Patents
Literature
55results about How to "High strength material" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
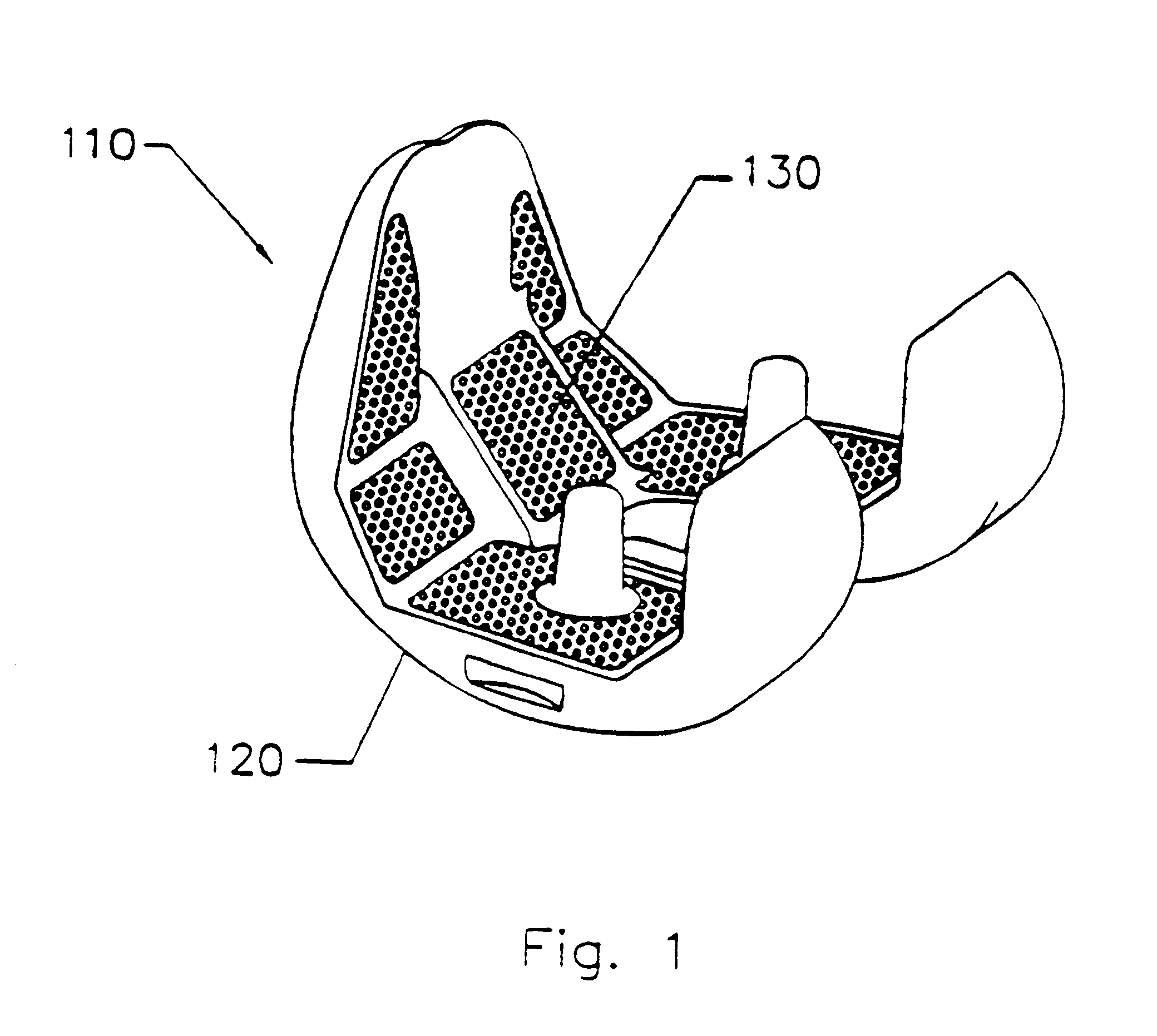
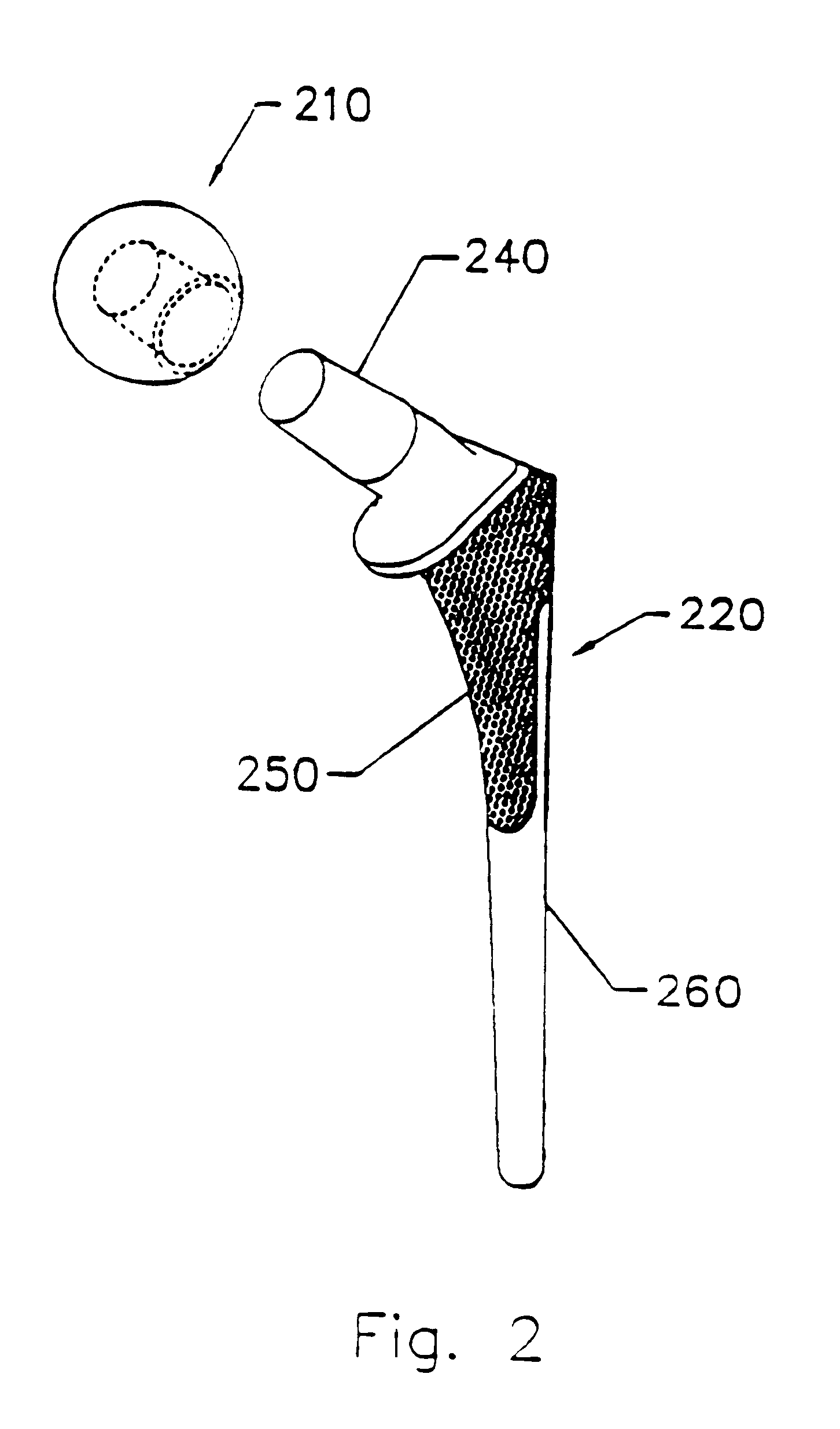
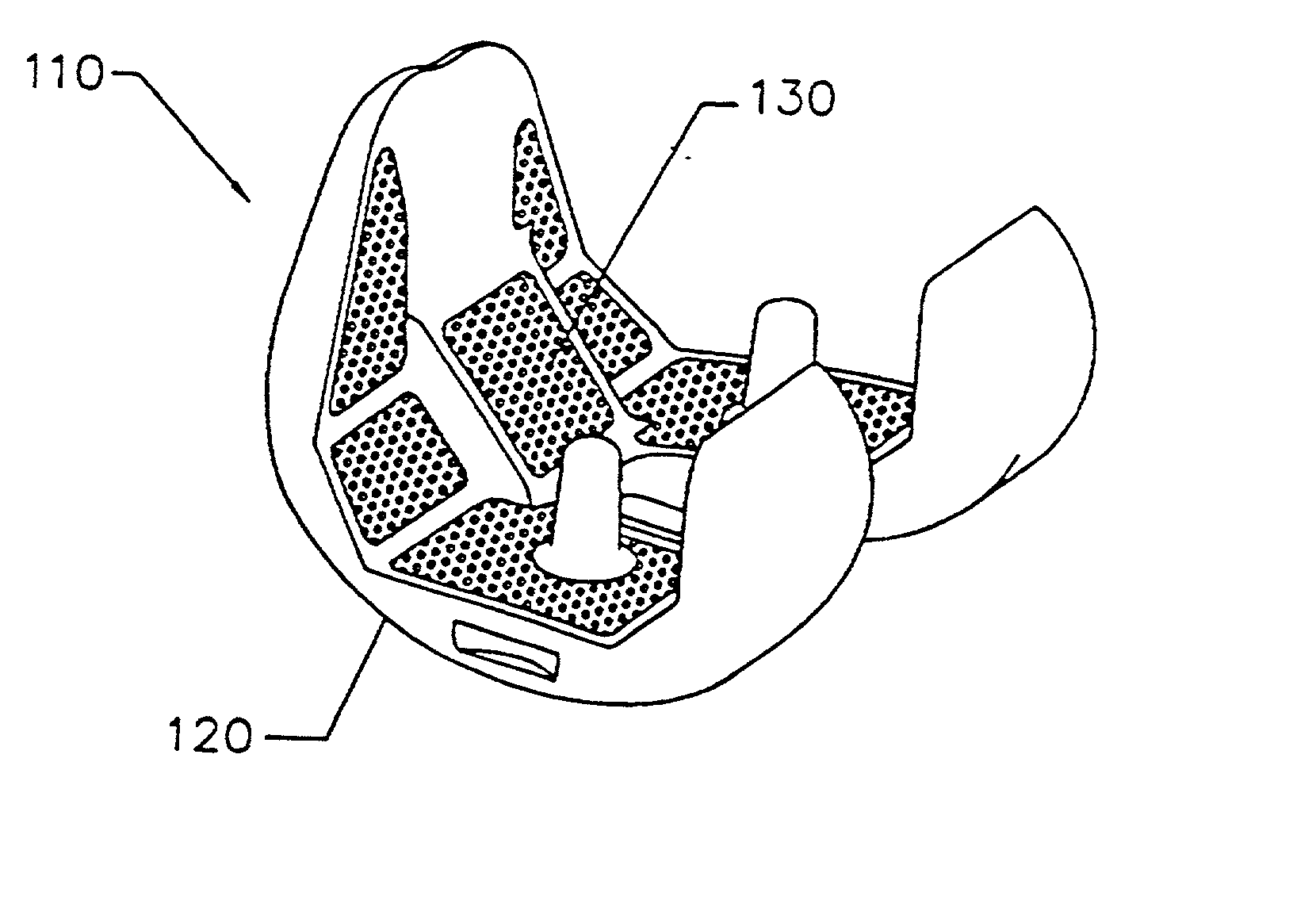
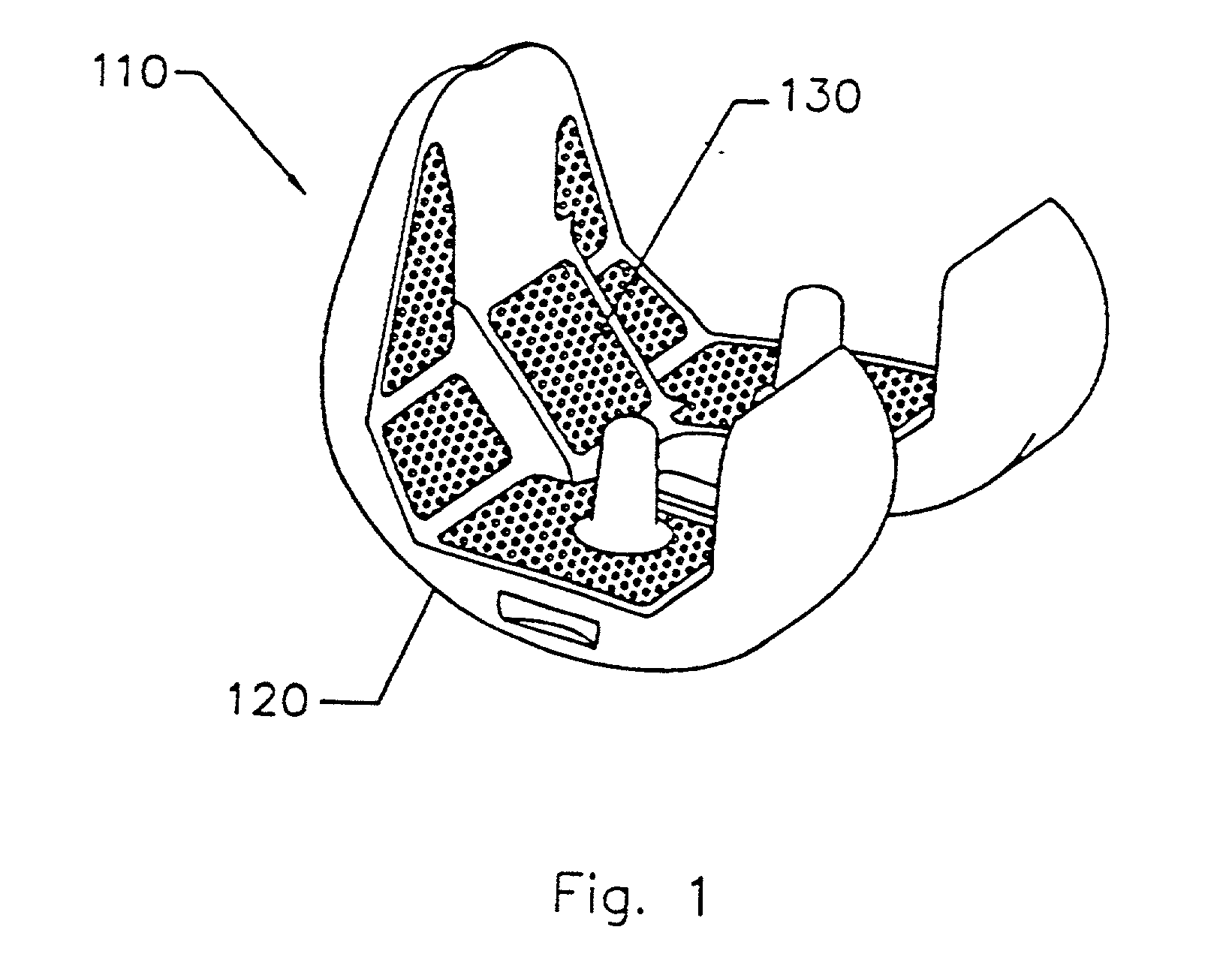
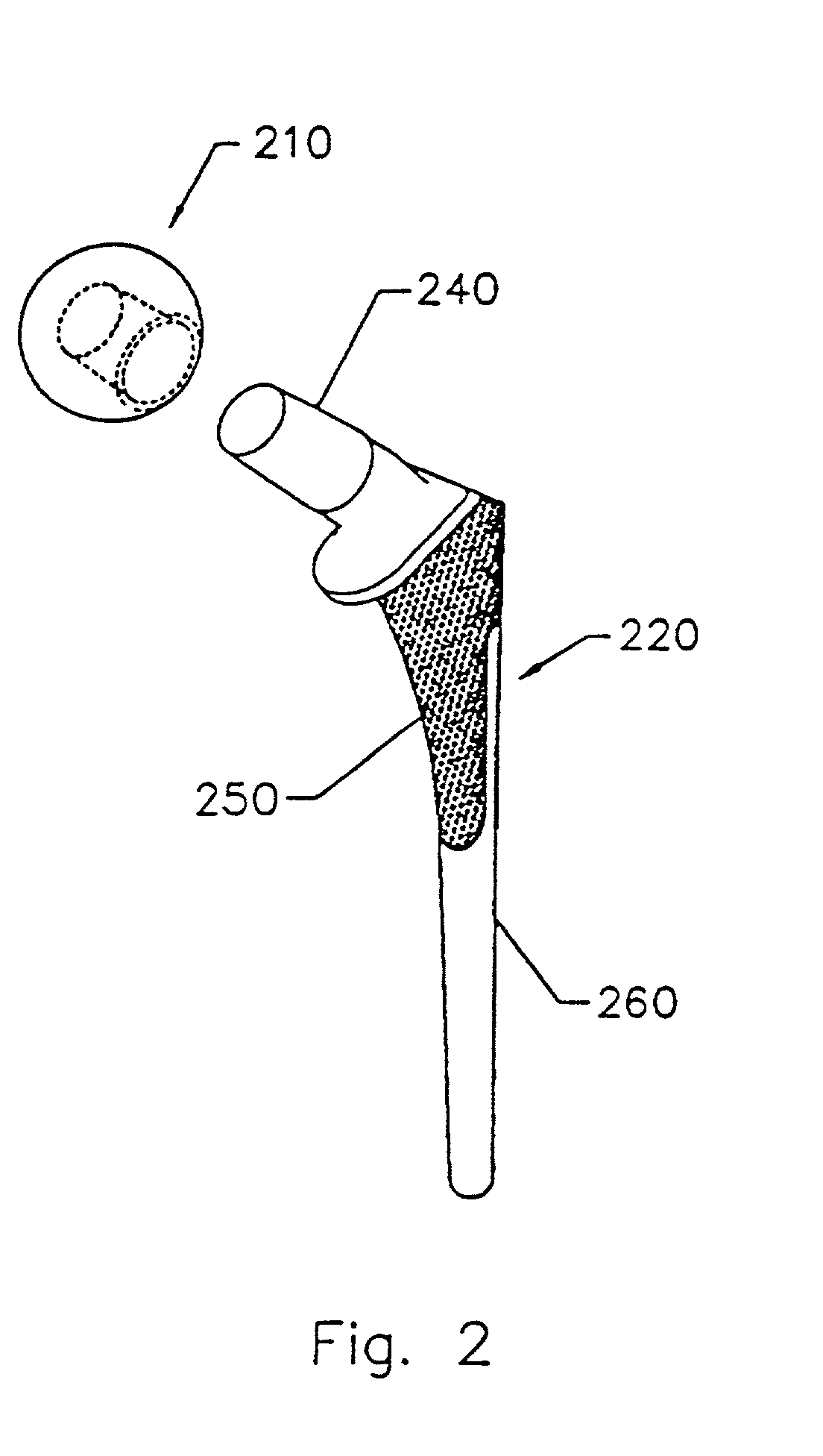
Implant with composite coating
InactiveUS6261322B1High strengthCost effectiveImpression capsBone implantBiocompatible coatingBiocompatibility Testing
Systems and methods are described for implants with composite coatings to promote tissue in-growth and / or on-growth. An implant includes: a substrate; a structured surface formed on at least a portion of the substrate; and a biocompatible coating deposited on at least a fraction of the structured surface. The systems and methods provide advantages in that the implant has good biocompatibility while the biocompatible coating has good strength.
Owner:SHALBY ADVANCED TECH INC
Implant with composite coating
InactiveUS20020016635A1High strengthCost effectiveImpression capsBone implantBiocompatible coatingBiocompatibility Testing
Systems and methods are described for implants with composite coatings to promote tissue in-growth and / or on-growth. An implant includes: a substrate; a structured surface formed on at least a portion of the substrate; and a biocompatible coating deposited on at least a fraction of the structured surface. The systems and methods provide advantages in that the implant has good biocompatibility while the biocompatible coating has good strength.
Owner:CONSENSUS ORTHOPEDICS
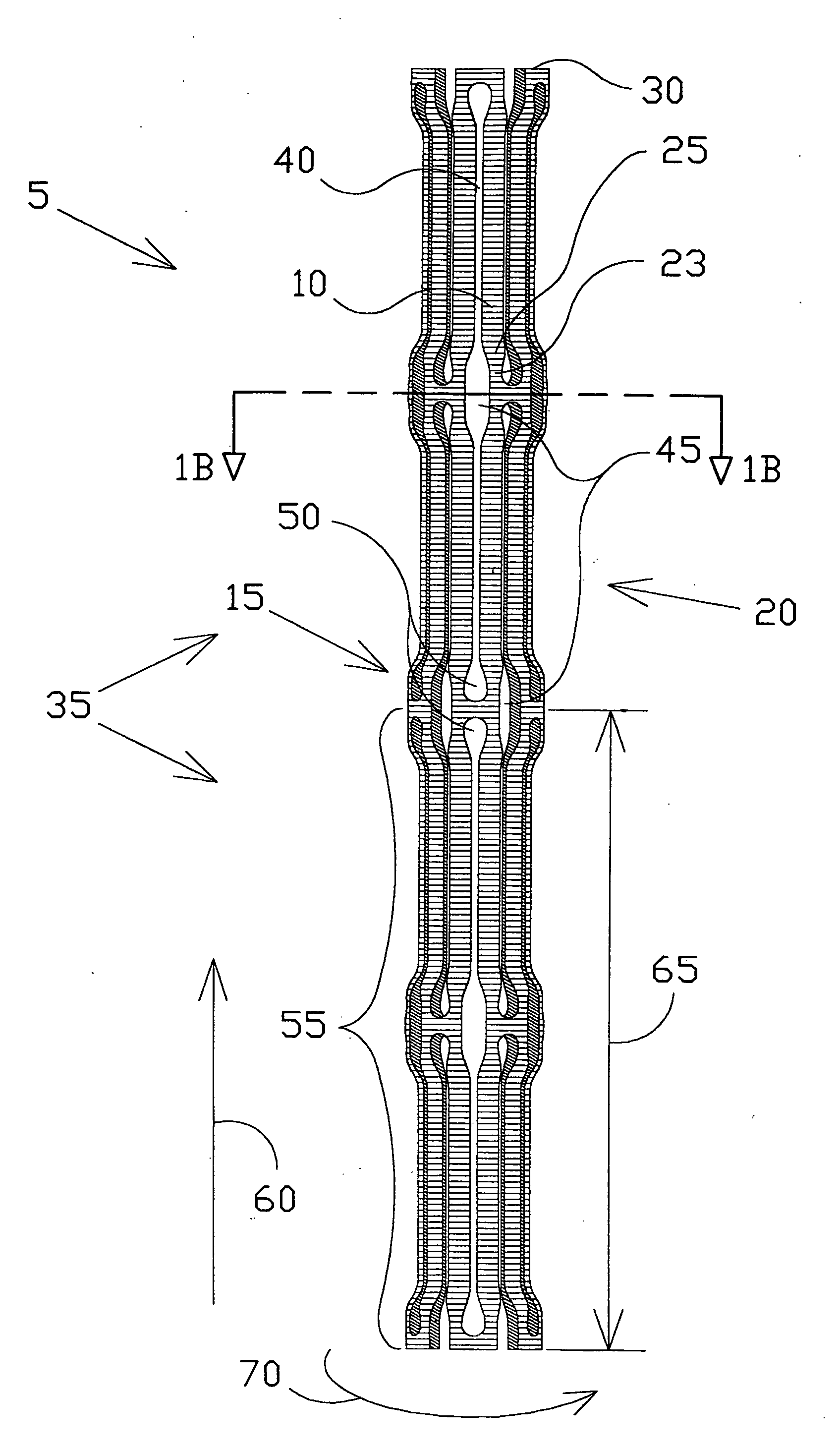
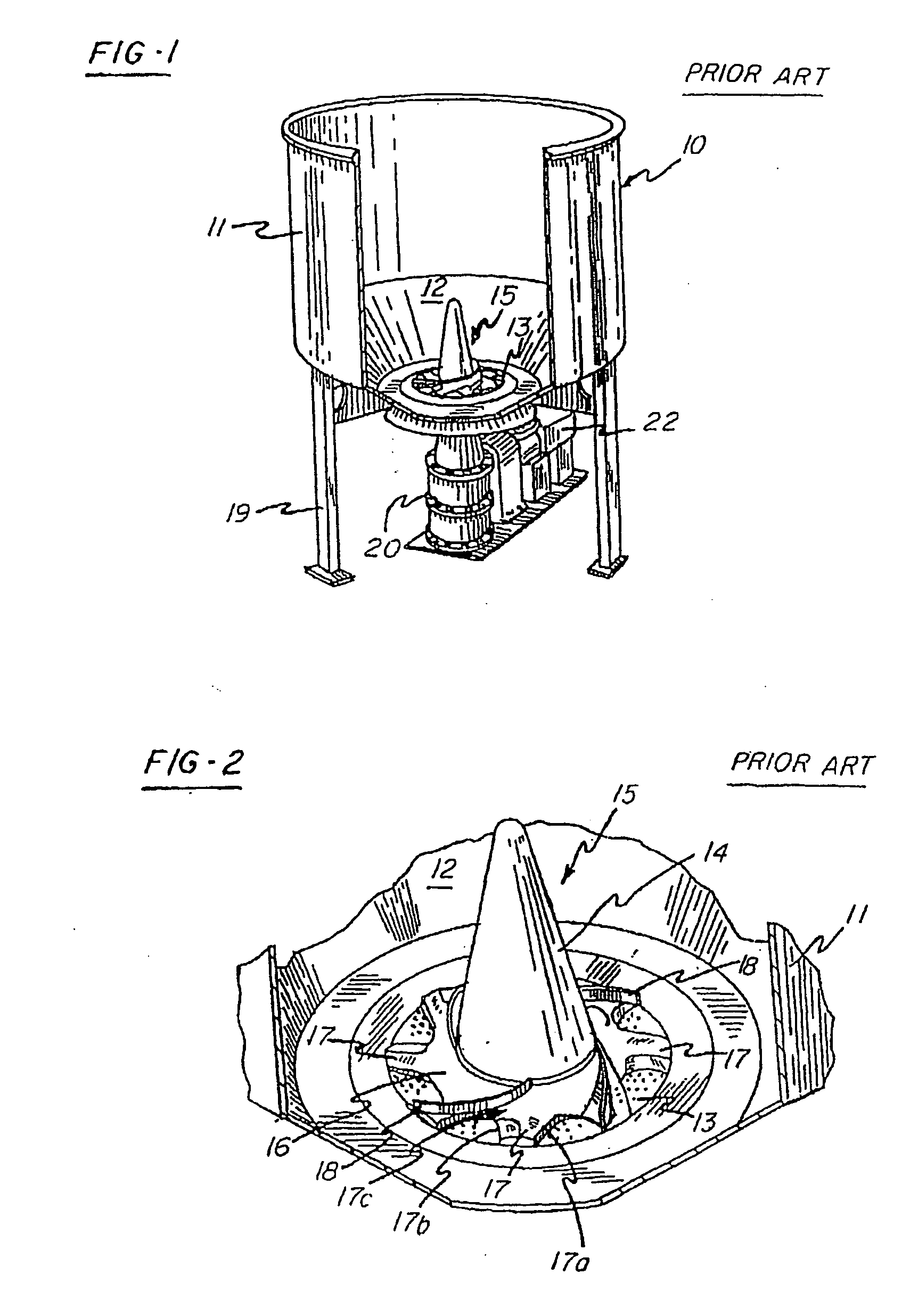
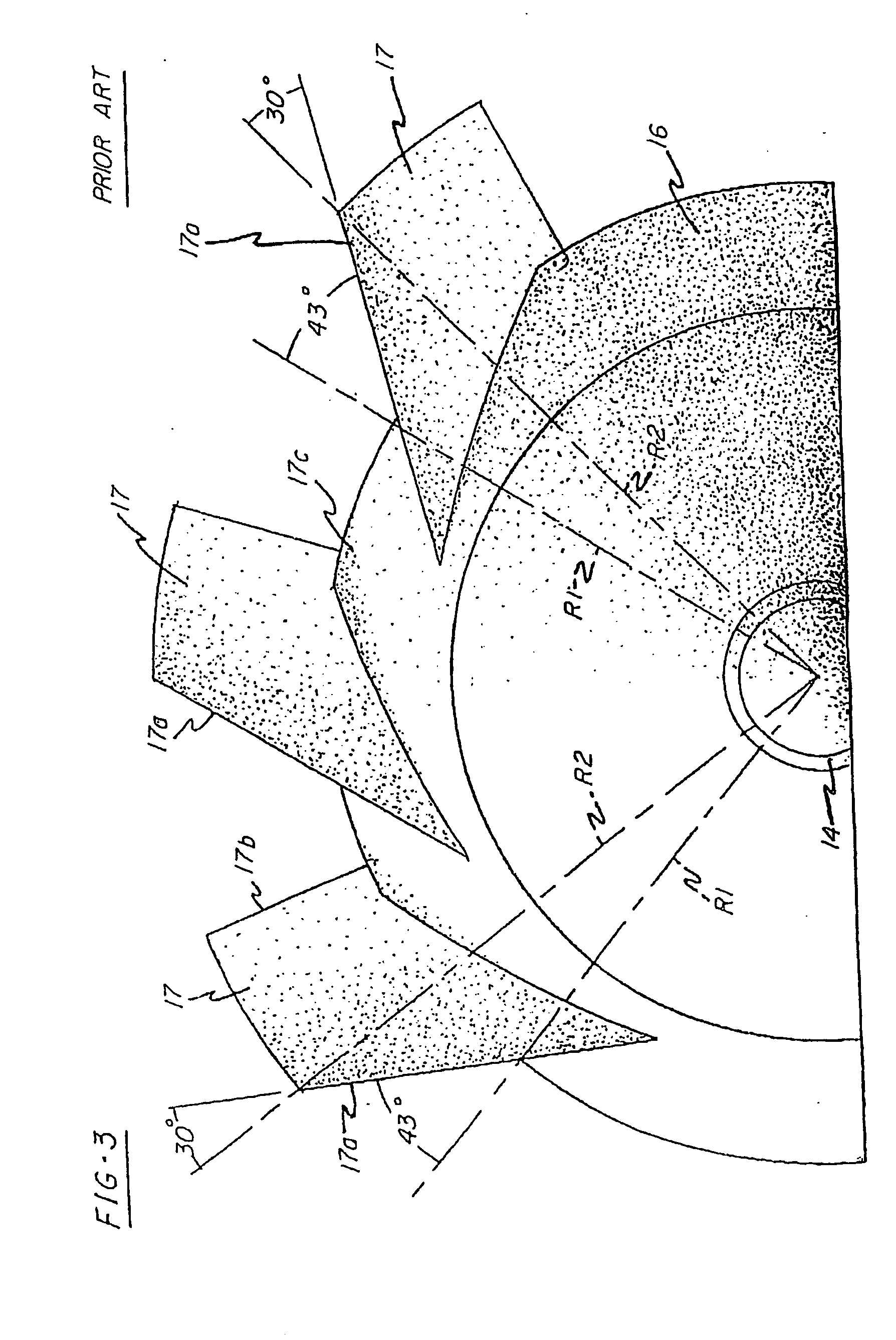
Intravascular hinge stent
A balloon or self expandable endoprosthesis formed of a single or multiple joined stent sections. Each section has a node and strut structure extending throughout in order to uncouple expansion forces of the stent to hold a blood vessel outward from crush forces that resist the formation of an oval shape during crush deformation. The hinge can bend in the direction of a uniformly curved surface of the stent but not in the radial direction. The strut can bend in the radial direction but not in the uniformly curved surface of the stent. The widths, lengths, and radial dimensions of the hinges and struts provide a balloon-expandable hinge stent that is non-crushable. For a self-expanding stent the hinge and strut dimensions provide expansion forces that are controlled independently from crush forces. Expansion properties are determined by the hinge dimensions and crush properties are determined independently by strut dimensions.
Owner:DRASLER WILLIAM JOSEPH +1
B-shaped beam with integrally-formed rib in face
InactiveUS20080093867A1Reduce “ offset ”Weight increaseBumpersMetal rolling arrangementsShaped beamEngineering
Owner:SHAPE CORP
Wood type golf club head
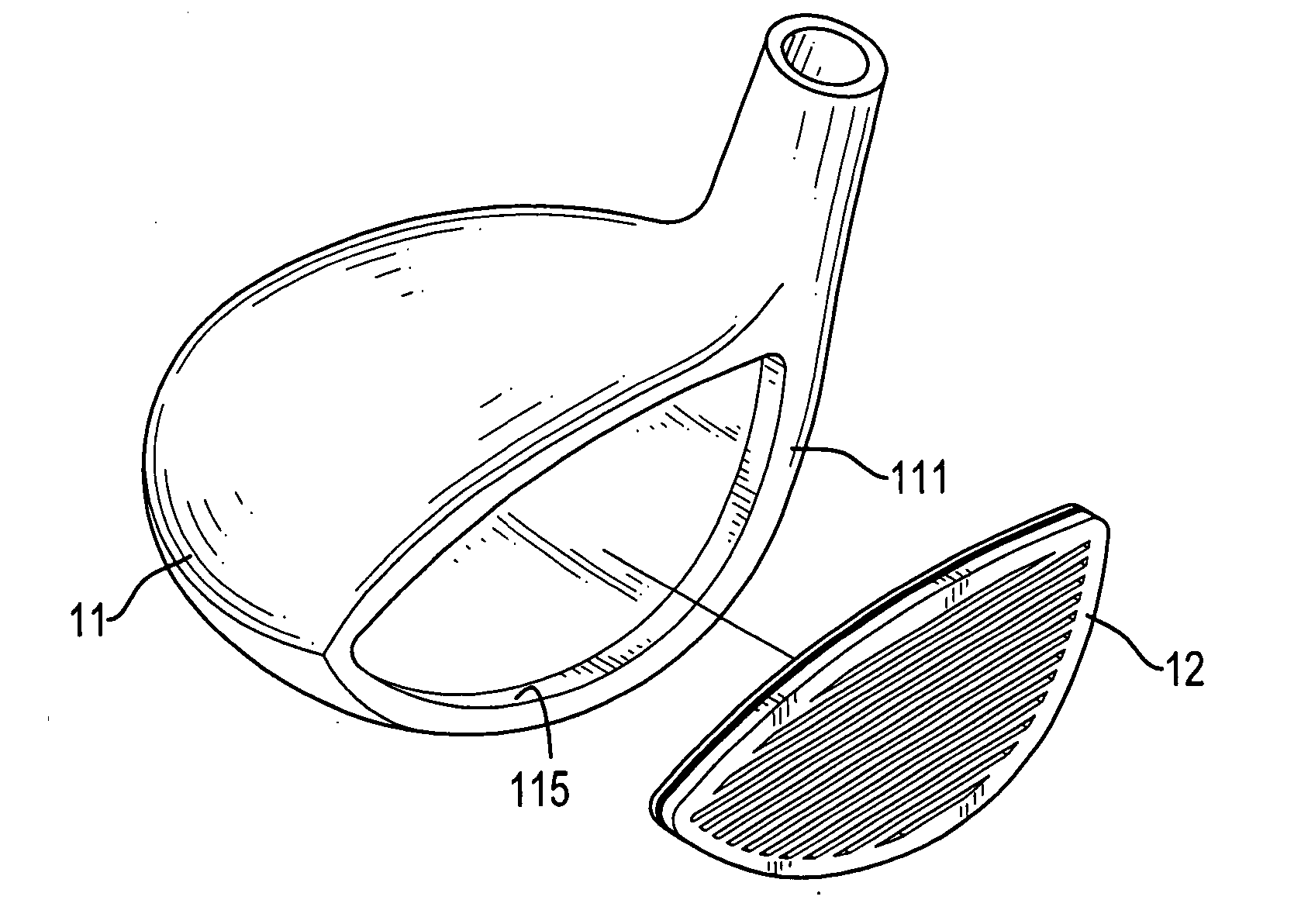
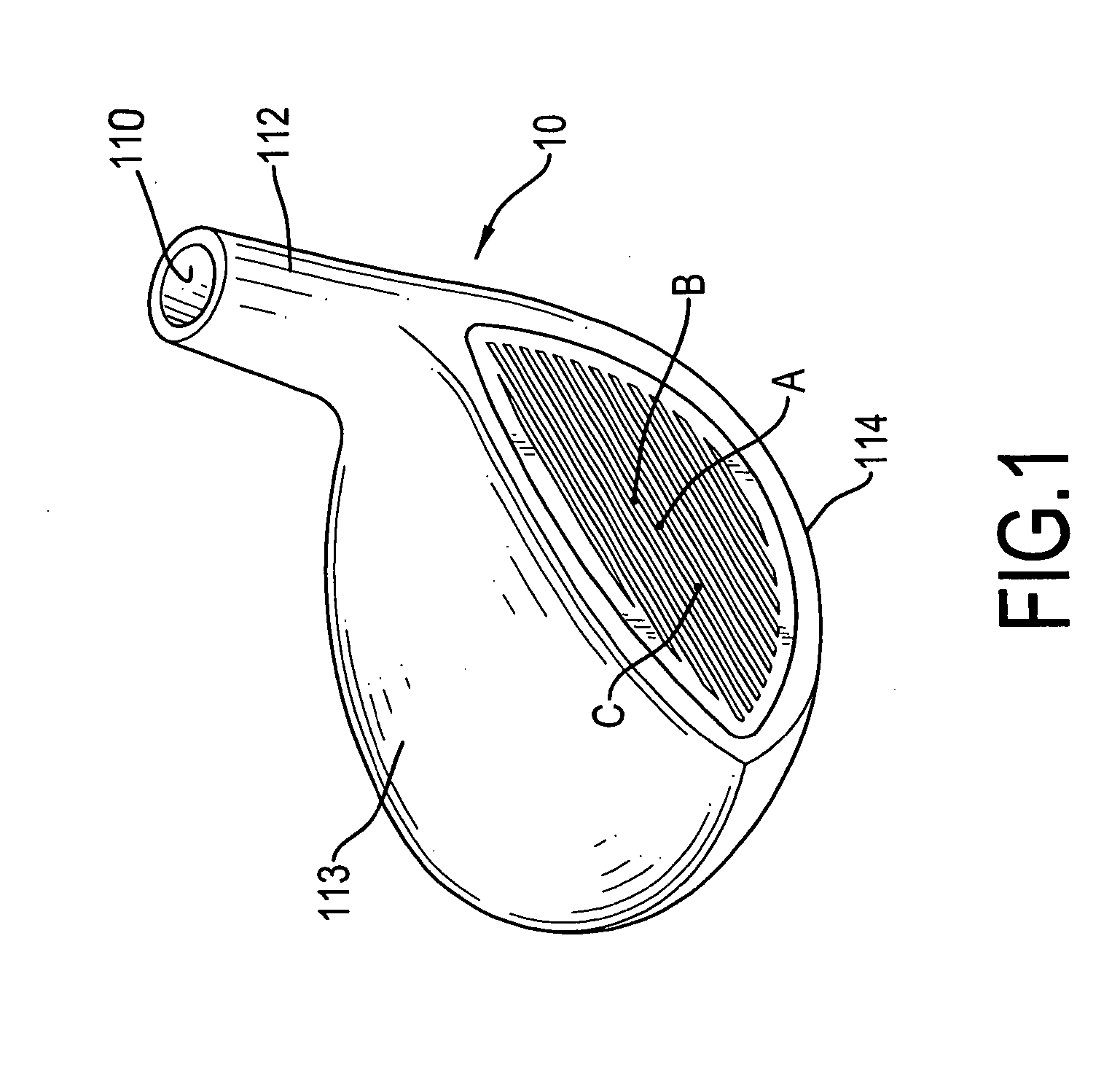
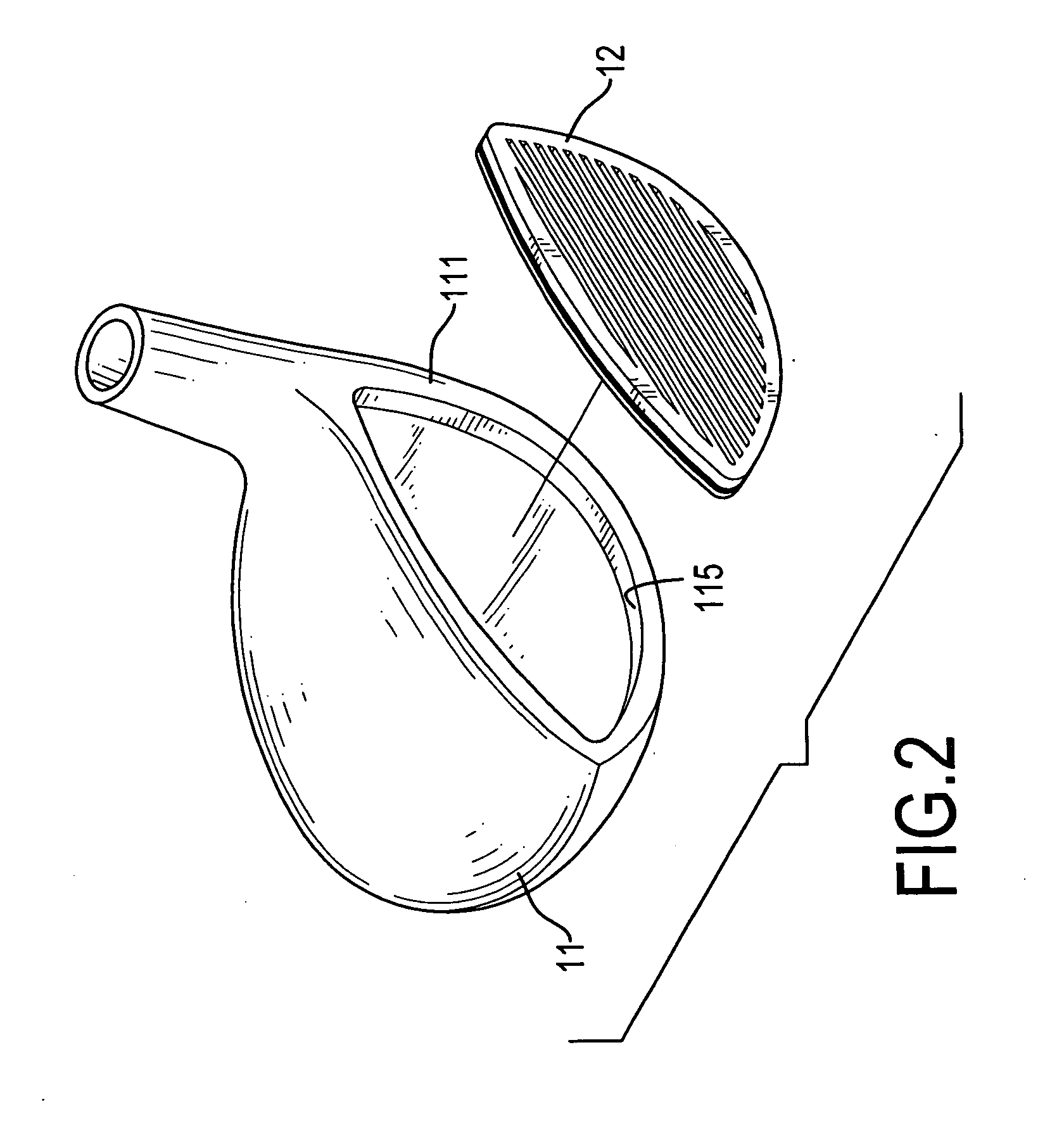
A golf club head for a metal wood includes a body and a faceplate. The body has a front face having a recess. The faceplate is mounted in the recess and includes on outside plate and an inside faceplate. The outside plate has a thickness that is 1.0 to 2.0 millimeters and is made of high strength material. The inside plate has a thickness that is 0.5 to 1.8 millimeters and is made of high strength material. The inside plate, which is hollow or meshed, is connected to the outside plate. Therefore, the COR of the faceplate will be more consistent, and the faceplate can be fabricated with a large area.
Owner:O TA PRECISION IND
Synthetic Nano-Sized Crystalline Calcium Phosphate and Method of Production
ActiveUS20080220233A1Easy to depositMaximize adherence to surfaceMaterial nanotechnologyNanostructure manufactureLiquid crystallineSolvent
Synthetic nano-sized crystalline calcium phosphate, particularly hydroxyapatite, having a specific surface area in the range of 150 m2 / g to 300 m2 / g, is described. The nano-sized crystalline calcium phosphate may be in the form of a powder or in the form of a coating on a surface. A method of producing a nano-sized crystalline calcium phosphate powder or coating is also described. The method comprises formation of a liquid crystalline phase in a water solution of calcium, phosphor and a surfactant, placing the phase in an ammonia atmosphere so that nano-sized crystals are formed, followed by either removal of the surfactant with a solvent and recovering the nano-sized crystals to obtain the powder, or diluting the ammonia-treated liquid crystalline phase with a hydrophobic organic solvent to create a microemulsion of the nano-sized crystals in water, dipping an oxide layer-coated surface of an object into the microemulsion, or alternatively saving the step of ammonia treatment of the liquid crystalline phase until after the dipping of the surface of an object into the microemulsion, followed by removal of the organic solvent and the surfactant from the surface to obtain the coating.
Owner:PROMIMIC
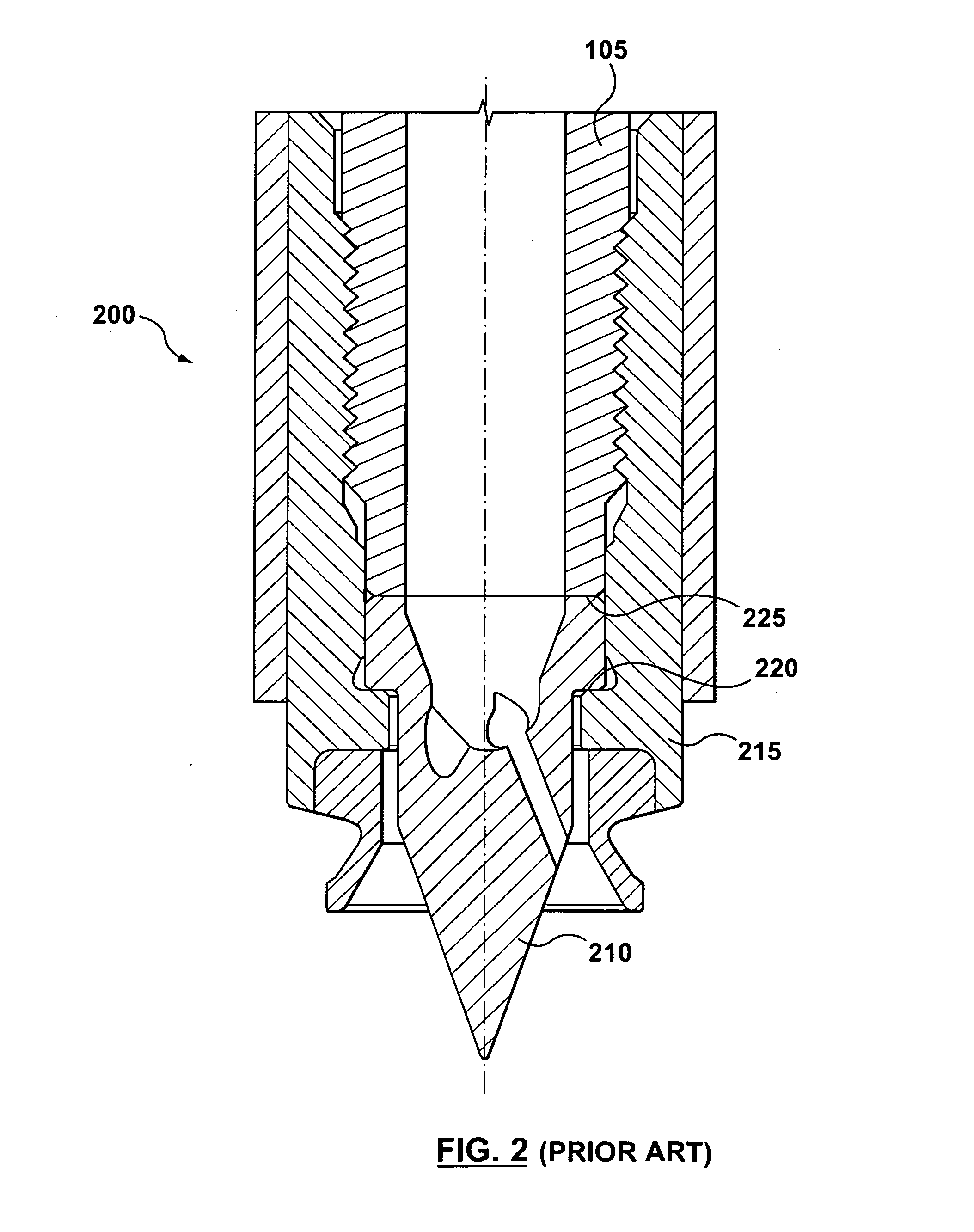
Hot Runner Nozzle System
InactiveUS20090220632A1Increase torquePrevent resin leakageConfectionerySweetmeatsHot runnerEngineering
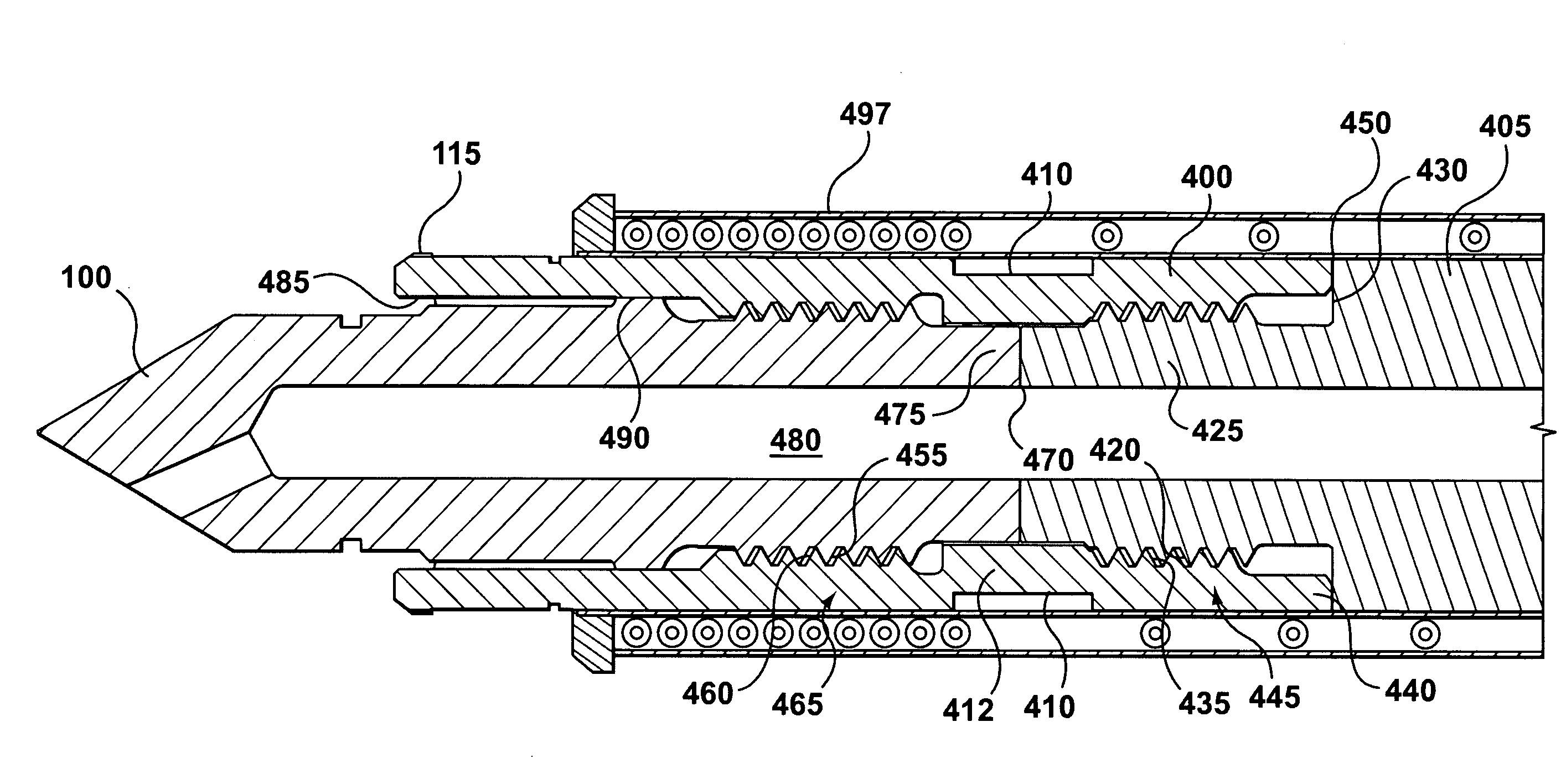
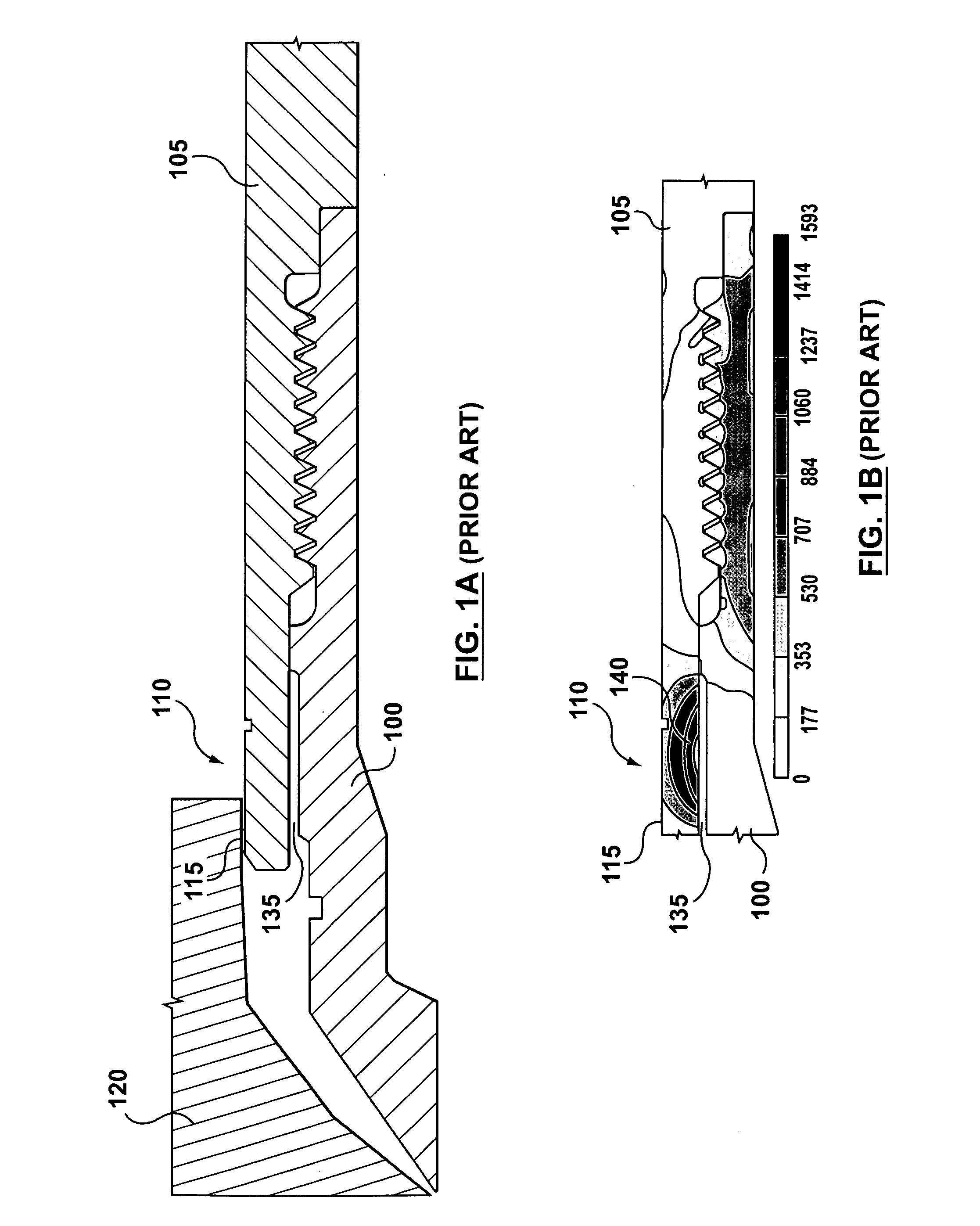
A hot runner nozzle system (500) comprising a nozzle tip (100) adjacent to a nozzle (405) in series, and a retainer (400) adjoining both the nozzle tip (100) and the nozzle (405) in parallel, the retainer (400) having a first retainer thread (435), for connection to a nozzle thread (420) to form a first seal (450) between the nozzle end (440) of the retainer (400) and a nozzle shoulder (430), a second retainer thread (460), for connection to a tip thread (455) to form a second seal (470) between the inlet end (475) of the nozzle tip (100) and the gate end (425) of the nozzle (405), a seal ring (115) for creating a gate seal (496) with a gate insert (120), and a plurality of flats (410) thereon to facilitate torquing of the retainer (400) to the nozzle (405), the torque value applied to the retainer (400) being about two to four times of that applied to the nozzle tip (100) to facilitate removal of the nozzle tip (100) independent of the retainer (400).
Owner:HUSKY INJECTION MOLDING SYST LTD
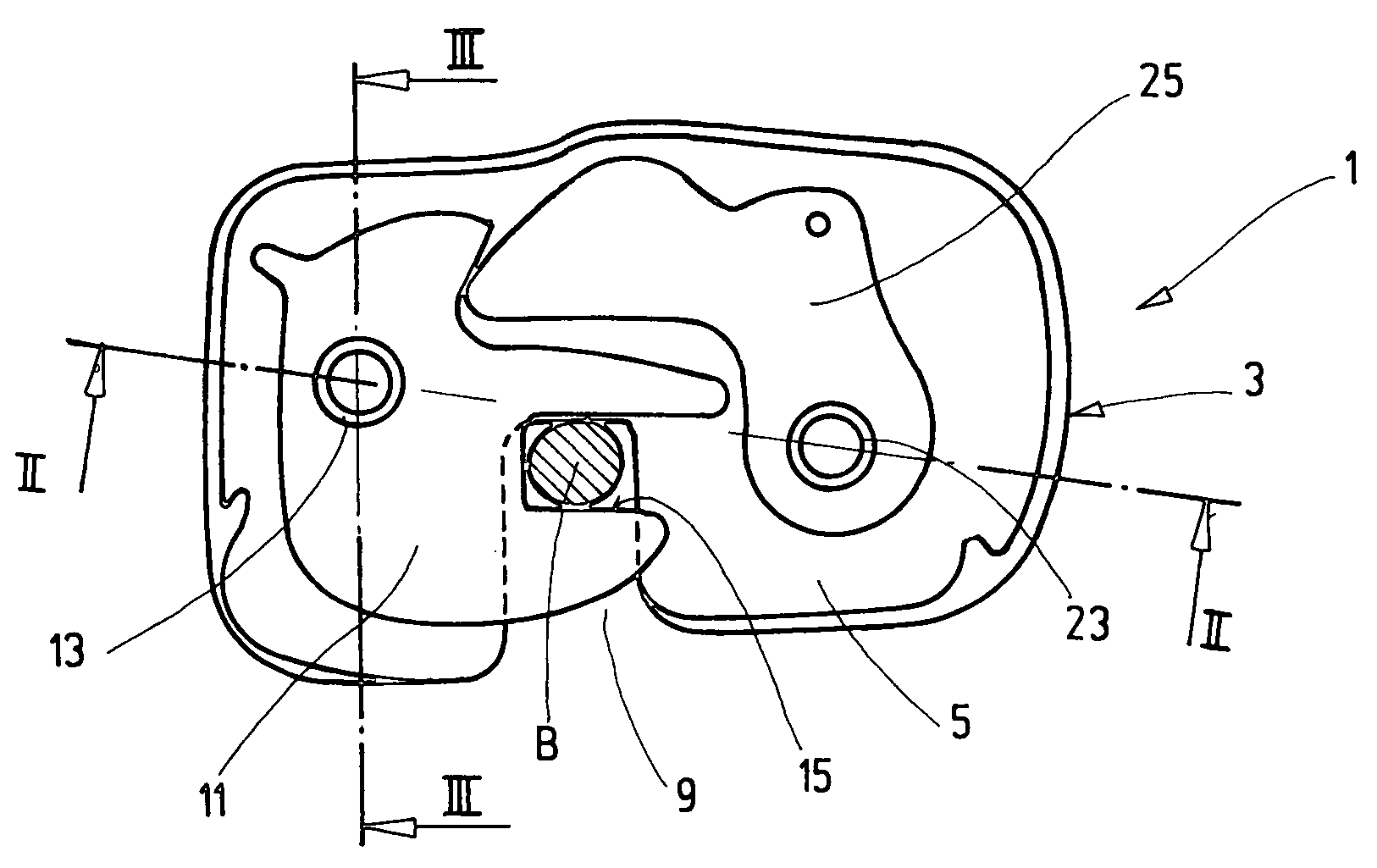
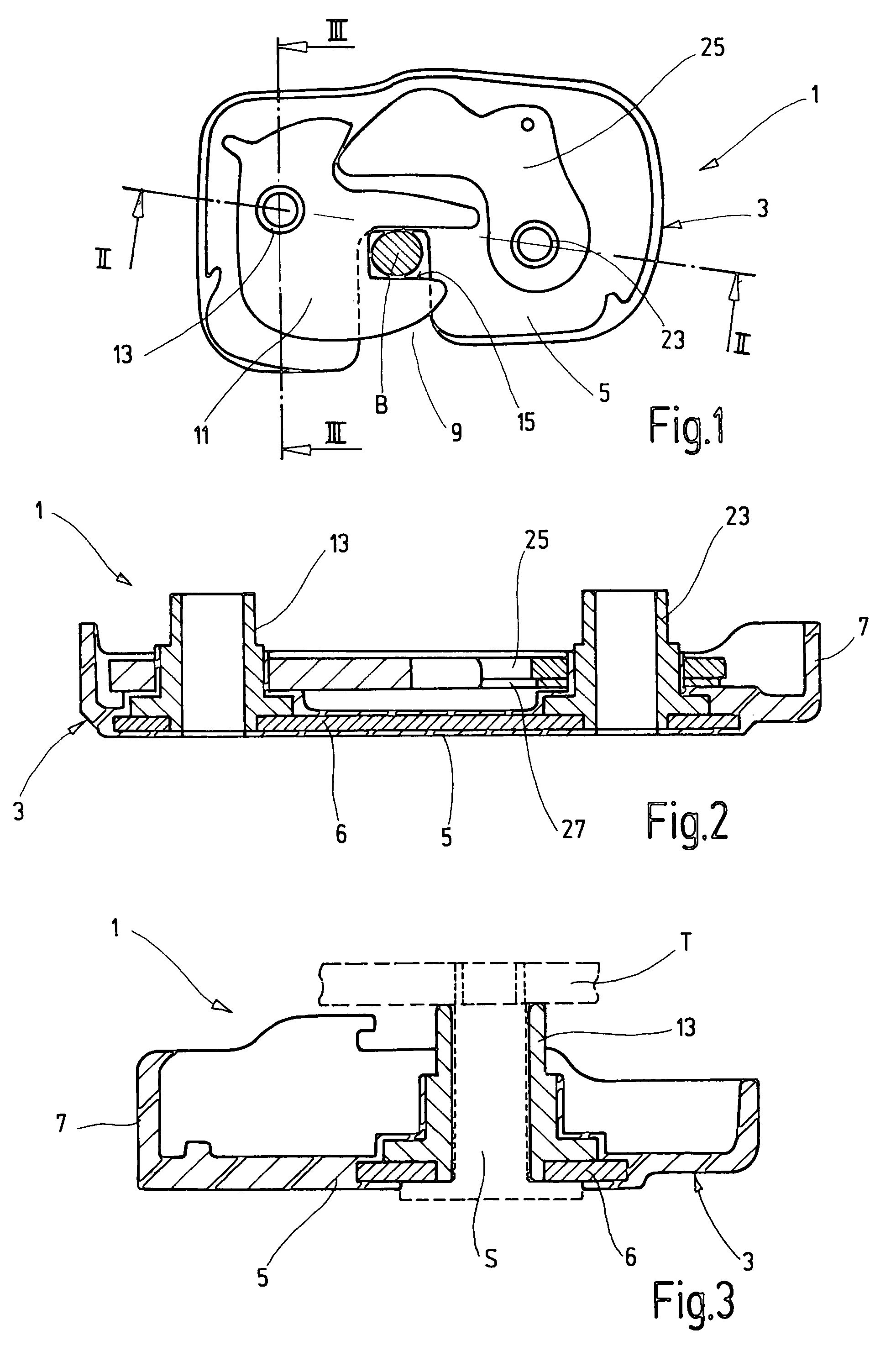
Locking device for a vehicle seat
ActiveUS7455336B2Reduce weightHigh strength materialOperating chairsFurniture partsEngineeringSoft materials
In the case of a locking device (1), in particular for a vehicle seat, with a housing (3); a latch (11) which is arranged in the housing (3), is pivotably mounted on a first bearing element (13) and is intended for locking with a mating element (B); and at least one securing element (25, 27) which is likewise arranged in the housing (3), is pivotably mounted on a second bearing element (23) and secures a locked state by interaction with the latch (11), the housing (3) has at least one insert (6) of a higher strength material, and at least one of the bearing elements (13, 23) and the insert (6) are joined together and are at least partially encased by a softer material, forming the housing (3).
Owner:KEIPER SEATING MECHANISMS CO LTD
High security opening apparatus for hermetically sealed containers
InactiveUS6902453B2Increased durabilityReduce the possibility of damageLife-raftsFlexible coversMechanical engineeringHigh security
Owner:SWITLIK PARACHUTE
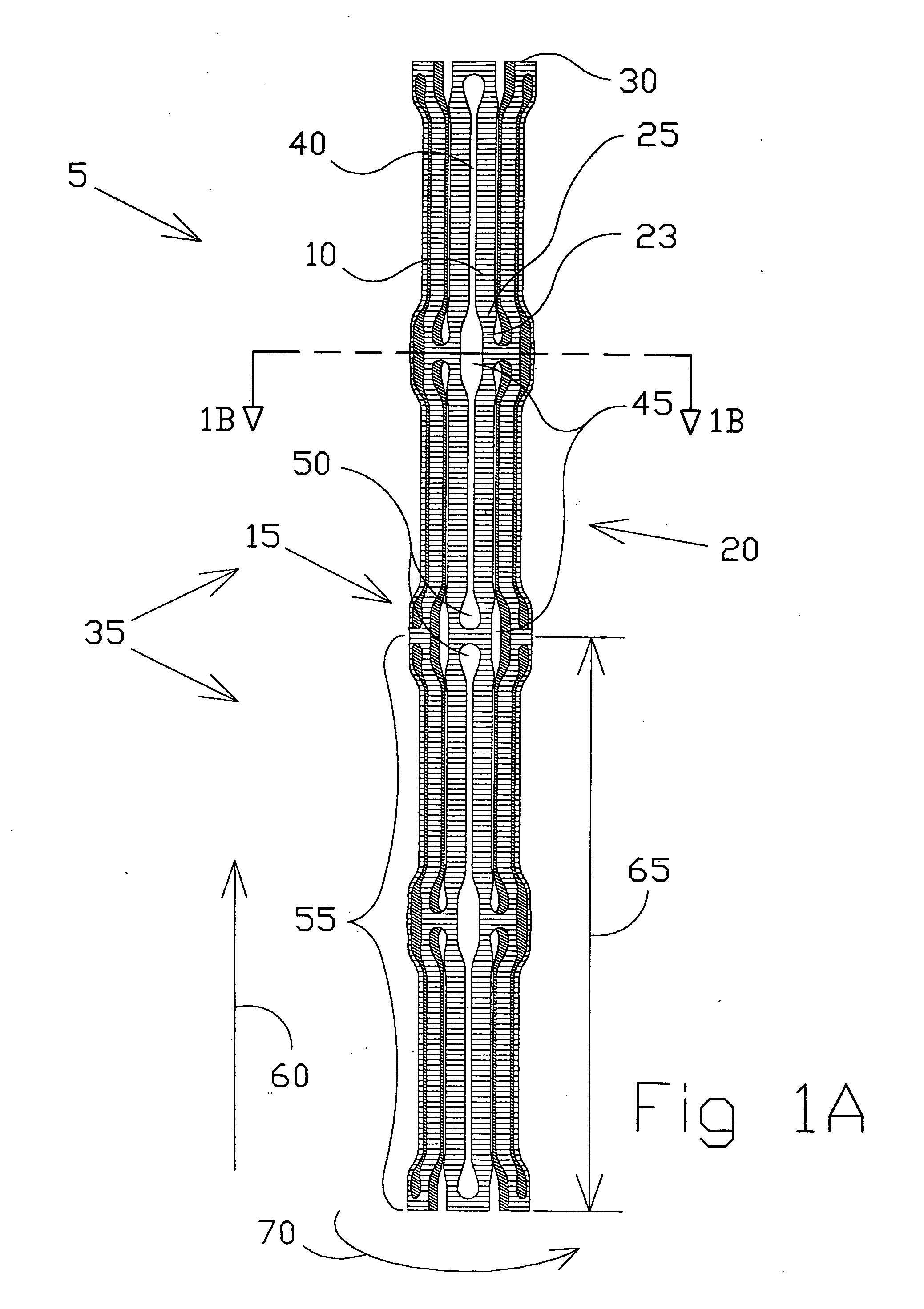
Intravascular hinge stent
A balloon or self expandable endoprosthesis formed of a single or multiple joined stent sections. Each section has a node and strut structure extending throughout in order to uncouple expansion forces of the stent to hold a blood vessel outward from crush forces that resist the formation of an oval shape during a crush deformation. The hinge can bend in the direction of a uniformly curved surface of the stent but not in the radial direction. The strut can bend in the radial direction but not in the uniformly curved surface of the stent. The widths, lengths, and radial dimensions of the hinges and struts provide a balloon-expandable hinge stent that is non-crushable. For a self-expandable stent the hinge and strut dimensions provide expansion forces that are controlled independently from crush forces. Expansion properties are determined by the hinge dimensions and crush properties are determined independently by the strut dimensions.
Owner:DRASLER WILLIAM J +1
Abradable and/or abrasive coating and brush seal configuration
InactiveUS20060249911A1Improve rendering capabilitiesIncrease temperatureEngine sealsLeakage preventionBristleEngineering
An abradable coating and / or an abrasive coating applied to the rotor of a steam or gas turbine allows the temperature and pressure drop capabilities of conventional brush seal assemblies to be increased. The pressure drop capability of brush seals is closely related to fence height, i.e., the distance between the rotor and the bottom edge of the backing plate supporting the brush seal wire bristles. Application of the abradable or abrasive coating allows the fence height to be reduced and the pressure capability of a brush seal assembly increased commensurately because the risk of the backing plate rubbing against the surface of the rotor, so as to cause damage to the rotor, is reduced. In addition, the backing plate can be made from a higher strength material that can withstand higher temperatures because any contact between the backing plate and the rotor results in no damage to the rotor, but rather a gradual reduction of the backing plate as it wears.
Owner:GENERAL ELECTRIC CO
Compact and robust load and moment sensor
ActiveUS20110197682A1Extended functional lifeEasy to replaceForce measurementApparatus for force/torque/work measurementElectrical resistance and conductanceLoad rejection
The present invention relates to a load and moment sensor for a prosthetic device detecting load in a single direction and moment in a single plane. The load and moment sensor includes a sensing element, and a plurality of strain gauges placed in specific locations of the sensing element. The plurality of strain gauges is part of a plurality of resistor circuits such as Wheatstone bridges. While the strain gauges can be located on a single sensing element, some resistive elements of the Wheatstone bridges can be located elsewhere on the prosthetic device. The combination of the location of the strain gauges and the use of the Wheatstone bridges allows for good side load rejection which is load and moment not in the single direction or the single plane.
Owner:PROTEOR USA LLC
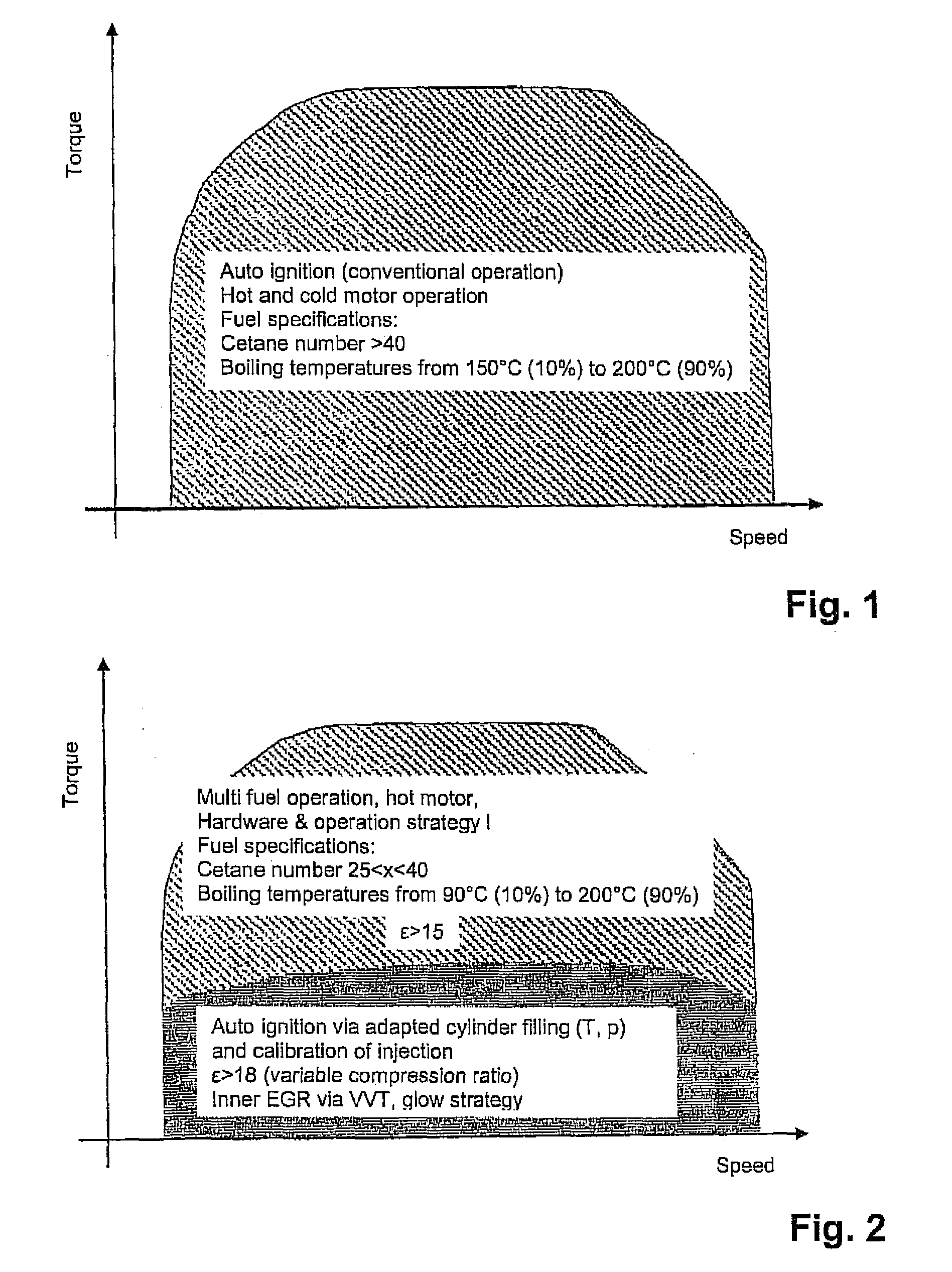
Multi-fuel diesel internal combustion engine
InactiveUS20110120416A1Reliable ignitionIncrease the compression ratioValve arrangementsElectrical controlCombustion chamberLow load
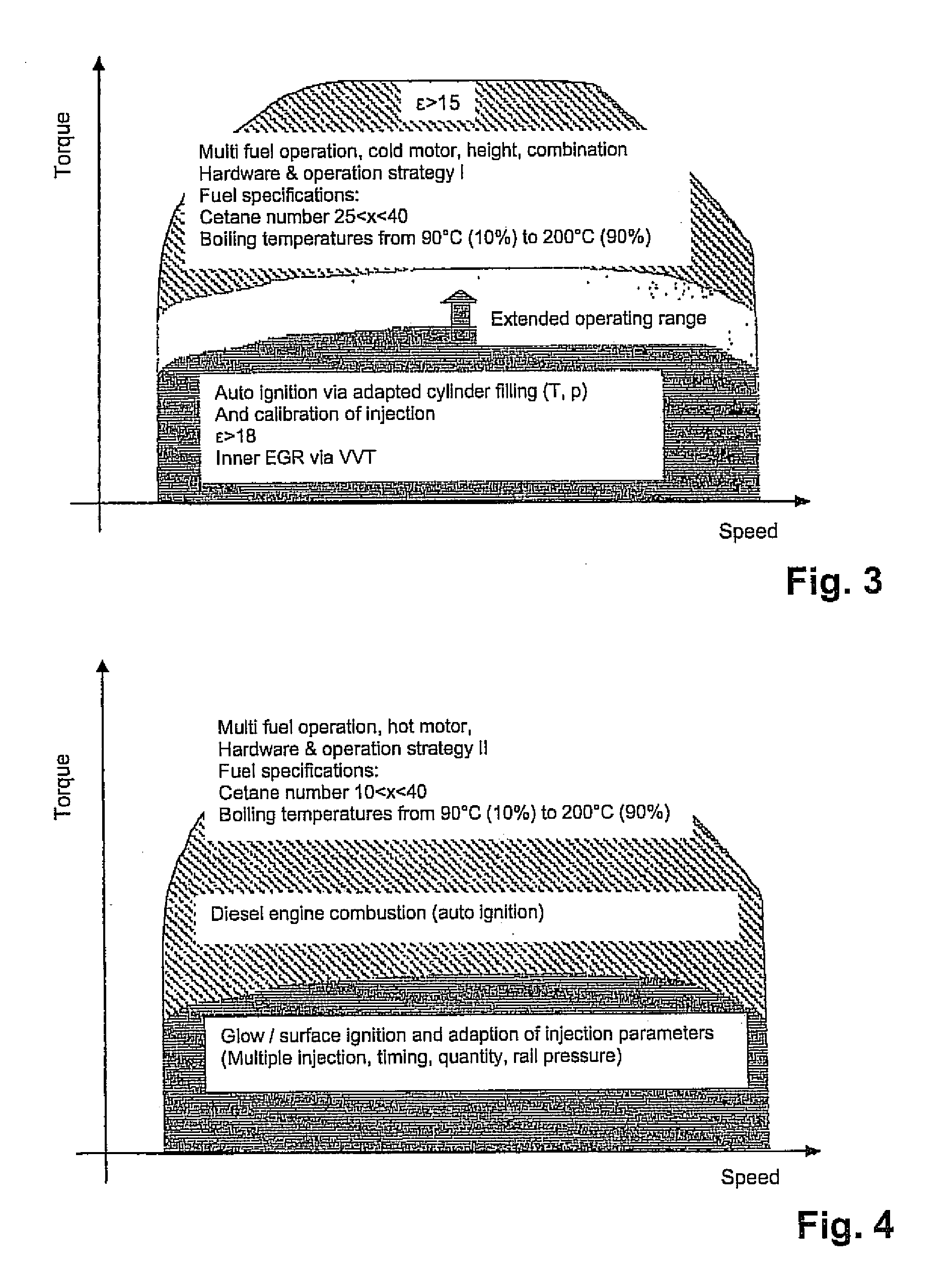
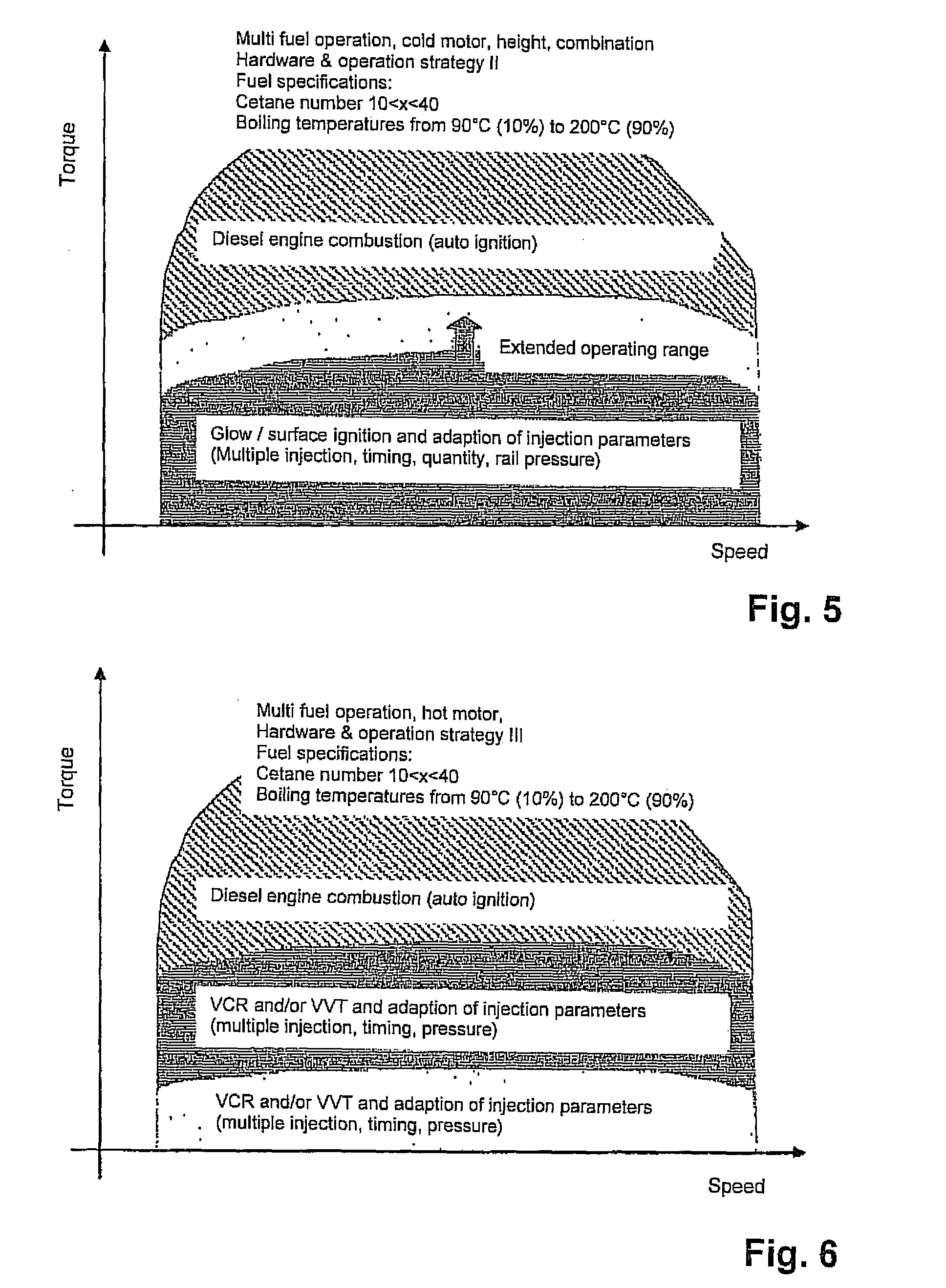
A diesel internal combustion engine that is operated advantageously at least predominantly according to the diesel principle, with a multi-fuel adaptation unit that reacts to various fuels of different mixtures and adapts an operating mode of the internal combustion engine to these fuels, wherein the multi-fuel adaptation unit comprises at least the following elements:a fuel characterization,a combustion-chamber filling that can be adapted as a function of a characteristic fuel,external ignition that can be allocated to each cylinder of the internal combustion engine,at least one control device that implements a first control strategy in which, in a lower load range, external ignition is performed, and a second control strategy in which automatic ignition is performed in a load range that is higher relative to the lower load range,a combustion-point control that can be adapted automatically to the characterized fuel, anda conversion-rate control that can be adapted automatically to the characterized fuel.
Owner:FEV
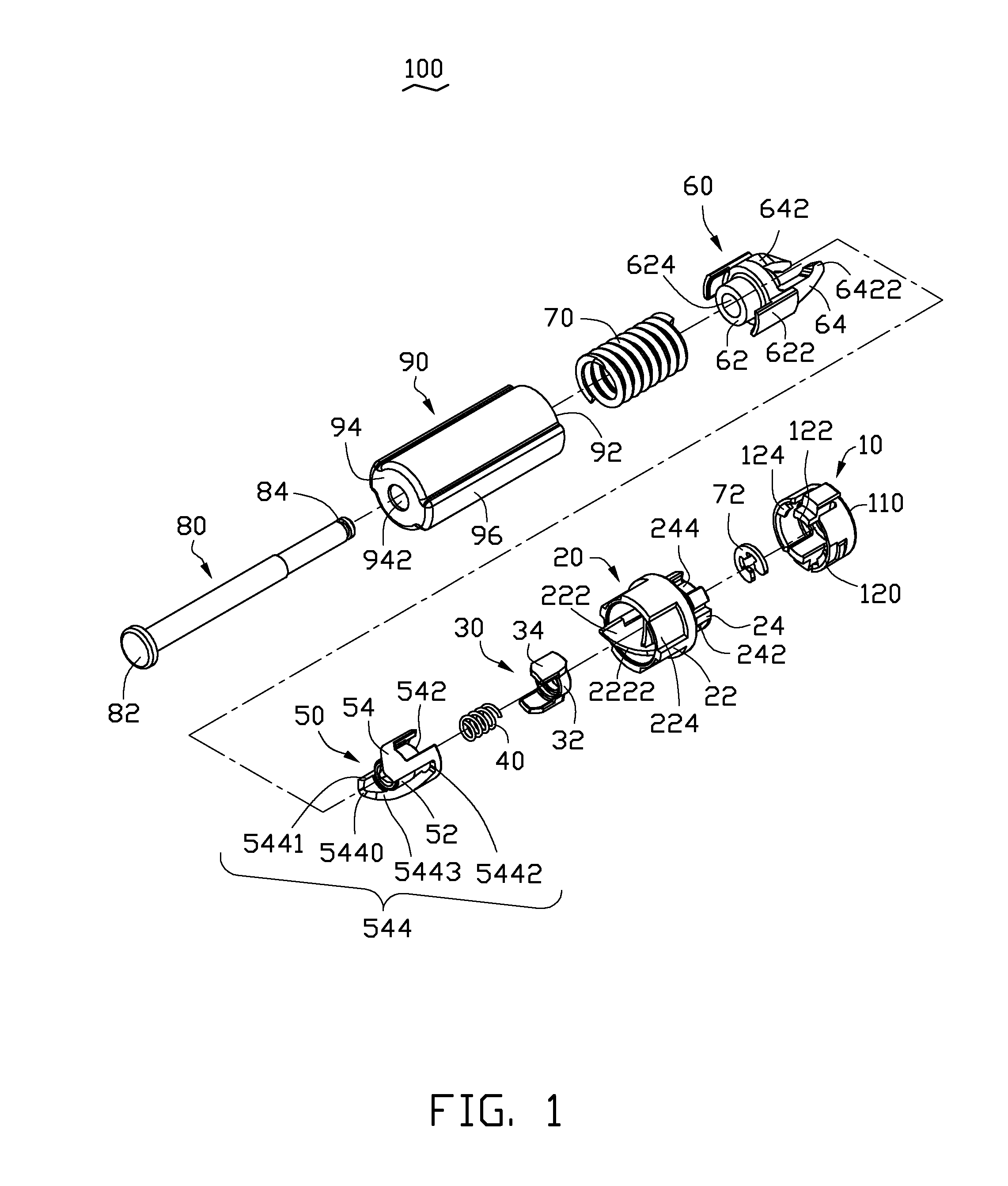
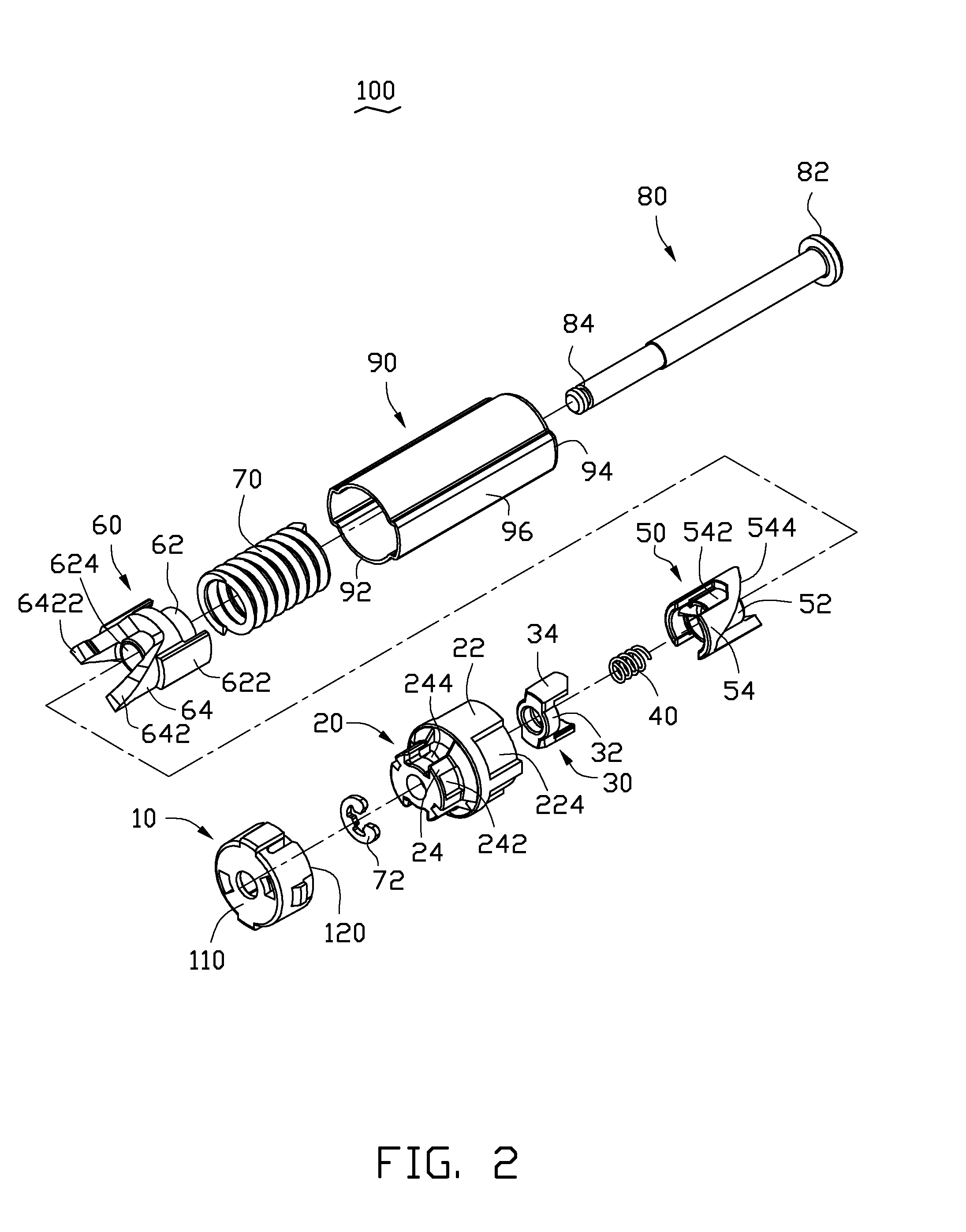
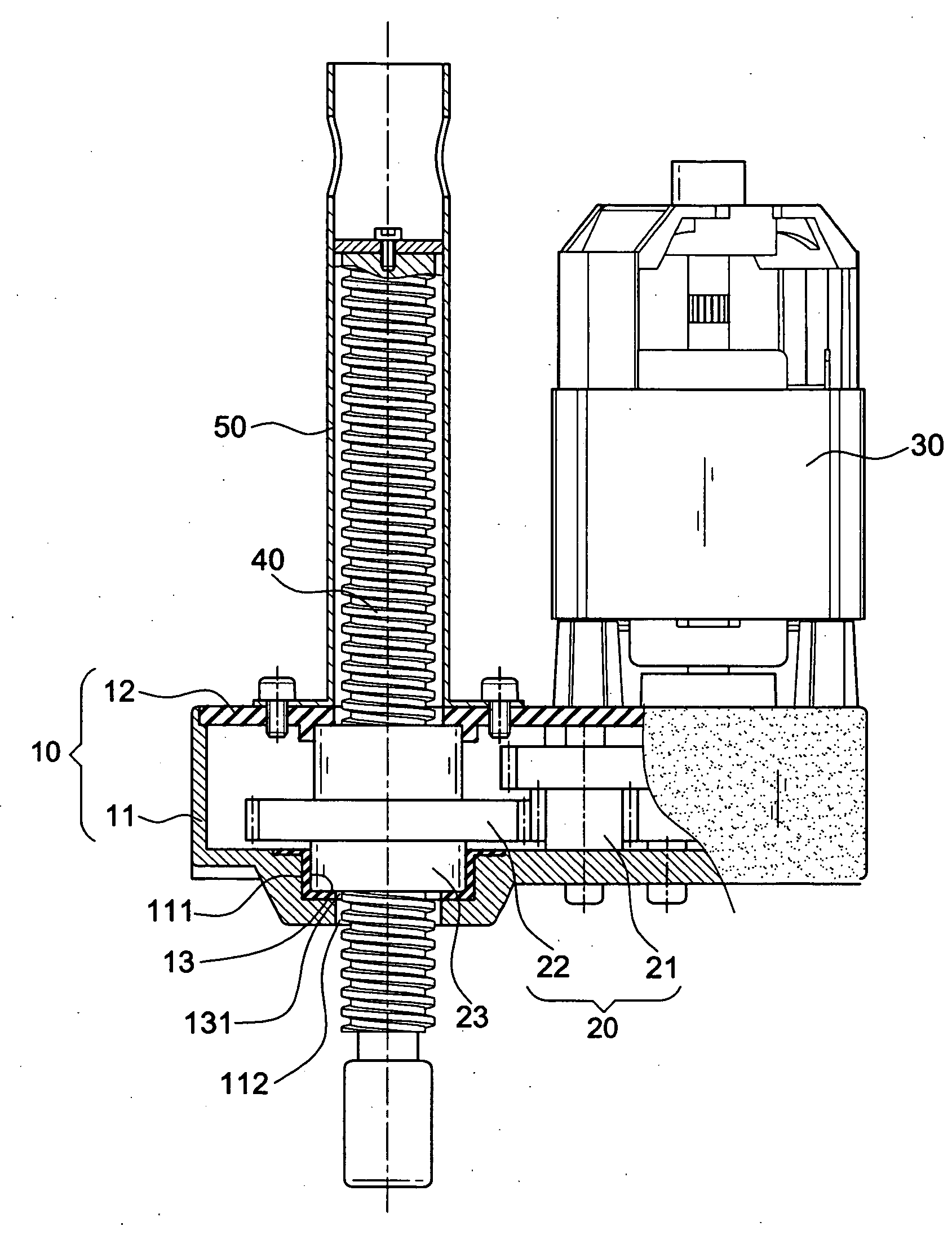
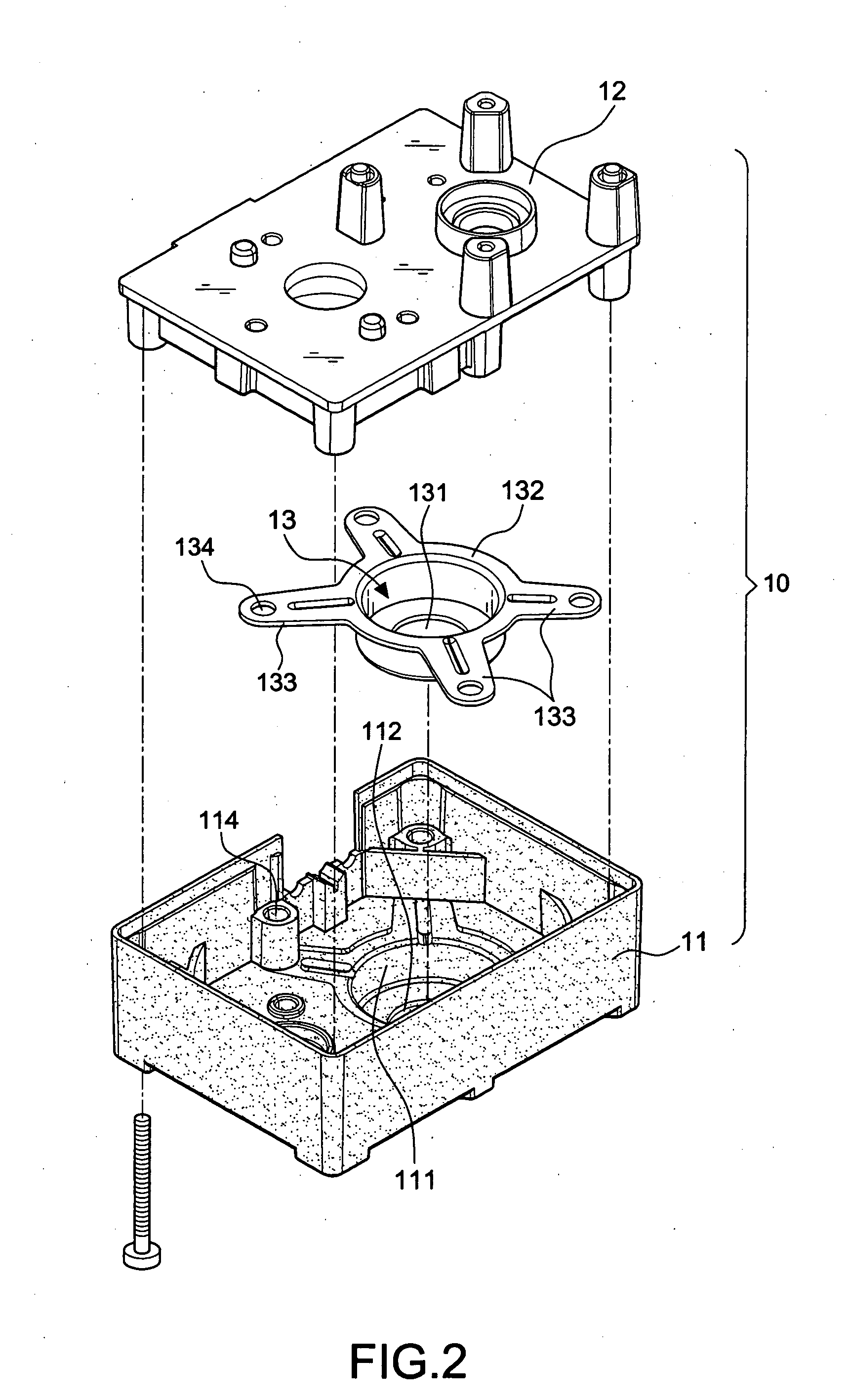
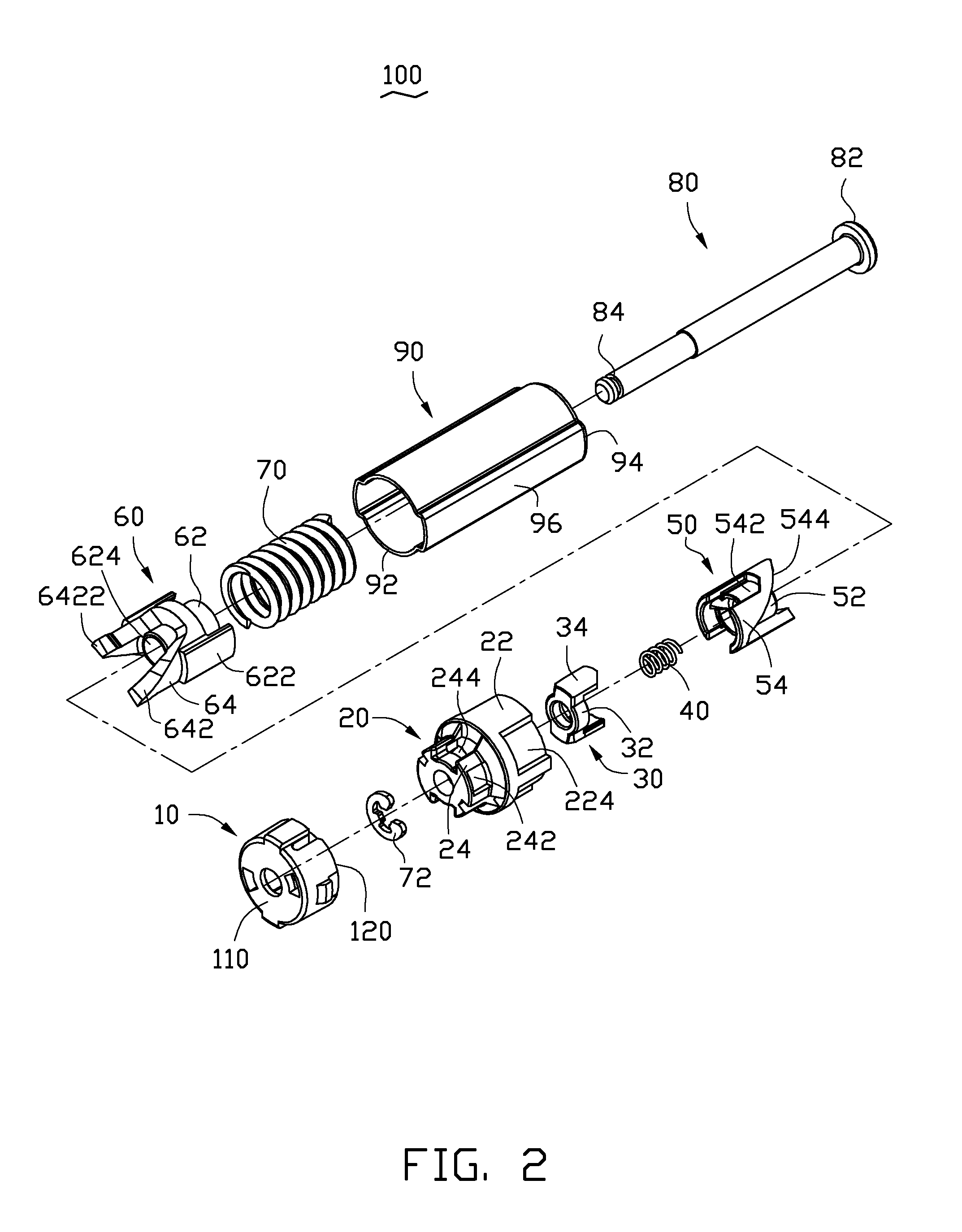
Button activated spring-loaded hinge assembly
InactiveUS20110286160A1Complicated to manufactureHigh strength materialSubstation equipmentHingesEngineeringCam
A hinge assembly includes a receiving seat, a control member, a clutch member, a follower, a main shaft and an elastic element. The receiving seat includes a first cam surface. The control member is releasably latched with the receiving seat. The clutch member includes at least one projection. The follower includes a second cam surface engaging with the at least one projection. The receiving seat, the control member, the clutch member and the follower are placed around the shaft. The elastic element provides an elastic force for the follower. After the control member is unlocked, the second cam surface engages with the first cam surface.
Owner:SHENZHEN FUTAIHONG PRECISION IND CO LTD +1
Systems and methods for power connection
InactiveUS20100216332A1Effective electricalRobust mechanical connectionCoupling device detailsCooling/ventillation arrangementElectric machineElectrical connection
The invention provides systems and methods for power connection, which may be a sealed power connection. The sealed power connection may be used with electric machines, which may be fluid cooled. The sealed power connection may provide an effective electrical connection while providing a robust mechanical connection. The sealed power connection may provide electrical insulation of the connection from the machine housing, and may also be sealed to provide for internal fluid cooling of the electric machine and fluid cooling of the connection.
Owner:CLEAN WAVE TECH
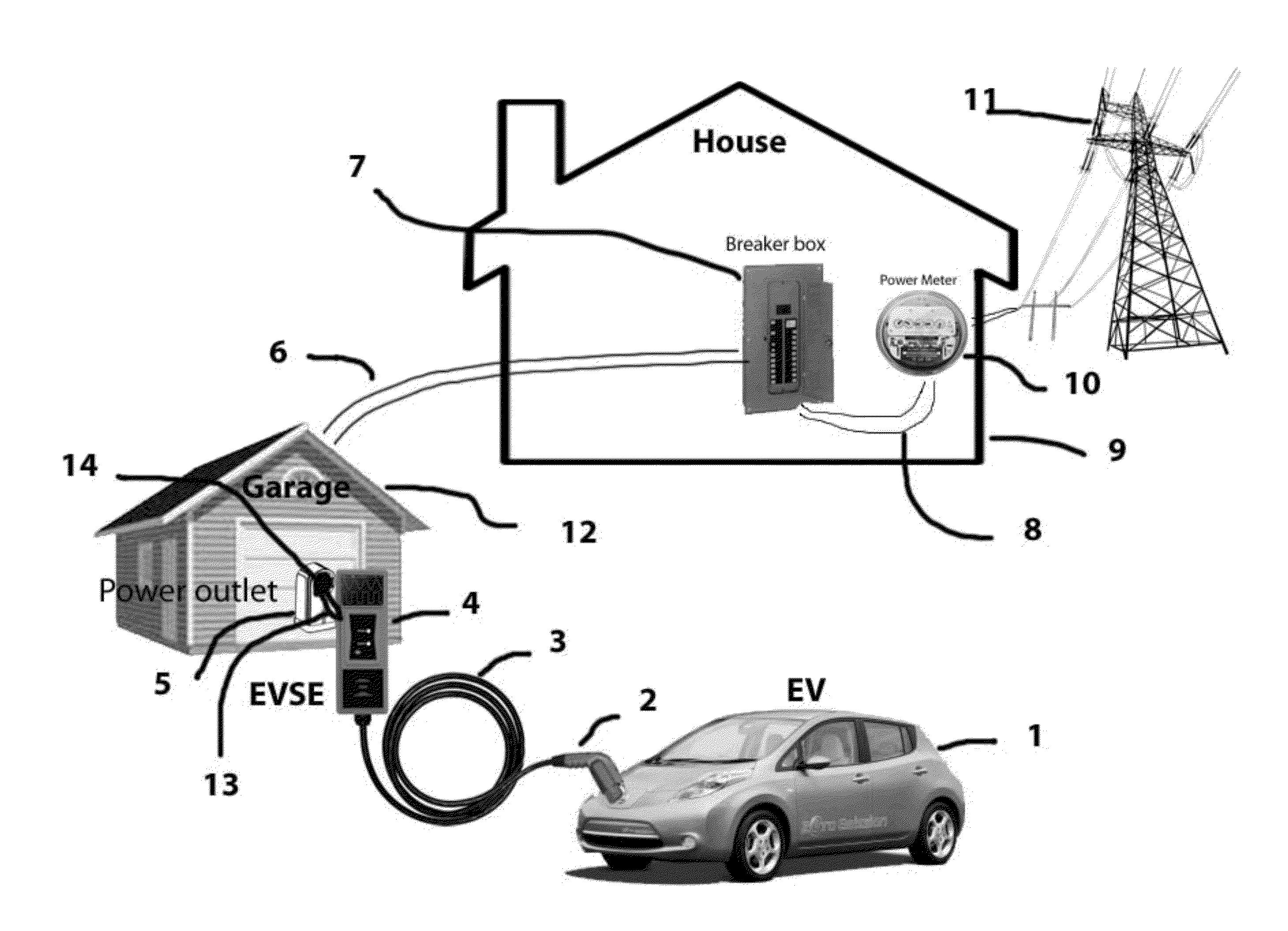
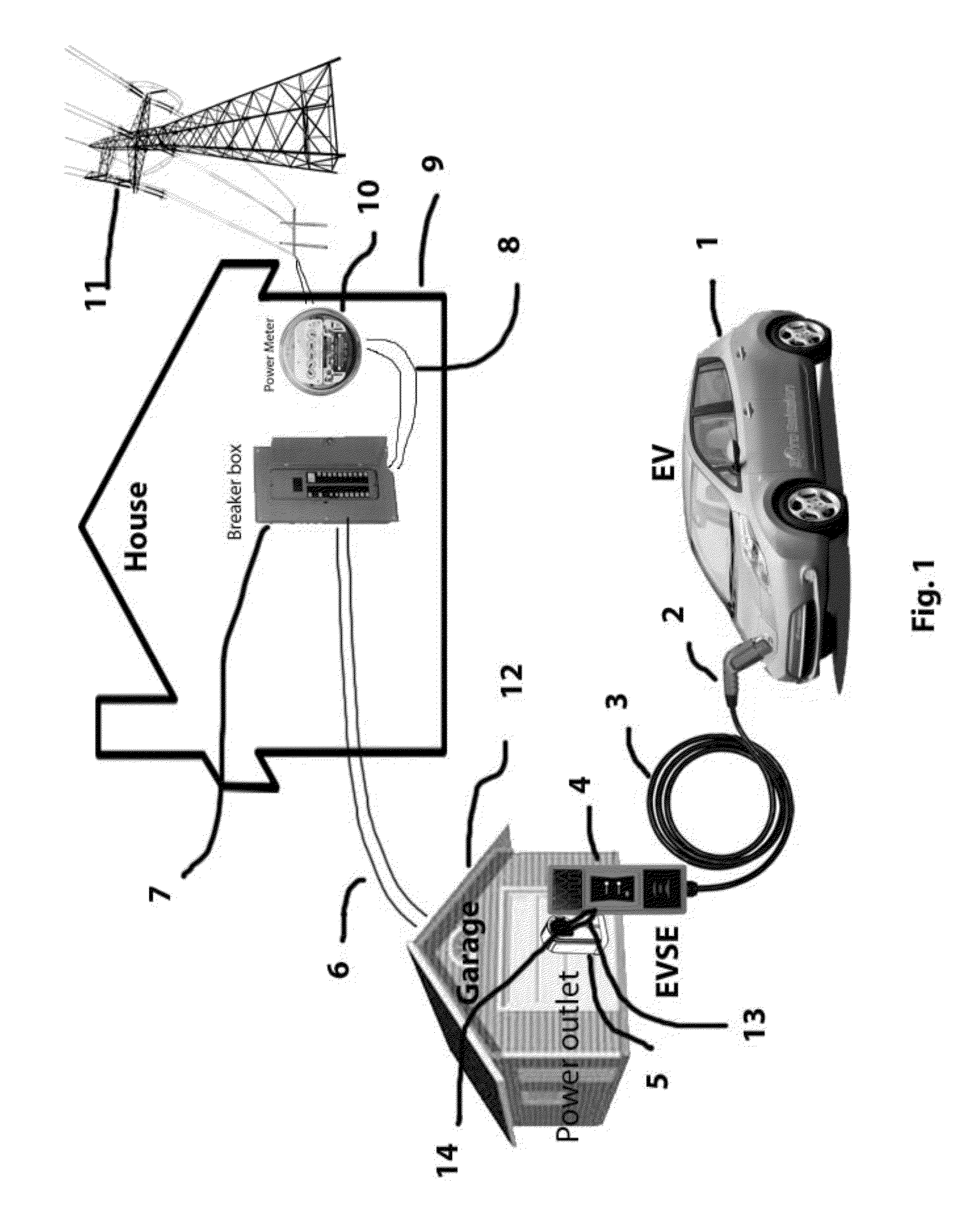
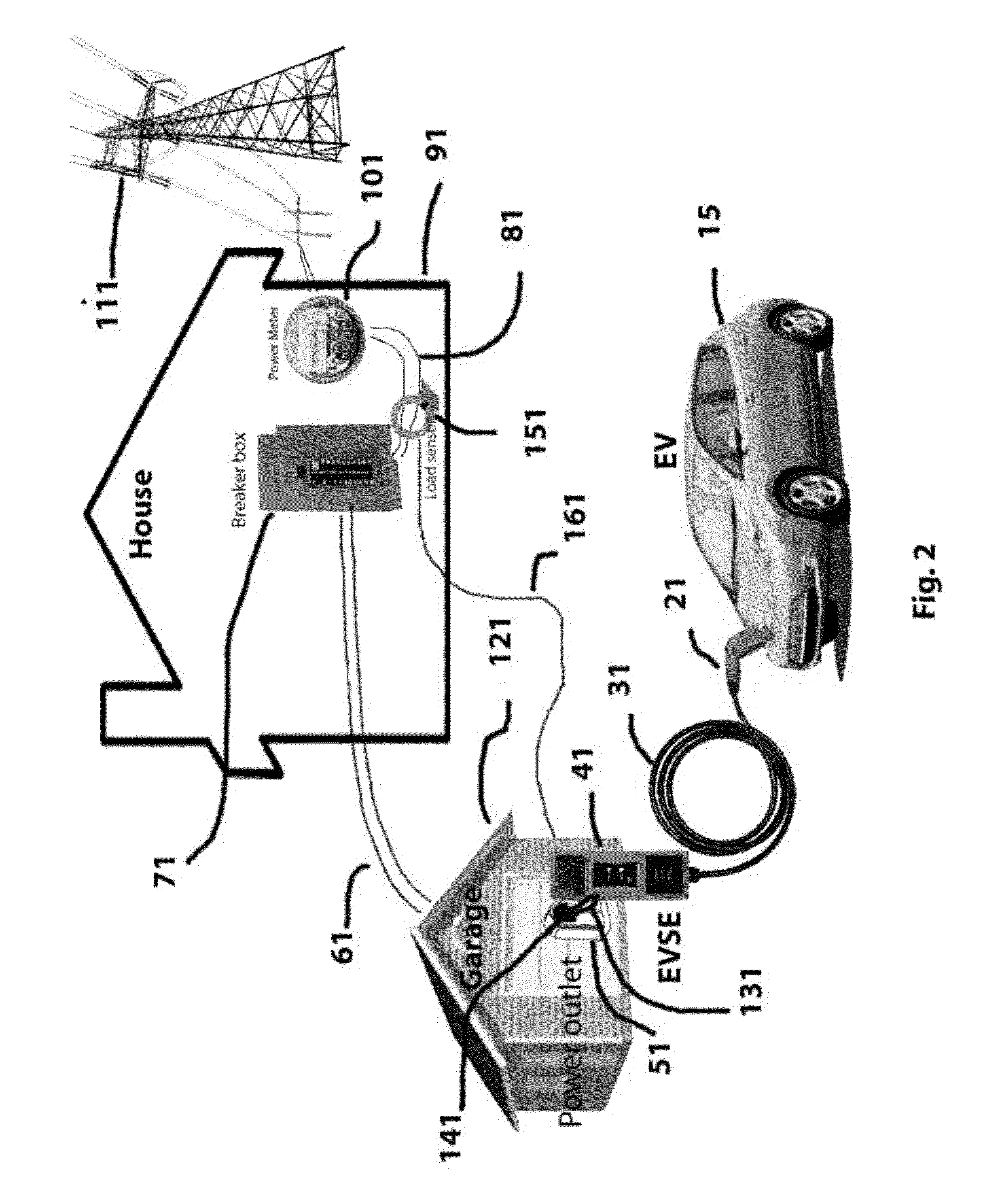
System and method for electric vehicle (EV) charging
InactiveUS20140077762A1High strength materialReal-time calibrationBatteries circuit arrangementsCharging stationsResidenceElectrical battery
A smart Electric Vehicle Supply Equipment (“EVSE”) that will accept input from different sources to determine the max rate of charge an Electric Vehicle (EV) can be safely charged at without overloading the electrical system of the residence. The system will also communication signals from the power company, fire department and police to reduce or eliminate the charging of the battery in the EV if the situation requires. The smart EVSE further comprises a non-invasive current and / or load sensing unit coupled to the power source for recharging the battery or batteries in the EV, cables to connect the load sensing unit to the EVSE, and optional wireless communication transmitter (sensor to EVSE), EVSE unit with compensating ability.
Owner:SPANOS MIKE
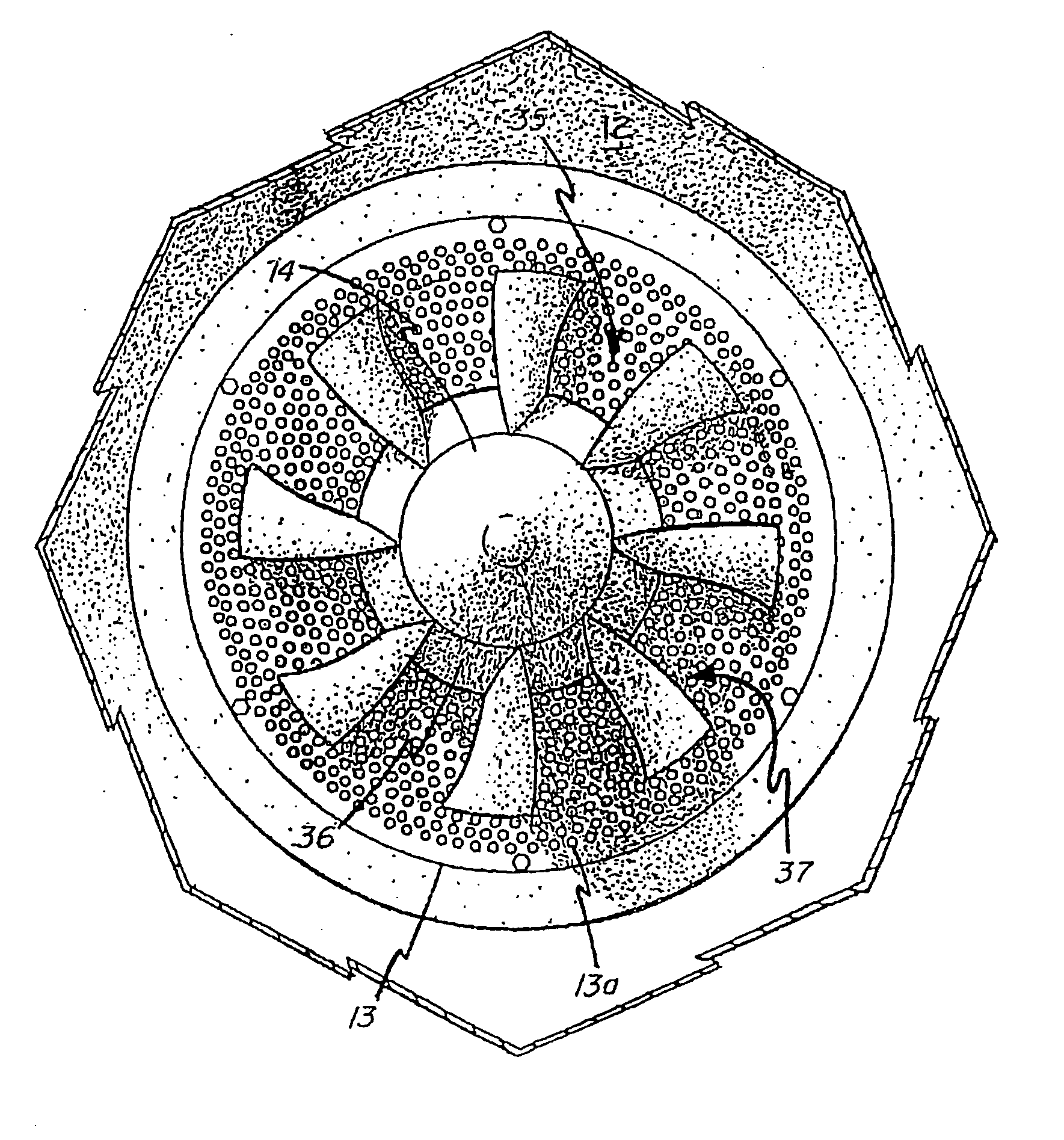
Pulper rotor and assembly
InactiveUS20050224610A1Promote circulationImprove the pumping effectGas current separationGrain millingTrailing edgeWear resistant
An improved pulper, mixer or defibering, rotor having a spar ring attached to a hub of the rotor with a series of curved vanes projecting from the spar ring. The curved vanes have a vane face and a trailing edge. The trailing edge may be unitary and integral with the vane, or may be segmented in combination with trailing edge portion provided on an underlying spar stub to which the vanes are attached. The hydrodynamic suction created by the trailing edge is enhanced by the addition of a dam at the vane tip end of the trailing edge zone. The vanes have a constant vane face angle relative to radians extending outward from the hub of the rotor. As a result of the constant vane face angle the pulp, or other materials, mixed by the vanes during rotation of the rotor are more consistently in contact with the vanes during rotation of the rotor. The vanes are also streamlined to reduce hydrodynamic drag especially at the vane tips where speed and therefore drag potential are at their highest levels. As a result, increased circulation and pumping effects with minimal power requirements are achieved. The vanes may be made of high wear resistant materials and are easily accessible for maintenance, repair or replacement.
Owner:THERMO BLACK CLAWSON
Systems and methods for power connection
InactiveUS20110045686A1Effective electricalRobust mechanical connectionPrinted circuit assemblingDustproof/splashproof/drip-proof/waterproof/flameproof connectionElectric machineElectrical connection
The invention provides systems and methods for power connection, which may be a sealed power connection. The sealed power connection may be used with an electric machine or any device that may require electrical and / or mechanical connection for power. The sealed power connection may provide an effective electrical connection while providing a robust mechanical connection. The electric machine or device may be fluid-sealed and / or fluid-cooled. The sealed power connection may provide for electrical insulation of the connection from the machine or device enclosure, and may also be sealed to provide for fluid sealing and / or internal fluid cooling of the electric machine or device, as well as fluid cooling of the connection.
Owner:CLEAN WAVE TECH
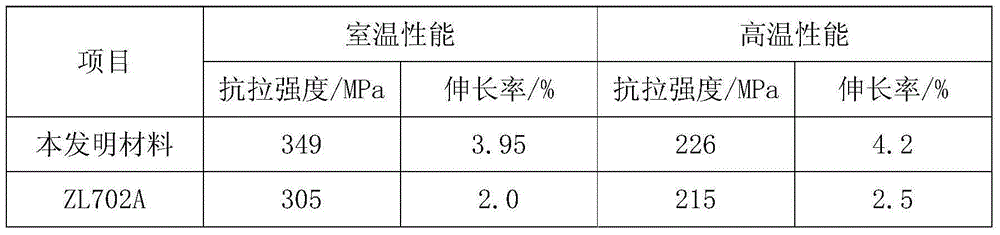
Castable aluminum alloy material for cylinder cover and preparation method thereof
The invention discloses a castable aluminum alloy material for a cylinder cover. The castable aluminum alloy material for the cylinder cover is characterized by comprising, by mass, 6.5%-7.5% of silicon, 0.55%-0.6% of zinc, 0.4%-0.5% of nickel, 0.2%-0.3% of vanadium, 0.22%-0.25% of magnesium, 0.2%-0.3% of iron, 0.1%-0.15% of tin, 45%-50% of scrap aluminum intermediate alloy powder, and the balance aluminum. Due to the facts that the proportions of the components of the alloy are optimized, the scrap aluminum intermediate alloy powder is used as the main component, and an appropriate amount of vanadium is added, the ductility of the aluminum alloy can be improved, and the castability and formability of the alloy are improved. According to the castable aluminum alloy material for the cylinder cover, a special treatment process is adopted, a refining agent made of porous materials is added, negative pressure is beneficial to degassing, and meanwhile materials in metal slag are separated; the prepared aluminum alloy material is high in strength and toughness, good in machinability and particularly suitable for manufacturing the engine cylinder cover.
Owner:FUYANG ANGU BOILER PRESSURE VESSEL MFG
Protective roof cover
InactiveUS20070266643A1Easy to installPrevention of hurricane-inflicted wind damageRoof coveringExtraordinary structuresSevere weatherHigh intensity
A protective roof cover which protects roof tiles and shingles from being torn from a roof during severe weather, such as a hurricane. The roof cover is aesthetically pleasing, durable, waterproof, and within code guidelines. The roof cover can be a variety of colors. The roof cover may be composed of a plurality of sheets of high strength material, wherein one sheet of high strength material is of sufficient length to cover a section said roof, at least one roll bar secured to a building below an overhang and attachable to the high strength material, and at least one wall channel secured to a building below an overhang for receiving the high strength material. Optionally, the roof cover can be a unitary sheet of durable, water proof material. The protective roof cover protects the roof against wind, rain and wind borne debris and other elements associated with storms.
Owner:COWVINS PRESTON
System for power connection
InactiveUS8334457B2Effective electricalRobust mechanical connectionPrinted circuit assemblingDustproof/splashproof/drip-proof/waterproof/flameproof connectionElectric machineElectrical connection
The invention provides systems and methods for power connection, which may be a sealed power connection. The sealed power connection may be used with an electric machine or any device that may require electrical and / or mechanical connection for power. The sealed power connection may provide an effective electrical connection while providing a robust mechanical connection. The electric machine or device may be fluid-sealed and / or fluid-cooled. The sealed power connection may provide for electrical insulation of the connection from the machine or device enclosure, and may also be sealed to provide for fluid sealing and / or internal fluid cooling of the electric machine or device, as well as fluid cooling of the connection.
Owner:CLEAN WAVE TECH
Two-step hot forming of steels
InactiveUS20180216205A1Shorten the timeSave energyHot-dipping/immersion processesFurnace typesAlloyUltimate tensile strength
Methods for press hardening steel alloys comprised of medium-Mn are provided. The press-hardened steel alloy may have an ultimate tensile strength (UTS) of at least 1,700 MPa and a tensile elongation of at least 8%. The press-hardened steel alloy may be formed in two forming steps above the martensitic finish temperature. The press-hardened steel may have a microstructure comprising martensite at greater than or equal to about 80% to less than or equal to about 98% and retained austenite at less than or equal to about 20% to greater than or equal to about 2%.
Owner:GM GLOBAL TECH OPERATIONS LLC
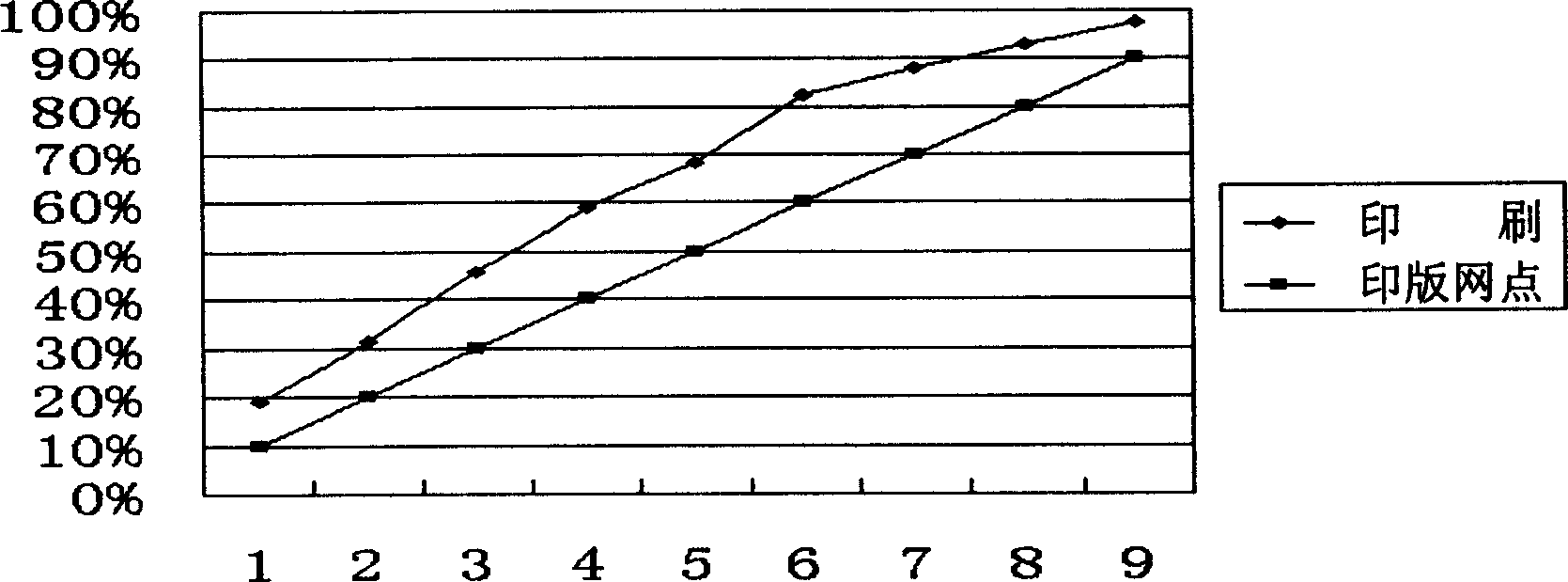
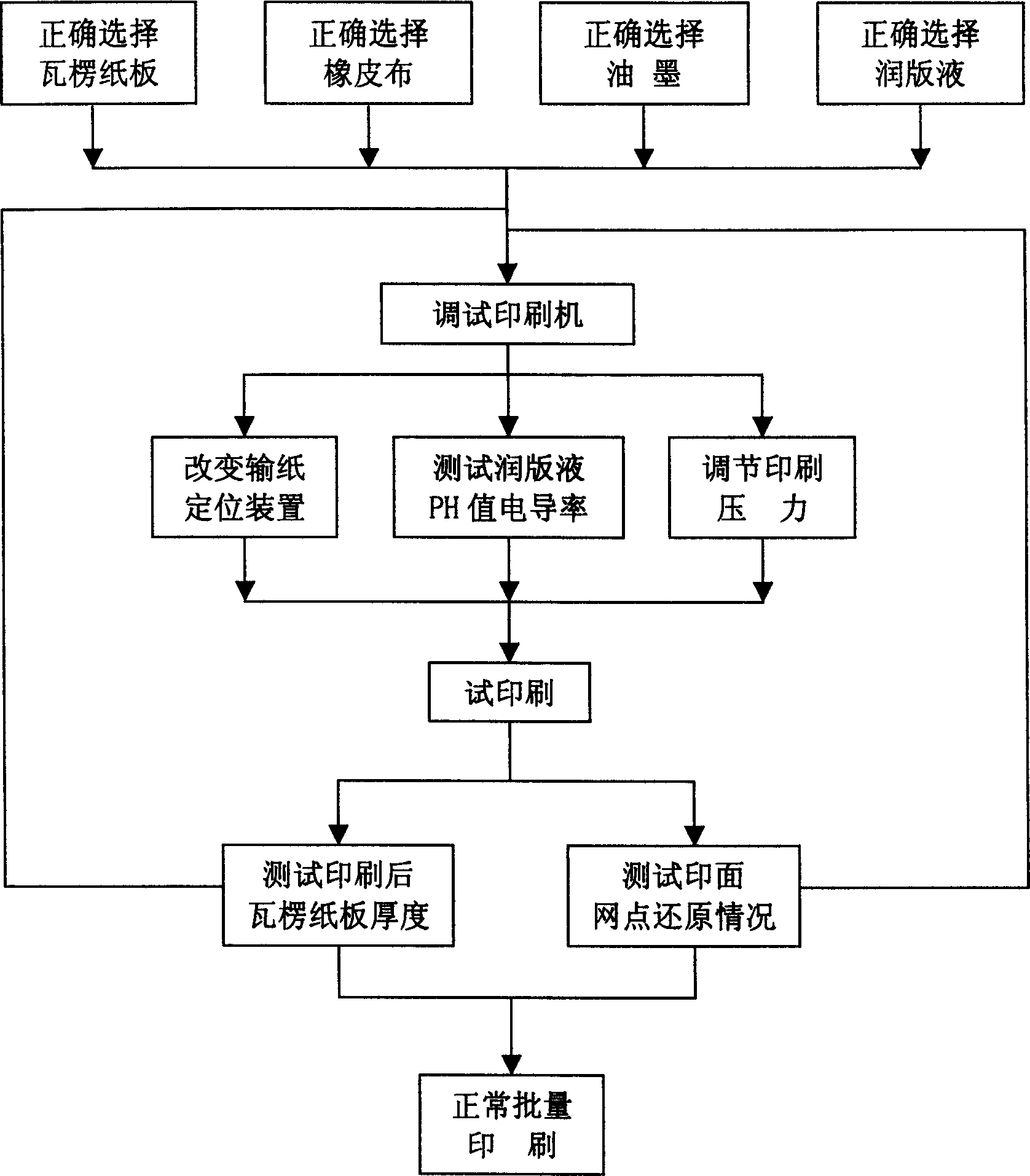
Method for printing miniature corrugated board by direct offset printing
InactiveCN1528591AInnovation in printing methodsGram weight highRotary pressesPrinting inkEngineering
The invention discloses a printing method of direct-hectographing micro corrugated board, its technical flow including: selecting printing corrugated board, mackintosh, printing ink, fountain solution and printing plate material; debugging the printer; adjusting printing pressure and monitoring the thickness of the micro corrugated board before and after printed and printing screen dots. Its character: the unit quality of the micro corrugated board is controlled at 380-440g / sq m, the material of the micro corrugated board is 185g / sq m heavily coated white kraft plus 90g / sq m corrugated original paper plus 140g / sq m white kraft; the water ratio is controlled at 7-9%; the corrugation is perpendicular to printing line pressure; the printing ink emulsifying rate is controlled at 15-18%; it selects the mackintosh whose screen dot restoring rate is not less than 95%; the printing plate material is a high-fidelity TP printing plate to truly restore screen dots. It has good presswork color restoring effect, clear image, etc.
Owner:上海外贸界龙彩印有限公司
Deformable restraint guide for use with child restraint system
ActiveUS7401859B2High strengthHigh-strength materialVehicle seatsSafety beltsSmall statureMechanical engineering
Deformable guides for use with smaller persons and / or in a child restraint system are provided. The guides are of at least two varieties, one for inclusion within a belt system, so as to absorb the impact and stresses of a collision or other abrupt vehicular motion and the other of a type useful to guide the restraint system of an automobile about the correct location on a smaller person. The second type permits a smaller individual to be properly secured to a restraint system in spite of his small stature. The guides are made of multiple layers of metal, plastic or other structurally significant materials, or combinations thereof, and designed to receive the energy of a collision such that a guide will deform, without breaking, absorbing the energy of a collision or other abrupt action.
Owner:KIM HOON Y +1
Stationary chain guide
InactiveUS20120295747A1Superior durabilityIncrease thicknessGearingEngineeringMechanical engineering
An elongated stationary guide for a traveling transmission chain in an engine such as a timing chain has a narrow reinforcing groove formed along the longitudinal direction of a metal base plate of the guide by surface-pressing, whereby the base plate exhibits plastic deformation. The narrow reinforcing groove extends toward the ends of the base plate past the locations at which the base plate is mounted on an engine block by mounting bolts.
Owner:TSUBAKIMOTO CHAIN CO
Compact and robust load and moment sensor
ActiveUS8746080B2Extended functional lifeEasy to replaceForce measurementApparatus for force/torque/work measurementElectrical resistance and conductanceLoad rejection
The present invention relates to a load and moment sensor for a prosthetic device detecting load in a single direction and moment in a single plane. The load and moment sensor includes a sensing element, and a plurality of strain gauges placed in specific locations of the sensing element. The plurality of strain gauges is part of a plurality of resistor circuits such as Wheatstone bridges. While the strain gauges can be located on a single sensing element, some resistive elements of the Wheatstone bridges can be located elsewhere on the prosthetic device. The combination of the location of the strain gauges and the use of the Wheatstone bridges allows for good side load rejection which is load and moment not in the single direction or the single plane.
Owner:PROTEOR USA LLC
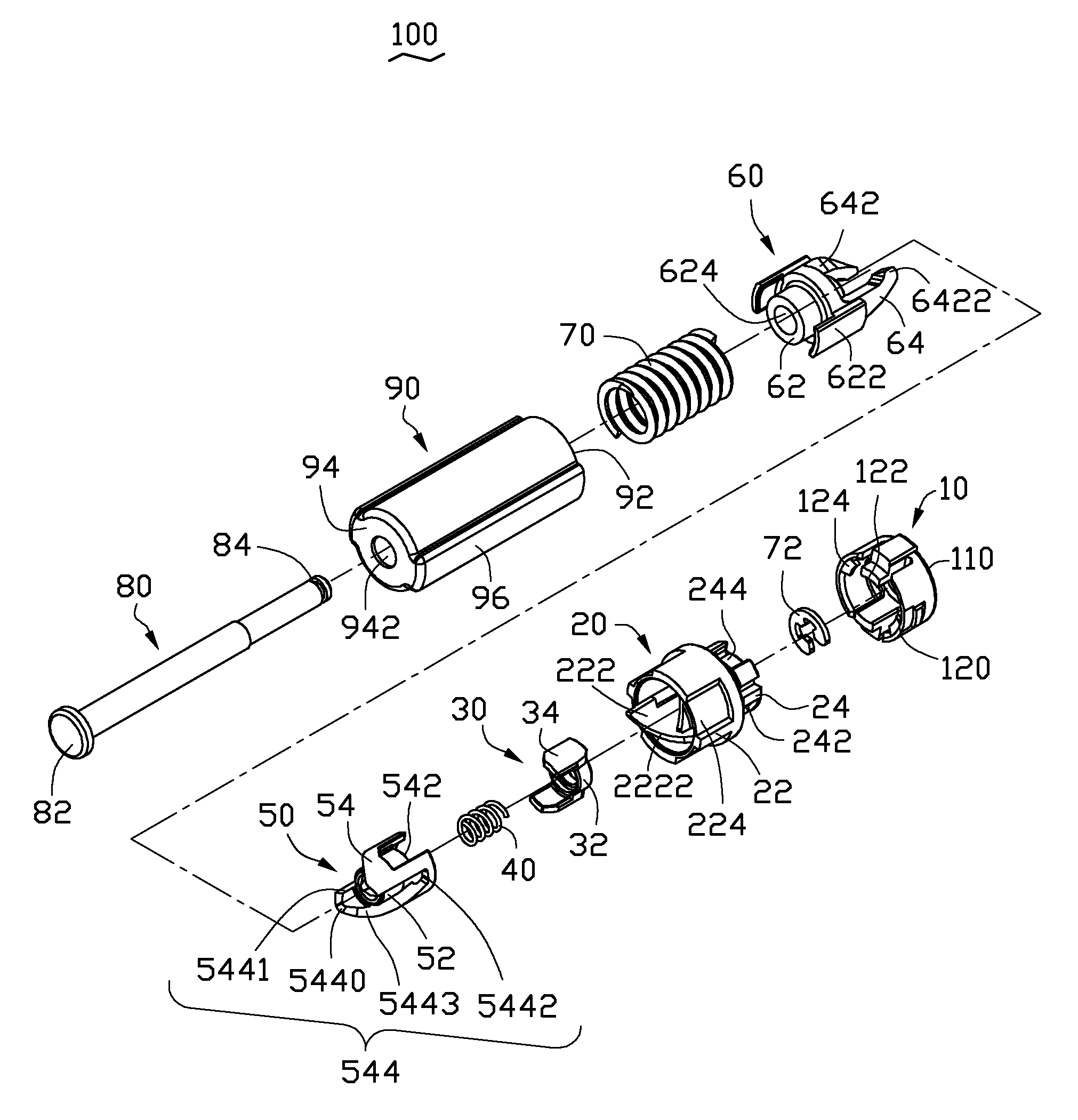
Button activated spring-loaded hinge assembly
InactiveUS20120140387A1Complicated to manufactureHigh strength materialDigital data processing detailsCasings/cabinets/drawers detailsCamClutch
A hinge assembly includes a receiving seat, a control member, a clutch member, a follower, a main shaft and an elastic element. The receiving seat includes a first cam surface. The control member is releasably latched with the receiving seat. The cam includes a second cam surface. The follower includes a third cam surface engaging with the first cam surface or the second cam surface. The receiving seat, the control member, the cam and the follower are placed around the shaft. The elastic element provides an elastic force for the follower. After the control member is unlocked, the second cam surface engages with the first cam surface.
Owner:SHENZHEN FUTAIHONG PRECISION IND CO LTD +1
Lifting mechanism for an exercise apparatus
InactiveUS20100083793A1Reduce weightExtended service lifeGearingSpace saving gamesFrame basedEngineering
A lifting mechanism for an exercise apparatus frame base includes a housing made of lightweight material with a positioning cavity having a through hole. A covering cap having a circular internal rim is integrally formed within the positioning cavity and made by high strength material an injection molding process is performed. The bottom of the covering cap includes an axle hole corresponding to the through hole. The top of the covering cap has an opening with a positioning flange extending outwards. In this way, the drawback that the inside of the conventional plastic housing is easily damaged by the transmission gear can be eliminated. As a result, the weight can be minimized and the strength can be maintained.
Owner:KO CHEN HUI
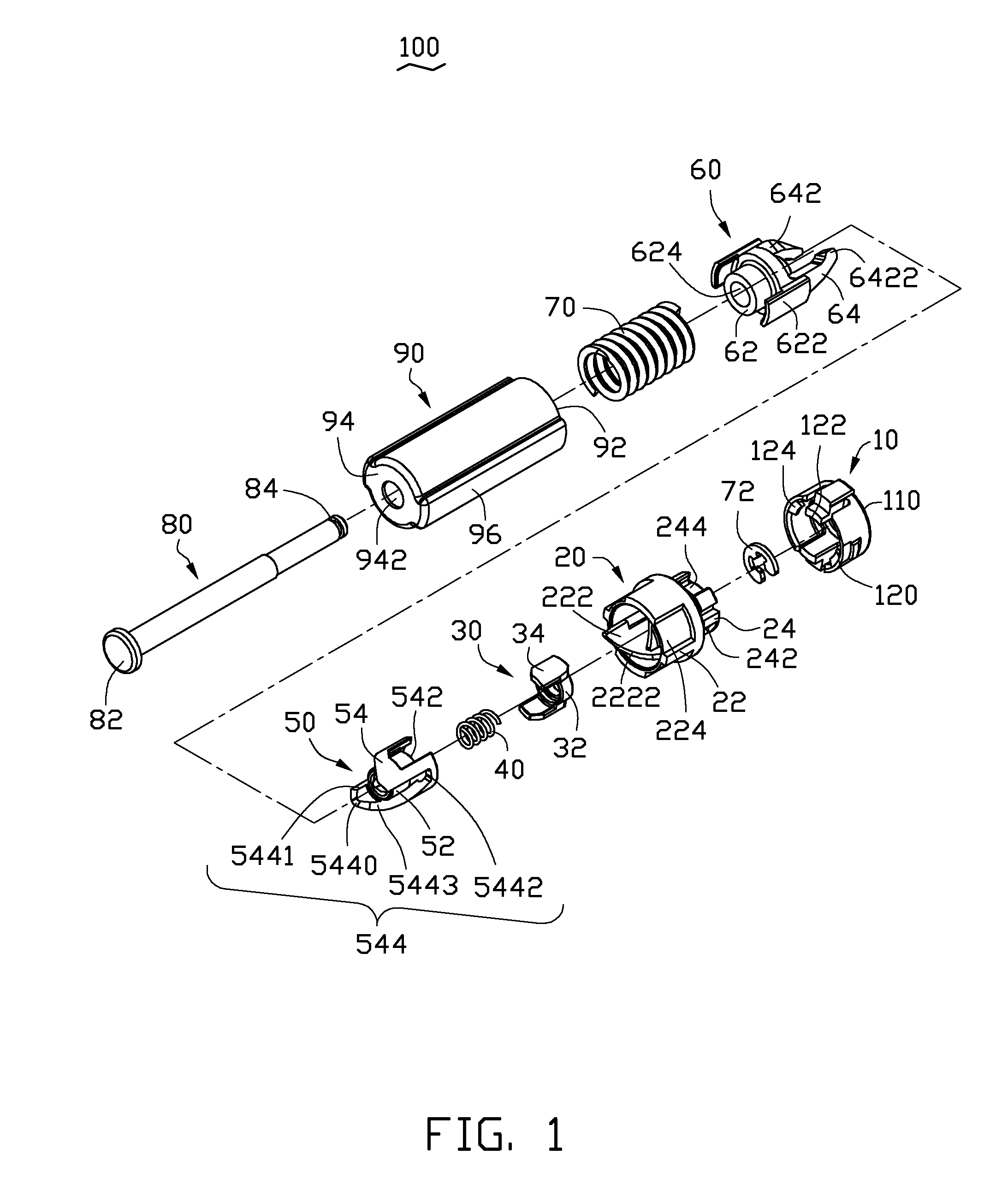
Button activated spring-loaded hinge assembly
InactiveUS8549709B2Complicated to manufactureHigh strength materialDigital data processing detailsWing openersEngineeringCam
A hinge assembly includes a receiving seat, a control member, a clutch member, a follower, a main shaft and an elastic element. The receiving seat includes a first cam surface. The control member is releasably latched with the receiving seat. The cam includes a second cam surface. The follower includes a third cam surface engaging with the first cam surface or the second cam surface. The receiving seat, the control member, the cam and the follower are placed around the shaft. The elastic element provides an elastic force for the follower. After the control member is unlocked, the second cam surface engages with the first cam surface.
Owner:SHENZHEN FUTAIHONG PRECISION IND CO LTD +1
Arrangement having a seal
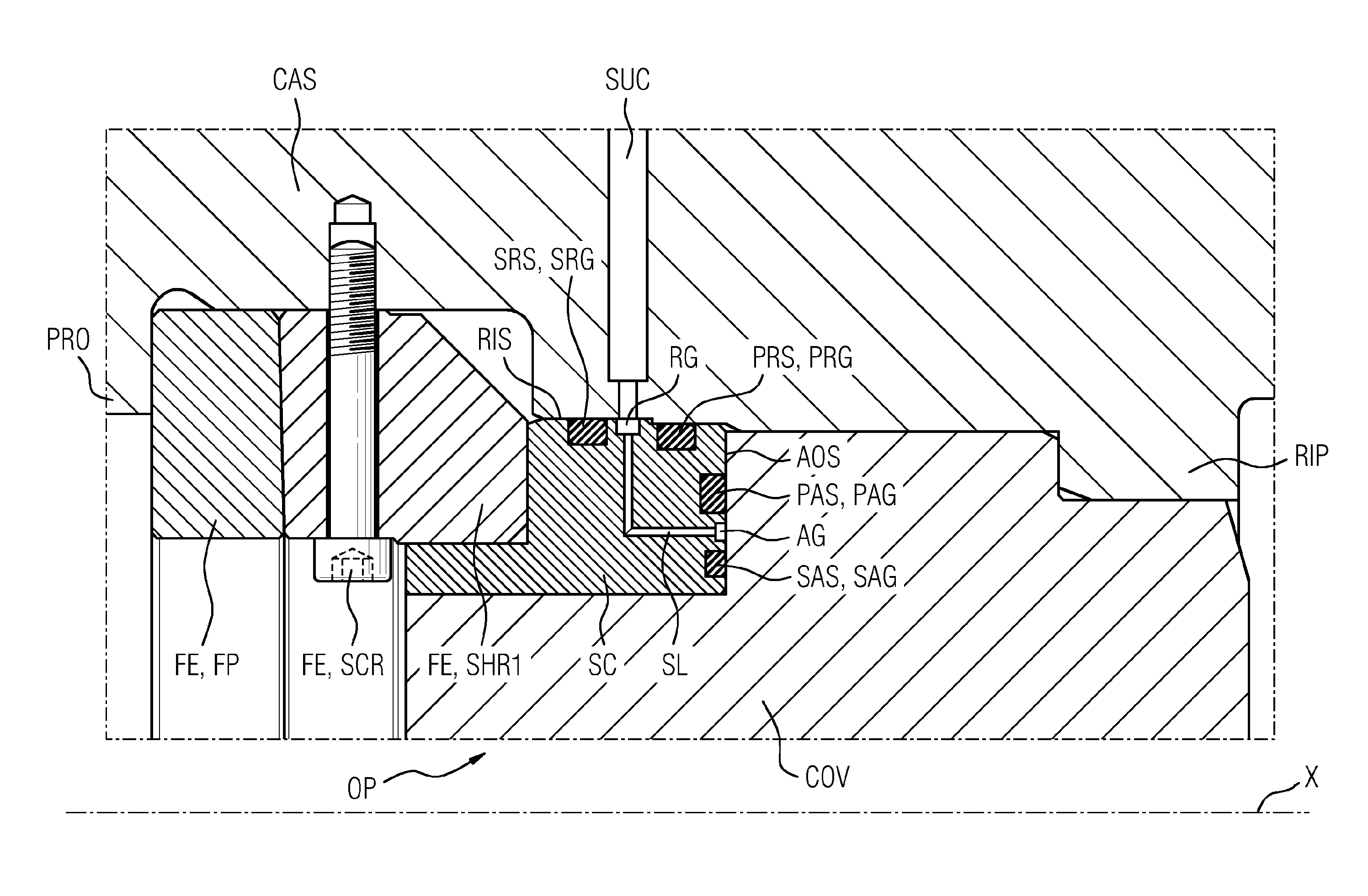
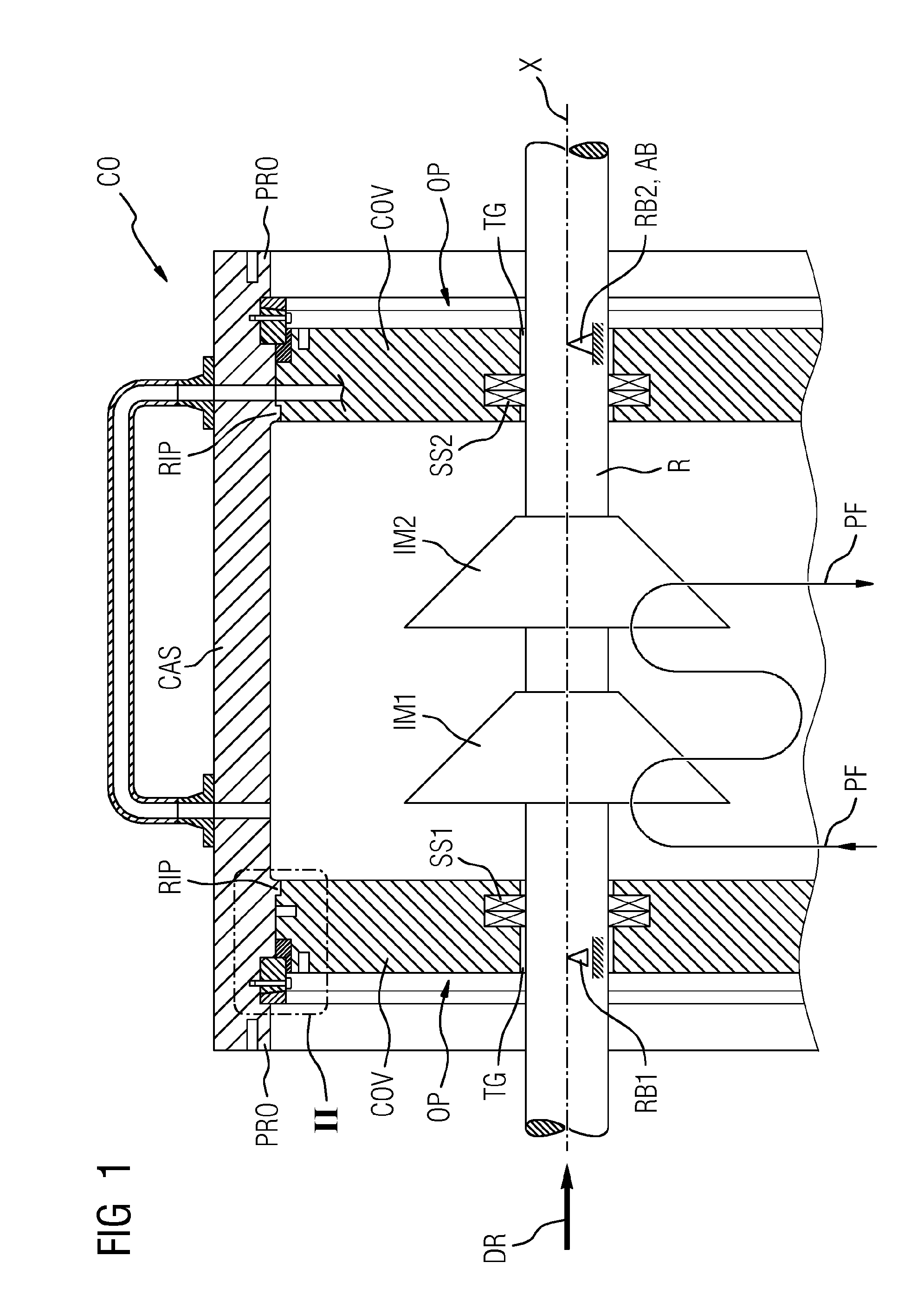
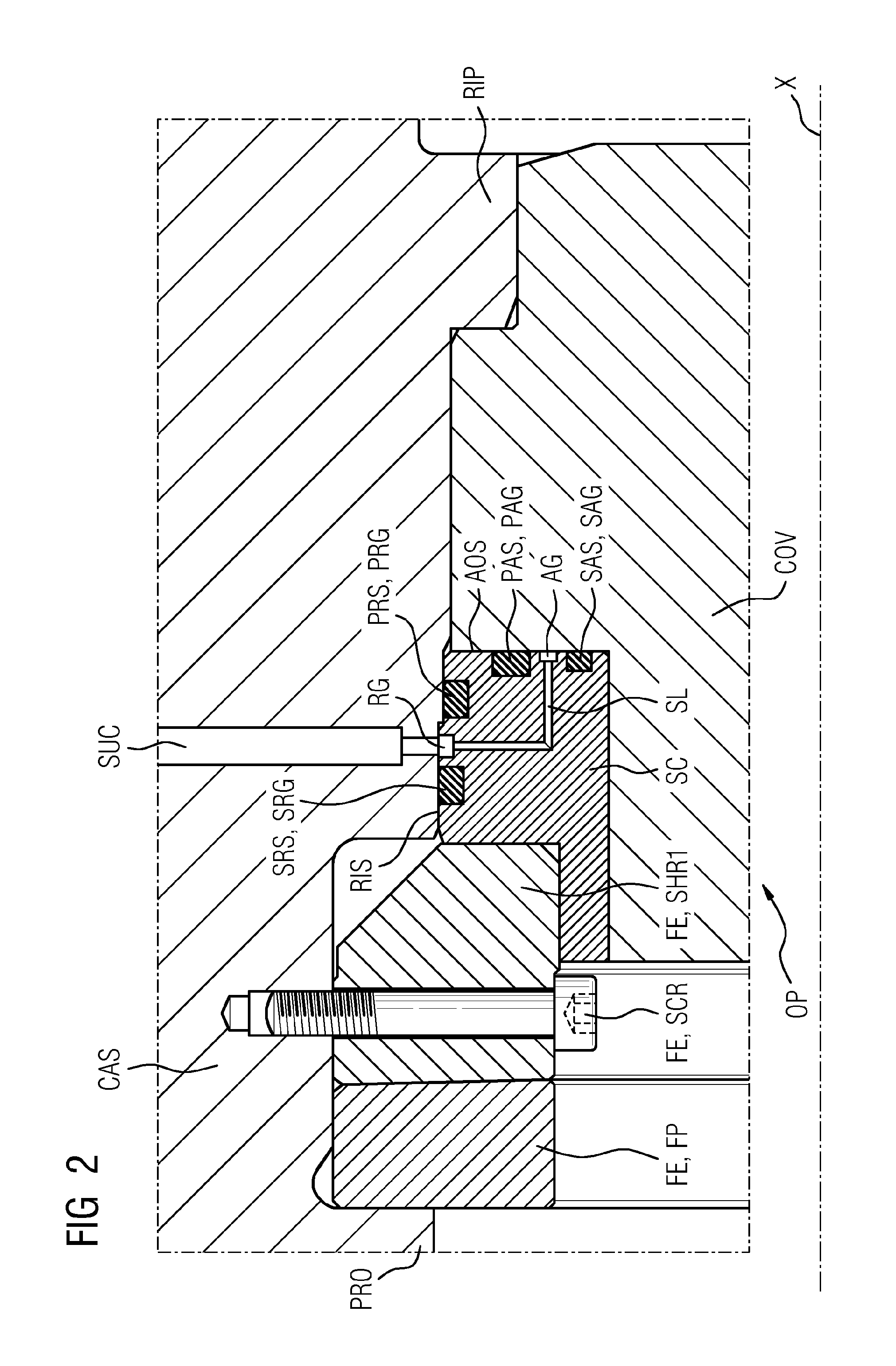
InactiveUS20160090990A1Avoid damageEasy to installEngine sealsPump componentsEngineeringDepth direction
An arrangement having a seal, includes: a casing, a cover, and at least one seal. In order to ensure damage-free installation of the cover and simultaneously simplify the installation process itself, as well as to guarantee good sealing, the arrangement has a seal carrier, the seal carrier has a first radial groove extending in the circumferential direction and having a radial depth direction, in which first radial groove a first radial seal is arranged, which seals against a radially inward surface of the casing, the seal carrier has a first axial groove extending in the circumferential direction and having an axial depth direction, in which first axial groove a first axial seal is arranged, which seals against an axial surface of the cover.
Owner:SIEMENS AG