Patents
Literature
442results about How to "Shorten the time to market" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
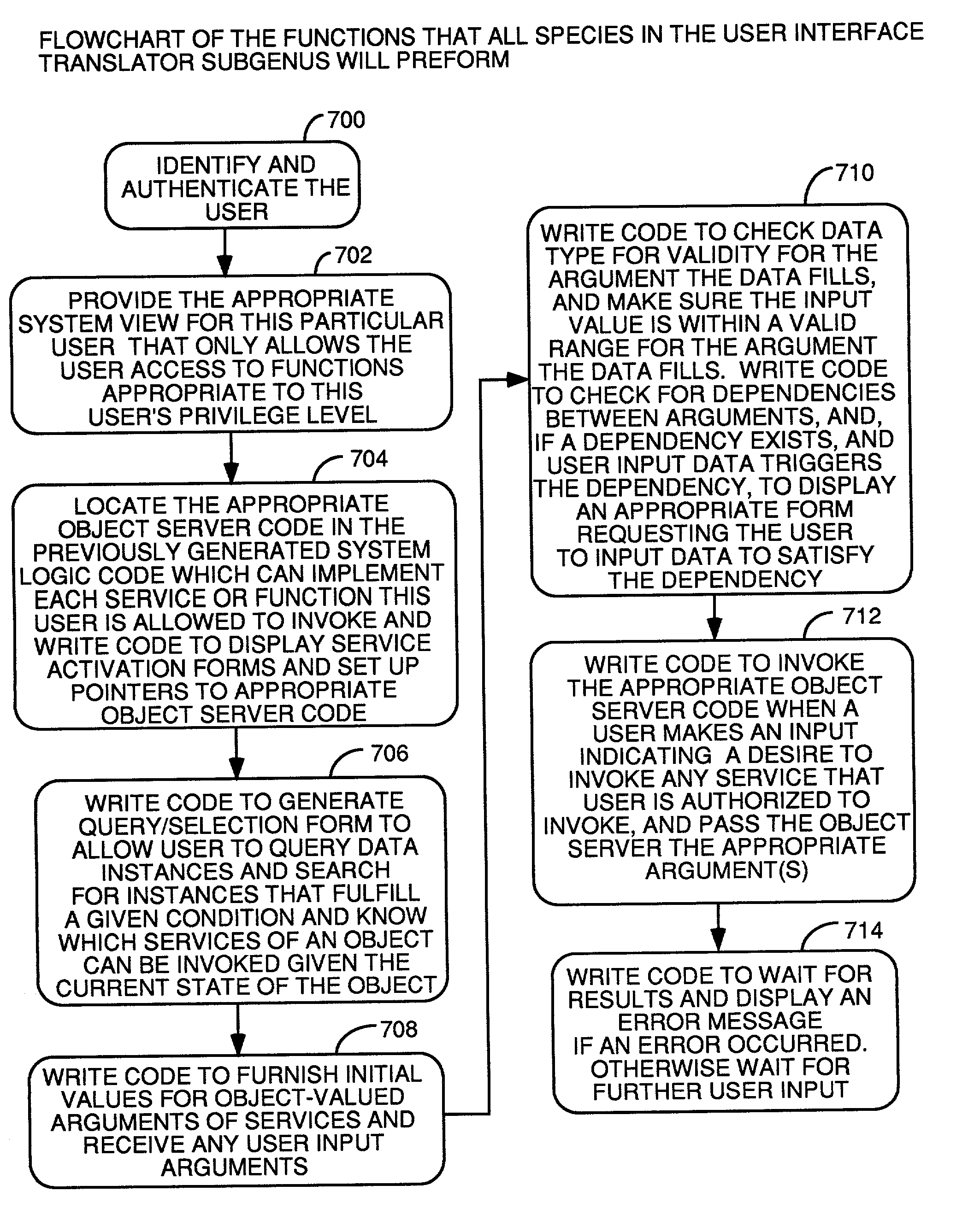
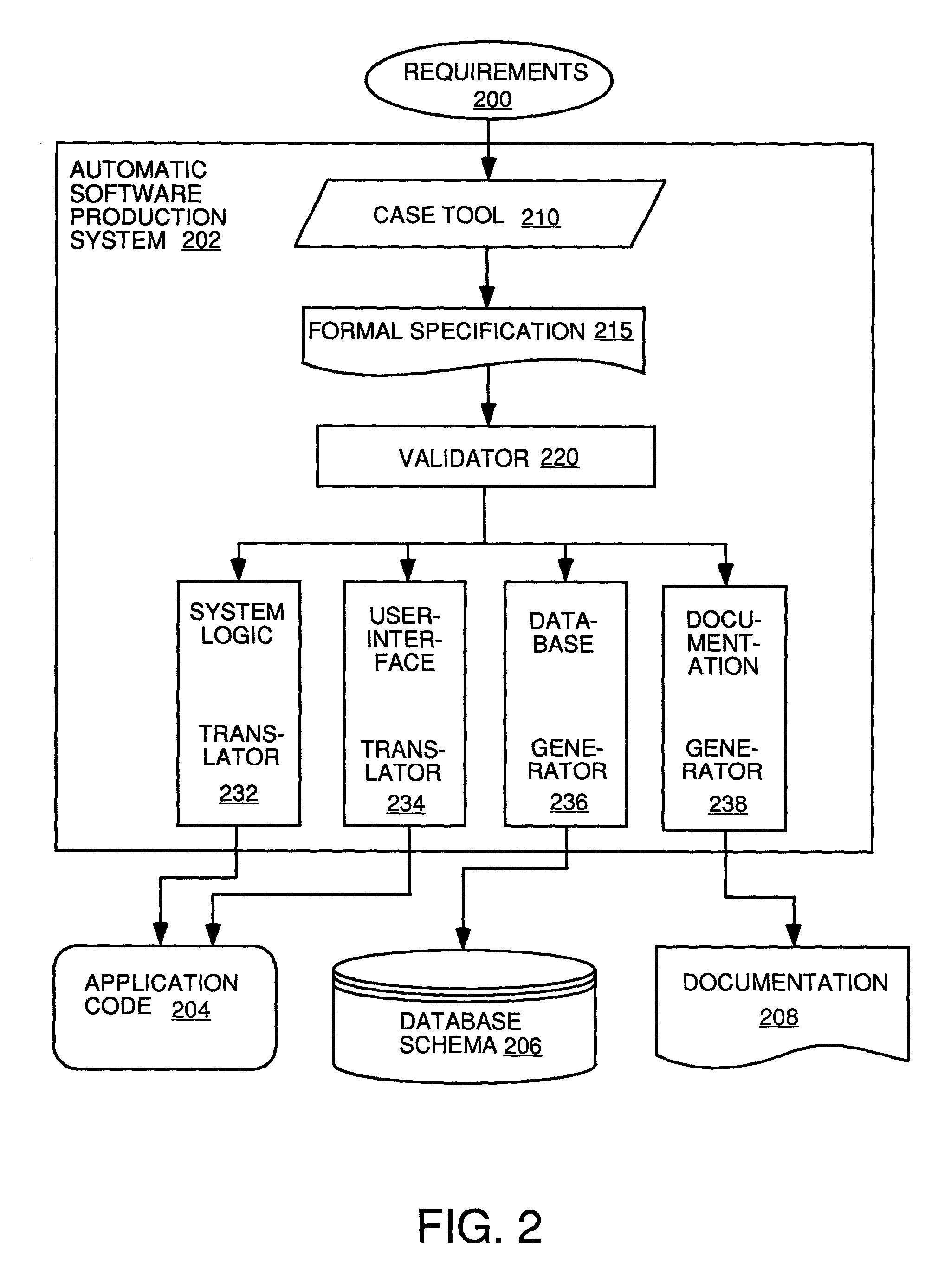
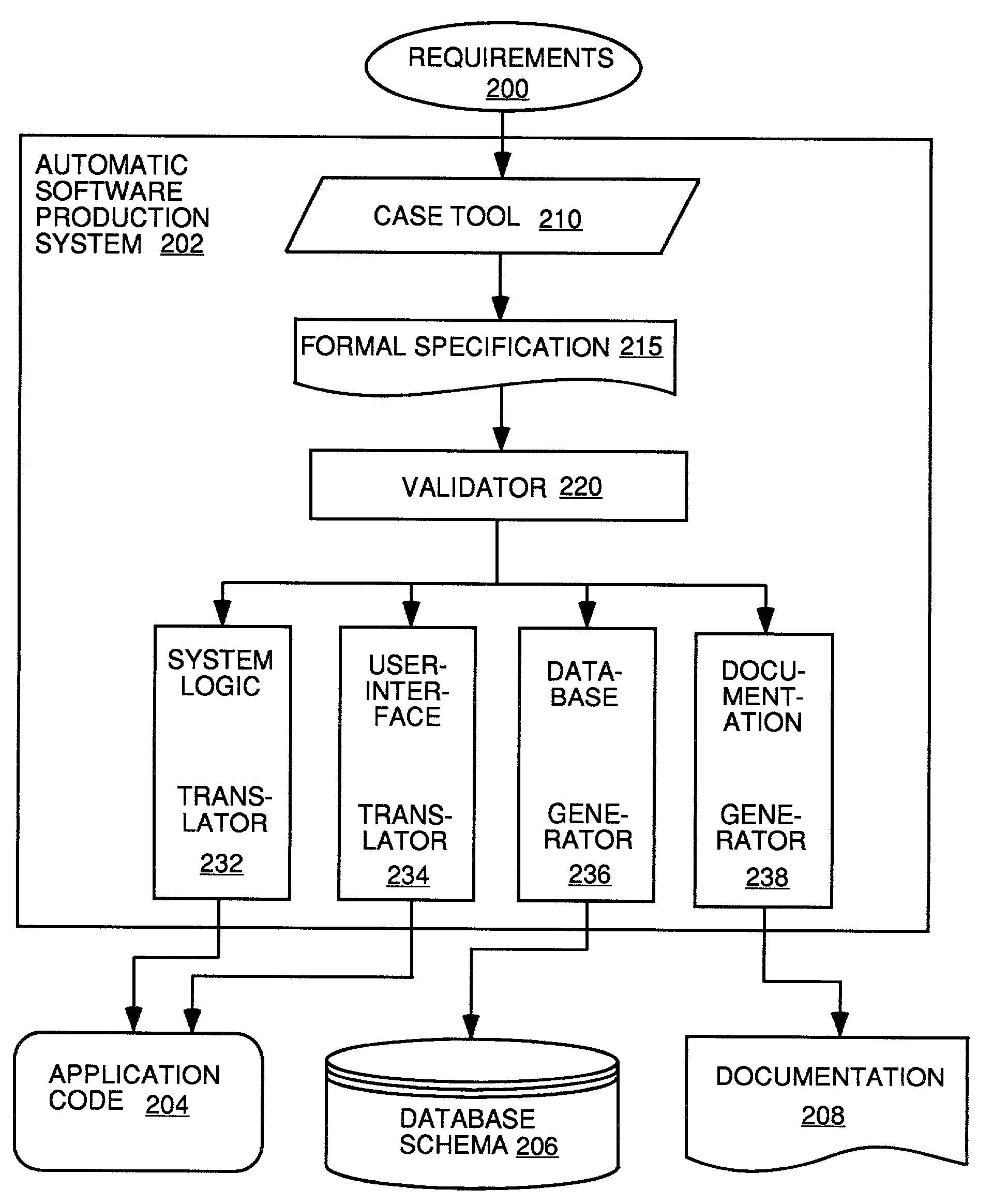
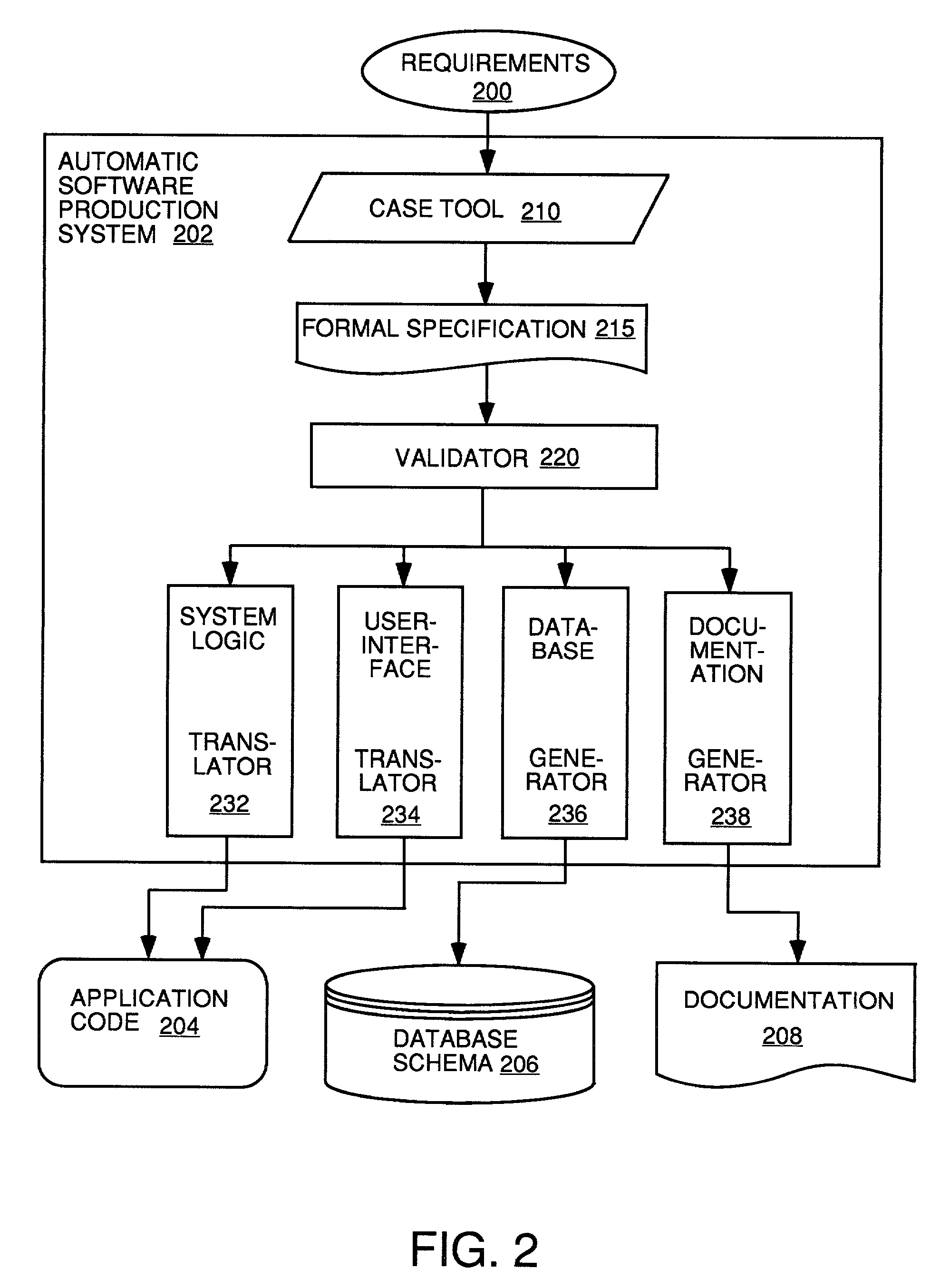
Automatic software production system
InactiveUS7137100B2Shorten the time to marketEasy to useDigital data information retrievalProgram documentationHandling CodeSystem requirements
Owner:SOSY
Software development tool for embedded computer systems
InactiveUS20030135842A1Shorten development timeShorten the time to marketVersion controlSpecific program execution arrangementsModularityApplication software
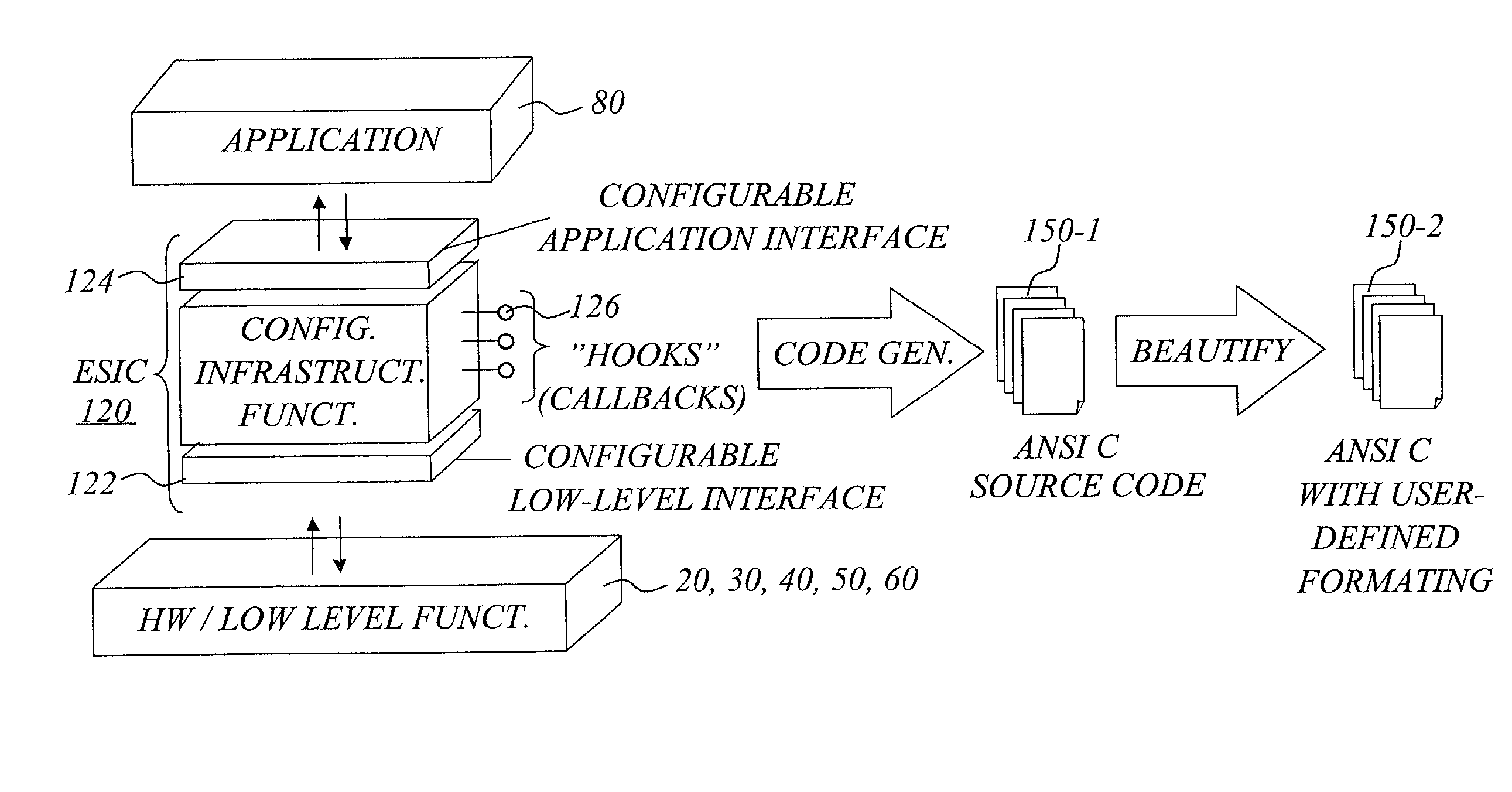
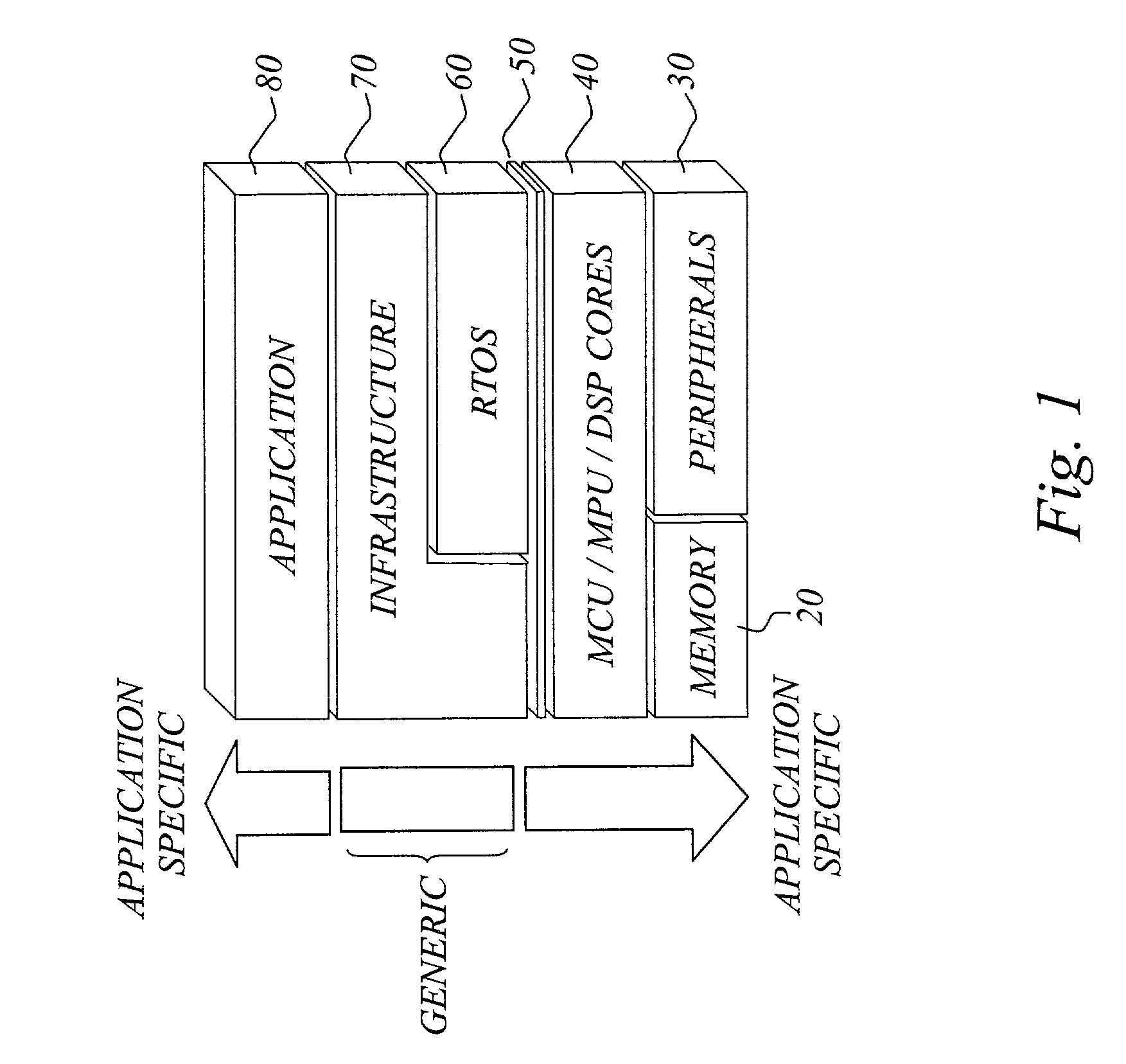
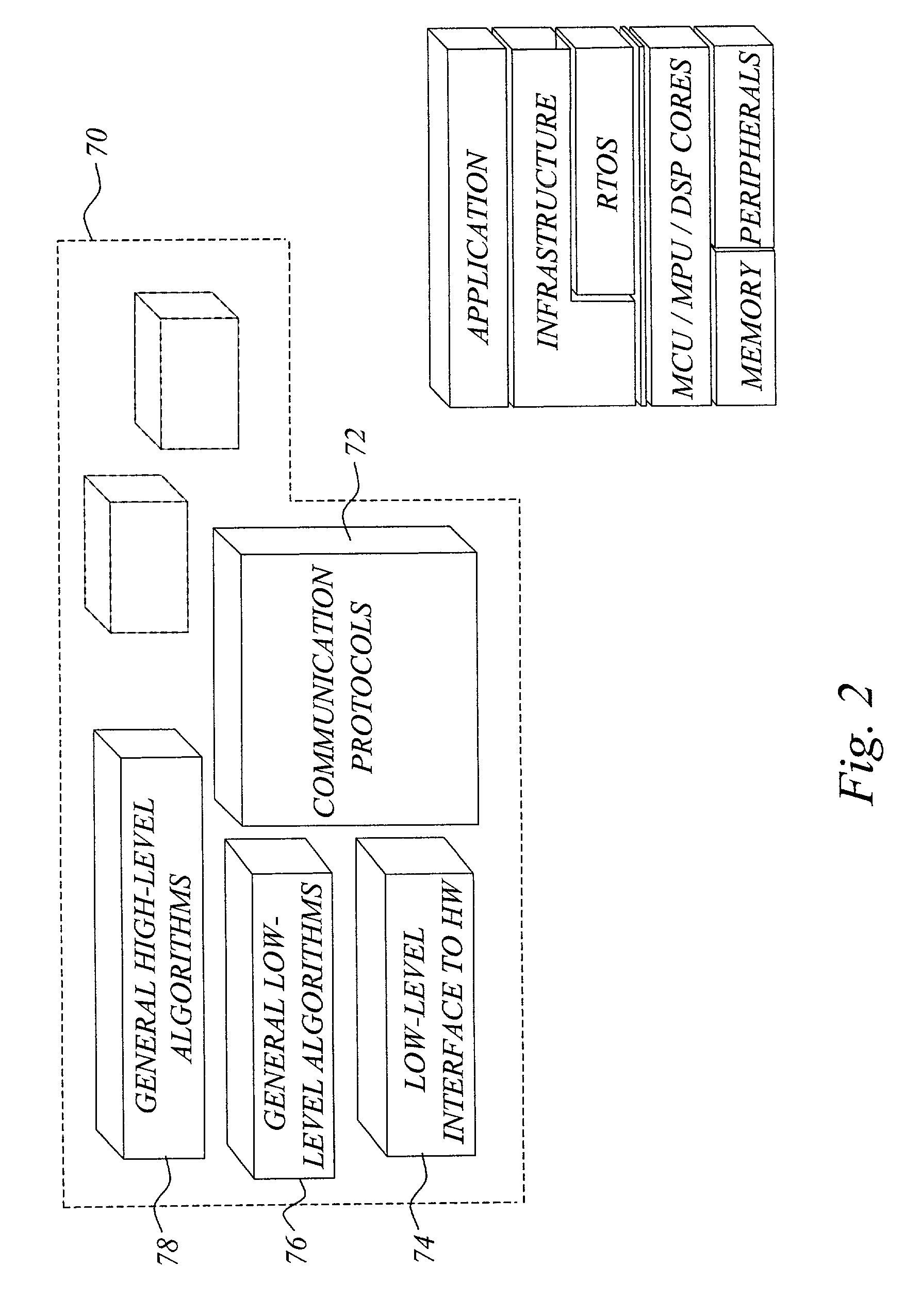
The invention concerns a software development tool for embedded computer systems, and is based on a repository of configurable, pre-programmed software components, together with associated tools for user selection and configuration of the components and a code generator for extracting relevant source code based on the configuration settings. Each software component, called embedded system infrastructure component (ESIC), is a self-contained object comprising a modular code base and associated configuration structure related to an infrastructure function in a hardware-independent, non-operating-system software infrastructure for an embedded computer system. For each ESIC, the configuration tool enables user configuration of the infrastructure function based on the configuration structure of the ESIC to match the requirements of the target application. For each ESIC, the code generator utilizes the configuration settings to extract source code for the infrastructure as a subset of the modular code base. This approach provides fast, efficient and flexible development of embedded system software infrastructures.
Owner:EMBEDDED ARTIST
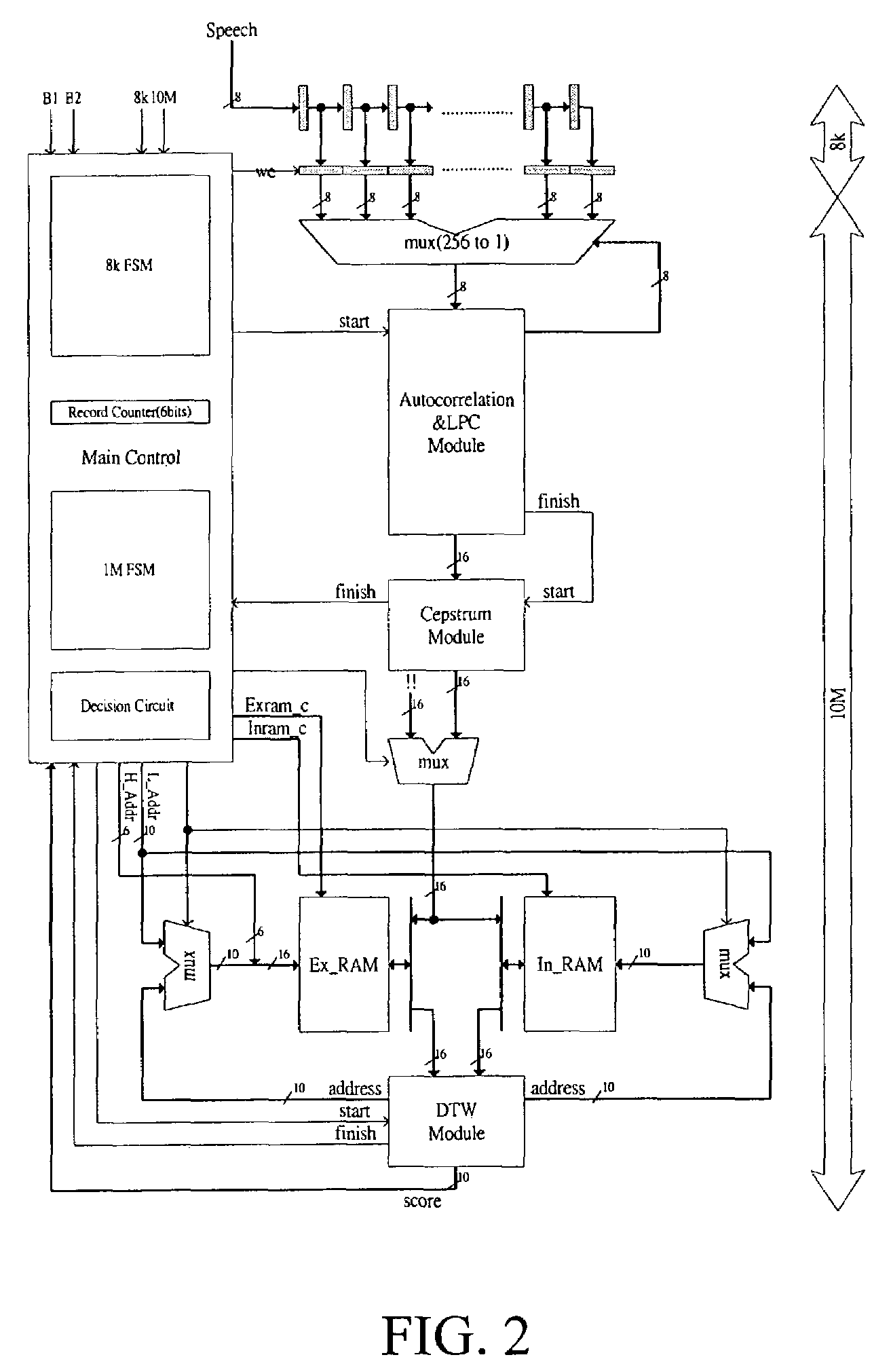
Speech recognition system
InactiveUS7266496B2Improve execution speedLow costSpeech recognitionLinearityLinear prediction coefficient
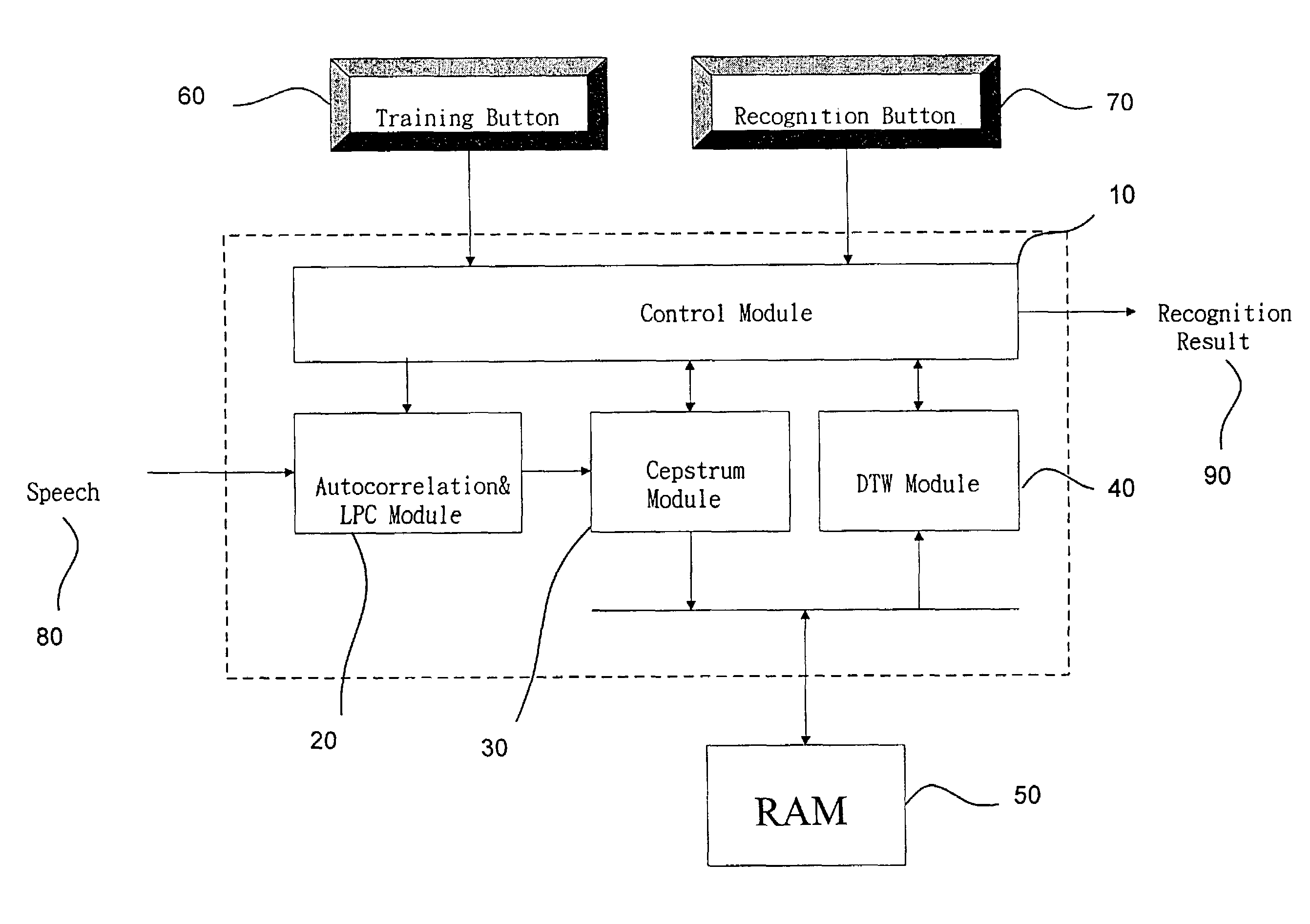
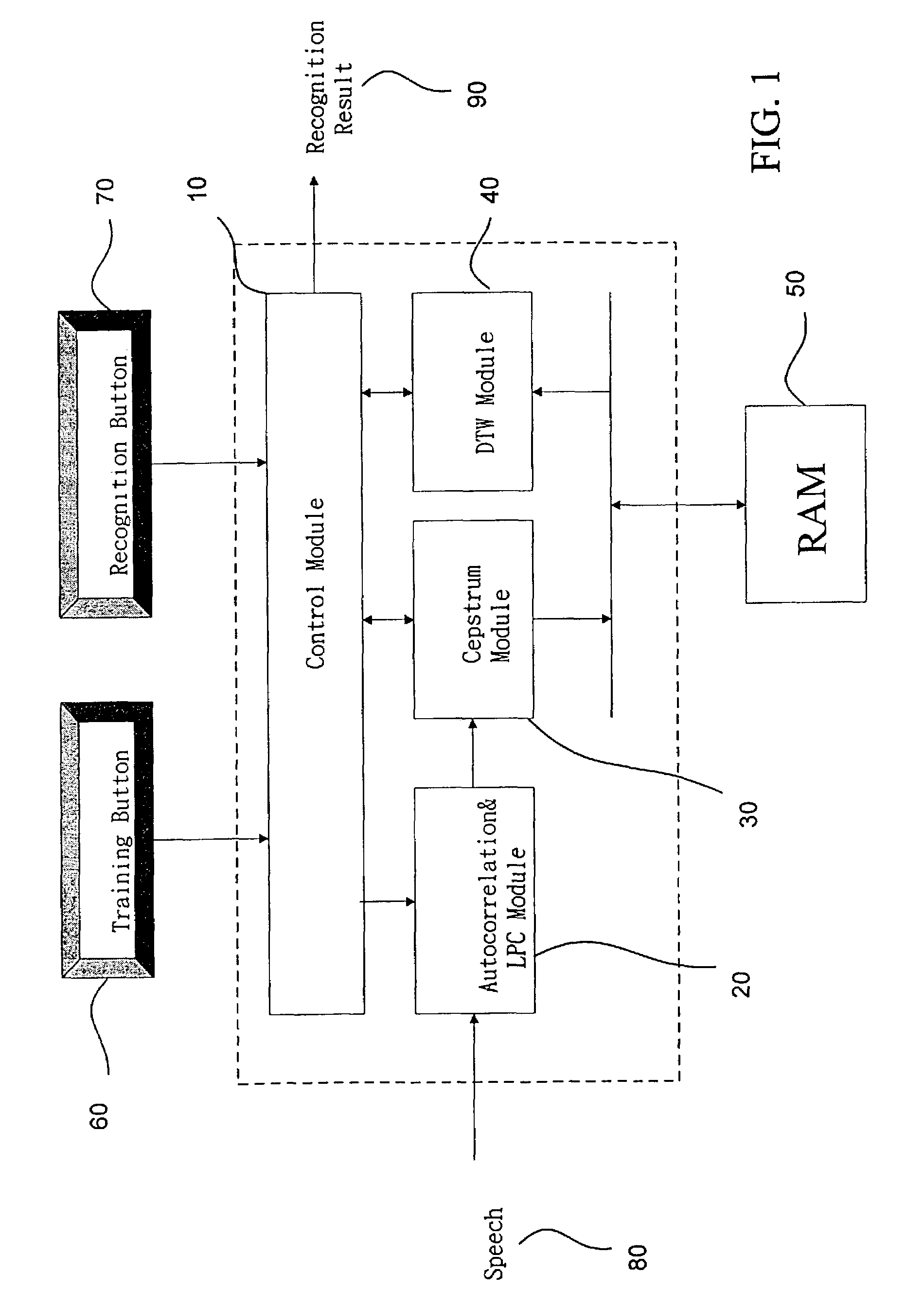
The present invention discloses a complete speech recognition system having a training button and a recognition button, and the whole system uses the application specific integrated circuit (ASIC) architecture for the design, and also uses the modular design to divide the speech processing into 4 modules: system control module, autocorrelation and linear predictive coefficient module, cepstrum module, and DTW recognition module. Each module forms an intellectual product (IP) component by itself. Each IP component can work with various products and application requirements for the design reuse to greatly shorten the time to market.
Owner:NAT CHENG KUNG UNIV
Automatic software production system
InactiveUS20030167455A1Shorten the time to marketEasy to useDigital data information retrievalProgram documentationHandling CodeSystem requirements
An automated software production system is provided, in which system requirements are captured, converted into a formal specification, and validated for correctness and completeness. In addition, a translator is provided to automatically generate a complete, robust software application based on the validated formal specification, including user-interface code and error handling code.
Owner:IBORRA JOSE +1
Navigation system for a vehicle
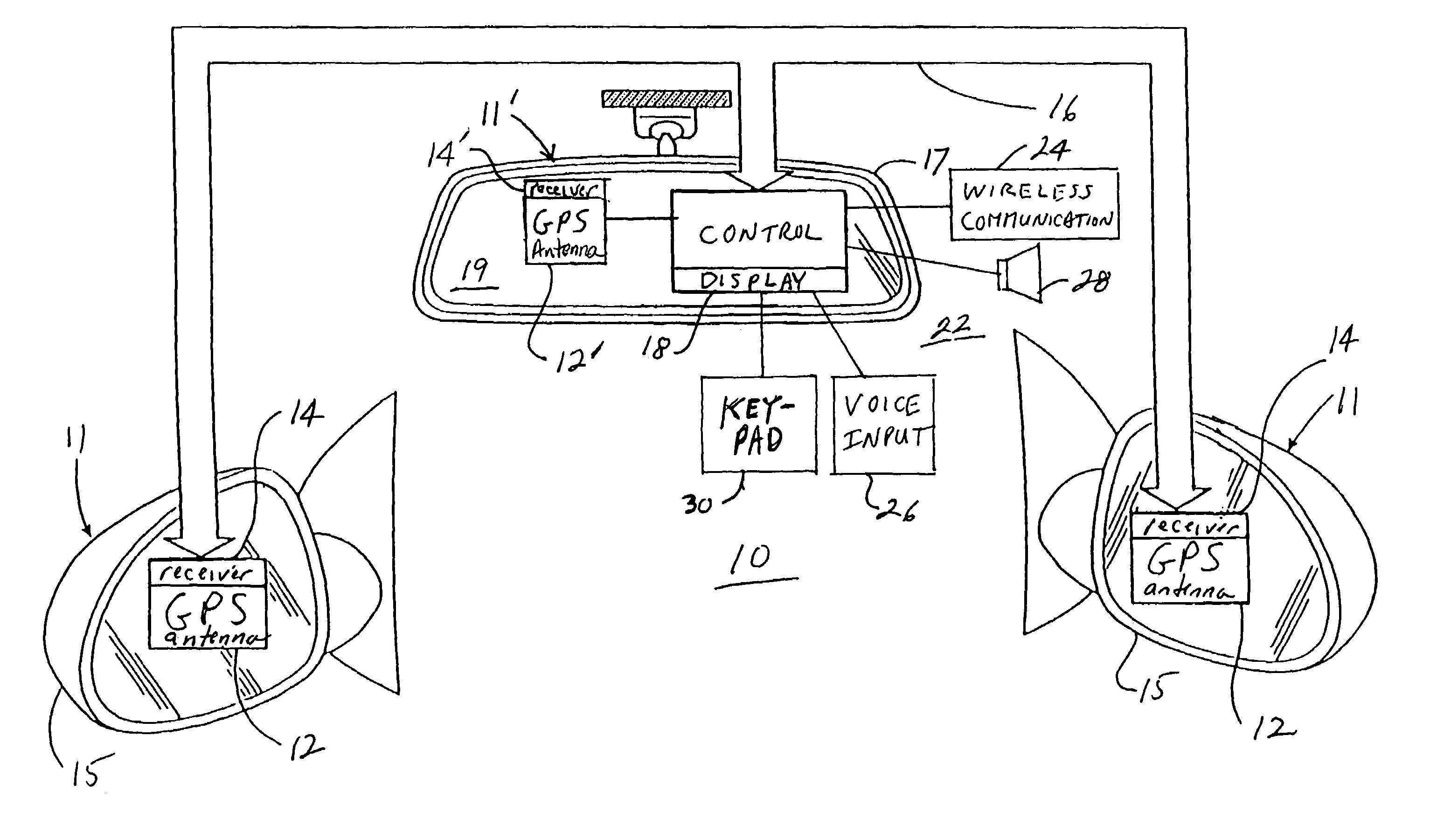
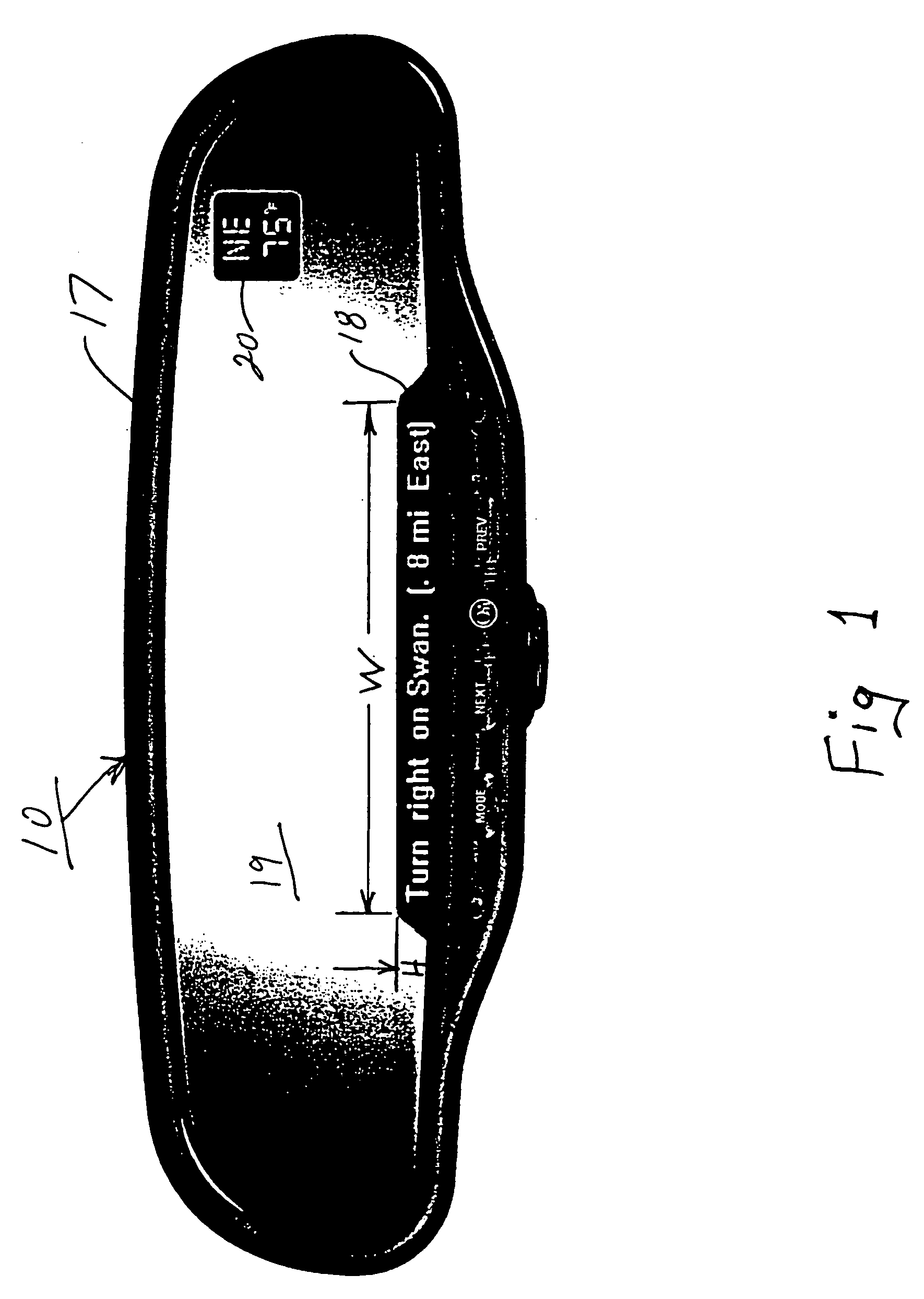
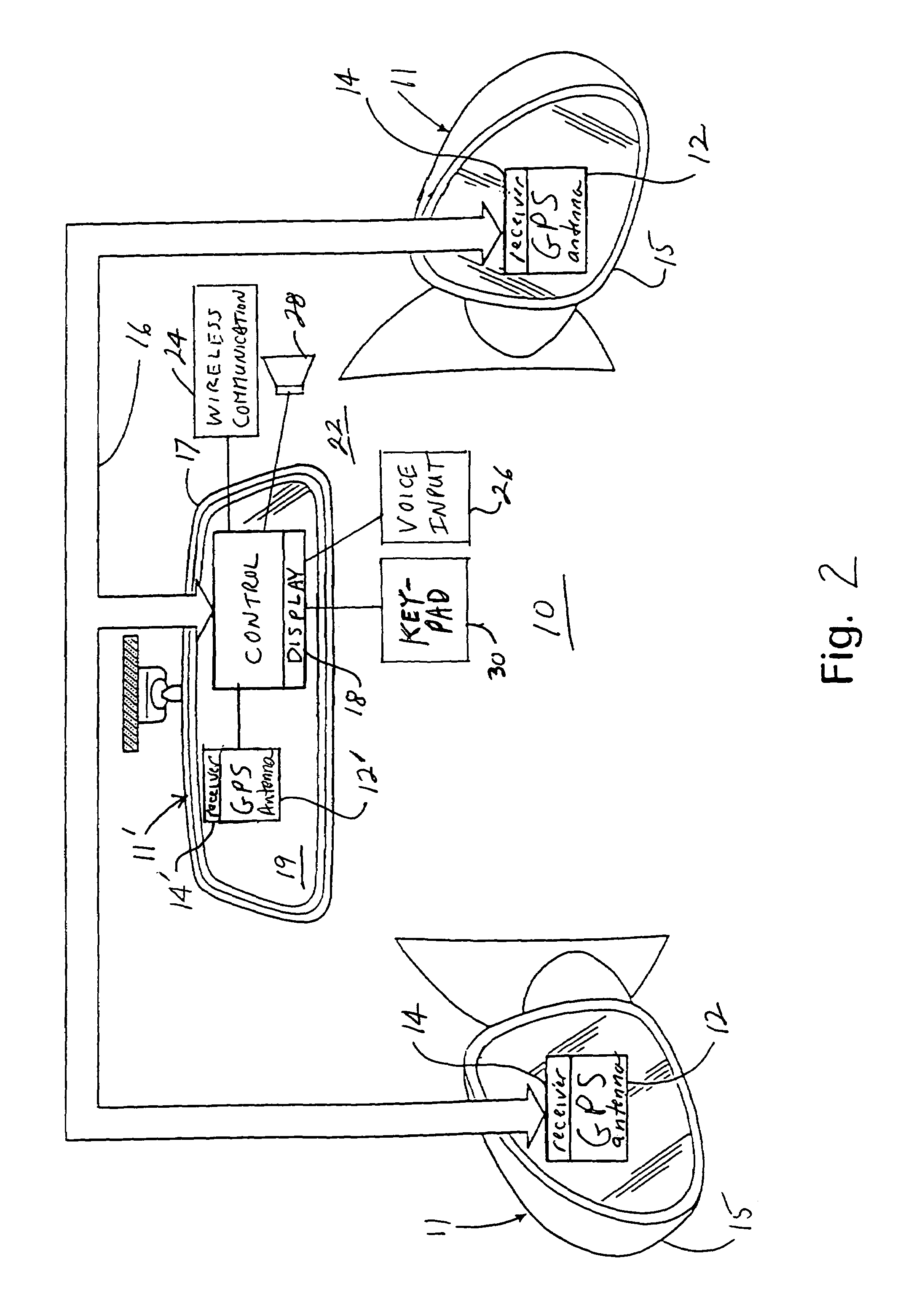
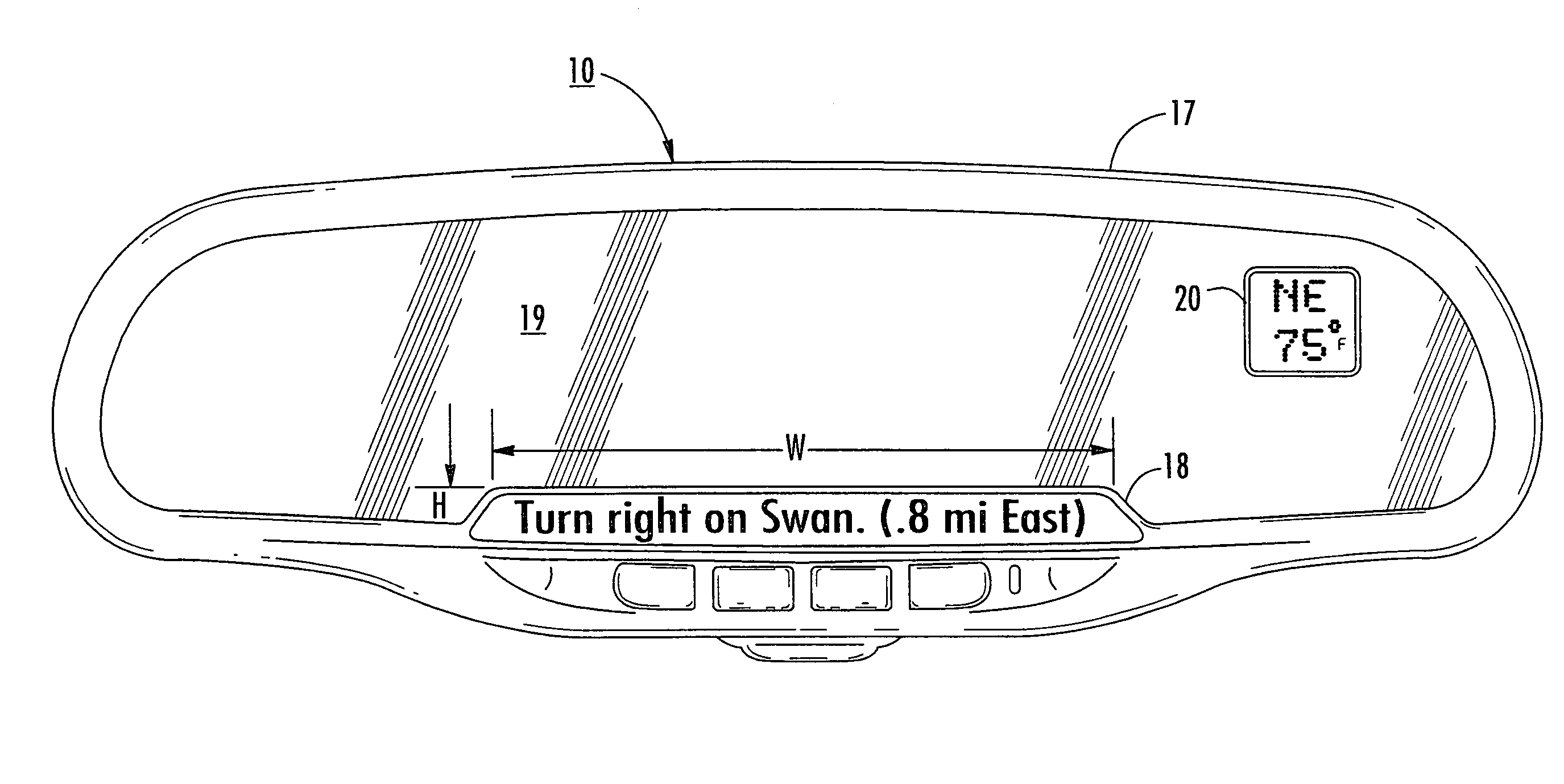
InactiveUS7158881B2Increase supplyImprove abilitiesInstruments for road network navigationMirrorsTelecommunications linkSatellite
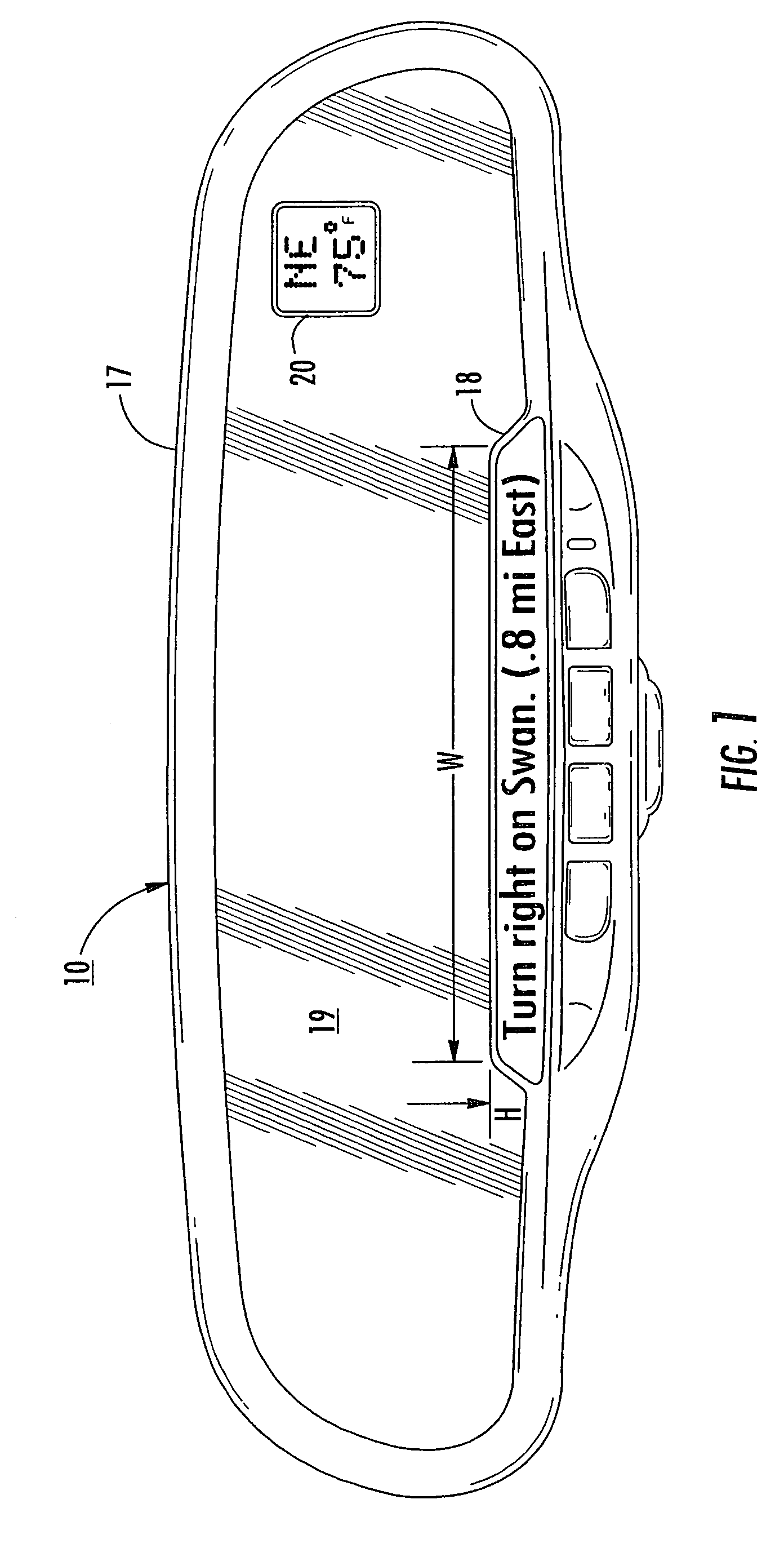
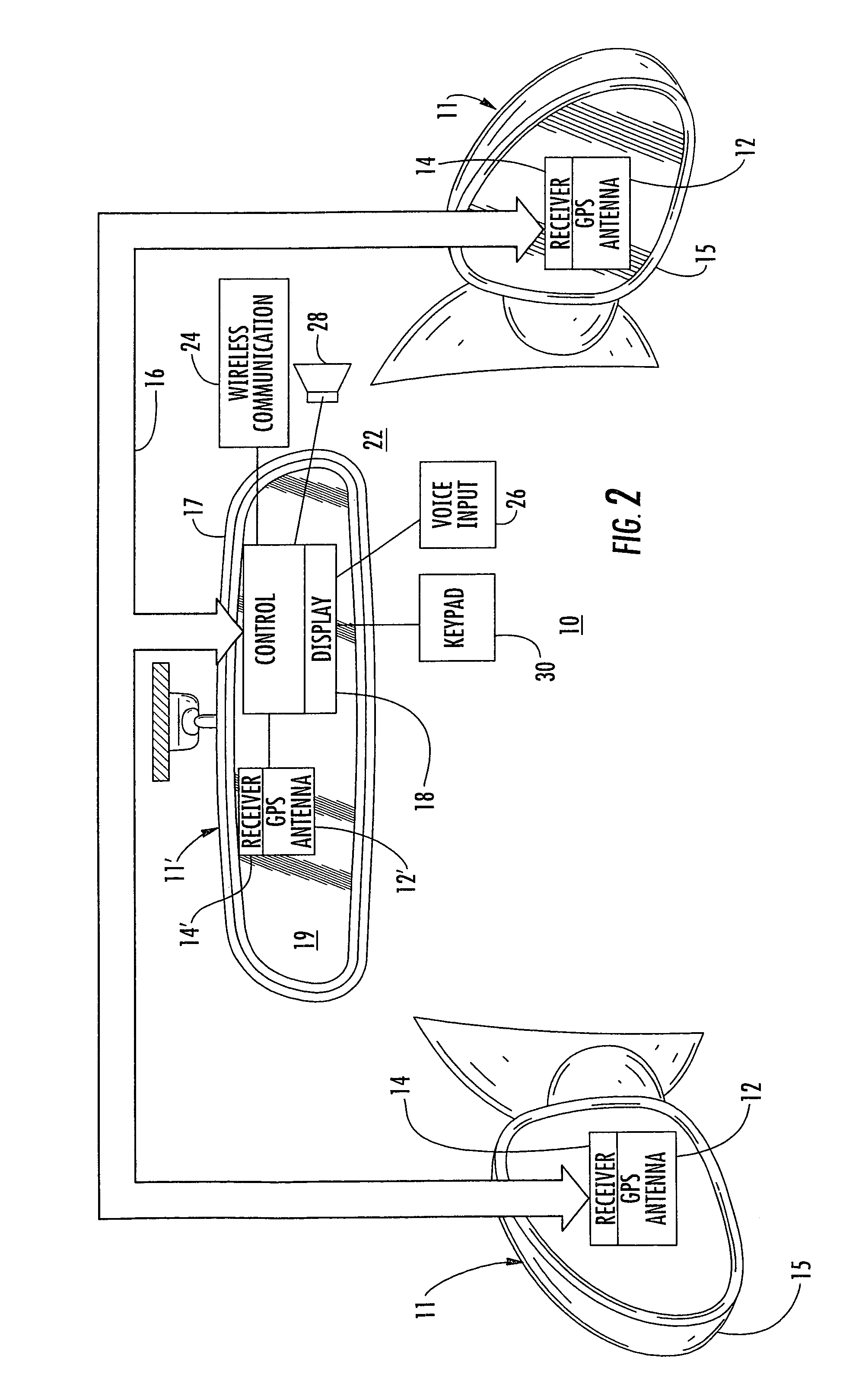
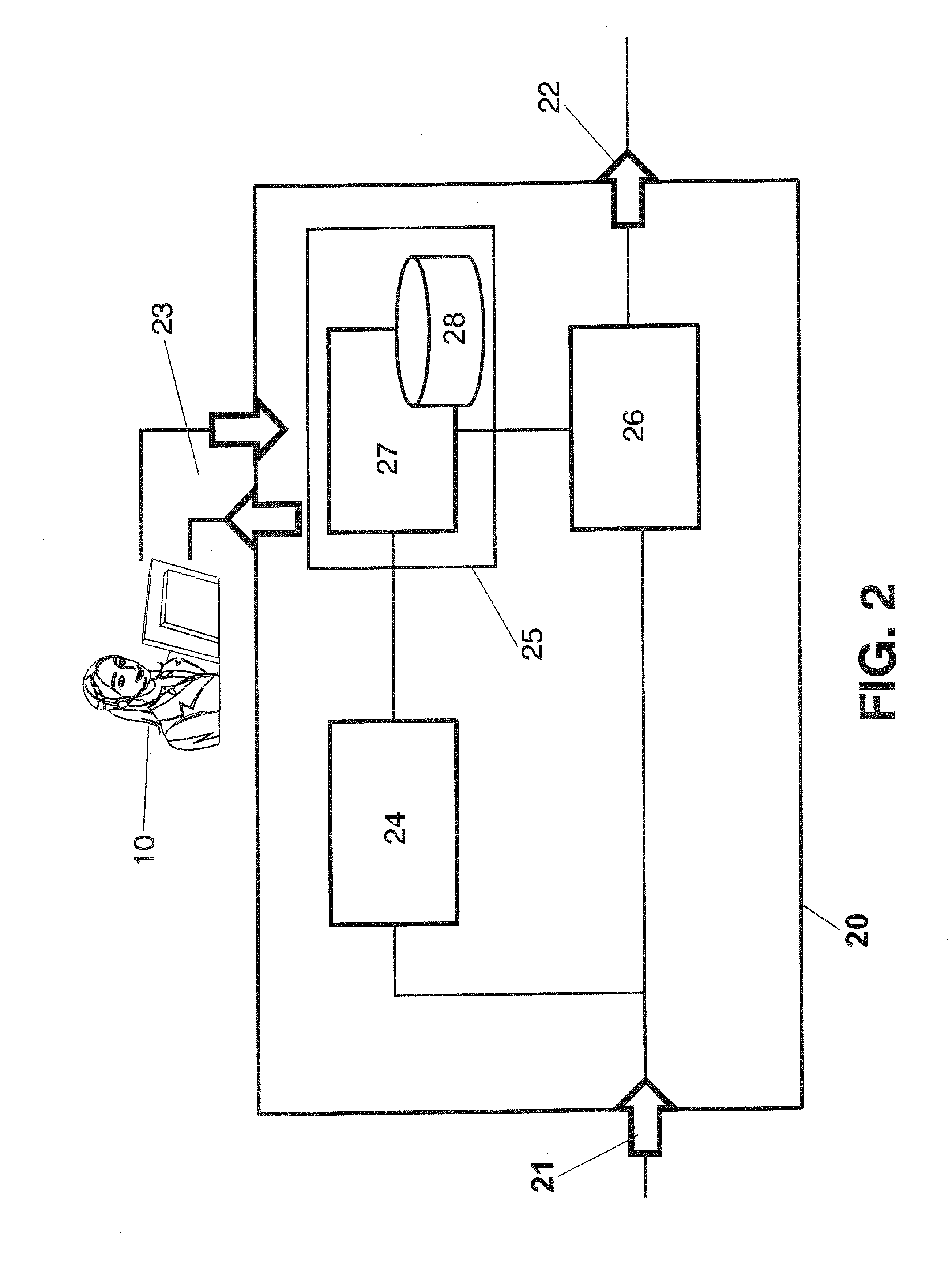
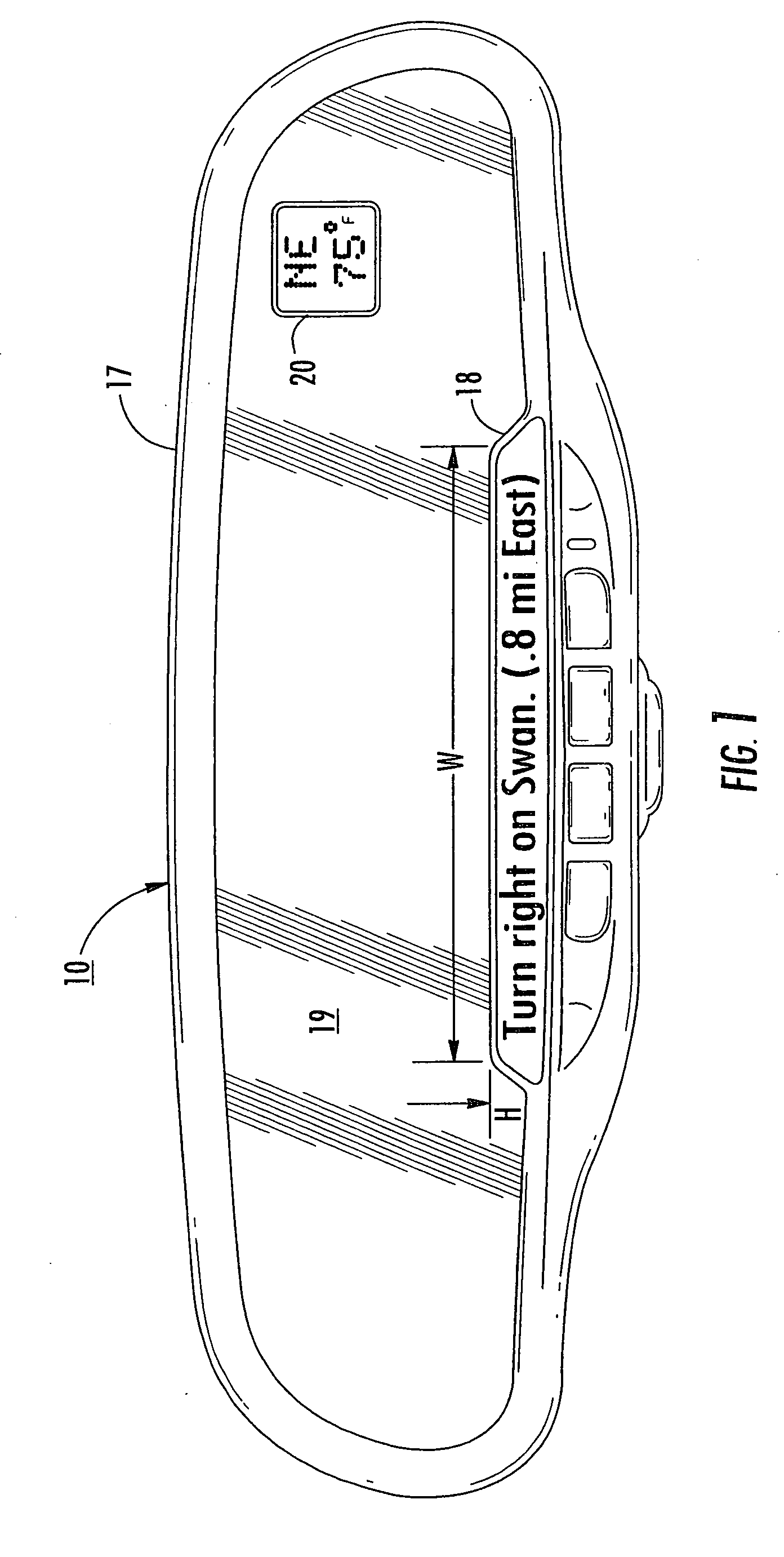
A navigation system suitable for use in a vehicle includes an interior rearview mirror assembly having an interior reflective element. A database at a site remote from the vehicle includes map data and / or directory data. A global-positioning system receiver is operable to receive signals from satellites external to said vehicles. A transceiver is operable to engage in a wireless communication with a remote transceiver located at a site remote from the vehicle to received data derived from the database. A user input allows a user to input a destination. The system responds to an input of a destination by wirelessly communicating the destination to the remote transceiver and downloading directions. A display that displays turn-by-turn instructions to the destination during the road journey based on the downloaded directions and on the signals received by the global-positioning system receiver. The turn-by-turn instructions may include (i) direction, (ii) when to turn, and / or (iii) how far until the turn. The navigation system preferably includes a short-range wireless communication link.
Owner:DONNELLY CORP
Navigation system for a vehicle
InactiveUS7328103B2Increase supplyImprove abilitiesInstruments for road network navigationRoad vehicles traffic controlTelecommunications linkGeolocation
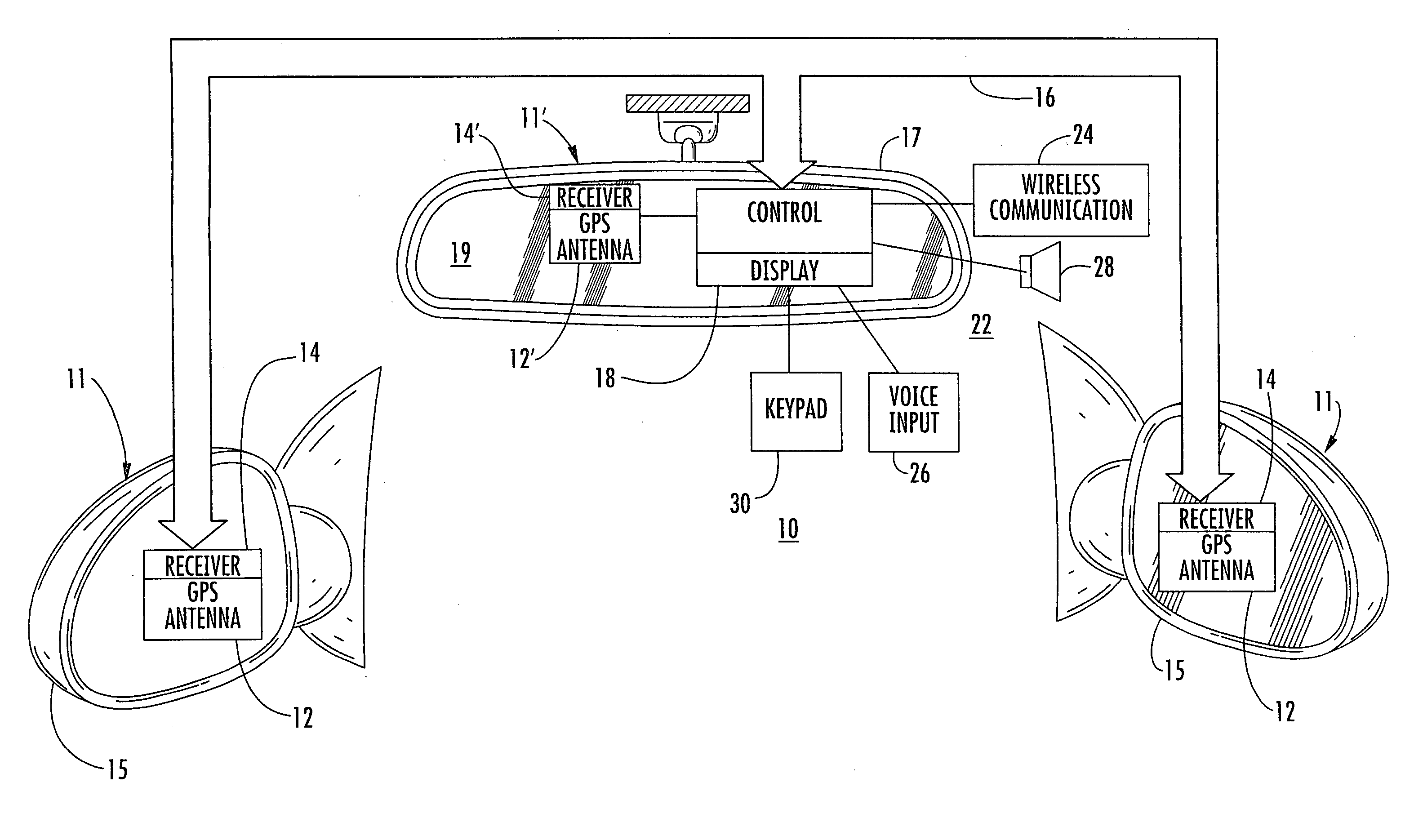
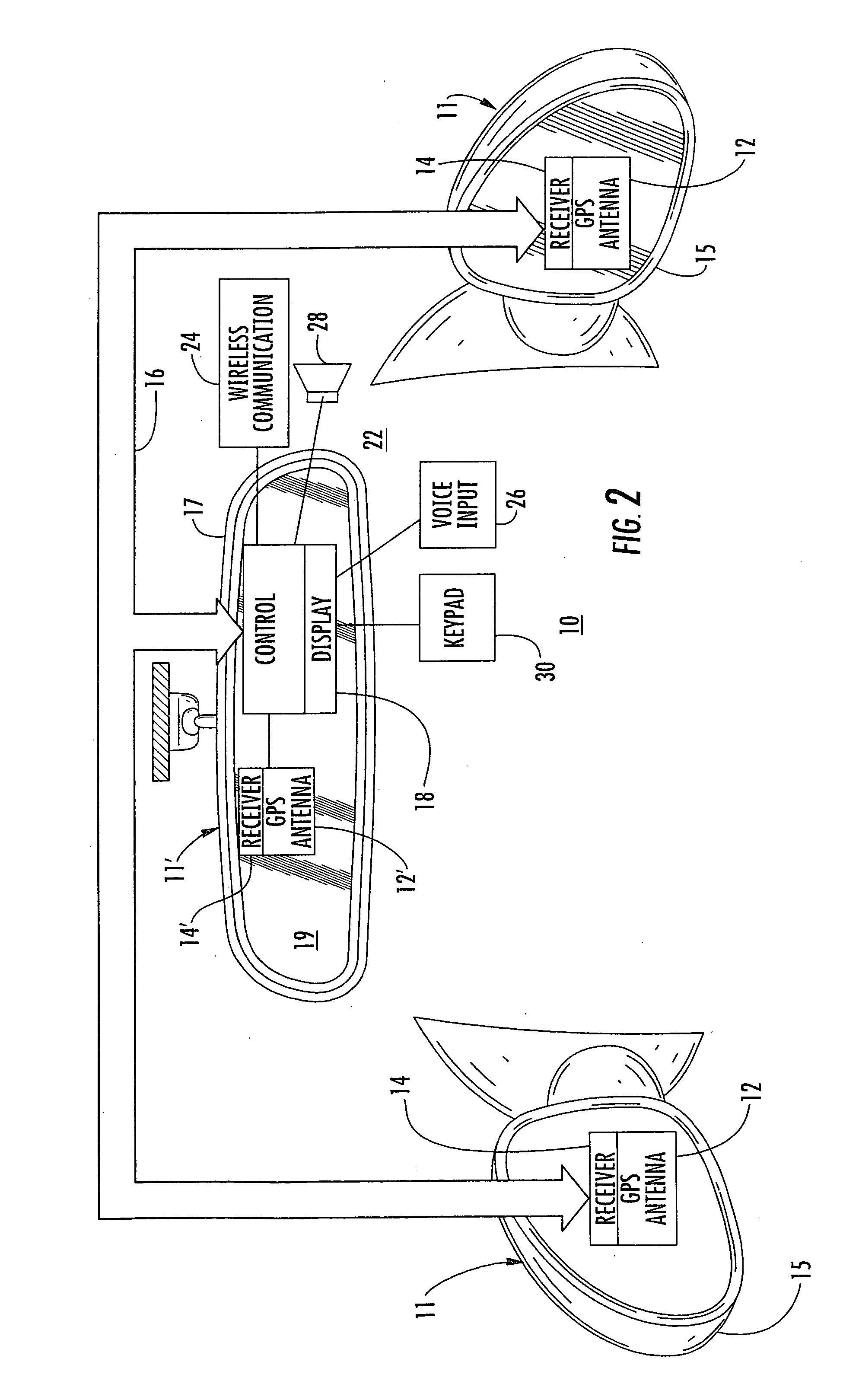
An interior rearview mirror system for a vehicle includes an interior rearview mirror assembly having a reflective element. An information display is at the interior rearview mirror assembly and displays information for viewing by a driver of the vehicle. The information display may be located behind the reflective element and viewable through the reflective element by the driver of the vehicle. The mirror system includes a handheld device having a position locator and being capable of establishing a geographical position of the handheld device. The information displayed by the information display is derived at least in part via a wireless communication link with the handheld device. The information display may include a display of turn-by-turn information for viewing by the driver of the vehicle. The information display may comprise a video screen.
Owner:DONNELLY CORP
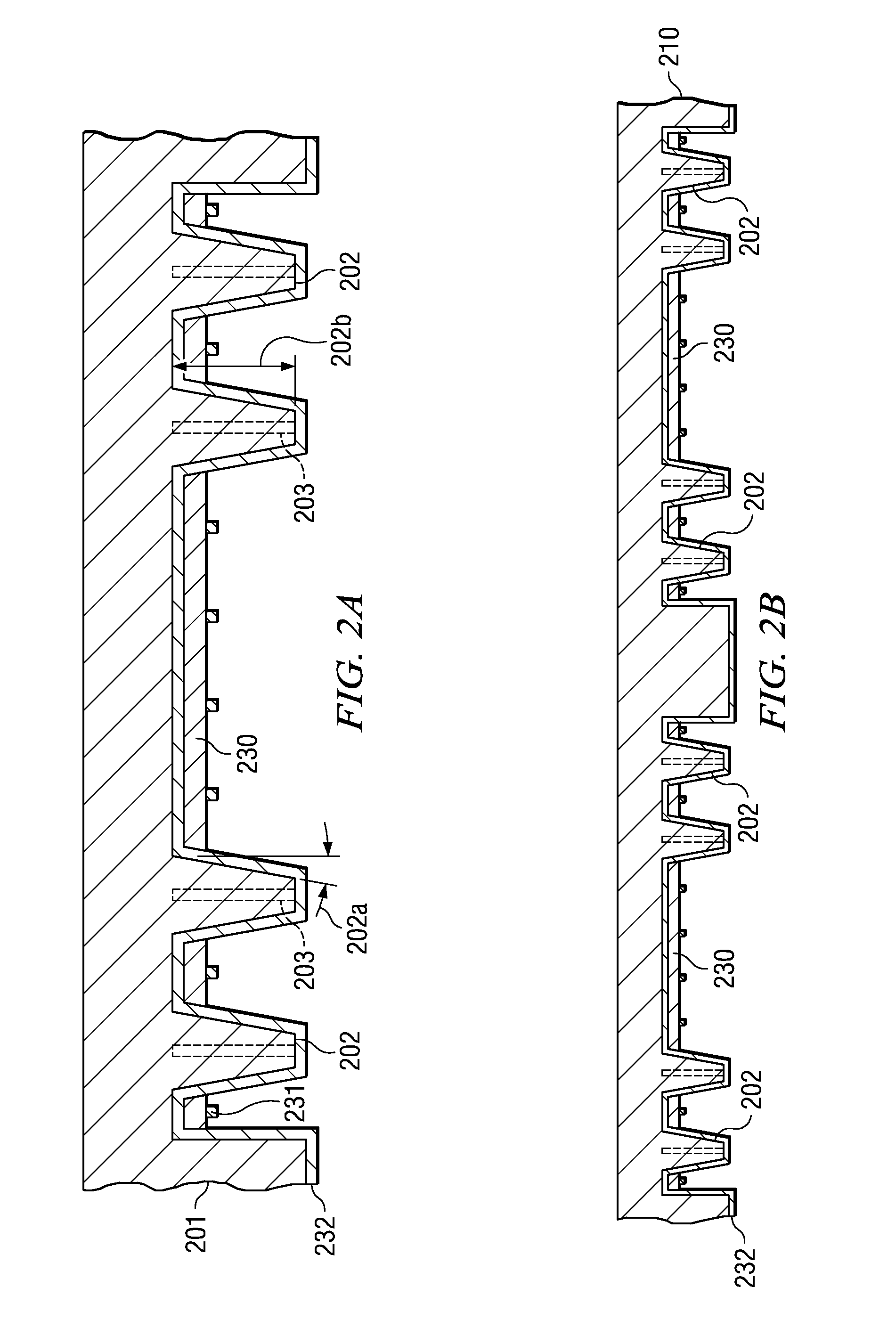
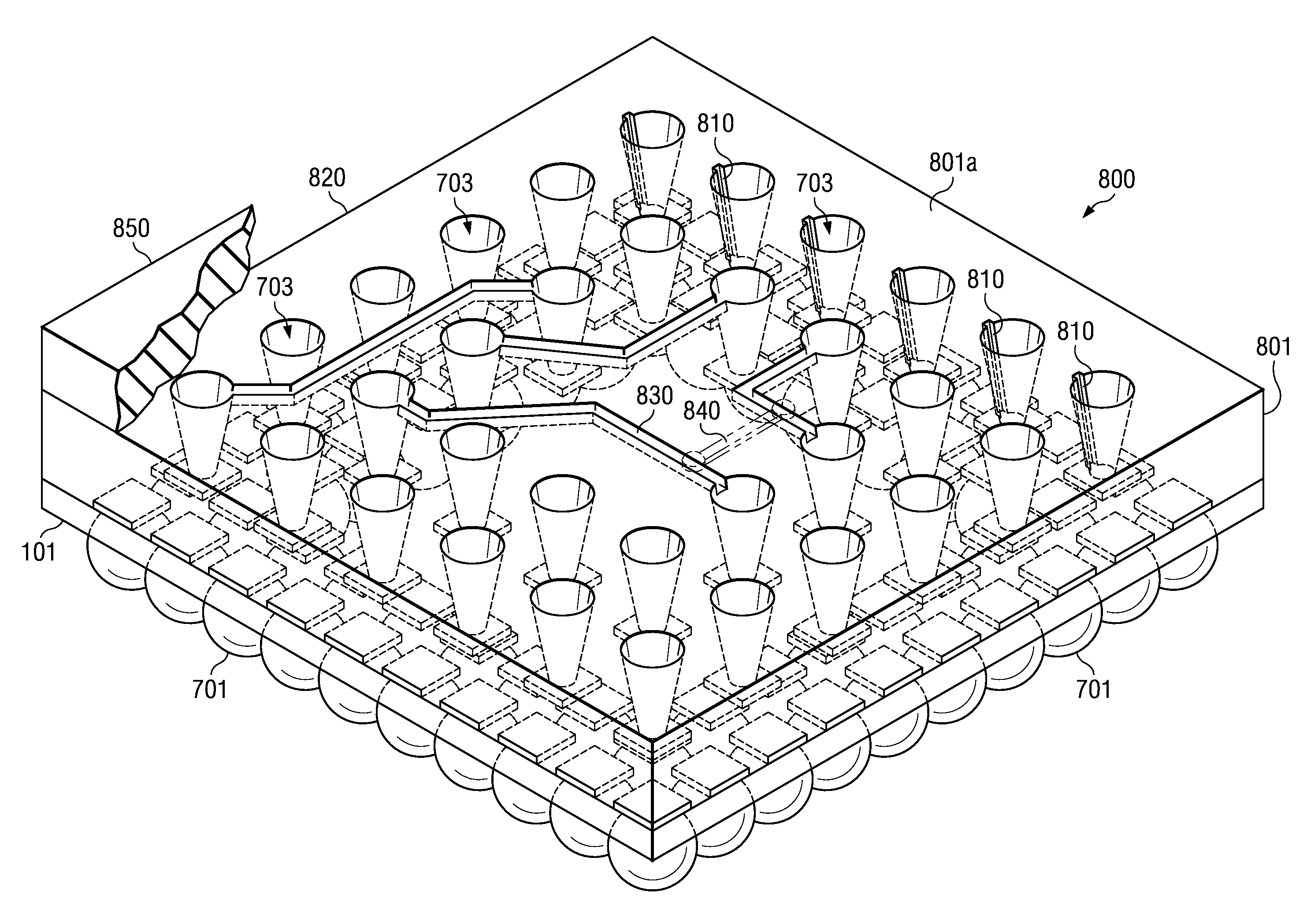
Array molded package-on-package having redistribution lines
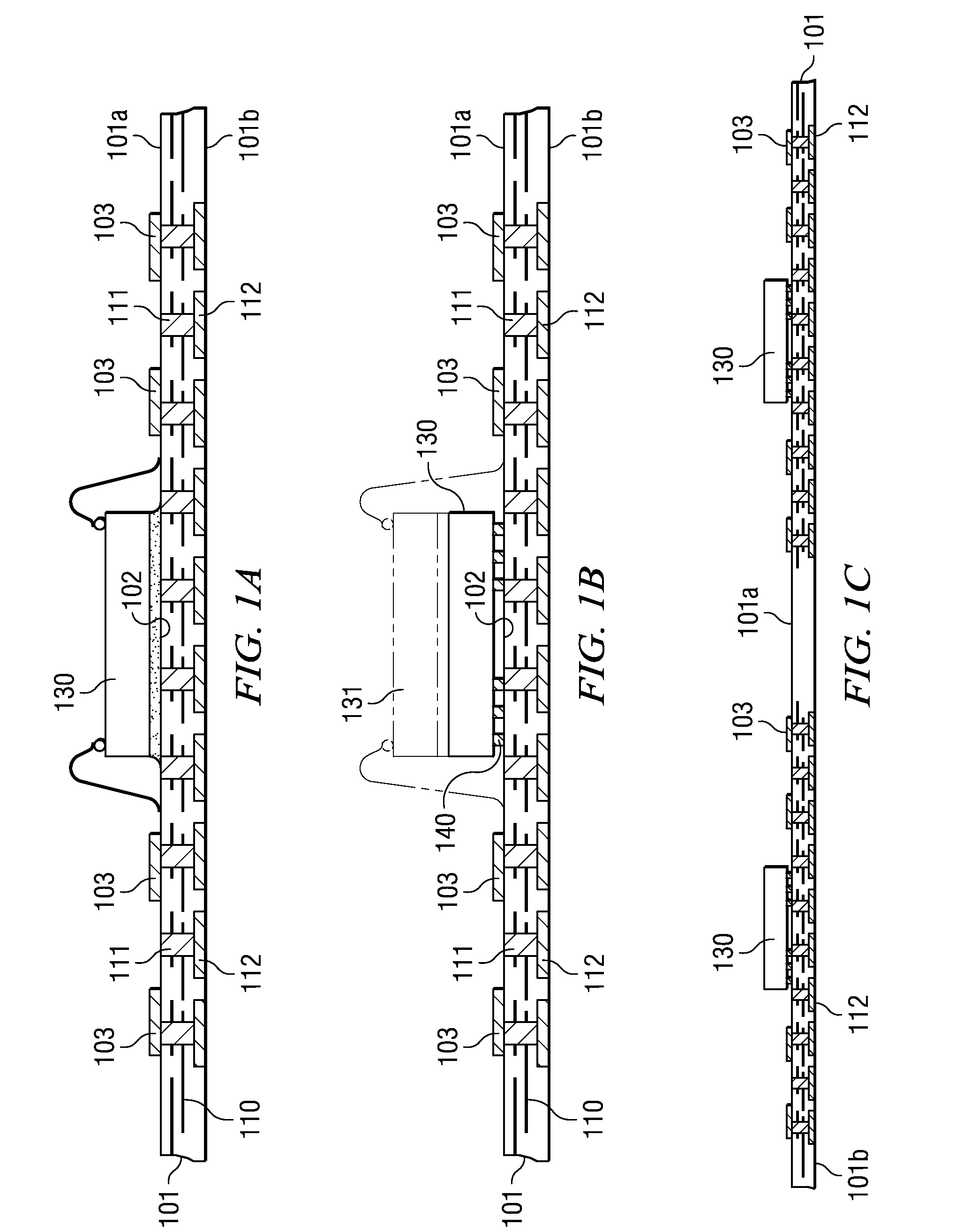
ActiveUS20080315385A1Shorten the time to marketSimple methodSemiconductor/solid-state device detailsSolid-state devicesContact padCoupling
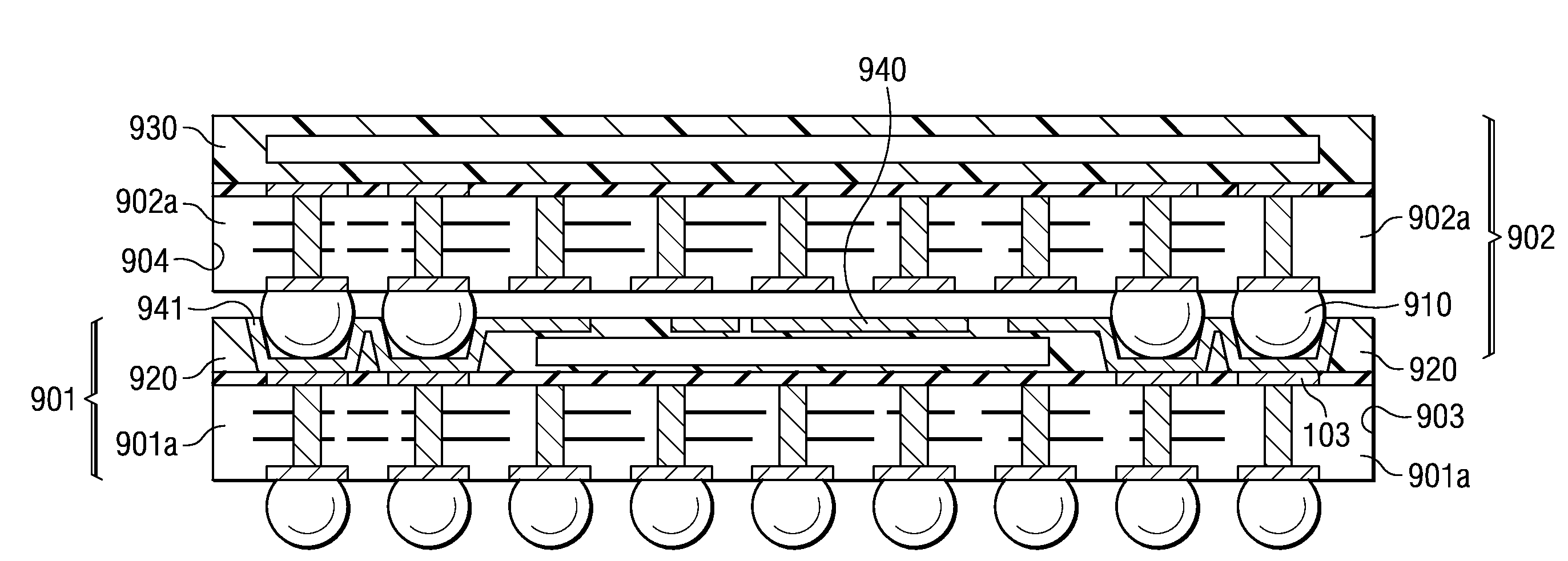
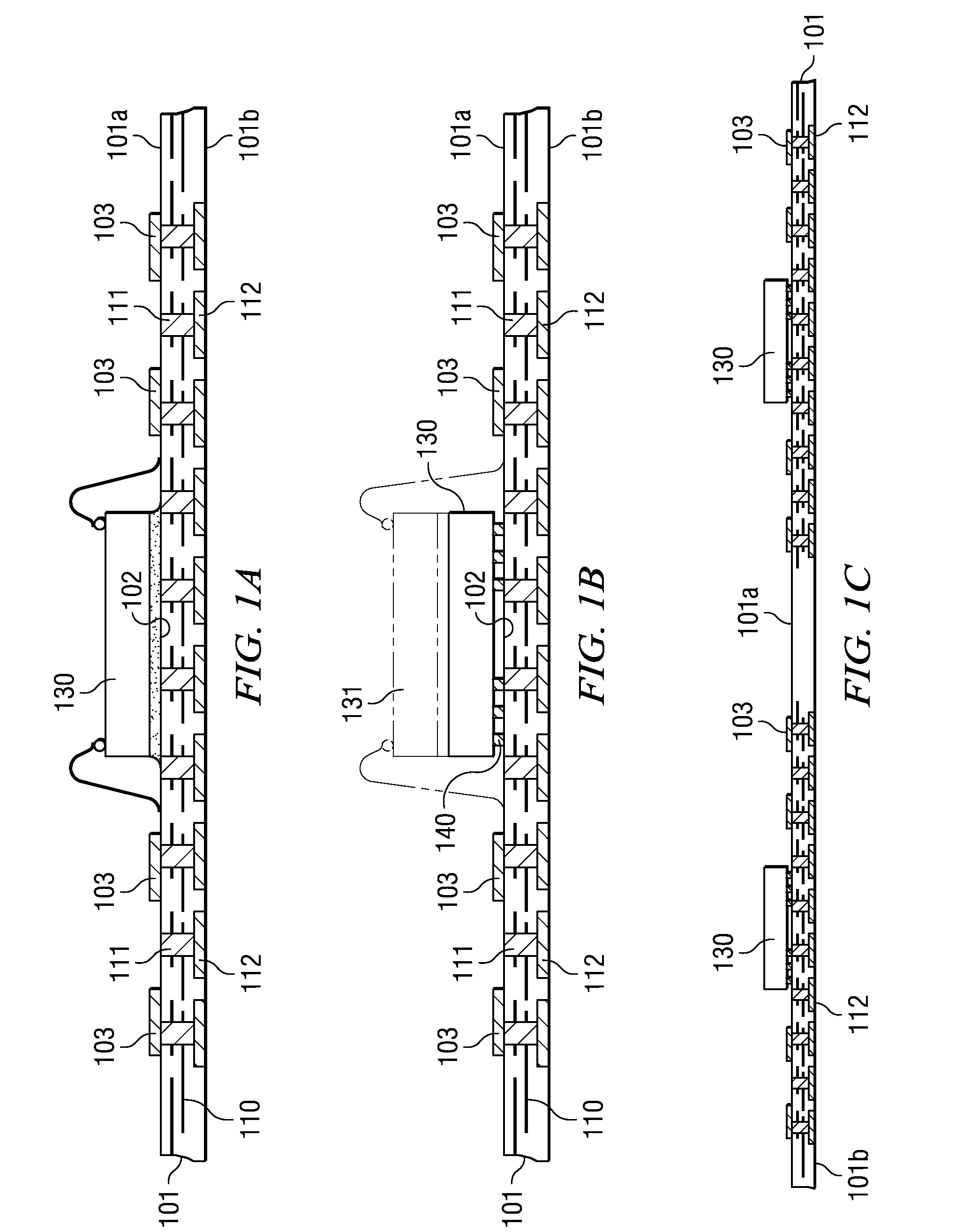
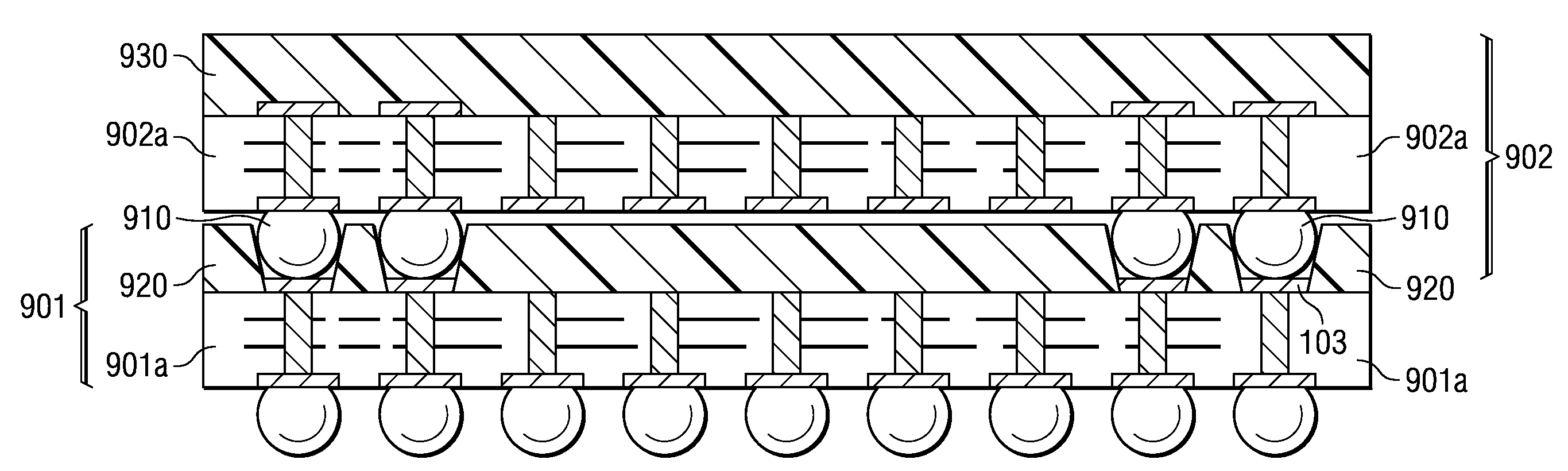
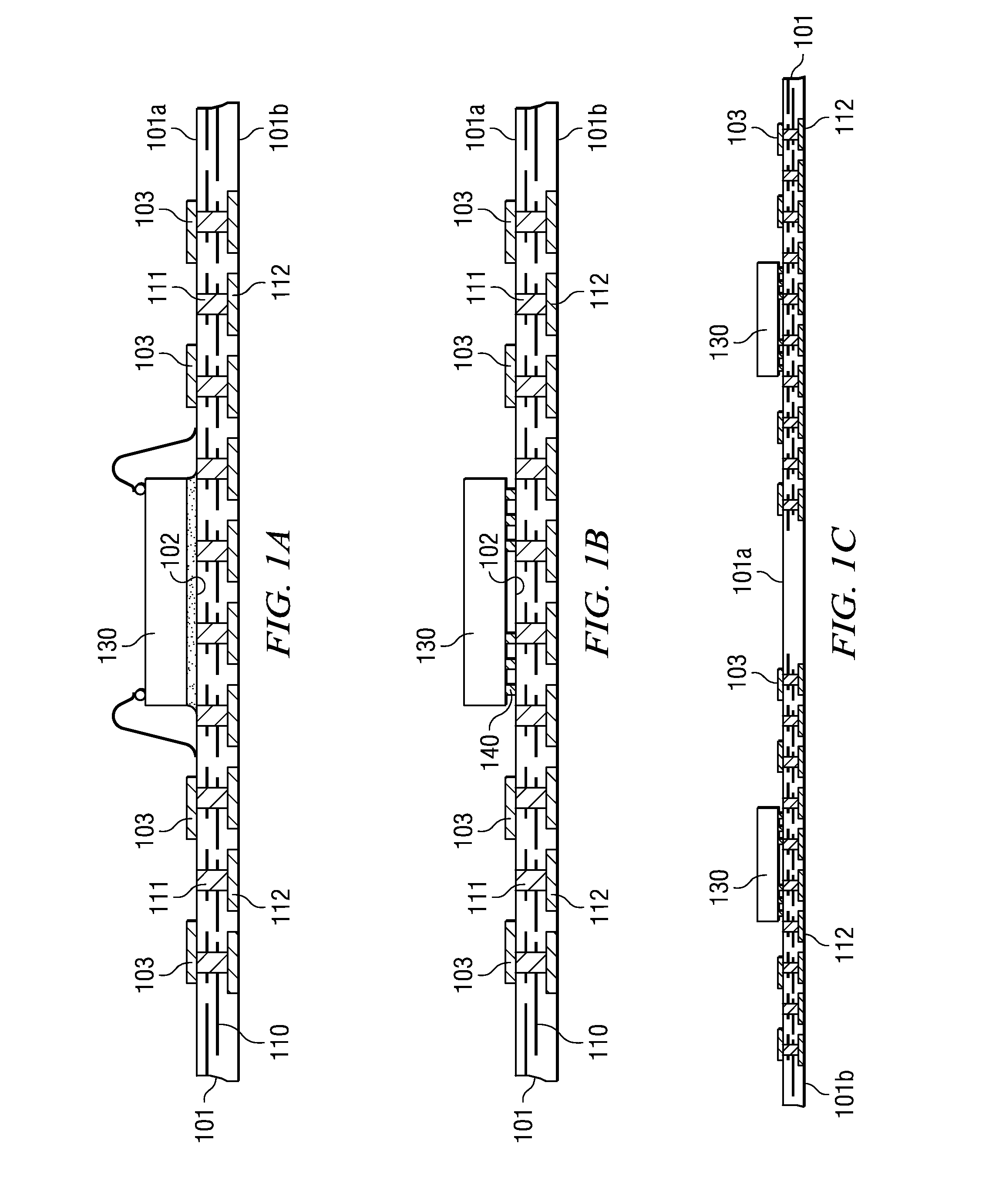
A semiconductor device with a sheet-like insulating substrate (101) integral with two or more patterned layers of conductive lines and vias, a chip attached to an assembly site, and contact pads (103) in pad locations has an encapsulated region on the top surface of the substrate, extending to the edge of the substrate, enclosing the chip, and having contact apertures (703) at the pad locations for external communication with the pad metal surfaces. The apertures may have not-smooth sidewall surfaces and may be filled with solder material (704) to contact the pads. Metal-filled surface grooves (710) in the encapsulated region, with smooth groove bottom and sidewalls, are selected to serve as customized routing interconnections, or redistribution lines, between selected apertures and thus to facilitate the coupling with another semiconductor device to form a package-on-package assembly.
Owner:TEXAS INSTR INC
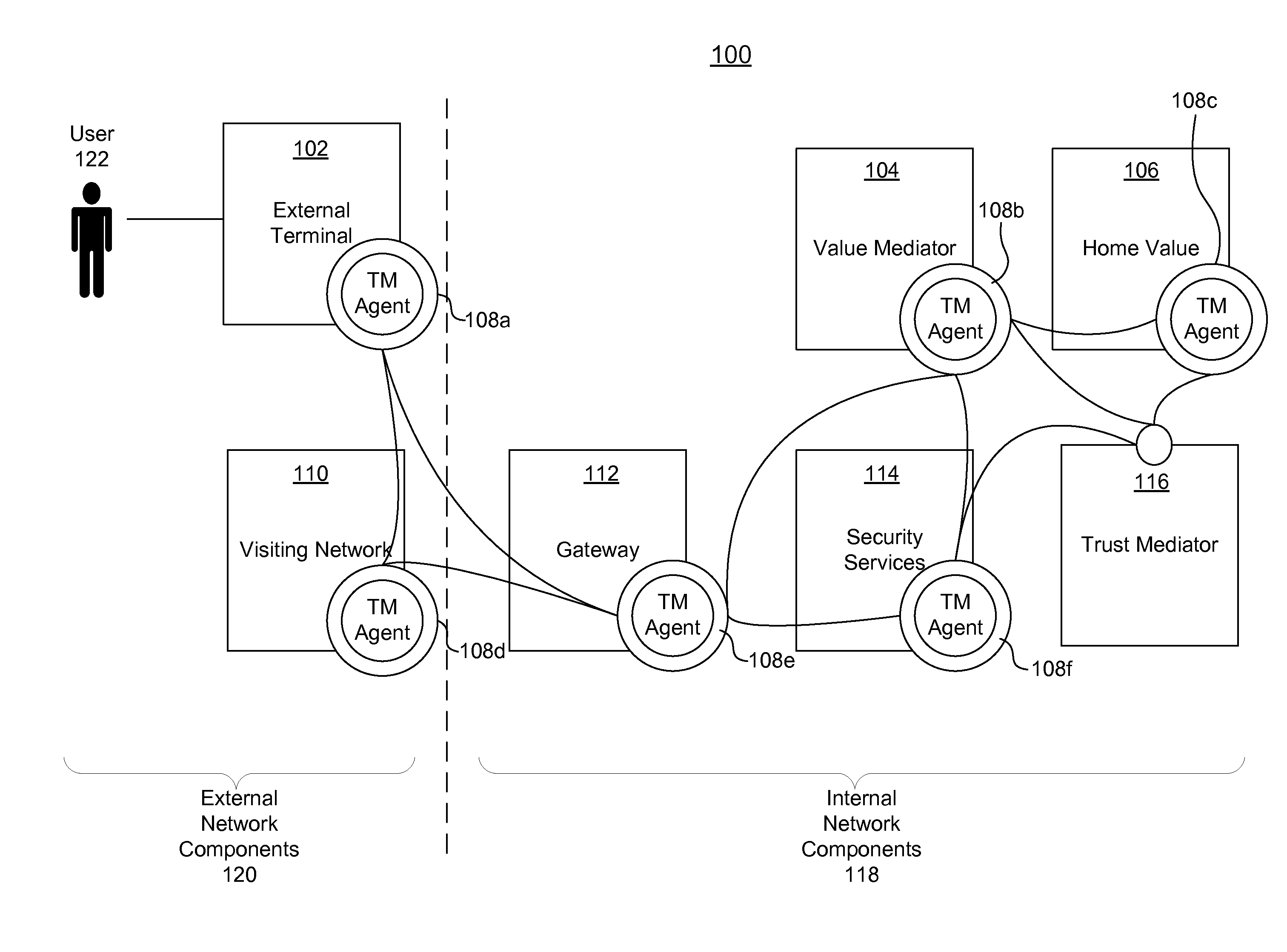
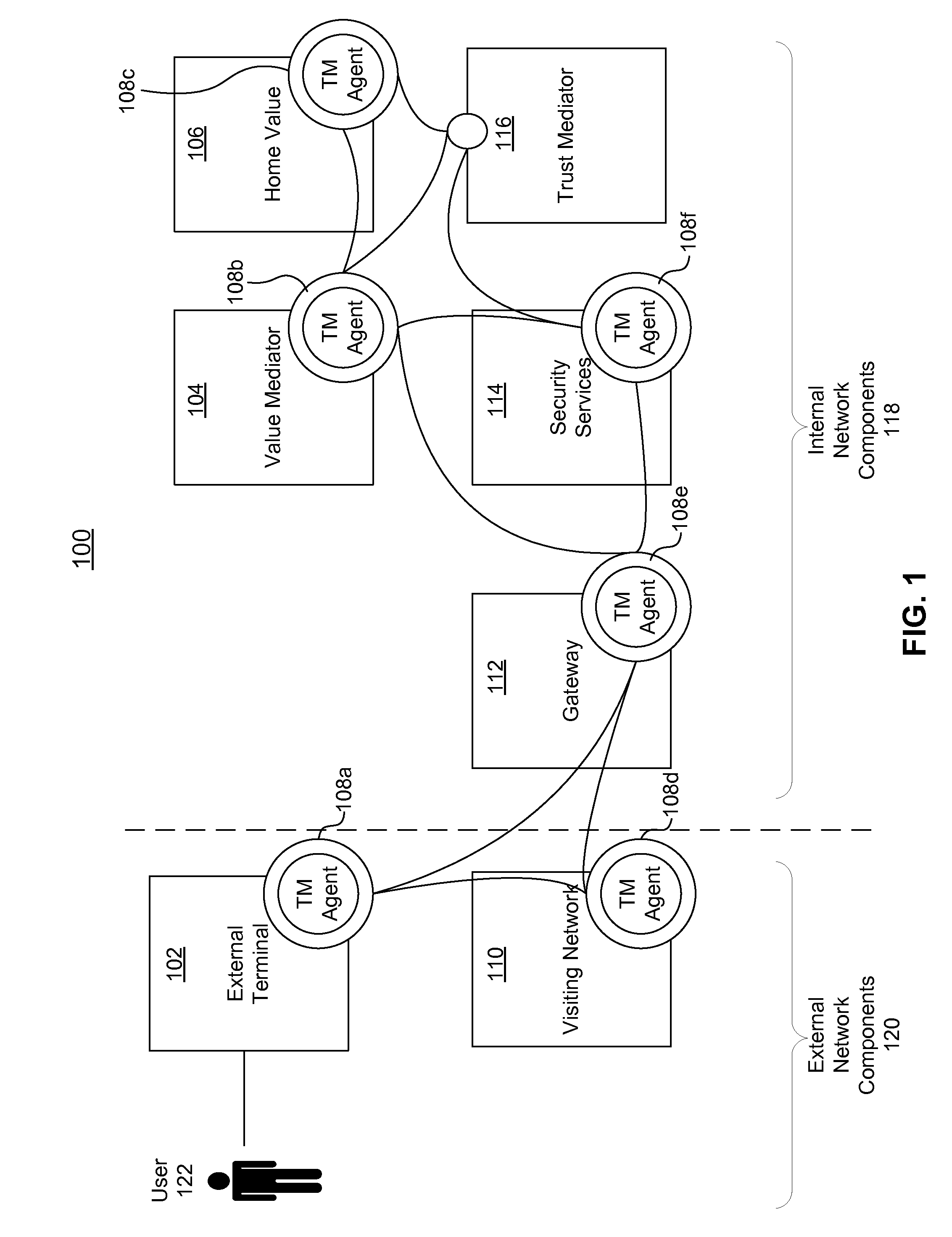
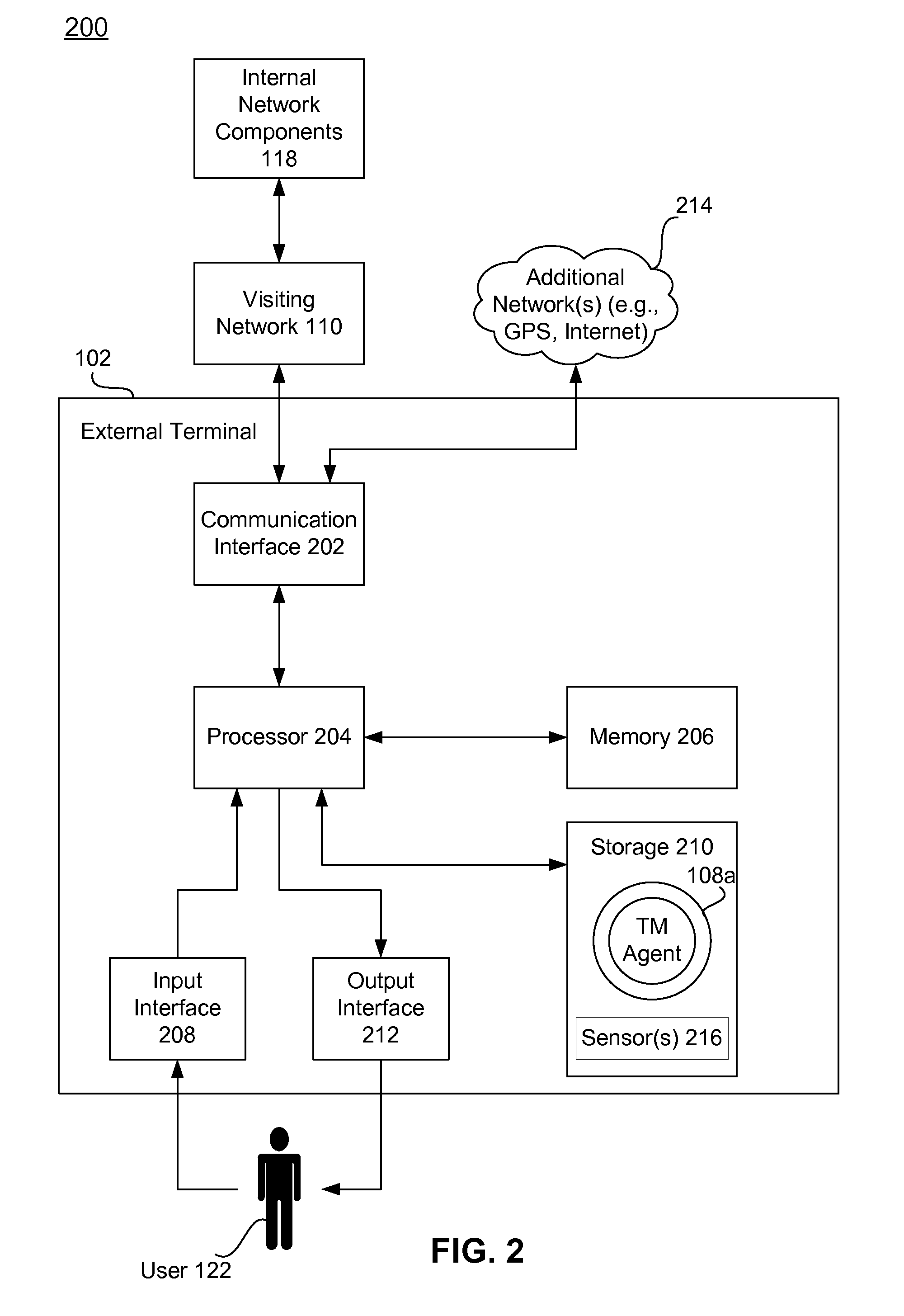
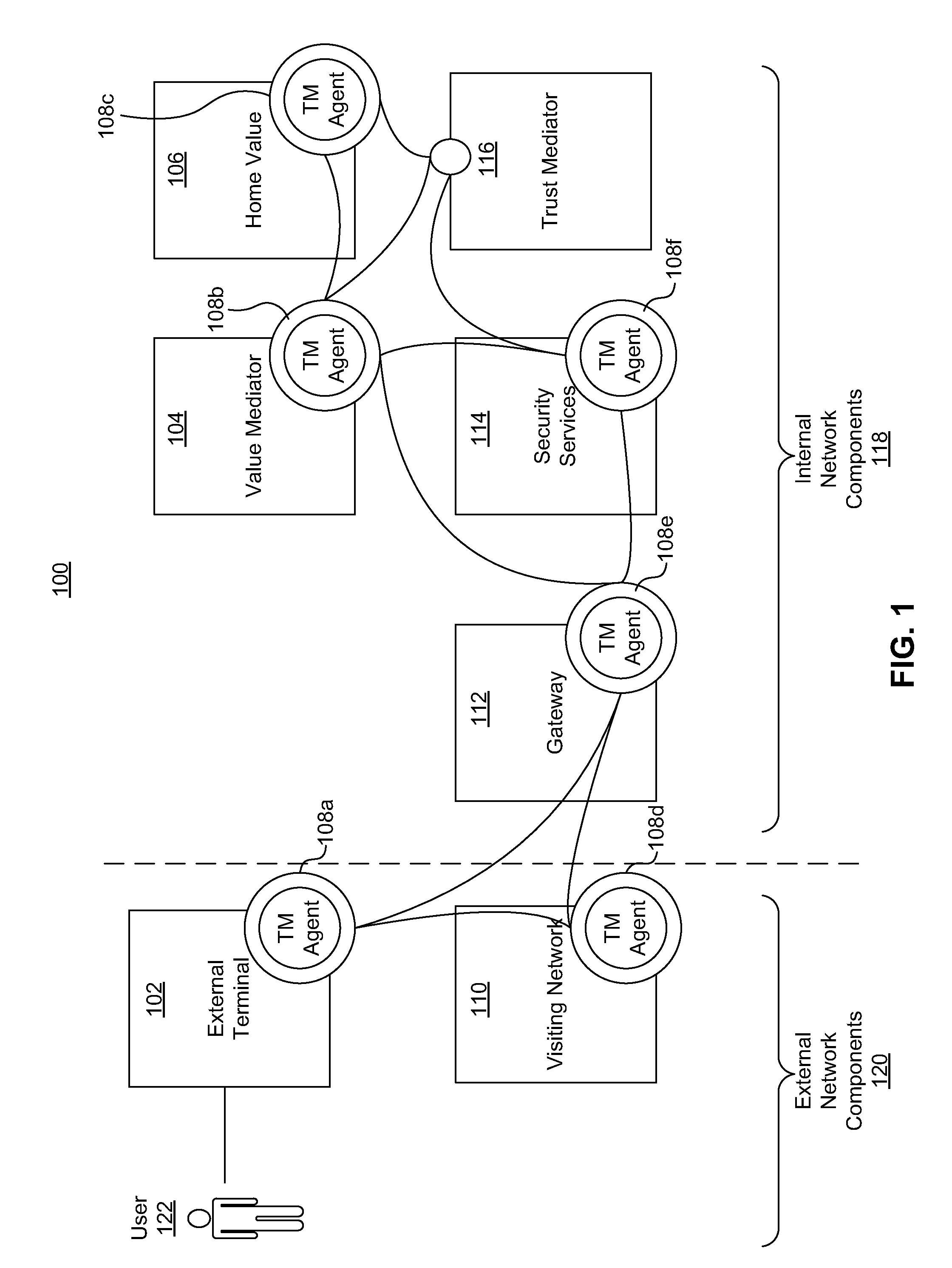
Systems, methods, and computer program products for collecting and reporting sensor data in a communication network
ActiveUS20110154497A1Shorten the time to marketIncrease propensityFinanceMemory loss protectionComputer networkEngineering
A system for collecting and reporting sensor data in a communication network includes a microprocessor coupled to a memory and an electronic storage device. The microprocessor receives sensor data from sensors, and stores the sensor data for each sensor in the electronic storage device. The microprocessor also receives, via the communication network, a data reporting instruction defining a data reporting technique corresponding to the sensor data associated with one or more of the sensors. The data reporting instruction is stored in the electronic storage device, and the microprocessor transmits, to a trust mediator over the communication network, at least a portion of the sensor data based on the data reporting instruction. The trust mediator maintains an acceptable level of security for data throughout the communication network by adjusting security safeguards based on the sensor data.
Owner:AMERICAN EXPRESS TRAVEL RELATED SERVICES CO INC
Method for Fabricating Array-Molded Package-On-Package
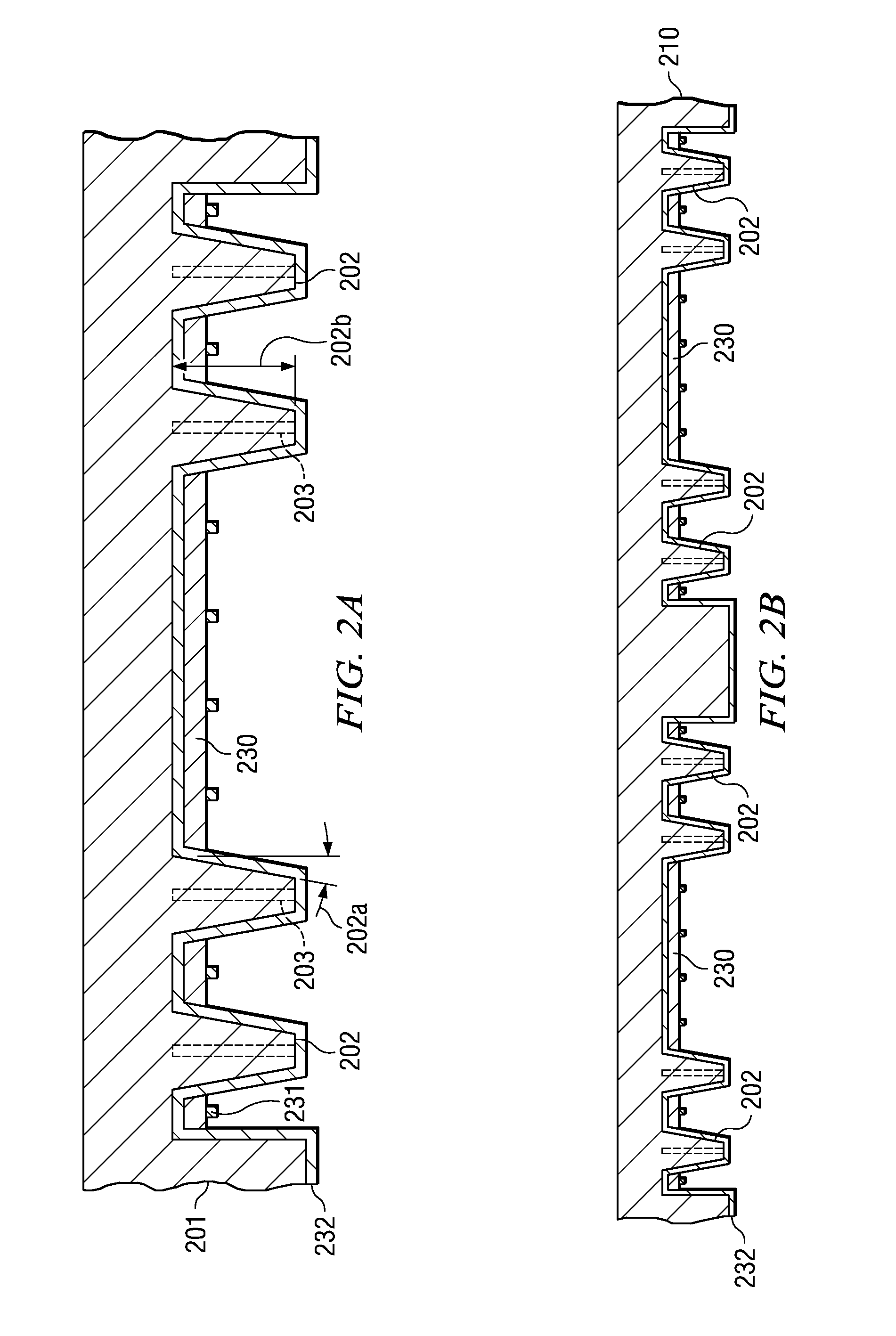
InactiveUS20080284045A1Low-cost and simplifyImprove testability and thus yieldLiquid surface applicatorsSemiconductor/solid-state device detailsContact padEngineering
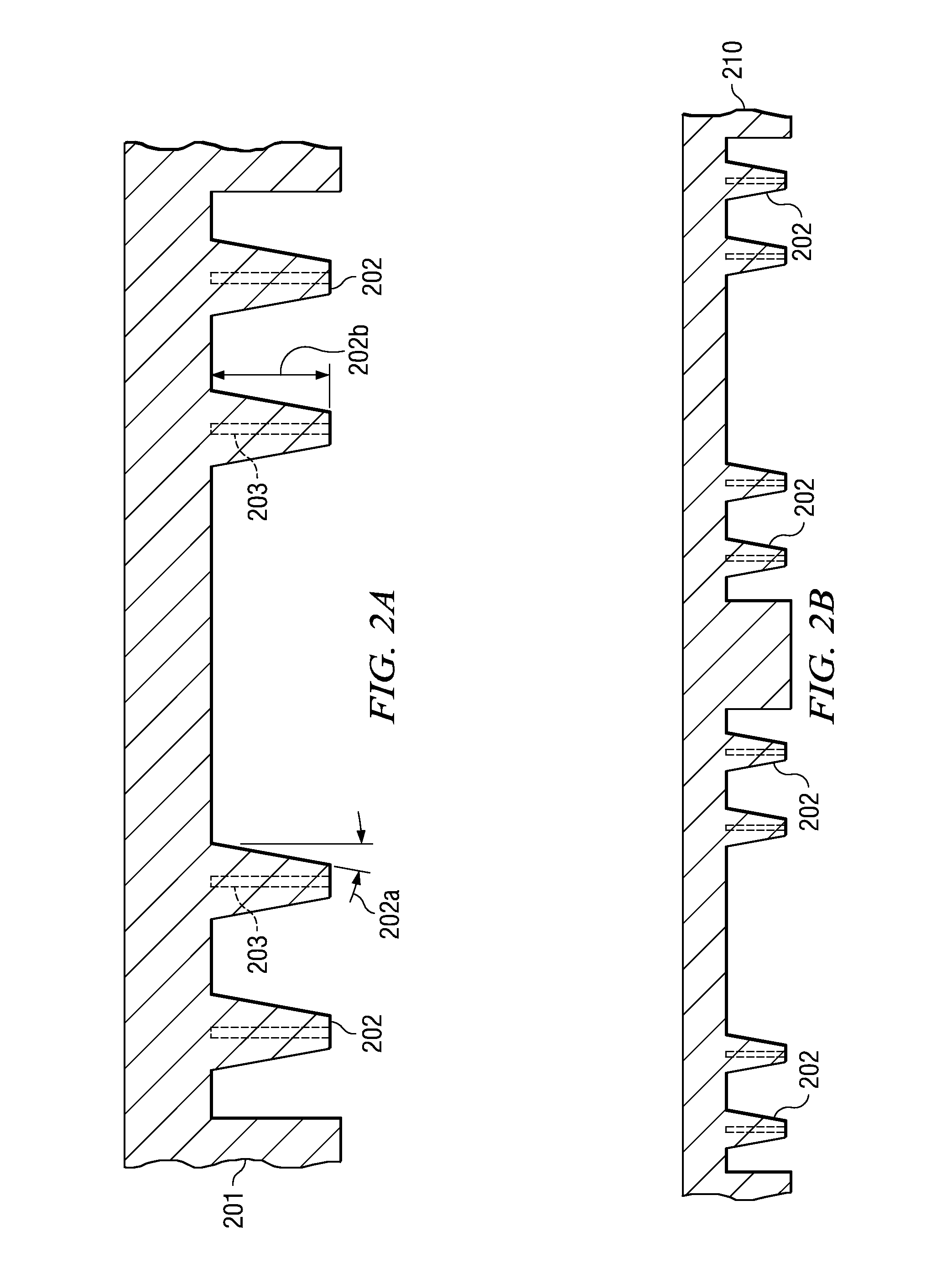
A method and apparatus for fabricating a semiconductor device are disclosed. The method attaches semiconductor chips (130) on a sheet-like insulating substrate (101) integral with two or more patterned layers of conductive lines and vias and with contact pads (103) in pad locations. A mold is provided, which has a top portion (210) with metal protrusions (202) at locations matching the pad locations. The protrusions are shaped as truncated cones of a height suitable to approach the pad metal surface in the closed mold cavity. The substrate and the chip are loaded onto the bottom mold portion (310); the mold is closed by clamping the top portion onto the bottom portion so that the protrusions are aligned with the contact pads, approaching the pad surface. After pressuring encapsulation compound into the cavity, the mold is opened; the encapsulated device has apertures to the pad locations. Any residual compound formed on the pads is removed by laser, plasma, or chemical to expose the metal surface.
Owner:TEXAS INSTR INC
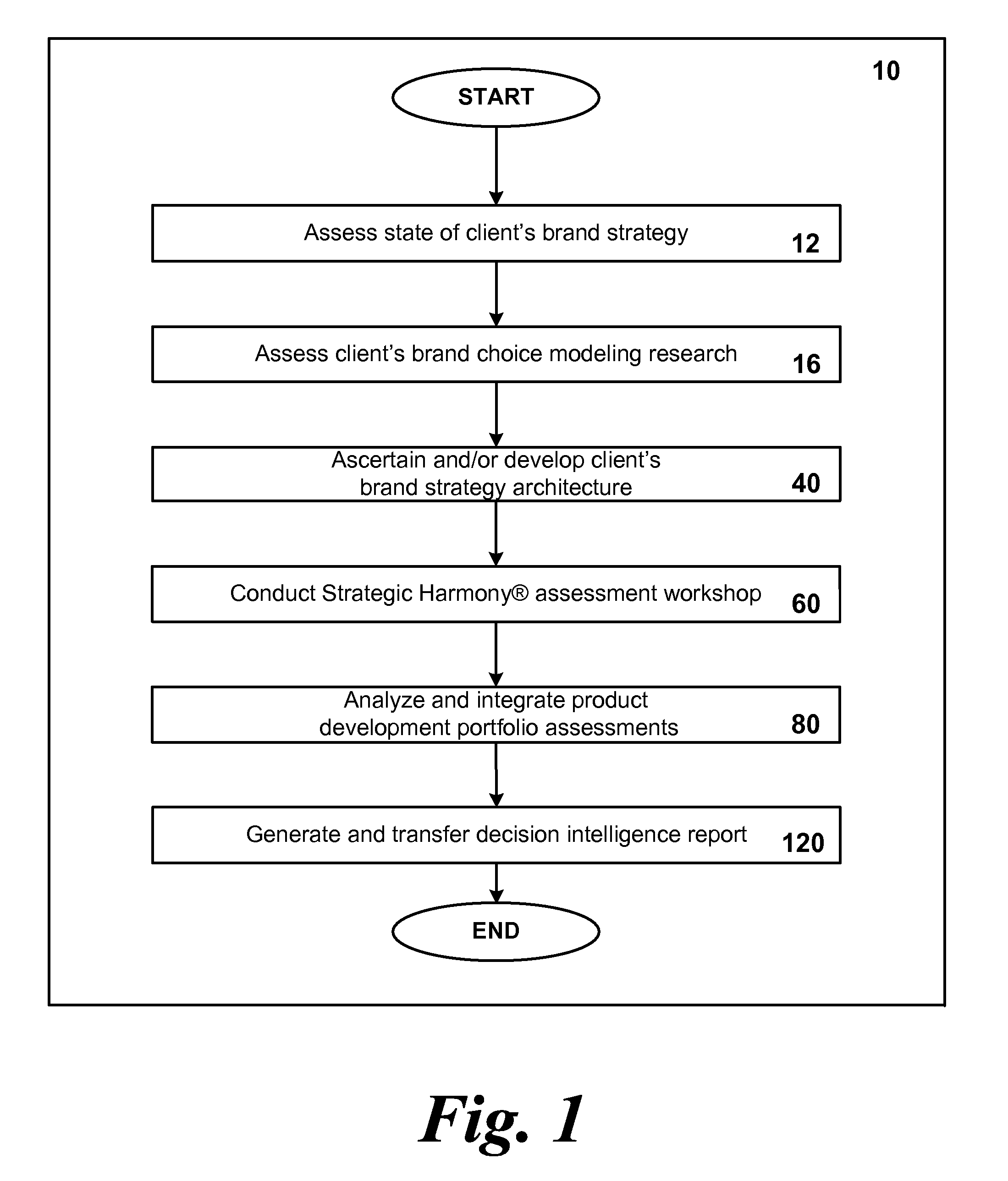
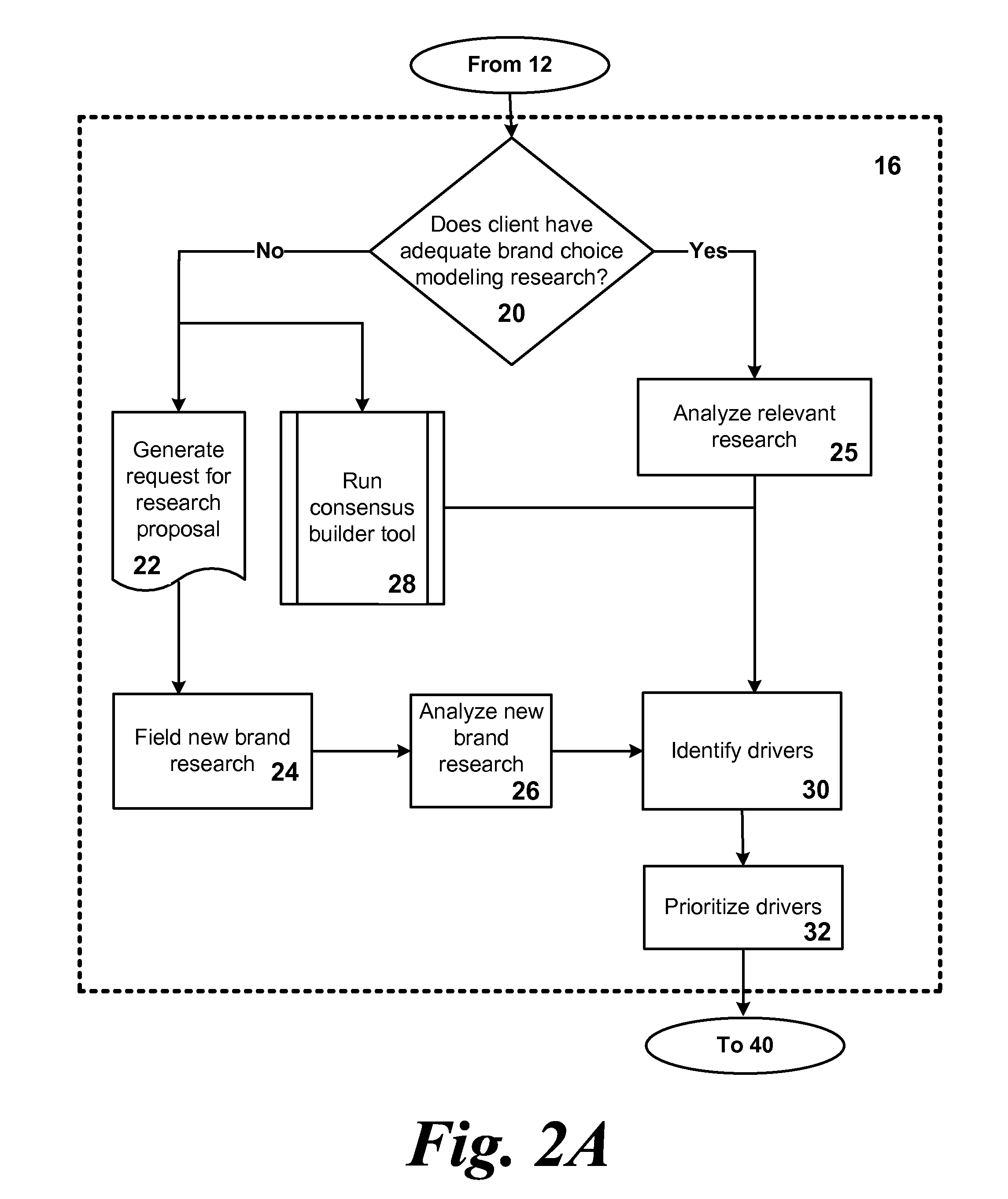
System and method for optimizing product development portfolios and integrating product strategy with brand strategy
InactiveUS20070192170A1Shorten the time to marketImprove business performanceFinanceResourcesTime to marketService composition
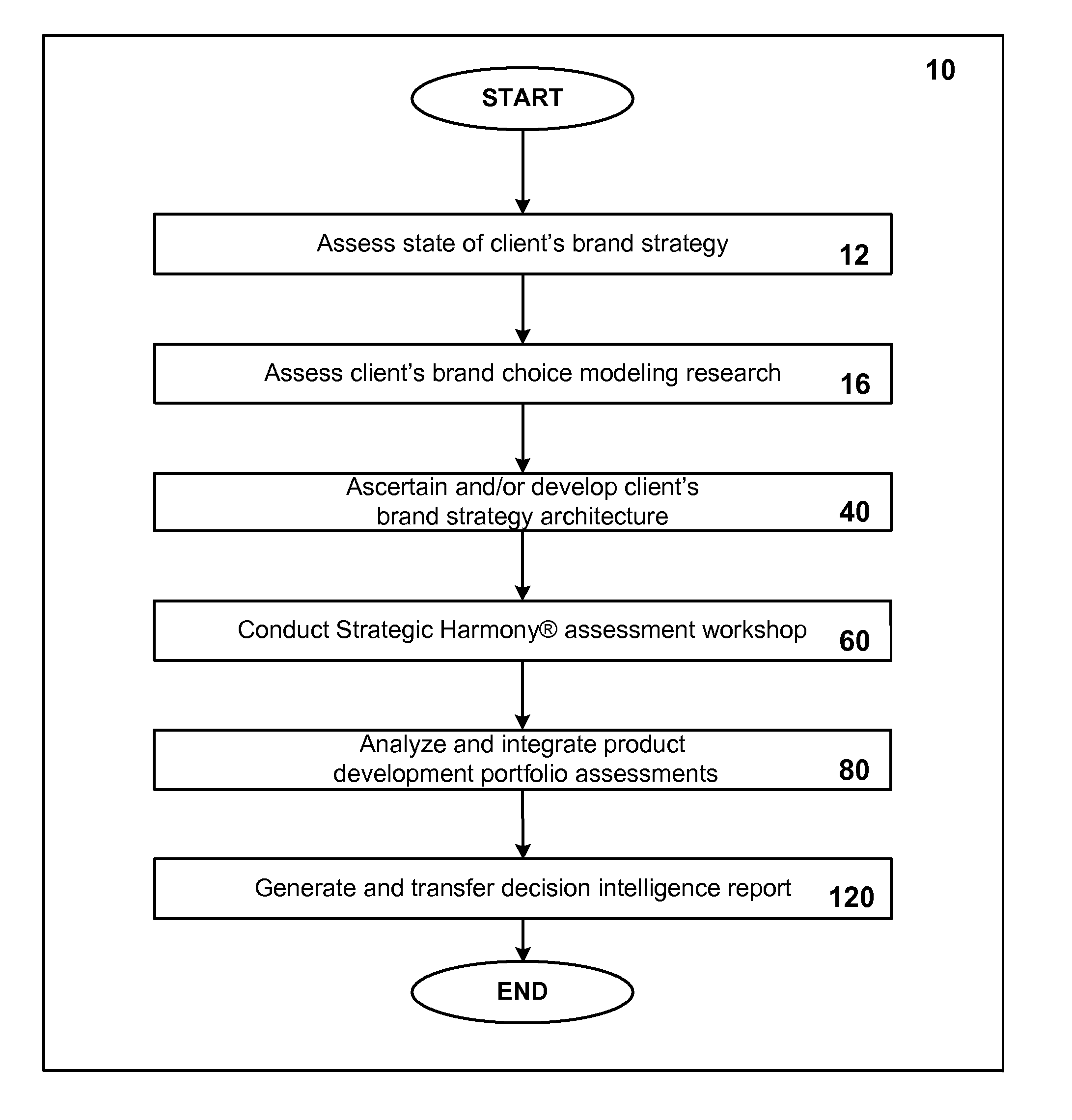
A business and software method to cost-effectively optimize product and / or service development portfolios, to reduce time to market, and to better integrate and align product or service strategy with brand strategy. The business and software method includes defining in detail the product and service attributes that characterize the ideal customer experience, categorizing the attributes, assigning a numerical value of importance to the attributes, and applying those values to statistical analysis of each assessed product development initiative in terms of alignment with ideal experience and potential competitive impact relative to the resources and risks required to bring each initiative to market. A prioritization for product development resource allocation is developed based upon these analyses. The prioritization is presented in the form of decision intelligence tools for an organization to use and reach informed judgments concerning resource allocation to develop, maintain, or optimize a given product or service portfolio. The decision intelligence tools serve to improve business performance, increase market impact, and build brand equity for products and services of a given organization by improving alignment between what the organization promises customers and what it actually delivers.
Owner:CRISTOL STEVEN M
Method and device for quality measuring of streaming media services
InactiveUS20140201330A1Easy to mergeLow costMultiple digital computer combinationsTransmissionMass parameterQuality measurement
Method and probe device for quality measuring in IP streaming of audio, video, or a synchronized mix of both, performing:receiving a streaming media flow at a user's end,measuring at least one network parameter which indicates QoS and / or QoE,extracting frames from the streaming at the user's end,analyzing the frames at the user's end by searching for determined errors and delivering at least a quality parameter defined by certain results of said searching;correlating each measured network parameter and each delivered quality parameter at the user's end and returning the results to the IP network operator though a control and configuration interface.The operator uses the control and configuration interface to configure at the user's end how to perform the correlation between the parameters, taking into account in said correlation the user's preferences described by an ontology.
Owner:TELEFONICA SA
Navigation system for a vehicle
InactiveUS20070162229A1Increase supplyImprove abilitiesInstruments for road network navigationRoad vehicles traffic controlTelecommunications linkGeolocation
An interior rearview mirror system for a vehicle includes an interior rearview mirror assembly having a reflective element. An information display is at the interior rearview mirror assembly and displays information for viewing by a driver of the vehicle. The information display may be located behind the reflective element and viewable through the reflective element by the driver of the vehicle. The mirror system includes a handheld device having a position locator and being capable of establishing a geographical position of the handheld device. The information displayed by the information display is derived at least in part via a wireless communication link with the handheld device. The information display may include a display of turn-by-turn information for viewing by the driver of the vehicle. The information display may comprise a video screen.
Owner:DONNELLY CORP
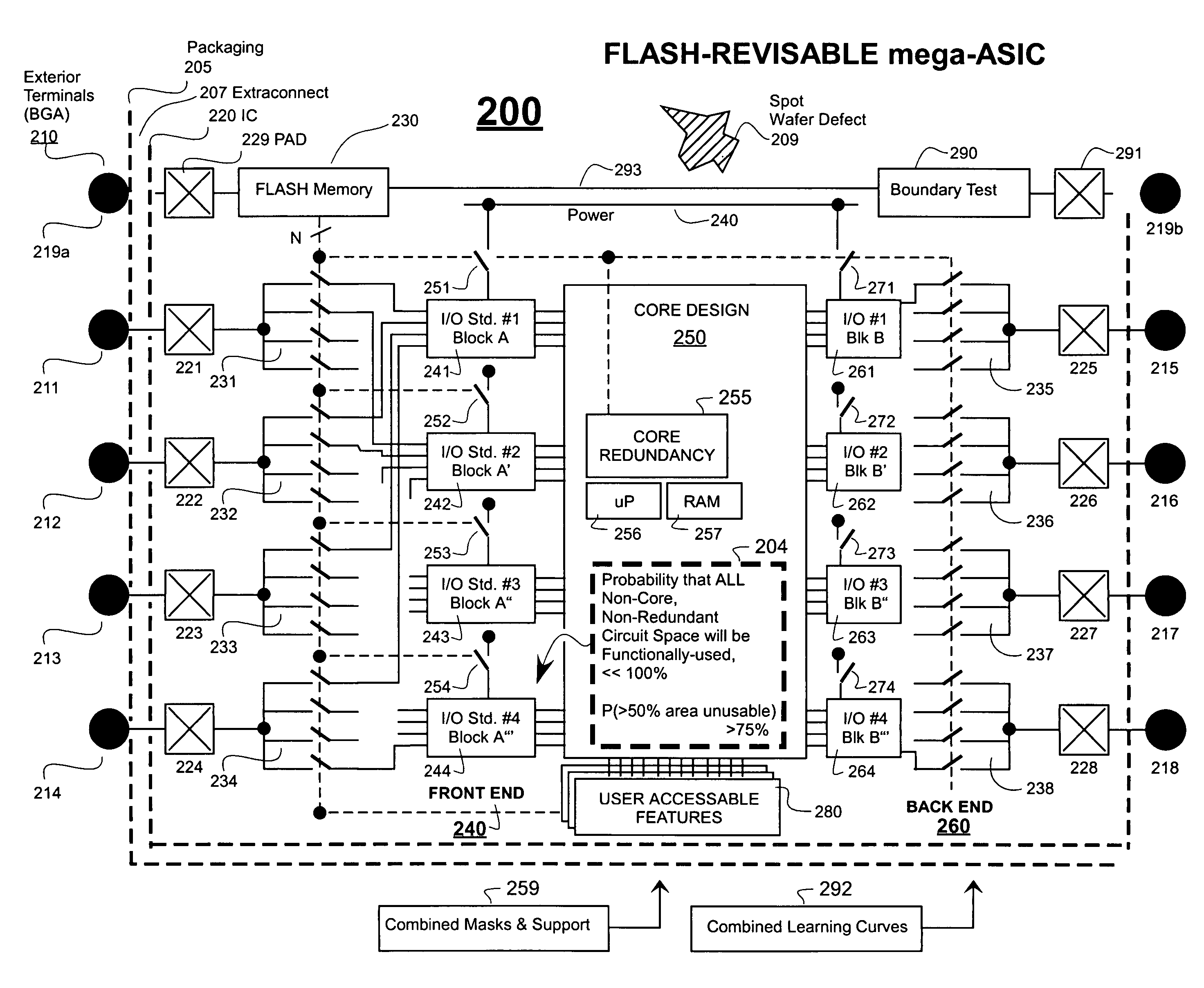
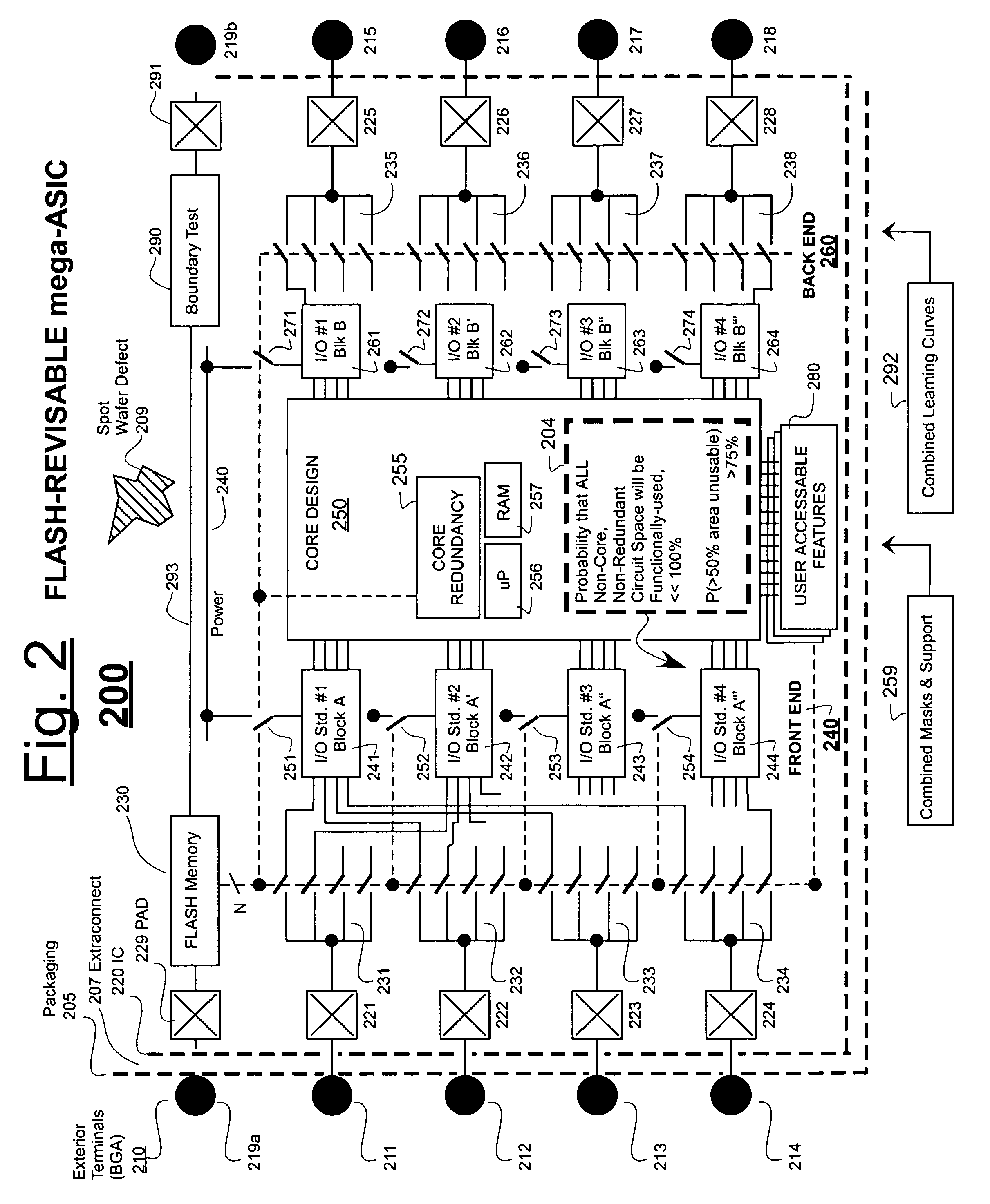
ASICs having more features than generally usable at one time and methods of use
ActiveUS7251805B2Offset drawbackShorten the time to marketSolid-state devicesCAD circuit designChipsetEmbedded system
Owner:SHEYU GROUP
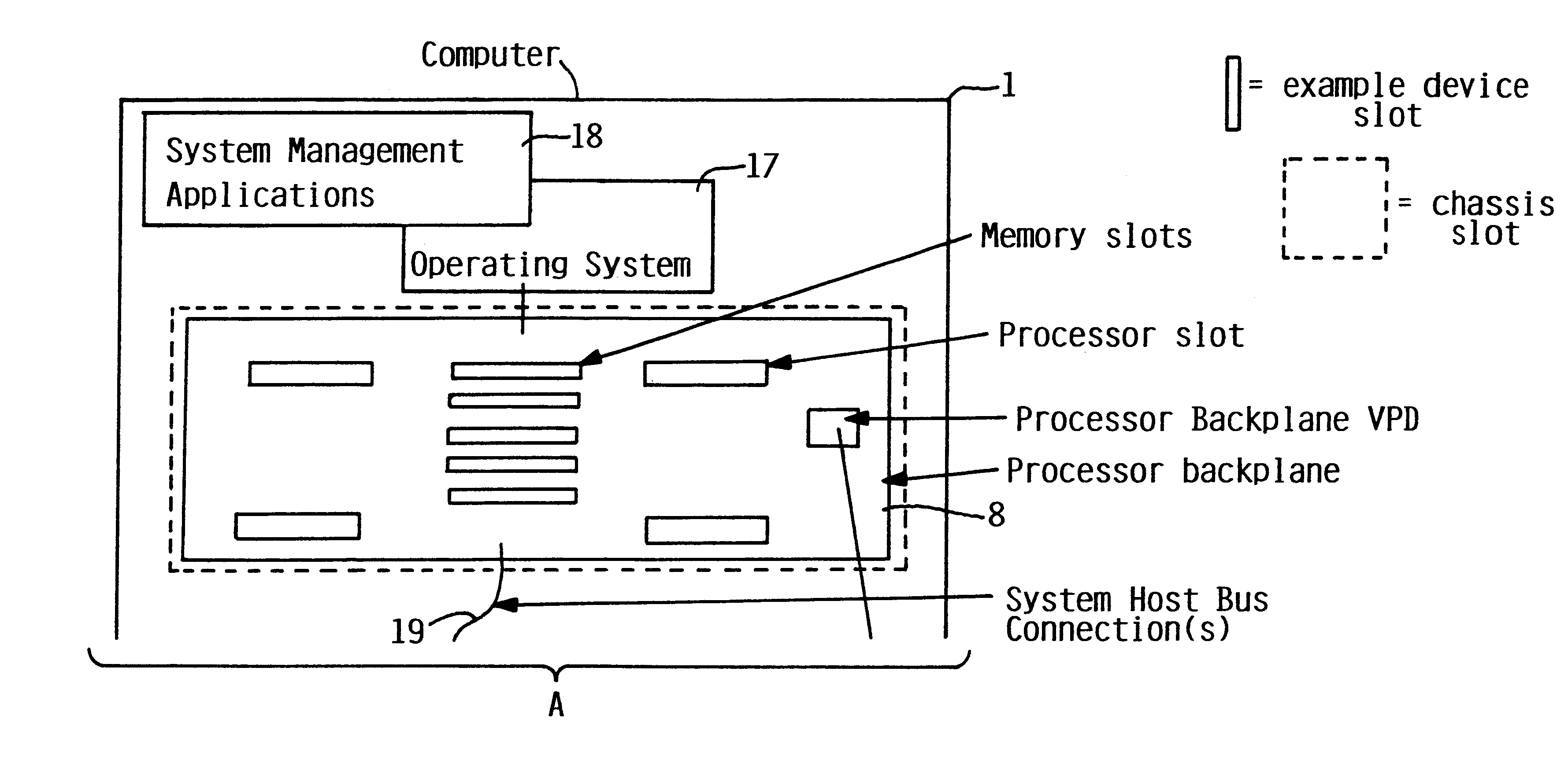
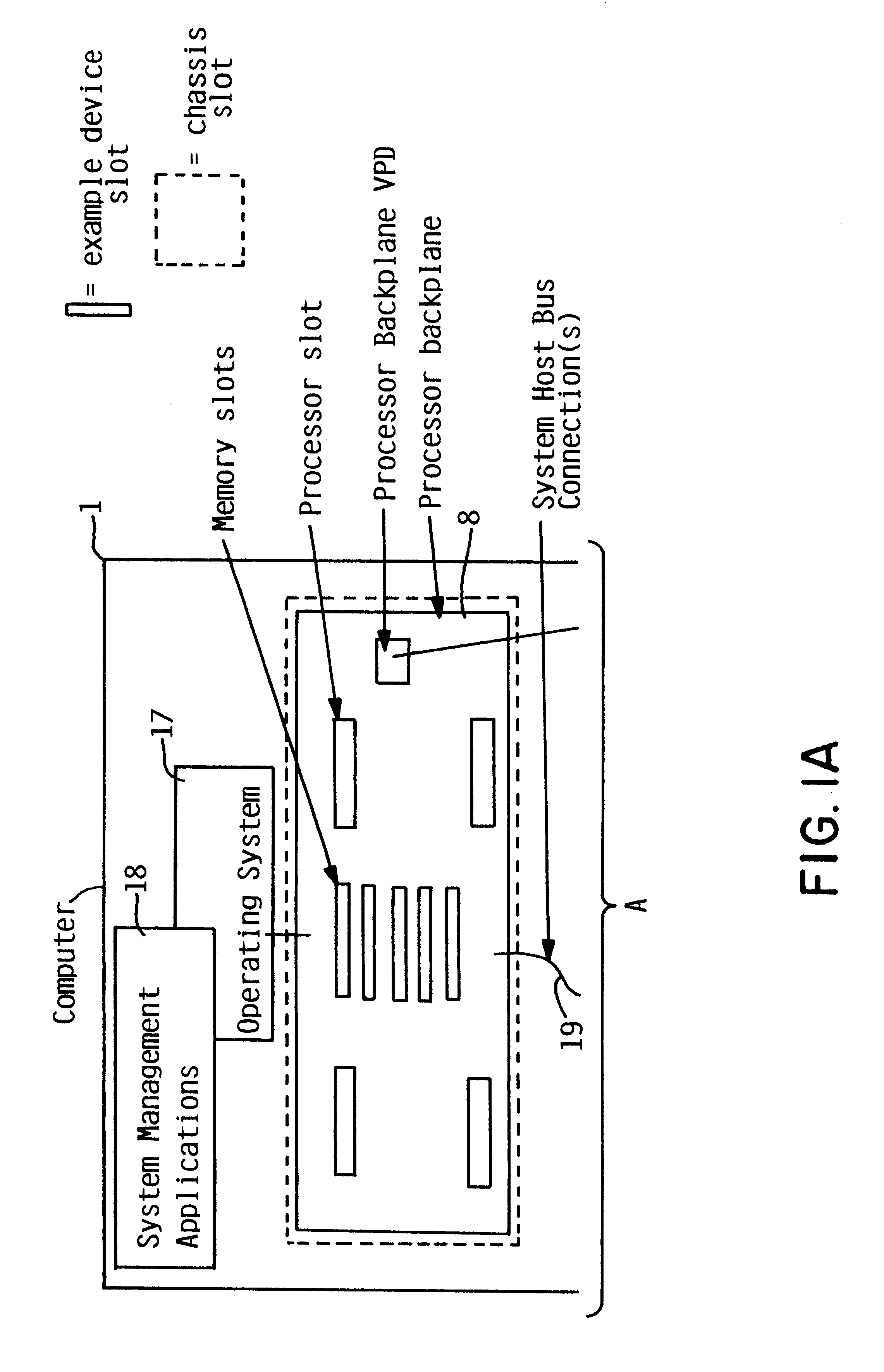
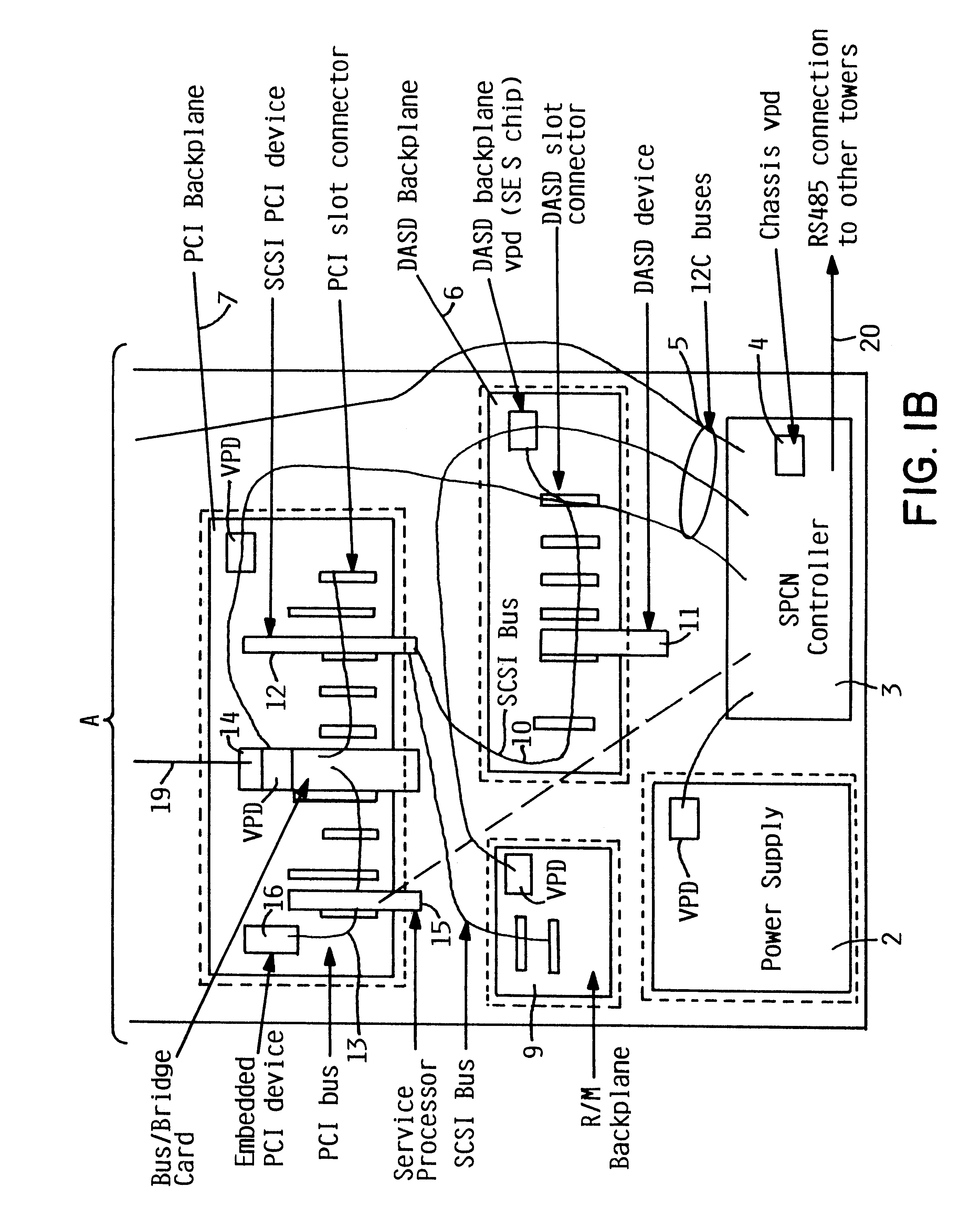
Addition of slot, backplane, chassis and device parametric properties to vital product data (VPD) in a computer system
InactiveUS6289405B1Easy to resolveEasily resolvedInput/output processes for data processingVital Product DataElectrical devices
In an electrical system having a chassis with at least one chassis slot for receiving a chassis electrical device, and at least one connector board with at least one board slot thereon for receiving a connector board electrical device therein, respective memory is provided for storing vital product parametric data associated with the at least one chassis slot, the at least one connector board slot, and the respective electrical devices. The stored vital product parametric data can be accessed by the electrical system to check compatibility.
Owner:IBM CORP
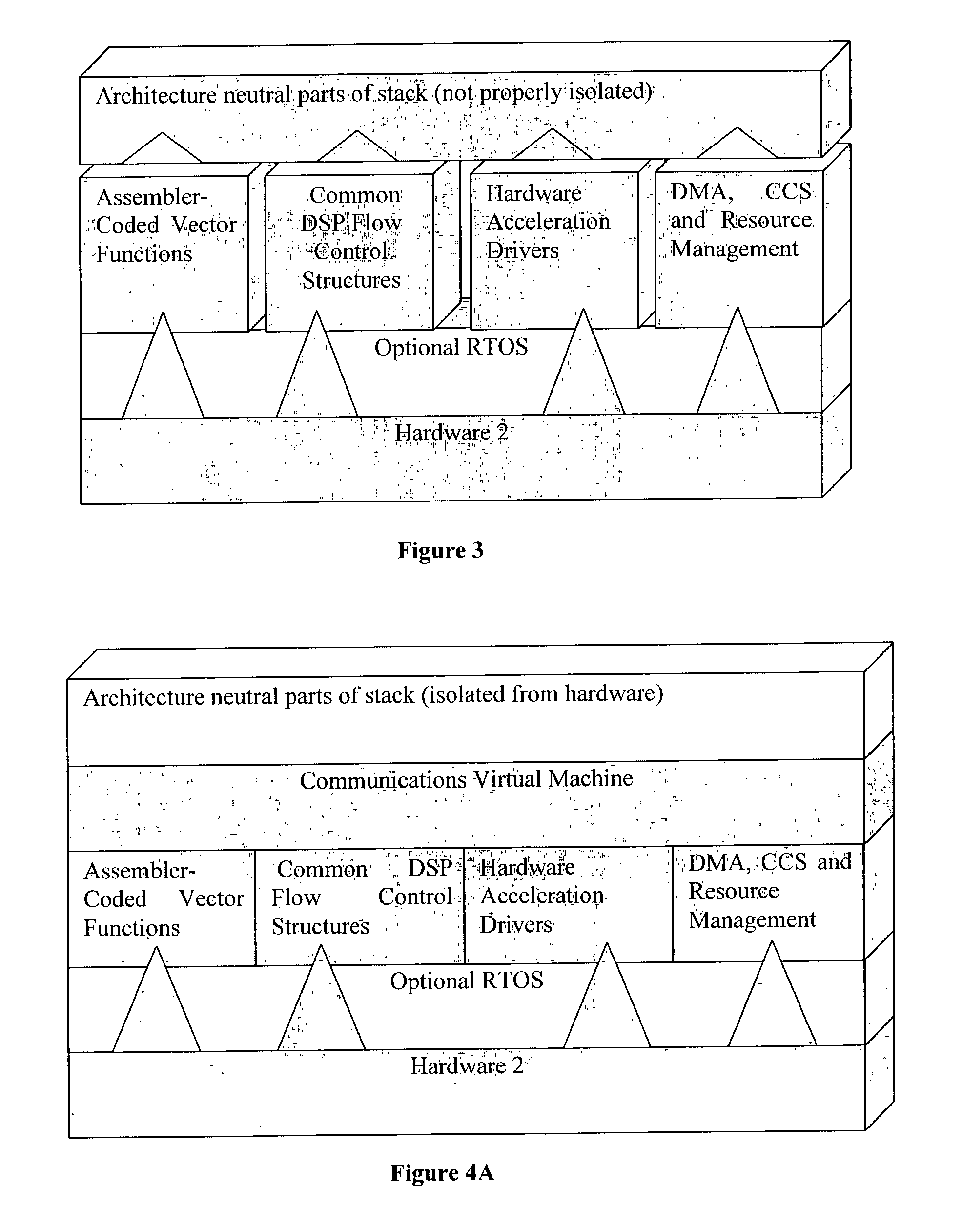
Method of designing, modelling or fabricating a communications baseband stack
InactiveUS20030004697A1Improve design qualityReduce chanceAnalogue computers for electric apparatusPhase-modulated carrier systemsComputer architectureBaseband
A method of designing, modelling or fabricating a communications baseband stack, comprising the steps of: (a) creating a description of one or more of the following parameters of the baseband stack: (i) resource requirements; (ii) capabilities; (iii) behavior; and (b) using that description as an input to software comprising a virtual machine layer optimised for a communications DSP in order to generate an emulation of the baseband stack to be designed, modelled or fabricated.
Owner:RADIOSCAPE
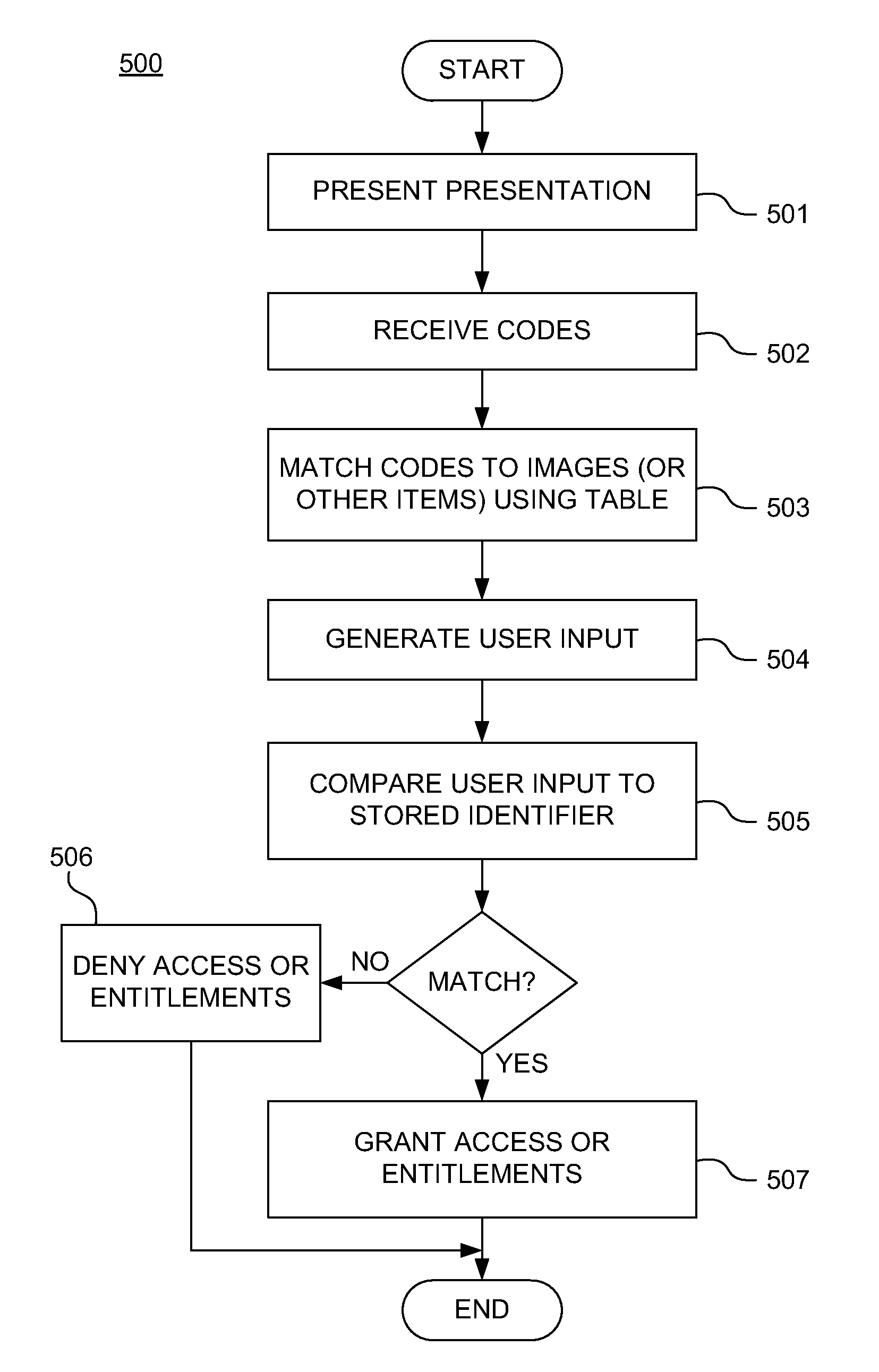
Adaptive policies and protections for securing financial transaction data at rest
ActiveUS20110314529A1Shorten the time to marketAcceptable levelDigital data processing detailsUser identity/authority verificationTransaction dataDisplay device
A system for challenge-response authentication is provided by receiving, from an external terminal over a communication network, a request for access to a service. A plurality of objects is presented to a user via a display. A plurality of codes is received over the communication network, each of the plurality of codes corresponding to one of the plurality of objects. The plurality of codes are matched to a plurality of alphanumeric characters according to a predetermined table. An alphanumeric string is generated from the plurality of alphanumeric characters and the alphanumeric string is compared to a user identifier stored in a database. Based on the comparing, a determination is made as to whether to grant the user access to the service.
Owner:AMERICAN EXPRESS TRAVEL RELATED SERVICES CO INC
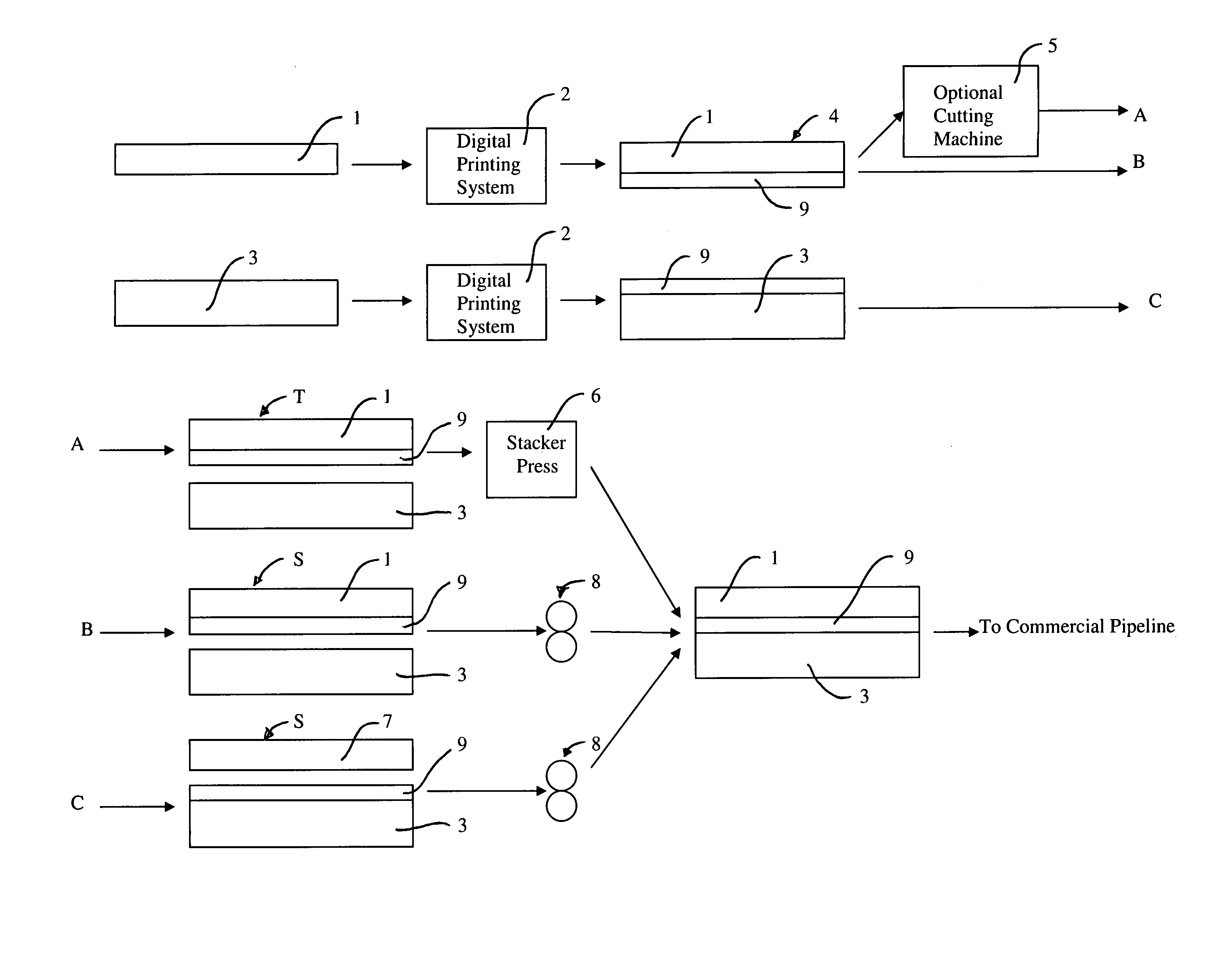
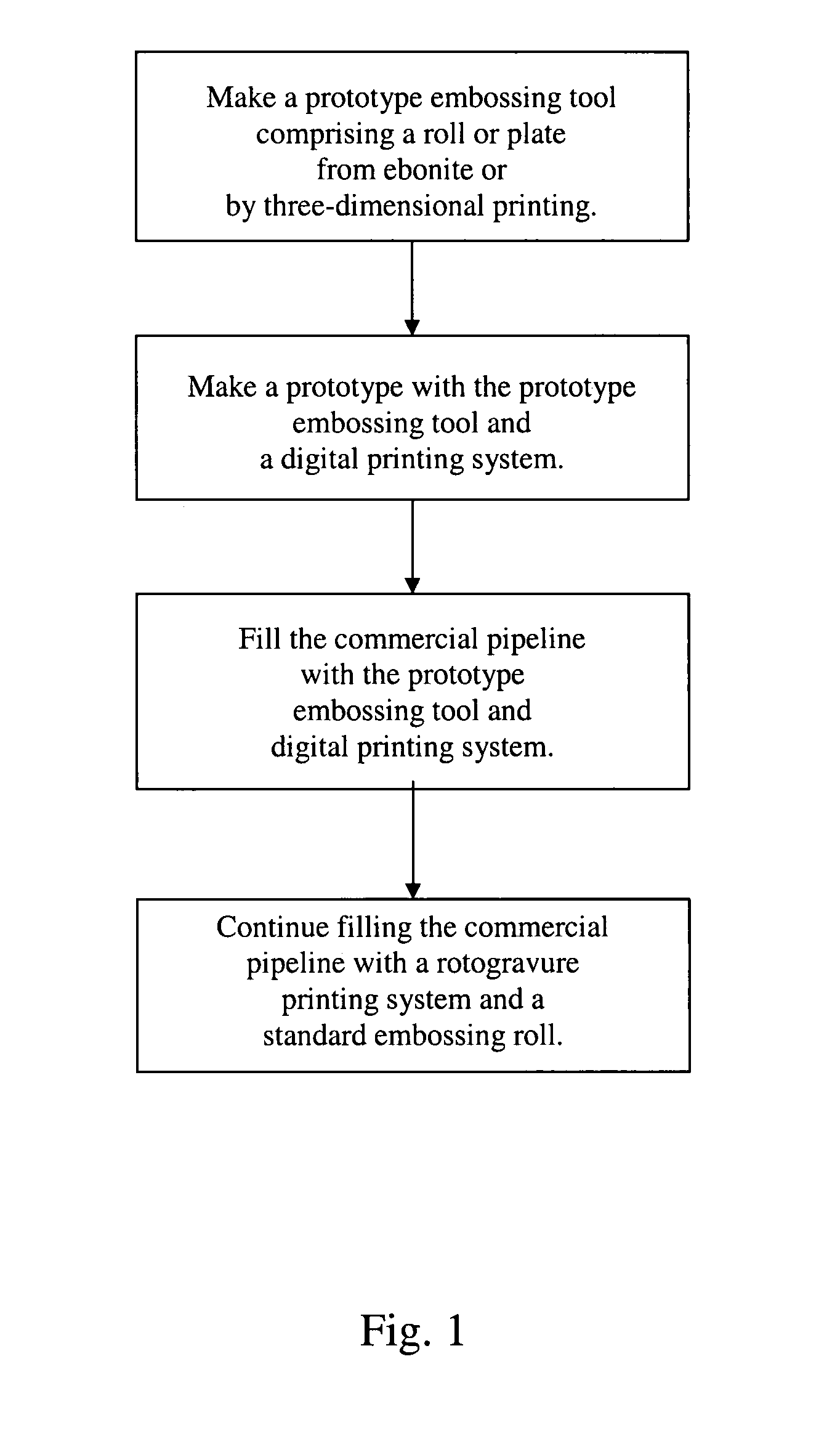
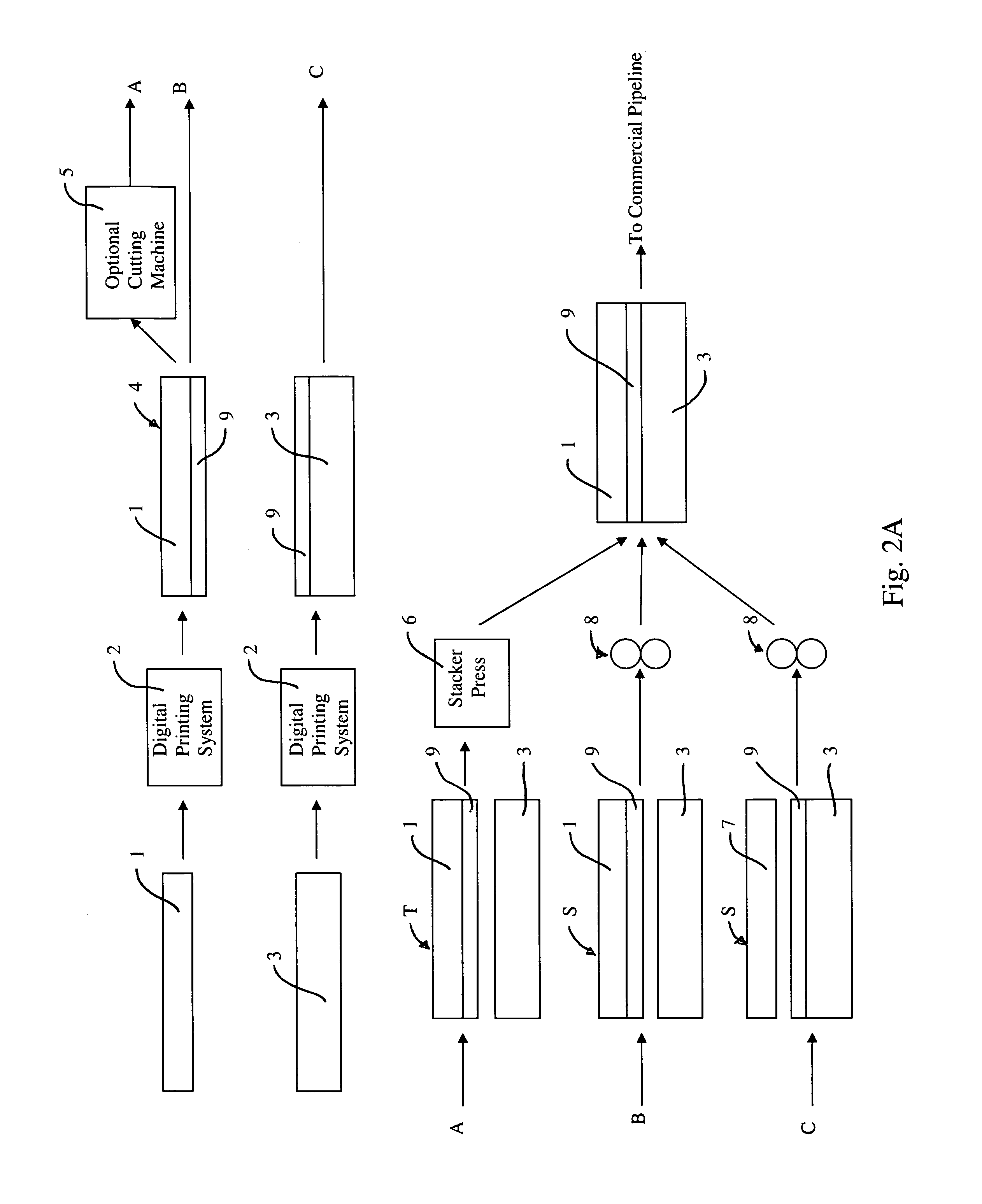
Rapid prototyping and filling commercial pipeline
InactiveUS7383768B2Quick changeCheap and quickAdditive manufacturing apparatusMechanical working/deformationEngineeringPrinting press
Methods and apparatus provide a means to rapidly develop and modify prototype surface coverings for customer review and approval, and fill the commercial pipeline while more conventional production equipment is obtained, installed and tested. The present apparatus to rapid prototype and fill the commercial pipeline includes a digital printing system to print a film and a press for laminating and embossing the printed film to a substrate. The press uses an embossing plate or roll, which is made from ebonite or by three-dimensional printing equipment. These plates and rolls can be produced within two or three days or less.
Owner:AFI LICENSING
Array molded package-on-package having redistribution lines
ActiveUS7944034B2Shorten the time to marketSimple methodLiquid surface applicatorsSemiconductor/solid-state device detailsContact padCoupling
A semiconductor device with a sheet-like insulating substrate (101) integral with two or more patterned layers of conductive lines and vias, a chip attached to an assembly site, and contact pads (103) in pad locations has an encapsulated region on the top surface of the substrate, extending to the edge of the substrate, enclosing the chip, and having contact apertures (703) at the pad locations for external communication with the pad metal surfaces. The apertures may have not-smooth sidewall surfaces and may be filled with solder material (704) to contact the pads. Metal-filled surface grooves (710) in the encapsulated region, with smooth groove bottom and sidewalls, are selected to serve as customized routing interconnections, or redistribution lines, between selected apertures and thus to facilitate the coupling with another semiconductor device to form a package-on-package assembly.
Owner:TEXAS INSTR INC
Publication of informational messages to software applications in a computing environment
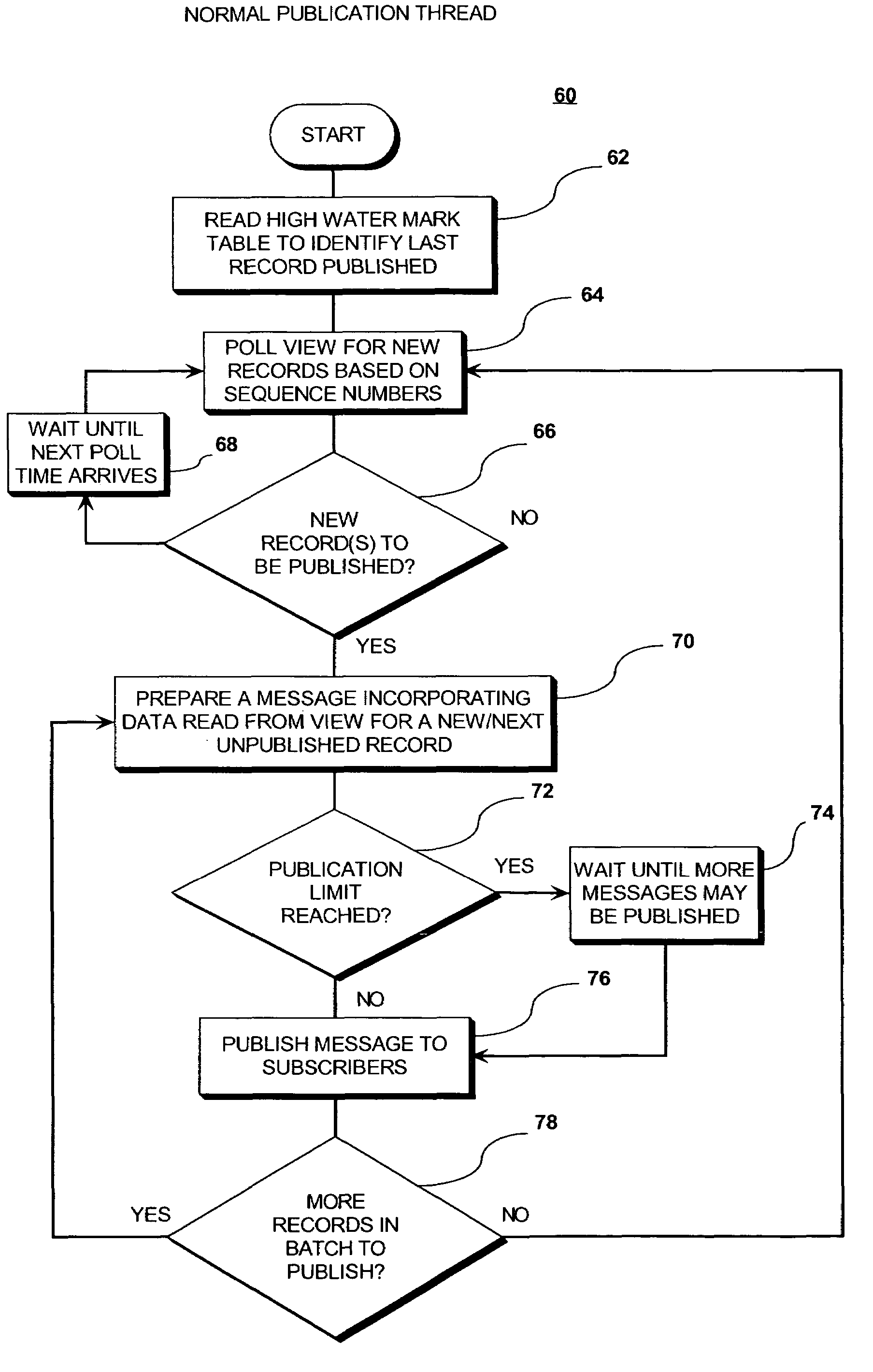
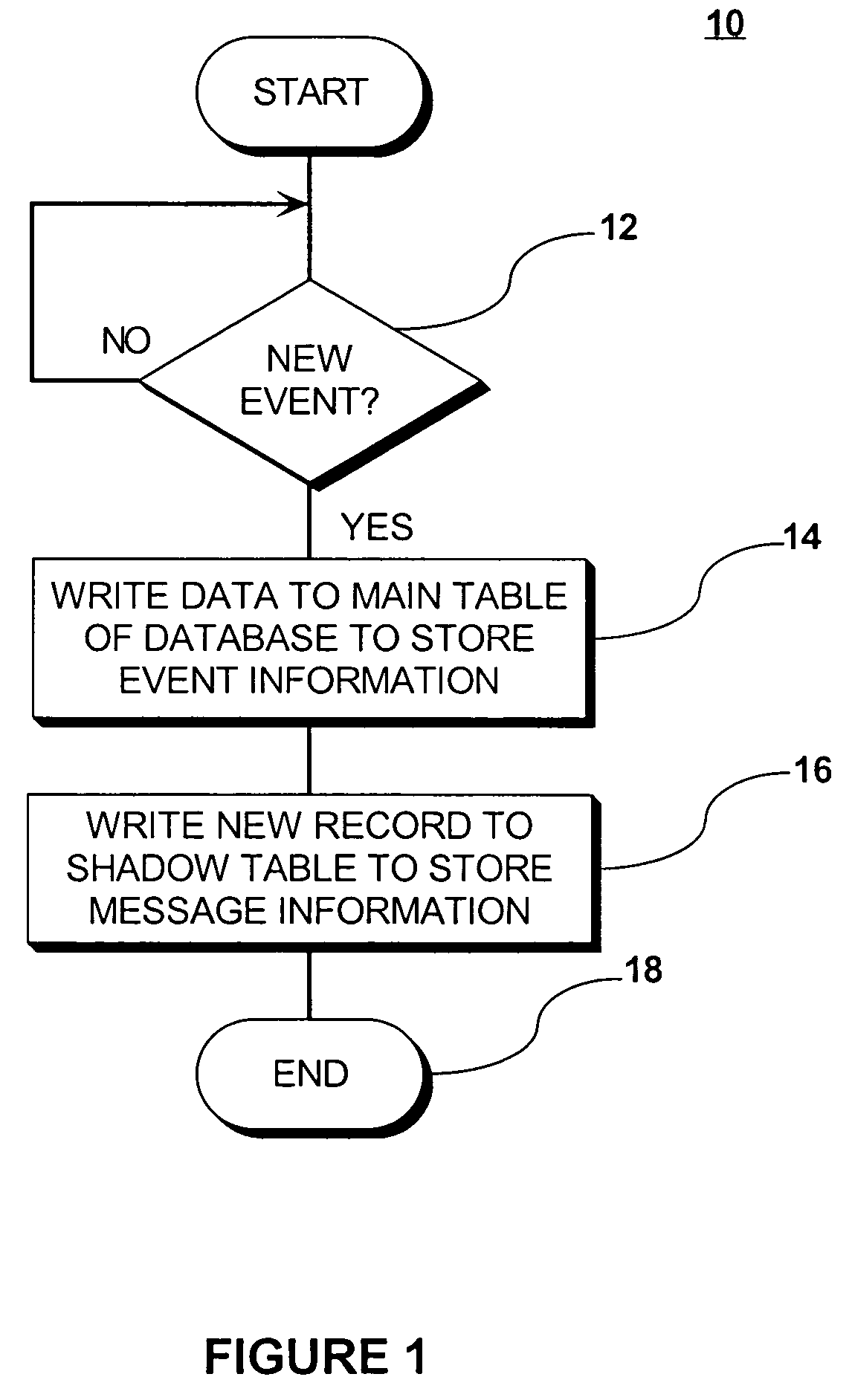
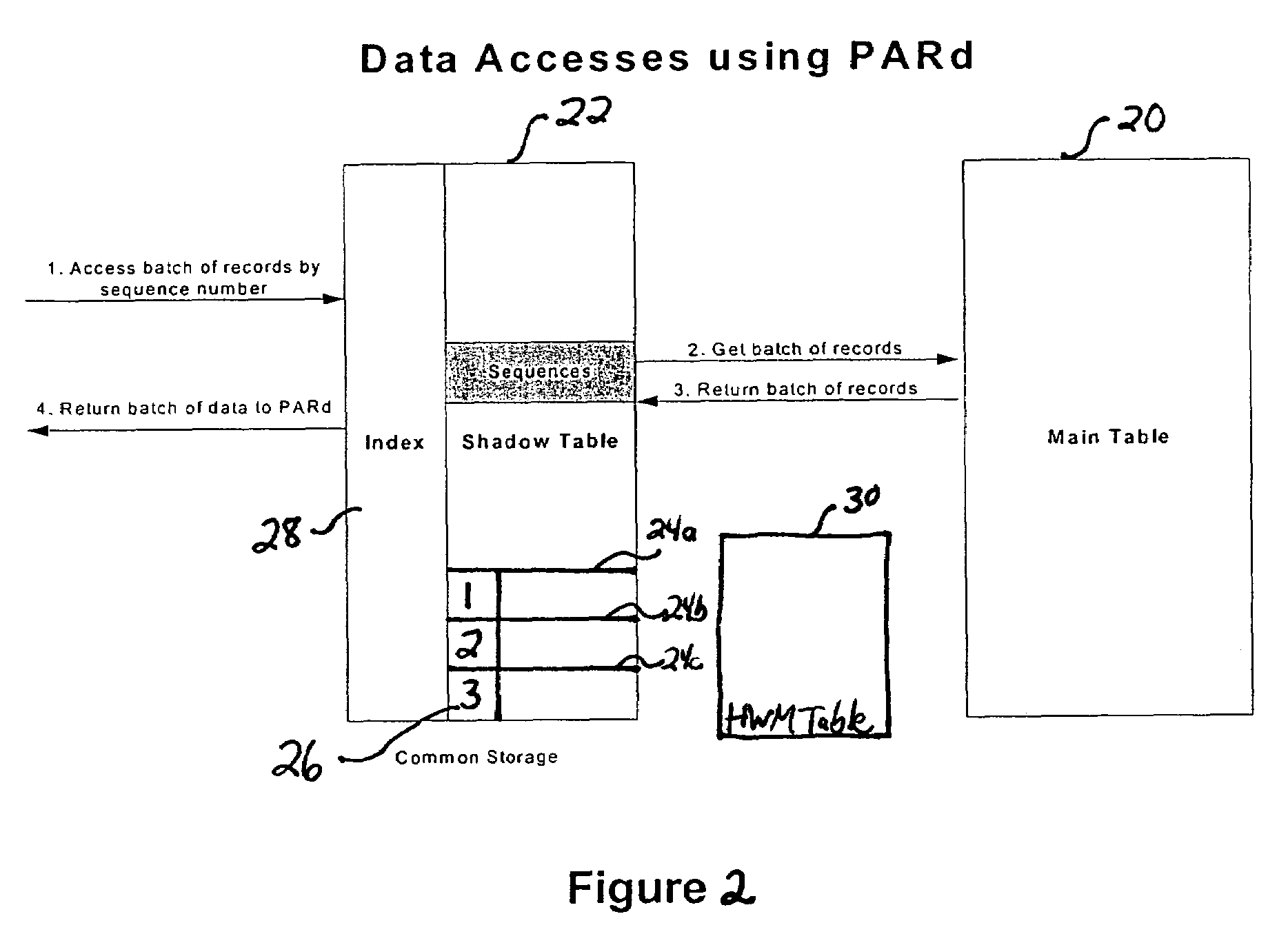
InactiveUS7571195B2Eliminate needEasy to eliminateOffice automationBuying/selling/leasing transactionsMessage structureData shipping
A “push” based publication model in which messages are sent automatically to communicate event related data to other system applications when an event occurs that causes a change to a persistent data store. A shadow table stores records relating to information stored in the main table. Each record is assigned a sequentially ordered identification code. A high water mark table stores the identification number of the last message published for a group of messages, and thus points to where publication last stopped. By using sequentially ordered codes, it is readily apparent from a gap in the sequence codes whether a record / message is missing. The shadow table may be configured as an Index Ordered Table to eliminate a query “hop” from the index table to the shadow table. The persistent store is queried to identify changes in persistent store structure, and the message structure is modified automatically to reflect any changes.
Owner:EBAY INC
Common interface framework for developing field programmable device based applications independent of a target circuit board
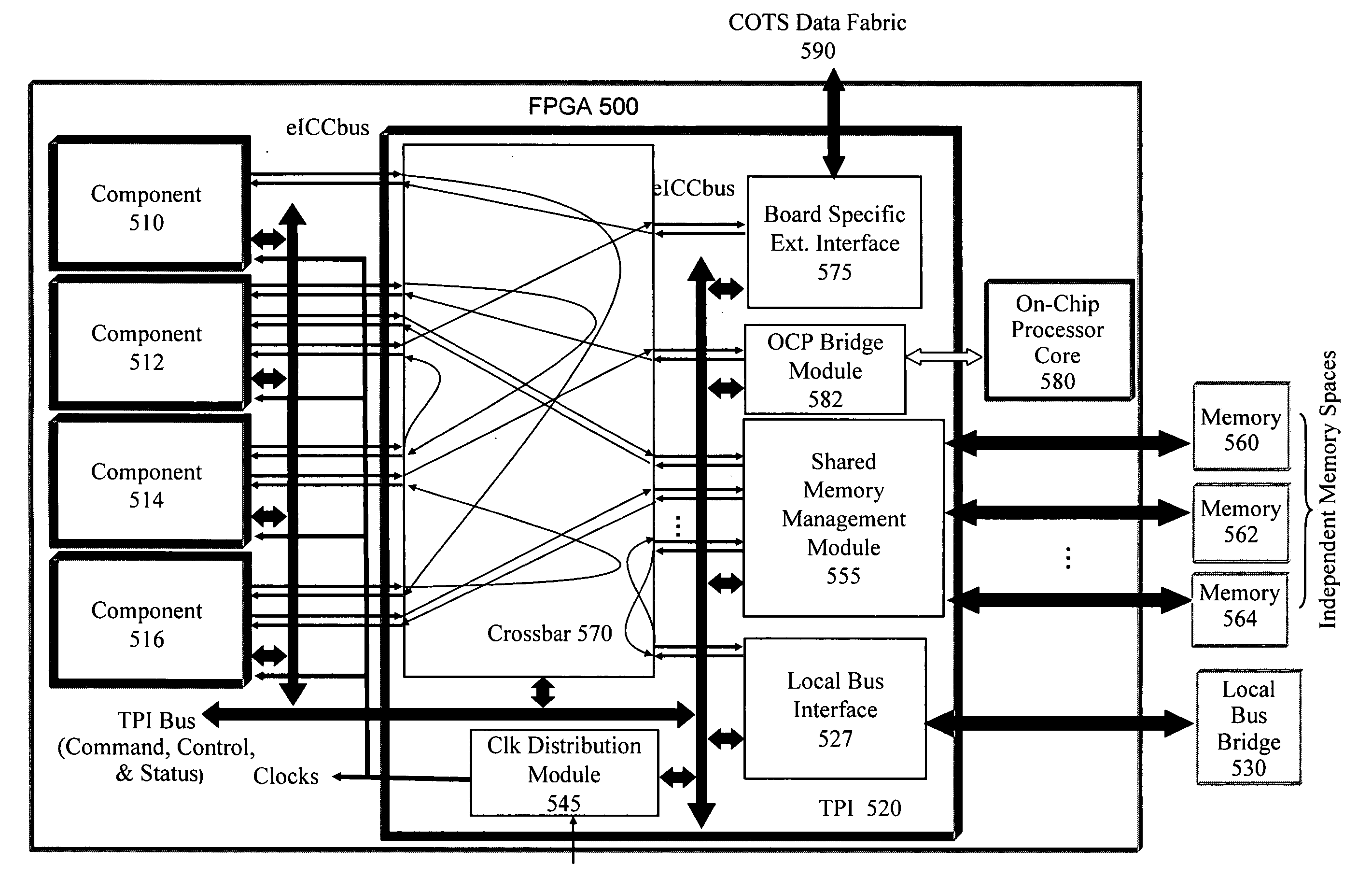
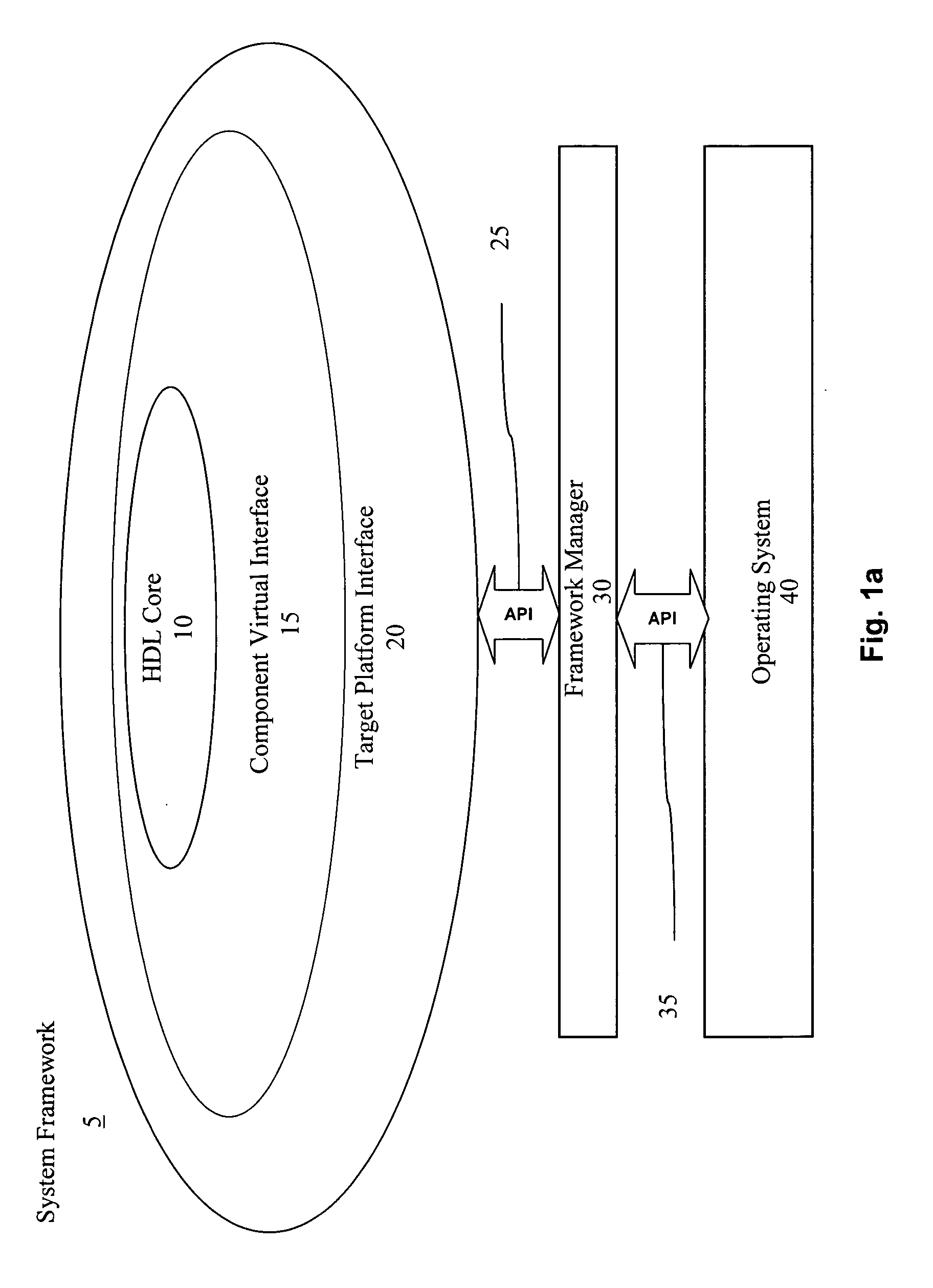
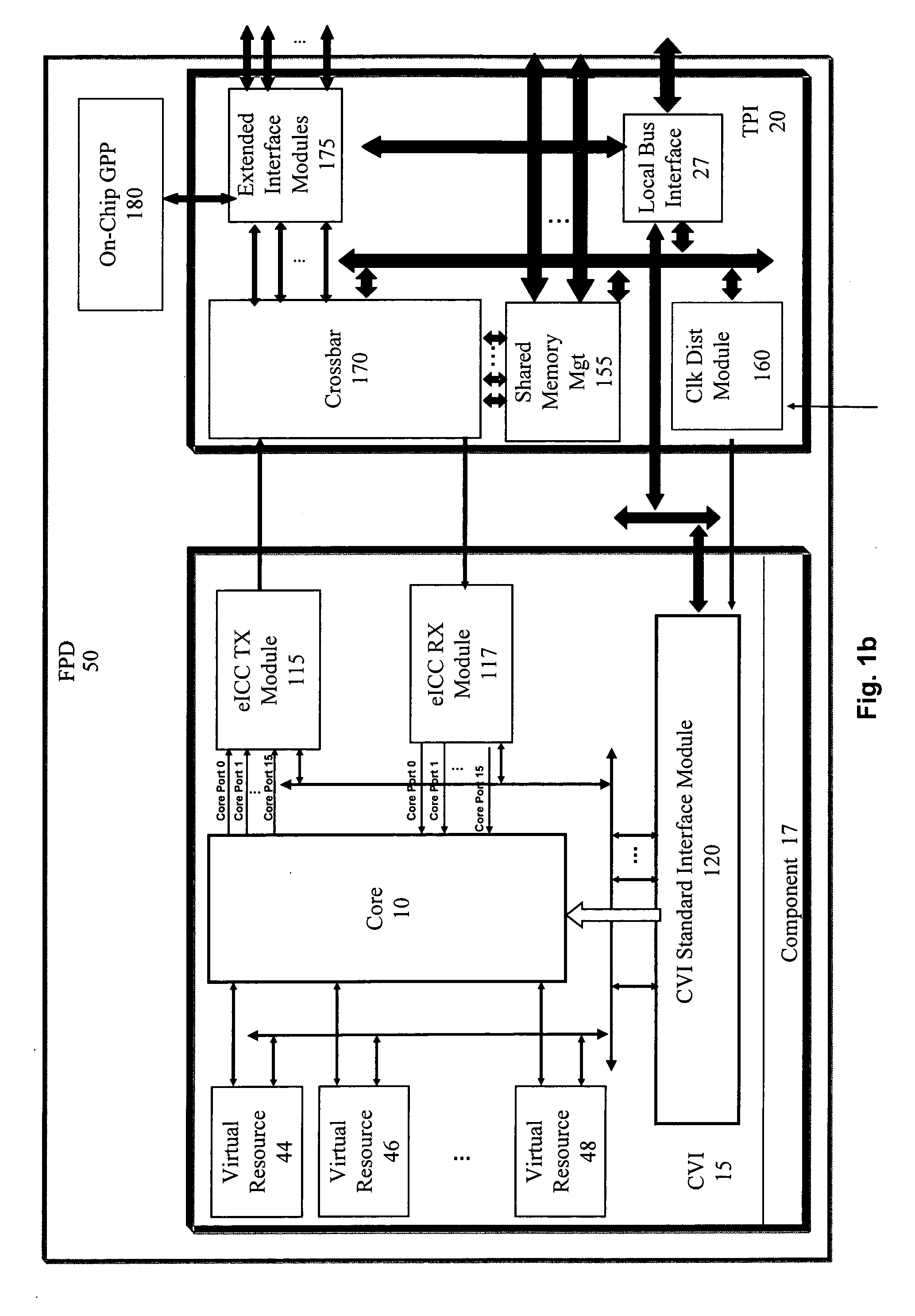
ActiveUS20060190904A1Raise the possibilityShorten the time to marketCAD circuit designSpecific program execution arrangementsComputer hardwareOperational system
A multi-level framework that allows an application to be developed independent of the chip or board, and any dependency is built in as part of the framework of the field programmable device (FPD). According to one embodiment, a field programmable device (FPD) comprises at least one hardware design language (HDL) application core with at least one component virtual interface respectively coupled to the HDL core, wherein the HDL core and the component virtual interface form a component, and wherein the component virtual interface has a plurality of pre-defined interfaces for communicating with the core. The component is hardware independent of the FPD. There is a target platform interface coupled to the component virtual interface wherein the target platform interface incorporates hardware specific information. A framework manager is coupled to the target platform interface and communicates with at least one operating system.
Owner:CALLAHAN CELLULAR L L C
Distributed software development tool
ActiveUS7331034B2Shorten the time to marketVersion controlComputer security arrangementsSoftware development processThe Internet
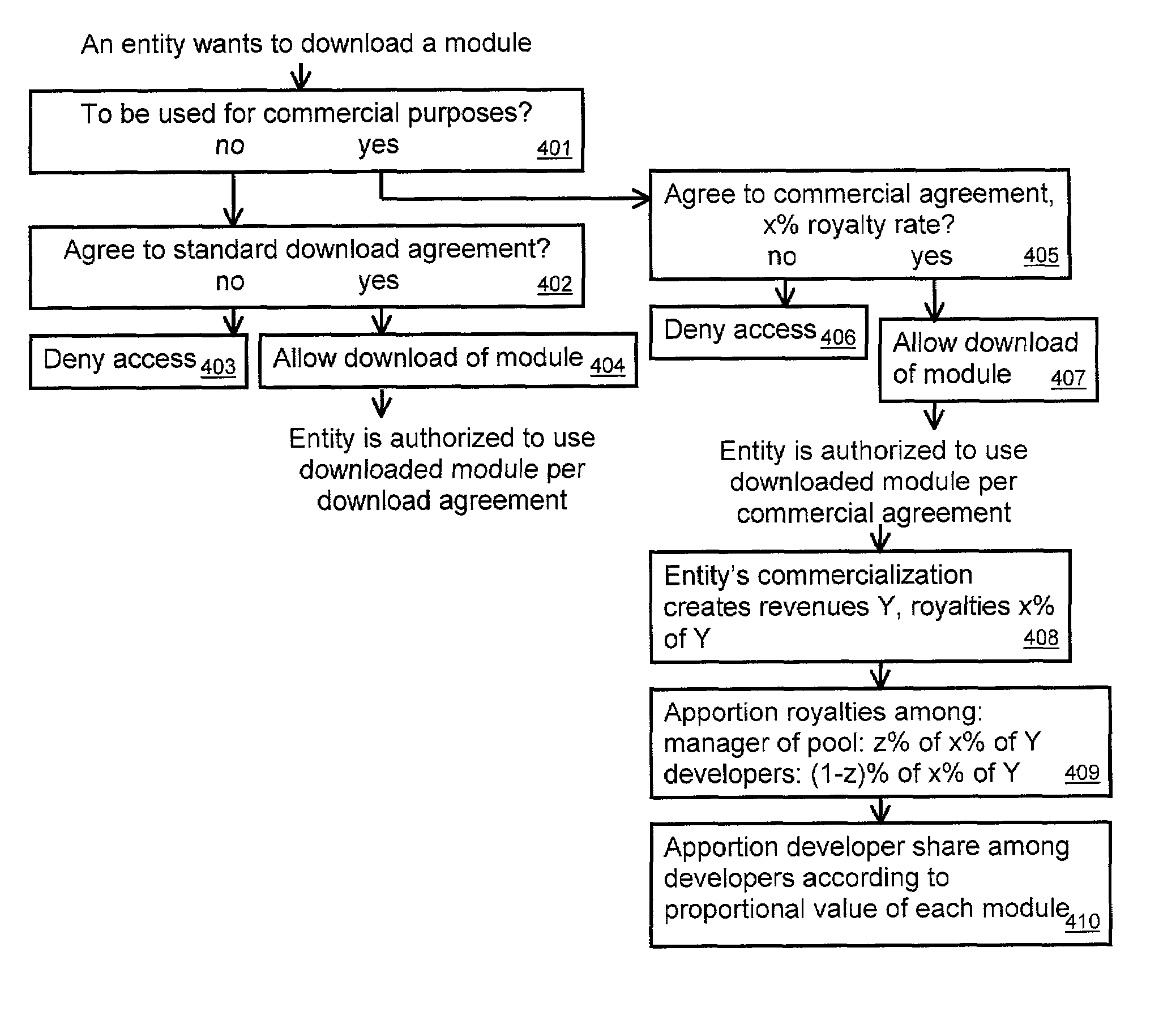
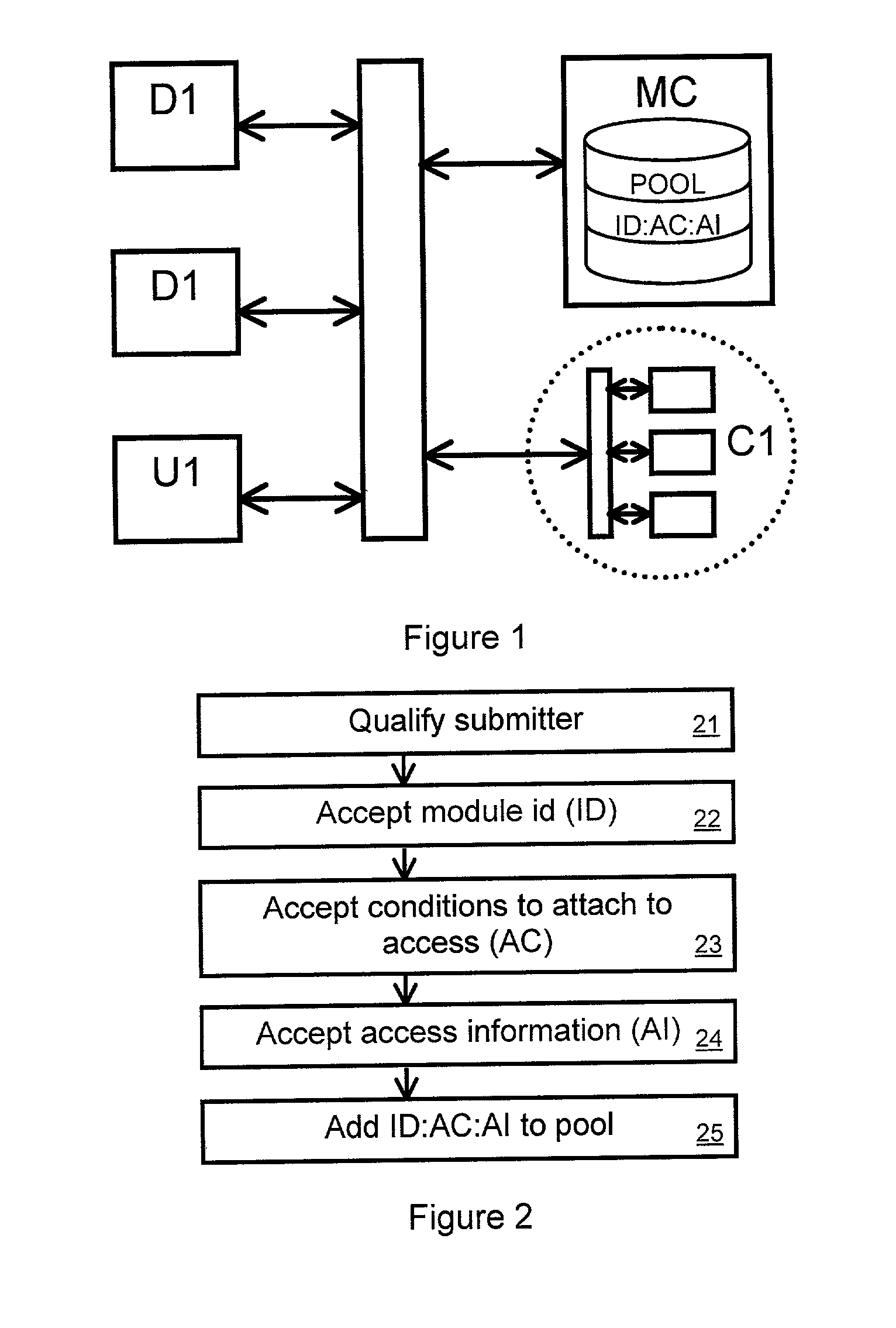
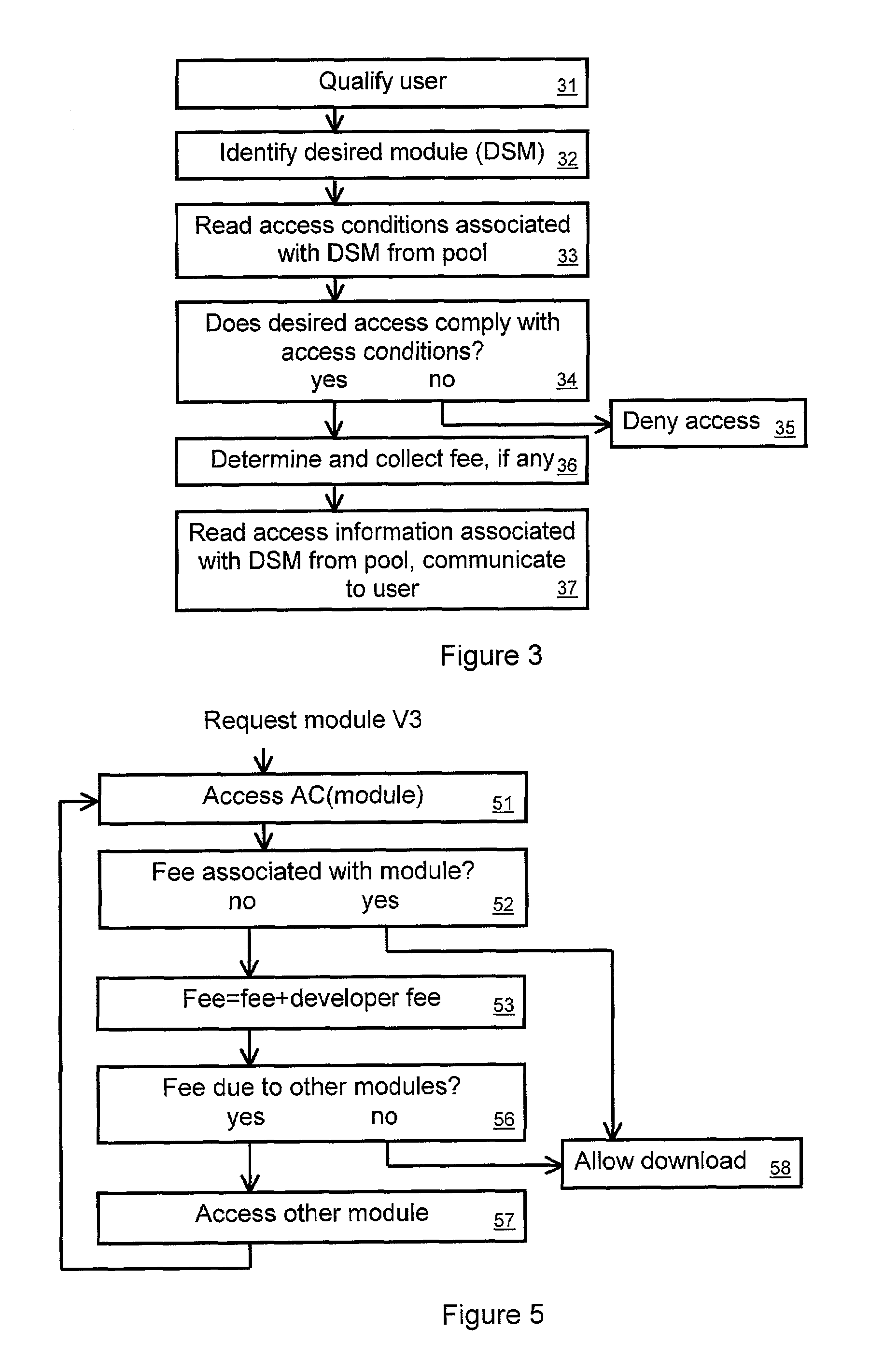
A tool for distributed software development maintains a pool of software modules. A module manager manages submissions to and downloads from the pool. The module manager can be a computer system with programming and resources suited for managing the pool. Developers and users can access the module manager via a computer network such as the internet, or via other transmission mechanisms. A developer intending to submit a module to the pool can specify access conditions associated with the module, and transmit information needed to access the module. The module manager can add the access information and access conditions to the pool. A developer or user intending to access a module from the pool can identify a desired module. The module manager can then determine if the intended access complies with the access conditions associated with the desired module. If it does, then the module manager can transmit the access information.
Owner:LEVERAGE ROCK LLC
Supply chain control method and system and storage medium
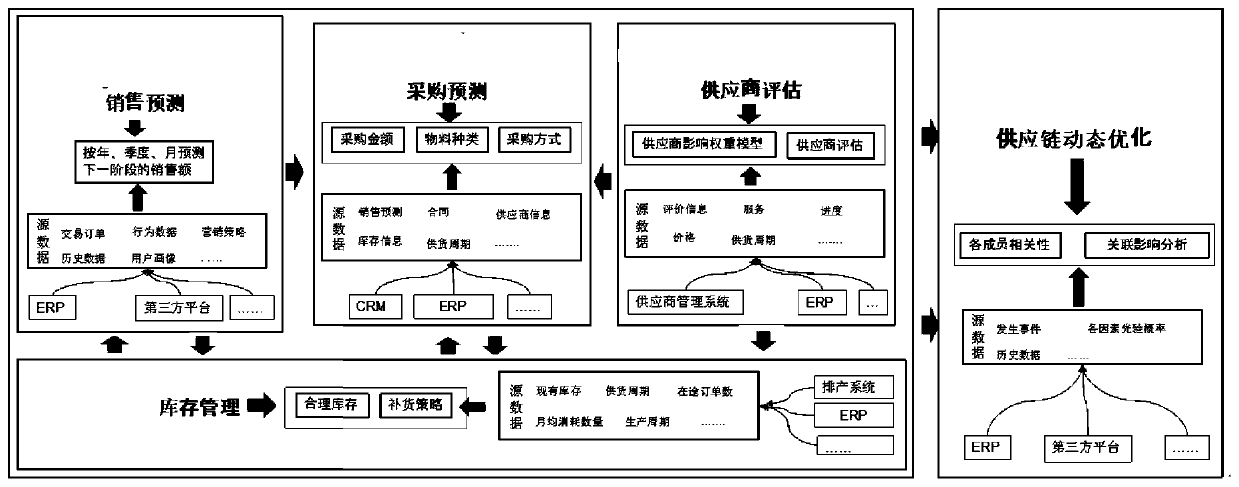
ActiveCN109784806AImprove operational efficiencyLow running costCharacter and pattern recognitionNeural architecturesData informationInventory management
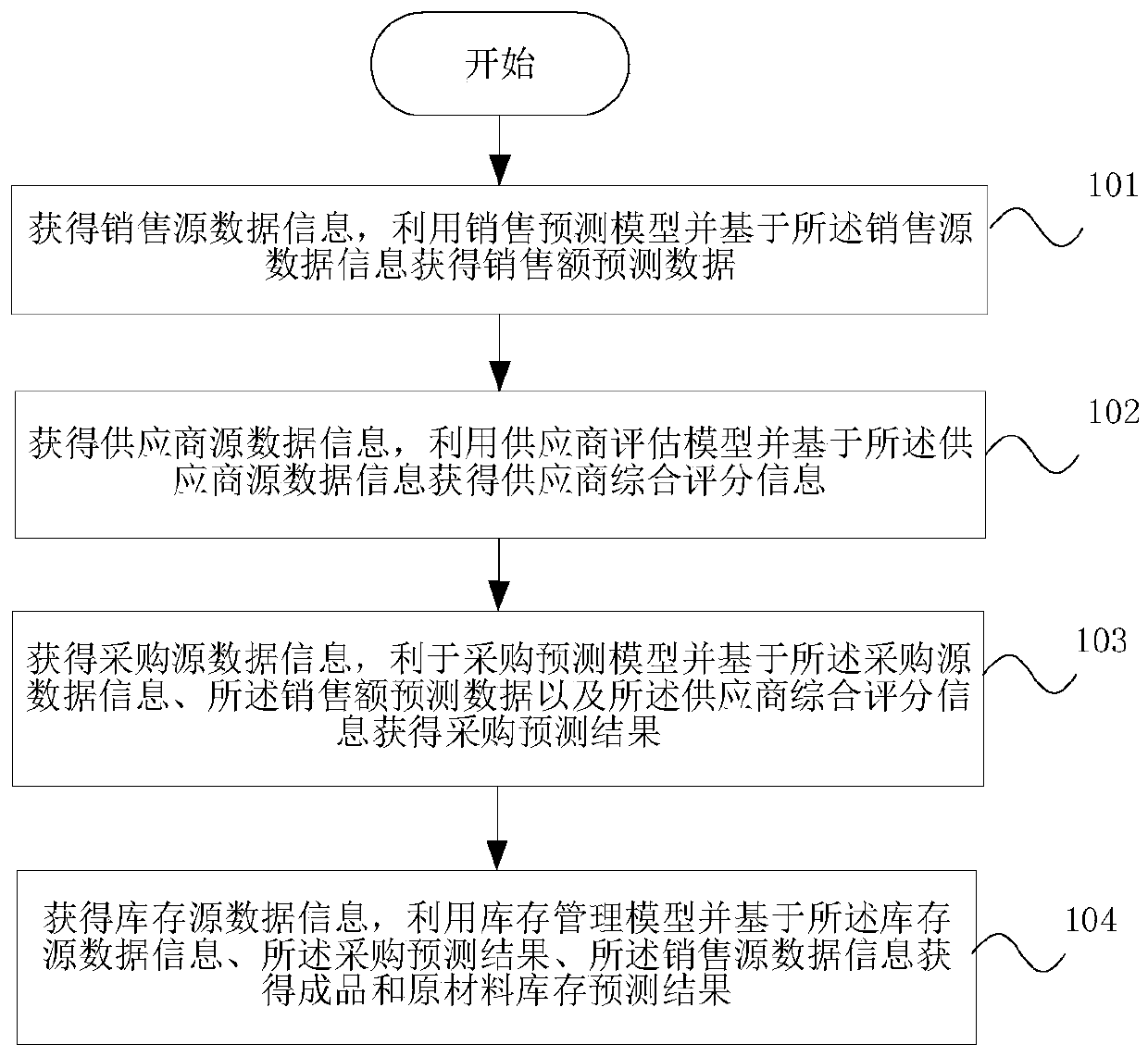
The invention discloses a supply chain control method and system and a storage medium. The method comprises: using a sales prediction model, and sales prediction data is obtained based on sales sourcedata information; utilizing the supplier evaluation model and obtaining supplier comprehensive scoring information based on the supplier source data information; A purchase prediction model is facilitated, and a purchase prediction result is obtained based on the purchase source data information, the sales prediction data and the supplier comprehensive scoring information; obtaining a finished product and raw material inventory prediction result by utilizing the inventory management model and based on the inventory source data information, the purchase prediction result and the sales source data information; according to the supply chain control method and system and the storage medium, the overall operation efficiency of the supply chain can be improved, and direct guidance can be provided for finished product and raw material inventory control; after an abnormal event occurs, the probability of occurrence of abnormality in other links can be predicted, early warning is given in time, the operation cost of the supply chain is reduced, and the operation efficiency of an enterprise is comprehensively improved.
Owner:北京航天智造科技发展有限公司
Test method for detecting reliability of product
ActiveCN101900788AImprove sturdinessImprove reliabilityElectrical testingDependabilityProduct design
Owner:BEIJING SHENGTAOPING TEST ENG TECH RES INST
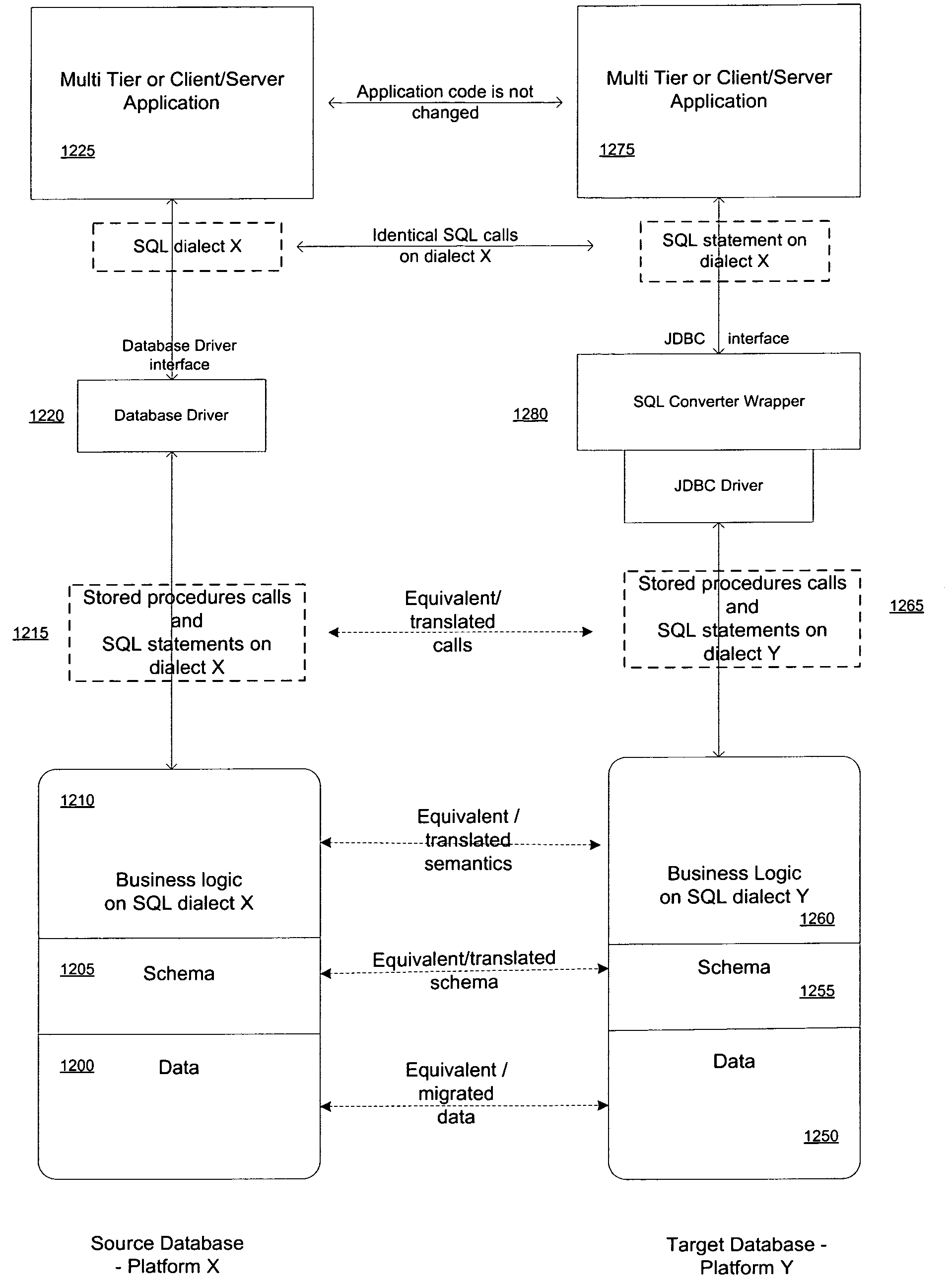
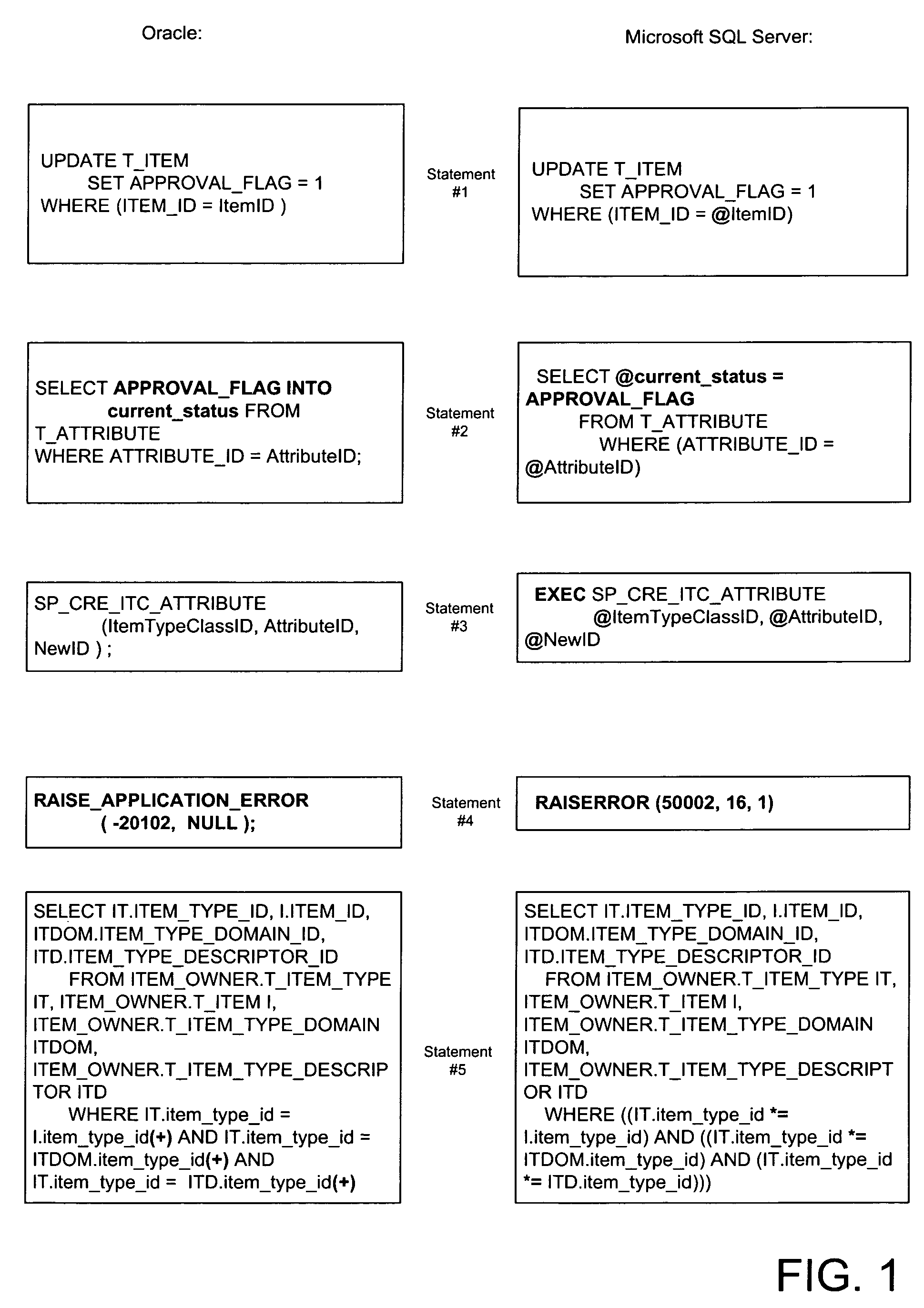
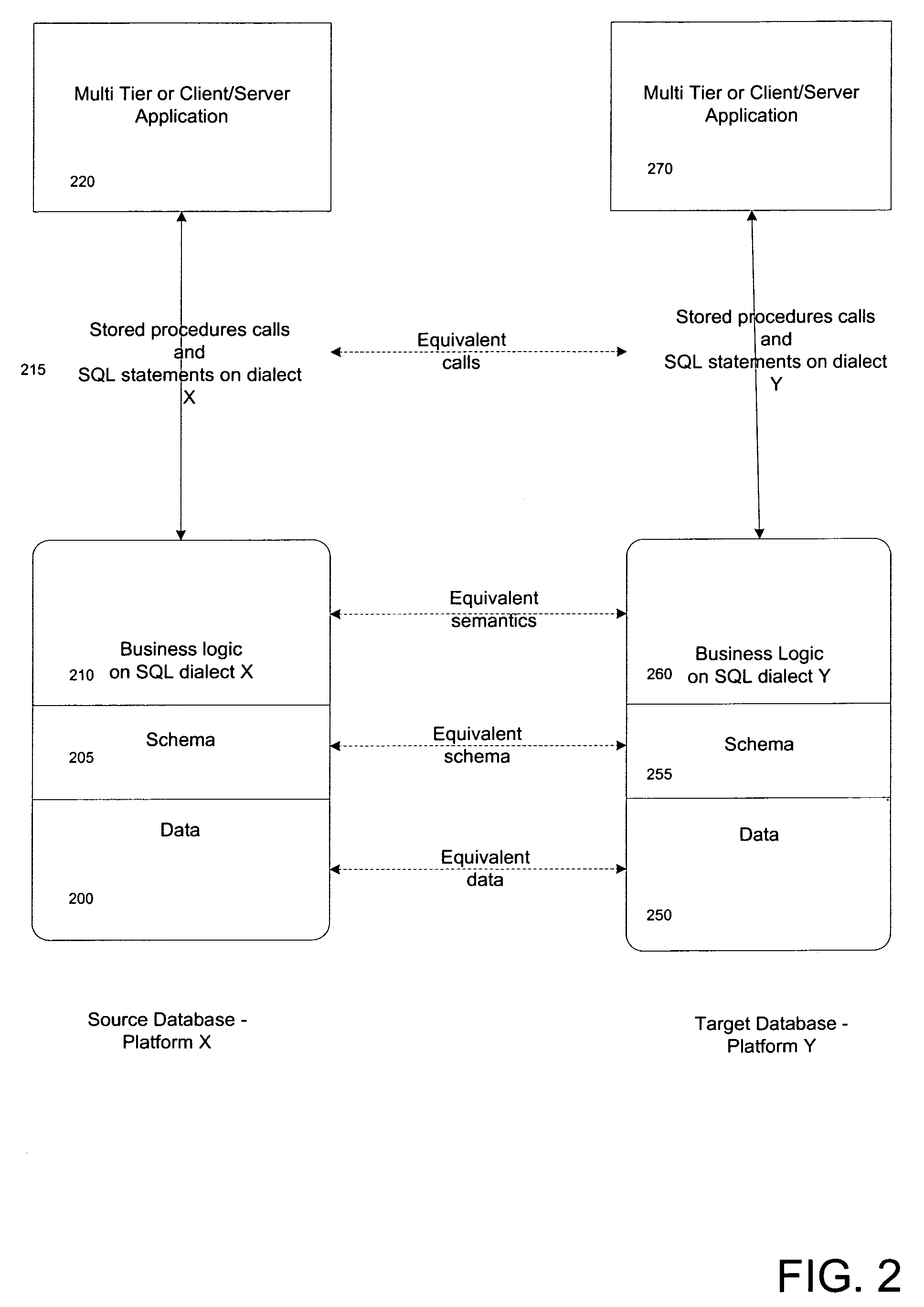
Apparatus and method for database migration
ActiveUS7664795B2Shorten the time to marketDigital data information retrievalDigital data processing detailsRelational databaseSql server
Systems and methods consistent with concepts of the present embodiment of the inventive concept provide mechanisms for database migration from one database (source database platform) platform to another (target database platform). The target and source databases can be any relational databases including, without limitation, Oracle, SQL Server (Microsoft), DB2 UDB (IBM), Sybase, MySQL, PostgreSQL, and the like. The inventive methodology will work well for migration of any relational database that implements SQL language from one platform to another. A feature of the invention is the provision of a virtual engine enabling the running of an original application on a migrated database.
Owner:MICROSOFT TECH LICENSING LLC
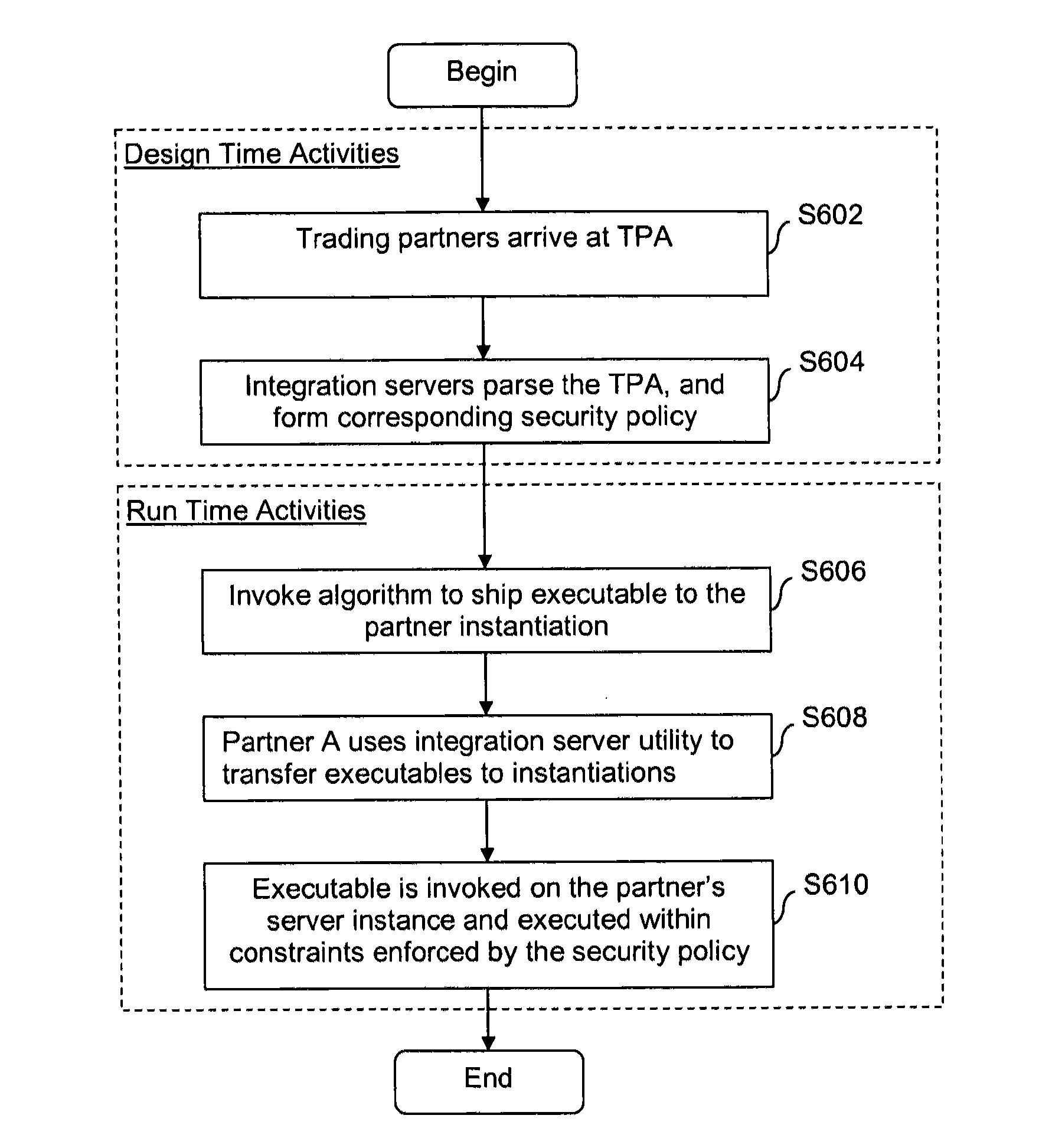
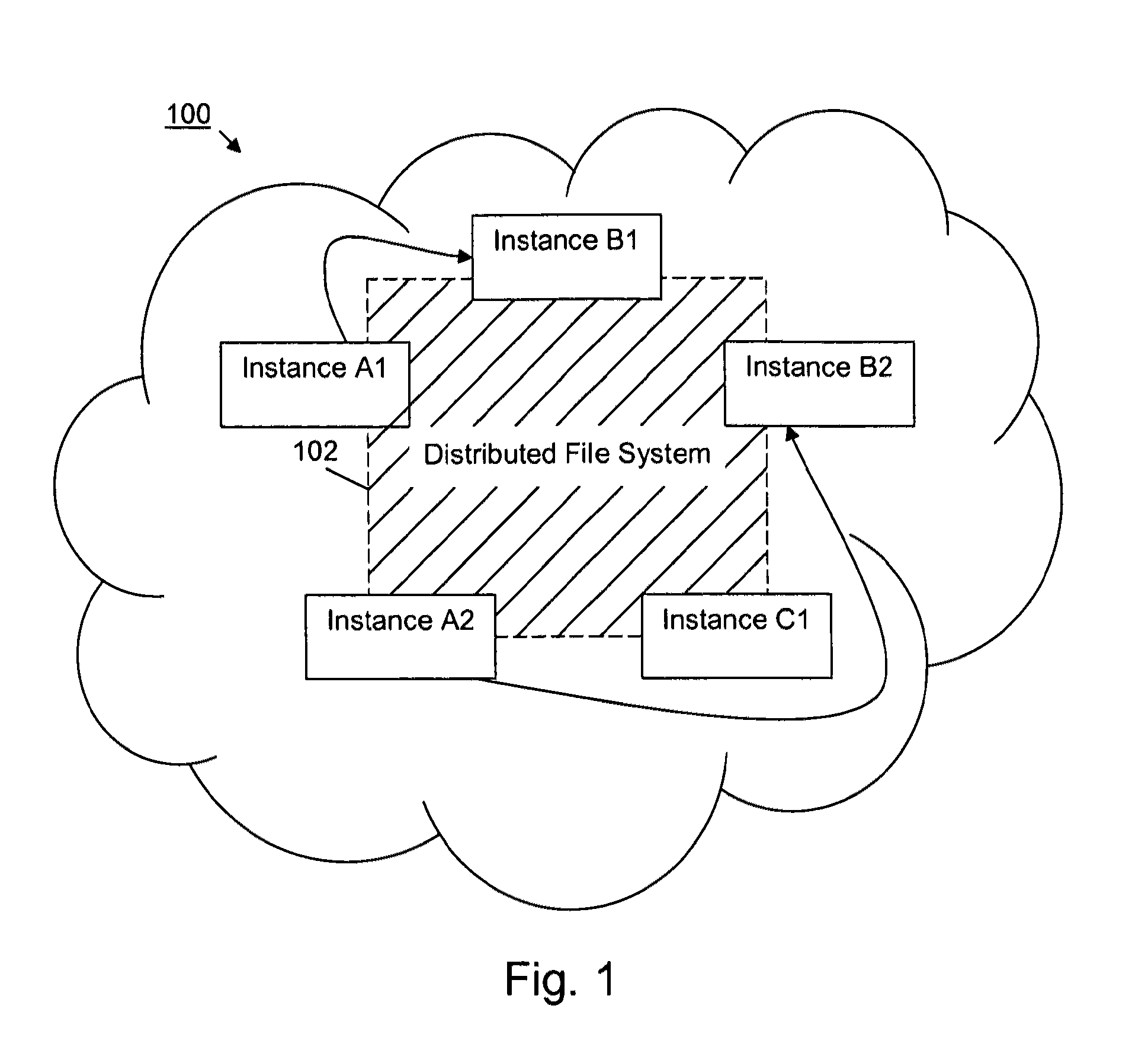
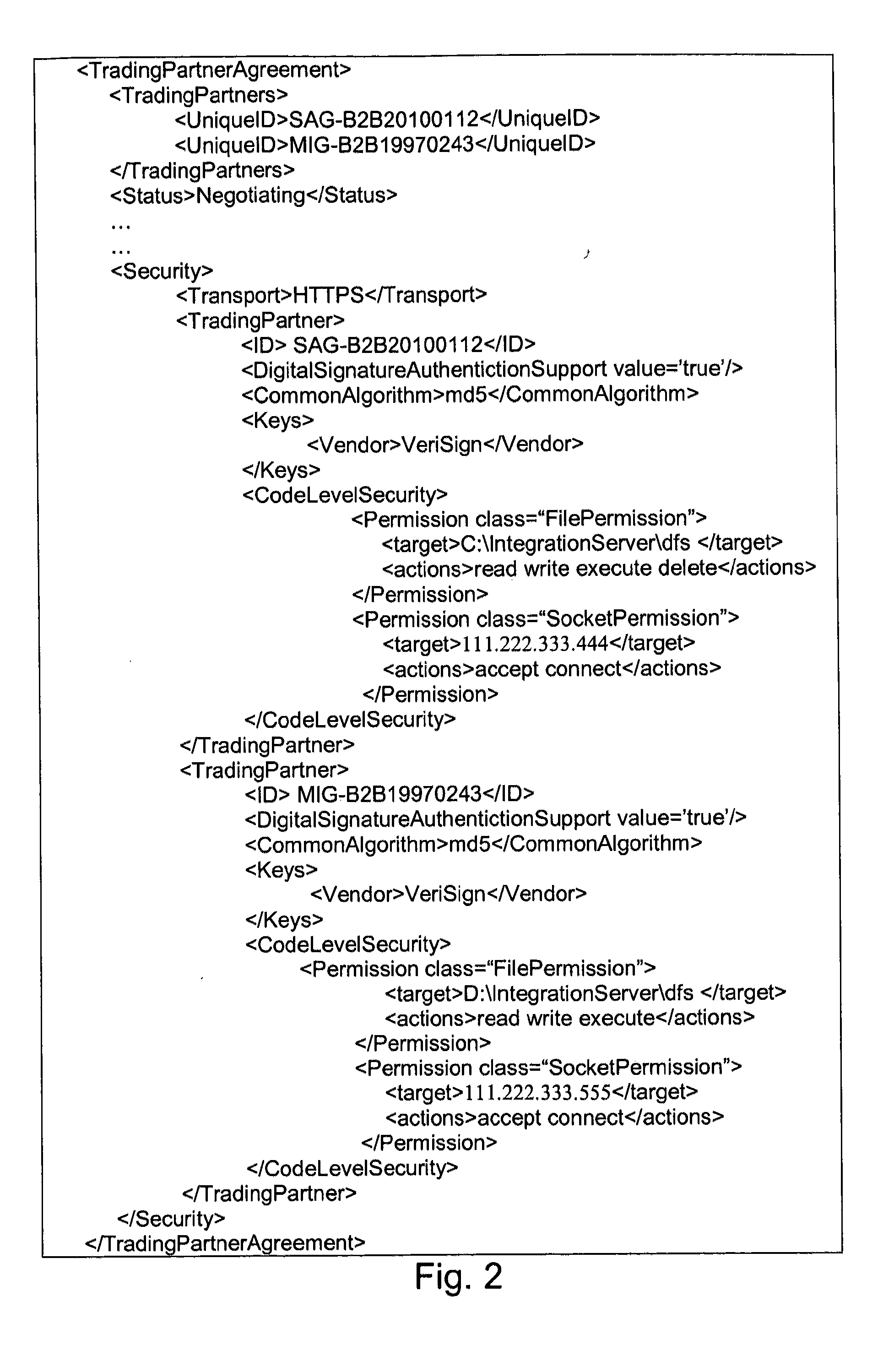
Security systems and/or methods for cloud computing environments
ActiveUS20120116782A1Improve application performanceQuickly deliver business resultComputer security arrangementsTransmissionSecurity policyShared resource
Certain example embodiments described herein relate to security systems and / or methods for cloud computing environments. More particularly, certain example embodiments described herein relate to the negotiation and subsequent use of Trading Partner Agreements (TPAs) between partners in a Virtual Organization, the TPAs enabling resources to be shared between the partners in a secure manner. In certain example embodiments, TPAs are negotiated, an algorithm is executed to determine where an executable is to be run, the resource is transferred to the location where it is to be run, and it is executed—with the TPAs collectively defining a security policy that constrains how and where it can be executed, the resources it can use, etc. The executable may be transferred to a location in a multipart (e.g., SMIME) message, along with header information and rights associated with the executable.
Owner:SOFTWARE AG
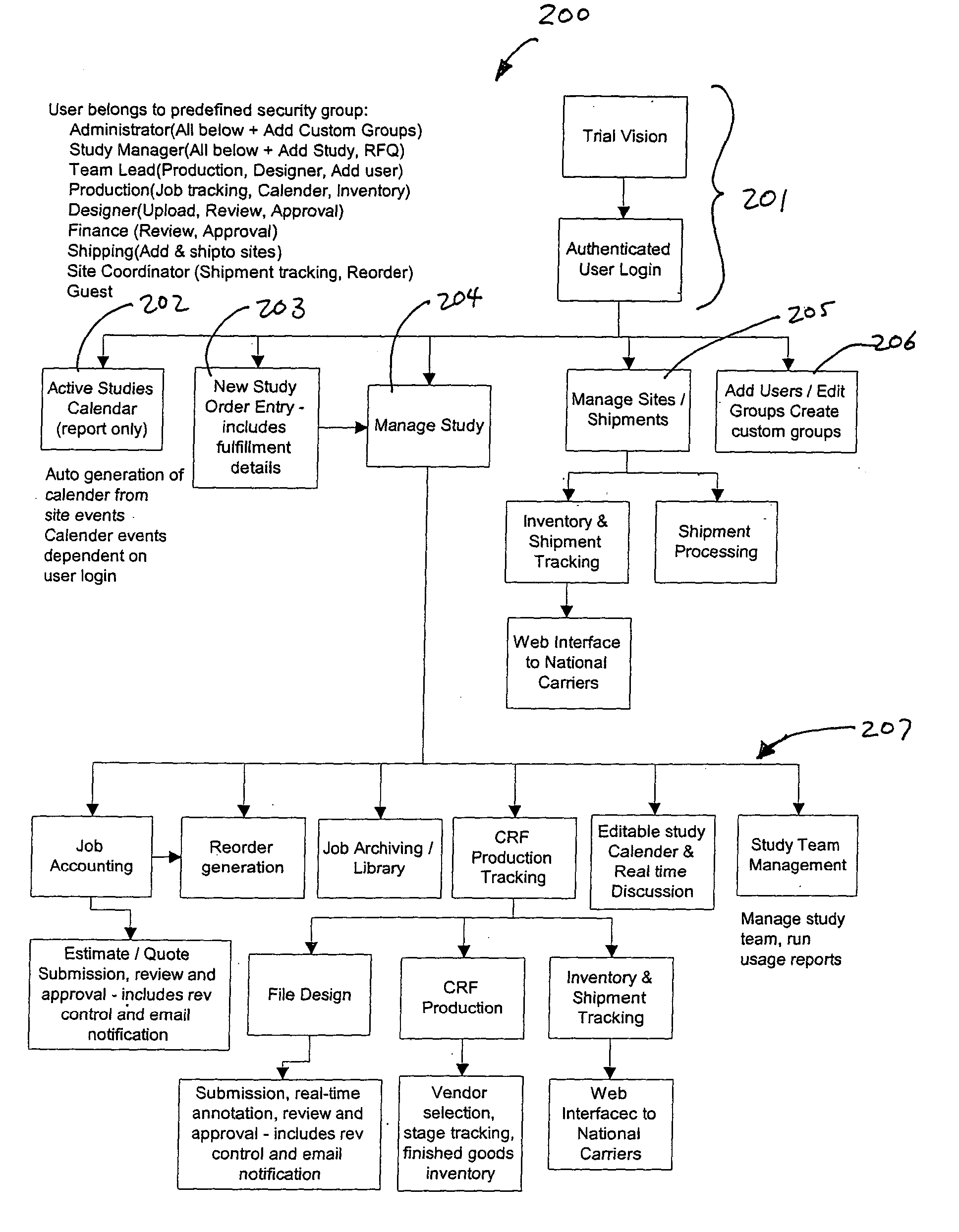
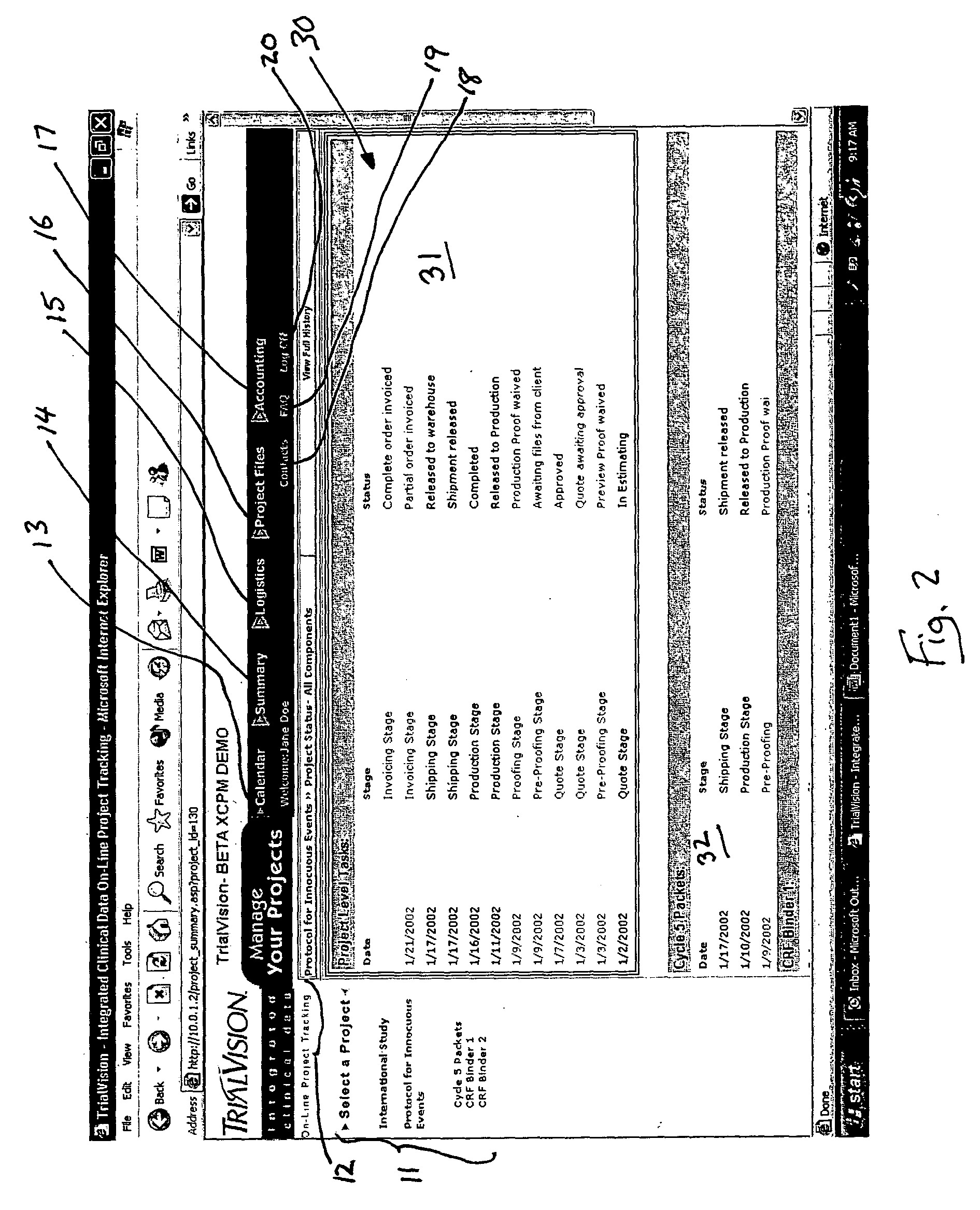
Apparatus, system and method for clinical documentation and data management
InactiveUS20050055241A1Facilitate coordinationEasy to trackOffice automationMedical equipmentFacilitated communicationDocumentation procedure
A system, method and apparatus for management of clinical studies to facilitate the tracking of the studies by personnel involved in the study, such as managers, coordinators, and vendors, wherever they are located, as well as placing orders, and receiving financial information, the invention being suitable for managing clinical trial documentation in connection with the pharmaceutical industry, and for facilitating communications and delivery of clinical data and documentation between clients who initiate a clinical study and a vendor who may, for example, provide data capture, data packaging, and submitting the study documentation to the appropriate authority, such as, for example, the FDA or WHO.
Owner:HORSTMANN STEPHEN P
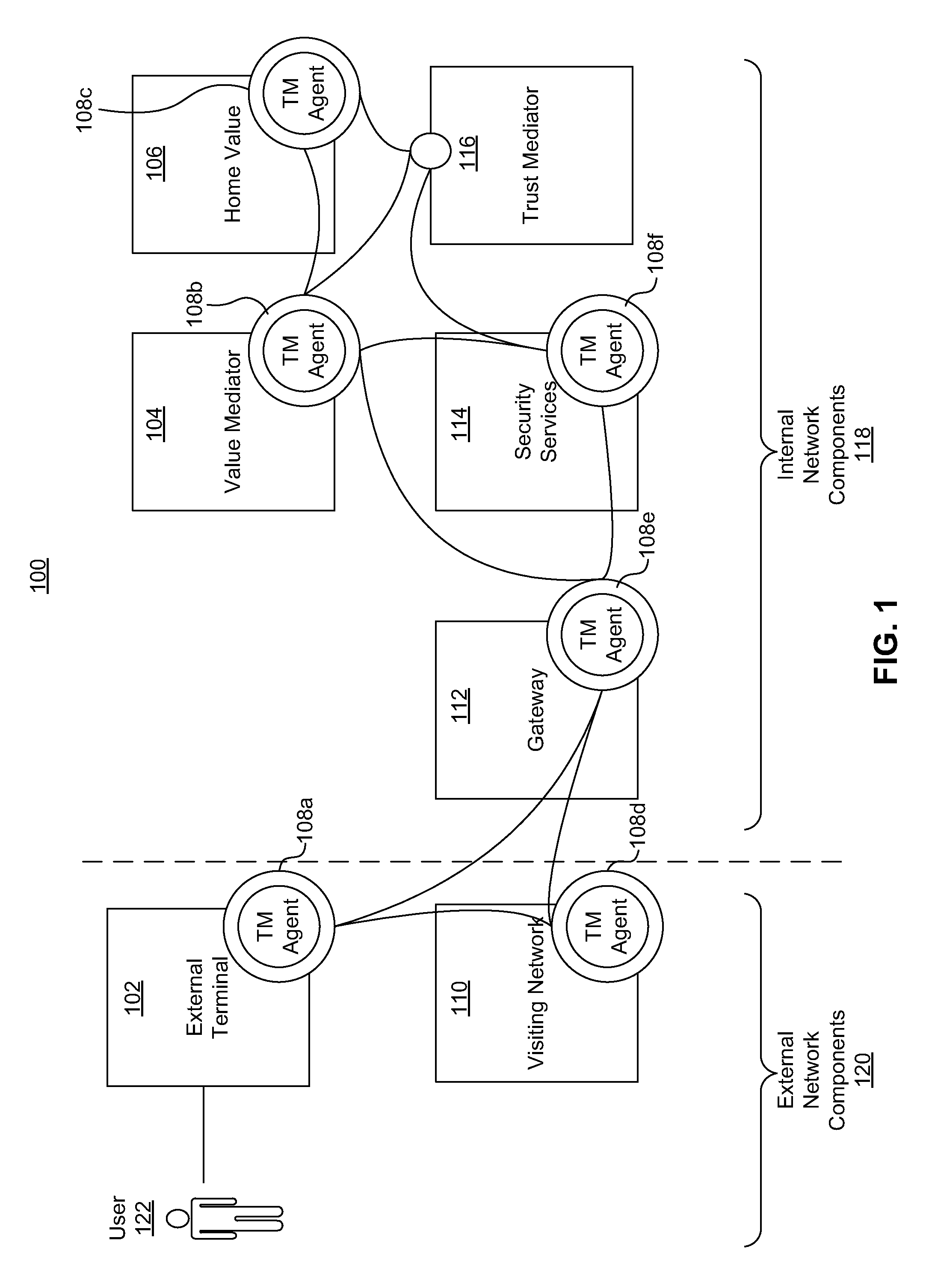
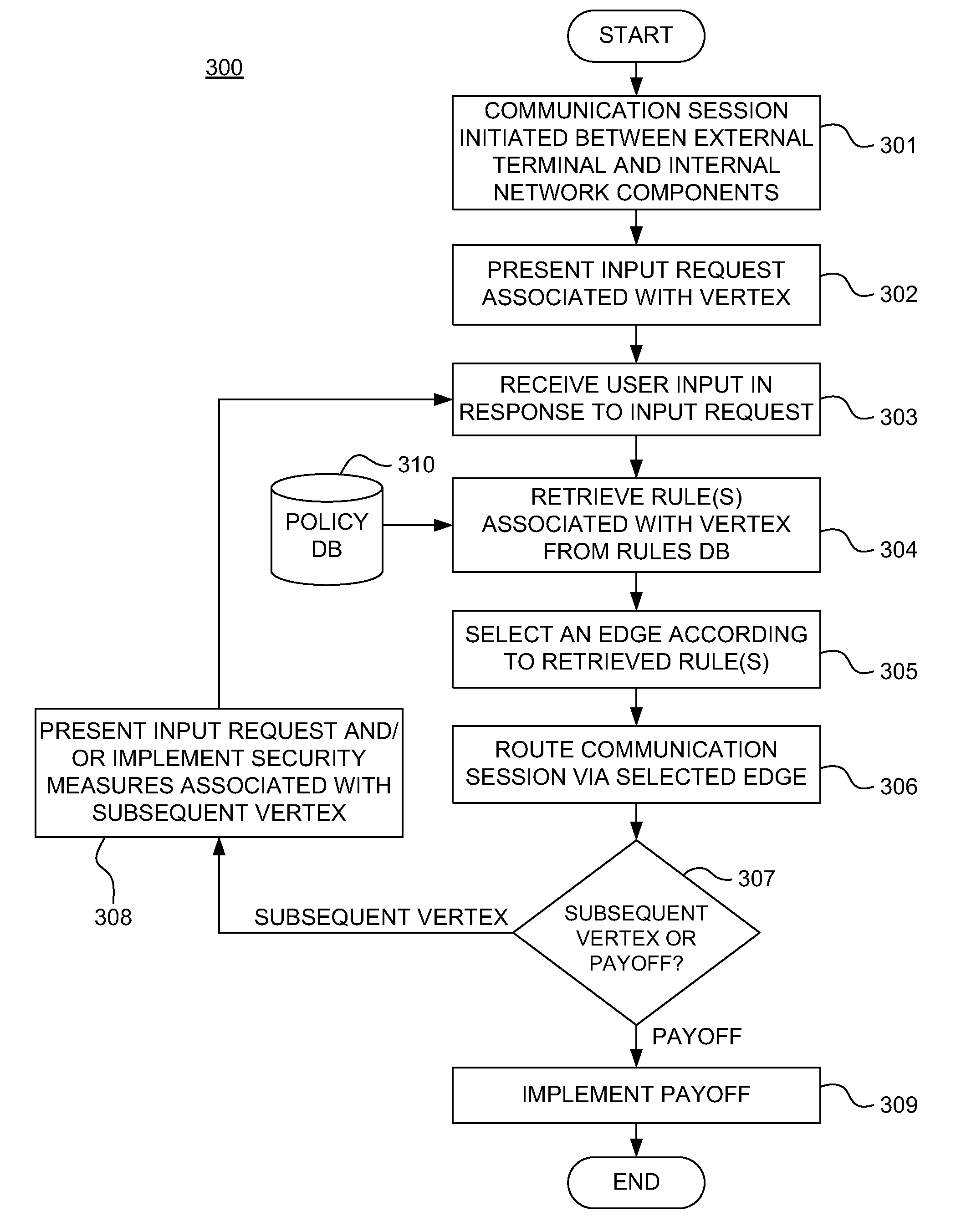
Dynamically adaptive policy management for securing mobile financial transactions
ActiveUS20110313930A1Shorten the time to marketIncrease propensityFinanceDigital data processing detailsAdaptive securityFinancial trading
A system for securing a mobile financial transaction using an adaptive security policy is provided by presenting, via an external terminal, an input request associated with a vertex of the security policy. User input is received via the external terminal in response to the input request. A rule associated with the vertex is retrieved from a database. An edge is selected from a plurality of edges associated with the vertex according to the retrieved rule. A communication session of the external terminal is routed to a subsequent vertex via the selected edge.
Owner:AMERICAN EXPRESS TRAVEL RELATED SERVICES CO INC
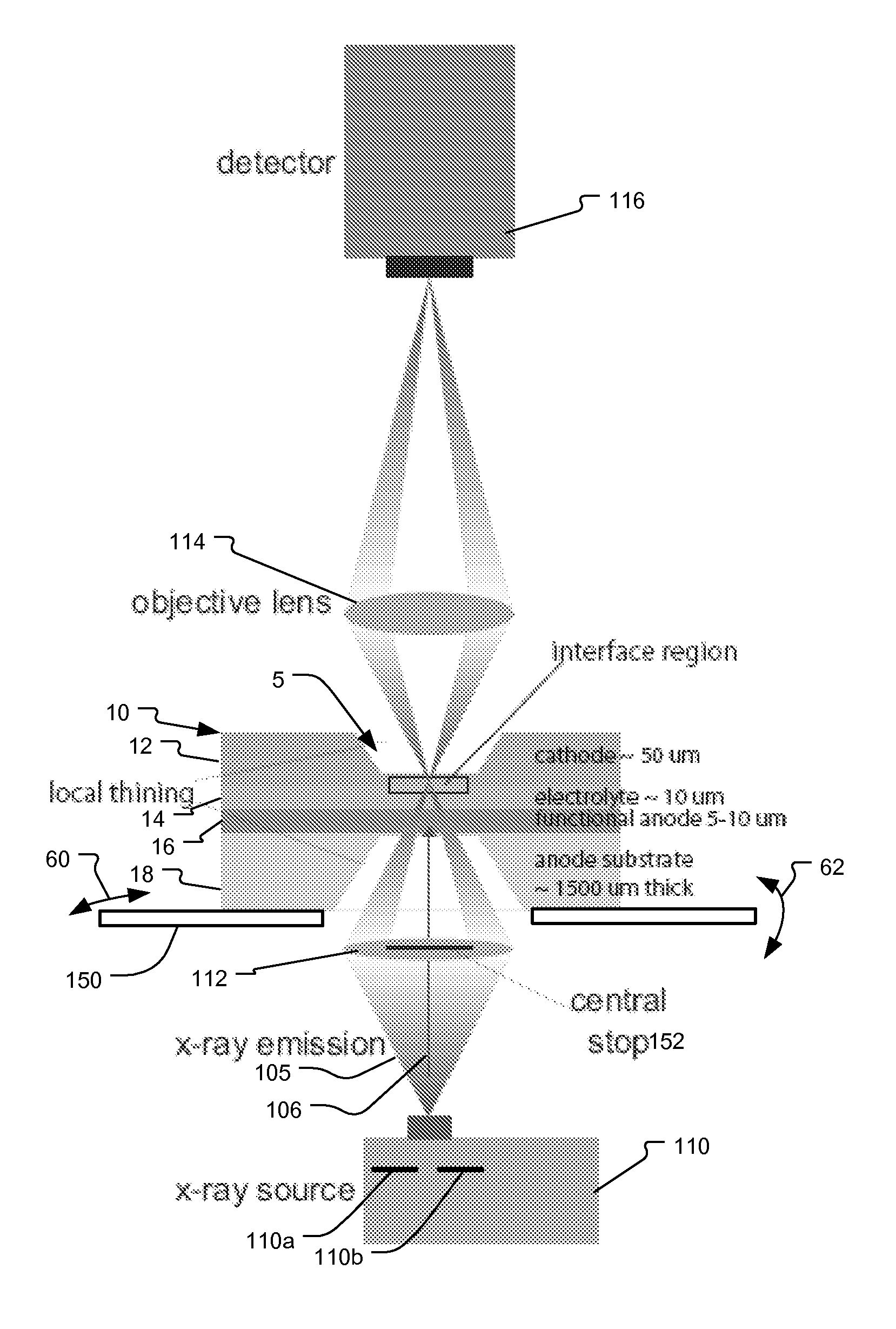
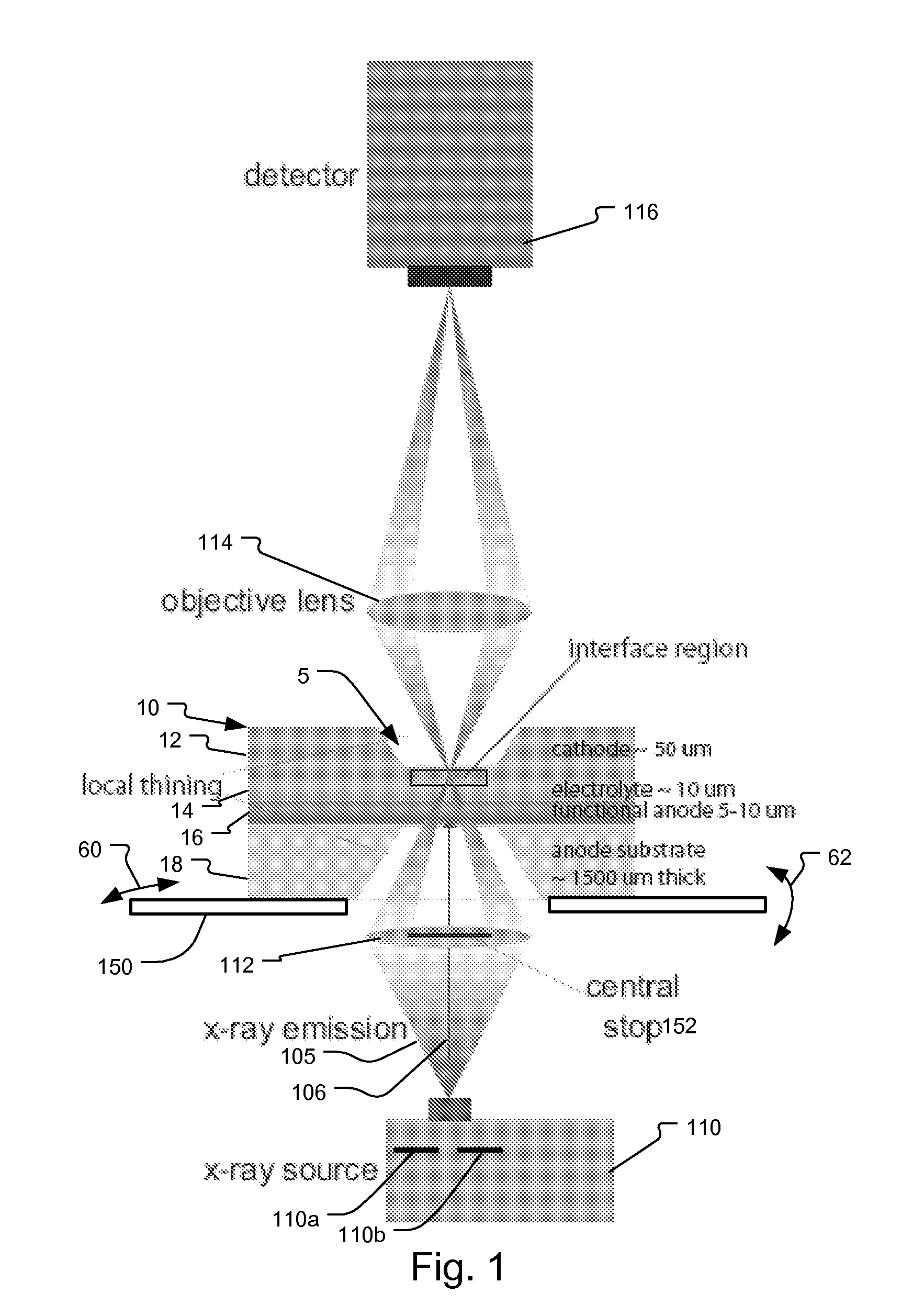
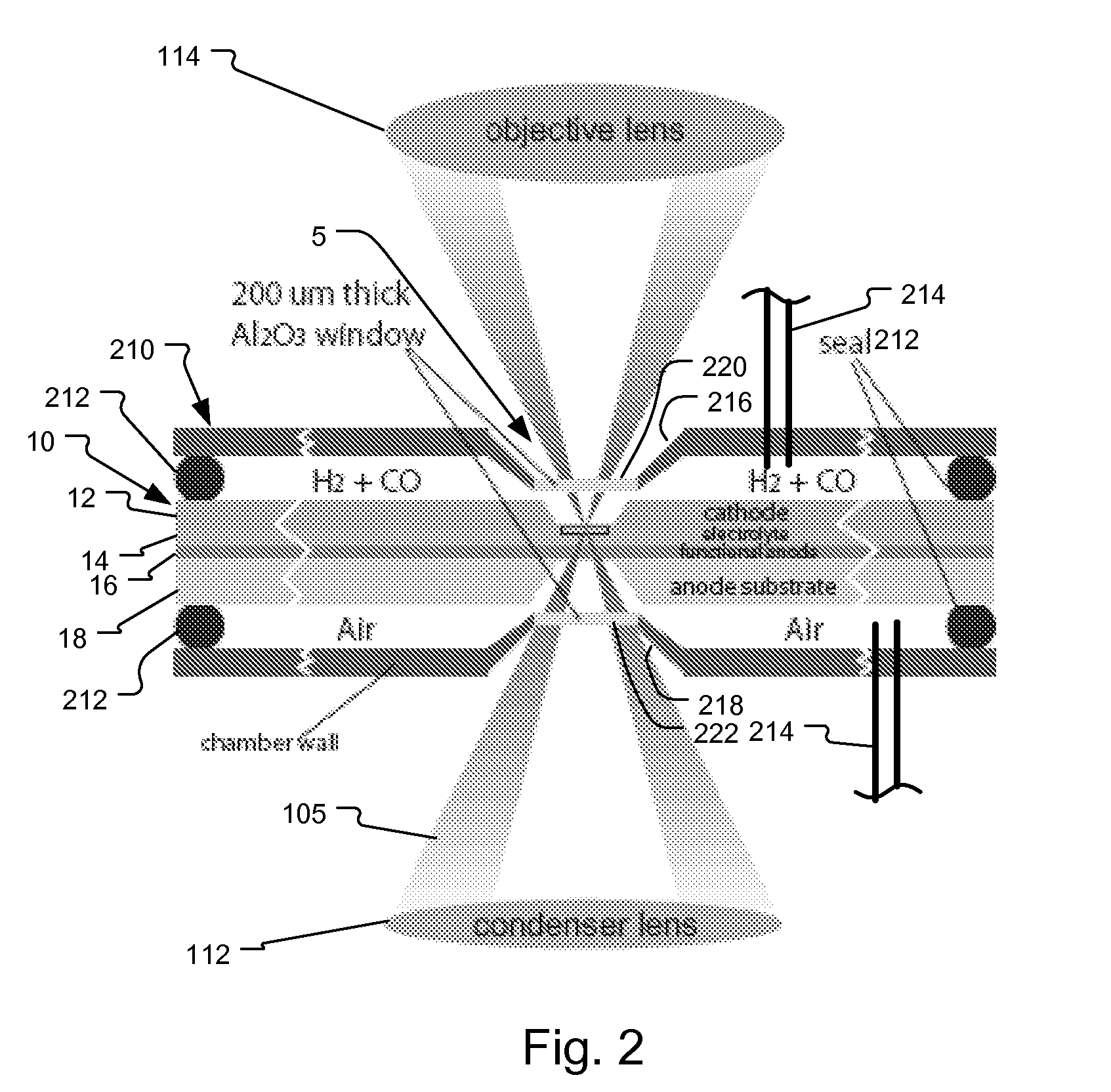
System and method for fuel cell material x-ray analysis
ActiveUS7499521B2Shorten development timeImprove reliabilityRadiation/particle handlingUsing wave/particle radiation meansHard X-raysMetrology
An imaging technology for fuel cells is based on x-ray microscopy. A metrology system images the electro-chemical interaction areas of solid-oxide fuel cells (SOFC) in-situ. This system takes advantage of both the penetrating power and elemental absorption contrast of hard x-ray radiation to image the internal interaction areas in a SOFC. The technology can further take advantage of the strong dependence of the x-ray absorption on material type and energy to distinguish the four major material types: cathode, electrolyte, air, and low-Z contaminants such as sulfur.
Owner:CARL ZEISS X RAY MICROSCOPY
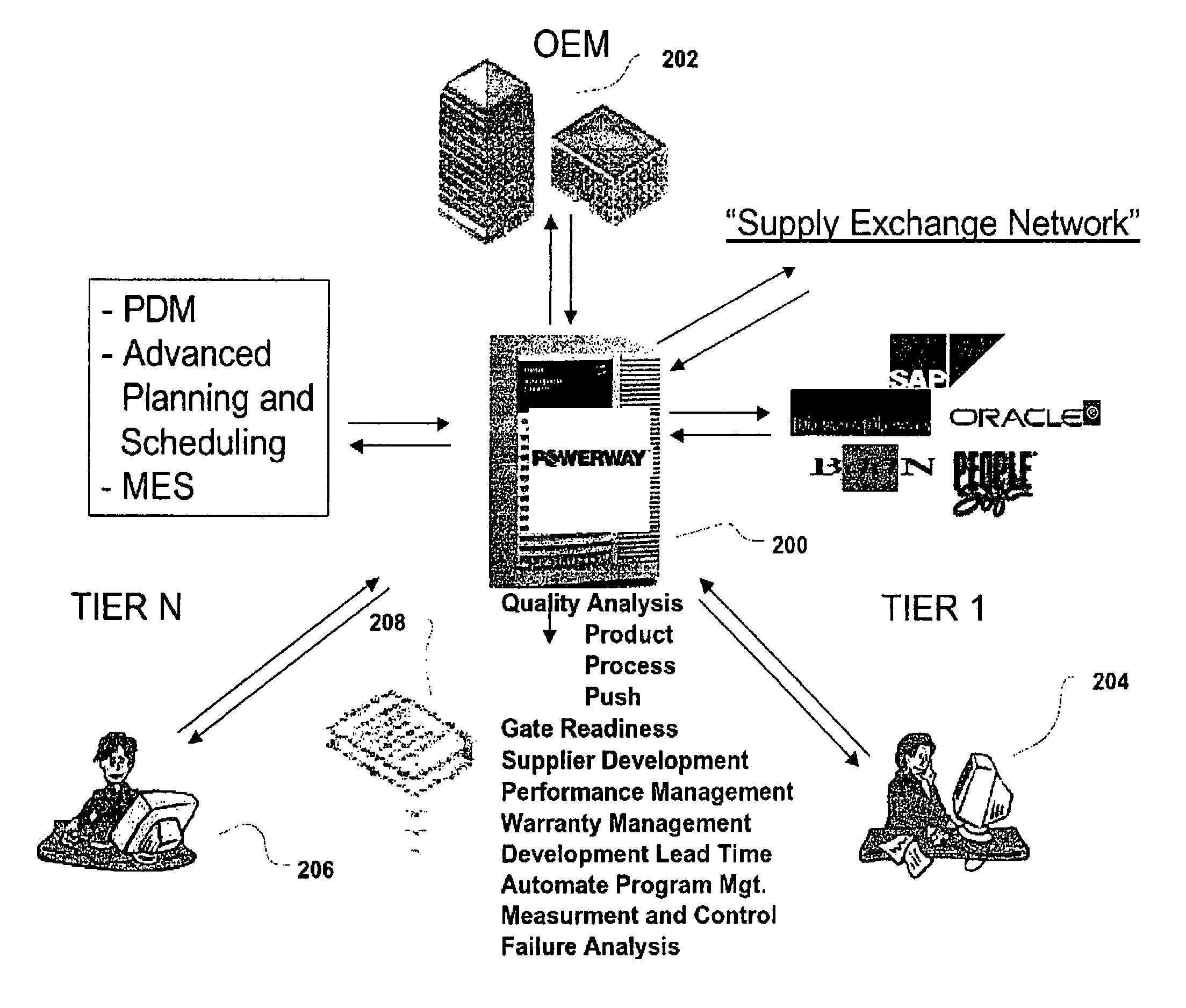
Method and system for supply chain product and process development collaboration
InactiveUS7747572B2Appropriately produceLow costDigital data processing detailsResourcesCollaboration toolProcess development
A method and system for supply chain product and process development collaboration. The supply chain is comprised of at least one project, each project having a part (or family of parts), a supplier of the part, and a customer. The system may include a data storage and retrieval device operably connected to a processor. The system's functions include collaboration tools and security based on the users' project roles. The method uses a methodology that ensures certain requirements are met prior to satisfying the methodology. The system and method of the present invention ensures that current, applicable methodologies are applied to a specific part and allows for collaboration between the customer and supplier during all phases of the project. Because all projects in a supply chain are present, a user can navigate throughout the supply chain. The supply chain's reporting functions permit determination of potential performance risks and the source thereof.
Owner:WAYPOINT GLOBAL II
Dynamic pairing system for securing a trusted communication channel
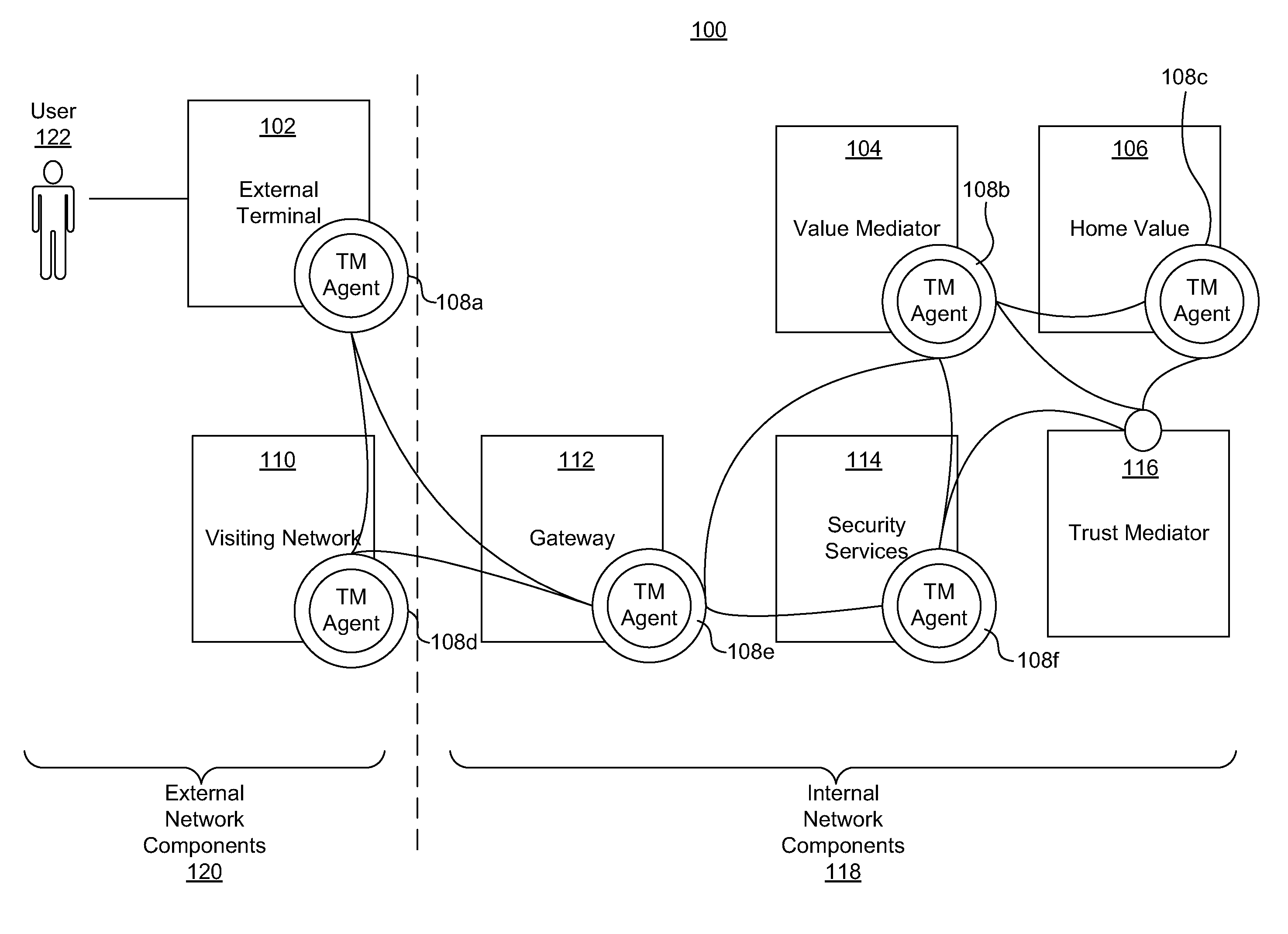
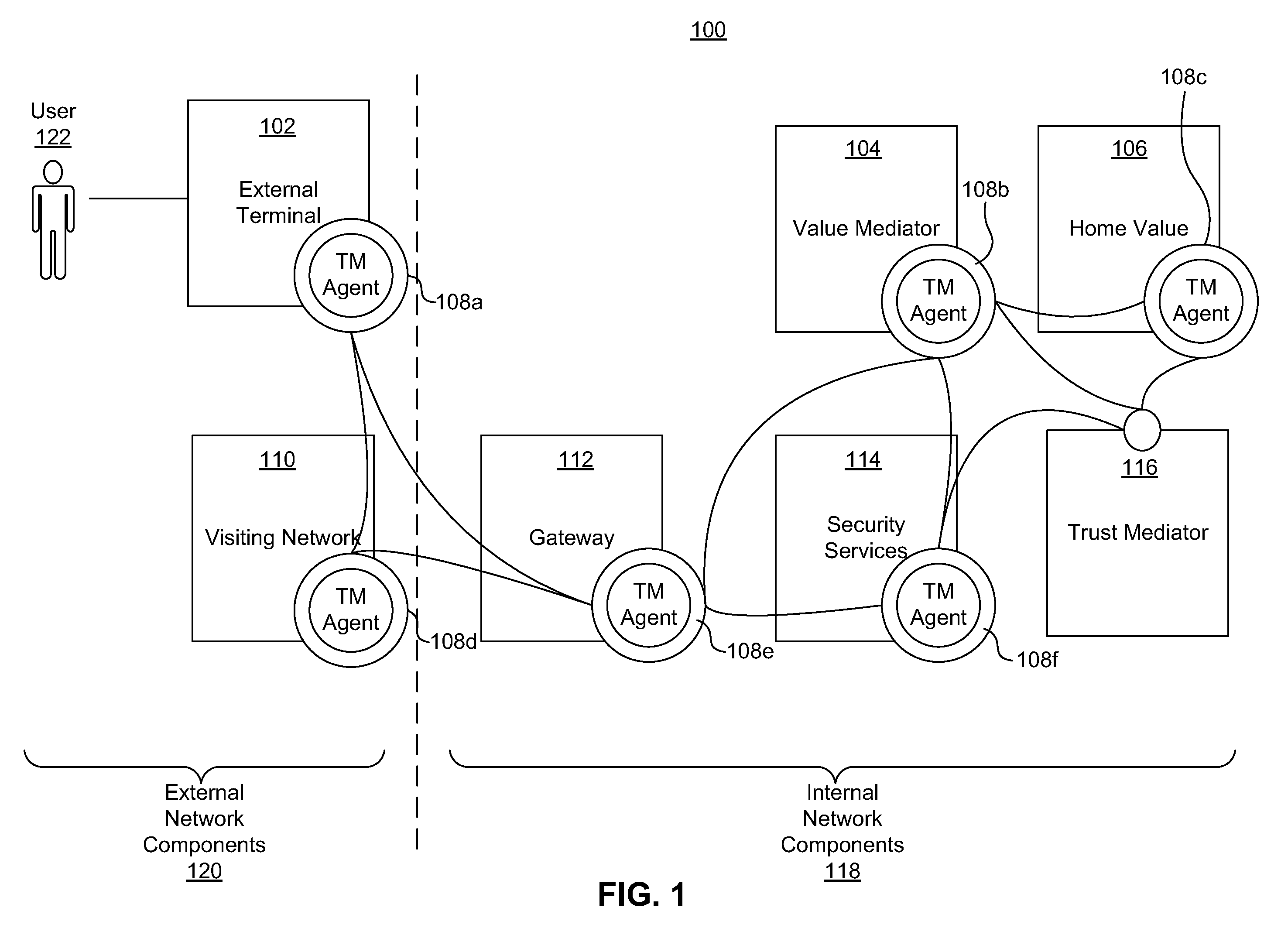
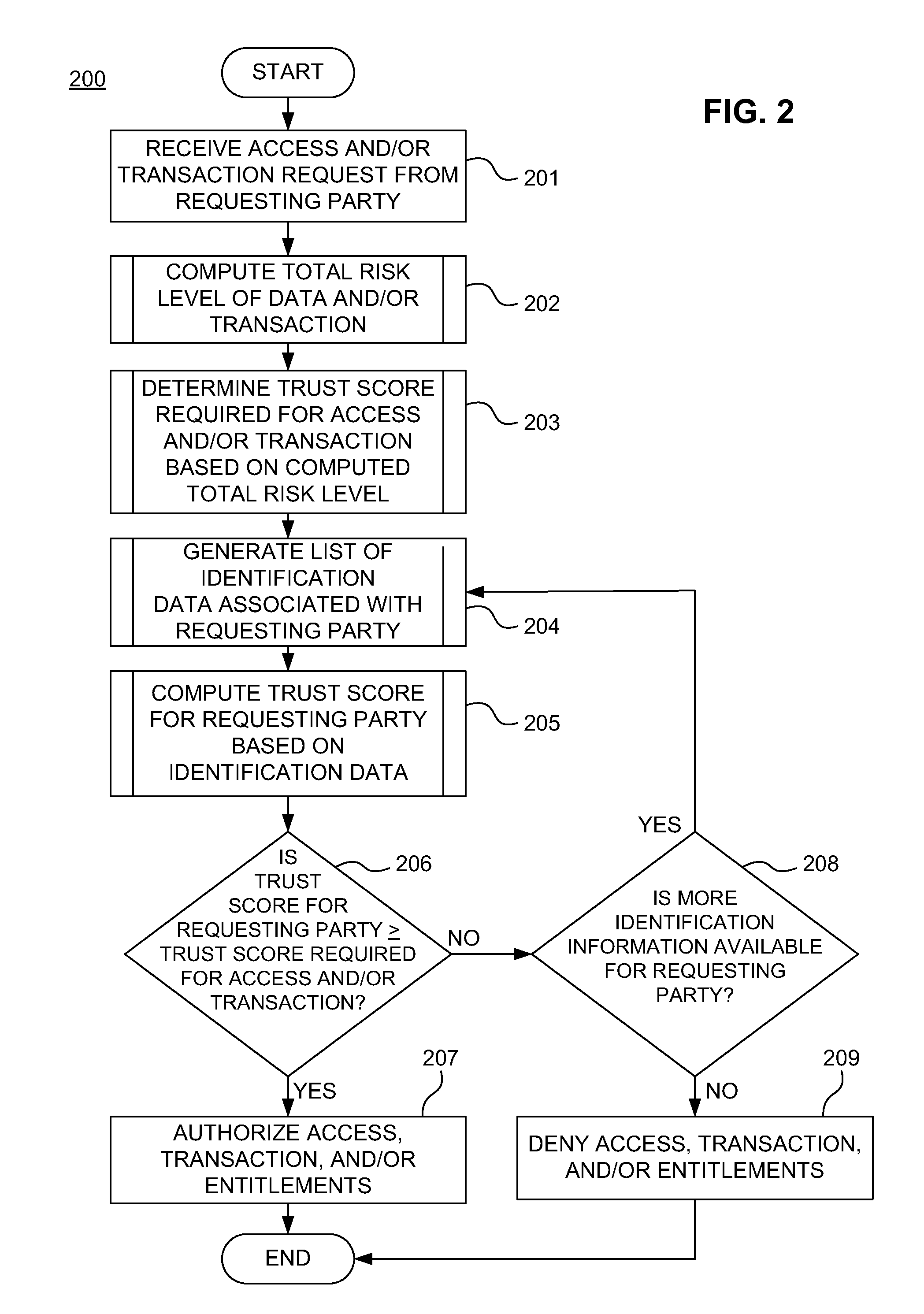
ActiveUS20110313925A1Shorten the time to marketIncrease propensityFinancePayment architectureTotal riskInternet privacy
A system for securing a trusted communications channel for a mobile financial transaction is provided by receiving, from a user via an external terminal, a request for an access control entitlement to complete a financial transaction. A total risk level associated with the financial transaction is computed. A required trust score is determined based on the total risk level. User identification data associated with the user is received from one or more data sources. The user identification data is validated. A user trust score associated with the user is computed based on the validated identification data. The user trust score is compared to the required trust score. The access control entitlement is transmitted to the user via the external terminal if the user trust score is greater than or equal to the required trust score.
Owner:AMERICAN EXPRESS TRAVEL RELATED SERVICES CO INC
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com