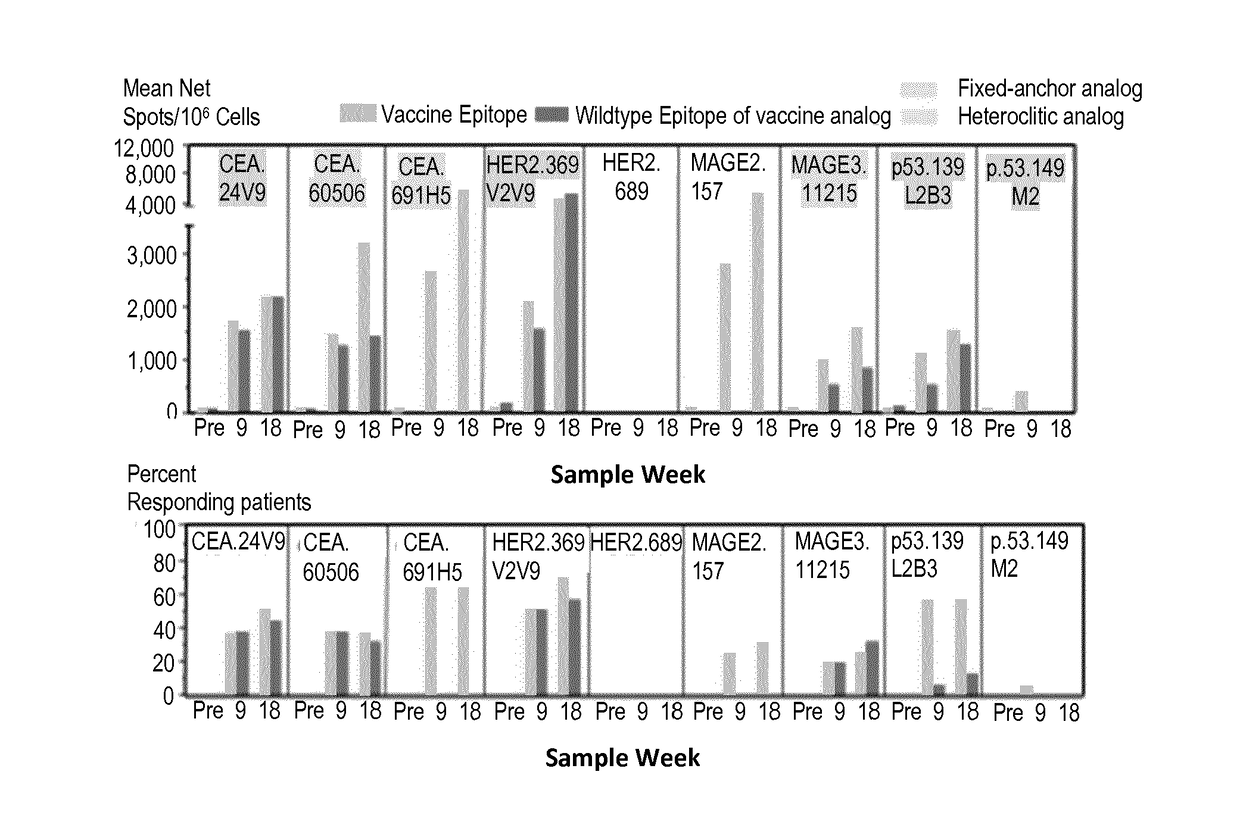
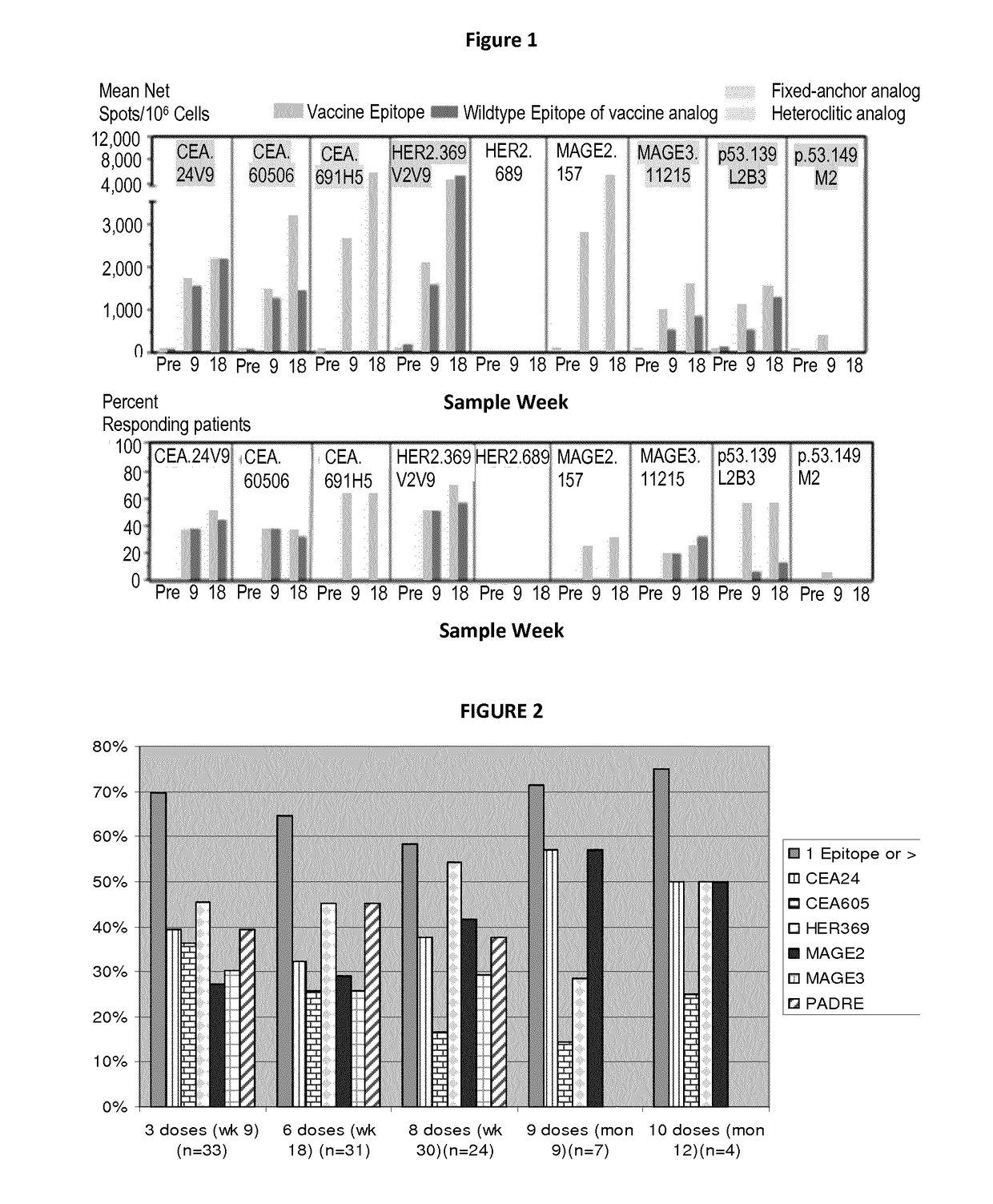
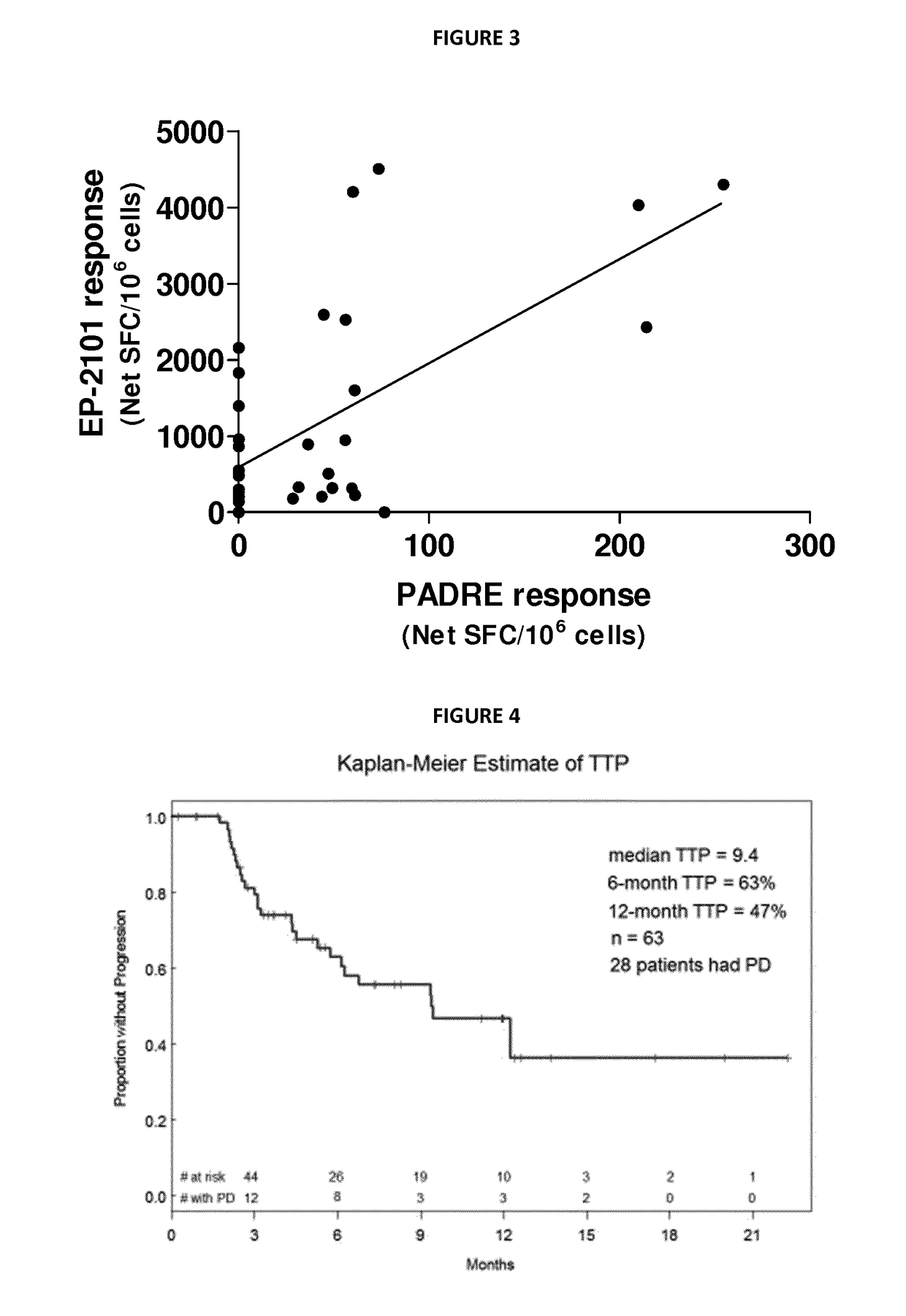
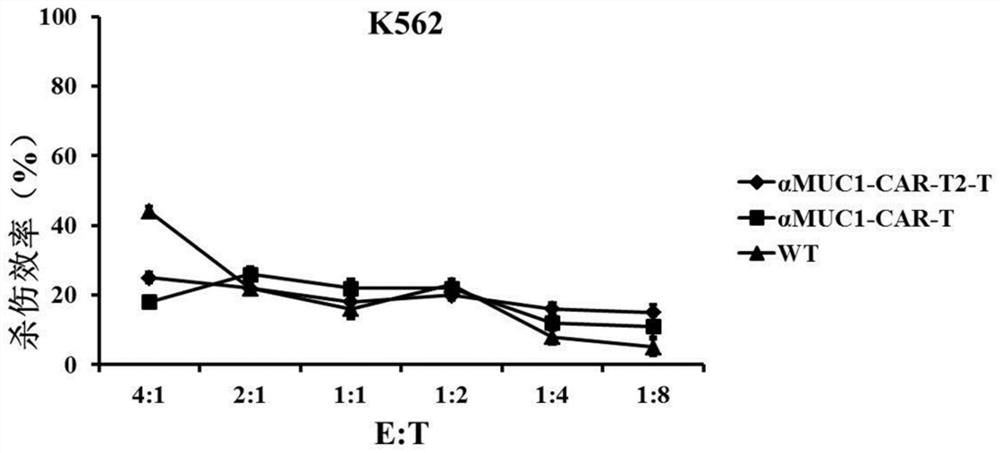
Patents
Literature
117 results about "Memory T cell" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Memory T cells are a subset of infection- and cancer-fighting T cells (also known as a T lymphocyte) that have previously encountered and responded to their cognate antigen; thus, the term antigen-experienced T cell is often applied. Such T cells can recognize foreign invaders, such as bacteria or viruses, as well as cancer cells. Memory T cells have become "experienced" by having encountered antigen during a prior infection, encounter with cancer, or previous vaccination. At a second encounter with the invader, memory T cells can reproduce to mount a faster and stronger immune response than the first time the immune system responded to the pathogen that entered the body. This behaviour is utilized in T lymphocyte proliferation assays, which can reveal exposure to specific antigens.
Composition capable of stimulating expansion of T cells
InactiveCN105543170AIncrease the number ofRaise the ratioBlood/immune system cellsCell culture active agentsCentral Memory T-CellHalf-life
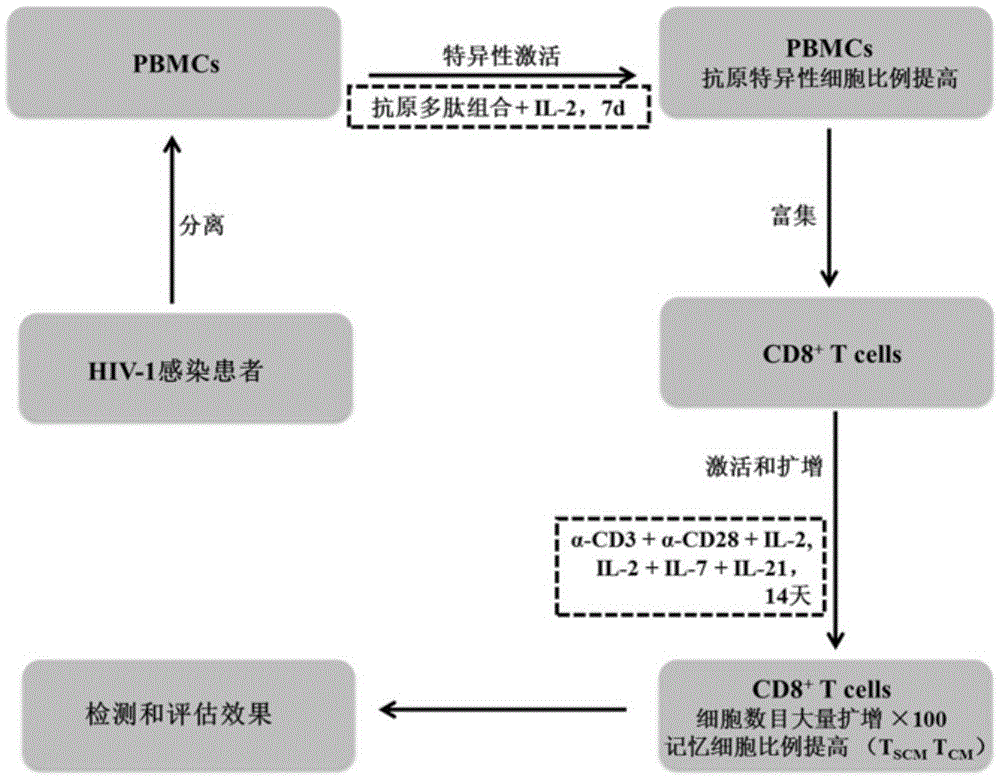
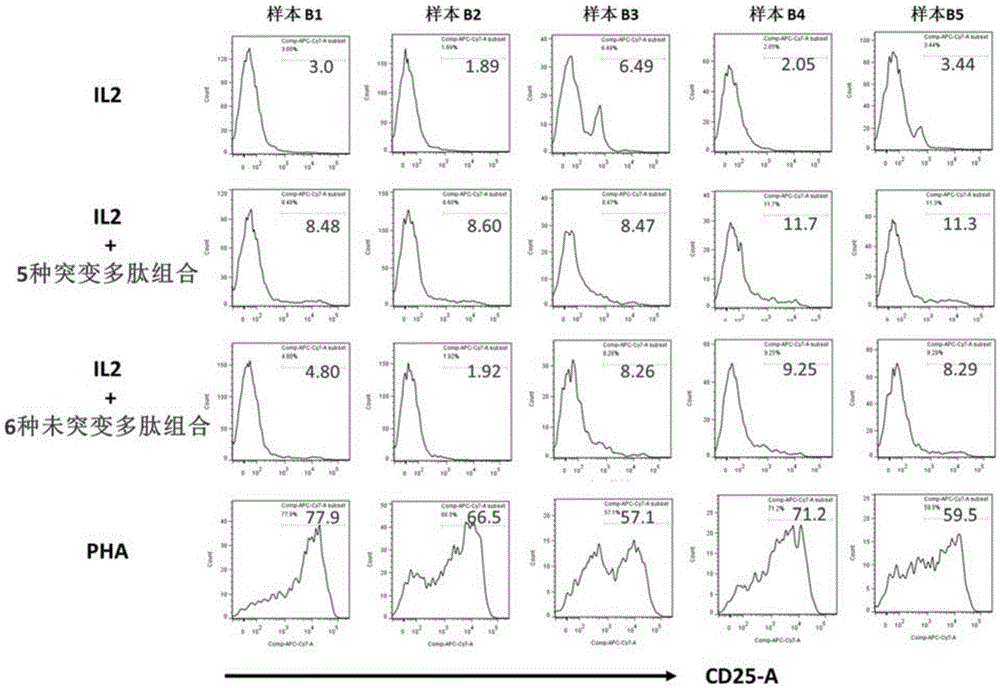
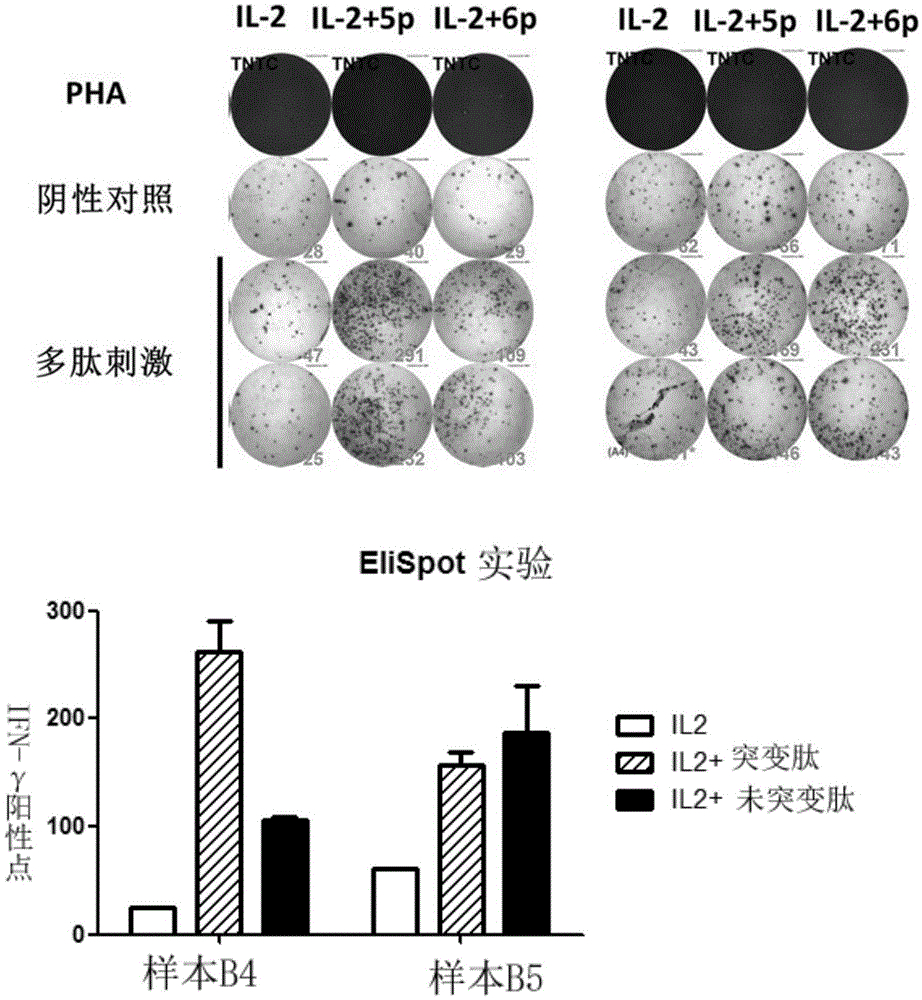
The invention discloses a composition capable of stimulating expansion of T cells. Active factors of the composition comprise an Anti-CD3 antibody, an Anti-CD28 antibody, IL-2, IL-7 and IL-21. Through an HIV-1 specificity polypeptide combination and a multiple cell factor combination, in-vitro expansion of the antigen-specific memory T cells (TSCM and TCM) of an HIV-1 infected person can be effectively and continuously stimulated, and meanwhile the proportion of the expanded CD8<+> memory T cells, especially the CD8<+> TSCM is increased. The half-life period of adoptive immunotherapy reinjection cells can be prolonged, therefore, the immune surveillance achieving ageing of the adoptive immunotherapy reinjection cells is prolonged, and the potential of controlling HIV-1 latent infection for a long term is achieved. According to the method, the advantages that operation is simple, and materials are easy to obtain are achieved, and good technical support is supplied to wide development of anti-infection adoptive immunotherapy.
Owner:SUN YAT SEN UNIV
Antigen and TLR agonist targeting co-loaded cationic phospholipid-polymer hybrid nanoparticle vaccine adjuvant, and preparation method and application thereof
ActiveCN108992666AStrong activationRealize the loadCancer antigen ingredientsPharmaceutical non-active ingredientsDendritic cellPhospholipid
The invention relates to an antigen and TLR agonist targeting co-loaded cationic phospholipid-polymer hybrid nanoparticle vaccine adjuvant, and a preparation method and an application thereof. A hydrophobic inner core is encapsulated with a TLR7 agonist, a phospholipid layer is encapsulated with a TLR4 agonist, an antigen is adsorbed by cationic phospholipids in the phospholipid layer, and ligation of a mannose ligand makes the vaccine adjuvant like a specific targeting antigen to have the dendritic cell presenting ability, so the DC cell intake and the maturation promoting effect are enhanced; and the antigen is protected by hybridized nanoparticles, the antigen uptake by DC cells is improved, immune response after antigen stimulation is significantly enhanced by the TLR agonist, and thecross-presentation of the antigen is significantly improved, so vaccine adjuvant has a strong potent T cell killing effect, can induce cytokine secretion, has a long-acting memory T cell response, andhas an excellent prevention effect on tumors.
Owner:INST OF BIOMEDICAL ENG CHINESE ACAD OF MEDICAL SCI
Methods for eliminating at least a substantial portion of a clonal antigen-specific memory T cell subpopulation
InactiveUS8617884B2Reduce riskReduce severityMammal material medical ingredientsArtificial cell constructsCentral Memory T-CellPartial cloning
The present invention relates generally to methods for stimulating T cells, and more particularly, to methods to eliminate undesired (e.g., autoreactive, alloreactive, pathogenic) subpopulations of T cells from a mixed population of T cells, thereby restoring the normal immune repertoire of said T cells. The present invention also relates to compositions of cells, including stimulated T cells having restored immune repertoire and uses thereof.
Owner:LIFE TECH CORP
Cationic phospholipid-polymer hybridized nanoparticle vaccine adjuvant of common-carrier antigen, MPLA (Monophosphoryl Lipid A) and IMQ (Imiquimod) as well as preparation method and application thereof
ActiveCN108743939AGood biocompatibilityPromote degradationPharmaceutical non-active ingredientsAntibody medical ingredientsDendritic cellPhospholipid
The invention relates to a cationic phospholipid-polymer hybridized nanoparticle vaccine adjuvant of a common-carrier antigen, MPLA (Monophosphoryl Lipid A) and IMQ (Imiquimod) as well as a preparation method and application thereof. The vaccine adjuvant is characterized in that the IMQ as a TLR7 agonist is loaded on a hydrophobic core; the MPLA as a TLR4 agonist is loaded in a phopholipid layer;cationic phospholipid DOTAP (1,2-dioleoy-3-trimethylammonium-propane) in the phopholipid layer is used for adsorbing an antigen; the antigen is protected through hybridized nanoparticles, and the ingestion of the antigen by dendritic cells is improved; immune response after antigen stimulation is improved remarkably through the TLR agonist, and cross-presentation of the antigen is improved remarkably. The hybridized nanoparticles as the vaccine adjuvant can load the antigen and different types of TLR agonists simultaneously, can deliver the antigen through a plurality of immune paths, and promotes the DC activation and maturation. The cross-presentation level is raised, a strong and powerful T-cell killing effect is achieved, cell factor secretion is induced, a long-term memory T-cell reaction is generated, and higher prevention capability for tumors is achieved.
Owner:INST OF BIOMEDICAL ENG CHINESE ACAD OF MEDICAL SCI
Method to improve the immune function of t cells
InactiveUS20150017185A1Enhance immune responseEnhancing function of T cellOrganic active ingredientsImmunoglobulins against cell receptors/antigens/surface-determinantsCentral Memory T-CellP38map kinase
The present invention provides a method for enhancing the immune function of a memory T cell which comprises the step of coinhibting signalling via an inhibitory receptor which regulates T cell exhaustion and via the p38 MAP kinase signalling pathway in the T cell, and a method for treating and / or preventing an immune condition in a subject, which comprises the step of enhancing the immune function of a memory T cell in the subject by such a method. There is also provided a pharmaceutical composition or kit comprising an agent capable of inhibiting signalling via an inhibitory receptor which regulates T cell exhaustion, such as PD-1, and an agent capable of inhibiting the p38 MAP kinase signalling pathway.
Owner:UCL BUSINESS PLC
Methods for activating t cells
InactiveUS20130045491A1Promote activationFacilitate functioningGenetically modified cellsBlood/immune system cellsCentral Memory T-CellLymphocyte
The present invention is directed to a method for promoting function, activation and proliferation of T helper lymphocytes such as those that express IL17 and IL22 (Th-IL17+ and Th-IL22+ T cells). The method features isolating a population of T cells from a subject such as a human and incubating the population of T cells in a serum-free culture medium that contains one or more cytokine. The T cells may be memory T cells (TM) such as, for example, CCR6+CD45RO+ memory T cells (TM), and the one or more cytokine may be, for instance, one or more of IL-2, IL-7 and IL-15. Likewise, the invention provides a method for expanding an activated population of T helper lymphocytes by the same means.
Owner:NEW YORK UNIV
Methods and compositions to enhance immune responses via recall antigens
InactiveUS20060263389A1Reduce capacityAvoid confusionBacterial antigen ingredientsAntibody mimetics/scaffoldsCentral Memory T-CellRecall antigen
The present invention provides a composition comprising a chimeric polypeptide comprising a recall antigen that reactivates memory T cells in a subject and a new antigen that activates naïve B cells in a subject and a composition comprising a nucleic acid encoding a chimeric polypeptide comprising a recall antigen that reactivates memory T cells in a subject and a new antigen that activates naïve B cells in a subject. Further provided are methods of modulating an immune response, as well as treating and / or preventing disease in subjects in whom the ability to mount an immune response to a new antigen is impaired, by administering the compositions of this invention.
Owner:BOARD OF RGT THE UNIV OF TEXAS SYST
Method for in-vitro culture and enrichment of CD8+ T cells
ActiveCN106566806APromote proliferationGood energyBlood/immune system cellsCell culture active agentsHuman bodyCentral Memory T-Cell
The invention belongs to the field of cell culture, particularly relates to a method for in-vitro culture and enrichment of CD8+ T cells, and more particularly relates to a method for directional amplification of a large amount of CD8+ T cells in peripheral blood mononuclear cells of a human body by using an irritant. The T cells obtained by the technical scheme are large in proliferation number and high in activity; the ratio of the CD8+ T cells to CD4+ T cells can be increased to 2-5 : 1 from about 1 : 2 in a normal physiological status; and in a target culture, the content of central memory T cells exceeds 30% based on the total cell number.
Owner:英威福赛生物技术(天津)有限公司
Construction method of human stem memory T cell bank
InactiveCN105821483ALow costEasy to operateCulture processDead animal preservationCentral Memory T-CellPeripheral blood mononuclear cell
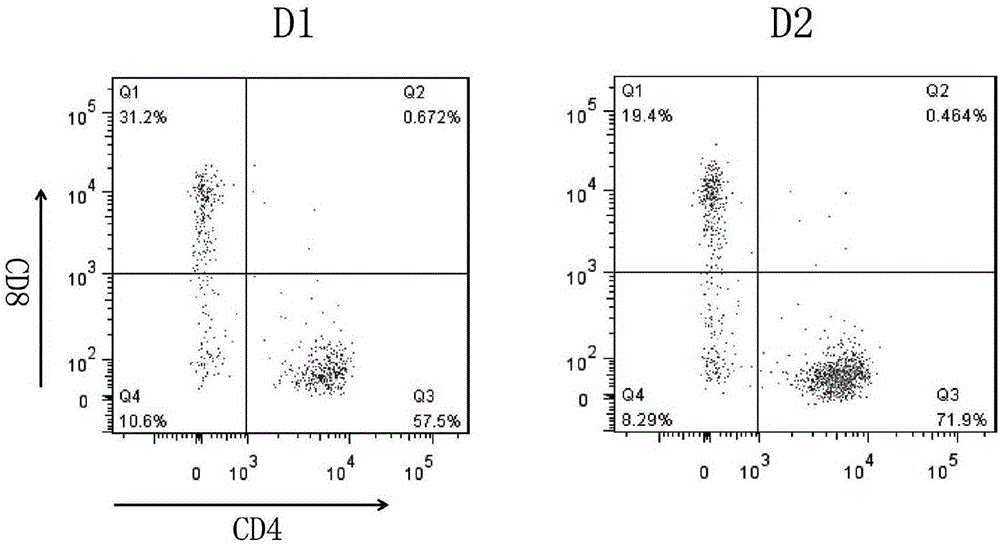
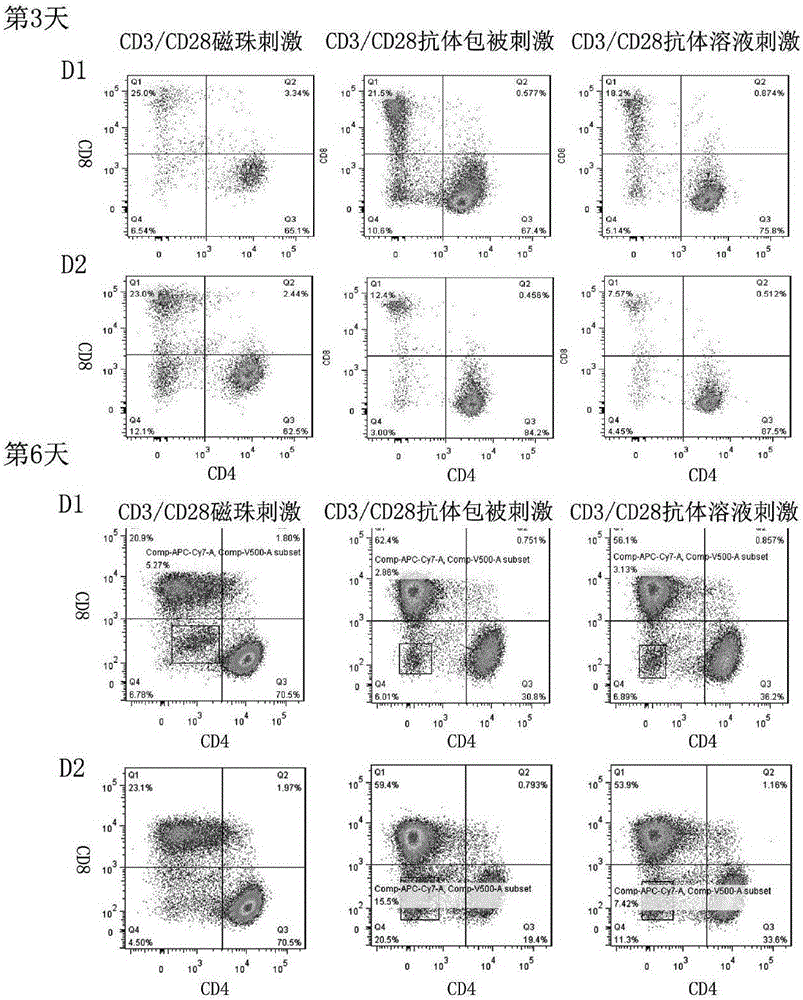
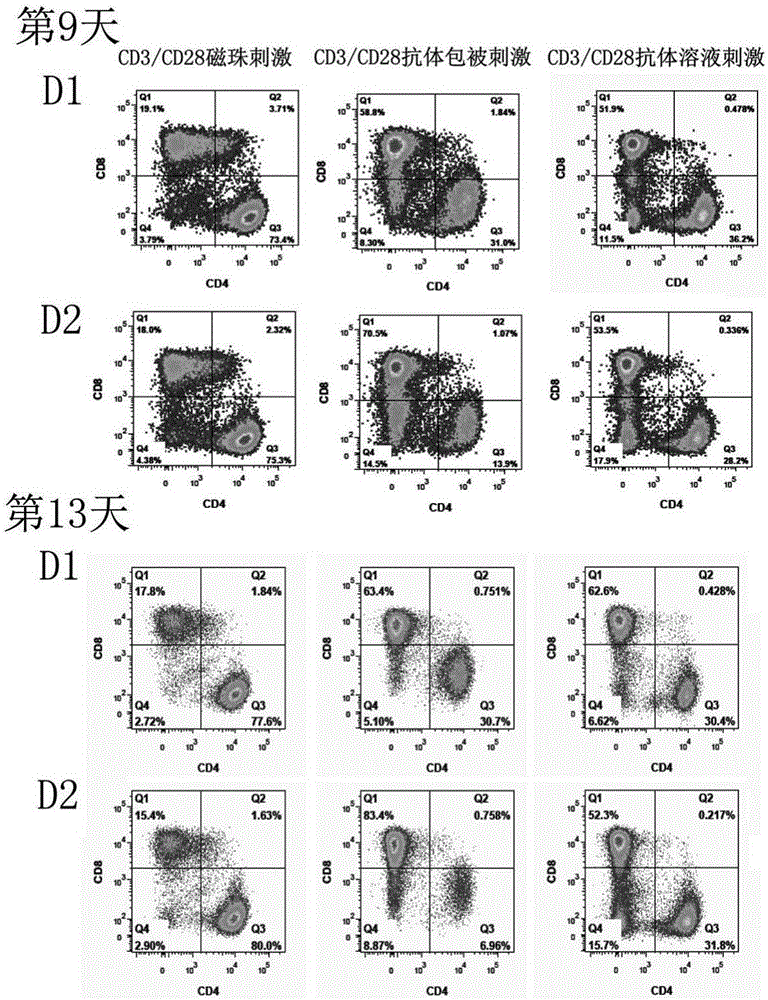
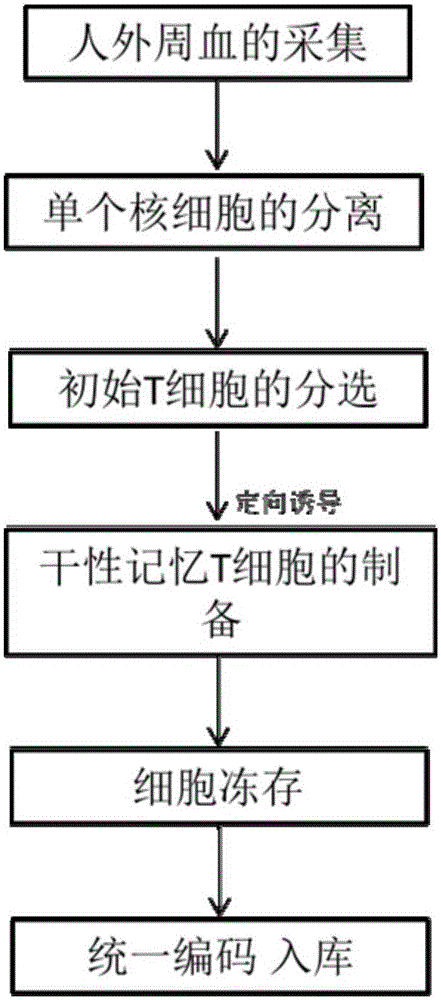
The invention relates to a construction method of a human stem memory T cell bank. The method comprises the following steps of human peripheral blood collection, peripheral blood mononuclear cell separation, initial T cell sorting, stem memory T cell preparation, cell cryopreservation and coding and storage. According to the construction method of the human stem memory T cell bank, a method of conducting directionally inducing in vitro is adopted, enough stem memory T cells (TSCM) can be obtained in a short time and subjected to bank construction, the volume of needed peripheral blood is small, and the construction method of the human stem memory T cell bank has the advantages of being low in cost, low in requirement on lab condition, easy to operate and the like.
Owner:SHENZHEN HUADA GENE INST
Compositions and methods for the prevention and treatment of cancer
Conventional cancer immunotherapy falls short at efficiently expanding T cells that specifically target cancerous cells in numbers sufficient to significantly reduce the tumor size or cancerous cell number in vivo. To overcome this limitation, provided herein are nanoparticles coated with MHC class I and / or class II molecules presenting tumor-specific antigens and co-stimulatory molecules and their use to expand antigen-specific anti-tumorigenic T cellsto levels not achieved in current immunotherapeutic techniques. These antigen-specific anti-tumorigenic T cells include cytotoxic T cells, effector T cells, memory T cells, and helper T cells that are necessary to initiate and maintain a substantial immune response against metastatic or non- metastatic cancerous, pre-cancerous, or neoplastic cells in vivo.; The present invention describes a systemic approach to targeting cancerous or pre-cancerous cells that are circulating cells, as in lymphomas, migratory metastatic cells, and solid tumors.
Owner:UTI LLP
Enhancing immunity to tuberculosis
ActiveUS9821047B2Enhancing the immune systemEnhance phagocytosisSsRNA viruses negative-senseBacterial antigen ingredientsCentral Memory T-CellPhagocytic response
The invention is directed to compositions and methods for generating or enhancing the immune system of a patient against infection by a pathogen, and in particular MTB. Compositions of the invention contain one or more non-naturally occurring antigens that generate an effective cellular or humoral immune response to MTB and / or antibodies that are specifically reactive to mycolic acid or to the surface of MTB. The greater activity of the immune system generated by a vaccine of the invention involve an conjugation of peptides to increase in the generation of memory T cells that provide for a greater and / or longer lived or extended response to an MTB infection. Preferably a response involves an increased generation of antibodies that enhance immunity against MTB infection and promote an enhanced phagocytic response.
Owner:LONGHORN VACCINES & DIAGNOSTICS LLC
Method for preparing memory T cells
InactiveCN109423478ASolve bottlenecksProlong survival timeMammal material medical ingredientsBlood/immune system cellsPeripheral blood mononuclear cellCentral Memory T-Cell
The present invention provides a method for efficiently preparing memory T cells by combining TWS119 and cytokines IL-7 and IL-21. The method includes collecting human peripheral blood; performing density gradient centrifugation to obtain human peripheral blood mononuclear cells; sorting CD8<+>T cells with a magnetic separation sorter; adding the CD8<+>T cells into a medium containing a stimulantCD3 / CD28 conjugated magnetic beads, cytokines IL-7and IL-21 and TWS119; and performing amplification so that a high number of CD8<+>T cells having Tcm and Tscm phenotypes can be obtained in the seventh day.
Owner:THE FIRST AFFILIATED HOSPITAL OF ZHENGZHOU UNIV
PBMC induced TCM and application
InactiveCN107365748ATargetedSolve the poor proliferation ability of memory T cellsImmunoglobulin superfamilyGenetically modified cellsAbnormal tissue growthClinical efficacy
The present invention relates to the technical field of immune cell targeted therapy, in particular to a kind of human peripheral blood mononuclear cells induced to differentiate into memory immune cells. TCM cells induced by frozen peripheral blood mononuclear cells provide a basis for clinical tumor treatment, and can Resuscitate cells at any time according to the treatment plan of tumor patients, which solves the problem of poor T cell proliferation and immune incompetence in tumor patients; at present, it is difficult for the reinfused immune cells, especially cloned effector T cells, to survive for a long time in the body, which greatly affects the adoptiveness Clinical efficacy of cell therapy. Memory T cells have long-term memory, and TCM cells are transfected with TCR genes to endow TCM cells with targeting.
Owner:路春光
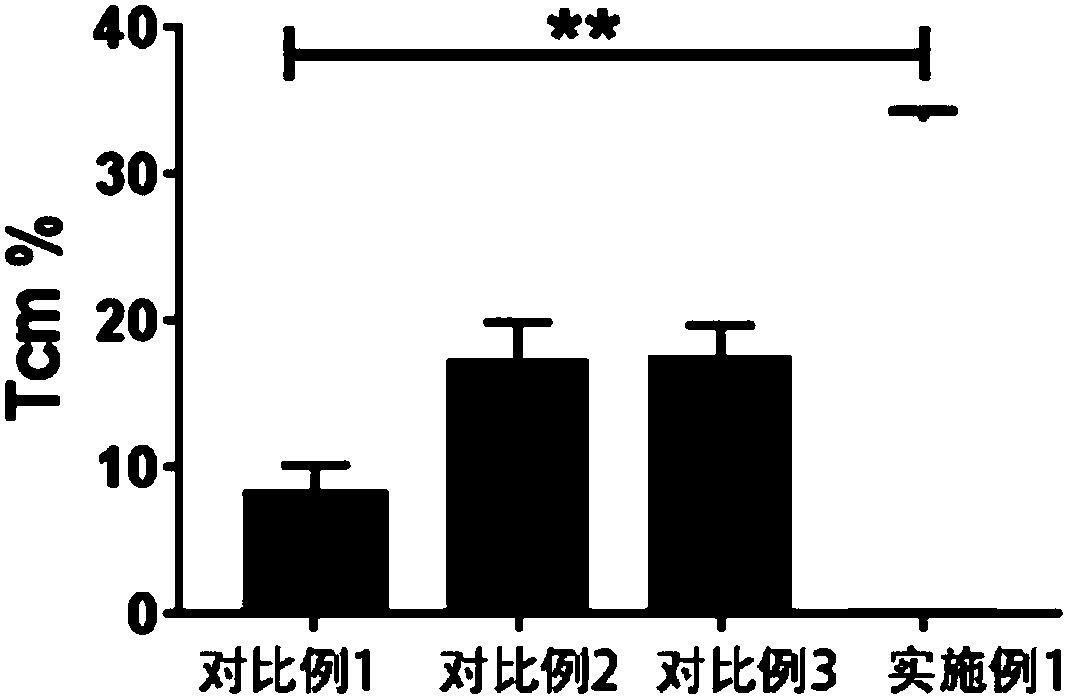
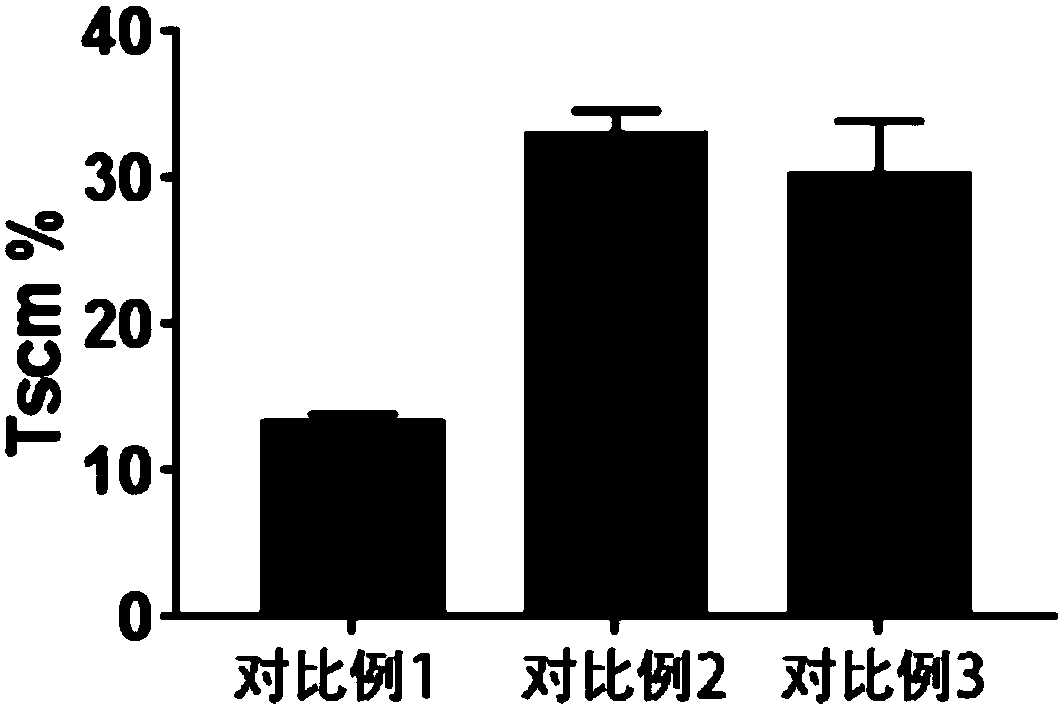
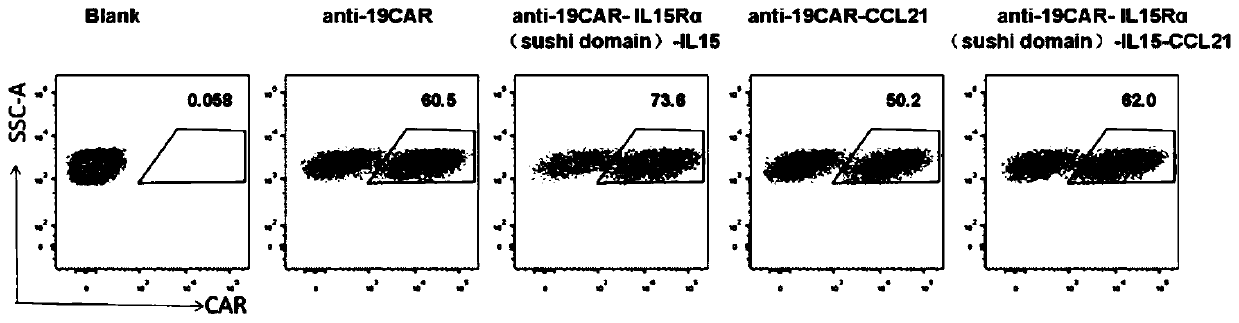
Chimeric antigen receptor-T cells secreting and expressing IL15RA-IL15 fusion proteins and CCL21 chemotactic factors, and applications thereof
ActiveCN110818803AHigh activityPromote proliferationPolypeptide with localisation/targeting motifImmunoglobulin superfamilyDendritic cellNatural Killer Cell Inhibitory Receptors
The invention discloses chimeric antigen receptor-T cells secreting and expressing IL15RA-IL15 fusion proteins and CCL21 chemotactic factors as well as application thereof. Super IL15 can promote activation and proliferation of T (especially memory T) cells, NK cells and NKT cells; and CCL21 can recruit peripheral CCR7-positive initial T cells, memory T cells and dendritic cells (DC) into lymphoidtissue or tumor lesions so as to activate active anti-tumor immune response (such as cancer memory T cells) in the body, thereby significantly improving efficiency and remission rate, as well as reducing recurrence rate after remission.
Owner:浙江启新生物技术有限公司
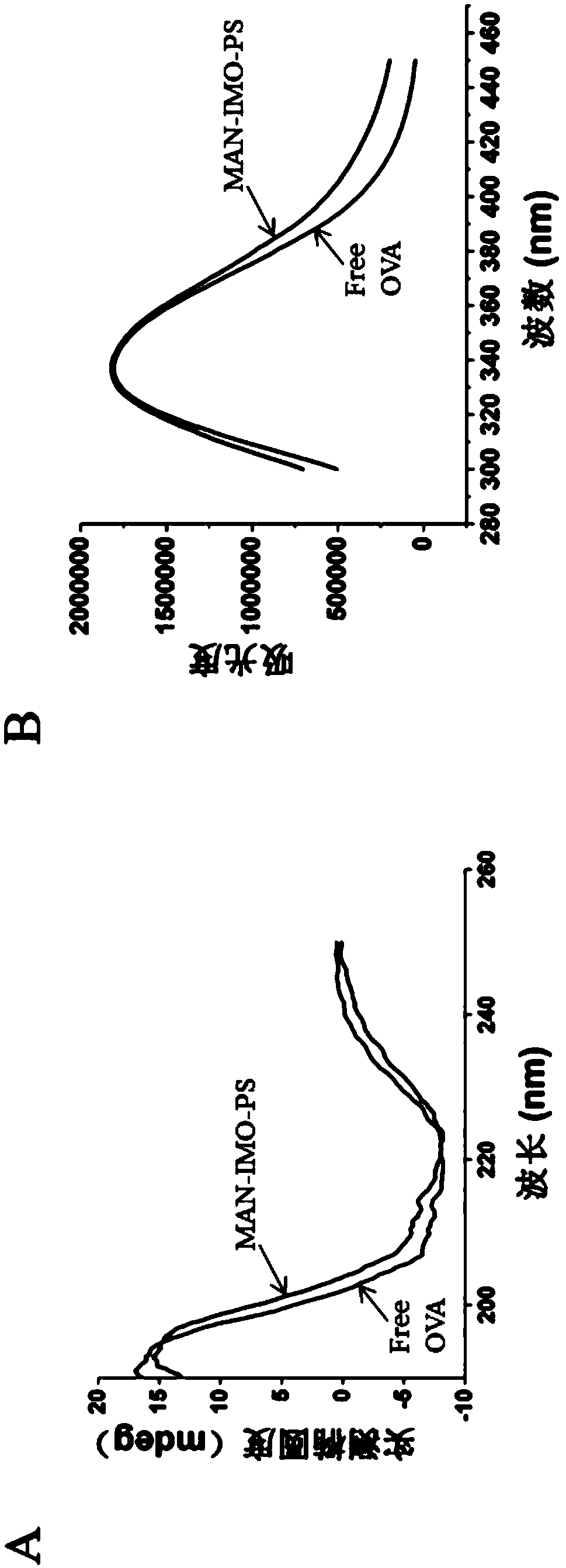
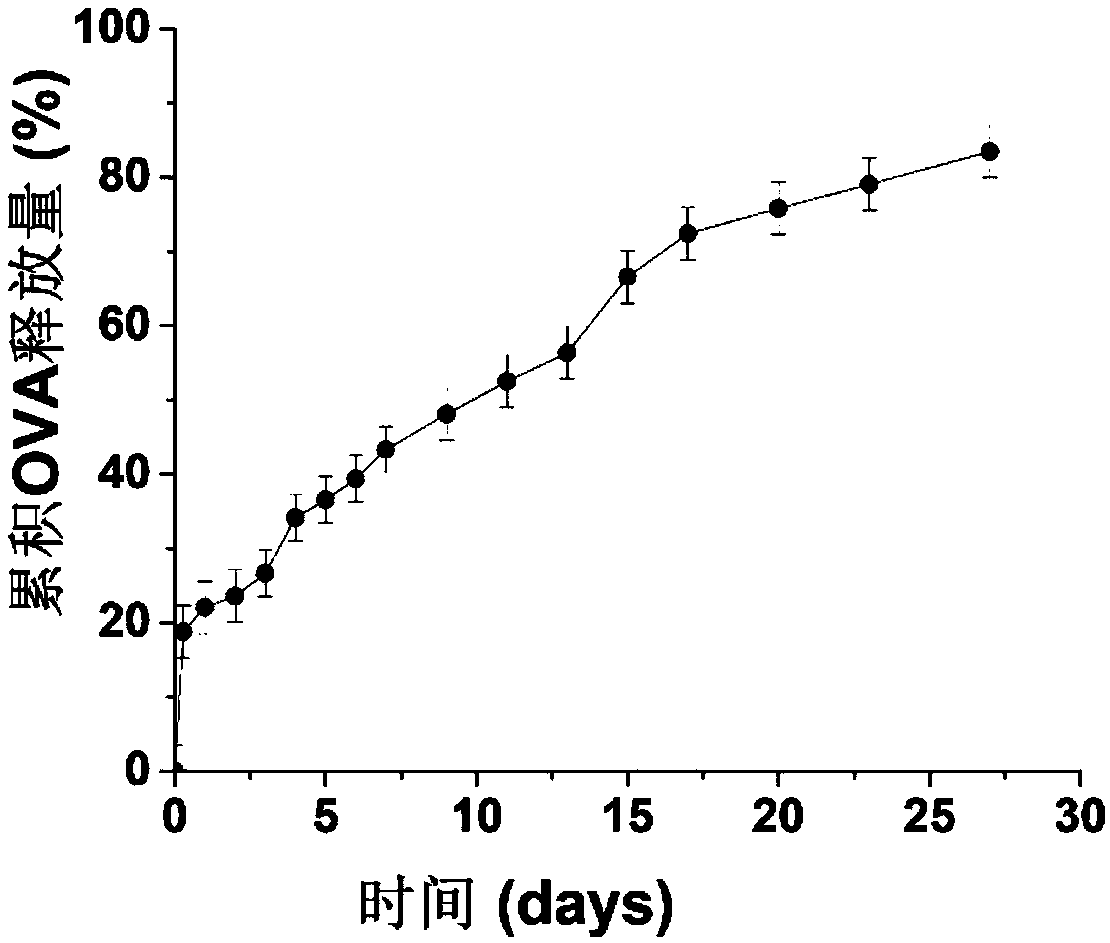
Mannose modified co-loaded antigen and double immuno-agonist phospholipid hybrid polymer vesicle as well as preparation method and application thereof
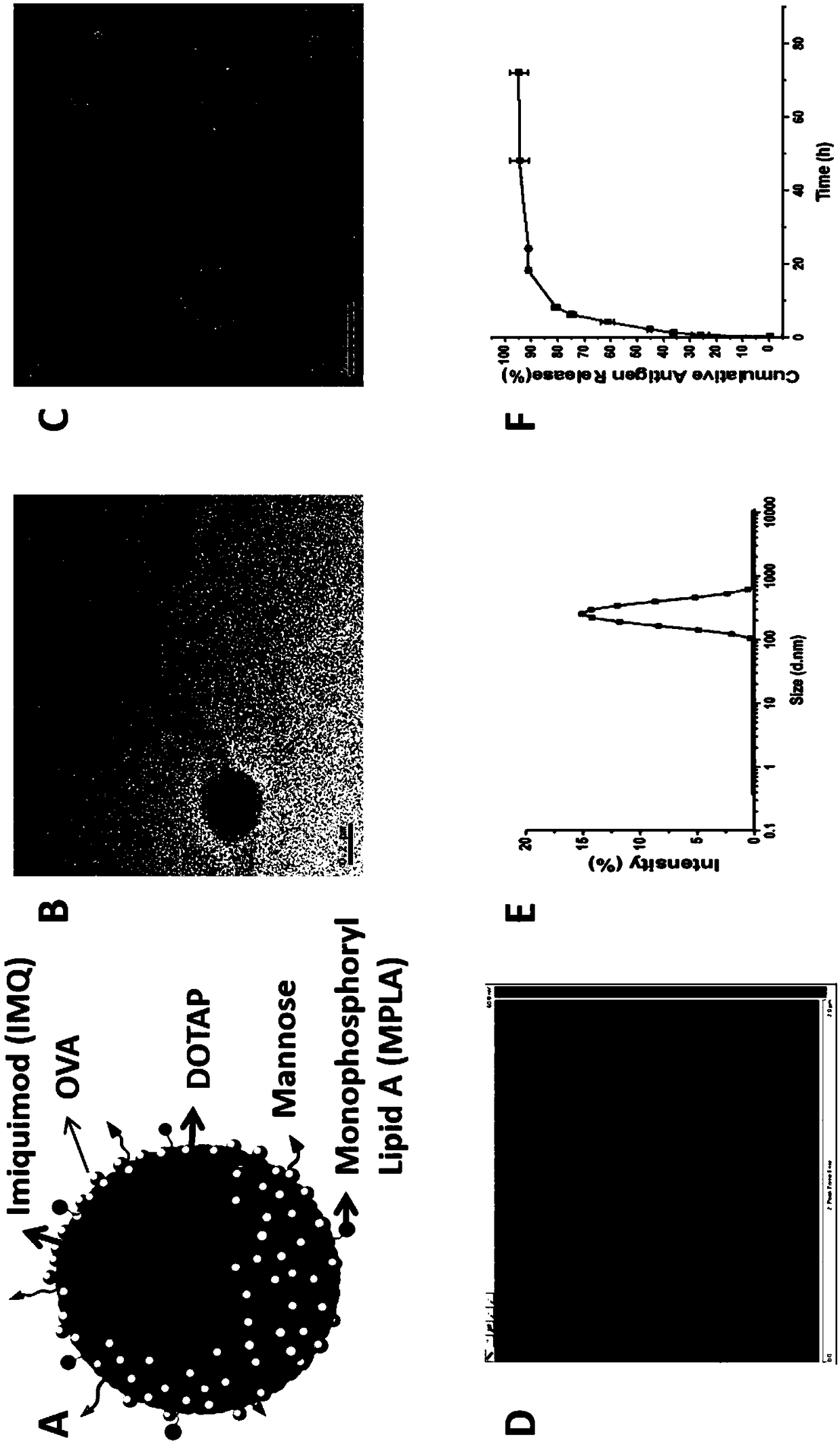
ActiveCN108969771AMaximize Targeting EffectMaximize the effect of targeted immunotherapyAntibacterial agentsBacterial antigen ingredientsAdjuvantBiocompatibility Testing
The invention relates to a mannose modified co-loaded antigen and a double immuno-agonist phospholipid hybrid polymer vesicle as well as a preparation method and application thereof. A vaccine carriertakes an amphiphilic triblock copolymer as material, OVA is loaded in the hydrophilic inner cavity, IMQ is loaded in the hydrophobic membrane layer, DOTAP cationic layer is fitted with MPLA, and cationic lipid outer layer adsorbs outer layer OVA; phospholipid with reactive group is introduced to pass the targeted mannose ligand through covalently linked to the PEG-active distal end of the polymer-loaded vesicle surface, integrating the functions of active targeting of tumors, co-delivery of antigens and adjuvants and the like; the vesicle has the characteristics of small particle size, good dispersion, high antigen loading and good biocompatibility, etc., which can promote antigen uptake, DC activation and maturation, antigen cross-presentation, antigenic lymph node migration, lymphocyteactivation, effector T cell immune response, CD8<+> T and CD4<+> T cell responses, and memory T cells immune response.
Owner:INST OF BIOMEDICAL ENG CHINESE ACAD OF MEDICAL SCI
Detection, localization and staging of tumors using labeled activated lymphocytes directed to a tumor specific epitope
InactiveUS20060171883A1Bind more effectivelyGood effectDisease diagnosisRadioactive preparation formsAbnormal tissue growthCell specific
A Disclosed are methods for detecting and localizing a cell-specific antigen in a mammal, such as a human subject, comprising exposing peripheral blood mononuclear cells (PBMCs) of the mammal to an immunogenic peptide epitope of the antigen, under conditions for antigen-specific activation of T lymphocytes in the PBMCs, thereby producing antigen-specific T lymphocytes that at least bind to the cell-specific antigen. Labeled antigen-specific T lymphocytes are administered to the mammal, typically with-out IL-2, either intraperitoneally or intravenously. The distribution of these cells in the mammal is determined by imaging, thereby detecting and localizing cell-specific antigen in the mammal. Exposing PBMCs to the immunogenic peptide typically involves a cell-free peptide preparation and interleukin-2 (IL-2), but no additional cells such as antigen presenting cells (APC) separately pulsed with antigen. The antigen-specific T lymphocytes typically are cytolytic for cells expressing the cell-specific antigen and may comprise CD4+, CD8+, and / or CD45RO+ memory T cells.
Owner:PHILLIPS CATHERINE A DR +1
Modulated Immunodominance Therapy
InactiveUS20140099341A1Substantial therapeutic benefitAlter immunodominance hierarchyAntipyreticAnalgesicsBiological bodyCellular homeostasis
The invention involves generating a T cell response to subdominant antigens and using the cells to therapeutically change the cellular homeostasis and nature of the immune response. In a preferred embodiment, the cells are generated outside of the patient avoiding the influence of the patient's immunologic milieu. By stimulating and growing the T cells from a patient in a tissue culture to one or more subdominant antigens and the transplanting them into the patient, if enough cells are expanded and transplanted, the transplanted cells overwhelm the endogenous dominant T cells in the response to either break or induce immune tolerance or otherwise modify the immune response to the cells or organism expressing that antigen. When the memory cells are established they are then reflective of this new immunodominance hierarchy so that the desired therapeutic effect is long lasting. In effect, the transplantation exogenously generated T cells reactive to the subdominant antigens is recapitulating priming and rebalancing the patient's immune response to target previously subdominant antigens in the cells or organism to produce a therapeutic benefit.
Owner:GENEIUS BIOTECH
Stem-cell-like memorability T cell in-vitro inducer and method
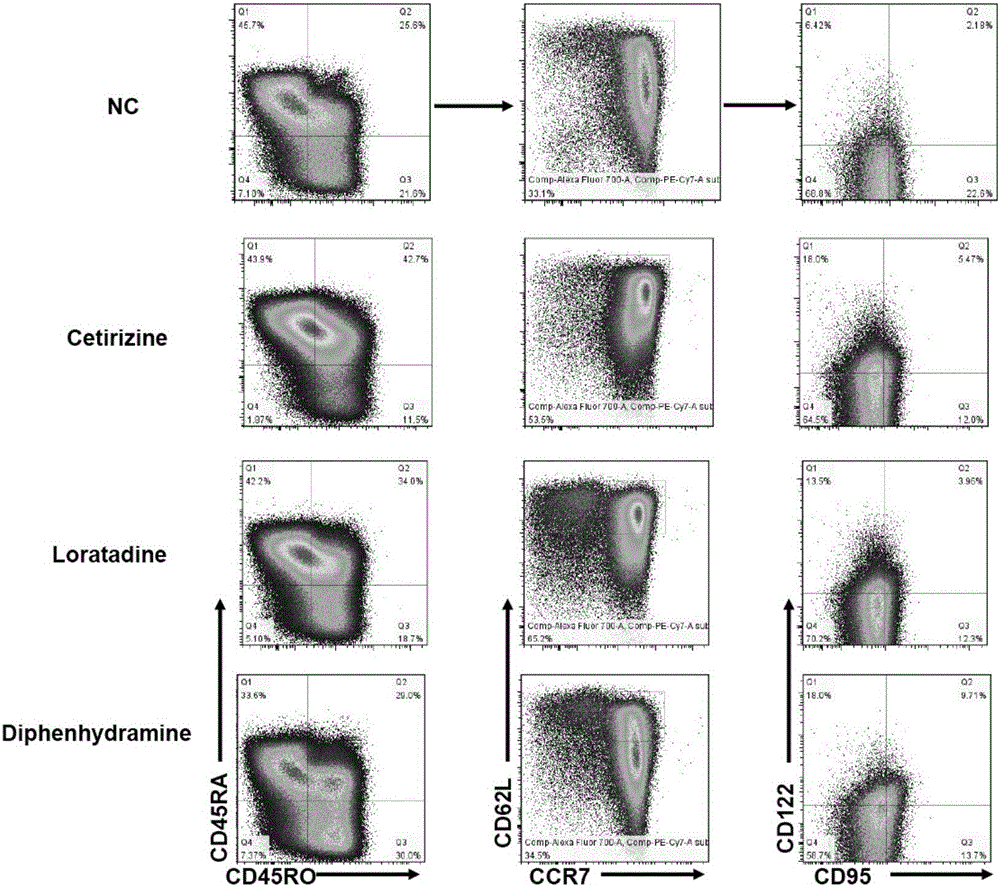
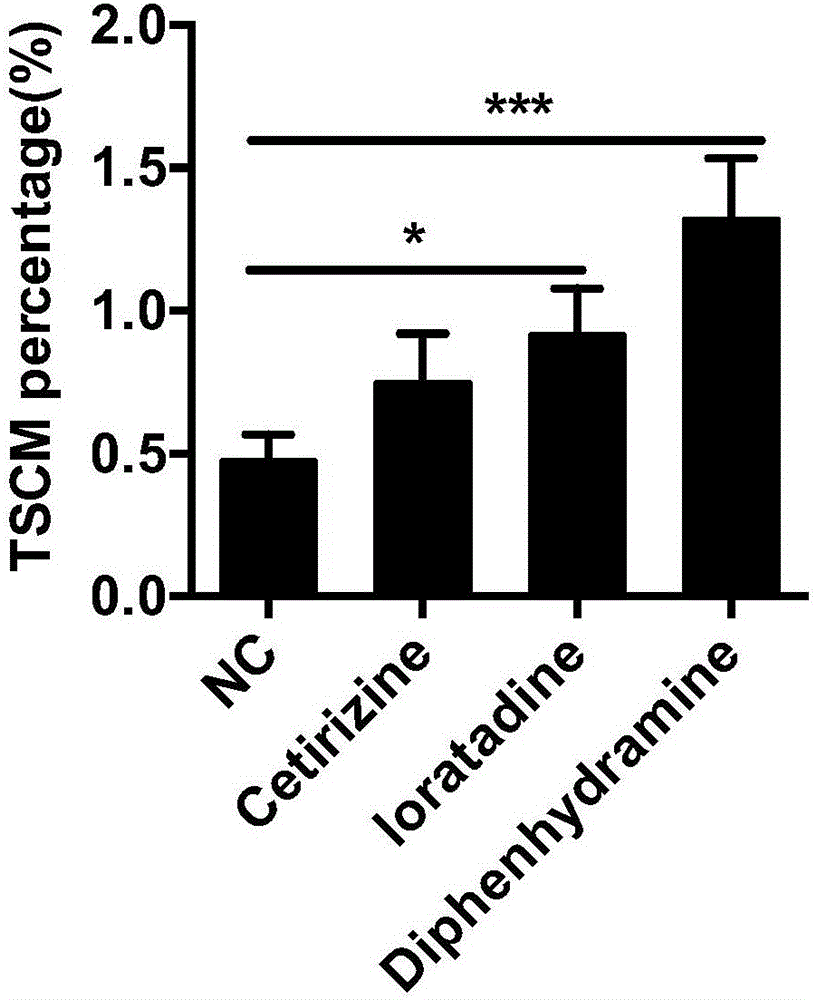
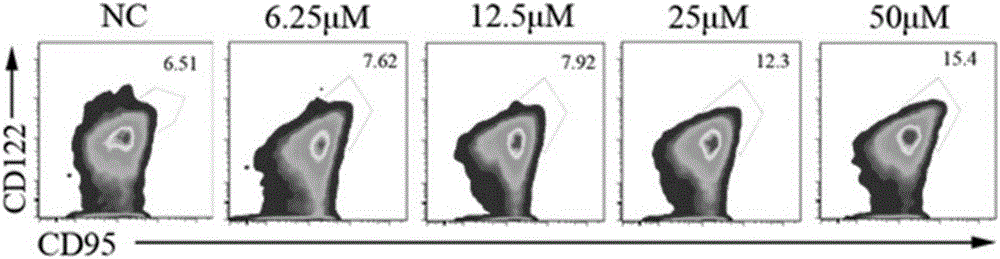
ActiveCN106399243AImprove securityLow costCulture processBlood/immune system cellsHistamine H1 Receptor AntagonistsAbsolute number
The invention discloses a stem-cell-like memorability T cell in-vitro inducer and a method. The inventor finds through experiments that a CD8 initial T cell can be rapidly induced, differentiated and amplified into TSCM cells in vitro if a histamine H1 receptor antagonist is additionally added into a T cell induction medium, the ratio of the TSCM cells to the CD8 cell can be remarkably increased, and the absolute number of the TSCM cells can be remarkably increased. Properties and functions of the induced, differentiated and amplified TSCM cell are maintained. The security of the histamine H1 receptor antagonist to human bodies is already tested through clinical practice, and a long research and development cycle of a new drug can be avoided. Meanwhile, as a developed medicine, the histamine H1 receptor antagonist has the advantages of low cost and good security, the TSCM cells induced, differentiated and amplified with the histamine H1 receptor antagonist can be re-transfused without special treatment, a powerful practice basis can be provided for tumor cell treatment, and significant development values and popularization meanings can be achieved.
Owner:SHENZHEN CITY OF REGENERATION BIOLOGICAL PHARMA TECH CO LTD
Use of common γ chain cytokines for the visualization, isolation and genetic modification of memory T lymphocytes
ActiveUS8999715B2Convenient treatmentRegulate immune responseAntibacterial agentsGenetic therapy composition manufactureMemory T-LymphocyteCentral Memory T-Cell
It is described in vitro methods for expanding, detecting or isolating rare populations of antigen specific memory T cells. It is also described an in vitro method for obtaining a genetically modified memory T cell population. Uses of cells so obtained are also disclosed.
Owner:OSPEDALE SAN RAFFAELE SRL
TCM (central memory T cell) induced by peripheral blood mononuclear cells, and application
InactiveCN109082410ALong-lasting memoryAntibacterial agentsImmunoglobulin superfamilyAbnormal tissue growthClinical efficacy
The invention relates to the technical field of targeted therapy of immune cells, in particular to a TCM (central memory T cell) obtained through induced differentiation of human peripheral blood mononuclear cells. The basis is provided for application of the TCM induced by the frozen peripheral blood mononuclear cells to clinical tumor treatment, the cell can be resuscitated at any time accordingto treatment plans of patients with tumors, and the problems of poor proliferation capability and immune incompetence of T cells of the patients with the tumors are solved; currently, the reinfused immune cells, especially cloning effector T cells, are difficult to survive in the long-term, and the clinical curative effect of the adoptive cell therapy is affected greatly; the TCM has a long-lasting memory, and is transfected through a TCR gene to achieve TCM cell targeting.
Owner:路春光
Anti-WT1/HLA bi-specific antibody
ActiveUS10239952B2Potent therapeutic activityImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsMyeloid leukemiaAntiendomysial antibodies
Disclosed herein is a bi-specific form of a T cell receptor mimic (TCRm) mAb with reactivity to human immune effector cell antigen and a WT1 peptide / HLA-A epitope. This antibody selectively bound to leukemias and solid tumor cells expressing WT1 and HLA-A as well as activated resting human T cells to release interferon-(IFN-γ) and to kill the target cancer cells in vitro. Importantly, the antibody mediated autologous T cell proliferation and directed potent cytotoxicity against fresh ovarian cancer cells. Therapeutic activity in vivo of the antibody was demonstrated in NOD SCID SCID Yc*(NSG) mice with three different human cancers expressing WT1 / HLA-A2 including disseminated Ph+ acute lymphocytic leukemia (ALL), disseminated acute myeloid leukemia, and peritoneal mesothelioma. In both of the leukemia xenograft models, mice that received the antibody and T cells also showed longer survival and delayed limb paralysis. Also provided are methods for stimulating a primary T cell response comprising stimulating cytotoxic T cells against a first tumor antigen and a secondary T cell response comprising stimulating effector T cells and / or memory T cells against a first tumor antigen and / or against a second tumor antigen using the bi-specific antibodies described herein.
Owner:EUREKA THERAPEUTICS INC +1
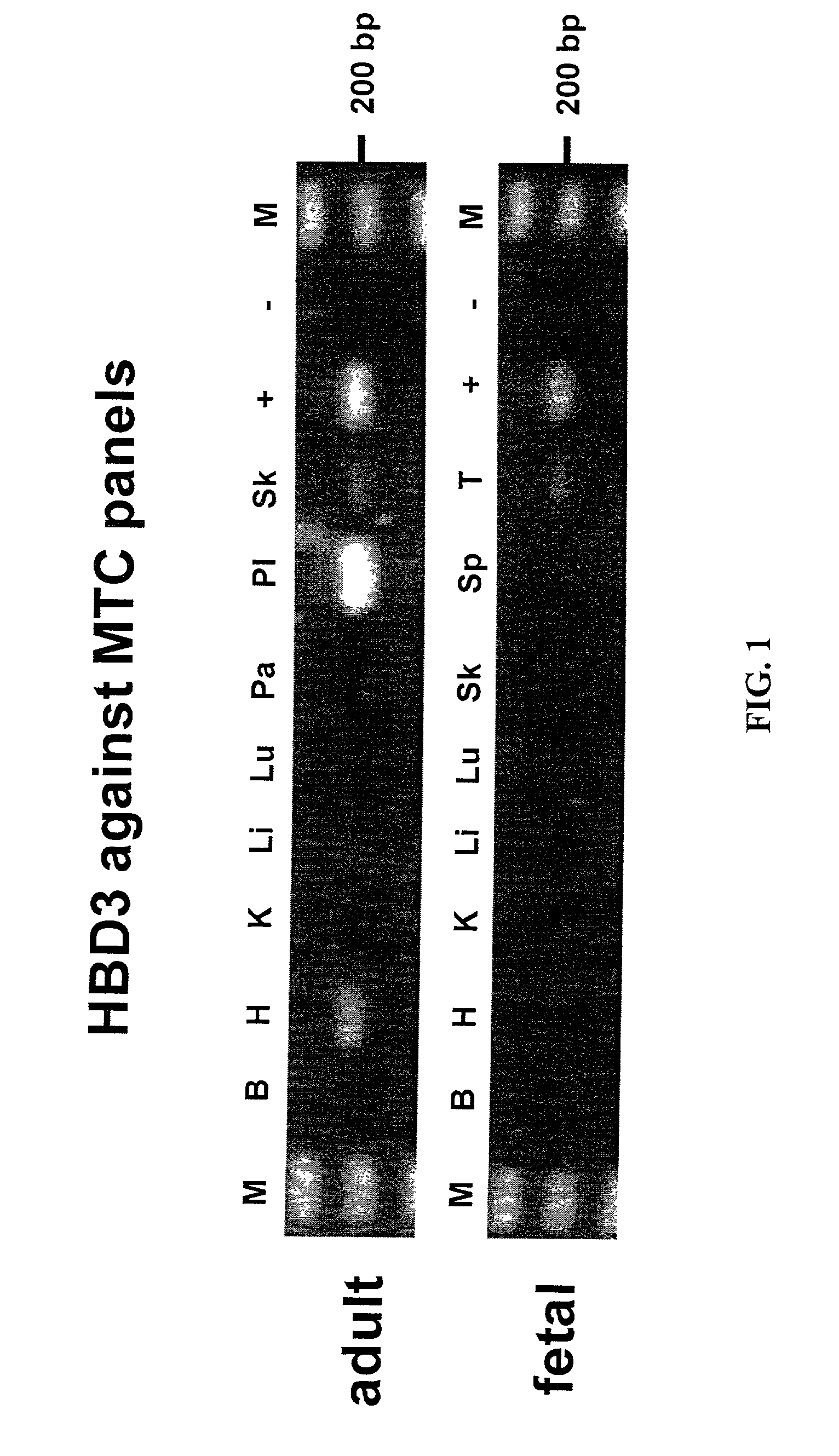
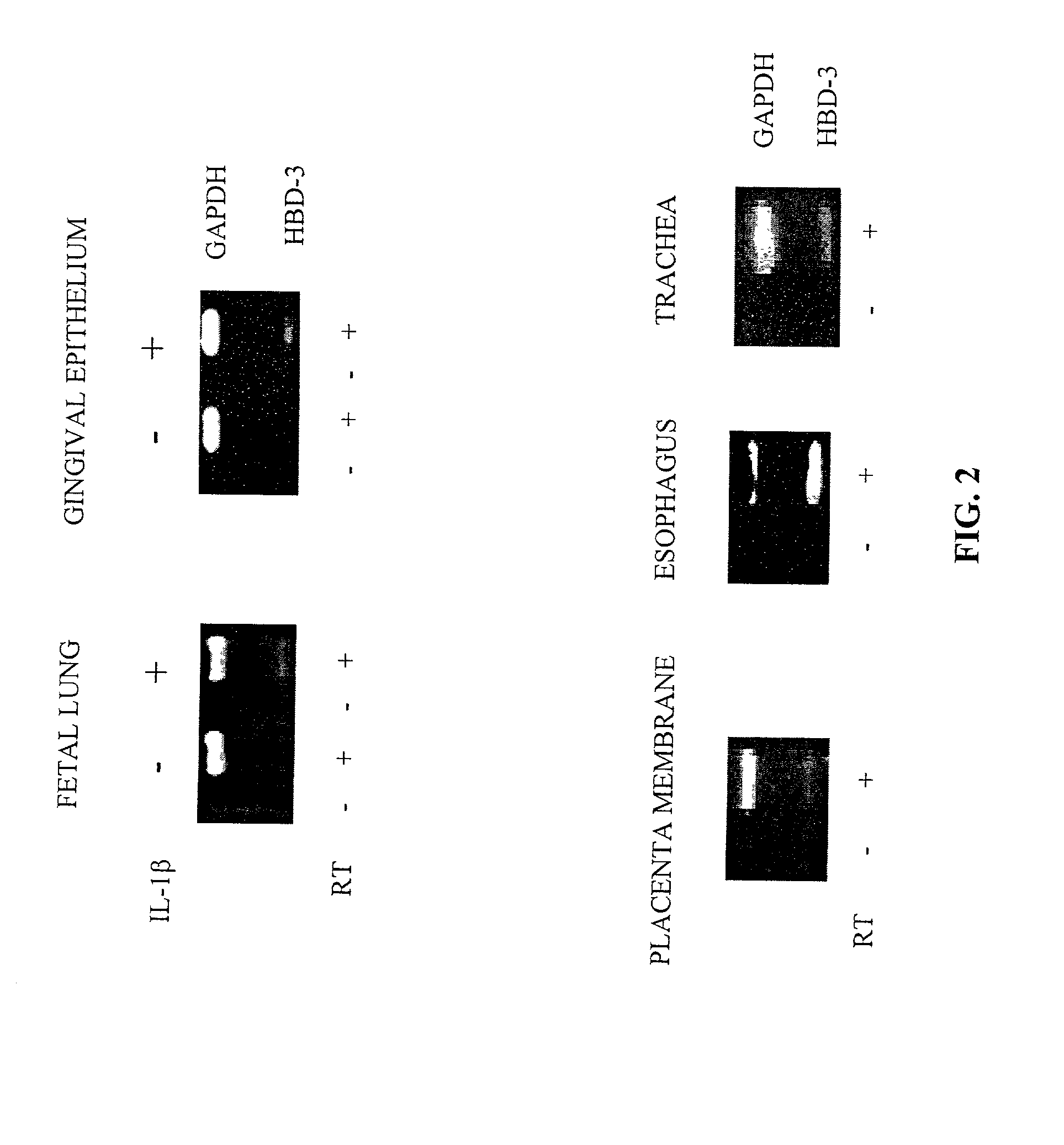
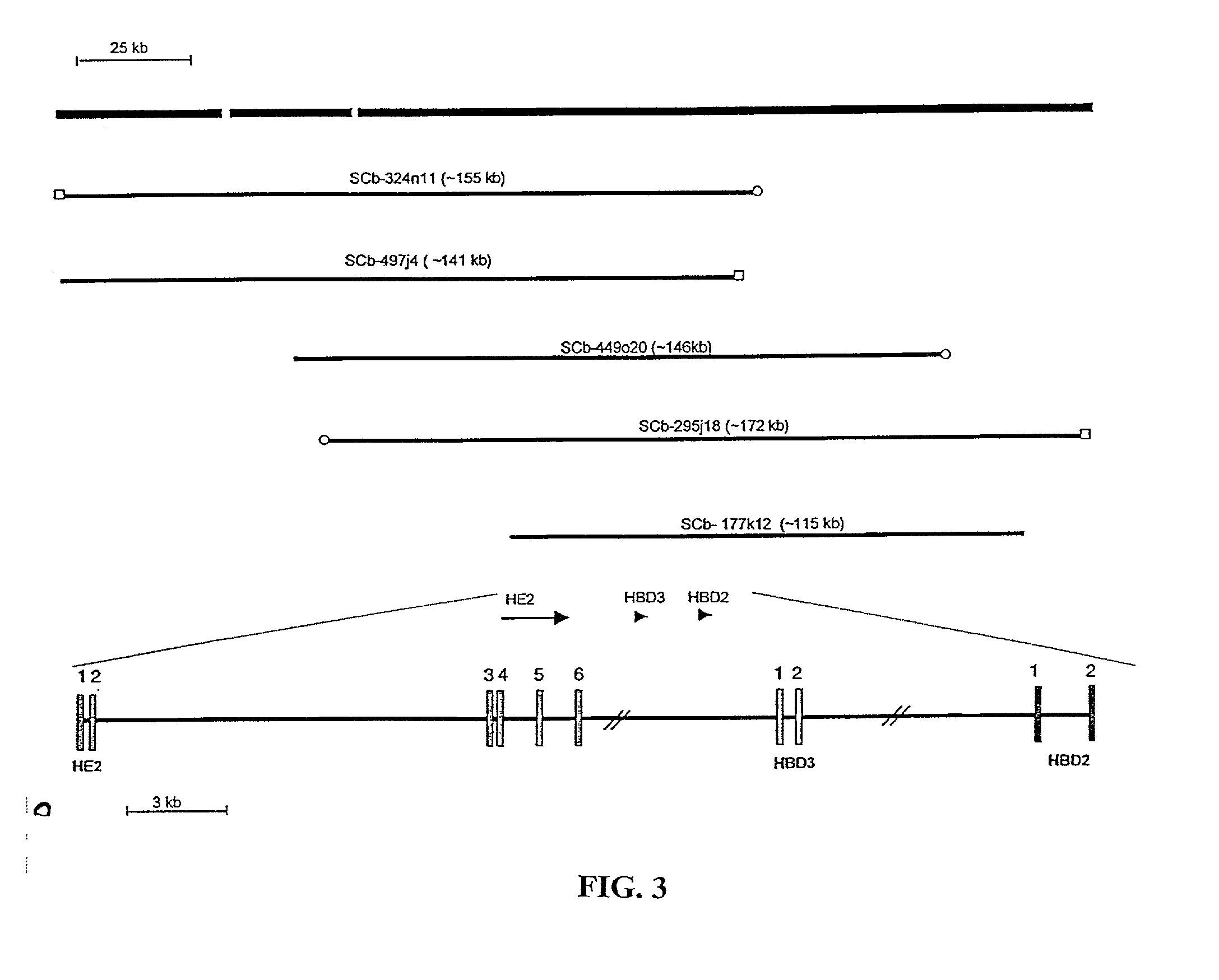
Human beta-defensin-3 (HBD-3), a highly cationic beta-defensin antimicrobial peptide
The present invention relates a novel antimicrobial peptide HBD-3 and derivatives thereof as well as the gene encoding the peptide. The invention further relates to methods of use of the HBD-3 peptide including a method of inhibiting microbial growth by administering an effective amount of the HBD-3 peptide alone or in combinination with other antimicrobial agents or antibiotics. In addition, the immunomodulatory properties of the HBD-3 peptide also facilitate the manipulation of the immune response, i.e., as a chemoattractant for immature dentritic cells or memory T cells.
Owner:UNIV OF IOWA RES FOUND
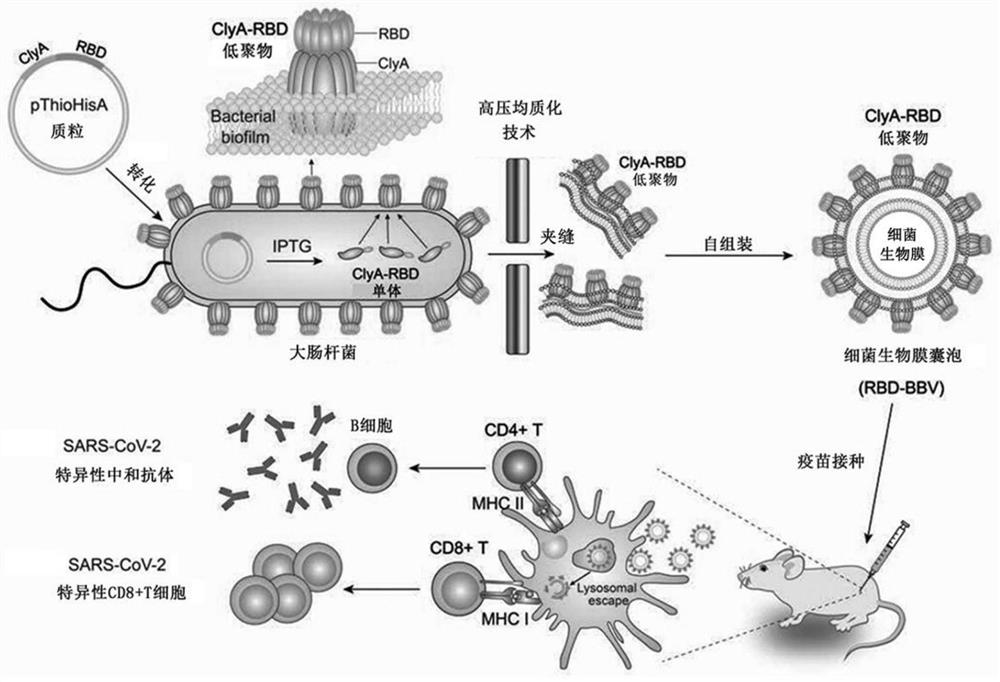
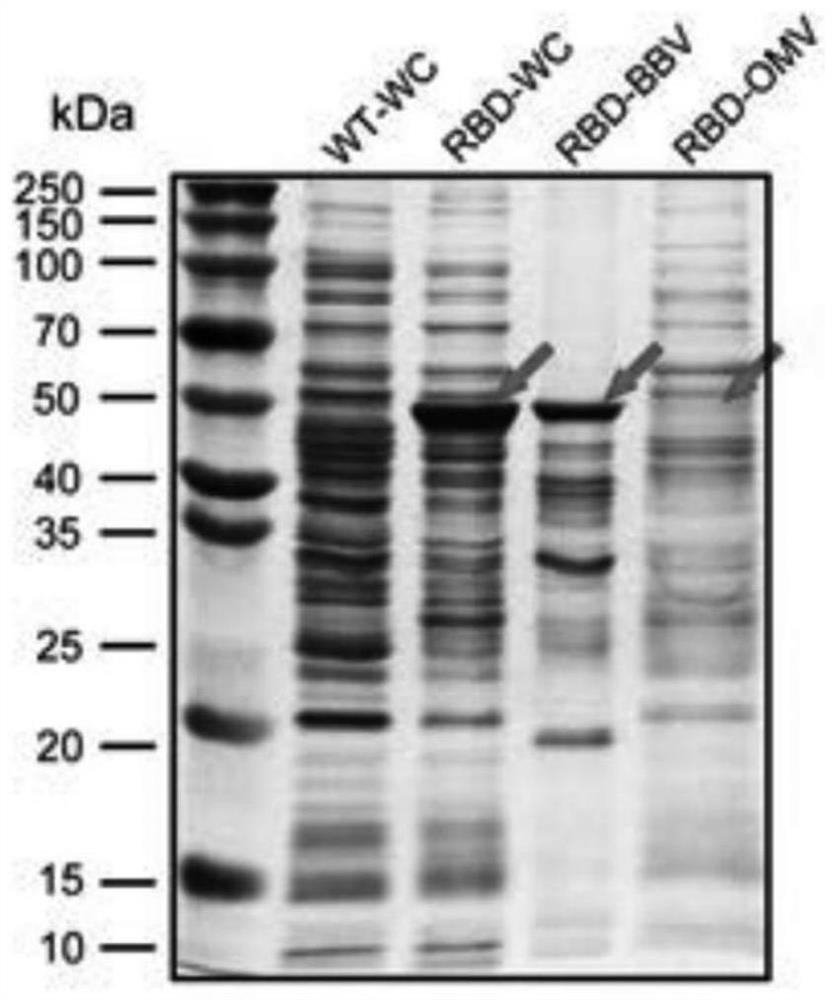
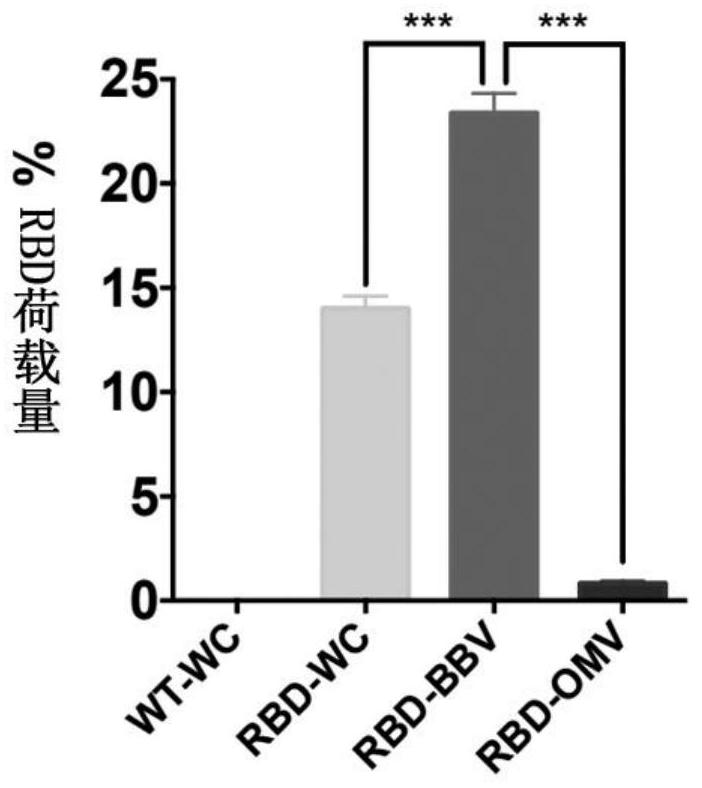
Construction method and application of bacterial biofilm vesicles (BBV) as vaccine vectors
ActiveCN112662695AIncrease productionIncrease intakeBacteriaMicroorganism based processesAntigenBiofilm
The invention provides a construction method and application of bacterial biofilm vesicles (BBV) as a vaccine vector, an engineering modified bacterial biofilm is driven to complete high-efficiency self-assembly by utilizing ultrahigh pressure for the first time, the ability of ClyA to assemble pores on a bacterial outer film is used for carrying RBD to construct burred bacterial vesicles (RBD-BBV) in a prokaryotic system, and the RBD-BBV efficiently exposes the correctly folded RBD on the surface of the vesicles and shows the burred vesicle structure. The RBD-BBV can be efficiently taken up by DC cells and stimulates the maturation of DC cells, and meanwhile, the bacterial biofilm mediates the lysosome escape of the RBD. After RBD-BBV immunization of mice, SARS-COV-2 specific neutralizing antibodies and SARS-COV-2 specific CD4+ T and CD8+ T cell responses are induced in the mice. The RBD-BBV also enhances the local stability of the antigen in vivo and induces the generation of memory T cells. The RBDBBV vaccine form disclosed by the invention has the unique characteristics that many other vaccine forms cannot be realized, and a new thought can be possibly provided for the development of SARSCoV2 vaccines.
Owner:INST OF MEDICAL BIOLOGY CHINESE ACAD OF MEDICAL SCI
Generation and use in adoptive immunotherapy of stem cell-like memory t cells
ActiveUS20200009189A1Significant cytotoxicityAntibacterial agentsAntimycoticsCentral Memory T-CellHuman patient
Provided herein are methods of generating antigen-specific T cells for therapeutic administration to a human patient having or suspected of having a pathogen or cancer, utilizing stem cell-like memory T cells (TSCM cells). Also disclosed are antigen-specific T cells generated by such methods, and methods of treating a human patient using such antigen-specific T cells.
Owner:MEMORIAL SLOAN KETTERING CANCER CENT
Central memory T cell body and in-vitro culture method
ActiveCN108165529ARaise the ratioIncrease lethalityBlood/immune system cellsCell culture active agentsCentral Memory T-CellCentral Memory T-Lymphocyte
The invention discloses an in-vitro culture method of a central memory T lymphocyte. The method comprises the following steps: adding IL-21 into an initial medium, and allowing the oxygen concentration in a culture environment to be 1 + / - 0.1%; and adding the IL-21 and rapamycin into an amplification medium when the culturing is performed for 7 days. The in-vitro culture method using the combination of the rapamycin, interleukin 21 and low-oxygen environment in a proper culture process to improve the proportion of the central memory T cell, inhibit the differentiation of the central memory T cell into effector cells and enhance the killing effect on tumor cells is disclosed for the first time.
Owner:BEIHAO STEM CELL & REGENERATIVE MEDICINE RES INST CO LTD
Peripheral blood memory T cell culture method
ActiveCN110093315AEfficient amplificationRaise the ratioBlood/immune system cellsCell culture active agentsAntigenCytokine
The invention relates to a peripheral blood memory T cell culture method. The method comprises: a) magnetically separating the PBMC isolated from the peripheral blood to obtain CD8<+> T cells; and inoculating the CD8<+> T cells in a culture container coated with a CD3 antibody and a CD28 antibody for culture; wherein a culture system contains autologous plasma, and the cytokine mainly includes IFN-gamma and IL-2; b) changing liquid for amplification culture, wherein a novel culture system contains autologous plasma and the cytokines mainly include IL-2, IL-1a, IL-7, and IL-15; and c) co-culturing the expanded T cells with DC cells; wherein the DC cells are previously co-cultured with an autologous tumor antigen. The method can effectively amplify memory T cells in vitro and effectively increase the proportion of active memory T cells.
Owner:新疆西部赛澳生物科技有限责任公司
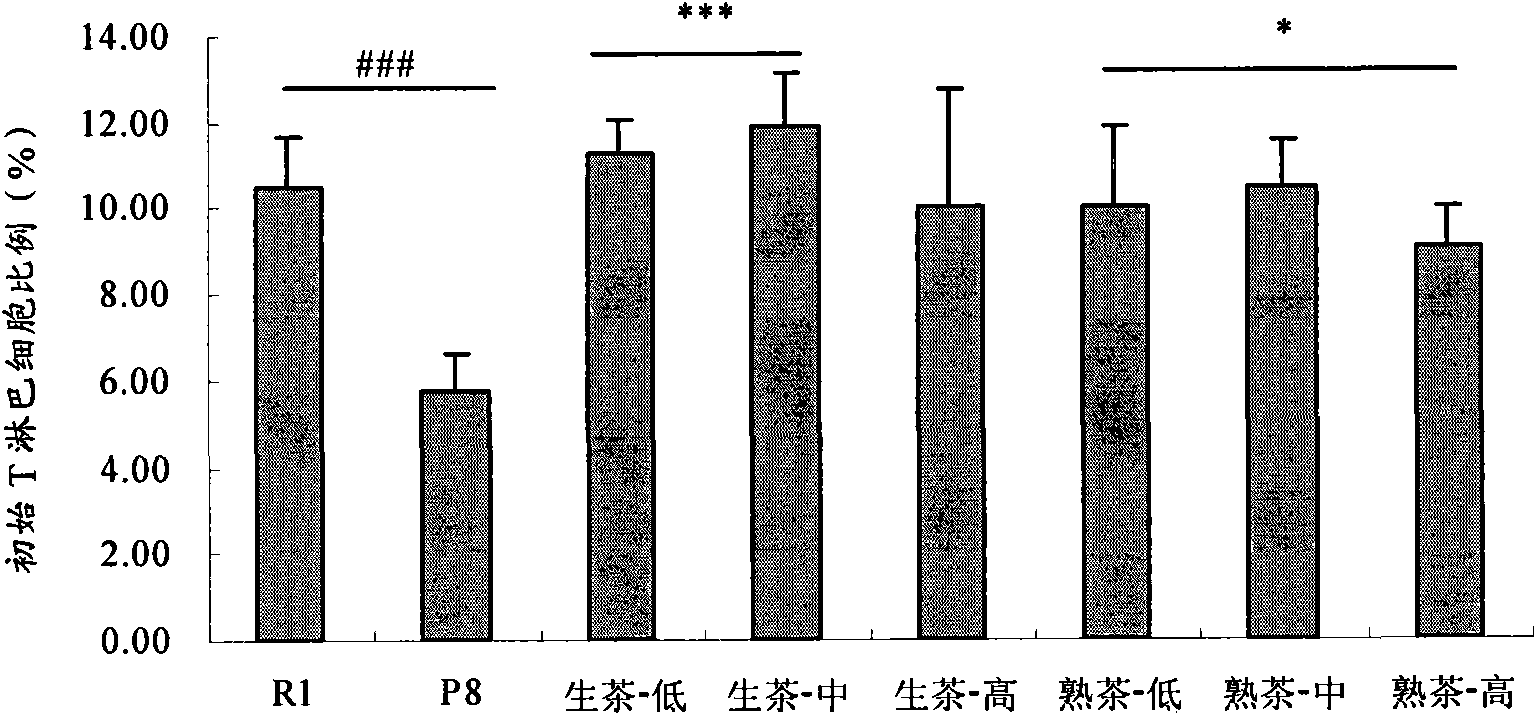
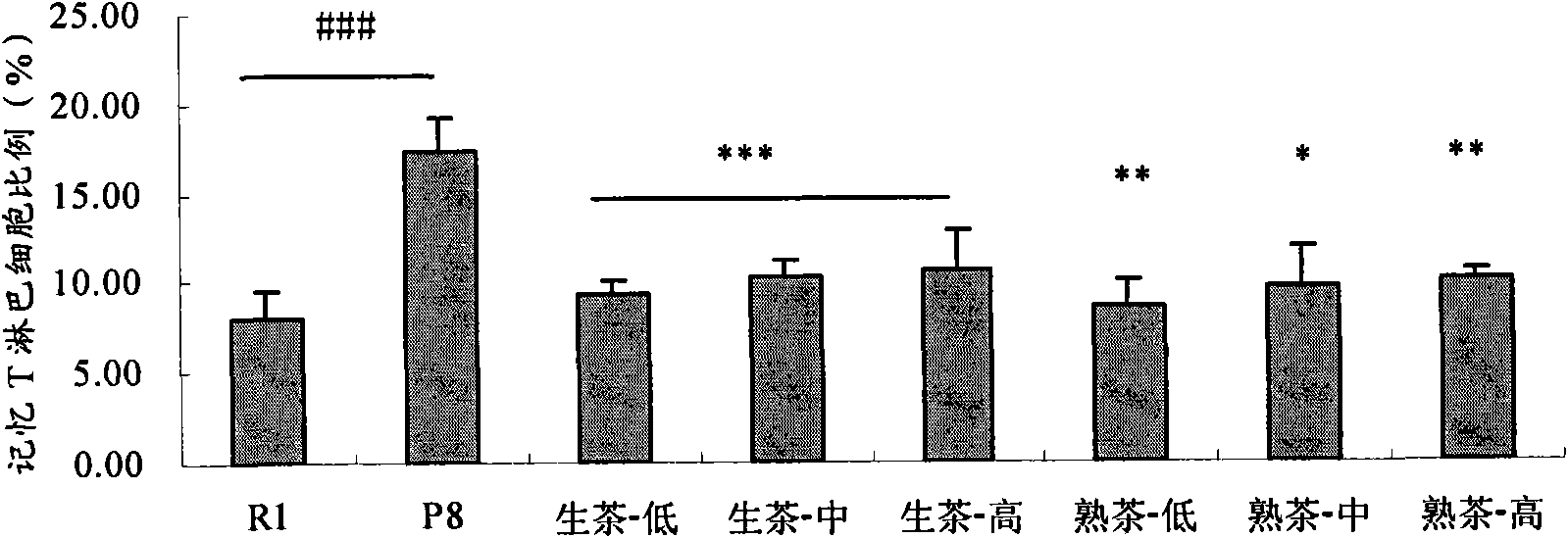
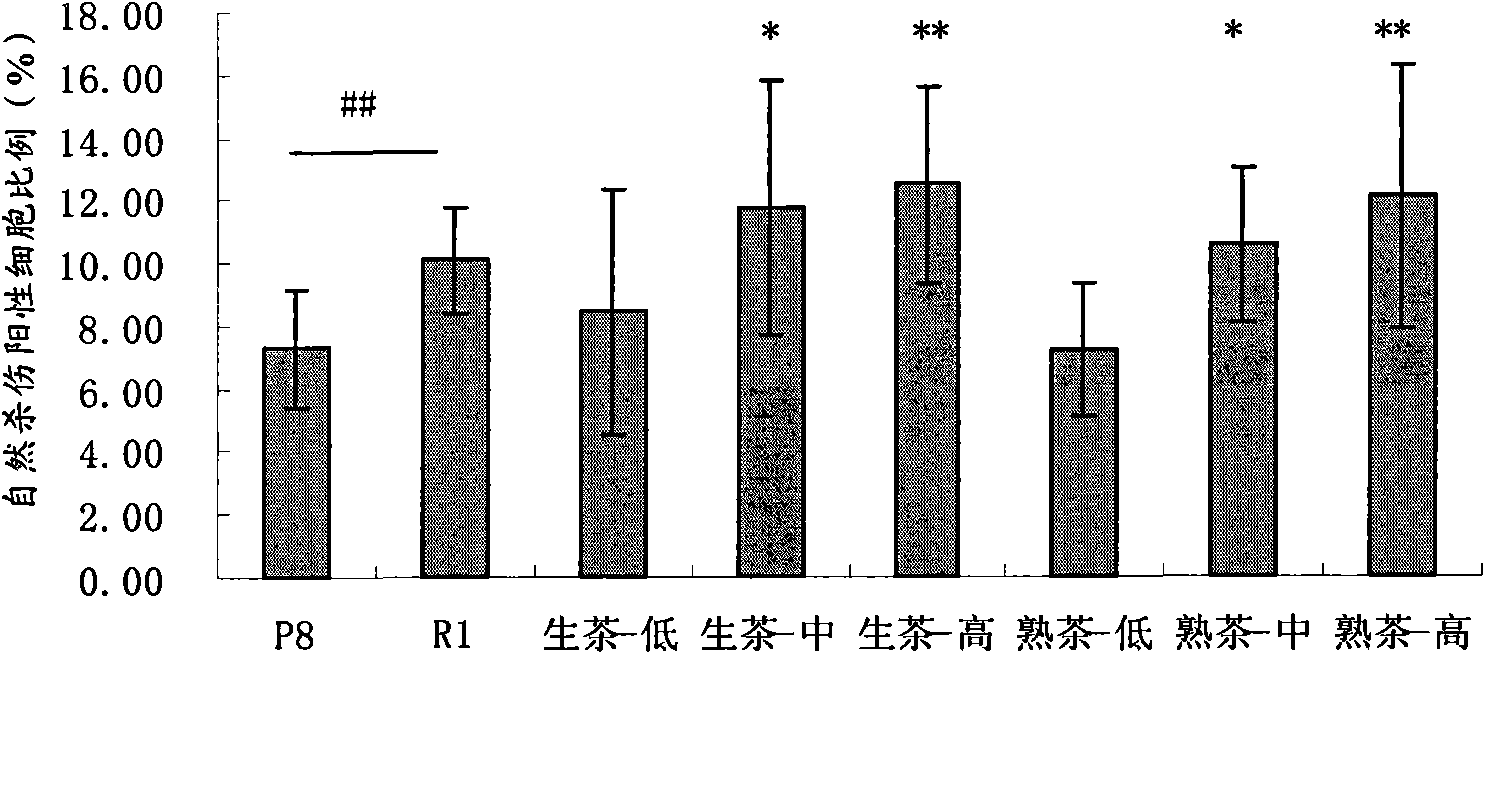
Application of Puer tea in preparing drugs for resisting immunosenescence or other relevant diseases
InactiveCN101856434AEasy to solveGood for healthAntinoxious agentsPlant ingredientsDiseaseNatural Killer Cell Inhibitory Receptors
The invention provides an application of Puer tea in preparing drugs for resisting immunosenescence or other relevant diseases. The invention makes research and analysis on the anti-immunosenescence fuction and the theory thereof of the Puer tea, proves that the Puer tea or an extractive thereof has dual functions of enhancing recessive immunity and restraining excessive inflammatory reaction of an aged organism under the action of reversing aged immunocyte ( reducing memorability T cells and increasing NK cells), realizes the purpose of preventing immunosenescence, expands the application range of the Puer tea or the extractive thereof, and provides a drug or health care product for effectivly preventing and / or treating the immunosenescence or the other relevant diseases.
Owner:马治中 +2
Method for producing lymphocyte cell group consisting mainly of memory t cells
ActiveCN104204195AMaster proliferationGrasp the status of shareMammal material medical ingredientsAntiviralsCentral Memory T-CellLymphocyte
Approximately 50% of lymphocytic cells derived from peripheral blood is occupied by naive T cells before an activation culture is performed, but the ratio decreases through the activation culture. The occupancy of the memory T cells, which is less than half before the start of the culture, is increased by an activation culture to reach 95%, but decreases if the culture continues. An object of the present invention is to prepare CD45RO positive memory T cell groups in quantity without having to use an antibody or the like, conveniently, and rapidly, and to provide a pharmaceutical composition consisting mainly of the memory T cell groups prepared by the method. According to the present invention, harvested lymphocytic cells are activation-cultured, and the occupancy of the memory T cells in the lymphocyte cell group reaching a desired value is detected through the index of the density of the cultured lymphocyte cell group exceeding a predetermined threshold, the related cell group is harvested for free preservation, and the related free-preserved cell is thawed, cultured, and grown so as to produce the lymphocyte cell groups consisting mainly of the memory T cells in quantity.
Owner:LYMPHOTEC
Method for inducing early t memory response with short peptides Anti-tumor vaccine
ActiveUS20180169200A1Large capacityEffective immunityCancer antigen ingredientsAntigen/adjuvant combination ingredientsPeptide TWhite blood cell
The present invention relates to a therapeutic peptide T specific immune therapy for use in the treatment of a cancer of an HLA-A2 (Human Leukocyte Antigen A2) positive patient, said treatment comprises a priming period consisting in two to three administrations of said therapeutic peptide T specific immune therapy, thereby inducing a memory T cell response.
Owner:OSE IMMUNOTHERAPEUTICS
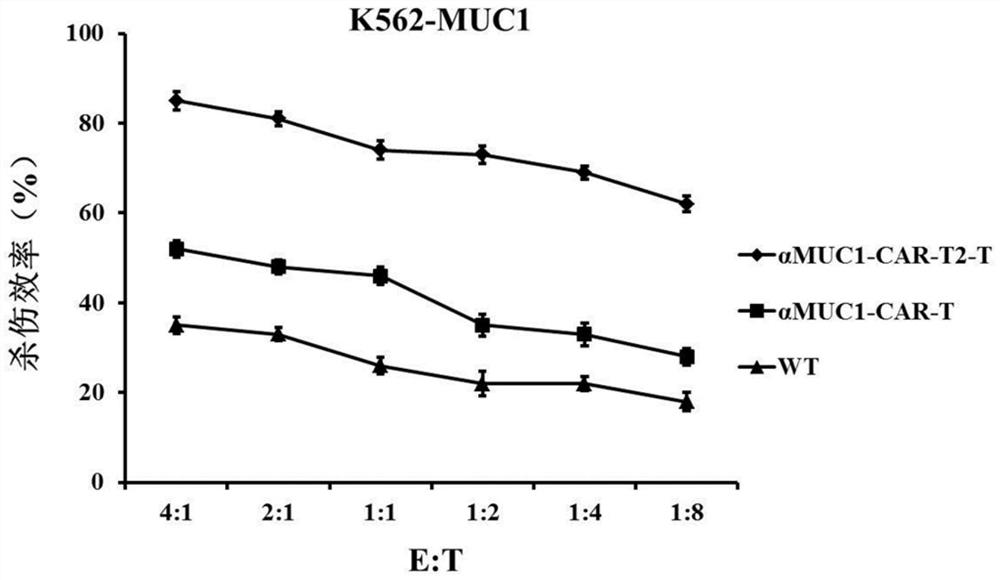
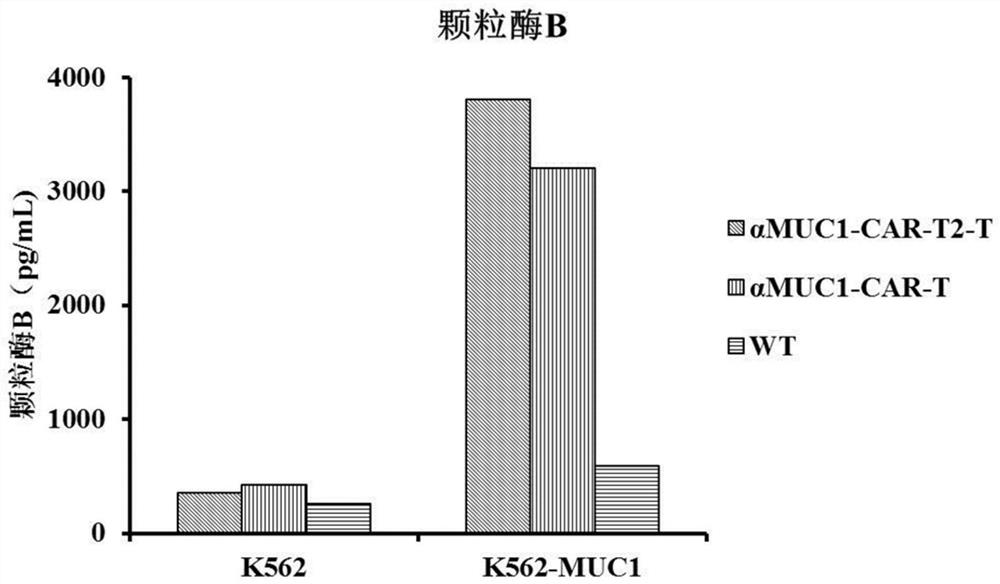
Enhanced MUC1 targeting chimeric antigen receptor and application thereof
InactiveCN111875712AStrong targetingEfficient activationPolypeptide with localisation/targeting motifImmunoglobulin superfamilyAntiendomysial antibodiesRegulatory T cell
The invention provides an enhanced MUC1 targeting chimeric antigen receptor and an application thereof. The chimeric antigen receptor comprises an antigen binding structural domain, a transmembrane structural domain and a signal transduction structural domain, wherein the antigen binding structural domain comprises an anti-MUC1 single-chain antibody and the signal transduction structural domain comprises 4-1BB, CD3 [zeta] and TLR2. The chimeric antigen receptor disclosed by the invention not only has a targeting effect on MUC1 positive tumor cells, but also can efficiently activate T cells, eliminate the immunosuppressive effect of regulatory T cells and promote the formation of memory T cells, and the constructed T cells for expressing the chimeric antigen receptor have remarkably enhanced tumor cell killing capacity.
Owner:GUANGDONG ZHAOTAI INVIVO BIOMEDICINE CO LTD