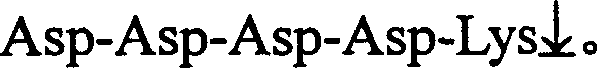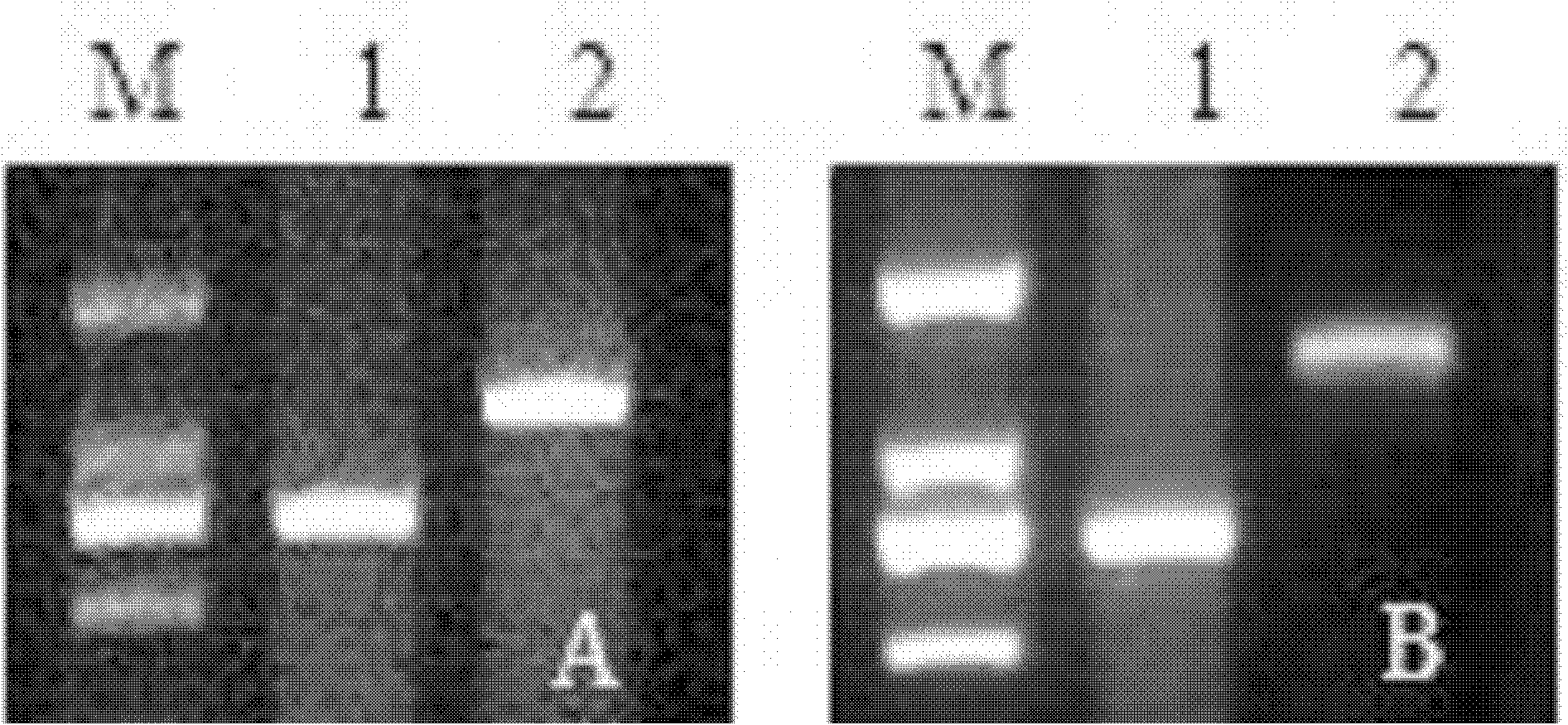Patents
Literature
52 results about "Light chain gene" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
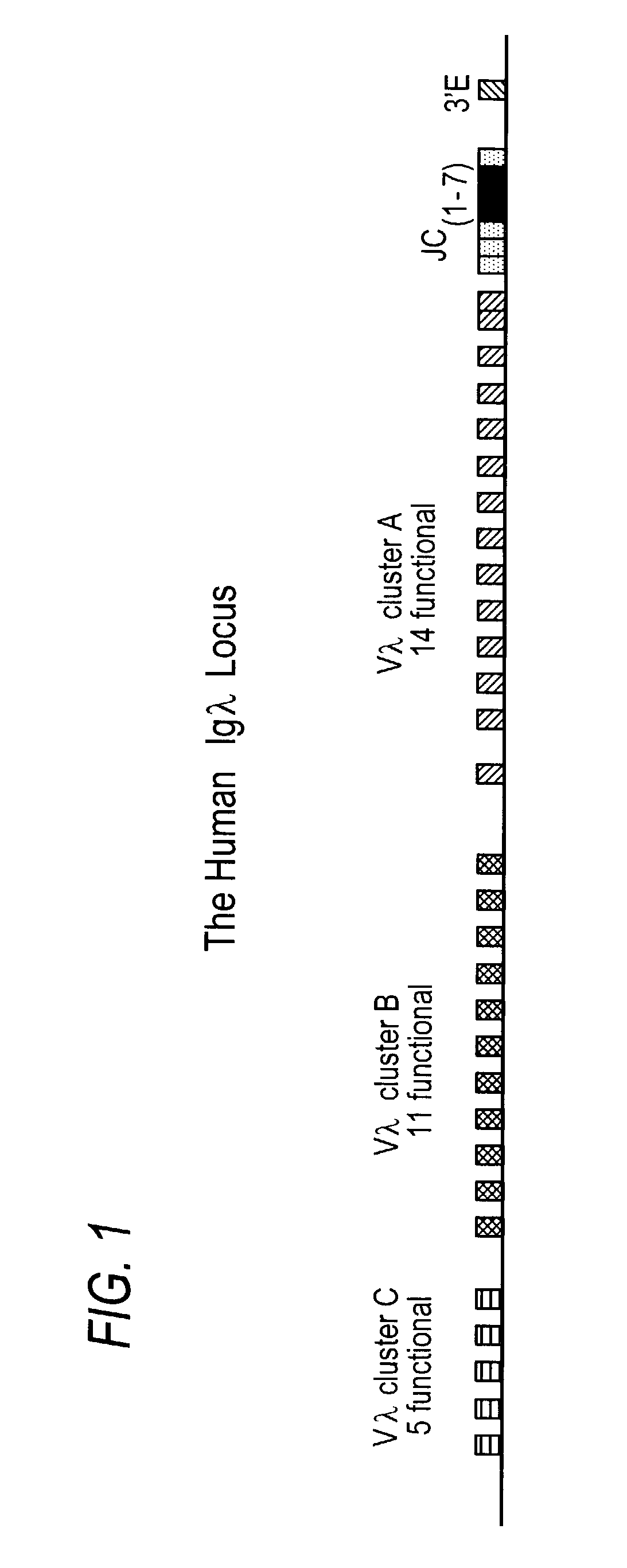
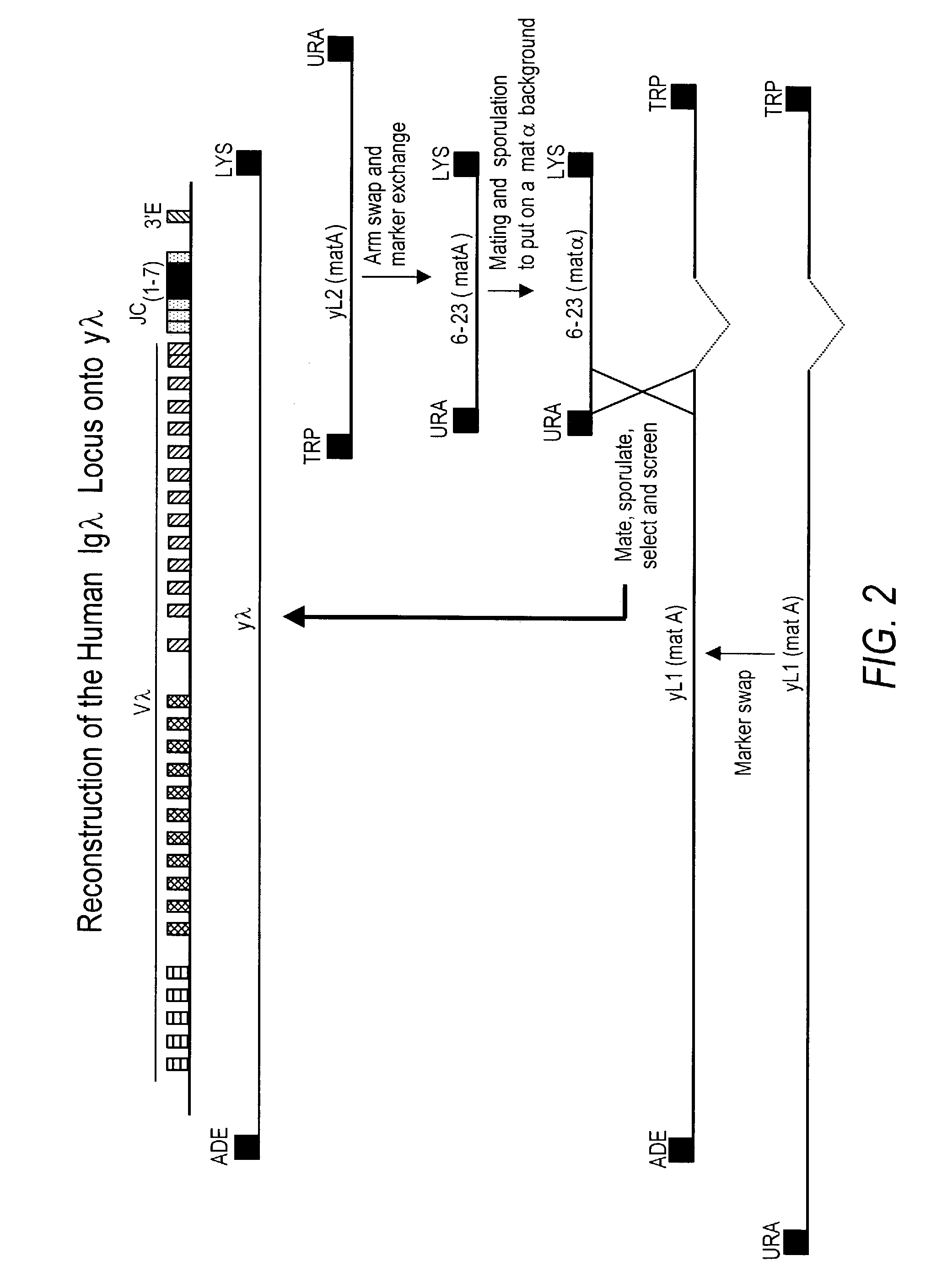
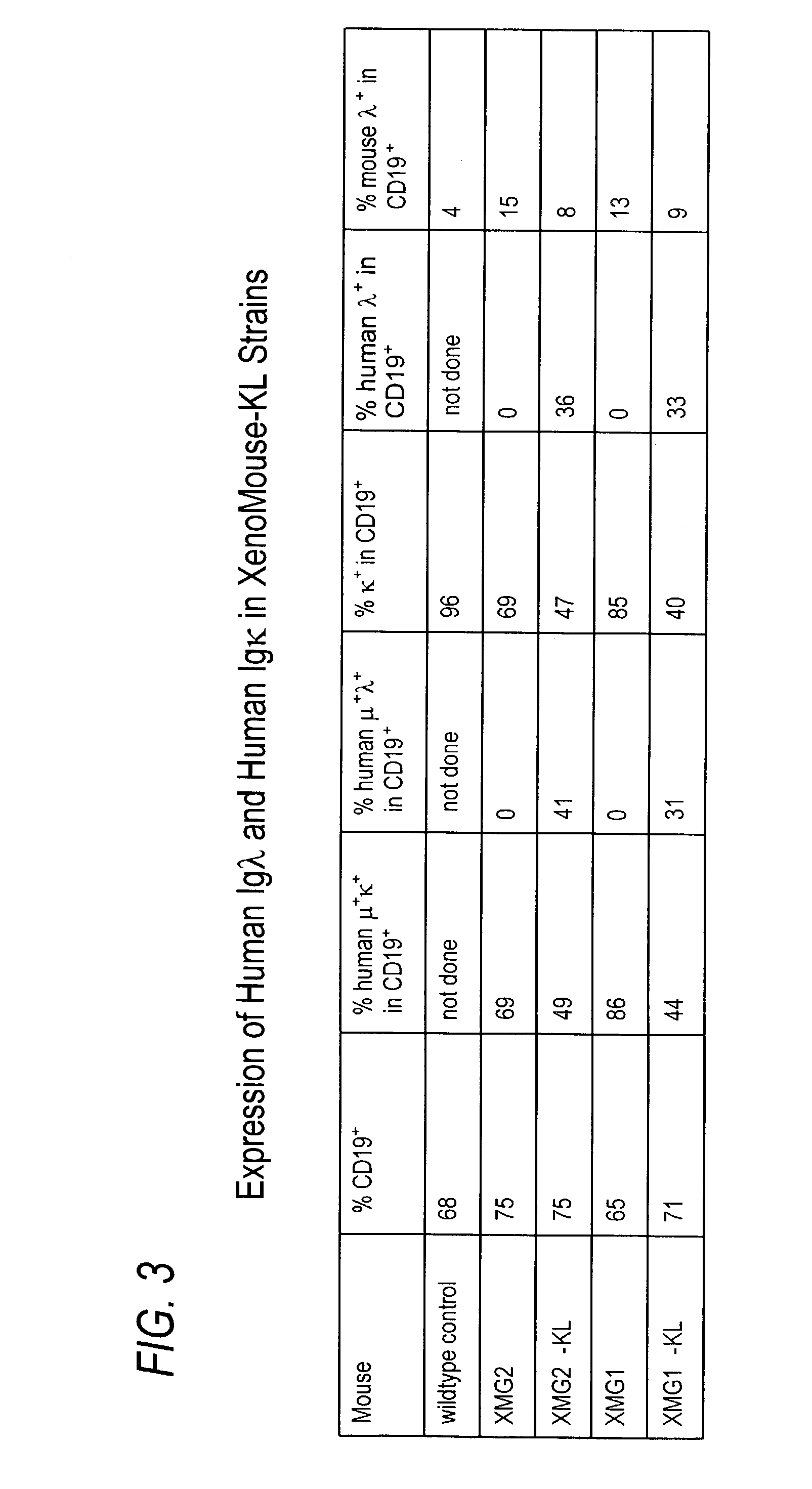
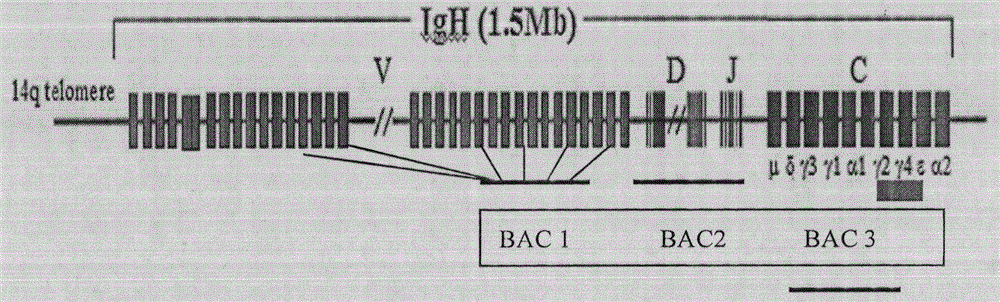
Transgenic animals bearing human Iglambda light chain genes
ActiveUS7435871B2Restore normal B-cell developmentAnimal cellsSerum immunoglobulinsPhysiologyLambda Light Chain Gene
Owner:AMGEN FREMONT INC
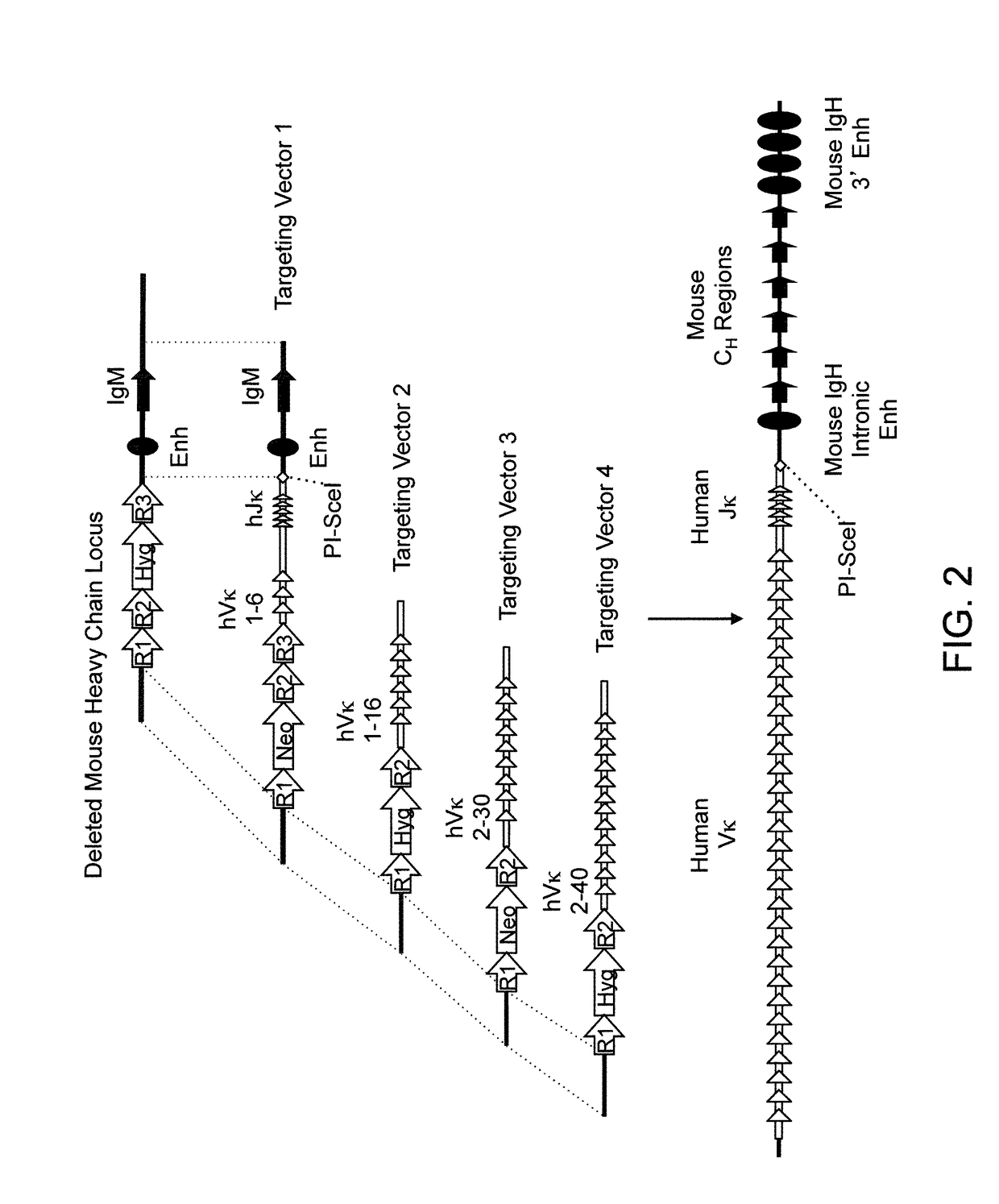
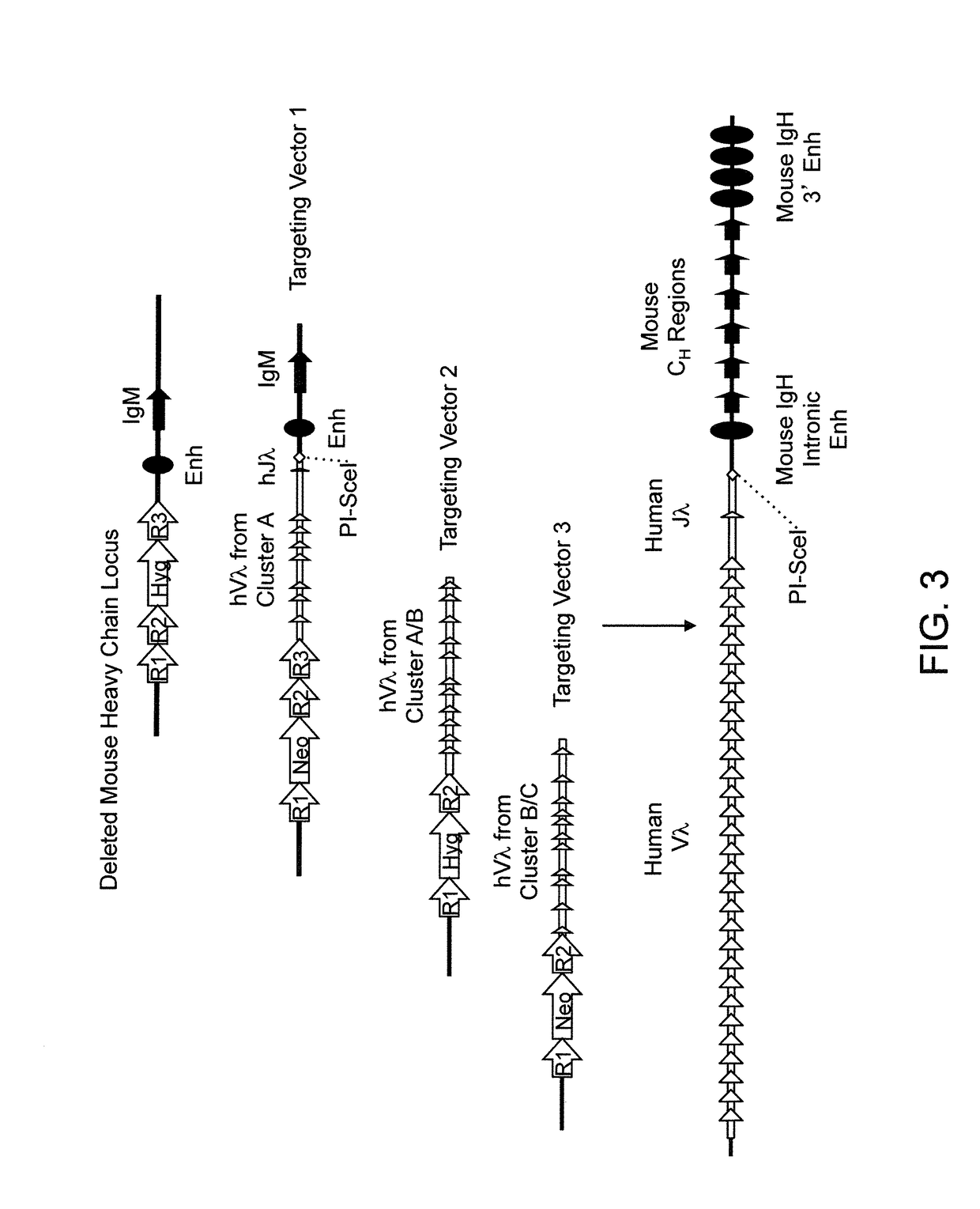
Humanized Rodents that Express Heavy Chain Containing VL Domains
InactiveUS20130212719A1Reduced fertilityImprove fertilityAnimal cellsHybrid immunoglobulinsGenetic MaterialsVariable domain
Non-human animals, tissues, cells, and genetic material are provided that comprise a modification of an endogenous non-human heavy chain immunoglobulin sequence and that comprise an ADAM6 activity functional in a rodent (e.g., a mouse), wherein the non-human animals rearrange human immunoglobulin light chain gene segments in the context of heavy chain constant regions and express immunoglobulin-like molecules comprising human immunoglobulin light chain variable domains fused to heavy chain constant domains that are cognate with human immunoglobulin light chain variable domains fused to light chain constant domains.
Owner:REGENERON PHARM INC
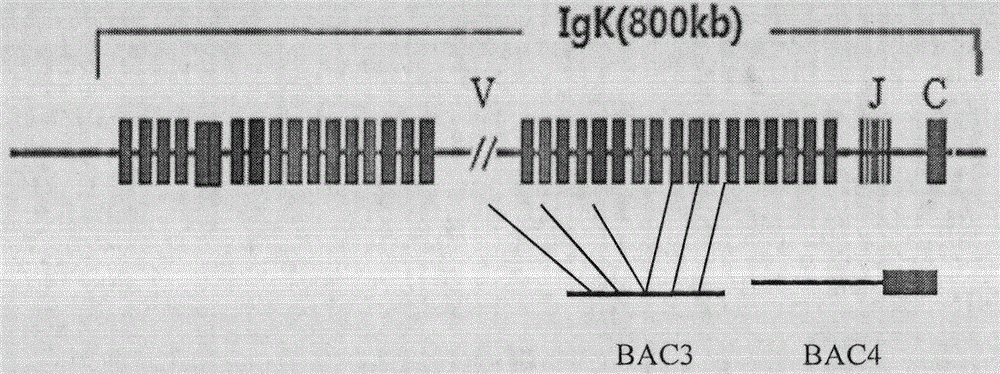
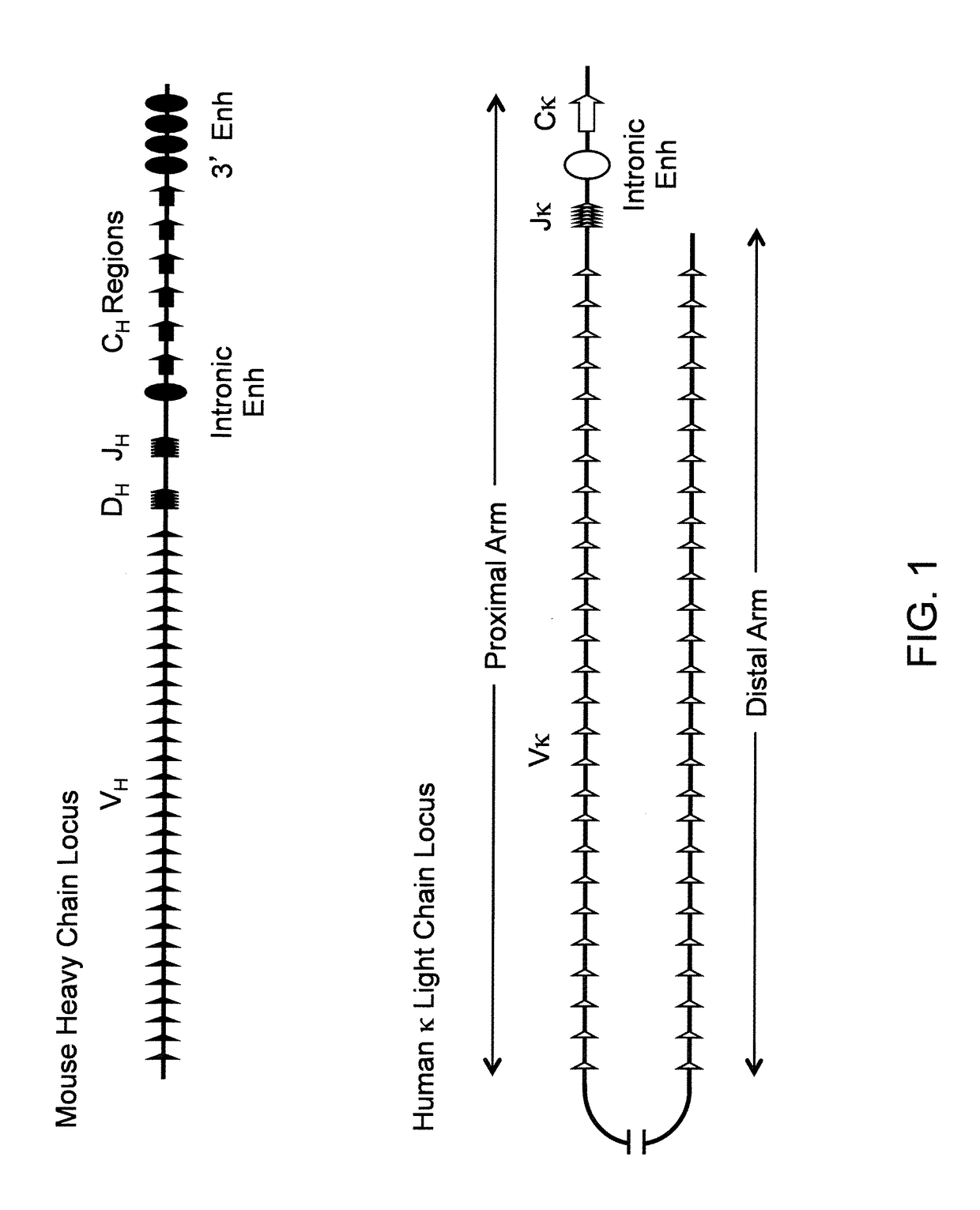
Antibody production
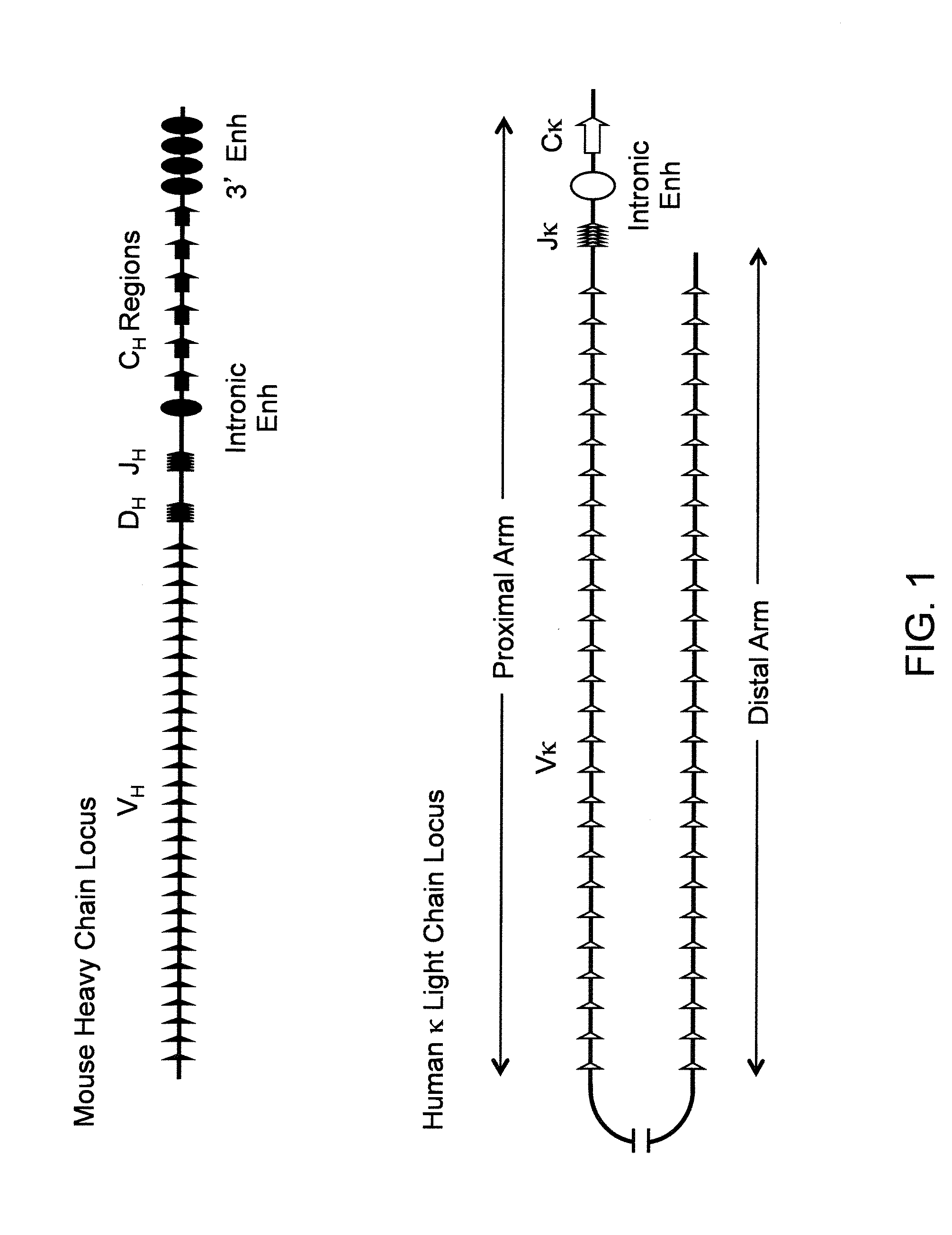
InactiveUS20110314563A1Antibody repertoire and diversityIncrease probabilityImmunoglobulinsImmunological disordersImmunoglobulin heavy chainAntigen challenge
A non-human mammal containing an endogenous lambda light chain gene locus, an endogenous kappa light chain gene locus and an endogenous heavy chain gene locus, each of which can re-arrange so that immunoglobulin heavy and light chain genes are formed and expressed in B-cells following antigen challenge but said loci have been mutated so that the ability to form functional immunoglobulin tetramers comprising re-arranged heavy and light chains produced from said mutated loci has been substantially reduced or eliminated.
Owner:ERASMUS UNIV MEDICAL CENT ROTTERDAM ERASMUS MC
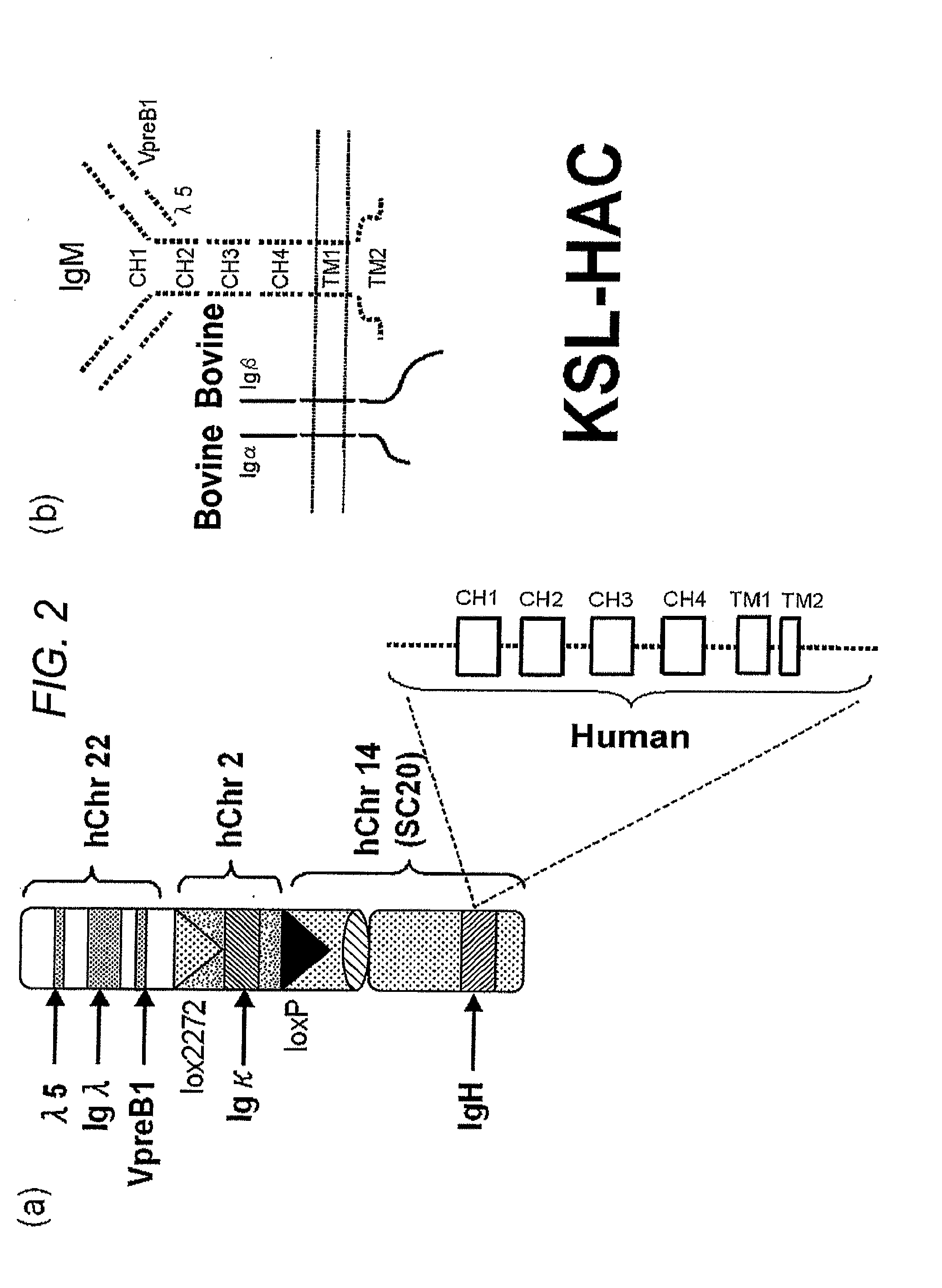
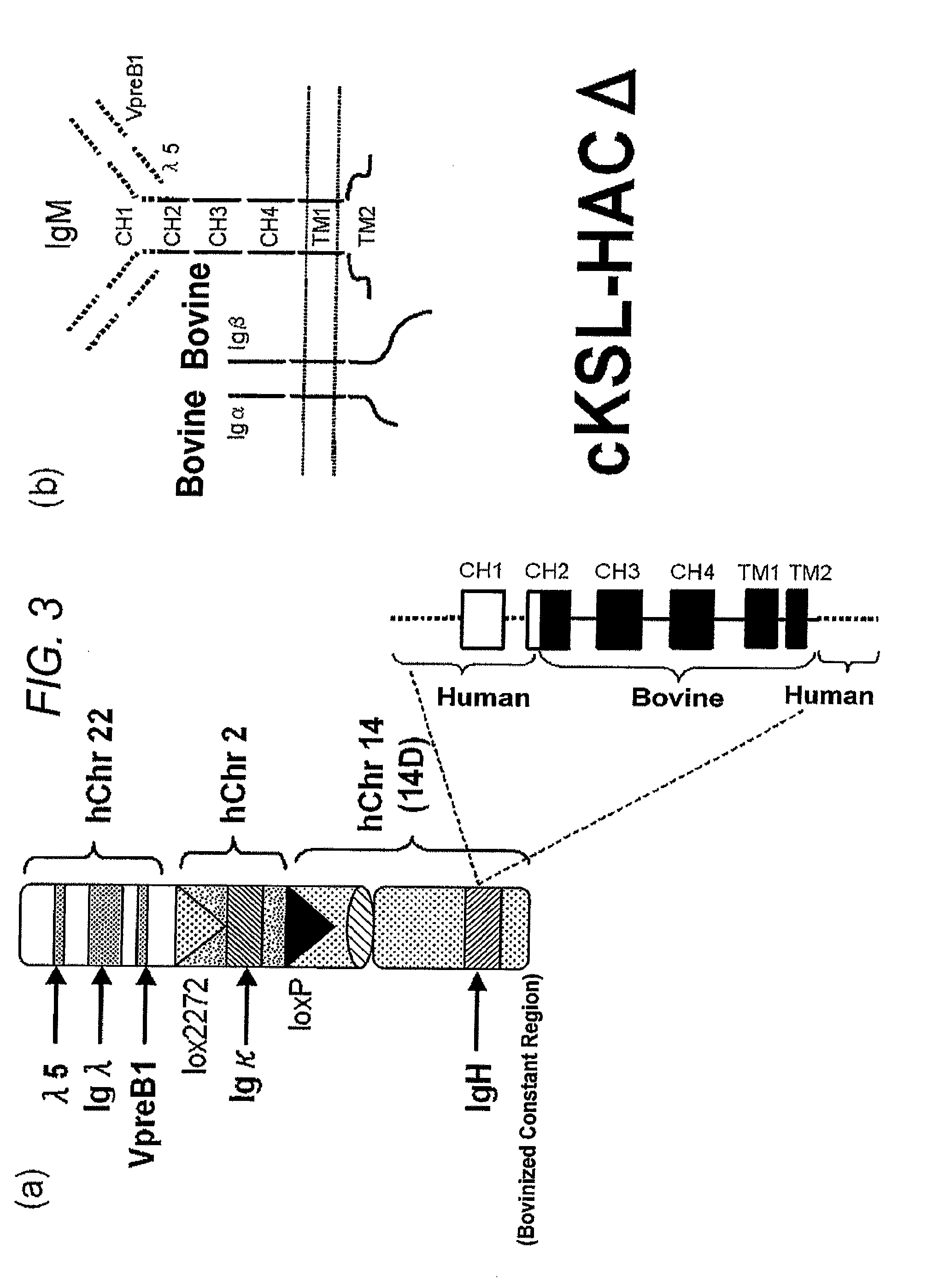
Human artificial chromosome vector
InactiveUS20120233715A1Efficient productionImprove efficiencyNucleic acid vectorImmunoglobulinsHuman artificial chromosomeGene
A human artificial chromosome vector comprising a human antibody heavy chain gene, a human antibody light chain gene, and a human antibody surrogate light chain gene.
Owner:SANFORD APPLIED BIOSCI
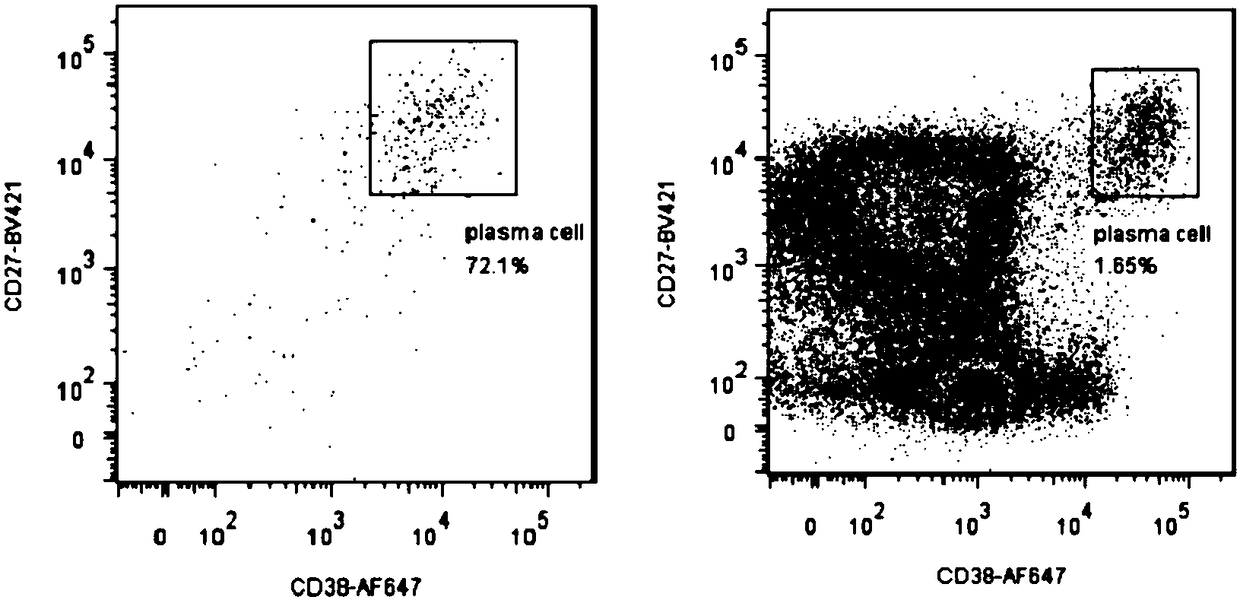
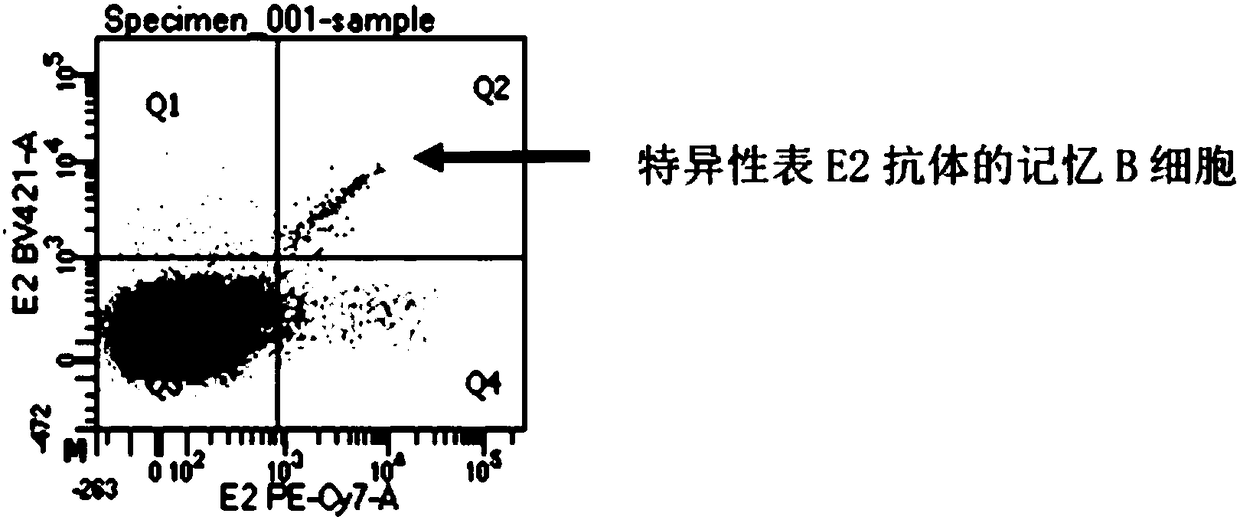
Preparation method and application of a high-throughput fully human antibody
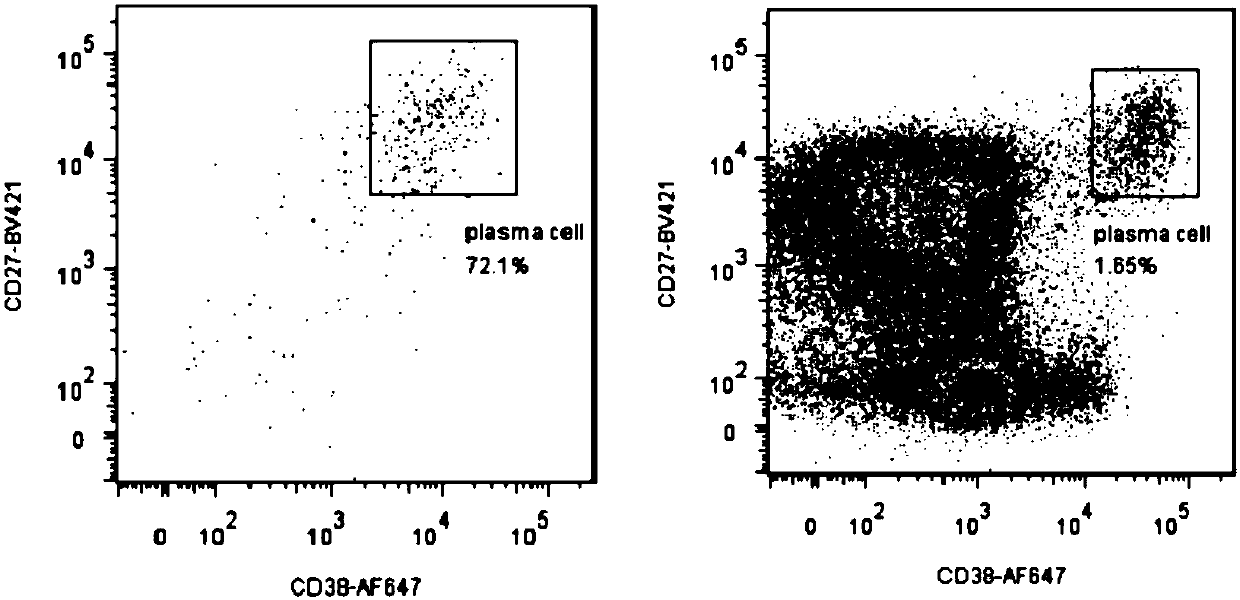
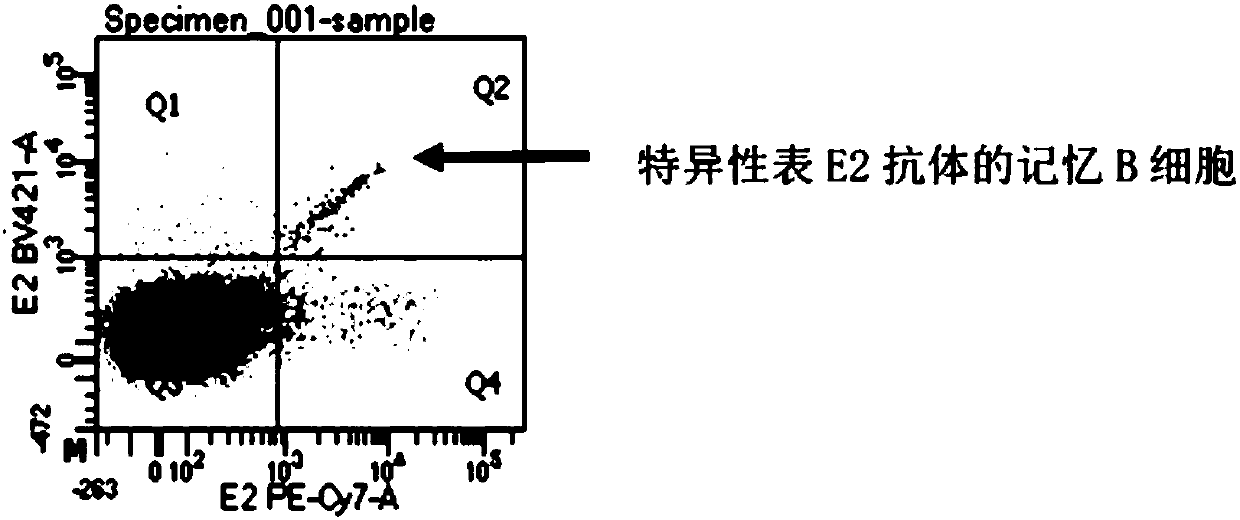
ActiveCN107760690BHigh affinityHigh potencyMicrobiological testing/measurementImmunoglobulinsPlasma cellHumanized antibody
The invention relates to gene engineering and antibody preparation and specifically discloses a preparation method and application of a high-throughput fully-humanized antibody. The preparation methodcomprises the following steps: separating peripheral blood monouclear cells; separating single cells of plasma cells or antigen-specificity memory B blymphocytes; amplifying the heavy-chain and light-chain gene variable regions of single B cell antibody by use of a primer provided by the inventor; establishing an expression system containing antibody heavy-chain and light-chain genes by use of alinear expression system containing a heavy-chain fragment or light-chain fragment constant area; finally, separating and purifying a fully humanized monoclonal antibody. By adopting the primer combination provided by the invention to amplify the genes in the antibody heavy-chain and light-chain variable areas, natural pairing of the light-chain and heavy-chain variable areas can be maintained, and the preparation method has the advantages of high gene diversity, high titer, full humanization, high antibody affinity, strong specificity, no heterologous serum reaction, no risk of propagating other infectious diseases, etc.
Owner:ZHUHAI TRINOMAB BIOTECHNOLOGY CO LTD
Preparation method and application of high-throughput fully-humanized antibody
ActiveCN107760690AHigh affinityHigh potencyMicrobiological testing/measurementImmunoglobulinsDiseaseAntibody Affinities
The invention relates to gene engineering and antibody preparation and specifically discloses a preparation method and application of a high-throughput fully-humanized antibody. The preparation methodcomprises the following steps: separating peripheral blood monouclear cells; separating single cells of plasma cells or antigen-specificity memory B blymphocytes; amplifying the heavy-chain and light-chain gene variable regions of single B cell antibody by use of a primer provided by the inventor; establishing an expression system containing antibody heavy-chain and light-chain genes by use of alinear expression system containing a heavy-chain fragment or light-chain fragment constant area; finally, separating and purifying a fully humanized monoclonal antibody. By adopting the primer combination provided by the invention to amplify the genes in the antibody heavy-chain and light-chain variable areas, natural pairing of the light-chain and heavy-chain variable areas can be maintained, and the preparation method has the advantages of high gene diversity, high titer, full humanization, high antibody affinity, strong specificity, no heterologous serum reaction, no risk of propagating other infectious diseases, etc.
Owner:ZHUHAI TRINOMAB BIOTECHNOLOGY CO LTD
Preparation method of transgenic animal capable of expressing human antibody
ActiveCN103571872AVector-based foreign material introductionAnimal husbandryGenomic DNAGene conversion
The invention relates to a preparation method of a mouse capable of producing a human antibody. The preparation method of the mouse capable of producing the human antibody comprises the step of carrying out hybridization on a transgenic mouse carrying a fragment containing a human antibody heavy chain gene locus part and a mouse carrying a fragment which is inserted into mouse genomic DNA (deoxyribonucleic acid) and contains an non-rearranged human antibody light chain gene locus part, wherein gene rearrangement and gene conversion can be carried out in the transgenic mouse by virtue of the human antibody heavy chain gene locus and the human antibody light chain gene locus, so as to produce various human immune globulins, and a mouse endogenous antibody heavy chain gene locus, a mouse endogenous antibody kappa light chain gene locus and a Lamda light chain gene locus are inactivated.
Owner:SHANDONG BIOANTY BIOLOGICAL TECH CO LTD
Enterpeptidase light chain variant with high activity and high stability
The present invention is characterized by that making the cysteine of 112th site of enterokinase light chain gene produce site-specific mutation, using colibacillus and microzyme to express enterokinase light chain variant, and utilizing Zn-Sepharose and STI-Sepharose affinity chromatography so as to obtain high-purity enterokinase light chain variant protein. As compared with wild enterokinase light chain said varient has higher stability and enzyme activity.
Owner:SUZHOU LANDING BIOPHARM CO LTD
Synthetic method of enterokinase light chain gene and preparation method of expression product of enterokinase light chain gene
InactiveCN102061302AImprove biological activityReduce degradationTransferasesMicroorganism based processesPurification methodsA-DNA
The invention relates to a synthetic method of an enterokinase light chain gene and a preparation method of an expression product of the enterokinase light chain gene, in particular to cow enterokinase light chain protein produced by adopting a DNA (Deoxyribonucleic Acid) recombination technology. The encoding gene of the protein is shown in a sequence (3) of a sequence list. The invention further discloses a fermentation and purification process of the cow enterokinase light chain protein. The optimized fermentation and purification method of the invention is utilized to successfully express the cow enterokinase light chain protein with high biological activity in a secretory expression mode. The cow enterokinase light chain protein is used as a tool protease for the specific cutting of fusion protein and is particularly suitable for researching the biological engineering pharmaceutical industry and gene engineering, biochemistry, molecular biology, and the like.
Owner:YANGTZE RIVER PHARMA GRP BEIJING HAIYAN PHARMA
Method for producing fluorescent antibacterial silks from transgenic silkworms
InactiveCN102321650AAchieving a Silky TransformationFunction increaseHybrid peptidesVector-based foreign material introductionFluorescenceChemical factor
The invention discloses a method for producing fluorescent antibacterial silks from transgenic silkworms. Gene targeting transgenic vectors fused with silkworm light-chain genes to express fluorescent proteins and antibacterial peptides are constructed on a vector of a homologous arm with silkworm silk fibroin light-chain genes; gene targeting is carried out through a transgenic method; the transgenic silkworms for producing fluorescent antibacterial silks are acquired after being subjected to the fluorescence screening and the identifying of molecular biology. According to the method disclosed by the invention, the quality of the silks produced from the silkworms is improved, the function of the silks is extended and the application value of the silks is increased; the fluorescent antibacterial silks are not polluted by DNA and other physical and chemical factors and have no potential harmfulness from transgenic organisms, so that the biological safety is high; and the simple preparation process and the low cost are achieved.
Owner:SUZHOU UNIV
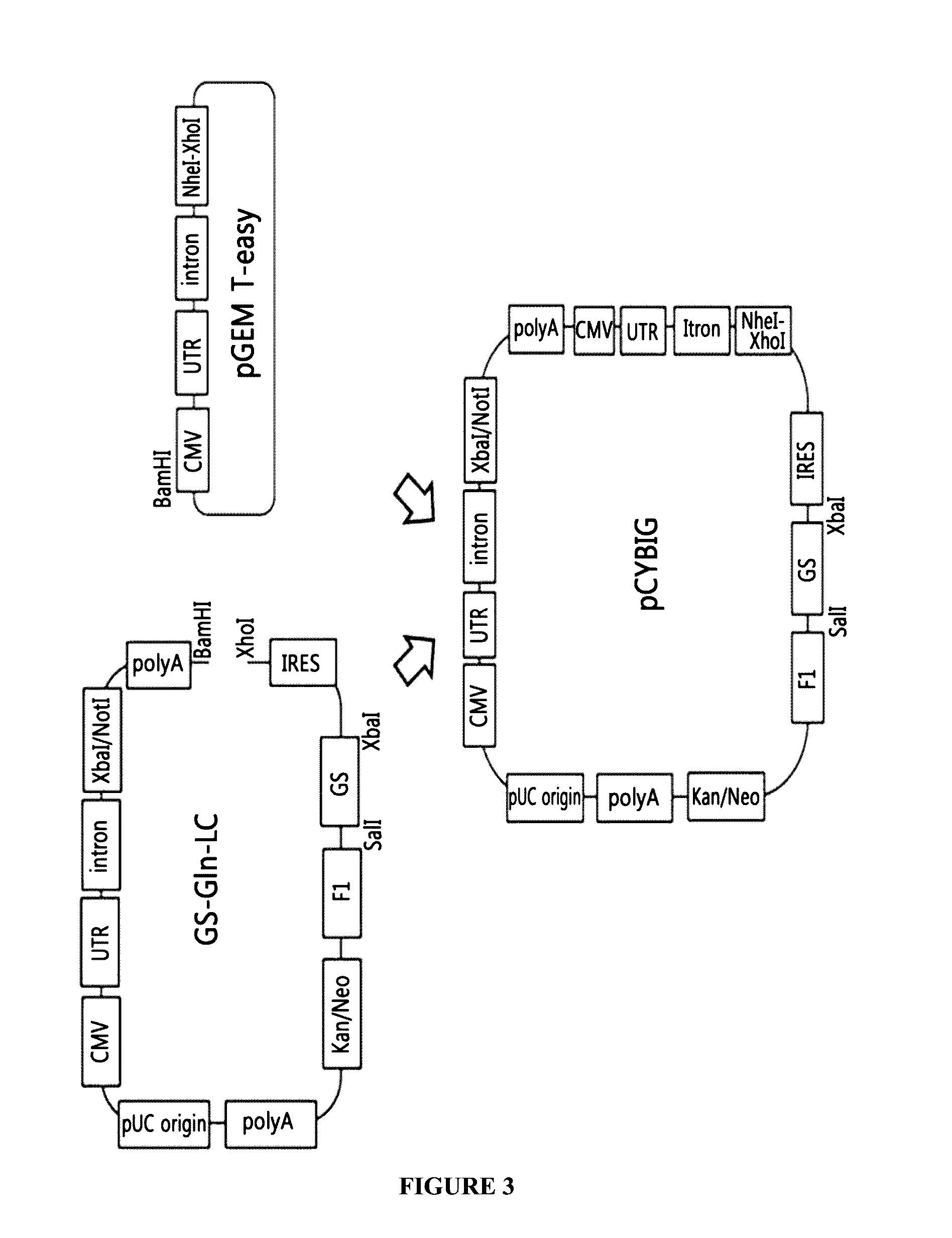
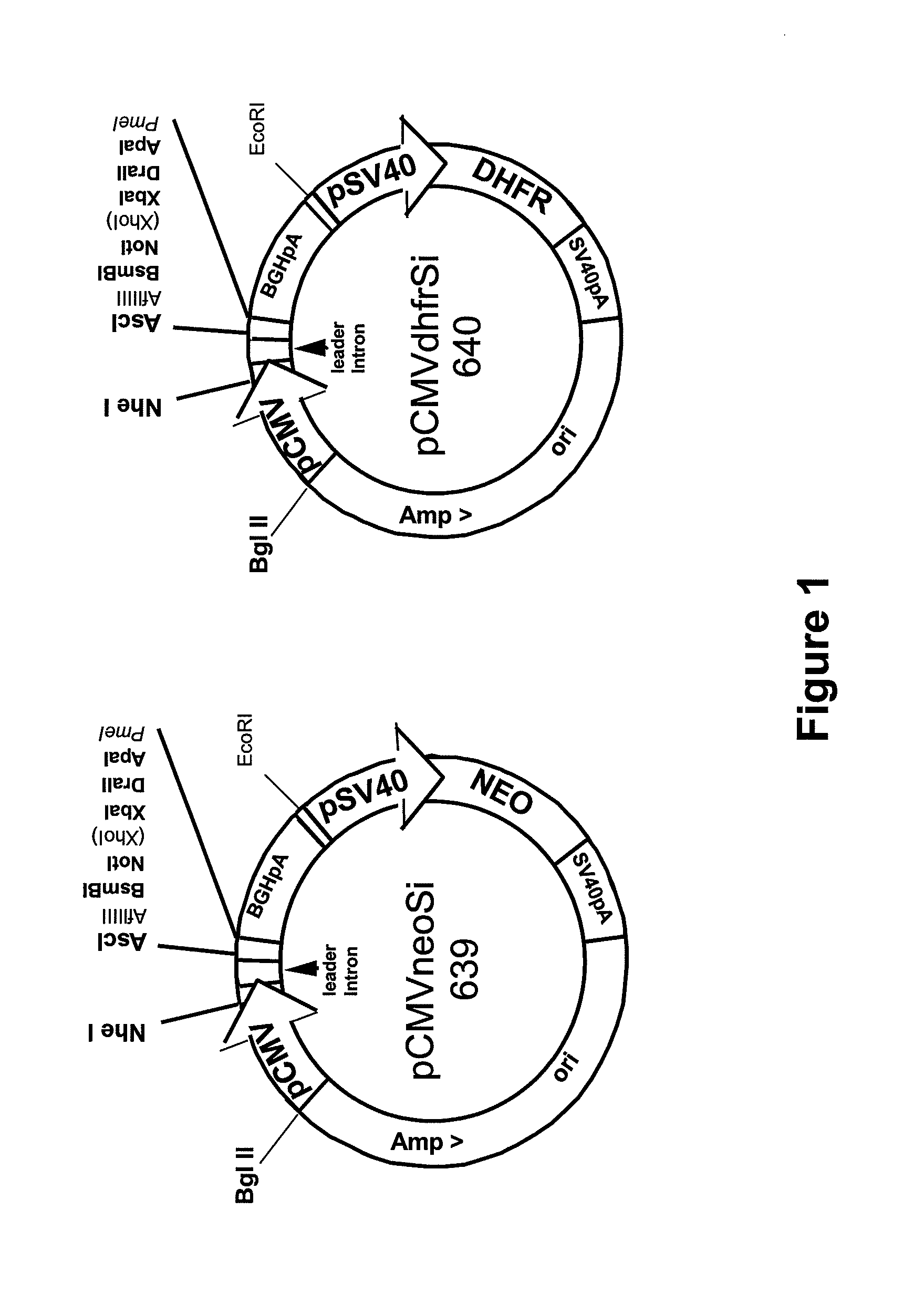
Bicistronic Expression Vector For Antibody Expression And Method For Producing Antibody Using Same
ActiveUS20160281106A1Improve expression efficiencyImprove efficiencyVectorsGenetically modified cellsAntibody expressionIntein
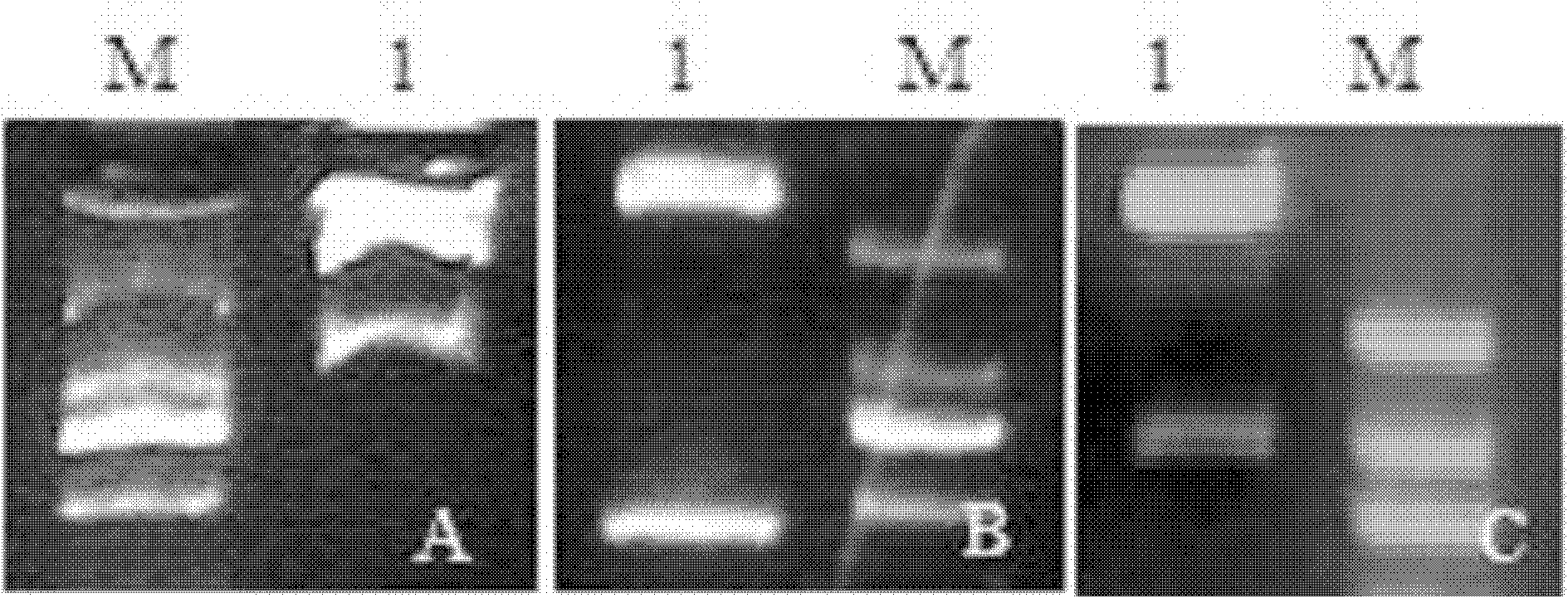
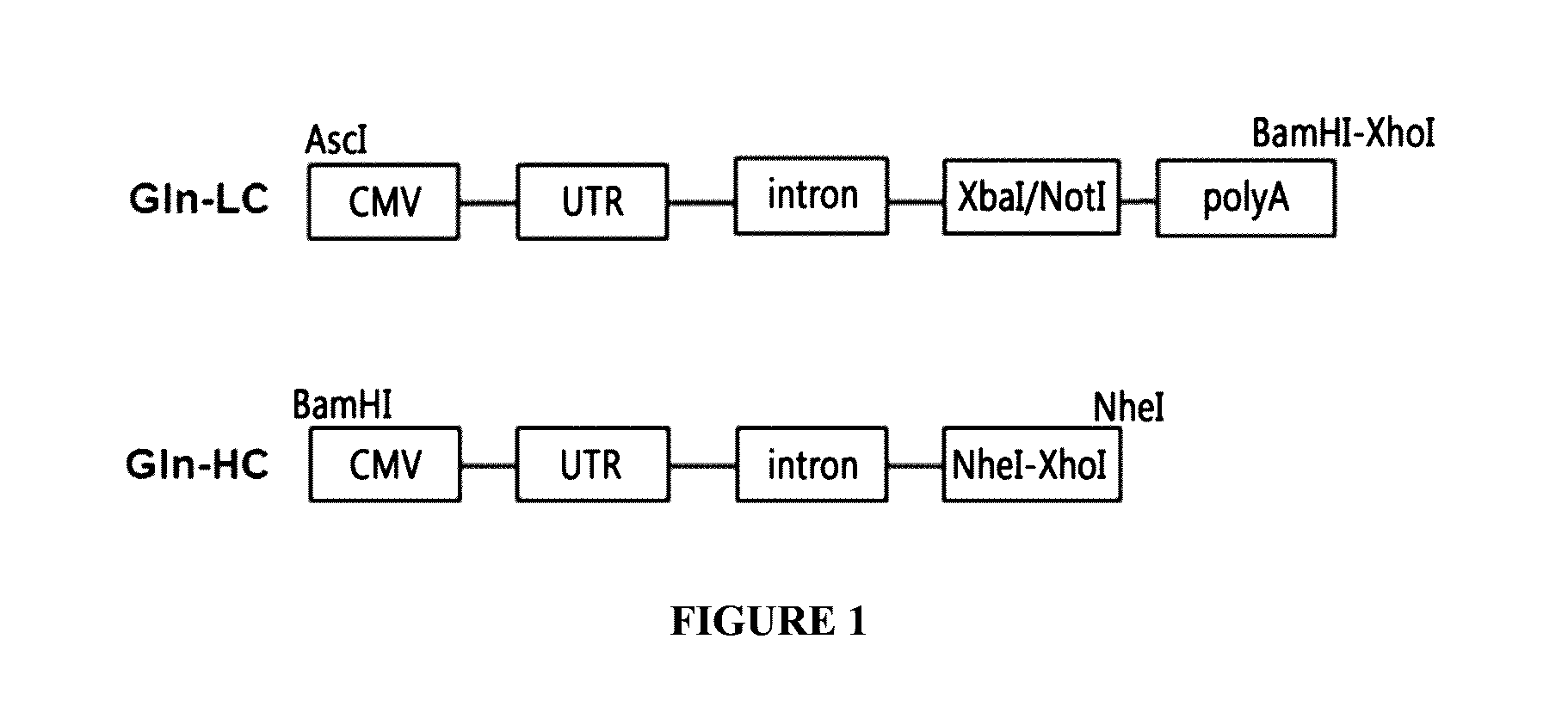
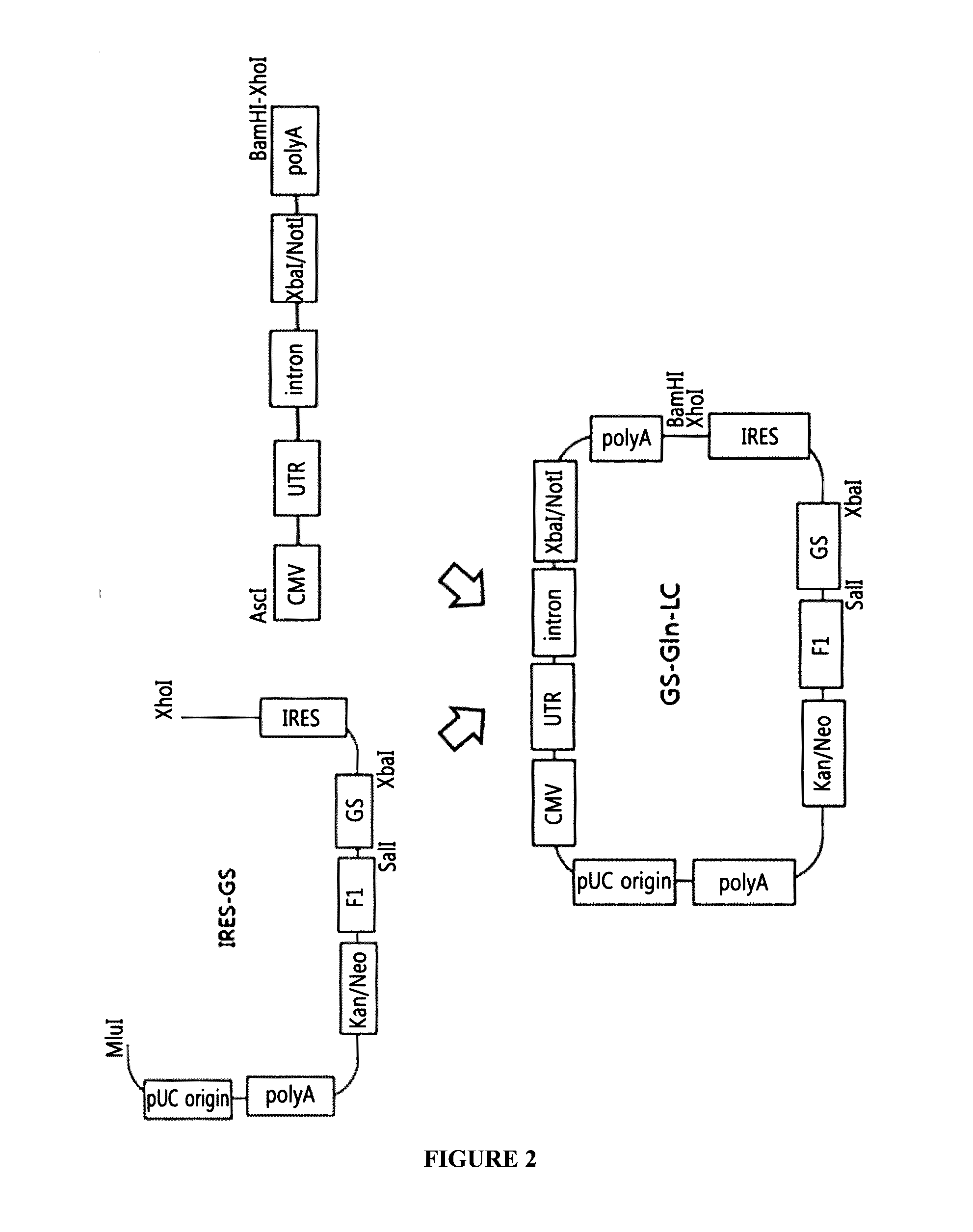
The present invention relates to a bicistronic expression vector for antibody expression, an animal cell transfected with the expression vector, and a method for producing an antibody including culturing the animal cell, in which the expression vector includes a first expression cassette including ‘promoter-UTR-intron-antibody light chain gene-polyA’ and a second expression cassette including ‘promoter-UTR-intron-antibody heavy chain gene-internal ribosome entry site (IRES)-amplification gene-polyA’. An expression vector capable of expressing a desired antibody with high efficiency can be constructed using the bicistronic expression vector including an intron for antibody expression according to the present invention, and the expression vector can produce the antibody by culturing the transfected animal cell with stability and high efficiency.
Owner:PRESTIGE BIOPHARMA
A method for preparing transgenic animals capable of expressing human antibodies
ActiveCN103571872BVector-based foreign material introductionAnimal husbandryAntibody fragmentsGene conversion
The present invention relates to a method for preparing a mouse capable of producing human antibodies, the method comprising combining a transgenic mouse carrying a partial fragment of a heavy chain locus of a human antibody, and a human antibody carrying an unrearranged human antibody inserted into the genomic DNA of the mouse. Mouse crosses of partial fragments of antibody light chain loci that can undergo gene rearrangement and gene conversion in transgenic mice to produce various human immunoglobulins. Among them, the mouse endogenous antibody heavy chain locus, the mouse endogenous antibody κ light chain locus and the Lamda light chain locus were inactivated.
Owner:SHANDONG BIOANTY BIOLOGICAL TECH CO LTD
Humanized rodents that express heavy chains containing vl domains
InactiveUS20170094955A1Reduces and eliminates ADAM activityEliminates bindingHybrid immunoglobulinsNucleic acid vectorHuman animalGenetic Materials
Non-human animals, tissues, cells, and genetic material are provided that comprise a modification of an endogenous non-human heavy chain immunoglobulin sequence and that comprise an ADAM6 activity functional in a rodent (e.g., a mouse), wherein the non-human animals rearrange human immunoglobulin light chain gene segments in the context of heavy chain constant regions and express immunoglobulin-like molecules comprising human immunoglobulin light chain variable domains fused to heavy chain constant domains that are cognate with human immunoglobulin light chain variable domains fused to light chain constant domains.
Owner:REGENERON PHARM INC
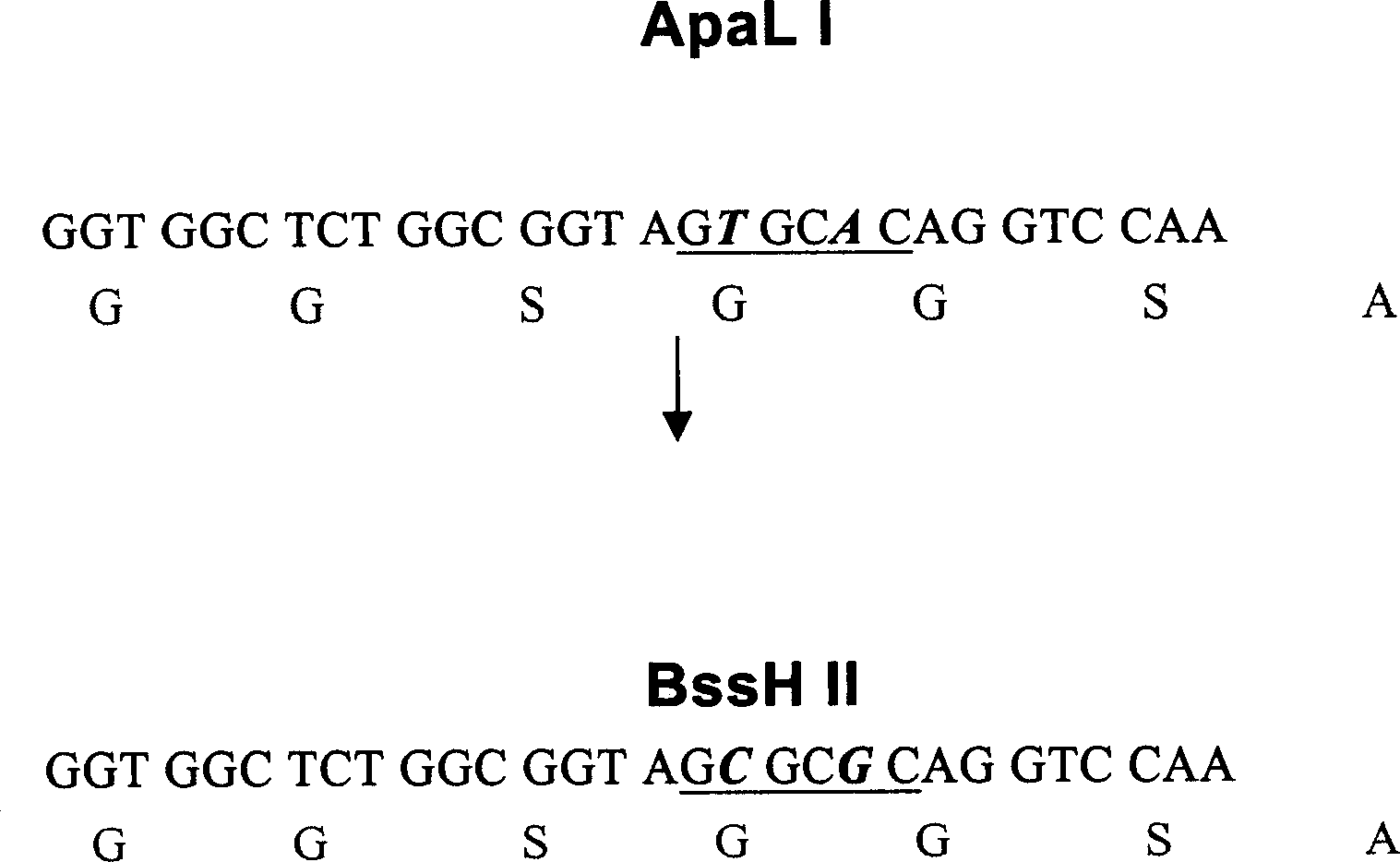
Improved bacteriophage expression carrrier
An improved expression carrier pHEN2 used to screen the phagemid in single-chain antibody by phage display method features that the enzyme severing site ApaL1 in its multiple enzyme severing sites is removed or replaced by BssH II, so more products coded by the light-chain gene of human immunoglobulin can be effectively inserted in single-chain antibody library to ensure its diversity and high volume.
Owner:CHINESE NAT HUMAN GENOME CENT AT SHANGHAI
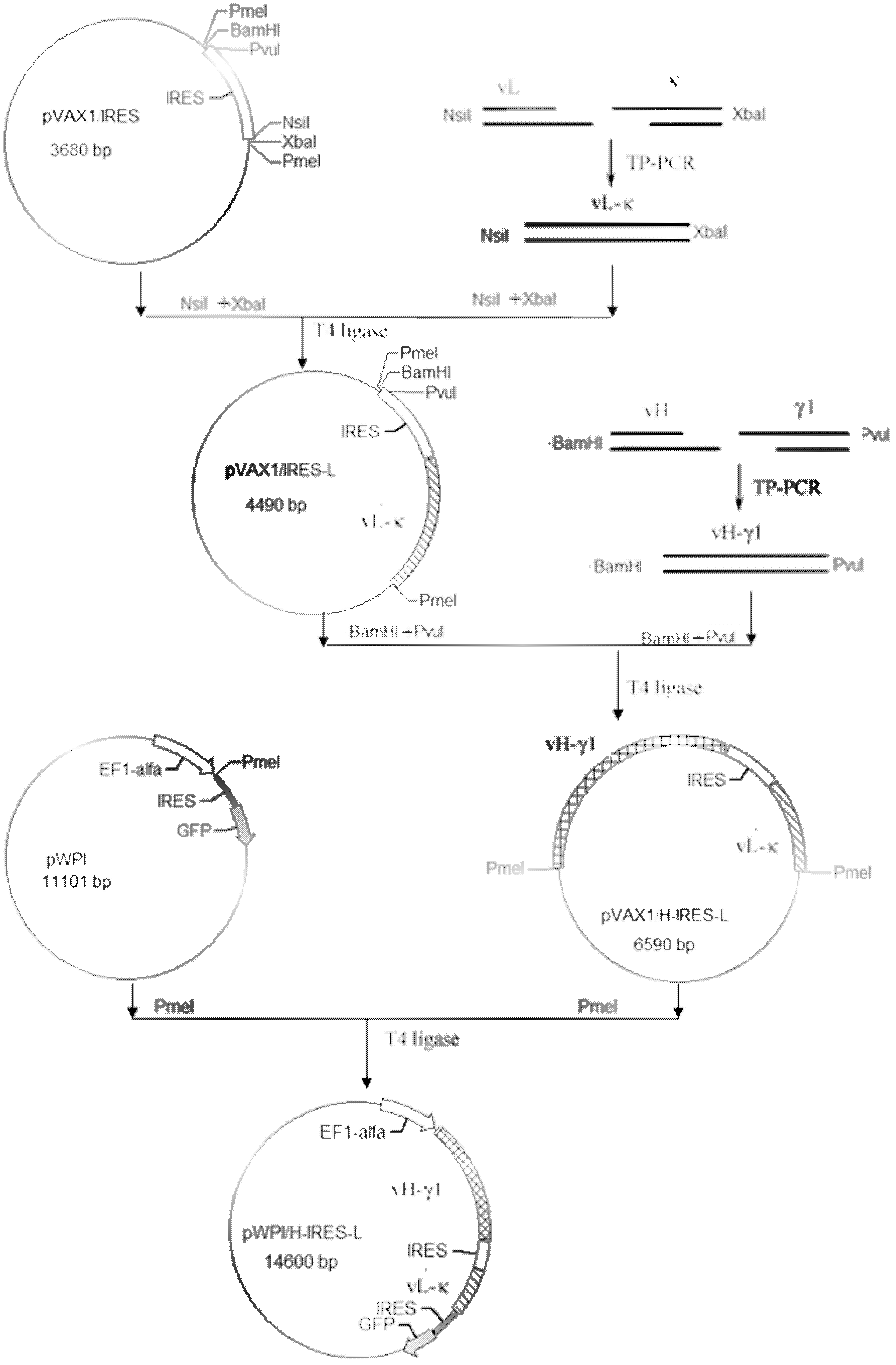
Anti-p185<erbB2> human-mouse chimeric antibody lentivirus expression vector and construction method thereof
InactiveCN102618582ALarge capacityNo toxicityFermentationVector-based foreign material introductionEnzyme digestionChimeric antibody
The invention discloses an anti-p185<erbB2> human-mouse chimeric antibody lentivirus expression vector and a construction method thereof. Antihuman-p185<erbB2> mouse-derived monoclonal antibody variable region genes (vH and vL) and human-IgG1 constant region genes (gamma 1 and kappa) are obtained by amplification by PCR (polymerase chain reaction) process. The vH is spliced with the gamma 1 and the vL is spliced with the kappa by three-primer PCR process so as to obtain a chimeric heavy chain (H) gene and a chimeric light chain (L) gene. The chimeric heavy chain (H) gene and the chimeric light chain (L) gene are inserted onto an IRES (internal ribosome entry site) element of plasmid pVAX1 / IRES downstream and upstream respectively. H-IRES-L is cut off from the pVAX1 / H-IRES-L using endonucleases and is inserted into the lentivirus expression vector pWPI so as to construct a lentivirus expression pWPI / H-IRES-L. Corresponding enzyme digestion and sequencing appraisal show that the expression is consistent to that of the expected design. After transfection of cells 293T, the chimeric heavy chain (H) gene and the chimeric light chain (L) gene are co-expressed, and the chimeric antibody can be combined with p185<erbB2> molecular specificity. By the vector and the construction method, basis is laid for anti-p185<erbB2> engineering antibodies in the future.
Owner:广西壮族自治区肿瘤防治研究所
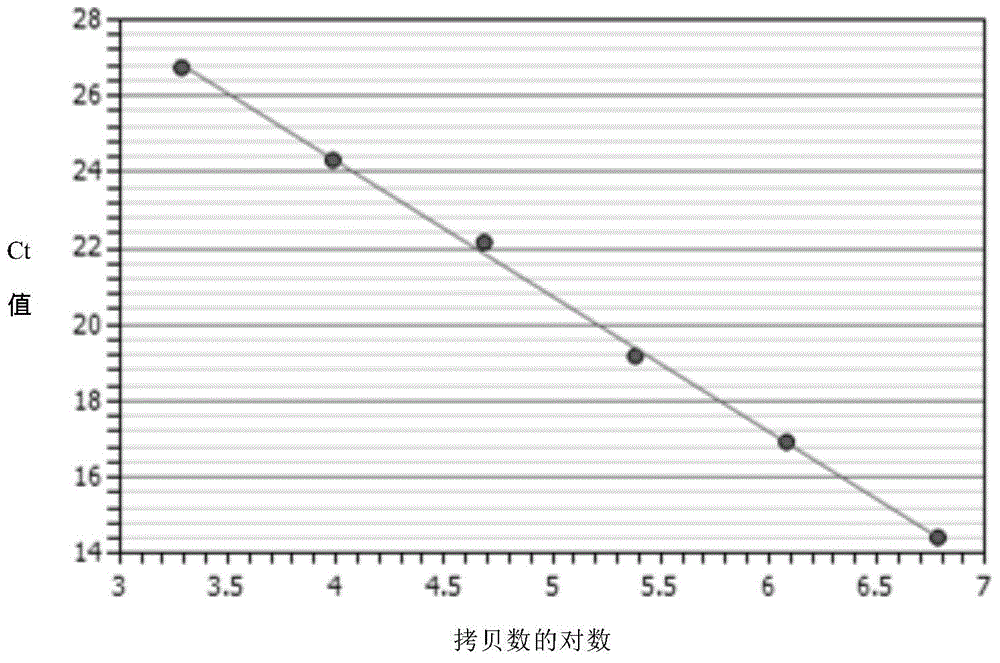
Method for quantitative PCR detection of exogenous gene copy number in transgenic cell
InactiveCN106191216AHigh detection sensitivityWide linear rangeMicrobiological testing/measurementHigh fluxPcr method
The invention relates to a method for quantitative detection of the light and heavy chain gene copy numbers of antibody molecules in a transgenic animal cell CHO. An SYBR-Green quantitative PCR method is adopted to realize quantitative, accurate and high-flux detection of the light chain gene copy number and the heavy chain gene copy number of the antibody molecules. The method has the advantages of high detection sensitivity, wide linear range and reliable result, can be used for researching the stability of antibody cell strains and screening stable high-expression engineering cell strains.
Owner:GENOR BIOPHARMA
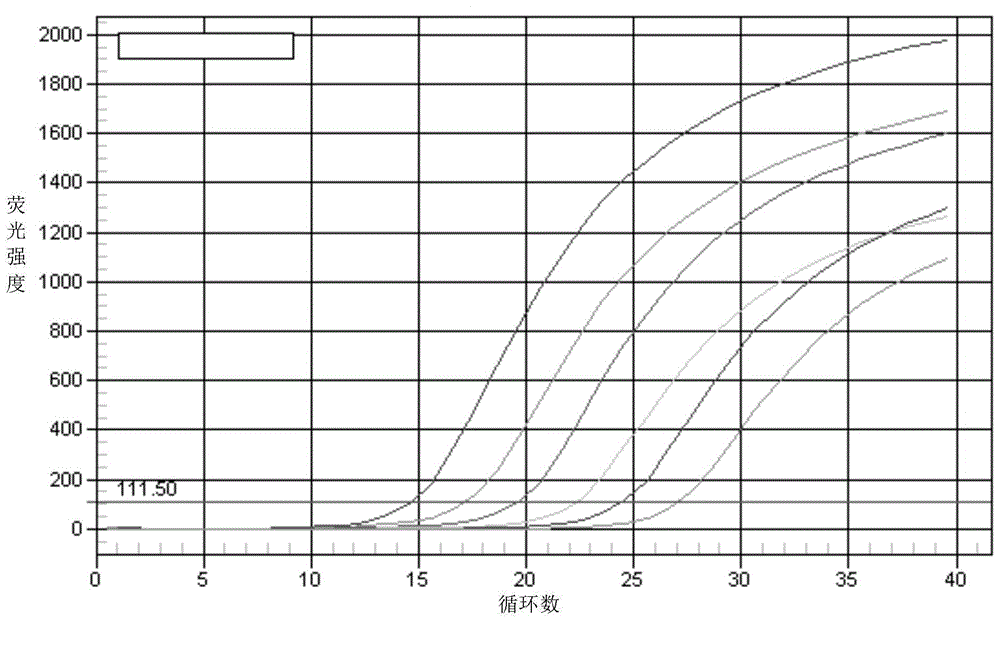
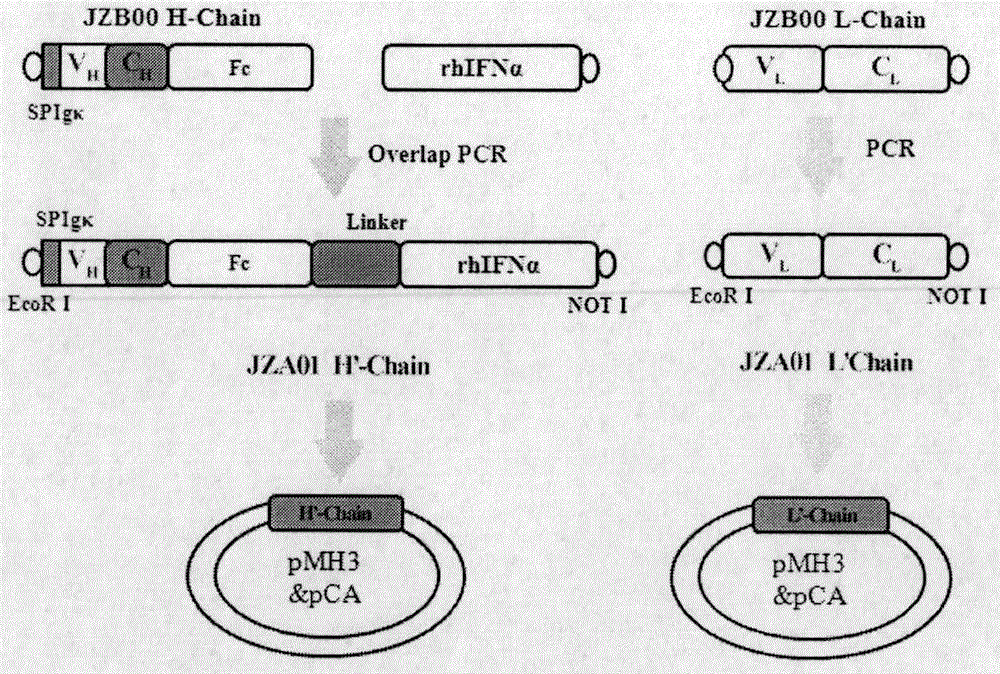
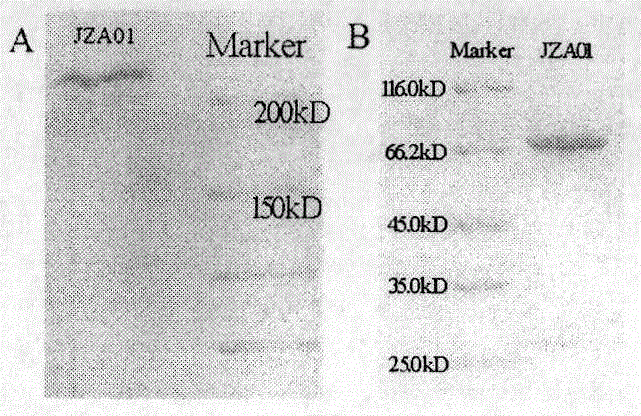
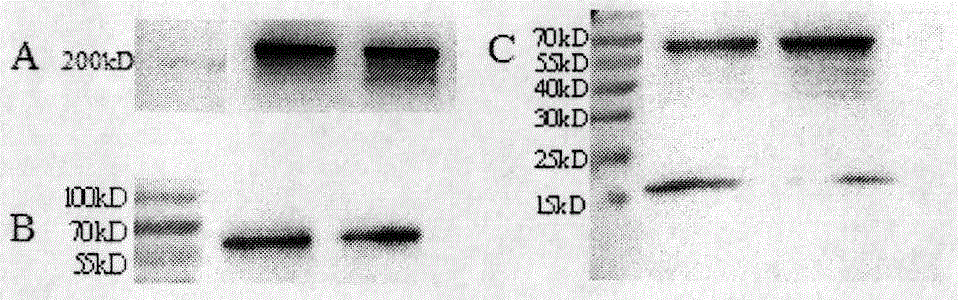
Preparation and application of cytokine fusion antibody
Belonging to the technical field of genetic engineering antibodies, the invention specifically discloses a vascular endothelial growth factor receptor 2 (VEGFR2 or KDR) targeted fusion antibody JZA01of antibody JZB00 and interferon alpha (IFN alpha), a preparation method and application thereof. The invention also discloses the amino acid sequences of heavy chain and light chain immunoglobulin molecules of JZA01. The invention also provides a construction method of JZA01 heavy chain and light chain gene, CHO cells are transfected, monoclone is picked by limited dilution method, then by means of eukaryotic cell secretory expression and affinity chromatography purification, the fusion antibody can be obtained. The fusion antibody provided by the invention can specifically bind VEGFR2 and inhibit tumor angiogenesis, the coupled interferon part also can also play a direct tumor killing and immunoregulation role, and no toxic or side effect is generated at the same time, thereby better inhibiting tumor growth. The fusion antibody can inhibit the proliferation of human umbilical vein endothelial cells (HUVEC) and some tumor cells in vitro.
Owner:CHINA PHARM UNIV
Cell strain constructing method for preparing anti-EGFR completely-humanized monoclonal antibody
PendingCN109988751ASEC promotionIncreased pI-cIEFGenetically modified cellsImmunoglobulinsDihydrofolate reductaseChinese hamster
The invention discloses a cell strain constructing method for preparing an anti-EGFR completely-humanized monoclonal antibody. The method comprises the following steps: in a POOL screening step aftercells of Chinese hamster ovary cell dihydrofolate reductase deficient strains CHO / DHFR- complete a transfection step, screening cell strains integrated with a heavy chain gene by using an SFM4CHO culture medium without containing HT in the first stage, and screening out the cell strains integrated with a light chain gene by using Blasticidin in the culture medium at the same time; and in the second stage, adding MTX to increase copy numbers of the light and heavy chain genes integrated into cell strain genomes, as to improve an expression quantity. An efficient stable cell strain, of which theexpression quantity reaches 2.0-3.0 g / L, suitable for a large trial production process is firstly cloned and screened out, product SEC, pI-cIEF, a de-sugaring molecular weight, and antibody activitybased on the cells are remarkably improved, so possibility is provided for realizing industrialization of recombining the anti-EGFR completely-humanized monoclonal antibody in China.
Owner:JINING UNIV
Human-derived anti-amylin antibody and preparation method thereof
InactiveCN106279414AImprove featuresHigh activityImmunoglobulins against hormonesFermentationAmylinBiology
The invention discloses a human-derived anti-amylin antibody and a preparation method thereof, and belongs to the field of immune globulin. The heavy chain variable region amino acid sequence of the antibody is shown as SEQ ID NO:1, the light chain variable region amino acid sequence of the antibody is shown as SEQ ID NO:2, the heavy chain variable region DNA sequence of the antibody is shown as SEQ ID NO:3, and the light chain variable region DNA sequence of the antibody is shown as SEQ ID NO:4. The heavy chain gene is an IgG1 subclass VH3 gene family, and the light chain gene is a Lambda gene family. The human-derived amylin antibody is screened out by establishing a high-capacity phage antibacterial library, the antibody has good antigen specificity and antigen combination activity, production of an amylin detecting kit is guaranteed, and wide application prospects are achieved.
Owner:GENERAL HOSPITAL OF TIANJIN MEDICAL UNIV
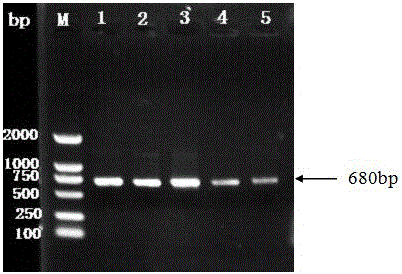
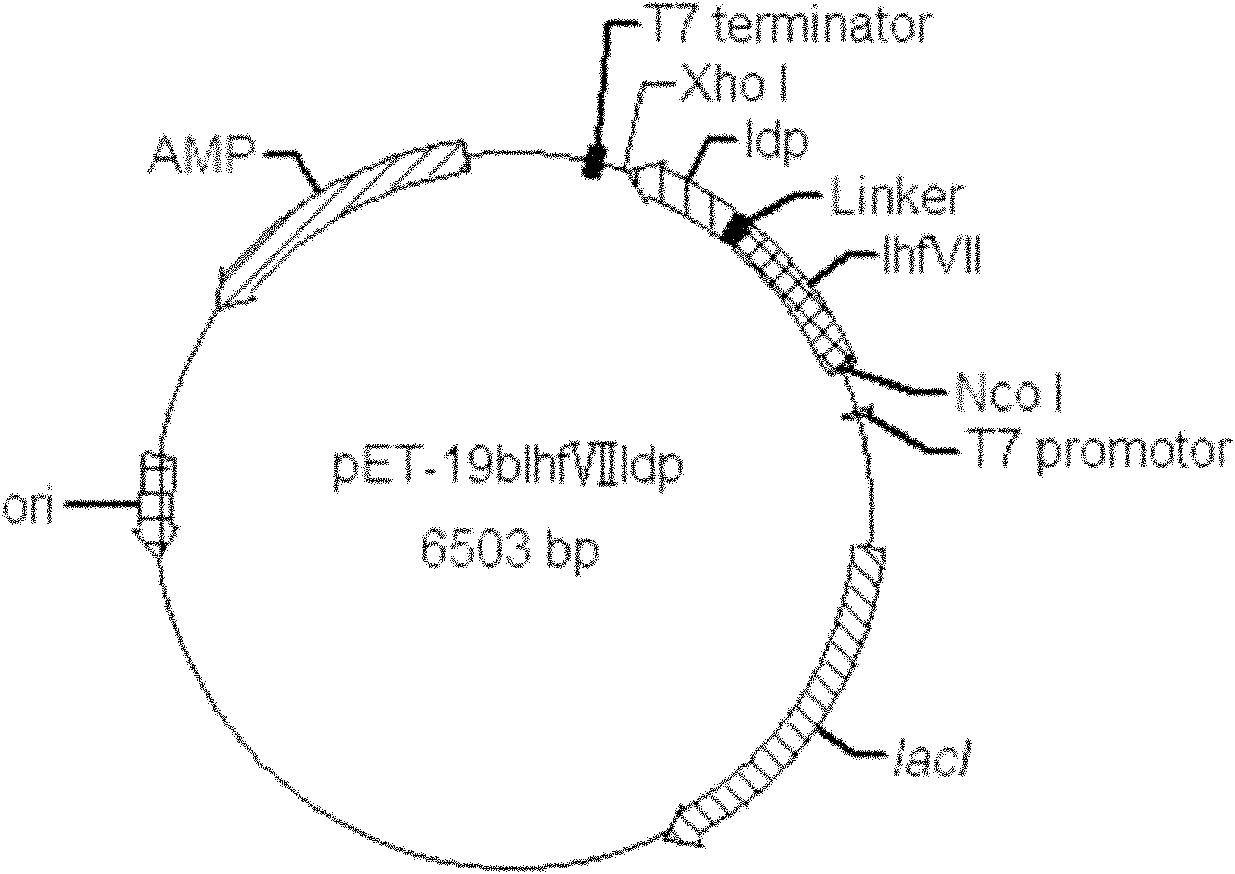
Fusion protein lhfvii-ldp and enhanced fusion protein lhfvii-ldp-ae and applications thereof
InactiveCN102277364AStrong targetingLow immunogenicityPeptide/protein ingredientsPharmaceutical non-active ingredientsNucleotideEukaryotic plasmids
A fusion gene encoding the fusion protein LhFVII-LDP is formed by linking the gene encoding the light chain of human blood coagulation factor VII and the gene encoding lidamycin prosthetic protein, and its nucleotide sequence is shown in SEQ ID NO. 3. A recombinant plasmid expressing the fusion protein LhFVII-LDP contains a fusion gene encoding the fusion protein LhFVII-LDP. The fusion protein LhFVII-LDP is expressed from the above-mentioned recombinant plasmid, and its amino acid sequence is shown in SEQ ID NO.4 in the sequence listing. The enhanced fusion protein LhFVII-LDP-AE is composed of the fusion protein LhFVII-LDP whose amino acid sequence is shown in SEQ ID NO.4 in the sequence listing and the enediyne chromophore AE. The fusion protein LhFVII-LDP and the strengthened fusion protein LhFVII-LDP-AE can be used in the preparation of medicines for treating tumors.
Owner:SICHUAN UNIV +1
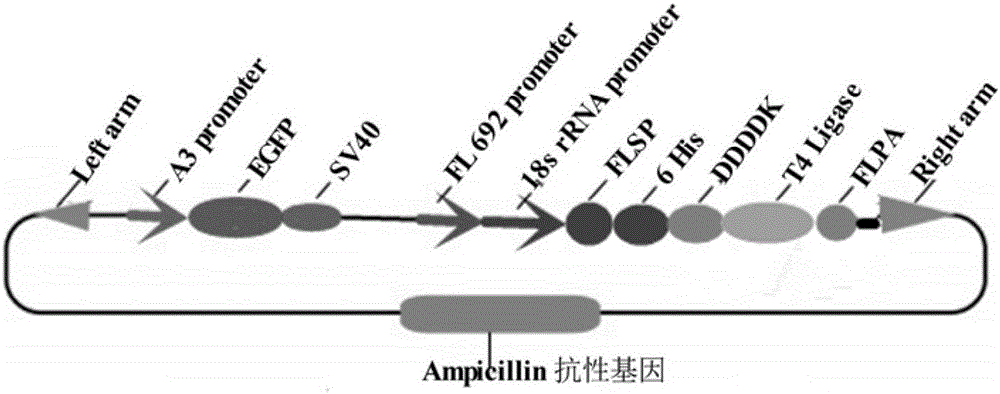
Dual-promoter universal plasmid for expressing T4 ligase of domestic silkworm middle silk gland bioreactor as well as application and method of dual-promoter universal plasmid
The invention discloses a dual-promoter universal plasmid for expressing T4 ligase of a domestic silkworm middle silk gland bioreactor as well as application and a method of the dual-promoter universal plasmid. The plasmid takes piggy Bac transposons as a foundation and carries an Amp resistance gene; the plasmid comprises function expression frames of a T4 ligase gene used as an exogenous gene and a green fluorescence EGFP (Enhanced Green Fluorescent Protein) gene used as a marker gene; the plasmid is constructed by utilizing a molecular biology method and two special restriction enzyme cutting sites containing ApaI and NheI are formed between DDDDK and a fibroin light-chain gene polyA; the universal plasmid is subjected to dual-enzyme digestion by adopting the ApaI and the NheI; after the universal plasmid is connected with the T4 ligase gene, the universal plasmid and an auxiliary plasmid are commonly injected into a domestic silkworm fertilized ovum; the green fluorescence protein gene and the T4 ligase gene are transferred into a domestic silkworm genome through utilizing properties of the transposons and are stably inherited and expressed to obtain transgenic domestic silkworms. The transgenic domestic silkworms are screened with the help of a fluorescence marker gene and domestic silkworm silk gland cells are used for specifically synthesizing and secreting T4 ligase protein.
Owner:ZHEJIANG UNIV
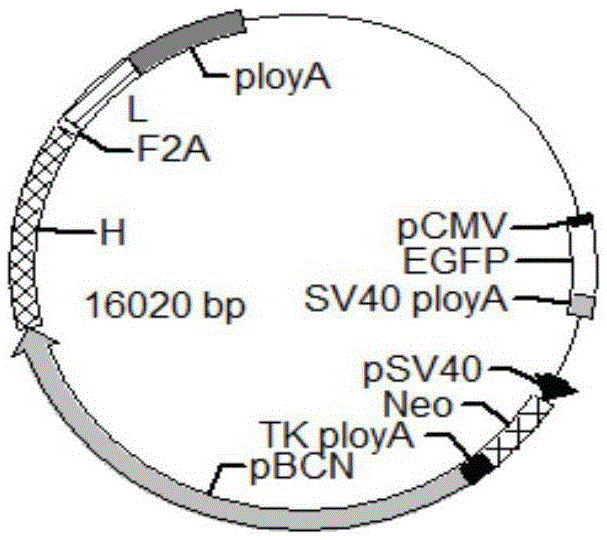
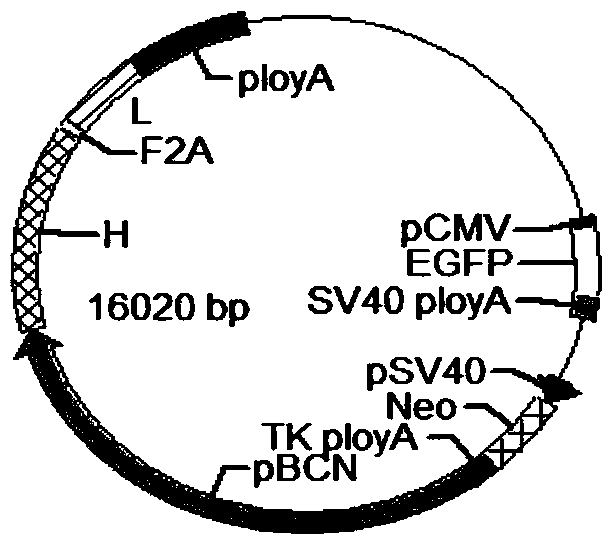
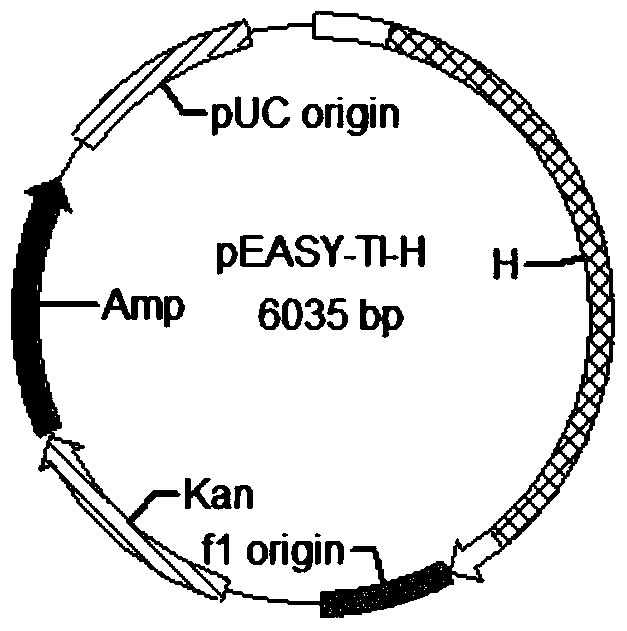
Anti-p185<erbB2> human-mouse chimeric antibody ChAb26, mammary gland specific expression vectors, transgenic FVB mouse, and preparation method of transgenic FVB mouse
InactiveCN105646704ALow immunogenicityAvoid reactionImmunoglobulins against animals/humansVector-based foreign material introductionHigh activityImmunogenicity
The invention discloses an anti-p185<erbB2> human-mouse chimeric antibody ChAb26. The anti-p185<erbB2> human-mouse chimeric antibody ChAb26 is mainly composed of a heavy chain gene H and a light chain gene L, and the heavy chain gene H and the light chain gene L are respectively connected to a mammary gland specific expression plasmid pBC1 in order to obtain p185<erbB2> human-mouse chimeric antibody ChAb26 mammary gland specific expression vectors pBC1-H and pBC1-L. The pBC1-H and pBC1-L are linearized, the linearized pBC1-H and pBC1-L are introduced to FVB mouse zygotes to produce a transgenic FVB mouse, and the anti-p185<erbB2> human-mouse chimeric antibody ChAb26 can be obtained through mammary gland specific expression with the transgenic FVB mouse as a mammary gland bioreactor. The antibody reduces the immunogenicity of the v monoclonal antibody, and a medicinal recombinant protein produced through the transgenic FVB mouse has the advantages of high output, low cost, high activity, easy large-scale formation, shortening of the manufacturing period of new medicines, and huge market potential.
Owner:GUANGXI MEDICAL UNIVERSITY
Gene fragment for controlling silking of major cotton leafroller as well as cloning method and application thereof
Owner:YANGZHOU UNIV
Bombyx mori posterior silk gland bioreactor dual-promoter universal plasmid for expressing T4 ligase and application and method thereof
The invention discloses a Bombyx mori posterior silk gland bioreactor dual-promoter universal plasmid for expressing T4 ligase and application and a method thereof. The plasmid takes a piggyBac transposon as a basis, carries an Amp resistant gene and contains a T4 ligase gene serving as an exogenous gene and a function expression box of a green fluorescent EGFP gene serving as a marker gene. The plasmid is constructed by using a molecular biology method, and two restriction enzyme cutting sites unique to ApaL and NheI are contained between DDDDK and a fibroin light chain gene poly. After the ApaL and NheI double restriction enzyme cutting universal plasmid is connected with the T4 ligase gene, the plasmid and an auxiliary plasmid are jointly injected into Bombyx mori zygote, and characteristics of the transposon are utilized to guide the green fluorescent protein gene and the T4 ligase gene into a Bombyx mori genome and to be stably inherited and expressed to obtain transgenic Bombyx mori. The transgenic Bombyx mori is screened with the help of the fluorescent marker gene, and a Bombyx mori silk gland cell is utilized to specifically synthesize T4 ligase protein.
Owner:ZHEJIANG UNIV
Single chain antibody of human anti-placenta growth factor
InactiveCN101643508AHigh affinityImmunoglobulins against growth factorsPeptide preparation methodsAntigenCDNA library
The invention relates to a single chain antibody of human anti-placenta growth factor, which is encoded by a heavy-chain gene with an SEQ NO.1 sequence and light-chain gene with an SEQ NO.2 sequence.The invention also provides a preparation method thereof, comprising the following steps in sequence: amplifying human total RNA heavy-chain variable region, light-chain variable region and constant region gene of the whole set by a PCR technique, splicing and constructing an overall-length single-chain antibody cDNA library; expressing the cDNA library by an externally-coupled transcription / translation system to obtain an antibody-ribosome-mRNA compound; performing affiliation selection and clution on the compound by a magnetic bead coated by specific antigen; amplifying selected destinationantibody gene by the PCR technique; and expressing the amplified antibody gene using the externally-coupled transcription / translation system repeatedly to obtain the single-chain antibody. The antibody is adapted to prepare medicament for treating ovarian cancer.
Owner:SHANGHAI TENTH PEOPLES HOSPITAL
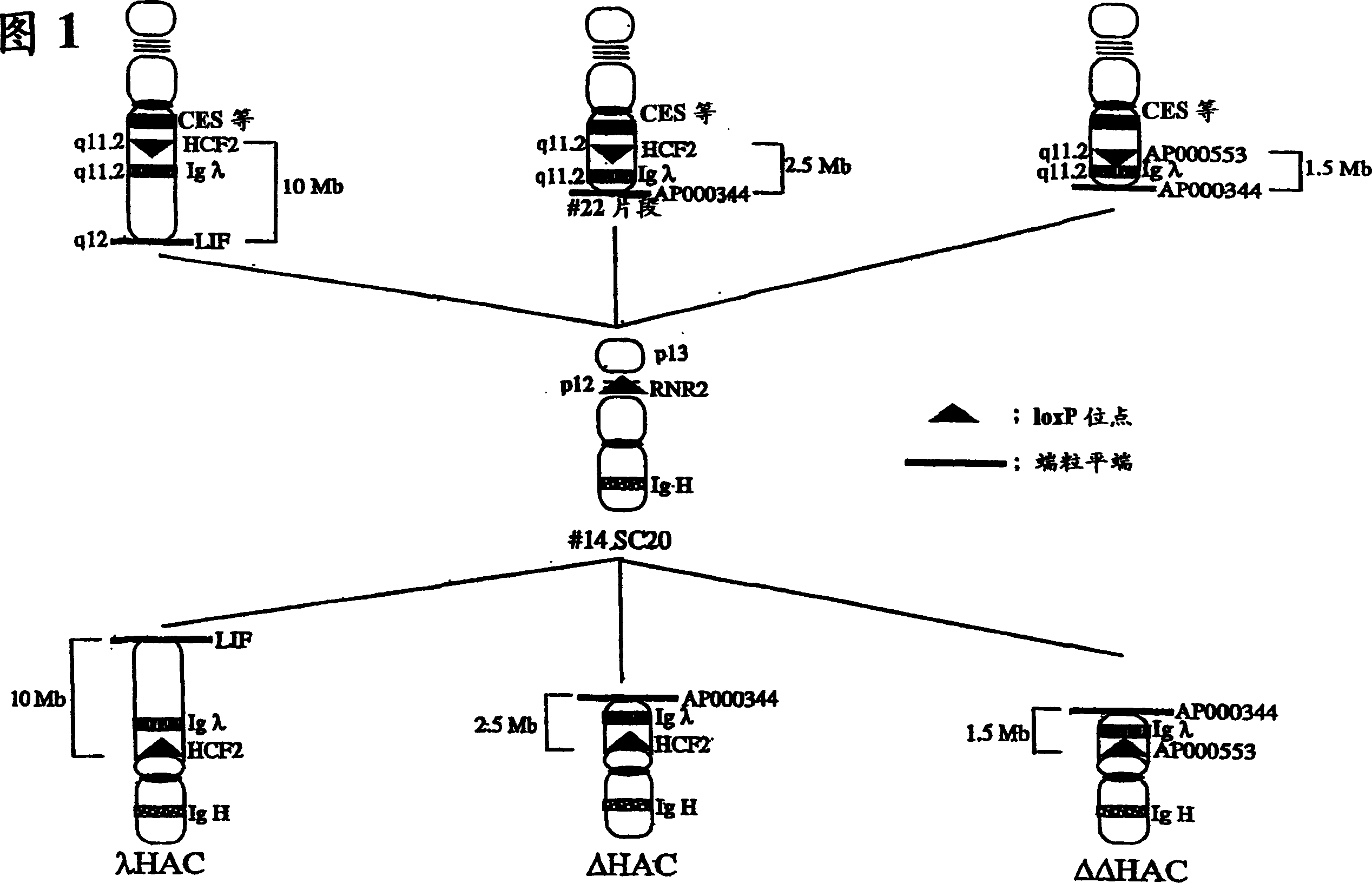
Human artificial chromosome containing human antibody lambda light chain
InactiveCN1789416AGenetic material ingredientsImmunoglobulins against cytokines/lymphokines/interferonsHuman animalHuman artificial chromosome
The present invention relates to a human artificial chromosome which is genetically transmissible to the next generation with high efficiency and the method for using the same. More specifically, the present invention relates to: a human artificial chromosome in which an about 3.5 Mb to about 1 Mb region containing an antibody » light chain gene derived from human chromosome 22 is bound to a chromosome fragment which is transmissible to a progeny through a germ line of a non-human animal, said chromosome fragment is derived from another human chromosome; a non-human animal carrying the human artificial chromosome and an offspring thereof; a method for producing the non-human animal; a method for producing a human antibody using the non-human animal or an offspring thereof; and a human antibody-producing mouse carrying the human artificial chromosome.
Owner:KYOWA HAKKO KIRIN CO LTD +1
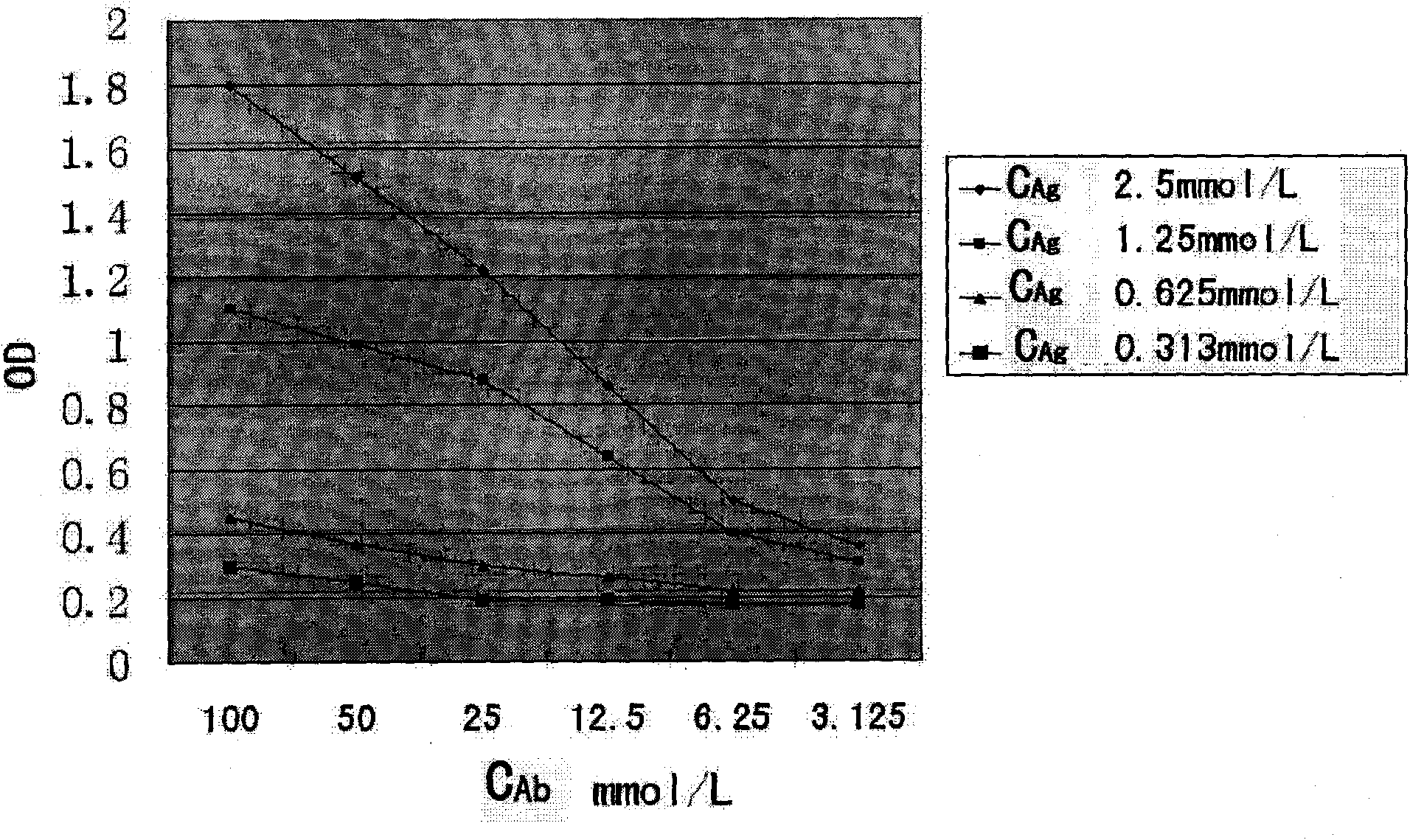
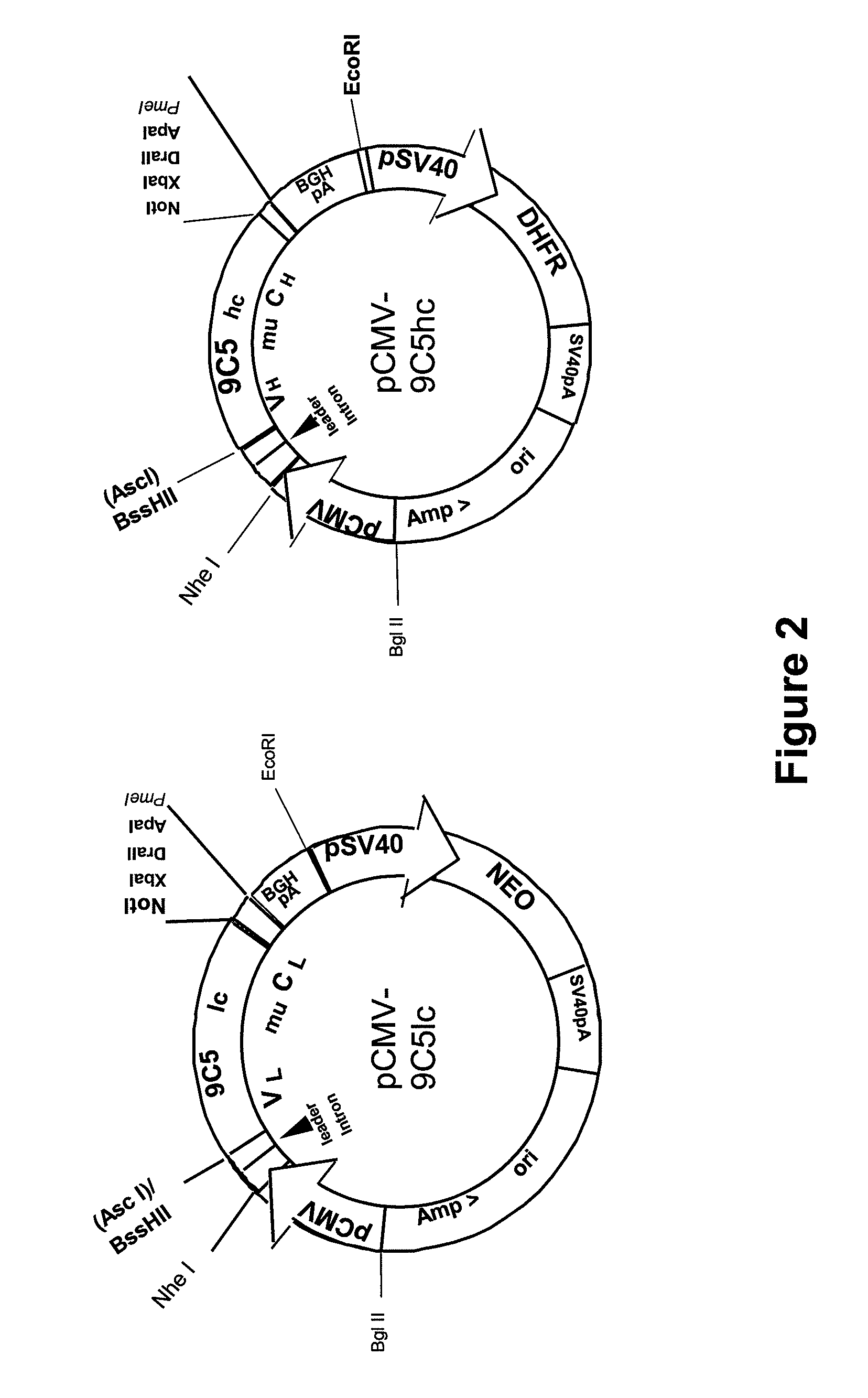
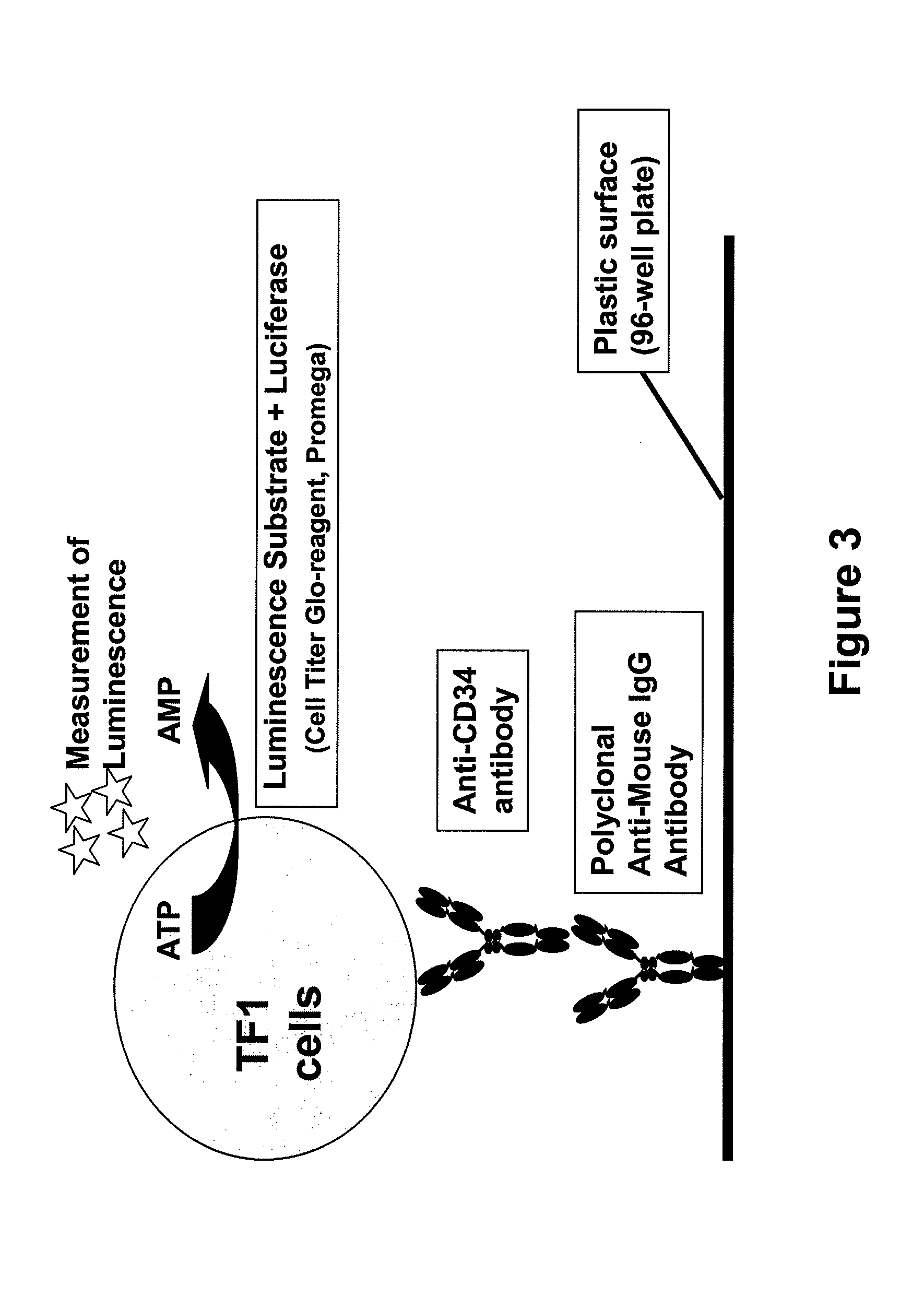
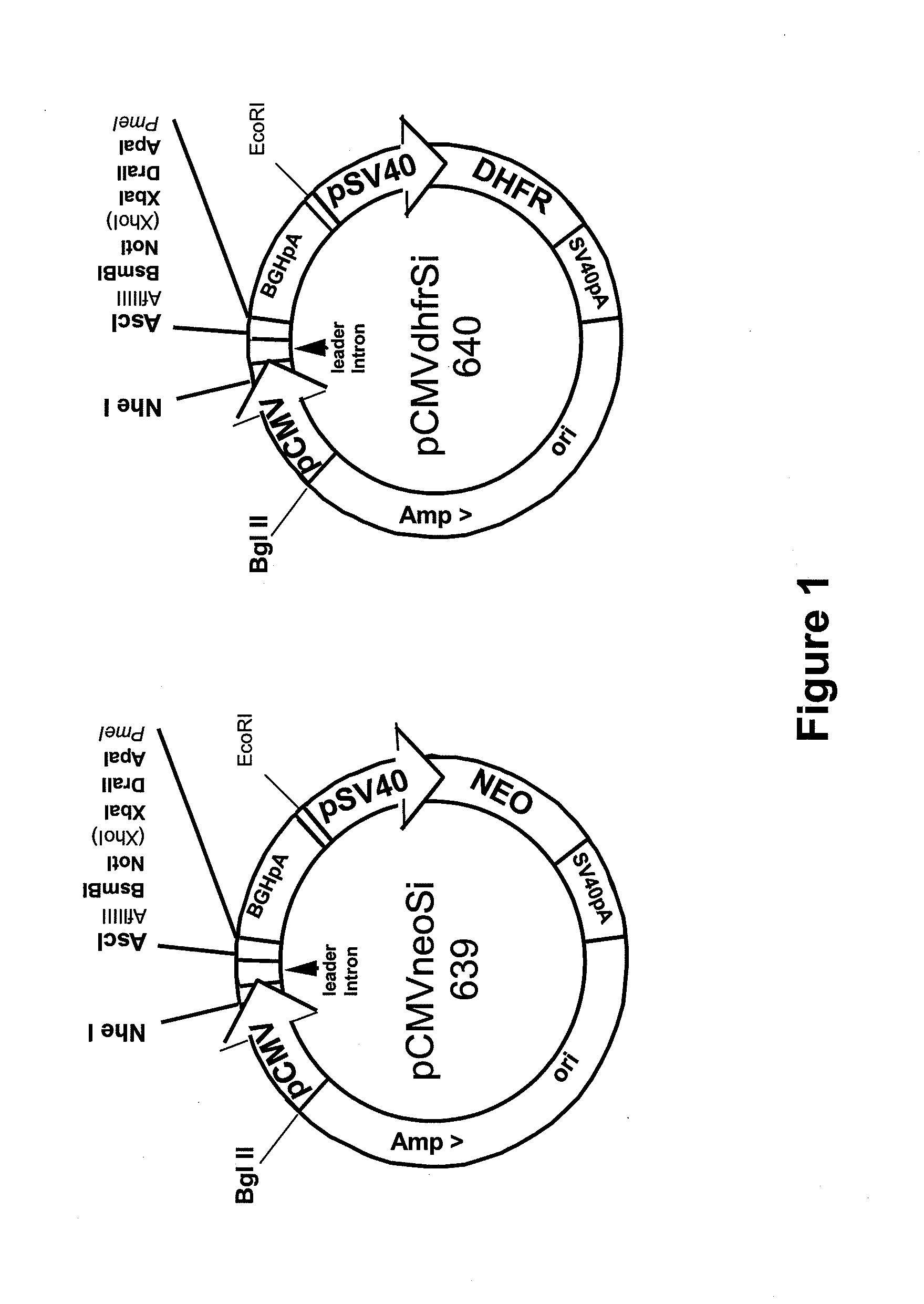
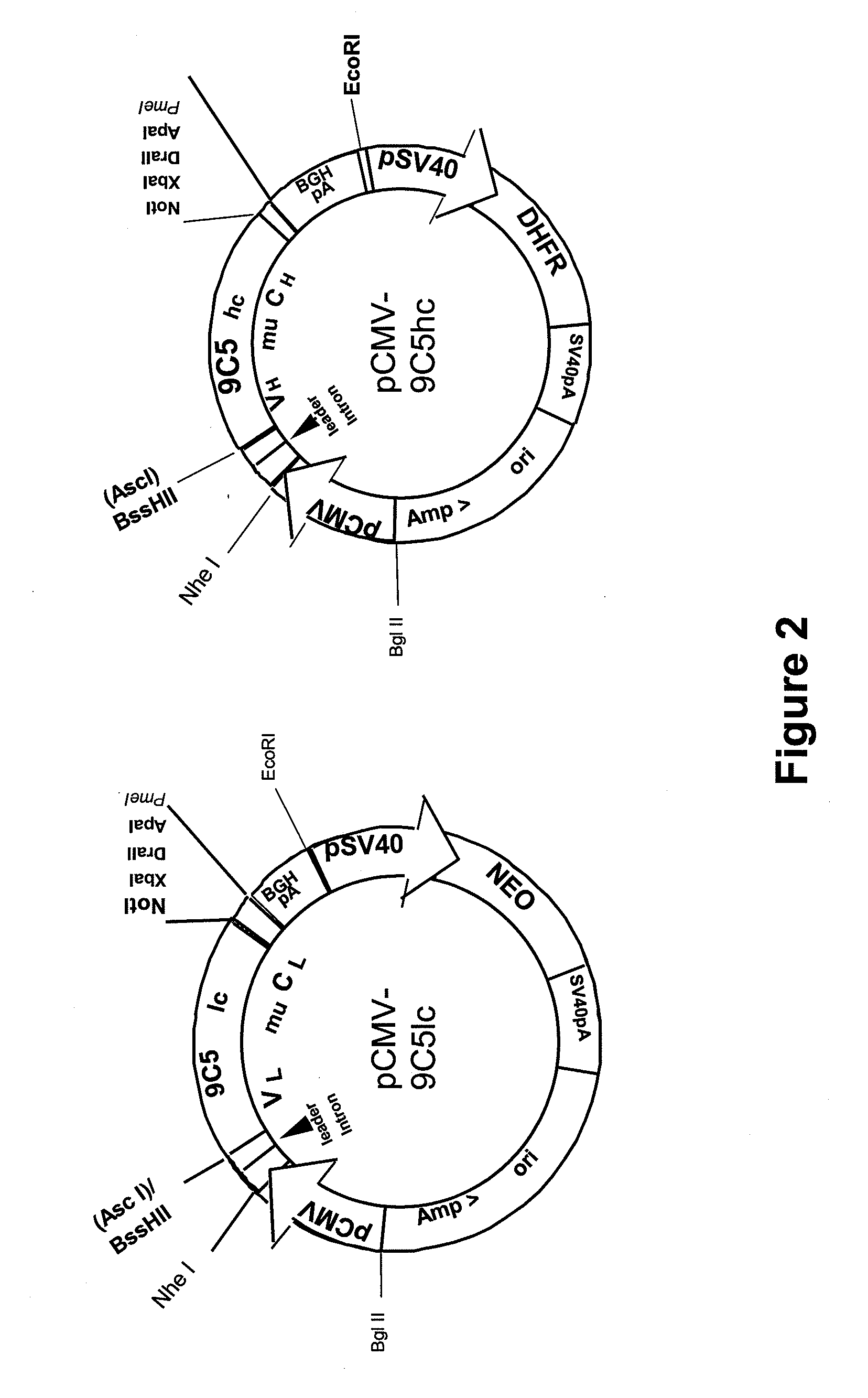
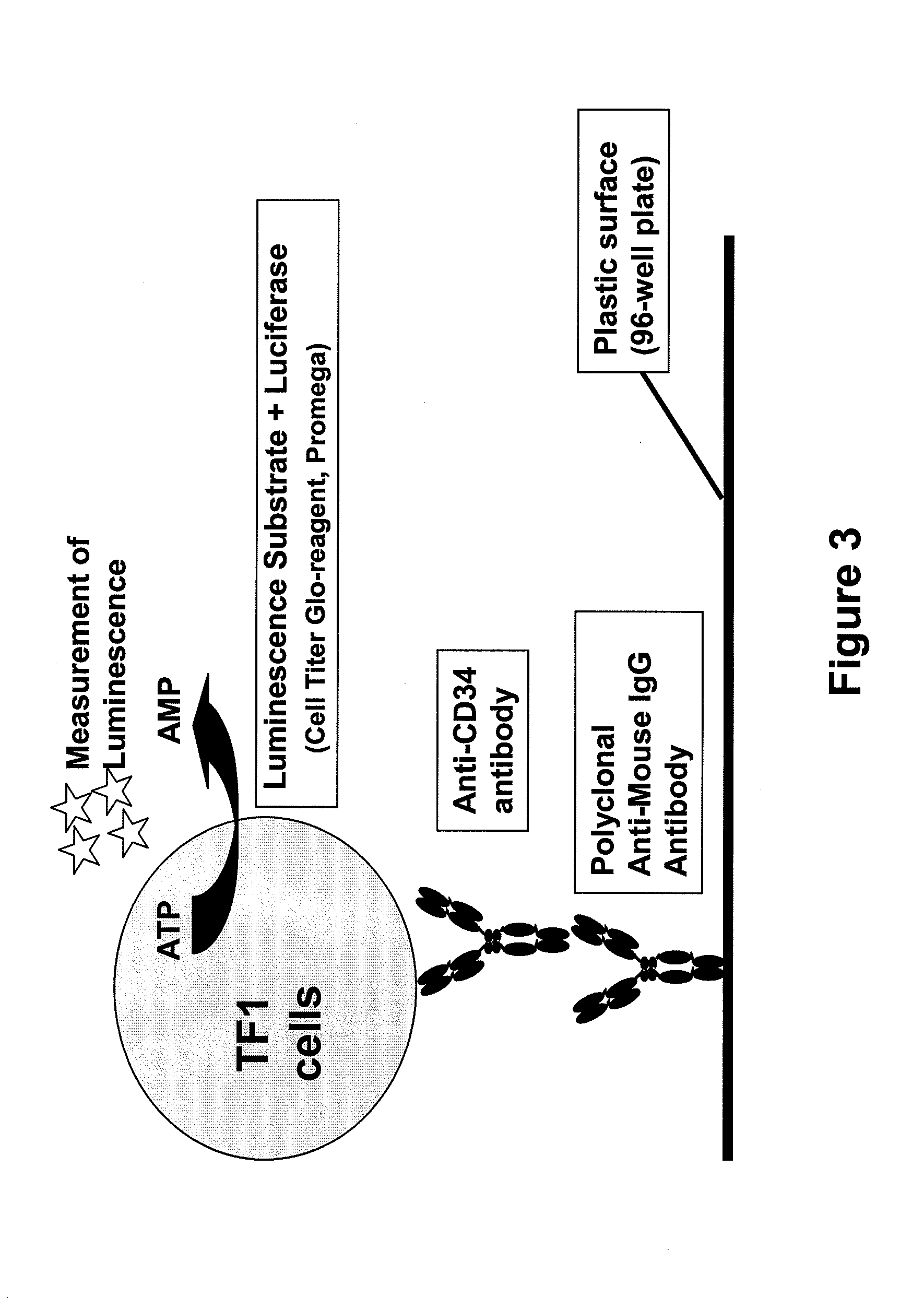
Procedure for the generation of a high producer cell line for the expression of a recombinant anti-CD34 antibody
The present invention relates to cell capture assay for the selection of a high producer cell line expressing anti-CD34 antibodies that recognize the CD34 membrane-protein in the cell membrane. The monoclonal antibody secreted by the hybridoma cell line 9C5 / 9069 binds to human CD34 and is used to isolate stem cells. The DNA sequences encoding for the antibody heavy and light chain have been identified, isolated from the hybridoma cells and cloned into appropriate expression vectors. After co-transfection of the heavy and light chain genes into HEK293T or in CHO cells either conditioned medium or purified antibody were assessed for binding to CD34 protein located in the cell membrane in different cell capture assays. The binding of the antibody to CD34-positive cells could be shown with these assays for several cell lines.
Owner:TAKEDA PHARMA CO LTD
Anti-p185erbb2 human-mouse chimeric antibody chab26, mammary gland-specific expression vector, transgenic fvb mouse and preparation method thereof
InactiveCN105646704BLow immunogenicityAvoid reactionImmunoglobulins against animals/humansVector-based foreign material introductionChimeric antibodyHigh activity
The invention discloses an anti-p185<erbB2> human-mouse chimeric antibody ChAb26. The anti-p185<erbB2> human-mouse chimeric antibody ChAb26 is mainly composed of a heavy chain gene H and a light chain gene L, and the heavy chain gene H and the light chain gene L are respectively connected to a mammary gland specific expression plasmid pBC1 in order to obtain p185<erbB2> human-mouse chimeric antibody ChAb26 mammary gland specific expression vectors pBC1-H and pBC1-L. The pBC1-H and pBC1-L are linearized, the linearized pBC1-H and pBC1-L are introduced to FVB mouse zygotes to produce a transgenic FVB mouse, and the anti-p185<erbB2> human-mouse chimeric antibody ChAb26 can be obtained through mammary gland specific expression with the transgenic FVB mouse as a mammary gland bioreactor. The antibody reduces the immunogenicity of the v monoclonal antibody, and a medicinal recombinant protein produced through the transgenic FVB mouse has the advantages of high output, low cost, high activity, easy large-scale formation, shortening of the manufacturing period of new medicines, and huge market potential.
Owner:GUANGXI MEDICAL UNIVERSITY
Procedure for the generation of a high producer cell line for the expression of a recombinant Anti-CD34 antibody
ActiveUS20090221003A1Animal cellsImmunoglobulins against cell receptors/antigens/surface-determinantsCell membraneCell Membrane Proteins
The present invention relates to cell capture assay for the selection of a high producer cell line expressing anti-CD34 antibodies that recognize the CD34 membrane-protein in the cell membrane. The monoclonal antibody secreted by the hybridoma cell line 9C5 / 9069 binds to human CD34 and is used to isolate stem cells. The DNA sequences encoding for the antibody heavy and light chain have been identified, isolated from the hybridoma cells and cloned into appropriate expression vectors. After co-transfection of the heavy and light chain genes into HEK293T or in CHO cells either conditioned medium or purified antibody were assessed for binding to CD34 protein located in the cell membrane in different cell capture assays. The binding of the antibody to CD34-positive cells could be shown with these assays for several cell lines.
Owner:TAKEDA PHARMA CO LTD
Antibody production
ActiveUS20130330771A1Increase probabilityImmunoglobulinsImmunological disordersImmunoglobulin heavy chainHeterologous
A transgenic non-human mammal containing a heterologous lambda light chain gene locus, and / or a heterologous kappa light chain gene locus, and / or a heterologous heavy chain gene locus, each of which can re-arrange so that immunoglobulin heavy and light chain genes are formed and expressed in B-cells following antigen challenge.
Owner:ERASMUS UNIV MEDICAL CENT ROTTERDAM ERASMUS MC