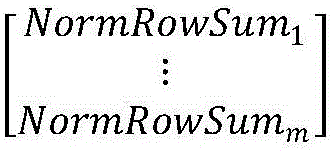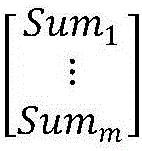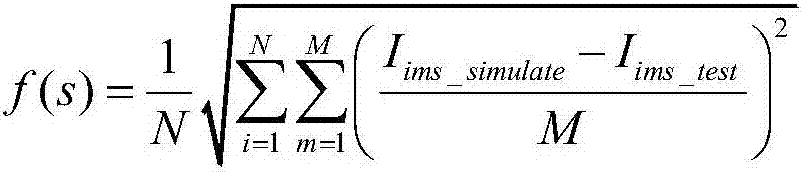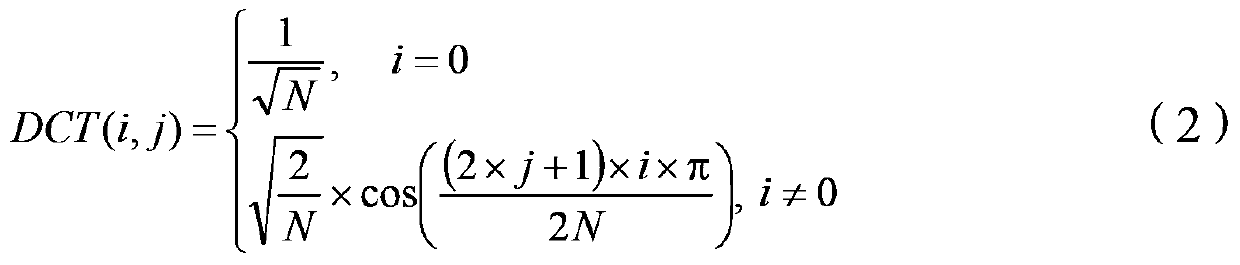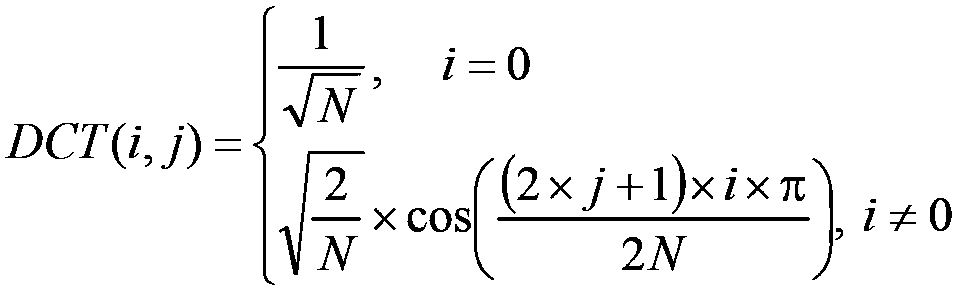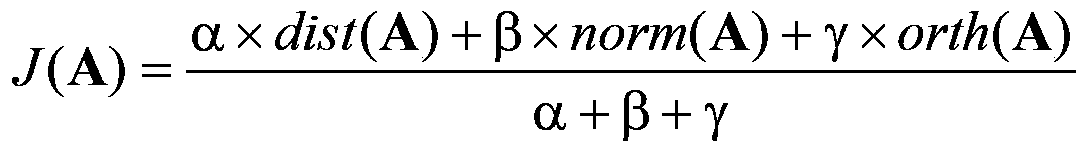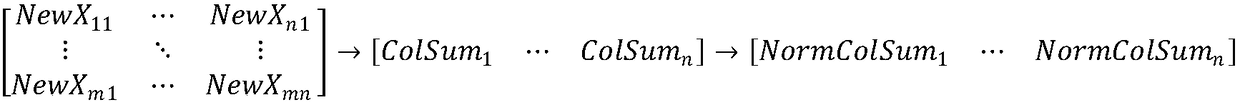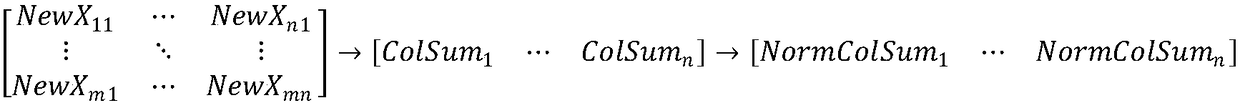Patents
Literature
33 results about "Transformation property" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
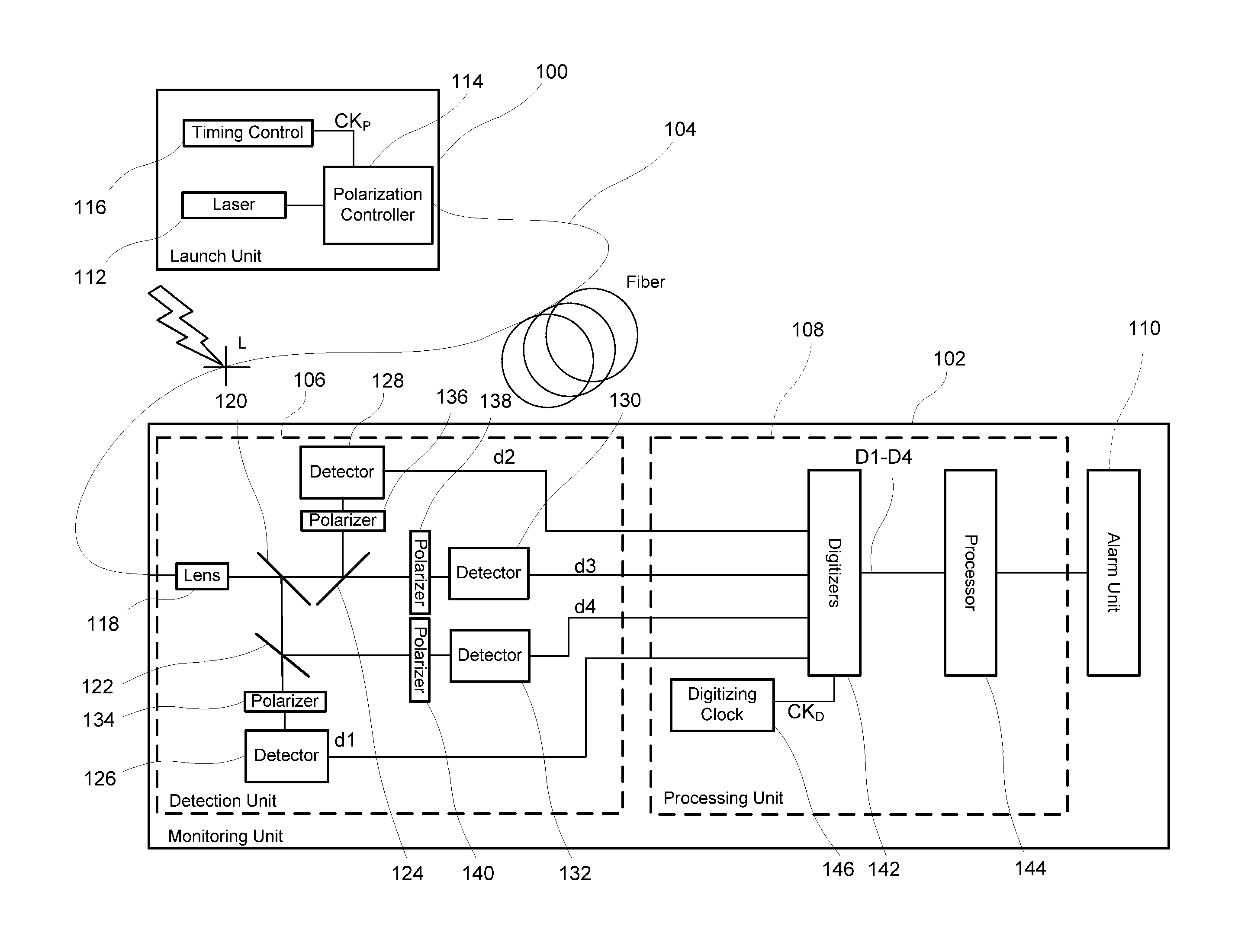
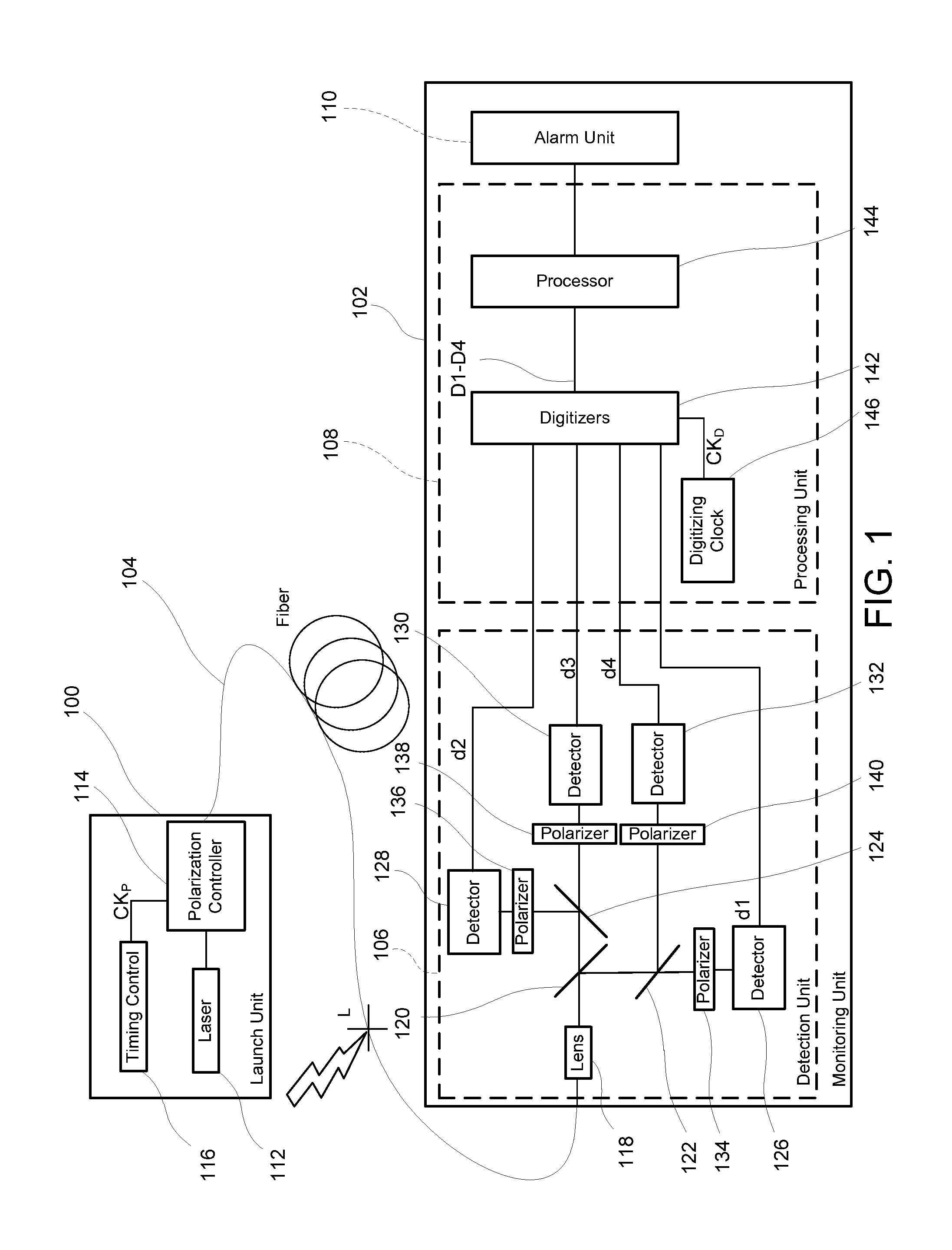
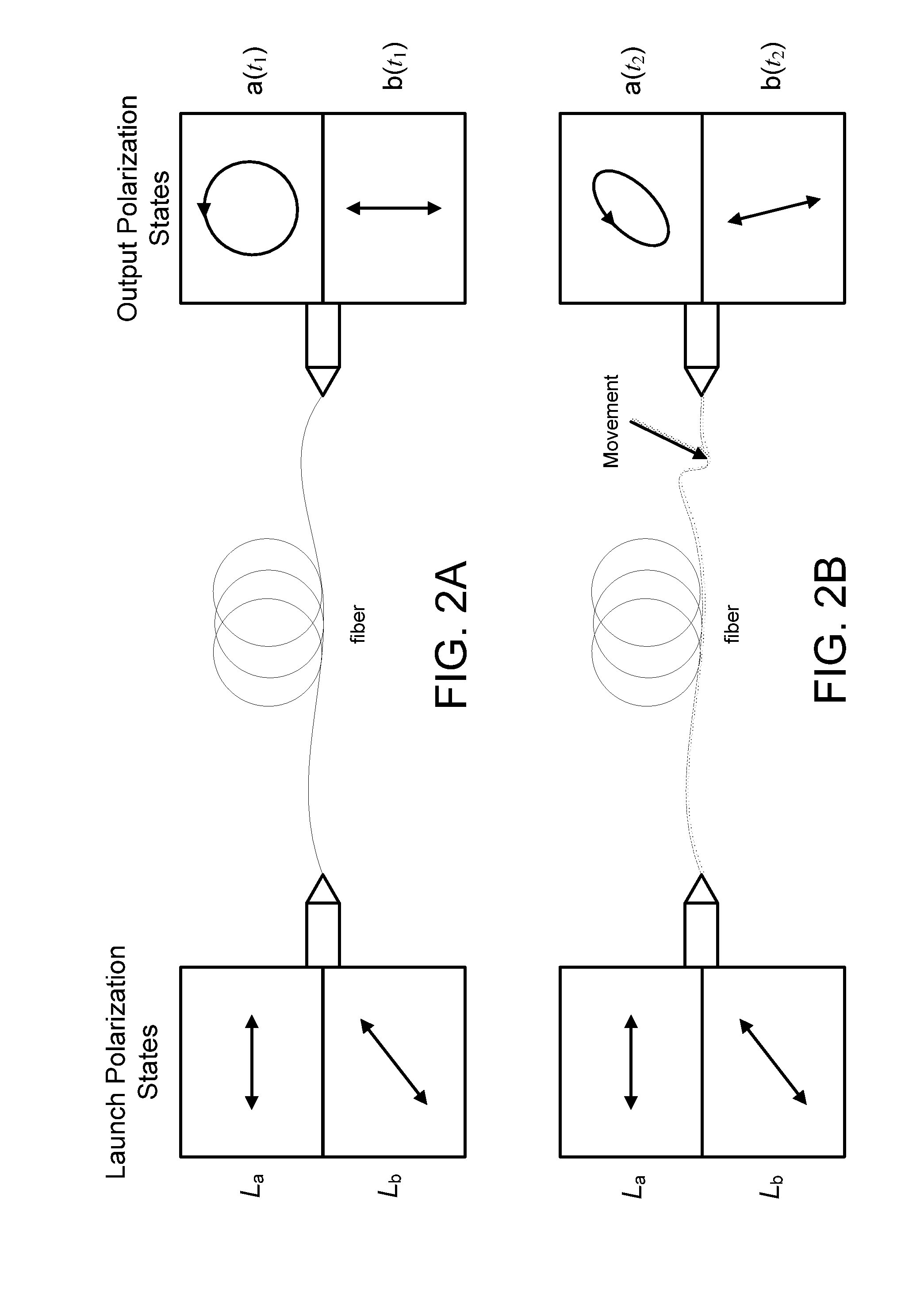
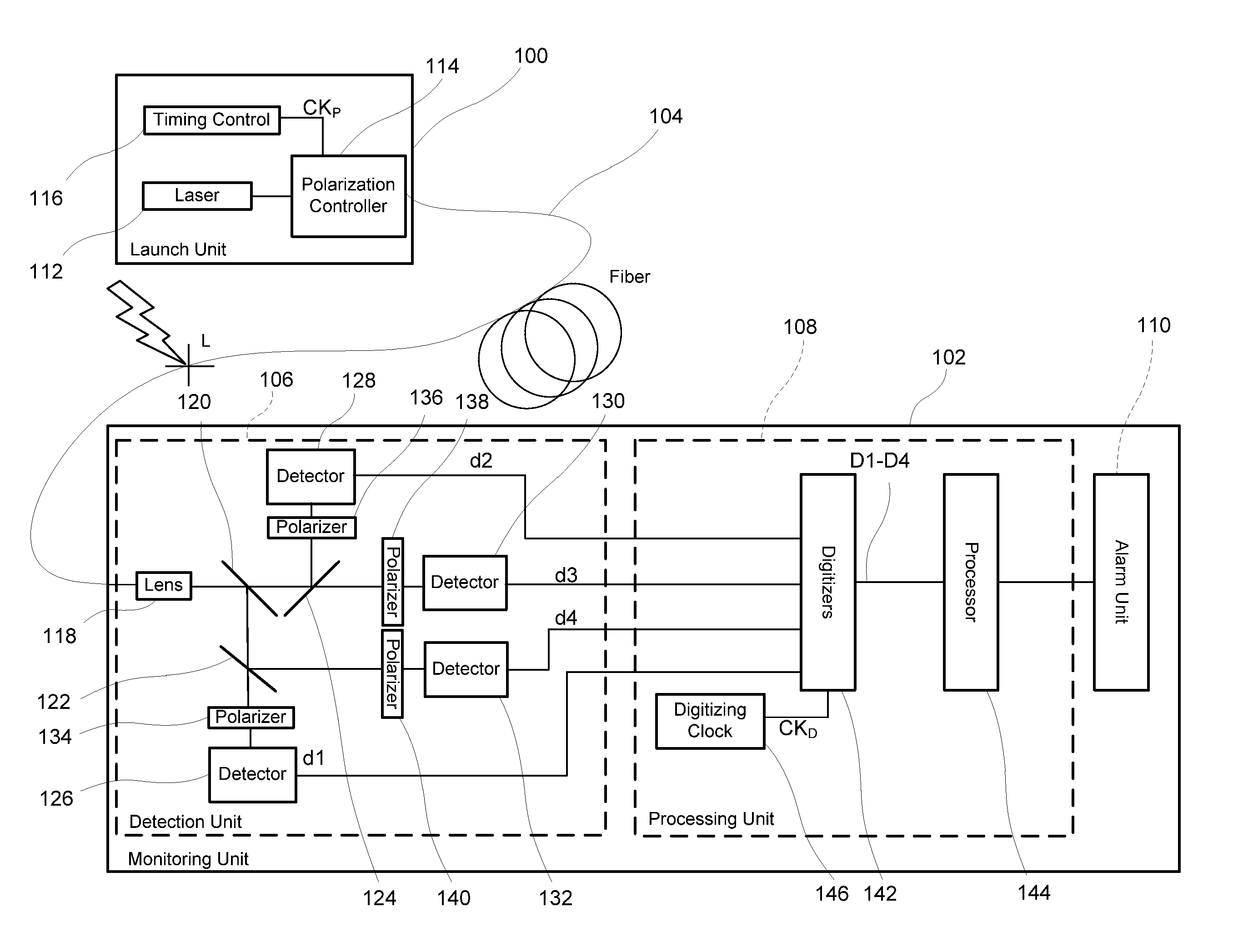
Monitoring for disturbance of optical fiber
ActiveUS20130188176A1Reduce deficiencyPolarisation-affecting propertiesTesting fibre optics/optical waveguide devicesFadingPhysics
Problems of excessive fading in systems for monitoring single-mode optical fiber for physical disturbances are addressed by launching into the fiber polarized light having at least two different predetermined launch states of polarization whose respective Stokes vectors are linearly-independent of each other; downstream from the first location, receiving the light from the fiber; analyzing the received light using polarization state analyzer means having at least two different analyzer states of polarization that are characterized by respective Stokes vectors that are linearly-independent of each other and detecting the analyzed light to provide corresponding detection signals; deriving from the detection signals measures of changes in polarization transformation properties of the fiber between different times that are substantially independent of said launch states and said detection states; and, on the basis of predefined acceptable physical disturbance criteria determining whether or not the measures are indicative of a reportable physical disturbance.
Owner:NETWORK INTEGRITY SYST
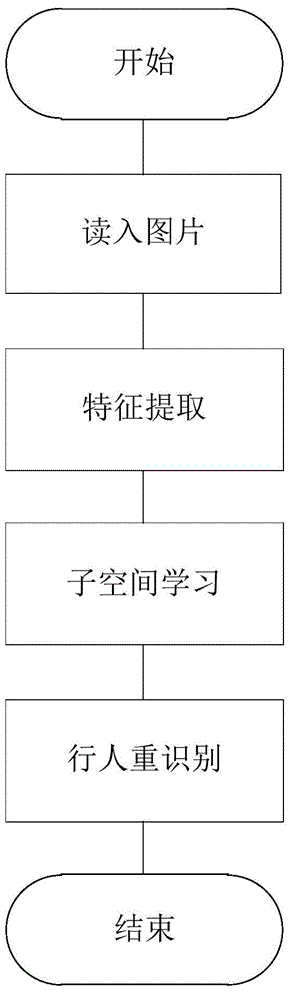
Method and system for re-identifying pedestrian based on view angle self-adaptive subspace learning algorithm
The invention provides a method and a system for re-identifying a pedestrian based on view angle self-adaptive subspace algorithm. With the method, a rectangular image only comprising a single pedestrian or a target rectangular frame cut from an original video image is used as an input image; a feature vector is extracted from the input image; a data set is divided into a training data set and a testing data set; a transformation matrix is acquired on the training data set by learning the view angle self-adaptive subspace learning algorithm; and the transformation matrix acquired on the testing data set by learning the view angle self-adaptive subspace learning algorithm is utilized to calculate the distance and re-identify the pedestrian. According to the invention, the fact that different cameras have different properties is taken into account; and different transformations are used to compensate the transformation properties of different cameras, thus the optimum transformation relationship between each pair of the cameras can be acquired more flexibly through learning; and the features under different cameras after the cameras are transformed can be closer to the ideal feature distribution.
Owner:合肥的卢深视科技有限公司
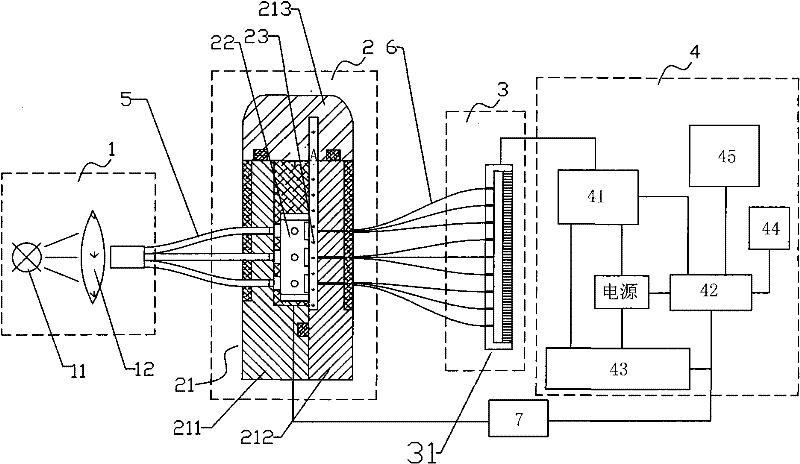
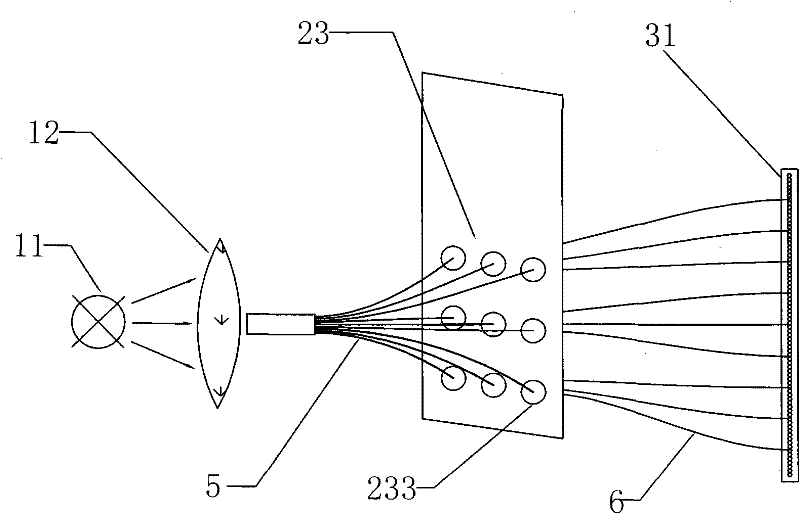
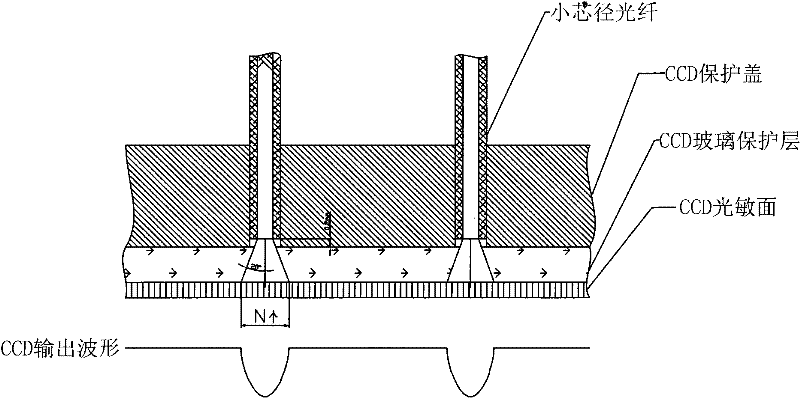
Mini-array poisonous gas detecting instrument
InactiveCN101726465AImprove sealingEasy loading and unloadingColor measuring devicesWithdrawing sample devicesGas detectorTransmitted light
The invention discloses a mini-array poisonous gas detecting instrument which can exhibit different colors when an air sensor reacts with poisonous gases, and the color of transmitted light can change correspondingly because incident light is spectrally absorbed. The mini-array poisonous gas detecting instrument comprises an incident light source system, an air sensing array system, a transmitted light acquisition system and a data analyzing and processing system, wherein the air sensing array system is provided with a sealed cabin which is characterized by good sealing effect, convenient loading and unloading, high stability and reasonable structural design, the transmitted light acquisition system uses a colored linear array CCD which is characterized by good sensibility and photoelectric transformation property, the data analyzing and processing system takes a microprocessor as a core element to form a perfect system of element controlling, data receiving, processing and analyzing, and is characterized by high automation degree, quick analysis speed and precise result. The mini-array poisonous gas detecting instrument is compact in structure and small in size, is especially beneficial to quickly detecting the slight quantity of extremely poisonous gas, and has good developing prospect.
Owner:CHONGQING UNIV
Method and device for investigation of phase transformations in metals and alloys
ActiveUS7473028B1Reliable and fast and inexpensive monitoringImprove bindingInvestigating phase/state changeAlloyMaterials science
A device and method for investigating phase transformation properties and structural changes of materials. In one form, the device simulates actual thermal processing conditions, while the method can be used in both simulations as well as in actual processing conditions. An analysis using at least one of the device and method is referred to as a single sensor differential thermal analysis, as it compares the temperature recorded in a measured specimen against a reference thermal history without requiring the derivation of the reference thermal history from measured reference temperatures.
Owner:THE OHIO STATES UNIV
Zanamivir nasal in situ jellies with phase variation property and preparing method thereof
InactiveCN101229122AEffective absorptionExtended stayOrganic active ingredientsAerosol deliveryNasal cavityWhole body
The invention relates to the new dosage form of zanamivir, which is a nasal in-situ gel with phase transformation property and preparation methods, the invention is made of the original drug of zanamivir, hydrophilic gel materials which are sensitive to environments and assistant materials which can be accepted in pharmacy, after being absorbed by nasal mucosa, the invention can have systemic functions and improve the partial concentration of drugs in respiratory tracts, when room temperature is about 20 DEG C and the invention is in storing state, the invention is in the state of liquid, while after being put into the physiological conditions of nasal cavities, the invention can be quickly formed into gel on the surface of nasal mucosa, so that the detained time of drugs can be prolonged, biological availability can be improved and the compliance of patients can be perfected, and the viscosity of the invention is proper, after being formed into gel, the invention can be detained on the surface of the nasal cavity for a rather long time, which has no nasal ciliary toxicity.
Owner:SHANGHAI INST OF MATERIA MEDICA CHINESE ACAD OF SCI
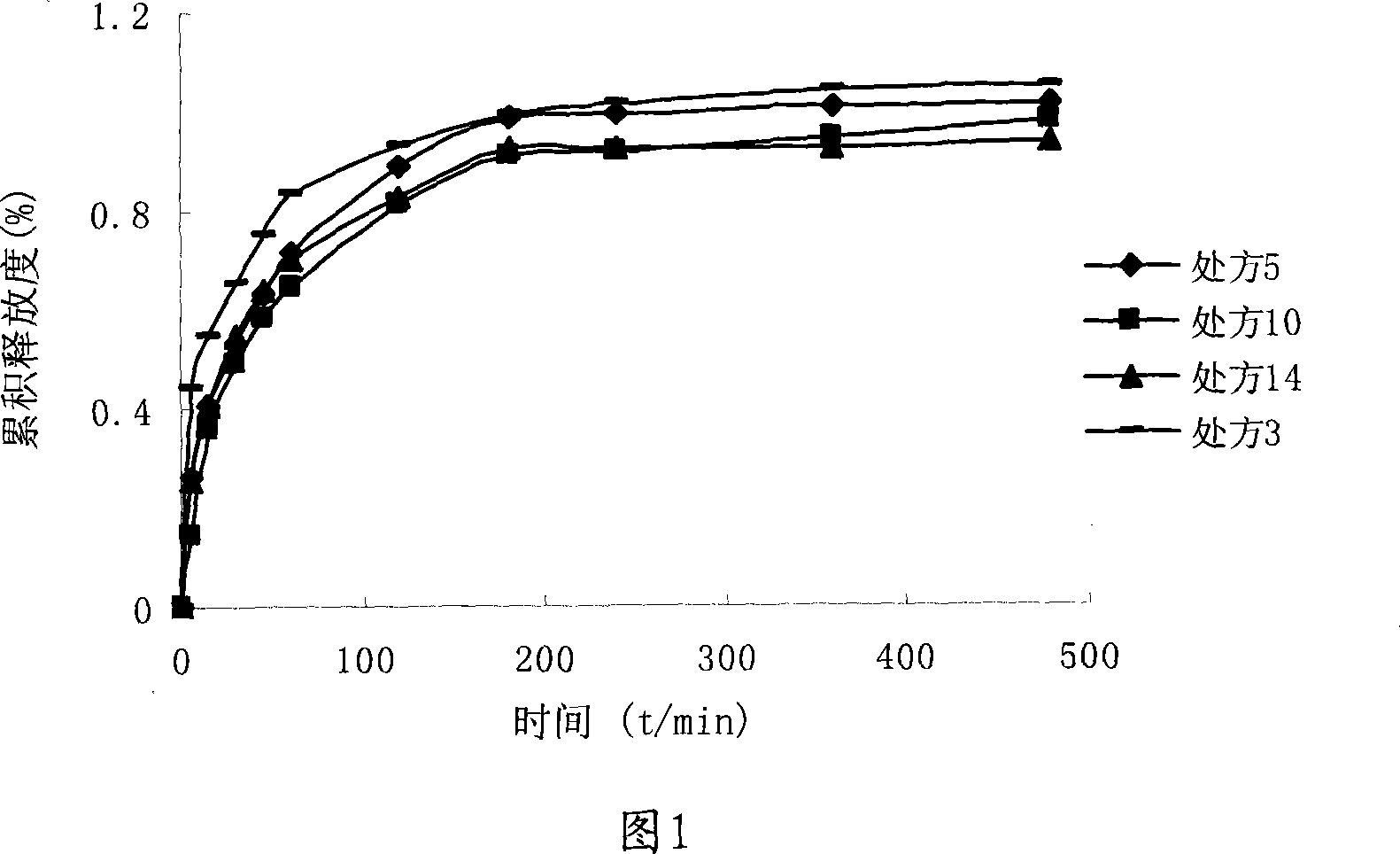
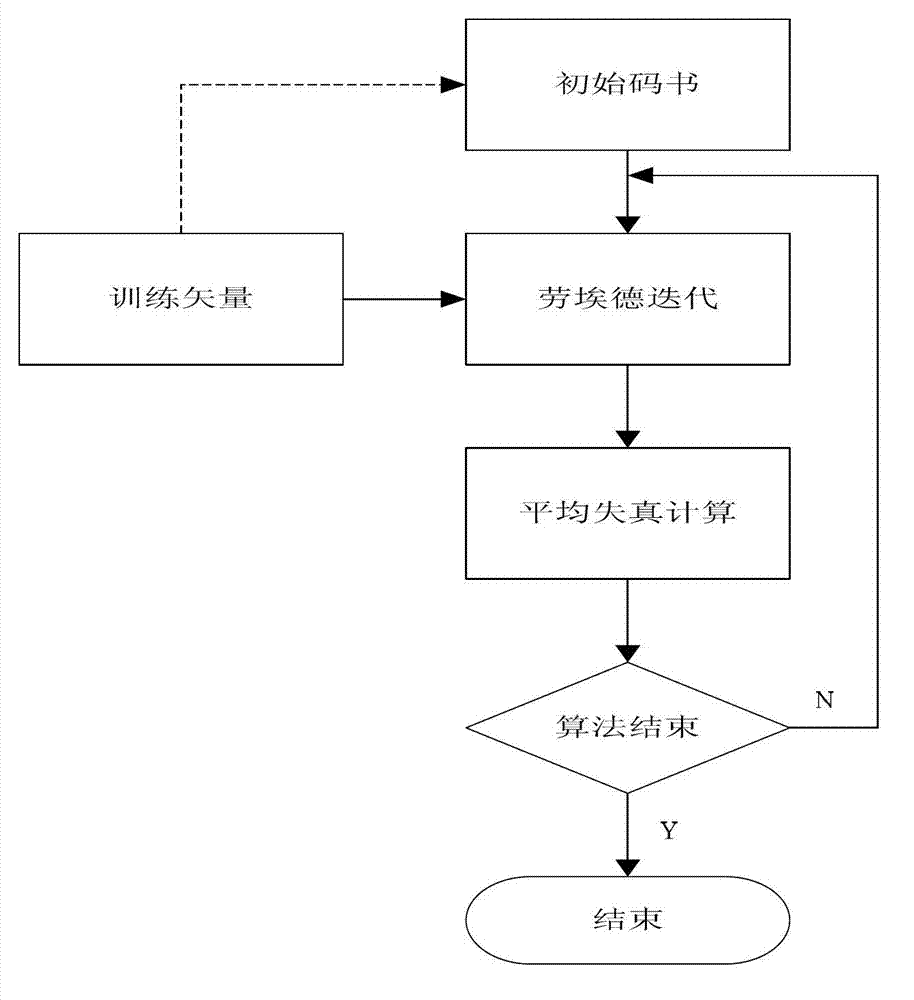
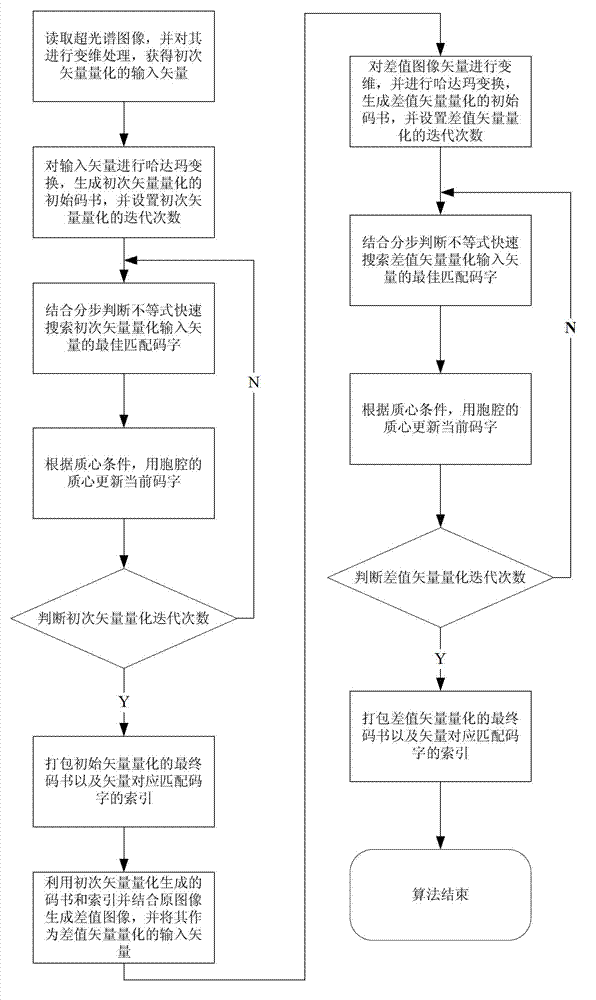
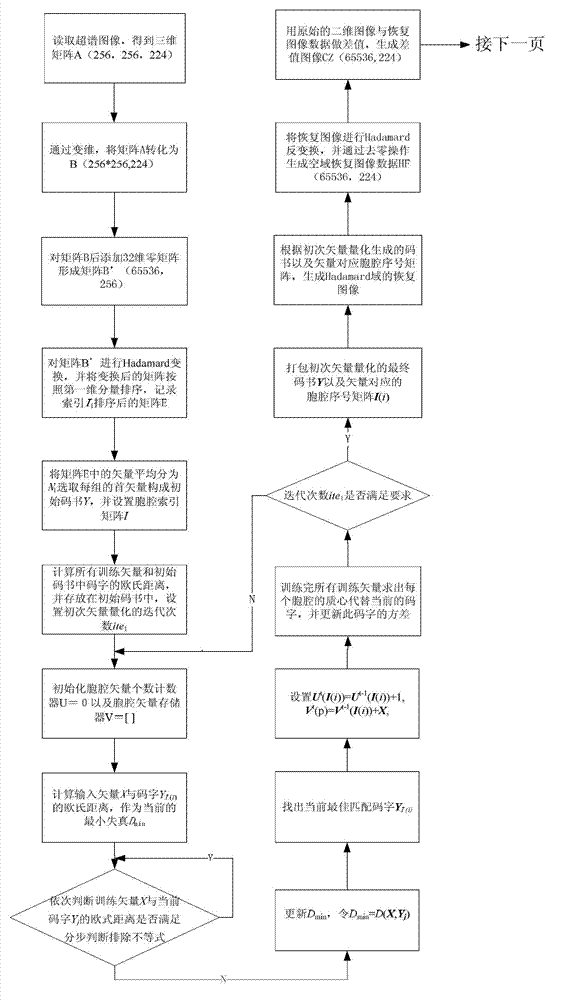
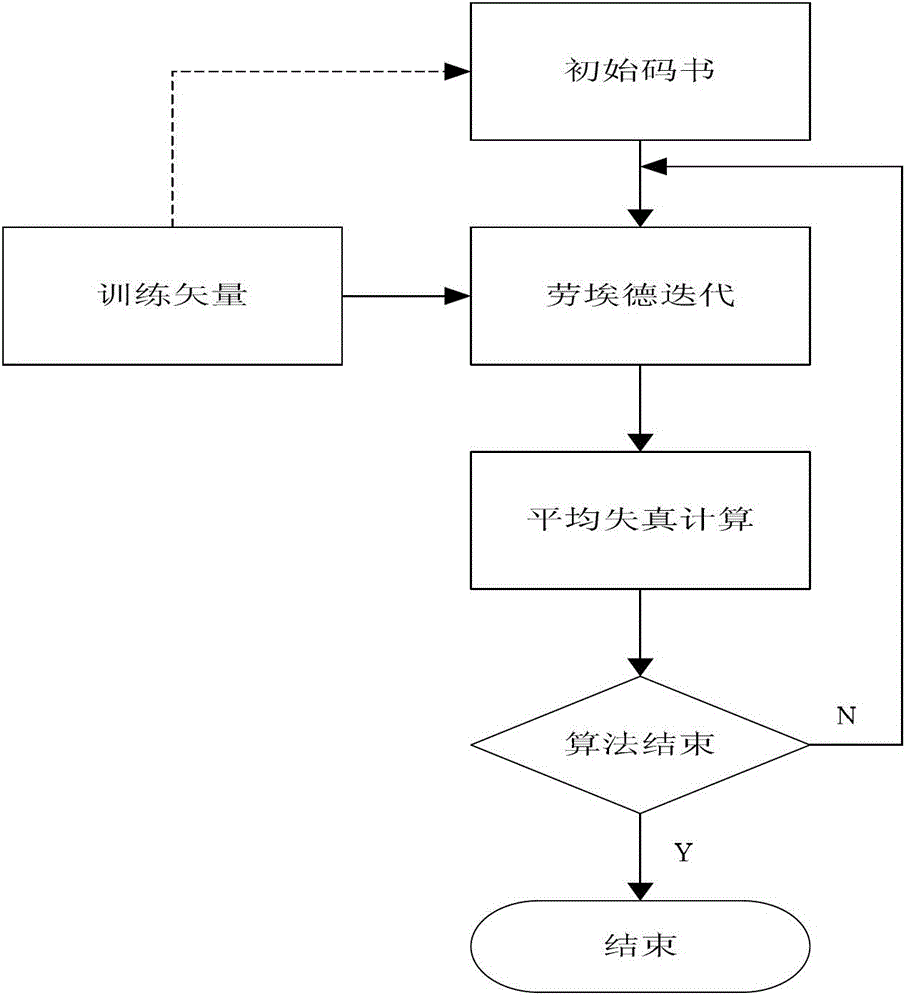
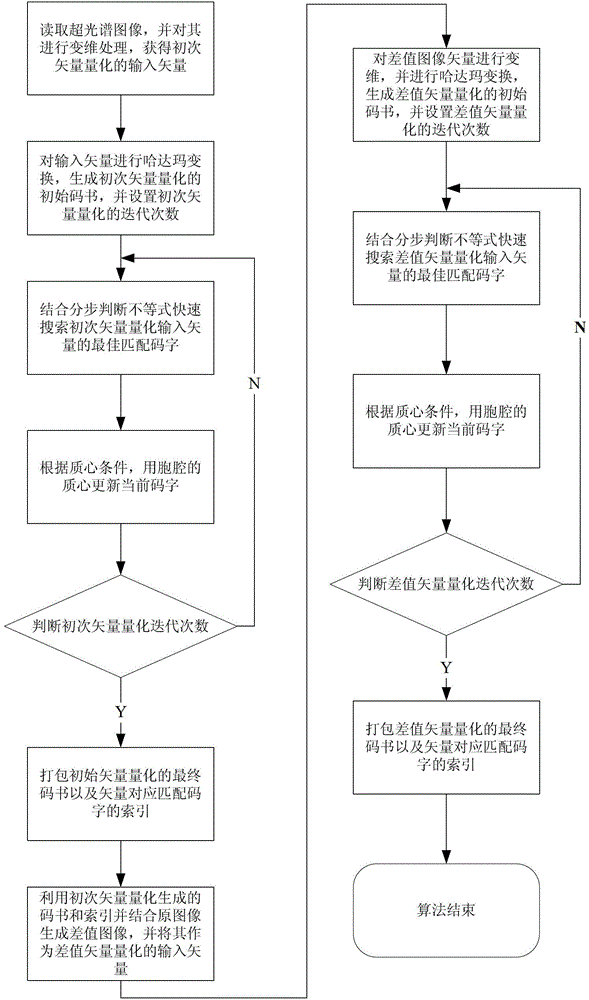
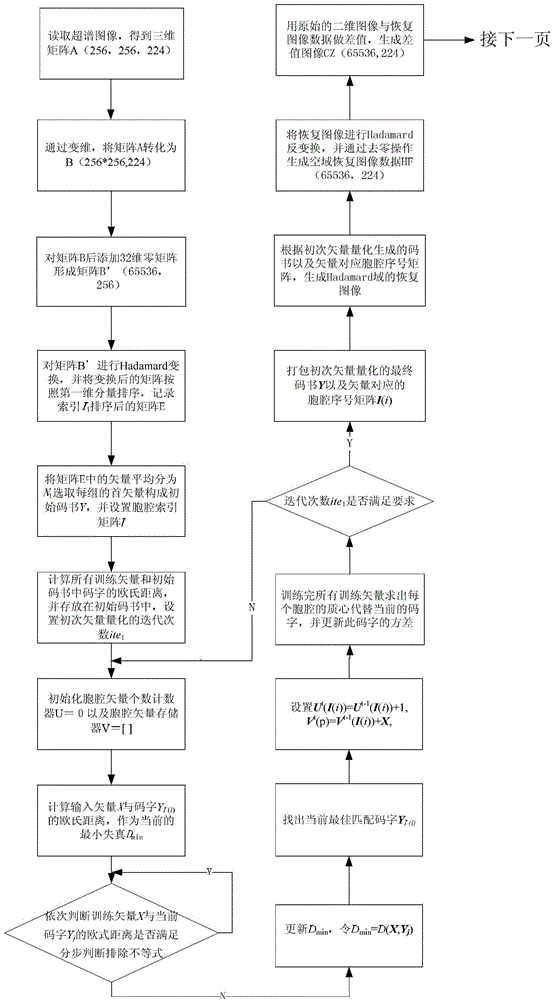
Quick difference value vector quantitative compression coding method of ultra-spectrum signal
ActiveCN102905137AFlexible data handlingFlexible transmissionTelevision systemsDigital video signal modificationCode bookAlgorithm
The invention provides a quick difference value vector quantitative compression scheme of an ultra-spectrum signal, which comprises the steps of: in a Hadamard domain, constructing an initial code book of initial vector quantization, carrying out quick clustering, generating a final code book and an index of initial vector quantization; obtaining difference value image data from the restored image of the initial vector quantization and an original image to be used as an input vector of the difference value vector quantization, carrying out Hadamard transformation on the input vector; and constructing an initial code book of the difference value vector quantization, and carrying out quick clustering to generate a final code book and an index of the difference value vector quantization. The quick difference value vector quantitative compression scheme provided by the invention is combined with the Hadamard transformation property and the characteristic of reducing the quantization error by the difference value vector quantization; and compared with the traditional LBG algorithm, the quick difference value vector quantitative compression scheme has the advantages that under the condition of same compression ratio, the computing quantity can be greatly reduced, the image restoring quality can be improved, and the purpose of quick compression and coding is achieved.
Owner:CHONGQING UNIV OF POSTS & TELECOMM
Rapid preparation method of monoclinic phase VO2 metal-insulator phase transformation ceramic material
InactiveCN106083044AHas a single-phase structureEnrich physical property transformationMetal insulatorCeramic
The invention relates to a rapid preparation method of a monoclinic phase VO2 metal-insulator phase transformation ceramic material. The method comprises the following steps: reducing V2O5 with citric acid by a hydrothermal method, carrying out centrifugal cleaning to obtain an orthorhombic phase VO2 nanopowder, carrying out high-temperature heat treatment to obtain a monoclinic phase VO2 powder, and finally compressing and sintering to obtain the monoclinic phase VO2 metal-insulator phase transformation ceramic. The process is simple; preparation efficiency is high; sintering temperature is low; energy consumption is low; cost is low; the method is environment friendly, nontoxic and pollution-free; and the prepared ceramic has good phase transformation property and stable and uniform structure.
Owner:HUAZHONG UNIV OF SCI & TECH
Method and device for investigation of phase transformations in metals and alloys
ActiveUS20090119057A1Reliable and fast and inexpensive monitoringImprove bindingDigital computer detailsThermometer testing/calibrationAlloyMetal
A device and method for investigating phase transformation properties and structural changes of materials. In one form, the device simulates actual thermal processing conditions, while the method can be used in both simulations as well as in actual processing conditions. An analysis using at least one of the device and method is referred to as a single sensor differential thermal analysis, as it compares the temperature recorded in a measured specimen against a reference thermal history without requiring the derivation of the reference thermal history from measured reference temperatures.
Owner:THE OHIO STATES UNIV
Nanosecond reversible phase transformation material and method for measuring phase transformation mechanism of same
ActiveCN102255044AFast reversible phase transitionMaterial analysis using wave/particle radiationElectrical apparatusPolyatomic ionNanosecond
The invention discloses a nanosecond reversible phase transformation material and method for measuring a phase transformation mechanism of the same. The nanosecond reversible phase transformation material has the property of generating nanosecond reversible transformation under the single action of multiple external factors or combined action of the multiple external factors. The multiple external factors include electric field, pressure and temperature; the nanosecond reversible transformation is reversible change between order distribution and disorder distribution of atoms, atom groups or molecules inside the nanosecond reversible phase transformation material so that the nanosecond reversible phase transformation material macroscopically presents a phase transformation process generated between a crystalline state and an amorphous state, the time spent by the phase transformation process is within 0.1-1000 nanoseconds, wherein the crystalline state of the material is of a lattice structure of a cubic system; meanwhile, the material has a stable high-temperature solid phase, a stoichiometric ratio of electric neutrality, P orbit ionization degree of 0.02<r sigma'<0.31 and s-p orbit hybridization degree of 1.6<rpi-1<2.5. The invention also provides a method for measuring the phase transformation mechanism by adopting experiment means, which can be applied to the explanation of the phase transformation mechanism of the material and prediction of the phase transformation property of the material and design of a novel phase transformation material.
Owner:SHANGHAI INST OF MICROSYSTEM & INFORMATION TECH CHINESE ACAD OF SCI
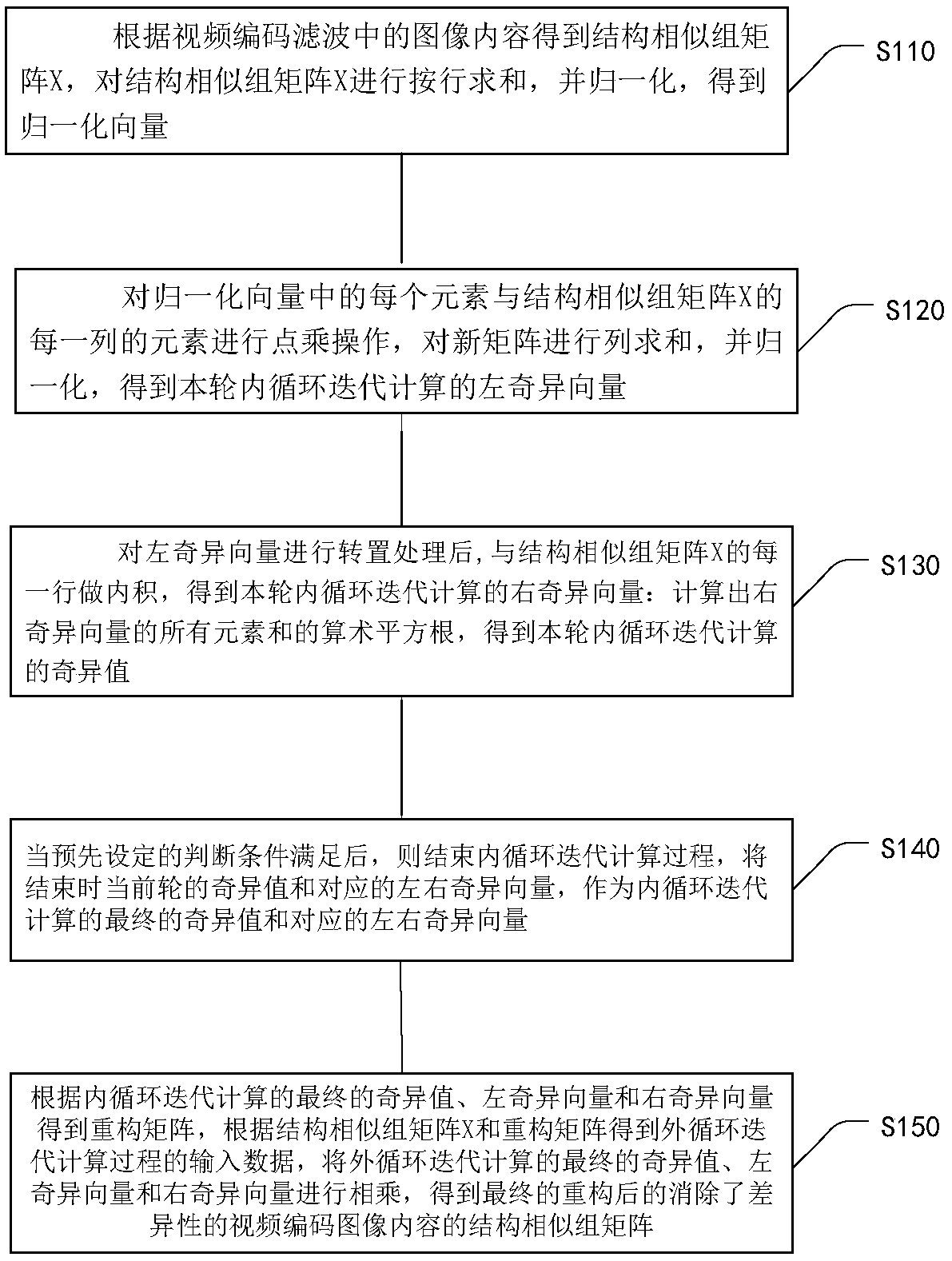
Method for eliminating block effect of video coding images
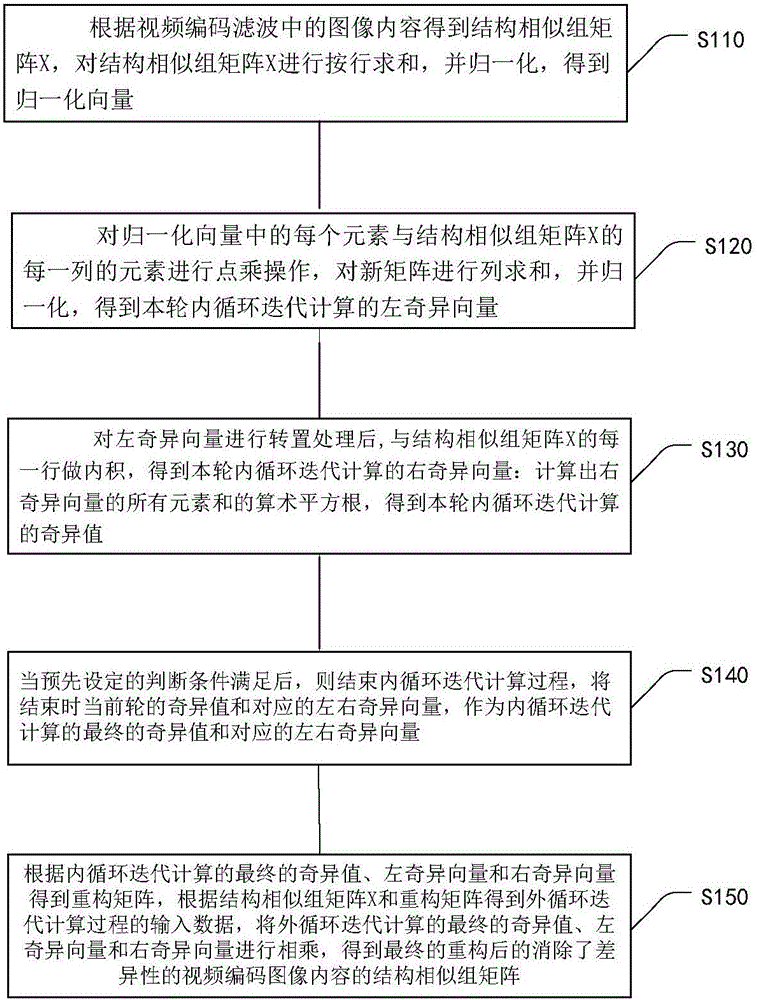
ActiveCN105763880AQuick calculationEliminate blockingDigital video signal modificationSingular value decompositionPattern recognition
The embodiment of the invention provides a method for eliminating the block effect of video coding images. According to the method, summation normalization operation is performed on a matrix from rows and columns based on the linear transformation property of the matrix, the maximum singular value and the corresponding left and right singular vectors of a current matrix module are worked out through iteration, and the purpose of quick singular value calculation is achieved. Through the method, singular value decomposition filtering operation can be carried out under the condition that the matrix is not decomposed completely, the algorithm complexity is reduced effectively during low-rank matrix processing, and the accuracy of the singular value can be adjusted according to the actual need in the algorithm. The method can be effectively applied to matrix singular value decomposition operation in the process of video coding and filtering requiring high real-time performance to effectively eliminate the difference between rows and between columns in a video coding image matrix, improve the visual effect of video coding images and improve the coding performance.
Owner:PEKING UNIV
Rearrangement ST-based low-signal-noise-ratio micro-seismic event identification method
ActiveCN107944469AImprove classification accuracyEfficient retrievalCharacter and pattern recognitionFrequency spectrumTime frequency spectrum
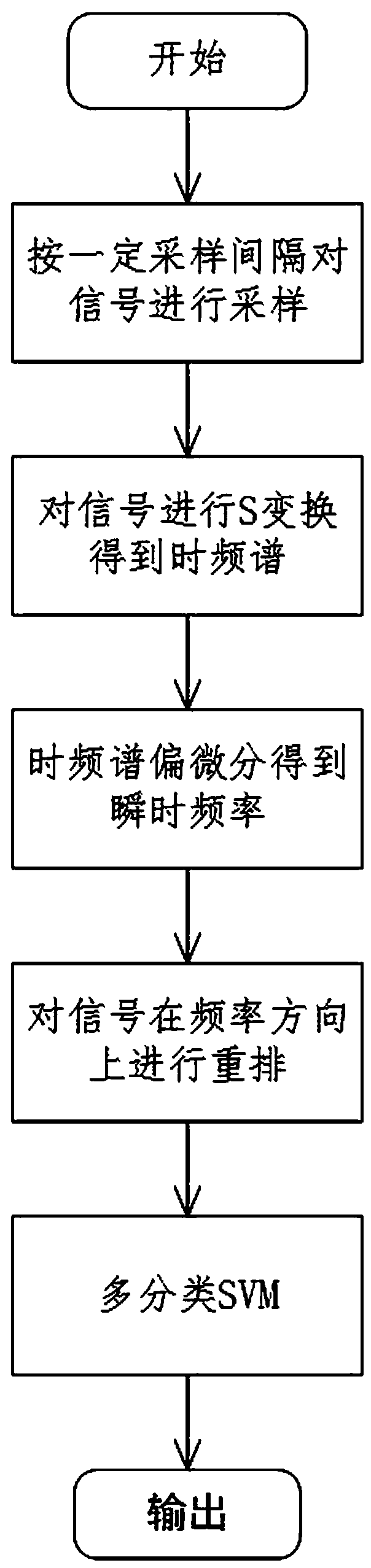
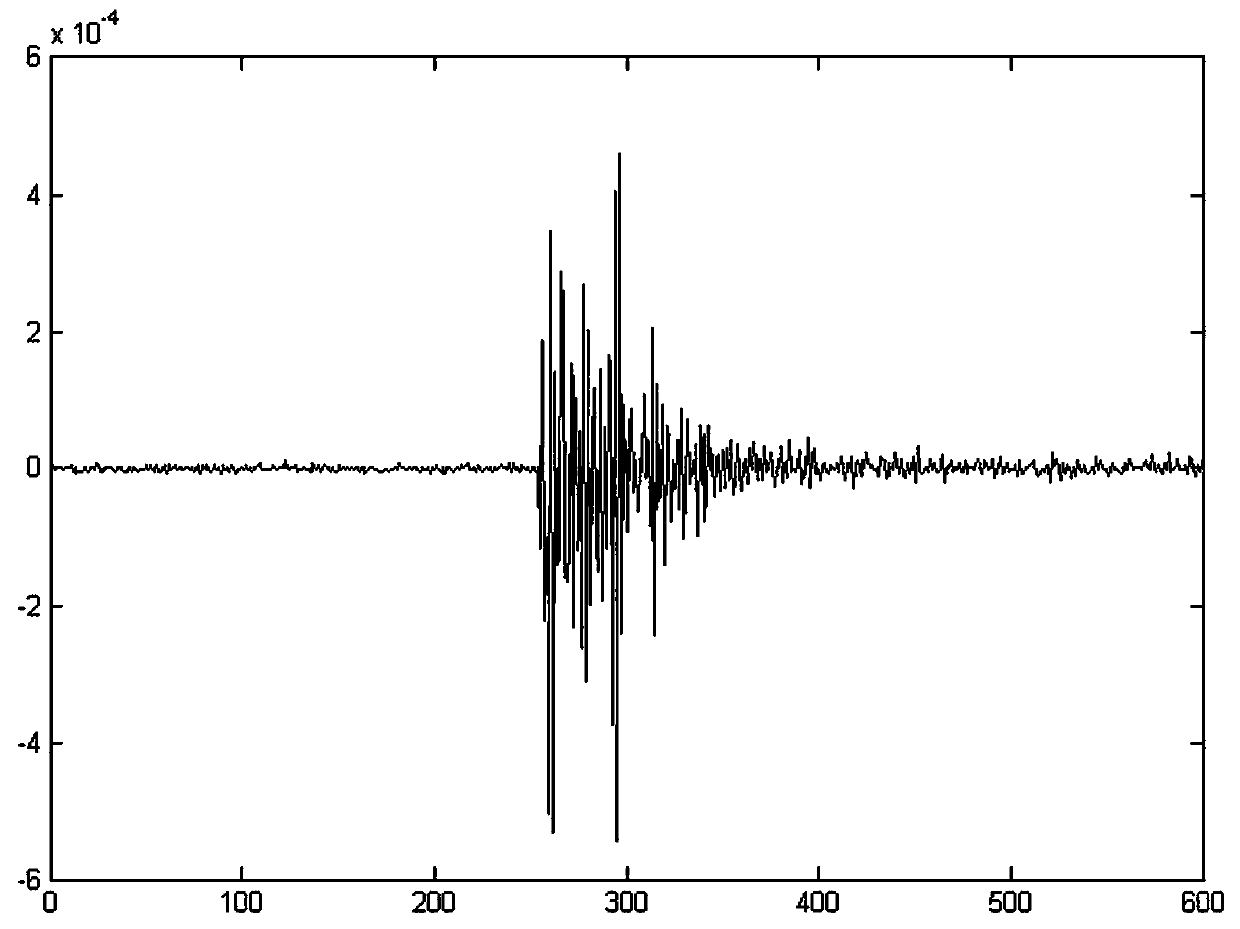
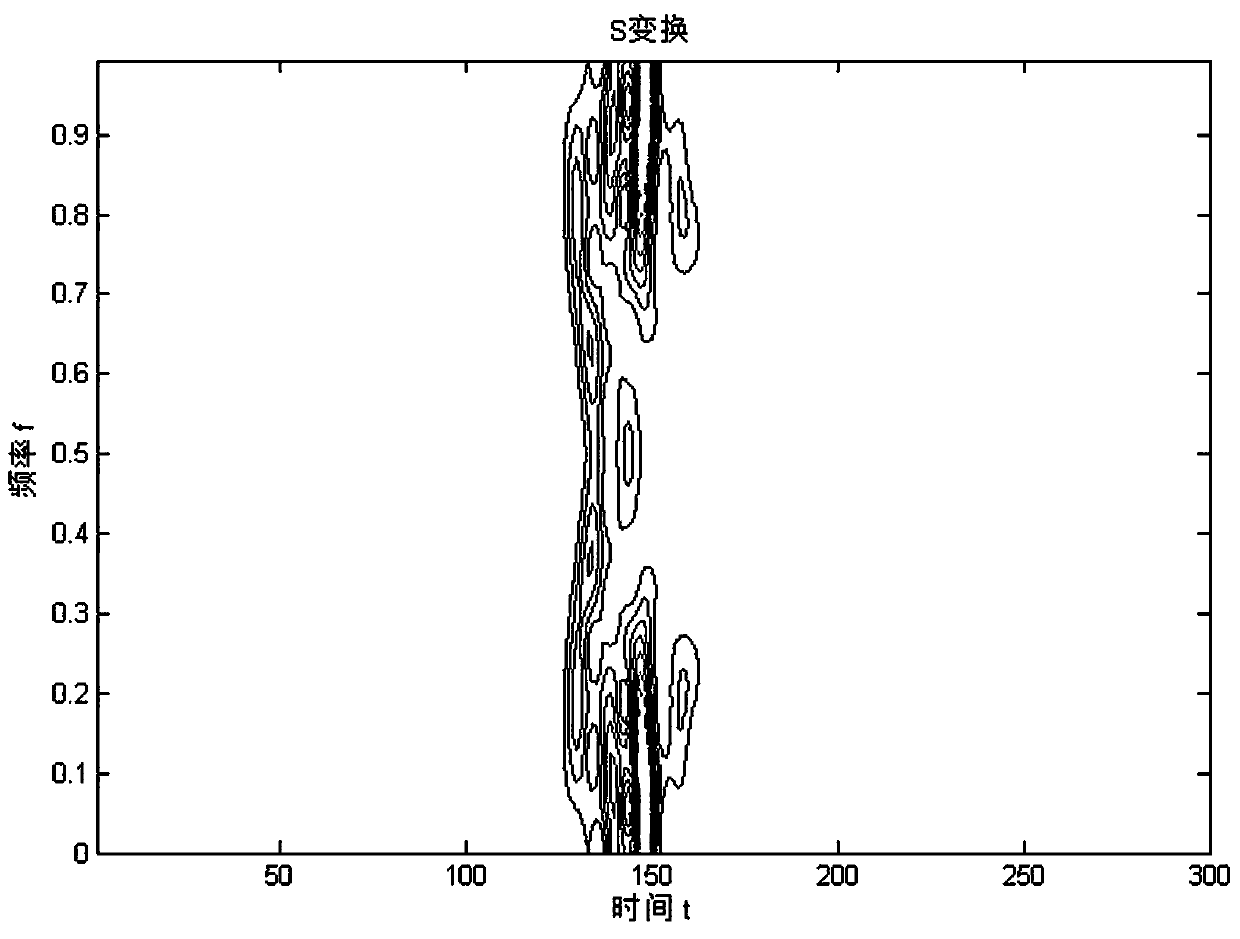
The invention discloses a rearrangement ST-based low-signal-noise-ratio micro-seismic event identification method. The method comprises the steps of firstly, performing ST on micro-seismic data to obtain time frequency spectrums of signals; secondly, according to a Parserval theorem and rules on scale transformation and translation in Fourier transformation properties, performing partial differential operation on the time frequency spectrums to obtain instantaneous frequencies of the signals; thirdly, converting a value calculated at any point for a spectrogram to a gravity center of energy distribution, thereby obtaining a rearranged time frequency matrix of the time frequency spectrums of the signals in a frequency direction; and finally, building a multi-classification SVM for realizingclassification of micro-seismic signals, blast signals and mechanical noises. The problems of low identification rate, rough classification and low classification accuracy of low-signal-noise-ratio micro-seismic signals in the prior art are solved, so that the time frequency resolution of the low-signal-noise-ratio micro-seismic signals is remarkably improved, the classification is accurate and the classification of multiple weak signals is realized; and the method can be well applied to the mine safety production and coal mine illegal mining monitoring technologies.
Owner:GUILIN UNIV OF ELECTRONIC TECH
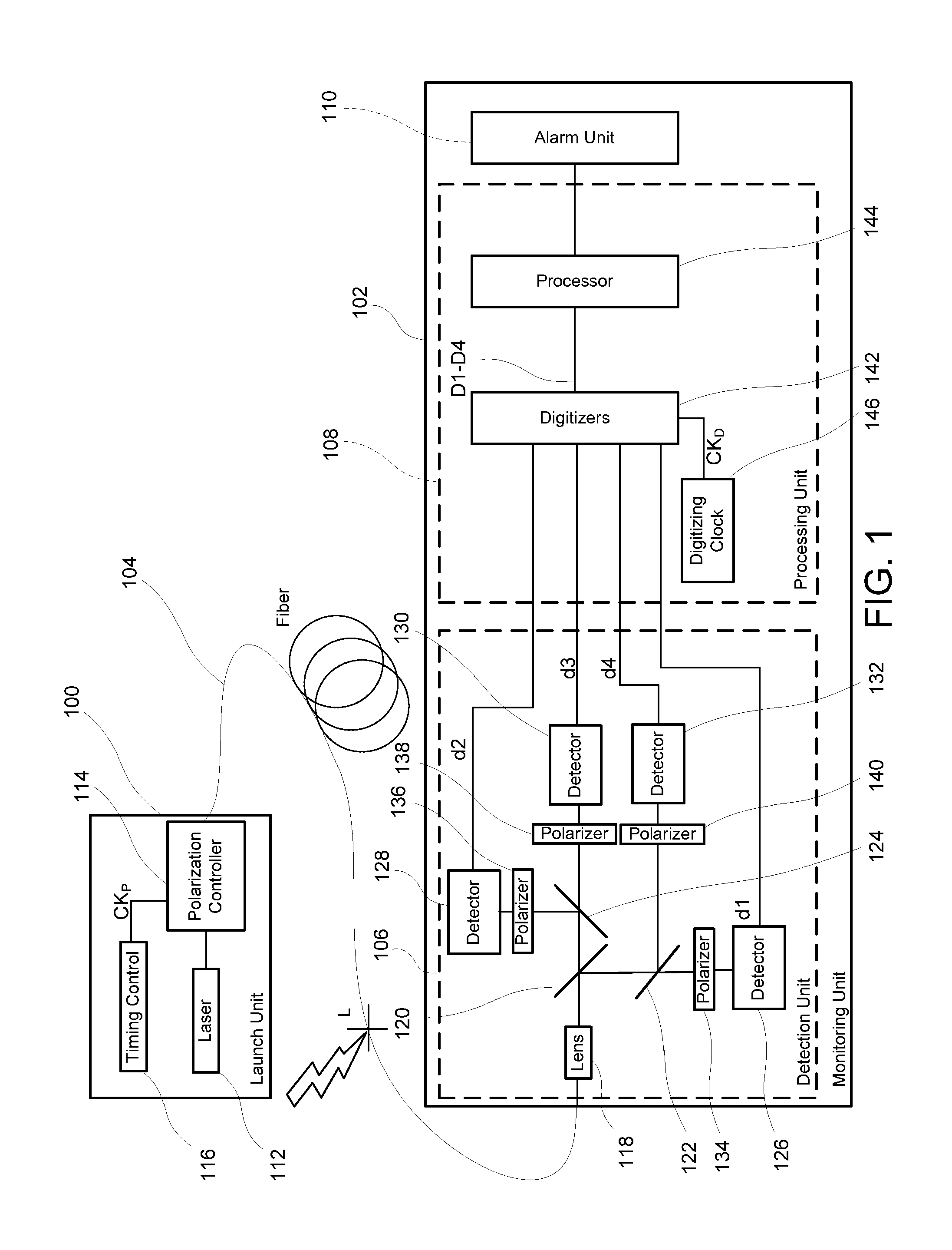
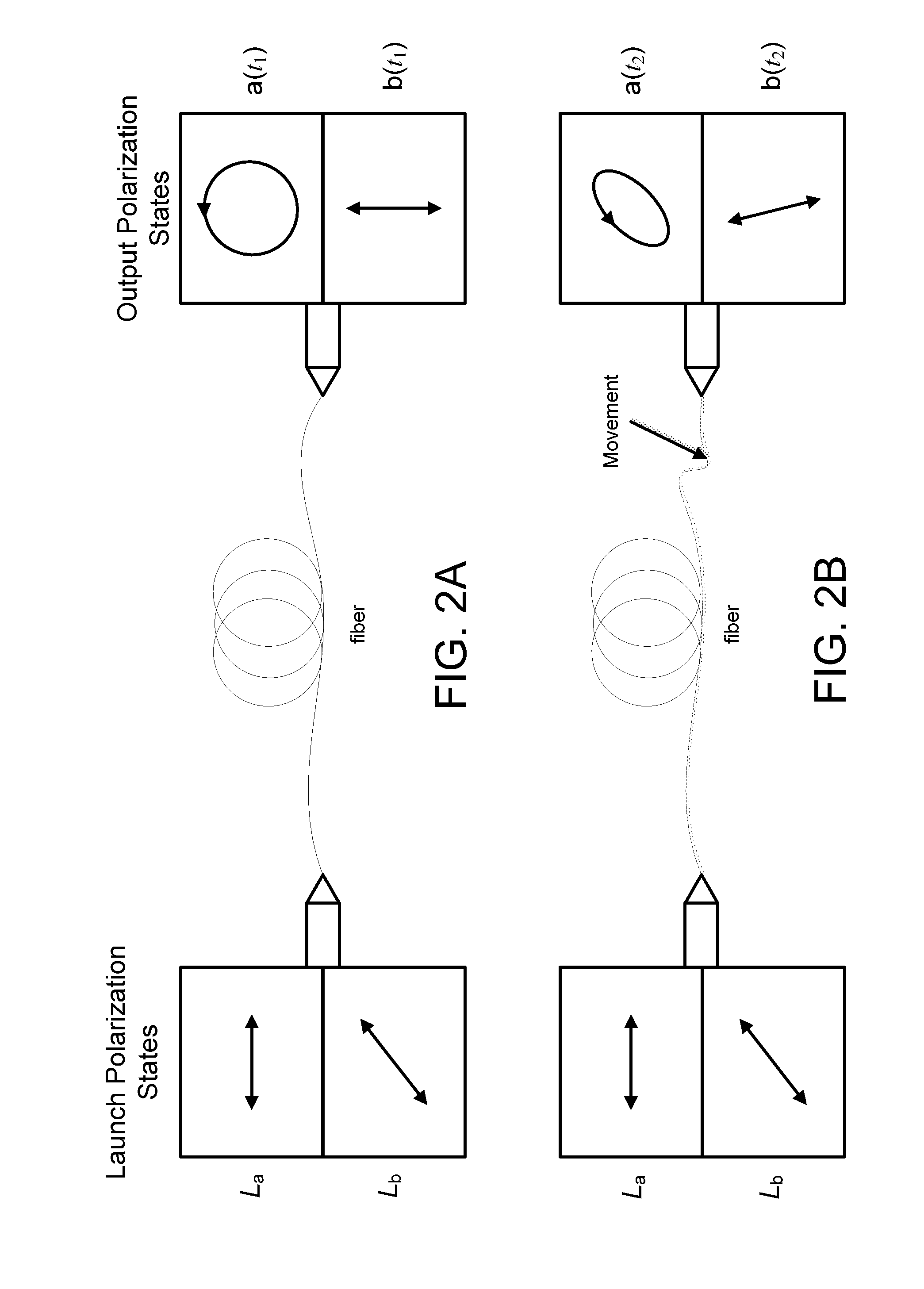
Monitoring for disturbance of optical fiber
ActiveUS8736826B2Material analysis by optical meansTransmission monitoringFadingSingle-mode optical fiber
Problems of excessive fading in systems for monitoring single-mode optical fiber for physical disturbances are addressed by launching into the fiber polarized light having at least two different predetermined launch states of polarization whose respective Stokes vectors are linearly-independent of each other; downstream from the first location, receiving the light from the fiber; analyzing the received light using polarization state analyzer means having at least two different analyzer states of polarization that are characterized by respective Stokes vectors that are linearly-independent of each other and detecting the analyzed light to provide corresponding detection signals; deriving from the detection signals measures of changes in polarization transformation properties of the fiber between different times that are substantially independent of said launch states and said detection states; and, on the basis of predefined acceptable physical disturbance criteria determining whether or not the measures are indicative of a reportable physical disturbance.
Owner:NETWORK INTEGRITY SYST
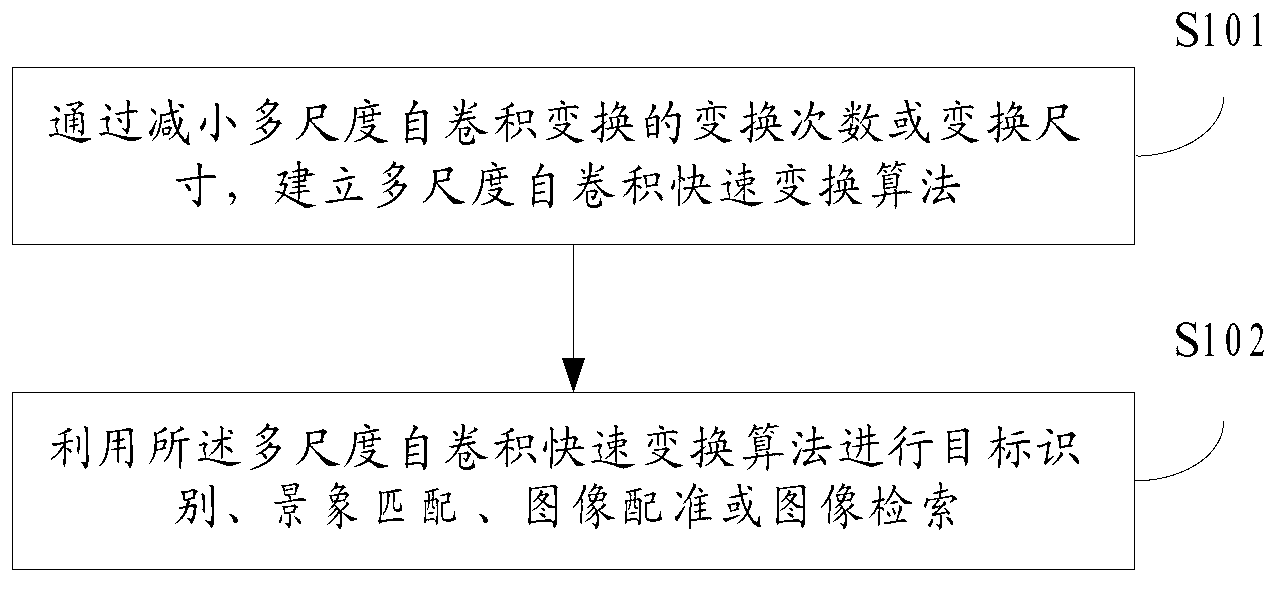
Fast transform algorithm application method and apparatus for multi-scale auto-convolution
InactiveCN103295183AReduce the number of transformationsTransform size upImage data processing detailsSystems analysisFast algorithm
Owner:RECONNAISSANCE INTELLIGENCE EQUIP INST OF ARMAMENT ACADEMY OF THE PEOPLES LIBERATION ARMY AIR FORCE
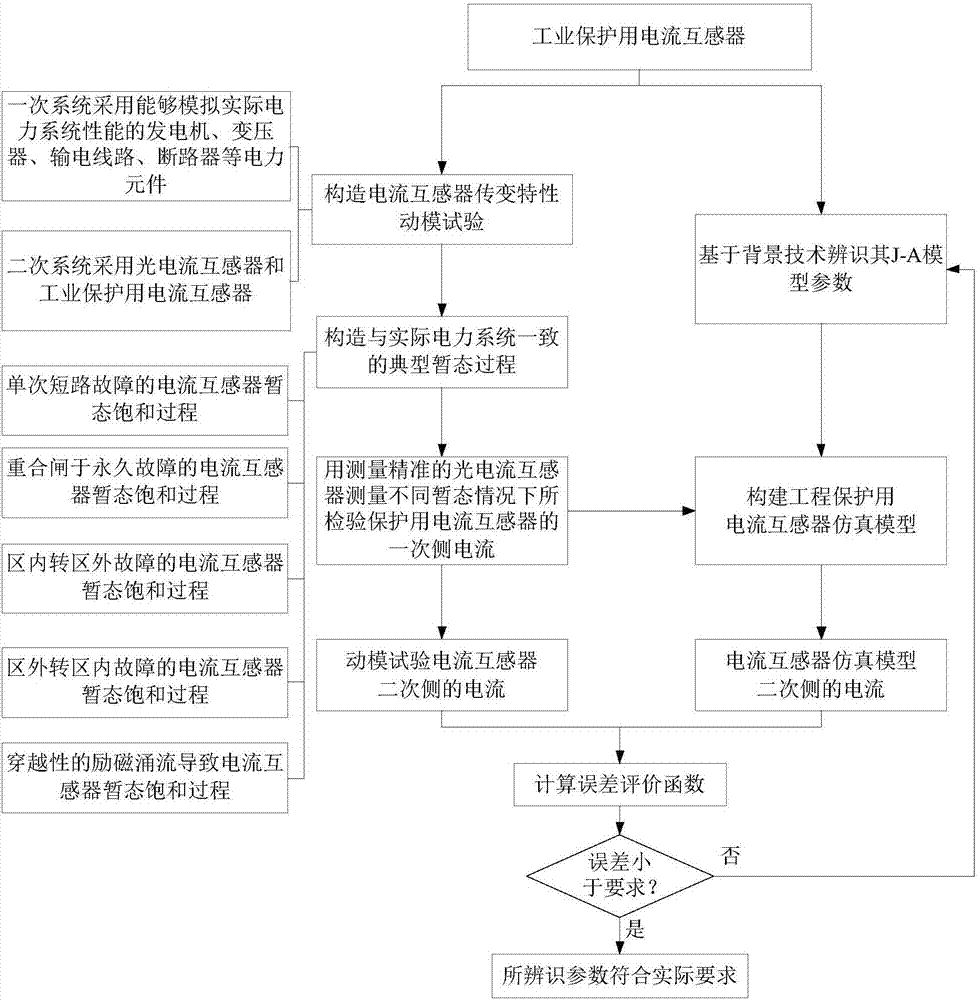
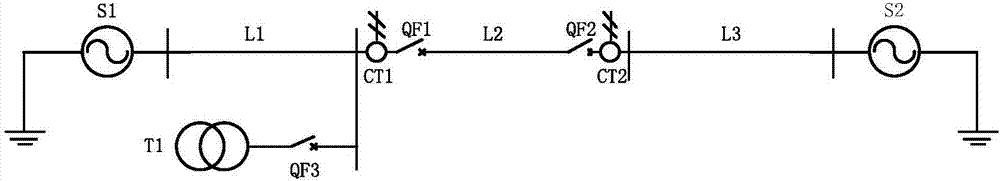
J-A model parameter test method of current transformer for protection
The invention discloses a J-A model parameter test method of a current transformer for protection. The method comprises model parameter identification, multi-transient state dynamic test, current transformer simulation, error function evaluation and the like and can judge whether identified J-A model parameters of the current transformer are in line with actual needs. According to the J-A model parameter test method of the current transformer for protection provided by the invention, test and simulation are combined, and therefore, the defects of the identified parameters caused by intelligent algorithms or experimental conditions can be effectively discovered. With the method adopted, whether the transformation properties of the current transformer which are reflected by the model parameters of the current transformer are consistent with actual conditions can be verified under typical power system fault transient state conditions; and suggestions for judging whether the model parameters of the current transformer accord with actual requirements can be provided from the aspect of application, and therefore, a sound foundation can be conveniently laid for the guidance of accident analysis of a site and protection responsive strategies from the perspective of simulation.
Owner:STATE GRID CORP OF CHINA +2
Low-toxicity high-flame-retardance low-shrinkage resin for vehicles and manufacturing method thereof
The invention provides a low-toxic, high-flame-retardant, low-shrinkage automotive resin and a manufacturing method thereof. The low-toxic, high-flame-retardant, low-shrinkage automotive resin includes vinyl resin or alkyd resin, cyclopentadiene resin, Shrinkage agent, white carbon black, dispersant, wetting agent, triethyl phosphate, phenylethylamine, 10% cobalt water, polymerization inhibitor, aluminum hydroxide, low volatility control agent. The invention can effectively control the volatilization of styrene, not only further reduce defects such as shrinkage, warping and deformability of products, improve the quality of downstream products, but also reduce the production links of customers and greatly reduce the production costs of customers.
Owner:ZHANGJIAGANG JIULI NEW MATERIAL TECH CO LTD
Bi-CWSAR (Bistatic Continuous Wave Synthetic Aperture Radar) remote sensing imaging method
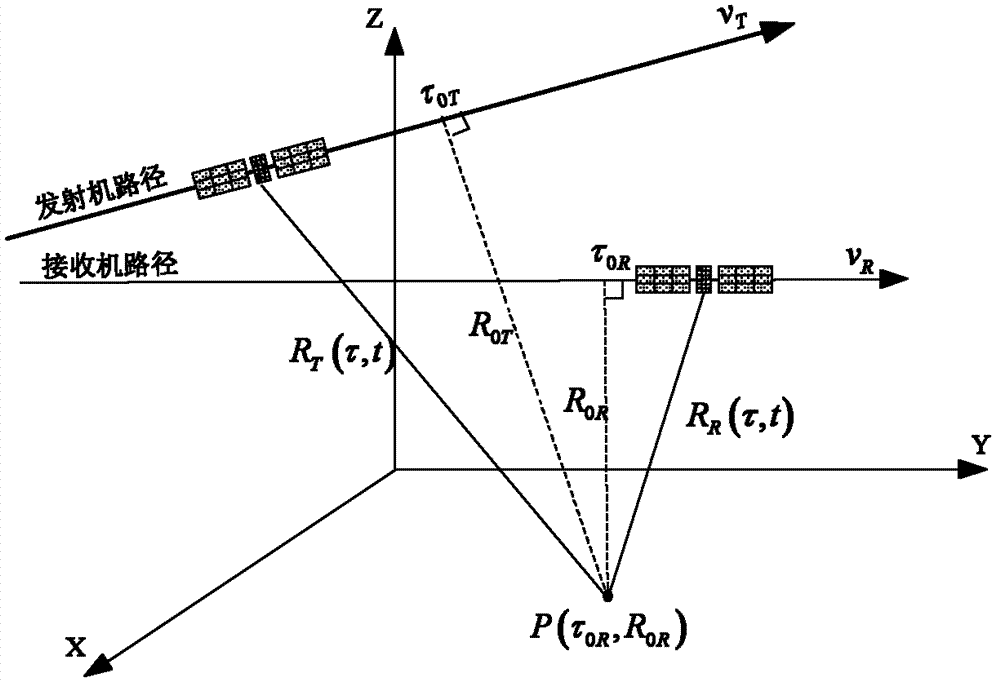
ActiveCN103245947ARealize the solutionRadio wave reradiation/reflectionSynthetic aperture radarFourier transform on finite groups
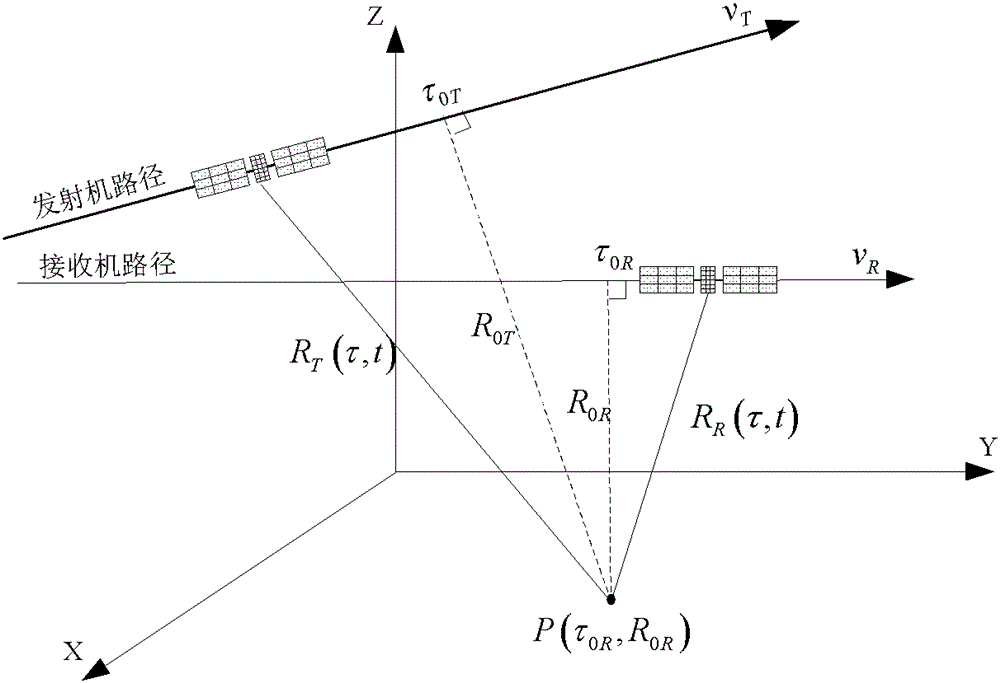
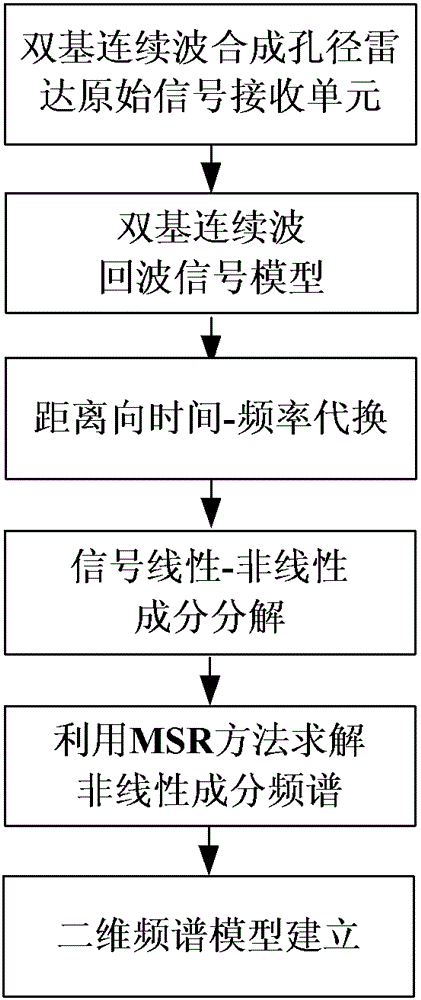
The invention discloses a Bi-CWSAR (Bistatic Continuous Wave Synthetic Aperture Radar) remote sensing imaging method. According to the Bi-CWSAR remote sensing imaging method, a sending and receiving antenna division mode is adopted for solving an antenna coupling problem, a Bi-CWSAR time domain echo signal model is established by accurately analyzing a signal process and solving echo delay time, a series inversion method and a Fourier transformation property are ingeniously used for establishing a two-dimensional analysis spectrum, and accordingly, foundations and supports are provided for a Bi-CWSAR remote sensing imaging technology.
Owner:INST OF ELECTRONICS CHINESE ACAD OF SCI
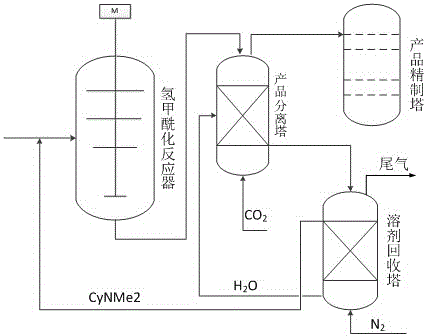
Polar-reversible transformation solvent for isobutylene hydroformylation reaction
ActiveCN104557488AEasy to separateSeparation increases the realizationPreparation by carbon monoxide reactionCarbonyl compound separation/purificationChemical industryChemical reaction
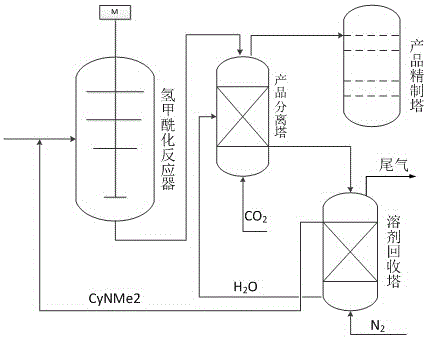
The invention belongs to the field of chemical industry, relates to solvents with different polarities used in different steps of the chemical reaction process, and in particular relates to a polar reversible transformation solvent for an isobutylene hydroformylation reaction. According to the reaction, N,N-dimethyl cyclohexyl amine (CyNMe2) serves as a solvent for generating isovaleraldehyde in the isobutylene hydroformylation reaction, and the polarity of the CyNMe2 is increased in a production separation tower under the actions of water and CO2 so as to realize separation of the product from the solvent; in a solvent recycling tower, CyNMe2 is dissociated from the aqueous phase by virtue of the gas stripping action of inert gases such as N2, and the solvent is recovered so as to be recycled. The traditional solvent and product separation process needs to be finished by distillation, and therefore the problems such as high energy consumption, solvent volatilization and loss and the like are caused. By utilizing the polar reversible transformation properties of the solvent under the normal temperature and normal pressure conditions, the aims of separating the solvent from the product and recycling the solvent are achieved, the energy consumption is reduced, and the solvent loss is avoided.
Owner:SOUTHWEST RES & DESIGN INST OF CHEM IND
Doxycycline temperature-sensitive in situ gel uterus perfusion fluid for sows and preparation method thereof
ActiveCN109568262ASpecial reverse thermogelling propertiesGood biocompatibilityAntibacterial agentsTetracycline active ingredientsDiseaseGynecology
The invention discloses doxycycline temperature-sensitive in situ gel uterus perfusion fluid for sows and a preparation method thereof. The doxycycline temperature-sensitive in situ gel uterus perfusion fluid is prepared from a main drug, a temperature-sensitive material, a temperature regulator, a metal complexing agent, an antioxidant, a pH (Potential of Hydrogen) regulator and ultrapure water.A preparation has a solution-gel transformation property and biological adhesion performance; compared with conventional sow uterus perfusion fluid, the preparation can be rapidly transformed into semi-solid gel from liquid at drug administration parts, the perfusion fluid can reach deeper parts of a uterus and can be tightly adhered to the surface of a uterine mucosa; the preparation can act on the drug administration parts for a long time, sudden release of a medicine is effectively prevented and uterus positioning and slow release effects can be realized; the clinical drug administration effect is improved and the clinical drug administration convenience for diseases is extremely improved. The preparation has the advantages that the preparation technology is simple and a product is stable and clarified, can be stored at room temperature, and has no toxicity, small irritation and good biocompatibility; the preparation has a good popularization and application prospect.
Owner:SOUTH CHINA AGRI UNIV
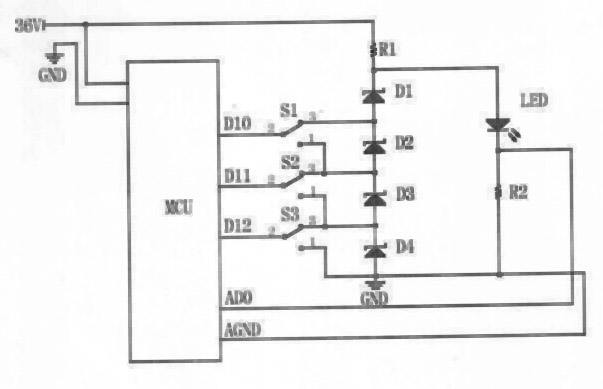
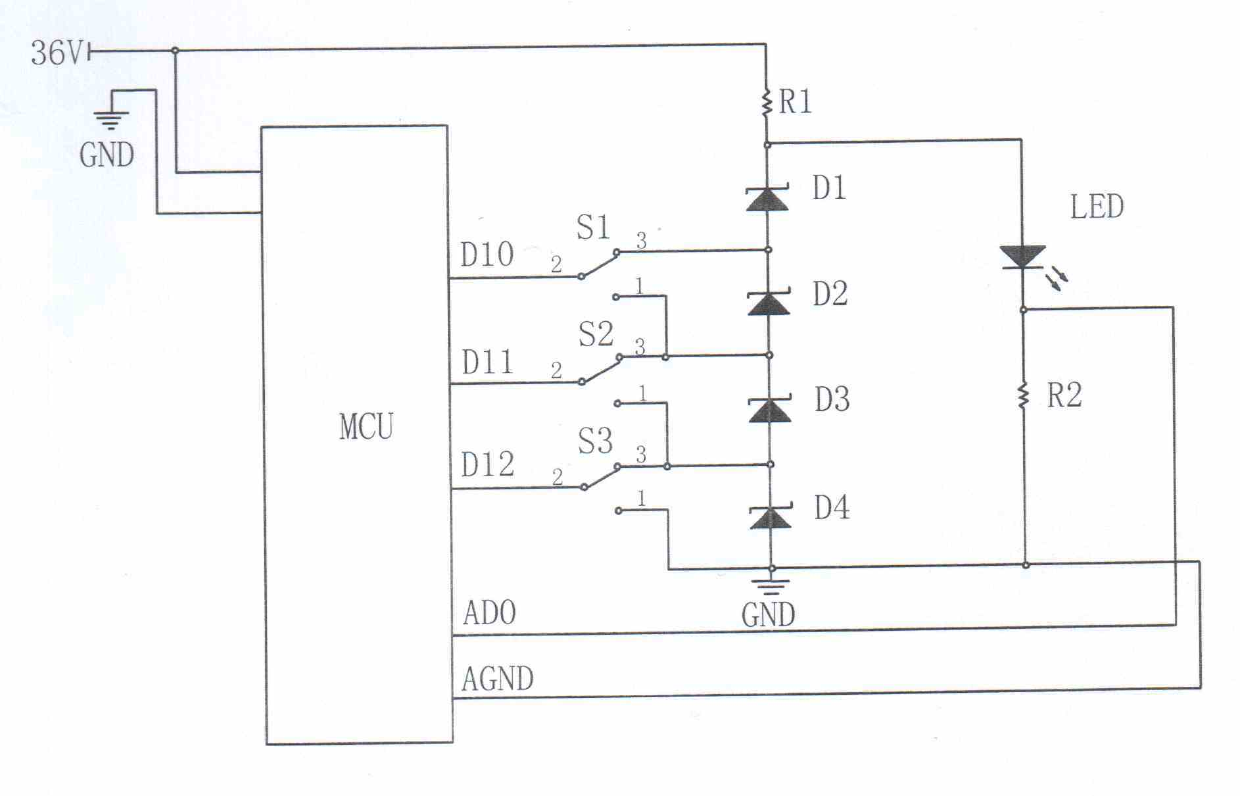
Solar LED lighting driving circuit
InactiveCN102685986AImprove efficiencySimple structureElectric light circuit arrangementCapacitanceElectronic switch
The invention relates to a solar LED (light-emitting diode) lighting driving circuit. Positive 36V direct current power is accessed into a micro-controller unit (MCU) and connected with a voltage-stabilizing tube via an electronic switch; and the positive 36V direct current power is connected with an LED through a current-limiting resistor R1, and the LED is grounded through a current-limiting resistor R2. According to the solar LED lighting driving circuit disclosed by the invention, on the premise of not requiring an inductor and a capacitor, the step-up and step-down voltage transformation property can be achieved, and the solar LED lighting driving circuit has the advantages of high efficiency, simple structure, low manufacturing cost, low calorific value, easiness in maintenance and long service life, and is suitable for being used on circuits of solar LED lighting devices.
Owner:SUIZHONG ZHONGWEI TECH
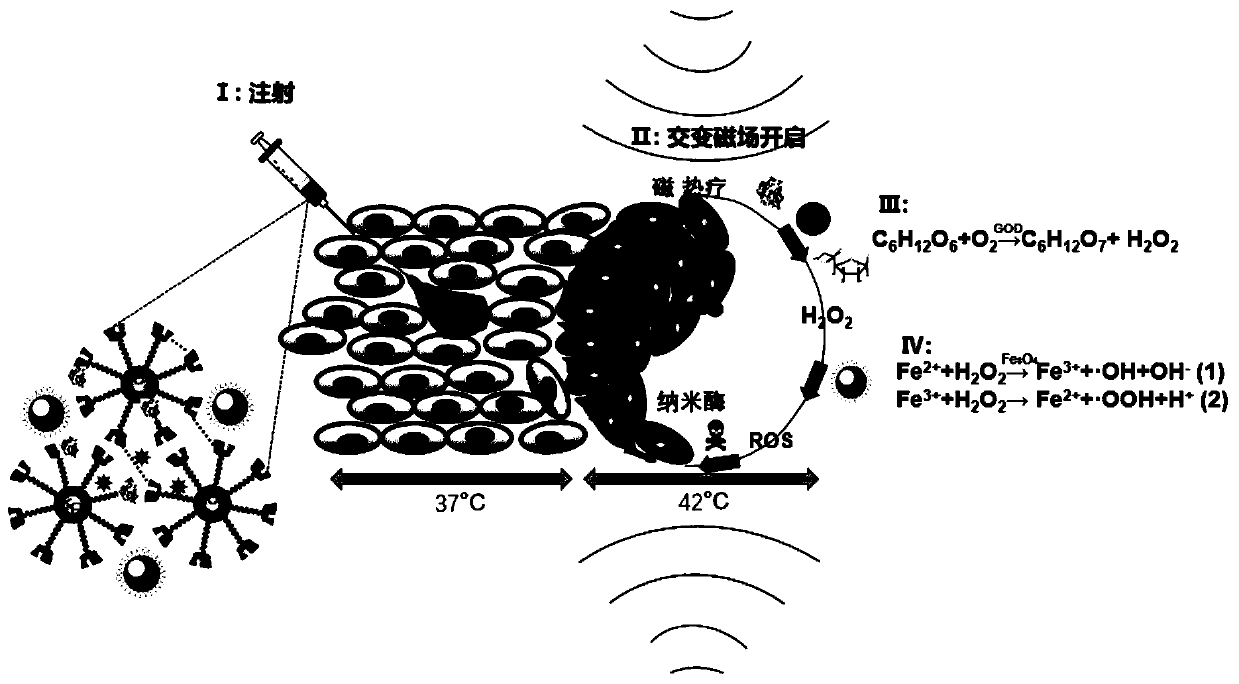
Magnetic heat triggered cascaded enzyme reaction supermolecular gel and preparing method and application thereof
ActiveCN110292641APromote liquefaction and dispersionSimple and fast operationHeavy metal active ingredientsOrganic active ingredientsPeroxidaseTumor cells
The invention discloses magnetic heat triggered cascaded enzyme reaction supermolecular gel and a preparing method and application thereof. The preparing method includes the steps of mixing an oxygen-rich PFOB nano-emulsion with glucose-loaded PLGA nano-capsules to form a solution A, adding alpha-cyclodextrin, glucose oxidase and mannitol to an Fe3O4@PEI solution to obtain a solution B, and mixingthe solution A with the solution B to obtain the magnetic heat triggered cascaded enzyme reaction supermolecular gel. The prepared gel has the shear thinning injection and temperature-sensitive reversible phase transformation properties, and the multi-stage structure of the gel can carry glucose, oxygen, glucose oxidase and mannitol; after the gel is injected into tumor tissue, ferroferric oxidenano-particles generate heat under the alternating magnetic field, the tumor tissue is heated, and the liquefying and dispersing of the gel are promoted, so tumor cell gaps are filled with the gel; meanwhile, the ferroferric oxide nano-particles as similar peroxidase participate in subsequent cascaded enzyme reaction to generate free radicals ROS, and the duplex functions of magnetic induction thermal therapy and nano-enzyme catalysis treatment are realized.
Owner:SOUTHEAST UNIV
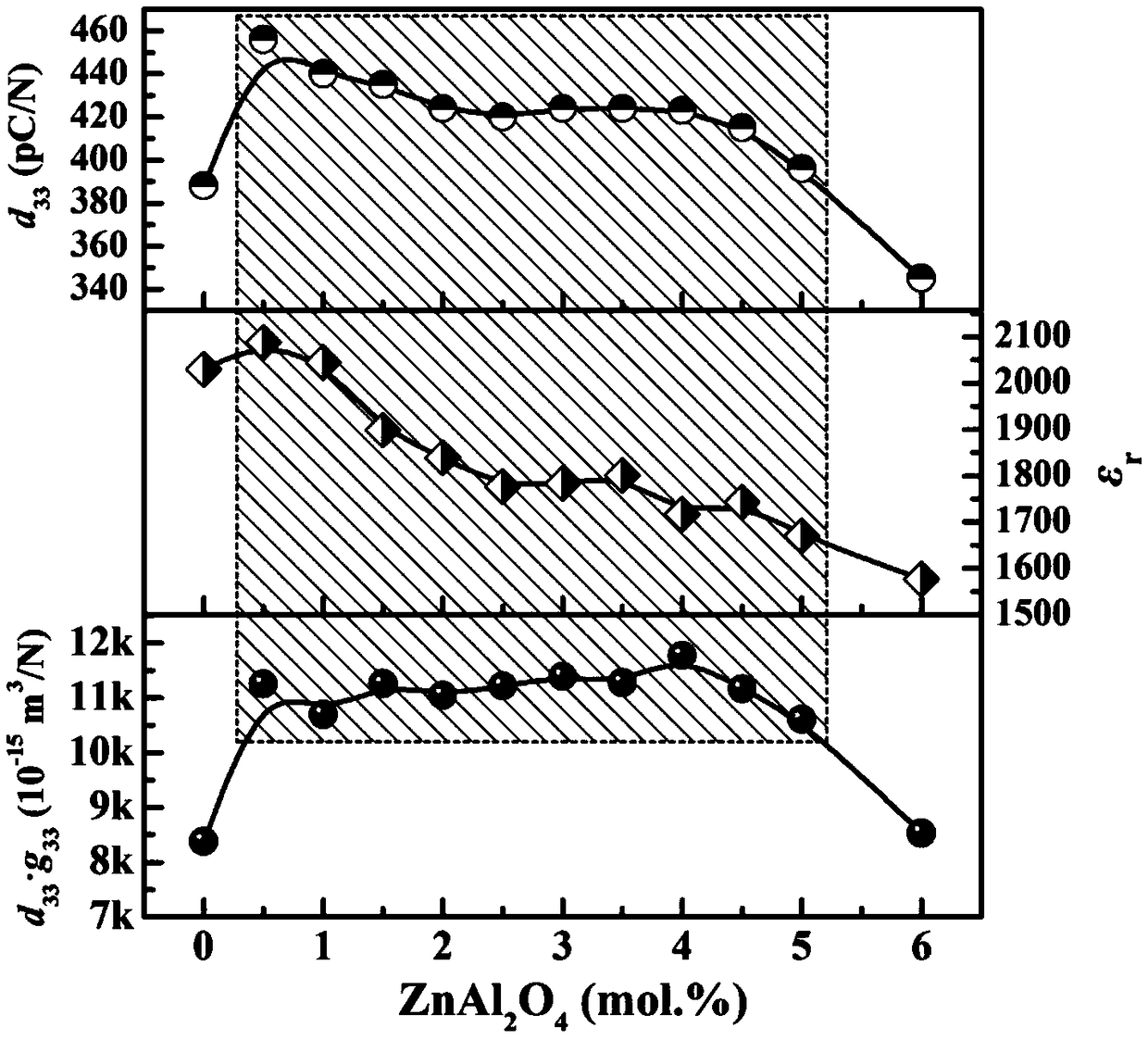
Wide component window and high transduction coefficient ceramic material for piezoelectric energy collection and preparation
ActiveCN108727021ASolve for piezoelectric charge constantSolving the problem of synergistic changes in relative permittivityElectricityRelative Change
The invention discloses a wide component window and high transduction coefficient ceramic material for piezoelectric energy collection and preparation and belongs to the field of piezoelectric ceramicmaterials. The material has a chemical formula of 0.2Pb(Zn1 / 2Nb2 / 3)O3-0.8Pb(Zr1 / 2Ti1 / 2)O3 (abbreviated as 0.2PZN-0.8PZT), AlN with a mole fraction x of mol.% is doped, and x is 0.00-6.00. The preparation comprises the following steps: preparing 0.2PZN-0.8PZT, further adding AlN, and carrying out steps of wet milling, drying, pelletizing, pressing molding and sintering. By adopting the material, relative change trend of piezoelectric charge constants and dielectric constants is effectively regulated and controlled, energy increase coefficients are increased, component windows of high transduction coefficients are greatly widened, dependency of optimal properties upon specific single components is relieved, and great promotion functions can be achieved for further improving electro-mechanical transformation properties and industrial application of piezoelectric energy collection devices.
Owner:昆山昱翌辉华电子科技有限公司
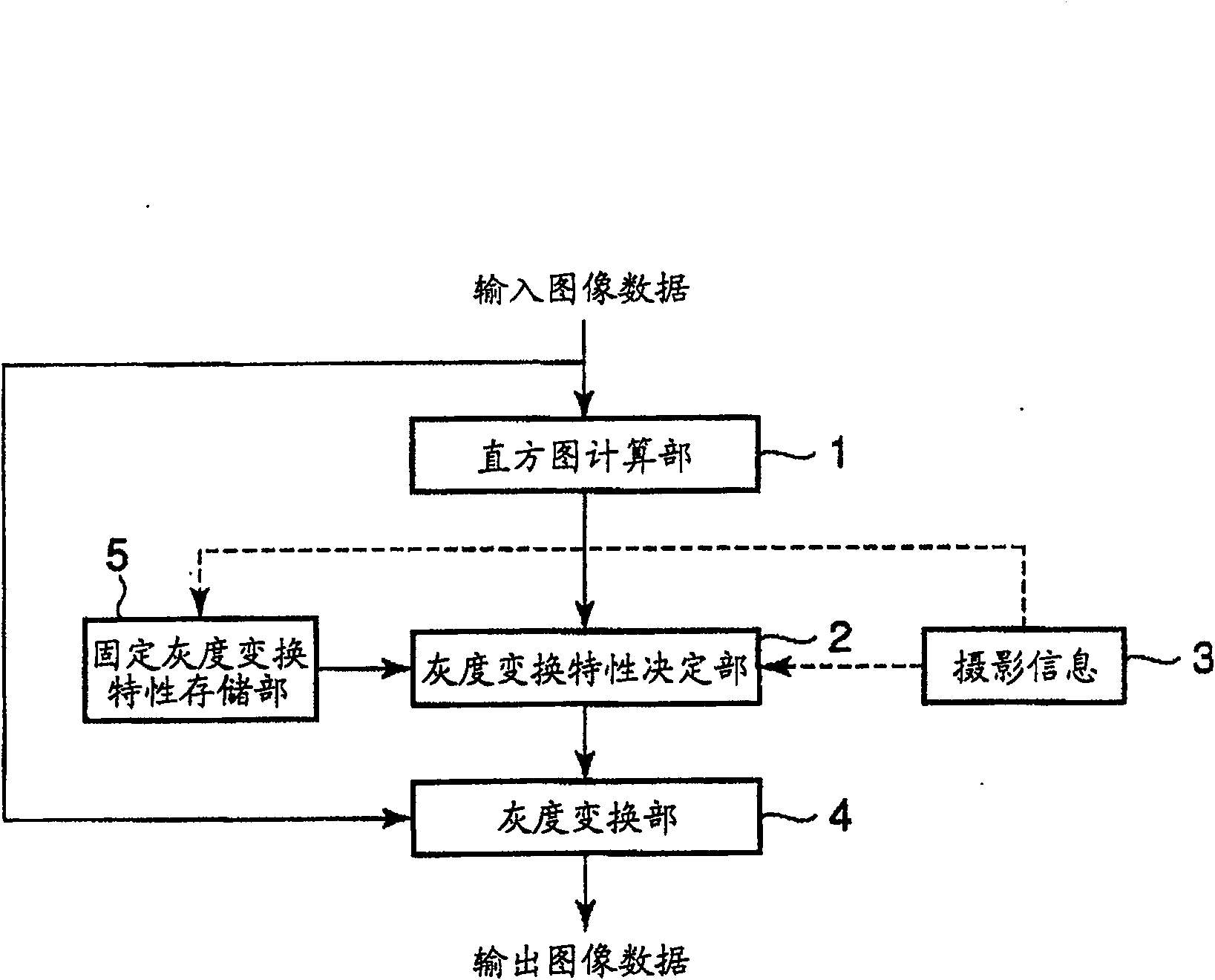
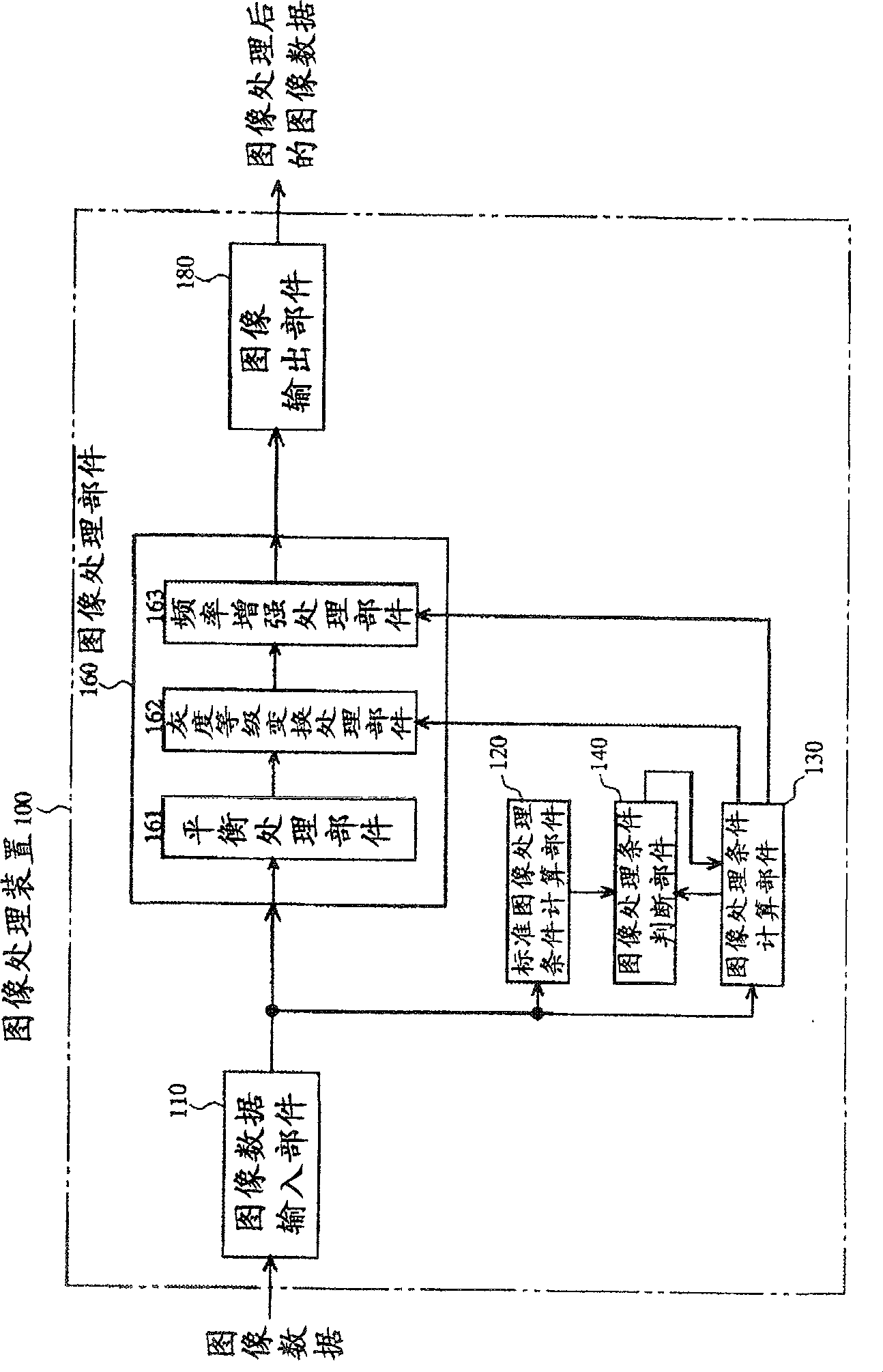
Image processing apparatus, image recording apparatus and image processing method
InactiveCN100563307CTelevision system detailsColor television detailsImaging processingComputer graphics (images)
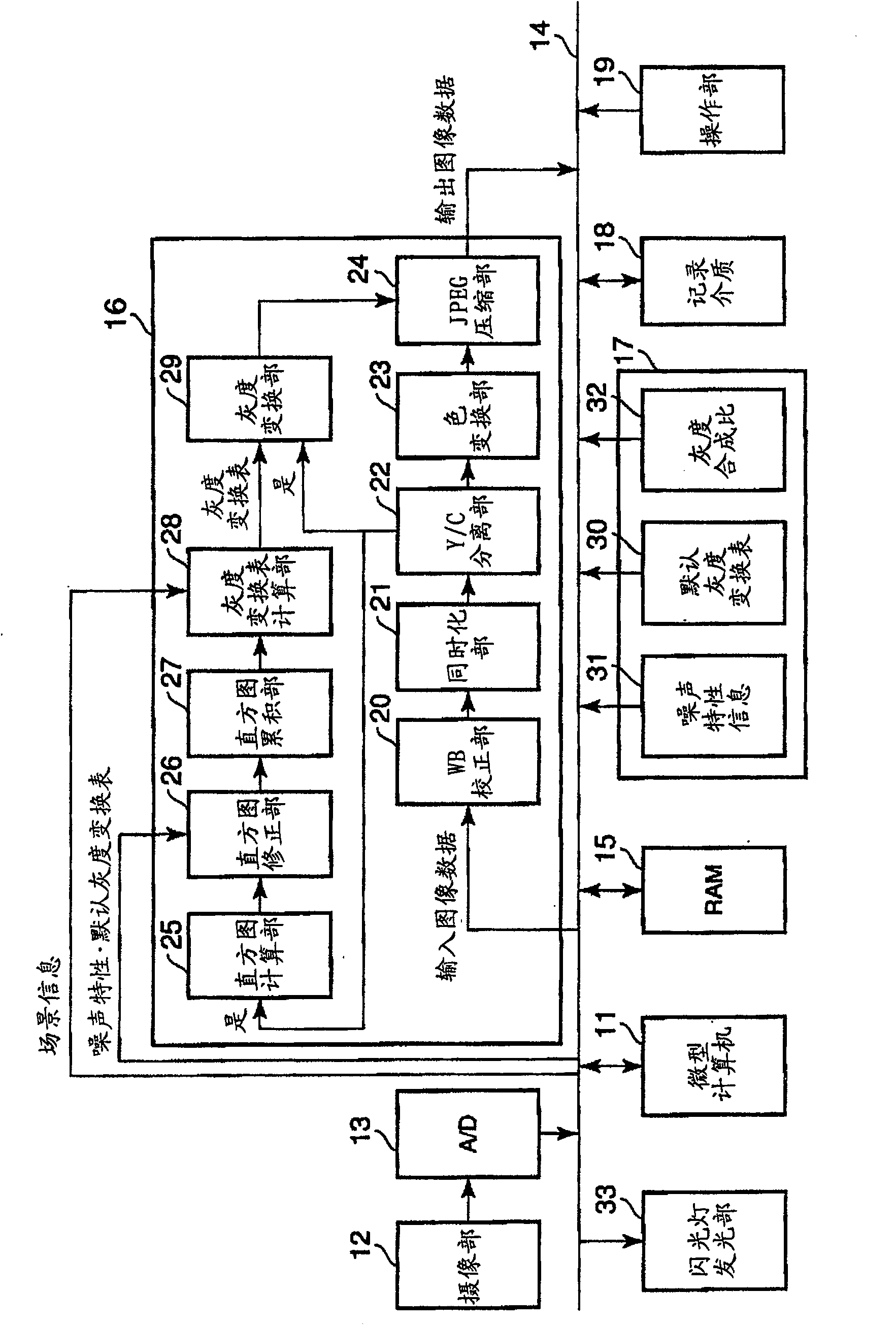
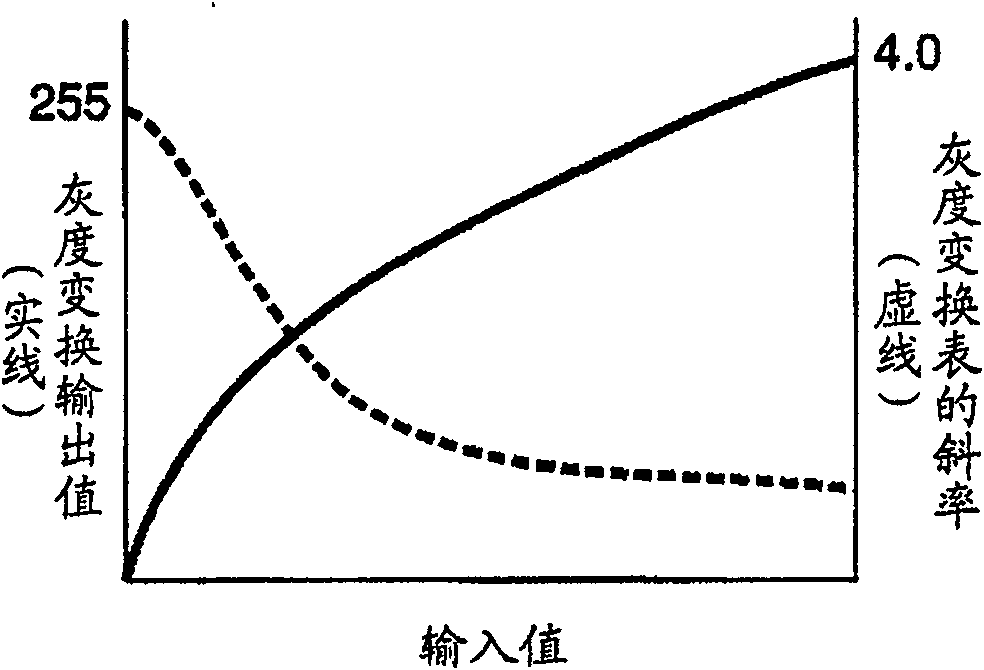
Image processing device, image recording device and image processing method. An object of the present invention is to provide an image processing device, an image recording device, and an image processing method capable of performing gradation conversion processing optimal for a scene at the time of shooting regardless of shooting conditions. A histogram of pixel values of the input image data is calculated in the histogram calculation unit (1), and the calculated histogram is input to the gradation transformation characteristic determination unit (2). In the gradation transformation characteristic determination unit (2), the histogram calculated in the histogram calculation unit (1) for photographic information synthesis and the fixed gradation stored in the fixed gradation transformation characteristic storage unit (5) are referred to. Transformation characteristics to determine the grayscale transformation characteristics. The gradation conversion of the image data is performed in a gradation conversion unit (4) based on the determined gradation conversion characteristics.
Owner:OLYMPUS CORP
Zanamivir nasal in situ jellies with phase variation property and preparing method thereof
InactiveCN101229122BEffective absorptionExtended stayOrganic active ingredientsAerosol deliveryNasal cavityWhole body
The invention relates to the new dosage form of zanamivir, which is a nasal in-situ gel with phase transformation property and preparation methods, the invention is made of the original drug of zanamivir, hydrophilic gel materials which are sensitive to environments and assistant materials which can be accepted in pharmacy, after being absorbed by nasal mucosa, the invention can have systemic functions and improve the partial concentration of drugs in respiratory tracts, when room temperature is about 20 DEG C and the invention is in storing state, the invention is in the state of liquid, while after being put into the physiological conditions of nasal cavities, the invention can be quickly formed into gel on the surface of nasal mucosa, so that the detained time of drugs can be prolonged,biological availability can be improved and the compliance of patients can be perfected, and the viscosity of the invention is proper, after being formed into gel, the invention can be detained on the surface of the nasal cavity for a rather long time, which has no nasal ciliary toxicity.
Owner:SHANGHAI INST OF MATERIA MEDICA CHINESE ACAD OF SCI
Mini-array poisonous gas detecting instrument
InactiveCN101726465BImprove sealingEasy loading and unloadingColor measuring devicesWithdrawing sample devicesGas detectorTransmitted light
The invention discloses a mini-array poisonous gas detecting instrument which can exhibit different colors when an air sensor reacts with poisonous gases, and the color of transmitted light can change correspondingly because incident light is spectrally absorbed. The mini-array poisonous gas detecting instrument comprises an incident light source system, an air sensing array system, a transmittedlight acquisition system and a data analyzing and processing system, wherein the air sensing array system is provided with a sealed cabin which is characterized by good sealing effect, convenient loading and unloading, high stability and reasonable structural design, the transmitted light acquisition system uses a colored linear array CCD which is characterized by good sensibility and photoelectric transformation property, the data analyzing and processing system takes a microprocessor as a core element to form a perfect system of element controlling, data receiving, processing and analyzing,and is characterized by high automation degree, quick analysis speed and precise result. The mini-array poisonous gas detecting instrument is compact in structure and small in size, is especially beneficial to quickly detecting the slight quantity of extremely poisonous gas, and has good developing prospect.
Owner:CHONGQING UNIV
Image processing apparatus and image processing method
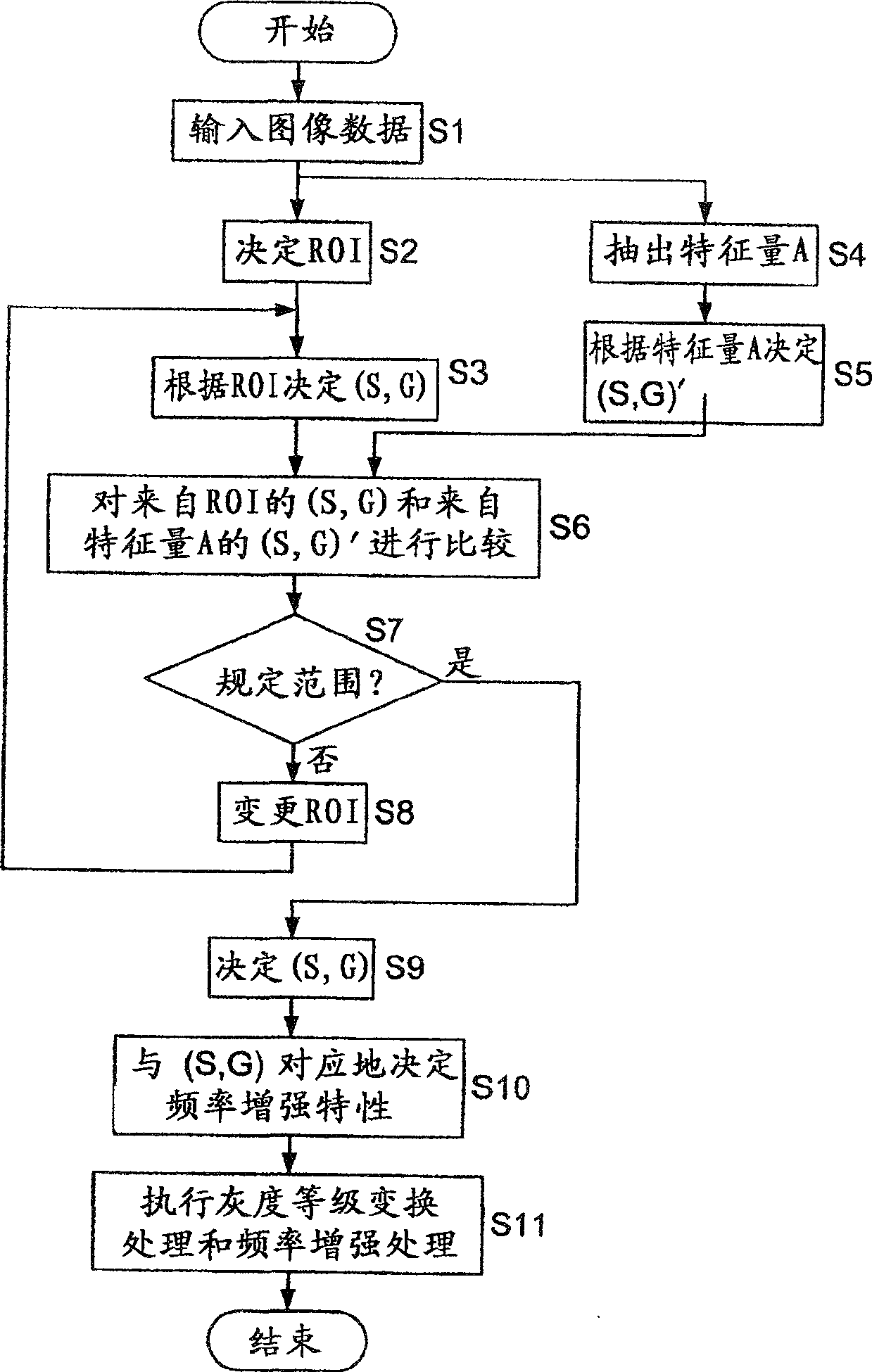
InactiveCN100502782CFull contrastSuppressing fluctuations in gray scaleImage enhancementImage analysisImaging processingFrequency characteristic
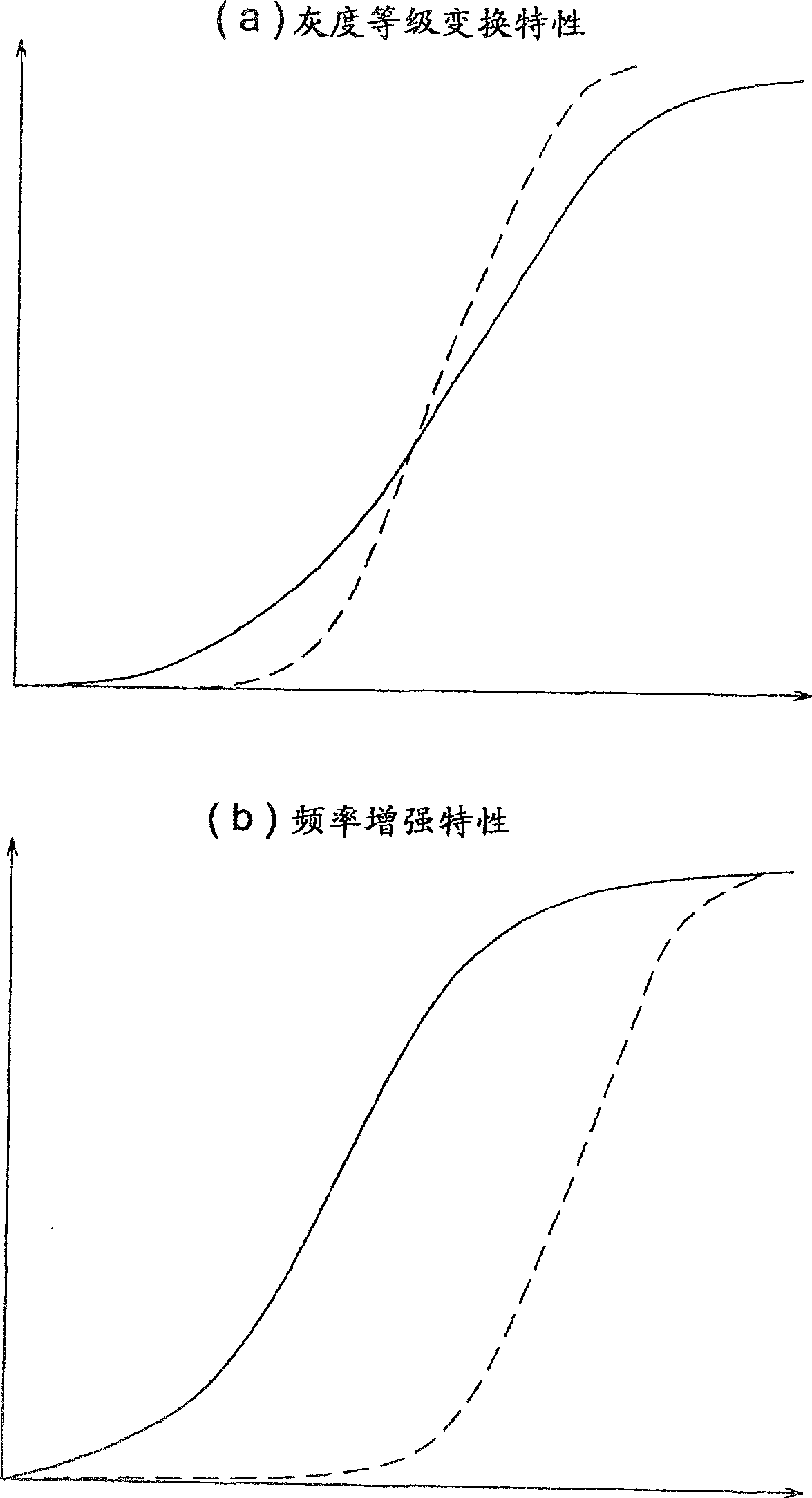
The present invention realizes an image processing apparatus capable of performing appropriate image processing in a state of high versatility regardless of the part of the subject or the setting of the operator. Equipped with: grayscale conversion processing means for performing grayscale conversion processing on image data obtained by photographing a subject based on the grayscale conversion characteristics having a predetermined offset (S) and a predetermined slope (G) (162); For the image data that has implemented the above-mentioned grayscale conversion process, according to the frequency enhancement characteristics as the characteristics of the enhancement degree of each frequency, implement the frequency enhancement processing part (163) of the frequency enhancement process; in determining the above-mentioned grayscale Image processing condition calculation means (130) for calculating the above-mentioned frequency enhancement characteristic from the grayscale transformation characteristic while calculating the grayscale transformation characteristic in a state in which the slope (G) is reduced The above-mentioned frequency enhancement characteristics are calculated from the gradation transformation characteristics to enable execution of frequency enhancement processing in which enhancement is performed from a low-frequency region.
Owner:KONICA MINOLTA MEDICAL & GRAPHICS INC
Method for improving digital signal conversion performance, digital signal conversion method and device
ActiveCN104244010BImprove transform performanceDigital video signal modificationDigital signal processingComputer science
The present invention proposes a method and device for improving digital signal conversion performance and a digital signal conversion method and device, wherein the method for improving digital signal conversion performance includes: constructing a transformation kernel matrix A with N rows and N columns, and making the transformation kernel matrix A satisfies two conditions (a) and (b) at the same time: (a)[DCT(i,j)×Factor]‑2≤A(i,j)≤[DCT(i,j)×Factor]+2( b) J(A)<TH; where, J ( A ) = α × dist ( A ) + β × the norm ( A ) + gamma × north ( A ) α + β + gamma The invention can ensure higher conversion performance for digital signal processing.
Owner:ZHEJIANG UNIV +2
Method for Eliminating Blocking Effect of Video Coding Image
ActiveCN105763880BQuick calculationEliminate blockingDigital video signal modificationSingular value decompositionPattern recognition
Owner:PEKING UNIV
A Reversible Polarity Switching Solvent for Isobutene Hydroformylation
ActiveCN104557488BEasy to separateSeparation increases the realizationPreparation by carbon monoxide reactionCarbonyl compound separation/purificationChemical industryChemical reaction
The invention belongs to the field of chemical industry, relates to solvents with different polarities used in different steps of the chemical reaction process, and in particular relates to a polar reversible transformation solvent for an isobutylene hydroformylation reaction. According to the reaction, N,N-dimethyl cyclohexyl amine (CyNMe2) serves as a solvent for generating isovaleraldehyde in the isobutylene hydroformylation reaction, and the polarity of the CyNMe2 is increased in a production separation tower under the actions of water and CO2 so as to realize separation of the product from the solvent; in a solvent recycling tower, CyNMe2 is dissociated from the aqueous phase by virtue of the gas stripping action of inert gases such as N2, and the solvent is recovered so as to be recycled. The traditional solvent and product separation process needs to be finished by distillation, and therefore the problems such as high energy consumption, solvent volatilization and loss and the like are caused. By utilizing the polar reversible transformation properties of the solvent under the normal temperature and normal pressure conditions, the aims of separating the solvent from the product and recycling the solvent are achieved, the energy consumption is reduced, and the solvent loss is avoided.
Owner:SOUTHWEST RES & DESIGN INST OF CHEM IND
A Bistatic Continuous Wave Synthetic Aperture Radar Remote Sensing Imaging Method
ActiveCN103245947BRealize the solutionRadio wave reradiation/reflectionSynthetic aperture radarFourier transform on finite groups
The invention discloses a Bi-CWSAR (Bistatic Continuous Wave Synthetic Aperture Radar) remote sensing imaging method. According to the Bi-CWSAR remote sensing imaging method, a sending and receiving antenna division mode is adopted for solving an antenna coupling problem, a Bi-CWSAR time domain echo signal model is established by accurately analyzing a signal process and solving echo delay time, a series inversion method and a Fourier transformation property are ingeniously used for establishing a two-dimensional analysis spectrum, and accordingly, foundations and supports are provided for a Bi-CWSAR remote sensing imaging technology.
Owner:INST OF ELECTRONICS CHINESE ACAD OF SCI
A Fast Difference Vector Quantization Compression Coding Method for Hyperspectral Signals
ActiveCN102905137BFlexible data handlingFlexible transmissionDigital video signal modificationSignal compressionCompression ratio
Owner:CHONGQING UNIV OF POSTS & TELECOMM