Patents
Literature
235 results about "Aids diagnostics" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
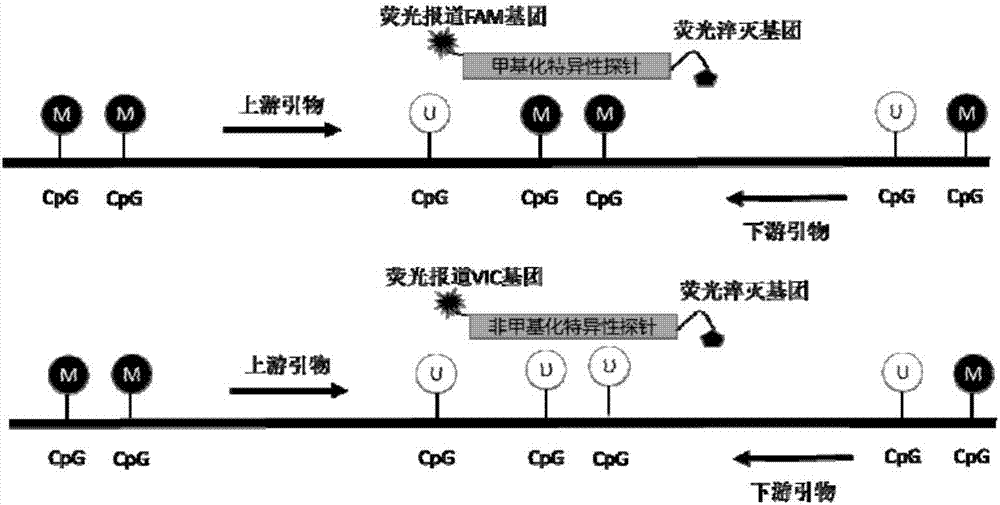
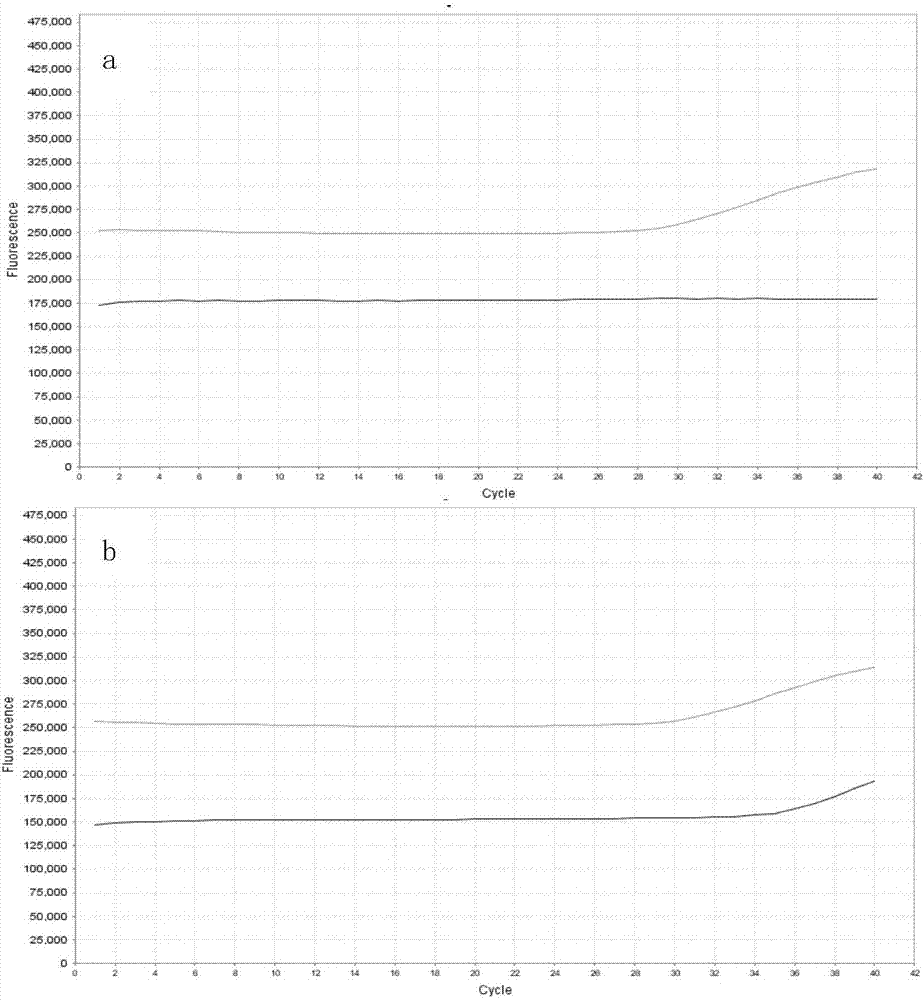
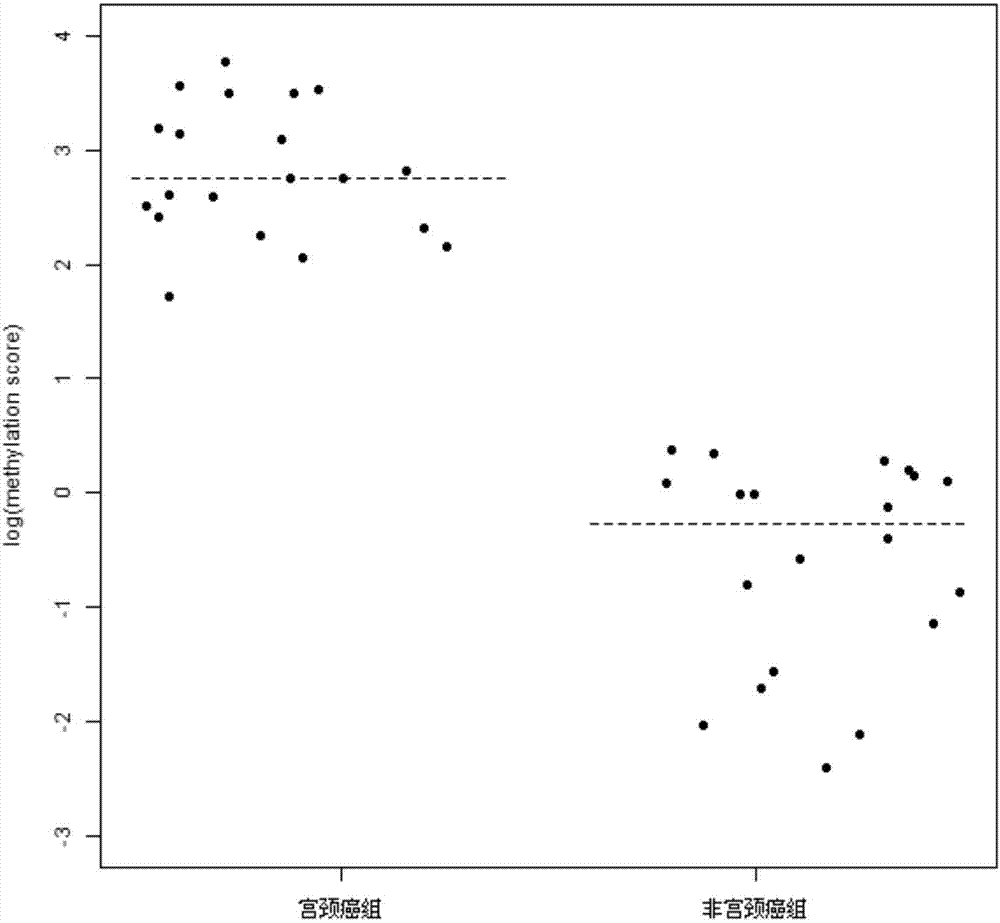
Detection primer for methylation degree of related genes of cervical cancer, probe, kit and application thereof
InactiveCN107287294AReliable resultsHigh sensitivityMicrobiological testing/measurementDNA/RNA fragmentationCervical tissueBiology
The invention discloses a detection primer for methylation degree of related genes of cervical cancer, a probe, a kit and an application thereof. The invention finds that the methylation degree of promoter zones of four genes, namely, PAX1, FAM19A4, PHACTR3 and DAPK in cervical tissue is obviously related to the cervical cancer; the detection primer can be used as a biomarker for assisting in cervical cancer diagnosis; a methylation probe and a non-methylation probe are designed, so that the influence of non-methylation DNA is avoided and the result is more reliable. Compared with the conventional cervical cancer diagnostic method, the method provided by the invention has the advantages of high speed, high sensitivity, high specificity, and the like, can timely diagnose human cervical carcinoma in early stage, can early accurate diagnose the cervical cancer and can avoid the waste of medical resources.
Owner:广州中心法则生物科技有限公司
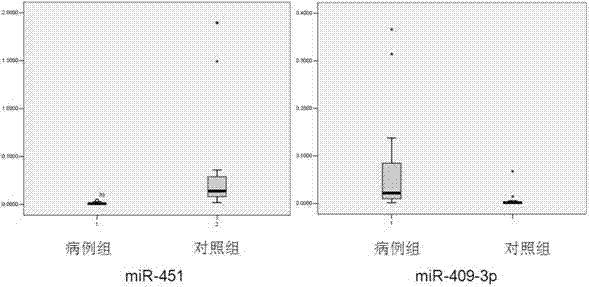
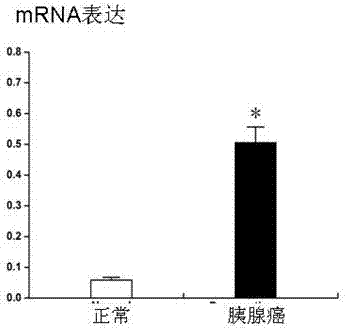
Blood serum/blood plasma micro ribonucleic acid (miRNA) marker relevant with pancreatic cancer and application thereof
ActiveCN102876676AAids in diagnosisEasy to detectMicrobiological testing/measurementDNA/RNA fragmentationBlood plasmaGenetic engineering
The invention belongs to the fields of genetic engineering and oncology, and discloses a blood serum / blood plasma micro ribonucleic acid (miRNA) marker relevant with pancreatic cancer and application thereof. The marker is formed by combining miR-451 and miR-490-3p. The marker and primers of the marker can be used for preparing a diagnostic kit and are used for the auxiliary diagnosis of pancreatic cancer.
Owner:NANJING MEDICAL UNIV
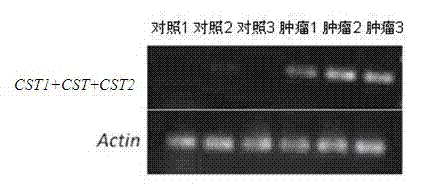
Tumor marker and application thereof to preparation of colorectal cancer diagnostic reagent
ActiveCN103740854AMicrobiological testing/measurementProtease inhibitorsDiagnosis earlyGenetic engineering
The invention belongs to the field of genetic engineering and medicines, and relates to a novel tumor marker for early diagnosis of colorectal cancer, in particular to a tumor marker related to the colorectal cancer and application thereof. The marker is a composition of CystatinSN (CST1), CystatinS (CST4) and CystatinSA (CST2). The marker as well as a primer and an antibody thereof can be applied to a diagnostic kit for the auxiliary diagnosis of the colorectal cancer.
Owner:THE THIRD AFFILIATED HOSPITAL OF SUN YAT SEN UNIV
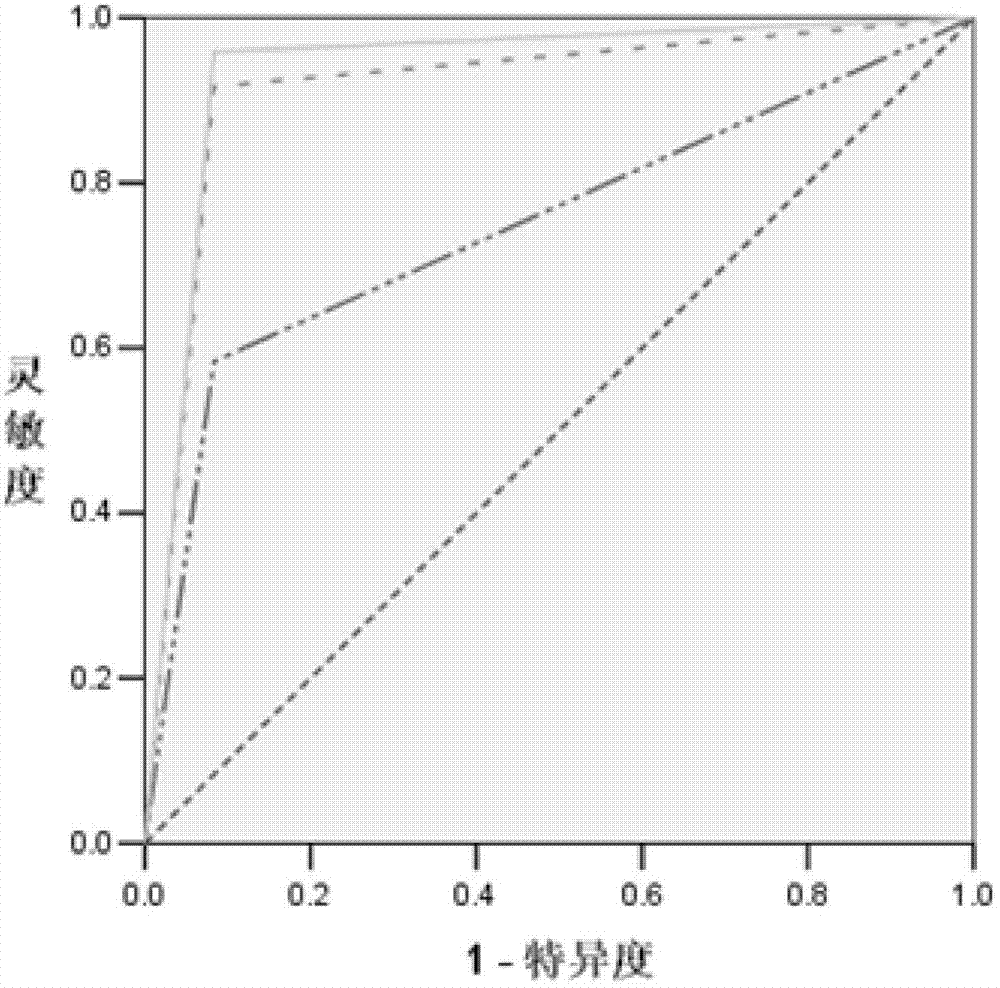
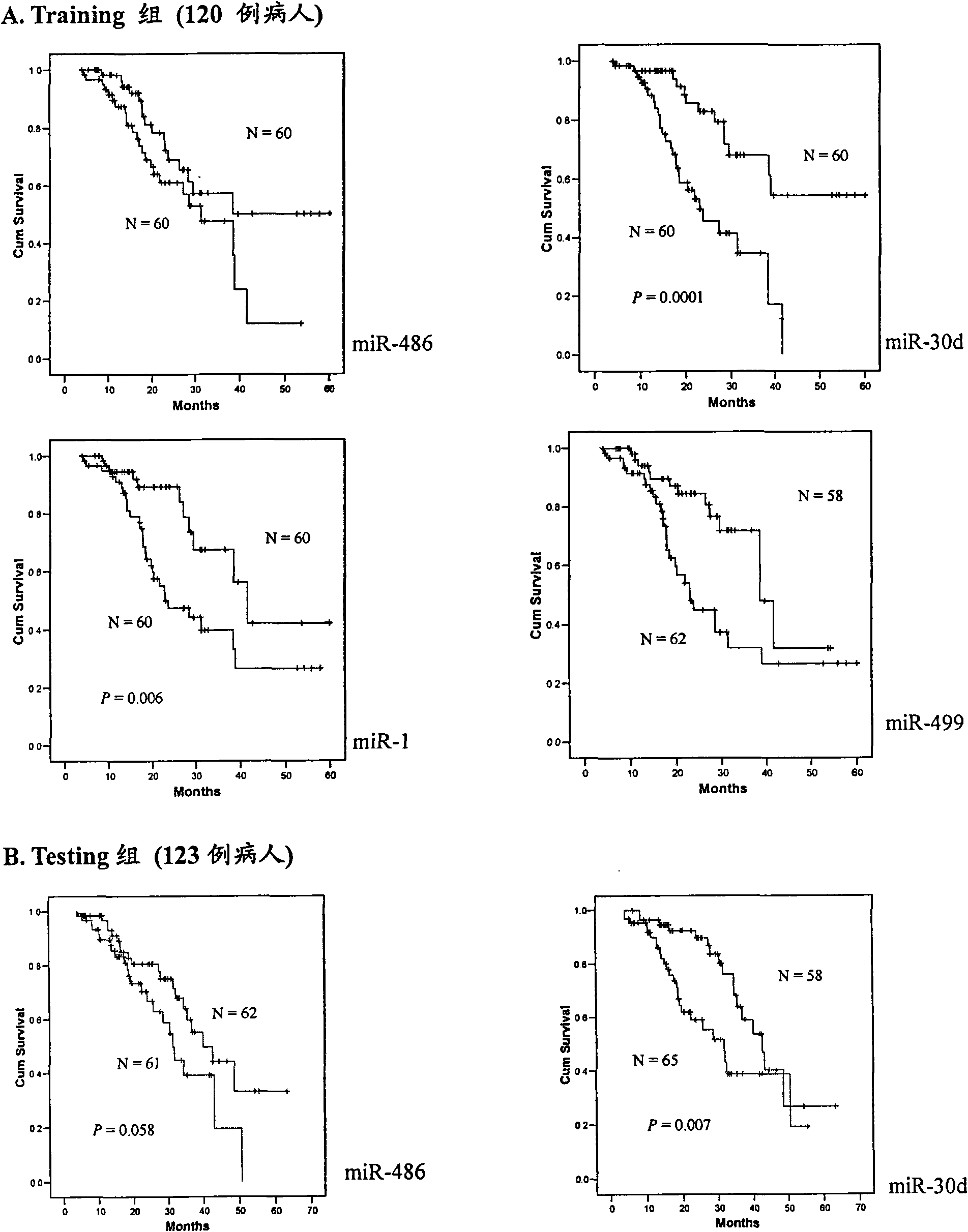
Blood serum/blood plasma miRNA marker related to non-small cell lung cancer (SCLC) prognosis and application thereof
InactiveCN101638656AIncreased sensitivityImprove featuresGenetic material ingredientsMicrobiological testing/measurementBlood plasmaGenetic engineering
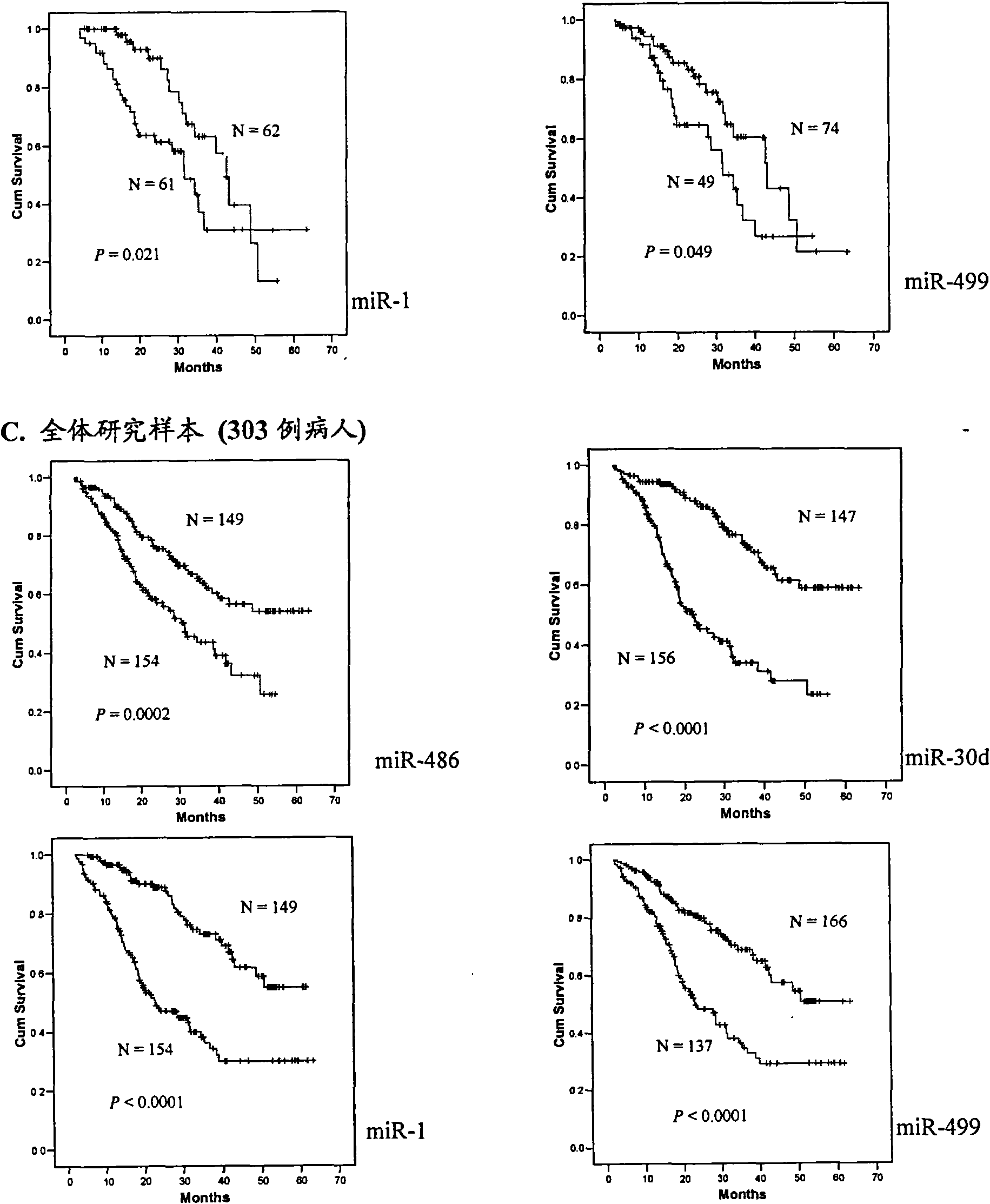
The invention belongs to the field of genetic engineering and phymatology, in particular to a blood serum / blood plasma miRNA marker related to non-small cell lung cancer (SCLC) prognosis and an application thereof. The marker is one or more of miR-486, miR30d, miR-1 or miR-499 and can be used for preparing an auxiliary diagnostic reagent kit for the non-SCLC prognosis or a medicine for treating the SCLC.
Owner:NANJING MEDICAL UNIV
Kawasaki disease classifying and predicting method based on medical data modeling
PendingCN106295229AReduce misdiagnosis rateImprove the treatment processSpecial data processing applicationsFeature screeningKawasaki disease
The invention provides a kawasaki disease classifying and predicting method based on medical data modeling. The method includes the following steps of firstly, selecting data samples, and extracting an effective sample for modeling from a sample data set; secondly, screening characteristics, and screening out 19 characteristics meeting on-site medical assistance diagnosis application from a characteristic set for establishing sample data for modeling; thirdly, establishing and evaluating a kawasaki disease classifying model. The method is used for systematical analyzing and modeling of kawasaki disease related data and giving model prediction; by means of the mode, effective assistance diagnosis can be conducted on the kawasaki disease of a patient based on the kawasaki disease data, effective prevention, interference and treatment are conducted in the early period of disease attack, and a basis is provided for the optimal treatment effect.
Owner:QINGDAO UNIV
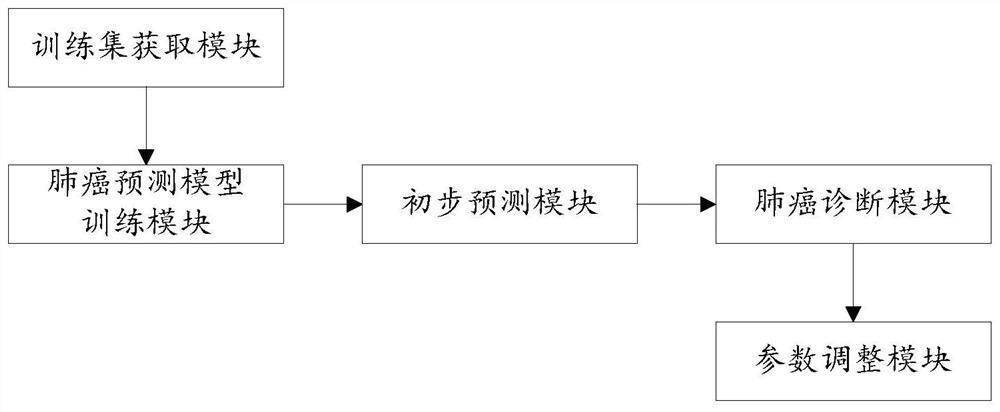
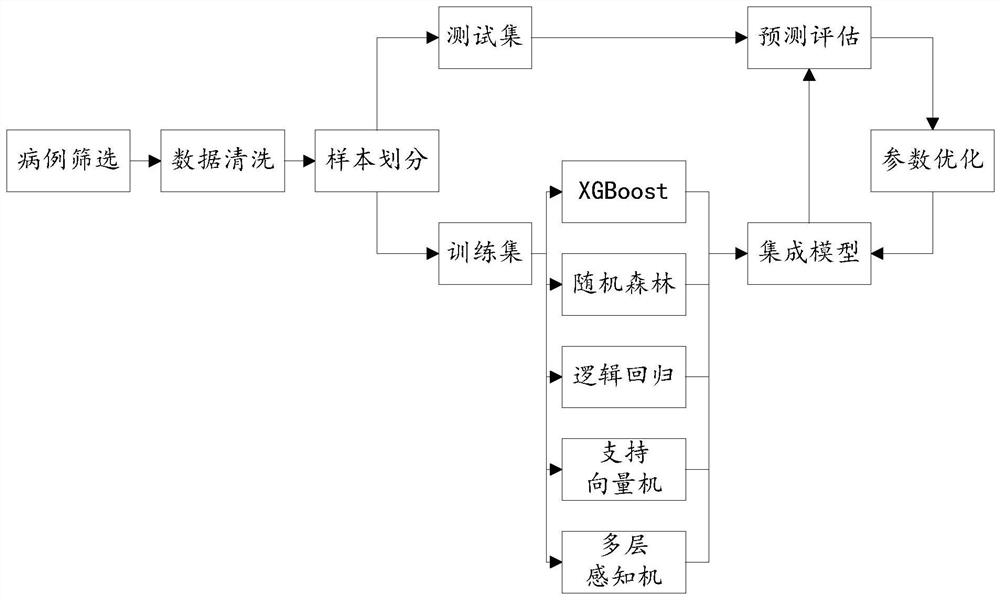
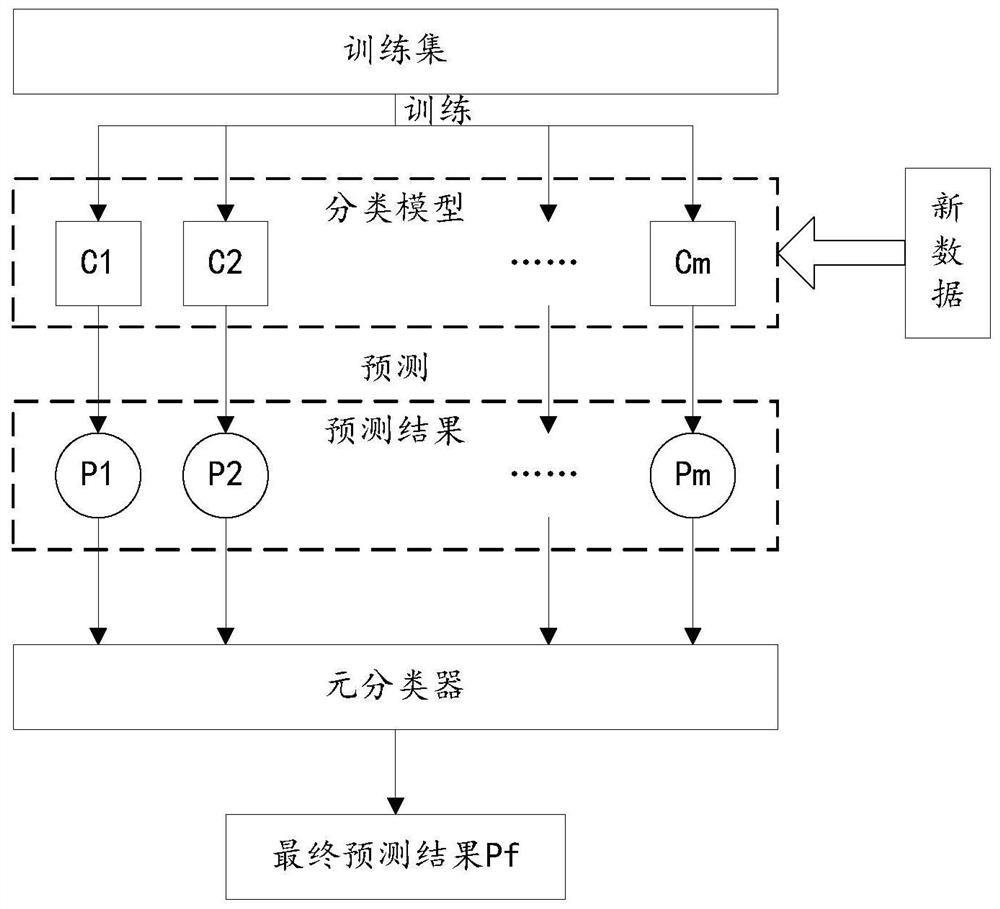
Lung cancer diagnosis system based on multiple machine learning algorithms
PendingCN112259221AEasy diagnosisImprove accuracyKernel methodsCharacter and pattern recognitionAids diagnosticsOncology
The invention discloses a lung cancer diagnosis system based on multiple machine learning algorithms, and relates to the field of medical instruments, and the system comprises a preliminary predictionmodule which is used for employing a plurality of trained lung cancer prediction models to perform lung cancer preliminary prediction processing of lung clinical data of a to-be-diagnosed patient, and obtaining a plurality of lung cancer preliminary prediction results, and a lung cancer diagnosis module used for carrying out lung cancer classification processing on the plurality of lung cancer preliminary prediction results by utilizing a trained lung cancer meta-classifier, and determining whether the patient to be diagnosed is a lung cancer patient or not. The system can be applied to the process of clinical auxiliary diagnosis of lung cancer and helps clinicians to make decisions, information can be shared to a plurality of centers through a network platform, diagnosis suggestions canbe provided for clinicians lacking experience, and the overall clinical lung cancer diagnosis level is improved.
Owner:PEKING UNIV FIRST HOSPITAL +1
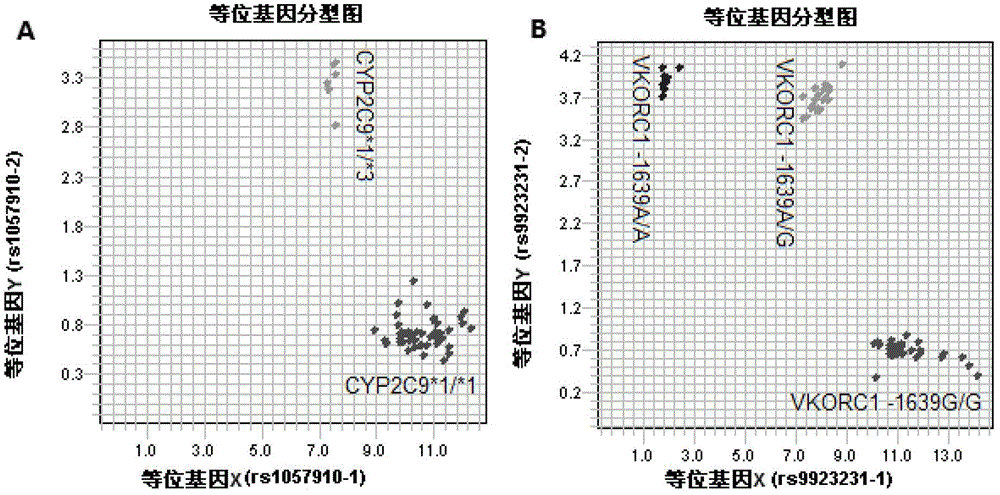
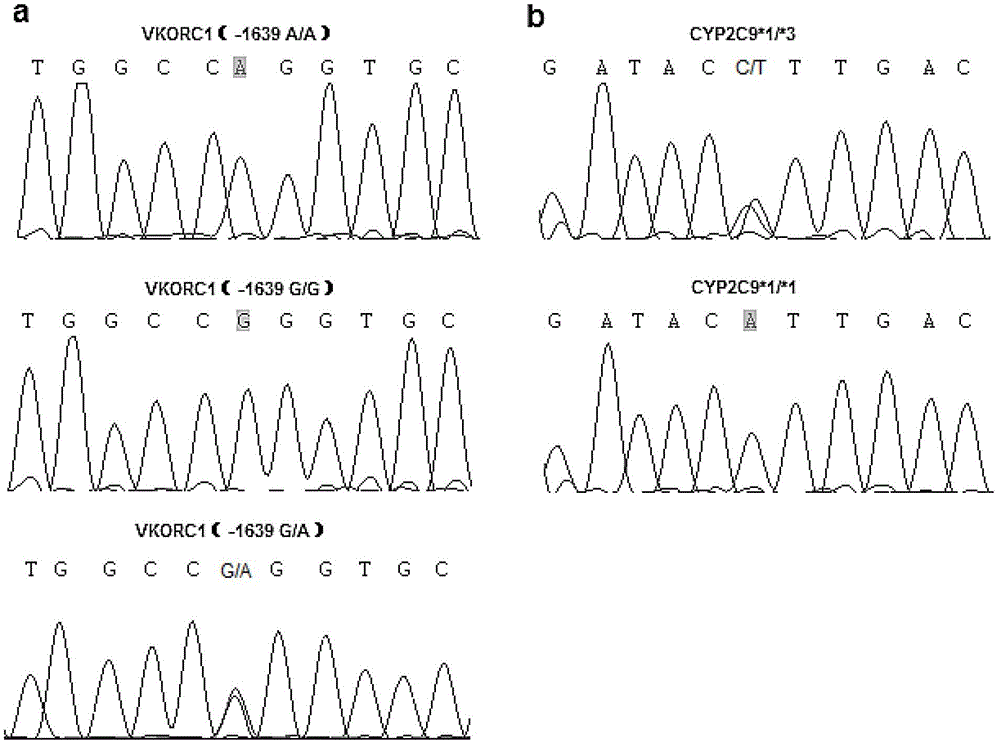
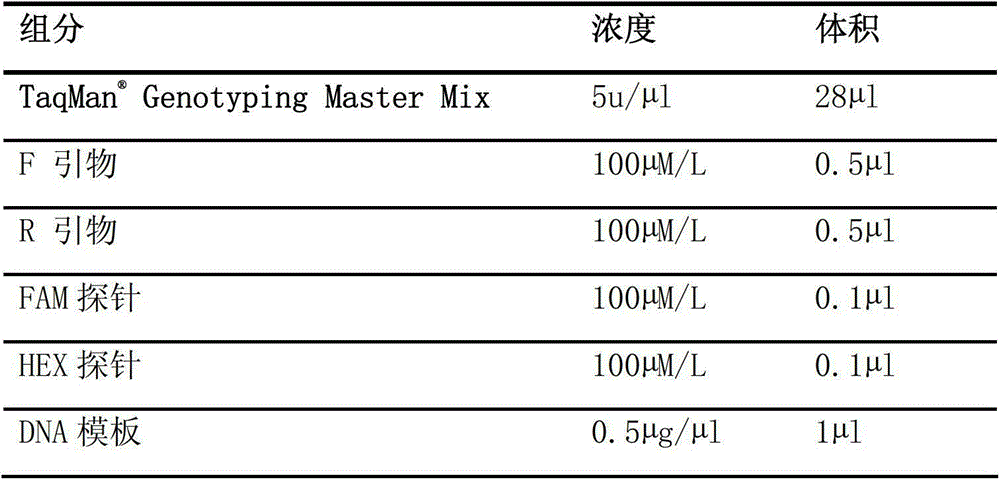
Kit for rapidly detecting warfarin individualized medication related gene SNP sites, and its detection method
The invention relates to a kit for rapidly detecting warfarin individualized medication related gene SNP sites, and its detection method, and belongs to a genetic detection technology in the clinic detection technique of the biomedical field. The genetic typing of two polymorphic sites comprising CYP2C9*3(1061A / C) and VKORC1(-1639G / A) is rapidly and accurately carried out through human peripheral blood DNA drawing, specific PCR amplification by a Taq man probe and fluorescence signal analysis. The invention provides primers, probes and kits which are used for detecting the polymorphic sites, a use of the primers and the probes in the preparation of the kits, and a use of the determination of the warfarin application amount of a patient according to the detection results of the polymorphic sites. The determination use can effectively reduce the generation of the warfarin application amount related adverse events and prevent thrombotic diseases. The detection method can be used for the auxiliary diagnosis and treatment of various patients needing warfarin clinically.
Owner:丁虎 +1
Circular RNA marker hsa_circ_0001788 and application thereof
ActiveCN109295218AImprove stabilityEasy to degradeMicrobiological testing/measurementDNA/RNA fragmentationDiagnosis earlyCraniosynostosis
The invention establishes a circRNA expression profile for a patient with craniosynostosis, provides, provides serum circRNA marker hsa_circ_0001788 for the patient with craniosynostosis, discloses diagnostic value of hsa_circ_0001788 of serum for craniosynostosis, applies the marker herein to prepare a diagnostic kit for the patient with craniosynostosis, and provides supporting for clinical early discovery and treatment of the patient with craniosynostosis. Serum circRNA has good stability, degradation difficulty and the other advantage, has high value in auxiliary diagnosis, and is easy toclinically popularize and use. A circRNA chip is used herein to carry out detecting to obtain disease-specific and abnormally-expressed serum circRNA expression profiles; verifying is performed by means of qRT-PCR; a strict design and evaluation system is provided. It is discovered for the first time that the marker hsa_circ_0001788 in serum is an important biological detection index which causesminimal invasion and has low cost and which is applicable to clinical detection; the marker hsa_circ_0001788 is important to the clinical early diagnosis and differential diagnosis of craniosynostosis; a new idea is provided for the diagnosis of craniosynostosis in molecular level, and it is expected that early diagnostic rate of craniosynostosis is increased.
Owner:NANJING MEDICAL UNIV
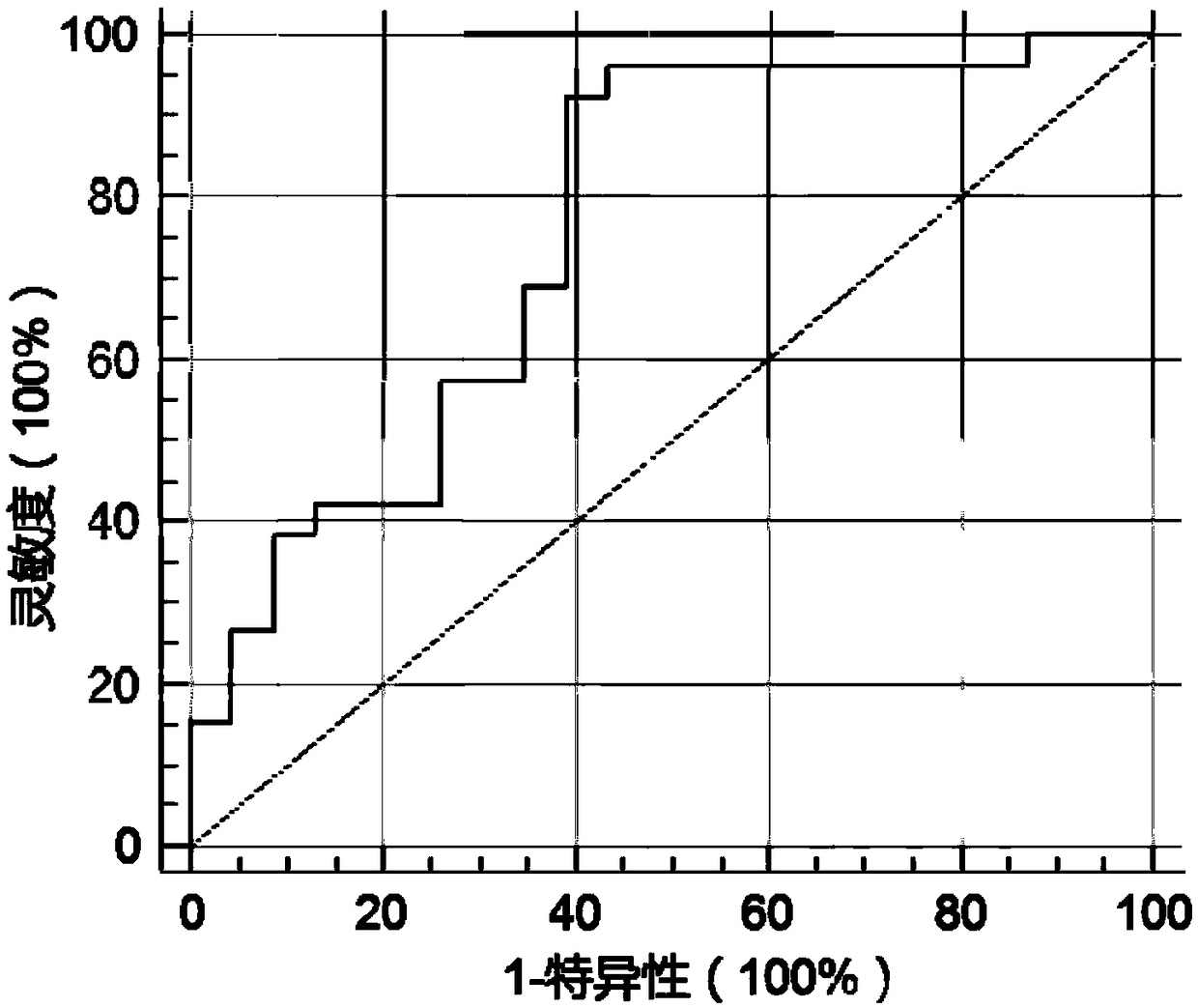
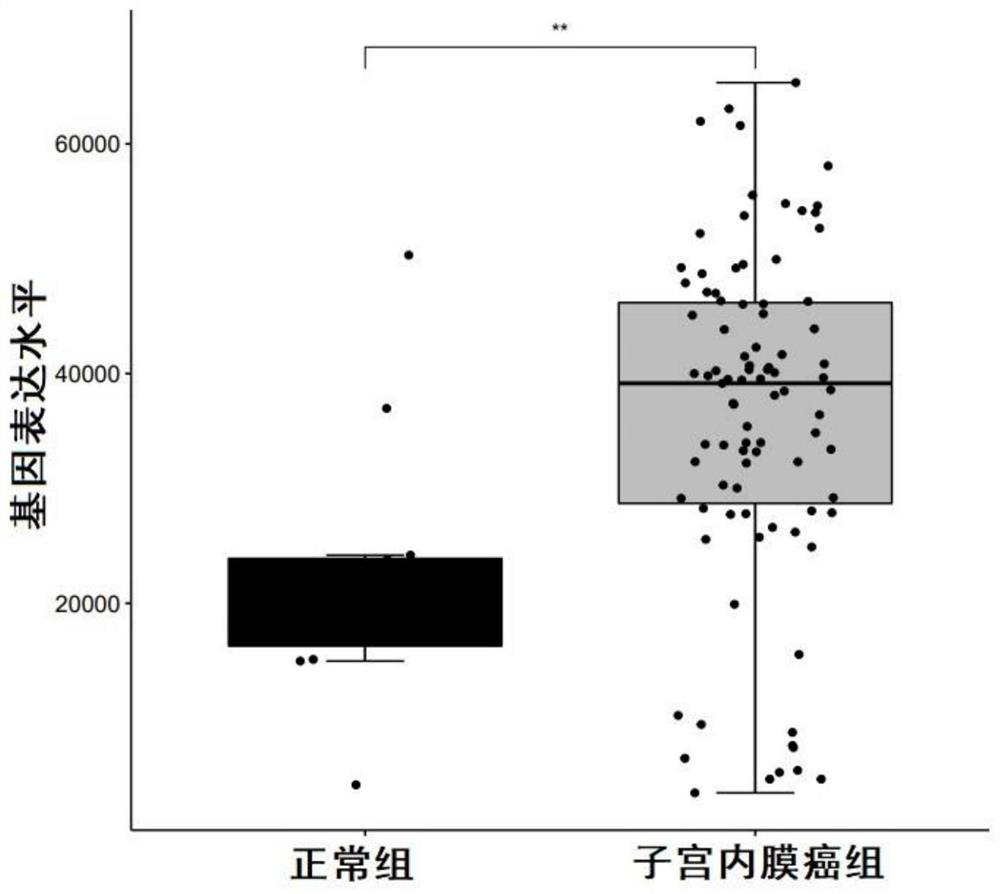
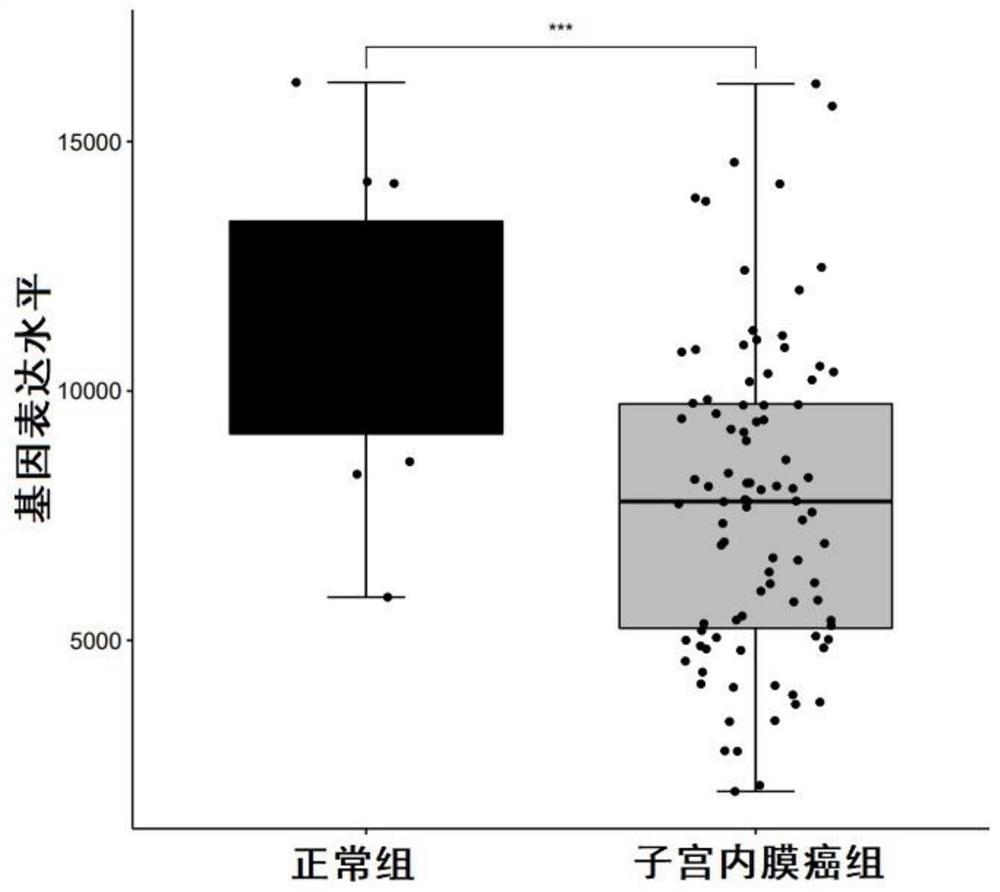
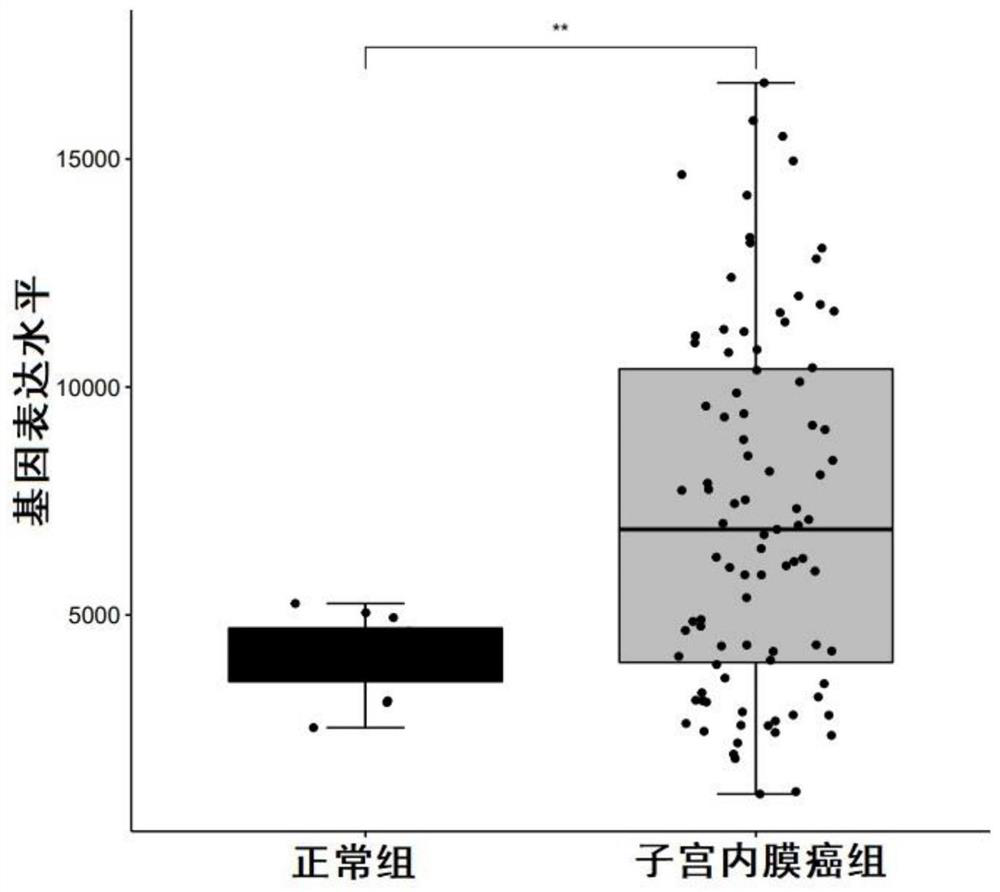
Biomarkers for diagnosing endometrial cancer as well as product and application thereof
PendingCN113249491AEasy diagnosisMicrobiological testing/measurementAntineoplastic agentsEndometrial CarcinomasAids diagnostics
The invention discloses biomarkers for diagnosing endometrial cancer as well as a product and application thereof, and provides application of reagents for detecting biomarkers TMSB10, EIF3E and PDIA6 in a sample to be detected in preparation of the product for diagnosing and / or assisting in diagnosing endometrial cancer. Verification results show that the combination of TMSB10, EIF3E and PDIA6 has relatively good diagnosis efficiency, whether a subject suffers from the endometrial cancer or the risk of suffering from the endometrial cancer or not can be diagnosed in an early stage according to the combination of TMSB10, EIF3E and PDIA6, and good clinical application value is achieved.
Owner:SICHUAN ACADEMY OF MEDICAL SCI SICHUAN PROVINCIAL PEOPLES HOSPITAL
Tumor markers associated with pancreatic cancer and application thereof
ActiveCN103667444AMicrobiological testing/measurementBiological material analysisTissues tumorGenetic engineering
The invention belongs to the field of genetic engineering and medicine, relates to a novel tumor marker for early diagnosis of pancreatic cancer, and more specifically discloses a new serum / tissue tumor marker associated with pancreatic cancer and application thereof. The marker is a combination of CystatinSN (CST1), CystatinS (CST4) and CystatinSA (CST2). The marker, primers and antibodies can be used for a diagnostic kit for the auxiliary diagnosis of pancreatic cancer.
Owner:THE THIRD AFFILIATED HOSPITAL OF SUN YAT SEN UNIV
Gastric cancer RNA molecular marker and application thereof
ActiveCN106148348AEasy to operateEasy to get materialsMicrobiological testing/measurementDNA/RNA fragmentationCancers diagnosisBlood plasma
The invention belongs to the field of biological medicine and specifically relates to various RNAs, various combinations of tumor markers thereof and an application thereof in preparing a gastric cancer diagnosis reagent. The invention provides a set of new RNA molecular markers for gastric cancer diagnosis. The content of the set of RNA in gastric cancer tissue is higher than the content of para-carcinoma tissue and the content of the set of RNA in the plasma of gastric cancer patient is higher than the content thereof in the plasma of healthy people. The invention also provides a method for taking one or different combinations of the set of RNA as the tumor markers and the application thereof. The set of RNA molecular marker and the diagnostic method of different combinations of the set of RNA markers adopted for diagnosing gastric cancer are simple in operation, are convenient in material taking and have the characteristics of high specificity, high sensitivity and easiness in mass screening. The set of RNA molecular marker is fit for screening of gastric cancer high-risk groups and auxiliary diagnosis of gastric cancer.
Owner:INST OF RADIATION MEDICINE ACAD OF MILITARY MEDICAL SCI OF THE PLA
Marker used for assisted detection of abrupt highland advancing low pressure low oxygen environment related myocardial damages
The invention discloses a marker used for assisted detection of abrupt highland advancing low pressure low oxygen environment related myocardial damages, and applications of a product used for detection of miR-144-3p expression level in preparation of kits used for auxiliary diagnosis of myocardial damages caused by low pressure low oxygen. According to the applications, a low-pressure oxygen cabin is adopted for stimulation of highland low pressure low oxygen environment, and establishment of rat animal models with low pressure low oxygen myocardial damages. High flux sequencing technology and bioinformatics methods are adopted, key miRNA related to highland low pressure low oxygen myocardial damages are screened, and fluorogenic quantitative PCR is adopted for experiment verifying of thekey miRNAs in cardiac muscle tissues and blood plasma. The miRNAs disclosed in the invention can be used for assisted detection of abrupt highland advancing low pressure low oxygen environment related myocardial damages, and possess important meaning in diagnosis and assessment of abrupt highland advancing low pressure low oxygen environment related myocardial damages.
Owner:中国人民解放军总医院第七医学中心
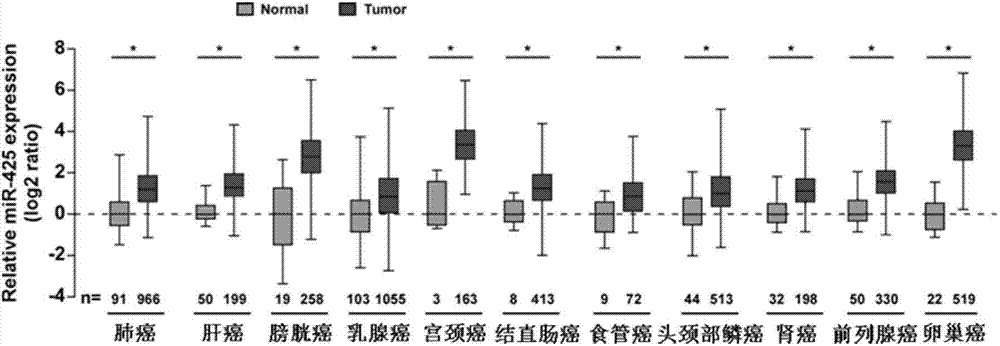
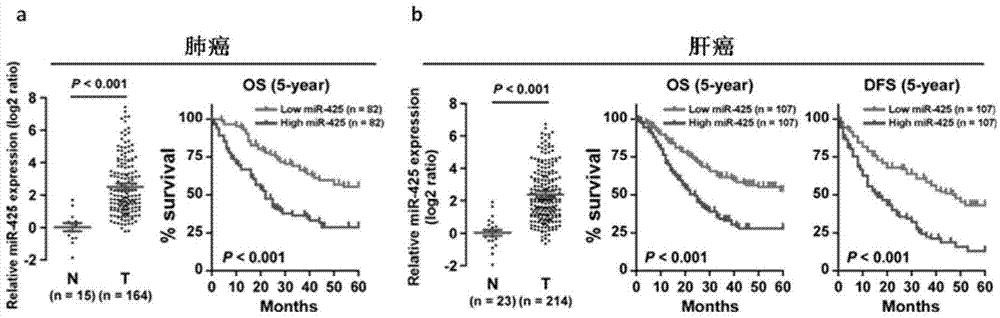
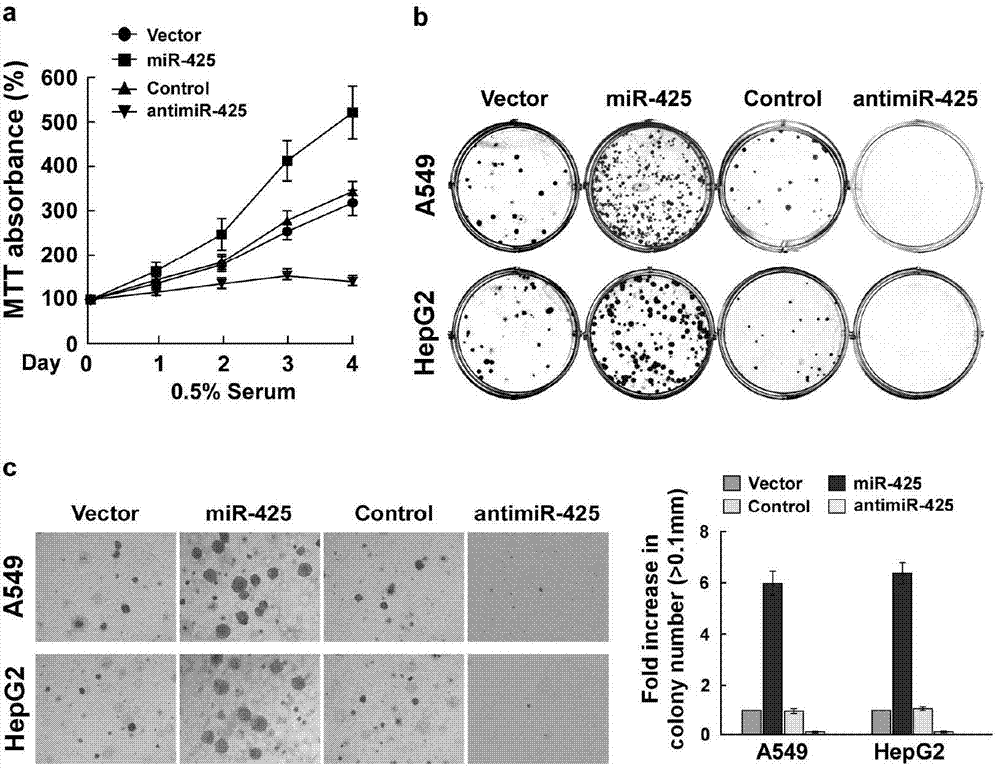
Application of miR-425 in tumor diagnosis, treatment and prognosis
ActiveCN104726584APrevent proliferationInhibit transferGenetic material ingredientsMicrobiological testing/measurementTumor therapyApoptosis
The invention discloses application of miR-425 in tumor diagnosis, treatment and prognosis. Multiple biological characteristics of tumor including apoptosis, proliferation, drug susceptibility and the like disclosed by the invention are closely related to the expression quantity of miR-425. MiR-425 antagonist antimiR-425 can be used for significantly inhibiting proliferation of multiple types of tumor cells and the tumorigenic ability of the tumor cells in a nude mouse, and enhancing the tumor treatment capacity of chemotherapy drug cisplatin and the like in the nude mouse. The invention discloses a kit for tumor assisted diagnosis and patient survival prognosis, and the kit contains a primer sequence for the quantitative determination of miR-425; the invention also discloses a pharmaceutical composition for treating tumor, wherein the composition contains the miR-425 antagonist antimiR-425. The invention provides a novel method for assisted diagnosis and prognosis diagnosis of cancer. The invention discovers that the antimiR-425 has high clinical application value in the preparation of a tumor-treating medicine, and particularly provides a novel medicine and a treatment method for the effective treatment of lung cancer and liver cancer.
Owner:SUN YAT SEN UNIV CANCER CENT
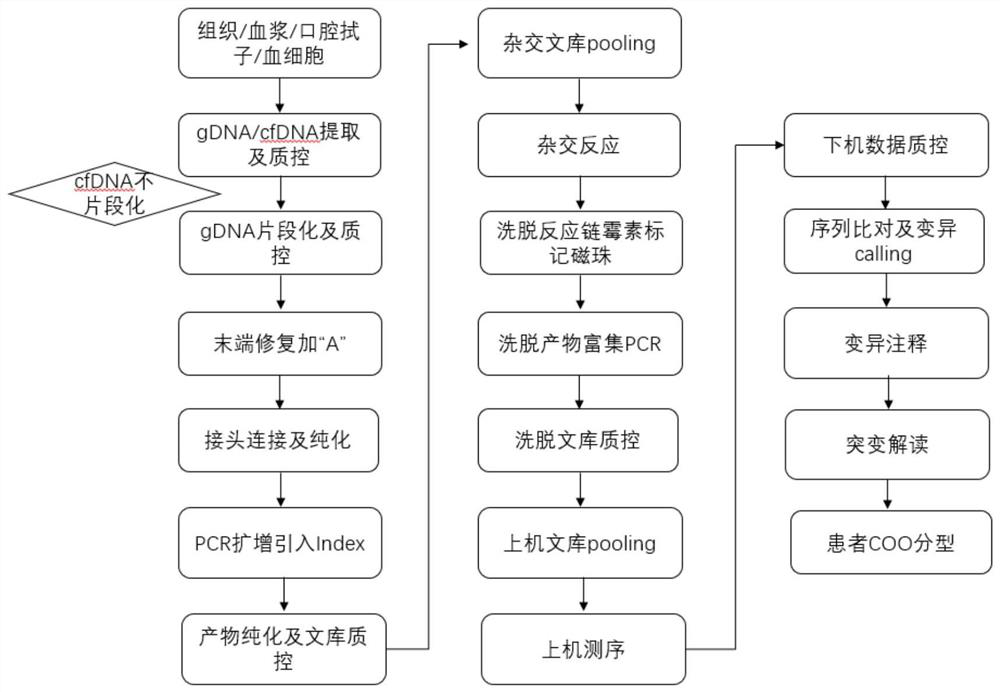
Reagent kit for detecting lymphoma genovariation and application of reagent kit
ActiveCN111662983AEasy to coverHigh sensitivityMicrobiological testing/measurementProteomicsAids diagnosticsBCL6
The invention discloses a reagent kit for detecting lymphoma genovariation and an application of the reagent kit, and particularly discloses the reagent kit for detection or auxiliary detection of variation of a lymphoma related gene. The reagent kit comprises a substance for detecting BCL2,BCL6, MYC and / or gene IGH fusion, the substance is a complete set of DNA probes, and the complete set of theDNA probes comprises 376 probes as shown in SEQID NO:1 to SEQ ID NO:376. The reagent kit can be used for detection or auxiliary detection of the variation of the lymphoma related gene, substyping ofcells of origin (cell of origin, COO) of patients suffering from diffuse large B-cell lymphoma, and auxiliary diagnosis, prognostication judging and / or targeted drug prediction of the patients suffering from lymphoma.
Owner:BEIJING GENEPLUS TECH +1
Diagnostic kits and methods for oesophageal abnormalities
InactiveCN101291630AQuick collectionReduce monitoringSurgical needlesVaccination/ovulation diagnosticsAids diagnosticsNon squamous
The invention relates to kits and methods for aiding the diagnosis of Barrett's oesophagus or Barrett's associated dysplasia. Preferred is a method comprising assaying cells from the surface of a subject's oesophagus for a non-squamous cellular marker, wherein detection of such a marker indicates increased likelihood of the presence of Barrett's or Barrett's associated dysplasia, preferably wherein said sample of cells is not directed to a particular site within the oesophagus. The invention also encompasses a method comprising sampling the cellular surface of the oesophagus of said subject. The invention also relates to a kit comprising a swallowable device comprising abrasive material capable of collecting cells from the surface of the oesophagus, together with printed instructions for its use in detection of Barrett's oesophagus or Barrett's associated dysplasia. Preferably said device comprises a capsule sponge.
Owner:MEDICAL RESEARCH COUNCIL +1
Detection marker of steroid-induced osteonecrosis of femoral head necrosis and application thereof
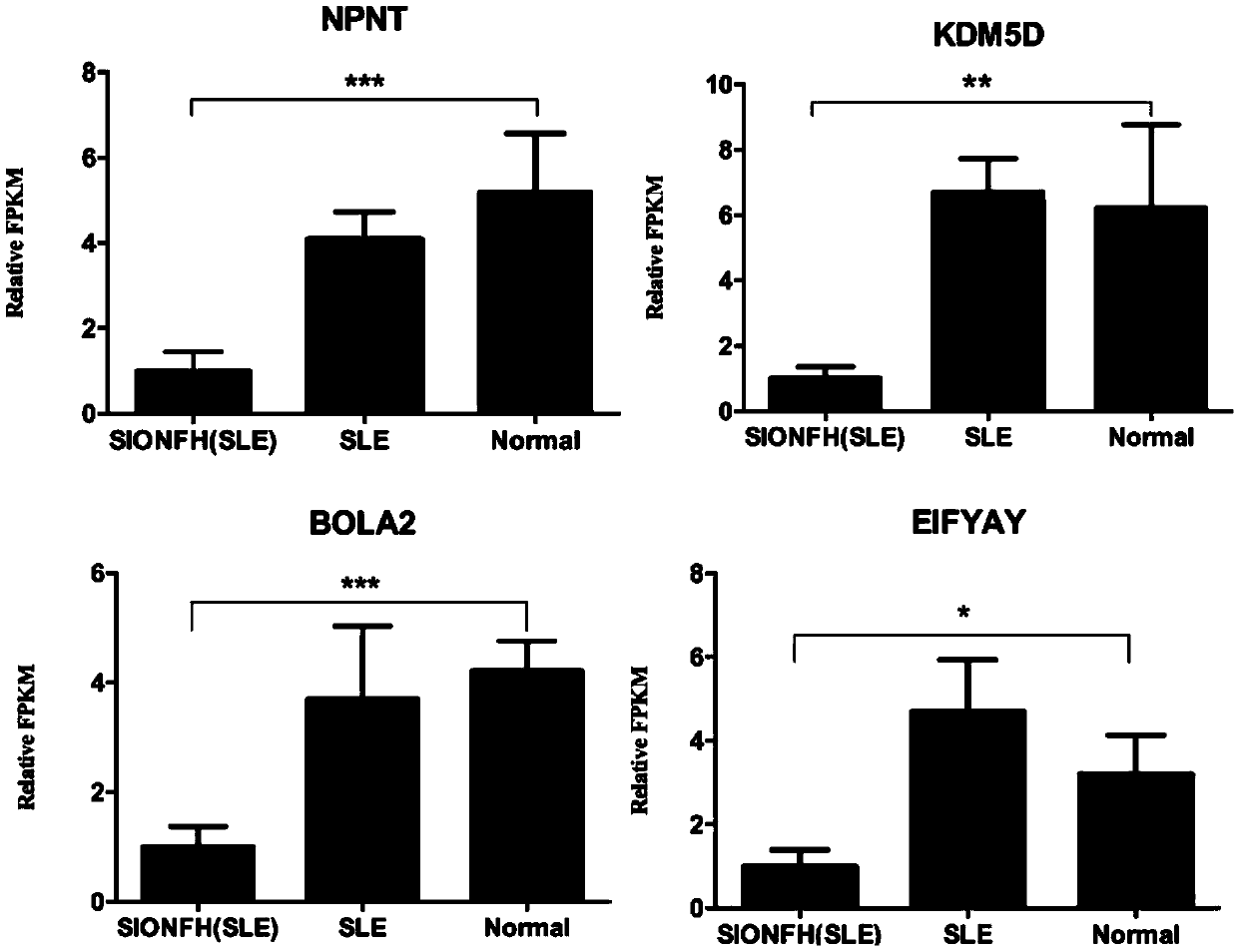
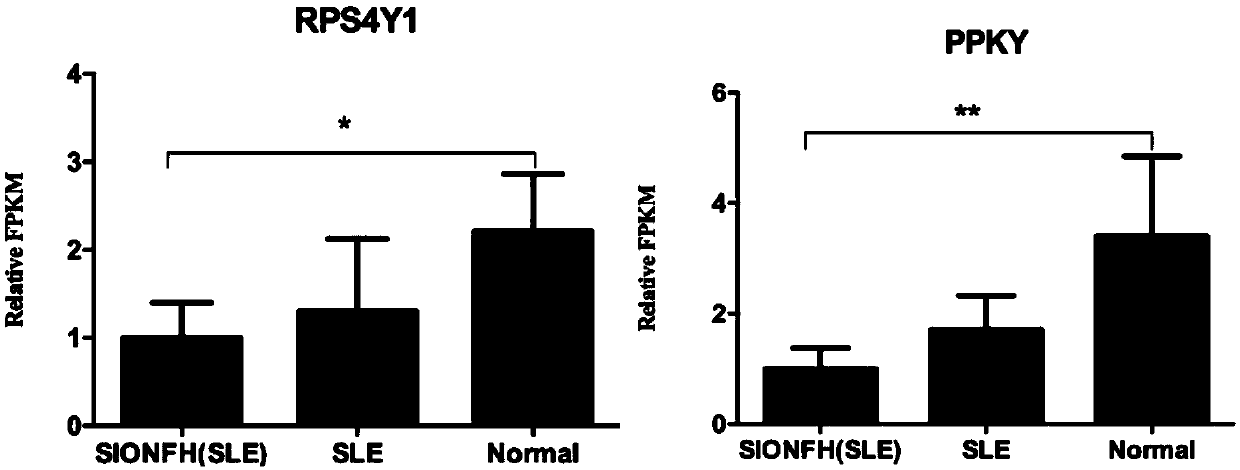
InactiveCN109554458ADownregulation of RNA expression levelsDownregulation of expression levelMicrobiological testing/measurementDNA/RNA fragmentationRna expressionSteroid induced osteonecrosis
The present invention discloses a detection marker of steroid-induced osteonecrosis of femoral head necrosis and an application thereof. RNA expression levels of 6 genes of NPNT, KDM5D, BOLA2, EIFYAY,RPS4Y1 and PPKY are down-regulated in SIONFH patients for the first time. The detection marker lays a good foundation on preparing a kit for diagnosis or auxiliary diagnosis of SIONFH.
Owner:陈雷雷
Method for rapidly detecting human leucocyte antigen B27 (HLA-B27) and kit thereof
ActiveCN102443625AStrong specificityIncreased sensitivityMicrobiological testing/measurementAntigenFluorescence
The invention relates to a method for rapidly detecting a human leucocyte antigen B27 (HLA-B27) and an in-vitro diagnosis kit, which belong to the technical field of medical biology. In the kit, a pair of HLA-B27 specific primers and a specific fluorescent probe are adopted for rapidly detecting the HLA-B27. In the kit, the pair of HLA-B27 specific primer and the specific fluorescent probe can beused for rapidly and accurately detecting HLA-B27 genes in samples such as human peripheral blood and the like, and the kit has the advantages of high specificity, high sensitivity, time saving, labor saving, low cost, high flux and the like, and can be applied to auxiliary diagnosis of multiple spondyloarthropathies which severely endanger human health such as clinical ankylosing spondylitis, Reiter syndrome, psoriatic arthritis, ulcerative colitis accompanying arthropathy, acute anterior tract uveal, and the like.
Owner:浙江夸克生物科技有限公司
Detection kit for detecting biomarker of bladder cancer
InactiveCN111154879ARealize Auxiliary DiagnosisRealize functionMicrobiological testing/measurementDNA/RNA fragmentationReference genesAids diagnostics
The invention provides a detection kit for detecting a biomarker of the bladder cancer. The detection kit includes a primer and probe for detecting the biomarker of the bladder cancer, the biomarker of the bladder cancer is at least one selected from genes of CA9, IGF2, CDK1, UBE2C, CRH, RPS21 and HOXA13. Furthermore, the detection kit further includes a primer and probe for detecting a referencegene, the reference gene is selected from SLC25A6, ACTB or GAPDH. The invention also provides a bladder cancer-diagnosing diagnostic kit including the detection kit. The diagnostic kit is capable of diagnosing whether an individual has the bladder cancer or not and the possibility of recurrence accurately, and has high sensitivity, and the functions of auxiliary diagnosis and recurrence monitor ofnon-invasive bladder cancer are achieved.
Owner:北京恩泽康泰生物科技有限公司
Sample dilution composition for detecting sugar antigen 72-4, detection reagent and kit containing detection reagent
InactiveCN110824175AShorten the timeEliminate distractionsBiological material analysisBiological testingDiseaseEnzyme binding
The invention relates to the field of medical examination, and particularly relates to a sample dilution composition for detecting a sugar antigen 72-4, a detection reagent and a kit containing the detection reagent. The kit mainly comprises a magnetic particle suspension coated with a CA72-4 antibody, an enzyme conjugate, a sample diluent and a calibrator. The sample dilution composition can be used for auxiliary diagnosis and postoperative monitoring of tumor diseases such as gastric cancer, ovarian cancer, colorectal cancer, pancreatic cancer and cervical cancer. The kit is short in detection time, and the time of an inspector is greatly saved; the kit adopts a double-antibody sandwich method, is wide in linear range and high in clinical practicability; the kit adopts the specific sample diluent, so that the difference between new and old samples can be eliminated, false positive interference caused by HAMA is effectively reduced, and the detection accuracy of CA72-4 is greatly improved; and the kit adopts a two-step method, so that the risk of HOOK in clinical practice is reduced.
Owner:AUTOBIO DIAGNOSTICS CO LTD
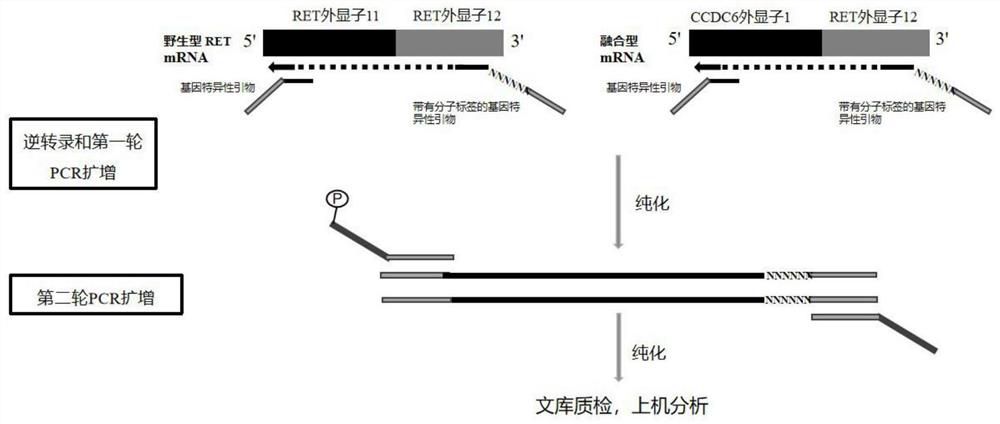
Human thyroid cancer gene fusion kit and detection method
InactiveCN112011615AImprove detection accuracyReduced open tube/pipetting riskMicrobiological testing/measurementFresh TissueOncology
The invention discloses a human thyroid cancer gene fusion kit and a detection method in the technical field of molecular biology. According to the kit, a multiple PCR capture technology and an NGS sequencing technology are adopted; the kit is used for qualitatively detecting various variants of CCC6-RET, NCOA4-RET, PAX8 / PPARG and ETV6-NTRK3 fusion genes in a thyroid nodule needle biopsy cytological uncertainty fresh tissue sample, and detection threshold values are determined according to molecular tags. Assistance can be provided for auxiliary diagnosis and treatment of thyroid cancer through detection of fusion sites of the kit in combination with clinical pathological analysis results.
Owner:上海睿璟生物科技有限公司
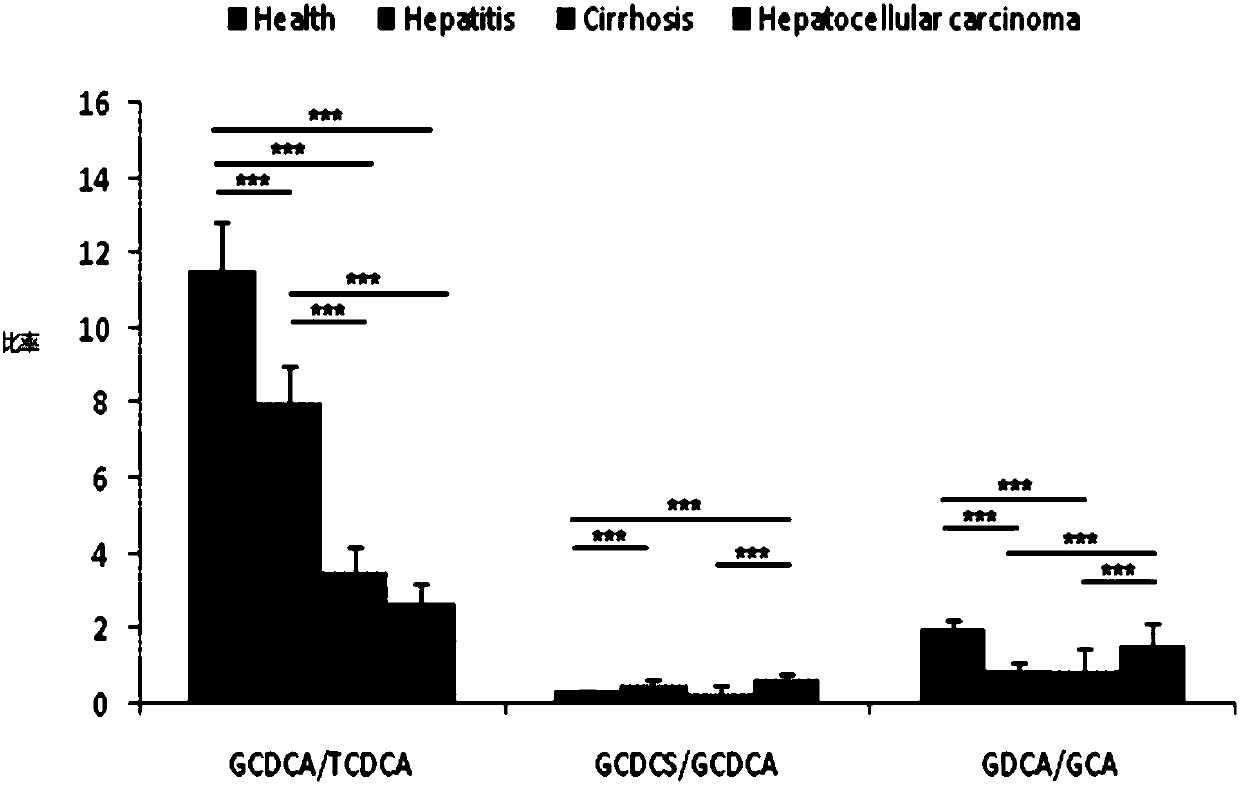
Use of combined serum metabolic marker in preparation of kit for diagnosing progress of hepatopathy, kit, and method using kit to screen serum metabolic markers
InactiveCN107656007AAlleviate the problem of difficult development monitoringRapid diagnosisComponent separationSerum igeMetabolite
The invention provides a use of a combined serum metabolic marker in the preparation of a kit for diagnosing the progress of hepatopathy, the kit, and a method using the kit to screen serum metabolicmarkers, and relates to the technical field of serum metabolic markers. The combined serum metabolic marker is mainly composed of sodium glycochenodeoxycholate sulfate, glycochenodeoxycholic acid, glycodesoxycholic acid, glycocholic acid and taurochenodeoxycholic acid. The kit executes combined screening detection on the content of above serum metabolites in serum in order to realize assisted diagnosis of the progress of the hepatopathy of hepatopathy patients, so the difficult monitoring problem of the occurrence and development of the hepatopathy in existing clinic therapy of the hepatopathyis effectively alleviated.
Owner:HANGZHOU HEALTH BANK MEDICAL LAB CO LTD
Auxiliary diagnose method, reagent kit and system of bladder cancer through union of drive gene point mutation and methylation, and application
ActiveCN110628910AReduce demandEasy to operateMicrobiological testing/measurementAgainst vector-borne diseasesBladder cancerAids diagnostics
The invention provides a method, reagent kit and system for screening, or detection or auxiliary diagnose of bladder cancer, and an application. FGFR3, TERT and PLEKHS1 point mutation and OTX1, NID2 and NRN1 methylation level can be detected at the same time, united detection of gene point mutation and methylation detection can be skillfully realized, the requirement quantity of samples, the experiment operation steps and the detection period can be reduced, and the detection specificity and the detection sensitivity are improved.
Owner:HUNAN YEARTH BIOTECHNOLOGICAL CO LTD
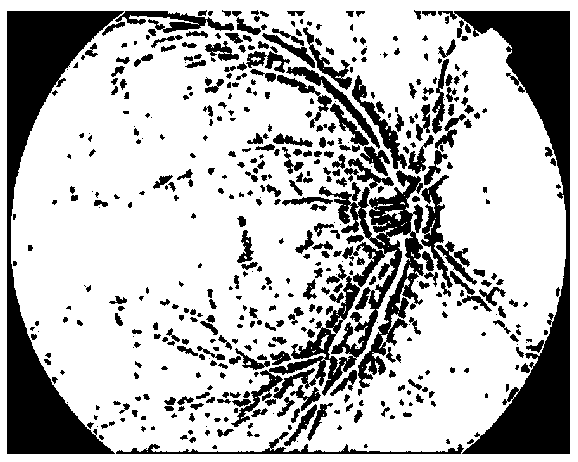
Blood vessel, artery and vein identification method for fundus photography
The invention discloses a blood vessel, artery and vein identification method for fundus photography. Each position in the fundus photography is automatically positioned and measured to achieve an effect on pre-screening diseases, pictures with lesion suspicions can be automatically screened so as to accurately and effectively carry out mark identification on blood vessel contours and blood vesselarteries and veins, then, the diameter ratio of arteries and veins can be calculated to assist in diagnosing diseases, including retina blood vessels and the like, and the workloads of doctors are reduced. In addition, the result of the method does not depend on the experience of doctors and is more objective, doctors can be effectively assisted in diagnosing diseases, and a purpose of remote consultation is realized.
Owner:SUN YAT SEN UNIV +1
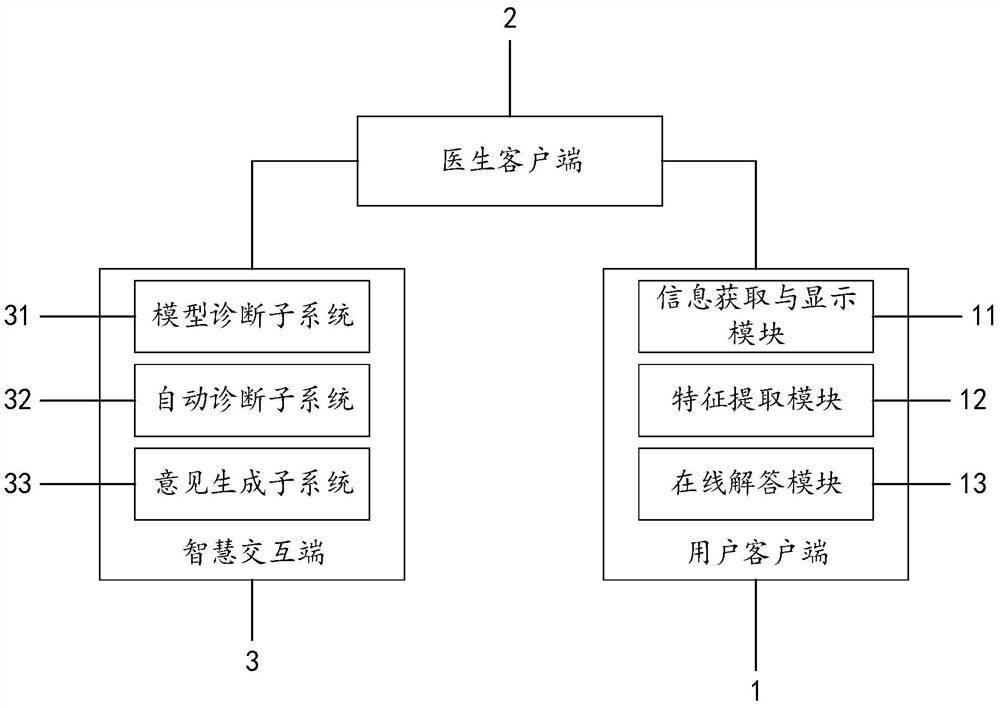
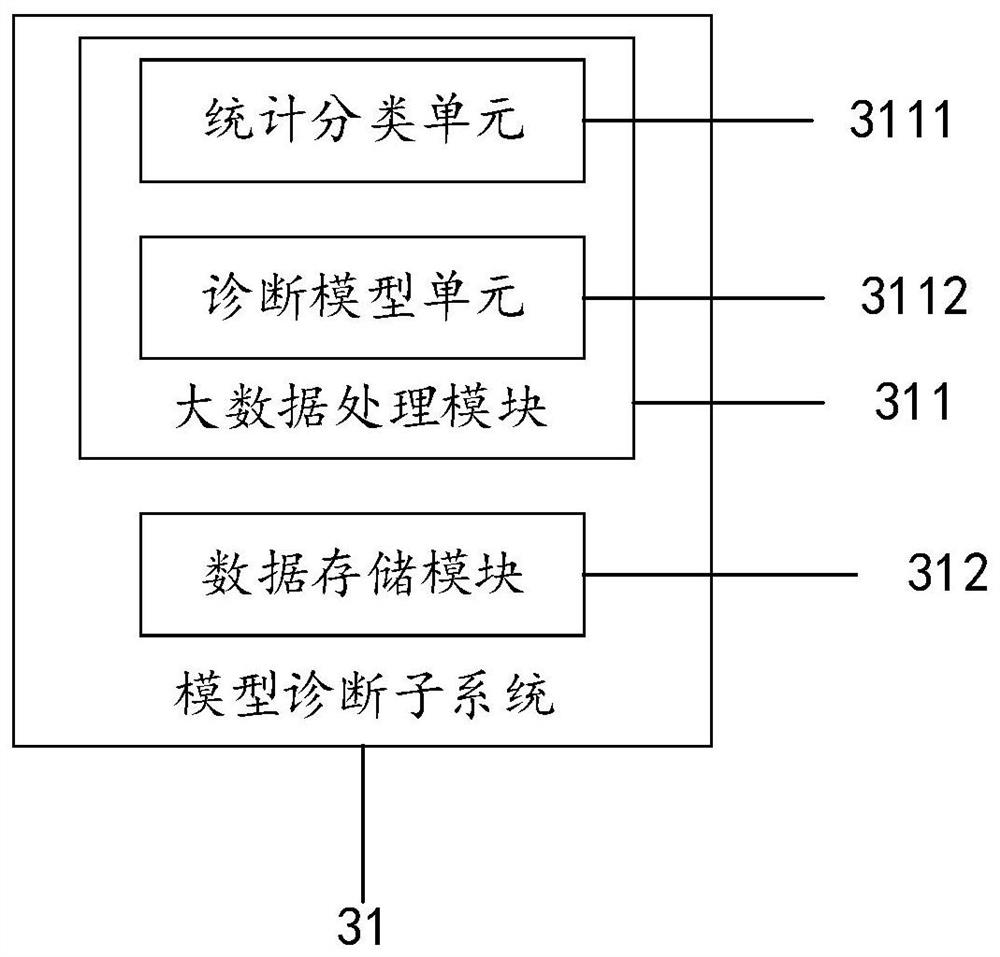
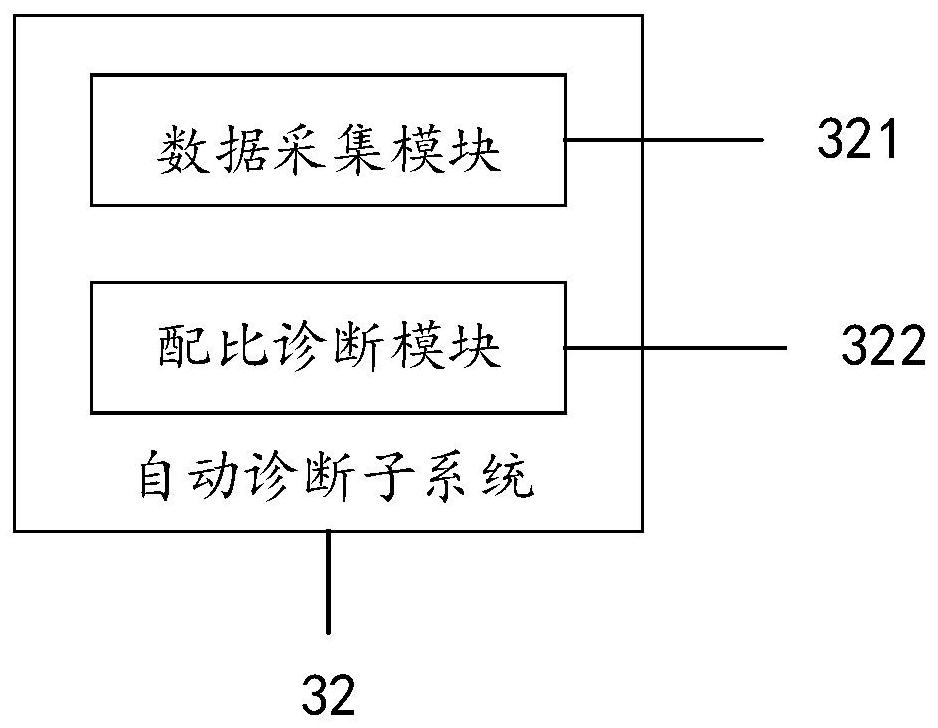
Large health medical disease auxiliary diagnosis system and method based on intelligent terminal
The invention discloses a large health medical disease auxiliary diagnosis system and method based on an intelligent terminal. The system comprises a user client, a doctor client, and an intelligent interaction terminal. The method comprises the steps: firstly carrying out the statistical division of large health medical data stored in the intelligent interaction terminal; constructing a corresponding definite diagnosis symptom set; acquiring user information through the user client, performing feature extraction, performing similarity calculation on the extracted feature information and the definite diagnosis symptom set, and returning diagnosis information; based on the diagnosis information, in combination with a doctor client, generating corresponding diagnosis suggestions and transmitting back to the user client to be displayed to complete auxiliary diagnosis. If online diagnosis is needed, the doctor client can conduct online diagnosis according to the diagnosis suggestions and asent consultation request, and the accuracy of a diagnosis result is improved.
Owner:深圳市唐仁医疗科技有限公司
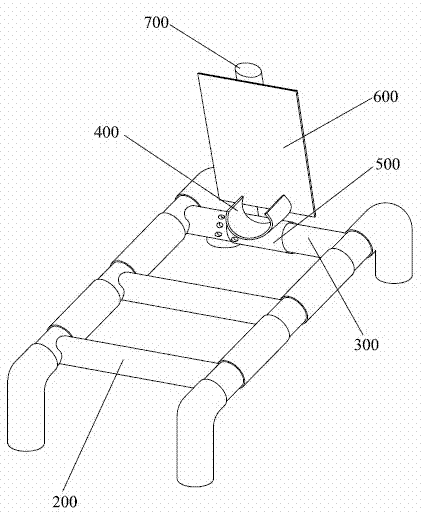
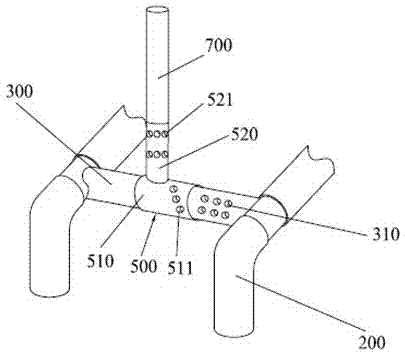
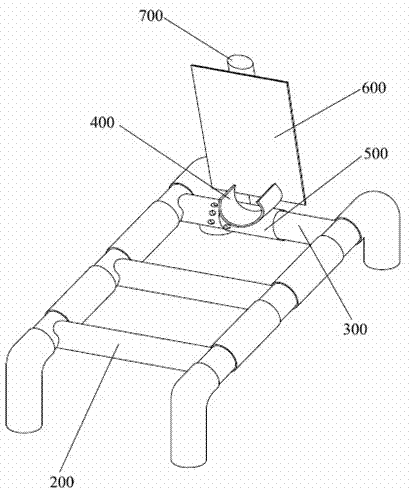
Stent for assisting CT diagnosis of tarsometatarsal joint injury
InactiveCN102512197AGuaranteed bending orientation requirementsGuaranteed accuracyPatient positioning for diagnosticsComputerised tomographsAids diagnosticsFirst tarsometatarsal joint
The invention discloses a stent for assisting computed tomography (CT) diagnosis of tarsometatarsal joint injury. The stent comprises a stent main body, a foot fixer, a pedal, an inner tube and an adjusting piece, wherein the inner tube is fixedly arranged on the stent main body; the adjusting piece is arranged on the inner tube in a rotatable and sliding mode; and both the foot fixer and the pedal are fixedly connected with the adjusting piece. The stent for assisting the CT diagnosis of the tarsometatarsal joint injury can assist a patient in maintaining a foot at a plantar flexion ectropion position in the CT diagnosis process, so that diagnosis is facilitated, the requirement of the CT diagnosis for the flexion direction of the foot of the patient is met, the CT diagnosis operation can be accurately performed, and the accuracy and the precision of the diagnosis result are ensured.
Owner:WEST CHINA HOSPITAL SICHUAN UNIV
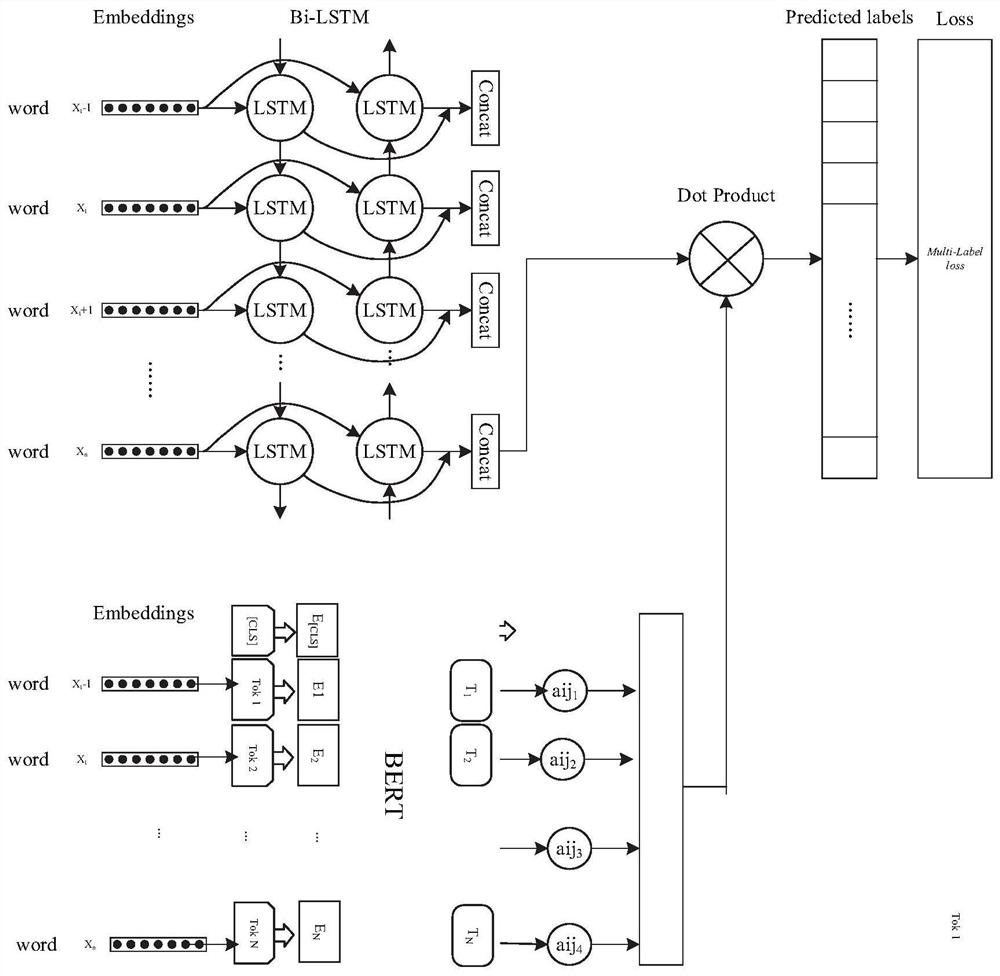
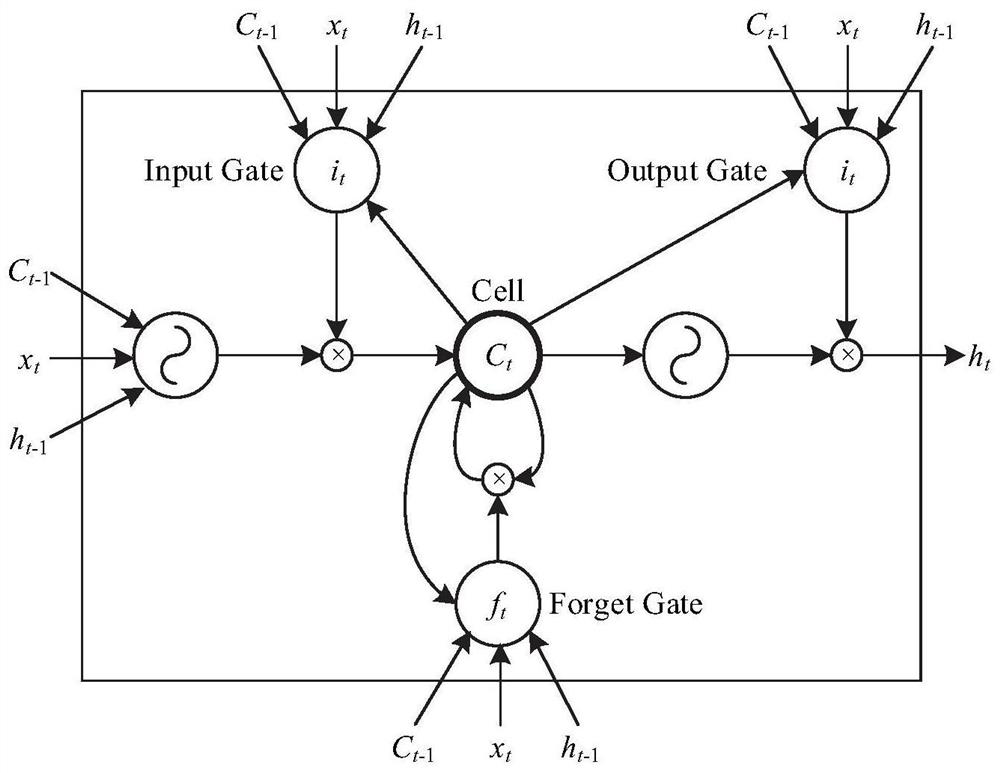
Traditional Chinese medicine auxiliary diagnosis system
PendingCN112002409AAvoid word segmentation problemsSolve underutilized problemsSemantic analysisMedical automated diagnosisDiseaseAids diagnostics
The invention discloses a traditional Chinese medicine auxiliary diagnosis system. The system comprises a data acquisition module used for acquiring a traditional Chinese medicine diagnosis text and acorresponding disease and constructing a training set; a model construction module used for respectively training the bidirectional LSTM network and the BERT network according to the training set, and constructing a traditional Chinese medicine auxiliary diagnosis model according to the trained bidirectional LSTM network and BERT network; and a disease prediction module used for predicting the to-be-detected traditional Chinese medicine diagnosis text according to the traditional Chinese medicine auxiliary diagnosis model and outputting a predicted disease. A traditional Chinese medicine auxiliary diagnosis model is constructed in combination with a bidirectional LSTM network and a BERT network, the problems that traditional Chinese medicine professional vocabulary expression is limited in traditional Chinese medicine diagnosis data and contextual information in traditional Chinese medicine texts cannot be fully utilized are solved, and corresponding diseases are predicted and diagnosed according to traditional Chinese medicine clinical symptoms.
Owner:SHANDONG NORMAL UNIV
Diagnostic agent for diagnosis or auxiliary diagnosis of renal insufficiency or renal injury, kit and application
ActiveCN110904214AMicrobiological testing/measurementDisease diagnosisDiagnostic agentAids diagnostics
The invention discloses a diagnostic agent for diagnosis or auxiliary diagnosis of renal insufficiency or renal injury, a kit and application, and relates to the technical field of renal insufficiencyor renal injury diagnosis. The diagnostic agent disclosed by the invention comprises a detection reagent for detecting the expression level of RPS7 gene and / or the SRP14 gene. Researches find that the RPS7 gene and the SRP14 gene have the characteristic of high expression in a renal insufficiency or renal injury group, can be used as markers for renal insufficiency or renal injury diagnosis or auxiliary diagnosis, and provide a new thought and method for renal insufficiency or renal injury diagnosis or auxiliary diagnosis.
Owner:SICHUAN PROVINCIAL PEOPLES HOSPITAL
Specific primer combination for identifying RET gene mutation of medullary thyroid carcinoma as well as kit and application of specific primer combination
ActiveCN109439752AAccurate coverageStrong specificityMicrobiological testing/measurementDNA/RNA fragmentationFluorescenceMicrobiology
The invention discloses a specific primer combination for identifying RET gene mutation of medullary thyroid carcinoma. The specific primer combination is characterized in that the combination is usedfor amplifying a sequence shown in SEQ ID NO.1 in RET gene mutation of medullary thyroid carcinoma. The primer combination comprises a combination of a primer RET8, a primer RET10, a primer RET11, aprimer RET13, a primer RET14, a primer RET15 and a primer RET16. The invention further discloses a kit of the primer combination and an application of the primer combination. The primer combination ishigh in specificity, covers targeted area fragments accurately and detects various known mutant sites. By adopting a mixed solution of the specific primers and marks with fluorescent edges to carry out PCR amplification on batched samples and sanger sequencing analysis, a lot of samples can be analyzed at one time, and various known and unknown mutation can be detected, so that the operating steps are reduced, the resources are saved and the efficiency is improved. A kit provided by the invention can be widely applied to assisted diagnostic detection of clinical medullary thyroid carcinoma and has a wide application prospect.
Owner:SHANGHAI PERSONAL BIOTECH
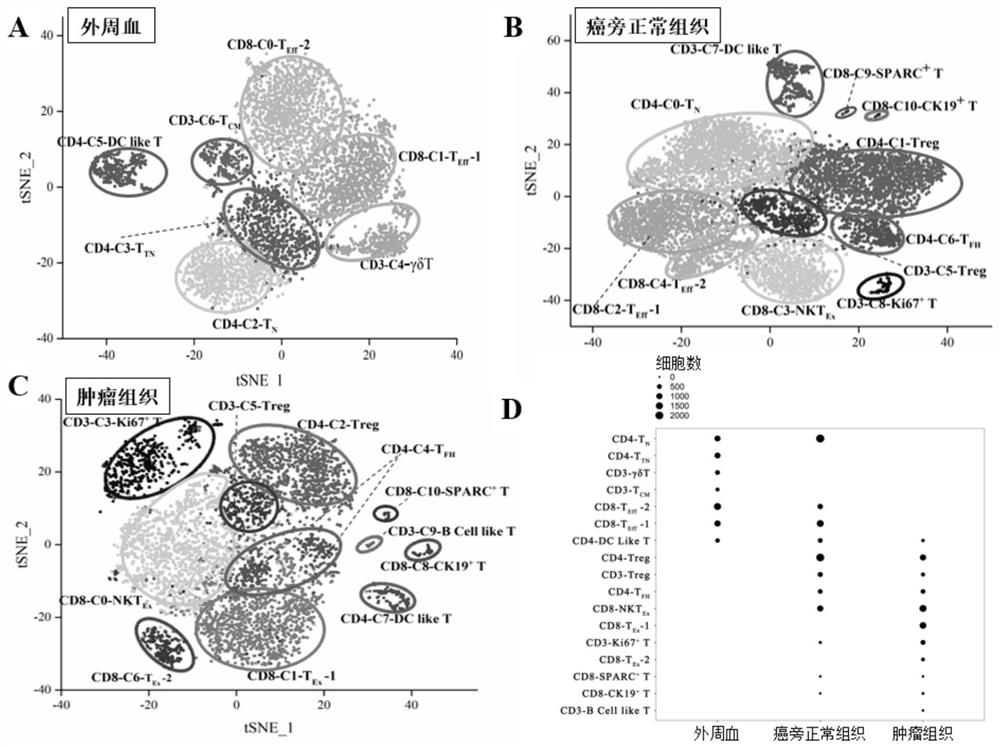
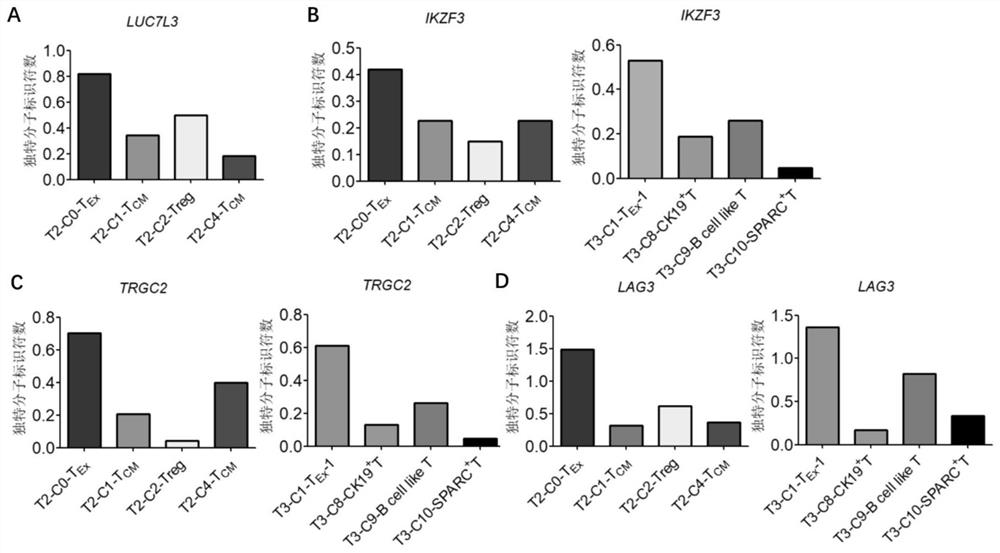
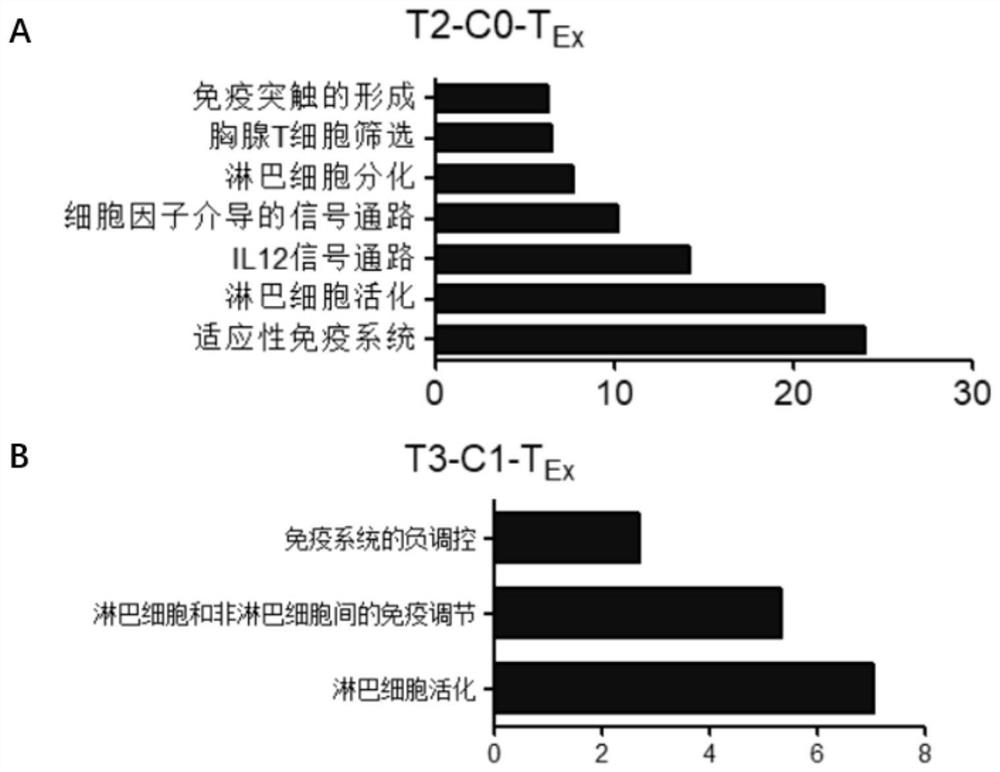
Bladder cancer exhausted T cell subset as well as characteristic genes and application thereof
InactiveCN111748627AMicrobiological testing/measurementBlood/immune system cellsSingle cell transcriptomeCD8
According to the invention, single-cell transcriptome sequencing is carried out on peripheral blood, normal bladder tissues and bladder cancer tissue-derived T cells of an infiltrated bladder cancer patient by utilizing a single-cell transcriptome sequencing analysis technology, a bladder cancer specific T cell subset, namely CD8<+> exhausted T cells, is identified, and characteristic genes, namely genes LUC7L3, IKZF3 and TRGC2, expressed by the CD8<+> exhausted T cells are determined. The characteristic genes can be used for identifying or authenticating CD8<+> exhausted T cells from bladdercancer, can be further used for auxiliary diagnosis, prognosis and immunotherapy of bladder cancer, and provide related detection reagents or kits.
Owner:BEIJING UNIV OF CHEM TECH
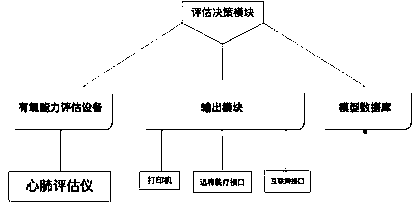
Automatic evaluation system for aerobic capacity and cardiopulmonary clinic symptom
PendingCN109935323ASave manpower and material resourcesQuick output conclusionMedical communicationHealth-index calculationThe InternetData acquisition
The invention discloses an automatic evaluation system for aerobic capability and a cardiopulmonary clinic symptom. The system comprises the following modules of: aerobic capability evaluating equipment, a network connecting module, an evaluating and decision module, a model database, and result output modules (printer, remote medical interface, and Internet interface). A doctor can use the aerobic capability evaluating equipment for evaluating a patient. Evaluation data are uploaded to the remote evaluating and decision module through a wired or wireless relay network. The evaluating and decision module generates auxiliary diagnosis information according to a data acquisition result and the model database and outputs the auxiliary diagnosis information to related personnel.
Owner:南京怡得健康管理有限公司