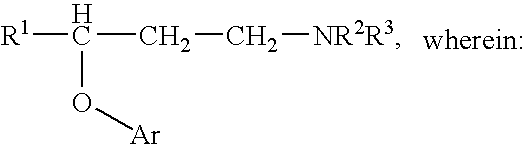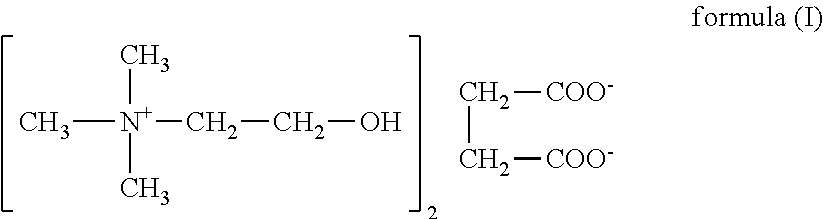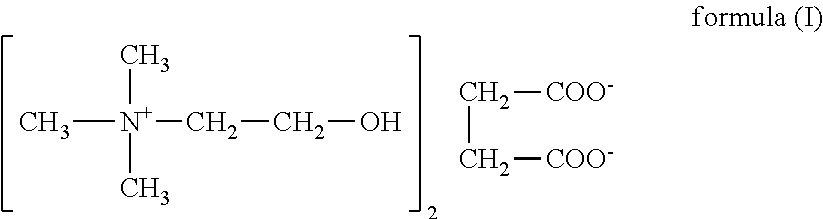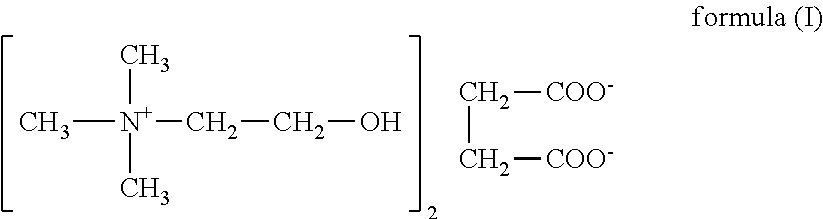Patents
Literature
127 results about "Isotopologue" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Isotopologues are molecules that differ only in their isotopic composition. They have the same chemical formula and bonding arrangement of atoms, but at least one atom has a different number of neutrons than the parent.
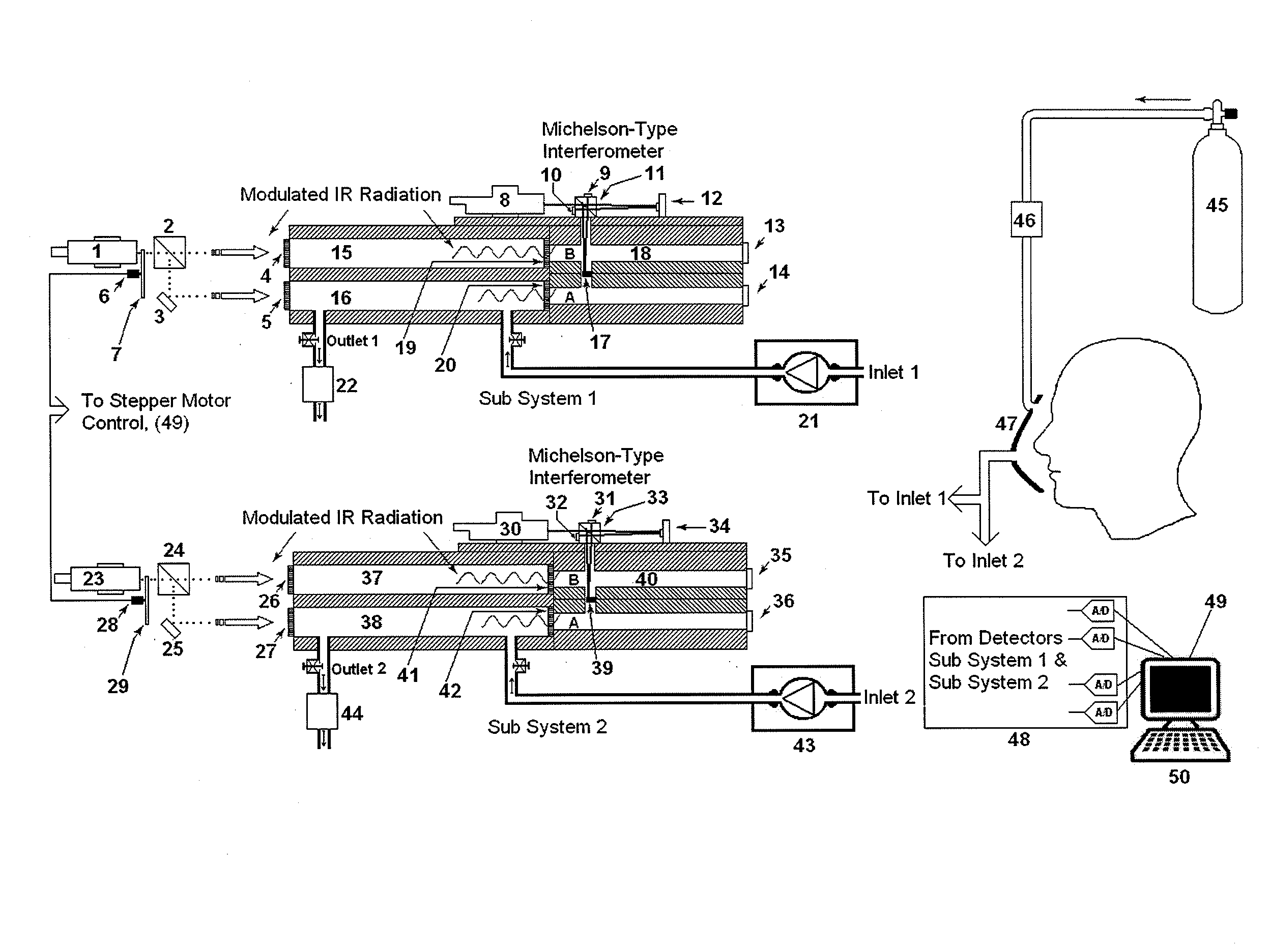
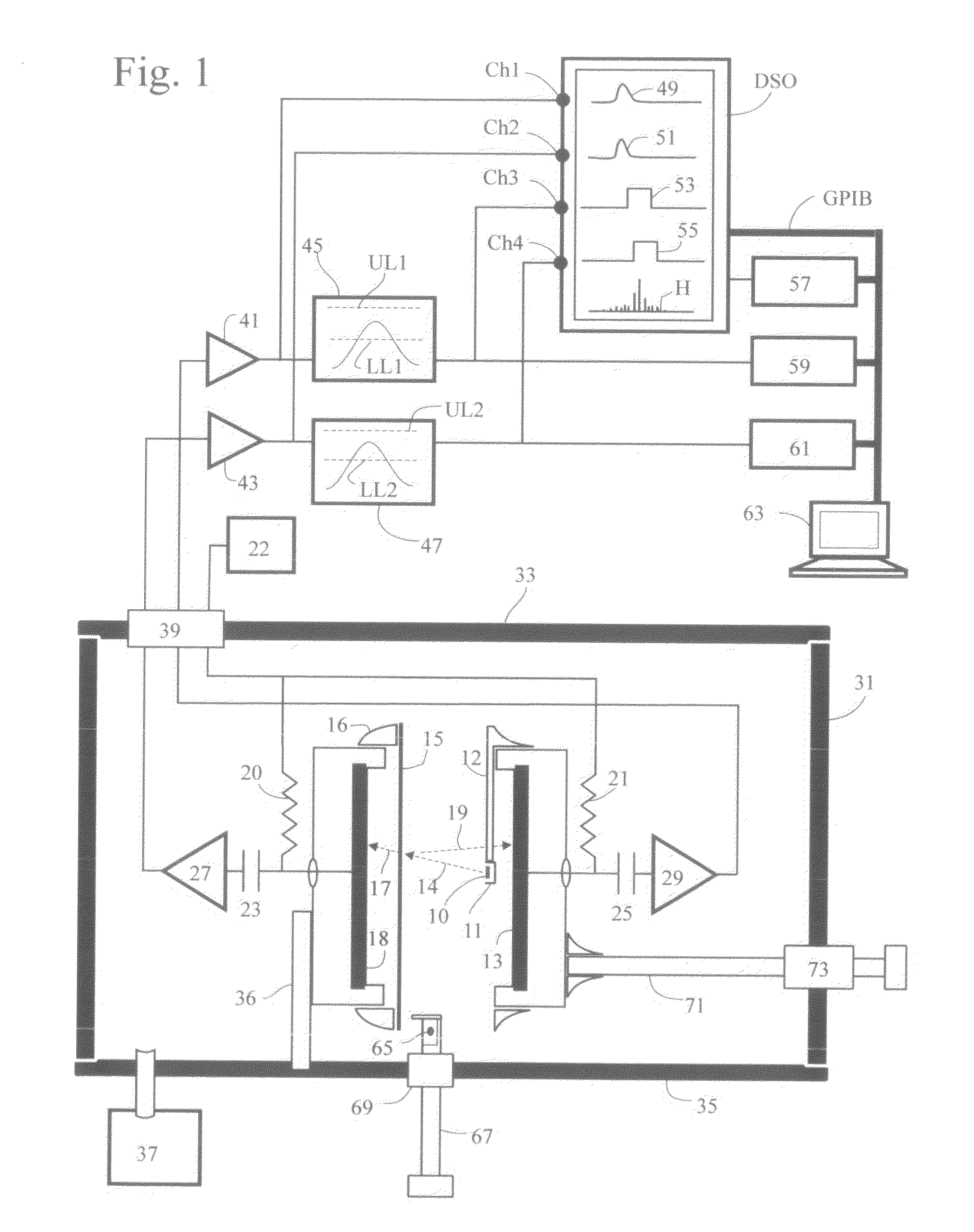
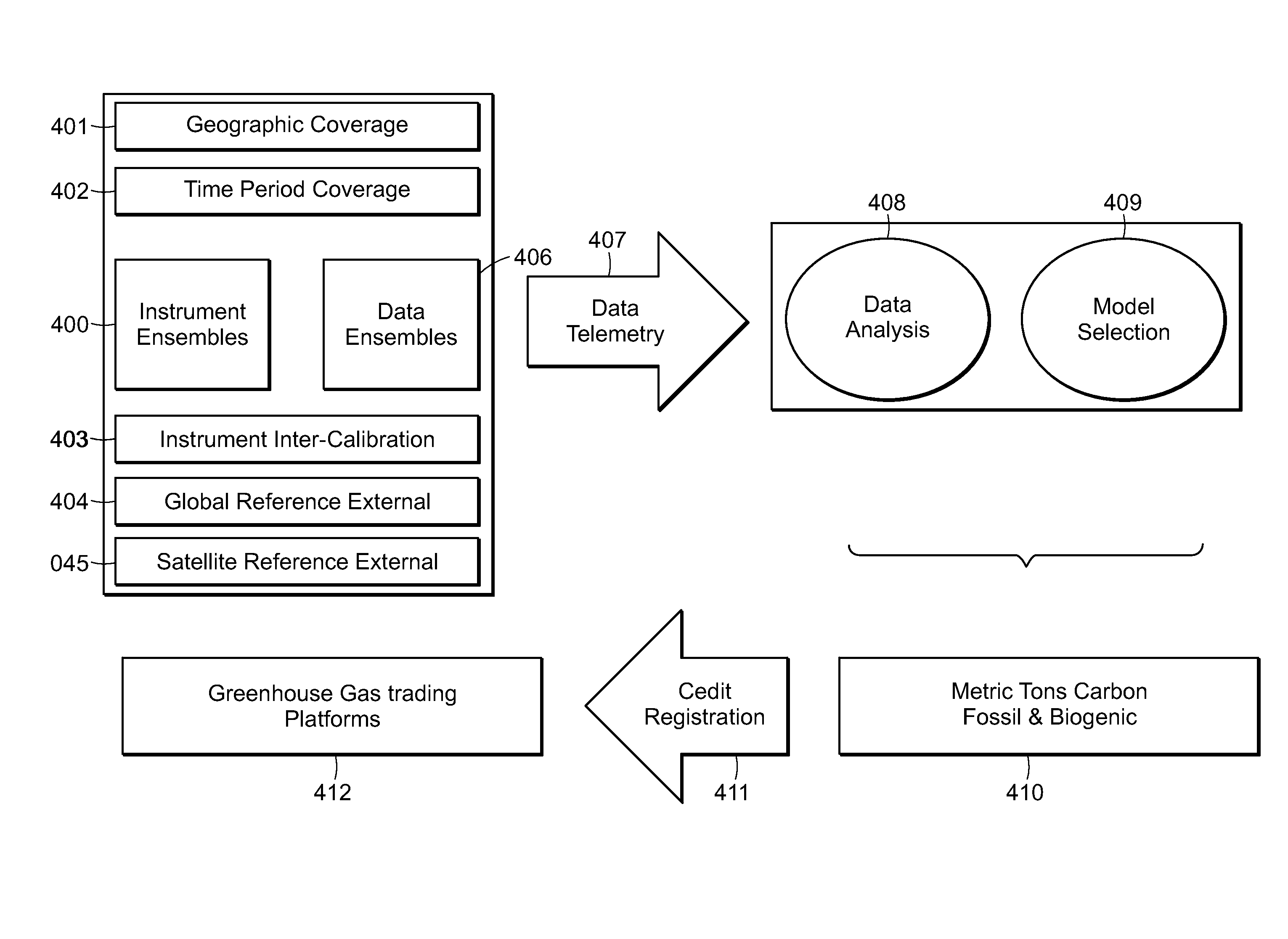
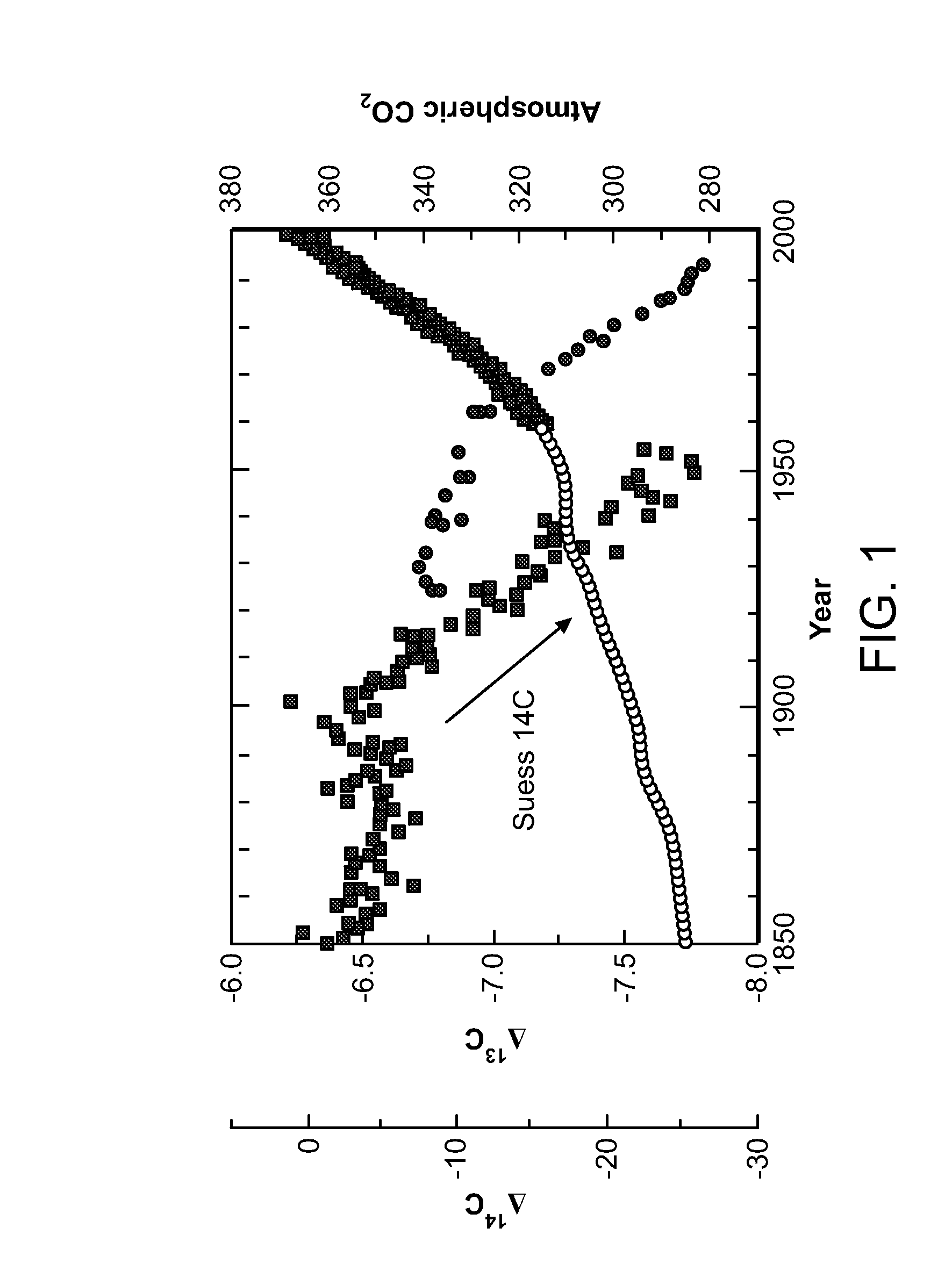
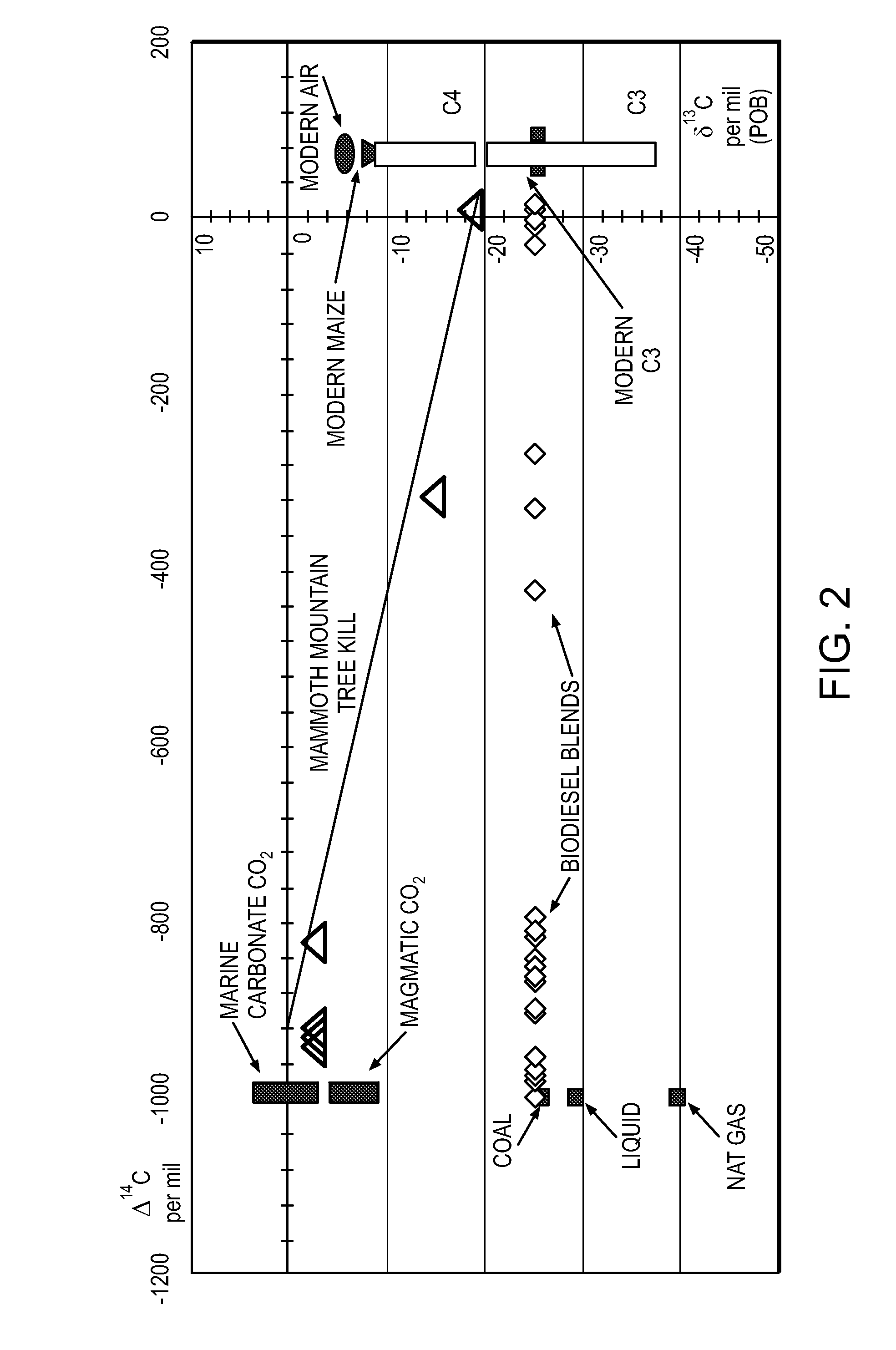
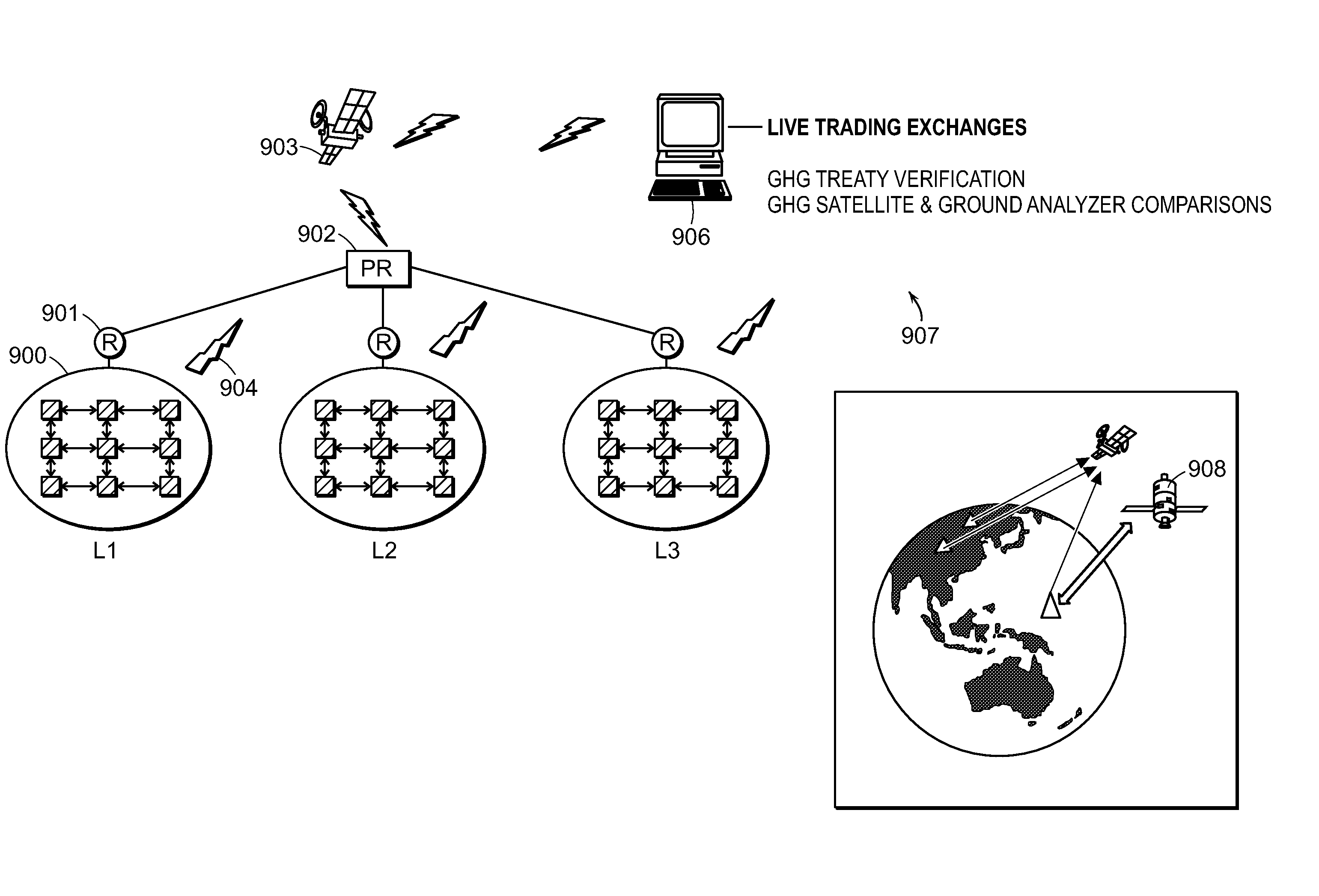
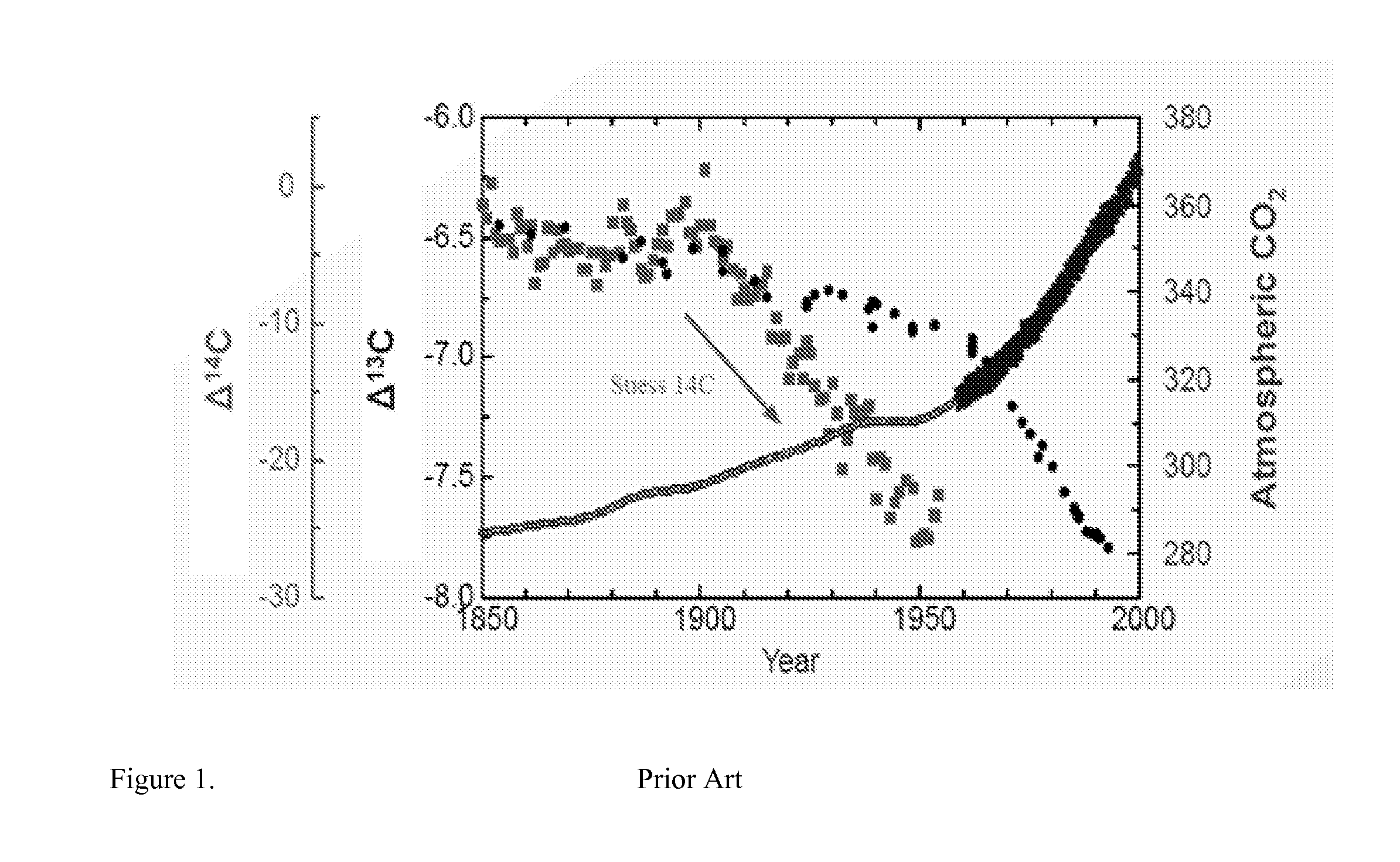
System of systems for monitoring greenhouse gas fluxes
ActiveUS20100198736A1High precisionReduce uncertaintySustainable waste treatmentCarbon compoundsNatural sourceGreenhouse gas flux
A system of systems to monitor data for carbon flux, for example, at scales capable of managing regional net carbon flux and pricing carbon financial instruments is disclosed. The system of systems can monitor carbon flux in forests, soils, agricultural areas, body of waters, flue gases, and the like. The system includes a means to identify and quantify sources of carbon based on simultaneous measurement of isotopologues of carbon dioxide, for example, industrial, agricultural or natural sources, offering integration of same in time and space. Carbon standards are employed at multiple scales to ensure harmonization of data and carbon financial instruments.
Owner:PLANETARY EMISSIONS MANAGEMENT
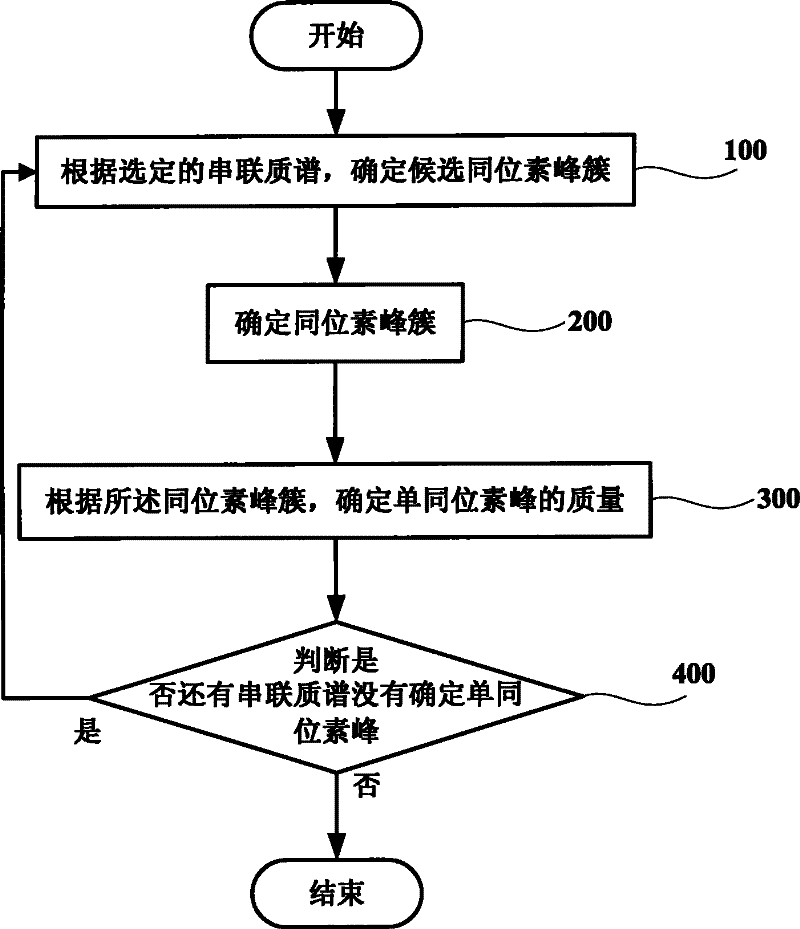
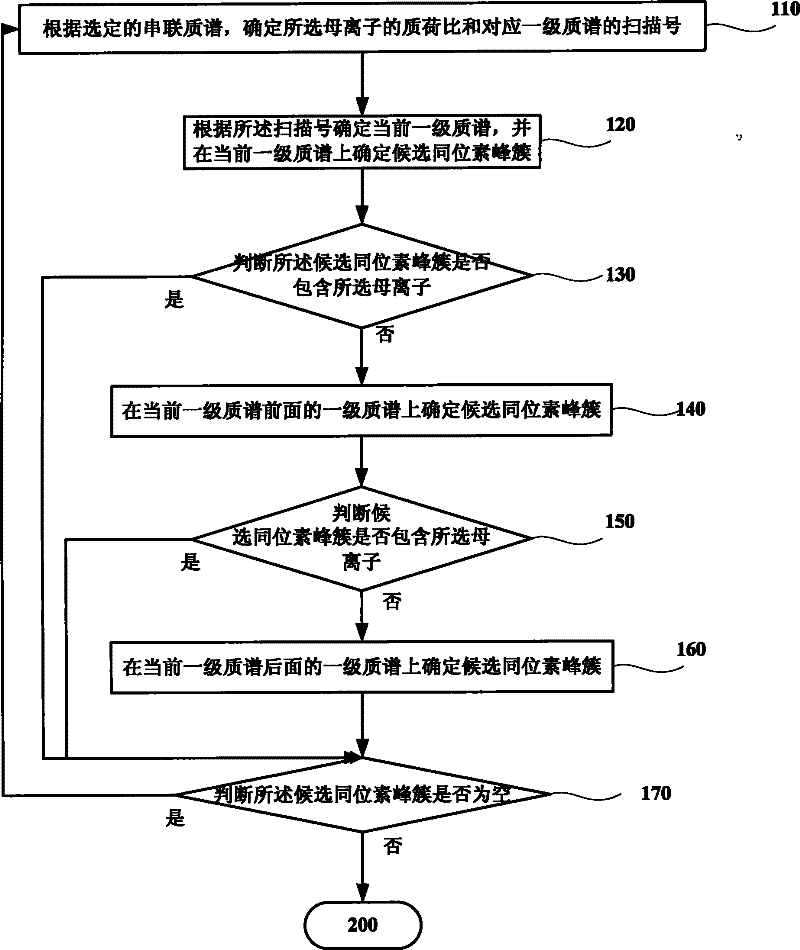
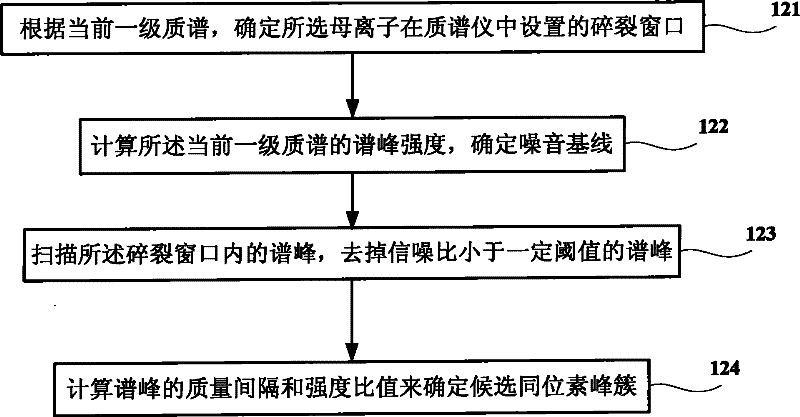
Method and system for increasing judgment accuracy of monoisotopic peaks
ActiveCN102445544AReduce mismatchSolve the problem of inaccurate intensity distributionMaterial analysis by electric/magnetic meansBiological testingComputer scienceTandem mass spectrometry
The invention discloses a method and system for increasing judgment accuracy of monoisotopic peaks. The method comprises the following steps: according to selected tandem mass spectrums, determining candidate isotopic peak clusters; according to the similarity and the strength ratio of the chromatographic outflow curves of the candidate isotopic peak clusters, determining the start and stop of the isotopic peak clusters; according to the start and stop of the isotopic peak clusters, determining the quality of the monoisotopic peaks; and judging whether the monoisotopic peaks of the tandem mass spectrums are not determined or not, if yes, returning and selecting a new tandem mass spectrum to determine a candidate isotopic peak cluster, and if not, ending.
Owner:INST OF COMPUTING TECH CHINESE ACAD OF SCI
System of systems for monitoring greenhouse gas fluxes
ActiveUS8595020B2Provide real-timeEasy to understandPigmenting treatmentSustainable waste treatmentNatural sourceGreenhouse gas flux
A system of systems to monitor data for carbon flux, for example, at scales capable of managing regional net carbon flux and pricing carbon financial instruments is disclosed. The system of systems can monitor carbon flux in forests, soils, agricultural areas, body of waters, flue gases, and the like. The system includes a means to identify and quantify sources of carbon based on simultaneous measurement of isotopologues of carbon dioxide, for example, industrial, agricultural or natural sources, offering integration of same in time and space. Carbon standards are employed at multiple scales to ensure harmonization of data and carbon financial instruments.
Owner:PLANETARY EMISSIONS MANAGEMENT
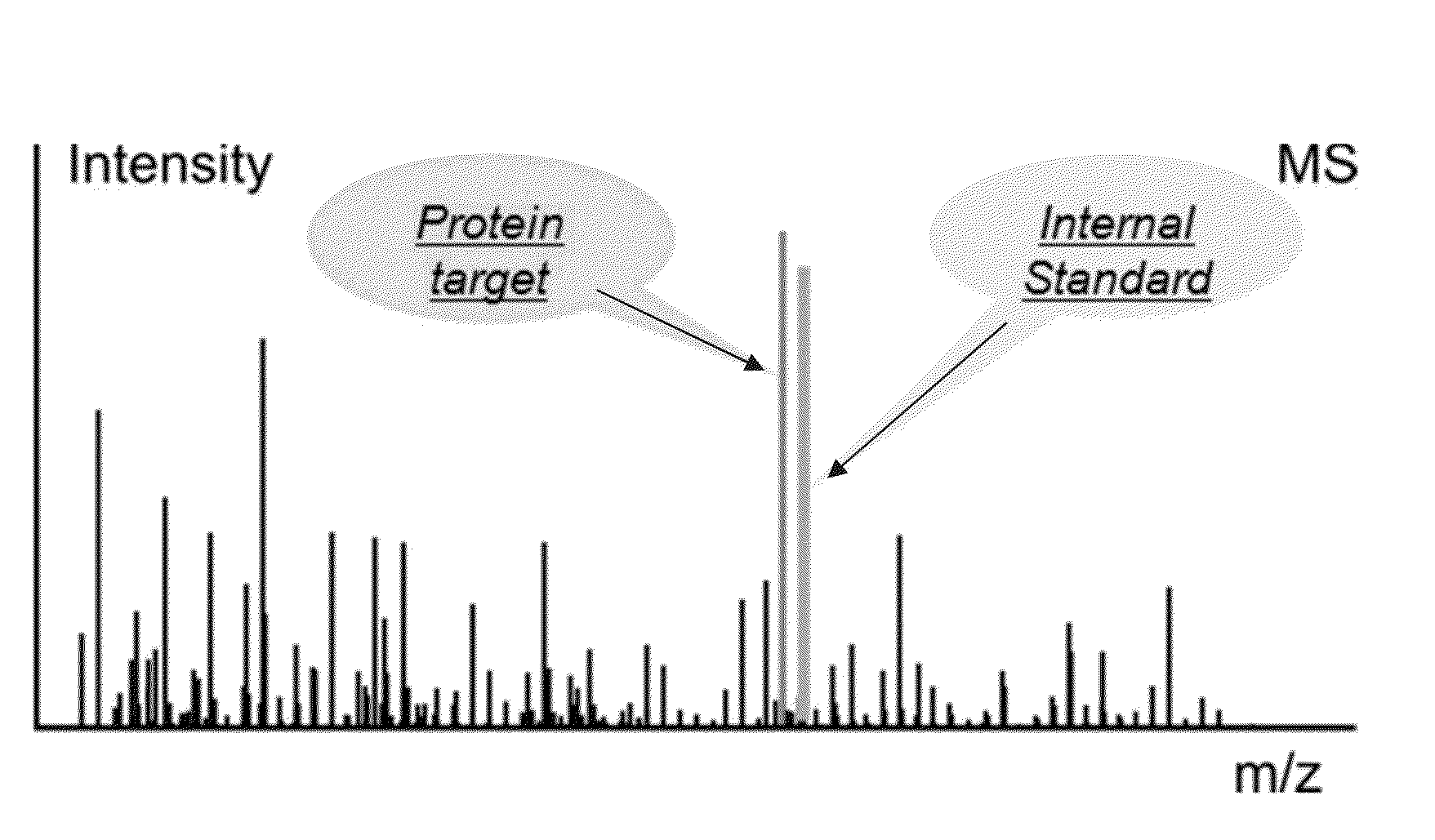
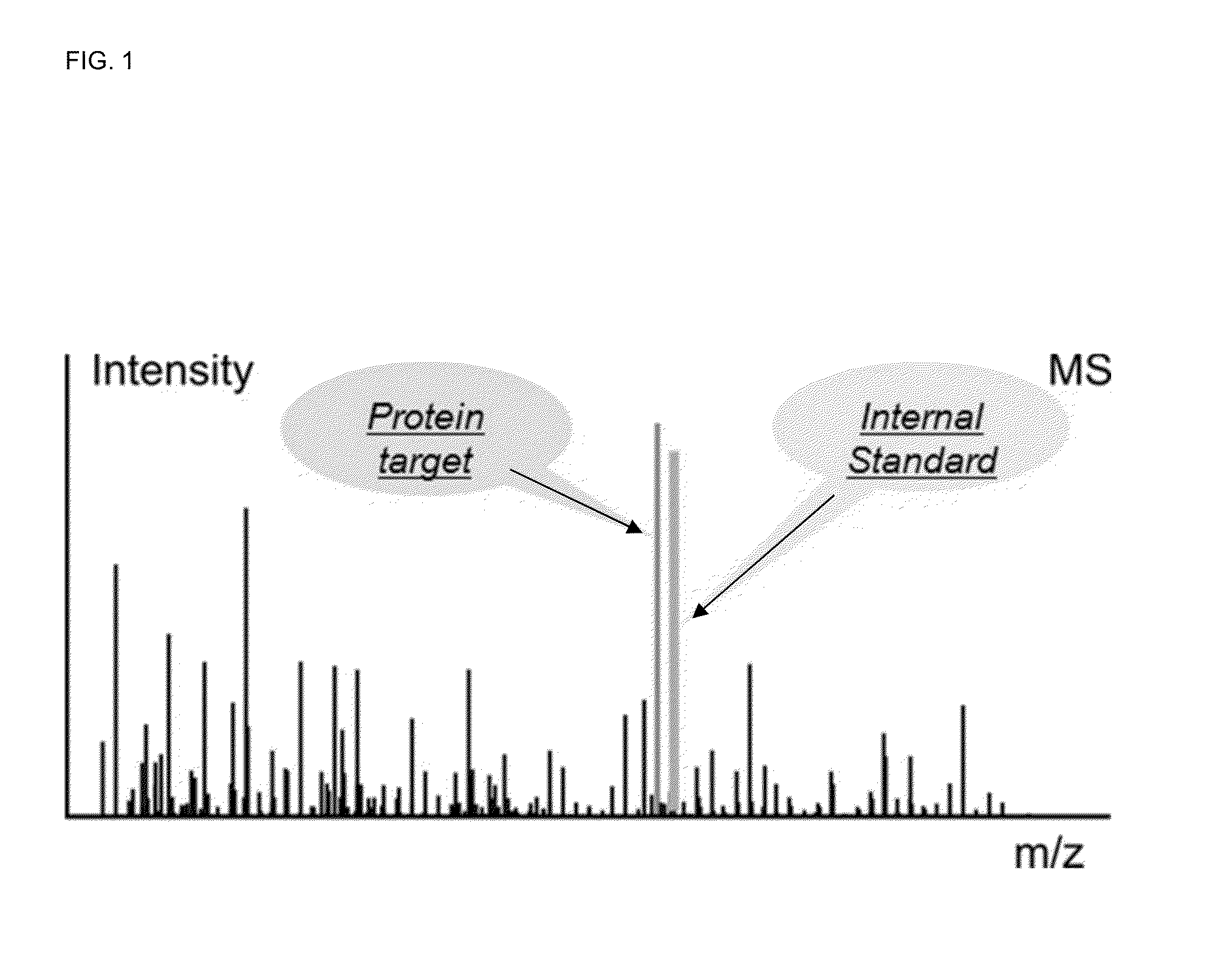
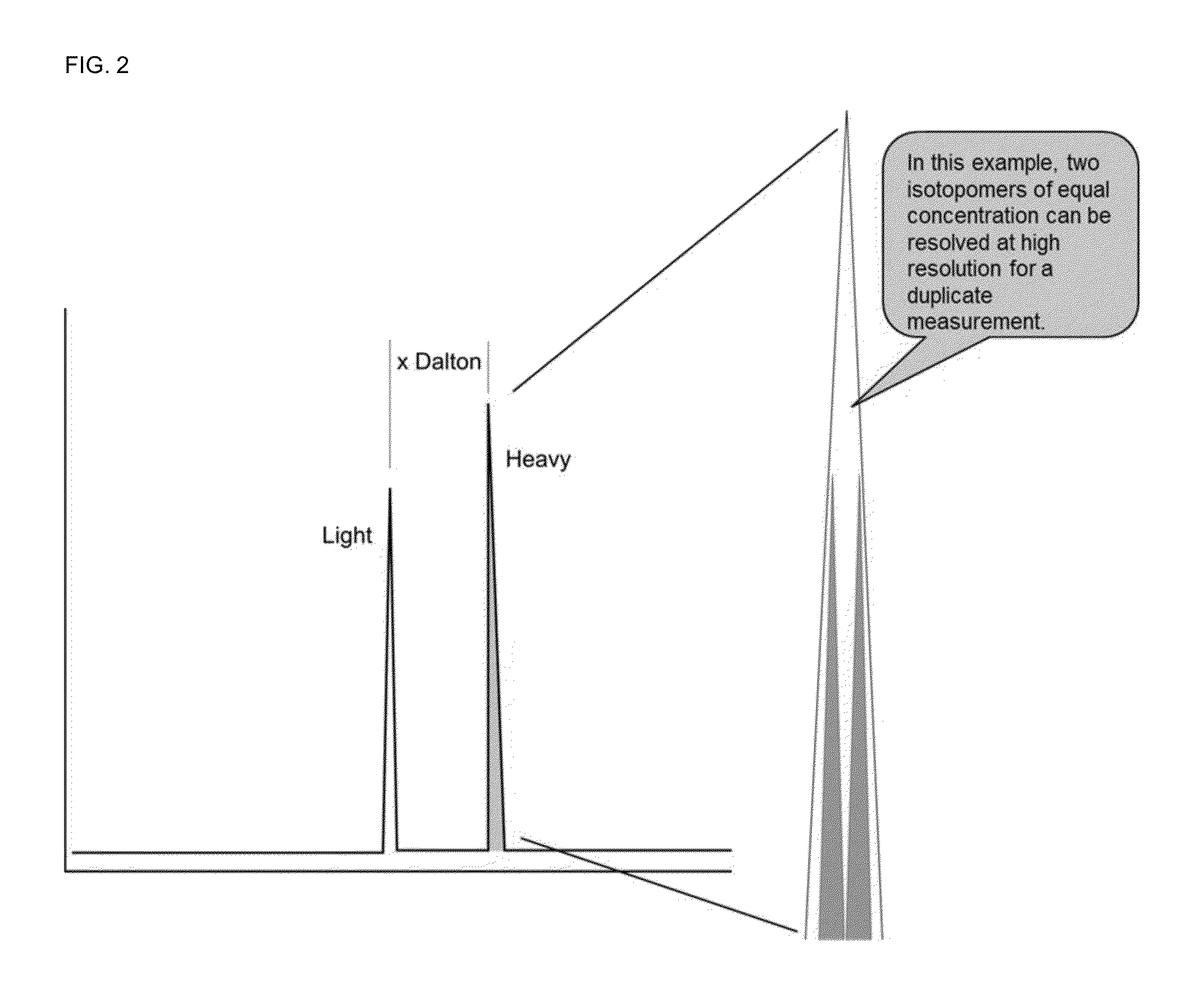
Absolute quantitation of proteins and protein modifications by mass spectrometry with multiplexed internal standards
ActiveUS20140364337A1Improve accuracyHigh sensitivityPeptide librariesLibrary screeningSpectroscopyMass spectrometry imaging
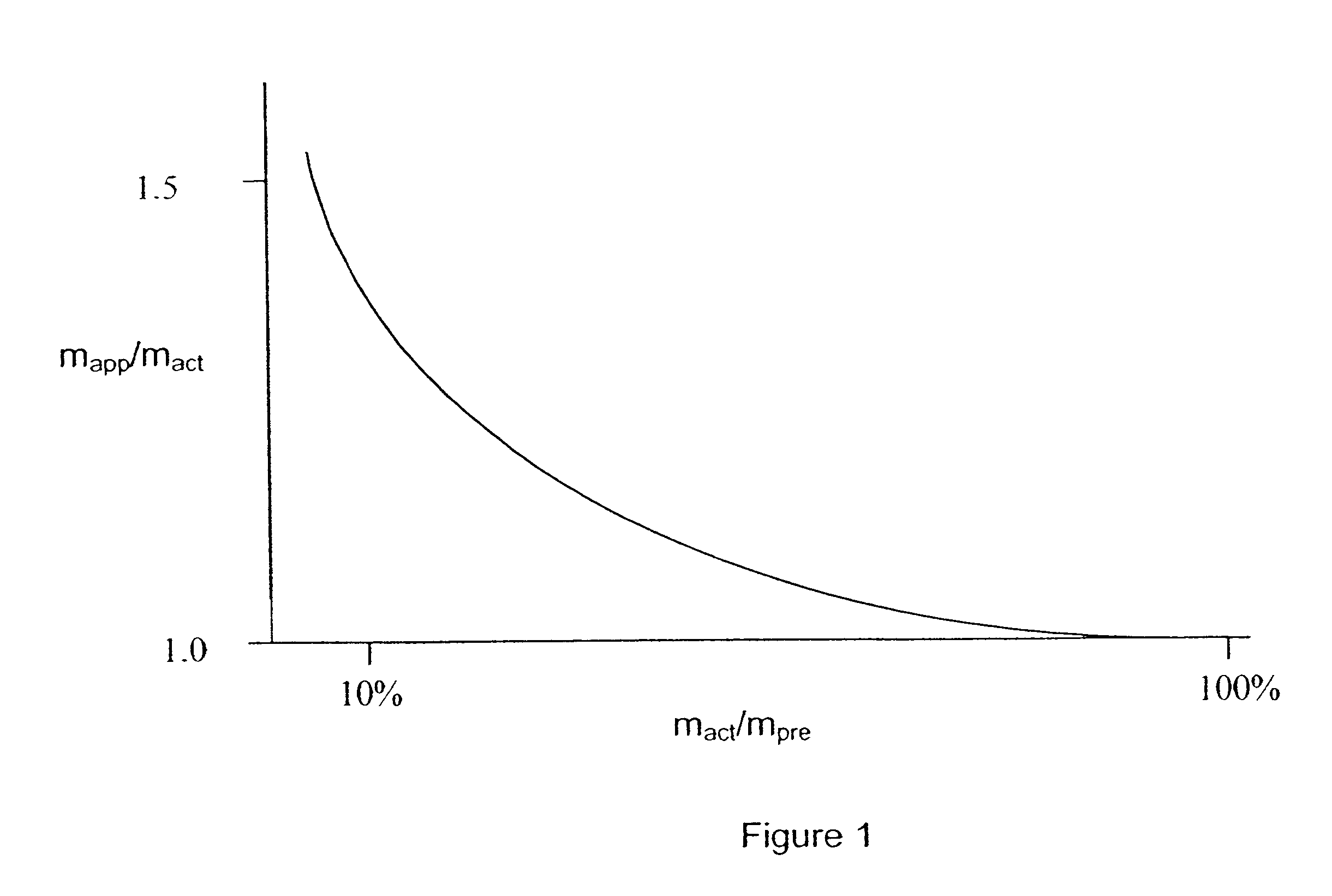
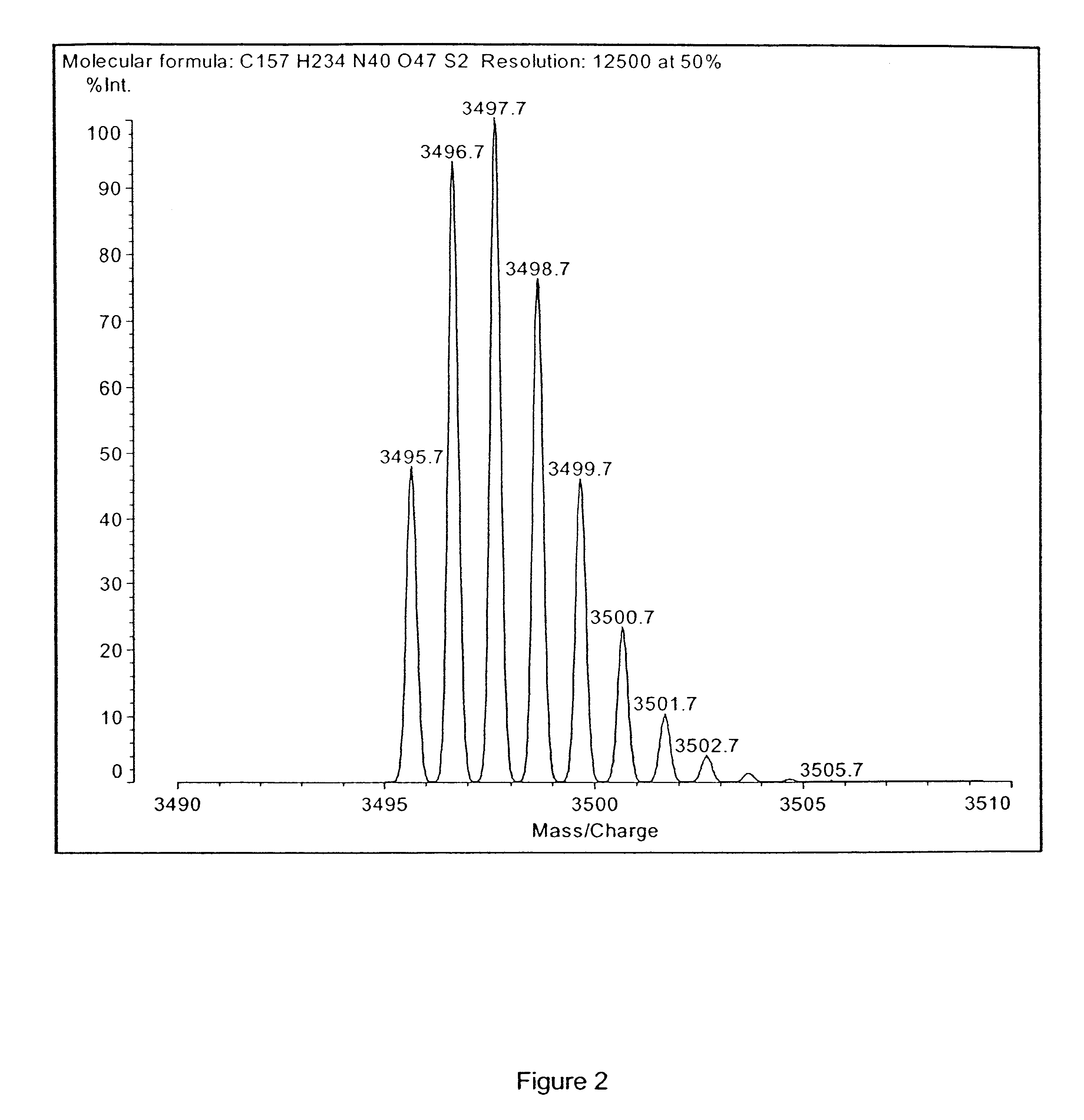
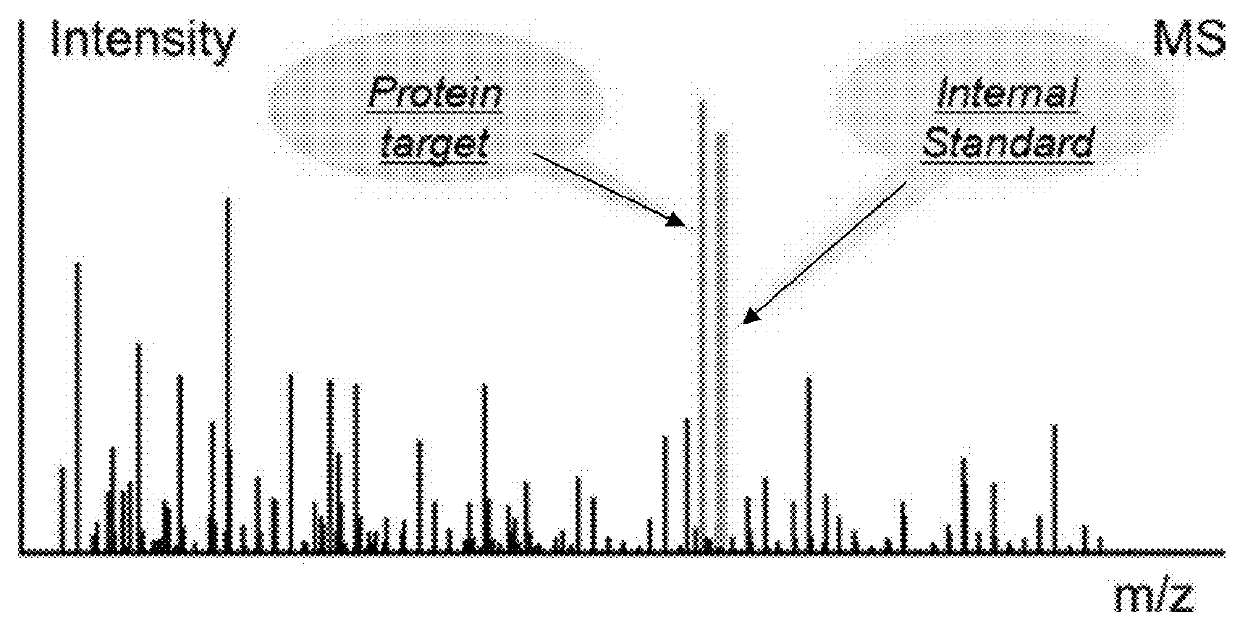
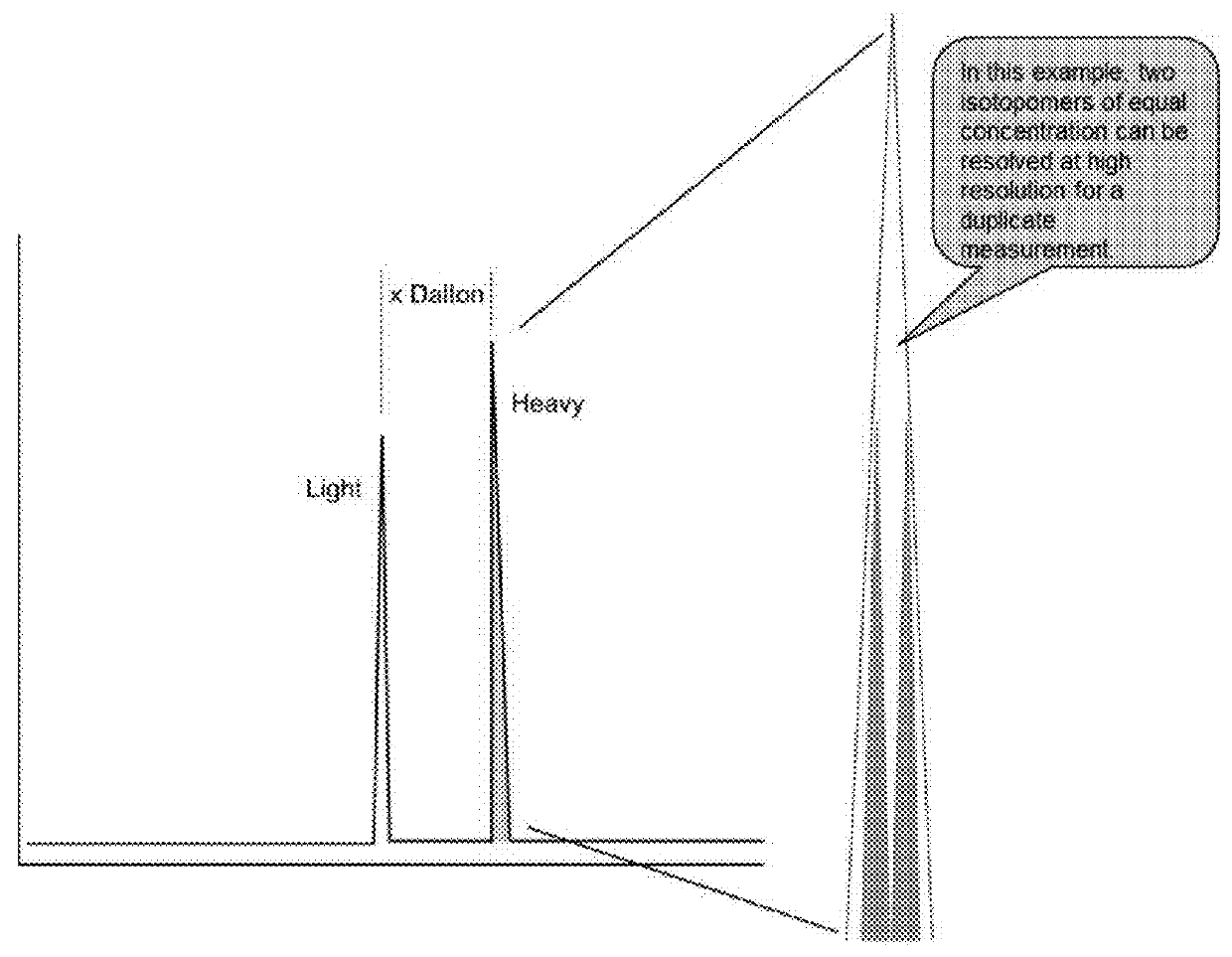
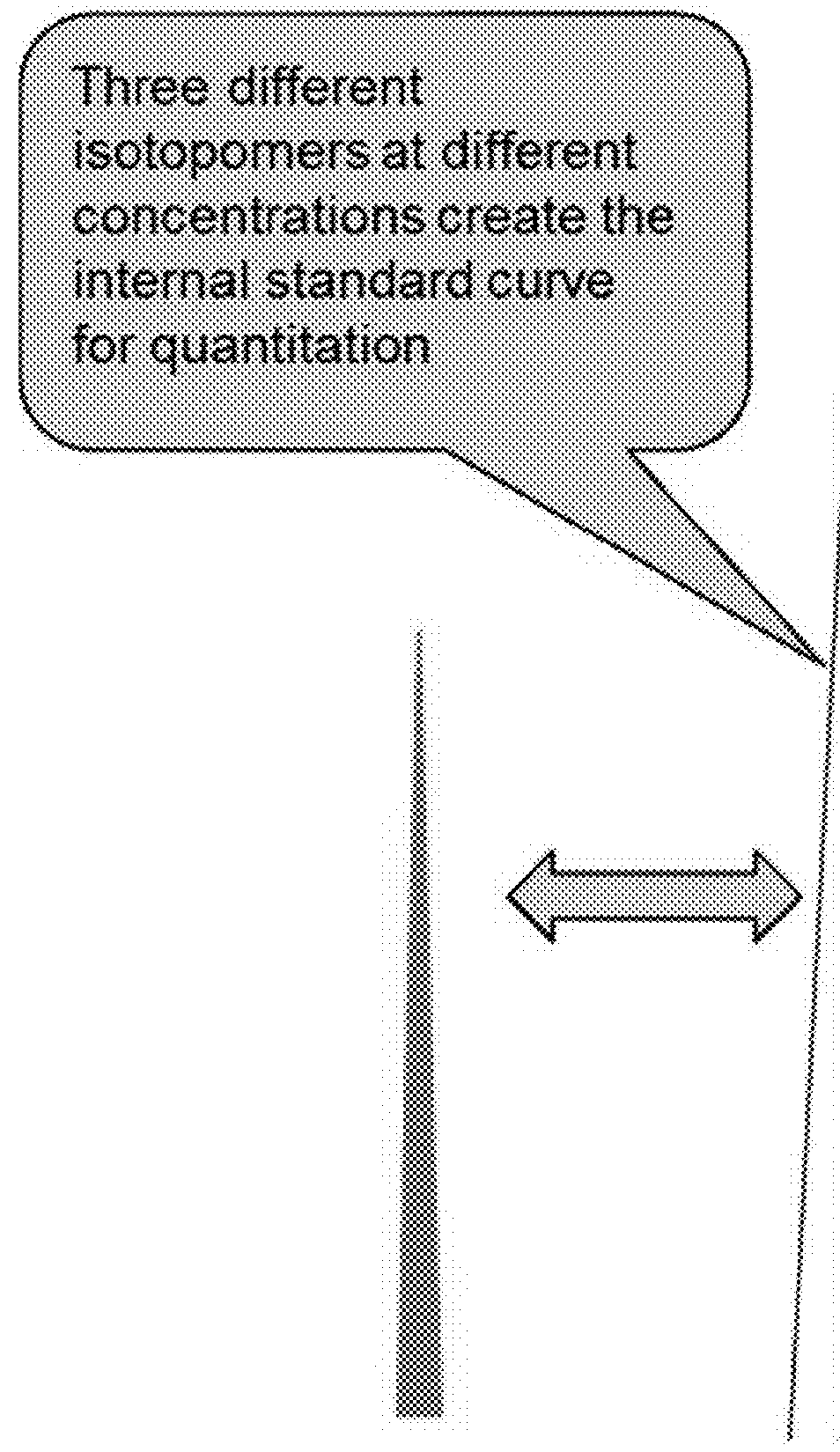
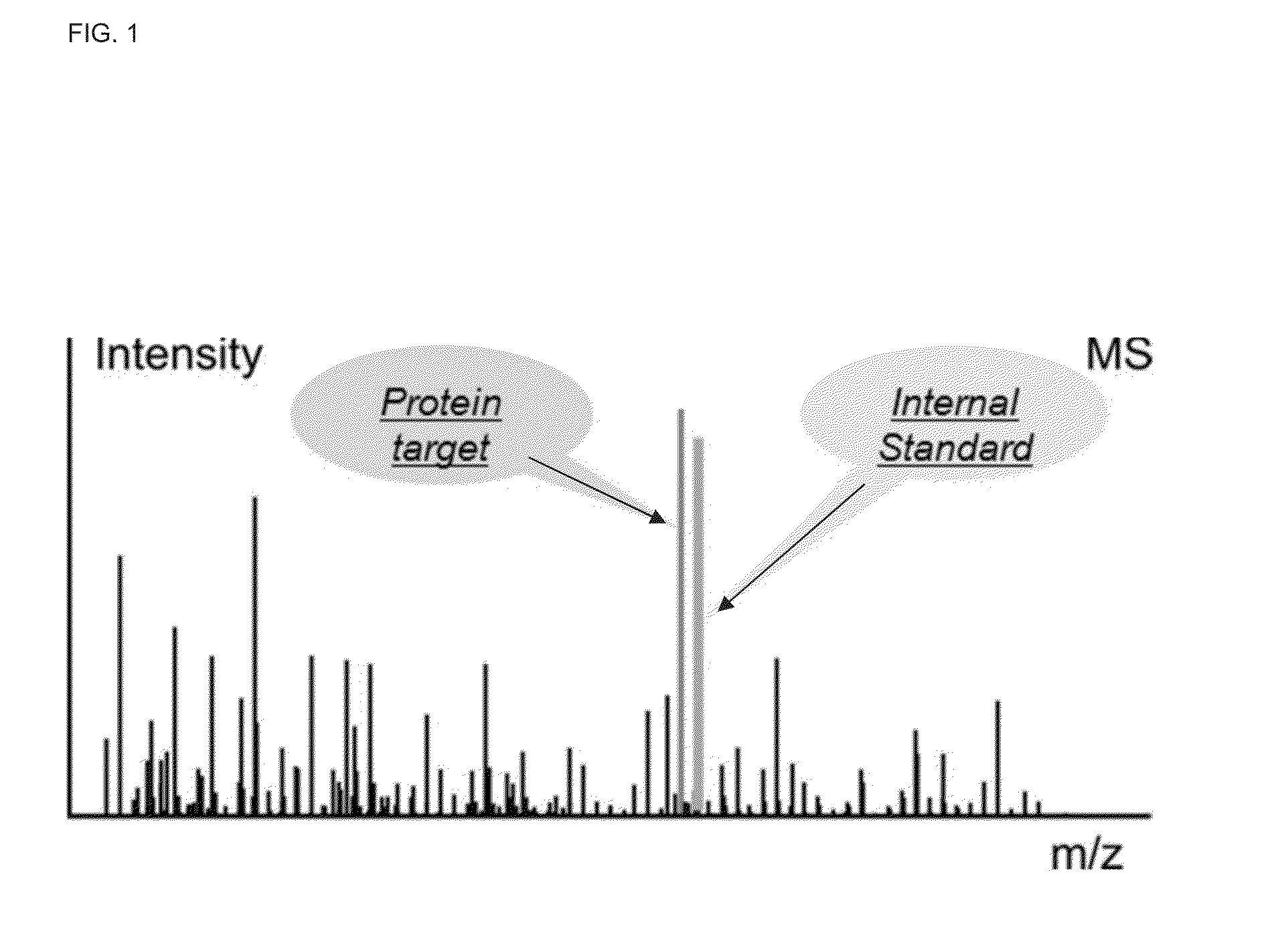
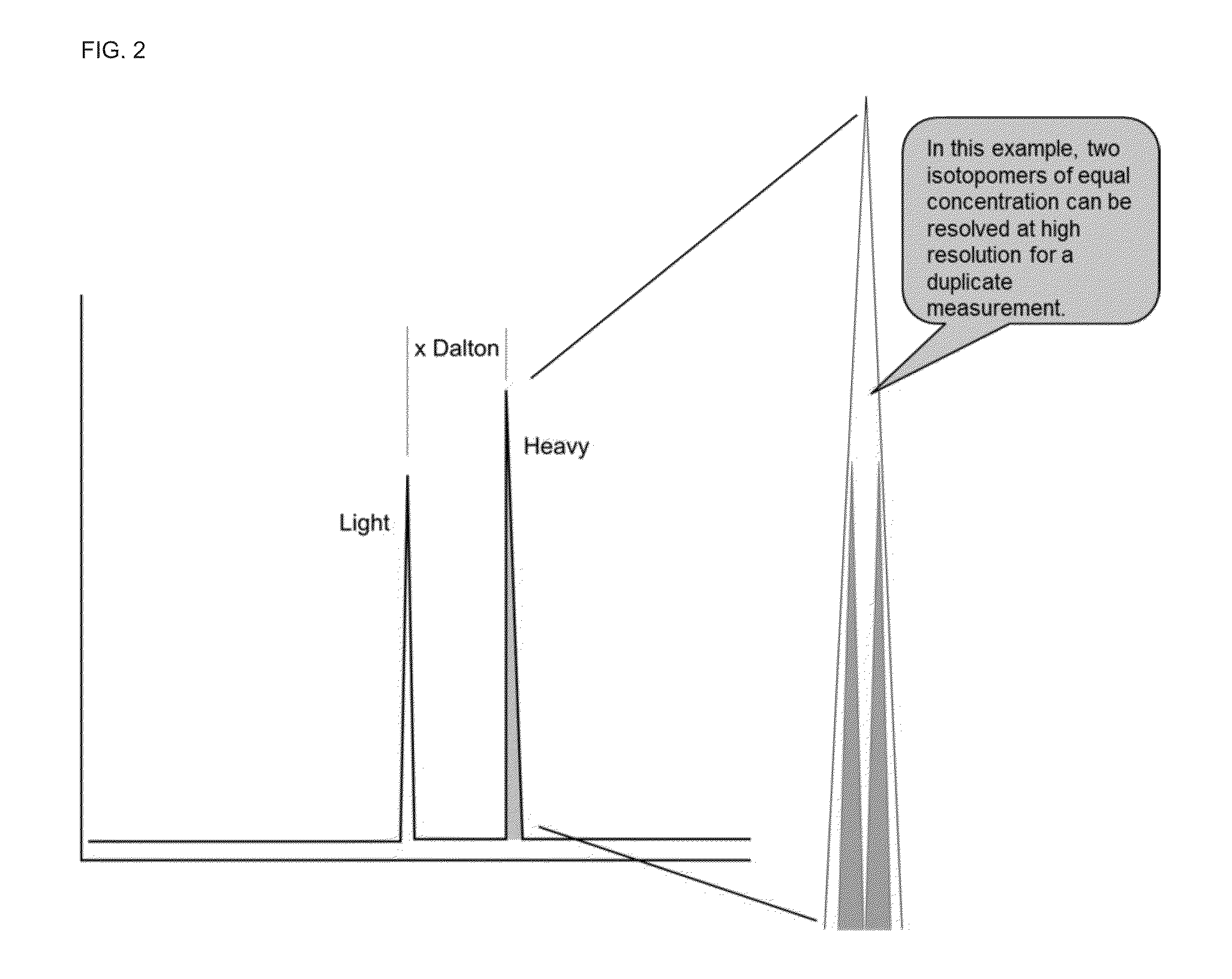
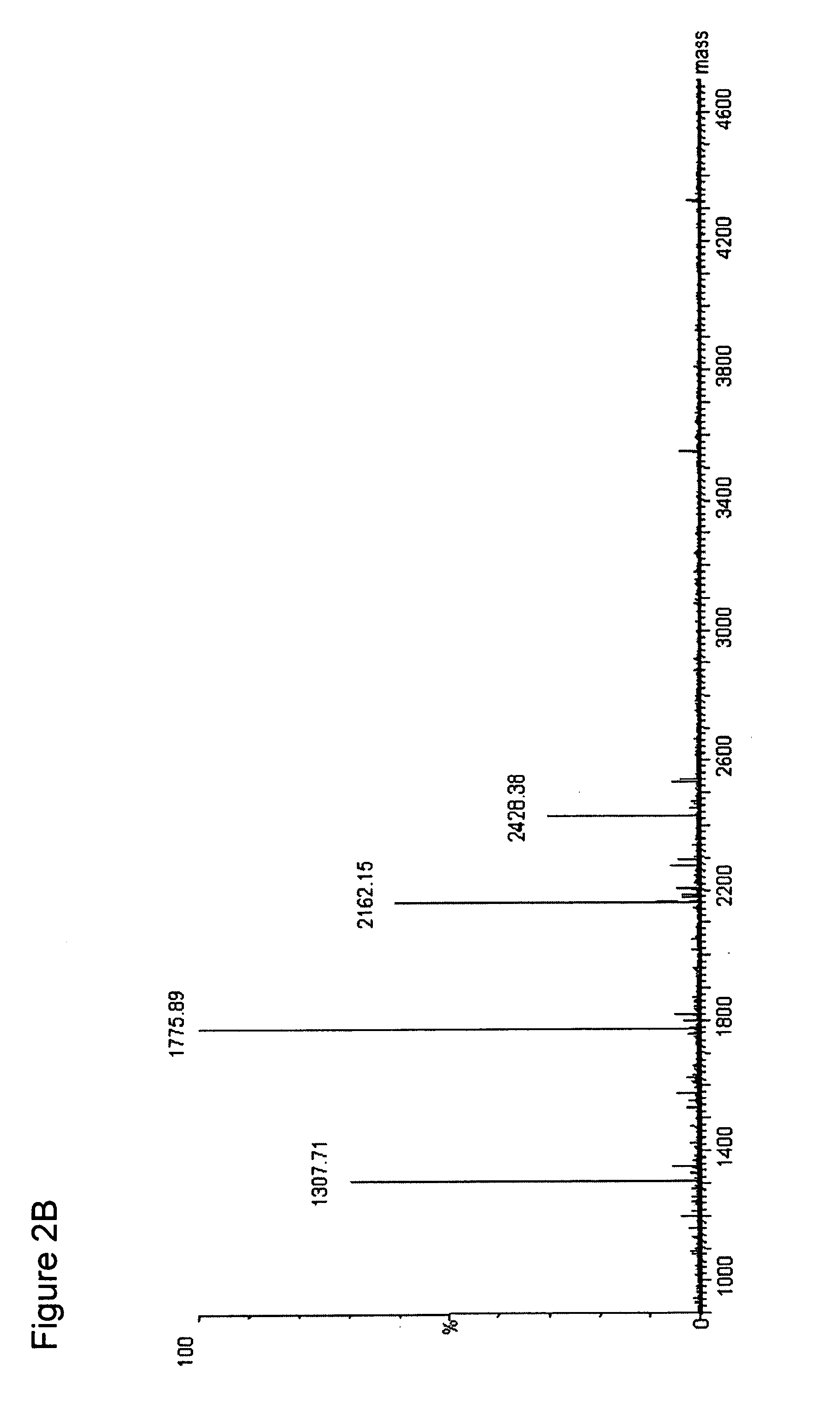
A method for absolute protein or peptide quantitation by mass spectroscopy. A sample containing a protein or peptide of interest is prepared for mass spectroscopy analysis. The sample is subjected to mass spectroscopy analysis at low resolution whereby a single additive mass spectroscopy peak is obtained, then is subjected to high resolution mass spectroscopy analysis whereby a plurality of mass spectroscopy peaks are obtained. The intensity of each of the plurality of mass spectroscopy peaks is quantitated either by comparison to an internal standard set, or by using a standard curve generated for each isotopologue set. Quantitation using a standard curve enhances quantitation across a dynamic range of analyte.
Owner:PIERCE BIOTECHNOLOGY
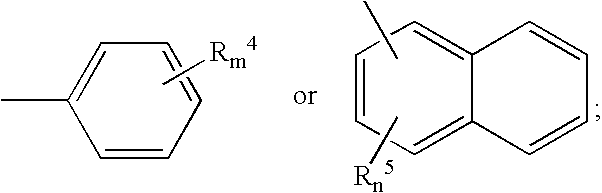
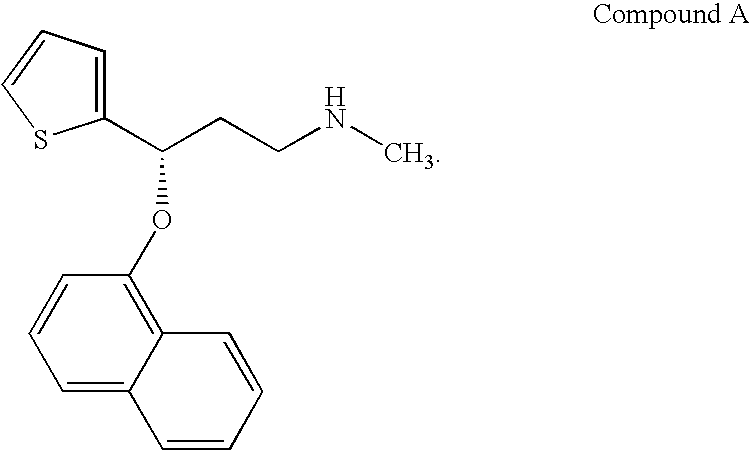
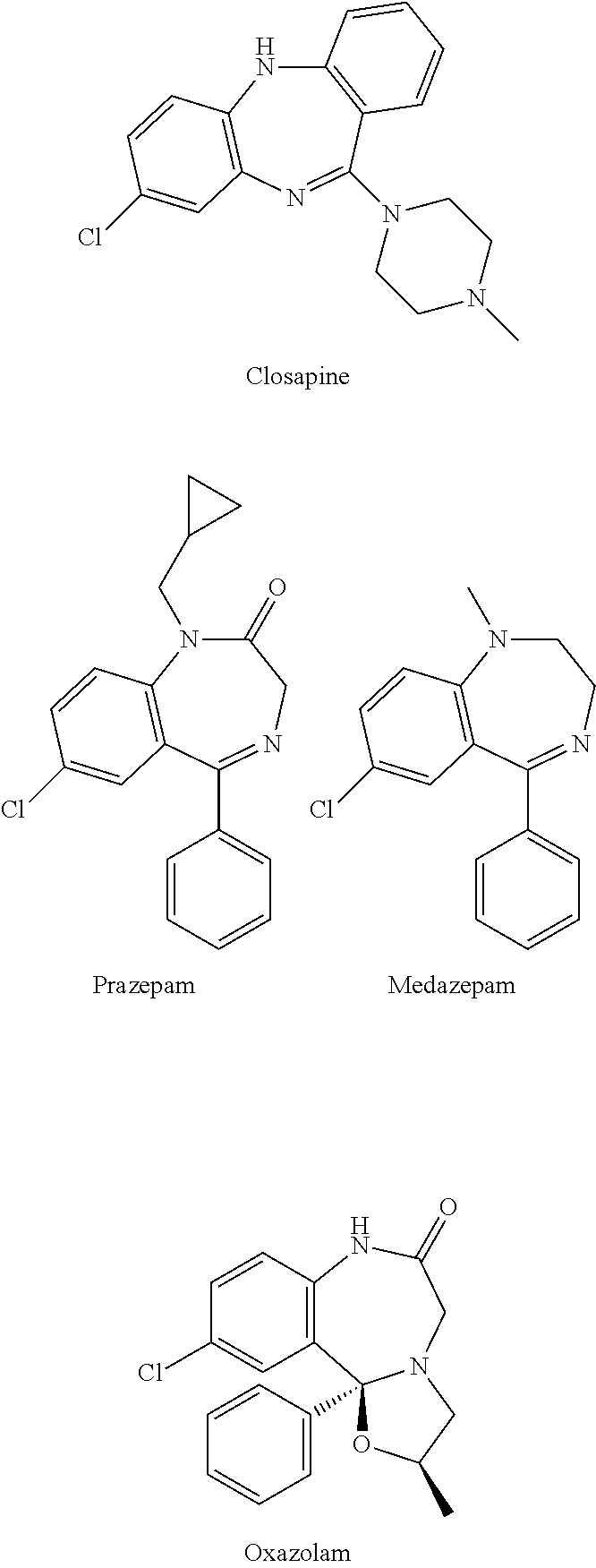
Novel benzo[D][1,3]-dioxol derivatives
InactiveUS20070191432A1Low efficiencyReduce probabilityBiocideSenses disorderBenzeneSerotonin reuptake
Owner:SUN PHARMA IND INC
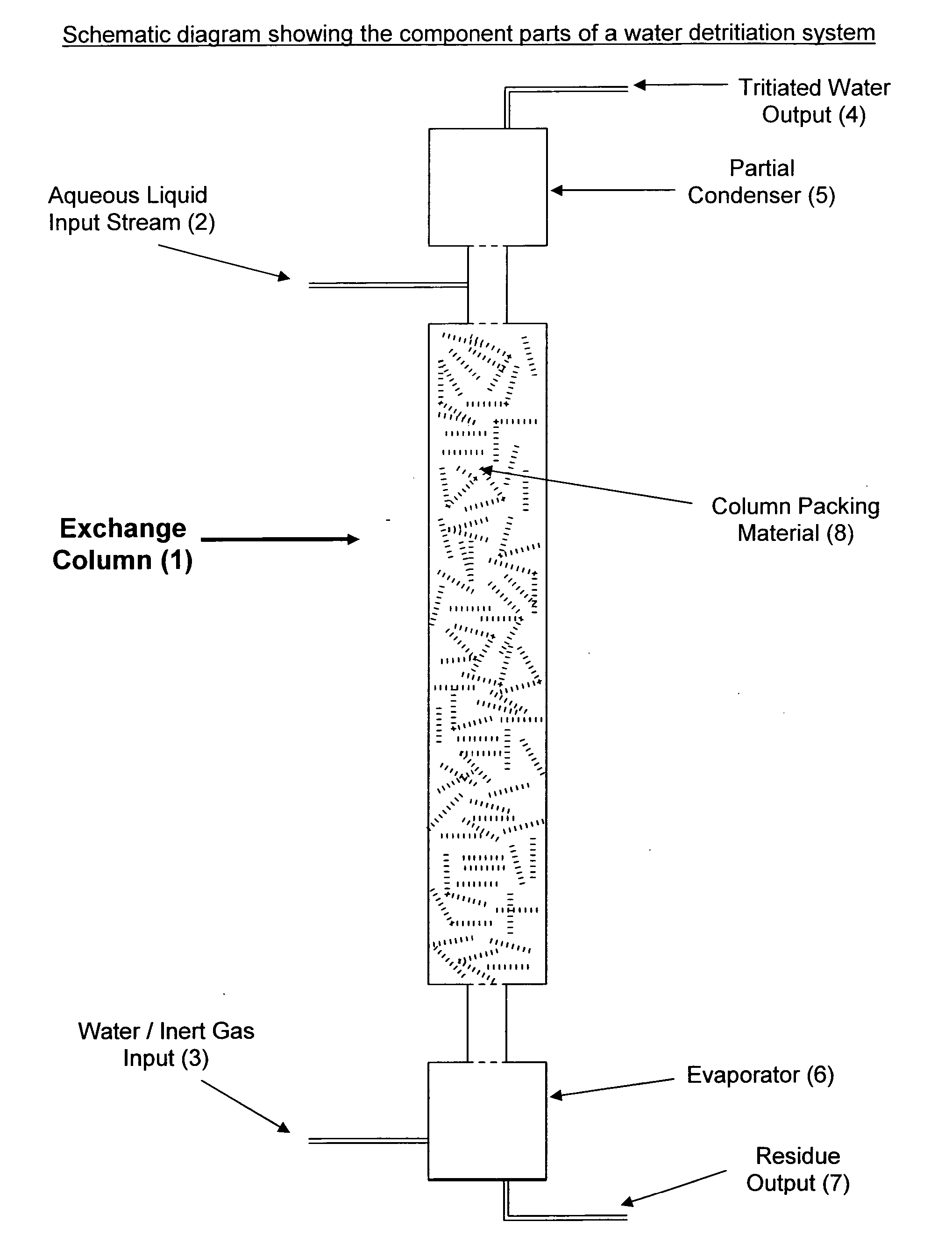
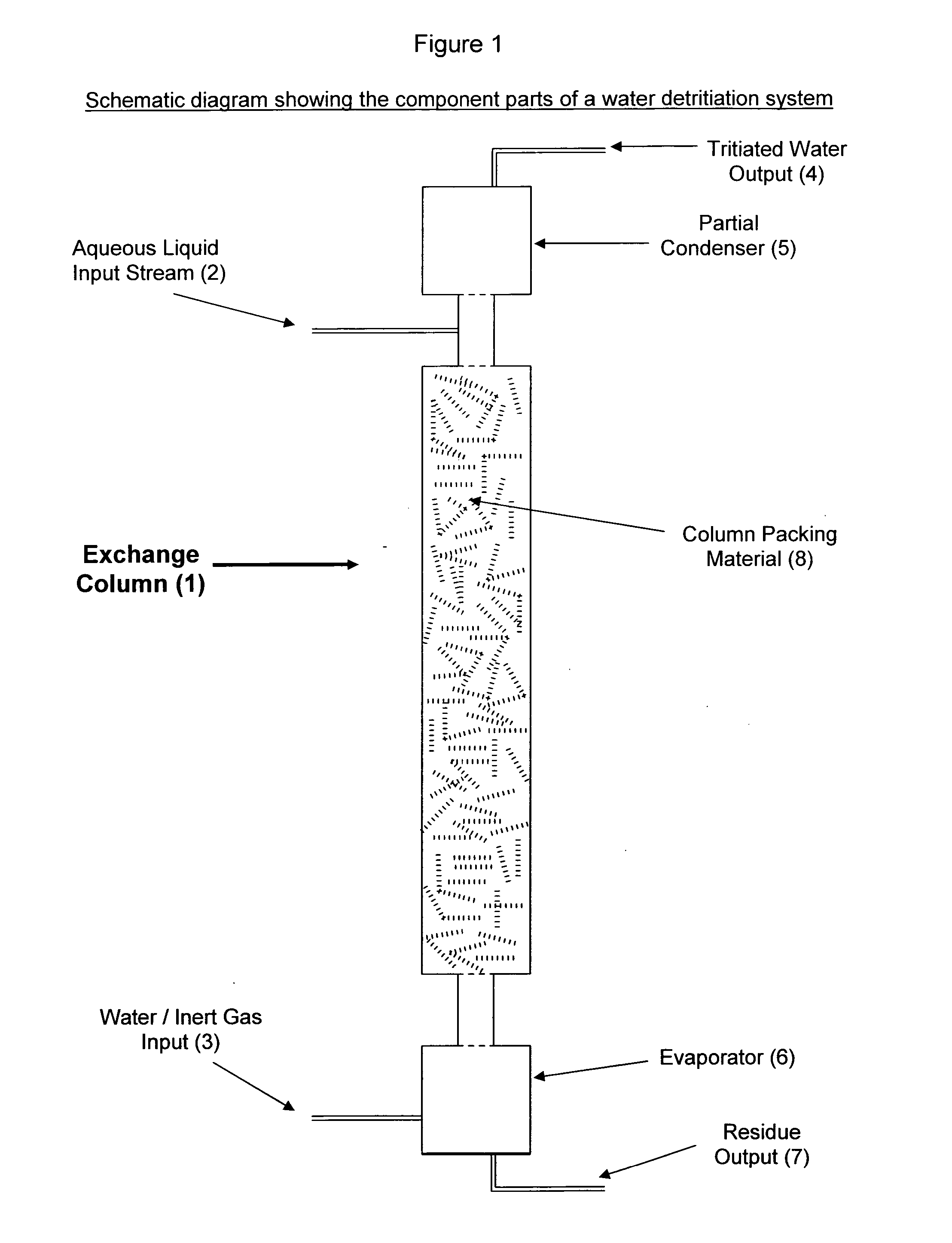
Method and device for production of radio-isotopes from a target
InactiveUS20050069076A1Precise temperature controlEasy to useConversion outside reactor/acceleratorsRadio isotopesIsotope
The invention relates to a method for production of a radio-isotope (4) from a target (3), containing a precursor (1) of said radio-isotope (4), using a beam of accelerated particles, comprising the following method steps: preparation of a target (3), containing the precursor (1) of the radioisotope (4), irradiation of said target (3) within an irradiation chamber (10) with a beam of accelerated particles in order to induce the transmutation of the precursor (1) into the radio-isotope (4), heating said target (3) in order to bring about the efflux of the radio-isotope (4) from the target (3), collection of said radio-isotope (4), extracted as a gas and condensation of said radio-isotope (4) into a solid or liquid. The invention further relates to a device for carrying out the above method and use of the device and method for the production of palladium 103 from rhodium 103.
Owner:ION BEAM APPL
Calibration method
InactiveUS6717134B2Time-of-flight spectrometersSamples introduction/extractionPeak valueTime of flight
In its most general terms the invention compensates for the effect of the mass offset in the prior art calibration method. This can be achieved either by correcting for the offset or assigning mass to the peaks in such a way that the offset is avoided. Accordingly in a first aspect there is provided a method of calibrating a reflectron time-of-flight mass spectrometer using a spectrum generated by fragment ions wherein the mass of the fragment ion is assigned using the mono-isotopic peak only. In other words a value corresponding to the mass of the fragment ion used for calibration is assigned using the fragment ion mono-isotopic peak only and said value is used to calibrate the spectrometer.
Owner:KRATOS ANALYTICAL
Absolute quantitation of proteins and protein modifications by mass spectrometry with multiplexed internal standards
ActiveUS9252003B2Fast and accurate and precise quantitationImprove accuracyParticle spectrometer methodsMass spectrometersSpectroscopyMass spectrometry imaging
A method for absolute protein or peptide quantitation by mass spectroscopy. A sample containing a protein or peptide of interest is prepared for mass spectroscopy analysis. The sample is subjected to mass spectroscopy analysis at low resolution whereby a single additive mass spectroscopy peak is obtained, then is subjected to high resolution mass spectroscopy analysis whereby a plurality of mass spectroscopy peaks are obtained. The intensity of each of the plurality of mass spectroscopy peaks is quantitated either by comparison to an internal standard set, or by using a standard curve generated for each isotopologue set. Quantitation using a standard curve enhances quantitation across a dynamic range of analyte.
Owner:PIERCE BIOTECHNOLOGY
Application of chlorogenic acid and derivative in preparing sensitizer for tumor immunotherapeutic drug
InactiveCN106890169AIncrease percentageProliferation effect is goodOrganic active ingredientsAntibody ingredientsChlorogenic acidHalogen
The invention discloses application of a compound as shown in a formula I, or a stereoisomer thereof, or a pharmaceutically acceptable salt thereof, or a solvate thereof, or a crystal form thereof, or an isotopologue thereof in preparing a sensitizer for a tumor immunotherapeutic drug, wherein R1-R3 are independently selected from hydroxyl, C1-6 alkoxy, substituents as shown in a formula II or cis-isomers thereof; and R4 and R5 are independently selected from hydrogen, halogen, C1-6 alkyl, hydroxyl or C1-6 alkoxy. A pharmacological experiment result verifies that the chlorogenic acid and the derivative thereof which are combined in use with the tumor immunotherapeutic drug can better play a role of killing tumor cells, and the sensitizer as the tumor immunotherapeutic drug has wide clinical application prospects.
Owner:SICHUAN JIUZHANG BIO TECH CO LTD
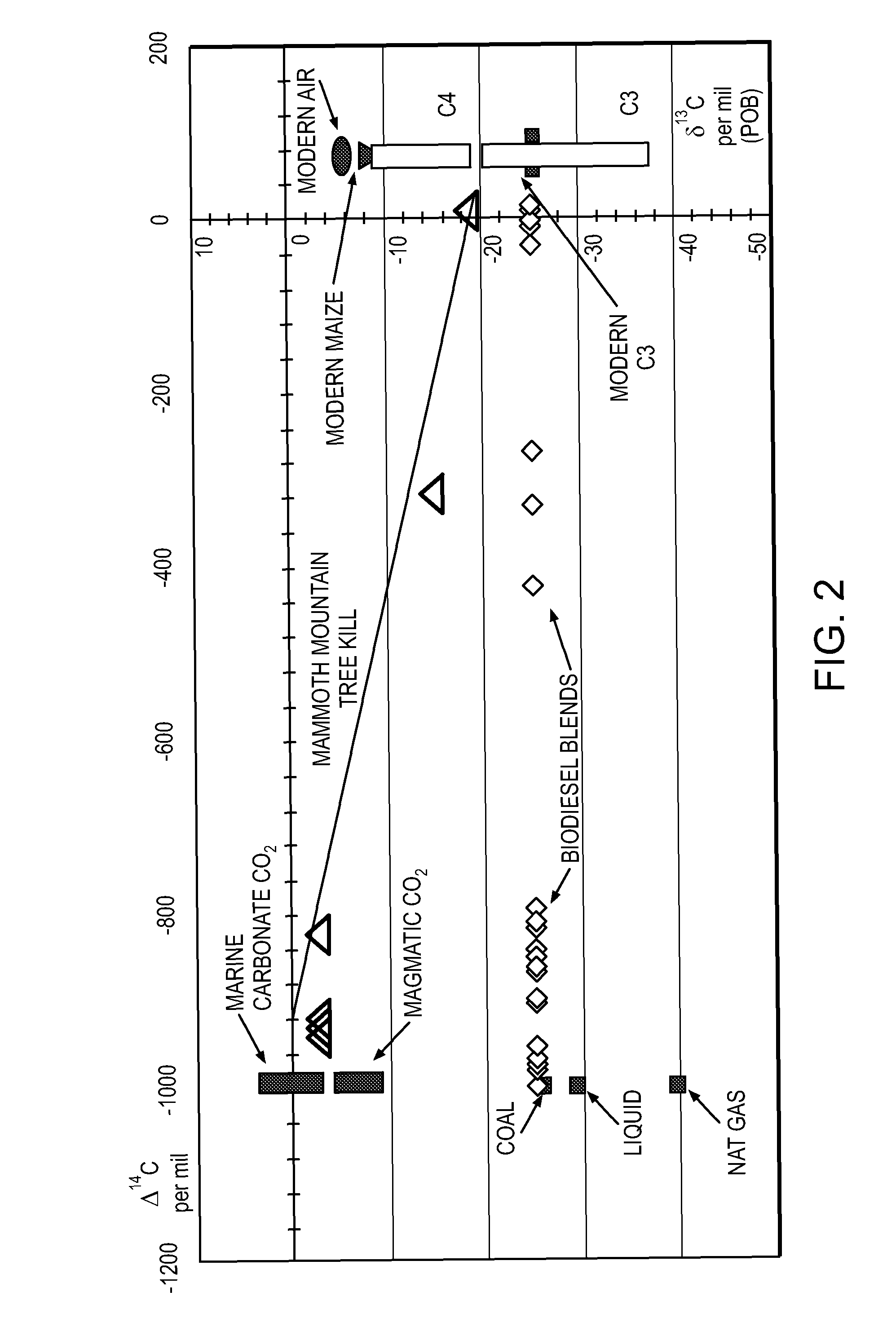
System and method for isotopic signature and mass analysis
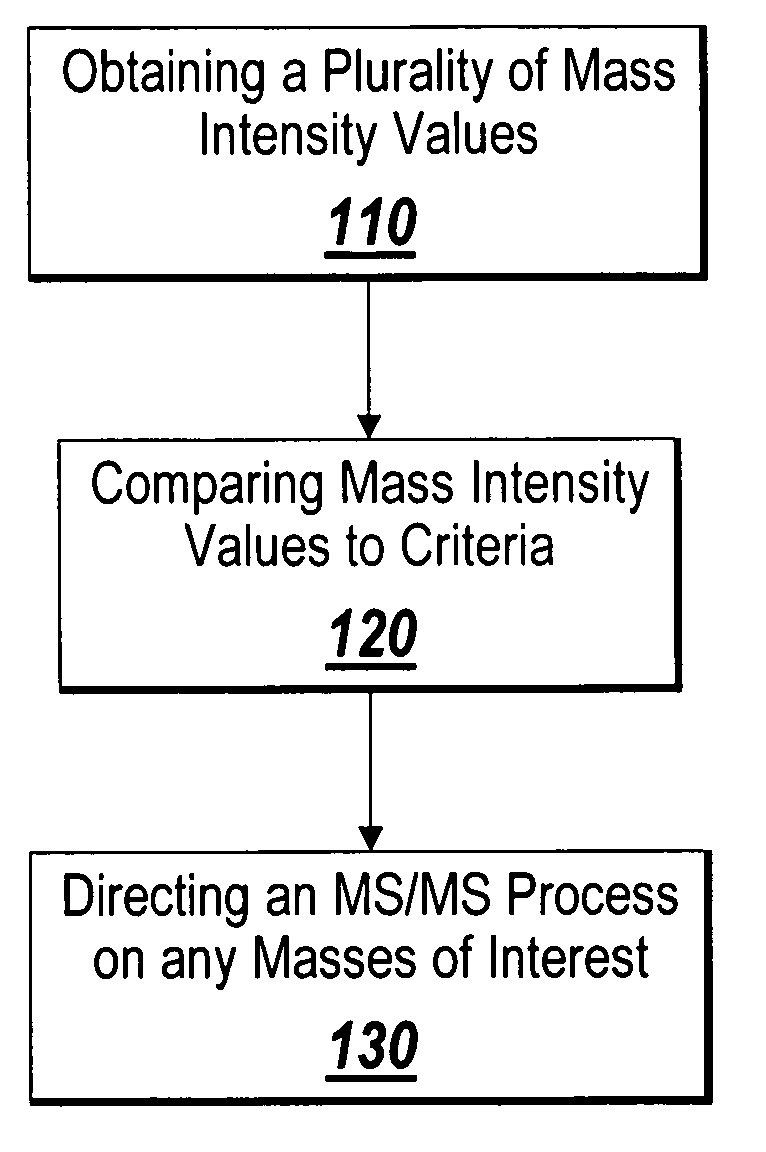
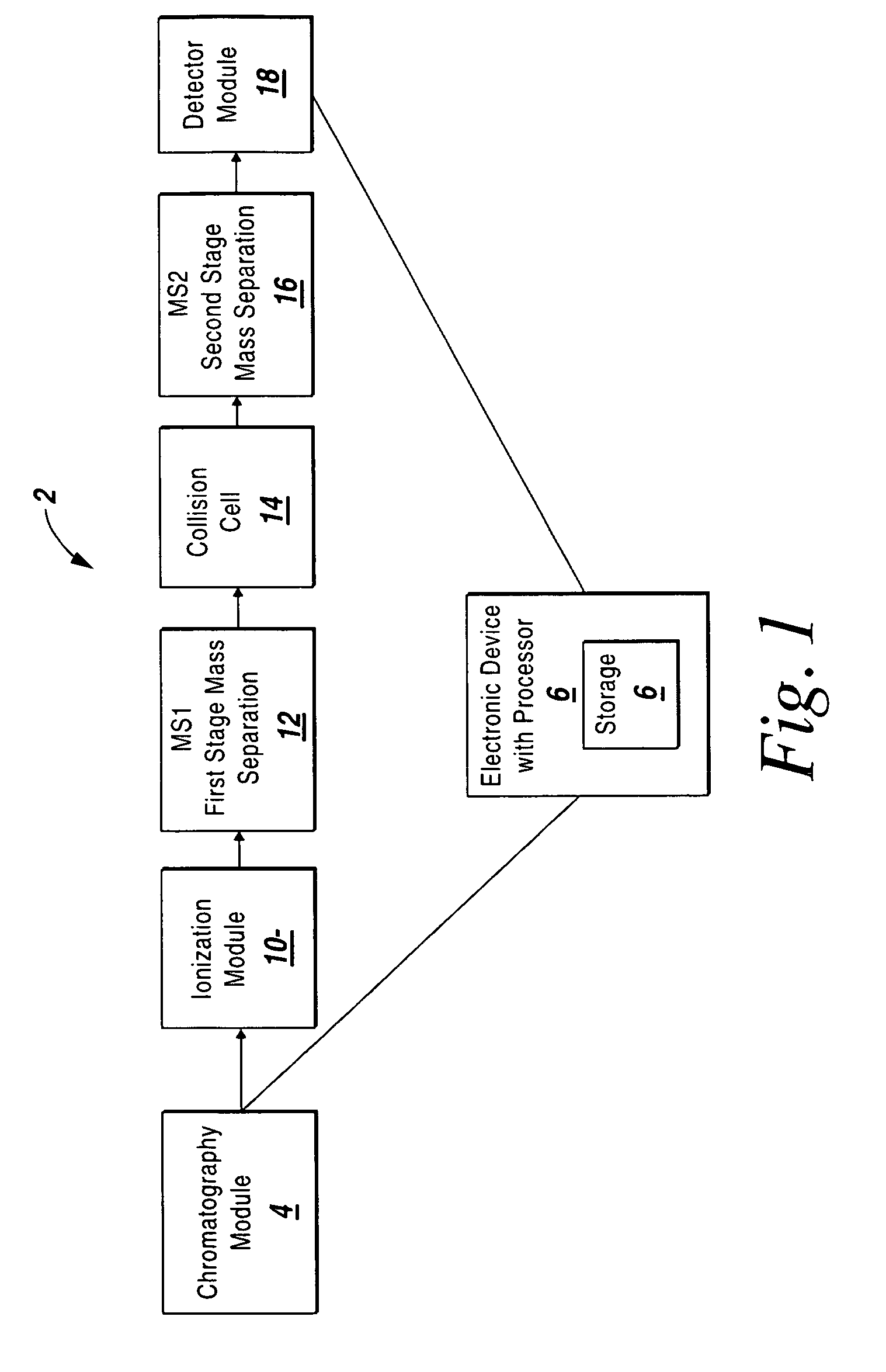
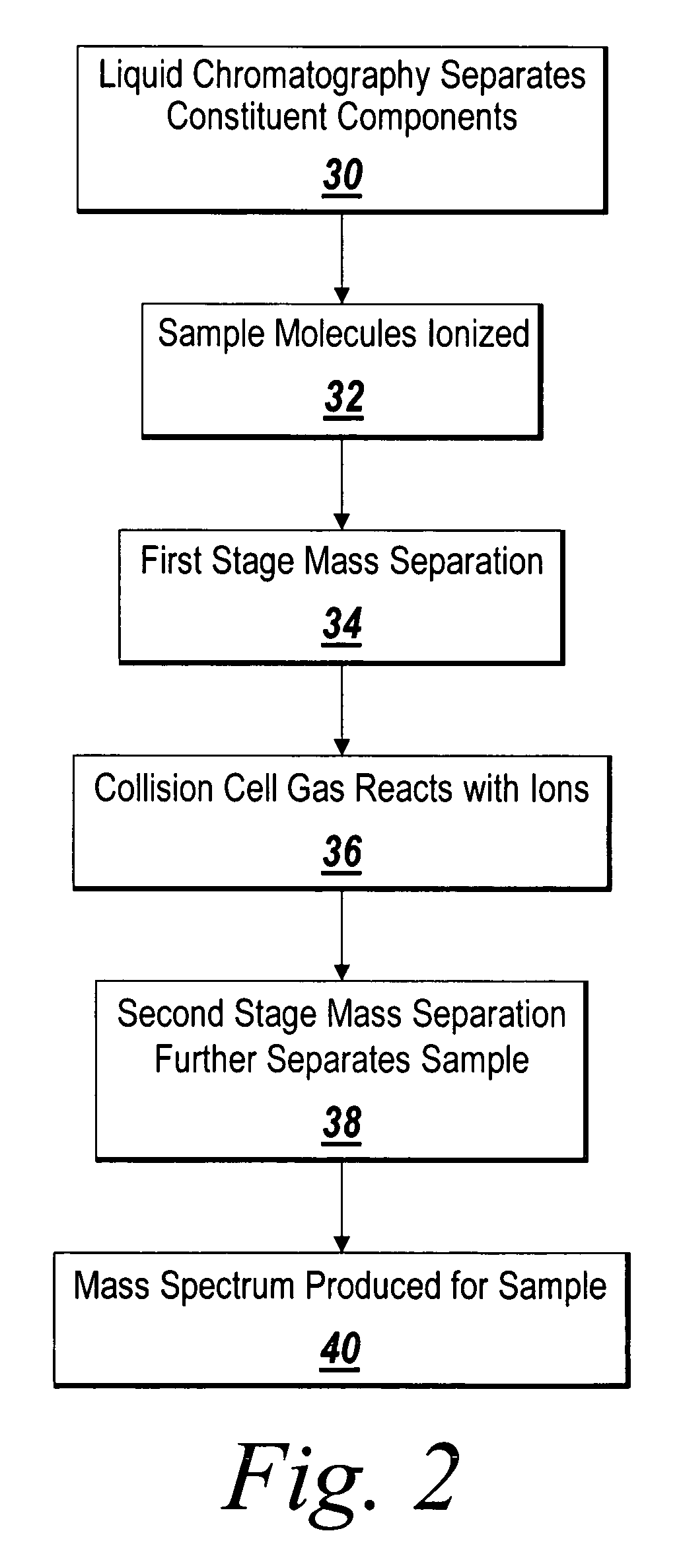
Mass intensity values, isotopic ratios and exact mass differences between isotopes may be obtained and analyzed to determine if there are one or more masses of interest. In one implementation, a method will look among the mass intensity values for peaks of interest. The mass intensity values, representative of isotopic signatures, will be compared to a criteria in order to determine which of the masses are of interest. Although the invention is not so limited, examples of such criteria include mass intensity values above a specified threshold, within a certain ratio of another mass intensity value, and / or separation between the masses themselves. Optionally, a tolerance may be applied to the criteria. If masses are found to be of interest, MS / MS can automatically be triggered for one or all of the masses of interest, when an analyzing system such as an LC-MS-MS, GC-MS-MS or MALDI-MS-MS system is used.
Owner:MICROMASS UK LTD
Unimodal 87 Rb isotopic atomic filter used for laser frequency stabilization and filter method thereof
ActiveCN103701030AReduce magnetic field strengthEasy to implementLaser detailsOptical elementsExternal cavity laserPrism
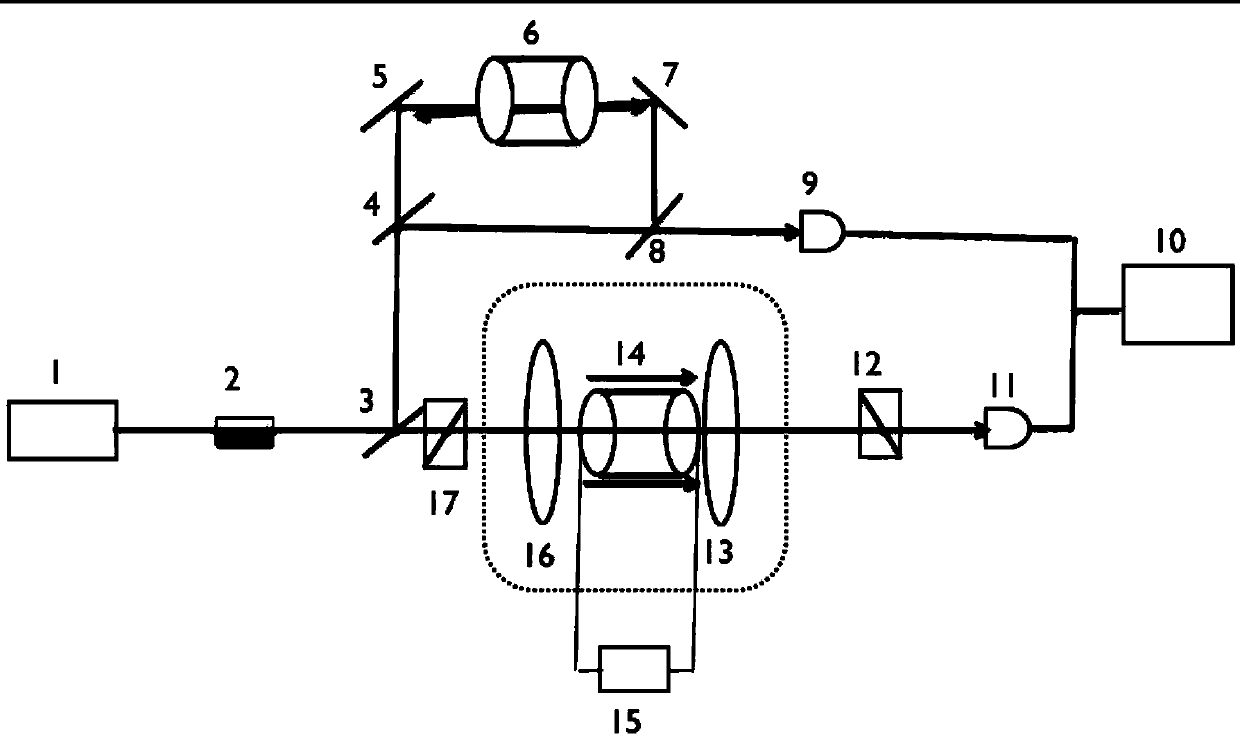
The invention relates to a unimodal 87 Rb isotopic atomic filter used for laser frequency stabilization and a method for filtering by adopting the atomic filter. The filter consists of components of an external cavity laser 1, an optical isolator 2, a first semipermeable semi-reflective mirror 3, a second semipermeable semi-reflective mirror 4, a first high reflectivity mirror 5, a natural rubidium cell 6 and the like. According to the unimodal 87 Rb isotopic atomic filter used for laser frequency stabilization and the filter method thereof, by using the Faraday anomalous dispersion effect of isotopic 87 Rb ground state atoms in a magnetic field, a uniform magnetic field is generated through a magnetic field source, such as a permanent magnet, and the laser and atoms are interacted, so the laser polarization direction is changed. The laser transmission polarization direction is limited by two polarizing prisms, the light wave with specific frequency is chosen, and the goal of filtering is achieved. A system is enabled to be in a good working condition through a temperature control system. The unimodal atomic filter has the advantages of simple structure, high filtering performance, work stability, long service life and the transmission rate of being more than 78.0%. The unimodal 87 Rb isotopic atomic filter used for laser frequency stabilization and the filter method thereof have important significance in the experiment with interaction between light and atoms.
Owner:PEKING UNIV
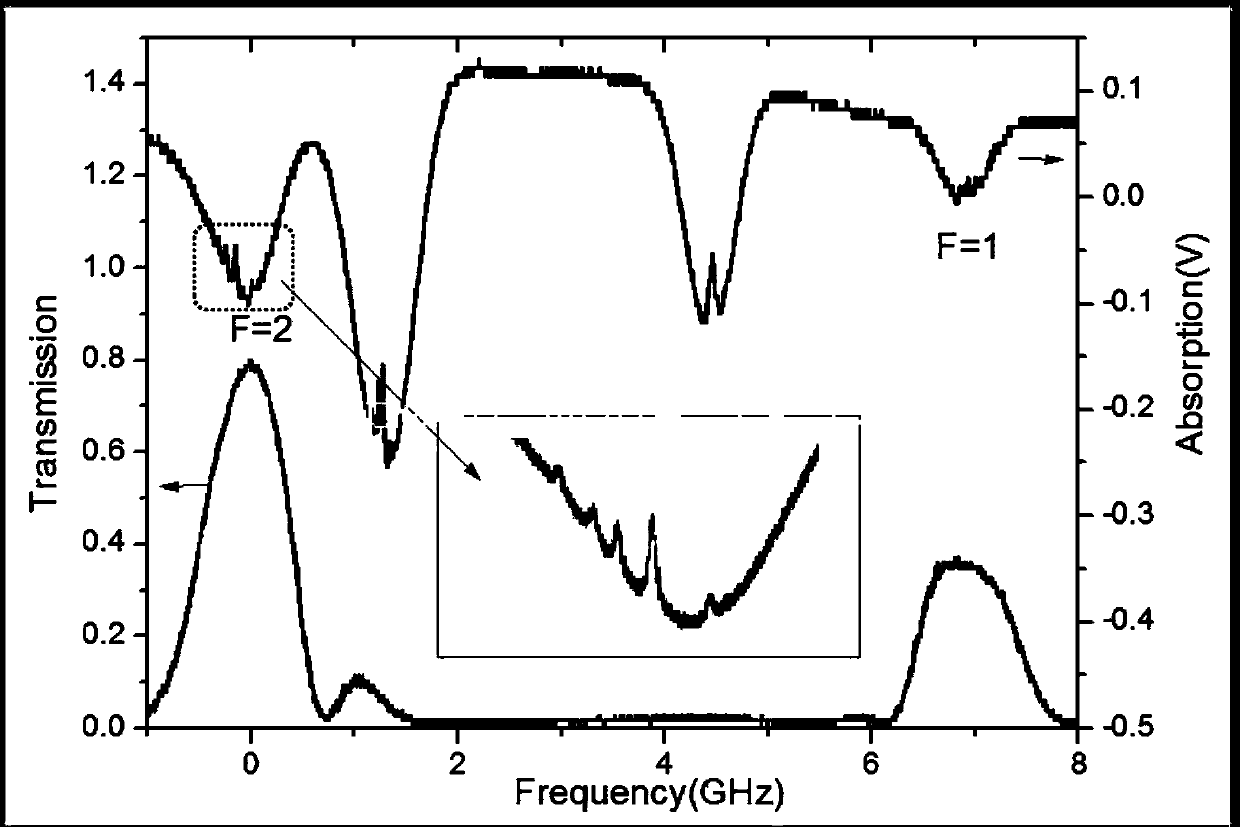
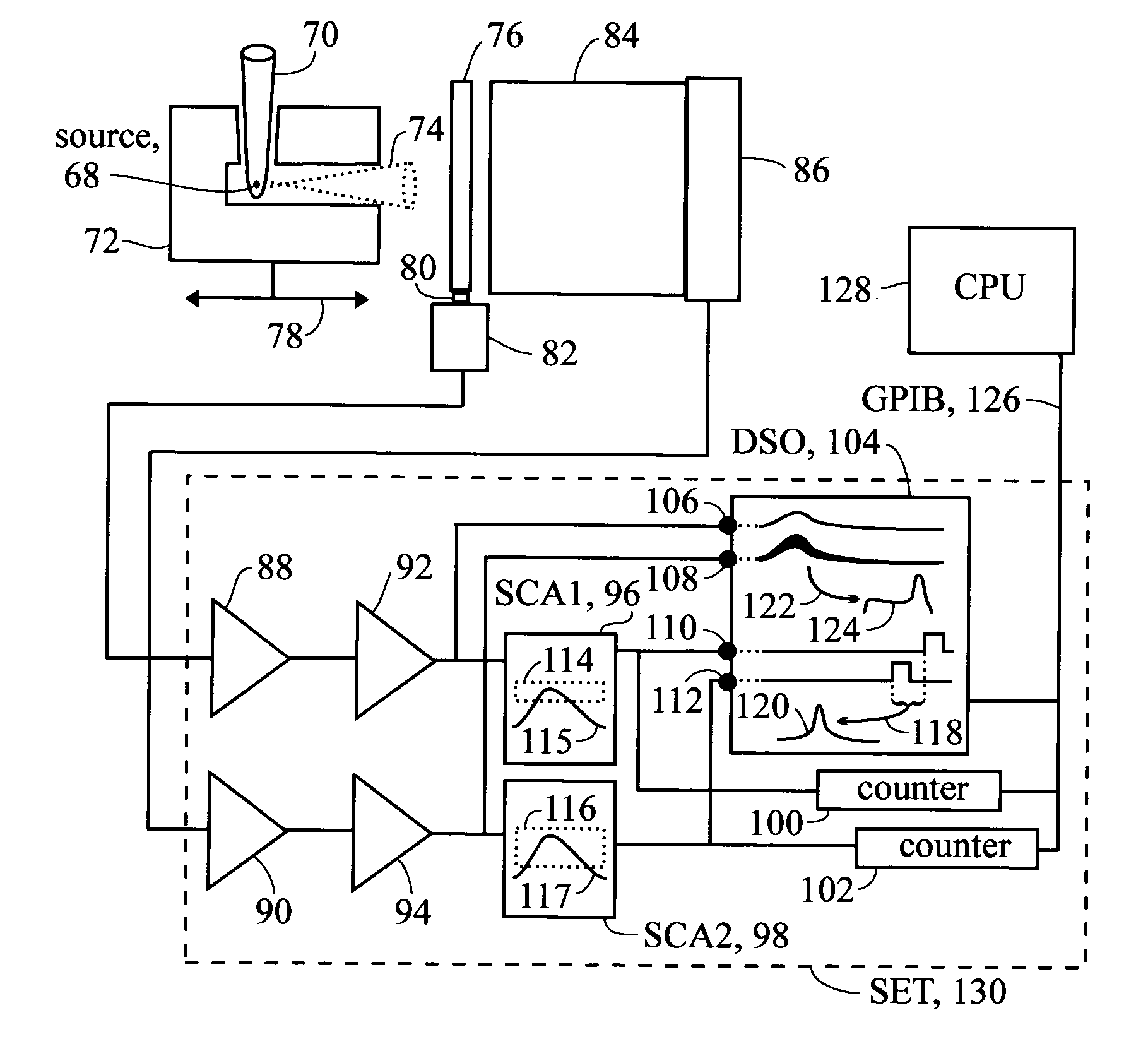
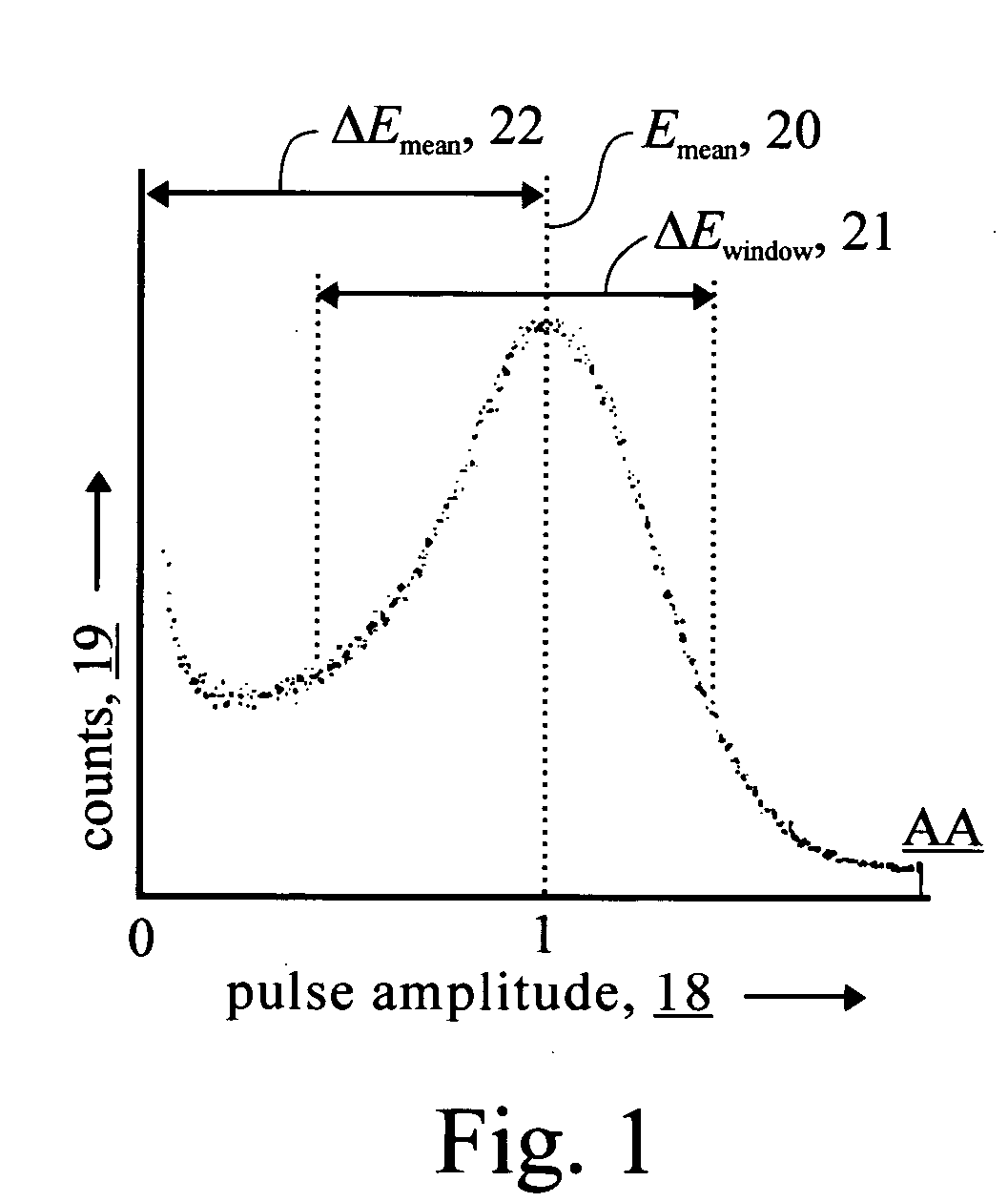
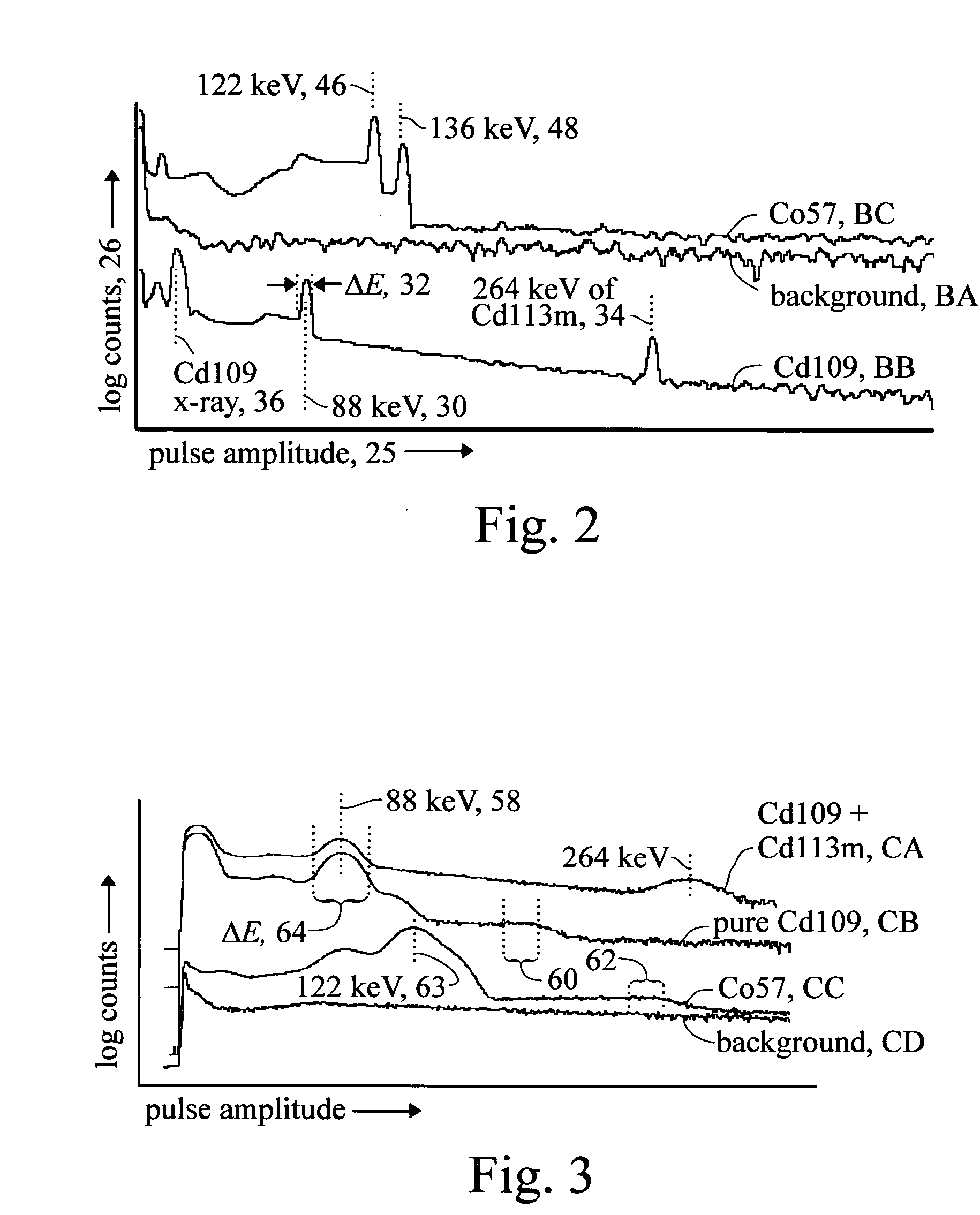
Photon violation spectroscopy
InactiveUS20050139776A1Reduce chanceAvoid smoothX-ray spectral distribution measurementMaterial analysis by optical meansBeam splitterSpectroscopy
The method typically uses spontaneous gamma rays from radioisotopes, either cadmium-109 at 88 keV or cobalt-57 at 122 keV, detected with NaI(Tl) or HPGe. After a two-part split, detection pulses are windowed for the characteristic gamma ray pulse amplitude and measured in coincidence. By using high resolution detectors and gamma rays that match the part of the spectrum where the detector has a high photoelectric effect efficiency, coincidence rates are found to substantially exceed the chance rate. This refutes the quantum mechanical prediction of energy quantization. This unquantum effect implies that photons are an illusion, and is explained by an extension of the abandoned loading theory of Planck to derive the photoelectric effect equation. In scattering gamma rays in a beam splitter geometry, changes in response to magnetic fields, temperature, and crystal orientation are tools for measuring properties of atomic bonds. With detectors in a tandem geometry where the first detector is both scatterer and absorber, tests reveal properties consistent with a classical gamma ray model. The method has also shown use in discovering that different crystalline states of the gamma ray source change the extent coincidence rates exceed chance, whereas conventional gamma ray spectroscopy shows no substantial dependence upon these applied variables.
Owner:REITER ERIC STANLEY
Novel aryloxypropanamines
InactiveUS20070116643A1Reduction in rate of metabolismSubstantial isotope effectIsotope introduction to heterocyclic compoundsHydrogen isotopesDisease causeDisease injury
The present invention relates to an aryloxypropanamine containing one or more deuterium or 13C in place of a hydrogen or carbon, respectively. These isotopic aryloxypropanamines of the invention are inhibitors of serotonin and norepinephrine uptake and possess unique biopharmaceutical and pharmacokinetic properties compared to the responding light atom isotopologues. The invention further provides compositions comprising these isotopic aryloxypropanamines and methods of treating diseases and conditions linked to reduced neurotransmission of norepinephrine and / or serotonin. It further provides methods for using these isotopic aryloxypropanamines to accurately determine the concentration of the light atom isotopologues in biological fluids and of studying the metabolism of the light atom isotopologues.
Owner:CONCERT PHARMA INC
Absolute Quantitation of Proteins and Protein Modifications by Mass Spectrometry with Multiplexed Internal Standards
InactiveUS20160154006A1Improve accuracyHigh sensitivityPeptide librariesLibrary screeningAnalyteImage resolution
A method for absolute protein or peptide quantitation by mass spectroscopy. A sample containing a protein or peptide of interest is prepared for mass spectroscopy analysis. The sample is subjected to mass spectroscopy analysis at low resolution whereby a single additive mass spectroscopy peak is obtained, then is subjected to high resolution mass spectroscopy analysis whereby a plurality of mass spectroscopy peaks are obtained. The intensity of each of the plurality of mass spectroscopy peaks is quantitated either by comparison to an internal standard set, or by using a standard curve generated for each isotopologue set. Quantitation using a standard curve enhances quantitation across a dynamic range of analyte.
Owner:PIERCE BIOTECHNOLOGY INC
Method of Treating Metabolic Syndrome
InactiveUS20080292719A1Elevated blood pressureElevated triglyceride levelBiocideMetabolism disorderMammalMedical treatment
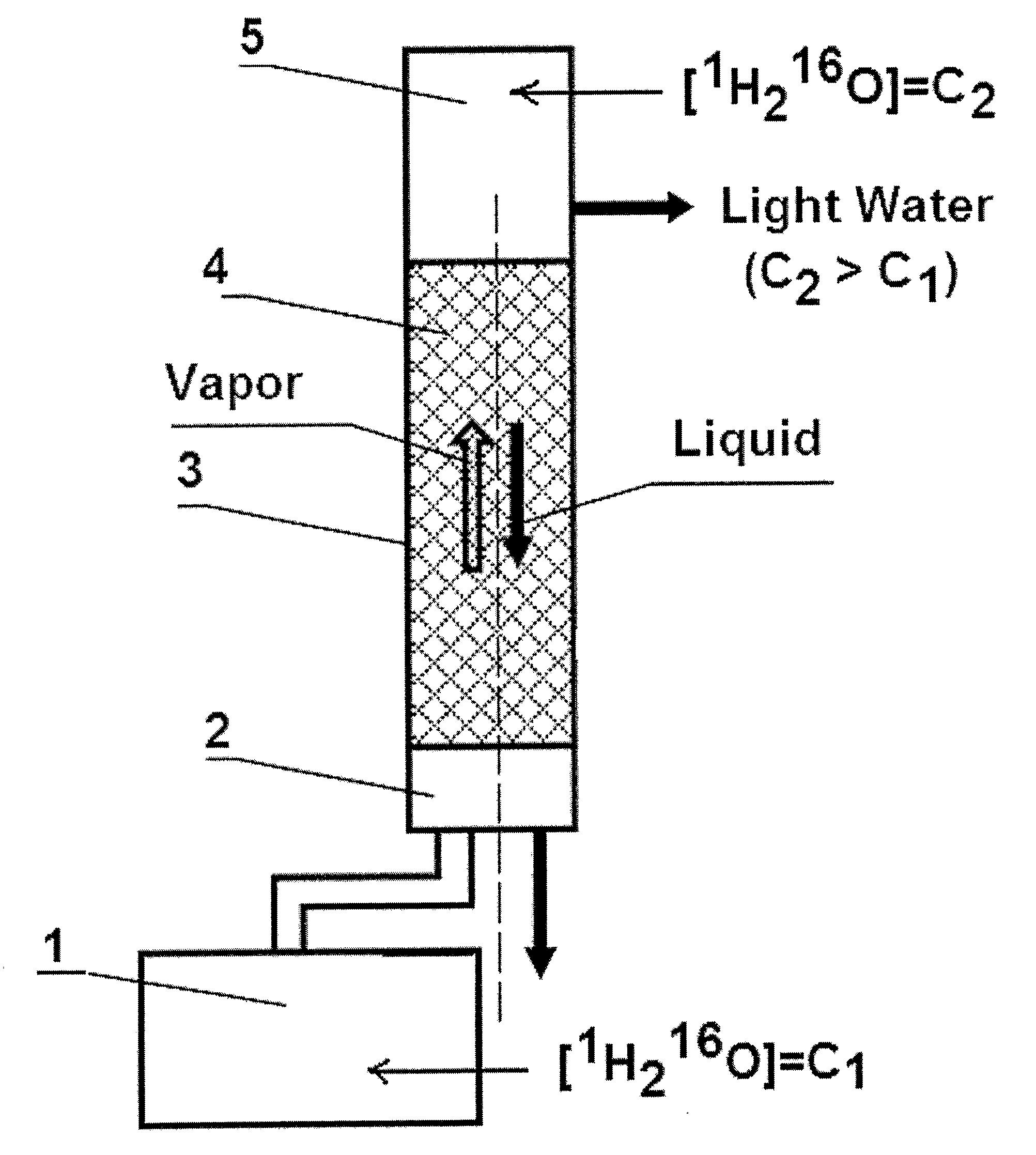
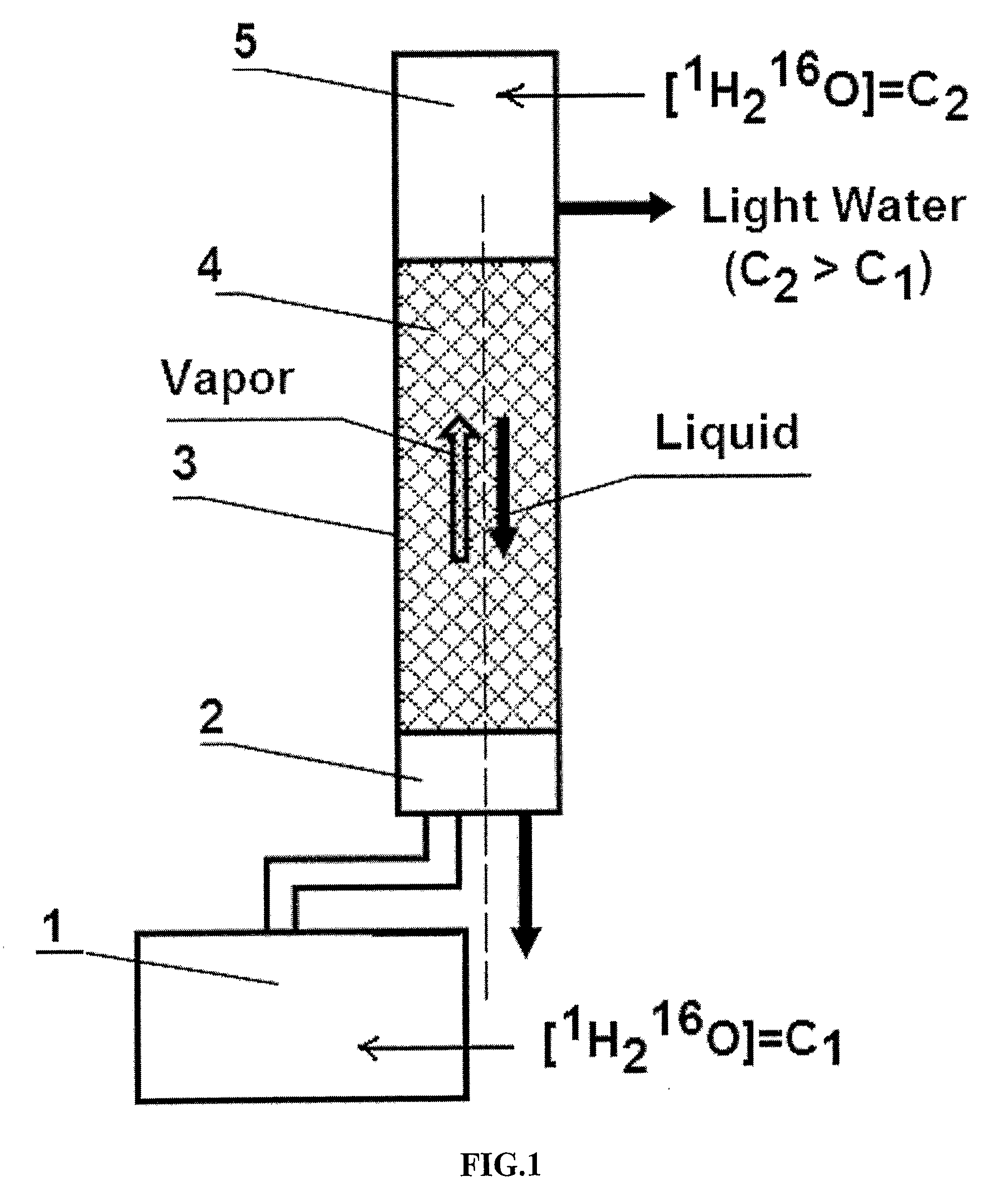
The invention relates to the method of treating metabolic syndrome in a mammal in need thereof comprising a step of administering to said mammal an effective amount of water comprising from 99.76 to 99.99 molecular % of isotopologue 1H216O. Further, invention relates to the medical food for treating metabolic syndrome in a mammal in need thereof which comprises water comprising from 99.76 to about 99.99 molecular % of isotopologue 1H216O. Further, invention relates to the use of water comprising from about 99.76 to about 99.99 molecular % of isotopologue 1H216O for the manufacture of a medical food for treating metabolic syndrome in a mammal in need thereof.
Owner:VADA CONSULTING LTD
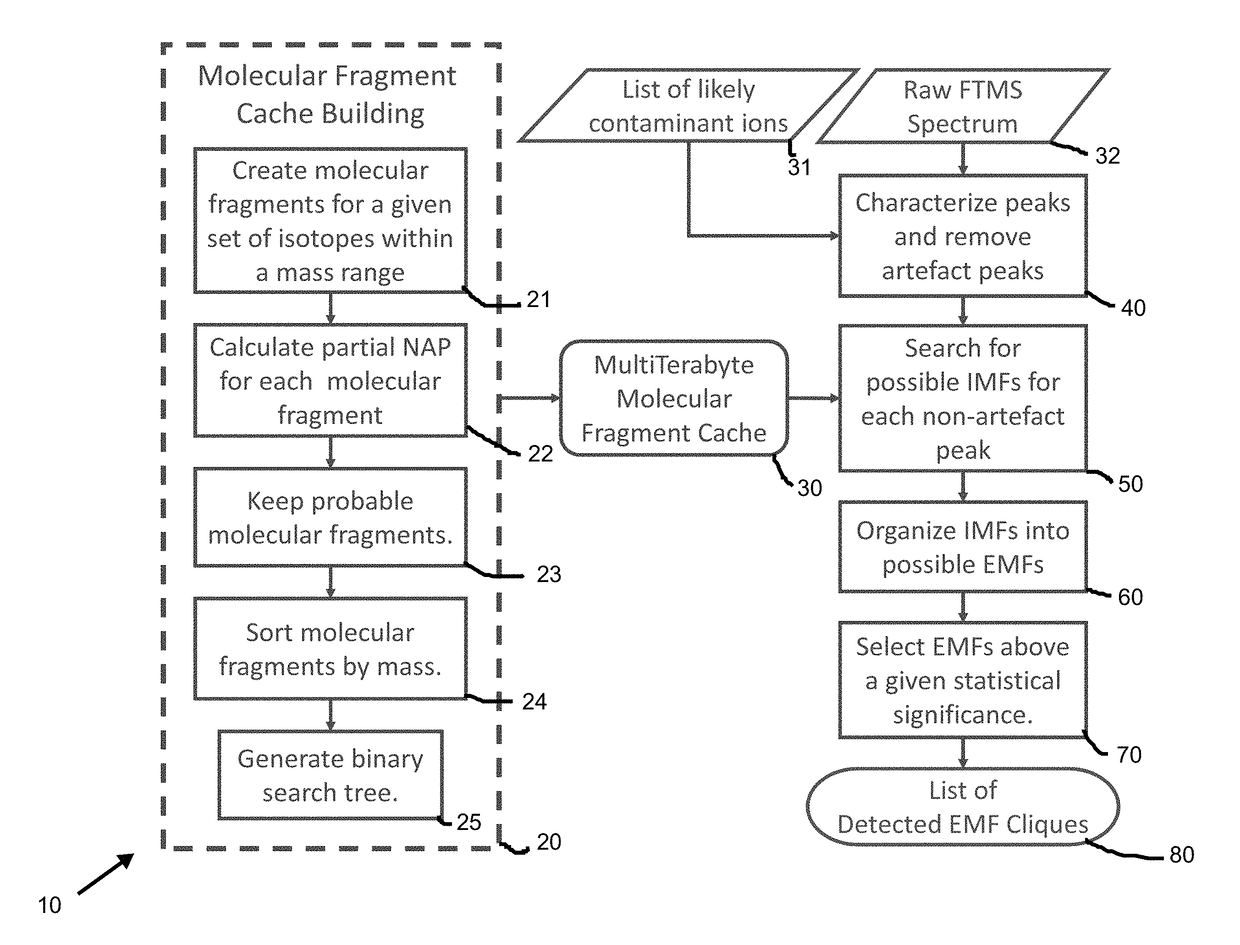
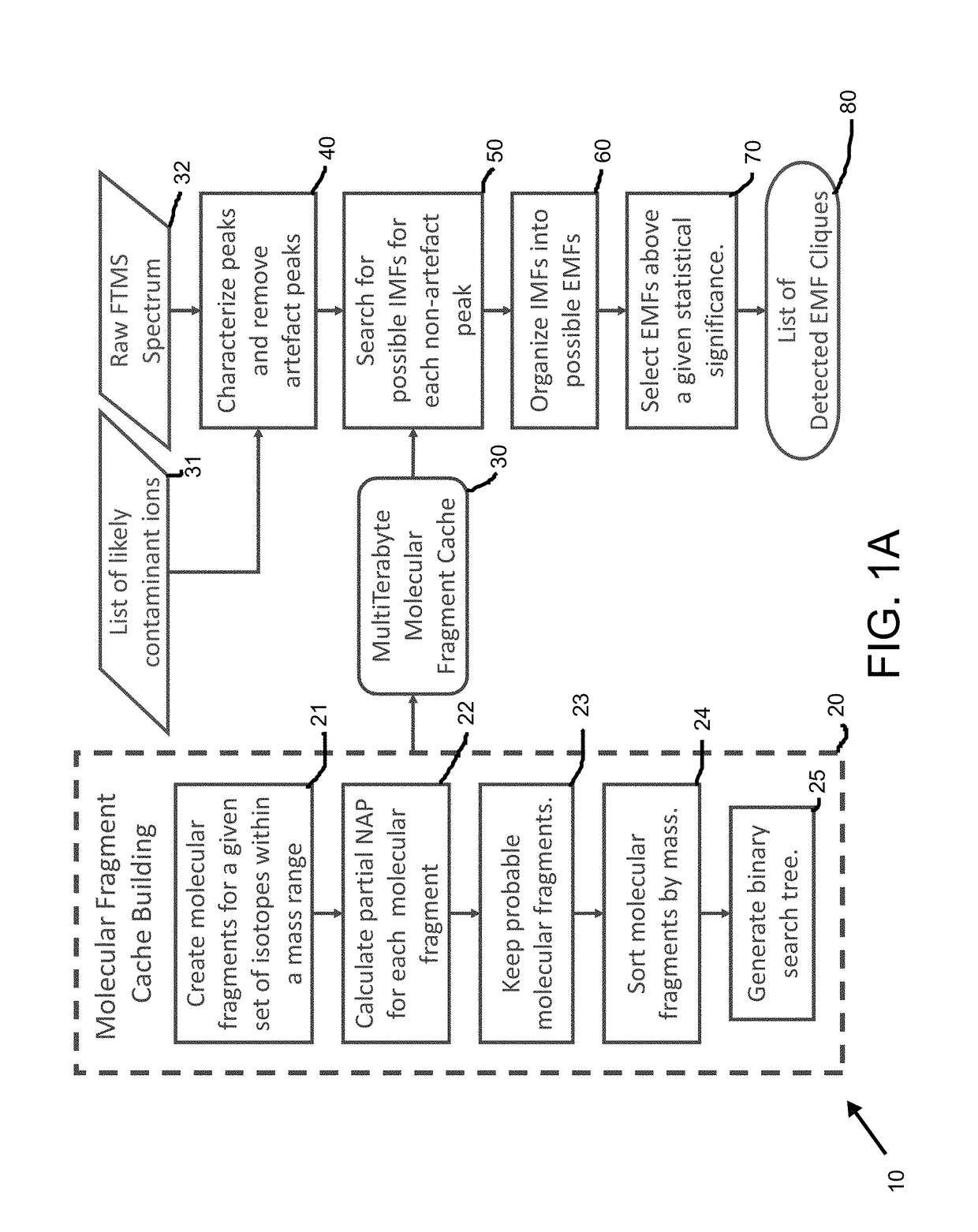
Method and system for identification of metabolites
ActiveUS20180011990A1减少已知的 FTMS 离子抑制效应Less error rateMolecular entity identificationBiostatisticsUltra high resolutionMass spectrum analysis
A method and system is provided for mass spectrometry for identification of a specific elemental formula for an unknown compound which includes but is not limited to a metabolite. The method includes calculating a natural abundance probability (NAP) of a given isotopologue for isotopes of non-labelling elements of an unknown compound. Molecular fragments for a subset of isotopes identified using the NAP are created and sorted into a requisite cache data structure to be subsequently searched. Peaks from raw spectrum data from mass spectrometry for an unknown compound. Sample-specific peaks of the unknown compound from various spectral artifacts in ultra-high resolution Fourier transform mass spectra are separated. A set of possible isotope-resolved molecular formula (IMF) are created by iteratively searching the molecular fragment caches and combining with additional isotopes and then statistically filtering the results based on NAP and mass-to-charge (m / 2) matching probabilities. An unknown compound is identified and its corresponding elemental molecular formula (EMF) from statistically-significant caches of isotopologues with compatible IMFs.
Owner:UNIV OF KENTUCKY RES FOUND
Method of Processing Spectrometric Data
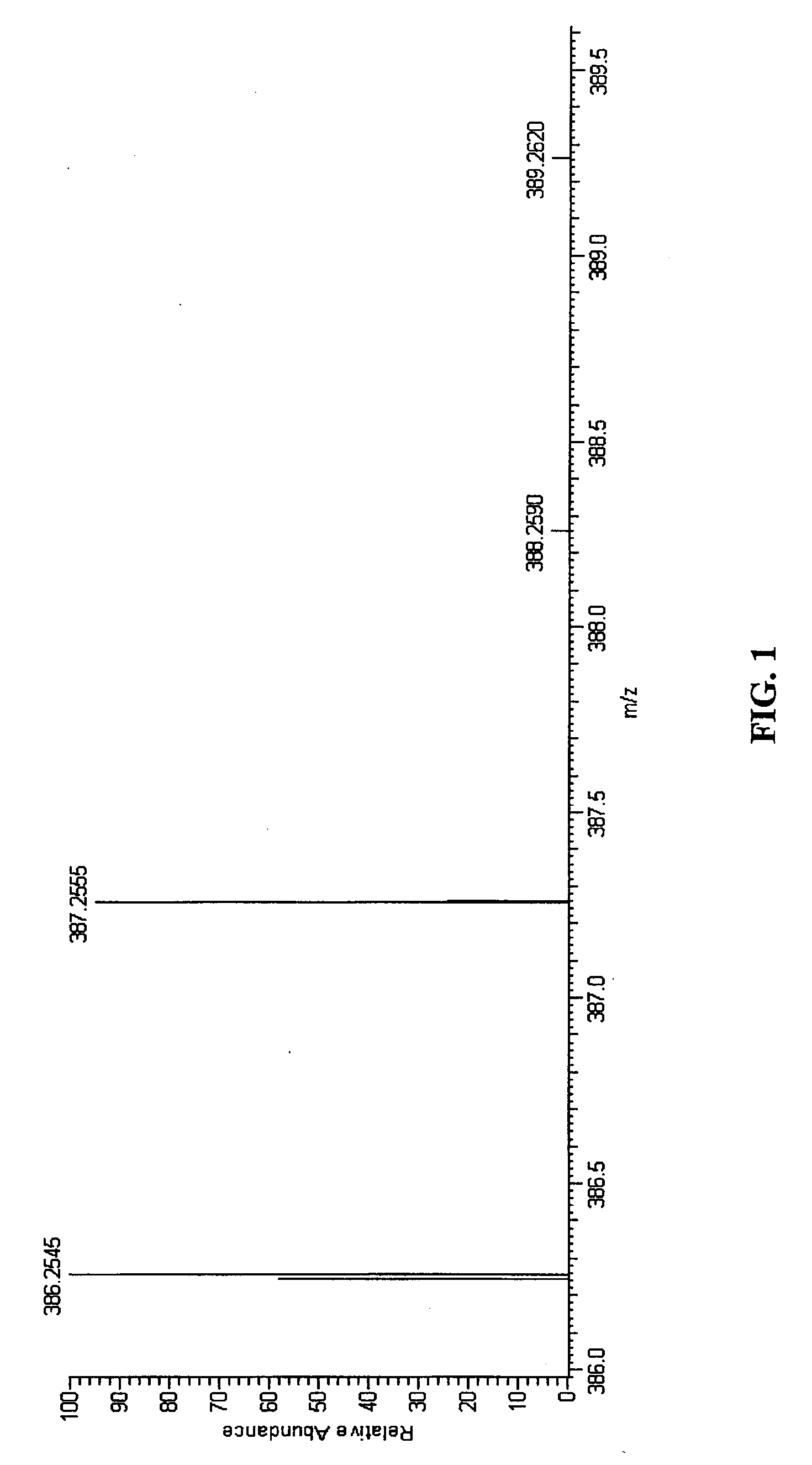
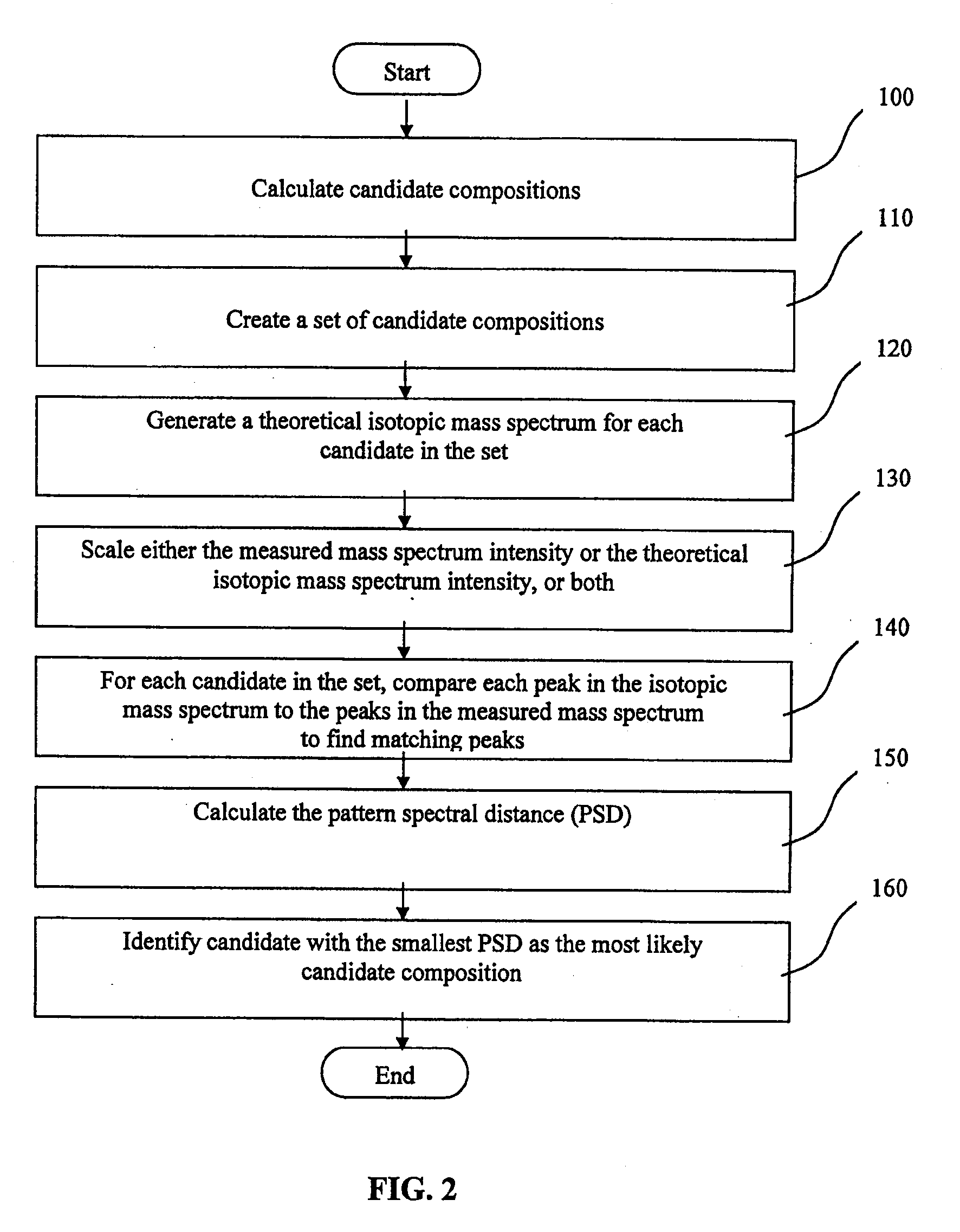
ActiveUS20090299653A1Increase valueReduce the differenceMolecular entity identificationIsotope separationPhysical chemistryMass Spectrometry-Mass Spectrometry
A method of characterising a sample from spectrometric data using calculation of spectral distance values is disclosed, for use in the field of mass spectrometry. Molecular formula assignment of peaks in mass spectral data is difficult and time-consuming, and the invention provides a computer implemented method of finding a most likely elemental composition of a measured spectral peak of interest. The method analyses isotopic peaks in a portion of the spectrum, using both their mass positions and intensities, to determine a spectral distance between those peaks and isotopic peaks of a candidate composition, finding peaks that match (140). A pattern spectral distance is determined (150) to provide a measure of the correspondence between a set of those peaks in the measured spectrum and peaks of each of a number of candidate compositions. The spectral fit is used to determine a most likely candidate composition.
Owner:THERMO FISHER SCI BREMEN
Light Pharmaceutical Water and Therapeutic Compositions and Methods Thereof
InactiveUS20080118463A1Antibacterial agentsOrganic active ingredientsPharmaceutical industryActive agent
This invention relates to pharmaceutical water, which comprises from 99.760 to 99.999% of light isotopologue 1H2 16O and up to 100% of residual isotopologues 1H2 17O, 1H2 18O, 1H2H16O, 1H2H17O, 1H2H 18O, 2H2 16O, 2H2 17O, and 2H2 18O, for the production of finished product, intermediate reagent preparation, and analytical processes in pharmaceutical industry. Further, the invention relates to pharmaceutical compositions comprising a biologically active agent and said pharmaceutical water, wherein said agent is selected from the group consisting of drugs, physiologically active peptides, physiologically active proteins, and nucleic acids. Further, the invention relates to method of administering a biologically active agent to a mammal in need thereof, which method comprises a step of administering to said mammal said therapeutic composition.
Owner:VADA CONSULTING LTD
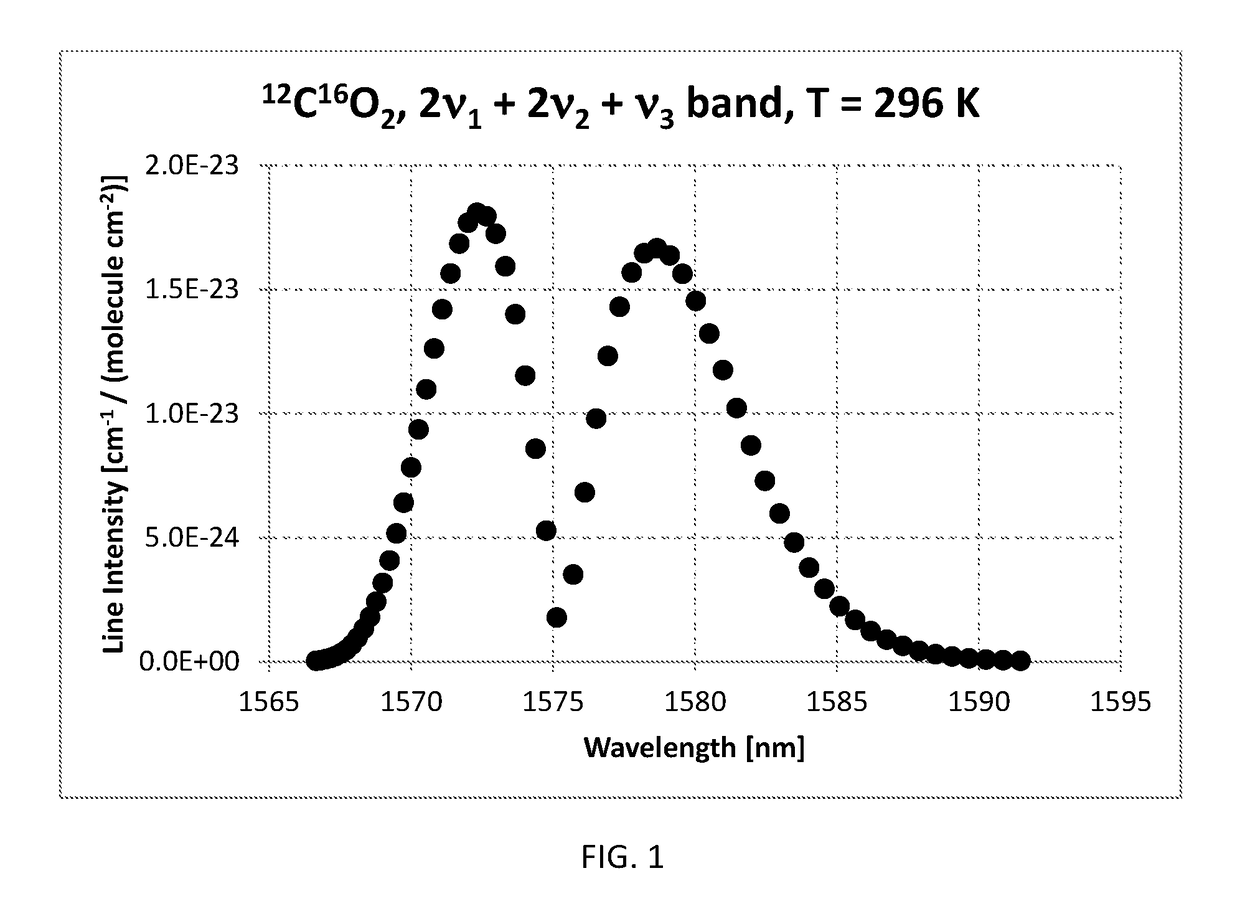
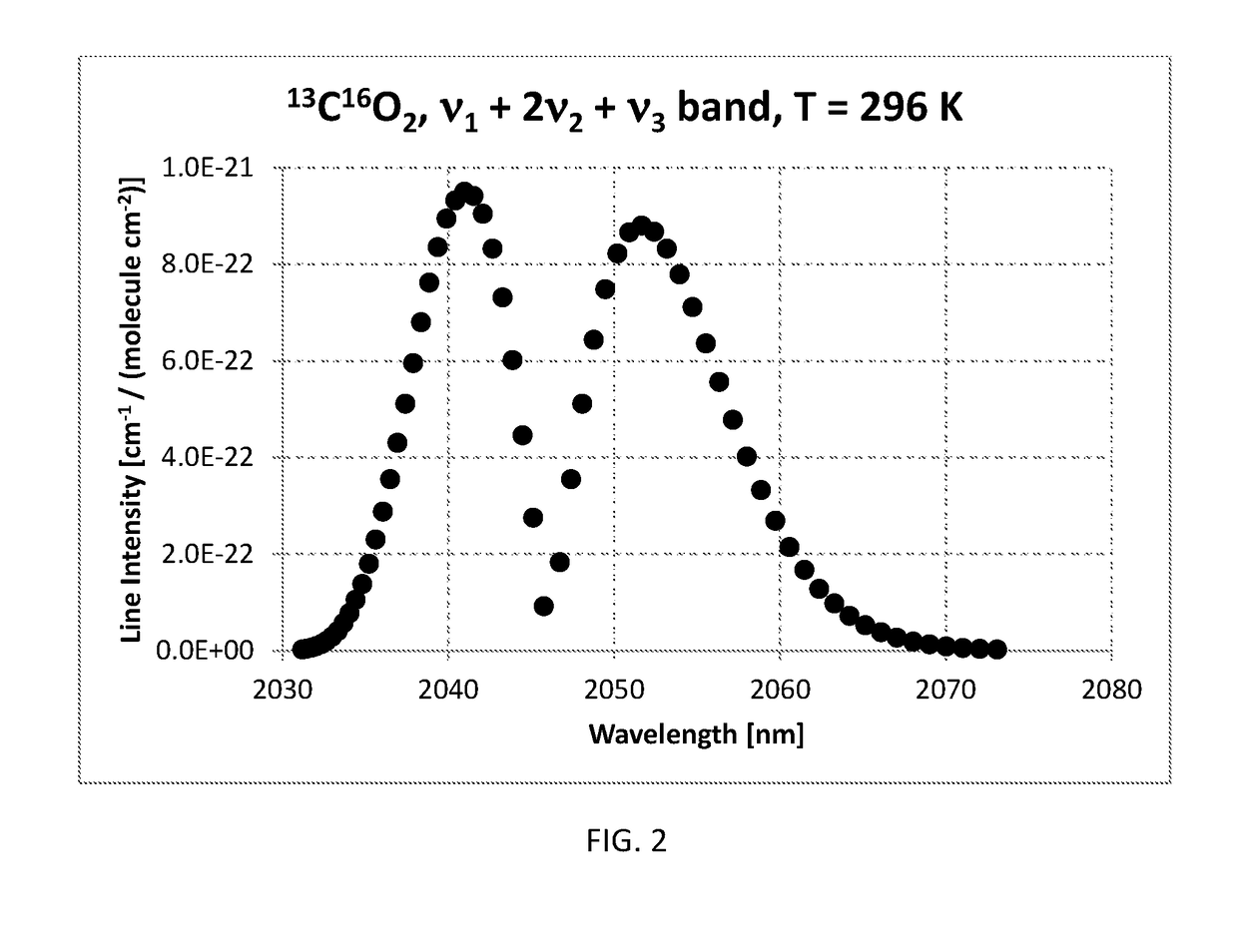
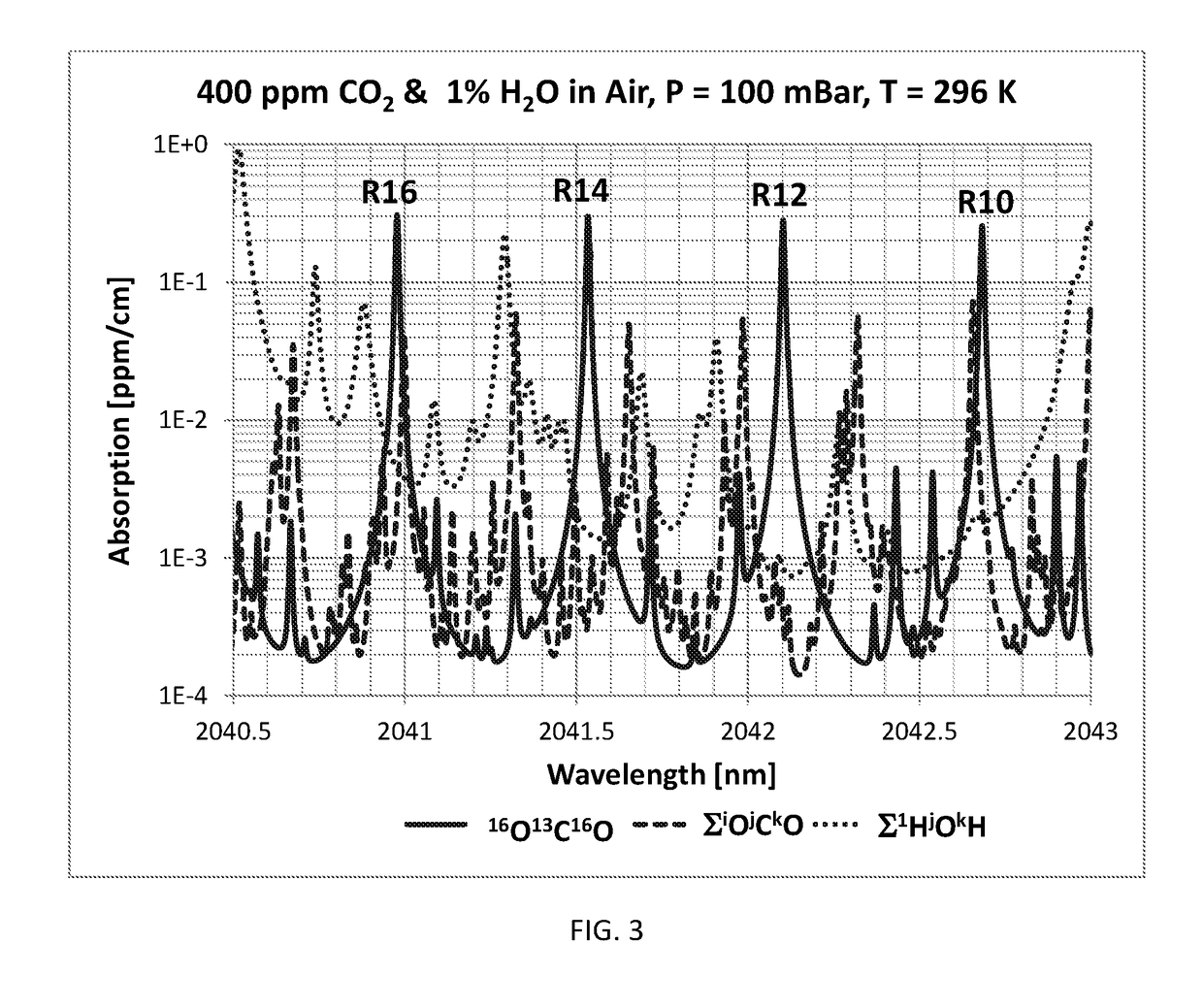
Method of measuring the ratio of isotopologue concentrations in the gas phase
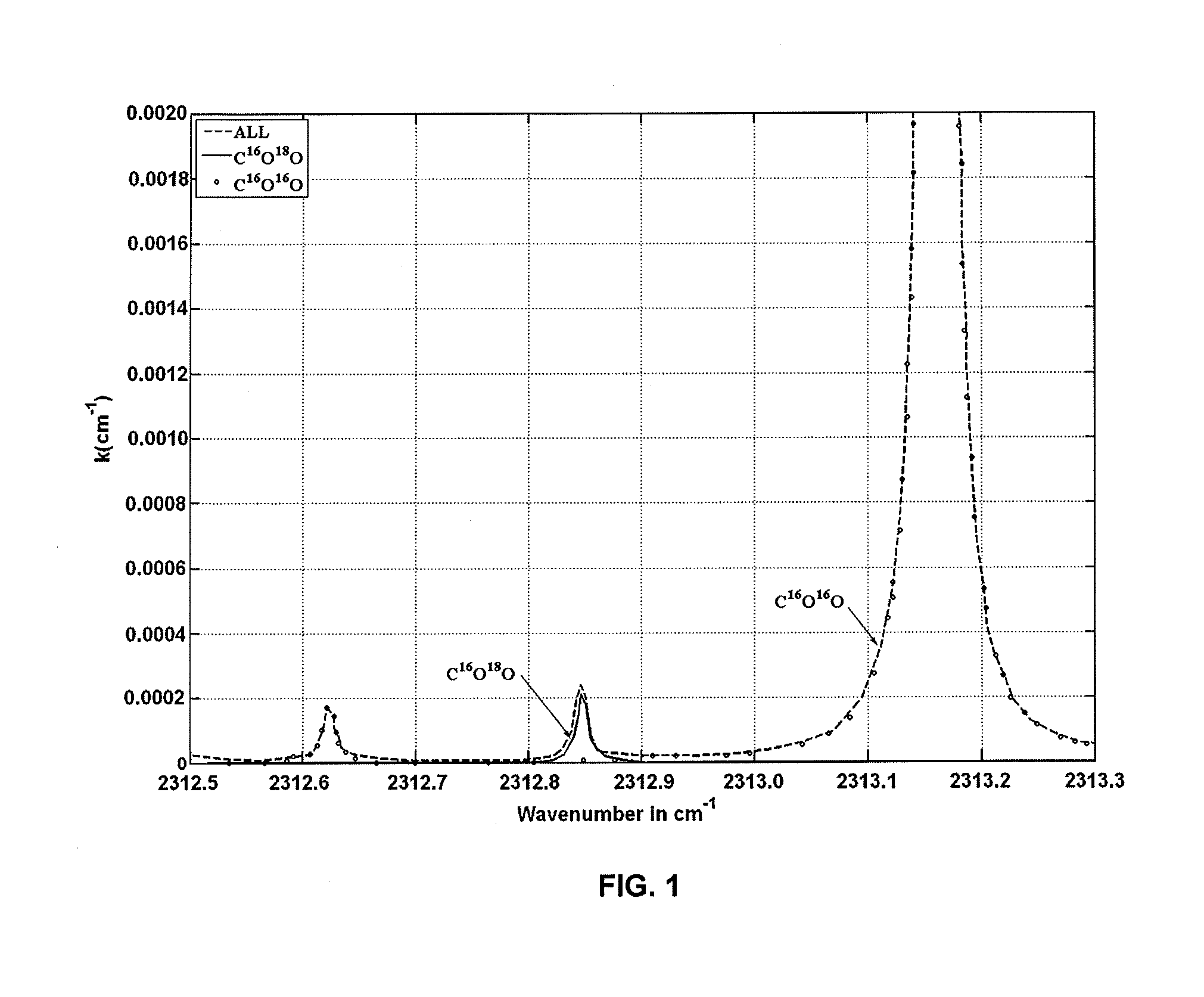
InactiveUS10234381B2Absorption/flicker/reflection spectroscopyColor/spectral properties measurementsChemical speciesGas phase
Method of measuring a ratio of concentrations of two isotopologues of a chemical species in a first gas mixture using an optical absorption spectroscopy based gas analyzer; the method includes measuring the line intensity of a rotational-vibrational line of one isotopologue and the line intensity of another rotational-vibrational line of another isotopologue. The method also includes determining the ratio of two line intensities and comparing it with another ratio measured using another gas analyzer for a gas mixture with known concentration of isotopologues. The second ratio can be measured for the same pair of lines or for different pair of lines. The second ratio can be measured at the same gas temperature or at different gas temperature. The method includes determining a ratio of concentrations of two isotopologues based on two ratios of line intensities and two temperatures.
Owner:KOULIKOV SERGUEI
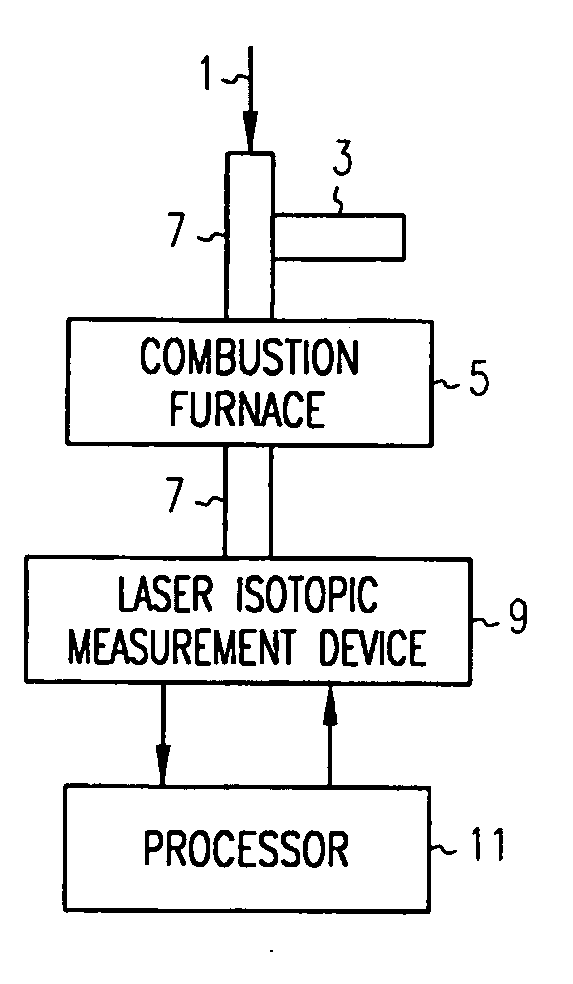
Method and apparatus for measuring isotopic characteristics
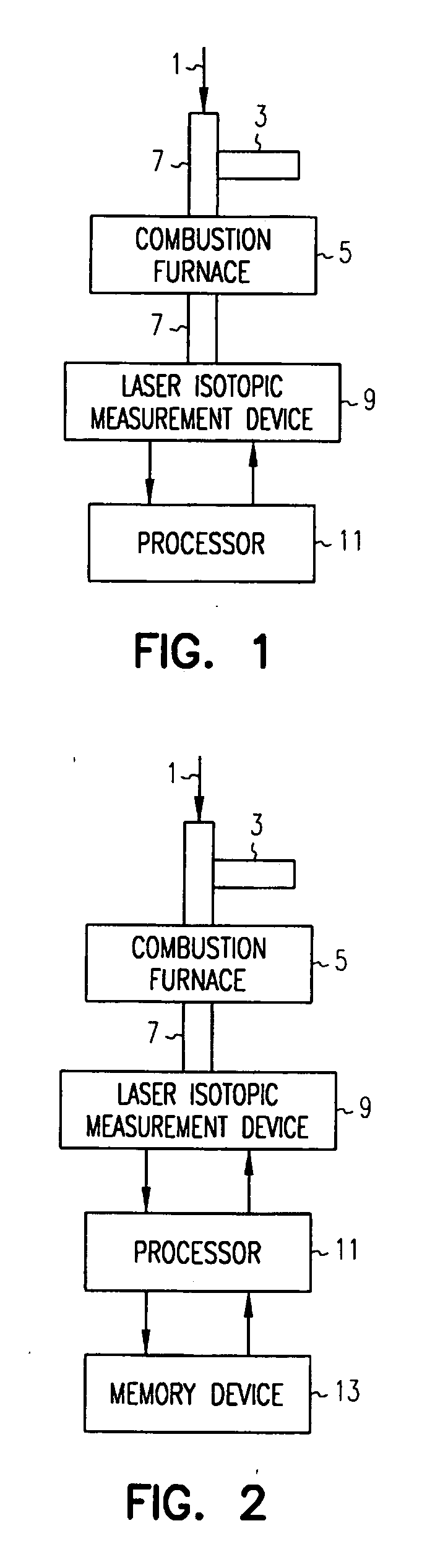
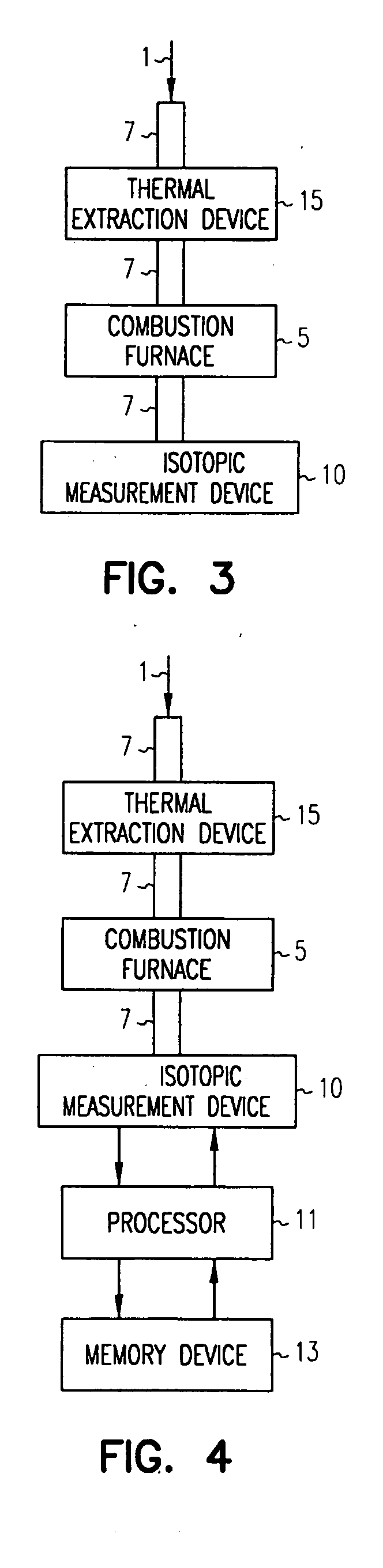
ActiveUS20070178011A1Chemical analysis using combustionMaterial analysis using wave/particle radiationElectricityCombustion
Methods and apparatus are provided to measure isotopic characteristics of a number of sample types. Embodiments of the invention combine novel and existing components to produce more accurate isotopic information. Further, embodiments of the invention allow for isotopic readings to be taken and analyzed outside of a laboratory. An example of such an embodiment is an apparatus comprising a combustion furnace; a reactant tube passing through the combustion furnace; an injector coupled to one, or a combination of, the combustion furnace, and reactant tube, to introduce a sample; a laser isotopic measurement device coupled to the reactant tube on the exit end; and a processor electrically coupled to one, or a combination of, the injector, the combustion furnace, the reactant tube, and the isotopic measurement device, in which a carrier gas transports the sample through the apparatus.
Owner:HALLIBURTON ENERGY SERVICES INC +1
Method of selective peptide isolation for the identification and quantitative analysis of proteins in complex mixtures
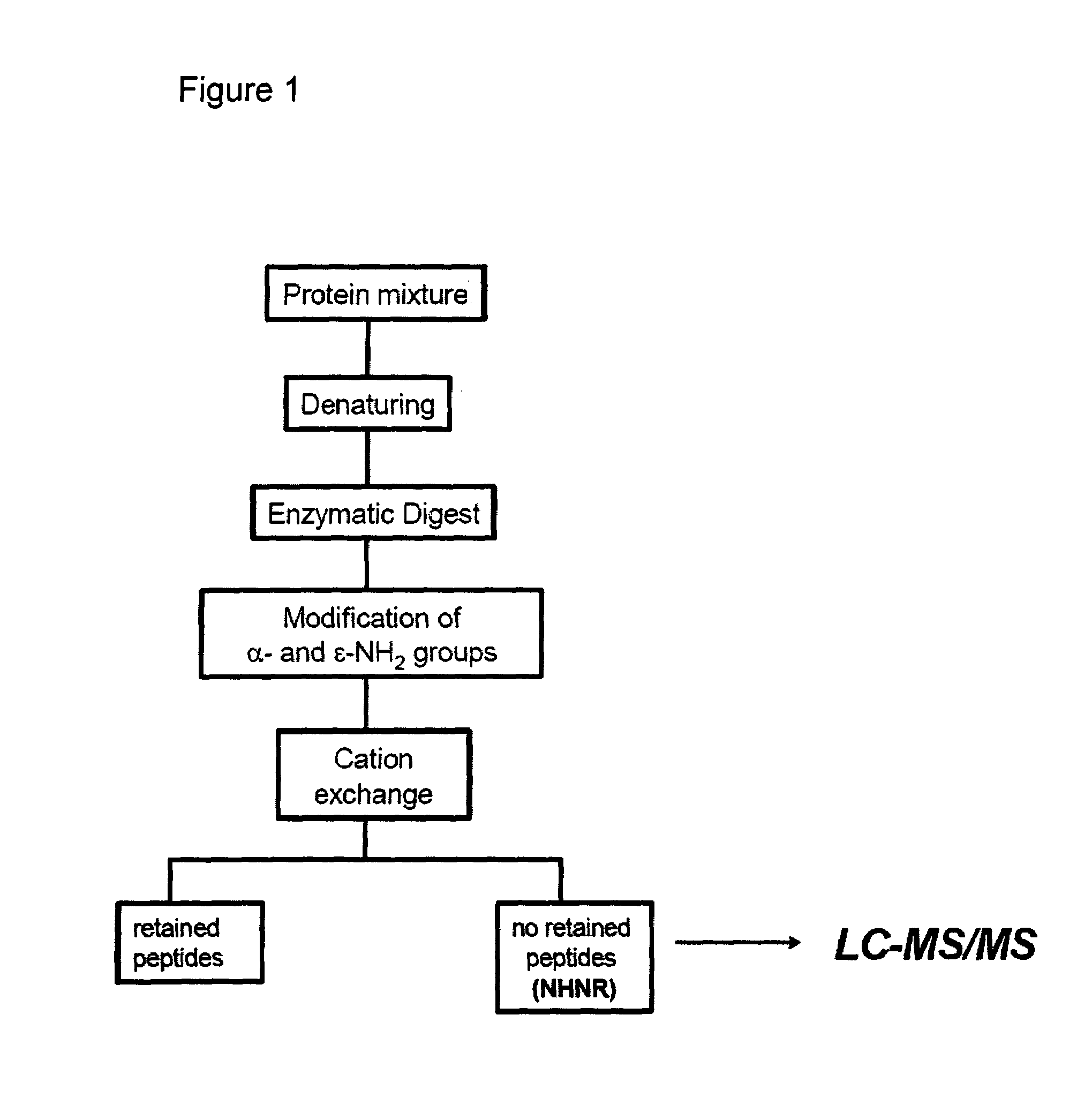
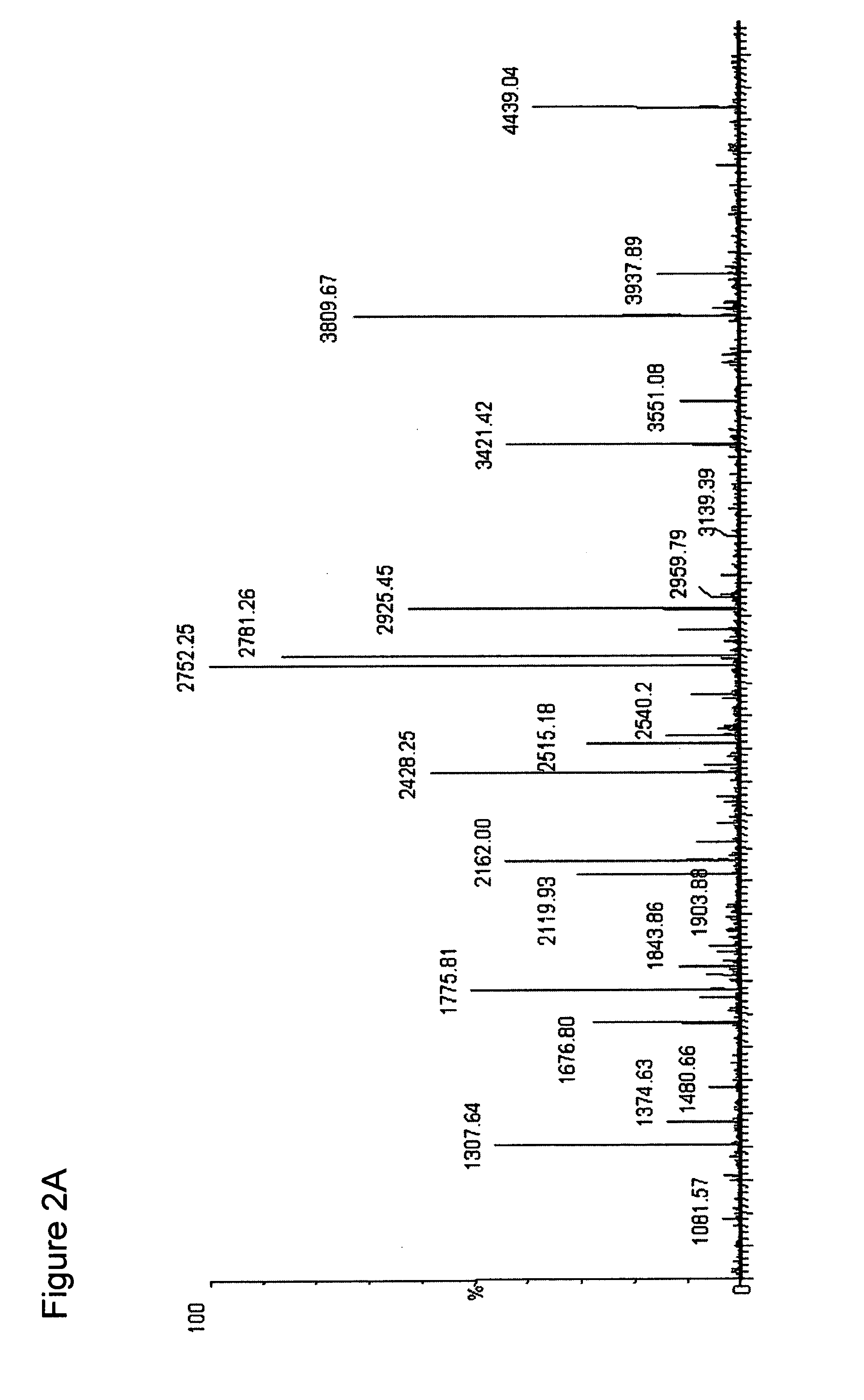
InactiveUS7244411B2Efficient identificationWidely distributedIn-vivo radioactive preparationsComponent separationProtein insertionArginine
The present invention describes a method of selective peptide isolation for the identification and quantitative analysis of proteins in complex mixture. The method comprises the selective isolation from every protein of those peptides that neither contain arginine nor histidine (NHNR peptides), and the determination of the relative concentrations of one or several proteins in different samples from the ratio between the areas of the estimated theoretical spectra for the NHNR peptides labeled with different isotopes in each sample. The determination of the relative concentration of proteins is valid for any type of isotopic label of the NHNR peptides. The method avoids the separation and purification of the proteins present in a complex mixture, and the analysis of all peptides generated from the enzymatic digest of the samples. The method is applicable to the identification of proteins with vacunal, therapeutic and diagnostic aims.
Owner:CENT DE ING GENETICA & BIOTECNOLOGIA
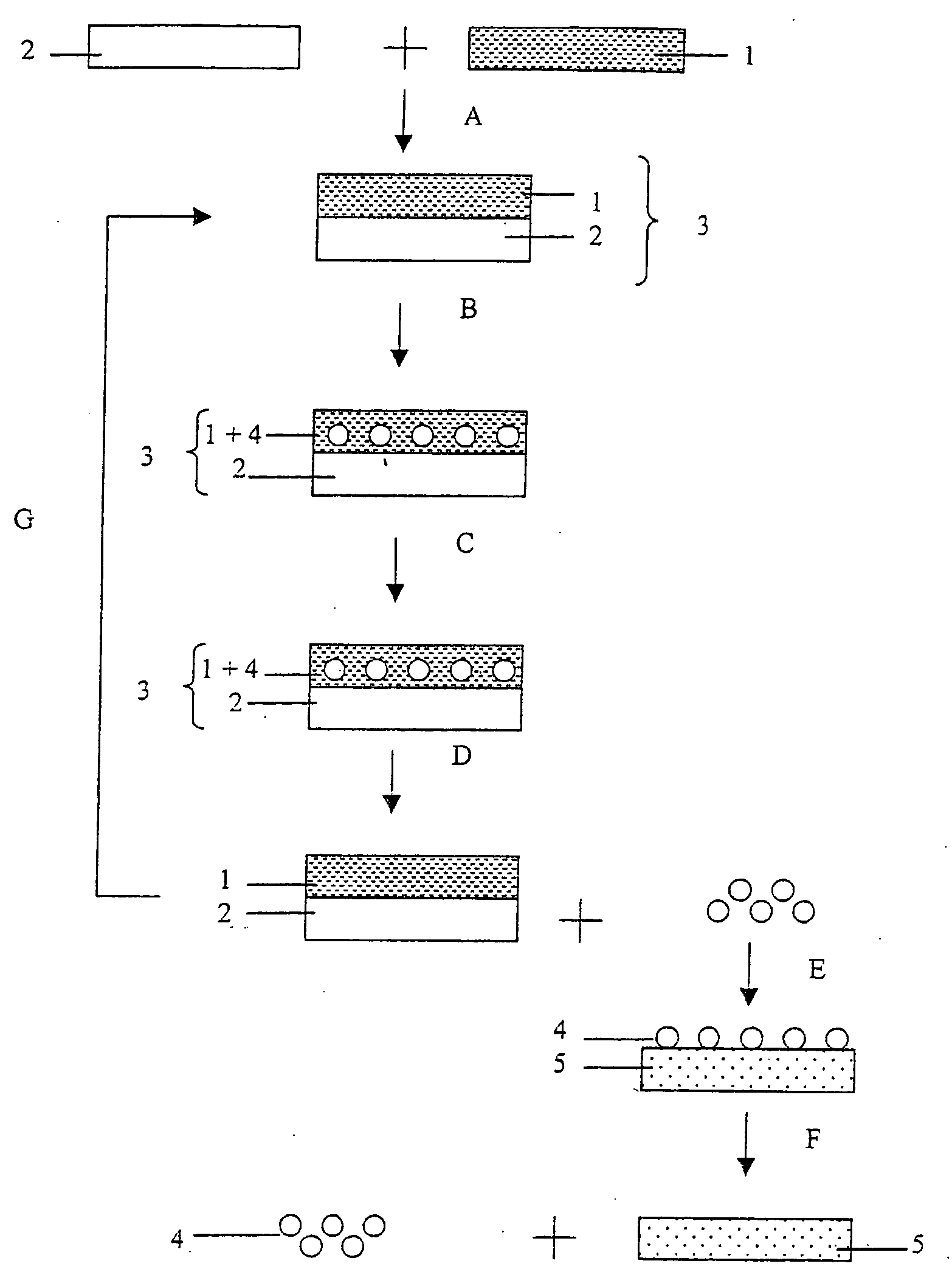
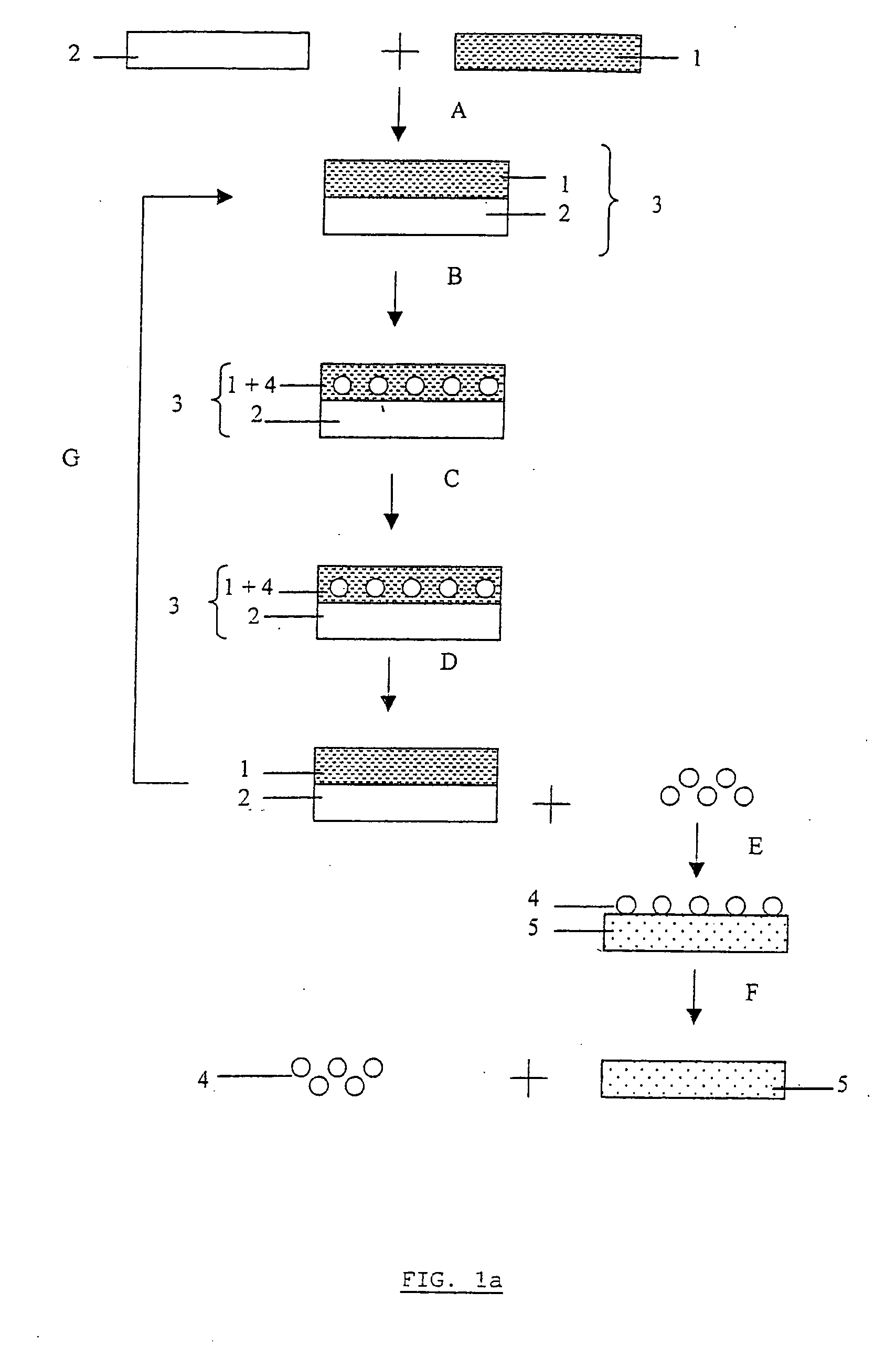
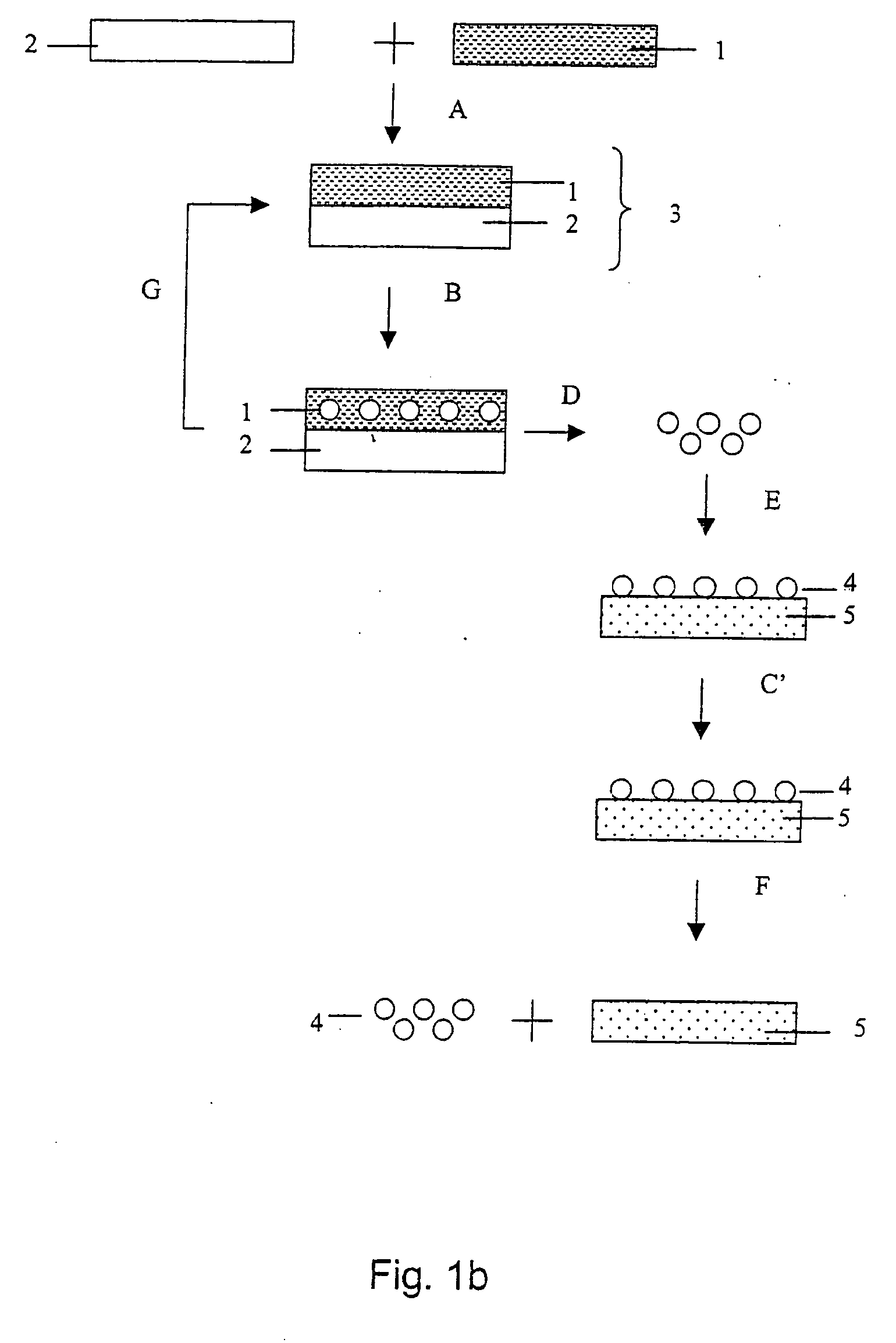
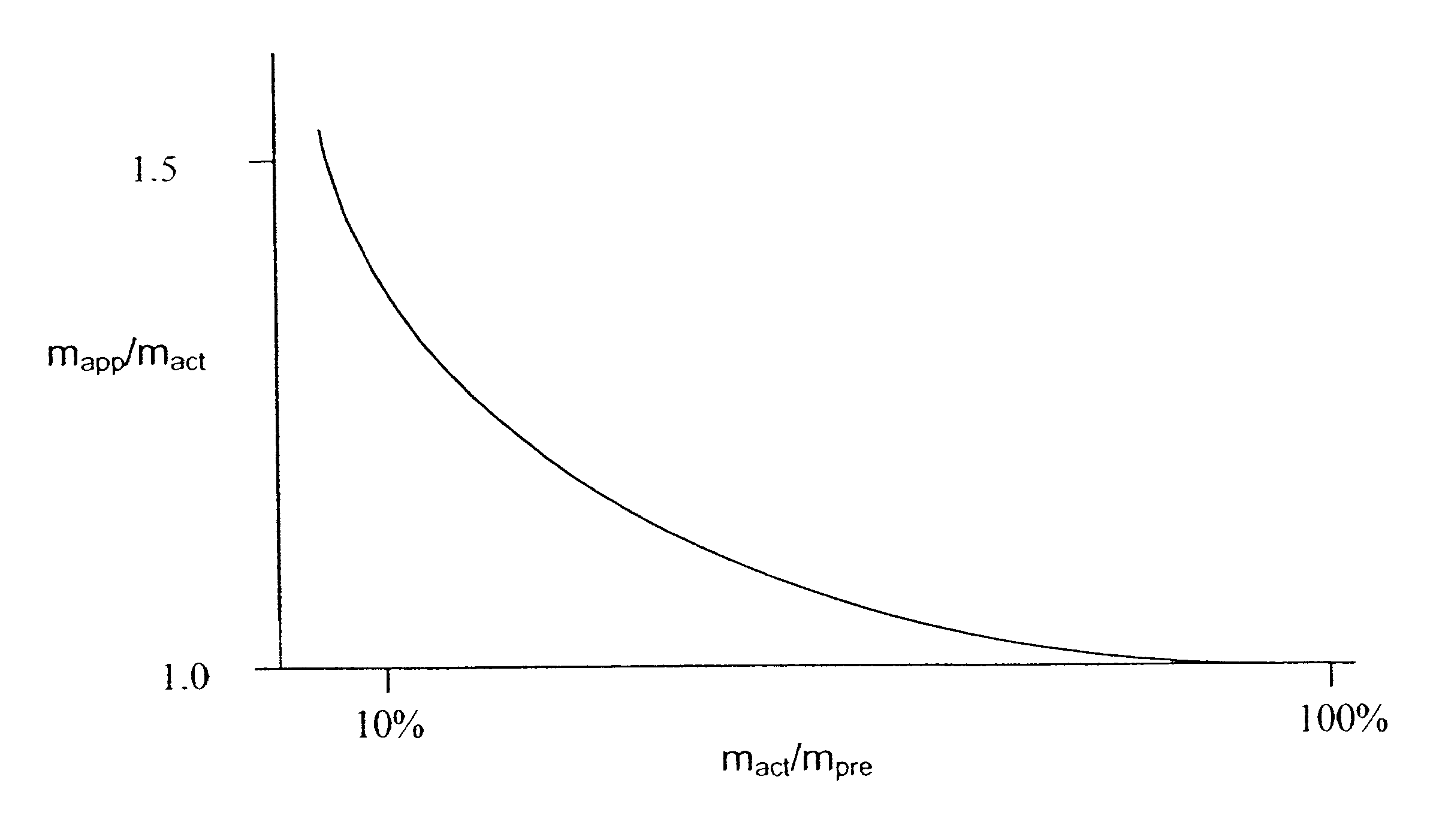
Methods, devices, and systems for the separation and concentration of isotopologues
Owner:EXELON GENERATION CO LLC
Nutrients solutions for enhancement of cognitive function
The present invention relates to dietary supplements, medical foods, or pharmaceutical compositions for use in the method of enhancement of cognitive function in healthy subjects and / or subjects person suffering from a cognitive deficit, comprising aqueous solution of a nutrient, wherein solution comprises a water having content of deuterium from 90 to about 135 ppm and 1H216O isotopologue up to 99.759%. In one embodiment of the invention relates to a succinate salt of choline as the nutrient.
Owner:MITOCHONDRIAL SUBSTRATE INVENTION
Stable isotopic biomarker measurement for the detection of cancer and the determination of efficacy of treatment in diagnosed cancer patients
InactiveUS20130011872A1Bioreactor/fermenter combinationsBiological substance pretreatmentsInfraredCancer cell
The present invention relates to a method of detecting the presence of cancer cells in an animal such as a human or other mammal, comprising administering a measured volume of air to the subject wherein 16O2 and 18O2 are present and then measuring the δ18O in the subject's exhaled CO2. A Photoacoustic Spectrometer system is used for measuring the amount of laser light (Infrared) absorbed by isotopes of CO2(C16O2, C18O2) in the exhalant.
Owner:GABRIEL PHILIP M
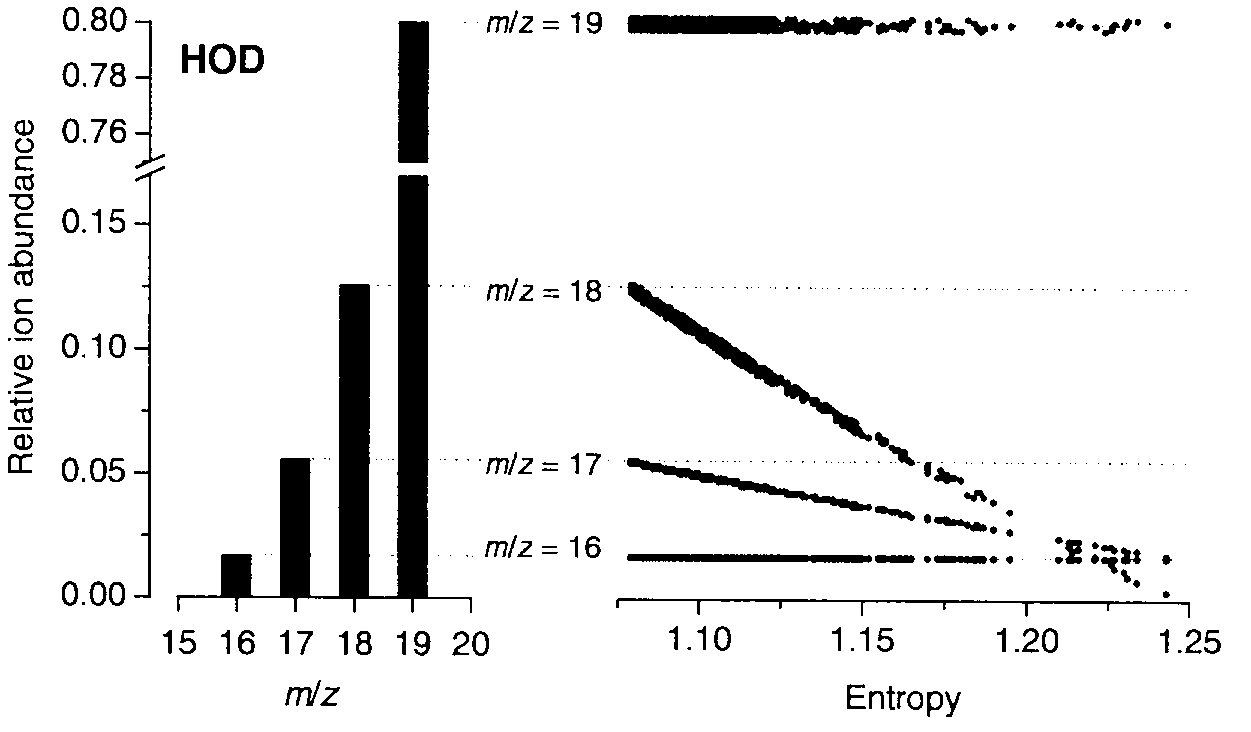
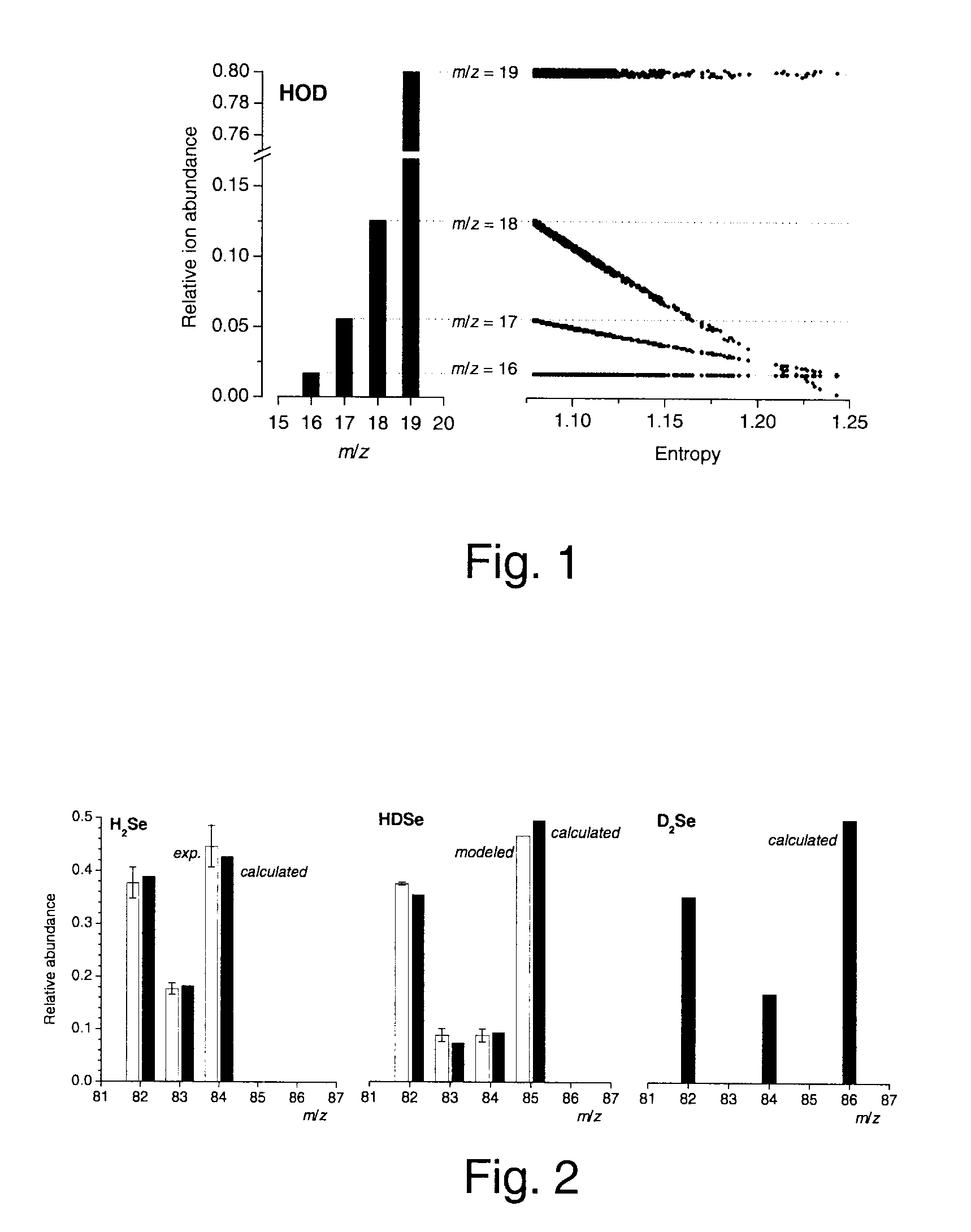
Blind extraction of pure component mass spectra from overlapping mass spectrometric peaks
InactiveUS7804062B2Increasing the instrumental duty cycleComponent separationIsotope separationConfocalHigh pressure
A method of obtaining pure component mass spectra or pure peak elution profiles from mass spectra of a mixture of components involves estimating number of components in the mixture, filtering noise, and extracting individual component mass spectra or pure peak elution profiles using blind entropy minimization with direct optimization (e.g. downhill simplex minimization). The method may be applied to deconvolution of pure GC / MS spectra of overlapping or partially overlapping isotopologues or other compounds, separation of overlapping or partially overlapping compounds in proteomics or metabolomics mass spectrometry applications, peptide sequencing using high voltage fragmentation followed by deconvolution of the obtained mixture mass spectra, deconvolution of MALDI mass spectra in the separation of multiple components present in a single solution, and specific compound monitoring in security and / or environmentally sensitive areas.
Owner:NAT RES COUNCIL OF CANADA
System for generation of useful electrical energy from isotopic electron emission
InactiveUS20100062288A1Safe and cost-effectiveTransit-tube cathodesCell component detailsHigh energyIsotope
Beta and alpha-ray particles emitted by radio-isotopic by-products of nuclear fission, such as nickel 63, are used as a power source at the cathode of a microwave generating magnetron. Such particles include high speed, high energy electrons having a large EMF associated therewith. In the magnetron, a radial electrical vector, between the cathode and anode, interacts with an axial magnetic vector to produce a cloud of electrons that rotates about the magnetron axis. The speed, geometry and density of the rotating cloud may be modulated by an external RF input or grids within the interaction space of the magnetron. At the periphery of the interaction space is a polar array of anode cavities into which the rotating field induces an LC equivalent parameter that includes high energy microwaves that may be used as an input for the generation of AC or DC power.
Owner:WEBER DAVID +1
Treatment of a hematologic malignancy with 2-(4-chlorophenyl)-n-((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide
Owner:CELGENE CORP
Test kit for the quantitative determination of narcotic drugs
A test kit for the quantitative determination of narcotic drugs comprising (A) series of sealed vessels, each vessel containing a deuterium free isotopologue of a narcotic drug in exactly defined concentrations and quantities, wherein the isotopologue differs from vessel to vessel and—wherein the quantities of the isotopologue differ from vessel to vessel or are the same for all vessels; and / or (B) series of sealed vessels, each vessel containing in exactly defined concentrations and quantities the same isotopologue in quantities which differ from vessel to vessel; wherein the free isotopologues are selected from narcotic drugs; prodrugs, salts, solvates, hydrates and polymorphs and contain at least three stable isotopes selected from the group consisting of 13C, 15N and 18O in the molecule with a degree of labeling of at least 95 mol-%; the use of the test kit and a method for quantitatively determining narcotic drugs.
Owner:CHIRON CORP
Process for recovery of water isotopologues from impure water
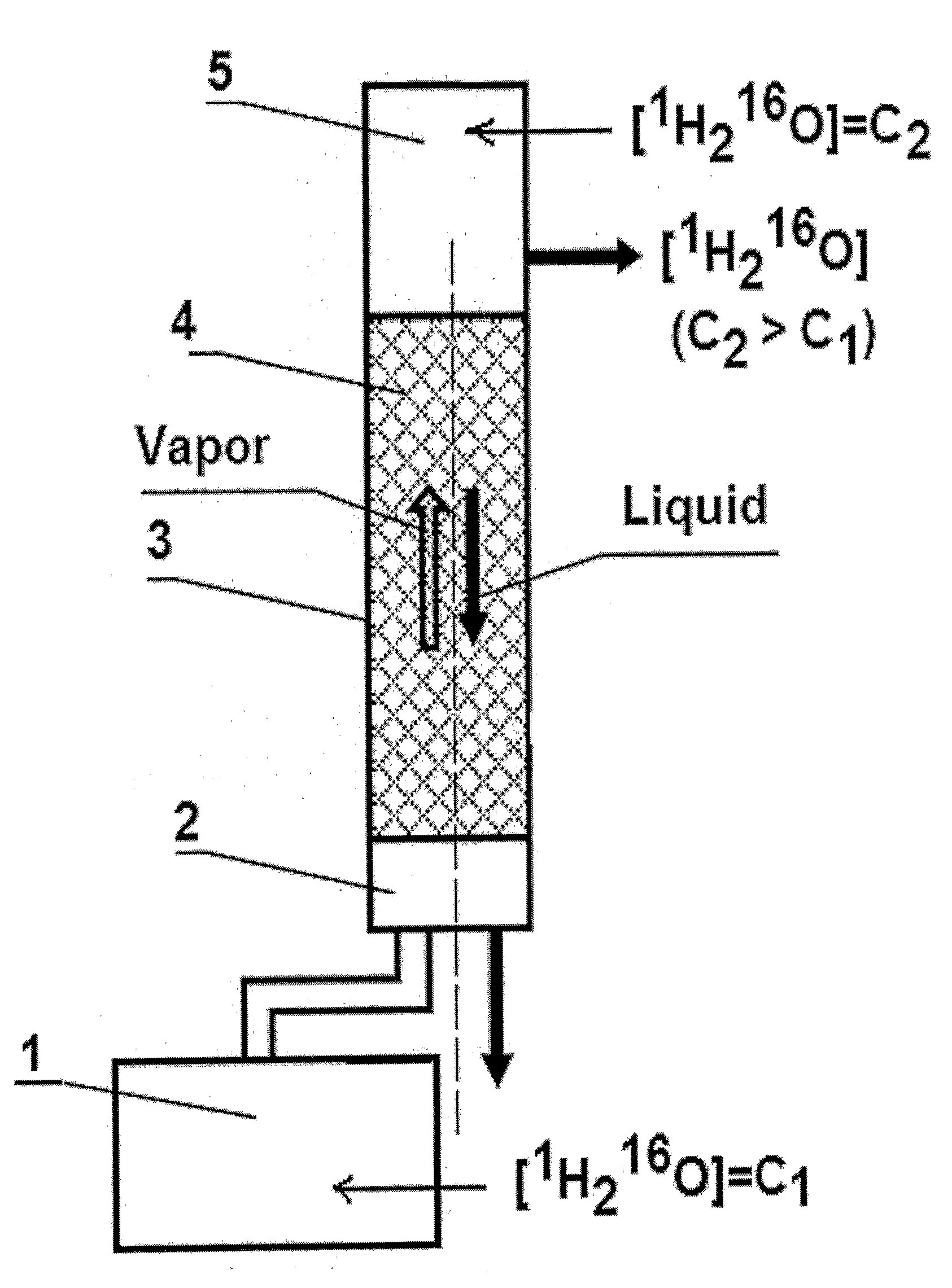
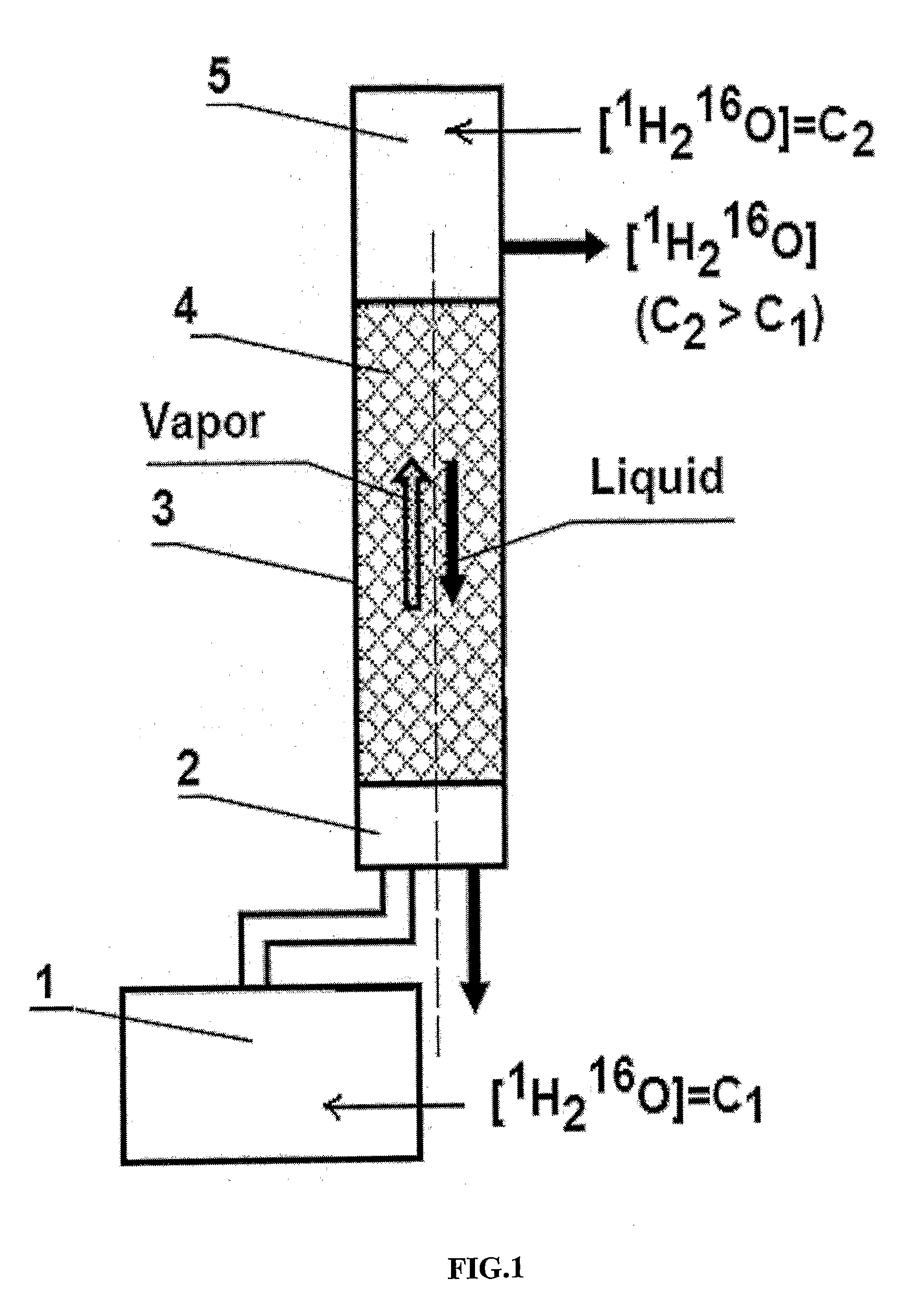
InactiveUS20100021372A1Simpler and effective processEfficient strippingIsotope separationHeavy waterImpurityChemistry
Owner:GE HEALTHCARE LTD
Particle violation spectroscopy
InactiveUS20080173825A1Material analysis using wave/particle radiationMaterial analysis by optical meansBeam splitterSurface barrier
The method shows how to violate predictions of quantum mechanics for matter. For light, the method has been disclosed in patent application Photon Violation Spectroscopy. The methods are different in specifying different methods for light and matter. For matter, the method typically uses the single 5.5 MeV alpha (He++) emitted from the radioisotope Americium-241 in spontaneous decay, a thin gold foil beam splitter, and two surface barrier alpha detectors. The detectors deliver a characteristic electrical pulse with amplitude proportional to matter wave energy. A circuit reads the coincidence rate and singles rates of pulses from the two detectors. Quantum mechanics predicts that the particle would go one way or the other at the beam splitter, and coincident detections of pulses characteristic of such a particle would occur only at an easily calculated chance rate. However, the method at hand shows such characteristic pulses occur in coincidence at a rate greatly exceeding chance. By exceeding chance the method demonstrates surpassing a binding energy threshold and predictions of quantum mechanics. The degree above chance is a new measure in fundamental physics and is usable as a material science probe of the beam splitter. An apparatus specially designed to test for the absence of true coincidences and then perform a beam splitting test to show split-beam coincidences becomes useful in applying the method of Particle Violation Spectroscopy to material science. Fundamental discoveries in physics have been made with this method; therefore it is a method of discovery in physics.
Owner:REITER ERIC STANLEY



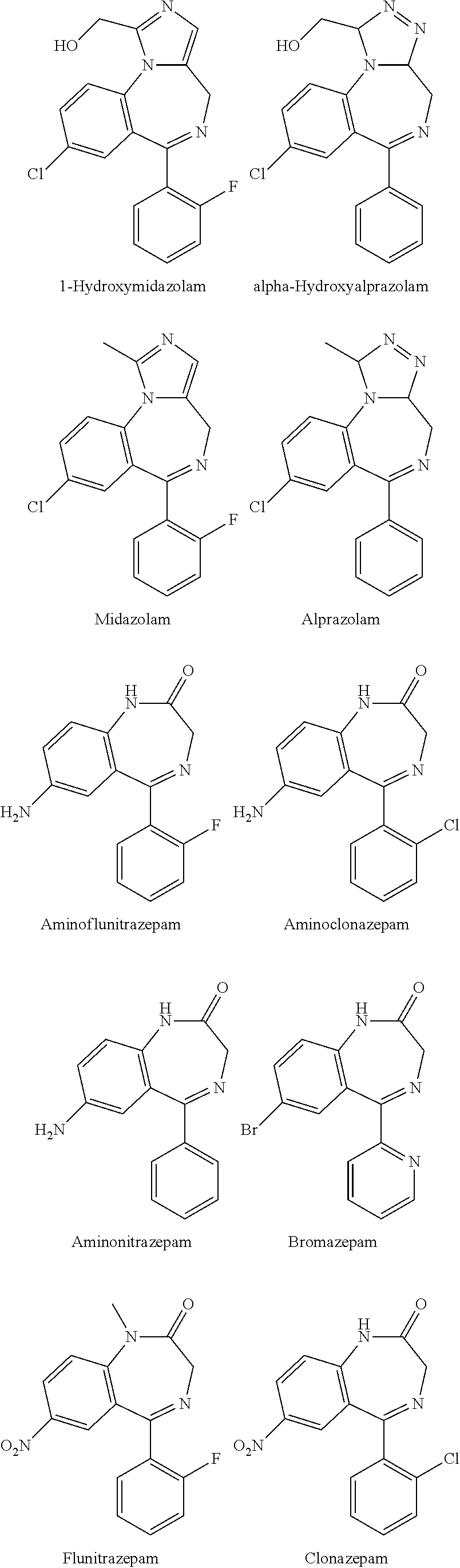
![Novel benzo[D][1,3]-dioxol derivatives Novel benzo[D][1,3]-dioxol derivatives](https://images-eureka.patsnap.com/patent_img/2501ca05-719b-4869-8497-d864d4741eae/US20070191432A1-20070816-C00001.png)
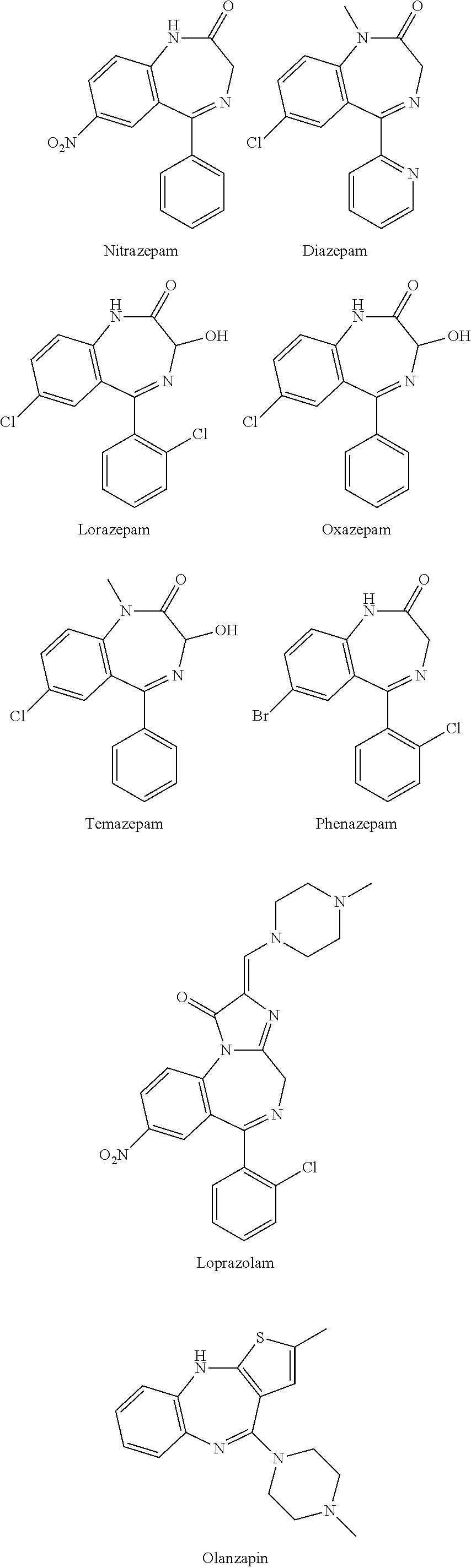
![Novel benzo[D][1,3]-dioxol derivatives Novel benzo[D][1,3]-dioxol derivatives](https://images-eureka.patsnap.com/patent_img/2501ca05-719b-4869-8497-d864d4741eae/US20070191432A1-20070816-C00002.png)
![Novel benzo[D][1,3]-dioxol derivatives Novel benzo[D][1,3]-dioxol derivatives](https://images-eureka.patsnap.com/patent_img/2501ca05-719b-4869-8497-d864d4741eae/US20070191432A1-20070816-C00003.png)