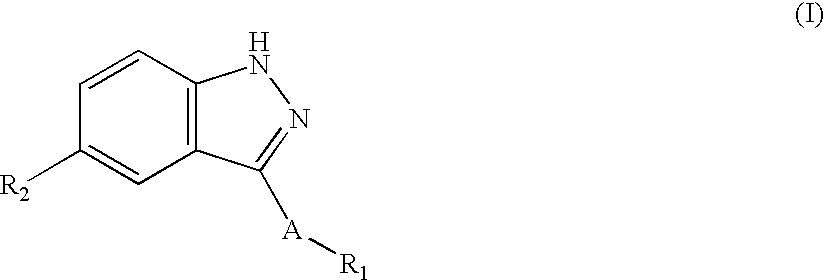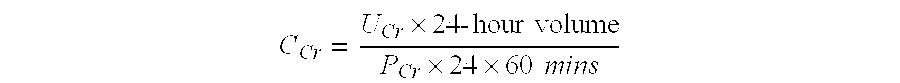Patents
Literature
340 results about "Kidney Failures" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Sorbent reactor for extracorporeal blood treatment systems, peritoneal dialysis systems, and other body fluid treatment systems
InactiveUS7169303B2Facilitate homogeneous suspensionReduce probabilitySolvent extractionHaemofiltrationFluid balancePeritoneal dialysis
Systems and methods for extracorporeal processing of blood or other body fluid for the treatment of conditions, such as sepsis, autoimmune disease, or toxemia related to kidney failure, liver failure, or drug overdose are provided. In an extracorporeal treatment system, a fraction of a body fluid is passed into a treatment fluid, at least a portion of which is then passed through a sorbent suspension reactor for treatment by a sorbent suspension. The treatment fluid circuit can be maintained at a fixed volume, which enables accurate fluid balance between the patient and the extracorporeal circuit. Some or all of the treatment fluid, optionally also containing nutrients and / or therapeutic agents, is returned to the patient. In a peritoneal dialysis system, dialysate is passed into a patient's peritoneal cavity, recovered from the cavity, passed through a sorbent suspension reactor in accordance with the invention, and returned to the cavity.
Owner:HEMOCLEANSE TECH
Sorbent reactor for extracorporeal blood treatment systems, peritoneal dialysis systems, and other body fluid treatment systems
InactiveUS20050006296A1Facilitate homogeneous suspensionReduce probabilityWater treatment parameter controlSemi-permeable membranesFluid balancePeritoneal dialysis
Systems and methods for extracorporeal processing of blood or other body fluid for the treatment of conditions, such as sepsis, autoimmune disease, or toxemia related to kidney failure, liver failure, or drug overdose are provided. In an extracorporeal treatment system, a fraction of a body fluid is passed into a treatment fluid, at least a portion of which is then passed through a sorbent suspension reactor for treatment by a sorbent suspension. The treatment fluid circuit can be maintained at a fixed volume, which enables accurate fluid balance between the patient and the extracorporeal circuit. Some or all of the treatment fluid, optionally also containing nutrients and / or therapeutic agents, is returned to the patient. In a peritoneal dialysis system, dialysate is passed into a patient's peritoneal cavity, recovered from the cavity, passed through a sorbent suspension reactor in accordance with the invention, and returned to the cavity.
Owner:HEMOCLEANSE TECH
Ion binding polymers and uses thereof
The present invention provides methods and compositions for the treatment of ion imbalances. In particular, the invention provides compositions comprising potassium binding polymers and pharmaceutical compositions thereof. Methods of use of the polymeric and pharmaceutical compositions for therapeutic and / or prophylactic benefits are disclosed herein. Examples of these methods include the treatment of hyperkalemia, such as hyperkalemia caused by renal failure and / or the use of hyperkalemia causing drugs.
Owner:VIFOR INT AG
Machine and Procedure For Extracorporeal Treatment of Blood
ActiveUS20080215247A1Shorten treatment timeIncrease the number ofOther blood circulation devicesHaemofiltrationBlood treatmentsBody fluid
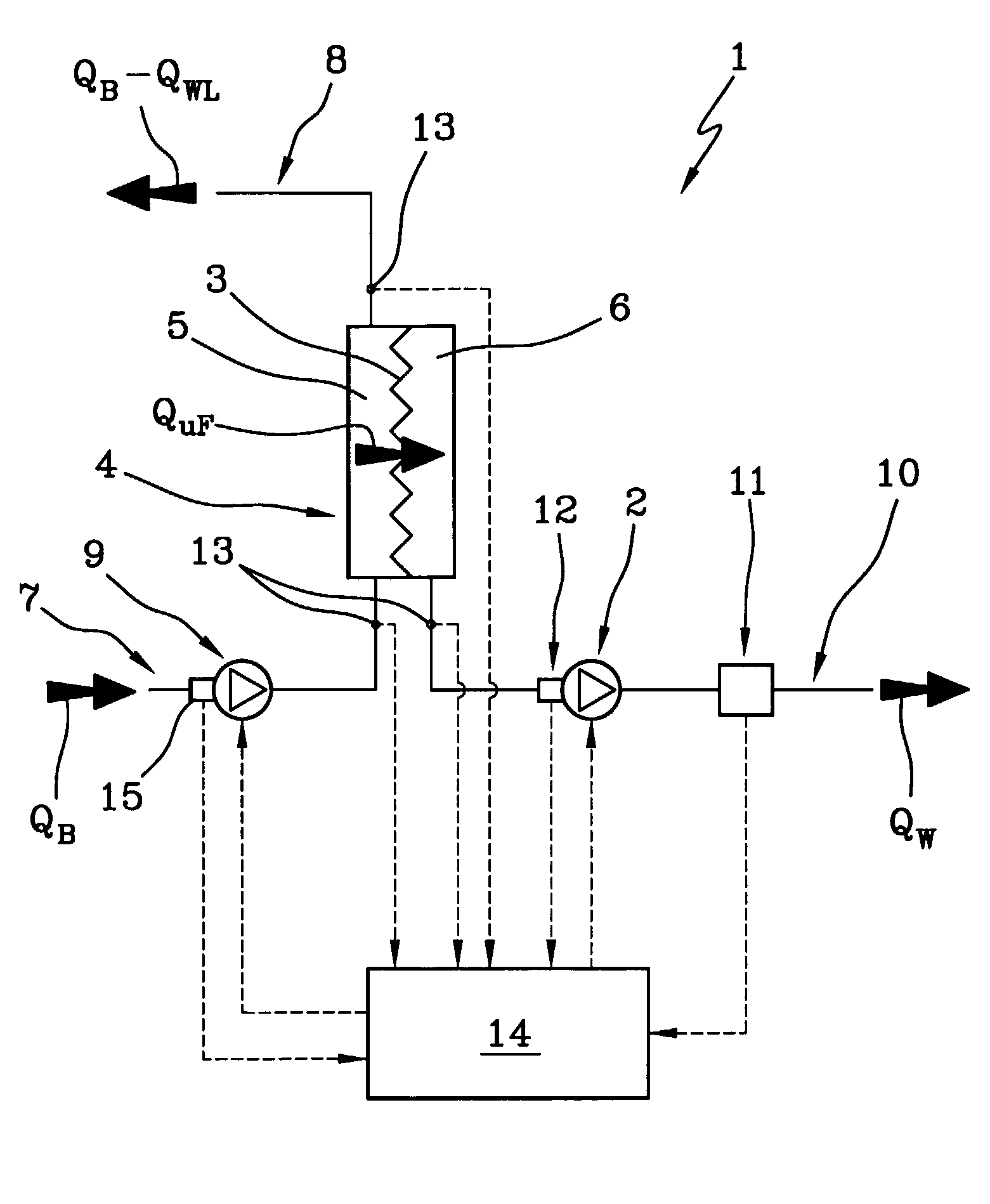
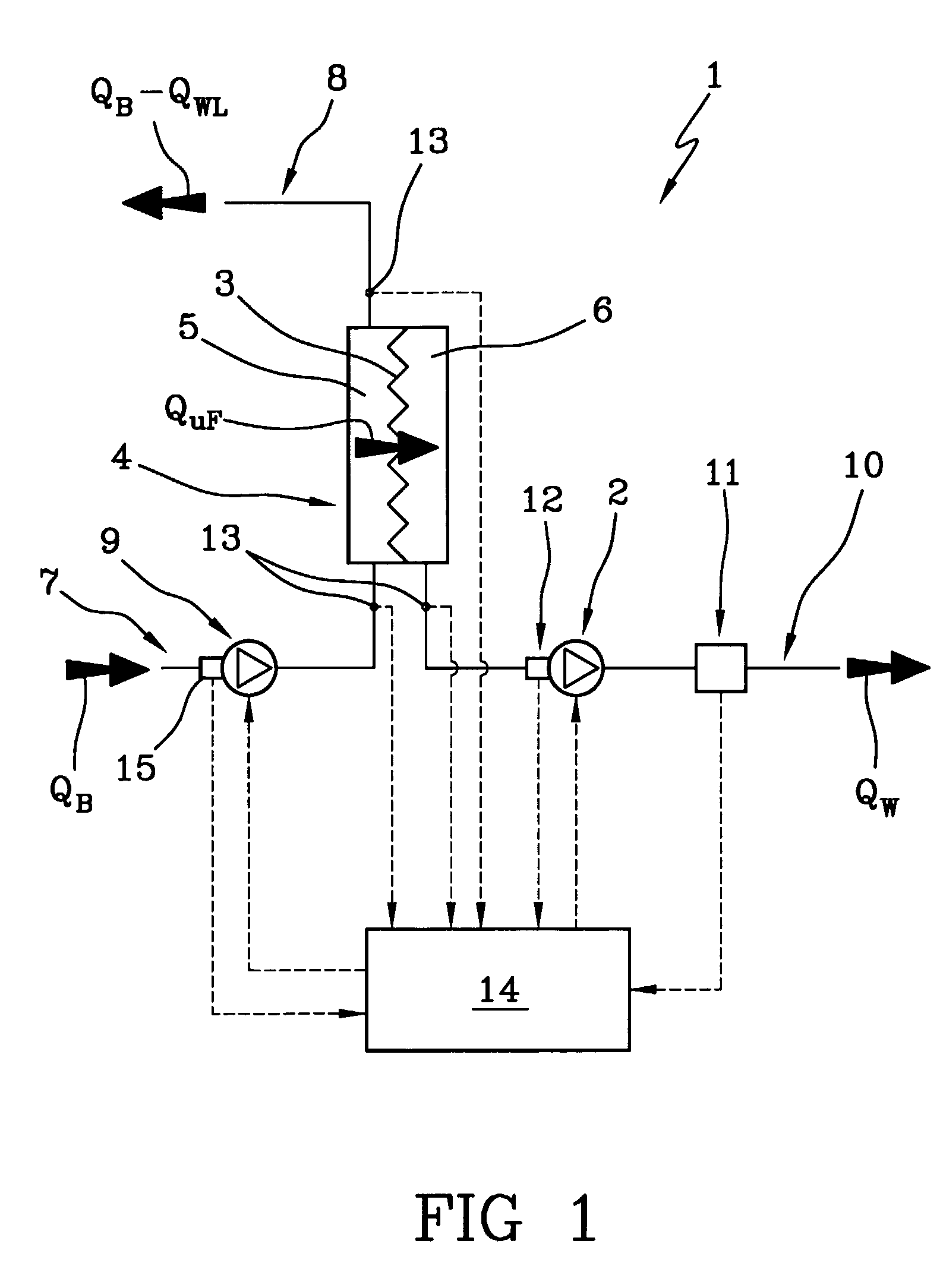
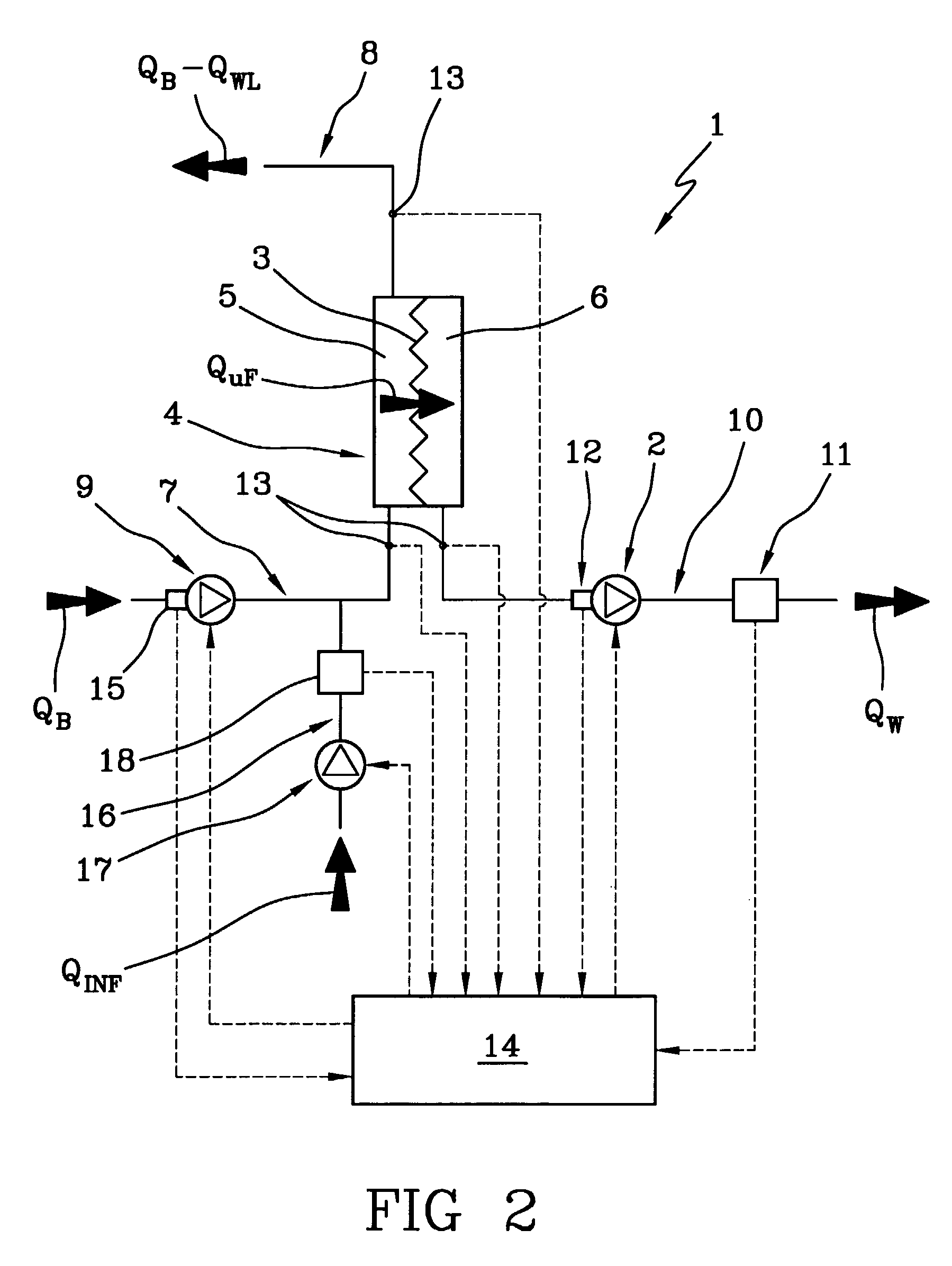
A machine (1) for extracorporeal blood treatment comprises a device (2) for ultrafiltration of a liquid through a semi-permeable membrane (3) of a blood treatment device (4), a first sensor (11) for ultrafiltration rate through the membrane, a second group of sensors (13) for measuring a trans-membrane pressure, and a programmed controller (14) for calculating a maximum ultrafiltration rate as a function of the transmembrane pressure. The machine is suitable for kidney failure treatments, such as hemofiltration and hemodiafiltration. In a relatively short time the machine enables a large quantity of corporeal blood to be removed from the patient.
Owner:GAMBRO LUNDIA AB
Method for the early detection of renal injury
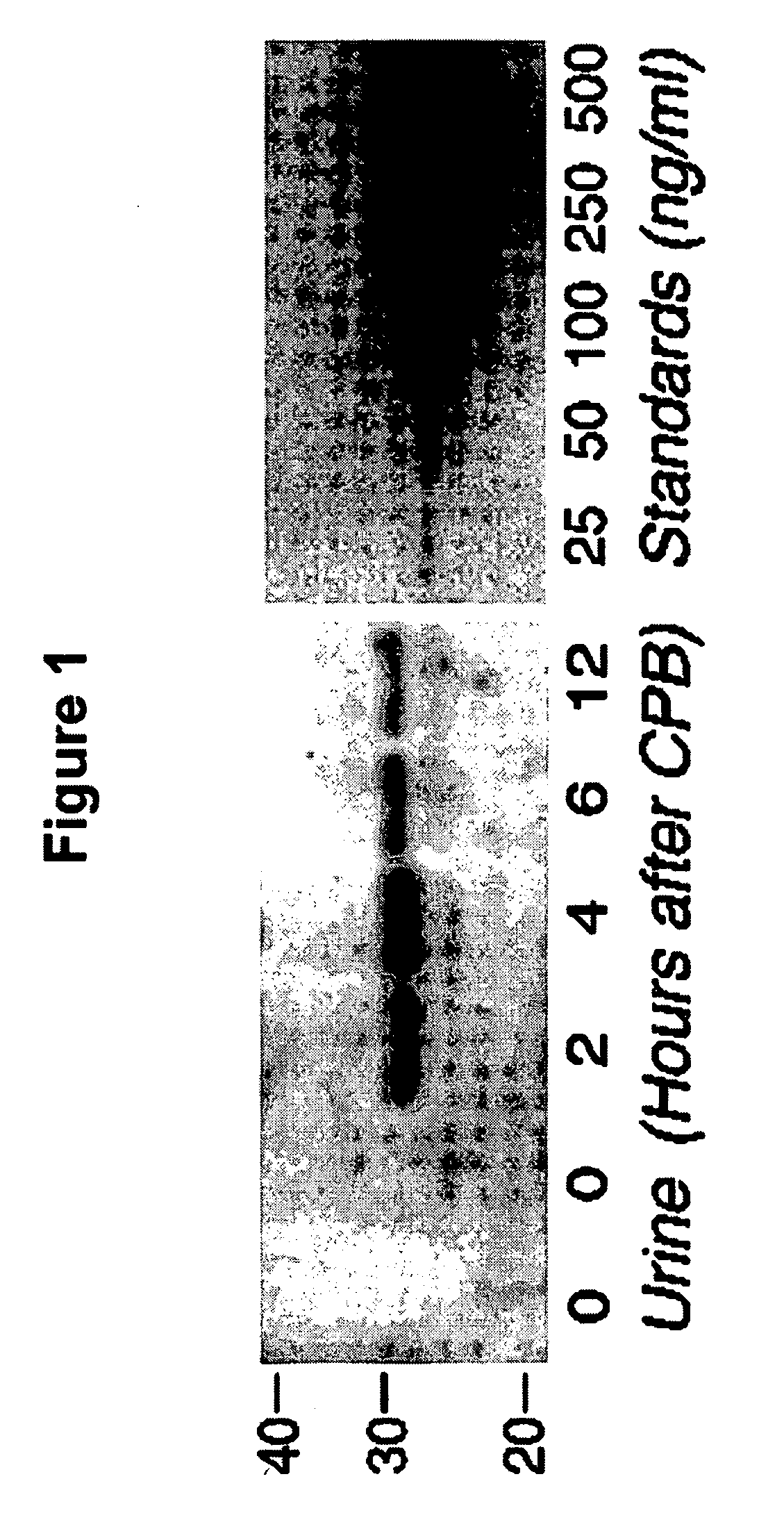
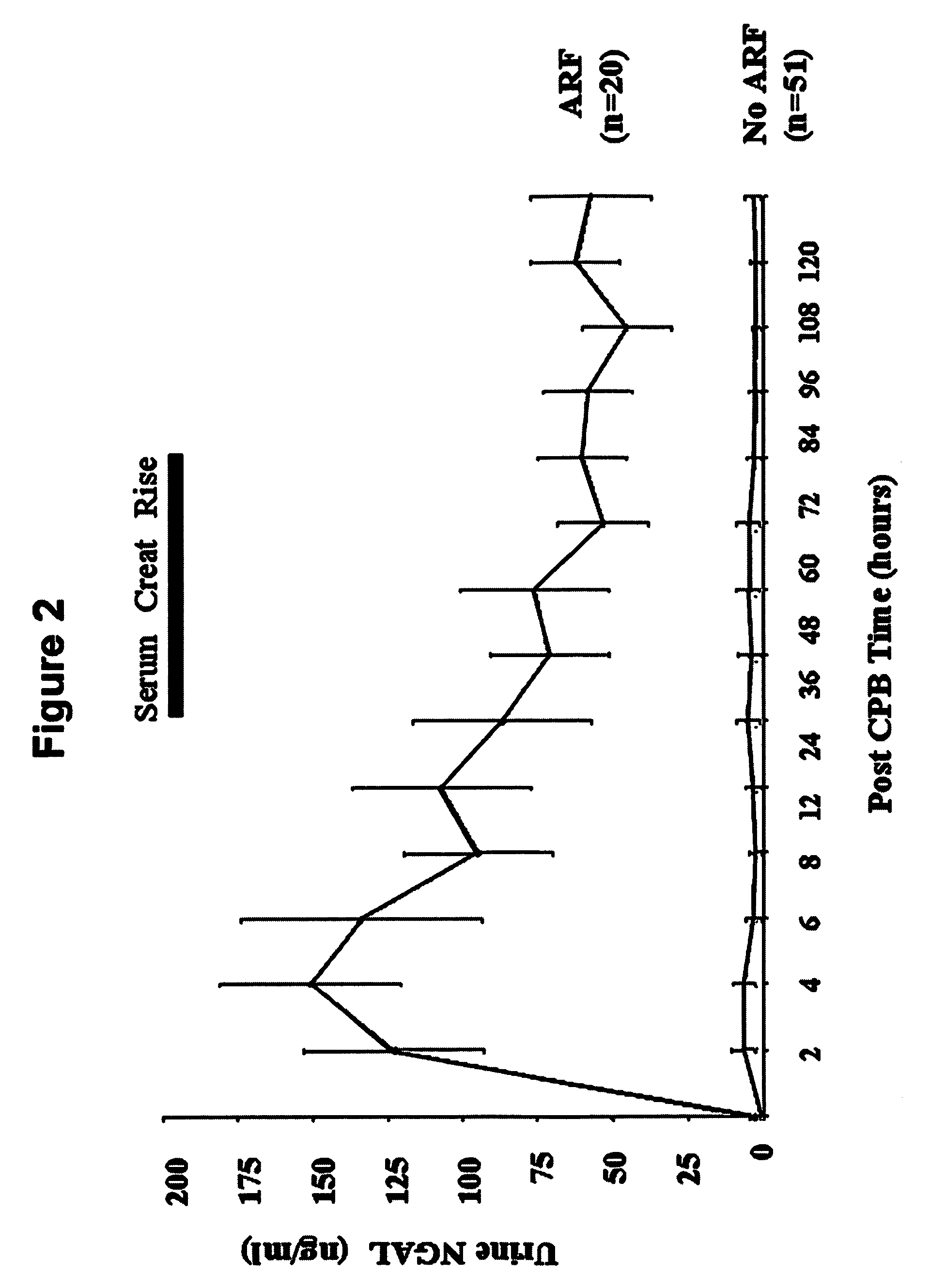
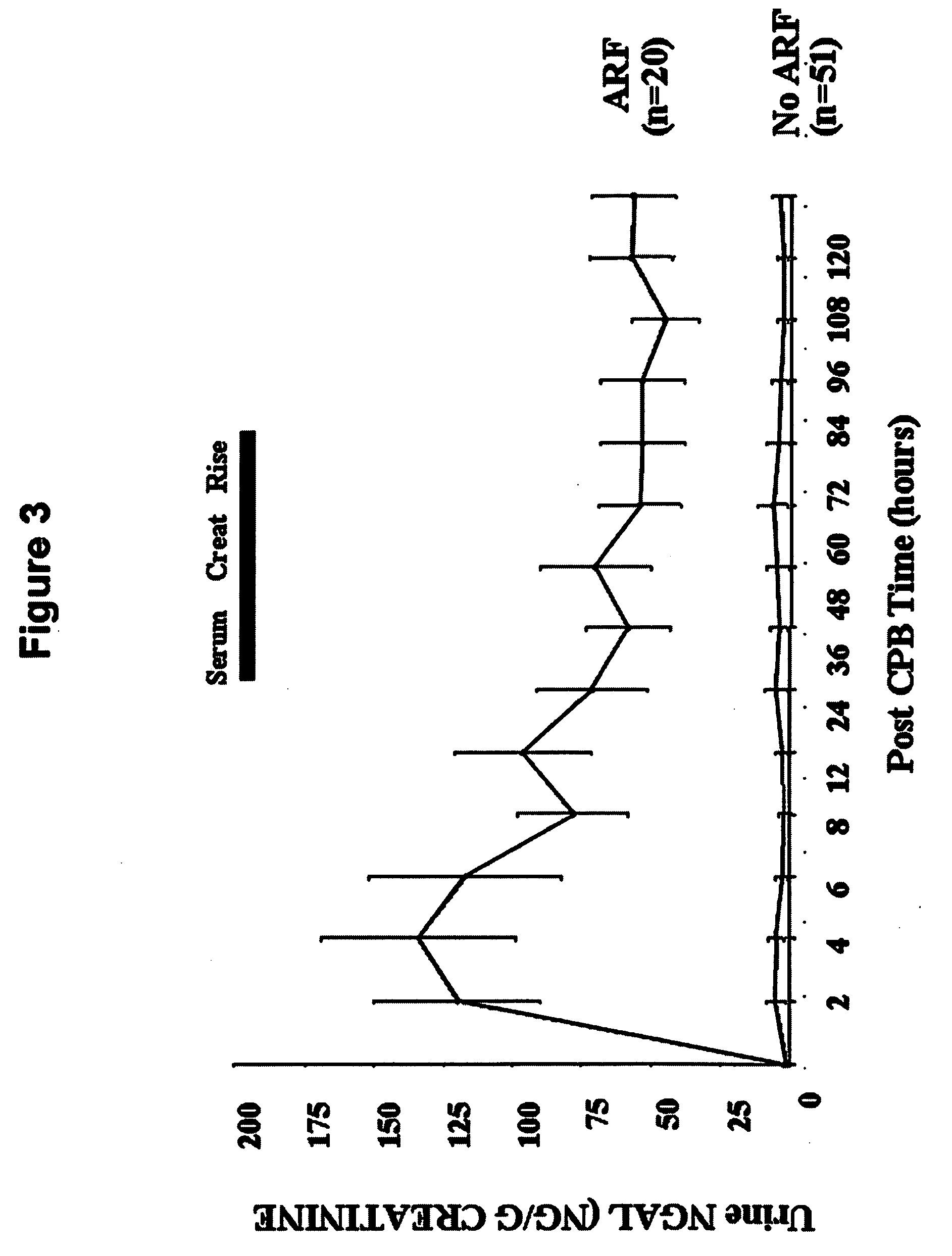
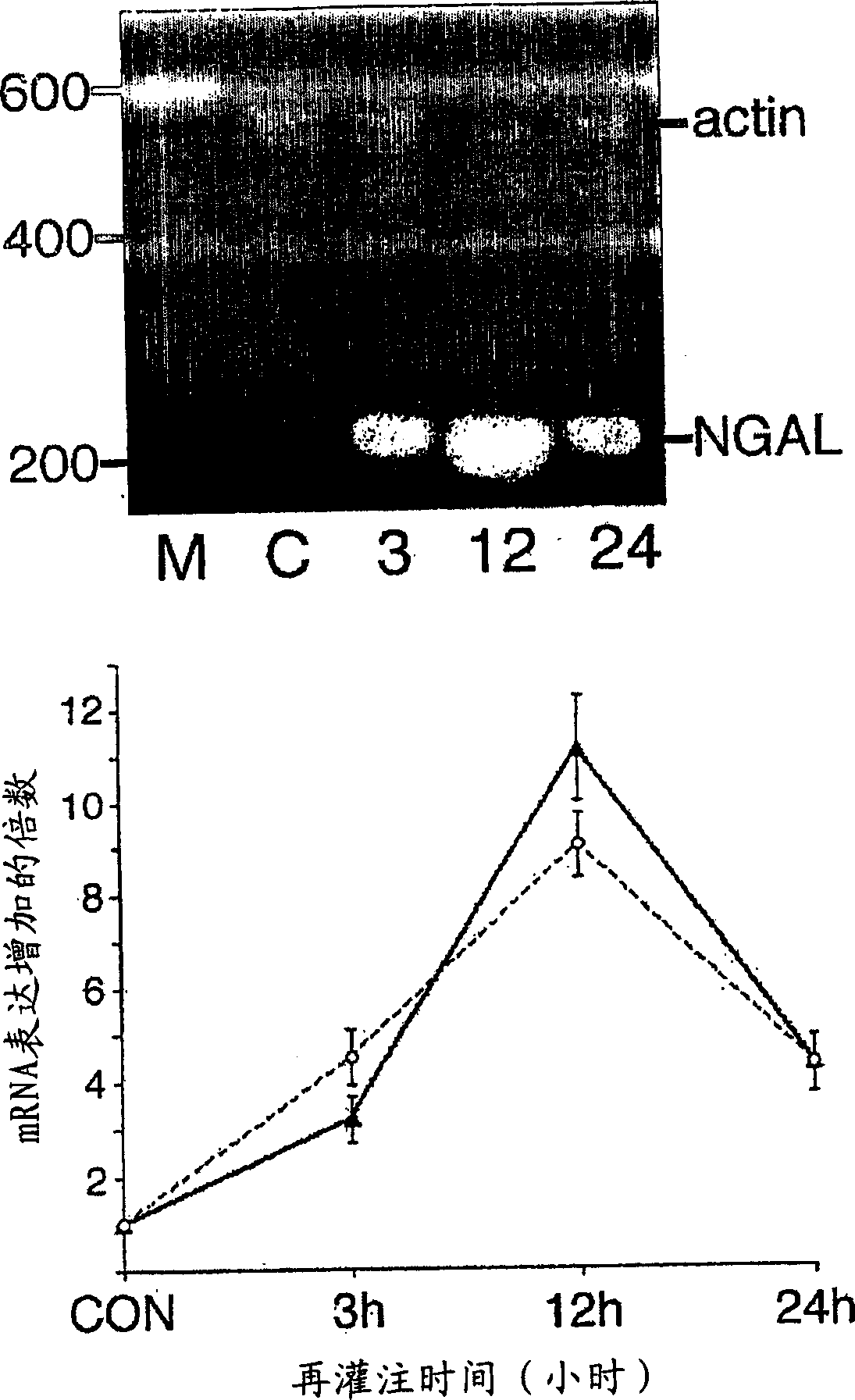
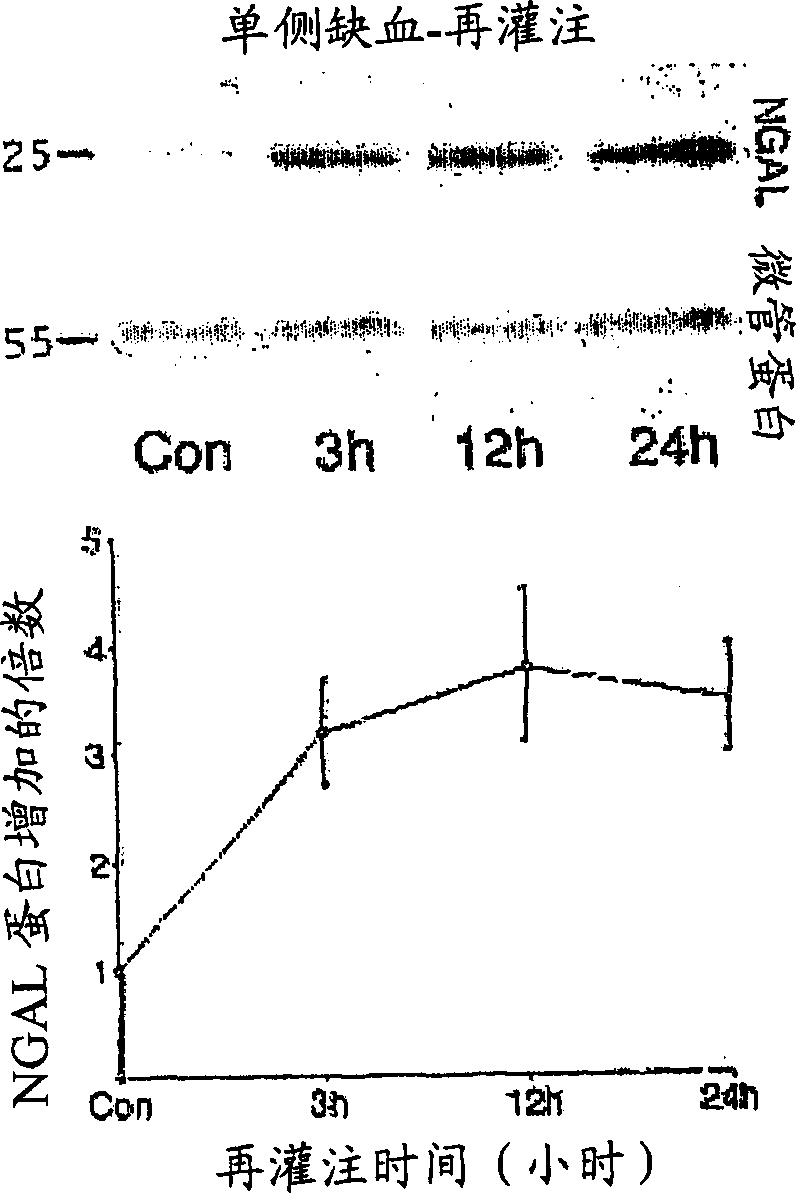
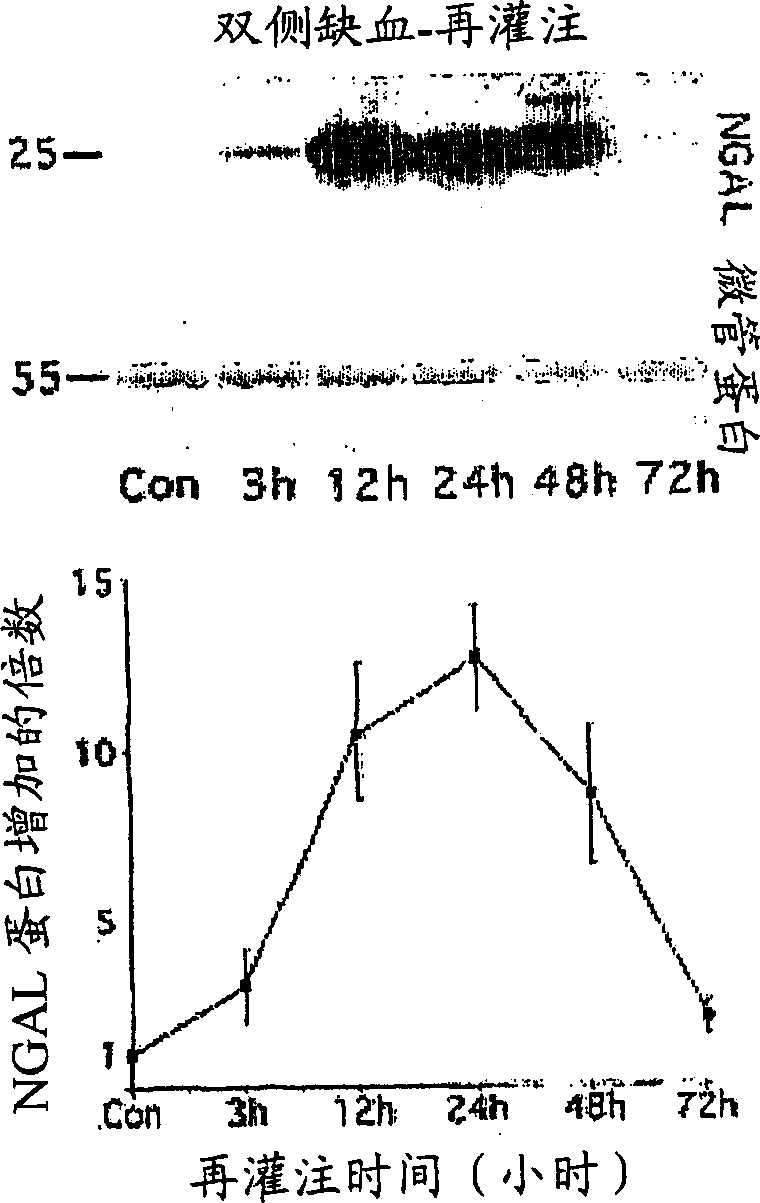
A method and kit for detecting the immediate or early onset of renal disease and injury, including renal tubular cell injury, utilizing NGAL as an immediate or early on-set biomarker in a sample of blood serum. NGAL is a small secreted polypeptide that is protease resistant and consequently readily detected in the blood serum following renal tubule cell injury. NGAL protein expression is detected predominantly in proximal tubule cells, in a punctuate cytoplasmic distribution reminiscent of a secreted protein. The appearance NGAL in the serum is related to the dose and duration of renal ischemia and nephrotoxemia, and is diagnostic of renal tubule cell injury and renal failure. NGAL detection is also a useful marker for monitoring the nephrotoxic side effects of drugs or other therapeutic agents.
Owner:CHILDRENS HOSPITAL MEDICAL CENT CINCINNATI +1
Machine and procedure for extracorporeal treatment of blood
ActiveUS8105260B2Shorten treatment timeIncrease the number ofSemi-permeable membranesOther blood circulation devicesBlood treatmentsBody fluid
A machine (1) for extracorporeal blood treatment comprises a device (2) for ultrafiltration of a liquid through a semi-permeable membrane (3) of a blood treatment device (4), a first sensor (11) for ultrafiltration rate through the membrane, a second group of sensors (13) for measuring a trans-membrane pressure, and a programmed controller (14) for calculating a maximum ultrafiltration rate as a function of the transmembrane pressure. The machine is suitable for kidney failure treatments, such as hemofiltration and hemodiafiltration. In a relatively short time the machine enables a large quantity of corporeal blood to be removed from the patient.
Owner:GAMBRO LUNDIA AB
Bioartificial filtration device for filtering blood to mimic kidney function
InactiveUS6942879B2Long life-timeImmobilised enzymesBioreactor/fermenter combinationsBioartificial liver deviceUltrafiltration
A novel cell seeded hollow fiber bioreactor is described as a potential bioartificial kidney. Endothelial cells along with pericyte, vascular smooth muscle, and / or mesangial cells or any mesenchymally derived support cells are seeded along a hollow fiber in a perfused bioreactor to reproduce the ultrafiltration function and transport function of the kidney. Maintenance of tissue specific function and ultrastructure suggest that this bioreactor provides an economical device for treating renal failure.
Owner:RGT UNIV OF MICHIGAN
Method and composition for treatment of renal failure with antibodies and their equivalents as partial or complete replacement for dialysis
InactiveUS7504106B2Lower Level RequirementsSlow onsetBiocidePeptide/protein ingredientsInterleukin 6Creatinine rise
A method for treating patients with renal failure includes administering to them an effective amount of antibody or of a functional equivalent thereof to at least two of urea, creatinine, tumor necrosis factor alpha, interferon gamma, interleukin 6 and interleukin 1 beta. Soluble cytokine receptors also can be employed. The method can be used as a supplement to or as partial or complete replacement for dialysis. A pharmaceutical composition includes antibody or functional equivalent thereof to urea, creatinine, or both; antibody, functional equivalent or soluble cytokine receptor to tumor necrosis factor alpha, interferon gamma, interleukin 6, interleukin 1 beta or any combination thereof The composition can be included in a kit.
Owner:SKURKOVICH BORIS +2
A method and kit for detecting the early onset of renal tubular cell injury
An early-onset method and kit for detecting renal tubular cell injury, using NGAL as an early urinary biomarker. NGAL is a small secreted polypeptide that is protease resistant and thus readily detected in urine following tubular cell injury. NGAL protein expression was mainly detected in the proximal tubule cells, which resembled a secreted protein with a punctate distribution in the cytoplasm. Apparent NGAL in urine correlates with the amount and duration of renal ischemia and nephrotoxicemia and is a diagnostic feature of tubular cell injury and renal failure. NGAL detection is also a useful marker for monitoring nephrotoxic side effects of drugs or other therapeutic agents.
Owner:CHILDRENS HOSPITAL MEDICAL CENT CINCINNATI +1
Chronic access system for extracorporeal treatment of blood including a continously wearable hemodialyzer
ActiveUS8109893B2More convenientEliminates the need for time-consuming, sedentary, hospital or clinic dialysisSemi-permeable membranesSolvent extractionIntravenous cannulaHaemodialysis machine
Owner:LANDE ARNOLD J
Ion sensor for long term use in complex medium
Devices and methods for measuring a target ion concentration uses an electrode pair. The pair includes a working electrode and a reference electrode. The working and reference electrodes are ion-selective electrodes (ISEs). The reference ISE can include a sodium ISE. The ISE pair interacts with body fluids where a target ion concentration changes more than sodium ion concentration over time. Some ISE membranes of a pair vary essentially only in the ionophore. An ISE pair can determine the ratio of a target ion concentration to sodium ion concentration in vivo. Periodic measurement of sodium concentration in drawn blood can be used to calibrate an ISE pair and provide target ion concentration as an output. Or, a potassium / sodium ISE pair beneficially monitors potassium concentration changes over time in heart- or kidney-failure patients. Then, manual or automatic titration of a diuretic material can be implemented to maintain a desired potassium concentration.
Owner:MEDTRONIC INC
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
ActiveUS20110201038A1Easy to adaptMicrobiological testing/measurementDisease diagnosisInterleukin 10Soluble P-Selectin
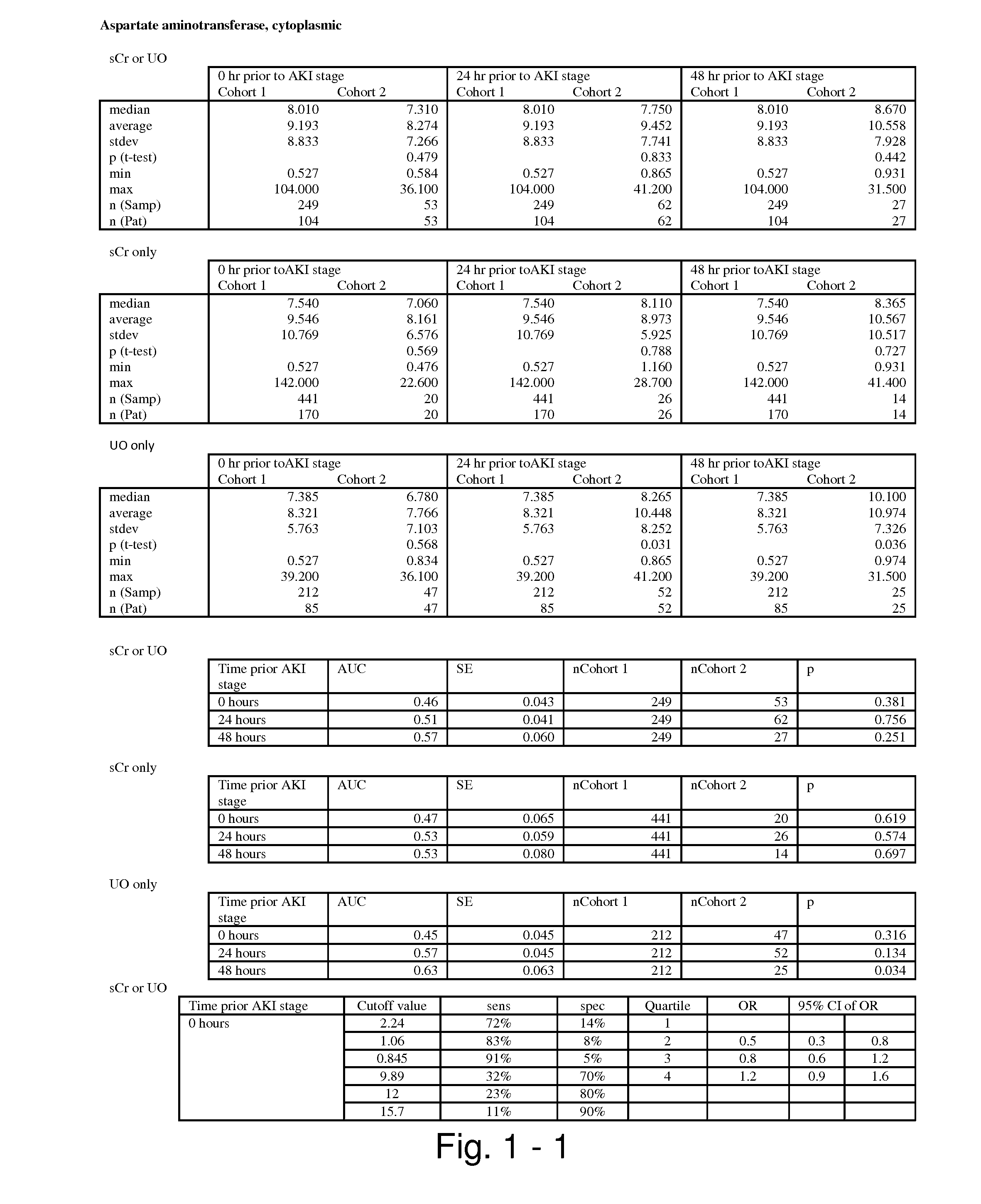
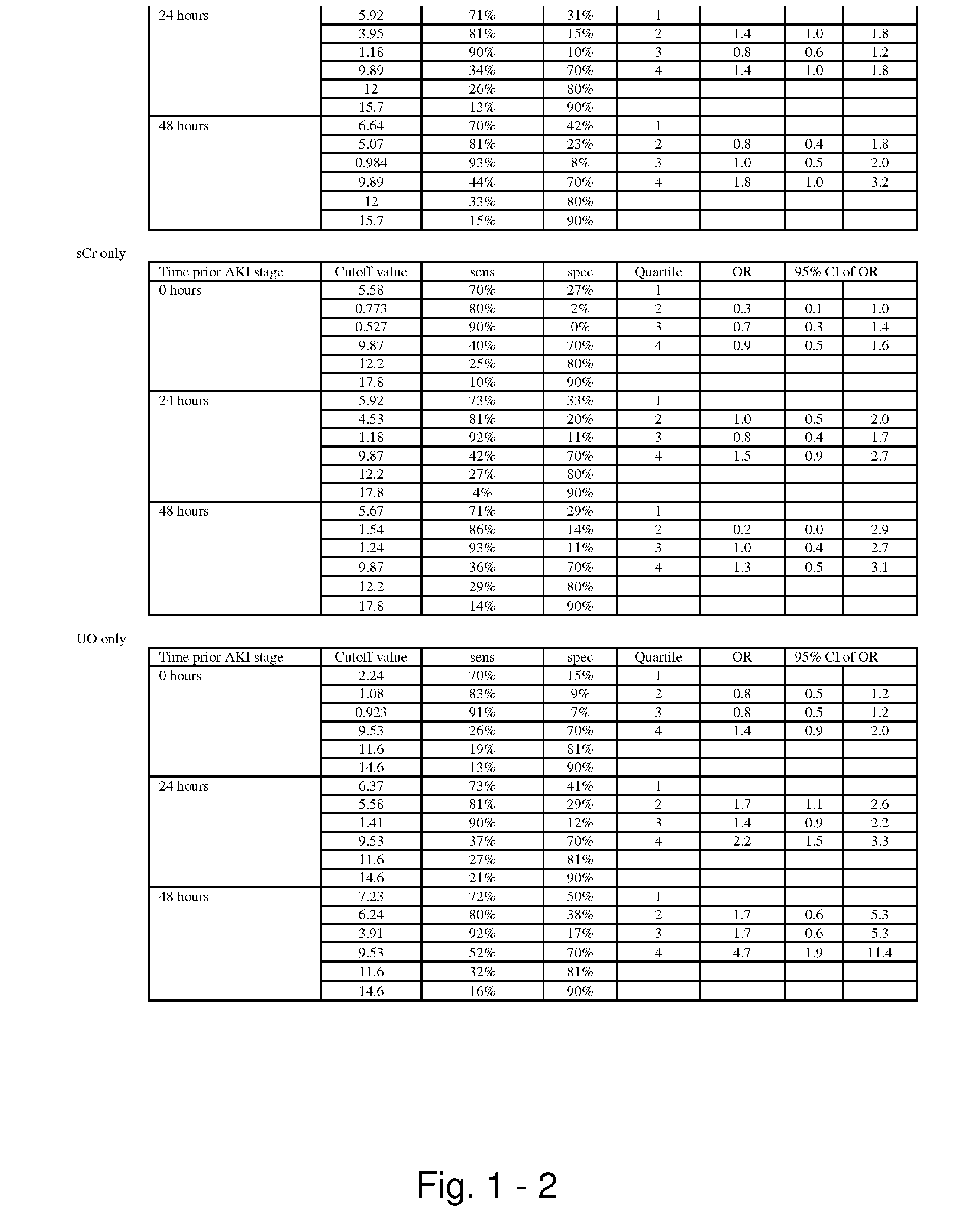
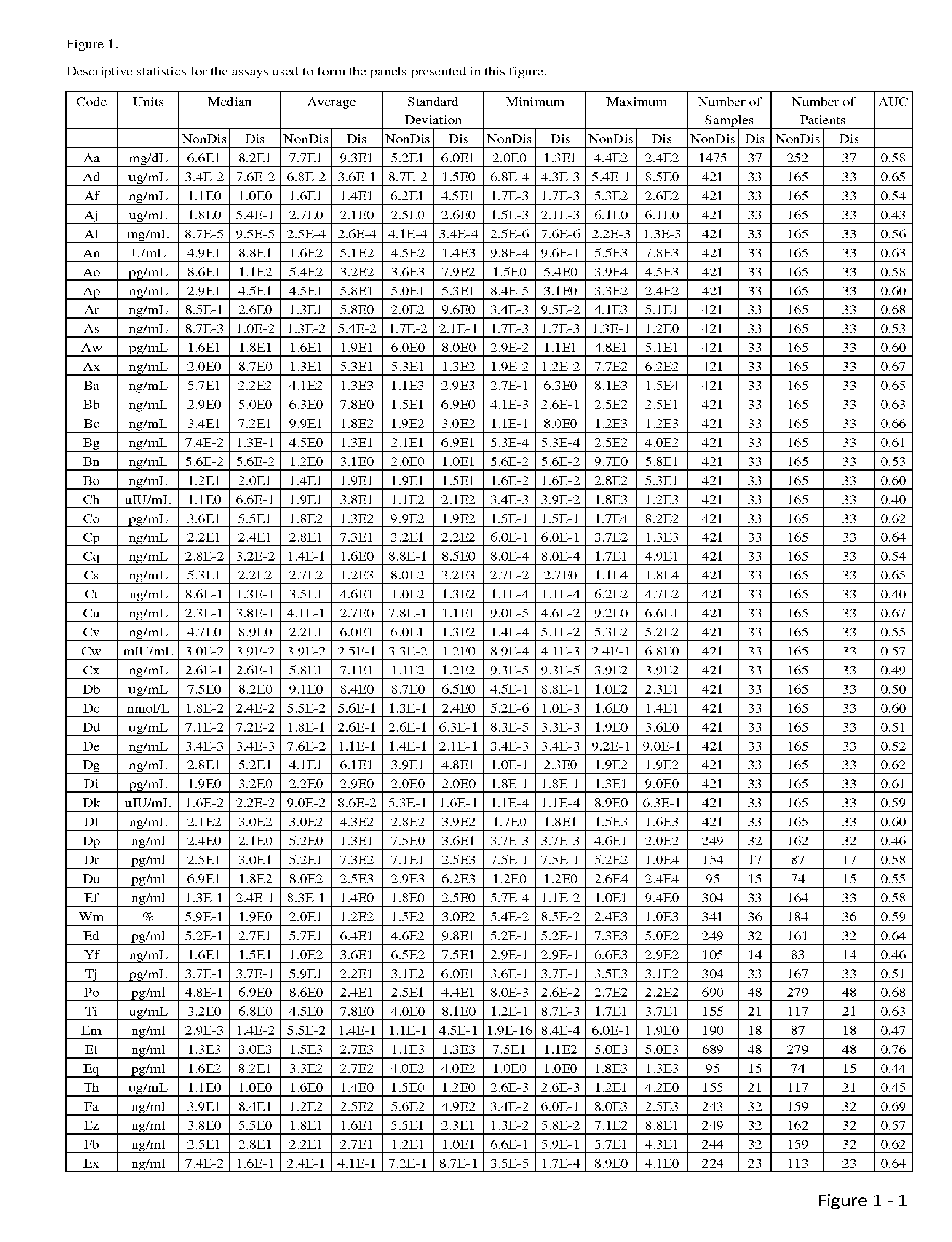
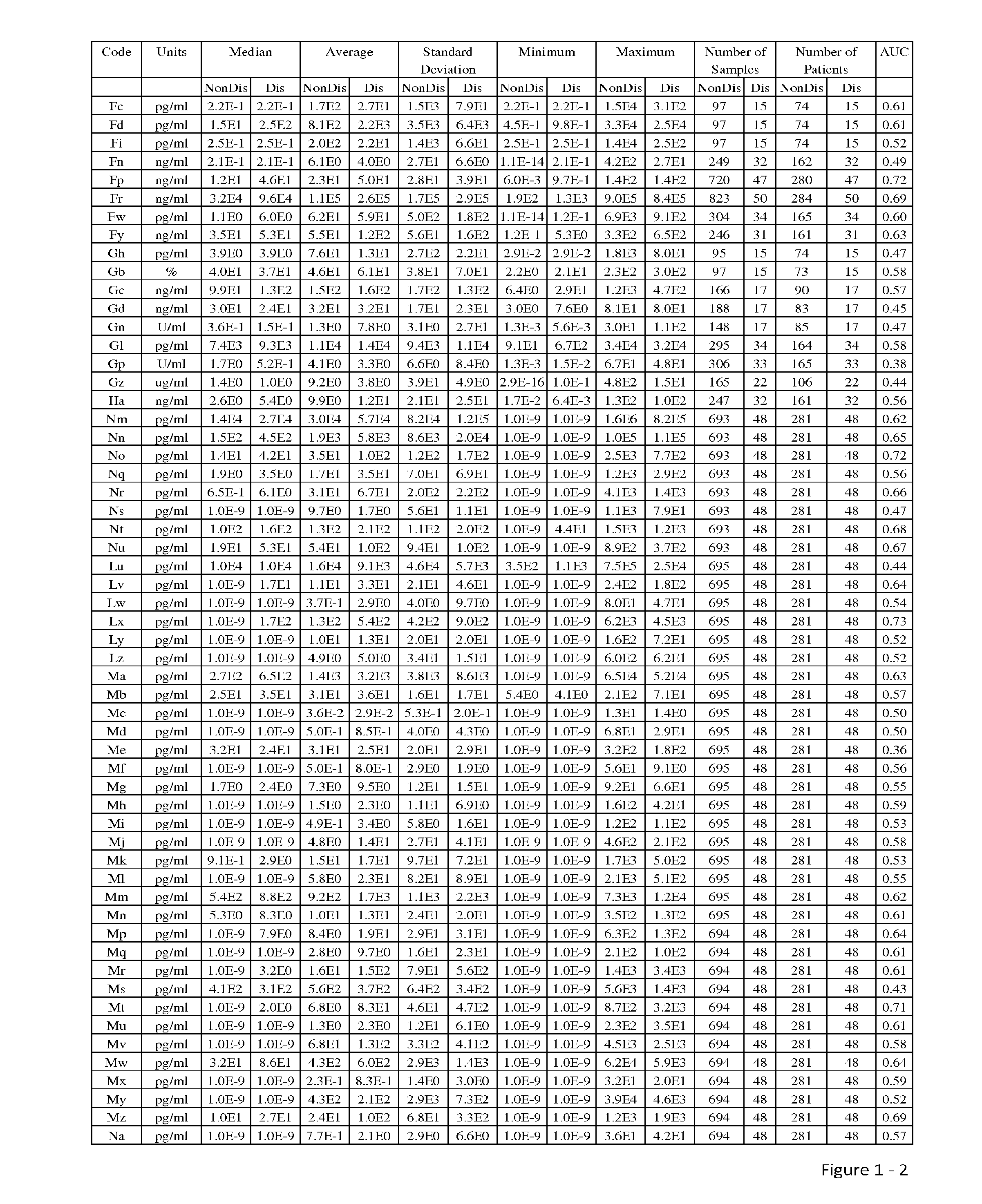
The present invention relates to methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in subjects suffering from or suspected of having a renal injury. In particular, the invention relates to using assays that detect one or more markers selected from the group consisting of Cytoplasmic aspartate aminotransferase, soluble Tumor necrosis factor receptor superfamily member 5, soluble CD40 Ligand, soluble C-X-C Motif chemokine 16, S100-A12, Eotaxin, soluble E-selectin, Fibronectin, Granulocyte colony-stimulating factor, Granulocyte-macrophage colony-stimulating factor, Heparin-binding growth factor 2, soluble Hepatocyte growth factor receptor, Interleukin-1 receptor antagonist, Interleukin-1 beta, Interleukin-10, Interleukin-15, Interleukin-3, Myeloperoxidase, Nidogen-1, soluble Oxidized low-density lipoprotein receptor 1, Pappalysin-1, soluble P-selectin glycoprotein ligand 1, Antileukoproteinase, soluble Kit ligand, Tissue inhibitor of metalloproteinase 1, Tissue inhibitor of metalloproteinase 2, soluble Tumor necrosis factor, soluble Vascular cell adhesion molecule 1, and Vascular endothelial growth factor A as diagnostic and prognostic biomarkers in renal injuries.
Owner:ASTUTE MEDICAL
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
InactiveUS20120190044A1Eliminate needBioreactor/fermenter combinationsBiological substance pretreatmentsGlycoproteinMetalloproteinase inhibitor
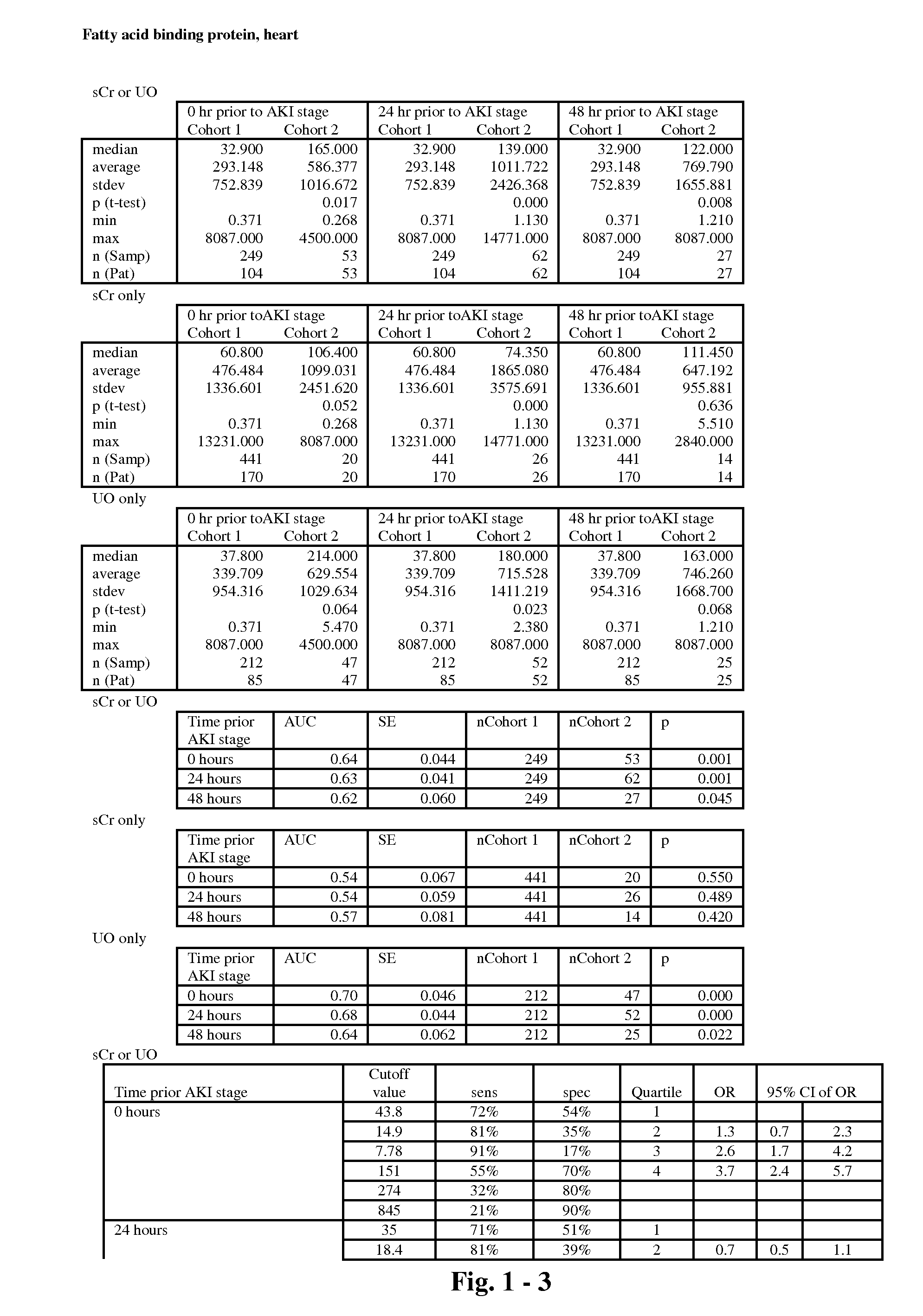
The present invention relates to methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in subjects suffering from or suspected of having a renal injury. In particular, the invention relates to using a plurality of assays, one or more of which is configured to detect a kidney injury marker selected from the group consisting of metalloproteinase inhibitor 2, soluble oxidized low-density lipoprotein receptor 1, interleukin-2, von Willebrand factor, granulocyte-macrophage colony-stimulating factor, tumor necrosis factor receptor superfamily member 11B, neutrophil elastase, interleukin-1 beta, heart-type fatty acid-binding protein, beta-2-glycoprotein 1, soluble CD40 ligand, coagulation factor VII, C—C motif chemokine 2, IgM, CA 19-9, IL-10, TNF-α, and myoglobin as diagnostic and prognostic biomarkers in renal injuries.
Owner:ASTUTE MEDICAL
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
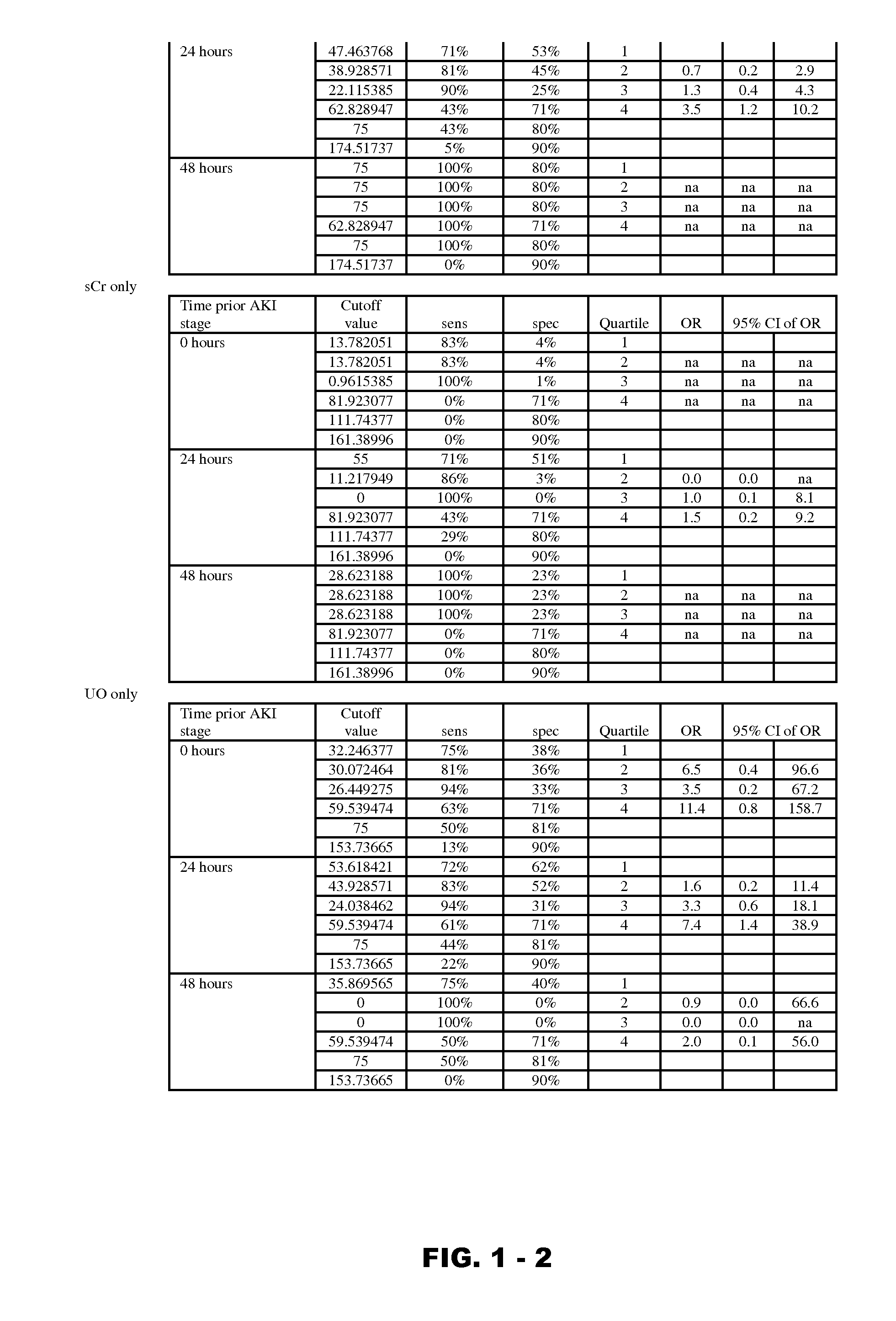
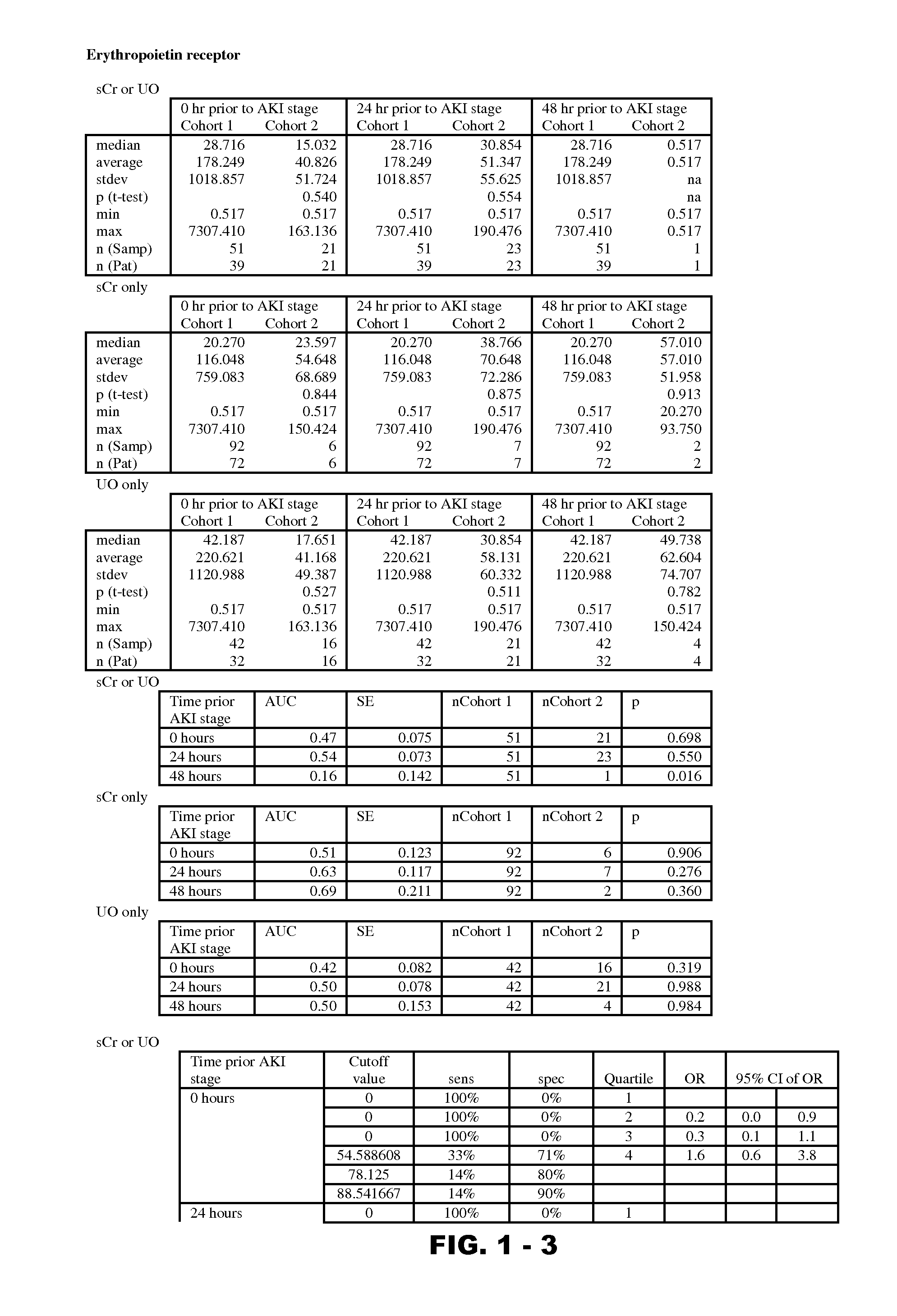
InactiveUS20120190051A1Eliminate needPeptide/protein ingredientsTransferrinsErythropoietin receptorFactor VIII vWF
Disclosed are methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in subjects suffering from or suspected of having a renal injury. In particular, disclosed are assays that detect one or more markers selected from the group consisting of Prostatic acid phosphatase, Lactotransfenin, Soluble erythropoietin receptor, Von Willebrand factor, Soluble endothelial protein C receptor, and Beta-2-glycoprotein 1 as diagnostic and prognostic biomarkers in renal injuries.
Owner:ASTUTE MEDICAL
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
InactiveUS20130210041A1Disease diagnosisColor/spectral properties measurementsRenal FailuresRenal injury
The present invention relates to methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in subjects suffering from or suspected of having a renal injury. In particular, the invention relates to using a plurality of assays configured to detect a kidney injury marker as diagnostic and prognostic biomarkers in renal injuries.
Owner:ASTUTE MEDICAL
Treatment for renal failure
InactiveUS20140058372A1Ultrasound therapySpinal electrodesProphylactic treatmentContrast-induced nephropathy
A method of increasing renal function in a patient operates by stimulation of perivascular sympathetic nerves found in the vicinity of the hepatic portal vein and the hepatic artery. The method can be used as a treatment for renal failure or chronic kidney disease. Alternatively, the method can be used as a prophylactic treatment for preventing contrast-induced nephropathy or any other toxic nephropathy, which can result in renal failure. The perivascular sympathetic nerves can be stimulated by applying energy, such as electrical energy, light, vibration, and ultrasonic vibration, to the perivascular sympathetic nerves. Various methods are described for stimulating the perivascular sympathetic nerves using electrodes that are placed using minimally-invasive techniques.
Owner:SUPERRENAL
Cyclohexane analogues as gpr119 agonists
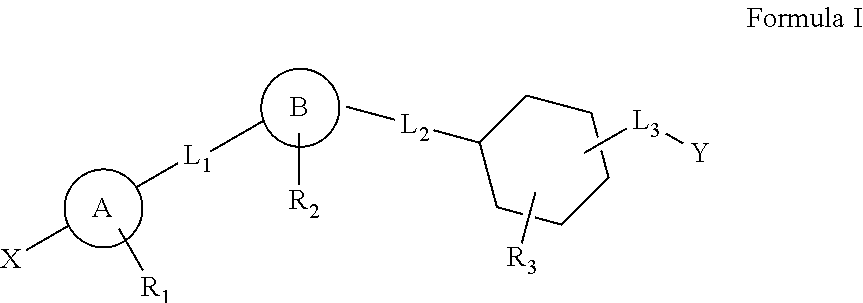
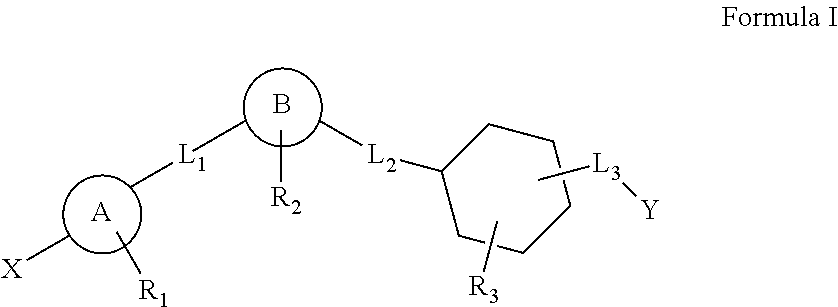
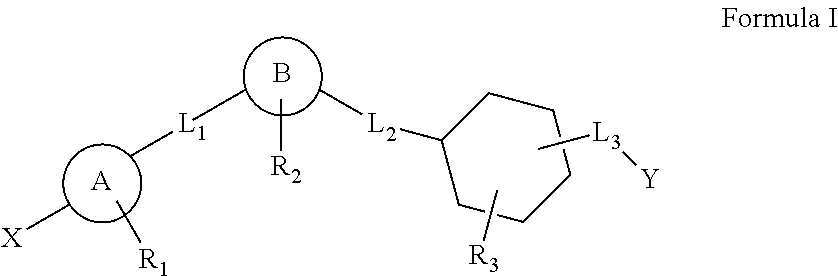
This invention relates to a series of substituted cyclohexane containing analogues which are agonists of GPR119 intended to treat metabolic diseases mediated by GPR119 including Type I & II diabetes mellitus. Diabetes mellitus is an ever-increasing threat to human health causing various complications (blindness, kidney failure, neuropathy, heart attack, stroke, etc.). Recently it was found that activation of GPR119 which is highly expressed in pancreatic beta cells causes glucose dependent insulin secretion and GLP-1 release. Many pharmaceuticals are currently developing GPR119 agonists and herein we disclose alternative GPR119 agonists. Our invention describes GPR119 agonists having structural Formula (I), pharmaceutically acceptable salt or solvate of Formula (I), isomer or prodrug of Formula (I), and combination therapy of Formula (I) with other anti-diabetic drugs like DPP-IV inhibitors and / or insulin sensitizers.
Owner:THE ASAN FOUND
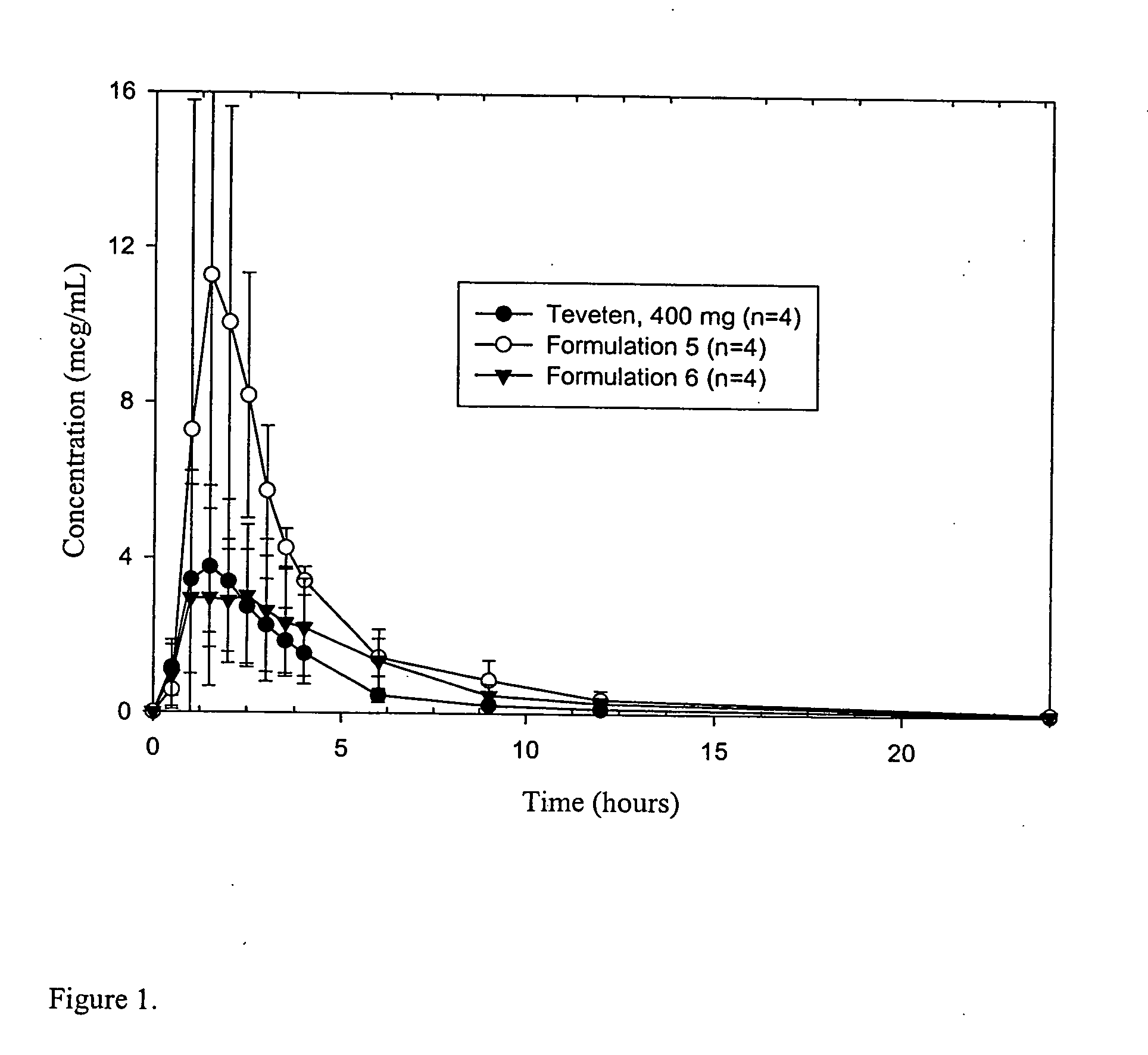
Novel formulations of eprosartan with enhanced bioavailability
InactiveUS20060099230A1Improve oral bioavailabilityBiocideOrganic active ingredientsAngiotensin receptorRenal Failures
The invention relates to the composition of a novel formulation of eprosartan with enhanced bioavailability. The formulation comprises of eprosartan or a salt, solvate, or hydrate thereof, a solubilizer, and an emulsifier. A process for manufacturing, and methods of using the formulation to block angiotensin II receptors and to treat hypertension, congestive heart failure and renal failure are also provided.
Owner:CHIANG CHIN CHIH
Methods for using JNK inhibitors for treating or preventing disease-related wasting
InactiveUS20040034084A1Inhibit progressPrevent worseningAntibacterial agentsBiocideChronic inflammatory diseaseKidney Failures
The present invention relates to methods useful for the treatment or prevention of disease-related wasting. The methods of the invention comprise the administration of an effective amount of a JNK Inhibitor. In one embodiment, the disease is HIV, AIDS, cancer, end-stage renal disease, kidney failure, chronic heart disease, obstructive pulmonary disease or tuberculosis. The methods can further comprise the administration of a therapeutic or prophylactic agent useful for the treatment or prevention of HIV, AIDS, cancer, end-stage renal disease, kidney failure, chronic heart disease, obstructive pulmonary disease, chronic infectious diseases (e.g., osteoarthritis and bacterial endocarditis), chronic inflammatory diseases (e.g., scleroderma and mixed connective tissue disease) or tuberculosis.
Owner:CELGENE CORP
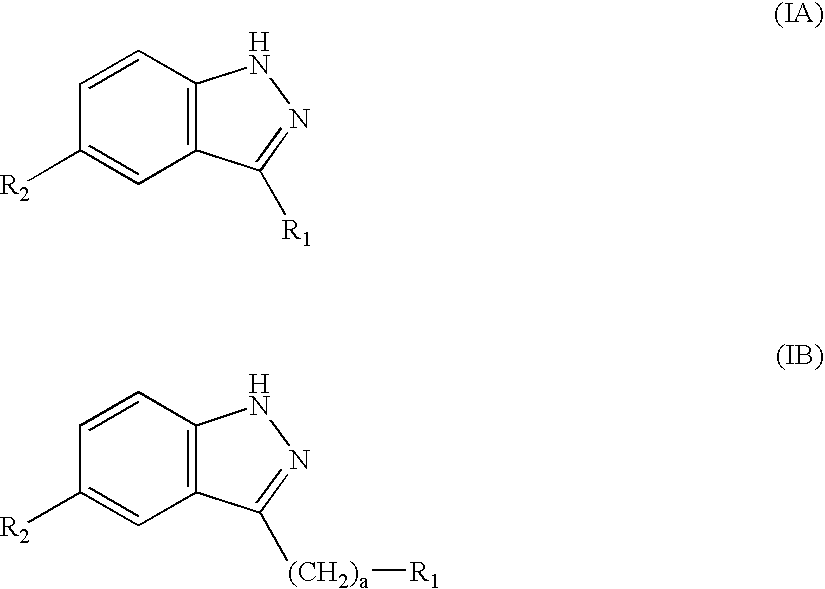
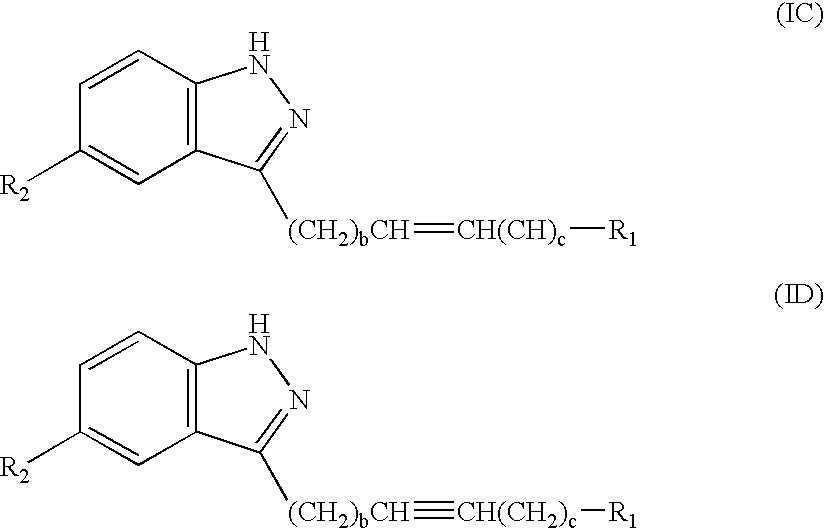
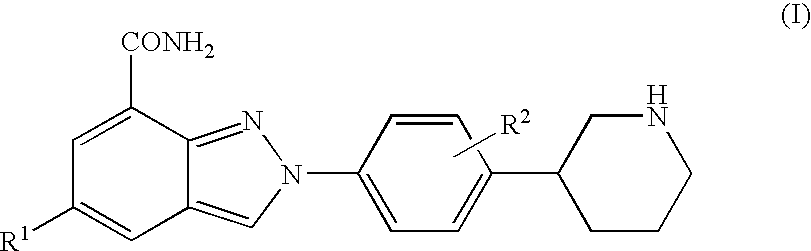
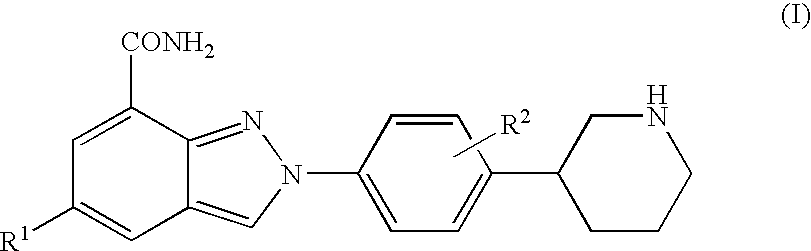
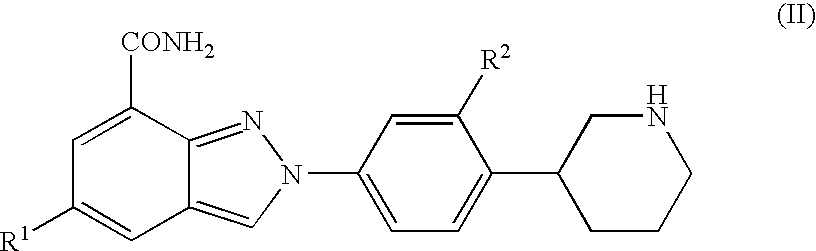
Amide substituted indazoles as poly(ADP-ribose)polymerase(PARP) inhibitors
The present invention relates to compounds of formula I:and pharmaceutically acceptable salts, stereoisomers or tautomers thereof which are inhibitors of poly (ADP-ribose) polymerase (PARP) and thus useful for the treatment of cancer, inflammatory diseases, reperfusion injuries, ischemic conditions, stroke, renal failure, cardiovascular diseases, vascular diseases other than cardiovascular diseases, diabetes, neurodegenerative diseases, retroviral infection, retinal damage or skin senescence and UV-induced skin damage, and as chemo- and / or radiosensitizers for cancer treatment.
Owner:MERCK SHARP & DOHME LLC
Gdf-15 as biomarker in type 1 diabetes
InactiveUS20110033886A1Bioreactor/fermenter combinationsBiological substance pretreatmentsFAILURE KIDNEYRenal Failures
Owner:ROCHE DIAGNOSTICS OPERATIONS INC
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
The present invention relates to methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in sepsis patients. In particular, the invention relates to using assays that detect one or more biomarkers selected from the group consisting of Insulin-like growth factor-binding protein 7, Beta-2-glycoprotein 1, Metalloproteinase inhibitor 2, Alpha-1 Antitrypsin, Leukocyte elastase, Serum Amyloid P Component, C-X-C motif chemokine 6, Immunoglobulin A, Immunoglobulin G subclass I, C-C motif chemokine 24, Neutrophil collagenase, Cathepsin D, C-X-C motif chemokine 13, Involucrin, Interleukin-6 receptor subunit beta, Hepatocyte Growth Factor, CXCL-1, -2, -3, Immunoglobulin G subclass II, Metalloproteinase inhibitor 4, C-C motif chemokine 18, Matrilysin, C-X-C motif chemokine 11, and Antileukoproteinase as diagnostic and prognostic biomarker assays of renal injury in the sepsis patient.
Owner:ASTUTE MEDICAL
Apparatus and methods for diagnosing renal disorders
ActiveUS20110244584A1Improve accuracyHigh precision diagnosisMaterial thermal conductivityWithdrawing sample devicesRenal disorderNephrosis
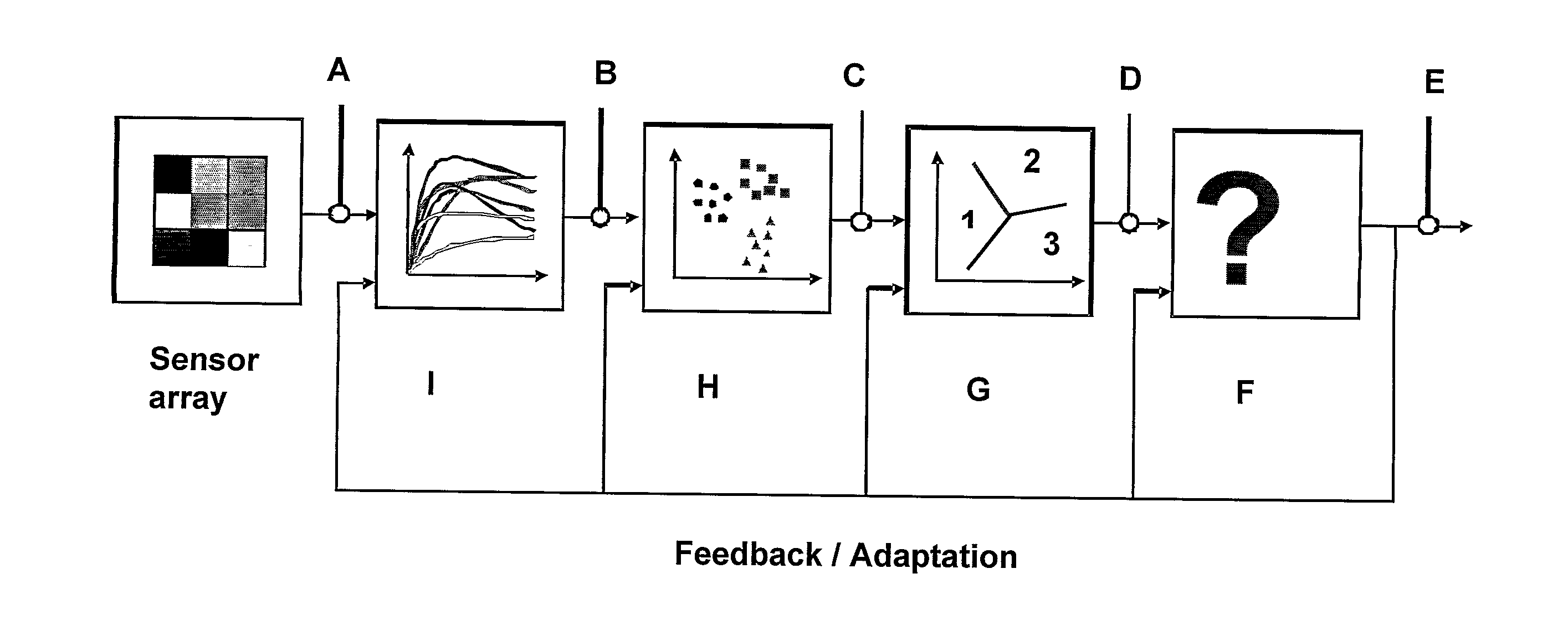
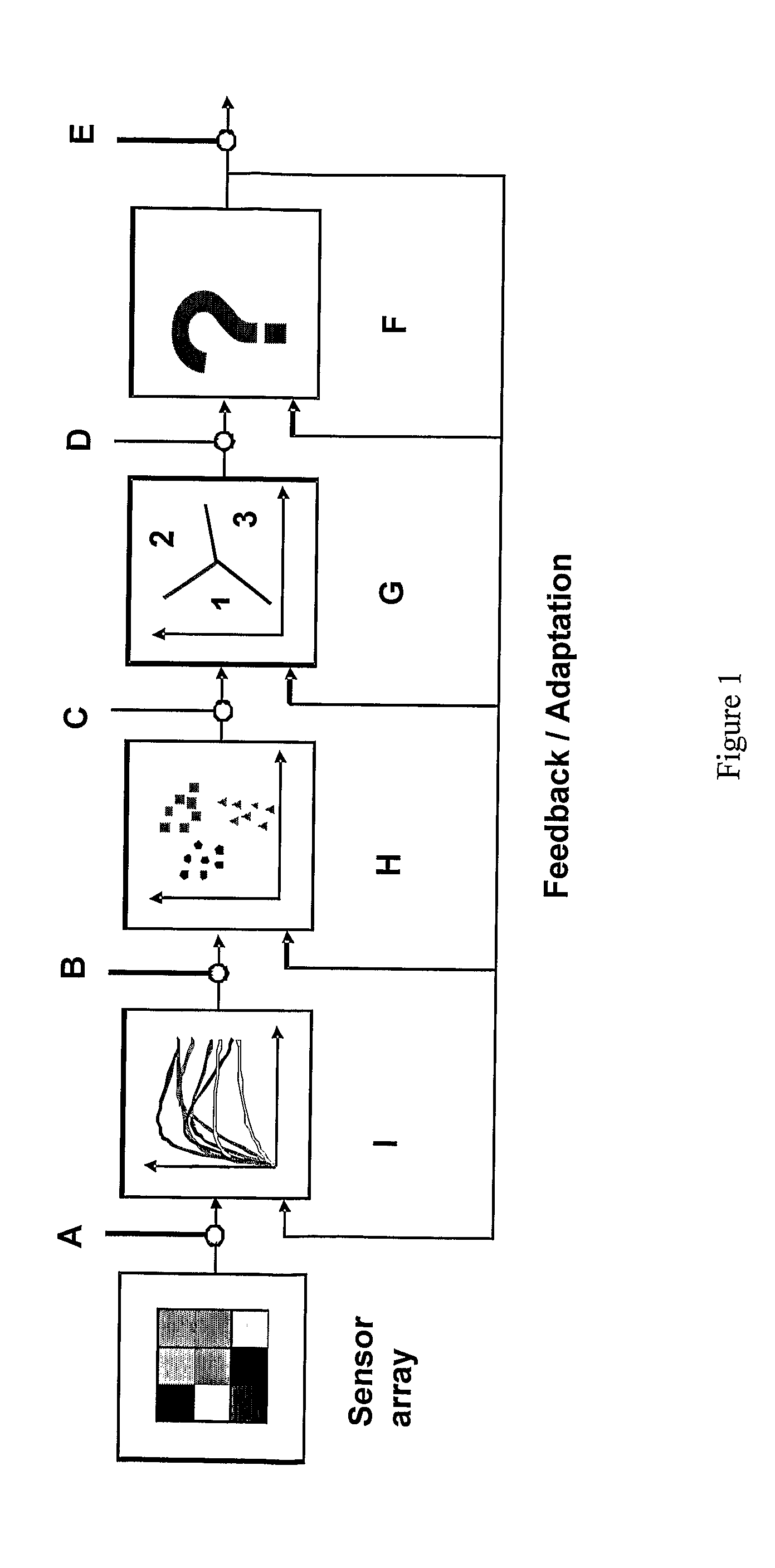
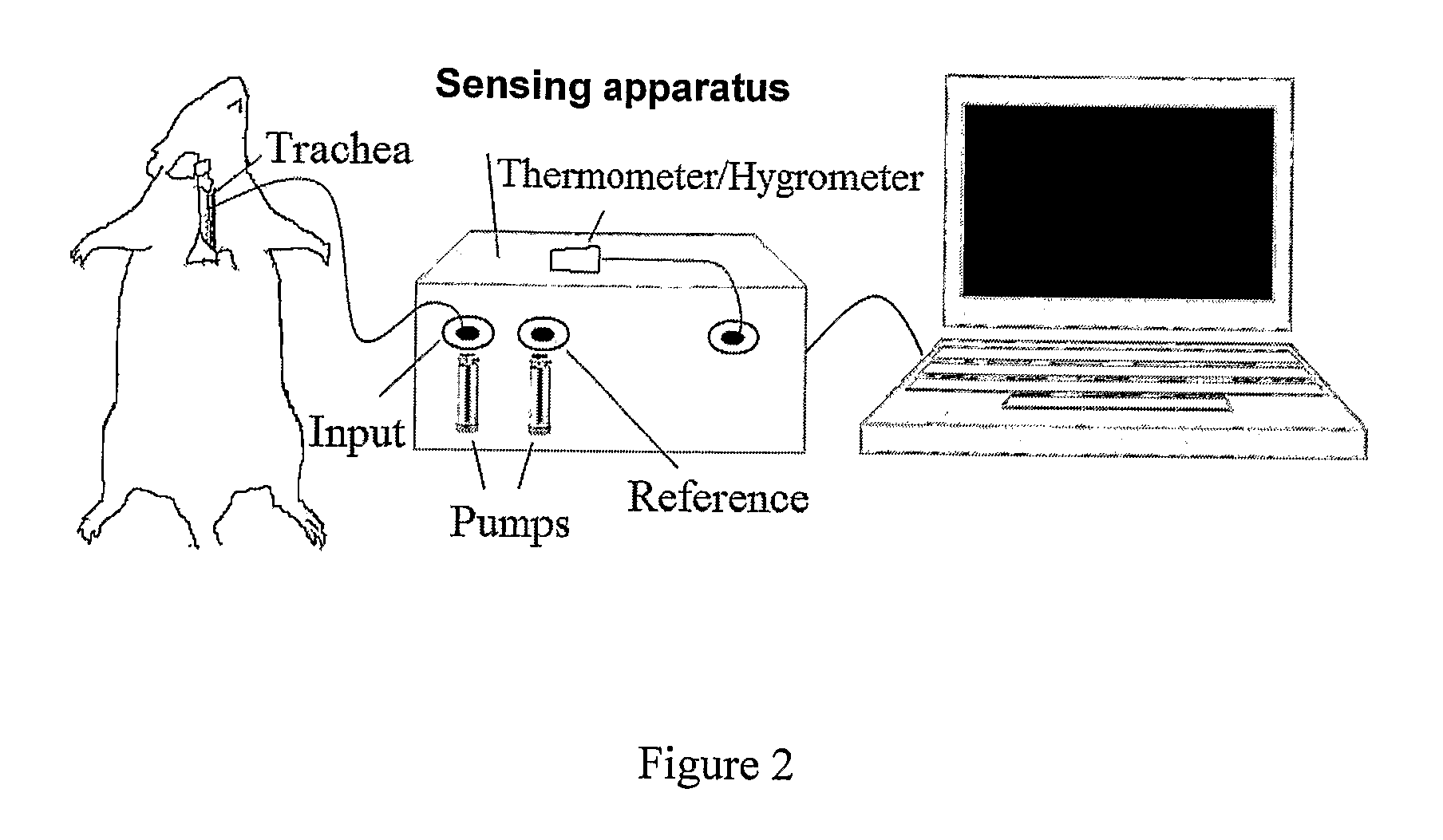
The present invention provides a system comprising an array of chemically sensitive sensors based on coated single walled carbon nanotubes, for measuring volatile organic compounds indicative of renal failure. Methods of breath analysis for diagnosing chronic, acute and end-stage renal failure are disclosed.
Owner:TECHNION RES & DEV FOUND LTD
Treatment of chronic renal failure and other conditions in domestic animals: compositions and methods
InactiveUS20080058250A1BiocideHeavy metal active ingredientsAge related diseaseCombined Modality Therapy
The present invention generally concerns the management of age-related diseases in domestic animals. Specifically, the present invention is directed to combination therapies for the treatment of progressive renal diseases (e.g., Chronic Renal Failure) and their accompanying secondary disease states. In a composition aspect, the present invention provides a composition comprising a phosphate binder and another pharmaceutically active ingredient. The other pharmaceutically active ingredient is selected from a group consisting of antihypertensives, calcitrol, vitamin D analogues, lipid restriction products, potassium salts, treatments for anemia and alkalization compounds.
Owner:SPECTRUM PHARMA INC
Novel therapies for chronic renal failure
InactiveUS20050143304A1Preventing delaying needReducing necessary frequencyOrganic active ingredientsPeptide/protein ingredientsOsteogenic proteinsMorphogenesis
The present invention provides methods for the treatment, and pharmaceuticals for use in the treatment, of mammalian subjects in, or at risk of chronic renal failure, or at risk of a need for renal replacement therapy. The methods involve the administration of certain proteins of, or based upon, the osteogenic protein / bone morphogenetic protein (OP / BMP) family within the TGF-β superfamily of proteins.
Owner:MARIEL THERAPEUTICS
Staple type oligonucleotide and drug comprising the same
ActiveUS20060276421A1Improve in instabilityReduce doOrganic active ingredientsNervous disorderPsoriasisArthritis
Conventional oligonucleotides are opened at both ends and thereby unstable. The stability of them against catabolic enzymes is increased by phosphorothioate modification, but such phosphorothioate causes toxicity. The present invention provides oligonucleotides and medicaments in which these problems are improved. That is, it provides a staple oligonucleotides and medicaments containing the same as the active ingredient. Specifically, it provides transcription factor inhibitors, antisense oligonucleotides and siRNAs. More specifically, it provides agents for preventing, treating or improving inflammation, autoimmune diseases, central diseases, reperfusion injury in ischaemic diseases, worsened prognosis after organ transplantation or organ surgery, or restenosis after PTCA. Further specifically, it provides agents for preventing, treating or improving arthritis, dermatitis, nephritis, hepatitis, renal failure, cystitis, prostatitis, urethritis, ulcerative colitis, Crohn disease, chronic rheumatoid arthritis, osteoarthritis, atopic dermatitis, contact dermatitis, psoriasis, cutaneous ulcer or decubitus.
Owner:ANGES MG INC
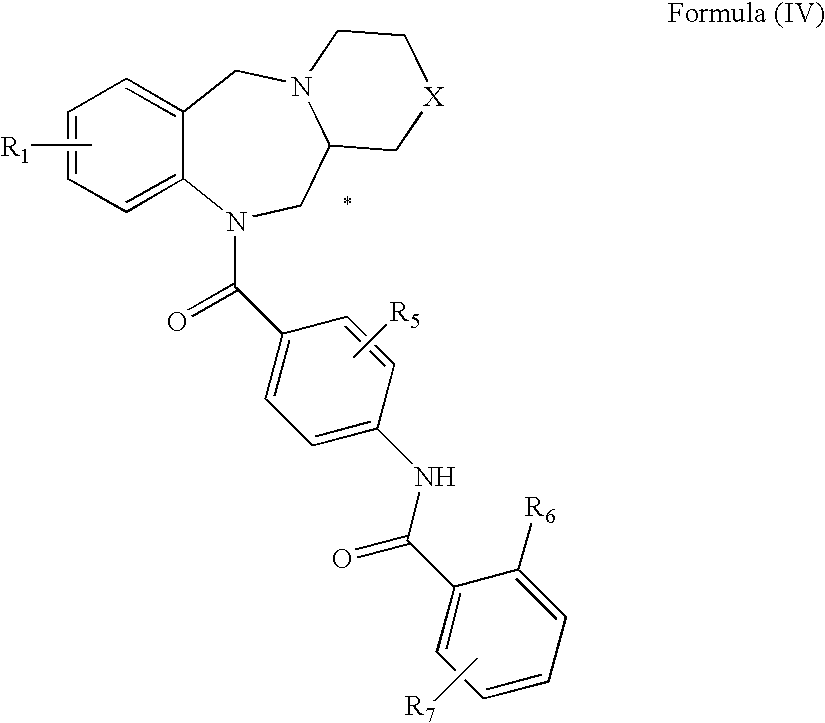
Tricyclic benzodiazepines as vasopressin receptor antagonists
The invention is directed to tricyclic benzodiazepines useful as vasopressin receptor antagonists for treating conditions involving increased vascular resistance and cardiac insufficiency. Pharmaceutical compositions comprising tricyclic benzodiazepines of the present invention and methods of treating conditions such as hypertension, congestive heart failure, cardiac insufficiency, coronary vasospasm, cardiac ischemia, liver cirrhosis, renal vasospasm, renal failure, cerebral edema and ischemia, stroke, thrombosis, or water retention are also disclosed.
Owner:ORTHO MCNEIL PHARM INC
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
InactiveUS9229010B2Peptide/protein ingredientsMammal material medical ingredientsFactor VIII vWFErythropoietin receptor
Disclosed are methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in subjects suffering from or suspected of having a renal injury. In particular, disclosed are assays that detect one or more markers selected from the group consisting of Prostatic acid phosphatase, Lactotransfenin, Soluble erythropoietin receptor, Von Willebrand factor, Soluble endothelial protein C receptor, and Beta-2-glycoprotein 1 as diagnostic and prognostic biomarkers in renal injuries.
Owner:ASTUTE MEDICAL
Methods and compositions for diagnosis and prognosis of renal injury and renal failure
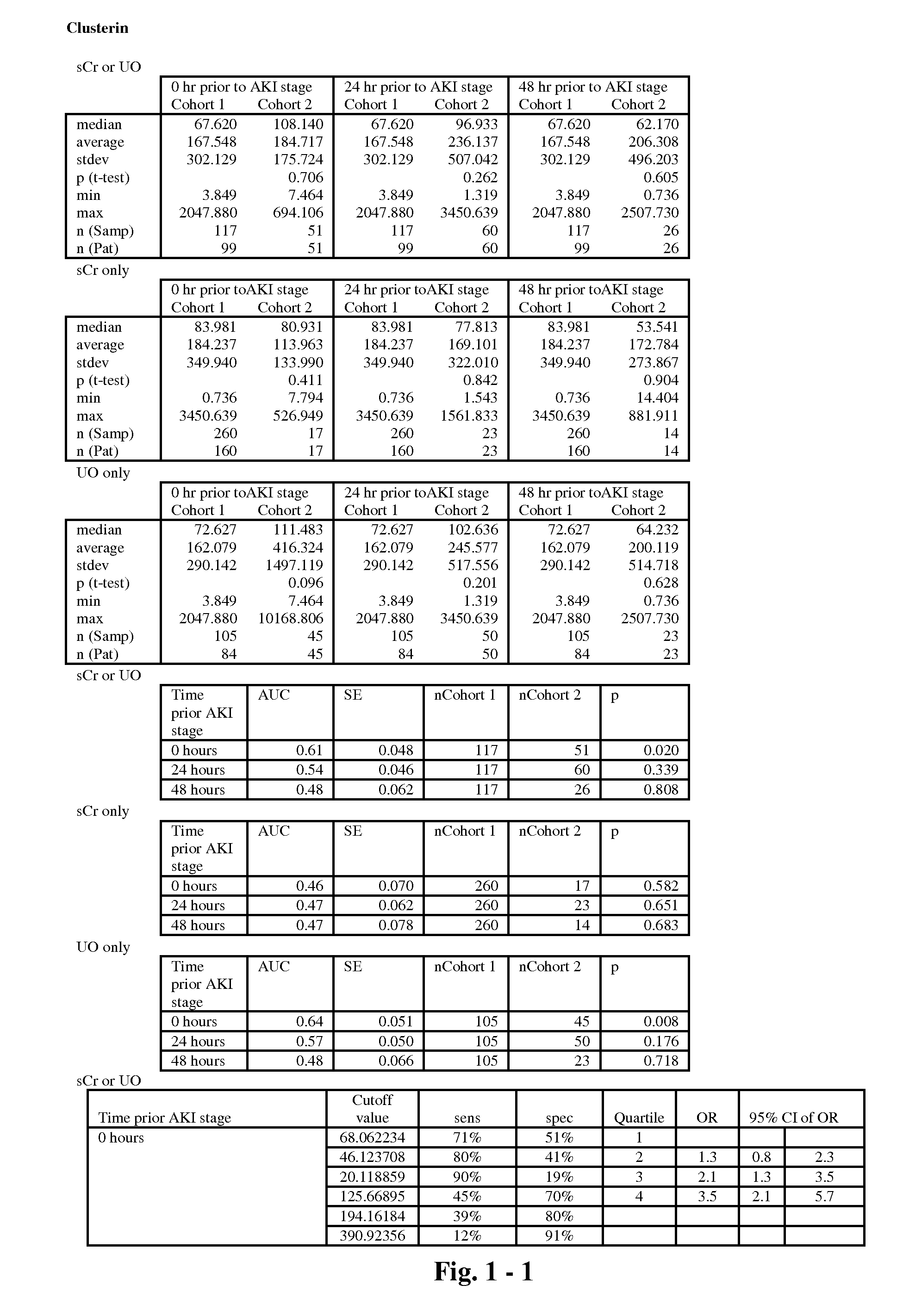
The present invention relates to methods and compositions for monitoring, diagnosis, prognosis, and determination of treatment regimens in subjects suffering from or suspected of having a renal injury. In particular, the invention relates to using assays that detect one or more markers selected from the group consisting of Clusterin, Heart-type fatty acid binding protein, Hepatocyte growth factor, Interferon gamma, Interleukin-12 subunit beta, Interleukin-16, Interleukin-2, 72 kDa type IV collagenase, Matrix metalloproteinase-9, Midkine, and Serum amyloid P-component as diagnostic and prognostic biomarkers in renal injuries.
Owner:ASTUTE MEDICAL
Medicinal composition containing calcium passage paralysor and B family vitamin and its use
ActiveCN1824320AImprove compliancePrevent or delay damageOrganic active ingredientsMetabolism disorderPatient complianceAngina
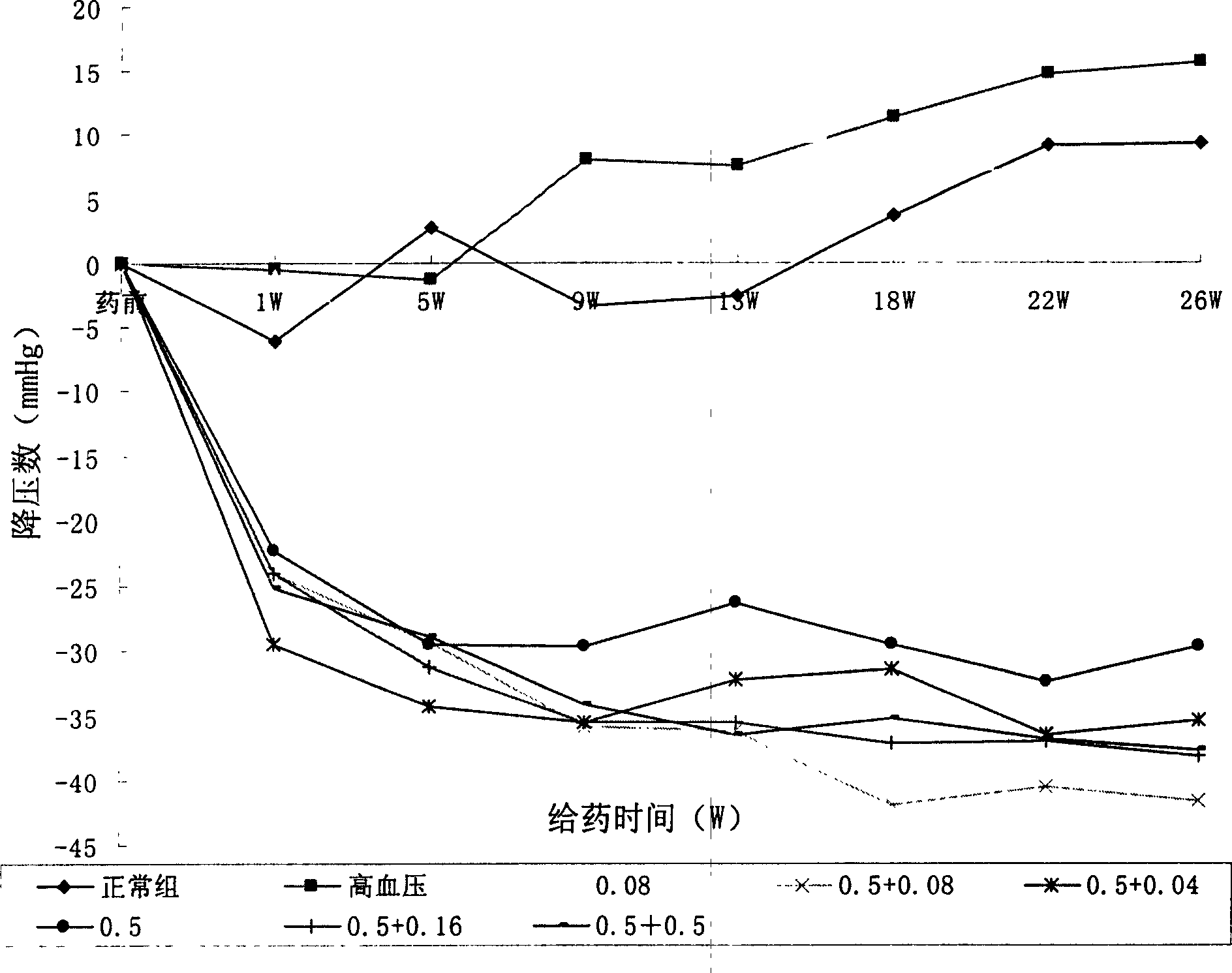
A composite medicine for improving the curative effect of the antihypertensive medicine containing Ca channel retardant and preventing the complications such as eyeground hemorrhage, angina pectoris, myocardial infarction, cerebral apoplexy, heart failure and kidney failure is composed of Ca channel vetardant or its precursor or its active metabolite or its salt, one or more of vitamins in B family and the pharmacologically acceptable carrier.
Owner:SHENZHEN AUSA PHARMA +1