Patents
Literature
65 results about "Chronic patient" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Formulation for sustained delivery
Disclosed is an extended or controlled release dosage form of citalopram or its related forms and other newer antidepressants for oral administration to treat chronic patients suffering from depression and to minimize the side effects associated with the current drug treatment.
Owner:CHALLAPALLI PRASAD V N +2
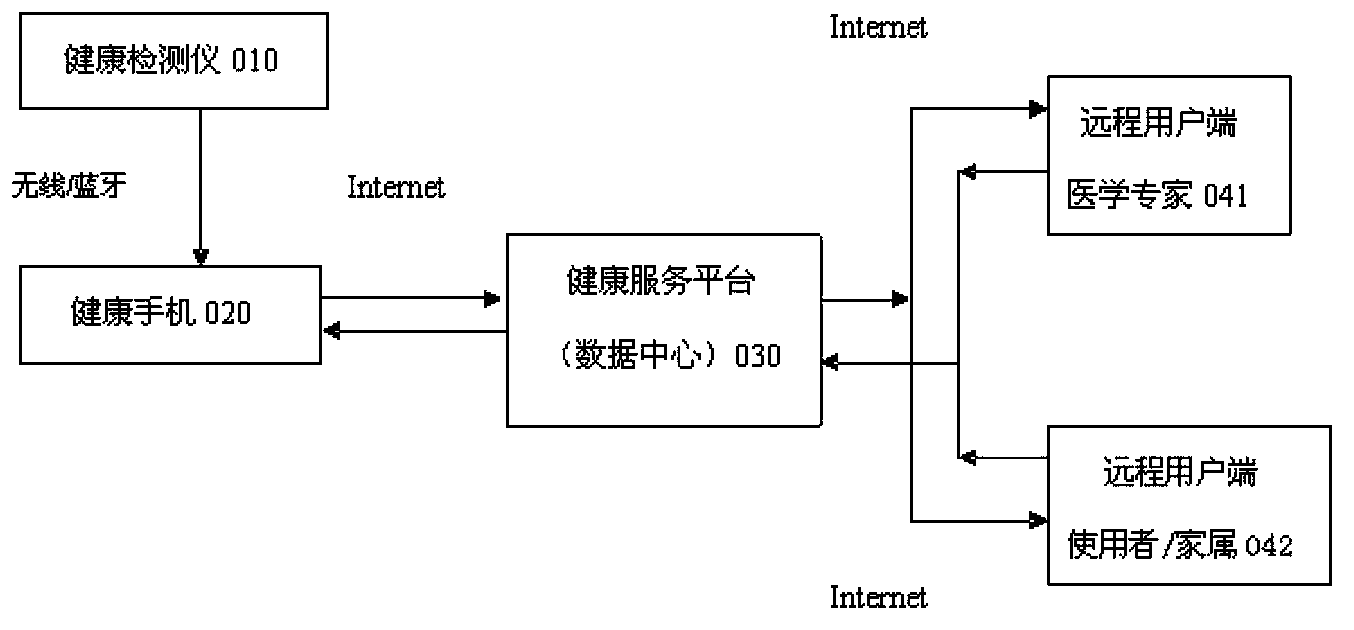
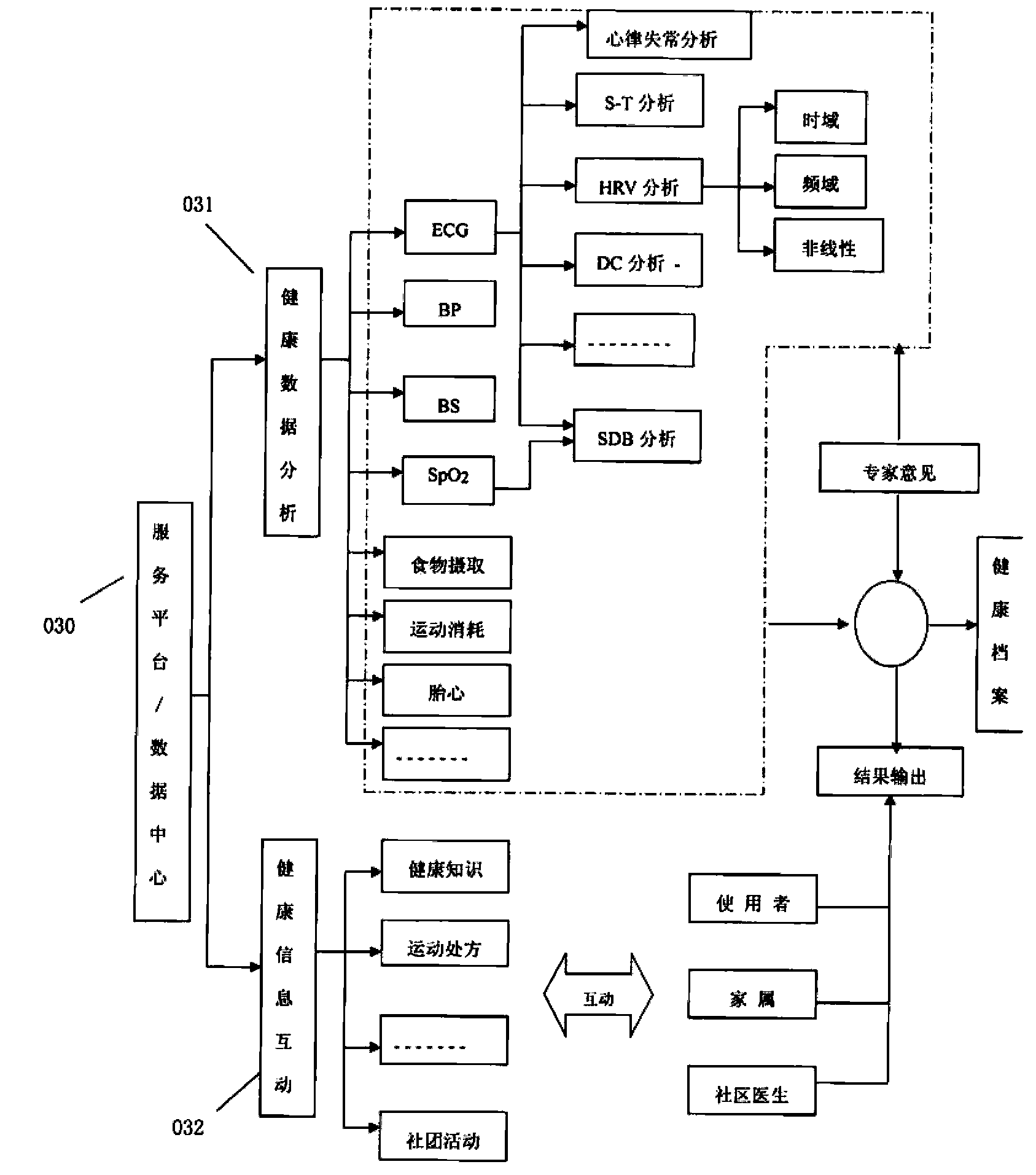
Health service platform system based on real-time monitoring
InactiveCN103400332ATimely medical guidanceHealth care anytime, anywhereData processing applicationsThe InternetHealth profile
The aging of population and the continuous rise in the number of chronic disease crowd have become prominent problems for the present society. A nonprofessional person, however, cannot do anything about the vital sign data detected at home. According to a health service platform system based on real-time monitoring, original vital sign data detected by a health detector is sent to a cellphone on which health monitoring software is preinstalled (health cellphone) in a wireless or Bluetooth manner; the vital sign data is received and displayed by the cellphone, and is transmitted to the health service platform through the Internet; a result is created through analysis and processing, and a personal health profile is created; the analysis result and a health hint are fed back to the health cellphone; a professional instruction can be given timely by a remote client (doctor) through logging in the health service platform; health management information can also be acquired by a user or a relative through logging in the health service platform. The personal health profile serves as a basis for health tendency analysis, and can judge health tendency timely through dynamic analysis of historical data, so as to assist in preventing the occurrences of major illness and emergency illness.
Owner:宁波天瑞医疗器械有限公司
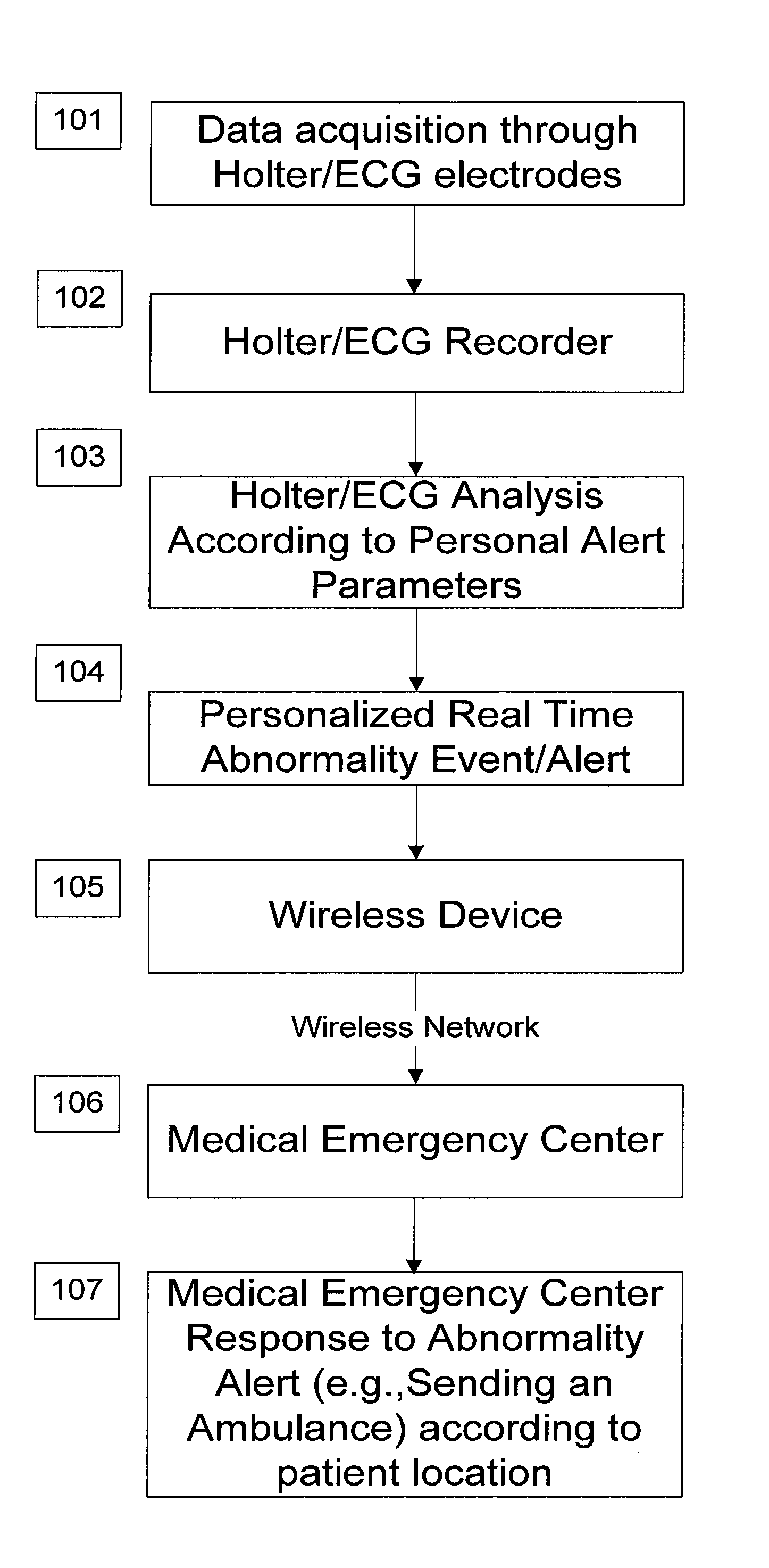
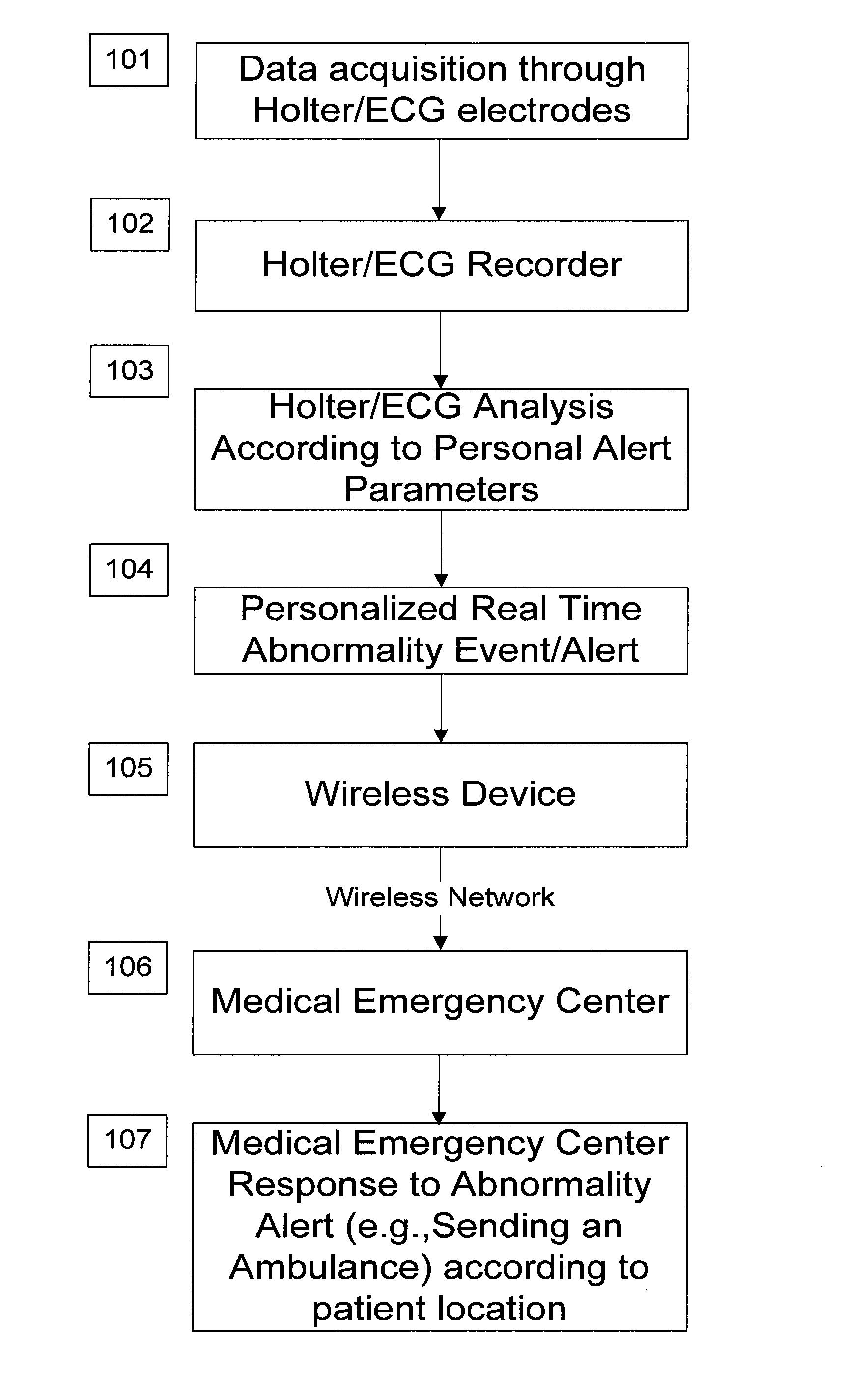
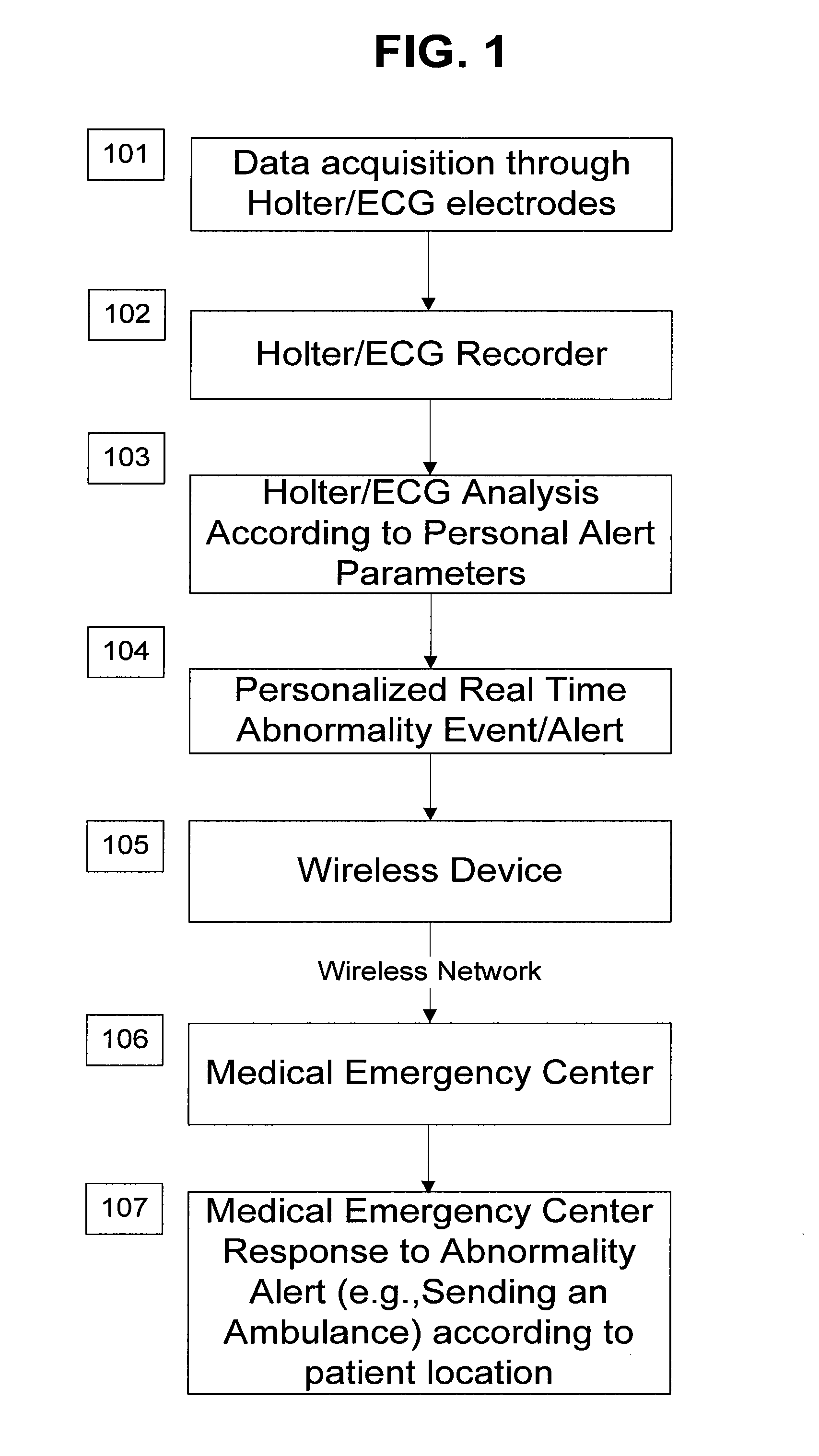
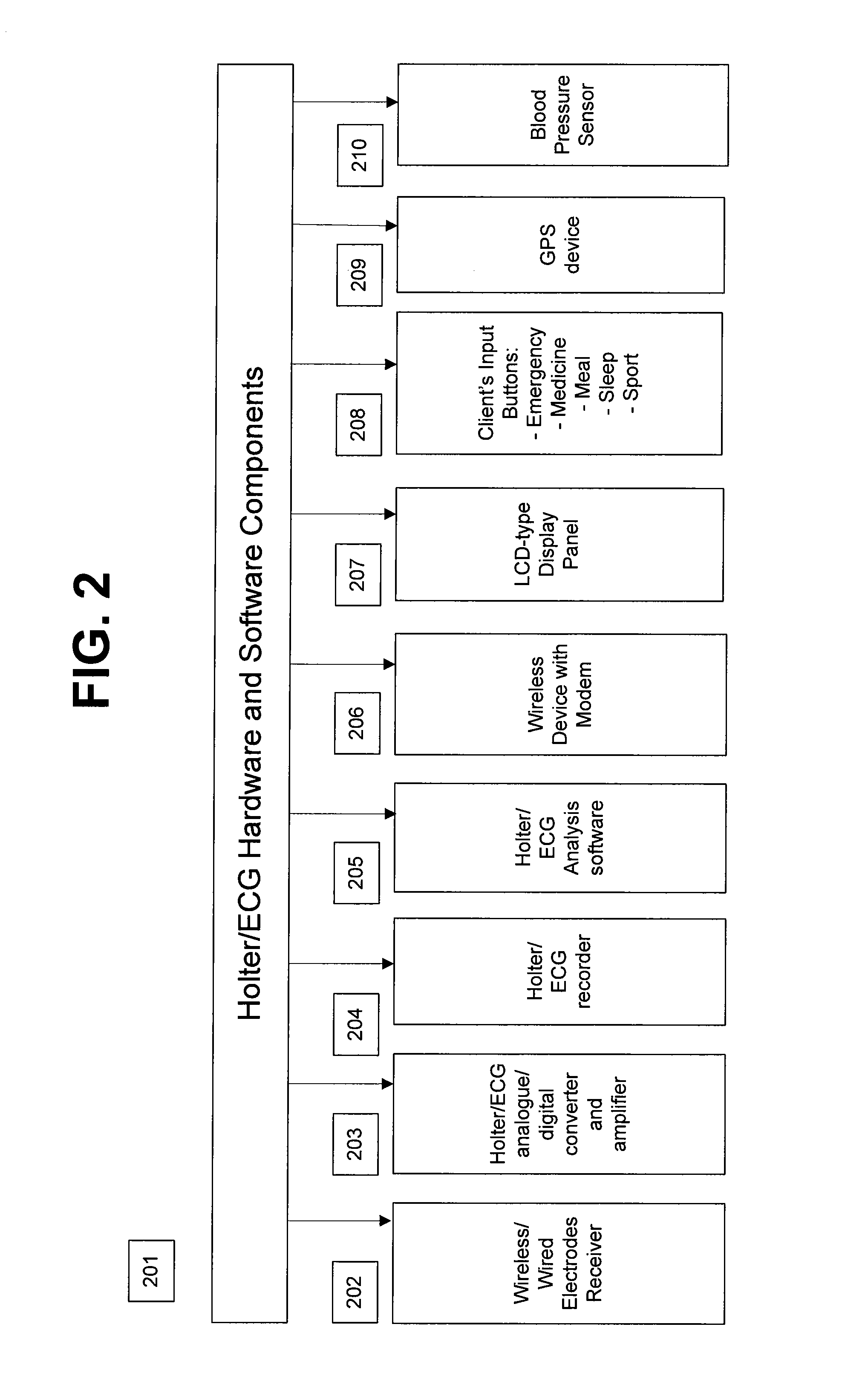
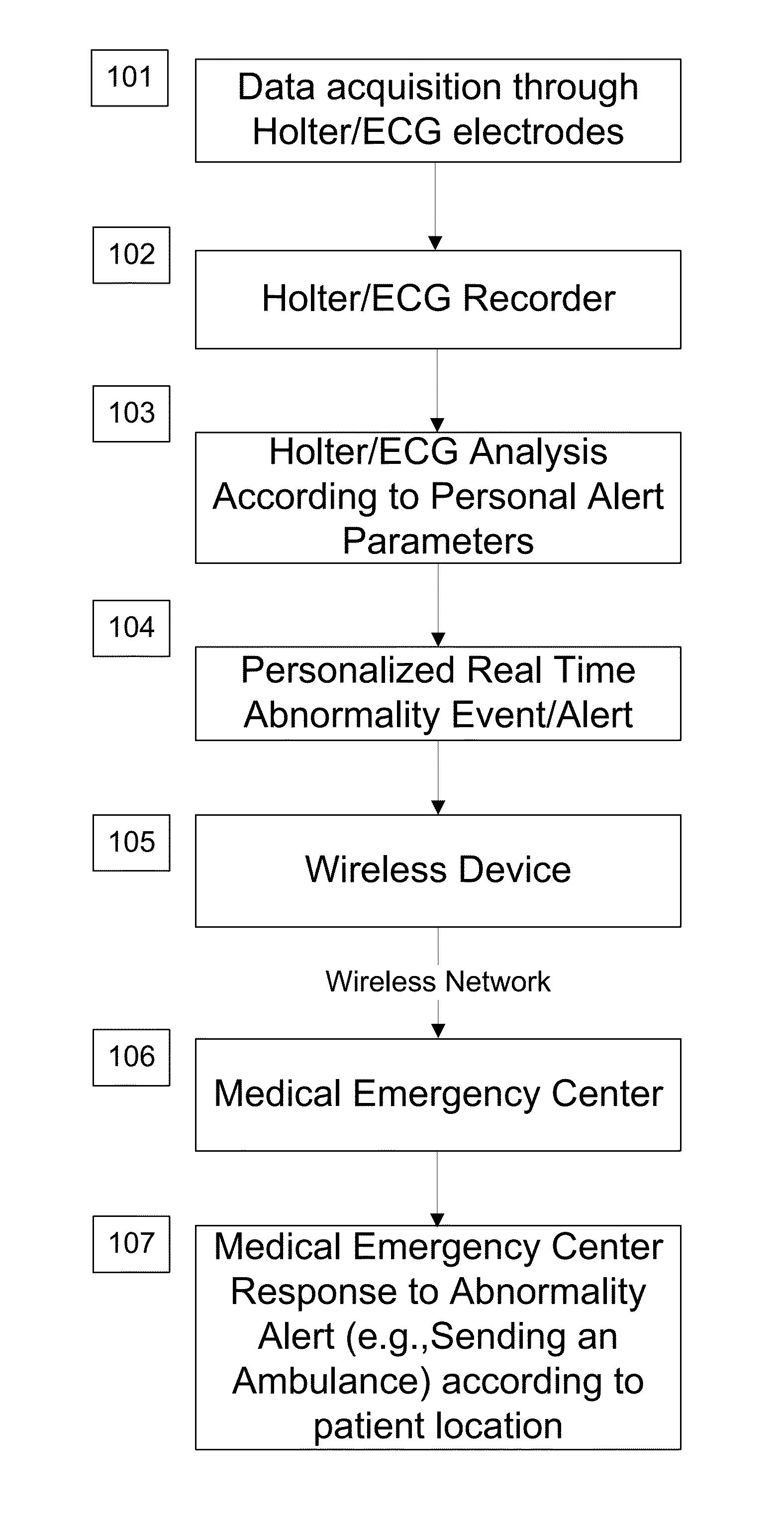
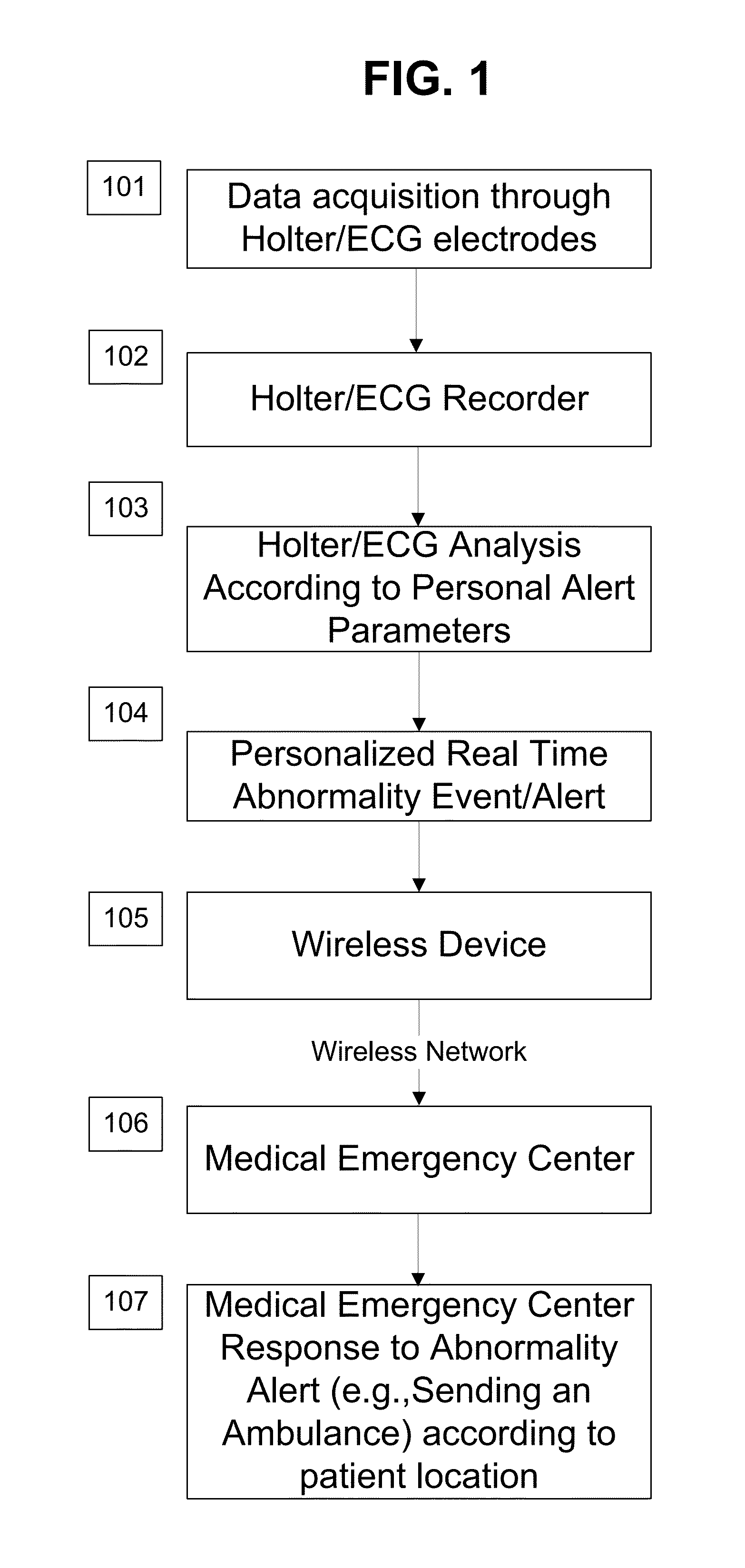
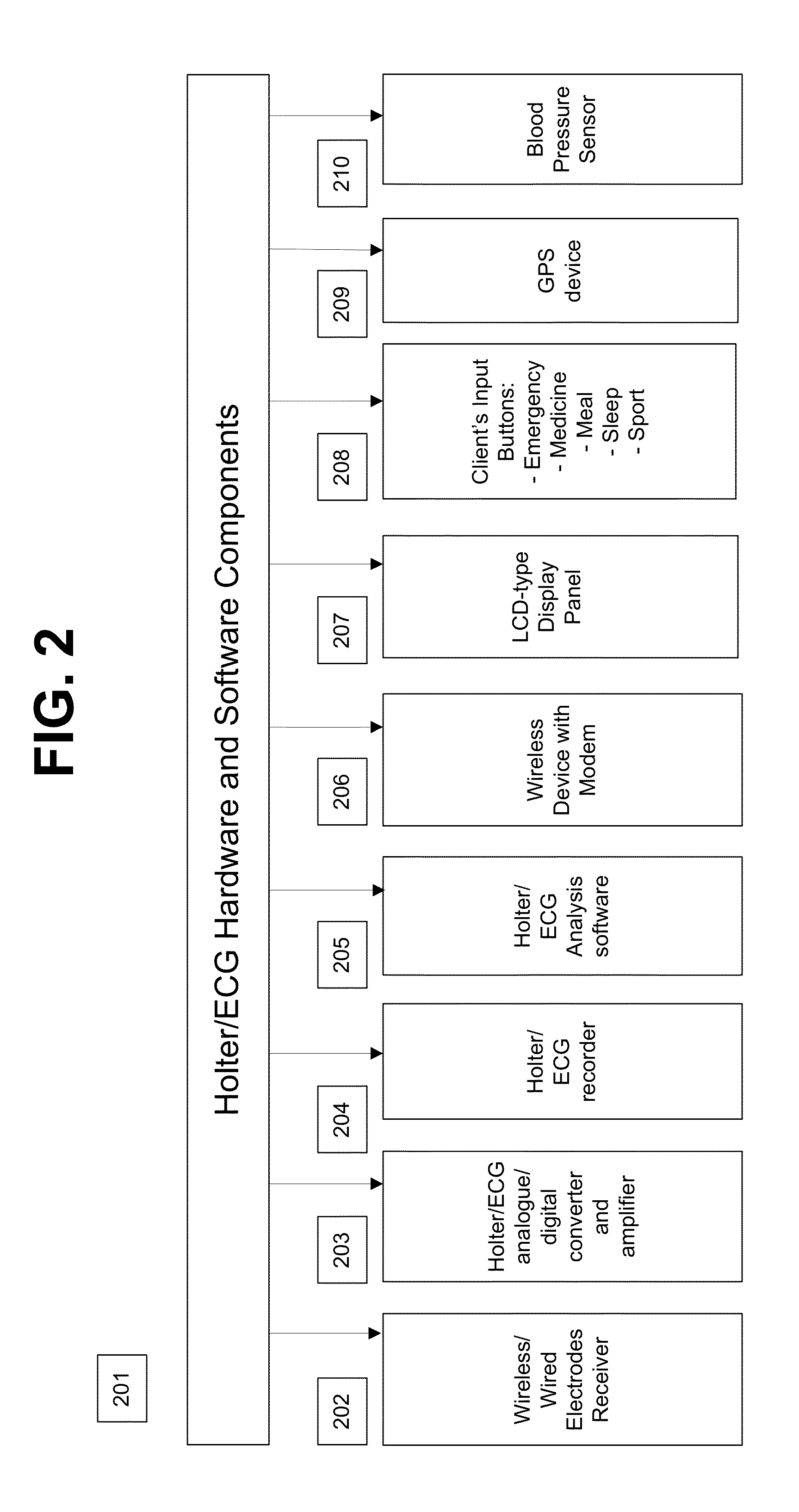
System and apparatus for providing diagnosis and personalized abnormalities alerts and for providing adaptive responses in clinical trials
InactiveUS8838217B2ElectrocardiographyEvaluation of blood vesselsHolter ElectrocardiographyClinical trial
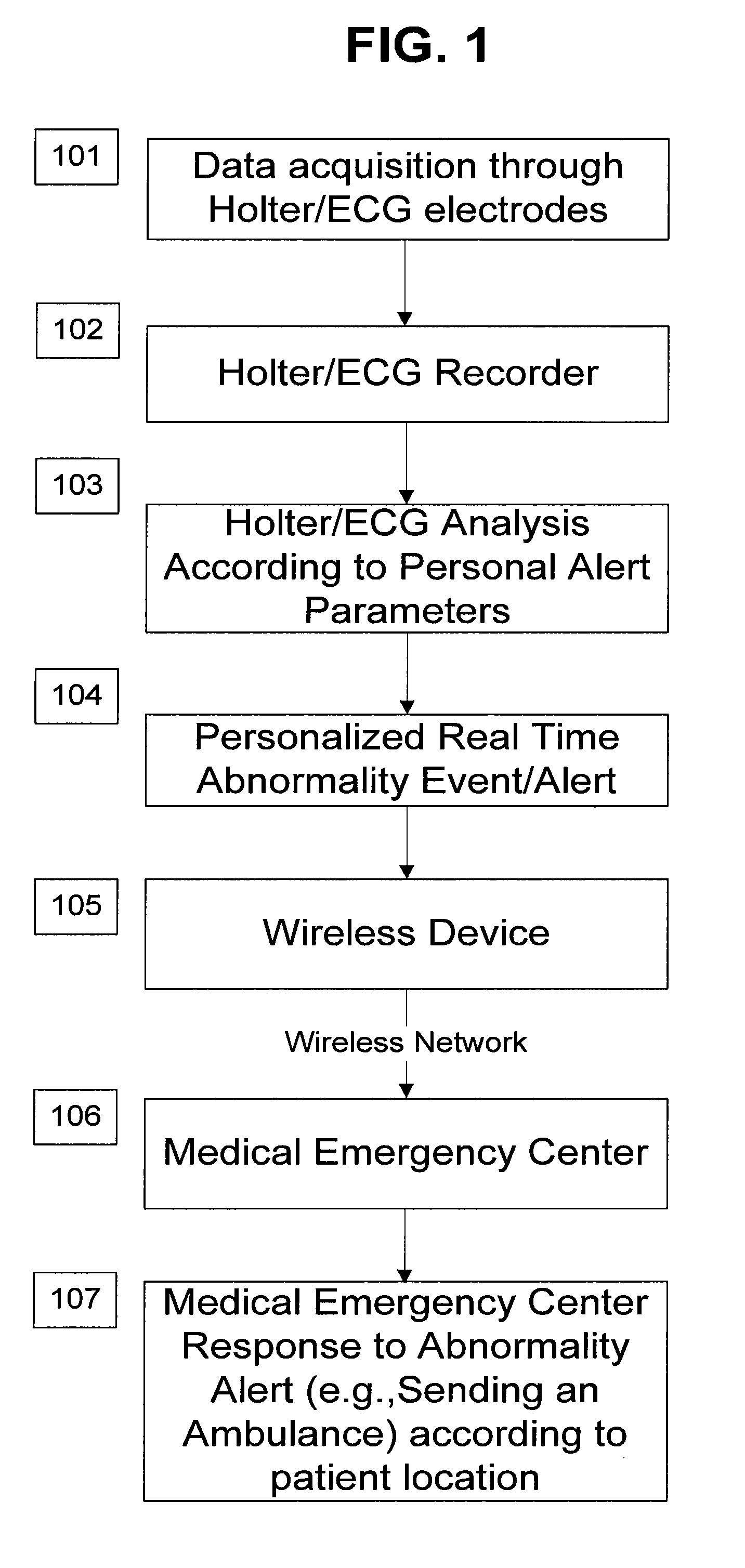
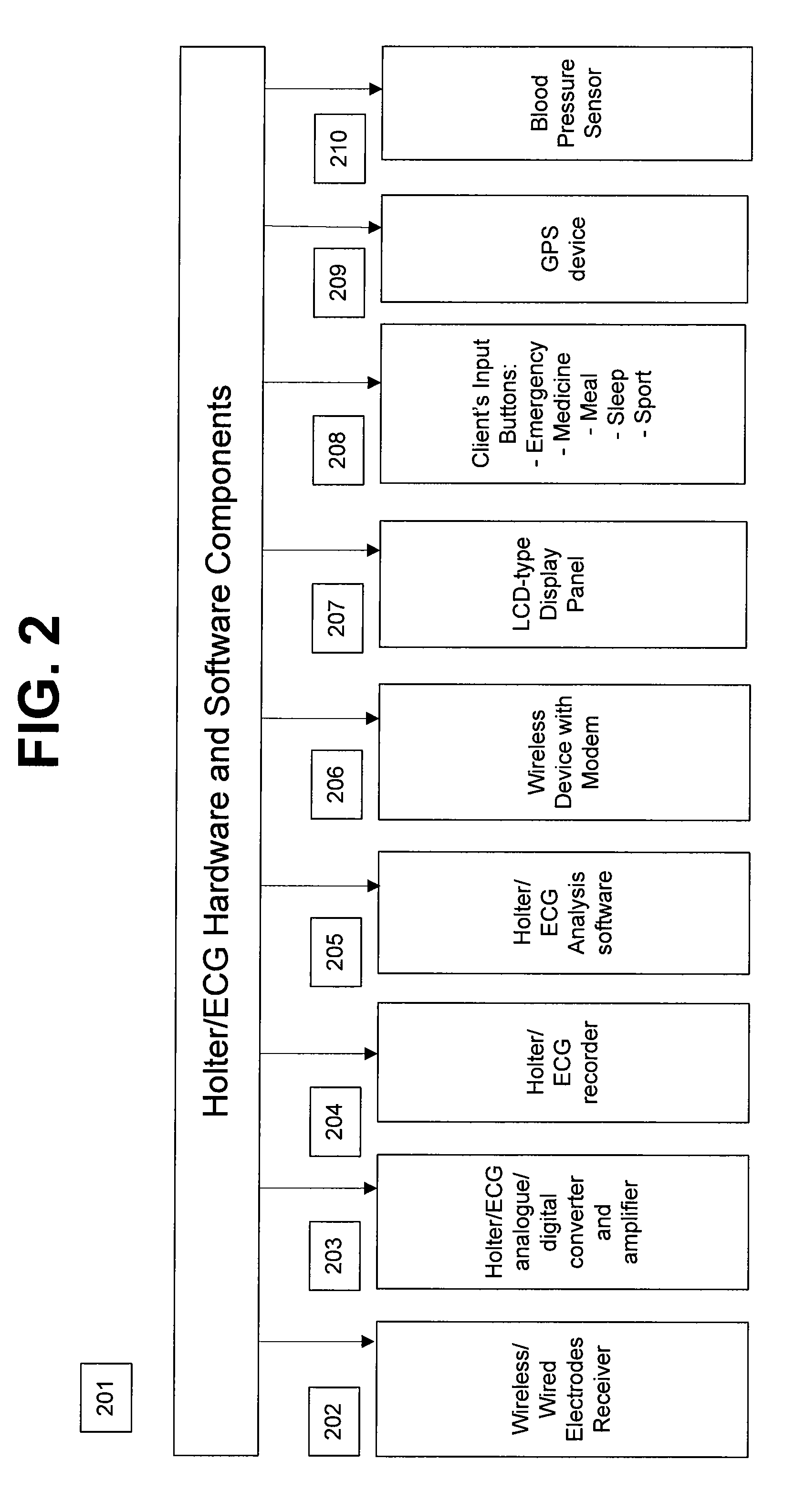
A personalized real-time automated cardiovascular monitoring system monitors abnormalities in a patient's cardiovascular activity data through the use of individually adjusted electrocardiogram Holter apparatus (Holter / ECG device) that provides an automatic medical diagnosis of cardiac abnormalities and generates abnormality alert signals representative of certain abnormalities in patient's cardiac activities. The signals are transmitted using a wireless network to a medical dispatcher center. A response is generated according to the abnormality detected. Individual parameters indicative of patient's cardio activities are personalized to allow for adjustments of chronic patients. A base Holter / ECG unit, includes the wireless / electric electrodes and their respective wireless / electric connections, and a Holter / ECG recording unit affixed to the base unit. Automatic real-time medical response may be provided based on automatic cardiac abnormality alert detection from the Holter / ECG data.
Owner:MAKOR ISSUES & RIGHTS
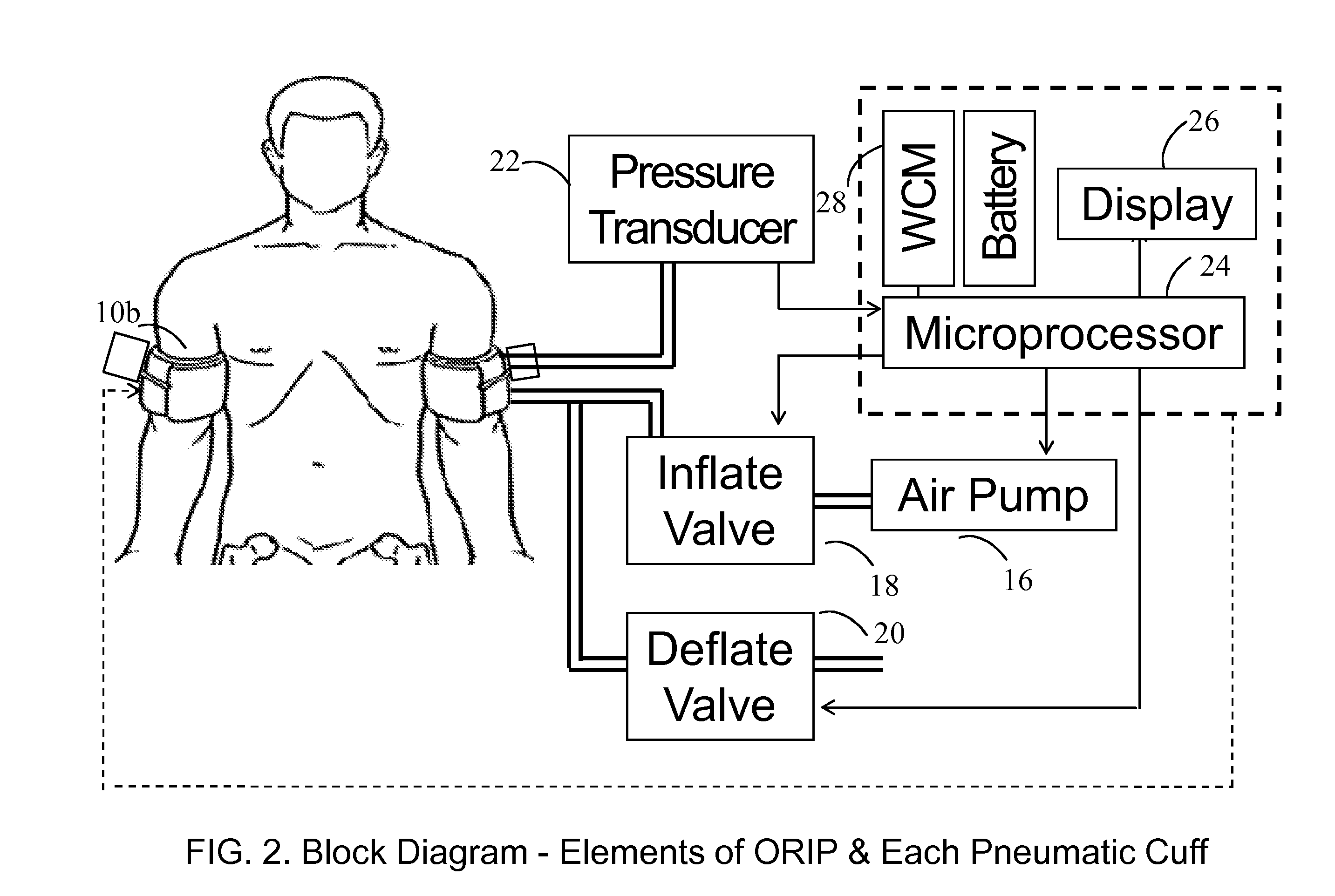
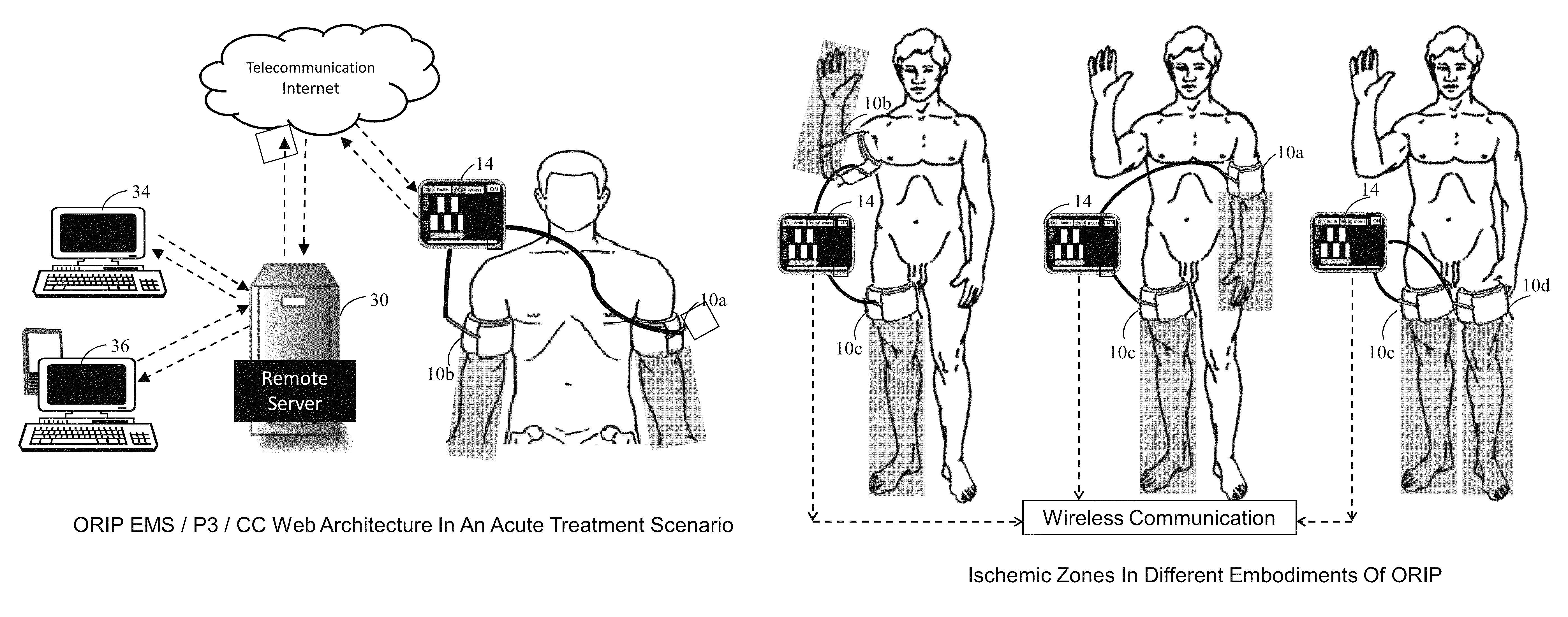
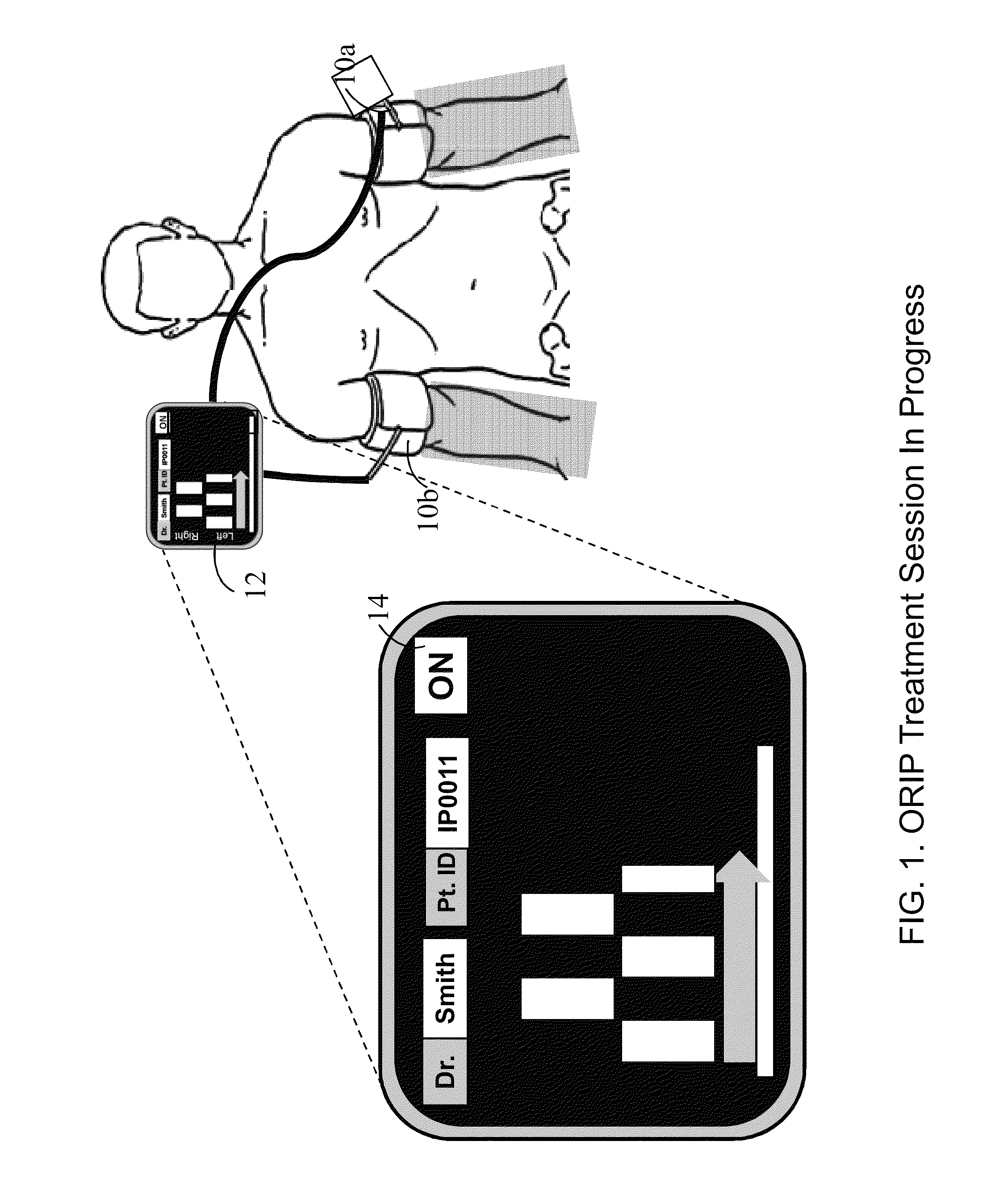
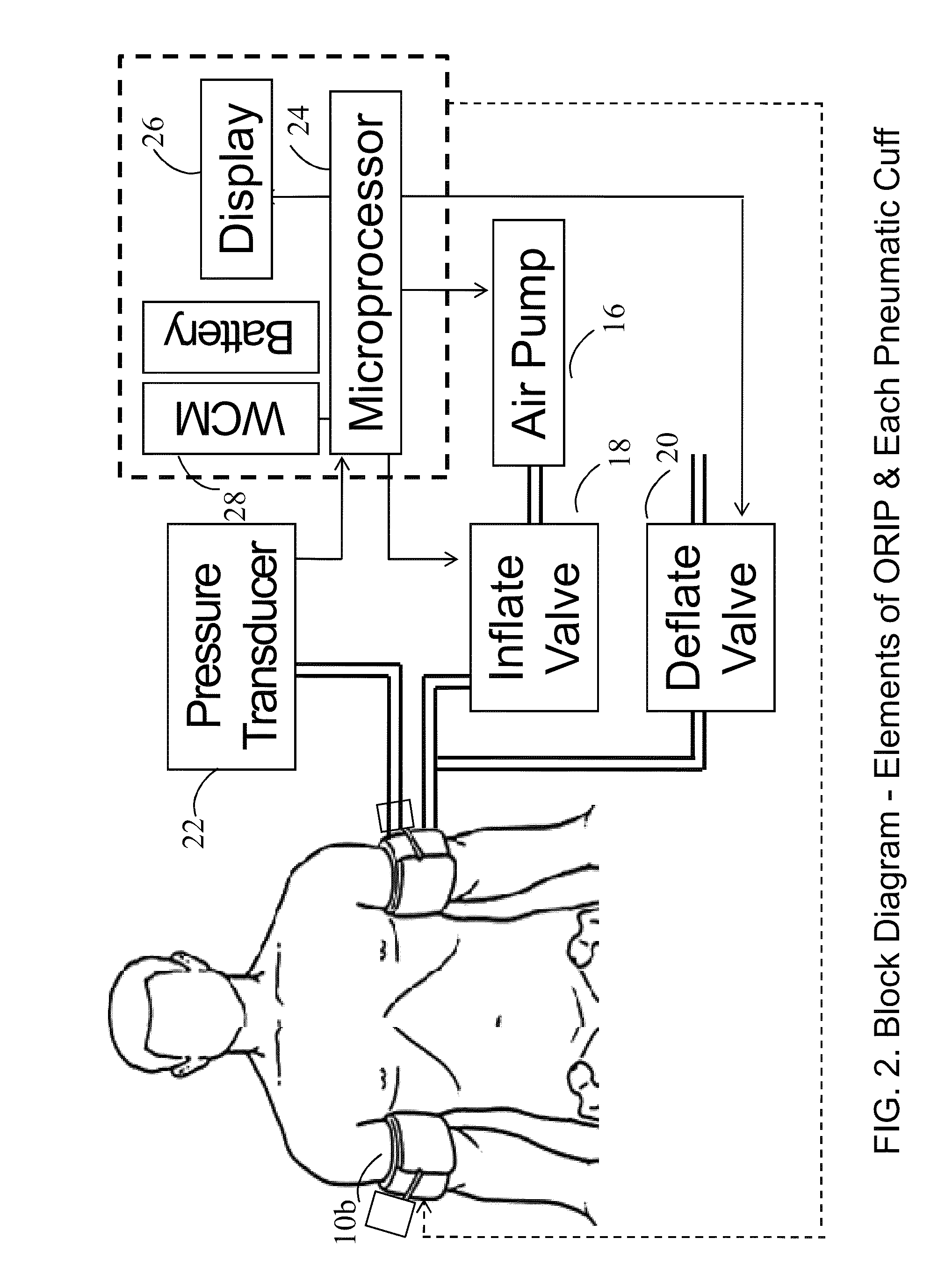
Methods and apparatus for optimal remote ischemic preconditioning (ORIP) for preventing ischemia-reperfusion injuries to organs
ActiveUS20110238107A1Preventing ischemia-reperfusion injuryMinimize consequencesEvaluation of blood vesselsSensorsReperfusion injuryPercutaneous angioplasty
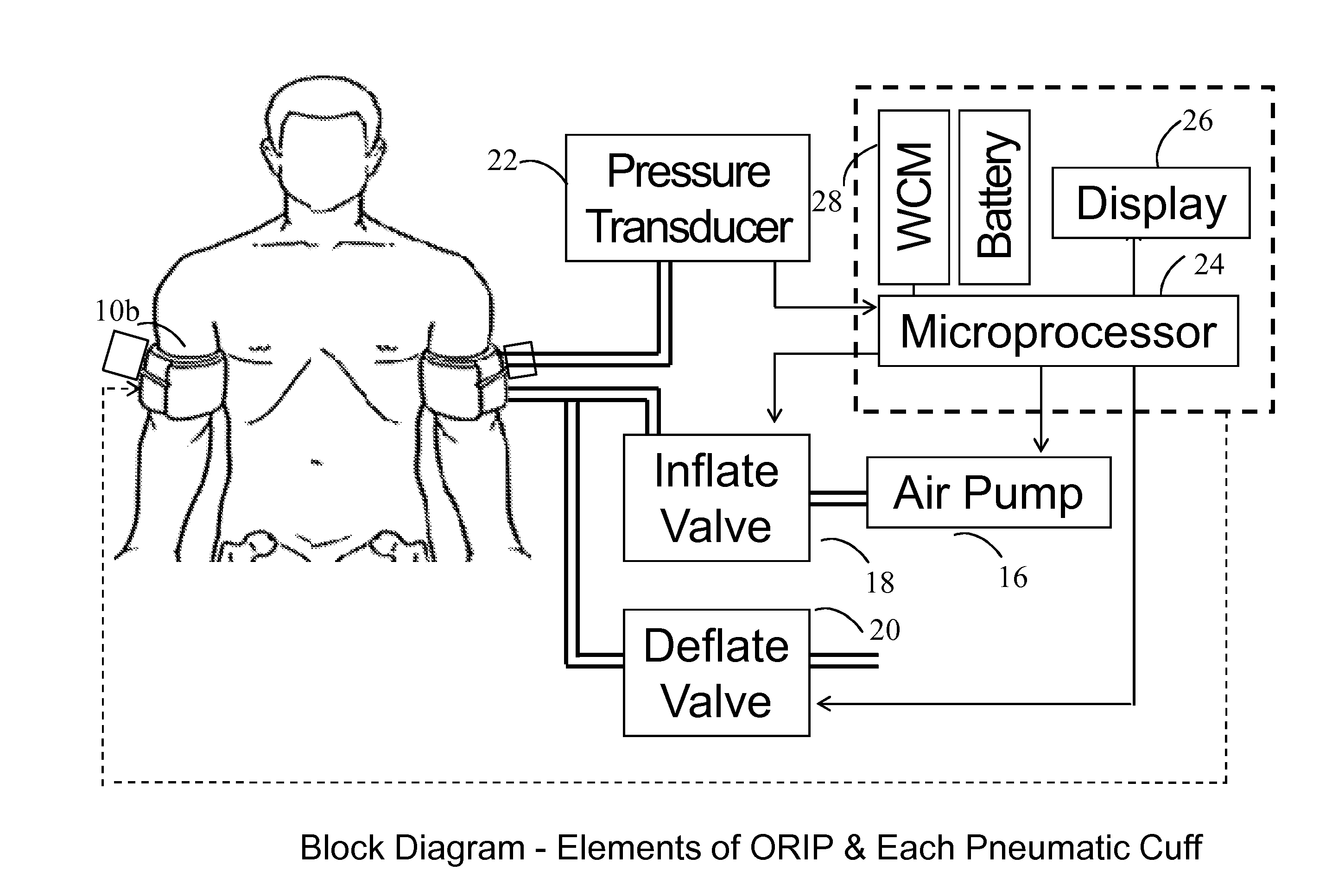
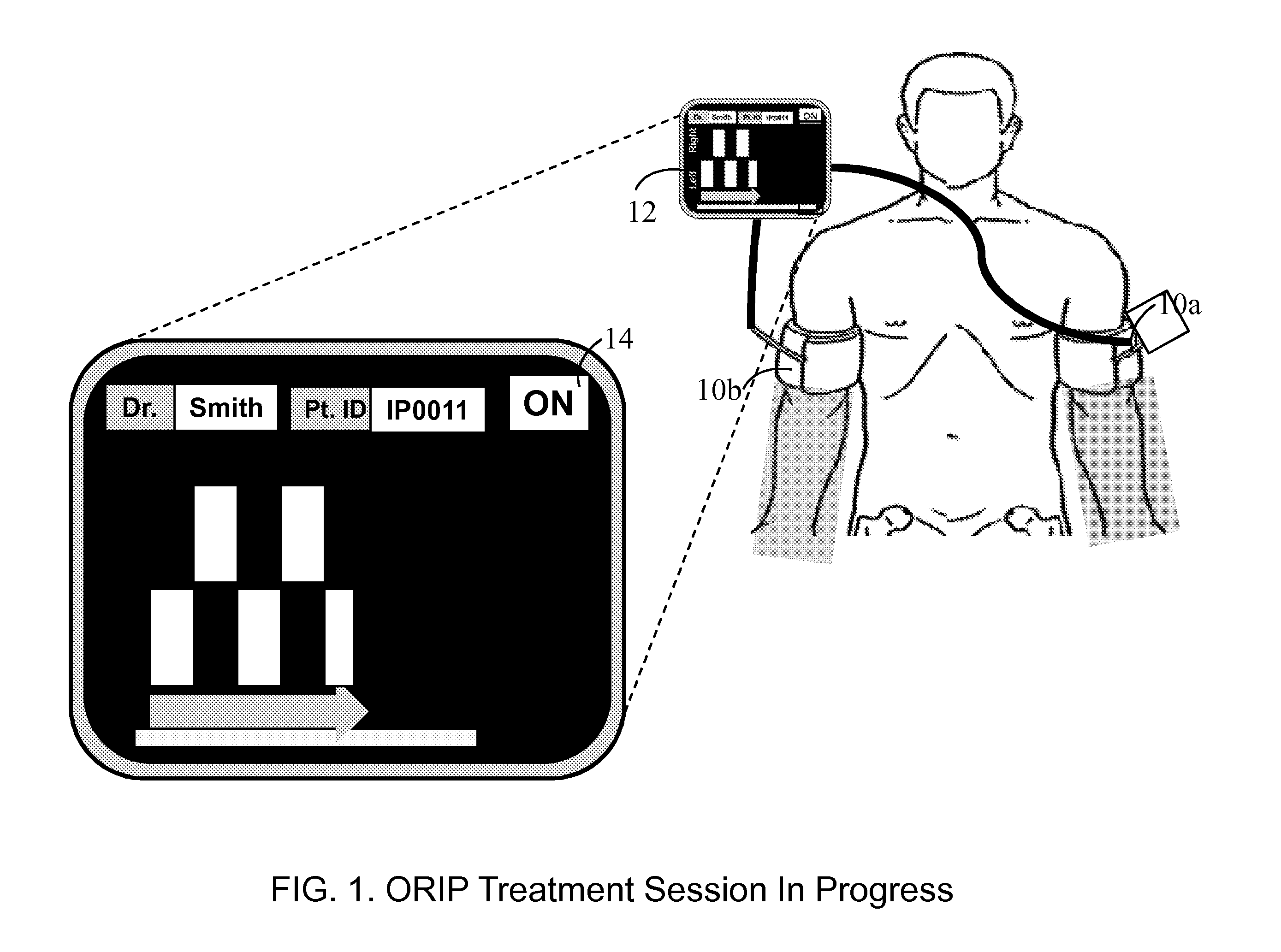
Ischemia-reperfusion injury commonly results from any surgical procedure requiring stopping of blood supply to an organ followed by reperfusion such as in heart bypass, angioplasty or organ transplant. The invention discloses a method to harness the innate power of repetitive transient ischemia in protecting organs against imminent ischemia-reperfusion, or any patho-physiological insults. This method of optimal remote ischemic preconditioning (ORIP) comprises of utilizing a pair of programmable pneumatic cuffs that inflate / deflate alternately occluding blood circulation to each of the limbs for pre-defined time intervals. The apparatus delivers maximal ORIP dose in shortest possible time either as an EMS procedure during patient transportation to hospital, as elective pre-surgery treatment, or in critical care for preventing multiple-organ-dysfunction-syndrome. ORIP can be self-administered and remotely monitored by clinician especially in chronic patients for homeostasis of malfunctioning target organs. ORIP may also be deployed as adjunct in angioplasty, gene / stem cell heart repair therapies.
Owner:NEOCARDIUM
System and apparatus for providing diagnosis and personalized abnormalities alerts and for providing adaptive responses in clinical trials
A personalized real-time automated cardiovascular monitoring system monitors abnormalities in a patient's cardiovascular activity data through the use of individually adjusted electrocardiogram Holter apparatus (Holter / ECG device) that provides an automatic medical diagnosis of cardiac abnormalities and generates abnormality alert signals representative of certain abnormalities in patient's cardiac activities. The signals are transmitted using a wireless network through a bi-directional wireless protocol to a medical dispatcher center. A response is generated according to the abnormality detected. Individual parameters indicative of patient's cardio activities are personalized to allow for adjustments of chronic patients. A base Holter / ECG unit, includes the wireless / electric electrodes and their respective wireless / electric connections, and a Holter / ECG recording unit affixed to the base unit. The Holter / ECG apparatus can be integrated as a chip into a wireless device, such device being in a wireless phone network. Automatic real-time medical response may be provided to detection of risks of adverse effects resulting from use of at least one medication, based on automatic cardiac abnormality alert detection from the Holter / ECG data.
Owner:MAKOR ISSUES & RIGHTS
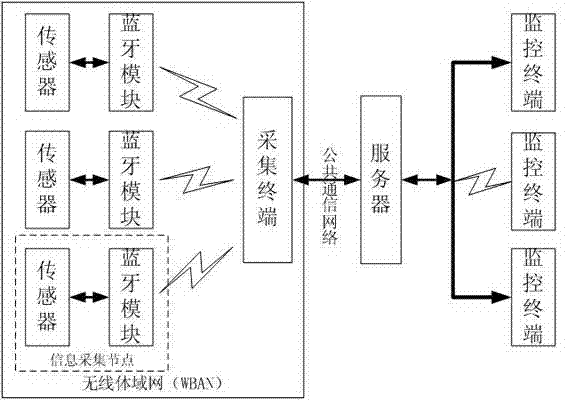
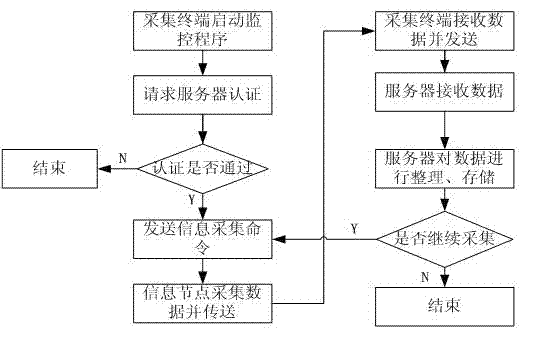
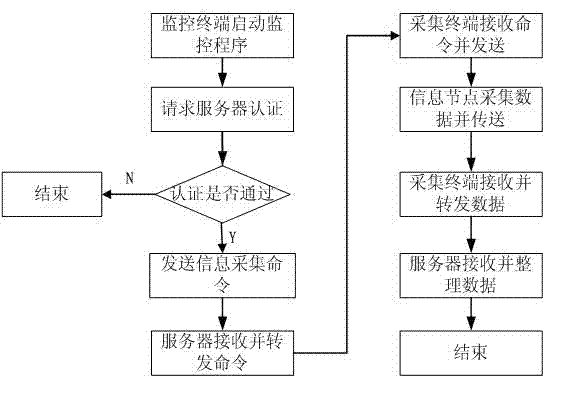
Bluetooth-based medical information acquisition system and acquisition method
ActiveCN102894963AReduce volumeEasy to collectNear-field transmissionEvaluation of blood vesselsComputer terminalBluetooth
The invention provides a Bluetooth-based medical information acquisition system. The acquisition system comprises an information acquisition node, an information acquisition terminal, a network server and a monitoring terminal, wherein the information acquisition terminal and the monitoring terminal are connected with the network server respectively; and the information acquisition node is connected with the information acquisition terminal. The invention also provides an acquisition method based on the system. Acquisition, processing, transmission, storage and management of remote medical information are performed by establishing the Bluetooth-based information acquisition system, so that remote acquisition of medical information is realized, the state of an illness is monitored in real time, the physiological status of a patient can be monitored conveniently, and the method is particularly suitable for a chronic patient. The medical quality can be obviously improved, the medical cost can be reduced, and broad application range and market space are realized.
Owner:JIANGSU ZEBONE SMARTCITY INFORMATION SYST
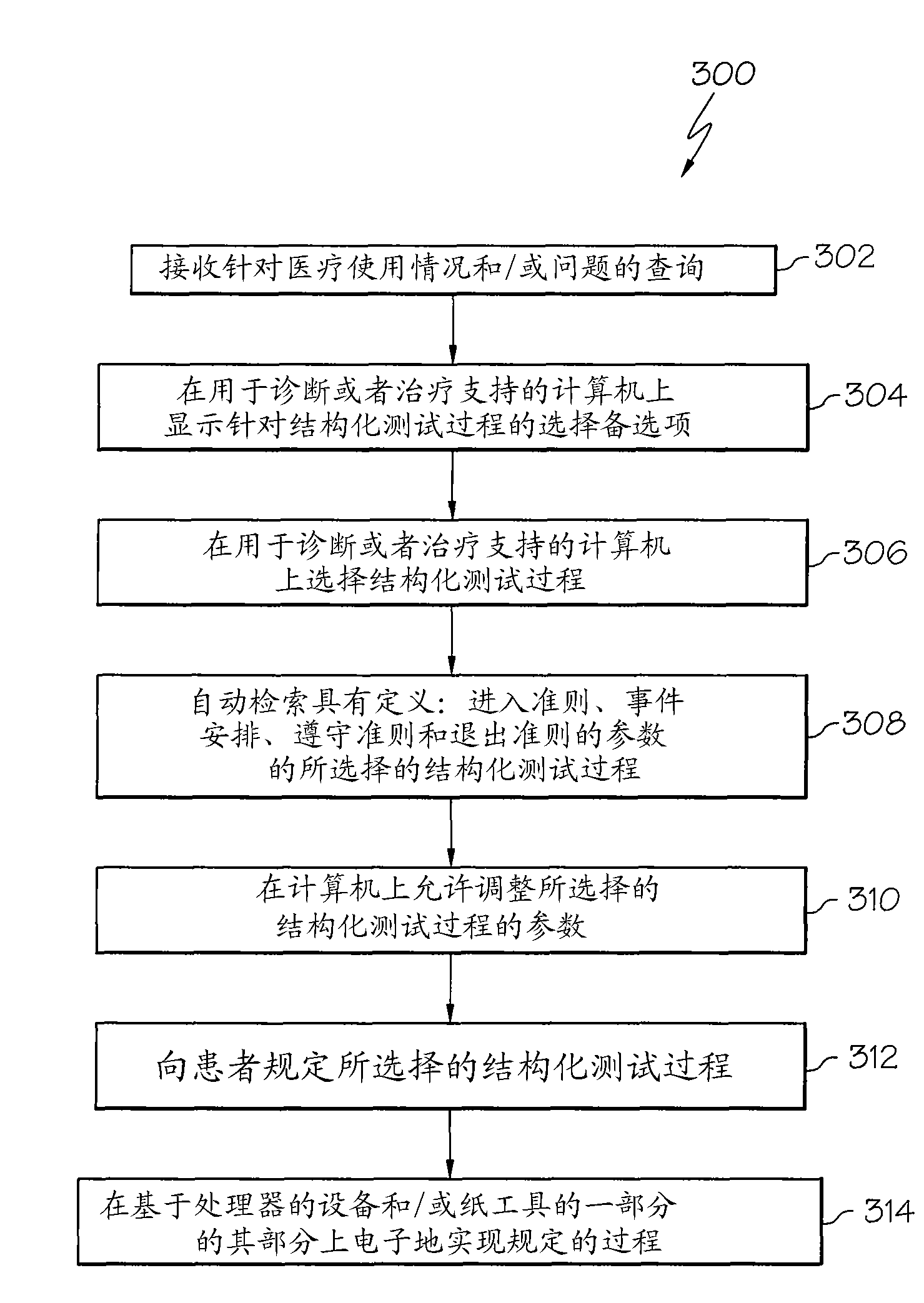
Structured testing method and device for diagnosis or treatment support of chronically ill patients
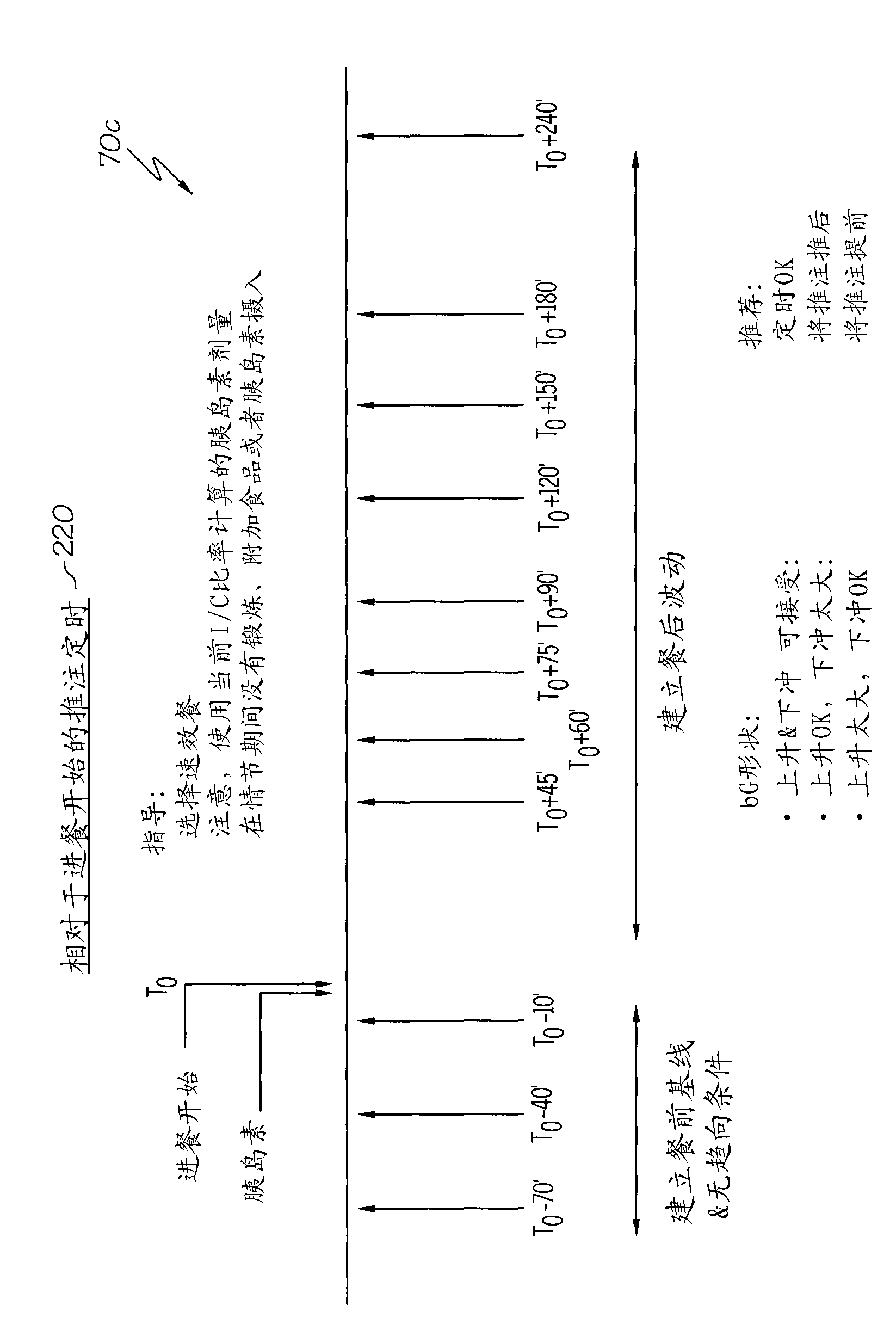
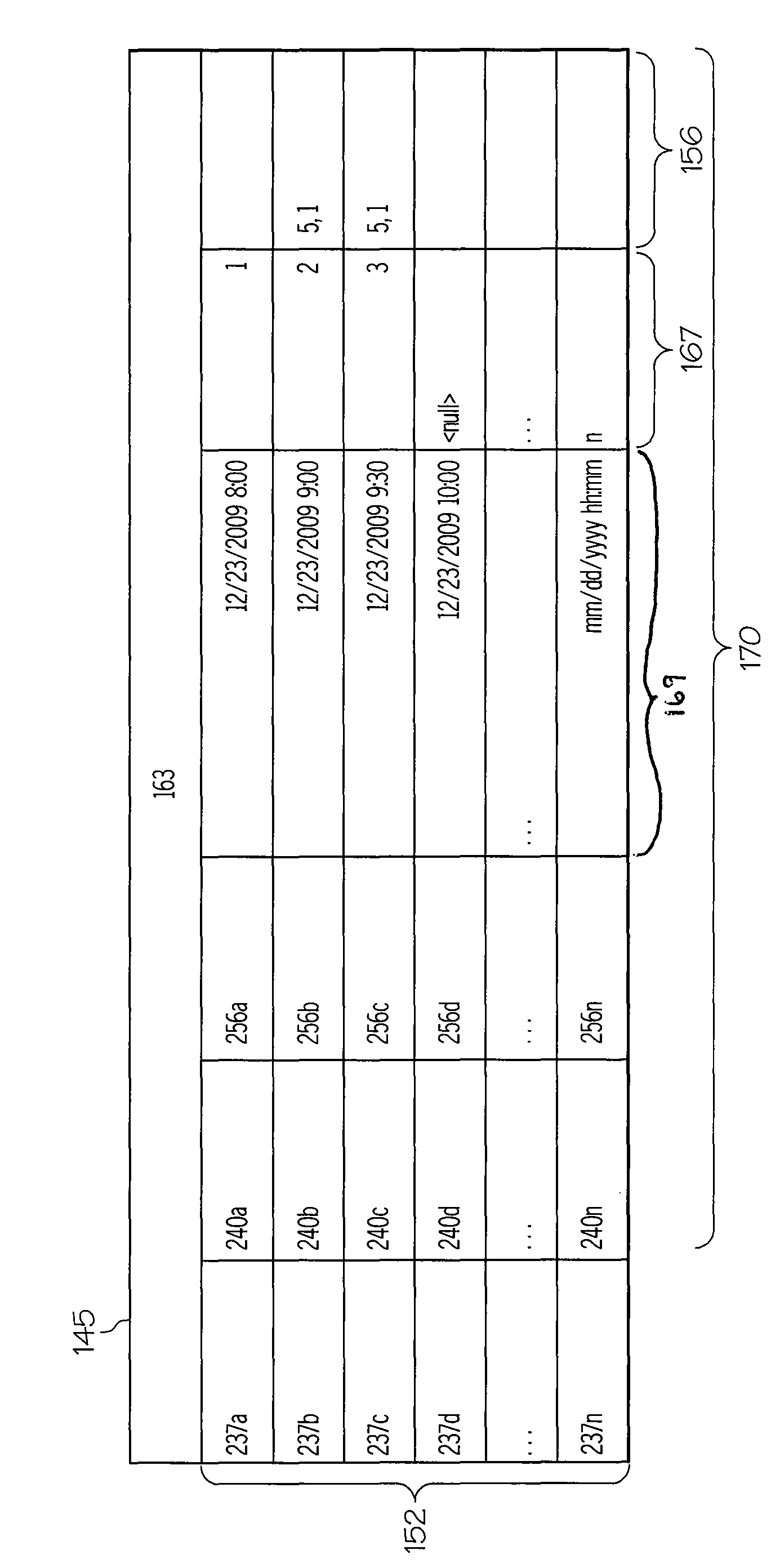
A structured testing method (200, 300, 388) and device (18) for diagnosis or treatment support of a chronic disease patient (12) are disclosed. The device (18) implements a medical usage and / or question based structured collection process (70) that provides at least one or more parameters (220, 222, 224, 226, 228, 230, 232, 237, 238, 240), said parameters (220, 222, 224, 226, 228, 230, 232, 237, 238, 240) define at least one entry criterion (226), event schedule (222), at least A compliance criterion (224) and at least one exit criterion (228). Entry criteria (226) establish conditions that need to be met before obtaining biomarker data (256) from patient (12). Each event (237) in the event schedule (222) may include a delivery time (238), instructions (230) for carrying out the event (237), a request for information from the patient (12) (240), an At least one or more of a request (240) for an action and a request (240) for collection of at least one type of biomarker data (256) from the patient (12). Adherence criteria (224) are used to qualitatively assess whether events (237) delivered against event schedules (222) provide acceptable data (256, 170, 145) to address medical usage and / or issues, and exit criteria (228 ) establishes the conditions that need to be met before the structured collection process (70) exits.
Owner:F HOFFMANN LA ROCHE & CO AG
Methods and apparatus for optimal remote ischemic preconditioning (ORIP) for preventing ischemia-reperfusion injuries to organs
ActiveUS8911469B2Strong responsePreventing ischemia-reperfusionEvaluation of blood vesselsCatheterSurgical departmentHeart bypass
Ischemia-reperfusion injury commonly results from any surgical procedure requiring stopping of blood supply to an organ followed by reperfusion such as in heart bypass, angioplasty or organ transplant. The invention discloses a method to harness the innate power of repetitive transient ischemia in protecting organs against imminent ischemia-reperfusion, or any patho-physiological insults. This method of optimal remote ischemic preconditioning (ORIP) comprises of utilizing a pair of programmable pneumatic cuffs that inflate / deflate alternately occluding blood circulation to each of the limbs for pre-defined time intervals. The apparatus delivers maximal ORIP dose in shortest possible time either as an EMS procedure during patient transportation to hospital, as elective pre-surgery treatment, or in critical care for preventing multiple-organ-dysfunction-syndrome. ORIP can be self-administered and remotely monitored by clinician especially in chronic patients for homeostasis of malfunctioning target organs. ORIP may also be deployed as adjunct in angioplasty, gene / stem cell heart repair therapies.
Owner:NEOCARDIUM
System and apparatus for providing diagnosis and personalized abnormalities alerts and for providing adaptive responses in clinical trials
InactiveUS20140221858A1ElectrocardiographyEvaluation of blood vesselsPersonalizationStatistical analysis
A personalized real-time automated cardiovascular monitoring system monitors abnormalities in a patient's cardiovascular activity data through the use of individually adjusted electrocardiogram Holter apparatus (Holter / ECG device) that provides an automatic medical diagnosis of cardiac abnormalities and generates abnormality alert signals representative of certain abnormalities in patient's cardiac activities. The signals are transmitted using a wireless network through a bi-directional wireless protocol. Individual parameters indicative of patient's cardio activities are personalized to allow for adjustments of chronic patients. A base Holter / ECG unit, includes the wireless / electric electrodes and their respective wireless / electric connections, and a Holter / ECG recording unit affixed to the base unit. Data is collected and subjected to statistical analysis.
Owner:MAKOR ISSUES & RIGHTS
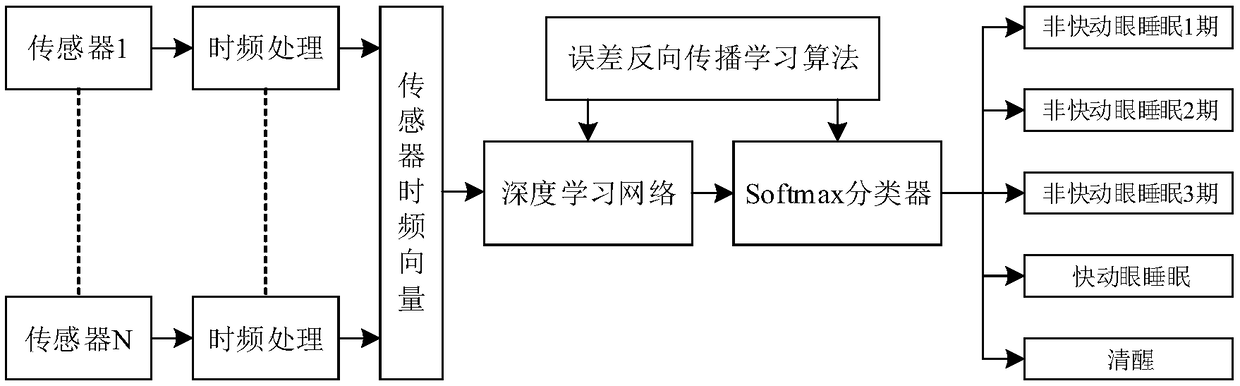
Human body sleep stage estimating method based on deep learning network
InactiveCN109316166AImprove estimation performanceRealize monitoringDiagnostic recording/measuringSensorsHuman bodyPulse rate
The invention provides a human body sleep stage estimating method based on a deep learning network, and belongs to the technical field of medical treatment and health and information. According to themethod, the sleep stage where the human body is in can be estimated, and an effective method for analyzing the sleep stage is provided for the fields of chronic patients, elder monitoring, health surveillance and the like. According to the method, by means of one or more kinds of information of the human body heart rate, respiratory rate, body moving, snores, electroencephalogram signals and thelike collected by a sensor, the optimal features of the information are extracted through the deep learning network, and a Softmax classifier is adopted for estimating the sleep stage based on the optimal features. According to the method, the estimating performance of the human body sleep stage can be improved, and the health state of the human body is effectively monitored.
Owner:DALIAN UNIV OF TECH
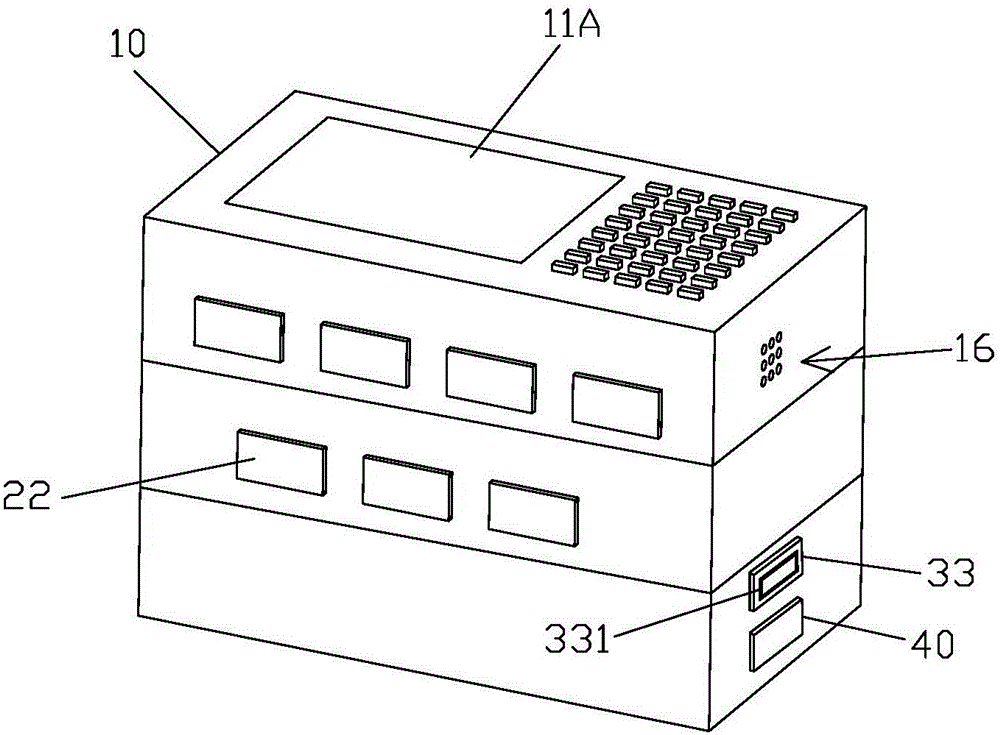
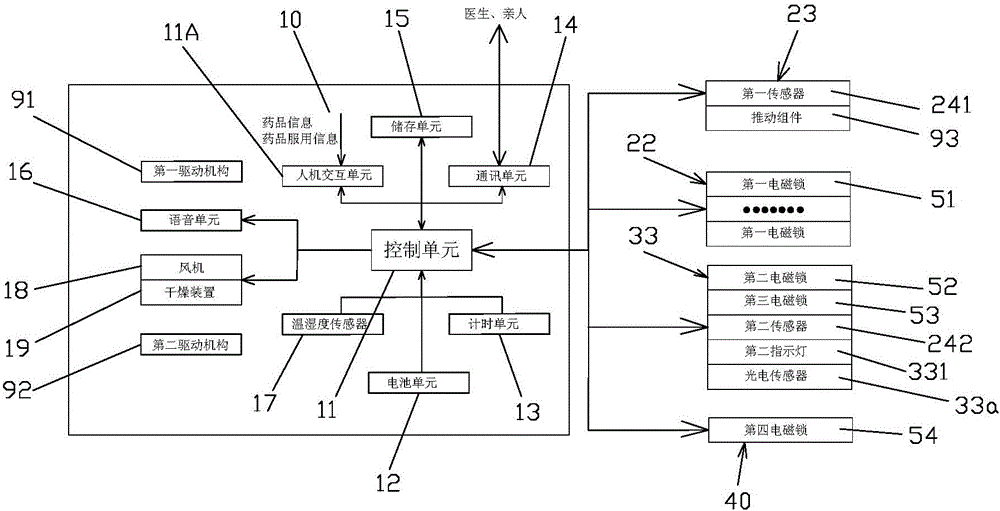
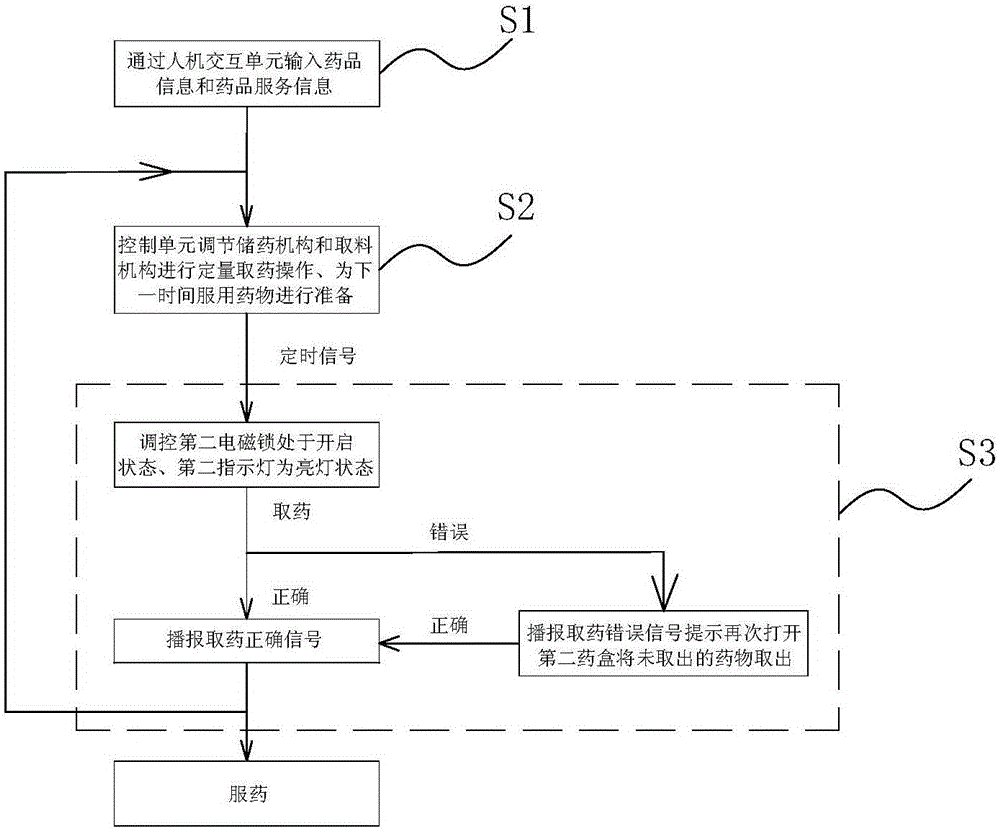
Medicine box prompting chronic patient to accurately take medicine
InactiveCN105668016APrecision medicineAvoid confusionSmall article dispensingOral administration deviceEmergency medicineChronic patient
The invention relates to a medicine box prompting a chronic patient to accurately take medicine. The medicine box comprises a box body. The box body is provided with medicine storage units used for storing and getting various medicines, and a medicine collecting unit used for collecting the medicines gotten from the medicine storage units for the patient to take. Each medicine storage unit comprises a medicine storage mechanism used for storing the medicines, and a medicine getting mechanism used for getting the medicines in the medicine storage mechanism out according to the dosage. Every time the patient takes medicine, the patient only needs to get out all the medicines in a second medicine case and takes medicine, it can be guaranteed that the patient poor in consciousness can accurately get and take medicine, and the phenomena that medicine fetching is neglected, the patient takes less medicine and the patient takes more medicine are avoided.
Owner:王丽
Multifunctional intelligent medicine box
ActiveCN111467230ASolve forgetting to take medicineSolve the problem of taking multiple types of medicinesPharmaceutical containersMedical packagingControl cellChronic patient
The invention discloses a multifunctional intelligent medicine box. The medicine box includes a medicine box body, a medicine delivery mechanism, a medicine getting drawer and a control unit, whereinthe medicine delivery mechanism and the medicine getting drawer are arranged inside the medicine box body; the medicine delivery mechanism includes a medicine storage box, a medicine falling disc, fan-shaped rotating medicine taking clips and medicine falling slide ways which are arranged from top to bottom, and provides a medicine delivery channel for the medicine getting drawer; and the controlunit is connected to the medicine delivery mechanism and used for controlling the medicine delivery mechanism to put single medicines into the medicine getting drawer. Through the adopting of the medicine box, the precise amount of medicine taken can be put into the medicine getting drawer, and patients can be timely reminded to take medicines at the time of medication; the problems of forgettingto take medicines and taking many kinds of the medicines can be effectively solved; and therefore, the medicine box is especially suitable for chronic patients and the patients who take many medicines.
Owner:HOHAI UNIV CHANGZHOU
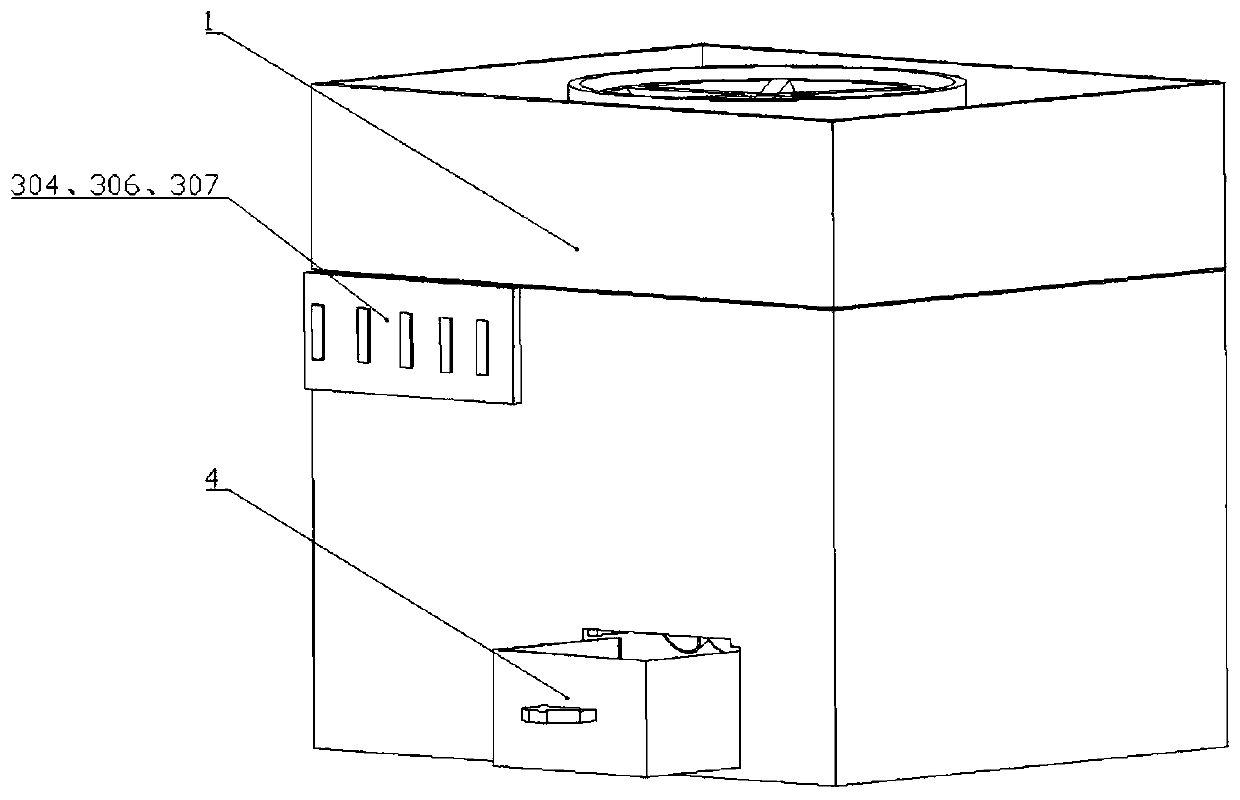
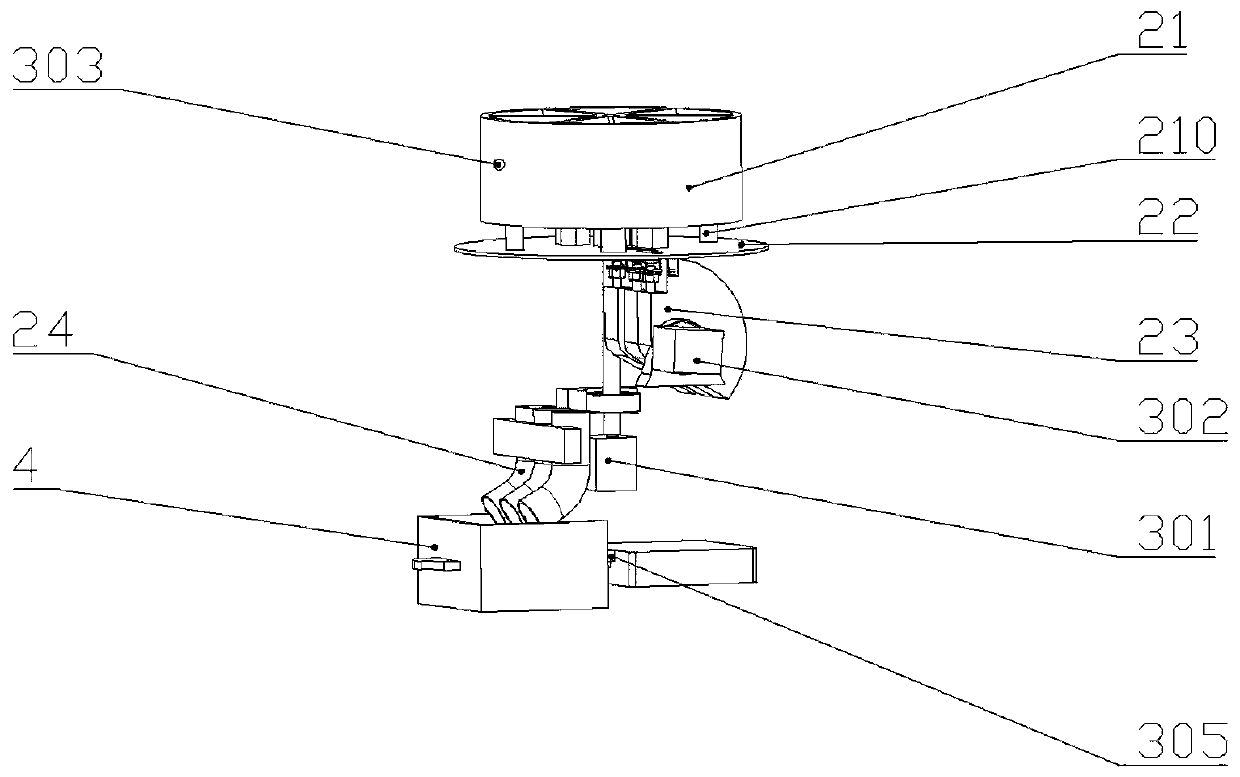
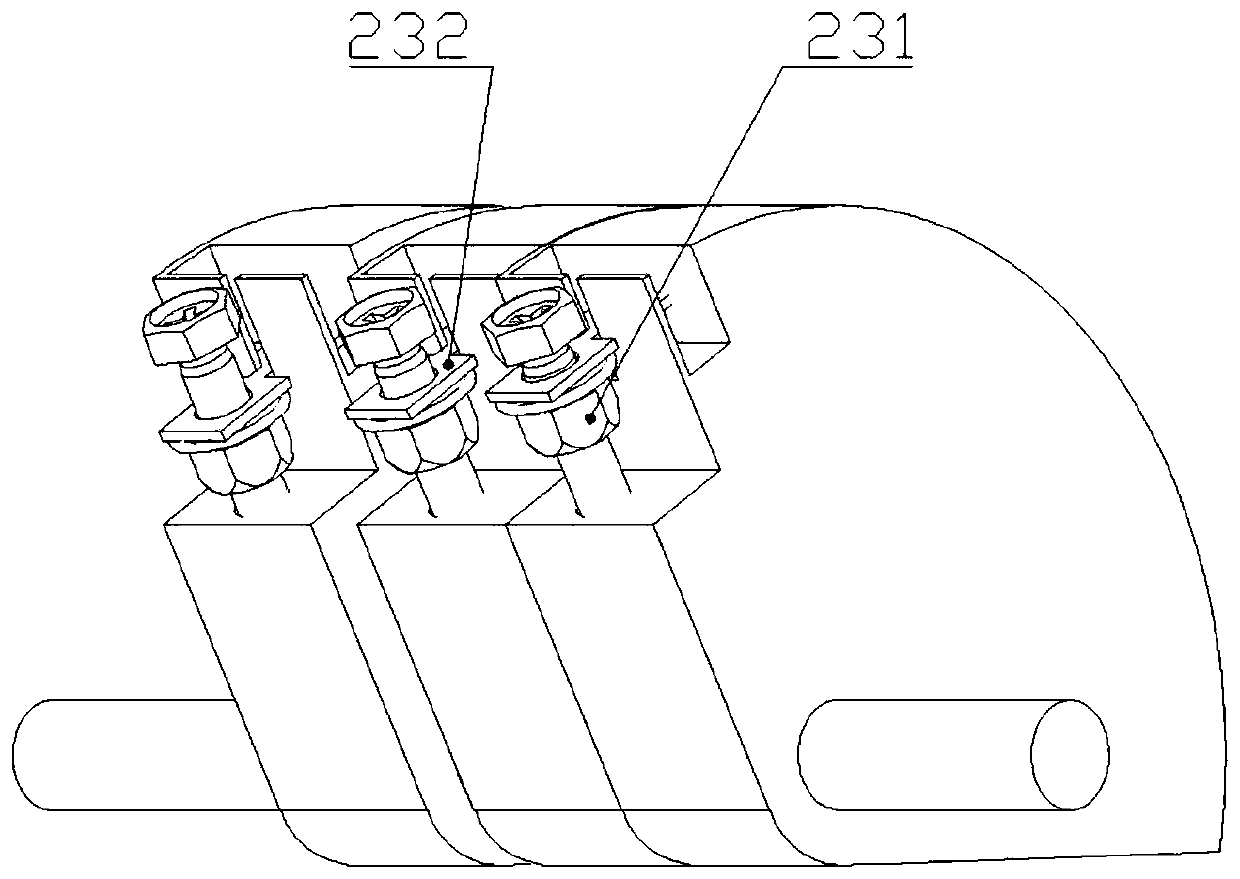
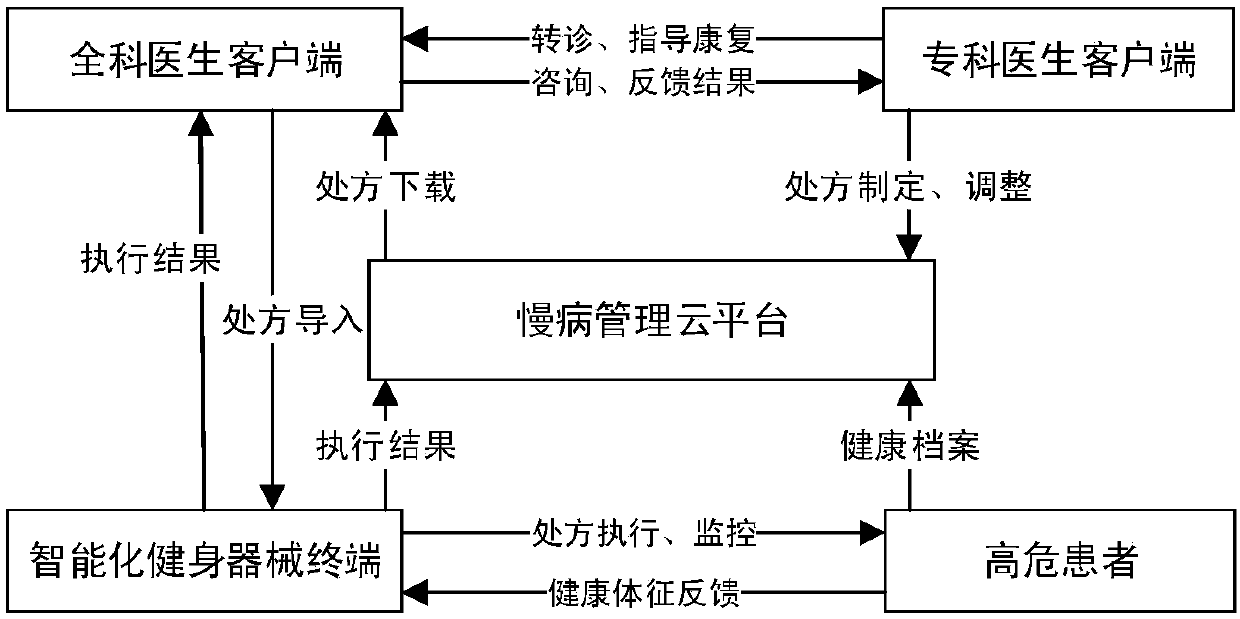
High-risk chronic patient exercise rehabilitation service system and method
InactiveCN108039204ASafe Sports Rehabilitation ManagementEffective Sports Rehabilitation ManagementPhysical therapies and activitiesHealth-index calculationGeneral practionerDisease
The invention discloses a high-risk chronic patient exercise rehabilitation service system and method. A specialist doctor client is used to complete the diagnosis and exercise risk assessment of a superior specialist to the disease of a high-risk patient, and develop a personalized exercise rehabilitation program for a high-risk group whose disease is stably controlled. A general practitioner client is used to assist a general practitioner in a lower-level community hospital to implement the exercise rehabilitation program, supervise the implementation of the exercise rehabilitation program,prevent and control the exercise risk in the implementation process, and feed back the implementation performance of the exercise rehabilitation program. An intelligent fitness equipment terminal is used to provide means for the implementation of the exercise rehabilitation program. According to the invention, safe and effective exercise rehabilitation management for the high-risk patient is realized; the work pressure of the superior specialist is relieved; at the same time, through the basic-level community hospital and intelligent fitness equipment, exercise rehabilitation of the high-riskgroup is realized; and the exercise rehabilitation benefit of the high-risk patient is improved.
Owner:HEFEI INSTITUTES OF PHYSICAL SCIENCE - CHINESE ACAD OF SCI
Method for making saffron crocus sugar-free candied dates
The invention provides a method for making saffron crocus sugar-free candied dates. According to the method, effective constituents of saffron crocus are integrated into Chinese dates, so that the blood-activating and nourishing functions of the candied dates are added, and cane sugar is replaced with stevioside, so that sweetness of the candied dates is enhanced, the problems that traditional candied dates are high in cane sugar and high in caloric value are solved, and microorganism breeding is inhibited through epsilon-polylysine. The saffron crocus sugar-free candied dates are made through the high-voltage pulsed electric field technology, vacuum impregnation and other methods, and the sugar-free candied dates which are convenient to carry, convenient to preserve and easy to eat and have the health care function are provided for particular persons, especially for obesity patients, chronic patients and diabetic patients.
Owner:杨军
Kit capable of detecting expression quantity of BAALC (brain and acute leukemia cytoplasmic) gene mRNA (Messenger Ribose Nucleic Acid)
InactiveCN103014154AIncreased sensitivityStrong specificityMicrobiological testing/measurementFluorescence/phosphorescenceReference genesMyeloid leukemia
The invention discloses a kit capable of detecting expression quantity of BAALC (brain and acute leukemia cytoplasmic) gene mRNA (Messenger Ribose Nucleic Acid), belonging to the field of biotechnology. The kit comprises a detection primer and fluorescence probe, a cDNA first strand synthesis reagent, fluorogenic quantitative PCR (Polymerase Chain Reaction) mixed liquor, negative control and positive control, wherein the primers and fluorescence probes for detection include a BAALC gene primer, a reference gene ABL primer and a Taqman fluorescence probe. The BAALC gene is a sign of progenitor cell of early hematopoietic cell, and is abnormally expressed in acute and chronic patients with AML (Acute Myelocytic Leukemia), ALL (Acute Lymphocytic Leukemia) and CML (Chronic Myeloid Leukemia). According to the embodiment of the invention, the mRNA level of the BAALC gene is detected by fluorogenic quantitative PCR with high sensitivity and specificity, and the specificity and sensitivity of the detection result are both obviously improved. The kit can provide a brandnew, fast, simple and convenient gene diagnosis technology for clinically evaluating the diagnosis, prognosis and reappearance period of acute lymphocytic leukemia.
Owner:李艳 +1
Bovine bone collagen peptide capable of improving immunity, and preparation method thereof
InactiveCN108785660AImprove immunityStimulate endocrine immune functionPowder deliveryPeptide/protein ingredientsLife qualityAdditive ingredient
The invention provides a bovine bone collagen peptide capable of improving immunity, and a preparation method thereof. The bovine bone collagen peptide capable of improving immunity comprises bovine bone collagen peptide powder, and a certain amount of a traditional Chinese medicine component, is capable of invigorating energy and blood, nourishing spleen and stomach, nourishing kidney, strengthening healthy, securing the exterior, and expelling evil; synergistic effect of the added traditional Chinese medicine component with the bovine bone collagen peptide is achieved, absorption is more convenient, the effect is much better than the superposition of the effects of single component medical ingredients, human self endocrine immunity function is activated, the bovine bone collagen peptideis suitable for people with poor immunity or chronic diseases to eat for long terms, is capable of improving immunity, conditioning chronic diseases, and improving life quality and working efficiency.
Owner:石丰
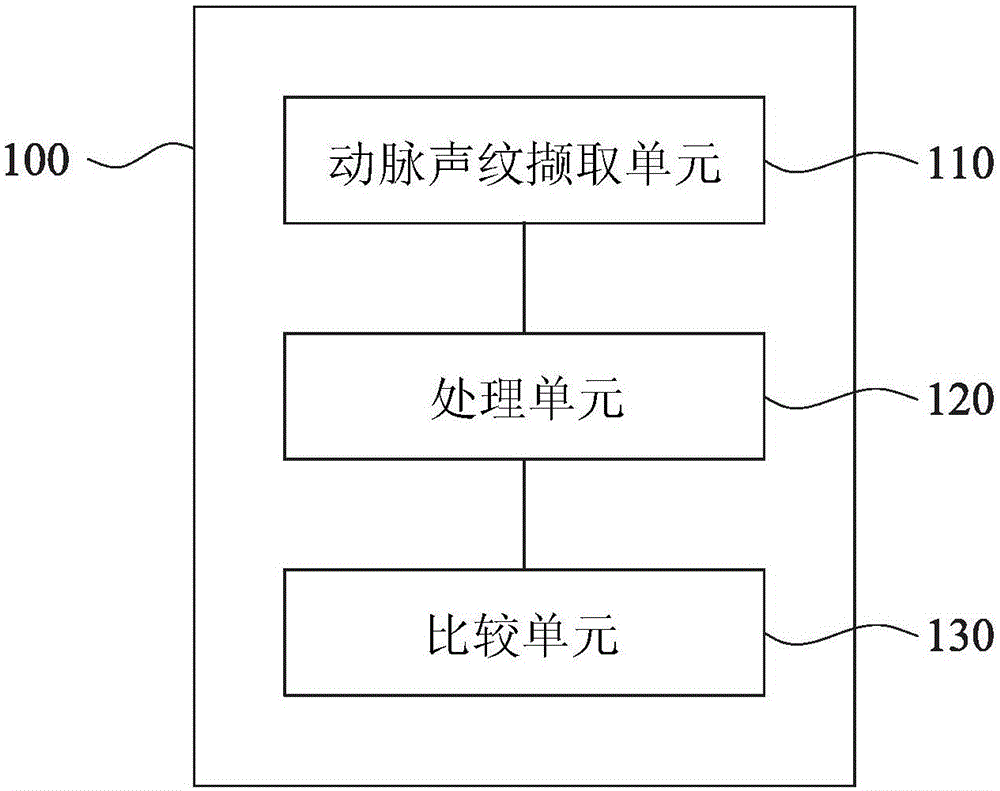
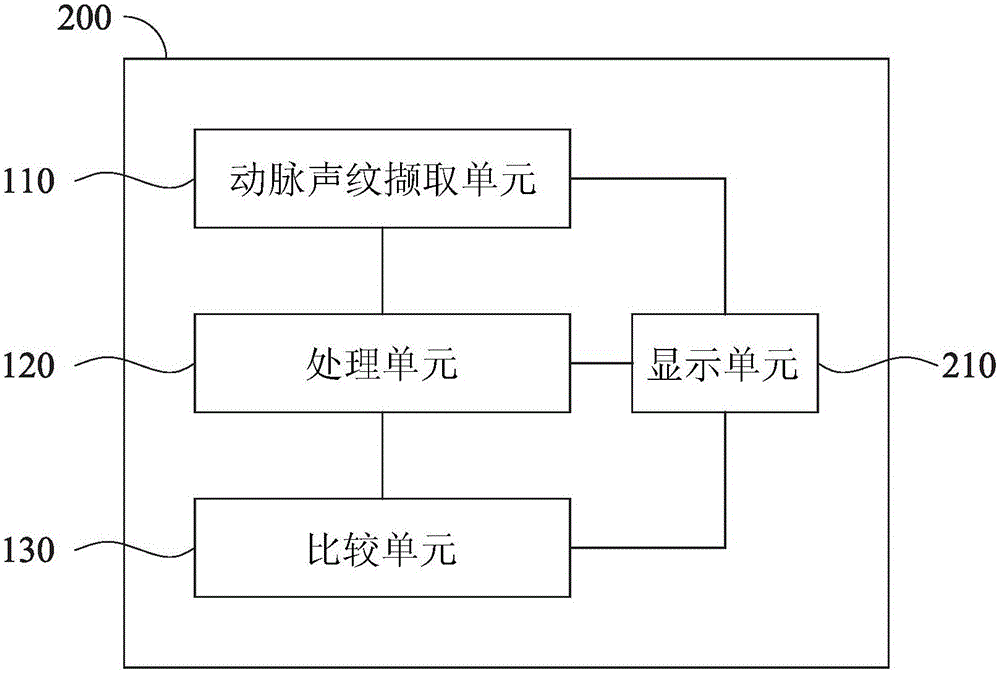
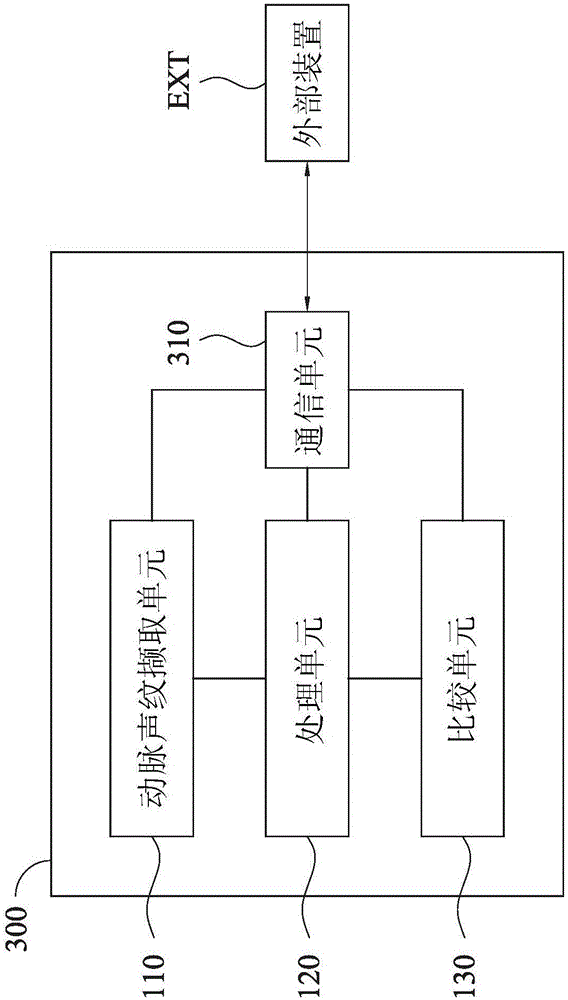
Health management device, health management method and health management system
InactiveCN106530165AReal-time understanding of health statusReduce hardware costsData processing applicationsBiological bodyLong-term care
The invention discloses a health management device, a health management method and a health management system. The health management device comprises an arterial voiceprint capturing unit, a processing unit and a comparison unit. The arterial voiceprint capturing unit is used for capturing an arterial voiceprint signal of an organism. The processing unit is used for processing the arterial voiceprint signal, and acquiring physiological data of the organism based on the arterial voiceprint signal. The comparison unit is used for comparing the physiological data with reference physiological data, and generating a health status report. According to the health management device, the health management method and the health management system, the health status report is obtained through capturing the arterial voiceprint signal and directly processing the arterial voiceprint signal, the user can be informed about own health status immediately, the hardware cost can be reduced, and the operation can be simplified. The health management device, the health management method and the health management system can be applied to many respects, such as long-term care for chronic patients, care for the elderly, care for pregnant women, TCM pulse diagnosis and the like, or applied to the evaluation of the effect of treatment or prescription.
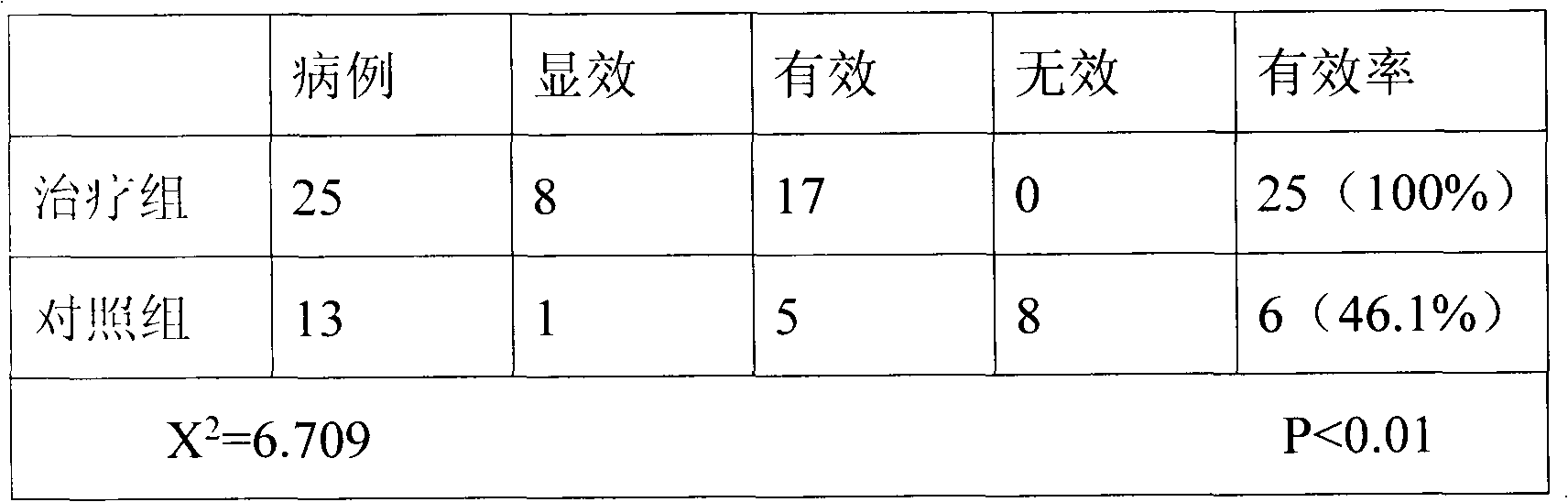
Owner:李冠瑢
Jam prepared from blueberry fruits, Chinese yams, coix seeds and red beans
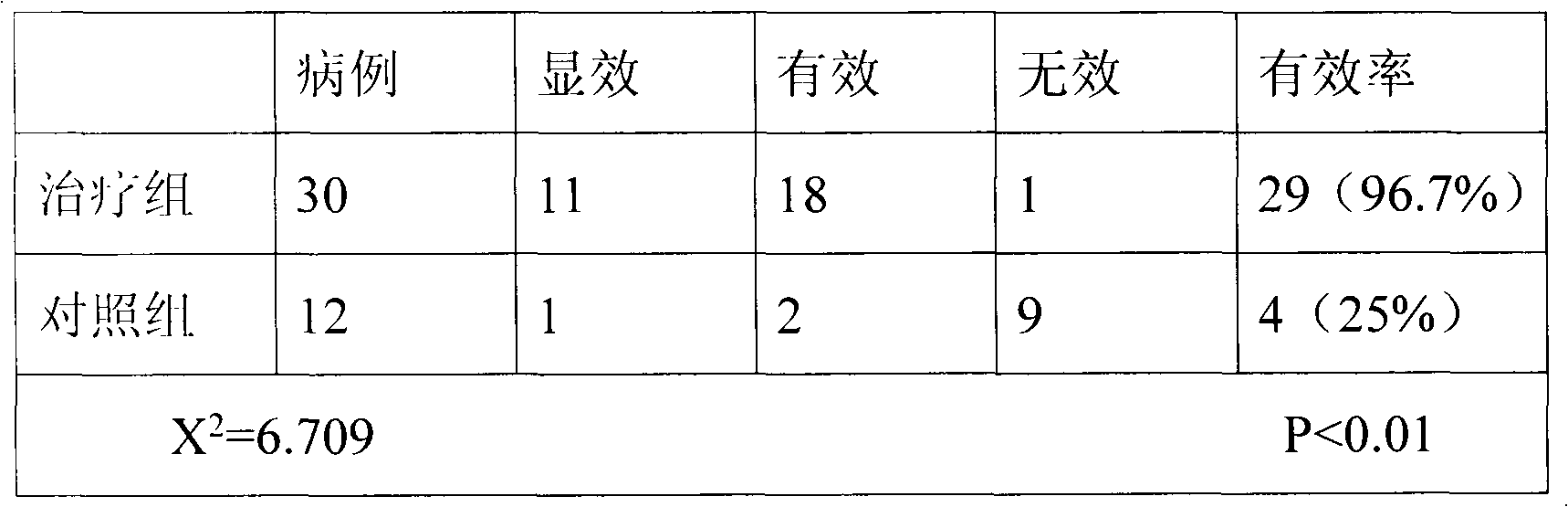
The invention relates to jam prepared from blueberry fruits, Chinese yams, coix seeds and red beans. The jam is prepared from the following raw materials of blueberry fruits, Chinese yams, coix seeds, red beans, crystal sugar and an appropriate amount of drinking water. A production method of the jam comprises the following steps of preparing materials, making blueberry fruit juice, grinding the Chinese yams, the coix seeds and the red beans into soybean milk, performing heating, performing boiling, performing inspection, performing disinfection and performing bottling. After the jam is eaten by 48 clinical chronic patients for 42 days, results indicate that 8 patients are healed, the healing proportion is 17%, the jam is effective for 12 patients, the effective proportion is 25%, 25 patients after eating the jam turn good, the proportion of turning good is 52%, the jam is ineffective to 3 patients, the ineffective proportion is 6%, and the total effective rate is 94%. Results prove that the health-care jam is a new health-care food variety being unique in flavor, moderate in sour and sweet degrees, delicious and rich in nutrition, has functions of being health-care and assisting in treating chronic diseases, can meet new pursuit of people for health-care foods, and is suitable for the chronic patients and people being desirable to build the body and achieve the effect of health care to eat for a long term.
Owner:申小维
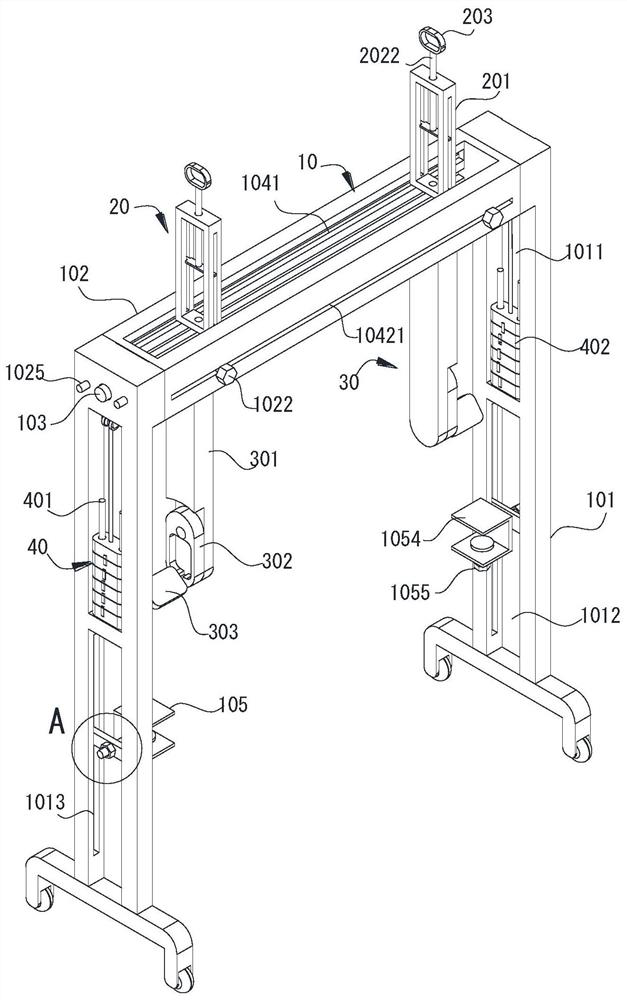
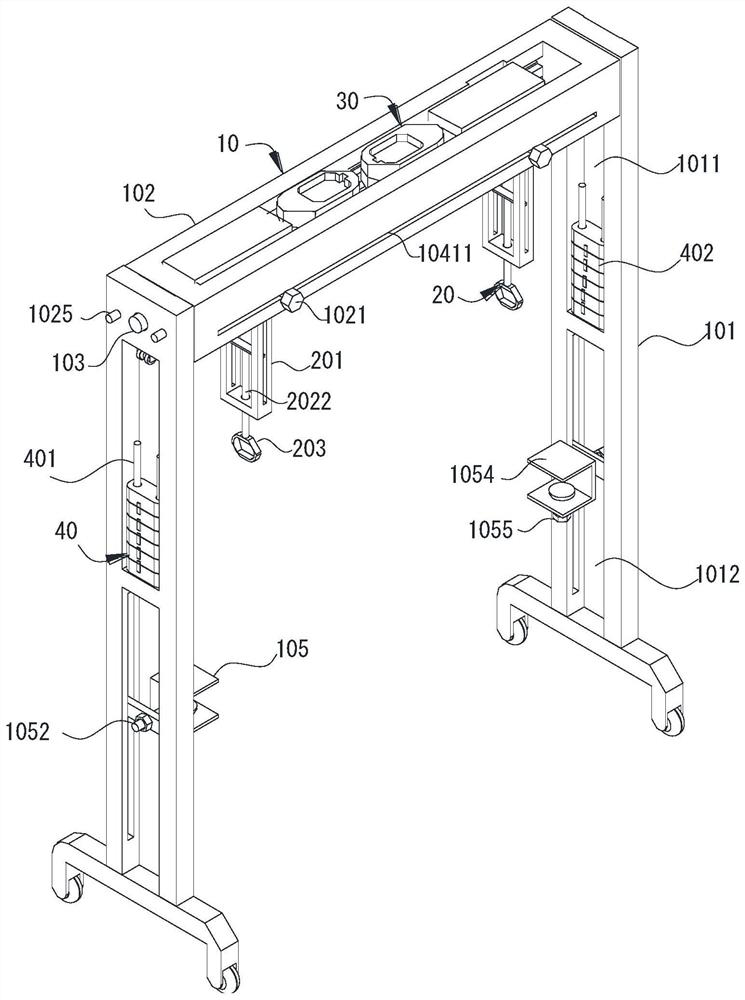
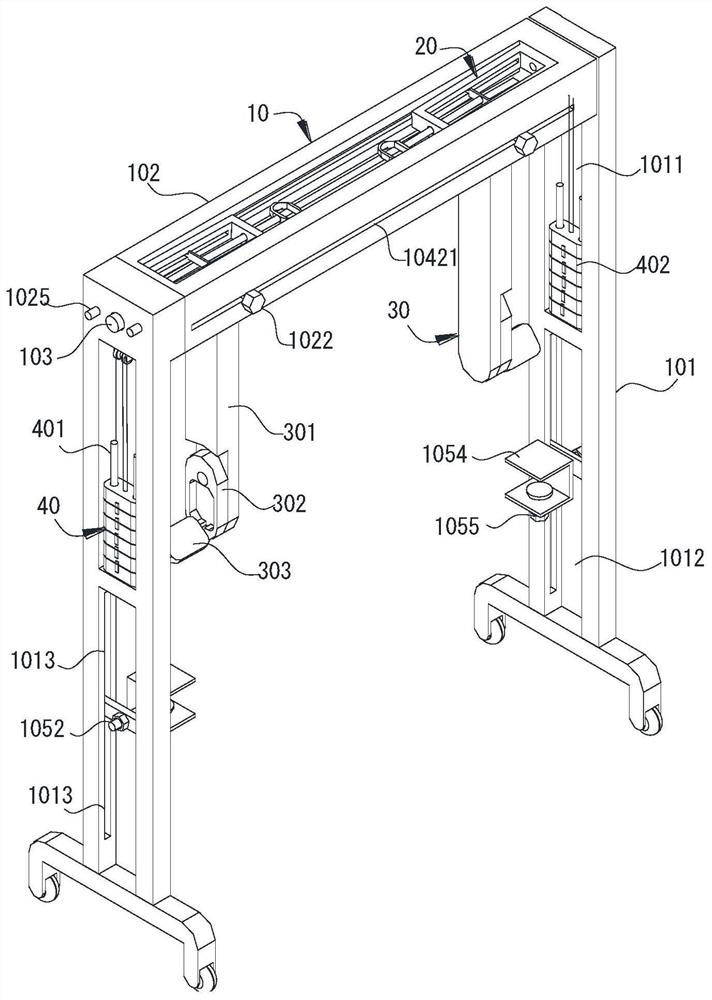
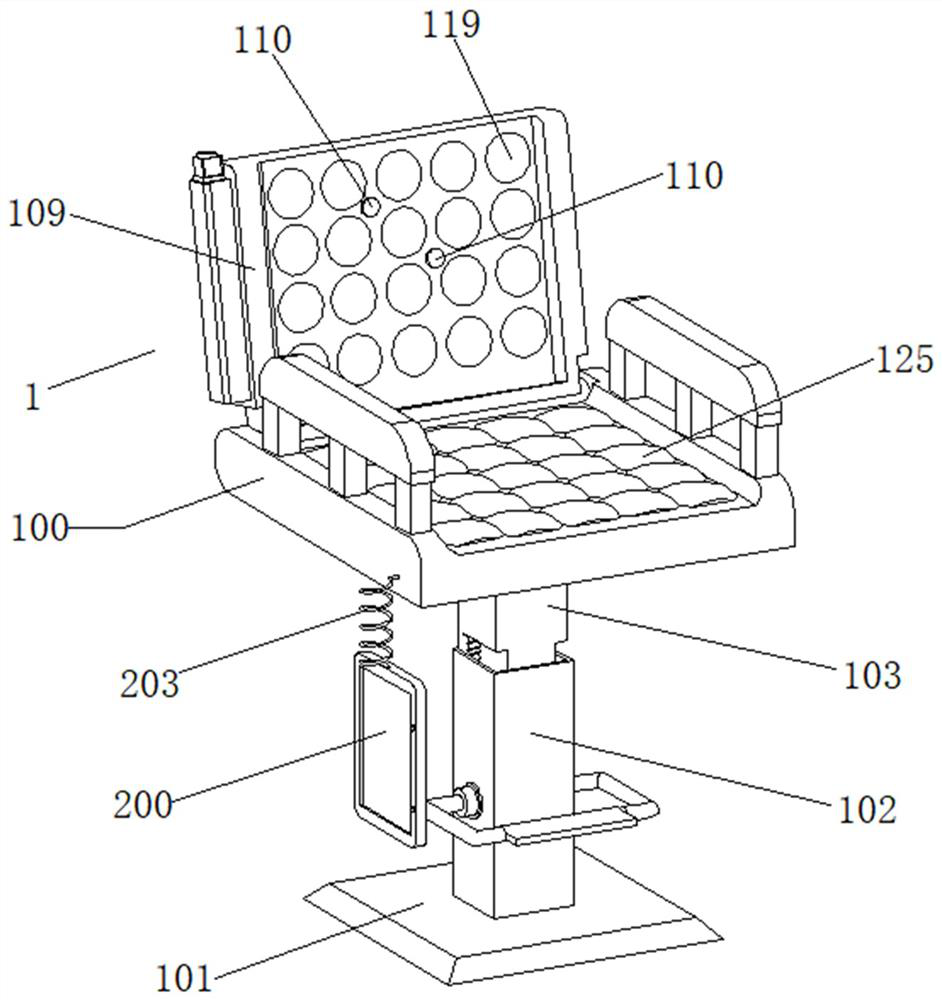
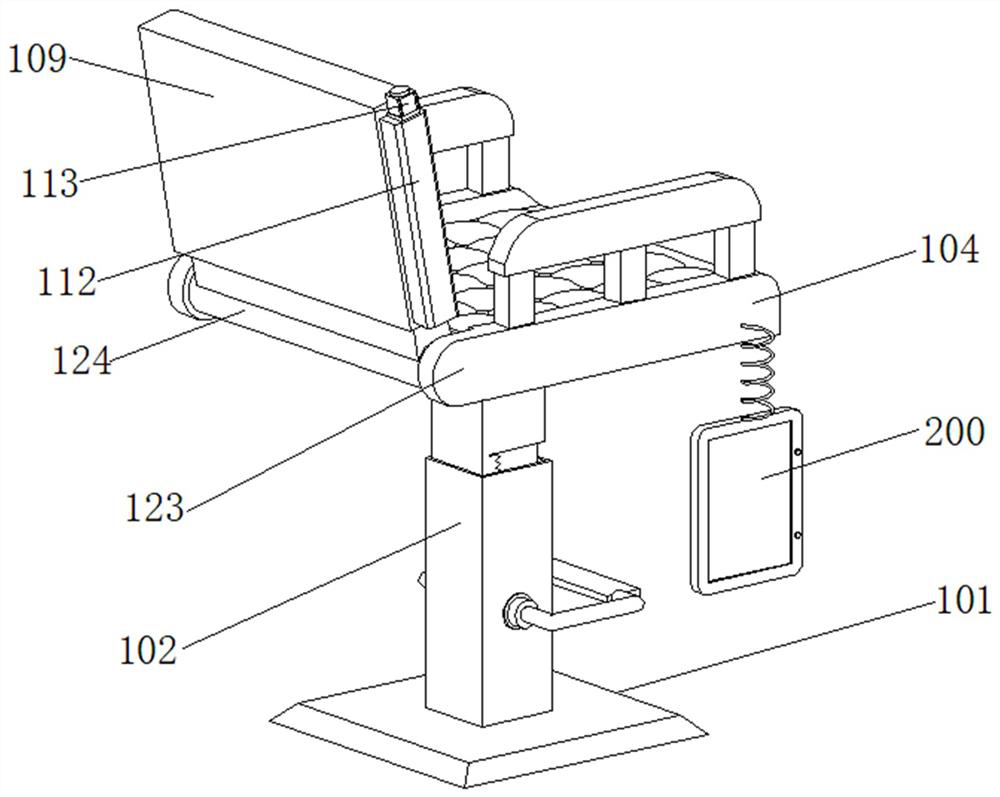
Rehabilitation nursing frame for chronic patients
A rehabilitation nursing frame for chronic patients is disclosed. The rehabilitation nursing frame comprises a supporting frame, two hand exercising components, two leg exercising components and two balance weight components, wherein the supporting frame comprises two supporting rods and a cross beam; the two supporting rods are oppositely arranged in the left-right direction; the two ends of the cross beam are connected to the upper ends of the two supporting rods through rotating shafts respectively; the cross beam can rotate along the axis of the rotating shafts, the lower portions of the two supporting rods are each provided with a fixing piece used for fixing the supporting frame to the bed edge; the two hand exercising components are arranged above the cross beam in a left-right opposite mode and used for exercising the hands of a patient; the two hand exercising components can be rotationally stored in storage grooves; the two leg exercising components are arranged below the cross beam in a left-right opposite mode, are used for exercising legs of a patient, and can be rotationally stored in the storage grooves; the two balance weight components are arranged in the middles of the two supporting rods respectively and used for adjusting resistance of the two hand exercising components and the two leg exercising components. The rehabilitation nursing frame is convenient to use and good in exercise effect and can effectively improve the physical rehabilitation progress of the patient.
Owner:PEKING UNIV SHENZHEN HOSPITAL
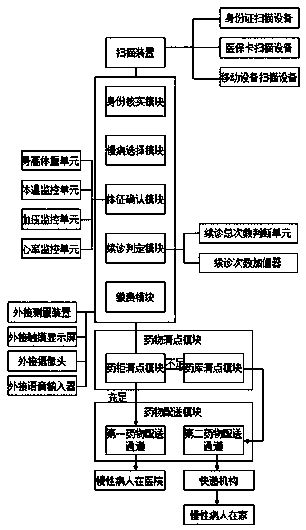
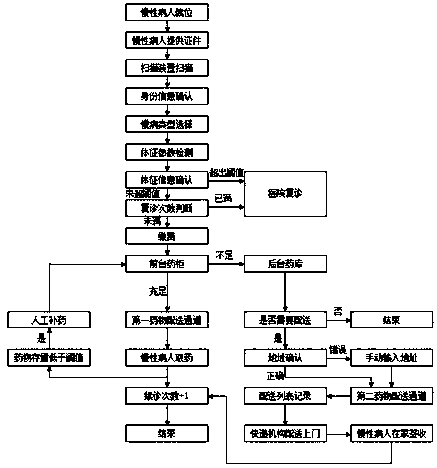
Automatic renewal and drug collection method suitable for chronic patients
ActiveCN108550397ARealize automatic renewalRealize automatic paymentMedical automated diagnosisDrug referencesPaymentMedicine
The invention relates to an automatic renewal and drug collection method suitable for chronic patients. The method is advantaged in that automatic renewal, automatic payment and automatic drug collection of the chronic patients can be realized, utilization efficiency of public resources of hospitals is improved, and the condition of the chronic patients is kept to be stable.
Owner:AFFILIATED HOSPITAL OF JIANGSU UNIV
Effervescent tablets with anti-fatigue effect and preparation method of effervescent tablets
InactiveCN104432065AEasy to takeTake resistancePharmaceutical delivery mechanismAntinoxious agentsSodium bicarbonateEffervescent tablet
Owner:郑州拓洋生物工程有限公司
Traditional Chinese medicine composition for improving immunity
InactiveCN105664102AImprove immunityGood effectAnthropod material medical ingredientsOintment deliveryLycium barbarum fruitSemen
The invention provides a traditional Chinese medicine composition for improving immunity. The traditional Chinese medicine composition is mainly composed of, by weight, bee pollen, lemon, red dates, fruits of Chinese wolfberry, fresh ginger, orange peel, fructus alpiniae oxyphyllae, endothelium corneum gigeriae galli, radix puerariae, semen ginkgo, folium mori, ginseng and the like. The traditional Chinese medicine composition can effectively improve immunity of the human body and is safe, unbiased and suitable for being taken by crowds with low immunity or chronic patients; besides, the invention further provides traditional Chinese medicine paste and a preparation method of the traditional Chinese medicine composition, the traditional Chinese medicine paste is stable in medicine, high in concentration, convenient to take and convenient to carry, the method that raw materials are processed into condensed juice and traditional Chinese medicine powder ground to be superfine is adopted so that the loss of nutrients can be reduced, the pesticide effect is better, and the taste is better.
Owner:BEIJING YUXINTANG BIOTECH CO LTD
Chronic disease follow-up tracking method
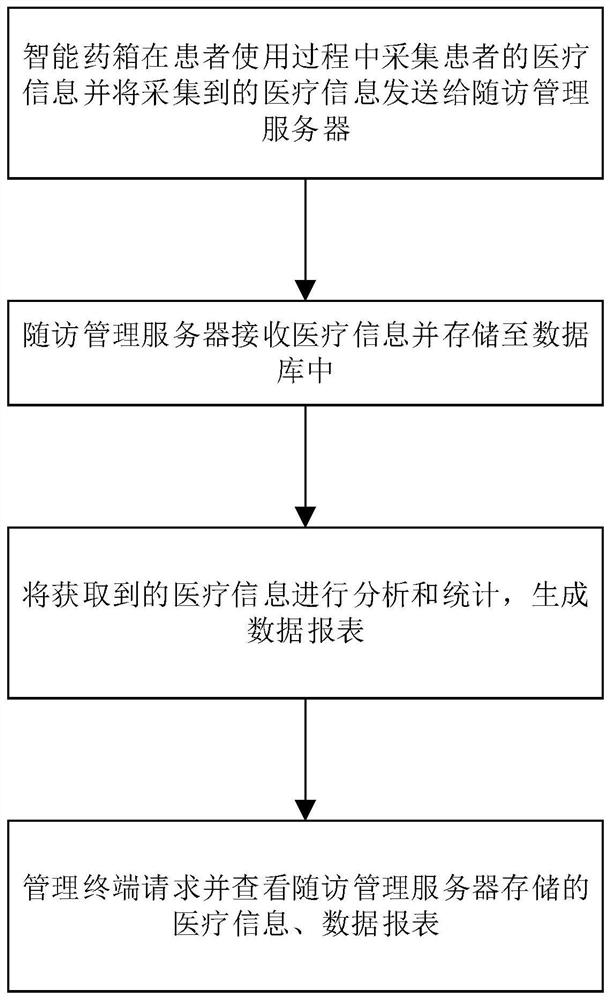
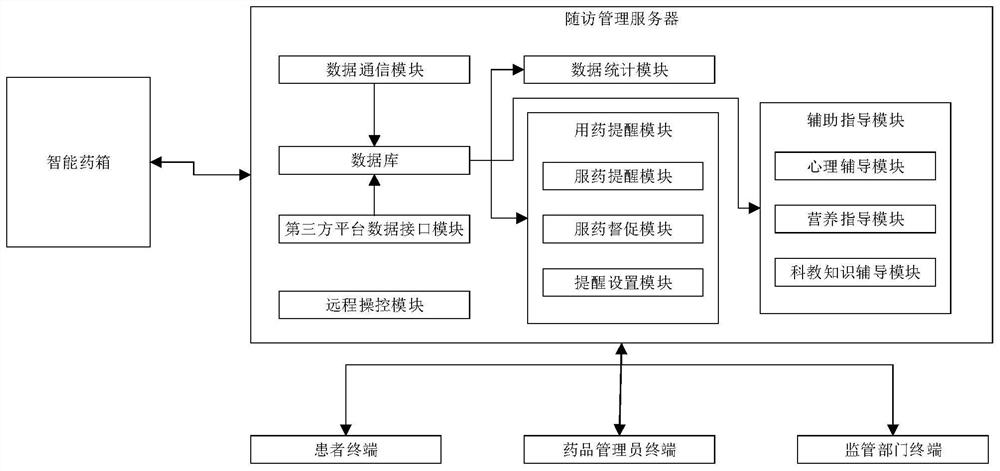
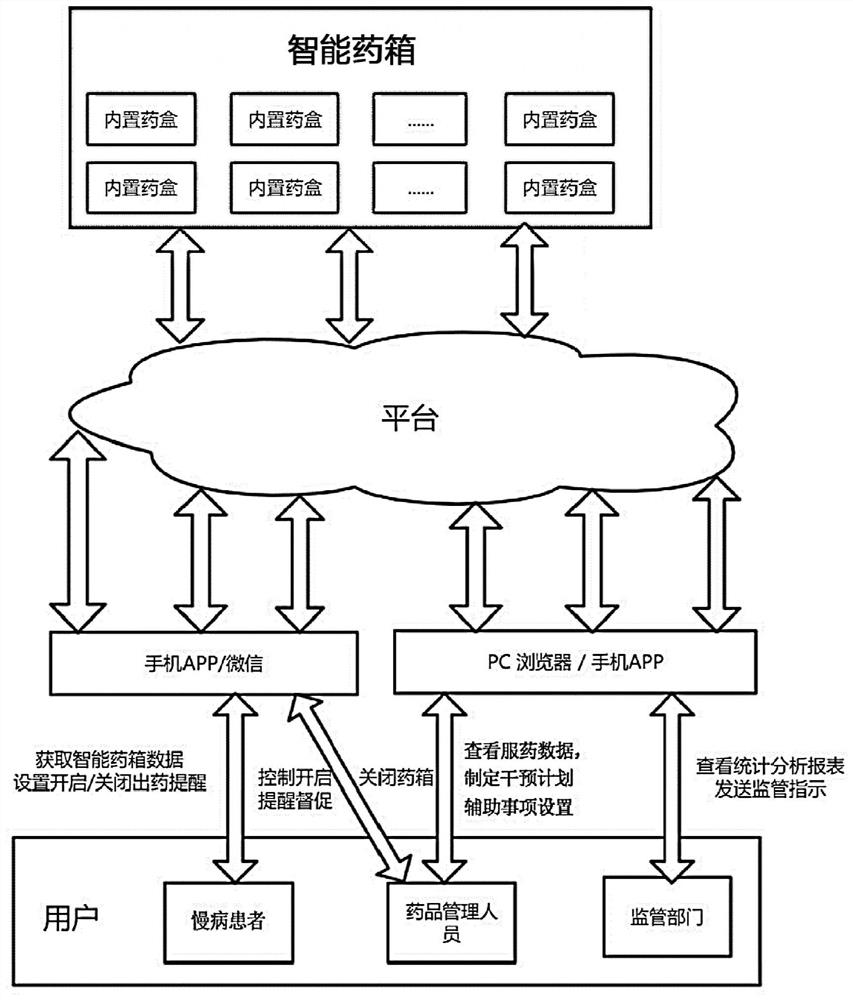
The invention relates to the technical field of medical follow-up visit, in particular to a chronic disease follow-up visit tracking method. The method comprises the steps that an intelligent medicine kit collects medical information of a patient and sends the collected medical information to a follow-up visit management server, wherein the medical information comprises medication data; the follow-up management server receives the medical information and stores the medical information in a database; the management terminal requests and checks the medical information stored in the follow-up management server, the medication data comprises medication time, dosage, medication name and medication adverse reaction data, and the medication adverse reaction data comprises adverse reaction attack time, adverse reaction expression and adverse reaction duration. According to the chronic disease follow-up visit tracking method provided by the invention, various medication data of the patient can be acquired in real time, the objectivity and authenticity of the follow-up visit data are improved, timely and accurate illness state judgment and analysis can be provided for a medical team through the acquired various data information, and a data support is provided for precise treatment of a chronic patient group.
Owner:重庆未来健康管理有限公司 +1
Kidney and yang tonifying medicine and preparation method thereof
InactiveCN103505705ADefinite curative effectBalanced effectDigestive systemPlant ingredientsWhite blood cell numberGLYCYRRHIZA EXTRACT
The invention discloses a kidney and yang tonifying medicine which comprises active components and accessories and is characterized in that the active components include 160-240 parts of herba epimedii, 160-240 parts of radix acanthopanacis semticosi, 40-60 parts of dog kidney, 160-240 parts of radix ophiopogonis, 160-240 parts of black bean, 80-120 parts of Chinese date, 80-120 parts of radix aconiti carmichaeli (prepared), 80-120 parts of cinnamon, 80-120 parts of dried ginger, 80-120 parts of liquorice and 15-25 parts of ginseng stem leaf total saponins all by weight. The medicine can be prepared into granules, tablets, capsules and the like. The composition disclosed by the invention has the functions of tonifying the kidney and yang, warming meridian / channel and replenishing qi and strengthening sinew and bones, and is applied to the 'deficiency of kidney yang' symptom caused by excessive consumption of physical strength, the 'deficiency of kidney yang' symptom of a chronic patient, and the reduction of leucocytes, hemachrome, blood platelets and the like caused by 'deficiency of kidney yang'.
Owner:SHAANXI PANLONG PHARMACEUTICAL GROUP LIMITED BY SHARE LTD
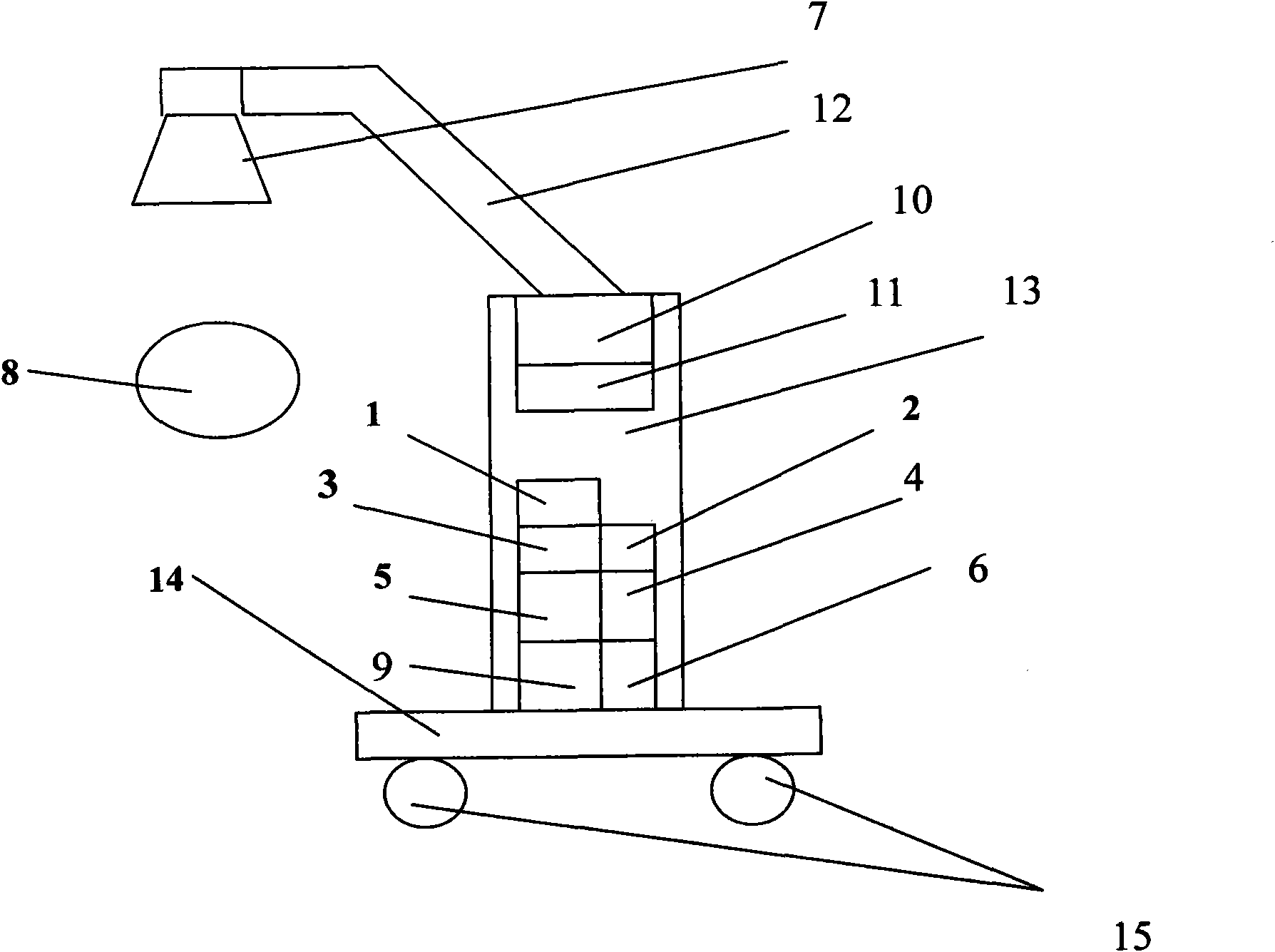
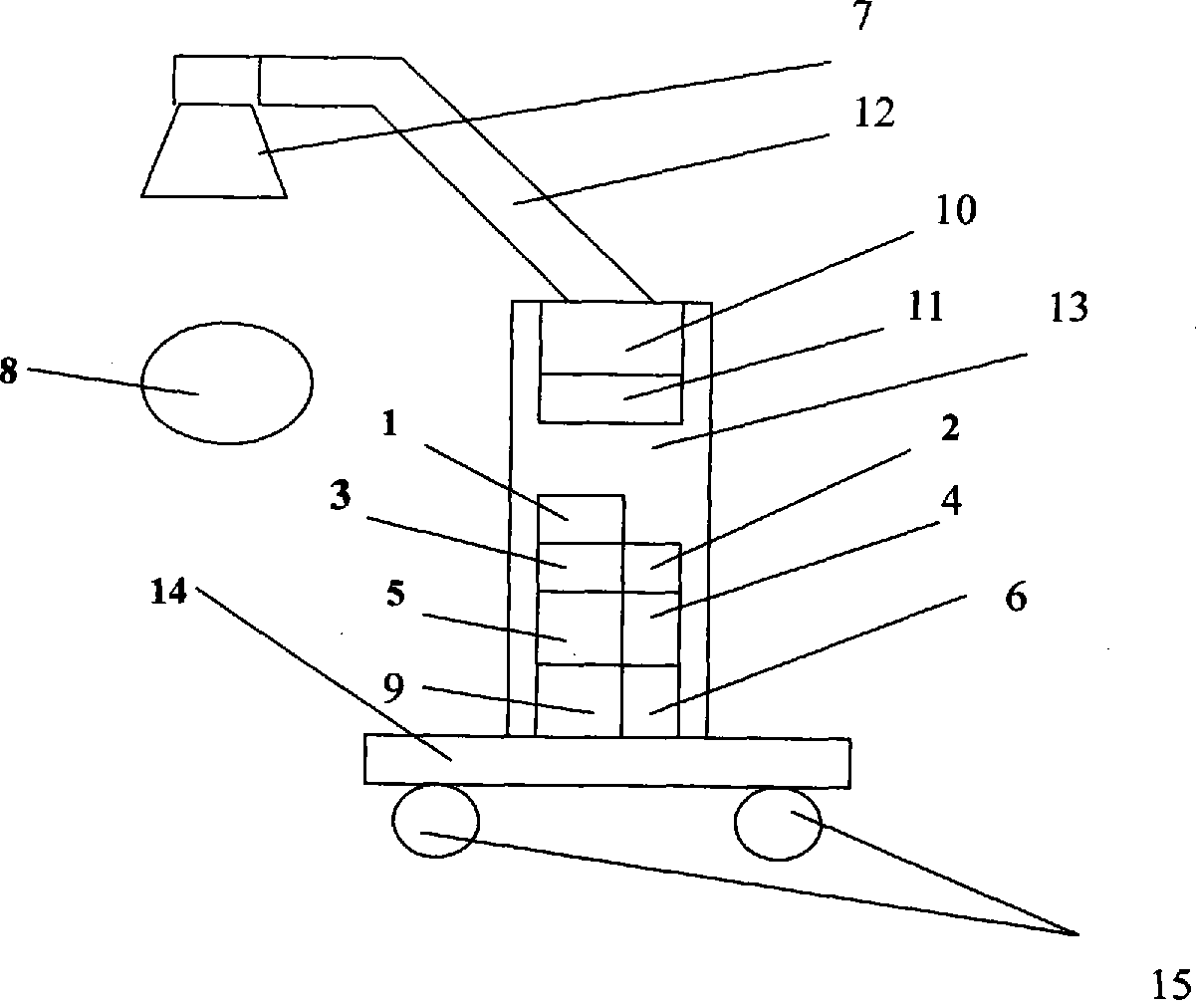
Near infrared ray biological pulse therapeutic apparatus
InactiveCN102580253ANo side effectsAlleviate cirrhotic liver diseaseLight therapySide effectInfrared lamp
The invention relates to a near infrared ray biological pulse therapeutic apparatus which comprises a near infrared lamp, a movable arm, a frame, a base, an electronic circuit, and a power circuit and is characterized in that the near infrared lamp is connected with one end of the movable arm, the other end of the movable arm is connected with the upper end of the frame, the lower end of the frame is mounted on the base of a trundle, the electronic circuit and the power circuit are mounted inside the frame, and the electronic circuit, the power circuit and the near infrared lamp are in electric connection. The near infrared ray biological pulse therapeutic apparatus can quickly relieve and effectively cure various paints, is suitable for rehabilitation of medicochirurgic patients, is noninvasive, painless, safe and effective, has no toxic side effect of medicine, is convenient to operate, is economical and practical, not only suitable for pains and rehabilitation in hospitals, sanitariums and sports teams, and is particularly convenient for household aged and chronic patients to use.
Owner:蒋德林
Intelligent nutrition management service plate special for chronic patient
InactiveCN112331301AEasy to controlReduce the problem of wrong intake of sugarNutrition controlMedical equipmentSugar intakeNutrition
The invention relates to the technical field of diet management, and particularly discloses an intelligent nutrition management service plate special for a chronic patient. The intelligent nutrition management service plate comprises a tray and dining lattices, a plurality of dining lattices capable of being taken out are arranged on the tray, a main control unit, a weighing module, a voice interaction module, a communication module and a power supply module are arranged in the tray, and an indicator lamp is arranged on the outer side of the tray; according to the invention, the weight of meals in the dining lattices can be collected through the weighing module, the varieties of the meals of a patient are collected through the voice interaction module and uploaded to the cloud server to beanalyzed, the dining situation, the nutrient content and the sugar intake can be accurately controlled, and the health hidden danger caused by diet of the diabetic patient is reduced from the source.
Owner:深检数据科技(江苏)有限公司
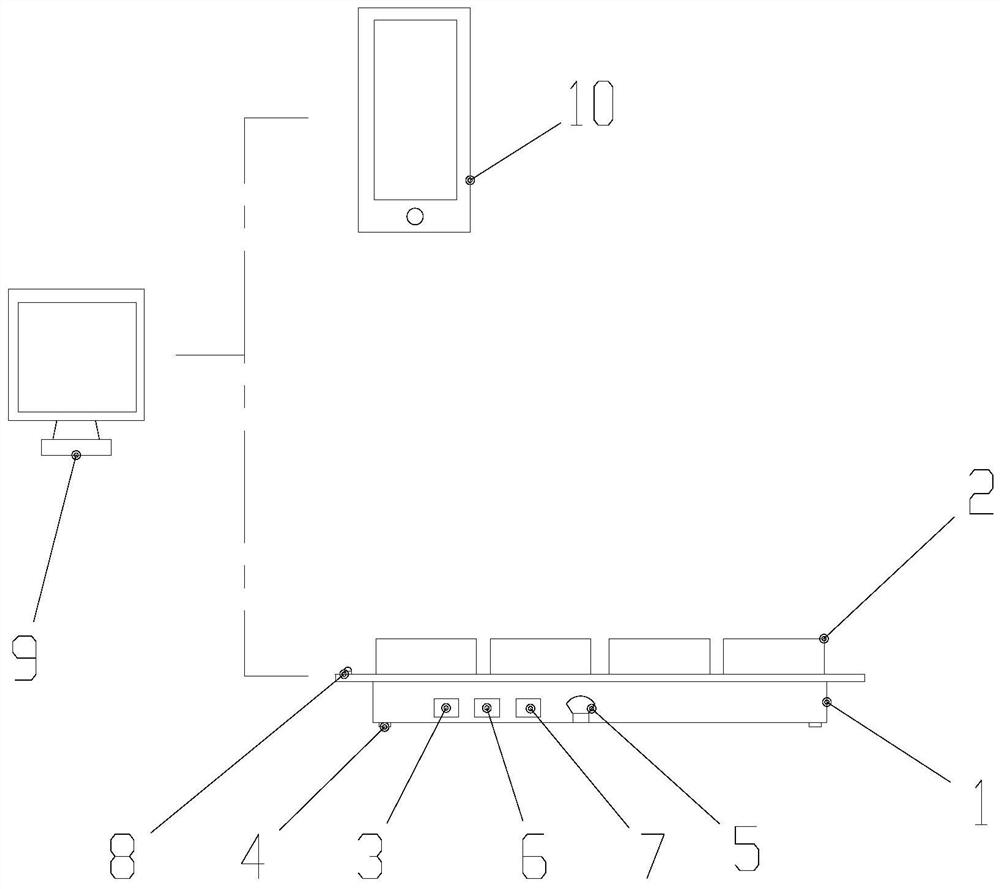
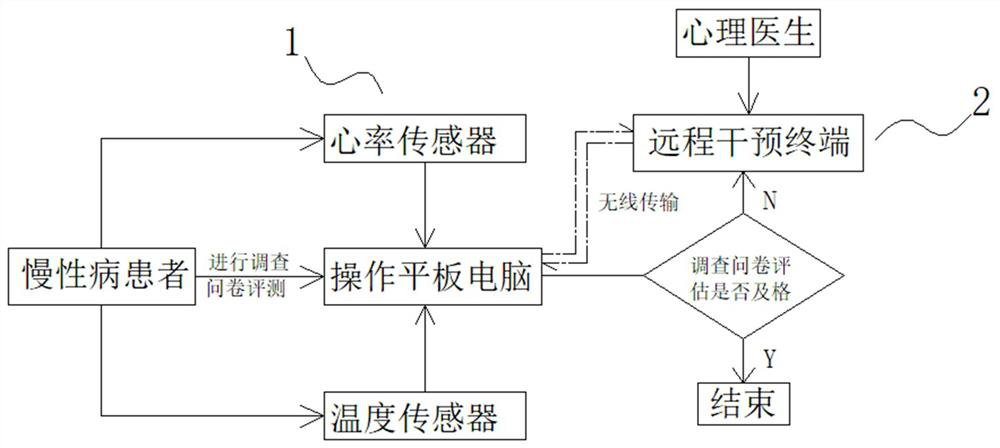
Cerebral stroke patient psychological assessment system based on Internet terminal automatic data acquisition
PendingCN113499072AHelp with assessmentAids in channelingParticular environment based servicesSensorsPhysical medicine and rehabilitationThe Internet
The invention relates to the technical field of psychological management, and particularly discloses a cerebral apoplexy patient psychological assessment system based on Internet terminal automatic data acquisition. The cerebral apoplexy patient psychological assessment system includes a psychological assessment device and a remote intervention terminal, the psychological assessment device and the remote intervention terminal are connected through a wireless network, the psychological assessment device comprises an assessment data acquisition seat body and an operation tablet computer, and the operation tablet computer is connected with the acquisition seat body through an elastic connecting line. A related questionnaire about binary pressure evaluation is also arranged in the operation tablet computer; the design of the whole device disclosed by the invention effectively makes up the blank of lack of binary pressure assessment equipment in the market, can contribute to psychological assessment and dredging of chronic patients, and is excellent in use effect and diversified in function.
Owner:ZHENGZHOU UNIV
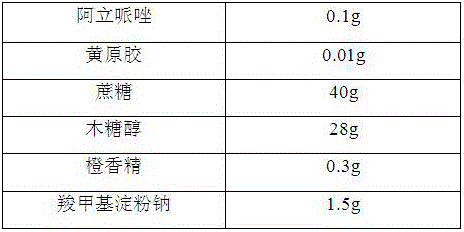
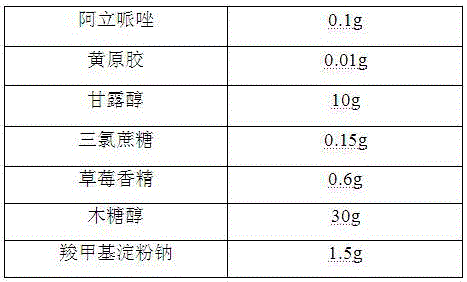
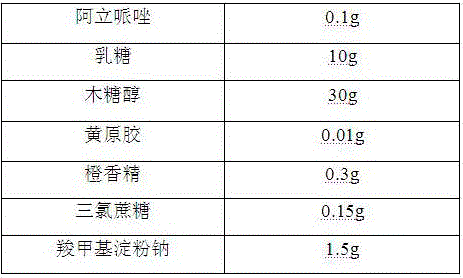
Oral aripiprazole liquid dry suspension agent and preparation method thereof
InactiveCN106389343AGood disintegrationPromote dissolutionOrganic active ingredientsNervous disorderSuspending AgentsBULK ACTIVE INGREDIENT
The invention belongs to the technical field of medicines, and relates to an oral aripiprazole liquid dry suspension agent which is used for treating schizophrenia and is effective for acute recurrent patients, chronic patients and affective schizophrenia patients, and a preparation method thereof. The medicine is prepared from active ingredients of aripiprazole and other pharmaceutically acceptable auxiliary materials of filling agents, suspending agents, corrigents, bonding agents and disintegrating agents. The oral aripiprazole liquid dry suspension agent provided by the invention solves the problems of medicine taking difficultly and poor medicine taking compliance of some patients with weak swallowing ability; in addition, the quality is enabled to be controllable; the stability is high; the taking is convenient; the mouthfeel is good; the clinic requirements can be well met. Compared with known aripiprazole tablets, the oral aripiprazole liquid dry suspension agent has the advantages of high medication speed, fast effect taking, stable absorption, high bioavailability and the like.
Owner:万全万特制药江苏有限公司
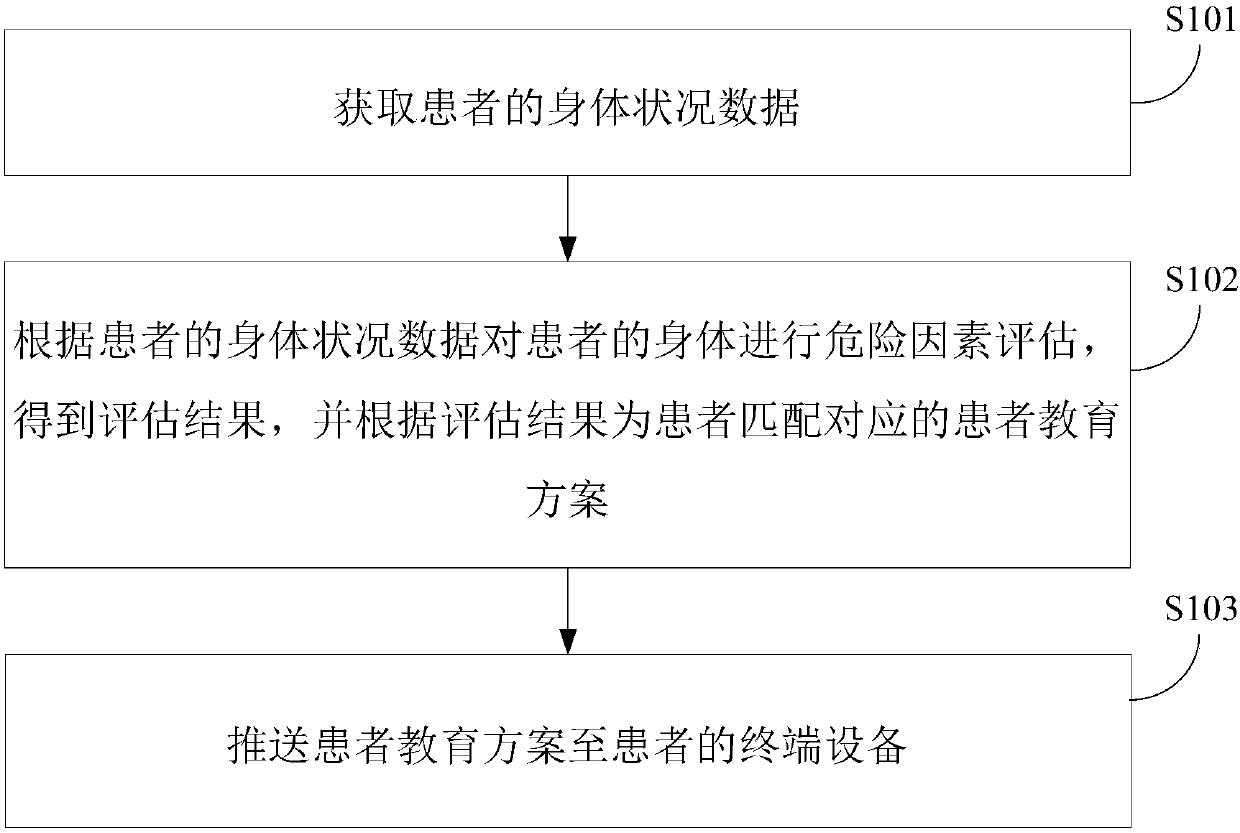
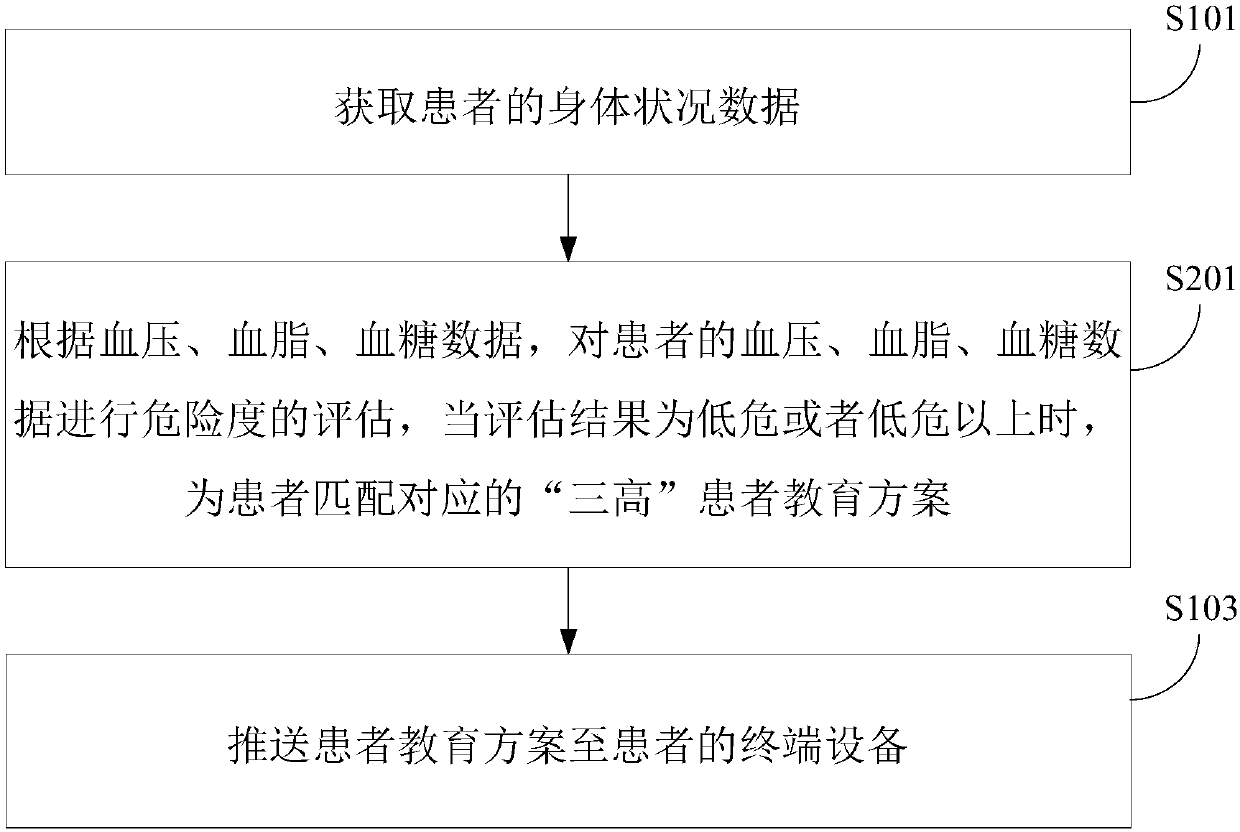
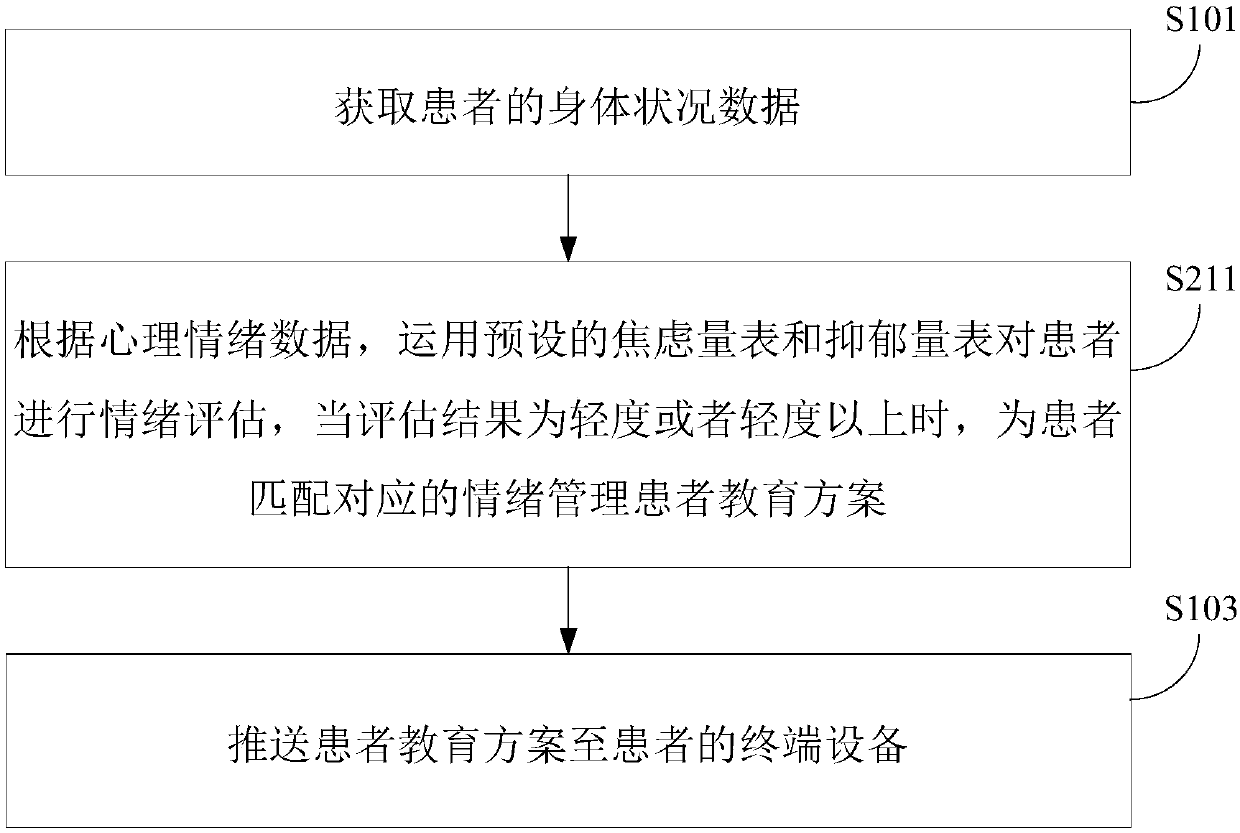
Pushing method and device of chronic patient education management scheme
InactiveCN107680681AIncrease awarenessEasy to controlData processing applicationsWeb data indexingDiseaseCognition
The invention relates to a pushing method and device for a chronic patient education management scheme. The method comprises acquiring the physical condition data of a patient; performing risk factorevaluation on the body of the patient according to the body condition data of the patient to obtain the evaluation result, and matching a corresponding patient education scheme for the patient according to the evaluation result; and pushing the patient education scheme to the terminal device of the patient. According to the technical scheme, the cognition of patients to diseases can be effectivelyimproved, so that the patient can be more actively involved in the disease management, the compliance is improved, and the disease is better controlled.
Owner:BEIJING JEEJEN ERA SOFTWARE TECH
Medication monitoring and recording method for senile chronic diseases
PendingCN111710438AImprove recognitionImprove satisfactionMedical communicationHealth-index calculationPharmacist interventionsDisease
The invention discloses a medication monitoring and recording method for senile chronic diseases, and belongs to the technical field of disease medication monitoring and recording methods. Whether from a hospital angle or from a patient angle, a pharmaceutical working mode is switched to a pharmaceutical service, and a pharmacist goes into a community from a hospital and goes into a family, so that the safe medication awareness of old chronic disease patients is improved to a great extent, the importance of guaranteeing safe medication of the patient by the pharmacist is brought into full play, the reasonable medication property of the senile chronic patient is improved by applying refined pharmacist intervention, the approval and satisfaction of the patient to the pharmacist are also improved, the economic benefits and social benefits of the patient are improved, and it is also the necessity to apply a refined pharmacist intervention pattern to facilitate pharmacist transformation.
Owner:周后凤