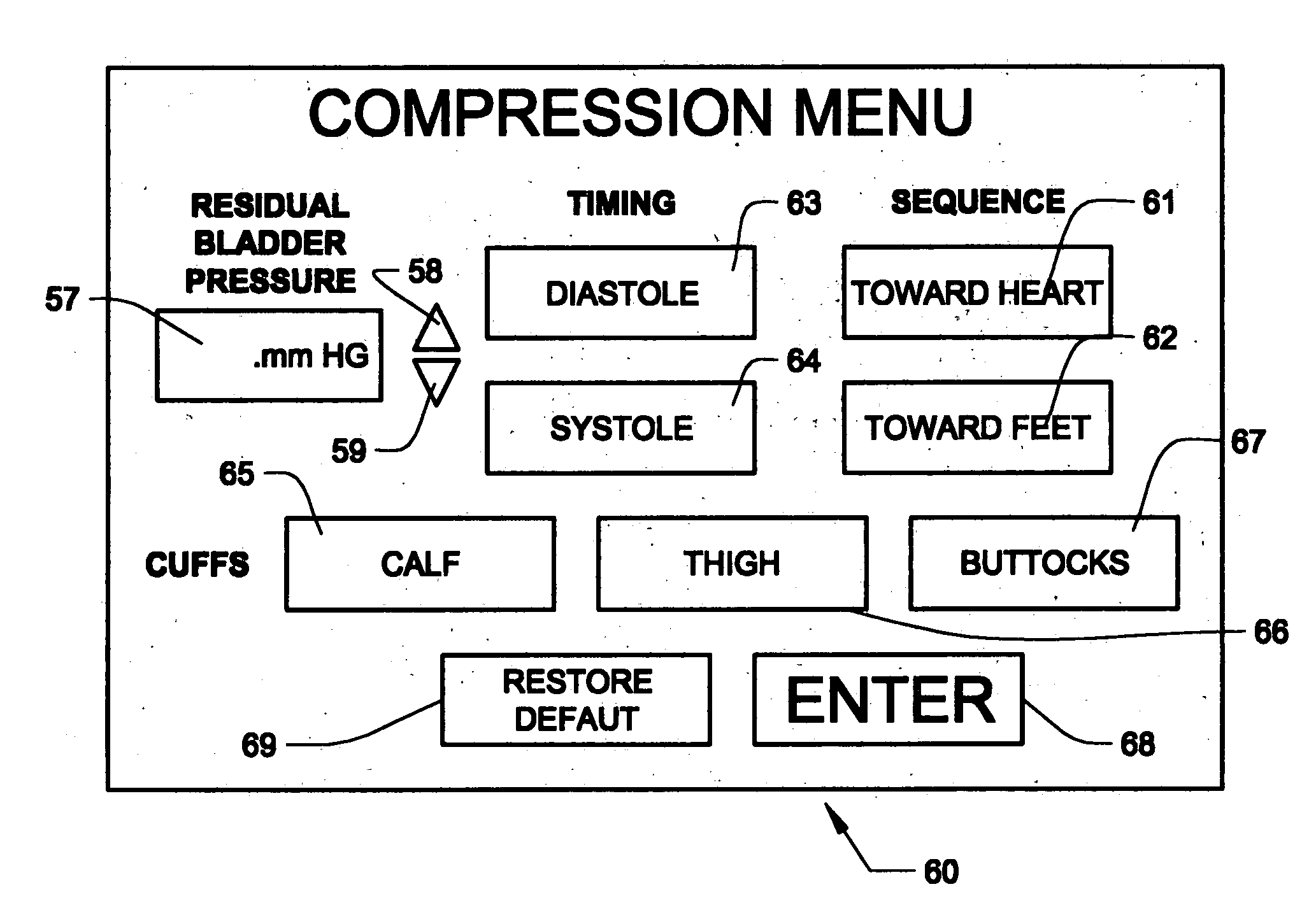
Devices and methods for non-invasively improving blood circulation
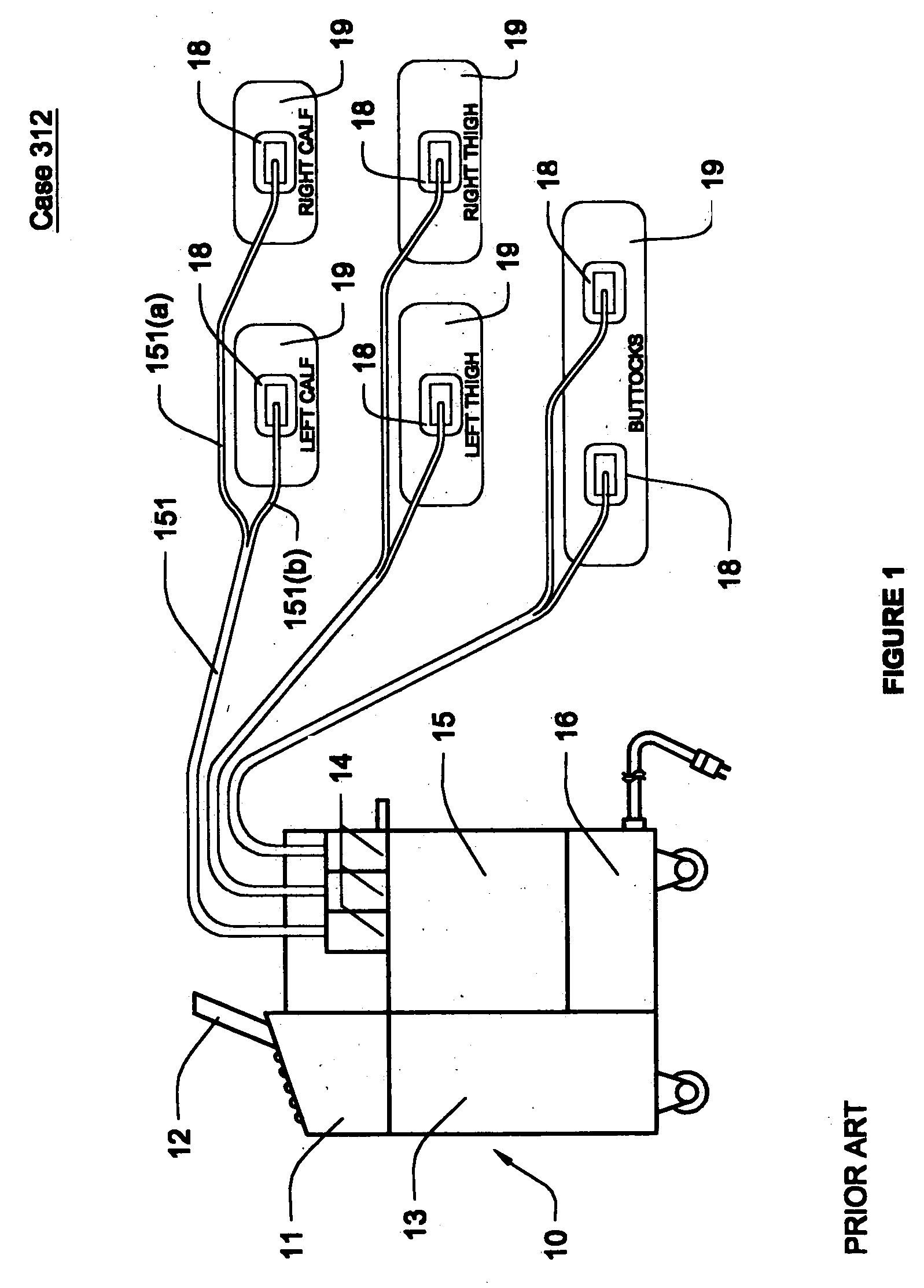
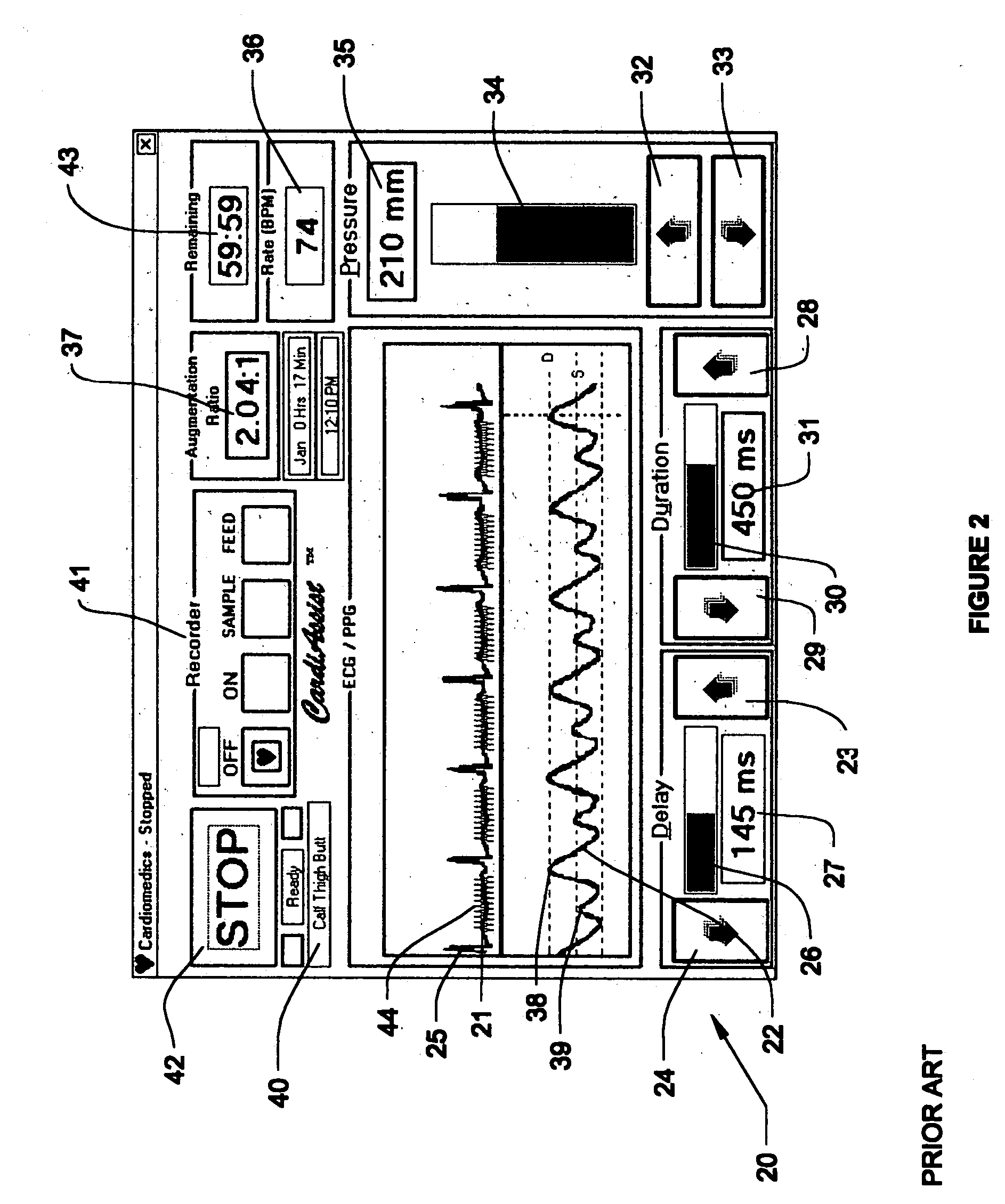
Circulatory assistance is provided in a non-invasive procedure safely and effectively using a microprocessor of an external counter pulsation device programmed to control the actuation of any or all of a plurality of valves, each of which is mounted on and in fluid communication with one of a plurality of individual inflatable bladders disposed in pockets within cuffs encasing the calves, thighs, buttocks, abdomen and / or chest of a person and an optional valve in fluid communication with the person's airway, in any desired sequence or order, toward the heart or toward the feet, either during diastole or systole, at desired inception times during the cardiac cycle, for selected durations and at chosen pressures, for treating a variety of cardiac, non-cardiac and circulatory conditions.
Owner:CARDIOMEDICS +1
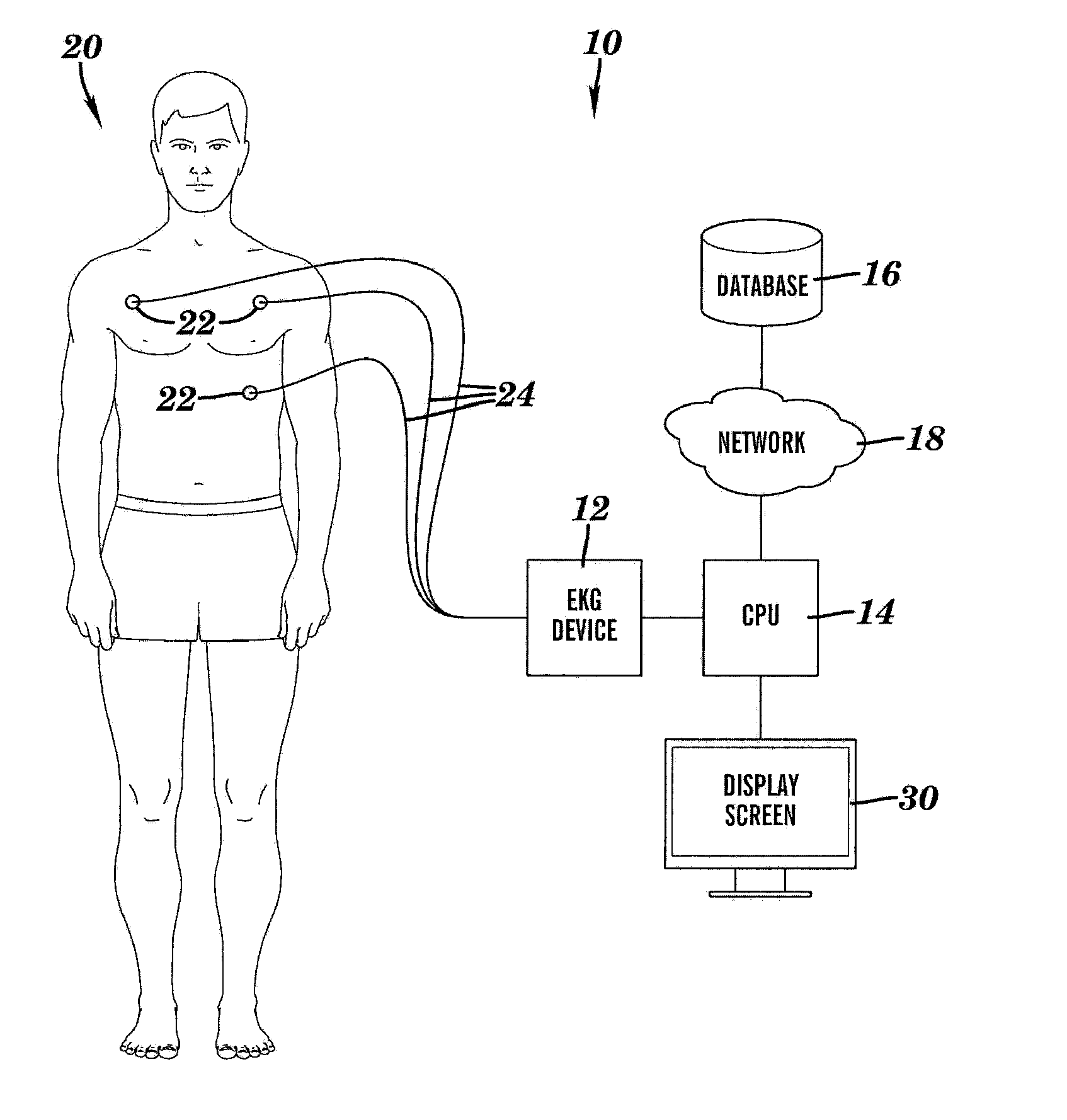
System and method for inferring disease similarity by shape matching of ECG time series
A method for inferring disease similarity by similarity retrieval of electrocardiogram time-series, comprising: acquiring user ECG waveforms correspondingly depicting many cardiac cycles of the heart of many users stored in a database; pre-processing each of the user ECG waveforms through pre-processing steps to isolate sets of single cardiac cycles corresponding to different heart-rates detected for each of the user ECG waveforms, each single cardiac cycle within the many cardiac cycles of the heart of many users corresponds to one single heart-rate detected. acquiring patient ECG waveforms depicting multiple cardiac cycles of the heart of a query patient; pre-processing the patient ECG waveforms through pre-processing steps to isolate sets of single cardiac cycles corresponding to different heart-rates detected for each of the patient ECG waveforms of the query patient, each single cardiac cycle within the multiple cardiac cycles of the heart of the query patient corresponds to one single heart-rate detected.
Owner:LINKEDIN
Synchronization of vagus nerve stimulation with the cardiac cycle of a patient
ActiveUS20070233194A1Facilitate conductionIncrease variabilityElectroencephalographySpinal electrodesIncreased heart rateRR interval
Disclosed herein are methods, systems, and apparatus for treating a medical condition of a patient, involving detecting a physiological cycle or cycles of the patient and applying an electrical signal to a portion of the patient's vagus nerve through an electrode at a selected point in the physiological cycle(s). The physiological cycle can be the cardiac and / or respiratory cycle. The selected point can be a point in the cardiac cycle correlated with increased afferent conduction on the vagus nerve, such as a point from about 10 msec to about 800 msec after an R-wave of the patient's ECG, optionally during inspiration by the patient. The selected point can be a point in the cardiac cycle when said applying increases heart rate variability, such as a point from about 10 msec to about 800 msec after an R-wave of the patient's ECG, optionally during expiration by the patient.
Owner:CATHOLIC HEALTHCARE WEST ST JOSEPHS HOSPITAL
Cardiac resynchronization therapy optimization using mechanical dyssynchrony and shortening parameters from realtime electrode motion tracking
Therapy optimization includes tracking electrode motion using an electroanatomic mapping system and generating, based on tracked electrode motion, one or more mechanical dyssynchrony metrics to thereby guide a clinician in therapy optimization (e.g., via optimal electrode sites, optimal therapy parameters, etc.). Such a method may include a vector analysis of electrode motion with respect to factors such as times in cardiac cycle, phases of a cardiac cycle, and therapy conditions, e.g., pacing sites, pacing parameters and pacing or no pacing. Differences in position-with-respect-to-time data for electrodes may also be used to provide measurements of mechanical dyssynchrony.
Owner:PACESETTER INC
Non-invasive method and device to monitor cardiac parameters
A method of and a device for non-invasively measuring the hemodynamic state of a subject or a human patient involve steps and units of non-invasively measuring cardiac cycle period, electrical-mechanical interval, mean arterial pressure, and ejection interval and converting the measured electrical-mechanical interval, mean arterial pressure and ejection interval into the cardiac parameters such as Preload, Afterload and Contractility, which are the common cardiac parameters used by an anesthesiologist.The converted hemodynamic state of a patient is displayed on a screen as a three-dimensional vector with each of its three coordinates respectively representing Preload, Afterload and Contractility. Therefore, a medical practitioner looks at the screen and quickly obtains the important and necessary information.
Owner:HIRSH ROBERT
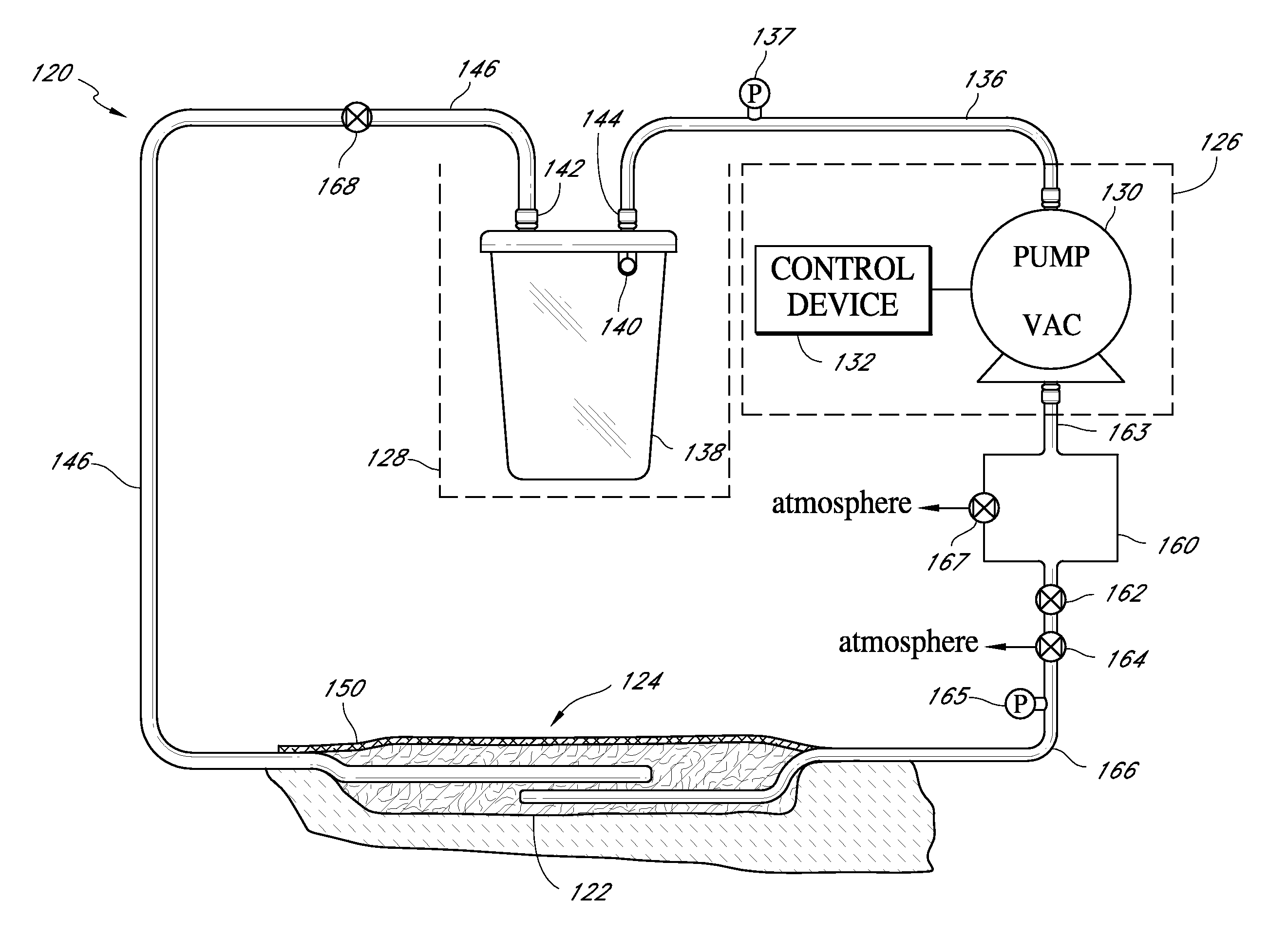
Sustained variable negative pressure wound treatment and method of controlling same
ActiveUS8366692B2Rapid and efficient mannerAssisted healingElectrocardiographyWound drainsWound dressingCardiac cycle
Owner:SMITH & NEPHEW PLC
Method and apparatus for creating a high resolution map of the electrical and mechanical properties of the heart
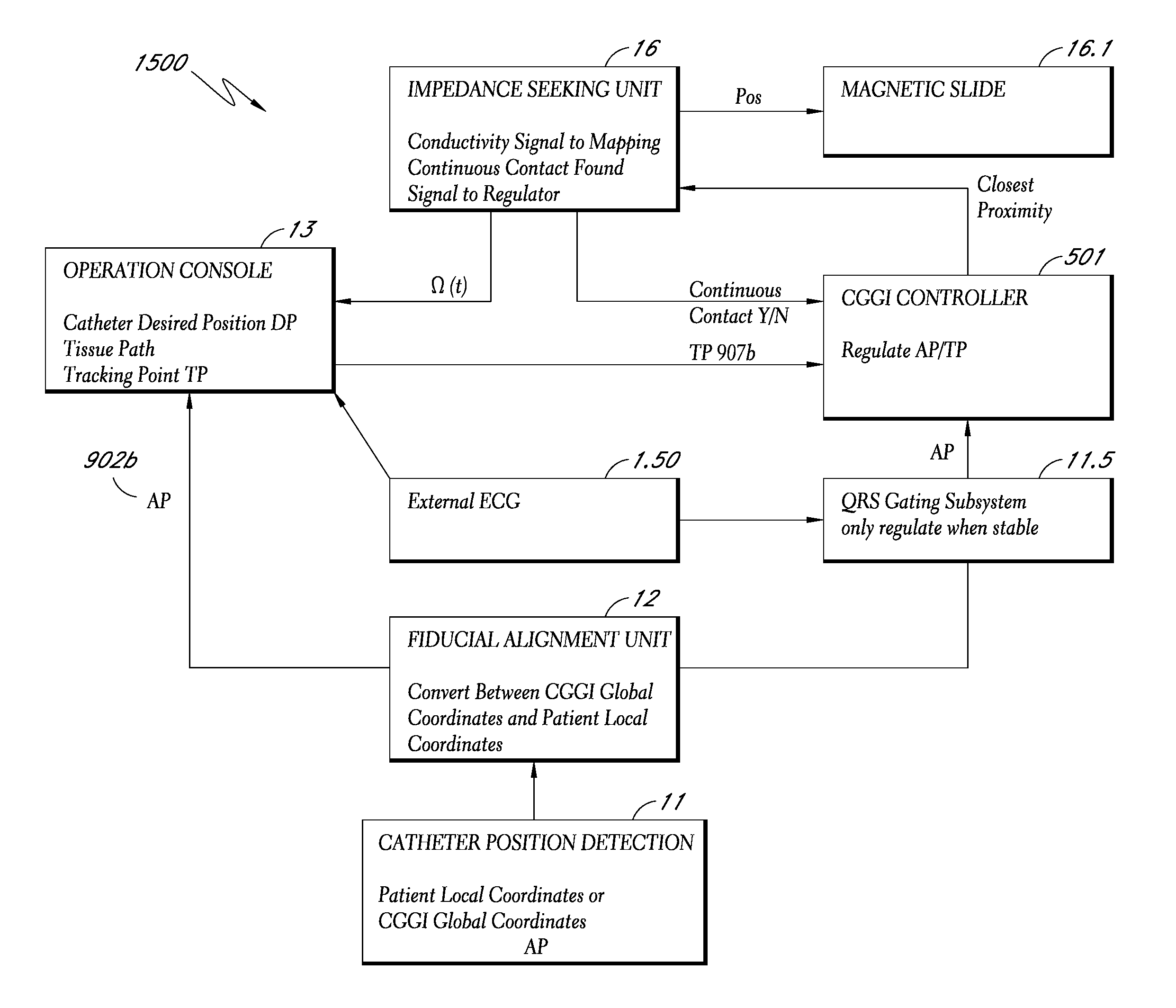
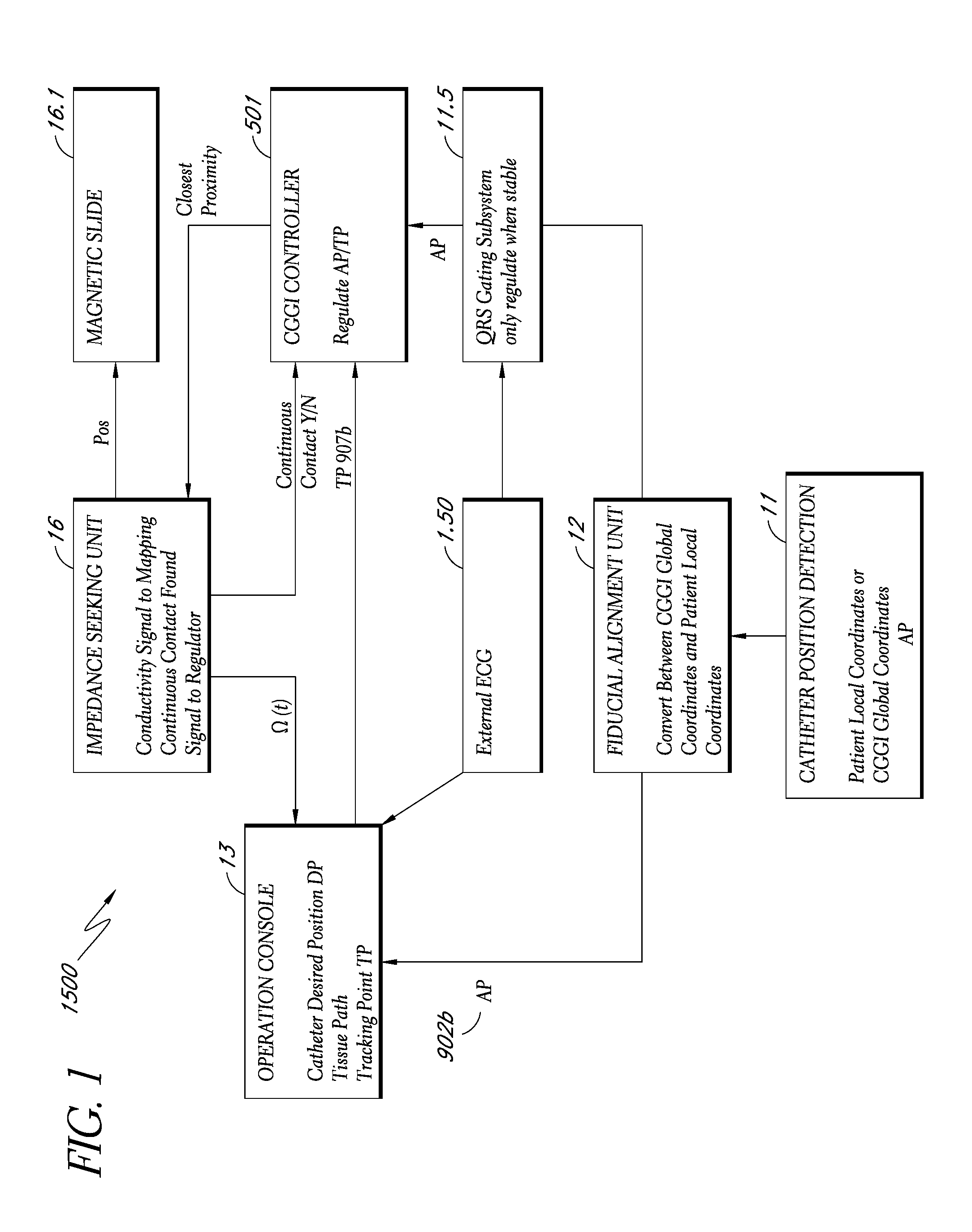
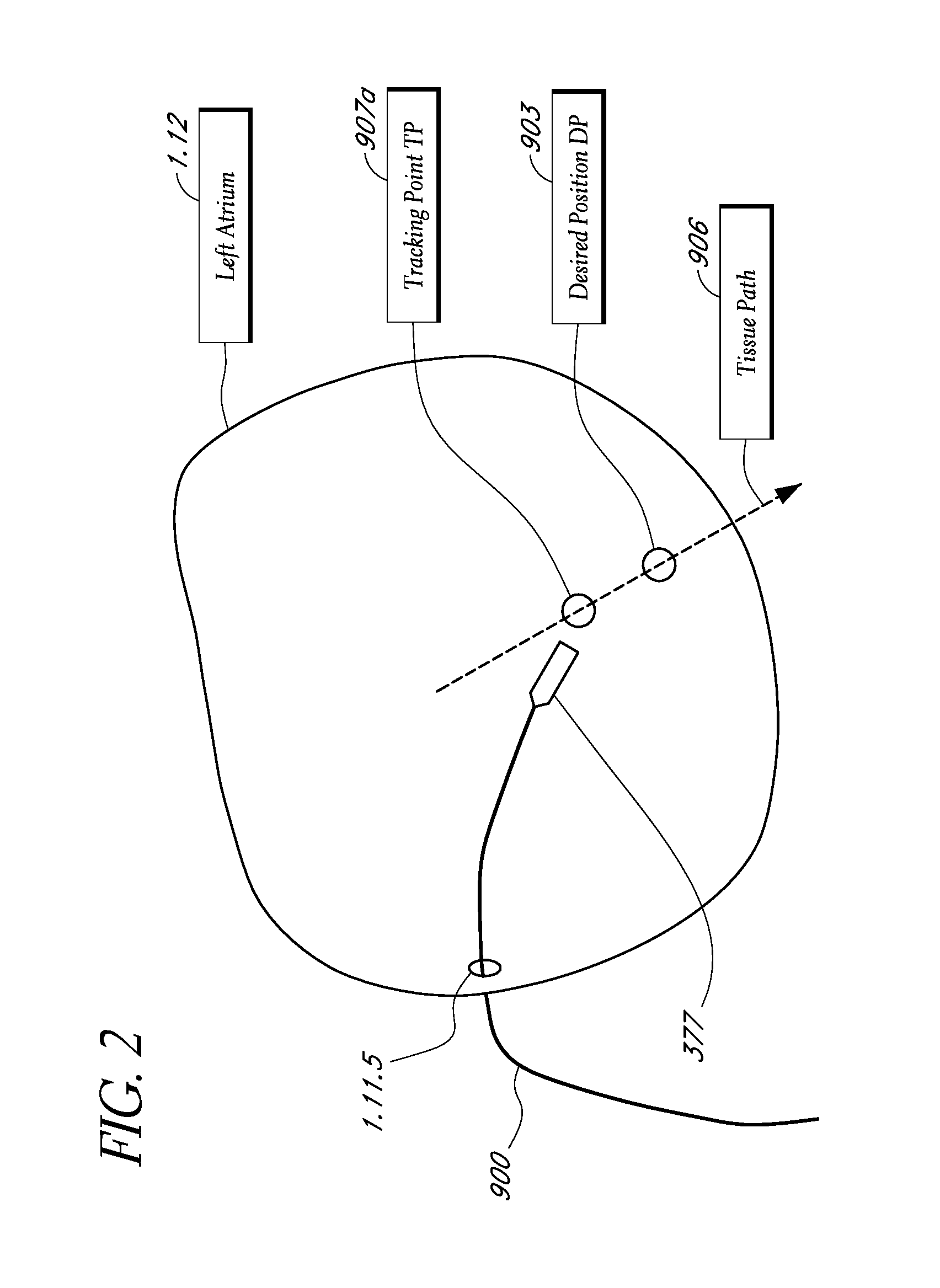
A system method that tracks one or more points on the surface of a cardiac tissue throughout a cardiac cycle and collect various types of data points which are then subsequently used to generate a corresponding model of the tissue and display the model as a 3D color coded image is described. In one embodiment, the system determines the position and orientation of a distal tip of a catheter, manipulates the catheter tip so as to maintain constant contact between the tip and a region of cardiac tissue using the impedance method, acquires positional and electrical data of the tip-tissue configuration through an entire heartbeat cycle, repeats the measurements as many times as needed in different tissue regions, and forms a 3D color coded map displaying various mechanical and electrical properties of the heart using the acquired data.
Owner:NEURO KINESIS CORP
Method and device to administer anesthetic and or vosactive agents according to non-invasively monitored cardiac and or neurological parameters
InactiveUS20090124867A1Efficiently safely teachingPumping capacity is overwhelmedMedical simulationRespiratorsCardiac cycleWhole body
A method of and a device for non-invasively measuring the neurological depressed state and the hemodynamic state of a human patient and involving steps and units of non-invasively measuring EEG, cardiac cycle period, electrical-mechanical interval, mean arterial pressure, and ejection interval and converting the EEG into a neurological index as well as converting the measured electrical-mechanical interval, mean arterial pressure and ejection interval into the cardiac parameters such as Preload, Afterload and Contractility, which are the common cardiac parameters used by an anesthesiologist. A general anesthetic is administered based upon the converted neurological index. A vasoactive agent is independently administered based upon the converted cardiac parameters as necessary in order to restore cardiovascular homeostasis in the patient. The converted neurological and hemodynamic state of a patient are displayed on a screen as an index value and a three-dimensional vector with each of its three coordinates respectively representing Preload, Afterload and Contractility. Therefore, a medical practitioner looks at the screen and quickly obtains the important and necessary information.
Owner:THE COOPER HEALTH SYST
Mechanical ventricular pacing capture detection for a post extrasystolic potentiation (PESP) pacing therapy using at least one lead-based accelerometer
InactiveUS20080234771A1Improve cardiac perfusionDecrease in cardiac performanceCatheterHeart stimulatorsPost extrasystolic potentiationAccelerometer
A system and method for monitoring at least one chamber of a heart (e.g., a left ventricular chamber) during delivery of extrasystolic stimulation to determine if the desired extra-systole (i.e., ventricular mechanical capture following refractory period expiration) occurs. The system includes an implantable or external cardiac stimulation device in association with a set of leads such as epicardial, endocardial, and / or coronary sinus leads equipped with motion sensor(s). The device receives and processes acceleration sensor signals to determine a signal characteristic indicative of chamber capture resulting from one or more pacing stimulus delivered closely following expiration of the refractory period. A threshold optimization method optionally evaluates capture and at least one of: runs an iterative routine to establish or re-establish chamber capture for the PESP therapy, sets a logical flag relating to chamber capture status and stores parameter(s) relating to successful chamber capture for one or more subsequent cardiac cycles.
Owner:MEDTRONIC INC
System and Method for Promoting Intrinsic Conduction Through Atrial Timing
An atrial based pacing protocol promotes intrinsic conduction. An entire cardiac cycle is monitored for ventricular activity and permitted to lapse with ventricular activity. Ventricular pacing is available in a cardiac cycle immediately subsequent to such a skipped beat. When monitoring for intrinsic ventricular events, an event is expected within a given window. If no such event is detected, the cardiac cycle in truncated, leading to a shorter cycle that is devoid of ventricular activity. The subsequent cycle has a high likelihood of a ventricular sensed event and a greater than normal AV interval is provided prior to pacing.
Owner:MEDTRONIC INC
Cardiac Resynchronization Therapy Optimization Using Vector Measurements Obtained From Realtime Electrode Position Tracking
An exemplary method includes selecting a first pair of electrodes to define a first vector and selecting a second pair of electrodes to define a second vector; acquiring position information during one or more cardiac cycles for the first and second pairs of electrodes wherein the acquiring comprises using each of the electrodes for measuring one or more electrical potentials in an electrical localization field established in the patient; and determining a dyssynchrony index by applying a cross-covariance technique to the position information for the first and the second vectors. Another method includes determining a phase shift based on the acquired position information for the first and the second vectors; and determining an interventricular delay based at least in part on the phase shift.
Owner:PACESETTER INC
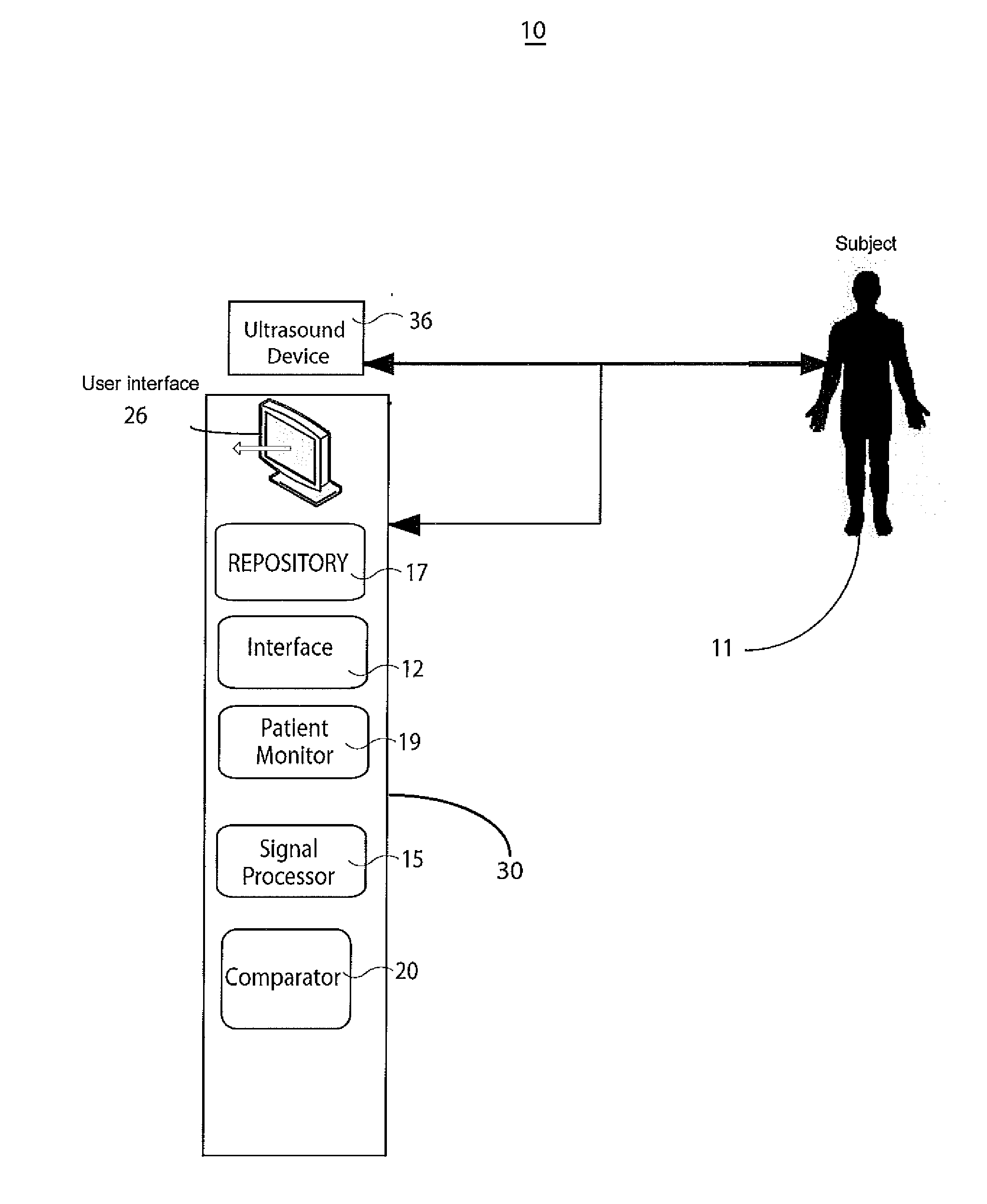
System for Cardiac Condition Detection and Characterization
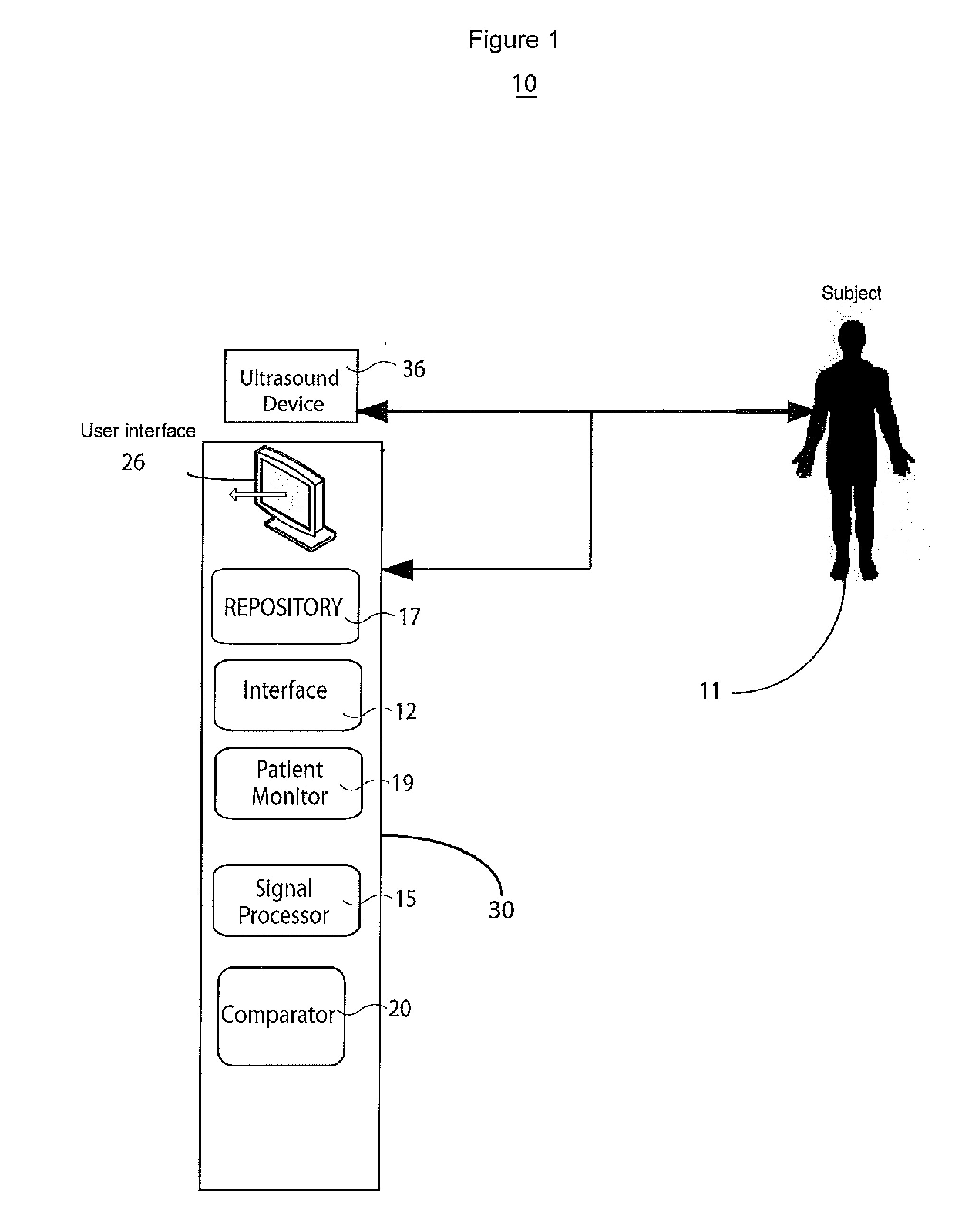
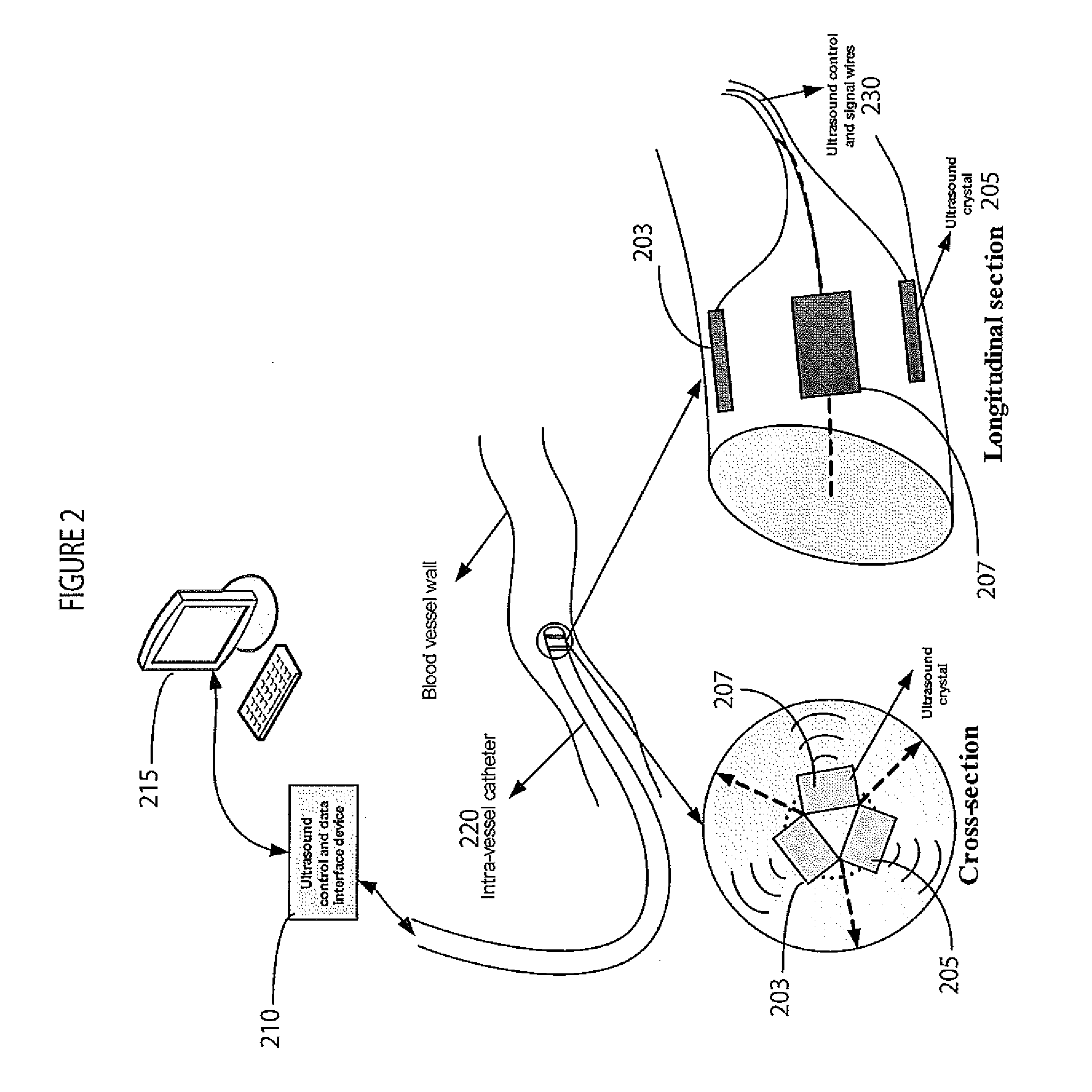
InactiveUS20110245669A1Little changeUltrasonic/sonic/infrasonic diagnosticsCatheterUltrasound deviceCardiac cycle
A system monitors and characterizes internal elasticity of a blood vessel to detect abnormality. A catheter system for heart performance characterization and abnormality detection, comprises an ultrasound device for emitting ultrasound wave signals within patient anatomy and acquiring corresponding ultrasound echo signals. A signal processor processes the ultrasound echo signals to, determine a signal indicating displacement of a tissue wall over at least one heart cycle and identify a displacement value in the displacement signal. The displacement value indicates a tissue wall displacement occurring at a point within a heart cycle. A comparator compares the tissue wall displacement value with a threshold value to provide a comparison indicator. A patient monitor, in response to the comparison indicator indicating the tissue wall displacement value exceeds the threshold value, generates an alert message associated with the threshold.
Owner:SIEMENS HEALTHCARE GMBH
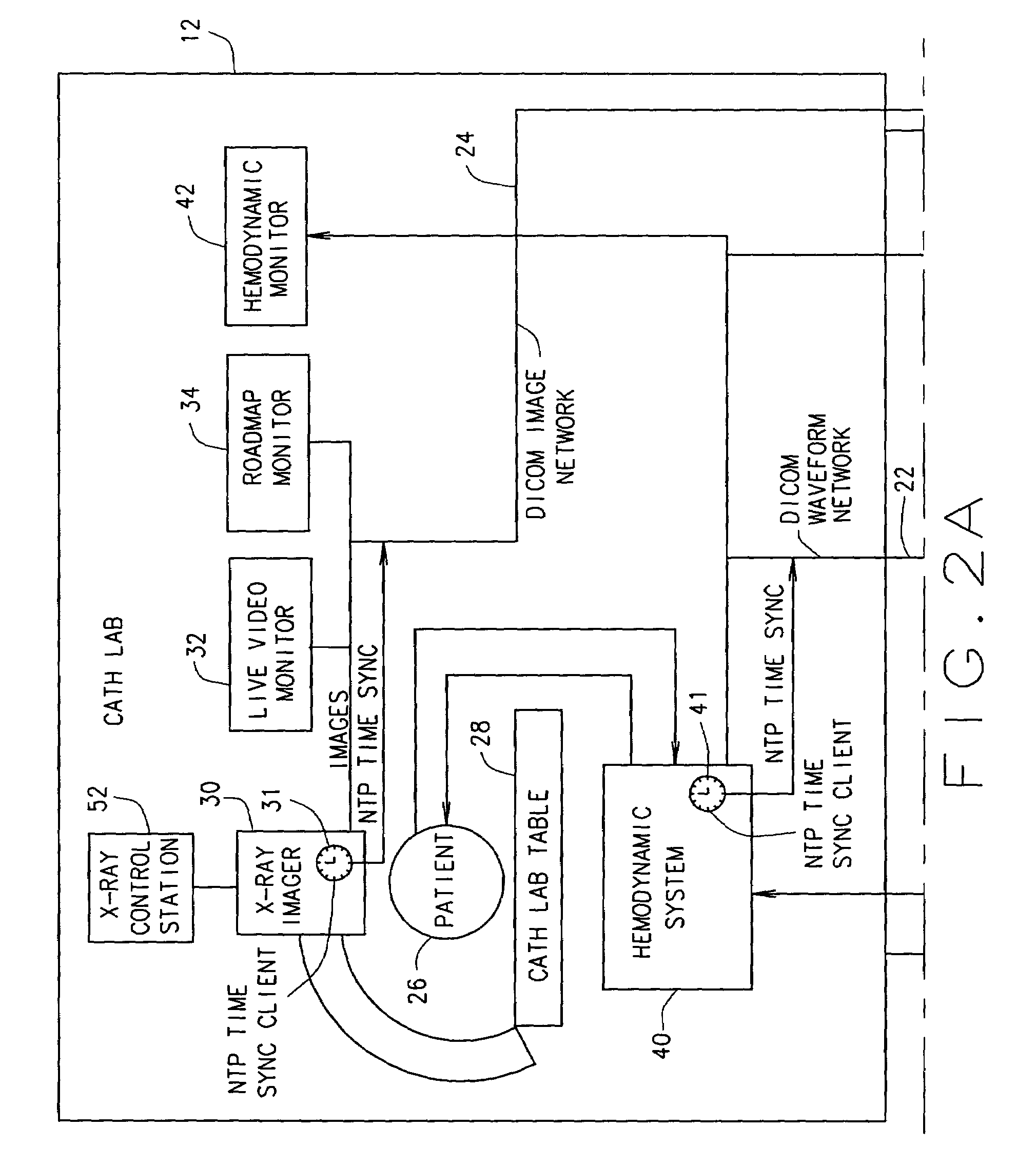
Methods and apparatus for analysis of angiographic and other cyclical images
A method for analyzing stored frames of time-stamped imaging data and time-stamped physiologic data obtained during a study of a patient during a plurality of cardiac cycles. The imaging data includes frames representative of a succession of images of the patient's heart. These configurations use a computer to analyze the stored physiologic data to determine times of a selected phase in the plurality of cardiac cycles, select, in the plurality of cardiac cycles, stored images corresponding to the determined times of the selected phase, and align and average images corresponding to the selected phase across the plurality of cardiac cycles to produce a single representative frame of image data.
Owner:GE MEDICAL SYST GLOBAL TECH CO LLC